
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 96
d-PHENOTHRIN
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
Published under the joint sponsorship of
the United Nations Environment Programme,
the International Labour Organisation,
and the World Health Organization
World Health Orgnization
Geneva, 1990
The International Programme on Chemical Safety (IPCS) is a
joint venture of the United Nations Environment Programme, the
International Labour Organisation, and the World Health
Organization. The main objective of the IPCS is to carry out and
disseminate evaluations of the effects of chemicals on human health
and the quality of the environment. Supporting activities include
the development of epidemiological, experimental laboratory, and
risk-assessment methods that could produce internationally
comparable results, and the development of manpower in the field of
toxicology. Other activities carried out by the IPCS include the
development of know-how for coping with chemical accidents,
coordination of laboratory testing and epidemiological studies, and
promotion of research on the mechanisms of the biological action of
chemicals.
WHO Library Cataloguing in Publication Data
d-Phenothrin.
(Environmental health criteria ; 96)
1.Pyrethrins I.Series
ISBN 92 4 154296 9 (NLM Classification: WA 240)
ISSN 0250-863X
The World Health Organization welcomes requests for permission
to reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1990
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material
in this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR d-PHENOTHRIN
INTRODUCTION
1. SUMMARY, EVALUATION, CONCLUSIONS, AND RECOMMENDATIONS
1.1. Summary and evaluation
1.1.1. Identity, physical and chemical properties,
analytical methods
1.1.2. Production and use
1.1.3. Human exposure
1.1.4. Environmental fate
1.1.5. Kinetics and metabolism
1.1.6. Effects on organisms in the environment
1.1.7. Effects on experimental animals and in vitro test
systems
1.1.8. Effects on human beings
1.2. Conclusions
1.2.1. General population
1.2.2. Occupational exposure
1.2.3. Environment
1.3. Recommendations
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1. Identity
2.2. Physical and chemical properties
2.3. Analytical methods
3. SOURCES AND LEVELS OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Industrial production
3.2. Use patterns
3.3. Residues in food
3.4. Residues in the environment
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
4.1. Transport and distribution between media
4.2. Photodegradation
4.3. Degradation in plants and soils
4.4. Degradation on stored foods
5. KINETICS AND METABOLISM
5.1. Metabolism in mammals
5.2. Enzymatic systems for biotransformation
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1. Aquatic organisms
6.2. Terrestrial organisms
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1. Single and short-term exposures
7.2. Long-term exposures and carcinogenicity
7.2.1. Rat
7.2.2. Mouse
7.2.3. Dog
7.3. Mutagenicity
7.4. Reproduction, embryotoxicity, and teratogenicity
7.4.1. Embryotoxicity and teratogenicity
7.4.2. Reproduction studies
7.5. Neurotoxicity
7.6. Miscellaneous effects
7.7. Mechanism of toxicity - mode of action
8. EFFECTS ON HUMANS
8.1. Clinical studies
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
APPENDIX I
FRENCH TRANSLATION OF SUMMARY, EVALUATION, CONCLUSIONS, AND
RECOMMENDATIONS
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR D-PHENOTHRIN
Members
Dr V. Benes, Toxicology and Reference Laboratory, Institute of
Hygiene and Epidemiology, Prague, Czechoslovakia
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood
Experimental Station, Huntingdon, United Kingdom
Dr Y. Hayashi, Division of Pathology, National Institute of
Hygienic Sciences, Tokyo, Japan
Dr S. Johnson, Hazard Evaluation Division, Office of Pesticide
Programme, US Environmental Protection Agency, Washington DC,
USA (Chairman)
Dr S.K. Kashyap, National Institute of Occupational Health
(I.C.M.R.) Ahmedabad, India (Vice Chairman)
Dr Yu. I. Kundiev, Research Institute of Labour, Hygiene, and
Occupational Diseases, Kiev, USSR
Dr J.P. Leahey, ICI Agrochemicals, Jealotts Hill Research Station,
Bracknell, United Kingdom (Rapporteur)
Dr J. Miyamoto, Takarazuka Research Centre, Sumitomo Chemical
Company, Takarazuka, Hyogo, Japan
Dr Y. Takenaka, Division of Information on Chemical Safety, Tokyo,
Japan
Representatives of other Organizations
Dr M. Ikeda, International Commission on Occupational Health,
Department of Environmental Health, Tohoku University, School
of Medicine, Sendai, Japan
Dr H. Naito, World Federation of Poison Control Centres and
Clinical Toxicology, Institute of Clinical Medicine, University
of Tsukuba, Tsukuba-Shi, Ibaraki, Japan
Dr N. Punja, Groupement International des Associations Nationales
de Fabricants de Produits Agrochimiques (GIFAP), ICI Plant
Protection Division, Fenhurst, Haslemere, United Kingdom
Observers
Dr M. Matsuo, Sumitomo Chemical Company, Biochemistry & Toxicology
Laboratory, Osaka, Japan
Dr Y. Okuno, Sumitomo Chemical Company, Biochemistry & Toxicology
Laboratory, Osaka, Japan
Secretariat
Dr K.W. Jager, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland (Secretary)
Dr R. Plestina, Division of Vector Control, Delivery and Management
of Vector Control, World Health Organization, Geneva,
Switzerland
Dr J. Sekizawa, Section of Information and Investigation, Division
of Information on Chemical Safety, National Institute of
Hygienic Sciences, Tokyo, Japan (Rapporteur)
NOTE TO READERS OF THE CRITERIA DOCUMENTS
Every effort has been made to present information in the
criteria documents as accurately as possible without unduly
delaying their publication. In the interest of all users of the
environmental health criteria documents, readers are kindly
requested to communicate any errors that may have occurred to the
Manager of the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland, in order that they may be
included in corrigenda, which will appear in subsequent volumes.
* * *
A detailed data profile and a legal file can be obtained from
the International Register of Potentially Toxic Chemicals, Palais
des Nations, 1211 Geneva 10, Switzerland (Telephone No. 7988400-
7985850).
* * *
The proprietary information contained in this document cannot
replace documentation for registration purposes, because the latter
has to be closely linked to the source, the manufacturing route and
the purity/impurities of the substance to be registered. The data
should be used in accordance with para. 82-84 and recommendations
para. 90 of the Second FAO Government Consultation (1982).
ENVIRONMENTAL HEALTH CRITERIA FOR D-PHENOTHRIN
A WHO Task Group on Environmental Health Criteria for
Fenvalerate, Permethrin, and d-Phenothrin met in Tokyo from 4 to 8
July 1988. This meeting was convened with the financial assistance
of the Ministry of Health and Welfare, Tokyo, Japan, and was hosted
by the National Institute of Hygienic Sciences (NIHS) in Tokyo.
Dr T. Furukawa and Dr K. Shirota opened the meeting on behalf
of the Ministry of Health and Welfare, and Dr A. Tanimura,
Director-General of NIHS welcomed the participants to the
institute. Dr M. Mercier, Manager of the IPCS, welcomed the
participants on behalf of the three IPCS cooperating organizations
(UNEP/ILO/WHO). The group reviewed and revised the draft monograph
and made an evaluation of the risks for human health and the
environment from exposure to d-phenothrin.
The first draft of this document was prepared by Dr J. MIYAMOTO
and Dr MATSUO of Sumitomo Chemical Company, with the assistance of
the staff of the National Institute of Hygienic Sciences, Tokyo,
Japan. Dr I. Yamamoto of the Tokyo University of Agriculture and Dr
M. Eto of Kyushu University, Japan, assisted with the finalization
of this draft. The second draft was prepared by Dr J. SEKIZAWA,
NIHS, Tokyo, incorporating comments received following circulation
of the first draft to the IPCS contact points for Environmental
Health Criteria documents. Dr K.W. Jager and Dr P.G. Jenkins, both
members of the IPCS Central Unit, were responsible for the
technical development and editing, respectively, of this monograph.
The assistance of the Sumitomo Chemical Company, Japan, in
making available to the IPCS and the Task Group their toxicological
proprietary information on d-phenothrin is gratefully acknowledged.
This allowed the Task Group to make its evaluation on this basis of
more complete data.
INTRODUCTION
SYNTHETIC PYRETHROIDS - A PROFILE
1. During investigations to modify the chemical structures of
natural pyrethrins, a certain number of synthetic pyrethroids
were produced with improved physical and chemical properties
and greater biological activity. Several of the earlier
synthetic pyrethroids were successfully commercialized, mainly
for the control of household insects. Other more recent
pyrethroids have been introduced as agricultural insecticides
because of their excellent activity against a wide range of
insect pests and their non-persistence in the environment.
2. The pyrethroids constitute another group of insecticides in
addition to organochlorine, organophosphorus, carbamate, and
other compounds. Pyrethroids commercially available to date
include allethrin, resmethrin, d-phenothrin, and tetramethrin
(for insects of public health importance), and cypermethrin,
deltamethrin, fenvalerate, and permethrin (mainly for
agricultural insects). Other pyrethroids are also available
including furamethrin, kadethrin, and tellallethrin (usually
for household insects), fenpropathrin, tralomethrin,
cyhalothrin, lambda-cyhalothrin, tefluthrin, cufluthrin,
flucythrinate, fluvalinate, and biphenate (for agricultural
insects).
3. Toxicological evaluations of several synthetic pyrethroids have
been performed by the FAO/WHO Joint Meeting on Pesticide
Residues (JMPR). The acceptable daily intake (ADI) has been
estimated by the JMPR for cypermethrin, deltamethrin,
fenvalerate, permethrin, d-phenothrin, cyfluthrin, cyhalothrin,
and flucythrinate.
4. Chemically, synthetic pyrethroids are esters of specific acids
(e.g., chrysanthemic acid, halo-substituted chrysanthemic acid,
2-(4-chlorophenyl)-3-methylbutyric acid) and alcohols (e.g.,
allethrolone, 3-phenoxybenzyl alcohol). For certain
pyrethroids, asymmetric centre(s) exist in the acid and/or
alcohol moiety, and the commercial products sometimes consist
of a mixture of both optical (1R/1S or d/1) and geometric
(cis/trans) isomers. However, most of the insecticidal
activity of such products may reside in only one or two
isomers. Some of the products (e.g., d-phenothrin,
deltamethrin) consist only of such active isomer(s).
5. Synthetic pyrethroids are neuropoisons acting on the axons in
the peripheral and central nervous systems by interacting with
sodium channels in mammals and/or insects. A single dose
produces toxic signs in mammals, such as tremors,
hyperexcitability, salivation, choreoathetosis, and paralysis.
The signs disappear fairly rapidly, and the animals recover,
generally within a week. At near-lethal dose levels, synthetic
pyrethroids cause transient changes in the nervous system, such
as axonal swelling and/or breaks and myelin degeneration in
sciatic nerves. They are not considered to cause delayed
neurotoxicity of the kind induced by some organophosphorus
compounds. The mechanism of toxicity of synthetic pyrethroids
and their classification into two types are discussed in the
Appendix.
6. Some pyrethroids (e.g., deltamethrin, fenvalerate,
flucythrinate, and cypermethrin) may cause a transient itching
and/or burning sensation in exposed human skin.
7. Synthetic pyrethroids are generally metabolized in mammals
through ester hydrolysis, oxidation, and conjugation, and there
is no tendency to accumulate in tissues. In the environment,
synthetic pyrethroids are fairly rapidly degraded in soil and
in plants. Ester hydrolysis and oxidation at various sites on
the molecule are the major degradation processes. The
pyrethroids are strongly adsorbed on soil and sediments, and
hardly eluted with water. There is little tendency for
bioaccumulation in organisms.
8. Because of low application rates and rapid degradation in the
environment, residues in food are generally low.
9. Synthetic pyrethroids have been shown to be toxic for fish,
aquatic arthropods, and honey-bees in laboratory tests. But,
in practical usage, no serious adverse effects have been
noticed because of the low rates of application and lack of
persistence in the environment. The toxicity of synthetic
pyrethroids in birds and domestic animals is low.
10. In addition to the evaluation documents of FAO/WHO, there are
several good reviews and books on the chemistry, metabolism,
mammalian toxicity, environmental effects, etc. of synthetic
pyrethroids, including those by Elliott (1977), Miyamoto
(1981), Miyamoto & Kearney (1983), and Leahey (1985).
1. SUMMARY, EVALUATION, CONCLUSIONS, AND RECOMMENDATIONS
1.1. Summary and Evaluation
1.1.1. Identity, physical and chemical properties, analytical methods
Racemic phenothrin was first synthesized in 1969. Chemically,
it is an ester of chrysanthemic acid (2,2-dimethyl-3-(2,2-
dimethylvinyl)-cyclopropanecarboxylic acid) and 3-phenoxybenzyl
alcohol (PBalc). It is a mixture of four stereoisomers, i.e., the
[1R,trans], [1R,cis], [1S,trans], and [1S,cis] isomers.
d-Phenothrin is the 1:4 mixture of the [1R,cis] and [1R,trans]
isomers and is nowadays the only technical product commercially
available. The [1R,trans] isomer is the most insecticidally active
isomer, followed by the [1R,cis] isomer.
Technical grade d-phenothrin is a pale yellow to yellow-brown
liquid and is 92.5-94.5% pure. The specific gravity is 1.058-1.061
at 25°C, and the vapour pressure is 0.16 mPa at 20°C. It is
sparingly soluble in water (2 mg/litre at 25°C) but is soluble in
organic solvents such as acetone, xylene, and hexane. It is
fairly stable in air but is unstable to light, although it is not
photodegraded as rapidly as natural pyrethrins. It is unstable in
alkaline media.
Residue analysis can be carried out by determination using
high-performance liquid chromatography with UV detector, the
minimum detectable concentration being 0.05 mg/kg. A gas
chromatograph equipped with flame ionization detector is used for
the analysis of the technical product.
1.1.2. Production and use
d-Phenothrin has been in use since 1977. It is estimated that
70-80 tonnes of d-phenothrin are used annually worldwide, mainly to
control noxious insects in the household and insects of public
health concern and to protect stored grain. It is used either
alone or in combination with other insecticides and/or synergists,
and it is formulated in aerosols, oil, dust formulations, and
emulsifiable concentrate. d-Phenothrin is also used to control
human lice, in which case it is formulated as a powder, shampoo, or
lotion.
1.1.3. Human exposure
Conventional household aerosol spraying is not expected to lead
to aerial levels of d-phenothrin greater than 0.5 mg/m3. Residues
of up to 4 mg/kg might be present in stored wheat, but this
decreases, after milling, to 0.8 mg/kg in flour and to 0.6 mg/kg
after baking.
To control lice, d-phenothrin is applied to human hair, e.g.,
three doses of 32 mg at 3-day intervals. No data are available on
occupational exposure to d-phenothrin.
The exposure of the general population is expected to be very
low, but precise data are lacking.
1.1.4. Environmental fate
Phenothrin degrades readily, with a half-life of less than 1
day, on plants and other surfaces. There is little translocation
of d-phenothrin or its degradation products to the untreated parts
of the plants. Limited uptake of radiolabelled products into bean
plants took place from soils treated with 14C-phenothrin. When
soils were treated with [1R, trans ]- or [1R, cis ]-phenothrin
(1 mg/kg), both isomers decomposed rapidly with initial half-lives
of 1-2 days, but under flooded conditions the degradation was much
slower, with initial half-lives of 2-4 weeks (trans isomer) and 1-2
months (cis isomer). Very little movement (approximately 2%) of
either trans- or cis- phenothrin was observed through soil
columns when leaching was started immediately or 14 days after
treatment with the insecticide.
In general, the degradative processes that occur in the
environment lead to less toxic products.
1.1.5. Kinetics and metabolism
After rats were given single or repeated oral exposure or
dermal treatment with radiolabelled phenothrin, the radiolabel was
rapidly and almost completely excreted in urine and faeces within
3-7 days. The major metabolic pathways of both trans- and cis-
phenothrin in rats were ester cleavage and oxidation at the 4'-
position of the alcohol moiety or the isobutenyl group of the acid
moiety. Ester-cleaved metabolites (excreted mainly in the urine)
were the principal products of the trans isomer, whereas ester-form
metabolites (excreted mainly in the faeces) were mostly formed from
the cis isomer.
1.1.6. Effects on organisms in the environment
Phenothrin has been tested on few groups of non-target
organisms and on only a few species within each group. The 96-h
LC50 for racemic phenothrin and (1R) stereoisomers in fish ranged
from 17 to 200 µg/litre. A single study on aquatic invertebrates
demonstrated 3-h LC50 values for Daphnia pulex of 25-50 mg/litre
for all isomers and for racemic phenothrin.
A single field study applying phenothrin to ponds showed no
effect on aquatic arthropods.
Toxicity to birds is low with an acute oral LD50 for bobwhite
quail of >2500 mg/kg body weight and a dietary LC50 for mallard
duck and bobwhite quail of >5000 mg/kg diet.
Since phenothrin breaks down rapidly in sunlight and is used
principally on stored grain, environmental exposure is expected to
be very low. Therefore, effects on the environment are extremely
unlikely.
1.1.7. Effects on experimental animals and in vitro test systems
The acute toxicity of d-phenothrin is extremely low, the LD50
being >5000 mg/kg body weight in the rat and mouse (via the oral,
subcutaneous, dermal, and intraperitoneal routes) and the
inhalation LC50 >3760 mg/m3 in the rat. d-Phenothrin causes a
poisoning syndrome of hyperexcitability, prostration, tremor,
ataxia, and paralysis. From these symptoms and the results of
electrophysiological studies of cockroach cercal sensory nerves, it
is classified as a Type I pyrethroid.
When rats were exposed to d-phenothrin by inhalation at
concentrations of up to 210 mg/m3 for 4 h per day for 4 weeks or
orally for 5 consecutive days at a dose level of 5000 mg/kg body
weight, no adverse toxicological effects were observed.
Several feeding studies of phenothrin (racemic or d-phenothrin
from 200 to 10 000 mg/kg diet) in rats and mice, with exposure
periods of 6 months to 2 years, have been performed. The no-
observed-effect levels (NOEL) obtained in these studies were 300-
1000 mg/kg diet, which correspond to approximately 40-160 mg/kg
body weight per day. In two studies on dogs in which d-phenothrin
was given at doses of 100-3000 mg/kg diet, with exposure periods of
26-52 weeks, the NOEL was 300 mg/kg diet, corresponding to 7-8
mg/kg body weight per day.
d-Phenothrin is not mutagenic in a variety of in vivo and in
vitro systems that test for gene mutations, DNA damage, DNA repair,
and chromosomal effects.
In 2-year studies, d-phenothrin was not oncogenic to rats and
mice at dietary levels of up to 3000 mg/kg diet.
Neither teratogenicity nor embryotoxicity was observed in
fetuses of rabbits and mice orally administered d-phenothrin at up
to 1000 and 3000 mg/kg body weight, respectively. In a 2-
generation rat reproduction study, the NOEL was 1000 mg/kg diet.
Rats exposed by inhalation to very high doses of d-phenothrin
(up to 3760 mg/m3) for 4 h or orally to a dose of 5000 mg/kg body
weight per day for 5 days showed no myelin degeneration or axon
disruption in the sciatic nerve.
1.1.8. Effects on human beings
Although d-phenothrin has been in use for more than 10 years,
no cases of human poisoning have been reported.
There are no indications that d-phenothrin, when used as
recommended, has an adverse effect on human beings.
1.2. Conclusions
1.2.1. General population
The exposure of the general population to d-phenothrin is
expected to be very low and is not likely to present a hazard when
it is used as recommended.
1.2.2. Occupational exposure
With reasonable work practices, hygiene measures and safety
precautions, d-phenothrin is unlikely to be an occupational hazard.
1.2.3. Environment
The rapid breakdown of phenothrin in sunlight and its use
principally on stored grain imply that environmental exposure
should be very low. Environmental effects of the compound are,
therefore, extremely unlikely.
1.3. Recommendations
When d-phenothrin is used as recommended, exposure levels are
expected to be very low. However, monitoring studies should be
continued.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1. Identity
Molecular formula: C23H26O3
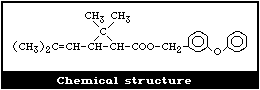 Racemic phenothrin was first synthesized by Itaya et al.
(1969). It is prepared by esterifying (1RS, cis,trans )-2,2-
dimethyl-3-(2,2-dimethylvinyl)cyclopropanecarboxylic acid
(chrysanthemic acid) with 3-phenoxybenzyl alcohol (Fujimoto et al.,
1973). Phenothrin is thus a mixture of four stereoisomers (Fig. 1).
The cis:trans isomer ratio is 1:4 and the optical ratio of 1R:1S is
1:1 (racemic). Thus the isomers 1, 2, 3, and 4 are present in the
approximate ratio of 4:1:4:1 (Table 1). d-Phenothrin is the
(1R,cis,trans) preparation (i.e., a mixture of isomers 2 and 1),
the cis:trans ratio being 1:4. The technical grade is 92.5-94.5%
pure. The major impurities found in seven d-phenothrin
preparations (average purity, 94.0%) were ethyl chrysanthemate
(2.31%), 3-phenoxy-6-bromobenzyl cis,trans-chrysanthemate (0.66%),
3-phenoxytoluene (0.43%), and 4-phenoxybenzyl cis,trans-
chrysanthemate (0.39%) (Miyamoto et al., 1984).
Racemic phenothrin was first synthesized by Itaya et al.
(1969). It is prepared by esterifying (1RS, cis,trans )-2,2-
dimethyl-3-(2,2-dimethylvinyl)cyclopropanecarboxylic acid
(chrysanthemic acid) with 3-phenoxybenzyl alcohol (Fujimoto et al.,
1973). Phenothrin is thus a mixture of four stereoisomers (Fig. 1).
The cis:trans isomer ratio is 1:4 and the optical ratio of 1R:1S is
1:1 (racemic). Thus the isomers 1, 2, 3, and 4 are present in the
approximate ratio of 4:1:4:1 (Table 1). d-Phenothrin is the
(1R,cis,trans) preparation (i.e., a mixture of isomers 2 and 1),
the cis:trans ratio being 1:4. The technical grade is 92.5-94.5%
pure. The major impurities found in seven d-phenothrin
preparations (average purity, 94.0%) were ethyl chrysanthemate
(2.31%), 3-phenoxy-6-bromobenzyl cis,trans-chrysanthemate (0.66%),
3-phenoxytoluene (0.43%), and 4-phenoxybenzyl cis,trans-
chrysanthemate (0.39%) (Miyamoto et al., 1984).
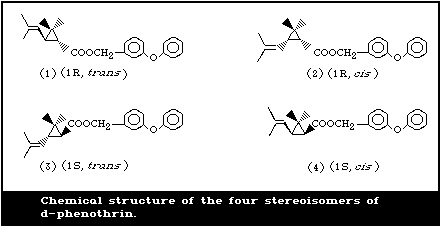 Table 1. Chemical identity of racemic phenothrin and d-phenothrin
--------------------------------------------------------------------------------------------
Common name/ CAS Index name (9CI)/ Stereoisomeric Synonyms and
CAS Registry no./ Stereospecific nameb,c compositiond trade names
RTECS Registry no.a
--------------------------------------------------------------------------------------------
Phenothrin (racemic) Cyclopropanecarboxylic acid, (1):(2):(3):(4) Phenoxythrin,
26002-80-2 2,2-dimethyl-3-(2-methyl-1-propenyl)-, =4:1:4:1 S-2539
GZ1975000 (3-phenoxyphenyl)methyl ester
3-Phenoxybenzyl (1RS, cis, trans )-,
2,2-dimethyl-3-(2,2-dimethylvinyl)-
cyclopropanecarboxylate
or
3-Phenoxybenzyl (1RS, cis, trans )-
chrysanthemate
(+) -cis, trans- Cyclopropanecarboxylic acid, (1):(2) Sumithrin,
Phenothrin 2,2-dimethyl-3-(2-methyl-1-propenyl)-, =4:1 S-2539 Forte
GZ2002000 (3-phenoxyphenyl)methyl ester d-Phenothrin
3-Phenoxybenzyl (1R, cis, trans )-
chrysanthemate
--------------------------------------------------------------------------------------------
a (NIOSH, 1983).
b (1R), d, (+) or (1S), l, (-) in the acid part of the compound signify the same
stereospecific conformation, respectively.
c Chrysanthemic acid is a name of the acid which forms the acid part of the compound.
d Numbers in parentheses identify the structures shown in Fig. 1.
2.2. Physical and Chemical Properties
Certain physical and chemical properties of d-phenothrin are
given in Table 2. It is poorly soluble in water, but is soluble in
organic solvents. d-Phenothrin is fairly stable in air but
unstable to light and in alkaline media. However, it is not
photodegraded as rapidly as the natural pyrethrins (FAO/WHO, 1980;
Worthing & Walker, 1987).
Table 2. Some physical and chemical properties of
d-phenothrin
-------------------------------------------------------
Physical state liquid
Colour pale yellow to yellow-brown
Relative molecular mass 350.5
Water solubility (25°C) 2 mg/litre
Solubility in organic solublea
solvents
25
Relative density d 1.058-1.061
25
Vapour pressure (20°C) 0.16 mPa
-------------------------------------------------------
a Hexane (>1 kg/kg), acetone, methanol (>1 kg/kg),
xylene (>1 kg/kg).
2.3. Analytical Methods
Examples of residue and product analyses of racemic phenothrin
and d-phenothrin are shown in Table 3.
To analyse technical grade racemic phenothrin or various
formulations, Sakaue et al. (1981) dissolved the product in
acetone, together with di-(2-ethylhexyl) phthalate (an internal
standard), and injected the solution into a gas chromatograph
equipped with a flame ionization detector (GC-FID). d-Phenothrin
was separated as a single peak in the analysis of formulations by a
high-performance liquid chromatography with UV detector (HPLC-UV)
system (utilizing a µ-Bondapak phenyl column eluted with
acetonitrile as water mobile phase). Murano (1972) and
Papadopoulou-Mourkidou et al. (1981) analyzed technical grade
phenothrin, and separated the cis and trans isomers of racemic or
d-phenothrin by GC-FID with a AW-DMCS chromosorb W column or by
HPLC-IR with a Partisil 10 column, respectively.
The Joint FAO/WHO Codex Alimentarius Committee has published
recommendations for methods of analysis of d-phenothrin residues
(FAO/WHO, 1985b).
Table 3. Analytical methods for racemic phenothrin
--------------------------------------------------------------------------------------------------------------------
Sample Sample preparation Determination MDCb % Recovery Refer-
Extraction Partition Clean up GLC or HPLC condition; (mg/kg) (fortification enced
solvent detector, column, level, mg/kg)c
column elution carrier flow,
retention time
--------------------------------------------------------------------------------------------------------------------
Residue analysis
apple n-hexane ext.sol.a silica gel CH2Cl2 HPLC UV-206nm, 25cm 0.05 62(0.1), 92(1.0) 1
acetone /H2O ODS, propan-2-o1,
(1/1) 1 ml/min
pear silica gel CH2Cl2 as for apple 0.05 85(0.1), 96(1.0)
cabbage silica gel CH2Cl2 as for apple 0.05 72(0.1), 88(1.0)
potato silica gel CH2Cl2 as for apple 0.05 77(0.1), 98(1.0)
wheat methanol n-hexane alumina HPLC, 235nm, 30cm, 87(2.0) 2
grain µBondapak C18,
methanol/H2O
(4/1), 2.5ml/min
cooked petroleum colorimetric method, 87(3.1) 3
rice ether 680nm 91(1.9)
ethanol
Product analysis
Technical acetone FID-GC, 1m, 2% PEG-20M 4
grade 210°C
--------------------------------------------------------------------------------------------------------------------
Table 3 (contd.)
--------------------------------------------------------------------------------------------------------------------
Sample Sample preparation Determination MDCb % Recovery Refer-
Extraction Partition Clean up GLC or HPLC condition; (mg/kg) (fortification enced
solvent detector, column, level, mg/kg)c
column elution carrier flow,
retention time
--------------------------------------------------------------------------------------------------------------------
Technical acetone FID-GC, 1m, 2% PEG-20M 5
grade or 2% DEGS-H3PO4
(total d-phenothrin)
Technical acetone FID-GC, 2m, 2% QF-1
grade (separation of
geometrical isomers)
Technical acetone HPLC, 230 nm, 50cm
grade Sumipax OA-2000
hexane/1,2-dichloro-
ethane (500/1)
(separation of
optical isomers)
--------------------------------------------------------------------------------------------------------------------
a extraction solvent
b minimum detectable concentration
c fortification level indicates the concentration of d-phenothrin added to control samples for the measurement of
recovery
d 1. Baker & Bottomley (1982), 2. Noble et al. (1982), 3. Desmarchelier (1980), 4. Sakaue et al. (1981),
5. Doi et al. (1985)
3. SOURCES AND LEVELS OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Industrial Production
Racemic phenothrin was first marketed in 1977 (Hayashi, 1977), but
is no longer commercially available. As for d-phenothrin, although
no production data are publicly available, the annual world-wide
production level is probably 70-80 tonnes.
3.2. Use Patterns
The main use of d-phenothrin is in aerosol formulations to
control household and public health insects, alone or in combination
with other insecticides (e.g., tetramethrin or d-allethrin) or
synergists (e.g., piperonyl butoxide). Oil and dust formulations are
used for the same purpose, as are emulsifiable concentrates.
d-Phenothrin is also formulated in powders, shampoos, and lotions,
mixed with a synergist (e.g., piperonyl butoxide), to control human
lice. In addition, it is used to protect stored grains.
3.3. Residues in Food
Phenothrin, being photodegradable, has a relatively short residue
time on plants.
Many residue studies (e.g., post-harvest treatment of stored
grains) have been carried out (FAO/WHO 1980, 1988a).
In supervised trials on several crops, emulsifiable formulations
of racemic phenothrin (0.375-0.50 kg ai/ha) were applied to rice,
green pepper, and cabbage, 3 to 9 times with 3 to 10 days interval
(Takimoto et al., 1977). The resultant residues were 0.005-0.008
mg/kg in cabbage (3-21 days after treatment), 0.125-1.26 mg/kg in
green pepper (1-7 days after treatment), and 0.86-2.54 mg/kg and
0.012-0.25 mg/kg in straw and hulled rice, respectively (7-14 days
after treatment in both cases).
Analyses of residues of racemic phenothrin and d-phenothrin
used for the protection of stored grains (e.g., wheat, barley, and
sorghum) have been carried out. The [1R,trans] or [1R,cis] isomer
of [methylene-14C]-phenothrin was applied at 4 mg/kg to wheat
grains (11% moisture content) and stored at 15°C or 30°C in the dark.
Both trans and cis isomers decomposed slowly; 79% and 87%,
respectively, of the applied radiocarbon remained intact in the grain
after it had been stored for one year at 30°C. The joint application
of either trans- or cis-phenothrin with piperonyl butoxide (20
mg/kg) or piperonyl butoxide plus fenitrothion (4 mg/kg) did not
significantly affect the residue levels of either isomer over a
period of 12 months. The phenothrin isomers and their decomposition
products were mainly located in the seed coat after storage for one
year, and the residue levels of both isomers in flour and bran were
0.77 and 11.4 mg/kg, respectively. The phenothrin residues in flour
decreased somewhat during the baking process, leaving 0.57 mg/kg in
bread (Nambu et al., 1981).
After wheat in a silo was treated with approximately 0.55 mg
d-phenothrin/kg, there was no evidence of residue loss during storage
for 25 weeks. Residue levels in white flour ranged from 0.15-0.22
mg/kg (approximately one third of the residues in grain), and in
white bread from 0.06-0.17 mg/kg. The decline in phenothrin residues
during baking was almost all accounted for by dilution (Turnbull &
Ardley, 1987; Ohnishi et al., 1987).
3.4. Residues in the Environment
No data are available on actual residue levels in air and water.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
Appraisal
Phenothrin undergoes rapid photodegradation under outdoor
conditions. It is transported to a very minor extent from the
site of application on plants and in soils. However, very
limited movement of phenothrin and its degradation products
from soil into bean plants was detected using radiolabelled
compounds. Phenothrin remains almost intact on stored grains
in the dark for up to 12 months.
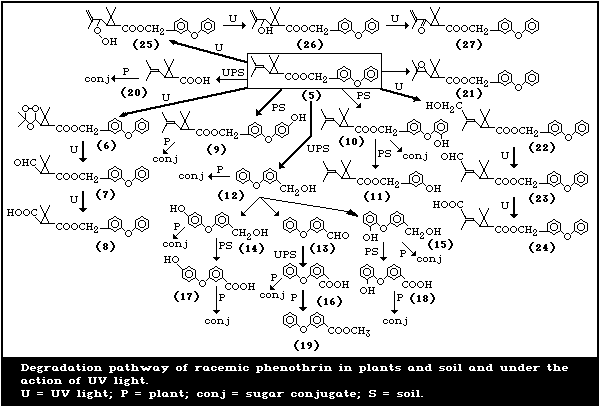
The degradation pathways of phenothrin under environmental
conditions are summarized in Fig. 2.
Table 1. Chemical identity of racemic phenothrin and d-phenothrin
--------------------------------------------------------------------------------------------
Common name/ CAS Index name (9CI)/ Stereoisomeric Synonyms and
CAS Registry no./ Stereospecific nameb,c compositiond trade names
RTECS Registry no.a
--------------------------------------------------------------------------------------------
Phenothrin (racemic) Cyclopropanecarboxylic acid, (1):(2):(3):(4) Phenoxythrin,
26002-80-2 2,2-dimethyl-3-(2-methyl-1-propenyl)-, =4:1:4:1 S-2539
GZ1975000 (3-phenoxyphenyl)methyl ester
3-Phenoxybenzyl (1RS, cis, trans )-,
2,2-dimethyl-3-(2,2-dimethylvinyl)-
cyclopropanecarboxylate
or
3-Phenoxybenzyl (1RS, cis, trans )-
chrysanthemate
(+) -cis, trans- Cyclopropanecarboxylic acid, (1):(2) Sumithrin,
Phenothrin 2,2-dimethyl-3-(2-methyl-1-propenyl)-, =4:1 S-2539 Forte
GZ2002000 (3-phenoxyphenyl)methyl ester d-Phenothrin
3-Phenoxybenzyl (1R, cis, trans )-
chrysanthemate
--------------------------------------------------------------------------------------------
a (NIOSH, 1983).
b (1R), d, (+) or (1S), l, (-) in the acid part of the compound signify the same
stereospecific conformation, respectively.
c Chrysanthemic acid is a name of the acid which forms the acid part of the compound.
d Numbers in parentheses identify the structures shown in Fig. 1.
2.2. Physical and Chemical Properties
Certain physical and chemical properties of d-phenothrin are
given in Table 2. It is poorly soluble in water, but is soluble in
organic solvents. d-Phenothrin is fairly stable in air but
unstable to light and in alkaline media. However, it is not
photodegraded as rapidly as the natural pyrethrins (FAO/WHO, 1980;
Worthing & Walker, 1987).
Table 2. Some physical and chemical properties of
d-phenothrin
-------------------------------------------------------
Physical state liquid
Colour pale yellow to yellow-brown
Relative molecular mass 350.5
Water solubility (25°C) 2 mg/litre
Solubility in organic solublea
solvents
25
Relative density d 1.058-1.061
25
Vapour pressure (20°C) 0.16 mPa
-------------------------------------------------------
a Hexane (>1 kg/kg), acetone, methanol (>1 kg/kg),
xylene (>1 kg/kg).
2.3. Analytical Methods
Examples of residue and product analyses of racemic phenothrin
and d-phenothrin are shown in Table 3.
To analyse technical grade racemic phenothrin or various
formulations, Sakaue et al. (1981) dissolved the product in
acetone, together with di-(2-ethylhexyl) phthalate (an internal
standard), and injected the solution into a gas chromatograph
equipped with a flame ionization detector (GC-FID). d-Phenothrin
was separated as a single peak in the analysis of formulations by a
high-performance liquid chromatography with UV detector (HPLC-UV)
system (utilizing a µ-Bondapak phenyl column eluted with
acetonitrile as water mobile phase). Murano (1972) and
Papadopoulou-Mourkidou et al. (1981) analyzed technical grade
phenothrin, and separated the cis and trans isomers of racemic or
d-phenothrin by GC-FID with a AW-DMCS chromosorb W column or by
HPLC-IR with a Partisil 10 column, respectively.
The Joint FAO/WHO Codex Alimentarius Committee has published
recommendations for methods of analysis of d-phenothrin residues
(FAO/WHO, 1985b).
Table 3. Analytical methods for racemic phenothrin
--------------------------------------------------------------------------------------------------------------------
Sample Sample preparation Determination MDCb % Recovery Refer-
Extraction Partition Clean up GLC or HPLC condition; (mg/kg) (fortification enced
solvent detector, column, level, mg/kg)c
column elution carrier flow,
retention time
--------------------------------------------------------------------------------------------------------------------
Residue analysis
apple n-hexane ext.sol.a silica gel CH2Cl2 HPLC UV-206nm, 25cm 0.05 62(0.1), 92(1.0) 1
acetone /H2O ODS, propan-2-o1,
(1/1) 1 ml/min
pear silica gel CH2Cl2 as for apple 0.05 85(0.1), 96(1.0)
cabbage silica gel CH2Cl2 as for apple 0.05 72(0.1), 88(1.0)
potato silica gel CH2Cl2 as for apple 0.05 77(0.1), 98(1.0)
wheat methanol n-hexane alumina HPLC, 235nm, 30cm, 87(2.0) 2
grain µBondapak C18,
methanol/H2O
(4/1), 2.5ml/min
cooked petroleum colorimetric method, 87(3.1) 3
rice ether 680nm 91(1.9)
ethanol
Product analysis
Technical acetone FID-GC, 1m, 2% PEG-20M 4
grade 210°C
--------------------------------------------------------------------------------------------------------------------
Table 3 (contd.)
--------------------------------------------------------------------------------------------------------------------
Sample Sample preparation Determination MDCb % Recovery Refer-
Extraction Partition Clean up GLC or HPLC condition; (mg/kg) (fortification enced
solvent detector, column, level, mg/kg)c
column elution carrier flow,
retention time
--------------------------------------------------------------------------------------------------------------------
Technical acetone FID-GC, 1m, 2% PEG-20M 5
grade or 2% DEGS-H3PO4
(total d-phenothrin)
Technical acetone FID-GC, 2m, 2% QF-1
grade (separation of
geometrical isomers)
Technical acetone HPLC, 230 nm, 50cm
grade Sumipax OA-2000
hexane/1,2-dichloro-
ethane (500/1)
(separation of
optical isomers)
--------------------------------------------------------------------------------------------------------------------
a extraction solvent
b minimum detectable concentration
c fortification level indicates the concentration of d-phenothrin added to control samples for the measurement of
recovery
d 1. Baker & Bottomley (1982), 2. Noble et al. (1982), 3. Desmarchelier (1980), 4. Sakaue et al. (1981),
5. Doi et al. (1985)
3. SOURCES AND LEVELS OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Industrial Production
Racemic phenothrin was first marketed in 1977 (Hayashi, 1977), but
is no longer commercially available. As for d-phenothrin, although
no production data are publicly available, the annual world-wide
production level is probably 70-80 tonnes.
3.2. Use Patterns
The main use of d-phenothrin is in aerosol formulations to
control household and public health insects, alone or in combination
with other insecticides (e.g., tetramethrin or d-allethrin) or
synergists (e.g., piperonyl butoxide). Oil and dust formulations are
used for the same purpose, as are emulsifiable concentrates.
d-Phenothrin is also formulated in powders, shampoos, and lotions,
mixed with a synergist (e.g., piperonyl butoxide), to control human
lice. In addition, it is used to protect stored grains.
3.3. Residues in Food
Phenothrin, being photodegradable, has a relatively short residue
time on plants.
Many residue studies (e.g., post-harvest treatment of stored
grains) have been carried out (FAO/WHO 1980, 1988a).
In supervised trials on several crops, emulsifiable formulations
of racemic phenothrin (0.375-0.50 kg ai/ha) were applied to rice,
green pepper, and cabbage, 3 to 9 times with 3 to 10 days interval
(Takimoto et al., 1977). The resultant residues were 0.005-0.008
mg/kg in cabbage (3-21 days after treatment), 0.125-1.26 mg/kg in
green pepper (1-7 days after treatment), and 0.86-2.54 mg/kg and
0.012-0.25 mg/kg in straw and hulled rice, respectively (7-14 days
after treatment in both cases).
Analyses of residues of racemic phenothrin and d-phenothrin
used for the protection of stored grains (e.g., wheat, barley, and
sorghum) have been carried out. The [1R,trans] or [1R,cis] isomer
of [methylene-14C]-phenothrin was applied at 4 mg/kg to wheat
grains (11% moisture content) and stored at 15°C or 30°C in the dark.
Both trans and cis isomers decomposed slowly; 79% and 87%,
respectively, of the applied radiocarbon remained intact in the grain
after it had been stored for one year at 30°C. The joint application
of either trans- or cis-phenothrin with piperonyl butoxide (20
mg/kg) or piperonyl butoxide plus fenitrothion (4 mg/kg) did not
significantly affect the residue levels of either isomer over a
period of 12 months. The phenothrin isomers and their decomposition
products were mainly located in the seed coat after storage for one
year, and the residue levels of both isomers in flour and bran were
0.77 and 11.4 mg/kg, respectively. The phenothrin residues in flour
decreased somewhat during the baking process, leaving 0.57 mg/kg in
bread (Nambu et al., 1981).
