
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 199
Chlordimeform
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
Environmental Health Criteria 199
First draft prepared by Dr P.J. Abbott, Australia and New Zealand Food
Authority, Canberra, Australia
Published under the joint sponsorship of the United Nations
Environment Programme, the International Labour Organisation, and the
World Health Organization
World Health Organization
Geneva, 1998
The International Programme on Chemical Safety (IPCS) is a joint
venture of the United Nations Environment Programme, the International
Labour Organisation, and the World Health Organization. The main
objective of the IPCS is to carry out and disseminate evaluations of
the effects of chemicals on human health and the quality of the
environment. Supporting activities include the development of
epidemiological, experimental laboratory, and risk-assessment methods
that could produce internationally comparable results, and the
development of manpower in the field of toxicology. Other activities
carried out by the IPCS include the development of know-how for coping
with chemical accidents, coordination of laboratory testing and
epidemiological studies, and promotion of research on the mechanisms
of the biological action of chemicals.
WHO Library Cataloguing in Publication Data
(Environmental health criteria ; 199)
1.Chlorphenamidine - toxicity 2.Chlorphenamidine - adverse effects
3.Environmental exposure 4.Occupational exposure
I.International Programme on Chemical Safety II.Series
ISBN 92 4 157199 3 (NLM Classification: QU 61)
ISSN 0250-863X
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1998
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved. The designations
employed and the presentation of the material in this publication do
not imply the expression of any opinion whatsoever on the part of the
Secretariat of the World Health Organization concerning the legal
status of any country, territory, city or area or of its authorities,
or concerning the delimitation of its frontiers or boundaries. The
mention of specific companies or of certain manufacturers' products
does not imply that they are endorsed or recommended by the World
Health Organization in preference to others of a similar nature that
are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
PREAMBLE
ABBREVIATIONS
1. SUMMARY
1.1. Identity, physical and chemical properties, and analytical
methods
1.2. Sources of human and environmental exposure
1.3. Environmental transport, distribution and transformation
1.4. Environmental levels and human exposure
1.5. Kinetics and metabolism in laboratory animals and humans
1.6. Effects on laboratory mammals and in vitro test systems
1.7. Effects on humans
1.8. Effects on other organisms in the laboratory and field
1.9. Evaluation of human health risks and effects on the
environment
1.10. Conclusions and recommendations
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL
METHODS
2.1. Identity
2.2. Physical and chemical properties
2.3. Conversion factors
2.4. Analytical methods
2.4.1. Plants
2.4.2. Soil
2.4.3. Water
2.4.4. Formulations
2.4.5. Air
2.4.6. Urine
2.4.7. Tissues
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Natural occurrence
3.2. Anthropogenic sources
3.2.1. Production levels and processes
3.2.2. Uses
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1. Transport and distribution between media
4.1.1. Air
4.1.2. Water
4.1.3. Soil
4.1.4. Vegetation and wildlife
4.1.5. Entry into food chain
4.2. Biotransformation
4.2.1. Degradation in plants
4.2.2. Degradation in soils
4.2.3. Bioaccumulation
4.3. Ultimate fate following use
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1. Environmental levels
5.1.1. Air and water
5.1.2. Soil
5.2. General population exposure
5.2.1. Environmental sources
5.2.2. Residues in raw produce
5.2.3. Residues in processed food
5.3. Occupational exposure during manufacture, formulation
or use
5.3.1. Exposure during manufacture and formulation
5.3.2. Exposure during use
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1. Absorption, distribution and excretion
6.1.1. Mouse and rat
6.1.2. Other species
6.1.3. Human
6.2. Metabolic transformation
6.2.1. Mouse and rat
6.2.2. Other species
6.2.3. In vitro studies
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1. Single exposure
7.1.1. Oral
7.1.2. Other routes
7.2. Short-term exposure
7.2.1. Dietary
7.2.1.1 Mouse
7.2.1.2 Rat
7.2.1.3 Dog
7.2.2. Intubation
7.2.2.1 Rat
7.3. Long-term dietary exposure
7.3.1. Mouse
7.3.2. Rat
7.4. Skin and eye irritation; skin sensitization
7.5. Reproductive toxicity, embryotoxicity and
teratogenicity
7.5.1. Reproductive toxicity
7.5.1.1 Rat
7.5.1.2 Hamster
7.5.2. Embryotoxicity and teratology
7.5.2.1 Rat
7.5.2.2 Rabbit
7.6. Mutagenicity and related end-points
7.6.1. DNA damage and repair
7.6.2. Mutation
7.6.3. Chromosome damage
7.6.4. Cell transformation
7.7. Carcinogenicity
7.7.1. Mouse
7.7.2. Rat
7.8. Other special studies
7.8.1. Immunotoxicity
7.8.2. Behavioural effects
7.8.3. Pharmacological and biochemical effects
7.9. Factors modifying toxicity
7.10. Mechanisms of toxicity - mode of action
7.10.1. Mechanism of acute toxicity
7.10.2. Mechanism of carcinogenicity
8. EFFECTS ON HUMANS
8.1. General population exposure
8.1.1. Acute poisoning incidents
8.2. Occupational exposure
8.2.1. Acute poisoning incidents
8.2.2. Effects of long-term exposure
8.2.3. Epidemiological studies
8.2.3.1 4-Chloro- o-toluidine
8.2.3.2 Chlordimeform
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1. Laboratory experiments
9.1.1. Microorganisms
9.1.2. Aquatic organisms
9.1.2.1 Plants
9.1.2.2 Invertebrates
9.1.2.3 Vertebrates
9.1.3. Terrestrial organisms
9.1.3.1 Plants
9.1.3.2 Invertebrates
9.1.3.3 Vertebrates
9.2. Field observations
9.2.1. Microorganisms
9.2.2. Aquatic organisms
9.2.3. Terrestrial organisms
9.2.3.1 Plants
9.2.3.2 Invertebrates
9.2.3.3 Vertebrates
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1. Evaluation of human health risks
10.1.1. Exposure
10.1.2. Toxicity
10.1.3. Risk evaluation
10.2. Evaluation of effects on the environment
11. CONCLUSIONS AND RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
AND THE ENVIRONMENT
11.1. Conclusions
11.2. Recommendations for protection of human health and the
environment
12. FURTHER RESEARCH
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
R�SUM�
RESUMEN
NOTE TO READERS OF THE CRITERIA MONOGRAPHS
Every effort has been made to present information in the criteria
monographs as accurately as possible without unduly delaying their
publication. In the interest of all users of the Environmental Health
Criteria monographs, readers are requested to communicate any errors
that may have occurred to the Director of the International Programme
on Chemical Safety, World Health Organization, Geneva, Switzerland, in
order that they may be included in corrigenda.
* * *
A detailed data profile and a legal file can be obtained from the
International Register of Potentially Toxic Chemicals, Case postale
356, 1219 Ch�telaine, Geneva, Switzerland (telephone no. + 41 22 -
9799111, fax no. + 41 22 - 7973460, E-mail irptc@unep.ch).
* * *
This publication was made possible by grant number
5 U01 ES02617-15 from the National Institute of Environmental Health
Sciences, National Institutes of Health, USA, and by financial support
from the European Commission.
Environmental Health Criteria
PREAMBLE
Objectives
In 1973 the WHO Environmental Health Criteria Programme was
initiated with the following objectives:
(i) to assess information on the relationship between exposure to
environmental pollutants and human health, and to provide
guidelines for setting exposure limits;
(ii) to identify new or potential pollutants;
(iii) to identify gaps in knowledge concerning the health effects of
pollutants;
(iv) to promote the harmonization of toxicological and
epidemiological methods in order to have internationally
comparable results.
The first Environmental Health Criteria (EHC) monograph, on
mercury, was published in 1976 and since that time an ever-increasing
number of assessments of chemicals and of physical effects have been
produced. In addition, many EHC monographs have been devoted to
evaluating toxicological methodology, e.g., for genetic, neurotoxic,
teratogenic and nephrotoxic effects. Other publications have been
concerned with epidemiological guidelines, evaluation of short-term
tests for carcinogens, biomarkers, effects on the elderly and so
forth.
Since its inauguration the EHC Programme has widened its scope,
and the importance of environmental effects, in addition to health
effects, has been increasingly emphasized in the total evaluation of
chemicals.
The original impetus for the Programme came from World Health
Assembly resolutions and the recommendations of the 1972 UN Conference
on the Human Environment. Subsequently the work became an integral
part of the International Programme on Chemical Safety (IPCS), a
cooperative programme of UNEP, ILO and WHO. In this manner, with the
strong support of the new partners, the importance of occupational
health and environmental effects was fully recognized. The EHC
monographs have become widely established, used and recognized
throughout the world.
The recommendations of the 1992 UN Conference on Environment and
Development and the subsequent establishment of the Intergovernmental
Forum on Chemical Safety with the priorities for action in the six
programme areas of Chapter 19, Agenda 21, all lend further weight to
the need for EHC assessments of the risks of chemicals.
Scope
The criteria monographs are intended to provide critical reviews
on the effect on human health and the environment of chemicals and of
combinations of chemicals and physical and biological agents. As
such, they include and review studies that are of direct relevance for
the evaluation. However, they do not describe every study carried
out. Worldwide data are used and are quoted from original studies,
not from abstracts or reviews. Both published and unpublished reports
are considered and it is incumbent on the authors to assess all the
articles cited in the references. Preference is always given to
published data. Unpublished data are only used when relevant
published data are absent or when they are pivotal to the risk
assessment. A detailed policy statement is available that describes
the procedures used for unpublished proprietary data so that this
information can be used in the evaluation without compromising its
confidential nature (WHO (1990) Revised Guidelines for the
Preparation of Environmental Health Criteria Monographs. PCS/90.69,
Geneva, World Health Organization).
In the evaluation of human health risks, sound human data,
whenever available, are preferred to animal data. Animal and
in vitro studies provide support and are used mainly to supply
evidence missing from human studies. It is mandatory that research on
human subjects is conducted in full accord with ethical principles,
including the provisions of the Helsinki Declaration.
The EHC monographs are intended to assist national and
international authorities in making risk assessments and subsequent
risk management decisions. They represent a thorough evaluation of
risks and are not, in any sense, recommendations for regulation or
standard setting. These latter are the exclusive purview of national
and regional governments.
Content
The layout of EHC monographs for chemicals is outlined
below.
* Summary - a review of the salient facts and the risk evaluation
of the chemical
* Identity - physical and chemical properties, analytical methods
* Sources of exposure
* Environmental transport, distribution and transformation
* Environmental levels and human exposure
* Kinetics and metabolism in laboratory animals and humans
* Effects on laboratory mammals and in vitro test systems
* Effects on humans
* Effects on other organisms in the laboratory and field
* Evaluation of human health risks and effects on the environment
* Conclusions and recommendations for protection of human health
and the environment
* Further research
* Previous evaluations by international bodies, e.g., IARC, JECFA,
JMPR
Selection of chemicals
Since the inception of the EHC Programme, the IPCS has organized
meetings of scientists to establish lists of priority chemicals for
subsequent evaluation. Such meetings have been held in: Ispra, Italy,
1980; Oxford, United Kingdom, 1984; Berlin, Germany, 1987; and North
Carolina, USA, 1995. The selection of chemicals has been based on the
following criteria: the existence of scientific evidence that the
substance presents a hazard to human health and/or the environment;
the possible use, persistence, accumulation or degradation of the
substance shows that there may be significant human or environmental
exposure; the size and nature of populations at risk (both human and
other species) and risks for environment; international concern, i.e.
the substance is of major interest to several countries; adequate data
on the hazards are available.
If an EHC monograph is proposed for a chemical not on the
priority list, the IPCS Secretariat consults with the Cooperating
Organizations and all the Participating Institutions before embarking
on the preparation of the monograph.
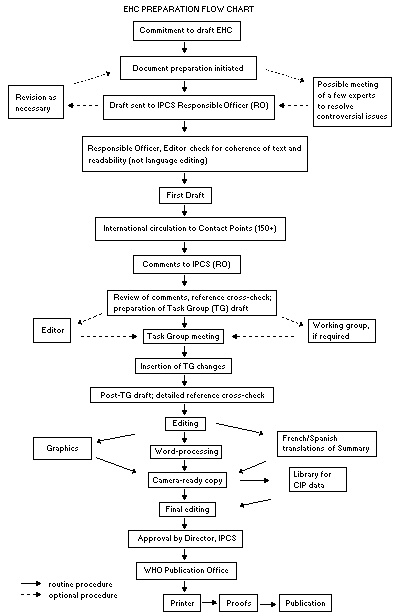
Procedures
The order of procedures that result in the publication of an EHC
monograph is shown in the flow chart. A designated staff member of
IPCS, responsible for the scientific quality of the document, serves
as Responsible Officer (RO). The IPCS Editor is responsible for
layout and language. The first draft, prepared by consultants or,
more usually, staff from an IPCS Participating Institution, is based
initially on data provided from the International Register of
Potentially Toxic Chemicals, and reference data bases such as Medline
and Toxline.
The draft document, when received by the RO, may require an
initial review by a small panel of experts to determine its scientific
quality and objectivity. Once the RO finds the document acceptable as
a first draft, it is distributed, in its unedited form, to well over
150 EHC contact points throughout the world who are asked to comment
on its completeness and accuracy and, where necessary, provide
additional material. The contact points, usually designated by
governments, may be Participating Institutions, IPCS Focal Points, or
individual scientists known for their particular expertise. Generally
some four months are allowed before the comments are considered by the
RO and author(s). A second draft incorporating comments received and
approved by the Director, IPCS, is then distributed to Task Group
members, who carry out the peer review, at least six weeks before
their meeting.
The Task Group members serve as individual scientists, not as
representatives of any organization, government or industry. Their
function is to evaluate the accuracy, significance and relevance of
the information in the document and to assess the health and
environmental risks from exposure to the chemical. A summary and
recommendations for further research and improved safety aspects are
also required. The composition of the Task Group is dictated by the
range of expertise required for the subject of the meeting and by the
need for a balanced geographical distribution.
The three cooperating organizations of the IPCS recognize the
important role played by nongovernmental organizations.
Representatives from relevant national and international associations
may be invited to join the Task Group as observers. While observers
may provide a valuable contribution to the process, they can only
speak at the invitation of the Chairperson. Observers do not
participate in the final evaluation of the chemical; this is the sole
responsibility of the Task Group members. When the Task Group
considers it to be appropriate, it may meet in camera.
All individuals who as authors, consultants or advisers
participate in the preparation of the EHC monograph must, in addition
to serving in their personal capacity as scientists, inform the RO if
at any time a conflict of interest, whether actual or potential, could
be perceived in their work. They are required to sign a conflict of
interest statement. Such a procedure ensures the transparency and
probity of the process.
When the Task Group has completed its review and the RO is
satisfied as to the scientific correctness and completeness of the
document, it then goes for language editing, reference checking, and
preparation of camera-ready copy. After approval by the Director,
IPCS, the monograph is submitted to the WHO Office of Publications for
printing. At this time a copy of the final draft is sent to the
Chairperson and Rapporteur of the Task Group to check for any errors.
 It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
Members
Dr P.J. Abbott, Australia and New Zealand Food Authority
(ANZFA), Canberra, Australia
Dr K. Barab�s, Department of Public Health, Albert Szent-Gyorgyi,
University Medical School, Szeged, Hungary
Dr A.L. Black, Woden, ACT, Australia
Professor J.F. Borzelleca, Pharmacology and Toxicology,
Richmond, Virginia, USA
Dr P.J. Campbell, Pesticides Safety Directorate, Ministry of
Agriculture, Fisheries and Food, Kings Pool, York,
United Kingdom
Professor L.G. Costa, Department of Environmental Health,
University of Washington, Seattle, USA
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood,
Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Dr I. Dewhurst, Mammalian Toxicology Branch, Pesticides Safety
Directorate, Ministry of Agriculture, Fisheries and Food,
Kings Pool, York, United Kingdom
Dr V. Drevenkar, Institute for Medical Research and Occupational
Health, Zagreb, Croatia
Dr W. Erickson, Environmental Fate and Effects Division,
US Environmental Protection Agency, Washington, D.C., USA
Dr A. Finizio, Group of Ecotoxicology, Institute of Agricultural
Entomology, University of Milan, Milan, Italy
Mr K. Garvey, Office of Pesticide Programs (7501C),
US Environmental Protection Agency, Washington, D.C., USA
Dr A.B. Kocialski, Health Effects Division, Office of Pesticide
Programs, US Environmental Protection Agency,
Washington, D.C., USA
Dr A. Moretto, Institute of Occupational Medicine, University of
Padua, Padua, Italy
Professor O. Pelkonen, Department of Pharmacology and
Toxicology, University of Oulu, Oulu, Finland
Dr D. Ray, Medical Research Council Toxicology Unit, University
of Leicester, Leicester, United Kingdom
Dr J.H.M. Temmink, Department of Toxicology, Wageningen
Agricultural University, Wageningen, The Netherlands
Observers
Dr J.W. Adcock, AgrEvo UK Limited, Chesterford Park, Saffron,
Waldon, Essex, United Kingdom
Mr D. Arnold, Environmental Sciences, AgrEvo UK Ltd.,
Chesterford Park, Saffron Waldon, Essex, United Kingdom
Dr E. Bellet, CCII, Overland Park, Kansas, USA
Mr Jan Chart, AMVAC Chemical Corporation, Newport Beach,
California, USA
Dr H. Egli, Novartis Crop Protection AG, Basel, Switzerland
Dr P. Harvey, AgrEvo UK Ltd., Chesterford Park, Saffron Walden,
Essex, United Kingdom
Dr G. Krinke, Novartis Crop Protection AG, Basel, Switzerland
Dr A. McReath, DowElanco Limited, Letcombe Regis, Wantage,
Oxford, United Kingdom
Dr H. Scheffler, Novartis Crop Protection AG, Basel, Switzerland
Dr A.E. Smith, Novartis Crop Protection AG, Basel, Switzerland
Secretariat
Dr L. Harrison, Health and Safety Executive, Bootle, Merseyside,
United Kingdom
Dr J.L. Herrman, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr P.G. Jenkins, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr D. McGregor, Unit of Carcinogen Identification and Evaluation,
International Agency for Research on Cancer, Lyon, France
Dr R. Plestina, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr E. Smith, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
Dr P. Toft, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
IPCS TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
The Core Assessment Group (CAG) of the Joint Meeting on
Pesticides (JMP) met at the Institute for Environment and Health,
Leicester, United Kingdom, from 3 to 8 March 1997. Dr L.L. Smith
welcomed the participants on behalf of the Institute, and
Dr R. Plestina on behalf of the three IPCS cooperating organizations
(UNEP/ILO/WHO). The CAG reviewed and revised the draft monograph and
made an evaluation of the risks for human health and the environment
from exposure to chlordimeform.
