
FLUSILAZOLE
EXPLANATION
Flusilazole is a fungicide. It was reviewed for the first time
at the present meeting.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
BIOLOGICAL DATA
Biochemical aspects
Absorption, distribution and excretion
14C-Flusilazole (uniformly phenyl labelled) was administered
orally to two male and two female Charles River CD rats/group by one
of three regimens: 1) a single dose of about 8 mg/kg bw with no
preconditioning: 2) a single dose of about 8 mg/kg bw following
preconditioning for 21 days on a diet containing 100 ppm
flusilazole; or 3) a single dose of about 200 mg/kg bw with no
preconditioning. By 96 (regimen 1) and 168 hours (regimens 2 and
3), 90% of the administered radioactivity was recovered in the urine
and feces in both sexes. Preconditioning did not affect excretion.
Excretion t1/2 was 31 hours in males and 37 hours in females.
There was a sex difference in the amount of material excreted in
feces compared to urine. In males total fecal recovery accounted
for 87% of the dose with 8% in urine, but in females the
corresponding figures were 59% and 23%. No significant amount of
radioactivity was expired as CO2 or volatile metabolites. Tissue
residues accounted for less than 25% of the dose with highest
amounts in carcass, GI tract and liver (means less than 1%).
An additional 1 male and 1 female rat were given a single dose
of 14C-flusilazole (triazole-3-labelled) of about 8 mg/kg bw
without preconditioning. As with the phenyl-labelled material, 88%
of the administered radioactivity was recovered in urine and feces
within 96 hours. However, the pattern of excretion was different
with the predominant route being the urine. In males, urinary
radioactivity accounted for 78% of the dose, with 11% in feces; in
females, the respective figures were 59% and 26%. Total tissue
residues were about 3% of the dose with the highest levels in
carcass (approx. 2% of dose), skin, GI tract and liver (all less
than .5% of dose (Anderson et al. 1986).
In another study, 14C-flusilazole (triazole-3-labelled) was
given orally to groups of 5 male and 5 female Charles River
Crl:CD(SD)BR rats by one of the following regimens: 1) a single dose
of about 8 mg/kg bw without preconditioning; 2) a single dose of
about 8 mg/kg bw following a 14-day treatment period with doses
about 8 mg non-labelled material/kg bw/day by gavage; 3) a single
dose of about 224 mg/kg bw without preconditioning. Total recovery
of radioactivity at 96 hours (regimen 1) or 120 hours (regimens 2
and 3) was 92.6 to 99.2%. Sex and regimen differences were not
observed in this study. Urinary excretion accounted for about 72%
and fecal excretion for 17%. About 90% of the material excreted was
recovered in the first 48 hours with all regimens. Tissue levels
were low and excluding carcass were less than .2% of the dose.
Carcass residues accounted for less than 3% of the dose (Cheng,
1986).
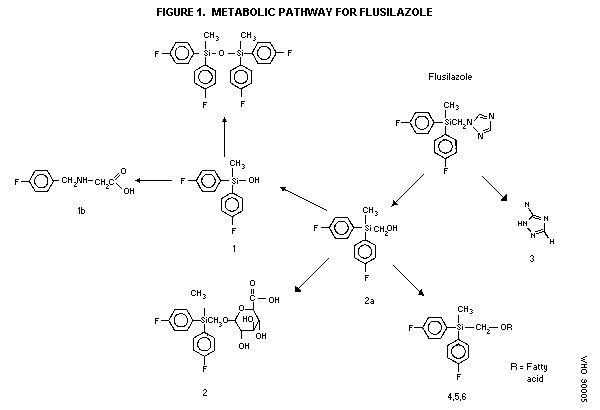
Biotransformation
14C-Flusilazole was extensively metabolized when administered
orally to Charles River CD rats. Recovered parent compound
accounted for 2-11% of the dose in all groups of animals and was
round almost exclusively in feces; urinary levels were less than 1%
of the dose. The chief fecal metabolites identified following
dosing with phenyl labelled flusilazole were:
[Bis(4-fluorophenyl)methyl] silanol (Metabolite 1) (about 30% of
dose in males, 19% in females); [Bis(fluorophenyl)methylsily]
methanol (Metabolite 2a) (about 9% of dose in both sexes); fatty
acid conjugates of the latter (19% of dose in males, 10% in
females); and disiloxane (about 11% of dose in males, 7% in
females). The same metabolites except for the fatty acid conjugates
were found in urine but in smaller amounts; none exceeded 1% of the
dose in males. In females, urine contained 7.5% of the dose as
metabolite 1 and 2.2% of the dose as metabolite 2a, and 1.9% of the
dose as siloxane.
Following dosing with triazole labelled flusilazole, the chief
metabolite identified was 1H,1,2,4-triazole (metabolite 3) which was
found predominantly in urine (63.8% of dose in males, 51.6% in
females) with smaller amounts in feces (4.0% of dose in males, 17.2%
of dose in females).
The proposed metabolic pathway is shown in Figure 1 (Anderson,
1986).
Metabolite 1: [Bis(4-fluorophenyl)methyl]silano
Metabolite 2a [Bis(4-fluorophenyl)methyl silyl]methanol
Metabolite 2 1-0-[Bis(4-fluorophenyl)methyl silyl]methyl]-ß-D-
glucopyranuronic acid
Metabolite 3 1H-1,2,4-Triazole
Metabolite 4 N-((4-fluorophenyl)methyl) glycine.
 Toxicological studies
Acute toxicity
The acute toxicity of flusilazole to several animal species is
given in Table 1. Signs of toxicity following oral administration
included weight loss, weakness, lethargy and, at higher doses,
laboured breathing, convulsions and prostration. Inhalation effects
were chiefly laboured breathing and lung sounds. Dermal
administration resulted only in some local erythema.
TABLE 1. RESULTS OF ACUTE TOXICITY ASSAYS WITH FLUSILAZOLE
SPECIES SEX ROUTE LD50a/ ALDa/ REFERENCES
LC50b ALCb
Technical flusilazole
Rat M oral 1500 Wylie et al. 1983
Rabbit M&F oral 350 Redgate et al. 1985
Mouse M oral 680 Wylie et al. 1985
F oral 1000 Wylie et al. 1985
Rat M oral 1110 Wylie et al. 1984a
F oral 674 Wylie et al. 1984a
Rabbit M&F dermal >2000 Gargus et al. 1983
Rat M inhal. 2.7b,c Poindexter et al. 1984
F inhal. 3.7b,c Poindexter et al. 1984
Rat M&F inhal. >5.0b Turner et al. 1985
Formulation - 40% flusilazole
Rat M oral 1865 Wylie et al. 1984b
F oral 1272 Wylie et al. 1984b
a mg/kg bw
b mg/l
c test material was 99% pure.
Short-term studies
Oral studies
Rat
Groups of six male Crl:CD rats were given 0 or 300 mg
flusilazole (95.5%) in corn oil/kg bw/day by oral gavage for 2 weeks
(5 days per week). After the tenth dose 3 rats/group were killed and
examined for histopathology. The remaining rats were killed after a
14-day recovery period. One treated rat died after the fifth dose
(found Day 7). The treated rats died lost weight during the first
week of treatment and showed alopecia, diarrhea, stained face and
perineal area, salivation and hypersensitivity during the treatment
period only. Liver vacuolation was seen in all treated animals. In
urinary bladder transitional epithelium hyperplasia and vacuolation
were seen in all sacrificed treated animals with an increase in
mitotic figures seen in two of three males killed immediately
following the treatment period. Two treated males had atypical
giant cells in the lumen of the seminiferous tubules and necrosis
and cellular degeneration of the germinal epithelium in the testes.
Hyperplasia and vacuolation of the transitional epithelium in the
kidney pelvis was seen in two treated males. Lesions tended to be
more severe in animals killed immediately after the treatment period
than in those from the recovery phase (Wylie et al. 1984c).
A 90-day feeding study was carried out in Charles River (CD)
rats with dietary levels of flusilazole (96.7%) of 0, 25, 125 and
375 ppm fed to groups of 16 rats/sex and 750 ppm fed to 10 rats/sex.
Groups of 10/sex/dose comprised the standard 90-day study while the
extra 6/sex/dose continued on diet for a one generation one litter
reproduction study and then received control diet for a 4-month
recovery phase. Body weight gains were reduced only in the females
given 750 ppm. There were no effects on food consumption, clinical
signs of toxicity, hematological parameters or urinalyses. Serum
cholesterol levels were increased in both males and females given
750 ppm. Absolute and relative liver weights were increased in both
sexes at 750 ppm. Mild urothelial hyperplasia was observed in
urinary bladders of 5 males and 8 females at 750 ppm and 1 male and
4 females at 375 ppm. After the 4-month recovery phase following
dosing with 0-375 ppm flusilazole, no evidence of bladder
hyperplasia was observed. At 750 ppm 5 males had liver lesions,
chiefly hepatocyte hypertrophy in 5 males and fatty
change/hepatocytolysis in 4 males. The NOAEL was 125 ppm (equal to
10 mg/kg bw/day) (Pastoor et al. 1983).
Mouse
Groups of 20 male and 20 female Crl:CD-1 mice were given diets
containing 0, 25, 75, 225, 500 or 1000 ppm of flusilazole (96.7%
pure). After four weeks on these diets 10 mice/sex/group were
sacrificed. The remaining 10 mice/sex/group were sacrificed after
90 days of treatment. There were no effects noted on body weight,
food consumption or clinical signs of toxicity in animals sacrificed
at either interval. At 90 days high-dose males and females had
slightly reduced Hb, Hct and RBC counts which were statistically
significant for at least one of these parameters in each sex. No
other hematological changes were noted. At both 4 weeks and 90 days
absolute and relative liver weights were increased in male mice at
225 ppm and higher and in female mice at 75 ppm and higher. The
absolute and relative kidney weights in males at 1000 ppm were
decreased. At 90 days hepatocellular hypertrophy was observed in
4/10 females at 1000 ppm and 8-10/10 males at 225, 500, and
1000 ppm. Hepatocellular vacuolar cytoplasmic changes were observed
in both sexes (incidences 0, 0, 0, 2, 7, 10 in males and 0, 0, 1, 3,
9, and 8 in females, at 0, 25, 75, 225, 500, and 1000 ppm,
respectively). Urothelial cell hyperplasia in urinary bladder was
observed in 1/10 males and 7/10 males at 500 and 1000 ppm,
respectively, and 2/10, 3/10, and 6/10 females at 225, 500 and
1000 ppm, respectively. No treatment-related kidney lesions were
observed. Tissues from animals killed at 4 weeks were not examined.
The NOAEL in this study was 25 ppm (equal to 4.5 mg/kg bw/day)
(Pastoor et al. 1984).
Dog
Four beagle dogs/sex/group were given diet containing 0, 25,
125, or 750/500 ppm flusilazole (93% pure) for 3 months. At the
highest dose level the dogs were given diet containing 750 ppm
flusilazole for 3 weeks, control diet for one week and then a dose
level of 500 ppm for the remainder of the study. The dose level was
reduced because of body weight loss and reduced food intake at
750 ppm. At 500 ppm the males gained a mean of 0.2 kg while the
females lost a mean of 0.5 kg body weight. The other two groups
were comparable to controls (gains of 1.6-1.9 kg in both sexes).
Food consumption remained lower than controls at the 750/500 ppm
dose level. Clinical signs related to treatment were observed in
one male and two females at 500 ppm (weakness or tremors). One
female at 25 ppm was sacrificed in extremis week 5. No other
animal died. In males at 750/500 ppm WBC count and monocyte counts
were slightly but statistically significantly increased and
cholesterol levels, total protein and albumen levels were lower than
controls. In both sexes at 750/500 ppm and males at 125 ppm alanine
amino transerase levels were higher than controls. Absolute and
relative liver weights were increased in both sexes at 750/500 ppm
and were considered related to treatment. Other organ weight
differences were probably related to the decreased body weights at
750/500 ppm. Mild mucosal hyperplasia in the urinary bladder was
observed in all males and females at 500 ppm and in 2 males at
125 ppm. In the stomach, hyperlasia of lymphoid follicles in the
pyloric granular mucosa was observed in 0/4, 3/4, 4/3, and 4/4 males
and 0/4, 0/4, 3/4, and 4/4 females at 0, 25, 125, and 750/500 ppm,
respectively. On the basis of this effect in stomach a NOAEL was
not demonstrated in this study (Richard et al. 1983).
Groups of 5 beagle dogs/sex were given diets containing 0, 5,
20, or 75 ppm flusilazole (95.8% pure) for one year. Although all
treated groups of females had lower weight gains than controls, this
did not appear to be treatment-related. No body weight effects were
seen in males. No treatment-related effects were noted in food
consumption or clinical signs of toxicity. WBC counts were higher
in both sexes at 75 ppm than in the controls. Alkaline phosphates
levels were higher than controls and cholesterol levels lower in
both sexes at 75 ppm but the difference was statistically
significant only in males. The 75 ppm males also had lower total
protein and albumen levels in serum. Relative liver weights were
increased in both sexes at 75 ppm and relative kidney weight in
females at 75 ppm. There was centrilobular hepatocyte enlargement
at 20 and 75 ppm, in both sexes with distinct vacuolation in 3
high-dose males. There were also a higher degree of centrilobular
inflammation in all of the high-dose dogs both with regard to
relative numbers of inflammatory cells and to increased number of
hepatic veins involved. All dogs in all groups including controls
had lymphoid hyperplasia in the gastric mucosa but the degree
increased with dose level and was moderate only in high-dose males
and females. There were no treatment-related kidney or urinary
bladder effects. The NOAEL in this study was 5 ppm (equal to
0.14 mg/kg bw/day) (O'Neal et al. 1985).
Dermal studies
Rabbit
A study in New Zealand White rabbits in which groups of 5-10
rabbits/sex were treated dermally with 0, 25, 250 or 2000 mg
flusilazole/kg bw/day (95.8% pure material) was terminated after 6
days exposure because of serve erythema with necrosis in animals at
250 and 2000 mg/kg bw/day. The erythema was first observed Day 4 of
test.
A range-finding study with 175 or 250 mg flusilazole (94.9%) kg
bw/day produced only mild erythema at both levels.
A 21-day study was performed in groups of 5 New Zealand White
rabbits/sex/dose at dose levels of 0, 1, 5, 25, and 200 mg
flusilazole (94.9% pure)/kg bw/day. For application the test
material was moistened with distilled water. The exposure sites
were occluded for 6 hours and then washed free of remaining test
material. There were no apparent treatment-related effects on body
weights: hematology; clinical chemistry; or liver, kidney and spleen
weights. At 200 mg/kg bw/day, four males and 2 females had
persistent mild erythema beginning Days 6 to 12. At termination
slight to mild diffuse hyperplasia/thickening of epidermis was
observed in the skin of one male and two females at 25 mg/kg bw/day
and three males and two females at 200 mg/kg bw/day (Sarver et al.
1986).
Long-term/carcinogenicity studies
Rats
Groups of 70 male and 70 female Crl:CD(SD)BR rats were given
diets containing 0, 10, 50 or 250 ppm flusilazole (95.6% pure) for
two years. Additionally, groups of 10/sex were given the same dose
levels for 6 months and 1 year. After about 100 days on test, 20
rats/sex/group were mated for the reproduction phase of the study.
These rats were returned to the long-term study after weaning of the
second litter. Body weights, food consumption and clinical
condition were monitored throughout the study. Hematology and
clinical chemistry parameters were examined at 3, 6, 9, 12 and 24
months on fasted animals. The animals sacrificed at 6 months were
examined for bladder lesions only. About 40 tissues/organs were
examined for control and high-dose animals and liver, bladder,
kidney, lung and gross lesions for the low and intermediate dose
groups sacrificed after one year. For animals that died on test or
were sacrificed at two years all tissues from all animals in all
groups were examined.
Survival to two years was 39-57% in the various groups with no
treatment-relationship. Survival did not drop below 50% in any
group until Week 98. The high-dose males had slightly lower body
weights from 6 months to the end of the study but there was no
similar effect in females. There were no treatment-related effects
on food consumption, clinical signs of toxicity, hematology,
clinical chemistry or urinalysis. In females, relative liver
weights were increased at both one and two years at 250 ppm and at
one year only at 50 ppm. No effect was observed in males. No
treatment-related effects on the urinary bladder were observed at 6
months. After one year there was an increased incidence of
hepatocellular hypertrophy in females (0/10, 0/10, 1/10, and 7/10 at
0, 10, 50, and 250 ppm, respectively) and at 250 ppm polyploidy
(increased number of multinuclear hepatocytes) was observed in two
females with none in any other group. There were no liver changes in
males at one year. There was an increased incidence of
hydronephrosis in males (0/10, 0/10, 3/10, and 4/10 at 0, 10, 50 and
250 ppm). Urothelial hyperplasia in renal pelvis was observed in
one male at 50 pm and one at 250 ppm, but not in any other group.
Treatment-related lesions were not observed in urinary bladders of
males. Females did not show kidney or bladder lesions after one
year. Among rats dying on test or sacrificed after two years there
were increased incidences in livers of females of acidophilic foci
of cellular alterations (3, 3, 4, and 13 at 0, 10, 50, and 250 ppm,
respectively) and diffuse fatty change (9, 9, 12, and 23,
respectively, in 63-68 /group). There were no treatment-related
liver lesions in males. In females, there was an increased
incidence of pyelonephritis in kidney (3, 3, 8, and 10,
respectively), but no treatment-related lesions were observed in
males. The only tumours which were increased in incidence in treated
groups were squamous cell carcinomas of the oral and nasal cavities
in males (0/66, 1/63, 0/67, and 3/64, respectively). Historical
control data from six two-year feeding studies showed five studies
which no squamous cell carcinomas of oral or nasal origin while the
remaining study had an incidence of 2/60. These data suggest that
these tumours may occur randomly as clusters and that the incidence
in this study was not treatment-related. There was, therefore, no
evidence of oncogenicity. It is noted, however, that the effects
noted were relatively mild and higher doses might have been
tolerated. The NOAEL for non-neoplastic effects was 10 ppm (equal
to 0.45 mg/kg bw/day) (Pastoor et al. 1986).
Mouse
Groups of 80 Crl:CD-1(ICR)BR mice/sex were given a diet
containing 0, 5, 25, or 200 ppm flusilazole (93%-95.6% purity) for
18 months. Additional groups of 10/sex/dose were given the same
diets for 6 months. Survival to 18 months was 76-86% in males and
57-80% in females with the highest survival at the high-dose level.
There were no treatment-related effects on body weight, food
consumption, hematology or serum protein. There was a higher
incidence of skin sores in high-dose males and females but these
were mainly ear lesions which were noted at the beginning of the
study and appeared to be related to the animal identification
process. High-dose males also had an increased incidence of ruffled
fur. Absolute and relative liver weights were increased in males at
200 ppm at both 6 and 18 months and in 200 ppm females at 6 months.
Absolute kidney weight only was decreased in females at 200 ppm at
18 months but the biological significance of this observation was
not clear. There were no treatment-related lesions in the kidney.
In the liver there was an increased incidence of hepatocellular
fatty change in both males and females at 200 ppm at 18 months
(40/80 and 24/80, respectively, compared to 4/80 and 2/79 in male
and female controls. One 200 ppm male at 6 months also had this
lesion, although 8/80 males at the 25 ppm levels also had
hepatoceullar fatty changes. This incidence was not statistically
significantly different from controls. There was no treatment-
related increase in incidence of any tumour type in this study. The
NOAEL in this study was 25 ppm (equal to 3.4 mg/kg bw/day) (Brock
et al. 1985).
Reproduction study
Rat
Groups of 6 Crl:CD(SD)BR rats/sex/dose level from the 90 day
feeding study were mated within the same dose level for a period of
15 days. Dose levels were 0, 25, 125, and 375 ppm flusilazole
(96.7% pure). The females were examined daily for evidence of a
copulation plug. After the mating period, the females were housed
individually.
The number of pregnant females/group was 3-6. The total number
of pups born did not show a treatment-related effect but two of the
five litters at 375 ppm were all dead at birth. The remaining three
litters at this dose level were 93-100% viable similar to the
litters in the other groups including controls. Mean pup weights at
Day 4 were lower in the 375 ppm group than the other three groups.
At 375 ppm (equal to 29 mg/kg bw/day) pup weights remained slightly
lower than the other groups to weaning (Pastoor et al. 1983).
Because of the small group size, the usefulness of this study
is limited.
Groups of 20 Crl:CD(SD)BR rats/sex/dose level from the chronic
toxicity/oncogenicity study were mated within the same dose level
for a period of 15 days after a dosing period of about 100 days.
Dose levels were 0, 10, 50, and 150 ppm flusilazole (95.6% pure).
The females were examined daily throughout the mating period for
evidence of a copulatory plug. At the end of the mating period the
females were housed individually and allowed to give birth and raise
their litters to weaning. About one week after weaning of the F1a
litters the females were sacrificed without pathological
examination. At weaning of the F1b litters 20 weanlings/sex/group
were selected as parents for the F2 generation. These weanlings
were maintained on the same diets as their parents for a 90 day
period before mating. Mating procedures were the same for the F0 -
F1 litters. It is stated that sibling matings were avoided. For
each litter the number of pups born, the number of pups born alive,
the number alive at 24 hours, 4 days, 12 days and 21 days, the
litter weight at 24 hours, and 4 days and the weight of male and
female pups at 21 days were recorded. At Day 4 all litters with
more than 10 pups were culled to 10, with equal numbers of males and
females where possible. Pups found dead were discarded. At weaning
of the F2b pups 10/sex/group were randomly selected and necropsied.
Liver, kidney, testes and brain were weighed and about 26
tissues/organs were examined for histopathology.
Food consumption and body weights were recorded only for the
pre-mating periods for the F0 and F1b animals. There were no
consistent differences during this period in F0 animals but F1b
males given 250 ppm had lower body weights. No differences in food
consumption were noted. Pregnancy rates were variable in all mating
periods but with no dose-relationship. There was no evidence of a
treatment-related effect on male reproductive parameters. There was
some indication that gestation length might be longer in the
high-dose group but this could not be determined unequivocally
because of the number of animals for which gestation lengths could
not be determined. The number of pups dead at birth was increased
in the 250 ppm group in all litters and in the 50 ppm group in the
F2a litter. Survival to Day 4 was reduced at 250 ppm in the F1a,
F1b, and F2a litters. Loss of the entire litter perinatally was
observed in 2, 2, 2 and 1 dams at 250 ppm in the F1a, F1b, F2a and
F2b litters, respectively, and in 1 dam at 50 ppm in the F2a
litter. Survival after 4 days was similar in all groups. The sex
of pups is indicated only at weaning after culling of the litters so
the sex ratio cannot be determined. Pup weights were similar in all
groups except for reduced weights at weaning in male and female pups
in the F1b and F2a litters. In the F2b weanling pups the absolute
and relative liver weights in males in the 250 ppm group were
increased. No effect was observed in females. The only
pathological lesion of note was hydronephrosis in female weanlings.
The incidence of unilateral and bilateral lesions combined was 1/10,
4/10, 3/10, and 5/10 at 0, 10, 50, and 250 ppm, respectively. The
incidences in the 10 and 250 ppm groups are slightly higher than
those observed in 10 historical control groups over the period
1982-1986. However, neither the severity nor the incidence of the
lesions showed a dose-related pattern so it is unlikely that this is
treatment-related effect. The NOAEL in this study was 10 ppm (equal
to 0.85 mg/kg bw/day on the basis of pre-mating intake data)
(Pastoor et al. 1986).
Special studies on mutagenicity
Flusilazole was negative in seven mutagenicity studies (Table
2).
Special studies on teratology
Rat
In a pilot study in which groups of seven pregnant rats were
given doses of 0, 100 or 300 mg flusilazole (99% pure)/kg bw/day
Days 7-16 of gestation, 300 mg/kg bw was overtly toxic and 100 mg/kg
bw slightly toxic to the dams. Both levels were embryotoxic and
300 mg/kg bw resulted in cleft palates in about 51% of the fetuses
in each litter.
TABLE 2. RESULTS OF MUTAGENICITY ASSAYS ON FLUSILAZOLE
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Ames testa Salmonella typhimurium 1-250 ug/plate Negative Donovan & Irr, 1982
TA1535, TA1537, TA98, (90%)
TA100
Ames testa Salmonella typhimurium 5-250 ug/plate Negative Arce et al. 1988
TA1535, TA97, TA98, (97.7%)
TA100
Unscheduled DNA Rat hepatocytes Crl (SD) 1 x 10-5 - Negative Chromey et al. 1983
synthesis 1.1 x 102 mM
(95.5%)
CHO/HGPRT assay Chinese hamster ovary 0.04 - 0.15 mM Negative McCooey et al. 1983
for gene mutationa cells (CHO-K1/BH4 clone) 0.05 - 0.275 mM
with and without
activation,
respectively
(95.5%)
Micronucleus test Mouse (CD-1) 375 mg/kg bw in Negative Sorg et al. 1984
(bone marrow) corn oil
(91.5%)
Chromosome aberration Human lymphocytes 1.7 - 100 ug/ml Negative Vlachos et al. 1986
assaya (94.85%)
TABLE 2 (CONTD).
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Chromosome assay Rat (Crl:CD (SD)) 50 - 500 mg/kg bw in Negative Farrow et al. 1983
(bone marrow) corn oil (not given)
a with and without metabolic activation.
Groups of 25 female Crl:CD(SD)BR rats mated to males of the
same strain were given doses of 0, 10, 50, or 250 mg flusilazole
(95.6%)/kg bw/day in corn oil by gavage Days 7-16 of gestation. The
day a copulation plug was observed was designated Day 1 of
gestation. Body weight, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation, the
rats were killed and examined for gross pathology, liver and uterine
weights, and numbers of corpora lutea, implantations, live and dead
fetuses. The uterus of any apparently non-pregnant females was
stained with ammonium sulfide to detect for early resorptions. The
fetuses were weighed and examined for external, visceral and
skeletal abnormalities.