After wheat in a silo was treated with approximately 0.55 mg
d-phenothrin/kg, there was no evidence of residue loss during storage
for 25 weeks. Residue levels in white flour ranged from 0.15-0.22
mg/kg (approximately one third of the residues in grain), and in
white bread from 0.06-0.17 mg/kg. The decline in phenothrin residues
during baking was almost all accounted for by dilution (Turnbull &
Ardley, 1987; Ohnishi et al., 1987).
3.4. Residues in the Environment
No data are available on actual residue levels in air and water.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
Appraisal
Phenothrin undergoes rapid photodegradation under outdoor
conditions. It is transported to a very minor extent from the
site of application on plants and in soils. However, very
limited movement of phenothrin and its degradation products
from soil into bean plants was detected using radiolabelled
compounds. Phenothrin remains almost intact on stored grains
in the dark for up to 12 months.
The degradation pathways of phenothrin under environmental
conditions are summarized in Fig. 2.
 4.1. Transport and Distribution between Media
The degree of leaching of [1RS, trans]- or [1RS, cis]-phenothrin
has been studied under laboratory conditions. Very little movement
(less than 2%) of trans- and cis-phenothrin through soil columns
occurred when leaching was started either immediately or 14 days
after treatment (Nambu et al., 1980).
4.2. Photodegradationa
Although d-phenothrin is more resistant to photolysis than
pyrethrin I, allethrin, and resmethrin (due to a more stable
alcohol moiety), it still possesses the photo-labile isobutenyl
group in the molecule and therefore is easily photodecomposed.
Ruzo et al. (1982) investigated the photodegradation of
[1RS, trans]-phenothrin (5)a in oxygenated benzene solution (10-3
mol/litre) under UV light (360 nm) or in a thin film (0.1-0.3 mg/cm2)
under sunlight (Fig. 2). Exposure to sunlight resulted in 30%
conversion, the major photoproducts being:
* the (1RS) epoxides (21) (22% of the reaction mixture), the
alcohol (22) (9%), and the aldehyde (23) (13%) derivatives from
oxidation at the (E)-methyl group;
* the caronaldehyde derivative (7) (4%) from ozonolysis;
* the hydroperoxide (25) (27%) from hydroperoxidation, including
migration of the double bond at the 1'-position of the
isobutenyl moiety (Fig. 2).
Minor products (26) and (27) (3%) resulted from further oxidation
of the hydroperoxide (25) and from cis/trans isomerization and
ester cleavage. Trace amounts of trans-chrysanthemic acid (20)
were detected. 3-Phenoxybenzyl alcohol (12) underwent further
oxidation to form the aldehyde (13) and carboxylic acid (16).
Unidentified photoproducts accounted for 16.6% of the total. A
similar product distribution was obtained in benzene solution.
4.3. Degradation in Plants and Soils
In studies by Nambu et al. (1980), 14C-methylene-labelled
[1R, trans]-or [1R, cis ]-phenothrin (each 10 mg/kg) disappeared from
the treated leaves of kidney bean or rice plants with half-lives of
less than one day under greenhouse conditions. The residue levels
in both plants were 0.04-0.28 mg/kg for trans- phenothrin and 0.10-
0.30 mg/kg for cis- phenothrin after 30 days, compared with
approximately 10 mg/kg immediately after treatment. Both isomers
primarily underwent ozonolysis at the isobutenyl double bond,
probably via the photochemical reactions indicated previously
(Fig. 2). The resultant ozonides (6) were detected soon after
treatment but they were rapidly decomposed to the corresponding
aldehyde (7) and carboxylic acid (8) (3-phenoxy-benzyl-2,2-
dimethyl-3-carboxy-cyclopropanecarboxylate) derivatives. Cleavage
of the ester linkage also occurred, together with hydroxylation at
the 2'-position (10) or 4'-position (9) of the alcohol moiety.
Conjugation of the acids and alcohols with sugars was also
observed, and the formation of polar products was more extensive in
rice than in bean plants. There was little translocation of trans-
------------------------------------------------------------------------
a The numbers in brackets following a chemical name refer to the
numbers given in Fig. 2.
or cis-phenothrin or of its degradation products to the untreated
parts of the plants. Limited uptake of radiolabelled products into
bean plants took place from light clay, sandy loam soil, and from
sand treated with 14C-methylene-labelled trans- or cis-phenothrin.
The degradation of 14C-labelled [1R, trans]- or [1R, cis]-
phenothrin in two soils was investigated by Nambu et al. (1980).
Both isomers decomposed rapidly under upland conditions, with
initial half-lives of 1-2 days, but under flooded conditions the
degradation was much slower, with initial half-lives of 2-4 weeks
and 1-2 months for the trans and cis isomers, respectively.
Analysis of the soil extracts revealed unchanged parent isomers,
(16), 3-(4'-hydroxyphenoxy)benzoic acid (17), (12), 3-(4'-
hydroxyphenoxy)benzyl alcohol (14), (9), and 3-hydroxy-benzyl-2,2-
dimethyl-3-(2,2-dimethylvinyl)-cyclopropanecarboxylate (11). These
degradation products were not persistent and underwent further
degradation in soil, under both upland and flooded conditions, to
yield large amounts of 14CO2 and unextractably bound residues.
More 14C carbon dioxide was formed in soils under upland conditions
than under flooded conditions and more was formed from trans-
phenothrin than from the cis isomer. Bound 14C residues were
associated mainly with the humic acid and fulvic acid fractions of
soil organic matter. The fulvic acid fraction contained small
amounts of the same degradation products as in the soil extracts.
4.4. Degradation on Stored Foods
When 14C-[1R, trans ]- or 14C-[1R, cis ]-phenothrin was applied at
4 mg/kg to wheat grains of 11% moisture content and the crop was
stored at 15 or 30°C in the dark for 12 months, most of the
phenothrin remained intact. Major metabolites were formed by
hydrolysis of the ester linkage and oxidation of 3-phenoxybenzyl
alcohol to 3-phenoxybenzoic acid. The methyl ester of 3-
phenoxybenzoic acid was also produced. After storage at 30°C for
12 months, these metabolites amounted to 13.9% in the case of the
trans isomer and 6.3% in the case of the cis isomer (Nambu et al.,
1981).
5. KINETICS AND METABOLISM
Appraisal
The pathways by which phenothrin is metabolized in mammals are
summarized in Fig. 3.
Comparative metabolism studies have shown that the metabolism
of racemic phenothrin is similar to that of [1R,cis,trans]-phenothrin
(d-phenothrin).
No information is available on the in vivo metabolism of the acid
moieties of trans([1R,trans])- or cis([1R,cis])-phenothrin. However,
the acid moiety liberated in vivo (chrysanthemic acid) is the same
as that of resmethrin and tetramethrin and, therefore, its fate can
be predicted from the resmethrin and tetramethrin data.
4.1. Transport and Distribution between Media
The degree of leaching of [1RS, trans]- or [1RS, cis]-phenothrin
has been studied under laboratory conditions. Very little movement
(less than 2%) of trans- and cis-phenothrin through soil columns
occurred when leaching was started either immediately or 14 days
after treatment (Nambu et al., 1980).
4.2. Photodegradationa
Although d-phenothrin is more resistant to photolysis than
pyrethrin I, allethrin, and resmethrin (due to a more stable
alcohol moiety), it still possesses the photo-labile isobutenyl
group in the molecule and therefore is easily photodecomposed.
Ruzo et al. (1982) investigated the photodegradation of
[1RS, trans]-phenothrin (5)a in oxygenated benzene solution (10-3
mol/litre) under UV light (360 nm) or in a thin film (0.1-0.3 mg/cm2)
under sunlight (Fig. 2). Exposure to sunlight resulted in 30%
conversion, the major photoproducts being:
* the (1RS) epoxides (21) (22% of the reaction mixture), the
alcohol (22) (9%), and the aldehyde (23) (13%) derivatives from
oxidation at the (E)-methyl group;
* the caronaldehyde derivative (7) (4%) from ozonolysis;
* the hydroperoxide (25) (27%) from hydroperoxidation, including
migration of the double bond at the 1'-position of the
isobutenyl moiety (Fig. 2).
Minor products (26) and (27) (3%) resulted from further oxidation
of the hydroperoxide (25) and from cis/trans isomerization and
ester cleavage. Trace amounts of trans-chrysanthemic acid (20)
were detected. 3-Phenoxybenzyl alcohol (12) underwent further
oxidation to form the aldehyde (13) and carboxylic acid (16).
Unidentified photoproducts accounted for 16.6% of the total. A
similar product distribution was obtained in benzene solution.
4.3. Degradation in Plants and Soils
In studies by Nambu et al. (1980), 14C-methylene-labelled
[1R, trans]-or [1R, cis ]-phenothrin (each 10 mg/kg) disappeared from
the treated leaves of kidney bean or rice plants with half-lives of
less than one day under greenhouse conditions. The residue levels
in both plants were 0.04-0.28 mg/kg for trans- phenothrin and 0.10-
0.30 mg/kg for cis- phenothrin after 30 days, compared with
approximately 10 mg/kg immediately after treatment. Both isomers
primarily underwent ozonolysis at the isobutenyl double bond,
probably via the photochemical reactions indicated previously
(Fig. 2). The resultant ozonides (6) were detected soon after
treatment but they were rapidly decomposed to the corresponding
aldehyde (7) and carboxylic acid (8) (3-phenoxy-benzyl-2,2-
dimethyl-3-carboxy-cyclopropanecarboxylate) derivatives. Cleavage
of the ester linkage also occurred, together with hydroxylation at
the 2'-position (10) or 4'-position (9) of the alcohol moiety.
Conjugation of the acids and alcohols with sugars was also
observed, and the formation of polar products was more extensive in
rice than in bean plants. There was little translocation of trans-
------------------------------------------------------------------------
a The numbers in brackets following a chemical name refer to the
numbers given in Fig. 2.
or cis-phenothrin or of its degradation products to the untreated
parts of the plants. Limited uptake of radiolabelled products into
bean plants took place from light clay, sandy loam soil, and from
sand treated with 14C-methylene-labelled trans- or cis-phenothrin.
The degradation of 14C-labelled [1R, trans]- or [1R, cis]-
phenothrin in two soils was investigated by Nambu et al. (1980).
Both isomers decomposed rapidly under upland conditions, with
initial half-lives of 1-2 days, but under flooded conditions the
degradation was much slower, with initial half-lives of 2-4 weeks
and 1-2 months for the trans and cis isomers, respectively.
Analysis of the soil extracts revealed unchanged parent isomers,
(16), 3-(4'-hydroxyphenoxy)benzoic acid (17), (12), 3-(4'-
hydroxyphenoxy)benzyl alcohol (14), (9), and 3-hydroxy-benzyl-2,2-
dimethyl-3-(2,2-dimethylvinyl)-cyclopropanecarboxylate (11). These
degradation products were not persistent and underwent further
degradation in soil, under both upland and flooded conditions, to
yield large amounts of 14CO2 and unextractably bound residues.
More 14C carbon dioxide was formed in soils under upland conditions
than under flooded conditions and more was formed from trans-
phenothrin than from the cis isomer. Bound 14C residues were
associated mainly with the humic acid and fulvic acid fractions of
soil organic matter. The fulvic acid fraction contained small
amounts of the same degradation products as in the soil extracts.
4.4. Degradation on Stored Foods
When 14C-[1R, trans ]- or 14C-[1R, cis ]-phenothrin was applied at
4 mg/kg to wheat grains of 11% moisture content and the crop was
stored at 15 or 30°C in the dark for 12 months, most of the
phenothrin remained intact. Major metabolites were formed by
hydrolysis of the ester linkage and oxidation of 3-phenoxybenzyl
alcohol to 3-phenoxybenzoic acid. The methyl ester of 3-
phenoxybenzoic acid was also produced. After storage at 30°C for
12 months, these metabolites amounted to 13.9% in the case of the
trans isomer and 6.3% in the case of the cis isomer (Nambu et al.,
1981).
5. KINETICS AND METABOLISM
Appraisal
The pathways by which phenothrin is metabolized in mammals are
summarized in Fig. 3.
Comparative metabolism studies have shown that the metabolism
of racemic phenothrin is similar to that of [1R,cis,trans]-phenothrin
(d-phenothrin).
No information is available on the in vivo metabolism of the acid
moieties of trans([1R,trans])- or cis([1R,cis])-phenothrin. However,
the acid moiety liberated in vivo (chrysanthemic acid) is the same
as that of resmethrin and tetramethrin and, therefore, its fate can
be predicted from the resmethrin and tetramethrin data.
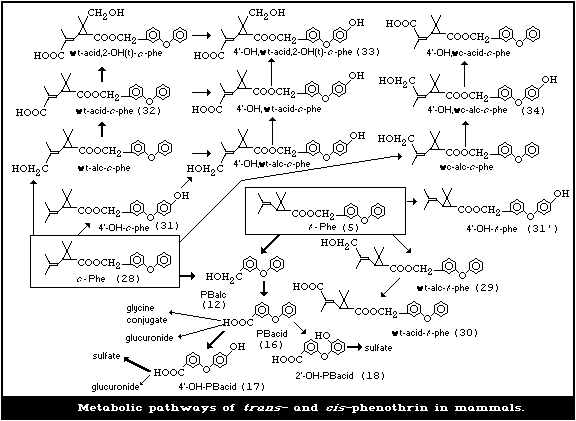 5.1. Metabolism in Mammals
Appraisal
After rats are treated with radiolabelled phenothrin, either by
single or repeated oral exposure, or dermally, the radioactivity is
rapidly (and almost totally) excreted into urine and faeces within
3 to 7 days. The major metabolic pathways of both trans- and cis-
phenothrin in rats are oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety and cleavage of
the ester linkage (see Fig. 3). Cleavage of the ester bond is more
difficult in the cis isomer than in the trans isomer. Thus, ester-
form metabolites oxidized in the various positions of the molecule are
the major metabolites of the cis isomer and are excreted mostly in the
faeces. However, ester-cleaved metabolites are the major products from
the trans isomer and are mostly excreted in urine.
When [1R, trans ]-phenothrin labelled with 14C at the methylene
moiety was given as a single oral dose (200 mg/kg body weight) to
Sprague Dawley male rats, the radiocarbon was rapidly eliminated,
57% and 43% being recovered within 3 days in urine and faeces,
respectively (Miyamoto et al., 1974). There was no detectable
radiocarbon in the expired air. Similarly, Sprague Dawley male
rats given a single oral dose (10 mg/kg body weight) of 14C-
[1R, trans ]-phenothrin, excreted the radiocarbon rapidly in the
urine (75%) and faeces (21%) (Kaneko et al., 1981).
In a study by Isobe et al. (1987), Sprague Dawley male and
female rats were given a single oral administration of 14C-
[1R, trans ]-phenothrin in corn oil at 4 and 200 mg/kg body weight.
Within 7 days the radiocarbon was almost completely eliminated in
the urine and faeces. The % elimination was as follows:
-----------------------------------------
Dose Urine Faeces
(mg/kg) Males Females Males Females
4 38 40 61 60
200 39 25 56 60
-----------------------------------------
Sprague Dawley male rats eliminated 65% of the dosed radiocarbon
in the faeces over 3 days after a single oral administration (200
mg/kg body weight) of 14C-[1R, cis ]-phenothrin (28) (Suzuki et al.,
1976), and 22 and 74% of the dose into urine and faeces,
respectively, 7 days after a single oral dose of 10 mg/kg body
weight (Kaneko et al., 1981).
When 14C-[1R, cis ]-phenothrin in corn oil was administered once
orally to Sprague Dawley male and female rats at 4 or 200 mg/kg
body weight, the radiocarbon was excreted into the urine (11-18%)
and faeces (81-87%) within 7 days. Similarly, when Sprague Dawley
rats were treated repeatedly with 14C-[1R,trans] or 14C-[1R,cis]
isomers at 4 mg/kg body weight per day for 14 days, the radiocarbon
was rapidly and almost completely excreted: 75-70% in urine and 24-
29% in faeces for the trans isomer, and 24% in urine and 72-73% in
faeces for the cis isomer (Isobe et. al., 1987).
The tissue residues in rats 7 days after a single oral dose of
14C-[1R, cis ]- or 14C-[1R, trans ]-phenothrin at 10 mg/kg body
weight were generally very low although the fat showed somewhat
higher residue levels (1-2.5 mg/kg) (Kaneko et al., 1981).
Similarly, high 14C residue levels (up to 23 mg/kg) were found in
the fat, 7 days after a single oral dose of the [1R,cis] isomer at
200 mg/kg body weight (Isobe et al., 1987).
The major metabolite of the trans isomer, when given as a
single oral dose of 200 mg/kg to rats, was 3-(4'-hydroxyphenoxy)-
benzoic acid (17) (4'OH-PBacid, 54%). There were smaller amounts
of 3-phenoxybenzoic acid (16) (PBacid, 9.5%) and its glycine
conjugate (Miyamoto et al., 1974).
When [1R, trans ]-phenothrin was given to rats at 4, 10, or 200
mg/kg body weight (oral single dose) or 4 mg/kg body weight
(repetitive oral dose for 14 days), the sulfate conjugate of 4'-OH-
PBacid was predominant, accounting for 28, 43, 28, and 55%,
respectively, of the dose. In addition, PBacid (4, 10, 5, and 6%),
its glycine conjugate (1,3,2, and 2%) and glucuronide (2,3,1, and
3%), and free 4'-OH-PBacid (2,11,3, and 3%) were found. The
sulfate conjugate of 3-(2'-hydroxyphenoxy)benzoic acid (18) (2'-
OH-PBacid) was also found as a minor metabolite (Kaneko et al.,
1981; Isobe et al., 1987).
When rats were given 14C-[1R, trans ]-phenothrin (10 mg/kg body
weight), the unmetabolized compound and two ester-form metabolites
were detected in their faeces in small amounts (0.4-1.2%), which
had hydroxymethyl (29) ( wt-alc- t-phe) or carboxyl group (30) wt-
acid- t-phe) (see Fig. 2) at the position of the trans methyl group
of chrysanthemic acid (Kaneko et al., 1981).
When Sprague Dawley rats were administered a single oral dose
of [1R, trans ]-phenothrin at 4 or 200 mg/kg body weight level or
given an oral dose of 4 mg/kg body weight per day for 14 days,
unmetabolized compound was found in the faeces (44-45, 44-60, and
14-16% of the dose, respectively). An ester-form metabolite, the
4'-hydroxy w(t)-acid derivative of trans- phenothrin, was also
detected (0.4-0.6%) (Isobe et al., 1987).
When male Sprague Dawley rats were given cis-phenothrin (200
mg/kg body weight), three ester-form metabolites, which accounted
for 14% of the dosed radioactivity, were found in the faeces.
These were 4'-hydroxy- cis-phenothrin (31) (4-OH- c-phe), an ester-
form derivative with the trans methyl of the isobutenyl group being
oxidized to carboxyl group (32) ( wt-acid- c-phe), and a compound
with the geminal-dimethyl groups oxidized (2-OH-) in addition to
both of the above modifications (33) (4'-OH, wt-acid, 2-OH(t)- c-phe)
(Suzuki et al., 1976).
In addition to this, the cis isomer gave rise to nine ester-
form metabolites in the faeces varying in amounts from 2% (4'-OH,
wc-alc-c-phe (34)) to 13% (4'-OH, wt-acid,2-OH(t)- c-phe (33)) of
the dosed radiocarbon after single oral administration. These
ester-form metabolites were transformed by oxidation reactions at
any of the following positions: 4'-position of the phenoxy group,
the trans or cis methyl of the isobutenyl groups, and the trans
methyl of the geminal-dimethyl group (Kaneko et al., 1981).
When Sprague Dawley rats were given a single oral dose of
[1R, cis ]-phenothrin at 4 or 200 mg/kg body weight level or an oral
dose of [1R, cis ]-phenothrin at 4 mg/kg body weight per day for 14
days, ester-form metabolites (1-9% of the dosed radioactivity) were
found, in addition to unmetabolized compound (17-59% of the dose).
The urine contained 4'-OH-PBacid as a sulfate conjugate (7-18%) and
in the free form (0.3-1%), and PBacid as glycine or glucuronide
conjugates and in the free form (0.3-1%) (Isobe et al., 1987).
Following the dermal treatment of male Sprague Dawley rats with
dust or emulsifiable concentrates (E.C.) of either 14C-[1R, trans ]-
or 14C-[1R, cis ]-phenothrin at 10 mg/kg body weight, the 14C
absorption into the body was estimated to be 3-7% of the initial
dose with dust and 8-17% with the E.C. After both dust and E.C.
treatments, the radiocarbon excreta (as a percentage of the initial
dose) recovered in the urine was 2.6-8.7% for the trans isomer, and
1.5-4.8% for the cis isomer, and in the faeces was 0.6-2.2% for the
trans isomer, and 3.0-12.3% for the cis isomer. Since the same
metabolites are formed following either oral exposure or dermal
treatment, it appears that both phenothrin isomers undergo the same
metabolism once in the systemic circulation, regardless of the
route of administration (Kaneko et al., 1981; Isobe et al., 1987).
Information concerning the comparative metabolism of racemic
(1RS) phenothrin and its d-isomer (1R) was obtained through a study
of CD rats and ddY mice given a single oral dose of either
[1R, trans ]-, [1S, trans ]-, [1RS, trans ]-, [1R, cis ]-, [1S, cis ]-,
or [1RS, cis ]-phenothrin. The radiocarbon derived from each isomer
was almost completely eliminated from the rats and mice within six
days after dosing. The trans isomers were mainly eliminated in the
urine (rat, 85-88%; mice, 65-75%) and the cis isomers mainly in the
faeces (rat, 57-71%; mice, 54-71%). The amounts of 14C in the
urine and faeces of rats and mice treated with the [1R,trans] and
[1R,cis] isomers did not differ significantly from those
corresponding to the [1RS,trans] and [1RS,cis] isomers, respectively.
The 14C tissue residues were very low, except in the fat. There
were no striking differences in 14C levels among the three trans
isomers and the three cis isomers. The 14C levels of the cis
isomers in fat (maximum 3.5 mg/kg) were three to seven times higher
than those of the trans isomers (less than 1 mg/ kg). The major
urinary and faecal metabolites were remarkably similar in both rats
and mice. In both rats and mice, there were virtually no
differences in the metabolic fate of the [1R,trans] and [1RS,
trans] isomers or of the [1R,cis] and [1RS,cis] isomers (Izumi et
al., 1984).
5.2. Enzymatic Systems for Biotransformation
In studies by Miyamoto et al. (1974), [IR, trans ]-phenothrin
(1 mmol/litre) was incubated with the 8000-g supernatant from a
liver homogenate of rats, mice, guinea-pigs, rabbits, or dogs at
37°C for 60 min in the absence of NADPH. The supernatant from the
guinea-pig was the most active in degrading [1R, trans ]-phenothrin,
followed by that of dog, rabbit, rat, and mouse. The major
metabolite in all the mammalian species tested was 3-phenoxybenzyl
alcohol (12) (PBalc). Smaller amounts of PBacid (16) and trace
amounts of 4'-OH-PBacid (17) were also found. However, in the
presence of NADPH, the amounts of PBacid and unidentified ether-
soluble metabolites increased in all species except dog. In
contrast to [1R, trans ]-phenothrin, [1R, cis ]-phenothrin was hardly
metabolized at all by the rat liver preparation in the absence of
NADPH. NADPH enhanced the degradation rate of the cis isomer,
leading to the formation of unidentified metabolites, while ester-
cleaved metabolites such as PBacid (16), PBalc (12) and 4'-OH-
PBacid (17) were found in very small amounts. When [1R, trans ]-,
[1R, cis ]-, [1S, trans ]-, and [1S, cis ]-phenothrin were incubated
with rat liver microsomes at 37.5°C for 30-60 min to estimate Km
and Vmax using a Lineweaver-Burk plot, the values for Km (0.11-0.17
mmol/litre) were similar for the four isomers, whereas the values
for Vmax were different; both the trans isomers yielded values for
Vmax 20-30 times larger than did the cis isomers (Miyamoto et al.,
1974).
In studies by Suzuki & Miyamoto (1978), pyrethroid
carboxyesterase(s) that hydrolyze esters of chrysanthemic acid
were purified from rat liver microsomes by cholic acid
solubilization, ammonium sulfate fractionation, heat treatment, and
DEAE-Sephadex A-50 column chromatography. The 45-fold-purified
enzyme (38% yield) probably consisted of a single protein with a
relative molecular mass of approximately 74 000, a Km of 0.21
mmol/litre for [1R, trans ]-phenothrin, and an optimum pH of 7-9.
It was susceptible to inhibition by organophosphate and carbamate
insecticides and insensitive to PCMB ( p-chloromercurybenzoic acid),
and mercuric and cupric ions. The enzyme seemed to require neither
coenzymes nor cofactors and hydrolysed the trans isomers of several
synthetic pyrethroids (tetramethrin, resmethrin, phenothrin, and
permethrin) well, at more or less similar rates. On the other hand,
the cis isomers were hydrolysed at rates 5-10 times lower than
their trans counterparts. The purified pyrethroid carboxyesterase
was apparently identical in nature to malathion carboxyesterase
and p-nitrophenyl acetate carboxyesterase.
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
Data on the acute toxicity of racemic and isomeric phenothrin
for aquatic organisms are summarized in Table 4.
6.1. Aquatic Organisms
Racemic phenothrin yielded 96-h LC50 values of 17-200 µg/litre
for the fish species tested (Table 4). The (1S)-optical isomers
were relatively non-toxic with LC50 values of 10 000 µg/litre,
whereas the (1R)-optical isomer and racemic phenothrin were of
similar toxicity with LC50 values between 120 and 200 µg/litre for
the killifish (Oryzias latipes) (Miyamoto, 1976).
When d-phenothrin was applied to ponds at the rates of 28 or 56
g/ha to control mosquito larvae, mayfly naiads were most affected
but no other arthropods (damselfly, dragonfly naiads, ostracods, or
diving beetle larvae) were seriously affected (Mulla et al., 1980).
6.2. Terrestrial Organisms
The available toxicity data for non-target terrestrial
organisms are very limited.
Phenothrin has low toxicity (acute oral dosage) for bobwhite
quail with an LD50 >2510 mg/kg body weight (Worthing & Walker,
1987). An 8-day feeding study with d-phenothrin on mallard duck
and bobwhite quail indicated LD50 values of >5620 and >5000 mg/kg
diet, respectivelya.
----------------------------------------------------------------------
a Personal communication from J.L. Noles, Ecological Effects
Branch, Hazard Evaluation Division, US Environmental Protection
Agency, November 1987.
Table 4. The acute toxicity of racemic and isomeric phenothrin to non-target aquatic organisms
-----------------------------------------------------------------------------------------------
Species Size Parameter Toxic Stereo- Systema Temper- Refer-
concentration isomeric ature enceb
(µg/litre) composition (°C)
-----------------------------------------------------------------------------------------------
Fish
Killifish adult 48-h LC50 200 racemic S 25 1
(Oryzias latipes) adult 48-h LC50 120 (+)-trans S 25 1
adult 48-h LC50 170 (+)-cis S 25 1
adult 48-h LC50 10 000 (-)-trans S 25 1
adult 48-h LC50 10 000 (-)-cis S 25 1
Rainbow trout 96-h LC50 17 racemic 2
(Salmo gairdneri)
Bluegill 96-h LC50 18 racemic 2
(Lepomis macrochirus)
Arthropods
Daphnia pulex 3-h LC50 50 000 racemic S 25 1
3-h LC50 25 000-50 000 (+)-trans S 25 1
3-h LC50 50 000 (+)-cis S 25 1
3-h LC50 50 000 (-)-trans S 25 1
3-h LC50 50 000 (-)-cis S 25 1
-----------------------------------------------------------------------------------------------
a s = static
b 1. Miyamoto (1976) 2. Worthing & Walker (1987)
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
The 1984 Joint FAO/WHO Meeting on Pesticide Residues concluded
that the toxicological data for racemic phenothrin can be used to
support that for d-phenothrin, owing to the similarity in
metabolism and toxicity between the two compounds (FAO/WHO,
1985b).
7.1. Single and Short-Term Exposures
The acute toxicity in rats and mice is extremely low. The LD50
values were >5000 mg/kg body weight when d-phenothrin was
administered orally, subcutaneously, dermally, or by
intraperitoneal injection to male and female Sprague Dawley rats
(Segawa, 1979a) and ddY mice (Segawa, 1979b) (Table 5).
Table 5. The acute toxicity of racemic phenothrin and d-phenothrin
to rats and micea
------------------------------------------------------------------
Compound/Route LD50 (mg/kg)
Rat Mice
Male Female Male Female
------------------------------------------------------------------
Phenothrin (racemic)
Oral > 5000 > 5000 > 5000 > 5000
Subcutaneous > 5000 > 5000 > 5000 > 5000
Dermal > 5000 > 5000 > 5000 > 5000
Intraperitoneal > 5000 > 5000 > 5000 > 5000
Inhalation > 1210b > 1210b > 1210b > 1210b
d-Phenothrin
Oral > 10 000 > 10 000 > 10 000 > 10 000
Subcutaneous > 10 000 > 10 000 > 10 000 > 10 000
Dermal > 10 000 > 10 000 > 5000 > 5000
Intraperitoneal > 10 000 > 10 000 > 10 000 > 10 000
Inhalation > 3760b > 3760b > 1180b > 1180b
------------------------------------------------------------------
a From: Segawa (1976; 1979a,b) and Khoda et al. (1977; 1979b; 1980)
b These are values for 4-h LC50 (mg/m3)
There were no differences between the effects on male and
female rats in acute toxicity studies carried out by Segawa (1976).
Signs of poisoning appeared rapidly following the intravenous
administration of phenothrin. These included fibrillation, tremor,
slow respiration, salivation, lacrimation, ataxia, and paralysis.
These signs, evident within 30 min to one hour following
administration, diminished rapidly to the point where, after 24 h,
there were no signs of toxicity.
No differences in the oral toxicity between racemic phenothrin
and d-phenothrin were detected by Segawa (1976; 1979a,b) (Table 5).
The intravenous LD50 values for racemic phenothrin in ICR mice
were 470 and 600 mg/kg for males and females, respectively, while
those for d-phenothrin were 265 and 315 mg/kg (Hiromori et al., 1984).
Following a 4-h acute inhalation exposure (whole body), the
LC50 values were >1210 mg/m3 for racemic phenothrin (particle size
5 µm) with both Sprague Dawley rats and ICR mice (Kohda et al., 1979b)
and >3760 and 1180 mg/m3 for d-phenothrin with Sprague Dawley rats
and ICR mice, respectively (Kohda et al., 1977; 1979b) (see Table 5).
At the higher concentration (3760 mg/m3), the mean particle size
was considered to be 0.72 µm and the cumulative distribution of
particles having a diameter between 0.46 and 1.09 µm was 88.9%,
according to a particle-size distribution experiment (Kohda et al.,
1980).
In studies by Kohda et al. (1979b), Sprague Dawley rats (15 of
each sex) were exposed (whole body) by inhalation to racemic
phenothrin at concentrations of 0, 43, or 220 mg/m3 (4 h per day, 5
days per week) for 4 weeks. After this treatment, animals (10/sex
per group) were sacrificed and the remaining rats were kept for 3
weeks without treatment. There were no adverse toxicological
effects on the animals exposed to the highest dose. Under the same
conditions (i.e. 220 mg/m3 for 4 weeks), racemic phenothrin did not
produce any adverse effects in male or female ICR mice. Similarly,
d-phenothrin also produced no adverse toxicological effects in
Sprague Dawley rats or ICR mice (both male and female) following
whole body exposure at 210 mg/m3 (particle size 5 µm) for 4 weeks
(Kohda et al., 1979b).
The acute oral and intraperitoneal toxicity of phenothrin
metabolites in rats and mice are shown in Table 6 and 7 (Kohda et
al., 1979a; FAO/WHO, 1981).
Table 6. The acute oral toxicity of phenothrin
metabolites
-----------------------------------------------------
Chemical No.a Species LD50
(mg/kg)
-----------------------------------------------------
3-Phenoxybenzyl alcohol 12 Rat 1330
3-Phenoxybenzaldehyde 13 Rat 600
-----------------------------------------------------
a Chemical identification number used in Fig. 2
Table 7. The acute intraperitoneal toxicity of several phenothrin
metabolites in mice
----------------------------------------------------------------------
Chemical No.a LD50 (mg/kg)
Male Female
----------------------------------------------------------------------
3-Phenoxybenzyl alcohol 12 371 424
3-(4'-hydroxyphenoxy)benzyl alcohol 14 750-1000 750-1000
3-(2'-hydroxyphenoxy)benzyl alcohol 15 876 778
3-Phenoxybenzoic acid 16 154 169
3-(4'-hydroxyphenoxy)benzoic acid 17 783 745
3-(2'-hydroxyphenoxy)benzoic acid 18 859 912
3-Phenoxybenzaldehyde 13 415 416
----------------------------------------------------------------------
a Chemical identification number used in Fig. 2
7.2. Long-Term Exposures and Carcinogenicity
Appraisal
There have been several long-term feeding studies with racemic
phenothrin or d-phenothrin (dose levels ranging from 200 to 10 000
mg/kg diet) in rats and mice, the length of exposure ranging from 6
months to 2 years. A slight increase in liver weight and a
significant difference in some clinical/chemistry parameters from those
of controls were observed at high doses. However, the no-observed-
effect level (NOEL) values obtained in these studies were high (300-
1000 mg/kg diet, corresponding to around 40-160 mg/kg body weight) and
no tumorigenicity was observed. Two feeding studies with d-phenothrin
(doses of 100-3000 mg/kg diet with exposure period 26 and 52 weeks) in
dogs revealed similar results, the NOEL being 300 mg/kg diet
(corresponding to around 7-8 mg/kg body weight per day). No
tumorigenicity relating to phenothrin feeding was detected in these
studies.
7.2.1. Rat
In a study by Murakami et al. (1981), [1R, cis]-phenothrin
(d-phenothrin) was administered to Sprague Dawley rats (20 of each
sex per group) at dose levels of 0, 1, 3, or 10 g/kg diet for 6
months. Ten rats of each sex per group were sacrificed after 3
months. d-Phenothrin had no significant effect on mortality,
clinical signs, ophthalmology, urinalysis, or gross and
histopathological findings. The serum albumin level was elevated
after 3 months in males fed 10 g/kg and in females fed 3 or 10
g/kg, and after 6 months in males fed 3 or 10 g/kg. The albumin-
globulin ratio was raised after 3 months in males fed 3 or 10 g/kg
and in females fed 10 g/kg, and in both males and females fed 10
g/kg after 6 months. Absolute and relative liver weights in both
males and females fed 3 or 10 g/kg were increased. Based on these
data, it was concluded that the NOEL for d-phenothrin in this study
was 1 g/kg diet for both sexes (55.4 mg/kg body weight per day for
males and 63.3 mg/kg body weight per day for females).
In a standard oncogenicity study, Fisher-344 rats (50 of each
sex per group) were fed d-phenothrin at dose levels of 0, 300, 1000
or 3000 mg/kg diet for at least 105 weeks in males and at least 118
weeks in females. Additional rats (30 of each sex per group) were
assigned to a chronic toxicity study with a 52-week interim
sacrifice. There were no significant effects on clinical signs,
mortality, food and water consumption, ophthalmology, blood
biochemistry, haematology, or urinalysis. However, the body weight
gain in females fed d-phenothrin at 3000 mg/kg was reduced, and the
relative liver weight was increased in females fed 3000 mg/kg for
52 weeks and, at the end of the oncogenicity study, in males fed
3000 mg/kg. Microscopic examination revealed that the incidence of
cystic dilatation of the sinuses of the mesenteric lymph nodes and
of periacinar hepatocytic hypertrophy was higher in males fed 3000
mg/kg for at least 105 weeks. d-Phenothrin did not show any
oncogenic activity to rats at up to 3000 mg/kg. Although at this
dose an increase in the incidence of adenomas and carcinomas of the
preputial gland was seen in males, the 1988 Joint FAO/WHO Meeting
on Pesticide Residues (FAO/WHO, 1988b) considered it unlikely that
this finding was of toxicological significance. The NOEL was 1000
mg/kg diet for both sexes (47 mg/kg body weight per day for males
and 56 mg/kg body weight per day for females) (Martin et al.,
1987).
When Sprague Dawley rats (50 of each sex per group) were fed a
diet containing racemic phenothrin (0, 200, 600, 2000, and 6000
mg/kg diet) for 2 years, body weight and food consumption were
slightly depressed at 6000 mg/kg in both males and females.
Ophthalmological examinations, haematological studies, urinalyses,
and clinical chemistry studies were performed at various intervals.
At the end of the study all animals were sacrificed and examined
for gross abnormalities, and extensive microscopic examinations
were conducted on a variety of tissues and organs. There were no
abnormal clinical or behavioral problems associated with
phenothrin administration. The survival rate of all groups of
treated rats was similar to that of controls. Males fed 6000 mg/kg
showed a significant increase in serum glutamine-pyruvate
aminotransferase activity. Ophthalmological examinations revealed
some abnormalities, all of which appeared to be age related.
Histopathological examination revealed no significant differences
between the treated groups and the control group with respect to
severity of lesions. No histopathological changes suggestive of
oncogenicity resulting from phenothrin treatment were found
(Hiromori et al., 1980).
7.2.2. Mouse
When Swiss White mice (50 of each sex per group) were fed
racemic phenothrin for 18 months at dose levels of 0, 300, 1000, or
3000 mg/kg diet, there were no significant effects on mortality,
clinical signs, haematologic values, clinical chemistry parameters,
or gross pathological findings. Slight body weight depression
occurred in males fed 3000 mg/kg, and increased liver weight was
found at the highest dose level in both males and females. There
was a statistically significant difference (compared with the
controls) in lung amyloidosis in the 1000 and 3000 mg/kg dose
groups, but no significant increase in tumours attributable to
phenothrin ingestion (Murakami et al., 1980).
In studies by Amyes et al. (1987), B6C3F1 hybrid mice (90 of
each sex per group) were fed d-phenothrin in the diet at dose
levels of 0, 300, 1000, or 3000 mg/kg. Fifty of each sex per group
were allocated to a standard oncogenicity study lasting 104 weeks.
The remaining mice were assigned to a chronic toxicity study, where
10 of each sex per group were sacrificed for interim study after 26
or 53 weeks and the remaining animals were examined after 78 weeks
of treatment. There were no compound-related effects on clinical
signs, mortality, ophthalmology, blood biochemistry, haematology,
or urinalysis. However, body weight gains for males fed d-
phenothrin at 3000 mg/kg were reduced and relative liver weights
were increased in both sexes fed 3000 mg/kg and in males receiving
1000 mg/kg. Microscopic examination revealed that the incidence of
periacinar hepatocyte hypertrophy with cytoplasmic eosinophilia was
higher in males fed 3000 mg/kg. The incidence of liver tumors
appeared higher in phenothrin-treated female mice than in control
females, but the difference was not statistically significant. It
was concluded, therefore, that administration of d-phenothrin to
mice for 2 years at dietary levels of up to 3000 mg/kg diet did not
significantly disturb the tumour burden or tumour profile of B6C3F1
hybrid mice. The NOEL in this study was 300 mg/kg diet for males
(40 mg/kg body weight per day) and 1000 mg/kg diet for females (164
mg/kg body weight per day).
7.2.3. Dog
When beagle dogs (six of each sex per group) were fed d-
phenothrin at dose levels of 0, 100, 300, or 1000 mg/kg diet for 26
weeks, there were no compound-related abnormal findings in
mortality, clinical signs, body weight, food consumption,
ophthalmology, gross or microscopic pathology, haematology, or
urinalysis studies. However, the alkaline phosphatase activity in
males fed 300 mg/kg and males and females fed 1000 mg/kg was
elevated and a slight increase in the mean relative liver weight in
males fed 1000 mg/kg was noted. The NOEL in this study was 300
mg/kg (Pence et al., 1981).
In a study by Cox et al. (1987), beagle dogs (four of each sex
per group) were fed d-phenothrin at dose levels of 0, 100, 300,
1000, or 3000 mg/kg diet for 52 weeks. There were no significant
effects on clinical signs, body weight, food consumption,
ophthalmology, or urinalysis. However, decreases in erythrocyte
count, haemoglobin concentration, haematocrit, and total blood
protein were noted in both male and female dogs fed 3000 mg/kg,
whereas mean absolute and relative liver weights increased.
Compound-related histopathological alterations were noted in the
adrenal glands and liver. Focal degeneration of the adrenal cortex
with cytoplasmic deposition of crystalline material was seen in one
male dog fed 1000 mg/kg and four dogs fed 3000 mg/kg. The chemical
nature or biological significance of this crystalline material was
not recorded. Hepatocytes appeared to enlargen slightly in one
male dog fed 1000 mg/kg and seven dogs fed 3000 mg/kg. The NOEL in
this study was 300 mg/kg diet for males and 1000 mg/kg for females
(8.24 and 26.77 mg/kg body weight per day for males and females,
respectively).
7.3. Mutagenicity
The results obtained in vivo and in vitro test system indicate
that d-phenothrin does not exhibit any mutagenic properties or
cause chromosomal or DNA damage.
In a DNA-repair test with Bacillus subtilis (M45 rec- and H17
wild type strains) using dose levels of up to 5 mg/disk per plate,
d-phenothrin did not inhibit the growth of any strain at any dose
level, whereas the positive control, mitomycin C, showed a clear
effect. The negative control gave a result similar to that of d-
phenothrin (Kishida & Suzuki, 1981a).
