
FAO/PL:1968/M/9/1
WHO/FOOD ADD./69.35
1968 EVALUATIONS OF SOME PESTICIDE RESIDUES IN FOOD
THE MONOGRAPHS
Issued jointly by FAO and WHO
The content of this document is the result of the deliberations of the
Joint Meeting of the FAO Working Party of Experts and the WHO Expert
Committee on Pesticide Residues, which met in Geneva, 9-16 December,
1968.
FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS
WORLD HEALTH ORGANIZATION
Geneva, 1969
CHLOROBENZILATE
This pesticide was evaluated for acceptable daily intake by the 1965
Joint Meeting of the FAO Committee on Pesticides in Agriculture and
the WHO Expert Committee on Pesticide Residues (FAO/WHO, 1965). Since
the previous evaluation, additional data has become available and the
monograph has been greatly expanded and is reproduced in its entirety.
IDENTITY
Biological activity
acaricide
Chemical names
ethyl 4,4'-dichlorobenzilate
ethyl 2-hydroxy-2,2-di(p-chlorophenyl)-acetate. (IUPAC)
Synonyms
Acaraben(R), Akar(R), Gesaspint(R)
Structural formula
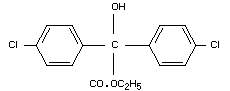 Other information on identity and properties
The technical material consists of a yellow-brown oily liquid with a
purity of at least 90 per cent. No data have been submitted on the
composition of the technical product.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
Biochemical aspects
Three groups, each containing one male and one female dog, were given
0, 12.8, or 64.1 mg/kg/day for five consecutive days. At autopsy, five
days after the termination of treatment, no residues could be detected
in whole blood, liver, kidney, muscle, fat, or brain tissues. At 12.8
mg/kg/day, the male excreted 31 per cent of the total dose in the
urine within 10 days. Urinary excretion in the female accounted for 41
per cent. Faecal levels contained 5.6 per cent and 6.6 per cent for
the male and female respectively. At 64.1 mg/kg/day urinary and faecal
elimination was 31 per cent and 1.7 per cent respectively for the
male, and 23 per cent and 26 per cent for the female. The method of
analysis would not detect conjugates of 4,4'-dichlorobenzilic acid
which is suggested to be the major metabolite (Hazleton Laboratories
Inc., 1964). Dichlorobenzilic acid has been qualitatively identified
in dog urine by thin-layer chromatographic techniques (Mattson et al.,
1965).
Acute toxicity (oral)
Animal Route Compound or LD50 (mg/kg Reference
solvent body-weight)
Mouse Oral Suspension in 4 850 Gasser, 1952
gum arabic
Mouse Oral Technical 729 Horn et al., 1955
material
Rat Oral Suspension in 3 100 Gasser, 1952
gum arabic
Rat Oral Technical 702 Horn et al., 1955
material
Rat Oral 25 per cent. 735 Horn et al., 1955
xylene
emulsion
Short-term studies
Avian wildlife
Groups of 10 Mallard ducks (Aras platyrhynchus) were fed 0, 0.25,
0.45 and 0.8 per cent of chlorobenzilate for five consecutive days.
The LD50 was greater than 0.8 per cent. Body-weight gain and food
intake was slightly reduced in all test groups (Woodard, 1965a).
Groups of 10 quail (Colinus virginianus) were fed 0, 0.14, 0.25
and 0.5 per cent of chlorobenzilate in their diet for seven days. The
LD50 was 0.34 per cent. Bodyweight, food intake and gross pathology
of the survivors appeared unaffected (Woodard, 1965a).
Rat
Groups, each of 20 male rats, were fed 0, 40 and 800 ppm technical
chlorobenzilate in their diet for 48, 48 and 44 weeks, respectively.
Mortality was slightly higher in the 800 ppm group than in the control
group. The growth of the animals fed 800 ppm was retarded, and several
animals exhibited red or swollen eyelids and soft faeces. Organ to
body-weight ratios for the 40 ppm group were significantly greater
than for the controls, in the case of liver, kidney and testes.
Similar data for the 800 ppm group were not evaluated statistically,
but organ to body-weight ratios in the case of the liver and kidney
appear to be greater than for the controls. Gross pathological
examination revealed no changes attributable to compound
administration, but upon histological examination non-specific changes
were observed in the pancreas and adrenals of animals in both test
groups, and increased hemopoietic activity in the spleens of the 800
ppm groups. Tissue analysis indicated that chlorobenzilate was not
stored in the animal to any appreciable extent (Hazleton Laboratories,
1953a).
Groups of five male rats received 0, 1, 3 or 5 per cent technical
chlorobenzilate in the diet, and groups of five female rats received
0, or 3 per cent, in the diet. All animals receiving chlorobenzilate
at these levels died, those receiving one per cent within 59 days,
three per cent within eight days, and five per cent within six days.
Reduced food consumption, and marked loss of body-weight preceded
death.(Hazleton Laboratories, 1953b).
A 15 week feeding study, utilizing groups of five male rats at dose
levels of 0, 500, 1000, or 5000 ppm technical chlorobenzilate resulted
in reduced food consumption, and growth retardation in all test
groups. Mortality was 40 per cent at 1000 ppm, and 100 per cent at
5000 ppm (Hazleton Laboratories, 1953b).
In a 17 week feeding study using groups of 20 male rats fed 0, 50, and
500 ppm, and 20 female rats at 0, and 500 ppm, body weight gain was
depressed in the male only, at 500 ppm. Food intake was not
significantly depressed (Hazleton Laboratories, 1953b).
Groups of 20 male and 20 female rats treated for 99 days, with a 20
per cent chlorobenzilate powder mixed in their diet at dose levels of
0, 20, 100, 500, and 2500 ppm active ingredient did not show any toxic
effects as judged by food consumption, body-weight, mortality, organ
weights, gross, or histopathology except at the top dose level. At
2500 ppm active ingredient, food intake was reduced initially,
body-weight gain was reduced, absolute testes and spleen weights were
below control levels (insufficient data available to calculate organ
to body-weight ratios), and histopathological examination revealed
three rats with increased fatty deposits in the liver lobules, as well
as five rats with spermatogenentic injury, and testicular atrophy
(Domenjoz, 1965a).
Dog
Pairs of dogs (one male and one female) were fed 12.8, or 64.1 mg/kg
five times weekly for 35 weeks, except for two periods of five days
when excretion studies were performed. No toxic effects were apparent,
judging by growth, general appearance, hematology, urinalysis, liver
function test, and gross and microscopic pathology. The excretion
studies showed that the majority of administered material was excreted
in the urine, the metabolite having a 4,4'-dichlorodiphenyl methyl
structure. No chlorobenzilate was present in the urine three days
after withdrawal (Horn et al., 1955).
In a two year feeding study utilizing the 25 per cent powder
formulation, groups of three male and three female dogs were fed 0,
100, or 500 ppm active ingredient in the diet. A further group of
three male and three female dogs were fed 5000 ppm active ingredient
for 14 weeks. One male and one female were maintained at this dose
level for 20 weeks when they were sacrificed. The remainder, after a
five week withdrawal period, were replaced in the study by a 3000 ppm
dose level, until the completion of the test. At 100, and 500 ppm, the
parameters considered were comparable to the control group. At 3000
ppm, body-weight depression, mild anaemia and increased liver and
spleen to body-weight ratios were observed in both sexes. At 5000 ppm,
in addition to these effects, food intake was depressed, anaemia was
moderately severe, serum alkaline phosphatase values were elevated,
and the albumen to globulin ratio was reversed. The histopathology,
presumably performed at 104 weeks, indicates no compound related
effects at 100 and 500 ppm. At the top level, extramedullary
haematopoiesis was evident in liver and spleen as well as erythroid
hyperplasia of the bone marrow (Hazleton Laboratories Inc., 1965).
Sheep and beef-cattle
Sheep were exposed to chlorobenzilate 25 per cent wettable powder in
the diet for four weeks at dose levels of 0 (two females), 3.6 (two
females), 8.7 (two females) or 29 (one male and four females) ppm
active ingredient, and beef-cattle at dose levels of 0 (one male and
one female), 3.2 (one male and one female), 8.8 (one male and one
female), or 27 (one male and one female) ppm active ingredient. The
sheep and cattle failed to show compound related toxic symptoms as
judged by food consumption, bowel evacuations, physical condition and
behaviour, haematology, or gross necropsy observation. One cow dosed
at 3.2 ppm aborted a six-month foetus on day four. However, a second
cow dosed at 8.8 ppm carried a fully-developed foetus until sacrificed
(Woodard, 1965c).
Long-term studies
Rat
A two year study, using technical chlorobenzilate at 0 (20 male and 20
female), 50 (20 male) and 500 ppm (20 male and 20 female) showed
decreased body-weight gain, as well as unthriftiness, and blood tinged
crusts about the nose and eyes at 500 ppm. An increased incidence of
testicular atrophy was noted at both 50 and 500 ppm without
histological change (Horn et al., 1955).
A second two year study utilizing groups of 30 male and 30 female rats
at dose levels of 0, 40, 125 and 400 ppm active ingredient as the 25
per cent wettable formulation showed no significant changes in
body-weight gain, or food intake. Haematologic values did not show any
consistent changes related to dose level, or length of exposure to the
test material. Incidence of neoplasms was unrelated to the
administration of the test substances. Adverse effects on organ to
body-weight ratios were noted in the case of liver at 400 ppm. In the
histopathological examination of tissues, changes were observed in
liver, kidney and testes at 400 ppm, and in kidney and testes at 125
ppm. Gross autopsy observations indicated testicular changes at 125
and 400 ppm (Woodard, 1966).
Special studies
(a) Reproduction
A three generation study, utilizing 20 male and 20 female rats at 0,
and 50 ppm active ingredients as the 25 per cent wettable formulation
in the first generation, and, 10 male and 20 female rats at 0, 25 and
50 ppm active ingredient in the second and third generations failed to
reveal any toxic effects on adult body-weight, resorption rates, or
histology (performed on F1b adults). Absolute testes weights for the
F1b adults (the only group reported) showed a significant decrease at
50 ppm active ingredient. The litters all appeared normal as judged by
litter size, birth weight, stillbirths, survival to weaning, weanling
weights, and F3b weanling histopathology. Adult fertility, as judged
by number of litters per group appeared normal, but chlorobenzilate
withdrawal at weaning for 15 days, and at weaning for 28 days in F1b
and F2b weanlings, respectively, leaves this parameter open to
question. No teratogenic changes were observed at any stage in the
study (Woodard, 1965b).
(b) Studies on the metabolite
The metabolite, dichlorbenzilic acid was fed to groups of 20 male and
20 female rats for 99 days at dose levels of 0, 20, 100, 500 and 2500
ppm active ingredient in a 20 per cent powder. Food consumption
body-weight gain, mortality, organ weights, and gross and
histopathology showed no significant changes from the controls, except
possibly for a slight depression of kidney and testes weight at 2500
ppm (Domenjoz, 1965b).
Comments
Adequate data have been presented on short-term and long-term studies
in rats and dogs. Biochemical data on excretion, and a tentative
identification of the major metabolite are also available. All the
studies appear to be reliable and sufficiently adequate to permit
evaluation. The slight doubts regarding the incidence of neoplasms
observed in the long-term study on the rat makes it desirable to
obtain information from a second species.
TOXOCOLOGICAL EVALUATION
Level causing no significant toxicological effect
Rat: 40 ppm in the diet, equivalent to 2 mg/kg body-weight
Dog: 500 ppm in the dry diet, equivalent to 12.5 mg/kg
body-weight
Estimate of acceptable daily intake for man
0-0.02 mg/kg body-weight
RESIDUES IN FOOD AND THEIR EVALUATION
Use pattern
Pre-harvest treatments
Chlorobenzilate is used as a contact acaricide for the control of
adult spider mites; it is also effective against summer eggs and all
post-embryonic stages. The compound is used in many countries
throughout the world for the control of several mite species on
apples, pears, stone fruits, citrus fruits, soft fruit, grapes,
olives, vegetables, coffee, tea, cotton and ornamentals.
Chlorobenzilate is less effective at lower temperatures, and in
countries with a temperate climate, it is primarily used on
glass-house crops and only to a small extent outdoors. In view of its
specific acaricidal activity and its non-toxicity to bees, it may be
usefully applied in integrated control schemes. In certain instances
it is being recommended when resistance to other acaricides has
developed.
The recommended rates of application range from 25-50 g/100 l, and the
mount of spray liquid applied is generally 500-1000 l/ha for low-grown
crops (vegetables, etc) and 2000-2500 l/ha for high-grown crops (e.g.
fruit orchards). (U.S. Dept. of Agric., 1967)
Post-harvest treatments
No post-harvest treatment is recommended.
Other uses
Chlorobenzilate is being used for the control of mites on ornamental
plants. The compound is also used as a smoke for the treatment of
bee-hives against tracheal mites (Gubler, 1953).
Residues resulting from supervised trials
Residue data are available from supervised trials on several food
crops, grown under various conditions, using various rates of
application and pre-harvest intervals (Geigy, 1968). These data refer
to apples, citrus, nuts and vegetables (melons, cantaloupes). In most
cases normal dosage rates were applied in accordance with label
recommendations; in a few experiments higher dosages were included.
Results mainly refer to work in the United States of America. Only
limited data are available from other countries.
The following table summarizes typical residue data.