Chromodacryorrhea, chromorhinorrhea, wet underbodies, or wet or
yellow or brown stained perineal areas were observed at the
250 mg/kg bw dose level with all but two females showing at least
one of these signs. The severity increased with continued dosing
and decreased following cessation of dosing. Some females at this
dose level had red vaginal discharges or stains and there was a
slightly increased incidence of focal alopecia. Two of the dams
died (Days 11 and 18). At 50 mg/kg bw one dam had red vaginal
discharge and one ruffled abdominal fur. Food consumption was
reduced during dosing at 50 and 250 mg/kg bw. At sacrifice relative
liver weights were increased in the dams at 50 and 250 mg/kg bw. At
250 mg/kg bw mean fetal body weight was reduced, incidence of
resorptions was increased and the number of live fetuses per litter
was reduced. Stunted fetuses were observed only in treated groups
at incidences of 1, 4, and 3 fetuses at 10, 50, and 250 mg/kg bw.
Cleft palate and absence of renal papilla were observed in fetuses
(12% and 9%, respectively) from dams given 250 mg/kg bw but not at
lower doses. At 50 mg/kg bw/day two fetuses (from 2 litters) had no
innominate artery and one (from the same litter as one of the
fetuses with no innominate artery) had a great vessel malformation
in the heart. These lesions were not seen in any other group so
were probably incidental. In all groups including controls there
was a high incidence of hydrocephalus and/or dilated lateral
ventricles of the brain. The incidence was not dose-related and no
hydrocephalus was observed in subsequent studies so this finding was
not considered to be treatment-related. The incidence of misaligned
sternebra, extra ossification centres in ribs and ossification delay
in sternebra were increased at all dose levels compared to controls.
There were increased incidences of rudimentary ribs at 50 and
250 mg/kg bw and extra ribs at 250 mg/kg bw. A NOEL was not
demonstrated in this study since skeletal variations were increased
at 10 mg/kg bw, the lowest dose tested. Maternal toxicity was seen
at 50 mg/kg bw. Teratogenic effects were observed at 250 mg/kg bw
(Lamontia et al. 1984a).
A second study included groups of 24 mated female Crl:CD(SD)BR
rats given doses of 0, 0.4, 2.0, 10.0, 50.0 or 250.0 (10 females) mg
flusilazole (95.6%)/kg bw/day in corn oil by gavage Days 7-16 of
gestation. The day a copulation plug was observed was designated
Day 1 of gestation. Body weight, food consumption and clinical
condition were monitored throughout the study. On Day 21 of
gestation the rats were killed and examined as in the study above.
At 50 and 250 mg/kg bw/day the dams had reduced body weight
gains during the early part of the dosing period but the difference
was statistically significant only at 250 mg/kg bw. Both groups had
reduced food intakes during dosing. Clinical signs of toxicity were
observed at 250 mg/kg bw: alopoecia, brown stains of face and limbs
and yellow staining of perineum. Relative liver weights were
increased in the 50 and 250 mg/kg bw groups. The incidence of early
resorptions was similar in all groups; however, the number of dams
with median or late resorptions was higher in groups given 10 mg/kg
bw or more. The mean number of live fetuses/litter was similar in
all groups. The incidence of stunted fetuses was increased at dose
levels of 10 mg/kg bw and higher. Cleft palate was observed in
fetuses from dams given 250 mg/kg bw but not at lower doses.
Absence of renal papilla was observed only in treated groups but
there was no dose-relationship and the observation was not
considered to be treatment-related. Hydrocephalus was not observed
in any group. Visceral (large renal pelvis and small renal papilla)
and skeletal (ribs anomalies) variations were observed at 10 mg/kg
bw and higher. Absence of the innominate artery was observed in
4(3) fetuses (litters) at 50 mg/kg bw and 1 fetus at 250 mg/kg bw.
Retarded development (delayed ossification of sternebra and
vertebral arch) was also evident at dose levels of 10 mg/kg bw and
higher. The incidence of red foci in bladder of fetuses was
increased at dose levels of 10 mg/kg bw and higher; however, these
lesions were non-specific and were considered to be of no biological
significance. The NOAEL in this study was 2 mg/kg bw/day (Lamontia
et al. 1984b).
In a dietary study, groups of 24 mated Crl:CD(SD)BR female rats
were given diets containing 0, 50, 100, and 300 or 900 ppm
flusilazole (94.8%) Days 7-16 of gestation. The day a copulation
plug was observed was designated Day 1 of gestation. Body weights,
food consumption and clinical condition were monitored throughout
the study. On Gestation Day 21 the rats were sacrificed and
examined for gross pathology, liver and uterine weights and uterine
contents. Fetuses were weighed, sexed and examined for external
visceral and skeletal abnormalities.
One female at 300 ppm died on Day 19 of gestation. The death
was not considered to be related to treatment. During Days 7-11 of
gestation at the beginning of dosing weight gains in all treated
groups were lower than in controls in a dose-related manner.
Significant reductions were noted Days 7-9 at 300 and 900 ppm. Food
consumption during dosing was lower than controls in all treated
groups but reached significant reductions only at 300 and 900 ppm.
There were no treatment-related clinical signs of toxicity.
Relative liver weights did not indicate a significant treatment-
related effect. Pregnancy rate was high in all groups. One dam at
900 ppm totally resorbed its litter. The incidence of early
resorptions was similar in all groups. However, there was an
increased incidence (dose-related) of median and late resorptions at
100 ppm and higher. Litter size was inversely dose-related and
significantly smaller than in controls in the 900 ppm group. Small
litters (less than 10 fetuses) were only seen at dose levels of
100 ppm and above. The 50 ppm group was similar to controls. The
incidence of stunted fetuses was increased at 300 and 900 ppm. Mean
fetal weights were not affected. No treatment-related malformations
were observed. No fetuses showed cleft palate and incidence of
absence of renal papilla was not dose or treatment-related. Two
fetuses from different litters in the 900 ppm group had slightly
domed heads but hydrocephalus was not confirmed on visceral
examination. There was a significant dose-related increase in
skeletal variations at dose levels of 100 ppm and higher with extra
ossification of the sternebra at all 3 dose levels, and rudimentary
ribs, extra ossification in cervical ribs and delayed ossification
in the vertebral arches (cervical) at 300 and 900 ppm. Red foci
were observed in the bladder of a few fetuses at all dose levels
with a slightly higher but not dose-related incidence at 100 ppm and
above. As in the previous study, these were not considered to be
biologically significant. The NOAEL in this study was 50 ppm (equal
to 4.6 mg/kg bw) (Alvarez et al. 1984).
A study of prenatal and postnatal toxicity in rats was
conducted in two phases. For the prenatal study (phase 1), groups
of 24 female Crl:CD(SD)BR rats were mated on a 1:1 basis with males
of the same strain. The day a copulation plug was observed was
designated Day 1 of gestation. On Days 7 through 16 the females
were given a dose of 0, 0.2, 0.4, 2, 10, or 100 mg flusilazole
(94.8% pure in 0.5% aqueous methyl cellulose)/kg bw by oral
intubation. Body weights, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation the
females of one group at each dose level were killed for examination
of the uterine contents. The extra control and 100 mg/kg bw groups
were killed Day 22 of gestation in order to determine whether
absence of renal papillae was a compound-related effect or an
anomaly.
Analyses of the dosing solutions indicates that the solution
prepared for the first day of dosing contained only 6-19% of the
nominal concentrations. Since solutions were prepared daily this
may have been given to only the one female/group that had reached
Gestation Day 7 on that day and only two of these (one at 2 and one
at 100 mg/kg) were pregnant. However, the next analyses given were
done on solutions prepared six days later and it is unknown whether
the technical error causing the low concentrations was corrected
before this date. Subsequent analyses indicated concentrations
75-110% of nominal. Assuming the worst case, 13-16 pregnant
females/group were given 1-6 doses which were about 10% of the
intended dose level (mean duration of low doses was 3.4-3.7 days).
In each group there were 7-8 pregnant females which did not receive
the low doses.
Body weight gains and food consumption were lower than controls
during dosing in the females given 100 mg/kg bw. Stained chin was
observed in many of the 100 mg/kg bw females and some showed
perinasal staining and/or wet perineum. Absolute and relative liver
weights were increased in dams at 100 mg/kg bw. Pregnancy rate was
83-96% in treated animals and 96-100% in controls; there was no dose
relationship. The number of early resorptions and dams with early
resorptions did not differ among the groups. However, there was an
increased incidence of median and late resorptions at 100 mg/kg bw.
The numbers of stunted fetuses and dams with stunted fetuses were
increased at 10 and 100 mg/kg bw (statistically significant only at
100 mg/kg bw). One fetus in each of the 100 mg/kg bw groups had no
renal papilla unilaterally. One other high-dose fetus had
malformations: no tail and imperforate anus (group killed Gestation
Day 22). There was an increased incidence of bladder foci in the
two 100 mg/kg bw groups compared to their respective controls.
However, the Gestation Day 22 control incidence was higher than the
100 mg/kg bw group killed on Gestation Day 21. Groups given 0.4,
2.0 and 10.0 mg/kg bw had slightly higher incidences of bladder
foci. Incidence of small papilla in the kidney was increased at 10
and 100 mg/kg bw but in the groups killed Gestation Day 21 the
increase was not dose-related. Incidence of distended ureter was
increased at 10 and 100 mg/kg bw when expressed as % affected per
litter. The number of litters affected was slightly higher than
controls in the Day 21 group at 100 mg/kg bw but not in the Day 22
group.
In the postnatal study (phase II), groups of 22 females
Crl:CD(SD)BR rats were mated and dosed as in phase I except that
only one control and one 100 mg/kg bw group were included. The
females were permitted to have their litters and raise them to
weaning. Body weights, food consumption and clinical signs were
monitored throughout the gestation and lactation periods. Offspring
were counted and weighted by sex Days 0, 4 (before and after
culling), 7 and 14 post partum. Pups were weighed individually Day
21. At weaning the pups were sacrificed and subjected to gross
necropsy.
The dosing solutions were adequately prepared in this phase of
the study. At 100 mg/kg bw five dams died; four on Gestation Day 24
or 25 (with apparently normal litters) and one four days post partum
after delivering a litter (all dead) Day 25. At 2.0 mg/kg bw one
dam died Day 24 during delivery. At 100 mg/kg body weight gain and
food consumption were reduced during the first few days of dosing
but were similar to controls for the remainder of the study. A few
dams at 100 mg/kg bw are stated to have shown pallor (4), hunching
(2), weakness (2) and dystocia (1) during the puturition and
lactation periods. There was an increase in mean gestational length
at 10 and 100 mg/kg bw. In controls, gestation did not exceed 22
days. In treated groups a gestation period of 23 days was seen in
2, 1, 4, 12, and 4 dams at 0.2, 0.4, 2.0, 10.0, and 100 mg/kg bw,
respectively, and at 100 mg/kg bw 11 dams did not deliver until 24
days gestation. At 100 mg/kg the mean gestation length was 23.7
days compared to 21.8 days in controls. Among the 11 high-dose dams
with parturition at Gestation Day 24, six had no live fetuses and
the other five had 1-5 live pups (mean 3) and 1-7 dead pups (mean
4.8). At 10 mg/kg bw the longest gestation period was 23 days and
there was no apparent effect on pup survival at birth. At this dose
four dams had one dead pup (of 6-15 pups in litter) at birth; two
delivered Day 22 and two delivered Day 23. This latter incidence
of dead pups at birth was similar to that in controls. Survival to
Day 4 was decreased at 100 mg/kg bw but was similar to controls at
the other dose levels. At 100 mg/kg bw 18% of the pups alive Day 0
of lactation had died by Day 4 compared to 0.4-1.7% of the pups in
the control and lower dose groups. There was no effect on survival
for the rest of the gestation period. Litter size was reduced at
100 mg/kg bw even when both live and dead pups in the litters were
considered. Mean litter size was slightly lower than controls in
the 10 mg/kg bw group reaching statistical significance after
culling. This was the result of an increased number of litters with
less than 10 pups (4 cf 0 in controls and 1-2 in the other treated
groups). Pup weights at birth were somewhat higher in the three
highest dose groups than in controls. At 2 and 10 mg/kg bw a few
litters with higher weights were among those with a 23 day gestation
length. At 100 mg/kg bw mean, live litter weights were not as high
as at 10 mg/kg bw mean, live litter weights were not as high as at
10 mg/kg bw but other effects confound any comparisons. No
treatment-related effects on sex were apparent. The only
alterations noted in the weanlings were dilatation of the renal
pelvis and distended ureter. The incidence was not unequivocally
dose-related although somewhat higher at 10 mg/kg bw/day. It is
stated that among the dead pups, absence of renal papilla was
observed in only 2/42 pups and small papilla (size 1 or 2) was seen
in only 4 pups (2 of each size). Most of the dead pups were in the
high-dose group so it is presumed that these observations were from
that group. In phase II the NOAEL was 2 mg/kg bw with the only
effects being a slightly longer gestation period with a higher mean
weight in a few animals (Alvarez et al. 1985a).
Rabbit
Groups of 18 New Zealand White female rabbits were inseminated
with semen from one of the two male rabbits of the same strain from
the same source. Day of insemination was designated Gestation Day
0. On Days 7-19 the females were given nominal doses of 0, 2, 5, or
12 mg flusilazole (94.8%)/kg bw/day by gavage in corn oil (actual
1.9, 4.8 and 10.1 mg/kg bw/day). There were two deaths during
treatment: one control female Day 19 and one high-dose female Day
16. Another control female died Day 23. Periodic anorexia was
observed in some rabbits in all groups and was not related to
treatment. No treatment-related effects were noted on clinical
observations, body weight, mean food consumption, abortions (2 in
controls, 3 in each of the treated groups), number of resorptions,
number of live or dead (none in any group) fetuses, fetal weights,
maternal liver weights or maternal pathology. The total number of
malformed fetuses was not increased by treatment. The incidence of
hydrocephalus was 1(1), 2(1), 4(2), and 4(3) fetuses (litters) in
the nominal 0, 2, 5, or 12 mg/kg bw/day level groups, respectively.
There was no increase in any other malformation nor of any
variation. In three subsequent studies hydrocephalus was observed
only in one fetus at 35 mg/kg bw/day and in none at any lower dose
or in controls. The incidence of hydrocephalus in this study,
therefore, was not confirmed and could not be regarded as related to
treatment. The NOAEL in this study was the nominal 12 mg/kg bw/day
level (actual 10.1 mg/kg bw/day) (Solomon et al. 1984).
A second study with 20 New Zealand White female rabbits/group
artificially inseminated with sperm from one of three male rabbits
tested dose levels of 0, 12, and 35 mg flusilazole (95%)/kg bw/day
administered Days 7-19 of gestation by gavage. In this study
pregnancy rates were low particularly in the two treated groups (not
dose-related). At 35 mg/kg bw/day, 2 of the does aborted and 10
totally resorbed their litters with only one doe with a live litter
at examination Day 29. There was an increased incidence of vaginal
discharge, discharge in cage and stained tail at this dose level as
well as an increased incidence of periodic anorexia. Since there
was only one litter, no teratologic effects could be determined.
One fetus did have hydrocephalus. Reproductive parameters at
12 mg/kg bw/day did not differ from controls and there was no
evidence of maternal toxicity at this level. The NOAEL in this
study was the nominal level of 12 mg/kg bw/day (actual 11 mg/kg
bw/day) (Zellers et al. 1985).
A pilot dietary study in New Zealand White rabbits included 7
females/group offered diets containing 0, 500, 1000, or 2000 ppm
flulilazole (94.8%) on Days 7-19 of gestation. Full results are not
given but pregnancy rate was low at 500 and 1000 ppm /3/7 at each
level and incidence of total resorption was high (4/7) at 2000 ppm.
Dietary levels of 0, 300, 600, and 1200 ppm flusilazole (94.8%)
were given to groups of 20 New Zealand White female rabbits days
7-19 of gestation following artificial insemination (day of
insemination designated Day 0 of gestation). Body weights, food
consumption and clinical signs of toxicity were monitored throughout
the study. The rabbits were sacrificed Day 29 of gestation and
examined for gross pathology, corpora lutea, numbers of live and
dead fetuses or resorption sites. The fetuses were examined for
external, visceral and skeletal abnormalities.
Body weights and food consumption were reduced during dosing.
There were no treatment-related clinical signs, pathological lesions
or changes in liver weights. Pregnancy rate was reduced in all
treated groups with the lowest rate in the high-dose group. The
number of dams with total resorptions was increased in the 600 and
1200 ppm groups. There were no treatment-related effects on mean
number of live fetuses, resorptions (in dams with fetuses) or fetal
weights. The number of litters available at 600 and 1200 ppm was
small (3 at each dose level), so the validity of the incidence of
fetal malformations and variations is uncertain at these levels.
There were no apparent effects at 300 ppm.
Since the pregnancy rate was reduced in all treated groups
another dietary study was performed with levels of 0, 30, 100, and
300 ppm flusilazole in diet offered Days 7-19 of gestation to groups
of 18 or 25 (300 ppm only) female New Zealand White rabbits
artificially inseminated with sperm from proven male rabbits. In
this study pregnancy rate was low in all groups but particularly in
the controls. In the 0 and 300 ppm groups 25% and 29%,
respectively, of the pregnant does totally resorbed their litters
but there were no total resorptions in the other two groups.
Because of the low number of dams with litters available in the
control group, the data in this study could not be adequately
assessed and a NOAEL cannot be determined (Alvarez et al. 1985b).
It has been suggested by the company that the reproductive
effects observed in these studies (reduced pregnancy and increased
total resorptions) were the result of inadequate technique during
artificial insemination. This might explain the effects observed in
the control group in the second dietary study.
Special studies on skin irritation and sensitization
Guinea Pig
In young adult Hartley guinea pigs, a range-finding study
indicated mild erythema, 24 hours after topical application to the
skin of the test material (90% pure flusilazole). The primary
irritation study conducted with 5 and 50% solutions in demethyl
phthalate did not demonstrate irritation in any of the 10 animals
exposed. Intradermal injections of 1% solutions of flusilazole (90%
pure) in dimethyl phthalate weekly for 4 weeks in the same animals
caused erythema and edema with necrotic centres at the injection
sites at 24 hours. Challenge with topical applications of 5 and 50%
solutions did not demonstrate sensitization (Wylie et al. 1984d).
In another study using 97.7% pure flusilazole, young adult
Duncan Hartley albino guinea pits (10/sex) were given 3 weekly
dermal applications of the test material (slightly moistened with
dimethyl phthalate) which was covered with plastic wrap for 6 hours.
No signs of irritation were observed in any of the animals after any
of these induction applications. Two weeks after the last induction
treatment, the guinea pigs were challenged with a dermal application
of test material on an untreated site which was then covered for 6
hours. No reaction was observed in any of the animals. A positive
control groups treated with 1-chloro,-2,4-dinitrobenzene (DNCB)
showed several erythema with necrosis after the second and third
induction applications and severe erythema after the challenge dose
two weeks later (Brock et al. 1988).
Special study of eye irritation
Rabbit
Two male New Zealand White rabbits were given doses of 0.01 ml
of undiluted flusilazole (90% pure) in the right conjunctival sac.
Both rabbits shoed mild conjunctival redness and chemosis at 1-4
hours. The washed eye also showed some discharge at one hour but no
other effects. The unwashed eye showed slight corneal opacity at
1-4 hours and slight cloudiness on biomicroscope examination at
24-48 hours. The eye was normal at 72 hours. The test material
produced only transient mild irritation (Hall et al. (1984).
COMMENTS
Flusilazole administered orally to rats was rapidly excreted in
urine and feces. The label (14C) from phenyl-labelled material was
excreted predominantly in feces, with a sex difference in that males
excreted higher amounts in feces than did females. However,
substantial variability existed. When triazole-labelled material
was given, the 14C was recovered predominantly in urine. Although
one study indicated a sex difference, with males excreting more in
urine than females, a sex difference was not observed in a second
study with more animals. Tissue levels were low. The highest
levels were found in the carcass (about 2% of the dose after 96-120
hours). Flusilazole was extensively metabolized in the rat.
Recovered parent compound accounted to 2-11% of the dose and was
recovered almost exclusively in feces.
Flusilazole has a low acute toxicity in the species examined.
In short-term (90-day) studies, urothelial hyperplasia in the
urinary bladder was observed in rats, mice and dogs at dietary
levels of 375, 225, and 125 ppm, respectively. Liver lesions,
including hepatocellular hypertrophy and hepatocellular vacuolar
cytoplasmic changes, were also observed in the 90-day studies in
rats and mice (at 750 and 225 ppm, respectively) and in a one-year
study in dogs at 20 ppm.
In a one-year feeding study in beagle dogs, centrilobular
hepatocyte enlargement was observed at 20 and 75 ppm. The NOAEL was
5 ppm, equal to 0.14 mg/kg bw/day.
In long-term feeding studies in rats and mice no bladder
lesions were observed. There was urothelial hyperplasia in the
renal pelvis in 1 of 10 male rats at 50 ppm and 1 of 10 male rats at
250 ppm for one year, but in none of the rats (7 of 10) given
250 ppm at one year (hepatocellular hypertrophy) and two years
(acidophilic foci of cellular alteration: 3/66, 6/63, 4/68, and
13/65 at 0, 10, 50, and 250 ppm, respectively). Male and female
mice given 200 ppm for 18 months developed hepatocellular fatty
changes (40/80 and 24/80, respectively, versus 4/80 and 2/70 in
respective controls). This effect was also reported in 8/80 male
mice at 25 ppm, which was not statistically significantly different
from controls. There was no evidence of oncogenicity in either
species. The NOAEL in the rat study was 10 ppm (equal to 0.45 mg/kg
bw/day). The NOAEL in the mouse study was 5 ppm (equal to
0.79 mg/kg bw/day).
In a 2-generation, 2 litters per generation reproduction study
in rats, flusilazole caused perinatal mortality in both F1 and F2
litters of dams given dietary levels of 250 ppm. There was some
indication of increased gestation length at this dose level, an
effect which was also observed in a post-natal toxicity study in
rats at doses of 10 mg/kg bw/day (gavage) and higher. Perinatal
mortality was observed at 100 mg/kg bw/day in this study. The
biological significance of prolonged gestation in this study is
uncertain. The NOAEL was 10 ppm (equal to 0.895 mg/kg bw/day) but
not at lower doses. The NOAEL in rats was 2 mg/kg bw/day. No
teratogenic effects were seen in rabbits. The NOAEL was 100 ppm
(equal to 2.8 mg/kg bw/day) based on maternal toxicity.
After reviewing all available in vitro and in vivo
short-term tests, the Meeting concluded that there was no evidence
of genotoxicity.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effect
Mouse: 25 ppm in the diet, equal to 3.4 mg/kg bw/day
Rat: 10 ppm in the diet, equal to 0.45 mg/kg bw/day
Dog: 5 ppm in the diet, equal to 0.14 mg/kg bw/day.
Estimate of acceptable daily intake for humans
0-0.001 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
1. Observations in humans.
2. Toxicity of the biotransformation products.
REFERENCES
Alvarez, L., Krauss, W.C. & Staples, R.E. (1984) Developmental
toxicity study in rats given INH-6573-66 in the diet on Days 7-16 of
gestation. Unpublished Report No. 432-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., & Kaplan, A.M. (1985a) INH-6573.
Prenatal and postnatal toxicity study in rats dosed by gavage on
Days 7-16 of gestation. Unpublished Report No. 654-85 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., Driscoll, C.D. & Kaplan, A.M. (1985b)
INH-6573. Developmental toxicity study in rabbits treated by diet
on Days 7-19 of gestation. Unpublished Report No. 337-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Anderson, J.J., Stadalius, M.A. & Schlueter, D.D. (1986) Metabolism
of 14C-DPX-H6573 in rats. Unpublished Report No. AMR-196-84 from
E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Arce, G.T., Matarese, C.C. & Sarrif, A.M. (1988) Mutagenicity
testing of INH-6573-21 in Salmonella typhimurium plate
incorporation assay. Unpublished Report No. 59-88 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Brock, W.J., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
Long-term feeding study in mice with INH-6573. Unpublished Report
No. 278-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Brock, W.J., Vick, D.A. & Chromey, N.C. (1988) Closed-patch
repeated insult dermal sensitization study (Buehler method) with
INH-6573-21 in guinea pigs. Unpublished Report No. 34-88 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Cheng, T. (1986) Rat metabolism study of [Triazole-3-14C]
DPX-H6573. Unpublished final report 6129-128 from Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Chromey, N.C., Horst, A.L., McCooey, K.T. & Sarrif, A.M. (1983)
Unscheduled DNA synthesis/rat hepatocytes in vitro. Unpublished
Report No. 209-83 from Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Donovan, S.M. & Irr, J.D. (1982) Mutagenicity evaluation in
Salmonella typhimurium. Unpublished Report No. 611-82 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Farrow, M.G., Cortina, T. & Padilla-Nash, H. (1983) In vivo bone
marrow chromosome study in rats - H # 14,728 - Final Report.
