
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 17
ENDOSULFAN
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA
This is a companion volume to Environmental Health Criteria 40:
Endosulfan
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
ISBN 92 4 154340 X
ISSN 0259 - 7268
(c) World Health Organization 1988
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Endosulfan toxicity
2.2. Human exposure to endosulfan
2.3. Evaluation of health risks for man
2.4. Fate in the environment
2.5. Evaluation of effects on the environment
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.2. Recommendations
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Main human health hazards, prevention and
protection, first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 Medical advice
4.1.2. Health surveillance advice
4.2. Safety in use
4.3. Explosion and fire hazards
4.3.1. Explosion hazards
4.3.2. Fire hazards
4.4. Storage
4.4.1. Leaking containers in store
4.5. Transport
4.6. Spillage and disposal
4.6.1. Spillage
4.6.1.1 Solid products
4.6.1.2 Liquid products
4.6.1.3 All products
4.6.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. INTERNATIONAL CHEMICAL SAFETY CARD
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Waste disposal
7.6. Other measures
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is an International Chemical Safety Card
which should be readily available, and should be clearly explained, to
all who could come into contact with the chemical. The section on
regulatory information has been extracted from the legal file of the
International Register of Potentially Toxic Chemicals (IRPTC) and from
other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: Endosulfan (ISO, BSO, ANSI, ESA),
benzoepin (JMAF)
Chemical structure:
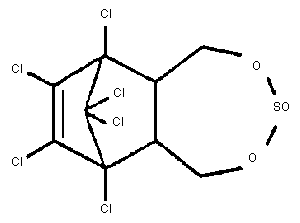 Molecular formula: C9H6Cl6O3S
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,
9,-9a-hexahydro-6,9-methano-2,4,
3-benzodioxathiepin-3-oxide.
Trade names: Beosit, Malix, Thiodan, Thiofor,
Thiomul, Thionex, Thimul
Formulations under other trade names may
also exist
Development codes: HOE 002671, NIA 5462
CAS registry number: 115-29-7
RTECS registry number: RB 9275000
Relative molecular mass: 406.9
Technical endosulfan is a brown crystalline substance consisting of
alpha- and beta-isomers in the ratio of approximately 70:30. Its
purity is 94-96%.
1.2 Physical and Chemical Properties
Pure grade endosulfan is a colourless crystalline solid. Its
solubility in water is low, being less than 0.5 mg/litre at 20°C, but
increases with decreasing pH. Solubility in other solvents varies
from 5-65%.
Technical endosulfan is usually sold in the form of brown crystalline
flakes with a faint odour of sulfur dioxide. It is stable to
sunlight, sensitive to moisture, unstable in alkaline media, and
subject to slow hydrolysis.
Endosulfan is available as an emulsifiable concentrate, water
dispersible powder, dispersion, dust, granules, and as an
ultra-low-volume (ULV) formulation.
Some physical and chemical properties of pure and technical endosulfan
are listed in the International Chemical Safety Card on pages 22-25.
1.3 Analytical Methods
Standard analytical methods for the determination of endosulfan in the
technical product, and in formulations, include saponification and
titration of the sulfur dioxide formed with iodine, or gas
chromatography. The method of choice for the determination of
residues is gas chromatography with electron capture detection. In
considering residue levels, the sum of the alpha- and beta-isomers
plus the metabolite endosulfan sulfate, which is similar in toxicity
to the parent compound, have to be considered.
1.4 Production and Uses
Present world production of technical grade endosulfan is estimated to
be of the order of 10 000 tonnes per year.
Endosulfan is used in a formulation as a non-systemic contact and
stomach insecticide, mainly in agriculture, in the control of the
tsetse fly, and in the control of home garden pests.
Endosulfan controls a wide range of sucking and chewing insect pests,
notably of the orders of Lepidoptera, Coleoptera, Heteroptera,
Homoptera, Thysanoptera, Diptera, and some species belonging to the
order of Acarina. It is especially used on non-food crops, such as
cotton and tobacco, and on food crops, such as vegetables, fruits,
corn, cereals, oilseeds, potatoes, tea, and coffee. It is also used
on numerous other crops.
2. SUMMARY AND EVALUATION
2.1 Endosulfan toxicity
WHO (1986) classified endosulfan in the category of technical products
that are moderately hazardous, based on an oral LD50 in the rat of
80 mg/kg body weight.
Endosulfan can be absorbed following ingestion, inhalation, or skin
contact. It is readily metabolized and eliminated and does not
accumulate in the body.
Acute intoxication may result in neurological manifestations, such as
irritability, restlessness, muscular twitchings, and convulsions that
may end in death.
The long-term, no-observed-adverse-effect level in rats was 30 mg/kg
diet (equivalent to 1.5 mg/kg body weight) and 0.75 mg/kg body weight
in dogs. At higher dose levels, testicular and renal damage occurred
in the rat.
Endosulfan was essentially negative in short-term tests for genetic
activity. It did not show any carcinogenic activity in mice or rats,
but studies were limited by inadequate reporting or survival.a
Several cases of suicidal and occupational poisoning have been
reported, the latter resulting, in most cases, from neglecting safety
precautions.
2.2 Human exposure to endosulfan
Food is the main source of exposure of the general population to
endosulfan. Endosulfan residues in food (the sum of its alpha- and
beta-isomers and endosulfan sulfate) have been found to be generally
well below FAO/WHO maximum residue limits.
In occupationally-exposed persons, both skin contact and inhalation
can be important routes of absorption, when adequate safety
precautions are not taken.
a Adequate proprietary data on reproductive toxicity,
teratogenicity, and neurotoxicity have become available since
this evaluation was made in 1984; new carcinogenicity studies on
the rat and the mouse are in progress.
2.3 Evaluation of Health Risks for Man
The main hazard associated with endosulfan is acute intoxication
through overexposure. Such situations may be due to intentional or
accidental overexposure or to gross negligence in occupational
situations.
In all other exposure situations, especially as far as the general
population is concerned, the toxicity profile and the present exposure
pattern do not indicate any appreciable hazard.
2.4 Fate in the Environment
Both endosulfan isomers are fairly resistant to photodegradation, but
the metabolites endosulfan sulfate and endosulfan diol are susceptible
to photolysis. The half-life of endosulfan in water is estimated to
be 4 days, but anaerobic conditions and/or a low pH will lengthen the
half-life. In water, it is mainly degraded to endosulfan diol.
In soil, the alpha-isomer disappears more rapidly than the
beta-isomer. Endosulfan sulfate, the major degradation product in
soil, is relatively persistent. These compounds are not prone to
leaching.
Biodegradation in soil and water is dependent on climatic conditions
and on the types of microorganism present.
2.5 Evaluation of Effects on the Environment
Endosulfan does not appear to be a problem with regard to persistence
in biota. It is not readily bioaccumulated. In aquatic organisms,
loss soon balances uptake and a fairly low plateau level of residues
is achieved.
Fish are extremely sensitive to endosulfan and the killing of fish has
been reported as a result of the discharge of endosulfan into rivers.
Agricultural run-off has not caused such a problem. However,
application of endosulfan to wetlands at recommended rates may well
result in the killing of fish. Large-scale field experience with
endosulfan has not resulted in any long-term adverse effects on the
environment.
Because there is little or no biomagnification, endosulfan applied at
recommended rates is not hazardous for terrestrial animals. Toxicity
for bees is low to moderate. Toxicity for birds is high in a
laboratory setting, but no poisonings have been reported under field
conditions.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
(a) The general population does not appear to be at risk from
endosulfan residues in food. Exposure of the general population
via air and drinking-water is generally low.
(b) Occupational exposure has resulted in some incidents of
poisoning. These appear to have occurred, however, only when
adequate safety precautions were not taken.
(c) In terms of the general environment, endosulfan is highly toxic
for some aquatic species, particularly fish. Endosulfan is
moderately toxic for honey bees.
(d) Endosulfan does not accumulate in food-chains and is eliminated
rapidly from the body.
3.2 Recommendations
(a) Precautions should be taken to avoid contamination of surface and
drinking-water supplies during spraying. Where necessary,
residue levels of endosulfan in drinking-water should be reduced
by proper water treatment.
(b) In countries where endosulfan is used for tsetse fly control,
exposed populations should be monitored for potential adverse
health effects.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First
Aid
Endosulfan is an organochlorine insecticide. It is toxic and can be
hazardous for human beings if incorrectly or carelessly handled. It
is therefore essential that the correct precautions are observed in
handling and use. For details see the International Chemical Safety
Card on pages 22-25.
4.1.1 Advice to Physicians
4.1.1.1 Symptoms of poisoning
Endosulfan is hazardous by mouth, by skin contact (especially liquid
formulations) and, to a lesser extent, by inhalation. It acts as a
stimulant of the central nervous system.
Following accidental ingestion or gross overexposure, symptoms may
include headache, dizziness, nausea, vomiting, weakness in the legs,
and convulsions, sometimes leading to death.
4.1.1.2 Medical advice
Medical treatment is largely symptomatic and supportive and directed
against convulsions and anoxaemia.
If endosulfan is swallowed, vomiting should not be induced and emetics
are contraindicated, because many liquid formulations contain
hydrocarbons and there is a risk of aspiration pneumonia. Instead,
the stomach should be emptied as soon as possible by careful gastric
lavage (with a cuffed endotracheal tube already in place), avoiding
aspiration into the lungs. This should be followed by intragastric
administration of 3-4 tablespoons of activated charcoal and 30 g
magnesium or sodium sulfate in a 30% aqueous solution. Oily
purgatives are contraindicated. No fats, oils, or milk should be
given.