Rate of Post treatment Residue, ***
Crop application Number of interval whole fruit
(g/100 1) treatments (days) basis (ppm)
Apples 31 1 1 2.24
31 1 8 1.79
62 1 1 4.98
62 1 8 4.33
25 1 6 0.72
25 1 35 0.41
60 1 39 0.90
Grapefruit 30 1 1 1.10*
30 1 22 2.04*
Oranges 30 1 1 1.22*
30 1 22 1.99*
Grapes 60 1 17 0.62*
60 1 34 1.06*
120 1 17 1.20*
120 1 34 1.15*
Almonds 45 1-2 0-63 < 0.1
Melons 0.56** 1 1 < 0.04-0.4
0.56** 1 14 < 0.04-0.13
2.24** 1 1 0.08-1.1
2.24** 1 14 0.09-0.39
* Based on peel; analysis of pulp showed residues of less than 0.1 ppm
in the case of apples - no data on residues in pulp of grapes.
** kg/ha
*** UV method (Blinn and Gunther, 1963) except GLC (see below) for grapes,
almonds and melons.
Fate of residues
General comments
Chorobenzilate can be considered as a persistent compound. More
information is needed on the nature of terminal residues in plants,
animals and their products.
In soils
No information available.
In plants
Experiments have been carried out to examine if and to what extent
breakdown products derived from chlorobenzilate could be found, with
particular reference to the possible occurrence of
4,4'-dichlorobenzilic acid after apples were sprayed with 2 lbs/ 100
gal and samples were taken 3, 14 and 21 days. The sensitivity of the
analytical method was 0.1 ppm. In recovery studies,
4,4'-dichlorobenzilic acid could be detected without interference of
chlorobenzilate. It appears that chlorobenzilate is found mainly on
the outer surface of the treated apples. No unchanged chlorobenzilate
could be detected in the pulp of the apples. None of the treated
apples contained detectable amounts of 4,4'-dichlorobenzilic acid
(Murphy et al., 1966).
In animals
In experiments on sheep and cattle (Mattson and Schneller, 1966) 16
per cent protein mixed grain containing chlorobenzilate was given in
addition to a daily ration of mixed timothy and clover hay to eight
sheep and six cattle (both sexes) over a period of four weeks. The
dosage levels were 10, 24 and 80 mg chlorobenzilate per animal per day
for sheep and 40, 110 and 340 mg chlorobenzilate per animal for
cattle.
Residues of unchanged chlorobenzilate were only found in cattle at the
highest feeding level (340 mg/animal/day).
Residues in cattle
(ppm in fat)
Males Females
subcutaneous fat 0.54 0.32
omental fat 0.98 0.69
perirenal fat 0.54 0.69
In another experiment (Woodard, 1960) chlorobenzilate was administered
to six dairy cows at dietary levels of 20, 48 and 160 ppm in the feed
(intake 4 lbs of treated feed per day) for a period of 15 days. After
16 days, treatment with chlorobenzilate was terminated and the animals
were given the same type of feed, without chlorobenzilate, to permit
post-treatment analysis of milk samples. Milk samples were taken on
the second, fourth, sixth, ninth and fifteenth days of the experiment,
and again on the twentieth, twenty-first and twenty-second day.
Chlorobenzilate in the milk was found in one "high-level" cow (160
ppm) on the second day (0.15 ppm) and in one "medium-level" cow (48
ppm) on the fourth day (0.15 ppm). On the fifteenth day, residues did
not exceed 0.06 ppm. Since these levels are approaching the limit of
sensitivity of the analytical method used, there is no distinct
correlation between residue and feeding levels, and a number of
negative values were obtained, the few positive values may be
fortuitous. No 4,4'-dichlorobenzilic acid was found in the milk.
Limited recovery studies show that this compound, if present, would be
detected with the methods used.
In the above short-term feeding experiments rather high dosages have
been used, and more information is required on the possible occurrence
of residues in milk after feeding normal dosage levels for a prolonged
period.
In storage and processing
No data are available on the fate of residues during storage and
processing. Since the residue remains mainly on the outer side of
fruit and migrates only to a very small extent or not at all into the
pulp, it may be expected that washing and peeling of treated fruit
will remove most of the residue. No data are available on the effect
of cooking.
Evidence of residues in food in commerce or at consumption
Food moving in commerce
No information available.
Food at the time of consumption
In "market basket" or "total diet" studies (Cummings, 1966), carried
out by the United States Food and Drug Administration, multidetection
methods were used for residue analysis. The analytical procedure used
for all samples in this study enabled the detection of about 54
pesticide chemicals, including chlorobenzilate. So far no residues of
chlorobenzilate have been found.
Methods of residue analysis
Several methods are available for residue analysis of chlorobenzilate,
e.g. ultra-violet or infra-red spectrophotometry and gas-liquid
chromotography. In former years the ultra-violet method, as described
by Blinn and Gunther (1963) was used for determination of
chlorobenzilate residues on apples, pears and citrus fruit. In
addition Harris (1955) developed a modified Schechter-Haller infra-red
spectrophotometric method for residue analysis of chlorobenzilate.
This method was suitable for residue analysis on apples, pears and
strawberries. A modification of this method is described by Margot and
Stammbach (1964).
At present a gas chromatographic method appears to be most suitable
for use as a referee method, and should be developed for this purpose
through collaborative studies, in particular, the comparative
evaluation of the different detectors.
The following is a brief description of the gas chromatographic
determination of chlorobenzilate and chloropropylate in fruits,
vegetables and nuts. The method is sensitive to 0.05 ppm:
Chlorobenzilate and chloropropylate are extracted from the chopped
material with petroleum ether (boiling range 30-60°C). Part of the
extract is evaporated to dryness, taken up in benzene and transferred
to an alumina column (basic, activity grade V). Interfering materials
are removed by eluting with hexane or with 10 per cent benzene in
hexane. Chlorobenzilate and chloropropylate are eluted with
hexane/benzene (1:1) and the content is determined by gas
chromotography using a glass column (4 ft × 0.25 in) packed with five
per cent silicone gum GE XE - 60 (nitrile) supported on Anakrom ABS
(50-60 mesh) with nitrogen as the carrier gas and a microcoulometric
detector. Conditions: oven temperature, 215-220°C; block temperature,
220-225°C; furnace temperature, 810-820°C; nitrogen flow, 80 ml/min;
oxygen flow, 100 ml/min; pure nitrogen flow, 110 ml/min.
Conditioning of the column is important and can be achieved by three
to four injections of 30 micrograms of the acaricide. Retention times
are approximately 4 min. Separation of chlorobenzilate and
chloropropylate can be obtained by using a column loading of 10 per
cent XE-60 and a column length of 6 ft (retention times approximately
18 min for chlorobenzilate and 20 min for chloropropylate).
Apples, pears, stone fruits, citrus fruits, berries, nuts, vegetables
and cotton have been analysed for chlorobenzilate.
Apples, pears, stone fruits, citrus fruits, almonds, cucumbers and
beans have been analysed for chloropropylate.
Typical recovery data with fortified samples (sample weight 1-5 g)
are:
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Green beans 73-100
(continued)
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Maize (ears) 80-120
Maize (foliage) 80-95
Ground nuts 78-80
Citrus (peel) 89-110
Citrus (pulp) 70-80
Pears (peel) 90-100
Pears (pulp) 100-110
For oleagenous samples such as nuts, seeds and citrus peel, an
additional petroleum ether-acetonitrile partitioning step is
necessary.
Using the described clean-up procedure no interference from DDT and
DDD is encountered.
National tolerances
Country Crop Tolerance (ppm)
Canada apples, pears, citrus fruit,
cantaloupes 8
Netherlands fruit and vegetables 2
New Zealand fruit and vegetables 5
United States
of America apples, pears, citrus fruit,
melons 5
almonds, walnuts 0.2
almond hulls 15
cotton-seed 0.5
meat, fat and meat by-products 0.5
of cattle and sheep
(United States of America Federal Register)
RECOMMENDATIONS FOR TOLERANCES AND PRACTICAL RESIDUE LIMITS
Appraisal
Chlorobenzilate is a persistent and specific acaricide used on a
fairly wide range of crops. In many instances it is active against
mites which have developed resistance to other acaricides.
The compound is non-toxic to bees, and may be usefully applied in
integrated control schemes. Although the compound is registered in
many countries, little information was available on the extent of the
use in these countries. Many of the data furnished are based upon
experiments which have been conducted in the United States of America.
Furthermore, no information was available on the total composition of
the technical product.
The compound has not been detected in total diet studies in the United
States of America. There was a lack of data on the disappearance of
the compound and the disappearance of residues during storage and
processing. Information is required on the possible carry-over of the
residue into wine before tolerances for grapes can be recommended.
Although data were provided on residues in milk following feeding at
excessively high dosages to dairy cattle for a short period of time
(16 days), there were no data furnished on possible residues in milk
following the feeding of treated feed at normal residue levels for
longer periods. However, the residue occurring as the result of these
high-level feeding trials was at the limit of detection of the
analytical method (0.05 ppm) and it would not be expected that a
higher residue level would result from feeding lower levels for longer
periods in view of the fact that the compound is not cumulative.
In animals the compound is metabolized to form 4,4'-dichlorobenzilic
acid. No information is available on the nature of the terminal
residues in plants and in animal products.
A referee method of analysis has not been established, although
gas-liquid chromatography would seem to be most suitable for this
purpose, but must be further developed by collaborative studies.
Comparative evaluation of the different detectors used in gas
chromatographic methods, and evaluation of different methods of
extraction are needed.
Recommendations
The following temporary tolerances (to be in effect until 1972) are to
apply to raw agricultural products moving in commerce unless otherwise
indicated. In the case of fruits and vegetables, the tolerances should
be applied as soon as practicable after harvest and in any event prior
to actual retail to the public. In the case of commodities entering
international trade, the tolerances should be applied by the importing
country at the point of entry or as soon as practicable thereafter.
The tolerances are for the parent compound only.
apples, pears (whole fruit basis) 5.0 ppm
citrus (whole fruit basis) 1.0 ppm
melons, cantaloupes 1.0 ppm
almonds, walnuts 0.2 ppm
Further work or information
Required before 30 June 1972
1. Information on the composition of the technical product.
2. Information on the nature of terminal residues in plants, animals,
and their products.
3. Data from countries other than the United States of America on the
required rates and frequencies of application, pre-harvest intervals,
and the resultant residues.
4. Further data on the disappearance of residues in soils, in plants,
and in plant products during storage and processing.
5. Data on the possible carry-over of residues into wine as a result
of the treatment of grapes.
6. Further data on the occurrence of residues in milk after feeding
dairy cows at normal residue levels of the compound in the feed.
7. Comparative evaluation of the different detectors used in gas
liquid chromatographic methods and of different methods of extraction
for regulatory purposes.
Desirable
1. Collaborative studies to establish a referee method.
2. Metabolic studies in animals.
3. Investigation of possible testicular effects and long-term studies
in species other than the rat on the incidence of neoplasms.
REFERENCES
Blinn, R. C. and Gunther, F. A. (1963) The utilization of infrared and
ultraviolet spectrophotometric procedures for assay of pesticide
residues. Residue Reviews 2: 99-152
Cummings, J. G. (1966) Pesticides in the total diet. Residue Reviews
16: 30-45
Domenjoz, R. (1965a) Chlorbenzilat Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Bonn. Unpublished report.
Domenjoz, R. (1965b) Dichlorbenzilsäure Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Boon. Unpublished report.
FAO/WHO. (1965) Evaluation of the toxicity of pesticide residues in
food. FAO Mtg Rept No. PL:1965/10/1; WHO/Food Add./27.65
Gasser, R. (1952) Über zwei neue Akarizide aus der Gruppe der Di-(p-
chlorphenyl)-karbinole. Experientia, 8: 65-67
Geigy S.A., J.R. (1968) Chlorobenzilate working paper. Unpublished
report.
Gubler, W. et al. (1953) Schweizerische Bienenzeitung, 7: 268
Harris, H. J. (1955) Colorimetric determination of ethyl
4,4'-dichlorbenzilate (chlorobenzilate) as a spray residue. J. Agr.
Food Chem., 3: 939-941
Hazleton Laboratories Inc. (1953a) Geigy 338, technical. Chronic
feeding - rats. Unpublished report
Hazleton Laboratories Inc. (1953b) Geigy 338, technical feeding
studies - rats. Unpublished report
Hazleton Laboratories Inc. (1964) Method and development for the
analysis of chloropropylate and chlorobenzilate in biological fluids
and tissue specimens. Metabolic distribution and excretion of
chloropropylate and chlorobenzilate in dogs. Unpublished report
Hazleton Laboratories Inc. (1965) Chlorobenzilate. Two year dietary
feeding study - purebred beagles. Unpublished report
Horn, H. J., Bruce, R. B, and Paynter, O.E. (1955) Toxicology of
chlorobenzilate. J. Agr. Food Chem., 3: 752-756
Industrial Bio-Test Laboratories. (1965) Report to Geigy Chemical
Corp. Acute toxicity studies on chlorobenzilate 25W. Unpublished
report
Margot, A. and Stammbach, K. (1964) Chlorobenzilate; in: Zweig, G.,
1964. Analytical methods for pesticides, plant regulators and food
additives. Academic Press, London and New York, Vol. 2, 65-73
Mattson, A. M., Beaudoin, R. L. and Schneller, J. (1965) Comparison of
urinary metabolites in dogs after administration of chlorobenzilate or
chloropropylate. Analytical Dept. of Geigy Research Laboratories.
Unpublished report
Mattson, A. M. and Schneller, J. (1966) Chlorobenzilate residues in
sheep and cattle tissues. Unpublished report of the Analytical
Department of Geigy Research Laboratories, Division of Geigy Chem.
Corp., Ardsley, New York.
Murphy, R., Kahrs, R. and Mattson, A. M. (1966) Dissipation of
residues of chlorobenzilate and chloropropylate on apples. Unpublished
report of the Analytical Department of Geigy Research Laboratories,
Division of Geigy Chem. Corp., Ardsley, New York
United States of America Federal Register, 27, F.R. 12092, pav.
