
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 78
CARBARYL
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1992
This is a companion volume to Environmental Health Criteria
153: Carbaryl
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the
United Nations Environment Programme, the International Labour
Organisation, and the World Health Organization)
This report contains the collective views of an international group
of experts and does not necessarily represent the decisions or the
stated policy of the United Nations Environment Programme, the
International Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Carbaryl : health and safety guide.
(Health and safety guide ; no. 78)
1.Carbaryl - standards 2.Carbaryl - toxicity 3.Environmental exposure
4.Hazardous substances - standards I.Series
ISBN 92 4 151078 1 (NLM Classification: WA 240)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1993
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Environmental transport, distribution, and
transformation
2.2. Environmental levels and human exposure
2.3. Kinetics and metabolism
2.4. Effects on organisms in the environment
2.5. Effects on experimental animals and in vitro
test systems
2.5.1. Reproduction effects
2.5.2. Mutagenicity
2.5.3. Carcinogenicity
2.5.4. Effects on different organs and systems
2.5.5. Primary mechanism of toxicity
2.6. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.1.1. General population
3.1.2. Subpopulations at high risk
3.1.3. Occupational exposure
3.1.4. Environmental effects
3.2. Recommendations
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION,
EMERGENCY ACTION
4.1. Human health hazards, prevention and
protection, first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 edical treatment
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
6.1. Previous evaluations by international bodies
6.2. Exposure limit values
6.3. Specific restrictions
6.4. Labelling, packaging, and transport
6.5. Waste disposal
BIBLIOGRAPHY
ANNEX. Treatment of carbamate poisoning in man
INTRODUCTION
The Environmental Health Criteria (EHC) monographs produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding
EHC. Section 4 includes advice on preventive and protective measures
and emergency action; health workers should be thoroughly familiar
with the medical information to ensure that they can act efficiently
in an emergency. The section on regulatory information has been
extracted from the legal file of the International Register of
Potentially Toxic Chemicals (IRPTC) and from other United Nations
sources.
The target readership includes occupational health services, those
in ministries, governmental agencies, industry, and trade unions who
are involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: Carbaryl
Chemical structure:
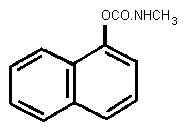 Molecular formula: C12H11NO2
Common name: Carbaryl (BSI)
CAS chemical name: 1-naphthalenyl methylcarbamate
IUPAC chemical name: 1-naphthyl methylcarbamate
Trade names: Arylat, Arylam, Atoxan, Bercema,
Carbaryl, Carbamate, Carpolin,
Carbotox, Denapon, Dicarbam, Hexavin,
Monsur, Murrin, Panam, Pamex,
Prosevor, Rayvon, Sevimol, Sevin,
Viaxon, etc.
CAS registry number: 63-25-2
RTECS registry number: FC5950000
Relative molecular mass: 201
Conversion factor: 1 mg/m3=8.22 ppm
1 ppm=0.12 mg/m3 at 35 °C
1.2 Physical and chemical properties
Carbaryl is an odourless, white, crystalline solid, with a low
volatility. It is practically insoluble in water, but soluble in
most organic solvents, stable to light and heat up to 70 °C, and
easily hydrolysed by alkaline materials. It is non-corrosive.
The technical product is principally manufactured in the USA;
however, small amounts are produced in other parts of the world.
The technical product, manufactured in the USA, is produced to a
minimum purity of 99% w/w carbaryl with a specified content of the
impurity, 2-naphthyl carbamate isomer (sometimes known as
beta-carbaryl), of <0.05% w/w.
The Food and Agriculture Organization of the United Nations (FAO)
specifies a minimum purity of 98% w/w carbaryl with a specified w/w
content of the impurity, 2-naphthyl carbamate isomer, of <0.05%.
Some physical properties of carbaryl are given in Table 1.
Table 1. Physical properties
Melting point (°C) 142
Boiling point (°C) decomposing
Solubility in water (30 °C) 40 mg/litre
Specific density (20 °C) 1.23
Relative vapour density -
Vapour pressure 1.17 x 10-6-3.1 x 10-7 mmHg at
24-25 °C
Flash point 193 °C
Octanol/water partition
coefficient (log Kow) 1.59-2.3
Flammability (explosive)
limits -
Relative molecular mass 201
1.3 Analytical methods
Product analysis can be carried out using infrared spectroscopy.
Residues can be determined by GLC and TLC. The Joint FAO/WHO Codex
Alimentarius Commission has given recommendations for the methods of
analysis to be used for the determination of carbaryl residues
(FAO/WHO, 1986a).
1.4 Production and uses
Carbaryl is used as a broad spectrum contact and stomach insecticide
with some systemic properties on crops, trees, and ornamentals. It
is also used in public health and veterinary practice. In
agriculture, the usual application rate is 0.25-2 kg/ha, but for
tree fruit, up to 10 kg/ha.
Formulations of carbaryl include dusts (1.75-50%), wettable powders
(50-85%), oil- and water-based liquid suspensions (40-50%), granules
(5 and 10%), and baits.
2. SUMMARY AND EVALUATION
2.1 Environmental transport, distribution, and transformation
Under most conditions, carbaryl is not persistent in the
environment. In water, the hydrolysis half-life is dependent on
temperature, pH, and the initial concentration, and varies from
several minutes to several weeks. The major degradation product is
1-naphthol.
Accumulation of carbaryl, expressed as a bioconcentration factor in
the aquatic environment, has been found to be in the range of 14-75
for freshwater fish. Carbaryl is adsorbed more readily on soil with
a high organic content than on sandy soils. At the usual application
rates, under "good agricultural practice", dissipation is rapid,
with a half-life of 8 days-1 month, under normal conditions.
Carbaryl may occasionally be carried, by rainfall and soil
cultivation, from the surface of the soil, into the subsoil (one
metre from the surface).
Carbaryl contaminates vegetation either during spraying or by
migrating through contaminated soil into plants.
The degradation of carbaryl in the environment is determined by the
extent of the volatilization, photodecomposition, and chemical and
microbial degradation that occur in the soil, water, and plants. The
rate of decomposition is more rapid under hot climatic conditions.
2.2 Environmental levels and human exposure
Food represents the major source of carbaryl intake for the general
population. Residues in total dietary samples are relatively low,
ranging from trace amounts to 0.05 mg/kg. In the USA, the daily
intake during the first years of carbaryl application was 0.15
mg/day per person (in 7.4% of the composites) and decreased to 0.003
mg/day per person in 1969 (in only 0.8% of the composites). During
the carbaryl application period, it may be found occasionally in
surface water and reservoirs.
The general population can be exposed to carbaryl during pest
control operations in the home and in recreation areas.
Workers can be exposed to carbaryl during its manufacture,
formulation, packing, transportation, storage, and during and after
application. Concentrations in the working-air environment during
production varied from <1 mg/m3 to 30 mg/m3. Significant dermal
exposure of industrial and agricultural workers may occur, if
protective measures are inadequate.
2.3 Kinetics and metabolism
Carbaryl is rapidly absorbed in the lungs and digestive tract. In
human volunteers, dermal absorption of 45% of an applied dose in
acetone occurred in 8 h. However, in vitro dermal penetration data
and toxicity data indicate that dermal absorption usually occurs at
a much lower rate.
The principal metabolic pathways of carbaryl are ring hydroxylation
and hydrolysis. As a result, numerous metabolites are formed and
subjected to conjugation, with the formation of water-soluble
sulfates, glucuronides, and mercapturates, which are excreted in the
urine. Hydrolysis results in the formation of 1-naphthol, carbon
dioxide, and methylamine. Hydroxylation produces 4-hydroxycarbaryl,
5-hydroxycarbaryl, N-hydroxy-methylcarbaryl,
5-6-dihydro-5-6-dihydroxycarbaryl, and 1,4-naphthalendiol. The
principal metabolite in human beings is 1-naphthol.
Under normal exposure conditions, accumulation of carbaryl in
animals is unlikely. Carbaryl is excreted primarily via the urine,
since its product of hydrolysis, 1-naphthol, is mainly detoxified to
water-soluble conjugates. Enterohepatic cycling of carbaryl
metabolites is also considerable, especially after oral
administration.
The hydrolysis product, N-naphthol carbamic acid, is spontaneously
decomposed to methylamine and carbon dioxide. The methylamine moiety
is later demethylated to carbon dioxide and formate, the latter
being mainly excreted in the urine.
Carbaryl metabolites are also present in a small percentage of the
absorbed doses in saliva and milk.
2.4 Effects on organisms in the environment
LC50 values for crustacea vary from 5 to 9 µg/litre (water fleas,
mysid shrimps), 8 to 25 µg/litre (scud), and 500 to 2500 µg/litre
(crayfish). Aquatic insects have a similar range of sensitivity.
Plecoptera and Ephemeroptera (stoneflies and mayflies) are the most
sensitive groups. Molluscs are less susceptible with EC50s in the
range of a few mg/litre. For fish, most LC50 values are between 1-
30 mg/litre. Salmonids are the most sensitive group.
The acute toxicity of carbonyl for birds is low. The LD50 for
waterfowl and game birds is >1000 mg/kg. The red-winged blackbird
is the most susceptible bird tested (LD50=56 mg/kg). There was no
evidence for field effects on birds in forest areas sprayed with 1.1
kg carbaryl/ha.
Carbaryl is very toxic for earthworms and honey bees, the oral
LD50 for the latter being 0.18 µg/bee (about 1-2 mg/kg).
There are indications that carbaryl may temporarily influence the
species composition of both terrestrial and aquatic ecosystems. For
instance, one study showed that effects on certain terrestrial
invertebrate communities may persist for at least 10 months
following a single application.
2.5 Effects on experimental animals and in vitro test systems
The acute toxicity, expressed as the LD50, varies considerably
according to species, formulation, and vehicle. Estimates for the
oral LD50 for rats range from 200 to 850 mg/kg. Cats are more
sensitive, with an LD50 of 150 mg/kg. Pigs and monkeys are less
sensitive, with an LD50 of >1000 mg/kg.
