
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 161
PHENOL
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
First draft prepared by Ms G.K. Montizaan,
National Institute of Public Health and
Environmental Hygiene, Bilthoven, Netherlands
Published under the joint sponsorship of
the United Nations Environment Programme,
the International Labour Organisation,
and the World Health Organization
World Health Orgnization
Geneva, 1994
The International Programme on Chemical Safety (IPCS) is a
joint venture of the United Nations Environment Programme, the
International Labour Organisation, and the World Health
Organization. The main objective of the IPCS is to carry out and
disseminate evaluations of the effects of chemicals on human health
and the quality of the environment. Supporting activities include
the development of epidemiological, experimental laboratory, and
risk-assessment methods that could produce internationally
comparable results, and the development of manpower in the field of
toxicology. Other activities carried out by the IPCS include the
development of know-how for coping with chemical accidents,
coordination of laboratory testing and epidemiological studies, and
promotion of research on the mechanisms of the biological action of
chemicals.
WHO Library Cataloguing in Publication Data
Phenol.
(Environmental health criteria ; 161)
1.Phenols - standards 2.Environmental exposure
I.Series
ISBN 92 4 157161 6 (NLM Classification: QD 341.P5)
ISSN 0250-863X
The World Health Organization welcomes requests for permission
to reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1994
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material
in this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR PHENOL
1. SUMMARY
1.1. Identity, physical and chemical properties, analytical
methods
1.2. Sources of human and environmental exposure
1.3. Environmental transport, distribution and transformation
1.4. Environmental levels and human exposure
1.5. Kinetics and metabolism
1.6. Effects on laboratory mammals, and in vitro test
systems
1.7. Effects on humans
1.8. Effects on organisms in the environment
1.9. Summary of evaluation
1.9.1. Human health
1.9.2. Environment
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1. Identity
2.2. Physical and chemical properties
2.3. Conversion factors
2.4. Analytical methods
2.4.1. Sampling and pre-treatment
2.4.2. Analysis
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Natural sources
3.2. Anthropogenic sources
3.2.1. Production
3.2.2. Industrial processes
3.2.3. Non-industrial sources
3.3. Endogenous sources
3.4. Uses
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1. Transport and distribution between media
4.2. Abiotic degradation
4.2.1. Air
4.2.2. Water
4.3. Biodegradation
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1. Environmental levels
5.1.1. Air
5.1.2. Water and sediment
5.2. Occupational exposure
5.2.1. Production
5.2.2. Application of phenolic resins
5.2.3. Other occupational situations
5.3. General population exposure
5.3.1. Indoor air
5.3.2. Food and drinking-water
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1. Absorption
6.1.1. Animal uptake studies
6.1.1.1 Pulmonary
6.1.1.2 Dermal
6.1.1.3 Intestinal
6.1.2. Human uptake studies
6.1.2.1 Pulmonary
6.1.2.2 Dermal
6.2. Distribution
6.3. Metabolic transformation
6.3.1. Metabolite identification
6.3.2. Covalent binding to macromolecules
6.3.3. Location
6.4. Elimination and excretion
6.5. Biological monitoring
7. EFFECTS ON LABORATORY MAMMALS, AND IN VITRO TEST SYSTEMS
7.1. Single exposure
7.1.1. LD50 values
7.1.2. Effects
7.2. Short-term exposure
7.2.1. Oral exposure
7.2.2. Dermal exposure
7.2.3. Inhalation exposure
7.2.4. Subcutaneous exposure
7.2.5. Ear exposure
7.3. Skin and eye irritation; sensitization
7.4. Long-term exposure
7.5. Reproduction, embryotoxicity and teratogenicity
7.5.1. Reproductive toxicity
7.5.2. Embryotoxicity/teratogenicity
7.5.2.1 In vivo studies
7.5.2.2 In vitro studies
7.6. Mutagenicity and related end-points
7.6.1. Mutagenicity studies
7.6.1.1 Bacterial systems
7.6.1.2 Non-mammalian eukaryotic systems
7.6.1.3 Mammalian in vitro systems
7.6.1.4 Mammalian in vivo systems: somatic
cells
7.6.1.5 Mammalian in vivo systems: germ cells
7.7. Carcinogenicity
7.7.1. Oral exposure
7.7.2. Dermal exposure
7.7.3. Inhalation exposure
7.7.4. Two-stage carcinogenicity studies
7.8. Special studies
7.8.1. Neurotoxicity
7.8.2. Myelotoxicity
7.8.3. Immunotoxicology
7.8.4. Biochemical effects
8. EFFECTS ON HUMANS
8.1. General population exposure
8.1.1. Controlled studies
8.1.2. Case reports
8.1.2.1 Dermal exposure
8.1.2.2 Oral exposure
8.1.2.3 Inhalation exposure
8.1.2.4 Exposure by injection
8.2. Occupational exposure
8.3. Organoleptic data
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1. Microorganisms
9.2. Aquatic organisms
9.2.1. Freshwater organisms
9.2.1.1 Short-term studies
9.2.1.2 Long-term studies
9.2.2. Marine organisms
9.2.2.1 Short-term studies
9.2.2.2 Long-term studies
9.2.3. Accumulation
9.2.4. Metabolism
9.3. Terrestrial organisms
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1. Evaluation of human health risks
10.1.1. Exposure
10.1.2. Toxicity
10.1.3. Evaluation
10.2. Evaluation of effects on the environment
10.2.1. Environmental levels
10.2.2. Toxicity
10.2.3. Evaluation
11. FURTHER RESEARCH
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
RESUME
RESUMEN
WHO TASK GROUP ON ENVIRONMENTAL HEALTH
CRITERIA FOR PHENOL
Members
Dr L.E. Hansen, dk-Teknik, Soeborg, Denmark
Dr R.J. Kavlock, Developmental Toxicology Division, Health Effects
Research Laboratory, US Environmental Protection Agency,
Research Triangle Park, North Carolina, USA
Dr C.J. Price, Neurotoxicology Program Development, Center for Life
Sciences and Toxicology, Research Triangle Institute, Research
Triangle Park, North Carolina, USA
Mr D. Renshaw, Department of Health, Elephant and Castle, London,
United Kingdom
Dr A. Smith, Health and Safety Executive, Toxicology Unit, Bootle,
Merseyside, United Kingdom ( Joint Rapporteur)
Professor J.A. Sokal, Institute of Occupational Medicine and
Environmental Health, Sosnowiec, Poland ( Chairman)
Dr S.H.H. Swierenga, Health and Welfare Canada, Drugs Directorate,
Ottawa, Ontario, Canada ( Joint Rapporteur)
Dr T. Vermeire, National Institute of Public Health and
Environmental Protection, Toxicology Advisory Centre,
Bilthoven, The Netherlands
Secretariat
Professor F. Valic, IPCS Consultant, World Health Organization,
Geneva, Switzerland, also Vice-Rector, University of Zagreb,
Zagreb, Croatia ( Responsible Officer and Secretary)
NOTE TO READERS OF THE CRITERIA MONOGRAPHS
Every effort has been made to present information in the
criteria monographs as accurately as possible without unduly
delaying their publication. In the interest of all users of the
Environmental Health Criteria monographs, readers are kindly
requested to communicate any errors that may have occurred to the
Director of the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland, in order that they may be
included in corrigenda.
* * *
A detailed data profile and a legal file can be obtained from
the International Register of Potentially Toxic Chemicals, Case
postale 356, 1219 Châtelaine, Geneva, Switzerland (Telephone No.
9799111).
* * *
This publication was made possible by grant number 5 U01
ES02617-14 from the National Institute of Environmental Health
Sciences, National Institutes of Health, USA.
ENVIRONMENTAL HEALTH CRITERIA FOR PHENOL
A Task Group on Environmental Health Criteria for Phenol met at
the British Industrial and Biological Research Association (BIBRA)
Toxicology International, Carshalton, United Kingdom from 26 to 30
April 1993. Dr D. Anderson welcomed the participants on behalf of
the host institution, and Professor F. Valic opened the Meeting on
behalf of the three cooperating organizations of the IPCS
(UNEP/ILO/WHO). The Task Group reviewed and revised the draft
monograph and made an evaluation of the risks for human health and
the environment from exposure to phenol.
The first draft of this monograph was prepared by Ms G.K.
Montizaan, National Institute of Public Health and Environmental
Hygiene, Bilthoven, the Netherlands.
Professor F. Valic was responsible for the overall scientific
content of the monograph and for the organization of the meeting,
and Dr P.G. Jenkins, IPCS, was responsible for the technical editing
of the monograph.
The efforts of all who helped in the preparation and
finalization of the monograph are gratefully acknowledged.
ABBREVIATIONS
DMBA dimethylbenzathraline
EEC European Economic Community
LOAEL lowest-observed-adverse-effect level
MATC maximum acceptable tolerance concentration
NOAEL no-observed-adverse-effect level
NOEL no-observed-effect level
NOLC no-observed lethal concentration
PCE polychromatic erythrocytes
TT toxicity threshold
1. SUMMARY
1.1 Identity, physical and chemical properties, analytical methods
Phenol is a white crystalline solid which melts at 43 °C and
liquefies upon contact with water. It has a characteristic acrid
odour and a sharp burning taste. It is soluble in most organic
solvents; its solubility in water is limited at room temperature;
above 68 °C it is entirely water-soluble. Phenol is moderately
volatile at room temperature. It is a weak acid, and in its ionized
form very sensitive to electrophile substitution reactions and
oxidation.
Phenol may be collected from environmental samples by
absorption in NaOH solution or onto solid sorbents. Desorption is
achieved by acidification, steam distillation and ether extraction
(from solutions) or by thermal or liquid desorption (from solid
sorbents). The most important analytical techniques are gas
chromatography in combination with flame ionization/electron capture
detection, and high-performance liquid chromatography in combination
with ultraviolet detection. The lowest reported detection limit for
air is 0.1 µg/m3. Phenol can be measured in blood and urine; in
urine samples a detection limit of 0.5 µg/litre has been reported.
1.2 Sources of human and environmental exposure
Phenol is a constituent of coal tar, and is formed during the
natural decomposition of organic materials. The major part of phenol
present in the environment, however, is of anthropogenic origin.
Production and use of phenol and its products, especially phenolic
resins and caprolactam, exhaust gases, residential wood burning and
cigarette smoke are potential sources. Another potential source is
the atmospheric degradation of benzene under the influence of light,
whereas the presence of phenol in liquid manure may also contribute
considerably to its atmospheric levels. Benzene and phenol
derivatives may, by in vivo conversion, form a source of
endogenous human phenol exposure.
The worldwide production of phenol appeared to be fairly
constant throughout the 1980s, the USA being the most important
producer. Its major use is as a feedstock for phenolic resins,
bisphenol A and caprolactam. Some medical and pharmaceutical
applications are also known.
1.3 Environmental transport, distribution and transformation
The main emissions of phenol occur to air. The major part of
phenol in the atmosphere will be degraded by photochemical reactions
to dihydroxybenzenes, nitrophenols and ring cleavage products, with
an estimated half-life of 4-5 h. A minor part will disappear from
the air by wet deposition (rain). Phenol is expected to be highly
mobile in soil, but transport and reactivity may be affected by pH.
Phenol in water and soil may be degraded by abiotic reactions
as well as microbial activity to a number of compounds, the most
important being carbon dioxide and methane. The proportion of
biodegradation to the overall degradation of phenol is determined by
many factors, such as concentration, acclimation, temperature, and
the presence of other compounds.
1.4 Environmental levels and human exposure
No data are available on atmospheric phenol levels. Background
levels are expected to be less than 1 ng/m3. Urban/suburban levels
vary from 0.1 to 8 µg/m3, while concentrations in source-dominated
areas (industry) were reported to be up to two orders of magnitude
higher. Phenol has been detected in rain, surface water and ground
water, but data are very scarce. Elevated phenol levels have been
reported in sediments and ground waters due to industrial pollution.
Occupational exposure to phenol may occur during the production
of phenol and its products, during the application of phenolic
resins (wood and iron/steel industry) and during a number of other
industrial activities. The highest concentration (up to 88 mg/m3)
was reported for workers in the ex-USSR quenching coke with
phenol-containing waste water. Most other reported concentrations
did not exceed 19 mg/m3.
For the general population, cigarette smoke and smoked food
products are the most important sources of phenol exposure, apart
from the exposure via air. Exposure by way of drinking-water and
inadvertently contaminated food products should be low; phenol has
an objectionable smell and taste, which in most cases leads to
non-acceptance by the consumer.
1.5 Kinetics and metabolism
Phenol is readily absorbed by all routes of exposure. After
absorption, the substance is rapidly distributed to all tissues.
Absorbed phenol mainly conjugates with glucuronic acid and
sulfuric acid and, to a lesser extent, hydroxylates into catechol
and hydroquinone. Phosphate conjugation also occurs. The formation
of reactive metabolites (4,4-biphenol and diphenoquinone) has been
demonstrated in in vitro studies with activated human neutrophils
and leucocytes.
The relative amounts of glucuronide and sulfate conjugates vary
with dose and animal species. A shift from sulfation to
glucuronidation was observed in rats after increasing the phenol
dose.
The liver, the lung, and the gastrointestinal mucosa are the
most important sites of phenol metabolism. The relative role played
by these tissues depends on route of administration and dose.
In vivo and in vitro studies have demonstrated covalent
binding of phenol to tissue and plasma proteins. Some phenol
metabolites also bind to proteins.
Urinary excretion is the major route of phenol elimination in
animals and humans. The rate of urinary excretion varies with dose,
route of administration, and species. A minor part is excreted in
the faeces and expired air.
1.6 Effects on laboratory mammals, and in vitro test systems
Phenol has moderate acute toxicity for mammals. Oral LD50
values in rodents range from 300 to 600 mg phenol/kg body weight.
Dermal LD50 values for rats and rabbits range from 670 to 1400
mg/kg body weight, respectively, and the 8-h LC50 for rats by
inhalation is more than 900 mg phenol/m3. Clinical symptoms after
acute exposure are neuromuscular hyperexcitability and severe
convulsions, necrosis of skin and mucous membranes of the throat,
and effects on lungs, nerve fibres, kidneys, liver, and the pupil
response to light.
Solutions of phenol are corrosive to skin and eyes. Phenol
vapours can irritate the respiratory tract. There is evidence that
phenol is not a skin sensitizer.
The most important effects reported in short-term animal
studies were neurotoxicity, liver and kidney damage, respiratory
effects and growth retardation. Toxic effects in rat kidney have
been reported to occur at oral dose levels of 40 mg/kg per day or
more. Liver toxicity was evident in rats administered at least 100
mg/kg per day. In a limited 14-day study in rats, an oral
no-observed-adverse-effect level (NOAEL) of 12 mg/kg per day was
reported, based on kidney effects. In this experiment miosis (an
iris response to light) was still inhibited at 4 mg/kg per day;
however, the health significance of this finding is not clear. Some
biological changes were reported to occur in the intestinal mucosa
and kidneys of mice at dose levels below 1 mg/kg per day, a finding
of uncertain toxicological significance.
There are no adequate studies on the reproductive toxicity of
phenol. Phenol has been identified as a developmental toxicant in
studies with rats and mice. In two multiple dose rat studies, NOAEL
values of 40 mg/kg per day (the lowest-observed-adverse-effect level
(LOAEL) was 53 mg/kg per day) and 60 mg/kg per day (the LOAEL was
120 mg/kg per day) have been reported. In the mouse, the NOAEL was
140 mg/kg per day (the LOAEL was 280 mg/kg per day).
The majority of bacterial mutagenicity tests have given
negative results. Mutations, chromosomal damage and DNA effects have
been observed in mammalian cells in vitro. Phenol has no effect on
intercellular communication (measured as metabolic cooperation) in
cultured mammalian cells. Induction of micro-nuclei in bone marrow
cells of mice has been observed in some studies. No micronuclei were
observed in mice studies at lower doses.
Two carcinogenicity studies have been conducted with male and
female rats and mice receiving phenol in their drinking-water.
Malignancies (e.g., C-cell thyroid carcinoma, leukaemia) were only
seen in low-dose male rats. No adequate dermal or inhalation
carcinogenicity studies have been conducted. Two-stage
carcinogenicity studies have shown that phenol, applied repeatedly
to mouse skin, has promoting activity.
1.7 Effects on humans
A wide range of adverse effects has been reported following
well-documented human exposure to phenol by the dermal, oral or
intravenous routes. Gastrointestinal irritation has been reported
following ingestion. Local effects following dermal exposure range
from painless blanching or erythema to corrosion and deep necrosis.
Systemic effects include cardiac dysrhythmias, metabolic acidosis,
hyperventilation, respiratory distress, acute renal failure, renal
damage, dark urine, methaemoglobinaemia, neurological effects
(including convulsions), cardiovascular shock, coma and death. The
lowest reported dose resulting in a human death was 4.8 g by
ingestion; death occurred within 10 min.
The potential for poisoning through inhalation of phenol
vapours has long been recognized, but no cases of death following
this route of exposure have been reported. Symptoms associated with
inhalation of phenol included anorexia, weight loss, headache,
vertigo, salivation and dark urine.
Phenol is not a sensitizing agent.
The human odour threshold for phenol has been reported to range
from 0.021 to 20 mg/m3 in air. The odour threshold for phenol in
water has been reported to be 7.9 mg/litre, and the taste threshold
0.3 mg/litre in water.
Adequate human data on the carcinogenicity of phenol are not
available.
1.8 Effects on organisms in the environment
In studies on single bacteria species, the EC50 values found
for growth inhibition varied from 244 to 1600 mg phenol/litre. A
toxicity threshold of 64 mg phenol/litre was found. Values for
protozoa and fungi were of the same order of magnitude as for
bacteria; for algae, they were somewhat lower.
Phenol is toxic to higher freshwater organisms. The lowest
LC50 or EC50 values for crustaceans and fish lie between 3 and 7
mg phenol/litre. The data on the acute toxicity to marine organisms
are comparable with those for freshwater organisms. In long-term
studies on crustacea and fish species, a remarkable difference in
sensitivity has been observed; the LC1 values from embryo-larval
tests on Salmo and Carassius proved to be much lower (0.2 and 2
µg phenol/litre, respectively) than the corresponding values for
other fish species (NOLC 2.2-6.1 mg/litre) and amphibia, or from
reproduction tests on crustacea (NOLC 10 mg phenol/litre). Data from
long-term tests on marine organisms are not available.
The bioconcentration factors of phenol in various types of
aquatic organisms are in general very low (< 1-10), although some
higher values (up to 2200) have also been reported. Phenol,
therefore, is not expected to bioaccumulate significantly.
The available data concerning the fate and effects of phenol in
terrestrial organisms are very scarce. A 120-h EC50 for millet was
found to be 120-170 mg/litre, and in a contact test the LC50 for
earthworm species was 2.4-10.6 µg/cm2.
1.9 Summary of evaluation
1.9.1 Human health
The general population is primarily exposed to phenol by
inhalation. Repeated oral exposure may arise from consumption of
smoked food or drinking-water.
Data are inadequate to determine the degree of exposure of the
general population, but an upper-limit estimate of the daily intake
can be made. On the basis of "the worst case scenario", an estimate
can be made assuming that an individual will be maximally exposed to
phenol through continuous inhalation of heavily contaminated air
with frequent consumption of smoked food items and of drinking-water
containing phenol up to the taste threshold. The estimated maximal
total daily intake of phenol for such a 70-kg individual is
calculated to be 0.1 mg/kg body weight per day.
The lowest NOAEL values identified in animal experiments are
for kidney and developmental effects, and in rats lie within the
range of 12-40 mg/kg body weight per day. Using an uncertainty
factor of 200, a range of 60-200 µg/kg body weight per day is
recommended as the upper limit of the total daily intake (TDI).
Considering the upper-limit estimate of human daily intake of 100
µg/kg body weight per day, it is concluded that the average general
population exposure to phenol from all sources is below this range.
A reason for concern is some evidence that phenol may be
genotoxic and the fact that there is insufficient data to discount
with certainty the possible carcinogenicity of the compound. The
evaluation must be kept under periodic review.
1.9.2 Environment
Phenol is not expected to bioaccumulate significantly. Phenol
is toxic to aquatic organisms; an environmental concern level of
0.02 µg/litre can be determined applying the modified US EPA method.
Adequate data on plants and terrestrial organisms are lacking.
Intercompartmental transport of phenol mainly occurs by wet
deposition and by leaching through soil. Generally, the compound is
not likely to persist in the environment. The scarce exposure data
do not allow the evaluation of the risk from phenol to either
aquatic or terrestrial ecosystems. However, in view of the derived
environmental concern level for water, it is reasonable to assume
that aquatic organisms may be at risk in any surface or sea water
contaminated with phenol.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1 Identity
Chemical formula: C6H6O
Chemical structure:
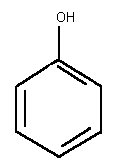 Relative molecular mass: 94.11
Common name: phenol
Common synonyms: acidum carbolicum, acidum phenolicum,
acidum phenylicum, benzaphenol, benzene
phenol, benzenol carbolic acid,
hydroxybenzene (IUPAC), oxybenzene,
monohydroxybenzene, monophenol, phenic
acid, phenol alcohol, phenyl hydrate,
phenyl hydroxide, phenylic acid
Common trade names: carbololie (NLD), fenololie (NLD),
kristalliertes Kreosot (GER),
Steinkohlenkreosot (GER),
Steinkohlenteerkreosot (GER), venzénol
(FRA), ENT 1814.
CAS registry number: 108-95-2
CAS chemical name: phenol
2.2 Physical and chemical properties
Some physical and chemical properties of phenol are given in
Table 1.
Table 1. Some physical and chemical properties of phenola
Boiling point (101.3 Pa) 181.75 °C
Melting point 43 °C
40.9 °C (ultrapure material)
Relative density (20 °/4 °)b 1.071
Relative vapour density (air = 1) 3.24
Vapour pressure (20 °C) 0.357 mmHg
(50 °C) 2.48 mmHg
(100 °C) 41.3 mmHg
Saturation concentration in air (20°C) 0.77 g/m3
Solubility in water (16 °C) 67 g/litrec
Log n-octanol/water partition
coefficient (log Pow) 1.46d
Dissociation constant in water
at 20 °C (Ka) 1.28 x 10-10
Flash-point (closed cup) 80 °C
(open cup) 79 °C
85 °Ce
Flammability limits 1.3-9.5%
a From: Kirk-Othmer (1980); RIVM (1986)
b Weast (1987)
c Above 68.4 °C phenol is entirely soluble in water
d The Pow of phenol is very much dependent on pH; pH at log
Pow = 1.46 was not given
e Budavari et al. (1989)
Phenol has a melting point of 43 °C and forms white to
colourless crystals (Budavari et al., 1989). It has also been
described as a colourless to pink solid or thick liquid (NIOSH,
1985a). Phenol has a characteristic acrid smell and a sharp burning
taste. Odour and taste threshold values are reported in section 8.3.
In the molten state, it is a clear, colourless liquid with a low
viscosity. A solution with approximately 10% water is called
phenolum liquefactum, as this mixture is liquid at room temperature.
Phenol is soluble in most organic solvents (aromatic hydrocarbons,
alcohols, ketones, ethers, acids, halogenated hydrocarbons). The
solubility is limited in aliphatic solvents.
The chemical properties of phenol are affected by the resonance
stabilization possibilities of phenol and, in particular, of the
phenolate ion. Because of this, phenol reacts as a mild acid. In the
presence of electrophilic groups (meta-indicators), the acidic
properties are emphasized.
Phenol is sensitive to oxidizing agents. Splitting of the
hydrogen atom from the phenolic hydroxyl group is followed by
resonance stabilization of the resulting phenyloxy radical. The
radical formed can easily be further oxidized. Depending on the
oxidizing agent applied and the reaction conditions, various
products, such as dihydroxy- and trihydroxybenzenes and quinones are
formed. These properties make phenol suitable as an antioxidant,
functioning as a radical trapping agent. Phenol undergoes numerous
electrophilic substitution reactions, such as halogenation and
sulfonation. It also reacts with carbonyl compounds in both acidic
and alkaline media. In the presence of formaldehyde, phenol is
readily hydroxymethylated with subsequent condensation to resins.
2.3 Conversion factors
1 mg/m3 = 0.26 ppm
1 ppm = 3.84 mg/m3
2.4 Analytical methods
Analytical methods for phenol are shown in Table 2.
2.4.1 Sampling and pre-treatment
Phenol in air samples may be collected by absorption in NaOH
solution contained in wash bottles or on filters impregnated with
NaOH solution. Phenol in air, water and solid waste samples may be
collected (directly or after extraction) in tubes containing solid
sorbent (Tenax, silica gel or, less commonly, carbon) (IARC, 1989).
For large air volumes, the NaOH method is usually preferred, whereas
for smaller quantities (personal air sampling, for instance) solid
sorbent tubes have been reported to be more practical (RIVM, 1986).
Release of phenol from aqueous solutions (including NaOH
sorbent, and also urine) is achieved by acidification, steam
distillation and/or ether extraction. After adsorption onto Tenax,
thermal desorption at 250 °C is usually preferred (the whole sample
may be inserted directly into a gas chromatograph), whereas, in the
case of silica gel, liquid desorption with chloroform is generally
applied. There is a small possibility of chemical conversion during
heating, whereas Tenax may react with ozone to form small quantities
of phenol. Only one analysis per sample is possible in the case of
thermal desorption. The use of liquid desorption allows more
analyses per sample, but because of the unavoidable dilution the
detection limit is higher (RIVM, 1986).
Table 2. Methods for the detection of phenol in air
Sampling Volume of air Pre-treatment Analysis Detection limit Reference
(litre) before analysis (µg/m3)
Absorbance in NaOH solution 100 acidification GC and FID 10 µg per NIOSH (1984)
in a wash bottle; 1 litre/min sample
Absorbance in NaOH solution 25 000 acidification and GC and FID 4 Katz (1977)
in an impinger; 20 litre/min steam distillation
Glass fibre filter impregnated with 600 acidification and GC and FID 13 Kifune (1979)
NaOH and glycerol; 120 litre/min extraction with ether
Absorbance in NaOH solution in 1000 acidification and colorimetry with 2 Katz (1977)
an impinger; 28 litre/min steam distillation 4-amino antipyrine
Absorbance in NaOH solution - none colorimetry with 700 Hensehler (1975)
in a wash bottle; 1 litre/min 4-amino antipyrine
Absorbance in NaOH solution in 150 conversion in an HPLC with UV detector 0.2 Kuwata et al.
a sinter wash bottle; 1-2 litre/min azophenol derivate (254 nm) (1980)
Absorbance in Na2CO3 solution; 30 calibration at pH = 10 UV at 235 nm at 2 pH 160 Zavorovskaya &
1 litre/min and pH = 7 values Nekhorosheva (1981)
Absorbance in NaOH solution in 300 calibration at pH = 12 UV at 241 and 295 nm 20 Bergshoeff (1960)
an impinger; 28 litre/min and at pH = 6
Tenax with and without KOH; 5 thermal desorption; GC and FID 0.1 Hoshika & Muto
0.25 litre/min 250 °C (1979)
Tenax; 0.1 litre/min 4 thermal desorption; GC and FID 1 Russell (1975)
260 °C
Tenax; 0.5-1 litre/min 70 thermal desorption; GC and MS 0.3 Hagemann et
250 °C al. (1978)
Table 2 (contd)
Sampling Volume of air Pre-treatment Analysis Detection limit Reference
(litre) before analysis (µg/m3)
Tenax; 0.75 litre/min 90 thermal desorption; IR with 20 m gas 300 Podolak et al.
300 °C cuvette (1981)
Silica gel 25 liquid desorption GC and FID 3 Dimitriev &
with chloroform Mishchikhin (1983)
Silica gel; 0.05-2 litre/min 10 liquid desorption HPLC with UV detector 50 Oomems &
with chloroform (275 nm) Schuurhuis (1983)
Drägen tube gas detection 0.5 none reading of colorisation 19 000 Leichnitz (1982)
2.4.2 Analysis
The most important analytical techniques for the detection of
phenol are gas chromatography (GC) in combination with flame
ionization detection (FID), and high-performance liquid
chromatography (HPLC) in combination with ultraviolet (UV)
detection. The identification of phenol by GC/FID has been improved
by reaction of phenols with bromide or pentafluoro-benzyl bromide,
and the use of electron capture detection (Hoshika & Muto, 1979; US
EPA, 1986a). The identification of phenol using HPLC can be improved
by reaction with, for example, p-nitrobenzene diazonium
tetrafluoroborate to form azo derivatives (Kuwata et al., 1980).
Detection limits of the above techniques for air samples are
given in Table 2. For the GC/FID detection of phenol in water, using
electron capture detection following derivatization with
pentafluorobenzyl bromide, a detection limit of approximately 0.2
µg/litre has been reported (US EPA, 1986a).
GC in combination with mass spectrometry (MS) is more sensitive
than with FID, but is more expensive. This technique, using either
packed or capillary columns, was reported to have practical
quantitative limits of approximately 1 mg phenol/kg wet weight for
soil/sediment samples, 1-200 mg phenol/kg for wastes, and 10
µg/litre for groundwater samples (US EPA, 1986b,c).
Another reported analytical technique is colorimetry after
reaction of phenol with 4-amino antipyrine, in the presence of
potassium ferricyanide, to form an antipyrine dye. The detection
limit of this technique for water samples, after steam distillation,
was reported to be 1 µg/litre (American Public Health Association,
1985). For air samples of 1 m3, the detection limit was reported
to be 2 µg/m3 (see Table 2). The interference by para-substituted
phenols and chlorophenols is low (RIVM, 1986).
Infrared (IR) detection of phenol is a rather insensitive
method, and is highly susceptible to interference by other compounds
such as water vapour. However, it is a rapid and specific method
which allows directly readable continuous measurement. It is
considered to be attractive only at air concentrations of more than
1000 µg/m3, for example in leakage tests and industrial warning
systems (RIVM, 1986). Also directly readable is the Dräger gas
detection tube (see Table 2); however, the detection limit is very
high (> 19 000 µg/m3).
For the GC/FID detection of total phenol in urine samples,
after acidification and ether extraction, a detection limit of 0.5
µg/litre was estimated (NIOSH, 1985b). Colorimetric methods for the
determination of free phenol in both urine and blood are available.
In one method, phenol reacts with p-nitroaniline following
deproteinization and extraction with diethyl ether. Other phenols
will interfere (Müting et al., 1970). In another method, phenol
reacts with ammonia and N-chlorosuccinimide in alkaline media with
sodium nitroprusside as a catalyst. This method was found to be
applicable in the range of 3-24 mg/litre, using spiked samples of
urine (Amlathe et al., 1987). The concentration of total phenol in
urine and plasma can be determined by GC/MS following hydrolysis of
glucuronide and sulfate conjugates with sulfuric acid and
derivatization with propanoic anhydride. The detection limit is
reported to be 10 µg/litre (Pierce & Nerland, 1988).
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural sources
Phenol is a constituent of coal tar, and is formed during the
natural decomposition of organic materials. Increased environmental
levels may result from forest fires (Hubble et al., 1981).
Phenol has been detected among the volatile components from
liquid manure at concentrations of 7-55 µg/kg dry weight (Spoelstra,
1978). In the Netherlands, for example, the contribution from this
source to the overall phenol emission into air in 1983 has been
calculated to be 15%, assuming complete volatilization of phenol and
an average phenol concentration in manure of 30 µg/kg dry weight
(RIVM, 1986).
3.2 Anthropogenic sources
3.2.1 Production
The most commonly used production method for phenol, on a
worldwide scale, is from cumene (isopropylbenzene). In the USA, for
example, more than 98% of phenol is produced by this method (IARC,
1989). Phenol is also produced from chlorobenzene and toluene. A
small but steady supply of phenol is recovered as a by-product of
metallurgical coke manufacture (IARC, 1989). The emission factor of
phenol into air during production by the cumene process has been
reported to be 0.16 g phenol emitted per kg phenol produced (UBA,
1981).
In Table 3, information is presented concerning the production
of phenol in various countries in 1981. This information was derived
from the open literature (Chemfacts, 1978-1981; United Nations,
1980; SRI, 1982; CID-TNO, 1984; IARC, 1989) and, where necessary,
was extrapolated to 1981. There have been no major production
changes according to data available up to 1986 (IARC, 1989).
3.2.2 Industrial processes
Phenol is the basic feedstock from which a number of
commercially important materials are made, including phenolic
resins, bisphenol A (2,2-bis-1-hydroxyphenylpropane), capro-lactam,
alkyl phenols, as well as chlorophenols such as pentachlorophenol
(IARC, 1989).
Table 3. Production of phenol in 1981 and 1986 (kilotonne/year)
Country Production Production
in 1981a in 1986b
Brazil 50
Bulgaria 35
Czechoslovakia 44 46
Finland 32
France 150
Germany, Federal
Republic of 247
Italy 223
India 14
Japan 215 260
Mexico 20
The Netherlands 166
Poland 66
Romania 66
Spain 55 70
United Kingdomc 110 53
USA 1350 1413
USSR 497 515
Other countries 34
European Community (total) 920
Total 3374
a Chemfacts (1978-1981); United Nations (1980); SRI (1982); CID-TNO
(1984); IARC (1989)
b From IARC (1989)
c Phenol is no longer produced in the UK
The most important phenol emissions result from the use of
phenolic resins. Phenolic resins are used as a binding material in,
for example, insulation materials, chipboard and triplex, paints and
casting sand foundries. Their contents vary from 2-3% for insulation
material to > 50% for moulds (Bollig & Decker, 1980). Emissions are
approximately proportional to the concentration of free phenol,
which is present as a monomer in these materials (1-5%) (Bollig &
Decker, 1980). In addition, phenol may be released as a result of
thermal decomposition of the resins.
In foundries, phenol emissions develop both during the
production of moulds and kernels and during founding (TNO, 1978).
The content of free phenol may rise by up to 12% (Ryser & Ulmer,
1980). Emission factors reported by RIVM (1986) were 0.35 g phenol
emitted per kg used casting sand, 2-5 g phenol emitted per kg resin
in the production of casting sand, and 10 g phenol emitted per kg
resin during the production of moulds by the "hot-box" procedure.
Other industrial activities in which phenol may be emitted to
the air, as well as some of their reported emission factors, are
listed below:
* production of phenol resins (0-0.5 g phenol emitted per kg
resin produced) (RIVM, 1986)
* production of phenols and phenol derivatives
* production of caprolactam (0.02-0.05 g phenol emitted per kg
cyclohexanone (an intermediate) produced (RIVM, 1986)
* production of cokes
* production of insulation materials
* process emissions
Emissions to water may also result from processing.
3.2.3 Non-industrial sources
Phenol has been detected in the exhaust gases of private cars
at concentrations of 0.3 ppm (approximately 1.2 mg/m3) to 1.4-2.0
ppm (5.4-7.7 mg/m3) (Kuwata et al., 1980; Verschueren, 1983). It
has also been identified in cigarette smoke, in quantities that are
comparable to an average emission of 0.4 mg/cigarette (Groenen,
1978). Emission gases from all material incinerators and home fires,
especially wood-burning, may contain substantial quantities of
phenol (Den Boeft et al., 1984).
Another potential source of phenol is the atmospheric
degradation of benzene under the influence of light (Hoshino &
Akimoto, 1978).
Phenols have been detected in smoked foods (section 5.3.2).
3.3 Endogenous sources
An important additional source of human phenol exposure may be
the in vivo formation from various xenobiotics, e.g., benzene
(Pekari et al., 1992).
3.4 Uses
The largest single use of phenol is the production of phenolic
resins. Next is its use in the production of caprolactam, an
intermediate in the production of nylon 6, and 2,2-bis-1-
hydroxyphenylpropane (bisphenol A), which is mainly used in the
production of phenolic resins (Kirk-Othmer, 1980).
The various applications of phenol as a percentage of total
1981 consumption, in the USA and western Europe, are summarized in
Table 4 (Kirk-Othmer, 1980). The data presented are in close
agreement with the 1986 USA data reported by IARC (1989).
Table 4. Use of phenol in 1981 (% total consumption)a
Production of USA West Europe
Phenolic resins 48 36
Bisphenol A(2,2-bis-1-
hydroxyphenylpropane) 18 17
Caprolactam 15 28
Other products 19 19
Total 100 100
a From: Kirk-Othmer (1980)
Phenol was widely used in the 19th century for wound treatment
and as an antiseptic and local anaesthetic. The medical uses of
phenol today include incorporation into disinfectants, antiseptics,
lotions, salves and ointments (IARC, 1989). Another medical
application of phenol is its use as a neurolytic agent, applied in
order to relieve spasm and chronic pain (Wood, 1978).
In addition to the applications mentioned in section 3.2.2,
phenol is used in the manufacture of paint and varnish removers,
lacquers, paints, rubber, ink, illuminating gases, tanning dyes,
perfumes, soaps and toys (IARC, 1989).
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1 Transport and distribution between media
No data have been found concerning wet and dry deposition of
phenol. Since phenol in air is present almost exclusively in the gas
phase, dry deposition (by particle deposition) is expected to be
negligible. Wet deposition may contribute to the disappearance of
phenol from the atmosphere: when phenol was measured during seven
episodes of rain in Portland, Oregon, USA, relatively high
concentrations were found in the rain water (Leuenberger et al.,
1985).
Based on its relatively high solubility in water and the
relatively low vapour pressure at room temperature, phenol is
expected to end up largely in the water phase upon distribution
between air and water. Consequently, transport from air to soil and
water is likely (RIVM, 1986). Volatilization from dry near-surface
soil should be relatively rapid (Howard, 1989).
Theoretical deposition rates for phenol were estimated assuming
a behaviour similar to SO2, and comparing with the rate of reaction
of phenol with hydroxyl radicals (see below). Based on this
comparison, it was concluded that most phenol in the atmosphere is
degraded chemically, rather than transported (RIVM, 1986).
Partition coefficient (Koc) values of phenol for two silt
loams were reported to be 39 and 91 dm3/kg. Based on these Koc
values, phenol would be expected to be highly mobile in soil, and
therefore may leach to ground water (Howard, 1989). This was
confirmed by Scott et al. (1982) who found low adsorption of
phenol to two sterile silt loams (pH 5.4, organic matter content 1.1
and 3.6, respectively), as shown by Freundlich K values of 0.57 and
1.19, respectively. Based on the pKa (log (1.28 x 10 -10)),
phenol exists in a partially dissociated state in water and moist
soils and, therefore, its transport and reactivity may be affected
by pH (Howard, 1989). Upon measurement of the sorption and
desorption of phenol from water to surface sediment (pH 6.21-6.35;
organic matter content of fine fraction (< 2 µM) was 10.2%), phenol
appeared to bind strongly to the soil. The estimated Koc was 2900
dm3/kg (Isaacson & Frink, 1984). However, no correction was made
for any degradation occurring during the experiments. The adsorption
of phenol onto soil or microbial biomass may be decreased by the
presence of phenol derivatives (Boyd, 1982; Selvakumar & Hsieh,
1988). Phenol has been detected in ground water as a result of
leaching (see section 5.1.2).
4.2 Abiotic degradation
4.2.1 Air
Phenol may react in air with hydroxyl and NO3 radicals, and
undergo other photochemical reactions to form dihydroxy-benzenes,
nitrophenols, and ring cleavage products (Atkinson et al., 1979;
Bruce et al., 1987). The half-life for phenol in air was found to
be 4-5 h under photochemically reactive conditions in a smog chamber
(Spicer et al., 1985); this is in good agreement with the
estimated half-life of phenol in air of 5 h based on its estimated
reaction rate with hydroxyl radicals (RIVM, 1986). Howard (1989)
reported an estimated half-life of 15 h for the reaction of phenol
with hydroxyl radicals in air. The reaction of phenol with nitrate
radicals during the night may be a significant removal process; a
half-life of 15 min has been estimated at an atmospheric
concentration of 2x108 nitrate radicals per cm3 (Howard, 1989).
Phenol absorbs light in the region of 290-330 nm and therefore
could photolyse (Howard, 1989).
4.2.2 Water
Phenols generally react in sunlit natural water via reaction
with photochemically produced hydroxyl and peroxy radicals; typical
half-lives were reported to be 100 and 19.2 h, respectively (Howard,
1989).
Phenol was found to be oxidized to carbon dioxide in water
under experimental conditions (temperature approximately 50 °C), in
the presence of oxygen and sunlight, at a rate of 11% per 24 h
(Knoevenagel & Himmelreich, 1976). It was reported to react with
nitrate ions in dilute aqueous solutions to form dihydroxybenzenes,
nitrophenols, nitrosophenol and nitroquinone, presumably by a
radical mechanism involving hydroxyl and phenoxyl radicals (Niessen
et al., 1988). Phenol has been found to react with nitrous acid in
waste water to form cyanide (Adachi et al., 1987), and to form
chlorophenols in chlorinated drinking-water (Jarvis et al., 1985)
and p-benzoquinone in the presence of chlorine dioxide (Wajon et
al., 1982).
4.3 Biodegradation
Bacteria play a major role in the degradation of phenol in
soil, sediment and water. The number of bacteria capable of
utilizing phenol is usually a small percentage of the total
population present in, for example, a soil sample (Hickman & Novak,
1989). However, repeated phenol exposure may result in acclimation
(the promotion of strains capable of utilizing phenol as food)
(Young & Rivera, 1985; Colvin & Rozich, 1986; Shimp & Pfaender,
1987; Wiggins & Alexander, 1988; Tibbles & Baecker, 1989).
Phenol may be converted by bacteria under aerobic conditions to
carbon dioxide (Southworth et al., 1985; Ursin, 1985; Aelion et
al., 1987; Dobbins et al., 1987; Aquino et al., 1988), and
under anaerobic conditions to carbon dioxide (Bak & Widdell, 1986;
Tschech & Fuchs, 1987) or methane (Healy & Young, 1979; Ehrlich et
al., 1982; Young & Rivera, 1985; Fedorak & Hrudey, 1986; Fedorak
et al., 1986). Benzoate, catechol, cis-cis-muconate,
ß-ketoadipate, succinate and acetate have all been identified as
intermediates in the biodegradation of phenol (Paris et al., 1982;
Krug et al., 1985; Fedorak et al., 1986; Knoll & Winter, 1987).
Some of the carbon derived from the degradation of phenol may be
incorporated into the bacterial biomass (Chesney et al., 1985).
Phenol may be degraded in its free form as well as after
adsorption onto soil or sediment, although the presence of sorbent
reduces the rate of biodegradation (Shimp & Young, 1987; Knezovich
et al., 1988).
When phenol is the only carbon source, it can be degraded in a
biofilm reactor with first-order kinetics at concentrations below
about 20 µg/litre at 10 °C. The first-order rate constants are 3 to
30 times higher than those of easily degraded organic compounds at
100- to 1000-fold higher concentrations (Arvin et al., 1991).
Reported phenol degradation rates suggest rapid aerobic degradation
in sewage (typically > 90% with an 8-h retention time), soil
(typically complete biodegradation in 2-5 days), fresh water
(typically complete biodegradation in < 1 day), and sea water
(typically 50% in 9 days) (Howard, 1989). Anaerobic biodegradation
is slower (Baker & Mayfield, 1980).
The contribution of bacteria to the overall rate of degradation
may be affected by a number of factors such as phenol concentration
(Baker & Mayfield, 1980; Ursin, 1985; Hwang et al., 1989),
temperature (Baker & Mayfield, 1980; Bak & Widdell, 1986; Hwang et
al., 1986; Thornton-Manning et al, 1987; Gurujeyalashmi & Oriel,
1989), sunlight (Hwang et al., 1986), soil depth (Dobbins et
al., 1987; Federle, 1988), the presence of other nutrients
required for bacterial growth (Rubin & Alexander, 1983; Fedorak &
Hrudey, 1986; Rozich & Colvin, 1986; Thorton-Manning et al.,
1987), the presence of other pollutants (Southworth et al., 1985;
Hoffmann & Vogt, 1988; Wang et al., 1988; Namkoong et al., 1989)
and bacterial abundance (Tranvik et al., 1991).
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air
No data are available for background levels of phenol in air,
away from emission sources. They are expected to be low (< 1 ng
phenol/m3) (RIVM, 1986).
Higher levels of phenol in air may be expected for urban areas,
mainly due to traffic emissions. Urban phenol concentrations have
been reported for Osaka, Japan (1-4 µg phenol/m3; Kuwata et al.,
1980), Nagoya, Japan (0.2-8 µg phenol/m3 with an average of 1.7 µg
phenol/m3; Hoshika & Muto, 1979, 1980), Paris, France (0.7-8 µg
phenol/m3; Hagemann et al., 1978), and Portland, USA (0.22 to
0.42 µg phenol/m3; Leuenberger et al., 1985). Despite
differences in analytical techniques, the first three series of
measurements showed good agreement. The Portland results were lower,
but came from air samples taken during rain periods; phenol was also
detected in rain water (see section 5.1.2).
Ambient air levels of phenol have been extensively monitored in
the highly industrialized and urbanized Upper Silesia region of
Poland (Sanitary Epidemiological Station, Katowice, 1991). Levels
during 1990 ranged from 3.8 to 26.6 µg/m3, the highest values
being in the areas of greatest industrial concentration.
Hoshika & Muto (1979, 1980) reported a phenol level of
approximately 190 µg/m3 "near" a phenolic resin factory (no
details). Kuwata et al. (1980) found phenol levels of 0.8-3.5
mg/m3 in foundry emissions (no details).
Based on limited data, median ambient atmospheric levels of
phenol (based on estimated 24-h averages) were estimated by
Brodzinsky & Singh (1982) to be 0.12 µg/m3 for urban/suburban
areas (7 samples) in the USA (which is lower than reported above for
several cities), and 104 µg/m3 (2-170 µg/m3) for source-
dominated areas (83 samples) in the USA.
5.1.2 Water and sediment
Levels of phenol dissolved in rain water from Portland, USA,
were found to range from 0.08 to 1.2 µg/litre and averaged above
0.28 µg/litre; gas phase concentrations ranged from 220-410 ng/m3
(Leuenberger et al., 1985).
Concentrations reported for surface water in the Netherlands
were 2.5-6.5 µg/litre for two major rivers, 0.3-7 µg/litre for
lakes, and 1.5 µg/litre for coastal waters (the given concentrations
include other phenolic substances) (RIVM, 1986). Industrial rivers
in the USA were reported to contain 0-5 µg/litre, but 3-24 µg/litre
was reported for Lake Huron. Phenol was also detected in 2/100 raw
water supplies in 1977 in the US EPA National Organics Monitoring
Survey (Howard, 1989).
Drinking-water levels of phenol in the USA have been reported
to be around 1 µg/litre or otherwise below the detection limit
(summarized by Howard, 1989). Phenol was detected (no quantitative
data) in drinking-water in the USA from 5 out of 14 drinking-water
plants surveyed and in Great Britain in 2 out of 4 sites (Fielding
et al., 1981). Higher groundwater levels have been reported
following industrial activity (e.g. 6.5-10 000 µg/litre in two
aquifers 15 months after a coal gasification project; summarized by
Howard, 1989). Phenol was detected at a maximum concentration of
1130 mg/litre in nine wells in Wisconsin after a spill, and was
detectable for at least 1.5 years after the spill (Delfino & Dube,
1976).
Phenol was not detected in water samples from three areas in
Japan analysed for an environmental survey; however, levels of
0.03-0.04 mg/litre were detected in 3 out of 9 bottom sediment
samples from the same regions (Fujii, 1978).
Other sediment concentrations reported were 13 000 µg/kg in
samples from Lake Huron, "not detected" in an unspecified industrial
river in the USA, < 1000 µg/kg (dry weight) as the median
concentration in 9% of sediment samples from 318 data points in the
USA, and 10 µg/kg (dry weight) in samples collected 6 km from a
wastewater treatment discharge zone in California (summarized by
Howard, 1989).
Phenol was detected in 63 out of 165 sediments from sampling
areas in the Puget Sound region (Tetra Tech. Inc., 1986). Half the
samples had a concentration of phenol below 40 µg/kg sediment (dry
weight); the maximum level was 1700 µg/kg.
Levels of phenol with means of 0.01-5.7 mg/litre (maximum up to
53 mg/litre) have been reported in effluents from various industrial
sources (summarized by Howard, 1989). Highest levels were associated
with the iron and steel industry. Limited quantitative data from the
VIEW Database (ATSDR, 1989) for ground water at hazardous waste
sites indicated maximum levels of 2.48 to 85 000 µg/litre (average
33 800 µg/litre, 6 data points).
No data have been found indicating the presence of phenol in
soil. Phenol is not likely to persist in soil because of rapid
biodegradation (section 4.3) or transport to ground water or air
(section 4.1).
5.2 Occupational exposure
Occupational exposure to phenol may occur during the production
of phenol and phenol derivatives, during the production of phenolic
resins and other products derived from phenol, during processing of
the latter materials, and during a number of other activities.
5.2.1 Production
Personal air samples of workers involved in the production of
phenol by the cumene process in the ex-USSR contained on average 5.8
mg phenol/m3. For workers occupied in the production of phenol
from chlorobenzene, the mean exposure level was 1.2 mg phenol/m3
(Mogilnicka & Piotrowski, 1974). Values reported in the same
publication for workers in two phenol resin-producing industries
were 0.6-3 mg phenol/m3.
5.2.2 Application of phenolic resins
Occupational exposure during the processing of phenolic resins
appears to be partly determined by the content of free phenol in the
applied resin (Bollig & Decker, 1980; Ryser & Ulmer, 1980).
In the wood industry, indoor phenol concentrations of 0.3 mg
per m3 (Winkler, 1981) and 1.5 mg/m3 (range 0.8-2.6) (Gilli et
al., 1980) have been reported. Concentrations in the breathing
zone of wood workers were 1.3-2.6 mg phenol/m3 (Gspan et al.,
1984). In another study, concentrations of < 0.04 to 1.9 mg
phenol/m3 were found at plywood plants (Kauppinen et al., 1986).
In iron and steel foundries, average hourly phenol
concentrations of 0.4-4.5 mg/m3 were reported in the manufacture
of moulds or kernels (Schütz & Wolf, 1980). Phenol concentrations of
1-4 mg/m3 were measured in a foundry in Osaka, Japan (Kuwata et
al., 1980). Local phenol concentrations were reported to be as
high as 75-420 mg/m3 due to the thermal degradation of the resin.
However, this effect of thermal degradation was not reflected in
hourly concentrations measured during the foundry process: values of
3-16 mg phenol/m3, with an average of 10 mg phenol/m3, were
reported by Schütz & Wolf (1980), and a maximum hourly average of
2.7 mg phenol/m3 was reported by Ryser & Ulmer (1980). (It is not
known whether these results were obtained in personal or area air
samples). Phenol concentrations during the operation of an electric
furnace in a steel factory in Pueblo, Colorado, USA, were 0.04, 0.18
and 0.20 mg/m3 in the vicinity of the furnace. General room air
samples taken during operation of a grey iron foundry were below the
detection limit (Gunter, 1987).
5.2.3 Other occupational situations
Exposure levels of 5-88 mg phenol/m3 have been reported for
employees in the ex-USSR who quenched coke with waste water
containing 0.3-0.8 g/litre phenol (Petrov, 1960).
Measurements at coal gasification and liquefaction plants in
the USA showed relatively low phenol concentrations (< 0.08 mg
per m3) at various sites (Dreibelbis & Hawthorne, 1985).
In a Japanese bakelite factory, area samples contained 0-12.5
mg phenol/m3 (Ohtsuji & Ikeda, 1972).
In a synthetic fibre factory in Japan, concentrations of 19 mg
phenol/m3 were measured, whereas in a USA fibrous glass wool
factory concentrations of 0.05-1.3 mg phenol/m3 were reported
(Dement et al., 1973; Ogata et al., 1986).
The concentrations of phenol in creosote vapour, analysed in
seven creosote impregnation plants in Finland, ranged from < 0.1 to
1.8 mg/m3 air (Heikkilä et al., 1987). The highest exposures
occurred during the cleaning of creosote warming chambers.
During the dissection of cadavers by dental students, phenol
breathing zone concentrations ranged from 5 to 19 mg phenol/m3.
(The high phenol concentrations resulted from the applied embalming
solution, in combination with inadequate ventilation) (Boiano,
1985).
5.3 General population exposure
5.3.1 Indoor air
Borovik & Dmitriev (1981) found a maximal concentration of 0.02
µg phenol/m3 in hospitals in the ex-USSR. It is, however, not
clear from where the phenol originated; it may have been used as a
disinfectant in these hospitals.
No information has been found with regard to phenol
concentrations in residential houses and apartments. Cigarette
smoking must be considered as the most important potential source in
dwellings. A distinction should be made between the main stream (the
smoke inhaled by the smoker) and the side stream (produced by the
smouldering cigarette itself). It was estimated that 0.01-0.22 mg
phenol per cigarette was released in the mainstream, while the
sidestream phenol content was 2.6 times higher. In the case of
various Japanese cigarettes, 0.3-0.4 mg phenol was emitted into air
during burning (Kuwata et al., 1980). For an unventilated room of
50 m3, the smoking of one cigarette would thus result in a phenol
concentration of 6-8 µg/m3.
5.3.2 Food and drinking-water
Phenol is found in smoked meat and fish products. The wood
smoke with which such products are treated contains, among other
ingredients, a wide range of phenols and phenol ethers, which
contribute significantly to the characteristic smoke aroma (smell
and taste) of the product.
Phenol is absorbed into the food products during smoking.
Quantitative data, however, are scarce, since phenols are usually
determined as a group. According to Toth (1982), the total phenol
content of smoked sausage is 70 mg/kg; Bratzler et al. (1969)
found a content of 37 mg/kg in the outer layer of the product, and
lower contents in the inner part. Luten et al. (1979) determined a
number of individual phenols in smoked herring and found a phenol
content, depending on the duration of smoking, of approximately
10-30 mg/kg. Potthast (1976, 1982) measured 2-18 mg/kg in smoked ham
and liver sausage.
If liquid smoke derivates are used in order to give a smoky
flavour to fish and meat products, the end product also contains
phenol. However, with regard to smell and taste aspects, phenol is
not the most important phenolic compound from wood smoke. In this
respect, methoxy and dimethoxy phenols are more important, together
with aliphatic fatty acids and carboxyl compounds.
Phenol may also enter food unintentionally by, for instance,
contamination in transport or from packaging materials, or contact
with other phenol-containing materials. However, these accidental
cases would probably be detected and lead to non-acceptance by the
consumer, owing to the conspicuous phenol smell and taste (see
section 8.3 for odour and taste thresholds).
Phenol has been found in botton pits fish from 5 sites in
Commencement Bay in Tacoma, USA, at a maximum average of 0.14 mg/kg
and an overall maximum of 0.22 mg/kg (Nicola et al., 1987).
Little information is available with respect to the occurrence
of phenol in drinking-water. Surface and ground waters intended for
the production of drinking-water in the Netherlands were reported to
contain 1-9 µg phenol/litre (phenol index, including other phenolic
compounds) (RIVM, 1986). Phenol was found in a domestic water supply
in the USA at a level of 1 µg/litre (Ramanathan, 1984). Cases of
drinking-water pollution with phenol have been reported in the UK
and the USA; the phenol water concentrations were reported to be
5-10 µg and 5-120 mg per litre, respectively (see section 8.1.2.2).
Chlorination of drinking-water may result in the formation of
chlorophenols from phenol, which greatly adds to the objectionable
smell and taste (Jarvis et al., 1985).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
Phenol and conjugated metabolites of phenol occur naturally in
animal and human tissue and can be detected in the urine, faeces,
saliva and sweat. The body's production of phenol depends on the
type of diet: a high protein or meat diet promotes phenol formation.
6.1 Absorption
Phenol is readily absorbed through all routes, such as the
lungs, intact and abraded skin, and the gastrointestinal tract of
both humans and animals (Von Oettingen, 1949; Deichmann & Keplinger,
1963).
6.1.1 Animal uptake studies
6.1.1.1 Pulmonary
There are no in vivo data on absorption of phenol following
inhalation exposure. However, in vitro studies by Hogg et al.
(1981), using 14C-phenol with excised trachea-lung preparations
and isolated perfused rat lung, demonstrated that phenol can be
rapidly and efficiently absorbed in the lungs.
6.1.1.2 Dermal
The extent of absorption of phenol through rabbit skin is more
strongly influenced by the area of the skin exposed than by the
concentration of the applied solution in water (Deichmann &
Witherup, 1944; Liao & Oehme, 1980).
In studies with the hairless mouse, phenol destroyed the
stratum corneum (Behl et al., 1983a). Similar effects were
reported by Huq et al. (1986) and Jetzer et al. (1986).
Absorption of phenol through thermally damaged mouse skin in vitro
was also reported to be greatly enhanced (Behl et al., 1983b). In
contrast, Deichmann et al. (1952) observed that injury of the
rabbit skin caused by phenol appeared to retard the rate of
absorption.
The permeability of mouse skin to phenol from aqueous solution
in vitro increased with increasing temperature of the carrier
solution (from 10 to 37 °C) (Jetzer et al., 1988).
Measurement of the permeation constant of phenol through
hairless mouse skin at 37 °C in vitro yielded a value of 18 800 ±
3000 cm/h (Huq et al., 1986; Jetzer et al., 1986).
6.1.1.3 Intestinal
When a single oral dose of 25 mg/kg body weight was
administered to rats, pigs or sheep, more than 95% was absorbed (Kao
et al., 1979).
In vitro studies showed that aqueous solutions of phenol
placed into ligated sections of the gastrointestinal tract had the
fastest absorption rate in the colon, followed by the ileum. The
absorption rate in the stomach was much slower (Deichmann &
Keplinger, 1963).
6.1.2 Human uptake studies
6.1.2.1 Pulmonary
The retention of phenol in the bodies of eight human volunteers
exposed to 6-20 mg/m3 by inhalation only for 8 h was 70%-80%
during the course of the study (Piotrowski, 1971). Ohtsuji & Ikeda
(1972) reported similar observations.
6.1.2.2 Dermal
Human skin absorption of phenol vapour (5-25 mg/m3) occurs
rapidly (Ruedemann & Deichmann, 1953). Fatal cases reflect the rapid
rate of absorption of phenol through the skin (Turtle & Dolan, 1922;
Duverneuil & Ravier, 1962; Hinkel & Kintzel, 1968; Lewin & Cleary,
1982). The retention in eight human volunteers, exposed to phenol
vapour at concentrations of 6-20 mg/m3, by skin only, for 6 h was
70-80% (Piotrowski, 1971). Piotrowski (1971) proposed the following
formula for calculating the absorption rate of phenol vapour through
the skin:
A = (0.35)C
where A is the amount of phenol absorbed in mg/h per unit area
and C is the phenol air concentration in mg/m3.
Concentrations of between 5 and 10% phenol denature epidermal
protein, and this can partly prevent absorption. The phenol-protein
complex is not stable and by dissociation of phenol the substance
may exert its action over a period of time (Schmidt & Maibach,
1981).
Phenol was detected in the urine of 4 out of 16 infants (2-5
months) with seborrhoeic eczema who were skin-painted twice daily
for 48 h with a commercial paint containing 4% (w/v) phenol and 8%
(w/v) resorcinol (Rogers et al., 1978). In adults, a single
topical application of 4 µg phenol/cm2 on 13 cm2 of the ventral
forearm, reportedly gave an absorption of 4.4% of the administered
dose (Feldman & Maibach, 1970). The period of exposure and the
concentration of phenol are both factors that determine the extent
of absorption (Piotrowski, 1971; Roberts et al., 1977;
Baranowska-Dutkiewicz, 1981).
In vitro studies have also shown that phenol from aqueous
solutions (1% w/v) readily penetrates human skin (Roberts et al.,
1977, 1978). A value of 8200 cm/h was obtained as the permeation
constant of phenol through human skin at 25 °C (Flynn & Yalkowsky,
1972). In an in vitro study with human abdominal skin, 10.9% of
the applied dose was absorbed. This study showed an excellent
qualitative, but a somewhat less accurate quantitative, agreement
between the in vivo and in vitro skin absorption of 12 compounds
(Franz, 1975).
6.2 Distribution
Phenol is rapidly distributed to all tissues in exposed
animals.
In rabbits, 15 min after oral administration of 0.5 g
phenol/kg, chemical analysis indicated that the liver contained the
highest concentration of total phenol followed by the central
nervous system, lungs and blood. After 82 min, phenol was fairly
uniformly distributed over all tissues. The proportion of free to
conjugated phenol changed with time, and, after 360 min, most of the
phenol was conjugated (Deichmann, 1944).
After a single oral administration of 14C-phenol (207 mg/kg)
to rats, the highest concentration ratios between tissue and plasma
were found in liver (42%), followed by spleen, kidney, adrenal,
thyroid and lungs, with a peak tissue level occurring after 0.5 h
(Liao, 1980; Liao & Oehme, 1981a).
Highest tissue residues were found after 2 h in the kidneys and
livers of mice and rats treated intravenously (Gbodi & Oehme, 1978;
Wheldrake et al., 1978; Greenlee et al., 1981).
6.3 Metabolic transformation
6.3.1 Metabolite identification
Studies employing several species have demonstrated that
conjugation with glucuronic acid and sulfate are major metabolic
pathways for phenol. Hydroxylation to hydroquinone and catechol also
occurs (Williams, 1938, 1959; Garton & Williams, 1949; Bray et
al., 1952a,b,c; Parke & Williams, 1953).
In vitro studies have shown the formation of 4, 4'-biphenol
and diphenoquinone by neutrophils, activated leucocytes and by
horseradish peroxidase following addition of phenol (Eastmond et
al., 1986).
Phenol metabolism in rabbits was studied by Deichmann &
Keplinger (1963). During the first 24 h following oral
administration of a sublethal dose of 300 mg phenol/kg body weight,
23% of the administered dose was recovered as exhaled carbon
dioxide. Trace amounts of catechol and hydroquinone were also
detected in the breath. Over the same period, 72% of the dose was
excreted in the urine (48% of which was excreted as free and 52% as
conjugated phenols), 1% was excreted in the faeces, 4% remained in
the carcass, and trace amounts were exhaled.
Oral administration of 14C-phenol (1.2 mg/kg) to rats
resulted in at least 80% excretion in urine within 24 h, with 68% as
phenyl sulfate and 12% as phenyl glucuronate (Edwards et al.,
1986).
A pronounced shift from sulfation to glucuronidation was
observed in rats after increasing the phenol dose (Koster et al.,
1981). This observed shift is apparently due to a saturation of the
overall sulfation process, rather than to a depletion of inorganic
sulfate (Weitering et al., 1979; Koster et al., 1981; Koster,
1982). A limited availability of 3-phosphoadenosine-5-phosphosulfate
may account for the decreased proportion of phenol conjugation to
sulphate at relatively high doses (Ramli & Wheldrake, 1981).
Repeated administration of phenol, however, did not affect
glucuronide synthesis in rats (Takemori & Glowacki, 1962).
The pig has limited ability for phenol sulfation. The domestic
cat lacks the ability for glucuronic acid conjugation of phenol. In
cats, phenyl phosphate has been detected as a metabolite in small
amounts, in addition to sulfate conjugates (Capel et al., 1974;
French et al., 1974).
Following oral administration of 14C-phenol (0.01 mg/kg) to
three men, 90% of the dose was excreted in the urine within 24 h,
mainly as phenyl sulfate (77%) and phenyl glucuronide. Small amounts
of guinol sulfate and guinol glucuronide were also present (Capel
et al., 1972b).
Several investigators have confirmed the above-mentioned
results using in vitro methods (DeMeio & Arnolt, 1944; Capel et
al., 1972b; Shirkey et al., 1979; Hogg et al., 1981; Koster
et al., 1981; Sawahata & Neal, 1983).
6.3.2 Covalent binding to macromolecules
Early pharmacokinetic studies (measuring distribution volumes)
in dogs, pigs and goats suggested that tissue binding occurs (Oehme,
1969). Further animal studies have indicated that phenol and/or its
metabolites bind covalently to tissue protein, mainly in the liver
(Bolt, 1977; Illing & House, 1980; Jergil et al., 1982; Smart &
Zannoni, 1984). Binding to rabbit bone marrow mitochondrial DNA in
studies with isolated cells has also been reported (Rushmore et
al., 1984). in vivo and in vitro studies have demonstrated
covalent binding of radiolabelled phenol to plasma proteins from
humans, dogs, rats and trout (Liao, 1980; Liao & Oehme, 1981a,b;
Judis, 1982; Schmieder & Henry, 1988). Reactive phenol metabolites
formed by peroxidases bind readily to proteins (Eastmond et al.,
1986, 1987a) and DNA (Subrahmanyam & O'Brien, 1985).
6.3.3 Location
Quantitatively, the most important sites of phenol conjugation
are the liver, lung and gastrointestinal mucosa. The relative roles
played by these tissues depend on the route of administration and
the dose.
The liver is an important site of phenol metabolism. After
direct administration of phenol into the hepatic circulation, the
liver showed considerable first-pass metabolism in rats (Cassidy &
Houston, 1980; Houston & Cassidy, 1982). Phenol-metabolizing enzymes
have been detected in rabbit hepatic microsomes (Koop et al.,
1989).
Other tissues, such as lungs, intestines and kidneys, also play
an important role in phenol metabolism (Quebbemann & Anders, 1973;
Powell et al., 1974; Houston & Cassidy, 1982). Phenol
sulfotransferases, which catalyse phenol sulfation, occur in a
variety of human tissues (intestinal wall, lungs, platelets, adrenal
glands, brain, placenta, etc.) (Campbell et al., 1987; Gibb et
al., 1987). After oral uptake of phenol, there is a very large
first-pass metabolism in the intestines. The lungs also show
considerable first-pass metabolism (as was established after direct
administration into the pulmonary circulation of rats) (Cassidy &
Houston, 1980; Houston & Cassidy, 1982). Due to saturation of
hepatic enzymes, extrahepatic tissues play an increasing role in the
conjugation of phenol as the dose of phenol increases; at doses
higher than 5 mg/kg body weight, intestinal conjugation in rats
exceeds the contribution of the hepatic and pulmonary enzymes
(Cassidy & Houston, 1984).
Myeloperoxidases isolated from human neutrophils and
peroxidative enzymes from activated human leucocytes mediate the
formation of reactive phenol metabolites including 4,4'-biphenol and
diphenoquinone. Myeloperoxidase-mediated hydroxylation occurs in
addition to hepatic cytochrome P-450 oxidation. In several species,
myeloperoxidase activity has been reported in bone marrow, where it
may play a role in phenol metabolism and toxicity (Eastmond et
al., 1986, 1987a; Subrahmanyam et al., 1991).
6.4 Elimination and excretion
Urinary excretion is the major route of phenol elimination in
animals and humans. The rate of excretion varies with dose, route of
administration and animal species (Deichmann, 1944; Capel et al.,
1972a,b). Of 18 animal species studied by Capel et al. (1972a,b),
the 24-h urinary excretion of phenol was greatest in the rat (95% of
the 25 mg/kg body weight oral dose) and the lowest in the squirrel
monkey (only 31% of the dose). Liao & Oehme (1981a,b) reported a
half-life of 4 h in rats.
Five days after oral gavage with 14C-phenol (0.1 mg/kg body
weight), only 0.3% of the applied dose was retained in rats (Freitag
et al., 1985).
Only minor amounts of unchanged phenol are excreted in exhaled
air or in faeces (Deichmann & Keplinger, 1963). Less than 1% of an
orally administered dose of 300 mg phenol/kg body weight to rabbits
was found in the faeces after 24 h (Deichmann, 1944).