A mutagenicity test with Escherichia coli (WP2 uvr) and
Salmonella typhimurium (TA 1535, TA 1537, TA 1538, TA 98, and TA
100) using d-phenothrin at dose levels of up to 5 mg/plate with and
without metabolizing enzyme system (S9 mix) yielded negative
results, whereas a positive control gave a significant number of
mutants (Kishida & Suzuki, 1981b).
In a host-mediated assay using S. typhimurium G46 (indicator
bacteria), d-phenothrin in corn oil was given orally (twice with a
24-h interval) to groups of six male ICR mice at dose levels of
2500 or 5000 mg/kg body weight. Soon after the last administration,
each mouse was injected intraperitoneally with the indicator cells.
Three hours later, the bacterial mutation frequency in d-
phenothrin-treated mice was no greater than that in the control
group (Kishida & Suzuki, 1981c).
Suzuki et al. (1981) examined d-phenothrin for its ability to
induce chromosomal aberrations in vivo using bone marrow cells.
ICR mice were treated intrapertioneally with single doses of 2500,
5000, or 10 000 mg/kg body weight and sacrificed 6, 24, or 48 h
after treatment. No chromosomal aberrations as a result of d-
phenothrin treatment were detected.
In an in vitro chromosomal aberration test, Chinese hamster
ovary cells (CHO-K1) were treated with d-phenothrin (dose levels:
2 x 10-5 to 2 x 10-4 mol/litre for 24 and 48 h in the absence of S9
mix; 5 x 10-5 to 5 x 10-4 mol/litre for 6 h in the presence of S9
mix). No significant increase in the number of cells with
chromosomal aberrations was observed (Kogiso et al., 1986).
The ability of d-phenothrin to induce sister-chromatid
exchanges (SCEs) were tested in cultured mouse embryonic cells in
vitro. At doses of 10-5, 10-4, and 10-3 mol/litre (with and
without S9 mix), d-phenothrin did not induce any increase in the
frequency of SCEs (Suzuki & Miyamoto, 1981).
In a study of unscheduled DNA synthesis, Hela S3 cells were
treated with d-phenothrin at dose levels of 0, 0.25, 0.5, 1.0, 2.0,
or 4.0 mg/ml in the presence of 3H-thymidine (with and without S9
mix) for 3 h, and the incorporation of 3H-thymidine into DNA was
measured. There was no significant increase in the radioactivity
of DNA from cells treated with d-phenothrin (Foster et al., 1984).
7.4. Reproduction, Embryotoxicity, and Teratogenicity
No teratogenicity or reproductive effects were observed when
phenothrin was fed to rabbits or mice during the major
organogenesis period of gestation or to rats in 3-generation
reproduction studies.
7.4.1. Embryotoxicity and teratogenicity
In studies by Ladd et al. (1976), pregnant New Zealand White
rabbits (17 per group) were administered racemic phenothrin orally
at dose levels of 0, 3, 10, or 30 mg/kg body weight on days 6 to 18
of gestation. They were sacrificed on day 29 and the young
obtained by caesarian section were examined. At 30 mg/kg, the body
weight of females decreased during gestation, and there was a
slight decrease in the number of live young and a slight reduction
in fetal weight. Racemic phenothrin had no apparent teratogenic
effect, as shown by a lack of gross internal or external somatic
abnormalities and by normal fetal skeletal development following
prenatal exposure.
Pregnant New Zealand White rabbits (15 per group) were orally
administered d-phenothrin by intubation (0, 10, 100, or 1000 mg/kg
body weight per day) on days 6 to 18 of gestation, and were
sacrificed on day 29 or 30. Following caesarian section, 50% of
the pups were maintained for 24 h to evaluate survival. No
abnormalities were observed among the does (body weight, food
consumption, clinical observations, and necropsy) or foetuses
(implantation sites, corpora lutea, resorption sites, weight,
condition, and viability). Data on foetal survival and from
internal and external examinations for abnormalities showed not
significant effects from administrating d-phenothrin during
gestation (Rutter, 1974).
In studies by Nakamoto et al. (1973), d-phenothrin was orally
administered to pregnant ICR mice (17 or 18 per group) at dose
levels of 0, 30, 300, or 3000 mg/kg body weight on days 7 to 12 of
gestation (not covering the whole period of organogenesis). The
dams were sacrificed on day 18 of gestation and the pups were
obtained by caesarian section. Other mice (7 per group) were given
d-phenothrin at dose levels of 0, 300, or 3000 mg/kg to evaluate
postnatal effect. These mice were allowed to deliver naturally and
the pups were kept for 29 days. At these levels, d-phenothrin
showed no adverse effects, as indicated by maternal growth, fetal
mortality and external and internal examination of fetuses for
teratogenic or embryotoxic effects.
7.4.2. Reproduction studies
In a standard 3-generation (2 litters per generation)
reproduction study, groups of rats (8 male and 16 female Charles
River albino rats per group) were fed racemic phenothrin at dose
levels of 0, 200, 600, or 2000 mg/kg diet. Various reproduction
indices (i.e. mating index, fecundity index, male fertility index,
female fertility index, and incidence of parturition) were
measured. The adult rats showed no significant mortality or
complications during the study, and the reproductive parameters
revealed no significant dose-related adverse effects attributable
to phenothrin. Gross and microscopic findings indicated no adverse
effect resulting from dietary phenothrin. It was concluded that
phenothrin had no effect on reproduction (Takatsuka et al., 1980).
In a study by Tesh et al. (1978), d-phenothrin was fed to
Charles River CD rats (30 of each sex per group) at dose levels of
0, 300, 1000, or 3000 mg/kg diet throughout two successive
generations and up to the maturation of a third generation. At 300
and 1000 mg/kg, there was no adverse effect upon mortality, somatic
growth, development, or reproductive performance. At 3000 mg/kg,
mortality, body weight, and reproductive performance showed no
significant response to treatment, and selected F2 animals reared
to maturity were in all respects comparable with control rats.
However, F0 and F1 females and selected F2B male and female
weanlings showed a slight but consistent increase in the relative
liver weight. The NOEL in this study was 1000 mg/kg diet.
7.5. Neurotoxicity
Sprague Dawley rats exposed to d-phenothrin by inhalation at
concentratons of up to 3760 mg/m3 for 4 h showed no toxic signs as
a result of exposure. Histopathologically, there were no compound
related alterations in the sciatic nerve (Kohda et al., 1977).
When d-phenothrin was given to Sprague Dawley rats orally for 5
consecutive days (5 g/kg body weight per day), one out of ten
female rats died after four doses and signs of poisoning
(piloerections and urinary incontinence) were noted in several of
the animals. However, these signs disappeared rapidly at the end
of the treatment and there were no other signs of poisoning such as
leg weakness or ataxia. All animals were sacrificed 3 days after
the final dose, and histopathologically examination of the sciatic
nerve revealed minute changes in axon and myelin, characterized by
very slight axonal swelling, axonal disintegration, and/or
demyelination. Since there were similar changes in the control
animals, it was suggested that they were not due to the d-
phenothrin. It was considered that the oral administration of very
high doses of d-phenothrin does not lead to the neurotoxic effects
observed with several other pyrethroid esters (Okuno et al., 1978).
7.6. Miscellaneous Effects
No d-phenothrin-attributable pharmacological effects were
detected in various tests (e.g., spontaneous movement of isolated
guinea-pig ileum, contraction of the rat phrenic nerve diaphragm
preparation, cardiopulmonary physiology of anaesthetized cats,
coordination, and spontaneous movement of mice, and rectal
temperature of rats) at doses of 100-300 µg/ml in vitro, 3 mg/kg
intravenous, or 100-300 mg/kg intraperitoneal. A tentative arousal
response was recorded in the electroencephalogram of cats given d-
phenothrin (4 mg/kg) intraperitoneally, as is commonly observed in
animals given synthetic pyrethroids (Hara et al., 1974).
7.7. Mechanisms of Toxicity - Mode of Action
Some synthetic pyrethroids given intravenously to rats cause
either tremor (T-syndrome) or choreoathetosis with salivation (CS-
syndrome) (Verschoyl & Aldridge, 1980). However, d-phenothrin
(>600 mg/kg body weight) injected intravenously into the lateral
tail vein caused neither T-syndrome nor CS-syndrome, due to its
very low acute toxicity. From a study involving intracerebral
dosing with [1R, cis]- or [1R, trans]-phenothrin in mice, both
compounds were classified as Type I pyrethroids based on the
occurrence of tremors (Lawrence & Casida, 1982) and on
neurophysiological studies in cockroach cercal sensory nerves
(Gammon et al., 1981).
8. EFFECTS ON HUMANS
Although d-phenothrin has been used for more than 10 years, no
toxic effects and no cases of poisoning have been reported.
8.1. Clinical Studies
In a study by Hashimoto et al. (1980), d-phenothrin (talc
powder formulation with Span 80 as a stabilizer) was applied to the
head hair and pudenda hair of eight male human volunteers (three
times at intervals of 3 days) at a dose of 32 mg/man per
administration (0.44 to 0.67 mg/kg body weight per day). d-
Phenothrin powder was washed off 1 h after application. There were
no significant abnormalities due to d-phenothrin in terms of dermal
irritation, clinical signs, or blood biochemical and haematological
parameters. The blood levels of d-phenothrin were below the
detection limit of 0.006 mg/kg.
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has
discussed and evaluated d-phenothrin at its meetings in 1979, 1980,
1984, and 1988 (FAO/WHO, 1980, 1981, 1985a, 1988b).
Since 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg
body weight has been established.
In the WHO Recommended Classification of Pesticides by Hazard,
technical phenothrin is classified as unlikely to present an acute
hazard in normal use (WHO, 1988).
REFERENCES
AMYES, S.J., MARTIN, P.A., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Oncogenicity and toxicity study
in mice, Suffolk, United Kingdom, Life Science Research (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of syn-
thetic pyrethroids in fruit and vegetables by gas-liquid and high-
performance liquid chromatography. Analyst, 107: 206-212.
COX, R.H., SUTHERLAND, J.D., VOELKER, R.W., ALSAKER, R.D.,
VARGAS, K.J., LEWIS, S.A., & HAGEN, W.J. (1987) Chronic toxicity study
in dogs with Sumithrin technical grade, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
DESMARCHELIER, J.M. (1980) Comparative study of analytical methods for
bioresmethrin, phenothrin, d-phenothrin, pyrethrum I, carbaryl,
fenitrothion, methacrifos, pirimifos-methyl and dichlorfos on various
grains. J. Pestic. Sci., 5: 521-532.
DOI, T., SAKAUE, S., & HORIBA, M. (1985) Analysis of d-phenothrin and
its optical isomers in technical preparations and formulations. J.
Assoc. Anal. Chem., 68(5): 911-916.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC, American
Chemical Society, p. 229 (ACS Symposium Series No. 42).
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to a
synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
FAO (1982) Second Government Consultation on International Harmoniz-
ation of Pesticide Registration Requirements, Rome, 11-15 October,
1982, Food and Agriculture Organization of the United Nations.
FAO/WHO (1980) Pesticide residues in food: 1979 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 427-452
(FAO Plant Production and Protection Paper 20 Sup.).
FAO/WHO (1981) Pesticide residues in food: 1980 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 394-403
(FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1985a) Pesticide residues in food: 1984 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 689-694
(FAO Plant Production and Protection Paper 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues, 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1988a) Pesticide residues in food: 1987 Evaluations. Part 1
- Residues, Rome, Food and Agriculture Organization of the United
Nations, pp. 109-113 (FAO Plant Production and Protection Paper 86.1).
FAO/WHO (1988b) Pesticide residues in food. Report of the 1988 Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues, Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper 92).
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous sensation
between synthetic pyrethroid insecticides. Contact Dermatitis, 13(3):
140-147.
FOSTER, R., TIPINS, R.S., DE VENEZIA, V., & NUNZIATA, A. (1984)
Unscheduled DNA synthesis in human cells, cell line: Hela S3, test
substance: Sumithrin, Suffolk, United Kingdom, Life Science Research,
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
FUJIMOTO, K., ITAYA, N., OKUNO, Y., KADOTA, T., & YAMAGUCHI,T. (1973)
A new insecticidal pyrethroid ester. Agric. biol. Chem., 37:2681-2682.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent class
antagonize GABA action at the crayfish neuromuscular junction.
Neurosci. Lett., 40:163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid toxi-
cology: Protective effects of diazepam and phenobarbital in the mouse
and the cockroach. Toxicol. appl. Pharmacol., 66: 290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural variations
affecting pyrethroid neurotoxicity. Neurobehav. Toxicol. Teratol., 4(6):
793-799.
HARA, Y., MIYAGISHI, A., OHTSUKA, M., ASAMI, Y., KUROKAWA, Y.,
& MIYAMOTO, J. (1974) Pharmacological study of Sumithrin: Effects on
respiration, blood pressure, and peripheral central nervous systems
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HASHIMOTO, S., OKANO, S., YAMADA, H., & MIYAMOTO, J. (1980)
Human volunteer study with phenothrin and d-phenothrin
powder (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
HAYASHI, A. (1977) [Safe use of insecticides (I)] [Bo-satsu-kyo News,]
No. 1 p. 10 (June) (in Japanese).
HIROMORI, T., KOYAMA, Y., OKUNO, Y., ARAI, M., ITO, N., &
MIYAMOTO, J. (1980) Two-year chronic toxicity study of S-2539 in
rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HIROMORI, T., HARA, S., KADOTA, T., & MIYAMOTO, J. (1984) Acute
intravenous toxicity of phenothrin and d-phenothrin in mice
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
ISOBE, N., MATSUNAGA, H., NAKATSUKA, I., & YOSITAKE, A.
(1987) Metabolism of [1R, trans]- and [1R, cis]- isomers of phenothrin
in rats (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
ITAYA, N., KAMOSHITA, K., KITAMURA, S., OKUNO, Y., MIZUTANI,
T., NAKAI, S., KAMEDA, N., & FUJIMOTO, K. (1969) Esters of cyclo-
propanecarboxylic acids. Ger. Offen., 1: 926, 433.
IZUMI, T., KANEKO, H., MATSUO, M., & MIYAMOTO, J. (1984)
Comparative metabolism of the six optical stereo isomers of phenothrin
in rats and mice. J. Pestic. Sci., 9: 259-267.
KANEKO, H., OHAKAWA, H., & MIYAMOTO, J. (1981) Absorption and
metabolism of dermally applied phenothrin in rats. J. Pestic. Sci., 6:
169-182.
KISHIDA, F. & SUZUKI, H. (1981a) Studies on DNA-damaging capacity of
Sumithrin with Bacillus subtilis (Unpublished Technical Report No. ET-
10-0069 submitted to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA. F. & SUZUKI, H. (1981b) Gene mutation test of Sumithrin in
bacterial system (Unpublished Technical Report No. ET-10-0068 submitted
to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA, F. & SUZUKI, H. (1981c) Mutagenicity test of Sumithrin in
host-mediated assay (Unpublished Technical Report No. ET-10-0067 sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
KOGISO, S., HARA, M., ITO, K., IWAWAKI, H., & YOSHITAKE, A.
(1986) In vitro chromosomal aberration test of S-2539F in Chinese
hamster ovary cells (CHO-K1) (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1977) Acute
inhalation toxicity of d-phenothrin (S-2539 Forte) in rats (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
KOHDA, H., KENEDO, H., OHKAWA, H., KADOTA, T., & MIYAMOTO, J.
(1979a) Acute intraperitoneal toxicity of permethrin metabolites in
mice (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
KOHDA, H., NISHIMOTO, K., KADOTA, K., & MIYAMOTO, J. (1979b)
Acute and subacute inhalation toxicity studies of S-2539 Forte in rats
and mice (Unpublished report submitted to WHO by Sumitomo Chemical
Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1980) Particle
size distribution and mean particle size in acute inhalation toxicity
study of d-phenothrin (S-2539 forte) in rats (Unpublished Technical
Report No. ET-00-0054 submitted to WHO by Sumitomo Chemial Co.,
Ltd).
LADD, R., SMITH, P.S., JENKINS, D.H., KENNEDY, G.L., Jr, KINOSHITA,
F.K., & KEPLINGER, M.L. (1976) Teratogenic study with S-2539 in albino
rabbits, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: mouse
intracerebral structure-toxicity relationship. Pestic. Biochem. Physiol.,
18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions of
pyrethroid insecticides with chloride ionophore-associated binding
sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticides, London, Taylor &
Francis Ltd, p. 440.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel modifi-
cation as the basis for the variation in the nerve membrane effects of
pyrethroids and DDT analogs. Pestic. Biochem. Physiol., 20: 203-216.
MARTIN, P.A., AMYES, S.J., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Combined toxicity and
oncogenicity study in rats, Suffolk, United Kingdom, Life Science
Research (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of synthetic
pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue analysis of
synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry-human welfare
and the environment. Proceedings of the Fifth International Congress of
Pesticide Chemistry, Kyoto, Japan, 29 August to 4 September, 1982, Oxford,
Pergamon Press, Vol. 1-4.
MIYAMOTO, J., SUZUKI, T., & NAKAE, C. (1974) Metabolism of phenothrin
or 3-phenoxybenzyl d-trans-chrysanthemate in mammals. Pestic. Biochem.
Physiol., 4: 438-450.
MIYAMOTO, J., KOGISO, S., & MATSUO, M. (1984) Additional toxicological
information for phenothrin (Unpublished Technical Report No. ET-40-0090
from Sumitomo Chemical Co., Ltd).
MULLA, M.S., DARWAZEH, H.A., & DHILLON, M.S. (1980) New
pyrethroids as mosquito larvicides and their effects on nontarget
organisms. Mosq. News, 40: 6-12.
MURAKAMI, M., HIROMORI, T., ITO, S., & HOSOKAWA, S. (1981)
Six month oral toxicity study of S2539 Forte (Sumithrin(R)) in rats
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURAKAMI, J., ITO, S., OKUNO, Y., ARAI, M., ITO, N., & MIYAMOTO, J.
(1980) Eighteen-month chronic oral toxicity and tumorigenicity study of
S-2539 in mice, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURANO, A. (1972) Determination of the optical isomers of insecticidal
pyrethroids by gas-liquid chromatography. Agric. biol. Chem., 36:
2203-2211.
NAKAMOTO, N., KATO, T., & MIYAMOTO, J. (1973) Teratogenicity study
of S-2539 Forte in mice (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
NAMBU, K., OHKAWA, H., & MIYAMOTO, J. (1980) Metabolic fate of
phenothrin in plants and soils. J. Pestic. Sci., 5: 177-197.
NAMBU, K., TAKIMOTO, Y., & MIYAMOTO, J. (1981) Degradation of
phenothrin in stored wheat grains. J. Pestic. Sci., 6: 183-191.
NIOSH (1983) Registry of toxic effects of chemical substances, 1981-
1982 edition, Cincinnati, Ohio, US National Institute for Occupational
Safety and Health, p. 1003.
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
OHNISHI, J., IKEDA, M., SUZUKI, Y., & MATSUDA, T. (1987) Residue
analysis of d-phenothrin, fenitrothion and piperonyl butoxide in stored
wheat, wheat milling fractions and bread (interim report) (Unpublished
Report No. ER-RD-8719 submitted to WHO by Sumitomo Chemical Co.,
Ltd).
OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1978) Neurotoxicity study
of d-phenothrin (S-2539 Forte(R)) in rats by repeated oral administration
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
PAPADOPOULOU-MOURKIDOU, E., IWATA, Y., & GUNTHER, F.A.
(1981) Utilization of an infrared detector for selective liquid
chromatographic analysis. 2. Formulation analysis of the pyrethroid
insecticides allethrin, decamethrin, cypermethrin, phenothrin and
tetramethrin. J. agric. food Chem., 29: 1105-1111.
PENCE, D.H., HAGEN, W.H., WLSAKER, R.D., HASTINGS, T.F.,
DAWKINS, B.G., TACEY, R.L., & MARSHALL, P.M. (1981) Subchronic
toxicity study of S2539-F in dogs, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids on a
nerve-muscle preparation of the clawed frog, Xenopus laevis. Pestic.
Biochem. Physiol., 25: 176-187.
RUTTER, H.A. (1974) Teratogenicity study in rabbits: S-2539 Forte,
Vienna, Virginia, Hazleton Laboratories Inc. (Unpublished report sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
RUZO, L.O., SMITH, I.H., & CASIDA, J.E. (1982) Pyrethroid
photochemistry: photooxidation reactions of the chrysanthemates,
phenothrin and tetramethrin. J. agric. food Chem., 30: 110-115.
SAKAUE, S., KITAJIMA, M., HORIBA, M., & YAMAMOTO, S. (1981) Gas
chromatographic and high performance liquid chromatographic
determination of d-phenothrin (Sumithrin) in formulations. Agric. biol.
Chem., 45: 1135-1140.
SEGAWA, T. (1976) Acute toxicity study of S-2539 in rats and mice,
Hiroshima, Hiroshima University School of Medicine (Unpublished report
submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979a) Acute oral, subcutaneous, dermal and intraperitoneal
toxicity studies of S-2539 Forte in rats, Hiroshima, Hiroshima
University School of Medicine (Unpublished Technical Report
No. ET-31-0033 submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979b) Acute oral, subcutaneous, dermal and
intraperitoneal toxicity studies of S-2539 Forte in mice, Hiroshima,
Hiroshima University School of Medicine (Unpublished Technical Report
No. ET-31-0032 submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H. & MIYAMOTO, J. (1981) In vitro sister chromatid
exchanges of Sumithrin (Unpublished Technical Report No. ET-10-0071
submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H., HARA, M., & MIYAMOTO, J. (1981) In vivo
chromosomal aberration test of Sumithrin on bone marrow cells of
mice (Unpublished Technical Report No. ET-10-0072 submitted to WHO by
Sumitomo Chemical Co., Ltd).
SUZUKI, T. & MIYAMOTO, J. (1978) Purification and properties of
pyrethroid carboxyesterase in rat liver microsome. Pestic. Biochem.
Physiol., 8: 186-198.
SUZUKI, T., OHNO, N., & MIYAMOTO, J. (1976) New metabolites of
(+)-cis-phenothrin, 3-phenoxybenzyl (+)-cis chrysanthemate, in rats.
J. Pestic. Sci., 1: 151-152.
TAKATSUKA, M., OKUNO, Y., SUZUKI, T., KADOTA, T., YASUDA, M.,
& MIYAMOTO, J. (1980) Three-generation reproduction study of S-2539
in rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
TAKIMOTO, Y., KATO, T., & MIYAMOTO, J. (1977) Residue analyses
of phenothrin (S-2539) in crops (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
TESH, J.M., WILLOUGHBY, C.R., & FOWLER, J.S.L. (1987) Sumithrin:
Effects upon reproductive performance of rats treated continuously
throughout two successive generations, Suffolk, United Kingdom, Life
Science Research (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
TURNBULL, S.R. & ARDLEY, J.H. (1987) Determination of d-phenothrin,
piperonyl butoxide and fenitrothion residues on wheat, derived
fractions and breads, at approximately one, three and six months post-
treatment, Australia, The Wellcome Foundation Ltd (Unpublished Report
No. AESH 87-11).
VAN DEN BERCKEN, J. (1977) The action of allethrin on the peripheral
nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels in
frog myelinated nerve membrane. In: Insect: Neurobiology and pesticide
action, London, Society of Chemical Industry, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER
ZALM, J.M. (1973) DDT-like action of allethrin in the sensory nervous
system of Xenopus laevis. Eur. J. Phamacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A.
(1979) Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VERSCHOYLE, R.D. & ALDRIDGE, W.N.(1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45: 325-
329.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Phamacol., 58: 501-504.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system. Neuropathol.
appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J.
(1982a) Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed frog,
Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN
BERCKEN, J. (1982b) Similar mode of action of pyrethroids and DDT on
sodium channel gating in mylinated nerves. Nature (Lond.), 295: 601-
603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF,
R.G.D.M., & VAN DEN BERCKEN, J. (1983) Temperature and structure-
dependent interaction of pyrethroids with the sodium channels in frog
node of Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WHO (1979) WHO Technical Report Series, No. 634 (Safe use of
pesticides. Third Report of the WHO Expert Committee on Vector Biology
and Control), pp 18-23.
WHO (1988) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1988-1989, WHO, World Health
Organization (WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1987) d-Phenothrin. In: The pesticide
manual, 8th ed., Croydon, British Crop Protection Council, pp. 654-
655.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
APPENDIX
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs ( Xenopus laevis; Rana temporaria, and
Rana Escuelenta) it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these two classes of pyrethroids has been made
on the basis of the symptoms of toxicity in mammals and insects
(Van den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge,
1980; Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group (allethrin, d-
phenothrin, permethrin, tetramethrin, cismethrin, and bioresmethrin)
(Type I: T-syndrome).
The pyrethroids that do not contain an alpha-cyano group give
rise to pronouced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3-10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organs and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5°C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of the "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 milliseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently, it was demonstrated that allethrin and DDT have
essentially the same effect on sodium channels in frog myelinated
nerve membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittently, cause large
depolarizing after-potentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl alcohol
(deltamethrin, cypermethrin, fenvalerate, and fenpropanate) (Type II:
CS-syndrome).
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependant depression of the
nervous impulse, brought about by a progressive depolarization
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, persumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
The results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
crayfish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered
pyrethroids closely approximates that of the convulsant picrotoxin
(PTX). Deltamethrin inhibits the binding of the [3H]-
dihydropicrotoxin to rat brain synaptic membranes, whereas the non-
toxic R epimer of deltamethrin is inactive. These findings suggest
a possible relation between the Type II pyrethroid action and the
GABA receptor complex. The stereospecific correlation between the
toxicity of Type II pyrethroids and their potency to inhibit the
[35S]-TBPS binding was established using a radioligand, [35S]= t-
butylbicyclophosphorothionate [35S]-TBPS. Studies with 37
pyrethroids revealed an absolute correlation, without any false
positive or negative, between mouse intracerebral toxicity and in
vitro inhibition: all toxic cyano compounds including deltamethrin,
1R, cis-cypermethrin, 1R, trans-cypermethrin, and [2S,alpha]-
fenvalerate were inhibitors, but their non-toxic stereoisomers were
not; non-cyano pyrethroids were much less potent or were inactive
(Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S,alphaS]-fenvalerate) increased the input resistance of crayfish
claw opener muscle fibres bathed in GABA. In contrast, two non-
insecticidal stereoisomers and Type I pyrethroids (permethrin,
resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis ). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action. It
may be related to the long-lasting repetitive activity in sense
organs and possibly in other parts of the nervous system, which, in
a more advance state of poisoning, may be accompanied by a
frequency-dependent depressoin of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vunerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
RESUME, EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1. Résumé et évaluation
1.1 Identité, propriétés physiques et chimiques, et méthodes
d'analyse
La phénothrine racémique a été synthétisée pour la première
fois en 1969. Du point de vue chimique, il s'agit de l'ester de
l'acide diméthyl-2,2 (diméthyl-2,2 vinyl)-3 cyclopropanecarboxylique
et de l'alcool phénoxy-3 benzylique (PBalc). Il se présente sous
la forme d'un mélange de quatre stéréoisomères : [1R,trans],
[1R,cis], [1S,trans], [1S,cis]. La d-phénothrine est un mélange
d'une partie d'isomère [1R,cis] pour quatre parties d'isomère
[1R,trans], et elle est à l'heure actuelle le seul produit
technique sur le marché. C'est l'isomère [1R,trans] qui est
l'insecticide le plus actif; vient ensuite l'isomère [1R,cis].
La d-phénothrine de qualité technique se présente sous la forme
d'un liquide jaune pâle à brun jaune et son degré de pureté est de
92,5 à 94,5%. Sa densité est de 1,058-1,061 à 25°C et sa tension
de vapeur de 0,16 mPa à 20°C. Elle est difficilement soluble dans
l'eau (2 mg par litre à 25°C) mais soluble dans les solvants
organiques tels que l'acétone, le xylène et l'hexane. Elle est
assez stable à l'air mais instable à la lumière, encore que sa
photodégradation ne soit pas aussi rapide que celle des pyréthrines
naturelles. Elle est instable en milieu alcalin.
Le dosage des résidus peut s'effectuer par chromatographie en
phase liquide à haute performance avec détecteur ultra-violet, la
concentration minimale décelable étant de 0,05 mg par kg. Pour
l'analyse du produit technique on utilise la chromatographie en
phase gazeuse avec détection par ionisation de flamme.
1.2 Production et usage
La d-phénothrine est utilisée depuis 1977. On estime que l'on
utilise chaque année 70 à 80 tonnes de d-phénothrine dans le monde,
essentiellement pour détruire les insectes incommodants dans les
habitations, lutter contre les vecteurs de maladies et protéger les
céréales ensilées; le produit est utilisé seul ou en association
avec d'autres insecticides ou synergisants. Il est présenté sous
forme d'huiles pour aérosols, de poudres ou de concentrés
émulsionnables. La d-phénothrine est également utilisée pour
détruire les poux de l'homme, auquel cas elle est présentée sous
forme de poudre, de shampooing ou de lotion.
1.3 Exposition humaine
Les aérosols classiques à usage domestique ne devraient pas
conduire à des concentrations atmosphériques de d-phénothrine
supérieures à 0,5 mg/m3. Dans le blé ensilé, on peut trouver des
résidus allant jusqu'à 4 mg/kg, mais ces teneurs tombent à 0,8
mg/kg dans la farine après mouture, et à 0,6 mg/kg après
panification.
Pour détruire les poux, on applique la d-phénothrine sur la
chevelure, par exemple en trois doses de 32 mg tous les trois
jours. Il n'existe aucune donnée sur l'exposition professionnelle à
la d-phénothrine.
L'exposition de la population dans son ensemble devrait être
très faible, mais on manque de données précises sur ce point.
1.4 Destinée dans l'environnement
La phénothrine se dégrade facilement, sa demi-vie étant
inférieure à un jour sur les végétaux et autres surfaces. La d-
phénothrine ou ses produits de dégradation ne migrent que très peu
vers les zones non traitées des végétaux. On a constaté que des
haricots ne captaient que dans une faible mesure les prouits
radiomarqués provenant de sols traités par de la phénothrine
marquée au carbone-14. Après traitement des sols avec de la
[1R, trans ]- ou de la [1R, cis]-phénothrine à raison de 1 mg/kg, on
a constaté que les deux isomères se décomposaient rapidement, la
demi-vie initiale étant de un à deux jours, mais que, en cas
d'inondation, la dégradation était considérablement ralentie, la
demi-vie initiale étant de deux à quatre semaines pour l'isomère
trans et de un à deux mois pour l'isomère cis. On a observé que
les isomères trans ou cis de la phénothrine se déplaçaient
relativement peu (environ 2%) à travers des colonnes de terre,
lorsque le lessivage commençait immédiatement ou deux semaines
après le traitement.
En général, la dégradation qui se produit dans l'environnement
conduit à des produits moins toxiques.
1.5 Cinétique et métabolisme
Après avoir reçu une dose unique ou des doses répétées de
phénothrine radiomarquée par voie orale ou percutanée, des rats ont
rapidement et presque complètement excrété la fraction marquée dans
leurs urines et leurs déjections en trois à sept jours. Les
principales voies métaboliques des isomères cis et trans chez le
rat consistent en une rupture de la liaison ester et l'oxydation en
position 4 du reste alcool ou du groupement isobutényle du reste
acide. Les métabolites résultant du clivage de l'ester (qui sont
excrétées essentiellement dans les urines) constituent les
principaux produits de dégradation de l'isomère trans alors que les
métabolites restant sous forme d'ester (excrétées essentiellement
dans les déjections) proviennent pour la plupart de l'isomère cis.
1.6 Effets sur les êtres vivant dans leur milieu naturel
La phénothrine a été expérimentée sur quelques groupes
d'organismes non visés et dans chaque groupe sur quelques espèces
seulement. Chez les poissons, la CL50 à 96 heures de la
phénothrine racémique et des stéréoisomères (1R) va de 17 à 200
microgrammes par litre. Une étude, portant sur des invertébrés
aquatiques, a montré que chez Daphnia pulex la CL50 à 3 heures
était de 25 à 50 mg/litre pour tous les isomères et la phénothrine
racémique.
Une seule et unique étude au cours de laquelle de la
phénothrine a été appliquée à des étangs n'a révélé aucun effet sur
les arthropodes aquatiques.
La toxicité pour les oiseaux est faible, avec une DL50 aiguë
par voie orale supérieure à 2500 mg/kg de poids corporel chez le
colin de Virginie et une CL50 par voie alimentaire supérieure à
5000 mg/kg de nourriture chez ce volatile et chez le colvert.
Etant donné que la phénothrine se dégrade rapidement à la
lumière solaire et qu'on l'utilise principalement pour traiter des
céréales ensilées, l'exposition environnementale est
vraisemblablement très faible. Dans ces conditions, des effets sur
l'environnement sont tout-à-fait improbables.
1.7 Effets sur les animaux d'expérience et sur les systèmes
d'épreuve in vitro
La toxicité aiguë de la d-phénothrine est extrêmement faible,
la DL50 étant supérieure à 5000 mg/kg de poids corporel chez le rat
et la souris (par voie orale, sous-cutanée, dermique et
intrapéritonéale) et la CL50 inhalatoire supérieure à 3760 mg/m3
chez le rat. Le syndrome d'intoxication se caractérise par une
hyperexcitabilité, une prostration, des tremblements, de l'ataxie
et une paralysie. Sur la base de ces symptômes et d'après les
résultats des études électrophysiologiques sur les nerfs sensoriels
des cerques de la blatte, la phénothrine est classée parmi les
pyréthroides du type I.
Exposés à de la d-phénothrine par inhalation à des
concentrations allant jusqu'à 210 mg/m3, quatre heures par jour
pendant quatre semaines, ou par voie orale cinq jours de suite à
raison de 5000 mg/kg de poids corporel, des rats n'ont présenté
aucun effet toxicologique indésirable.
Plusieurs études d'alimentation ont été effectuées sur des rats
et des souris recevant de la d-phénothrine ou de la phénothrine
racémique à des doses allant de 200 à 10 000 mg par kg de
nourriture; la période d'exposition allait de six mois à deux ans.
Ces études ont permis d'établir une dose sans effet observable
allant de 300 à 1000 mg par kg de nourriture, ce qui correspond à
peu près à 40-160 mg/kg de poids corporel et par jour. Chez des
chiens à qui l'on avait administré de la d-phénothrine à des doses
de 100 à 3000 mg/kg de nourriture avec des périodes d'exposition de
26 à 52 semaines, deux études ont permis d'obtenir une dose sans
effet observable de 300 mg par kg de nourriture, soit 7 à 8 mg/kg
de poids corporel et par jour.
Divers systèmes permettant d'étudier in vivo et in vitro les
mutations géniques, les lésions et les réparations de l'ADN ainsi
que les effets chromosomiques, ont permis de constater que la d-
phénothrine n'avait pas d'effet mutagène.
Des études de deux ans ont également montré que la d-
phénothrine n'était pas cancérogène pour le rat ni la souris à des
doses allant jusqu'à 3000 mg par kg de nourriture. Aucune
tératogénicité ni embryotoxicité n'a été observée chez des foetus
de lapins et de souris dont les mères avaient reçu de la d-
phénotrine à des doses allant jusqu'à 1000 et 3000 mg par kg de
poids corporel, respectivement. Lors d'une étude de reproduction
chez le rat, portant sur deux générations, on a établi que la dose
sans effet observable était de 1000 mg par kg de nourriture.
Des rats exposés par inhalation à des doses très élevées de d-
phénothrine (jusqu'à 3760 mg/m3) pendant quatre heures ou,
quotidiennement, par voie orale, à une dose de 5000 mg par kg de
poids corporel, cinq jours durant, n'ont présenté aucune
dégénérescence myélinique ni désagrégation de l'axone au niveau du
nerf sciatique.
1.8 Effets sur les êtres humains
La d-phénothrine est utilisée depuis plus de 10 ans sans que
l'on ait signalé d'intoxication humaine.
Rien n'indique que cette substance puisse avoir des effets
nocifs sur l'homme pour peu qu'elle soit utilisée conformément aux
recommandations.
2. Conclusions
2.1 Population générale
L'exposition de la population dans son ensemble à la d-
phénothrine est vraisemblablement très faible et il n'y a
probablement aucun risque à cet égard si le produit est utilisé
conformément aux recommandations.
2.2 Exposition professionnelle
Si elle est utilisée de manière convenable, moyennant un
certain nombre de mesures d'hygiène et de sécurité, la d-
phénothrine ne devrait pas présenter de risque pour les personnes
qui lui sont exposées de par leur profession.
2.3. Environnement
Du fait qu'elle se dégrade rapidement à la lumière solaire et
qu'elle est principalement utilisée pour traiter les céréales
ensilées, l'exposition environnementale à la phénothrine est
vraisemblablement très faible. Dans ces conditions tout effet sur
l'environnement est extrêmement improbable.
3. Recommandations
Les niveaux d'exposition résultant d'une utilisation conforme
aux recommandations sont censés être extrêmement faibles, toutefois
on pourrait envisager de confirmer cette hypothèse en étendant la
surveillance à la d-phénothrine.
5.1. Metabolism in Mammals
Appraisal
After rats are treated with radiolabelled phenothrin, either by
single or repeated oral exposure, or dermally, the radioactivity is
rapidly (and almost totally) excreted into urine and faeces within
3 to 7 days. The major metabolic pathways of both trans- and cis-
phenothrin in rats are oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety and cleavage of
the ester linkage (see Fig. 3). Cleavage of the ester bond is more
difficult in the cis isomer than in the trans isomer. Thus, ester-
form metabolites oxidized in the various positions of the molecule are
the major metabolites of the cis isomer and are excreted mostly in the
faeces. However, ester-cleaved metabolites are the major products from
the trans isomer and are mostly excreted in urine.
When [1R, trans ]-phenothrin labelled with 14C at the methylene
moiety was given as a single oral dose (200 mg/kg body weight) to
Sprague Dawley male rats, the radiocarbon was rapidly eliminated,
57% and 43% being recovered within 3 days in urine and faeces,
respectively (Miyamoto et al., 1974). There was no detectable
radiocarbon in the expired air. Similarly, Sprague Dawley male
rats given a single oral dose (10 mg/kg body weight) of 14C-
[1R, trans ]-phenothrin, excreted the radiocarbon rapidly in the
urine (75%) and faeces (21%) (Kaneko et al., 1981).
In a study by Isobe et al. (1987), Sprague Dawley male and
female rats were given a single oral administration of 14C-
[1R, trans ]-phenothrin in corn oil at 4 and 200 mg/kg body weight.
Within 7 days the radiocarbon was almost completely eliminated in
the urine and faeces. The % elimination was as follows:
-----------------------------------------
Dose Urine Faeces
(mg/kg) Males Females Males Females
4 38 40 61 60
200 39 25 56 60
-----------------------------------------
Sprague Dawley male rats eliminated 65% of the dosed radiocarbon
in the faeces over 3 days after a single oral administration (200
mg/kg body weight) of 14C-[1R, cis ]-phenothrin (28) (Suzuki et al.,
1976), and 22 and 74% of the dose into urine and faeces,
respectively, 7 days after a single oral dose of 10 mg/kg body
weight (Kaneko et al., 1981).
When 14C-[1R, cis ]-phenothrin in corn oil was administered once
orally to Sprague Dawley male and female rats at 4 or 200 mg/kg
body weight, the radiocarbon was excreted into the urine (11-18%)
and faeces (81-87%) within 7 days. Similarly, when Sprague Dawley
rats were treated repeatedly with 14C-[1R,trans] or 14C-[1R,cis]
isomers at 4 mg/kg body weight per day for 14 days, the radiocarbon
was rapidly and almost completely excreted: 75-70% in urine and 24-
29% in faeces for the trans isomer, and 24% in urine and 72-73% in
faeces for the cis isomer (Isobe et. al., 1987).
The tissue residues in rats 7 days after a single oral dose of
14C-[1R, cis ]- or 14C-[1R, trans ]-phenothrin at 10 mg/kg body
weight were generally very low although the fat showed somewhat
higher residue levels (1-2.5 mg/kg) (Kaneko et al., 1981).
Similarly, high 14C residue levels (up to 23 mg/kg) were found in
the fat, 7 days after a single oral dose of the [1R,cis] isomer at
200 mg/kg body weight (Isobe et al., 1987).
The major metabolite of the trans isomer, when given as a
single oral dose of 200 mg/kg to rats, was 3-(4'-hydroxyphenoxy)-
benzoic acid (17) (4'OH-PBacid, 54%). There were smaller amounts
of 3-phenoxybenzoic acid (16) (PBacid, 9.5%) and its glycine
conjugate (Miyamoto et al., 1974).
When [1R, trans ]-phenothrin was given to rats at 4, 10, or 200
mg/kg body weight (oral single dose) or 4 mg/kg body weight
(repetitive oral dose for 14 days), the sulfate conjugate of 4'-OH-
PBacid was predominant, accounting for 28, 43, 28, and 55%,
respectively, of the dose. In addition, PBacid (4, 10, 5, and 6%),
its glycine conjugate (1,3,2, and 2%) and glucuronide (2,3,1, and
3%), and free 4'-OH-PBacid (2,11,3, and 3%) were found. The
sulfate conjugate of 3-(2'-hydroxyphenoxy)benzoic acid (18) (2'-
OH-PBacid) was also found as a minor metabolite (Kaneko et al.,
1981; Isobe et al., 1987).
When rats were given 14C-[1R, trans ]-phenothrin (10 mg/kg body
weight), the unmetabolized compound and two ester-form metabolites
were detected in their faeces in small amounts (0.4-1.2%), which
had hydroxymethyl (29) ( wt-alc- t-phe) or carboxyl group (30) wt-
acid- t-phe) (see Fig. 2) at the position of the trans methyl group
of chrysanthemic acid (Kaneko et al., 1981).