120-129
United States of America Department of Agriculture. (1966) Summary of
registered pesticide chemical cases. 1st October
Woodard. (1960) Examination of milk for residues following feeding of
chlorobenzilate to dairy cows. Unpublished report, Woodard Research
Corp.
Woodard. (1965a) Chlorobenzilate. Safety evaluation on fish and wild
life. (Bobwhite quail, mallard ducks, rainbow trout, sunfish,
goldfish, oysters). Unpublished report, Woodard Research Corp.
Woodard. (1965b) Chlorobenzilate. Three generation reproduction study
in the rat. Unpublished report, Woodard Research Corp.
Woodard. (1965c) Safety evaluation and determination of the amount of
chlorobenzilate that might appear in the tissues of sheep and
beef-cattle fed this material in the diet for four weeks. Unpublished
report, Woodard Research Corp.
Woodard. (1966) Chlorobenzilate. Safety evaluation by dietary feeding
to rats for 104 weeks. Unpublished report, Woodard Research Corp.
Other information on identity and properties
The technical material consists of a yellow-brown oily liquid with a
purity of at least 90 per cent. No data have been submitted on the
composition of the technical product.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
Biochemical aspects
Three groups, each containing one male and one female dog, were given
0, 12.8, or 64.1 mg/kg/day for five consecutive days. At autopsy, five
days after the termination of treatment, no residues could be detected
in whole blood, liver, kidney, muscle, fat, or brain tissues. At 12.8
mg/kg/day, the male excreted 31 per cent of the total dose in the
urine within 10 days. Urinary excretion in the female accounted for 41
per cent. Faecal levels contained 5.6 per cent and 6.6 per cent for
the male and female respectively. At 64.1 mg/kg/day urinary and faecal
elimination was 31 per cent and 1.7 per cent respectively for the
male, and 23 per cent and 26 per cent for the female. The method of
analysis would not detect conjugates of 4,4'-dichlorobenzilic acid
which is suggested to be the major metabolite (Hazleton Laboratories
Inc., 1964). Dichlorobenzilic acid has been qualitatively identified
in dog urine by thin-layer chromatographic techniques (Mattson et al.,
1965).
Acute toxicity (oral)
Animal Route Compound or LD50 (mg/kg Reference
solvent body-weight)
Mouse Oral Suspension in 4 850 Gasser, 1952
gum arabic
Mouse Oral Technical 729 Horn et al., 1955
material
Rat Oral Suspension in 3 100 Gasser, 1952
gum arabic
Rat Oral Technical 702 Horn et al., 1955
material
Rat Oral 25 per cent. 735 Horn et al., 1955
xylene
emulsion
Short-term studies
Avian wildlife
Groups of 10 Mallard ducks (Aras platyrhynchus) were fed 0, 0.25,
0.45 and 0.8 per cent of chlorobenzilate for five consecutive days.
The LD50 was greater than 0.8 per cent. Body-weight gain and food
intake was slightly reduced in all test groups (Woodard, 1965a).
Groups of 10 quail (Colinus virginianus) were fed 0, 0.14, 0.25
and 0.5 per cent of chlorobenzilate in their diet for seven days. The
LD50 was 0.34 per cent. Bodyweight, food intake and gross pathology
of the survivors appeared unaffected (Woodard, 1965a).
Rat
Groups, each of 20 male rats, were fed 0, 40 and 800 ppm technical
chlorobenzilate in their diet for 48, 48 and 44 weeks, respectively.
Mortality was slightly higher in the 800 ppm group than in the control
group. The growth of the animals fed 800 ppm was retarded, and several
animals exhibited red or swollen eyelids and soft faeces. Organ to
body-weight ratios for the 40 ppm group were significantly greater
than for the controls, in the case of liver, kidney and testes.
Similar data for the 800 ppm group were not evaluated statistically,
but organ to body-weight ratios in the case of the liver and kidney
appear to be greater than for the controls. Gross pathological
examination revealed no changes attributable to compound
administration, but upon histological examination non-specific changes
were observed in the pancreas and adrenals of animals in both test
groups, and increased hemopoietic activity in the spleens of the 800
ppm groups. Tissue analysis indicated that chlorobenzilate was not
stored in the animal to any appreciable extent (Hazleton Laboratories,
1953a).
Groups of five male rats received 0, 1, 3 or 5 per cent technical
chlorobenzilate in the diet, and groups of five female rats received
0, or 3 per cent, in the diet. All animals receiving chlorobenzilate
at these levels died, those receiving one per cent within 59 days,
three per cent within eight days, and five per cent within six days.
Reduced food consumption, and marked loss of body-weight preceded
death.(Hazleton Laboratories, 1953b).
A 15 week feeding study, utilizing groups of five male rats at dose
levels of 0, 500, 1000, or 5000 ppm technical chlorobenzilate resulted
in reduced food consumption, and growth retardation in all test
groups. Mortality was 40 per cent at 1000 ppm, and 100 per cent at
5000 ppm (Hazleton Laboratories, 1953b).
In a 17 week feeding study using groups of 20 male rats fed 0, 50, and
500 ppm, and 20 female rats at 0, and 500 ppm, body weight gain was
depressed in the male only, at 500 ppm. Food intake was not
significantly depressed (Hazleton Laboratories, 1953b).
Groups of 20 male and 20 female rats treated for 99 days, with a 20
per cent chlorobenzilate powder mixed in their diet at dose levels of
0, 20, 100, 500, and 2500 ppm active ingredient did not show any toxic
effects as judged by food consumption, body-weight, mortality, organ
weights, gross, or histopathology except at the top dose level. At
2500 ppm active ingredient, food intake was reduced initially,
body-weight gain was reduced, absolute testes and spleen weights were
below control levels (insufficient data available to calculate organ
to body-weight ratios), and histopathological examination revealed
three rats with increased fatty deposits in the liver lobules, as well
as five rats with spermatogenentic injury, and testicular atrophy
(Domenjoz, 1965a).
Dog
Pairs of dogs (one male and one female) were fed 12.8, or 64.1 mg/kg
five times weekly for 35 weeks, except for two periods of five days
when excretion studies were performed. No toxic effects were apparent,
judging by growth, general appearance, hematology, urinalysis, liver
function test, and gross and microscopic pathology. The excretion
studies showed that the majority of administered material was excreted
in the urine, the metabolite having a 4,4'-dichlorodiphenyl methyl
structure. No chlorobenzilate was present in the urine three days
after withdrawal (Horn et al., 1955).
In a two year feeding study utilizing the 25 per cent powder
formulation, groups of three male and three female dogs were fed 0,
100, or 500 ppm active ingredient in the diet. A further group of
three male and three female dogs were fed 5000 ppm active ingredient
for 14 weeks. One male and one female were maintained at this dose
level for 20 weeks when they were sacrificed. The remainder, after a
five week withdrawal period, were replaced in the study by a 3000 ppm
dose level, until the completion of the test. At 100, and 500 ppm, the
parameters considered were comparable to the control group. At 3000
ppm, body-weight depression, mild anaemia and increased liver and
spleen to body-weight ratios were observed in both sexes. At 5000 ppm,
in addition to these effects, food intake was depressed, anaemia was
moderately severe, serum alkaline phosphatase values were elevated,
and the albumen to globulin ratio was reversed. The histopathology,
presumably performed at 104 weeks, indicates no compound related
effects at 100 and 500 ppm. At the top level, extramedullary
haematopoiesis was evident in liver and spleen as well as erythroid
hyperplasia of the bone marrow (Hazleton Laboratories Inc., 1965).
Sheep and beef-cattle
Sheep were exposed to chlorobenzilate 25 per cent wettable powder in
the diet for four weeks at dose levels of 0 (two females), 3.6 (two
females), 8.7 (two females) or 29 (one male and four females) ppm
active ingredient, and beef-cattle at dose levels of 0 (one male and
one female), 3.2 (one male and one female), 8.8 (one male and one
female), or 27 (one male and one female) ppm active ingredient. The
sheep and cattle failed to show compound related toxic symptoms as
judged by food consumption, bowel evacuations, physical condition and
behaviour, haematology, or gross necropsy observation. One cow dosed
at 3.2 ppm aborted a six-month foetus on day four. However, a second
cow dosed at 8.8 ppm carried a fully-developed foetus until sacrificed
(Woodard, 1965c).
Long-term studies
Rat
A two year study, using technical chlorobenzilate at 0 (20 male and 20
female), 50 (20 male) and 500 ppm (20 male and 20 female) showed
decreased body-weight gain, as well as unthriftiness, and blood tinged
crusts about the nose and eyes at 500 ppm. An increased incidence of
testicular atrophy was noted at both 50 and 500 ppm without
histological change (Horn et al., 1955).
A second two year study utilizing groups of 30 male and 30 female rats
at dose levels of 0, 40, 125 and 400 ppm active ingredient as the 25
per cent wettable formulation showed no significant changes in
body-weight gain, or food intake. Haematologic values did not show any
consistent changes related to dose level, or length of exposure to the
test material. Incidence of neoplasms was unrelated to the
administration of the test substances. Adverse effects on organ to
body-weight ratios were noted in the case of liver at 400 ppm. In the
histopathological examination of tissues, changes were observed in
liver, kidney and testes at 400 ppm, and in kidney and testes at 125
ppm. Gross autopsy observations indicated testicular changes at 125
and 400 ppm (Woodard, 1966).
Special studies
(a) Reproduction
A three generation study, utilizing 20 male and 20 female rats at 0,
and 50 ppm active ingredients as the 25 per cent wettable formulation
in the first generation, and, 10 male and 20 female rats at 0, 25 and
50 ppm active ingredient in the second and third generations failed to
reveal any toxic effects on adult body-weight, resorption rates, or
histology (performed on F1b adults). Absolute testes weights for the
F1b adults (the only group reported) showed a significant decrease at
50 ppm active ingredient. The litters all appeared normal as judged by
litter size, birth weight, stillbirths, survival to weaning, weanling
weights, and F3b weanling histopathology. Adult fertility, as judged
by number of litters per group appeared normal, but chlorobenzilate
withdrawal at weaning for 15 days, and at weaning for 28 days in F1b
and F2b weanlings, respectively, leaves this parameter open to
question. No teratogenic changes were observed at any stage in the
study (Woodard, 1965b).
(b) Studies on the metabolite
The metabolite, dichlorbenzilic acid was fed to groups of 20 male and
20 female rats for 99 days at dose levels of 0, 20, 100, 500 and 2500
ppm active ingredient in a 20 per cent powder. Food consumption
body-weight gain, mortality, organ weights, and gross and
histopathology showed no significant changes from the controls, except
possibly for a slight depression of kidney and testes weight at 2500
ppm (Domenjoz, 1965b).
Comments
Adequate data have been presented on short-term and long-term studies
in rats and dogs. Biochemical data on excretion, and a tentative
identification of the major metabolite are also available. All the
studies appear to be reliable and sufficiently adequate to permit
evaluation. The slight doubts regarding the incidence of neoplasms
observed in the long-term study on the rat makes it desirable to
obtain information from a second species.
TOXOCOLOGICAL EVALUATION
Level causing no significant toxicological effect
Rat: 40 ppm in the diet, equivalent to 2 mg/kg body-weight
Dog: 500 ppm in the dry diet, equivalent to 12.5 mg/kg
body-weight
Estimate of acceptable daily intake for man
0-0.02 mg/kg body-weight
RESIDUES IN FOOD AND THEIR EVALUATION
Use pattern
Pre-harvest treatments
Chlorobenzilate is used as a contact acaricide for the control of
adult spider mites; it is also effective against summer eggs and all
post-embryonic stages. The compound is used in many countries
throughout the world for the control of several mite species on
apples, pears, stone fruits, citrus fruits, soft fruit, grapes,
olives, vegetables, coffee, tea, cotton and ornamentals.
Chlorobenzilate is less effective at lower temperatures, and in
countries with a temperate climate, it is primarily used on
glass-house crops and only to a small extent outdoors. In view of its
specific acaricidal activity and its non-toxicity to bees, it may be
usefully applied in integrated control schemes. In certain instances
it is being recommended when resistance to other acaricides has
developed.
The recommended rates of application range from 25-50 g/100 l, and the
mount of spray liquid applied is generally 500-1000 l/ha for low-grown
crops (vegetables, etc) and 2000-2500 l/ha for high-grown crops (e.g.
fruit orchards). (U.S. Dept. of Agric., 1967)
Post-harvest treatments
No post-harvest treatment is recommended.
Other uses
Chlorobenzilate is being used for the control of mites on ornamental
plants. The compound is also used as a smoke for the treatment of
bee-hives against tracheal mites (Gubler, 1953).
Residues resulting from supervised trials
Residue data are available from supervised trials on several food
crops, grown under various conditions, using various rates of
application and pre-harvest intervals (Geigy, 1968). These data refer
to apples, citrus, nuts and vegetables (melons, cantaloupes). In most
cases normal dosage rates were applied in accordance with label
recommendations; in a few experiments higher dosages were included.
Results mainly refer to work in the United States of America. Only
limited data are available from other countries.
The following table summarizes typical residue data.
Rate of Post treatment Residue, ***
Crop application Number of interval whole fruit
(g/100 1) treatments (days) basis (ppm)
Apples 31 1 1 2.24
31 1 8 1.79
62 1 1 4.98
62 1 8 4.33
25 1 6 0.72
25 1 35 0.41
60 1 39 0.90
Grapefruit 30 1 1 1.10*
30 1 22 2.04*
Oranges 30 1 1 1.22*
30 1 22 1.99*
Grapes 60 1 17 0.62*
60 1 34 1.06*
120 1 17 1.20*
120 1 34 1.15*
Almonds 45 1-2 0-63 < 0.1
Melons 0.56** 1 1 < 0.04-0.4
0.56** 1 14 < 0.04-0.13
2.24** 1 1 0.08-1.1
2.24** 1 14 0.09-0.39
* Based on peel; analysis of pulp showed residues of less than 0.1 ppm
in the case of apples - no data on residues in pulp of grapes.