Phenol conjugates may also be excreted in the bile of rats
(4.6% of a 50 mg/kg dose) (Abou-el-Makarem et al., 1967). It has
been suggested that biliary excretion of phenol plays an important
role when urinary excretion is impeded. Rats, whose kidneys were
ligated, showed a marked increase in biliary excretion of phenol
metabolites (Weitering et al., 1979). Furthermore, it has been
reported that phenol and its metabolites can undergo enterohepatic
circulation in rats (Gbodi & Oehme, 1978).
Urinary excretion of phenol in human volunteers exposed to
phenol vapour via inhalation (chamber studies) or skin, occurred
with an excretion rate constant of k = 0.2/h. For a one-compartment
model, this corresponds to a half-life of approximately 3.5 h
(Piotrowski, 1971).
6.5 Biological monitoring
The US ACGIH has listed a biological exposure index for phenol
of 250 mg/g creatinine for end-of-shift urine samples (ACGIH, 1991).
The excretion of phenol and phenol conjugates in the urine may
be used as an index of exposure, but it should be noted that there
are other causes that may lead to phenol excretion in the urine. One
of these is benzene exposure; other possible significant sources are
food and drugs (Docter & Zielhuis, 1967; Ikeda & Ohtsuji, 1969;
Fishbeck et al., 1975; Paradowski et al., 1981). Elevated
urinary phenol excretion is thus not a specific index of exposure to
phenol. Furthermore, the large range of "normal" urine values
(phenol concentrations have been found to vary from 0.5 to 81.5
mg/litre) (Deichmann & Schafer, 1942; Docter & Zielhuis, 1967;
Piotrowski, 1971; Gspan et al., 1984; Pekari et al., 1992) would
appear to limit the usefulness of urinary phenol excretion as an
accurate index of low occupational exposure levels.
In volunteers, after a single 8-h exposure to phenol vapour
concentrations of up to 6.8 mg/m3, the phenol excretion in urine
increased up to a maximum of 100 mg total phenol/litre (Piotrowski,
1971). In workers occupationally exposed to 10 mg phenol/m3,
concentrations in urine of up to 262 mg/litre were reported
(Ohtsuji & Ikeda, 1972). However, another recent study, using more
specific methods of analysis, showed good correlation (R=0.91) between
exposure levels in the range 5-17 mg/m3 and the total concentration
of phenyl sulfate and phenyl glucuronide in the urine at the end of the
workshift (Ogata et al., 1986).
7. EFFECTS ON LABORATORY MAMMALS, AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 LD50 values
After oral administration of phenol to mice, rats and rabbits,
LD50 values ranged from 300-600 mg phenol/kg body weight. No
LC50 values have been reported in the published literature.
However, after inhalation of 900 mg phenol/m3 by rats for 8 h, no
deaths were observed. The dermal LD50 (by occlusive and
non-occlusive techniques) was 670 mg/kg body weight for rats and
850-1400 mg phenol/kg body weight for rabbits. LD50 values for
intraperitoneal injection were in the range of 127-223 mg phenol/kg
body weight for rats.
A summary of LD50 values is given in Table 5.
7.1.2 Effects
The acute lethality of phenol, associated with exposure to high
concentrations, is generally attributed to a depressing effect on
the central nervous system (see also section 7.8.1). The clinical
effects of phenol poisoning are independent of the route of
administration. Reported symptoms include neuromuscular
hyperexcitability, including twitching and severe convulsions. Heart
rate at first increases, then becomes slow and irregular. Blood
pressure at first increases slightly, then falls markedly.
Salivation, marked dyspnoea and a decrease in body temperature are
also among the effects reported (Deichmann & Witherup, 1944; Von
Oettingen & Sharples, 1946; Farquharson et al., 1958; Ernst et
al., 1961; Deichmann & Keplinger, 1963; Oehme & Davis, 1970;
Pullin et al., 1978; Liao & Oehme, 1980; Reid et al., 1982).
After oral ingestion, the mucous membranes of the throat and
oesophagus showed swelling, corrosion, and necrosis, with
haemorrhages (Deichmann & Keplinger,1963).
In a study by Schlicht et al. (1992), female Fischer-344 rats
were administered 0, 12, 40, 120 or 224 mg phenol/kg body weight by
gavage in a water vehicle. Animals were examined for clinical signs,
and neurotoxicity and systemic (liver, kidney, adrenal and thymus)
effects, 4-20 h after treatment. Tremors were observed 1-2 min after
dosing in the two highest dose groups. The pupil response to light
(miosis) was significantly inhibited at all dose levels at 24 h
after exposure. Locomotor activity was reduced at 224 mg/kg. At this
dose level, 2/6 animals had hepatocyte necrosis, 4/6 had renal
vascular stasis and 4/6 had necrosis of the thymus. At 120 mg/kg,
liver necrosis was present in 1/7 animals, as was necrosis of the
thymus gland.
Table 5. Acute animal toxicity of phenol LD50 values
Species Route of LD50 values Vehicle Reference
administration (mg/kg body
weight)
Mouse oral 300 Von Oettingen &
Sharples (1946)
Mouse oral 427 Kostovetskii &
Zholdakova (1971)
Rat oral 340-530 2-7% in
water Deichmann &
Witherup (1944)
Rat oral 512 Kostovetskii &
Zholdakova (1971)
Rat oral 445-520 water Thompson & Gibson
(1984)
Rat oral 400 water Schlicht et al.
(1992)
Rat dermal 670 undiluted Conning & Hayes
(570-780) (1970); Brown et al.
(1975)
Rat intraperitoneal 127-223 water or Thompson & Gibson
undiluted
(1984)
Rabbit oral 400-600 2-7% in Deichmann &
water Witherup (1944)
Rabbit dermal 850 Flickinger (1976)
(600-1200)
Rabbit dermal 1400 Vernot et al. (1977)
(740-2670)
In various animal species, inhalation of phenol adversely
affected the lungs, causing hyperaemia, infarcts, bronchopneumonia,
purulent bronchitis and hyperplasia of the peribronchial tissues
(Von Oettingen, 1949).
Sensory irritation was measured in mice by the Alarie assay. A
50% decrease in respiratory rate (RD50) was found at 638 mg
phenol/m3 (De Ceaurriz et al., 1981).
Ocular and nasal irritation, tremors and incoordination were
reported in rats exposed via inhalation to 906 mg/m3 for 8 h
(Flickinger, 1976).
Other pathological abnormalities induced by phenol by various
routes of administration included demyelination of nerve fibres (see
also section 7.8.1), myocardial degeneration and necrosis (Deichmann
& Keplinger, 1963; Liao & Oehme, 1980). Kidney damage (vacuolization
and enlargement of cells) and liver damage (e.g., enlargement of
hepatic cells) were also observed (Oehme & Davis, 1970; Coan et
al., 1982). Urine was usually dark or "smoky" in appearance,
probably due to oxidation products of phenol (Solliman, 1957).
7.2 Short-term exposure
7.2.1 Oral exposure
In a study by Schlicht et al. (1992), groups of eight female
Fischer-344 rats received oral doses of phenol in a water vehicle of
0, 4, 12, 40 or 120 mg/kg body weight daily for 14 days. Tremors
were apparent only after the first dose at the highest level.
Exposure to 120 mg/kg per day was lethal to all rats within 11 days.
The pupil response (miosis) was decreased one day after the last
dose for all but the highest surviving dose group (the incidences
were 100%, 50%, 62% and 76% for the 0, 4, 12 and 40 mg/kg groups,
respectively). Locomotor activity was not affected after the 4th,
9th or 14th dose. No hepatic effects were observed at 40 mg/kg per
day, while 3/8 animals had renal vascular stasis. There were no
histological effects at 12 mg/kg per day. At 40 mg/kg per day, the
pathological changes in the kidneys included two animals with
tubular degeneration in the papillar region, and one with protein
casts in the tubules. The pathological report attributed these
findings to decreased vascular perfusion (MacPhail, personal
communication to the IPCS).
Rats were administered, by gavage, 20 daily doses of 10, 50 or
100 mg phenol/kg body weight. At necropsy, slight effects on liver
and kidneys were reported at 100 mg phenol/kg body weight (Dow
Chemical Company, 1976).
Rats receiving 50 or 100 mg phenol/kg body weight, by gavage,
over a 6-month period (135 doses, presumably daily, 5 days/week)
were reported to show slight to moderate kidney damage.
Administration of 100 mg phenol/kg body weight apparently resulted
in slight liver changes (Dow Chemical Company, 1976).
In a range-finding study, carried out prior to a long-term
carcinogenicity study, mice and rats were provided with tap water
containing 0, 100, 300, 1000, 3000 or 10 000 mg phenol/litre for 13
weeks. Mean body weight gain was decreased only in mice and rats
receiving 10 000 mg phenol/litre (NCI, 1980). In these
drinking-water studies, the highest daily doses were calculated to
be approximately 2000 mg phenol/kg body weight for mice and 1000 mg
phenol/kg body weight for rats.
Phenol was provided to rats in drinking-water for 12 months at
0, 800, 1200, 1600, 2000 and 2400 mg phenol/litre. Depressed weight
gain was observed in rats receiving doses > 2000 mg/litre. The
corresponding daily dose was calculated by the authors of the study
to be > 200 mg/kg body weight (Deichmann & Oesper, 1940).
7.2.2 Dermal exposure
In a study by Deichmann et al. (1950), rabbits were exposed
to 1.18-7.12% phenol in water (64-380 mg phenol/kg body weight) for
5 h/day, 5 days/week, for 18 days. Dose-related systemic effects
(tremors, death) were observed in rabbits exposed to > 2.37%
phenol (130 mg phenol/kg body weight), while skin irritation
(hyperaemia, tissue necrosis) occurred at doses of > 3.56% phenol
(190 mg phenol/kg body weight). This effect was particularly
apparent when the application sites were bandaged.
7.2.3 Inhalation exposure
No studies reported or conducted according to contemporary
standards were available.
In a study by Deichmann et al. (1944), rats, rabbits and
guinea-pigs were exposed to concentrations of 100-200 mg phenol
vapour/m3, 7 h/day, for 5 days/week. Rats exposed for a period of
74 days did not show any gross or microscopic evidence of injury.
Rabbits survived a 3-month exposure but, at autopsy, lung and heart
damage and indications of liver and kidney damage were found.
Guinea-pigs were the most susceptible. Five out of twelve died after
12 days of exposure, and the remaining seven were killed after 29
days of exposure. Prior to death, guinea-pigs showed weight loss,
respiratory difficulties, and signs of paralysis. At autopsy, there
was evidence of acute lobular pneumonia, vascular damage, and
hepatic and renal damage; the total (free and conjugated) phenol
content of the blood was 14 mg/litre. The rabbits had similar, but
less severe, symptoms.
Groups of 10 monkeys, 50 rats and 100 mice were exposed to 19
mg phenol/m3, 8 h/day, 5 days/week for 90 days. Concurrent control
groups were exposed to fresh air only. No deaths occurred and there
was no reduction in weight gain of treated animals. There were no
statistically significant adverse effects observed when the animals
were assessed by a stress test involving swimming performance. A
range of clinical chemistry, haematology and urinalysis parameters
were not affected by exposure to phenol. Routine histology was
performed on the liver, lungs, kidneys, brain and heart. The results
of the percentage of animals showing evidence of "pathological
change" indicated effects in the liver and kidneys of exposed
animals. However, the author of the study concluded that no clinical
or pathological changes occurred that were of toxicological
importance. It is not clear if the upper respiratory tract was
examined in this study in order to look for evidence of irritation
(Sandage, 1961).
Continuous exposure to 100 mg phenol/m3 for 15 days
significantly affected the central nervous system of rats, as was
demonstrated by their performance in the "tilted plane" test. Plasma
levels of potassium, magnesium, lactate dehydrogenase, aspartate
aminotransferase (ASAT), alanine aminotransferase (ALAT) and
glutamate dehydrogenase were elevated. Haemoglobin, haematocrit, and
plasma sodium, calcium and chloride levels were unaffected (Dalin &
Kristofferson, 1974).
7.2.4 Subcutaneous exposure
Subcutaneous exposure to phenol was studied principally to
obtain information about neurological or haematopoietic effects (see
sections 7.8.1 and 7.8.2). No other effects were reported.
7.2.5 Ear exposure
Instillation of phenol (form and amount not specified) into the
inner ear round window of Sprague-Dawley rats caused morphological
damage to the organ of Corti in the basal coil. The outer hair cells
appeared to be more sensitive to phenol than the inner hair cells,
which were mostly intact. As a result of the damage, impairment of
inner ear function was noted (as determined by auditory brain stem
recordings) which was regressive for lower sound frequencies, but
appeared to be permanent for higher frequencies (Anniko et al.,
1988).
7.3 Skin and eye irritation; sensitization
Local damage to the skin, following exposure to phenol, was
found to include erythema, inflammation, discoloration, eczema,
papillomas and necrosis (Deichmann, 1949; Deichmann et al., 1950;
Conning & Hayes, 1970; Pullin et al., 1978). For example, in
rabbits, 0.5 g phenol, moistened with physiological saline, produced
necrosis of both the intact and abraded skin (Flickinger, 1976).
Solutions of 10-14% (v/v) phenol in water have been reported to
cause transient delayed erythema (after 0.5-5 h) and acute vascular
permeability, as assessed by exudation of intravenously injected
Evans blue, in guinea-pigs after dermal treatment for 1 min (Steele
& Wilhelm, 1966).
In one study, an increase in ear thickness was used as an index
of skin irritation (inflammation). Maximal responses to phenol were
observed one hour after application of 1-2 mg phenol to the ear of
female ICR mice. Significant thickening could still be detected 6
weeks after exposure (Patrick et al., 1985).
When phenol, in glycerine dilutions down to 10% or 5% aqueous
solutions, was applied to the rabbit eye, severe damage (complete
destruction to opaque corneas) was seen. Immediate water irrigation
was very effective in preventing the opacity. A delay of 10 seconds
reduced this effectiveness (Murphy et al., 1982).
Fourteen days after the application of 0.1 g phenol to the
rabbit eye, all eyes exhibited keratoconus and pannus formation
(Flickinger, 1976).
Phenol gave negative results in a Magnussen and Kligman skin
sensitization test (Itoh, 1982).
7.4 Long-term exposure
No adequate data are available. Studies on carcinogenicity are
presented in section 7.7.
7.5 Reproduction, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
No adequate studies conducted according to current protocols
are available.
Heller & Pursell (1938) exposed rats to 100-12 000 mg
phenol/litre drinking-water, corresponding to calculated approximate
daily oral doses of 10-1200 mg phenol/kg body weight. General
appearance, growth and fecundity were normal for rats exposed to
100-1000 mg/litre for five generations and to 3000 or 5000 mg/litre
for three generations. Stunted growth was noted in the offspring of
rats exposed to 7000 mg/litre. Many of the offspring died at levels
of 8000 mg/litre because of maternal neglect. At 10 000 mg/litre,
the offspring died at birth, and at 12 000 mg/litre there was no
reproduction.
7.5.2 Embryotoxicity/teratogenicity
7.5.2.1 In vivo studies
Phenol was evaluated for maternal and developmental toxicity in
timed-pregnant Sprague-Dawley rats (20-22 confirmed pregnancies per
group). Distilled water (vehicle) or phenol (30, 60 or 120 mg/kg per
day) was administered daily by gavage in a volume of 5 ml/kg of body
weight throughout the period of major organogenesis (gestational
days 6-15). Dams were weighed on the day of sperm detection
(gestational day 0), prior to daily dosing, and at termination
(gestational day 20); observations for clinical signs of toxicity
were conducted during the treatment period. At termination
(gestational day 20), maternal liver weight, gravid uterine weight
and status of uterine implantation sites (i.e. number of implants,
resorptions, late fetal deaths and live fetuses) for each dam were
recorded. Each live fetus was weighed, sexed and examined for
external morphological abnormalities. Visceral examination of each
fetus was performed using a fresh tissue dissection method;
approximately one-half of the fetal heads from each litter were
fixed (Bouins' solution) and sectioned free-hand for examination of
internal structures; carcasses (one-half without heads) were cleared
and stained with Alizarin Red S prior to skeletal examination. All
control and phenol-treated dams survived to scheduled sacrifice, and
no distinctive treatment-related signs of toxicity were noted.
Pregnancy rates at termination were high (95-100% per group) and no
litters were totally resorbed, so that a total of 20-22 live litters
per group (268-293 fetus per group) was available for examination.
No significant dose-related changes were noted for the following
end-points: maternal body weight (gestational day 0, 6, 11, 15 or
20), maternal body weight gain (treatment period, gestational period
or gestational period corrected for gravid uterine weight), maternal
liver weight, gravid uterine weight, prenatal mortality, live litter
size or incidence of morphological abnormalities (malformations or
variations). However, average fetal body weight per litter was
significantly reduced at the high-dose (93% of average control
weight) (Jones-Price et al., 1983a).
In a study by Kavlock (1990), phenol was administered by oral
gavage to groups of Sprague-Dawley rats on day 11 of gestation (day
1 : sperm plug) at 0, 100, 333, 667 and 1000 mg phenol/kg body
weight. The vehicle used in this study was a 4:4:1:1 mixture of
water, Tween 20, propylene glycol and ethanol. Maternal toxicity
(decreased weight gain) was seen at the two highest doses. Offspring
viability and growth were not affected up to postnatal day 6,but
hind limb paralysis was observed in some offspring in the two
highest dose groups.
In a screening assay, groups of 17-21 Fischer-344 rats received
0, 40 or 53.3 mg phenol/kg body weight by gavage in water on
gestation days 6-15. There were no significant effects on maternal
body weight gain. One of 15 pregnant females resorbed the entire
litter at 40 mg/kg and 2 of 16 did so at 53.3 mg/kg (there were no
similar effects in 153 control litters in the study). All three
females had severe respiratory syndromes (rales and dyspnoea). One
high-dose female with symptoms of respiratory toxicity delivered a
low weight litter that had poor viability. Kinked tails were present
in 2 of 4 surviving pups in that litter. Litter size on postnatal
days 1 and 6 was significantly reduced at 53.3 mg/kg but not at 40
mg/kg. There were no effects on pup body weights on postnatal days 1
or 6 (Narotsky & Kavlock, 1993).
Phenol was evaluated for maternal and developmental toxicity in
timed-pregnant Swiss albino (CD-1) mice (22-29 confirmed pregnancies
per group). Distilled water (vehicle) or phenol (70, 140 or 280
mg/kg per day) was administered daily by gavage in a volume of 10
ml/kg of body weight throughout the period of major organogenesis
(gestational days 6-15). Dams were weighed on the day of vaginal
plug detection (gestational day 0), prior to daily dosing
(gestational days 6-15), and at termination (gestational day 17);
observations for clinical signs of toxicity were conducted during
the treatment period. Evaluation of maternal and developmental
end-points at termination (gestational day 17) were the same as for
rats (see description from the study by Jones-Price et al., 1983a,
above). Toxicity observed at the high-dose level included 11%
mortality (4/36 treated females), clinical signs (especially tremor
and ataxia), reduced maternal body weight (gestational day 17),
reduced maternal body weight gain (treatment period, gestational
period and gestational period corrected for gravid uterine weight),
and a trend only toward reduced maternal liver weight. Pregnancy
rates at termination ranged from 71 to 84%; no litters were totally
resorbed so that 22-29 live litters (214-308 fetuses) were available
for examination. No dose-related changes were noted for prenatal
mortality, live litter size or incidence of morphological
abnormalities, except for an apparent increase in cleft palate
(8/214 fetuses in the high dose versus 0/308 among controls). (It
should be noted that cleft palate is a malformation to which the
CD-1 mouse is predisposed under conditions of maternal stress).
Average fetal body weight per litter was significantly reduced (82%
of average control weight) in the highest dose group (Jones-Price
et al., 1983b).
In a study by Minor & Becker (1971), groups of Sprague-Dawley
rats were given 20, 63, or 200 mg phenol/kg body weight
intraperitoneally on days 9-11 or 12-14 of gestation. Fetal body
weight was reduced in the highest dose group treated on days 12-14.
No gross anomalies were observed, and intrauterine death was not
increased at any dose level.
7.5.2.2 In vitro studies
In the chick embryotoxicity screening test (CHEST), 130
substances were tested. For each compound, 120 selected White
Leghorn Fowl embryos, aged 1.5, 2, 3 and 4 days of incubation, were
used. Phenol did not exhibit embryotoxic properties in this test up
to 100 µg, and was one of the least embryotoxic compounds tested
(Jelinek et al., 1985).
In a study by Oglesby et al. (1992), phenol was added to
cultures of five rat embryos on gestational day 10 at
concen-trations of 0 to 100 µg/ml. Embryos were examined 42 h later
for viability, growth and morphology. Viability was not affected at
any concentration, but a low incidence of tail defects was observed
at 100 µg/ml, and embryonic growth was decreased at 75 and 100
µg/ml. When hepatocytes isolated from pregnant rats were co-cultured
with the embryos, the toxicity to the embryos was increased. Tail
defects were observed at 25 and 50 µg/ml, and growth was reduced at
these concentrations. Without the presence of hepatocytes, phenol
was the least toxic of 13 para-substituted phenols tested in this
system; however, it was the only one which became more embryotoxic
when hepatocytes were present.
When phenol was added to cultures of human embryonic palatal
mesenchyme cells, cell growth was 50% inhibited at a concentration
(IC50) of 0.8 mM (78 µg/litre) (Pratt & Willis, 1985).
7.6 Mutagenicity and related end-points
Data on mutagenicity and related end-points are summarized in
Tables 6, 7, 8 and 9, respectively.
7.6.1 Mutagenicity studies
7.6.1.1 Bacterial systems
Phenol has been tested for mutagenicity by a number of authors
in various strains of Salmonella typhimurium and was shown to be
reproducibly negative, both with and without metabolic activation
(Epler et al., 1979; Gilbert et al., 1980; Rapson et al.,
1980; Pool & Lin, 1982; Haworth et al., 1983). A positive effect
was observed in strain TA98 in the presence of an exogenous
metabolic activation system in a study employing a modified culture
medium (Wild et al., 1980; Gocke et al., 1981).
A positive effect was reported for phenol in a fluctuation test
with strain TA100 after metabolic activation, but no data were given
on toxicity (Koike et al., 1988; abstract). A positive result was
reported in a mutation test with Escherichia coli B/Sd-4; however,
the applied dose levels were highly toxic (Demerec et al., 1951).
7.6.1.2 Non-mammalian eukaryotic systems
Negative data were obtained in the absence of exogenous
metabolic activation with the eukaryotic microorganism
Saccharomyces cerevisiae. At high doses and in the presence of an
activation system, a positive result was obtained (Cotruvo et al.,
1977). Phenol induced mitotic segregation in Aspergillus nidulans
(Crebelli et al., 1987).
Table 6. Tests for genotoxicity in bacteria
Species Strain Measured end-point Test conditions Metabolic Resultsb Reference
activationa
Escherichia coli Sol-4 reverse mutation 0.1-0.2%; 3-24 h - + Demerec et al.
(survival less than 2%) (1951)
AB1899 nm filamentation 10-500 µg/ml; 3-4 h - Nagel et al.
(1982)
Salmonella TA100 reverse mutation fluctuation test - - Koike et al.
typhimurium 0-500 ng/well + (rat) + (no data (1988)
on toxicity)
TA98 reverse mutation 1000-fold concentration + and - - Epler et al.
TA100 range in DMSOc (1979)
TA1535 reverse mutation 0-100 µg/plate - - Gilbert et al.
(1980)
TA1538 reverse mutation 0-50 µg/plate - -
TA98 reverse mutation 0-3333 µg/plate in - - Haworth et al.
TA100 DMSO; preincubation + (rat) - (1983)
TA1535 + (hamster) -
TA1537
TA98 reverse mutation 0-3333 µg/plate in - - Haworth et al.
TA100 preincubation H2O; + (rat) - (1983)
TA1535 + (hamster) -
TA1537
TA98 reverse mutation 0.5-5000 µg/plate in + and - (rat) - Pool & Lin
TA100 DMSO (5000 µg toxic) (1982)
TA1535
TA1537
TA1538
Table 6 (contd).
Species Strain Measured end-point Test conditions Metabolic Resultsb Reference
activationa
TA100 reverse mutation 0.1-1000 µg/plate - - Rapson et al.
(1980)
TA98 reverse mutation 0-100 µmol/plate (99.5% - - Wild et al.
purity with 0.15% + (rat) + (1980)
cresols as main impurity,
non-standard media)
a + = present; - = absent
b + = positive; - = negative
c DMSO = dimethyl sulfoxide
Table 7. Tests for genotoxicity in non-mammalian eukaryotic systems
Species Strain Measured end-point Test conditions Metabolic Resultsa Reference
activation
Fungi
Saccharomyces D3 mitotic recombination 10-5, 10-3 dilution of - - Cotruvo et al.
cerevisiae phenol in saline + (rat) + (1977)
Aspergillus mitotic segregation 5-20 mM - + Crebelli et al.
nidulans (1987)
Insects
Drosophila Oregon-R SLRLb phenol vapour 24 h; - Sturtevant
melanogaster 0.2, 0.25, 0.5% in - (1952)
saline, injection
0.01, 0.1, 1.2% in -
Holtfreter solution;
vaginal douch
Berlin K SLRL 50 nM in 5% saccharose; - Wild et al.
feeding, 3 broods F1 (1980); Gocke
generation et al. (1981)
injection - Woodruff et al.
(1985)
Fish
Salmo gairdneri chromosomal aberrations 0.3-0.6 µl/litre, 72 h + Al-Sabti (1985)
a + = positive; - = negative
b SLRL = sex-linked recessive lethal mutations
Table 8. In vitro phenol genotoxicity in mammalian cells
Species Cell type End-pointa Conditions Activationb Resultc Reference
Chinese hamster CHO-WBL CA 500-800 µg/ml - - Ivett et al. (1989)
2000-3000 µg/ml + (rat) +
Chinese hamster V79 lung forward mutation 0-500 µg/ml + (mouse) + Pashin & Bakhitova
HPRT (500 µg/ml toxic) (1982)
Chinese hamster CHO-WBL SCE 300-400 µg/ml - + Ivett et al. (1989)
2000-3000 µg/ml + (rat) +
Chinese hamster V79 lung intercellular not reported - - Chen et al. (1984)
communication
V79 lung intercellular 250 µg/ml - - Malcolm et al. (1985)
communication
V79 lung intercellular 10-75 µg/ml - - Bohrman et al. (1988)
communication
Mouse L5178Y lymphoma forward mutation 600-1800 µg/ml + and - ? McGregor et al.
TK (1988)
L5178Y forward mutation 180-890 µg/ml - + Wangenheim &
(530 µg/ml toxic) Bolcsfoldi (1988)
5.6-41 µg/ml + (rat) +
(20 µg/ml toxic)
Mouse L5178Y DNA synthesis 9.4-940 µg/ml - + Pellack-Walker
inhibition et al. (1985)
L5178Y DNA strand breaks 16-470 µg/ml - - Garberg et al.
16-470 µg/ml + (rat) + (1988)
Table 8 (contd).
Species Cell type End-pointa Conditions Activationb Resultc Reference
L5178Y DNA strand breaks 94 µg/ml - - Pellack-Walker & Blumer
(1986)
Human T-lymphocytes SCE 0.47-282 µg/ml - + Erexson et al. (1985)
lymphocytes SCE 188 µg/ml - - Jansson et al. (1986)
lymphocytes SCE 1.7-470 µg/ml + (rat) + Morimoto & Wolff
(470 µg/ml toxic) (1980)
lymphocytes SCE 282 µg/ml + (rat) + Morimoto et al.
(1983)
Human fibroblast DNA repair 0.094-9400 µg/ml + Poirier et al. (1975)
HeLa DNA synthesis 188 µg/ml + (rat) + Painter & Howard
inhibition (1982)
WI-38 DNA synthesis 0.094-9400 µg/ml + Poirier et al. (1975)
inhibition
a CA = chromosome aberrations; HPRT = hypoxanthine guanine phosphoribosyl transferase locus; TK = thymidine kinase locus;
SCE = sister chromatid exchange
b - = absent; + = present;
c - = negative; + = positive
Table 9. Phenol genotoxicity in in vivo mammalian systems
Species/Strain Measured end-point Test conditions Remarks Resultsa Reference
(sampling times)
Mouse/CD-1 micronuclei in bone 265 mg/kg, oral bone marrow + Ciranni et al.
marrow (0, 18, 24, 42 or 48 h) depression (1988a)
Mouse/CD-1 micronuclei in maternal bone gestation day 13, 265 mg/kg, maternal bone marrow + Ciranni et al.
marrow and fetal liver oral (15, 18, 24, 30, 36 or 40 h) depression (1988a)
Mouse/CD-1 micronuclei in bone 250 mg/kg, oral (30 h) convulsive seizures - Gad-El Karim et
marrow al. (1986)
Mouse/CD-1 micronuclei in bone 265 mg/kg, i.p. bone marrow + Ciranni et al.
marrow (18, 24, 42 or 48 h) depression (1988a)
Mouse/CD-1 micronuclei in bone 40, 80 or 160 mg/kg, i.p. no bone marrow - Barale et al.
marrow (18 h) depression (1990)
Mouse/NMRI micronuclei in bone 47, 94 or 188 mg/kg, i.p. no information on - Gocke et al.
marrow 0, 24 h (30 h) toxicity (1981)
Mouse/Porton chromosomal aberrations in 2 ml of 0.08, 0.8 or 8 mg/litre + Bulsiewicz (1977)
spermatogonia, primary solution, oral, daily for five
spermatocytes generations
Rat/Sprague- chromosomal aberrations in 72-180 mg/kg, i.p. LD1-LD30, no change - Thompson &
Dawley bone marrow 300-510 mg/kg, oral (20 h) in mitotic index Gibson (1984)
Rat/Sprague- DNA strand breaks (alkaline 7.9, 26 or 79 mg/kg, i.p. (2.6 - Skare & Schrotel
Dawley elution in rat testis) or 24 h) 4, 13.2 or 39.5 mg/kg, (1984)
i.p. for 5 days
a + = positive; - = negative; i.p. = intraperitoneal injection
In Drosophila melanogaster, no statistically significant
sex-linked recessive lethals were obtained after exposure to phenol
via a variety of techniques (Sturtevant, 1952; Wild et al., 1980;
Gocke et al., 1981; Woodruff et al., 1985). However, when an
unusual technique was used, i.e. exposing isolated gonads in vitro
and implanting them in host larvae, several types of mutations were
induced (Hadorn & Niggli, 1946).
When rainbow trout ( Salmo gairdneri) were exposed to phenol
for 72 h at concentrations of 0.3 and 0.6 µl phenol/litre water, the
percentage of chromosomal aberrations in gill and kidney tissue was
significantly increased, at both concentrations, in a dose-related
way. Of these aberrations, 30% were structural, 45% consisted of
aneuploidy, and 25% were non-specified metaphases (Al-Sabti, 1985).
7.6.1.3 Mammalian in vitro systems
Data on in vitro genotoxicity in mammalian cells are given in
Table 8.
In a Chinese hamster V79 lung cell/HPRT mutation test, phenol
gave a positive result with metabolic activation. The highest dose
decreased survival by approximately 50% (Pashin & Bakhitova, 1982).
In a mouse lymphoma L5178Y cell/TK mutation test, there were
statistically significant and dose-related increases in mutation
frequency in the presence and absence of metabolic activation
(Wangenheim & Bolcsfoldi, 1988). However, in another laboratory this
test yielded non-conclusive results (McGregor et al., 1988).
As part of the US NTP testing program, phenol was evaluated for
induction of chromosomal aberrations and sister chromatid exchange
(SCE) in Chinese hamster ovary (CHO) cells (Ivett et al., 1989).
At a delayed harvest time (22.5 h), there were significantly
increased incidences of aberrations in cultures that included a
metabolic activation system from induced rat liver. Although a
dose-response effect was seen, the frequency of aberrations in the
absence of activation was low and the authors reported a negative
result. Regarding SCE induction, positive results were obtained,
both with and without activation. In additional studies, phenol
induced SCE in human lymphocytes in vitro, both in the presence
and in the absence of metabolic activation (Morimoto & Wolff, 1980;
Morimoto et al., 1983; Erexson et al., 1985). Negative results
(SCE) have also been reported (Jansson et al., 1986).
Phenol gave negative results in three studies in Chinese
hamster V79 cell metabolic cooperation assays (Chen et al., 1984;
Malcolm et al., 1985; Bohrman et al., 1988).
7.6.1.4 Mammalian in vivo system: somatic cells
In a bone marrow micronucleus test, groups of four Swiss CD-1
mice (sex not specified) were orally administered 265 mg phenol/kg
body weight and were sacrificed at 0, 18, 24, 42 and 48 h. Bone
marrow depression (decreased polychromatic erythrocytes/normocytes
(PCE/NCE) ratio) persisted at least up to 48 h after dosing. A
slight, but statistically significant, increase in the number of
micronuclei was seen at 24 h (3 micronuclei/1000 cells versus 1.5
micronuclei/1000 cells; 3000 cells scored per mouse) (Ciranni et
al., 1988b).
In a further study to asses the transplacental clastogenicity
of phenol, groups of 4 pregnant Swiss CD-1 mice received 265 mg
phenol/kg/body weight by oral gavage on day 13 of gestation. After
15, 18, 24, 30, 36 or 40 h, animals were sacrificed and adult bone
marrow cells and fetal liver cells were scored for micronuclei.
Slight, but statistically significant, increases in the frequency of
micronucleated PCE in adult bone marrow were observed at 15, 18 and
24 h (3.8, 4.0 and 5.0 micronuclei/1000 cells, respectively,
compared with 2/1000 for negative controls). A statistically
significant reduction in the PCE/NCE ratio was seen at 18 and 36 h.
Phenol had no effect on the frequency of micronuclei in fetal liver
(Ciranni et al., 1988a).
Bone marrow liver cells were also evaluated for the formation
of micronuclei, 30 h after oral administration of 0 or 250 mg
phenol/kg body weight to groups of five males Swiss CD-1 mice
(Gad-El Karim et al., 1986). Uptake was indicated by convulsive
seizures in all mice receiving phenol (1000 PCEs scored per mouse).
Phenol (265 mg/kg body weight), administered to Swiss CD-1 mice
by a single intraperitoneal injection, was reported to increase the
frequency of micronuclei in bone marrow PCEs 18 h post-treatment (7
micronuclei/1000 cells). The increased frequency decreased at 24 h
and was no longer statistically significant at 42 h. A decreased
PCE/NCE ratio persisted in tests up to 48 h post-treatment (Ciranni
et al., 1988b).
Barale et al. (1990) reported a negative result in a bone
marrow micronucleus test in Swiss CD-1 mice 18 h after treatment
with phenol. There was no effect on the PCE/NCE ratio.
Gocke et al. (1981) briefly reported a negative result in a
bone marrow micronucleus test, in which NMRI mice were sampled at 30
h, following i.p. injection of 47-188 mg phenol/kg. No information
on toxicity was given.
The results of these and other studies are summarized in Table
9.
7.6.1.5 Mammalian in vivo systems: germ cells
Skare & Schrotel (1984) obtained negative results in studies of
DNA strand breakage in rat testis. In one experiment, rats received
by intraperitoneal injection 0, 7.9, 26 or 79 mg phenol/kg body
weight and were sacrificed at 2.6 or 24 h post-treatment. Similar
results were obtained with further groups of rats that received 4,
13.2 and 39.5 mg phenol/kg body weight daily for 5 days before
sacrifice.
In an unconventional study involving dosing (0, 6.4, 64 and 640
mg phenol/kg body weight) of five successive generations of male and
female mice, large numbers of structural and numerical chromosomal
aberrations were reported in spermatocytes and spermatozoa, with
dose- and generation-related increases. The study was carried out
with 138 male mice from an inbred stock after skin testing
(Bulsiewicz, 1977).
7.7 Carcinogenicity
The evidence for the carcinogenicity of phenol in experimental
animals, based on the studies summarized below, was recently
considered by the IARC (1989) to be inadequate. Phenol was
classified by US EPA in Group D (data inadequate for evaluating the
carcinogenic potential) (Bruce et al., 1987).
7.7.1 Oral exposure
In an NCI (1980) study, groups of 50 male and 50 female
B6C3F1 mice were given drinking-water containing 0, 2500 or 5000
mg phenol/litre for 103 weeks. As matched controls, groups of 50
male and 50 female mice received tap water. There was a dose-related
decrease in water consumption and mean body weight gain in all
groups of mice. In mice receiving 5000 mg phenol/litre, an increase
in the number of uterine endometrial stromal polyps (5/48 = 10%) was
observed (in matched controls the incidence was 1/50 = 2%). There
was no evidence of an increased incidence of malignant tumours. The
other observed neoplasms were of the usual number and type found in
mice of this strain and age (NCI, 1980).
Groups of 50 male and 50 female Fischer-344 rats received 0,
2500 or 5000 mg phenol/litre drinking-water for 103 weeks, while the
matched control group received tap water. Male and female rats given
5000 mg/litre showed a decrease in mean body weight from week 20
onwards. There were statistically significant increased incidences
of phaeochromocytomas, leukaemias, lymphomas and C-cell thyroid
carcinomas in males of the low-dose group (NCI, 1980). NTP
considered this study negative for carcinogenicity due to the lack
of a dose-response for the neoplasms and the lack of response in
females.
7.7.2 Dermal exposure
Three studies examined the potential carcinogenicity of phenol
following dermal application (Rusch et al., 1955, Boutwell et
al., 1956; Bernard & Salt, 1982). However, none is considered
adequate for the evaluation of carcinogenicity due to the short
duration of exposure and/or use of inappropriate vehicles.
7.7.3 Inhalation exposure
No studies have been reported for this route of exposure.
7.7.4 Two-stage carcinogenicity studies
A dose of 3 mg phenol in acetone was applied to ICR/Ha Swiss
mice 3 times per week for 52 weeks after initiation with 150 µg
DMBA. Papilloma development was enhanced, compared with that of mice
exposed to DMBA alone (Van Duuren et al., 1968; Van Duuren &
Goldschmidt, 1976). These observations were in agreement with those
from earlier reports on the promotional activity of phenol (Boutwell
et al., 1955, 1956; Salamon & Glendenning, 1957; Boutwell & Bosch,
1959; Wynder & Hoffmann, 1961).
A dose of 3 mg phenol in acetone applied 3 times weekly for 460
days to female ICR/Ha Swiss mice after initiation with 5 µg
benzo[a]pyrene had a slight promoting activity. Simultaneous
application of both agents showed a partial reduction in carcinomas
compared with mice treated with benzo[ a]pyrene alone (Van Duuren
et al., 1971, 1973; Van Duuren & Goldschmidt, 1976).
7.8 Special studies
7.8.1 Neurotoxicity
Tremors, convulsions, coma and death were reported after
intraperitoneal and subcutaneous doses of phenol (Deichmann &
Witherup, 1944; Ernst et al., 1961; Windus-Podehl et al., 1983).
The tremors were enhanced by prior monoamine depletion following
reserpine treatment (Suzuki & Kisara, 1985).
Upon histological examination of rats in which convulsions had
been induced by subcutaneous phenol injection (200 mg/kg body weight
given once a week for two weeks), two out of six animals showed
spinal cord and spinal root degeneration (Veronesi et al., 1986).
Phenol, given either intravenously or intra-arterially (250
µg), facilitated neuromuscular transmission and antagonized
neuromuscular blockade by D-tubocurarine in cats. The effect was
determined to be pre-synaptic in origin (Blaber & Gallagher, 1971).
Phenol caused diminution of the compound action potential in
preparations of the saphenous nerve after acute and chronic
perfusion (Schaumburg et al., 1970).
Groups of five male CD-1 mice were supplied with drinking-water
containing 0, 4.7, 19.5 or 95.2 mg phenol/litre for 4 weeks, at the
end of which the concentrations of various neurotransmitters and
their metabolites were measured in different parts of the brain. The
largest effects were seen in levels of noradrenaline in the
hypothalamus (significant decreases of 29 and 40% in the mid- and
high-dose groups) and of dopamine in the corpus striatum
(significant decreases of 21, 26 and 35% in the low-, mid- and
high-dose groups). There were dose-related, but not always
statistically significant, decreases in all of the neurochemicals
measured in the hypothalamus: noradrenaline, dopamine,
vanillylmandelic acid (VMA), 3,4-dihydroxy-phenylacetic acid
(dopac), homovanillic acid (HVA), serotonin (5-HT) and
5-hydroxyindoleacetic acid (5-HIAA). There were significant
decreases in VMA in the midbrain, corpus striatum and cortex, 5-HT
in the midbrain, corpus striatum and medulla oblongata, and dopac in
the cerebellum in the high-dose group only. There were also
significant decreases in 5-HT and 5-HIAA in the hypothalamus of the
mid- and high-dose groups (Hsieh et al., 1992).
Continuous exposure of rats to 0.012, 0.12 and 5.3 mg
phenol/m3 for 61 days caused a shorter extensor muscle chronaxy
and an increase in whole-blood cholinesterase activities in rats at
concentrations above 0.012 mg phenol/m3 (Mukhitov, 1964).
7.8.2 Myelotoxicity
Because phenol is an important metabolite of benzene, which is
known to exert a toxic effect on bone marrow (including leukaemia)
after metabolic activation, many studies have been performed to
investigate the possible myelotoxic action of phenol.
In an in vitro assay for toxicity to primary murine
haematopoietic cell cultures, phenol showed slight and variable
activity at a concentration of 0.4 mM, but there was marked toxicity
at 2 mM. In comparison, the phenol metabolites catechol and
hydroquinone exhibited marked toxicity at 0.04 mM (Seidel et al.,
1991).
Subcutaneous treatment with phenol (245 mg/kg body weight)
significantly inhibited erythropoiesis in mice 48 h after treatment,
as indicated by a 59Fe uptake assay (Bolcsak & Nerland, 1983).
Intraperitoneal injection of 0-150 mg phenol/kg body weight to
male B6C3F1 mice, twice daily for 12 days, did not result in a
suppression of bone marrow cellularity. However, simultaneous
treatment of mice with 75 mg phenol/kg body weight and 25-75 mg
hydroquinone/kg body weight (another benzene metabolite), produced a
dose-related decrease in bone marrow cellularity, which was much
more pronounced than after treatment with hydroquinone only. The
observed effect closely resembled the myelotoxic effect of benzene
(Eastmond et al., 1987b). Subsequent in vitro studies by these
and other investigators confirmed that phenol (0.01-1 mM) stimulates
further bioactivation of hydroquinone to myelotoxic compounds in
bone-marrow cells (Eastmond et al., 1987b; Subrahmanyam et al.,
1989).
No haematopoietic toxicity was found in rats after daily
subcutaneous injections of 250-750 mg phenol/kg body weight for 1
week. Of the rats receiving 750 mg phenol/kg body weight, 50% died
(Mitchell, 1972).
Six consecutive subcutaneous injections of 50 mg phenol/kg body
weight to mice resulted in a slightly but significantly reduced
number of granulopoietic stem cells and bone marrow cellularity in
the tibia (Tunek et al., 1981).
7.8.3 Immunotoxicology
One immunological study has been reported. Female CD1 mice were
exposed to 19 mg phenol/m3 (5 ppm), either as a single 3-h
exposure or as five daily 3-h exposures. Neither the susceptibility
of the animals to experimentally induced streptococcus aerosol
infection nor their pulmonary bactericidal activity was
significantly affected (Aranyi et al., 1986).
Groups of five male CD-1 mice were supplied with drinking-water
containing 0, 4.7, 19.5 or 95.2 mg phenol/litre for 4 weeks, at the
end of which various haematological and immunological parameters
were measured. The erythrocyte count was statistically significantly
decreased, compared with control values, in all treated groups in a
dose-related manner, but total and differential leucocyte counts
were unaffected. Total spleen cellularity was decreased in a
non-significant dose-related manner. The highest dose suppressed the
stimulation of cultured splenic lymphocytes by the B-cell mitogen
lipopolysaccharide, the T-cell mitogen phytohaemagglutinin, and the
T- and B-cell mitogen pokeweed, but not by concanavatin. The mid and
high doses suppressed the animals' antibody production in response
to a T-cell-dependent antigen, i.e. sheep erythrocytes (Hsieh et
al., 1992).
7.8.4 Biochemical effects
In a study on the biochemistry of intestinal mucosa, mice were
provided with 0,5, 50 or 500 mg phenol/litre drinking-water
(calculated by the authors to be 1, 10 and 100 mg phenol/kg body
weight) for 5 days or 5 months at 1-day intervals. An increase in
glucose-6-phosphatase, succinate dehydrogenase and cytochrome
oxidase activities in the intestinal mucosa was observed in mice
receiving > 0.02 mg phenol/kg body weight for 5 days. A decrease
in these activities was seen at 2 mg phenol/kg body weight. After 5
months administration of 0.02 and 0.2 mg phenol/kg body weight, the
enzyme activity had returned to normal, but the highest dose group
showed a decline (or even a lack) of activity in the cells of the
intestinal mucosa (Olowska et al., 1980).
In another study of biochemical effects, mice were provided for
5 days (killed 24 h after the last application) and 35 days (killed
30 days after the last application) with aqueous solutions of 0.08,
0.8 or 8 mg phenol/litre. The authors of the study calculated the
doses to be approximately 0.016, 0.16 and 1.6 mg phenol/kg body
weight per day, respectively. Only the lowest dose of 0.08 mg
phenol/litre evoked considerable changes in the localization of
glucose-6-phosphatase, 5 days after treatment. Changes in alkaline
phosphatase localization in the kidney were seen at 0.8 and 8 mg
phenol/litre. Full recovery occurred after 30 days (Laszczynska et
al., 1983).
Inhalation exposure of 50 male white rats to 0.4 mg
phenol/m3, 24 h/day, 7 days/week, for 3 months, resulted in some
inhibition of oxidative phosphorylation in the lungs, liver and
kidneys. An increase in the rate of glycolysis was also observed in
the lungs and kidneys (Skvortsova & Vysochina, 1976).
8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Controlled studies
In the Kligman maximization test, phenol did not cause
sensitization in 24 human volunteers (Kligman, 1966).
It was reported by Rea et al. (1987) that, in a group of 134
"chemically sensitive" patients where several volatile organic
chemicals were detected in the blood, 107 (80%) reacted adversely
after a challenge exposure to phenol alone (0.008 mg/m3). The
criteria used to identify "sensitive patients" and "adverse
reactions" were not specified. The toxicological significance of
this finding is not known.
Mukhitov (1964) reported that six 5-min inhalation exposures to
phenol at 0.015 mg/m3 produced an increased sensitivity to light
in each of 3 dark-adapted subjects.
8.1.2 Case reports
Various reports have appeared on the adverse effects of phenol
in individuals or groups of humans after intentional (e.g.,
therapeutic) as well as accidental short-term exposure to phenol.
8.1.2.1 Dermal exposure
The use of phenol as a disinfectant and antiseptic was
introduced by Lister (1867). However, its use has been restricted by
intoxications caused by these applications (Table 10).
Local effects after dermal phenol exposure consisted of
erythema or painless blanching (Dreisbach, 1983), and, in more
severe cases, corrosion (Schmidt & Maibach, 1981) and necrosis. The
use of 5-10% phenol dressings for antiseptic purposes, for example,
has led to many cases of necrosis of the skin and underlying
tissues. When fingers and toes have been involved, amputation has
sometimes been necessary. Due to their high toxicity, these
dressings are no longer used (Cronin & Brauer, 1949; Deichmann,
1949; De Groote & Lambotte, 1960; Abraham, 1972).
Phenol chemical peel is a technique which has been used in
superficial surgery of the skin for the last 30 years (Ersek, 1991).
The phenolic mixture used classically is 3 ml of 50% phenol, 2 ml of
water, 8 drops of soap and 8 drops of croton oil. This is applied to
the skin to reduce pigmentation. Topical use of phenol as a chemical
face-peel has been reported as being associated with cardiac
dysrhythmias in "up to 30% of adults" (Morrison et al., 1991), but
only a single case report has been published (Warner & Harper,
1985). This report concerned a 10-year-old boy who had a solution,
consisting of 40% phenol and 0.8% croton oil in hexachlorophene soap
and water, applied to a large nevus covering 1.9% of his body
surface whilst under anaesthesia (60% nitrous oxide and 3%
halothane) and receiving a total of 200 ml of lactated Ringer's
solution intravenously. After 55 min of treatment, multifocal and
coupled premature ventricular complexes were detected by ECG, but
blood pressure remained stable and plasma sodium and potassium
concentrations were normal. An intravenous infusion of 250 mg
bretylium sulfate suppressed the dysrhythmia and the boy had an
uneventful recovery.
Systemic intoxication can occur very rapidly after absorption
of phenol through the skin (Table 10). Most significantly,
cardiovascular shock (sometimes resulting in death) and severe
metabolic acidosis occur. Truppman & Ellenby (1979) observed cardiac
arrhythmias (supraventricular as well as ventricular) in 10 out of
42 patients within 10 min after the application of approximately 5%
phenol on half of the face for cosmetic treatment. Hyperventilation,
kidney damage and methaemoglobinaemia have also been observed in
several cases of exposure to phenol.
Table 10. Human dermal toxicity of phenol
Concentration Medium Contact duration Circumstances Most severe response References
(%)
100 crystals 30 min in glove grangrene Abraham (1972)
80-100 water 20 min spill on hip, thigh, death Turtle & Dolan
scrotum (1922)
80-100 water 2-4 days closed dressings on 11 persons exposed: Lister (1867)
open wounds 1 death, 8 gas gangrene,
11 tissue necrosis
78 water 2-5 min 4-5 litres spilt on coma Duverneuil &
upper half of body Ravier (1962)
43.5 waste water 1 min spill on lower half of body shock Evans (1952)
5 ointment 7 days closed dressing on cut gangrene Schussler &
Stern (1911)
2 water 2.5 days moist dressing over burns death Cronin & Brauer
on 30% of body surface (1949)
2 water 11 h closed bandage on death Hinkel & Kintzel
infant umbilicus (1968)
Foxall et al. (1991) reported a case of acute renal failure
following an industrial accident in which a man was partially
submerged for a few seconds in a solution of 20% phenol in
dichloromethane. He immediately showered, but was subsequently found
in a state of collapse. His extremities were cold and he had 50%
body burns. He developed nausea and vomiting after taking fluids.
Anuria ensued, with a rise in plasma creatinine, but treatment with
intravenous furosemide and haemodyalisis (daily for seven days, then
with decreasing intervals for a further 18 days), allowed adequate
urinary volumes to be produced. Respiratory distress required
intensive care treatment. Marginal polyuria persisted one year after
the accident.
8.1.2.2 Oral exposure
Cases of oral intoxication have occurred as a result of
accidental and intentional ingestion. Local and systemic effects
have been described in the literature, symptoms being similar to
those following dermal exposure. Case reports have been published
(Model, 1889; Stajduhar-Caric, 1968; Haddad et al., 1979).
Death occurred within 10 min of ingestion of 4.8 g phenol
(Andersen, 1869). However, ingestion of 56.7 g of a phenol-saline
mixture was reported to have occurred without complaints (Leider &
Moser, 1961), and an individual survived the ingestion of 57 g
phenol (88%) after intensive treatment. Symptoms in the latter study
included severe gastrointestinal irritation, as well as the expected
cardiovascular and respiratory effects (Bennett et al., 1950).
A severe accidental phenol spill in Wisconsin in 1974
contaminated ground water which was being used as drinking-water.
Approximately one month later, several people living near the spill
complained of health effects. Six months after the spill, medical
histories were taken from 100 people who had consumed
phenol-contaminated water (the authors estimated the daily exposure
to be 10-240 mg phenol/person). In retrospect, a statistically
significant increase was found in diarrhoea, mouth sores, dark urine
and burning of the mouth, which had persisted for an average of 2
weeks. No significant abnormalities were found 6 months after
initial exposure upon physical examination or laboratory analysis.
Urinary phenol levels were not elevated (Delfino & Dube, 1976; Baker
et al., 1978).
A river in North Wales, United Kingdom, used for the
preparation of drinking-water, was accidentally contaminated with
phenol (Jarvis et al., 1985). When the water was chlorinated,
various chlorophenols appeared to have been formed. A retrospective
postal survey of 344 households that received the contaminated tap
water and 250 control households was carried out. Significantly more
gastrointestinal illnesses, as well as other symptoms, were claimed
in the contaminated areas than in the unexposed areas. Phenol
concentrations in drinking-water were conservatively estimated to
have been 4.7-10.3 µg/litre for some days (Jarvis et al., 1985).
8.1.2.3 Inhalation exposure
Very few cases of adverse effects after short-term phenol
vapour exposure have been reported.
Hospital outbreaks of severe idiopathic neonatal unconjugated
hyperbilirubinaemia have been associated with the phenol-containing
disinfectant used for cleaning the nursery equipment, floors and
walls. When the disinfectant was no longer used, the epidemic
subsided (Daum et al., 1976; Wysowski et al., 1978; Doan et
al., 1979).
Studies of occupational inhalation exposure are described in
section 8.2.
8.1.2.4 Exposure by injection
Phenol has been used as a neuron blocking agent in patients
suffering from spasm following, for example, spinal cord damage or
cerebrovascular stroke (Wood, 1978, review; Nathan, 1959; Cooper et
al., 1965; Khalili & Betts, 1967; Gibson, 1987). It has also been
used to relieve chronic pain (Wood, 1978; Benzon, 1979; Smith,
1984). Treatment involved administering the phenol by intravenous
injection or perfusion, or by direct injection into the spinal cord.
Reported side-effects of phenol therapy were convulsions, transient
paraesthesia, leg weakness, urinary and fecal incontinence, one case
of a severe arterial block in the upper arm requiring amputation,
and one case of acute bronchospasm (Wood, 1978, review; Benzon,
1979; Gibson, 1987; Atkinson & Skupak, 1989). In addition, there
have been reports of phenol-induced cardiac dysrhythmia in adults
(Forrest & Ramage, 1987) and in children, in whom an incidence of
19% was reported (Morrison et al., 1991).
8.2 Occupational exposure
Poisoning due to chronic inhalation of phenol was known 100
years ago, primarily as a disorder in physicians and their helpers,
under the term "carbol marasmus" (Lister, 1867). A classical case of
phenol marasmus was described in a worker employed for 13´ years in
a laboratory boiling phenol solutions. Symptoms were anorexia,
weight loss, headache, vertigo, salivation and dark urine (Merliss,
1972).
A few studies are available concerning occupational exposure of
workers in bakelite factories. Workers were exposed to phenol, and
simultaneous exposure to formaldehyde occurred. Elevated phenol
urine levels, unspecified complaints, and chronic airway obstruction
were observed (Schoenberg & Mitchell, 1975; Knapik et al., 1980).
Twenty-nine cases of poisonings among workers, who, during a
3-year period, quenched coke with a waste-water solution containing
0.3-0.8 g phenol/litre, were attributed to phenol intoxication.
Phenol vapour concentrations in the air ranged from 0.5 to 12.2
mg/m3. The number of workers and the symptoms of intoxication were
not specified. The author did not consider the potential of dermal
absorption (Petrov, 1960).
A case-control study was carried out on 57 cases among 3805
workers from the Finnish wood industry (particle board, plywood,
sawmill or formaldehyde glue) suffering from respiratory cancer. The
inhalatory exposure level to phenol and frequency of multiple
exposure to pesticides were found to be significantly higher for
cancer cases, but the exposure to wood dust was not significantly
different between cases and controls (Kauppinen et al., 1986). The
number of cases in this study was small, and confounding exposures
were inadequately controlled.
In a case-control study of 6678 rubber workers, employed in
areas where phenol was used, exposure to phenol was not associated
with increased risks of cancer of the respiratory tract, stomach or
prostate or of lymphosarcoma or lymphatic leukaemia (Wilcosky et
al., 1984).
A mortality study was conducted among 14 861 white male workers
engaged in the production or use of phenol and formaldehyde in five
companies within the USA. The follow-up comprised more than 360 000
man-years. Mortality rates from all causes combined were similar to
those in the general population of the USA. Excesses of cancer of
the oesophagus, cancer of the kidney and Hodgkin's disease were
observed among the workers exposed to phenol, but these did not show
any exposure-response relationship and were not statistically
significant. Reduced mortality ratios were observed for cancer of
the buccal cavity and pharynx, cancer of the stomach, cancer of the
brain, arteriosclerotic heart disease, emphysema, disease of the
digestive system and cirrhosis of the liver, although these
reductions were not statistically significant. For arteriosclerotic
heart disease, emphysema and cirrhosis of the liver, there were
inverse relationships between mortality rates and duration of phenol
exposure and cumulative phenol exposure levels (Dosemeci et al.,
1991).
A cardiovascular disease (CVD) mortality study was conducted
among 1282 white male production workers in a large rubber- and
tyre-manufacturing plant. Exposure estimates for 25 solvents were
available (concentrations were not measured). The CVD mortality
during 15-year follow-up period was analyzed in exposed and not
exposed workers. The known association between CS2 exposures and
ischemic heart diseases (IHD) was confirmed, and two other solvents,
ethanol and phenol, were also found to be predictors of IHD. Phenol
showed the strongest association with CVD mortality. However, some
confounders (cigarette smoking, hypertension and high serum
cholesterol) were not controlled and unrecognized chemical
atherogens could also, according to the authors, influence the
results (Wilcosky & Tyroler, 1983).
8.3 Organoleptic data
The odour threshold for phenol has been reported to range from
0.021 to 20 mg/m3 (Van Gemert & Nettenbreijer, 1977; Van Gemert,
1984).
The geometric mean of 16 air odour thresholds and 6 water odour
thresholds for phenol was reported by Amoore & Hautala (1983) to be
0.16 mg/m3 (0.040 ppm, with a standard error of 0.026 ppm). In
this calculation, the original literature was reviewed and values
which diverged more than 100-fold from the nearest of two or more
other thresholds were eliminated. Both detection and recognition
values were included. The water detection threshold for phenol,
based upon multiplying the calculated air odour threshold by the
water-air distribution ratio, was reported by the same authors to be
7.9 mg/litre.
A taste threshold value of 0.3 mg/litre in water has been
reported (US EPA, 1992).
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
The toxicity of phenol has been studied in microorganisms
(e.g., bacteria, fungi, algae and protozoa) and numerous aquatic
invertebrates and vertebrates (Buikema et al., 1979). Because of
this vast amount of data, a selection has been made, based on the
reliability of the data and the relevance of the test organisms.
Details of acute and long-term aquatic toxicity studies, considered
to be adequately performed and reported, are included in Tables 11
and 12. Less adequate studies are reported in the text only.
9.1 Microorganisms
Reliable phenol toxicity data for microorganisms are given in
Table 11.
In microorganisms, growth inhibition is usually observed after
phenol exposure. In studies on single bacterial species, the EC50
values (EC50 = calculated concentration affecting 50% of test
population) found for growth inhibition varied from 244 mg
phenol/litre in a newly developed, 6-h test with Pseudomonas putida
(Slabbert, 1986) to 1600 mg phenol/litre after 18 h of exposure in a
more conventional test with Aeromonas hydrophila (Dutka & Kwann,
1981). Bringmann & Kühn (1977) reported a toxicity threshold of 64
mg/litre after 16 h. EC50 values for reduced photoluminescence in
Photobacterium phosphoreum of 28-34 mg phenol/litre (Dutka et
al., 1983) and 40 mg phenol/litre (Curtis et al., 1982) have
been reported. In activated sludge, the EC50 for a reduced oxygen
uptake was reported to be 520-1500 mg phenol/litre, whereas a lower
value was found for substrate consumption inhibition (104 mg
phenol/litre) (Miksch & Schürmann, 1988; Volskay & Grady, 1988). The
lowest reported concentration affecting activated sludge was 10 mg
phenol/litre; 1 mg phenol/litre had no effect (Baird et al.,
1974).
Reported toxicity thresholds for protozoa were of the same
order of magnitude as for bacteria: 33-144 mg phenol/litre
(Bringmann & Kühn, 1959, 1980; Dive & LeClerq, 1977; Bringmann et
al., 1980). For algae, values were somewhat lower, but were
observed after a longer exposure period: 6 mg phenol/litre for
cyanobacteria (blue-green algae) and 8 mg phenol/litre for green
algae, after 7-8 days of exposure (Bringmann & Kühn, 1978, 1980).
The IC50 values (concentration causing 50% growth inhibition)
reported for various fungi by Kwasniewska & Kaiser (1983) were of
the same order of magnitude as the above EC50 values for bacterial
growth inhibition: 460-1000 mg phenol/litre. These values are also
within the range of concentrations observed by Babich & Stotzky
(1985) to cause initial or complete growth inhibition in various
fungi (100-1000 mg phenol/litre and 750->1000 mg phenol/litre,
respectively).
Table 11. Acute aquatic toxicity of phenol
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Freshwater Organisms
Bacteria
Photobacterium 15 6.5-6.7 S 5, 10, EC50c 28, 32, 34 Dutka et al.
phosphoreum 15 min (1983)
Pseudomonas putida 25 7.0 80.1 S 16 h TTc 64 Bringmann &
Kuhn (1977)
27 7.2 S 6 h EC50 244 Slabbert
(1986)
Cyanobacteria
Microcystis aeruginosa 27 7.0 72.3 S 8 days TT 6 Bringmann &
Kuhn (1978)
Green algae
Scenedesmus 27 7.0 72.3 S 7 days TT 8 Bringmann &
quadricauda Kuhn (1980)
Protozoa
Chilomonas 20 6.9 S 48 h TT 65 Bringmann et
paramaecium al. (1980)
Colpidium campylum 20 S 43 h TTd 100 Dive & LeClerq
(1977)
Entosiphon sulcatum 25 6.9 80.1 S 72 h TT 33 Bringmann &
Kuhn (1980)
Microregma heterostoma 27 7.5-7.8 213.6 S 28 h TT 30e Bringmann &
Kuhn (1959)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Uronema parduczi 25 7.3 S 20 h TT 144 Bringmann &
Kuhn (1980)
Crustacea
Asellus aquaticus 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 230, 200, Green et al.
96 h 180f (1985)
Cypris subglobosa 20.4 7.9 8.4 204 S 12, 24, 48, LC50 173, 167, Rao et al.
72, 96 h 137, 122, 72 (1983)
Daphnia magna 19.8-20.9 7.7-8.3 157 ± 4 S 48 h LC50 13 Gersich et
al. (1986)
19 ± 1 8.2 ± 0.3 199.4 S 48 h LC50 100f Hermens (1984)
19 ± 1 8.2 ± 0.3 199.4 S 48 h EC50g 9f Hermens (1984)
17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 24 h LC50h 13 Holcombe et
al. (1987)
S 48 h EC50g 7 Keen & Baillod
(1985)
22 ± 1 6.8-7.8 7.6 ± 0.2 146 ± 15 S 48 h LC50 8 Lewis (1983)
Gammarus pulex 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 106, 85, Green et al.
96 h 69f (1985)
7 ± 1 8.3 10.9 250 R 24, 48, LC50 100, 89, Stephenson
72, 96 h 67, 51 (1983)
Ceriodaphnia dubia 25 ± 1 8.18 ± 0.04 57.1 ± 4.1 S 48 h LC50 3.1 Oris et al.
(1991)
Mollusca
Indoplanorbis S 12, 24, 48, LC50 265, 215, 200, Agrawal (1987)
exustus 72, 96 h 156, 126
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Lymnaea acuminata 20 ± 2 7.9 ± 0.2 5.5 ± 1.5 190-223 R 12, 24, 48, LC50 270, 219, 205, Gupta & Rao
72, 96 h 158, 129 (1982)
Worms
Limnodrilus 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 960, 870, Green et al.
hoffmeisteri 96 h 780f (1985)
Polycelis felina 18 7-8.5 -i** 300-500 S 96 h LC50 64f Erben et al.
(1983)
Polycelis tenuis 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 230, 200, Green et al.
96 h 88f (1985)
Fish
Brachydanio rerio 25 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 31, 29f Fogels &
Sprague (1977)
24 ± 1 > 6 64 R 6, 12, 24, LC50 35, 31, 28, Razani et al.
(12 h) 48, 72, 96 h 26, 25, 25f (1986a)
Campostoma anomalum 23 -i R 48 h LC50 18f Chagnon &
(24 h) Hlohowskyj
(1989)
Catostomus 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 11 Holcombe et
commersoni al. (1987)
Jordanella floridae 25 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 36, 36f Fogels &
Sprague (1977)
Lebistes reticulatus 28-31.8 7.8-8.2 5.7-7.2 218-239 R 12, 24, 48, LC50 103, 83, 64, Gupta et al.
(24 h) 72, 96 h 50, 48 (1982a)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Lepomis macrochirus 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 17 Holcombe et
al. (1987)
Leuciscus idus 20 S 48 h LC50 14, 25 Jünke &
melanotus Lüdemann
(1978)
Notopterus notopterus 23-26.5 6.8-7.6 5.9-7.8 60-70 S 24, 48, 72, LC50 14, 14, 13, Verma et al.
96 h 13 (1980)
Pimephales promelas 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 25 Holcombe et
al. (1987)
25 ± 2 6.2-8.2 43-49 CF 96, 192 h LC50 29, 23f Phipps et al.
(1981)
Rasbora 20 7.2 250 S 24, 48 h LC50 8, 7 Alabaster
heteromorpha (1969)
Rutilus rutilus 10.3 ± 0.3 7.8 ± 0.02 257-260 S 48 h LC50 10f Solbé et al.
(1985)
Salmo gairdneri 15 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 12 12f Fogels &
Sprague (1977)
17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50i 11 Holcombe et
al. (1987)
Insects
Baetis rhodani 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 19, 19, 16f Green et al.
96 h (1985)
Chironomus riparius 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 1050, 500, Green et al.
96 h 240f (1985)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Hydropsyche 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 940, 720, Green et al.
angustipennis 96 h 260f (1985)
Marine Organisms
Crustacea
Artemia salina S 24, 48 h LC50j 157, 56 Price et al.
(1974)
Canthocamptus synthetic medium according to Cairns S 48 h LC50j 9 Rao & Nath
(1983)
Gammarus duebeni 5 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 183f Oksama &
Kristofferson
16 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 89f (1979)
Mesidotea entomon 5 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 176f Oksama &
Kristofferson
(1979)
10 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 186f
Panopeus herbstii 25 25 pptk R 96 h LC50 53 Key & Scott
24 h (1986)
Mollusca
Crassostrea 24 ± 1 seak R 48 h (eggs) LC50 58 Davis & Hidu
virginica (1969)
Mercenaria 24 ± 1 seak R 48 h (eggs)- LC50j 53-55 Davis & Hidu
mercenaria 12 days (1969)
(larvae)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Worms
Ophryotrocha 21 seak S 48 h LC50 100-330 Parker (1984)
diadema
Fish
Phoxinus phoxinus 5 ± 0.7 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 10 Oksama &
Kristofferson
(1979)
a S = static test; CF = continuous flow test; R = renewal test (semi-static test)
b EC50 = median effect concentration = calculated concentration causing effect in 50% of population; LC50 = median lethal concentration
TT = toxicity threshold, i.e. concentration affecting growth in > 3% of population
c effect: reduction in photoluminescence
d minimal concentration affecting growth
e river water
f concentration of test compound analysed during assay
g immobility
h simultaneous testing of 8 species
i no data; aerated
j reported was TLm, or median toxicity limit
k salinity; sea = sea water
Table 12. Long-term aquatic toxicity of phenol
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
Freshwater Organisms
Crustacea
Daphnia 19 ± 1 8.2 199 R 16 days NOEC (growth) 0.16 De Neer et
magna (48 h) al. (1988)
19 ± 1 8.2 ± 0.3 199 R 21 days NOLC 10c Hermens
NOEC (repro) 3.2c (1984)
NOEC (growth) 3.2c
Ceriodaphnia 25 ± 1 8.18 ± 0.04 57.1 ± 4.1 R 4 days NOEC (repro) 4c Oris et al.
dubia (48 h) 7 days NOEC (survival)d 4c (1991)
Fish
Brachydanyo 24 ± 1 6.1-6.5 6.4-8.5 57-61 R 3 months (adults) NOLC 4.9 Razani et
rerio (24 h) al. (1986b)
NOEC (spawning) < 2.2
R 2 months (test started NOEC (growth) 2.2
(48 h) with eggs from adults NOLC < 2.2
exposed to same
concentrations; all
eggs hatched after
2-3 days)
Table 12 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
Carassius 19-24 7.78 6.2-9.0 197.5 CF 8 days after hatching LC1 (at hatch) 0.002c Birge et
auratus (test was started with al. (1979)
eggs (1-2 h after
spawning) which
hatched completely
within 3.5 days)
Pimephales 25 ± 1 8.0 5.3 725.3 CF 30 days after hatching NOEC (hatching) 83.2c De Graeve
promelas et al.