The first draft of the monograph was prepared by Dr P. Abbott,
Canberra, Australia. Extensive scientific comments were received
following circulation of the first draft to the IPCS contact points
for Environmental Health Criteria monographs and these comments were
incorporated into the second draft by the Secretariat.
Dr R. Plestina and Dr P.G. Jenkins, both members of the IPCS
Central Unit, were responsible for the overall scientific content and
technical editing, respectively. The efforts of all who helped in the
preparation and finalization of the monograph are gratefully
acknowledged.
ABBREVIATIONS
ACTH adrenocorticotropic hormone
ADI acceptable daily intake
a.i. active ingredient
BSP bromosulfophthalein
CIMS chemical ionization mass spectrometry
CNS central nervous system
CORT corticosteroid
DNA deoxyribonucleic acid
EC emulsifiable concentrate
ECG electrocardiography
GC gas chromatography
HPLC high performance liquid chromatography
IgM immunoglobulin M
JMPR Joint FAO/WHO Meeting on Pesticide Residues
MRL maximum residue limit
Mu Chinese measure of an area equivalent to 1/15 acre
or 1/60 ha or 166 m2
MS mass spectroscopy
NADPH reduced nicotinamide adenine dinucleotide
NC cell activity natural cytotoxic cell activity
NK cell activity natural killer cell activity
NOEL no-observable-effect level
PL prolactin
SAP serum alkaline phosphatase
SGOT serum glutamate-oxalate transaminase
SGPT serum glutamate-pyruvate transaminase
SIR standard incidence rate
SMR standardized mortality ratio
SPF specific pathogen free
TLC thin layer chromatography
TLm median tolerance limit
UV ultraviolet
1. SUMMARY
1.1 Identity, physical and chemical properties, and analytical
methods
Chlordimeform is a base of medium strength and forms stable salts
with strong acids. Both chlordimeform and its hydrochloride salt in
the pure state are colourless crystalline solids. Chlordimeform base
has a melting point of 32�C, while the hydrochloride salt has a
melting point of 225-227�C. Chlordimeform base is sparingly soluble in
water (250 mg/litre) and readily soluble in organic solvents, whereas
the hydrochloride salt is readily soluble in water but less soluble in
organic solvents. Chlordimeform base has a vapour pressure at 20�C of
48 mPa and a log Kow of 2.89. A wide range of analytical methods are
available for detection and quantification of chlordimeform in plants,
soil, water and urine.
1.2 Sources of human and environmental exposure
Chlordimeform does not occur naturally. It is manufactured
commercially by condensation of the Vilsmeier reagent (obtained by
reaction of dimethylformamide with POCl3, SOCl2 or COCl2) either
with 4-chloro- o-toluidine or with o-toluidine and subsequent
chlorination of the resulting intermediate . It has been used as a
broad spectrum acaricide and is active mainly against motile forms of
mites and ticks and against eggs and early instars of some
Lepidoptera insects. It is active in the vapour phase as well as by
contact. In the early period of its use, it was used on a wide variety
of crops such as pome fruits, stone fruits, cole crops, vegetables,
grapes, hops, citrus fruits, apples, pears, cherries and strawberries.
It was also used in cattle dips for the control of cattle ticks. In
the latter years, its use was generally restricted to cotton, although
in some countries, there was continued use on rice. Its registration
was voluntarily withdrawn in 1988/1989 in most countries. In China,
production stopped in 1992 and sales ceased in 1993.
1.3 Environmental transport, distribution and transformation
Chlordimeform has a moderate vapour pressure but its evaporation
from plant surfaces is less than would be expected. The hydrolytic
stability of chlordimeform is strongly pH-dependent; it is stable in
acid conditions but rapidly hydrolysed in alkaline conditions.
Chlordimeform has the potential to adsorb to dissolved organic matter.
In soils, chlordimeform is primarily dissipated by microbial
action with some contribution by chemical hydrolysis. There is little
evidence of leaching despite its water solubility, which may be
due to its adsorption to clay minerals, soil organic matter and
biodegradation. The principal metabolites are N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine.
There is a low but measurable uptake of chlordimeform into plants
from soil, sufficient to affect plant-feeding pests. When applied to
the leaves, chlordimeform has only limited capacity to penetrate the
cuticular layers. Chlordimeform is degraded rapidly in plants. The
principal metabolites are demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine, though not all plants
studied produced the 4-chloro- o-toluidine.
In soils, chlordimeform and its metabolites are dissipated
according to first-order kinetics with a half-life of 20-40 days.
Bioaccumulation studies have demonstrated low uptake of
chlordimeform by aquatic organisms and rapid depuration on transfer to
clean water.
1.4 Environmental levels and human exposure
Levels have not been measured in air and water. Following
applications to paddy fields residues of up to 2900 �g/kg in the top
5 cm of soil and 150 �g/kg in the next 5 cm have been found.
Maximum residue levels were set for a wide range of raw produce
and, in some cases, the residues carried over into processed food. The
Codex maximum residue limits for chlordimeform have been withdrawn.
Occupational exposure to chlordimeform has taken place during
manufacture, formulation and application. In recent years, total
urinary levels of chlordimeform and its metabolites have been used as
a monitor for exposure, and the urine level correlates well with the
degree of skin contamination. Where agricultural workers in the cotton
industry have undergone extensively surveillance for urinary excretion
of chlordimeform, the highest exposure levels were in loaders, washers
and mechanics, with lower levels in flagmen and pilots.
1.5 Kinetics and metabolism in laboratory animals and humans
Chlordimeform is readily absorbed from the gastrointestinal tract
and through the skin of mammals. Rapid excretion follows, with
approximately 80% in the urine and 10-15% in faeces. Low residue
levels are evident in all tissues after approximately 10 days, and
there is no evidence of bioaccumulation. Following dermal
administration in humans, similar rapid excretion through the urine is
observed.
Several oxidized and conjugated metabolites of chlordimeform are
excreted in the urine, demethylchlordimeform, N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine being the major metabolites.
In in vitro studies, the same metabolites are formed,
4-chloro- o-toluidine being the major metabolite.
1.6 Effects on laboratory mammals and in vitro test systems
Chlordimeform has moderate acute toxicity when tested in several
species by oral and dermal routes of administration. The major
metabolites have low oral toxicity when tested in rats. Chlordimeform
causes only slight skin and eye irritation in rabbits. Following
either short- or long-term exposure in both mice and rats with either
chlordimeform or its metabolites, treatment-related changes can be
observed in haematological parameters, and there is some evidence of
hyperplasia of the epithelium of the bile duct and urinary bladder at
the high dose levels. Chlordimeform does not cause an increase in
tumour incidence in rats. In mice, following dietary administration
of either chlordimeform, N-formyl-4-chloro- o-toluidine or
4-chloro- o-toluidine, there is a dose-related increase in
haemorrhagic malignant tumours of vascular origin classified as
malignant haemangioendotheliomas, which cause a dose-related increase
in mortality.
Chlordimeform does not affect reproductive parameters, nor does
it have any teratogenic potential.
Chlordimeform has been tested in a broad range of in vitro and
in vivo genotoxicity assays. No positive responses have been
reported with any of these tests in which unformulated chlordimeform
was tested. In addition, there have been several sporadic and
unconfirmed reports of mutagenic activity induced by N-formyl-
4-chloro- o-toluidine and 4-chloro- o-toluidine. A single report
describes cell transformation induction by both chlordimeform and
4-chloro- o-toluidine. Binding to DNA occurs in the liver of dosed
mice and rats. One major hydrophobic adduct is found at a much higher
level in mice than in rats.
Chlordimeform induces a variety of pharmacological and
biochemical effects in animals, including cardiovascular changes,
hypothermia, hyperexcitability, effects on central visual and auditory
functions, and modulation of biogenic amines and drug-metabolizing
enzymes.
1.7 Effects on humans
Acute poisoning causes fatigue, nausea and loss of appetite, and,
in more severe cases, somnolence, cyanosis, urgency in urination,
cystitis, cardiovascular effects (tachycardia, bradycardia, ECG
changes), coma and shock. Generally, there is complete recovery from
acute intoxication.
Following chronic exposure to chlordimeform, additional symptoms
include abdominal pain, skin itching and rashes (dermal exposure), and
gross and microscopic haematuria. A large number of cases with
clinical symptoms of chronic exposure have been reported in both
chlordimeform-manufacturing plants as well as in agricultural workers.
Following occupational exposure, epidemiological evidence has
provided a strong association between exposure to the metabolite
4-chloro- o-toluidine and the incidence of human urinary bladder
cancer. There is currently only weak evidence for an association
between exposure to chlordimeform and human bladder cancer.
1.8 Effects on other organisms in the laboratory and field
There were no significant effects on populations of soil fungi,
bacteria or actinomycetes following application of chlordimeform to
soil.
There are no laboratory toxicity data on freshwater
invertebrates. Growth of larval oysters was inhibited by chlordimeform
with an EC50 of 5.7 mg/litre. The 96-h LC50 for pink shrimp, the only
crustacean studied, was 7.1 mg/litre and the 96-h LC50 values for
fish ranged from 1 to 54 mg/litre. There are no chronic aquatic
toxicity data available. A mixture of laboratory and field data shows
that chlordimeform is toxic to a wide range of terrestrial non-target
arthropods.
The contact toxicity LD50 for bees has been reported to be
120 �g/g and that for oral toxicity 187 �g/g. There was no mortality
in the field following exposure of species of bees to residues on
alfalfa 3 h after spraying.
The dietary LC50 for various birds species ranged from >1000 to
>5000 mg/kg diet.
1.9 Evaluation of human health risks and effects on the environment
Heavy exposure during manufacture or use, possibly resulting from
inadequate safety precautions, has led to signs of acute poisoning in
workers. Since both production and use are reported to have ceased
worldwide, acute poisoning should no longer occur. The risk associated
with chronic exposure, however, particularly the risk of bladder
cancer, will continue to be of concern for many years. Health
screening of significantly exposed individuals from manufacturing
plants from those rural communities where chlordimeform was
extensively used should be continued.
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
1.10 Conclusions and recommendations
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine and, to a lesser
extent, chlordimeform.
Chlordimeform does not persist in the environment, and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Identity
Common name: Chlordimeform
Chemical structure:
It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
Members
Dr P.J. Abbott, Australia and New Zealand Food Authority
(ANZFA), Canberra, Australia
Dr K. Barab�s, Department of Public Health, Albert Szent-Gyorgyi,
University Medical School, Szeged, Hungary
Dr A.L. Black, Woden, ACT, Australia
Professor J.F. Borzelleca, Pharmacology and Toxicology,
Richmond, Virginia, USA
Dr P.J. Campbell, Pesticides Safety Directorate, Ministry of
Agriculture, Fisheries and Food, Kings Pool, York,
United Kingdom
Professor L.G. Costa, Department of Environmental Health,
University of Washington, Seattle, USA
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood,
Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Dr I. Dewhurst, Mammalian Toxicology Branch, Pesticides Safety
Directorate, Ministry of Agriculture, Fisheries and Food,
Kings Pool, York, United Kingdom
Dr V. Drevenkar, Institute for Medical Research and Occupational
Health, Zagreb, Croatia
Dr W. Erickson, Environmental Fate and Effects Division,
US Environmental Protection Agency, Washington, D.C., USA
Dr A. Finizio, Group of Ecotoxicology, Institute of Agricultural
Entomology, University of Milan, Milan, Italy
Mr K. Garvey, Office of Pesticide Programs (7501C),
US Environmental Protection Agency, Washington, D.C., USA
Dr A.B. Kocialski, Health Effects Division, Office of Pesticide
Programs, US Environmental Protection Agency,
Washington, D.C., USA
Dr A. Moretto, Institute of Occupational Medicine, University of
Padua, Padua, Italy
Professor O. Pelkonen, Department of Pharmacology and
Toxicology, University of Oulu, Oulu, Finland
Dr D. Ray, Medical Research Council Toxicology Unit, University
of Leicester, Leicester, United Kingdom
Dr J.H.M. Temmink, Department of Toxicology, Wageningen
Agricultural University, Wageningen, The Netherlands
Observers
Dr J.W. Adcock, AgrEvo UK Limited, Chesterford Park, Saffron,
Waldon, Essex, United Kingdom
Mr D. Arnold, Environmental Sciences, AgrEvo UK Ltd.,
Chesterford Park, Saffron Waldon, Essex, United Kingdom
Dr E. Bellet, CCII, Overland Park, Kansas, USA
Mr Jan Chart, AMVAC Chemical Corporation, Newport Beach,
California, USA
Dr H. Egli, Novartis Crop Protection AG, Basel, Switzerland
Dr P. Harvey, AgrEvo UK Ltd., Chesterford Park, Saffron Walden,
Essex, United Kingdom
Dr G. Krinke, Novartis Crop Protection AG, Basel, Switzerland
Dr A. McReath, DowElanco Limited, Letcombe Regis, Wantage,
Oxford, United Kingdom
Dr H. Scheffler, Novartis Crop Protection AG, Basel, Switzerland
Dr A.E. Smith, Novartis Crop Protection AG, Basel, Switzerland
Secretariat
Dr L. Harrison, Health and Safety Executive, Bootle, Merseyside,
United Kingdom
Dr J.L. Herrman, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr P.G. Jenkins, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr D. McGregor, Unit of Carcinogen Identification and Evaluation,
International Agency for Research on Cancer, Lyon, France
Dr R. Plestina, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr E. Smith, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
Dr P. Toft, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
IPCS TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
The Core Assessment Group (CAG) of the Joint Meeting on
Pesticides (JMP) met at the Institute for Environment and Health,
Leicester, United Kingdom, from 3 to 8 March 1997. Dr L.L. Smith
welcomed the participants on behalf of the Institute, and
Dr R. Plestina on behalf of the three IPCS cooperating organizations
(UNEP/ILO/WHO). The CAG reviewed and revised the draft monograph and
made an evaluation of the risks for human health and the environment
from exposure to chlordimeform.
The first draft of the monograph was prepared by Dr P. Abbott,
Canberra, Australia. Extensive scientific comments were received
following circulation of the first draft to the IPCS contact points
for Environmental Health Criteria monographs and these comments were
incorporated into the second draft by the Secretariat.
Dr R. Plestina and Dr P.G. Jenkins, both members of the IPCS
Central Unit, were responsible for the overall scientific content and
technical editing, respectively. The efforts of all who helped in the
preparation and finalization of the monograph are gratefully
acknowledged.