** kg/ha
*** UV method (Blinn and Gunther, 1963) except GLC (see below) for grapes,
almonds and melons.
Fate of residues
General comments
Chorobenzilate can be considered as a persistent compound. More
information is needed on the nature of terminal residues in plants,
animals and their products.
In soils
No information available.
In plants
Experiments have been carried out to examine if and to what extent
breakdown products derived from chlorobenzilate could be found, with
particular reference to the possible occurrence of
4,4'-dichlorobenzilic acid after apples were sprayed with 2 lbs/ 100
gal and samples were taken 3, 14 and 21 days. The sensitivity of the
analytical method was 0.1 ppm. In recovery studies,
4,4'-dichlorobenzilic acid could be detected without interference of
chlorobenzilate. It appears that chlorobenzilate is found mainly on
the outer surface of the treated apples. No unchanged chlorobenzilate
could be detected in the pulp of the apples. None of the treated
apples contained detectable amounts of 4,4'-dichlorobenzilic acid
(Murphy et al., 1966).
In animals
In experiments on sheep and cattle (Mattson and Schneller, 1966) 16
per cent protein mixed grain containing chlorobenzilate was given in
addition to a daily ration of mixed timothy and clover hay to eight
sheep and six cattle (both sexes) over a period of four weeks. The
dosage levels were 10, 24 and 80 mg chlorobenzilate per animal per day
for sheep and 40, 110 and 340 mg chlorobenzilate per animal for
cattle.
Residues of unchanged chlorobenzilate were only found in cattle at the
highest feeding level (340 mg/animal/day).
Residues in cattle
(ppm in fat)
Males Females
subcutaneous fat 0.54 0.32
omental fat 0.98 0.69
perirenal fat 0.54 0.69
In another experiment (Woodard, 1960) chlorobenzilate was administered
to six dairy cows at dietary levels of 20, 48 and 160 ppm in the feed
(intake 4 lbs of treated feed per day) for a period of 15 days. After
16 days, treatment with chlorobenzilate was terminated and the animals
were given the same type of feed, without chlorobenzilate, to permit
post-treatment analysis of milk samples. Milk samples were taken on
the second, fourth, sixth, ninth and fifteenth days of the experiment,
and again on the twentieth, twenty-first and twenty-second day.
Chlorobenzilate in the milk was found in one "high-level" cow (160
ppm) on the second day (0.15 ppm) and in one "medium-level" cow (48
ppm) on the fourth day (0.15 ppm). On the fifteenth day, residues did
not exceed 0.06 ppm. Since these levels are approaching the limit of
sensitivity of the analytical method used, there is no distinct
correlation between residue and feeding levels, and a number of
negative values were obtained, the few positive values may be
fortuitous. No 4,4'-dichlorobenzilic acid was found in the milk.
Limited recovery studies show that this compound, if present, would be
detected with the methods used.
In the above short-term feeding experiments rather high dosages have
been used, and more information is required on the possible occurrence
of residues in milk after feeding normal dosage levels for a prolonged
period.
In storage and processing
No data are available on the fate of residues during storage and
processing. Since the residue remains mainly on the outer side of
fruit and migrates only to a very small extent or not at all into the
pulp, it may be expected that washing and peeling of treated fruit
will remove most of the residue. No data are available on the effect
of cooking.
Evidence of residues in food in commerce or at consumption
Food moving in commerce
No information available.
Food at the time of consumption
In "market basket" or "total diet" studies (Cummings, 1966), carried
out by the United States Food and Drug Administration, multidetection
methods were used for residue analysis. The analytical procedure used
for all samples in this study enabled the detection of about 54
pesticide chemicals, including chlorobenzilate. So far no residues of
chlorobenzilate have been found.
Methods of residue analysis
Several methods are available for residue analysis of chlorobenzilate,
e.g. ultra-violet or infra-red spectrophotometry and gas-liquid
chromotography. In former years the ultra-violet method, as described
by Blinn and Gunther (1963) was used for determination of
chlorobenzilate residues on apples, pears and citrus fruit. In
addition Harris (1955) developed a modified Schechter-Haller infra-red
spectrophotometric method for residue analysis of chlorobenzilate.
This method was suitable for residue analysis on apples, pears and
strawberries. A modification of this method is described by Margot and
Stammbach (1964).
At present a gas chromatographic method appears to be most suitable
for use as a referee method, and should be developed for this purpose
through collaborative studies, in particular, the comparative
evaluation of the different detectors.
The following is a brief description of the gas chromatographic
determination of chlorobenzilate and chloropropylate in fruits,
vegetables and nuts. The method is sensitive to 0.05 ppm:
Chlorobenzilate and chloropropylate are extracted from the chopped
material with petroleum ether (boiling range 30-60°C). Part of the
extract is evaporated to dryness, taken up in benzene and transferred
to an alumina column (basic, activity grade V). Interfering materials
are removed by eluting with hexane or with 10 per cent benzene in
hexane. Chlorobenzilate and chloropropylate are eluted with
hexane/benzene (1:1) and the content is determined by gas
chromotography using a glass column (4 ft × 0.25 in) packed with five
per cent silicone gum GE XE - 60 (nitrile) supported on Anakrom ABS
(50-60 mesh) with nitrogen as the carrier gas and a microcoulometric
detector. Conditions: oven temperature, 215-220°C; block temperature,
220-225°C; furnace temperature, 810-820°C; nitrogen flow, 80 ml/min;
oxygen flow, 100 ml/min; pure nitrogen flow, 110 ml/min.
Conditioning of the column is important and can be achieved by three
to four injections of 30 micrograms of the acaricide. Retention times
are approximately 4 min. Separation of chlorobenzilate and
chloropropylate can be obtained by using a column loading of 10 per
cent XE-60 and a column length of 6 ft (retention times approximately
18 min for chlorobenzilate and 20 min for chloropropylate).
Apples, pears, stone fruits, citrus fruits, berries, nuts, vegetables
and cotton have been analysed for chlorobenzilate.
Apples, pears, stone fruits, citrus fruits, almonds, cucumbers and
beans have been analysed for chloropropylate.
Typical recovery data with fortified samples (sample weight 1-5 g)
are:
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Green beans 73-100
(continued)
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Maize (ears) 80-120
Maize (foliage) 80-95
Ground nuts 78-80
Citrus (peel) 89-110
Citrus (pulp) 70-80
Pears (peel) 90-100
Pears (pulp) 100-110
For oleagenous samples such as nuts, seeds and citrus peel, an
additional petroleum ether-acetonitrile partitioning step is
necessary.
Using the described clean-up procedure no interference from DDT and
DDD is encountered.
National tolerances
Country Crop Tolerance (ppm)
Canada apples, pears, citrus fruit,
cantaloupes 8
Netherlands fruit and vegetables 2
New Zealand fruit and vegetables 5
United States
of America apples, pears, citrus fruit,
melons 5
almonds, walnuts 0.2
almond hulls 15
cotton-seed 0.5
meat, fat and meat by-products 0.5
of cattle and sheep
(United States of America Federal Register)
RECOMMENDATIONS FOR TOLERANCES AND PRACTICAL RESIDUE LIMITS
Appraisal
Chlorobenzilate is a persistent and specific acaricide used on a
fairly wide range of crops. In many instances it is active against
mites which have developed resistance to other acaricides.
The compound is non-toxic to bees, and may be usefully applied in
integrated control schemes. Although the compound is registered in
many countries, little information was available on the extent of the
use in these countries. Many of the data furnished are based upon
experiments which have been conducted in the United States of America.
Furthermore, no information was available on the total composition of
the technical product.
The compound has not been detected in total diet studies in the United
States of America. There was a lack of data on the disappearance of
the compound and the disappearance of residues during storage and
processing. Information is required on the possible carry-over of the
residue into wine before tolerances for grapes can be recommended.
Although data were provided on residues in milk following feeding at
excessively high dosages to dairy cattle for a short period of time
(16 days), there were no data furnished on possible residues in milk
following the feeding of treated feed at normal residue levels for
longer periods. However, the residue occurring as the result of these
high-level feeding trials was at the limit of detection of the
analytical method (0.05 ppm) and it would not be expected that a
higher residue level would result from feeding lower levels for longer
periods in view of the fact that the compound is not cumulative.
In animals the compound is metabolized to form 4,4'-dichlorobenzilic
acid. No information is available on the nature of the terminal
residues in plants and in animal products.
A referee method of analysis has not been established, although
gas-liquid chromatography would seem to be most suitable for this
purpose, but must be further developed by collaborative studies.
Comparative evaluation of the different detectors used in gas
chromatographic methods, and evaluation of different methods of
extraction are needed.
Recommendations
The following temporary tolerances (to be in effect until 1972) are to
apply to raw agricultural products moving in commerce unless otherwise
indicated. In the case of fruits and vegetables, the tolerances should
be applied as soon as practicable after harvest and in any event prior
to actual retail to the public. In the case of commodities entering
international trade, the tolerances should be applied by the importing
country at the point of entry or as soon as practicable thereafter.
The tolerances are for the parent compound only.
apples, pears (whole fruit basis) 5.0 ppm
citrus (whole fruit basis) 1.0 ppm
melons, cantaloupes 1.0 ppm
almonds, walnuts 0.2 ppm
Further work or information
Required before 30 June 1972
1. Information on the composition of the technical product.
2. Information on the nature of terminal residues in plants, animals,
and their products.
3. Data from countries other than the United States of America on the
required rates and frequencies of application, pre-harvest intervals,
and the resultant residues.
4. Further data on the disappearance of residues in soils, in plants,
and in plant products during storage and processing.
5. Data on the possible carry-over of residues into wine as a result
of the treatment of grapes.
6. Further data on the occurrence of residues in milk after feeding
dairy cows at normal residue levels of the compound in the feed.
7. Comparative evaluation of the different detectors used in gas
liquid chromatographic methods and of different methods of extraction
for regulatory purposes.
Desirable
1. Collaborative studies to establish a referee method.
2. Metabolic studies in animals.
3. Investigation of possible testicular effects and long-term studies
in species other than the rat on the incidence of neoplasms.
REFERENCES
Blinn, R. C. and Gunther, F. A. (1963) The utilization of infrared and
ultraviolet spectrophotometric procedures for assay of pesticide
residues. Residue Reviews 2: 99-152
Cummings, J. G. (1966) Pesticides in the total diet. Residue Reviews
16: 30-45
Domenjoz, R. (1965a) Chlorbenzilat Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Bonn. Unpublished report.
Domenjoz, R. (1965b) Dichlorbenzilsäure Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Boon. Unpublished report.
FAO/WHO. (1965) Evaluation of the toxicity of pesticide residues in
food. FAO Mtg Rept No. PL:1965/10/1; WHO/Food Add./27.65
Gasser, R. (1952) Über zwei neue Akarizide aus der Gruppe der Di-(p-
chlorphenyl)-karbinole. Experientia, 8: 65-67
Geigy S.A., J.R. (1968) Chlorobenzilate working paper. Unpublished
report.
Gubler, W. et al. (1953) Schweizerische Bienenzeitung, 7: 268
Harris, H. J. (1955) Colorimetric determination of ethyl
4,4'-dichlorbenzilate (chlorobenzilate) as a spray residue. J. Agr.
Food Chem., 3: 939-941
Hazleton Laboratories Inc. (1953a) Geigy 338, technical. Chronic
feeding - rats. Unpublished report
Hazleton Laboratories Inc. (1953b) Geigy 338, technical feeding
studies - rats. Unpublished report
Hazleton Laboratories Inc. (1964) Method and development for the
analysis of chloropropylate and chlorobenzilate in biological fluids
and tissue specimens. Metabolic distribution and excretion of
chloropropylate and chlorobenzilate in dogs. Unpublished report
Hazleton Laboratories Inc. (1965) Chlorobenzilate. Two year dietary
feeding study - purebred beagles. Unpublished report
Horn, H. J., Bruce, R. B, and Paynter, O.E. (1955) Toxicology of
chlorobenzilate. J. Agr. Food Chem., 3: 752-756
Industrial Bio-Test Laboratories. (1965) Report to Geigy Chemical
Corp. Acute toxicity studies on chlorobenzilate 25W. Unpublished
report
Margot, A. and Stammbach, K. (1964) Chlorobenzilate; in: Zweig, G.,
1964. Analytical methods for pesticides, plant regulators and food
additives. Academic Press, London and New York, Vol. 2, 65-73
Mattson, A. M., Beaudoin, R. L. and Schneller, J. (1965) Comparison of
urinary metabolites in dogs after administration of chlorobenzilate or
chloropropylate. Analytical Dept. of Geigy Research Laboratories.
Unpublished report
Mattson, A. M. and Schneller, J. (1966) Chlorobenzilate residues in
sheep and cattle tissues. Unpublished report of the Analytical
Department of Geigy Research Laboratories, Division of Geigy Chem.
Corp., Ardsley, New York.
Murphy, R., Kahrs, R. and Mattson, A. M. (1966) Dissipation of
residues of chlorobenzilate and chloropropylate on apples. Unpublished
report of the Analytical Department of Geigy Research Laboratories,
Division of Geigy Chem. Corp., Ardsley, New York
United States of America Federal Register, 27, F.R. 12092, pav.
120-129
United States of America Department of Agriculture. (1966) Summary of
registered pesticide chemical cases. 1st October
Woodard. (1960) Examination of milk for residues following feeding of
chlorobenzilate to dairy cows. Unpublished report, Woodard Research
Corp.
Woodard. (1965a) Chlorobenzilate. Safety evaluation on fish and wild
life. (Bobwhite quail, mallard ducks, rainbow trout, sunfish,
goldfish, oysters). Unpublished report, Woodard Research Corp.