The maximum achievable aerosol concentration of carbaryl of 792 mg
a.i./m3, over a 4-h exposure, resulted in the death of 1 out of 5
female rats. A single 4-h exposure to carbaryl aerosol, at a
concentration of 20 mg/m3, decreased cholinesterase (ChE) activity
in cats. However, this concentration did not produce any observable
effects in rats.
Carbaryl is a mild eye irritant and has little or no sensitizing
potential. In long-term studies, the NOEL for rats was 10 mg/kg body
weight (200 mg/kg diet), and that for dogs, 1.8 mg/kg body weight
(100 mg/kg diet). The long-term inhalation NOEL for cats was 0.16
mg/m3. The cumulative potential of carbaryl is low.
2.5.1 Reproduction effects
Carbaryl has been shown to affect mammalian reproduction and
perinatal development adversely in a number of species. Effects on
reproduction include impairment of fertility, decreased litter size,
and reduced postnatal viability. Developmental toxicity is seen as
increased in utero death, reduced fetal weight, and the
occurrence of malformation. With the exception of a small number of
studies, all adverse reproductive and developmental effects have
been noted only at doses that cause overt maternal toxicity; in a
number of cases, the maternal animal is more sensitive to carbaryl
than the conceptus. The maternal toxic effects include lethality,
decreased growth, and dystocia. Data indicate that the reproductive
and developmental processes of mammals are not especially sensitive
to carbaryl when compared to the susceptibility of the adult
organism.
2.5.2 Mutagenicity
Carbaryl has been evaluated for its potential mutagenicity in a
number of in vitro and in vivo tests, including tests on
bacterial, yeast, plant, insect, and mammalian systems, with a
variety of end-points.
The available evidence indicates that carbaryl does not have any
DNA-damaging properties. There are no confirmed reports of the
induction of mitotic recombination, gene conversion, and UDS in
prokaryotes ( H. influenzae, B. subtilis) and eukaryotes ( S.
cerevisiae, A. nidulans, cultured human lymphocytes, and rat
hepatocytes) in vitro.
Tests for gene mutations in a large number of bacterial assays, with
the exception of 2 cases, have given negative results. In several
studies of gene mutations in mammalian cells in vitro, carbaryl
only produced one equivocal positive result in a cell culture study.
However, the study had several shortcomings and the result has not
been confirmed in any other comparable studies.
Chromosomal damage with high dosages of carbaryl, has been reported
in in vitro studies on human, rat, and hamster cells and in
plants. No such effects have been observed in mammalian tests in
vivo, even at doses as high as 1000 mg/kg.
Carbaryl has been shown to induce disturbances in the spindle fibre
mechanism in plant and mammalian cells in vitro. The relevance of
plant assays for extrapolation to humans is unclear.
It can be concluded that the available data do not support the
presumption that carbaryl poses a risk of inducing genetic changes
in either somatic or germinal tissue of humans.
The nitrosated product of carbaryl, N-nitrosocarbaryl, is capable
of inducing mitotic recombination and gene conversion in prokaryotes
( H. influenzae, B. subtilis) and eukaryotes ( S. cerevisiae), in
vitro, and gives positive results in Escherichia coli spot tests.
Furthermore, experimental results indicate that N-nitrosocarbaryl
binds to DNA, causing alkali-sensitive bonds and single-strand
breakage.
Nitrosocarbaryl has not been established as a clastogen in vivo
(bone marrow and germ cells), even at high toxic doses.
2.5.3 Carcinogenicity
Carbaryl has been studied for its carcinogenic potential in numerous
studies on rats and mice. The results of most of these studies were
negative, but the studies are old and do not meet contemporary
standards. However, new studies meeting modern standards are in
progress on mice and rats. The latest IARC evaluation (IARC, 1987)
concluded that, as there are no data on cancer in humans, and
evidence of carcinogenicity in experimental animals is inadequate,
carbaryl could not be classified as to its carcinogenicity for
humans (Group 3).
N-nitrosocarbaryl has been shown to induce tumours locally in rats
(either sarcoma at the site of injection or forestomach squamous
cell carcinoma, when given by the oral route). Given the human
chemistry of carbaryl, the risk of N-nitrosocarbaryl
carcinogenicity from carbaryl exposure can be judged as negligible.
2.5.4 Effects on different organs and systems
(a) Nervous sysytem
The effects of carbaryl on the nervous system are primarily related
to cholinesterase inhibition and are usually transitory. The effect
on the central nervous system was studied in rats and monkeys. Oral
doses of 10-20 mg carbaryl/kg for 50 days have been reported to
disrupt learning and performance in rats.
In a small study on pigs, carbaryl (150 mg/kg body weight)
administered in the diet for 72-82 days, was reported to produce a
number of neuromuscular effects. Reversible leg weakness was noticed
in chickens that were given high doses of carbaryl. No evidence of
demyelination was observed in brain, sciatic nerve, or in spinal
cord sections examined microscopically. Similar effects were not
observed in long-term rodent studies.
(b) Immune system
Carbaryl, when administered in vivo at doses causing overt
clinical signs, has been reported to produce a variety of effects on
the immune system. Many of the effects described were detected at
doses close to the LD50. Most studies on rabbits and mice, at
doses permitting survival, have not produced significant effects on
the immune system. Shortcomings of several of these studies were a
lack of consistency and sometimes overt contradiction between
results, which prevents the description of a defined immunotoxic
mechanism.
(c) Blood
Carbaryl has been reported to affect coagulation, but there are
conflicts in the direction of the effect. In glucose-6-phosphate
dehydrogenase deficient sheep erythrocytes, carbaryl produces a
dose-dependent increase in Met-Hb formation. Human serum albumin
reacts in vitro with the ester group of carbaryl. Carbaryl binds
free blood amino acids.
(d) Liver metabolism
Disturbances in the carbohydrate metabolism and protein synthesis
and detoxification function of the liver in mammals have been
reported. Carbaryl is a weak inducer of hepatic microsomal
drug-metabolizing activity. It reduces phenobarbital sleeping time.
The hepatic levels of cytochrome P-450 and b5 are increased.
Changes in liver metabolism may account, in part, for the trifold
increase of the carbaryl LD50 in carbaryl pretreated rats.
(e) Gonadotropic function
Carbaryl has been reported to increase the gonadotropic function of
the hypophysis in rats.
2.5.5 Primary mechanism of toxicity
Carbaryl is an inhibitor of cholinesterase activity. This effect was
dose-related and quickly reversible. There was no aging of the
carbamylated cholinesterase. All identified metabolites of carbaryl
are appreciably less active as cholinesterase inhibitors than
carbaryl itself.
2.6 Effects on human beings
Carbaryl is easily absorbed through inhalation and via the oral
route, but less readily absorbed via the dermal route. Since the
inhibition of cholinesterase (ChE) is the principal mechanism of
carbaryl action, the clinical picture of intoxication is dominated
by ChE inhibition symptoms, such as: increased bronchial secretion,
excessive sweating, salivation, and lacrimation; pinpoint pupils,
bronchoconstriction, abdominal cramps (vomiting and diarrhoea);
bradycardia; fasciculation of fine muscles (in severe cases,
diaphragm and respiratory muscles also involved); tachycardia;
headache, dizziness, anxiety, mental confusion, convulsions, and
coma; and depression of the respiratory centre. Signs of
intoxication develop quickly after absorption and disappear rapidly
after exposure ends.
In controlled studies on human volunteers, single doses of less than
2 mg/kg were well-tolerated. A single dose of 250 mg (2.8 mg/kg)
produced moderate ChE inhibition symptoms (epigastric pain and
sweating) within 20 min. Complete recovery occurred within 2 h of
treatment with atropine sulfate.
In cases of occupational overexposure to carbaryl, mild symptoms are
observed long before a dangerous dose is absorbed, which is why
severe cases of occupational intoxication with carbaryl are rare.
During agricultural application, dermal exposure may play an
important role. Local irritative effects are not usually observed;
however, the appearance of a skin rash after accidental splashing
with carbaryl formulations has been described.
There are conflicting data about effects of carbaryl on sperm count
and changes in sperm morphology in plant workers. No adverse effect
on reproduction has been reported.
The most sensitive biological indicator of carbaryl exposure is the
appearance of 1-naphthol in the urine and the decrease of ChE
activity in the blood. Levels of 1-naphthol in the urine can be used
as a biological indicator, if there is no 1-naphthol in the working
environment. During occupational exposure, 40% of the urine samples
contained more than 10 mg total 1-naphthol/litre. In one case of
acute intoxication, a level of 31 mg/litre was found in the urine.
The hazard level is >10 mg 1-naphthol/litre and the symptomatic
level, 30 mg/litre, in urine (WHO, 1978).
Measurement of the ChE activity can be a very sensitive test for
monitoring, provided that measurement is conducted soon after
exposure.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
The health hazard of carbaryl for human beings is judged to be low,
because of its low vapour pressure, rapid degradation, the rapid
spontaneous recovery of inhibited cholinesterase, and the fact that
symptoms usually appear well before a dangerous dose has accumulated
in the body. Good carcinogenicity studies, which meet modern
standards, are not yet available.
3.1.1 General population
Residue levels of carbaryl in food and drinking-water, remaining
after its normal use in agriculture, are far below the ADI (0.01
mg/kg body weight per day), and not likely to produce health hazards
in the general population.
3.1.2 Subpopulations at high risk
Use of carbaryl for public health purposes in the home or in
recreation areas may create overexposure, if the rules for its
application are neglected.
3.1.3 Occupational exposure
By enforcing reasonable work practices, including safety
precautions, personal protection, and proper supervision,
occupational exposure during the manufacture, formulation, and
application of carbaryl will not create hazards. Undiluted
concentrations must be handled with great care, because improper
work practices may cause skin contamination. Work air concentrations
should not exceed 5 mg/m3.
3.1.4 Environmental effects
Carbaryl is toxic for honey bees and earthworms and should not be
applied to crops during flowering.
With normal use, carbaryl should not cause environmental concern.
Carbaryl is adsorbed on soil particles to a great extent and does
not readily leach into groundwater. It is rapidly degraded in the
environment and therefore is not persistent. Use of carbaryl should
not result in harmful short-term effects on the ecosystem.
