
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 68
POLYCHLORINATED BIPHENYLS (PCBs) AND
POLYCHLORINATED TERPHENYLS (PCTs)
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1992
This is a companion volume to Environmental Health Criteria 140:
Polychlorinated Biphenyls and Terphenyls (Second Edition)
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the
United Nations Environment Programme, the International Labour
Organisation, and the World Health Organization)
This report contains the collective views of an international group
of experts and does not necessarily represent the decisions or the
stated policy of the United Nations Environment Programme, the
International Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Polychlorinated biphenyls, polychlorinated terphenyls
(PCBs and PCTs) : health and safety guide.
(Health and safety guide ; no. 68)
1. Polychlorobiphenyl compounds - poisoning
2. Polychlorobiphenyl compounds - standards
3. Polychloroterphenyl compounds - poisoning
4. Polychloroterphenyl compounds - standards
5. Environmental exposure 6. Environmental pollutants
7. Hazardous substances I. Series
ISBN 92 4 151068 4 (NLM Classification: QV 633)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1992
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.1.1. Polychlorinated biphenyls - PCBs
1.1.2. Polychlorinated terphenyls - PCTs
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Uses
2. SUMMARY AND EVALUATION
2.1. Environmental transport, distribution, and transformation
2.2. Environmental levels and human exposure
2.3. Kinaetics and metabolism
2.4. Effects on organisms in the environment
2.4.1. Laboratory studies
2.4.2. Field studies
2.5. Effects on experimental animals and in vitro systems
2.5.1. Single exposure
2.5.2. Short-term exposure
2.5.3. Reproduction, embryotoxicity, and teratogenicity
2.5.4. Mutagenicity
2.5.5. Carcinogenicity
2.5.6. Special studies
2.5.7. Factors modifying toxicity; mode of action
2.6. Effects in humans
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.1.1. Distribution
3.1.2. Effects on experimental animals
3.1.3. Effects on humans
3.1.4. Effects on the environment
3.2. Recommendations
4. HEALTH HAZARDS FOR MAN, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 Medical advice
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.3.1. Leaking containers in store
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
5.1. Hazards
5.2. Prevention
6 SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding
EHC. Section 4 includes advice on preventive and protective measures
and emergency action; health workers should be thoroughly familiar
with the medical information to ensure that they can act efficiently
in an emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those
in ministries, governmental agencies, industry, and trade unions who
are involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING
POINT TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
1.1.1 Polychlorinated biphenyls - PCBs
Chemical formula - chemical structure
The chlorination of biphenyl can lead to the replacement of 1-10
hydrogen atoms by chlorine.
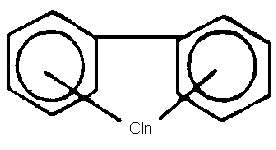 The chemical formula can be presented as C12H10-nCln, where n is
the number of chlorine atoms in the molecule.
Chemical composition
The PCBs are chlorinated hydrocarbons that are manufactured
commercially by the progressive chlorination of biphenyl in the
presence of a suitable catalyst (e.g., iron chloride). Depending on
the reaction conditions, the degree of chlorination can vary between
21 and 68% (w/w). The yield is always a mixture of different
compounds and congeners. Thus, a total of 209 different chemical
components may exist, but only about 130 of these are likely to
occur in commercial products or mixtures of these compounds.
Individual PCBs have been synthesized for use as reference samples
in the identification of gas-liquid chromatographic peaks, for
toxicological investigations, and in order to study their metabolic
fate in living organisms.
Purity and impurities
Commercial PCBs are sold on the basis of their physical properties,
not their chemical composition. Different batches may vary somewhat
in their composition. The impurities known to be present in
commercial PCBs include chlorinated naphthalenes and small
quantities of the highly toxic polychlorinated dibenzofurans
(PCDFs). There are no authenticated reports of the presence of
polychlorinated dibenzo- p-dioxins (PCDDs) in commercial PCBs.
Common name: Polychlorinated biphenyls - PCBs.
CAS registry number: 1336-36-3
RTECS registry number: TQ1350000
Relative molecular mass: Depends on degree of chlorination and
composition of the mixture.
Major trade names
Apirolio (t,c) Disconon (c) PCBs
Areclor (t) Dk (t,c) Phenoclor (t,c)
Aroclor Duconol (c) Polychlorinated biphenyl
Arubren Dykanol (t,c) Polychlorobiphenyl
Asbestol (t,c) EEC-18 Pydraul
Askarel Elemex (t,c) Pyralene (t,c)
Bakola 131 (t,c) Eucarel Pyranol (t,c)
Biclor (c) Fenchlor (t,c) Pyroclor (t)
Chlorextol (t) Hivar (c) Saf-T-Kuhl (t,c)
Chlorinated Biphenyl Hydol (t,c) Santotherm FR a
Chlorinated Diphenyl Inclor Santovac 1 and 2
Chlorinol Interteen (t,c) Siclonyl (c)
Chlorobiphenyl Kanechlor (t,c) Solvol (t,c)
Clophen (t,c) Kennechlor Sovol
Chlorphen (t) Montar Therminol FR a
Delor Nepolin
Diaclor (t,c) No-Flamol (t,c)
Dialor (c) PCB
a Previous products (FR-series) used as heat transfer fluids
contained PCBs, but since 1972 current products are different
series and do not contain PCBs.
(t) used in transformers.
(c) used in capacitors.
1.1.2 Polychlorinated terphenyls - PCTs
Chemical formula - chemical structure
The chemical formula can be presented as C12H10-nCln, where n is
the number of chlorine atoms in the molecule.
Chemical composition
The PCBs are chlorinated hydrocarbons that are manufactured
commercially by the progressive chlorination of biphenyl in the
presence of a suitable catalyst (e.g., iron chloride). Depending on
the reaction conditions, the degree of chlorination can vary between
21 and 68% (w/w). The yield is always a mixture of different
compounds and congeners. Thus, a total of 209 different chemical
components may exist, but only about 130 of these are likely to
occur in commercial products or mixtures of these compounds.
Individual PCBs have been synthesized for use as reference samples
in the identification of gas-liquid chromatographic peaks, for
toxicological investigations, and in order to study their metabolic
fate in living organisms.
Purity and impurities
Commercial PCBs are sold on the basis of their physical properties,
not their chemical composition. Different batches may vary somewhat
in their composition. The impurities known to be present in
commercial PCBs include chlorinated naphthalenes and small
quantities of the highly toxic polychlorinated dibenzofurans
(PCDFs). There are no authenticated reports of the presence of
polychlorinated dibenzo- p-dioxins (PCDDs) in commercial PCBs.
Common name: Polychlorinated biphenyls - PCBs.
CAS registry number: 1336-36-3
RTECS registry number: TQ1350000
Relative molecular mass: Depends on degree of chlorination and
composition of the mixture.
Major trade names
Apirolio (t,c) Disconon (c) PCBs
Areclor (t) Dk (t,c) Phenoclor (t,c)
Aroclor Duconol (c) Polychlorinated biphenyl
Arubren Dykanol (t,c) Polychlorobiphenyl
Asbestol (t,c) EEC-18 Pydraul
Askarel Elemex (t,c) Pyralene (t,c)
Bakola 131 (t,c) Eucarel Pyranol (t,c)
Biclor (c) Fenchlor (t,c) Pyroclor (t)
Chlorextol (t) Hivar (c) Saf-T-Kuhl (t,c)
Chlorinated Biphenyl Hydol (t,c) Santotherm FR a
Chlorinated Diphenyl Inclor Santovac 1 and 2
Chlorinol Interteen (t,c) Siclonyl (c)
Chlorobiphenyl Kanechlor (t,c) Solvol (t,c)
Clophen (t,c) Kennechlor Sovol
Chlorphen (t) Montar Therminol FR a
Delor Nepolin
Diaclor (t,c) No-Flamol (t,c)
Dialor (c) PCB
a Previous products (FR-series) used as heat transfer fluids
contained PCBs, but since 1972 current products are different
series and do not contain PCBs.
(t) used in transformers.
(c) used in capacitors.
1.1.2 Polychlorinated terphenyls - PCTs
Chemical formula - chemical structure
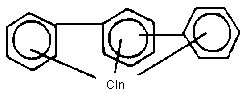 The chemical formula can be written as C18H14-nCln, in which n
is the number of chlorine atoms, which can range from 1-14.
Chemical composition
The theoretically possible number of different PCTs is several
orders of magnitude greater than the number of PCBs, but in
practice, as with PCBs, PCTs are sold on the basis of their physical
properties, which depend on the degree of chlorination, and not
their chemical composition.
Common name: Polychlorinated terphenyls - PCTs.
CAS registry number: 61788-33-8
RTECS registry number: WZ6500000
Relative molecular mass: Depends on degree of chlorination and
composition of the mixture.
Main trade names
PCTs are known by a variety of trade names, some of which are
similar to those given for PCBs in Section 1.1.1. In the Aroclor
series, terphenyls are indicated by 54 in the first two places of
the four digit code. In Japan, the PCTs are coded Kanechlor KC-C.
1.2 Physical and chemical properties
Individual, pure congeners of PCB and PCT are colourless, often
crystalline compounds, but commercial PCBs are mixtures of these
congeners with a clear, light yellow or dark colour, and range from
oily liquids to waxy or hard solids. They do not crystallize at low
temperatures, but turn into solid resins. Because of the chlorine
atoms in the molecule, the compounds have a fairly high density. In
practice, PCBs are fire resistant, and have a fairly high
flash-point (170-380 °C). They form vapours that are heavier than
air, but they do not form an explosive mixture with air. The
electrical conductivity of PCBs and PCTs is very low, and their
resistance to thermal breakdown is extremely high. It is on the
basis of these properties that they are used as cooling liquids in
electrical equipment. The physical properties of some Aroclors are
shown in Table 1.
PCBs are chemically stable under normal conditions. They are very
resistant to a range of oxidants and other chemicals, and they
remain chemically unchanged even in the presence of oxygen, or some
active metals, at high temperatures (up to 170 °C), and for
protracted periods.
PCBs are practically insoluble in water; however, they dissolve
easily in hydrocarbons, fats, and other organic compounds, and they
are readily adsorbed by fatty tissues.
The partition coefficient (log Kow) values for all 209 PCB
congeners range from 4.46 to 8.18.
1.3 Analytical methods
Only a small number of laboratories in the world have access to, and
experience of working with, the complicated techniques necessary for
a reproducible determination of PCBs and PCTs.
One probable source of error is incomplete extraction and clean-up
of the PCBs. The method used to quantify the gas-liquid
chromatographic peaks also gives rise to variation between
laboratories.
Data on concentrations of PCBs must be interpreted with the greatest
care. Comparisons can only be made between data from the same
laboratory, obtained using the same validated technique over a long
period. Comparisons between data from different laboratories are
possible in only the very few cases, in which very strict
interlaboratory checks have been made on the basis of the same
sampling and analytical techniques. Indications about trends can
only be obtained when these basic considerations are taken into
account.
Gas-liquid chromatography (GLC) with packed or capillary columns, is
generally used for the analysis, and comparison of peak patterns,
and various PCB standard formulations are used for quantification.
Different approaches are used for the summing-up of individual
peaks.
Analytical methods are discussed in more detail in the WHO/EURO
(1987) document.
Individual congeners are identified using GLC, with either hydrogen
flame ionization detection (HFID) or electron capture detection
(ECD), and mass spectrometry.
Table 1. Physical properties of some Aroclors
Substance Water Vapour Density Appearance Henry's Law Refractive index Boiling point
Aroclor solubility pressure g/cm3 constant (distillation
(mg/litre, (torr, (25 °C) (atm-m3/mol range)
25 °C) 25 °C) at 25 °C) (750 torr, °C)
1016 0.42 4.0 x 10-4 1.33 Clear, 2.9 x 10-4 1.6215-1.6135 325-356
mobile oil (at 25 °C)
1221 0.59 6.7 x 10-3 1.15 Clear, 3.5 x 10-3 1.617-1.618 275-320
mobile oil
1232 0.45 4.1 x 10-3 1.24 Clear, unknown unknown 290-325
mobile oil
1242 0.24 4.1 x 10-3 1.35 Clear, 5.2 x 10-4 1.627-1.629 325-366
mobile oil (at 20 °C)
1248 0.054 4.9 x 10-4 1.41 Clear, 28 x 10-3 unknown 340-375
mobile oil
1254 0.021 7.7 x 10-5 1.50 Light yellow, 2.0 x 10-3 1.6375-1.6415 365-390
viscous oil (at 25 °C)
1260 0.0027 4.0 x 10-5 1.58 Light yellow, 4.6 x 10-3 unknown 385-420
sticky resin
1.4 Uses
The industrial usefulness of PCBs and PCTs depends on their chemical
inertness, resistance to heat, non-flammability, low vapour pressure
(particularly with the higher chlorinated compounds), and high
dielectric constant.
The main uses are (or were):
(a) as dielectrics in transformers and large capacitors (considered
to be closed systems);
(b) in heat transfer and hydraulic systems (nominally closed
systems);
(c) in the formulation of lubricating and cutting oils;
(d) as plasticizers in paints, carbonless copying paper, adhesives,
sealants, and plastics.
Both c and d are open-ended applications.
2. SUMMARY AND EVALUATION
2.1 Environmental transport, distribution, and transformation
In the atmosphere, PCBs exist primarily in the vapour phase; the
tendency to adsorb to particulates increases with the degree of
chlorination. The virtually universal distribution of PCBs suggests
that they are transported in air.
At present, the major source of PCB exposure for the general
population appears to be as a consequence of the redistribution of
PCBs previously introduced into the environment. This redistribution
involves volatilization from soil and water into the atmosphere,
with subsequent transport in air and removal from the atmosphere
through wet or dry deposition (of PCBs bound to particulates), and
then re-volatilization. The concentrations of PCBs in precipitation
range from 0.001 to 0.25 µg/litre. Since the volatilization and
degradation rates of PCBs vary among the different congeners, this
redistribution leads to an alteration in the composition of PCB
mixtures in the environment.
In water, PCBs are adsorbed to sediments and other organic matter;
experimental and monitoring data have shown that PCB concentrations
are higher in sediment and suspended matter than in the associated
water columns. Strong adsorption to sediment, especially in the case
of the higher chlorinated PCBs, decreases the rate of
volatilization. On the basis of their water solubilities and
n-octanol-water partition coefficients, the lower chlorinated PCB
congeners will sorb less strongly than the higher chlorinated
isomers. Although adsorption can immobilize PCBs for relatively long
periods in the aquatic environment, desorption into the water column
has been shown to occur by both the abiotic and biotic routes. The
substantial quantities of PCBs in aquatic sediments therefore act as
both an environmental sink and a reservoir of PCBs for subsequent
recycling in the environment. Most of the PCBs in the environment
are in the aquatic sediment.
The low solubility of PCBs, and their strong adsorption to soil
particles, limits leaching in soil; the lower chlorinated PCBs will
tend to leach more than the highly chlorinated PCBs.
Degradation of PCBs in the environment depends on the degree of
chlorination of the biphenyl. In general, the persistence of PCB
congeners increases as the degree of chlorination increases. In the
atmosphere, the reaction of PCBs in the vapour phase with hydroxyl
radicals (which are photochemically formed by sunlight) may be the
most important transformation process. The estimated half-life of
this reaction in the atmosphere ranges from about 10 days to 1.5
years for a monochlorobiphenyl and a heptachlorobiphenyl,
respectively.
In the aquatic environment, PCBs are not significantly degraded by
hydrolysis and oxidation. Photolysis appears to be the only abiotic
degradation process in water; however, insufficient experimental
data are available to determine its rate or its importance in the
environment.
Microorganisms degrade mono-, di-, and trichlorinated biphenyls
relatively rapidly, and tetrachlorobiphenyls slowly, while higher
chlorinated biphenyls are resistant to biodegradation. The chlorine
substitution position on the biphenyl ring appears to be important
in determining the biodegradation rate. PCBs containing chlorine
atoms in the para position are preferentially biodegraded. Higher
chlorinated congeners are biotransformed anaerobically, by reductive
dechlorination, to lower chlorinated PCBs, which may then be
biodegradable by aerobic processes.
Several factors determine the degree of bioaccumulation in adipose
tissues: duration and level of exposure, chemical structure of the
compound, and position and pattern of substitution. In general, the
higher chlorinated congeners are accumulated more readily.
The bioconcentration factors of various PCBs determined
experimentally in aquatic species (fish, shrimp, oyster) range from
200 up to 70 000 or higher. In the open ocean, there is
bioaccumulation of PCBs in the higher trophic levels, with an
increased proportion of higher chlorinated biphenyls in the higher
ranking predators.
Transfer of PCBs from soil to vegetation takes place mainly through
adsorption onto the external surfaces of terrestrial plants; little
translocation takes place.
2.2 Environmental levels and human exposure
Because they persist for a long time, and because of other
physicochemical properties, PCBs are present in the environment all
over the world.
PCBs are found in air, all over the world, at concentrations of
0.002 up to 15 ng/m3. In industrial areas, higher levels of up to
micrograms/m3 are found. In rain-water and snow, PCBs are found in
the range of not detectable (<ng/litre) to 250 ng/litre.
Under workplace conditions, the levels in the air may be much
higher. In the manufacturing of transformers or capacitors, for
instance, levels of up to 1000 µg/m3 have been observed. In
emergency situations, concentrations of up to 16 mg/m3 have been
measured. In the case of fires and/or explosions, the soot may
contain high levels of PCBs; concentrations of 8000 mg PCBs/kg soot
have been found. In fires and explosions, PCDFs will also be present
in the soot. In accidents with transformers in which chlorinated
benzenes are present in addition to PCBs, polychlorinated dioxins
(PCDDs) will also be found.
In these emergency situations, skin contamination with soot is
possible, and ingestion or inhalation of soot particles may occur
and result in serious exposures of personnel. However, the exposure
of the general population through the air will be very low.
Surface water may be contaminated by PCBs by atmospheric fall-out,
or by direct emissions from point sources, or waste disposal. Under
certain conditions, levels of up to 100-500 ng/litre of water have
been measured. In the water of oceans, levels of 0.05-0.6 ng/litre
have been found.
In non-contaminated areas, drinking-water usually contains less than
1 ng PCBs/litre, but levels up to 5 ng/litre have been reported.
Soil and sediments normally contain concentrations of PCBs in the
range of <0.01-2.0 mg/kg. In polluted areas, the levels in soils
have been much higher, up to 500 mg/kg.