When Sprague Dawley rats were administered a single oral dose
of [1R, trans ]-phenothrin at 4 or 200 mg/kg body weight level or
given an oral dose of 4 mg/kg body weight per day for 14 days,
unmetabolized compound was found in the faeces (44-45, 44-60, and
14-16% of the dose, respectively). An ester-form metabolite, the
4'-hydroxy w(t)-acid derivative of trans- phenothrin, was also
detected (0.4-0.6%) (Isobe et al., 1987).
When male Sprague Dawley rats were given cis-phenothrin (200
mg/kg body weight), three ester-form metabolites, which accounted
for 14% of the dosed radioactivity, were found in the faeces.
These were 4'-hydroxy- cis-phenothrin (31) (4-OH- c-phe), an ester-
form derivative with the trans methyl of the isobutenyl group being
oxidized to carboxyl group (32) ( wt-acid- c-phe), and a compound
with the geminal-dimethyl groups oxidized (2-OH-) in addition to
both of the above modifications (33) (4'-OH, wt-acid, 2-OH(t)- c-phe)
(Suzuki et al., 1976).
In addition to this, the cis isomer gave rise to nine ester-
form metabolites in the faeces varying in amounts from 2% (4'-OH,
wc-alc-c-phe (34)) to 13% (4'-OH, wt-acid,2-OH(t)- c-phe (33)) of
the dosed radiocarbon after single oral administration. These
ester-form metabolites were transformed by oxidation reactions at
any of the following positions: 4'-position of the phenoxy group,
the trans or cis methyl of the isobutenyl groups, and the trans
methyl of the geminal-dimethyl group (Kaneko et al., 1981).
When Sprague Dawley rats were given a single oral dose of
[1R, cis ]-phenothrin at 4 or 200 mg/kg body weight level or an oral
dose of [1R, cis ]-phenothrin at 4 mg/kg body weight per day for 14
days, ester-form metabolites (1-9% of the dosed radioactivity) were
found, in addition to unmetabolized compound (17-59% of the dose).
The urine contained 4'-OH-PBacid as a sulfate conjugate (7-18%) and
in the free form (0.3-1%), and PBacid as glycine or glucuronide
conjugates and in the free form (0.3-1%) (Isobe et al., 1987).
Following the dermal treatment of male Sprague Dawley rats with
dust or emulsifiable concentrates (E.C.) of either 14C-[1R, trans ]-
or 14C-[1R, cis ]-phenothrin at 10 mg/kg body weight, the 14C
absorption into the body was estimated to be 3-7% of the initial
dose with dust and 8-17% with the E.C. After both dust and E.C.
treatments, the radiocarbon excreta (as a percentage of the initial
dose) recovered in the urine was 2.6-8.7% for the trans isomer, and
1.5-4.8% for the cis isomer, and in the faeces was 0.6-2.2% for the
trans isomer, and 3.0-12.3% for the cis isomer. Since the same
metabolites are formed following either oral exposure or dermal
treatment, it appears that both phenothrin isomers undergo the same
metabolism once in the systemic circulation, regardless of the
route of administration (Kaneko et al., 1981; Isobe et al., 1987).
Information concerning the comparative metabolism of racemic
(1RS) phenothrin and its d-isomer (1R) was obtained through a study
of CD rats and ddY mice given a single oral dose of either
[1R, trans ]-, [1S, trans ]-, [1RS, trans ]-, [1R, cis ]-, [1S, cis ]-,
or [1RS, cis ]-phenothrin. The radiocarbon derived from each isomer
was almost completely eliminated from the rats and mice within six
days after dosing. The trans isomers were mainly eliminated in the
urine (rat, 85-88%; mice, 65-75%) and the cis isomers mainly in the
faeces (rat, 57-71%; mice, 54-71%). The amounts of 14C in the
urine and faeces of rats and mice treated with the [1R,trans] and
[1R,cis] isomers did not differ significantly from those
corresponding to the [1RS,trans] and [1RS,cis] isomers, respectively.
The 14C tissue residues were very low, except in the fat. There
were no striking differences in 14C levels among the three trans
isomers and the three cis isomers. The 14C levels of the cis
isomers in fat (maximum 3.5 mg/kg) were three to seven times higher
than those of the trans isomers (less than 1 mg/ kg). The major
urinary and faecal metabolites were remarkably similar in both rats
and mice. In both rats and mice, there were virtually no
differences in the metabolic fate of the [1R,trans] and [1RS,
trans] isomers or of the [1R,cis] and [1RS,cis] isomers (Izumi et
al., 1984).
5.2. Enzymatic Systems for Biotransformation
In studies by Miyamoto et al. (1974), [IR, trans ]-phenothrin
(1 mmol/litre) was incubated with the 8000-g supernatant from a
liver homogenate of rats, mice, guinea-pigs, rabbits, or dogs at
37°C for 60 min in the absence of NADPH. The supernatant from the
guinea-pig was the most active in degrading [1R, trans ]-phenothrin,
followed by that of dog, rabbit, rat, and mouse. The major
metabolite in all the mammalian species tested was 3-phenoxybenzyl
alcohol (12) (PBalc). Smaller amounts of PBacid (16) and trace
amounts of 4'-OH-PBacid (17) were also found. However, in the
presence of NADPH, the amounts of PBacid and unidentified ether-
soluble metabolites increased in all species except dog. In
contrast to [1R, trans ]-phenothrin, [1R, cis ]-phenothrin was hardly
metabolized at all by the rat liver preparation in the absence of
NADPH. NADPH enhanced the degradation rate of the cis isomer,
leading to the formation of unidentified metabolites, while ester-
cleaved metabolites such as PBacid (16), PBalc (12) and 4'-OH-
PBacid (17) were found in very small amounts. When [1R, trans ]-,
[1R, cis ]-, [1S, trans ]-, and [1S, cis ]-phenothrin were incubated
with rat liver microsomes at 37.5°C for 30-60 min to estimate Km
and Vmax using a Lineweaver-Burk plot, the values for Km (0.11-0.17
mmol/litre) were similar for the four isomers, whereas the values
for Vmax were different; both the trans isomers yielded values for
Vmax 20-30 times larger than did the cis isomers (Miyamoto et al.,
1974).
In studies by Suzuki & Miyamoto (1978), pyrethroid
carboxyesterase(s) that hydrolyze esters of chrysanthemic acid
were purified from rat liver microsomes by cholic acid
solubilization, ammonium sulfate fractionation, heat treatment, and
DEAE-Sephadex A-50 column chromatography. The 45-fold-purified
enzyme (38% yield) probably consisted of a single protein with a
relative molecular mass of approximately 74 000, a Km of 0.21
mmol/litre for [1R, trans ]-phenothrin, and an optimum pH of 7-9.
It was susceptible to inhibition by organophosphate and carbamate
insecticides and insensitive to PCMB ( p-chloromercurybenzoic acid),
and mercuric and cupric ions. The enzyme seemed to require neither
coenzymes nor cofactors and hydrolysed the trans isomers of several
synthetic pyrethroids (tetramethrin, resmethrin, phenothrin, and
permethrin) well, at more or less similar rates. On the other hand,
the cis isomers were hydrolysed at rates 5-10 times lower than
their trans counterparts. The purified pyrethroid carboxyesterase
was apparently identical in nature to malathion carboxyesterase
and p-nitrophenyl acetate carboxyesterase.
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
Data on the acute toxicity of racemic and isomeric phenothrin
for aquatic organisms are summarized in Table 4.
6.1. Aquatic Organisms
Racemic phenothrin yielded 96-h LC50 values of 17-200 µg/litre
for the fish species tested (Table 4). The (1S)-optical isomers
were relatively non-toxic with LC50 values of 10 000 µg/litre,
whereas the (1R)-optical isomer and racemic phenothrin were of
similar toxicity with LC50 values between 120 and 200 µg/litre for
the killifish (Oryzias latipes) (Miyamoto, 1976).
When d-phenothrin was applied to ponds at the rates of 28 or 56
g/ha to control mosquito larvae, mayfly naiads were most affected
but no other arthropods (damselfly, dragonfly naiads, ostracods, or
diving beetle larvae) were seriously affected (Mulla et al., 1980).
6.2. Terrestrial Organisms
The available toxicity data for non-target terrestrial
organisms are very limited.
Phenothrin has low toxicity (acute oral dosage) for bobwhite
quail with an LD50 >2510 mg/kg body weight (Worthing & Walker,
1987). An 8-day feeding study with d-phenothrin on mallard duck
and bobwhite quail indicated LD50 values of >5620 and >5000 mg/kg
diet, respectivelya.
----------------------------------------------------------------------
a Personal communication from J.L. Noles, Ecological Effects
Branch, Hazard Evaluation Division, US Environmental Protection
Agency, November 1987.
Table 4. The acute toxicity of racemic and isomeric phenothrin to non-target aquatic organisms
-----------------------------------------------------------------------------------------------
Species Size Parameter Toxic Stereo- Systema Temper- Refer-
concentration isomeric ature enceb
(µg/litre) composition (°C)
-----------------------------------------------------------------------------------------------
Fish
Killifish adult 48-h LC50 200 racemic S 25 1
(Oryzias latipes) adult 48-h LC50 120 (+)-trans S 25 1
adult 48-h LC50 170 (+)-cis S 25 1
adult 48-h LC50 10 000 (-)-trans S 25 1
adult 48-h LC50 10 000 (-)-cis S 25 1
Rainbow trout 96-h LC50 17 racemic 2
(Salmo gairdneri)
Bluegill 96-h LC50 18 racemic 2
(Lepomis macrochirus)
Arthropods
Daphnia pulex 3-h LC50 50 000 racemic S 25 1
3-h LC50 25 000-50 000 (+)-trans S 25 1
3-h LC50 50 000 (+)-cis S 25 1
3-h LC50 50 000 (-)-trans S 25 1
3-h LC50 50 000 (-)-cis S 25 1
-----------------------------------------------------------------------------------------------
a s = static
b 1. Miyamoto (1976) 2. Worthing & Walker (1987)
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
The 1984 Joint FAO/WHO Meeting on Pesticide Residues concluded
that the toxicological data for racemic phenothrin can be used to
support that for d-phenothrin, owing to the similarity in
metabolism and toxicity between the two compounds (FAO/WHO,
1985b).
7.1. Single and Short-Term Exposures
The acute toxicity in rats and mice is extremely low. The LD50
values were >5000 mg/kg body weight when d-phenothrin was
administered orally, subcutaneously, dermally, or by
intraperitoneal injection to male and female Sprague Dawley rats
(Segawa, 1979a) and ddY mice (Segawa, 1979b) (Table 5).
Table 5. The acute toxicity of racemic phenothrin and d-phenothrin
to rats and micea
------------------------------------------------------------------
Compound/Route LD50 (mg/kg)
Rat Mice
Male Female Male Female
------------------------------------------------------------------
Phenothrin (racemic)
Oral > 5000 > 5000 > 5000 > 5000
Subcutaneous > 5000 > 5000 > 5000 > 5000
Dermal > 5000 > 5000 > 5000 > 5000
Intraperitoneal > 5000 > 5000 > 5000 > 5000
Inhalation > 1210b > 1210b > 1210b > 1210b
d-Phenothrin
Oral > 10 000 > 10 000 > 10 000 > 10 000
Subcutaneous > 10 000 > 10 000 > 10 000 > 10 000
Dermal > 10 000 > 10 000 > 5000 > 5000
Intraperitoneal > 10 000 > 10 000 > 10 000 > 10 000
Inhalation > 3760b > 3760b > 1180b > 1180b
------------------------------------------------------------------
a From: Segawa (1976; 1979a,b) and Khoda et al. (1977; 1979b; 1980)
b These are values for 4-h LC50 (mg/m3)
There were no differences between the effects on male and
female rats in acute toxicity studies carried out by Segawa (1976).
Signs of poisoning appeared rapidly following the intravenous
administration of phenothrin. These included fibrillation, tremor,
slow respiration, salivation, lacrimation, ataxia, and paralysis.
These signs, evident within 30 min to one hour following
administration, diminished rapidly to the point where, after 24 h,
there were no signs of toxicity.
No differences in the oral toxicity between racemic phenothrin
and d-phenothrin were detected by Segawa (1976; 1979a,b) (Table 5).
The intravenous LD50 values for racemic phenothrin in ICR mice
were 470 and 600 mg/kg for males and females, respectively, while
those for d-phenothrin were 265 and 315 mg/kg (Hiromori et al., 1984).
Following a 4-h acute inhalation exposure (whole body), the
LC50 values were >1210 mg/m3 for racemic phenothrin (particle size
5 µm) with both Sprague Dawley rats and ICR mice (Kohda et al., 1979b)
and >3760 and 1180 mg/m3 for d-phenothrin with Sprague Dawley rats
and ICR mice, respectively (Kohda et al., 1977; 1979b) (see Table 5).
At the higher concentration (3760 mg/m3), the mean particle size
was considered to be 0.72 µm and the cumulative distribution of
particles having a diameter between 0.46 and 1.09 µm was 88.9%,
according to a particle-size distribution experiment (Kohda et al.,
1980).
In studies by Kohda et al. (1979b), Sprague Dawley rats (15 of
each sex) were exposed (whole body) by inhalation to racemic
phenothrin at concentrations of 0, 43, or 220 mg/m3 (4 h per day, 5
days per week) for 4 weeks. After this treatment, animals (10/sex
per group) were sacrificed and the remaining rats were kept for 3
weeks without treatment. There were no adverse toxicological
effects on the animals exposed to the highest dose. Under the same
conditions (i.e. 220 mg/m3 for 4 weeks), racemic phenothrin did not
produce any adverse effects in male or female ICR mice. Similarly,
d-phenothrin also produced no adverse toxicological effects in
Sprague Dawley rats or ICR mice (both male and female) following
whole body exposure at 210 mg/m3 (particle size 5 µm) for 4 weeks
(Kohda et al., 1979b).
The acute oral and intraperitoneal toxicity of phenothrin
metabolites in rats and mice are shown in Table 6 and 7 (Kohda et
al., 1979a; FAO/WHO, 1981).
Table 6. The acute oral toxicity of phenothrin
metabolites
-----------------------------------------------------
Chemical No.a Species LD50
(mg/kg)
-----------------------------------------------------
3-Phenoxybenzyl alcohol 12 Rat 1330
3-Phenoxybenzaldehyde 13 Rat 600
-----------------------------------------------------
a Chemical identification number used in Fig. 2
Table 7. The acute intraperitoneal toxicity of several phenothrin
metabolites in mice
----------------------------------------------------------------------
Chemical No.a LD50 (mg/kg)
Male Female
----------------------------------------------------------------------
3-Phenoxybenzyl alcohol 12 371 424
3-(4'-hydroxyphenoxy)benzyl alcohol 14 750-1000 750-1000
3-(2'-hydroxyphenoxy)benzyl alcohol 15 876 778
3-Phenoxybenzoic acid 16 154 169
3-(4'-hydroxyphenoxy)benzoic acid 17 783 745
3-(2'-hydroxyphenoxy)benzoic acid 18 859 912
3-Phenoxybenzaldehyde 13 415 416
----------------------------------------------------------------------
a Chemical identification number used in Fig. 2
7.2. Long-Term Exposures and Carcinogenicity
Appraisal
There have been several long-term feeding studies with racemic
phenothrin or d-phenothrin (dose levels ranging from 200 to 10 000
mg/kg diet) in rats and mice, the length of exposure ranging from 6
months to 2 years. A slight increase in liver weight and a
significant difference in some clinical/chemistry parameters from those
of controls were observed at high doses. However, the no-observed-
effect level (NOEL) values obtained in these studies were high (300-
1000 mg/kg diet, corresponding to around 40-160 mg/kg body weight) and
no tumorigenicity was observed. Two feeding studies with d-phenothrin
(doses of 100-3000 mg/kg diet with exposure period 26 and 52 weeks) in
dogs revealed similar results, the NOEL being 300 mg/kg diet
(corresponding to around 7-8 mg/kg body weight per day). No
tumorigenicity relating to phenothrin feeding was detected in these
studies.
7.2.1. Rat
In a study by Murakami et al. (1981), [1R, cis]-phenothrin
(d-phenothrin) was administered to Sprague Dawley rats (20 of each
sex per group) at dose levels of 0, 1, 3, or 10 g/kg diet for 6
months. Ten rats of each sex per group were sacrificed after 3
months. d-Phenothrin had no significant effect on mortality,
clinical signs, ophthalmology, urinalysis, or gross and
histopathological findings. The serum albumin level was elevated
after 3 months in males fed 10 g/kg and in females fed 3 or 10
g/kg, and after 6 months in males fed 3 or 10 g/kg. The albumin-
globulin ratio was raised after 3 months in males fed 3 or 10 g/kg
and in females fed 10 g/kg, and in both males and females fed 10
g/kg after 6 months. Absolute and relative liver weights in both
males and females fed 3 or 10 g/kg were increased. Based on these
data, it was concluded that the NOEL for d-phenothrin in this study
was 1 g/kg diet for both sexes (55.4 mg/kg body weight per day for
males and 63.3 mg/kg body weight per day for females).
In a standard oncogenicity study, Fisher-344 rats (50 of each
sex per group) were fed d-phenothrin at dose levels of 0, 300, 1000
or 3000 mg/kg diet for at least 105 weeks in males and at least 118
weeks in females. Additional rats (30 of each sex per group) were
assigned to a chronic toxicity study with a 52-week interim
sacrifice. There were no significant effects on clinical signs,
mortality, food and water consumption, ophthalmology, blood
biochemistry, haematology, or urinalysis. However, the body weight
gain in females fed d-phenothrin at 3000 mg/kg was reduced, and the
relative liver weight was increased in females fed 3000 mg/kg for
52 weeks and, at the end of the oncogenicity study, in males fed
3000 mg/kg. Microscopic examination revealed that the incidence of
cystic dilatation of the sinuses of the mesenteric lymph nodes and
of periacinar hepatocytic hypertrophy was higher in males fed 3000
mg/kg for at least 105 weeks. d-Phenothrin did not show any
oncogenic activity to rats at up to 3000 mg/kg. Although at this
dose an increase in the incidence of adenomas and carcinomas of the
preputial gland was seen in males, the 1988 Joint FAO/WHO Meeting
on Pesticide Residues (FAO/WHO, 1988b) considered it unlikely that
this finding was of toxicological significance. The NOEL was 1000
mg/kg diet for both sexes (47 mg/kg body weight per day for males
and 56 mg/kg body weight per day for females) (Martin et al.,
1987).
When Sprague Dawley rats (50 of each sex per group) were fed a
diet containing racemic phenothrin (0, 200, 600, 2000, and 6000
mg/kg diet) for 2 years, body weight and food consumption were
slightly depressed at 6000 mg/kg in both males and females.
Ophthalmological examinations, haematological studies, urinalyses,
and clinical chemistry studies were performed at various intervals.
At the end of the study all animals were sacrificed and examined
for gross abnormalities, and extensive microscopic examinations
were conducted on a variety of tissues and organs. There were no
abnormal clinical or behavioral problems associated with
phenothrin administration. The survival rate of all groups of
treated rats was similar to that of controls. Males fed 6000 mg/kg
showed a significant increase in serum glutamine-pyruvate
aminotransferase activity. Ophthalmological examinations revealed
some abnormalities, all of which appeared to be age related.
Histopathological examination revealed no significant differences
between the treated groups and the control group with respect to
severity of lesions. No histopathological changes suggestive of
oncogenicity resulting from phenothrin treatment were found
(Hiromori et al., 1980).
7.2.2. Mouse
When Swiss White mice (50 of each sex per group) were fed
racemic phenothrin for 18 months at dose levels of 0, 300, 1000, or
3000 mg/kg diet, there were no significant effects on mortality,
clinical signs, haematologic values, clinical chemistry parameters,
or gross pathological findings. Slight body weight depression
occurred in males fed 3000 mg/kg, and increased liver weight was
found at the highest dose level in both males and females. There
was a statistically significant difference (compared with the
controls) in lung amyloidosis in the 1000 and 3000 mg/kg dose
groups, but no significant increase in tumours attributable to
phenothrin ingestion (Murakami et al., 1980).
In studies by Amyes et al. (1987), B6C3F1 hybrid mice (90 of
each sex per group) were fed d-phenothrin in the diet at dose
levels of 0, 300, 1000, or 3000 mg/kg. Fifty of each sex per group
were allocated to a standard oncogenicity study lasting 104 weeks.
The remaining mice were assigned to a chronic toxicity study, where
10 of each sex per group were sacrificed for interim study after 26
or 53 weeks and the remaining animals were examined after 78 weeks
of treatment. There were no compound-related effects on clinical
signs, mortality, ophthalmology, blood biochemistry, haematology,
or urinalysis. However, body weight gains for males fed d-
phenothrin at 3000 mg/kg were reduced and relative liver weights
were increased in both sexes fed 3000 mg/kg and in males receiving
1000 mg/kg. Microscopic examination revealed that the incidence of
periacinar hepatocyte hypertrophy with cytoplasmic eosinophilia was
higher in males fed 3000 mg/kg. The incidence of liver tumors
appeared higher in phenothrin-treated female mice than in control
females, but the difference was not statistically significant. It
was concluded, therefore, that administration of d-phenothrin to
mice for 2 years at dietary levels of up to 3000 mg/kg diet did not
significantly disturb the tumour burden or tumour profile of B6C3F1
hybrid mice. The NOEL in this study was 300 mg/kg diet for males
(40 mg/kg body weight per day) and 1000 mg/kg diet for females (164
mg/kg body weight per day).
7.2.3. Dog
When beagle dogs (six of each sex per group) were fed d-
phenothrin at dose levels of 0, 100, 300, or 1000 mg/kg diet for 26
weeks, there were no compound-related abnormal findings in
mortality, clinical signs, body weight, food consumption,
ophthalmology, gross or microscopic pathology, haematology, or
urinalysis studies. However, the alkaline phosphatase activity in
males fed 300 mg/kg and males and females fed 1000 mg/kg was
elevated and a slight increase in the mean relative liver weight in
males fed 1000 mg/kg was noted. The NOEL in this study was 300
mg/kg (Pence et al., 1981).
In a study by Cox et al. (1987), beagle dogs (four of each sex
per group) were fed d-phenothrin at dose levels of 0, 100, 300,
1000, or 3000 mg/kg diet for 52 weeks. There were no significant
effects on clinical signs, body weight, food consumption,
ophthalmology, or urinalysis. However, decreases in erythrocyte
count, haemoglobin concentration, haematocrit, and total blood
protein were noted in both male and female dogs fed 3000 mg/kg,
whereas mean absolute and relative liver weights increased.
Compound-related histopathological alterations were noted in the
adrenal glands and liver. Focal degeneration of the adrenal cortex
with cytoplasmic deposition of crystalline material was seen in one
male dog fed 1000 mg/kg and four dogs fed 3000 mg/kg. The chemical
nature or biological significance of this crystalline material was
not recorded. Hepatocytes appeared to enlargen slightly in one
male dog fed 1000 mg/kg and seven dogs fed 3000 mg/kg. The NOEL in
this study was 300 mg/kg diet for males and 1000 mg/kg for females
(8.24 and 26.77 mg/kg body weight per day for males and females,
respectively).
7.3. Mutagenicity
The results obtained in vivo and in vitro test system indicate
that d-phenothrin does not exhibit any mutagenic properties or
cause chromosomal or DNA damage.
In a DNA-repair test with Bacillus subtilis (M45 rec- and H17
wild type strains) using dose levels of up to 5 mg/disk per plate,
d-phenothrin did not inhibit the growth of any strain at any dose
level, whereas the positive control, mitomycin C, showed a clear
effect. The negative control gave a result similar to that of d-
phenothrin (Kishida & Suzuki, 1981a).
A mutagenicity test with Escherichia coli (WP2 uvr) and
Salmonella typhimurium (TA 1535, TA 1537, TA 1538, TA 98, and TA
100) using d-phenothrin at dose levels of up to 5 mg/plate with and
without metabolizing enzyme system (S9 mix) yielded negative
results, whereas a positive control gave a significant number of
mutants (Kishida & Suzuki, 1981b).
In a host-mediated assay using S. typhimurium G46 (indicator
bacteria), d-phenothrin in corn oil was given orally (twice with a
24-h interval) to groups of six male ICR mice at dose levels of
2500 or 5000 mg/kg body weight. Soon after the last administration,
each mouse was injected intraperitoneally with the indicator cells.
Three hours later, the bacterial mutation frequency in d-
phenothrin-treated mice was no greater than that in the control
group (Kishida & Suzuki, 1981c).
Suzuki et al. (1981) examined d-phenothrin for its ability to
induce chromosomal aberrations in vivo using bone marrow cells.
ICR mice were treated intrapertioneally with single doses of 2500,
5000, or 10 000 mg/kg body weight and sacrificed 6, 24, or 48 h
after treatment. No chromosomal aberrations as a result of d-
phenothrin treatment were detected.
In an in vitro chromosomal aberration test, Chinese hamster
ovary cells (CHO-K1) were treated with d-phenothrin (dose levels:
2 x 10-5 to 2 x 10-4 mol/litre for 24 and 48 h in the absence of S9
mix; 5 x 10-5 to 5 x 10-4 mol/litre for 6 h in the presence of S9
mix). No significant increase in the number of cells with
chromosomal aberrations was observed (Kogiso et al., 1986).
The ability of d-phenothrin to induce sister-chromatid
exchanges (SCEs) were tested in cultured mouse embryonic cells in
vitro. At doses of 10-5, 10-4, and 10-3 mol/litre (with and
without S9 mix), d-phenothrin did not induce any increase in the
frequency of SCEs (Suzuki & Miyamoto, 1981).
In a study of unscheduled DNA synthesis, Hela S3 cells were
treated with d-phenothrin at dose levels of 0, 0.25, 0.5, 1.0, 2.0,
or 4.0 mg/ml in the presence of 3H-thymidine (with and without S9
mix) for 3 h, and the incorporation of 3H-thymidine into DNA was
measured. There was no significant increase in the radioactivity
of DNA from cells treated with d-phenothrin (Foster et al., 1984).
7.4. Reproduction, Embryotoxicity, and Teratogenicity
No teratogenicity or reproductive effects were observed when
phenothrin was fed to rabbits or mice during the major
organogenesis period of gestation or to rats in 3-generation
reproduction studies.
7.4.1. Embryotoxicity and teratogenicity
In studies by Ladd et al. (1976), pregnant New Zealand White
rabbits (17 per group) were administered racemic phenothrin orally
at dose levels of 0, 3, 10, or 30 mg/kg body weight on days 6 to 18
of gestation. They were sacrificed on day 29 and the young
obtained by caesarian section were examined. At 30 mg/kg, the body
weight of females decreased during gestation, and there was a
slight decrease in the number of live young and a slight reduction
in fetal weight. Racemic phenothrin had no apparent teratogenic
effect, as shown by a lack of gross internal or external somatic
abnormalities and by normal fetal skeletal development following
prenatal exposure.
Pregnant New Zealand White rabbits (15 per group) were orally
administered d-phenothrin by intubation (0, 10, 100, or 1000 mg/kg
body weight per day) on days 6 to 18 of gestation, and were
sacrificed on day 29 or 30. Following caesarian section, 50% of
the pups were maintained for 24 h to evaluate survival. No
abnormalities were observed among the does (body weight, food
consumption, clinical observations, and necropsy) or foetuses
(implantation sites, corpora lutea, resorption sites, weight,
condition, and viability). Data on foetal survival and from
internal and external examinations for abnormalities showed not
significant effects from administrating d-phenothrin during
gestation (Rutter, 1974).
In studies by Nakamoto et al. (1973), d-phenothrin was orally
administered to pregnant ICR mice (17 or 18 per group) at dose
levels of 0, 30, 300, or 3000 mg/kg body weight on days 7 to 12 of
gestation (not covering the whole period of organogenesis). The
dams were sacrificed on day 18 of gestation and the pups were
obtained by caesarian section. Other mice (7 per group) were given
d-phenothrin at dose levels of 0, 300, or 3000 mg/kg to evaluate
postnatal effect. These mice were allowed to deliver naturally and
the pups were kept for 29 days. At these levels, d-phenothrin
showed no adverse effects, as indicated by maternal growth, fetal
mortality and external and internal examination of fetuses for
teratogenic or embryotoxic effects.
7.4.2. Reproduction studies
In a standard 3-generation (2 litters per generation)
reproduction study, groups of rats (8 male and 16 female Charles
River albino rats per group) were fed racemic phenothrin at dose
levels of 0, 200, 600, or 2000 mg/kg diet. Various reproduction
indices (i.e. mating index, fecundity index, male fertility index,
female fertility index, and incidence of parturition) were
measured. The adult rats showed no significant mortality or
complications during the study, and the reproductive parameters
revealed no significant dose-related adverse effects attributable
to phenothrin. Gross and microscopic findings indicated no adverse
effect resulting from dietary phenothrin. It was concluded that
phenothrin had no effect on reproduction (Takatsuka et al., 1980).
In a study by Tesh et al. (1978), d-phenothrin was fed to
Charles River CD rats (30 of each sex per group) at dose levels of
0, 300, 1000, or 3000 mg/kg diet throughout two successive
generations and up to the maturation of a third generation. At 300
and 1000 mg/kg, there was no adverse effect upon mortality, somatic
growth, development, or reproductive performance. At 3000 mg/kg,
mortality, body weight, and reproductive performance showed no
significant response to treatment, and selected F2 animals reared
to maturity were in all respects comparable with control rats.
However, F0 and F1 females and selected F2B male and female
weanlings showed a slight but consistent increase in the relative
liver weight. The NOEL in this study was 1000 mg/kg diet.
7.5. Neurotoxicity
Sprague Dawley rats exposed to d-phenothrin by inhalation at
concentratons of up to 3760 mg/m3 for 4 h showed no toxic signs as
a result of exposure. Histopathologically, there were no compound
related alterations in the sciatic nerve (Kohda et al., 1977).
When d-phenothrin was given to Sprague Dawley rats orally for 5
consecutive days (5 g/kg body weight per day), one out of ten
female rats died after four doses and signs of poisoning
(piloerections and urinary incontinence) were noted in several of
the animals. However, these signs disappeared rapidly at the end
of the treatment and there were no other signs of poisoning such as
leg weakness or ataxia. All animals were sacrificed 3 days after
the final dose, and histopathologically examination of the sciatic
nerve revealed minute changes in axon and myelin, characterized by
very slight axonal swelling, axonal disintegration, and/or
demyelination. Since there were similar changes in the control
animals, it was suggested that they were not due to the d-
phenothrin. It was considered that the oral administration of very
high doses of d-phenothrin does not lead to the neurotoxic effects
observed with several other pyrethroid esters (Okuno et al., 1978).
7.6. Miscellaneous Effects
No d-phenothrin-attributable pharmacological effects were
detected in various tests (e.g., spontaneous movement of isolated
guinea-pig ileum, contraction of the rat phrenic nerve diaphragm
preparation, cardiopulmonary physiology of anaesthetized cats,
coordination, and spontaneous movement of mice, and rectal
temperature of rats) at doses of 100-300 µg/ml in vitro, 3 mg/kg
intravenous, or 100-300 mg/kg intraperitoneal. A tentative arousal
response was recorded in the electroencephalogram of cats given d-
phenothrin (4 mg/kg) intraperitoneally, as is commonly observed in
animals given synthetic pyrethroids (Hara et al., 1974).
7.7. Mechanisms of Toxicity - Mode of Action
Some synthetic pyrethroids given intravenously to rats cause
either tremor (T-syndrome) or choreoathetosis with salivation (CS-
syndrome) (Verschoyl & Aldridge, 1980). However, d-phenothrin
(>600 mg/kg body weight) injected intravenously into the lateral
tail vein caused neither T-syndrome nor CS-syndrome, due to its
very low acute toxicity. From a study involving intracerebral
dosing with [1R, cis]- or [1R, trans]-phenothrin in mice, both
compounds were classified as Type I pyrethroids based on the
occurrence of tremors (Lawrence & Casida, 1982) and on
neurophysiological studies in cockroach cercal sensory nerves
(Gammon et al., 1981).
8. EFFECTS ON HUMANS
Although d-phenothrin has been used for more than 10 years, no
toxic effects and no cases of poisoning have been reported.
8.1. Clinical Studies
In a study by Hashimoto et al. (1980), d-phenothrin (talc
powder formulation with Span 80 as a stabilizer) was applied to the
head hair and pudenda hair of eight male human volunteers (three
times at intervals of 3 days) at a dose of 32 mg/man per
administration (0.44 to 0.67 mg/kg body weight per day). d-
Phenothrin powder was washed off 1 h after application. There were
no significant abnormalities due to d-phenothrin in terms of dermal
irritation, clinical signs, or blood biochemical and haematological
parameters. The blood levels of d-phenothrin were below the
detection limit of 0.006 mg/kg.
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has
discussed and evaluated d-phenothrin at its meetings in 1979, 1980,
1984, and 1988 (FAO/WHO, 1980, 1981, 1985a, 1988b).
Since 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg
body weight has been established.
In the WHO Recommended Classification of Pesticides by Hazard,
technical phenothrin is classified as unlikely to present an acute
hazard in normal use (WHO, 1988).
REFERENCES
AMYES, S.J., MARTIN, P.A., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Oncogenicity and toxicity study
in mice, Suffolk, United Kingdom, Life Science Research (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of syn-
thetic pyrethroids in fruit and vegetables by gas-liquid and high-
performance liquid chromatography. Analyst, 107: 206-212.
COX, R.H., SUTHERLAND, J.D., VOELKER, R.W., ALSAKER, R.D.,
VARGAS, K.J., LEWIS, S.A., & HAGEN, W.J. (1987) Chronic toxicity study
in dogs with Sumithrin technical grade, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
DESMARCHELIER, J.M. (1980) Comparative study of analytical methods for
bioresmethrin, phenothrin, d-phenothrin, pyrethrum I, carbaryl,
fenitrothion, methacrifos, pirimifos-methyl and dichlorfos on various
grains. J. Pestic. Sci., 5: 521-532.
DOI, T., SAKAUE, S., & HORIBA, M. (1985) Analysis of d-phenothrin and
its optical isomers in technical preparations and formulations. J.
Assoc. Anal. Chem., 68(5): 911-916.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC, American
Chemical Society, p. 229 (ACS Symposium Series No. 42).
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to a
synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
FAO (1982) Second Government Consultation on International Harmoniz-
ation of Pesticide Registration Requirements, Rome, 11-15 October,
1982, Food and Agriculture Organization of the United Nations.
FAO/WHO (1980) Pesticide residues in food: 1979 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 427-452
(FAO Plant Production and Protection Paper 20 Sup.).
FAO/WHO (1981) Pesticide residues in food: 1980 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 394-403
(FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1985a) Pesticide residues in food: 1984 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 689-694
(FAO Plant Production and Protection Paper 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues, 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1988a) Pesticide residues in food: 1987 Evaluations. Part 1
- Residues, Rome, Food and Agriculture Organization of the United
Nations, pp. 109-113 (FAO Plant Production and Protection Paper 86.1).
FAO/WHO (1988b) Pesticide residues in food. Report of the 1988 Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues, Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper 92).
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous sensation
between synthetic pyrethroid insecticides. Contact Dermatitis, 13(3):
140-147.
FOSTER, R., TIPINS, R.S., DE VENEZIA, V., & NUNZIATA, A. (1984)
Unscheduled DNA synthesis in human cells, cell line: Hela S3, test
substance: Sumithrin, Suffolk, United Kingdom, Life Science Research,
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
FUJIMOTO, K., ITAYA, N., OKUNO, Y., KADOTA, T., & YAMAGUCHI,T. (1973)
A new insecticidal pyrethroid ester. Agric. biol. Chem., 37:2681-2682.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent class
antagonize GABA action at the crayfish neuromuscular junction.
Neurosci. Lett., 40:163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid toxi-
cology: Protective effects of diazepam and phenobarbital in the mouse
and the cockroach. Toxicol. appl. Pharmacol., 66: 290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural variations
affecting pyrethroid neurotoxicity. Neurobehav. Toxicol. Teratol., 4(6):
793-799.
HARA, Y., MIYAGISHI, A., OHTSUKA, M., ASAMI, Y., KUROKAWA, Y.,
& MIYAMOTO, J. (1974) Pharmacological study of Sumithrin: Effects on
respiration, blood pressure, and peripheral central nervous systems
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HASHIMOTO, S., OKANO, S., YAMADA, H., & MIYAMOTO, J. (1980)
Human volunteer study with phenothrin and d-phenothrin
powder (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
HAYASHI, A. (1977) [Safe use of insecticides (I)] [Bo-satsu-kyo News,]
No. 1 p. 10 (June) (in Japanese).
HIROMORI, T., KOYAMA, Y., OKUNO, Y., ARAI, M., ITO, N., &
MIYAMOTO, J. (1980) Two-year chronic toxicity study of S-2539 in
rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HIROMORI, T., HARA, S., KADOTA, T., & MIYAMOTO, J. (1984) Acute
intravenous toxicity of phenothrin and d-phenothrin in mice
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
ISOBE, N., MATSUNAGA, H., NAKATSUKA, I., & YOSITAKE, A.
(1987) Metabolism of [1R, trans]- and [1R, cis]- isomers of phenothrin
in rats (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
ITAYA, N., KAMOSHITA, K., KITAMURA, S., OKUNO, Y., MIZUTANI,
T., NAKAI, S., KAMEDA, N., & FUJIMOTO, K. (1969) Esters of cyclo-
propanecarboxylic acids. Ger. Offen., 1: 926, 433.
IZUMI, T., KANEKO, H., MATSUO, M., & MIYAMOTO, J. (1984)
Comparative metabolism of the six optical stereo isomers of phenothrin
in rats and mice. J. Pestic. Sci., 9: 259-267.
KANEKO, H., OHAKAWA, H., & MIYAMOTO, J. (1981) Absorption and
metabolism of dermally applied phenothrin in rats. J. Pestic. Sci., 6:
169-182.
KISHIDA, F. & SUZUKI, H. (1981a) Studies on DNA-damaging capacity of
Sumithrin with Bacillus subtilis (Unpublished Technical Report No. ET-
10-0069 submitted to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA. F. & SUZUKI, H. (1981b) Gene mutation test of Sumithrin in
bacterial system (Unpublished Technical Report No. ET-10-0068 submitted
to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA, F. & SUZUKI, H. (1981c) Mutagenicity test of Sumithrin in
host-mediated assay (Unpublished Technical Report No. ET-10-0067 sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
KOGISO, S., HARA, M., ITO, K., IWAWAKI, H., & YOSHITAKE, A.
(1986) In vitro chromosomal aberration test of S-2539F in Chinese
hamster ovary cells (CHO-K1) (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1977) Acute
inhalation toxicity of d-phenothrin (S-2539 Forte) in rats (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
KOHDA, H., KENEDO, H., OHKAWA, H., KADOTA, T., & MIYAMOTO, J.
(1979a) Acute intraperitoneal toxicity of permethrin metabolites in
mice (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
KOHDA, H., NISHIMOTO, K., KADOTA, K., & MIYAMOTO, J. (1979b)
Acute and subacute inhalation toxicity studies of S-2539 Forte in rats
and mice (Unpublished report submitted to WHO by Sumitomo Chemical
Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1980) Particle
size distribution and mean particle size in acute inhalation toxicity
study of d-phenothrin (S-2539 forte) in rats (Unpublished Technical
Report No. ET-00-0054 submitted to WHO by Sumitomo Chemial Co.,
Ltd).
LADD, R., SMITH, P.S., JENKINS, D.H., KENNEDY, G.L., Jr, KINOSHITA,
F.K., & KEPLINGER, M.L. (1976) Teratogenic study with S-2539 in albino
rabbits, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: mouse
intracerebral structure-toxicity relationship. Pestic. Biochem. Physiol.,
18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions of
pyrethroid insecticides with chloride ionophore-associated binding
sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticides, London, Taylor &
Francis Ltd, p. 440.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel modifi-
cation as the basis for the variation in the nerve membrane effects of
pyrethroids and DDT analogs. Pestic. Biochem. Physiol., 20: 203-216.
MARTIN, P.A., AMYES, S.J., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Combined toxicity and
oncogenicity study in rats, Suffolk, United Kingdom, Life Science
Research (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of synthetic
pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue analysis of
synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry-human welfare
and the environment. Proceedings of the Fifth International Congress of
Pesticide Chemistry, Kyoto, Japan, 29 August to 4 September, 1982, Oxford,
Pergamon Press, Vol. 1-4.
MIYAMOTO, J., SUZUKI, T., & NAKAE, C. (1974) Metabolism of phenothrin
or 3-phenoxybenzyl d-trans-chrysanthemate in mammals. Pestic. Biochem.
Physiol., 4: 438-450.
MIYAMOTO, J., KOGISO, S., & MATSUO, M. (1984) Additional toxicological
information for phenothrin (Unpublished Technical Report No. ET-40-0090
from Sumitomo Chemical Co., Ltd).
MULLA, M.S., DARWAZEH, H.A., & DHILLON, M.S. (1980) New
pyrethroids as mosquito larvicides and their effects on nontarget
organisms. Mosq. News, 40: 6-12.
MURAKAMI, M., HIROMORI, T., ITO, S., & HOSOKAWA, S. (1981)
Six month oral toxicity study of S2539 Forte (Sumithrin(R)) in rats
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURAKAMI, J., ITO, S., OKUNO, Y., ARAI, M., ITO, N., & MIYAMOTO, J.