NOEC (growth) 0.75c (1980)
Pimephales 25 ± 1 8.0 5.3 725.3 CF NOLC 6.1c,e
promelas 25 ± 2 7.2-7.9 7.7 46.0 CF 38 days after hatching NOLC 3.57c,f Holcombe
(test was started with NOEC (growth) 1.83c et al.
eggs within 24 h after NOEC (hatching) 3.57c,f (1982)
spawning)
Salmo 12-14 7.78 6.2-9.0 197.5 CF 8 days after hatching LC1 (at hatch) 0.0002c Birge et
gairdneri (test was started with al. (1979)
eggs (20 min after
fertilization) which
hatched completely
within 22 days)
Table 12 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
13.3-14.2 7.4-8.1 8.6-10.2 100 CF 4 days after hatching NOLC 0.009c,e Black et
(test was started with al. (1983)
eggs (20 min after NOEC (hatching) 0.009c,f
fertilization) which
hatched within
23 days)
7.8 5.7 579.9 CF 58 days after hatching NOEC (growth) 0.1c,g De Graeve
(test was started with et al.
eyed eggs which hatched (1980)
completely within 48 h)
a S = static test; CF = continuous flow test; R = renewal test (semi-static)
b NOEC = no-observed-effect concentration = highest tested concentration without observed effect; NOLC = no-observed-lethal concentration.
An LC1 is a calculated value which is, to some extent, comparable to the observed NOLC value used in other studies.
c phenol concentration analysed during test
d survival was a more sensitive end-point than reproduction
e calculated from results by Task Group
f highest concentration tested
g extrapolated by authors
An increase in salinity (0-30%) increased the toxicity of
phenol to fungi (Babich & Stotzky, 1985).
9.2 Aquatic organisms
9.2.1 Freshwater organisms
9.2.1.1 Short-term studies
The most important sublethal acute effects observed in
freshwater species after phenol exposure were a reduced heart rate
and damage to the epithelium of gills (with loss of function),
liver, kidneys, intestines and blood vessels. One study reported the
occurrence of severe seizures, mediated by the central nervous
system, in Salmo gairdneri after exposure to sublethal phenol
concentrations (Bradbury et al., 1989). In invertebrates, growth
inhibition was usually observed. Some EC50 values for the latter
organisms are given in Table 11.
Most toxicity studies concentrated on lethal effects. Death was
usually preceded by immobility, loss of equilibrium, paralysis and
respiratory distress (Razani et al., 1986a; Tonapi & Varghese,
1987; Green et al., 1988; Chagnon & Hlohowskyj, 1989). Toxicity
testing, where the same species was used by different research
workers in different waters, resulted in LC50 values (LC50 :
calculated concentration causing death in 50% of test group) that
varied widely, as can be seen from Table 11.
Environmental factors may affect the toxicity of phenol (Brown
et al., 1967; Miller & Ogilvie, 1975; Ruessink & Smith, 1975;
Cairns et al., 1976; Reynolds et al., 1978; Birge et al.,
1979; Dalela et al., 1980; Gluth & Hanke, 1983; Gupta et al.,
1983a,b; Stephenson, 1983). Hardness and pH, however, do not appear
to have a large impact on phenol toxicity. The toxicity for various
fungi and fish species, for example, did not change significantly
over the pH range of 5-8; toxicity for fungi and some fish species
was not influenced at all by hardness, whereas phenol was slightly
more toxic in soft than in hard waters for the carp (Herbert, 1962;
Pickering & Henderson, 1966; EIFAC, 1972; Babich & Stotzky, 1985).
The effect of temperature appeared to be variable (Cairns et al.,
1978; Babich & Stotzky, 1985). Since temperature influences both the
uptake and the detoxification (conjugation) of phenol (Green et
al., 1988), phenol toxicity could be enhanced, as well as
diminished, by increasing temperature, depending on which parameter
was influenced most.
Several biological factors also influence the response of the
biota to phenol, e.g., strain type, nutritional status, size,
embryonal or developmental stage, crowding and physiological
adaptation (Dowden & Benett, 1965; Alexander & Clarke, 1978; Birge
et al., 1979; Flerov, 1979; De Graeve et al., 1980; Kordylewska,
1980; Gupta et al., 1982b; Mayes et al., 1982; Black et al.,
1983; Lewis, 1983).
Comparison of 48-h LC50 values from Table 11 shows that, in
general, fish are the most sensitive freshwater species with respect
to phenol toxicity. The 48-h LC50 values for some selected fish
species ranged from 7 to 64 mg/litre. For crustaceans, this range
was 3.1-200, and for molluscs it was 200-205 mg/litre; for insects,
it was 19-720 mg/litre and for worms 200-870 mg/litre. Upon
simultaneous testing of eight species at concentrations up to 51 mg
phenol/litre, no toxicity was observed for larvae of the amphibian
Xenopus laevis, for the snail Aplexa hypnorum or the insect
Tanytarsus dissimilis. Where toxicity was observed, LC50 values
were included in Table 11 (Holcombe et al., 1987). The data
presented in Table 11 are in good agreement with the order of
increasing tolerance to phenol proposed by Alekseyev & Antipin
(1976): fish-crustaceans-tolerant insects-worms-molluscs-highly
tolerant insects.
9.2.1.2 Long-term studies
Most long-term studies with freshwater species have concerned
growth, reproduction and/or mortality; these studies are discussed
below. Studies considered to be adequately performed and reported
are included in Table 12.
A few long-term studies with freshwater fish have been designed
to detect sublethal effects of phenol exposure. Increased
proteolysis as a result of stress, mild kidney damage, and an
inhibitory effect on the development and maturation of the ovary,
secondary to a liver dysfunction, were some of the effects reported
(Dangé, 1986; Gupta & Dalela, 1987; Kumar & Mukerjee, 1988).
In a life-cycle test using Daphnia magna, the maximum
acceptable tolerance concentration (MATC) proved to be 1.5-6.3 mg
phenol/litre (US EPA, 1980). These results are comparable with the
no-observed-effect concentration (NOEC) values for growth and
reproduction (both 3.2 mg phenol/litre) found by Hermens (1984) and
Oris et al. (1991). De Neer et al. (1988), however, found a
considerably lower NOEC value of 0.16 mg phenol/litre for growth of
Daphnia magna under similar experimental conditions.
Exposure of adult Brachydanio rerio to 2.2, 4.9 or 24 mg
phenol/litre for 3 months resulted in 67% mortality at the highest
concentration; the no-observed-lethal concentration (NOLC) was 4.9
mg phenol/litre. At 24 mg phenol/litre, only immature oocytes were
found in surviving fish; at the two lower concentrations both
immature and mature oocytes were observed, whereas spawning was
delayed. In subsequent embryo-larval tests, starting with the eggs
of exposed adults, mortality appeared to be maximal during embryonic
development and the initial larval stage. All larvae died within 12
days at 4.9 mg phenol/litre. At 2.2 mg phenol/litre, larval
mortality was still slightly increased, but surviving animals showed
normal growth and development (Razani et al., 1986b).
In two embryo-larval bioassays on Pimephales promelas, growth
proved to be the most sensitive criterion: the NOEC values were 0.75
and 1.83 mg phenol/litre (De Graeve et al., 1980; Holcombe et
al., 1982).
The results of the embryo-larval test on Salmo gairdneri
given by Birge et al. (1979) (LC1:0.2 µg/litre) and Black et
al. (1983) (NOEC:9 µg/litre) are much lower than those obtained by
De Graeve et al. (1980) (NOECgrowth: 0.1 mg/litre), probably
because the latter test was started with eyed eggs, whereas the
former two tests were started with just-fertilized eggs.
In addition, Birge et al. (1979) studied the influence of
phenol in an embryo-larval test on Carassius auratus and found a
LC1 of 2.0 µg phenol/litre.
Dumpert (1987) reported a NOLC value of 10 mg phenol/litre for
larvae of the amphibian Xenopus laevis; larval mortality was 100%
within 3 weeks at 50 mg phenol/litre. Larval growth was slightly,
but not significantly, retarded at 5 and 10 mg phenol/litre.
Hatching was normal at all tested concentrations. However, the
results may not be reliable because test solutions were renewed only
once a week, whereas aeration may also have contributed to
undetected loss of phenol. In other embryo-larval bioassays on five
amphibian species, Rana ripiens and Rana catesbeiana were the
least tolerant. These species exhibited about equal sensitivity to
phenol (LC1: 1.0 and 1.1 mg phenol/litre, respectively; LC10:
5.2 and 8.5 mg phenol/litre, respectively (Birge et al., 1980).
9.2.2 Marine organisms
9.2.2.1 Short-term studies
In acute toxicity studies on some marine organisms
(crustaceans, worms, snails and fish), the LC50 values ranged from
8.8-330 mg phenol/litre (see Table 11). In general, the
sensitivities of marine and freshwater organisms for phenol were
similar.
At a sublethal phenol concentration, activities of some enzymes
appeared to be decreased in the brain, liver and muscle tissue of
Sarotherodon mossambicus; this effect was independent of salinity
(Ravichandran & Anantharaj, 1984).
9.2.2.2 Long-term studies
No adequate data are available on long-term toxicity to marine
organisms.
9.2.3 Accumulation
The bioconcentration factor of phenol may be calculated, using
a log Pow value of 1.46 (pH not stated, see Table 1) and the
formula log BCF = 0.79 log Pow -0.40 (Veith & Kosian, 1983). This
yields a bioconcentration factor of 5.7, which is very low and does
not indicate any potential for bioaccumulation.
The experimental bioconcentration factors, reported by Hardy
et al. (1985) for algae, by Erben (1983) for flatworms, by Erben
(1982) for snails, by Key & Scott (1986) for crabs, and by Call et
al. (1980) and Kobayashi & Akitake (1975) for fish, are in
agreement with the calculated value. Other studies, however,
reported higher values. The bioconcentration factor for Daphnia
magna, as assessed from 14C measurements, was reported to be
1375 after 24 h, with an estimated half-life upon depuration of 8 h.
A lower bioconcentration factor (277) was calculated from uptake and
depuration rate constants (Dauble et al., 1986). The
bioconcentration factors for phenol, as determined by 14C
measurements in a 5-day experiment with activated sludge, in a 24
h-experiment with the alga Chlorella fusca, and a 3-day experiment
with the fish Leuciscus idus melanotus, were 2200, 200 and 20,
respectively (Freitag et al., 1985).
Uptake was usually complete (the equilibrium level reached)
within 1-2 days. Initially, excretion was also rapid, but it usually
slowed down after some hours, and depuration was reported to be
incomplete. The amount of unchanged phenol still present after 24 h
in the alga Scenedesmus quadricauda, for example, was 22% (Hardy
et al., 1985), whereas radioactivity from 14C showed a retention
of 80% after 96 h of depuration in the mud crab Panopeus herbstii
(Key & Scott, 1986).
Based on reported and estimated bioconcentration factors for
aquatic organisms, phenol is not expected to bioaccumulate
significantly.
9.2.4 Metabolism
The metabolism of phenol in fish yields the known phenyl
conjugates (phenyl sulfate and phenyl glucuronide) and quinol
sulfate (Kobayashi et al., 1976; Layiwola & Linnecar, 1981; Nagel,
1983; Nagel & Urich, 1983; Kasokat et al., 1987). In the excreta
of the frog Rana temporaria, the same metabolites were found after
phenol injection, together with catechol, catechol sulfate and
traces of resorcinol. Xenopus laevis, another amphibian, appeared
to be unable to glucuronidate phenol, but compensated this by
increasing the production of other metabolites compared to Rana
temporaria (more quinol sulfate and phenyl sulfate, and, in
addition, resorcinol sulfate) (Beyer & Frank, 1985; Görge et al.,
1987).
Phenol metabolism may be induced by prior exposure to phenol or
phenol derivatives, as was observed for sulfate conjugation in the
clam Ruditapes phillippinarum (Kobayashi et al., 1987).
9.3 Terrestrial organisms
Phenol may be taken up by, and stored in, the cuticle membranes
of various plants, such as tomatoes and green pepper fruits, and
rubber leaves (Shafer & Schönherr, 1985). Labelled phenol was
demonstrated to be taken up by soybean roots. The label stayed in
the roots, and was not transported to the shoot, which was
attributed to the metabolism of phenol by the plant into immobile
compounds (McFarlane et al., 1987). Millet seeds appeared to be
more sensitive to the toxicity of phenolic compounds than lettuce
and cucumber seeds. The 120-h EC50 value for root elongation
inhibition in millet was determined to be 120-170 mg phenol/litre
(Wang, 1986).
The toxicity of phenols for four earthworm species (Neuhauser
et al., 1986) was compared with those of other chemicals using two
standardized tests developed by the EEC, the 2-day contact test and
the 14-day artificial soil test. Of ten classes of chemicals, phenol
was the most toxic in the contact test, with LC50 values of
2.4-10.6 µg/cm2, regardless of species. A lower relative toxicity
was reported by the same authors using the artificial soil test.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
The main way in which the general population can be exposed, on
a long-term basis, to phenol present in ambient air is as a result
of industrial emissions and various combustion processes. Other
inhalation sources include the decomposition of organic materials,
liquid manure, and the atmospheric degradation of benzene.
Inhalation and dermal exposure may arise from contact with
contaminated water or consumer products containing phenol. Indirect
exposure of man through the food chain is not likely to add
significantly to long-term inhalation exposure, in view of the short
life-time of the compound in the environment (section 10.2.1.).
Individuals may ingest phenol via drinking-water from contaminated
surface water or ground water. Repeated oral exposure may arise from
the consumption of smoked food items. Endogenous production of
phenol may be influenced by the diet and exposure to certain drugs
and other xenobiotics.
The exposure data available are inadequate to determine the
degree of exposure of the general population or of specific groups
at risk, including workers.
An upper-limit estimate of the daily intake can be made for
long-term exposure of the general population. In this hypothetical
case, it is assumed that an individual will be maximally exposed to
phenol through continuous inhalation of air from a heavily
industrialized area, with frequent consumption of smoked food items
with a high phenol content, and of drinking-water containing phenol
up to the taste threshold. The estimate is summarized in the table
below:
Source Quantity of source Phenol Phenol intake
concentration
Air 20 m3/day 200 µg/m3 4 mg/day
Smoked food 200 g/week 70 mg/kg 2 mg/day
items
Drinking-water 2 litres/day 300 µg/litre 0.6 mg/day
Assuming an average body weight of 70 kg, the total daily
intake of this maximally exposed individual will be 0.1 mg/kg body
weight per day. The daily intake by the general population can be
expected to be much less than this figure.
10.1.2 Toxicity
Phenol has moderately acute toxicity for animals. The oral
LD50 for various animal species range from 300 to 600 mg phenol/kg
body weight, and the LC50 for rats by inhalation is more than 900
mg phenol/m3.
In humans, the lowest acutely lethal oral dose was reported to
be 4.8 g, which is approximately 70 mg/kg body weight. Local, as
well as systemic, effects have been reported in humans, consisting
of irritation, necrosis, cardiovascular effects, metabolic acidosis,
neurological effects and methaemoglobinaemia. Several fatal cases
have been reported after oral or dermal intoxication. No documented
cases of death by inhalation of phenol have been found.
Solutions of phenol are corrosive to the skin and eyes. Phenol
vapours can irritate the respiratory tract. Phenol is not a skin
sensitiser in guinea-pigs or humans.
The most important effects reported in short-term animal
studies were neurotoxicity, liver and kidney damage, respiratory
effects and growth retardation. Toxic effects in rat kidney have
been reported to occur at oral dose levels of 40 mg/kg per day or
more. Liver toxicity was evident in rats administered at least 100
mg/kg per day. In a limited 14-day study on rats, an oral NOAEL of
12 mg/kg per day was reported based on kidney effects. In this
experiment, miosis (an iris response to light) was inhibited at 4
mg/kg per day (the lowest dose tested). However, the health
significance of this finding is not clear. Some biochemical changes
have been reported to occur in the intestinal mucosa and kidneys of
mice at dose levels below 1 mg/kg per day. The toxicological value
of these insufficiently reported biochemical observations is not
known.
There have been no long-term general toxicity studies in
animals or adequate epidemiological studies.
No adequate studies on the reproductive toxicity of phenol have
been reported. Phenol has been identified as a developmental
toxicant in studies with rats and mice. In two multiple-dose rat
studies, NOAELs of 40 mg/kg per day (the LOAEL was 53 mg/kg per day)
and 60 mg/kg per day (the LOAEL was 120 mg/kg per day) have been
reported. For the mouse, the NOAEL was 140 mg/kg per day (the LOAEL
was 280 mg/kg per day).
There is some evidence that phenol is genotoxic to mammalian
cells in vitro. Based on the induction of bone marrow micronuclei
in several studies with mice, phenol may have genotoxic potential.
Oral (drinking-water) animal carcinogenicity bioassays did not
give evidence of a carcinogenic potential of phenol. No animal
inhalation or adequate dermal carcinogenicity studies are available.
Two-stage carcinogenicity studies with mice showed that phenol
applied to the skin does have tumour-promoting activity. Adequate
human data on carcinogenicity are not available.
10.1.3 Evaluation
Accidental high exposure to phenol may cause severe local
effects, systemic intoxications and even death. The available data
do not suggest a strong potential for cumulative health effects from
chronic exposure.
The lowest NOAELs identified are for kidney and developmental
effects, and in rats are in the range of 12-40 mg/kg body weight per
day. The Task Group decided to derive a tolerable daily intake
(TDI), taking into consideration this range. An uncertainty factor
of 200 (including factors of 10 for interspecies variation, 10 for
intraspecies variation, and 2 to account for the limited data base
on the toxicity of phenol in animals) was considered appropriate. A
range of 60-200 µg/kg per day was recommended as the upper limit of
the TDI by the Task Group. As the Task Group's upper-limit estimate
of human daily intake is 100 µg/kg body weight per day (section
10.1.1), it can be concluded that the average general population
exposure to phenol from all sources will be well below this range.
There remain, however, two reasons for concern. The available
data suggest that phenol may be genotoxic, and there is insufficient
data to discount the possibility that phenol is carcinogenic. For
these reasons, it is particularly important that this evaluation of
phenol be kept under periodic review.
10.2 Evaluation of effects on the environment
10.2.1 Environmental levels
Once released into the environment, intercompartmental
transport of phenol may occur by wet deposition from air to sea
water and surface water and soil, and, as the compound can be
expected to be highly mobile in soil, by leaching through soil.
Evaporation will be slow from water and can only be expected
following contamination of relatively dry soil.
Phenol, however, is generally not likely to persist in either
air, sea or surface water, soil or sewage. It readily reacts
photochemically and is rapidly biodegraded aerobically, mainly to
carbon dioxide. Anaerobic degradation to carbon dioxide or methane
also occur. Half-lives will be in the range of several hours for
photodegradation and in the range of hours to days for aerobic
biodegradation. Anaerobic biodegradation also occurs, albeit at a
slower rate. Low removal rates of phenol in ground water and soil
may occur, e.g., following spills, with subsequent inhibition of the
microbial populations.
The scarce environmental exposure data available give some
support for the above conclusions:
* reported ambient air levels are low (< 8 µg/m3 for urban
areas; < 200 µg/m3 for heavily industrialized areas);
* phenol has been detected in rain water;
* reported surface water levels are low (< 24 µg/litre);
* levels in ground water have only been found at highly
contaminated sites.
10.2.2 Toxicity
Based on reported and estimated bioconcentration factors for
aquatic organism, phenol is not expected to bioaccumulate
significantly. The data base on aquatic toxicity is considered
adequate for evaluation. Phenol is toxic to aquatic organisms: the
lowest EC50 for water organisms is estimated to be 3.1 mg/litre
(48-h LC50 for Ceriodaphnia dubia). The lowest chronic NOEC is
estimated to be 0.2 µg/litre (8-day LC1 for Salmo gairdneri).
Applying the modified US EPA method, an Environmental Concern Level
of 0.02 µg/litre can be derived for water. In general, fish are the
most sensitive species and the sensitivities of marine and
freshwater organisms are similar. Adequate data on plants and
terrestrial organisms are not available.
10.2.3 Evaluation
The scarce exposure data available do not allow any firm
conclusions with regard to the degree of risk from phenol to either
aquatic or terrestrial ecosystems. However, in view of the derived
Environmental Concern Level of phenol for aquatic organisms, it is
reasonable to assume that these organisms may be at risk in any
surface or sea water subject to phenol contamination, in spite of
the rapid degradation of this compound.
11. FURTHER RESEARCH
There is a need for the following items:
a) further investigation of the in vivo genotoxicity of phenol;
b) more animal toxicology studies, including 90-day oral and
inhalation studies, carcinogenicity bio-assays by the
inhalation route, and neurotoxicity and multigeneration
reproductive toxicity studies (including evaluation in
offspring);
c) assessment of environmental and occupational exposures and
evaluation of health effects in occupational populations;
d) further evaluation of the dose-duration-effect relationships,
reversibility/persistence and health significance of the
reported phenol-induced inhibition of the pupillary response to
light;
e) further data on the toxicity of phenol for plants and
terrestrial organisms.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic risk of phenol was evaluated in 1989 by the
International Agency for Research on Cancer (IARC, 1989). The
summary evaluation from the IARC Monograph is reproduced here.
"Exposures
Phenol is a basic feedstock for the production of phenolic
resins, bisphenol A, caprolactam, chlorophenols and several
alkylphenols and xylenols. Phenol is also used in disinfectants and
antiseptics. Occupational exposure to phenol has been reported
during its production and use, as well as in the use of phenolic
resins in the wood products industry. It has also been detected in
automotive exhaust and tobacco smoke.
Experimental carcinogenicity data
Phenol was tested for carcinogenicity by oral administration in
drinking-water in one strain of mice and one strain of rats. No
treatment-related increase in the incidence of tumours was observed
in mice or in female rats. In male rats, an increase in the
incidence of leukaemia was observed at the lower dose, but not at
the higher dose. Phenol was tested extensively in the two-stage
mouse skin model and showed promoting activity.
Human carcinogenicity data
In one case-control study of workers in various wood
industries, an increased risk was seen for tumours of the mouth and
respiratory tract in association with exposure to phenol; however,
the number of cases was small and confounding exposures were
inadequately controlled.
Other relevant data
In humans, phenol poisoning can occur after skin absorption,
inhalation of vapours or ingestion. Acute local effects are severe
tissue irritation and necrosis. At high doses, the most prominent
systemic effect is central nervous system depression. Phenol causes
irritation, dermatitis, central nervous system effects and liver and
kidney toxicity in experimental animals.
Phenol induced micronuclei in female mice and sister chromatid
exchange in cultured human cells. It did not inhibit intercellular
communication in cultured animal cells. It induced mutation but not
DNA damage in cultured animal cells. It did not induce recessive
lethal mutation in Drosophila. It had a weak effect in inducing
mitotic segregation in Aspergillus nidulans. Phenol did not induce
mutation in bacteria.
Evaluation
There is inadequate evidence for the carcinogenicity of phenol
in humans.
There is inadequate evidence for the carcinogenicity of phenol
in experimental animals.
Overall evaluation
Phenol is not classifiable as to its carcinogenicity to humans
(Group 3)".
REFERENCES
Abou-el-Makarem MM, Millburn P, Smith RL, & Williams RT (1967)
Biliary excretion of foreign compounds. Biochem J, 105: 1269-1274.
Abraham AJ (1972) A case of carbolic acid gangrene of the thumb. Br
J Plast Surg, 25: 282-284.
ACGIH (1991) Threshold limit values for chemical substances and
physical agents and biological exposure indices for 1991-1992.
Cincinnati, Ohio, American Conference of Environmental Industrial
Hygienists.
Adachi A, Asaka Y, Ozasa M, Sawai N, & Kobayashi T (1987) [Formation
of cyanide ion by the reaction of phenol with nitrous acid in waste
water.] Eisei Kagaku, 33(6): 445-448 (in Japanese, with English
abstract).
Aelion CM, Swindoll CM, & Pfaender FK (1987) Adaptation to and
biodegradation of xenobiotic compounds by microbial communities from
a pristine aquifer. Appl Environ Microbiol, 53(9): 2212-2217.
Agrawal HP (1987) Evaluation of the toxicity of phenol and sodium
pentachlorophenate to the snail Indoplanorbis exustus (Deshayes).
J Anim Morphol Physiol, 34(1-2): 107-112.
Alabaster JS (1969) Survival of fish in 164 herbicides,
insecticides, fungicides, wetting agents and miscellaneous
substances. Int Pest Control, March/April: 29-35.
Alekseyev VA & Antipin BN (1976) Toxicological characteristics and
symptoms of acute phenol poisoning in some freshwater crustaceans
and molluscs. Hydrobiol J, 12(2): 27-33.
Alexander DG & Clarke RMcV (1978) The selection and limitations of
phenol as a reference toxicant to detect differences in sensitivity
among groups of rainbow trout ( Salmo gairdneri). Water Res, 12:
1085-1090.
Al-Sabti K (1985) Frequency of chromosomal aberrations in the
rainbow trout, Salmo gairdneri, exposed to 5 pollutants. J Fish
Biol, 26(1): 13-20.
American Public Health Association, American Waterworks Association,
Water Pollution Control Federation (1985) Standard methods for the
examination of water and wastewater - Phenol, 16th ed. Washington,
DC, American Public Health Association, pp 556-570.
Amlathe S, Upadhyay S, & Gupta VK (1987) Spectrophotometric
determination of trace amounts of phenol in waste water and
biological fluids. Analyst, 112: 1463-1465.
Amoore JE & Hautala E (1983) Odor as an aid to chemical safety; odor
thresholds compared with threshold limit values and volatilities for
214 industrial chemicals in air and water dilution. J Appl Toxicol,
3: 272-290.
Andersen W (1869) Fatal misadventure with carbolic acid. Lancet, 1:
179.
Anniko M, Hellstrom S, Schmidt SH, & Spandow O (1988) Toxic effects
on inner ear of noxious agents passing through the round window
membrane. Acta Otolaryngol (Stockholm), 457(Suppl): 49-56.
Aquino MD, Korol S, Santini P, & Moretton J (1988) Biodegradation of
phenolic compounds: I. Improved degradation of phenol and benzoate
by indigenous strains of Acinobacter and Pseudomonas. Rev Latinoam
Microbiol, 30(3): 283-288.
Aranyi C, O'Shea WJ, Graham JA, & Miller FJ (1986) The effects of
inhalation of organic chemical air contaminants on murine lung host
defences. Fumdam Appl Toxicol, 6(4): 713-720.
Arvin E, Jensen BK, & Gundersen TA (1991) Biodegradation kinetics of
phenol in an aerobic biofilm at low concentrations. Water Sci
Technol, 23: 1375-1384.
Atkinson GL & Shupak RC (1989) Acute bronchospasm complicating
intercostal nerve block. Anesth Analg, 68: 400-401.
Atkinson R, Darnall KR, Lloyd AC, Winer AM, & Pitts JN Jr (1979)
Kinetics and mechanisms of the reactions of the hydroxyl radical
with organic compounds int he gas phase. Adv in Photochem, 11: 375.
ATSDR (1989) Toxicological profile for phenol. Atlanta, Georgia,
Agency for Toxic Substances and Disease Registry, 111 pp
(ATSDR/TP-89/20).
Babich H & Stotzky G (1985) A microbial assay for determining the
influence of physicochemical environmental factors on the toxicity
of organics: phenol. Arch Environ Contam Toxicol, 14(4): 409-416.
Baird RB, Kuo CL, Shapiro JS, & Yanko WA (1974) The fate of
phenolics in waste water - determination by direct injection GLC and
Warburg respirometry. Arch Environ Contam Toxicol, 2: 165-169.
Bak F & Widdell F (1986) Anaerobic degradation of phenol and phenol
derivatives by Desulfobacterium phenolicum, new species. Arch
Microbiol, 146(2): 177-180.
Baker MD & Mayfield CI (1980) Microbial and nonbiological
decomposition of chlorophenols and phenol in soil. Water Air Soil
Pollut, 13: 411-424.
Baker EL, Bertozzi PE, Field PH, Basteyns BJ, & Skinner HG (1978)
Phenol poisoning due to contaminated drinking water. Arch Environ
Health, 33: 89-94.
Barale R, Marrazzini A, Betti C, Vangelisti F, Loprieno N, & Barrai
J (1990) Genotoxicity of two metabolites of benzene: phenol and
hydroquinone show strong synergistic effects in vivo. Mutat Res,
244: 15-20.
Baranowska-Dutkiewicz B (1981) Skin absorption of phenol from
aqueous solutions in men. Int Arch Occup Environ Health, 49: 99-104.
Behl CR, Linn EE, Flynn GL, Pierson CL, Higuchi WI, & Ho NFH (1983a)
Permeation of skin and eschar by antiseptics: I. Baseline studies
with phenol. J Pharm Sci, 72(4): 391-397.
Behl CR, Linn EE, Flynn GL, Ho NFH, Higuchi WI, & Pierson CL (1983b)
Permeation of skin and eschar by antiseptics: II. Influence of
controlled burns on the permeation of phenol. J Pharm Sci, 72(4):
397-400.
Bennett IL, James DF, & Golden A (1950) Severe acidosis due to
phenol poisoning. Report of two cases. Ann Intern Med, 32: 324-327.
Benzon HT (1979) Convulsions secondary to intravascular phenol: a
hazard of celiac plexus block. Anesth Analg, 58(2): 150-151.
Beyer J & Frank G (1985) Hydroxylation and conjugation of phenol by
the frog, Rana temporaria. Xenobiotica, 15(4): 277-280.
Bergshoeff G (1960) [Determination of phenols in air and water. Part
III-A. Instructions for the method using p-nitroaniline and for the
method using ultraviolet spectrophotometry.] Delft, The Netherlands,
Dutch Organization for Applied Scientific Research (TNO-IMG) (Report
No. F765) (in Dutch).
Bernard DP & Salt DB (1982) Carcinogen specificity of gamma-glutamyl
transpeptidase-rich foci chemically induced in hamster buccal pouch
epithelium. 1st Int. Conf., Boston, Mass., 173-182.
Birge WJ, Black JA, Hudson JE, & Bruser DM ((1979) Embryo larval
toxicity test with organic compounds - Aquatic Toxicology. In:
Proceedings of the Second Annual Symposium on Aquatic Toxicology, 31
October-1 November 1977. pp 131-147.
Birge WJ, Black JA, & Kuehne RA (1980) Effects of organic compounds
on amphibial reproduction. Lexington, Kentucky Water Resources
Research Institute, 50 pp.
Blaber LC & Gallagher JP (1971) The facilitory effects of catechol
and phenol at the neuromuscular junction of the cat.
Neuropharmacology, 10: 153-159.
Black JA, Birge WJ, Westerman AC, & Francis PC (1983) Comparative
aquatic toxicology of aromatic hydrocarbons. Fundam Appl Toxicol,
3(5): 353-358.
Bohrman JS, Burg JR, Elmore E, Gulati DK, Barfknecht TR, Niemeier
RW, Dames BL, Toraason M, & Langenbach R (1988) Interlaboratory
studies with the Chinese hamster V79 cell metabolic cooperation
assay to detect tumor-promoting agents. Environ Mol Mutagen, 12(1):
33-51.
Boiano JM (1985) Health hazard evaluation report, University of
Pennsylvania, Medical Education Building, Philadelphia,
Pennsylvania. Cincinnati, OH, National Institute for Occupational
Safety and Health (HETA 84-098-1497; NTIS/PB85-220861).
Bolcsak LE & Nerland DE (1983) Inhibition of erythropoiesis by
benzene and benzene metabolites. Toxicol Appl Pharmacol, 69(3):
363-368.
Bollig FJ & Decker KH (1980) [Phenol resins.] Kunststoffe, 70: 672
(in German).
Bolt HM (1977) Structural modifications in contraceptive steroids
altering their metabolism and toxicity. Arch Toxicol, 39: 13-19.
Borovik EB & Dmitriev MT (1981) [Principles of air interchange as a
basis for the hygienic standardization of the air inside hospitals.]
Gig i Sanit, 2: 22-25 (in Russian).
Boutwell RK & Bosch DK (1959) The tumour promoting action of phenol
and related compounds for mouse skin. Cancer Res, 19: 413-424.
Boutwell RK, Rusch HP, & Bosch DK (1955) The action of phenol and
related compounds in tumour formation. Proc Am Assoc Cancer Res, 2:
6-7.
Boutwell RK, Rusch HP, & Booth B (1956) Tumour production by phenol
and related compounds. Proc Am Assoc Cancer Res, 2: 96.
Boyd SA (1982) Adsorption of substituted phenols by soil. Soil Sci,
134(5): 337-343.
Bradbury SP, Henry TR, Niemi GJ, Carlson RW, & Snarski VM (1989) Use
of respiratory-cardiovascular responses of rainbow trout ( Salmo
gairdneri) in identifying acute toxicity syndromes in fish: Part
3. Polar narcotics. Environ Toxicol Chem, 9(3): 247-262.
Bratzler LJ, Spooner ME, Weatherspoon JB, & Maxey JA (1969) Smoke
flavour as related to phenol, carbonyl and acid content of Bologna.
J Food Sci, 34: 146-148.
Bray HG, Humphris BG, Thorpe WV, White K, & Wood PB (1952a) Kinetic
studies of the metabolism of foreign organic compounds. 3. The
conjugation of phenol with glucuronic acid. Biochem J, 52: 416-419.
Bray HG, Humphris BG, Thorpe WV, White K, & Wood PB (1952b) Kinetic
studies of the metabolism of foreign organic compounds. 4. The
conjugation of phenol with sulfuric acid. Biochem J, 52: 419-423.
Bray HG, Thorpe WV, & White K (1952c) Kinetic studies of the
metabolism of foreign organic compounds. 5. A mathematical model
expressing the metabolic fate of phenols, benzoic acids and their
precursors. Biochem J, 52: 423-430.
Bringmann G & Kuhn R (1959) [Studies on water toxicology using
protozoa as test organisms.] Gesundheits-Ing, 80: 239-242 (in
German).
Bringmann G & Kuhn R (1977) [Threshold values for the harmful effect
of water pollutants on bacteria ( Ps. putida) and green algae ( Sc.
quadricauda) in the cell reproduction inhibition test.] Z Wasser
Abwasser Forsch, 80: 87-98 (in German).
Bringmann G & Kuhn R (1978) [Threshold values for the harmful effect
of water pollutants on blue algae ( Microcystis aeruginosa) and
green algae ( Scenedesmus quadricauda) in the cell reproduction
inhibition test.] Vom Wasser, 80: 45-60 (in German).
Bringmann G & Kuhn R (1980) Comparison of the toxicity thresholds of
water pollutants to bacteria, algae, and protozoa in the cell
multiplication inhibition test. Water Res, 14(3): 231-241.
Bringmann G, Kuhn R, & Winter A (1980) [Determination of the harmful
biological action of water endangering substances on protozoa. III.
Saprozic flagellates.] Z Wasser Abwasser Forsch, 13(5): 170-173 (in
German).
Brodzinsky R & Singh HB (1982) Volatile organic chemicals in the
atmosphere: an assessment of available data. Research Triangle Park,
North Carolina, US Environmental Protection Agency, Environmental
Sciences Research Laboratory (EPA/600/3-83/027a; NTIS PB83-195503).
Brown VM, Jordan DHM, & Tiller BA (1967) The effects of temperature
on the acute toxicity of phenol to rainbow trout in hard water.
Water Res, 1: 587-594.
Brown VKH, Box VL, & Simpson BJ (1975) Decontamination procedures
for skin exposed to phenolic substances. Arch Environ Health, 30:
1-6.
Bruce RM, Santodonato J, & Neal MW (1987) Summary review of the
health effects associated with phenol. Toxicol Ind Health, 3(4):
535-568.
Budavari S, O'Neil MJ, Smith A, & Heckelman PE ed. (1989) The Merck
index. Rahway, New Jersey, Merck & Co., Inc., p 1150.
Buikema AL, McGinness MJ, & Cairns J (1979) Phenolics in aquatic
ecosystems. A selected review of recent literature. Mar Environ Res,
2(2): 87-181.
Bulsiewicz H (1977) The influence of phenol on chromosomes of mice
in the process of spermatogenesis. Folia Morphol (Warsaw), 36:
13-22.
Cairns J, Messenger DJ, & Calhoun WF (1976) Invertebrate response to
thermal shock following exposure to acutely sub-lethal
concentrations of chemicals. Arch Hydrobiol, 77(2): 164-175.
Cairns J, Buikema AL Jr, Heath AG, & Parker BC (1978) Effects of
temperature on aquatic organism sensitivity to selected chemicals.
Blackburg, Virginia, Virginia Water Resources Research Center
(Bulletin 106).
Call DJ, Brooke LT, & Lu PY (1980) Uptake, elimination, and
metabolism of three phenols by Fathead minnow. Arch Environ Contam
Toxicol, 9(6): 699-714.
Campbell NRC, Van Loon JA, & Weinshilboum RM (1987) Human liver
phenol sulfotransferase: assay conditions, biochemical properties
and partial purification of isozymes of the thermostable form.
Biochem Pharmacol, 36(9): 1435-1446.
Capel ID, French MR, Millburn P, Smith RL, & Williams RT (1972a)
Species variations in phenol metabolism. Biochem J, 127: 25-26.
Capel ID, French MR, Millburn P, Smith RL, & Williams RT (1972b) The
fate of 14C phenol in various species. Xenobiotica, 2: 25-34.
Capel ID, Millburn P, & Williams RT (1974) Monophenyl phosphate, a
new conjugate of phenol in the cat. Biochem Soc Trans, 2: 305-306.
Cassidy MK & Houston JB (1980) In vivo assessment of extrahepatic
conjugative metabolism in first pass effects using the model
compound phenol. J Pharm Pharmacol, 32(5): 57-59.
Cassidy MK & Houston JB (1984) In vivo capacity of hepatic and
extrahepatic enzymes to conjugate phenol. Drug Metab Dispos, 12:
619-624.
Chagnon N & Hlohowskyj I (1989) Effects of phenol exposure on the
thermal tolerance ability of the central stoneroller minnow. Bull
Environ Contam Toxicol, 42(4): 614-619.
Chemfacts (1978-1981) Chemical Data Sources - Chemfacts.
Publications for the Netherlands, Belgium, West-Germany, France,
Great Britain, Spain, Italy, Portugal and Scandinavia.
Chen TH, Kavanagh TJ, Chang CC, & Trosko JE (1984) Inhibition of
metabolic cooperation in Chinese hamster V79 cells by various
organic solvents and simple compounds. Cell Biol Toxicol, 1(1):
155-171.
Chesney RH, Sollitti P, & Rubin HE (1985) Incorporation of phenol
carbon at trace concentrations by phenol-mineralizing microorganisms
in fresh water. Appl Environ Microbiol, 49(1): 15-18.
CID-TNO (1984) Databases ECDIN and PROMT.
Ciranni R, Barale R, Ghelardini G, & Loprieno N (1988a) Benzene and
the genotoxicity of its metabolites. I. Transplacental activity in
mouse fetuses and in their dams. Mutat Res, 208: 61-67.
Ciranni R, Barale R, Ghelardini G, & Loprieno N (1988b) Benzene and
the genotoxicity of its metabolites. II. The effect of the route of
administration on the micronuclei and bone marrow depression in
mouse bone marrow cells. Mutat Res, 209: 23-28.
Coan ML, Baggs RB, & Bosmann HB (1982) Demonstration of direct
toxicity of phenol on kidney. Chem Pathol Pharmacol Res Commun,
36(2): 229-239.
Colvin RJ & Rozich AR (1986) Phenol growth kinetics of heterogenous
populations in a two-stage continuous culture system. Water Pollut
Control Fed, 58(4): 326-332.
Conning DM & Hayes MJ (1970) The dermal toxicity of phenol, and
investigation of the most effective first-aid measures. Br J Ind
Med, 27: 155-159.
Cooper IS, Hirose T, Matsuoka S, Roth D, Waltz JM, & Ericsson AD
(1965) Specific neurotic perfusion: a new approach to selected cases
of pain and spasticity. Neurology, 15: 985.
Cotruvo JA, Simmon VF, & Sponggord RJ (1977) Investigation of
mutagenic effects of products of ozonation reactions in water. Ann
NY Acad Sci, 298: 124-140.
Crebelli R, Conti G, & Carere A (1987) On the mechanism of mitotic
segregation induction in Aspergillus nidulans by benzene hydroxy
metabolites. Mutagenesis, 2(3): 235-238.
Cronin TD & Brauer RO (1949) Death due to phenol contained in
Foille. J Am Med Assoc, 139: 777-779.
Curtis C, Lima A, Lozano SJ, & Veith GD (1982) Philadelphia,
Pennsylvania, American Society for Testing and Materials, p 170
(ASTM Special Technical Publication No. 766).
Dalela RC, Rani S, & Verna SR (1980) Influence of pH on the toxicity
of phenol and its 2 derivatives pentachlorophenol and dinitrophenol
to some fresh water teleosts. Acta Hydrochim Hydrobiol, 8(6):
623-628.
Dalin NM & Kristofferson R (1974) Physiological effects of a
sublethal concentration of inhaled phenol on the rat. Ann Zool Fenn,
11: 193-199.
Dangé AD (1986) Metabolic effects of naphthalene, toluene or phenol
intoxication in the cichlid fish tilapia, Oreochromis mossambicus:
changes in aminotransferase activities. Environ Pollut, A42:
311-323.
Dauble DD, Carlile DW, & Hanf RW Jr (1986) Bioaccumulation of fossil
fuel components during single-compound and complex-mixture exposures
of Daphnia magna. Bull Environ Contam Toxicol, 37: 125-132.
Daum F, Cohen MJ, & McNamara H (1976) Experimental toxicological
studies on a phenol detergent associated with neonatal
hyperbilirubinemia. J Pediatr, 89: 853-854.
Davis HC & Hidu H (1969) Effects of pesticides on embryonic
development of clams and oysters and on survival and growth of the
larvae. US Dept. Inter., Fish Wildl. Bull., 67: 393.
De Ceaurriz JC, Micillino JC, Bonnet P, & Guenier JP (1981) Sensory
irritation caused by various industrial airborne chemicals. Toxicol
Lett, 9: 137-143.
De Graeve GM, Geiger WL, Meyer JS, & Bergman HL (1980) Acute and
embryo-larval toxicity of phenolic compounds to aquatic biota. Arch
Environ Contam Toxicol, 9: 557-568.
De Groote V & Lambotte C (1960) A case of fatal phenol poisoning in
a newborn infant. Ann Méd Lég, 40: 288-290.
Deichmann WB (1944) Phenol studies. V. The distribution,
detoxification, and excretion of phenol in the mammalian body. Arch
Biochem, 2: 345-355.
Deichmann WB (1949) Local and systemic effects following skin
contact with phenol: a review of the literature. J Ind Hyg Toxicol,
31: 146-154.
Deichmann WB & Keplinger ML (1963) Phenols and phenolic compounds.
In: Patty FA ed. Industrial hygiene and toxicology. New York,
Interscience Publishers, pp 2567-2627.
Deichmann WB & Oesper P (1940) Ingestion of phenol - effects of the
albino rat. Ind Med, 9: 296.
Deichmann WB & Schafer LJ (1942) Phenols studies - I: Review of the
literature. II: Quantitative spectrophotometric estimation of free
and conjugated phenol in tissues and fluids. III: Phenol content of
normal human tissues and fluids. Am J Clin Pathol, 12: 129-143.
Deichmann WB & Witherup S (1944) Phenol studies - VI: The acute and
comparative toxicity of phenol and o-, m-, and p-cresols for
experimental animals. J Pharmacol Exp Ther, 80: 233-240.
Deichmann WB, Kitzmiller KV, & Witherup S (1944) Phenols studies -
VII: Chronic phenol poisoning, with special reference to the effects
upon experimental animals of the inhalation of phenol vapour. Am J
Clin Pathol, 14: 273-277.
Deichmann WB, Miller T, & Roberts JB (1950) Local and systemic
effects following application of dilute solutions of phenol in water
and in camphor-liquid petrolatum on the skin of animals. Arch Ind
Hyg Occup Med, 2: 454-461.
Deichmann WB, Witherup S, & Dierker M (1952) Phenol studies - XII:
The percutaneous and alimentary absorption of phenol by rabbits with
recommendations for the removal of phenol from the alimentary tract
or skin of persons suffering exposure. J Pharmacol Exp Ther, 105:
265-272.
Delfino JJ & Dube DJ (1976) Persistent contamination of ground water
by phenol. J Environ Sci Health, pp 345-355.
DeMeio RH & Arnolt RI (1944) Phenol conjugation - II: The
conjugation by rat and cat tissues in vitro. J Biol Chem, 156:
577-583.
Dement J, Wallingford K, & Zumwalde R (1973) Industrial hygiene
survey of Owens-Corning Fiberglas, Kansas, City, Kansas. Cincinnati,
Ohio, National Institute for Occupational Safety and Health (Report
No. IW 35.16).
Demerec M, Bertani G, & Flint J (1951) A survey of chemicals for
mutagenic action on E. coli. Am Nat, 85: 119-136.
Den Boeft J, Kruiswijk FJ, & Schulting FL (1984) [Air pollution by
combustion of solid fuels.] The Hague, Ministry of Housing, Physical
Planning and Environment (Publication Lucht No. 37) (in Dutch).
De Neer JW, Seinen W, & Hermens JLM (1988) Growth of Daphnia magna
exposed to mixtures of chemicals with diverse modes of action.
Ecotoxicol Environ Saf, 15(1): 72-77.
Dimitriev MT & Mischchikhin VA (1983) [Gas chromatographic
determination of phenol in air.] Gig i Sanit, 5: 42-44 (in Russian).
Dive O & Leclerc H (1977) Utilisation du protozoaire cilie colpidum
campylum pour la meure de la toxicité et de l'accumulation des
micropollutants: analyse critique et applications. Environ Pollut,
14: 169-186.
Doan HMcK, Keith L, & Shennan AT (1979) Phenol and neonatal
jaundice. Pediatrics, 64(3): 324-325.
Dobbins DC, Thornton-Manning JR, Jones DD, & Federle TW (1987)
Mineralization potential for phenol in subsurface soils. J Environ
Qual, 16(1): 54-58.
Docter HJ & Zielhuis RL (1967) Phenol excretion as a measure of
benzene exposure. Ann Occup Hyg, 10: 317-326.
Dosemeci M, Blair A, Stewart, PA, Chandeler J, & Trush MA (1991)
Mortality among industrial workers exposed to phenol. Epidemiology,
2, 188-193.
Dow Chemical Company (1976) References and literature review
pertaining to toxicological properties of phenol. Dow Chemical
Company, Toxicological Research Laboratory (unpublished manuscript).
Dowden BF & Benett HJ (1965) Toxicity of selected chemicals to
certain animals. J Water Pollut Control Fed, 37: 1308-1316.
Dreibelbis WG & Hawthorne AR (1985) Worker exposure potential at
coal conversion facilities. Oak Ridge, Tennessee, Oak Ridge National
Laboratory (NTIS/DE85006822).
Dreisbach RH (1983) Handbook of poisoning: Prevention, diagnosis &
treatment, 11th ed. Los Altos, California, Lange Medical
Publications, pp 401-405.
Dumpert K (1987) Embryotoxic effects of environmental chemicals:
tests with the South African clawed toad ( Xenopus laevis).
Ecotoxicol Environ Saf, 13(3): 324-338.
Dutka BJ & Kwann KK (1981) Comparison of three microbial toxicity
screening tests with the Microtox test. Bull Environ Contam Toxicol,
27: 753-757.
Dutka BJ, Nyholm N, & Peterson J (1983) Comparison of several
microbiological toxicity screening tests. Water Res, 17(10):
1363-1368.
Duverneuil G & Ravier E (1962) Toxicité suraiguë du phénol par voie
transcutanée. Arch Mal Prof, 23: 830-833.
Eastmond DA, Smith MT, Ruzo LG, & Ross D (1986) Metabolic activation
of phenol by human myeloperoxidase and horseradish peroxidase. Mol
Pharmacol, 30(6): 674-679.
Eastmond DA, French RG, Ross D, & Smith MT (1987a) Metabolic
activation of 1-naphthol and phenol by a simple
superoxide-generating system and human leukocytes. Chem-Biol
Interact, 63(1): 47-62.
Eastmond DA, Smith MT, & Irons RD (1987b) An interaction of benzene
metabolites reproduces the myelotoxicity observed with benzene
exposure. Toxicol Appl Pharmacol, 91: 85-95.
Edwards VT, Jones BC, & Hudson DH (1986) A comparison of the
metabolic fate of phenol, phenyl glucoside and phenyl
6-o-malonyl-glucoside in the rat. Xenobiotica, 16: 801-807.
Ehrlich GG, Goelitz DF, & Godsy EM (1982) Degradation of phenolic
contaminants in ground water by anaerobic bacteria: St. Louis Park,
MN. Ground Water, 20: 703-710.
EIFAC (European Inland Fisheries Advisory Commission) (1972) Water
quality criteria for European freshwater fish. Report on monohydric
phenols and inland fisheries. Rome, Food and Agriculture
Organization of the United Nations (Technical Paper No. 15).
Epler JL, Rao TK, & Guerin MR (1979) Evaluation of feasibility of
mutagenic listing of shale oil products and effluents. Environ
Health Perspect, 30: 179-184.
Erben R (1982) The effect of phenol and its accumulation on snails
Physa fontinalis and Radix peregra mull (Pulmonata). Period
Biol, 84(4): 403-410.
Erben R, Munjko J, & Jovanovic V (1983) Influence of phenol
accumulation on the species Polycelis felina daly (Turbellaria).
Biologica (Bratislava), 38: 283-288.
Erexson GL, Wilmer JL, & Kligerman AD (1985) Sister chromatid
exchange induction in human lymphocytes exposed to benzene and its
metabolites in vitro. Cancer Res, 45(6): 2471-2477.
Ernst MR, Klesmer R, Huebner RA, & Martin JE (1961) Susceptibility
of cats to phenol. J Am Vet Med Assoc, 138: 197-199.
Ersek RA (1991) Comparative study of dermabrasion, phenol peel, and
acetic acid peel. Anesth Plast Surg, 15: 241-243.
Evans SJ (1952) Acute phenol poisoning. Br J Ind Med, 9: 227-229.
Farquharson ME, Gage JC, & Northover J (1958) The biological action
of chlorophenols. Br J Pharmacol, 13: 20-24.
Federle TW (1988) Mineralization of monosubstituted aromatic
compounds in unsaturated and saturated subsurface soils. Can J
Microbiol, 34(9): 1037-1042.
Fedorak PM & Hrudey SE (1986) Nutrient requirements for the
methanogenic degradation of phenol and p-cresol in anaerobic draw
and feed cultures. Water Res, 20(7): 929-934.
Fedorak PM, Roberts DJ, & Hrudey SE (1986) The effects of cyanide on
the methanogenic degradation of phenolic compounds. Water Res,
20(10): 1315-1320.
Feldman RJ & Maibach HI (1970) Absorption of some organic compounds
through the skin in man. J Invest Dermatol, 54: 399-404.
Fielding M, Gibson TM, James HA, et al (1981) Organic
micropollutants in drinking water. Medmenham, UK, Water Research
Centre (Report No. TR-159).
Fishbeck WA, Langner RR, & Kociba RJ (1975) Elevated urinary
phenol levels not related to benzene exposure. Am Ind Hyg Assoc J,
36(11): 820-824.
Flerov BA (1979) Physiological mechanisms of the action of toxic
substances and adaptation of aquatic animals to them. Hydrobiol J,
13(4): 70-74.
Flickinger CW (1976) The benzenediols: catechol, resorcinol and
hydroquinone - a review of the industrial toxicology and current
industrial exposure limits. Am Ind Hyg Assoc J, 37: 596-606.
Flynn GL & Yalkowsky SH (1972) Correlation and prediction of mass
transport across membranes. I. Influence of alkyl chain length on
flux-determining properties of barrier and diffusant. J Pharm Sci,
61: 838-852.
Fogels A & Sprague JB (1977) Comparative short-term tolerance of
zebrafish, flagfish, and rainbow trout to five poisons including
potential reference toxicants. Water Res, 11(9): 811-817.
Forrest F & Ramage DTO (1987) Cardiac dysrhythmia after subtrigonal
phenol. Anaesthesia, 42(7): 777-778.
Foxall PJD, Bending MR, Gartland KPR, & Nicholson JK (1991) Acute
renal failure following accidental cutaneous absorption of phenol.
In: Bach PH ed. Nephrotoxicity: Mechanisms, early diagnosis and
therapeutic management (Fourth International Symposium on
Nephrotoxicity, Guildford, UK, 1989). New York, Basel, Marcel
Dekker, Inc., pp 55-59.
Franz TJ (1975) Percutaneous absorption. On the relevance of in
vitro data. J Invest Dermatol, 64: 190-195.
Freitag D, Ballhorn L, Geyer H, & Korte F (1985) Environmental
hazard profile of organic chemicals. An experimental method for the
assessment of the behaviour of organic chemicals in the ecosphere by
means of simple laboratory tests with 14C-labelled chemicals.
Chemosphere, 14(10): 1589-1616.
French MR, Bababunmi EA, Golding RR, Bassir O, Caldwell J, Smith RL,
& Williams RT (1974) The conjugation of phenol, benzoic acid,
1-naphthylacetic acid and sulphadimethoxine in the lion, civet and
genet. FEBS Lett, 46(1): 134-137.
Fujii M (1978) Environmental monitoring of chemicals - Environmental
survey report of FY 1977. Tokyo, Environment Agency Japan,
Department of Environmental Health.
Gad-El Karim MM, Ramanujam VMS, Ahmed AE, & Legator MS (1965)
Benzene mycloclastogenicity: A function of its metabolism. Am J Ind
Med, 7: 475-484.
Gad-El Karim MM, Ramanujam VMS, & Legator MS (1966) Correlation
between the induction of micronuclei in bone marrow by benzene
exposure and the excretion of metabolites in urine of CD-1 mice.
Toxicol Appl Pharmacol, 85: 464-477.
Garberg P, Akerblom EL, & Bolcsfoldi G (1988) Evaluation of a
genotoxicity test measuring DNA-strand breaks in mouse lymphoma
cells by alkaline unwinding and hydroxyapatite elution. Mutat Res,
203: 155-176.
Garton GA & Williams RT (1949) Studies in detoxification. 26. The
fate of phenol, phenyl sulfuric acids and phenylglucuronide in the
rabbit in relation to the metabolism of benzene. Biochem J, 45:
158-163.
Gbodi TA & Oehme FW (1978) The fate of phenol, o-phenylphenol and
diisophenol in rats. Toxicol Appl Pharmacol, 45: 219-362.
Gersich FM, Blanchard FA, Applegath SL, & Park CN (1986) The
precision of daphnid ( Daphnia magna Straus, 1820) static acute
toxicity tests. Arch Environ Contam Toxicol, 15: 741-749.
Gibb C, Glover V, & Sandler M (1987) In vitro inhibition of phenol
sulphotransferase by food and drink constituents. Biochem Pharmacol,
36(14): 2325-2330.
Gibson II (1987) Phenol block in the treatment of spasticity.
Gerontology, 33(5): 327-330.
Gilbert P, Rondelet J, Poncelet F, & Mercier M (1980) Mutagenicity
of p-nitrosophenol. Food Cosmet Toxicol, 18: 523-525.
Gilli G, Palin L, & Scursatore E (1980) [Exposition to phenol.] Arch
Sci Med, 137: 41-44 (in Italian).
Gluth G & Hanke W (1983) The effect of temperature on physiological
changes in Carp Cyprinus carpio L. induced by phenol. Ecotoxicol
Environ Saf, 7(4): 373-389.
Gocke E, King MT, Eckhardt K, & Wild D (1981) Mutagenicity of
cosmetics, ingredients licensed by the European Communities. Mutat
Res, 90: 91-109.
Görge G, Beyer J, & Urich K (1987) Excretion and metabolism of
phenol, 4-nitrophenol and 2-methylphenol by the frogs Rana
temporaria and Xenopus laevis. Xenobiotica, 17(11): 1293-1298.
Green DWJ, Williams KA, & Pascoe D (1985) Studies on the acute
toxicity of pollutants to freshwater macroinvertebrates: 2. Phenol.
Arch Hydrobiol, 103(1): 75-82.
Green DWJ, Williams KA, Hughes DRL, Shaik GAR, & Pascoe D (1988)
Toxicity of phenol to Asellus aquaticus (L.): effects of
temperature and episodic exposure. Water Res, 22(2): 225-232.
Greenlee WF, Gross EA, & Irons RW (1981) Relationship between
benzene toxicity and the disposition of 14C-labelled benzene
metabolites in the rat. Chem-Biol Interact, 33: 285-299.
Groenen PJ (1978) [Components of tobacco smoke. Nature and quantity;
potential influence on health.] Zeist, The Netherlands, CIVO-TNO
Institute (Report No. R/5787) (in Dutch).
Gspan P, Jersic A, & Cadez E (1984) [Relationship between phenol
concentration in urine and work environment.] Staub-Reinhalt Luft,
44: 314-316 (in German).
Gunter BJ (1987) Health hazard evaluation report: CF & I Steel,
Pueblo, Colorado. Cincinnati, Ohio, National Institute for
Occupational Safety and Health (HETA 86-034-1712; NTIS/PB87-117016).
Gupta S & Dalela RC (1987) Kidney damage in Notopterus notopterus
(Pallas) following exposure to phenolic compounds. J Environ Biol,
8(2): 167-172.
Gupta PK & Rao PS (1982) Toxicity of phenol, pentachlorophenol and
sodium pentachlorophenolate to a freshwater pulmonate snail Lymnaea
acuminata (Lamarck). Arch Hydrobiol, 94: 210-217.
Gupta PK, Mujumdar VS, Rao PS, & Durve VS (1982a) Toxicity of
phenol, pentachlorophenol and sodium pentachlorophenolate to a
freshwater teleost Lebistes reticulatus (Peters). Acta Hydrochim
Hydrobiol, 10(2): 177-181.
Gupta S, Verma Sr, & Saxena PK (1982b) Toxicity of phenolic
compounds in relation to the size of a freshwater fish, Notopterus
notopterus (Pallas). Ecotoxicol Environ Saf, 6: 433-438.
Gupta S, Dalela RC, & Saxena PK (1983a) Influence of dissolved
oxygen levels on acute toxicity of phenolic compounds to a
freshwater teleost, Notopterus notopterus (Pallas). W.A.s. Pol.,
19(3): 223-228.
Gupta S, Dalela RC, & Saxena PK (1983b) Influence of temperature on
the toxicity of phenol and its chloro and nitro-derivates to the
fish Notopterus notopterus (Pallas). Acta Hydrochim Hydrobiol,
11(2): 187-192.
Gurujeyalashmi S & Oriel P (1989) Isolation of phenol-degrading
Bacillus stearothermophilus and partial characterization of the
phenol hydroxylase. Appl Environ Microbiol, 55(2): 500-502.
Haddad LM, Dimend KA, & Schweistris JE (1979) Phenol poisoning.
JACEP, 8(7): 267-269.
Hadorn E & Niggli N (1946) Mutations in Drosophila after chemical
treatment of gonads in vitro. Nature (Lond), 157: 162-163.
Hagemann R, Virelizier H, & Gaudin D (1978) Analyse de composés
organiques dans une atmosphère urbaine. Analusis, 6(9): 401-405.
Hardy JT, Dauble DD, & Felice LJ (1985) Aquatic fate of synfuel
residuals: bioaccumulation of aniline and phenol by the freshwater
phytoplankton Scenedesmus quadricauda. Environ Toxicol Chem, 4(1):
29-35.
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(Suppl): 3-142.
Healy JB Jr & Young LY (1979) Anaerobic biodegradation of eleven
aromatic compounds to methane. Appl Environ Microbiol, 28: 84-89.
Heikkilä P, Hämeilä M, Pyy L, & Raunu P (1987) Exposure to creosote
in the impregnation and handling of wood. Scand J Work Environ
Health, 13: 431-437.
Heller VG & Pursell L (1938) Phenol-contaminated waters and their
physiological action. J Pharmacol Exp Ther, 63: 99-107.
Henschler D ed. (1975) [Phenol.] In: [Analytical methods for the
testing of working media harmful to health. Volume 1: Air analyses.]
Weinheim, Verlag Chemie (in German).
Herbert DWM (1962) The toxicity to rainbow trout of spent still
liquors from the distillation of coal. Ann Appl Biol, 50: 755-777.
Hermens J (1984) Joint effects of a mixture of 14 chemicals on
mortality and inhibition of reproduction of Daphnia magna.
(Thesis).
Hickman GT & Novak JT (1989) Relationship between subsurface
biodegradation rates and microbial density. Environ Sci Technol,
23(5): 524-532.
Hinkel GK & Kintzel HW (1968) Phenol poisoning of a newborn through
skin resorption. Dtsch Gesundh, 23: 2420-2422.
Hoffmann KH & Vogt U (1988) [Degradation of phenol by yeasts in the
presence of n-hexadecane under growth conditions in a stirred
reactor.] Zent.bl Mikrobiol, 143(2): 87-91 (in German, with English
abstract).
Hogg SI, Curtis CG, Upshall DG, & Powell GM (1981) Conjugation of
phenol by rat lung. Biochem Pharmacol, 30(12): 1551-1555.
Holcombe GW, Phipps GL, & Fiandt JT (1982) Effects of phenol,
2,4-dimethylphenol, 2,4-dichlorophenol and pentachlorophenol on
embryo, larval and early-juvenile fathead minnows ( Pimephales
promelas). Arch Environ Contam Toxicol, 11: 73-78.
Holcombe GW, Phipps GL, Sulaiman AH, & Hoffman AD (1987)
Simultaneous multiple species testing: acute toxicity of 13
chemicals to 12 diverse freshwater amphibian, fish, and invertebrate
families. Arch Environ Contam Toxicol, 16(6): 697-710.
Hoshika Y & Muto G (1979) Sensitive gas chromatographic
determination of phenols as bromophenols using electron capture
detection. J Chromatogr, 179: 105-111.
Hoshika Y & Muto G (1980) Gas-liquid-solid chromatographic
determination of phenols in air using Tenax-GC and alkaline
precolumns. J Chromatogr, 187: 277-284.
Hoshino M & Akimoto H (1978) Photochemical oxidation of benzene,
toluene and ethylbenzene initiated by OH radicals in the gas phase.
Bull Chem Soc Jpn, 51: 718.
Houston JB & Cassidy MK (1982) Sites of phenol conjugation in the
rat in sulfate metabolism and sulfate conjugation. In: Proceedings
of an international workshop held at Noordwijkerhout, The
Netherlands, 20-23 September 1981. Taylor & Francis, pp 270-278.
Howard PH (1989) Handbook of environmental fate and exposure data
for organic chemicals. Chelsea, Michigan, Lewis Publishers, vol 1,
pp 468-476.
Hsieh GC, Sharma RP, Parker RDR, & Coulombe RA Jr (1992)
Immunological and neurobiochemical alterations induced by repeated
oral exposure of phenol in mice. Eur J Pharmacol - Environ Toxicol
Pharmacol Sect, 228: 107-114.
Hubble BR, Stetter JR, Gebert E, Harkness JBL, & Flotard RD (1981)
Experimental measurements from residential wood-burning stoves.
Proceedings of the International Conference on Residential Solid
Fuels: Environmental Impacts and Solutions, John A. Cooper & Dorothy
Malek.
Huq AS, Ho NFH, Husari N, Flynn GL, Jetzer WE, & Condie L (1986)
Permeation of water contaminative phenols through hairless mouse
skin. Arch Environ Contam Toxicol, 15(5): 557-566.
Hwang HM, Hodson RE, & Lee RF (1986) Degradation of phenol and
chlorophenols by sunlight and microbes in estuarine water. Environ
Sci Technol, 20(10): 1002-1007.
Hwang HM, Hodson RE, & Lewis DL (1989) Assessing interactions of
organic compounds during biodegradation of complex waste mixtures by
naturally occurring bacterial assemblages. Environ Toxicol Chem,
8(3): 209-214.
IARC (1989) Phenol. In: Some organic solvents, resin monomers and
related compounds, pigments and occupational exposures in paint
manufacture and painting. Lyon, International Agency for Research on
Cancer, pp 263-287 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 47).
Ikeda M & Ohtsuji H (1969) Hippuric acid, phenol and trichloroacetic
acid levels in the urine of Japanese subjects with no known exposure
to organic solvents. Br J Ind Med, 26: 162-164.
Illing HPA & House ESA (1980) Binding of (14C) phenol to rat liver
high-speed supernatant. Biochem Soc Trans, 8(1): 117-118.
Isaacson PJ & Frink GR (1984) Nonreversible sorption of phenolic
compounds by sediment fractions: the role of sediment organic
matter. Environ Sci Technol, 18: 43-48.
Itoh M (1982) Sensitisation potency of some phenolic compounds. J
Dermatol, 9(3): 223-283.
Ivett JL, Brown BM, Rodgers C, Anderson BE, Resnick MA, & Zeiger E
(1989) Chromosomal aberrations and sister chromatid exchange tests
in Chinese hamster ovary cells in vitro. IV. Results with 15
chemicals. Environ Mol Mutagen, 14: 165-187.
Jansson T, Curvall M, Hedin A, & Enzell CR (1986) In vitro studies
of biological effects of cigarette smoke condensate. II. Induction
of sister chromatid exchanges in human lymphocytes by weakly acidic,
semivolatile constituents. Mutat Res, 169: 129-139.
Jarvis SN, Straube RC, Williams ALJ, & Bartlett CLR (1985) Illness
associated with contamination of drinking water supplies with
phenol. Br Med J, 290: 1800-1802.
Jelinek R, Peterka M, & Rychter Z (1985) Chick embryotoxicity
screening test - 130 substances tested. Indian J Exp Biol, 23:
588-595.
Jergil B, Schelin C, & Tunek A (1982) Covalent binding of
metabolically activated hydrocarbons to specific microsomal
proteins. Adv Exp Med Biol, 136A: 341-348.
Jetzer WE, Huq AS, Ho NFH, Flynn GL, Duraiswamy N, & Condie L (1986)
Permeation of mouse skin and silicone rubber membranes by phenols:
relationship to in vitro partitioning. J Pharm Sci, 75(11):
1098-1103.
Jetzer WE, Hou SY, Huq AS, Duraiswamy N, Ho NFH, & Flynn GL (1988)
Temperature dependency of skin permeation of waterborne organic
compounds. Pharm Acta Helv, 63(7): 197-201.