If convulsions occur, anticonvulsants should be given immediately,
e.g., 10 mg of diazepam, slowly, intravenously (children 1-5 mg),
repeated as necessary; or thiopental sodium or hexobarbital sodium
slowly, intravenously, in a dose of 10 mg/kg with a maximum total dose
of up to 750 mg for an adult, or paraldehyde 5 ml by intramuscular
injection. These short-acting anticonvulsants should always be
followed by phenobarbital given orally at 3 mg/kg (up to 200 mg for an
adult), or phenobarbital sodium given intramuscularly at 3 mg/kg (also
up to 200 mg for an adult).
Morphine and its derivatives, epinephrine, and noradrenaline should
never be given.
An unobstructed airway must be maintained. Respiratory inadequacy,
which may be accentuated by barbiturate anticonvulsants, should be
corrected and oxygen and/or artificial ventilation may be needed.
4.1.2 Health Surveillance Advice
A complete medical history and physical examination of regularly
exposed workers should be made on an annual basis.
4.2 Safety in Use
Handling liquid formulations: Wear protective neoprene or PVC
gloves, cotton overalls, rubber
boots, and face shield.
Handling powder formulations: Avoid raising a dust cloud. Wear
protective gloves and dust mask.
Follow the advice relating to
personal hygiene.
Application in the field
Aerial application: Ensure that flag-men (markers) do
not stand in the spray-path of the
aircraft; do not spray over surface
waters and avoid spraying over
ditches, canals, rivers, streams,
ponds, or lakes.
Ground spraying: Wear suitable protective clothing
(i.e., cap or hat, cotton overalls
or long-sleeved cotton shirt and
long trousers, boots or shoes);
when spraying tall crops or when
there is a risk of accidental
contamination by the spray, also
wear impermeable hood and jacket;
at all times avoid exposure to the
spray mist; do not spray into the
wind.
Hand-held ULV application: Wear suitable protective clothing
(i.e., cap or hat, cotton overalls
or long-sleeved cotton shirt and
long trousers, boots or shoes; read
and observe the instructions that
apply to the equipment being used;
pay proper attention to wind speed
and direction; always spray in a
down-wind direction; do not spray
if there are other people
immediately down-wind.
After application: Take off heavily splashed or
contaminated clothing; wash hands
and exposed skin before eating,
drinking, or smoking; wash
overalls, boots, hat, and other
protective clothing thoroughly,
especially the inside of gloves;
keep application equipment in good
condition, and free from leaks and
external contamination; keep
contents tightly closed in original
labelled container when not fully
used; do not reuse empty container
for any other purpose; keep
container in a safe place away from
food, children, and animals; empty
containers must be washed out and
disposed of as advised in sections
4.4.1 and 4.6.2.
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
The explosion hazard will depend on the solvent used in the
formulation, or on the characteristics of the dust.
4.3.2 Fire hazards
Liquid formulations containing organic solvents may be flammable.
Extinguish fires with alcohol-resistant foam, carbon dioxide, or
powder. With sufficient burning or external heat, endosulfan will
decompose, emitting toxic fumes. Fire-fighters should be equipped
with self-contained breathing apparatus, eye protection, and full
protective clothing.
The use of water spray should be confined to the cooling of unaffected
stock, thus avoiding the accumulation of polluted run-off from the
site.
4.4 Storage
Products should be stored in locked buildings -- preferably buildings
dedicated to insecticides.
Keep the products out of reach of children and unauthorized personnel.
Do not store near foodstuffs or animal feed.
4.4.1 Leaking containers in store
Take precautions and use appropriate personal protection (section
4.2). Empty any product remaining in damaged/leaking containers into
a clean empty drum, which should then be tightly closed and suitably
labelled.
Sweep up spillage with sawdust, sand, or earth (moisten for powders),
and dispose of safely.
After emptying, the leaking containers should be rinsed with at least
1 litre water per 20-litre drum. Swirl round to rinse the walls,
empty, and add the rinsings to the sawdust or earth. Puncture the
container to prevent reuse.
4.5 Transport
Comply with any local requirements regarding movement of hazardous
goods. Do not transport in the same compartment as foodstuffs. Check
that containers are sound and labels undamaged before despatch.
4.6 Spillage and Disposal
4.6.1 Spillage
Before dealing with any spillage, precautions should be taken as
required, and appropriate personal protection should be used (section
4.2).
4.6.1.1 Solid products
Sweep up and absorb remaining spilled product with moist sawdust,
sand, or earth, and transfer the sweepings in a suitable container to
a safe place for disposal.
4.6.1.2 Liquid products
Prevent the liquid from spreading or contaminating other cargo,
vegetation, or waterways, by making a barrier of the most suitable
available material, e.g., earth or sand.
Absorb spilled liquid with sawdust, sand, or earth, sweep up and place
it in a closeable container for later transfer to a safe place for
disposal.
4.6.1.3 All products
As soon as possible after the spillage and before reuse, cover all
contaminated areas with damp sawdust, sand, or earth. Sweep up and
place in a closeable container for later transfer to a safe place for
disposal. Since this insecticide is highly toxic for fish, care
should be taken to avoid run-off into water courses.
4.6.2 Disposal
Any surplus product, contaminated absorbents, and containers should be
disposed of in an appropriate way. Waste material should be burned in
a proper incinerator designed for organochlorine waste disposal
(1000°C and 30 min residence time with effluent gas scrubbing). If
this is not possible, bury in an approved dump or landfill where there
is no risk of contamination of surface or ground water. Comply with
any local legislation regarding disposal of toxic wastes. Puncture
container to prevent reuse.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Endosulfan is highly toxic for fish and some other aquatic organisms,
and moderately toxic for honey bees. It is not readily bioaccumulated
and it does not persist in the environment.
Industrial discharges from manufacture, formulation, and technical
applications should not be allowed to pollute the environment and
should be properly treated (section 4.6.2).
Any spillage or unused product should be prevented from spreading to
vegetation or waterways and should be treated and disposed of
properly.
6. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers concerned
with, and users of, endosulfan. It should be displayed at, or near,
entrances to areas where there is potential exposure to endosulfan,
and on processing equipment and containers. The card should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions on the
chemical safety card clearly explained. Space is available on the
card for insertion of the National Occupational Exposure Limit, the
address and telephone number of the National Poison Control Centre,
and for local trade names.
ENDOSULFAN
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,9,9a-hexahydro-6,9-methano-2,4,3-benzodioxathiepin-3-oxide
Molecular formula: C9H6Cl6O3S
RTECS registry number: RB 9275000 CAS Registry number: 115-29-7
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Pure substance:
Melting point of alpha-isomer 109.2°C Endosulfan is a non-systemic contact and stomach
of beta-isomer 213.3°C insecticide effective against a wide range of sucking and
Relative density at 20°C 1.745 chewing insect pests in agriculture, and public health
Vapour pressure <0.1 µPa at 20°C and technical fields of applications
<1.4 µPa at 40°C
Relative molecular mass 406.9
Solubility in water 0.5 mg/litre at 20°C Formulations include:
in most organic emulsifiable concentrates water dispersible powders,
solvents 5-65 g/100 ml at 20°C dispersions, dusts, granules, and ultra-low-volume
formulations
Technical grade:
Melting point 80°C
Appearance brown crystalline flakes
Odour faint odour of sulfur
dioxide
Ratio of isomers alpha- to beta-endosulfan
approx. 70:30
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: may cause poisoning in Avoid contact with skin; After contact with skin, wash immediately
contact with skin wear suitable protective with plenty of water and soap; immediately
clothing and gloves remove all contaminated clothing
EYES: may cause irritation to Avoid contact with eyes; wear In case of contact with eyes, rinse
eyes eye protection immediately with plenty of water and seek
medical advice
INHALATION: dusts may cause Do not breathe dusts or sprays
poisoning by inhalation
INGESTION: unlikely occupational Do not eat, drink or smoke during
hazard work; wash hands before eating,
drinking or smoking
Accidental or intentional ingestion If swallowed, seek medical advice
may cause poisoning immediately and show container or label;
do not induce vomiting where organic
solvents are present in emulsifiable
concentrates; keep at rest, lying face
downwards, and ensure a clear airway
ENVIRONMENT: Dangerous for Do not contaminate surface waters
aquatic life, particularly fish
SPILLAGE STORAGE FIRE AND EXPLOSION
Take appropriate personal Products should be stored in Liquid products containing organic solvents
precautions; prevent liquid locked buildings preferably may be flammable. Extinguish fires with
from spreading or contaminating dedicated to insecticides; keep alcohol-resistant foam, CO2 or powder.
other cargo, vegetation, or surface out of reach of children; keep With sufficient burning or external heat
waters and drainage systems, away from food, drink and animal endosulfan will decompose, emitting toxic
with a barrier of most suitable feeding stuffs fumes. Fire-fighters should be equipped
material, e.g., earth or sand with self-contained breathing apparatus,
eye protection, and full protective clothing;
Absorb spilled liquid with confine the use of water spray to cooling of
sawdust, sand, or earth; sweep up and unaffected stock, thus avoiding the
place it in a closeable container accumulation of polluted run-off from the site
for later safe disposal.
Since this insecticide is dangerous
for fish, care should be taken to
avoid run-off into water courses
WASTE DISPOSAL NATIONAL INFORMATION
Endosulfan waste material should National Occupational Exposure UN No. 2761, 2762, 2995, 2996
be burned in a proper incinerator Limit:
designed for organochlorine waste
disposal; if this is not possible, National Poison Control Centre:
bury in an approved dump or
landfill where there is no risk of Local Trade Names:
contamination of surface or ground
water; comply with any local
legislation regarding disposal of
toxic wastes
Molecular formula: C9H6Cl6O3S
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,
9,-9a-hexahydro-6,9-methano-2,4,
3-benzodioxathiepin-3-oxide.