(1980) Eighteen-month chronic oral toxicity and tumorigenicity study of
S-2539 in mice, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURANO, A. (1972) Determination of the optical isomers of insecticidal
pyrethroids by gas-liquid chromatography. Agric. biol. Chem., 36:
2203-2211.
NAKAMOTO, N., KATO, T., & MIYAMOTO, J. (1973) Teratogenicity study
of S-2539 Forte in mice (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
NAMBU, K., OHKAWA, H., & MIYAMOTO, J. (1980) Metabolic fate of
phenothrin in plants and soils. J. Pestic. Sci., 5: 177-197.
NAMBU, K., TAKIMOTO, Y., & MIYAMOTO, J. (1981) Degradation of
phenothrin in stored wheat grains. J. Pestic. Sci., 6: 183-191.
NIOSH (1983) Registry of toxic effects of chemical substances, 1981-
1982 edition, Cincinnati, Ohio, US National Institute for Occupational
Safety and Health, p. 1003.
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
OHNISHI, J., IKEDA, M., SUZUKI, Y., & MATSUDA, T. (1987) Residue
analysis of d-phenothrin, fenitrothion and piperonyl butoxide in stored
wheat, wheat milling fractions and bread (interim report) (Unpublished
Report No. ER-RD-8719 submitted to WHO by Sumitomo Chemical Co.,
Ltd).
OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1978) Neurotoxicity study
of d-phenothrin (S-2539 Forte(R)) in rats by repeated oral administration
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
PAPADOPOULOU-MOURKIDOU, E., IWATA, Y., & GUNTHER, F.A.
(1981) Utilization of an infrared detector for selective liquid
chromatographic analysis. 2. Formulation analysis of the pyrethroid
insecticides allethrin, decamethrin, cypermethrin, phenothrin and
tetramethrin. J. agric. food Chem., 29: 1105-1111.
PENCE, D.H., HAGEN, W.H., WLSAKER, R.D., HASTINGS, T.F.,
DAWKINS, B.G., TACEY, R.L., & MARSHALL, P.M. (1981) Subchronic
toxicity study of S2539-F in dogs, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids on a
nerve-muscle preparation of the clawed frog, Xenopus laevis. Pestic.
Biochem. Physiol., 25: 176-187.
RUTTER, H.A. (1974) Teratogenicity study in rabbits: S-2539 Forte,
Vienna, Virginia, Hazleton Laboratories Inc. (Unpublished report sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
RUZO, L.O., SMITH, I.H., & CASIDA, J.E. (1982) Pyrethroid
photochemistry: photooxidation reactions of the chrysanthemates,
phenothrin and tetramethrin. J. agric. food Chem., 30: 110-115.
SAKAUE, S., KITAJIMA, M., HORIBA, M., & YAMAMOTO, S. (1981) Gas
chromatographic and high performance liquid chromatographic
determination of d-phenothrin (Sumithrin) in formulations. Agric. biol.
Chem., 45: 1135-1140.
SEGAWA, T. (1976) Acute toxicity study of S-2539 in rats and mice,
Hiroshima, Hiroshima University School of Medicine (Unpublished report
submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979a) Acute oral, subcutaneous, dermal and intraperitoneal
toxicity studies of S-2539 Forte in rats, Hiroshima, Hiroshima
University School of Medicine (Unpublished Technical Report
No. ET-31-0033 submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979b) Acute oral, subcutaneous, dermal and
intraperitoneal toxicity studies of S-2539 Forte in mice, Hiroshima,
Hiroshima University School of Medicine (Unpublished Technical Report
No. ET-31-0032 submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H. & MIYAMOTO, J. (1981) In vitro sister chromatid
exchanges of Sumithrin (Unpublished Technical Report No. ET-10-0071
submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H., HARA, M., & MIYAMOTO, J. (1981) In vivo
chromosomal aberration test of Sumithrin on bone marrow cells of
mice (Unpublished Technical Report No. ET-10-0072 submitted to WHO by
Sumitomo Chemical Co., Ltd).
SUZUKI, T. & MIYAMOTO, J. (1978) Purification and properties of
pyrethroid carboxyesterase in rat liver microsome. Pestic. Biochem.
Physiol., 8: 186-198.
SUZUKI, T., OHNO, N., & MIYAMOTO, J. (1976) New metabolites of
(+)-cis-phenothrin, 3-phenoxybenzyl (+)-cis chrysanthemate, in rats.
J. Pestic. Sci., 1: 151-152.
TAKATSUKA, M., OKUNO, Y., SUZUKI, T., KADOTA, T., YASUDA, M.,
& MIYAMOTO, J. (1980) Three-generation reproduction study of S-2539
in rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
TAKIMOTO, Y., KATO, T., & MIYAMOTO, J. (1977) Residue analyses
of phenothrin (S-2539) in crops (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
TESH, J.M., WILLOUGHBY, C.R., & FOWLER, J.S.L. (1987) Sumithrin:
Effects upon reproductive performance of rats treated continuously
throughout two successive generations, Suffolk, United Kingdom, Life
Science Research (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
TURNBULL, S.R. & ARDLEY, J.H. (1987) Determination of d-phenothrin,
piperonyl butoxide and fenitrothion residues on wheat, derived
fractions and breads, at approximately one, three and six months post-
treatment, Australia, The Wellcome Foundation Ltd (Unpublished Report
No. AESH 87-11).
VAN DEN BERCKEN, J. (1977) The action of allethrin on the peripheral
nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels in
frog myelinated nerve membrane. In: Insect: Neurobiology and pesticide
action, London, Society of Chemical Industry, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER
ZALM, J.M. (1973) DDT-like action of allethrin in the sensory nervous
system of Xenopus laevis. Eur. J. Phamacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A.
(1979) Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VERSCHOYLE, R.D. & ALDRIDGE, W.N.(1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45: 325-
329.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Phamacol., 58: 501-504.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system. Neuropathol.
appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J.
(1982a) Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed frog,
Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN
BERCKEN, J. (1982b) Similar mode of action of pyrethroids and DDT on
sodium channel gating in mylinated nerves. Nature (Lond.), 295: 601-
603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF,
R.G.D.M., & VAN DEN BERCKEN, J. (1983) Temperature and structure-
dependent interaction of pyrethroids with the sodium channels in frog
node of Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WHO (1979) WHO Technical Report Series, No. 634 (Safe use of
pesticides. Third Report of the WHO Expert Committee on Vector Biology
and Control), pp 18-23.
WHO (1988) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1988-1989, WHO, World Health
Organization (WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1987) d-Phenothrin. In: The pesticide
manual, 8th ed., Croydon, British Crop Protection Council, pp. 654-
655.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
APPENDIX
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs ( Xenopus laevis; Rana temporaria, and
Rana Escuelenta) it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these two classes of pyrethroids has been made
on the basis of the symptoms of toxicity in mammals and insects
(Van den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge,
1980; Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group (allethrin, d-
phenothrin, permethrin, tetramethrin, cismethrin, and bioresmethrin)
(Type I: T-syndrome).
The pyrethroids that do not contain an alpha-cyano group give
rise to pronouced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3-10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organs and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5°C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of the "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 milliseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently, it was demonstrated that allethrin and DDT have
essentially the same effect on sodium channels in frog myelinated
nerve membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittently, cause large
depolarizing after-potentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl alcohol
(deltamethrin, cypermethrin, fenvalerate, and fenpropanate) (Type II:
CS-syndrome).
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependant depression of the
nervous impulse, brought about by a progressive depolarization
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, persumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
The results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
crayfish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered
pyrethroids closely approximates that of the convulsant picrotoxin
(PTX). Deltamethrin inhibits the binding of the [3H]-
dihydropicrotoxin to rat brain synaptic membranes, whereas the non-
toxic R epimer of deltamethrin is inactive. These findings suggest
a possible relation between the Type II pyrethroid action and the
GABA receptor complex. The stereospecific correlation between the
toxicity of Type II pyrethroids and their potency to inhibit the
[35S]-TBPS binding was established using a radioligand, [35S]= t-
butylbicyclophosphorothionate [35S]-TBPS. Studies with 37
pyrethroids revealed an absolute correlation, without any false
positive or negative, between mouse intracerebral toxicity and in
vitro inhibition: all toxic cyano compounds including deltamethrin,
1R, cis-cypermethrin, 1R, trans-cypermethrin, and [2S,alpha]-
fenvalerate were inhibitors, but their non-toxic stereoisomers were
not; non-cyano pyrethroids were much less potent or were inactive
(Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S,alphaS]-fenvalerate) increased the input resistance of crayfish
claw opener muscle fibres bathed in GABA. In contrast, two non-
insecticidal stereoisomers and Type I pyrethroids (permethrin,
resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis ). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action. It
may be related to the long-lasting repetitive activity in sense
organs and possibly in other parts of the nervous system, which, in
a more advance state of poisoning, may be accompanied by a
frequency-dependent depressoin of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vunerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
RESUME, EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1. Résumé et évaluation
1.1 Identité, propriétés physiques et chimiques, et méthodes
d'analyse
La phénothrine racémique a été synthétisée pour la première
fois en 1969. Du point de vue chimique, il s'agit de l'ester de
l'acide diméthyl-2,2 (diméthyl-2,2 vinyl)-3 cyclopropanecarboxylique
et de l'alcool phénoxy-3 benzylique (PBalc). Il se présente sous
la forme d'un mélange de quatre stéréoisomères : [1R,trans],
[1R,cis], [1S,trans], [1S,cis]. La d-phénothrine est un mélange
d'une partie d'isomère [1R,cis] pour quatre parties d'isomère
[1R,trans], et elle est à l'heure actuelle le seul produit
technique sur le marché. C'est l'isomère [1R,trans] qui est
l'insecticide le plus actif; vient ensuite l'isomère [1R,cis].
La d-phénothrine de qualité technique se présente sous la forme
d'un liquide jaune pâle à brun jaune et son degré de pureté est de
92,5 à 94,5%. Sa densité est de 1,058-1,061 à 25°C et sa tension
de vapeur de 0,16 mPa à 20°C. Elle est difficilement soluble dans
l'eau (2 mg par litre à 25°C) mais soluble dans les solvants
organiques tels que l'acétone, le xylène et l'hexane. Elle est
assez stable à l'air mais instable à la lumière, encore que sa
photodégradation ne soit pas aussi rapide que celle des pyréthrines
naturelles. Elle est instable en milieu alcalin.
Le dosage des résidus peut s'effectuer par chromatographie en
phase liquide à haute performance avec détecteur ultra-violet, la
concentration minimale décelable étant de 0,05 mg par kg. Pour
l'analyse du produit technique on utilise la chromatographie en
phase gazeuse avec détection par ionisation de flamme.
1.2 Production et usage
La d-phénothrine est utilisée depuis 1977. On estime que l'on
utilise chaque année 70 à 80 tonnes de d-phénothrine dans le monde,
essentiellement pour détruire les insectes incommodants dans les
habitations, lutter contre les vecteurs de maladies et protéger les
céréales ensilées; le produit est utilisé seul ou en association
avec d'autres insecticides ou synergisants. Il est présenté sous
forme d'huiles pour aérosols, de poudres ou de concentrés
émulsionnables. La d-phénothrine est également utilisée pour
détruire les poux de l'homme, auquel cas elle est présentée sous
forme de poudre, de shampooing ou de lotion.
1.3 Exposition humaine
Les aérosols classiques à usage domestique ne devraient pas
conduire à des concentrations atmosphériques de d-phénothrine
supérieures à 0,5 mg/m3. Dans le blé ensilé, on peut trouver des
résidus allant jusqu'à 4 mg/kg, mais ces teneurs tombent à 0,8
mg/kg dans la farine après mouture, et à 0,6 mg/kg après
panification.
Pour détruire les poux, on applique la d-phénothrine sur la
chevelure, par exemple en trois doses de 32 mg tous les trois
jours. Il n'existe aucune donnée sur l'exposition professionnelle à
la d-phénothrine.
L'exposition de la population dans son ensemble devrait être
très faible, mais on manque de données précises sur ce point.
1.4 Destinée dans l'environnement
La phénothrine se dégrade facilement, sa demi-vie étant
inférieure à un jour sur les végétaux et autres surfaces. La d-
phénothrine ou ses produits de dégradation ne migrent que très peu
vers les zones non traitées des végétaux. On a constaté que des
haricots ne captaient que dans une faible mesure les prouits
radiomarqués provenant de sols traités par de la phénothrine
marquée au carbone-14. Après traitement des sols avec de la
[1R, trans ]- ou de la [1R, cis]-phénothrine à raison de 1 mg/kg, on
a constaté que les deux isomères se décomposaient rapidement, la
demi-vie initiale étant de un à deux jours, mais que, en cas
d'inondation, la dégradation était considérablement ralentie, la
demi-vie initiale étant de deux à quatre semaines pour l'isomère
trans et de un à deux mois pour l'isomère cis. On a observé que
les isomères trans ou cis de la phénothrine se déplaçaient
relativement peu (environ 2%) à travers des colonnes de terre,
lorsque le lessivage commençait immédiatement ou deux semaines
après le traitement.
En général, la dégradation qui se produit dans l'environnement
conduit à des produits moins toxiques.
1.5 Cinétique et métabolisme
Après avoir reçu une dose unique ou des doses répétées de
phénothrine radiomarquée par voie orale ou percutanée, des rats ont
rapidement et presque complètement excrété la fraction marquée dans
leurs urines et leurs déjections en trois à sept jours. Les
principales voies métaboliques des isomères cis et trans chez le
rat consistent en une rupture de la liaison ester et l'oxydation en
position 4 du reste alcool ou du groupement isobutényle du reste
acide. Les métabolites résultant du clivage de l'ester (qui sont
excrétées essentiellement dans les urines) constituent les
principaux produits de dégradation de l'isomère trans alors que les
métabolites restant sous forme d'ester (excrétées essentiellement
dans les déjections) proviennent pour la plupart de l'isomère cis.
1.6 Effets sur les êtres vivant dans leur milieu naturel
La phénothrine a été expérimentée sur quelques groupes
d'organismes non visés et dans chaque groupe sur quelques espèces
seulement. Chez les poissons, la CL50 à 96 heures de la
phénothrine racémique et des stéréoisomères (1R) va de 17 à 200
microgrammes par litre. Une étude, portant sur des invertébrés
aquatiques, a montré que chez Daphnia pulex la CL50 à 3 heures
était de 25 à 50 mg/litre pour tous les isomères et la phénothrine
racémique.
Une seule et unique étude au cours de laquelle de la
phénothrine a été appliquée à des étangs n'a révélé aucun effet sur
les arthropodes aquatiques.
La toxicité pour les oiseaux est faible, avec une DL50 aiguë
par voie orale supérieure à 2500 mg/kg de poids corporel chez le
colin de Virginie et une CL50 par voie alimentaire supérieure à
5000 mg/kg de nourriture chez ce volatile et chez le colvert.
Etant donné que la phénothrine se dégrade rapidement à la
lumière solaire et qu'on l'utilise principalement pour traiter des
céréales ensilées, l'exposition environnementale est
vraisemblablement très faible. Dans ces conditions, des effets sur
l'environnement sont tout-à-fait improbables.
1.7 Effets sur les animaux d'expérience et sur les systèmes
d'épreuve in vitro
La toxicité aiguë de la d-phénothrine est extrêmement faible,
la DL50 étant supérieure à 5000 mg/kg de poids corporel chez le rat
et la souris (par voie orale, sous-cutanée, dermique et
intrapéritonéale) et la CL50 inhalatoire supérieure à 3760 mg/m3
chez le rat. Le syndrome d'intoxication se caractérise par une
hyperexcitabilité, une prostration, des tremblements, de l'ataxie
et une paralysie. Sur la base de ces symptômes et d'après les
résultats des études électrophysiologiques sur les nerfs sensoriels
des cerques de la blatte, la phénothrine est classée parmi les
pyréthroides du type I.
Exposés à de la d-phénothrine par inhalation à des
concentrations allant jusqu'à 210 mg/m3, quatre heures par jour
pendant quatre semaines, ou par voie orale cinq jours de suite à
raison de 5000 mg/kg de poids corporel, des rats n'ont présenté
aucun effet toxicologique indésirable.
Plusieurs études d'alimentation ont été effectuées sur des rats
et des souris recevant de la d-phénothrine ou de la phénothrine
racémique à des doses allant de 200 à 10 000 mg par kg de
nourriture; la période d'exposition allait de six mois à deux ans.
Ces études ont permis d'établir une dose sans effet observable
allant de 300 à 1000 mg par kg de nourriture, ce qui correspond à
peu près à 40-160 mg/kg de poids corporel et par jour. Chez des
chiens à qui l'on avait administré de la d-phénothrine à des doses
de 100 à 3000 mg/kg de nourriture avec des périodes d'exposition de
26 à 52 semaines, deux études ont permis d'obtenir une dose sans
effet observable de 300 mg par kg de nourriture, soit 7 à 8 mg/kg
de poids corporel et par jour.
Divers systèmes permettant d'étudier in vivo et in vitro les
mutations géniques, les lésions et les réparations de l'ADN ainsi
que les effets chromosomiques, ont permis de constater que la d-
phénothrine n'avait pas d'effet mutagène.
Des études de deux ans ont également montré que la d-
phénothrine n'était pas cancérogène pour le rat ni la souris à des
doses allant jusqu'à 3000 mg par kg de nourriture. Aucune
tératogénicité ni embryotoxicité n'a été observée chez des foetus
de lapins et de souris dont les mères avaient reçu de la d-
phénotrine à des doses allant jusqu'à 1000 et 3000 mg par kg de
poids corporel, respectivement. Lors d'une étude de reproduction
chez le rat, portant sur deux générations, on a établi que la dose
sans effet observable était de 1000 mg par kg de nourriture.
Des rats exposés par inhalation à des doses très élevées de d-
phénothrine (jusqu'à 3760 mg/m3) pendant quatre heures ou,
quotidiennement, par voie orale, à une dose de 5000 mg par kg de
poids corporel, cinq jours durant, n'ont présenté aucune
dégénérescence myélinique ni désagrégation de l'axone au niveau du
nerf sciatique.
1.8 Effets sur les êtres humains
La d-phénothrine est utilisée depuis plus de 10 ans sans que
l'on ait signalé d'intoxication humaine.
Rien n'indique que cette substance puisse avoir des effets
nocifs sur l'homme pour peu qu'elle soit utilisée conformément aux
recommandations.
2. Conclusions
2.1 Population générale
L'exposition de la population dans son ensemble à la d-
phénothrine est vraisemblablement très faible et il n'y a
probablement aucun risque à cet égard si le produit est utilisé
conformément aux recommandations.
2.2 Exposition professionnelle
Si elle est utilisée de manière convenable, moyennant un
certain nombre de mesures d'hygiène et de sécurité, la d-
phénothrine ne devrait pas présenter de risque pour les personnes
qui lui sont exposées de par leur profession.
2.3. Environnement
Du fait qu'elle se dégrade rapidement à la lumière solaire et
qu'elle est principalement utilisée pour traiter les céréales
ensilées, l'exposition environnementale à la phénothrine est
vraisemblablement très faible. Dans ces conditions tout effet sur
l'environnement est extrêmement improbable.
3. Recommandations
Les niveaux d'exposition résultant d'une utilisation conforme
aux recommandations sont censés être extrêmement faibles, toutefois
on pourrait envisager de confirmer cette hypothèse en étendant la
surveillance à la d-phénothrine.
 Racemic phenothrin was first synthesized by Itaya et al.
(1969). It is prepared by esterifying (1RS, cis,trans )-2,2-
dimethyl-3-(2,2-dimethylvinyl)cyclopropanecarboxylic acid
(chrysanthemic acid) with 3-phenoxybenzyl alcohol (Fujimoto et al.,
1973). Phenothrin is thus a mixture of four stereoisomers (Fig. 1).
The cis:trans isomer ratio is 1:4 and the optical ratio of 1R:1S is
1:1 (racemic). Thus the isomers 1, 2, 3, and 4 are present in the
approximate ratio of 4:1:4:1 (Table 1). d-Phenothrin is the
(1R,cis,trans) preparation (i.e., a mixture of isomers 2 and 1),
the cis:trans ratio being 1:4. The technical grade is 92.5-94.5%
pure. The major impurities found in seven d-phenothrin
preparations (average purity, 94.0%) were ethyl chrysanthemate
(2.31%), 3-phenoxy-6-bromobenzyl cis,trans-chrysanthemate (0.66%),
3-phenoxytoluene (0.43%), and 4-phenoxybenzyl cis,trans-
chrysanthemate (0.39%) (Miyamoto et al., 1984).
Racemic phenothrin was first synthesized by Itaya et al.
(1969). It is prepared by esterifying (1RS, cis,trans )-2,2-
dimethyl-3-(2,2-dimethylvinyl)cyclopropanecarboxylic acid
(chrysanthemic acid) with 3-phenoxybenzyl alcohol (Fujimoto et al.,
1973). Phenothrin is thus a mixture of four stereoisomers (Fig. 1).
The cis:trans isomer ratio is 1:4 and the optical ratio of 1R:1S is
1:1 (racemic). Thus the isomers 1, 2, 3, and 4 are present in the
approximate ratio of 4:1:4:1 (Table 1). d-Phenothrin is the
(1R,cis,trans) preparation (i.e., a mixture of isomers 2 and 1),
the cis:trans ratio being 1:4. The technical grade is 92.5-94.5%
pure. The major impurities found in seven d-phenothrin
preparations (average purity, 94.0%) were ethyl chrysanthemate
(2.31%), 3-phenoxy-6-bromobenzyl cis,trans-chrysanthemate (0.66%),
3-phenoxytoluene (0.43%), and 4-phenoxybenzyl cis,trans-
chrysanthemate (0.39%) (Miyamoto et al., 1984).
 Table 1. Chemical identity of racemic phenothrin and d-phenothrin
--------------------------------------------------------------------------------------------
Common name/ CAS Index name (9CI)/ Stereoisomeric Synonyms and
CAS Registry no./ Stereospecific nameb,c compositiond trade names
RTECS Registry no.a
--------------------------------------------------------------------------------------------
Phenothrin (racemic) Cyclopropanecarboxylic acid, (1):(2):(3):(4) Phenoxythrin,
Table 1. Chemical identity of racemic phenothrin and d-phenothrin
--------------------------------------------------------------------------------------------
Common name/ CAS Index name (9CI)/ Stereoisomeric Synonyms and
CAS Registry no./ Stereospecific nameb,c compositiond trade names
RTECS Registry no.a
--------------------------------------------------------------------------------------------
Phenothrin (racemic) Cyclopropanecarboxylic acid, (1):(2):(3):(4) Phenoxythrin,
 4.1. Transport and Distribution between Media
The degree of leaching of [1RS, trans]- or [1RS, cis]-phenothrin
has been studied under laboratory conditions. Very little movement
(less than 2%) of trans- and cis-phenothrin through soil columns
occurred when leaching was started either immediately or 14 days
after treatment (Nambu et al., 1980).
4.2. Photodegradationa
Although d-phenothrin is more resistant to photolysis than
pyrethrin I, allethrin, and resmethrin (due to a more stable
alcohol moiety), it still possesses the photo-labile isobutenyl
group in the molecule and therefore is easily photodecomposed.
Ruzo et al. (1982) investigated the photodegradation of
[1RS, trans]-phenothrin (5)a in oxygenated benzene solution (10-3
mol/litre) under UV light (360 nm) or in a thin film (0.1-0.3 mg/cm2)
under sunlight (Fig. 2). Exposure to sunlight resulted in 30%
conversion, the major photoproducts being:
* the (1RS) epoxides (21) (22% of the reaction mixture), the
alcohol (22) (9%), and the aldehyde (23) (13%) derivatives from
oxidation at the (E)-methyl group;
* the caronaldehyde derivative (7) (4%) from ozonolysis;
* the hydroperoxide (25) (27%) from hydroperoxidation, including
migration of the double bond at the 1'-position of the
isobutenyl moiety (Fig. 2).
Minor products (26) and (27) (3%) resulted from further oxidation
of the hydroperoxide (25) and from cis/trans isomerization and
ester cleavage. Trace amounts of trans-chrysanthemic acid (20)
were detected. 3-Phenoxybenzyl alcohol (12) underwent further
oxidation to form the aldehyde (13) and carboxylic acid (16).
Unidentified photoproducts accounted for 16.6% of the total. A
similar product distribution was obtained in benzene solution.
4.3. Degradation in Plants and Soils
In studies by Nambu et al. (1980), 14C-methylene-labelled
[1R, trans]-or [1R, cis ]-phenothrin (each 10 mg/kg) disappeared from
the treated leaves of kidney bean or rice plants with half-lives of
less than one day under greenhouse conditions. The residue levels
in both plants were 0.04-0.28 mg/kg for trans- phenothrin and 0.10-
0.30 mg/kg for cis- phenothrin after 30 days, compared with
approximately 10 mg/kg immediately after treatment. Both isomers
primarily underwent ozonolysis at the isobutenyl double bond,
probably via the photochemical reactions indicated previously
(Fig. 2). The resultant ozonides (6) were detected soon after
treatment but they were rapidly decomposed to the corresponding
aldehyde (7) and carboxylic acid (8) (3-phenoxy-benzyl-2,2-
dimethyl-3-carboxy-cyclopropanecarboxylate) derivatives. Cleavage
of the ester linkage also occurred, together with hydroxylation at
the 2'-position (10) or 4'-position (9) of the alcohol moiety.
Conjugation of the acids and alcohols with sugars was also
observed, and the formation of polar products was more extensive in
rice than in bean plants. There was little translocation of trans-
------------------------------------------------------------------------
a The numbers in brackets following a chemical name refer to the
numbers given in Fig. 2.
or cis-phenothrin or of its degradation products to the untreated
parts of the plants. Limited uptake of radiolabelled products into
bean plants took place from light clay, sandy loam soil, and from
sand treated with 14C-methylene-labelled trans- or cis-phenothrin.
The degradation of 14C-labelled [1R, trans]- or [1R, cis]-
phenothrin in two soils was investigated by Nambu et al. (1980).
Both isomers decomposed rapidly under upland conditions, with
initial half-lives of 1-2 days, but under flooded conditions the
degradation was much slower, with initial half-lives of 2-4 weeks
and 1-2 months for the trans and cis isomers, respectively.
Analysis of the soil extracts revealed unchanged parent isomers,
(16), 3-(4'-hydroxyphenoxy)benzoic acid (17), (12), 3-(4'-
hydroxyphenoxy)benzyl alcohol (14), (9), and 3-hydroxy-benzyl-2,2-
dimethyl-3-(2,2-dimethylvinyl)-cyclopropanecarboxylate (11). These
degradation products were not persistent and underwent further
degradation in soil, under both upland and flooded conditions, to
yield large amounts of 14CO2 and unextractably bound residues.
More 14C carbon dioxide was formed in soils under upland conditions
than under flooded conditions and more was formed from trans-
phenothrin than from the cis isomer. Bound 14C residues were
associated mainly with the humic acid and fulvic acid fractions of
soil organic matter. The fulvic acid fraction contained small
amounts of the same degradation products as in the soil extracts.
4.4. Degradation on Stored Foods
When 14C-[1R, trans ]- or 14C-[1R, cis ]-phenothrin was applied at
4 mg/kg to wheat grains of 11% moisture content and the crop was
stored at 15 or 30°C in the dark for 12 months, most of the
phenothrin remained intact. Major metabolites were formed by
hydrolysis of the ester linkage and oxidation of 3-phenoxybenzyl
alcohol to 3-phenoxybenzoic acid. The methyl ester of 3-
phenoxybenzoic acid was also produced. After storage at 30°C for
12 months, these metabolites amounted to 13.9% in the case of the
trans isomer and 6.3% in the case of the cis isomer (Nambu et al.,
1981).
5. KINETICS AND METABOLISM
Appraisal
The pathways by which phenothrin is metabolized in mammals are
summarized in Fig. 3.
Comparative metabolism studies have shown that the metabolism
of racemic phenothrin is similar to that of [1R,cis,trans]-phenothrin
(d-phenothrin).
No information is available on the in vivo metabolism of the acid
moieties of trans([1R,trans])- or cis([1R,cis])-phenothrin. However,
the acid moiety liberated in vivo (chrysanthemic acid) is the same
as that of resmethrin and tetramethrin and, therefore, its fate can
be predicted from the resmethrin and tetramethrin data.
4.1. Transport and Distribution between Media
The degree of leaching of [1RS, trans]- or [1RS, cis]-phenothrin
has been studied under laboratory conditions. Very little movement
(less than 2%) of trans- and cis-phenothrin through soil columns
occurred when leaching was started either immediately or 14 days
after treatment (Nambu et al., 1980).
4.2. Photodegradationa
Although d-phenothrin is more resistant to photolysis than
pyrethrin I, allethrin, and resmethrin (due to a more stable
alcohol moiety), it still possesses the photo-labile isobutenyl
group in the molecule and therefore is easily photodecomposed.
Ruzo et al. (1982) investigated the photodegradation of
[1RS, trans]-phenothrin (5)a in oxygenated benzene solution (10-3
mol/litre) under UV light (360 nm) or in a thin film (0.1-0.3 mg/cm2)
under sunlight (Fig. 2). Exposure to sunlight resulted in 30%
conversion, the major photoproducts being:
* the (1RS) epoxides (21) (22% of the reaction mixture), the
alcohol (22) (9%), and the aldehyde (23) (13%) derivatives from
oxidation at the (E)-methyl group;
* the caronaldehyde derivative (7) (4%) from ozonolysis;
* the hydroperoxide (25) (27%) from hydroperoxidation, including
migration of the double bond at the 1'-position of the
isobutenyl moiety (Fig. 2).
Minor products (26) and (27) (3%) resulted from further oxidation
of the hydroperoxide (25) and from cis/trans isomerization and
ester cleavage. Trace amounts of trans-chrysanthemic acid (20)
were detected. 3-Phenoxybenzyl alcohol (12) underwent further
oxidation to form the aldehyde (13) and carboxylic acid (16).
Unidentified photoproducts accounted for 16.6% of the total. A
similar product distribution was obtained in benzene solution.
4.3. Degradation in Plants and Soils
In studies by Nambu et al. (1980), 14C-methylene-labelled
[1R, trans]-or [1R, cis ]-phenothrin (each 10 mg/kg) disappeared from
the treated leaves of kidney bean or rice plants with half-lives of
less than one day under greenhouse conditions. The residue levels
in both plants were 0.04-0.28 mg/kg for trans- phenothrin and 0.10-
0.30 mg/kg for cis- phenothrin after 30 days, compared with
approximately 10 mg/kg immediately after treatment. Both isomers
primarily underwent ozonolysis at the isobutenyl double bond,
probably via the photochemical reactions indicated previously
(Fig. 2). The resultant ozonides (6) were detected soon after
treatment but they were rapidly decomposed to the corresponding
aldehyde (7) and carboxylic acid (8) (3-phenoxy-benzyl-2,2-
dimethyl-3-carboxy-cyclopropanecarboxylate) derivatives. Cleavage
of the ester linkage also occurred, together with hydroxylation at
the 2'-position (10) or 4'-position (9) of the alcohol moiety.
Conjugation of the acids and alcohols with sugars was also
observed, and the formation of polar products was more extensive in
rice than in bean plants. There was little translocation of trans-
------------------------------------------------------------------------
a The numbers in brackets following a chemical name refer to the
numbers given in Fig. 2.
or cis-phenothrin or of its degradation products to the untreated
parts of the plants. Limited uptake of radiolabelled products into
bean plants took place from light clay, sandy loam soil, and from
sand treated with 14C-methylene-labelled trans- or cis-phenothrin.
The degradation of 14C-labelled [1R, trans]- or [1R, cis]-
phenothrin in two soils was investigated by Nambu et al. (1980).
Both isomers decomposed rapidly under upland conditions, with
initial half-lives of 1-2 days, but under flooded conditions the
degradation was much slower, with initial half-lives of 2-4 weeks
and 1-2 months for the trans and cis isomers, respectively.
Analysis of the soil extracts revealed unchanged parent isomers,
(16), 3-(4'-hydroxyphenoxy)benzoic acid (17), (12), 3-(4'-
hydroxyphenoxy)benzyl alcohol (14), (9), and 3-hydroxy-benzyl-2,2-
dimethyl-3-(2,2-dimethylvinyl)-cyclopropanecarboxylate (11). These
degradation products were not persistent and underwent further
degradation in soil, under both upland and flooded conditions, to
yield large amounts of 14CO2 and unextractably bound residues.
More 14C carbon dioxide was formed in soils under upland conditions
than under flooded conditions and more was formed from trans-
phenothrin than from the cis isomer. Bound 14C residues were
associated mainly with the humic acid and fulvic acid fractions of
soil organic matter. The fulvic acid fraction contained small
amounts of the same degradation products as in the soil extracts.
4.4. Degradation on Stored Foods
When 14C-[1R, trans ]- or 14C-[1R, cis ]-phenothrin was applied at
4 mg/kg to wheat grains of 11% moisture content and the crop was
stored at 15 or 30°C in the dark for 12 months, most of the
phenothrin remained intact. Major metabolites were formed by
hydrolysis of the ester linkage and oxidation of 3-phenoxybenzyl
alcohol to 3-phenoxybenzoic acid. The methyl ester of 3-
phenoxybenzoic acid was also produced. After storage at 30°C for
12 months, these metabolites amounted to 13.9% in the case of the
trans isomer and 6.3% in the case of the cis isomer (Nambu et al.,
1981).
5. KINETICS AND METABOLISM
Appraisal
The pathways by which phenothrin is metabolized in mammals are
summarized in Fig. 3.
Comparative metabolism studies have shown that the metabolism
of racemic phenothrin is similar to that of [1R,cis,trans]-phenothrin
(d-phenothrin).
No information is available on the in vivo metabolism of the acid
moieties of trans([1R,trans])- or cis([1R,cis])-phenothrin. However,
the acid moiety liberated in vivo (chrysanthemic acid) is the same
as that of resmethrin and tetramethrin and, therefore, its fate can
be predicted from the resmethrin and tetramethrin data.
 5.1. Metabolism in Mammals
Appraisal
After rats are treated with radiolabelled phenothrin, either by
single or repeated oral exposure, or dermally, the radioactivity is
rapidly (and almost totally) excreted into urine and faeces within
3 to 7 days. The major metabolic pathways of both trans- and cis-
phenothrin in rats are oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety and cleavage of
the ester linkage (see Fig. 3). Cleavage of the ester bond is more
difficult in the cis isomer than in the trans isomer. Thus, ester-
form metabolites oxidized in the various positions of the molecule are
the major metabolites of the cis isomer and are excreted mostly in the
faeces. However, ester-cleaved metabolites are the major products from
the trans isomer and are mostly excreted in urine.
When [1R, trans ]-phenothrin labelled with 14C at the methylene
moiety was given as a single oral dose (200 mg/kg body weight) to
Sprague Dawley male rats, the radiocarbon was rapidly eliminated,
57% and 43% being recovered within 3 days in urine and faeces,
respectively (Miyamoto et al., 1974). There was no detectable
radiocarbon in the expired air. Similarly, Sprague Dawley male
rats given a single oral dose (10 mg/kg body weight) of 14C-
[1R, trans ]-phenothrin, excreted the radiocarbon rapidly in the
urine (75%) and faeces (21%) (Kaneko et al., 1981).
In a study by Isobe et al. (1987), Sprague Dawley male and
female rats were given a single oral administration of 14C-
[1R, trans ]-phenothrin in corn oil at 4 and 200 mg/kg body weight.
Within 7 days the radiocarbon was almost completely eliminated in
the urine and faeces. The % elimination was as follows:
-----------------------------------------
Dose Urine Faeces
(mg/kg) Males Females Males Females
4 38 40 61 60
200 39 25 56 60
-----------------------------------------
Sprague Dawley male rats eliminated 65% of the dosed radiocarbon
in the faeces over 3 days after a single oral administration (200
mg/kg body weight) of 14C-[1R, cis ]-phenothrin (28) (Suzuki et al.,
1976), and 22 and 74% of the dose into urine and faeces,
respectively, 7 days after a single oral dose of 10 mg/kg body
weight (Kaneko et al., 1981).
When 14C-[1R, cis ]-phenothrin in corn oil was administered once
orally to Sprague Dawley male and female rats at 4 or 200 mg/kg
body weight, the radiocarbon was excreted into the urine (11-18%)
and faeces (81-87%) within 7 days. Similarly, when Sprague Dawley
rats were treated repeatedly with 14C-[1R,trans] or 14C-[1R,cis]
isomers at 4 mg/kg body weight per day for 14 days, the radiocarbon
was rapidly and almost completely excreted: 75-70% in urine and 24-
29% in faeces for the trans isomer, and 24% in urine and 72-73% in
faeces for the cis isomer (Isobe et. al., 1987).
The tissue residues in rats 7 days after a single oral dose of
14C-[1R, cis ]- or 14C-[1R, trans ]-phenothrin at 10 mg/kg body
weight were generally very low although the fat showed somewhat
higher residue levels (1-2.5 mg/kg) (Kaneko et al., 1981).
Similarly, high 14C residue levels (up to 23 mg/kg) were found in
the fat, 7 days after a single oral dose of the [1R,cis] isomer at
200 mg/kg body weight (Isobe et al., 1987).
The major metabolite of the trans isomer, when given as a
single oral dose of 200 mg/kg to rats, was 3-(4'-hydroxyphenoxy)-
benzoic acid (17) (4'OH-PBacid, 54%). There were smaller amounts
of 3-phenoxybenzoic acid (16) (PBacid, 9.5%) and its glycine
conjugate (Miyamoto et al., 1974).
When [1R, trans ]-phenothrin was given to rats at 4, 10, or 200
mg/kg body weight (oral single dose) or 4 mg/kg body weight
(repetitive oral dose for 14 days), the sulfate conjugate of 4'-OH-
PBacid was predominant, accounting for 28, 43, 28, and 55%,
respectively, of the dose. In addition, PBacid (4, 10, 5, and 6%),
its glycine conjugate (1,3,2, and 2%) and glucuronide (2,3,1, and
3%), and free 4'-OH-PBacid (2,11,3, and 3%) were found. The
sulfate conjugate of 3-(2'-hydroxyphenoxy)benzoic acid (18) (2'-
OH-PBacid) was also found as a minor metabolite (Kaneko et al.,
1981; Isobe et al., 1987).
When rats were given 14C-[1R, trans ]-phenothrin (10 mg/kg body
weight), the unmetabolized compound and two ester-form metabolites
were detected in their faeces in small amounts (0.4-1.2%), which
had hydroxymethyl (29) ( wt-alc- t-phe) or carboxyl group (30) wt-
acid- t-phe) (see Fig. 2) at the position of the trans methyl group
of chrysanthemic acid (Kaneko et al., 1981).
When Sprague Dawley rats were administered a single oral dose
of [1R, trans ]-phenothrin at 4 or 200 mg/kg body weight level or
given an oral dose of 4 mg/kg body weight per day for 14 days,
unmetabolized compound was found in the faeces (44-45, 44-60, and
14-16% of the dose, respectively). An ester-form metabolite, the
4'-hydroxy w(t)-acid derivative of trans- phenothrin, was also
detected (0.4-0.6%) (Isobe et al., 1987).
When male Sprague Dawley rats were given cis-phenothrin (200
mg/kg body weight), three ester-form metabolites, which accounted
for 14% of the dosed radioactivity, were found in the faeces.
These were 4'-hydroxy- cis-phenothrin (31) (4-OH- c-phe), an ester-
form derivative with the trans methyl of the isobutenyl group being
oxidized to carboxyl group (32) ( wt-acid- c-phe), and a compound
with the geminal-dimethyl groups oxidized (2-OH-) in addition to
both of the above modifications (33) (4'-OH, wt-acid, 2-OH(t)- c-phe)
(Suzuki et al., 1976).
In addition to this, the cis isomer gave rise to nine ester-
form metabolites in the faeces varying in amounts from 2% (4'-OH,
wc-alc-c-phe (34)) to 13% (4'-OH, wt-acid,2-OH(t)- c-phe (33)) of
the dosed radiocarbon after single oral administration. These
ester-form metabolites were transformed by oxidation reactions at
any of the following positions: 4'-position of the phenoxy group,
the trans or cis methyl of the isobutenyl groups, and the trans
methyl of the geminal-dimethyl group (Kaneko et al., 1981).
When Sprague Dawley rats were given a single oral dose of
[1R, cis ]-phenothrin at 4 or 200 mg/kg body weight level or an oral
dose of [1R, cis ]-phenothrin at 4 mg/kg body weight per day for 14
days, ester-form metabolites (1-9% of the dosed radioactivity) were
found, in addition to unmetabolized compound (17-59% of the dose).
The urine contained 4'-OH-PBacid as a sulfate conjugate (7-18%) and
in the free form (0.3-1%), and PBacid as glycine or glucuronide
conjugates and in the free form (0.3-1%) (Isobe et al., 1987).
Following the dermal treatment of male Sprague Dawley rats with
dust or emulsifiable concentrates (E.C.) of either 14C-[1R, trans ]-
or 14C-[1R, cis ]-phenothrin at 10 mg/kg body weight, the 14C
absorption into the body was estimated to be 3-7% of the initial
dose with dust and 8-17% with the E.C. After both dust and E.C.
treatments, the radiocarbon excreta (as a percentage of the initial
dose) recovered in the urine was 2.6-8.7% for the trans isomer, and
1.5-4.8% for the cis isomer, and in the faeces was 0.6-2.2% for the
trans isomer, and 3.0-12.3% for the cis isomer. Since the same
metabolites are formed following either oral exposure or dermal
treatment, it appears that both phenothrin isomers undergo the same
metabolism once in the systemic circulation, regardless of the
route of administration (Kaneko et al., 1981; Isobe et al., 1987).