Unpublished Report No. 201-636 (Haskell No. 288-83) by Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Hall, J.A., Dashiell, O.L. & Kennedy, G.L. (1984) Eye irritation
test in rabbits. Unpublished Report No. 582-82 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984a) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 444-83 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984b) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 142-84 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
McCooey, K.T., Chromey, N.C., Sarrif, A.M. & Hemingway, R.E. (1983)
CHO/HGPRT assay for gene mutation. Unpublished Report No. 449-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
O'Neal, F.O., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
One-year feeding study in dogs with INH-6573. Unpublished Report
No. 461-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Krahn, D.F. & Gibson, J.R. (1983)
Ninety-day feeding and one-generation reproduction study in rats
with Silane [Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-
ylmethyl) (INH-6573). Unpublished Report No. 483-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Drahn, D.F. & Aftosmis, J.G. (1984)
Four-week range finding and ninety-day feeding study in mice with
Silane [Bis(4-fluorophenyl)] (methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Revised Unpublished Report No. 341-83 1-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Rickard, R.W., Sykes, G.P., Kaplan, A.M. & Gibson,
J.R. (1986) Long-term feeding (combined chronic
toxicity/oncogenicity study) and two-generation, four litter
reproduction study in rats with INH-6573. Unpublished Report
No. 32-86 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Poindexter, G.L., Henry, J.E., Kenney, L.A., Burgess, B.A. &
Kennedy, G.L. (1984) Inhalation median lethal concentration
(LC50) of INH-6573-41 by EPA Protocol. Unpublished Report
No. 553-83 by Haskell Laboratory, Newark, DE, USA. Submitted to WHO
by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Redgate, D., Sarver, J.W. & Chromey, N.C. (1985) Approximate lethal
dose (ALD) of INH-6573-66 in rabbits. Unpublished Report No. 54-85
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Richard, R.W., Wood, C.K., Krahn, D.F. & Aftosmis, J.G. (1983)
Three-month feeding study in dogs with Silane
[Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Unpublished Report No. 461-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Sarver, W., Vick, D.A., Valentine, R., Chromey, N.C. & Kaplan, A.M.
(1986) Twenty-one dose dermal toxicity study with INH-6573-82 in
rabbits. Unpublished Report No. 744-86 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Solomon, H.M., Alvarez, L., Staples, R.E. & Hamill, J.C. (1984)
Developmental toxicity study in rabbits given INH-6573 by gavage on
Days 7-19 of gestation. Unpublished Report No. 333-84 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Sorg, R.M., Naismith, R.W. & Mathews, R.J. (1984) Micronucleus test
(MNT) OECD H # 15,314. Unpublished Report No. PH 309A-DU-001-84
from Pharmakon Research International (Haskell Report HLO-437-84).
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Turner, R.J., Kinney, L.A. & Chromey, N.C. (1985) Inhalation median
lethal concentration (LC50) of INH-6573 by EPA Guidelines.
Unpublished Report No. 1-85 by Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Vlachos, D.A., Covell, D.L. & Sarrif, A.M. (1986) Evaluation of
INH-6573-82 in the in vitro assay for chromosome aberrations in
human lymphocytes. Unpublished Report No. 745-86 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Burgess, B.A. & Kennedy, G.L. (1983) Acute oral test
in rats. Unpublished Report No. 78-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984a) Median lethal dose (LD50) in rats - EPA proposed
guidelines, Newark, DE, USA. Unpublished Report No. 433-83 by
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984b) Median lethal dose (LD50) in rats - EPA proposed
guidelines. Unpublished Report No. 412-83 by Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Burgess, B.A. & Kennedy, G.L. (1984c)
Ten-dose oral subacute test in rats. Unpublished Report No. 78-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Dashiell, O.L. & Kennedy, G.L. (1984d)
Primary skin irritation and sensitization test on guinea pigs.
Unpublished Report No. 626-82 from Haskell Laboratory, Newark, DE,
USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Wylie, C.N., Redgate, D., Warheit, D.B. & Chromey, N.C. (1985)
Approximate lethal dose (ALD) of INH-6573-66 in male and female
mice. Unpublished Report No. 531-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Zellers, J.E., Staples, R.E., Alvarez, L. & Kaplan, A.M. (1985)
Developmental toxicity study (supplemental) in rabbits dosed by
gavage on Days 7-19 of gestation. Unpublished Report No. 699-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Toxicological studies
Acute toxicity
The acute toxicity of flusilazole to several animal species is
given in Table 1. Signs of toxicity following oral administration
included weight loss, weakness, lethargy and, at higher doses,
laboured breathing, convulsions and prostration. Inhalation effects
were chiefly laboured breathing and lung sounds. Dermal
administration resulted only in some local erythema.
TABLE 1. RESULTS OF ACUTE TOXICITY ASSAYS WITH FLUSILAZOLE
SPECIES SEX ROUTE LD50a/ ALDa/ REFERENCES
LC50b ALCb
Technical flusilazole
Rat M oral 1500 Wylie et al. 1983
Rabbit M&F oral 350 Redgate et al. 1985
Mouse M oral 680 Wylie et al. 1985
F oral 1000 Wylie et al. 1985
Rat M oral 1110 Wylie et al. 1984a
F oral 674 Wylie et al. 1984a
Rabbit M&F dermal >2000 Gargus et al. 1983
Rat M inhal. 2.7b,c Poindexter et al. 1984
F inhal. 3.7b,c Poindexter et al. 1984
Rat M&F inhal. >5.0b Turner et al. 1985
Formulation - 40% flusilazole
Rat M oral 1865 Wylie et al. 1984b
F oral 1272 Wylie et al. 1984b
a mg/kg bw
b mg/l
c test material was 99% pure.
Short-term studies
Oral studies
Rat
Groups of six male Crl:CD rats were given 0 or 300 mg
flusilazole (95.5%) in corn oil/kg bw/day by oral gavage for 2 weeks
(5 days per week). After the tenth dose 3 rats/group were killed and
examined for histopathology. The remaining rats were killed after a
14-day recovery period. One treated rat died after the fifth dose
(found Day 7). The treated rats died lost weight during the first
week of treatment and showed alopecia, diarrhea, stained face and
perineal area, salivation and hypersensitivity during the treatment
period only. Liver vacuolation was seen in all treated animals. In
urinary bladder transitional epithelium hyperplasia and vacuolation
were seen in all sacrificed treated animals with an increase in
mitotic figures seen in two of three males killed immediately
following the treatment period. Two treated males had atypical
giant cells in the lumen of the seminiferous tubules and necrosis
and cellular degeneration of the germinal epithelium in the testes.
Hyperplasia and vacuolation of the transitional epithelium in the
kidney pelvis was seen in two treated males. Lesions tended to be
more severe in animals killed immediately after the treatment period
than in those from the recovery phase (Wylie et al. 1984c).
A 90-day feeding study was carried out in Charles River (CD)
rats with dietary levels of flusilazole (96.7%) of 0, 25, 125 and
375 ppm fed to groups of 16 rats/sex and 750 ppm fed to 10 rats/sex.
Groups of 10/sex/dose comprised the standard 90-day study while the
extra 6/sex/dose continued on diet for a one generation one litter
reproduction study and then received control diet for a 4-month
recovery phase. Body weight gains were reduced only in the females
given 750 ppm. There were no effects on food consumption, clinical
signs of toxicity, hematological parameters or urinalyses. Serum
cholesterol levels were increased in both males and females given
750 ppm. Absolute and relative liver weights were increased in both
sexes at 750 ppm. Mild urothelial hyperplasia was observed in
urinary bladders of 5 males and 8 females at 750 ppm and 1 male and
4 females at 375 ppm. After the 4-month recovery phase following
dosing with 0-375 ppm flusilazole, no evidence of bladder
hyperplasia was observed. At 750 ppm 5 males had liver lesions,
chiefly hepatocyte hypertrophy in 5 males and fatty
change/hepatocytolysis in 4 males. The NOAEL was 125 ppm (equal to
10 mg/kg bw/day) (Pastoor et al. 1983).
Mouse
Groups of 20 male and 20 female Crl:CD-1 mice were given diets
containing 0, 25, 75, 225, 500 or 1000 ppm of flusilazole (96.7%
pure). After four weeks on these diets 10 mice/sex/group were
sacrificed. The remaining 10 mice/sex/group were sacrificed after
90 days of treatment. There were no effects noted on body weight,
food consumption or clinical signs of toxicity in animals sacrificed
at either interval. At 90 days high-dose males and females had
slightly reduced Hb, Hct and RBC counts which were statistically
significant for at least one of these parameters in each sex. No
other hematological changes were noted. At both 4 weeks and 90 days
absolute and relative liver weights were increased in male mice at
225 ppm and higher and in female mice at 75 ppm and higher. The
absolute and relative kidney weights in males at 1000 ppm were
decreased. At 90 days hepatocellular hypertrophy was observed in
4/10 females at 1000 ppm and 8-10/10 males at 225, 500, and
1000 ppm. Hepatocellular vacuolar cytoplasmic changes were observed
in both sexes (incidences 0, 0, 0, 2, 7, 10 in males and 0, 0, 1, 3,
9, and 8 in females, at 0, 25, 75, 225, 500, and 1000 ppm,
respectively). Urothelial cell hyperplasia in urinary bladder was
observed in 1/10 males and 7/10 males at 500 and 1000 ppm,
respectively, and 2/10, 3/10, and 6/10 females at 225, 500 and
1000 ppm, respectively. No treatment-related kidney lesions were
observed. Tissues from animals killed at 4 weeks were not examined.
The NOAEL in this study was 25 ppm (equal to 4.5 mg/kg bw/day)
(Pastoor et al. 1984).
Dog
Four beagle dogs/sex/group were given diet containing 0, 25,
125, or 750/500 ppm flusilazole (93% pure) for 3 months. At the
highest dose level the dogs were given diet containing 750 ppm
flusilazole for 3 weeks, control diet for one week and then a dose
level of 500 ppm for the remainder of the study. The dose level was
reduced because of body weight loss and reduced food intake at
750 ppm. At 500 ppm the males gained a mean of 0.2 kg while the
females lost a mean of 0.5 kg body weight. The other two groups
were comparable to controls (gains of 1.6-1.9 kg in both sexes).
Food consumption remained lower than controls at the 750/500 ppm
dose level. Clinical signs related to treatment were observed in
one male and two females at 500 ppm (weakness or tremors). One
female at 25 ppm was sacrificed in extremis week 5. No other
animal died. In males at 750/500 ppm WBC count and monocyte counts
were slightly but statistically significantly increased and
cholesterol levels, total protein and albumen levels were lower than
controls. In both sexes at 750/500 ppm and males at 125 ppm alanine
amino transerase levels were higher than controls. Absolute and
relative liver weights were increased in both sexes at 750/500 ppm
and were considered related to treatment. Other organ weight
differences were probably related to the decreased body weights at
750/500 ppm. Mild mucosal hyperplasia in the urinary bladder was
observed in all males and females at 500 ppm and in 2 males at
125 ppm. In the stomach, hyperlasia of lymphoid follicles in the
pyloric granular mucosa was observed in 0/4, 3/4, 4/3, and 4/4 males
and 0/4, 0/4, 3/4, and 4/4 females at 0, 25, 125, and 750/500 ppm,
respectively. On the basis of this effect in stomach a NOAEL was
not demonstrated in this study (Richard et al. 1983).
Groups of 5 beagle dogs/sex were given diets containing 0, 5,
20, or 75 ppm flusilazole (95.8% pure) for one year. Although all
treated groups of females had lower weight gains than controls, this
did not appear to be treatment-related. No body weight effects were
seen in males. No treatment-related effects were noted in food
consumption or clinical signs of toxicity. WBC counts were higher
in both sexes at 75 ppm than in the controls. Alkaline phosphates
levels were higher than controls and cholesterol levels lower in
both sexes at 75 ppm but the difference was statistically
significant only in males. The 75 ppm males also had lower total
protein and albumen levels in serum. Relative liver weights were
increased in both sexes at 75 ppm and relative kidney weight in
females at 75 ppm. There was centrilobular hepatocyte enlargement
at 20 and 75 ppm, in both sexes with distinct vacuolation in 3
high-dose males. There were also a higher degree of centrilobular
inflammation in all of the high-dose dogs both with regard to
relative numbers of inflammatory cells and to increased number of
hepatic veins involved. All dogs in all groups including controls
had lymphoid hyperplasia in the gastric mucosa but the degree
increased with dose level and was moderate only in high-dose males
and females. There were no treatment-related kidney or urinary
bladder effects. The NOAEL in this study was 5 ppm (equal to
0.14 mg/kg bw/day) (O'Neal et al. 1985).
Dermal studies
Rabbit
A study in New Zealand White rabbits in which groups of 5-10
rabbits/sex were treated dermally with 0, 25, 250 or 2000 mg
flusilazole/kg bw/day (95.8% pure material) was terminated after 6
days exposure because of serve erythema with necrosis in animals at
250 and 2000 mg/kg bw/day. The erythema was first observed Day 4 of
test.
A range-finding study with 175 or 250 mg flusilazole (94.9%) kg
bw/day produced only mild erythema at both levels.
A 21-day study was performed in groups of 5 New Zealand White
rabbits/sex/dose at dose levels of 0, 1, 5, 25, and 200 mg
flusilazole (94.9% pure)/kg bw/day. For application the test
material was moistened with distilled water. The exposure sites
were occluded for 6 hours and then washed free of remaining test
material. There were no apparent treatment-related effects on body
weights: hematology; clinical chemistry; or liver, kidney and spleen
weights. At 200 mg/kg bw/day, four males and 2 females had
persistent mild erythema beginning Days 6 to 12. At termination
slight to mild diffuse hyperplasia/thickening of epidermis was
observed in the skin of one male and two females at 25 mg/kg bw/day
and three males and two females at 200 mg/kg bw/day (Sarver et al.
1986).
Long-term/carcinogenicity studies
Rats
Groups of 70 male and 70 female Crl:CD(SD)BR rats were given
diets containing 0, 10, 50 or 250 ppm flusilazole (95.6% pure) for
two years. Additionally, groups of 10/sex were given the same dose
levels for 6 months and 1 year. After about 100 days on test, 20
rats/sex/group were mated for the reproduction phase of the study.
These rats were returned to the long-term study after weaning of the
second litter. Body weights, food consumption and clinical
condition were monitored throughout the study. Hematology and
clinical chemistry parameters were examined at 3, 6, 9, 12 and 24
months on fasted animals. The animals sacrificed at 6 months were
examined for bladder lesions only. About 40 tissues/organs were
examined for control and high-dose animals and liver, bladder,
kidney, lung and gross lesions for the low and intermediate dose
groups sacrificed after one year. For animals that died on test or
were sacrificed at two years all tissues from all animals in all
groups were examined.
Survival to two years was 39-57% in the various groups with no
treatment-relationship. Survival did not drop below 50% in any
group until Week 98. The high-dose males had slightly lower body
weights from 6 months to the end of the study but there was no
similar effect in females. There were no treatment-related effects
on food consumption, clinical signs of toxicity, hematology,
clinical chemistry or urinalysis. In females, relative liver
weights were increased at both one and two years at 250 ppm and at
one year only at 50 ppm. No effect was observed in males. No
treatment-related effects on the urinary bladder were observed at 6
months. After one year there was an increased incidence of
hepatocellular hypertrophy in females (0/10, 0/10, 1/10, and 7/10 at
0, 10, 50, and 250 ppm, respectively) and at 250 ppm polyploidy
(increased number of multinuclear hepatocytes) was observed in two
females with none in any other group. There were no liver changes in
males at one year. There was an increased incidence of
hydronephrosis in males (0/10, 0/10, 3/10, and 4/10 at 0, 10, 50 and
250 ppm). Urothelial hyperplasia in renal pelvis was observed in
one male at 50 pm and one at 250 ppm, but not in any other group.
Treatment-related lesions were not observed in urinary bladders of
males. Females did not show kidney or bladder lesions after one
year. Among rats dying on test or sacrificed after two years there
were increased incidences in livers of females of acidophilic foci
of cellular alterations (3, 3, 4, and 13 at 0, 10, 50, and 250 ppm,
respectively) and diffuse fatty change (9, 9, 12, and 23,
respectively, in 63-68 /group). There were no treatment-related
liver lesions in males. In females, there was an increased
incidence of pyelonephritis in kidney (3, 3, 8, and 10,
respectively), but no treatment-related lesions were observed in
males. The only tumours which were increased in incidence in treated
groups were squamous cell carcinomas of the oral and nasal cavities
in males (0/66, 1/63, 0/67, and 3/64, respectively). Historical
control data from six two-year feeding studies showed five studies
which no squamous cell carcinomas of oral or nasal origin while the
remaining study had an incidence of 2/60. These data suggest that
these tumours may occur randomly as clusters and that the incidence
in this study was not treatment-related. There was, therefore, no
evidence of oncogenicity. It is noted, however, that the effects
noted were relatively mild and higher doses might have been
tolerated. The NOAEL for non-neoplastic effects was 10 ppm (equal
to 0.45 mg/kg bw/day) (Pastoor et al. 1986).
Mouse
Groups of 80 Crl:CD-1(ICR)BR mice/sex were given a diet
containing 0, 5, 25, or 200 ppm flusilazole (93%-95.6% purity) for
18 months. Additional groups of 10/sex/dose were given the same
diets for 6 months. Survival to 18 months was 76-86% in males and
57-80% in females with the highest survival at the high-dose level.
There were no treatment-related effects on body weight, food
consumption, hematology or serum protein. There was a higher
incidence of skin sores in high-dose males and females but these
were mainly ear lesions which were noted at the beginning of the
study and appeared to be related to the animal identification
process. High-dose males also had an increased incidence of ruffled
fur. Absolute and relative liver weights were increased in males at
200 ppm at both 6 and 18 months and in 200 ppm females at 6 months.
Absolute kidney weight only was decreased in females at 200 ppm at
18 months but the biological significance of this observation was
not clear. There were no treatment-related lesions in the kidney.
In the liver there was an increased incidence of hepatocellular
fatty change in both males and females at 200 ppm at 18 months
(40/80 and 24/80, respectively, compared to 4/80 and 2/79 in male
and female controls. One 200 ppm male at 6 months also had this
lesion, although 8/80 males at the 25 ppm levels also had
hepatoceullar fatty changes. This incidence was not statistically
significantly different from controls. There was no treatment-
related increase in incidence of any tumour type in this study. The
NOAEL in this study was 25 ppm (equal to 3.4 mg/kg bw/day) (Brock
et al. 1985).
Reproduction study
Rat
Groups of 6 Crl:CD(SD)BR rats/sex/dose level from the 90 day
feeding study were mated within the same dose level for a period of
15 days. Dose levels were 0, 25, 125, and 375 ppm flusilazole
(96.7% pure). The females were examined daily for evidence of a
copulation plug. After the mating period, the females were housed
individually.
The number of pregnant females/group was 3-6. The total number
of pups born did not show a treatment-related effect but two of the
five litters at 375 ppm were all dead at birth. The remaining three
litters at this dose level were 93-100% viable similar to the
litters in the other groups including controls. Mean pup weights at
Day 4 were lower in the 375 ppm group than the other three groups.
At 375 ppm (equal to 29 mg/kg bw/day) pup weights remained slightly
lower than the other groups to weaning (Pastoor et al. 1983).
Because of the small group size, the usefulness of this study
is limited.
Groups of 20 Crl:CD(SD)BR rats/sex/dose level from the chronic
toxicity/oncogenicity study were mated within the same dose level
for a period of 15 days after a dosing period of about 100 days.
Dose levels were 0, 10, 50, and 150 ppm flusilazole (95.6% pure).
The females were examined daily throughout the mating period for
evidence of a copulatory plug. At the end of the mating period the
females were housed individually and allowed to give birth and raise
their litters to weaning. About one week after weaning of the F1a
litters the females were sacrificed without pathological
examination. At weaning of the F1b litters 20 weanlings/sex/group
were selected as parents for the F2 generation. These weanlings
were maintained on the same diets as their parents for a 90 day
period before mating. Mating procedures were the same for the F0 -
F1 litters. It is stated that sibling matings were avoided. For
each litter the number of pups born, the number of pups born alive,
the number alive at 24 hours, 4 days, 12 days and 21 days, the
litter weight at 24 hours, and 4 days and the weight of male and
female pups at 21 days were recorded. At Day 4 all litters with
more than 10 pups were culled to 10, with equal numbers of males and
females where possible. Pups found dead were discarded. At weaning
of the F2b pups 10/sex/group were randomly selected and necropsied.
Liver, kidney, testes and brain were weighed and about 26
tissues/organs were examined for histopathology.
Food consumption and body weights were recorded only for the
pre-mating periods for the F0 and F1b animals. There were no
consistent differences during this period in F0 animals but F1b
males given 250 ppm had lower body weights. No differences in food
consumption were noted. Pregnancy rates were variable in all mating
periods but with no dose-relationship. There was no evidence of a
treatment-related effect on male reproductive parameters. There was
some indication that gestation length might be longer in the
high-dose group but this could not be determined unequivocally
because of the number of animals for which gestation lengths could
not be determined. The number of pups dead at birth was increased
in the 250 ppm group in all litters and in the 50 ppm group in the
F2a litter. Survival to Day 4 was reduced at 250 ppm in the F1a,
F1b, and F2a litters. Loss of the entire litter perinatally was
observed in 2, 2, 2 and 1 dams at 250 ppm in the F1a, F1b, F2a and
F2b litters, respectively, and in 1 dam at 50 ppm in the F2a
litter. Survival after 4 days was similar in all groups. The sex
of pups is indicated only at weaning after culling of the litters so
the sex ratio cannot be determined. Pup weights were similar in all
groups except for reduced weights at weaning in male and female pups
in the F1b and F2a litters. In the F2b weanling pups the absolute
and relative liver weights in males in the 250 ppm group were
increased. No effect was observed in females. The only
pathological lesion of note was hydronephrosis in female weanlings.
The incidence of unilateral and bilateral lesions combined was 1/10,
4/10, 3/10, and 5/10 at 0, 10, 50, and 250 ppm, respectively. The
incidences in the 10 and 250 ppm groups are slightly higher than
those observed in 10 historical control groups over the period
1982-1986. However, neither the severity nor the incidence of the
lesions showed a dose-related pattern so it is unlikely that this is
treatment-related effect. The NOAEL in this study was 10 ppm (equal
to 0.85 mg/kg bw/day on the basis of pre-mating intake data)
(Pastoor et al. 1986).
Special studies on mutagenicity
Flusilazole was negative in seven mutagenicity studies (Table
2).
Special studies on teratology
Rat
In a pilot study in which groups of seven pregnant rats were
given doses of 0, 100 or 300 mg flusilazole (99% pure)/kg bw/day
Days 7-16 of gestation, 300 mg/kg bw was overtly toxic and 100 mg/kg
bw slightly toxic to the dams. Both levels were embryotoxic and
300 mg/kg bw resulted in cleft palates in about 51% of the fetuses
in each litter.
TABLE 2. RESULTS OF MUTAGENICITY ASSAYS ON FLUSILAZOLE
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Ames testa Salmonella typhimurium 1-250 ug/plate Negative Donovan & Irr, 1982
TA1535, TA1537, TA98, (90%)
TA100
Ames testa Salmonella typhimurium 5-250 ug/plate Negative Arce et al. 1988
TA1535, TA97, TA98, (97.7%)
TA100
Unscheduled DNA Rat hepatocytes Crl (SD) 1 x 10-5 - Negative Chromey et al. 1983
synthesis 1.1 x 102 mM
(95.5%)
CHO/HGPRT assay Chinese hamster ovary 0.04 - 0.15 mM Negative McCooey et al. 1983
for gene mutationa cells (CHO-K1/BH4 clone) 0.05 - 0.275 mM
with and without
activation,
respectively
(95.5%)
Micronucleus test Mouse (CD-1) 375 mg/kg bw in Negative Sorg et al. 1984
(bone marrow) corn oil
(91.5%)
Chromosome aberration Human lymphocytes 1.7 - 100 ug/ml Negative Vlachos et al. 1986
assaya (94.85%)
TABLE 2 (CONTD).
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Chromosome assay Rat (Crl:CD (SD)) 50 - 500 mg/kg bw in Negative Farrow et al. 1983
(bone marrow) corn oil (not given)
a with and without metabolic activation.
Groups of 25 female Crl:CD(SD)BR rats mated to males of the
same strain were given doses of 0, 10, 50, or 250 mg flusilazole
(95.6%)/kg bw/day in corn oil by gavage Days 7-16 of gestation. The
day a copulation plug was observed was designated Day 1 of
gestation. Body weight, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation, the
rats were killed and examined for gross pathology, liver and uterine
weights, and numbers of corpora lutea, implantations, live and dead
fetuses. The uterus of any apparently non-pregnant females was
stained with ammonium sulfide to detect for early resorptions. The
fetuses were weighed and examined for external, visceral and
skeletal abnormalities.
Chromodacryorrhea, chromorhinorrhea, wet underbodies, or wet or
yellow or brown stained perineal areas were observed at the
250 mg/kg bw dose level with all but two females showing at least
one of these signs. The severity increased with continued dosing
and decreased following cessation of dosing. Some females at this
dose level had red vaginal discharges or stains and there was a
slightly increased incidence of focal alopecia. Two of the dams
died (Days 11 and 18). At 50 mg/kg bw one dam had red vaginal
discharge and one ruffled abdominal fur. Food consumption was
reduced during dosing at 50 and 250 mg/kg bw. At sacrifice relative
liver weights were increased in the dams at 50 and 250 mg/kg bw. At
250 mg/kg bw mean fetal body weight was reduced, incidence of
resorptions was increased and the number of live fetuses per litter
was reduced. Stunted fetuses were observed only in treated groups
at incidences of 1, 4, and 3 fetuses at 10, 50, and 250 mg/kg bw.
Cleft palate and absence of renal papilla were observed in fetuses
(12% and 9%, respectively) from dams given 250 mg/kg bw but not at
lower doses. At 50 mg/kg bw/day two fetuses (from 2 litters) had no
innominate artery and one (from the same litter as one of the
fetuses with no innominate artery) had a great vessel malformation
in the heart. These lesions were not seen in any other group so
were probably incidental. In all groups including controls there
was a high incidence of hydrocephalus and/or dilated lateral
ventricles of the brain. The incidence was not dose-related and no
hydrocephalus was observed in subsequent studies so this finding was
not considered to be treatment-related. The incidence of misaligned
sternebra, extra ossification centres in ribs and ossification delay
in sternebra were increased at all dose levels compared to controls.
