
FAO Meeting Report No. PL/1965/10/1
WHO/Food Add./27.65
EVALUATION OF THE TOXICITY OF PESTICIDE RESIDUES IN FOOD
The content of this document is the result of the deliberations of the
Joint Meeting of the FAO Committee on Pesticides in Agriculture and
the WHO Expert Committee on Pesticide Residues, which met in Rome,
15-22 March 19651
Food and Agriculture Organization of the United Nations
World Health Organization
1965
1 Report of the second joint meeting of the FAO Committee on
Pesticides in Agriculture and the WHO Expert Committee on Pesticide
Residues, FAO Meeting Report No. PL/1965/10; WHO/Food Add./26.65
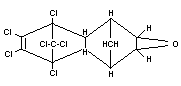
ENDRIN
Chemical name
1,2,3,4,10-hexachloro-6,7-epoxy-1,4,4a,5,6,7,8,8a-octahydro-endo-
1,4-endo-5,8,-dimethanonaphthalene;
1,2,3,4,10,10-hexachloro-6,7-epoxy-1,4,4a-5,6,7,8,8a-octahydro-exo
1,4-exo-5,8-dimethanonaphthalene
Empirical formula
C12H8OCl6
Structural formula
 BIOLOGICAL DATA
Biochemical aspects
Endrin fed to animals is stored unchanged in the tissues
(Kiigemagi et al., 1958, Street et al., 1957; Terriere et al., 1958;
Treon et al., 1955) and excreted in milk and eggs (Ely et al., 1957;
Street et al., 1957, Terriere et al., 1958). The chief organ of
storage is the fat, with a storage ratio of about 15 when the
concentration in the diet is 1 ppm (Lehman, 1956). Endrin is slowly
excreted from the tissues (Nelson et al., 1956. Zavon, 1961).
Serum alkaline phosphatase levels in rats increased as the
concentration of endrin in the diet was increased from 1 ppm to 100
ppm (Nelson et al., 1956).
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Adult rat (female) Oral 7.3 Treon et al., 1955
Young rat (female) Oral 16.8 Treon et al., 1955
Adult rat (male) Oral 40-43.4 Speck & Maaske, 1958
Treon et al., 1955
(cont'd)
Animal Route LD50 mg/kg References
body-weight
Young rat (male) Oral 28.8 Treon et al., 1955
Rabbit (female) Oral 7-10 Treon et al., 1955
Guinea-pig Oral approx. 16-36 Treon et al., 1955
Monkey Oral approx. 3 Treon et al., 1955
Man. In one incident 59 people became ill from the ingestion of
bread containing 150 ppm of endrin, but there were no fatalities
(Davies & Lewis, 1956). The maximum amount consumed was estimated to
have been 1 mg/kg (Zavon, 1961).
Short-term studies
Rat. Adult rats, 5 males and 5 females per group, were given
diets containing 1, 5, 25, 50 and 100 ppm of endrin for 16 weeks. Only
2 rats on 50 ppm and 3 on 25 ppm survived. Three male rats died in the
group on 5 ppm. There was loss of weight and hypersensitivity to
tactile stimuli in all groups ingesting endrin (Nelson et al., 1956).
Dog. In a series of experiments, dogs were fed diets containing
from 1 to 50 ppm endrin. Two of 4 fed a diet containing 8 ppm and the
one dog on 5 ppm died. The 2 surviving dogs on 8 ppm were kept on the
diet for about 6 months and then killed; increased organ/body-weight
ratios for the liver, kidney and brain were found, and
histopathological examination showed slight degeneration of kidney
tissue. Three dogs on 4 ppm of endrin survived (Treon et al., 1955).
In an experiment of about 19 months' duration, groups of 4
beagles (2 males and 2 females) were placed on diets containing 1 and
3 ppm of endrin. All dogs on 3 ppm had increased organ/body-weight
ratios for the kidney and heart. At 1 ppm the females showed
questionable tubular vacuolation in the kidneys (Treon et al., 1955).
Cattle and sheep. Cattle and sheep were not affected by 5 ppm
of endrin in their diet for 112 days (Radeleff, 1956).
Fowl. Seven-day-old chicks were unaffected by a ration
containing endrin at levels of 1.5 and 3 ppm. When the concentration
of endrin was either 6 or 12 ppm the chicks became highly excitable,
failed to gain as much weight as the controls and the survival rates
for the 12 weeks were 85% and 5% respectively (controls 100%) (Sherman
& Rosenberg, 1954).