Jones-Price C, Kimmel CA, Ledoux TA, Reel JR, Fisher PW,
Langhoff-Paschke L, & Marr MC (1983a) Final study report -
Teratologic evaluation of phenol (CAS No. 108-95-2) in CD rats (NTP
Study No. TER-81-104). Springfield, Virginia, National Technical
Information Service (NTIS/PB83-247726).
Jones-Price C, Kimmel CA, Ledoux TA, Reel JR, Langhoff-Paschke L, &
Marr MC (1983b) Final study report - Teratologic evaluation of
phenol (CAS No. 108-95-2) in CD-1 rats (NTP Study No. TER-80-129).
Springfield, Virginia, National Technical Information Service (NTIS/
PB85-104461.
Judis J (1982) Binding of selected phenol derivatives to human serum
proteins. J Pharm Sci, 71(110): 1145-1147.
Jünke I & Lüdemann D (1978) [Results of the testing of 200 chemical
compounds for acute toxicity for fish by the golden orfe test.] Z
Wasser Abwasser Forsch, 11(5): 161-164 (in German).
Kao J, Bridges JW, & Faulkner JK (1979) Metabolism of 14C phenol
by sheep, pig and rat. Xenobiotica, 9(3): 141-147.
Kasokat T, Nagel R, & Urich K (1987) The metabolism of phenol and
substituted phenols in zebra fish. Xenobiotica, 17(10): 1215-1222.
Katz M ed. (1977) Tentative method of analysis for determination of
phenolic compounds in the atmosphere with the 4-amino antipyrine
method - Methods of air sampling and analysis, 2nd ed. Washington,
DC, American Public Health Association, pp 324-332.
Kauppinen TP, Partanen TJ, Nurminen MM, Nickels JI, Hernberg, SG,
Hakulinen TR, Pukkala EI, & Savonen ET (1986) Respiratory cancers
and chemical exposures in the wood industry: a nested case-control
study. Br J Ind Med, 43(2): 84-90.
Kavlock RJ (1990) Structure-activity relationships in the
developmental toxicity of substituted phenols: in vivo effects.
Teratology, 41: 43-59.
Keen R & Baillod CR (1985) Toxicity to Daphnia of the end products
of wet oxidation of phenol and substituted phenols. Water Res,
19(6): 767-772.
Key PB & Scott GI (1986) Lethal and sublethal effects of chlorine,
phenol, and chlorine-phenol mixtures on the mud crab Panopeus
herbstii. Environ Health Perspect, 69: 307-312.
Khalili AA & Betts HB (1967) Peripheral nerve block with phenol in
the management of spasticity. J Am Med Assoc, 200: 1155.
Kifune I (1979) [Determination of micro amounts of phenol and
cresols in the atmosphere by using a filter impregnated with a
solvent of alkali and glycerol.] Bunseki Kagaku, 28(11): 638-642 (in
Japanese).
Kirk RE & Othmer DF (1980) Encyclopedia of chemical toxicology, 3rd
ed. New York, John Wiley and Sons, vol 17, pp 373-379.
Kligman AM (1966) The identification of contact allergens by human
assays. III. The maximization test: A procedure for screening and
rating contact sensitizers. J Invest Dermatol, 47(5): 393-409.
Knapik Z, Hanczyc H, Lubczynska-Kowalska W, Menzel-Lipinska M, Cader
J, Paradowski L, & Borowka Z (1980) Use of subclinical forms of the
toxic action of phenol. Z Gesamte Hyg Grenzgeb, 26(8): 585-587.
Knezovich JP, Hirabayashi JM, Bishop DJ, & Harrison FL (1988) The
influence of different soil types on the fate of phenol and its
biodegradation products. Chemosphere, 17(11): 2199-2206.
Knoevenagel K & Himmelreich R (1976) Degradation of compounds
containing carbon atoms by photooxidation in the presence of water.
Arch Environ Contam Toxicol, 4: 324-333.
Knoll G & Winter J (1987) Anaerobic degradation of phenol in sewage
sludge: benzoate formation from phenol and carbon dioxide in the
presence of hydrogen. Appl Microbiol Biotechnol, 25(4): 384-391.
Kobayashi K & Akitake K (1975) Metabolism of chlorophenols in fish.
VIV Absorption, and excretion of phenol by goldfish. Bull Jpn Soc
Sci Fish, 41: 1271-1276.
Kobayashi K, Akitake H, & Kimura S (1976) Studies on the metabolism
of chlorophenols in fish. VI Turnover of absorbed phenol in
goldfish. Bull Jpn Soc Sci Fish, 42: 45-50.
Kobayashi K, Oshima Y, Hamada S, & Taguchi C (1987) Induction of
phenol-sulfate conjugating activity by exposure to phenols and
duration of its induced activity in short-necked clam. Bull Jpn Soc
Sci Fish, 53(11): 2073-2076.
Koike N, Haga S, Ubukata N, Sakurai M, Shimizu H, & Sato A (1988)
[Mutagenicity of benzene metabolites by fluctuation test.] Jpn. J.
Ind. Health 30(6): 475-480 (in Japanese, with English abstract).
Koop DR, Laethem CL, & Schnier GG (1989) Identification of
ethanol-inducible P450 isozyme 3a (P450IIE1) as a benzene and phenol
hydroxylase. Toxicol Appl Pharmacol, 98: 278-288.
Kordylewska A (1980) Developmental disturbances of Limnaea
stagnalis due to phenol intoxication. Acta Biol (Cracow) Ser Zool,
22(1): 105-130.
Koster HJ, Halsema J, Scholtens E, Knippers M, & Mulder GJ (1981)
Dose-dependent shifts in the sulfation and glucuronidation of
phenolic compounds in the rat in vivo and in isolated hepatocytes.
The role of saturation of phenolsulfotransferase. Biochem Pharmacol,
30(18): 2569-2575.
Koster HJ (1982) Sulfuration and glucuronidation in the rat in vivo
and in vitro. The balance between two enzyme systems competing for
a mutual substrate. University of Groningen (Thesis).
Kostoveckii YA & Zholdakova Z (1971) [On hygienic norm-setting in
waterbodies.] Gig i Sanit, 7: 7-10 (in Russian).
Krug M, Ziegler H, & Straube G (1985) Degradation of phenolic
compounds by the yeast Candida tropicalis HP-15: 1. Physiology and
growth and substrate utilization. J Basic Microbiol, 25(2): 103-110.
Kumar V & Mukerjee D (1988) Phenol and sulfide induced changes in
the ovary and liver of sexually maturing common carp, Cyprinus
carpio. Aquat Toxicol, 13(1): 53-60.
Kuwata K, Uebori M, & Yamazaki Y (1980) Determination of phenol in
polluted air as p-nitrobenzene azophenol derivative by reversed
phase high performance liquid chromatography. Anal Chem, 52(6):
857-860.
Kwasniewska K & Kaiser KLE (1983) Toxicities of selected phenols to
fermentative and oxidative yeasts. Bull Environ Contam Toxicol,
31(2): 188-194.
Laszczynska M, Barcew B, Wyczarska-Srak K, & Wozniak D (1983)
Reversibility of histoenzymatic changes caused by phenol in the
mouse kidney and liver. Folia Biol (Cracow), 31(1): 65-73.
Layiwola PJ & Linnecar DFC (1981) The biotransformation of 14C
phenol in some freshwater fish. Xenobiotica, 11(3): 167-171.
Leichnitz K ed. (1982) [Dräger test tubes phenol 5/a.] In: [Test
tube pocket book; testing of air and technical analysis of gas using
Dräger test tubes], 5th ed. Lubeck, Drägerwerk AG, p 114 (in
German).
Leider M & Moser HS (1961) Toxicology of topical dermatology
preparations. Arch Dermatol, 83: 928-929.
Leuenberger C, Ligocki MP, & Pankow JF (1985) Trace organic
compounds in rain: 4. Identities, concentrations, and scavenging
mechanisms for phenols in urban air and rain. Environ Sci Technol,
19(11): 1053-1058.
Lewin JF & Cleary WT (1982) An accidental death caused by the
absorption of phenol through skin. A case report. Forensic Sci Int,
19(2): 177-179.
Lewis MA (1983) Effect of loading density on the acute toxicities of
surfactants, copper and phenol to Daphnia magna Straus. Arch
Environ Contam Toxicol, 12: 51-55.
Liao JTF (1980) Investigations of phenol tissue distribution, plasma
protein binding, and liver subcellular fraction affinity. Diss Abstr
Int, B41(5): 1736.
Liao JTF & Oehme FW (1980) Literature reviews of phenolic compounds.
I. Phenol. Vet Hum Toxicol, 22: 160-164.
Liao JTF & Oehme FW (1981a) Tissues distribution and plasma protein
binding of (14C) phenol in rats. Toxicol Appl Pharmacol, 57(2):
220-225.
Liao JTF & Oehme FW (1981b) Plasma protein binding of phenol in dogs
and rats as determined by equilibrium dialysis and ultrafiltration.
Toxicol Appl Pharmacol., 57(2): 226-230.
Lister J (1867; republished 1937) On a new method of treating
compound fracture, abcess, etc., with observations on the condition
of suppuration. Med Classics, 2: 28-71.
Luten JB, Ritskes JM, & Weseman JM (1979) Determination of phenol,
guaiacol and 4-methylguaiacol in wood smoke and smoked fish-products
by gas-liquid chromatography. Z Lebensm.unters Forsch, 168: 289-292.
McFarlane JC, Pfleeger T, & Fletcher J (1987) Transpiration effect
on the uptake and distribution of bromacil, nitrobenzene and phenol
in soybean plants. J Environ Qual, 16(4): 372-376.
McGregor DB, Brown A, Cattanach P, Edwards I, McBride D, Riach C, &
Caspary WJ (1988) Responses of the L5178Y tk+/tk- mouse lymphoma
cell forward mutation assay: III. 72 Coded chemicals. Environ Mol
Mutagen, 12: 85-154.
Malcolm AR, Mills LJ, & Trosko JE (1985) Effects of ethanol, phenol,
formaldehyde, and selected metabolites on metabolic cooperation
between Chinese hamster V79 lung fibroblasts. Carcinogenesis: A
compr survey, 8: 305-318.
Mayes ME, Alexander HC, & Dill DC (1982) A study to assess the
influence of age on the response of Fathead minnows in static acute
toxicity tests. Bull Environ Contam Toxicol, 31(2) 139-147.
Merliss RR (1972) Phenol marasmus. J Occup Med, 14: 55-56.
Miksch K & Schürmann B (1988) [Toxicity assessment of zinc sulphate,
copper sulphate and phenol by different methods.] Z Wasser Abwasser
Forsch, 21: 193-198 (in German with English abstract).
Miller DL & Ogilvie DM (1975) Temperature selection in brook trout
( Salvelinus fontinalis) following exposure to DDT, PCB or phenol.
Bull Environ Contam Toxicol, 14: 545-551.
Minor JK & Becker BA (1971) A comparison of the teratogenic
properties of sodium salycate, sodium benzoate and phenol. Abstracts
of the 10th Annual Meeting of the Society of Toxicology No. 40.
Toxicol Appl Pharmacol, 19: 373.
Mitchell JR (1972) Mechanism of benzene-induced aplastic anaemia.
Fed Proc, 30: 561.
Model A (1889) Poisoning with concentrated phenol in a child
suffering from diphtheria. Ther Mon.heft, 3: 483-485.
Mogilnicka EM & Piotrowski JK (1974) [The exposure test for phenol
in the light of field study.] Med Prac, 25: 137-141 (in Hungarian).
Morimoto K & Wolff S (1980) Increase of sister chromatid exchanges
and perturbations of cell division kinetics in human lymphocytes by
benzene metabolites. Cancer Res, 40: 1189-1193.
Morimoto K, Wolff S, & Koizumi A (1983) Induction of sister
chromatid exchanges in human lymphocytes by microsomal activation of
benzene metabolites. Mutat Res, 119(3-4): 355-360.
Morrison JE, Matthews D, Washington R, Fennessey PV, & Harrison M
(1991) Phenol motor point blocks in children: Plasma concentrations
and cardiac dysrhythmias. Anesthetiology, 75: 359-362.
Mukhitov B (1964) The effect of low phenol concentrations on the
organism of man or animals and their hygienic evaluation. In: Levine
BS ed. USSR literature on air pollution and related occupational
diseases. Springfield, Virginia, US Department of Commerce, National
Technical Information Service, pp 185-199 (NTIS/PB 64-11574).
Murphy JC, Osterberg RE, Seabaugh UM, & Bierbower GW (1982) Ocular
irritancy responses to various pHs of acids and bases with and
without irrigation. Toxicology, 23(4): 281-291.
Müting D, Keller HE, & Kraus W (1970) Quantitative colorimetric
determination of free phenols in serum and urine of healthy adults
using modified diaro-reactions. Clin Chem Acta, 27: 177-180.
Nagel R (1983) Species differences, influence of dose and
application on biotransformation of phenol in fish. Xenobiotica,
13(2): 101-106.
Nagel R & Urich K (1983) Quinol sulfate, a new conjugate of phenol
in goldfish. Xenobiotica, 13(2): 97-100.
Nagel R, Adler HJ, & Rao TK (1982) Induction of filamentation by
mutagens and carcinogens in a lon mutant of Escherichia coli.
Mutat Res, 105: 309-312.
Namkoong W, Loehr RC, & Malina JF Jr (1989) Effects of mixture and
acclimation on removal of phenolic compounds in soil. J Water Pollut
Control Fed, 61(2): 242-250.
Narotsky MG & Kavlock RJ (1993) A multidisciplinary approach to
toxicological screening. II. Development toxicity. Research Triangle
Park, North Carolina, US Environmental Protection Agency, Health
Effects Research Laboratory (Report No. MS 91-237).
Nathan PW (1959) Intrathecal phenol to relieve spasticity in
paraplegia. Lancet, 2: 1099.
NCI (1980) Bioassay of phenol for possible carcinogenicity.
Bethesda, Maryland, US Department of Health Services, National
Cancer Institute (Technical Report Series No. NCI-CG-TR-203).
Neuhauser EF, Durking PR, Malecki MR, & Anatra M (1986) Comparative
toxicity of ten organic chemicals to four earthworm species. Comp
Biochem Physiol, C83: 197-200.
Nicola RM, Branchflower R, & Pierce D (1987) Chemical contaminants
in bottonfish. J Environ Health, 49: 342-347.
Niessen R, Lenoir D, & Boule P (1988) Phototransformation of phenol
induced by excitation of nitrate ions. Chemosphere, 17(19):
1977-1984.
NIOSH (1984) NIOSH manual of analytical methods, 3rd ed. Cincinnati,
Ohio, National Institute for Occupational Safety and Health.
NIOSH (1985a) NIOSH pocket guide to chemical hazards. Cincinnati,
Ohio, National Institute for Occupational Safety and Health.
NIOSH (1985b) Method 8305. NIOSH manual of analytical methods, 3rd
ed. Cincinnati, Ohio, Institute for Occupational Safety and Health.
Oehme FW (1969) A comparative study of the biotransformation and
excretion of phenol. A doctoral dissertation. University of
Missouri.
Oehme FW & Davis LE (1970) The comparative toxicity and
biotransformation of phenol. Toxicol Appl Pharmacol, 17: 283.
Ogata M, Yamasaki Y, & Kawai T (1986) Significance of urinary phenyl
sulfate and phenyl glucuronide as indices of exposure to phenol. Int
Arch Occup Environ Health, 58: 197-202.
Oglesbi LA, Ebron-McCoy MT, Logson TR, Copeland F, Beyer PE, &
Kavlock RJ (1992) in vitro embryotoxicity of a series of
para-substituted phenols: structure, activity, and correlation with
in vivo data. Teratology, 45: 11-33.
Ohtsuji H & Ikeda M (1972) Quantitative relationship between
atmospheric phenol vapour and phenol in the urine of employees in
Bakelite factories. Br J Ind Med, 29: 70-73.
Oksama M & Kristofferson R (1979) The toxicity of phenol to
Phoxinus phoxinus, Gammarus ducbeni and Mesidota entomon in
brackish water. Ann Zool Fenn, 16(3): 209-216.
Olowska L, Oledzka-Slotwinska H, Lazczymska M, & Wozniak W (1980)
Effect of phenol on hydrolytic and respiratory enzymes of the small
intestinal mucosa of white mouse. Folia Morphol (Warsaw), 39(2):
149-158.
Oomens AC & Schuurhuis FG (1983) A method for the collection and
determination of phenol and bisphenol A in air. Int Arch Occup
Environ Health, 52(1): 43-48.
Oris JT, Winner RW, & Moore MV (1991) A four-day survival and
reproduction toxicity test for Ceriodaphnia dubia. Environ Toxicol
Chem, 10: 217-224.
Painter RB & Howard R (1982) The Hela DNA-synthesis inhibition test
as a rapid screen for mutagenic carcinogens. Mutat Res, 92(1-2):
427-437.
Paradowski M, Anderzeiewski S, Zauisza B, & Wybrak-Wrobel T (1981)
Excretion of phenol and p-cresol in urine as an element of
evaluation of professional exposure to benzene and phenol in
employees of refining and petrochemicals works in Plock. Przeglad
Lekarski, 38(9): 663-666.
Paris DF, Wolfe NL, & Steen WC (1982) Structure-activity
relationships in microbial transformation of phenols. Appl Environ
Microbiol, 44: 153-158.
Parke DV & Williams RT (1953) Studies in detoxification. 54. The
metabolism of benzene: (a) The formation of phenylglucuronide and
phenylsulfuric acid from 14C benzene. (b) The metabolism of 14C
phenol. Biochem J, 55: 337-340.
Parker JG (1984) The effects of selected chemicals and water quality
on the marine polychaete Ophryotrocha diadema. Water Res, 18(7):
865-868.
Pashin YV & Bakhitova LM (1982) Mutagenicity of benzo(a)pyrene and
the antioxidant phenol at the HGPRT locus of V79 Chinese Hamster
cells. Mutat Res, 104(6): 389-393.
Patrick E, Maibach HI, & Burkhalter A (1985) Mechanisms of
chemically induced skin irritation. I. Studies of time course, dose
response, and components of inflammation in the laboratory mouse.
Toxicol Appl Pharmacol, 81: 476-490.
Pekari K, Vainotalo S, Heikkila P, Palotie A, Luotamo M, & Riihimaki
V (1992) Biological monitoring of occupational exposure to low
levels of benzene. Scand J Work Environ Health, 18: 317-322.
Pellack-Walker P & Blumer JL (1986) DNA damage in L5178YS cells
following exposure to benzene metabolites. Mol Pharmacol, 30: 42-47.
Pellack-Walker P, Walker JK, Evans HH, & Blumer JL (1985)
Relationship between the oxidation potential of benzene metabolites
and their inhibitory effect on DNA synthesis in L5178YS cells. Mol
Pharmacol, 28: 560-566.
Petrov VI (1960) [Cases of phenol poisoning during coke slaking with
phenol water.] Gig i Sanit, 25: 60-62 (in Russian).
Phipps GL, Holcombe GW, & Fiandt JT (1981) Acute toxicity of phenol
and substituted phenols to the fathead minnow. Bull Environ Contam
Toxicol, 26: 585-593.
Pickering GH & Henderson C (1966) Acute toxicity of some important
petrochemicals to fish. J Water Pollut Control Fed, 38: 1419-1429.
Pierce WM & Nerland DE (1988) Qualitative and quantitative analyses
of phenol, phenylglucuronide, and phenylsulfate in urine and plasma
by gas chromatography mass spectrometry. J Anal Toxicol, 12:
344-347.
Piotrowski JK (1971) Evaluation of exposure to phenol, absorption of
phenol vapour in the lungs and through the skin and excretion of
phenol in urine. Br J Ind Med, 28: 172-178.
Podolak GE, McKenzie RM, Rinehart DS, & Mazur JF (1981) A rapid
technique for collection and analysis of phenol vapours. Am Ind Hyg
Assoc J, 42(10) 734-738.
Poirier MC, De Cicco BT, & Lieiberman MW (1975) Non-specific
inhibition of DNA repair synthesis by tumour promotors on human
diploid fibroblasts damaged with N-acetoxy-acetyl-aminofluorene.
Cancer Res, 35: 1392-1397.
Pool BL & Lin PZ (1982) Mutagenicity testing in the Salmonella
typhimurium assay of phenolic compounds and phenolic fractions
obtained from smoke-house condensates. Food Chem Toxicol, 22:
383-391.
Potthast K (1976) Determination of phenols in smoked meat products.
Advances in Smoking of Foods, Intern. Joint IUPAC/IUFoSt Symp.,
Warschau, pp. 39-44.
Potthast K (1982) Dark smoking at high smokehouse temperatures.
Fleischwirtschaft, 62: 1578-1582.
Powell GM, Miller JJ, Olavesen AH, & Curtis CG (1974) Liver as a
major organ of phenol detoxification? Nature (Lond), 252: 234-235.
Pratt RM & Willis WD (1985) in vitro screening assay for
teratogens using growth inhibition of human embryonic cells. Proc
Natl Acad Sci (USA), 82: 5791-5794.
Price KS, Waggy GT, & Conway RA (1974) Brine shrimp bioassay and
seawater BOD of petrochemicals. J Water Pollut Control Fed, 46:
63-76.
Pullin TG, Pinkerton MN, Johnson RV, & Kilian DJ (1978)
Decontamination of the skin of swine following phenol exposure: A
comparison of the relative efficacy of water versus polyethylene
glycol/industrial methylated spirits. Toxicol Appl Pharmacol, 43:
199-206.
Quebbemann AJ & Anders MW (1973) Renal tubular conjugation and
excretion of phenol and p-nitrophenol in the chicken: differing
mechanism of renal transfer. J Pharmacol Exp Ther, 184(3): 695-708.
Ramanathan M (1984) Water pollution. In: Mark HF, Othmer DF,
Overberger CG, Seaborg GT, & Grayson M ed. Kirk-Othmer encyclopedia
of chemical technology, 3rd ed. New York, Chichester Brisbane,
Toronto, John Wiley & Sons, vol 24, p 299.
Ramli JB & Wheldrake JF (1981) Phenol conjugation in the desert
hopping mouse, Notomys alexis. Comp Biochem Physiol, C69(2):
379-381.
Rao RSV & Nath KJ (1983) Biological effect of some poisons on
Cantocamptus (Crustacea). Int J Environ Stud, 21(3-4): 271-275.
Rao, PS, Durve US, Khangarot BS, & Srekhawat SS (1983) Acute
toxicity of phenol, pentachlorophenol and sodium pentachlorophenate
to a freshwater ostracod Cypris subglubosa. Acta Hydrochim
Hydrobiol, 11(4): 457-465.
Rapson WH, Nazar MA, & Butsky V (1980) Mutagenicity produced by
aqueous chlorination of organic compounds. Bull Environ Contam
Toxicol, 24: 590-596.
Ravichandran S & Anantharaj B (1984) Effect of phenol on the
phosphomonoesterases and ATPase activity in the fish Sarotherodon
mossambicus (Peters) in saline waters. Proc Indian Acad Sci (Anim
Sci), 93(6): 557-563.
Razani H, Nanba K, & Murachi S (1986a) Acute toxic effect of phenol
on zebra fish Brachydanio rerio. Bull Jpn Soc Sci Fish, 52(9):
1547-1552.
Razani H, Nanba K, & Murachi S (1986b) Chronic toxic effect of
phenol on zebra fish Brachydanio rerio. Bull Jpn Soc Sci Fish,
52(9): 1553-1558.
Rea WJ, Pan Y, Laseter JL, Johnson AR, & Fenyves EJ (1987) Toxic
volatile organic hydrocarbons in chemically sensitive patients. Clin
Ecol, 5(2): 70-74.
Reid GG, Ketterer PJ, Mawkinney H, & Glover P (1982) Exposure to
phenol and endrin as a cause of skin ulcerations and nervous signs
in pigs. Aust Vet J, 59: 160.
Reynolds JH, Middlebrooks EJ, Porcella WB, & Grenney WJ (1978)
Comparison of semi-continuous and continuous-flow bioassay. Progress
in Water Technology, 9(4): 897-909.
RIVM (1986) [Criteria Document: Phenol]. Bilthoven, The Netherlands,
National Institute of Public Health and Environmental Protection
(Document No. 738513002) (in Dutch; toxicology also available in
English).
Roberts MS, Anderson RA, & Swarlrich J (1977) Permeability of human
epidermis to phenolic compounds. J Pharm Pharmacol, 29(11): 677-683.
Roberts MS, Anderson RA, Swarlrich J, & Moore DE (1978) The
percutaneous absorption of phenolic compounds: the mechanism of
diffusion across the stratum corneum. J Pharm Pharmacol, 30:
486-490.
Rogers SCF, Burrows D, & Neill D (1978) Percutaneous absorption of
phenol and methyl alcohol in magenta paint B.C.P. Br J Dermatol, 98:
559-560.
Rozich AF & Colvin RJ (1986) Effects of glucose on phenol
biodegradation by heterogenous populations. Biotechnol Bioeng,
29(7): 965-971.
Rubin HE & Alexander M (1983) Effect of nutrients on the rates of
mineralization of trace concentrations of phenol and
p-nitrophenol. Environ Sci Technol, 17: 104-107.
Ruedemann R & Deichmann WB (1953) Blood phenol level after topical
application of phenol containing preparations. J Am Med Assoc, 152:
506-509.
Ruessink RG & Smith LL (1975) The relationship of the 96 h LC50 of
the lethal threshold concentration of hexavalent chromium, phenol
and sodium pentachlorophenate for fathead minnows ( Pimephales
promelas Rafinesque). Trans Am Fish Soc, 3: 570.
Rusch HP, Bosch MS, & Boutwell RK (1955) The influence of irritants
on mitotic activity and tumour formation in mouse epidermus.
International Union against Cancer Newsletter, 11: 699-703.
Rushmore TH, Snyder R, & Kalf G (1984) Covalent binding of benzene
and its metabolites to DNA in rabbit bone marrow mitochondria in
vitro. Chem-Biol Interact, 49: 133-154.
Russell JW (1975) Analysis of air pollutants using sampling tubes
and gas chromatography. Environ Sci Technol, 9(13): 1175-1178.
Ryser S & Ulmer G (1980) Etude de la pollution gazeuse par les
résines synthétiques utilisées en fonderie. Fonderie, 35: 313-324.
Salamon MH & Glendenning OM (1957) Tumour promotion in mouse skin by
sclerosing agents. Br J Cancer, 11: 434-444.
Sandage C (1961) Tolerance criteria for continuous inhalation
exposure to toxic material. I. Effects on animals of 90-day exposure
to phenol, CCl4, and a mixture of indole, skatole, H2S, and methyl
mercaptan. Dayton, Ohio, Wright-Patterson Air Force Base, US Air
Force Systems Command, Aeronautical Systems Division (ASD Technical
Report 61-519 (I); NTIS AD-268783).
Sanitary Epidemiological Station, Katowice (1991) Air pollution in
the Katowice voivodeship in 1988-1990. Katowice, Poland, Regional
Sanitary Epidemiological Station.
Sawahata T & Neal RA (1983) Biotransformation of phenol to
hydroquinone and catechol by rat liver microsomes. Mol Pharmacol,
23(21): 453-460.
Schaumburg HH, Byck R, & Weller RO (1970) The effect of phenol on
peripheral nerve. A histological and electrophysiological study. J
Neuropathol Exp Neurol, 29: 615-630.
Schlicht MP, Moser VC, Sumrell BM, Berman E, & MacPhail RC (1992)
Systemic and neurotoxic effects of acute and repeated phenol
administration (Abstract No. 1047). Toxicologist, 12, 274.
Schmidt R & Maibach H (1981) Immediate and delayed onset "skip area"
dermatitis presumed secondary to topical phenol exposure. Contact
Dermatitis, 7(4): 199-202.
Schmieder PK & Henry TR (1988) Plasma binding of 1-butanol, phenol,
nitrobenzene and pentachlorophenol in the rainbow trout and rat: a
comparative study. Comp Biochem Physiol, C91(2): 413-418.
Schoenberg JB & Mitchell CA (1975) Airway disease caused by phenolic
(phenol-formaldehyde) resin exposure. Arch Environ Health, 30:
574-577.
Schussler OF & Stern MA (1911) Gangrene of finger caused by five
percent phenol ointment. J Am Med Assoc, 57: 628.
Schütz A & Wolfe D (1980) [Gases and vapours in foundries;
measurement; evaluation; control.] Giesserei, 67: 68-73 (in German).
Seidel HJ, Barthel E, Schäfer F, Schad H, & Weber L (1991) Action of
benzene metabolites on murine hematopoietic colony-forming cells in
vitro. Toxicol Appl Pharmacol, 111, 128-131.
Selvakumar A & Hsieh HN (1988) Competitive adsorption of organic
compounds by microbial biomass. J Environ Sci Health, A23(8):
729-744.
Schlicht MP, Moser VP, Sumrell BM, Berman E, & MacPhail RC (1992)
Systematic and neurotoxic effects of acute and repeated phenol
administration. Toxicologist, 12(1): 274.
Scott HD, Wolf DC, & Lavy TL (1982) Apparent absorption and
microbial degradation of phenol by soil. J Environ Qual, 11:
107-112.
Shafer WE & Schönherr J (1985) Accumulation and transport of phenol,
2-nitrophenol, and 4-nitrophenol in plant cuticles. Ecotoxicol
Environ Saf, 10(2): 239-252.
Shimp RJ & Pfaender FK (1987) Effect of adaptation to phenol on
biodegradation of monosubstituted phenols by aquatic microbial
communities. Appl Environ Microbiol, 53(7): 1496-1499.
Shimp RJ & Young RL (1987) Availability of organic chemicals for
biodegradation in settled bottom sediments. Ecotoxicol Environ Saf,
15(1): 31-45.
Shirkey RJ, Kao J, Fry JR, & Bridges JW (1979) A comparison of
xenobiotic metabolism in cells isolated from rat liver and small
intestinal mucosa. Biochem Pharmacol, 28: 1461-1466.
Skare JA & Schrotel KR (1984) Alkaline elution of rat testicular
DNA: Detection of DNA strand breaks after in vivo treatment with
chemical mutagens. Mutat Res, 130: 283-294.
Skvortsova NN & Vysochina IV (1976) Changes in biochemical and
physiological indices in animals produced by the combined effect of
benz[a]pyrene and phenol. Environ Health Perspect, 13: 101-106.
Slabbert JL (1986) Improved bacterial growth test for rapid water
toxicity screening. Bull Environ Contam Toxicol, 37: 565-569.
Smart AC & Zannoni VC (1984) D.T. Diaphorase and peroxidase
influence the covalent binding of the metabolites of phenol, the
major metabolite of benzene. Mol Pharmacol, 26: 105-111.
Smith MC (1984) Histological findings following intrathecal
injections of phenol solutions for relief of pain. Br J Anaesth, 36:
387-406.
Solbé JFLG, Cooper VA, Willis CA, & Mallett MJ (1985) Effects of
pollutants in fresh waters on European non-salmoid fish. I:
Non-metals. J Fish Biol, 27(Suppl A): 197-207.
Solliman T (1957) A manual of pharmacology and its applications to
therapeutics and toxicology, 8th ed. New York, Philadelphia, W.B.
Saunders Company, pp 805-809.
Steele RH & Wilhelm DL (1966) The inflammatory reaction in chemical
injury. I. Increased vascular permeability and erythema induced by
various chemicals. Br J Exp Pathol, 47: 612-623.
Southworth GR, Herbes SE, Franco PJ, & Giddings JM (1985)
Persistence of phenols in aquatic microcosms receiving chronic
inputs of coal-derived oil. Water Air Soil Pollut, 24(3): 283-296.
Spicer CW, Riggin RM, Holdren MW, DeRoos FL, & Lee RN (1985)
Atmospheric reaction products from hazardous air pollutant
degradation. Research Triangle Park, North Carolina, US
Environmental Protection Agency (EPA-600/3-85-028; NTIS
PB85-185841/XAB).
Spoelstra SF (1978) Degradation of tyrosine in anaerobically stored
piggery wastes and in pig feces. Appl Environ Microbiol, 36:
631-638.
Stajduhar-Caric Z (1968) Acute phenol poisoning. J Forensic Med, 15:
41-42.
Stephenson RR (1983) Effects of water hardness, water temperature
and size of the test organism on the susceptibility of the
freshwater shrimp Gammarus pulex (L.) to toxicants. Bull Environ
Contam Toxicol, 31(4): 459-466.
Sturtevant MJ (1952) Studies on the mutagenicity of phenol. Journal
of Heredity, 43: 217-219.
SRI (1982) Economics handbook. Stanford, Stanford Research
Institute.
Subrahmanyam VV & O'Brien PJ (1985) Peroxidase-catalyzed binding of
uniformly carbon-14-labeled phenol to DNA. Xenobiotica, 15(10):
859-872.
Subrahmanyam V, Sadler A, Suba E, & Ross D (1989) Stimulation of in
vitro bioactivation of hydroquinone by phenol in bone marrow
cells. Drug Metab Dispos, 17(3): 348-349.
Subrahmanyam VV, Kolachana P, & Smith MT (1991) Hydroxylation of
phenol to hydroquinone catalyzed by a human
myeloperoxidase-superoxide complex: possible implications in
benzene-induced myelotoxicity. Free Radic Res Commun, 15, 285-296.
Suzuki T & Kisara K (1985) Enhancement of phenol-induced tremor
caused by central monoamine depletion. Pharmacol Biochem Behav, 22:
153-155.
Takemori AE & Glowacki GA (1962) Studies on glucuronide synthesis in
rats chronically treated with morphine and phenol. Biochem
Pharmacol, 11: 867-870.
Tetra Tech. Inc. (1986) Development of sediment quality values for
Pudget Sound (unnamed report prepared for Pudget Sound Dredged
Disposal Analysis and Pudget Sound Estuary Program). Bellevue,
Washington, Tetra Tech. Inc.
Thompson ED & Gibson DP (1984) A method for determining the maximum
tolerated dose for acute in vivo cytogenetic studies. Food Chem
Toxicol, 22(8): 665-676.
Thornton-Manning JR, Jones DD, & Federle TW (1987) Effects of
experimental manipulation of environmental factors on phenol
mineralization in soil. Environ Toxicol Chem, 6: 615-621.
Tibbles BJ & Baecker AAW (1989) Effects and fate of phenol in
simulated landfill sites. Microb Ecol, 17(2): 201-206.
TNO (1978) [Emissions during various processes in foundries.] Delft,
The Netherlands, Dutch Organization for Applied Scientific Research
(TNO) (Report No. M78/40/07536/Jul/SCI) (in Dutch).
Tonapi GT & Varghese G (1987) Cardio-physiological responses of some
selected cladocerans to three common pollutants. Arch Hydrobiol,
110(1): 59-65.
Toth L (1982) [Transfer of phenols from tobacco smoke into meat
products.] Fleischwirtschaft, 62: 1389-1402 (in German).
Tranvik L, Larsson P, Okla L, & Regnell O (1991) In situ
mineralization of chlorinated phenols by pelagic bacteria in lakes
of different humic content. Environ Toxicol Chem, 10: 195-200.
Truppman ES & Ellenby JD (1979) Major electrocardiograph changes
during chemical face peeling. Plast Reconstr Surg, 63: 44-48.
Tschech A & Fuchs G (1987) Anaerobic degradation of phenol by pure
cultures of newly isolated denitrifying pseudomonads. Arch
Microbiol, 148(3): 213-217.
Tunek A, Olofsson J, & Berlin M (1981) Toxic effects of benzene and
benzene metabolites on granulopoietic stem cells and bone marrow
cellularity in mice. Toxicol Appl Pharmacol, 59(1): 149-156.
Turtle WRM & Dolan T (1922) A case of rapid and fatal absorption of
carbolic acid through the skin. Lancet, 2: 1273-1274.
UBA (1981) [Air pollution control '81. Developments, state of the
art, tendencies.] Berlin, Federal Office for the Environment (in
German).
United Nations (1980) Yearbook or industrial statistics. Volume II:
Commodity product data. New York, United Nations.
Ursin C (1985) Degradation of organic chemicals at trace levels in
seawater and marine sediment. The effects of concentration of the
initial fractional turnover rate. Chemosphere, 14(10): 1539-1550.
US EPA (1980) Phenol. Ambient water quality criteria. Washington,
DC, US Environmental Protection Agency (EPA 440/5-80-066).
US EPA (1986a) Method 8010. Phenols. In: Test methods for evaluating
solid waste -Physical/chemical methods, 3rd ed. Washington, DC, US
Environmental Protection Agency, Office of Solid Waste and Emergency
Response, pp 8040/1-8050/17 (EPA Report No. SW-846).
US EPA (1986b) Method 8250. Gas chromatography/mass spectrometry for
semi-volatile organics: Packed column technique. In: Test methods
for evaluating solid waste -Physical/chemical methods, 3rd ed.
Washington, DC, US Environmental Protection Agency, Office of Solid
Waste and Emergency Response, pp 8250/1-8250/32 (EPA Report No.
SW-846).
US EPA (1986c) Method 8270. Gas chromatography/mass spectrometry for
semi-volatile organics: Capillary column technique. In: Test methods
for evaluating solid waste -Physical/chemical methods, 3rd ed.
Washington, DC, US Environmental Protection Agency, Office of Solid
Waste and Emergency Response, pp 8270/1-8270/34 (EPA Report No.
SW-846).
US EPA (1992) Phenol - Drinking water health advisory. Washington,
DC, US Environmental Protection Agency, Office of Water.
Van Duuren BL & Goldschmidt BM (1976) Carcinogenic and
tumour-promoting agents in tobacco carcinogenesis. J Natl Cancer
Inst, 56: 1237-1242.
Van Duuren BL, Sivah A, Langretti J, & Goldschmidt BM (1968)
Initiators and promotors in tobacco carcinogenesis. Natl Cancer Inst
Monogr, 28: 173-180.
Van Duuren BL, Blazej T, Goldschmidt BM, Katz C, Melchionne S, &
Sivah A (1971) Carcinogenesis studies on mouse skin and inhibition
of tumour induction. J Natl Cancer Inst, 46: 1039-1044.
Van Duuren BL, Katz C, & Goldschmidt BM (1973) Cocarcinogenesis
agents in tobacco carcinogenesis. J Natl Cancer Inst, 51: 703-705.
Van Gemert LJ (1984) Compilation of odour threshold values in air:
Supplement V. Zeist, The Netherlands, CIVO-TNO (Report No. 84.220).
Van Gemert LJ & Van Nettenbreijer (1977) Compilation of odour
threshold values in air and water. Zeist, The Netherlands, CIVO/RID.
Veith GD & Kosian P (1983) Estimating bioconcentration potential
from octanol/water partition coefficients. In: Physical behaviour of
PCB's in the Great Lakes. Ann. Arbor, Michigan, Ann Arbor Science
Publishers, Inc., pp 269-282.
Verma SR, Rani S, Tyagi AK, & Dalela RC (1980) Evaluation of acute
toxicity of phenol and its chloro- and nitro-derivates to certain
teleosts. Water Pollut, 14: 95-102.
Vernot EH, MacEwen JD, Haun CC, & Kinkead ER (1977) Acute toxicity
and skin corrosion data for some organic and inorganic compounds and
aqueous solutions. Toxicol Appl Pharmacol, 42: 417-423.
Veronesi B, Padilla S, & Newland D (1986) Biochemical and
neuropathological assessment of triphenyl phosphite in rats. Toxicol
Appl Pharmacol, 83: 203-210.
Verschueren K (1983) Handbook of environmental data on organic
chemicals, 2nd ed. New York, Van Nostrand Reinhold Company.
Volskay VT Jr & Grady CPL (1988) Toxicity of selected RCRA compounds
to activated sludge microorganisms. J Water Pollut Control Fed,.
60(10): 1850-1856.
Von Oettingen WF (1949) Phenol and its derivatives. The relation
between their chemical constitution and their effect on the
organisms. Washington, DC, National Institutes of Health Bulletin.
Von Oettingen WF & Sharples NE (1946) The toxicity and toxic
manifestation of 2,2 bis-(p-chlorophenyl)-1,1,1-trichloroethane
(DDT) as influenced by chemical changes in the molecule. J Pharmacol
Exp Ther, 88: 400-413.
Wajon JE, Rosenblatt DH, & Burrows EP (1982) Oxidation of phenol and
hydroquinone by chlorine dioxide. Environ Sci Technol, 16: 396-402.
Wang WC (1986) Comparative toxicology of phenolic compounds using
root elongation method. Environ Toxicol Chem, 5(10): 891-896.
Wang YT, Suidan MT, Pfeiffer JT, & Najm I (1988) Effects of some
alkyl phenols on methanogenic degradation of phenol. Appl Environ
Microbiol, 54(5): 1277-1279.
Wangenheim J & Bolcsfoldi G (1988) Mouse lymphoma L5178Y thymidine
kinase locus assay of 50 compounds. Mutagenesis, 3(3): 193-205.
Warner MA & Harper JV (1985) Cardiac dysrhythmias associated with
chemical peeling with phenol. Anesthesiology, 62: 366-367.
Weast RC, ed. (1987) Handbook of chemistry and physics, 68th ed.
Boca Raton, Florida, CRC Press, Inc., p C-406.
Weitering JG, Krijgsheld KR, & Mulder GJ (1979) The availability of
inorganic sulfate as a rate limiting factor in the sulfate
conjugation or xenobiotics in the rat? Biochem Pharmacol, 28:
757-762.
Wheldrake JF, Baudinette RV, & Hewitt S (1978) The metabolism of
phenol in a desert rodent Notomys alexis. Comp Biochem Physiol,
C61: 103-107.
Wiggins BA & Alexander M (1988) Role of chemical concentration and
second carbon sources in acclimation of microbial communities for
biodegradation. Appl Environ Microbiol, 54(11): 2803-2807.
Wilcosky TC & Tyroler HA (1983) Mortality from heart disease among
workers exposed to solvents. J Occup Med, 25: 879-885.
Wilcosky TC, Checkoway H, Marshall EG, & Tyroler HA (1984) Cancer
mortality and solvent exposures in the rubber industry. Am Ind Hyg
Assoc J, 45: 809-811.
Wild D, Eckhardt K, Gocke E, & King MT (1980) Comparative results on
short-term in vitro and in vivo mutagenicity tests obtained with
selected environmental chemicals. Berlin, Heidelberg, New York,
Springer Verlag, pp 170-178.
Williams RT (1938) Studies in detoxification. I. The influence of
(a) dose and (b) o, m, p, substitution on the sulfate detoxification
of phenol in the rabbit. Biochem J, 32: 878-887.
Williams RT (1959) Detoxication mechanisms, 2nd ed. New York,
Chichester, Brisbane, Toronto, John Wiley and Sons, pp 237-295.
Windus-Podehl G, Lyftog C, Zieve L, & Brunner G (1983)
Encephalopathic effect in rats. J Lab Clin Med, 101(4): 586-592.
Winkler HD (1981) [Methods for reduction of emission of air
pollutants in production of chipboards.] Gesundheits-Ing, 102:
295-299 (in German).
Wood KA (1978) The use of phenol as a neurolytic agent: a review.
Pain, 5: 205-225.
Woodruff RC, Mason JM, Valencia R, & Zimmering S (1985) Chemical
mutagenesis testing in Drosophila. V. Results of 53 coded compounds
tested for the National Toxicology Program. Environ Mutagen, 7:
677-702.
Wynder EL & Hoffmann D (1961) A study of tobacco carcinogenesis -
the role of the acid fractions as promoters. Cancer, 14: 1306-1315.
Wysowski DK, Flynt JW, Goldfield M, Altman R, & Davis AT (1978)
Epidemic neonatal hyperbilirubinemia and use of a phenolic
disinfectant detergent. Pediatrics, 61(2): 165-170.
Young LY & Rivera MD (1985) Methanogenic degradation of 4 phenolic
compounds. Water Res, 19(10): 1325-1332.
Zavorovskaya NA & Nekhorosheva EV (1981) [Spectrometric
determination of phenol and aniline in air and solutions simulating
biological media.] Zh Anal Khim, 36(9): 1808-1812 (in Russian).
RESUME
1. Identité, propriétés physiques et chimiques et méthodes
d'analyse
Le phénol se présente sous la forme d'un solide cristallin
blanc qui fond à 43 °C et se liquéfie par contact avec l'eau. Il
dégage une odeur âcre caractéristique et possède une saveur forte et
piquante. Il est soluble dans la plupart des solvants organiques; sa
solubilité dans l'eau est limitée à la température ambiante;
au-dessus de 68 °C il est entièrement soluble dans l'eau. Le phénol
est modérément volatil à la température ambiante. C'est un acide
faible, et, sous sa forme ionisée, il est très sensible aux
réactions de substitution électrophile et à l'oxydation.
On peut recueillir le phénol dans des échantillons prélevés
dans l'environnement par absorption dans une solution de soude ou
par adsorption sur un solide. La désorption s'effectue par
acidification, entraînement à la vapeur et extraction à l'éther (à
partir des solutions) ou par voie thermique ou en phase liquide
(lorsqu'il est adsorbé sur un solide). Les méthodes d'analyse les
plus importantes sont la chromatographie en phase gazeuse avec
détection par ionisation de flamme ou capture d'électrons ou encore
la chromatographie liquide à haute performance avec détection en
lumière ultra-violette. La limite de détection la plus basse qui ait
été signalée dans l'air est de 0,1 µg par m3. On peut doser le
phénol dans le sang et les urines; dans les échantillons d'urine, on
a fait état d'une limite de détection de 0.5 µg/litre.
2. Sources d'exposition humaine et environnementale
Le phénol est un constituant du goudron de houille et il se
forme au cours de la décomposition naturelle des substances
organiques. La majeure partie du phénol présent dans l'environnement
provient cependant de l'activité humaine. Les sources potentielles
en sont la production et l'utilisation, tel quel ou sous forme de
dérivés, en particulier de résines phénoliques et de caprolactame,
les gaz d'échappement, la combustion de bois de construction et la
fumée de cigarette. Une autre source potentielle est constituée par
la dégradation atmosphérique du benzène sous l'action de la lumière,
la présence de phénol dans le purin pouvant également fortement
contribuer à sa concentration dans l'atmosphère. Les dérivés du
benzène et du phénol peuvent, par conversion in vivo, constituer
une source d'exposition humaine endogène.
La production de phénol dans le monde s'est montrée
relativement constante pendant les années 1980, les Etats-Unis
d'Amérique en étant le premier producteur. Il est principalement
utilisé pour la fabrication des résines phénoliques, du bisphénol A
et de la caprolactame. On en connaît également un certain nombre
d'applications médicales et pharmaceutiques.
3. Transport, distribution et transformation dans l'environnement
Les principales émissions de phénol se produisent dans l'air.
La majeure partie du phénol présent dans l'atmosphère finit par être
dégradée par voie photochimique en dihydroxybenzènes, nitrophénols
et dérivés résultant de l'ouverture du cycle, avec une demi-vie
estimative de 4 à 5 heures. Une petite quantité est éliminée de
l'air par dépôt humide (pluie). Le phénol devrait présenter une
forte mobilité dans le sol mais son transport et sa réactivité
peuvent être affectés par le pH.
Le phénol présent dans l'eau et le sol peut être décomposé par
des réactions abiotiques ainsi que par l'activité microbienne en un
certain nombre de produits, dont les plus importants sont le dioxyde
de carbone et le méthane. La part des réactions biologiques dans la
décomposition globale du phénol dépend de nombreux facteurs tels que
la concentration, l'acclimatation, la température et la présence
d'autres composés.
4. Concentrations dans l'environnement et exposition humaine
On ne possède aucune donnée sur la concentration atmosphérique
du phénol. La concentration de fond est vraisemblablement inférieure
à 1 ng/m3. Les valeurs en milieu urbain et suburbain varient de
0,1 à 8 µg/m3 alors que dans les zones où prédominent les sources
de phénol (zones industrielles) les concentrations signalées peuvent
être jusqu'à 100 fois plus élevées. On a décelé du phénol dans l'eau
de pluie, les eaux superficielles et les eaux souterraines mais les
données sont très rares. On a fait état de concentrations élevées de
phénol dans des sédiments et des eaux souterraines par suite de
pollution industrielle.
Il peut y avoir exposition professionnelle au phénol lors de la
production de ce produit et de ses dérivés, lors de l'enduction avec
des résines phénoliques (industrie du bois et métallurgie) et lors
d'un certain nombre d'autres activités industrielles. La
concentration la plus élevée qui ait été signalée (jusqu'à 88
mg/m3) concernait des ouvriers de l'ex-URSS employés à
l'extinction du coke avec des eaux usées contenant du phénol. La
plupart des autres concentrations évoquées ne dépassaient 19
mg/m3.
En ce qui concerne la population dans son ensemble, c'est la
fumée de cigarette et les aliments fumés qui constituent les plus
importantes sources d'exposition au phénol, si l'on excepte
l'exposition par voie atmosphérique. L'exposition par l'eau de
boisson ou la consommation par inadvertance de produits alimentaires
contaminés devraient rester faibles; le phénol a en effet une odeur
et une saveur désagréables, ce qui dans la plupart des cas devrait
alarmer le consommateur.
5. Cinétique et métabolisme
Le phénol est facilement absorbé par toutes les voies
d'exposition. Après absorption, il se répartit rapidement dans
l'ensemble des tissus.
Une fois résorbé, il forme essentiellement des conjugués avec
l'acide glucuronique et l'acide sulfurique, et, dans une moindre
mesure, des hydroxylates avec le catéchol et l'hydroquinone. Il y a
également conjugaison avec les phosphates. La formation de
métabolites réactifs (4,4-biphénol et diphénoquinone) a été mise en
évidence lors d'études in vitro portant sur des neutrophiles et
des leucocytes humains activés.
La proportion relative de glucuronides et de sulfo-conjugués
varie avec la dose et l'espèce animale. Chez le rat, on a observé
qu'en augmentant la dose de phénol, la formation de sulfo-conjugués
l'emportait sur celle de glucuro-conjugués.
C'est essentiellement dans le foie, les poumons et au niveau de
la muqueuse gastro-intestinale que le phénol est métabolisé. Le rôle
relatif joué par ces divers tissus dépend de la voie
d'administration et de la dose.
Des études in vivo et in vitro ont montré que le phénol se
fixait aux protéines tissulaires et plasmatiques par liaison
covalente. Certains métabolites du phénol se lient également aux
protéines.
C'est principalement par la voie urinaire que la phénol est
excrété chez l'animal et l'homme. Le taux d'excrétion urinaire varie
selon la dose, la voie d'administration et l'espèce. Une faible
proportion est excrétée dans les matières fécales et l'air expiré.
6. Effets sur les animaux d'expérience et les systèmes
d'épreuve in vitro
Le phénol présente une toxicité aiguë modérée pour les
mammifères. La DL50 par voie orale varie chez les rongeurs de 300
à 600 mg de phénol/kg de poids corporel. La DL50 dermique varie
respectivement de 670 à 1400 mg/kg de poids corporel chez le rat et
le lapin et pour le rat, la CL50 à 8h. par voie respiratoire est
supérieure à 900 mg de phénol/m3. Après exposition aiguë, les
symptômes cliniques sont une hyperexcitabilité neuromusculaire, des
convulsions graves, une nécrose de la peau et des muqueuses de la
gorge et l'on note également des effets au niveau des poumons, des
fibres nerveuses, des reins, du foie et de la pupille (réflexe
photomoteur).
Les solutions de phénol sont agressives pour la peau et les
yeux. A l'état de vapeur, le phénol peut irriter les voies
respiratoires. On est fondé à croire que le phénol n'agit pas comme
sensibilisateur cutané.
Les effets les plus importants relevés lors d'études à court
terme sur l'animal consistaient en neurotoxicité, lésions hépatiques
et rénales, troubles respiratoires et retard de croissance. A des
doses orales quotidiennes de 40 mg/kg ou davantage on a observé des
effets néphrotoxiques chez le rat. Chez la même espèce, il y avait
une évidente hépatotoxicité aux doses supérieures ou égales à 100
mg/kg/jour. Lors d'une étude limitée de 14 jours sur des rats, on a
obtenu, pour la dose par voie orale sans effets nocifs observables,
une valeur de 12 mg/kg/jour, le critère retenu étant les effets sur
le rein. Dans cette expérience, il y avait encore inhibition du
myosis (réaction de l'iris à un stimulus lumineux) à la dose
quotidienne de 4 mg/kg; toutefois, l'importance médicale de cette
observation demeure incertaine. On a signalé la présence de
certaines altérations biologiques au niveau de la muqueuse
intestinale et des reins chez des souris recevant des doses
quotidiennes inférieures à 1 mg/kg, observation dont l'importance
toxicologique n'est pas non plus bien claire.
Il n'y a pas eu d'études satisfaisantes sur la toxicité du
phénol pour la fonction de reproduction. Toutefois, la toxicité du
phénol paraît se manifester par son action délétère sur le
développement du rat et de la souris. Lors de deux études au cours
desquelles des rats ont reçu des doses multiples de phénol, on a
obtenu, pour la dose sans effets nocifs observables, une valeur de
40 mg/kg/jour (pour la dose la plus faible sans effets nocifs
observables, cette valeur était de 53 mg/kg/jour) et de 60
mg/kg/jour respectivement (dans ce cas, la dose la plus faible sans
effets nocifs observables était de 120 mg/kg/jour). Chez la souris,
la dose sans effets nocifs observables était de 140 mg/kg/jour (dose
minimale sans effets nocifs observables: 280 mg/kg/jour).
La plupart des tests de mutagénicité bactérienne ont donné des
résultats négatifs. Cependant, des épreuves effectuées in vitro
sur des cellules mammaliennes ont révélé la présence de mutations,
de lésions chromosomiques et d'effets sur l'ADN. Le phénol est sans
effet sur la communication intercellulaire (mesurée par la
coopération métabolique) dans des cultures de cellules mammaliennes.
Un certain nombre d'études ont mis en évidence l'induction de
micro-noyaux dans des cellules de moelle osseuse murine. Toutefois,
les études sur la souris n'ont pas révélé la présence de
micro-noyaux à doses plus faibles.
Deux études de cancérogénicité ont été effectuées sur des rats
et des souris mâles et femelles à qui l'on administrait du phénol
mêlé à leur eau de boisson. On n'a observé d'affections malignes (à
savoir cancers médullaires de la thyroïde, leucémies) que chez les
rats mâles soumis à de faibles doses. On n'a pas effectué d'études
de cancérogénicité en bonne et due forme utilisant la voie
percutanée ou la voie respiratoire. Des études de cancérogénicité en
deux phases ont montré que le phénol pouvait se comporter comme un
agent tumoro-promoteur lorsqu'on l'appliquait à plusieurs reprises
sur la peau de la souris.
7. Effets sur l'homme
Des cas bien documentés d'exposition humaine au phénol par la
voie percutanée, buccale ou intraveineuse, ont donné lieu à
l'observation d'effets indésirables très divers. Il a été fait état
d'une irritation des voies gastro-intestinales après ingestion de
phénol. Après exposition de la peau, les effets observés localement
vont d'un blémissement cutané indolore ou d'un érythème à la
corrosion et à la nécrose profonde. Parmi les effets généraux, on a
noté les troubles suivants: arythmies cardiaques, acidose
métabolique, hyperventilation, détresse respiratoire, insuffisance
rénale aiguë, lésions rénales, urines foncées, méthémoglobinémie,
troubles neurologiques (notamment des convulsions), choc
cardio-vasculaire, coma et mort. La dose orale la plus faible qui
ait entraîné un décès humain était de 4,8 g; la mort est survenue
dans les 10 minutes.
Le risque d'intoxication par inhalation de vapeurs de phénol
est connu depuis longtemps, mais on n'a pas signalé de décès
consécutif à ce type d'accident. Les symptômes produits par
l'inhalation de phénol consistent notamment en anorexie, perte de
poids, maux de tête, vertiges, salivation et coloration foncée des
urines.
Le phénol n'est pas un agent sensibilisateur.
Le seuil olfactif pour l'homme serait 0,021 à 20 mg/m3 d'air.
Pour le phénol en solution aqueuse, on a fait état d'un seuil
olfactif de 9 mg/litre, le seuil gustatif étant de 0,3 mg/litre
d'eau.
On ne dispose pas de données suffisantes sur le pouvoir
cancérogène du phénol.
8. Effets sur les êtres vivants dans leur milieu naturel
Lors d'études portant sur une seule espèce de bactéries, on a
obtenu, pour la CE50 relative à l'inhibition de la croissance, des
valeurs allant de 244 à 1600 mg de phénol/litre. On a constaté que
le seuil de toxicité se situait à 64 mg de phénol/litre. Pour les
protozoaires et les champignons, les valeurs étaient du même ordre
que pour les bactéries; pour les algues elles étaient un peu
inférieures. Le phénol est toxique pour les organismes dulçaquicoles
supérieurs. Pour les crustacés et les poissons, les valeurs les plus
faibles de la CL50 ou de CE50 se situent entre 3 et 7 mg de
phénol/litre. Les données concernant la toxicité aiguë du phénol
pour les organismes marins sont comparables à celles dont on dispose
au sujet des organismes d'eau douce. Des études à long terme sur des
crustacés et diverses espèces de poissons ont révélé des différences
de sensibilité remarquables; c'est ainsi que les valeurs de la CL1
provenant d'épreuves sur des embryons et des larves de Salmo et de
Carassius se sont révélées très inférieures (respectivement 0,2 et
2 µg de phénol/litre) aux valeurs correspondantes pour d'autres
espèces de poissons (concentration sans effet létal observable,
2,2-6,1 mg/litre) et d'amphibiens, ou tirées d'études sur la
reproduction des crustacés (concentration sans effet létal
observable, 10 mg de phénol/litre). On ne dispose pas de données sur
des épreuves à long terme qui auraient été pratiquées sur des
organismes marins.
En général le facteur de bioconcentration du phénol chez les
divers types d'organismes aquatiques est très bas (<1-10) encore
qu'on ait signalé parfois des valeurs plus élevées (jusqu'à 2200).
Il est donc vraisemblable que le phénol ne subit pas d'accumulation
biologique importante.
Les données dont on dispose au sujet de la destinée et des
effets du phénol chez les organismes terrestres sont très peu
nombreuses. La CE50 à 120 h. est de 120 à 170 mg/litre pour le
millet et lors d'une épreuve par contact, on a obtenu pour la CL50
chez le lombric, une valeur comprise entre 2,4 et 10,6 µg/cm3.
9. Résumé de l'évaluation
9.1 Santé humaine
La population dans son ensemble est essentiellement exposée au
phénol par la voie respiratoire. Par voie orale, il peut y avoir
exposition répétée par suite de la consommation d'eau de boisson
contaminée ou d'aliments fumés.
On ne dispose pas de données suffisantes pour déterminer
l'ampleur de l'exposition de la population générale, mais on peut
donner une limite estimative supérieure de l'absorption quotidienne.
En mettant les choses au pire, on peut considérer que l'exposition
maximale se produit chez un individu qui inhale en permanence de
l'air fortement contaminé et consomme souvent des aliments fumés ou
de l'eau de boisson qui contient du phénol à des concentrations
atteignant le seuil gustatif. On a calculé que la dose quotidienne
ingérée maximale totale estimative pour un individu de ce genre
pesant 70 kg était de 0,1 mg/kg de poids corporel.
Les valeurs de la dose sans effets nocifs observables obtenues
par expérimentation animale en prenant comme critères les troubles
rénaux et les effets sur le développement étaient, chez le rat, de
l'ordre de 12 à 40 mg/kg de poids corporel et par jour. En utilisant
un coefficient d'incertitude de 200, on peut recommander comme
limite supérieure de la dose journalière totale une valeur située
entre 60 et 200 µg/kg de poids corporel. En prenant pour l'homme une
dose quotidienne limite de 100 µg/kg de poids corporel, on peut
conclure que l'exposition de toutes origines au phénol de la
population dans son ensemble se situe en-dessous de ces valeurs.
On peut être préoccupé par le fait que, selon certaines
données, le phénol pourrait être génotoxique et que d'autre part, on
ne possède pas suffisamment de résultats pour écarter avec certitude
l'éventualité que le phénol soit cancérogène. L'évaluation de ce
composé doit être revue périodiquement.
9.2 Environnement
Le phénol ne subit probablement pas d'accumulation biologique
importante. Il est toxique pour les organismes aquatiques; en
appliquant la méthode modifiée de l'Agence de protection de
l'environnement des Etats-Unis, on peut considérer que la
concentration préoccupante de cette substance dans l'environnement
est de 0,02 µg/litre. On manque de données suffisantes sur son
action chez les plantes et les organismes terrestres.
Il peut y avoir transport du phénol d'un compartiment à l'autre
de l'environnement par dépôt humide ou par lessivage du sol. En
général, ce composé ne devrait pas persister dans l'environnement.
Les rares données dont on dispose sur l'exposition ne permettent pas
d'évaluer le risque qu'il présente pour les écosystèmes aquatiques
ou terrestres. Toutefois, en tenant compte de la valeur de sa
concentration préoccupante pour l'environnement aquatique, il est
raisonnable de considérer qu'en cas de contamination par le phénol
des eaux de surface ou des eaux marines, il y a un risque pour les
organismes aquatiques.
RESUMEN
1. Identidad, propiedades físicas y químicas, métodos analíticos
El fenol es un sólido cristalino, blanco, que funde a 43 oC y
se licúa al contacto con el agua. Posee un olor acre característico
y un sabor ardiente fuerte. Es soluble en la mayor parte de los
disolventes orgánicos. A temperatura ambiente, su solubilidad en
agua es limitada; por encima de 68 oC es completamente hidrosoluble.
El fenol es moderadamente volátil a temperatura ambiente. Es un
ácido débil y, en su forma ionizada, muy sensible a las reacciones
de sustitución electrofílica y a la oxidación.
El fenol se puede obtener a partir de muestras ambientales por
absorción en una solución de NaOH o en contacto con sorbentes
sólidos. La desorción se lleva a cabo por acidificación, destilación
al vapor y extracción con éter (a partir de soluciones) o mediante
desorción térmica o líquida (a partir de sorbentes sólidos). Las
técnicas analíticas más importantes son la cromatografía de gases en
combinación con la detección de ionización por conductor y de
captura de electrones, y la cromatografía en fase líquida, de alta
presión, en combinación con la detección por luz ultravioleta. En el
aire, el límite de detección más bajo que se haya notificado es de
0,1 µg/m3. Se puede determinar la presencia de fenol en la sangre
y la orina; en muestras de orina se ha registrado un límite de
detección de 0,5 µg/litro.
2. Fuentes de exposición humana y ambiental
El fenol es uno de los componentes del alquitrán de hulla y se
forma durante la descomposición natural de materiales orgánicos. No
obstante, la mayor parte del fenol presente en el medio ambiente es
de origen antropogénico. Algunas fuentes potenciales son la
producción y el uso de fenol y de sus productos, especialmente
plásticos fenólicos y caprolactama, los gases de escape, la quema de
leña y el humo de los cigarrillos. Otra fuente potencial es la
degradación atmosférica del benceno por la influencia de la luz, si
bien la presencia del fenol en los purines puede asimismo tener
considerable influencia en sus niveles atmosféricos. Los derivados
del benceno y del fenol pueden, mediante una conversión in vivo,
constituir una fuente de exposición humana endógena a fenol.
Según parece, la producción mundial de fenol fue bastante
regular a lo largo del decenio de 1980, en que los Estados Unidos
fueron el productor más importante. Se usa principalmente como
materia básica de las resinas fenólicas, del bisfenol A y de la
caprolactama. También se le conocen algunas aplicaciones médicas y
farmacéuticas.
3. Transporte, distribución y transformación en el medio ambiente
Las principales emisiones de fenol van al aire. La mayor parte
del fenol existente en la atmósfera se degradará mediante reacciones
fotoquímicas frente a los dihidroxibencenos, los nitrofenoles y los
productos de rotura del anillo, con una semivida, estimada en 4 a 5
hs. Una parte menor desaparecerá del aire por deposición hídrica
(lluvia). Se piensa que el fenol es móvil en el suelo, pero el pH
puede influir en el transporte y la reactividad.
El fenol presente en el agua y el suelo puede degradarse por
reacciones abióticas, así como por la actividad microbiana, dando
lugar a un número de compuestos, los más importantes de los cuales
son el dióxido de carbono y el metano. La proporción entre la
biodegradación y la degradación general del fenol está determinada
por múltiples factores, como la concentración, la aclimación, la
temperatura y la presencia de otros compuestos.
4. Niveles ambientales y exposición humana
No se dispone de datos sobre los niveles atmosféricos de fenol.
Se supone que los niveles básicos son inferiores a 1 ng/m3. Los
niveles urbanos y suburbanos oscilan entre 0,1 y 8 µg/m3, mientras
que se ha notificado que las concentraciones en las zonas próximas
al foco de emisión (industria) alcanzan magnitudes cien veces
superiores. Se ha detectado fenol en la lluvia y en las aguas
superficiales y subterráneas, pero los datos son muy escasos. En
sedimentos y aguas subterráneas se han notificado niveles elevados
de fenol debidos a la contaminación industrial.
La exposición profesional al fenol puede tener lugar durante la
producción del mismo y de sus derivados, la aplicación de resinas
fenólicas (industrias maderera y siderúrgica) y algunas otras
actividades industriales. La concentración más alta (hasta 88
mg/m3) se ha notificado en relación con trabajadores de la antigua
Unión Soviética que apagaban el coque con aguas residuales que
contenían fenol. La mayor parte de las restantes concentraciones
notificadas no rebasan los 19 mg/m3.
Para la población en general, el humo de cigarrillo y los
alimentos ahumados constituyen las fuentes más importantes de
exposición al fenol, aparte de la exposición a través del aire. La
exposición a través del agua potable y de los alimentos contaminados
por inadvertencia probablemente sea baja; el fenol tiene un olor y
un sabor desagradables, que en la mayor parte de los casos provocan
el rechazo del consumidor.
5. Cinética y metabolismo
El fenol se absorbe fácilmente por todas las vías de
exposición. Tras la absorción, la sustancia se distribuye
rápidamente a todos los tejidos.
El fenol absorbido se conjuga principalmente con el ácido
glucurónico y el ácido sulfúrico y, en menor medida, se hidroxila en
pirocatequina e hidroquinona. También se conjuga con los fosfatos.
La formación de metabolitos reactivos (4,4-bifenol y difenoquinona)
se ha demostrado en estudios in vitro con neutrófilos y leucocitos
humanos activados.
Las cantidades relativas de glucurono y sulfoconjugados varían
según la dosis y la especie animal. Tras aumentar la dosis de fenol,
se observó en las ratas un cambio de la sulfatación a la
glucuronidación.
El hígado, los pulmones y la mucosa gastrointestinal
constituyen los sitios más importantes del metabolismo fenólico. La
función relativa desempeñada por esos tejidos depende de la vía de
administración y de la dosis.
Estudios in vivo e in vitro han demostrado la unión
covalente del fenol con las proteínas tisulares y plasmáticas.
Algunos metabolitos fenólicos se unen asimismo a las proteínas.
La excreción por la orina es la principal vía de eliminación
del fenol en los animales y en los seres humanos. La tasa de
excreción urinaria varía en función de la dosis, de la vía de
administración y de la especie. Una parte menor se excreta a través
de las heces y del aire espirado.
6. Efectos en mamíferos de laboratorio y en sistemas de
prueba in vitro
El fenol tiene una toxicidad aguda moderada en los mamíferos.