There were increased incidences of rudimentary ribs at 50 and
250 mg/kg bw and extra ribs at 250 mg/kg bw. A NOEL was not
demonstrated in this study since skeletal variations were increased
at 10 mg/kg bw, the lowest dose tested. Maternal toxicity was seen
at 50 mg/kg bw. Teratogenic effects were observed at 250 mg/kg bw
(Lamontia et al. 1984a).
A second study included groups of 24 mated female Crl:CD(SD)BR
rats given doses of 0, 0.4, 2.0, 10.0, 50.0 or 250.0 (10 females) mg
flusilazole (95.6%)/kg bw/day in corn oil by gavage Days 7-16 of
gestation. The day a copulation plug was observed was designated
Day 1 of gestation. Body weight, food consumption and clinical
condition were monitored throughout the study. On Day 21 of
gestation the rats were killed and examined as in the study above.
At 50 and 250 mg/kg bw/day the dams had reduced body weight
gains during the early part of the dosing period but the difference
was statistically significant only at 250 mg/kg bw. Both groups had
reduced food intakes during dosing. Clinical signs of toxicity were
observed at 250 mg/kg bw: alopoecia, brown stains of face and limbs
and yellow staining of perineum. Relative liver weights were
increased in the 50 and 250 mg/kg bw groups. The incidence of early
resorptions was similar in all groups; however, the number of dams
with median or late resorptions was higher in groups given 10 mg/kg
bw or more. The mean number of live fetuses/litter was similar in
all groups. The incidence of stunted fetuses was increased at dose
levels of 10 mg/kg bw and higher. Cleft palate was observed in
fetuses from dams given 250 mg/kg bw but not at lower doses.
Absence of renal papilla was observed only in treated groups but
there was no dose-relationship and the observation was not
considered to be treatment-related. Hydrocephalus was not observed
in any group. Visceral (large renal pelvis and small renal papilla)
and skeletal (ribs anomalies) variations were observed at 10 mg/kg
bw and higher. Absence of the innominate artery was observed in
4(3) fetuses (litters) at 50 mg/kg bw and 1 fetus at 250 mg/kg bw.
Retarded development (delayed ossification of sternebra and
vertebral arch) was also evident at dose levels of 10 mg/kg bw and
higher. The incidence of red foci in bladder of fetuses was
increased at dose levels of 10 mg/kg bw and higher; however, these
lesions were non-specific and were considered to be of no biological
significance. The NOAEL in this study was 2 mg/kg bw/day (Lamontia
et al. 1984b).
In a dietary study, groups of 24 mated Crl:CD(SD)BR female rats
were given diets containing 0, 50, 100, and 300 or 900 ppm
flusilazole (94.8%) Days 7-16 of gestation. The day a copulation
plug was observed was designated Day 1 of gestation. Body weights,
food consumption and clinical condition were monitored throughout
the study. On Gestation Day 21 the rats were sacrificed and
examined for gross pathology, liver and uterine weights and uterine
contents. Fetuses were weighed, sexed and examined for external
visceral and skeletal abnormalities.
One female at 300 ppm died on Day 19 of gestation. The death
was not considered to be related to treatment. During Days 7-11 of
gestation at the beginning of dosing weight gains in all treated
groups were lower than in controls in a dose-related manner.
Significant reductions were noted Days 7-9 at 300 and 900 ppm. Food
consumption during dosing was lower than controls in all treated
groups but reached significant reductions only at 300 and 900 ppm.
There were no treatment-related clinical signs of toxicity.
Relative liver weights did not indicate a significant treatment-
related effect. Pregnancy rate was high in all groups. One dam at
900 ppm totally resorbed its litter. The incidence of early
resorptions was similar in all groups. However, there was an
increased incidence (dose-related) of median and late resorptions at
100 ppm and higher. Litter size was inversely dose-related and
significantly smaller than in controls in the 900 ppm group. Small
litters (less than 10 fetuses) were only seen at dose levels of
100 ppm and above. The 50 ppm group was similar to controls. The
incidence of stunted fetuses was increased at 300 and 900 ppm. Mean
fetal weights were not affected. No treatment-related malformations
were observed. No fetuses showed cleft palate and incidence of
absence of renal papilla was not dose or treatment-related. Two
fetuses from different litters in the 900 ppm group had slightly
domed heads but hydrocephalus was not confirmed on visceral
examination. There was a significant dose-related increase in
skeletal variations at dose levels of 100 ppm and higher with extra
ossification of the sternebra at all 3 dose levels, and rudimentary
ribs, extra ossification in cervical ribs and delayed ossification
in the vertebral arches (cervical) at 300 and 900 ppm. Red foci
were observed in the bladder of a few fetuses at all dose levels
with a slightly higher but not dose-related incidence at 100 ppm and
above. As in the previous study, these were not considered to be
biologically significant. The NOAEL in this study was 50 ppm (equal
to 4.6 mg/kg bw) (Alvarez et al. 1984).
A study of prenatal and postnatal toxicity in rats was
conducted in two phases. For the prenatal study (phase 1), groups
of 24 female Crl:CD(SD)BR rats were mated on a 1:1 basis with males
of the same strain. The day a copulation plug was observed was
designated Day 1 of gestation. On Days 7 through 16 the females
were given a dose of 0, 0.2, 0.4, 2, 10, or 100 mg flusilazole
(94.8% pure in 0.5% aqueous methyl cellulose)/kg bw by oral
intubation. Body weights, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation the
females of one group at each dose level were killed for examination
of the uterine contents. The extra control and 100 mg/kg bw groups
were killed Day 22 of gestation in order to determine whether
absence of renal papillae was a compound-related effect or an
anomaly.
Analyses of the dosing solutions indicates that the solution
prepared for the first day of dosing contained only 6-19% of the
nominal concentrations. Since solutions were prepared daily this
may have been given to only the one female/group that had reached
Gestation Day 7 on that day and only two of these (one at 2 and one
at 100 mg/kg) were pregnant. However, the next analyses given were
done on solutions prepared six days later and it is unknown whether
the technical error causing the low concentrations was corrected
before this date. Subsequent analyses indicated concentrations
75-110% of nominal. Assuming the worst case, 13-16 pregnant
females/group were given 1-6 doses which were about 10% of the
intended dose level (mean duration of low doses was 3.4-3.7 days).
In each group there were 7-8 pregnant females which did not receive
the low doses.
Body weight gains and food consumption were lower than controls
during dosing in the females given 100 mg/kg bw. Stained chin was
observed in many of the 100 mg/kg bw females and some showed
perinasal staining and/or wet perineum. Absolute and relative liver
weights were increased in dams at 100 mg/kg bw. Pregnancy rate was
83-96% in treated animals and 96-100% in controls; there was no dose
relationship. The number of early resorptions and dams with early
resorptions did not differ among the groups. However, there was an
increased incidence of median and late resorptions at 100 mg/kg bw.
The numbers of stunted fetuses and dams with stunted fetuses were
increased at 10 and 100 mg/kg bw (statistically significant only at
100 mg/kg bw). One fetus in each of the 100 mg/kg bw groups had no
renal papilla unilaterally. One other high-dose fetus had
malformations: no tail and imperforate anus (group killed Gestation
Day 22). There was an increased incidence of bladder foci in the
two 100 mg/kg bw groups compared to their respective controls.
However, the Gestation Day 22 control incidence was higher than the
100 mg/kg bw group killed on Gestation Day 21. Groups given 0.4,
2.0 and 10.0 mg/kg bw had slightly higher incidences of bladder
foci. Incidence of small papilla in the kidney was increased at 10
and 100 mg/kg bw but in the groups killed Gestation Day 21 the
increase was not dose-related. Incidence of distended ureter was
increased at 10 and 100 mg/kg bw when expressed as % affected per
litter. The number of litters affected was slightly higher than
controls in the Day 21 group at 100 mg/kg bw but not in the Day 22
group.
In the postnatal study (phase II), groups of 22 females
Crl:CD(SD)BR rats were mated and dosed as in phase I except that
only one control and one 100 mg/kg bw group were included. The
females were permitted to have their litters and raise them to
weaning. Body weights, food consumption and clinical signs were
monitored throughout the gestation and lactation periods. Offspring
were counted and weighted by sex Days 0, 4 (before and after
culling), 7 and 14 post partum. Pups were weighed individually Day
21. At weaning the pups were sacrificed and subjected to gross
necropsy.
The dosing solutions were adequately prepared in this phase of
the study. At 100 mg/kg bw five dams died; four on Gestation Day 24
or 25 (with apparently normal litters) and one four days post partum
after delivering a litter (all dead) Day 25. At 2.0 mg/kg bw one
dam died Day 24 during delivery. At 100 mg/kg body weight gain and
food consumption were reduced during the first few days of dosing
but were similar to controls for the remainder of the study. A few
dams at 100 mg/kg bw are stated to have shown pallor (4), hunching
(2), weakness (2) and dystocia (1) during the puturition and
lactation periods. There was an increase in mean gestational length
at 10 and 100 mg/kg bw. In controls, gestation did not exceed 22
days. In treated groups a gestation period of 23 days was seen in
2, 1, 4, 12, and 4 dams at 0.2, 0.4, 2.0, 10.0, and 100 mg/kg bw,
respectively, and at 100 mg/kg bw 11 dams did not deliver until 24
days gestation. At 100 mg/kg the mean gestation length was 23.7
days compared to 21.8 days in controls. Among the 11 high-dose dams
with parturition at Gestation Day 24, six had no live fetuses and
the other five had 1-5 live pups (mean 3) and 1-7 dead pups (mean
4.8). At 10 mg/kg bw the longest gestation period was 23 days and
there was no apparent effect on pup survival at birth. At this dose
four dams had one dead pup (of 6-15 pups in litter) at birth; two
delivered Day 22 and two delivered Day 23. This latter incidence
of dead pups at birth was similar to that in controls. Survival to
Day 4 was decreased at 100 mg/kg bw but was similar to controls at
the other dose levels. At 100 mg/kg bw 18% of the pups alive Day 0
of lactation had died by Day 4 compared to 0.4-1.7% of the pups in
the control and lower dose groups. There was no effect on survival
for the rest of the gestation period. Litter size was reduced at
100 mg/kg bw even when both live and dead pups in the litters were
considered. Mean litter size was slightly lower than controls in
the 10 mg/kg bw group reaching statistical significance after
culling. This was the result of an increased number of litters with
less than 10 pups (4 cf 0 in controls and 1-2 in the other treated
groups). Pup weights at birth were somewhat higher in the three
highest dose groups than in controls. At 2 and 10 mg/kg bw a few
litters with higher weights were among those with a 23 day gestation
length. At 100 mg/kg bw mean, live litter weights were not as high
as at 10 mg/kg bw mean, live litter weights were not as high as at
10 mg/kg bw but other effects confound any comparisons. No
treatment-related effects on sex were apparent. The only
alterations noted in the weanlings were dilatation of the renal
pelvis and distended ureter. The incidence was not unequivocally
dose-related although somewhat higher at 10 mg/kg bw/day. It is
stated that among the dead pups, absence of renal papilla was
observed in only 2/42 pups and small papilla (size 1 or 2) was seen
in only 4 pups (2 of each size). Most of the dead pups were in the
high-dose group so it is presumed that these observations were from
that group. In phase II the NOAEL was 2 mg/kg bw with the only
effects being a slightly longer gestation period with a higher mean
weight in a few animals (Alvarez et al. 1985a).
Rabbit
Groups of 18 New Zealand White female rabbits were inseminated
with semen from one of the two male rabbits of the same strain from
the same source. Day of insemination was designated Gestation Day
0. On Days 7-19 the females were given nominal doses of 0, 2, 5, or
12 mg flusilazole (94.8%)/kg bw/day by gavage in corn oil (actual
1.9, 4.8 and 10.1 mg/kg bw/day). There were two deaths during
treatment: one control female Day 19 and one high-dose female Day
16. Another control female died Day 23. Periodic anorexia was
observed in some rabbits in all groups and was not related to
treatment. No treatment-related effects were noted on clinical
observations, body weight, mean food consumption, abortions (2 in
controls, 3 in each of the treated groups), number of resorptions,
number of live or dead (none in any group) fetuses, fetal weights,
maternal liver weights or maternal pathology. The total number of
malformed fetuses was not increased by treatment. The incidence of
hydrocephalus was 1(1), 2(1), 4(2), and 4(3) fetuses (litters) in
the nominal 0, 2, 5, or 12 mg/kg bw/day level groups, respectively.
There was no increase in any other malformation nor of any
variation. In three subsequent studies hydrocephalus was observed
only in one fetus at 35 mg/kg bw/day and in none at any lower dose
or in controls. The incidence of hydrocephalus in this study,
therefore, was not confirmed and could not be regarded as related to
treatment. The NOAEL in this study was the nominal 12 mg/kg bw/day
level (actual 10.1 mg/kg bw/day) (Solomon et al. 1984).
A second study with 20 New Zealand White female rabbits/group
artificially inseminated with sperm from one of three male rabbits
tested dose levels of 0, 12, and 35 mg flusilazole (95%)/kg bw/day
administered Days 7-19 of gestation by gavage. In this study
pregnancy rates were low particularly in the two treated groups (not
dose-related). At 35 mg/kg bw/day, 2 of the does aborted and 10
totally resorbed their litters with only one doe with a live litter
at examination Day 29. There was an increased incidence of vaginal
discharge, discharge in cage and stained tail at this dose level as
well as an increased incidence of periodic anorexia. Since there
was only one litter, no teratologic effects could be determined.
One fetus did have hydrocephalus. Reproductive parameters at
12 mg/kg bw/day did not differ from controls and there was no
evidence of maternal toxicity at this level. The NOAEL in this
study was the nominal level of 12 mg/kg bw/day (actual 11 mg/kg
bw/day) (Zellers et al. 1985).
A pilot dietary study in New Zealand White rabbits included 7
females/group offered diets containing 0, 500, 1000, or 2000 ppm
flulilazole (94.8%) on Days 7-19 of gestation. Full results are not
given but pregnancy rate was low at 500 and 1000 ppm /3/7 at each
level and incidence of total resorption was high (4/7) at 2000 ppm.
Dietary levels of 0, 300, 600, and 1200 ppm flusilazole (94.8%)
were given to groups of 20 New Zealand White female rabbits days
7-19 of gestation following artificial insemination (day of
insemination designated Day 0 of gestation). Body weights, food
consumption and clinical signs of toxicity were monitored throughout
the study. The rabbits were sacrificed Day 29 of gestation and
examined for gross pathology, corpora lutea, numbers of live and
dead fetuses or resorption sites. The fetuses were examined for
external, visceral and skeletal abnormalities.
Body weights and food consumption were reduced during dosing.
There were no treatment-related clinical signs, pathological lesions
or changes in liver weights. Pregnancy rate was reduced in all
treated groups with the lowest rate in the high-dose group. The
number of dams with total resorptions was increased in the 600 and
1200 ppm groups. There were no treatment-related effects on mean
number of live fetuses, resorptions (in dams with fetuses) or fetal
weights. The number of litters available at 600 and 1200 ppm was
small (3 at each dose level), so the validity of the incidence of
fetal malformations and variations is uncertain at these levels.
There were no apparent effects at 300 ppm.
Since the pregnancy rate was reduced in all treated groups
another dietary study was performed with levels of 0, 30, 100, and
300 ppm flusilazole in diet offered Days 7-19 of gestation to groups
of 18 or 25 (300 ppm only) female New Zealand White rabbits
artificially inseminated with sperm from proven male rabbits. In
this study pregnancy rate was low in all groups but particularly in
the controls. In the 0 and 300 ppm groups 25% and 29%,
respectively, of the pregnant does totally resorbed their litters
but there were no total resorptions in the other two groups.
Because of the low number of dams with litters available in the
control group, the data in this study could not be adequately
assessed and a NOAEL cannot be determined (Alvarez et al. 1985b).
It has been suggested by the company that the reproductive
effects observed in these studies (reduced pregnancy and increased
total resorptions) were the result of inadequate technique during
artificial insemination. This might explain the effects observed in
the control group in the second dietary study.
Special studies on skin irritation and sensitization
Guinea Pig
In young adult Hartley guinea pigs, a range-finding study
indicated mild erythema, 24 hours after topical application to the
skin of the test material (90% pure flusilazole). The primary
irritation study conducted with 5 and 50% solutions in demethyl
phthalate did not demonstrate irritation in any of the 10 animals
exposed. Intradermal injections of 1% solutions of flusilazole (90%
pure) in dimethyl phthalate weekly for 4 weeks in the same animals
caused erythema and edema with necrotic centres at the injection
sites at 24 hours. Challenge with topical applications of 5 and 50%
solutions did not demonstrate sensitization (Wylie et al. 1984d).
In another study using 97.7% pure flusilazole, young adult
Duncan Hartley albino guinea pits (10/sex) were given 3 weekly
dermal applications of the test material (slightly moistened with
dimethyl phthalate) which was covered with plastic wrap for 6 hours.
No signs of irritation were observed in any of the animals after any
of these induction applications. Two weeks after the last induction
treatment, the guinea pigs were challenged with a dermal application
of test material on an untreated site which was then covered for 6
hours. No reaction was observed in any of the animals. A positive
control groups treated with 1-chloro,-2,4-dinitrobenzene (DNCB)
showed several erythema with necrosis after the second and third
induction applications and severe erythema after the challenge dose
two weeks later (Brock et al. 1988).
Special study of eye irritation
Rabbit
Two male New Zealand White rabbits were given doses of 0.01 ml
of undiluted flusilazole (90% pure) in the right conjunctival sac.
Both rabbits shoed mild conjunctival redness and chemosis at 1-4
hours. The washed eye also showed some discharge at one hour but no
other effects. The unwashed eye showed slight corneal opacity at
1-4 hours and slight cloudiness on biomicroscope examination at
24-48 hours. The eye was normal at 72 hours. The test material
produced only transient mild irritation (Hall et al. (1984).
COMMENTS
Flusilazole administered orally to rats was rapidly excreted in
urine and feces. The label (14C) from phenyl-labelled material was
excreted predominantly in feces, with a sex difference in that males
excreted higher amounts in feces than did females. However,
substantial variability existed. When triazole-labelled material
was given, the 14C was recovered predominantly in urine. Although
one study indicated a sex difference, with males excreting more in
urine than females, a sex difference was not observed in a second
study with more animals. Tissue levels were low. The highest
levels were found in the carcass (about 2% of the dose after 96-120
hours). Flusilazole was extensively metabolized in the rat.
Recovered parent compound accounted to 2-11% of the dose and was
recovered almost exclusively in feces.
Flusilazole has a low acute toxicity in the species examined.
In short-term (90-day) studies, urothelial hyperplasia in the
urinary bladder was observed in rats, mice and dogs at dietary
levels of 375, 225, and 125 ppm, respectively. Liver lesions,
including hepatocellular hypertrophy and hepatocellular vacuolar
cytoplasmic changes, were also observed in the 90-day studies in
rats and mice (at 750 and 225 ppm, respectively) and in a one-year
study in dogs at 20 ppm.
In a one-year feeding study in beagle dogs, centrilobular
hepatocyte enlargement was observed at 20 and 75 ppm. The NOAEL was
5 ppm, equal to 0.14 mg/kg bw/day.
In long-term feeding studies in rats and mice no bladder
lesions were observed. There was urothelial hyperplasia in the
renal pelvis in 1 of 10 male rats at 50 ppm and 1 of 10 male rats at
250 ppm for one year, but in none of the rats (7 of 10) given
250 ppm at one year (hepatocellular hypertrophy) and two years
(acidophilic foci of cellular alteration: 3/66, 6/63, 4/68, and
13/65 at 0, 10, 50, and 250 ppm, respectively). Male and female
mice given 200 ppm for 18 months developed hepatocellular fatty
changes (40/80 and 24/80, respectively, versus 4/80 and 2/70 in
respective controls). This effect was also reported in 8/80 male
mice at 25 ppm, which was not statistically significantly different
from controls. There was no evidence of oncogenicity in either
species. The NOAEL in the rat study was 10 ppm (equal to 0.45 mg/kg
bw/day). The NOAEL in the mouse study was 5 ppm (equal to
0.79 mg/kg bw/day).
In a 2-generation, 2 litters per generation reproduction study
in rats, flusilazole caused perinatal mortality in both F1 and F2
litters of dams given dietary levels of 250 ppm. There was some
indication of increased gestation length at this dose level, an
effect which was also observed in a post-natal toxicity study in
rats at doses of 10 mg/kg bw/day (gavage) and higher. Perinatal
mortality was observed at 100 mg/kg bw/day in this study. The
biological significance of prolonged gestation in this study is
uncertain. The NOAEL was 10 ppm (equal to 0.895 mg/kg bw/day) but
not at lower doses. The NOAEL in rats was 2 mg/kg bw/day. No
teratogenic effects were seen in rabbits. The NOAEL was 100 ppm
(equal to 2.8 mg/kg bw/day) based on maternal toxicity.
After reviewing all available in vitro and in vivo
short-term tests, the Meeting concluded that there was no evidence
of genotoxicity.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effect
Mouse: 25 ppm in the diet, equal to 3.4 mg/kg bw/day
Rat: 10 ppm in the diet, equal to 0.45 mg/kg bw/day
Dog: 5 ppm in the diet, equal to 0.14 mg/kg bw/day.
Estimate of acceptable daily intake for humans
0-0.001 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
1. Observations in humans.
2. Toxicity of the biotransformation products.
REFERENCES
Alvarez, L., Krauss, W.C. & Staples, R.E. (1984) Developmental
toxicity study in rats given INH-6573-66 in the diet on Days 7-16 of
gestation. Unpublished Report No. 432-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., & Kaplan, A.M. (1985a) INH-6573.
Prenatal and postnatal toxicity study in rats dosed by gavage on
Days 7-16 of gestation. Unpublished Report No. 654-85 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., Driscoll, C.D. & Kaplan, A.M. (1985b)
INH-6573. Developmental toxicity study in rabbits treated by diet
on Days 7-19 of gestation. Unpublished Report No. 337-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Anderson, J.J., Stadalius, M.A. & Schlueter, D.D. (1986) Metabolism
of 14C-DPX-H6573 in rats. Unpublished Report No. AMR-196-84 from
E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Arce, G.T., Matarese, C.C. & Sarrif, A.M. (1988) Mutagenicity
testing of INH-6573-21 in Salmonella typhimurium plate
incorporation assay. Unpublished Report No. 59-88 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Brock, W.J., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
Long-term feeding study in mice with INH-6573. Unpublished Report
No. 278-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Brock, W.J., Vick, D.A. & Chromey, N.C. (1988) Closed-patch
repeated insult dermal sensitization study (Buehler method) with
INH-6573-21 in guinea pigs. Unpublished Report No. 34-88 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Cheng, T. (1986) Rat metabolism study of [Triazole-3-14C]
DPX-H6573. Unpublished final report 6129-128 from Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Chromey, N.C., Horst, A.L., McCooey, K.T. & Sarrif, A.M. (1983)
Unscheduled DNA synthesis/rat hepatocytes in vitro. Unpublished
Report No. 209-83 from Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Donovan, S.M. & Irr, J.D. (1982) Mutagenicity evaluation in
Salmonella typhimurium. Unpublished Report No. 611-82 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Farrow, M.G., Cortina, T. & Padilla-Nash, H. (1983) In vivo bone
marrow chromosome study in rats - H # 14,728 - Final Report.
Unpublished Report No. 201-636 (Haskell No. 288-83) by Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Hall, J.A., Dashiell, O.L. & Kennedy, G.L. (1984) Eye irritation
test in rabbits. Unpublished Report No. 582-82 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984a) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 444-83 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984b) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 142-84 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
McCooey, K.T., Chromey, N.C., Sarrif, A.M. & Hemingway, R.E. (1983)
CHO/HGPRT assay for gene mutation. Unpublished Report No. 449-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
O'Neal, F.O., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
One-year feeding study in dogs with INH-6573. Unpublished Report
No. 461-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Krahn, D.F. & Gibson, J.R. (1983)
Ninety-day feeding and one-generation reproduction study in rats
with Silane [Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-
ylmethyl) (INH-6573). Unpublished Report No. 483-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Drahn, D.F. & Aftosmis, J.G. (1984)
Four-week range finding and ninety-day feeding study in mice with
Silane [Bis(4-fluorophenyl)] (methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Revised Unpublished Report No. 341-83 1-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Rickard, R.W., Sykes, G.P., Kaplan, A.M. & Gibson,
J.R. (1986) Long-term feeding (combined chronic
toxicity/oncogenicity study) and two-generation, four litter
reproduction study in rats with INH-6573. Unpublished Report
No. 32-86 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Poindexter, G.L., Henry, J.E., Kenney, L.A., Burgess, B.A. &
Kennedy, G.L. (1984) Inhalation median lethal concentration
(LC50) of INH-6573-41 by EPA Protocol. Unpublished Report
No. 553-83 by Haskell Laboratory, Newark, DE, USA. Submitted to WHO
by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Redgate, D., Sarver, J.W. & Chromey, N.C. (1985) Approximate lethal
dose (ALD) of INH-6573-66 in rabbits. Unpublished Report No. 54-85
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Richard, R.W., Wood, C.K., Krahn, D.F. & Aftosmis, J.G. (1983)
Three-month feeding study in dogs with Silane
[Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Unpublished Report No. 461-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Sarver, W., Vick, D.A., Valentine, R., Chromey, N.C. & Kaplan, A.M.
(1986) Twenty-one dose dermal toxicity study with INH-6573-82 in
rabbits. Unpublished Report No. 744-86 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Solomon, H.M., Alvarez, L., Staples, R.E. & Hamill, J.C. (1984)
Developmental toxicity study in rabbits given INH-6573 by gavage on
Days 7-19 of gestation. Unpublished Report No. 333-84 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Sorg, R.M., Naismith, R.W. & Mathews, R.J. (1984) Micronucleus test
(MNT) OECD H # 15,314. Unpublished Report No. PH 309A-DU-001-84
from Pharmakon Research International (Haskell Report HLO-437-84).