Long-term studies
Rat. In a 2-year experiment, groups of rats of 20 males and 20
females each were given diets containing 1, 5, 25, 50 and 100 ppm of
endrin. Concentrations of 50 and 100 ppm were lethal within a few
weeks. The concentration of 25 ppm increased the mortality rate of the
females. Non-survivors at the 3 higher levels exhibited diffuse
degeneration of the brain, liver, kidneys and adrenal glands. The
survivors in the 2 higher levels showed degenerative changes in the
liver only, while those fed at the lower levels had normal viscera.
The level of 5 ppm caused an increase in liver/body-weight ratio in
males and an increase in kidney/body-weight ratio in females. There
was no effect at the 1 ppm level (Treon et al., 1955).
Comments on experimental studies reported
Studies of 2 years' duration in rats and 19 months' duration in
dogs have been published. Other studies of shorter duration have been
published on the toxicity of endrin to rats, fowl, sheep and cattle.
In one experiment, the no-effect level in rats was 1 ppm. In
another study this dose level caused an increase in the serum alkaline
phosphatase level. A questionable effect occurred in female dogs on 1
ppm of endrin.
EVALUATION
Level causing no significant toxicological effect
In neither the dog nor the rat has a no-effect level been
demonstrated.
Acceptable daily intake for man
From the data presented, an acceptable daily intake for man
cannot be estimated. Until further evidence is forthcoming, every
effort should be made to see that the intake of endrin for man is kept
at the lowest possible level.
Further work required
Additional long-term studies including reproduction studies.
Determination of a no-effect level in more than one species.
REFERENCES
Devise, G. M. & Lewis, I. (1956) Brit. med. J., ii, 393
Ely, R. E., Moore, L. A., Carter, R. H. & App, B. E. (1957) J. econ.
Ent., 20, 348
Kiigemagi, U., Sprowls, R. G. & Terriere, L. C. (1958) J. Agr. Food
Chem., 6, 518
Lehman, A. J. (1956) Quart. Bull. Assoc. Food and Drug Officials
U.S., 20, 95
Nelson, S. C., Bahler, T. L., Hartwell, W. V., Greenwood, D. A. &
Harris, L. E. (1956) J. Agr. Food Chem., 4, 696
Radeleff, R. D. (1956) Mosquito News, 16, 79
Sherman, M. & Rosenberg, M. M. (1954) J. econ. Ent., 47, 1082
Speck, L. B. & Maaske, C. A. (1958) A.M.A. Arch. industr. Hlth,
18, 268
Street, J. C., Butcher, J. E., Raleigh, R. J. & Clanton, D. C. (1957)
Proc. Western Sec. Amer. Soc. for Anim. Production, XLVI, 1
Terriere, L. C., Kiigemagi, U. & England, D. C. (1958) J. Agr. Food
Chem., 6, 516
Treon, J. F., Cleveland, F. P. & Cappel, J. (1955) J. Agr. Food.
Chem., 3, 842
Zavon, M. R. (1961) Unpublished data from the Kettering Laboratory,
University of Cincinnati, Cincinnati, Ohio
BIOLOGICAL DATA
Biochemical aspects
Endrin fed to animals is stored unchanged in the tissues
(Kiigemagi et al., 1958, Street et al., 1957; Terriere et al., 1958;
Treon et al., 1955) and excreted in milk and eggs (Ely et al., 1957;
Street et al., 1957, Terriere et al., 1958). The chief organ of
storage is the fat, with a storage ratio of about 15 when the
concentration in the diet is 1 ppm (Lehman, 1956). Endrin is slowly
excreted from the tissues (Nelson et al., 1956. Zavon, 1961).
Serum alkaline phosphatase levels in rats increased as the
concentration of endrin in the diet was increased from 1 ppm to 100
ppm (Nelson et al., 1956).
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Adult rat (female) Oral 7.3 Treon et al., 1955
Young rat (female) Oral 16.8 Treon et al., 1955
Adult rat (male) Oral 40-43.4 Speck & Maaske, 1958
Treon et al., 1955
(cont'd)
Animal Route LD50 mg/kg References
body-weight
Young rat (male) Oral 28.8 Treon et al., 1955
Rabbit (female) Oral 7-10 Treon et al., 1955
Guinea-pig Oral approx. 16-36 Treon et al., 1955
Monkey Oral approx. 3 Treon et al., 1955
Man. In one incident 59 people became ill from the ingestion of
bread containing 150 ppm of endrin, but there were no fatalities
(Davies & Lewis, 1956). The maximum amount consumed was estimated to
have been 1 mg/kg (Zavon, 1961).