ABBREVIATIONS
ACTH adrenocorticotropic hormone
ADI acceptable daily intake
a.i. active ingredient
BSP bromosulfophthalein
CIMS chemical ionization mass spectrometry
CNS central nervous system
CORT corticosteroid
DNA deoxyribonucleic acid
EC emulsifiable concentrate
ECG electrocardiography
GC gas chromatography
HPLC high performance liquid chromatography
IgM immunoglobulin M
JMPR Joint FAO/WHO Meeting on Pesticide Residues
MRL maximum residue limit
Mu Chinese measure of an area equivalent to 1/15 acre
or 1/60 ha or 166 m2
MS mass spectroscopy
NADPH reduced nicotinamide adenine dinucleotide
NC cell activity natural cytotoxic cell activity
NK cell activity natural killer cell activity
NOEL no-observable-effect level
PL prolactin
SAP serum alkaline phosphatase
SGOT serum glutamate-oxalate transaminase
SGPT serum glutamate-pyruvate transaminase
SIR standard incidence rate
SMR standardized mortality ratio
SPF specific pathogen free
TLC thin layer chromatography
TLm median tolerance limit
UV ultraviolet
1. SUMMARY
1.1 Identity, physical and chemical properties, and analytical
methods
Chlordimeform is a base of medium strength and forms stable salts
with strong acids. Both chlordimeform and its hydrochloride salt in
the pure state are colourless crystalline solids. Chlordimeform base
has a melting point of 32�C, while the hydrochloride salt has a
melting point of 225-227�C. Chlordimeform base is sparingly soluble in
water (250 mg/litre) and readily soluble in organic solvents, whereas
the hydrochloride salt is readily soluble in water but less soluble in
organic solvents. Chlordimeform base has a vapour pressure at 20�C of
48 mPa and a log Kow of 2.89. A wide range of analytical methods are
available for detection and quantification of chlordimeform in plants,
soil, water and urine.
1.2 Sources of human and environmental exposure
Chlordimeform does not occur naturally. It is manufactured
commercially by condensation of the Vilsmeier reagent (obtained by
reaction of dimethylformamide with POCl3, SOCl2 or COCl2) either
with 4-chloro- o-toluidine or with o-toluidine and subsequent
chlorination of the resulting intermediate . It has been used as a
broad spectrum acaricide and is active mainly against motile forms of
mites and ticks and against eggs and early instars of some
Lepidoptera insects. It is active in the vapour phase as well as by
contact. In the early period of its use, it was used on a wide variety
of crops such as pome fruits, stone fruits, cole crops, vegetables,
grapes, hops, citrus fruits, apples, pears, cherries and strawberries.
It was also used in cattle dips for the control of cattle ticks. In
the latter years, its use was generally restricted to cotton, although
in some countries, there was continued use on rice. Its registration
was voluntarily withdrawn in 1988/1989 in most countries. In China,
production stopped in 1992 and sales ceased in 1993.
1.3 Environmental transport, distribution and transformation
Chlordimeform has a moderate vapour pressure but its evaporation
from plant surfaces is less than would be expected. The hydrolytic
stability of chlordimeform is strongly pH-dependent; it is stable in
acid conditions but rapidly hydrolysed in alkaline conditions.
Chlordimeform has the potential to adsorb to dissolved organic matter.
In soils, chlordimeform is primarily dissipated by microbial
action with some contribution by chemical hydrolysis. There is little
evidence of leaching despite its water solubility, which may be
due to its adsorption to clay minerals, soil organic matter and
biodegradation. The principal metabolites are N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine.
There is a low but measurable uptake of chlordimeform into plants
from soil, sufficient to affect plant-feeding pests. When applied to
the leaves, chlordimeform has only limited capacity to penetrate the
cuticular layers. Chlordimeform is degraded rapidly in plants. The
principal metabolites are demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine, though not all plants
studied produced the 4-chloro- o-toluidine.
In soils, chlordimeform and its metabolites are dissipated
according to first-order kinetics with a half-life of 20-40 days.
Bioaccumulation studies have demonstrated low uptake of
chlordimeform by aquatic organisms and rapid depuration on transfer to
clean water.
1.4 Environmental levels and human exposure
Levels have not been measured in air and water. Following
applications to paddy fields residues of up to 2900 �g/kg in the top
5 cm of soil and 150 �g/kg in the next 5 cm have been found.
Maximum residue levels were set for a wide range of raw produce
and, in some cases, the residues carried over into processed food. The
Codex maximum residue limits for chlordimeform have been withdrawn.
Occupational exposure to chlordimeform has taken place during
manufacture, formulation and application. In recent years, total
urinary levels of chlordimeform and its metabolites have been used as
a monitor for exposure, and the urine level correlates well with the
degree of skin contamination. Where agricultural workers in the cotton
industry have undergone extensively surveillance for urinary excretion
of chlordimeform, the highest exposure levels were in loaders, washers
and mechanics, with lower levels in flagmen and pilots.
1.5 Kinetics and metabolism in laboratory animals and humans
Chlordimeform is readily absorbed from the gastrointestinal tract
and through the skin of mammals. Rapid excretion follows, with
approximately 80% in the urine and 10-15% in faeces. Low residue
levels are evident in all tissues after approximately 10 days, and
there is no evidence of bioaccumulation. Following dermal
administration in humans, similar rapid excretion through the urine is
observed.
Several oxidized and conjugated metabolites of chlordimeform are
excreted in the urine, demethylchlordimeform, N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine being the major metabolites.
In in vitro studies, the same metabolites are formed,
4-chloro- o-toluidine being the major metabolite.
1.6 Effects on laboratory mammals and in vitro test systems
Chlordimeform has moderate acute toxicity when tested in several
species by oral and dermal routes of administration. The major
metabolites have low oral toxicity when tested in rats. Chlordimeform
causes only slight skin and eye irritation in rabbits. Following
either short- or long-term exposure in both mice and rats with either
chlordimeform or its metabolites, treatment-related changes can be
observed in haematological parameters, and there is some evidence of
hyperplasia of the epithelium of the bile duct and urinary bladder at
the high dose levels. Chlordimeform does not cause an increase in
tumour incidence in rats. In mice, following dietary administration
of either chlordimeform, N-formyl-4-chloro- o-toluidine or
4-chloro- o-toluidine, there is a dose-related increase in
haemorrhagic malignant tumours of vascular origin classified as
malignant haemangioendotheliomas, which cause a dose-related increase
in mortality.
Chlordimeform does not affect reproductive parameters, nor does
it have any teratogenic potential.
Chlordimeform has been tested in a broad range of in vitro and
in vivo genotoxicity assays. No positive responses have been
reported with any of these tests in which unformulated chlordimeform
was tested. In addition, there have been several sporadic and
unconfirmed reports of mutagenic activity induced by N-formyl-
4-chloro- o-toluidine and 4-chloro- o-toluidine. A single report
describes cell transformation induction by both chlordimeform and
4-chloro- o-toluidine. Binding to DNA occurs in the liver of dosed
mice and rats. One major hydrophobic adduct is found at a much higher
level in mice than in rats.
Chlordimeform induces a variety of pharmacological and
biochemical effects in animals, including cardiovascular changes,
hypothermia, hyperexcitability, effects on central visual and auditory
functions, and modulation of biogenic amines and drug-metabolizing
enzymes.
1.7 Effects on humans
Acute poisoning causes fatigue, nausea and loss of appetite, and,
in more severe cases, somnolence, cyanosis, urgency in urination,
cystitis, cardiovascular effects (tachycardia, bradycardia, ECG
changes), coma and shock. Generally, there is complete recovery from
acute intoxication.
Following chronic exposure to chlordimeform, additional symptoms
include abdominal pain, skin itching and rashes (dermal exposure), and
gross and microscopic haematuria. A large number of cases with
clinical symptoms of chronic exposure have been reported in both
chlordimeform-manufacturing plants as well as in agricultural workers.
Following occupational exposure, epidemiological evidence has
provided a strong association between exposure to the metabolite
4-chloro- o-toluidine and the incidence of human urinary bladder
cancer. There is currently only weak evidence for an association
between exposure to chlordimeform and human bladder cancer.
1.8 Effects on other organisms in the laboratory and field
There were no significant effects on populations of soil fungi,
bacteria or actinomycetes following application of chlordimeform to
soil.
There are no laboratory toxicity data on freshwater
invertebrates. Growth of larval oysters was inhibited by chlordimeform
with an EC50 of 5.7 mg/litre. The 96-h LC50 for pink shrimp, the only
crustacean studied, was 7.1 mg/litre and the 96-h LC50 values for
fish ranged from 1 to 54 mg/litre. There are no chronic aquatic
toxicity data available. A mixture of laboratory and field data shows
that chlordimeform is toxic to a wide range of terrestrial non-target
arthropods.
The contact toxicity LD50 for bees has been reported to be
120 �g/g and that for oral toxicity 187 �g/g. There was no mortality
in the field following exposure of species of bees to residues on
alfalfa 3 h after spraying.
The dietary LC50 for various birds species ranged from >1000 to
>5000 mg/kg diet.
1.9 Evaluation of human health risks and effects on the environment
Heavy exposure during manufacture or use, possibly resulting from
inadequate safety precautions, has led to signs of acute poisoning in
workers. Since both production and use are reported to have ceased
worldwide, acute poisoning should no longer occur. The risk associated
with chronic exposure, however, particularly the risk of bladder
cancer, will continue to be of concern for many years. Health
screening of significantly exposed individuals from manufacturing
plants from those rural communities where chlordimeform was
extensively used should be continued.
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
1.10 Conclusions and recommendations
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine and, to a lesser
extent, chlordimeform.
Chlordimeform does not persist in the environment, and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Identity
Common name: Chlordimeform
Chemical structure:
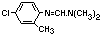 Chemical formula: C10H13ClN2
Relative molecular mass: 196.7
CAS name: N'(4-chloro-2-methylphenyl)-
N, N-dimethyl-methanimidamide
IUPAC name: N2-(4-Chloro- o-tolyl)-
N1, N1-dimethylformamidine
CAS registry number: 6164-98-3 (chlordimeform)
19750-95-9 (chlordimeform hydrochloride)
RTECS number: LQ4375000
Common synonyms: Chlorphenamidine; chlorfenamidine;
chlorophedine; chlorophenamide;
chlorophenamidin; chlorophenamidine;
N'-(4-chloro- o-tolyl)- N,
N-dimethylformamidine;
N, N-dimethyl- N'-(2-methyl-4-
chlorophenyl)-formamidine;
N, N-dimethyl- N'-(2-methyl-4-
chlorophenyl)formadin;
ENT 27335; ENT 27567; EP-333;
N'-(2-methyl-4-chlorophenyl)- N,
N-dimethylformamidine
Trade names: Acaron; Bellotion Especial; Bermat;
Bermatchlorfenamidine; C8514; Carzol;
CDM; CDMS; CGS500; CGS800SP;
Chlorfenamidine; Ciba 8514; Ciba C8514;
COTIP 500EC; Fundal; Fundal 500; Fundex;
Galecron; OMS-1209; Ovatoxion; OVINA;
OVITIX; RS 141; Schering 36268;
Sn 36268; Spanon; Spanone;
SPIKE ULVAIR.
Technical grade chlordimeform is greater than 95% pure and
contains the following impurities: N-formyl-4-chloro-2-toluidine
( N-formyl-4-chloro- o-toluidine), 4-chloro-2-toluidine
(4-chloro- o-toluidine hydrochloride) and sodium chloride.
Chlordimeform free base has been formulated as a 500 g/litre
emulsifiable concentrate. Chlordimeform hydrochloride has been
formulated as a 300 or 800 g/kg water-soluble powder, a 20 g/kg dust
or as 50 g/kg granules.
2.2 Physical and chemical properties
Some of the physical and chemical properties of chlordimeform
base and chlordimeform HCl are shown in Table 1. The molecular
structure of chlordimeform has been investigated by Gifkins & Jacobson
(1980) using single crystal X-ray diffraction.
Table 1. Some physical and chemical properties of chlordimeform
basea
Physical state colourless crystalline solid
Boiling point at 14 mmHg 163 - 165�C
Melting point 32�C
Log Kow 2.89
Vapour pressure at 20�C 48 mPa (3.5 � 10-4 mmHg)
Density (d30) 1.10
Solubility in water at 20�C 250 mg/litre
Solubility in acetone, benzene,
chloroform, ethyl acetate, hexane,
methanol at 20�C >200 g/litre
Half-life at pH 7
(30�C in water, 5% methanol) 42 h
Half-life at pH 9
(30�C in water, 5% methanol) 5 h
Reactivity Forms salt with acids
a From: Worthing (1979); IARC (1978)
Chlordimeform has a solubility in water of 250 mg/litre but is
readily soluble in organic solvents. It forms salts with acids and the
hydrochloride salt is readily soluble in water. When pure,
chlordimeform forms colourless crystals.
Chlordimeform is a base of medium strength with pKa of 6.8 in
50% aqueous methanol (Voss et al., 1973) and forms stable salts with
strong acids.
Chlordimeform is sensitive to light, especially in alkali, and
slowly decomposes in neutral and alkaline aqueous solution. The pH
dependence of photodecomposition of chlordimeform was noted by Su &
Zabik (1972), who observed that an aqueous solution of chlordimeform
hydrochloride (pH 3.1) was unaffected by mercury lamp irradiation for
up to 12 days at 25�C, while a solution of the free base at pH 7-8
decomposed in the same period to a mixture consisting of N-formyl-4-
chloro- o-toluidine and a bis-formamidine. Photo-decomposition of
chlordimeform has also been studied on silica gel chromatographic
plates with irradiation by long- and short-wave ultraviolet light,
fluorescent light and sunlight (under glass) for periods of 10 to 20 h
(Knowles & Sen Gupta, 1969). The major degradation product was again
N-formyl-4-chloro- o-toluidine with either sunlight or UV light.
Fluorescent light caused little decomposition. Sunlight resulted in
12% decomposition in 10 h, while UV resulted in 25% decomposition in
20 h. When 4-chloro- o-toluidine was irradiated with UV light,
numerous decomposition products were found but these were not
characterized further.
Chlordimeform has relatively high volatility and is thus capable
of efficient fumigation action. The hydrochloride salt has negligible
volatility.
2.3 Conversion factors
1 ppm = 8.04 mg/m3 1 mg/m3 = 0.12 ppm
2.4 Analytical methods
2.4.1 Plants
Geissb�hler et al. (1971) described in detail a method for the
determination of total residues of chlordimeform and its metabolites,
which can be used for routine analysis of plant and soil samples. In
this method, chlordimeform and its metabolites are hydrolysed to
4-chloro- o-toluidine by successive treatments with acetic acid and
sodium hydroxide, respectively. The hydrolysis product is then steam
distilled, extracted with isooctane, diazotized and coupled with
N-ethyl-1-naphthylamine yielding a purple dye, which, after column
chromatography on cellulose, is determined by colorimetry. Interfering
azo-dyes from aromatic plants or soil are removed by chromatography on
a cellulose column. This colorimetric method has a limit of detection
of 0.05 mg/kg. If required, the identity of the residues can be
verified by thin-layer chromatography on a cellulose column. This
procedure is sensitive to about 0.1 mg/kg. Alternatively, the
hydrolysis product, 4-chloro- o-toluidine, is diazotized and
iodinated, and the iodinated derivative is measured by electron-
capture gas chromatography. This alternative method has a limit of
detection of 0.05 mg/kg.
Kossmann et al. (1971) refined the method of Geissb�hler et al.
(1971) to permit separate determination of residue quantities of the
parent compound and its potential degradation products in plant
materials. In this procedure, plant material is subject to a two-fold
extraction, the first with methanol/hydrochloric acid and the second
with the lipophilic mixture, methanol/methylene chloride. Separation
of chlordimeform and its degradation products is accomplished by
thin-layer chromatography. The separated eluants are converted to
4-chloro- o-toluidine and analysed as described by Geissb�hler et al.
(1971). The limits of detection for the separated compounds,
chlordimeform, demethylchlordimeform and 4-chloro- o-toluidine are
0.02 to 0.03 mg/kg.
Gr�bner (1977) described a thin-layer chromatographic method for
the determination of chlordimeform residues alone or together with its
metabolite, 4-chloro- o-toluidine, in cucumbers and apples. The
limits of detection for chlordimeform and 4-chloro -o-toluidine were
0.1 and 0.05 mg/kg, respectively. The rates of recovery were 76-85 and
90-105%, respectively.
Fan & Ge (1982) described an alkali flame ionization
gas-chromatographic method for the determination of chlordimeform and
three potential metabolites in cargo rice and husk. Residues of
chlordimeform and its metabolites were extracted with absolute alcohol
or hexane and cleaned up on neutral alumina columns, before being
chromatographed in a column of 1% DEGS coated on 60-80 mesh
405 support (PEG 20M bonded phase). The detection limits for
chlordimeform, 4-chloro- o-toluidine, 2,2'-dimethyl-4,
4'-dichloroazobenzene, and N-formyl-4-chloro- o-toluidine were
0.03, 0.028, 0.11 and 0.43 mg/kg, respectively, for cargo rice and
0.03, 0.028, 0.22 and 0.43 mg/kg, respectively, for husk. Recovery for
chlordimeform was 81-93% for cargo rice and 103-104% for husk.
Recovery for 4-chloro- o-toluidine was 71-73% for both cargo rice
and husk. Recovery for 2,2'-dimethyl-4,4'-dichloroazobenzene was
81.8-112% for cargo rice and 109-118% for husk. Recovery for
N-formyl-4-chloro- o-toluidine was 66% for husk. Mattern et al.