Woodard. (1965b) Chlorobenzilate. Three generation reproduction study
in the rat. Unpublished report, Woodard Research Corp.
Woodard. (1965c) Safety evaluation and determination of the amount of
chlorobenzilate that might appear in the tissues of sheep and
beef-cattle fed this material in the diet for four weeks. Unpublished
report, Woodard Research Corp.
Woodard. (1966) Chlorobenzilate. Safety evaluation by dietary feeding
to rats for 104 weeks. Unpublished report, Woodard Research Corp.
 Other information on identity and properties
The technical material consists of a yellow-brown oily liquid with a
purity of at least 90 per cent. No data have been submitted on the
composition of the technical product.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
Biochemical aspects
Three groups, each containing one male and one female dog, were given
0, 12.8, or 64.1 mg/kg/day for five consecutive days. At autopsy, five
days after the termination of treatment, no residues could be detected
in whole blood, liver, kidney, muscle, fat, or brain tissues. At 12.8
mg/kg/day, the male excreted 31 per cent of the total dose in the
urine within 10 days. Urinary excretion in the female accounted for 41
per cent. Faecal levels contained 5.6 per cent and 6.6 per cent for
the male and female respectively. At 64.1 mg/kg/day urinary and faecal
elimination was 31 per cent and 1.7 per cent respectively for the
male, and 23 per cent and 26 per cent for the female. The method of
analysis would not detect conjugates of 4,4'-dichlorobenzilic acid
which is suggested to be the major metabolite (Hazleton Laboratories
Inc., 1964). Dichlorobenzilic acid has been qualitatively identified
in dog urine by thin-layer chromatographic techniques (Mattson et al.,
1965).
Acute toxicity (oral)
Animal Route Compound or LD50 (mg/kg Reference
solvent body-weight)
Mouse Oral Suspension in 4 850 Gasser, 1952
gum arabic
Mouse Oral Technical 729 Horn et al., 1955
material
Rat Oral Suspension in 3 100 Gasser, 1952
gum arabic
Rat Oral Technical 702 Horn et al., 1955
material
Rat Oral 25 per cent. 735 Horn et al., 1955
xylene
emulsion
Short-term studies
Avian wildlife
Groups of 10 Mallard ducks (Aras platyrhynchus) were fed 0, 0.25,
0.45 and 0.8 per cent of chlorobenzilate for five consecutive days.
The LD50 was greater than 0.8 per cent. Body-weight gain and food
intake was slightly reduced in all test groups (Woodard, 1965a).
Groups of 10 quail (Colinus virginianus) were fed 0, 0.14, 0.25
and 0.5 per cent of chlorobenzilate in their diet for seven days. The
LD50 was 0.34 per cent. Bodyweight, food intake and gross pathology
of the survivors appeared unaffected (Woodard, 1965a).
Rat
Groups, each of 20 male rats, were fed 0, 40 and 800 ppm technical
chlorobenzilate in their diet for 48, 48 and 44 weeks, respectively.
Mortality was slightly higher in the 800 ppm group than in the control
group. The growth of the animals fed 800 ppm was retarded, and several
animals exhibited red or swollen eyelids and soft faeces. Organ to
body-weight ratios for the 40 ppm group were significantly greater
than for the controls, in the case of liver, kidney and testes.
Similar data for the 800 ppm group were not evaluated statistically,
but organ to body-weight ratios in the case of the liver and kidney
appear to be greater than for the controls. Gross pathological
examination revealed no changes attributable to compound
administration, but upon histological examination non-specific changes
were observed in the pancreas and adrenals of animals in both test
groups, and increased hemopoietic activity in the spleens of the 800
ppm groups. Tissue analysis indicated that chlorobenzilate was not
stored in the animal to any appreciable extent (Hazleton Laboratories,
1953a).
Groups of five male rats received 0, 1, 3 or 5 per cent technical
chlorobenzilate in the diet, and groups of five female rats received
0, or 3 per cent, in the diet. All animals receiving chlorobenzilate
at these levels died, those receiving one per cent within 59 days,
three per cent within eight days, and five per cent within six days.
Reduced food consumption, and marked loss of body-weight preceded
death.(Hazleton Laboratories, 1953b).
A 15 week feeding study, utilizing groups of five male rats at dose
levels of 0, 500, 1000, or 5000 ppm technical chlorobenzilate resulted
in reduced food consumption, and growth retardation in all test
groups. Mortality was 40 per cent at 1000 ppm, and 100 per cent at
5000 ppm (Hazleton Laboratories, 1953b).
In a 17 week feeding study using groups of 20 male rats fed 0, 50, and
500 ppm, and 20 female rats at 0, and 500 ppm, body weight gain was
depressed in the male only, at 500 ppm. Food intake was not
significantly depressed (Hazleton Laboratories, 1953b).
Groups of 20 male and 20 female rats treated for 99 days, with a 20
per cent chlorobenzilate powder mixed in their diet at dose levels of
0, 20, 100, 500, and 2500 ppm active ingredient did not show any toxic
effects as judged by food consumption, body-weight, mortality, organ
weights, gross, or histopathology except at the top dose level. At
2500 ppm active ingredient, food intake was reduced initially,
body-weight gain was reduced, absolute testes and spleen weights were
below control levels (insufficient data available to calculate organ
to body-weight ratios), and histopathological examination revealed
three rats with increased fatty deposits in the liver lobules, as well
as five rats with spermatogenentic injury, and testicular atrophy
(Domenjoz, 1965a).
Dog
Pairs of dogs (one male and one female) were fed 12.8, or 64.1 mg/kg
five times weekly for 35 weeks, except for two periods of five days
when excretion studies were performed. No toxic effects were apparent,
judging by growth, general appearance, hematology, urinalysis, liver
function test, and gross and microscopic pathology. The excretion
studies showed that the majority of administered material was excreted
in the urine, the metabolite having a 4,4'-dichlorodiphenyl methyl
structure. No chlorobenzilate was present in the urine three days
after withdrawal (Horn et al., 1955).
In a two year feeding study utilizing the 25 per cent powder
formulation, groups of three male and three female dogs were fed 0,
100, or 500 ppm active ingredient in the diet. A further group of
three male and three female dogs were fed 5000 ppm active ingredient
for 14 weeks. One male and one female were maintained at this dose
level for 20 weeks when they were sacrificed. The remainder, after a
five week withdrawal period, were replaced in the study by a 3000 ppm
dose level, until the completion of the test. At 100, and 500 ppm, the
parameters considered were comparable to the control group. At 3000
ppm, body-weight depression, mild anaemia and increased liver and
spleen to body-weight ratios were observed in both sexes. At 5000 ppm,
in addition to these effects, food intake was depressed, anaemia was
moderately severe, serum alkaline phosphatase values were elevated,
and the albumen to globulin ratio was reversed. The histopathology,
presumably performed at 104 weeks, indicates no compound related
effects at 100 and 500 ppm. At the top level, extramedullary
haematopoiesis was evident in liver and spleen as well as erythroid
hyperplasia of the bone marrow (Hazleton Laboratories Inc., 1965).
Sheep and beef-cattle
Sheep were exposed to chlorobenzilate 25 per cent wettable powder in
the diet for four weeks at dose levels of 0 (two females), 3.6 (two
females), 8.7 (two females) or 29 (one male and four females) ppm
active ingredient, and beef-cattle at dose levels of 0 (one male and
one female), 3.2 (one male and one female), 8.8 (one male and one
female), or 27 (one male and one female) ppm active ingredient. The
sheep and cattle failed to show compound related toxic symptoms as
judged by food consumption, bowel evacuations, physical condition and
behaviour, haematology, or gross necropsy observation. One cow dosed
at 3.2 ppm aborted a six-month foetus on day four. However, a second
cow dosed at 8.8 ppm carried a fully-developed foetus until sacrificed
(Woodard, 1965c).
Long-term studies
Rat
A two year study, using technical chlorobenzilate at 0 (20 male and 20
female), 50 (20 male) and 500 ppm (20 male and 20 female) showed
decreased body-weight gain, as well as unthriftiness, and blood tinged
crusts about the nose and eyes at 500 ppm. An increased incidence of
testicular atrophy was noted at both 50 and 500 ppm without
histological change (Horn et al., 1955).
A second two year study utilizing groups of 30 male and 30 female rats
at dose levels of 0, 40, 125 and 400 ppm active ingredient as the 25
per cent wettable formulation showed no significant changes in
body-weight gain, or food intake. Haematologic values did not show any
consistent changes related to dose level, or length of exposure to the
test material. Incidence of neoplasms was unrelated to the
administration of the test substances. Adverse effects on organ to
body-weight ratios were noted in the case of liver at 400 ppm. In the
histopathological examination of tissues, changes were observed in
liver, kidney and testes at 400 ppm, and in kidney and testes at 125
ppm. Gross autopsy observations indicated testicular changes at 125
and 400 ppm (Woodard, 1966).
Special studies
(a) Reproduction
A three generation study, utilizing 20 male and 20 female rats at 0,
and 50 ppm active ingredients as the 25 per cent wettable formulation
in the first generation, and, 10 male and 20 female rats at 0, 25 and
50 ppm active ingredient in the second and third generations failed to
reveal any toxic effects on adult body-weight, resorption rates, or
histology (performed on F1b adults). Absolute testes weights for the
F1b adults (the only group reported) showed a significant decrease at
50 ppm active ingredient. The litters all appeared normal as judged by
litter size, birth weight, stillbirths, survival to weaning, weanling
weights, and F3b weanling histopathology. Adult fertility, as judged
by number of litters per group appeared normal, but chlorobenzilate
withdrawal at weaning for 15 days, and at weaning for 28 days in F1b
and F2b weanlings, respectively, leaves this parameter open to
question. No teratogenic changes were observed at any stage in the
study (Woodard, 1965b).
(b) Studies on the metabolite
The metabolite, dichlorbenzilic acid was fed to groups of 20 male and
20 female rats for 99 days at dose levels of 0, 20, 100, 500 and 2500
ppm active ingredient in a 20 per cent powder. Food consumption
body-weight gain, mortality, organ weights, and gross and
histopathology showed no significant changes from the controls, except
possibly for a slight depression of kidney and testes weight at 2500
ppm (Domenjoz, 1965b).
Comments
Adequate data have been presented on short-term and long-term studies
in rats and dogs. Biochemical data on excretion, and a tentative
identification of the major metabolite are also available. All the
studies appear to be reliable and sufficiently adequate to permit
evaluation. The slight doubts regarding the incidence of neoplasms
observed in the long-term study on the rat makes it desirable to
obtain information from a second species.
TOXOCOLOGICAL EVALUATION
Level causing no significant toxicological effect
Rat: 40 ppm in the diet, equivalent to 2 mg/kg body-weight
Dog: 500 ppm in the dry diet, equivalent to 12.5 mg/kg
body-weight
Estimate of acceptable daily intake for man
0-0.02 mg/kg body-weight
RESIDUES IN FOOD AND THEIR EVALUATION
Use pattern
Pre-harvest treatments
Chlorobenzilate is used as a contact acaricide for the control of
adult spider mites; it is also effective against summer eggs and all
post-embryonic stages. The compound is used in many countries
throughout the world for the control of several mite species on
apples, pears, stone fruits, citrus fruits, soft fruit, grapes,
olives, vegetables, coffee, tea, cotton and ornamentals.
Chlorobenzilate is less effective at lower temperatures, and in
countries with a temperate climate, it is primarily used on
glass-house crops and only to a small extent outdoors. In view of its
specific acaricidal activity and its non-toxicity to bees, it may be
usefully applied in integrated control schemes. In certain instances
it is being recommended when resistance to other acaricides has
developed.
The recommended rates of application range from 25-50 g/100 l, and the
mount of spray liquid applied is generally 500-1000 l/ha for low-grown
crops (vegetables, etc) and 2000-2500 l/ha for high-grown crops (e.g.
fruit orchards). (U.S. Dept. of Agric., 1967)
Post-harvest treatments
No post-harvest treatment is recommended.
Other uses
Chlorobenzilate is being used for the control of mites on ornamental
plants. The compound is also used as a smoke for the treatment of
bee-hives against tracheal mites (Gubler, 1953).
Residues resulting from supervised trials
Residue data are available from supervised trials on several food
crops, grown under various conditions, using various rates of
application and pre-harvest intervals (Geigy, 1968). These data refer
to apples, citrus, nuts and vegetables (melons, cantaloupes). In most
cases normal dosage rates were applied in accordance with label
recommendations; in a few experiments higher dosages were included.
Results mainly refer to work in the United States of America. Only
limited data are available from other countries.
The following table summarizes typical residue data.
Rate of Post treatment Residue, ***
Crop application Number of interval whole fruit
(g/100 1) treatments (days) basis (ppm)
Apples 31 1 1 2.24
31 1 8 1.79
62 1 1 4.98
62 1 8 4.33
25 1 6 0.72
25 1 35 0.41
60 1 39 0.90
Grapefruit 30 1 1 1.10*
30 1 22 2.04*
Oranges 30 1 1 1.22*
30 1 22 1.99*
Grapes 60 1 17 0.62*
60 1 34 1.06*
120 1 17 1.20*
120 1 34 1.15*
Almonds 45 1-2 0-63 < 0.1
Melons 0.56** 1 1 < 0.04-0.4
0.56** 1 14 < 0.04-0.13
2.24** 1 1 0.08-1.1
2.24** 1 14 0.09-0.39
* Based on peel; analysis of pulp showed residues of less than 0.1 ppm
in the case of apples - no data on residues in pulp of grapes.
** kg/ha
*** UV method (Blinn and Gunther, 1963) except GLC (see below) for grapes,
almonds and melons.
Fate of residues
General comments
Chorobenzilate can be considered as a persistent compound. More
information is needed on the nature of terminal residues in plants,
animals and their products.