3.2 Recommendations
* The handling and application of carbaryl should be accomplished
with the care given to all pesticides. Instructions for proper
usage, provided on the package containing the chemical, should
be carefully followed.
* Manufacture, formulation, use, and disposal of carbaryl should
be carefully managed to minimize contamination of the
environment.
* Regularly exposed workers should receive periodic health
evaluations.
* Application of carbaryl should be timed to avoid effects on
non-target species.
* Carcinogenicity studies, which meet modern standards, should be
conducted.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Human health hazards, prevention and protection, first aid
Carbaryl is a carbamate insecticide with anticholinesterase
activity. It is moderately toxic and hazardous (the acute oral
LD50 for the rat is approximately 300 mg/kg (WHO, 1990). It can be
absorbed via the gastrointestinal tract and through inhalation, and,
to a lesser extent, through the skin. Because of its rapid
metabolism and excretion, it does not accumulate in tissues. It can
be hazardous for human beings if handled incorrectly and carelessly.
It is, therefore, essential that the correct precautions should be
observed during handling and use.
The human health hazards associated with certain types of exposure
to carbaryl, together with preventive and protective measures and
first aid and are listed in Table 2.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
Carbaryl is a short-acting anticholinesterase agent, with the
important characteristic of rapid reversibility of enzyme
inhibition. It inactivates cholinesterase, resulting in the
accumulation of acetylcholine at synapses in the nervous system, at
neuromuscular junctions of the skeletal and smooth muscles, and in
secretory glands. Signs and symptoms of overexposure may include
headache, tiredness, pinpoint pupils, blurred vision, lacrimation,
excessive nasal discharge or salivation, sweating, dizziness,
stomach pain, nausea, vomiting, diarrhoea, tremor, difficulties in
breathing, cyanosis, convulsions,and unconsciousness. Symptoms
appear immediately or within 12 h of exposure.
For a more complete treatise on the mode of action and the effects
of carbamate pesticides, especially their effects on the nervous
system, refer to EHC 64: Carbamate pesticides - a general
introduction (WHO, 1986).
4.1.1.2 Medical treatment
If more than 1 g carbaryl equivalent has been ingested (unless the
patient is vomiting) rapid gastric lavage should be performed using
5% sodium bicarbonate, if available. In case of skin contact, the
skin should be washed with soap and water. Contaminated eyes should
be washed with isotonic saline or water. Since the symptoms of
poisoning with carbaryl disappear comparatively rapidly, atropine
treatment is often not necessary by the time the patient reaches the
place where the antidote is available. In case of accidental
poisoning or of manifest symptoms, 2 mg of atropine (adult dose) may
be given im, or even iv, and repeated if necessary. Care should be
taken to avoid overdosage with atropine in the case of carbaryl
poisoning, especially in children. Mechanical respiratory assistance
with oxygen may be required in extreme cases, if the patient is
unconscious or in respiratory distress.
Oximes, such as pralidoxime, barbiturates, and central
stimulants of all kinds are contraindicated.
If the acute toxic effect is survived, the chances of complete
recovery are very good.
More information on the treatment of carbamate pesticide poisoning
is given in the Annex to this Guide.
4.1.2 Health surveillance advice
Pre-exposure, and annual, general medical examinations should be
carried out on workers exposed to carbaryl. In the pre-employment
examinations, medical fitness for work with carbamates and a
baseline cholinesterase level should be established.
Overexposure to carbamates can be monitored by the measurement of
erythrocyte- and whole blood-ChE activity, but, in the case of
carbamates, return to normal values is generally rapid. Moreover,
physiological variations in erythrocyte- and whole blood-ChE values
occur in healthy persons.
In exposed workers, the urinary excretion of 1-naphthol should be
monitored; 1.5-4 mg/litre can be considered as acceptable. A hazard
level is >10 mg/litre, and, at >30 mg/litre, toxic signs and
symptoms can be expected.
Table 2. Health hazards, prevention and protection, first aid
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Readily absorbed
via skin, ingestion, and
inhalation; may cause carbamate
poisoning: excessive sweating,
headache, weakness, giddiness,
nausea, vomiting, stomach pains,
blurred vision, slurred speech,
tremors, and convulsions
SKIN: Irritation; skin rash Wear impervious clothing, gloves, Remove contaminated clothing and shoes;
face shields wash the skin with plenty of water and soap
(or mild detergent); get medical attention
attention immediately
EYES: Irritation Wear face shield or safety goggles Wash eyes immediately with clean water for at
least 15 min, lifting the lower and upper
lids occasionally; if irritation persists, get
medical attention immediately
INHALATION: Irritation Use proper exhaust ventilation Move the exposed person to fresh air at once;
and suitable respiratory protection if breathing has stopped, perform artificial
respiration; keep the affected person warm and
at rest; get medical attention immediately
INGESTION: May lead to severe Do not eat, drink, or smoke when Obtain medical attention immediately; in case
poisoning working with the compound of accidental or intentional ingestion, and if
the patient is still conscious, induce vomiting
immediately: if breathing has stopped, apply
artificial respiration
4.2 Explosion and fire hazards
Technical grade material will not burn, but formulations in organic
solvents may be highly flammable. Use dry powder, carbon dioxide, or
alcohol-resistant foam, sand, or earth for dealing with fires.
If carbaryl is involved in a major fire or in a fire involving other
products, advise the fire service that protective clothing and
breathing apparatus should be worn, because toxic decomposition
products, such as nitrogen oxides, methylamine, and carbon monoxide
may be formed. Also, warn the authorities that carbaryl is toxic for
fish, and that the use of water should be confined to the cooling of
unaffected stock, thus avoiding the accumulation of polluted run-off
from the site.
4.3 Storage
Formulations should be stored in locked buildings, preferably
dedicated to insecticides. They should be kept in clearly labelled,
leakproof containers.
Keep formulations and dilutions out of reach of children and
unauthorized persons. Do not store near foodstuffs or animal feed.
4.4 Transport
Comply with any local requirements regarding the movement of
hazardous goods. Do not load with foodstuffs or animal feed. Before
dispatch, check that containers are sound and labels undamaged.
4.5 Spillage and disposal
4.5.1 Spillage
Avoid exposure, if possible, by the use of appropriate protective
clothing and masks.
Empty any product remaining in damaged or leaking containers into a
clean, empty drum, and label.
Absorb spillage with lime, damp sawdust, sand, or earth and dispose
of safely (see below). If spillage is large, contain it by building
a barrier of earth or sandbags.
Decontaminate empty, damaged, or leaking containers with a 10%
sodium carbonate solution added at the rate of at least 1 litre per
20-litre drum. Puncture and crush containers to prevent reuse.
4.5.2 Disposal
Waste that contains carbaryl should be burned in a high-temperature
incinerator with effluent gas scrubbing.
Where no incinerator is available, contaminated absorbents or
surplus products should be decomposed by hydrolysis at pH12 or
above. Contact with a suitable hydrolysing agent is required to
ensure the degradation of the active ingredient to a safe level. For
emulsifiable material: 5% sodium hydroxide (caustic soda) solution
or saturated (7-10%) sodium carbonate (washing soda) solution can be
used. For non-emulsifiable material, use a 1:1 mixture (by volume)
of either of the above solutions and a water/oil-soluble solvent,
such as denatured alcohol, monoethylene glycol, hexylene glycol, or
isopropanol. Cover the material with a hydrolysing agent and put
aside for 24 h. Before disposal of the resultant waste, the material
must be analysed to ensure that the active ingredient has been
degraded to a safe level.
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage into streams,
watercourses, open waterways, ditches, fields with drainage systems,
or the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
At worst, carbaryl has only limited persistence in the environment.
In water, it is easily degraded under most conditions; the half-life
ranges from a few minutes to several weeks. In soil, the half-life
ranges from a few days to several weeks. Carbaryl is adsorbed on
soil and leaching into water is not of great significance.
There is no evidence of general phytotoxicity when used at
recommended rates, but damage to apples, pears, and water-melons has
been reported. Excessive application may retard the germination of
grasses.
Carbaryl is toxic for earthworms. It is hazardous for honey bees and
it should not be applied to crops during flowering.
Avoid spraying over bodies of water. Do not contaminate ponds,
waterways, or ditches with the product or used containers, as
carbaryl has some toxicity for some aquatic species.
In case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
6.1 Previous evaluations by international bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) evaluated
carbaryl at its meetings in 1963, 1965, 1966, 1967, 1968, 1969,
1970, 1971, 1973, 1975, 1976, 1977, 1979, and 1984. In 1973, an
Acceptable Daily Intake (ADI) of 0-0.01 mg/kg body weight was
established. In addition, Maximum Residue Limits (MRLs) have been
set for specified crops (see Table 3).
The International Agency for Research on Cancer (IARC), evaluated
carbaryl in 1976 and concluded that the available data did not allow
an evaluation of the carcinogenicity of carbaryl to be made.
In the WHO recommended classification of pesticides by hazard,
technical carbaryl is classified in Class II as moderately hazardous
in normal use. WHO has issued a data sheet on carbaryl (No. 3).
6.2 Exposure limit values
Some exposure limit values are given in the Table 3.