(1991) described a rapid analytical procedure for 17 pesticides,
including chlordimeform, using gas chromatography/chemical ionization
mass spectrometry (GC/CIMS) for detection in various commodities
including peppers, spinach, lettuce and snap beans. Percentage
recoveries for chlordimeform were 87.8% (peppers), 72.6% (spinach),
99.7% (lettuce) and 94.7% (beans). The limits of detection for
chlordimeform were 0.05 mg/kg (beans), 0.05 mg/kg (lettuce),
0.05 mg/kg (peppers) and 0.10 mg/kg (spinach).
2.4.2 Soil
The method of Geissb�hler et al. (1971) described in section
2.4.1 for plants can equally be applied to the determination of total
residues of chlordimeform in soil.
2.4.3 Water
Machin & Dingle (1977) described a UV spectrographic method for
the determination of chlordimeform in cattle dipping baths and
sprays. Preliminary clean-up removes UV-absorbing impurities and
converts chlordimeform to its hydrochloride. Following silica gel
chromatography, the absorbance of the non-eluted material is measured
at 240 nm to determine chlordimeform content. Optimum results are
obtained in the concentration range of 0.02-0.06% (w/v) chlordimeform.
2.4.4 Formulations
Voss et al. (1973) described two methods for the determination of
chlordimeform in formulations. The first relies on acid titration of
the free base with hydrochloric acid. The hydrochloride salt is
converted into the free chlordimeform base, which is extracted into an
organic solvent. After evaporation of the solvent, the active
ingredient is determined potentiometrically. The second method makes
use of gas chromatography, and in this case the chlordimeform
hydrochloride preparations have to be converted into the base form
prior to injection into the gas chromatograph.
Gale & Hofberg (1985) described a gas chromatographic procedure
for the determination of chlordimeform in emulsifiable concentrate
formulations. Chlordimeform was extracted with methylene chloride,
chromatographed on CBWX-20M and detected by flame ionization.
2.4.5 Air
There are no published methods described for the determination of
chlordimeform in air.
2.4.6 Urine
Liu & Mao (1980) described a method for the gas chromatographic
separation of chlordimeform, demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine in urine. Optimum
separation was achieved on a column with 1% polyvinylpyrolidone and 8%
PEG 20M on 80-100 mesh white diatomeous support no. 101 (acid and base
washed). The column was suitable for both qualitative and quantitative
analysis.
A method to analyse urinary residues of workers occupationally
exposed to chlordimeform was developed by Ciba-Geigy in 1980
(Anonymous, 1980a). The method relies on the hydrolysis of
chlordimeform and other residues to 4-chloro- o-toluidine with sodium
hydroxide, followed by extraction with hexane and separation on
reverse-phase liquid chromatography fitted with a UV detector. A
published version of this method was prepared by Geyer & Fattal (1987)
in which the alkaline hydrolysate of urine is extracted with hexane,
the solvent is evaporated, and the hydrolysate is reconstituted with
aqueous acetonitrile. Separation was performed on a reverse-phase Novo
Pak 5 mm C18 column with a UV absorbance detector equipped with a 254
nm filter. A similar method was described by Cheung et al. (1989) for
the analysis of chlordimeform from urine of field workers. Ross &
Leisten (1989) have refined this method with the use of synchronous
spectral data which provides a improved signal-to-noise ratio, which
gives lower minimum detectable levels while still allowing a
well-resolved spectrum. This system may allow detection of levels
equivalent to 1 mg/litre in urine.
2.4.7 Tissues
A gas chromatographic method for the determination of residues of
chlordimeform in animal tissues was first described in the early 1970s
(Anonymous, 1971a). The method involves hydrolysis of chlordimeform to
4-chloro- o-toluidine by successive treatments with acetic acid and
sodium hydroxide. The hydrolysis product is steam distilled and
extracted into isooctane. Following diazotization of the 4-chloro-
o-toluidine, the diazo-moiety is exchanged for iodine by potassium
iodide treatment. The iodinated derivative is gas chromatographed
using electron-capture detection. The limit of detection using this
method is 0.02 mg/kg.
Rieger et al. (1985) have described a gas chromatography/flame
ionization detection method for the determination of chlordimeform and
its major metabolite, demethylchlordimeform, from human tissue
samples, namely, human whole blood and human liver (1:1 aqueous
homogenate). Tissues were first extracted with an organic solvent,
transferred to an acid aqueous medium (0.1M hydrochloric acid),
re-extracted into a small volume of organic solvent and separated on
GC or GC/MS. Using extraction with either chloroform or n-butanol,
recoveries of 81 and 75%, respectively, were obtained.
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Chlordimeform does not occur naturally.
3.2 Anthropogenic sources
3.2.1 Production levels and processes
Chlordimeform was first commercialized in 1966. It can be
manufactured commercially by two methods (Voss et al., 1973), both
starting with the conversion of dimethylformamide to the Vilsmeier
reagent by reaction with POCl3, SOCl2 or COCl2.
By the first method, condensation of the Vilsmeier reagent with
4-chloro-amino-toluene (or 5-chloro-2-aminotoluene, 5-CAT) leads
directly to chlordimeform hydrochloride. Treatment with a strong base
gives the free chlordimeform base.
By the second method, the Vilsmeier reagent is reacted with
o-toluidine to give phenamidine, which is chlorinated in a second
step. The chlorination gives rise to a certain amount of isomers as
unwanted side-products. The crude chlordimeform so obtained has to be
purified either by recrystallization of its chlorohydrate or by
rectification of the free base.
Chlordimeform has been produced at various times in Switzerland,
Germany, United Kingdom, USA, Italy, Argentina and China.
Little information is available on the production levels of
chlordimeform. Information from the US International Trade Commission
(IARC, 1983) indicated that imports of chlordimeform to the USA
through the principal US customs districts amounted to 745 tonnes in
1979 and 198 tonnes in 1980.
In 1974, total usage of chlordimeform in the USA is estimated to
have been 590 tonnes, 77% of which was used on cotton, 15% on
deciduous fruits and nuts, and 8% on vegetables. In 1976, the US
Department of Agriculture reported that 2000 tonnes of chlordimeform
was used in the USA on major crops (IARC, 1983). In 1980, total usage
in the USA was 227 tonnes, all of which was used on cotton to control
budworm/bollworm.
Chlordimeform has been used in China throughout the 1970s and the
1980s at the rate of approximately 10 000 to 15 000 tonnes per year
(Xue, personal communication). In the Chinese province of Hu-bei, the
average annual usage during the period 1984-1988 was 3276 tonnes
(Huang et al., 1989).
3.2.2 Uses
Chlordimeform is a broad spectrum acaricide and is active mainly
against eggs and motile forms of mites and ticks and against eggs and
early instars of some Lepidoptera insects. It kills eggs, larvae and
adults not only by contact but also in the vapour phase. The major use
initially was in the control of mites on deciduous fruit.
In 1971, chlordimeform products were registered in many countries
for use on a wide variety of crops such as pome fruits, stone fruits,
cole crops, vegetables, grapes, hops, citrus, apples, pears, cherries
and strawberries. Chlordimeform also had important veterinary uses as
an acaricide. In Australia, chlordimeform was registered for use in
cattle dips for the control of cattle ticks (Boophilis mictopus), in
combination with organophosphorus acaricides (FAO/WHO, 1972).
In 1975, it was reported that the use pattern of chlordimeform
had been extended to include control of stemborers in irrigated rice,
control of Lepidoptera larvae on cotton, and control of a wide range
of Lepidoptera larvae on cabbage and tomatoes (FAO/WHO, 1976). At
this time, the control of stemborers in irrigated rice proved to be
one of the most important uses of chlordimeform. In the case of
cotton, chlordimeform became one of the most important substitutes for
DDT and other organochlorine pesticides.
Chlordimeform has had no significant usage in non-crop situations
other than on ornamentals.
In 1976, the manufacturers temporarily suspended the sale of
chlordimeform from all markets worldwide, on the basis of adverse
carcinogenicity findings in chronic mouse studies.
In 1978, having completed a number of toxicology, metabolism and
residue studies, the manufacturers re-applied in a number of countries
for registration to allow limited commercial use in cotton crops only.
The proposal was to use chlordimeform by aerial application under
supervised conditions that limited the uptake by operators and
by-standers. Chlordimeform was re-introduced for insect control in
cotton in USA, Central America, Columbia, Israel, Australia and China.
Guidelines for the handling and use of chlordimeform were set in
Australia, Columbia, Israel and USA (California). Application rates
were set to minimize the occurrence of residues in cotton fibres and
cotton seed oil. In China, extensive use of chlordimeform continued
through the 1980s on rice and cotton.
Use of chlordimeform ceased in most countries in the mid to late
1980. The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) withdrew
its temporary Acceptable Daily Intake (ADI) in 1987 and recommended
that chlordimeform should not be used where its residues, or those of
its metabolite, 4-chloro- o-toluidine, could arise in food. (FAO/WHO,
1988).
In 1988-1989, Ciba-Geigy and Schering voluntarily and finally
halted marketing of chlordimeform and decided to withdraw registration
worldwide. In China, production stopped at the end of 1992, and sales
ceased in June 1993.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1 Transport and distribution between media
4.1.1 Air
Chlordimeform has relatively high volatility, and thus when
sprayed on crops considerable evaporation would be expected from plant
surfaces as well as from the soil. Studies in plants, however,
indicate a lower rate of evaporation than expected. In bean plants,
disappearance from the surface in the first few hours was found to be
of the order of only 30-40% of the original dose applied (FAO/WHO,
1972). This result was obtained when either chlordimeform or its
hydrochloride salt was used and is considered to be due to the
buffering capacity of plant exudates with a resulting equilibrium
between the free base and salts. The low volatility from plant
surfaces was confirmed by Sen Gupta & Knowles (1969) on apple
seedlings and by Ehrhardt & Knowles (1970) on grapefruit seedlings. In
cotton plants, approximately 55% of the dose applied to leaves was
volatilized from the surface of the leaves within 2 h (Bull, 1973).
No studies are available on the volatilization of chlordimeform
from soil surfaces, but it is likely to be at least as high as from
leaf surfaces.
4.1.2 Water
While chlordimeform base has only low solubility in water, the
solubility of the hydrochloride salt is relatively high. Its stability
in water, however, is highly pH-dependent, and in the normally neutral
to slightly alkaline conditions of rivers and lakes its half-life
would be relatively short.
It also has the potential to adsorb readily to dissolved organic
matter resulting in precipitation (Maqueda et al., 1989).
The hydrolytic stability of chlordimeform is highly pH-dependent.
It slowly hydrolyses in neutral pH and is stable in strongly acid
conditions. The half-life at 10�C is about 38 days at pH 7, compared
to 8 days at pH 8. At 30�C, these values are reduced to about 3 and
0.5 days, respectively. A solution of the hydrochloride salt (pH 3-4)
showed no appreciable hydrolysis over several days (Su & Zabik, 1972).
The principal product of hydrolysis is N-formyl-4-chloro- o-
toluidine, which at room temperature is very slowly converted to
4-chloro- o-toluidine by further hydrolysis. The second step may be
accelerated by heating with strong acid or alkali.
4.1.3 Soil
Hydrolysis of chlordimeform to N-formyl-4-chloro- o-toluidine
would be expected to be significant under the slightly acid or
slightly alkaline conditions that normally prevail in soils.
Despite the reasonably high solubility of the hydrochloride salt
of chlordimeform, there appears to be little leaching from the site of
application in the soil (FAO/WHO, 1972).
In the studies by Fischer & Cassidy on the uptake of
chlordimeform from soil into cotton plants, the levels of
chlordimeform in the soil were also analysed (FAO/WHO, 1979). Soil was
treated when the cotton reached 10 weeks of maturity. Radioactivity in
the top 75-mm layer of silt loam soil accounted for 1.23 mg/kg
chlordimeform equivalents after treatment. At 7 weeks, this level had
decreased to 0.33 mg/kg and at 13 weeks to 0.20 mg/kg. Extraction of
this layer revealed partition of 32% into the organic layer and 20%
into the polar fraction, and 44% was non-extractable, indicating rapid
degradation. For all but one sample, the level of radioactivity as
chlordimeform equivalents in the lower soil levels, 75-150 mm and
150-200 mm, was less than 0.01 mg/kg, indicating that leaching did not
occur in silt loam. In later experiments with regular over-the-top
spray treatment throughout the maturation of the cotton plants, the
same rapid decrease in radioactivity (as chlordimeform equivalents)
was seen in the top 75 mm of soil. Radioactivity in deeper layers was
again equivalent to less than 0.01 mg/kg. At harvest of the cotton
plants, up to 91% of the radioactivity in the soil could be converted
to 4-chloro- o-toluidine.
The nature of the non-extractable portion of chlordimeform in
soil was investigated by Perez-Rodriguez & Hermosin (1979) and by
Hermosin & Perez-Rodriguez (1981) in experiments examining the
interaction of chlordimeform with clay minerals, montmorillonite,
kaolinite, illite and vermiculite. The earlier work indicated that the
adsorption of chlordimeform on clay is essentially a cation-exchange
reaction and that chlordimeform ions lie between the silicate layers,
thus being difficult to disperse with water or aqueous solutions of
inorganic cations. In the later study, chlordimeform adsorption to the
clay minerals montmorillonite, illite and vermiculite was found to be
an irreversible process, whereas chlordimeform adsorbed on kaolinite
is only weakly bonded and easily removed by washing with water.
The role of soil organic matter in the adsorption and degradation
of chlordimeform in soil was examined in experiments by Maqueda et al.
(1983, 1989). In the first study, the interaction of chlordimeform
with humic acid extracted from the top 20 cm of a clay soil classified
as Typic Chromozerert soil was examined. Adsorption is essentially a
cation-exchange process, although other mechanisms, such as charge
transfer, H-bonding, and van der Waals forces may contribute to the
high adsorption capacity. The variety of mechanism may make it
difficult to ascertain the long-term fate in the environment. In the
second study, the interaction of chlordimeform and other pesticides
with fulvic acids extracted from a spodosol soil was examined. Fulvic
acids are the fraction of humic substances that dissolves in both acid
and alkaline media, and thus are readily found solubilized in lakes
and rivers. The adsorption of chlordimeform was again shown to be a
cation-exchange process, together with H-bonding and charge transfer
mechanisms. Precipitation occurred upon interaction of chlordimeform
with fulvic acids. The amount of precipitate increased in a
dose-related manner up to levels of 100 mmol chlordimeform/litre.
4.1.4 Vegetation and wildlife
Benezet & Knowles (1981) examined the degradation of
chlordimeform by two algal types, Chlorella, the green alga,
and Oscillatoria, a cyanobacterium. In the presence of either
Chlorella or Oscillatoria, chlordimeform was hydrolysed to
N-formyl-4-chloro- o-toluidine, probably by a largely non-enzymatic
reaction. Further reaction formed 4-chloro- o-toluidine and some
CO2. Oxidative N-demethylation was not a major path for chlordimeform
degradation by algae.
The solubility of chlordimeform was sufficient to allow uptake
by the roots of bean and rice plants and to be transported to
plant-feeding pests, as demonstrated by the efficacy experiments of
Dittrich (1967) and Dittrich & Loncarevic (1971).
The ability of plants to take up chlordimeform from soil was
further demonstrated by the experiments of Fischer & Cassidy
(FAO/WHO, 1979), where the soil of a cotton field was treated with
[14C]-chlordimeform when the cotton was 10 weeks old. Uptake of the
radioactivity by the cotton plant was noted to occur in small
quantities, and the highest levels were found in the seeds and fibres.
Biphasic extraction showed 42% in the organic fraction and 34% in the
polar fraction, and 24% was not extractable. Thirteen weeks after
treatment, the mature cotton contained 0.09 mg/kg in the leaves.
The low level of translocation of chlordimeform in plants was
demonstrated by Sen Gupta & Knowles (1969) in experiments where
[14C]-chlordimeform was injected into the stem of apple seedlings
followed by analysis of stem and leaf radioactivity for a period of 20
days. For the first 4 days after injection, 95% of the radioactivity
was localized in the stems, predominantly as the parent compound.
After 20 days, 71.6% of the radioactivity still remained in the stem,
with 25.4% in the leaves, and only 17.9% remained as the parent
compound. The major portion of the radioactivity in the stems after 20
days was unextractable with chloroform and acetone.
In the experiments of Ehrhardt & Knowles (1970) with grapefruit
seedlings, there was no detectable movement of radioactivity into
adjacent stems and leaves 8 days after application of [14C]-
chlordimeform to two upper leaves or two lower leaves. Considerable
movement into stems and leaves was noted when [14C]-chlordimeform was
injected into the main stem, and also to the periphery of grapefruit
leaves when it is applied centrally. Thus, movement of chlordimeform
occurred mainly in the direction of the xylematic transpiration
stream.
Application of chlordimeform directly to the leaves of apple
seedlings (Sen Gupta & Knowles, 1969) or the leaves of grapefruit
seedlings (Ehrhardt & Knowles, 1970) demonstrated the limited capacity
of chlordimeform to penetrate the cuticular layers. Ercegovich et al.