Over the years, many thousands of samples of different foodstuffs
have been analysed, in several countries, for contaminants,
including PCBs. Most samples have been taken from individual food
items, especially fish and other foods of animal origin, such as
meat and milk. Food becomes contaminated with PCBs by three main
routes:
a) uptake from the environment by fish, birds, livestock (via
food-chains), and crops;
b) migration from packaging materials into food (mainly less than 1
mg/kg, but in some cases up to 10 mg/kg);
c) direct contamination of foodstuffs or animal feed as the result
of an industrial accident.
For the most important food items that contain PCBs, the following
concentrations have been found: animal fat, 20-240 µg/kg; cow's
milk, 5-200 µg/kg; butter, 30-80 µg/kg; fish, 10-500 µg/kg, on fat
basis. Certain fish species (eel) and fish products (fish liver and
fish oils) contained much higher levels, up to 10 mg PCBs/kg. Levels
of <10 µg/kg were found in vegetables, cereals, fruits, and a
number of other products. Fish, shellfish, meat, milk, and other
dairy products are the main foods that give rise to concern as
regards levels of PCBs. The median levels reported in fish, in
various countries, are in the order of 100 µg/kg (on fat basis);
however, it appears that the levels of PCBs in fish are slowly
decreasing.
PCBs accumulate in human adipose tissue and breast milk. The
concentrations of PCBs in different organs and tissues depend upon
the lipid content of the organ or tissue, with the exception of the
brain. The levels of PCB residues in adipose tissue of the general
population in industrialized countries range from <1 to 5 mg/kg, on
fat basis.
The average concentration of total PCBs in human milk is in the
range of 0.5 to 1.5 mg/kg fat, depending on the donor's place of
residence, life-style, and the analytical methods used. Women living
in heavily industrialized urban areas, or with a high fish
consumption (especially fish from heavily contaminated waters), may
have higher PCB concentrations in breast milk.
The composition of most PCB extracts from environmental samples does
not resemble that of the commercial PCB mixtures. High-resolution
gas chromatography (GC) analysis shows that the congener
composition, and relative concentrations of the individual
components, in adipose tissues and breast milk differ markedly from
the composition of commercial PCBs. The GC-patterns of PCBs in human
adipose tissues and breast milk indicate relatively high
concentrations of mainly the higher chlorinated PCBs, such as,
2,4,5,3',4'-pentachlorobiphenyl; 2,4,5,2',4',5'-hexa-
chlorobiphenyl; and 2,3,4,2',4',5'-hexachlorobiphenyl;
2,3,4,5,2',4',5'-hepta- and 2,3,4,5,2',3',4'-heptachlorobiphenyl. A
few other PCB congeners are present at much lower concentrations,
including the most toxic, coplanar PCBs: 3,4,3',4'-tetra-,
3,4,5,3',4'-penta- and 3,4,5,3',4',5'-hexahlorobiphenyl.
The daily intake of PCBs by infants from breast milk is of the order
of 4.2 µg/kg body weight (5.2 µg/100 kcal consumed) (WHO/EURO,
1987). The average total quantity of PCBs ingested in breast milk
during the first 6 months of life is 4.5 mg compared with a
calculated intake of 357 mg of PCBs over the subsequent life-time
(0.2 µg/kg per day in the diet of a 70-kg person over a 70-year
life-time). Therefore, the nursing period contributes about 1.3% of
the life-time intake, which is not large in light of the benefits of
breast-feeding (WHO/EURO, 1987).
On the basis of the evaluated background data, the average dietary
intake of PCBs for adults amounts to a maximum of 100 µg/week, or
approximately 14 µg/person per day. For a 70-kg person, this is an
intake equivalent to a maximum of 0.2 µg/kg body weight per day
(WHO/EURO, 1987).
2.3 Kinetics and metabolism
Animal studies have been reported involving mainly oral, inhalation,
and dermal exposures to both PCB mixtures and individual congeners.
In general, PCBs appear to be rapidly absorbed, particularly by the
gastro-intestinal tract after oral exposure. It is clear that
absorption does occur in humans, but information on the rates of
absorption of PCBs in humans is limited.
From the available studies, the data on the distribution of PCBs,
suggest a biphasic kinetic process with rapid clearance from blood,
and accumulation in the liver and the adipose tissue of various
organs. There is also evidence of placental transport, fetal
accumulation, and distribution to milk. In some studies with humans,
the skin was a tissue with a high concentration of PCBs but the
concentration in the brain was lower than would be expected on the
basis of the lipid content.
Mobilization of PCBs from fat appears to depend largely on the rates
of metabolism of the individual PCB congeners. Excretion depends on
the metabolism of PCBs to more polar compounds, such as phenols,
conjugates of thiol compounds, and other water-soluble derivatives.
Metabolic pathways include hydroxylation, conjugation with thiols
and other water-soluble derivatives, some of which can involve
reactive intermediates, such as the arene oxides. The rates of
metabolism have been shown to depend on the PCB structure and
reflect both the degree and position of the chlorine substituents.
The polar metabolites of the more highly chlorinated PCBs appear to
be eliminated primarily in the faeces, but excretion in the urine
can also be significant. An important elimination route, is via
(breast) milk. Certain PCB congeners can also be eliminated via the
hair.
The available kinetic studies indicate that there is a wide
divergence in biological half-life among the individual congeners
and this can reflect differences in structure-dependent metabolism,
tissue affinities, and other factors, affecting mobilization from
storage sites.
Persistence in tissues is not always correlated with high toxicity
and differences in toxicity between PCB congeners may be associated
with specific metabolites and/or their intermediates.
2.4 Effects on organisms in the environment
PCBs are universal environmental contaminants and are present
globally in most environmental compartments, both abiotic and
biotic. Since many countries have controlled both use and release,
new input into the environment is on a much smaller scale, compared
with the past. However, the available evidence suggests that the
cycling of PCBs is causing a gradual redistribution of some
congeners towards the marine environment. There is a trend for the
highest chlorinated congeners to accumulate preferentially. While a
large proportion of the PCBs is adsorbed onto particulates in
sediment, it is still bioavailable to organisms and will continue to
be accumulated in the higher trophic levels.
2.4.1. Laboratory studies
Effects of PCB mixtures on microorganisms are highly variable, with
some species being adversely affected by a concentration of
0.1 mg/litre and others being unaffected by 100 mg/litre; effects on
different species do not vary consistently with the degree of
chlorination of the mixtures.
Almost all of the studies of the effects of PCBs on aquatic
organisms have involved Aroclor mixtures. Results are extremely
variable with no consistent relationship between percentage
chlorination or environmental conditions and toxicity, even with
closely-related organisms. Over 96 h under static conditions, LC50
values ranged between 12 µg/litre and >10 mg/litre for various
aquatic invertebrate species and different Aroclor mixtures.
Flow-through conditions increased the toxicity of the PCBs.
Generally, the most toxic mixtures were Aroclors in the mid-range of
chlorination; low and high percentage chlorination mixtures were
less toxic. This was also true for sublethal effects, such as
reproduction of Daphnia. Crustaceans seem to be more susceptible
to PCBs during moult. In model populations, the community structure
of estuarine species changed on exposure to Aroclor 1254, with
amphipods, bryozoans, crabs, and molluscs decreasing in
representation and annelids, brachyopods, coelenterates, echinoderms
and nemerines being unaffected. Too few of the groups have been
included in acute tests to determine whether the result represents
variation in susceptibility to PCBs or differences in interaction
between species.
There is similar variation in the toxicity of PCB mixtures for fish
with 96-h LC50s varying between 0.008 and >100 mg/litre. Long-term
tests show that acute exposure, particularly under static
conditions, considerably underestimates the toxicity of the PCB.
Rainbow trout were particularly susceptible, with embryo-larval
stages showing a 22-day LC50 of 0.32 µg/litre for Aroclor 1254. The
no-observed-effect level (NOEL) over 22-days for rainbow trout
embryo-larval stages was 0.01 g/litre for Aroclors 1016, 1242, and
1254.
Freshwater fathead minnow showed NOELs of 5.4, 0.1, 1.8, and 1.3
µg/litre for Aroclors 1242, 1248, 1254, and 1260, respectively. The
estuarine sheephead minnow showed NOELs of 3.4 and 0.06 µg/litre for
Aroclors 1016 and 1254, respectively.
Experimental evidence has confirmed field observations demonstrating
reproductive impairment in seals fed on fish containing PCBs,
accumulated in the wild. The effect occurs late in reproduction,
preventing implantation of the embryo in the uterine wall.
In short-term tests, the toxicity of Aroclor for birds increases
with increasing percentage chlorination; 5-day dietary LC50s ranged
from 604 to >6000 mg/kg diet. The main reproductive effects in
birds are reduced hatchability of eggs and embryotoxicity. These
effects of the PCB continue after dosing has ended, as the hens
reduce their PCB load via the eggs. There is no evidence that
Aroclors cause egg-shell thinning directly; effects on the food
consumption and body weight of hens have an indirect effect on shell
thickness. Sublethal effects on behaviour and hormone secretion have
been reported.
For mink, the acute toxicity of Aroclors decreases with increasing
percentage chlorination, acute oral LD50s varying between >750 and
4000 mg/kg body weight; the ferret is less sensitive. Aroclors
reduce food consumption and, thus, the growth rate of young mink.
Reproduction in mink is reduced or eliminated by Aroclors, either
given directly, or as natural contaminants in fish. Aroclors with a
higher percentage chlorination (notably 1254) have a greater effect.
The reproductive rate returns to normal after feeding with Aroclor
is stopped.
Bats are susceptible to Aroclor released from their fat during
migration.
The majority of laboratory tests on aquatic and terrestrial
organisms have been carried out using PCB mixtures and it has not
been possible to identify the specific components of mixtures
responsible for the effects. Similarly, because tests have been
conducted under environmentally unrealistic conditions (e.g., beyond
the solubility of congeners, and, in aquatic tests, without the
presence of sediment) it is difficult to extrapolate from laboratory
tests to the field.
2.4.2 Field studies
Results suggesting that PCBs affect fish populations in the field
are inconclusive. Interpretation of field data on birds is
difficult, since residues of many different organochlorines are also
present. Most authors have shown a correlation between
embryotoxicity and total organochlorine residues. The levels of PCB
residues correlate best with the effects on embryos, but these
results cannot be regarded as proof of a field effect of the PCBs.
There is evidence (confirmed in laboratory studies) that PCBs reduce
the reproductive capacity of sea mammals. The effect is on
implantation of the embryo, but PCBs can also lead to physical
changes in the female reproductive tract.
It is not possible to extrapolate from the results of acute and
short-term laboratory tests to effects on populations in the field.
Uncertainties about which components of the PCB mixtures cause
effects, the specific congeners present in the environment, and the
bioavailability of PCB components to organisms, all combine to make
it difficult to estimate the probable environmental exposure and
effects. The effects on populatons of sea mammals can be regarded as
proved, but it is not yet known which component(s) of the PCB
mixtures are responsible.
Given the trends towards increased contamination of the marine
environment, attention should be concentrated on effects on marine
organisms. There is clear laboratory and field evidence of
reproductive effects of PCBs on populations of sea mammals in
heavily polluted areas, and PCB residues and their effects are
likely to increase in the future. It is less clear whether effects
will be seen in other organisms, such as birds that feed on marine
prey.
On the evidence of laboratory studies, population and community
effects on lower organisms, i.e., phytoplankton and zooplankton,
would be expected to occur. Both the degree and significance of such
effects are difficult to assess. From currently available
information, effects on fish populations would not be expected,
though fish will act as a route of exposure of fish-eating mammals
and birds.
Previously reported effects on terrestrial species, fish-eating
freshwater mammals and migratory bats for example, should be less
evident as the residues of PCBs are redistributed. Residues in
terrestrial biota currently show little decline overall, but
information on changes in congeners is scarce or absent. Levels of
the higher chlorinated congeners would only be expected to decrease
slowly.
2.5 Effects on experimental animals and in vitro systems
2.5.1 Single exposure
The acute toxicity of Aroclors for rats after a single oral exposure
is generally low. Young animals appear to be more sensitive (LD50,
1.3-2.5 g/kg body weight) than adults (LD50, 4-11 g/kg body
weight). The lowest LD50 reported for Aroclor 1254 in adult rats
was 1.0 g/kg body weight. No sex differences were observed.
Dermal LD50s in rabbits ranged from >1.26 to <2 g/kg body weight
for Aroclor 1260 (in corn oil) and from 0.79 to <3.17 g/kg body
weight for some other undiluted PCB mixtures. Intravenous
application demonstrated an LD50 of 0.4 g/kg body weight for
Aroclor 1254 in rats; the LD50, after intraperitoneal injection, in
the mouse varied from 0.9 to 1.2 g/kg body weight.
2.5.2 Short-term exposure
The main targets in mammals with short-term oral exposure to PCB
mixtures or congeners are the liver, the skin, the immune system,
and the reproductive system. The rhesus monkey is the most sensitive
species tested, females being more sensitive than males. Adult
female rhesus monkeys exposed to a diet containing Aroclor 1248 at a
level of 2.5 mg/kg diet, or 0.09 mg/kg body weight per day for 6
months showed an increased mortality rate, growth retardation,
alopecia, acne, swelling of the Meibomian glands, and possibly
immunosuppression. Microscopically, enlarged fatty liver with focal
necrosis, and epithelial hyperplasia and keratinization of hair
follicles were found. At higher exposure levels, microscopic changes
have also been observed in other epithelial tissues, such as
sebaceous and Meibomian glands, gastric mucosa, gall bladder, bile
duct, nail beds, and ameloblast. Serum levels of total lipid
triglycerides and cholesterol were decreased. Short-term exposure to
commercial PCB mixtures induced an increase in the contents and
concentrations of total lipids, triglycerides, cholesterol, and/or
phospholipids in the liver. Among the PCB congeners,
3,4,3',4'-tetrachlorobiphenyl, 3,4,5,3',4',5'-, and
2,4,6,2',4',6'-hexachlorobiphenyl were the most potent. Aroclor
1254, at a dose level of 0.2 mg/kg body weight per day, also showed
several other effects, such as lymphoreticular lesions, fingernail
detachment, and gingival effects, but no acne and alopecia. A
no-observed-effect level (NOEL) for the general toxicity of Aroclor
1242 of 0.04 mg/kg body weight per day was established in rhesus
monkeys. Relatively mild effects were shown in suckling rhesus
monkeys exposed to a much higher dose of Aroclor 1248 of 35 mg/kg
body weight per day. Effects in the liver have been investigated
most thoroughly in rats, and include hypertrophy, fatty
degeneration, proliferation of the endoplasmic reticulum, porphyria,
adeno-fibrosis, bile duct hyperplasia, cysts, and preneoplastic and
neoplastic changes. In studies on rats and mice, individual PCB
congeners caused effects in the liver, spleen, and thymus, the
planar congeners being most toxic. In monkeys, planar congeners, at
doses of 1-3 mg/kg diet, induced effects similar in character and
severity to those seen with Aroclor 1242 at a dose of 100 mg/kg diet
and with Aroclor 1248 at a dose of 25 mg/kg diet.
Following dermal exposure of rabbits and mice, PCB mixtures, and
some congeners, caused effects on the skin and liver similar to
those found after oral exposure. In rabbits, thymic atrophy, a
reduction in the germinal centres of the lymph nodes, and leukopenia
were also observed.
2.5.3 Reproduction, embryotoxicity, and teratogenicity
(a) Reproduction and embryotoxicity
Comprehensive reproduction and teratogenicity studies have not been
conducted. In a two-generation reproduction study on rats, a NOEL of
0.32 mg/kg body weight was established for Aroclor 1254 and a NOEL
of 7.5 mg/kg body weight for Aroclor 1260. However, the lowest
tested dose (0.06 mg/kg body weight) resulted in increased relative
liver weights in weanlings.
In rhesus monkeys exposed to Aroclor 1016, a NOEL of 0.03 mg/kg body
weight was established on the basis of reproductive parameters.
However, decreased birth weight was observed at this level and the
lowest dose tested (0.01 mg/kg body weight) resulted in skin
hyperpigmentation.
In rhesus monkeys, a NOEL of 0.09 mg/kg body weight was established
for Aroclor 1248 (containing PCDFs) 1 year after exposure ceased.
(b) Teratogenicity
Available studies on rats and monkeys did not indicate teratogenic
effects when animals were dosed orally during organogenesis. A NOEL
of 50 mg/kg body weight was demonstrated in rats for Aroclor 1254,
with regard to pup weight, and a lowest-observed-effect level of 2.5
mg/kg body weight, based on fetotoxicity (lesions in thyroid
follicular cells), could be assumed.
In teratogenicity tests of individual congeners on mice, rats, and
Rhesus monkeys, no NOEL was demonstrated. In Rhesus monkeys, a dose
of 0.07 mg/kg body weight indicated maternal toxicity
(3,4,3',4'-tetra-chlorobiphenyl).
2.5.4 Mutagenicity
PCB mixtures did not cause mutation or chromosomal damage in a
variety of test systems. Chromosome breakage was induced in human
lymphocytes in vitro by 3,4,3',4'-tetrachlorobiphenyl. High
concentrations of PCB mixtures may cause primary DNA damage, as
indicated by DNA single strand breaks in alkaline elution assays.
2.5.5 Carcinogenicity
The interpretation of the available animal data involving commercial
PCB mixtures is often complicated by lack of information concerning
the presence or contribution of chlorinated dibenzofuran impurities,
as well as variations in congener composition.
A number of long-term carcinogenicity studies have been carried out
in mice and rats. The PCB mixtures used were Kanechlors 300, 400,
and 500, Aroclors 1254 and 1260, and Clophens A30 and A60. Except
for the Clophens, which were reported to be free of polychlorinated
dibenzo-furans (PCDFs), no data were provided on the purity of the
PCB mixtures used.
A significant increase in hepatocellular adenomas and/or carcinomas
was observed in mice fed with a diet containing Kanechlor 500 and
Aroclor 1254 at a dose level of approximately 15-25 mg/kg body
weight. No neoplasms could be detected in mice treated with
Kanechlors 300 and 400.
In rats, an increase in hepatocellular adenomas and/or carcinomas
was noted in the studies on Aroclors 1254 and 1260 and Clophen A30,
with an exposure period of more than 1 year. The increase in
occurrence of tumours in animals in these studies was not considered
to be statistically significant; however, this was the case in two
other studies. An increase in the incidence of hepatocellular
(trabecular) carcinomas and adenocarcinomas was demonstrated with
Aroclor 1260 and Clophen A60, at a dose level of approximately 5
mg/kg body weight.