Trade names: Beosit, Malix, Thiodan, Thiofor,
Thiomul, Thionex, Thimul
Formulations under other trade names may
also exist
Development codes: HOE 002671, NIA 5462
CAS registry number: 115-29-7
RTECS registry number: RB 9275000
Relative molecular mass: 406.9
Technical endosulfan is a brown crystalline substance consisting of
alpha- and beta-isomers in the ratio of approximately 70:30. Its
purity is 94-96%.
1.2 Physical and Chemical Properties
Pure grade endosulfan is a colourless crystalline solid. Its
solubility in water is low, being less than 0.5 mg/litre at 20°C, but
increases with decreasing pH. Solubility in other solvents varies
from 5-65%.
Technical endosulfan is usually sold in the form of brown crystalline
flakes with a faint odour of sulfur dioxide. It is stable to
sunlight, sensitive to moisture, unstable in alkaline media, and
subject to slow hydrolysis.
Endosulfan is available as an emulsifiable concentrate, water
dispersible powder, dispersion, dust, granules, and as an
ultra-low-volume (ULV) formulation.
Some physical and chemical properties of pure and technical endosulfan
are listed in the International Chemical Safety Card on pages 22-25.
1.3 Analytical Methods
Standard analytical methods for the determination of endosulfan in the
technical product, and in formulations, include saponification and
titration of the sulfur dioxide formed with iodine, or gas
chromatography. The method of choice for the determination of
residues is gas chromatography with electron capture detection. In
considering residue levels, the sum of the alpha- and beta-isomers
plus the metabolite endosulfan sulfate, which is similar in toxicity
to the parent compound, have to be considered.
1.4 Production and Uses
Present world production of technical grade endosulfan is estimated to
be of the order of 10 000 tonnes per year.
Endosulfan is used in a formulation as a non-systemic contact and
stomach insecticide, mainly in agriculture, in the control of the
tsetse fly, and in the control of home garden pests.
Endosulfan controls a wide range of sucking and chewing insect pests,
notably of the orders of Lepidoptera, Coleoptera, Heteroptera,
Homoptera, Thysanoptera, Diptera, and some species belonging to the
order of Acarina. It is especially used on non-food crops, such as
cotton and tobacco, and on food crops, such as vegetables, fruits,
corn, cereals, oilseeds, potatoes, tea, and coffee. It is also used
on numerous other crops.
2. SUMMARY AND EVALUATION
2.1 Endosulfan toxicity
WHO (1986) classified endosulfan in the category of technical products
that are moderately hazardous, based on an oral LD50 in the rat of
80 mg/kg body weight.
Endosulfan can be absorbed following ingestion, inhalation, or skin
contact. It is readily metabolized and eliminated and does not
accumulate in the body.
Acute intoxication may result in neurological manifestations, such as
irritability, restlessness, muscular twitchings, and convulsions that
may end in death.
The long-term, no-observed-adverse-effect level in rats was 30 mg/kg
diet (equivalent to 1.5 mg/kg body weight) and 0.75 mg/kg body weight
in dogs. At higher dose levels, testicular and renal damage occurred
in the rat.
Endosulfan was essentially negative in short-term tests for genetic
activity. It did not show any carcinogenic activity in mice or rats,
but studies were limited by inadequate reporting or survival.a
Several cases of suicidal and occupational poisoning have been
reported, the latter resulting, in most cases, from neglecting safety
precautions.
2.2 Human exposure to endosulfan
Food is the main source of exposure of the general population to
endosulfan. Endosulfan residues in food (the sum of its alpha- and
beta-isomers and endosulfan sulfate) have been found to be generally
well below FAO/WHO maximum residue limits.
In occupationally-exposed persons, both skin contact and inhalation
can be important routes of absorption, when adequate safety
precautions are not taken.
a Adequate proprietary data on reproductive toxicity,
teratogenicity, and neurotoxicity have become available since
this evaluation was made in 1984; new carcinogenicity studies on
the rat and the mouse are in progress.
2.3 Evaluation of Health Risks for Man
The main hazard associated with endosulfan is acute intoxication
through overexposure. Such situations may be due to intentional or
accidental overexposure or to gross negligence in occupational
situations.
In all other exposure situations, especially as far as the general
population is concerned, the toxicity profile and the present exposure
pattern do not indicate any appreciable hazard.
2.4 Fate in the Environment
Both endosulfan isomers are fairly resistant to photodegradation, but
the metabolites endosulfan sulfate and endosulfan diol are susceptible
to photolysis. The half-life of endosulfan in water is estimated to
be 4 days, but anaerobic conditions and/or a low pH will lengthen the
half-life. In water, it is mainly degraded to endosulfan diol.
In soil, the alpha-isomer disappears more rapidly than the
beta-isomer. Endosulfan sulfate, the major degradation product in
soil, is relatively persistent. These compounds are not prone to
leaching.
Biodegradation in soil and water is dependent on climatic conditions
and on the types of microorganism present.
2.5 Evaluation of Effects on the Environment
Endosulfan does not appear to be a problem with regard to persistence
in biota. It is not readily bioaccumulated. In aquatic organisms,
loss soon balances uptake and a fairly low plateau level of residues
is achieved.
Fish are extremely sensitive to endosulfan and the killing of fish has
been reported as a result of the discharge of endosulfan into rivers.
Agricultural run-off has not caused such a problem. However,
application of endosulfan to wetlands at recommended rates may well
result in the killing of fish. Large-scale field experience with
endosulfan has not resulted in any long-term adverse effects on the
environment.
Because there is little or no biomagnification, endosulfan applied at
recommended rates is not hazardous for terrestrial animals. Toxicity
for bees is low to moderate. Toxicity for birds is high in a
laboratory setting, but no poisonings have been reported under field
conditions.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
(a) The general population does not appear to be at risk from
endosulfan residues in food. Exposure of the general population
via air and drinking-water is generally low.
(b) Occupational exposure has resulted in some incidents of
poisoning. These appear to have occurred, however, only when
adequate safety precautions were not taken.
(c) In terms of the general environment, endosulfan is highly toxic
for some aquatic species, particularly fish. Endosulfan is
moderately toxic for honey bees.
(d) Endosulfan does not accumulate in food-chains and is eliminated
rapidly from the body.
3.2 Recommendations
(a) Precautions should be taken to avoid contamination of surface and
drinking-water supplies during spraying. Where necessary,
residue levels of endosulfan in drinking-water should be reduced
by proper water treatment.
(b) In countries where endosulfan is used for tsetse fly control,
exposed populations should be monitored for potential adverse
health effects.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First
Aid
Endosulfan is an organochlorine insecticide. It is toxic and can be
hazardous for human beings if incorrectly or carelessly handled. It
is therefore essential that the correct precautions are observed in
handling and use. For details see the International Chemical Safety
Card on pages 22-25.
4.1.1 Advice to Physicians
4.1.1.1 Symptoms of poisoning
Endosulfan is hazardous by mouth, by skin contact (especially liquid
formulations) and, to a lesser extent, by inhalation. It acts as a
stimulant of the central nervous system.
Following accidental ingestion or gross overexposure, symptoms may
include headache, dizziness, nausea, vomiting, weakness in the legs,
and convulsions, sometimes leading to death.
4.1.1.2 Medical advice
Medical treatment is largely symptomatic and supportive and directed
against convulsions and anoxaemia.
If endosulfan is swallowed, vomiting should not be induced and emetics
are contraindicated, because many liquid formulations contain
hydrocarbons and there is a risk of aspiration pneumonia. Instead,
the stomach should be emptied as soon as possible by careful gastric
lavage (with a cuffed endotracheal tube already in place), avoiding
aspiration into the lungs. This should be followed by intragastric
administration of 3-4 tablespoons of activated charcoal and 30 g
magnesium or sodium sulfate in a 30% aqueous solution. Oily
purgatives are contraindicated. No fats, oils, or milk should be
given.
If convulsions occur, anticonvulsants should be given immediately,
e.g., 10 mg of diazepam, slowly, intravenously (children 1-5 mg),
repeated as necessary; or thiopental sodium or hexobarbital sodium
slowly, intravenously, in a dose of 10 mg/kg with a maximum total dose
of up to 750 mg for an adult, or paraldehyde 5 ml by intramuscular
injection. These short-acting anticonvulsants should always be
followed by phenobarbital given orally at 3 mg/kg (up to 200 mg for an
adult), or phenobarbital sodium given intramuscularly at 3 mg/kg (also
up to 200 mg for an adult).
Morphine and its derivatives, epinephrine, and noradrenaline should
never be given.
An unobstructed airway must be maintained. Respiratory inadequacy,
which may be accentuated by barbiturate anticonvulsants, should be
corrected and oxygen and/or artificial ventilation may be needed.
4.1.2 Health Surveillance Advice
A complete medical history and physical examination of regularly
exposed workers should be made on an annual basis.
4.2 Safety in Use
Handling liquid formulations: Wear protective neoprene or PVC
gloves, cotton overalls, rubber
boots, and face shield.
Handling powder formulations: Avoid raising a dust cloud. Wear
protective gloves and dust mask.
Follow the advice relating to
personal hygiene.
Application in the field
Aerial application: Ensure that flag-men (markers) do
not stand in the spray-path of the
aircraft; do not spray over surface
waters and avoid spraying over
ditches, canals, rivers, streams,
ponds, or lakes.