(1972) reported that chlordimeform appeared to adhere to the outer
surface of fruit and did not appear to translocate readily to the
fleshy parts. The chief factors which seem to account for the decrease
of chlordimeform residues in fruit appear to be volatilization,
weathering and growth dilution.
Similarly, the application of [14C]-chlordimeform to cotton
leaves resulted in little movement of radioactivity (and none of
chlordimeform itself) into the untreated plant parts. The small amount
of translocated radioactivity consisted exclusively of polar, mainly
non-extractable substances (Gross, 1977).
In a field experiment, Fischer & Cassidy treated a cotton field
plot over-the-top with [14C]-chlordimeform at a rate of 1 kg/ha when
plants were 12-14 weeks old (FAO/WHO, 1979). Radioactivity in the
cotton plants immediately after treatment was the equivalent of
2.44 mg/kg chlordimeform. At harvest, the radioactivity calculated as
[14C]-chlordimeform was 12.91 mg/kg in the leaves, 0.99 mg/kg in
the stalks, 0.03 mg/kg in the fibre, and 0.26 mg/kg in the seed, with
0.07 mg/kg in the oil and 0.19 mg/kg in the meal. Parent chlordimeform
accounted for 31% and 45.2% in the leaves and stalks, respectively.
The data indicated that although leaf radioactivity is high, there is
still little translocation of [14C]-chlordimeform metabolites to the
seed or fibre.
Supervised residue trials to determine the residue levels in
cottonseed and cottonseed products have been conducted (FAO/WHO,
1979). In general, there is a correlation between the application rate
and the residue level but the interval between the last application
and the harvest also has a strong influence on the residue level. The
decrease of residues with time was most pronounced during the first 10
days after treatment of the cotton plants. At the maximum application
rate of 1 kg/ha, the residue level rarely exceeded 2 mg/kg in
cottonseed, seed meal or crude oil.
When used for the control of rice stem borer in Japan,
chlordimeform resulted in low levels of residues in rice grains and
straws. In rice grain after three treatments, the residue levels of
chlordimeform, demethylchlordimeform, N-formyl-4-chloro- o-
toluidine and 4-chloro- o-toluidine were 48, 0.4, 15 and 53 �g/kg,
respectively. The results indicate a low level of penetration of
chlordimeform into rice plants. The chlordimeform that entered the
plant was gradually degraded to 4-chloro- o-toluidine (Iizuka &
Masuda, 1979).
There have been no studies conducted on the uptake of
chlordimeform by wildlife. Studies with experimental animals suggest
rapid metabolism and excretion, with negligible retention.
4.1.5 Entry into food chain
Potential routes of entry of chlordimeform into the human diet
include the direct consumption of crops containing chlordimeform
residues, the consumption of processed food prepared from treated
crops, or the consumption of animal products derived from animals
treated topically with chlordimeform or raised on chlordimeform-
containing feed such as cottonseed.
Since the temporary withdrawal of the use of chlordimeform from
the market in 1976 in most countries and the later restriction to use
on cotton, dietary consumption of chlordimeform residues on crops in
these countries has virtually ceased. However, dietary consumption of
chlordimeform residues is likely to have continued at least until the
late 1980s in some areas (see section 5.2.2). The maximum residue
levels (MRLs) which were used for chlordimeform are discussed in
section 5.2.2.
4.2 Biotransformation
4.2.1 Degradation in plants
Data reviewed by JMPR (FAO/WHO, 1972) demonstrated that
chlordimeform was quite rapidly degraded in plants with a high
inherent metabolic activity (e.g., bean plants) but was only slowly
degraded in ripe fruits. Green fruits (e.g., grapes) and stems have
an intermediate rate of degradation of chlordimeform. Tentative
identification of the observed metabolites indicated that in
leaves both N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform) and N-formyl-4-chloro- o-toluidine were
major metabolites. In ripe apple and pear fruit, however, only
N'-formyl-4-chloro- o-toluidine was detected. In all tissues,
4-chloro- o-toluidine was either not detected or present in small
quantities, even when six-fold overdose treatment was used.
In the experiments of Sen Gupta & Knowles (1969), [3H]- or
[14C]-chlordimeform was applied to apple seedlings by either leaf
treatment or stem injection. The half-life of degradation was about
12-16 days, and after 20 days 40% of the radioactivity was still
unchanged chlordimeform. Organosoluble degradation products were
identified as demethylchlordimeform, N-formyl-4-chloro- o-toluidine
and 4-chloro- o-toluidine, with the last two representing less than
1% of the total radioactivity. Non-extractable radioactivity, possibly
chlordimeform degradation products complexed with polymeric cell
constituents, was observed only after stem application.
In the experiments of Ehrhardt & Knowles (1970), both
[14C]-chlordimeform and [14C]-chlordimeform hydrochloride were
applied to the leaf surface of growing grapefruit seedlings. After 20
days, only 10-20% of total radioactivity was recovered, possibly due
to evaporation from leaves, and only 1% of radioactivity was unchanged
chlordimeform. The pattern of metabolites was essentially the same as
in apple seedlings, but the levels were smaller.
Witkonton & Ercegovich (1972) examined the metabolites found in
six different fruits (apples, pears, cherries, plums, strawberries and
peaches) following treatment at varying rates with chlordimeform.
Samples of the fruit were collected at various intervals after the
last application from orchards and plants that had been treated with
aqueous sprays of chlordimeform. Of the three potential degradation
products analysed for, only one, namely, N-formyl-4-chloro- o-
toluidine, was detected, together with the parent compound. The other
potential degradation products, namely, demethylchlordimeform and
4-chloro- o-toluidine, were not detected. There was no correlation
between the amount of chlordimeform and 4-chloro- o-toluidine and the
application rate or the sampling interval. The nature of the fruit and
environmental factors were accredited as the major contributing
factors governing the formation and retention of 4-chloro- o-
toluidine. At harvest, the total residue in all crops was
approximately 1 mg/kg, except in peaches, which had approximately
2 mg/kg of total residue. The chief factors which appeared to account
for the decrease in chlordimeform residues were weathering and growth
dilution, rather than chemical or enzymatic degradation.
The potential formation of azo-derivatives of chlordimeform or
its metabolite, 4-chloro- o-toluidine, in treated fruit and
vegetables under field conditions was investigated by Geissbuhler et
al. (1971) using a sensitive gas-chromatographic residue method that
allowed the detection of 0.01 mg/kg of 2,2'-dimethyl-4,4'-
dichloroazobenzene. At 20, 30 or 40 days after a 4-fold overdose
treatment by chlordimeform to apple fruits and leaves, residues of the
azobenzene compound were either not detectable or detected at very low
levels (0.04 mg/kg) in leaves. At normal levels of treatment, residues
of azobenzene compounds would be unlikely to be detected. This result
is supported by the experiments of Witkonton (1973), who analysed
the residues on apple surfaces 60 days after treatment with
[14C]-chlordimeform. The results of these experiments do not support
the in vitro studies of Rose (1969a,b), which indicate the potential
formation of azobenzene derivatives in plants by plant peroxidases.
The metabolism of chlordimeform in cotton plants was first
examined by Bull (1973) following treatment of individual leaves with
[14C]-chlordimeform by petiole injection or by foliar application.
About 45% of the applied dose was absorbed by the leaves, and the
balance volatilized from the leaf surface within 2 h. Tentative
identification of metabolites included demethylchlordimeform,
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine. After
1 h, only 2% of the applied dose could be recovered from leaf
surfaces. The unextractable radioactivity was considered to represent
decomposition products bound to insoluble plant material.
Gross (1977) studied the metabolism of [14C]-chlordimeform in
greenhouse-grown cotton plants following treatment of leaves at a rate
equivalent to 0.6 kg a.i./ha. Metabolites were extracted into hexane,
methylene chloride and water-soluble fractions at various times up to
11 weeks after treatment. The radioactivity in the organic fractions
consisted of at least seven substances. Four were characterized by TLC
as chlordimeform, N-demethylchlordimeform, 4-chloro- o-toluidine
and N-formyl-4-chloro- o-toluidine. Fifty-six percent of the dose
was found in the plant after one week, the balance being lost by
volatilization. The main degradation pathway was hydrolysis,
demethylation only being significant at later sampling times. The loss
of chlordimeform from the surface of leaves was confirmed by
Wolfenbarger et al. (1979) who noted that 24 h after cotton leaves
were treated topically with chlordimeform, only 5% of the EC form was
recovered, whereas 25% of the HCl salt was recovered.
Fischer & Cassidy (FAO/WHO, 1979) identified the metabolites in
leaves after [14C]-chlordimeform was sprayed over-the-top on cotton
plants. At mature harvest, the radioactivity in the leaves consisted
of chlordimeform (60.3%), demethylchlordimeform (4.1%), 4-chloro-
o-toluidine ((7.6%) and N-formyl-4-chloro- o-toluidine (7%). The
results indicate that the parent chlordimeform will be the major
chemical residue in the mature cotton foliage.
Honeycutt & Cassidy (1977) investigated the metabolism of
chlordimeform in cottonseed following injection of [14C]-
chlordimeform into the stem of a growing cotton plant. Forty percent
of the radioactivity in the cottonseed was not extractable. Total
hydrolysis of the radioactivity in the cottonseed showed that a total
of 19.8% of the radioactivity could be converted to 4-chloro- o-
toluidine. The data indicated that the metabolism of chlordimeform in
cottonseed is extensive and results in conjugation to natural
products.
4.2.2 Degradation in soils
The potential for microbial degradation of chlordimeform in
the soil was first identified by Johnson & Knowles (1970), who
demonstrated the capability of several bacteria (Aerobacter
aerogenes and Serratia marcesens), actinomycetes (Streptomyces
griseus) and fungi (Fusarium moniliforme and Rhizopus nigricans)
in culture media to degrade chlordimeform extensively. The
principal metabolite of the bacterial and fungal species was
N-formyl-4-chloro- o-toluidine, while for the actinomycete,
Streptomyces griseus, the principal metabolite was 4-chloro- o-
toluidine. 4-Chloro- o-toluidine was also formed by the bacteria and
fungi. None of the microbes formed symmetrical azo-compounds.
The metabolic fate of chlordimeform in sandy loam over a one-year
period was examined by Iwan & Goller (1975). Soil samples containing
2 �Ci of either [14C- ring]- or [14C- tolyl]-chlordimeform were
prepared in an environmental chamber and methanol/benzene extracts
examined at various intervals. Extractability decreased to 50% within
7 days and was less than 2% after 360 days. In sterilized soil
samples, on the other hand, extractability decreased only slowly, and
70% was still extractable after 180 days. This result indicates that
microbial activity plays a major role in soil degradation of
chlordimeform to non-extractable components. Even though bound to
soil, degradation of chlordimeform continued, as shown by the release
of CO2 as a consequence of oxidative attack upon the tolyl group.
Little CO2 was released under anaerobic conditions and no CO2 was
released from sterile samples. The major pathway of metabolism was
through hydrolysis to 4-chloro- o-toluidine but oxidative
N-demethylation was also a significant pathway leading to
4-chloro- o-toluidine. Further hydrolysis steps followed. The azo
compound, 2,2'-dimethyl-4,4'-dichloroazobenzene, was formed in small
amounts only when the initial chlordimeform concentration was
200 mg/kg in the soil samples. Anaerobic conditions produced the same
metabolic products with the exception of oxidative products such as
demethylchlordimeform. The data suggests that even under sterile
conditions, the degradation of chlordimeform is rapid and its
half-life in non-sterile soils should not exceed one month.
In a further study, Iwan et al. (1976) isolated from
chlordimeform-treated soil four coupling products formed by one-
electron oxidation of 4-chloro- o-toluidine by soil microorganisms.
The four products, one of which is 2,2'-dimethyl-4,4'-
dichloroazobenzene, are formed only from high concentrations of
chlordimeform (70-100 mg/kg), which are at least 10 times higher than
the levels occurring after field application.
4.2.3 Bioaccumulation
There is no data to indicate that chlordimeform bioaccumulates in
plant or animal tissues. However, with a low Kow of 2.89, this
indicates a moderate potential to bioaccumulate.
4.3 Ultimate fate following use
Chlordimeform in the air and in water would be expected to
undergo photodecomposition. In water as well as in soil, chemical
hydrolysis occurs together with adsorption to organic and clay
materials. In plants, residues form complexes with polymeric cell
constituents.
Chlordimeform can be hydrolysed readily to 4-chloro- o-toluidine
by heating with alkali. For the disposal of small quantities of unused
pesticide, the following method is recommended: mix with excess lime
(CaO) or sodium hydroxide (NaOH) and sand and bury at least 0.5 m
below the surface in clay soils. Commercial formulations require
0.5-1.0 kg alkali per kg of pesticide. Alkali can be reduced by 50%
for dilute formulations, e.g., 1% solution or dust. For very
concentrated pesticides (> 50% a.i.), double the amount of alkali and
mix the pesticide with soapy water, before reaction with alkali. Test
reaction on small scale to discover whether or not it will be too
vigorous. Larger quantities should be treated in small batches or
burned in a high-temperature incinerator equipped with effluent gas
scrubbing (IRPTC, 1992).
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air and water
There are no specific data available on the monitoring of
chlordimeform levels in air and water. In neutral and alkaline
solutions, relatively rapid degradation would be expected owing to
hydrolytic instability. Under acidic conditions, slower degradation
would be expected (Su & Zabik, 1972). Adsorption to organic matter in
water would also be expected under field conditions. In both media,
there would be degradation due to photodecomposition (Knowles & Sen
Gupta, 1969).
5.1.2 Soil
Chlordimeform deposited inadvertently on soil surfaces after
spray application may be expected to dissipate by the following
processes: volatilization, chemical hydrolysis, photodecomposition and
microbial degradation. Under field conditions, chlordimeform and its
4-chloro- o-toluidine-containing metabolites are dissipated according
to first-order reactions with half-lives ranging from 20 to 40 days
(Guth & Senn, 1969; FAO/WHO, 1972). The conclusion from these
experiments is that accumulation of chlordimeform in the soil would
not be expected.
Following three applications to rice paddy fields for the control
of rice stem borer, residues of chlordimeform, demethylchlordimeform,
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
2900, 9, 190 and 68 �g/kg, respectively, in the top 5 cm of soil, and
were 150, 1, 8 and 20 �g/kg, respectively, in the 5-10 cm level of
soil. These results indicate the presence of chlordimeform and its
degradation products mainly in the upper layer with minimal movement
downward (Iizuka & Masuda, 1979).
5.2 General population exposure
5.2.1 Environmental sources
There are no longer any environmental sources for exposure of the
general population to chlordimeform. While chlordimeform was being
used on cotton, there was potential for general population exposure to
spray drift from aerial application. The persistence of residues of
chlordimeform on the leaves of cotton also raised the possibility of
exposure through contact with the leaves during the growing period or
during harvesting.
5.2.2 Residues in raw produce
Prior to the temporary suspension of its use in 1976,
chlordimeform was used on a wide variety of crops and on livestock.
The temporary maximum residue levels (MRLs) shown in Table 2 were
established at the 1971 meeting of the Joint Meeting on Pesticide
Residues (JMPR) (FAO/WHO, 1972) as a result of numerous residue trials
in various countries. Residue trials indicated that whilst there was a
sharp drop in the residue level between the day of application and the
second or third day post-treatment, thereafter the rate of decline was
remarkably slow with a half-life on apples, grapes, pears and tomatoes
exceeding 21 days.
Table 2. Temporary tolerances for chlordimeform established in 1971
(FAO/WHO, 1972)
Temporary tolerance mg/kg
Pears, peaches, prunes 5
Apples, grapes, plums, strawberries 3
Brassicas, cherries, citrus fruit, cotton seed oil
(crude and refined), cotton seed 2
Beans 0.5
Fat, meat and meat products of cattle 0.5
Milk (whole) 0.05
Butter 0.5
In 1975, the temporary MRL for pears was raised to 10 mg/kg, and
new temporary MRLs were established for tomatoes (1 mg/kg) and hulled
rice (0.1 mg/kg) (FAO/WHO, 1976). In 1978, the JMPR meeting retained
only the MRLs for cottonseed and recommended that for cottonseed oil
(edible), meat of cattle, pigs, poultry and sheep, and milk and milk
products no residues should occur at the current limit of detection
(0.05 mg/kg) (FAO/WHO, 1979).
The proportion of metabolites and parent compound in the residues
remaining on fruits at various times after application have been
determined in numerous trials. In general, the parent compound
represents the major residue (>80%), followed by N-formyl-4-chloro-
o-toluidine, N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform) and 4-chloro- o-toluidine.
In Chinese residue trials, chlordimeform residues on green
cabbage after application by direct spraying of a 800-fold dilution of
25% chlor-dimeform formulation were 20.9 mg/kg after 4 h, 11.5 mg/kg
after 2 days, 4.2 mg/kg after 7 days and 0.02 mg/kg after 14 days
(Anonymous, 1980b).