Short-term studies
Rat. Adult rats, 5 males and 5 females per group, were given
diets containing 1, 5, 25, 50 and 100 ppm of endrin for 16 weeks. Only
2 rats on 50 ppm and 3 on 25 ppm survived. Three male rats died in the
group on 5 ppm. There was loss of weight and hypersensitivity to
tactile stimuli in all groups ingesting endrin (Nelson et al., 1956).
Dog. In a series of experiments, dogs were fed diets containing
from 1 to 50 ppm endrin. Two of 4 fed a diet containing 8 ppm and the
one dog on 5 ppm died. The 2 surviving dogs on 8 ppm were kept on the
diet for about 6 months and then killed; increased organ/body-weight
ratios for the liver, kidney and brain were found, and
histopathological examination showed slight degeneration of kidney
tissue. Three dogs on 4 ppm of endrin survived (Treon et al., 1955).
In an experiment of about 19 months' duration, groups of 4
beagles (2 males and 2 females) were placed on diets containing 1 and
3 ppm of endrin. All dogs on 3 ppm had increased organ/body-weight
ratios for the kidney and heart. At 1 ppm the females showed
questionable tubular vacuolation in the kidneys (Treon et al., 1955).
Cattle and sheep. Cattle and sheep were not affected by 5 ppm
of endrin in their diet for 112 days (Radeleff, 1956).
Fowl. Seven-day-old chicks were unaffected by a ration
containing endrin at levels of 1.5 and 3 ppm. When the concentration
of endrin was either 6 or 12 ppm the chicks became highly excitable,
failed to gain as much weight as the controls and the survival rates
for the 12 weeks were 85% and 5% respectively (controls 100%) (Sherman
& Rosenberg, 1954).
Long-term studies
Rat. In a 2-year experiment, groups of rats of 20 males and 20
females each were given diets containing 1, 5, 25, 50 and 100 ppm of
endrin. Concentrations of 50 and 100 ppm were lethal within a few
weeks. The concentration of 25 ppm increased the mortality rate of the
females. Non-survivors at the 3 higher levels exhibited diffuse
degeneration of the brain, liver, kidneys and adrenal glands. The
survivors in the 2 higher levels showed degenerative changes in the
liver only, while those fed at the lower levels had normal viscera.
The level of 5 ppm caused an increase in liver/body-weight ratio in
males and an increase in kidney/body-weight ratio in females. There
was no effect at the 1 ppm level (Treon et al., 1955).
Comments on experimental studies reported
Studies of 2 years' duration in rats and 19 months' duration in
dogs have been published. Other studies of shorter duration have been
published on the toxicity of endrin to rats, fowl, sheep and cattle.
In one experiment, the no-effect level in rats was 1 ppm. In
another study this dose level caused an increase in the serum alkaline
phosphatase level. A questionable effect occurred in female dogs on 1
ppm of endrin.
EVALUATION
Level causing no significant toxicological effect
In neither the dog nor the rat has a no-effect level been
demonstrated.
Acceptable daily intake for man
From the data presented, an acceptable daily intake for man
cannot be estimated. Until further evidence is forthcoming, every
effort should be made to see that the intake of endrin for man is kept
at the lowest possible level.
Further work required
Additional long-term studies including reproduction studies.
Determination of a no-effect level in more than one species.
REFERENCES
Devise, G. M. & Lewis, I. (1956) Brit. med. J., ii, 393
Ely, R. E., Moore, L. A., Carter, R. H. & App, B. E. (1957) J. econ.
Ent., 20, 348
Kiigemagi, U., Sprowls, R. G. & Terriere, L. C. (1958) J. Agr. Food
Chem., 6, 518
Lehman, A. J. (1956) Quart. Bull. Assoc. Food and Drug Officials
U.S., 20, 95
Nelson, S. C., Bahler, T. L., Hartwell, W. V., Greenwood, D. A. &
Harris, L. E. (1956) J. Agr. Food Chem., 4, 696
Radeleff, R. D. (1956) Mosquito News, 16, 79
Sherman, M. & Rosenberg, M. M. (1954) J. econ. Ent., 47, 1082
Speck, L. B. & Maaske, C. A. (1958) A.M.A. Arch. industr. Hlth,
18, 268
Street, J. C., Butcher, J. E., Raleigh, R. J. & Clanton, D. C. (1957)
Proc. Western Sec. Amer. Soc. for Anim. Production, XLVI, 1
Terriere, L. C., Kiigemagi, U. & England, D. C. (1958) J. Agr. Food
Chem., 6, 516
Treon, J. F., Cleveland, F. P. & Cappel, J. (1955) J. Agr. Food.