The liver tumours concerned were considered to be non-aggressive
(benign or weakly malignant, no metastasis) and not life-shortening.
Adenofibrosis, a pre-neoplastic lesion, and/or neoplastic nodules in
the liver were reported in some of the studies. In one test with
Aroclor 1254, dose-related increases in intestinal metaplasia and
adenocarcinomas of the glandular stomach were demonstrated in the
rat. There is a substantial body of evidence indicating that PCBs
increase the incidence of liver carcinogenesis in rodents
pre-treated with hepatocarcinogens. There is weak evidence that PCB
mixtures initiate carcinogenesis in rodents. From the genotoxicity
studies reported, it can be concluded that PCB-mixtures are
non-genotoxic. These results imply that the association of liver
tumours with administration of PCBs in rodents is attributable to
some epigenitic mechanisms involving enforcement of cell
proliferation in the liver and other manifestations of liver
toxicity, hence a threshold approach can be followed in the
evaluation of PCB toxicity. The possibility that PCBs might increase
carcinogenesis in tissues other than liver in animals pre-exposed to
various tissue-specific carcinogens needs to be addressed. The
anticarcinogenic activities of PCBs shown in some studies, where
PCBs were given to animals during, and prior to, the administration
of carcinogens, may be related to microsomal enzyme-inducing
properties of PCBs that result in an increase of detoxication.
Overall, there is reason to exercise caution in extrapolating to
humans the available animal data on the carcinogenic potential of
PCBs.
2.5.6 Special studies
The lesions induced by exposure to PCB mixtures or individual
congeners concern the liver, skin, immune system, reproductive
system, oedema, and disturbances of the gastrointestinal tract and
thyroid gland.
PCBs are able to induce various enzymes in the liver. This has been
demonstrated in rats, mice, guinea-pigs, rabbits, dogs, and monkeys
for Aroclor 1248, 1254, 1260, and Kanechlor 400 (induction of
cytochrome P450 and P448). The inducing ability increases with the
chlorine content of the molecule; it is also dependent on the
congener composition, where congeners with chlorine in the para
and meta positions induce the P450 enzyme. For AHH (aryl
hydrocarbon hydroxylase) induction, the position of the chlorine
seems to be more important than the degree of chlorination.
Congeners with both para and at least two meta positions
substituted by chlorine are the most potent inducers of AHH.
Distinct interspecies variations have been demonstrated. The lowest
NOEL of 0.025 mg/kg body weight was found for Aroclor 1260 in
Osborn-Mendel rats.
Effects have been demonstrated on the endocrine system, seen as
alterations in hormonal receptor-binding and alterations in steroid
hormone balance. Direct and indirect evidence for a weak estrogenic
activity of various Aroclors has been observed. Decreased levels of
gonadal hormones and increased relative testes weights were found in
rats exposed to 75 mg Aroclor 1242/kg diet for 36 weeks. Decreased
plasma corticosteroid levels without increased adrenal weight, were
found in female mice exposed to Aroclor 1254 (25 mg/kg diet) for 3
weeks. Increased adrenal weight was found in another strain given a
diet containing 200 mg/kg for 2 weeks.
PCB mixtures have been shown to have an immunosuppressive effect in
various animal species. Monkeys and rabbits were the most sensitive
species. The lowest NOEL in monkeys was 0.1 mg/kg body weight, and
that in rabbits, 0.18 mg/kg body weight.
Depressed motor-activity was seen in mice exposed to a single oral
dose of 500 mg Aroclor (1254/kg body weight). This was probably
related to inhibition of uptake and release of neurotransmitters.
PCB mixtures have been found to decrease the levels of vitamins A
and B1 in the blood and liver of rats. Decreased levels of vitamins
A, B1, B2, and B6 were seen in rats and mice exposed to PCB
mixtures.
2.5.7 Factors modifying toxicity; mode of action
Commercial PCBs produce a spectrum of toxic responses, partly
resembling those of polychlorinated dibenzodioxins (PCDDs) and
PCDFs. In addition, the analogous structure-activity relations of
PCB congeners, with respect to most of their toxic responses and to
their potency in inducing P448-dependent AHH (aryl hydrocarbon
hydroxylase), indicate that PCB congeners that are approximate
stereoisomers of 2,3,7,8,-TCDD are the most active. These findings
suggest a common mechanism of action based on the affinity of these
compounds for the cytosolic Ah-receptor protein. Toxic equivalence
factors relating to 2,3,7,8-TCDD have been proposed for these
coplanar PCB congeners. The nature of the likely interactions
between PCBs, PCDFs, and PCDDs has not been investigated adequately.
As PCBs can stimulate microsomal enzyme activity, they can influence
the action of other chemicals that undergo microsomal metabolism.
Other so-called non-planar PCB congeners may cause other more subtle
toxicities. In addition, PCB congeners, especially the lower
chlorinated ones, may be metabolized through arene oxide
intermediates and methylsulfonyl metabolites.
2.6 Effects in humans
The toxicological evaluation of PCBs presents many problems. PCBs
usually occur as mixtures of many congeners, and many of the data on
the toxicity of the PCBs are based on the testing of such mixtures.
Some components of the mixtures are more easily degraded in the
environment than others. Thus, exposure of the general population
may be to mixtures different from those to which the workers are
exposed.
The general population is exposed to PCBs mainly through
contaminated food (aquatic organisms, meat, and dairy products). In
most of the industrialized countries, the daily intake of PCBs is of
the order of some micrograms per person. Such exposure has not been
associated with disease. Infants are exposed to PCBs through their
mothers' milk, and their daily intake of PCBs may be some
micrograms/kg body weight.
There are great difficulties in assessing the human health effects
separately for PCBs, PCDFs, or PCDDs, since, quite frequently, PCBs
contain PCDFs, and occasionally PCDDs have been detected in the
mixtures involved in certain accidents. Commercial PCBs have been
shown to be contaminated with PCDFs and, therefore, in many cases it
is unclear whether effects are attributable to the PCBs or to the
much more toxic PCDFs. Thus, much of the data that can be retrieved
from large intoxication episodes in humans, e.g., the Yusho,
Yu-Cheng, and other intoxications, probably reflect effects of
exposure to both PCDFs and PCBs.
The signs of intoxication in the Yusho and Yu-Cheng patients were
hypersecretion of the Meibomian glands of the eyes, swelling of the
eyelids, and pigmentation of the nails and mucous membranes,
occasionally associated with fatigue, nausea, and vomiting. This was
usually followed by hyperkeratosis and darkening of the skin, with
follicular enlargement and acneform eruptions. In addition, oedema
of the arms and legs, liver enlargement and liver disorders, central
nervous system disturbances, respiratory problems, e.g.,
bronchitis-like disturbances, and changes in the immune status of
the patients, were observed. In children of the Yusho and Yu-Cheng
patients, diminished growth, dark pigmentation of the skin and
mucous membranes, gingival hyperplasia, xenophthalmic oedematous
eyes, dentition at birth, abnormal calcification of the skull,
rocker bottom heel, and a high incidence of low birth weight were
observed. Whether or not a correlation exists between the exposure
and the occurrence of malignant neoplasms in these patients cannot
be definitely concluded, because the number of deaths was too small.
However, a statistically significant increase was observed in male
patients, as regards mortality from all neoplasms, and liver and
lung cancer.
Under occupational conditions, skin rash has occurred a few hours
after acute exposure. Furthermore, itching, burning sensations,
irritation of the conjunctivae, pigmentation of fingers and nails,
and chloracne were found after exposure to high PCB concentrations.
Chloracne is one of the most prevalent findings among PCB-exposed
workers. Besides these dermal signs of intoxication, different
authors have found liver disturbances, immunosuppressive changes,
transient irritation of the mucous membranes of the respiratory
tract, and neurological and unspecific psychological or
psychosomatic effects, such as headache, dizziness, depression,
sleep and memory disturbances, nervousness, fatigue, and impotence.
The overall conclusion is that continuous occupational exposure to
high PCB and PCDF concentrations may result in effects on the skin
and liver.
Two large mortality studies have been carried out with cohorts of
workers exposed to Aroclor 1254, 1242, and 1016. Increased mortality
from cancer of the liver and gall bladder was observed in one study
and from neoplasms and cancer of the gastrointestinal tract in the
other. None of the available epidemiological studies provide
conclusive evidence of an association between PCB exposure and
increased cancer mortality owing to the small number of deaths in
the exposed populations, the lack of dose relationships, and the
problems with contaminants in the PCB mixtures.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
3.1.1 Distribution
Because of their physical and chemical properties, PCBs have become
dispersed globally throughout the environment.
PCBs are almost universally present in organisms in the environment
and are readily bioaccumulated. Biomagnification in food-chains has
also been demonstrated.
Higher chlorinated congeners accumulate preferentially.
3.1.2 Effects on experimental animals
Animal studies suggest that PCBs are immunosuppressive, as assessed
by alterations in gross measures of immune function (spleen weight,
thymus weight, or lymphocyte count). NOELs have been estimated in
monkeys at 100 µg/kg body weight for Aroclor 1248 and <100 µg/kg
body weight for Aroclor 1254. Immunosuppression appears to be a
congener-specific effect.
Reproductive toxicity is, in general, only seen at doses producing
systemic toxicity in the mother. Neonates feeding on contaminated
mother's milk (in monkeys and other animal species used as models)
appear to be particularly sensitive to PCBs and show reduced growth,
with other toxic symptoms. The NOEL for Aroclor 1016 in monkeys (on
the basis of reproductive effects) is 30 µg/kg body weight; no NOEL
could be established for reproductive effects of Aroclor 1248.
PCBs are not genotoxic and the evidence for any action as tumour
initiators is inconclusive. PCBs do act as tumour promoters. The
toxicity of PCB mixtures can be evaluated on a threshold basis.
3.1.3 Effects on humans
Exposure of the general population to PCBs is principally through
food items. Babies are exposed through the mother's milk.
Two large intoxication episodes in humans have occurred in Japan
(Yusho) and Taiwan (Yu-Cheng). The main symptoms of the Yusho and
Yu-Cheng patients have frequently been attributed mainly to the
contaminants of the PCB mixtures; specifically to PCDFs. The Task
Group concluded that the symptoms may have been caused by the
combined exposure to PCBs and PCDFs. Some of the symptoms, and
principally the chronic respiratory effects, may have been caused
specifically by the methylsulfone metabolites of certain PCB
congeners.
3.1.4 Effects on the environment
While there have been reports of effects on populations of birds,
the most important effect of PCBs on organisms in the environment is
reproductive failure in sea mammals. This has been observed
principally in semi-enclosed seas and has led to local decreases in
populations. The prediction that residues of PCBs in the environment
will gradually be redistributed towards the marine environment
indicates an increasing hazard for sea mammals in the future.
3.2 Recommendations
* International agreement on analytical procedures to improve the
comparability of results of monitoring programmes is
recommended. Development of methods for congener-specific
analysis should be continued, though the value of analysis
based on mixtures is recognized.
* In order to ensure reliability of analytical data,
inter-laboratory quality control studies are strongly
recommended. It is also recommended that an international
network of technical support and supervision should be
established to allow developing countries to participate in
monitoring.
* Long-term studies using specific congeners and studies on the
mechanism of action of constituents of PCBs mixtures, with
special regard to tumour promotion, are recommended to improve
the precision of risk assessment of PCBs.
* Epidemiological studies to improve the assessment of the risk
to neonates are required, since newborn infants appear to be
the most vulnerable sector of the general population, because
of high exposure through milk.
* Sensitive and specific biomarkers for some of the more subtle
aspects of PCB toxicity (such as reproductive, immunological,
and neural toxicity) should be developed for use in future
epidemiological studies
* PCBs should be disposed of by incineration in properly designed
and run facilities that can guarantee the constant high
temperatures (above 1000 °C), residence time, and turbulence
needed to ensure complete breakdown.
* Methods to remove PCBs already contained in landfills should be
investigated.
* Worldwide monitoring of PCBs in the environment and in wildlife
should be encouraged, to monitor the expected redistribution of
residues already present.
* Marine mammals show evidence of reproductive failure as a
result of PCB contamination. Studies on population size and
reproductive success of cetaceans should be encouraged,
together with further research to establish those congeners
responsible for the effects.
4. HEALTH HAZARDS FOR MAN, PREVENTION AND PROTECTION, EMERGENCY
ACTION
For a more detailed treatment of prevention and control of
accidental and environmental exposures to PCBs and PCTs, the reader
should refer to the WHO/EURO document listed in the Bibliography. A
detailed description of the human and environmental hazards of PCBs
is given in EHC 140 (WHO, in preparation).
4.1 Main human health hazards, prevention and protection, first aid
PCBs and PCTs are highly chlorinated organic substances. They are
very persistent and may be hazardous for human beings if incorrectly
or carelessly handled. It is, therefore, essential that the correct
precautions are observed during handling, use, and disposal.
For details see the Summary of Chemical Safety Information in
section 6.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
The acute oral and dermal toxicity is low, but under occupational
conditions skin rash may occur a few hours after acute exposure.
Furthermore, itching, burning sensations, irritation of the
conjunctivae, pigmentation of fingers and nails, and (chlor)acne
were found after exposure to high PCB concentrations for long
periods. Massive doses can cause hepatitis, facial oedema, numbness,
and weakness of the extremities. Chloracne is one of the most
prevalent findings among workers exposed to PCBs, but may be due to
the presence of PCDFs in the technical PCB mixtures. In addition to
these dermal signs of intoxication, liver disturbances,
immunosuppressive changes, transient irritation of the mucous
membranes of the respiratory tract, neurological and unspecific
effects, such as headache, dizziness, depression, sleep and memory
disturbances, nervousness, fatigue, and impotence, have been
reported.
4.1.1.2 Medical advice
Medical treatment is symptomatic and supportive.
4.1.2 Health surveillance advice
A complete medical history and physical examination of workers
regularly exposed to PCBs should be made annually. Special attention
should be paid to the skin and to liver function.
4.2 Explosion and fire hazards
Fires and explosions involving PCBs have been reported mainly from
their use in electrical equipment, such as transformers and
capacitors, but PCBs may also be involved in fires during storage
and transport. Fires may lead to the formation of highly toxic
polychlorinated dibenzofurans (PCDFs). In dielectric fluid
formulations, which also contain various tri- or
tetra-chlorobenzenes, polychlorinated dibenzo- p-dioxins (PCDDs)
may be formed upon fire or explosion.
Fires should be extinguished with alcohol-resistant foam, carbon
dioxide, or powder. With sufficient burning or external heat, PCBs
will decompose, emitting very toxic fumes. Fire-fighters should be
equipped with self-contained breathing apparatus, eye protection,
and full protective clothing.
The use of water sprays should be confined to the cooling of
unaffected stock, thus avoiding the accumulation of polluted run-off
from the site.
4.3 Storage
Products should be stored in well ventilated, locked buildings, out
of the reach of children and unauthorized personnel. Do not store
near foodstuffs or animal feed.
4.3.1 Leaking containers in store
Take precautions, and use appropriate personal protection (see
section 6). Empty any product remaining in damaged or leaking
containers into a clean empty drum, which should then be tightly
closed and suitably labelled. Sweep up spillage with sawdust, sand,
or earth (moisten for powders), and dispose of safely.
4.4 Transport
Comply with any local requirements regarding the movement of
hazardous goods. Do not transport in the same compartment as
foodstuffs. Check that the containers are sound and labels undamaged
before despatch.
4.5 Spillage and disposal
4.5.1 Spillage
Before dealing with any spillage, precautions should be taken, as
required, and appropriate personal protection should be used (see
section 6).
Prevent material from spreading or contaminating other cargo,
vegetation, or waterways, by making a barrier of the most suitable
available material, e.g., earth or sand. Absorb the spilled liquid
with sawdust, sand, or earth, sweep up and place the contaminated
material in a closeable container for later transfer to a safe place
for disposal. Care should be taken to avoid run-off into water
courses.
4.5.2 Disposal
Dielectric fluids containing PCBs in transformers and capacitors
should be recovered and sent for destruction.
Any surplus product, contaminated absorbants, and containers should
be disposed of in an appropriate way. Waste material should be
burned in a proper incinerator designed for organochlorine waste
disposal, with effluent gas-scrubbing. For PCB wastes, incineration
must be for more than 2 seconds at 1200 °C or higher. Cement kilns
may meet the required temperature/time conditions and may be
properly constructed for this purpose. If the PCB content of the
waste is less than 500 mg/kg, any proper waste incinerator can be
used as long as temperature exceeds 800 °C for 0.5 seconds.
Combustion of PCBs can produce dibenzofurans; PCB dielectric fluids
also containing tri- or tetrachlorobenzenes can also produce
dioxins. If proper incineration is not possible, bury in an approved
dump or landfill where there is no risk of contamination of surface
or ground water. Decomposition of PCBs is extremely slow.a
Comply with any local legislation regarding disposal of toxic
wastes. Puncture and crush containers, to prevent re-use.
a For a more complete treatment of the subject, refer to
WHO/Euro, 1987.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
5.1 Hazards
PCBs and PCTs are very resistant to degradation and hence very
persistent in the environment. Because they are very soluble in
lipids they bioaccumulate, especially in the fatty tissues of all
living organisms, and biomagnify in the higher trophic levels of the
food-chain.
Although their acute toxicity is relatively low, bioaccumulation and
biomagnification may lead to lethal effects, especially at the
highest trophic levels. Reduced growth and reproduction may affect
populations.
5.2 Prevention
PCBs and PCTs should be replaced by alternative products wherever
practicable.
Industrial discharges occurring during manufacture, formulation, or
technical applications should not be allowed to pollute the
environment and should be treated properly.
Any spillage or unused product should be prevented from spreading to
vegetation or waterways, and should be treated and disposed of
properly.
In all cases, immediate remedial action is essential.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, PCBs and PCTs. It should be
displayed at, or near, entrances to areas where there is
potential exposure to PCBs and PCTs, and on processing
equipment and containers. The summary should be translated
into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions in
the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the
National Poison Control Centre, and local trade names.
SUMMARY OF CHEMICAL SAFETY INFORMATION
Polychlorinated biphenyls (PCBs) Polychlorinated terphenyls (PCTs)
Molecular formula: C12H10-nCln Molecular formula: C18H14-nCln
CAS registry number: 1336-36-3 CAS registry number: 61788-33-8
RTECS registry number: TQ1350000 RTECS registry number: WZ6500000
PHYSICAL PROPERTIES AND OTHER CHARACTERISTICS
Commercial PCBs and PCTs are complex mixtures of many different congeners with various degrees of chlorination. They are
not produced to a composition specification; the criteria for use are based on physical properties. They are clear, light
yellow, or dark liquids that may turn into solid resin at low temperatures.