In a paper by the Chinese Special Task Group on the residues of
chlordimeform (Anonymous, 1981), the residues of chlordimeform in rice
plants during the period 1974-1980 were examined. In the period
1974-1975, after a single application of 25% aqueous chlordimeform
(9-11 litre/ha) the residue levels on rice harvested after 33-40 days
were 0.25-0.28 mg/kg. When applied at half this rate, residue levels
on rice harvested after 20-74 days were 0.17-0.71 mg/kg. In
field studies in Beijing in 1977, with the same single rate of
application, residue levels on rice harvested after 19-42 days were
0.37-0.51 mg/kg. If 2-3 applications were used, the residue levels on
rice harvested after 19-31 days were 1.3-1.8 mg/kg. The authors noted
the difficulty in meeting the requirement for a residue level of
0.1 mg/kg regardless of the pattern of application. In field studies
in Hu-bei Province in 1978 with the same application rate, the residue
levels in rice harvested after 25-42 days were 0.19-1.20 mg/kg. In
field studies in Zhe-jiang Province in 1978, residue levels in rice
when harvested after 30 days were 0.080-0.112 mg/kg, while residues in
rice harvested after 80 days were 0.039-0.100 mg/kg. In field studies
in Guang-dong Province in 1978, residues in rice harvested after 30
days were 0.042-0.149 mg/kg. In other field studies in the Guang-dong
Province in 1980, residue levels on rice harvested after 56 days were
0.185 mg/kg, but when the harvest was performed at 72 days, the
residue level was less than 0.10 mg/kg (Anonymous, 1981).
Huang et al. (1989) reported the residues of chlordimeform on
both rice and cotton plants in the Hu-bei Province of China between
1984 and 1988. With 1-3 applications to rice plants, followed by
harvest after 25-55 days, the chlordimeform residues were generally in
the range of 0.066-0.820 mg/kg for the rice, 7.70-22.30 mg/kg for the
husk, and 16.5-21.2 mg/kg for the stem. The authors noted that the
residue levels seldom met the 1975 JMPR recommended MRL of 0.10 mg/kg
for hulled rice (FAO/WHO, 1976). In further work on rice plants, it
was noted that the residue levels for late rice were generally higher
(approximately 2-fold) in late rice compared to early rice, and that
the residue levels in both the rice and the husk reduced by more than
90% when the time to harvest was increased from 26 to 72 days. With a
72-day harvest, the residue level in the rice was 0.065 mg/kg. The
residue levels in the stem (18-41 mg/kg), on the other hand, remained
relatively unchanged over the 72-day period. With 1-3 applications to
cotton plants, followed by harvest after 40 days, the chlordimeform
residues were 0.053-0.151 mg/kg in the kernel and 0.118 mg/kg in the
bracket.
Chlordimeform residues were also found in 8/15 honey samples
(Huang et al., 1989). The highest residue found was 32.2 mg/kg, and
the majority of the samples contained less than half this level. In
1994 the US FDA collected and analysed samples of honey imported from
the People's Republic of China. Of 60 samples analysed, 39 had
detectable residues, the highest being 0.058 mg/kg (Krick, 1994).
Moore (1971) summarized the results of residue trials on the use
of chlordimeform as an acaricide in cattle dips in Australia. The
residues were examined in cattle muscle, fat and liver as well as in
milk and butter from the first milking. Chlordimeform was used at
concentrations of 0.0125-0.1% in buffered cattle dips. Residues in
muscle, fat and liver did not increase greatly with increasing dose
of chlordimeform, and showed significant reductions between day 1
and day 3 post-treatment. The maximum residue levels found at day
3 post-treatment in muscle, fat and liver were 0.33, 0.51 and
0.69 mg/kg, respectively. At the first milking, the residues levels
showed a closer relationship with the concentration of chlordimeform
in the dip. The residue levels in milk and butter at a concentration
of 0.0125% were 0.01 and 0.30 mg/kg, respectively. The maximum
residue levels in milk and butter, which were found at the highest
concentration used (0.2 %), were 0.31 and 1.6 mg/kg, respectively.
In the study by Burkhard (1971), cows washed with a 0.5%
solution of chlordimeform to the hindquarters (3 treatments at 7-day
intervals), had total residue levels in milk, meat and fat below the
level of detection (0.03 mg/litre), except in milk on the day after
treatment when the levels rose to 1 mg/kg. In a further study by Voss
& Burkhard (1971), when cows were fed a concentrate containing
40-240 mg/kg chlordimeform for periods up to 42 days, the total
residues of chlordimeform and its metabolites in all milk, meat
and fat samples were below the limit of detection (0.03 mg/litre or
mg/kg). In liver and kidney samples, residues rose to a peak between
14 and 21 days (0.58 mg/kg in liver and 0.13 mg/kg in kidney), which
was followed by a slow decline.
In a study by Palmer et al. (1977), residues of chlordimeform
were determined in tissues and milk of cattle after spray application
to control cattle tick. In subcutaneous fat from animals sprayed with
0.45, 0.15 or 0.05% chlordimeform, the residue levels were 2.88, 0.46
and 0.15 mg/kg, respectively. The half-life of disappearance in all
cases was 2.46 days. Lower residue levels were found in six other
tissues, including kidney, muscle and liver. Residue levels in whole
milk of lactating cows at the three treatment levels were 1.42, 0.28
and 0.03 mg/litre, respectively. The half-life of disappearance from
milk was 0.45 days.
White Leghorn hens fed a laying mash containing chlordimeform at
levels of 0.25, 0.75 or 1.0 mg/kg were examined for residues in eggs
and tissues (breast, fat and liver) for periods of up to 28 days. No
residues were detected in breast meat. Residues were detected in fat
(0.22 mg/kg) at the 21 days only. Residues in the liver were highest
between 7 and 14 days (0.20 mg/kg) and reduced rapidly upon withdrawal
from the chlordimeform-containing feed. There were no detectable
residues of chlordimeform in eggs (FAO/WHO, 1972).
Residue trails on cotton were conducted between the years 1969
and 1978 (FAO/WHO, 1979). The application rates ranged from 0.125 to
3.6 kg/ha and resulted in mean residue levels of 0.1 to 13.1 mg/kg in
cottonseed when it was harvested immediately after application. The
final residue level was dependent on a number of factors including
application rate, number of applications, and length of waiting period
before harvest. The application rate had the largest influence.
5.2.3 Residues in processed food
Total residues of chlordimeform and its metabolites do not reduce
substantially during cooking processes, since while the proportion of
parent compound is reduced, there is an increase in the hydrolysis
product, N-formyl-4-chloro- o-toluidine. Residues of chlordimeform
itself in crops decrease through hydrolysis, but volatilization in
steam during cooking is not an important factor. The rate of
hydrolysis of chlordimeform is a function of pH and occurs much more
rapidly in weakly acid or neutral crops such as cauliflower (pH 6) or
green beans (pH 5) than in strongly acid crops such as apples (pH 2.5)
or tomatoes (pH 3). These results have been derived from studies in
different crops such as apples, grapes, tomatoes, cauliflower, beans
and sugar beet. These studies have also shown that residues of
chlordimeform and its metabolites are located in the outer parts of
crops, such as fruit peel. Excessive residues might therefore be
removed by peeling fruit (apples, citrus) or trimming the outer leaves
of leaf crops. In general, washing will remove only a small part of
the total residue (FAO/WHO, 1972).
Chlordimeform residues in whole apples reduced to approximately
40% of this level in pressed apple juice, while the level in the wet
pomace doubled (FAO/WHO, 1972) This is consistent with studies that
have shown that the residue level in the skin and outer layer is
approximately 50-fold higher than that found in the flesh (FAO/WHO,
1972).
Chlordimeform residues in tea leaves were found to be extractable
into tea prepared from these leaves to the extent of approximately 50%
of the total residues (Blass, 1972a).
Chlordimeform residues in grapes reduced to approximately 60% of
this level in grape juice (Blass, 1972b). This is consistent with
studies that have shown that the residue level in the grape skin was
between 60 and 76% of total residues (FAO/WHO, 1972). Fermentation
of the grape juice over a period of 72 days yielded a wine that
contained residue levels similar to those in grape juice (Blass,
1972c), indicating that the fermentation process does not
significantly lower the total chlordimeform residue level.
Chlordimeform residues in green hop cones, when used to prepare
beer, were found to be reduced to levels below the level of detection
(0.03 mg/kg) (Voss, 1971).
Residues associated with the processing of cottonseed have been
reported (FAO/WHO, 1979). Separation of the cottonseed oil leaves the
majority of the residues in the hulk and meal, although a significant
residue still remains in the crude oil. Additional refining processes
including bleaching, hydrogenating and deodorizing reduce the residue
level to below the level of detection. Cottonseed oil for human
consumption is subject to the bleaching and deodorizing processes and
thus residues of chlordimeform will be virtually zero.
5.3 Occupational exposure during manufacture, formulation or use
5.3.1 Exposure during manufacture and formulation
In the cases described by Folland et al. (1978) of
hospitalization of three factory workers in the USA who were exposed
to chlordimeform, the urinary levels of chlordimeform plus 4-chloro-
o-toluidine were 1.29, 6.32 and 4.85 mg/litre, respectively, three
days after exposure. This report is described in more detail in
section 8.2.2.
In a study on workers in the USA engaged in chlordimeform
production and packaging in 1976, urine was monitored in more than
100 workers. In more than 800 individual urine samples, total urinary
levels ranged from 0.05 to 50 mg/litre (personal communication by J.W.
Barnett, Ciba-Geigy Agricultural Division, Greenborough, North
Carolina, USA, to California Department of Food and Agriculture).
In China, there have been several studies in which the level of
exposure of workers to chlordimeform in chemical factories has been
examined together with a medical examination to detect any evidence of
toxicity in these workers. These are described in section 8.2.
In the study by Lu et al. (1981), the air concentrations
in 1974 in a chlordimeform-producing factory were generally below
0.036 mg/m3, with shorter periods at higher levels (0.108-
0.33 mg/m3), during specific tasks. Skin contamination on hands and
forearms was 9.1 mg/h for chemical operators and 964.2 mg/h for
packers. The urinary excretion levels for chlordimeform and
4-chloro- o-toluidine in controls were 0.015 and 0.042 mg/litre,
respectively, in chemical operators were 0.065 and 0.108 mg/litre,
respectively, and in packers were 0.263 and 0.398 mg/litre,
respectively.
In the study by Li et al. (1985b), 24 packers (9 male, 15 female)
in a chlordimeform manufacturing plant in Jiang-su Province of China,
were exposed to chlordimeform air concentrations (9 samples over 3
consecutive days) of 0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin
contamination of the hands and forearms was 110 �g/100 cm2
(S.D. 39 �g/100 cm2). Urinary chlordimeform levels were
0.20 � 0.13 mg/litre, and urinary 4-chloro- o-toluidine levels
were 0.48 � 0.29 mg/litre.
In a further study (Anonymous, 1985a) in a chlordimeform
manufacturing factory in China, packers had the highest urinary
chlordimeform and 4-chloro- o-toluidine levels at 0.39 mg/litre which
significantly correlated with skin contamination but not with air
concentration.
In the study by Tao et al. (1985), 61 employees (25 chemical
operators, 36 packers) of a pesticide factory in China were exposed to
air levels in the range 0.074 to 0.160 mg/m3. Skin contamination of
packers (2.99 mg/day) was higher than for chemical operators
(0.784 mg/day). The urinary excretion rate of chlordimeform and
4-chloro- o-toluidine in packers was also higher (0.513 mg/litre)
than for chemical operators (0.206 mg/litre) or controls
(0.055 mg/litre).
5.3.2 Exposure during use
In a company report by Kossmann (1980), summary data was provided
on the results of occupational exposure surveillance programmes on
agricultural workers associated with chlordimeform in nine countries.
Surveys of aerial pesticide applications to cotton entailed the
monitoring of about 600 airstrips in 1979 in the nine countries. Over
28 000 urine samples were analysed from workers in all phases of the
application situation. The urine was monitored and residue data
expressed as chlordimeform equivalents. In 1% of the assays,
substantial chlordimeform urinary residues indicated a significant
occupational exposure. Over 75% of the samples were at or below the
lowest level of analytical detection. This report states that, in
general, the conditions in two countries, the USA and Australia, were
indicative of favourable working conditions where only about 1% of the
samples contained a residue level indicating a higher-than-desired
level of exposure. In a subsequent report by Kenyon et al. (1993),
however, it is stated that at least 20% of the urine samples in
agricultural workers associated with chlordimeform in New South Wales,
Australia, exceeded the maximum permissible exposure level for
chlordimeform equivalents in urine, which was set at 0.2 mg/litre.
Operators who exceeded this level were required to be withdrawn from
the site until the urinary level fell below 0.1 mg/litre. The mean
sample assays for both ground rig operators and workers involved in
aerial application exceeded the set level in 1984-1985. Furthermore, a
number of workers experienced exposures that exceeded the limit on
multiple occasions. The urine monitoring programme in operation in New
South Wales, Australia, also grossly underestimated the worker
exposure levels since its protocol did not allow urine sample
collection in the first 24 h following potential exposure (Kenyon et
al., 1993). In the report by Kossman (1980), it is stated that working
conditions in some other countries (i.e., Colombia, El Salvador,
Guatemala and Honduras) were less favourable and thus exposure was
higher. However, in some areas where flagmen were unavoidably exposed,
the urinary residue levels were low, indicating that with precautions
exposure can be controlled. In New South Wales and Israel, urine
monitoring for agricultural workers was mandatory, while in the USA,
urine monitoring was conducted on a voluntary basis.
In a report by Henderson (1985), monitoring studies on operator
exposure during the 1984-1985 cotton season in NSW, Australia, were
summarized. Urine samples were examined in operators involved in
application of chlordimeform by both ground-rig (Strong & Bull, 1985a)
and aerial (Strong & Bull, 1985b) methods. Chlordimeform application
by ground-rig to 26 444 hectares involved 48 people. A total of 85
urine samples were examined; in 78.8% of samples the chlordimeform
level was below 0.20 mg/litre, and in 90.5% of samples it was below
0.50 mg/litre. The mean sample assay was 0.21 mg/litre. Chlordimeform
application by aerial spraying to 315 694 hectares involved
222 people. A total of 919 urine samples were examined and in 80.3% of
samples, the chlordimeform level was below 0.20 mg/litre, and in 89.8%
of samples was below 0.50 mg/litre. The mean sample assay was
0.24 mg/litre.
The exposure data for chlordimeform used on cotton in seven
countries (Australia, Columbia, El Salvador, Guatemala, Mexico,
Nicaragua, USA) for the period 1980-1984 has been compiled in a
company report by Limmer (1985). Urine samples indicated that in all
countries, the chlordimeform level was less than 0.3 mg/litre for
between 70 and 92% of the exposed workers, and was >5 mg/litre in
less than 2% of workers. The highest levels were recorded in the
loaders, washers and mechanics, while the lower levels were found in
the pilots and flagmen.
In a study by Jiang et al. (1985), exposure of workers engaged
in spraying chlordimeform with fine mist sprayers in both rice fields
and cotton fields was examined. The air concentration of chlordimeform
surrounding the workers during spraying was 0.80 mg/m3. Skin
contamination from spraying in a rice field was 0.777 mg/100 cm2/h
(16 samples), and from spraying in a cotton field was 0.445 mg/100
cm2/h for one group (40 samples) and 1.216 mg/100cm2/h for a
second group (40 samples). Urinary excretion of chlordimeform and
4-chloro- o-toluidine together was 0.756 mg/litre for rice workers,
and 0.490, 0.465 and 1.125 mg/litre in three separate groups (40 each)
for cotton workers. Good correlation was noted between skin
contamination and urinary excretion. It was noted that contamination
of the lower extremities of the body was significantly different
between workers with protection (0.490 mg/100 cm2 per h) and those
without (1.179 mg/100 cm2 per h).
In a study by Ling et al. (1986) and Zhang et al. (1986a),
excretion of chlordimeform and 4-chloro- o-toluidine was examined as
a measure of occupational exposure. Chlordimeform applicators (7 male,
6 female; 20-41 years) were examined during spraying of cotton for
three consecutive days for 4.7, 3.0 and 4.4 h respectively in July
1985. Protective measures included gauze mask, plastic gloves and
plastic apron, although it was noted that extensive contamination
occurred. Air levels in the breathing zone on each of the three days
were 0.011, 0.014, 0.011 mg/m3, respectively. Skin contamination on
each of the three days was estimated by the method of Zhang et al.
(1986b) to be 10.99, 4.32, and 4.45 mg/day, respectively. Urinary
chlordimeform and 4-chloro- o-toluidine together were measured over
the 3 days of exposure and for 7 days after cessation of exposure.
Urinary levels ranged from a peak of 2.408 mg/litre during exposure to
0.036 mg /litre after 7 days. Excretion of chlordimeform occurred very
rapidly and the highest level was detected in the sample collected at
the end of each shift. There was a close correlation between skin
contamination and urinary excretion. Metabolism occurred very rapidly
since 4-chloro- o-toluidine usually accounted for 70-93 % of the
total amount in the urine. The authors concluded that the level of
urinary chlordimeform plus 4-chloro- o-toluidine is an accurate index
of chlordimeform exposure.