Chem., 3, 842
Zavon, M. R. (1961) Unpublished data from the Kettering Laboratory,
University of Cincinnati, Cincinnati, Ohio
 BIOLOGICAL DATA
Biochemical aspects
Endrin fed to animals is stored unchanged in the tissues
(Kiigemagi et al., 1958, Street et al., 1957; Terriere et al., 1958;
Treon et al., 1955) and excreted in milk and eggs (Ely et al., 1957;
Street et al., 1957, Terriere et al., 1958). The chief organ of
storage is the fat, with a storage ratio of about 15 when the
concentration in the diet is 1 ppm (Lehman, 1956). Endrin is slowly
excreted from the tissues (Nelson et al., 1956. Zavon, 1961).
Serum alkaline phosphatase levels in rats increased as the
concentration of endrin in the diet was increased from 1 ppm to 100
ppm (Nelson et al., 1956).
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Adult rat (female) Oral 7.3 Treon et al., 1955
Young rat (female) Oral 16.8 Treon et al., 1955
Adult rat (male) Oral 40-43.4 Speck & Maaske, 1958
Treon et al., 1955
(cont'd)
Animal Route LD50 mg/kg References
body-weight
Young rat (male) Oral 28.8 Treon et al., 1955
Rabbit (female) Oral 7-10 Treon et al., 1955
Guinea-pig Oral approx. 16-36 Treon et al., 1955
Monkey Oral approx. 3 Treon et al., 1955
Man. In one incident 59 people became ill from the ingestion of
bread containing 150 ppm of endrin, but there were no fatalities
(Davies & Lewis, 1956). The maximum amount consumed was estimated to
have been 1 mg/kg (Zavon, 1961).
Short-term studies
Rat. Adult rats, 5 males and 5 females per group, were given
diets containing 1, 5, 25, 50 and 100 ppm of endrin for 16 weeks. Only
2 rats on 50 ppm and 3 on 25 ppm survived. Three male rats died in the
group on 5 ppm. There was loss of weight and hypersensitivity to
tactile stimuli in all groups ingesting endrin (Nelson et al., 1956).
Dog. In a series of experiments, dogs were fed diets containing
from 1 to 50 ppm endrin. Two of 4 fed a diet containing 8 ppm and the
one dog on 5 ppm died. The 2 surviving dogs on 8 ppm were kept on the
diet for about 6 months and then killed; increased organ/body-weight
ratios for the liver, kidney and brain were found, and
histopathological examination showed slight degeneration of kidney
tissue. Three dogs on 4 ppm of endrin survived (Treon et al., 1955).
In an experiment of about 19 months' duration, groups of 4
beagles (2 males and 2 females) were placed on diets containing 1 and
3 ppm of endrin. All dogs on 3 ppm had increased organ/body-weight
ratios for the kidney and heart. At 1 ppm the females showed
questionable tubular vacuolation in the kidneys (Treon et al., 1955).
Cattle and sheep. Cattle and sheep were not affected by 5 ppm
of endrin in their diet for 112 days (Radeleff, 1956).
Fowl. Seven-day-old chicks were unaffected by a ration
containing endrin at levels of 1.5 and 3 ppm. When the concentration
of endrin was either 6 or 12 ppm the chicks became highly excitable,
failed to gain as much weight as the controls and the survival rates
for the 12 weeks were 85% and 5% respectively (controls 100%) (Sherman
& Rosenberg, 1954).
Long-term studies
Rat. In a 2-year experiment, groups of rats of 20 males and 20
females each were given diets containing 1, 5, 25, 50 and 100 ppm of
endrin. Concentrations of 50 and 100 ppm were lethal within a few
weeks. The concentration of 25 ppm increased the mortality rate of the
females. Non-survivors at the 3 higher levels exhibited diffuse
degeneration of the brain, liver, kidneys and adrenal glands. The
survivors in the 2 higher levels showed degenerative changes in the
liver only, while those fed at the lower levels had normal viscera.