In soils
No information available.
In plants
Experiments have been carried out to examine if and to what extent
breakdown products derived from chlorobenzilate could be found, with
particular reference to the possible occurrence of
4,4'-dichlorobenzilic acid after apples were sprayed with 2 lbs/ 100
gal and samples were taken 3, 14 and 21 days. The sensitivity of the
analytical method was 0.1 ppm. In recovery studies,
4,4'-dichlorobenzilic acid could be detected without interference of
chlorobenzilate. It appears that chlorobenzilate is found mainly on
the outer surface of the treated apples. No unchanged chlorobenzilate
could be detected in the pulp of the apples. None of the treated
apples contained detectable amounts of 4,4'-dichlorobenzilic acid
(Murphy et al., 1966).
In animals
In experiments on sheep and cattle (Mattson and Schneller, 1966) 16
per cent protein mixed grain containing chlorobenzilate was given in
addition to a daily ration of mixed timothy and clover hay to eight
sheep and six cattle (both sexes) over a period of four weeks. The
dosage levels were 10, 24 and 80 mg chlorobenzilate per animal per day
for sheep and 40, 110 and 340 mg chlorobenzilate per animal for
cattle.
Residues of unchanged chlorobenzilate were only found in cattle at the
highest feeding level (340 mg/animal/day).
Residues in cattle
(ppm in fat)
Males Females
subcutaneous fat 0.54 0.32
omental fat 0.98 0.69
perirenal fat 0.54 0.69
In another experiment (Woodard, 1960) chlorobenzilate was administered
to six dairy cows at dietary levels of 20, 48 and 160 ppm in the feed
(intake 4 lbs of treated feed per day) for a period of 15 days. After
16 days, treatment with chlorobenzilate was terminated and the animals
were given the same type of feed, without chlorobenzilate, to permit
post-treatment analysis of milk samples. Milk samples were taken on
the second, fourth, sixth, ninth and fifteenth days of the experiment,
and again on the twentieth, twenty-first and twenty-second day.
Chlorobenzilate in the milk was found in one "high-level" cow (160
ppm) on the second day (0.15 ppm) and in one "medium-level" cow (48
ppm) on the fourth day (0.15 ppm). On the fifteenth day, residues did
not exceed 0.06 ppm. Since these levels are approaching the limit of
sensitivity of the analytical method used, there is no distinct
correlation between residue and feeding levels, and a number of
negative values were obtained, the few positive values may be
fortuitous. No 4,4'-dichlorobenzilic acid was found in the milk.
Limited recovery studies show that this compound, if present, would be
detected with the methods used.
In the above short-term feeding experiments rather high dosages have
been used, and more information is required on the possible occurrence
of residues in milk after feeding normal dosage levels for a prolonged
period.
In storage and processing
No data are available on the fate of residues during storage and
processing. Since the residue remains mainly on the outer side of
fruit and migrates only to a very small extent or not at all into the
pulp, it may be expected that washing and peeling of treated fruit
will remove most of the residue. No data are available on the effect
of cooking.
Evidence of residues in food in commerce or at consumption
Food moving in commerce
No information available.
Food at the time of consumption
In "market basket" or "total diet" studies (Cummings, 1966), carried
out by the United States Food and Drug Administration, multidetection
methods were used for residue analysis. The analytical procedure used
for all samples in this study enabled the detection of about 54
pesticide chemicals, including chlorobenzilate. So far no residues of
chlorobenzilate have been found.
Methods of residue analysis
Several methods are available for residue analysis of chlorobenzilate,
e.g. ultra-violet or infra-red spectrophotometry and gas-liquid
chromotography. In former years the ultra-violet method, as described
by Blinn and Gunther (1963) was used for determination of
chlorobenzilate residues on apples, pears and citrus fruit. In
addition Harris (1955) developed a modified Schechter-Haller infra-red
spectrophotometric method for residue analysis of chlorobenzilate.
This method was suitable for residue analysis on apples, pears and
strawberries. A modification of this method is described by Margot and
Stammbach (1964).
At present a gas chromatographic method appears to be most suitable
for use as a referee method, and should be developed for this purpose
through collaborative studies, in particular, the comparative
evaluation of the different detectors.
The following is a brief description of the gas chromatographic
determination of chlorobenzilate and chloropropylate in fruits,
vegetables and nuts. The method is sensitive to 0.05 ppm:
Chlorobenzilate and chloropropylate are extracted from the chopped
material with petroleum ether (boiling range 30-60°C). Part of the
extract is evaporated to dryness, taken up in benzene and transferred
to an alumina column (basic, activity grade V). Interfering materials
are removed by eluting with hexane or with 10 per cent benzene in
hexane. Chlorobenzilate and chloropropylate are eluted with
hexane/benzene (1:1) and the content is determined by gas
chromotography using a glass column (4 ft × 0.25 in) packed with five
per cent silicone gum GE XE - 60 (nitrile) supported on Anakrom ABS
(50-60 mesh) with nitrogen as the carrier gas and a microcoulometric
detector. Conditions: oven temperature, 215-220°C; block temperature,
220-225°C; furnace temperature, 810-820°C; nitrogen flow, 80 ml/min;
oxygen flow, 100 ml/min; pure nitrogen flow, 110 ml/min.
Conditioning of the column is important and can be achieved by three
to four injections of 30 micrograms of the acaricide. Retention times
are approximately 4 min. Separation of chlorobenzilate and
chloropropylate can be obtained by using a column loading of 10 per
cent XE-60 and a column length of 6 ft (retention times approximately
18 min for chlorobenzilate and 20 min for chloropropylate).
Apples, pears, stone fruits, citrus fruits, berries, nuts, vegetables
and cotton have been analysed for chlorobenzilate.
Apples, pears, stone fruits, citrus fruits, almonds, cucumbers and
beans have been analysed for chloropropylate.
Typical recovery data with fortified samples (sample weight 1-5 g)
are:
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Green beans 73-100
(continued)
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Maize (ears) 80-120
Maize (foliage) 80-95
Ground nuts 78-80
Citrus (peel) 89-110
Citrus (pulp) 70-80
Pears (peel) 90-100
Pears (pulp) 100-110
For oleagenous samples such as nuts, seeds and citrus peel, an
additional petroleum ether-acetonitrile partitioning step is
necessary.
Using the described clean-up procedure no interference from DDT and
DDD is encountered.
National tolerances
Country Crop Tolerance (ppm)
Canada apples, pears, citrus fruit,
cantaloupes 8
Netherlands fruit and vegetables 2
New Zealand fruit and vegetables 5
United States
of America apples, pears, citrus fruit,
melons 5
almonds, walnuts 0.2
almond hulls 15
cotton-seed 0.5
meat, fat and meat by-products 0.5
of cattle and sheep
(United States of America Federal Register)
RECOMMENDATIONS FOR TOLERANCES AND PRACTICAL RESIDUE LIMITS
Appraisal
Chlorobenzilate is a persistent and specific acaricide used on a
fairly wide range of crops. In many instances it is active against
mites which have developed resistance to other acaricides.
The compound is non-toxic to bees, and may be usefully applied in
integrated control schemes. Although the compound is registered in
many countries, little information was available on the extent of the
use in these countries. Many of the data furnished are based upon
experiments which have been conducted in the United States of America.
Furthermore, no information was available on the total composition of
the technical product.
The compound has not been detected in total diet studies in the United
States of America. There was a lack of data on the disappearance of
the compound and the disappearance of residues during storage and
processing. Information is required on the possible carry-over of the
residue into wine before tolerances for grapes can be recommended.
Although data were provided on residues in milk following feeding at
excessively high dosages to dairy cattle for a short period of time
(16 days), there were no data furnished on possible residues in milk
following the feeding of treated feed at normal residue levels for
longer periods. However, the residue occurring as the result of these
high-level feeding trials was at the limit of detection of the
analytical method (0.05 ppm) and it would not be expected that a
higher residue level would result from feeding lower levels for longer
periods in view of the fact that the compound is not cumulative.
In animals the compound is metabolized to form 4,4'-dichlorobenzilic
acid. No information is available on the nature of the terminal
residues in plants and in animal products.
A referee method of analysis has not been established, although
gas-liquid chromatography would seem to be most suitable for this
purpose, but must be further developed by collaborative studies.
Comparative evaluation of the different detectors used in gas
chromatographic methods, and evaluation of different methods of
extraction are needed.
Recommendations
The following temporary tolerances (to be in effect until 1972) are to
apply to raw agricultural products moving in commerce unless otherwise
indicated. In the case of fruits and vegetables, the tolerances should
be applied as soon as practicable after harvest and in any event prior
to actual retail to the public. In the case of commodities entering
international trade, the tolerances should be applied by the importing
country at the point of entry or as soon as practicable thereafter.
The tolerances are for the parent compound only.
apples, pears (whole fruit basis) 5.0 ppm
citrus (whole fruit basis) 1.0 ppm
melons, cantaloupes 1.0 ppm
almonds, walnuts 0.2 ppm
Further work or information
Required before 30 June 1972
1. Information on the composition of the technical product.
2. Information on the nature of terminal residues in plants, animals,
and their products.
3. Data from countries other than the United States of America on the
required rates and frequencies of application, pre-harvest intervals,
and the resultant residues.
4. Further data on the disappearance of residues in soils, in plants,
and in plant products during storage and processing.
5. Data on the possible carry-over of residues into wine as a result
of the treatment of grapes.
6. Further data on the occurrence of residues in milk after feeding
dairy cows at normal residue levels of the compound in the feed.
7. Comparative evaluation of the different detectors used in gas
liquid chromatographic methods and of different methods of extraction
for regulatory purposes.
Desirable
1. Collaborative studies to establish a referee method.
2. Metabolic studies in animals.
3. Investigation of possible testicular effects and long-term studies
in species other than the rat on the incidence of neoplasms.
REFERENCES
Blinn, R. C. and Gunther, F. A. (1963) The utilization of infrared and
ultraviolet spectrophotometric procedures for assay of pesticide
residues. Residue Reviews 2: 99-152
Cummings, J. G. (1966) Pesticides in the total diet. Residue Reviews
16: 30-45
Domenjoz, R. (1965a) Chlorbenzilat Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Bonn. Unpublished report.
Domenjoz, R. (1965b) Dichlorbenzilsäure Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Boon. Unpublished report.
FAO/WHO. (1965) Evaluation of the toxicity of pesticide residues in
food. FAO Mtg Rept No. PL:1965/10/1; WHO/Food Add./27.65
Gasser, R. (1952) Über zwei neue Akarizide aus der Gruppe der Di-(p-
chlorphenyl)-karbinole. Experientia, 8: 65-67
Geigy S.A., J.R. (1968) Chlorobenzilate working paper. Unpublished
report.
Gubler, W. et al. (1953) Schweizerische Bienenzeitung, 7: 268
Harris, H. J. (1955) Colorimetric determination of ethyl
4,4'-dichlorbenzilate (chlorobenzilate) as a spray residue. J. Agr.
Food Chem., 3: 939-941
Hazleton Laboratories Inc. (1953a) Geigy 338, technical. Chronic
feeding - rats. Unpublished report
Hazleton Laboratories Inc. (1953b) Geigy 338, technical feeding
studies - rats. Unpublished report
Hazleton Laboratories Inc. (1964) Method and development for the
analysis of chloropropylate and chlorobenzilate in biological fluids
and tissue specimens. Metabolic distribution and excretion of
chloropropylate and chlorobenzilate in dogs. Unpublished report
Hazleton Laboratories Inc. (1965) Chlorobenzilate. Two year dietary
feeding study - purebred beagles. Unpublished report
Horn, H. J., Bruce, R. B, and Paynter, O.E. (1955) Toxicology of
chlorobenzilate. J. Agr. Food Chem., 3: 752-756
Industrial Bio-Test Laboratories. (1965) Report to Geigy Chemical
Corp. Acute toxicity studies on chlorobenzilate 25W. Unpublished
report
Margot, A. and Stammbach, K. (1964) Chlorobenzilate; in: Zweig, G.,
1964. Analytical methods for pesticides, plant regulators and food
additives. Academic Press, London and New York, Vol. 2, 65-73
Mattson, A. M., Beaudoin, R. L. and Schneller, J. (1965) Comparison of
urinary metabolites in dogs after administration of chlorobenzilate or
chloropropylate. Analytical Dept. of Geigy Research Laboratories.
Unpublished report
Mattson, A. M. and Schneller, J. (1966) Chlorobenzilate residues in
sheep and cattle tissues. Unpublished report of the Analytical
Department of Geigy Research Laboratories, Division of Geigy Chem.
Corp., Ardsley, New York.
Murphy, R., Kahrs, R. and Mattson, A. M. (1966) Dissipation of
residues of chlorobenzilate and chloropropylate on apples. Unpublished
report of the Analytical Department of Geigy Research Laboratories,
Division of Geigy Chem. Corp., Ardsley, New York
United States of America Federal Register, 27, F.R. 12092, pav.
120-129
United States of America Department of Agriculture. (1966) Summary of
registered pesticide chemical cases. 1st October
Woodard. (1960) Examination of milk for residues following feeding of
chlorobenzilate to dairy cows. Unpublished report, Woodard Research
Corp.
Woodard. (1965a) Chlorobenzilate. Safety evaluation on fish and wild
life. (Bobwhite quail, mallard ducks, rainbow trout, sunfish,
goldfish, oysters). Unpublished report, Woodard Research Corp.
Woodard. (1965b) Chlorobenzilate. Three generation reproduction study
in the rat. Unpublished report, Woodard Research Corp.
Woodard. (1965c) Safety evaluation and determination of the amount of
chlorobenzilate that might appear in the tissues of sheep and
beef-cattle fed this material in the diet for four weeks. Unpublished
report, Woodard Research Corp.