Maddy et al. (1986) reported the results between 1982 and 1985 of
a programme of monitoring the urine of more than 200 workers who had
received training in the use of chlordimeform on cotton in California.
Protective clothing was required for all employees who handled
containers, prepared mixtures, loaded application vehicles, applied
chemical, flagged or did repair work on equipment exposed to
chlordimeform. This included cloth overalls, washable hat, waterproof
boots, waterproof gloves, and a full-face shield. Chlordimeform was
detectable in urine as early as 4 h after dermal exposure, but did not
increase during the work season. The chlordimeform concentrations
averaged about 90 �g/litre, with the highest levels found in
mixer-loaders and somewhat less in equipment washers, and the lowest
levels in pilots and flaggers. Urinary levels in the 8-10 h following
a work shift gave a good indication of exposure for the shift just
completed.
Kurtz et al. (1987) reported the results of a monitoring
programme of agricultural workers exposed to chlordimeform when used
on cotton in Imperial Valley, California, during the 1982 season. More
than 1000 urine samples were taken from 132 workers, including pilots,
mixers/loaders, flaggers and equipment maintenance workers.
Chlordimeform metabolites were detected in all workers at some time
during the study despite the use of protective clothing. The level of
urinary metabolites was positively correlated with the length of
exposure and the nature of job activity as shown in Table 3.
Mixer/loaders and maintenance workers had the highest levels.
Metabolites appeared in urine within 4 h and approximately 75% of
urinary excretion occurred within the first 24 h.
Table 3. Chlordimeform metabolite concentrations in urine (mg/litre)
of agricultural workers during an 11-week application period
(Kurtz et al., 1987)
Work group Immediately post-work Following morning
No. Mean SD No. Mean SD
All groups 535 0.12 0.41 572 0.10 0.23
Pilots 145 0.08 0.10 163 0.08 0.10
Mixers/Loaders 156 0.19a 0.71 162 0.15b 0.36
Flaggers 202 0.07 0.08 213 0.07 0.09
Others 32 0.25 0.45 34 0.21c 0.36
a Significantly greater versus flagger group (P<0.01)
b Significantly greater versus pilots (P<0.01) and flaggers
(P<0.001)
c Significantly greater versus pilots (P<0.001) and flaggers
(P<0.001)
Lemesch et al. (1987) provided the results of monitoring for
chlordimeform exposure in agricultural workers in Israel during
1980-1985. Chlordimeform was used only on cotton by aerial application
and all workers were monitored for urinary chlordimeform and its
metabolites on a weekly basis. The results indicated 86.8% of the
urine samples contained less 0.05 mg/litre, and 1.4% contained more
than 0.30 mg/litre. Overall, the loaders had the highest exposure
followed by the mechanic and then the pilots (see Table 4).
Table 4. Chlordimeform metabolite concentrations in urine (mg/litre)
of agricultural workers in Israel during 1980-1985
according to occupation (Lemesch et al., 1987)
Occupation < 0.05 0.05 - 0.30 > 0.30 Total
No. % No. % No. %
Loaders 666 79.0 157 18.6 20 2.4 843
Mechanics 383 94.8 19 4.7 2 0.5 404
Pilots 287 98.2 5 1.7 - - 292
Total 1336 86.8 181 11.8 22 1.4 1539
Balu (1989) has provided the results of monitoring field worker
exposure to chlordimeform from aerial application on cotton. During
the years 1978-1984, urine samples using a grab sample technique from
approximately 4600 field workers were examined. For mixer/loaders,
between 0.5 and 1.9% had levels >5 mg/litre, and between 2.1 and 18%
had levels of 1.0-5.0 mg/litre. The majority (46-78%) had levels in
the range <0.05-0.10 mg/litre. There was no apparent change in the
proportion of workers in the various exposure levels over the course
of the study. For the pilots, between 0.3 and 0.7% had levels
>5.0 mg/litre, while 63-90% had levels between <0.05 and
0.10 mg/litre.
The clinical signs associated with chlordimeform exposure in
these studies are described in section 8.2.2.
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption, distribution and excretion
6.1.1 Mouse and rat
The earliest investigations on the kinetics and distribution of
chlordimeform were performed in a series of studies on rats (FAO/WHO,
1972). Four male and four female rats were treated orally with 270 �g
[3H-phenyl]-chlordimeform. Over a 24 h period, 52.8% (range
41.8-59.6%) of the radioactivity was eliminated in urine and 2.5%
(range 0.13-5.30%) in faeces, while 19-23% of the dose was excreted
into the bile. Following intravenous injection of 270 �g [3H-phenyl]-
chlordimeform in rat, elimination of radioactivity over 24 h consisted
of 53.7% (range 52.0-55.6%) in urine and 1.42% (range 1.19-1.84%) in
faeces.
Oral dosing of male rats with 270 �g [3H-phenyl]-chlordimeform
resulted in residues in liver (0.78 mg/kg), kidney (0.59 mg/kg)
and lymph nodes (0.35 mg/kg) after 8 h. After 24 h, residues in
gastrointestinal tract (and contents) and liver were 0.95 and
0.35 mg/kg, respectively. All other tissues contained residue levels
of <0.16 mg/kg at 8 h, and <0.27 mg/kg at 24 h (FAO/WHO, 1972).
Oral dosing of male rats with 270 �g [3H-phenyl]-chlordimeform
for seven consecutive days resulted in excretion of 59% of the
administered label in urine and 10% in faeces during the dosing
period. Tissue residues at the termination of dosing were less than
0.03% of the administered dose (FAO/WHO, 1972).
Knowles & Sen Gupta (1970) further studied the toxicokinetics
in rats. A group of two male and two female rats was given
[14C-tolyl]-chlordimeform (3 �Ci) orally (dose unspecified). Over a
72-h period, 88% of the administered radioactivity was eliminated in
the urine, with the highest concentration occurring at 12 h, and 7.5%
was eliminated in the faeces. At sacrifice (72 h), tissue levels based
upon [14C]-label levels were 0.21 mg/kg in liver, 0.15 mg/kg in
muscle, 0.11 mg/kg in fat and less than 0.1 mg/kg in other tissues. As
part of the same study (Knowles & Sen Gupta, 1970), a similar group of
male and female rats received an oral dose of [14C-methyl]-4-chloro-
o-toluidine. Tissue levels based upon [14C]-label levels at 72 h
after dosing were 0.33 mg/kg in fat, 0.26 mg/kg in liver, 0.2 mg/kg in
kidney and oviduct, 0.1 mg/kg in brain, and less than 0.1 mg/kg in
other tissues.
In a more recent study by Watanabe & Matsumura (1987) concerning
the comparative metabolism of chlordimeform and sulfamidine in rats,
it was found that after administration of [14C]-chlordimeform as a
single oral dose (130 mg/kg), radioactivity was eliminated in the
urine (87%) and faeces (8%) within 3 days. Most of the radioactivity
was excreted within 2 days. After 5 daily doses of [14C]-
chlordimeform (26 mg/kg), 78% of the radioactivity was excreted in the
urine and 15% in the faeces. After 10 days, the residue level in all
tissues, except blood and liver, was below 1 mg/kg.
In a study by Ifflaender (1977a), groups of mice (8/sex; strain
Tif:MAG f) and rats (3/sex; strain TIF:RAI f) were administered
[14C- ring]-chlordimeform orally at a dose of 25 mg/kg body weight.
The general excretion pattern was similar for both mice and rats with
more than 70% of the [14C]-label being excreted within 24 h. Of the
excreted dose, 80-95% was excreted through the urine, while 10-15% was
excreted through the faeces. After 144 h, 95-113% of the administered
dose was recovered. Over the period of the experiment (144 h), the
levels of radioactivity in the urine were found to range from 82-97%
of the administered dose. Residues of chlordimeform were found in
liver, kidney and blood, with the highest level found to be 1 mg/kg.
Slightly higher residue levels were found to be present in females
than in males. In a subsequent study by Ifflaender (1977b) to
determine the quantitative differences between mice and rats, animals
were administered 25 mg/kg body weight [14C- ring]-chlordimeform.
Rapid urinary excretion of chlordimeform was again observed in both
mice (85%) and rats (75%) within 24 h.
In a more detailed toxicokinetic study by Kopp et al. (1977),
chlordimeform was administered orally to female mice at two dose
levels (1.2 or 120 mg/kg body weight) using either a single acute or
multiple daily administration for up to 21 days. The results again
indicated rapid excretion of chlordimeform and/or its metabolites
through the urine and did not provide any indication of
bioaccumulation at either dose level. At the high dose level, a
slightly reduced 24-h excretion pattern of the radioactivity was
observed following a single administration. This pattern returned to
normal within two to three doses in the multiple dosing regime. The
percentage of excretion was the same after a period of 21 days,
irrespective of the dose level. The authors concluded that
chlordimeform excretion was largely complete within 24 h of
discontinuation of administration. No accumulation of residues was
evident.
Knowles & Benezet (1977) studied the kinetics of chlordimeform in
mice following intraperitoneal injection of 0.6 �Ci [14C-tolyl]-
chlordimeform. Over the 96-h period, 95.5% of the administered dose
was eliminated, with 42.5% in the urine and 53% in the faeces. In the
first 3 h, 43.7% was eliminated, with 27.3% in urine and 16.4% in
faeces.
In a study in mice by Crowder & Whitson (1980), the excretion and
retention of [14C]-chlordimeform in mice was found not to be affected
by oral co-administration of toxaphene or methyl parathion. Low
residue levels of chlordimeform were evident at 196 h in all tissues
following oral administration.
6.1.2 Other species
In a study by Sen Gupta & Knowles (1970), two female dogs (18 and
20 kg) were given 10 �Ci [14C]-chlordimeform as a single oral dose
(0.3 mg/kg), and one male dog (12 kg), which had undergone cannulation
of the gall-bladder and ligation of the bile duct, was given 20 �Ci
[14C]-chlordimeform orally. Urine was collected (by catheterization)
at 1, 3, 6, 12, 24, 48 and 72 h. Faeces were collected at similar time
intervals. Of the administered [14C] label, 85% was recovered in
urine, 0.6% in faeces, and 5% in the bile within 72 h. In the same
study, two brush goats, a male (36 kg) and a lactating female (39 kg)
were administered 10 �Ci [14C]-chlordimeform orally. The male goat
eliminated 87% of the administered dose through the urine within 48 h,
while the lactating female eliminated only 67% during the same period.
Only about 0.3% of the applied dose was eliminated in the milk within
96 h.
In a review by Knowles (1970), the metabolites found in three
species, namely, rat, dog and goat, were compared. In all three
species, oral treatment with radioactive chlordimeform resulted
primarily in elimination through the urine. Cumulative percentages of
the dose excreted in the urine 24 h after treatment were 85% for rats,
70% and 80% for the two dogs, 65% for a lactating goat, and 80% for a
male goat. Rats eliminated 7.5% of the dose in the faeces by 72 h, and
only 0.6% and 1.8% of the administered radioactivity was accounted for
in dog and goat faeces, respectively. The rate of degradation of
chlordimeform was also different in the three species. By 24 h after
treatment, 25% of the radioactive material in rat urine was
organosoluble and partitioned into chloroform, but in the dog and goat
urine less than 10% was organosoluble. Levels of chlordimeform
expressed as percentages of organosoluble urinary radioactivity at 24
and 72 h post-treatment were 9.9 and 2.1% for the rat, 1.3 and 0.2%
for the dog, and 0.1 and <0.1% for the goat.
6.1.3 Human
In a study by Nixon & Neal (1983), the excretion of chlordimeform
residues was examined in eight volunteers following dermal
application. A dose of 24.75 mg chlordimeform was applied to the
forearm via a patch which was removed after 4 h and the application
site washed in propanol followed by detergent. The average absorbed
dose was calculated to be 7.95 mg. Urine was collected for 72 h
following treatment. During this period, an average of 38.3% of the
absorbed dose was recovered from the urine. The half-life for
excretion was between 5.9 and 12.1 h, with an average of 8.8 h.
A number of studies have been conducted that monitored the urine
of workers exposed to chlordimeform during use (see section 5.3.2).
The data indicate rapid metabolism of chlordimeform to 4-chloro- o-
toluidine, followed by urinary excretion. Detection in the urine was
as early as 4 h after exposure, and approximately 75% was excreted
within 24 h.
6.2 Metabolic transformation
6.2.1 Mouse and rat
In an early study (FAO/WHO, 1972), the urine from a male rat
collected over 72 h subsequent to oral administration of 1.1 mg
[3H-phenyl]-chlordimeform contained free extractables representing
22% of the [3H] label, of which 10% was in the water phase and 17%
was extractable glucuronides. The free extractable [3H]-label
comprised chlordimeform, 4-chloro- o-toluidine, N-formyl-4-chloro-
o-toluidine, and N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform). Glucuronides were based on the same compounds
found as free extractables.
In a study by Knowles & Sen Gupta (1970), pairs of male and
female rats were treated orally with 3 �Ci [14C-tolyl]-chlordimeform.
Urine and faeces were collected at 3, 12, 24, 48 and 72 h after
dosing. Urinary and faecal elimination of [14C] label after 72 h
comprised 88% and 7.5% of the administered dose of [14C]-
chlordimeform, and 71 and 24.5% of the administered [14C]-4-
chloro- o-toluidine. Chloroform extraction removed 30% of the
radioactivity from the urine of [14C]-chlordimeform-treated rats, the
extract containing chlordimeform, N'-(4-chloro- o-tolyl)-
N-methylformamidine (demethylchlordimeform), N-formyl-4-chloro-
o-toluidine, and 4-chloro- o-toluidine, in addition to three
unidentified metabolites. A considerable amount of radioactivity
remained at the point of origin of the chromatograph, with the amount
remaining increasing with time, (30% at 3 h and 75% at 72 h). At 3 h,
the four identified compounds were present in approximately equal
amounts. By 12 h, the level of N'-(4-chloro- o-tolyl)- N-
methylform-amidine had decreased to approximately 25% of the level of
any of the other three compounds. By 48 h, chlordimeform levels were
half those of the other two compounds, and, by 72 h, N-formyl-
4-chloro- o-toluidine was present in the greatest proportion.
As part of the same study (Knowles & Sen Gupta, 1970), a similar
group of male and female rats received an oral dose of [14C-methyl]-
4-chloro- o-toluidine. The metabolites found in ethyl acetate-
extracted urine comprised 5-chloroanthranilic acid, and N-formyl-
5-chloroanthranilic acid increased. The level of 5-chloroanthranilic
acid remained constant. A large amount (20-50%) of the radioactivity
remained at the origin of the chromatograph. Five unidentified
compounds were noted.
The metabolic transformation of the metabolite
demethyl-chlordimeform ( N'-(4-chloro- o-tolyl)- N-
methylformamidine) in the rat was investigated by Benezet & Knowles
(1976). Eight Sprague-Dawley rats were each administered 1.5 �Ci
[14C]-demethylchlordimeform by oral intubation. Urine and faeces were
analysed over a 72-h period. The majority of the radioactivity was
eliminated through the faeces (64%) but significant amounts were also
eliminated in the urine (35%). The peak level of radioactivity
occurred in the urine between 12 and 24 h, and in the faeces between
18 and 48 h. Of the urinary radioactivity, 16-26% could be extracted
with ethyl acetate. Compounds present included demethylchlordimeform,
N'-(4-chloro- o-tolyl)formamidine, N-formyl-4-chloro- o-
toluidine, 4-chloro- o-toluidine and several unidentified compounds.
The aqueous fraction remaining after ethyl acetate extraction (74-85%
of the total radioactivity) was largely acid-labile and probably
consisted of conjugates, possible glucuronides and ethereal sulfates.
Approximately 25% of the total radioactivity of the faeces was
extractable with ethyl acetate, and similar metabolites were present.
Ifflaender (1977b) examined the quantitative differences in
urinary metabolites between mice and rats following oral
administration of [14C]-chlordimeform at a dose level of 25 mg/kg
body weight. Little quantitative difference in individual
metabolites was observed between the species. Of the total
metabolites, N'-(4-chloro- o-tolyl)- N-methyl formamidine
(demethylchlordimeform) represented 11.3% in rats and 2.4% in mice,
while 4-chloro-2-methyl-phenylurea represented 6.3% in rats and 1.2%
in mice. Sulfuric acid conjugates represented 20.8% in mice compared
to 14.0% in rats. Glucuronic acid conjugates (representing 28% of
metabolites) and all other minor metabolites were in similar amounts
in the urine of rats and mice. Acid hydrolysis of the urine released
degradation products in similar amounts in the urine of rats and mice.