The level of 5 ppm caused an increase in liver/body-weight ratio in
males and an increase in kidney/body-weight ratio in females. There
was no effect at the 1 ppm level (Treon et al., 1955).
Comments on experimental studies reported
Studies of 2 years' duration in rats and 19 months' duration in
dogs have been published. Other studies of shorter duration have been
published on the toxicity of endrin to rats, fowl, sheep and cattle.
In one experiment, the no-effect level in rats was 1 ppm. In
another study this dose level caused an increase in the serum alkaline
phosphatase level. A questionable effect occurred in female dogs on 1
ppm of endrin.
EVALUATION
Level causing no significant toxicological effect
In neither the dog nor the rat has a no-effect level been
demonstrated.
Acceptable daily intake for man
From the data presented, an acceptable daily intake for man
cannot be estimated. Until further evidence is forthcoming, every
effort should be made to see that the intake of endrin for man is kept
at the lowest possible level.
Further work required
Additional long-term studies including reproduction studies.
Determination of a no-effect level in more than one species.
REFERENCES
Devise, G. M. & Lewis, I. (1956) Brit. med. J., ii, 393
Ely, R. E., Moore, L. A., Carter, R. H. & App, B. E. (1957) J. econ.
Ent., 20, 348
Kiigemagi, U., Sprowls, R. G. & Terriere, L. C. (1958) J. Agr. Food
Chem., 6, 518
Lehman, A. J. (1956) Quart. Bull. Assoc. Food and Drug Officials
U.S., 20, 95
Nelson, S. C., Bahler, T. L., Hartwell, W. V., Greenwood, D. A. &
Harris, L. E. (1956) J. Agr. Food Chem., 4, 696
Radeleff, R. D. (1956) Mosquito News, 16, 79
Sherman, M. & Rosenberg, M. M. (1954) J. econ. Ent., 47, 1082
Speck, L. B. & Maaske, C. A. (1958) A.M.A. Arch. industr. Hlth,
18, 268
Street, J. C., Butcher, J. E., Raleigh, R. J. & Clanton, D. C. (1957)
Proc. Western Sec. Amer. Soc. for Anim. Production, XLVI, 1
Terriere, L. C., Kiigemagi, U. & England, D. C. (1958) J. Agr. Food
Chem., 6, 516
Treon, J. F., Cleveland, F. P. & Cappel, J. (1955) J. Agr. Food.
Chem., 3, 842
Zavon, M. R. (1961) Unpublished data from the Kettering Laboratory,
University of Cincinnati, Cincinnati, Ohio
BIOLOGICAL DATA
Biochemical aspects
Endrin fed to animals is stored unchanged in the tissues
(Kiigemagi et al., 1958, Street et al., 1957; Terriere et al., 1958;
Treon et al., 1955) and excreted in milk and eggs (Ely et al., 1957;
Street et al., 1957, Terriere et al., 1958). The chief organ of
storage is the fat, with a storage ratio of about 15 when the
concentration in the diet is 1 ppm (Lehman, 1956). Endrin is slowly
excreted from the tissues (Nelson et al., 1956. Zavon, 1961).
Serum alkaline phosphatase levels in rats increased as the
concentration of endrin in the diet was increased from 1 ppm to 100
ppm (Nelson et al., 1956).
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Adult rat (female) Oral 7.3 Treon et al., 1955
Young rat (female) Oral 16.8 Treon et al., 1955
Adult rat (male) Oral 40-43.4 Speck & Maaske, 1958
Treon et al., 1955
(cont'd)
Animal Route LD50 mg/kg References
body-weight
Young rat (male) Oral 28.8 Treon et al., 1955
Rabbit (female) Oral 7-10 Treon et al., 1955
Guinea-pig Oral approx. 16-36 Treon et al., 1955
Monkey Oral approx. 3 Treon et al., 1955
Man. In one incident 59 people became ill from the ingestion of
bread containing 150 ppm of endrin, but there were no fatalities
(Davies & Lewis, 1956). The maximum amount consumed was estimated to
have been 1 mg/kg (Zavon, 1961).
Short-term studies
Rat. Adult rats, 5 males and 5 females per group, were given
diets containing 1, 5, 25, 50 and 100 ppm of endrin for 16 weeks. Only
2 rats on 50 ppm and 3 on 25 ppm survived. Three male rats died in the
group on 5 ppm. There was loss of weight and hypersensitivity to
tactile stimuli in all groups ingesting endrin (Nelson et al., 1956).