Ground spraying: Wear suitable protective clothing
(i.e., cap or hat, cotton overalls
or long-sleeved cotton shirt and
long trousers, boots or shoes);
when spraying tall crops or when
there is a risk of accidental
contamination by the spray, also
wear impermeable hood and jacket;
at all times avoid exposure to the
spray mist; do not spray into the
wind.
Hand-held ULV application: Wear suitable protective clothing
(i.e., cap or hat, cotton overalls
or long-sleeved cotton shirt and
long trousers, boots or shoes; read
and observe the instructions that
apply to the equipment being used;
pay proper attention to wind speed
and direction; always spray in a
down-wind direction; do not spray
if there are other people
immediately down-wind.
After application: Take off heavily splashed or
contaminated clothing; wash hands
and exposed skin before eating,
drinking, or smoking; wash
overalls, boots, hat, and other
protective clothing thoroughly,
especially the inside of gloves;
keep application equipment in good
condition, and free from leaks and
external contamination; keep
contents tightly closed in original
labelled container when not fully
used; do not reuse empty container
for any other purpose; keep
container in a safe place away from
food, children, and animals; empty
containers must be washed out and
disposed of as advised in sections
4.4.1 and 4.6.2.
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
The explosion hazard will depend on the solvent used in the
formulation, or on the characteristics of the dust.
4.3.2 Fire hazards
Liquid formulations containing organic solvents may be flammable.
Extinguish fires with alcohol-resistant foam, carbon dioxide, or
powder. With sufficient burning or external heat, endosulfan will
decompose, emitting toxic fumes. Fire-fighters should be equipped
with self-contained breathing apparatus, eye protection, and full
protective clothing.
The use of water spray should be confined to the cooling of unaffected
stock, thus avoiding the accumulation of polluted run-off from the
site.
4.4 Storage
Products should be stored in locked buildings -- preferably buildings
dedicated to insecticides.
Keep the products out of reach of children and unauthorized personnel.
Do not store near foodstuffs or animal feed.
4.4.1 Leaking containers in store
Take precautions and use appropriate personal protection (section
4.2). Empty any product remaining in damaged/leaking containers into
a clean empty drum, which should then be tightly closed and suitably
labelled.
Sweep up spillage with sawdust, sand, or earth (moisten for powders),
and dispose of safely.
After emptying, the leaking containers should be rinsed with at least
1 litre water per 20-litre drum. Swirl round to rinse the walls,
empty, and add the rinsings to the sawdust or earth. Puncture the
container to prevent reuse.
4.5 Transport
Comply with any local requirements regarding movement of hazardous
goods. Do not transport in the same compartment as foodstuffs. Check
that containers are sound and labels undamaged before despatch.
4.6 Spillage and Disposal
4.6.1 Spillage
Before dealing with any spillage, precautions should be taken as
required, and appropriate personal protection should be used (section
4.2).
4.6.1.1 Solid products
Sweep up and absorb remaining spilled product with moist sawdust,
sand, or earth, and transfer the sweepings in a suitable container to
a safe place for disposal.
4.6.1.2 Liquid products
Prevent the liquid from spreading or contaminating other cargo,
vegetation, or waterways, by making a barrier of the most suitable
available material, e.g., earth or sand.
Absorb spilled liquid with sawdust, sand, or earth, sweep up and place
it in a closeable container for later transfer to a safe place for
disposal.
4.6.1.3 All products
As soon as possible after the spillage and before reuse, cover all
contaminated areas with damp sawdust, sand, or earth. Sweep up and
place in a closeable container for later transfer to a safe place for
disposal. Since this insecticide is highly toxic for fish, care
should be taken to avoid run-off into water courses.
4.6.2 Disposal
Any surplus product, contaminated absorbents, and containers should be
disposed of in an appropriate way. Waste material should be burned in
a proper incinerator designed for organochlorine waste disposal
(1000°C and 30 min residence time with effluent gas scrubbing). If
this is not possible, bury in an approved dump or landfill where there
is no risk of contamination of surface or ground water. Comply with
any local legislation regarding disposal of toxic wastes. Puncture
container to prevent reuse.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Endosulfan is highly toxic for fish and some other aquatic organisms,
and moderately toxic for honey bees. It is not readily bioaccumulated
and it does not persist in the environment.
Industrial discharges from manufacture, formulation, and technical
applications should not be allowed to pollute the environment and
should be properly treated (section 4.6.2).
Any spillage or unused product should be prevented from spreading to
vegetation or waterways and should be treated and disposed of
properly.
6. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers concerned
with, and users of, endosulfan. It should be displayed at, or near,
entrances to areas where there is potential exposure to endosulfan,
and on processing equipment and containers. The card should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions on the
chemical safety card clearly explained. Space is available on the
card for insertion of the National Occupational Exposure Limit, the
address and telephone number of the National Poison Control Centre,
and for local trade names.
ENDOSULFAN
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,9,9a-hexahydro-6,9-methano-2,4,3-benzodioxathiepin-3-oxide
Molecular formula: C9H6Cl6O3S
RTECS registry number: RB 9275000 CAS Registry number: 115-29-7
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Pure substance:
Melting point of alpha-isomer 109.2°C Endosulfan is a non-systemic contact and stomach
of beta-isomer 213.3°C insecticide effective against a wide range of sucking and
Relative density at 20°C 1.745 chewing insect pests in agriculture, and public health
Vapour pressure <0.1 µPa at 20°C and technical fields of applications
<1.4 µPa at 40°C
Relative molecular mass 406.9
Solubility in water 0.5 mg/litre at 20°C Formulations include:
in most organic emulsifiable concentrates water dispersible powders,
solvents 5-65 g/100 ml at 20°C dispersions, dusts, granules, and ultra-low-volume
formulations
Technical grade:
Melting point 80°C
Appearance brown crystalline flakes
Odour faint odour of sulfur
dioxide
Ratio of isomers alpha- to beta-endosulfan
approx. 70:30
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: may cause poisoning in Avoid contact with skin; After contact with skin, wash immediately
contact with skin wear suitable protective with plenty of water and soap; immediately
clothing and gloves remove all contaminated clothing
EYES: may cause irritation to Avoid contact with eyes; wear In case of contact with eyes, rinse
eyes eye protection immediately with plenty of water and seek
medical advice
INHALATION: dusts may cause Do not breathe dusts or sprays
poisoning by inhalation
INGESTION: unlikely occupational Do not eat, drink or smoke during
hazard work; wash hands before eating,
drinking or smoking
Accidental or intentional ingestion If swallowed, seek medical advice
may cause poisoning immediately and show container or label;
do not induce vomiting where organic
solvents are present in emulsifiable
concentrates; keep at rest, lying face
downwards, and ensure a clear airway
ENVIRONMENT: Dangerous for Do not contaminate surface waters
aquatic life, particularly fish
SPILLAGE STORAGE FIRE AND EXPLOSION
Take appropriate personal Products should be stored in Liquid products containing organic solvents
precautions; prevent liquid locked buildings preferably may be flammable. Extinguish fires with
from spreading or contaminating dedicated to insecticides; keep alcohol-resistant foam, CO2 or powder.
other cargo, vegetation, or surface out of reach of children; keep With sufficient burning or external heat
waters and drainage systems, away from food, drink and animal endosulfan will decompose, emitting toxic
with a barrier of most suitable feeding stuffs fumes. Fire-fighters should be equipped
material, e.g., earth or sand with self-contained breathing apparatus,
eye protection, and full protective clothing;
Absorb spilled liquid with confine the use of water spray to cooling of
sawdust, sand, or earth; sweep up and unaffected stock, thus avoiding the
place it in a closeable container accumulation of polluted run-off from the site
for later safe disposal.
Since this insecticide is dangerous
for fish, care should be taken to
avoid run-off into water courses
WASTE DISPOSAL NATIONAL INFORMATION
Endosulfan waste material should National Occupational Exposure UN No. 2761, 2762, 2995, 2996
be burned in a proper incinerator Limit:
designed for organochlorine waste
disposal; if this is not possible, National Poison Control Centre:
bury in an approved dump or
landfill where there is no risk of Local Trade Names:
contamination of surface or ground
water; comply with any local
legislation regarding disposal of
toxic wastes
 7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other UN sources. Its intention is to give the reader a
representative but non-exhaustive overview of current regulations,
guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
The FAO/WHO Joint Meeting on Pesticide Residues (JMPR) has reviewed
residues and toxicity data on endosulfan on several occasions in the
past: 1965, 1967, 1968, 1971, 1974, 1975, 1978, 1982, and 1985.
The 1985 meeting discussed the situation with regard to endosulfan and
found it to be unsatisfactory. The meeting concluded that a complete
reevaluation was necessary, which should be based on current use
patterns, with toxicological data obtained following up-to-date
protocols and residue data obtained using the present analytical
methods. The reevaluation should also take into account the relevant
information in the Environmental Health Criteria document on
endosulfan. The required data should be submitted for study by the
1989 JMPR. Nevertheless, because of the absence of any indications of
potential major adverse toxicological effects in man, the temporary
acceptable daily intake (TADI) of 0.008 mg/kg body weight was
extended.
WHO (1975) issued a data sheet on endosulfan (No. 15) in its series of
"Data sheets on chemical pesticides" Based on a brief review of use,
exposure, and toxicity, practical advice is given on labelling,
safe-handling, transport, storage, disposal, decontamination,
selection, training, and medical supervision of workers, and first aid
and medical treatment.