Their distillation range is in general above 250 °C. Their relative molecular mass and density depend on the degree of
chlorination. They are very fire-resistant, with flash-points above 170 °C. Their vapours are heavier than air, but do not
form explosive mixtures. Their electrical conductivity is very low and their resistance to thermal breakdown extremely
high. They are practically insoluble in water, easily miscible with most organic solvents, and accumulate in fatty tissues.
They have high n-octanol/water partition coefficents.
They are mainly used as dielectrics in transformers and capacitors, in heat transfer and hydraulic systems, and, to a
lesser extent, in lubricating and cutting oils, carbonless copying paper, adhesives, sealants, plastics, and as
plasticizers in paints.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: May cause irritation Avoid contact with skin; wear After contact with skin, wash immediately
and chloracne suitable protective clothing with plenty of water and soap; immediately
and gloves remove all contaminated clothing and
launder before reuse
EYES: May cause irritation Avoid contact with eyes; wear In case of contact with eyes, rinse
eye protection immediately with plenty of water and seek
medical advice
INHALATION: May cause irritation Adequate ventilation;
do not breathe vapours
INGESTION: Unlikely occupational Do not eat, drink, or smoke
hazard during work; wash hands before
eating, drinking, or smoking
Accidental or intentional If swallowed, seek medical advice
ingestion may cause poisoning immediately and show container or label
ENVIRONMENT: Bioaccumulates Strictly avoid environmental
and biomagnifies pollution
SPILLAGE STORAGEFIRE AND EXPLOSION
Take appropriate personal Products should be stored in Extinguish fires with alcohol-resistant
precautions; prevent liquid from well ventilated locked buildings; foam, CO2, or powder; with sufficient
spreading or contaminating other keep out of reach of children; burning or external heat, PCBs and
cargo, vegetation, or surface keep away from food, drink, and PCTs will decompose, emitting toxic fumes;
waters and drainage systems, animal feeding stuffs firefighters should be equipped with
with a barrier of most suitable self-contained breathing apparatus, eye
suitable material, e.g., earth or protection, and full protective clothing;
sand confine the use of water spray to cooling
of unaffected stock, thus avoiding the
Absorb spilled liquid with saw- accumulation of polluted run-off from the
dust, sand, or earth; sweep up and site
place it in a closeable container
for later safe disposal; care
should be taken to avoid run-off
into water courses
WASTE DISPOSAL NATIONAL INFORMATION
PCBs and PCTs waste material National Occupational Exposure UN No. 2315
should be burned in a proper Limit:
incinerator designed for
organochlorine waste disposal; National Poison Control Centre:
if this is not possible, bury in
an approved dump or landfill Local trade names:
where there is no risk of
contamination of surface or
ground water; comply with any
local legislation regarding
disposal of toxic wastes
7. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC. When no effective date appears in the
IRPTC legal file, the year of the reference from which the data are
taken is indicated in the table by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
7.1 Previous evaluations by international bodies
PCBs have been evaluated by IARC in 1978 and 1987 (IARC, 1978, 1987
(Supplement)). It was concluded that there was sufficient
experimental evidence to indicate a carcinogenic effect of some PCBs
in rodents, and that epidemiological data provided suggestive
evidence of a relationship between exposure to PCBs and the
development of certain cancers in man. PCBs were classified in group
2A: the agent is probably carcinogenic for humans. For practical
purposes, PCBs should be regarded as if they were carcinogenic for
human beings.
7.2 Exposure limit values
Some exposure limit values for PCBs are given in the table on pages
43-45.
CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Exposure limit values
Medium Specification Country Exposure limit description Value Effective
date
AIR Workplace Czechoslovakia Maximum permissible concentration
- time-weighted average (TWA) 0.5 mg/m3 1985
- short-term exposure limit (STEL) 1.0 mg/m3
Japan Maximum permissible concentration
- time-weighted average (TWA) 0.1 mg/m3 a 1985
Netherlands Maximum allowable concentration (MAC)
- time-weighted average (TWA) 0.5 mg/m3 -
Sweden Occupational exposure limit
- time-weighted average (TWA) 0.01 mg/m3 a 1985
- short-term exposure limit (STEL) 0.03 mg/m3
USSR Maximum permissible concentration
- time-weighted average (TWA) (vapour) 1.0 mg/m3 1977
United Kingdom Recommended limit
- time-weighted average (TWA) 0.5 mg/m3 -
- short-term exposure limit (STEL) 1.0 mg/m3
(10-min TWA)
Medium Specification Country Exposure limit description Value Effective
date
USA (ACGIH) Threshold limit value (TLV)
- time-weighted average (TWA) 0.5 mg/m3 -
- short-term exposure limit (STEL) 1.0 mg/m3
FOOD Animal Germany, Maximum residue limit (MRL) 0.008-0.6 1988
Federal (specified products) mg/kg
Republic of
Sweden Maximum residue limit (MRL) (Specified) 0.05-2 mg/kg 1983
FOOD Animal & France Maximum residue limit (MRL) (fish) 2 mg/kg -
plant
Japan Maximum residue limit (MRL) (Specified) 0.2-3 mg/kg -
Netherlands Maximum residue limit (MRL) (Specified) 0.3-1 mg/kg -
Switzerland Maximum residue limit (MRL) (Specified) 0.5-2 mg/kg -
USA Temporary residue tolerance (Specified) 0.2-3 mg/kg 1981
FEED Japan Maximum residue limit (MRL) (Specified) 0.05-3 mg/kg -
Netherlands Maximum residue limit (MRL) (Specified) 0.3 mg/kg -
USA Maximum residue limit (MRL) (Specified) 0.2-2 mg/kg -
WATER Drinking- EEC Maximum permissible concentration
(total PCBs + PCTs) 0.5 µg/litre 1982
Medium Specification Country Exposure limit description Value Effective
date
Environment Japan Environment water quality standard not detectable 1981
Surface water USSR Permissible limit 0.0 mg/litre 1978
for fishing
Effluent Japan Effluent standard 0.003 mg/litre 1981
GOODS Packaging USA Temporary residue tolerance 10 mg/kg 1983
material for
food or feed
a Skin absorption.
7.3 Specific restrictions
Several intergovernmental organizations have been active in
providing directives or recommendations for regulatory measures to
control PCBs.
On 13 February 1973, the Council of the Organisation for Economic
Cooperation and Development (OECD) adopted a Decision on the
Protection of the Environment by Control of Polychlorinated
Biphenyls (C(73)1). The Council decided that PCBs would not be used
for industrial or commercial purposes, except for five essentially
closed purposes. These are: dielectric fluids in transformers; large
power-correcting capacitors; heat-transfer fluids (but only in
installations that do not process food, feed, pharmaceuticals, or
veterinary products); hydraulic fluids (but only in mining
equipment); and in small capacitors (though Member Countries have
recommended working towards the elimination of this last use). PCBs
should only be used in the exempted applications where
non-flammability requirements outweigh the environmental protection
considerations and where sufficient controls are exercised to
minimize risk to the environment. The Council also made some
recommendations concerning the elimination of other uses of PCBs and
PCB replacements. It also provided for certain administrative and
engineering control measures for PCBs still in use, and for the
disposal of PCB wastes. The OECD Council decision provided for an
exchange of information on PCBs between Member Countries within the
framework of the OECD Environment Committee and information on PCBs
was exchanged annually between 1974 and 1980. The information
exchanged and the experience gained by Member Countries was
summarized in a report (OECD, 1982). This report indicated that,
while considerable progress had been made in reducing environmental
contamination by PCBs, some important problems remained.
An extensive synopsis of national regulatory measures in a number of
countries was prepared by the OECD in 1982. Countries have rather
complex and very different systems to control PCBs in the general
environment. Most regulations impose usage restrictions and
prescriptions for transportation and labelling, require notification
of production and/or importation, and provide rules for the disposal
of PCB-containing wastes.
On 13 February 1987, the OECD Council adopted a further
Decision-Recommendation (C(87)2(final)) on "Further measures for the
protection of the environment by control of polychlorinated
biphenyls". With this Decision-Recommendation, the OECD Member
Countries committed themselves to ban virtually all new uses for
PCBs, accelerate the phasing out of PCBs from existing uses, control
PCBs in contaminated products, articles, or equipment, and ensure
appropriate disposal methods for wastes containing PCBs.
In the countries of the European Economic Community, the use of PCBs
and PCTs is prohibited by Directive 85/467/EEC (6th Amendment (PCBs
and PCTs) Directive 76/769/EEC) but until 30 June 1986 the following
uses were excepted: (a) closed-system electrical equipment; (b)
large condensers; (c) small condensers (provided that the PCB has a
maximum chlorine content of 43% and does not contain more than 3.5%
of penta- and higher chlorinated biphenyls); (d) heat-transmitting
fluids in closed-circuit heat-transfer installations; (e) hydraulic
fluids used in underground mining equipment; (f) primary and
intermediate products for further processing into other products
which are not prohibited under the Directive. The use of equipment,
plant, and fluids referred to in points a to f above that were in
service on 30 June 1986 shall continue to be authorized until they
are disposed of or reach the end of their service life. Derogations
considered to have no deleterious effects on health or the
environment could be granted after 30 June 1986. These provisions
apply to PCBs and PCTs (except mono- and dichlorinated biphenyls)
and preparations with a PCB or PCT content higher than 0.01% by
weight.
Apart from the above restrictions in OECD and EEC countries, several
other countries have similar, more or less severe restrictions on
the use of PCBs (and PCTs). In Japan, the manufacture and import of
all PCBs is prohibited without authorization from the Government. In
the USA, the manufacture, processing, distribution in commerce and
use of PCBs is prohibited without Government authorization.
7.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies PCBs in:
Hazard Class 9: Miscellaneous dangerous substance.
Packing Group II: Substances presenting medium danger.
The European Economic Community legislation requires the labelling
of PCBs and PCTs as harmful substances using the symbol:
The chemical formula can be written as C18H14-nCln, in which n
is the number of chlorine atoms, which can range from 1-14.
Chemical composition
The theoretically possible number of different PCTs is several
orders of magnitude greater than the number of PCBs, but in
practice, as with PCBs, PCTs are sold on the basis of their physical
properties, which depend on the degree of chlorination, and not
their chemical composition.
Common name: Polychlorinated terphenyls - PCTs.
CAS registry number: 61788-33-8
RTECS registry number: WZ6500000
Relative molecular mass: Depends on degree of chlorination and
composition of the mixture.
Main trade names
PCTs are known by a variety of trade names, some of which are
similar to those given for PCBs in Section 1.1.1. In the Aroclor
series, terphenyls are indicated by 54 in the first two places of
the four digit code. In Japan, the PCTs are coded Kanechlor KC-C.
1.2 Physical and chemical properties
Individual, pure congeners of PCB and PCT are colourless, often
crystalline compounds, but commercial PCBs are mixtures of these
congeners with a clear, light yellow or dark colour, and range from
oily liquids to waxy or hard solids. They do not crystallize at low
temperatures, but turn into solid resins. Because of the chlorine
atoms in the molecule, the compounds have a fairly high density. In
practice, PCBs are fire resistant, and have a fairly high
flash-point (170-380 °C). They form vapours that are heavier than
air, but they do not form an explosive mixture with air. The
electrical conductivity of PCBs and PCTs is very low, and their
resistance to thermal breakdown is extremely high. It is on the
basis of these properties that they are used as cooling liquids in
electrical equipment. The physical properties of some Aroclors are
shown in Table 1.
PCBs are chemically stable under normal conditions. They are very
resistant to a range of oxidants and other chemicals, and they
remain chemically unchanged even in the presence of oxygen, or some
active metals, at high temperatures (up to 170 °C), and for
protracted periods.
PCBs are practically insoluble in water; however, they dissolve
easily in hydrocarbons, fats, and other organic compounds, and they
are readily adsorbed by fatty tissues.
The partition coefficient (log Kow) values for all 209 PCB
congeners range from 4.46 to 8.18.
1.3 Analytical methods
Only a small number of laboratories in the world have access to, and
experience of working with, the complicated techniques necessary for
a reproducible determination of PCBs and PCTs.
One probable source of error is incomplete extraction and clean-up
of the PCBs. The method used to quantify the gas-liquid
chromatographic peaks also gives rise to variation between
laboratories.
Data on concentrations of PCBs must be interpreted with the greatest
care. Comparisons can only be made between data from the same
laboratory, obtained using the same validated technique over a long
period. Comparisons between data from different laboratories are
possible in only the very few cases, in which very strict
interlaboratory checks have been made on the basis of the same
sampling and analytical techniques. Indications about trends can
only be obtained when these basic considerations are taken into
account.
Gas-liquid chromatography (GLC) with packed or capillary columns, is
generally used for the analysis, and comparison of peak patterns,
and various PCB standard formulations are used for quantification.
Different approaches are used for the summing-up of individual
peaks.
Analytical methods are discussed in more detail in the WHO/EURO
(1987) document.
Individual congeners are identified using GLC, with either hydrogen
flame ionization detection (HFID) or electron capture detection
(ECD), and mass spectrometry.
Table 1. Physical properties of some Aroclors
Substance Water Vapour Density Appearance Henry's Law Refractive index Boiling point
Aroclor solubility pressure g/cm3 constant (distillation
(mg/litre, (torr, (25 °C) (atm-m3/mol range)
25 °C) 25 °C) at 25 °C) (750 torr, °C)
1016 0.42 4.0 x 10-4 1.33 Clear, 2.9 x 10-4 1.6215-1.6135 325-356
mobile oil (at 25 °C)
1221 0.59 6.7 x 10-3 1.15 Clear, 3.5 x 10-3 1.617-1.618 275-320
mobile oil
1232 0.45 4.1 x 10-3 1.24 Clear, unknown unknown 290-325
mobile oil
1242 0.24 4.1 x 10-3 1.35 Clear, 5.2 x 10-4 1.627-1.629 325-366
mobile oil (at 20 °C)
1248 0.054 4.9 x 10-4 1.41 Clear, 28 x 10-3 unknown 340-375
mobile oil
1254 0.021 7.7 x 10-5 1.50 Light yellow, 2.0 x 10-3 1.6375-1.6415 365-390
viscous oil (at 25 °C)
1260 0.0027 4.0 x 10-5 1.58 Light yellow, 4.6 x 10-3 unknown 385-420
sticky resin
1.4 Uses
The industrial usefulness of PCBs and PCTs depends on their chemical
inertness, resistance to heat, non-flammability, low vapour pressure
(particularly with the higher chlorinated compounds), and high
dielectric constant.
The main uses are (or were):
(a) as dielectrics in transformers and large capacitors (considered
to be closed systems);
(b) in heat transfer and hydraulic systems (nominally closed
systems);
(c) in the formulation of lubricating and cutting oils;
(d) as plasticizers in paints, carbonless copying paper, adhesives,
sealants, and plastics.
Both c and d are open-ended applications.
2. SUMMARY AND EVALUATION
2.1 Environmental transport, distribution, and transformation
In the atmosphere, PCBs exist primarily in the vapour phase; the
tendency to adsorb to particulates increases with the degree of
chlorination. The virtually universal distribution of PCBs suggests
that they are transported in air.
At present, the major source of PCB exposure for the general
population appears to be as a consequence of the redistribution of
PCBs previously introduced into the environment. This redistribution
involves volatilization from soil and water into the atmosphere,
with subsequent transport in air and removal from the atmosphere
through wet or dry deposition (of PCBs bound to particulates), and
then re-volatilization. The concentrations of PCBs in precipitation
range from 0.001 to 0.25 µg/litre. Since the volatilization and
degradation rates of PCBs vary among the different congeners, this
redistribution leads to an alteration in the composition of PCB
mixtures in the environment.
In water, PCBs are adsorbed to sediments and other organic matter;
experimental and monitoring data have shown that PCB concentrations
are higher in sediment and suspended matter than in the associated
water columns. Strong adsorption to sediment, especially in the case
of the higher chlorinated PCBs, decreases the rate of
volatilization. On the basis of their water solubilities and
n-octanol-water partition coefficients, the lower chlorinated PCB
congeners will sorb less strongly than the higher chlorinated
isomers. Although adsorption can immobilize PCBs for relatively long
periods in the aquatic environment, desorption into the water column
has been shown to occur by both the abiotic and biotic routes. The
substantial quantities of PCBs in aquatic sediments therefore act as
both an environmental sink and a reservoir of PCBs for subsequent
recycling in the environment. Most of the PCBs in the environment
are in the aquatic sediment.
The low solubility of PCBs, and their strong adsorption to soil
particles, limits leaching in soil; the lower chlorinated PCBs will
tend to leach more than the highly chlorinated PCBs.
Degradation of PCBs in the environment depends on the degree of
chlorination of the biphenyl. In general, the persistence of PCB
congeners increases as the degree of chlorination increases. In the
atmosphere, the reaction of PCBs in the vapour phase with hydroxyl
radicals (which are photochemically formed by sunlight) may be the
most important transformation process. The estimated half-life of
this reaction in the atmosphere ranges from about 10 days to 1.5
years for a monochlorobiphenyl and a heptachlorobiphenyl,
respectively.
In the aquatic environment, PCBs are not significantly degraded by
hydrolysis and oxidation. Photolysis appears to be the only abiotic
degradation process in water; however, insufficient experimental
data are available to determine its rate or its importance in the
environment.
Microorganisms degrade mono-, di-, and trichlorinated biphenyls
relatively rapidly, and tetrachlorobiphenyls slowly, while higher
chlorinated biphenyls are resistant to biodegradation. The chlorine
substitution position on the biphenyl ring appears to be important
in determining the biodegradation rate. PCBs containing chlorine
atoms in the para position are preferentially biodegraded. Higher
chlorinated congeners are biotransformed anaerobically, by reductive
dechlorination, to lower chlorinated PCBs, which may then be
biodegradable by aerobic processes.
Several factors determine the degree of bioaccumulation in adipose
tissues: duration and level of exposure, chemical structure of the
compound, and position and pattern of substitution. In general, the
higher chlorinated congeners are accumulated more readily.
The bioconcentration factors of various PCBs determined
experimentally in aquatic species (fish, shrimp, oyster) range from
200 up to 70 000 or higher. In the open ocean, there is
bioaccumulation of PCBs in the higher trophic levels, with an
increased proportion of higher chlorinated biphenyls in the higher
ranking predators.