Information concerning the comparative metabolism of racemic
(1RS) phenothrin and its d-isomer (1R) was obtained through a study
of CD rats and ddY mice given a single oral dose of either
[1R, trans ]-, [1S, trans ]-, [1RS, trans ]-, [1R, cis ]-, [1S, cis ]-,
or [1RS, cis ]-phenothrin. The radiocarbon derived from each isomer
was almost completely eliminated from the rats and mice within six
days after dosing. The trans isomers were mainly eliminated in the
urine (rat, 85-88%; mice, 65-75%) and the cis isomers mainly in the
faeces (rat, 57-71%; mice, 54-71%). The amounts of 14C in the
urine and faeces of rats and mice treated with the [1R,trans] and
[1R,cis] isomers did not differ significantly from those
corresponding to the [1RS,trans] and [1RS,cis] isomers, respectively.
The 14C tissue residues were very low, except in the fat. There
were no striking differences in 14C levels among the three trans
isomers and the three cis isomers. The 14C levels of the cis
isomers in fat (maximum 3.5 mg/kg) were three to seven times higher
than those of the trans isomers (less than 1 mg/ kg). The major
urinary and faecal metabolites were remarkably similar in both rats
and mice. In both rats and mice, there were virtually no
differences in the metabolic fate of the [1R,trans] and [1RS,
trans] isomers or of the [1R,cis] and [1RS,cis] isomers (Izumi et
al., 1984).
5.2. Enzymatic Systems for Biotransformation
In studies by Miyamoto et al. (1974), [IR, trans ]-phenothrin
(1 mmol/litre) was incubated with the 8000-g supernatant from a
liver homogenate of rats, mice, guinea-pigs, rabbits, or dogs at
37°C for 60 min in the absence of NADPH. The supernatant from the
guinea-pig was the most active in degrading [1R, trans ]-phenothrin,
followed by that of dog, rabbit, rat, and mouse. The major
metabolite in all the mammalian species tested was 3-phenoxybenzyl
alcohol (12) (PBalc). Smaller amounts of PBacid (16) and trace
amounts of 4'-OH-PBacid (17) were also found. However, in the
presence of NADPH, the amounts of PBacid and unidentified ether-
soluble metabolites increased in all species except dog. In
contrast to [1R, trans ]-phenothrin, [1R, cis ]-phenothrin was hardly
metabolized at all by the rat liver preparation in the absence of
NADPH. NADPH enhanced the degradation rate of the cis isomer,
leading to the formation of unidentified metabolites, while ester-
cleaved metabolites such as PBacid (16), PBalc (12) and 4'-OH-
PBacid (17) were found in very small amounts. When [1R, trans ]-,
[1R, cis ]-, [1S, trans ]-, and [1S, cis ]-phenothrin were incubated
with rat liver microsomes at 37.5°C for 30-60 min to estimate Km
and Vmax using a Lineweaver-Burk plot, the values for Km (0.11-0.17
mmol/litre) were similar for the four isomers, whereas the values
for Vmax were different; both the trans isomers yielded values for
Vmax 20-30 times larger than did the cis isomers (Miyamoto et al.,
1974).
In studies by Suzuki & Miyamoto (1978), pyrethroid
carboxyesterase(s) that hydrolyze esters of chrysanthemic acid
were purified from rat liver microsomes by cholic acid
solubilization, ammonium sulfate fractionation, heat treatment, and
DEAE-Sephadex A-50 column chromatography. The 45-fold-purified
enzyme (38% yield) probably consisted of a single protein with a
relative molecular mass of approximately 74 000, a Km of 0.21
mmol/litre for [1R, trans ]-phenothrin, and an optimum pH of 7-9.
It was susceptible to inhibition by organophosphate and carbamate
insecticides and insensitive to PCMB ( p-chloromercurybenzoic acid),
and mercuric and cupric ions. The enzyme seemed to require neither
coenzymes nor cofactors and hydrolysed the trans isomers of several
synthetic pyrethroids (tetramethrin, resmethrin, phenothrin, and
permethrin) well, at more or less similar rates. On the other hand,
the cis isomers were hydrolysed at rates 5-10 times lower than
their trans counterparts. The purified pyrethroid carboxyesterase
was apparently identical in nature to malathion carboxyesterase
and p-nitrophenyl acetate carboxyesterase.
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
Data on the acute toxicity of racemic and isomeric phenothrin
for aquatic organisms are summarized in Table 4.
6.1. Aquatic Organisms
Racemic phenothrin yielded 96-h LC50 values of 17-200 µg/litre
for the fish species tested (Table 4). The (1S)-optical isomers
were relatively non-toxic with LC50 values of 10 000 µg/litre,
whereas the (1R)-optical isomer and racemic phenothrin were of
similar toxicity with LC50 values between 120 and 200 µg/litre for
the killifish (Oryzias latipes) (Miyamoto, 1976).
When d-phenothrin was applied to ponds at the rates of 28 or 56
g/ha to control mosquito larvae, mayfly naiads were most affected
but no other arthropods (damselfly, dragonfly naiads, ostracods, or
diving beetle larvae) were seriously affected (Mulla et al., 1980).
6.2. Terrestrial Organisms
The available toxicity data for non-target terrestrial
organisms are very limited.
Phenothrin has low toxicity (acute oral dosage) for bobwhite
quail with an LD50 >2510 mg/kg body weight (Worthing & Walker,
1987). An 8-day feeding study with d-phenothrin on mallard duck
and bobwhite quail indicated LD50 values of >5620 and >5000 mg/kg
diet, respectivelya.
----------------------------------------------------------------------
a Personal communication from J.L. Noles, Ecological Effects
Branch, Hazard Evaluation Division, US Environmental Protection
Agency, November 1987.
Table 4. The acute toxicity of racemic and isomeric phenothrin to non-target aquatic organisms
-----------------------------------------------------------------------------------------------
Species Size Parameter Toxic Stereo- Systema Temper- Refer-
concentration isomeric ature enceb
(µg/litre) composition (°C)
-----------------------------------------------------------------------------------------------
Fish
Killifish adult 48-h LC50 200 racemic S 25 1
(Oryzias latipes) adult 48-h LC50 120 (+)-trans S 25 1
adult 48-h LC50 170 (+)-cis S 25 1
adult 48-h LC50 10 000 (-)-trans S 25 1
adult 48-h LC50 10 000 (-)-cis S 25 1
Rainbow trout 96-h LC50 17 racemic 2
(Salmo gairdneri)
Bluegill 96-h LC50 18 racemic 2
(Lepomis macrochirus)
Arthropods
Daphnia pulex 3-h LC50 50 000 racemic S 25 1
3-h LC50 25 000-50 000 (+)-trans S 25 1
3-h LC50 50 000 (+)-cis S 25 1
3-h LC50 50 000 (-)-trans S 25 1
3-h LC50 50 000 (-)-cis S 25 1
-----------------------------------------------------------------------------------------------
a s = static
b 1. Miyamoto (1976) 2. Worthing & Walker (1987)
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
The 1984 Joint FAO/WHO Meeting on Pesticide Residues concluded
that the toxicological data for racemic phenothrin can be used to
support that for d-phenothrin, owing to the similarity in
metabolism and toxicity between the two compounds (FAO/WHO,
1985b).
7.1. Single and Short-Term Exposures
The acute toxicity in rats and mice is extremely low. The LD50
values were >5000 mg/kg body weight when d-phenothrin was
administered orally, subcutaneously, dermally, or by
intraperitoneal injection to male and female Sprague Dawley rats
(Segawa, 1979a) and ddY mice (Segawa, 1979b) (Table 5).
Table 5. The acute toxicity of racemic phenothrin and d-phenothrin
to rats and micea
------------------------------------------------------------------
Compound/Route LD50 (mg/kg)
Rat Mice
Male Female Male Female
------------------------------------------------------------------
Phenothrin (racemic)
Oral > 5000 > 5000 > 5000 > 5000
Subcutaneous > 5000 > 5000 > 5000 > 5000
Dermal > 5000 > 5000 > 5000 > 5000
Intraperitoneal > 5000 > 5000 > 5000 > 5000
Inhalation > 1210b > 1210b > 1210b > 1210b
d-Phenothrin
Oral > 10 000 > 10 000 > 10 000 > 10 000
Subcutaneous > 10 000 > 10 000 > 10 000 > 10 000
Dermal > 10 000 > 10 000 > 5000 > 5000
Intraperitoneal > 10 000 > 10 000 > 10 000 > 10 000
Inhalation > 3760b > 3760b > 1180b > 1180b
------------------------------------------------------------------
a From: Segawa (1976; 1979a,b) and Khoda et al. (1977; 1979b; 1980)
b These are values for 4-h LC50 (mg/m3)
There were no differences between the effects on male and
female rats in acute toxicity studies carried out by Segawa (1976).
Signs of poisoning appeared rapidly following the intravenous
administration of phenothrin. These included fibrillation, tremor,
slow respiration, salivation, lacrimation, ataxia, and paralysis.
These signs, evident within 30 min to one hour following
administration, diminished rapidly to the point where, after 24 h,
there were no signs of toxicity.
No differences in the oral toxicity between racemic phenothrin
and d-phenothrin were detected by Segawa (1976; 1979a,b) (Table 5).
The intravenous LD50 values for racemic phenothrin in ICR mice
were 470 and 600 mg/kg for males and females, respectively, while
those for d-phenothrin were 265 and 315 mg/kg (Hiromori et al., 1984).
Following a 4-h acute inhalation exposure (whole body), the
LC50 values were >1210 mg/m3 for racemic phenothrin (particle size
5 µm) with both Sprague Dawley rats and ICR mice (Kohda et al., 1979b)
and >3760 and 1180 mg/m3 for d-phenothrin with Sprague Dawley rats
and ICR mice, respectively (Kohda et al., 1977; 1979b) (see Table 5).
At the higher concentration (3760 mg/m3), the mean particle size
was considered to be 0.72 µm and the cumulative distribution of
particles having a diameter between 0.46 and 1.09 µm was 88.9%,
according to a particle-size distribution experiment (Kohda et al.,
1980).
In studies by Kohda et al. (1979b), Sprague Dawley rats (15 of
each sex) were exposed (whole body) by inhalation to racemic
phenothrin at concentrations of 0, 43, or 220 mg/m3 (4 h per day, 5
days per week) for 4 weeks. After this treatment, animals (10/sex
per group) were sacrificed and the remaining rats were kept for 3
weeks without treatment. There were no adverse toxicological
effects on the animals exposed to the highest dose. Under the same
conditions (i.e. 220 mg/m3 for 4 weeks), racemic phenothrin did not
produce any adverse effects in male or female ICR mice. Similarly,
d-phenothrin also produced no adverse toxicological effects in
Sprague Dawley rats or ICR mice (both male and female) following
whole body exposure at 210 mg/m3 (particle size 5 µm) for 4 weeks
(Kohda et al., 1979b).
The acute oral and intraperitoneal toxicity of phenothrin
metabolites in rats and mice are shown in Table 6 and 7 (Kohda et
al., 1979a; FAO/WHO, 1981).
Table 6. The acute oral toxicity of phenothrin
metabolites
-----------------------------------------------------
Chemical No.a Species LD50
(mg/kg)
-----------------------------------------------------
3-Phenoxybenzyl alcohol 12 Rat 1330
3-Phenoxybenzaldehyde 13 Rat 600
-----------------------------------------------------
a Chemical identification number used in Fig. 2
Table 7. The acute intraperitoneal toxicity of several phenothrin
metabolites in mice
----------------------------------------------------------------------
Chemical No.a LD50 (mg/kg)
Male Female
----------------------------------------------------------------------
3-Phenoxybenzyl alcohol 12 371 424
3-(4'-hydroxyphenoxy)benzyl alcohol 14 750-1000 750-1000
3-(2'-hydroxyphenoxy)benzyl alcohol 15 876 778
3-Phenoxybenzoic acid 16 154 169
3-(4'-hydroxyphenoxy)benzoic acid 17 783 745
3-(2'-hydroxyphenoxy)benzoic acid 18 859 912
3-Phenoxybenzaldehyde 13 415 416
----------------------------------------------------------------------
a Chemical identification number used in Fig. 2
7.2. Long-Term Exposures and Carcinogenicity
Appraisal
There have been several long-term feeding studies with racemic
phenothrin or d-phenothrin (dose levels ranging from 200 to 10 000
mg/kg diet) in rats and mice, the length of exposure ranging from 6
months to 2 years. A slight increase in liver weight and a
significant difference in some clinical/chemistry parameters from those
of controls were observed at high doses. However, the no-observed-
effect level (NOEL) values obtained in these studies were high (300-
1000 mg/kg diet, corresponding to around 40-160 mg/kg body weight) and
no tumorigenicity was observed. Two feeding studies with d-phenothrin
(doses of 100-3000 mg/kg diet with exposure period 26 and 52 weeks) in
dogs revealed similar results, the NOEL being 300 mg/kg diet
(corresponding to around 7-8 mg/kg body weight per day). No
tumorigenicity relating to phenothrin feeding was detected in these
studies.
7.2.1. Rat
In a study by Murakami et al. (1981), [1R, cis]-phenothrin
(d-phenothrin) was administered to Sprague Dawley rats (20 of each
sex per group) at dose levels of 0, 1, 3, or 10 g/kg diet for 6
months. Ten rats of each sex per group were sacrificed after 3
months. d-Phenothrin had no significant effect on mortality,
clinical signs, ophthalmology, urinalysis, or gross and
histopathological findings. The serum albumin level was elevated
after 3 months in males fed 10 g/kg and in females fed 3 or 10
g/kg, and after 6 months in males fed 3 or 10 g/kg. The albumin-
globulin ratio was raised after 3 months in males fed 3 or 10 g/kg
and in females fed 10 g/kg, and in both males and females fed 10
g/kg after 6 months. Absolute and relative liver weights in both
males and females fed 3 or 10 g/kg were increased. Based on these
data, it was concluded that the NOEL for d-phenothrin in this study
was 1 g/kg diet for both sexes (55.4 mg/kg body weight per day for
males and 63.3 mg/kg body weight per day for females).
In a standard oncogenicity study, Fisher-344 rats (50 of each
sex per group) were fed d-phenothrin at dose levels of 0, 300, 1000
or 3000 mg/kg diet for at least 105 weeks in males and at least 118
weeks in females. Additional rats (30 of each sex per group) were
assigned to a chronic toxicity study with a 52-week interim
sacrifice. There were no significant effects on clinical signs,
mortality, food and water consumption, ophthalmology, blood
biochemistry, haematology, or urinalysis. However, the body weight
gain in females fed d-phenothrin at 3000 mg/kg was reduced, and the
relative liver weight was increased in females fed 3000 mg/kg for
52 weeks and, at the end of the oncogenicity study, in males fed
3000 mg/kg. Microscopic examination revealed that the incidence of
cystic dilatation of the sinuses of the mesenteric lymph nodes and
of periacinar hepatocytic hypertrophy was higher in males fed 3000
mg/kg for at least 105 weeks. d-Phenothrin did not show any
oncogenic activity to rats at up to 3000 mg/kg. Although at this
dose an increase in the incidence of adenomas and carcinomas of the
preputial gland was seen in males, the 1988 Joint FAO/WHO Meeting
on Pesticide Residues (FAO/WHO, 1988b) considered it unlikely that
this finding was of toxicological significance. The NOEL was 1000
mg/kg diet for both sexes (47 mg/kg body weight per day for males
and 56 mg/kg body weight per day for females) (Martin et al.,
1987).
When Sprague Dawley rats (50 of each sex per group) were fed a
diet containing racemic phenothrin (0, 200, 600, 2000, and 6000
mg/kg diet) for 2 years, body weight and food consumption were
slightly depressed at 6000 mg/kg in both males and females.
Ophthalmological examinations, haematological studies, urinalyses,
and clinical chemistry studies were performed at various intervals.
At the end of the study all animals were sacrificed and examined
for gross abnormalities, and extensive microscopic examinations
were conducted on a variety of tissues and organs. There were no
abnormal clinical or behavioral problems associated with
phenothrin administration. The survival rate of all groups of
treated rats was similar to that of controls. Males fed 6000 mg/kg
showed a significant increase in serum glutamine-pyruvate
aminotransferase activity. Ophthalmological examinations revealed
some abnormalities, all of which appeared to be age related.
Histopathological examination revealed no significant differences
between the treated groups and the control group with respect to
severity of lesions. No histopathological changes suggestive of
oncogenicity resulting from phenothrin treatment were found
(Hiromori et al., 1980).
7.2.2. Mouse
When Swiss White mice (50 of each sex per group) were fed
racemic phenothrin for 18 months at dose levels of 0, 300, 1000, or
3000 mg/kg diet, there were no significant effects on mortality,
clinical signs, haematologic values, clinical chemistry parameters,
or gross pathological findings. Slight body weight depression
occurred in males fed 3000 mg/kg, and increased liver weight was
found at the highest dose level in both males and females. There
was a statistically significant difference (compared with the
controls) in lung amyloidosis in the 1000 and 3000 mg/kg dose
groups, but no significant increase in tumours attributable to
phenothrin ingestion (Murakami et al., 1980).
In studies by Amyes et al. (1987), B6C3F1 hybrid mice (90 of
each sex per group) were fed d-phenothrin in the diet at dose
levels of 0, 300, 1000, or 3000 mg/kg. Fifty of each sex per group
were allocated to a standard oncogenicity study lasting 104 weeks.
The remaining mice were assigned to a chronic toxicity study, where
10 of each sex per group were sacrificed for interim study after 26
or 53 weeks and the remaining animals were examined after 78 weeks
of treatment. There were no compound-related effects on clinical
signs, mortality, ophthalmology, blood biochemistry, haematology,
or urinalysis. However, body weight gains for males fed d-
phenothrin at 3000 mg/kg were reduced and relative liver weights
were increased in both sexes fed 3000 mg/kg and in males receiving
1000 mg/kg. Microscopic examination revealed that the incidence of
periacinar hepatocyte hypertrophy with cytoplasmic eosinophilia was
higher in males fed 3000 mg/kg. The incidence of liver tumors
appeared higher in phenothrin-treated female mice than in control
females, but the difference was not statistically significant. It
was concluded, therefore, that administration of d-phenothrin to
mice for 2 years at dietary levels of up to 3000 mg/kg diet did not
significantly disturb the tumour burden or tumour profile of B6C3F1
hybrid mice. The NOEL in this study was 300 mg/kg diet for males
(40 mg/kg body weight per day) and 1000 mg/kg diet for females (164
mg/kg body weight per day).
7.2.3. Dog
When beagle dogs (six of each sex per group) were fed d-
phenothrin at dose levels of 0, 100, 300, or 1000 mg/kg diet for 26
weeks, there were no compound-related abnormal findings in
mortality, clinical signs, body weight, food consumption,
ophthalmology, gross or microscopic pathology, haematology, or
urinalysis studies. However, the alkaline phosphatase activity in
males fed 300 mg/kg and males and females fed 1000 mg/kg was
elevated and a slight increase in the mean relative liver weight in
males fed 1000 mg/kg was noted. The NOEL in this study was 300
mg/kg (Pence et al., 1981).
In a study by Cox et al. (1987), beagle dogs (four of each sex
per group) were fed d-phenothrin at dose levels of 0, 100, 300,
1000, or 3000 mg/kg diet for 52 weeks. There were no significant
effects on clinical signs, body weight, food consumption,
ophthalmology, or urinalysis. However, decreases in erythrocyte
count, haemoglobin concentration, haematocrit, and total blood
protein were noted in both male and female dogs fed 3000 mg/kg,
whereas mean absolute and relative liver weights increased.
Compound-related histopathological alterations were noted in the
adrenal glands and liver. Focal degeneration of the adrenal cortex
with cytoplasmic deposition of crystalline material was seen in one
male dog fed 1000 mg/kg and four dogs fed 3000 mg/kg. The chemical
nature or biological significance of this crystalline material was
not recorded. Hepatocytes appeared to enlargen slightly in one
male dog fed 1000 mg/kg and seven dogs fed 3000 mg/kg. The NOEL in
this study was 300 mg/kg diet for males and 1000 mg/kg for females
(8.24 and 26.77 mg/kg body weight per day for males and females,
respectively).
7.3. Mutagenicity
The results obtained in vivo and in vitro test system indicate
that d-phenothrin does not exhibit any mutagenic properties or
cause chromosomal or DNA damage.
In a DNA-repair test with Bacillus subtilis (M45 rec- and H17
wild type strains) using dose levels of up to 5 mg/disk per plate,
d-phenothrin did not inhibit the growth of any strain at any dose
level, whereas the positive control, mitomycin C, showed a clear
effect. The negative control gave a result similar to that of d-
phenothrin (Kishida & Suzuki, 1981a).
A mutagenicity test with Escherichia coli (WP2 uvr) and
Salmonella typhimurium (TA 1535, TA 1537, TA 1538, TA 98, and TA
100) using d-phenothrin at dose levels of up to 5 mg/plate with and
without metabolizing enzyme system (S9 mix) yielded negative
results, whereas a positive control gave a significant number of
mutants (Kishida & Suzuki, 1981b).
In a host-mediated assay using S. typhimurium G46 (indicator
bacteria), d-phenothrin in corn oil was given orally (twice with a
24-h interval) to groups of six male ICR mice at dose levels of
2500 or 5000 mg/kg body weight. Soon after the last administration,
each mouse was injected intraperitoneally with the indicator cells.
Three hours later, the bacterial mutation frequency in d-
phenothrin-treated mice was no greater than that in the control
group (Kishida & Suzuki, 1981c).
Suzuki et al. (1981) examined d-phenothrin for its ability to
induce chromosomal aberrations in vivo using bone marrow cells.
ICR mice were treated intrapertioneally with single doses of 2500,
5000, or 10 000 mg/kg body weight and sacrificed 6, 24, or 48 h
after treatment. No chromosomal aberrations as a result of d-
phenothrin treatment were detected.
In an in vitro chromosomal aberration test, Chinese hamster
ovary cells (CHO-K1) were treated with d-phenothrin (dose levels:
2 x 10-5 to 2 x 10-4 mol/litre for 24 and 48 h in the absence of S9
mix; 5 x 10-5 to 5 x 10-4 mol/litre for 6 h in the presence of S9
mix). No significant increase in the number of cells with
chromosomal aberrations was observed (Kogiso et al., 1986).
The ability of d-phenothrin to induce sister-chromatid
exchanges (SCEs) were tested in cultured mouse embryonic cells in
vitro. At doses of 10-5, 10-4, and 10-3 mol/litre (with and
without S9 mix), d-phenothrin did not induce any increase in the
frequency of SCEs (Suzuki & Miyamoto, 1981).
In a study of unscheduled DNA synthesis, Hela S3 cells were
treated with d-phenothrin at dose levels of 0, 0.25, 0.5, 1.0, 2.0,
or 4.0 mg/ml in the presence of 3H-thymidine (with and without S9
mix) for 3 h, and the incorporation of 3H-thymidine into DNA was
measured. There was no significant increase in the radioactivity
of DNA from cells treated with d-phenothrin (Foster et al., 1984).
7.4. Reproduction, Embryotoxicity, and Teratogenicity
No teratogenicity or reproductive effects were observed when
phenothrin was fed to rabbits or mice during the major
organogenesis period of gestation or to rats in 3-generation
reproduction studies.
7.4.1. Embryotoxicity and teratogenicity
In studies by Ladd et al. (1976), pregnant New Zealand White
rabbits (17 per group) were administered racemic phenothrin orally
at dose levels of 0, 3, 10, or 30 mg/kg body weight on days 6 to 18
of gestation. They were sacrificed on day 29 and the young
obtained by caesarian section were examined. At 30 mg/kg, the body
weight of females decreased during gestation, and there was a
slight decrease in the number of live young and a slight reduction
in fetal weight. Racemic phenothrin had no apparent teratogenic
effect, as shown by a lack of gross internal or external somatic
abnormalities and by normal fetal skeletal development following
prenatal exposure.
Pregnant New Zealand White rabbits (15 per group) were orally
administered d-phenothrin by intubation (0, 10, 100, or 1000 mg/kg
body weight per day) on days 6 to 18 of gestation, and were
sacrificed on day 29 or 30. Following caesarian section, 50% of
the pups were maintained for 24 h to evaluate survival. No
abnormalities were observed among the does (body weight, food
consumption, clinical observations, and necropsy) or foetuses
(implantation sites, corpora lutea, resorption sites, weight,
condition, and viability). Data on foetal survival and from
internal and external examinations for abnormalities showed not
significant effects from administrating d-phenothrin during
gestation (Rutter, 1974).
In studies by Nakamoto et al. (1973), d-phenothrin was orally
administered to pregnant ICR mice (17 or 18 per group) at dose
levels of 0, 30, 300, or 3000 mg/kg body weight on days 7 to 12 of
gestation (not covering the whole period of organogenesis). The
dams were sacrificed on day 18 of gestation and the pups were
obtained by caesarian section. Other mice (7 per group) were given
d-phenothrin at dose levels of 0, 300, or 3000 mg/kg to evaluate
postnatal effect. These mice were allowed to deliver naturally and
the pups were kept for 29 days. At these levels, d-phenothrin
showed no adverse effects, as indicated by maternal growth, fetal
mortality and external and internal examination of fetuses for
teratogenic or embryotoxic effects.
7.4.2. Reproduction studies
In a standard 3-generation (2 litters per generation)
reproduction study, groups of rats (8 male and 16 female Charles
River albino rats per group) were fed racemic phenothrin at dose
levels of 0, 200, 600, or 2000 mg/kg diet. Various reproduction
indices (i.e. mating index, fecundity index, male fertility index,
female fertility index, and incidence of parturition) were
measured. The adult rats showed no significant mortality or
complications during the study, and the reproductive parameters
revealed no significant dose-related adverse effects attributable
to phenothrin. Gross and microscopic findings indicated no adverse
effect resulting from dietary phenothrin. It was concluded that
phenothrin had no effect on reproduction (Takatsuka et al., 1980).
In a study by Tesh et al. (1978), d-phenothrin was fed to
Charles River CD rats (30 of each sex per group) at dose levels of
0, 300, 1000, or 3000 mg/kg diet throughout two successive
generations and up to the maturation of a third generation. At 300
and 1000 mg/kg, there was no adverse effect upon mortality, somatic
growth, development, or reproductive performance. At 3000 mg/kg,
mortality, body weight, and reproductive performance showed no
significant response to treatment, and selected F2 animals reared
to maturity were in all respects comparable with control rats.
However, F0 and F1 females and selected F2B male and female
weanlings showed a slight but consistent increase in the relative
liver weight. The NOEL in this study was 1000 mg/kg diet.
7.5. Neurotoxicity
Sprague Dawley rats exposed to d-phenothrin by inhalation at
concentratons of up to 3760 mg/m3 for 4 h showed no toxic signs as
a result of exposure. Histopathologically, there were no compound
related alterations in the sciatic nerve (Kohda et al., 1977).
When d-phenothrin was given to Sprague Dawley rats orally for 5
consecutive days (5 g/kg body weight per day), one out of ten
female rats died after four doses and signs of poisoning
(piloerections and urinary incontinence) were noted in several of
the animals. However, these signs disappeared rapidly at the end
of the treatment and there were no other signs of poisoning such as
leg weakness or ataxia. All animals were sacrificed 3 days after
the final dose, and histopathologically examination of the sciatic
nerve revealed minute changes in axon and myelin, characterized by
very slight axonal swelling, axonal disintegration, and/or
demyelination. Since there were similar changes in the control
animals, it was suggested that they were not due to the d-
phenothrin. It was considered that the oral administration of very
high doses of d-phenothrin does not lead to the neurotoxic effects
observed with several other pyrethroid esters (Okuno et al., 1978).
7.6. Miscellaneous Effects
No d-phenothrin-attributable pharmacological effects were
detected in various tests (e.g., spontaneous movement of isolated
guinea-pig ileum, contraction of the rat phrenic nerve diaphragm
preparation, cardiopulmonary physiology of anaesthetized cats,
coordination, and spontaneous movement of mice, and rectal
temperature of rats) at doses of 100-300 µg/ml in vitro, 3 mg/kg
intravenous, or 100-300 mg/kg intraperitoneal. A tentative arousal
response was recorded in the electroencephalogram of cats given d-
phenothrin (4 mg/kg) intraperitoneally, as is commonly observed in
animals given synthetic pyrethroids (Hara et al., 1974).
7.7. Mechanisms of Toxicity - Mode of Action
Some synthetic pyrethroids given intravenously to rats cause
either tremor (T-syndrome) or choreoathetosis with salivation (CS-
syndrome) (Verschoyl & Aldridge, 1980). However, d-phenothrin
(>600 mg/kg body weight) injected intravenously into the lateral
tail vein caused neither T-syndrome nor CS-syndrome, due to its
very low acute toxicity. From a study involving intracerebral
dosing with [1R, cis]- or [1R, trans]-phenothrin in mice, both
compounds were classified as Type I pyrethroids based on the
occurrence of tremors (Lawrence & Casida, 1982) and on
neurophysiological studies in cockroach cercal sensory nerves
(Gammon et al., 1981).
8. EFFECTS ON HUMANS
Although d-phenothrin has been used for more than 10 years, no
toxic effects and no cases of poisoning have been reported.
8.1. Clinical Studies
In a study by Hashimoto et al. (1980), d-phenothrin (talc
powder formulation with Span 80 as a stabilizer) was applied to the
head hair and pudenda hair of eight male human volunteers (three
times at intervals of 3 days) at a dose of 32 mg/man per
administration (0.44 to 0.67 mg/kg body weight per day). d-
Phenothrin powder was washed off 1 h after application. There were
no significant abnormalities due to d-phenothrin in terms of dermal
irritation, clinical signs, or blood biochemical and haematological
parameters. The blood levels of d-phenothrin were below the
detection limit of 0.006 mg/kg.
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has
discussed and evaluated d-phenothrin at its meetings in 1979, 1980,
1984, and 1988 (FAO/WHO, 1980, 1981, 1985a, 1988b).
Since 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg
body weight has been established.
In the WHO Recommended Classification of Pesticides by Hazard,
technical phenothrin is classified as unlikely to present an acute
hazard in normal use (WHO, 1988).
REFERENCES
AMYES, S.J., MARTIN, P.A., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Oncogenicity and toxicity study
in mice, Suffolk, United Kingdom, Life Science Research (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of syn-
thetic pyrethroids in fruit and vegetables by gas-liquid and high-
performance liquid chromatography. Analyst, 107: 206-212.
COX, R.H., SUTHERLAND, J.D., VOELKER, R.W., ALSAKER, R.D.,
VARGAS, K.J., LEWIS, S.A., & HAGEN, W.J. (1987) Chronic toxicity study
in dogs with Sumithrin technical grade, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
DESMARCHELIER, J.M. (1980) Comparative study of analytical methods for
bioresmethrin, phenothrin, d-phenothrin, pyrethrum I, carbaryl,
fenitrothion, methacrifos, pirimifos-methyl and dichlorfos on various
grains. J. Pestic. Sci., 5: 521-532.
DOI, T., SAKAUE, S., & HORIBA, M. (1985) Analysis of d-phenothrin and
its optical isomers in technical preparations and formulations. J.
Assoc. Anal. Chem., 68(5): 911-916.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC, American
Chemical Society, p. 229 (ACS Symposium Series No. 42).
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to a
synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
FAO (1982) Second Government Consultation on International Harmoniz-
ation of Pesticide Registration Requirements, Rome, 11-15 October,
1982, Food and Agriculture Organization of the United Nations.
FAO/WHO (1980) Pesticide residues in food: 1979 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 427-452
(FAO Plant Production and Protection Paper 20 Sup.).
FAO/WHO (1981) Pesticide residues in food: 1980 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 394-403
(FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1985a) Pesticide residues in food: 1984 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 689-694
(FAO Plant Production and Protection Paper 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues, 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1988a) Pesticide residues in food: 1987 Evaluations. Part 1
- Residues, Rome, Food and Agriculture Organization of the United
Nations, pp. 109-113 (FAO Plant Production and Protection Paper 86.1).
FAO/WHO (1988b) Pesticide residues in food. Report of the 1988 Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues, Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper 92).
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous sensation
between synthetic pyrethroid insecticides. Contact Dermatitis, 13(3):
140-147.
FOSTER, R., TIPINS, R.S., DE VENEZIA, V., & NUNZIATA, A. (1984)
Unscheduled DNA synthesis in human cells, cell line: Hela S3, test
substance: Sumithrin, Suffolk, United Kingdom, Life Science Research,
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
FUJIMOTO, K., ITAYA, N., OKUNO, Y., KADOTA, T., & YAMAGUCHI,T. (1973)
A new insecticidal pyrethroid ester. Agric. biol. Chem., 37:2681-2682.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent class
antagonize GABA action at the crayfish neuromuscular junction.
Neurosci. Lett., 40:163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid toxi-
cology: Protective effects of diazepam and phenobarbital in the mouse
and the cockroach. Toxicol. appl. Pharmacol., 66: 290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural variations
affecting pyrethroid neurotoxicity. Neurobehav. Toxicol. Teratol., 4(6):
793-799.
HARA, Y., MIYAGISHI, A., OHTSUKA, M., ASAMI, Y., KUROKAWA, Y.,
& MIYAMOTO, J. (1974) Pharmacological study of Sumithrin: Effects on
respiration, blood pressure, and peripheral central nervous systems
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HASHIMOTO, S., OKANO, S., YAMADA, H., & MIYAMOTO, J. (1980)
Human volunteer study with phenothrin and d-phenothrin
powder (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
HAYASHI, A. (1977) [Safe use of insecticides (I)] [Bo-satsu-kyo News,]
No. 1 p. 10 (June) (in Japanese).
HIROMORI, T., KOYAMA, Y., OKUNO, Y., ARAI, M., ITO, N., &
MIYAMOTO, J. (1980) Two-year chronic toxicity study of S-2539 in
rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HIROMORI, T., HARA, S., KADOTA, T., & MIYAMOTO, J. (1984) Acute
intravenous toxicity of phenothrin and d-phenothrin in mice
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
ISOBE, N., MATSUNAGA, H., NAKATSUKA, I., & YOSITAKE, A.
(1987) Metabolism of [1R, trans]- and [1R, cis]- isomers of phenothrin
in rats (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
ITAYA, N., KAMOSHITA, K., KITAMURA, S., OKUNO, Y., MIZUTANI,
T., NAKAI, S., KAMEDA, N., & FUJIMOTO, K. (1969) Esters of cyclo-
propanecarboxylic acids. Ger. Offen., 1: 926, 433.
IZUMI, T., KANEKO, H., MATSUO, M., & MIYAMOTO, J. (1984)
Comparative metabolism of the six optical stereo isomers of phenothrin
in rats and mice. J. Pestic. Sci., 9: 259-267.
KANEKO, H., OHAKAWA, H., & MIYAMOTO, J. (1981) Absorption and
metabolism of dermally applied phenothrin in rats. J. Pestic. Sci., 6:
169-182.
KISHIDA, F. & SUZUKI, H. (1981a) Studies on DNA-damaging capacity of
Sumithrin with Bacillus subtilis (Unpublished Technical Report No. ET-
10-0069 submitted to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA. F. & SUZUKI, H. (1981b) Gene mutation test of Sumithrin in
bacterial system (Unpublished Technical Report No. ET-10-0068 submitted
to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA, F. & SUZUKI, H. (1981c) Mutagenicity test of Sumithrin in
host-mediated assay (Unpublished Technical Report No. ET-10-0067 sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
KOGISO, S., HARA, M., ITO, K., IWAWAKI, H., & YOSHITAKE, A.
(1986) In vitro chromosomal aberration test of S-2539F in Chinese
hamster ovary cells (CHO-K1) (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1977) Acute
inhalation toxicity of d-phenothrin (S-2539 Forte) in rats (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
KOHDA, H., KENEDO, H., OHKAWA, H., KADOTA, T., & MIYAMOTO, J.
(1979a) Acute intraperitoneal toxicity of permethrin metabolites in
mice (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
KOHDA, H., NISHIMOTO, K., KADOTA, K., & MIYAMOTO, J. (1979b)
Acute and subacute inhalation toxicity studies of S-2539 Forte in rats
and mice (Unpublished report submitted to WHO by Sumitomo Chemical
Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1980) Particle
size distribution and mean particle size in acute inhalation toxicity
study of d-phenothrin (S-2539 forte) in rats (Unpublished Technical
Report No. ET-00-0054 submitted to WHO by Sumitomo Chemial Co.,
Ltd).
LADD, R., SMITH, P.S., JENKINS, D.H., KENNEDY, G.L., Jr, KINOSHITA,
F.K., & KEPLINGER, M.L. (1976) Teratogenic study with S-2539 in albino
rabbits, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: mouse
intracerebral structure-toxicity relationship. Pestic. Biochem. Physiol.,
18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions of
pyrethroid insecticides with chloride ionophore-associated binding
sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticides, London, Taylor &
Francis Ltd, p. 440.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel modifi-
cation as the basis for the variation in the nerve membrane effects of
pyrethroids and DDT analogs. Pestic. Biochem. Physiol., 20: 203-216.
MARTIN, P.A., AMYES, S.J., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Combined toxicity and
oncogenicity study in rats, Suffolk, United Kingdom, Life Science
Research (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of synthetic
pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue analysis of
synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry-human welfare
and the environment. Proceedings of the Fifth International Congress of
Pesticide Chemistry, Kyoto, Japan, 29 August to 4 September, 1982, Oxford,
Pergamon Press, Vol. 1-4.
MIYAMOTO, J., SUZUKI, T., & NAKAE, C. (1974) Metabolism of phenothrin
or 3-phenoxybenzyl d-trans-chrysanthemate in mammals. Pestic. Biochem.
Physiol., 4: 438-450.
MIYAMOTO, J., KOGISO, S., & MATSUO, M. (1984) Additional toxicological
information for phenothrin (Unpublished Technical Report No. ET-40-0090
from Sumitomo Chemical Co., Ltd).
MULLA, M.S., DARWAZEH, H.A., & DHILLON, M.S. (1980) New
pyrethroids as mosquito larvicides and their effects on nontarget
organisms. Mosq. News, 40: 6-12.
MURAKAMI, M., HIROMORI, T., ITO, S., & HOSOKAWA, S. (1981)
Six month oral toxicity study of S2539 Forte (Sumithrin(R)) in rats
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURAKAMI, J., ITO, S., OKUNO, Y., ARAI, M., ITO, N., & MIYAMOTO, J.
(1980) Eighteen-month chronic oral toxicity and tumorigenicity study of
S-2539 in mice, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURANO, A. (1972) Determination of the optical isomers of insecticidal
pyrethroids by gas-liquid chromatography. Agric. biol. Chem., 36:
2203-2211.
NAKAMOTO, N., KATO, T., & MIYAMOTO, J. (1973) Teratogenicity study
of S-2539 Forte in mice (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
NAMBU, K., OHKAWA, H., & MIYAMOTO, J. (1980) Metabolic fate of
phenothrin in plants and soils. J. Pestic. Sci., 5: 177-197.
NAMBU, K., TAKIMOTO, Y., & MIYAMOTO, J. (1981) Degradation of
phenothrin in stored wheat grains. J. Pestic. Sci., 6: 183-191.
NIOSH (1983) Registry of toxic effects of chemical substances, 1981-
1982 edition, Cincinnati, Ohio, US National Institute for Occupational
Safety and Health, p. 1003.
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
OHNISHI, J., IKEDA, M., SUZUKI, Y., & MATSUDA, T. (1987) Residue
analysis of d-phenothrin, fenitrothion and piperonyl butoxide in stored
wheat, wheat milling fractions and bread (interim report) (Unpublished
Report No. ER-RD-8719 submitted to WHO by Sumitomo Chemical Co.,
Ltd).
OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1978) Neurotoxicity study
of d-phenothrin (S-2539 Forte(R)) in rats by repeated oral administration
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
PAPADOPOULOU-MOURKIDOU, E., IWATA, Y., & GUNTHER, F.A.
(1981) Utilization of an infrared detector for selective liquid
chromatographic analysis. 2. Formulation analysis of the pyrethroid
insecticides allethrin, decamethrin, cypermethrin, phenothrin and
tetramethrin. J. agric. food Chem., 29: 1105-1111.
PENCE, D.H., HAGEN, W.H., WLSAKER, R.D., HASTINGS, T.F.,
DAWKINS, B.G., TACEY, R.L., & MARSHALL, P.M. (1981) Subchronic
toxicity study of S2539-F in dogs, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids on a
nerve-muscle preparation of the clawed frog, Xenopus laevis. Pestic.
Biochem. Physiol., 25: 176-187.
RUTTER, H.A. (1974) Teratogenicity study in rabbits: S-2539 Forte,
Vienna, Virginia, Hazleton Laboratories Inc. (Unpublished report sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
RUZO, L.O., SMITH, I.H., & CASIDA, J.E. (1982) Pyrethroid
photochemistry: photooxidation reactions of the chrysanthemates,
phenothrin and tetramethrin. J. agric. food Chem., 30: 110-115.
SAKAUE, S., KITAJIMA, M., HORIBA, M., & YAMAMOTO, S. (1981) Gas
chromatographic and high performance liquid chromatographic
determination of d-phenothrin (Sumithrin) in formulations. Agric. biol.
Chem., 45: 1135-1140.