6.3 Specific restrictions
Carbaryl is approved for use as a pesticide in many countries, in
each of which, specific uses are defined, as well as limitations and
precautions.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Workplace Argentina Maximum permissible concentration (MPC) 1979
- time-weighted average (TWA) 5 mg/m3
- short-term exposure limit 10 mg/m3
Bulgaria Maximum permissible concentration (MPC) 1 mg/m3 1971
Germany Maximum work-site concentration (MAK) 1986
- 8-h time-weighted averageb (TWA) 5 mg/m3
Hungary Maximum acceptable concentration (MAK) 1 mg/m3 1988
United Occupational exposure standard (OES) 1989
Kingdom - 8-h time-weighted average (TWA) 5 mg/m3
- 10-min short-term exposure level (STEL) 10 mg/m3
USA (OSHA) Permissible exposure limit (PEL) 1989
- 8-h time-weighted average (TWA) 5 mg/m3
USA (NIOSH) Recommended exposure limit (REL)
- time-weighted average (TWA) 5 mg/m3 1976
USA (ACGIH) Threshold limit value (TLV) 1986
- 8-h time weighted average (TWA) 5 mg/m3
USSR Ceiling value (aerosol) 1 mg/m3 1977
WHO Maximum permissible concentrationb 5 mg/m3 1982
(MPC) (tentative)
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration 1977
- average per day 0.01 mg/m3
- once per day 0.02 mg/m3
WATER Surface USSR Maximum allowable concentration (MAC) 0.1 mg/litre 1983
Surface for USSR Maximum allowable concentration (MAC) 0.0005 mg/litre 1983
fishing
SOIL USSR Maximum allowable concentration (MAC) 0.05 mg/kg 1973
FOOD Intake FAO/WHO Acceptable daily intake (ADI) 0-0.01 mg/kg 1973
body weight
Residues FAO/WHO Maximum residue limit (MRL)
(Codex (in specified products): 0.1-100 mg/kg 1986
Alimentarius) animal feedstuffs (green), alfalfa, bean
and pea vines, colver corn, forage
cowpea, foliage, grasses, peanut hay,
sorghum forage, soybean foliage,
sugarbeet tops 100 mg/kg
bran 20 mg/kg
apricots, asparagus, blackberries,
boysenberries, cherries, kiwi fruit
leafy vegetables (except brassicas),
nectarines, nuts (whole), okra, olives
(fresh), peaches, plums, raspberries,
sorghum grain 10 mg/kg
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
blueberries, citrus fruit, cranberries
strawberries 7 mg/kg
apples, aubergines, bananas, barley,
beans, brassicas, grapes, oats, pears,
peas (including pod), peppers, poultry
(skin), rice (in husks and hulled),
rye, tomatoes, wheat 5 mg/kg
cucurbits (including melons) 3 mg/kg
peanuts (whole), root crops (beets,
carrots, parsnips, radishes, rutabagas),
wholemeal flour 2 mg/kg
cottonseed (whole), cowpeas, nuts (shelled),
olives (processed), soybeans, (dry),
sweetcorn (kernels) 1 mg/kg
eggs (shell-free), poultry meat 0.5 mg/kg
meat of cattle, goats and sheep, 0.2 mg/kg
potatoes, sugarbeet, wheat flour (white)
milk and milk products 0.1 mg/kg
a TWA - time-weighted average over one working day (usually 8 h).
b Skin absorption.
6.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transport of
Dangerous Goods classifies carbaryl in:
- Hazard Class 6.1: poisonous substances
- Packing Group III: a substance that has a relatively low risk
of poisoning during transport (solid >30%; liquid >10%).
The following label should be used:
Molecular formula: C12H11NO2
Common name: Carbaryl (BSI)
CAS chemical name: 1-naphthalenyl methylcarbamate
IUPAC chemical name: 1-naphthyl methylcarbamate
Trade names: Arylat, Arylam, Atoxan, Bercema,
Carbaryl, Carbamate, Carpolin,
Carbotox, Denapon, Dicarbam, Hexavin,
Monsur, Murrin, Panam, Pamex,
Prosevor, Rayvon, Sevimol, Sevin,
Viaxon, etc.
CAS registry number: 63-25-2
RTECS registry number: FC5950000
Relative molecular mass: 201
Conversion factor: 1 mg/m3=8.22 ppm
1 ppm=0.12 mg/m3 at 35 °C
1.2 Physical and chemical properties
Carbaryl is an odourless, white, crystalline solid, with a low
volatility. It is practically insoluble in water, but soluble in
most organic solvents, stable to light and heat up to 70 °C, and
easily hydrolysed by alkaline materials. It is non-corrosive.
The technical product is principally manufactured in the USA;
however, small amounts are produced in other parts of the world.
The technical product, manufactured in the USA, is produced to a
minimum purity of 99% w/w carbaryl with a specified content of the
impurity, 2-naphthyl carbamate isomer (sometimes known as
beta-carbaryl), of <0.05% w/w.
The Food and Agriculture Organization of the United Nations (FAO)
specifies a minimum purity of 98% w/w carbaryl with a specified w/w
content of the impurity, 2-naphthyl carbamate isomer, of <0.05%.
Some physical properties of carbaryl are given in Table 1.
Table 1. Physical properties
Melting point (°C) 142
Boiling point (°C) decomposing
Solubility in water (30 °C) 40 mg/litre
Specific density (20 °C) 1.23
Relative vapour density -
Vapour pressure 1.17 x 10-6-3.1 x 10-7 mmHg at
24-25 °C
Flash point 193 °C
Octanol/water partition
coefficient (log Kow) 1.59-2.3
Flammability (explosive)
limits -
Relative molecular mass 201
1.3 Analytical methods
Product analysis can be carried out using infrared spectroscopy.
Residues can be determined by GLC and TLC. The Joint FAO/WHO Codex
Alimentarius Commission has given recommendations for the methods of
analysis to be used for the determination of carbaryl residues
(FAO/WHO, 1986a).
1.4 Production and uses
Carbaryl is used as a broad spectrum contact and stomach insecticide
with some systemic properties on crops, trees, and ornamentals. It
is also used in public health and veterinary practice. In
agriculture, the usual application rate is 0.25-2 kg/ha, but for
tree fruit, up to 10 kg/ha.
Formulations of carbaryl include dusts (1.75-50%), wettable powders
(50-85%), oil- and water-based liquid suspensions (40-50%), granules
(5 and 10%), and baits.
2. SUMMARY AND EVALUATION
2.1 Environmental transport, distribution, and transformation
Under most conditions, carbaryl is not persistent in the
environment. In water, the hydrolysis half-life is dependent on
temperature, pH, and the initial concentration, and varies from
several minutes to several weeks. The major degradation product is
1-naphthol.
Accumulation of carbaryl, expressed as a bioconcentration factor in
the aquatic environment, has been found to be in the range of 14-75
for freshwater fish. Carbaryl is adsorbed more readily on soil with
a high organic content than on sandy soils. At the usual application
rates, under "good agricultural practice", dissipation is rapid,
with a half-life of 8 days-1 month, under normal conditions.
Carbaryl may occasionally be carried, by rainfall and soil
cultivation, from the surface of the soil, into the subsoil (one
metre from the surface).
Carbaryl contaminates vegetation either during spraying or by
migrating through contaminated soil into plants.
The degradation of carbaryl in the environment is determined by the
extent of the volatilization, photodecomposition, and chemical and
microbial degradation that occur in the soil, water, and plants. The
rate of decomposition is more rapid under hot climatic conditions.
2.2 Environmental levels and human exposure
Food represents the major source of carbaryl intake for the general
population. Residues in total dietary samples are relatively low,
ranging from trace amounts to 0.05 mg/kg. In the USA, the daily
intake during the first years of carbaryl application was 0.15
mg/day per person (in 7.4% of the composites) and decreased to 0.003
mg/day per person in 1969 (in only 0.8% of the composites). During
the carbaryl application period, it may be found occasionally in
surface water and reservoirs.
The general population can be exposed to carbaryl during pest
control operations in the home and in recreation areas.
Workers can be exposed to carbaryl during its manufacture,
formulation, packing, transportation, storage, and during and after
application. Concentrations in the working-air environment during
production varied from <1 mg/m3 to 30 mg/m3. Significant dermal
exposure of industrial and agricultural workers may occur, if
protective measures are inadequate.
2.3 Kinetics and metabolism
Carbaryl is rapidly absorbed in the lungs and digestive tract. In
human volunteers, dermal absorption of 45% of an applied dose in
acetone occurred in 8 h. However, in vitro dermal penetration data
and toxicity data indicate that dermal absorption usually occurs at
a much lower rate.
The principal metabolic pathways of carbaryl are ring hydroxylation
and hydrolysis. As a result, numerous metabolites are formed and
subjected to conjugation, with the formation of water-soluble
sulfates, glucuronides, and mercapturates, which are excreted in the
urine. Hydrolysis results in the formation of 1-naphthol, carbon
dioxide, and methylamine. Hydroxylation produces 4-hydroxycarbaryl,
5-hydroxycarbaryl, N-hydroxy-methylcarbaryl,
5-6-dihydro-5-6-dihydroxycarbaryl, and 1,4-naphthalendiol. The
principal metabolite in human beings is 1-naphthol.
Under normal exposure conditions, accumulation of carbaryl in
animals is unlikely. Carbaryl is excreted primarily via the urine,
since its product of hydrolysis, 1-naphthol, is mainly detoxified to
water-soluble conjugates. Enterohepatic cycling of carbaryl
metabolites is also considerable, especially after oral
administration.
The hydrolysis product, N-naphthol carbamic acid, is spontaneously
decomposed to methylamine and carbon dioxide. The methylamine moiety
is later demethylated to carbon dioxide and formate, the latter
being mainly excreted in the urine.
Carbaryl metabolites are also present in a small percentage of the
absorbed doses in saliva and milk.
2.4 Effects on organisms in the environment
LC50 values for crustacea vary from 5 to 9 µg/litre (water fleas,
mysid shrimps), 8 to 25 µg/litre (scud), and 500 to 2500 µg/litre
(crayfish). Aquatic insects have a similar range of sensitivity.
Plecoptera and Ephemeroptera (stoneflies and mayflies) are the most
sensitive groups. Molluscs are less susceptible with EC50s in the
range of a few mg/litre. For fish, most LC50 values are between 1-
30 mg/litre. Salmonids are the most sensitive group.
The acute toxicity of carbonyl for birds is low. The LD50 for
waterfowl and game birds is >1000 mg/kg. The red-winged blackbird
is the most susceptible bird tested (LD50=56 mg/kg). There was no
evidence for field effects on birds in forest areas sprayed with 1.1
kg carbaryl/ha.
Carbaryl is very toxic for earthworms and honey bees, the oral
LD50 for the latter being 0.18 µg/bee (about 1-2 mg/kg).
There are indications that carbaryl may temporarily influence the
species composition of both terrestrial and aquatic ecosystems. For
instance, one study showed that effects on certain terrestrial
invertebrate communities may persist for at least 10 months
following a single application.