En los roedores, los valores de la DL50 por vía oral oscilan entre
300 y 600 mg de fenol/kg de peso corporal. Los valores de la
DL50por vía cutánea para ratas y conejos oscilan entre 670 y 1400
mg/kg de peso corporal, respectivamente, y el valor de la CL50 por
inhalación a las 8 horas en las ratas es superior a los 900 mg de
fenol/m3. Los síntomas clínicos después de la exposición aguda son
hiperexcitabilidad neuromuscular y convulsiones graves, necrosis de
la piel y de las mucosas de la garganta y efectos en los pulmones,
fibras nerviosas, riñones, hígado y en la respuesta pupilar a la
luz.
Las soluciones de fenol son corrosivas para la piel y los ojos.
Los vapores de fenol pueden irritar las vías respiratorias. Existen
pruebas de que el fenol no produce sensibilización cutánea.
Los efectos más importantes notificados a partir de estudios de
corta duración en animales fueron neurotoxicidad, lesiones hepáticas
y renales, trastornos respiratorios y retraso del crecimiento. Se
han notificado efectos tóxicos en el riñón de las ratas con dosis
por vía oral de 40 mg/kg al día o más. La toxicidad en el hígado
resultó evidente en las ratas a las que se habían administrado al
menos 100 mg/kg diarios. En un estudio limitado de 14 días de
duración realizado en ratas se notificó un nivel sin efectos
adversos observados (NOAEL) de 12 mg/kg al día por vía oral, basado
en los efectos renales. En este experimento, la miosis (respuesta
del iris a la luz) se mantuvo inhibida con 4 mg/kg al día; sin
embargo, no está claro el significado médico de este hallazgo. Se
notificó la existencia de algunos cambios biológicos en la mucosa
intestinal y los riñones de ratones con dosis inferiores a 1 mg/kg
al día, dato de significado toxicológico incierto.
No hay estudios adecuados sobre la toxicidad reproductiva del
fenol. En estudios con ratas y ratones el fenol ha sido identificado
como tóxico del desarrollo. En dos estudios de dosis múltiples en
ratas, se han notificado NOAEL de 40 mg/kg al día (el más bajo nivel
sin efectos adversos observados (LOAEL) fue de 53 mg/kg al día) y de
60 mg/kg al día (el LOAEL fue de 120 mg/kg al día). En el ratón, el
NOAEL fue de 140 mg/kg al día (el LOAEL fue de 280 mg/kg al día).
La mayor parte de las pruebas de mutagenicidad bacteriana han
dado resultados negativos. En células in vitro de mamíferos se han
observado mutaciones, lesiones cromosómicas y efectos en el ADN. El
fenol no tiene efectos en la comunicación intercelular (medida como
cooperación metabólica) en cultivos de células de mamíferos. En
algunos estudios se ha observado la inducción de micronúcleos en
células de médula ósea de ratones. Con dosis más bajas no se
observaron micronúcleos en estudios con ratones.
Se han llevado a cabo dos estudios de carcinogenicidad con
ratas y ratones machos y hembras a los que se administró fenol con
el agua de beber. Sólo se observó malignidad (por ejemplo, carcinoma
de células C de la tiroides y leucemia) en ratas macho con dosis
bajas. No se han realizado estudios adecuados de carcinogenicidad
por vía dérmica o por inhalación. Estudios de carcinogenicidad de
dos fases han mostrado que el fenol, aplicado repetidamente a la
piel del ratón, tiene efectos activadores.
7. Efectos en el ser humano
Se ha notificado una larga serie de efectos adversos en el ser
humano resultantes de la exposición bien documentada al fenol por
vía cutánea, oral o intravenosa. Se ha notificado irritación
gastrointestinal tras su ingestión. Los efectos locales de la
exposición cutánea van desde el emblanquecimiento o el eritema
indoloros hasta la corrosión y la necrosis profunda. Entre los
efectos sistémicos cabe citar disritmias, acidosis metabólica,
hiperventilación, disnea, insuficiencia renal aguda, lesiones
renales, orinas oscuras, metahemoglobinemia, trastornos neurológicos
(incluidas convulsiones), choque cardiovascular, coma y muerte. La
dosis mínima reportada como causante de muerte en el ser humano es
de 4,8 g por ingestión; la muerte se produjo en menos de 10 minutos.
Durante mucho tiempo se ha reconocido la posibilidad de
envenenamiento por inhalación de los vapores de fenol, pero no se
han reportado casos mortales relacionados con esta vía de
exposición. Los síntomas que se asocian a la inhalación de fenol
consisten, entre otros, en anorexia, pérdida de peso, dolor de
cabeza, vértigo, salivación y orinas oscuras.
El fenol no es un agente sensibilizante.
El umbral de percepción del fenol por el olfato humano oscila
entre 0,021 y 20 mg/m3 en el aire. Se ha notificado un umbral de
percepción del fenol en el agua de 7,9 mg/litro, y un umbral de
percepción por el gusto de 0,3 mg/litro en el agua.
No se dispone de datos adecuados sobre la carcinogenicidad del
fenol en el ser humano.
8. Efectos en los seres vivos del medio ambiente
En estudios con bacterias de especie única, los valores de la
CE50 con inhibición del crecimiento oscilaron entre 244 y 1600 mg
de fenol/litro. Se comprobó un umbral de toxicidad de 64 mg de
fenol/litro. Los valores para los protozoarios y los hongos fueron
de la misma cuantía que para las bacterias, mientras que para las
algas fueron ligeramente inferiores.
El fenol es tóxico para los organismos superiores de agua
dulce. Los valores más bajos de la CL50 o la CE50, para
crustáceos y peces se sitúan entre 3 y 7 mg de fenol/litro. Los
datos sobre la toxicidad aguda para organismos marinos son
comparables a los correspondientes a organismos de agua dulce. En
estudios de larga duración sobre especies de crustáceos y de peces
se han observado notables diferencias de sensibilidad; los valores
de la CL1 en pruebas con embriones y larvas de Salmo y
Carassius resultaron mucho más bajos (0,2 y 2 µg de fenol/litro,
respectivamente) que los valores correspondientes a otras especies
de peces (NOLC de 2,2-6,1 mg/litro) y anfibios, o que los obtenidos
en pruebas de reproducción en crustáceos (NOLC de 10 mg de
fenol/litro). No se dispone de datos acerca de pruebas de larga
duración realizadas en organismos marinos.
Los factores de bioconcentración del fenol en diversos tipos de
organismos acuáticos son en general muy bajos (< 1-10), aunque se
han notificado también algunos valores más altos (hasta 2200). Así
pues, no se prevé que la bioacumulación del fenol sea significativa.
Los datos sobre el destino y los efectos del fenol en
organismos terrestres son muy escasos. En el mijo se determinó una
CE50 a las 120 horas de 120-170 mg/litro mientras que, en una
prueba de contacto, la CL50 para especies de lombrices resultó ser
de 2,4-10,6 µg/cm2.
9. Resumen de la evaluación
9.1 Salud humana
La población en general está expuesta al fenol principalmente
por inhalación. La exposición repetida por vía oral puede producirse
por el consumo de alimentos ahumados o de agua potable.
No existen datos suficientes para determinar el grado de
exposición de la población en general, pero se puede calcular la
cantidad máxima ingerida diariamente. Basándose en "la peor de las
hipótesis" se puede realizar una estimación suponiendo que un
individuo estará expuesto en grado máximo al fenol mediante la
inhalación continua de aire intensamente contaminado acompañada de
un consumo frecuente de productos alimenticios ahumados y de agua
que contenga fenol hasta niveles de percepción por el gusto. En
total, la ingesta máxima diaria de fenol en un individuo de 70 kg se
calcula en 0,1 mg/kg de peso corporal al día.
Los valores de NOAEL más bajos identificados en experimentos
con animales se refieren a efectos en el riñón y en el desarrollo, y
en las ratas se sitúan dentro de un margen de variación de 12-40
mg/kg de peso corporal al día. Utilizando un factor de incertidumbre
de 200, se recomienda como límite máximo de la ingesta diaria total
(IDT) entre 60 y 200 µg/kg de peso corporal al día. Teniendo en
cuenta que el límite máximo de la ingesta diaria en seres humanos se
calcula en 100 µg/kg de peso corporal al día, se llega a la
conclusión de que la exposición media de la población en general al
fenol, sea cual fuere la fuente, se encuentra por debajo de este
intervalo.
Son motivo de preocupación algunas indicaciones de que el fenol
podría ser genotóxico y el hecho de que no haya datos suficientes
para descartar con seguridad la posible carcinogenicidad del
compuesto. La evaluación debe mantenerse sujeta a revisión
periódica.
9.2 Medio ambiente
No se prevé una bioacumulación importante del fenol. Este
compuesto es tóxico para los organismos acuáticos; mediante la
aplicación del método modificado de la Agencia de los EE.UU. para la
Protección del Medio Ambiente, se puede determinar un nivel en medio
ambiente de preocupación de 0,02 µg/litro. Se carece de datos
adecuados sobre plantas y organismos terrestres.
El transporte de fenol entre compartimientos se produce
principalmente por deposición hídrica y filtración a través del
suelo. En general, es poco probable que el compuesto persista en el
medio ambiente. La escasez de datos sobre la exposición no permiten
evaluar el riesgo que representa el fenol para los ecosistemas tanto
acuáticos como terrestres. Sin embargo, habida cuenta del nivel de
preocupación ambiental que se ha establecido en relación con el
agua, es razonable suponer que los organismos acuáticos pueden
correr riesgo en cualquier agua superficial o marina contaminada con
fenol.
Relative molecular mass: 94.11
Common name: phenol
Common synonyms: acidum carbolicum, acidum phenolicum,
acidum phenylicum, benzaphenol, benzene
phenol, benzenol carbolic acid,
hydroxybenzene (IUPAC), oxybenzene,
monohydroxybenzene, monophenol, phenic
acid, phenol alcohol, phenyl hydrate,
phenyl hydroxide, phenylic acid
Common trade names: carbololie (NLD), fenololie (NLD),
kristalliertes Kreosot (GER),
Steinkohlenkreosot (GER),
Steinkohlenteerkreosot (GER), venzénol
(FRA), ENT 1814.
CAS registry number: 108-95-2
CAS chemical name: phenol
2.2 Physical and chemical properties
Some physical and chemical properties of phenol are given in
Table 1.
Table 1. Some physical and chemical properties of phenola
Boiling point (101.3 Pa) 181.75 °C
Melting point 43 °C
40.9 °C (ultrapure material)
Relative density (20 °/4 °)b 1.071
Relative vapour density (air = 1) 3.24
Vapour pressure (20 °C) 0.357 mmHg
(50 °C) 2.48 mmHg
(100 °C) 41.3 mmHg
Saturation concentration in air (20°C) 0.77 g/m3
Solubility in water (16 °C) 67 g/litrec
Log n-octanol/water partition
coefficient (log Pow) 1.46d
Dissociation constant in water
at 20 °C (Ka) 1.28 x 10-10
Flash-point (closed cup) 80 °C
(open cup) 79 °C
85 °Ce
Flammability limits 1.3-9.5%
a From: Kirk-Othmer (1980); RIVM (1986)
b Weast (1987)
c Above 68.4 °C phenol is entirely soluble in water
d The Pow of phenol is very much dependent on pH; pH at log
Pow = 1.46 was not given
e Budavari et al. (1989)
Phenol has a melting point of 43 °C and forms white to
colourless crystals (Budavari et al., 1989). It has also been
described as a colourless to pink solid or thick liquid (NIOSH,
1985a). Phenol has a characteristic acrid smell and a sharp burning
taste. Odour and taste threshold values are reported in section 8.3.
In the molten state, it is a clear, colourless liquid with a low
viscosity. A solution with approximately 10% water is called
phenolum liquefactum, as this mixture is liquid at room temperature.
Phenol is soluble in most organic solvents (aromatic hydrocarbons,
alcohols, ketones, ethers, acids, halogenated hydrocarbons). The
solubility is limited in aliphatic solvents.
The chemical properties of phenol are affected by the resonance
stabilization possibilities of phenol and, in particular, of the
phenolate ion. Because of this, phenol reacts as a mild acid. In the
presence of electrophilic groups (meta-indicators), the acidic
properties are emphasized.
Phenol is sensitive to oxidizing agents. Splitting of the
hydrogen atom from the phenolic hydroxyl group is followed by
resonance stabilization of the resulting phenyloxy radical. The
radical formed can easily be further oxidized. Depending on the
oxidizing agent applied and the reaction conditions, various
products, such as dihydroxy- and trihydroxybenzenes and quinones are
formed. These properties make phenol suitable as an antioxidant,
functioning as a radical trapping agent. Phenol undergoes numerous
electrophilic substitution reactions, such as halogenation and
sulfonation. It also reacts with carbonyl compounds in both acidic
and alkaline media. In the presence of formaldehyde, phenol is
readily hydroxymethylated with subsequent condensation to resins.
2.3 Conversion factors
1 mg/m3 = 0.26 ppm
1 ppm = 3.84 mg/m3
2.4 Analytical methods
Analytical methods for phenol are shown in Table 2.
2.4.1 Sampling and pre-treatment
Phenol in air samples may be collected by absorption in NaOH
solution contained in wash bottles or on filters impregnated with
NaOH solution. Phenol in air, water and solid waste samples may be
collected (directly or after extraction) in tubes containing solid
sorbent (Tenax, silica gel or, less commonly, carbon) (IARC, 1989).
For large air volumes, the NaOH method is usually preferred, whereas
for smaller quantities (personal air sampling, for instance) solid
sorbent tubes have been reported to be more practical (RIVM, 1986).
Release of phenol from aqueous solutions (including NaOH
sorbent, and also urine) is achieved by acidification, steam
distillation and/or ether extraction. After adsorption onto Tenax,
thermal desorption at 250 °C is usually preferred (the whole sample
may be inserted directly into a gas chromatograph), whereas, in the
case of silica gel, liquid desorption with chloroform is generally
applied. There is a small possibility of chemical conversion during
heating, whereas Tenax may react with ozone to form small quantities
of phenol. Only one analysis per sample is possible in the case of
thermal desorption. The use of liquid desorption allows more
analyses per sample, but because of the unavoidable dilution the
detection limit is higher (RIVM, 1986).
Table 2. Methods for the detection of phenol in air
Sampling Volume of air Pre-treatment Analysis Detection limit Reference
(litre) before analysis (µg/m3)
Absorbance in NaOH solution 100 acidification GC and FID 10 µg per NIOSH (1984)
in a wash bottle; 1 litre/min sample
Absorbance in NaOH solution 25 000 acidification and GC and FID 4 Katz (1977)
in an impinger; 20 litre/min steam distillation
Glass fibre filter impregnated with 600 acidification and GC and FID 13 Kifune (1979)
NaOH and glycerol; 120 litre/min extraction with ether
Absorbance in NaOH solution in 1000 acidification and colorimetry with 2 Katz (1977)
an impinger; 28 litre/min steam distillation 4-amino antipyrine
Absorbance in NaOH solution - none colorimetry with 700 Hensehler (1975)
in a wash bottle; 1 litre/min 4-amino antipyrine
Absorbance in NaOH solution in 150 conversion in an HPLC with UV detector 0.2 Kuwata et al.
a sinter wash bottle; 1-2 litre/min azophenol derivate (254 nm) (1980)
Absorbance in Na2CO3 solution; 30 calibration at pH = 10 UV at 235 nm at 2 pH 160 Zavorovskaya &
1 litre/min and pH = 7 values Nekhorosheva (1981)
Absorbance in NaOH solution in 300 calibration at pH = 12 UV at 241 and 295 nm 20 Bergshoeff (1960)
an impinger; 28 litre/min and at pH = 6
Tenax with and without KOH; 5 thermal desorption; GC and FID 0.1 Hoshika & Muto
0.25 litre/min 250 °C (1979)
Tenax; 0.1 litre/min 4 thermal desorption; GC and FID 1 Russell (1975)
260 °C
Tenax; 0.5-1 litre/min 70 thermal desorption; GC and MS 0.3 Hagemann et
250 °C al. (1978)
Table 2 (contd)
Sampling Volume of air Pre-treatment Analysis Detection limit Reference
(litre) before analysis (µg/m3)
Tenax; 0.75 litre/min 90 thermal desorption; IR with 20 m gas 300 Podolak et al.
300 °C cuvette (1981)
Silica gel 25 liquid desorption GC and FID 3 Dimitriev &
with chloroform Mishchikhin (1983)
Silica gel; 0.05-2 litre/min 10 liquid desorption HPLC with UV detector 50 Oomems &
with chloroform (275 nm) Schuurhuis (1983)
Drägen tube gas detection 0.5 none reading of colorisation 19 000 Leichnitz (1982)
2.4.2 Analysis
The most important analytical techniques for the detection of
phenol are gas chromatography (GC) in combination with flame
ionization detection (FID), and high-performance liquid
chromatography (HPLC) in combination with ultraviolet (UV)
detection. The identification of phenol by GC/FID has been improved
by reaction of phenols with bromide or pentafluoro-benzyl bromide,
and the use of electron capture detection (Hoshika & Muto, 1979; US
EPA, 1986a). The identification of phenol using HPLC can be improved
by reaction with, for example, p-nitrobenzene diazonium
tetrafluoroborate to form azo derivatives (Kuwata et al., 1980).
Detection limits of the above techniques for air samples are
given in Table 2. For the GC/FID detection of phenol in water, using
electron capture detection following derivatization with
pentafluorobenzyl bromide, a detection limit of approximately 0.2
µg/litre has been reported (US EPA, 1986a).
GC in combination with mass spectrometry (MS) is more sensitive
than with FID, but is more expensive. This technique, using either
packed or capillary columns, was reported to have practical
quantitative limits of approximately 1 mg phenol/kg wet weight for
soil/sediment samples, 1-200 mg phenol/kg for wastes, and 10
µg/litre for groundwater samples (US EPA, 1986b,c).
Another reported analytical technique is colorimetry after
reaction of phenol with 4-amino antipyrine, in the presence of
potassium ferricyanide, to form an antipyrine dye. The detection
limit of this technique for water samples, after steam distillation,
was reported to be 1 µg/litre (American Public Health Association,
1985). For air samples of 1 m3, the detection limit was reported
to be 2 µg/m3 (see Table 2). The interference by para-substituted
phenols and chlorophenols is low (RIVM, 1986).
Infrared (IR) detection of phenol is a rather insensitive
method, and is highly susceptible to interference by other compounds
such as water vapour. However, it is a rapid and specific method
which allows directly readable continuous measurement. It is
considered to be attractive only at air concentrations of more than
1000 µg/m3, for example in leakage tests and industrial warning
systems (RIVM, 1986). Also directly readable is the Dräger gas
detection tube (see Table 2); however, the detection limit is very
high (> 19 000 µg/m3).
For the GC/FID detection of total phenol in urine samples,
after acidification and ether extraction, a detection limit of 0.5
µg/litre was estimated (NIOSH, 1985b). Colorimetric methods for the
determination of free phenol in both urine and blood are available.
In one method, phenol reacts with p-nitroaniline following
deproteinization and extraction with diethyl ether. Other phenols
will interfere (Müting et al., 1970). In another method, phenol
reacts with ammonia and N-chlorosuccinimide in alkaline media with
sodium nitroprusside as a catalyst. This method was found to be
applicable in the range of 3-24 mg/litre, using spiked samples of
urine (Amlathe et al., 1987). The concentration of total phenol in
urine and plasma can be determined by GC/MS following hydrolysis of
glucuronide and sulfate conjugates with sulfuric acid and
derivatization with propanoic anhydride. The detection limit is
reported to be 10 µg/litre (Pierce & Nerland, 1988).
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural sources
Phenol is a constituent of coal tar, and is formed during the
natural decomposition of organic materials. Increased environmental
levels may result from forest fires (Hubble et al., 1981).
Phenol has been detected among the volatile components from
liquid manure at concentrations of 7-55 µg/kg dry weight (Spoelstra,
1978). In the Netherlands, for example, the contribution from this
source to the overall phenol emission into air in 1983 has been
calculated to be 15%, assuming complete volatilization of phenol and
an average phenol concentration in manure of 30 µg/kg dry weight
(RIVM, 1986).
3.2 Anthropogenic sources
3.2.1 Production
The most commonly used production method for phenol, on a
worldwide scale, is from cumene (isopropylbenzene). In the USA, for
example, more than 98% of phenol is produced by this method (IARC,
1989). Phenol is also produced from chlorobenzene and toluene. A
small but steady supply of phenol is recovered as a by-product of
metallurgical coke manufacture (IARC, 1989). The emission factor of
phenol into air during production by the cumene process has been
reported to be 0.16 g phenol emitted per kg phenol produced (UBA,
1981).
In Table 3, information is presented concerning the production
of phenol in various countries in 1981. This information was derived
from the open literature (Chemfacts, 1978-1981; United Nations,
1980; SRI, 1982; CID-TNO, 1984; IARC, 1989) and, where necessary,
was extrapolated to 1981. There have been no major production
changes according to data available up to 1986 (IARC, 1989).
3.2.2 Industrial processes
Phenol is the basic feedstock from which a number of
commercially important materials are made, including phenolic
resins, bisphenol A (2,2-bis-1-hydroxyphenylpropane), capro-lactam,
alkyl phenols, as well as chlorophenols such as pentachlorophenol
(IARC, 1989).
Table 3. Production of phenol in 1981 and 1986 (kilotonne/year)
Country Production Production
in 1981a in 1986b
Brazil 50
Bulgaria 35
Czechoslovakia 44 46
Finland 32
France 150
Germany, Federal
Republic of 247
Italy 223
India 14
Japan 215 260
Mexico 20
The Netherlands 166
Poland 66
Romania 66
Spain 55 70
United Kingdomc 110 53
USA 1350 1413
USSR 497 515
Other countries 34
European Community (total) 920
Total 3374
a Chemfacts (1978-1981); United Nations (1980); SRI (1982); CID-TNO
(1984); IARC (1989)
b From IARC (1989)
c Phenol is no longer produced in the UK
The most important phenol emissions result from the use of
phenolic resins. Phenolic resins are used as a binding material in,
for example, insulation materials, chipboard and triplex, paints and
casting sand foundries. Their contents vary from 2-3% for insulation
material to > 50% for moulds (Bollig & Decker, 1980). Emissions are
approximately proportional to the concentration of free phenol,
which is present as a monomer in these materials (1-5%) (Bollig &
Decker, 1980). In addition, phenol may be released as a result of
thermal decomposition of the resins.
In foundries, phenol emissions develop both during the
production of moulds and kernels and during founding (TNO, 1978).
The content of free phenol may rise by up to 12% (Ryser & Ulmer,
1980). Emission factors reported by RIVM (1986) were 0.35 g phenol
emitted per kg used casting sand, 2-5 g phenol emitted per kg resin
in the production of casting sand, and 10 g phenol emitted per kg
resin during the production of moulds by the "hot-box" procedure.
Other industrial activities in which phenol may be emitted to
the air, as well as some of their reported emission factors, are
listed below:
* production of phenol resins (0-0.5 g phenol emitted per kg
resin produced) (RIVM, 1986)
* production of phenols and phenol derivatives
* production of caprolactam (0.02-0.05 g phenol emitted per kg
cyclohexanone (an intermediate) produced (RIVM, 1986)
* production of cokes
* production of insulation materials
* process emissions
Emissions to water may also result from processing.
3.2.3 Non-industrial sources
Phenol has been detected in the exhaust gases of private cars
at concentrations of 0.3 ppm (approximately 1.2 mg/m3) to 1.4-2.0
ppm (5.4-7.7 mg/m3) (Kuwata et al., 1980; Verschueren, 1983). It
has also been identified in cigarette smoke, in quantities that are
comparable to an average emission of 0.4 mg/cigarette (Groenen,
1978). Emission gases from all material incinerators and home fires,
especially wood-burning, may contain substantial quantities of
phenol (Den Boeft et al., 1984).
Another potential source of phenol is the atmospheric
degradation of benzene under the influence of light (Hoshino &
Akimoto, 1978).
Phenols have been detected in smoked foods (section 5.3.2).
3.3 Endogenous sources
An important additional source of human phenol exposure may be
the in vivo formation from various xenobiotics, e.g., benzene
(Pekari et al., 1992).
3.4 Uses
The largest single use of phenol is the production of phenolic
resins. Next is its use in the production of caprolactam, an
intermediate in the production of nylon 6, and 2,2-bis-1-
hydroxyphenylpropane (bisphenol A), which is mainly used in the
production of phenolic resins (Kirk-Othmer, 1980).
The various applications of phenol as a percentage of total
1981 consumption, in the USA and western Europe, are summarized in
Table 4 (Kirk-Othmer, 1980). The data presented are in close
agreement with the 1986 USA data reported by IARC (1989).
Table 4. Use of phenol in 1981 (% total consumption)a
Production of USA West Europe
Phenolic resins 48 36
Bisphenol A(2,2-bis-1-
hydroxyphenylpropane) 18 17
Caprolactam 15 28
Other products 19 19
Total 100 100
a From: Kirk-Othmer (1980)
Phenol was widely used in the 19th century for wound treatment
and as an antiseptic and local anaesthetic. The medical uses of
phenol today include incorporation into disinfectants, antiseptics,
lotions, salves and ointments (IARC, 1989). Another medical
application of phenol is its use as a neurolytic agent, applied in
order to relieve spasm and chronic pain (Wood, 1978).
In addition to the applications mentioned in section 3.2.2,
phenol is used in the manufacture of paint and varnish removers,
lacquers, paints, rubber, ink, illuminating gases, tanning dyes,
perfumes, soaps and toys (IARC, 1989).
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1 Transport and distribution between media
No data have been found concerning wet and dry deposition of
phenol. Since phenol in air is present almost exclusively in the gas
phase, dry deposition (by particle deposition) is expected to be
negligible. Wet deposition may contribute to the disappearance of
phenol from the atmosphere: when phenol was measured during seven
episodes of rain in Portland, Oregon, USA, relatively high
concentrations were found in the rain water (Leuenberger et al.,
1985).
Based on its relatively high solubility in water and the
relatively low vapour pressure at room temperature, phenol is
expected to end up largely in the water phase upon distribution
between air and water. Consequently, transport from air to soil and
water is likely (RIVM, 1986). Volatilization from dry near-surface
soil should be relatively rapid (Howard, 1989).
Theoretical deposition rates for phenol were estimated assuming
a behaviour similar to SO2, and comparing with the rate of reaction
of phenol with hydroxyl radicals (see below). Based on this
comparison, it was concluded that most phenol in the atmosphere is
degraded chemically, rather than transported (RIVM, 1986).
Partition coefficient (Koc) values of phenol for two silt
loams were reported to be 39 and 91 dm3/kg. Based on these Koc
values, phenol would be expected to be highly mobile in soil, and
therefore may leach to ground water (Howard, 1989). This was
confirmed by Scott et al. (1982) who found low adsorption of
phenol to two sterile silt loams (pH 5.4, organic matter content 1.1
and 3.6, respectively), as shown by Freundlich K values of 0.57 and
1.19, respectively. Based on the pKa (log (1.28 x 10 -10)),
phenol exists in a partially dissociated state in water and moist
soils and, therefore, its transport and reactivity may be affected
by pH (Howard, 1989). Upon measurement of the sorption and
desorption of phenol from water to surface sediment (pH 6.21-6.35;
organic matter content of fine fraction (< 2 µM) was 10.2%), phenol
appeared to bind strongly to the soil. The estimated Koc was 2900
dm3/kg (Isaacson & Frink, 1984). However, no correction was made
for any degradation occurring during the experiments. The adsorption
of phenol onto soil or microbial biomass may be decreased by the
presence of phenol derivatives (Boyd, 1982; Selvakumar & Hsieh,
1988). Phenol has been detected in ground water as a result of
leaching (see section 5.1.2).
4.2 Abiotic degradation
4.2.1 Air
Phenol may react in air with hydroxyl and NO3 radicals, and
undergo other photochemical reactions to form dihydroxy-benzenes,
nitrophenols, and ring cleavage products (Atkinson et al., 1979;
Bruce et al., 1987). The half-life for phenol in air was found to
be 4-5 h under photochemically reactive conditions in a smog chamber
(Spicer et al., 1985); this is in good agreement with the
estimated half-life of phenol in air of 5 h based on its estimated
reaction rate with hydroxyl radicals (RIVM, 1986). Howard (1989)
reported an estimated half-life of 15 h for the reaction of phenol
with hydroxyl radicals in air. The reaction of phenol with nitrate
radicals during the night may be a significant removal process; a
half-life of 15 min has been estimated at an atmospheric
concentration of 2x108 nitrate radicals per cm3 (Howard, 1989).
Phenol absorbs light in the region of 290-330 nm and therefore
could photolyse (Howard, 1989).
4.2.2 Water
Phenols generally react in sunlit natural water via reaction
with photochemically produced hydroxyl and peroxy radicals; typical
half-lives were reported to be 100 and 19.2 h, respectively (Howard,
1989).
Phenol was found to be oxidized to carbon dioxide in water
under experimental conditions (temperature approximately 50 °C), in
the presence of oxygen and sunlight, at a rate of 11% per 24 h
(Knoevenagel & Himmelreich, 1976). It was reported to react with
nitrate ions in dilute aqueous solutions to form dihydroxybenzenes,
nitrophenols, nitrosophenol and nitroquinone, presumably by a
radical mechanism involving hydroxyl and phenoxyl radicals (Niessen
et al., 1988). Phenol has been found to react with nitrous acid in
waste water to form cyanide (Adachi et al., 1987), and to form
chlorophenols in chlorinated drinking-water (Jarvis et al., 1985)
and p-benzoquinone in the presence of chlorine dioxide (Wajon et
al., 1982).
4.3 Biodegradation
Bacteria play a major role in the degradation of phenol in
soil, sediment and water. The number of bacteria capable of
utilizing phenol is usually a small percentage of the total
population present in, for example, a soil sample (Hickman & Novak,
1989). However, repeated phenol exposure may result in acclimation
(the promotion of strains capable of utilizing phenol as food)
(Young & Rivera, 1985; Colvin & Rozich, 1986; Shimp & Pfaender,
1987; Wiggins & Alexander, 1988; Tibbles & Baecker, 1989).
Phenol may be converted by bacteria under aerobic conditions to
carbon dioxide (Southworth et al., 1985; Ursin, 1985; Aelion et
al., 1987; Dobbins et al., 1987; Aquino et al., 1988), and
under anaerobic conditions to carbon dioxide (Bak & Widdell, 1986;
Tschech & Fuchs, 1987) or methane (Healy & Young, 1979; Ehrlich et
al., 1982; Young & Rivera, 1985; Fedorak & Hrudey, 1986; Fedorak
et al., 1986). Benzoate, catechol, cis-cis-muconate,
ß-ketoadipate, succinate and acetate have all been identified as
intermediates in the biodegradation of phenol (Paris et al., 1982;
Krug et al., 1985; Fedorak et al., 1986; Knoll & Winter, 1987).
Some of the carbon derived from the degradation of phenol may be
incorporated into the bacterial biomass (Chesney et al., 1985).
Phenol may be degraded in its free form as well as after
adsorption onto soil or sediment, although the presence of sorbent
reduces the rate of biodegradation (Shimp & Young, 1987; Knezovich
et al., 1988).
When phenol is the only carbon source, it can be degraded in a
biofilm reactor with first-order kinetics at concentrations below
about 20 µg/litre at 10 °C. The first-order rate constants are 3 to
30 times higher than those of easily degraded organic compounds at
100- to 1000-fold higher concentrations (Arvin et al., 1991).
Reported phenol degradation rates suggest rapid aerobic degradation
in sewage (typically > 90% with an 8-h retention time), soil
(typically complete biodegradation in 2-5 days), fresh water
(typically complete biodegradation in < 1 day), and sea water
(typically 50% in 9 days) (Howard, 1989). Anaerobic biodegradation
is slower (Baker & Mayfield, 1980).
The contribution of bacteria to the overall rate of degradation
may be affected by a number of factors such as phenol concentration
(Baker & Mayfield, 1980; Ursin, 1985; Hwang et al., 1989),
temperature (Baker & Mayfield, 1980; Bak & Widdell, 1986; Hwang et
al., 1986; Thornton-Manning et al, 1987; Gurujeyalashmi & Oriel,
1989), sunlight (Hwang et al., 1986), soil depth (Dobbins et
al., 1987; Federle, 1988), the presence of other nutrients
required for bacterial growth (Rubin & Alexander, 1983; Fedorak &
Hrudey, 1986; Rozich & Colvin, 1986; Thorton-Manning et al.,
1987), the presence of other pollutants (Southworth et al., 1985;
Hoffmann & Vogt, 1988; Wang et al., 1988; Namkoong et al., 1989)
and bacterial abundance (Tranvik et al., 1991).
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air
No data are available for background levels of phenol in air,
away from emission sources. They are expected to be low (< 1 ng
phenol/m3) (RIVM, 1986).
Higher levels of phenol in air may be expected for urban areas,
mainly due to traffic emissions. Urban phenol concentrations have
been reported for Osaka, Japan (1-4 µg phenol/m3; Kuwata et al.,
1980), Nagoya, Japan (0.2-8 µg phenol/m3 with an average of 1.7 µg
phenol/m3; Hoshika & Muto, 1979, 1980), Paris, France (0.7-8 µg
phenol/m3; Hagemann et al., 1978), and Portland, USA (0.22 to
0.42 µg phenol/m3; Leuenberger et al., 1985). Despite
differences in analytical techniques, the first three series of
measurements showed good agreement. The Portland results were lower,
but came from air samples taken during rain periods; phenol was also
detected in rain water (see section 5.1.2).
Ambient air levels of phenol have been extensively monitored in
the highly industrialized and urbanized Upper Silesia region of
Poland (Sanitary Epidemiological Station, Katowice, 1991). Levels
during 1990 ranged from 3.8 to 26.6 µg/m3, the highest values
being in the areas of greatest industrial concentration.
Hoshika & Muto (1979, 1980) reported a phenol level of
approximately 190 µg/m3 "near" a phenolic resin factory (no
details). Kuwata et al. (1980) found phenol levels of 0.8-3.5
mg/m3 in foundry emissions (no details).
Based on limited data, median ambient atmospheric levels of
phenol (based on estimated 24-h averages) were estimated by
Brodzinsky & Singh (1982) to be 0.12 µg/m3 for urban/suburban
areas (7 samples) in the USA (which is lower than reported above for
several cities), and 104 µg/m3 (2-170 µg/m3) for source-
dominated areas (83 samples) in the USA.
5.1.2 Water and sediment
Levels of phenol dissolved in rain water from Portland, USA,
were found to range from 0.08 to 1.2 µg/litre and averaged above
0.28 µg/litre; gas phase concentrations ranged from 220-410 ng/m3
(Leuenberger et al., 1985).
Concentrations reported for surface water in the Netherlands
were 2.5-6.5 µg/litre for two major rivers, 0.3-7 µg/litre for
lakes, and 1.5 µg/litre for coastal waters (the given concentrations
include other phenolic substances) (RIVM, 1986). Industrial rivers
in the USA were reported to contain 0-5 µg/litre, but 3-24 µg/litre
was reported for Lake Huron. Phenol was also detected in 2/100 raw
water supplies in 1977 in the US EPA National Organics Monitoring
Survey (Howard, 1989).
Drinking-water levels of phenol in the USA have been reported
to be around 1 µg/litre or otherwise below the detection limit
(summarized by Howard, 1989). Phenol was detected (no quantitative
data) in drinking-water in the USA from 5 out of 14 drinking-water
plants surveyed and in Great Britain in 2 out of 4 sites (Fielding
et al., 1981). Higher groundwater levels have been reported
following industrial activity (e.g. 6.5-10 000 µg/litre in two
aquifers 15 months after a coal gasification project; summarized by
Howard, 1989). Phenol was detected at a maximum concentration of
1130 mg/litre in nine wells in Wisconsin after a spill, and was
detectable for at least 1.5 years after the spill (Delfino & Dube,
1976).
Phenol was not detected in water samples from three areas in
Japan analysed for an environmental survey; however, levels of
0.03-0.04 mg/litre were detected in 3 out of 9 bottom sediment
samples from the same regions (Fujii, 1978).
Other sediment concentrations reported were 13 000 µg/kg in
samples from Lake Huron, "not detected" in an unspecified industrial
river in the USA, < 1000 µg/kg (dry weight) as the median
concentration in 9% of sediment samples from 318 data points in the
USA, and 10 µg/kg (dry weight) in samples collected 6 km from a
wastewater treatment discharge zone in California (summarized by
Howard, 1989).
Phenol was detected in 63 out of 165 sediments from sampling
areas in the Puget Sound region (Tetra Tech. Inc., 1986). Half the
samples had a concentration of phenol below 40 µg/kg sediment (dry
weight); the maximum level was 1700 µg/kg.
Levels of phenol with means of 0.01-5.7 mg/litre (maximum up to
53 mg/litre) have been reported in effluents from various industrial
sources (summarized by Howard, 1989). Highest levels were associated
with the iron and steel industry. Limited quantitative data from the
VIEW Database (ATSDR, 1989) for ground water at hazardous waste
sites indicated maximum levels of 2.48 to 85 000 µg/litre (average
33 800 µg/litre, 6 data points).
No data have been found indicating the presence of phenol in
soil. Phenol is not likely to persist in soil because of rapid
biodegradation (section 4.3) or transport to ground water or air
(section 4.1).
5.2 Occupational exposure
Occupational exposure to phenol may occur during the production
of phenol and phenol derivatives, during the production of phenolic
resins and other products derived from phenol, during processing of
the latter materials, and during a number of other activities.
5.2.1 Production
Personal air samples of workers involved in the production of
phenol by the cumene process in the ex-USSR contained on average 5.8
mg phenol/m3. For workers occupied in the production of phenol
from chlorobenzene, the mean exposure level was 1.2 mg phenol/m3
(Mogilnicka & Piotrowski, 1974). Values reported in the same
publication for workers in two phenol resin-producing industries
were 0.6-3 mg phenol/m3.
5.2.2 Application of phenolic resins
Occupational exposure during the processing of phenolic resins
appears to be partly determined by the content of free phenol in the
applied resin (Bollig & Decker, 1980; Ryser & Ulmer, 1980).
In the wood industry, indoor phenol concentrations of 0.3 mg
per m3 (Winkler, 1981) and 1.5 mg/m3 (range 0.8-2.6) (Gilli et
al., 1980) have been reported. Concentrations in the breathing
zone of wood workers were 1.3-2.6 mg phenol/m3 (Gspan et al.,
1984). In another study, concentrations of < 0.04 to 1.9 mg
phenol/m3 were found at plywood plants (Kauppinen et al., 1986).
In iron and steel foundries, average hourly phenol
concentrations of 0.4-4.5 mg/m3 were reported in the manufacture
of moulds or kernels (Schütz & Wolf, 1980). Phenol concentrations of
1-4 mg/m3 were measured in a foundry in Osaka, Japan (Kuwata et
al., 1980). Local phenol concentrations were reported to be as
high as 75-420 mg/m3 due to the thermal degradation of the resin.
However, this effect of thermal degradation was not reflected in
hourly concentrations measured during the foundry process: values of
3-16 mg phenol/m3, with an average of 10 mg phenol/m3, were
reported by Schütz & Wolf (1980), and a maximum hourly average of
2.7 mg phenol/m3 was reported by Ryser & Ulmer (1980). (It is not
known whether these results were obtained in personal or area air
samples). Phenol concentrations during the operation of an electric
furnace in a steel factory in Pueblo, Colorado, USA, were 0.04, 0.18
and 0.20 mg/m3 in the vicinity of the furnace. General room air
samples taken during operation of a grey iron foundry were below the
detection limit (Gunter, 1987).
5.2.3 Other occupational situations
Exposure levels of 5-88 mg phenol/m3 have been reported for
employees in the ex-USSR who quenched coke with waste water
containing 0.3-0.8 g/litre phenol (Petrov, 1960).
Measurements at coal gasification and liquefaction plants in
the USA showed relatively low phenol concentrations (< 0.08 mg
per m3) at various sites (Dreibelbis & Hawthorne, 1985).
In a Japanese bakelite factory, area samples contained 0-12.5
mg phenol/m3 (Ohtsuji & Ikeda, 1972).
In a synthetic fibre factory in Japan, concentrations of 19 mg
phenol/m3 were measured, whereas in a USA fibrous glass wool
factory concentrations of 0.05-1.3 mg phenol/m3 were reported
(Dement et al., 1973; Ogata et al., 1986).
The concentrations of phenol in creosote vapour, analysed in
seven creosote impregnation plants in Finland, ranged from < 0.1 to
1.8 mg/m3 air (Heikkilä et al., 1987). The highest exposures
occurred during the cleaning of creosote warming chambers.
During the dissection of cadavers by dental students, phenol
breathing zone concentrations ranged from 5 to 19 mg phenol/m3.
(The high phenol concentrations resulted from the applied embalming
solution, in combination with inadequate ventilation) (Boiano,
1985).
5.3 General population exposure
5.3.1 Indoor air
Borovik & Dmitriev (1981) found a maximal concentration of 0.02
µg phenol/m3 in hospitals in the ex-USSR. It is, however, not
clear from where the phenol originated; it may have been used as a
disinfectant in these hospitals.
No information has been found with regard to phenol
concentrations in residential houses and apartments. Cigarette
smoking must be considered as the most important potential source in
dwellings. A distinction should be made between the main stream (the
smoke inhaled by the smoker) and the side stream (produced by the
smouldering cigarette itself). It was estimated that 0.01-0.22 mg
phenol per cigarette was released in the mainstream, while the
sidestream phenol content was 2.6 times higher. In the case of
various Japanese cigarettes, 0.3-0.4 mg phenol was emitted into air
during burning (Kuwata et al., 1980). For an unventilated room of
50 m3, the smoking of one cigarette would thus result in a phenol
concentration of 6-8 µg/m3.
5.3.2 Food and drinking-water
Phenol is found in smoked meat and fish products. The wood
smoke with which such products are treated contains, among other
ingredients, a wide range of phenols and phenol ethers, which
contribute significantly to the characteristic smoke aroma (smell
and taste) of the product.
Phenol is absorbed into the food products during smoking.
Quantitative data, however, are scarce, since phenols are usually
determined as a group. According to Toth (1982), the total phenol
content of smoked sausage is 70 mg/kg; Bratzler et al. (1969)
found a content of 37 mg/kg in the outer layer of the product, and
lower contents in the inner part. Luten et al. (1979) determined a
number of individual phenols in smoked herring and found a phenol
content, depending on the duration of smoking, of approximately
10-30 mg/kg. Potthast (1976, 1982) measured 2-18 mg/kg in smoked ham
and liver sausage.
If liquid smoke derivates are used in order to give a smoky
flavour to fish and meat products, the end product also contains
phenol. However, with regard to smell and taste aspects, phenol is
not the most important phenolic compound from wood smoke. In this
respect, methoxy and dimethoxy phenols are more important, together
with aliphatic fatty acids and carboxyl compounds.
Phenol may also enter food unintentionally by, for instance,
contamination in transport or from packaging materials, or contact
with other phenol-containing materials. However, these accidental
cases would probably be detected and lead to non-acceptance by the
consumer, owing to the conspicuous phenol smell and taste (see
section 8.3 for odour and taste thresholds).
Phenol has been found in botton pits fish from 5 sites in
Commencement Bay in Tacoma, USA, at a maximum average of 0.14 mg/kg
and an overall maximum of 0.22 mg/kg (Nicola et al., 1987).
Little information is available with respect to the occurrence
of phenol in drinking-water. Surface and ground waters intended for
the production of drinking-water in the Netherlands were reported to
contain 1-9 µg phenol/litre (phenol index, including other phenolic
compounds) (RIVM, 1986). Phenol was found in a domestic water supply
in the USA at a level of 1 µg/litre (Ramanathan, 1984). Cases of
drinking-water pollution with phenol have been reported in the UK
and the USA; the phenol water concentrations were reported to be
5-10 µg and 5-120 mg per litre, respectively (see section 8.1.2.2).
Chlorination of drinking-water may result in the formation of
chlorophenols from phenol, which greatly adds to the objectionable
smell and taste (Jarvis et al., 1985).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
Phenol and conjugated metabolites of phenol occur naturally in
animal and human tissue and can be detected in the urine, faeces,
saliva and sweat. The body's production of phenol depends on the
type of diet: a high protein or meat diet promotes phenol formation.
6.1 Absorption
Phenol is readily absorbed through all routes, such as the
lungs, intact and abraded skin, and the gastrointestinal tract of
both humans and animals (Von Oettingen, 1949; Deichmann & Keplinger,
1963).
6.1.1 Animal uptake studies
6.1.1.1 Pulmonary
There are no in vivo data on absorption of phenol following
inhalation exposure. However, in vitro studies by Hogg et al.
(1981), using 14C-phenol with excised trachea-lung preparations
and isolated perfused rat lung, demonstrated that phenol can be
rapidly and efficiently absorbed in the lungs.
6.1.1.2 Dermal
The extent of absorption of phenol through rabbit skin is more
strongly influenced by the area of the skin exposed than by the
concentration of the applied solution in water (Deichmann &
Witherup, 1944; Liao & Oehme, 1980).
In studies with the hairless mouse, phenol destroyed the
stratum corneum (Behl et al., 1983a). Similar effects were
reported by Huq et al. (1986) and Jetzer et al. (1986).
Absorption of phenol through thermally damaged mouse skin in vitro
was also reported to be greatly enhanced (Behl et al., 1983b). In
contrast, Deichmann et al. (1952) observed that injury of the
rabbit skin caused by phenol appeared to retard the rate of
absorption.
The permeability of mouse skin to phenol from aqueous solution
in vitro increased with increasing temperature of the carrier
solution (from 10 to 37 °C) (Jetzer et al., 1988).
Measurement of the permeation constant of phenol through
hairless mouse skin at 37 °C in vitro yielded a value of 18 800 ±
3000 cm/h (Huq et al., 1986; Jetzer et al., 1986).
6.1.1.3 Intestinal
When a single oral dose of 25 mg/kg body weight was
administered to rats, pigs or sheep, more than 95% was absorbed (Kao
et al., 1979).
In vitro studies showed that aqueous solutions of phenol
placed into ligated sections of the gastrointestinal tract had the
fastest absorption rate in the colon, followed by the ileum. The
absorption rate in the stomach was much slower (Deichmann &
Keplinger, 1963).
6.1.2 Human uptake studies
6.1.2.1 Pulmonary
The retention of phenol in the bodies of eight human volunteers
exposed to 6-20 mg/m3 by inhalation only for 8 h was 70%-80%
during the course of the study (Piotrowski, 1971). Ohtsuji & Ikeda
(1972) reported similar observations.
6.1.2.2 Dermal
Human skin absorption of phenol vapour (5-25 mg/m3) occurs
rapidly (Ruedemann & Deichmann, 1953). Fatal cases reflect the rapid
rate of absorption of phenol through the skin (Turtle & Dolan, 1922;
Duverneuil & Ravier, 1962; Hinkel & Kintzel, 1968; Lewin & Cleary,
1982). The retention in eight human volunteers, exposed to phenol
vapour at concentrations of 6-20 mg/m3, by skin only, for 6 h was
70-80% (Piotrowski, 1971). Piotrowski (1971) proposed the following
formula for calculating the absorption rate of phenol vapour through
the skin:
A = (0.35)C
where A is the amount of phenol absorbed in mg/h per unit area
and C is the phenol air concentration in mg/m3.
Concentrations of between 5 and 10% phenol denature epidermal
protein, and this can partly prevent absorption. The phenol-protein
complex is not stable and by dissociation of phenol the substance
may exert its action over a period of time (Schmidt & Maibach,
1981).
Phenol was detected in the urine of 4 out of 16 infants (2-5
months) with seborrhoeic eczema who were skin-painted twice daily
for 48 h with a commercial paint containing 4% (w/v) phenol and 8%
(w/v) resorcinol (Rogers et al., 1978). In adults, a single
topical application of 4 µg phenol/cm2 on 13 cm2 of the ventral
forearm, reportedly gave an absorption of 4.4% of the administered
dose (Feldman & Maibach, 1970). The period of exposure and the
concentration of phenol are both factors that determine the extent
of absorption (Piotrowski, 1971; Roberts et al., 1977;
Baranowska-Dutkiewicz, 1981).
In vitro studies have also shown that phenol from aqueous
solutions (1% w/v) readily penetrates human skin (Roberts et al.,
1977, 1978). A value of 8200 cm/h was obtained as the permeation
constant of phenol through human skin at 25 °C (Flynn & Yalkowsky,
1972). In an in vitro study with human abdominal skin, 10.9% of
the applied dose was absorbed. This study showed an excellent
qualitative, but a somewhat less accurate quantitative, agreement
between the in vivo and in vitro skin absorption of 12 compounds
(Franz, 1975).
6.2 Distribution
Phenol is rapidly distributed to all tissues in exposed
animals.
In rabbits, 15 min after oral administration of 0.5 g
phenol/kg, chemical analysis indicated that the liver contained the
highest concentration of total phenol followed by the central
nervous system, lungs and blood. After 82 min, phenol was fairly
uniformly distributed over all tissues. The proportion of free to
conjugated phenol changed with time, and, after 360 min, most of the
phenol was conjugated (Deichmann, 1944).
After a single oral administration of 14C-phenol (207 mg/kg)
to rats, the highest concentration ratios between tissue and plasma
were found in liver (42%), followed by spleen, kidney, adrenal,
thyroid and lungs, with a peak tissue level occurring after 0.5 h
(Liao, 1980; Liao & Oehme, 1981a).
Highest tissue residues were found after 2 h in the kidneys and
livers of mice and rats treated intravenously (Gbodi & Oehme, 1978;
Wheldrake et al., 1978; Greenlee et al., 1981).
6.3 Metabolic transformation
6.3.1 Metabolite identification
Studies employing several species have demonstrated that
conjugation with glucuronic acid and sulfate are major metabolic
pathways for phenol. Hydroxylation to hydroquinone and catechol also
occurs (Williams, 1938, 1959; Garton & Williams, 1949; Bray et
al., 1952a,b,c; Parke & Williams, 1953).
In vitro studies have shown the formation of 4, 4'-biphenol
and diphenoquinone by neutrophils, activated leucocytes and by
horseradish peroxidase following addition of phenol (Eastmond et
al., 1986).
Phenol metabolism in rabbits was studied by Deichmann &
Keplinger (1963). During the first 24 h following oral
administration of a sublethal dose of 300 mg phenol/kg body weight,
23% of the administered dose was recovered as exhaled carbon
dioxide. Trace amounts of catechol and hydroquinone were also
detected in the breath. Over the same period, 72% of the dose was
excreted in the urine (48% of which was excreted as free and 52% as
conjugated phenols), 1% was excreted in the faeces, 4% remained in
the carcass, and trace amounts were exhaled.
Oral administration of 14C-phenol (1.2 mg/kg) to rats
resulted in at least 80% excretion in urine within 24 h, with 68% as
phenyl sulfate and 12% as phenyl glucuronate (Edwards et al.,
1986).
A pronounced shift from sulfation to glucuronidation was
observed in rats after increasing the phenol dose (Koster et al.,
1981). This observed shift is apparently due to a saturation of the
overall sulfation process, rather than to a depletion of inorganic
sulfate (Weitering et al., 1979; Koster et al., 1981; Koster,
1982). A limited availability of 3-phosphoadenosine-5-phosphosulfate
may account for the decreased proportion of phenol conjugation to
sulphate at relatively high doses (Ramli & Wheldrake, 1981).
Repeated administration of phenol, however, did not affect
glucuronide synthesis in rats (Takemori & Glowacki, 1962).
The pig has limited ability for phenol sulfation. The domestic
cat lacks the ability for glucuronic acid conjugation of phenol. In
cats, phenyl phosphate has been detected as a metabolite in small
amounts, in addition to sulfate conjugates (Capel et al., 1974;
French et al., 1974).
Following oral administration of 14C-phenol (0.01 mg/kg) to
three men, 90% of the dose was excreted in the urine within 24 h,
mainly as phenyl sulfate (77%) and phenyl glucuronide. Small amounts
of guinol sulfate and guinol glucuronide were also present (Capel
et al., 1972b).
Several investigators have confirmed the above-mentioned
results using in vitro methods (DeMeio & Arnolt, 1944; Capel et
al., 1972b; Shirkey et al., 1979; Hogg et al., 1981; Koster
et al., 1981; Sawahata & Neal, 1983).
6.3.2 Covalent binding to macromolecules
Early pharmacokinetic studies (measuring distribution volumes)
in dogs, pigs and goats suggested that tissue binding occurs (Oehme,
1969). Further animal studies have indicated that phenol and/or its
metabolites bind covalently to tissue protein, mainly in the liver
(Bolt, 1977; Illing & House, 1980; Jergil et al., 1982; Smart &
Zannoni, 1984). Binding to rabbit bone marrow mitochondrial DNA in
studies with isolated cells has also been reported (Rushmore et
al., 1984). in vivo and in vitro studies have demonstrated
covalent binding of radiolabelled phenol to plasma proteins from
humans, dogs, rats and trout (Liao, 1980; Liao & Oehme, 1981a,b;
Judis, 1982; Schmieder & Henry, 1988). Reactive phenol metabolites
formed by peroxidases bind readily to proteins (Eastmond et al.,
1986, 1987a) and DNA (Subrahmanyam & O'Brien, 1985).
6.3.3 Location
Quantitatively, the most important sites of phenol conjugation
are the liver, lung and gastrointestinal mucosa. The relative roles
played by these tissues depend on the route of administration and
the dose.
The liver is an important site of phenol metabolism. After
direct administration of phenol into the hepatic circulation, the
liver showed considerable first-pass metabolism in rats (Cassidy &
Houston, 1980; Houston & Cassidy, 1982). Phenol-metabolizing enzymes
have been detected in rabbit hepatic microsomes (Koop et al.,
1989).
Other tissues, such as lungs, intestines and kidneys, also play
an important role in phenol metabolism (Quebbemann & Anders, 1973;
Powell et al., 1974; Houston & Cassidy, 1982). Phenol
sulfotransferases, which catalyse phenol sulfation, occur in a
variety of human tissues (intestinal wall, lungs, platelets, adrenal
glands, brain, placenta, etc.) (Campbell et al., 1987; Gibb et
al., 1987). After oral uptake of phenol, there is a very large
first-pass metabolism in the intestines. The lungs also show
considerable first-pass metabolism (as was established after direct
administration into the pulmonary circulation of rats) (Cassidy &
Houston, 1980; Houston & Cassidy, 1982). Due to saturation of
hepatic enzymes, extrahepatic tissues play an increasing role in the
conjugation of phenol as the dose of phenol increases; at doses
higher than 5 mg/kg body weight, intestinal conjugation in rats
exceeds the contribution of the hepatic and pulmonary enzymes
(Cassidy & Houston, 1984).
Myeloperoxidases isolated from human neutrophils and
peroxidative enzymes from activated human leucocytes mediate the
formation of reactive phenol metabolites including 4,4'-biphenol and
diphenoquinone. Myeloperoxidase-mediated hydroxylation occurs in
addition to hepatic cytochrome P-450 oxidation. In several species,
myeloperoxidase activity has been reported in bone marrow, where it
may play a role in phenol metabolism and toxicity (Eastmond et
al., 1986, 1987a; Subrahmanyam et al., 1991).
6.4 Elimination and excretion
Urinary excretion is the major route of phenol elimination in
animals and humans. The rate of excretion varies with dose, route of
administration and animal species (Deichmann, 1944; Capel et al.,
1972a,b). Of 18 animal species studied by Capel et al. (1972a,b),
the 24-h urinary excretion of phenol was greatest in the rat (95% of
the 25 mg/kg body weight oral dose) and the lowest in the squirrel
monkey (only 31% of the dose). Liao & Oehme (1981a,b) reported a
half-life of 4 h in rats.
Five days after oral gavage with 14C-phenol (0.1 mg/kg body
weight), only 0.3% of the applied dose was retained in rats (Freitag
et al., 1985).
Only minor amounts of unchanged phenol are excreted in exhaled
air or in faeces (Deichmann & Keplinger, 1963). Less than 1% of an
orally administered dose of 300 mg phenol/kg body weight to rabbits
was found in the faeces after 24 h (Deichmann, 1944).
Phenol conjugates may also be excreted in the bile of rats
(4.6% of a 50 mg/kg dose) (Abou-el-Makarem et al., 1967). It has
been suggested that biliary excretion of phenol plays an important
role when urinary excretion is impeded. Rats, whose kidneys were
ligated, showed a marked increase in biliary excretion of phenol
metabolites (Weitering et al., 1979). Furthermore, it has been
reported that phenol and its metabolites can undergo enterohepatic
circulation in rats (Gbodi & Oehme, 1978).
Urinary excretion of phenol in human volunteers exposed to
phenol vapour via inhalation (chamber studies) or skin, occurred
with an excretion rate constant of k = 0.2/h. For a one-compartment
model, this corresponds to a half-life of approximately 3.5 h
(Piotrowski, 1971).
6.5 Biological monitoring
The US ACGIH has listed a biological exposure index for phenol
of 250 mg/g creatinine for end-of-shift urine samples (ACGIH, 1991).
The excretion of phenol and phenol conjugates in the urine may
be used as an index of exposure, but it should be noted that there
are other causes that may lead to phenol excretion in the urine. One
of these is benzene exposure; other possible significant sources are
food and drugs (Docter & Zielhuis, 1967; Ikeda & Ohtsuji, 1969;
Fishbeck et al., 1975; Paradowski et al., 1981). Elevated
urinary phenol excretion is thus not a specific index of exposure to
phenol. Furthermore, the large range of "normal" urine values
(phenol concentrations have been found to vary from 0.5 to 81.5
mg/litre) (Deichmann & Schafer, 1942; Docter & Zielhuis, 1967;
Piotrowski, 1971; Gspan et al., 1984; Pekari et al., 1992) would
appear to limit the usefulness of urinary phenol excretion as an
accurate index of low occupational exposure levels.
In volunteers, after a single 8-h exposure to phenol vapour
concentrations of up to 6.8 mg/m3, the phenol excretion in urine
increased up to a maximum of 100 mg total phenol/litre (Piotrowski,
1971). In workers occupationally exposed to 10 mg phenol/m3,
concentrations in urine of up to 262 mg/litre were reported
(Ohtsuji & Ikeda, 1972). However, another recent study, using more
specific methods of analysis, showed good correlation (R=0.91) between
exposure levels in the range 5-17 mg/m3 and the total concentration
of phenyl sulfate and phenyl glucuronide in the urine at the end of the
workshift (Ogata et al., 1986).
7. EFFECTS ON LABORATORY MAMMALS, AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 LD50 values
After oral administration of phenol to mice, rats and rabbits,
LD50 values ranged from 300-600 mg phenol/kg body weight. No
LC50 values have been reported in the published literature.
However, after inhalation of 900 mg phenol/m3 by rats for 8 h, no
deaths were observed. The dermal LD50 (by occlusive and
non-occlusive techniques) was 670 mg/kg body weight for rats and
850-1400 mg phenol/kg body weight for rabbits. LD50 values for
intraperitoneal injection were in the range of 127-223 mg phenol/kg
body weight for rats.
A summary of LD50 values is given in Table 5.
7.1.2 Effects
The acute lethality of phenol, associated with exposure to high
concentrations, is generally attributed to a depressing effect on
the central nervous system (see also section 7.8.1). The clinical
effects of phenol poisoning are independent of the route of
administration. Reported symptoms include neuromuscular
hyperexcitability, including twitching and severe convulsions. Heart
rate at first increases, then becomes slow and irregular. Blood
pressure at first increases slightly, then falls markedly.
Salivation, marked dyspnoea and a decrease in body temperature are
also among the effects reported (Deichmann & Witherup, 1944; Von
Oettingen & Sharples, 1946; Farquharson et al., 1958; Ernst et
al., 1961; Deichmann & Keplinger, 1963; Oehme & Davis, 1970;
Pullin et al., 1978; Liao & Oehme, 1980; Reid et al., 1982).
After oral ingestion, the mucous membranes of the throat and
oesophagus showed swelling, corrosion, and necrosis, with
haemorrhages (Deichmann & Keplinger,1963).
In a study by Schlicht et al. (1992), female Fischer-344 rats
were administered 0, 12, 40, 120 or 224 mg phenol/kg body weight by
gavage in a water vehicle. Animals were examined for clinical signs,
and neurotoxicity and systemic (liver, kidney, adrenal and thymus)
effects, 4-20 h after treatment. Tremors were observed 1-2 min after
dosing in the two highest dose groups. The pupil response to light
(miosis) was significantly inhibited at all dose levels at 24 h
after exposure. Locomotor activity was reduced at 224 mg/kg. At this
dose level, 2/6 animals had hepatocyte necrosis, 4/6 had renal
vascular stasis and 4/6 had necrosis of the thymus. At 120 mg/kg,
liver necrosis was present in 1/7 animals, as was necrosis of the
thymus gland.
Table 5. Acute animal toxicity of phenol LD50 values
Species Route of LD50 values Vehicle Reference
administration (mg/kg body
weight)
Mouse oral 300 Von Oettingen &
Sharples (1946)
Mouse oral 427 Kostovetskii &
Zholdakova (1971)
Rat oral 340-530 2-7% in
water Deichmann &
Witherup (1944)
Rat oral 512 Kostovetskii &
Zholdakova (1971)
Rat oral 445-520 water Thompson & Gibson
(1984)
Rat oral 400 water Schlicht et al.
(1992)
Rat dermal 670 undiluted Conning & Hayes
(570-780) (1970); Brown et al.
(1975)
Rat intraperitoneal 127-223 water or Thompson & Gibson
undiluted
(1984)
Rabbit oral 400-600 2-7% in Deichmann &
water Witherup (1944)
Rabbit dermal 850 Flickinger (1976)
(600-1200)
Rabbit dermal 1400 Vernot et al. (1977)
(740-2670)
In various animal species, inhalation of phenol adversely
affected the lungs, causing hyperaemia, infarcts, bronchopneumonia,
purulent bronchitis and hyperplasia of the peribronchial tissues
(Von Oettingen, 1949).
Sensory irritation was measured in mice by the Alarie assay. A
50% decrease in respiratory rate (RD50) was found at 638 mg
phenol/m3 (De Ceaurriz et al., 1981).
Ocular and nasal irritation, tremors and incoordination were
reported in rats exposed via inhalation to 906 mg/m3 for 8 h
(Flickinger, 1976).
Other pathological abnormalities induced by phenol by various
routes of administration included demyelination of nerve fibres (see
also section 7.8.1), myocardial degeneration and necrosis (Deichmann
& Keplinger, 1963; Liao & Oehme, 1980). Kidney damage (vacuolization
and enlargement of cells) and liver damage (e.g., enlargement of
hepatic cells) were also observed (Oehme & Davis, 1970; Coan et
al., 1982). Urine was usually dark or "smoky" in appearance,
probably due to oxidation products of phenol (Solliman, 1957).
7.2 Short-term exposure
7.2.1 Oral exposure
In a study by Schlicht et al. (1992), groups of eight female
Fischer-344 rats received oral doses of phenol in a water vehicle of
0, 4, 12, 40 or 120 mg/kg body weight daily for 14 days. Tremors
were apparent only after the first dose at the highest level.
Exposure to 120 mg/kg per day was lethal to all rats within 11 days.
The pupil response (miosis) was decreased one day after the last
dose for all but the highest surviving dose group (the incidences
were 100%, 50%, 62% and 76% for the 0, 4, 12 and 40 mg/kg groups,
respectively). Locomotor activity was not affected after the 4th,
9th or 14th dose. No hepatic effects were observed at 40 mg/kg per
day, while 3/8 animals had renal vascular stasis. There were no
histological effects at 12 mg/kg per day. At 40 mg/kg per day, the
pathological changes in the kidneys included two animals with
tubular degeneration in the papillar region, and one with protein
casts in the tubules. The pathological report attributed these
findings to decreased vascular perfusion (MacPhail, personal
communication to the IPCS).
Rats were administered, by gavage, 20 daily doses of 10, 50 or
100 mg phenol/kg body weight. At necropsy, slight effects on liver
and kidneys were reported at 100 mg phenol/kg body weight (Dow
Chemical Company, 1976).
Rats receiving 50 or 100 mg phenol/kg body weight, by gavage,
over a 6-month period (135 doses, presumably daily, 5 days/week)
were reported to show slight to moderate kidney damage.
Administration of 100 mg phenol/kg body weight apparently resulted
in slight liver changes (Dow Chemical Company, 1976).
In a range-finding study, carried out prior to a long-term
carcinogenicity study, mice and rats were provided with tap water
containing 0, 100, 300, 1000, 3000 or 10 000 mg phenol/litre for 13
weeks. Mean body weight gain was decreased only in mice and rats
receiving 10 000 mg phenol/litre (NCI, 1980). In these
drinking-water studies, the highest daily doses were calculated to
be approximately 2000 mg phenol/kg body weight for mice and 1000 mg
phenol/kg body weight for rats.
Phenol was provided to rats in drinking-water for 12 months at
0, 800, 1200, 1600, 2000 and 2400 mg phenol/litre. Depressed weight
gain was observed in rats receiving doses > 2000 mg/litre. The
corresponding daily dose was calculated by the authors of the study
to be > 200 mg/kg body weight (Deichmann & Oesper, 1940).
7.2.2 Dermal exposure
In a study by Deichmann et al. (1950), rabbits were exposed
to 1.18-7.12% phenol in water (64-380 mg phenol/kg body weight) for
5 h/day, 5 days/week, for 18 days. Dose-related systemic effects
(tremors, death) were observed in rabbits exposed to > 2.37%
phenol (130 mg phenol/kg body weight), while skin irritation
(hyperaemia, tissue necrosis) occurred at doses of > 3.56% phenol
(190 mg phenol/kg body weight). This effect was particularly
apparent when the application sites were bandaged.
7.2.3 Inhalation exposure
No studies reported or conducted according to contemporary
standards were available.
In a study by Deichmann et al. (1944), rats, rabbits and
guinea-pigs were exposed to concentrations of 100-200 mg phenol
vapour/m3, 7 h/day, for 5 days/week. Rats exposed for a period of
74 days did not show any gross or microscopic evidence of injury.
Rabbits survived a 3-month exposure but, at autopsy, lung and heart
damage and indications of liver and kidney damage were found.
Guinea-pigs were the most susceptible. Five out of twelve died after
12 days of exposure, and the remaining seven were killed after 29
days of exposure. Prior to death, guinea-pigs showed weight loss,
respiratory difficulties, and signs of paralysis. At autopsy, there
was evidence of acute lobular pneumonia, vascular damage, and
hepatic and renal damage; the total (free and conjugated) phenol
content of the blood was 14 mg/litre. The rabbits had similar, but
less severe, symptoms.
Groups of 10 monkeys, 50 rats and 100 mice were exposed to 19
mg phenol/m3, 8 h/day, 5 days/week for 90 days. Concurrent control
groups were exposed to fresh air only. No deaths occurred and there
was no reduction in weight gain of treated animals. There were no
statistically significant adverse effects observed when the animals
were assessed by a stress test involving swimming performance. A
range of clinical chemistry, haematology and urinalysis parameters
were not affected by exposure to phenol. Routine histology was
performed on the liver, lungs, kidneys, brain and heart. The results
of the percentage of animals showing evidence of "pathological
change" indicated effects in the liver and kidneys of exposed
animals. However, the author of the study concluded that no clinical
or pathological changes occurred that were of toxicological
importance. It is not clear if the upper respiratory tract was
examined in this study in order to look for evidence of irritation
(Sandage, 1961).