Transfer of PCBs from soil to vegetation takes place mainly through
adsorption onto the external surfaces of terrestrial plants; little
translocation takes place.
2.2 Environmental levels and human exposure
Because they persist for a long time, and because of other
physicochemical properties, PCBs are present in the environment all
over the world.
PCBs are found in air, all over the world, at concentrations of
0.002 up to 15 ng/m3. In industrial areas, higher levels of up to
micrograms/m3 are found. In rain-water and snow, PCBs are found in
the range of not detectable (<ng/litre) to 250 ng/litre.
Under workplace conditions, the levels in the air may be much
higher. In the manufacturing of transformers or capacitors, for
instance, levels of up to 1000 µg/m3 have been observed. In
emergency situations, concentrations of up to 16 mg/m3 have been
measured. In the case of fires and/or explosions, the soot may
contain high levels of PCBs; concentrations of 8000 mg PCBs/kg soot
have been found. In fires and explosions, PCDFs will also be present
in the soot. In accidents with transformers in which chlorinated
benzenes are present in addition to PCBs, polychlorinated dioxins
(PCDDs) will also be found.
In these emergency situations, skin contamination with soot is
possible, and ingestion or inhalation of soot particles may occur
and result in serious exposures of personnel. However, the exposure
of the general population through the air will be very low.
Surface water may be contaminated by PCBs by atmospheric fall-out,
or by direct emissions from point sources, or waste disposal. Under
certain conditions, levels of up to 100-500 ng/litre of water have
been measured. In the water of oceans, levels of 0.05-0.6 ng/litre
have been found.
In non-contaminated areas, drinking-water usually contains less than
1 ng PCBs/litre, but levels up to 5 ng/litre have been reported.
Soil and sediments normally contain concentrations of PCBs in the
range of <0.01-2.0 mg/kg. In polluted areas, the levels in soils
have been much higher, up to 500 mg/kg.
Over the years, many thousands of samples of different foodstuffs
have been analysed, in several countries, for contaminants,
including PCBs. Most samples have been taken from individual food
items, especially fish and other foods of animal origin, such as
meat and milk. Food becomes contaminated with PCBs by three main
routes:
a) uptake from the environment by fish, birds, livestock (via
food-chains), and crops;
b) migration from packaging materials into food (mainly less than 1
mg/kg, but in some cases up to 10 mg/kg);
c) direct contamination of foodstuffs or animal feed as the result
of an industrial accident.
For the most important food items that contain PCBs, the following
concentrations have been found: animal fat, 20-240 µg/kg; cow's
milk, 5-200 µg/kg; butter, 30-80 µg/kg; fish, 10-500 µg/kg, on fat
basis. Certain fish species (eel) and fish products (fish liver and
fish oils) contained much higher levels, up to 10 mg PCBs/kg. Levels
of <10 µg/kg were found in vegetables, cereals, fruits, and a
number of other products. Fish, shellfish, meat, milk, and other
dairy products are the main foods that give rise to concern as
regards levels of PCBs. The median levels reported in fish, in
various countries, are in the order of 100 µg/kg (on fat basis);
however, it appears that the levels of PCBs in fish are slowly
decreasing.
PCBs accumulate in human adipose tissue and breast milk. The
concentrations of PCBs in different organs and tissues depend upon
the lipid content of the organ or tissue, with the exception of the
brain. The levels of PCB residues in adipose tissue of the general
population in industrialized countries range from <1 to 5 mg/kg, on
fat basis.
The average concentration of total PCBs in human milk is in the
range of 0.5 to 1.5 mg/kg fat, depending on the donor's place of
residence, life-style, and the analytical methods used. Women living
in heavily industrialized urban areas, or with a high fish
consumption (especially fish from heavily contaminated waters), may
have higher PCB concentrations in breast milk.
The composition of most PCB extracts from environmental samples does
not resemble that of the commercial PCB mixtures. High-resolution
gas chromatography (GC) analysis shows that the congener
composition, and relative concentrations of the individual
components, in adipose tissues and breast milk differ markedly from
the composition of commercial PCBs. The GC-patterns of PCBs in human
adipose tissues and breast milk indicate relatively high
concentrations of mainly the higher chlorinated PCBs, such as,
2,4,5,3',4'-pentachlorobiphenyl; 2,4,5,2',4',5'-hexa-
chlorobiphenyl; and 2,3,4,2',4',5'-hexachlorobiphenyl;
2,3,4,5,2',4',5'-hepta- and 2,3,4,5,2',3',4'-heptachlorobiphenyl. A
few other PCB congeners are present at much lower concentrations,
including the most toxic, coplanar PCBs: 3,4,3',4'-tetra-,
3,4,5,3',4'-penta- and 3,4,5,3',4',5'-hexahlorobiphenyl.
The daily intake of PCBs by infants from breast milk is of the order
of 4.2 µg/kg body weight (5.2 µg/100 kcal consumed) (WHO/EURO,
1987). The average total quantity of PCBs ingested in breast milk
during the first 6 months of life is 4.5 mg compared with a
calculated intake of 357 mg of PCBs over the subsequent life-time
(0.2 µg/kg per day in the diet of a 70-kg person over a 70-year
life-time). Therefore, the nursing period contributes about 1.3% of
the life-time intake, which is not large in light of the benefits of
breast-feeding (WHO/EURO, 1987).
On the basis of the evaluated background data, the average dietary
intake of PCBs for adults amounts to a maximum of 100 µg/week, or
approximately 14 µg/person per day. For a 70-kg person, this is an
intake equivalent to a maximum of 0.2 µg/kg body weight per day
(WHO/EURO, 1987).
2.3 Kinetics and metabolism
Animal studies have been reported involving mainly oral, inhalation,
and dermal exposures to both PCB mixtures and individual congeners.
In general, PCBs appear to be rapidly absorbed, particularly by the
gastro-intestinal tract after oral exposure. It is clear that
absorption does occur in humans, but information on the rates of
absorption of PCBs in humans is limited.
From the available studies, the data on the distribution of PCBs,
suggest a biphasic kinetic process with rapid clearance from blood,
and accumulation in the liver and the adipose tissue of various
organs. There is also evidence of placental transport, fetal
accumulation, and distribution to milk. In some studies with humans,
the skin was a tissue with a high concentration of PCBs but the
concentration in the brain was lower than would be expected on the
basis of the lipid content.
Mobilization of PCBs from fat appears to depend largely on the rates
of metabolism of the individual PCB congeners. Excretion depends on
the metabolism of PCBs to more polar compounds, such as phenols,
conjugates of thiol compounds, and other water-soluble derivatives.
Metabolic pathways include hydroxylation, conjugation with thiols
and other water-soluble derivatives, some of which can involve
reactive intermediates, such as the arene oxides. The rates of
metabolism have been shown to depend on the PCB structure and
reflect both the degree and position of the chlorine substituents.
The polar metabolites of the more highly chlorinated PCBs appear to
be eliminated primarily in the faeces, but excretion in the urine
can also be significant. An important elimination route, is via
(breast) milk. Certain PCB congeners can also be eliminated via the
hair.
The available kinetic studies indicate that there is a wide
divergence in biological half-life among the individual congeners
and this can reflect differences in structure-dependent metabolism,
tissue affinities, and other factors, affecting mobilization from
storage sites.
Persistence in tissues is not always correlated with high toxicity
and differences in toxicity between PCB congeners may be associated
with specific metabolites and/or their intermediates.
2.4 Effects on organisms in the environment
PCBs are universal environmental contaminants and are present
globally in most environmental compartments, both abiotic and
biotic. Since many countries have controlled both use and release,
new input into the environment is on a much smaller scale, compared
with the past. However, the available evidence suggests that the
cycling of PCBs is causing a gradual redistribution of some
congeners towards the marine environment. There is a trend for the
highest chlorinated congeners to accumulate preferentially. While a
large proportion of the PCBs is adsorbed onto particulates in
sediment, it is still bioavailable to organisms and will continue to
be accumulated in the higher trophic levels.
2.4.1. Laboratory studies
Effects of PCB mixtures on microorganisms are highly variable, with
some species being adversely affected by a concentration of
0.1 mg/litre and others being unaffected by 100 mg/litre; effects on
different species do not vary consistently with the degree of
chlorination of the mixtures.
Almost all of the studies of the effects of PCBs on aquatic
organisms have involved Aroclor mixtures. Results are extremely
variable with no consistent relationship between percentage
chlorination or environmental conditions and toxicity, even with
closely-related organisms. Over 96 h under static conditions, LC50
values ranged between 12 µg/litre and >10 mg/litre for various
aquatic invertebrate species and different Aroclor mixtures.
Flow-through conditions increased the toxicity of the PCBs.
Generally, the most toxic mixtures were Aroclors in the mid-range of
chlorination; low and high percentage chlorination mixtures were
less toxic. This was also true for sublethal effects, such as
reproduction of Daphnia. Crustaceans seem to be more susceptible
to PCBs during moult. In model populations, the community structure
of estuarine species changed on exposure to Aroclor 1254, with
amphipods, bryozoans, crabs, and molluscs decreasing in
representation and annelids, brachyopods, coelenterates, echinoderms
and nemerines being unaffected. Too few of the groups have been
included in acute tests to determine whether the result represents
variation in susceptibility to PCBs or differences in interaction
between species.
There is similar variation in the toxicity of PCB mixtures for fish
with 96-h LC50s varying between 0.008 and >100 mg/litre. Long-term
tests show that acute exposure, particularly under static
conditions, considerably underestimates the toxicity of the PCB.
Rainbow trout were particularly susceptible, with embryo-larval
stages showing a 22-day LC50 of 0.32 µg/litre for Aroclor 1254. The
no-observed-effect level (NOEL) over 22-days for rainbow trout
embryo-larval stages was 0.01 g/litre for Aroclors 1016, 1242, and
1254.
Freshwater fathead minnow showed NOELs of 5.4, 0.1, 1.8, and 1.3
µg/litre for Aroclors 1242, 1248, 1254, and 1260, respectively. The
estuarine sheephead minnow showed NOELs of 3.4 and 0.06 µg/litre for
Aroclors 1016 and 1254, respectively.
Experimental evidence has confirmed field observations demonstrating
reproductive impairment in seals fed on fish containing PCBs,
accumulated in the wild. The effect occurs late in reproduction,
preventing implantation of the embryo in the uterine wall.
In short-term tests, the toxicity of Aroclor for birds increases
with increasing percentage chlorination; 5-day dietary LC50s ranged
from 604 to >6000 mg/kg diet. The main reproductive effects in
birds are reduced hatchability of eggs and embryotoxicity. These
effects of the PCB continue after dosing has ended, as the hens
reduce their PCB load via the eggs. There is no evidence that
Aroclors cause egg-shell thinning directly; effects on the food
consumption and body weight of hens have an indirect effect on shell
thickness. Sublethal effects on behaviour and hormone secretion have
been reported.
For mink, the acute toxicity of Aroclors decreases with increasing
percentage chlorination, acute oral LD50s varying between >750 and
4000 mg/kg body weight; the ferret is less sensitive. Aroclors
reduce food consumption and, thus, the growth rate of young mink.
Reproduction in mink is reduced or eliminated by Aroclors, either
given directly, or as natural contaminants in fish. Aroclors with a
higher percentage chlorination (notably 1254) have a greater effect.
The reproductive rate returns to normal after feeding with Aroclor
is stopped.
Bats are susceptible to Aroclor released from their fat during
migration.
The majority of laboratory tests on aquatic and terrestrial
organisms have been carried out using PCB mixtures and it has not
been possible to identify the specific components of mixtures
responsible for the effects. Similarly, because tests have been
conducted under environmentally unrealistic conditions (e.g., beyond
the solubility of congeners, and, in aquatic tests, without the
presence of sediment) it is difficult to extrapolate from laboratory
tests to the field.
2.4.2 Field studies
Results suggesting that PCBs affect fish populations in the field
are inconclusive. Interpretation of field data on birds is
difficult, since residues of many different organochlorines are also
present. Most authors have shown a correlation between
embryotoxicity and total organochlorine residues. The levels of PCB
residues correlate best with the effects on embryos, but these
results cannot be regarded as proof of a field effect of the PCBs.
There is evidence (confirmed in laboratory studies) that PCBs reduce
the reproductive capacity of sea mammals. The effect is on
implantation of the embryo, but PCBs can also lead to physical
changes in the female reproductive tract.
It is not possible to extrapolate from the results of acute and
short-term laboratory tests to effects on populations in the field.
Uncertainties about which components of the PCB mixtures cause
effects, the specific congeners present in the environment, and the
bioavailability of PCB components to organisms, all combine to make
it difficult to estimate the probable environmental exposure and
effects. The effects on populatons of sea mammals can be regarded as
proved, but it is not yet known which component(s) of the PCB
mixtures are responsible.
Given the trends towards increased contamination of the marine
environment, attention should be concentrated on effects on marine
organisms. There is clear laboratory and field evidence of
reproductive effects of PCBs on populations of sea mammals in
heavily polluted areas, and PCB residues and their effects are
likely to increase in the future. It is less clear whether effects
will be seen in other organisms, such as birds that feed on marine
prey.
On the evidence of laboratory studies, population and community
effects on lower organisms, i.e., phytoplankton and zooplankton,
would be expected to occur. Both the degree and significance of such
effects are difficult to assess. From currently available
information, effects on fish populations would not be expected,
though fish will act as a route of exposure of fish-eating mammals
and birds.
Previously reported effects on terrestrial species, fish-eating
freshwater mammals and migratory bats for example, should be less
evident as the residues of PCBs are redistributed. Residues in
terrestrial biota currently show little decline overall, but
information on changes in congeners is scarce or absent. Levels of
the higher chlorinated congeners would only be expected to decrease
slowly.
2.5 Effects on experimental animals and in vitro systems
2.5.1 Single exposure
The acute toxicity of Aroclors for rats after a single oral exposure
is generally low. Young animals appear to be more sensitive (LD50,
1.3-2.5 g/kg body weight) than adults (LD50, 4-11 g/kg body
weight). The lowest LD50 reported for Aroclor 1254 in adult rats
was 1.0 g/kg body weight. No sex differences were observed.
Dermal LD50s in rabbits ranged from >1.26 to <2 g/kg body weight
for Aroclor 1260 (in corn oil) and from 0.79 to <3.17 g/kg body
weight for some other undiluted PCB mixtures. Intravenous
application demonstrated an LD50 of 0.4 g/kg body weight for
Aroclor 1254 in rats; the LD50, after intraperitoneal injection, in
the mouse varied from 0.9 to 1.2 g/kg body weight.
2.5.2 Short-term exposure
The main targets in mammals with short-term oral exposure to PCB
mixtures or congeners are the liver, the skin, the immune system,
and the reproductive system. The rhesus monkey is the most sensitive
species tested, females being more sensitive than males. Adult
female rhesus monkeys exposed to a diet containing Aroclor 1248 at a
level of 2.5 mg/kg diet, or 0.09 mg/kg body weight per day for 6
months showed an increased mortality rate, growth retardation,
alopecia, acne, swelling of the Meibomian glands, and possibly
immunosuppression. Microscopically, enlarged fatty liver with focal
necrosis, and epithelial hyperplasia and keratinization of hair
follicles were found. At higher exposure levels, microscopic changes
have also been observed in other epithelial tissues, such as
sebaceous and Meibomian glands, gastric mucosa, gall bladder, bile
duct, nail beds, and ameloblast. Serum levels of total lipid
triglycerides and cholesterol were decreased. Short-term exposure to
commercial PCB mixtures induced an increase in the contents and
concentrations of total lipids, triglycerides, cholesterol, and/or
phospholipids in the liver. Among the PCB congeners,
3,4,3',4'-tetrachlorobiphenyl, 3,4,5,3',4',5'-, and
2,4,6,2',4',6'-hexachlorobiphenyl were the most potent. Aroclor
1254, at a dose level of 0.2 mg/kg body weight per day, also showed
several other effects, such as lymphoreticular lesions, fingernail
detachment, and gingival effects, but no acne and alopecia. A
no-observed-effect level (NOEL) for the general toxicity of Aroclor
1242 of 0.04 mg/kg body weight per day was established in rhesus
monkeys. Relatively mild effects were shown in suckling rhesus
monkeys exposed to a much higher dose of Aroclor 1248 of 35 mg/kg
body weight per day. Effects in the liver have been investigated
most thoroughly in rats, and include hypertrophy, fatty
degeneration, proliferation of the endoplasmic reticulum, porphyria,
adeno-fibrosis, bile duct hyperplasia, cysts, and preneoplastic and
neoplastic changes. In studies on rats and mice, individual PCB
congeners caused effects in the liver, spleen, and thymus, the
planar congeners being most toxic. In monkeys, planar congeners, at
doses of 1-3 mg/kg diet, induced effects similar in character and
severity to those seen with Aroclor 1242 at a dose of 100 mg/kg diet
and with Aroclor 1248 at a dose of 25 mg/kg diet.
Following dermal exposure of rabbits and mice, PCB mixtures, and
some congeners, caused effects on the skin and liver similar to
those found after oral exposure. In rabbits, thymic atrophy, a
reduction in the germinal centres of the lymph nodes, and leukopenia
were also observed.
2.5.3 Reproduction, embryotoxicity, and teratogenicity
(a) Reproduction and embryotoxicity
Comprehensive reproduction and teratogenicity studies have not been
conducted. In a two-generation reproduction study on rats, a NOEL of
0.32 mg/kg body weight was established for Aroclor 1254 and a NOEL
of 7.5 mg/kg body weight for Aroclor 1260. However, the lowest
tested dose (0.06 mg/kg body weight) resulted in increased relative
liver weights in weanlings.
In rhesus monkeys exposed to Aroclor 1016, a NOEL of 0.03 mg/kg body
weight was established on the basis of reproductive parameters.
However, decreased birth weight was observed at this level and the
lowest dose tested (0.01 mg/kg body weight) resulted in skin
hyperpigmentation.
In rhesus monkeys, a NOEL of 0.09 mg/kg body weight was established
for Aroclor 1248 (containing PCDFs) 1 year after exposure ceased.
(b) Teratogenicity
Available studies on rats and monkeys did not indicate teratogenic
effects when animals were dosed orally during organogenesis. A NOEL
of 50 mg/kg body weight was demonstrated in rats for Aroclor 1254,
with regard to pup weight, and a lowest-observed-effect level of 2.5
mg/kg body weight, based on fetotoxicity (lesions in thyroid
follicular cells), could be assumed.