WHO has classified endosulfan as moderately hazardous (WHO, 1986).
a The regulations and guidelines of all countries are subject to
change and should always be verified withe appropriate regulatory
authorities before application.
7.2 Exposure Limit Values
Some exposure limit values are shown in the table on pages 28-31.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
The use of endosulfan is permitted with certain restrictions in
Argentina, Canada, Czechoslovakia, Finland, Japan, Norway, Portugal,
the USSR, the United Kingdom, Venezuela, and Yugoslavia.
Absorption through the skin is indicated as a potentially hazardous
route in the regulatory documents of Argentina, Australia, Belgium,
Canada, Finland, the Netherlands, Switzerland, the United Kingdom,
and the USA.
7.4 Labelling, Packaging and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies endosulfan in:
Hazard Class 6.1: poisonous substance
Packing Group II: substances and preparations presenting a
serious risk of poisoning, when the content
of active ingredient is 80-100%.
Packing Group III: substance presenting a relatively low risk of
poisoning in transport, when the content of
active ingredient is 20-80% (solid) or 8-80%
(liquid).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Work-place Argentina Maximum permissible concentration 1979
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure limit (STEL) 0.3 mg/m3
Australia Threshold limit value (TLV)
- time-weighted average (TWA) 0.1 mg/m3
Bulgaria Maximum permissible concentration 0.1 mg/m3
Finland Occupational exposure limit
- time-weighted average (TWA) 0.1 mg/m3 1981
- short-term exposure limit 0.3 mg/m3 1981
Netherlands Maximum limit
- time-weighted average (TWA) 0.1 mg/m3
Switzerland Maximum worksite concentration (MAK)
- time-weighted average (TWA) 0.1 mg/m3
United Kingdom Recommended limit
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure level (STEL) 0.3 mg/m3
(10 min - TWA)
USA Threshold limit value (TLV)
- time weighted average (TWA) 0.1 mg/m3
- short term exposure limit (STEL) 0.3 mg/m3
USSR Maximum allowable concentration (MAC)
- ceiling value (CLV) (vapour + aerosol) 0.1 mg/m3 1977
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- (1x/day) 0.017 mg/m3
- (av/day) 0.0017 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0.008 mg/kg body
weight
USSR Acceptable daily intake (ADI) 0.002 mg/kg 1983
FOOD Plant Argentina Maximum limit 0.5-2 mg/kg 1969
Brazil Acceptable limits 0.01-2 mg/kg
FAO/WHO Maximum residue limit 0.02-30 mg/kg 1987
Germany, Maximum residue limit 0.2-30.0 mg/kg 1984
Federal
Republic of
Finland Maximum residue limit 0.5-1.0 mg/kg 1987
Netherlands Maximum residue limit 0.05-30 mg/kg 1987
EEC Maximum residue limit 1 mg/kg 1984
(Root vegetables) 0.2 mg/kg 1976
India Maximum tolerable concentration 0.2-2.0 mg/kg
Kenya Maximum limit 0.1-30.0 mg/kg
Sweden Maximum tolerable concentration 0.2-0.5 mg/kg 1985
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD USA Acceptable residue limit 24 mg/kg
(in dried tea)
Acceptable residue limit 0.1-2 mg/kg
USSR Maximum residue limit for food 0.1-1.0 mg/kg 1984
products exported and imported by CMEA
countries
USSR Pesticide is prohibited in some food 1983
products
WATER Ambient Mexico Maximum permissible concentration
(coastal) 0.0002 mg/litre 1973
(estuarine) 0.002 mg/litre 1973
SOIL USSR Permissible limit 0.1 mg/kg
The label should be as follows:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other UN sources. Its intention is to give the reader a
representative but non-exhaustive overview of current regulations,
guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
The FAO/WHO Joint Meeting on Pesticide Residues (JMPR) has reviewed
residues and toxicity data on endosulfan on several occasions in the
past: 1965, 1967, 1968, 1971, 1974, 1975, 1978, 1982, and 1985.
The 1985 meeting discussed the situation with regard to endosulfan and
found it to be unsatisfactory. The meeting concluded that a complete
reevaluation was necessary, which should be based on current use
patterns, with toxicological data obtained following up-to-date
protocols and residue data obtained using the present analytical
methods. The reevaluation should also take into account the relevant
information in the Environmental Health Criteria document on
endosulfan. The required data should be submitted for study by the
1989 JMPR. Nevertheless, because of the absence of any indications of
potential major adverse toxicological effects in man, the temporary
acceptable daily intake (TADI) of 0.008 mg/kg body weight was
extended.
WHO (1975) issued a data sheet on endosulfan (No. 15) in its series of
"Data sheets on chemical pesticides" Based on a brief review of use,
exposure, and toxicity, practical advice is given on labelling,
safe-handling, transport, storage, disposal, decontamination,
selection, training, and medical supervision of workers, and first aid
and medical treatment.
WHO has classified endosulfan as moderately hazardous (WHO, 1986).
a The regulations and guidelines of all countries are subject to
change and should always be verified withe appropriate regulatory
authorities before application.
7.2 Exposure Limit Values
Some exposure limit values are shown in the table on pages 28-31.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
The use of endosulfan is permitted with certain restrictions in
Argentina, Canada, Czechoslovakia, Finland, Japan, Norway, Portugal,
the USSR, the United Kingdom, Venezuela, and Yugoslavia.
Absorption through the skin is indicated as a potentially hazardous
route in the regulatory documents of Argentina, Australia, Belgium,
Canada, Finland, the Netherlands, Switzerland, the United Kingdom,
and the USA.
7.4 Labelling, Packaging and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies endosulfan in:
Hazard Class 6.1: poisonous substance
Packing Group II: substances and preparations presenting a
serious risk of poisoning, when the content
of active ingredient is 80-100%.
Packing Group III: substance presenting a relatively low risk of
poisoning in transport, when the content of
active ingredient is 20-80% (solid) or 8-80%
(liquid).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Work-place Argentina Maximum permissible concentration 1979
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure limit (STEL) 0.3 mg/m3
Australia Threshold limit value (TLV)
- time-weighted average (TWA) 0.1 mg/m3
Bulgaria Maximum permissible concentration 0.1 mg/m3
Finland Occupational exposure limit
- time-weighted average (TWA) 0.1 mg/m3 1981
- short-term exposure limit 0.3 mg/m3 1981
Netherlands Maximum limit
- time-weighted average (TWA) 0.1 mg/m3
Switzerland Maximum worksite concentration (MAK)
- time-weighted average (TWA) 0.1 mg/m3
United Kingdom Recommended limit
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure level (STEL) 0.3 mg/m3
(10 min - TWA)
USA Threshold limit value (TLV)
- time weighted average (TWA) 0.1 mg/m3
- short term exposure limit (STEL) 0.3 mg/m3
USSR Maximum allowable concentration (MAC)
- ceiling value (CLV) (vapour + aerosol) 0.1 mg/m3 1977
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- (1x/day) 0.017 mg/m3
- (av/day) 0.0017 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0.008 mg/kg body
weight
USSR Acceptable daily intake (ADI) 0.002 mg/kg 1983
FOOD Plant Argentina Maximum limit 0.5-2 mg/kg 1969
Brazil Acceptable limits 0.01-2 mg/kg
FAO/WHO Maximum residue limit 0.02-30 mg/kg 1987
Germany, Maximum residue limit 0.2-30.0 mg/kg 1984
Federal
Republic of
Finland Maximum residue limit 0.5-1.0 mg/kg 1987
Netherlands Maximum residue limit 0.05-30 mg/kg 1987
EEC Maximum residue limit 1 mg/kg 1984
(Root vegetables) 0.2 mg/kg 1976
India Maximum tolerable concentration 0.2-2.0 mg/kg
Kenya Maximum limit 0.1-30.0 mg/kg
Sweden Maximum tolerable concentration 0.2-0.5 mg/kg 1985
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD USA Acceptable residue limit 24 mg/kg
(in dried tea)
Acceptable residue limit 0.1-2 mg/kg
USSR Maximum residue limit for food 0.1-1.0 mg/kg 1984
products exported and imported by CMEA
countries
USSR Pesticide is prohibited in some food 1983
products
WATER Ambient Mexico Maximum permissible concentration
(coastal) 0.0002 mg/litre 1973
(estuarine) 0.002 mg/litre 1973
SOIL USSR Permissible limit 0.1 mg/kg
The label should be as follows:
 The FAO specifications for plant protection products containing
endosulfan specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
The endosulfan content should be stated and may not differ by more
than 2% from this for the technical product (and up to 10% for some
formulations). The isomeric composition of the total endosulfan
should be:
64-67% alpha-isomer and 29-32% beta-isomer.
In the WHO interim specifications for endosulfan technical and
endosulfan emulsifiable concentrate, similar specifications and
checking methods are given for its use in public health.
The WHO specifications include the following provisions:
(a) Technical endosulfan shall consist essentially of a mixture of the
alpha- and beta-isomers (same isomeric composition as above for FAO)
of endosulfan, together with related manufacturing compounds, and
shall be in the form of beige to dark brown flakes with the tendency
to agglomeration, free from extraneous impurities and added modifying
agents.
(b) Technical endosulfan shall be packed in suitable, clean
containers, as specified in the order.
All packages shall bear, durably and legibly marked on the container
the following:
Manufacturer's name
Endosulfan to Interim Specification WHO/IS/1.0205-1
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
Endosulfan is a sulfurous acid ester of a chlorinated cyclic
diol. It is poisonous if swallowed or inhaled as dusts or
mists. Avoid eye and skin contact; wear safety goggles,
protective gloves, clean protective clothing, and a respirator
when handling the material. Wash thoroughly with soap and water
after using. Keep the material out of reach of children and
well away from foodstuffs, animal feed, and their containers.