Continuous exposure to 100 mg phenol/m3 for 15 days
significantly affected the central nervous system of rats, as was
demonstrated by their performance in the "tilted plane" test. Plasma
levels of potassium, magnesium, lactate dehydrogenase, aspartate
aminotransferase (ASAT), alanine aminotransferase (ALAT) and
glutamate dehydrogenase were elevated. Haemoglobin, haematocrit, and
plasma sodium, calcium and chloride levels were unaffected (Dalin &
Kristofferson, 1974).
7.2.4 Subcutaneous exposure
Subcutaneous exposure to phenol was studied principally to
obtain information about neurological or haematopoietic effects (see
sections 7.8.1 and 7.8.2). No other effects were reported.
7.2.5 Ear exposure
Instillation of phenol (form and amount not specified) into the
inner ear round window of Sprague-Dawley rats caused morphological
damage to the organ of Corti in the basal coil. The outer hair cells
appeared to be more sensitive to phenol than the inner hair cells,
which were mostly intact. As a result of the damage, impairment of
inner ear function was noted (as determined by auditory brain stem
recordings) which was regressive for lower sound frequencies, but
appeared to be permanent for higher frequencies (Anniko et al.,
1988).
7.3 Skin and eye irritation; sensitization
Local damage to the skin, following exposure to phenol, was
found to include erythema, inflammation, discoloration, eczema,
papillomas and necrosis (Deichmann, 1949; Deichmann et al., 1950;
Conning & Hayes, 1970; Pullin et al., 1978). For example, in
rabbits, 0.5 g phenol, moistened with physiological saline, produced
necrosis of both the intact and abraded skin (Flickinger, 1976).
Solutions of 10-14% (v/v) phenol in water have been reported to
cause transient delayed erythema (after 0.5-5 h) and acute vascular
permeability, as assessed by exudation of intravenously injected
Evans blue, in guinea-pigs after dermal treatment for 1 min (Steele
& Wilhelm, 1966).
In one study, an increase in ear thickness was used as an index
of skin irritation (inflammation). Maximal responses to phenol were
observed one hour after application of 1-2 mg phenol to the ear of
female ICR mice. Significant thickening could still be detected 6
weeks after exposure (Patrick et al., 1985).
When phenol, in glycerine dilutions down to 10% or 5% aqueous
solutions, was applied to the rabbit eye, severe damage (complete
destruction to opaque corneas) was seen. Immediate water irrigation
was very effective in preventing the opacity. A delay of 10 seconds
reduced this effectiveness (Murphy et al., 1982).
Fourteen days after the application of 0.1 g phenol to the
rabbit eye, all eyes exhibited keratoconus and pannus formation
(Flickinger, 1976).
Phenol gave negative results in a Magnussen and Kligman skin
sensitization test (Itoh, 1982).
7.4 Long-term exposure
No adequate data are available. Studies on carcinogenicity are
presented in section 7.7.
7.5 Reproduction, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
No adequate studies conducted according to current protocols
are available.
Heller & Pursell (1938) exposed rats to 100-12 000 mg
phenol/litre drinking-water, corresponding to calculated approximate
daily oral doses of 10-1200 mg phenol/kg body weight. General
appearance, growth and fecundity were normal for rats exposed to
100-1000 mg/litre for five generations and to 3000 or 5000 mg/litre
for three generations. Stunted growth was noted in the offspring of
rats exposed to 7000 mg/litre. Many of the offspring died at levels
of 8000 mg/litre because of maternal neglect. At 10 000 mg/litre,
the offspring died at birth, and at 12 000 mg/litre there was no
reproduction.
7.5.2 Embryotoxicity/teratogenicity
7.5.2.1 In vivo studies
Phenol was evaluated for maternal and developmental toxicity in
timed-pregnant Sprague-Dawley rats (20-22 confirmed pregnancies per
group). Distilled water (vehicle) or phenol (30, 60 or 120 mg/kg per
day) was administered daily by gavage in a volume of 5 ml/kg of body
weight throughout the period of major organogenesis (gestational
days 6-15). Dams were weighed on the day of sperm detection
(gestational day 0), prior to daily dosing, and at termination
(gestational day 20); observations for clinical signs of toxicity
were conducted during the treatment period. At termination
(gestational day 20), maternal liver weight, gravid uterine weight
and status of uterine implantation sites (i.e. number of implants,
resorptions, late fetal deaths and live fetuses) for each dam were
recorded. Each live fetus was weighed, sexed and examined for
external morphological abnormalities. Visceral examination of each
fetus was performed using a fresh tissue dissection method;
approximately one-half of the fetal heads from each litter were
fixed (Bouins' solution) and sectioned free-hand for examination of
internal structures; carcasses (one-half without heads) were cleared
and stained with Alizarin Red S prior to skeletal examination. All
control and phenol-treated dams survived to scheduled sacrifice, and
no distinctive treatment-related signs of toxicity were noted.
Pregnancy rates at termination were high (95-100% per group) and no
litters were totally resorbed, so that a total of 20-22 live litters
per group (268-293 fetus per group) was available for examination.
No significant dose-related changes were noted for the following
end-points: maternal body weight (gestational day 0, 6, 11, 15 or
20), maternal body weight gain (treatment period, gestational period
or gestational period corrected for gravid uterine weight), maternal
liver weight, gravid uterine weight, prenatal mortality, live litter
size or incidence of morphological abnormalities (malformations or
variations). However, average fetal body weight per litter was
significantly reduced at the high-dose (93% of average control
weight) (Jones-Price et al., 1983a).
In a study by Kavlock (1990), phenol was administered by oral
gavage to groups of Sprague-Dawley rats on day 11 of gestation (day
1 : sperm plug) at 0, 100, 333, 667 and 1000 mg phenol/kg body
weight. The vehicle used in this study was a 4:4:1:1 mixture of
water, Tween 20, propylene glycol and ethanol. Maternal toxicity
(decreased weight gain) was seen at the two highest doses. Offspring
viability and growth were not affected up to postnatal day 6,but
hind limb paralysis was observed in some offspring in the two
highest dose groups.
In a screening assay, groups of 17-21 Fischer-344 rats received
0, 40 or 53.3 mg phenol/kg body weight by gavage in water on
gestation days 6-15. There were no significant effects on maternal
body weight gain. One of 15 pregnant females resorbed the entire
litter at 40 mg/kg and 2 of 16 did so at 53.3 mg/kg (there were no
similar effects in 153 control litters in the study). All three
females had severe respiratory syndromes (rales and dyspnoea). One
high-dose female with symptoms of respiratory toxicity delivered a
low weight litter that had poor viability. Kinked tails were present
in 2 of 4 surviving pups in that litter. Litter size on postnatal
days 1 and 6 was significantly reduced at 53.3 mg/kg but not at 40
mg/kg. There were no effects on pup body weights on postnatal days 1
or 6 (Narotsky & Kavlock, 1993).
Phenol was evaluated for maternal and developmental toxicity in
timed-pregnant Swiss albino (CD-1) mice (22-29 confirmed pregnancies
per group). Distilled water (vehicle) or phenol (70, 140 or 280
mg/kg per day) was administered daily by gavage in a volume of 10
ml/kg of body weight throughout the period of major organogenesis
(gestational days 6-15). Dams were weighed on the day of vaginal
plug detection (gestational day 0), prior to daily dosing
(gestational days 6-15), and at termination (gestational day 17);
observations for clinical signs of toxicity were conducted during
the treatment period. Evaluation of maternal and developmental
end-points at termination (gestational day 17) were the same as for
rats (see description from the study by Jones-Price et al., 1983a,
above). Toxicity observed at the high-dose level included 11%
mortality (4/36 treated females), clinical signs (especially tremor
and ataxia), reduced maternal body weight (gestational day 17),
reduced maternal body weight gain (treatment period, gestational
period and gestational period corrected for gravid uterine weight),
and a trend only toward reduced maternal liver weight. Pregnancy
rates at termination ranged from 71 to 84%; no litters were totally
resorbed so that 22-29 live litters (214-308 fetuses) were available
for examination. No dose-related changes were noted for prenatal
mortality, live litter size or incidence of morphological
abnormalities, except for an apparent increase in cleft palate
(8/214 fetuses in the high dose versus 0/308 among controls). (It
should be noted that cleft palate is a malformation to which the
CD-1 mouse is predisposed under conditions of maternal stress).
Average fetal body weight per litter was significantly reduced (82%
of average control weight) in the highest dose group (Jones-Price
et al., 1983b).
In a study by Minor & Becker (1971), groups of Sprague-Dawley
rats were given 20, 63, or 200 mg phenol/kg body weight
intraperitoneally on days 9-11 or 12-14 of gestation. Fetal body
weight was reduced in the highest dose group treated on days 12-14.
No gross anomalies were observed, and intrauterine death was not
increased at any dose level.
7.5.2.2 In vitro studies
In the chick embryotoxicity screening test (CHEST), 130
substances were tested. For each compound, 120 selected White
Leghorn Fowl embryos, aged 1.5, 2, 3 and 4 days of incubation, were
used. Phenol did not exhibit embryotoxic properties in this test up
to 100 µg, and was one of the least embryotoxic compounds tested
(Jelinek et al., 1985).
In a study by Oglesby et al. (1992), phenol was added to
cultures of five rat embryos on gestational day 10 at
concen-trations of 0 to 100 µg/ml. Embryos were examined 42 h later
for viability, growth and morphology. Viability was not affected at
any concentration, but a low incidence of tail defects was observed
at 100 µg/ml, and embryonic growth was decreased at 75 and 100
µg/ml. When hepatocytes isolated from pregnant rats were co-cultured
with the embryos, the toxicity to the embryos was increased. Tail
defects were observed at 25 and 50 µg/ml, and growth was reduced at
these concentrations. Without the presence of hepatocytes, phenol
was the least toxic of 13 para-substituted phenols tested in this
system; however, it was the only one which became more embryotoxic
when hepatocytes were present.
When phenol was added to cultures of human embryonic palatal
mesenchyme cells, cell growth was 50% inhibited at a concentration
(IC50) of 0.8 mM (78 µg/litre) (Pratt & Willis, 1985).
7.6 Mutagenicity and related end-points
Data on mutagenicity and related end-points are summarized in
Tables 6, 7, 8 and 9, respectively.
7.6.1 Mutagenicity studies
7.6.1.1 Bacterial systems
Phenol has been tested for mutagenicity by a number of authors
in various strains of Salmonella typhimurium and was shown to be
reproducibly negative, both with and without metabolic activation
(Epler et al., 1979; Gilbert et al., 1980; Rapson et al.,
1980; Pool & Lin, 1982; Haworth et al., 1983). A positive effect
was observed in strain TA98 in the presence of an exogenous
metabolic activation system in a study employing a modified culture
medium (Wild et al., 1980; Gocke et al., 1981).
A positive effect was reported for phenol in a fluctuation test
with strain TA100 after metabolic activation, but no data were given
on toxicity (Koike et al., 1988; abstract). A positive result was
reported in a mutation test with Escherichia coli B/Sd-4; however,
the applied dose levels were highly toxic (Demerec et al., 1951).
7.6.1.2 Non-mammalian eukaryotic systems
Negative data were obtained in the absence of exogenous
metabolic activation with the eukaryotic microorganism
Saccharomyces cerevisiae. At high doses and in the presence of an
activation system, a positive result was obtained (Cotruvo et al.,
1977). Phenol induced mitotic segregation in Aspergillus nidulans
(Crebelli et al., 1987).
Table 6. Tests for genotoxicity in bacteria
Species Strain Measured end-point Test conditions Metabolic Resultsb Reference
activationa
Escherichia coli Sol-4 reverse mutation 0.1-0.2%; 3-24 h - + Demerec et al.
(survival less than 2%) (1951)
AB1899 nm filamentation 10-500 µg/ml; 3-4 h - Nagel et al.
(1982)
Salmonella TA100 reverse mutation fluctuation test - - Koike et al.
typhimurium 0-500 ng/well + (rat) + (no data (1988)
on toxicity)
TA98 reverse mutation 1000-fold concentration + and - - Epler et al.
TA100 range in DMSOc (1979)
TA1535 reverse mutation 0-100 µg/plate - - Gilbert et al.
(1980)
TA1538 reverse mutation 0-50 µg/plate - -
TA98 reverse mutation 0-3333 µg/plate in - - Haworth et al.
TA100 DMSO; preincubation + (rat) - (1983)
TA1535 + (hamster) -
TA1537
TA98 reverse mutation 0-3333 µg/plate in - - Haworth et al.
TA100 preincubation H2O; + (rat) - (1983)
TA1535 + (hamster) -
TA1537
TA98 reverse mutation 0.5-5000 µg/plate in + and - (rat) - Pool & Lin
TA100 DMSO (5000 µg toxic) (1982)
TA1535
TA1537
TA1538
Table 6 (contd).
Species Strain Measured end-point Test conditions Metabolic Resultsb Reference
activationa
TA100 reverse mutation 0.1-1000 µg/plate - - Rapson et al.
(1980)
TA98 reverse mutation 0-100 µmol/plate (99.5% - - Wild et al.
purity with 0.15% + (rat) + (1980)
cresols as main impurity,
non-standard media)
a + = present; - = absent
b + = positive; - = negative
c DMSO = dimethyl sulfoxide
Table 7. Tests for genotoxicity in non-mammalian eukaryotic systems
Species Strain Measured end-point Test conditions Metabolic Resultsa Reference
activation
Fungi
Saccharomyces D3 mitotic recombination 10-5, 10-3 dilution of - - Cotruvo et al.
cerevisiae phenol in saline + (rat) + (1977)
Aspergillus mitotic segregation 5-20 mM - + Crebelli et al.
nidulans (1987)
Insects
Drosophila Oregon-R SLRLb phenol vapour 24 h; - Sturtevant
melanogaster 0.2, 0.25, 0.5% in - (1952)
saline, injection
0.01, 0.1, 1.2% in -
Holtfreter solution;
vaginal douch
Berlin K SLRL 50 nM in 5% saccharose; - Wild et al.
feeding, 3 broods F1 (1980); Gocke
generation et al. (1981)
injection - Woodruff et al.
(1985)
Fish
Salmo gairdneri chromosomal aberrations 0.3-0.6 µl/litre, 72 h + Al-Sabti (1985)
a + = positive; - = negative
b SLRL = sex-linked recessive lethal mutations
Table 8. In vitro phenol genotoxicity in mammalian cells
Species Cell type End-pointa Conditions Activationb Resultc Reference
Chinese hamster CHO-WBL CA 500-800 µg/ml - - Ivett et al. (1989)
2000-3000 µg/ml + (rat) +
Chinese hamster V79 lung forward mutation 0-500 µg/ml + (mouse) + Pashin & Bakhitova
HPRT (500 µg/ml toxic) (1982)
Chinese hamster CHO-WBL SCE 300-400 µg/ml - + Ivett et al. (1989)
2000-3000 µg/ml + (rat) +
Chinese hamster V79 lung intercellular not reported - - Chen et al. (1984)
communication
V79 lung intercellular 250 µg/ml - - Malcolm et al. (1985)
communication
V79 lung intercellular 10-75 µg/ml - - Bohrman et al. (1988)
communication
Mouse L5178Y lymphoma forward mutation 600-1800 µg/ml + and - ? McGregor et al.
TK (1988)
L5178Y forward mutation 180-890 µg/ml - + Wangenheim &
(530 µg/ml toxic) Bolcsfoldi (1988)
5.6-41 µg/ml + (rat) +
(20 µg/ml toxic)
Mouse L5178Y DNA synthesis 9.4-940 µg/ml - + Pellack-Walker
inhibition et al. (1985)
L5178Y DNA strand breaks 16-470 µg/ml - - Garberg et al.
16-470 µg/ml + (rat) + (1988)
Table 8 (contd).
Species Cell type End-pointa Conditions Activationb Resultc Reference
L5178Y DNA strand breaks 94 µg/ml - - Pellack-Walker & Blumer
(1986)
Human T-lymphocytes SCE 0.47-282 µg/ml - + Erexson et al. (1985)
lymphocytes SCE 188 µg/ml - - Jansson et al. (1986)
lymphocytes SCE 1.7-470 µg/ml + (rat) + Morimoto & Wolff
(470 µg/ml toxic) (1980)
lymphocytes SCE 282 µg/ml + (rat) + Morimoto et al.
(1983)
Human fibroblast DNA repair 0.094-9400 µg/ml + Poirier et al. (1975)
HeLa DNA synthesis 188 µg/ml + (rat) + Painter & Howard
inhibition (1982)
WI-38 DNA synthesis 0.094-9400 µg/ml + Poirier et al. (1975)
inhibition
a CA = chromosome aberrations; HPRT = hypoxanthine guanine phosphoribosyl transferase locus; TK = thymidine kinase locus;
SCE = sister chromatid exchange
b - = absent; + = present;
c - = negative; + = positive
Table 9. Phenol genotoxicity in in vivo mammalian systems
Species/Strain Measured end-point Test conditions Remarks Resultsa Reference
(sampling times)
Mouse/CD-1 micronuclei in bone 265 mg/kg, oral bone marrow + Ciranni et al.
marrow (0, 18, 24, 42 or 48 h) depression (1988a)
Mouse/CD-1 micronuclei in maternal bone gestation day 13, 265 mg/kg, maternal bone marrow + Ciranni et al.
marrow and fetal liver oral (15, 18, 24, 30, 36 or 40 h) depression (1988a)
Mouse/CD-1 micronuclei in bone 250 mg/kg, oral (30 h) convulsive seizures - Gad-El Karim et
marrow al. (1986)
Mouse/CD-1 micronuclei in bone 265 mg/kg, i.p. bone marrow + Ciranni et al.
marrow (18, 24, 42 or 48 h) depression (1988a)
Mouse/CD-1 micronuclei in bone 40, 80 or 160 mg/kg, i.p. no bone marrow - Barale et al.
marrow (18 h) depression (1990)
Mouse/NMRI micronuclei in bone 47, 94 or 188 mg/kg, i.p. no information on - Gocke et al.
marrow 0, 24 h (30 h) toxicity (1981)
Mouse/Porton chromosomal aberrations in 2 ml of 0.08, 0.8 or 8 mg/litre + Bulsiewicz (1977)
spermatogonia, primary solution, oral, daily for five
spermatocytes generations
Rat/Sprague- chromosomal aberrations in 72-180 mg/kg, i.p. LD1-LD30, no change - Thompson &
Dawley bone marrow 300-510 mg/kg, oral (20 h) in mitotic index Gibson (1984)
Rat/Sprague- DNA strand breaks (alkaline 7.9, 26 or 79 mg/kg, i.p. (2.6 - Skare & Schrotel
Dawley elution in rat testis) or 24 h) 4, 13.2 or 39.5 mg/kg, (1984)
i.p. for 5 days
a + = positive; - = negative; i.p. = intraperitoneal injection
In Drosophila melanogaster, no statistically significant
sex-linked recessive lethals were obtained after exposure to phenol
via a variety of techniques (Sturtevant, 1952; Wild et al., 1980;
Gocke et al., 1981; Woodruff et al., 1985). However, when an
unusual technique was used, i.e. exposing isolated gonads in vitro
and implanting them in host larvae, several types of mutations were
induced (Hadorn & Niggli, 1946).
When rainbow trout ( Salmo gairdneri) were exposed to phenol
for 72 h at concentrations of 0.3 and 0.6 µl phenol/litre water, the
percentage of chromosomal aberrations in gill and kidney tissue was
significantly increased, at both concentrations, in a dose-related
way. Of these aberrations, 30% were structural, 45% consisted of
aneuploidy, and 25% were non-specified metaphases (Al-Sabti, 1985).
7.6.1.3 Mammalian in vitro systems
Data on in vitro genotoxicity in mammalian cells are given in
Table 8.
In a Chinese hamster V79 lung cell/HPRT mutation test, phenol
gave a positive result with metabolic activation. The highest dose
decreased survival by approximately 50% (Pashin & Bakhitova, 1982).
In a mouse lymphoma L5178Y cell/TK mutation test, there were
statistically significant and dose-related increases in mutation
frequency in the presence and absence of metabolic activation
(Wangenheim & Bolcsfoldi, 1988). However, in another laboratory this
test yielded non-conclusive results (McGregor et al., 1988).
As part of the US NTP testing program, phenol was evaluated for
induction of chromosomal aberrations and sister chromatid exchange
(SCE) in Chinese hamster ovary (CHO) cells (Ivett et al., 1989).
At a delayed harvest time (22.5 h), there were significantly
increased incidences of aberrations in cultures that included a
metabolic activation system from induced rat liver. Although a
dose-response effect was seen, the frequency of aberrations in the
absence of activation was low and the authors reported a negative
result. Regarding SCE induction, positive results were obtained,
both with and without activation. In additional studies, phenol
induced SCE in human lymphocytes in vitro, both in the presence
and in the absence of metabolic activation (Morimoto & Wolff, 1980;
Morimoto et al., 1983; Erexson et al., 1985). Negative results
(SCE) have also been reported (Jansson et al., 1986).
Phenol gave negative results in three studies in Chinese
hamster V79 cell metabolic cooperation assays (Chen et al., 1984;
Malcolm et al., 1985; Bohrman et al., 1988).
7.6.1.4 Mammalian in vivo system: somatic cells
In a bone marrow micronucleus test, groups of four Swiss CD-1
mice (sex not specified) were orally administered 265 mg phenol/kg
body weight and were sacrificed at 0, 18, 24, 42 and 48 h. Bone
marrow depression (decreased polychromatic erythrocytes/normocytes
(PCE/NCE) ratio) persisted at least up to 48 h after dosing. A
slight, but statistically significant, increase in the number of
micronuclei was seen at 24 h (3 micronuclei/1000 cells versus 1.5
micronuclei/1000 cells; 3000 cells scored per mouse) (Ciranni et
al., 1988b).
In a further study to asses the transplacental clastogenicity
of phenol, groups of 4 pregnant Swiss CD-1 mice received 265 mg
phenol/kg/body weight by oral gavage on day 13 of gestation. After
15, 18, 24, 30, 36 or 40 h, animals were sacrificed and adult bone
marrow cells and fetal liver cells were scored for micronuclei.
Slight, but statistically significant, increases in the frequency of
micronucleated PCE in adult bone marrow were observed at 15, 18 and
24 h (3.8, 4.0 and 5.0 micronuclei/1000 cells, respectively,
compared with 2/1000 for negative controls). A statistically
significant reduction in the PCE/NCE ratio was seen at 18 and 36 h.
Phenol had no effect on the frequency of micronuclei in fetal liver
(Ciranni et al., 1988a).
Bone marrow liver cells were also evaluated for the formation
of micronuclei, 30 h after oral administration of 0 or 250 mg
phenol/kg body weight to groups of five males Swiss CD-1 mice
(Gad-El Karim et al., 1986). Uptake was indicated by convulsive
seizures in all mice receiving phenol (1000 PCEs scored per mouse).
Phenol (265 mg/kg body weight), administered to Swiss CD-1 mice
by a single intraperitoneal injection, was reported to increase the
frequency of micronuclei in bone marrow PCEs 18 h post-treatment (7
micronuclei/1000 cells). The increased frequency decreased at 24 h
and was no longer statistically significant at 42 h. A decreased
PCE/NCE ratio persisted in tests up to 48 h post-treatment (Ciranni
et al., 1988b).
Barale et al. (1990) reported a negative result in a bone
marrow micronucleus test in Swiss CD-1 mice 18 h after treatment
with phenol. There was no effect on the PCE/NCE ratio.
Gocke et al. (1981) briefly reported a negative result in a
bone marrow micronucleus test, in which NMRI mice were sampled at 30
h, following i.p. injection of 47-188 mg phenol/kg. No information
on toxicity was given.
The results of these and other studies are summarized in Table
9.
7.6.1.5 Mammalian in vivo systems: germ cells
Skare & Schrotel (1984) obtained negative results in studies of
DNA strand breakage in rat testis. In one experiment, rats received
by intraperitoneal injection 0, 7.9, 26 or 79 mg phenol/kg body
weight and were sacrificed at 2.6 or 24 h post-treatment. Similar
results were obtained with further groups of rats that received 4,
13.2 and 39.5 mg phenol/kg body weight daily for 5 days before
sacrifice.
In an unconventional study involving dosing (0, 6.4, 64 and 640
mg phenol/kg body weight) of five successive generations of male and
female mice, large numbers of structural and numerical chromosomal
aberrations were reported in spermatocytes and spermatozoa, with
dose- and generation-related increases. The study was carried out
with 138 male mice from an inbred stock after skin testing
(Bulsiewicz, 1977).
7.7 Carcinogenicity
The evidence for the carcinogenicity of phenol in experimental
animals, based on the studies summarized below, was recently
considered by the IARC (1989) to be inadequate. Phenol was
classified by US EPA in Group D (data inadequate for evaluating the
carcinogenic potential) (Bruce et al., 1987).
7.7.1 Oral exposure
In an NCI (1980) study, groups of 50 male and 50 female
B6C3F1 mice were given drinking-water containing 0, 2500 or 5000
mg phenol/litre for 103 weeks. As matched controls, groups of 50
male and 50 female mice received tap water. There was a dose-related
decrease in water consumption and mean body weight gain in all
groups of mice. In mice receiving 5000 mg phenol/litre, an increase
in the number of uterine endometrial stromal polyps (5/48 = 10%) was
observed (in matched controls the incidence was 1/50 = 2%). There
was no evidence of an increased incidence of malignant tumours. The
other observed neoplasms were of the usual number and type found in
mice of this strain and age (NCI, 1980).
Groups of 50 male and 50 female Fischer-344 rats received 0,
2500 or 5000 mg phenol/litre drinking-water for 103 weeks, while the
matched control group received tap water. Male and female rats given
5000 mg/litre showed a decrease in mean body weight from week 20
onwards. There were statistically significant increased incidences
of phaeochromocytomas, leukaemias, lymphomas and C-cell thyroid
carcinomas in males of the low-dose group (NCI, 1980). NTP
considered this study negative for carcinogenicity due to the lack
of a dose-response for the neoplasms and the lack of response in
females.
7.7.2 Dermal exposure
Three studies examined the potential carcinogenicity of phenol
following dermal application (Rusch et al., 1955, Boutwell et
al., 1956; Bernard & Salt, 1982). However, none is considered
adequate for the evaluation of carcinogenicity due to the short
duration of exposure and/or use of inappropriate vehicles.
7.7.3 Inhalation exposure
No studies have been reported for this route of exposure.
7.7.4 Two-stage carcinogenicity studies
A dose of 3 mg phenol in acetone was applied to ICR/Ha Swiss
mice 3 times per week for 52 weeks after initiation with 150 µg
DMBA. Papilloma development was enhanced, compared with that of mice
exposed to DMBA alone (Van Duuren et al., 1968; Van Duuren &
Goldschmidt, 1976). These observations were in agreement with those
from earlier reports on the promotional activity of phenol (Boutwell
et al., 1955, 1956; Salamon & Glendenning, 1957; Boutwell & Bosch,
1959; Wynder & Hoffmann, 1961).
A dose of 3 mg phenol in acetone applied 3 times weekly for 460
days to female ICR/Ha Swiss mice after initiation with 5 µg
benzo[a]pyrene had a slight promoting activity. Simultaneous
application of both agents showed a partial reduction in carcinomas
compared with mice treated with benzo[ a]pyrene alone (Van Duuren
et al., 1971, 1973; Van Duuren & Goldschmidt, 1976).
7.8 Special studies
7.8.1 Neurotoxicity
Tremors, convulsions, coma and death were reported after
intraperitoneal and subcutaneous doses of phenol (Deichmann &
Witherup, 1944; Ernst et al., 1961; Windus-Podehl et al., 1983).
The tremors were enhanced by prior monoamine depletion following
reserpine treatment (Suzuki & Kisara, 1985).
Upon histological examination of rats in which convulsions had
been induced by subcutaneous phenol injection (200 mg/kg body weight
given once a week for two weeks), two out of six animals showed
spinal cord and spinal root degeneration (Veronesi et al., 1986).
Phenol, given either intravenously or intra-arterially (250
µg), facilitated neuromuscular transmission and antagonized
neuromuscular blockade by D-tubocurarine in cats. The effect was
determined to be pre-synaptic in origin (Blaber & Gallagher, 1971).
Phenol caused diminution of the compound action potential in
preparations of the saphenous nerve after acute and chronic
perfusion (Schaumburg et al., 1970).
Groups of five male CD-1 mice were supplied with drinking-water
containing 0, 4.7, 19.5 or 95.2 mg phenol/litre for 4 weeks, at the
end of which the concentrations of various neurotransmitters and
their metabolites were measured in different parts of the brain. The
largest effects were seen in levels of noradrenaline in the
hypothalamus (significant decreases of 29 and 40% in the mid- and
high-dose groups) and of dopamine in the corpus striatum
(significant decreases of 21, 26 and 35% in the low-, mid- and
high-dose groups). There were dose-related, but not always
statistically significant, decreases in all of the neurochemicals
measured in the hypothalamus: noradrenaline, dopamine,
vanillylmandelic acid (VMA), 3,4-dihydroxy-phenylacetic acid
(dopac), homovanillic acid (HVA), serotonin (5-HT) and
5-hydroxyindoleacetic acid (5-HIAA). There were significant
decreases in VMA in the midbrain, corpus striatum and cortex, 5-HT
in the midbrain, corpus striatum and medulla oblongata, and dopac in
the cerebellum in the high-dose group only. There were also
significant decreases in 5-HT and 5-HIAA in the hypothalamus of the
mid- and high-dose groups (Hsieh et al., 1992).
Continuous exposure of rats to 0.012, 0.12 and 5.3 mg
phenol/m3 for 61 days caused a shorter extensor muscle chronaxy
and an increase in whole-blood cholinesterase activities in rats at
concentrations above 0.012 mg phenol/m3 (Mukhitov, 1964).
7.8.2 Myelotoxicity
Because phenol is an important metabolite of benzene, which is
known to exert a toxic effect on bone marrow (including leukaemia)
after metabolic activation, many studies have been performed to
investigate the possible myelotoxic action of phenol.
In an in vitro assay for toxicity to primary murine
haematopoietic cell cultures, phenol showed slight and variable
activity at a concentration of 0.4 mM, but there was marked toxicity
at 2 mM. In comparison, the phenol metabolites catechol and
hydroquinone exhibited marked toxicity at 0.04 mM (Seidel et al.,
1991).
Subcutaneous treatment with phenol (245 mg/kg body weight)
significantly inhibited erythropoiesis in mice 48 h after treatment,
as indicated by a 59Fe uptake assay (Bolcsak & Nerland, 1983).
Intraperitoneal injection of 0-150 mg phenol/kg body weight to
male B6C3F1 mice, twice daily for 12 days, did not result in a
suppression of bone marrow cellularity. However, simultaneous
treatment of mice with 75 mg phenol/kg body weight and 25-75 mg
hydroquinone/kg body weight (another benzene metabolite), produced a
dose-related decrease in bone marrow cellularity, which was much
more pronounced than after treatment with hydroquinone only. The
observed effect closely resembled the myelotoxic effect of benzene
(Eastmond et al., 1987b). Subsequent in vitro studies by these
and other investigators confirmed that phenol (0.01-1 mM) stimulates
further bioactivation of hydroquinone to myelotoxic compounds in
bone-marrow cells (Eastmond et al., 1987b; Subrahmanyam et al.,
1989).
No haematopoietic toxicity was found in rats after daily
subcutaneous injections of 250-750 mg phenol/kg body weight for 1
week. Of the rats receiving 750 mg phenol/kg body weight, 50% died
(Mitchell, 1972).
Six consecutive subcutaneous injections of 50 mg phenol/kg body
weight to mice resulted in a slightly but significantly reduced
number of granulopoietic stem cells and bone marrow cellularity in
the tibia (Tunek et al., 1981).
7.8.3 Immunotoxicology
One immunological study has been reported. Female CD1 mice were
exposed to 19 mg phenol/m3 (5 ppm), either as a single 3-h
exposure or as five daily 3-h exposures. Neither the susceptibility
of the animals to experimentally induced streptococcus aerosol
infection nor their pulmonary bactericidal activity was
significantly affected (Aranyi et al., 1986).
Groups of five male CD-1 mice were supplied with drinking-water
containing 0, 4.7, 19.5 or 95.2 mg phenol/litre for 4 weeks, at the
end of which various haematological and immunological parameters
were measured. The erythrocyte count was statistically significantly
decreased, compared with control values, in all treated groups in a
dose-related manner, but total and differential leucocyte counts
were unaffected. Total spleen cellularity was decreased in a
non-significant dose-related manner. The highest dose suppressed the
stimulation of cultured splenic lymphocytes by the B-cell mitogen
lipopolysaccharide, the T-cell mitogen phytohaemagglutinin, and the
T- and B-cell mitogen pokeweed, but not by concanavatin. The mid and
high doses suppressed the animals' antibody production in response
to a T-cell-dependent antigen, i.e. sheep erythrocytes (Hsieh et
al., 1992).
7.8.4 Biochemical effects
In a study on the biochemistry of intestinal mucosa, mice were
provided with 0,5, 50 or 500 mg phenol/litre drinking-water
(calculated by the authors to be 1, 10 and 100 mg phenol/kg body
weight) for 5 days or 5 months at 1-day intervals. An increase in
glucose-6-phosphatase, succinate dehydrogenase and cytochrome
oxidase activities in the intestinal mucosa was observed in mice
receiving > 0.02 mg phenol/kg body weight for 5 days. A decrease
in these activities was seen at 2 mg phenol/kg body weight. After 5
months administration of 0.02 and 0.2 mg phenol/kg body weight, the
enzyme activity had returned to normal, but the highest dose group
showed a decline (or even a lack) of activity in the cells of the
intestinal mucosa (Olowska et al., 1980).
In another study of biochemical effects, mice were provided for
5 days (killed 24 h after the last application) and 35 days (killed
30 days after the last application) with aqueous solutions of 0.08,
0.8 or 8 mg phenol/litre. The authors of the study calculated the
doses to be approximately 0.016, 0.16 and 1.6 mg phenol/kg body
weight per day, respectively. Only the lowest dose of 0.08 mg
phenol/litre evoked considerable changes in the localization of
glucose-6-phosphatase, 5 days after treatment. Changes in alkaline
phosphatase localization in the kidney were seen at 0.8 and 8 mg
phenol/litre. Full recovery occurred after 30 days (Laszczynska et
al., 1983).
Inhalation exposure of 50 male white rats to 0.4 mg
phenol/m3, 24 h/day, 7 days/week, for 3 months, resulted in some
inhibition of oxidative phosphorylation in the lungs, liver and
kidneys. An increase in the rate of glycolysis was also observed in
the lungs and kidneys (Skvortsova & Vysochina, 1976).
8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Controlled studies
In the Kligman maximization test, phenol did not cause
sensitization in 24 human volunteers (Kligman, 1966).
It was reported by Rea et al. (1987) that, in a group of 134
"chemically sensitive" patients where several volatile organic
chemicals were detected in the blood, 107 (80%) reacted adversely
after a challenge exposure to phenol alone (0.008 mg/m3). The
criteria used to identify "sensitive patients" and "adverse
reactions" were not specified. The toxicological significance of
this finding is not known.
Mukhitov (1964) reported that six 5-min inhalation exposures to
phenol at 0.015 mg/m3 produced an increased sensitivity to light
in each of 3 dark-adapted subjects.
8.1.2 Case reports
Various reports have appeared on the adverse effects of phenol
in individuals or groups of humans after intentional (e.g.,
therapeutic) as well as accidental short-term exposure to phenol.
8.1.2.1 Dermal exposure
The use of phenol as a disinfectant and antiseptic was
introduced by Lister (1867). However, its use has been restricted by
intoxications caused by these applications (Table 10).
Local effects after dermal phenol exposure consisted of
erythema or painless blanching (Dreisbach, 1983), and, in more
severe cases, corrosion (Schmidt & Maibach, 1981) and necrosis. The
use of 5-10% phenol dressings for antiseptic purposes, for example,
has led to many cases of necrosis of the skin and underlying
tissues. When fingers and toes have been involved, amputation has
sometimes been necessary. Due to their high toxicity, these
dressings are no longer used (Cronin & Brauer, 1949; Deichmann,
1949; De Groote & Lambotte, 1960; Abraham, 1972).
Phenol chemical peel is a technique which has been used in
superficial surgery of the skin for the last 30 years (Ersek, 1991).
The phenolic mixture used classically is 3 ml of 50% phenol, 2 ml of
water, 8 drops of soap and 8 drops of croton oil. This is applied to
the skin to reduce pigmentation. Topical use of phenol as a chemical
face-peel has been reported as being associated with cardiac
dysrhythmias in "up to 30% of adults" (Morrison et al., 1991), but
only a single case report has been published (Warner & Harper,
1985). This report concerned a 10-year-old boy who had a solution,
consisting of 40% phenol and 0.8% croton oil in hexachlorophene soap
and water, applied to a large nevus covering 1.9% of his body
surface whilst under anaesthesia (60% nitrous oxide and 3%
halothane) and receiving a total of 200 ml of lactated Ringer's
solution intravenously. After 55 min of treatment, multifocal and
coupled premature ventricular complexes were detected by ECG, but
blood pressure remained stable and plasma sodium and potassium
concentrations were normal. An intravenous infusion of 250 mg
bretylium sulfate suppressed the dysrhythmia and the boy had an
uneventful recovery.
Systemic intoxication can occur very rapidly after absorption
of phenol through the skin (Table 10). Most significantly,
cardiovascular shock (sometimes resulting in death) and severe
metabolic acidosis occur. Truppman & Ellenby (1979) observed cardiac
arrhythmias (supraventricular as well as ventricular) in 10 out of
42 patients within 10 min after the application of approximately 5%
phenol on half of the face for cosmetic treatment. Hyperventilation,
kidney damage and methaemoglobinaemia have also been observed in
several cases of exposure to phenol.
Table 10. Human dermal toxicity of phenol
Concentration Medium Contact duration Circumstances Most severe response References
(%)
100 crystals 30 min in glove grangrene Abraham (1972)
80-100 water 20 min spill on hip, thigh, death Turtle & Dolan
scrotum (1922)
80-100 water 2-4 days closed dressings on 11 persons exposed: Lister (1867)
open wounds 1 death, 8 gas gangrene,
11 tissue necrosis
78 water 2-5 min 4-5 litres spilt on coma Duverneuil &
upper half of body Ravier (1962)
43.5 waste water 1 min spill on lower half of body shock Evans (1952)
5 ointment 7 days closed dressing on cut gangrene Schussler &
Stern (1911)
2 water 2.5 days moist dressing over burns death Cronin & Brauer
on 30% of body surface (1949)
2 water 11 h closed bandage on death Hinkel & Kintzel
infant umbilicus (1968)
Foxall et al. (1991) reported a case of acute renal failure
following an industrial accident in which a man was partially
submerged for a few seconds in a solution of 20% phenol in
dichloromethane. He immediately showered, but was subsequently found
in a state of collapse. His extremities were cold and he had 50%
body burns. He developed nausea and vomiting after taking fluids.
Anuria ensued, with a rise in plasma creatinine, but treatment with
intravenous furosemide and haemodyalisis (daily for seven days, then
with decreasing intervals for a further 18 days), allowed adequate
urinary volumes to be produced. Respiratory distress required
intensive care treatment. Marginal polyuria persisted one year after
the accident.
8.1.2.2 Oral exposure
Cases of oral intoxication have occurred as a result of
accidental and intentional ingestion. Local and systemic effects
have been described in the literature, symptoms being similar to
those following dermal exposure. Case reports have been published
(Model, 1889; Stajduhar-Caric, 1968; Haddad et al., 1979).
Death occurred within 10 min of ingestion of 4.8 g phenol
(Andersen, 1869). However, ingestion of 56.7 g of a phenol-saline
mixture was reported to have occurred without complaints (Leider &
Moser, 1961), and an individual survived the ingestion of 57 g
phenol (88%) after intensive treatment. Symptoms in the latter study
included severe gastrointestinal irritation, as well as the expected
cardiovascular and respiratory effects (Bennett et al., 1950).
A severe accidental phenol spill in Wisconsin in 1974
contaminated ground water which was being used as drinking-water.
Approximately one month later, several people living near the spill
complained of health effects. Six months after the spill, medical
histories were taken from 100 people who had consumed
phenol-contaminated water (the authors estimated the daily exposure
to be 10-240 mg phenol/person). In retrospect, a statistically
significant increase was found in diarrhoea, mouth sores, dark urine
and burning of the mouth, which had persisted for an average of 2
weeks. No significant abnormalities were found 6 months after
initial exposure upon physical examination or laboratory analysis.
Urinary phenol levels were not elevated (Delfino & Dube, 1976; Baker
et al., 1978).
A river in North Wales, United Kingdom, used for the
preparation of drinking-water, was accidentally contaminated with
phenol (Jarvis et al., 1985). When the water was chlorinated,
various chlorophenols appeared to have been formed. A retrospective
postal survey of 344 households that received the contaminated tap
water and 250 control households was carried out. Significantly more
gastrointestinal illnesses, as well as other symptoms, were claimed
in the contaminated areas than in the unexposed areas. Phenol
concentrations in drinking-water were conservatively estimated to
have been 4.7-10.3 µg/litre for some days (Jarvis et al., 1985).
8.1.2.3 Inhalation exposure
Very few cases of adverse effects after short-term phenol
vapour exposure have been reported.
Hospital outbreaks of severe idiopathic neonatal unconjugated
hyperbilirubinaemia have been associated with the phenol-containing
disinfectant used for cleaning the nursery equipment, floors and
walls. When the disinfectant was no longer used, the epidemic
subsided (Daum et al., 1976; Wysowski et al., 1978; Doan et
al., 1979).
Studies of occupational inhalation exposure are described in
section 8.2.
8.1.2.4 Exposure by injection
Phenol has been used as a neuron blocking agent in patients
suffering from spasm following, for example, spinal cord damage or
cerebrovascular stroke (Wood, 1978, review; Nathan, 1959; Cooper et
al., 1965; Khalili & Betts, 1967; Gibson, 1987). It has also been
used to relieve chronic pain (Wood, 1978; Benzon, 1979; Smith,
1984). Treatment involved administering the phenol by intravenous
injection or perfusion, or by direct injection into the spinal cord.
Reported side-effects of phenol therapy were convulsions, transient
paraesthesia, leg weakness, urinary and fecal incontinence, one case
of a severe arterial block in the upper arm requiring amputation,
and one case of acute bronchospasm (Wood, 1978, review; Benzon,
1979; Gibson, 1987; Atkinson & Skupak, 1989). In addition, there
have been reports of phenol-induced cardiac dysrhythmia in adults
(Forrest & Ramage, 1987) and in children, in whom an incidence of
19% was reported (Morrison et al., 1991).
8.2 Occupational exposure
Poisoning due to chronic inhalation of phenol was known 100
years ago, primarily as a disorder in physicians and their helpers,
under the term "carbol marasmus" (Lister, 1867). A classical case of
phenol marasmus was described in a worker employed for 13´ years in
a laboratory boiling phenol solutions. Symptoms were anorexia,
weight loss, headache, vertigo, salivation and dark urine (Merliss,
1972).
A few studies are available concerning occupational exposure of
workers in bakelite factories. Workers were exposed to phenol, and
simultaneous exposure to formaldehyde occurred. Elevated phenol
urine levels, unspecified complaints, and chronic airway obstruction
were observed (Schoenberg & Mitchell, 1975; Knapik et al., 1980).
Twenty-nine cases of poisonings among workers, who, during a
3-year period, quenched coke with a waste-water solution containing
0.3-0.8 g phenol/litre, were attributed to phenol intoxication.
Phenol vapour concentrations in the air ranged from 0.5 to 12.2
mg/m3. The number of workers and the symptoms of intoxication were
not specified. The author did not consider the potential of dermal
absorption (Petrov, 1960).
A case-control study was carried out on 57 cases among 3805
workers from the Finnish wood industry (particle board, plywood,
sawmill or formaldehyde glue) suffering from respiratory cancer. The
inhalatory exposure level to phenol and frequency of multiple
exposure to pesticides were found to be significantly higher for
cancer cases, but the exposure to wood dust was not significantly
different between cases and controls (Kauppinen et al., 1986). The
number of cases in this study was small, and confounding exposures
were inadequately controlled.
In a case-control study of 6678 rubber workers, employed in
areas where phenol was used, exposure to phenol was not associated
with increased risks of cancer of the respiratory tract, stomach or
prostate or of lymphosarcoma or lymphatic leukaemia (Wilcosky et
al., 1984).
A mortality study was conducted among 14 861 white male workers
engaged in the production or use of phenol and formaldehyde in five
companies within the USA. The follow-up comprised more than 360 000
man-years. Mortality rates from all causes combined were similar to
those in the general population of the USA. Excesses of cancer of
the oesophagus, cancer of the kidney and Hodgkin's disease were
observed among the workers exposed to phenol, but these did not show
any exposure-response relationship and were not statistically
significant. Reduced mortality ratios were observed for cancer of
the buccal cavity and pharynx, cancer of the stomach, cancer of the
brain, arteriosclerotic heart disease, emphysema, disease of the
digestive system and cirrhosis of the liver, although these
reductions were not statistically significant. For arteriosclerotic
heart disease, emphysema and cirrhosis of the liver, there were
inverse relationships between mortality rates and duration of phenol
exposure and cumulative phenol exposure levels (Dosemeci et al.,
1991).
A cardiovascular disease (CVD) mortality study was conducted
among 1282 white male production workers in a large rubber- and
tyre-manufacturing plant. Exposure estimates for 25 solvents were
available (concentrations were not measured). The CVD mortality
during 15-year follow-up period was analyzed in exposed and not
exposed workers. The known association between CS2 exposures and
ischemic heart diseases (IHD) was confirmed, and two other solvents,
ethanol and phenol, were also found to be predictors of IHD. Phenol
showed the strongest association with CVD mortality. However, some
confounders (cigarette smoking, hypertension and high serum
cholesterol) were not controlled and unrecognized chemical
atherogens could also, according to the authors, influence the
results (Wilcosky & Tyroler, 1983).
8.3 Organoleptic data
The odour threshold for phenol has been reported to range from
0.021 to 20 mg/m3 (Van Gemert & Nettenbreijer, 1977; Van Gemert,
1984).
The geometric mean of 16 air odour thresholds and 6 water odour
thresholds for phenol was reported by Amoore & Hautala (1983) to be
0.16 mg/m3 (0.040 ppm, with a standard error of 0.026 ppm). In
this calculation, the original literature was reviewed and values
which diverged more than 100-fold from the nearest of two or more
other thresholds were eliminated. Both detection and recognition
values were included. The water detection threshold for phenol,
based upon multiplying the calculated air odour threshold by the
water-air distribution ratio, was reported by the same authors to be
7.9 mg/litre.
A taste threshold value of 0.3 mg/litre in water has been
reported (US EPA, 1992).
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
The toxicity of phenol has been studied in microorganisms
(e.g., bacteria, fungi, algae and protozoa) and numerous aquatic
invertebrates and vertebrates (Buikema et al., 1979). Because of
this vast amount of data, a selection has been made, based on the
reliability of the data and the relevance of the test organisms.
Details of acute and long-term aquatic toxicity studies, considered
to be adequately performed and reported, are included in Tables 11
and 12. Less adequate studies are reported in the text only.
9.1 Microorganisms
Reliable phenol toxicity data for microorganisms are given in
Table 11.
In microorganisms, growth inhibition is usually observed after
phenol exposure. In studies on single bacterial species, the EC50
values (EC50 = calculated concentration affecting 50% of test
population) found for growth inhibition varied from 244 mg
phenol/litre in a newly developed, 6-h test with Pseudomonas putida
(Slabbert, 1986) to 1600 mg phenol/litre after 18 h of exposure in a
more conventional test with Aeromonas hydrophila (Dutka & Kwann,
1981). Bringmann & Kühn (1977) reported a toxicity threshold of 64
mg/litre after 16 h. EC50 values for reduced photoluminescence in
Photobacterium phosphoreum of 28-34 mg phenol/litre (Dutka et
al., 1983) and 40 mg phenol/litre (Curtis et al., 1982) have
been reported. In activated sludge, the EC50 for a reduced oxygen
uptake was reported to be 520-1500 mg phenol/litre, whereas a lower
value was found for substrate consumption inhibition (104 mg
phenol/litre) (Miksch & Schürmann, 1988; Volskay & Grady, 1988). The
lowest reported concentration affecting activated sludge was 10 mg
phenol/litre; 1 mg phenol/litre had no effect (Baird et al.,
1974).
Reported toxicity thresholds for protozoa were of the same
order of magnitude as for bacteria: 33-144 mg phenol/litre
(Bringmann & Kühn, 1959, 1980; Dive & LeClerq, 1977; Bringmann et
al., 1980). For algae, values were somewhat lower, but were
observed after a longer exposure period: 6 mg phenol/litre for
cyanobacteria (blue-green algae) and 8 mg phenol/litre for green
algae, after 7-8 days of exposure (Bringmann & Kühn, 1978, 1980).
The IC50 values (concentration causing 50% growth inhibition)
reported for various fungi by Kwasniewska & Kaiser (1983) were of
the same order of magnitude as the above EC50 values for bacterial
growth inhibition: 460-1000 mg phenol/litre. These values are also
within the range of concentrations observed by Babich & Stotzky
(1985) to cause initial or complete growth inhibition in various
fungi (100-1000 mg phenol/litre and 750->1000 mg phenol/litre,
respectively).
Table 11. Acute aquatic toxicity of phenol
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Freshwater Organisms
Bacteria
Photobacterium 15 6.5-6.7 S 5, 10, EC50c 28, 32, 34 Dutka et al.
phosphoreum 15 min (1983)
Pseudomonas putida 25 7.0 80.1 S 16 h TTc 64 Bringmann &
Kuhn (1977)
27 7.2 S 6 h EC50 244 Slabbert
(1986)
Cyanobacteria
Microcystis aeruginosa 27 7.0 72.3 S 8 days TT 6 Bringmann &
Kuhn (1978)
Green algae
Scenedesmus 27 7.0 72.3 S 7 days TT 8 Bringmann &
quadricauda Kuhn (1980)
Protozoa
Chilomonas 20 6.9 S 48 h TT 65 Bringmann et
paramaecium al. (1980)
Colpidium campylum 20 S 43 h TTd 100 Dive & LeClerq
(1977)
Entosiphon sulcatum 25 6.9 80.1 S 72 h TT 33 Bringmann &
Kuhn (1980)
Microregma heterostoma 27 7.5-7.8 213.6 S 28 h TT 30e Bringmann &
Kuhn (1959)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Uronema parduczi 25 7.3 S 20 h TT 144 Bringmann &
Kuhn (1980)
Crustacea
Asellus aquaticus 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 230, 200, Green et al.
96 h 180f (1985)
Cypris subglobosa 20.4 7.9 8.4 204 S 12, 24, 48, LC50 173, 167, Rao et al.
72, 96 h 137, 122, 72 (1983)
Daphnia magna 19.8-20.9 7.7-8.3 157 ± 4 S 48 h LC50 13 Gersich et
al. (1986)
19 ± 1 8.2 ± 0.3 199.4 S 48 h LC50 100f Hermens (1984)
19 ± 1 8.2 ± 0.3 199.4 S 48 h EC50g 9f Hermens (1984)
17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 24 h LC50h 13 Holcombe et
al. (1987)
S 48 h EC50g 7 Keen & Baillod
(1985)
22 ± 1 6.8-7.8 7.6 ± 0.2 146 ± 15 S 48 h LC50 8 Lewis (1983)
Gammarus pulex 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 106, 85, Green et al.
96 h 69f (1985)
7 ± 1 8.3 10.9 250 R 24, 48, LC50 100, 89, Stephenson
72, 96 h 67, 51 (1983)
Ceriodaphnia dubia 25 ± 1 8.18 ± 0.04 57.1 ± 4.1 S 48 h LC50 3.1 Oris et al.
(1991)
Mollusca
Indoplanorbis S 12, 24, 48, LC50 265, 215, 200, Agrawal (1987)
exustus 72, 96 h 156, 126
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Lymnaea acuminata 20 ± 2 7.9 ± 0.2 5.5 ± 1.5 190-223 R 12, 24, 48, LC50 270, 219, 205, Gupta & Rao
72, 96 h 158, 129 (1982)
Worms
Limnodrilus 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 960, 870, Green et al.
hoffmeisteri 96 h 780f (1985)
Polycelis felina 18 7-8.5 -i** 300-500 S 96 h LC50 64f Erben et al.
(1983)
Polycelis tenuis 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 230, 200, Green et al.
96 h 88f (1985)
Fish
Brachydanio rerio 25 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 31, 29f Fogels &
Sprague (1977)
24 ± 1 > 6 64 R 6, 12, 24, LC50 35, 31, 28, Razani et al.
(12 h) 48, 72, 96 h 26, 25, 25f (1986a)
Campostoma anomalum 23 -i R 48 h LC50 18f Chagnon &
(24 h) Hlohowskyj
(1989)
Catostomus 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 11 Holcombe et
commersoni al. (1987)
Jordanella floridae 25 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 36, 36f Fogels &
Sprague (1977)
Lebistes reticulatus 28-31.8 7.8-8.2 5.7-7.2 218-239 R 12, 24, 48, LC50 103, 83, 64, Gupta et al.
(24 h) 72, 96 h 50, 48 (1982a)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Lepomis macrochirus 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 17 Holcombe et
al. (1987)
Leuciscus idus 20 S 48 h LC50 14, 25 Jünke &
melanotus Lüdemann
(1978)
Notopterus notopterus 23-26.5 6.8-7.6 5.9-7.8 60-70 S 24, 48, 72, LC50 14, 14, 13, Verma et al.
96 h 13 (1980)
Pimephales promelas 17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50h 25 Holcombe et
al. (1987)
25 ± 2 6.2-8.2 43-49 CF 96, 192 h LC50 29, 23f Phipps et al.
(1981)
Rasbora 20 7.2 250 S 24, 48 h LC50 8, 7 Alabaster
heteromorpha (1969)
Rutilus rutilus 10.3 ± 0.3 7.8 ± 0.02 257-260 S 48 h LC50 10f Solbé et al.
(1985)
Salmo gairdneri 15 ± 0.5 8.0-8.3 -i 350-375 CF 48, 96 h LC50 12 12f Fogels &
Sprague (1977)
17.2 ± 0.5 7.4 ± 0.2 8.7 ± 1.1 44.7 S 96 h LC50i 11 Holcombe et
al. (1987)
Insects
Baetis rhodani 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 19, 19, 16f Green et al.
96 h (1985)
Chironomus riparius 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 1050, 500, Green et al.
96 h 240f (1985)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Hydropsyche 11 ± 1 7.5-8.1 99.5 ± 7.7 S 24, 48, LC50 940, 720, Green et al.
angustipennis 96 h 260f (1985)
Marine Organisms
Crustacea
Artemia salina S 24, 48 h LC50j 157, 56 Price et al.
(1974)
Canthocamptus synthetic medium according to Cairns S 48 h LC50j 9 Rao & Nath
(1983)
Gammarus duebeni 5 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 183f Oksama &
Kristofferson
16 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 89f (1979)
Mesidotea entomon 5 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 176f Oksama &
Kristofferson
(1979)
10 ± 0.6 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 186f
Panopeus herbstii 25 25 pptk R 96 h LC50 53 Key & Scott
24 h (1986)
Mollusca
Crassostrea 24 ± 1 seak R 48 h (eggs) LC50 58 Davis & Hidu
virginica (1969)
Mercenaria 24 ± 1 seak R 48 h (eggs)- LC50j 53-55 Davis & Hidu
mercenaria 12 days (1969)
(larvae)
Table 11 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test Parameterb Concentration Reference
(°C) oxygen (mg CaCO3/ duration (mg/litre)
(mg/litre) litre)
Worms
Ophryotrocha 21 seak S 48 h LC50 100-330 Parker (1984)
diadema
Fish
Phoxinus phoxinus 5 ± 0.7 7.7 ± 0.1 8.4 ± 0.3 0.6%k CF 96 h LC50 10 Oksama &
Kristofferson
(1979)
a S = static test; CF = continuous flow test; R = renewal test (semi-static test)
b EC50 = median effect concentration = calculated concentration causing effect in 50% of population; LC50 = median lethal concentration
TT = toxicity threshold, i.e. concentration affecting growth in > 3% of population
c effect: reduction in photoluminescence
d minimal concentration affecting growth
e river water
f concentration of test compound analysed during assay
g immobility
h simultaneous testing of 8 species
i no data; aerated
j reported was TLm, or median toxicity limit
k salinity; sea = sea water
Table 12. Long-term aquatic toxicity of phenol
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
Freshwater Organisms
Crustacea
Daphnia 19 ± 1 8.2 199 R 16 days NOEC (growth) 0.16 De Neer et
magna (48 h) al. (1988)
19 ± 1 8.2 ± 0.3 199 R 21 days NOLC 10c Hermens
NOEC (repro) 3.2c (1984)
NOEC (growth) 3.2c
Ceriodaphnia 25 ± 1 8.18 ± 0.04 57.1 ± 4.1 R 4 days NOEC (repro) 4c Oris et al.
dubia (48 h) 7 days NOEC (survival)d 4c (1991)
Fish
Brachydanyo 24 ± 1 6.1-6.5 6.4-8.5 57-61 R 3 months (adults) NOLC 4.9 Razani et
rerio (24 h) al. (1986b)
NOEC (spawning) < 2.2
R 2 months (test started NOEC (growth) 2.2
(48 h) with eggs from adults NOLC < 2.2
exposed to same
concentrations; all
eggs hatched after
2-3 days)
Table 12 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
Carassius 19-24 7.78 6.2-9.0 197.5 CF 8 days after hatching LC1 (at hatch) 0.002c Birge et
auratus (test was started with al. (1979)
eggs (1-2 h after
spawning) which
hatched completely
within 3.5 days)
Pimephales 25 ± 1 8.0 5.3 725.3 CF 30 days after hatching NOEC (hatching) 83.2c De Graeve
promelas et al.
NOEC (growth) 0.75c (1980)
Pimephales 25 ± 1 8.0 5.3 725.3 CF NOLC 6.1c,e
promelas 25 ± 2 7.2-7.9 7.7 46.0 CF 38 days after hatching NOLC 3.57c,f Holcombe
(test was started with NOEC (growth) 1.83c et al.
eggs within 24 h after NOEC (hatching) 3.57c,f (1982)
spawning)
Salmo 12-14 7.78 6.2-9.0 197.5 CF 8 days after hatching LC1 (at hatch) 0.0002c Birge et
gairdneri (test was started with al. (1979)
eggs (20 min after
fertilization) which
hatched completely
within 22 days)
Table 12 (contd).
Organism Temperature pH Dissolved Hardness Methoda Test duration Parameterb Concentration Reference
(°C) oxygen (mg/litre (mg/litre)
(mg/litre) CaCO3)
13.3-14.2 7.4-8.1 8.6-10.2 100 CF 4 days after hatching NOLC 0.009c,e Black et
(test was started with al. (1983)
eggs (20 min after NOEC (hatching) 0.009c,f
fertilization) which
hatched within
23 days)
7.8 5.7 579.9 CF 58 days after hatching NOEC (growth) 0.1c,g De Graeve
(test was started with et al.
eyed eggs which hatched (1980)
completely within 48 h)
a S = static test; CF = continuous flow test; R = renewal test (semi-static)
b NOEC = no-observed-effect concentration = highest tested concentration without observed effect; NOLC = no-observed-lethal concentration.
An LC1 is a calculated value which is, to some extent, comparable to the observed NOLC value used in other studies.
c phenol concentration analysed during test
d survival was a more sensitive end-point than reproduction
e calculated from results by Task Group
f highest concentration tested
g extrapolated by authors
An increase in salinity (0-30%) increased the toxicity of
phenol to fungi (Babich & Stotzky, 1985).
9.2 Aquatic organisms
9.2.1 Freshwater organisms
9.2.1.1 Short-term studies
The most important sublethal acute effects observed in
freshwater species after phenol exposure were a reduced heart rate
and damage to the epithelium of gills (with loss of function),
liver, kidneys, intestines and blood vessels. One study reported the
occurrence of severe seizures, mediated by the central nervous
system, in Salmo gairdneri after exposure to sublethal phenol
concentrations (Bradbury et al., 1989). In invertebrates, growth
inhibition was usually observed. Some EC50 values for the latter
organisms are given in Table 11.
Most toxicity studies concentrated on lethal effects. Death was
usually preceded by immobility, loss of equilibrium, paralysis and
respiratory distress (Razani et al., 1986a; Tonapi & Varghese,
1987; Green et al., 1988; Chagnon & Hlohowskyj, 1989). Toxicity
testing, where the same species was used by different research
workers in different waters, resulted in LC50 values (LC50 :
calculated concentration causing death in 50% of test group) that
varied widely, as can be seen from Table 11.
Environmental factors may affect the toxicity of phenol (Brown
et al., 1967; Miller & Ogilvie, 1975; Ruessink & Smith, 1975;
Cairns et al., 1976; Reynolds et al., 1978; Birge et al.,
1979; Dalela et al., 1980; Gluth & Hanke, 1983; Gupta et al.,
1983a,b; Stephenson, 1983). Hardness and pH, however, do not appear
to have a large impact on phenol toxicity. The toxicity for various
fungi and fish species, for example, did not change significantly
over the pH range of 5-8; toxicity for fungi and some fish species
was not influenced at all by hardness, whereas phenol was slightly
more toxic in soft than in hard waters for the carp (Herbert, 1962;
Pickering & Henderson, 1966; EIFAC, 1972; Babich & Stotzky, 1985).
The effect of temperature appeared to be variable (Cairns et al.,
1978; Babich & Stotzky, 1985). Since temperature influences both the
uptake and the detoxification (conjugation) of phenol (Green et
al., 1988), phenol toxicity could be enhanced, as well as
diminished, by increasing temperature, depending on which parameter
was influenced most.
Several biological factors also influence the response of the
biota to phenol, e.g., strain type, nutritional status, size,
embryonal or developmental stage, crowding and physiological
adaptation (Dowden & Benett, 1965; Alexander & Clarke, 1978; Birge
et al., 1979; Flerov, 1979; De Graeve et al., 1980; Kordylewska,
1980; Gupta et al., 1982b; Mayes et al., 1982; Black et al.,
1983; Lewis, 1983).
Comparison of 48-h LC50 values from Table 11 shows that, in
general, fish are the most sensitive freshwater species with respect
to phenol toxicity. The 48-h LC50 values for some selected fish
species ranged from 7 to 64 mg/litre. For crustaceans, this range
was 3.1-200, and for molluscs it was 200-205 mg/litre; for insects,
it was 19-720 mg/litre and for worms 200-870 mg/litre. Upon
simultaneous testing of eight species at concentrations up to 51 mg
phenol/litre, no toxicity was observed for larvae of the amphibian
Xenopus laevis, for the snail Aplexa hypnorum or the insect
Tanytarsus dissimilis. Where toxicity was observed, LC50 values
were included in Table 11 (Holcombe et al., 1987). The data
presented in Table 11 are in good agreement with the order of
increasing tolerance to phenol proposed by Alekseyev & Antipin
(1976): fish-crustaceans-tolerant insects-worms-molluscs-highly
tolerant insects.
9.2.1.2 Long-term studies
Most long-term studies with freshwater species have concerned
growth, reproduction and/or mortality; these studies are discussed
below. Studies considered to be adequately performed and reported
are included in Table 12.
A few long-term studies with freshwater fish have been designed
to detect sublethal effects of phenol exposure. Increased
proteolysis as a result of stress, mild kidney damage, and an
inhibitory effect on the development and maturation of the ovary,
secondary to a liver dysfunction, were some of the effects reported
(Dangé, 1986; Gupta & Dalela, 1987; Kumar & Mukerjee, 1988).
In a life-cycle test using Daphnia magna, the maximum
acceptable tolerance concentration (MATC) proved to be 1.5-6.3 mg
phenol/litre (US EPA, 1980). These results are comparable with the
no-observed-effect concentration (NOEC) values for growth and
reproduction (both 3.2 mg phenol/litre) found by Hermens (1984) and
Oris et al. (1991). De Neer et al. (1988), however, found a
considerably lower NOEC value of 0.16 mg phenol/litre for growth of
Daphnia magna under similar experimental conditions.
Exposure of adult Brachydanio rerio to 2.2, 4.9 or 24 mg
phenol/litre for 3 months resulted in 67% mortality at the highest
concentration; the no-observed-lethal concentration (NOLC) was 4.9
mg phenol/litre. At 24 mg phenol/litre, only immature oocytes were
found in surviving fish; at the two lower concentrations both
immature and mature oocytes were observed, whereas spawning was
delayed. In subsequent embryo-larval tests, starting with the eggs
of exposed adults, mortality appeared to be maximal during embryonic
development and the initial larval stage. All larvae died within 12
days at 4.9 mg phenol/litre. At 2.2 mg phenol/litre, larval
mortality was still slightly increased, but surviving animals showed
normal growth and development (Razani et al., 1986b).
In two embryo-larval bioassays on Pimephales promelas, growth
proved to be the most sensitive criterion: the NOEC values were 0.75
and 1.83 mg phenol/litre (De Graeve et al., 1980; Holcombe et
al., 1982).
The results of the embryo-larval test on Salmo gairdneri
given by Birge et al. (1979) (LC1:0.2 µg/litre) and Black et
al. (1983) (NOEC:9 µg/litre) are much lower than those obtained by
De Graeve et al. (1980) (NOECgrowth: 0.1 mg/litre), probably
because the latter test was started with eyed eggs, whereas the
former two tests were started with just-fertilized eggs.
In addition, Birge et al. (1979) studied the influence of
phenol in an embryo-larval test on Carassius auratus and found a
LC1 of 2.0 µg phenol/litre.
Dumpert (1987) reported a NOLC value of 10 mg phenol/litre for
larvae of the amphibian Xenopus laevis; larval mortality was 100%
within 3 weeks at 50 mg phenol/litre. Larval growth was slightly,
but not significantly, retarded at 5 and 10 mg phenol/litre.
Hatching was normal at all tested concentrations. However, the
results may not be reliable because test solutions were renewed only
once a week, whereas aeration may also have contributed to
undetected loss of phenol. In other embryo-larval bioassays on five
amphibian species, Rana ripiens and Rana catesbeiana were the
least tolerant. These species exhibited about equal sensitivity to
phenol (LC1: 1.0 and 1.1 mg phenol/litre, respectively; LC10:
5.2 and 8.5 mg phenol/litre, respectively (Birge et al., 1980).
9.2.2 Marine organisms
9.2.2.1 Short-term studies
In acute toxicity studies on some marine organisms
(crustaceans, worms, snails and fish), the LC50 values ranged from
8.8-330 mg phenol/litre (see Table 11). In general, the
sensitivities of marine and freshwater organisms for phenol were
similar.
At a sublethal phenol concentration, activities of some enzymes
appeared to be decreased in the brain, liver and muscle tissue of
Sarotherodon mossambicus; this effect was independent of salinity
(Ravichandran & Anantharaj, 1984).
9.2.2.2 Long-term studies
No adequate data are available on long-term toxicity to marine
organisms.
9.2.3 Accumulation
The bioconcentration factor of phenol may be calculated, using
a log Pow value of 1.46 (pH not stated, see Table 1) and the
formula log BCF = 0.79 log Pow -0.40 (Veith & Kosian, 1983). This
yields a bioconcentration factor of 5.7, which is very low and does
not indicate any potential for bioaccumulation.