2.5 Effects on experimental animals and in vitro test systems
The acute toxicity, expressed as the LD50, varies considerably
according to species, formulation, and vehicle. Estimates for the
oral LD50 for rats range from 200 to 850 mg/kg. Cats are more
sensitive, with an LD50 of 150 mg/kg. Pigs and monkeys are less
sensitive, with an LD50 of >1000 mg/kg.
The maximum achievable aerosol concentration of carbaryl of 792 mg
a.i./m3, over a 4-h exposure, resulted in the death of 1 out of 5
female rats. A single 4-h exposure to carbaryl aerosol, at a
concentration of 20 mg/m3, decreased cholinesterase (ChE) activity
in cats. However, this concentration did not produce any observable
effects in rats.
Carbaryl is a mild eye irritant and has little or no sensitizing
potential. In long-term studies, the NOEL for rats was 10 mg/kg body
weight (200 mg/kg diet), and that for dogs, 1.8 mg/kg body weight
(100 mg/kg diet). The long-term inhalation NOEL for cats was 0.16
mg/m3. The cumulative potential of carbaryl is low.
2.5.1 Reproduction effects
Carbaryl has been shown to affect mammalian reproduction and
perinatal development adversely in a number of species. Effects on
reproduction include impairment of fertility, decreased litter size,
and reduced postnatal viability. Developmental toxicity is seen as
increased in utero death, reduced fetal weight, and the
occurrence of malformation. With the exception of a small number of
studies, all adverse reproductive and developmental effects have
been noted only at doses that cause overt maternal toxicity; in a
number of cases, the maternal animal is more sensitive to carbaryl
than the conceptus. The maternal toxic effects include lethality,
decreased growth, and dystocia. Data indicate that the reproductive
and developmental processes of mammals are not especially sensitive
to carbaryl when compared to the susceptibility of the adult
organism.
2.5.2 Mutagenicity
Carbaryl has been evaluated for its potential mutagenicity in a
number of in vitro and in vivo tests, including tests on
bacterial, yeast, plant, insect, and mammalian systems, with a
variety of end-points.
The available evidence indicates that carbaryl does not have any
DNA-damaging properties. There are no confirmed reports of the
induction of mitotic recombination, gene conversion, and UDS in
prokaryotes ( H. influenzae, B. subtilis) and eukaryotes ( S.
cerevisiae, A. nidulans, cultured human lymphocytes, and rat
hepatocytes) in vitro.
Tests for gene mutations in a large number of bacterial assays, with
the exception of 2 cases, have given negative results. In several
studies of gene mutations in mammalian cells in vitro, carbaryl
only produced one equivocal positive result in a cell culture study.
However, the study had several shortcomings and the result has not
been confirmed in any other comparable studies.
Chromosomal damage with high dosages of carbaryl, has been reported
in in vitro studies on human, rat, and hamster cells and in
plants. No such effects have been observed in mammalian tests in
vivo, even at doses as high as 1000 mg/kg.
Carbaryl has been shown to induce disturbances in the spindle fibre
mechanism in plant and mammalian cells in vitro. The relevance of
plant assays for extrapolation to humans is unclear.
It can be concluded that the available data do not support the
presumption that carbaryl poses a risk of inducing genetic changes
in either somatic or germinal tissue of humans.
The nitrosated product of carbaryl, N-nitrosocarbaryl, is capable
of inducing mitotic recombination and gene conversion in prokaryotes
( H. influenzae, B. subtilis) and eukaryotes ( S. cerevisiae), in
vitro, and gives positive results in Escherichia coli spot tests.
Furthermore, experimental results indicate that N-nitrosocarbaryl
binds to DNA, causing alkali-sensitive bonds and single-strand
breakage.
Nitrosocarbaryl has not been established as a clastogen in vivo
(bone marrow and germ cells), even at high toxic doses.
2.5.3 Carcinogenicity
Carbaryl has been studied for its carcinogenic potential in numerous
studies on rats and mice. The results of most of these studies were
negative, but the studies are old and do not meet contemporary
standards. However, new studies meeting modern standards are in
progress on mice and rats. The latest IARC evaluation (IARC, 1987)
concluded that, as there are no data on cancer in humans, and
evidence of carcinogenicity in experimental animals is inadequate,
carbaryl could not be classified as to its carcinogenicity for
humans (Group 3).
N-nitrosocarbaryl has been shown to induce tumours locally in rats
(either sarcoma at the site of injection or forestomach squamous
cell carcinoma, when given by the oral route). Given the human
chemistry of carbaryl, the risk of N-nitrosocarbaryl
carcinogenicity from carbaryl exposure can be judged as negligible.
2.5.4 Effects on different organs and systems
(a) Nervous sysytem
The effects of carbaryl on the nervous system are primarily related
to cholinesterase inhibition and are usually transitory. The effect
on the central nervous system was studied in rats and monkeys. Oral
doses of 10-20 mg carbaryl/kg for 50 days have been reported to
disrupt learning and performance in rats.
In a small study on pigs, carbaryl (150 mg/kg body weight)
administered in the diet for 72-82 days, was reported to produce a
number of neuromuscular effects. Reversible leg weakness was noticed
in chickens that were given high doses of carbaryl. No evidence of
demyelination was observed in brain, sciatic nerve, or in spinal
cord sections examined microscopically. Similar effects were not
observed in long-term rodent studies.
(b) Immune system
Carbaryl, when administered in vivo at doses causing overt
clinical signs, has been reported to produce a variety of effects on
the immune system. Many of the effects described were detected at
doses close to the LD50. Most studies on rabbits and mice, at
doses permitting survival, have not produced significant effects on
the immune system. Shortcomings of several of these studies were a
lack of consistency and sometimes overt contradiction between
results, which prevents the description of a defined immunotoxic
mechanism.
(c) Blood
Carbaryl has been reported to affect coagulation, but there are
conflicts in the direction of the effect. In glucose-6-phosphate
dehydrogenase deficient sheep erythrocytes, carbaryl produces a
dose-dependent increase in Met-Hb formation. Human serum albumin
reacts in vitro with the ester group of carbaryl. Carbaryl binds
free blood amino acids.
(d) Liver metabolism
Disturbances in the carbohydrate metabolism and protein synthesis
and detoxification function of the liver in mammals have been
reported. Carbaryl is a weak inducer of hepatic microsomal
drug-metabolizing activity. It reduces phenobarbital sleeping time.
The hepatic levels of cytochrome P-450 and b5 are increased.
Changes in liver metabolism may account, in part, for the trifold
increase of the carbaryl LD50 in carbaryl pretreated rats.
(e) Gonadotropic function
Carbaryl has been reported to increase the gonadotropic function of
the hypophysis in rats.
2.5.5 Primary mechanism of toxicity
Carbaryl is an inhibitor of cholinesterase activity. This effect was
dose-related and quickly reversible. There was no aging of the
carbamylated cholinesterase. All identified metabolites of carbaryl
are appreciably less active as cholinesterase inhibitors than
carbaryl itself.
2.6 Effects on human beings
Carbaryl is easily absorbed through inhalation and via the oral
route, but less readily absorbed via the dermal route. Since the
inhibition of cholinesterase (ChE) is the principal mechanism of
carbaryl action, the clinical picture of intoxication is dominated
by ChE inhibition symptoms, such as: increased bronchial secretion,
excessive sweating, salivation, and lacrimation; pinpoint pupils,
bronchoconstriction, abdominal cramps (vomiting and diarrhoea);
bradycardia; fasciculation of fine muscles (in severe cases,
diaphragm and respiratory muscles also involved); tachycardia;
headache, dizziness, anxiety, mental confusion, convulsions, and
coma; and depression of the respiratory centre. Signs of
intoxication develop quickly after absorption and disappear rapidly
after exposure ends.
In controlled studies on human volunteers, single doses of less than
2 mg/kg were well-tolerated. A single dose of 250 mg (2.8 mg/kg)
produced moderate ChE inhibition symptoms (epigastric pain and
sweating) within 20 min. Complete recovery occurred within 2 h of
treatment with atropine sulfate.
In cases of occupational overexposure to carbaryl, mild symptoms are
observed long before a dangerous dose is absorbed, which is why
severe cases of occupational intoxication with carbaryl are rare.
During agricultural application, dermal exposure may play an
important role. Local irritative effects are not usually observed;
however, the appearance of a skin rash after accidental splashing
with carbaryl formulations has been described.
There are conflicting data about effects of carbaryl on sperm count
and changes in sperm morphology in plant workers. No adverse effect
on reproduction has been reported.
The most sensitive biological indicator of carbaryl exposure is the
appearance of 1-naphthol in the urine and the decrease of ChE
activity in the blood. Levels of 1-naphthol in the urine can be used
as a biological indicator, if there is no 1-naphthol in the working
environment. During occupational exposure, 40% of the urine samples
contained more than 10 mg total 1-naphthol/litre. In one case of
acute intoxication, a level of 31 mg/litre was found in the urine.
The hazard level is >10 mg 1-naphthol/litre and the symptomatic
level, 30 mg/litre, in urine (WHO, 1978).
Measurement of the ChE activity can be a very sensitive test for
monitoring, provided that measurement is conducted soon after
exposure.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
The health hazard of carbaryl for human beings is judged to be low,
because of its low vapour pressure, rapid degradation, the rapid
spontaneous recovery of inhibited cholinesterase, and the fact that
symptoms usually appear well before a dangerous dose has accumulated
in the body. Good carcinogenicity studies, which meet modern
standards, are not yet available.
3.1.1 General population
Residue levels of carbaryl in food and drinking-water, remaining
after its normal use in agriculture, are far below the ADI (0.01
mg/kg body weight per day), and not likely to produce health hazards
in the general population.
3.1.2 Subpopulations at high risk
Use of carbaryl for public health purposes in the home or in
recreation areas may create overexposure, if the rules for its
application are neglected.
3.1.3 Occupational exposure
By enforcing reasonable work practices, including safety
precautions, personal protection, and proper supervision,
occupational exposure during the manufacture, formulation, and
application of carbaryl will not create hazards. Undiluted
concentrations must be handled with great care, because improper
work practices may cause skin contamination. Work air concentrations
should not exceed 5 mg/m3.