SEGAWA, T. (1976) Acute toxicity study of S-2539 in rats and mice,
Hiroshima, Hiroshima University School of Medicine (Unpublished report
submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979a) Acute oral, subcutaneous, dermal and intraperitoneal
toxicity studies of S-2539 Forte in rats, Hiroshima, Hiroshima
University School of Medicine (Unpublished Technical Report
No. ET-31-0033 submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979b) Acute oral, subcutaneous, dermal and
intraperitoneal toxicity studies of S-2539 Forte in mice, Hiroshima,
Hiroshima University School of Medicine (Unpublished Technical Report
No. ET-31-0032 submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H. & MIYAMOTO, J. (1981) In vitro sister chromatid
exchanges of Sumithrin (Unpublished Technical Report No. ET-10-0071
submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H., HARA, M., & MIYAMOTO, J. (1981) In vivo
chromosomal aberration test of Sumithrin on bone marrow cells of
mice (Unpublished Technical Report No. ET-10-0072 submitted to WHO by
Sumitomo Chemical Co., Ltd).
SUZUKI, T. & MIYAMOTO, J. (1978) Purification and properties of
pyrethroid carboxyesterase in rat liver microsome. Pestic. Biochem.
Physiol., 8: 186-198.
SUZUKI, T., OHNO, N., & MIYAMOTO, J. (1976) New metabolites of
(+)-cis-phenothrin, 3-phenoxybenzyl (+)-cis chrysanthemate, in rats.
J. Pestic. Sci., 1: 151-152.
TAKATSUKA, M., OKUNO, Y., SUZUKI, T., KADOTA, T., YASUDA, M.,
& MIYAMOTO, J. (1980) Three-generation reproduction study of S-2539
in rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
TAKIMOTO, Y., KATO, T., & MIYAMOTO, J. (1977) Residue analyses
of phenothrin (S-2539) in crops (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
TESH, J.M., WILLOUGHBY, C.R., & FOWLER, J.S.L. (1987) Sumithrin:
Effects upon reproductive performance of rats treated continuously
throughout two successive generations, Suffolk, United Kingdom, Life
Science Research (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
TURNBULL, S.R. & ARDLEY, J.H. (1987) Determination of d-phenothrin,
piperonyl butoxide and fenitrothion residues on wheat, derived
fractions and breads, at approximately one, three and six months post-
treatment, Australia, The Wellcome Foundation Ltd (Unpublished Report
No. AESH 87-11).
VAN DEN BERCKEN, J. (1977) The action of allethrin on the peripheral
nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels in
frog myelinated nerve membrane. In: Insect: Neurobiology and pesticide
action, London, Society of Chemical Industry, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER
ZALM, J.M. (1973) DDT-like action of allethrin in the sensory nervous
system of Xenopus laevis. Eur. J. Phamacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A.
(1979) Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VERSCHOYLE, R.D. & ALDRIDGE, W.N.(1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45: 325-
329.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Phamacol., 58: 501-504.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system. Neuropathol.
appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J.
(1982a) Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed frog,
Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN
BERCKEN, J. (1982b) Similar mode of action of pyrethroids and DDT on
sodium channel gating in mylinated nerves. Nature (Lond.), 295: 601-
603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF,
R.G.D.M., & VAN DEN BERCKEN, J. (1983) Temperature and structure-
dependent interaction of pyrethroids with the sodium channels in frog
node of Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WHO (1979) WHO Technical Report Series, No. 634 (Safe use of
pesticides. Third Report of the WHO Expert Committee on Vector Biology
and Control), pp 18-23.
WHO (1988) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1988-1989, WHO, World Health
Organization (WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1987) d-Phenothrin. In: The pesticide
manual, 8th ed., Croydon, British Crop Protection Council, pp. 654-
655.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
APPENDIX
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs ( Xenopus laevis; Rana temporaria, and
Rana Escuelenta) it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these two classes of pyrethroids has been made
on the basis of the symptoms of toxicity in mammals and insects
(Van den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge,
1980; Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group (allethrin, d-
phenothrin, permethrin, tetramethrin, cismethrin, and bioresmethrin)
(Type I: T-syndrome).
The pyrethroids that do not contain an alpha-cyano group give
rise to pronouced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3-10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organs and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5°C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of the "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 milliseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently, it was demonstrated that allethrin and DDT have
essentially the same effect on sodium channels in frog myelinated
nerve membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittently, cause large
depolarizing after-potentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl alcohol
(deltamethrin, cypermethrin, fenvalerate, and fenpropanate) (Type II:
CS-syndrome).
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependant depression of the
nervous impulse, brought about by a progressive depolarization
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, persumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
The results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
crayfish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered
pyrethroids closely approximates that of the convulsant picrotoxin
(PTX). Deltamethrin inhibits the binding of the [3H]-
dihydropicrotoxin to rat brain synaptic membranes, whereas the non-
toxic R epimer of deltamethrin is inactive. These findings suggest
a possible relation between the Type II pyrethroid action and the
GABA receptor complex. The stereospecific correlation between the
toxicity of Type II pyrethroids and their potency to inhibit the
[35S]-TBPS binding was established using a radioligand, [35S]= t-
butylbicyclophosphorothionate [35S]-TBPS. Studies with 37
pyrethroids revealed an absolute correlation, without any false
positive or negative, between mouse intracerebral toxicity and in
vitro inhibition: all toxic cyano compounds including deltamethrin,
1R, cis-cypermethrin, 1R, trans-cypermethrin, and [2S,alpha]-
fenvalerate were inhibitors, but their non-toxic stereoisomers were
not; non-cyano pyrethroids were much less potent or were inactive
(Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S,alphaS]-fenvalerate) increased the input resistance of crayfish
claw opener muscle fibres bathed in GABA. In contrast, two non-
insecticidal stereoisomers and Type I pyrethroids (permethrin,
resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis ). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action. It
may be related to the long-lasting repetitive activity in sense
organs and possibly in other parts of the nervous system, which, in
a more advance state of poisoning, may be accompanied by a
frequency-dependent depressoin of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vunerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
RESUME, EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1. Résumé et évaluation
1.1 Identité, propriétés physiques et chimiques, et méthodes
d'analyse
La phénothrine racémique a été synthétisée pour la première
fois en 1969. Du point de vue chimique, il s'agit de l'ester de
l'acide diméthyl-2,2 (diméthyl-2,2 vinyl)-3 cyclopropanecarboxylique
et de l'alcool phénoxy-3 benzylique (PBalc). Il se présente sous
la forme d'un mélange de quatre stéréoisomères : [1R,trans],
[1R,cis], [1S,trans], [1S,cis]. La d-phénothrine est un mélange
d'une partie d'isomère [1R,cis] pour quatre parties d'isomère
[1R,trans], et elle est à l'heure actuelle le seul produit
technique sur le marché. C'est l'isomère [1R,trans] qui est
l'insecticide le plus actif; vient ensuite l'isomère [1R,cis].
La d-phénothrine de qualité technique se présente sous la forme
d'un liquide jaune pâle à brun jaune et son degré de pureté est de
92,5 à 94,5%. Sa densité est de 1,058-1,061 à 25°C et sa tension
de vapeur de 0,16 mPa à 20°C. Elle est difficilement soluble dans
l'eau (2 mg par litre à 25°C) mais soluble dans les solvants
organiques tels que l'acétone, le xylène et l'hexane. Elle est
assez stable à l'air mais instable à la lumière, encore que sa
photodégradation ne soit pas aussi rapide que celle des pyréthrines
naturelles. Elle est instable en milieu alcalin.
Le dosage des résidus peut s'effectuer par chromatographie en
phase liquide à haute performance avec détecteur ultra-violet, la
concentration minimale décelable étant de 0,05 mg par kg. Pour
l'analyse du produit technique on utilise la chromatographie en
phase gazeuse avec détection par ionisation de flamme.
1.2 Production et usage
La d-phénothrine est utilisée depuis 1977. On estime que l'on
utilise chaque année 70 à 80 tonnes de d-phénothrine dans le monde,
essentiellement pour détruire les insectes incommodants dans les
habitations, lutter contre les vecteurs de maladies et protéger les
céréales ensilées; le produit est utilisé seul ou en association
avec d'autres insecticides ou synergisants. Il est présenté sous
forme d'huiles pour aérosols, de poudres ou de concentrés
émulsionnables. La d-phénothrine est également utilisée pour
détruire les poux de l'homme, auquel cas elle est présentée sous
forme de poudre, de shampooing ou de lotion.
1.3 Exposition humaine
Les aérosols classiques à usage domestique ne devraient pas
conduire à des concentrations atmosphériques de d-phénothrine
supérieures à 0,5 mg/m3. Dans le blé ensilé, on peut trouver des
résidus allant jusqu'à 4 mg/kg, mais ces teneurs tombent à 0,8
mg/kg dans la farine après mouture, et à 0,6 mg/kg après
panification.
Pour détruire les poux, on applique la d-phénothrine sur la
chevelure, par exemple en trois doses de 32 mg tous les trois
jours. Il n'existe aucune donnée sur l'exposition professionnelle à
la d-phénothrine.
L'exposition de la population dans son ensemble devrait être
très faible, mais on manque de données précises sur ce point.
1.4 Destinée dans l'environnement
La phénothrine se dégrade facilement, sa demi-vie étant
inférieure à un jour sur les végétaux et autres surfaces. La d-
phénothrine ou ses produits de dégradation ne migrent que très peu
vers les zones non traitées des végétaux. On a constaté que des
haricots ne captaient que dans une faible mesure les prouits
radiomarqués provenant de sols traités par de la phénothrine
marquée au carbone-14. Après traitement des sols avec de la
[1R, trans ]- ou de la [1R, cis]-phénothrine à raison de 1 mg/kg, on
a constaté que les deux isomères se décomposaient rapidement, la
demi-vie initiale étant de un à deux jours, mais que, en cas
d'inondation, la dégradation était considérablement ralentie, la
demi-vie initiale étant de deux à quatre semaines pour l'isomère
trans et de un à deux mois pour l'isomère cis. On a observé que
les isomères trans ou cis de la phénothrine se déplaçaient
relativement peu (environ 2%) à travers des colonnes de terre,
lorsque le lessivage commençait immédiatement ou deux semaines
après le traitement.
En général, la dégradation qui se produit dans l'environnement
conduit à des produits moins toxiques.
1.5 Cinétique et métabolisme
Après avoir reçu une dose unique ou des doses répétées de
phénothrine radiomarquée par voie orale ou percutanée, des rats ont
rapidement et presque complètement excrété la fraction marquée dans
leurs urines et leurs déjections en trois à sept jours. Les
principales voies métaboliques des isomères cis et trans chez le
rat consistent en une rupture de la liaison ester et l'oxydation en
position 4 du reste alcool ou du groupement isobutényle du reste
acide. Les métabolites résultant du clivage de l'ester (qui sont
excrétées essentiellement dans les urines) constituent les
principaux produits de dégradation de l'isomère trans alors que les
métabolites restant sous forme d'ester (excrétées essentiellement
dans les déjections) proviennent pour la plupart de l'isomère cis.
1.6 Effets sur les êtres vivant dans leur milieu naturel
La phénothrine a été expérimentée sur quelques groupes
d'organismes non visés et dans chaque groupe sur quelques espèces
seulement. Chez les poissons, la CL50 à 96 heures de la
phénothrine racémique et des stéréoisomères (1R) va de 17 à 200
microgrammes par litre. Une étude, portant sur des invertébrés
aquatiques, a montré que chez Daphnia pulex la CL50 à 3 heures
était de 25 à 50 mg/litre pour tous les isomères et la phénothrine
racémique.
Une seule et unique étude au cours de laquelle de la
phénothrine a été appliquée à des étangs n'a révélé aucun effet sur
les arthropodes aquatiques.
La toxicité pour les oiseaux est faible, avec une DL50 aiguë
par voie orale supérieure à 2500 mg/kg de poids corporel chez le
colin de Virginie et une CL50 par voie alimentaire supérieure à
5000 mg/kg de nourriture chez ce volatile et chez le colvert.
Etant donné que la phénothrine se dégrade rapidement à la
lumière solaire et qu'on l'utilise principalement pour traiter des
céréales ensilées, l'exposition environnementale est
vraisemblablement très faible. Dans ces conditions, des effets sur
l'environnement sont tout-à-fait improbables.
1.7 Effets sur les animaux d'expérience et sur les systèmes
d'épreuve in vitro
La toxicité aiguë de la d-phénothrine est extrêmement faible,
la DL50 étant supérieure à 5000 mg/kg de poids corporel chez le rat
et la souris (par voie orale, sous-cutanée, dermique et
intrapéritonéale) et la CL50 inhalatoire supérieure à 3760 mg/m3
chez le rat. Le syndrome d'intoxication se caractérise par une
hyperexcitabilité, une prostration, des tremblements, de l'ataxie
et une paralysie. Sur la base de ces symptômes et d'après les
résultats des études électrophysiologiques sur les nerfs sensoriels
des cerques de la blatte, la phénothrine est classée parmi les
pyréthroides du type I.
Exposés à de la d-phénothrine par inhalation à des
concentrations allant jusqu'à 210 mg/m3, quatre heures par jour
pendant quatre semaines, ou par voie orale cinq jours de suite à
raison de 5000 mg/kg de poids corporel, des rats n'ont présenté
aucun effet toxicologique indésirable.
Plusieurs études d'alimentation ont été effectuées sur des rats
et des souris recevant de la d-phénothrine ou de la phénothrine
racémique à des doses allant de 200 à 10 000 mg par kg de
nourriture; la période d'exposition allait de six mois à deux ans.
Ces études ont permis d'établir une dose sans effet observable
allant de 300 à 1000 mg par kg de nourriture, ce qui correspond à
peu près à 40-160 mg/kg de poids corporel et par jour. Chez des
chiens à qui l'on avait administré de la d-phénothrine à des doses
de 100 à 3000 mg/kg de nourriture avec des périodes d'exposition de
26 à 52 semaines, deux études ont permis d'obtenir une dose sans
effet observable de 300 mg par kg de nourriture, soit 7 à 8 mg/kg
de poids corporel et par jour.
Divers systèmes permettant d'étudier in vivo et in vitro les
mutations géniques, les lésions et les réparations de l'ADN ainsi
que les effets chromosomiques, ont permis de constater que la d-
phénothrine n'avait pas d'effet mutagène.
Des études de deux ans ont également montré que la d-
phénothrine n'était pas cancérogène pour le rat ni la souris à des
doses allant jusqu'à 3000 mg par kg de nourriture. Aucune
tératogénicité ni embryotoxicité n'a été observée chez des foetus
de lapins et de souris dont les mères avaient reçu de la d-
phénotrine à des doses allant jusqu'à 1000 et 3000 mg par kg de
poids corporel, respectivement. Lors d'une étude de reproduction
chez le rat, portant sur deux générations, on a établi que la dose
sans effet observable était de 1000 mg par kg de nourriture.
Des rats exposés par inhalation à des doses très élevées de d-
phénothrine (jusqu'à 3760 mg/m3) pendant quatre heures ou,
quotidiennement, par voie orale, à une dose de 5000 mg par kg de
poids corporel, cinq jours durant, n'ont présenté aucune
dégénérescence myélinique ni désagrégation de l'axone au niveau du
nerf sciatique.
1.8 Effets sur les êtres humains
La d-phénothrine est utilisée depuis plus de 10 ans sans que
l'on ait signalé d'intoxication humaine.
Rien n'indique que cette substance puisse avoir des effets
nocifs sur l'homme pour peu qu'elle soit utilisée conformément aux
recommandations.
2. Conclusions
2.1 Population générale
L'exposition de la population dans son ensemble à la d-
phénothrine est vraisemblablement très faible et il n'y a
probablement aucun risque à cet égard si le produit est utilisé
conformément aux recommandations.
2.2 Exposition professionnelle
Si elle est utilisée de manière convenable, moyennant un
certain nombre de mesures d'hygiène et de sécurité, la d-
phénothrine ne devrait pas présenter de risque pour les personnes
qui lui sont exposées de par leur profession.
2.3. Environnement
Du fait qu'elle se dégrade rapidement à la lumière solaire et
qu'elle est principalement utilisée pour traiter les céréales
ensilées, l'exposition environnementale à la phénothrine est
vraisemblablement très faible. Dans ces conditions tout effet sur
l'environnement est extrêmement improbable.
3. Recommandations
Les niveaux d'exposition résultant d'une utilisation conforme
aux recommandations sont censés être extrêmement faibles, toutefois
on pourrait envisager de confirmer cette hypothèse en étendant la
surveillance à la d-phénothrine.
5.1. Metabolism in Mammals
Appraisal
After rats are treated with radiolabelled phenothrin, either by
single or repeated oral exposure, or dermally, the radioactivity is
rapidly (and almost totally) excreted into urine and faeces within
3 to 7 days. The major metabolic pathways of both trans- and cis-
phenothrin in rats are oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety and cleavage of
the ester linkage (see Fig. 3). Cleavage of the ester bond is more
difficult in the cis isomer than in the trans isomer. Thus, ester-
form metabolites oxidized in the various positions of the molecule are
the major metabolites of the cis isomer and are excreted mostly in the
faeces. However, ester-cleaved metabolites are the major products from
the trans isomer and are mostly excreted in urine.
When [1R, trans ]-phenothrin labelled with 14C at the methylene
moiety was given as a single oral dose (200 mg/kg body weight) to
Sprague Dawley male rats, the radiocarbon was rapidly eliminated,
57% and 43% being recovered within 3 days in urine and faeces,
respectively (Miyamoto et al., 1974). There was no detectable
radiocarbon in the expired air. Similarly, Sprague Dawley male
rats given a single oral dose (10 mg/kg body weight) of 14C-
[1R, trans ]-phenothrin, excreted the radiocarbon rapidly in the
urine (75%) and faeces (21%) (Kaneko et al., 1981).
In a study by Isobe et al. (1987), Sprague Dawley male and
female rats were given a single oral administration of 14C-
[1R, trans ]-phenothrin in corn oil at 4 and 200 mg/kg body weight.
Within 7 days the radiocarbon was almost completely eliminated in
the urine and faeces. The % elimination was as follows:
-----------------------------------------
Dose Urine Faeces
(mg/kg) Males Females Males Females
4 38 40 61 60
200 39 25 56 60
-----------------------------------------
Sprague Dawley male rats eliminated 65% of the dosed radiocarbon
in the faeces over 3 days after a single oral administration (200
mg/kg body weight) of 14C-[1R, cis ]-phenothrin (28) (Suzuki et al.,
1976), and 22 and 74% of the dose into urine and faeces,
respectively, 7 days after a single oral dose of 10 mg/kg body
weight (Kaneko et al., 1981).
When 14C-[1R, cis ]-phenothrin in corn oil was administered once
orally to Sprague Dawley male and female rats at 4 or 200 mg/kg
body weight, the radiocarbon was excreted into the urine (11-18%)
and faeces (81-87%) within 7 days. Similarly, when Sprague Dawley
rats were treated repeatedly with 14C-[1R,trans] or 14C-[1R,cis]
isomers at 4 mg/kg body weight per day for 14 days, the radiocarbon
was rapidly and almost completely excreted: 75-70% in urine and 24-
29% in faeces for the trans isomer, and 24% in urine and 72-73% in
faeces for the cis isomer (Isobe et. al., 1987).
The tissue residues in rats 7 days after a single oral dose of
14C-[1R, cis ]- or 14C-[1R, trans ]-phenothrin at 10 mg/kg body
weight were generally very low although the fat showed somewhat
higher residue levels (1-2.5 mg/kg) (Kaneko et al., 1981).
Similarly, high 14C residue levels (up to 23 mg/kg) were found in
the fat, 7 days after a single oral dose of the [1R,cis] isomer at
200 mg/kg body weight (Isobe et al., 1987).
The major metabolite of the trans isomer, when given as a
single oral dose of 200 mg/kg to rats, was 3-(4'-hydroxyphenoxy)-
benzoic acid (17) (4'OH-PBacid, 54%). There were smaller amounts
of 3-phenoxybenzoic acid (16) (PBacid, 9.5%) and its glycine
conjugate (Miyamoto et al., 1974).
When [1R, trans ]-phenothrin was given to rats at 4, 10, or 200
mg/kg body weight (oral single dose) or 4 mg/kg body weight
(repetitive oral dose for 14 days), the sulfate conjugate of 4'-OH-
PBacid was predominant, accounting for 28, 43, 28, and 55%,
respectively, of the dose. In addition, PBacid (4, 10, 5, and 6%),
its glycine conjugate (1,3,2, and 2%) and glucuronide (2,3,1, and
3%), and free 4'-OH-PBacid (2,11,3, and 3%) were found. The
sulfate conjugate of 3-(2'-hydroxyphenoxy)benzoic acid (18) (2'-
OH-PBacid) was also found as a minor metabolite (Kaneko et al.,
1981; Isobe et al., 1987).
When rats were given 14C-[1R, trans ]-phenothrin (10 mg/kg body
weight), the unmetabolized compound and two ester-form metabolites
were detected in their faeces in small amounts (0.4-1.2%), which
had hydroxymethyl (29) ( wt-alc- t-phe) or carboxyl group (30) wt-
acid- t-phe) (see Fig. 2) at the position of the trans methyl group
of chrysanthemic acid (Kaneko et al., 1981).
When Sprague Dawley rats were administered a single oral dose
of [1R, trans ]-phenothrin at 4 or 200 mg/kg body weight level or
given an oral dose of 4 mg/kg body weight per day for 14 days,
unmetabolized compound was found in the faeces (44-45, 44-60, and
14-16% of the dose, respectively). An ester-form metabolite, the
4'-hydroxy w(t)-acid derivative of trans- phenothrin, was also
detected (0.4-0.6%) (Isobe et al., 1987).
When male Sprague Dawley rats were given cis-phenothrin (200
mg/kg body weight), three ester-form metabolites, which accounted
for 14% of the dosed radioactivity, were found in the faeces.
These were 4'-hydroxy- cis-phenothrin (31) (4-OH- c-phe), an ester-
form derivative with the trans methyl of the isobutenyl group being
oxidized to carboxyl group (32) ( wt-acid- c-phe), and a compound
with the geminal-dimethyl groups oxidized (2-OH-) in addition to
both of the above modifications (33) (4'-OH, wt-acid, 2-OH(t)- c-phe)
(Suzuki et al., 1976).
In addition to this, the cis isomer gave rise to nine ester-
form metabolites in the faeces varying in amounts from 2% (4'-OH,
wc-alc-c-phe (34)) to 13% (4'-OH, wt-acid,2-OH(t)- c-phe (33)) of
the dosed radiocarbon after single oral administration. These
ester-form metabolites were transformed by oxidation reactions at
any of the following positions: 4'-position of the phenoxy group,
the trans or cis methyl of the isobutenyl groups, and the trans
methyl of the geminal-dimethyl group (Kaneko et al., 1981).
When Sprague Dawley rats were given a single oral dose of
[1R, cis ]-phenothrin at 4 or 200 mg/kg body weight level or an oral
dose of [1R, cis ]-phenothrin at 4 mg/kg body weight per day for 14
days, ester-form metabolites (1-9% of the dosed radioactivity) were
found, in addition to unmetabolized compound (17-59% of the dose).
The urine contained 4'-OH-PBacid as a sulfate conjugate (7-18%) and
in the free form (0.3-1%), and PBacid as glycine or glucuronide
conjugates and in the free form (0.3-1%) (Isobe et al., 1987).
Following the dermal treatment of male Sprague Dawley rats with
dust or emulsifiable concentrates (E.C.) of either 14C-[1R, trans ]-
or 14C-[1R, cis ]-phenothrin at 10 mg/kg body weight, the 14C
absorption into the body was estimated to be 3-7% of the initial
dose with dust and 8-17% with the E.C. After both dust and E.C.
treatments, the radiocarbon excreta (as a percentage of the initial
dose) recovered in the urine was 2.6-8.7% for the trans isomer, and
1.5-4.8% for the cis isomer, and in the faeces was 0.6-2.2% for the
trans isomer, and 3.0-12.3% for the cis isomer. Since the same
metabolites are formed following either oral exposure or dermal
treatment, it appears that both phenothrin isomers undergo the same
metabolism once in the systemic circulation, regardless of the
route of administration (Kaneko et al., 1981; Isobe et al., 1987).
Information concerning the comparative metabolism of racemic
(1RS) phenothrin and its d-isomer (1R) was obtained through a study
of CD rats and ddY mice given a single oral dose of either
[1R, trans ]-, [1S, trans ]-, [1RS, trans ]-, [1R, cis ]-, [1S, cis ]-,
or [1RS, cis ]-phenothrin. The radiocarbon derived from each isomer
was almost completely eliminated from the rats and mice within six
days after dosing. The trans isomers were mainly eliminated in the
urine (rat, 85-88%; mice, 65-75%) and the cis isomers mainly in the
faeces (rat, 57-71%; mice, 54-71%). The amounts of 14C in the
urine and faeces of rats and mice treated with the [1R,trans] and
[1R,cis] isomers did not differ significantly from those
corresponding to the [1RS,trans] and [1RS,cis] isomers, respectively.
The 14C tissue residues were very low, except in the fat. There
were no striking differences in 14C levels among the three trans
isomers and the three cis isomers. The 14C levels of the cis
isomers in fat (maximum 3.5 mg/kg) were three to seven times higher
than those of the trans isomers (less than 1 mg/ kg). The major
urinary and faecal metabolites were remarkably similar in both rats
and mice. In both rats and mice, there were virtually no
differences in the metabolic fate of the [1R,trans] and [1RS,
trans] isomers or of the [1R,cis] and [1RS,cis] isomers (Izumi et
al., 1984).
5.2. Enzymatic Systems for Biotransformation
In studies by Miyamoto et al. (1974), [IR, trans ]-phenothrin
(1 mmol/litre) was incubated with the 8000-g supernatant from a
liver homogenate of rats, mice, guinea-pigs, rabbits, or dogs at
37°C for 60 min in the absence of NADPH. The supernatant from the
guinea-pig was the most active in degrading [1R, trans ]-phenothrin,
followed by that of dog, rabbit, rat, and mouse. The major
metabolite in all the mammalian species tested was 3-phenoxybenzyl
alcohol (12) (PBalc). Smaller amounts of PBacid (16) and trace
amounts of 4'-OH-PBacid (17) were also found. However, in the
presence of NADPH, the amounts of PBacid and unidentified ether-
soluble metabolites increased in all species except dog. In
contrast to [1R, trans ]-phenothrin, [1R, cis ]-phenothrin was hardly
metabolized at all by the rat liver preparation in the absence of
NADPH. NADPH enhanced the degradation rate of the cis isomer,
leading to the formation of unidentified metabolites, while ester-
cleaved metabolites such as PBacid (16), PBalc (12) and 4'-OH-
PBacid (17) were found in very small amounts. When [1R, trans ]-,
[1R, cis ]-, [1S, trans ]-, and [1S, cis ]-phenothrin were incubated
with rat liver microsomes at 37.5°C for 30-60 min to estimate Km
and Vmax using a Lineweaver-Burk plot, the values for Km (0.11-0.17
mmol/litre) were similar for the four isomers, whereas the values
for Vmax were different; both the trans isomers yielded values for
Vmax 20-30 times larger than did the cis isomers (Miyamoto et al.,
1974).
In studies by Suzuki & Miyamoto (1978), pyrethroid
carboxyesterase(s) that hydrolyze esters of chrysanthemic acid
were purified from rat liver microsomes by cholic acid
solubilization, ammonium sulfate fractionation, heat treatment, and
DEAE-Sephadex A-50 column chromatography. The 45-fold-purified
enzyme (38% yield) probably consisted of a single protein with a
relative molecular mass of approximately 74 000, a Km of 0.21
mmol/litre for [1R, trans ]-phenothrin, and an optimum pH of 7-9.
It was susceptible to inhibition by organophosphate and carbamate
insecticides and insensitive to PCMB ( p-chloromercurybenzoic acid),
and mercuric and cupric ions. The enzyme seemed to require neither
coenzymes nor cofactors and hydrolysed the trans isomers of several
synthetic pyrethroids (tetramethrin, resmethrin, phenothrin, and
permethrin) well, at more or less similar rates. On the other hand,
the cis isomers were hydrolysed at rates 5-10 times lower than
their trans counterparts. The purified pyrethroid carboxyesterase
was apparently identical in nature to malathion carboxyesterase
and p-nitrophenyl acetate carboxyesterase.
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
Data on the acute toxicity of racemic and isomeric phenothrin
for aquatic organisms are summarized in Table 4.
6.1. Aquatic Organisms
Racemic phenothrin yielded 96-h LC50 values of 17-200 µg/litre
for the fish species tested (Table 4). The (1S)-optical isomers
were relatively non-toxic with LC50 values of 10 000 µg/litre,
whereas the (1R)-optical isomer and racemic phenothrin were of
similar toxicity with LC50 values between 120 and 200 µg/litre for
the killifish (Oryzias latipes) (Miyamoto, 1976).
When d-phenothrin was applied to ponds at the rates of 28 or 56
g/ha to control mosquito larvae, mayfly naiads were most affected
but no other arthropods (damselfly, dragonfly naiads, ostracods, or
diving beetle larvae) were seriously affected (Mulla et al., 1980).
6.2. Terrestrial Organisms
The available toxicity data for non-target terrestrial
organisms are very limited.
Phenothrin has low toxicity (acute oral dosage) for bobwhite
quail with an LD50 >2510 mg/kg body weight (Worthing & Walker,
1987). An 8-day feeding study with d-phenothrin on mallard duck
and bobwhite quail indicated LD50 values of >5620 and >5000 mg/kg
diet, respectivelya.
----------------------------------------------------------------------
a Personal communication from J.L. Noles, Ecological Effects
Branch, Hazard Evaluation Division, US Environmental Protection
Agency, November 1987.
Table 4. The acute toxicity of racemic and isomeric phenothrin to non-target aquatic organisms
-----------------------------------------------------------------------------------------------
Species Size Parameter Toxic Stereo- Systema Temper- Refer-
concentration isomeric ature enceb
(µg/litre) composition (°C)
-----------------------------------------------------------------------------------------------
Fish
Killifish adult 48-h LC50 200 racemic S 25 1
(Oryzias latipes) adult 48-h LC50 120 (+)-trans S 25 1
adult 48-h LC50 170 (+)-cis S 25 1
adult 48-h LC50 10 000 (-)-trans S 25 1
adult 48-h LC50 10 000 (-)-cis S 25 1
Rainbow trout 96-h LC50 17 racemic 2
(Salmo gairdneri)
Bluegill 96-h LC50 18 racemic 2
(Lepomis macrochirus)
Arthropods
Daphnia pulex 3-h LC50 50 000 racemic S 25 1
3-h LC50 25 000-50 000 (+)-trans S 25 1
3-h LC50 50 000 (+)-cis S 25 1
3-h LC50 50 000 (-)-trans S 25 1
3-h LC50 50 000 (-)-cis S 25 1
-----------------------------------------------------------------------------------------------
a s = static
b 1. Miyamoto (1976) 2. Worthing & Walker (1987)
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
The 1984 Joint FAO/WHO Meeting on Pesticide Residues concluded
that the toxicological data for racemic phenothrin can be used to
support that for d-phenothrin, owing to the similarity in
metabolism and toxicity between the two compounds (FAO/WHO,
1985b).
7.1. Single and Short-Term Exposures
The acute toxicity in rats and mice is extremely low. The LD50
values were >5000 mg/kg body weight when d-phenothrin was
administered orally, subcutaneously, dermally, or by
intraperitoneal injection to male and female Sprague Dawley rats
(Segawa, 1979a) and ddY mice (Segawa, 1979b) (Table 5).
Table 5. The acute toxicity of racemic phenothrin and d-phenothrin
to rats and micea
------------------------------------------------------------------
Compound/Route LD50 (mg/kg)
Rat Mice
Male Female Male Female
------------------------------------------------------------------
Phenothrin (racemic)
Oral > 5000 > 5000 > 5000 > 5000
Subcutaneous > 5000 > 5000 > 5000 > 5000
Dermal > 5000 > 5000 > 5000 > 5000
Intraperitoneal > 5000 > 5000 > 5000 > 5000
Inhalation > 1210b > 1210b > 1210b > 1210b
d-Phenothrin
Oral > 10 000 > 10 000 > 10 000 > 10 000
Subcutaneous > 10 000 > 10 000 > 10 000 > 10 000
Dermal > 10 000 > 10 000 > 5000 > 5000
Intraperitoneal > 10 000 > 10 000 > 10 000 > 10 000
Inhalation > 3760b > 3760b > 1180b > 1180b
------------------------------------------------------------------
a From: Segawa (1976; 1979a,b) and Khoda et al. (1977; 1979b; 1980)
b These are values for 4-h LC50 (mg/m3)
There were no differences between the effects on male and
female rats in acute toxicity studies carried out by Segawa (1976).
Signs of poisoning appeared rapidly following the intravenous
administration of phenothrin. These included fibrillation, tremor,
slow respiration, salivation, lacrimation, ataxia, and paralysis.
These signs, evident within 30 min to one hour following
administration, diminished rapidly to the point where, after 24 h,
there were no signs of toxicity.
No differences in the oral toxicity between racemic phenothrin
and d-phenothrin were detected by Segawa (1976; 1979a,b) (Table 5).
The intravenous LD50 values for racemic phenothrin in ICR mice
were 470 and 600 mg/kg for males and females, respectively, while
those for d-phenothrin were 265 and 315 mg/kg (Hiromori et al., 1984).
Following a 4-h acute inhalation exposure (whole body), the
LC50 values were >1210 mg/m3 for racemic phenothrin (particle size
5 µm) with both Sprague Dawley rats and ICR mice (Kohda et al., 1979b)
and >3760 and 1180 mg/m3 for d-phenothrin with Sprague Dawley rats
and ICR mice, respectively (Kohda et al., 1977; 1979b) (see Table 5).
At the higher concentration (3760 mg/m3), the mean particle size
was considered to be 0.72 µm and the cumulative distribution of
particles having a diameter between 0.46 and 1.09 µm was 88.9%,
according to a particle-size distribution experiment (Kohda et al.,
1980).
In studies by Kohda et al. (1979b), Sprague Dawley rats (15 of
each sex) were exposed (whole body) by inhalation to racemic
phenothrin at concentrations of 0, 43, or 220 mg/m3 (4 h per day, 5
days per week) for 4 weeks. After this treatment, animals (10/sex
per group) were sacrificed and the remaining rats were kept for 3
weeks without treatment. There were no adverse toxicological
effects on the animals exposed to the highest dose. Under the same
conditions (i.e. 220 mg/m3 for 4 weeks), racemic phenothrin did not
produce any adverse effects in male or female ICR mice. Similarly,
d-phenothrin also produced no adverse toxicological effects in
Sprague Dawley rats or ICR mice (both male and female) following
whole body exposure at 210 mg/m3 (particle size 5 µm) for 4 weeks
(Kohda et al., 1979b).
The acute oral and intraperitoneal toxicity of phenothrin
metabolites in rats and mice are shown in Table 6 and 7 (Kohda et
al., 1979a; FAO/WHO, 1981).
Table 6. The acute oral toxicity of phenothrin
metabolites
-----------------------------------------------------
Chemical No.a Species LD50
(mg/kg)
-----------------------------------------------------
3-Phenoxybenzyl alcohol 12 Rat 1330
3-Phenoxybenzaldehyde 13 Rat 600
-----------------------------------------------------
a Chemical identification number used in Fig. 2
Table 7. The acute intraperitoneal toxicity of several phenothrin
metabolites in mice
----------------------------------------------------------------------
Chemical No.a LD50 (mg/kg)
Male Female
----------------------------------------------------------------------
3-Phenoxybenzyl alcohol 12 371 424
3-(4'-hydroxyphenoxy)benzyl alcohol 14 750-1000 750-1000
3-(2'-hydroxyphenoxy)benzyl alcohol 15 876 778
3-Phenoxybenzoic acid 16 154 169
3-(4'-hydroxyphenoxy)benzoic acid 17 783 745
3-(2'-hydroxyphenoxy)benzoic acid 18 859 912
3-Phenoxybenzaldehyde 13 415 416
----------------------------------------------------------------------
a Chemical identification number used in Fig. 2
7.2. Long-Term Exposures and Carcinogenicity
Appraisal
There have been several long-term feeding studies with racemic
phenothrin or d-phenothrin (dose levels ranging from 200 to 10 000
mg/kg diet) in rats and mice, the length of exposure ranging from 6
months to 2 years. A slight increase in liver weight and a
significant difference in some clinical/chemistry parameters from those
of controls were observed at high doses. However, the no-observed-
effect level (NOEL) values obtained in these studies were high (300-
1000 mg/kg diet, corresponding to around 40-160 mg/kg body weight) and
no tumorigenicity was observed. Two feeding studies with d-phenothrin
(doses of 100-3000 mg/kg diet with exposure period 26 and 52 weeks) in
dogs revealed similar results, the NOEL being 300 mg/kg diet
(corresponding to around 7-8 mg/kg body weight per day). No
tumorigenicity relating to phenothrin feeding was detected in these
studies.
7.2.1. Rat
In a study by Murakami et al. (1981), [1R, cis]-phenothrin
(d-phenothrin) was administered to Sprague Dawley rats (20 of each
sex per group) at dose levels of 0, 1, 3, or 10 g/kg diet for 6
months. Ten rats of each sex per group were sacrificed after 3
months. d-Phenothrin had no significant effect on mortality,
clinical signs, ophthalmology, urinalysis, or gross and
histopathological findings. The serum albumin level was elevated
after 3 months in males fed 10 g/kg and in females fed 3 or 10
g/kg, and after 6 months in males fed 3 or 10 g/kg. The albumin-
globulin ratio was raised after 3 months in males fed 3 or 10 g/kg
and in females fed 10 g/kg, and in both males and females fed 10
g/kg after 6 months. Absolute and relative liver weights in both
males and females fed 3 or 10 g/kg were increased. Based on these
data, it was concluded that the NOEL for d-phenothrin in this study
was 1 g/kg diet for both sexes (55.4 mg/kg body weight per day for
males and 63.3 mg/kg body weight per day for females).
In a standard oncogenicity study, Fisher-344 rats (50 of each
sex per group) were fed d-phenothrin at dose levels of 0, 300, 1000
or 3000 mg/kg diet for at least 105 weeks in males and at least 118
weeks in females. Additional rats (30 of each sex per group) were
assigned to a chronic toxicity study with a 52-week interim
sacrifice. There were no significant effects on clinical signs,
mortality, food and water consumption, ophthalmology, blood
biochemistry, haematology, or urinalysis. However, the body weight
gain in females fed d-phenothrin at 3000 mg/kg was reduced, and the
relative liver weight was increased in females fed 3000 mg/kg for
52 weeks and, at the end of the oncogenicity study, in males fed
3000 mg/kg. Microscopic examination revealed that the incidence of
cystic dilatation of the sinuses of the mesenteric lymph nodes and
of periacinar hepatocytic hypertrophy was higher in males fed 3000
mg/kg for at least 105 weeks. d-Phenothrin did not show any
oncogenic activity to rats at up to 3000 mg/kg. Although at this
dose an increase in the incidence of adenomas and carcinomas of the
preputial gland was seen in males, the 1988 Joint FAO/WHO Meeting
on Pesticide Residues (FAO/WHO, 1988b) considered it unlikely that
this finding was of toxicological significance. The NOEL was 1000
mg/kg diet for both sexes (47 mg/kg body weight per day for males
and 56 mg/kg body weight per day for females) (Martin et al.,
1987).
When Sprague Dawley rats (50 of each sex per group) were fed a
diet containing racemic phenothrin (0, 200, 600, 2000, and 6000
mg/kg diet) for 2 years, body weight and food consumption were
slightly depressed at 6000 mg/kg in both males and females.
Ophthalmological examinations, haematological studies, urinalyses,
and clinical chemistry studies were performed at various intervals.
At the end of the study all animals were sacrificed and examined
for gross abnormalities, and extensive microscopic examinations
were conducted on a variety of tissues and organs. There were no
abnormal clinical or behavioral problems associated with
phenothrin administration. The survival rate of all groups of
treated rats was similar to that of controls. Males fed 6000 mg/kg
showed a significant increase in serum glutamine-pyruvate
aminotransferase activity. Ophthalmological examinations revealed
some abnormalities, all of which appeared to be age related.
Histopathological examination revealed no significant differences
between the treated groups and the control group with respect to
severity of lesions. No histopathological changes suggestive of
oncogenicity resulting from phenothrin treatment were found
(Hiromori et al., 1980).
7.2.2. Mouse
When Swiss White mice (50 of each sex per group) were fed
racemic phenothrin for 18 months at dose levels of 0, 300, 1000, or
3000 mg/kg diet, there were no significant effects on mortality,
clinical signs, haematologic values, clinical chemistry parameters,
or gross pathological findings. Slight body weight depression
occurred in males fed 3000 mg/kg, and increased liver weight was
found at the highest dose level in both males and females. There
was a statistically significant difference (compared with the
controls) in lung amyloidosis in the 1000 and 3000 mg/kg dose
groups, but no significant increase in tumours attributable to
phenothrin ingestion (Murakami et al., 1980).
In studies by Amyes et al. (1987), B6C3F1 hybrid mice (90 of
each sex per group) were fed d-phenothrin in the diet at dose
levels of 0, 300, 1000, or 3000 mg/kg. Fifty of each sex per group
were allocated to a standard oncogenicity study lasting 104 weeks.