The experimental bioconcentration factors, reported by Hardy
et al. (1985) for algae, by Erben (1983) for flatworms, by Erben
(1982) for snails, by Key & Scott (1986) for crabs, and by Call et
al. (1980) and Kobayashi & Akitake (1975) for fish, are in
agreement with the calculated value. Other studies, however,
reported higher values. The bioconcentration factor for Daphnia
magna, as assessed from 14C measurements, was reported to be
1375 after 24 h, with an estimated half-life upon depuration of 8 h.
A lower bioconcentration factor (277) was calculated from uptake and
depuration rate constants (Dauble et al., 1986). The
bioconcentration factors for phenol, as determined by 14C
measurements in a 5-day experiment with activated sludge, in a 24
h-experiment with the alga Chlorella fusca, and a 3-day experiment
with the fish Leuciscus idus melanotus, were 2200, 200 and 20,
respectively (Freitag et al., 1985).
Uptake was usually complete (the equilibrium level reached)
within 1-2 days. Initially, excretion was also rapid, but it usually
slowed down after some hours, and depuration was reported to be
incomplete. The amount of unchanged phenol still present after 24 h
in the alga Scenedesmus quadricauda, for example, was 22% (Hardy
et al., 1985), whereas radioactivity from 14C showed a retention
of 80% after 96 h of depuration in the mud crab Panopeus herbstii
(Key & Scott, 1986).
Based on reported and estimated bioconcentration factors for
aquatic organisms, phenol is not expected to bioaccumulate
significantly.
9.2.4 Metabolism
The metabolism of phenol in fish yields the known phenyl
conjugates (phenyl sulfate and phenyl glucuronide) and quinol
sulfate (Kobayashi et al., 1976; Layiwola & Linnecar, 1981; Nagel,
1983; Nagel & Urich, 1983; Kasokat et al., 1987). In the excreta
of the frog Rana temporaria, the same metabolites were found after
phenol injection, together with catechol, catechol sulfate and
traces of resorcinol. Xenopus laevis, another amphibian, appeared
to be unable to glucuronidate phenol, but compensated this by
increasing the production of other metabolites compared to Rana
temporaria (more quinol sulfate and phenyl sulfate, and, in
addition, resorcinol sulfate) (Beyer & Frank, 1985; Görge et al.,
1987).
Phenol metabolism may be induced by prior exposure to phenol or
phenol derivatives, as was observed for sulfate conjugation in the
clam Ruditapes phillippinarum (Kobayashi et al., 1987).
9.3 Terrestrial organisms
Phenol may be taken up by, and stored in, the cuticle membranes
of various plants, such as tomatoes and green pepper fruits, and
rubber leaves (Shafer & Schönherr, 1985). Labelled phenol was
demonstrated to be taken up by soybean roots. The label stayed in
the roots, and was not transported to the shoot, which was
attributed to the metabolism of phenol by the plant into immobile
compounds (McFarlane et al., 1987). Millet seeds appeared to be
more sensitive to the toxicity of phenolic compounds than lettuce
and cucumber seeds. The 120-h EC50 value for root elongation
inhibition in millet was determined to be 120-170 mg phenol/litre
(Wang, 1986).
The toxicity of phenols for four earthworm species (Neuhauser
et al., 1986) was compared with those of other chemicals using two
standardized tests developed by the EEC, the 2-day contact test and
the 14-day artificial soil test. Of ten classes of chemicals, phenol
was the most toxic in the contact test, with LC50 values of
2.4-10.6 µg/cm2, regardless of species. A lower relative toxicity
was reported by the same authors using the artificial soil test.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
The main way in which the general population can be exposed, on
a long-term basis, to phenol present in ambient air is as a result
of industrial emissions and various combustion processes. Other
inhalation sources include the decomposition of organic materials,
liquid manure, and the atmospheric degradation of benzene.
Inhalation and dermal exposure may arise from contact with
contaminated water or consumer products containing phenol. Indirect
exposure of man through the food chain is not likely to add
significantly to long-term inhalation exposure, in view of the short
life-time of the compound in the environment (section 10.2.1.).
Individuals may ingest phenol via drinking-water from contaminated
surface water or ground water. Repeated oral exposure may arise from
the consumption of smoked food items. Endogenous production of
phenol may be influenced by the diet and exposure to certain drugs
and other xenobiotics.
The exposure data available are inadequate to determine the
degree of exposure of the general population or of specific groups
at risk, including workers.
An upper-limit estimate of the daily intake can be made for
long-term exposure of the general population. In this hypothetical
case, it is assumed that an individual will be maximally exposed to
phenol through continuous inhalation of air from a heavily
industrialized area, with frequent consumption of smoked food items
with a high phenol content, and of drinking-water containing phenol
up to the taste threshold. The estimate is summarized in the table
below:
Source Quantity of source Phenol Phenol intake
concentration
Air 20 m3/day 200 µg/m3 4 mg/day
Smoked food 200 g/week 70 mg/kg 2 mg/day
items
Drinking-water 2 litres/day 300 µg/litre 0.6 mg/day
Assuming an average body weight of 70 kg, the total daily
intake of this maximally exposed individual will be 0.1 mg/kg body
weight per day. The daily intake by the general population can be
expected to be much less than this figure.
10.1.2 Toxicity
Phenol has moderately acute toxicity for animals. The oral
LD50 for various animal species range from 300 to 600 mg phenol/kg
body weight, and the LC50 for rats by inhalation is more than 900
mg phenol/m3.
In humans, the lowest acutely lethal oral dose was reported to
be 4.8 g, which is approximately 70 mg/kg body weight. Local, as
well as systemic, effects have been reported in humans, consisting
of irritation, necrosis, cardiovascular effects, metabolic acidosis,
neurological effects and methaemoglobinaemia. Several fatal cases
have been reported after oral or dermal intoxication. No documented
cases of death by inhalation of phenol have been found.
Solutions of phenol are corrosive to the skin and eyes. Phenol
vapours can irritate the respiratory tract. Phenol is not a skin
sensitiser in guinea-pigs or humans.
The most important effects reported in short-term animal
studies were neurotoxicity, liver and kidney damage, respiratory
effects and growth retardation. Toxic effects in rat kidney have
been reported to occur at oral dose levels of 40 mg/kg per day or
more. Liver toxicity was evident in rats administered at least 100
mg/kg per day. In a limited 14-day study on rats, an oral NOAEL of
12 mg/kg per day was reported based on kidney effects. In this
experiment, miosis (an iris response to light) was inhibited at 4
mg/kg per day (the lowest dose tested). However, the health
significance of this finding is not clear. Some biochemical changes
have been reported to occur in the intestinal mucosa and kidneys of
mice at dose levels below 1 mg/kg per day. The toxicological value
of these insufficiently reported biochemical observations is not
known.
There have been no long-term general toxicity studies in
animals or adequate epidemiological studies.
No adequate studies on the reproductive toxicity of phenol have
been reported. Phenol has been identified as a developmental
toxicant in studies with rats and mice. In two multiple-dose rat
studies, NOAELs of 40 mg/kg per day (the LOAEL was 53 mg/kg per day)
and 60 mg/kg per day (the LOAEL was 120 mg/kg per day) have been
reported. For the mouse, the NOAEL was 140 mg/kg per day (the LOAEL
was 280 mg/kg per day).
There is some evidence that phenol is genotoxic to mammalian
cells in vitro. Based on the induction of bone marrow micronuclei
in several studies with mice, phenol may have genotoxic potential.
Oral (drinking-water) animal carcinogenicity bioassays did not
give evidence of a carcinogenic potential of phenol. No animal
inhalation or adequate dermal carcinogenicity studies are available.
Two-stage carcinogenicity studies with mice showed that phenol
applied to the skin does have tumour-promoting activity. Adequate
human data on carcinogenicity are not available.
10.1.3 Evaluation
Accidental high exposure to phenol may cause severe local
effects, systemic intoxications and even death. The available data
do not suggest a strong potential for cumulative health effects from
chronic exposure.
The lowest NOAELs identified are for kidney and developmental
effects, and in rats are in the range of 12-40 mg/kg body weight per
day. The Task Group decided to derive a tolerable daily intake
(TDI), taking into consideration this range. An uncertainty factor
of 200 (including factors of 10 for interspecies variation, 10 for
intraspecies variation, and 2 to account for the limited data base
on the toxicity of phenol in animals) was considered appropriate. A
range of 60-200 µg/kg per day was recommended as the upper limit of
the TDI by the Task Group. As the Task Group's upper-limit estimate
of human daily intake is 100 µg/kg body weight per day (section
10.1.1), it can be concluded that the average general population
exposure to phenol from all sources will be well below this range.
There remain, however, two reasons for concern. The available
data suggest that phenol may be genotoxic, and there is insufficient
data to discount the possibility that phenol is carcinogenic. For
these reasons, it is particularly important that this evaluation of
phenol be kept under periodic review.
10.2 Evaluation of effects on the environment
10.2.1 Environmental levels
Once released into the environment, intercompartmental
transport of phenol may occur by wet deposition from air to sea
water and surface water and soil, and, as the compound can be
expected to be highly mobile in soil, by leaching through soil.
Evaporation will be slow from water and can only be expected
following contamination of relatively dry soil.
Phenol, however, is generally not likely to persist in either
air, sea or surface water, soil or sewage. It readily reacts
photochemically and is rapidly biodegraded aerobically, mainly to
carbon dioxide. Anaerobic degradation to carbon dioxide or methane
also occur. Half-lives will be in the range of several hours for
photodegradation and in the range of hours to days for aerobic
biodegradation. Anaerobic biodegradation also occurs, albeit at a
slower rate. Low removal rates of phenol in ground water and soil
may occur, e.g., following spills, with subsequent inhibition of the
microbial populations.
The scarce environmental exposure data available give some
support for the above conclusions:
* reported ambient air levels are low (< 8 µg/m3 for urban
areas; < 200 µg/m3 for heavily industrialized areas);
* phenol has been detected in rain water;
* reported surface water levels are low (< 24 µg/litre);
* levels in ground water have only been found at highly
contaminated sites.
10.2.2 Toxicity
Based on reported and estimated bioconcentration factors for
aquatic organism, phenol is not expected to bioaccumulate
significantly. The data base on aquatic toxicity is considered
adequate for evaluation. Phenol is toxic to aquatic organisms: the
lowest EC50 for water organisms is estimated to be 3.1 mg/litre
(48-h LC50 for Ceriodaphnia dubia). The lowest chronic NOEC is
estimated to be 0.2 µg/litre (8-day LC1 for Salmo gairdneri).
Applying the modified US EPA method, an Environmental Concern Level
of 0.02 µg/litre can be derived for water. In general, fish are the
most sensitive species and the sensitivities of marine and
freshwater organisms are similar. Adequate data on plants and
terrestrial organisms are not available.
10.2.3 Evaluation
The scarce exposure data available do not allow any firm
conclusions with regard to the degree of risk from phenol to either
aquatic or terrestrial ecosystems. However, in view of the derived
Environmental Concern Level of phenol for aquatic organisms, it is
reasonable to assume that these organisms may be at risk in any
surface or sea water subject to phenol contamination, in spite of
the rapid degradation of this compound.
11. FURTHER RESEARCH
There is a need for the following items:
a) further investigation of the in vivo genotoxicity of phenol;
b) more animal toxicology studies, including 90-day oral and
inhalation studies, carcinogenicity bio-assays by the
inhalation route, and neurotoxicity and multigeneration
reproductive toxicity studies (including evaluation in
offspring);
c) assessment of environmental and occupational exposures and
evaluation of health effects in occupational populations;
d) further evaluation of the dose-duration-effect relationships,
reversibility/persistence and health significance of the
reported phenol-induced inhibition of the pupillary response to
light;
e) further data on the toxicity of phenol for plants and
terrestrial organisms.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic risk of phenol was evaluated in 1989 by the
International Agency for Research on Cancer (IARC, 1989). The
summary evaluation from the IARC Monograph is reproduced here.
"Exposures
Phenol is a basic feedstock for the production of phenolic
resins, bisphenol A, caprolactam, chlorophenols and several
alkylphenols and xylenols. Phenol is also used in disinfectants and
antiseptics. Occupational exposure to phenol has been reported
during its production and use, as well as in the use of phenolic
resins in the wood products industry. It has also been detected in
automotive exhaust and tobacco smoke.
Experimental carcinogenicity data
Phenol was tested for carcinogenicity by oral administration in
drinking-water in one strain of mice and one strain of rats. No
treatment-related increase in the incidence of tumours was observed
in mice or in female rats. In male rats, an increase in the
incidence of leukaemia was observed at the lower dose, but not at
the higher dose. Phenol was tested extensively in the two-stage
mouse skin model and showed promoting activity.
Human carcinogenicity data
In one case-control study of workers in various wood
industries, an increased risk was seen for tumours of the mouth and
respiratory tract in association with exposure to phenol; however,
the number of cases was small and confounding exposures were
inadequately controlled.
Other relevant data
In humans, phenol poisoning can occur after skin absorption,
inhalation of vapours or ingestion. Acute local effects are severe
tissue irritation and necrosis. At high doses, the most prominent
systemic effect is central nervous system depression. Phenol causes
irritation, dermatitis, central nervous system effects and liver and
kidney toxicity in experimental animals.
Phenol induced micronuclei in female mice and sister chromatid
exchange in cultured human cells. It did not inhibit intercellular
communication in cultured animal cells. It induced mutation but not
DNA damage in cultured animal cells. It did not induce recessive
lethal mutation in Drosophila. It had a weak effect in inducing
mitotic segregation in Aspergillus nidulans. Phenol did not induce
mutation in bacteria.
Evaluation
There is inadequate evidence for the carcinogenicity of phenol
in humans.
There is inadequate evidence for the carcinogenicity of phenol
in experimental animals.
Overall evaluation
Phenol is not classifiable as to its carcinogenicity to humans
(Group 3)".
REFERENCES
Abou-el-Makarem MM, Millburn P, Smith RL, & Williams RT (1967)
Biliary excretion of foreign compounds. Biochem J, 105: 1269-1274.
Abraham AJ (1972) A case of carbolic acid gangrene of the thumb. Br
J Plast Surg, 25: 282-284.
ACGIH (1991) Threshold limit values for chemical substances and
physical agents and biological exposure indices for 1991-1992.
Cincinnati, Ohio, American Conference of Environmental Industrial
Hygienists.
Adachi A, Asaka Y, Ozasa M, Sawai N, & Kobayashi T (1987) [Formation
of cyanide ion by the reaction of phenol with nitrous acid in waste
water.] Eisei Kagaku, 33(6): 445-448 (in Japanese, with English
abstract).
Aelion CM, Swindoll CM, & Pfaender FK (1987) Adaptation to and
biodegradation of xenobiotic compounds by microbial communities from
a pristine aquifer. Appl Environ Microbiol, 53(9): 2212-2217.
Agrawal HP (1987) Evaluation of the toxicity of phenol and sodium
pentachlorophenate to the snail Indoplanorbis exustus (Deshayes).
J Anim Morphol Physiol, 34(1-2): 107-112.
Alabaster JS (1969) Survival of fish in 164 herbicides,
insecticides, fungicides, wetting agents and miscellaneous
substances. Int Pest Control, March/April: 29-35.
Alekseyev VA & Antipin BN (1976) Toxicological characteristics and
symptoms of acute phenol poisoning in some freshwater crustaceans
and molluscs. Hydrobiol J, 12(2): 27-33.
Alexander DG & Clarke RMcV (1978) The selection and limitations of
phenol as a reference toxicant to detect differences in sensitivity
among groups of rainbow trout ( Salmo gairdneri). Water Res, 12:
1085-1090.
Al-Sabti K (1985) Frequency of chromosomal aberrations in the
rainbow trout, Salmo gairdneri, exposed to 5 pollutants. J Fish
Biol, 26(1): 13-20.
American Public Health Association, American Waterworks Association,
Water Pollution Control Federation (1985) Standard methods for the
examination of water and wastewater - Phenol, 16th ed. Washington,
DC, American Public Health Association, pp 556-570.
Amlathe S, Upadhyay S, & Gupta VK (1987) Spectrophotometric
determination of trace amounts of phenol in waste water and
biological fluids. Analyst, 112: 1463-1465.
Amoore JE & Hautala E (1983) Odor as an aid to chemical safety; odor
thresholds compared with threshold limit values and volatilities for
214 industrial chemicals in air and water dilution. J Appl Toxicol,
3: 272-290.
Andersen W (1869) Fatal misadventure with carbolic acid. Lancet, 1:
179.
Anniko M, Hellstrom S, Schmidt SH, & Spandow O (1988) Toxic effects
on inner ear of noxious agents passing through the round window
membrane. Acta Otolaryngol (Stockholm), 457(Suppl): 49-56.
Aquino MD, Korol S, Santini P, & Moretton J (1988) Biodegradation of
phenolic compounds: I. Improved degradation of phenol and benzoate
by indigenous strains of Acinobacter and Pseudomonas. Rev Latinoam
Microbiol, 30(3): 283-288.
Aranyi C, O'Shea WJ, Graham JA, & Miller FJ (1986) The effects of
inhalation of organic chemical air contaminants on murine lung host
defences. Fumdam Appl Toxicol, 6(4): 713-720.
Arvin E, Jensen BK, & Gundersen TA (1991) Biodegradation kinetics of
phenol in an aerobic biofilm at low concentrations. Water Sci
Technol, 23: 1375-1384.
Atkinson GL & Shupak RC (1989) Acute bronchospasm complicating
intercostal nerve block. Anesth Analg, 68: 400-401.
Atkinson R, Darnall KR, Lloyd AC, Winer AM, & Pitts JN Jr (1979)
Kinetics and mechanisms of the reactions of the hydroxyl radical
with organic compounds int he gas phase. Adv in Photochem, 11: 375.
ATSDR (1989) Toxicological profile for phenol. Atlanta, Georgia,
Agency for Toxic Substances and Disease Registry, 111 pp
(ATSDR/TP-89/20).
Babich H & Stotzky G (1985) A microbial assay for determining the
influence of physicochemical environmental factors on the toxicity
of organics: phenol. Arch Environ Contam Toxicol, 14(4): 409-416.
Baird RB, Kuo CL, Shapiro JS, & Yanko WA (1974) The fate of
phenolics in waste water - determination by direct injection GLC and
Warburg respirometry. Arch Environ Contam Toxicol, 2: 165-169.
Bak F & Widdell F (1986) Anaerobic degradation of phenol and phenol
derivatives by Desulfobacterium phenolicum, new species. Arch
Microbiol, 146(2): 177-180.
Baker MD & Mayfield CI (1980) Microbial and nonbiological
decomposition of chlorophenols and phenol in soil. Water Air Soil
Pollut, 13: 411-424.
Baker EL, Bertozzi PE, Field PH, Basteyns BJ, & Skinner HG (1978)
Phenol poisoning due to contaminated drinking water. Arch Environ
Health, 33: 89-94.
Barale R, Marrazzini A, Betti C, Vangelisti F, Loprieno N, & Barrai
J (1990) Genotoxicity of two metabolites of benzene: phenol and
hydroquinone show strong synergistic effects in vivo. Mutat Res,
244: 15-20.
Baranowska-Dutkiewicz B (1981) Skin absorption of phenol from
aqueous solutions in men. Int Arch Occup Environ Health, 49: 99-104.
Behl CR, Linn EE, Flynn GL, Pierson CL, Higuchi WI, & Ho NFH (1983a)
Permeation of skin and eschar by antiseptics: I. Baseline studies
with phenol. J Pharm Sci, 72(4): 391-397.
Behl CR, Linn EE, Flynn GL, Ho NFH, Higuchi WI, & Pierson CL (1983b)
Permeation of skin and eschar by antiseptics: II. Influence of
controlled burns on the permeation of phenol. J Pharm Sci, 72(4):
397-400.
Bennett IL, James DF, & Golden A (1950) Severe acidosis due to
phenol poisoning. Report of two cases. Ann Intern Med, 32: 324-327.
Benzon HT (1979) Convulsions secondary to intravascular phenol: a
hazard of celiac plexus block. Anesth Analg, 58(2): 150-151.
Beyer J & Frank G (1985) Hydroxylation and conjugation of phenol by
the frog, Rana temporaria. Xenobiotica, 15(4): 277-280.
Bergshoeff G (1960) [Determination of phenols in air and water. Part
III-A. Instructions for the method using p-nitroaniline and for the
method using ultraviolet spectrophotometry.] Delft, The Netherlands,
Dutch Organization for Applied Scientific Research (TNO-IMG) (Report
No. F765) (in Dutch).
Bernard DP & Salt DB (1982) Carcinogen specificity of gamma-glutamyl
transpeptidase-rich foci chemically induced in hamster buccal pouch
epithelium. 1st Int. Conf., Boston, Mass., 173-182.
Birge WJ, Black JA, Hudson JE, & Bruser DM ((1979) Embryo larval
toxicity test with organic compounds - Aquatic Toxicology. In:
Proceedings of the Second Annual Symposium on Aquatic Toxicology, 31
October-1 November 1977. pp 131-147.
Birge WJ, Black JA, & Kuehne RA (1980) Effects of organic compounds
on amphibial reproduction. Lexington, Kentucky Water Resources
Research Institute, 50 pp.
Blaber LC & Gallagher JP (1971) The facilitory effects of catechol
and phenol at the neuromuscular junction of the cat.
Neuropharmacology, 10: 153-159.
Black JA, Birge WJ, Westerman AC, & Francis PC (1983) Comparative
aquatic toxicology of aromatic hydrocarbons. Fundam Appl Toxicol,
3(5): 353-358.
Bohrman JS, Burg JR, Elmore E, Gulati DK, Barfknecht TR, Niemeier
RW, Dames BL, Toraason M, & Langenbach R (1988) Interlaboratory
studies with the Chinese hamster V79 cell metabolic cooperation
assay to detect tumor-promoting agents. Environ Mol Mutagen, 12(1):
33-51.
Boiano JM (1985) Health hazard evaluation report, University of
Pennsylvania, Medical Education Building, Philadelphia,
Pennsylvania. Cincinnati, OH, National Institute for Occupational
Safety and Health (HETA 84-098-1497; NTIS/PB85-220861).
Bolcsak LE & Nerland DE (1983) Inhibition of erythropoiesis by
benzene and benzene metabolites. Toxicol Appl Pharmacol, 69(3):
363-368.
Bollig FJ & Decker KH (1980) [Phenol resins.] Kunststoffe, 70: 672
(in German).
Bolt HM (1977) Structural modifications in contraceptive steroids
altering their metabolism and toxicity. Arch Toxicol, 39: 13-19.
Borovik EB & Dmitriev MT (1981) [Principles of air interchange as a
basis for the hygienic standardization of the air inside hospitals.]
Gig i Sanit, 2: 22-25 (in Russian).
Boutwell RK & Bosch DK (1959) The tumour promoting action of phenol
and related compounds for mouse skin. Cancer Res, 19: 413-424.
Boutwell RK, Rusch HP, & Bosch DK (1955) The action of phenol and
related compounds in tumour formation. Proc Am Assoc Cancer Res, 2:
6-7.
Boutwell RK, Rusch HP, & Booth B (1956) Tumour production by phenol
and related compounds. Proc Am Assoc Cancer Res, 2: 96.
Boyd SA (1982) Adsorption of substituted phenols by soil. Soil Sci,
134(5): 337-343.
Bradbury SP, Henry TR, Niemi GJ, Carlson RW, & Snarski VM (1989) Use
of respiratory-cardiovascular responses of rainbow trout ( Salmo
gairdneri) in identifying acute toxicity syndromes in fish: Part
3. Polar narcotics. Environ Toxicol Chem, 9(3): 247-262.
Bratzler LJ, Spooner ME, Weatherspoon JB, & Maxey JA (1969) Smoke
flavour as related to phenol, carbonyl and acid content of Bologna.
J Food Sci, 34: 146-148.
Bray HG, Humphris BG, Thorpe WV, White K, & Wood PB (1952a) Kinetic
studies of the metabolism of foreign organic compounds. 3. The
conjugation of phenol with glucuronic acid. Biochem J, 52: 416-419.
Bray HG, Humphris BG, Thorpe WV, White K, & Wood PB (1952b) Kinetic
studies of the metabolism of foreign organic compounds. 4. The
conjugation of phenol with sulfuric acid. Biochem J, 52: 419-423.
Bray HG, Thorpe WV, & White K (1952c) Kinetic studies of the
metabolism of foreign organic compounds. 5. A mathematical model
expressing the metabolic fate of phenols, benzoic acids and their
precursors. Biochem J, 52: 423-430.
Bringmann G & Kuhn R (1959) [Studies on water toxicology using
protozoa as test organisms.] Gesundheits-Ing, 80: 239-242 (in
German).
Bringmann G & Kuhn R (1977) [Threshold values for the harmful effect
of water pollutants on bacteria ( Ps. putida) and green algae ( Sc.
quadricauda) in the cell reproduction inhibition test.] Z Wasser
Abwasser Forsch, 80: 87-98 (in German).
Bringmann G & Kuhn R (1978) [Threshold values for the harmful effect
of water pollutants on blue algae ( Microcystis aeruginosa) and
green algae ( Scenedesmus quadricauda) in the cell reproduction
inhibition test.] Vom Wasser, 80: 45-60 (in German).
Bringmann G & Kuhn R (1980) Comparison of the toxicity thresholds of
water pollutants to bacteria, algae, and protozoa in the cell
multiplication inhibition test. Water Res, 14(3): 231-241.
Bringmann G, Kuhn R, & Winter A (1980) [Determination of the harmful
biological action of water endangering substances on protozoa. III.
Saprozic flagellates.] Z Wasser Abwasser Forsch, 13(5): 170-173 (in
German).
Brodzinsky R & Singh HB (1982) Volatile organic chemicals in the
atmosphere: an assessment of available data. Research Triangle Park,
North Carolina, US Environmental Protection Agency, Environmental
Sciences Research Laboratory (EPA/600/3-83/027a; NTIS PB83-195503).
Brown VM, Jordan DHM, & Tiller BA (1967) The effects of temperature
on the acute toxicity of phenol to rainbow trout in hard water.
Water Res, 1: 587-594.
Brown VKH, Box VL, & Simpson BJ (1975) Decontamination procedures
for skin exposed to phenolic substances. Arch Environ Health, 30:
1-6.
Bruce RM, Santodonato J, & Neal MW (1987) Summary review of the
health effects associated with phenol. Toxicol Ind Health, 3(4):
535-568.
Budavari S, O'Neil MJ, Smith A, & Heckelman PE ed. (1989) The Merck
index. Rahway, New Jersey, Merck & Co., Inc., p 1150.
Buikema AL, McGinness MJ, & Cairns J (1979) Phenolics in aquatic
ecosystems. A selected review of recent literature. Mar Environ Res,
2(2): 87-181.
Bulsiewicz H (1977) The influence of phenol on chromosomes of mice
in the process of spermatogenesis. Folia Morphol (Warsaw), 36:
13-22.
Cairns J, Messenger DJ, & Calhoun WF (1976) Invertebrate response to
thermal shock following exposure to acutely sub-lethal
concentrations of chemicals. Arch Hydrobiol, 77(2): 164-175.
Cairns J, Buikema AL Jr, Heath AG, & Parker BC (1978) Effects of
temperature on aquatic organism sensitivity to selected chemicals.
Blackburg, Virginia, Virginia Water Resources Research Center
(Bulletin 106).
Call DJ, Brooke LT, & Lu PY (1980) Uptake, elimination, and
metabolism of three phenols by Fathead minnow. Arch Environ Contam
Toxicol, 9(6): 699-714.
Campbell NRC, Van Loon JA, & Weinshilboum RM (1987) Human liver
phenol sulfotransferase: assay conditions, biochemical properties
and partial purification of isozymes of the thermostable form.
Biochem Pharmacol, 36(9): 1435-1446.
Capel ID, French MR, Millburn P, Smith RL, & Williams RT (1972a)
Species variations in phenol metabolism. Biochem J, 127: 25-26.
Capel ID, French MR, Millburn P, Smith RL, & Williams RT (1972b) The
fate of 14C phenol in various species. Xenobiotica, 2: 25-34.
Capel ID, Millburn P, & Williams RT (1974) Monophenyl phosphate, a
new conjugate of phenol in the cat. Biochem Soc Trans, 2: 305-306.
Cassidy MK & Houston JB (1980) In vivo assessment of extrahepatic
conjugative metabolism in first pass effects using the model
compound phenol. J Pharm Pharmacol, 32(5): 57-59.
Cassidy MK & Houston JB (1984) In vivo capacity of hepatic and
extrahepatic enzymes to conjugate phenol. Drug Metab Dispos, 12:
619-624.
Chagnon N & Hlohowskyj I (1989) Effects of phenol exposure on the
thermal tolerance ability of the central stoneroller minnow. Bull
Environ Contam Toxicol, 42(4): 614-619.
Chemfacts (1978-1981) Chemical Data Sources - Chemfacts.
Publications for the Netherlands, Belgium, West-Germany, France,
Great Britain, Spain, Italy, Portugal and Scandinavia.
Chen TH, Kavanagh TJ, Chang CC, & Trosko JE (1984) Inhibition of
metabolic cooperation in Chinese hamster V79 cells by various
organic solvents and simple compounds. Cell Biol Toxicol, 1(1):
155-171.
Chesney RH, Sollitti P, & Rubin HE (1985) Incorporation of phenol
carbon at trace concentrations by phenol-mineralizing microorganisms
in fresh water. Appl Environ Microbiol, 49(1): 15-18.
CID-TNO (1984) Databases ECDIN and PROMT.
Ciranni R, Barale R, Ghelardini G, & Loprieno N (1988a) Benzene and
the genotoxicity of its metabolites. I. Transplacental activity in
mouse fetuses and in their dams. Mutat Res, 208: 61-67.
Ciranni R, Barale R, Ghelardini G, & Loprieno N (1988b) Benzene and
the genotoxicity of its metabolites. II. The effect of the route of
administration on the micronuclei and bone marrow depression in
mouse bone marrow cells. Mutat Res, 209: 23-28.
Coan ML, Baggs RB, & Bosmann HB (1982) Demonstration of direct
toxicity of phenol on kidney. Chem Pathol Pharmacol Res Commun,
36(2): 229-239.
Colvin RJ & Rozich AR (1986) Phenol growth kinetics of heterogenous
populations in a two-stage continuous culture system. Water Pollut
Control Fed, 58(4): 326-332.
Conning DM & Hayes MJ (1970) The dermal toxicity of phenol, and
investigation of the most effective first-aid measures. Br J Ind
Med, 27: 155-159.
Cooper IS, Hirose T, Matsuoka S, Roth D, Waltz JM, & Ericsson AD
(1965) Specific neurotic perfusion: a new approach to selected cases
of pain and spasticity. Neurology, 15: 985.
Cotruvo JA, Simmon VF, & Sponggord RJ (1977) Investigation of
mutagenic effects of products of ozonation reactions in water. Ann
NY Acad Sci, 298: 124-140.
Crebelli R, Conti G, & Carere A (1987) On the mechanism of mitotic
segregation induction in Aspergillus nidulans by benzene hydroxy
metabolites. Mutagenesis, 2(3): 235-238.
Cronin TD & Brauer RO (1949) Death due to phenol contained in
Foille. J Am Med Assoc, 139: 777-779.
Curtis C, Lima A, Lozano SJ, & Veith GD (1982) Philadelphia,
Pennsylvania, American Society for Testing and Materials, p 170
(ASTM Special Technical Publication No. 766).
Dalela RC, Rani S, & Verna SR (1980) Influence of pH on the toxicity
of phenol and its 2 derivatives pentachlorophenol and dinitrophenol
to some fresh water teleosts. Acta Hydrochim Hydrobiol, 8(6):
623-628.
Dalin NM & Kristofferson R (1974) Physiological effects of a
sublethal concentration of inhaled phenol on the rat. Ann Zool Fenn,
11: 193-199.
Dangé AD (1986) Metabolic effects of naphthalene, toluene or phenol
intoxication in the cichlid fish tilapia, Oreochromis mossambicus:
changes in aminotransferase activities. Environ Pollut, A42:
311-323.
Dauble DD, Carlile DW, & Hanf RW Jr (1986) Bioaccumulation of fossil
fuel components during single-compound and complex-mixture exposures
of Daphnia magna. Bull Environ Contam Toxicol, 37: 125-132.
Daum F, Cohen MJ, & McNamara H (1976) Experimental toxicological
studies on a phenol detergent associated with neonatal
hyperbilirubinemia. J Pediatr, 89: 853-854.
Davis HC & Hidu H (1969) Effects of pesticides on embryonic
development of clams and oysters and on survival and growth of the
larvae. US Dept. Inter., Fish Wildl. Bull., 67: 393.
De Ceaurriz JC, Micillino JC, Bonnet P, & Guenier JP (1981) Sensory
irritation caused by various industrial airborne chemicals. Toxicol
Lett, 9: 137-143.
De Graeve GM, Geiger WL, Meyer JS, & Bergman HL (1980) Acute and
embryo-larval toxicity of phenolic compounds to aquatic biota. Arch
Environ Contam Toxicol, 9: 557-568.
De Groote V & Lambotte C (1960) A case of fatal phenol poisoning in
a newborn infant. Ann Méd Lég, 40: 288-290.
Deichmann WB (1944) Phenol studies. V. The distribution,
detoxification, and excretion of phenol in the mammalian body. Arch
Biochem, 2: 345-355.
Deichmann WB (1949) Local and systemic effects following skin
contact with phenol: a review of the literature. J Ind Hyg Toxicol,
31: 146-154.
Deichmann WB & Keplinger ML (1963) Phenols and phenolic compounds.
In: Patty FA ed. Industrial hygiene and toxicology. New York,
Interscience Publishers, pp 2567-2627.
Deichmann WB & Oesper P (1940) Ingestion of phenol - effects of the
albino rat. Ind Med, 9: 296.
Deichmann WB & Schafer LJ (1942) Phenols studies - I: Review of the
literature. II: Quantitative spectrophotometric estimation of free
and conjugated phenol in tissues and fluids. III: Phenol content of
normal human tissues and fluids. Am J Clin Pathol, 12: 129-143.
Deichmann WB & Witherup S (1944) Phenol studies - VI: The acute and
comparative toxicity of phenol and o-, m-, and p-cresols for
experimental animals. J Pharmacol Exp Ther, 80: 233-240.
Deichmann WB, Kitzmiller KV, & Witherup S (1944) Phenols studies -
VII: Chronic phenol poisoning, with special reference to the effects
upon experimental animals of the inhalation of phenol vapour. Am J
Clin Pathol, 14: 273-277.
Deichmann WB, Miller T, & Roberts JB (1950) Local and systemic
effects following application of dilute solutions of phenol in water
and in camphor-liquid petrolatum on the skin of animals. Arch Ind
Hyg Occup Med, 2: 454-461.
Deichmann WB, Witherup S, & Dierker M (1952) Phenol studies - XII:
The percutaneous and alimentary absorption of phenol by rabbits with
recommendations for the removal of phenol from the alimentary tract
or skin of persons suffering exposure. J Pharmacol Exp Ther, 105:
265-272.
Delfino JJ & Dube DJ (1976) Persistent contamination of ground water
by phenol. J Environ Sci Health, pp 345-355.
DeMeio RH & Arnolt RI (1944) Phenol conjugation - II: The
conjugation by rat and cat tissues in vitro. J Biol Chem, 156:
577-583.
Dement J, Wallingford K, & Zumwalde R (1973) Industrial hygiene
survey of Owens-Corning Fiberglas, Kansas, City, Kansas. Cincinnati,
Ohio, National Institute for Occupational Safety and Health (Report
No. IW 35.16).
Demerec M, Bertani G, & Flint J (1951) A survey of chemicals for
mutagenic action on E. coli. Am Nat, 85: 119-136.
Den Boeft J, Kruiswijk FJ, & Schulting FL (1984) [Air pollution by
combustion of solid fuels.] The Hague, Ministry of Housing, Physical
Planning and Environment (Publication Lucht No. 37) (in Dutch).
De Neer JW, Seinen W, & Hermens JLM (1988) Growth of Daphnia magna
exposed to mixtures of chemicals with diverse modes of action.
Ecotoxicol Environ Saf, 15(1): 72-77.
Dimitriev MT & Mischchikhin VA (1983) [Gas chromatographic
determination of phenol in air.] Gig i Sanit, 5: 42-44 (in Russian).
Dive O & Leclerc H (1977) Utilisation du protozoaire cilie colpidum
campylum pour la meure de la toxicité et de l'accumulation des
micropollutants: analyse critique et applications. Environ Pollut,
14: 169-186.
Doan HMcK, Keith L, & Shennan AT (1979) Phenol and neonatal
jaundice. Pediatrics, 64(3): 324-325.
Dobbins DC, Thornton-Manning JR, Jones DD, & Federle TW (1987)
Mineralization potential for phenol in subsurface soils. J Environ
Qual, 16(1): 54-58.
Docter HJ & Zielhuis RL (1967) Phenol excretion as a measure of
benzene exposure. Ann Occup Hyg, 10: 317-326.
Dosemeci M, Blair A, Stewart, PA, Chandeler J, & Trush MA (1991)
Mortality among industrial workers exposed to phenol. Epidemiology,
2, 188-193.
Dow Chemical Company (1976) References and literature review
pertaining to toxicological properties of phenol. Dow Chemical
Company, Toxicological Research Laboratory (unpublished manuscript).
Dowden BF & Benett HJ (1965) Toxicity of selected chemicals to
certain animals. J Water Pollut Control Fed, 37: 1308-1316.
Dreibelbis WG & Hawthorne AR (1985) Worker exposure potential at
coal conversion facilities. Oak Ridge, Tennessee, Oak Ridge National
Laboratory (NTIS/DE85006822).
Dreisbach RH (1983) Handbook of poisoning: Prevention, diagnosis &
treatment, 11th ed. Los Altos, California, Lange Medical
Publications, pp 401-405.
Dumpert K (1987) Embryotoxic effects of environmental chemicals:
tests with the South African clawed toad ( Xenopus laevis).
Ecotoxicol Environ Saf, 13(3): 324-338.
Dutka BJ & Kwann KK (1981) Comparison of three microbial toxicity
screening tests with the Microtox test. Bull Environ Contam Toxicol,
27: 753-757.
Dutka BJ, Nyholm N, & Peterson J (1983) Comparison of several
microbiological toxicity screening tests. Water Res, 17(10):
1363-1368.
Duverneuil G & Ravier E (1962) Toxicité suraiguë du phénol par voie
transcutanée. Arch Mal Prof, 23: 830-833.
Eastmond DA, Smith MT, Ruzo LG, & Ross D (1986) Metabolic activation
of phenol by human myeloperoxidase and horseradish peroxidase. Mol
Pharmacol, 30(6): 674-679.
Eastmond DA, French RG, Ross D, & Smith MT (1987a) Metabolic
activation of 1-naphthol and phenol by a simple
superoxide-generating system and human leukocytes. Chem-Biol
Interact, 63(1): 47-62.
Eastmond DA, Smith MT, & Irons RD (1987b) An interaction of benzene
metabolites reproduces the myelotoxicity observed with benzene
exposure. Toxicol Appl Pharmacol, 91: 85-95.
Edwards VT, Jones BC, & Hudson DH (1986) A comparison of the
metabolic fate of phenol, phenyl glucoside and phenyl
6-o-malonyl-glucoside in the rat. Xenobiotica, 16: 801-807.
Ehrlich GG, Goelitz DF, & Godsy EM (1982) Degradation of phenolic
contaminants in ground water by anaerobic bacteria: St. Louis Park,
MN. Ground Water, 20: 703-710.
EIFAC (European Inland Fisheries Advisory Commission) (1972) Water
quality criteria for European freshwater fish. Report on monohydric
phenols and inland fisheries. Rome, Food and Agriculture
Organization of the United Nations (Technical Paper No. 15).
Epler JL, Rao TK, & Guerin MR (1979) Evaluation of feasibility of
mutagenic listing of shale oil products and effluents. Environ
Health Perspect, 30: 179-184.
Erben R (1982) The effect of phenol and its accumulation on snails
Physa fontinalis and Radix peregra mull (Pulmonata). Period
Biol, 84(4): 403-410.
Erben R, Munjko J, & Jovanovic V (1983) Influence of phenol
accumulation on the species Polycelis felina daly (Turbellaria).
Biologica (Bratislava), 38: 283-288.
Erexson GL, Wilmer JL, & Kligerman AD (1985) Sister chromatid
exchange induction in human lymphocytes exposed to benzene and its
metabolites in vitro. Cancer Res, 45(6): 2471-2477.
Ernst MR, Klesmer R, Huebner RA, & Martin JE (1961) Susceptibility
of cats to phenol. J Am Vet Med Assoc, 138: 197-199.
Ersek RA (1991) Comparative study of dermabrasion, phenol peel, and
acetic acid peel. Anesth Plast Surg, 15: 241-243.
Evans SJ (1952) Acute phenol poisoning. Br J Ind Med, 9: 227-229.
Farquharson ME, Gage JC, & Northover J (1958) The biological action
of chlorophenols. Br J Pharmacol, 13: 20-24.
Federle TW (1988) Mineralization of monosubstituted aromatic
compounds in unsaturated and saturated subsurface soils. Can J
Microbiol, 34(9): 1037-1042.
Fedorak PM & Hrudey SE (1986) Nutrient requirements for the
methanogenic degradation of phenol and p-cresol in anaerobic draw
and feed cultures. Water Res, 20(7): 929-934.
Fedorak PM, Roberts DJ, & Hrudey SE (1986) The effects of cyanide on
the methanogenic degradation of phenolic compounds. Water Res,
20(10): 1315-1320.
Feldman RJ & Maibach HI (1970) Absorption of some organic compounds
through the skin in man. J Invest Dermatol, 54: 399-404.
Fielding M, Gibson TM, James HA, et al (1981) Organic
micropollutants in drinking water. Medmenham, UK, Water Research
Centre (Report No. TR-159).
Fishbeck WA, Langner RR, & Kociba RJ (1975) Elevated urinary
phenol levels not related to benzene exposure. Am Ind Hyg Assoc J,
36(11): 820-824.
Flerov BA (1979) Physiological mechanisms of the action of toxic
substances and adaptation of aquatic animals to them. Hydrobiol J,
13(4): 70-74.
Flickinger CW (1976) The benzenediols: catechol, resorcinol and
hydroquinone - a review of the industrial toxicology and current
industrial exposure limits. Am Ind Hyg Assoc J, 37: 596-606.
Flynn GL & Yalkowsky SH (1972) Correlation and prediction of mass
transport across membranes. I. Influence of alkyl chain length on
flux-determining properties of barrier and diffusant. J Pharm Sci,
61: 838-852.
Fogels A & Sprague JB (1977) Comparative short-term tolerance of
zebrafish, flagfish, and rainbow trout to five poisons including
potential reference toxicants. Water Res, 11(9): 811-817.
Forrest F & Ramage DTO (1987) Cardiac dysrhythmia after subtrigonal
phenol. Anaesthesia, 42(7): 777-778.
Foxall PJD, Bending MR, Gartland KPR, & Nicholson JK (1991) Acute
renal failure following accidental cutaneous absorption of phenol.
In: Bach PH ed. Nephrotoxicity: Mechanisms, early diagnosis and
therapeutic management (Fourth International Symposium on
Nephrotoxicity, Guildford, UK, 1989). New York, Basel, Marcel
Dekker, Inc., pp 55-59.
Franz TJ (1975) Percutaneous absorption. On the relevance of in
vitro data. J Invest Dermatol, 64: 190-195.
Freitag D, Ballhorn L, Geyer H, & Korte F (1985) Environmental
hazard profile of organic chemicals. An experimental method for the
assessment of the behaviour of organic chemicals in the ecosphere by
means of simple laboratory tests with 14C-labelled chemicals.
Chemosphere, 14(10): 1589-1616.
French MR, Bababunmi EA, Golding RR, Bassir O, Caldwell J, Smith RL,
& Williams RT (1974) The conjugation of phenol, benzoic acid,
1-naphthylacetic acid and sulphadimethoxine in the lion, civet and
genet. FEBS Lett, 46(1): 134-137.
Fujii M (1978) Environmental monitoring of chemicals - Environmental
survey report of FY 1977. Tokyo, Environment Agency Japan,
Department of Environmental Health.
Gad-El Karim MM, Ramanujam VMS, Ahmed AE, & Legator MS (1965)
Benzene mycloclastogenicity: A function of its metabolism. Am J Ind
Med, 7: 475-484.
Gad-El Karim MM, Ramanujam VMS, & Legator MS (1966) Correlation
between the induction of micronuclei in bone marrow by benzene
exposure and the excretion of metabolites in urine of CD-1 mice.
Toxicol Appl Pharmacol, 85: 464-477.
Garberg P, Akerblom EL, & Bolcsfoldi G (1988) Evaluation of a
genotoxicity test measuring DNA-strand breaks in mouse lymphoma
cells by alkaline unwinding and hydroxyapatite elution. Mutat Res,
203: 155-176.
Garton GA & Williams RT (1949) Studies in detoxification. 26. The
fate of phenol, phenyl sulfuric acids and phenylglucuronide in the
rabbit in relation to the metabolism of benzene. Biochem J, 45:
158-163.
Gbodi TA & Oehme FW (1978) The fate of phenol, o-phenylphenol and
diisophenol in rats. Toxicol Appl Pharmacol, 45: 219-362.
Gersich FM, Blanchard FA, Applegath SL, & Park CN (1986) The
precision of daphnid ( Daphnia magna Straus, 1820) static acute
toxicity tests. Arch Environ Contam Toxicol, 15: 741-749.
Gibb C, Glover V, & Sandler M (1987) In vitro inhibition of phenol
sulphotransferase by food and drink constituents. Biochem Pharmacol,
36(14): 2325-2330.
Gibson II (1987) Phenol block in the treatment of spasticity.
Gerontology, 33(5): 327-330.
Gilbert P, Rondelet J, Poncelet F, & Mercier M (1980) Mutagenicity
of p-nitrosophenol. Food Cosmet Toxicol, 18: 523-525.
Gilli G, Palin L, & Scursatore E (1980) [Exposition to phenol.] Arch
Sci Med, 137: 41-44 (in Italian).
Gluth G & Hanke W (1983) The effect of temperature on physiological
changes in Carp Cyprinus carpio L. induced by phenol. Ecotoxicol
Environ Saf, 7(4): 373-389.
Gocke E, King MT, Eckhardt K, & Wild D (1981) Mutagenicity of
cosmetics, ingredients licensed by the European Communities. Mutat
Res, 90: 91-109.
Görge G, Beyer J, & Urich K (1987) Excretion and metabolism of
phenol, 4-nitrophenol and 2-methylphenol by the frogs Rana
temporaria and Xenopus laevis. Xenobiotica, 17(11): 1293-1298.
Green DWJ, Williams KA, & Pascoe D (1985) Studies on the acute
toxicity of pollutants to freshwater macroinvertebrates: 2. Phenol.
Arch Hydrobiol, 103(1): 75-82.
Green DWJ, Williams KA, Hughes DRL, Shaik GAR, & Pascoe D (1988)
Toxicity of phenol to Asellus aquaticus (L.): effects of
temperature and episodic exposure. Water Res, 22(2): 225-232.
Greenlee WF, Gross EA, & Irons RW (1981) Relationship between
benzene toxicity and the disposition of 14C-labelled benzene
metabolites in the rat. Chem-Biol Interact, 33: 285-299.
Groenen PJ (1978) [Components of tobacco smoke. Nature and quantity;
potential influence on health.] Zeist, The Netherlands, CIVO-TNO
Institute (Report No. R/5787) (in Dutch).
Gspan P, Jersic A, & Cadez E (1984) [Relationship between phenol
concentration in urine and work environment.] Staub-Reinhalt Luft,
44: 314-316 (in German).
Gunter BJ (1987) Health hazard evaluation report: CF & I Steel,
Pueblo, Colorado. Cincinnati, Ohio, National Institute for
Occupational Safety and Health (HETA 86-034-1712; NTIS/PB87-117016).
Gupta S & Dalela RC (1987) Kidney damage in Notopterus notopterus
(Pallas) following exposure to phenolic compounds. J Environ Biol,
8(2): 167-172.
Gupta PK & Rao PS (1982) Toxicity of phenol, pentachlorophenol and
sodium pentachlorophenolate to a freshwater pulmonate snail Lymnaea
acuminata (Lamarck). Arch Hydrobiol, 94: 210-217.
Gupta PK, Mujumdar VS, Rao PS, & Durve VS (1982a) Toxicity of
phenol, pentachlorophenol and sodium pentachlorophenolate to a
freshwater teleost Lebistes reticulatus (Peters). Acta Hydrochim
Hydrobiol, 10(2): 177-181.
Gupta S, Verma Sr, & Saxena PK (1982b) Toxicity of phenolic
compounds in relation to the size of a freshwater fish, Notopterus
notopterus (Pallas). Ecotoxicol Environ Saf, 6: 433-438.
Gupta S, Dalela RC, & Saxena PK (1983a) Influence of dissolved
oxygen levels on acute toxicity of phenolic compounds to a
freshwater teleost, Notopterus notopterus (Pallas). W.A.s. Pol.,
19(3): 223-228.
Gupta S, Dalela RC, & Saxena PK (1983b) Influence of temperature on
the toxicity of phenol and its chloro and nitro-derivates to the
fish Notopterus notopterus (Pallas). Acta Hydrochim Hydrobiol,
11(2): 187-192.
Gurujeyalashmi S & Oriel P (1989) Isolation of phenol-degrading
Bacillus stearothermophilus and partial characterization of the
phenol hydroxylase. Appl Environ Microbiol, 55(2): 500-502.
Haddad LM, Dimend KA, & Schweistris JE (1979) Phenol poisoning.
JACEP, 8(7): 267-269.
Hadorn E & Niggli N (1946) Mutations in Drosophila after chemical
treatment of gonads in vitro. Nature (Lond), 157: 162-163.
Hagemann R, Virelizier H, & Gaudin D (1978) Analyse de composés
organiques dans une atmosphère urbaine. Analusis, 6(9): 401-405.
Hardy JT, Dauble DD, & Felice LJ (1985) Aquatic fate of synfuel
residuals: bioaccumulation of aniline and phenol by the freshwater
phytoplankton Scenedesmus quadricauda. Environ Toxicol Chem, 4(1):
29-35.
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(Suppl): 3-142.
Healy JB Jr & Young LY (1979) Anaerobic biodegradation of eleven
aromatic compounds to methane. Appl Environ Microbiol, 28: 84-89.
Heikkilä P, Hämeilä M, Pyy L, & Raunu P (1987) Exposure to creosote
in the impregnation and handling of wood. Scand J Work Environ
Health, 13: 431-437.
Heller VG & Pursell L (1938) Phenol-contaminated waters and their
physiological action. J Pharmacol Exp Ther, 63: 99-107.
Henschler D ed. (1975) [Phenol.] In: [Analytical methods for the
testing of working media harmful to health. Volume 1: Air analyses.]
Weinheim, Verlag Chemie (in German).
Herbert DWM (1962) The toxicity to rainbow trout of spent still
liquors from the distillation of coal. Ann Appl Biol, 50: 755-777.
Hermens J (1984) Joint effects of a mixture of 14 chemicals on
mortality and inhibition of reproduction of Daphnia magna.
(Thesis).
Hickman GT & Novak JT (1989) Relationship between subsurface
biodegradation rates and microbial density. Environ Sci Technol,
23(5): 524-532.
Hinkel GK & Kintzel HW (1968) Phenol poisoning of a newborn through
skin resorption. Dtsch Gesundh, 23: 2420-2422.
Hoffmann KH & Vogt U (1988) [Degradation of phenol by yeasts in the
presence of n-hexadecane under growth conditions in a stirred
reactor.] Zent.bl Mikrobiol, 143(2): 87-91 (in German, with English
abstract).
Hogg SI, Curtis CG, Upshall DG, & Powell GM (1981) Conjugation of
phenol by rat lung. Biochem Pharmacol, 30(12): 1551-1555.
Holcombe GW, Phipps GL, & Fiandt JT (1982) Effects of phenol,
2,4-dimethylphenol, 2,4-dichlorophenol and pentachlorophenol on
embryo, larval and early-juvenile fathead minnows ( Pimephales
promelas). Arch Environ Contam Toxicol, 11: 73-78.
Holcombe GW, Phipps GL, Sulaiman AH, & Hoffman AD (1987)
Simultaneous multiple species testing: acute toxicity of 13
chemicals to 12 diverse freshwater amphibian, fish, and invertebrate
families. Arch Environ Contam Toxicol, 16(6): 697-710.
Hoshika Y & Muto G (1979) Sensitive gas chromatographic
determination of phenols as bromophenols using electron capture
detection. J Chromatogr, 179: 105-111.
Hoshika Y & Muto G (1980) Gas-liquid-solid chromatographic
determination of phenols in air using Tenax-GC and alkaline
precolumns. J Chromatogr, 187: 277-284.
Hoshino M & Akimoto H (1978) Photochemical oxidation of benzene,
toluene and ethylbenzene initiated by OH radicals in the gas phase.
Bull Chem Soc Jpn, 51: 718.
Houston JB & Cassidy MK (1982) Sites of phenol conjugation in the
rat in sulfate metabolism and sulfate conjugation. In: Proceedings
of an international workshop held at Noordwijkerhout, The
Netherlands, 20-23 September 1981. Taylor & Francis, pp 270-278.
Howard PH (1989) Handbook of environmental fate and exposure data
for organic chemicals. Chelsea, Michigan, Lewis Publishers, vol 1,
pp 468-476.
Hsieh GC, Sharma RP, Parker RDR, & Coulombe RA Jr (1992)
Immunological and neurobiochemical alterations induced by repeated
oral exposure of phenol in mice. Eur J Pharmacol - Environ Toxicol
Pharmacol Sect, 228: 107-114.
Hubble BR, Stetter JR, Gebert E, Harkness JBL, & Flotard RD (1981)
Experimental measurements from residential wood-burning stoves.
Proceedings of the International Conference on Residential Solid
Fuels: Environmental Impacts and Solutions, John A. Cooper & Dorothy
Malek.
Huq AS, Ho NFH, Husari N, Flynn GL, Jetzer WE, & Condie L (1986)
Permeation of water contaminative phenols through hairless mouse
skin. Arch Environ Contam Toxicol, 15(5): 557-566.
Hwang HM, Hodson RE, & Lee RF (1986) Degradation of phenol and
chlorophenols by sunlight and microbes in estuarine water. Environ
Sci Technol, 20(10): 1002-1007.
Hwang HM, Hodson RE, & Lewis DL (1989) Assessing interactions of
organic compounds during biodegradation of complex waste mixtures by
naturally occurring bacterial assemblages. Environ Toxicol Chem,
8(3): 209-214.
IARC (1989) Phenol. In: Some organic solvents, resin monomers and
related compounds, pigments and occupational exposures in paint
manufacture and painting. Lyon, International Agency for Research on
Cancer, pp 263-287 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 47).
Ikeda M & Ohtsuji H (1969) Hippuric acid, phenol and trichloroacetic
acid levels in the urine of Japanese subjects with no known exposure
to organic solvents. Br J Ind Med, 26: 162-164.
Illing HPA & House ESA (1980) Binding of (14C) phenol to rat liver
high-speed supernatant. Biochem Soc Trans, 8(1): 117-118.
Isaacson PJ & Frink GR (1984) Nonreversible sorption of phenolic
compounds by sediment fractions: the role of sediment organic
matter. Environ Sci Technol, 18: 43-48.
Itoh M (1982) Sensitisation potency of some phenolic compounds. J
Dermatol, 9(3): 223-283.
Ivett JL, Brown BM, Rodgers C, Anderson BE, Resnick MA, & Zeiger E
(1989) Chromosomal aberrations and sister chromatid exchange tests
in Chinese hamster ovary cells in vitro. IV. Results with 15
chemicals. Environ Mol Mutagen, 14: 165-187.
Jansson T, Curvall M, Hedin A, & Enzell CR (1986) In vitro studies
of biological effects of cigarette smoke condensate. II. Induction
of sister chromatid exchanges in human lymphocytes by weakly acidic,
semivolatile constituents. Mutat Res, 169: 129-139.
Jarvis SN, Straube RC, Williams ALJ, & Bartlett CLR (1985) Illness
associated with contamination of drinking water supplies with
phenol. Br Med J, 290: 1800-1802.
Jelinek R, Peterka M, & Rychter Z (1985) Chick embryotoxicity
screening test - 130 substances tested. Indian J Exp Biol, 23:
588-595.
Jergil B, Schelin C, & Tunek A (1982) Covalent binding of
metabolically activated hydrocarbons to specific microsomal
proteins. Adv Exp Med Biol, 136A: 341-348.
Jetzer WE, Huq AS, Ho NFH, Flynn GL, Duraiswamy N, & Condie L (1986)
Permeation of mouse skin and silicone rubber membranes by phenols:
relationship to in vitro partitioning. J Pharm Sci, 75(11):
1098-1103.
Jetzer WE, Hou SY, Huq AS, Duraiswamy N, Ho NFH, & Flynn GL (1988)
Temperature dependency of skin permeation of waterborne organic
compounds. Pharm Acta Helv, 63(7): 197-201.
Jones-Price C, Kimmel CA, Ledoux TA, Reel JR, Fisher PW,
Langhoff-Paschke L, & Marr MC (1983a) Final study report -
Teratologic evaluation of phenol (CAS No. 108-95-2) in CD rats (NTP
Study No. TER-81-104). Springfield, Virginia, National Technical
Information Service (NTIS/PB83-247726).
Jones-Price C, Kimmel CA, Ledoux TA, Reel JR, Langhoff-Paschke L, &
Marr MC (1983b) Final study report - Teratologic evaluation of
phenol (CAS No. 108-95-2) in CD-1 rats (NTP Study No. TER-80-129).
Springfield, Virginia, National Technical Information Service (NTIS/
PB85-104461.
Judis J (1982) Binding of selected phenol derivatives to human serum
proteins. J Pharm Sci, 71(110): 1145-1147.
Jünke I & Lüdemann D (1978) [Results of the testing of 200 chemical
compounds for acute toxicity for fish by the golden orfe test.] Z
Wasser Abwasser Forsch, 11(5): 161-164 (in German).
Kao J, Bridges JW, & Faulkner JK (1979) Metabolism of 14C phenol
by sheep, pig and rat. Xenobiotica, 9(3): 141-147.
Kasokat T, Nagel R, & Urich K (1987) The metabolism of phenol and
substituted phenols in zebra fish. Xenobiotica, 17(10): 1215-1222.
Katz M ed. (1977) Tentative method of analysis for determination of
phenolic compounds in the atmosphere with the 4-amino antipyrine
method - Methods of air sampling and analysis, 2nd ed. Washington,
DC, American Public Health Association, pp 324-332.
Kauppinen TP, Partanen TJ, Nurminen MM, Nickels JI, Hernberg, SG,
Hakulinen TR, Pukkala EI, & Savonen ET (1986) Respiratory cancers
and chemical exposures in the wood industry: a nested case-control
study. Br J Ind Med, 43(2): 84-90.
Kavlock RJ (1990) Structure-activity relationships in the
developmental toxicity of substituted phenols: in vivo effects.
Teratology, 41: 43-59.
Keen R & Baillod CR (1985) Toxicity to Daphnia of the end products
of wet oxidation of phenol and substituted phenols. Water Res,
19(6): 767-772.
Key PB & Scott GI (1986) Lethal and sublethal effects of chlorine,
phenol, and chlorine-phenol mixtures on the mud crab Panopeus
herbstii. Environ Health Perspect, 69: 307-312.
Khalili AA & Betts HB (1967) Peripheral nerve block with phenol in
the management of spasticity. J Am Med Assoc, 200: 1155.
Kifune I (1979) [Determination of micro amounts of phenol and
cresols in the atmosphere by using a filter impregnated with a
solvent of alkali and glycerol.] Bunseki Kagaku, 28(11): 638-642 (in
Japanese).
Kirk RE & Othmer DF (1980) Encyclopedia of chemical toxicology, 3rd
ed. New York, John Wiley and Sons, vol 17, pp 373-379.
Kligman AM (1966) The identification of contact allergens by human
assays. III. The maximization test: A procedure for screening and
rating contact sensitizers. J Invest Dermatol, 47(5): 393-409.
Knapik Z, Hanczyc H, Lubczynska-Kowalska W, Menzel-Lipinska M, Cader
J, Paradowski L, & Borowka Z (1980) Use of subclinical forms of the
toxic action of phenol. Z Gesamte Hyg Grenzgeb, 26(8): 585-587.
Knezovich JP, Hirabayashi JM, Bishop DJ, & Harrison FL (1988) The
influence of different soil types on the fate of phenol and its
biodegradation products. Chemosphere, 17(11): 2199-2206.
Knoevenagel K & Himmelreich R (1976) Degradation of compounds
containing carbon atoms by photooxidation in the presence of water.
Arch Environ Contam Toxicol, 4: 324-333.
Knoll G & Winter J (1987) Anaerobic degradation of phenol in sewage
sludge: benzoate formation from phenol and carbon dioxide in the
presence of hydrogen. Appl Microbiol Biotechnol, 25(4): 384-391.
Kobayashi K & Akitake K (1975) Metabolism of chlorophenols in fish.
VIV Absorption, and excretion of phenol by goldfish. Bull Jpn Soc
Sci Fish, 41: 1271-1276.
Kobayashi K, Akitake H, & Kimura S (1976) Studies on the metabolism
of chlorophenols in fish. VI Turnover of absorbed phenol in
goldfish. Bull Jpn Soc Sci Fish, 42: 45-50.
Kobayashi K, Oshima Y, Hamada S, & Taguchi C (1987) Induction of
phenol-sulfate conjugating activity by exposure to phenols and
duration of its induced activity in short-necked clam. Bull Jpn Soc
Sci Fish, 53(11): 2073-2076.
Koike N, Haga S, Ubukata N, Sakurai M, Shimizu H, & Sato A (1988)
[Mutagenicity of benzene metabolites by fluctuation test.] Jpn. J.
Ind. Health 30(6): 475-480 (in Japanese, with English abstract).
Koop DR, Laethem CL, & Schnier GG (1989) Identification of
ethanol-inducible P450 isozyme 3a (P450IIE1) as a benzene and phenol
hydroxylase. Toxicol Appl Pharmacol, 98: 278-288.
Kordylewska A (1980) Developmental disturbances of Limnaea
stagnalis due to phenol intoxication. Acta Biol (Cracow) Ser Zool,
22(1): 105-130.
Koster HJ, Halsema J, Scholtens E, Knippers M, & Mulder GJ (1981)
Dose-dependent shifts in the sulfation and glucuronidation of
phenolic compounds in the rat in vivo and in isolated hepatocytes.
The role of saturation of phenolsulfotransferase. Biochem Pharmacol,
30(18): 2569-2575.
Koster HJ (1982) Sulfuration and glucuronidation in the rat in vivo
and in vitro. The balance between two enzyme systems competing for
a mutual substrate. University of Groningen (Thesis).
Kostoveckii YA & Zholdakova Z (1971) [On hygienic norm-setting in
waterbodies.] Gig i Sanit, 7: 7-10 (in Russian).
Krug M, Ziegler H, & Straube G (1985) Degradation of phenolic
compounds by the yeast Candida tropicalis HP-15: 1. Physiology and
growth and substrate utilization. J Basic Microbiol, 25(2): 103-110.
Kumar V & Mukerjee D (1988) Phenol and sulfide induced changes in
the ovary and liver of sexually maturing common carp, Cyprinus
carpio. Aquat Toxicol, 13(1): 53-60.
Kuwata K, Uebori M, & Yamazaki Y (1980) Determination of phenol in
polluted air as p-nitrobenzene azophenol derivative by reversed
phase high performance liquid chromatography. Anal Chem, 52(6):
857-860.
Kwasniewska K & Kaiser KLE (1983) Toxicities of selected phenols to
fermentative and oxidative yeasts. Bull Environ Contam Toxicol,
31(2): 188-194.
Laszczynska M, Barcew B, Wyczarska-Srak K, & Wozniak D (1983)
Reversibility of histoenzymatic changes caused by phenol in the
mouse kidney and liver. Folia Biol (Cracow), 31(1): 65-73.
Layiwola PJ & Linnecar DFC (1981) The biotransformation of 14C
phenol in some freshwater fish. Xenobiotica, 11(3): 167-171.
Leichnitz K ed. (1982) [Dräger test tubes phenol 5/a.] In: [Test
tube pocket book; testing of air and technical analysis of gas using
Dräger test tubes], 5th ed. Lubeck, Drägerwerk AG, p 114 (in
German).
Leider M & Moser HS (1961) Toxicology of topical dermatology
preparations. Arch Dermatol, 83: 928-929.
Leuenberger C, Ligocki MP, & Pankow JF (1985) Trace organic
compounds in rain: 4. Identities, concentrations, and scavenging
mechanisms for phenols in urban air and rain. Environ Sci Technol,
19(11): 1053-1058.
Lewin JF & Cleary WT (1982) An accidental death caused by the
absorption of phenol through skin. A case report. Forensic Sci Int,
19(2): 177-179.
Lewis MA (1983) Effect of loading density on the acute toxicities of
surfactants, copper and phenol to Daphnia magna Straus. Arch
Environ Contam Toxicol, 12: 51-55.
Liao JTF (1980) Investigations of phenol tissue distribution, plasma
protein binding, and liver subcellular fraction affinity. Diss Abstr
Int, B41(5): 1736.
Liao JTF & Oehme FW (1980) Literature reviews of phenolic compounds.
I. Phenol. Vet Hum Toxicol, 22: 160-164.
Liao JTF & Oehme FW (1981a) Tissues distribution and plasma protein
binding of (14C) phenol in rats. Toxicol Appl Pharmacol, 57(2):
220-225.
Liao JTF & Oehme FW (1981b) Plasma protein binding of phenol in dogs
and rats as determined by equilibrium dialysis and ultrafiltration.
Toxicol Appl Pharmacol., 57(2): 226-230.
Lister J (1867; republished 1937) On a new method of treating
compound fracture, abcess, etc., with observations on the condition
of suppuration. Med Classics, 2: 28-71.
Luten JB, Ritskes JM, & Weseman JM (1979) Determination of phenol,
guaiacol and 4-methylguaiacol in wood smoke and smoked fish-products
by gas-liquid chromatography. Z Lebensm.unters Forsch, 168: 289-292.
McFarlane JC, Pfleeger T, & Fletcher J (1987) Transpiration effect
on the uptake and distribution of bromacil, nitrobenzene and phenol
in soybean plants. J Environ Qual, 16(4): 372-376.
McGregor DB, Brown A, Cattanach P, Edwards I, McBride D, Riach C, &
Caspary WJ (1988) Responses of the L5178Y tk+/tk- mouse lymphoma
cell forward mutation assay: III. 72 Coded chemicals. Environ Mol
Mutagen, 12: 85-154.
Malcolm AR, Mills LJ, & Trosko JE (1985) Effects of ethanol, phenol,
formaldehyde, and selected metabolites on metabolic cooperation
between Chinese hamster V79 lung fibroblasts. Carcinogenesis: A
compr survey, 8: 305-318.
Mayes ME, Alexander HC, & Dill DC (1982) A study to assess the
influence of age on the response of Fathead minnows in static acute
toxicity tests. Bull Environ Contam Toxicol, 31(2) 139-147.
Merliss RR (1972) Phenol marasmus. J Occup Med, 14: 55-56.
Miksch K & Schürmann B (1988) [Toxicity assessment of zinc sulphate,
copper sulphate and phenol by different methods.] Z Wasser Abwasser
Forsch, 21: 193-198 (in German with English abstract).