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Turner, R.J., Kinney, L.A. & Chromey, N.C. (1985) Inhalation median
lethal concentration (LC50) of INH-6573 by EPA Guidelines.
Unpublished Report No. 1-85 by Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Vlachos, D.A., Covell, D.L. & Sarrif, A.M. (1986) Evaluation of
INH-6573-82 in the in vitro assay for chromosome aberrations in
human lymphocytes. Unpublished Report No. 745-86 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Burgess, B.A. & Kennedy, G.L. (1983) Acute oral test
in rats. Unpublished Report No. 78-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984a) Median lethal dose (LD50) in rats - EPA proposed
guidelines, Newark, DE, USA. Unpublished Report No. 433-83 by
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984b) Median lethal dose (LD50) in rats - EPA proposed
guidelines. Unpublished Report No. 412-83 by Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Burgess, B.A. & Kennedy, G.L. (1984c)
Ten-dose oral subacute test in rats. Unpublished Report No. 78-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Dashiell, O.L. & Kennedy, G.L. (1984d)
Primary skin irritation and sensitization test on guinea pigs.
Unpublished Report No. 626-82 from Haskell Laboratory, Newark, DE,
USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Wylie, C.N., Redgate, D., Warheit, D.B. & Chromey, N.C. (1985)
Approximate lethal dose (ALD) of INH-6573-66 in male and female
mice. Unpublished Report No. 531-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Zellers, J.E., Staples, R.E., Alvarez, L. & Kaplan, A.M. (1985)
Developmental toxicity study (supplemental) in rabbits dosed by
gavage on Days 7-19 of gestation. Unpublished Report No. 699-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
 Toxicological studies
Acute toxicity
The acute toxicity of flusilazole to several animal species is
given in Table 1. Signs of toxicity following oral administration
included weight loss, weakness, lethargy and, at higher doses,
laboured breathing, convulsions and prostration. Inhalation effects
were chiefly laboured breathing and lung sounds. Dermal
administration resulted only in some local erythema.
TABLE 1. RESULTS OF ACUTE TOXICITY ASSAYS WITH FLUSILAZOLE
SPECIES SEX ROUTE LD50a/ ALDa/ REFERENCES
LC50b ALCb
Technical flusilazole
Rat M oral 1500 Wylie et al. 1983
Rabbit M&F oral 350 Redgate et al. 1985
Mouse M oral 680 Wylie et al. 1985
F oral 1000 Wylie et al. 1985
Rat M oral 1110 Wylie et al. 1984a
F oral 674 Wylie et al. 1984a
Rabbit M&F dermal >2000 Gargus et al. 1983
Rat M inhal. 2.7b,c Poindexter et al. 1984
F inhal. 3.7b,c Poindexter et al. 1984
Rat M&F inhal. >5.0b Turner et al. 1985
Formulation - 40% flusilazole
Rat M oral 1865 Wylie et al. 1984b
F oral 1272 Wylie et al. 1984b
a mg/kg bw
b mg/l
c test material was 99% pure.
Short-term studies
Oral studies
Rat
Groups of six male Crl:CD rats were given 0 or 300 mg
flusilazole (95.5%) in corn oil/kg bw/day by oral gavage for 2 weeks
(5 days per week). After the tenth dose 3 rats/group were killed and
examined for histopathology. The remaining rats were killed after a
14-day recovery period. One treated rat died after the fifth dose
(found Day 7). The treated rats died lost weight during the first
week of treatment and showed alopecia, diarrhea, stained face and
perineal area, salivation and hypersensitivity during the treatment
period only. Liver vacuolation was seen in all treated animals. In
urinary bladder transitional epithelium hyperplasia and vacuolation
were seen in all sacrificed treated animals with an increase in
mitotic figures seen in two of three males killed immediately
following the treatment period. Two treated males had atypical
giant cells in the lumen of the seminiferous tubules and necrosis
and cellular degeneration of the germinal epithelium in the testes.
Hyperplasia and vacuolation of the transitional epithelium in the
kidney pelvis was seen in two treated males. Lesions tended to be
more severe in animals killed immediately after the treatment period
than in those from the recovery phase (Wylie et al. 1984c).
A 90-day feeding study was carried out in Charles River (CD)
rats with dietary levels of flusilazole (96.7%) of 0, 25, 125 and
375 ppm fed to groups of 16 rats/sex and 750 ppm fed to 10 rats/sex.
Groups of 10/sex/dose comprised the standard 90-day study while the
extra 6/sex/dose continued on diet for a one generation one litter
reproduction study and then received control diet for a 4-month
recovery phase. Body weight gains were reduced only in the females
given 750 ppm. There were no effects on food consumption, clinical
signs of toxicity, hematological parameters or urinalyses. Serum
cholesterol levels were increased in both males and females given
750 ppm. Absolute and relative liver weights were increased in both
sexes at 750 ppm. Mild urothelial hyperplasia was observed in
urinary bladders of 5 males and 8 females at 750 ppm and 1 male and
4 females at 375 ppm. After the 4-month recovery phase following
dosing with 0-375 ppm flusilazole, no evidence of bladder
hyperplasia was observed. At 750 ppm 5 males had liver lesions,
chiefly hepatocyte hypertrophy in 5 males and fatty
change/hepatocytolysis in 4 males. The NOAEL was 125 ppm (equal to
10 mg/kg bw/day) (Pastoor et al. 1983).
Mouse
Groups of 20 male and 20 female Crl:CD-1 mice were given diets
containing 0, 25, 75, 225, 500 or 1000 ppm of flusilazole (96.7%
pure). After four weeks on these diets 10 mice/sex/group were
sacrificed. The remaining 10 mice/sex/group were sacrificed after
90 days of treatment. There were no effects noted on body weight,
food consumption or clinical signs of toxicity in animals sacrificed
at either interval. At 90 days high-dose males and females had
slightly reduced Hb, Hct and RBC counts which were statistically
significant for at least one of these parameters in each sex. No
other hematological changes were noted. At both 4 weeks and 90 days
absolute and relative liver weights were increased in male mice at
225 ppm and higher and in female mice at 75 ppm and higher. The
absolute and relative kidney weights in males at 1000 ppm were
decreased. At 90 days hepatocellular hypertrophy was observed in
4/10 females at 1000 ppm and 8-10/10 males at 225, 500, and
1000 ppm. Hepatocellular vacuolar cytoplasmic changes were observed
in both sexes (incidences 0, 0, 0, 2, 7, 10 in males and 0, 0, 1, 3,
9, and 8 in females, at 0, 25, 75, 225, 500, and 1000 ppm,
respectively). Urothelial cell hyperplasia in urinary bladder was
observed in 1/10 males and 7/10 males at 500 and 1000 ppm,
respectively, and 2/10, 3/10, and 6/10 females at 225, 500 and
1000 ppm, respectively. No treatment-related kidney lesions were
observed. Tissues from animals killed at 4 weeks were not examined.
The NOAEL in this study was 25 ppm (equal to 4.5 mg/kg bw/day)
(Pastoor et al. 1984).
Dog
Four beagle dogs/sex/group were given diet containing 0, 25,
125, or 750/500 ppm flusilazole (93% pure) for 3 months. At the
highest dose level the dogs were given diet containing 750 ppm
flusilazole for 3 weeks, control diet for one week and then a dose
level of 500 ppm for the remainder of the study. The dose level was
reduced because of body weight loss and reduced food intake at
750 ppm. At 500 ppm the males gained a mean of 0.2 kg while the
females lost a mean of 0.5 kg body weight. The other two groups
were comparable to controls (gains of 1.6-1.9 kg in both sexes).
Food consumption remained lower than controls at the 750/500 ppm
dose level. Clinical signs related to treatment were observed in
one male and two females at 500 ppm (weakness or tremors). One
female at 25 ppm was sacrificed in extremis week 5. No other
animal died. In males at 750/500 ppm WBC count and monocyte counts
were slightly but statistically significantly increased and
cholesterol levels, total protein and albumen levels were lower than
controls. In both sexes at 750/500 ppm and males at 125 ppm alanine
amino transerase levels were higher than controls. Absolute and
relative liver weights were increased in both sexes at 750/500 ppm
and were considered related to treatment. Other organ weight
differences were probably related to the decreased body weights at
750/500 ppm. Mild mucosal hyperplasia in the urinary bladder was
observed in all males and females at 500 ppm and in 2 males at
125 ppm. In the stomach, hyperlasia of lymphoid follicles in the
pyloric granular mucosa was observed in 0/4, 3/4, 4/3, and 4/4 males
and 0/4, 0/4, 3/4, and 4/4 females at 0, 25, 125, and 750/500 ppm,
respectively. On the basis of this effect in stomach a NOAEL was
not demonstrated in this study (Richard et al. 1983).
Groups of 5 beagle dogs/sex were given diets containing 0, 5,
20, or 75 ppm flusilazole (95.8% pure) for one year. Although all
treated groups of females had lower weight gains than controls, this
did not appear to be treatment-related. No body weight effects were
seen in males. No treatment-related effects were noted in food
consumption or clinical signs of toxicity. WBC counts were higher
in both sexes at 75 ppm than in the controls. Alkaline phosphates
levels were higher than controls and cholesterol levels lower in
both sexes at 75 ppm but the difference was statistically
significant only in males. The 75 ppm males also had lower total
protein and albumen levels in serum. Relative liver weights were
increased in both sexes at 75 ppm and relative kidney weight in
females at 75 ppm. There was centrilobular hepatocyte enlargement
at 20 and 75 ppm, in both sexes with distinct vacuolation in 3
high-dose males. There were also a higher degree of centrilobular
inflammation in all of the high-dose dogs both with regard to
relative numbers of inflammatory cells and to increased number of
hepatic veins involved. All dogs in all groups including controls
had lymphoid hyperplasia in the gastric mucosa but the degree
increased with dose level and was moderate only in high-dose males
and females. There were no treatment-related kidney or urinary
bladder effects. The NOAEL in this study was 5 ppm (equal to
0.14 mg/kg bw/day) (O'Neal et al. 1985).
Dermal studies
Rabbit
A study in New Zealand White rabbits in which groups of 5-10
rabbits/sex were treated dermally with 0, 25, 250 or 2000 mg
flusilazole/kg bw/day (95.8% pure material) was terminated after 6
days exposure because of serve erythema with necrosis in animals at
250 and 2000 mg/kg bw/day. The erythema was first observed Day 4 of
test.
A range-finding study with 175 or 250 mg flusilazole (94.9%) kg
bw/day produced only mild erythema at both levels.
A 21-day study was performed in groups of 5 New Zealand White
rabbits/sex/dose at dose levels of 0, 1, 5, 25, and 200 mg
flusilazole (94.9% pure)/kg bw/day. For application the test
material was moistened with distilled water. The exposure sites
were occluded for 6 hours and then washed free of remaining test
material. There were no apparent treatment-related effects on body
weights: hematology; clinical chemistry; or liver, kidney and spleen
weights. At 200 mg/kg bw/day, four males and 2 females had
persistent mild erythema beginning Days 6 to 12. At termination
slight to mild diffuse hyperplasia/thickening of epidermis was
observed in the skin of one male and two females at 25 mg/kg bw/day
and three males and two females at 200 mg/kg bw/day (Sarver et al.
1986).
Long-term/carcinogenicity studies
Rats
Groups of 70 male and 70 female Crl:CD(SD)BR rats were given
diets containing 0, 10, 50 or 250 ppm flusilazole (95.6% pure) for
two years. Additionally, groups of 10/sex were given the same dose
levels for 6 months and 1 year. After about 100 days on test, 20
rats/sex/group were mated for the reproduction phase of the study.
These rats were returned to the long-term study after weaning of the
second litter. Body weights, food consumption and clinical
condition were monitored throughout the study. Hematology and
clinical chemistry parameters were examined at 3, 6, 9, 12 and 24
months on fasted animals. The animals sacrificed at 6 months were
examined for bladder lesions only. About 40 tissues/organs were
examined for control and high-dose animals and liver, bladder,
kidney, lung and gross lesions for the low and intermediate dose
groups sacrificed after one year. For animals that died on test or
were sacrificed at two years all tissues from all animals in all
groups were examined.
Survival to two years was 39-57% in the various groups with no
treatment-relationship. Survival did not drop below 50% in any
group until Week 98. The high-dose males had slightly lower body
weights from 6 months to the end of the study but there was no
similar effect in females. There were no treatment-related effects
on food consumption, clinical signs of toxicity, hematology,
clinical chemistry or urinalysis. In females, relative liver
weights were increased at both one and two years at 250 ppm and at
one year only at 50 ppm. No effect was observed in males. No
treatment-related effects on the urinary bladder were observed at 6
months. After one year there was an increased incidence of
hepatocellular hypertrophy in females (0/10, 0/10, 1/10, and 7/10 at
0, 10, 50, and 250 ppm, respectively) and at 250 ppm polyploidy
(increased number of multinuclear hepatocytes) was observed in two
females with none in any other group. There were no liver changes in
males at one year. There was an increased incidence of
hydronephrosis in males (0/10, 0/10, 3/10, and 4/10 at 0, 10, 50 and
250 ppm). Urothelial hyperplasia in renal pelvis was observed in
one male at 50 pm and one at 250 ppm, but not in any other group.
Treatment-related lesions were not observed in urinary bladders of
males. Females did not show kidney or bladder lesions after one
year. Among rats dying on test or sacrificed after two years there
were increased incidences in livers of females of acidophilic foci
of cellular alterations (3, 3, 4, and 13 at 0, 10, 50, and 250 ppm,
respectively) and diffuse fatty change (9, 9, 12, and 23,
respectively, in 63-68 /group). There were no treatment-related
liver lesions in males. In females, there was an increased
incidence of pyelonephritis in kidney (3, 3, 8, and 10,
respectively), but no treatment-related lesions were observed in
males. The only tumours which were increased in incidence in treated
groups were squamous cell carcinomas of the oral and nasal cavities
in males (0/66, 1/63, 0/67, and 3/64, respectively). Historical
control data from six two-year feeding studies showed five studies
which no squamous cell carcinomas of oral or nasal origin while the
remaining study had an incidence of 2/60. These data suggest that
these tumours may occur randomly as clusters and that the incidence
in this study was not treatment-related. There was, therefore, no
evidence of oncogenicity. It is noted, however, that the effects
noted were relatively mild and higher doses might have been
tolerated. The NOAEL for non-neoplastic effects was 10 ppm (equal
to 0.45 mg/kg bw/day) (Pastoor et al. 1986).
Mouse
Groups of 80 Crl:CD-1(ICR)BR mice/sex were given a diet
containing 0, 5, 25, or 200 ppm flusilazole (93%-95.6% purity) for
18 months. Additional groups of 10/sex/dose were given the same
diets for 6 months. Survival to 18 months was 76-86% in males and
57-80% in females with the highest survival at the high-dose level.
There were no treatment-related effects on body weight, food
consumption, hematology or serum protein. There was a higher
incidence of skin sores in high-dose males and females but these
were mainly ear lesions which were noted at the beginning of the
study and appeared to be related to the animal identification
process. High-dose males also had an increased incidence of ruffled
fur. Absolute and relative liver weights were increased in males at
200 ppm at both 6 and 18 months and in 200 ppm females at 6 months.
Absolute kidney weight only was decreased in females at 200 ppm at
18 months but the biological significance of this observation was
not clear. There were no treatment-related lesions in the kidney.
In the liver there was an increased incidence of hepatocellular
fatty change in both males and females at 200 ppm at 18 months
(40/80 and 24/80, respectively, compared to 4/80 and 2/79 in male
and female controls. One 200 ppm male at 6 months also had this
lesion, although 8/80 males at the 25 ppm levels also had
hepatoceullar fatty changes. This incidence was not statistically
significantly different from controls. There was no treatment-
related increase in incidence of any tumour type in this study. The
NOAEL in this study was 25 ppm (equal to 3.4 mg/kg bw/day) (Brock
et al. 1985).
Reproduction study
Rat
Groups of 6 Crl:CD(SD)BR rats/sex/dose level from the 90 day
feeding study were mated within the same dose level for a period of
15 days. Dose levels were 0, 25, 125, and 375 ppm flusilazole
(96.7% pure). The females were examined daily for evidence of a
copulation plug. After the mating period, the females were housed
individually.
The number of pregnant females/group was 3-6. The total number
of pups born did not show a treatment-related effect but two of the
five litters at 375 ppm were all dead at birth. The remaining three
litters at this dose level were 93-100% viable similar to the
litters in the other groups including controls. Mean pup weights at
Day 4 were lower in the 375 ppm group than the other three groups.
At 375 ppm (equal to 29 mg/kg bw/day) pup weights remained slightly
lower than the other groups to weaning (Pastoor et al. 1983).
Because of the small group size, the usefulness of this study
is limited.
Groups of 20 Crl:CD(SD)BR rats/sex/dose level from the chronic
toxicity/oncogenicity study were mated within the same dose level
for a period of 15 days after a dosing period of about 100 days.
Dose levels were 0, 10, 50, and 150 ppm flusilazole (95.6% pure).
The females were examined daily throughout the mating period for
evidence of a copulatory plug. At the end of the mating period the
females were housed individually and allowed to give birth and raise
their litters to weaning. About one week after weaning of the F1a
litters the females were sacrificed without pathological
examination. At weaning of the F1b litters 20 weanlings/sex/group
were selected as parents for the F2 generation. These weanlings
were maintained on the same diets as their parents for a 90 day
period before mating. Mating procedures were the same for the F0 -
F1 litters. It is stated that sibling matings were avoided. For
each litter the number of pups born, the number of pups born alive,
the number alive at 24 hours, 4 days, 12 days and 21 days, the
litter weight at 24 hours, and 4 days and the weight of male and
female pups at 21 days were recorded. At Day 4 all litters with
more than 10 pups were culled to 10, with equal numbers of males and
females where possible. Pups found dead were discarded. At weaning
of the F2b pups 10/sex/group were randomly selected and necropsied.
Liver, kidney, testes and brain were weighed and about 26
tissues/organs were examined for histopathology.
Food consumption and body weights were recorded only for the
pre-mating periods for the F0 and F1b animals. There were no
consistent differences during this period in F0 animals but F1b
males given 250 ppm had lower body weights. No differences in food
consumption were noted. Pregnancy rates were variable in all mating
periods but with no dose-relationship. There was no evidence of a
treatment-related effect on male reproductive parameters. There was
some indication that gestation length might be longer in the
high-dose group but this could not be determined unequivocally
because of the number of animals for which gestation lengths could
not be determined. The number of pups dead at birth was increased
in the 250 ppm group in all litters and in the 50 ppm group in the
F2a litter. Survival to Day 4 was reduced at 250 ppm in the F1a,
F1b, and F2a litters. Loss of the entire litter perinatally was
observed in 2, 2, 2 and 1 dams at 250 ppm in the F1a, F1b, F2a and
F2b litters, respectively, and in 1 dam at 50 ppm in the F2a
litter. Survival after 4 days was similar in all groups. The sex
of pups is indicated only at weaning after culling of the litters so
the sex ratio cannot be determined. Pup weights were similar in all
groups except for reduced weights at weaning in male and female pups
in the F1b and F2a litters. In the F2b weanling pups the absolute
and relative liver weights in males in the 250 ppm group were
increased. No effect was observed in females. The only
pathological lesion of note was hydronephrosis in female weanlings.
The incidence of unilateral and bilateral lesions combined was 1/10,
4/10, 3/10, and 5/10 at 0, 10, 50, and 250 ppm, respectively. The
incidences in the 10 and 250 ppm groups are slightly higher than
those observed in 10 historical control groups over the period
1982-1986. However, neither the severity nor the incidence of the
lesions showed a dose-related pattern so it is unlikely that this is
treatment-related effect. The NOAEL in this study was 10 ppm (equal
to 0.85 mg/kg bw/day on the basis of pre-mating intake data)
(Pastoor et al. 1986).
Special studies on mutagenicity
Flusilazole was negative in seven mutagenicity studies (Table
2).
Special studies on teratology
Rat
In a pilot study in which groups of seven pregnant rats were
given doses of 0, 100 or 300 mg flusilazole (99% pure)/kg bw/day
Days 7-16 of gestation, 300 mg/kg bw was overtly toxic and 100 mg/kg
bw slightly toxic to the dams. Both levels were embryotoxic and
300 mg/kg bw resulted in cleft palates in about 51% of the fetuses
in each litter.
TABLE 2. RESULTS OF MUTAGENICITY ASSAYS ON FLUSILAZOLE
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Ames testa Salmonella typhimurium 1-250 ug/plate Negative Donovan & Irr, 1982
TA1535, TA1537, TA98, (90%)
TA100
Ames testa Salmonella typhimurium 5-250 ug/plate Negative Arce et al. 1988
TA1535, TA97, TA98, (97.7%)
TA100
Unscheduled DNA Rat hepatocytes Crl (SD) 1 x 10-5 - Negative Chromey et al. 1983
synthesis 1.1 x 102 mM
(95.5%)
CHO/HGPRT assay Chinese hamster ovary 0.04 - 0.15 mM Negative McCooey et al. 1983
for gene mutationa cells (CHO-K1/BH4 clone) 0.05 - 0.275 mM
with and without
activation,
respectively
(95.5%)
Micronucleus test Mouse (CD-1) 375 mg/kg bw in Negative Sorg et al. 1984
(bone marrow) corn oil
(91.5%)
Chromosome aberration Human lymphocytes 1.7 - 100 ug/ml Negative Vlachos et al. 1986
assaya (94.85%)
TABLE 2 (CONTD).
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Chromosome assay Rat (Crl:CD (SD)) 50 - 500 mg/kg bw in Negative Farrow et al. 1983
(bone marrow) corn oil (not given)
a with and without metabolic activation.
Groups of 25 female Crl:CD(SD)BR rats mated to males of the
same strain were given doses of 0, 10, 50, or 250 mg flusilazole
(95.6%)/kg bw/day in corn oil by gavage Days 7-16 of gestation. The
day a copulation plug was observed was designated Day 1 of
gestation. Body weight, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation, the
rats were killed and examined for gross pathology, liver and uterine
weights, and numbers of corpora lutea, implantations, live and dead
fetuses. The uterus of any apparently non-pregnant females was
stained with ammonium sulfide to detect for early resorptions. The
fetuses were weighed and examined for external, visceral and
skeletal abnormalities.
Chromodacryorrhea, chromorhinorrhea, wet underbodies, or wet or
yellow or brown stained perineal areas were observed at the
250 mg/kg bw dose level with all but two females showing at least
one of these signs. The severity increased with continued dosing
and decreased following cessation of dosing. Some females at this
dose level had red vaginal discharges or stains and there was a
slightly increased incidence of focal alopecia. Two of the dams
died (Days 11 and 18). At 50 mg/kg bw one dam had red vaginal
discharge and one ruffled abdominal fur. Food consumption was
reduced during dosing at 50 and 250 mg/kg bw. At sacrifice relative
liver weights were increased in the dams at 50 and 250 mg/kg bw. At
250 mg/kg bw mean fetal body weight was reduced, incidence of
resorptions was increased and the number of live fetuses per litter
was reduced. Stunted fetuses were observed only in treated groups
at incidences of 1, 4, and 3 fetuses at 10, 50, and 250 mg/kg bw.
Cleft palate and absence of renal papilla were observed in fetuses
(12% and 9%, respectively) from dams given 250 mg/kg bw but not at
lower doses. At 50 mg/kg bw/day two fetuses (from 2 litters) had no
innominate artery and one (from the same litter as one of the
fetuses with no innominate artery) had a great vessel malformation
in the heart. These lesions were not seen in any other group so
were probably incidental. In all groups including controls there
was a high incidence of hydrocephalus and/or dilated lateral
ventricles of the brain. The incidence was not dose-related and no
hydrocephalus was observed in subsequent studies so this finding was
not considered to be treatment-related. The incidence of misaligned
sternebra, extra ossification centres in ribs and ossification delay
in sternebra were increased at all dose levels compared to controls.
There were increased incidences of rudimentary ribs at 50 and
250 mg/kg bw and extra ribs at 250 mg/kg bw. A NOEL was not
demonstrated in this study since skeletal variations were increased
at 10 mg/kg bw, the lowest dose tested. Maternal toxicity was seen
at 50 mg/kg bw. Teratogenic effects were observed at 250 mg/kg bw
(Lamontia et al. 1984a).
A second study included groups of 24 mated female Crl:CD(SD)BR
rats given doses of 0, 0.4, 2.0, 10.0, 50.0 or 250.0 (10 females) mg
flusilazole (95.6%)/kg bw/day in corn oil by gavage Days 7-16 of
gestation. The day a copulation plug was observed was designated
Day 1 of gestation. Body weight, food consumption and clinical
condition were monitored throughout the study. On Day 21 of
gestation the rats were killed and examined as in the study above.
At 50 and 250 mg/kg bw/day the dams had reduced body weight
gains during the early part of the dosing period but the difference
was statistically significant only at 250 mg/kg bw. Both groups had
reduced food intakes during dosing. Clinical signs of toxicity were
observed at 250 mg/kg bw: alopoecia, brown stains of face and limbs
and yellow staining of perineum. Relative liver weights were
increased in the 50 and 250 mg/kg bw groups. The incidence of early
resorptions was similar in all groups; however, the number of dams
with median or late resorptions was higher in groups given 10 mg/kg
bw or more. The mean number of live fetuses/litter was similar in
all groups. The incidence of stunted fetuses was increased at dose
levels of 10 mg/kg bw and higher. Cleft palate was observed in
fetuses from dams given 250 mg/kg bw but not at lower doses.