If poisoning occurs, call a physician. Endosulfan is highly
toxic to fish. Keep the material and the emptied containers
away from ground and surface water.
(c) The emulsifiable concentrate shall consist of technical endosulfan
dissolved in suitable solvents, with other necessary formulants added.
It shall be in the form of a stable liquid, free from suspended matter
and sediment. The technical endosulfan used in the manufacture of the
concentrate shall comply with the requirements of Interim
Specification WHO/IS/1.0205-1.
(d) Endosulfan emulsifiable concentrate shall be packed in suitable,
clean containers, as specified in the order. All packages shall bear,
durably and legibly marked on the container, the following:
Manufacturer's name
Endosulfan emulsifiable concentrate to Interim Specification
WHO/IS/3.0205-1
Endosulfan, ...... g/kg
Batch or reference number and date of test
Net weight of contents
Instructions for dilution
Date of formulation
and the same minimum cautionary notice as for the technical material.
The European Community legislation requires labelling as a dangerous
substance using the symbol:
The FAO specifications for plant protection products containing
endosulfan specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
The endosulfan content should be stated and may not differ by more
than 2% from this for the technical product (and up to 10% for some
formulations). The isomeric composition of the total endosulfan
should be:
64-67% alpha-isomer and 29-32% beta-isomer.
In the WHO interim specifications for endosulfan technical and
endosulfan emulsifiable concentrate, similar specifications and
checking methods are given for its use in public health.
The WHO specifications include the following provisions:
(a) Technical endosulfan shall consist essentially of a mixture of the
alpha- and beta-isomers (same isomeric composition as above for FAO)
of endosulfan, together with related manufacturing compounds, and
shall be in the form of beige to dark brown flakes with the tendency
to agglomeration, free from extraneous impurities and added modifying
agents.
(b) Technical endosulfan shall be packed in suitable, clean
containers, as specified in the order.
All packages shall bear, durably and legibly marked on the container
the following:
Manufacturer's name
Endosulfan to Interim Specification WHO/IS/1.0205-1
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
Endosulfan is a sulfurous acid ester of a chlorinated cyclic
diol. It is poisonous if swallowed or inhaled as dusts or
mists. Avoid eye and skin contact; wear safety goggles,
protective gloves, clean protective clothing, and a respirator
when handling the material. Wash thoroughly with soap and water
after using. Keep the material out of reach of children and
well away from foodstuffs, animal feed, and their containers.
If poisoning occurs, call a physician. Endosulfan is highly
toxic to fish. Keep the material and the emptied containers
away from ground and surface water.
(c) The emulsifiable concentrate shall consist of technical endosulfan
dissolved in suitable solvents, with other necessary formulants added.
It shall be in the form of a stable liquid, free from suspended matter
and sediment. The technical endosulfan used in the manufacture of the
concentrate shall comply with the requirements of Interim
Specification WHO/IS/1.0205-1.
(d) Endosulfan emulsifiable concentrate shall be packed in suitable,
clean containers, as specified in the order. All packages shall bear,
durably and legibly marked on the container, the following:
Manufacturer's name
Endosulfan emulsifiable concentrate to Interim Specification
WHO/IS/3.0205-1
Endosulfan, ...... g/kg
Batch or reference number and date of test
Net weight of contents
Instructions for dilution
Date of formulation
and the same minimum cautionary notice as for the technical material.
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
toxic by inhalation, in contact with skin and if swallowed;
irritating to eyes and skin; keep out of reach of children; keep
away from food, drink and animal feeding stuffs; if you feel
unwell, seek medical advice (show the label where possible).
The European Community legislation on labelling of pesticide
preparations classifies endosulfan in Class I/b for the purpose of
determining the label for preparations containing endosulfan and other
active ingredients.
7.5 Waste Disposal
In the USA, any non-domestic waste containing endosulfan must be
treated as a hazardous waste. Specific instructions are given for
notification and incineration. Under the Comprehensive Environmental
Response, Compensation, and Liability Act of 1980 (CERCLA), as
amended, unless in compliance with a specified permit or procedure,
owners/operators of vessels or onshore or offshore facilities must
notify the USA government (National Response Center) of any release of
endosulfan in or on navigable waters, adjoining shorelines, in the
contiguous zone or beyond the contiguous zone or to any other
environmental media (air, land or groundwater) in an amount equal to
or greater than one pound (0.454 kg).
In Finland, any waste containing endosulfan is classified as hazardous
waste and must be treated according to specific instructions.
7.6 Other Measures
The European Community legislation requires that Member States shall
prescribe that cereals (wheat, rye, barley, oats, paddy rice,
buckwheat, millet, grain sorghum, triticale) may not contain (in and
on cereals), from the time they are put into circulation, levels of
residues of endosulfan (sum of alfa- and beta-isomers and of
endosulfan sulfate, expressed as endosulfan) greater than maximum
0.1 mg/kg, and, in and on maize, levels of residues greater than 0.2 mg/kg
(applicable latest by June 1988).
The European Community legislation requires that Member States shall
prescribe that fruit and vegetables may not contain, from the time
they are put into circulation, levels of residues of endosulfan (sum
of alfa- and beta-isomers and of endosulfan sulfate, expressed as
endosulfan) greater than 0.2 mg/kg for root vegetables and root
fruits, and greater than 1 mg/kg for leaf vegetables.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice. Rome, Food and
Agriculture Organization of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1983) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished report No. VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods, 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1984) Environmental Health Criteria 40: Endosulfan. Geneva,
World Health Organization, 82 pp.
WHO (1986) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1986-87. Geneva, World Health
Organization (Unpublished report VBC/86.1).
WHO/FAO (1975-87) Data sheets on pesticides. Geneva, World Health
Organization (No. 15).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.
Lavenham, Lavenham Press Limited, British Crop Protection Council
The label must read:
toxic by inhalation, in contact with skin and if swallowed;
irritating to eyes and skin; keep out of reach of children; keep
away from food, drink and animal feeding stuffs; if you feel
unwell, seek medical advice (show the label where possible).
The European Community legislation on labelling of pesticide
preparations classifies endosulfan in Class I/b for the purpose of
determining the label for preparations containing endosulfan and other
active ingredients.
7.5 Waste Disposal
In the USA, any non-domestic waste containing endosulfan must be
treated as a hazardous waste. Specific instructions are given for
notification and incineration. Under the Comprehensive Environmental
Response, Compensation, and Liability Act of 1980 (CERCLA), as
amended, unless in compliance with a specified permit or procedure,
owners/operators of vessels or onshore or offshore facilities must
notify the USA government (National Response Center) of any release of
endosulfan in or on navigable waters, adjoining shorelines, in the
contiguous zone or beyond the contiguous zone or to any other
environmental media (air, land or groundwater) in an amount equal to
or greater than one pound (0.454 kg).
In Finland, any waste containing endosulfan is classified as hazardous
waste and must be treated according to specific instructions.
7.6 Other Measures
The European Community legislation requires that Member States shall
prescribe that cereals (wheat, rye, barley, oats, paddy rice,
buckwheat, millet, grain sorghum, triticale) may not contain (in and
on cereals), from the time they are put into circulation, levels of
residues of endosulfan (sum of alfa- and beta-isomers and of
endosulfan sulfate, expressed as endosulfan) greater than maximum
0.1 mg/kg, and, in and on maize, levels of residues greater than 0.2 mg/kg
(applicable latest by June 1988).
The European Community legislation requires that Member States shall
prescribe that fruit and vegetables may not contain, from the time
they are put into circulation, levels of residues of endosulfan (sum
of alfa- and beta-isomers and of endosulfan sulfate, expressed as
endosulfan) greater than 0.2 mg/kg for root vegetables and root
fruits, and greater than 1 mg/kg for leaf vegetables.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice. Rome, Food and
Agriculture Organization of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1983) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished report No. VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods, 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1984) Environmental Health Criteria 40: Endosulfan. Geneva,
World Health Organization, 82 pp.
WHO (1986) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1986-87. Geneva, World Health
Organization (Unpublished report VBC/86.1).
WHO/FAO (1975-87) Data sheets on pesticides. Geneva, World Health
Organization (No. 15).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.
Lavenham, Lavenham Press Limited, British Crop Protection Council
 Molecular formula: C9H6Cl6O3S
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,
9,-9a-hexahydro-6,9-methano-2,4,
3-benzodioxathiepin-3-oxide.
Trade names: Beosit, Malix, Thiodan, Thiofor,
Thiomul, Thionex, Thimul
Formulations under other trade names may
also exist
Development codes: HOE 002671, NIA 5462
CAS registry number:
Molecular formula: C9H6Cl6O3S
CAS chemical name: 6,7,8,9,10,10-hexachloro-1,5,5a,6,
9,-9a-hexahydro-6,9-methano-2,4,
3-benzodioxathiepin-3-oxide.
Trade names: Beosit, Malix, Thiodan, Thiofor,
Thiomul, Thionex, Thimul
Formulations under other trade names may
also exist
Development codes: HOE 002671, NIA 5462
CAS registry number:  7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other UN sources. Its intention is to give the reader a
representative but non-exhaustive overview of current regulations,
guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
The FAO/WHO Joint Meeting on Pesticide Residues (JMPR) has reviewed
residues and toxicity data on endosulfan on several occasions in the
past: 1965, 1967, 1968, 1971, 1974, 1975, 1978, 1982, and 1985.