3.1.4 Environmental effects
Carbaryl is toxic for honey bees and earthworms and should not be
applied to crops during flowering.
With normal use, carbaryl should not cause environmental concern.
Carbaryl is adsorbed on soil particles to a great extent and does
not readily leach into groundwater. It is rapidly degraded in the
environment and therefore is not persistent. Use of carbaryl should
not result in harmful short-term effects on the ecosystem.
3.2 Recommendations
* The handling and application of carbaryl should be accomplished
with the care given to all pesticides. Instructions for proper
usage, provided on the package containing the chemical, should
be carefully followed.
* Manufacture, formulation, use, and disposal of carbaryl should
be carefully managed to minimize contamination of the
environment.
* Regularly exposed workers should receive periodic health
evaluations.
* Application of carbaryl should be timed to avoid effects on
non-target species.
* Carcinogenicity studies, which meet modern standards, should be
conducted.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Human health hazards, prevention and protection, first aid
Carbaryl is a carbamate insecticide with anticholinesterase
activity. It is moderately toxic and hazardous (the acute oral
LD50 for the rat is approximately 300 mg/kg (WHO, 1990). It can be
absorbed via the gastrointestinal tract and through inhalation, and,
to a lesser extent, through the skin. Because of its rapid
metabolism and excretion, it does not accumulate in tissues. It can
be hazardous for human beings if handled incorrectly and carelessly.
It is, therefore, essential that the correct precautions should be
observed during handling and use.
The human health hazards associated with certain types of exposure
to carbaryl, together with preventive and protective measures and
first aid and are listed in Table 2.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
Carbaryl is a short-acting anticholinesterase agent, with the
important characteristic of rapid reversibility of enzyme
inhibition. It inactivates cholinesterase, resulting in the
accumulation of acetylcholine at synapses in the nervous system, at
neuromuscular junctions of the skeletal and smooth muscles, and in
secretory glands. Signs and symptoms of overexposure may include
headache, tiredness, pinpoint pupils, blurred vision, lacrimation,
excessive nasal discharge or salivation, sweating, dizziness,
stomach pain, nausea, vomiting, diarrhoea, tremor, difficulties in
breathing, cyanosis, convulsions,and unconsciousness. Symptoms
appear immediately or within 12 h of exposure.
For a more complete treatise on the mode of action and the effects
of carbamate pesticides, especially their effects on the nervous
system, refer to EHC 64: Carbamate pesticides - a general
introduction (WHO, 1986).
4.1.1.2 Medical treatment
If more than 1 g carbaryl equivalent has been ingested (unless the
patient is vomiting) rapid gastric lavage should be performed using
5% sodium bicarbonate, if available. In case of skin contact, the
skin should be washed with soap and water. Contaminated eyes should
be washed with isotonic saline or water. Since the symptoms of
poisoning with carbaryl disappear comparatively rapidly, atropine
treatment is often not necessary by the time the patient reaches the
place where the antidote is available. In case of accidental
poisoning or of manifest symptoms, 2 mg of atropine (adult dose) may
be given im, or even iv, and repeated if necessary. Care should be
taken to avoid overdosage with atropine in the case of carbaryl
poisoning, especially in children. Mechanical respiratory assistance
with oxygen may be required in extreme cases, if the patient is
unconscious or in respiratory distress.
Oximes, such as pralidoxime, barbiturates, and central
stimulants of all kinds are contraindicated.
If the acute toxic effect is survived, the chances of complete
recovery are very good.
More information on the treatment of carbamate pesticide poisoning
is given in the Annex to this Guide.
4.1.2 Health surveillance advice
Pre-exposure, and annual, general medical examinations should be
carried out on workers exposed to carbaryl. In the pre-employment
examinations, medical fitness for work with carbamates and a
baseline cholinesterase level should be established.
Overexposure to carbamates can be monitored by the measurement of
erythrocyte- and whole blood-ChE activity, but, in the case of
carbamates, return to normal values is generally rapid. Moreover,
physiological variations in erythrocyte- and whole blood-ChE values
occur in healthy persons.
In exposed workers, the urinary excretion of 1-naphthol should be
monitored; 1.5-4 mg/litre can be considered as acceptable. A hazard
level is >10 mg/litre, and, at >30 mg/litre, toxic signs and
symptoms can be expected.
Table 2. Health hazards, prevention and protection, first aid
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Readily absorbed
via skin, ingestion, and
inhalation; may cause carbamate
poisoning: excessive sweating,
headache, weakness, giddiness,
nausea, vomiting, stomach pains,
blurred vision, slurred speech,
tremors, and convulsions
SKIN: Irritation; skin rash Wear impervious clothing, gloves, Remove contaminated clothing and shoes;
face shields wash the skin with plenty of water and soap
(or mild detergent); get medical attention
attention immediately
EYES: Irritation Wear face shield or safety goggles Wash eyes immediately with clean water for at
least 15 min, lifting the lower and upper
lids occasionally; if irritation persists, get
medical attention immediately
INHALATION: Irritation Use proper exhaust ventilation Move the exposed person to fresh air at once;
and suitable respiratory protection if breathing has stopped, perform artificial
respiration; keep the affected person warm and
at rest; get medical attention immediately
INGESTION: May lead to severe Do not eat, drink, or smoke when Obtain medical attention immediately; in case
poisoning working with the compound of accidental or intentional ingestion, and if
the patient is still conscious, induce vomiting
immediately: if breathing has stopped, apply
artificial respiration
4.2 Explosion and fire hazards
Technical grade material will not burn, but formulations in organic
solvents may be highly flammable. Use dry powder, carbon dioxide, or
alcohol-resistant foam, sand, or earth for dealing with fires.
If carbaryl is involved in a major fire or in a fire involving other
products, advise the fire service that protective clothing and
breathing apparatus should be worn, because toxic decomposition
products, such as nitrogen oxides, methylamine, and carbon monoxide
may be formed. Also, warn the authorities that carbaryl is toxic for
fish, and that the use of water should be confined to the cooling of
unaffected stock, thus avoiding the accumulation of polluted run-off
from the site.
4.3 Storage
Formulations should be stored in locked buildings, preferably
dedicated to insecticides. They should be kept in clearly labelled,
leakproof containers.
Keep formulations and dilutions out of reach of children and
unauthorized persons. Do not store near foodstuffs or animal feed.
4.4 Transport
Comply with any local requirements regarding the movement of
hazardous goods. Do not load with foodstuffs or animal feed. Before
dispatch, check that containers are sound and labels undamaged.
4.5 Spillage and disposal
4.5.1 Spillage
Avoid exposure, if possible, by the use of appropriate protective
clothing and masks.
Empty any product remaining in damaged or leaking containers into a
clean, empty drum, and label.
Absorb spillage with lime, damp sawdust, sand, or earth and dispose
of safely (see below). If spillage is large, contain it by building
a barrier of earth or sandbags.
Decontaminate empty, damaged, or leaking containers with a 10%
sodium carbonate solution added at the rate of at least 1 litre per
20-litre drum. Puncture and crush containers to prevent reuse.
4.5.2 Disposal
Waste that contains carbaryl should be burned in a high-temperature
incinerator with effluent gas scrubbing.
Where no incinerator is available, contaminated absorbents or
surplus products should be decomposed by hydrolysis at pH12 or
above. Contact with a suitable hydrolysing agent is required to
ensure the degradation of the active ingredient to a safe level. For
emulsifiable material: 5% sodium hydroxide (caustic soda) solution
or saturated (7-10%) sodium carbonate (washing soda) solution can be
used. For non-emulsifiable material, use a 1:1 mixture (by volume)
of either of the above solutions and a water/oil-soluble solvent,
such as denatured alcohol, monoethylene glycol, hexylene glycol, or
isopropanol. Cover the material with a hydrolysing agent and put
aside for 24 h. Before disposal of the resultant waste, the material
must be analysed to ensure that the active ingredient has been
degraded to a safe level.
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage into streams,
watercourses, open waterways, ditches, fields with drainage systems,
or the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
At worst, carbaryl has only limited persistence in the environment.
In water, it is easily degraded under most conditions; the half-life
ranges from a few minutes to several weeks. In soil, the half-life
ranges from a few days to several weeks. Carbaryl is adsorbed on
soil and leaching into water is not of great significance.
There is no evidence of general phytotoxicity when used at
recommended rates, but damage to apples, pears, and water-melons has
been reported. Excessive application may retard the germination of
grasses.
Carbaryl is toxic for earthworms. It is hazardous for honey bees and
it should not be applied to crops during flowering.
Avoid spraying over bodies of water. Do not contaminate ponds,
waterways, or ditches with the product or used containers, as
carbaryl has some toxicity for some aquatic species.
In case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
6.1 Previous evaluations by international bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) evaluated
carbaryl at its meetings in 1963, 1965, 1966, 1967, 1968, 1969,
1970, 1971, 1973, 1975, 1976, 1977, 1979, and 1984. In 1973, an
Acceptable Daily Intake (ADI) of 0-0.01 mg/kg body weight was
established. In addition, Maximum Residue Limits (MRLs) have been
set for specified crops (see Table 3).
The International Agency for Research on Cancer (IARC), evaluated
carbaryl in 1976 and concluded that the available data did not allow
an evaluation of the carcinogenicity of carbaryl to be made.
In the WHO recommended classification of pesticides by hazard,
technical carbaryl is classified in Class II as moderately hazardous
in normal use. WHO has issued a data sheet on carbaryl (No. 3).
6.2 Exposure limit values
Some exposure limit values are given in the Table 3.