Absence of renal papilla was observed only in treated groups but
there was no dose-relationship and the observation was not
considered to be treatment-related. Hydrocephalus was not observed
in any group. Visceral (large renal pelvis and small renal papilla)
and skeletal (ribs anomalies) variations were observed at 10 mg/kg
bw and higher. Absence of the innominate artery was observed in
4(3) fetuses (litters) at 50 mg/kg bw and 1 fetus at 250 mg/kg bw.
Retarded development (delayed ossification of sternebra and
vertebral arch) was also evident at dose levels of 10 mg/kg bw and
higher. The incidence of red foci in bladder of fetuses was
increased at dose levels of 10 mg/kg bw and higher; however, these
lesions were non-specific and were considered to be of no biological
significance. The NOAEL in this study was 2 mg/kg bw/day (Lamontia
et al. 1984b).
In a dietary study, groups of 24 mated Crl:CD(SD)BR female rats
were given diets containing 0, 50, 100, and 300 or 900 ppm
flusilazole (94.8%) Days 7-16 of gestation. The day a copulation
plug was observed was designated Day 1 of gestation. Body weights,
food consumption and clinical condition were monitored throughout
the study. On Gestation Day 21 the rats were sacrificed and
examined for gross pathology, liver and uterine weights and uterine
contents. Fetuses were weighed, sexed and examined for external
visceral and skeletal abnormalities.
One female at 300 ppm died on Day 19 of gestation. The death
was not considered to be related to treatment. During Days 7-11 of
gestation at the beginning of dosing weight gains in all treated
groups were lower than in controls in a dose-related manner.
Significant reductions were noted Days 7-9 at 300 and 900 ppm. Food
consumption during dosing was lower than controls in all treated
groups but reached significant reductions only at 300 and 900 ppm.
There were no treatment-related clinical signs of toxicity.
Relative liver weights did not indicate a significant treatment-
related effect. Pregnancy rate was high in all groups. One dam at
900 ppm totally resorbed its litter. The incidence of early
resorptions was similar in all groups. However, there was an
increased incidence (dose-related) of median and late resorptions at
100 ppm and higher. Litter size was inversely dose-related and
significantly smaller than in controls in the 900 ppm group. Small
litters (less than 10 fetuses) were only seen at dose levels of
100 ppm and above. The 50 ppm group was similar to controls. The
incidence of stunted fetuses was increased at 300 and 900 ppm. Mean
fetal weights were not affected. No treatment-related malformations
were observed. No fetuses showed cleft palate and incidence of
absence of renal papilla was not dose or treatment-related. Two
fetuses from different litters in the 900 ppm group had slightly
domed heads but hydrocephalus was not confirmed on visceral
examination. There was a significant dose-related increase in
skeletal variations at dose levels of 100 ppm and higher with extra
ossification of the sternebra at all 3 dose levels, and rudimentary
ribs, extra ossification in cervical ribs and delayed ossification
in the vertebral arches (cervical) at 300 and 900 ppm. Red foci
were observed in the bladder of a few fetuses at all dose levels
with a slightly higher but not dose-related incidence at 100 ppm and
above. As in the previous study, these were not considered to be
biologically significant. The NOAEL in this study was 50 ppm (equal
to 4.6 mg/kg bw) (Alvarez et al. 1984).
A study of prenatal and postnatal toxicity in rats was
conducted in two phases. For the prenatal study (phase 1), groups
of 24 female Crl:CD(SD)BR rats were mated on a 1:1 basis with males
of the same strain. The day a copulation plug was observed was
designated Day 1 of gestation. On Days 7 through 16 the females
were given a dose of 0, 0.2, 0.4, 2, 10, or 100 mg flusilazole
(94.8% pure in 0.5% aqueous methyl cellulose)/kg bw by oral
intubation. Body weights, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation the
females of one group at each dose level were killed for examination
of the uterine contents. The extra control and 100 mg/kg bw groups
were killed Day 22 of gestation in order to determine whether
absence of renal papillae was a compound-related effect or an
anomaly.
Analyses of the dosing solutions indicates that the solution
prepared for the first day of dosing contained only 6-19% of the
nominal concentrations. Since solutions were prepared daily this
may have been given to only the one female/group that had reached
Gestation Day 7 on that day and only two of these (one at 2 and one
at 100 mg/kg) were pregnant. However, the next analyses given were
done on solutions prepared six days later and it is unknown whether
the technical error causing the low concentrations was corrected
before this date. Subsequent analyses indicated concentrations
75-110% of nominal. Assuming the worst case, 13-16 pregnant
females/group were given 1-6 doses which were about 10% of the
intended dose level (mean duration of low doses was 3.4-3.7 days).
In each group there were 7-8 pregnant females which did not receive
the low doses.
Body weight gains and food consumption were lower than controls
during dosing in the females given 100 mg/kg bw. Stained chin was
observed in many of the 100 mg/kg bw females and some showed
perinasal staining and/or wet perineum. Absolute and relative liver
weights were increased in dams at 100 mg/kg bw. Pregnancy rate was
83-96% in treated animals and 96-100% in controls; there was no dose
relationship. The number of early resorptions and dams with early
resorptions did not differ among the groups. However, there was an
increased incidence of median and late resorptions at 100 mg/kg bw.
The numbers of stunted fetuses and dams with stunted fetuses were
increased at 10 and 100 mg/kg bw (statistically significant only at
100 mg/kg bw). One fetus in each of the 100 mg/kg bw groups had no
renal papilla unilaterally. One other high-dose fetus had
malformations: no tail and imperforate anus (group killed Gestation
Day 22). There was an increased incidence of bladder foci in the
two 100 mg/kg bw groups compared to their respective controls.
However, the Gestation Day 22 control incidence was higher than the
100 mg/kg bw group killed on Gestation Day 21. Groups given 0.4,
2.0 and 10.0 mg/kg bw had slightly higher incidences of bladder
foci. Incidence of small papilla in the kidney was increased at 10
and 100 mg/kg bw but in the groups killed Gestation Day 21 the
increase was not dose-related. Incidence of distended ureter was
increased at 10 and 100 mg/kg bw when expressed as % affected per
litter. The number of litters affected was slightly higher than
controls in the Day 21 group at 100 mg/kg bw but not in the Day 22
group.
In the postnatal study (phase II), groups of 22 females
Crl:CD(SD)BR rats were mated and dosed as in phase I except that
only one control and one 100 mg/kg bw group were included. The
females were permitted to have their litters and raise them to
weaning. Body weights, food consumption and clinical signs were
monitored throughout the gestation and lactation periods. Offspring
were counted and weighted by sex Days 0, 4 (before and after
culling), 7 and 14 post partum. Pups were weighed individually Day
21. At weaning the pups were sacrificed and subjected to gross
necropsy.
The dosing solutions were adequately prepared in this phase of
the study. At 100 mg/kg bw five dams died; four on Gestation Day 24
or 25 (with apparently normal litters) and one four days post partum
after delivering a litter (all dead) Day 25. At 2.0 mg/kg bw one
dam died Day 24 during delivery. At 100 mg/kg body weight gain and
food consumption were reduced during the first few days of dosing
but were similar to controls for the remainder of the study. A few
dams at 100 mg/kg bw are stated to have shown pallor (4), hunching
(2), weakness (2) and dystocia (1) during the puturition and
lactation periods. There was an increase in mean gestational length
at 10 and 100 mg/kg bw. In controls, gestation did not exceed 22
days. In treated groups a gestation period of 23 days was seen in
2, 1, 4, 12, and 4 dams at 0.2, 0.4, 2.0, 10.0, and 100 mg/kg bw,
respectively, and at 100 mg/kg bw 11 dams did not deliver until 24
days gestation. At 100 mg/kg the mean gestation length was 23.7
days compared to 21.8 days in controls. Among the 11 high-dose dams
with parturition at Gestation Day 24, six had no live fetuses and
the other five had 1-5 live pups (mean 3) and 1-7 dead pups (mean
4.8). At 10 mg/kg bw the longest gestation period was 23 days and
there was no apparent effect on pup survival at birth. At this dose
four dams had one dead pup (of 6-15 pups in litter) at birth; two
delivered Day 22 and two delivered Day 23. This latter incidence
of dead pups at birth was similar to that in controls. Survival to
Day 4 was decreased at 100 mg/kg bw but was similar to controls at
the other dose levels. At 100 mg/kg bw 18% of the pups alive Day 0
of lactation had died by Day 4 compared to 0.4-1.7% of the pups in
the control and lower dose groups. There was no effect on survival
for the rest of the gestation period. Litter size was reduced at
100 mg/kg bw even when both live and dead pups in the litters were
considered. Mean litter size was slightly lower than controls in
the 10 mg/kg bw group reaching statistical significance after
culling. This was the result of an increased number of litters with
less than 10 pups (4 cf 0 in controls and 1-2 in the other treated
groups). Pup weights at birth were somewhat higher in the three
highest dose groups than in controls. At 2 and 10 mg/kg bw a few
litters with higher weights were among those with a 23 day gestation
length. At 100 mg/kg bw mean, live litter weights were not as high
as at 10 mg/kg bw mean, live litter weights were not as high as at
10 mg/kg bw but other effects confound any comparisons. No
treatment-related effects on sex were apparent. The only
alterations noted in the weanlings were dilatation of the renal
pelvis and distended ureter. The incidence was not unequivocally
dose-related although somewhat higher at 10 mg/kg bw/day. It is
stated that among the dead pups, absence of renal papilla was
observed in only 2/42 pups and small papilla (size 1 or 2) was seen
in only 4 pups (2 of each size). Most of the dead pups were in the
high-dose group so it is presumed that these observations were from
that group. In phase II the NOAEL was 2 mg/kg bw with the only
effects being a slightly longer gestation period with a higher mean
weight in a few animals (Alvarez et al. 1985a).
Rabbit
Groups of 18 New Zealand White female rabbits were inseminated
with semen from one of the two male rabbits of the same strain from
the same source. Day of insemination was designated Gestation Day
0. On Days 7-19 the females were given nominal doses of 0, 2, 5, or
12 mg flusilazole (94.8%)/kg bw/day by gavage in corn oil (actual
1.9, 4.8 and 10.1 mg/kg bw/day). There were two deaths during
treatment: one control female Day 19 and one high-dose female Day
16. Another control female died Day 23. Periodic anorexia was
observed in some rabbits in all groups and was not related to
treatment. No treatment-related effects were noted on clinical
observations, body weight, mean food consumption, abortions (2 in
controls, 3 in each of the treated groups), number of resorptions,
number of live or dead (none in any group) fetuses, fetal weights,
maternal liver weights or maternal pathology. The total number of
malformed fetuses was not increased by treatment. The incidence of
hydrocephalus was 1(1), 2(1), 4(2), and 4(3) fetuses (litters) in
the nominal 0, 2, 5, or 12 mg/kg bw/day level groups, respectively.
There was no increase in any other malformation nor of any
variation. In three subsequent studies hydrocephalus was observed
only in one fetus at 35 mg/kg bw/day and in none at any lower dose
or in controls. The incidence of hydrocephalus in this study,
therefore, was not confirmed and could not be regarded as related to
treatment. The NOAEL in this study was the nominal 12 mg/kg bw/day
level (actual 10.1 mg/kg bw/day) (Solomon et al. 1984).
A second study with 20 New Zealand White female rabbits/group
artificially inseminated with sperm from one of three male rabbits
tested dose levels of 0, 12, and 35 mg flusilazole (95%)/kg bw/day
administered Days 7-19 of gestation by gavage. In this study
pregnancy rates were low particularly in the two treated groups (not
dose-related). At 35 mg/kg bw/day, 2 of the does aborted and 10
totally resorbed their litters with only one doe with a live litter
at examination Day 29. There was an increased incidence of vaginal
discharge, discharge in cage and stained tail at this dose level as
well as an increased incidence of periodic anorexia. Since there
was only one litter, no teratologic effects could be determined.
One fetus did have hydrocephalus. Reproductive parameters at
12 mg/kg bw/day did not differ from controls and there was no
evidence of maternal toxicity at this level. The NOAEL in this
study was the nominal level of 12 mg/kg bw/day (actual 11 mg/kg
bw/day) (Zellers et al. 1985).
A pilot dietary study in New Zealand White rabbits included 7
females/group offered diets containing 0, 500, 1000, or 2000 ppm
flulilazole (94.8%) on Days 7-19 of gestation. Full results are not
given but pregnancy rate was low at 500 and 1000 ppm /3/7 at each
level and incidence of total resorption was high (4/7) at 2000 ppm.
Dietary levels of 0, 300, 600, and 1200 ppm flusilazole (94.8%)
were given to groups of 20 New Zealand White female rabbits days
7-19 of gestation following artificial insemination (day of
insemination designated Day 0 of gestation). Body weights, food
consumption and clinical signs of toxicity were monitored throughout
the study. The rabbits were sacrificed Day 29 of gestation and
examined for gross pathology, corpora lutea, numbers of live and
dead fetuses or resorption sites. The fetuses were examined for
external, visceral and skeletal abnormalities.
Body weights and food consumption were reduced during dosing.
There were no treatment-related clinical signs, pathological lesions
or changes in liver weights. Pregnancy rate was reduced in all
treated groups with the lowest rate in the high-dose group. The
number of dams with total resorptions was increased in the 600 and
1200 ppm groups. There were no treatment-related effects on mean
number of live fetuses, resorptions (in dams with fetuses) or fetal
weights. The number of litters available at 600 and 1200 ppm was
small (3 at each dose level), so the validity of the incidence of
fetal malformations and variations is uncertain at these levels.
There were no apparent effects at 300 ppm.
Since the pregnancy rate was reduced in all treated groups
another dietary study was performed with levels of 0, 30, 100, and
300 ppm flusilazole in diet offered Days 7-19 of gestation to groups
of 18 or 25 (300 ppm only) female New Zealand White rabbits
artificially inseminated with sperm from proven male rabbits. In
this study pregnancy rate was low in all groups but particularly in
the controls. In the 0 and 300 ppm groups 25% and 29%,
respectively, of the pregnant does totally resorbed their litters
but there were no total resorptions in the other two groups.
Because of the low number of dams with litters available in the
control group, the data in this study could not be adequately
assessed and a NOAEL cannot be determined (Alvarez et al. 1985b).
It has been suggested by the company that the reproductive
effects observed in these studies (reduced pregnancy and increased
total resorptions) were the result of inadequate technique during
artificial insemination. This might explain the effects observed in
the control group in the second dietary study.
Special studies on skin irritation and sensitization
Guinea Pig
In young adult Hartley guinea pigs, a range-finding study
indicated mild erythema, 24 hours after topical application to the
skin of the test material (90% pure flusilazole). The primary
irritation study conducted with 5 and 50% solutions in demethyl
phthalate did not demonstrate irritation in any of the 10 animals
exposed. Intradermal injections of 1% solutions of flusilazole (90%
pure) in dimethyl phthalate weekly for 4 weeks in the same animals
caused erythema and edema with necrotic centres at the injection
sites at 24 hours. Challenge with topical applications of 5 and 50%
solutions did not demonstrate sensitization (Wylie et al. 1984d).
In another study using 97.7% pure flusilazole, young adult
Duncan Hartley albino guinea pits (10/sex) were given 3 weekly
dermal applications of the test material (slightly moistened with
dimethyl phthalate) which was covered with plastic wrap for 6 hours.
No signs of irritation were observed in any of the animals after any
of these induction applications. Two weeks after the last induction
treatment, the guinea pigs were challenged with a dermal application
of test material on an untreated site which was then covered for 6
hours. No reaction was observed in any of the animals. A positive
control groups treated with 1-chloro,-2,4-dinitrobenzene (DNCB)
showed several erythema with necrosis after the second and third
induction applications and severe erythema after the challenge dose
two weeks later (Brock et al. 1988).
Special study of eye irritation
Rabbit
Two male New Zealand White rabbits were given doses of 0.01 ml
of undiluted flusilazole (90% pure) in the right conjunctival sac.
Both rabbits shoed mild conjunctival redness and chemosis at 1-4
hours. The washed eye also showed some discharge at one hour but no
other effects. The unwashed eye showed slight corneal opacity at
1-4 hours and slight cloudiness on biomicroscope examination at
24-48 hours. The eye was normal at 72 hours. The test material
produced only transient mild irritation (Hall et al. (1984).
COMMENTS
Flusilazole administered orally to rats was rapidly excreted in
urine and feces. The label (14C) from phenyl-labelled material was
excreted predominantly in feces, with a sex difference in that males
excreted higher amounts in feces than did females. However,
substantial variability existed. When triazole-labelled material
was given, the 14C was recovered predominantly in urine. Although
one study indicated a sex difference, with males excreting more in
urine than females, a sex difference was not observed in a second
study with more animals. Tissue levels were low. The highest
levels were found in the carcass (about 2% of the dose after 96-120
hours). Flusilazole was extensively metabolized in the rat.
Recovered parent compound accounted to 2-11% of the dose and was
recovered almost exclusively in feces.
Flusilazole has a low acute toxicity in the species examined.
In short-term (90-day) studies, urothelial hyperplasia in the
urinary bladder was observed in rats, mice and dogs at dietary
levels of 375, 225, and 125 ppm, respectively. Liver lesions,
including hepatocellular hypertrophy and hepatocellular vacuolar
cytoplasmic changes, were also observed in the 90-day studies in
rats and mice (at 750 and 225 ppm, respectively) and in a one-year
study in dogs at 20 ppm.
In a one-year feeding study in beagle dogs, centrilobular
hepatocyte enlargement was observed at 20 and 75 ppm. The NOAEL was
5 ppm, equal to 0.14 mg/kg bw/day.
In long-term feeding studies in rats and mice no bladder
lesions were observed. There was urothelial hyperplasia in the
renal pelvis in 1 of 10 male rats at 50 ppm and 1 of 10 male rats at
250 ppm for one year, but in none of the rats (7 of 10) given
250 ppm at one year (hepatocellular hypertrophy) and two years
(acidophilic foci of cellular alteration: 3/66, 6/63, 4/68, and
13/65 at 0, 10, 50, and 250 ppm, respectively). Male and female
mice given 200 ppm for 18 months developed hepatocellular fatty
changes (40/80 and 24/80, respectively, versus 4/80 and 2/70 in
respective controls). This effect was also reported in 8/80 male
mice at 25 ppm, which was not statistically significantly different
from controls. There was no evidence of oncogenicity in either
species. The NOAEL in the rat study was 10 ppm (equal to 0.45 mg/kg
bw/day). The NOAEL in the mouse study was 5 ppm (equal to
0.79 mg/kg bw/day).
In a 2-generation, 2 litters per generation reproduction study
in rats, flusilazole caused perinatal mortality in both F1 and F2
litters of dams given dietary levels of 250 ppm. There was some
indication of increased gestation length at this dose level, an
effect which was also observed in a post-natal toxicity study in
rats at doses of 10 mg/kg bw/day (gavage) and higher. Perinatal
mortality was observed at 100 mg/kg bw/day in this study. The
biological significance of prolonged gestation in this study is
uncertain. The NOAEL was 10 ppm (equal to 0.895 mg/kg bw/day) but
not at lower doses. The NOAEL in rats was 2 mg/kg bw/day. No
teratogenic effects were seen in rabbits. The NOAEL was 100 ppm
(equal to 2.8 mg/kg bw/day) based on maternal toxicity.
After reviewing all available in vitro and in vivo
short-term tests, the Meeting concluded that there was no evidence
of genotoxicity.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effect
Mouse: 25 ppm in the diet, equal to 3.4 mg/kg bw/day
Rat: 10 ppm in the diet, equal to 0.45 mg/kg bw/day
Dog: 5 ppm in the diet, equal to 0.14 mg/kg bw/day.
Estimate of acceptable daily intake for humans
0-0.001 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
1. Observations in humans.
2. Toxicity of the biotransformation products.
REFERENCES
Alvarez, L., Krauss, W.C. & Staples, R.E. (1984) Developmental
toxicity study in rats given INH-6573-66 in the diet on Days 7-16 of
gestation. Unpublished Report No. 432-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., & Kaplan, A.M. (1985a) INH-6573.
Prenatal and postnatal toxicity study in rats dosed by gavage on
Days 7-16 of gestation. Unpublished Report No. 654-85 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., Driscoll, C.D. & Kaplan, A.M. (1985b)
INH-6573. Developmental toxicity study in rabbits treated by diet
on Days 7-19 of gestation. Unpublished Report No. 337-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Anderson, J.J., Stadalius, M.A. & Schlueter, D.D. (1986) Metabolism
of 14C-DPX-H6573 in rats. Unpublished Report No. AMR-196-84 from
E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Arce, G.T., Matarese, C.C. & Sarrif, A.M. (1988) Mutagenicity
testing of INH-6573-21 in Salmonella typhimurium plate
incorporation assay. Unpublished Report No. 59-88 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Brock, W.J., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
Long-term feeding study in mice with INH-6573. Unpublished Report
No. 278-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Brock, W.J., Vick, D.A. & Chromey, N.C. (1988) Closed-patch
repeated insult dermal sensitization study (Buehler method) with
INH-6573-21 in guinea pigs. Unpublished Report No. 34-88 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Cheng, T. (1986) Rat metabolism study of [Triazole-3-14C]
DPX-H6573. Unpublished final report 6129-128 from Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Chromey, N.C., Horst, A.L., McCooey, K.T. & Sarrif, A.M. (1983)
Unscheduled DNA synthesis/rat hepatocytes in vitro. Unpublished
Report No. 209-83 from Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Donovan, S.M. & Irr, J.D. (1982) Mutagenicity evaluation in
Salmonella typhimurium. Unpublished Report No. 611-82 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Farrow, M.G., Cortina, T. & Padilla-Nash, H. (1983) In vivo bone
marrow chromosome study in rats - H # 14,728 - Final Report.
Unpublished Report No. 201-636 (Haskell No. 288-83) by Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Hall, J.A., Dashiell, O.L. & Kennedy, G.L. (1984) Eye irritation
test in rabbits. Unpublished Report No. 582-82 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984a) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 444-83 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984b) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 142-84 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
McCooey, K.T., Chromey, N.C., Sarrif, A.M. & Hemingway, R.E. (1983)
CHO/HGPRT assay for gene mutation. Unpublished Report No. 449-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
O'Neal, F.O., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
One-year feeding study in dogs with INH-6573. Unpublished Report
No. 461-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Krahn, D.F. & Gibson, J.R. (1983)
Ninety-day feeding and one-generation reproduction study in rats
with Silane [Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-
ylmethyl) (INH-6573). Unpublished Report No. 483-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Drahn, D.F. & Aftosmis, J.G. (1984)
Four-week range finding and ninety-day feeding study in mice with
Silane [Bis(4-fluorophenyl)] (methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Revised Unpublished Report No. 341-83 1-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Rickard, R.W., Sykes, G.P., Kaplan, A.M. & Gibson,
J.R. (1986) Long-term feeding (combined chronic
toxicity/oncogenicity study) and two-generation, four litter
reproduction study in rats with INH-6573. Unpublished Report
No. 32-86 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Poindexter, G.L., Henry, J.E., Kenney, L.A., Burgess, B.A. &
Kennedy, G.L. (1984) Inhalation median lethal concentration
(LC50) of INH-6573-41 by EPA Protocol. Unpublished Report
No. 553-83 by Haskell Laboratory, Newark, DE, USA. Submitted to WHO
by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Redgate, D., Sarver, J.W. & Chromey, N.C. (1985) Approximate lethal
dose (ALD) of INH-6573-66 in rabbits. Unpublished Report No. 54-85
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Richard, R.W., Wood, C.K., Krahn, D.F. & Aftosmis, J.G. (1983)
Three-month feeding study in dogs with Silane
[Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Unpublished Report No. 461-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Sarver, W., Vick, D.A., Valentine, R., Chromey, N.C. & Kaplan, A.M.
(1986) Twenty-one dose dermal toxicity study with INH-6573-82 in
rabbits. Unpublished Report No. 744-86 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Solomon, H.M., Alvarez, L., Staples, R.E. & Hamill, J.C. (1984)
Developmental toxicity study in rabbits given INH-6573 by gavage on
Days 7-19 of gestation. Unpublished Report No. 333-84 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Sorg, R.M., Naismith, R.W. & Mathews, R.J. (1984) Micronucleus test
(MNT) OECD H # 15,314. Unpublished Report No. PH 309A-DU-001-84
from Pharmakon Research International (Haskell Report HLO-437-84).
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Turner, R.J., Kinney, L.A. & Chromey, N.C. (1985) Inhalation median
lethal concentration (LC50) of INH-6573 by EPA Guidelines.
Unpublished Report No. 1-85 by Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Vlachos, D.A., Covell, D.L. & Sarrif, A.M. (1986) Evaluation of
INH-6573-82 in the in vitro assay for chromosome aberrations in
human lymphocytes. Unpublished Report No. 745-86 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Burgess, B.A. & Kennedy, G.L. (1983) Acute oral test
in rats. Unpublished Report No. 78-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984a) Median lethal dose (LD50) in rats - EPA proposed
guidelines, Newark, DE, USA. Unpublished Report No. 433-83 by
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984b) Median lethal dose (LD50) in rats - EPA proposed
guidelines. Unpublished Report No. 412-83 by Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Burgess, B.A. & Kennedy, G.L. (1984c)
Ten-dose oral subacute test in rats. Unpublished Report No. 78-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Dashiell, O.L. & Kennedy, G.L. (1984d)
Primary skin irritation and sensitization test on guinea pigs.