The 1985 meeting discussed the situation with regard to endosulfan and
found it to be unsatisfactory. The meeting concluded that a complete
reevaluation was necessary, which should be based on current use
patterns, with toxicological data obtained following up-to-date
protocols and residue data obtained using the present analytical
methods. The reevaluation should also take into account the relevant
information in the Environmental Health Criteria document on
endosulfan. The required data should be submitted for study by the
1989 JMPR. Nevertheless, because of the absence of any indications of
potential major adverse toxicological effects in man, the temporary
acceptable daily intake (TADI) of 0.008 mg/kg body weight was
extended.
WHO (1975) issued a data sheet on endosulfan (No. 15) in its series of
"Data sheets on chemical pesticides" Based on a brief review of use,
exposure, and toxicity, practical advice is given on labelling,
safe-handling, transport, storage, disposal, decontamination,
selection, training, and medical supervision of workers, and first aid
and medical treatment.
WHO has classified endosulfan as moderately hazardous (WHO, 1986).
a The regulations and guidelines of all countries are subject to
change and should always be verified withe appropriate regulatory
authorities before application.
7.2 Exposure Limit Values
Some exposure limit values are shown in the table on pages 28-31.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
The use of endosulfan is permitted with certain restrictions in
Argentina, Canada, Czechoslovakia, Finland, Japan, Norway, Portugal,
the USSR, the United Kingdom, Venezuela, and Yugoslavia.
Absorption through the skin is indicated as a potentially hazardous
route in the regulatory documents of Argentina, Australia, Belgium,
Canada, Finland, the Netherlands, Switzerland, the United Kingdom,
and the USA.
7.4 Labelling, Packaging and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies endosulfan in:
Hazard Class 6.1: poisonous substance
Packing Group II: substances and preparations presenting a
serious risk of poisoning, when the content
of active ingredient is 80-100%.
Packing Group III: substance presenting a relatively low risk of
poisoning in transport, when the content of
active ingredient is 20-80% (solid) or 8-80%
(liquid).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Work-place Argentina Maximum permissible concentration 1979
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure limit (STEL) 0.3 mg/m3
Australia Threshold limit value (TLV)
- time-weighted average (TWA) 0.1 mg/m3
Bulgaria Maximum permissible concentration 0.1 mg/m3
Finland Occupational exposure limit
- time-weighted average (TWA) 0.1 mg/m3 1981
- short-term exposure limit 0.3 mg/m3 1981
Netherlands Maximum limit
- time-weighted average (TWA) 0.1 mg/m3
Switzerland Maximum worksite concentration (MAK)
- time-weighted average (TWA) 0.1 mg/m3
United Kingdom Recommended limit
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure level (STEL) 0.3 mg/m3
(10 min - TWA)
USA Threshold limit value (TLV)
- time weighted average (TWA) 0.1 mg/m3
- short term exposure limit (STEL) 0.3 mg/m3
USSR Maximum allowable concentration (MAC)
- ceiling value (CLV) (vapour + aerosol) 0.1 mg/m3 1977
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- (1x/day) 0.017 mg/m3
- (av/day) 0.0017 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0.008 mg/kg body
weight
USSR Acceptable daily intake (ADI) 0.002 mg/kg 1983
FOOD Plant Argentina Maximum limit 0.5-2 mg/kg 1969
Brazil Acceptable limits 0.01-2 mg/kg
FAO/WHO Maximum residue limit 0.02-30 mg/kg 1987
Germany, Maximum residue limit 0.2-30.0 mg/kg 1984
Federal
Republic of
Finland Maximum residue limit 0.5-1.0 mg/kg 1987
Netherlands Maximum residue limit 0.05-30 mg/kg 1987
EEC Maximum residue limit 1 mg/kg 1984
(Root vegetables) 0.2 mg/kg 1976
India Maximum tolerable concentration 0.2-2.0 mg/kg
Kenya Maximum limit 0.1-30.0 mg/kg
Sweden Maximum tolerable concentration 0.2-0.5 mg/kg 1985
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD USA Acceptable residue limit 24 mg/kg
(in dried tea)
Acceptable residue limit 0.1-2 mg/kg
USSR Maximum residue limit for food 0.1-1.0 mg/kg 1984
products exported and imported by CMEA
countries
USSR Pesticide is prohibited in some food 1983
products
WATER Ambient Mexico Maximum permissible concentration
(coastal) 0.0002 mg/litre 1973
(estuarine) 0.002 mg/litre 1973
SOIL USSR Permissible limit 0.1 mg/kg
The label should be as follows:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other UN sources. Its intention is to give the reader a
representative but non-exhaustive overview of current regulations,
guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
The FAO/WHO Joint Meeting on Pesticide Residues (JMPR) has reviewed
residues and toxicity data on endosulfan on several occasions in the
past: 1965, 1967, 1968, 1971, 1974, 1975, 1978, 1982, and 1985.
The 1985 meeting discussed the situation with regard to endosulfan and
found it to be unsatisfactory. The meeting concluded that a complete
reevaluation was necessary, which should be based on current use
patterns, with toxicological data obtained following up-to-date
protocols and residue data obtained using the present analytical
methods. The reevaluation should also take into account the relevant
information in the Environmental Health Criteria document on
endosulfan. The required data should be submitted for study by the
1989 JMPR. Nevertheless, because of the absence of any indications of
potential major adverse toxicological effects in man, the temporary
acceptable daily intake (TADI) of 0.008 mg/kg body weight was
extended.
WHO (1975) issued a data sheet on endosulfan (No. 15) in its series of
"Data sheets on chemical pesticides" Based on a brief review of use,
exposure, and toxicity, practical advice is given on labelling,
safe-handling, transport, storage, disposal, decontamination,
selection, training, and medical supervision of workers, and first aid
and medical treatment.
WHO has classified endosulfan as moderately hazardous (WHO, 1986).
a The regulations and guidelines of all countries are subject to
change and should always be verified withe appropriate regulatory
authorities before application.
7.2 Exposure Limit Values
Some exposure limit values are shown in the table on pages 28-31.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
The use of endosulfan is permitted with certain restrictions in
Argentina, Canada, Czechoslovakia, Finland, Japan, Norway, Portugal,
the USSR, the United Kingdom, Venezuela, and Yugoslavia.
Absorption through the skin is indicated as a potentially hazardous
route in the regulatory documents of Argentina, Australia, Belgium,
Canada, Finland, the Netherlands, Switzerland, the United Kingdom,
and the USA.
7.4 Labelling, Packaging and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies endosulfan in:
Hazard Class 6.1: poisonous substance
Packing Group II: substances and preparations presenting a
serious risk of poisoning, when the content
of active ingredient is 80-100%.
Packing Group III: substance presenting a relatively low risk of
poisoning in transport, when the content of
active ingredient is 20-80% (solid) or 8-80%
(liquid).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Work-place Argentina Maximum permissible concentration 1979
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure limit (STEL) 0.3 mg/m3
Australia Threshold limit value (TLV)
- time-weighted average (TWA) 0.1 mg/m3
Bulgaria Maximum permissible concentration 0.1 mg/m3
Finland Occupational exposure limit
- time-weighted average (TWA) 0.1 mg/m3 1981
- short-term exposure limit 0.3 mg/m3 1981
Netherlands Maximum limit
- time-weighted average (TWA) 0.1 mg/m3
Switzerland Maximum worksite concentration (MAK)
- time-weighted average (TWA) 0.1 mg/m3
United Kingdom Recommended limit
- time-weighted average (TWA) 0.1 mg/m3
- short-term exposure level (STEL) 0.3 mg/m3
(10 min - TWA)
USA Threshold limit value (TLV)
- time weighted average (TWA) 0.1 mg/m3
- short term exposure limit (STEL) 0.3 mg/m3
USSR Maximum allowable concentration (MAC)
- ceiling value (CLV) (vapour + aerosol) 0.1 mg/m3 1977
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- (1x/day) 0.017 mg/m3
- (av/day) 0.0017 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0.008 mg/kg body
weight
USSR Acceptable daily intake (ADI) 0.002 mg/kg 1983
FOOD Plant Argentina Maximum limit 0.5-2 mg/kg 1969
Brazil Acceptable limits 0.01-2 mg/kg
FAO/WHO Maximum residue limit 0.02-30 mg/kg 1987
Germany, Maximum residue limit 0.2-30.0 mg/kg 1984
Federal
Republic of
Finland Maximum residue limit 0.5-1.0 mg/kg 1987
Netherlands Maximum residue limit 0.05-30 mg/kg 1987
EEC Maximum residue limit 1 mg/kg 1984
(Root vegetables) 0.2 mg/kg 1976
India Maximum tolerable concentration 0.2-2.0 mg/kg
Kenya Maximum limit 0.1-30.0 mg/kg
Sweden Maximum tolerable concentration 0.2-0.5 mg/kg 1985
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD USA Acceptable residue limit 24 mg/kg
(in dried tea)
Acceptable residue limit 0.1-2 mg/kg
USSR Maximum residue limit for food 0.1-1.0 mg/kg 1984
products exported and imported by CMEA
countries
USSR Pesticide is prohibited in some food 1983
products
WATER Ambient Mexico Maximum permissible concentration
(coastal) 0.0002 mg/litre 1973
(estuarine) 0.002 mg/litre 1973
SOIL USSR Permissible limit 0.1 mg/kg
The label should be as follows:

 The FAO specifications for plant protection products containing
endosulfan specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
The endosulfan content should be stated and may not differ by more
than 2% from this for the technical product (and up to 10% for some
formulations). The isomeric composition of the total endosulfan
should be:
64-67% alpha-isomer and 29-32% beta-isomer.