In teratogenicity tests of individual congeners on mice, rats, and
Rhesus monkeys, no NOEL was demonstrated. In Rhesus monkeys, a dose
of 0.07 mg/kg body weight indicated maternal toxicity
(3,4,3',4'-tetra-chlorobiphenyl).
2.5.4 Mutagenicity
PCB mixtures did not cause mutation or chromosomal damage in a
variety of test systems. Chromosome breakage was induced in human
lymphocytes in vitro by 3,4,3',4'-tetrachlorobiphenyl. High
concentrations of PCB mixtures may cause primary DNA damage, as
indicated by DNA single strand breaks in alkaline elution assays.
2.5.5 Carcinogenicity
The interpretation of the available animal data involving commercial
PCB mixtures is often complicated by lack of information concerning
the presence or contribution of chlorinated dibenzofuran impurities,
as well as variations in congener composition.
A number of long-term carcinogenicity studies have been carried out
in mice and rats. The PCB mixtures used were Kanechlors 300, 400,
and 500, Aroclors 1254 and 1260, and Clophens A30 and A60. Except
for the Clophens, which were reported to be free of polychlorinated
dibenzo-furans (PCDFs), no data were provided on the purity of the
PCB mixtures used.
A significant increase in hepatocellular adenomas and/or carcinomas
was observed in mice fed with a diet containing Kanechlor 500 and
Aroclor 1254 at a dose level of approximately 15-25 mg/kg body
weight. No neoplasms could be detected in mice treated with
Kanechlors 300 and 400.
In rats, an increase in hepatocellular adenomas and/or carcinomas
was noted in the studies on Aroclors 1254 and 1260 and Clophen A30,
with an exposure period of more than 1 year. The increase in
occurrence of tumours in animals in these studies was not considered
to be statistically significant; however, this was the case in two
other studies. An increase in the incidence of hepatocellular
(trabecular) carcinomas and adenocarcinomas was demonstrated with
Aroclor 1260 and Clophen A60, at a dose level of approximately 5
mg/kg body weight.
The liver tumours concerned were considered to be non-aggressive
(benign or weakly malignant, no metastasis) and not life-shortening.
Adenofibrosis, a pre-neoplastic lesion, and/or neoplastic nodules in
the liver were reported in some of the studies. In one test with
Aroclor 1254, dose-related increases in intestinal metaplasia and
adenocarcinomas of the glandular stomach were demonstrated in the
rat. There is a substantial body of evidence indicating that PCBs
increase the incidence of liver carcinogenesis in rodents
pre-treated with hepatocarcinogens. There is weak evidence that PCB
mixtures initiate carcinogenesis in rodents. From the genotoxicity
studies reported, it can be concluded that PCB-mixtures are
non-genotoxic. These results imply that the association of liver
tumours with administration of PCBs in rodents is attributable to
some epigenitic mechanisms involving enforcement of cell
proliferation in the liver and other manifestations of liver
toxicity, hence a threshold approach can be followed in the
evaluation of PCB toxicity. The possibility that PCBs might increase
carcinogenesis in tissues other than liver in animals pre-exposed to
various tissue-specific carcinogens needs to be addressed. The
anticarcinogenic activities of PCBs shown in some studies, where
PCBs were given to animals during, and prior to, the administration
of carcinogens, may be related to microsomal enzyme-inducing
properties of PCBs that result in an increase of detoxication.
Overall, there is reason to exercise caution in extrapolating to
humans the available animal data on the carcinogenic potential of
PCBs.
2.5.6 Special studies
The lesions induced by exposure to PCB mixtures or individual
congeners concern the liver, skin, immune system, reproductive
system, oedema, and disturbances of the gastrointestinal tract and
thyroid gland.
PCBs are able to induce various enzymes in the liver. This has been
demonstrated in rats, mice, guinea-pigs, rabbits, dogs, and monkeys
for Aroclor 1248, 1254, 1260, and Kanechlor 400 (induction of
cytochrome P450 and P448). The inducing ability increases with the
chlorine content of the molecule; it is also dependent on the
congener composition, where congeners with chlorine in the para
and meta positions induce the P450 enzyme. For AHH (aryl
hydrocarbon hydroxylase) induction, the position of the chlorine
seems to be more important than the degree of chlorination.
Congeners with both para and at least two meta positions
substituted by chlorine are the most potent inducers of AHH.
Distinct interspecies variations have been demonstrated. The lowest
NOEL of 0.025 mg/kg body weight was found for Aroclor 1260 in
Osborn-Mendel rats.
Effects have been demonstrated on the endocrine system, seen as
alterations in hormonal receptor-binding and alterations in steroid
hormone balance. Direct and indirect evidence for a weak estrogenic
activity of various Aroclors has been observed. Decreased levels of
gonadal hormones and increased relative testes weights were found in
rats exposed to 75 mg Aroclor 1242/kg diet for 36 weeks. Decreased
plasma corticosteroid levels without increased adrenal weight, were
found in female mice exposed to Aroclor 1254 (25 mg/kg diet) for 3
weeks. Increased adrenal weight was found in another strain given a
diet containing 200 mg/kg for 2 weeks.
PCB mixtures have been shown to have an immunosuppressive effect in
various animal species. Monkeys and rabbits were the most sensitive
species. The lowest NOEL in monkeys was 0.1 mg/kg body weight, and
that in rabbits, 0.18 mg/kg body weight.
Depressed motor-activity was seen in mice exposed to a single oral
dose of 500 mg Aroclor (1254/kg body weight). This was probably
related to inhibition of uptake and release of neurotransmitters.
PCB mixtures have been found to decrease the levels of vitamins A
and B1 in the blood and liver of rats. Decreased levels of vitamins
A, B1, B2, and B6 were seen in rats and mice exposed to PCB
mixtures.
2.5.7 Factors modifying toxicity; mode of action
Commercial PCBs produce a spectrum of toxic responses, partly
resembling those of polychlorinated dibenzodioxins (PCDDs) and
PCDFs. In addition, the analogous structure-activity relations of
PCB congeners, with respect to most of their toxic responses and to
their potency in inducing P448-dependent AHH (aryl hydrocarbon
hydroxylase), indicate that PCB congeners that are approximate
stereoisomers of 2,3,7,8,-TCDD are the most active. These findings
suggest a common mechanism of action based on the affinity of these
compounds for the cytosolic Ah-receptor protein. Toxic equivalence
factors relating to 2,3,7,8-TCDD have been proposed for these
coplanar PCB congeners. The nature of the likely interactions
between PCBs, PCDFs, and PCDDs has not been investigated adequately.
As PCBs can stimulate microsomal enzyme activity, they can influence
the action of other chemicals that undergo microsomal metabolism.
Other so-called non-planar PCB congeners may cause other more subtle
toxicities. In addition, PCB congeners, especially the lower
chlorinated ones, may be metabolized through arene oxide
intermediates and methylsulfonyl metabolites.
2.6 Effects in humans
The toxicological evaluation of PCBs presents many problems. PCBs
usually occur as mixtures of many congeners, and many of the data on
the toxicity of the PCBs are based on the testing of such mixtures.
Some components of the mixtures are more easily degraded in the
environment than others. Thus, exposure of the general population
may be to mixtures different from those to which the workers are
exposed.
The general population is exposed to PCBs mainly through
contaminated food (aquatic organisms, meat, and dairy products). In
most of the industrialized countries, the daily intake of PCBs is of
the order of some micrograms per person. Such exposure has not been
associated with disease. Infants are exposed to PCBs through their
mothers' milk, and their daily intake of PCBs may be some
micrograms/kg body weight.
There are great difficulties in assessing the human health effects
separately for PCBs, PCDFs, or PCDDs, since, quite frequently, PCBs
contain PCDFs, and occasionally PCDDs have been detected in the
mixtures involved in certain accidents. Commercial PCBs have been
shown to be contaminated with PCDFs and, therefore, in many cases it
is unclear whether effects are attributable to the PCBs or to the
much more toxic PCDFs. Thus, much of the data that can be retrieved
from large intoxication episodes in humans, e.g., the Yusho,
Yu-Cheng, and other intoxications, probably reflect effects of
exposure to both PCDFs and PCBs.
The signs of intoxication in the Yusho and Yu-Cheng patients were
hypersecretion of the Meibomian glands of the eyes, swelling of the
eyelids, and pigmentation of the nails and mucous membranes,
occasionally associated with fatigue, nausea, and vomiting. This was
usually followed by hyperkeratosis and darkening of the skin, with
follicular enlargement and acneform eruptions. In addition, oedema
of the arms and legs, liver enlargement and liver disorders, central
nervous system disturbances, respiratory problems, e.g.,
bronchitis-like disturbances, and changes in the immune status of
the patients, were observed. In children of the Yusho and Yu-Cheng
patients, diminished growth, dark pigmentation of the skin and
mucous membranes, gingival hyperplasia, xenophthalmic oedematous
eyes, dentition at birth, abnormal calcification of the skull,
rocker bottom heel, and a high incidence of low birth weight were
observed. Whether or not a correlation exists between the exposure
and the occurrence of malignant neoplasms in these patients cannot
be definitely concluded, because the number of deaths was too small.
However, a statistically significant increase was observed in male
patients, as regards mortality from all neoplasms, and liver and
lung cancer.
Under occupational conditions, skin rash has occurred a few hours
after acute exposure. Furthermore, itching, burning sensations,
irritation of the conjunctivae, pigmentation of fingers and nails,
and chloracne were found after exposure to high PCB concentrations.
Chloracne is one of the most prevalent findings among PCB-exposed
workers. Besides these dermal signs of intoxication, different
authors have found liver disturbances, immunosuppressive changes,
transient irritation of the mucous membranes of the respiratory
tract, and neurological and unspecific psychological or
psychosomatic effects, such as headache, dizziness, depression,
sleep and memory disturbances, nervousness, fatigue, and impotence.
The overall conclusion is that continuous occupational exposure to
high PCB and PCDF concentrations may result in effects on the skin
and liver.
Two large mortality studies have been carried out with cohorts of
workers exposed to Aroclor 1254, 1242, and 1016. Increased mortality
from cancer of the liver and gall bladder was observed in one study
and from neoplasms and cancer of the gastrointestinal tract in the
other. None of the available epidemiological studies provide
conclusive evidence of an association between PCB exposure and
increased cancer mortality owing to the small number of deaths in
the exposed populations, the lack of dose relationships, and the
problems with contaminants in the PCB mixtures.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
3.1.1 Distribution
Because of their physical and chemical properties, PCBs have become
dispersed globally throughout the environment.
PCBs are almost universally present in organisms in the environment
and are readily bioaccumulated. Biomagnification in food-chains has
also been demonstrated.
Higher chlorinated congeners accumulate preferentially.
3.1.2 Effects on experimental animals
Animal studies suggest that PCBs are immunosuppressive, as assessed
by alterations in gross measures of immune function (spleen weight,
thymus weight, or lymphocyte count). NOELs have been estimated in
monkeys at 100 µg/kg body weight for Aroclor 1248 and <100 µg/kg
body weight for Aroclor 1254. Immunosuppression appears to be a
congener-specific effect.
Reproductive toxicity is, in general, only seen at doses producing
systemic toxicity in the mother. Neonates feeding on contaminated
mother's milk (in monkeys and other animal species used as models)
appear to be particularly sensitive to PCBs and show reduced growth,
with other toxic symptoms. The NOEL for Aroclor 1016 in monkeys (on
the basis of reproductive effects) is 30 µg/kg body weight; no NOEL
could be established for reproductive effects of Aroclor 1248.
PCBs are not genotoxic and the evidence for any action as tumour
initiators is inconclusive. PCBs do act as tumour promoters. The
toxicity of PCB mixtures can be evaluated on a threshold basis.
3.1.3 Effects on humans
Exposure of the general population to PCBs is principally through
food items. Babies are exposed through the mother's milk.
Two large intoxication episodes in humans have occurred in Japan
(Yusho) and Taiwan (Yu-Cheng). The main symptoms of the Yusho and
Yu-Cheng patients have frequently been attributed mainly to the
contaminants of the PCB mixtures; specifically to PCDFs. The Task
Group concluded that the symptoms may have been caused by the
combined exposure to PCBs and PCDFs. Some of the symptoms, and
principally the chronic respiratory effects, may have been caused
specifically by the methylsulfone metabolites of certain PCB
congeners.
3.1.4 Effects on the environment
While there have been reports of effects on populations of birds,
the most important effect of PCBs on organisms in the environment is
reproductive failure in sea mammals. This has been observed
principally in semi-enclosed seas and has led to local decreases in
populations. The prediction that residues of PCBs in the environment
will gradually be redistributed towards the marine environment
indicates an increasing hazard for sea mammals in the future.
3.2 Recommendations
* International agreement on analytical procedures to improve the
comparability of results of monitoring programmes is
recommended. Development of methods for congener-specific
analysis should be continued, though the value of analysis
based on mixtures is recognized.
* In order to ensure reliability of analytical data,
inter-laboratory quality control studies are strongly
recommended. It is also recommended that an international
network of technical support and supervision should be
established to allow developing countries to participate in
monitoring.
* Long-term studies using specific congeners and studies on the
mechanism of action of constituents of PCBs mixtures, with
special regard to tumour promotion, are recommended to improve
the precision of risk assessment of PCBs.
* Epidemiological studies to improve the assessment of the risk
to neonates are required, since newborn infants appear to be
the most vulnerable sector of the general population, because
of high exposure through milk.
* Sensitive and specific biomarkers for some of the more subtle
aspects of PCB toxicity (such as reproductive, immunological,
and neural toxicity) should be developed for use in future
epidemiological studies
* PCBs should be disposed of by incineration in properly designed
and run facilities that can guarantee the constant high
temperatures (above 1000 °C), residence time, and turbulence
needed to ensure complete breakdown.
* Methods to remove PCBs already contained in landfills should be
investigated.
* Worldwide monitoring of PCBs in the environment and in wildlife
should be encouraged, to monitor the expected redistribution of
residues already present.
* Marine mammals show evidence of reproductive failure as a
result of PCB contamination. Studies on population size and
reproductive success of cetaceans should be encouraged,
together with further research to establish those congeners
responsible for the effects.
4. HEALTH HAZARDS FOR MAN, PREVENTION AND PROTECTION, EMERGENCY
ACTION
For a more detailed treatment of prevention and control of
accidental and environmental exposures to PCBs and PCTs, the reader
should refer to the WHO/EURO document listed in the Bibliography. A
detailed description of the human and environmental hazards of PCBs
is given in EHC 140 (WHO, in preparation).
4.1 Main human health hazards, prevention and protection, first aid
PCBs and PCTs are highly chlorinated organic substances. They are
very persistent and may be hazardous for human beings if incorrectly
or carelessly handled. It is, therefore, essential that the correct
precautions are observed during handling, use, and disposal.
For details see the Summary of Chemical Safety Information in
section 6.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
The acute oral and dermal toxicity is low, but under occupational
conditions skin rash may occur a few hours after acute exposure.
Furthermore, itching, burning sensations, irritation of the
conjunctivae, pigmentation of fingers and nails, and (chlor)acne
were found after exposure to high PCB concentrations for long
periods. Massive doses can cause hepatitis, facial oedema, numbness,
and weakness of the extremities. Chloracne is one of the most
prevalent findings among workers exposed to PCBs, but may be due to
the presence of PCDFs in the technical PCB mixtures. In addition to
these dermal signs of intoxication, liver disturbances,
immunosuppressive changes, transient irritation of the mucous
membranes of the respiratory tract, neurological and unspecific
effects, such as headache, dizziness, depression, sleep and memory
disturbances, nervousness, fatigue, and impotence, have been
reported.
4.1.1.2 Medical advice
Medical treatment is symptomatic and supportive.
4.1.2 Health surveillance advice
A complete medical history and physical examination of workers
regularly exposed to PCBs should be made annually. Special attention
should be paid to the skin and to liver function.
4.2 Explosion and fire hazards
Fires and explosions involving PCBs have been reported mainly from
their use in electrical equipment, such as transformers and
capacitors, but PCBs may also be involved in fires during storage
and transport. Fires may lead to the formation of highly toxic
polychlorinated dibenzofurans (PCDFs). In dielectric fluid
formulations, which also contain various tri- or
tetra-chlorobenzenes, polychlorinated dibenzo- p-dioxins (PCDDs)
may be formed upon fire or explosion.
Fires should be extinguished with alcohol-resistant foam, carbon
dioxide, or powder. With sufficient burning or external heat, PCBs
will decompose, emitting very toxic fumes. Fire-fighters should be
equipped with self-contained breathing apparatus, eye protection,
and full protective clothing.
The use of water sprays should be confined to the cooling of
unaffected stock, thus avoiding the accumulation of polluted run-off
from the site.
4.3 Storage
Products should be stored in well ventilated, locked buildings, out
of the reach of children and unauthorized personnel. Do not store
near foodstuffs or animal feed.
4.3.1 Leaking containers in store
Take precautions, and use appropriate personal protection (see
section 6). Empty any product remaining in damaged or leaking
containers into a clean empty drum, which should then be tightly
closed and suitably labelled. Sweep up spillage with sawdust, sand,
or earth (moisten for powders), and dispose of safely.
4.4 Transport
Comply with any local requirements regarding the movement of
hazardous goods. Do not transport in the same compartment as
foodstuffs. Check that the containers are sound and labels undamaged
before despatch.
4.5 Spillage and disposal
4.5.1 Spillage
Before dealing with any spillage, precautions should be taken, as
required, and appropriate personal protection should be used (see
section 6).
Prevent material from spreading or contaminating other cargo,
vegetation, or waterways, by making a barrier of the most suitable
available material, e.g., earth or sand. Absorb the spilled liquid
with sawdust, sand, or earth, sweep up and place the contaminated
material in a closeable container for later transfer to a safe place
for disposal. Care should be taken to avoid run-off into water
courses.
4.5.2 Disposal
Dielectric fluids containing PCBs in transformers and capacitors
should be recovered and sent for destruction.
Any surplus product, contaminated absorbants, and containers should
be disposed of in an appropriate way. Waste material should be
burned in a proper incinerator designed for organochlorine waste
disposal, with effluent gas-scrubbing. For PCB wastes, incineration
must be for more than 2 seconds at 1200 °C or higher. Cement kilns
may meet the required temperature/time conditions and may be
properly constructed for this purpose. If the PCB content of the
waste is less than 500 mg/kg, any proper waste incinerator can be
used as long as temperature exceeds 800 °C for 0.5 seconds.