Dog. In a series of experiments, dogs were fed diets containing
from 1 to 50 ppm endrin. Two of 4 fed a diet containing 8 ppm and the
one dog on 5 ppm died. The 2 surviving dogs on 8 ppm were kept on the
diet for about 6 months and then killed; increased organ/body-weight
ratios for the liver, kidney and brain were found, and
histopathological examination showed slight degeneration of kidney
tissue. Three dogs on 4 ppm of endrin survived (Treon et al., 1955).
In an experiment of about 19 months' duration, groups of 4
beagles (2 males and 2 females) were placed on diets containing 1 and
3 ppm of endrin. All dogs on 3 ppm had increased organ/body-weight
ratios for the kidney and heart. At 1 ppm the females showed
questionable tubular vacuolation in the kidneys (Treon et al., 1955).
Cattle and sheep. Cattle and sheep were not affected by 5 ppm
of endrin in their diet for 112 days (Radeleff, 1956).
Fowl. Seven-day-old chicks were unaffected by a ration
containing endrin at levels of 1.5 and 3 ppm. When the concentration
of endrin was either 6 or 12 ppm the chicks became highly excitable,
failed to gain as much weight as the controls and the survival rates
for the 12 weeks were 85% and 5% respectively (controls 100%) (Sherman
& Rosenberg, 1954).
Long-term studies
Rat. In a 2-year experiment, groups of rats of 20 males and 20
females each were given diets containing 1, 5, 25, 50 and 100 ppm of
endrin. Concentrations of 50 and 100 ppm were lethal within a few
weeks. The concentration of 25 ppm increased the mortality rate of the
females. Non-survivors at the 3 higher levels exhibited diffuse
degeneration of the brain, liver, kidneys and adrenal glands. The
survivors in the 2 higher levels showed degenerative changes in the
liver only, while those fed at the lower levels had normal viscera.
The level of 5 ppm caused an increase in liver/body-weight ratio in
males and an increase in kidney/body-weight ratio in females. There
was no effect at the 1 ppm level (Treon et al., 1955).
Comments on experimental studies reported
Studies of 2 years' duration in rats and 19 months' duration in
dogs have been published. Other studies of shorter duration have been
published on the toxicity of endrin to rats, fowl, sheep and cattle.
In one experiment, the no-effect level in rats was 1 ppm. In
another study this dose level caused an increase in the serum alkaline
phosphatase level. A questionable effect occurred in female dogs on 1
ppm of endrin.
EVALUATION
Level causing no significant toxicological effect
In neither the dog nor the rat has a no-effect level been
demonstrated.
Acceptable daily intake for man
From the data presented, an acceptable daily intake for man
cannot be estimated. Until further evidence is forthcoming, every
effort should be made to see that the intake of endrin for man is kept
at the lowest possible level.
Further work required
Additional long-term studies including reproduction studies.
Determination of a no-effect level in more than one species.
REFERENCES
Devise, G. M. & Lewis, I. (1956) Brit. med. J., ii, 393
Ely, R. E., Moore, L. A., Carter, R. H. & App, B. E. (1957) J. econ.
Ent., 20, 348
Kiigemagi, U., Sprowls, R. G. & Terriere, L. C. (1958) J. Agr. Food
Chem., 6, 518
Lehman, A. J. (1956) Quart. Bull. Assoc. Food and Drug Officials
U.S., 20, 95
Nelson, S. C., Bahler, T. L., Hartwell, W. V., Greenwood, D. A. &
Harris, L. E. (1956) J. Agr. Food Chem., 4, 696
Radeleff, R. D. (1956) Mosquito News, 16, 79
Sherman, M. & Rosenberg, M. M. (1954) J. econ. Ent., 47, 1082
Speck, L. B. & Maaske, C. A. (1958) A.M.A. Arch. industr. Hlth,
18, 268
Street, J. C., Butcher, J. E., Raleigh, R. J. & Clanton, D. C. (1957)
Proc. Western Sec. Amer. Soc. for Anim. Production, XLVI, 1
Terriere, L. C., Kiigemagi, U. & England, D. C. (1958) J. Agr. Food
Chem., 6, 516
Treon, J. F., Cleveland, F. P. & Cappel, J. (1955) J. Agr. Food.
Chem., 3, 842
Zavon, M. R. (1961) Unpublished data from the Kettering Laboratory,
University of Cincinnati, Cincinnati, Ohio