Miller DL & Ogilvie DM (1975) Temperature selection in brook trout
( Salvelinus fontinalis) following exposure to DDT, PCB or phenol.
Bull Environ Contam Toxicol, 14: 545-551.
Minor JK & Becker BA (1971) A comparison of the teratogenic
properties of sodium salycate, sodium benzoate and phenol. Abstracts
of the 10th Annual Meeting of the Society of Toxicology No. 40.
Toxicol Appl Pharmacol, 19: 373.
Mitchell JR (1972) Mechanism of benzene-induced aplastic anaemia.
Fed Proc, 30: 561.
Model A (1889) Poisoning with concentrated phenol in a child
suffering from diphtheria. Ther Mon.heft, 3: 483-485.
Mogilnicka EM & Piotrowski JK (1974) [The exposure test for phenol
in the light of field study.] Med Prac, 25: 137-141 (in Hungarian).
Morimoto K & Wolff S (1980) Increase of sister chromatid exchanges
and perturbations of cell division kinetics in human lymphocytes by
benzene metabolites. Cancer Res, 40: 1189-1193.
Morimoto K, Wolff S, & Koizumi A (1983) Induction of sister
chromatid exchanges in human lymphocytes by microsomal activation of
benzene metabolites. Mutat Res, 119(3-4): 355-360.
Morrison JE, Matthews D, Washington R, Fennessey PV, & Harrison M
(1991) Phenol motor point blocks in children: Plasma concentrations
and cardiac dysrhythmias. Anesthetiology, 75: 359-362.
Mukhitov B (1964) The effect of low phenol concentrations on the
organism of man or animals and their hygienic evaluation. In: Levine
BS ed. USSR literature on air pollution and related occupational
diseases. Springfield, Virginia, US Department of Commerce, National
Technical Information Service, pp 185-199 (NTIS/PB 64-11574).
Murphy JC, Osterberg RE, Seabaugh UM, & Bierbower GW (1982) Ocular
irritancy responses to various pHs of acids and bases with and
without irrigation. Toxicology, 23(4): 281-291.
Müting D, Keller HE, & Kraus W (1970) Quantitative colorimetric
determination of free phenols in serum and urine of healthy adults
using modified diaro-reactions. Clin Chem Acta, 27: 177-180.
Nagel R (1983) Species differences, influence of dose and
application on biotransformation of phenol in fish. Xenobiotica,
13(2): 101-106.
Nagel R & Urich K (1983) Quinol sulfate, a new conjugate of phenol
in goldfish. Xenobiotica, 13(2): 97-100.
Nagel R, Adler HJ, & Rao TK (1982) Induction of filamentation by
mutagens and carcinogens in a lon mutant of Escherichia coli.
Mutat Res, 105: 309-312.
Namkoong W, Loehr RC, & Malina JF Jr (1989) Effects of mixture and
acclimation on removal of phenolic compounds in soil. J Water Pollut
Control Fed, 61(2): 242-250.
Narotsky MG & Kavlock RJ (1993) A multidisciplinary approach to
toxicological screening. II. Development toxicity. Research Triangle
Park, North Carolina, US Environmental Protection Agency, Health
Effects Research Laboratory (Report No. MS 91-237).
Nathan PW (1959) Intrathecal phenol to relieve spasticity in
paraplegia. Lancet, 2: 1099.
NCI (1980) Bioassay of phenol for possible carcinogenicity.
Bethesda, Maryland, US Department of Health Services, National
Cancer Institute (Technical Report Series No. NCI-CG-TR-203).
Neuhauser EF, Durking PR, Malecki MR, & Anatra M (1986) Comparative
toxicity of ten organic chemicals to four earthworm species. Comp
Biochem Physiol, C83: 197-200.
Nicola RM, Branchflower R, & Pierce D (1987) Chemical contaminants
in bottonfish. J Environ Health, 49: 342-347.
Niessen R, Lenoir D, & Boule P (1988) Phototransformation of phenol
induced by excitation of nitrate ions. Chemosphere, 17(19):
1977-1984.
NIOSH (1984) NIOSH manual of analytical methods, 3rd ed. Cincinnati,
Ohio, National Institute for Occupational Safety and Health.
NIOSH (1985a) NIOSH pocket guide to chemical hazards. Cincinnati,
Ohio, National Institute for Occupational Safety and Health.
NIOSH (1985b) Method 8305. NIOSH manual of analytical methods, 3rd
ed. Cincinnati, Ohio, Institute for Occupational Safety and Health.
Oehme FW (1969) A comparative study of the biotransformation and
excretion of phenol. A doctoral dissertation. University of
Missouri.
Oehme FW & Davis LE (1970) The comparative toxicity and
biotransformation of phenol. Toxicol Appl Pharmacol, 17: 283.
Ogata M, Yamasaki Y, & Kawai T (1986) Significance of urinary phenyl
sulfate and phenyl glucuronide as indices of exposure to phenol. Int
Arch Occup Environ Health, 58: 197-202.
Oglesbi LA, Ebron-McCoy MT, Logson TR, Copeland F, Beyer PE, &
Kavlock RJ (1992) in vitro embryotoxicity of a series of
para-substituted phenols: structure, activity, and correlation with
in vivo data. Teratology, 45: 11-33.
Ohtsuji H & Ikeda M (1972) Quantitative relationship between
atmospheric phenol vapour and phenol in the urine of employees in
Bakelite factories. Br J Ind Med, 29: 70-73.
Oksama M & Kristofferson R (1979) The toxicity of phenol to
Phoxinus phoxinus, Gammarus ducbeni and Mesidota entomon in
brackish water. Ann Zool Fenn, 16(3): 209-216.
Olowska L, Oledzka-Slotwinska H, Lazczymska M, & Wozniak W (1980)
Effect of phenol on hydrolytic and respiratory enzymes of the small
intestinal mucosa of white mouse. Folia Morphol (Warsaw), 39(2):
149-158.
Oomens AC & Schuurhuis FG (1983) A method for the collection and
determination of phenol and bisphenol A in air. Int Arch Occup
Environ Health, 52(1): 43-48.
Oris JT, Winner RW, & Moore MV (1991) A four-day survival and
reproduction toxicity test for Ceriodaphnia dubia. Environ Toxicol
Chem, 10: 217-224.
Painter RB & Howard R (1982) The Hela DNA-synthesis inhibition test
as a rapid screen for mutagenic carcinogens. Mutat Res, 92(1-2):
427-437.
Paradowski M, Anderzeiewski S, Zauisza B, & Wybrak-Wrobel T (1981)
Excretion of phenol and p-cresol in urine as an element of
evaluation of professional exposure to benzene and phenol in
employees of refining and petrochemicals works in Plock. Przeglad
Lekarski, 38(9): 663-666.
Paris DF, Wolfe NL, & Steen WC (1982) Structure-activity
relationships in microbial transformation of phenols. Appl Environ
Microbiol, 44: 153-158.
Parke DV & Williams RT (1953) Studies in detoxification. 54. The
metabolism of benzene: (a) The formation of phenylglucuronide and
phenylsulfuric acid from 14C benzene. (b) The metabolism of 14C
phenol. Biochem J, 55: 337-340.
Parker JG (1984) The effects of selected chemicals and water quality
on the marine polychaete Ophryotrocha diadema. Water Res, 18(7):
865-868.
Pashin YV & Bakhitova LM (1982) Mutagenicity of benzo(a)pyrene and
the antioxidant phenol at the HGPRT locus of V79 Chinese Hamster
cells. Mutat Res, 104(6): 389-393.
Patrick E, Maibach HI, & Burkhalter A (1985) Mechanisms of
chemically induced skin irritation. I. Studies of time course, dose
response, and components of inflammation in the laboratory mouse.
Toxicol Appl Pharmacol, 81: 476-490.
Pekari K, Vainotalo S, Heikkila P, Palotie A, Luotamo M, & Riihimaki
V (1992) Biological monitoring of occupational exposure to low
levels of benzene. Scand J Work Environ Health, 18: 317-322.
Pellack-Walker P & Blumer JL (1986) DNA damage in L5178YS cells
following exposure to benzene metabolites. Mol Pharmacol, 30: 42-47.
Pellack-Walker P, Walker JK, Evans HH, & Blumer JL (1985)
Relationship between the oxidation potential of benzene metabolites
and their inhibitory effect on DNA synthesis in L5178YS cells. Mol
Pharmacol, 28: 560-566.
Petrov VI (1960) [Cases of phenol poisoning during coke slaking with
phenol water.] Gig i Sanit, 25: 60-62 (in Russian).
Phipps GL, Holcombe GW, & Fiandt JT (1981) Acute toxicity of phenol
and substituted phenols to the fathead minnow. Bull Environ Contam
Toxicol, 26: 585-593.
Pickering GH & Henderson C (1966) Acute toxicity of some important
petrochemicals to fish. J Water Pollut Control Fed, 38: 1419-1429.
Pierce WM & Nerland DE (1988) Qualitative and quantitative analyses
of phenol, phenylglucuronide, and phenylsulfate in urine and plasma
by gas chromatography mass spectrometry. J Anal Toxicol, 12:
344-347.
Piotrowski JK (1971) Evaluation of exposure to phenol, absorption of
phenol vapour in the lungs and through the skin and excretion of
phenol in urine. Br J Ind Med, 28: 172-178.
Podolak GE, McKenzie RM, Rinehart DS, & Mazur JF (1981) A rapid
technique for collection and analysis of phenol vapours. Am Ind Hyg
Assoc J, 42(10) 734-738.
Poirier MC, De Cicco BT, & Lieiberman MW (1975) Non-specific
inhibition of DNA repair synthesis by tumour promotors on human
diploid fibroblasts damaged with N-acetoxy-acetyl-aminofluorene.
Cancer Res, 35: 1392-1397.
Pool BL & Lin PZ (1982) Mutagenicity testing in the Salmonella
typhimurium assay of phenolic compounds and phenolic fractions
obtained from smoke-house condensates. Food Chem Toxicol, 22:
383-391.
Potthast K (1976) Determination of phenols in smoked meat products.
Advances in Smoking of Foods, Intern. Joint IUPAC/IUFoSt Symp.,
Warschau, pp. 39-44.
Potthast K (1982) Dark smoking at high smokehouse temperatures.
Fleischwirtschaft, 62: 1578-1582.
Powell GM, Miller JJ, Olavesen AH, & Curtis CG (1974) Liver as a
major organ of phenol detoxification? Nature (Lond), 252: 234-235.
Pratt RM & Willis WD (1985) in vitro screening assay for
teratogens using growth inhibition of human embryonic cells. Proc
Natl Acad Sci (USA), 82: 5791-5794.
Price KS, Waggy GT, & Conway RA (1974) Brine shrimp bioassay and
seawater BOD of petrochemicals. J Water Pollut Control Fed, 46:
63-76.
Pullin TG, Pinkerton MN, Johnson RV, & Kilian DJ (1978)
Decontamination of the skin of swine following phenol exposure: A
comparison of the relative efficacy of water versus polyethylene
glycol/industrial methylated spirits. Toxicol Appl Pharmacol, 43:
199-206.
Quebbemann AJ & Anders MW (1973) Renal tubular conjugation and
excretion of phenol and p-nitrophenol in the chicken: differing
mechanism of renal transfer. J Pharmacol Exp Ther, 184(3): 695-708.
Ramanathan M (1984) Water pollution. In: Mark HF, Othmer DF,
Overberger CG, Seaborg GT, & Grayson M ed. Kirk-Othmer encyclopedia
of chemical technology, 3rd ed. New York, Chichester Brisbane,
Toronto, John Wiley & Sons, vol 24, p 299.
Ramli JB & Wheldrake JF (1981) Phenol conjugation in the desert
hopping mouse, Notomys alexis. Comp Biochem Physiol, C69(2):
379-381.
Rao RSV & Nath KJ (1983) Biological effect of some poisons on
Cantocamptus (Crustacea). Int J Environ Stud, 21(3-4): 271-275.
Rao, PS, Durve US, Khangarot BS, & Srekhawat SS (1983) Acute
toxicity of phenol, pentachlorophenol and sodium pentachlorophenate
to a freshwater ostracod Cypris subglubosa. Acta Hydrochim
Hydrobiol, 11(4): 457-465.
Rapson WH, Nazar MA, & Butsky V (1980) Mutagenicity produced by
aqueous chlorination of organic compounds. Bull Environ Contam
Toxicol, 24: 590-596.
Ravichandran S & Anantharaj B (1984) Effect of phenol on the
phosphomonoesterases and ATPase activity in the fish Sarotherodon
mossambicus (Peters) in saline waters. Proc Indian Acad Sci (Anim
Sci), 93(6): 557-563.
Razani H, Nanba K, & Murachi S (1986a) Acute toxic effect of phenol
on zebra fish Brachydanio rerio. Bull Jpn Soc Sci Fish, 52(9):
1547-1552.
Razani H, Nanba K, & Murachi S (1986b) Chronic toxic effect of
phenol on zebra fish Brachydanio rerio. Bull Jpn Soc Sci Fish,
52(9): 1553-1558.
Rea WJ, Pan Y, Laseter JL, Johnson AR, & Fenyves EJ (1987) Toxic
volatile organic hydrocarbons in chemically sensitive patients. Clin
Ecol, 5(2): 70-74.
Reid GG, Ketterer PJ, Mawkinney H, & Glover P (1982) Exposure to
phenol and endrin as a cause of skin ulcerations and nervous signs
in pigs. Aust Vet J, 59: 160.
Reynolds JH, Middlebrooks EJ, Porcella WB, & Grenney WJ (1978)
Comparison of semi-continuous and continuous-flow bioassay. Progress
in Water Technology, 9(4): 897-909.
RIVM (1986) [Criteria Document: Phenol]. Bilthoven, The Netherlands,
National Institute of Public Health and Environmental Protection
(Document No. 738513002) (in Dutch; toxicology also available in
English).
Roberts MS, Anderson RA, & Swarlrich J (1977) Permeability of human
epidermis to phenolic compounds. J Pharm Pharmacol, 29(11): 677-683.
Roberts MS, Anderson RA, Swarlrich J, & Moore DE (1978) The
percutaneous absorption of phenolic compounds: the mechanism of
diffusion across the stratum corneum. J Pharm Pharmacol, 30:
486-490.
Rogers SCF, Burrows D, & Neill D (1978) Percutaneous absorption of
phenol and methyl alcohol in magenta paint B.C.P. Br J Dermatol, 98:
559-560.
Rozich AF & Colvin RJ (1986) Effects of glucose on phenol
biodegradation by heterogenous populations. Biotechnol Bioeng,
29(7): 965-971.
Rubin HE & Alexander M (1983) Effect of nutrients on the rates of
mineralization of trace concentrations of phenol and
p-nitrophenol. Environ Sci Technol, 17: 104-107.
Ruedemann R & Deichmann WB (1953) Blood phenol level after topical
application of phenol containing preparations. J Am Med Assoc, 152:
506-509.
Ruessink RG & Smith LL (1975) The relationship of the 96 h LC50 of
the lethal threshold concentration of hexavalent chromium, phenol
and sodium pentachlorophenate for fathead minnows ( Pimephales
promelas Rafinesque). Trans Am Fish Soc, 3: 570.
Rusch HP, Bosch MS, & Boutwell RK (1955) The influence of irritants
on mitotic activity and tumour formation in mouse epidermus.
International Union against Cancer Newsletter, 11: 699-703.
Rushmore TH, Snyder R, & Kalf G (1984) Covalent binding of benzene
and its metabolites to DNA in rabbit bone marrow mitochondria in
vitro. Chem-Biol Interact, 49: 133-154.
Russell JW (1975) Analysis of air pollutants using sampling tubes
and gas chromatography. Environ Sci Technol, 9(13): 1175-1178.
Ryser S & Ulmer G (1980) Etude de la pollution gazeuse par les
résines synthétiques utilisées en fonderie. Fonderie, 35: 313-324.
Salamon MH & Glendenning OM (1957) Tumour promotion in mouse skin by
sclerosing agents. Br J Cancer, 11: 434-444.
Sandage C (1961) Tolerance criteria for continuous inhalation
exposure to toxic material. I. Effects on animals of 90-day exposure
to phenol, CCl4, and a mixture of indole, skatole, H2S, and methyl
mercaptan. Dayton, Ohio, Wright-Patterson Air Force Base, US Air
Force Systems Command, Aeronautical Systems Division (ASD Technical
Report 61-519 (I); NTIS AD-268783).
Sanitary Epidemiological Station, Katowice (1991) Air pollution in
the Katowice voivodeship in 1988-1990. Katowice, Poland, Regional
Sanitary Epidemiological Station.
Sawahata T & Neal RA (1983) Biotransformation of phenol to
hydroquinone and catechol by rat liver microsomes. Mol Pharmacol,
23(21): 453-460.
Schaumburg HH, Byck R, & Weller RO (1970) The effect of phenol on
peripheral nerve. A histological and electrophysiological study. J
Neuropathol Exp Neurol, 29: 615-630.
Schlicht MP, Moser VC, Sumrell BM, Berman E, & MacPhail RC (1992)
Systemic and neurotoxic effects of acute and repeated phenol
administration (Abstract No. 1047). Toxicologist, 12, 274.
Schmidt R & Maibach H (1981) Immediate and delayed onset "skip area"
dermatitis presumed secondary to topical phenol exposure. Contact
Dermatitis, 7(4): 199-202.
Schmieder PK & Henry TR (1988) Plasma binding of 1-butanol, phenol,
nitrobenzene and pentachlorophenol in the rainbow trout and rat: a
comparative study. Comp Biochem Physiol, C91(2): 413-418.
Schoenberg JB & Mitchell CA (1975) Airway disease caused by phenolic
(phenol-formaldehyde) resin exposure. Arch Environ Health, 30:
574-577.
Schussler OF & Stern MA (1911) Gangrene of finger caused by five
percent phenol ointment. J Am Med Assoc, 57: 628.
Schütz A & Wolfe D (1980) [Gases and vapours in foundries;
measurement; evaluation; control.] Giesserei, 67: 68-73 (in German).
Seidel HJ, Barthel E, Schäfer F, Schad H, & Weber L (1991) Action of
benzene metabolites on murine hematopoietic colony-forming cells in
vitro. Toxicol Appl Pharmacol, 111, 128-131.
Selvakumar A & Hsieh HN (1988) Competitive adsorption of organic
compounds by microbial biomass. J Environ Sci Health, A23(8):
729-744.
Schlicht MP, Moser VP, Sumrell BM, Berman E, & MacPhail RC (1992)
Systematic and neurotoxic effects of acute and repeated phenol
administration. Toxicologist, 12(1): 274.
Scott HD, Wolf DC, & Lavy TL (1982) Apparent absorption and
microbial degradation of phenol by soil. J Environ Qual, 11:
107-112.
Shafer WE & Schönherr J (1985) Accumulation and transport of phenol,
2-nitrophenol, and 4-nitrophenol in plant cuticles. Ecotoxicol
Environ Saf, 10(2): 239-252.
Shimp RJ & Pfaender FK (1987) Effect of adaptation to phenol on
biodegradation of monosubstituted phenols by aquatic microbial
communities. Appl Environ Microbiol, 53(7): 1496-1499.
Shimp RJ & Young RL (1987) Availability of organic chemicals for
biodegradation in settled bottom sediments. Ecotoxicol Environ Saf,
15(1): 31-45.
Shirkey RJ, Kao J, Fry JR, & Bridges JW (1979) A comparison of
xenobiotic metabolism in cells isolated from rat liver and small
intestinal mucosa. Biochem Pharmacol, 28: 1461-1466.
Skare JA & Schrotel KR (1984) Alkaline elution of rat testicular
DNA: Detection of DNA strand breaks after in vivo treatment with
chemical mutagens. Mutat Res, 130: 283-294.
Skvortsova NN & Vysochina IV (1976) Changes in biochemical and
physiological indices in animals produced by the combined effect of
benz[a]pyrene and phenol. Environ Health Perspect, 13: 101-106.
Slabbert JL (1986) Improved bacterial growth test for rapid water
toxicity screening. Bull Environ Contam Toxicol, 37: 565-569.
Smart AC & Zannoni VC (1984) D.T. Diaphorase and peroxidase
influence the covalent binding of the metabolites of phenol, the
major metabolite of benzene. Mol Pharmacol, 26: 105-111.
Smith MC (1984) Histological findings following intrathecal
injections of phenol solutions for relief of pain. Br J Anaesth, 36:
387-406.
Solbé JFLG, Cooper VA, Willis CA, & Mallett MJ (1985) Effects of
pollutants in fresh waters on European non-salmoid fish. I:
Non-metals. J Fish Biol, 27(Suppl A): 197-207.
Solliman T (1957) A manual of pharmacology and its applications to
therapeutics and toxicology, 8th ed. New York, Philadelphia, W.B.
Saunders Company, pp 805-809.
Steele RH & Wilhelm DL (1966) The inflammatory reaction in chemical
injury. I. Increased vascular permeability and erythema induced by
various chemicals. Br J Exp Pathol, 47: 612-623.
Southworth GR, Herbes SE, Franco PJ, & Giddings JM (1985)
Persistence of phenols in aquatic microcosms receiving chronic
inputs of coal-derived oil. Water Air Soil Pollut, 24(3): 283-296.
Spicer CW, Riggin RM, Holdren MW, DeRoos FL, & Lee RN (1985)
Atmospheric reaction products from hazardous air pollutant
degradation. Research Triangle Park, North Carolina, US
Environmental Protection Agency (EPA-600/3-85-028; NTIS
PB85-185841/XAB).
Spoelstra SF (1978) Degradation of tyrosine in anaerobically stored
piggery wastes and in pig feces. Appl Environ Microbiol, 36:
631-638.
Stajduhar-Caric Z (1968) Acute phenol poisoning. J Forensic Med, 15:
41-42.
Stephenson RR (1983) Effects of water hardness, water temperature
and size of the test organism on the susceptibility of the
freshwater shrimp Gammarus pulex (L.) to toxicants. Bull Environ
Contam Toxicol, 31(4): 459-466.
Sturtevant MJ (1952) Studies on the mutagenicity of phenol. Journal
of Heredity, 43: 217-219.
SRI (1982) Economics handbook. Stanford, Stanford Research
Institute.
Subrahmanyam VV & O'Brien PJ (1985) Peroxidase-catalyzed binding of
uniformly carbon-14-labeled phenol to DNA. Xenobiotica, 15(10):
859-872.
Subrahmanyam V, Sadler A, Suba E, & Ross D (1989) Stimulation of in
vitro bioactivation of hydroquinone by phenol in bone marrow
cells. Drug Metab Dispos, 17(3): 348-349.
Subrahmanyam VV, Kolachana P, & Smith MT (1991) Hydroxylation of
phenol to hydroquinone catalyzed by a human
myeloperoxidase-superoxide complex: possible implications in
benzene-induced myelotoxicity. Free Radic Res Commun, 15, 285-296.
Suzuki T & Kisara K (1985) Enhancement of phenol-induced tremor
caused by central monoamine depletion. Pharmacol Biochem Behav, 22:
153-155.
Takemori AE & Glowacki GA (1962) Studies on glucuronide synthesis in
rats chronically treated with morphine and phenol. Biochem
Pharmacol, 11: 867-870.
Tetra Tech. Inc. (1986) Development of sediment quality values for
Pudget Sound (unnamed report prepared for Pudget Sound Dredged
Disposal Analysis and Pudget Sound Estuary Program). Bellevue,
Washington, Tetra Tech. Inc.
Thompson ED & Gibson DP (1984) A method for determining the maximum
tolerated dose for acute in vivo cytogenetic studies. Food Chem
Toxicol, 22(8): 665-676.
Thornton-Manning JR, Jones DD, & Federle TW (1987) Effects of
experimental manipulation of environmental factors on phenol
mineralization in soil. Environ Toxicol Chem, 6: 615-621.
Tibbles BJ & Baecker AAW (1989) Effects and fate of phenol in
simulated landfill sites. Microb Ecol, 17(2): 201-206.
TNO (1978) [Emissions during various processes in foundries.] Delft,
The Netherlands, Dutch Organization for Applied Scientific Research
(TNO) (Report No. M78/40/07536/Jul/SCI) (in Dutch).
Tonapi GT & Varghese G (1987) Cardio-physiological responses of some
selected cladocerans to three common pollutants. Arch Hydrobiol,
110(1): 59-65.
Toth L (1982) [Transfer of phenols from tobacco smoke into meat
products.] Fleischwirtschaft, 62: 1389-1402 (in German).
Tranvik L, Larsson P, Okla L, & Regnell O (1991) In situ
mineralization of chlorinated phenols by pelagic bacteria in lakes
of different humic content. Environ Toxicol Chem, 10: 195-200.
Truppman ES & Ellenby JD (1979) Major electrocardiograph changes
during chemical face peeling. Plast Reconstr Surg, 63: 44-48.
Tschech A & Fuchs G (1987) Anaerobic degradation of phenol by pure
cultures of newly isolated denitrifying pseudomonads. Arch
Microbiol, 148(3): 213-217.
Tunek A, Olofsson J, & Berlin M (1981) Toxic effects of benzene and
benzene metabolites on granulopoietic stem cells and bone marrow
cellularity in mice. Toxicol Appl Pharmacol, 59(1): 149-156.
Turtle WRM & Dolan T (1922) A case of rapid and fatal absorption of
carbolic acid through the skin. Lancet, 2: 1273-1274.
UBA (1981) [Air pollution control '81. Developments, state of the
art, tendencies.] Berlin, Federal Office for the Environment (in
German).
United Nations (1980) Yearbook or industrial statistics. Volume II:
Commodity product data. New York, United Nations.
Ursin C (1985) Degradation of organic chemicals at trace levels in
seawater and marine sediment. The effects of concentration of the
initial fractional turnover rate. Chemosphere, 14(10): 1539-1550.
US EPA (1980) Phenol. Ambient water quality criteria. Washington,
DC, US Environmental Protection Agency (EPA 440/5-80-066).
US EPA (1986a) Method 8010. Phenols. In: Test methods for evaluating
solid waste -Physical/chemical methods, 3rd ed. Washington, DC, US
Environmental Protection Agency, Office of Solid Waste and Emergency
Response, pp 8040/1-8050/17 (EPA Report No. SW-846).
US EPA (1986b) Method 8250. Gas chromatography/mass spectrometry for
semi-volatile organics: Packed column technique. In: Test methods
for evaluating solid waste -Physical/chemical methods, 3rd ed.
Washington, DC, US Environmental Protection Agency, Office of Solid
Waste and Emergency Response, pp 8250/1-8250/32 (EPA Report No.
SW-846).
US EPA (1986c) Method 8270. Gas chromatography/mass spectrometry for
semi-volatile organics: Capillary column technique. In: Test methods
for evaluating solid waste -Physical/chemical methods, 3rd ed.
Washington, DC, US Environmental Protection Agency, Office of Solid
Waste and Emergency Response, pp 8270/1-8270/34 (EPA Report No.
SW-846).
US EPA (1992) Phenol - Drinking water health advisory. Washington,
DC, US Environmental Protection Agency, Office of Water.
Van Duuren BL & Goldschmidt BM (1976) Carcinogenic and
tumour-promoting agents in tobacco carcinogenesis. J Natl Cancer
Inst, 56: 1237-1242.
Van Duuren BL, Sivah A, Langretti J, & Goldschmidt BM (1968)
Initiators and promotors in tobacco carcinogenesis. Natl Cancer Inst
Monogr, 28: 173-180.
Van Duuren BL, Blazej T, Goldschmidt BM, Katz C, Melchionne S, &
Sivah A (1971) Carcinogenesis studies on mouse skin and inhibition
of tumour induction. J Natl Cancer Inst, 46: 1039-1044.
Van Duuren BL, Katz C, & Goldschmidt BM (1973) Cocarcinogenesis
agents in tobacco carcinogenesis. J Natl Cancer Inst, 51: 703-705.
Van Gemert LJ (1984) Compilation of odour threshold values in air:
Supplement V. Zeist, The Netherlands, CIVO-TNO (Report No. 84.220).
Van Gemert LJ & Van Nettenbreijer (1977) Compilation of odour
threshold values in air and water. Zeist, The Netherlands, CIVO/RID.
Veith GD & Kosian P (1983) Estimating bioconcentration potential
from octanol/water partition coefficients. In: Physical behaviour of
PCB's in the Great Lakes. Ann. Arbor, Michigan, Ann Arbor Science
Publishers, Inc., pp 269-282.
Verma SR, Rani S, Tyagi AK, & Dalela RC (1980) Evaluation of acute
toxicity of phenol and its chloro- and nitro-derivates to certain
teleosts. Water Pollut, 14: 95-102.
Vernot EH, MacEwen JD, Haun CC, & Kinkead ER (1977) Acute toxicity
and skin corrosion data for some organic and inorganic compounds and
aqueous solutions. Toxicol Appl Pharmacol, 42: 417-423.
Veronesi B, Padilla S, & Newland D (1986) Biochemical and
neuropathological assessment of triphenyl phosphite in rats. Toxicol
Appl Pharmacol, 83: 203-210.
Verschueren K (1983) Handbook of environmental data on organic
chemicals, 2nd ed. New York, Van Nostrand Reinhold Company.
Volskay VT Jr & Grady CPL (1988) Toxicity of selected RCRA compounds
to activated sludge microorganisms. J Water Pollut Control Fed,.
60(10): 1850-1856.
Von Oettingen WF (1949) Phenol and its derivatives. The relation
between their chemical constitution and their effect on the
organisms. Washington, DC, National Institutes of Health Bulletin.
Von Oettingen WF & Sharples NE (1946) The toxicity and toxic
manifestation of 2,2 bis-(p-chlorophenyl)-1,1,1-trichloroethane
(DDT) as influenced by chemical changes in the molecule. J Pharmacol
Exp Ther, 88: 400-413.
Wajon JE, Rosenblatt DH, & Burrows EP (1982) Oxidation of phenol and
hydroquinone by chlorine dioxide. Environ Sci Technol, 16: 396-402.
Wang WC (1986) Comparative toxicology of phenolic compounds using
root elongation method. Environ Toxicol Chem, 5(10): 891-896.
Wang YT, Suidan MT, Pfeiffer JT, & Najm I (1988) Effects of some
alkyl phenols on methanogenic degradation of phenol. Appl Environ
Microbiol, 54(5): 1277-1279.
Wangenheim J & Bolcsfoldi G (1988) Mouse lymphoma L5178Y thymidine
kinase locus assay of 50 compounds. Mutagenesis, 3(3): 193-205.
Warner MA & Harper JV (1985) Cardiac dysrhythmias associated with
chemical peeling with phenol. Anesthesiology, 62: 366-367.
Weast RC, ed. (1987) Handbook of chemistry and physics, 68th ed.
Boca Raton, Florida, CRC Press, Inc., p C-406.
Weitering JG, Krijgsheld KR, & Mulder GJ (1979) The availability of
inorganic sulfate as a rate limiting factor in the sulfate
conjugation or xenobiotics in the rat? Biochem Pharmacol, 28:
757-762.
Wheldrake JF, Baudinette RV, & Hewitt S (1978) The metabolism of
phenol in a desert rodent Notomys alexis. Comp Biochem Physiol,
C61: 103-107.
Wiggins BA & Alexander M (1988) Role of chemical concentration and
second carbon sources in acclimation of microbial communities for
biodegradation. Appl Environ Microbiol, 54(11): 2803-2807.
Wilcosky TC & Tyroler HA (1983) Mortality from heart disease among
workers exposed to solvents. J Occup Med, 25: 879-885.
Wilcosky TC, Checkoway H, Marshall EG, & Tyroler HA (1984) Cancer
mortality and solvent exposures in the rubber industry. Am Ind Hyg
Assoc J, 45: 809-811.
Wild D, Eckhardt K, Gocke E, & King MT (1980) Comparative results on
short-term in vitro and in vivo mutagenicity tests obtained with
selected environmental chemicals. Berlin, Heidelberg, New York,
Springer Verlag, pp 170-178.
Williams RT (1938) Studies in detoxification. I. The influence of
(a) dose and (b) o, m, p, substitution on the sulfate detoxification
of phenol in the rabbit. Biochem J, 32: 878-887.
Williams RT (1959) Detoxication mechanisms, 2nd ed. New York,
Chichester, Brisbane, Toronto, John Wiley and Sons, pp 237-295.
Windus-Podehl G, Lyftog C, Zieve L, & Brunner G (1983)
Encephalopathic effect in rats. J Lab Clin Med, 101(4): 586-592.
Winkler HD (1981) [Methods for reduction of emission of air
pollutants in production of chipboards.] Gesundheits-Ing, 102:
295-299 (in German).
Wood KA (1978) The use of phenol as a neurolytic agent: a review.
Pain, 5: 205-225.
Woodruff RC, Mason JM, Valencia R, & Zimmering S (1985) Chemical
mutagenesis testing in Drosophila. V. Results of 53 coded compounds
tested for the National Toxicology Program. Environ Mutagen, 7:
677-702.
Wynder EL & Hoffmann D (1961) A study of tobacco carcinogenesis -
the role of the acid fractions as promoters. Cancer, 14: 1306-1315.
Wysowski DK, Flynt JW, Goldfield M, Altman R, & Davis AT (1978)
Epidemic neonatal hyperbilirubinemia and use of a phenolic
disinfectant detergent. Pediatrics, 61(2): 165-170.
Young LY & Rivera MD (1985) Methanogenic degradation of 4 phenolic
compounds. Water Res, 19(10): 1325-1332.
Zavorovskaya NA & Nekhorosheva EV (1981) [Spectrometric
determination of phenol and aniline in air and solutions simulating
biological media.] Zh Anal Khim, 36(9): 1808-1812 (in Russian).
RESUME
1. Identité, propriétés physiques et chimiques et méthodes
d'analyse
Le phénol se présente sous la forme d'un solide cristallin
blanc qui fond à 43 °C et se liquéfie par contact avec l'eau. Il
dégage une odeur âcre caractéristique et possède une saveur forte et
piquante. Il est soluble dans la plupart des solvants organiques; sa
solubilité dans l'eau est limitée à la température ambiante;
au-dessus de 68 °C il est entièrement soluble dans l'eau. Le phénol
est modérément volatil à la température ambiante. C'est un acide
faible, et, sous sa forme ionisée, il est très sensible aux
réactions de substitution électrophile et à l'oxydation.
On peut recueillir le phénol dans des échantillons prélevés
dans l'environnement par absorption dans une solution de soude ou
par adsorption sur un solide. La désorption s'effectue par
acidification, entraînement à la vapeur et extraction à l'éther (à
partir des solutions) ou par voie thermique ou en phase liquide
(lorsqu'il est adsorbé sur un solide). Les méthodes d'analyse les
plus importantes sont la chromatographie en phase gazeuse avec
détection par ionisation de flamme ou capture d'électrons ou encore
la chromatographie liquide à haute performance avec détection en
lumière ultra-violette. La limite de détection la plus basse qui ait
été signalée dans l'air est de 0,1 µg par m3. On peut doser le
phénol dans le sang et les urines; dans les échantillons d'urine, on
a fait état d'une limite de détection de 0.5 µg/litre.
2. Sources d'exposition humaine et environnementale
Le phénol est un constituant du goudron de houille et il se
forme au cours de la décomposition naturelle des substances
organiques. La majeure partie du phénol présent dans l'environnement
provient cependant de l'activité humaine. Les sources potentielles
en sont la production et l'utilisation, tel quel ou sous forme de
dérivés, en particulier de résines phénoliques et de caprolactame,
les gaz d'échappement, la combustion de bois de construction et la
fumée de cigarette. Une autre source potentielle est constituée par
la dégradation atmosphérique du benzène sous l'action de la lumière,
la présence de phénol dans le purin pouvant également fortement
contribuer à sa concentration dans l'atmosphère. Les dérivés du
benzène et du phénol peuvent, par conversion in vivo, constituer
une source d'exposition humaine endogène.
La production de phénol dans le monde s'est montrée
relativement constante pendant les années 1980, les Etats-Unis
d'Amérique en étant le premier producteur. Il est principalement
utilisé pour la fabrication des résines phénoliques, du bisphénol A
et de la caprolactame. On en connaît également un certain nombre
d'applications médicales et pharmaceutiques.
3. Transport, distribution et transformation dans l'environnement
Les principales émissions de phénol se produisent dans l'air.
La majeure partie du phénol présent dans l'atmosphère finit par être
dégradée par voie photochimique en dihydroxybenzènes, nitrophénols
et dérivés résultant de l'ouverture du cycle, avec une demi-vie
estimative de 4 à 5 heures. Une petite quantité est éliminée de
l'air par dépôt humide (pluie). Le phénol devrait présenter une
forte mobilité dans le sol mais son transport et sa réactivité
peuvent être affectés par le pH.
Le phénol présent dans l'eau et le sol peut être décomposé par
des réactions abiotiques ainsi que par l'activité microbienne en un
certain nombre de produits, dont les plus importants sont le dioxyde
de carbone et le méthane. La part des réactions biologiques dans la
décomposition globale du phénol dépend de nombreux facteurs tels que
la concentration, l'acclimatation, la température et la présence
d'autres composés.
4. Concentrations dans l'environnement et exposition humaine
On ne possède aucune donnée sur la concentration atmosphérique
du phénol. La concentration de fond est vraisemblablement inférieure
à 1 ng/m3. Les valeurs en milieu urbain et suburbain varient de
0,1 à 8 µg/m3 alors que dans les zones où prédominent les sources
de phénol (zones industrielles) les concentrations signalées peuvent
être jusqu'à 100 fois plus élevées. On a décelé du phénol dans l'eau
de pluie, les eaux superficielles et les eaux souterraines mais les
données sont très rares. On a fait état de concentrations élevées de
phénol dans des sédiments et des eaux souterraines par suite de
pollution industrielle.
Il peut y avoir exposition professionnelle au phénol lors de la
production de ce produit et de ses dérivés, lors de l'enduction avec
des résines phénoliques (industrie du bois et métallurgie) et lors
d'un certain nombre d'autres activités industrielles. La
concentration la plus élevée qui ait été signalée (jusqu'à 88
mg/m3) concernait des ouvriers de l'ex-URSS employés à
l'extinction du coke avec des eaux usées contenant du phénol. La
plupart des autres concentrations évoquées ne dépassaient 19
mg/m3.
En ce qui concerne la population dans son ensemble, c'est la
fumée de cigarette et les aliments fumés qui constituent les plus
importantes sources d'exposition au phénol, si l'on excepte
l'exposition par voie atmosphérique. L'exposition par l'eau de
boisson ou la consommation par inadvertance de produits alimentaires
contaminés devraient rester faibles; le phénol a en effet une odeur
et une saveur désagréables, ce qui dans la plupart des cas devrait
alarmer le consommateur.
5. Cinétique et métabolisme
Le phénol est facilement absorbé par toutes les voies
d'exposition. Après absorption, il se répartit rapidement dans
l'ensemble des tissus.
Une fois résorbé, il forme essentiellement des conjugués avec
l'acide glucuronique et l'acide sulfurique, et, dans une moindre
mesure, des hydroxylates avec le catéchol et l'hydroquinone. Il y a
également conjugaison avec les phosphates. La formation de
métabolites réactifs (4,4-biphénol et diphénoquinone) a été mise en
évidence lors d'études in vitro portant sur des neutrophiles et
des leucocytes humains activés.
La proportion relative de glucuronides et de sulfo-conjugués
varie avec la dose et l'espèce animale. Chez le rat, on a observé
qu'en augmentant la dose de phénol, la formation de sulfo-conjugués
l'emportait sur celle de glucuro-conjugués.
C'est essentiellement dans le foie, les poumons et au niveau de
la muqueuse gastro-intestinale que le phénol est métabolisé. Le rôle
relatif joué par ces divers tissus dépend de la voie
d'administration et de la dose.
Des études in vivo et in vitro ont montré que le phénol se
fixait aux protéines tissulaires et plasmatiques par liaison
covalente. Certains métabolites du phénol se lient également aux
protéines.
C'est principalement par la voie urinaire que la phénol est
excrété chez l'animal et l'homme. Le taux d'excrétion urinaire varie
selon la dose, la voie d'administration et l'espèce. Une faible
proportion est excrétée dans les matières fécales et l'air expiré.
6. Effets sur les animaux d'expérience et les systèmes
d'épreuve in vitro
Le phénol présente une toxicité aiguë modérée pour les
mammifères. La DL50 par voie orale varie chez les rongeurs de 300
à 600 mg de phénol/kg de poids corporel. La DL50 dermique varie
respectivement de 670 à 1400 mg/kg de poids corporel chez le rat et
le lapin et pour le rat, la CL50 à 8h. par voie respiratoire est
supérieure à 900 mg de phénol/m3. Après exposition aiguë, les
symptômes cliniques sont une hyperexcitabilité neuromusculaire, des
convulsions graves, une nécrose de la peau et des muqueuses de la
gorge et l'on note également des effets au niveau des poumons, des
fibres nerveuses, des reins, du foie et de la pupille (réflexe
photomoteur).
Les solutions de phénol sont agressives pour la peau et les
yeux. A l'état de vapeur, le phénol peut irriter les voies
respiratoires. On est fondé à croire que le phénol n'agit pas comme
sensibilisateur cutané.
Les effets les plus importants relevés lors d'études à court
terme sur l'animal consistaient en neurotoxicité, lésions hépatiques
et rénales, troubles respiratoires et retard de croissance. A des
doses orales quotidiennes de 40 mg/kg ou davantage on a observé des
effets néphrotoxiques chez le rat. Chez la même espèce, il y avait
une évidente hépatotoxicité aux doses supérieures ou égales à 100
mg/kg/jour. Lors d'une étude limitée de 14 jours sur des rats, on a
obtenu, pour la dose par voie orale sans effets nocifs observables,
une valeur de 12 mg/kg/jour, le critère retenu étant les effets sur
le rein. Dans cette expérience, il y avait encore inhibition du
myosis (réaction de l'iris à un stimulus lumineux) à la dose
quotidienne de 4 mg/kg; toutefois, l'importance médicale de cette
observation demeure incertaine. On a signalé la présence de
certaines altérations biologiques au niveau de la muqueuse
intestinale et des reins chez des souris recevant des doses
quotidiennes inférieures à 1 mg/kg, observation dont l'importance
toxicologique n'est pas non plus bien claire.
Il n'y a pas eu d'études satisfaisantes sur la toxicité du
phénol pour la fonction de reproduction. Toutefois, la toxicité du
phénol paraît se manifester par son action délétère sur le
développement du rat et de la souris. Lors de deux études au cours
desquelles des rats ont reçu des doses multiples de phénol, on a
obtenu, pour la dose sans effets nocifs observables, une valeur de
40 mg/kg/jour (pour la dose la plus faible sans effets nocifs
observables, cette valeur était de 53 mg/kg/jour) et de 60
mg/kg/jour respectivement (dans ce cas, la dose la plus faible sans
effets nocifs observables était de 120 mg/kg/jour). Chez la souris,
la dose sans effets nocifs observables était de 140 mg/kg/jour (dose
minimale sans effets nocifs observables: 280 mg/kg/jour).
La plupart des tests de mutagénicité bactérienne ont donné des
résultats négatifs. Cependant, des épreuves effectuées in vitro
sur des cellules mammaliennes ont révélé la présence de mutations,
de lésions chromosomiques et d'effets sur l'ADN. Le phénol est sans
effet sur la communication intercellulaire (mesurée par la
coopération métabolique) dans des cultures de cellules mammaliennes.
Un certain nombre d'études ont mis en évidence l'induction de
micro-noyaux dans des cellules de moelle osseuse murine. Toutefois,
les études sur la souris n'ont pas révélé la présence de
micro-noyaux à doses plus faibles.
Deux études de cancérogénicité ont été effectuées sur des rats
et des souris mâles et femelles à qui l'on administrait du phénol
mêlé à leur eau de boisson. On n'a observé d'affections malignes (à
savoir cancers médullaires de la thyroïde, leucémies) que chez les
rats mâles soumis à de faibles doses. On n'a pas effectué d'études
de cancérogénicité en bonne et due forme utilisant la voie
percutanée ou la voie respiratoire. Des études de cancérogénicité en
deux phases ont montré que le phénol pouvait se comporter comme un
agent tumoro-promoteur lorsqu'on l'appliquait à plusieurs reprises
sur la peau de la souris.
7. Effets sur l'homme
Des cas bien documentés d'exposition humaine au phénol par la
voie percutanée, buccale ou intraveineuse, ont donné lieu à
l'observation d'effets indésirables très divers. Il a été fait état
d'une irritation des voies gastro-intestinales après ingestion de
phénol. Après exposition de la peau, les effets observés localement
vont d'un blémissement cutané indolore ou d'un érythème à la
corrosion et à la nécrose profonde. Parmi les effets généraux, on a
noté les troubles suivants: arythmies cardiaques, acidose
métabolique, hyperventilation, détresse respiratoire, insuffisance
rénale aiguë, lésions rénales, urines foncées, méthémoglobinémie,
troubles neurologiques (notamment des convulsions), choc
cardio-vasculaire, coma et mort. La dose orale la plus faible qui
ait entraîné un décès humain était de 4,8 g; la mort est survenue
dans les 10 minutes.
Le risque d'intoxication par inhalation de vapeurs de phénol
est connu depuis longtemps, mais on n'a pas signalé de décès
consécutif à ce type d'accident. Les symptômes produits par
l'inhalation de phénol consistent notamment en anorexie, perte de
poids, maux de tête, vertiges, salivation et coloration foncée des
urines.
Le phénol n'est pas un agent sensibilisateur.
Le seuil olfactif pour l'homme serait 0,021 à 20 mg/m3 d'air.
Pour le phénol en solution aqueuse, on a fait état d'un seuil
olfactif de 9 mg/litre, le seuil gustatif étant de 0,3 mg/litre
d'eau.
On ne dispose pas de données suffisantes sur le pouvoir
cancérogène du phénol.
8. Effets sur les êtres vivants dans leur milieu naturel
Lors d'études portant sur une seule espèce de bactéries, on a
obtenu, pour la CE50 relative à l'inhibition de la croissance, des
valeurs allant de 244 à 1600 mg de phénol/litre. On a constaté que
le seuil de toxicité se situait à 64 mg de phénol/litre. Pour les
protozoaires et les champignons, les valeurs étaient du même ordre
que pour les bactéries; pour les algues elles étaient un peu
inférieures. Le phénol est toxique pour les organismes dulçaquicoles
supérieurs. Pour les crustacés et les poissons, les valeurs les plus
faibles de la CL50 ou de CE50 se situent entre 3 et 7 mg de
phénol/litre. Les données concernant la toxicité aiguë du phénol
pour les organismes marins sont comparables à celles dont on dispose
au sujet des organismes d'eau douce. Des études à long terme sur des
crustacés et diverses espèces de poissons ont révélé des différences
de sensibilité remarquables; c'est ainsi que les valeurs de la CL1
provenant d'épreuves sur des embryons et des larves de Salmo et de
Carassius se sont révélées très inférieures (respectivement 0,2 et
2 µg de phénol/litre) aux valeurs correspondantes pour d'autres
espèces de poissons (concentration sans effet létal observable,
2,2-6,1 mg/litre) et d'amphibiens, ou tirées d'études sur la
reproduction des crustacés (concentration sans effet létal
observable, 10 mg de phénol/litre). On ne dispose pas de données sur
des épreuves à long terme qui auraient été pratiquées sur des
organismes marins.
En général le facteur de bioconcentration du phénol chez les
divers types d'organismes aquatiques est très bas (<1-10) encore
qu'on ait signalé parfois des valeurs plus élevées (jusqu'à 2200).
Il est donc vraisemblable que le phénol ne subit pas d'accumulation
biologique importante.
Les données dont on dispose au sujet de la destinée et des
effets du phénol chez les organismes terrestres sont très peu
nombreuses. La CE50 à 120 h. est de 120 à 170 mg/litre pour le
millet et lors d'une épreuve par contact, on a obtenu pour la CL50
chez le lombric, une valeur comprise entre 2,4 et 10,6 µg/cm3.
9. Résumé de l'évaluation
9.1 Santé humaine
La population dans son ensemble est essentiellement exposée au
phénol par la voie respiratoire. Par voie orale, il peut y avoir
exposition répétée par suite de la consommation d'eau de boisson
contaminée ou d'aliments fumés.
On ne dispose pas de données suffisantes pour déterminer
l'ampleur de l'exposition de la population générale, mais on peut
donner une limite estimative supérieure de l'absorption quotidienne.
En mettant les choses au pire, on peut considérer que l'exposition
maximale se produit chez un individu qui inhale en permanence de
l'air fortement contaminé et consomme souvent des aliments fumés ou
de l'eau de boisson qui contient du phénol à des concentrations
atteignant le seuil gustatif. On a calculé que la dose quotidienne
ingérée maximale totale estimative pour un individu de ce genre
pesant 70 kg était de 0,1 mg/kg de poids corporel.
Les valeurs de la dose sans effets nocifs observables obtenues
par expérimentation animale en prenant comme critères les troubles
rénaux et les effets sur le développement étaient, chez le rat, de
l'ordre de 12 à 40 mg/kg de poids corporel et par jour. En utilisant
un coefficient d'incertitude de 200, on peut recommander comme
limite supérieure de la dose journalière totale une valeur située
entre 60 et 200 µg/kg de poids corporel. En prenant pour l'homme une
dose quotidienne limite de 100 µg/kg de poids corporel, on peut
conclure que l'exposition de toutes origines au phénol de la
population dans son ensemble se situe en-dessous de ces valeurs.
On peut être préoccupé par le fait que, selon certaines
données, le phénol pourrait être génotoxique et que d'autre part, on
ne possède pas suffisamment de résultats pour écarter avec certitude
l'éventualité que le phénol soit cancérogène. L'évaluation de ce
composé doit être revue périodiquement.
9.2 Environnement
Le phénol ne subit probablement pas d'accumulation biologique
importante. Il est toxique pour les organismes aquatiques; en
appliquant la méthode modifiée de l'Agence de protection de
l'environnement des Etats-Unis, on peut considérer que la
concentration préoccupante de cette substance dans l'environnement
est de 0,02 µg/litre. On manque de données suffisantes sur son
action chez les plantes et les organismes terrestres.
Il peut y avoir transport du phénol d'un compartiment à l'autre
de l'environnement par dépôt humide ou par lessivage du sol. En
général, ce composé ne devrait pas persister dans l'environnement.
Les rares données dont on dispose sur l'exposition ne permettent pas
d'évaluer le risque qu'il présente pour les écosystèmes aquatiques
ou terrestres. Toutefois, en tenant compte de la valeur de sa
concentration préoccupante pour l'environnement aquatique, il est
raisonnable de considérer qu'en cas de contamination par le phénol
des eaux de surface ou des eaux marines, il y a un risque pour les
organismes aquatiques.
RESUMEN
1. Identidad, propiedades físicas y químicas, métodos analíticos
El fenol es un sólido cristalino, blanco, que funde a 43 oC y
se licúa al contacto con el agua. Posee un olor acre característico
y un sabor ardiente fuerte. Es soluble en la mayor parte de los
disolventes orgánicos. A temperatura ambiente, su solubilidad en
agua es limitada; por encima de 68 oC es completamente hidrosoluble.
El fenol es moderadamente volátil a temperatura ambiente. Es un
ácido débil y, en su forma ionizada, muy sensible a las reacciones
de sustitución electrofílica y a la oxidación.
El fenol se puede obtener a partir de muestras ambientales por
absorción en una solución de NaOH o en contacto con sorbentes
sólidos. La desorción se lleva a cabo por acidificación, destilación
al vapor y extracción con éter (a partir de soluciones) o mediante
desorción térmica o líquida (a partir de sorbentes sólidos). Las
técnicas analíticas más importantes son la cromatografía de gases en
combinación con la detección de ionización por conductor y de
captura de electrones, y la cromatografía en fase líquida, de alta
presión, en combinación con la detección por luz ultravioleta. En el
aire, el límite de detección más bajo que se haya notificado es de
0,1 µg/m3. Se puede determinar la presencia de fenol en la sangre
y la orina; en muestras de orina se ha registrado un límite de
detección de 0,5 µg/litro.
2. Fuentes de exposición humana y ambiental
El fenol es uno de los componentes del alquitrán de hulla y se
forma durante la descomposición natural de materiales orgánicos. No
obstante, la mayor parte del fenol presente en el medio ambiente es
de origen antropogénico. Algunas fuentes potenciales son la
producción y el uso de fenol y de sus productos, especialmente
plásticos fenólicos y caprolactama, los gases de escape, la quema de
leña y el humo de los cigarrillos. Otra fuente potencial es la
degradación atmosférica del benceno por la influencia de la luz, si
bien la presencia del fenol en los purines puede asimismo tener
considerable influencia en sus niveles atmosféricos. Los derivados
del benceno y del fenol pueden, mediante una conversión in vivo,
constituir una fuente de exposición humana endógena a fenol.
Según parece, la producción mundial de fenol fue bastante
regular a lo largo del decenio de 1980, en que los Estados Unidos
fueron el productor más importante. Se usa principalmente como
materia básica de las resinas fenólicas, del bisfenol A y de la
caprolactama. También se le conocen algunas aplicaciones médicas y
farmacéuticas.
3. Transporte, distribución y transformación en el medio ambiente
Las principales emisiones de fenol van al aire. La mayor parte
del fenol existente en la atmósfera se degradará mediante reacciones
fotoquímicas frente a los dihidroxibencenos, los nitrofenoles y los
productos de rotura del anillo, con una semivida, estimada en 4 a 5
hs. Una parte menor desaparecerá del aire por deposición hídrica
(lluvia). Se piensa que el fenol es móvil en el suelo, pero el pH
puede influir en el transporte y la reactividad.
El fenol presente en el agua y el suelo puede degradarse por
reacciones abióticas, así como por la actividad microbiana, dando
lugar a un número de compuestos, los más importantes de los cuales
son el dióxido de carbono y el metano. La proporción entre la
biodegradación y la degradación general del fenol está determinada
por múltiples factores, como la concentración, la aclimación, la
temperatura y la presencia de otros compuestos.
4. Niveles ambientales y exposición humana
No se dispone de datos sobre los niveles atmosféricos de fenol.
Se supone que los niveles básicos son inferiores a 1 ng/m3. Los
niveles urbanos y suburbanos oscilan entre 0,1 y 8 µg/m3, mientras
que se ha notificado que las concentraciones en las zonas próximas
al foco de emisión (industria) alcanzan magnitudes cien veces
superiores. Se ha detectado fenol en la lluvia y en las aguas
superficiales y subterráneas, pero los datos son muy escasos. En
sedimentos y aguas subterráneas se han notificado niveles elevados
de fenol debidos a la contaminación industrial.
La exposición profesional al fenol puede tener lugar durante la
producción del mismo y de sus derivados, la aplicación de resinas
fenólicas (industrias maderera y siderúrgica) y algunas otras
actividades industriales. La concentración más alta (hasta 88
mg/m3) se ha notificado en relación con trabajadores de la antigua
Unión Soviética que apagaban el coque con aguas residuales que
contenían fenol. La mayor parte de las restantes concentraciones
notificadas no rebasan los 19 mg/m3.
Para la población en general, el humo de cigarrillo y los
alimentos ahumados constituyen las fuentes más importantes de
exposición al fenol, aparte de la exposición a través del aire. La
exposición a través del agua potable y de los alimentos contaminados
por inadvertencia probablemente sea baja; el fenol tiene un olor y
un sabor desagradables, que en la mayor parte de los casos provocan
el rechazo del consumidor.
5. Cinética y metabolismo
El fenol se absorbe fácilmente por todas las vías de
exposición. Tras la absorción, la sustancia se distribuye
rápidamente a todos los tejidos.
El fenol absorbido se conjuga principalmente con el ácido
glucurónico y el ácido sulfúrico y, en menor medida, se hidroxila en
pirocatequina e hidroquinona. También se conjuga con los fosfatos.
La formación de metabolitos reactivos (4,4-bifenol y difenoquinona)
se ha demostrado en estudios in vitro con neutrófilos y leucocitos
humanos activados.
Las cantidades relativas de glucurono y sulfoconjugados varían
según la dosis y la especie animal. Tras aumentar la dosis de fenol,
se observó en las ratas un cambio de la sulfatación a la
glucuronidación.
El hígado, los pulmones y la mucosa gastrointestinal
constituyen los sitios más importantes del metabolismo fenólico. La
función relativa desempeñada por esos tejidos depende de la vía de
administración y de la dosis.
Estudios in vivo e in vitro han demostrado la unión
covalente del fenol con las proteínas tisulares y plasmáticas.
Algunos metabolitos fenólicos se unen asimismo a las proteínas.
La excreción por la orina es la principal vía de eliminación
del fenol en los animales y en los seres humanos. La tasa de
excreción urinaria varía en función de la dosis, de la vía de
administración y de la especie. Una parte menor se excreta a través
de las heces y del aire espirado.
6. Efectos en mamíferos de laboratorio y en sistemas de
prueba in vitro
El fenol tiene una toxicidad aguda moderada en los mamíferos.
En los roedores, los valores de la DL50 por vía oral oscilan entre
300 y 600 mg de fenol/kg de peso corporal. Los valores de la
DL50por vía cutánea para ratas y conejos oscilan entre 670 y 1400
mg/kg de peso corporal, respectivamente, y el valor de la CL50 por
inhalación a las 8 horas en las ratas es superior a los 900 mg de
fenol/m3. Los síntomas clínicos después de la exposición aguda son
hiperexcitabilidad neuromuscular y convulsiones graves, necrosis de
la piel y de las mucosas de la garganta y efectos en los pulmones,
fibras nerviosas, riñones, hígado y en la respuesta pupilar a la
luz.
Las soluciones de fenol son corrosivas para la piel y los ojos.
Los vapores de fenol pueden irritar las vías respiratorias. Existen
pruebas de que el fenol no produce sensibilización cutánea.
Los efectos más importantes notificados a partir de estudios de
corta duración en animales fueron neurotoxicidad, lesiones hepáticas
y renales, trastornos respiratorios y retraso del crecimiento. Se
han notificado efectos tóxicos en el riñón de las ratas con dosis
por vía oral de 40 mg/kg al día o más. La toxicidad en el hígado
resultó evidente en las ratas a las que se habían administrado al
menos 100 mg/kg diarios. En un estudio limitado de 14 días de
duración realizado en ratas se notificó un nivel sin efectos
adversos observados (NOAEL) de 12 mg/kg al día por vía oral, basado
en los efectos renales. En este experimento, la miosis (respuesta
del iris a la luz) se mantuvo inhibida con 4 mg/kg al día; sin
embargo, no está claro el significado médico de este hallazgo. Se
notificó la existencia de algunos cambios biológicos en la mucosa
intestinal y los riñones de ratones con dosis inferiores a 1 mg/kg
al día, dato de significado toxicológico incierto.
No hay estudios adecuados sobre la toxicidad reproductiva del
fenol. En estudios con ratas y ratones el fenol ha sido identificado
como tóxico del desarrollo. En dos estudios de dosis múltiples en
ratas, se han notificado NOAEL de 40 mg/kg al día (el más bajo nivel
sin efectos adversos observados (LOAEL) fue de 53 mg/kg al día) y de
60 mg/kg al día (el LOAEL fue de 120 mg/kg al día). En el ratón, el
NOAEL fue de 140 mg/kg al día (el LOAEL fue de 280 mg/kg al día).
La mayor parte de las pruebas de mutagenicidad bacteriana han
dado resultados negativos. En células in vitro de mamíferos se han
observado mutaciones, lesiones cromosómicas y efectos en el ADN. El
fenol no tiene efectos en la comunicación intercelular (medida como
cooperación metabólica) en cultivos de células de mamíferos. En
algunos estudios se ha observado la inducción de micronúcleos en
células de médula ósea de ratones. Con dosis más bajas no se
observaron micronúcleos en estudios con ratones.
Se han llevado a cabo dos estudios de carcinogenicidad con
ratas y ratones machos y hembras a los que se administró fenol con
el agua de beber. Sólo se observó malignidad (por ejemplo, carcinoma
de células C de la tiroides y leucemia) en ratas macho con dosis
bajas. No se han realizado estudios adecuados de carcinogenicidad
por vía dérmica o por inhalación. Estudios de carcinogenicidad de
dos fases han mostrado que el fenol, aplicado repetidamente a la
piel del ratón, tiene efectos activadores.
7. Efectos en el ser humano
Se ha notificado una larga serie de efectos adversos en el ser
humano resultantes de la exposición bien documentada al fenol por
vía cutánea, oral o intravenosa. Se ha notificado irritación
gastrointestinal tras su ingestión. Los efectos locales de la
exposición cutánea van desde el emblanquecimiento o el eritema
indoloros hasta la corrosión y la necrosis profunda. Entre los
efectos sistémicos cabe citar disritmias, acidosis metabólica,
hiperventilación, disnea, insuficiencia renal aguda, lesiones
renales, orinas oscuras, metahemoglobinemia, trastornos neurológicos
(incluidas convulsiones), choque cardiovascular, coma y muerte. La
dosis mínima reportada como causante de muerte en el ser humano es
de 4,8 g por ingestión; la muerte se produjo en menos de 10 minutos.
Durante mucho tiempo se ha reconocido la posibilidad de
envenenamiento por inhalación de los vapores de fenol, pero no se
han reportado casos mortales relacionados con esta vía de
exposición. Los síntomas que se asocian a la inhalación de fenol
consisten, entre otros, en anorexia, pérdida de peso, dolor de
cabeza, vértigo, salivación y orinas oscuras.
El fenol no es un agente sensibilizante.
El umbral de percepción del fenol por el olfato humano oscila
entre 0,021 y 20 mg/m3 en el aire. Se ha notificado un umbral de
percepción del fenol en el agua de 7,9 mg/litro, y un umbral de
percepción por el gusto de 0,3 mg/litro en el agua.
No se dispone de datos adecuados sobre la carcinogenicidad del
fenol en el ser humano.
8. Efectos en los seres vivos del medio ambiente
En estudios con bacterias de especie única, los valores de la
CE50 con inhibición del crecimiento oscilaron entre 244 y 1600 mg
de fenol/litro. Se comprobó un umbral de toxicidad de 64 mg de
fenol/litro. Los valores para los protozoarios y los hongos fueron
de la misma cuantía que para las bacterias, mientras que para las
algas fueron ligeramente inferiores.
El fenol es tóxico para los organismos superiores de agua
dulce. Los valores más bajos de la CL50 o la CE50, para
crustáceos y peces se sitúan entre 3 y 7 mg de fenol/litro. Los
datos sobre la toxicidad aguda para organismos marinos son
comparables a los correspondientes a organismos de agua dulce. En
estudios de larga duración sobre especies de crustáceos y de peces
se han observado notables diferencias de sensibilidad; los valores
de la CL1 en pruebas con embriones y larvas de Salmo y
Carassius resultaron mucho más bajos (0,2 y 2 µg de fenol/litro,
respectivamente) que los valores correspondientes a otras especies
de peces (NOLC de 2,2-6,1 mg/litro) y anfibios, o que los obtenidos
en pruebas de reproducción en crustáceos (NOLC de 10 mg de
fenol/litro). No se dispone de datos acerca de pruebas de larga
duración realizadas en organismos marinos.
Los factores de bioconcentración del fenol en diversos tipos de
organismos acuáticos son en general muy bajos (< 1-10), aunque se
han notificado también algunos valores más altos (hasta 2200). Así
pues, no se prevé que la bioacumulación del fenol sea significativa.
Los datos sobre el destino y los efectos del fenol en
organismos terrestres son muy escasos. En el mijo se determinó una
CE50 a las 120 horas de 120-170 mg/litro mientras que, en una
prueba de contacto, la CL50 para especies de lombrices resultó ser
de 2,4-10,6 µg/cm2.
9. Resumen de la evaluación
9.1 Salud humana
La población en general está expuesta al fenol principalmente
por inhalación. La exposición repetida por vía oral puede producirse
por el consumo de alimentos ahumados o de agua potable.
No existen datos suficientes para determinar el grado de
exposición de la población en general, pero se puede calcular la
cantidad máxima ingerida diariamente. Basándose en "la peor de las
hipótesis" se puede realizar una estimación suponiendo que un
individuo estará expuesto en grado máximo al fenol mediante la
inhalación continua de aire intensamente contaminado acompañada de
un consumo frecuente de productos alimenticios ahumados y de agua
que contenga fenol hasta niveles de percepción por el gusto. En
total, la ingesta máxima diaria de fenol en un individuo de 70 kg se
calcula en 0,1 mg/kg de peso corporal al día.
Los valores de NOAEL más bajos identificados en experimentos
con animales se refieren a efectos en el riñón y en el desarrollo, y
en las ratas se sitúan dentro de un margen de variación de 12-40
mg/kg de peso corporal al día. Utilizando un factor de incertidumbre
de 200, se recomienda como límite máximo de la ingesta diaria total
(IDT) entre 60 y 200 µg/kg de peso corporal al día. Teniendo en
cuenta que el límite máximo de la ingesta diaria en seres humanos se
calcula en 100 µg/kg de peso corporal al día, se llega a la
conclusión de que la exposición media de la población en general al
fenol, sea cual fuere la fuente, se encuentra por debajo de este
intervalo.
Son motivo de preocupación algunas indicaciones de que el fenol
podría ser genotóxico y el hecho de que no haya datos suficientes
para descartar con seguridad la posible carcinogenicidad del
compuesto. La evaluación debe mantenerse sujeta a revisión
periódica.
9.2 Medio ambiente
No se prevé una bioacumulación importante del fenol. Este
compuesto es tóxico para los organismos acuáticos; mediante la
aplicación del método modificado de la Agencia de los EE.UU. para la
Protección del Medio Ambiente, se puede determinar un nivel en medio
ambiente de preocupación de 0,02 µg/litro. Se carece de datos
adecuados sobre plantas y organismos terrestres.
El transporte de fenol entre compartimientos se produce
principalmente por deposición hídrica y filtración a través del
suelo. En general, es poco probable que el compuesto persista en el
medio ambiente. La escasez de datos sobre la exposición no permiten
evaluar el riesgo que representa el fenol para los ecosistemas tanto
acuáticos como terrestres. Sin embargo, habida cuenta del nivel de
preocupación ambiental que se ha establecido en relación con el
agua, es razonable suponer que los organismos acuáticos pueden
correr riesgo en cualquier agua superficial o marina contaminada con
fenol.
 Relative molecular mass: 94.11
Common name: phenol
Common synonyms: acidum carbolicum, acidum phenolicum,
acidum phenylicum, benzaphenol, benzene
phenol, benzenol carbolic acid,
hydroxybenzene (IUPAC), oxybenzene,
monohydroxybenzene, monophenol, phenic
acid, phenol alcohol, phenyl hydrate,
phenyl hydroxide, phenylic acid
Common trade names: carbololie (NLD), fenololie (NLD),
kristalliertes Kreosot (GER),
Steinkohlenkreosot (GER),
Steinkohlenteerkreosot (GER), venzénol
(FRA), ENT 1814.
CAS registry number:
Relative molecular mass: 94.11
Common name: phenol
Common synonyms: acidum carbolicum, acidum phenolicum,
acidum phenylicum, benzaphenol, benzene
phenol, benzenol carbolic acid,
hydroxybenzene (IUPAC), oxybenzene,
monohydroxybenzene, monophenol, phenic
acid, phenol alcohol, phenyl hydrate,
phenyl hydroxide, phenylic acid
Common trade names: carbololie (NLD), fenololie (NLD),
kristalliertes Kreosot (GER),
Steinkohlenkreosot (GER),
Steinkohlenteerkreosot (GER), venzénol
(FRA), ENT 1814.
CAS registry number: 