Woodard. (1966) Chlorobenzilate. Safety evaluation by dietary feeding
to rats for 104 weeks. Unpublished report, Woodard Research Corp.
Other information on identity and properties
The technical material consists of a yellow-brown oily liquid with a
purity of at least 90 per cent. No data have been submitted on the
composition of the technical product.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
Biochemical aspects
Three groups, each containing one male and one female dog, were given
0, 12.8, or 64.1 mg/kg/day for five consecutive days. At autopsy, five
days after the termination of treatment, no residues could be detected
in whole blood, liver, kidney, muscle, fat, or brain tissues. At 12.8
mg/kg/day, the male excreted 31 per cent of the total dose in the
urine within 10 days. Urinary excretion in the female accounted for 41
per cent. Faecal levels contained 5.6 per cent and 6.6 per cent for
the male and female respectively. At 64.1 mg/kg/day urinary and faecal
elimination was 31 per cent and 1.7 per cent respectively for the
male, and 23 per cent and 26 per cent for the female. The method of
analysis would not detect conjugates of 4,4'-dichlorobenzilic acid
which is suggested to be the major metabolite (Hazleton Laboratories
Inc., 1964). Dichlorobenzilic acid has been qualitatively identified
in dog urine by thin-layer chromatographic techniques (Mattson et al.,
1965).
Acute toxicity (oral)
Animal Route Compound or LD50 (mg/kg Reference
solvent body-weight)
Mouse Oral Suspension in 4 850 Gasser, 1952
gum arabic
Mouse Oral Technical 729 Horn et al., 1955
material
Rat Oral Suspension in 3 100 Gasser, 1952
gum arabic
Rat Oral Technical 702 Horn et al., 1955
material
Rat Oral 25 per cent. 735 Horn et al., 1955
xylene
emulsion
Short-term studies
Avian wildlife
Groups of 10 Mallard ducks (Aras platyrhynchus) were fed 0, 0.25,
0.45 and 0.8 per cent of chlorobenzilate for five consecutive days.
The LD50 was greater than 0.8 per cent. Body-weight gain and food
intake was slightly reduced in all test groups (Woodard, 1965a).
Groups of 10 quail (Colinus virginianus) were fed 0, 0.14, 0.25
and 0.5 per cent of chlorobenzilate in their diet for seven days. The
LD50 was 0.34 per cent. Bodyweight, food intake and gross pathology
of the survivors appeared unaffected (Woodard, 1965a).
Rat
Groups, each of 20 male rats, were fed 0, 40 and 800 ppm technical
chlorobenzilate in their diet for 48, 48 and 44 weeks, respectively.
Mortality was slightly higher in the 800 ppm group than in the control
group. The growth of the animals fed 800 ppm was retarded, and several
animals exhibited red or swollen eyelids and soft faeces. Organ to
body-weight ratios for the 40 ppm group were significantly greater
than for the controls, in the case of liver, kidney and testes.
Similar data for the 800 ppm group were not evaluated statistically,
but organ to body-weight ratios in the case of the liver and kidney
appear to be greater than for the controls. Gross pathological
examination revealed no changes attributable to compound
administration, but upon histological examination non-specific changes
were observed in the pancreas and adrenals of animals in both test
groups, and increased hemopoietic activity in the spleens of the 800
ppm groups. Tissue analysis indicated that chlorobenzilate was not
stored in the animal to any appreciable extent (Hazleton Laboratories,
1953a).
Groups of five male rats received 0, 1, 3 or 5 per cent technical
chlorobenzilate in the diet, and groups of five female rats received
0, or 3 per cent, in the diet. All animals receiving chlorobenzilate
at these levels died, those receiving one per cent within 59 days,
three per cent within eight days, and five per cent within six days.
Reduced food consumption, and marked loss of body-weight preceded
death.(Hazleton Laboratories, 1953b).
A 15 week feeding study, utilizing groups of five male rats at dose
levels of 0, 500, 1000, or 5000 ppm technical chlorobenzilate resulted
in reduced food consumption, and growth retardation in all test
groups. Mortality was 40 per cent at 1000 ppm, and 100 per cent at
5000 ppm (Hazleton Laboratories, 1953b).
In a 17 week feeding study using groups of 20 male rats fed 0, 50, and
500 ppm, and 20 female rats at 0, and 500 ppm, body weight gain was
depressed in the male only, at 500 ppm. Food intake was not
significantly depressed (Hazleton Laboratories, 1953b).
Groups of 20 male and 20 female rats treated for 99 days, with a 20
per cent chlorobenzilate powder mixed in their diet at dose levels of
0, 20, 100, 500, and 2500 ppm active ingredient did not show any toxic
effects as judged by food consumption, body-weight, mortality, organ
weights, gross, or histopathology except at the top dose level. At
2500 ppm active ingredient, food intake was reduced initially,
body-weight gain was reduced, absolute testes and spleen weights were
below control levels (insufficient data available to calculate organ
to body-weight ratios), and histopathological examination revealed
three rats with increased fatty deposits in the liver lobules, as well
as five rats with spermatogenentic injury, and testicular atrophy
(Domenjoz, 1965a).
Dog
Pairs of dogs (one male and one female) were fed 12.8, or 64.1 mg/kg
five times weekly for 35 weeks, except for two periods of five days
when excretion studies were performed. No toxic effects were apparent,
judging by growth, general appearance, hematology, urinalysis, liver
function test, and gross and microscopic pathology. The excretion
studies showed that the majority of administered material was excreted
in the urine, the metabolite having a 4,4'-dichlorodiphenyl methyl
structure. No chlorobenzilate was present in the urine three days
after withdrawal (Horn et al., 1955).
In a two year feeding study utilizing the 25 per cent powder
formulation, groups of three male and three female dogs were fed 0,
100, or 500 ppm active ingredient in the diet. A further group of
three male and three female dogs were fed 5000 ppm active ingredient
for 14 weeks. One male and one female were maintained at this dose
level for 20 weeks when they were sacrificed. The remainder, after a
five week withdrawal period, were replaced in the study by a 3000 ppm
dose level, until the completion of the test. At 100, and 500 ppm, the
parameters considered were comparable to the control group. At 3000
ppm, body-weight depression, mild anaemia and increased liver and
spleen to body-weight ratios were observed in both sexes. At 5000 ppm,
in addition to these effects, food intake was depressed, anaemia was
moderately severe, serum alkaline phosphatase values were elevated,
and the albumen to globulin ratio was reversed. The histopathology,
presumably performed at 104 weeks, indicates no compound related
effects at 100 and 500 ppm. At the top level, extramedullary
haematopoiesis was evident in liver and spleen as well as erythroid
hyperplasia of the bone marrow (Hazleton Laboratories Inc., 1965).
Sheep and beef-cattle
Sheep were exposed to chlorobenzilate 25 per cent wettable powder in
the diet for four weeks at dose levels of 0 (two females), 3.6 (two
females), 8.7 (two females) or 29 (one male and four females) ppm
active ingredient, and beef-cattle at dose levels of 0 (one male and
one female), 3.2 (one male and one female), 8.8 (one male and one
female), or 27 (one male and one female) ppm active ingredient. The
sheep and cattle failed to show compound related toxic symptoms as
judged by food consumption, bowel evacuations, physical condition and
behaviour, haematology, or gross necropsy observation. One cow dosed
at 3.2 ppm aborted a six-month foetus on day four. However, a second
cow dosed at 8.8 ppm carried a fully-developed foetus until sacrificed
(Woodard, 1965c).
Long-term studies
Rat
A two year study, using technical chlorobenzilate at 0 (20 male and 20
female), 50 (20 male) and 500 ppm (20 male and 20 female) showed
decreased body-weight gain, as well as unthriftiness, and blood tinged
crusts about the nose and eyes at 500 ppm. An increased incidence of
testicular atrophy was noted at both 50 and 500 ppm without
histological change (Horn et al., 1955).
A second two year study utilizing groups of 30 male and 30 female rats
at dose levels of 0, 40, 125 and 400 ppm active ingredient as the 25
per cent wettable formulation showed no significant changes in
body-weight gain, or food intake. Haematologic values did not show any
consistent changes related to dose level, or length of exposure to the
test material. Incidence of neoplasms was unrelated to the
administration of the test substances. Adverse effects on organ to
body-weight ratios were noted in the case of liver at 400 ppm. In the
histopathological examination of tissues, changes were observed in
liver, kidney and testes at 400 ppm, and in kidney and testes at 125
ppm. Gross autopsy observations indicated testicular changes at 125
and 400 ppm (Woodard, 1966).
Special studies
(a) Reproduction
A three generation study, utilizing 20 male and 20 female rats at 0,
and 50 ppm active ingredients as the 25 per cent wettable formulation
in the first generation, and, 10 male and 20 female rats at 0, 25 and
50 ppm active ingredient in the second and third generations failed to
reveal any toxic effects on adult body-weight, resorption rates, or
histology (performed on F1b adults). Absolute testes weights for the
F1b adults (the only group reported) showed a significant decrease at
50 ppm active ingredient. The litters all appeared normal as judged by
litter size, birth weight, stillbirths, survival to weaning, weanling
weights, and F3b weanling histopathology. Adult fertility, as judged
by number of litters per group appeared normal, but chlorobenzilate
withdrawal at weaning for 15 days, and at weaning for 28 days in F1b
and F2b weanlings, respectively, leaves this parameter open to
question. No teratogenic changes were observed at any stage in the
study (Woodard, 1965b).
(b) Studies on the metabolite
The metabolite, dichlorbenzilic acid was fed to groups of 20 male and
20 female rats for 99 days at dose levels of 0, 20, 100, 500 and 2500
ppm active ingredient in a 20 per cent powder. Food consumption
body-weight gain, mortality, organ weights, and gross and
histopathology showed no significant changes from the controls, except
possibly for a slight depression of kidney and testes weight at 2500
ppm (Domenjoz, 1965b).
Comments
Adequate data have been presented on short-term and long-term studies
in rats and dogs. Biochemical data on excretion, and a tentative
identification of the major metabolite are also available. All the
studies appear to be reliable and sufficiently adequate to permit
evaluation. The slight doubts regarding the incidence of neoplasms
observed in the long-term study on the rat makes it desirable to
obtain information from a second species.
TOXOCOLOGICAL EVALUATION
Level causing no significant toxicological effect
Rat: 40 ppm in the diet, equivalent to 2 mg/kg body-weight
Dog: 500 ppm in the dry diet, equivalent to 12.5 mg/kg
body-weight
Estimate of acceptable daily intake for man
0-0.02 mg/kg body-weight
RESIDUES IN FOOD AND THEIR EVALUATION
Use pattern
Pre-harvest treatments
Chlorobenzilate is used as a contact acaricide for the control of
adult spider mites; it is also effective against summer eggs and all
post-embryonic stages. The compound is used in many countries
throughout the world for the control of several mite species on
apples, pears, stone fruits, citrus fruits, soft fruit, grapes,
olives, vegetables, coffee, tea, cotton and ornamentals.
Chlorobenzilate is less effective at lower temperatures, and in
countries with a temperate climate, it is primarily used on
glass-house crops and only to a small extent outdoors. In view of its
specific acaricidal activity and its non-toxicity to bees, it may be
usefully applied in integrated control schemes. In certain instances
it is being recommended when resistance to other acaricides has
developed.
The recommended rates of application range from 25-50 g/100 l, and the
mount of spray liquid applied is generally 500-1000 l/ha for low-grown
crops (vegetables, etc) and 2000-2500 l/ha for high-grown crops (e.g.
fruit orchards). (U.S. Dept. of Agric., 1967)
Post-harvest treatments
No post-harvest treatment is recommended.
Other uses
Chlorobenzilate is being used for the control of mites on ornamental
plants. The compound is also used as a smoke for the treatment of
bee-hives against tracheal mites (Gubler, 1953).
Residues resulting from supervised trials
Residue data are available from supervised trials on several food
crops, grown under various conditions, using various rates of
application and pre-harvest intervals (Geigy, 1968). These data refer
to apples, citrus, nuts and vegetables (melons, cantaloupes). In most
cases normal dosage rates were applied in accordance with label
recommendations; in a few experiments higher dosages were included.
Results mainly refer to work in the United States of America. Only
limited data are available from other countries.
The following table summarizes typical residue data.
Rate of Post treatment Residue, ***
Crop application Number of interval whole fruit
(g/100 1) treatments (days) basis (ppm)
Apples 31 1 1 2.24
31 1 8 1.79
62 1 1 4.98
62 1 8 4.33
25 1 6 0.72
25 1 35 0.41
60 1 39 0.90
Grapefruit 30 1 1 1.10*
30 1 22 2.04*
Oranges 30 1 1 1.22*
30 1 22 1.99*
Grapes 60 1 17 0.62*
60 1 34 1.06*
120 1 17 1.20*
120 1 34 1.15*
Almonds 45 1-2 0-63 < 0.1
Melons 0.56** 1 1 < 0.04-0.4
0.56** 1 14 < 0.04-0.13
2.24** 1 1 0.08-1.1
2.24** 1 14 0.09-0.39
* Based on peel; analysis of pulp showed residues of less than 0.1 ppm
in the case of apples - no data on residues in pulp of grapes.
** kg/ha
*** UV method (Blinn and Gunther, 1963) except GLC (see below) for grapes,
almonds and melons.
Fate of residues
General comments
Chorobenzilate can be considered as a persistent compound. More
information is needed on the nature of terminal residues in plants,
animals and their products.
In soils
No information available.