Unpublished Report No. 626-82 from Haskell Laboratory, Newark, DE,
USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Wylie, C.N., Redgate, D., Warheit, D.B. & Chromey, N.C. (1985)
Approximate lethal dose (ALD) of INH-6573-66 in male and female
mice. Unpublished Report No. 531-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Zellers, J.E., Staples, R.E., Alvarez, L. & Kaplan, A.M. (1985)
Developmental toxicity study (supplemental) in rabbits dosed by
gavage on Days 7-19 of gestation. Unpublished Report No. 699-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Toxicological studies
Acute toxicity
The acute toxicity of flusilazole to several animal species is
given in Table 1. Signs of toxicity following oral administration
included weight loss, weakness, lethargy and, at higher doses,
laboured breathing, convulsions and prostration. Inhalation effects
were chiefly laboured breathing and lung sounds. Dermal
administration resulted only in some local erythema.
TABLE 1. RESULTS OF ACUTE TOXICITY ASSAYS WITH FLUSILAZOLE
SPECIES SEX ROUTE LD50a/ ALDa/ REFERENCES
LC50b ALCb
Technical flusilazole
Rat M oral 1500 Wylie et al. 1983
Rabbit M&F oral 350 Redgate et al. 1985
Mouse M oral 680 Wylie et al. 1985
F oral 1000 Wylie et al. 1985
Rat M oral 1110 Wylie et al. 1984a
F oral 674 Wylie et al. 1984a
Rabbit M&F dermal >2000 Gargus et al. 1983
Rat M inhal. 2.7b,c Poindexter et al. 1984
F inhal. 3.7b,c Poindexter et al. 1984
Rat M&F inhal. >5.0b Turner et al. 1985
Formulation - 40% flusilazole
Rat M oral 1865 Wylie et al. 1984b
F oral 1272 Wylie et al. 1984b
a mg/kg bw
b mg/l
c test material was 99% pure.
Short-term studies
Oral studies
Rat
Groups of six male Crl:CD rats were given 0 or 300 mg
flusilazole (95.5%) in corn oil/kg bw/day by oral gavage for 2 weeks
(5 days per week). After the tenth dose 3 rats/group were killed and
examined for histopathology. The remaining rats were killed after a
14-day recovery period. One treated rat died after the fifth dose
(found Day 7). The treated rats died lost weight during the first
week of treatment and showed alopecia, diarrhea, stained face and
perineal area, salivation and hypersensitivity during the treatment
period only. Liver vacuolation was seen in all treated animals. In
urinary bladder transitional epithelium hyperplasia and vacuolation
were seen in all sacrificed treated animals with an increase in
mitotic figures seen in two of three males killed immediately
following the treatment period. Two treated males had atypical
giant cells in the lumen of the seminiferous tubules and necrosis
and cellular degeneration of the germinal epithelium in the testes.
Hyperplasia and vacuolation of the transitional epithelium in the
kidney pelvis was seen in two treated males. Lesions tended to be
more severe in animals killed immediately after the treatment period
than in those from the recovery phase (Wylie et al. 1984c).
A 90-day feeding study was carried out in Charles River (CD)
rats with dietary levels of flusilazole (96.7%) of 0, 25, 125 and
375 ppm fed to groups of 16 rats/sex and 750 ppm fed to 10 rats/sex.
Groups of 10/sex/dose comprised the standard 90-day study while the
extra 6/sex/dose continued on diet for a one generation one litter
reproduction study and then received control diet for a 4-month
recovery phase. Body weight gains were reduced only in the females
given 750 ppm. There were no effects on food consumption, clinical
signs of toxicity, hematological parameters or urinalyses. Serum
cholesterol levels were increased in both males and females given
750 ppm. Absolute and relative liver weights were increased in both
sexes at 750 ppm. Mild urothelial hyperplasia was observed in
urinary bladders of 5 males and 8 females at 750 ppm and 1 male and
4 females at 375 ppm. After the 4-month recovery phase following
dosing with 0-375 ppm flusilazole, no evidence of bladder
hyperplasia was observed. At 750 ppm 5 males had liver lesions,
chiefly hepatocyte hypertrophy in 5 males and fatty
change/hepatocytolysis in 4 males. The NOAEL was 125 ppm (equal to
10 mg/kg bw/day) (Pastoor et al. 1983).
Mouse
Groups of 20 male and 20 female Crl:CD-1 mice were given diets
containing 0, 25, 75, 225, 500 or 1000 ppm of flusilazole (96.7%
pure). After four weeks on these diets 10 mice/sex/group were
sacrificed. The remaining 10 mice/sex/group were sacrificed after
90 days of treatment. There were no effects noted on body weight,
food consumption or clinical signs of toxicity in animals sacrificed
at either interval. At 90 days high-dose males and females had
slightly reduced Hb, Hct and RBC counts which were statistically
significant for at least one of these parameters in each sex. No
other hematological changes were noted. At both 4 weeks and 90 days
absolute and relative liver weights were increased in male mice at
225 ppm and higher and in female mice at 75 ppm and higher. The
absolute and relative kidney weights in males at 1000 ppm were
decreased. At 90 days hepatocellular hypertrophy was observed in
4/10 females at 1000 ppm and 8-10/10 males at 225, 500, and
1000 ppm. Hepatocellular vacuolar cytoplasmic changes were observed
in both sexes (incidences 0, 0, 0, 2, 7, 10 in males and 0, 0, 1, 3,
9, and 8 in females, at 0, 25, 75, 225, 500, and 1000 ppm,
respectively). Urothelial cell hyperplasia in urinary bladder was
observed in 1/10 males and 7/10 males at 500 and 1000 ppm,
respectively, and 2/10, 3/10, and 6/10 females at 225, 500 and
1000 ppm, respectively. No treatment-related kidney lesions were
observed. Tissues from animals killed at 4 weeks were not examined.
The NOAEL in this study was 25 ppm (equal to 4.5 mg/kg bw/day)
(Pastoor et al. 1984).
Dog
Four beagle dogs/sex/group were given diet containing 0, 25,
125, or 750/500 ppm flusilazole (93% pure) for 3 months. At the
highest dose level the dogs were given diet containing 750 ppm
flusilazole for 3 weeks, control diet for one week and then a dose
level of 500 ppm for the remainder of the study. The dose level was
reduced because of body weight loss and reduced food intake at
750 ppm. At 500 ppm the males gained a mean of 0.2 kg while the
females lost a mean of 0.5 kg body weight. The other two groups
were comparable to controls (gains of 1.6-1.9 kg in both sexes).
Food consumption remained lower than controls at the 750/500 ppm
dose level. Clinical signs related to treatment were observed in
one male and two females at 500 ppm (weakness or tremors). One
female at 25 ppm was sacrificed in extremis week 5. No other
animal died. In males at 750/500 ppm WBC count and monocyte counts
were slightly but statistically significantly increased and
cholesterol levels, total protein and albumen levels were lower than
controls. In both sexes at 750/500 ppm and males at 125 ppm alanine
amino transerase levels were higher than controls. Absolute and
relative liver weights were increased in both sexes at 750/500 ppm
and were considered related to treatment. Other organ weight
differences were probably related to the decreased body weights at
750/500 ppm. Mild mucosal hyperplasia in the urinary bladder was
observed in all males and females at 500 ppm and in 2 males at
125 ppm. In the stomach, hyperlasia of lymphoid follicles in the
pyloric granular mucosa was observed in 0/4, 3/4, 4/3, and 4/4 males
and 0/4, 0/4, 3/4, and 4/4 females at 0, 25, 125, and 750/500 ppm,
respectively. On the basis of this effect in stomach a NOAEL was
not demonstrated in this study (Richard et al. 1983).
Groups of 5 beagle dogs/sex were given diets containing 0, 5,
20, or 75 ppm flusilazole (95.8% pure) for one year. Although all
treated groups of females had lower weight gains than controls, this
did not appear to be treatment-related. No body weight effects were
seen in males. No treatment-related effects were noted in food
consumption or clinical signs of toxicity. WBC counts were higher
in both sexes at 75 ppm than in the controls. Alkaline phosphates
levels were higher than controls and cholesterol levels lower in
both sexes at 75 ppm but the difference was statistically
significant only in males. The 75 ppm males also had lower total
protein and albumen levels in serum. Relative liver weights were
increased in both sexes at 75 ppm and relative kidney weight in
females at 75 ppm. There was centrilobular hepatocyte enlargement
at 20 and 75 ppm, in both sexes with distinct vacuolation in 3
high-dose males. There were also a higher degree of centrilobular
inflammation in all of the high-dose dogs both with regard to
relative numbers of inflammatory cells and to increased number of
hepatic veins involved. All dogs in all groups including controls
had lymphoid hyperplasia in the gastric mucosa but the degree
increased with dose level and was moderate only in high-dose males
and females. There were no treatment-related kidney or urinary
bladder effects. The NOAEL in this study was 5 ppm (equal to
0.14 mg/kg bw/day) (O'Neal et al. 1985).
Dermal studies
Rabbit
A study in New Zealand White rabbits in which groups of 5-10
rabbits/sex were treated dermally with 0, 25, 250 or 2000 mg
flusilazole/kg bw/day (95.8% pure material) was terminated after 6
days exposure because of serve erythema with necrosis in animals at
250 and 2000 mg/kg bw/day. The erythema was first observed Day 4 of
test.
A range-finding study with 175 or 250 mg flusilazole (94.9%) kg
bw/day produced only mild erythema at both levels.
A 21-day study was performed in groups of 5 New Zealand White
rabbits/sex/dose at dose levels of 0, 1, 5, 25, and 200 mg
flusilazole (94.9% pure)/kg bw/day. For application the test
material was moistened with distilled water. The exposure sites
were occluded for 6 hours and then washed free of remaining test
material. There were no apparent treatment-related effects on body
weights: hematology; clinical chemistry; or liver, kidney and spleen
weights. At 200 mg/kg bw/day, four males and 2 females had
persistent mild erythema beginning Days 6 to 12. At termination
slight to mild diffuse hyperplasia/thickening of epidermis was
observed in the skin of one male and two females at 25 mg/kg bw/day
and three males and two females at 200 mg/kg bw/day (Sarver et al.
1986).
Long-term/carcinogenicity studies
Rats
Groups of 70 male and 70 female Crl:CD(SD)BR rats were given
diets containing 0, 10, 50 or 250 ppm flusilazole (95.6% pure) for
two years. Additionally, groups of 10/sex were given the same dose
levels for 6 months and 1 year. After about 100 days on test, 20
rats/sex/group were mated for the reproduction phase of the study.
These rats were returned to the long-term study after weaning of the
second litter. Body weights, food consumption and clinical
condition were monitored throughout the study. Hematology and
clinical chemistry parameters were examined at 3, 6, 9, 12 and 24
months on fasted animals. The animals sacrificed at 6 months were
examined for bladder lesions only. About 40 tissues/organs were
examined for control and high-dose animals and liver, bladder,
kidney, lung and gross lesions for the low and intermediate dose
groups sacrificed after one year. For animals that died on test or
were sacrificed at two years all tissues from all animals in all
groups were examined.
Survival to two years was 39-57% in the various groups with no
treatment-relationship. Survival did not drop below 50% in any
group until Week 98. The high-dose males had slightly lower body
weights from 6 months to the end of the study but there was no
similar effect in females. There were no treatment-related effects
on food consumption, clinical signs of toxicity, hematology,
clinical chemistry or urinalysis. In females, relative liver
weights were increased at both one and two years at 250 ppm and at
one year only at 50 ppm. No effect was observed in males. No
treatment-related effects on the urinary bladder were observed at 6
months. After one year there was an increased incidence of
hepatocellular hypertrophy in females (0/10, 0/10, 1/10, and 7/10 at
0, 10, 50, and 250 ppm, respectively) and at 250 ppm polyploidy
(increased number of multinuclear hepatocytes) was observed in two
females with none in any other group. There were no liver changes in
males at one year. There was an increased incidence of
hydronephrosis in males (0/10, 0/10, 3/10, and 4/10 at 0, 10, 50 and
250 ppm). Urothelial hyperplasia in renal pelvis was observed in
one male at 50 pm and one at 250 ppm, but not in any other group.
Treatment-related lesions were not observed in urinary bladders of
males. Females did not show kidney or bladder lesions after one
year. Among rats dying on test or sacrificed after two years there
were increased incidences in livers of females of acidophilic foci
of cellular alterations (3, 3, 4, and 13 at 0, 10, 50, and 250 ppm,
respectively) and diffuse fatty change (9, 9, 12, and 23,
respectively, in 63-68 /group). There were no treatment-related
liver lesions in males. In females, there was an increased
incidence of pyelonephritis in kidney (3, 3, 8, and 10,
respectively), but no treatment-related lesions were observed in
males. The only tumours which were increased in incidence in treated
groups were squamous cell carcinomas of the oral and nasal cavities
in males (0/66, 1/63, 0/67, and 3/64, respectively). Historical
control data from six two-year feeding studies showed five studies
which no squamous cell carcinomas of oral or nasal origin while the
remaining study had an incidence of 2/60. These data suggest that
these tumours may occur randomly as clusters and that the incidence
in this study was not treatment-related. There was, therefore, no
evidence of oncogenicity. It is noted, however, that the effects
noted were relatively mild and higher doses might have been
tolerated. The NOAEL for non-neoplastic effects was 10 ppm (equal
to 0.45 mg/kg bw/day) (Pastoor et al. 1986).
Mouse
Groups of 80 Crl:CD-1(ICR)BR mice/sex were given a diet
containing 0, 5, 25, or 200 ppm flusilazole (93%-95.6% purity) for
18 months. Additional groups of 10/sex/dose were given the same
diets for 6 months. Survival to 18 months was 76-86% in males and
57-80% in females with the highest survival at the high-dose level.
There were no treatment-related effects on body weight, food
consumption, hematology or serum protein. There was a higher
incidence of skin sores in high-dose males and females but these
were mainly ear lesions which were noted at the beginning of the
study and appeared to be related to the animal identification
process. High-dose males also had an increased incidence of ruffled
fur. Absolute and relative liver weights were increased in males at
200 ppm at both 6 and 18 months and in 200 ppm females at 6 months.
Absolute kidney weight only was decreased in females at 200 ppm at
18 months but the biological significance of this observation was
not clear. There were no treatment-related lesions in the kidney.
In the liver there was an increased incidence of hepatocellular
fatty change in both males and females at 200 ppm at 18 months
(40/80 and 24/80, respectively, compared to 4/80 and 2/79 in male
and female controls. One 200 ppm male at 6 months also had this
lesion, although 8/80 males at the 25 ppm levels also had
hepatoceullar fatty changes. This incidence was not statistically
significantly different from controls. There was no treatment-
related increase in incidence of any tumour type in this study. The
NOAEL in this study was 25 ppm (equal to 3.4 mg/kg bw/day) (Brock
et al. 1985).
Reproduction study
Rat
Groups of 6 Crl:CD(SD)BR rats/sex/dose level from the 90 day
feeding study were mated within the same dose level for a period of
15 days. Dose levels were 0, 25, 125, and 375 ppm flusilazole
(96.7% pure). The females were examined daily for evidence of a
copulation plug. After the mating period, the females were housed
individually.
The number of pregnant females/group was 3-6. The total number
of pups born did not show a treatment-related effect but two of the
five litters at 375 ppm were all dead at birth. The remaining three
litters at this dose level were 93-100% viable similar to the
litters in the other groups including controls. Mean pup weights at
Day 4 were lower in the 375 ppm group than the other three groups.
At 375 ppm (equal to 29 mg/kg bw/day) pup weights remained slightly
lower than the other groups to weaning (Pastoor et al. 1983).
Because of the small group size, the usefulness of this study
is limited.
Groups of 20 Crl:CD(SD)BR rats/sex/dose level from the chronic
toxicity/oncogenicity study were mated within the same dose level
for a period of 15 days after a dosing period of about 100 days.
Dose levels were 0, 10, 50, and 150 ppm flusilazole (95.6% pure).
The females were examined daily throughout the mating period for
evidence of a copulatory plug. At the end of the mating period the
females were housed individually and allowed to give birth and raise
their litters to weaning. About one week after weaning of the F1a
litters the females were sacrificed without pathological
examination. At weaning of the F1b litters 20 weanlings/sex/group
were selected as parents for the F2 generation. These weanlings
were maintained on the same diets as their parents for a 90 day
period before mating. Mating procedures were the same for the F0 -
F1 litters. It is stated that sibling matings were avoided. For
each litter the number of pups born, the number of pups born alive,
the number alive at 24 hours, 4 days, 12 days and 21 days, the
litter weight at 24 hours, and 4 days and the weight of male and
female pups at 21 days were recorded. At Day 4 all litters with
more than 10 pups were culled to 10, with equal numbers of males and
females where possible. Pups found dead were discarded. At weaning
of the F2b pups 10/sex/group were randomly selected and necropsied.
Liver, kidney, testes and brain were weighed and about 26
tissues/organs were examined for histopathology.
Food consumption and body weights were recorded only for the
pre-mating periods for the F0 and F1b animals. There were no
consistent differences during this period in F0 animals but F1b
males given 250 ppm had lower body weights. No differences in food
consumption were noted. Pregnancy rates were variable in all mating
periods but with no dose-relationship. There was no evidence of a
treatment-related effect on male reproductive parameters. There was
some indication that gestation length might be longer in the
high-dose group but this could not be determined unequivocally
because of the number of animals for which gestation lengths could
not be determined. The number of pups dead at birth was increased
in the 250 ppm group in all litters and in the 50 ppm group in the
F2a litter. Survival to Day 4 was reduced at 250 ppm in the F1a,
F1b, and F2a litters. Loss of the entire litter perinatally was
observed in 2, 2, 2 and 1 dams at 250 ppm in the F1a, F1b, F2a and
F2b litters, respectively, and in 1 dam at 50 ppm in the F2a
litter. Survival after 4 days was similar in all groups. The sex
of pups is indicated only at weaning after culling of the litters so
the sex ratio cannot be determined. Pup weights were similar in all
groups except for reduced weights at weaning in male and female pups
in the F1b and F2a litters. In the F2b weanling pups the absolute
and relative liver weights in males in the 250 ppm group were
increased. No effect was observed in females. The only
pathological lesion of note was hydronephrosis in female weanlings.
The incidence of unilateral and bilateral lesions combined was 1/10,
4/10, 3/10, and 5/10 at 0, 10, 50, and 250 ppm, respectively. The
incidences in the 10 and 250 ppm groups are slightly higher than
those observed in 10 historical control groups over the period
1982-1986. However, neither the severity nor the incidence of the
lesions showed a dose-related pattern so it is unlikely that this is
treatment-related effect. The NOAEL in this study was 10 ppm (equal
to 0.85 mg/kg bw/day on the basis of pre-mating intake data)
(Pastoor et al. 1986).
Special studies on mutagenicity
Flusilazole was negative in seven mutagenicity studies (Table
2).
Special studies on teratology
Rat
In a pilot study in which groups of seven pregnant rats were
given doses of 0, 100 or 300 mg flusilazole (99% pure)/kg bw/day
Days 7-16 of gestation, 300 mg/kg bw was overtly toxic and 100 mg/kg
bw slightly toxic to the dams. Both levels were embryotoxic and
300 mg/kg bw resulted in cleft palates in about 51% of the fetuses
in each litter.
TABLE 2. RESULTS OF MUTAGENICITY ASSAYS ON FLUSILAZOLE
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Ames testa Salmonella typhimurium 1-250 ug/plate Negative Donovan & Irr, 1982
TA1535, TA1537, TA98, (90%)
TA100
Ames testa Salmonella typhimurium 5-250 ug/plate Negative Arce et al. 1988
TA1535, TA97, TA98, (97.7%)
TA100
Unscheduled DNA Rat hepatocytes Crl (SD) 1 x 10-5 - Negative Chromey et al. 1983
synthesis 1.1 x 102 mM
(95.5%)
CHO/HGPRT assay Chinese hamster ovary 0.04 - 0.15 mM Negative McCooey et al. 1983
for gene mutationa cells (CHO-K1/BH4 clone) 0.05 - 0.275 mM
with and without
activation,
respectively
(95.5%)
Micronucleus test Mouse (CD-1) 375 mg/kg bw in Negative Sorg et al. 1984
(bone marrow) corn oil
(91.5%)
Chromosome aberration Human lymphocytes 1.7 - 100 ug/ml Negative Vlachos et al. 1986
assaya (94.85%)
TABLE 2 (CONTD).
CONCENTRATION
TEST SYSTEM TEST ORGANISM OF FLUSILAZOLE RESULTS REFERENCE
(PURITY)
Chromosome assay Rat (Crl:CD (SD)) 50 - 500 mg/kg bw in Negative Farrow et al. 1983
(bone marrow) corn oil (not given)
a with and without metabolic activation.
Groups of 25 female Crl:CD(SD)BR rats mated to males of the
same strain were given doses of 0, 10, 50, or 250 mg flusilazole
(95.6%)/kg bw/day in corn oil by gavage Days 7-16 of gestation. The
day a copulation plug was observed was designated Day 1 of
gestation. Body weight, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation, the
rats were killed and examined for gross pathology, liver and uterine
weights, and numbers of corpora lutea, implantations, live and dead
fetuses. The uterus of any apparently non-pregnant females was
stained with ammonium sulfide to detect for early resorptions. The
fetuses were weighed and examined for external, visceral and
skeletal abnormalities.
Chromodacryorrhea, chromorhinorrhea, wet underbodies, or wet or
yellow or brown stained perineal areas were observed at the
250 mg/kg bw dose level with all but two females showing at least
one of these signs. The severity increased with continued dosing
and decreased following cessation of dosing. Some females at this
dose level had red vaginal discharges or stains and there was a
slightly increased incidence of focal alopecia. Two of the dams
died (Days 11 and 18). At 50 mg/kg bw one dam had red vaginal
discharge and one ruffled abdominal fur. Food consumption was
reduced during dosing at 50 and 250 mg/kg bw. At sacrifice relative
liver weights were increased in the dams at 50 and 250 mg/kg bw. At
250 mg/kg bw mean fetal body weight was reduced, incidence of
resorptions was increased and the number of live fetuses per litter
was reduced. Stunted fetuses were observed only in treated groups
at incidences of 1, 4, and 3 fetuses at 10, 50, and 250 mg/kg bw.
Cleft palate and absence of renal papilla were observed in fetuses
(12% and 9%, respectively) from dams given 250 mg/kg bw but not at
lower doses. At 50 mg/kg bw/day two fetuses (from 2 litters) had no
innominate artery and one (from the same litter as one of the
fetuses with no innominate artery) had a great vessel malformation
in the heart. These lesions were not seen in any other group so
were probably incidental. In all groups including controls there
was a high incidence of hydrocephalus and/or dilated lateral
ventricles of the brain. The incidence was not dose-related and no
hydrocephalus was observed in subsequent studies so this finding was
not considered to be treatment-related. The incidence of misaligned
sternebra, extra ossification centres in ribs and ossification delay
in sternebra were increased at all dose levels compared to controls.
There were increased incidences of rudimentary ribs at 50 and
250 mg/kg bw and extra ribs at 250 mg/kg bw. A NOEL was not
demonstrated in this study since skeletal variations were increased
at 10 mg/kg bw, the lowest dose tested. Maternal toxicity was seen
at 50 mg/kg bw. Teratogenic effects were observed at 250 mg/kg bw
(Lamontia et al. 1984a).
A second study included groups of 24 mated female Crl:CD(SD)BR
rats given doses of 0, 0.4, 2.0, 10.0, 50.0 or 250.0 (10 females) mg
flusilazole (95.6%)/kg bw/day in corn oil by gavage Days 7-16 of
gestation. The day a copulation plug was observed was designated
Day 1 of gestation. Body weight, food consumption and clinical
condition were monitored throughout the study. On Day 21 of
gestation the rats were killed and examined as in the study above.
At 50 and 250 mg/kg bw/day the dams had reduced body weight
gains during the early part of the dosing period but the difference
was statistically significant only at 250 mg/kg bw. Both groups had
reduced food intakes during dosing. Clinical signs of toxicity were
observed at 250 mg/kg bw: alopoecia, brown stains of face and limbs
and yellow staining of perineum. Relative liver weights were
increased in the 50 and 250 mg/kg bw groups. The incidence of early
resorptions was similar in all groups; however, the number of dams
with median or late resorptions was higher in groups given 10 mg/kg
bw or more. The mean number of live fetuses/litter was similar in
all groups. The incidence of stunted fetuses was increased at dose
levels of 10 mg/kg bw and higher. Cleft palate was observed in
fetuses from dams given 250 mg/kg bw but not at lower doses.
Absence of renal papilla was observed only in treated groups but
there was no dose-relationship and the observation was not
considered to be treatment-related. Hydrocephalus was not observed
in any group. Visceral (large renal pelvis and small renal papilla)
and skeletal (ribs anomalies) variations were observed at 10 mg/kg
bw and higher. Absence of the innominate artery was observed in
4(3) fetuses (litters) at 50 mg/kg bw and 1 fetus at 250 mg/kg bw.
Retarded development (delayed ossification of sternebra and
vertebral arch) was also evident at dose levels of 10 mg/kg bw and
higher. The incidence of red foci in bladder of fetuses was
increased at dose levels of 10 mg/kg bw and higher; however, these
lesions were non-specific and were considered to be of no biological
significance. The NOAEL in this study was 2 mg/kg bw/day (Lamontia
et al. 1984b).
In a dietary study, groups of 24 mated Crl:CD(SD)BR female rats
were given diets containing 0, 50, 100, and 300 or 900 ppm
flusilazole (94.8%) Days 7-16 of gestation. The day a copulation
plug was observed was designated Day 1 of gestation. Body weights,
food consumption and clinical condition were monitored throughout
the study. On Gestation Day 21 the rats were sacrificed and
examined for gross pathology, liver and uterine weights and uterine
contents. Fetuses were weighed, sexed and examined for external
visceral and skeletal abnormalities.
One female at 300 ppm died on Day 19 of gestation. The death
was not considered to be related to treatment. During Days 7-11 of
gestation at the beginning of dosing weight gains in all treated
groups were lower than in controls in a dose-related manner.
Significant reductions were noted Days 7-9 at 300 and 900 ppm. Food
consumption during dosing was lower than controls in all treated
groups but reached significant reductions only at 300 and 900 ppm.