Combustion of PCBs can produce dibenzofurans; PCB dielectric fluids
also containing tri- or tetrachlorobenzenes can also produce
dioxins. If proper incineration is not possible, bury in an approved
dump or landfill where there is no risk of contamination of surface
or ground water. Decomposition of PCBs is extremely slow.a
Comply with any local legislation regarding disposal of toxic
wastes. Puncture and crush containers, to prevent re-use.
a For a more complete treatment of the subject, refer to
WHO/Euro, 1987.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
5.1 Hazards
PCBs and PCTs are very resistant to degradation and hence very
persistent in the environment. Because they are very soluble in
lipids they bioaccumulate, especially in the fatty tissues of all
living organisms, and biomagnify in the higher trophic levels of the
food-chain.
Although their acute toxicity is relatively low, bioaccumulation and
biomagnification may lead to lethal effects, especially at the
highest trophic levels. Reduced growth and reproduction may affect
populations.
5.2 Prevention
PCBs and PCTs should be replaced by alternative products wherever
practicable.
Industrial discharges occurring during manufacture, formulation, or
technical applications should not be allowed to pollute the
environment and should be treated properly.
Any spillage or unused product should be prevented from spreading to
vegetation or waterways, and should be treated and disposed of
properly.
In all cases, immediate remedial action is essential.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, PCBs and PCTs. It should be
displayed at, or near, entrances to areas where there is
potential exposure to PCBs and PCTs, and on processing
equipment and containers. The summary should be translated
into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions in
the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the
National Poison Control Centre, and local trade names.
SUMMARY OF CHEMICAL SAFETY INFORMATION
Polychlorinated biphenyls (PCBs) Polychlorinated terphenyls (PCTs)
Molecular formula: C12H10-nCln Molecular formula: C18H14-nCln
CAS registry number: 1336-36-3 CAS registry number: 61788-33-8
RTECS registry number: TQ1350000 RTECS registry number: WZ6500000
PHYSICAL PROPERTIES AND OTHER CHARACTERISTICS
Commercial PCBs and PCTs are complex mixtures of many different congeners with various degrees of chlorination. They are
not produced to a composition specification; the criteria for use are based on physical properties. They are clear, light
yellow, or dark liquids that may turn into solid resin at low temperatures.
Their distillation range is in general above 250 °C. Their relative molecular mass and density depend on the degree of
chlorination. They are very fire-resistant, with flash-points above 170 °C. Their vapours are heavier than air, but do not
form explosive mixtures. Their electrical conductivity is very low and their resistance to thermal breakdown extremely
high. They are practically insoluble in water, easily miscible with most organic solvents, and accumulate in fatty tissues.
They have high n-octanol/water partition coefficents.
They are mainly used as dielectrics in transformers and capacitors, in heat transfer and hydraulic systems, and, to a
lesser extent, in lubricating and cutting oils, carbonless copying paper, adhesives, sealants, plastics, and as
plasticizers in paints.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: May cause irritation Avoid contact with skin; wear After contact with skin, wash immediately
and chloracne suitable protective clothing with plenty of water and soap; immediately
and gloves remove all contaminated clothing and
launder before reuse
EYES: May cause irritation Avoid contact with eyes; wear In case of contact with eyes, rinse
eye protection immediately with plenty of water and seek
medical advice
INHALATION: May cause irritation Adequate ventilation;
do not breathe vapours
INGESTION: Unlikely occupational Do not eat, drink, or smoke
hazard during work; wash hands before
eating, drinking, or smoking
Accidental or intentional If swallowed, seek medical advice
ingestion may cause poisoning immediately and show container or label
ENVIRONMENT: Bioaccumulates Strictly avoid environmental
and biomagnifies pollution
SPILLAGE STORAGEFIRE AND EXPLOSION
Take appropriate personal Products should be stored in Extinguish fires with alcohol-resistant
precautions; prevent liquid from well ventilated locked buildings; foam, CO2, or powder; with sufficient
spreading or contaminating other keep out of reach of children; burning or external heat, PCBs and
cargo, vegetation, or surface keep away from food, drink, and PCTs will decompose, emitting toxic fumes;
waters and drainage systems, animal feeding stuffs firefighters should be equipped with
with a barrier of most suitable self-contained breathing apparatus, eye
suitable material, e.g., earth or protection, and full protective clothing;
sand confine the use of water spray to cooling
of unaffected stock, thus avoiding the
Absorb spilled liquid with saw- accumulation of polluted run-off from the
dust, sand, or earth; sweep up and site
place it in a closeable container
for later safe disposal; care
should be taken to avoid run-off
into water courses
WASTE DISPOSAL NATIONAL INFORMATION
PCBs and PCTs waste material National Occupational Exposure UN No. 2315
should be burned in a proper Limit:
incinerator designed for
organochlorine waste disposal; National Poison Control Centre:
if this is not possible, bury in
an approved dump or landfill Local trade names:
where there is no risk of
contamination of surface or
ground water; comply with any
local legislation regarding
disposal of toxic wastes
7. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC. When no effective date appears in the
IRPTC legal file, the year of the reference from which the data are
taken is indicated in the table by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
7.1 Previous evaluations by international bodies
PCBs have been evaluated by IARC in 1978 and 1987 (IARC, 1978, 1987
(Supplement)). It was concluded that there was sufficient
experimental evidence to indicate a carcinogenic effect of some PCBs
in rodents, and that epidemiological data provided suggestive
evidence of a relationship between exposure to PCBs and the
development of certain cancers in man. PCBs were classified in group
2A: the agent is probably carcinogenic for humans. For practical
purposes, PCBs should be regarded as if they were carcinogenic for
human beings.
7.2 Exposure limit values
Some exposure limit values for PCBs are given in the table on pages
43-45.
CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Exposure limit values
Medium Specification Country Exposure limit description Value Effective
date
AIR Workplace Czechoslovakia Maximum permissible concentration
- time-weighted average (TWA) 0.5 mg/m3 1985
- short-term exposure limit (STEL) 1.0 mg/m3
Japan Maximum permissible concentration
- time-weighted average (TWA) 0.1 mg/m3 a 1985
Netherlands Maximum allowable concentration (MAC)
- time-weighted average (TWA) 0.5 mg/m3 -
Sweden Occupational exposure limit
- time-weighted average (TWA) 0.01 mg/m3 a 1985
- short-term exposure limit (STEL) 0.03 mg/m3
USSR Maximum permissible concentration
- time-weighted average (TWA) (vapour) 1.0 mg/m3 1977
United Kingdom Recommended limit
- time-weighted average (TWA) 0.5 mg/m3 -
- short-term exposure limit (STEL) 1.0 mg/m3
(10-min TWA)
Medium Specification Country Exposure limit description Value Effective
date
USA (ACGIH) Threshold limit value (TLV)
- time-weighted average (TWA) 0.5 mg/m3 -
- short-term exposure limit (STEL) 1.0 mg/m3
FOOD Animal Germany, Maximum residue limit (MRL) 0.008-0.6 1988
Federal (specified products) mg/kg
Republic of
Sweden Maximum residue limit (MRL) (Specified) 0.05-2 mg/kg 1983
FOOD Animal & France Maximum residue limit (MRL) (fish) 2 mg/kg -
plant
Japan Maximum residue limit (MRL) (Specified) 0.2-3 mg/kg -
Netherlands Maximum residue limit (MRL) (Specified) 0.3-1 mg/kg -
Switzerland Maximum residue limit (MRL) (Specified) 0.5-2 mg/kg -
USA Temporary residue tolerance (Specified) 0.2-3 mg/kg 1981
FEED Japan Maximum residue limit (MRL) (Specified) 0.05-3 mg/kg -
Netherlands Maximum residue limit (MRL) (Specified) 0.3 mg/kg -
USA Maximum residue limit (MRL) (Specified) 0.2-2 mg/kg -
WATER Drinking- EEC Maximum permissible concentration
(total PCBs + PCTs) 0.5 µg/litre 1982
Medium Specification Country Exposure limit description Value Effective
date
Environment Japan Environment water quality standard not detectable 1981
Surface water USSR Permissible limit 0.0 mg/litre 1978
for fishing
Effluent Japan Effluent standard 0.003 mg/litre 1981
GOODS Packaging USA Temporary residue tolerance 10 mg/kg 1983
material for
food or feed
a Skin absorption.
7.3 Specific restrictions
Several intergovernmental organizations have been active in
providing directives or recommendations for regulatory measures to
control PCBs.
On 13 February 1973, the Council of the Organisation for Economic
Cooperation and Development (OECD) adopted a Decision on the
Protection of the Environment by Control of Polychlorinated
Biphenyls (C(73)1). The Council decided that PCBs would not be used
for industrial or commercial purposes, except for five essentially
closed purposes. These are: dielectric fluids in transformers; large
power-correcting capacitors; heat-transfer fluids (but only in
installations that do not process food, feed, pharmaceuticals, or
veterinary products); hydraulic fluids (but only in mining
equipment); and in small capacitors (though Member Countries have
recommended working towards the elimination of this last use). PCBs
should only be used in the exempted applications where
non-flammability requirements outweigh the environmental protection
considerations and where sufficient controls are exercised to
minimize risk to the environment. The Council also made some
recommendations concerning the elimination of other uses of PCBs and
PCB replacements. It also provided for certain administrative and
engineering control measures for PCBs still in use, and for the
disposal of PCB wastes. The OECD Council decision provided for an
exchange of information on PCBs between Member Countries within the
framework of the OECD Environment Committee and information on PCBs
was exchanged annually between 1974 and 1980. The information
exchanged and the experience gained by Member Countries was
summarized in a report (OECD, 1982). This report indicated that,
while considerable progress had been made in reducing environmental
contamination by PCBs, some important problems remained.
An extensive synopsis of national regulatory measures in a number of
countries was prepared by the OECD in 1982. Countries have rather
complex and very different systems to control PCBs in the general
environment. Most regulations impose usage restrictions and
prescriptions for transportation and labelling, require notification
of production and/or importation, and provide rules for the disposal
of PCB-containing wastes.
On 13 February 1987, the OECD Council adopted a further
Decision-Recommendation (C(87)2(final)) on "Further measures for the
protection of the environment by control of polychlorinated
biphenyls". With this Decision-Recommendation, the OECD Member
Countries committed themselves to ban virtually all new uses for
PCBs, accelerate the phasing out of PCBs from existing uses, control
PCBs in contaminated products, articles, or equipment, and ensure
appropriate disposal methods for wastes containing PCBs.
In the countries of the European Economic Community, the use of PCBs
and PCTs is prohibited by Directive 85/467/EEC (6th Amendment (PCBs
and PCTs) Directive 76/769/EEC) but until 30 June 1986 the following
uses were excepted: (a) closed-system electrical equipment; (b)
large condensers; (c) small condensers (provided that the PCB has a
maximum chlorine content of 43% and does not contain more than 3.5%
of penta- and higher chlorinated biphenyls); (d) heat-transmitting
fluids in closed-circuit heat-transfer installations; (e) hydraulic
fluids used in underground mining equipment; (f) primary and
intermediate products for further processing into other products
which are not prohibited under the Directive. The use of equipment,
plant, and fluids referred to in points a to f above that were in
service on 30 June 1986 shall continue to be authorized until they
are disposed of or reach the end of their service life. Derogations
considered to have no deleterious effects on health or the
environment could be granted after 30 June 1986. These provisions
apply to PCBs and PCTs (except mono- and dichlorinated biphenyls)
and preparations with a PCB or PCT content higher than 0.01% by
weight.
Apart from the above restrictions in OECD and EEC countries, several
other countries have similar, more or less severe restrictions on
the use of PCBs (and PCTs). In Japan, the manufacture and import of
all PCBs is prohibited without authorization from the Government. In
the USA, the manufacture, processing, distribution in commerce and
use of PCBs is prohibited without Government authorization.
7.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies PCBs in:
Hazard Class 9: Miscellaneous dangerous substance.
Packing Group II: Substances presenting medium danger.
The European Economic Community legislation requires the labelling
of PCBs and PCTs as harmful substances using the symbol:
 The label must read:
Danger of cumulative effects. This material and its container
must be disposed of in a safe way. It should be stated on the
label whether the substance is a specific isomer or a mixture
of isomers.
7.5 Waste disposal
The following is an excerpt from the EEC Council Directive
84/631/EEC:
"EEC Member States shall: (1) prohibit the uncontrolled
discharge, dumping, and tipping of polychlorinated biphenyls
(PCBs) and polychlorinated terphenyls (PCTs) as well as
mixtures, objects, and equipment containing one or both of the
substances; (2) make compulsory the disposal of such waste; (3)
ensure that it is disposed of without endangering human health
or harming the environment; (4) promote the regeneration of PCB
and PCT; and (5) set up or designate installations which are
authorized for disposing of such waste.
"Waste containing or contaminated by polychlorinated biphenyls
is classified as 'hazardous waste'. Member States shall take
the necessary measures for the supervision and control, with a
view to human health and the environment, of the transfrontier
shipment of hazardous waste both within and if entering and/or
leaving the community. Where the holder of such waste intends
to have it shipped into, through or from one to another Member
State he shall notify the competent authorities through a
consignment note. He must provide satisfactory information in
particular on: (1) the source and composition; (2) provisions
made for routes and insurances; (3) measures to ensure safe
transport; (4) contractual agreement with the consignee of the
waste. The hazardous waste must: (a) be properly packed; (b)
have appropriate labels indicating nature, composition,
quantity and telephone numbers of persons from whom
instructions can be obtained; (c) instructions to be followed
in the event of danger or accident."
Under proposed EEC Council Directives, combustion gases in
combustion chambers must be kept at, at least 850 °C for 2 seconds,
and all plant must be fitted with auxiliary burners which come into
use automatically when combustion chamber gases fall below 850 °C.
These conditions must be met immediately by new plant and by 1994 by
existing plant (United Kingdom House of Lords paper 17, 1989, HMSO,
London).
In the USA, PCBs are classified as toxic pollutants and acute
hazardous wastes, subject to handling, transport, treatment,
storage, and disposal regulations, and permit and notification
requirements. An owner or operator of a hazardous waste incinerator
must achieve 99.9999% destruction and removal efficiency for these
substances. Effluent limitations and pre-treatment standards are set
for industries using PCBs. The interim emission standards for
incinerators are 0.25 mg/m3 (peak value) and 0.15 mg/m3 (peak
value from liquid PCB incinerators).
Under the Environmental Contaminants Act, the Canadian Ministry of
Environment published "Guidelines for the management of PCB
wastes". The Guidelines set out recommended procedures and
criteria for the safe storage, handling, and disposal of PCB wastes.
In Italy, an emission standard for PCBs and PCTs of 0.1 mg/m3 for
urban incinerators has been adopted by the Lombardy Region.
In Sweden, enterprises producing wastes that contain PCBs are
required to report the type, content, quantity, and handling of the
waste to the health authorities. Permission from the authorities is
required for the transport, handling, and export of such waste
(1975).
The Assembly of the Intergovernmental Maritime Consultative
Organization (IMCO) passed Resolution A 394(x) on 14 November 1977,
inviting governments to take steps to ensure that the operational
sea discharge of tank washings from incinerator ships containing
PCBs is prohibited, except where this is permitted under specific
regulations or technical guidelines adopted by the contracting
parties.
In 1972, the Final Act of the Intergovernmental Conference on the
Dumping of Wastes at Sea prohibited the dumping of PCBs at sea. The
Third Consultative Meeting organized by IMCO in 1978 adopted the
Amendments to the Convention Annexes which made incineration of
waste at sea subject to controls under the Convention.
The Convention on the Prevention of Marine Pollution by Dumping of
Wastes and other Matter (Oslo, 1972), concerned with the NE
Atlantic, came into force in 1974. It prohibits the dumping of
organohalogen (i.e., PCB-inclusive) sources.
At its sixth meeting (1979), the Interim Baltic Marine Environment
Protection Commission decided to draft a resolution concerning
regulation of the use of PCBs and the prevention of discharges
BIBLIOGRAPHY
BRETHERICK, L. (1986) Hazards in the chemical laboratory. 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
CEFIC. Transport emergency cards. Brussels, Conseil Européen des
Fédérations de l'Industrie Chimique.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1989) Handling chemicals
safely. 2nd ed. Amsterdam, Dutch Association of Safety Experts,
Dutch Safety Institute.
GOSSELIN, R.E., SMITH, R.P., HODGE, H.C., & BRADDOCK, J.E. (1984)
Clinical toxicology of commercial products. 5th ed. Baltimore,
Maryland, Williams and Wilkins company.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC. Data profile on individual chemical substances (unpublished
file). Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
OECD (1982) Report on the implementation by member countries of the
decision by the council on the protection of the environment by
control of polychlorinated biphenyls. Organisation of Economic
Co-operation and Development, Paris. ENV/CHEM/81.2 of November,
1981.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of
dangerous goods. 4th ed. New York, United Nations.
US NIOSH. Current intelligence bulletins. US Department of Health,
Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. 3
Vol. US National Institute for Occupational Safety and Health,
Cincinatti, Ohio, pp. 76-189.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington,
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication No.
85.114).
WHO (in preparation) EHC No. 140: Polychlorinated biphenyls and
terphenyls, 2nd ed. Geneva, World Health Organization.
WHO/EURO (1987) PCBs, PCDDs, and PCDFs: Prevention and control of
accidental and environmental exposures. Copenhagen, World Health
Organization Regional Office for Europe, pp. 227 (Environmental
Health Series No. 23).
The label must read:
Danger of cumulative effects. This material and its container
must be disposed of in a safe way. It should be stated on the
label whether the substance is a specific isomer or a mixture
of isomers.
7.5 Waste disposal
The following is an excerpt from the EEC Council Directive
84/631/EEC:
"EEC Member States shall: (1) prohibit the uncontrolled
discharge, dumping, and tipping of polychlorinated biphenyls
(PCBs) and polychlorinated terphenyls (PCTs) as well as
mixtures, objects, and equipment containing one or both of the
substances; (2) make compulsory the disposal of such waste; (3)
ensure that it is disposed of without endangering human health
or harming the environment; (4) promote the regeneration of PCB
and PCT; and (5) set up or designate installations which are
authorized for disposing of such waste.
"Waste containing or contaminated by polychlorinated biphenyls
is classified as 'hazardous waste'. Member States shall take
the necessary measures for the supervision and control, with a
view to human health and the environment, of the transfrontier
shipment of hazardous waste both within and if entering and/or
leaving the community. Where the holder of such waste intends
to have it shipped into, through or from one to another Member
State he shall notify the competent authorities through a
consignment note. He must provide satisfactory information in
particular on: (1) the source and composition; (2) provisions
made for routes and insurances; (3) measures to ensure safe
transport; (4) contractual agreement with the consignee of the
waste. The hazardous waste must: (a) be properly packed; (b)
have appropriate labels indicating nature, composition,
quantity and telephone numbers of persons from whom
instructions can be obtained; (c) instructions to be followed
in the event of danger or accident."