The remaining mice were assigned to a chronic toxicity study, where
10 of each sex per group were sacrificed for interim study after 26
or 53 weeks and the remaining animals were examined after 78 weeks
of treatment. There were no compound-related effects on clinical
signs, mortality, ophthalmology, blood biochemistry, haematology,
or urinalysis. However, body weight gains for males fed d-
phenothrin at 3000 mg/kg were reduced and relative liver weights
were increased in both sexes fed 3000 mg/kg and in males receiving
1000 mg/kg. Microscopic examination revealed that the incidence of
periacinar hepatocyte hypertrophy with cytoplasmic eosinophilia was
higher in males fed 3000 mg/kg. The incidence of liver tumors
appeared higher in phenothrin-treated female mice than in control
females, but the difference was not statistically significant. It
was concluded, therefore, that administration of d-phenothrin to
mice for 2 years at dietary levels of up to 3000 mg/kg diet did not
significantly disturb the tumour burden or tumour profile of B6C3F1
hybrid mice. The NOEL in this study was 300 mg/kg diet for males
(40 mg/kg body weight per day) and 1000 mg/kg diet for females (164
mg/kg body weight per day).
7.2.3. Dog
When beagle dogs (six of each sex per group) were fed d-
phenothrin at dose levels of 0, 100, 300, or 1000 mg/kg diet for 26
weeks, there were no compound-related abnormal findings in
mortality, clinical signs, body weight, food consumption,
ophthalmology, gross or microscopic pathology, haematology, or
urinalysis studies. However, the alkaline phosphatase activity in
males fed 300 mg/kg and males and females fed 1000 mg/kg was
elevated and a slight increase in the mean relative liver weight in
males fed 1000 mg/kg was noted. The NOEL in this study was 300
mg/kg (Pence et al., 1981).
In a study by Cox et al. (1987), beagle dogs (four of each sex
per group) were fed d-phenothrin at dose levels of 0, 100, 300,
1000, or 3000 mg/kg diet for 52 weeks. There were no significant
effects on clinical signs, body weight, food consumption,
ophthalmology, or urinalysis. However, decreases in erythrocyte
count, haemoglobin concentration, haematocrit, and total blood
protein were noted in both male and female dogs fed 3000 mg/kg,
whereas mean absolute and relative liver weights increased.
Compound-related histopathological alterations were noted in the
adrenal glands and liver. Focal degeneration of the adrenal cortex
with cytoplasmic deposition of crystalline material was seen in one
male dog fed 1000 mg/kg and four dogs fed 3000 mg/kg. The chemical
nature or biological significance of this crystalline material was
not recorded. Hepatocytes appeared to enlargen slightly in one
male dog fed 1000 mg/kg and seven dogs fed 3000 mg/kg. The NOEL in
this study was 300 mg/kg diet for males and 1000 mg/kg for females
(8.24 and 26.77 mg/kg body weight per day for males and females,
respectively).
7.3. Mutagenicity
The results obtained in vivo and in vitro test system indicate
that d-phenothrin does not exhibit any mutagenic properties or
cause chromosomal or DNA damage.
In a DNA-repair test with Bacillus subtilis (M45 rec- and H17
wild type strains) using dose levels of up to 5 mg/disk per plate,
d-phenothrin did not inhibit the growth of any strain at any dose
level, whereas the positive control, mitomycin C, showed a clear
effect. The negative control gave a result similar to that of d-
phenothrin (Kishida & Suzuki, 1981a).
A mutagenicity test with Escherichia coli (WP2 uvr) and
Salmonella typhimurium (TA 1535, TA 1537, TA 1538, TA 98, and TA
100) using d-phenothrin at dose levels of up to 5 mg/plate with and
without metabolizing enzyme system (S9 mix) yielded negative
results, whereas a positive control gave a significant number of
mutants (Kishida & Suzuki, 1981b).
In a host-mediated assay using S. typhimurium G46 (indicator
bacteria), d-phenothrin in corn oil was given orally (twice with a
24-h interval) to groups of six male ICR mice at dose levels of
2500 or 5000 mg/kg body weight. Soon after the last administration,
each mouse was injected intraperitoneally with the indicator cells.
Three hours later, the bacterial mutation frequency in d-
phenothrin-treated mice was no greater than that in the control
group (Kishida & Suzuki, 1981c).
Suzuki et al. (1981) examined d-phenothrin for its ability to
induce chromosomal aberrations in vivo using bone marrow cells.
ICR mice were treated intrapertioneally with single doses of 2500,
5000, or 10 000 mg/kg body weight and sacrificed 6, 24, or 48 h
after treatment. No chromosomal aberrations as a result of d-
phenothrin treatment were detected.
In an in vitro chromosomal aberration test, Chinese hamster
ovary cells (CHO-K1) were treated with d-phenothrin (dose levels:
2 x 10-5 to 2 x 10-4 mol/litre for 24 and 48 h in the absence of S9
mix; 5 x 10-5 to 5 x 10-4 mol/litre for 6 h in the presence of S9
mix). No significant increase in the number of cells with
chromosomal aberrations was observed (Kogiso et al., 1986).
The ability of d-phenothrin to induce sister-chromatid
exchanges (SCEs) were tested in cultured mouse embryonic cells in
vitro. At doses of 10-5, 10-4, and 10-3 mol/litre (with and
without S9 mix), d-phenothrin did not induce any increase in the
frequency of SCEs (Suzuki & Miyamoto, 1981).
In a study of unscheduled DNA synthesis, Hela S3 cells were
treated with d-phenothrin at dose levels of 0, 0.25, 0.5, 1.0, 2.0,
or 4.0 mg/ml in the presence of 3H-thymidine (with and without S9
mix) for 3 h, and the incorporation of 3H-thymidine into DNA was
measured. There was no significant increase in the radioactivity
of DNA from cells treated with d-phenothrin (Foster et al., 1984).
7.4. Reproduction, Embryotoxicity, and Teratogenicity
No teratogenicity or reproductive effects were observed when
phenothrin was fed to rabbits or mice during the major
organogenesis period of gestation or to rats in 3-generation
reproduction studies.
7.4.1. Embryotoxicity and teratogenicity
In studies by Ladd et al. (1976), pregnant New Zealand White
rabbits (17 per group) were administered racemic phenothrin orally
at dose levels of 0, 3, 10, or 30 mg/kg body weight on days 6 to 18
of gestation. They were sacrificed on day 29 and the young
obtained by caesarian section were examined. At 30 mg/kg, the body
weight of females decreased during gestation, and there was a
slight decrease in the number of live young and a slight reduction
in fetal weight. Racemic phenothrin had no apparent teratogenic
effect, as shown by a lack of gross internal or external somatic
abnormalities and by normal fetal skeletal development following
prenatal exposure.
Pregnant New Zealand White rabbits (15 per group) were orally
administered d-phenothrin by intubation (0, 10, 100, or 1000 mg/kg
body weight per day) on days 6 to 18 of gestation, and were
sacrificed on day 29 or 30. Following caesarian section, 50% of
the pups were maintained for 24 h to evaluate survival. No
abnormalities were observed among the does (body weight, food
consumption, clinical observations, and necropsy) or foetuses
(implantation sites, corpora lutea, resorption sites, weight,
condition, and viability). Data on foetal survival and from
internal and external examinations for abnormalities showed not
significant effects from administrating d-phenothrin during
gestation (Rutter, 1974).
In studies by Nakamoto et al. (1973), d-phenothrin was orally
administered to pregnant ICR mice (17 or 18 per group) at dose
levels of 0, 30, 300, or 3000 mg/kg body weight on days 7 to 12 of
gestation (not covering the whole period of organogenesis). The
dams were sacrificed on day 18 of gestation and the pups were
obtained by caesarian section. Other mice (7 per group) were given
d-phenothrin at dose levels of 0, 300, or 3000 mg/kg to evaluate
postnatal effect. These mice were allowed to deliver naturally and
the pups were kept for 29 days. At these levels, d-phenothrin
showed no adverse effects, as indicated by maternal growth, fetal
mortality and external and internal examination of fetuses for
teratogenic or embryotoxic effects.
7.4.2. Reproduction studies
In a standard 3-generation (2 litters per generation)
reproduction study, groups of rats (8 male and 16 female Charles
River albino rats per group) were fed racemic phenothrin at dose
levels of 0, 200, 600, or 2000 mg/kg diet. Various reproduction
indices (i.e. mating index, fecundity index, male fertility index,
female fertility index, and incidence of parturition) were
measured. The adult rats showed no significant mortality or
complications during the study, and the reproductive parameters
revealed no significant dose-related adverse effects attributable
to phenothrin. Gross and microscopic findings indicated no adverse
effect resulting from dietary phenothrin. It was concluded that
phenothrin had no effect on reproduction (Takatsuka et al., 1980).
In a study by Tesh et al. (1978), d-phenothrin was fed to
Charles River CD rats (30 of each sex per group) at dose levels of
0, 300, 1000, or 3000 mg/kg diet throughout two successive
generations and up to the maturation of a third generation. At 300
and 1000 mg/kg, there was no adverse effect upon mortality, somatic
growth, development, or reproductive performance. At 3000 mg/kg,
mortality, body weight, and reproductive performance showed no
significant response to treatment, and selected F2 animals reared
to maturity were in all respects comparable with control rats.
However, F0 and F1 females and selected F2B male and female
weanlings showed a slight but consistent increase in the relative
liver weight. The NOEL in this study was 1000 mg/kg diet.
7.5. Neurotoxicity
Sprague Dawley rats exposed to d-phenothrin by inhalation at
concentratons of up to 3760 mg/m3 for 4 h showed no toxic signs as
a result of exposure. Histopathologically, there were no compound
related alterations in the sciatic nerve (Kohda et al., 1977).
When d-phenothrin was given to Sprague Dawley rats orally for 5
consecutive days (5 g/kg body weight per day), one out of ten
female rats died after four doses and signs of poisoning
(piloerections and urinary incontinence) were noted in several of
the animals. However, these signs disappeared rapidly at the end
of the treatment and there were no other signs of poisoning such as
leg weakness or ataxia. All animals were sacrificed 3 days after
the final dose, and histopathologically examination of the sciatic
nerve revealed minute changes in axon and myelin, characterized by
very slight axonal swelling, axonal disintegration, and/or
demyelination. Since there were similar changes in the control
animals, it was suggested that they were not due to the d-
phenothrin. It was considered that the oral administration of very
high doses of d-phenothrin does not lead to the neurotoxic effects
observed with several other pyrethroid esters (Okuno et al., 1978).
7.6. Miscellaneous Effects
No d-phenothrin-attributable pharmacological effects were
detected in various tests (e.g., spontaneous movement of isolated
guinea-pig ileum, contraction of the rat phrenic nerve diaphragm
preparation, cardiopulmonary physiology of anaesthetized cats,
coordination, and spontaneous movement of mice, and rectal
temperature of rats) at doses of 100-300 µg/ml in vitro, 3 mg/kg
intravenous, or 100-300 mg/kg intraperitoneal. A tentative arousal
response was recorded in the electroencephalogram of cats given d-
phenothrin (4 mg/kg) intraperitoneally, as is commonly observed in
animals given synthetic pyrethroids (Hara et al., 1974).
7.7. Mechanisms of Toxicity - Mode of Action
Some synthetic pyrethroids given intravenously to rats cause
either tremor (T-syndrome) or choreoathetosis with salivation (CS-
syndrome) (Verschoyl & Aldridge, 1980). However, d-phenothrin
(>600 mg/kg body weight) injected intravenously into the lateral
tail vein caused neither T-syndrome nor CS-syndrome, due to its
very low acute toxicity. From a study involving intracerebral
dosing with [1R, cis]- or [1R, trans]-phenothrin in mice, both
compounds were classified as Type I pyrethroids based on the
occurrence of tremors (Lawrence & Casida, 1982) and on
neurophysiological studies in cockroach cercal sensory nerves
(Gammon et al., 1981).
8. EFFECTS ON HUMANS
Although d-phenothrin has been used for more than 10 years, no
toxic effects and no cases of poisoning have been reported.
8.1. Clinical Studies
In a study by Hashimoto et al. (1980), d-phenothrin (talc
powder formulation with Span 80 as a stabilizer) was applied to the
head hair and pudenda hair of eight male human volunteers (three
times at intervals of 3 days) at a dose of 32 mg/man per
administration (0.44 to 0.67 mg/kg body weight per day). d-
Phenothrin powder was washed off 1 h after application. There were
no significant abnormalities due to d-phenothrin in terms of dermal
irritation, clinical signs, or blood biochemical and haematological
parameters. The blood levels of d-phenothrin were below the
detection limit of 0.006 mg/kg.
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has
discussed and evaluated d-phenothrin at its meetings in 1979, 1980,
1984, and 1988 (FAO/WHO, 1980, 1981, 1985a, 1988b).
Since 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg
body weight has been established.
In the WHO Recommended Classification of Pesticides by Hazard,
technical phenothrin is classified as unlikely to present an acute
hazard in normal use (WHO, 1988).
REFERENCES
AMYES, S.J., MARTIN, P.A., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Oncogenicity and toxicity study
in mice, Suffolk, United Kingdom, Life Science Research (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of syn-
thetic pyrethroids in fruit and vegetables by gas-liquid and high-
performance liquid chromatography. Analyst, 107: 206-212.
COX, R.H., SUTHERLAND, J.D., VOELKER, R.W., ALSAKER, R.D.,
VARGAS, K.J., LEWIS, S.A., & HAGEN, W.J. (1987) Chronic toxicity study
in dogs with Sumithrin technical grade, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
DESMARCHELIER, J.M. (1980) Comparative study of analytical methods for
bioresmethrin, phenothrin, d-phenothrin, pyrethrum I, carbaryl,
fenitrothion, methacrifos, pirimifos-methyl and dichlorfos on various
grains. J. Pestic. Sci., 5: 521-532.
DOI, T., SAKAUE, S., & HORIBA, M. (1985) Analysis of d-phenothrin and
its optical isomers in technical preparations and formulations. J.
Assoc. Anal. Chem., 68(5): 911-916.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC, American
Chemical Society, p. 229 (ACS Symposium Series No. 42).
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to a
synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
FAO (1982) Second Government Consultation on International Harmoniz-
ation of Pesticide Registration Requirements, Rome, 11-15 October,
1982, Food and Agriculture Organization of the United Nations.
FAO/WHO (1980) Pesticide residues in food: 1979 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 427-452
(FAO Plant Production and Protection Paper 20 Sup.).
FAO/WHO (1981) Pesticide residues in food: 1980 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 394-403
(FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1985a) Pesticide residues in food: 1984 Evaluations, Rome,
Food and Agriculture Organization of the United Nations, pp. 689-694
(FAO Plant Production and Protection Paper 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues, 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1988a) Pesticide residues in food: 1987 Evaluations. Part 1
- Residues, Rome, Food and Agriculture Organization of the United
Nations, pp. 109-113 (FAO Plant Production and Protection Paper 86.1).
FAO/WHO (1988b) Pesticide residues in food. Report of the 1988 Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues, Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper 92).
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous sensation
between synthetic pyrethroid insecticides. Contact Dermatitis, 13(3):
140-147.
FOSTER, R., TIPINS, R.S., DE VENEZIA, V., & NUNZIATA, A. (1984)
Unscheduled DNA synthesis in human cells, cell line: Hela S3, test
substance: Sumithrin, Suffolk, United Kingdom, Life Science Research,
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
FUJIMOTO, K., ITAYA, N., OKUNO, Y., KADOTA, T., & YAMAGUCHI,T. (1973)
A new insecticidal pyrethroid ester. Agric. biol. Chem., 37:2681-2682.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent class
antagonize GABA action at the crayfish neuromuscular junction.
Neurosci. Lett., 40:163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid toxi-
cology: Protective effects of diazepam and phenobarbital in the mouse
and the cockroach. Toxicol. appl. Pharmacol., 66: 290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural variations
affecting pyrethroid neurotoxicity. Neurobehav. Toxicol. Teratol., 4(6):
793-799.
HARA, Y., MIYAGISHI, A., OHTSUKA, M., ASAMI, Y., KUROKAWA, Y.,
& MIYAMOTO, J. (1974) Pharmacological study of Sumithrin: Effects on
respiration, blood pressure, and peripheral central nervous systems
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HASHIMOTO, S., OKANO, S., YAMADA, H., & MIYAMOTO, J. (1980)
Human volunteer study with phenothrin and d-phenothrin
powder (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
HAYASHI, A. (1977) [Safe use of insecticides (I)] [Bo-satsu-kyo News,]
No. 1 p. 10 (June) (in Japanese).
HIROMORI, T., KOYAMA, Y., OKUNO, Y., ARAI, M., ITO, N., &
MIYAMOTO, J. (1980) Two-year chronic toxicity study of S-2539 in
rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
HIROMORI, T., HARA, S., KADOTA, T., & MIYAMOTO, J. (1984) Acute
intravenous toxicity of phenothrin and d-phenothrin in mice
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
ISOBE, N., MATSUNAGA, H., NAKATSUKA, I., & YOSITAKE, A.
(1987) Metabolism of [1R, trans]- and [1R, cis]- isomers of phenothrin
in rats (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
ITAYA, N., KAMOSHITA, K., KITAMURA, S., OKUNO, Y., MIZUTANI,
T., NAKAI, S., KAMEDA, N., & FUJIMOTO, K. (1969) Esters of cyclo-
propanecarboxylic acids. Ger. Offen., 1: 926, 433.
IZUMI, T., KANEKO, H., MATSUO, M., & MIYAMOTO, J. (1984)
Comparative metabolism of the six optical stereo isomers of phenothrin
in rats and mice. J. Pestic. Sci., 9: 259-267.
KANEKO, H., OHAKAWA, H., & MIYAMOTO, J. (1981) Absorption and
metabolism of dermally applied phenothrin in rats. J. Pestic. Sci., 6:
169-182.
KISHIDA, F. & SUZUKI, H. (1981a) Studies on DNA-damaging capacity of
Sumithrin with Bacillus subtilis (Unpublished Technical Report No. ET-
10-0069 submitted to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA. F. & SUZUKI, H. (1981b) Gene mutation test of Sumithrin in
bacterial system (Unpublished Technical Report No. ET-10-0068 submitted
to WHO by Sumitomo Chemical Co., Ltd).
KISHIDA, F. & SUZUKI, H. (1981c) Mutagenicity test of Sumithrin in
host-mediated assay (Unpublished Technical Report No. ET-10-0067 sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
KOGISO, S., HARA, M., ITO, K., IWAWAKI, H., & YOSHITAKE, A.
(1986) In vitro chromosomal aberration test of S-2539F in Chinese
hamster ovary cells (CHO-K1) (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1977) Acute
inhalation toxicity of d-phenothrin (S-2539 Forte) in rats (Unpublished
report submitted to WHO by Sumitomo Chemical Co., Ltd).
KOHDA, H., KENEDO, H., OHKAWA, H., KADOTA, T., & MIYAMOTO, J.
(1979a) Acute intraperitoneal toxicity of permethrin metabolites in
mice (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
KOHDA, H., NISHIMOTO, K., KADOTA, K., & MIYAMOTO, J. (1979b)
Acute and subacute inhalation toxicity studies of S-2539 Forte in rats
and mice (Unpublished report submitted to WHO by Sumitomo Chemical
Co., Ltd).
KOHDA, H., OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1980) Particle
size distribution and mean particle size in acute inhalation toxicity
study of d-phenothrin (S-2539 forte) in rats (Unpublished Technical
Report No. ET-00-0054 submitted to WHO by Sumitomo Chemial Co.,
Ltd).
LADD, R., SMITH, P.S., JENKINS, D.H., KENNEDY, G.L., Jr, KINOSHITA,
F.K., & KEPLINGER, M.L. (1976) Teratogenic study with S-2539 in albino
rabbits, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: mouse
intracerebral structure-toxicity relationship. Pestic. Biochem. Physiol.,
18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions of
pyrethroid insecticides with chloride ionophore-associated binding
sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticides, London, Taylor &
Francis Ltd, p. 440.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel modifi-
cation as the basis for the variation in the nerve membrane effects of
pyrethroids and DDT analogs. Pestic. Biochem. Physiol., 20: 203-216.
MARTIN, P.A., AMYES, S.J., ASHBY, R., LEE, P., BROWN, P.M., FOWLER,
J.S.L., & FINN, J.P. (1987) Sumithrin: Combined toxicity and
oncogenicity study in rats, Suffolk, United Kingdom, Life Science
Research (Unpublished report submitted to WHO by Sumitomo Chemical Co.,
Ltd).
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of synthetic
pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue analysis of
synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry-human welfare
and the environment. Proceedings of the Fifth International Congress of
Pesticide Chemistry, Kyoto, Japan, 29 August to 4 September, 1982, Oxford,
Pergamon Press, Vol. 1-4.
MIYAMOTO, J., SUZUKI, T., & NAKAE, C. (1974) Metabolism of phenothrin
or 3-phenoxybenzyl d-trans-chrysanthemate in mammals. Pestic. Biochem.
Physiol., 4: 438-450.
MIYAMOTO, J., KOGISO, S., & MATSUO, M. (1984) Additional toxicological
information for phenothrin (Unpublished Technical Report No. ET-40-0090
from Sumitomo Chemical Co., Ltd).
MULLA, M.S., DARWAZEH, H.A., & DHILLON, M.S. (1980) New
pyrethroids as mosquito larvicides and their effects on nontarget
organisms. Mosq. News, 40: 6-12.
MURAKAMI, M., HIROMORI, T., ITO, S., & HOSOKAWA, S. (1981)
Six month oral toxicity study of S2539 Forte (Sumithrin(R)) in rats
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURAKAMI, J., ITO, S., OKUNO, Y., ARAI, M., ITO, N., & MIYAMOTO, J.
(1980) Eighteen-month chronic oral toxicity and tumorigenicity study of
S-2539 in mice, Northbrook, Illinois, Industrial Bio-Test Laboratories
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
MURANO, A. (1972) Determination of the optical isomers of insecticidal
pyrethroids by gas-liquid chromatography. Agric. biol. Chem., 36:
2203-2211.
NAKAMOTO, N., KATO, T., & MIYAMOTO, J. (1973) Teratogenicity study
of S-2539 Forte in mice (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
NAMBU, K., OHKAWA, H., & MIYAMOTO, J. (1980) Metabolic fate of
phenothrin in plants and soils. J. Pestic. Sci., 5: 177-197.
NAMBU, K., TAKIMOTO, Y., & MIYAMOTO, J. (1981) Degradation of
phenothrin in stored wheat grains. J. Pestic. Sci., 6: 183-191.
NIOSH (1983) Registry of toxic effects of chemical substances, 1981-
1982 edition, Cincinnati, Ohio, US National Institute for Occupational
Safety and Health, p. 1003.
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
OHNISHI, J., IKEDA, M., SUZUKI, Y., & MATSUDA, T. (1987) Residue
analysis of d-phenothrin, fenitrothion and piperonyl butoxide in stored
wheat, wheat milling fractions and bread (interim report) (Unpublished
Report No. ER-RD-8719 submitted to WHO by Sumitomo Chemical Co.,
Ltd).
OKUNO, Y., KADOTA, T., & MIYAMOTO, J. (1978) Neurotoxicity study
of d-phenothrin (S-2539 Forte(R)) in rats by repeated oral administration
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
PAPADOPOULOU-MOURKIDOU, E., IWATA, Y., & GUNTHER, F.A.
(1981) Utilization of an infrared detector for selective liquid
chromatographic analysis. 2. Formulation analysis of the pyrethroid
insecticides allethrin, decamethrin, cypermethrin, phenothrin and
tetramethrin. J. agric. food Chem., 29: 1105-1111.
PENCE, D.H., HAGEN, W.H., WLSAKER, R.D., HASTINGS, T.F.,
DAWKINS, B.G., TACEY, R.L., & MARSHALL, P.M. (1981) Subchronic
toxicity study of S2539-F in dogs, Vienna, Virginia, Hazleton
Laboratories Inc. (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids on a
nerve-muscle preparation of the clawed frog, Xenopus laevis. Pestic.
Biochem. Physiol., 25: 176-187.
RUTTER, H.A. (1974) Teratogenicity study in rabbits: S-2539 Forte,
Vienna, Virginia, Hazleton Laboratories Inc. (Unpublished report sub-
mitted to WHO by Sumitomo Chemical Co., Ltd).
RUZO, L.O., SMITH, I.H., & CASIDA, J.E. (1982) Pyrethroid
photochemistry: photooxidation reactions of the chrysanthemates,
phenothrin and tetramethrin. J. agric. food Chem., 30: 110-115.
SAKAUE, S., KITAJIMA, M., HORIBA, M., & YAMAMOTO, S. (1981) Gas
chromatographic and high performance liquid chromatographic
determination of d-phenothrin (Sumithrin) in formulations. Agric. biol.
Chem., 45: 1135-1140.
SEGAWA, T. (1976) Acute toxicity study of S-2539 in rats and mice,
Hiroshima, Hiroshima University School of Medicine (Unpublished report
submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979a) Acute oral, subcutaneous, dermal and intraperitoneal
toxicity studies of S-2539 Forte in rats, Hiroshima, Hiroshima
University School of Medicine (Unpublished Technical Report
No. ET-31-0033 submitted to WHO by Sumitomo Chemical Co., Ltd).
SEGAWA, T. (1979b) Acute oral, subcutaneous, dermal and
intraperitoneal toxicity studies of S-2539 Forte in mice, Hiroshima,
Hiroshima University School of Medicine (Unpublished Technical Report
No. ET-31-0032 submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H. & MIYAMOTO, J. (1981) In vitro sister chromatid
exchanges of Sumithrin (Unpublished Technical Report No. ET-10-0071
submitted to WHO by Sumitomo Chemical Co., Ltd).
SUZUKI, H., HARA, M., & MIYAMOTO, J. (1981) In vivo
chromosomal aberration test of Sumithrin on bone marrow cells of
mice (Unpublished Technical Report No. ET-10-0072 submitted to WHO by
Sumitomo Chemical Co., Ltd).
SUZUKI, T. & MIYAMOTO, J. (1978) Purification and properties of
pyrethroid carboxyesterase in rat liver microsome. Pestic. Biochem.
Physiol., 8: 186-198.
SUZUKI, T., OHNO, N., & MIYAMOTO, J. (1976) New metabolites of
(+)-cis-phenothrin, 3-phenoxybenzyl (+)-cis chrysanthemate, in rats.
J. Pestic. Sci., 1: 151-152.
TAKATSUKA, M., OKUNO, Y., SUZUKI, T., KADOTA, T., YASUDA, M.,
& MIYAMOTO, J. (1980) Three-generation reproduction study of S-2539
in rats, Northbrook, Illinois, Industrial Bio-Test Laboratories Inc.
(Unpublished report submitted to WHO by Sumitomo Chemical Co., Ltd).
TAKIMOTO, Y., KATO, T., & MIYAMOTO, J. (1977) Residue analyses
of phenothrin (S-2539) in crops (Unpublished report submitted to WHO by
Sumitomo Chemical Co., Ltd).
TESH, J.M., WILLOUGHBY, C.R., & FOWLER, J.S.L. (1987) Sumithrin:
Effects upon reproductive performance of rats treated continuously
throughout two successive generations, Suffolk, United Kingdom, Life
Science Research (Unpublished report submitted to WHO by Sumitomo
Chemical Co., Ltd).
TURNBULL, S.R. & ARDLEY, J.H. (1987) Determination of d-phenothrin,
piperonyl butoxide and fenitrothion residues on wheat, derived
fractions and breads, at approximately one, three and six months post-
treatment, Australia, The Wellcome Foundation Ltd (Unpublished Report
No. AESH 87-11).
VAN DEN BERCKEN, J. (1977) The action of allethrin on the peripheral
nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels in
frog myelinated nerve membrane. In: Insect: Neurobiology and pesticide
action, London, Society of Chemical Industry, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER
ZALM, J.M. (1973) DDT-like action of allethrin in the sensory nervous
system of Xenopus laevis. Eur. J. Phamacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A.
(1979) Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VERSCHOYLE, R.D. & ALDRIDGE, W.N.(1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45: 325-
329.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Phamacol., 58: 501-504.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system. Neuropathol.
appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J.
(1982a) Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed frog,
Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN
BERCKEN, J. (1982b) Similar mode of action of pyrethroids and DDT on
sodium channel gating in mylinated nerves. Nature (Lond.), 295: 601-
603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF,
R.G.D.M., & VAN DEN BERCKEN, J. (1983) Temperature and structure-
dependent interaction of pyrethroids with the sodium channels in frog
node of Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WHO (1979) WHO Technical Report Series, No. 634 (Safe use of
pesticides. Third Report of the WHO Expert Committee on Vector Biology
and Control), pp 18-23.
WHO (1988) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1988-1989, WHO, World Health
Organization (WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1987) d-Phenothrin. In: The pesticide
manual, 8th ed., Croydon, British Crop Protection Council, pp. 654-
655.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
APPENDIX
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs ( Xenopus laevis; Rana temporaria, and
Rana Escuelenta) it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these two classes of pyrethroids has been made
on the basis of the symptoms of toxicity in mammals and insects
(Van den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge,
1980; Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group (allethrin, d-
phenothrin, permethrin, tetramethrin, cismethrin, and bioresmethrin)
(Type I: T-syndrome).
The pyrethroids that do not contain an alpha-cyano group give
rise to pronouced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3-10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organs and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5°C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of the "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 milliseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently, it was demonstrated that allethrin and DDT have
essentially the same effect on sodium channels in frog myelinated
nerve membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittently, cause large
depolarizing after-potentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl alcohol
(deltamethrin, cypermethrin, fenvalerate, and fenpropanate) (Type II:
CS-syndrome).
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependant depression of the
nervous impulse, brought about by a progressive depolarization
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, persumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
The results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
crayfish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered
pyrethroids closely approximates that of the convulsant picrotoxin
(PTX). Deltamethrin inhibits the binding of the [3H]-
dihydropicrotoxin to rat brain synaptic membranes, whereas the non-
toxic R epimer of deltamethrin is inactive. These findings suggest
a possible relation between the Type II pyrethroid action and the
GABA receptor complex. The stereospecific correlation between the
toxicity of Type II pyrethroids and their potency to inhibit the
[35S]-TBPS binding was established using a radioligand, [35S]= t-
butylbicyclophosphorothionate [35S]-TBPS. Studies with 37
pyrethroids revealed an absolute correlation, without any false
positive or negative, between mouse intracerebral toxicity and in
vitro inhibition: all toxic cyano compounds including deltamethrin,
1R, cis-cypermethrin, 1R, trans-cypermethrin, and [2S,alpha]-
fenvalerate were inhibitors, but their non-toxic stereoisomers were
not; non-cyano pyrethroids were much less potent or were inactive
(Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S,alphaS]-fenvalerate) increased the input resistance of crayfish
claw opener muscle fibres bathed in GABA. In contrast, two non-
insecticidal stereoisomers and Type I pyrethroids (permethrin,
resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog ( Xenopus laevis ). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action. It
may be related to the long-lasting repetitive activity in sense
organs and possibly in other parts of the nervous system, which, in
a more advance state of poisoning, may be accompanied by a
frequency-dependent depressoin of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vunerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
RESUME, EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1. Résumé et évaluation
1.1 Identité, propriétés physiques et chimiques, et méthodes
d'analyse
La phénothrine racémique a été synthétisée pour la première
fois en 1969. Du point de vue chimique, il s'agit de l'ester de
l'acide diméthyl-2,2 (diméthyl-2,2 vinyl)-3 cyclopropanecarboxylique
et de l'alcool phénoxy-3 benzylique (PBalc). Il se présente sous
la forme d'un mélange de quatre stéréoisomères : [1R,trans],
[1R,cis], [1S,trans], [1S,cis]. La d-phénothrine est un mélange
d'une partie d'isomère [1R,cis] pour quatre parties d'isomère
[1R,trans], et elle est à l'heure actuelle le seul produit
technique sur le marché. C'est l'isomère [1R,trans] qui est
l'insecticide le plus actif; vient ensuite l'isomère [1R,cis].
La d-phénothrine de qualité technique se présente sous la forme
d'un liquide jaune pâle à brun jaune et son degré de pureté est de
92,5 à 94,5%. Sa densité est de 1,058-1,061 à 25°C et sa tension
de vapeur de 0,16 mPa à 20°C. Elle est difficilement soluble dans
l'eau (2 mg par litre à 25°C) mais soluble dans les solvants
organiques tels que l'acétone, le xylène et l'hexane. Elle est
assez stable à l'air mais instable à la lumière, encore que sa
photodégradation ne soit pas aussi rapide que celle des pyréthrines
naturelles. Elle est instable en milieu alcalin.
Le dosage des résidus peut s'effectuer par chromatographie en
phase liquide à haute performance avec détecteur ultra-violet, la
concentration minimale décelable étant de 0,05 mg par kg. Pour
l'analyse du produit technique on utilise la chromatographie en
phase gazeuse avec détection par ionisation de flamme.
1.2 Production et usage
La d-phénothrine est utilisée depuis 1977. On estime que l'on
utilise chaque année 70 à 80 tonnes de d-phénothrine dans le monde,
essentiellement pour détruire les insectes incommodants dans les
habitations, lutter contre les vecteurs de maladies et protéger les
céréales ensilées; le produit est utilisé seul ou en association
avec d'autres insecticides ou synergisants. Il est présenté sous
forme d'huiles pour aérosols, de poudres ou de concentrés
émulsionnables. La d-phénothrine est également utilisée pour
détruire les poux de l'homme, auquel cas elle est présentée sous
forme de poudre, de shampooing ou de lotion.
1.3 Exposition humaine
Les aérosols classiques à usage domestique ne devraient pas
conduire à des concentrations atmosphériques de d-phénothrine
supérieures à 0,5 mg/m3. Dans le blé ensilé, on peut trouver des
résidus allant jusqu'à 4 mg/kg, mais ces teneurs tombent à 0,8
mg/kg dans la farine après mouture, et à 0,6 mg/kg après
panification.
Pour détruire les poux, on applique la d-phénothrine sur la
chevelure, par exemple en trois doses de 32 mg tous les trois
jours. Il n'existe aucune donnée sur l'exposition professionnelle à
la d-phénothrine.
L'exposition de la population dans son ensemble devrait être
très faible, mais on manque de données précises sur ce point.
1.4 Destinée dans l'environnement
La phénothrine se dégrade facilement, sa demi-vie étant
inférieure à un jour sur les végétaux et autres surfaces. La d-
phénothrine ou ses produits de dégradation ne migrent que très peu
vers les zones non traitées des végétaux. On a constaté que des
haricots ne captaient que dans une faible mesure les prouits
radiomarqués provenant de sols traités par de la phénothrine
marquée au carbone-14. Après traitement des sols avec de la
[1R, trans ]- ou de la [1R, cis]-phénothrine à raison de 1 mg/kg, on
a constaté que les deux isomères se décomposaient rapidement, la
demi-vie initiale étant de un à deux jours, mais que, en cas
d'inondation, la dégradation était considérablement ralentie, la
demi-vie initiale étant de deux à quatre semaines pour l'isomère
trans et de un à deux mois pour l'isomère cis. On a observé que
les isomères trans ou cis de la phénothrine se déplaçaient
relativement peu (environ 2%) à travers des colonnes de terre,
lorsque le lessivage commençait immédiatement ou deux semaines
après le traitement.
En général, la dégradation qui se produit dans l'environnement
conduit à des produits moins toxiques.
1.5 Cinétique et métabolisme
Après avoir reçu une dose unique ou des doses répétées de
phénothrine radiomarquée par voie orale ou percutanée, des rats ont
rapidement et presque complètement excrété la fraction marquée dans
leurs urines et leurs déjections en trois à sept jours. Les
principales voies métaboliques des isomères cis et trans chez le
rat consistent en une rupture de la liaison ester et l'oxydation en
position 4 du reste alcool ou du groupement isobutényle du reste
acide. Les métabolites résultant du clivage de l'ester (qui sont
excrétées essentiellement dans les urines) constituent les
principaux produits de dégradation de l'isomère trans alors que les
métabolites restant sous forme d'ester (excrétées essentiellement
dans les déjections) proviennent pour la plupart de l'isomère cis.
1.6 Effets sur les êtres vivant dans leur milieu naturel
La phénothrine a été expérimentée sur quelques groupes
d'organismes non visés et dans chaque groupe sur quelques espèces
seulement. Chez les poissons, la CL50 à 96 heures de la
phénothrine racémique et des stéréoisomères (1R) va de 17 à 200
microgrammes par litre. Une étude, portant sur des invertébrés
aquatiques, a montré que chez Daphnia pulex la CL50 à 3 heures
était de 25 à 50 mg/litre pour tous les isomères et la phénothrine
racémique.
Une seule et unique étude au cours de laquelle de la
phénothrine a été appliquée à des étangs n'a révélé aucun effet sur
les arthropodes aquatiques.
La toxicité pour les oiseaux est faible, avec une DL50 aiguë
par voie orale supérieure à 2500 mg/kg de poids corporel chez le
colin de Virginie et une CL50 par voie alimentaire supérieure à
5000 mg/kg de nourriture chez ce volatile et chez le colvert.
Etant donné que la phénothrine se dégrade rapidement à la
lumière solaire et qu'on l'utilise principalement pour traiter des
céréales ensilées, l'exposition environnementale est
vraisemblablement très faible. Dans ces conditions, des effets sur
l'environnement sont tout-à-fait improbables.
1.7 Effets sur les animaux d'expérience et sur les systèmes
d'épreuve in vitro
La toxicité aiguë de la d-phénothrine est extrêmement faible,
la DL50 étant supérieure à 5000 mg/kg de poids corporel chez le rat
et la souris (par voie orale, sous-cutanée, dermique et
intrapéritonéale) et la CL50 inhalatoire supérieure à 3760 mg/m3
chez le rat. Le syndrome d'intoxication se caractérise par une
hyperexcitabilité, une prostration, des tremblements, de l'ataxie
et une paralysie. Sur la base de ces symptômes et d'après les
résultats des études électrophysiologiques sur les nerfs sensoriels
des cerques de la blatte, la phénothrine est classée parmi les
pyréthroides du type I.
Exposés à de la d-phénothrine par inhalation à des
concentrations allant jusqu'à 210 mg/m3, quatre heures par jour
pendant quatre semaines, ou par voie orale cinq jours de suite à
raison de 5000 mg/kg de poids corporel, des rats n'ont présenté
aucun effet toxicologique indésirable.
Plusieurs études d'alimentation ont été effectuées sur des rats
et des souris recevant de la d-phénothrine ou de la phénothrine
racémique à des doses allant de 200 à 10 000 mg par kg de
nourriture; la période d'exposition allait de six mois à deux ans.
Ces études ont permis d'établir une dose sans effet observable
allant de 300 à 1000 mg par kg de nourriture, ce qui correspond à
peu près à 40-160 mg/kg de poids corporel et par jour. Chez des
chiens à qui l'on avait administré de la d-phénothrine à des doses
de 100 à 3000 mg/kg de nourriture avec des périodes d'exposition de
26 à 52 semaines, deux études ont permis d'obtenir une dose sans
effet observable de 300 mg par kg de nourriture, soit 7 à 8 mg/kg
de poids corporel et par jour.
Divers systèmes permettant d'étudier in vivo et in vitro les
mutations géniques, les lésions et les réparations de l'ADN ainsi
que les effets chromosomiques, ont permis de constater que la d-
phénothrine n'avait pas d'effet mutagène.
Des études de deux ans ont également montré que la d-
phénothrine n'était pas cancérogène pour le rat ni la souris à des
doses allant jusqu'à 3000 mg par kg de nourriture. Aucune
tératogénicité ni embryotoxicité n'a été observée chez des foetus
de lapins et de souris dont les mères avaient reçu de la d-
phénotrine à des doses allant jusqu'à 1000 et 3000 mg par kg de
poids corporel, respectivement. Lors d'une étude de reproduction
chez le rat, portant sur deux générations, on a établi que la dose
sans effet observable était de 1000 mg par kg de nourriture.
Des rats exposés par inhalation à des doses très élevées de d-
phénothrine (jusqu'à 3760 mg/m3) pendant quatre heures ou,
quotidiennement, par voie orale, à une dose de 5000 mg par kg de
poids corporel, cinq jours durant, n'ont présenté aucune
dégénérescence myélinique ni désagrégation de l'axone au niveau du
nerf sciatique.
1.8 Effets sur les êtres humains
La d-phénothrine est utilisée depuis plus de 10 ans sans que
l'on ait signalé d'intoxication humaine.
Rien n'indique que cette substance puisse avoir des effets
nocifs sur l'homme pour peu qu'elle soit utilisée conformément aux
recommandations.
2. Conclusions
2.1 Population générale
L'exposition de la population dans son ensemble à la d-
phénothrine est vraisemblablement très faible et il n'y a
probablement aucun risque à cet égard si le produit est utilisé
conformément aux recommandations.
2.2 Exposition professionnelle
Si elle est utilisée de manière convenable, moyennant un
certain nombre de mesures d'hygiène et de sécurité, la d-
phénothrine ne devrait pas présenter de risque pour les personnes
qui lui sont exposées de par leur profession.
2.3. Environnement
Du fait qu'elle se dégrade rapidement à la lumière solaire et
qu'elle est principalement utilisée pour traiter les céréales
ensilées, l'exposition environnementale à la phénothrine est
vraisemblablement très faible. Dans ces conditions tout effet sur
l'environnement est extrêmement improbable.
3. Recommandations
Les niveaux d'exposition résultant d'une utilisation conforme
aux recommandations sont censés être extrêmement faibles, toutefois
on pourrait envisager de confirmer cette hypothèse en étendant la
surveillance à la d-phénothrine.