In the WHO interim specifications for endosulfan technical and
endosulfan emulsifiable concentrate, similar specifications and
checking methods are given for its use in public health.
The WHO specifications include the following provisions:
(a) Technical endosulfan shall consist essentially of a mixture of the
alpha- and beta-isomers (same isomeric composition as above for FAO)
of endosulfan, together with related manufacturing compounds, and
shall be in the form of beige to dark brown flakes with the tendency
to agglomeration, free from extraneous impurities and added modifying
agents.
(b) Technical endosulfan shall be packed in suitable, clean
containers, as specified in the order.
All packages shall bear, durably and legibly marked on the container
the following:
Manufacturer's name
Endosulfan to Interim Specification WHO/IS/1.0205-1
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
Endosulfan is a sulfurous acid ester of a chlorinated cyclic
diol. It is poisonous if swallowed or inhaled as dusts or
mists. Avoid eye and skin contact; wear safety goggles,
protective gloves, clean protective clothing, and a respirator
when handling the material. Wash thoroughly with soap and water
after using. Keep the material out of reach of children and
well away from foodstuffs, animal feed, and their containers.
If poisoning occurs, call a physician. Endosulfan is highly
toxic to fish. Keep the material and the emptied containers
away from ground and surface water.
(c) The emulsifiable concentrate shall consist of technical endosulfan
dissolved in suitable solvents, with other necessary formulants added.
It shall be in the form of a stable liquid, free from suspended matter
and sediment. The technical endosulfan used in the manufacture of the
concentrate shall comply with the requirements of Interim
Specification WHO/IS/1.0205-1.
(d) Endosulfan emulsifiable concentrate shall be packed in suitable,
clean containers, as specified in the order. All packages shall bear,
durably and legibly marked on the container, the following:
Manufacturer's name
Endosulfan emulsifiable concentrate to Interim Specification
WHO/IS/3.0205-1
Endosulfan, ...... g/kg
Batch or reference number and date of test
Net weight of contents
Instructions for dilution
Date of formulation
and the same minimum cautionary notice as for the technical material.
The European Community legislation requires labelling as a dangerous
substance using the symbol:
The FAO specifications for plant protection products containing
endosulfan specify the composition and purity of the technical product
and its formulations. They also advise on methods for checking this.
The endosulfan content should be stated and may not differ by more
than 2% from this for the technical product (and up to 10% for some
formulations). The isomeric composition of the total endosulfan
should be:
64-67% alpha-isomer and 29-32% beta-isomer.
In the WHO interim specifications for endosulfan technical and
endosulfan emulsifiable concentrate, similar specifications and
checking methods are given for its use in public health.
The WHO specifications include the following provisions:
(a) Technical endosulfan shall consist essentially of a mixture of the
alpha- and beta-isomers (same isomeric composition as above for FAO)
of endosulfan, together with related manufacturing compounds, and
shall be in the form of beige to dark brown flakes with the tendency
to agglomeration, free from extraneous impurities and added modifying
agents.
(b) Technical endosulfan shall be packed in suitable, clean
containers, as specified in the order.
All packages shall bear, durably and legibly marked on the container
the following:
Manufacturer's name
Endosulfan to Interim Specification WHO/IS/1.0205-1
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
Endosulfan is a sulfurous acid ester of a chlorinated cyclic
diol. It is poisonous if swallowed or inhaled as dusts or
mists. Avoid eye and skin contact; wear safety goggles,
protective gloves, clean protective clothing, and a respirator
when handling the material. Wash thoroughly with soap and water
after using. Keep the material out of reach of children and
well away from foodstuffs, animal feed, and their containers.
If poisoning occurs, call a physician. Endosulfan is highly
toxic to fish. Keep the material and the emptied containers
away from ground and surface water.
(c) The emulsifiable concentrate shall consist of technical endosulfan
dissolved in suitable solvents, with other necessary formulants added.
It shall be in the form of a stable liquid, free from suspended matter
and sediment. The technical endosulfan used in the manufacture of the
concentrate shall comply with the requirements of Interim
Specification WHO/IS/1.0205-1.
(d) Endosulfan emulsifiable concentrate shall be packed in suitable,
clean containers, as specified in the order. All packages shall bear,
durably and legibly marked on the container, the following:
Manufacturer's name
Endosulfan emulsifiable concentrate to Interim Specification
WHO/IS/3.0205-1
Endosulfan, ...... g/kg
Batch or reference number and date of test
Net weight of contents
Instructions for dilution
Date of formulation
and the same minimum cautionary notice as for the technical material.
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
toxic by inhalation, in contact with skin and if swallowed;
irritating to eyes and skin; keep out of reach of children; keep
away from food, drink and animal feeding stuffs; if you feel
unwell, seek medical advice (show the label where possible).
The European Community legislation on labelling of pesticide
preparations classifies endosulfan in Class I/b for the purpose of
determining the label for preparations containing endosulfan and other
active ingredients.
7.5 Waste Disposal
In the USA, any non-domestic waste containing endosulfan must be
treated as a hazardous waste. Specific instructions are given for
notification and incineration. Under the Comprehensive Environmental
Response, Compensation, and Liability Act of 1980 (CERCLA), as
amended, unless in compliance with a specified permit or procedure,
owners/operators of vessels or onshore or offshore facilities must
notify the USA government (National Response Center) of any release of
endosulfan in or on navigable waters, adjoining shorelines, in the
contiguous zone or beyond the contiguous zone or to any other
environmental media (air, land or groundwater) in an amount equal to
or greater than one pound (0.454 kg).
In Finland, any waste containing endosulfan is classified as hazardous
waste and must be treated according to specific instructions.
7.6 Other Measures
The European Community legislation requires that Member States shall
prescribe that cereals (wheat, rye, barley, oats, paddy rice,
buckwheat, millet, grain sorghum, triticale) may not contain (in and
on cereals), from the time they are put into circulation, levels of
residues of endosulfan (sum of alfa- and beta-isomers and of
endosulfan sulfate, expressed as endosulfan) greater than maximum
0.1 mg/kg, and, in and on maize, levels of residues greater than 0.2 mg/kg
(applicable latest by June 1988).
The European Community legislation requires that Member States shall
prescribe that fruit and vegetables may not contain, from the time
they are put into circulation, levels of residues of endosulfan (sum
of alfa- and beta-isomers and of endosulfan sulfate, expressed as
endosulfan) greater than 0.2 mg/kg for root vegetables and root
fruits, and greater than 1 mg/kg for leaf vegetables.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice. Rome, Food and
Agriculture Organization of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1983) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished report No. VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods, 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1984) Environmental Health Criteria 40: Endosulfan. Geneva,
World Health Organization, 82 pp.
WHO (1986) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1986-87. Geneva, World Health
Organization (Unpublished report VBC/86.1).
WHO/FAO (1975-87) Data sheets on pesticides. Geneva, World Health
Organization (No. 15).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.
Lavenham, Lavenham Press Limited, British Crop Protection Council
The label must read:
toxic by inhalation, in contact with skin and if swallowed;
irritating to eyes and skin; keep out of reach of children; keep
away from food, drink and animal feeding stuffs; if you feel
unwell, seek medical advice (show the label where possible).
The European Community legislation on labelling of pesticide
preparations classifies endosulfan in Class I/b for the purpose of
determining the label for preparations containing endosulfan and other
active ingredients.
7.5 Waste Disposal
In the USA, any non-domestic waste containing endosulfan must be
treated as a hazardous waste. Specific instructions are given for
notification and incineration. Under the Comprehensive Environmental
Response, Compensation, and Liability Act of 1980 (CERCLA), as
amended, unless in compliance with a specified permit or procedure,
owners/operators of vessels or onshore or offshore facilities must
notify the USA government (National Response Center) of any release of
endosulfan in or on navigable waters, adjoining shorelines, in the
contiguous zone or beyond the contiguous zone or to any other
environmental media (air, land or groundwater) in an amount equal to
or greater than one pound (0.454 kg).
In Finland, any waste containing endosulfan is classified as hazardous
waste and must be treated according to specific instructions.
7.6 Other Measures
The European Community legislation requires that Member States shall
prescribe that cereals (wheat, rye, barley, oats, paddy rice,
buckwheat, millet, grain sorghum, triticale) may not contain (in and
on cereals), from the time they are put into circulation, levels of
residues of endosulfan (sum of alfa- and beta-isomers and of
endosulfan sulfate, expressed as endosulfan) greater than maximum
0.1 mg/kg, and, in and on maize, levels of residues greater than 0.2 mg/kg
(applicable latest by June 1988).
The European Community legislation requires that Member States shall
prescribe that fruit and vegetables may not contain, from the time
they are put into circulation, levels of residues of endosulfan (sum
of alfa- and beta-isomers and of endosulfan sulfate, expressed as
endosulfan) greater than 0.2 mg/kg for root vegetables and root
fruits, and greater than 1 mg/kg for leaf vegetables.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice. Rome, Food and
Agriculture Organization of the United Nations.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1983) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished report No. VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods, 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1984) Environmental Health Criteria 40: Endosulfan. Geneva,
World Health Organization, 82 pp.
WHO (1986) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1986-87. Geneva, World Health
Organization (Unpublished report VBC/86.1).
WHO/FAO (1975-87) Data sheets on pesticides. Geneva, World Health
Organization (No. 15).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.
Lavenham, Lavenham Press Limited, British Crop Protection Council