Under proposed EEC Council Directives, combustion gases in
combustion chambers must be kept at, at least 850 °C for 2 seconds,
and all plant must be fitted with auxiliary burners which come into
use automatically when combustion chamber gases fall below 850 °C.
These conditions must be met immediately by new plant and by 1994 by
existing plant (United Kingdom House of Lords paper 17, 1989, HMSO,
London).
In the USA, PCBs are classified as toxic pollutants and acute
hazardous wastes, subject to handling, transport, treatment,
storage, and disposal regulations, and permit and notification
requirements. An owner or operator of a hazardous waste incinerator
must achieve 99.9999% destruction and removal efficiency for these
substances. Effluent limitations and pre-treatment standards are set
for industries using PCBs. The interim emission standards for
incinerators are 0.25 mg/m3 (peak value) and 0.15 mg/m3 (peak
value from liquid PCB incinerators).
Under the Environmental Contaminants Act, the Canadian Ministry of
Environment published "Guidelines for the management of PCB
wastes". The Guidelines set out recommended procedures and
criteria for the safe storage, handling, and disposal of PCB wastes.
In Italy, an emission standard for PCBs and PCTs of 0.1 mg/m3 for
urban incinerators has been adopted by the Lombardy Region.
In Sweden, enterprises producing wastes that contain PCBs are
required to report the type, content, quantity, and handling of the
waste to the health authorities. Permission from the authorities is
required for the transport, handling, and export of such waste
(1975).
The Assembly of the Intergovernmental Maritime Consultative
Organization (IMCO) passed Resolution A 394(x) on 14 November 1977,
inviting governments to take steps to ensure that the operational
sea discharge of tank washings from incinerator ships containing
PCBs is prohibited, except where this is permitted under specific
regulations or technical guidelines adopted by the contracting
parties.
In 1972, the Final Act of the Intergovernmental Conference on the
Dumping of Wastes at Sea prohibited the dumping of PCBs at sea. The
Third Consultative Meeting organized by IMCO in 1978 adopted the
Amendments to the Convention Annexes which made incineration of
waste at sea subject to controls under the Convention.
The Convention on the Prevention of Marine Pollution by Dumping of
Wastes and other Matter (Oslo, 1972), concerned with the NE
Atlantic, came into force in 1974. It prohibits the dumping of
organohalogen (i.e., PCB-inclusive) sources.
At its sixth meeting (1979), the Interim Baltic Marine Environment
Protection Commission decided to draft a resolution concerning
regulation of the use of PCBs and the prevention of discharges
BIBLIOGRAPHY
BRETHERICK, L. (1986) Hazards in the chemical laboratory. 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
CEFIC. Transport emergency cards. Brussels, Conseil Européen des
Fédérations de l'Industrie Chimique.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1989) Handling chemicals
safely. 2nd ed. Amsterdam, Dutch Association of Safety Experts,
Dutch Safety Institute.
GOSSELIN, R.E., SMITH, R.P., HODGE, H.C., & BRADDOCK, J.E. (1984)
Clinical toxicology of commercial products. 5th ed. Baltimore,
Maryland, Williams and Wilkins company.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC. Data profile on individual chemical substances (unpublished
file). Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
OECD (1982) Report on the implementation by member countries of the
decision by the council on the protection of the environment by
control of polychlorinated biphenyls. Organisation of Economic
Co-operation and Development, Paris. ENV/CHEM/81.2 of November,
1981.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of
dangerous goods. 4th ed. New York, United Nations.
US NIOSH. Current intelligence bulletins. US Department of Health,
Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. 3
Vol. US National Institute for Occupational Safety and Health,
Cincinatti, Ohio, pp. 76-189.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington,
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication No.
85.114).
WHO (in preparation) EHC No. 140: Polychlorinated biphenyls and
terphenyls, 2nd ed. Geneva, World Health Organization.
WHO/EURO (1987) PCBs, PCDDs, and PCDFs: Prevention and control of
accidental and environmental exposures. Copenhagen, World Health
Organization Regional Office for Europe, pp. 227 (Environmental
Health Series No. 23).
 The chemical formula can be presented as C12H10-nCln, where n is
the number of chlorine atoms in the molecule.
Chemical composition
The PCBs are chlorinated hydrocarbons that are manufactured
commercially by the progressive chlorination of biphenyl in the
presence of a suitable catalyst (e.g., iron chloride). Depending on
the reaction conditions, the degree of chlorination can vary between
21 and 68% (w/w). The yield is always a mixture of different
compounds and congeners. Thus, a total of 209 different chemical
components may exist, but only about 130 of these are likely to
occur in commercial products or mixtures of these compounds.
Individual PCBs have been synthesized for use as reference samples
in the identification of gas-liquid chromatographic peaks, for
toxicological investigations, and in order to study their metabolic
fate in living organisms.
Purity and impurities
Commercial PCBs are sold on the basis of their physical properties,
not their chemical composition. Different batches may vary somewhat
in their composition. The impurities known to be present in
commercial PCBs include chlorinated naphthalenes and small
quantities of the highly toxic polychlorinated dibenzofurans
(PCDFs). There are no authenticated reports of the presence of
polychlorinated dibenzo- p-dioxins (PCDDs) in commercial PCBs.
Common name: Polychlorinated biphenyls - PCBs.
CAS registry number:
The chemical formula can be presented as C12H10-nCln, where n is
the number of chlorine atoms in the molecule.
Chemical composition
The PCBs are chlorinated hydrocarbons that are manufactured
commercially by the progressive chlorination of biphenyl in the
presence of a suitable catalyst (e.g., iron chloride). Depending on
the reaction conditions, the degree of chlorination can vary between
21 and 68% (w/w). The yield is always a mixture of different
compounds and congeners. Thus, a total of 209 different chemical
components may exist, but only about 130 of these are likely to
occur in commercial products or mixtures of these compounds.
Individual PCBs have been synthesized for use as reference samples
in the identification of gas-liquid chromatographic peaks, for
toxicological investigations, and in order to study their metabolic
fate in living organisms.
Purity and impurities
Commercial PCBs are sold on the basis of their physical properties,
not their chemical composition. Different batches may vary somewhat
in their composition. The impurities known to be present in
commercial PCBs include chlorinated naphthalenes and small
quantities of the highly toxic polychlorinated dibenzofurans
(PCDFs). There are no authenticated reports of the presence of
polychlorinated dibenzo- p-dioxins (PCDDs) in commercial PCBs.
Common name: Polychlorinated biphenyls - PCBs.
CAS registry number:  The chemical formula can be written as C18H14-nCln, in which n
is the number of chlorine atoms, which can range from 1-14.
Chemical composition
The theoretically possible number of different PCTs is several
orders of magnitude greater than the number of PCBs, but in
practice, as with PCBs, PCTs are sold on the basis of their physical
properties, which depend on the degree of chlorination, and not
their chemical composition.
Common name: Polychlorinated terphenyls - PCTs.
CAS registry number:
The chemical formula can be written as C18H14-nCln, in which n
is the number of chlorine atoms, which can range from 1-14.
Chemical composition
The theoretically possible number of different PCTs is several
orders of magnitude greater than the number of PCBs, but in
practice, as with PCBs, PCTs are sold on the basis of their physical
properties, which depend on the degree of chlorination, and not
their chemical composition.
Common name: Polychlorinated terphenyls - PCTs.
CAS registry number:  The label must read:
Danger of cumulative effects. This material and its container
must be disposed of in a safe way. It should be stated on the
label whether the substance is a specific isomer or a mixture
of isomers.
7.5 Waste disposal
The following is an excerpt from the EEC Council Directive
84/631/EEC:
"EEC Member States shall: (1) prohibit the uncontrolled
discharge, dumping, and tipping of polychlorinated biphenyls
(PCBs) and polychlorinated terphenyls (PCTs) as well as
mixtures, objects, and equipment containing one or both of the
substances; (2) make compulsory the disposal of such waste; (3)
ensure that it is disposed of without endangering human health
or harming the environment; (4) promote the regeneration of PCB
and PCT; and (5) set up or designate installations which are
authorized for disposing of such waste.
"Waste containing or contaminated by polychlorinated biphenyls
is classified as 'hazardous waste'. Member States shall take
the necessary measures for the supervision and control, with a
view to human health and the environment, of the transfrontier
shipment of hazardous waste both within and if entering and/or
leaving the community. Where the holder of such waste intends
to have it shipped into, through or from one to another Member
State he shall notify the competent authorities through a
consignment note. He must provide satisfactory information in
particular on: (1) the source and composition; (2) provisions
made for routes and insurances; (3) measures to ensure safe
transport; (4) contractual agreement with the consignee of the
waste. The hazardous waste must: (a) be properly packed; (b)
have appropriate labels indicating nature, composition,
quantity and telephone numbers of persons from whom
instructions can be obtained; (c) instructions to be followed
in the event of danger or accident."
Under proposed EEC Council Directives, combustion gases in
combustion chambers must be kept at, at least 850 °C for 2 seconds,
and all plant must be fitted with auxiliary burners which come into
use automatically when combustion chamber gases fall below 850 °C.
These conditions must be met immediately by new plant and by 1994 by
existing plant (United Kingdom House of Lords paper 17, 1989, HMSO,
London).
In the USA, PCBs are classified as toxic pollutants and acute
hazardous wastes, subject to handling, transport, treatment,
storage, and disposal regulations, and permit and notification
requirements. An owner or operator of a hazardous waste incinerator
must achieve 99.9999% destruction and removal efficiency for these
substances. Effluent limitations and pre-treatment standards are set
for industries using PCBs. The interim emission standards for
incinerators are 0.25 mg/m3 (peak value) and 0.15 mg/m3 (peak
value from liquid PCB incinerators).
Under the Environmental Contaminants Act, the Canadian Ministry of
Environment published "Guidelines for the management of PCB
wastes". The Guidelines set out recommended procedures and
criteria for the safe storage, handling, and disposal of PCB wastes.
In Italy, an emission standard for PCBs and PCTs of 0.1 mg/m3 for
urban incinerators has been adopted by the Lombardy Region.
In Sweden, enterprises producing wastes that contain PCBs are
required to report the type, content, quantity, and handling of the
waste to the health authorities. Permission from the authorities is
required for the transport, handling, and export of such waste
(1975).
The Assembly of the Intergovernmental Maritime Consultative
Organization (IMCO) passed Resolution A 394(x) on 14 November 1977,
inviting governments to take steps to ensure that the operational
sea discharge of tank washings from incinerator ships containing
PCBs is prohibited, except where this is permitted under specific
regulations or technical guidelines adopted by the contracting
parties.
In 1972, the Final Act of the Intergovernmental Conference on the
Dumping of Wastes at Sea prohibited the dumping of PCBs at sea. The
Third Consultative Meeting organized by IMCO in 1978 adopted the
Amendments to the Convention Annexes which made incineration of
waste at sea subject to controls under the Convention.
The Convention on the Prevention of Marine Pollution by Dumping of
Wastes and other Matter (Oslo, 1972), concerned with the NE
Atlantic, came into force in 1974. It prohibits the dumping of
organohalogen (i.e., PCB-inclusive) sources.
At its sixth meeting (1979), the Interim Baltic Marine Environment
Protection Commission decided to draft a resolution concerning
regulation of the use of PCBs and the prevention of discharges
BIBLIOGRAPHY
BRETHERICK, L. (1986) Hazards in the chemical laboratory. 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
CEFIC. Transport emergency cards. Brussels, Conseil Européen des
Fédérations de l'Industrie Chimique.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1989) Handling chemicals
safely. 2nd ed. Amsterdam, Dutch Association of Safety Experts,
Dutch Safety Institute.
GOSSELIN, R.E., SMITH, R.P., HODGE, H.C., & BRADDOCK, J.E. (1984)
Clinical toxicology of commercial products. 5th ed. Baltimore,
Maryland, Williams and Wilkins company.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC. Data profile on individual chemical substances (unpublished
file). Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
OECD (1982) Report on the implementation by member countries of the
decision by the council on the protection of the environment by
control of polychlorinated biphenyls. Organisation of Economic
Co-operation and Development, Paris. ENV/CHEM/81.2 of November,
1981.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of
dangerous goods. 4th ed. New York, United Nations.
US NIOSH. Current intelligence bulletins. US Department of Health,
Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. 3
Vol. US National Institute for Occupational Safety and Health,
Cincinatti, Ohio, pp. 76-189.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington,
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication No.
85.114).
WHO (in preparation) EHC No. 140: Polychlorinated biphenyls and
terphenyls, 2nd ed. Geneva, World Health Organization.
WHO/EURO (1987) PCBs, PCDDs, and PCDFs: Prevention and control of
accidental and environmental exposures. Copenhagen, World Health
Organization Regional Office for Europe, pp. 227 (Environmental
Health Series No. 23).
The label must read:
Danger of cumulative effects. This material and its container
must be disposed of in a safe way. It should be stated on the
label whether the substance is a specific isomer or a mixture
of isomers.
7.5 Waste disposal
The following is an excerpt from the EEC Council Directive
84/631/EEC:
"EEC Member States shall: (1) prohibit the uncontrolled
discharge, dumping, and tipping of polychlorinated biphenyls
(PCBs) and polychlorinated terphenyls (PCTs) as well as
mixtures, objects, and equipment containing one or both of the
substances; (2) make compulsory the disposal of such waste; (3)
ensure that it is disposed of without endangering human health
or harming the environment; (4) promote the regeneration of PCB
and PCT; and (5) set up or designate installations which are
authorized for disposing of such waste.
"Waste containing or contaminated by polychlorinated biphenyls
is classified as 'hazardous waste'. Member States shall take
the necessary measures for the supervision and control, with a
view to human health and the environment, of the transfrontier
shipment of hazardous waste both within and if entering and/or
leaving the community. Where the holder of such waste intends
to have it shipped into, through or from one to another Member
State he shall notify the competent authorities through a
consignment note. He must provide satisfactory information in
particular on: (1) the source and composition; (2) provisions
made for routes and insurances; (3) measures to ensure safe
transport; (4) contractual agreement with the consignee of the
waste. The hazardous waste must: (a) be properly packed; (b)
have appropriate labels indicating nature, composition,
quantity and telephone numbers of persons from whom
instructions can be obtained; (c) instructions to be followed
in the event of danger or accident."
Under proposed EEC Council Directives, combustion gases in
combustion chambers must be kept at, at least 850 °C for 2 seconds,
and all plant must be fitted with auxiliary burners which come into
use automatically when combustion chamber gases fall below 850 °C.
These conditions must be met immediately by new plant and by 1994 by
existing plant (United Kingdom House of Lords paper 17, 1989, HMSO,
London).
In the USA, PCBs are classified as toxic pollutants and acute
hazardous wastes, subject to handling, transport, treatment,
storage, and disposal regulations, and permit and notification
requirements. An owner or operator of a hazardous waste incinerator
must achieve 99.9999% destruction and removal efficiency for these
substances. Effluent limitations and pre-treatment standards are set
for industries using PCBs. The interim emission standards for
incinerators are 0.25 mg/m3 (peak value) and 0.15 mg/m3 (peak
value from liquid PCB incinerators).
Under the Environmental Contaminants Act, the Canadian Ministry of
Environment published "Guidelines for the management of PCB
wastes". The Guidelines set out recommended procedures and
criteria for the safe storage, handling, and disposal of PCB wastes.
In Italy, an emission standard for PCBs and PCTs of 0.1 mg/m3 for
urban incinerators has been adopted by the Lombardy Region.
In Sweden, enterprises producing wastes that contain PCBs are
required to report the type, content, quantity, and handling of the
waste to the health authorities. Permission from the authorities is
required for the transport, handling, and export of such waste
(1975).
The Assembly of the Intergovernmental Maritime Consultative
Organization (IMCO) passed Resolution A 394(x) on 14 November 1977,
inviting governments to take steps to ensure that the operational
sea discharge of tank washings from incinerator ships containing
PCBs is prohibited, except where this is permitted under specific
regulations or technical guidelines adopted by the contracting
parties.
In 1972, the Final Act of the Intergovernmental Conference on the
Dumping of Wastes at Sea prohibited the dumping of PCBs at sea. The
Third Consultative Meeting organized by IMCO in 1978 adopted the
Amendments to the Convention Annexes which made incineration of
waste at sea subject to controls under the Convention.
The Convention on the Prevention of Marine Pollution by Dumping of
Wastes and other Matter (Oslo, 1972), concerned with the NE
Atlantic, came into force in 1974. It prohibits the dumping of
organohalogen (i.e., PCB-inclusive) sources.
At its sixth meeting (1979), the Interim Baltic Marine Environment
Protection Commission decided to draft a resolution concerning
regulation of the use of PCBs and the prevention of discharges
BIBLIOGRAPHY
BRETHERICK, L. (1986) Hazards in the chemical laboratory. 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
CEFIC. Transport emergency cards. Brussels, Conseil Européen des
Fédérations de l'Industrie Chimique.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1989) Handling chemicals
safely. 2nd ed. Amsterdam, Dutch Association of Safety Experts,
Dutch Safety Institute.
GOSSELIN, R.E., SMITH, R.P., HODGE, H.C., & BRADDOCK, J.E. (1984)
Clinical toxicology of commercial products. 5th ed. Baltimore,
Maryland, Williams and Wilkins company.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC. Data profile on individual chemical substances (unpublished
file). Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
OECD (1982) Report on the implementation by member countries of the
decision by the council on the protection of the environment by
control of polychlorinated biphenyls. Organisation of Economic
Co-operation and Development, Paris. ENV/CHEM/81.2 of November,
1981.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of
dangerous goods. 4th ed. New York, United Nations.
US NIOSH. Current intelligence bulletins. US Department of Health,
Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. 3
Vol. US National Institute for Occupational Safety and Health,
Cincinatti, Ohio, pp. 76-189.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington,
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication No.
85.114).
WHO (in preparation) EHC No. 140: Polychlorinated biphenyls and
terphenyls, 2nd ed. Geneva, World Health Organization.
WHO/EURO (1987) PCBs, PCDDs, and PCDFs: Prevention and control of
accidental and environmental exposures. Copenhagen, World Health
Organization Regional Office for Europe, pp. 227 (Environmental
Health Series No. 23).