In plants
Experiments have been carried out to examine if and to what extent
breakdown products derived from chlorobenzilate could be found, with
particular reference to the possible occurrence of
4,4'-dichlorobenzilic acid after apples were sprayed with 2 lbs/ 100
gal and samples were taken 3, 14 and 21 days. The sensitivity of the
analytical method was 0.1 ppm. In recovery studies,
4,4'-dichlorobenzilic acid could be detected without interference of
chlorobenzilate. It appears that chlorobenzilate is found mainly on
the outer surface of the treated apples. No unchanged chlorobenzilate
could be detected in the pulp of the apples. None of the treated
apples contained detectable amounts of 4,4'-dichlorobenzilic acid
(Murphy et al., 1966).
In animals
In experiments on sheep and cattle (Mattson and Schneller, 1966) 16
per cent protein mixed grain containing chlorobenzilate was given in
addition to a daily ration of mixed timothy and clover hay to eight
sheep and six cattle (both sexes) over a period of four weeks. The
dosage levels were 10, 24 and 80 mg chlorobenzilate per animal per day
for sheep and 40, 110 and 340 mg chlorobenzilate per animal for
cattle.
Residues of unchanged chlorobenzilate were only found in cattle at the
highest feeding level (340 mg/animal/day).
Residues in cattle
(ppm in fat)
Males Females
subcutaneous fat 0.54 0.32
omental fat 0.98 0.69
perirenal fat 0.54 0.69
In another experiment (Woodard, 1960) chlorobenzilate was administered
to six dairy cows at dietary levels of 20, 48 and 160 ppm in the feed
(intake 4 lbs of treated feed per day) for a period of 15 days. After
16 days, treatment with chlorobenzilate was terminated and the animals
were given the same type of feed, without chlorobenzilate, to permit
post-treatment analysis of milk samples. Milk samples were taken on
the second, fourth, sixth, ninth and fifteenth days of the experiment,
and again on the twentieth, twenty-first and twenty-second day.
Chlorobenzilate in the milk was found in one "high-level" cow (160
ppm) on the second day (0.15 ppm) and in one "medium-level" cow (48
ppm) on the fourth day (0.15 ppm). On the fifteenth day, residues did
not exceed 0.06 ppm. Since these levels are approaching the limit of
sensitivity of the analytical method used, there is no distinct
correlation between residue and feeding levels, and a number of
negative values were obtained, the few positive values may be
fortuitous. No 4,4'-dichlorobenzilic acid was found in the milk.
Limited recovery studies show that this compound, if present, would be
detected with the methods used.
In the above short-term feeding experiments rather high dosages have
been used, and more information is required on the possible occurrence
of residues in milk after feeding normal dosage levels for a prolonged
period.
In storage and processing
No data are available on the fate of residues during storage and
processing. Since the residue remains mainly on the outer side of
fruit and migrates only to a very small extent or not at all into the
pulp, it may be expected that washing and peeling of treated fruit
will remove most of the residue. No data are available on the effect
of cooking.
Evidence of residues in food in commerce or at consumption
Food moving in commerce
No information available.
Food at the time of consumption
In "market basket" or "total diet" studies (Cummings, 1966), carried
out by the United States Food and Drug Administration, multidetection
methods were used for residue analysis. The analytical procedure used
for all samples in this study enabled the detection of about 54
pesticide chemicals, including chlorobenzilate. So far no residues of
chlorobenzilate have been found.
Methods of residue analysis
Several methods are available for residue analysis of chlorobenzilate,
e.g. ultra-violet or infra-red spectrophotometry and gas-liquid
chromotography. In former years the ultra-violet method, as described
by Blinn and Gunther (1963) was used for determination of
chlorobenzilate residues on apples, pears and citrus fruit. In
addition Harris (1955) developed a modified Schechter-Haller infra-red
spectrophotometric method for residue analysis of chlorobenzilate.
This method was suitable for residue analysis on apples, pears and
strawberries. A modification of this method is described by Margot and
Stammbach (1964).
At present a gas chromatographic method appears to be most suitable
for use as a referee method, and should be developed for this purpose
through collaborative studies, in particular, the comparative
evaluation of the different detectors.
The following is a brief description of the gas chromatographic
determination of chlorobenzilate and chloropropylate in fruits,
vegetables and nuts. The method is sensitive to 0.05 ppm:
Chlorobenzilate and chloropropylate are extracted from the chopped
material with petroleum ether (boiling range 30-60°C). Part of the
extract is evaporated to dryness, taken up in benzene and transferred
to an alumina column (basic, activity grade V). Interfering materials
are removed by eluting with hexane or with 10 per cent benzene in
hexane. Chlorobenzilate and chloropropylate are eluted with
hexane/benzene (1:1) and the content is determined by gas
chromotography using a glass column (4 ft × 0.25 in) packed with five
per cent silicone gum GE XE - 60 (nitrile) supported on Anakrom ABS
(50-60 mesh) with nitrogen as the carrier gas and a microcoulometric
detector. Conditions: oven temperature, 215-220°C; block temperature,
220-225°C; furnace temperature, 810-820°C; nitrogen flow, 80 ml/min;
oxygen flow, 100 ml/min; pure nitrogen flow, 110 ml/min.
Conditioning of the column is important and can be achieved by three
to four injections of 30 micrograms of the acaricide. Retention times
are approximately 4 min. Separation of chlorobenzilate and
chloropropylate can be obtained by using a column loading of 10 per
cent XE-60 and a column length of 6 ft (retention times approximately
18 min for chlorobenzilate and 20 min for chloropropylate).
Apples, pears, stone fruits, citrus fruits, berries, nuts, vegetables
and cotton have been analysed for chlorobenzilate.
Apples, pears, stone fruits, citrus fruits, almonds, cucumbers and
beans have been analysed for chloropropylate.
Typical recovery data with fortified samples (sample weight 1-5 g)
are:
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Green beans 73-100
(continued)
Per cent recovery
Crop
Chlorobenzilate Chloropropylate
Maize (ears) 80-120
Maize (foliage) 80-95
Ground nuts 78-80
Citrus (peel) 89-110
Citrus (pulp) 70-80
Pears (peel) 90-100
Pears (pulp) 100-110
For oleagenous samples such as nuts, seeds and citrus peel, an
additional petroleum ether-acetonitrile partitioning step is
necessary.
Using the described clean-up procedure no interference from DDT and
DDD is encountered.
National tolerances
Country Crop Tolerance (ppm)
Canada apples, pears, citrus fruit,
cantaloupes 8
Netherlands fruit and vegetables 2
New Zealand fruit and vegetables 5
United States
of America apples, pears, citrus fruit,
melons 5
almonds, walnuts 0.2
almond hulls 15
cotton-seed 0.5
meat, fat and meat by-products 0.5
of cattle and sheep
(United States of America Federal Register)
RECOMMENDATIONS FOR TOLERANCES AND PRACTICAL RESIDUE LIMITS
Appraisal
Chlorobenzilate is a persistent and specific acaricide used on a
fairly wide range of crops. In many instances it is active against
mites which have developed resistance to other acaricides.
The compound is non-toxic to bees, and may be usefully applied in
integrated control schemes. Although the compound is registered in
many countries, little information was available on the extent of the
use in these countries. Many of the data furnished are based upon
experiments which have been conducted in the United States of America.
Furthermore, no information was available on the total composition of
the technical product.
The compound has not been detected in total diet studies in the United
States of America. There was a lack of data on the disappearance of
the compound and the disappearance of residues during storage and
processing. Information is required on the possible carry-over of the
residue into wine before tolerances for grapes can be recommended.
Although data were provided on residues in milk following feeding at
excessively high dosages to dairy cattle for a short period of time
(16 days), there were no data furnished on possible residues in milk
following the feeding of treated feed at normal residue levels for
longer periods. However, the residue occurring as the result of these
high-level feeding trials was at the limit of detection of the
analytical method (0.05 ppm) and it would not be expected that a
higher residue level would result from feeding lower levels for longer
periods in view of the fact that the compound is not cumulative.
In animals the compound is metabolized to form 4,4'-dichlorobenzilic
acid. No information is available on the nature of the terminal
residues in plants and in animal products.
A referee method of analysis has not been established, although
gas-liquid chromatography would seem to be most suitable for this
purpose, but must be further developed by collaborative studies.
Comparative evaluation of the different detectors used in gas
chromatographic methods, and evaluation of different methods of
extraction are needed.
Recommendations
The following temporary tolerances (to be in effect until 1972) are to
apply to raw agricultural products moving in commerce unless otherwise
indicated. In the case of fruits and vegetables, the tolerances should
be applied as soon as practicable after harvest and in any event prior
to actual retail to the public. In the case of commodities entering
international trade, the tolerances should be applied by the importing
country at the point of entry or as soon as practicable thereafter.
The tolerances are for the parent compound only.
apples, pears (whole fruit basis) 5.0 ppm
citrus (whole fruit basis) 1.0 ppm
melons, cantaloupes 1.0 ppm
almonds, walnuts 0.2 ppm
Further work or information
Required before 30 June 1972
1. Information on the composition of the technical product.
2. Information on the nature of terminal residues in plants, animals,
and their products.
3. Data from countries other than the United States of America on the
required rates and frequencies of application, pre-harvest intervals,
and the resultant residues.
4. Further data on the disappearance of residues in soils, in plants,
and in plant products during storage and processing.
5. Data on the possible carry-over of residues into wine as a result
of the treatment of grapes.
6. Further data on the occurrence of residues in milk after feeding
dairy cows at normal residue levels of the compound in the feed.
7. Comparative evaluation of the different detectors used in gas
liquid chromatographic methods and of different methods of extraction
for regulatory purposes.
Desirable
1. Collaborative studies to establish a referee method.
2. Metabolic studies in animals.
3. Investigation of possible testicular effects and long-term studies
in species other than the rat on the incidence of neoplasms.
REFERENCES
Blinn, R. C. and Gunther, F. A. (1963) The utilization of infrared and
ultraviolet spectrophotometric procedures for assay of pesticide
residues. Residue Reviews 2: 99-152
Cummings, J. G. (1966) Pesticides in the total diet. Residue Reviews
16: 30-45
Domenjoz, R. (1965a) Chlorbenzilat Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Bonn. Unpublished report.
Domenjoz, R. (1965b) Dichlorbenzilsäure Toxizität bei chronischer
Verabreichung. Institute of Pharmacology, Rheinische
Friedrich-Wilhelm's University, Boon. Unpublished report.
FAO/WHO. (1965) Evaluation of the toxicity of pesticide residues in
food. FAO Mtg Rept No. PL:1965/10/1; WHO/Food Add./27.65
Gasser, R. (1952) Über zwei neue Akarizide aus der Gruppe der Di-(p-
chlorphenyl)-karbinole. Experientia, 8: 65-67
Geigy S.A., J.R. (1968) Chlorobenzilate working paper. Unpublished
report.
Gubler, W. et al. (1953) Schweizerische Bienenzeitung, 7: 268
Harris, H. J. (1955) Colorimetric determination of ethyl
4,4'-dichlorbenzilate (chlorobenzilate) as a spray residue. J. Agr.
Food Chem., 3: 939-941
Hazleton Laboratories Inc. (1953a) Geigy 338, technical. Chronic
feeding - rats. Unpublished report
Hazleton Laboratories Inc. (1953b) Geigy 338, technical feeding
studies - rats. Unpublished report
Hazleton Laboratories Inc. (1964) Method and development for the
analysis of chloropropylate and chlorobenzilate in biological fluids
and tissue specimens. Metabolic distribution and excretion of
chloropropylate and chlorobenzilate in dogs. Unpublished report
Hazleton Laboratories Inc. (1965) Chlorobenzilate. Two year dietary
feeding study - purebred beagles. Unpublished report
Horn, H. J., Bruce, R. B, and Paynter, O.E. (1955) Toxicology of
chlorobenzilate. J. Agr. Food Chem., 3: 752-756
Industrial Bio-Test Laboratories. (1965) Report to Geigy Chemical
Corp. Acute toxicity studies on chlorobenzilate 25W. Unpublished
report
Margot, A. and Stammbach, K. (1964) Chlorobenzilate; in: Zweig, G.,
1964. Analytical methods for pesticides, plant regulators and food
additives. Academic Press, London and New York, Vol. 2, 65-73
Mattson, A. M., Beaudoin, R. L. and Schneller, J. (1965) Comparison of
urinary metabolites in dogs after administration of chlorobenzilate or
chloropropylate. Analytical Dept. of Geigy Research Laboratories.
Unpublished report
Mattson, A. M. and Schneller, J. (1966) Chlorobenzilate residues in
sheep and cattle tissues. Unpublished report of the Analytical
Department of Geigy Research Laboratories, Division of Geigy Chem.
Corp., Ardsley, New York.
Murphy, R., Kahrs, R. and Mattson, A. M. (1966) Dissipation of
residues of chlorobenzilate and chloropropylate on apples. Unpublished
report of the Analytical Department of Geigy Research Laboratories,
Division of Geigy Chem. Corp., Ardsley, New York
United States of America Federal Register, 27, F.R. 12092, pav.
120-129
United States of America Department of Agriculture. (1966) Summary of
registered pesticide chemical cases. 1st October
Woodard. (1960) Examination of milk for residues following feeding of
chlorobenzilate to dairy cows. Unpublished report, Woodard Research
Corp.
Woodard. (1965a) Chlorobenzilate. Safety evaluation on fish and wild
life. (Bobwhite quail, mallard ducks, rainbow trout, sunfish,
goldfish, oysters). Unpublished report, Woodard Research Corp.
Woodard. (1965b) Chlorobenzilate. Three generation reproduction study
in the rat. Unpublished report, Woodard Research Corp.
Woodard. (1965c) Safety evaluation and determination of the amount of
chlorobenzilate that might appear in the tissues of sheep and
beef-cattle fed this material in the diet for four weeks. Unpublished
report, Woodard Research Corp.
Woodard. (1966) Chlorobenzilate. Safety evaluation by dietary feeding
to rats for 104 weeks. Unpublished report, Woodard Research Corp.