6.3 Specific restrictions
Carbaryl is approved for use as a pesticide in many countries, in
each of which, specific uses are defined, as well as limitations and
precautions.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Workplace Argentina Maximum permissible concentration (MPC) 1979
- time-weighted average (TWA) 5 mg/m3
- short-term exposure limit 10 mg/m3
Bulgaria Maximum permissible concentration (MPC) 1 mg/m3 1971
Germany Maximum work-site concentration (MAK) 1986
- 8-h time-weighted averageb (TWA) 5 mg/m3
Hungary Maximum acceptable concentration (MAK) 1 mg/m3 1988
United Occupational exposure standard (OES) 1989
Kingdom - 8-h time-weighted average (TWA) 5 mg/m3
- 10-min short-term exposure level (STEL) 10 mg/m3
USA (OSHA) Permissible exposure limit (PEL) 1989
- 8-h time-weighted average (TWA) 5 mg/m3
USA (NIOSH) Recommended exposure limit (REL)
- time-weighted average (TWA) 5 mg/m3 1976
USA (ACGIH) Threshold limit value (TLV) 1986
- 8-h time weighted average (TWA) 5 mg/m3
USSR Ceiling value (aerosol) 1 mg/m3 1977
WHO Maximum permissible concentrationb 5 mg/m3 1982
(MPC) (tentative)
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration 1977
- average per day 0.01 mg/m3
- once per day 0.02 mg/m3
WATER Surface USSR Maximum allowable concentration (MAC) 0.1 mg/litre 1983
Surface for USSR Maximum allowable concentration (MAC) 0.0005 mg/litre 1983
fishing
SOIL USSR Maximum allowable concentration (MAC) 0.05 mg/kg 1973
FOOD Intake FAO/WHO Acceptable daily intake (ADI) 0-0.01 mg/kg 1973
body weight
Residues FAO/WHO Maximum residue limit (MRL)
(Codex (in specified products): 0.1-100 mg/kg 1986
Alimentarius) animal feedstuffs (green), alfalfa, bean
and pea vines, colver corn, forage
cowpea, foliage, grasses, peanut hay,
sorghum forage, soybean foliage,
sugarbeet tops 100 mg/kg
bran 20 mg/kg
apricots, asparagus, blackberries,
boysenberries, cherries, kiwi fruit
leafy vegetables (except brassicas),
nectarines, nuts (whole), okra, olives
(fresh), peaches, plums, raspberries,
sorghum grain 10 mg/kg
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
blueberries, citrus fruit, cranberries
strawberries 7 mg/kg
apples, aubergines, bananas, barley,
beans, brassicas, grapes, oats, pears,
peas (including pod), peppers, poultry
(skin), rice (in husks and hulled),
rye, tomatoes, wheat 5 mg/kg
cucurbits (including melons) 3 mg/kg
peanuts (whole), root crops (beets,
carrots, parsnips, radishes, rutabagas),
wholemeal flour 2 mg/kg
cottonseed (whole), cowpeas, nuts (shelled),
olives (processed), soybeans, (dry),
sweetcorn (kernels) 1 mg/kg
eggs (shell-free), poultry meat 0.5 mg/kg
meat of cattle, goats and sheep, 0.2 mg/kg
potatoes, sugarbeet, wheat flour (white)
milk and milk products 0.1 mg/kg
a TWA - time-weighted average over one working day (usually 8 h).
b Skin absorption.
6.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transport of
Dangerous Goods classifies carbaryl in:
- Hazard Class 6.1: poisonous substances
- Packing Group III: a substance that has a relatively low risk
of poisoning during transport (solid >30%; liquid >10%).
The following label should be used:
 The European Community legislation requires labelling as a dangerous
substance using the symbol:
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
Harmful by inhalation and if swallowed; irritating to
respiratory system; keep out of reach of children; keep away
from food, drink, and animal feeding stuffs.
Pesticide preparations that contain carbaryl are classified in Class
IIA, i.e., when solid: as harmful at concentrations >5%, and when
liquid as toxic at concentrations >50% and as harmful at 2.5-50%.
Member States should ensure that they cannot be placed on the market
unless their packaging, fastenings, and labels comply with the
requirements laid down.
The FAO specifications for plant protection products containing
carbaryl specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
6.5 Waste disposal
In the USA, permits are required for the discharge of carbaryl from
any point source into national waters. This requirement contains
detailed instructions.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - Classification and
labelling in the European Communities. Vol. 1 & 2. Commission of
the European Communities, London, Graham & Trotman, Ltd.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and
use of pesticides. Rome, Food and Agriculture Organization of the
United Nations.
FAO (1988) Carbaryl, FAO specifications for plant protection
products. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986a) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 3rd ed. Rome, Codex Alimentarius Committee on
Pesticide Residues.
FAO/WHO (1986b) Codex Alimentarius Commission Codex maximum limits
for pesticide residues. Joint FAO/WHO Food Standards Programme,
FAO, CAC Vol. XIII, ed. 2. Rome, Food and Agriculture Organization
of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage, and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of
pesticide poisoning. Brussels, Groupement International des
Associations Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
HAYES, W.J., Jr. & LAWS, E.R., Jr. (1991) Handbook of pesticide
toxicology. 3 vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
for Potentially Toxic Chemicals, United Nations Environment
Programme.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme - Industry and Environment Office, 80 pp.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
UNITED NATIONS (1991) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 4th ed. New York,
United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol., Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 137 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-93. Geneva, World
Health Organization (Unpublished document WHO/PCS/92.14).
WHO (1993) EHC No. 153: Carbaryl. Geneva, World Health
Organization.
WHO/FAO (1978) Data sheets on pesticides: No. 3: Carbaryl.
(Unpublished WHO document VBC/DS/75.3, rev. 1).
WORTHING, C.R. & HANCE, R.J. (1991) The pesticide manual. 9th ed.
Old Woking, Surrey, British Crop Protection Council, Unwin Brothers
Ltd.
ANNEX
Treatment of carbamate pesticide poisoning in man
All cases of carbamate poisoning should be dealt with as an
emergency and the patient should be hospitalized as quickly as
possible.
Extensive descriptions of the treatment of poisoning by
anticholinesterase agents are given in several major references
(Kagan, 1977; Taylor, 1980; Plestina, 1984).
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in the cleaning of the skin area where venupuncture
is performed. Blood might be contaminated with carbamates and
therefore inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting can be induced, if the
patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml of water. However, this treatment is
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with the addition of bicarbonate solution
or activated charcoal) can also be performed, particularly in
unconscious patients, taking care to prevent aspiration of fluids
into the lungs (i.e., only after a tracheal tube has been put in
place).
The volumes of the fluids introduced in the stomach should be
recorded and samples of gastric lavage frozen and stored for
subsequent chemical analysis. If the formulation of the pesticide
involved is available, it should also be stored for further analysis
(i.e., detection of toxicologically relevant impurities). A purge to
remove the ingested compound can be administered.
a From: EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 1968.
General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
Special pharmacological treatment
Atropine
Atropine should be given, beginning with 2 mg iv repeated at 15-
30-min intervals. The dose and the frequency of atropine treatment
vary from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Oxime reactivators
There is no rational basis for using these drugs. Furthermore, some
unconfirmed reports suggest an increased toxicity of carbamates when
oximes have been administered.
Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required.
Other centrally acting drugs and drugs that may depress respiration
are not usually recommended in the absence of artificial respiration
procedures.
References to Annex
KAGAN, J.S. (1977) The toxicity of organophosphorus pesticides.
Moscow (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization.
(Unpublished document VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed.
New York, Macmillan Publishing Co., pp. 100-119.
The label must read:
Harmful by inhalation and if swallowed; irritating to
respiratory system; keep out of reach of children; keep away
from food, drink, and animal feeding stuffs.
Pesticide preparations that contain carbaryl are classified in Class
IIA, i.e., when solid: as harmful at concentrations >5%, and when
liquid as toxic at concentrations >50% and as harmful at 2.5-50%.
Member States should ensure that they cannot be placed on the market
unless their packaging, fastenings, and labels comply with the
requirements laid down.
The FAO specifications for plant protection products containing
carbaryl specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
6.5 Waste disposal
In the USA, permits are required for the discharge of carbaryl from
any point source into national waters. This requirement contains
detailed instructions.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - Classification and
labelling in the European Communities. Vol. 1 & 2. Commission of
the European Communities, London, Graham & Trotman, Ltd.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and
use of pesticides. Rome, Food and Agriculture Organization of the
United Nations.
FAO (1988) Carbaryl, FAO specifications for plant protection
products. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986a) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 3rd ed. Rome, Codex Alimentarius Committee on
Pesticide Residues.
FAO/WHO (1986b) Codex Alimentarius Commission Codex maximum limits
for pesticide residues. Joint FAO/WHO Food Standards Programme,
FAO, CAC Vol. XIII, ed. 2. Rome, Food and Agriculture Organization
of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage, and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of
pesticide poisoning. Brussels, Groupement International des
Associations Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
HAYES, W.J., Jr. & LAWS, E.R., Jr. (1991) Handbook of pesticide
toxicology. 3 vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
for Potentially Toxic Chemicals, United Nations Environment
Programme.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme - Industry and Environment Office, 80 pp.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
UNITED NATIONS (1991) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 4th ed. New York,
United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol., Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 137 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-93. Geneva, World
Health Organization (Unpublished document WHO/PCS/92.14).
WHO (1993) EHC No. 153: Carbaryl. Geneva, World Health
Organization.
WHO/FAO (1978) Data sheets on pesticides: No. 3: Carbaryl.
(Unpublished WHO document VBC/DS/75.3, rev. 1).
WORTHING, C.R. & HANCE, R.J. (1991) The pesticide manual. 9th ed.
Old Woking, Surrey, British Crop Protection Council, Unwin Brothers
Ltd.
ANNEX
Treatment of carbamate pesticide poisoning in man
All cases of carbamate poisoning should be dealt with as an
emergency and the patient should be hospitalized as quickly as
possible.
Extensive descriptions of the treatment of poisoning by
anticholinesterase agents are given in several major references
(Kagan, 1977; Taylor, 1980; Plestina, 1984).
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in the cleaning of the skin area where venupuncture
is performed. Blood might be contaminated with carbamates and
therefore inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting can be induced, if the
patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml of water. However, this treatment is
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with the addition of bicarbonate solution
or activated charcoal) can also be performed, particularly in
unconscious patients, taking care to prevent aspiration of fluids
into the lungs (i.e., only after a tracheal tube has been put in
place).