There were no treatment-related clinical signs of toxicity.
Relative liver weights did not indicate a significant treatment-
related effect. Pregnancy rate was high in all groups. One dam at
900 ppm totally resorbed its litter. The incidence of early
resorptions was similar in all groups. However, there was an
increased incidence (dose-related) of median and late resorptions at
100 ppm and higher. Litter size was inversely dose-related and
significantly smaller than in controls in the 900 ppm group. Small
litters (less than 10 fetuses) were only seen at dose levels of
100 ppm and above. The 50 ppm group was similar to controls. The
incidence of stunted fetuses was increased at 300 and 900 ppm. Mean
fetal weights were not affected. No treatment-related malformations
were observed. No fetuses showed cleft palate and incidence of
absence of renal papilla was not dose or treatment-related. Two
fetuses from different litters in the 900 ppm group had slightly
domed heads but hydrocephalus was not confirmed on visceral
examination. There was a significant dose-related increase in
skeletal variations at dose levels of 100 ppm and higher with extra
ossification of the sternebra at all 3 dose levels, and rudimentary
ribs, extra ossification in cervical ribs and delayed ossification
in the vertebral arches (cervical) at 300 and 900 ppm. Red foci
were observed in the bladder of a few fetuses at all dose levels
with a slightly higher but not dose-related incidence at 100 ppm and
above. As in the previous study, these were not considered to be
biologically significant. The NOAEL in this study was 50 ppm (equal
to 4.6 mg/kg bw) (Alvarez et al. 1984).
A study of prenatal and postnatal toxicity in rats was
conducted in two phases. For the prenatal study (phase 1), groups
of 24 female Crl:CD(SD)BR rats were mated on a 1:1 basis with males
of the same strain. The day a copulation plug was observed was
designated Day 1 of gestation. On Days 7 through 16 the females
were given a dose of 0, 0.2, 0.4, 2, 10, or 100 mg flusilazole
(94.8% pure in 0.5% aqueous methyl cellulose)/kg bw by oral
intubation. Body weights, food consumption and clinical condition
were monitored throughout the study. On Day 21 of gestation the
females of one group at each dose level were killed for examination
of the uterine contents. The extra control and 100 mg/kg bw groups
were killed Day 22 of gestation in order to determine whether
absence of renal papillae was a compound-related effect or an
anomaly.
Analyses of the dosing solutions indicates that the solution
prepared for the first day of dosing contained only 6-19% of the
nominal concentrations. Since solutions were prepared daily this
may have been given to only the one female/group that had reached
Gestation Day 7 on that day and only two of these (one at 2 and one
at 100 mg/kg) were pregnant. However, the next analyses given were
done on solutions prepared six days later and it is unknown whether
the technical error causing the low concentrations was corrected
before this date. Subsequent analyses indicated concentrations
75-110% of nominal. Assuming the worst case, 13-16 pregnant
females/group were given 1-6 doses which were about 10% of the
intended dose level (mean duration of low doses was 3.4-3.7 days).
In each group there were 7-8 pregnant females which did not receive
the low doses.
Body weight gains and food consumption were lower than controls
during dosing in the females given 100 mg/kg bw. Stained chin was
observed in many of the 100 mg/kg bw females and some showed
perinasal staining and/or wet perineum. Absolute and relative liver
weights were increased in dams at 100 mg/kg bw. Pregnancy rate was
83-96% in treated animals and 96-100% in controls; there was no dose
relationship. The number of early resorptions and dams with early
resorptions did not differ among the groups. However, there was an
increased incidence of median and late resorptions at 100 mg/kg bw.
The numbers of stunted fetuses and dams with stunted fetuses were
increased at 10 and 100 mg/kg bw (statistically significant only at
100 mg/kg bw). One fetus in each of the 100 mg/kg bw groups had no
renal papilla unilaterally. One other high-dose fetus had
malformations: no tail and imperforate anus (group killed Gestation
Day 22). There was an increased incidence of bladder foci in the
two 100 mg/kg bw groups compared to their respective controls.
However, the Gestation Day 22 control incidence was higher than the
100 mg/kg bw group killed on Gestation Day 21. Groups given 0.4,
2.0 and 10.0 mg/kg bw had slightly higher incidences of bladder
foci. Incidence of small papilla in the kidney was increased at 10
and 100 mg/kg bw but in the groups killed Gestation Day 21 the
increase was not dose-related. Incidence of distended ureter was
increased at 10 and 100 mg/kg bw when expressed as % affected per
litter. The number of litters affected was slightly higher than
controls in the Day 21 group at 100 mg/kg bw but not in the Day 22
group.
In the postnatal study (phase II), groups of 22 females
Crl:CD(SD)BR rats were mated and dosed as in phase I except that
only one control and one 100 mg/kg bw group were included. The
females were permitted to have their litters and raise them to
weaning. Body weights, food consumption and clinical signs were
monitored throughout the gestation and lactation periods. Offspring
were counted and weighted by sex Days 0, 4 (before and after
culling), 7 and 14 post partum. Pups were weighed individually Day
21. At weaning the pups were sacrificed and subjected to gross
necropsy.
The dosing solutions were adequately prepared in this phase of
the study. At 100 mg/kg bw five dams died; four on Gestation Day 24
or 25 (with apparently normal litters) and one four days post partum
after delivering a litter (all dead) Day 25. At 2.0 mg/kg bw one
dam died Day 24 during delivery. At 100 mg/kg body weight gain and
food consumption were reduced during the first few days of dosing
but were similar to controls for the remainder of the study. A few
dams at 100 mg/kg bw are stated to have shown pallor (4), hunching
(2), weakness (2) and dystocia (1) during the puturition and
lactation periods. There was an increase in mean gestational length
at 10 and 100 mg/kg bw. In controls, gestation did not exceed 22
days. In treated groups a gestation period of 23 days was seen in
2, 1, 4, 12, and 4 dams at 0.2, 0.4, 2.0, 10.0, and 100 mg/kg bw,
respectively, and at 100 mg/kg bw 11 dams did not deliver until 24
days gestation. At 100 mg/kg the mean gestation length was 23.7
days compared to 21.8 days in controls. Among the 11 high-dose dams
with parturition at Gestation Day 24, six had no live fetuses and
the other five had 1-5 live pups (mean 3) and 1-7 dead pups (mean
4.8). At 10 mg/kg bw the longest gestation period was 23 days and
there was no apparent effect on pup survival at birth. At this dose
four dams had one dead pup (of 6-15 pups in litter) at birth; two
delivered Day 22 and two delivered Day 23. This latter incidence
of dead pups at birth was similar to that in controls. Survival to
Day 4 was decreased at 100 mg/kg bw but was similar to controls at
the other dose levels. At 100 mg/kg bw 18% of the pups alive Day 0
of lactation had died by Day 4 compared to 0.4-1.7% of the pups in
the control and lower dose groups. There was no effect on survival
for the rest of the gestation period. Litter size was reduced at
100 mg/kg bw even when both live and dead pups in the litters were
considered. Mean litter size was slightly lower than controls in
the 10 mg/kg bw group reaching statistical significance after
culling. This was the result of an increased number of litters with
less than 10 pups (4 cf 0 in controls and 1-2 in the other treated
groups). Pup weights at birth were somewhat higher in the three
highest dose groups than in controls. At 2 and 10 mg/kg bw a few
litters with higher weights were among those with a 23 day gestation
length. At 100 mg/kg bw mean, live litter weights were not as high
as at 10 mg/kg bw mean, live litter weights were not as high as at
10 mg/kg bw but other effects confound any comparisons. No
treatment-related effects on sex were apparent. The only
alterations noted in the weanlings were dilatation of the renal
pelvis and distended ureter. The incidence was not unequivocally
dose-related although somewhat higher at 10 mg/kg bw/day. It is
stated that among the dead pups, absence of renal papilla was
observed in only 2/42 pups and small papilla (size 1 or 2) was seen
in only 4 pups (2 of each size). Most of the dead pups were in the
high-dose group so it is presumed that these observations were from
that group. In phase II the NOAEL was 2 mg/kg bw with the only
effects being a slightly longer gestation period with a higher mean
weight in a few animals (Alvarez et al. 1985a).
Rabbit
Groups of 18 New Zealand White female rabbits were inseminated
with semen from one of the two male rabbits of the same strain from
the same source. Day of insemination was designated Gestation Day
0. On Days 7-19 the females were given nominal doses of 0, 2, 5, or
12 mg flusilazole (94.8%)/kg bw/day by gavage in corn oil (actual
1.9, 4.8 and 10.1 mg/kg bw/day). There were two deaths during
treatment: one control female Day 19 and one high-dose female Day
16. Another control female died Day 23. Periodic anorexia was
observed in some rabbits in all groups and was not related to
treatment. No treatment-related effects were noted on clinical
observations, body weight, mean food consumption, abortions (2 in
controls, 3 in each of the treated groups), number of resorptions,
number of live or dead (none in any group) fetuses, fetal weights,
maternal liver weights or maternal pathology. The total number of
malformed fetuses was not increased by treatment. The incidence of
hydrocephalus was 1(1), 2(1), 4(2), and 4(3) fetuses (litters) in
the nominal 0, 2, 5, or 12 mg/kg bw/day level groups, respectively.
There was no increase in any other malformation nor of any
variation. In three subsequent studies hydrocephalus was observed
only in one fetus at 35 mg/kg bw/day and in none at any lower dose
or in controls. The incidence of hydrocephalus in this study,
therefore, was not confirmed and could not be regarded as related to
treatment. The NOAEL in this study was the nominal 12 mg/kg bw/day
level (actual 10.1 mg/kg bw/day) (Solomon et al. 1984).
A second study with 20 New Zealand White female rabbits/group
artificially inseminated with sperm from one of three male rabbits
tested dose levels of 0, 12, and 35 mg flusilazole (95%)/kg bw/day
administered Days 7-19 of gestation by gavage. In this study
pregnancy rates were low particularly in the two treated groups (not
dose-related). At 35 mg/kg bw/day, 2 of the does aborted and 10
totally resorbed their litters with only one doe with a live litter
at examination Day 29. There was an increased incidence of vaginal
discharge, discharge in cage and stained tail at this dose level as
well as an increased incidence of periodic anorexia. Since there
was only one litter, no teratologic effects could be determined.
One fetus did have hydrocephalus. Reproductive parameters at
12 mg/kg bw/day did not differ from controls and there was no
evidence of maternal toxicity at this level. The NOAEL in this
study was the nominal level of 12 mg/kg bw/day (actual 11 mg/kg
bw/day) (Zellers et al. 1985).
A pilot dietary study in New Zealand White rabbits included 7
females/group offered diets containing 0, 500, 1000, or 2000 ppm
flulilazole (94.8%) on Days 7-19 of gestation. Full results are not
given but pregnancy rate was low at 500 and 1000 ppm /3/7 at each
level and incidence of total resorption was high (4/7) at 2000 ppm.
Dietary levels of 0, 300, 600, and 1200 ppm flusilazole (94.8%)
were given to groups of 20 New Zealand White female rabbits days
7-19 of gestation following artificial insemination (day of
insemination designated Day 0 of gestation). Body weights, food
consumption and clinical signs of toxicity were monitored throughout
the study. The rabbits were sacrificed Day 29 of gestation and
examined for gross pathology, corpora lutea, numbers of live and
dead fetuses or resorption sites. The fetuses were examined for
external, visceral and skeletal abnormalities.
Body weights and food consumption were reduced during dosing.
There were no treatment-related clinical signs, pathological lesions
or changes in liver weights. Pregnancy rate was reduced in all
treated groups with the lowest rate in the high-dose group. The
number of dams with total resorptions was increased in the 600 and
1200 ppm groups. There were no treatment-related effects on mean
number of live fetuses, resorptions (in dams with fetuses) or fetal
weights. The number of litters available at 600 and 1200 ppm was
small (3 at each dose level), so the validity of the incidence of
fetal malformations and variations is uncertain at these levels.
There were no apparent effects at 300 ppm.
Since the pregnancy rate was reduced in all treated groups
another dietary study was performed with levels of 0, 30, 100, and
300 ppm flusilazole in diet offered Days 7-19 of gestation to groups
of 18 or 25 (300 ppm only) female New Zealand White rabbits
artificially inseminated with sperm from proven male rabbits. In
this study pregnancy rate was low in all groups but particularly in
the controls. In the 0 and 300 ppm groups 25% and 29%,
respectively, of the pregnant does totally resorbed their litters
but there were no total resorptions in the other two groups.
Because of the low number of dams with litters available in the
control group, the data in this study could not be adequately
assessed and a NOAEL cannot be determined (Alvarez et al. 1985b).
It has been suggested by the company that the reproductive
effects observed in these studies (reduced pregnancy and increased
total resorptions) were the result of inadequate technique during
artificial insemination. This might explain the effects observed in
the control group in the second dietary study.
Special studies on skin irritation and sensitization
Guinea Pig
In young adult Hartley guinea pigs, a range-finding study
indicated mild erythema, 24 hours after topical application to the
skin of the test material (90% pure flusilazole). The primary
irritation study conducted with 5 and 50% solutions in demethyl
phthalate did not demonstrate irritation in any of the 10 animals
exposed. Intradermal injections of 1% solutions of flusilazole (90%
pure) in dimethyl phthalate weekly for 4 weeks in the same animals
caused erythema and edema with necrotic centres at the injection
sites at 24 hours. Challenge with topical applications of 5 and 50%
solutions did not demonstrate sensitization (Wylie et al. 1984d).
In another study using 97.7% pure flusilazole, young adult
Duncan Hartley albino guinea pits (10/sex) were given 3 weekly
dermal applications of the test material (slightly moistened with
dimethyl phthalate) which was covered with plastic wrap for 6 hours.
No signs of irritation were observed in any of the animals after any
of these induction applications. Two weeks after the last induction
treatment, the guinea pigs were challenged with a dermal application
of test material on an untreated site which was then covered for 6
hours. No reaction was observed in any of the animals. A positive
control groups treated with 1-chloro,-2,4-dinitrobenzene (DNCB)
showed several erythema with necrosis after the second and third
induction applications and severe erythema after the challenge dose
two weeks later (Brock et al. 1988).
Special study of eye irritation
Rabbit
Two male New Zealand White rabbits were given doses of 0.01 ml
of undiluted flusilazole (90% pure) in the right conjunctival sac.
Both rabbits shoed mild conjunctival redness and chemosis at 1-4
hours. The washed eye also showed some discharge at one hour but no
other effects. The unwashed eye showed slight corneal opacity at
1-4 hours and slight cloudiness on biomicroscope examination at
24-48 hours. The eye was normal at 72 hours. The test material
produced only transient mild irritation (Hall et al. (1984).
COMMENTS
Flusilazole administered orally to rats was rapidly excreted in
urine and feces. The label (14C) from phenyl-labelled material was
excreted predominantly in feces, with a sex difference in that males
excreted higher amounts in feces than did females. However,
substantial variability existed. When triazole-labelled material
was given, the 14C was recovered predominantly in urine. Although
one study indicated a sex difference, with males excreting more in
urine than females, a sex difference was not observed in a second
study with more animals. Tissue levels were low. The highest
levels were found in the carcass (about 2% of the dose after 96-120
hours). Flusilazole was extensively metabolized in the rat.
Recovered parent compound accounted to 2-11% of the dose and was
recovered almost exclusively in feces.
Flusilazole has a low acute toxicity in the species examined.
In short-term (90-day) studies, urothelial hyperplasia in the
urinary bladder was observed in rats, mice and dogs at dietary
levels of 375, 225, and 125 ppm, respectively. Liver lesions,
including hepatocellular hypertrophy and hepatocellular vacuolar
cytoplasmic changes, were also observed in the 90-day studies in
rats and mice (at 750 and 225 ppm, respectively) and in a one-year
study in dogs at 20 ppm.
In a one-year feeding study in beagle dogs, centrilobular
hepatocyte enlargement was observed at 20 and 75 ppm. The NOAEL was
5 ppm, equal to 0.14 mg/kg bw/day.
In long-term feeding studies in rats and mice no bladder
lesions were observed. There was urothelial hyperplasia in the
renal pelvis in 1 of 10 male rats at 50 ppm and 1 of 10 male rats at
250 ppm for one year, but in none of the rats (7 of 10) given
250 ppm at one year (hepatocellular hypertrophy) and two years
(acidophilic foci of cellular alteration: 3/66, 6/63, 4/68, and
13/65 at 0, 10, 50, and 250 ppm, respectively). Male and female
mice given 200 ppm for 18 months developed hepatocellular fatty
changes (40/80 and 24/80, respectively, versus 4/80 and 2/70 in
respective controls). This effect was also reported in 8/80 male
mice at 25 ppm, which was not statistically significantly different
from controls. There was no evidence of oncogenicity in either
species. The NOAEL in the rat study was 10 ppm (equal to 0.45 mg/kg
bw/day). The NOAEL in the mouse study was 5 ppm (equal to
0.79 mg/kg bw/day).
In a 2-generation, 2 litters per generation reproduction study
in rats, flusilazole caused perinatal mortality in both F1 and F2
litters of dams given dietary levels of 250 ppm. There was some
indication of increased gestation length at this dose level, an
effect which was also observed in a post-natal toxicity study in
rats at doses of 10 mg/kg bw/day (gavage) and higher. Perinatal
mortality was observed at 100 mg/kg bw/day in this study. The
biological significance of prolonged gestation in this study is
uncertain. The NOAEL was 10 ppm (equal to 0.895 mg/kg bw/day) but
not at lower doses. The NOAEL in rats was 2 mg/kg bw/day. No
teratogenic effects were seen in rabbits. The NOAEL was 100 ppm
(equal to 2.8 mg/kg bw/day) based on maternal toxicity.
After reviewing all available in vitro and in vivo
short-term tests, the Meeting concluded that there was no evidence
of genotoxicity.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effect
Mouse: 25 ppm in the diet, equal to 3.4 mg/kg bw/day
Rat: 10 ppm in the diet, equal to 0.45 mg/kg bw/day
Dog: 5 ppm in the diet, equal to 0.14 mg/kg bw/day.
Estimate of acceptable daily intake for humans
0-0.001 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
1. Observations in humans.
2. Toxicity of the biotransformation products.
REFERENCES
Alvarez, L., Krauss, W.C. & Staples, R.E. (1984) Developmental
toxicity study in rats given INH-6573-66 in the diet on Days 7-16 of
gestation. Unpublished Report No. 432-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., & Kaplan, A.M. (1985a) INH-6573.
Prenatal and postnatal toxicity study in rats dosed by gavage on
Days 7-16 of gestation. Unpublished Report No. 654-85 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Alvarez, L., Staples, R.E., Driscoll, C.D. & Kaplan, A.M. (1985b)
INH-6573. Developmental toxicity study in rabbits treated by diet
on Days 7-19 of gestation. Unpublished Report No. 337-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Anderson, J.J., Stadalius, M.A. & Schlueter, D.D. (1986) Metabolism
of 14C-DPX-H6573 in rats. Unpublished Report No. AMR-196-84 from
E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Arce, G.T., Matarese, C.C. & Sarrif, A.M. (1988) Mutagenicity
testing of INH-6573-21 in Salmonella typhimurium plate
incorporation assay. Unpublished Report No. 59-88 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Brock, W.J., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
Long-term feeding study in mice with INH-6573. Unpublished Report
No. 278-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Brock, W.J., Vick, D.A. & Chromey, N.C. (1988) Closed-patch
repeated insult dermal sensitization study (Buehler method) with
INH-6573-21 in guinea pigs. Unpublished Report No. 34-88 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Cheng, T. (1986) Rat metabolism study of [Triazole-3-14C]
DPX-H6573. Unpublished final report 6129-128 from Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Chromey, N.C., Horst, A.L., McCooey, K.T. & Sarrif, A.M. (1983)
Unscheduled DNA synthesis/rat hepatocytes in vitro. Unpublished
Report No. 209-83 from Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Donovan, S.M. & Irr, J.D. (1982) Mutagenicity evaluation in
Salmonella typhimurium. Unpublished Report No. 611-82 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Farrow, M.G., Cortina, T. & Padilla-Nash, H. (1983) In vivo bone
marrow chromosome study in rats - H # 14,728 - Final Report.
Unpublished Report No. 201-636 (Haskell No. 288-83) by Hazleton
Laboratories America, Inc. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Hall, J.A., Dashiell, O.L. & Kennedy, G.L. (1984) Eye irritation
test in rabbits. Unpublished Report No. 582-82 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984a) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 444-83 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Lamontia, C.L., Staples, R.E. & Alvarez, L. (1984b) Embryo-fetal
toxicity and teratogenicity study of INH-6573-39 by gavage in the
rat. Unpublished Report No. 142-84 from Haskell Laboratory, Newark,
DE, USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
McCooey, K.T., Chromey, N.C., Sarrif, A.M. & Hemingway, R.E. (1983)
CHO/HGPRT assay for gene mutation. Unpublished Report No. 449-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
O'Neal, F.O., Rickard, R.W., Kaplan, A.M. & Gibson, J.R. (1985)
One-year feeding study in dogs with INH-6573. Unpublished Report
No. 461-85 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Krahn, D.F. & Gibson, J.R. (1983)
Ninety-day feeding and one-generation reproduction study in rats
with Silane [Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-
ylmethyl) (INH-6573). Unpublished Report No. 483-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Wood, C.K., Drahn, D.F. & Aftosmis, J.G. (1984)
Four-week range finding and ninety-day feeding study in mice with
Silane [Bis(4-fluorophenyl)] (methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Revised Unpublished Report No. 341-83 1-83 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Pastoor, T.P., Rickard, R.W., Sykes, G.P., Kaplan, A.M. & Gibson,
J.R. (1986) Long-term feeding (combined chronic
toxicity/oncogenicity study) and two-generation, four litter
reproduction study in rats with INH-6573. Unpublished Report
No. 32-86 from Haskell Laboratory, Newark, DE, USA. Submitted to
WHO by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Poindexter, G.L., Henry, J.E., Kenney, L.A., Burgess, B.A. &
Kennedy, G.L. (1984) Inhalation median lethal concentration
(LC50) of INH-6573-41 by EPA Protocol. Unpublished Report
No. 553-83 by Haskell Laboratory, Newark, DE, USA. Submitted to WHO
by E.I. du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Redgate, D., Sarver, J.W. & Chromey, N.C. (1985) Approximate lethal
dose (ALD) of INH-6573-66 in rabbits. Unpublished Report No. 54-85
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., DE, Wilmington, USA.
Richard, R.W., Wood, C.K., Krahn, D.F. & Aftosmis, J.G. (1983)
Three-month feeding study in dogs with Silane
[Bis(4-fluorophenyl)](methyl) (1H-1,2,4-triazol-1-ylmethyl)
(INH-6573). Unpublished Report No. 461-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Sarver, W., Vick, D.A., Valentine, R., Chromey, N.C. & Kaplan, A.M.
(1986) Twenty-one dose dermal toxicity study with INH-6573-82 in
rabbits. Unpublished Report No. 744-86 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Solomon, H.M., Alvarez, L., Staples, R.E. & Hamill, J.C. (1984)
Developmental toxicity study in rabbits given INH-6573 by gavage on
Days 7-19 of gestation. Unpublished Report No. 333-84 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Sorg, R.M., Naismith, R.W. & Mathews, R.J. (1984) Micronucleus test
(MNT) OECD H # 15,314. Unpublished Report No. PH 309A-DU-001-84
from Pharmakon Research International (Haskell Report HLO-437-84).
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Turner, R.J., Kinney, L.A. & Chromey, N.C. (1985) Inhalation median
lethal concentration (LC50) of INH-6573 by EPA Guidelines.
Unpublished Report No. 1-85 by Haskell Laboratory, Newark, DE, USA.
Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Vlachos, D.A., Covell, D.L. & Sarrif, A.M. (1986) Evaluation of
INH-6573-82 in the in vitro assay for chromosome aberrations in
human lymphocytes. Unpublished Report No. 745-86 from Haskell
Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du Pont de
Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Burgess, B.A. & Kennedy, G.L. (1983) Acute oral test
in rats. Unpublished Report No. 78-83 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984a) Median lethal dose (LD50) in rats - EPA proposed
guidelines, Newark, DE, USA. Unpublished Report No. 433-83 by
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Ferenz, R.L., Burgess, B.A. & Kennedy,
G.L. (1984b) Median lethal dose (LD50) in rats - EPA proposed
guidelines. Unpublished Report No. 412-83 by Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Burgess, B.A. & Kennedy, G.L. (1984c)
Ten-dose oral subacute test in rats. Unpublished Report No. 78-83
from Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I.
du Pont de Nemours and Co., Inc., Wilmington, DE, USA.
Wylie, C.N., Henry, J.E., Dashiell, O.L. & Kennedy, G.L. (1984d)
Primary skin irritation and sensitization test on guinea pigs.
Unpublished Report No. 626-82 from Haskell Laboratory, Newark, DE,
USA. Submitted to WHO by E.I. du Pont de Nemours and Co., Inc.,
Wilmington, DE, USA.
Wylie, C.N., Redgate, D., Warheit, D.B. & Chromey, N.C. (1985)
Approximate lethal dose (ALD) of INH-6573-66 in male and female
mice. Unpublished Report No. 531-84 from Haskell Laboratory,
Newark, DE, USA. Submitted to WHO by E.I. du Pont de Nemours and
Co., Inc., Wilmington, DE, USA.
Zellers, J.E., Staples, R.E., Alvarez, L. & Kaplan, A.M. (1985)
Developmental toxicity study (supplemental) in rabbits dosed by
gavage on Days 7-19 of gestation. Unpublished Report No. 699-85 from
Haskell Laboratory, Newark, DE, USA. Submitted to WHO by E.I. du
Pont de Nemours and Co., Inc., Wilmington, DE, USA.