The volumes of the fluids introduced in the stomach should be
recorded and samples of gastric lavage frozen and stored for
subsequent chemical analysis. If the formulation of the pesticide
involved is available, it should also be stored for further analysis
(i.e., detection of toxicologically relevant impurities). A purge to
remove the ingested compound can be administered.
a From: EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 1968.
General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
Special pharmacological treatment
Atropine
Atropine should be given, beginning with 2 mg iv repeated at 15-
30-min intervals. The dose and the frequency of atropine treatment
vary from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Oxime reactivators
There is no rational basis for using these drugs. Furthermore, some
unconfirmed reports suggest an increased toxicity of carbamates when
oximes have been administered.
Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required.
Other centrally acting drugs and drugs that may depress respiration
are not usually recommended in the absence of artificial respiration
procedures.
References to Annex
KAGAN, J.S. (1977) The toxicity of organophosphorus pesticides.
Moscow (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization.
(Unpublished document VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed.
New York, Macmillan Publishing Co., pp. 100-119.
 Molecular formula: C12H11NO2
Common name: Carbaryl (BSI)
CAS chemical name: 1-naphthalenyl methylcarbamate
IUPAC chemical name: 1-naphthyl methylcarbamate
Trade names: Arylat, Arylam, Atoxan, Bercema,
Carbaryl, Carbamate, Carpolin,
Carbotox, Denapon, Dicarbam, Hexavin,
Monsur, Murrin, Panam, Pamex,
Prosevor, Rayvon, Sevimol, Sevin,
Viaxon, etc.
CAS registry number:
Molecular formula: C12H11NO2
Common name: Carbaryl (BSI)
CAS chemical name: 1-naphthalenyl methylcarbamate
IUPAC chemical name: 1-naphthyl methylcarbamate
Trade names: Arylat, Arylam, Atoxan, Bercema,
Carbaryl, Carbamate, Carpolin,
Carbotox, Denapon, Dicarbam, Hexavin,
Monsur, Murrin, Panam, Pamex,
Prosevor, Rayvon, Sevimol, Sevin,
Viaxon, etc.
CAS registry number:  The European Community legislation requires labelling as a dangerous
substance using the symbol:
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
Harmful by inhalation and if swallowed; irritating to
respiratory system; keep out of reach of children; keep away
from food, drink, and animal feeding stuffs.
Pesticide preparations that contain carbaryl are classified in Class
IIA, i.e., when solid: as harmful at concentrations >5%, and when
liquid as toxic at concentrations >50% and as harmful at 2.5-50%.
Member States should ensure that they cannot be placed on the market
unless their packaging, fastenings, and labels comply with the
requirements laid down.
The FAO specifications for plant protection products containing
carbaryl specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
6.5 Waste disposal
In the USA, permits are required for the discharge of carbaryl from
any point source into national waters. This requirement contains
detailed instructions.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - Classification and
labelling in the European Communities. Vol. 1 & 2. Commission of
the European Communities, London, Graham & Trotman, Ltd.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and
use of pesticides. Rome, Food and Agriculture Organization of the
United Nations.
FAO (1988) Carbaryl, FAO specifications for plant protection
products. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986a) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 3rd ed. Rome, Codex Alimentarius Committee on
Pesticide Residues.
FAO/WHO (1986b) Codex Alimentarius Commission Codex maximum limits
for pesticide residues. Joint FAO/WHO Food Standards Programme,
FAO, CAC Vol. XIII, ed. 2. Rome, Food and Agriculture Organization
of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage, and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of
pesticide poisoning. Brussels, Groupement International des
Associations Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
HAYES, W.J., Jr. & LAWS, E.R., Jr. (1991) Handbook of pesticide
toxicology. 3 vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
for Potentially Toxic Chemicals, United Nations Environment
Programme.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme - Industry and Environment Office, 80 pp.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
UNITED NATIONS (1991) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 4th ed. New York,
United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol., Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 137 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-93. Geneva, World
Health Organization (Unpublished document WHO/PCS/92.14).
WHO (1993) EHC No. 153: Carbaryl. Geneva, World Health
Organization.
WHO/FAO (1978) Data sheets on pesticides: No. 3: Carbaryl.
(Unpublished WHO document VBC/DS/75.3, rev. 1).
WORTHING, C.R. & HANCE, R.J. (1991) The pesticide manual. 9th ed.
Old Woking, Surrey, British Crop Protection Council, Unwin Brothers
Ltd.
ANNEX
Treatment of carbamate pesticide poisoning in man
All cases of carbamate poisoning should be dealt with as an
emergency and the patient should be hospitalized as quickly as
possible.
Extensive descriptions of the treatment of poisoning by
anticholinesterase agents are given in several major references
(Kagan, 1977; Taylor, 1980; Plestina, 1984).
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in the cleaning of the skin area where venupuncture
is performed. Blood might be contaminated with carbamates and
therefore inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting can be induced, if the
patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml of water. However, this treatment is
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with the addition of bicarbonate solution
or activated charcoal) can also be performed, particularly in
unconscious patients, taking care to prevent aspiration of fluids
into the lungs (i.e., only after a tracheal tube has been put in
place).
The volumes of the fluids introduced in the stomach should be
recorded and samples of gastric lavage frozen and stored for
subsequent chemical analysis. If the formulation of the pesticide
involved is available, it should also be stored for further analysis
(i.e., detection of toxicologically relevant impurities). A purge to
remove the ingested compound can be administered.
a From: EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 1968.
General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
Special pharmacological treatment
Atropine
Atropine should be given, beginning with 2 mg iv repeated at 15-
30-min intervals. The dose and the frequency of atropine treatment
vary from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Oxime reactivators
There is no rational basis for using these drugs. Furthermore, some
unconfirmed reports suggest an increased toxicity of carbamates when
oximes have been administered.
Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required.
Other centrally acting drugs and drugs that may depress respiration
are not usually recommended in the absence of artificial respiration
procedures.
References to Annex
KAGAN, J.S. (1977) The toxicity of organophosphorus pesticides.
Moscow (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization.
(Unpublished document VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed.
New York, Macmillan Publishing Co., pp. 100-119.
The label must read:
Harmful by inhalation and if swallowed; irritating to
respiratory system; keep out of reach of children; keep away
from food, drink, and animal feeding stuffs.
Pesticide preparations that contain carbaryl are classified in Class
IIA, i.e., when solid: as harmful at concentrations >5%, and when
liquid as toxic at concentrations >50% and as harmful at 2.5-50%.
Member States should ensure that they cannot be placed on the market
unless their packaging, fastenings, and labels comply with the
requirements laid down.
The FAO specifications for plant protection products containing
carbaryl specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
6.5 Waste disposal
In the USA, permits are required for the discharge of carbaryl from
any point source into national waters. This requirement contains
detailed instructions.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - Classification and
labelling in the European Communities. Vol. 1 & 2. Commission of
the European Communities, London, Graham & Trotman, Ltd.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and
use of pesticides. Rome, Food and Agriculture Organization of the
United Nations.
FAO (1988) Carbaryl, FAO specifications for plant protection
products. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986a) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 3rd ed. Rome, Codex Alimentarius Committee on
Pesticide Residues.
FAO/WHO (1986b) Codex Alimentarius Commission Codex maximum limits
for pesticide residues. Joint FAO/WHO Food Standards Programme,
FAO, CAC Vol. XIII, ed. 2. Rome, Food and Agriculture Organization
of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage, and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of
pesticide poisoning. Brussels, Groupement International des
Associations Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
HAYES, W.J., Jr. & LAWS, E.R., Jr. (1991) Handbook of pesticide
toxicology. 3 vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
for Potentially Toxic Chemicals, United Nations Environment
Programme.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme - Industry and Environment Office, 80 pp.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
UNITED NATIONS (1991) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 4th ed. New York,
United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol., Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 137 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-93. Geneva, World
Health Organization (Unpublished document WHO/PCS/92.14).
WHO (1993) EHC No. 153: Carbaryl. Geneva, World Health
Organization.
WHO/FAO (1978) Data sheets on pesticides: No. 3: Carbaryl.
(Unpublished WHO document VBC/DS/75.3, rev. 1).
WORTHING, C.R. & HANCE, R.J. (1991) The pesticide manual. 9th ed.
Old Woking, Surrey, British Crop Protection Council, Unwin Brothers
Ltd.
ANNEX
Treatment of carbamate pesticide poisoning in man
All cases of carbamate poisoning should be dealt with as an
emergency and the patient should be hospitalized as quickly as
possible.
Extensive descriptions of the treatment of poisoning by
anticholinesterase agents are given in several major references
(Kagan, 1977; Taylor, 1980; Plestina, 1984).
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in the cleaning of the skin area where venupuncture
is performed. Blood might be contaminated with carbamates and
therefore inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting can be induced, if the
patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml of water. However, this treatment is
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with the addition of bicarbonate solution
or activated charcoal) can also be performed, particularly in
unconscious patients, taking care to prevent aspiration of fluids
into the lungs (i.e., only after a tracheal tube has been put in
place).
The volumes of the fluids introduced in the stomach should be
recorded and samples of gastric lavage frozen and stored for
subsequent chemical analysis. If the formulation of the pesticide
involved is available, it should also be stored for further analysis
(i.e., detection of toxicologically relevant impurities). A purge to
remove the ingested compound can be administered.
a From: EHC 64: Carbamate pesticides - a general introduction.
Geneva, World Health Organization, 1968.
General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
Special pharmacological treatment
Atropine
Atropine should be given, beginning with 2 mg iv repeated at 15-
30-min intervals. The dose and the frequency of atropine treatment
vary from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Oxime reactivators
There is no rational basis for using these drugs. Furthermore, some
unconfirmed reports suggest an increased toxicity of carbamates when
oximes have been administered.
Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required.
Other centrally acting drugs and drugs that may depress respiration
are not usually recommended in the absence of artificial respiration
procedures.
References to Annex
KAGAN, J.S. (1977) The toxicity of organophosphorus pesticides.
Moscow (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization.
(Unpublished document VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed.
New York, Macmillan Publishing Co., pp. 100-119.