Knowles & Benezet (1977) reassessed the metabolism of
chlordimeform in rat and also assessed the metabolism in mice. Ten
male rats were treated orally with 2 �Ci [14C]-chlordimeform and
urine samples collected at 12 and 24 h. Twelve male mice were injected
intraperitoneally with 0.6 �Ci [14C]-chlordimeform, and urine and
faeces samples were collected at various times up to 96 h. In rat
urine, the major organosoluble metabolites (>10%) included
3-(4-chloro- o-tolyl)urea, N-formyl-4-chloro- o-toluidine,
4-chloro- o-toluidine, and N-formyl-5-chloroanthranilic.
Demethylchlordimeform, didemethylchlordimeform, 1,1-dimethyl-3-
(4-chloro- o-tolyl)urea and 5-chloroanthranilic acid were minor
metabolites. In mouse urine, the majority of the radioactive material
was water soluble, probably consisting mainly of conjugates such as
glucuronides and ethereal sulfates (based on analogy with metabolism
in rats). The major organosoluble metabolites (>10%) were
N-formyl-4-chloro- o-toluidine, 4-chloro- o-toluidine and
N-formyl-5-chloroanthranilic acid. The minor metabolites identified
in rat urine were also present in mouse urine. The identity of the
major metabolites in rat urine were confirmed in the study of Watanabe
& Matsumura (1987).
Knowles & Benezet (1977) proposed the metabolic pathway for
chlordimeform metabolism in rats and mice shown in Fig. 1.
6.2.2 Other species
In the study of Sen Gupta & Knowles (1970) in dogs described in
section 6.1.2, chloroform extraction of the urine removed 10% of the
radioactivity. Thin-layer chromatography of the extract revealed
chlordimeform, N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethyl-chlordimeform) and 4-chloro- o-toluidine in about equal
quantities, but about four times as much N-formyl-4-chloro- o-
toluidine at 1 h after treatment. The level of unchanged chlordimeform
and N'-(4-chloro- o-tolyl)- N-methylformamidine decreased steadily
with time, whereas 4-chloro- o-toluidine and N-formyl-4-chloro- o-
toluidine rose to maximum levels between 6 and 12 h prior to tapering
off. Three unidentified metabolites were present. In addition, a lot
of the radioactivity remained at the origin of the chromatograph.
Re-runs of this material in polar solvents showed 5-chloroanthranilic
acid, N-formyl-5-chloroanthranilic acid and three unidentified
compounds were present. Some radioactivity still remained at the
origin. The urinary [14C] label not extracted by chloroform was
treated with enzymes (�-glucuronidase, �-glucu-ronidase-aryl
sulfatase) to form "aglycones". About 75% of the remaining [14C]
label was extracted in this manner (hydrochloric acid released 62%),
and thin-layer chromatography showed the same compounds as found in
the chloroform extract, the major metabolite being N-formyl-
4-chloro- o-toluidine. In addition, more of one of the unidentified
metabolites was present. Again re-chromatography of the 45% of the
radioactivity remaining at the origin with more polar solvents
revealed 5-chloroanthranilic acid to be the major product. In the
bile, peak concentration of radioactivity occurred at 8 h. About 10%
of this activity could be partitioned into ether, and thin-layer
chromato-graphy of the extract indicated the same four compounds seen
in urine chloroform extract. N'-(4-chloro- o-tolyl)- N-
methylformamidine (demethylchlordimeform), N-formyl-4-chloro-
o-toluidine and an unidentified compound accounted for most of the
activity at 2 h. By 6 h, 75% of the activity was due to N-formyl-4-
chloro- o-toluidine. Incubation of extracted bile with enzymes or
acid gave the same "aglycone" compounds as found in urine. Tissue
residues of [14C] label at 72 h ranged from 72 �g/kg in liver to
30 �g/kg (kidney), 13.5 �g/kg (lung), 11.9 �g/kg (spleen and brain)
and 5 �g/kg (heart and fat and pancreas).
Chemical formula: C10H13ClN2
Relative molecular mass: 196.7
CAS name: N'(4-chloro-2-methylphenyl)-
N, N-dimethyl-methanimidamide
IUPAC name: N2-(4-Chloro- o-tolyl)-
N1, N1-dimethylformamidine
CAS registry number: 6164-98-3 (chlordimeform)
19750-95-9 (chlordimeform hydrochloride)
RTECS number: LQ4375000
Common synonyms: Chlorphenamidine; chlorfenamidine;
chlorophedine; chlorophenamide;
chlorophenamidin; chlorophenamidine;
N'-(4-chloro- o-tolyl)- N,
N-dimethylformamidine;
N, N-dimethyl- N'-(2-methyl-4-
chlorophenyl)-formamidine;
N, N-dimethyl- N'-(2-methyl-4-
chlorophenyl)formadin;
ENT 27335; ENT 27567; EP-333;
N'-(2-methyl-4-chlorophenyl)- N,
N-dimethylformamidine
Trade names: Acaron; Bellotion Especial; Bermat;
Bermatchlorfenamidine; C8514; Carzol;
CDM; CDMS; CGS500; CGS800SP;
Chlorfenamidine; Ciba 8514; Ciba C8514;
COTIP 500EC; Fundal; Fundal 500; Fundex;
Galecron; OMS-1209; Ovatoxion; OVINA;
OVITIX; RS 141; Schering 36268;
Sn 36268; Spanon; Spanone;
SPIKE ULVAIR.
Technical grade chlordimeform is greater than 95% pure and
contains the following impurities: N-formyl-4-chloro-2-toluidine
( N-formyl-4-chloro- o-toluidine), 4-chloro-2-toluidine
(4-chloro- o-toluidine hydrochloride) and sodium chloride.
Chlordimeform free base has been formulated as a 500 g/litre
emulsifiable concentrate. Chlordimeform hydrochloride has been
formulated as a 300 or 800 g/kg water-soluble powder, a 20 g/kg dust
or as 50 g/kg granules.
2.2 Physical and chemical properties
Some of the physical and chemical properties of chlordimeform
base and chlordimeform HCl are shown in Table 1. The molecular
structure of chlordimeform has been investigated by Gifkins & Jacobson
(1980) using single crystal X-ray diffraction.
Table 1. Some physical and chemical properties of chlordimeform
basea
Physical state colourless crystalline solid
Boiling point at 14 mmHg 163 - 165�C
Melting point 32�C
Log Kow 2.89
Vapour pressure at 20�C 48 mPa (3.5 � 10-4 mmHg)
Density (d30) 1.10
Solubility in water at 20�C 250 mg/litre
Solubility in acetone, benzene,
chloroform, ethyl acetate, hexane,
methanol at 20�C >200 g/litre
Half-life at pH 7
(30�C in water, 5% methanol) 42 h
Half-life at pH 9
(30�C in water, 5% methanol) 5 h
Reactivity Forms salt with acids
a From: Worthing (1979); IARC (1978)
Chlordimeform has a solubility in water of 250 mg/litre but is
readily soluble in organic solvents. It forms salts with acids and the
hydrochloride salt is readily soluble in water. When pure,
chlordimeform forms colourless crystals.
Chlordimeform is a base of medium strength with pKa of 6.8 in
50% aqueous methanol (Voss et al., 1973) and forms stable salts with
strong acids.
Chlordimeform is sensitive to light, especially in alkali, and
slowly decomposes in neutral and alkaline aqueous solution. The pH
dependence of photodecomposition of chlordimeform was noted by Su &
Zabik (1972), who observed that an aqueous solution of chlordimeform
hydrochloride (pH 3.1) was unaffected by mercury lamp irradiation for
up to 12 days at 25�C, while a solution of the free base at pH 7-8
decomposed in the same period to a mixture consisting of N-formyl-4-
chloro- o-toluidine and a bis-formamidine. Photo-decomposition of
chlordimeform has also been studied on silica gel chromatographic
plates with irradiation by long- and short-wave ultraviolet light,
fluorescent light and sunlight (under glass) for periods of 10 to 20 h
(Knowles & Sen Gupta, 1969). The major degradation product was again
N-formyl-4-chloro- o-toluidine with either sunlight or UV light.
Fluorescent light caused little decomposition. Sunlight resulted in
12% decomposition in 10 h, while UV resulted in 25% decomposition in
20 h. When 4-chloro- o-toluidine was irradiated with UV light,
numerous decomposition products were found but these were not
characterized further.
Chlordimeform has relatively high volatility and is thus capable
of efficient fumigation action. The hydrochloride salt has negligible
volatility.
2.3 Conversion factors
1 ppm = 8.04 mg/m3 1 mg/m3 = 0.12 ppm
2.4 Analytical methods
2.4.1 Plants
Geissb�hler et al. (1971) described in detail a method for the
determination of total residues of chlordimeform and its metabolites,
which can be used for routine analysis of plant and soil samples. In
this method, chlordimeform and its metabolites are hydrolysed to
4-chloro- o-toluidine by successive treatments with acetic acid and
sodium hydroxide, respectively. The hydrolysis product is then steam
distilled, extracted with isooctane, diazotized and coupled with
N-ethyl-1-naphthylamine yielding a purple dye, which, after column
chromatography on cellulose, is determined by colorimetry. Interfering
azo-dyes from aromatic plants or soil are removed by chromatography on
a cellulose column. This colorimetric method has a limit of detection
of 0.05 mg/kg. If required, the identity of the residues can be
verified by thin-layer chromatography on a cellulose column. This
procedure is sensitive to about 0.1 mg/kg. Alternatively, the
hydrolysis product, 4-chloro- o-toluidine, is diazotized and
iodinated, and the iodinated derivative is measured by electron-
capture gas chromatography. This alternative method has a limit of
detection of 0.05 mg/kg.
Kossmann et al. (1971) refined the method of Geissb�hler et al.
(1971) to permit separate determination of residue quantities of the
parent compound and its potential degradation products in plant
materials. In this procedure, plant material is subject to a two-fold
extraction, the first with methanol/hydrochloric acid and the second
with the lipophilic mixture, methanol/methylene chloride. Separation
of chlordimeform and its degradation products is accomplished by
thin-layer chromatography. The separated eluants are converted to
4-chloro- o-toluidine and analysed as described by Geissb�hler et al.
(1971). The limits of detection for the separated compounds,
chlordimeform, demethylchlordimeform and 4-chloro- o-toluidine are
0.02 to 0.03 mg/kg.
Gr�bner (1977) described a thin-layer chromatographic method for
the determination of chlordimeform residues alone or together with its
metabolite, 4-chloro- o-toluidine, in cucumbers and apples. The
limits of detection for chlordimeform and 4-chloro -o-toluidine were
0.1 and 0.05 mg/kg, respectively. The rates of recovery were 76-85 and
90-105%, respectively.
Fan & Ge (1982) described an alkali flame ionization
gas-chromatographic method for the determination of chlordimeform and
three potential metabolites in cargo rice and husk. Residues of
chlordimeform and its metabolites were extracted with absolute alcohol
or hexane and cleaned up on neutral alumina columns, before being
chromatographed in a column of 1% DEGS coated on 60-80 mesh
405 support (PEG 20M bonded phase). The detection limits for
chlordimeform, 4-chloro- o-toluidine, 2,2'-dimethyl-4,
4'-dichloroazobenzene, and N-formyl-4-chloro- o-toluidine were
0.03, 0.028, 0.11 and 0.43 mg/kg, respectively, for cargo rice and
0.03, 0.028, 0.22 and 0.43 mg/kg, respectively, for husk. Recovery for
chlordimeform was 81-93% for cargo rice and 103-104% for husk.
Recovery for 4-chloro- o-toluidine was 71-73% for both cargo rice
and husk. Recovery for 2,2'-dimethyl-4,4'-dichloroazobenzene was
81.8-112% for cargo rice and 109-118% for husk. Recovery for
N-formyl-4-chloro- o-toluidine was 66% for husk. Mattern et al.
(1991) described a rapid analytical procedure for 17 pesticides,
including chlordimeform, using gas chromatography/chemical ionization
mass spectrometry (GC/CIMS) for detection in various commodities
including peppers, spinach, lettuce and snap beans. Percentage
recoveries for chlordimeform were 87.8% (peppers), 72.6% (spinach),
99.7% (lettuce) and 94.7% (beans). The limits of detection for
chlordimeform were 0.05 mg/kg (beans), 0.05 mg/kg (lettuce),
0.05 mg/kg (peppers) and 0.10 mg/kg (spinach).
2.4.2 Soil
The method of Geissb�hler et al. (1971) described in section
2.4.1 for plants can equally be applied to the determination of total
residues of chlordimeform in soil.
2.4.3 Water
Machin & Dingle (1977) described a UV spectrographic method for
the determination of chlordimeform in cattle dipping baths and
sprays. Preliminary clean-up removes UV-absorbing impurities and
converts chlordimeform to its hydrochloride. Following silica gel
chromatography, the absorbance of the non-eluted material is measured
at 240 nm to determine chlordimeform content. Optimum results are
obtained in the concentration range of 0.02-0.06% (w/v) chlordimeform.
2.4.4 Formulations
Voss et al. (1973) described two methods for the determination of
chlordimeform in formulations. The first relies on acid titration of
the free base with hydrochloric acid. The hydrochloride salt is
converted into the free chlordimeform base, which is extracted into an
organic solvent. After evaporation of the solvent, the active
ingredient is determined potentiometrically. The second method makes
use of gas chromatography, and in this case the chlordimeform
hydrochloride preparations have to be converted into the base form
prior to injection into the gas chromatograph.
Gale & Hofberg (1985) described a gas chromatographic procedure
for the determination of chlordimeform in emulsifiable concentrate
formulations. Chlordimeform was extracted with methylene chloride,
chromatographed on CBWX-20M and detected by flame ionization.
2.4.5 Air
There are no published methods described for the determination of
chlordimeform in air.
2.4.6 Urine
Liu & Mao (1980) described a method for the gas chromatographic
separation of chlordimeform, demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine in urine. Optimum
separation was achieved on a column with 1% polyvinylpyrolidone and 8%
PEG 20M on 80-100 mesh white diatomeous support no. 101 (acid and base
washed). The column was suitable for both qualitative and quantitative
analysis.
A method to analyse urinary residues of workers occupationally
exposed to chlordimeform was developed by Ciba-Geigy in 1980
(Anonymous, 1980a). The method relies on the hydrolysis of
chlordimeform and other residues to 4-chloro- o-toluidine with sodium
hydroxide, followed by extraction with hexane and separation on
reverse-phase liquid chromatography fitted with a UV detector. A
published version of this method was prepared by Geyer & Fattal (1987)
in which the alkaline hydrolysate of urine is extracted with hexane,
the solvent is evaporated, and the hydrolysate is reconstituted with
aqueous acetonitrile. Separation was performed on a reverse-phase Novo
Pak 5 mm C18 column with a UV absorbance detector equipped with a 254
nm filter. A similar method was described by Cheung et al. (1989) for
the analysis of chlordimeform from urine of field workers. Ross &
Leisten (1989) have refined this method with the use of synchronous
spectral data which provides a improved signal-to-noise ratio, which
gives lower minimum detectable levels while still allowing a
well-resolved spectrum. This system may allow detection of levels
equivalent to 1 mg/litre in urine.
2.4.7 Tissues
A gas chromatographic method for the determination of residues of
chlordimeform in animal tissues was first described in the early 1970s
(Anonymous, 1971a). The method involves hydrolysis of chlordimeform to
4-chloro- o-toluidine by successive treatments with acetic acid and
sodium hydroxide. The hydrolysis product is steam distilled and
extracted into isooctane. Following diazotization of the 4-chloro-
o-toluidine, the diazo-moiety is exchanged for iodine by potassium
iodide treatment. The iodinated derivative is gas chromatographed
using electron-capture detection. The limit of detection using this
method is 0.02 mg/kg.
Rieger et al. (1985) have described a gas chromatography/flame
ionization detection method for the determination of chlordimeform and
its major metabolite, demethylchlordimeform, from human tissue
samples, namely, human whole blood and human liver (1:1 aqueous
homogenate). Tissues were first extracted with an organic solvent,
transferred to an acid aqueous medium (0.1M hydrochloric acid),
re-extracted into a small volume of organic solvent and separated on
GC or GC/MS. Using extraction with either chloroform or n-butanol,
recoveries of 81 and 75%, respectively, were obtained.
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Chlordimeform does not occur naturally.
3.2 Anthropogenic sources
3.2.1 Production levels and processes
Chlordimeform was first commercialized in 1966. It can be
manufactured commercially by two methods (Voss et al., 1973), both
starting with the conversion of dimethylformamide to the Vilsmeier
reagent by reaction with POCl3, SOCl2 or COCl2.
By the first method, condensation of the Vilsmeier reagent with
4-chloro-amino-toluene (or 5-chloro-2-aminotoluene, 5-CAT) leads
directly to chlordimeform hydrochloride. Treatment with a strong base
gives the free chlordimeform base.
By the second method, the Vilsmeier reagent is reacted with
o-toluidine to give phenamidine, which is chlorinated in a second
step. The chlorination gives rise to a certain amount of isomers as
unwanted side-products. The crude chlordimeform so obtained has to be
purified either by recrystallization of its chlorohydrate or by
rectification of the free base.
Chlordimeform has been produced at various times in Switzerland,
Germany, United Kingdom, USA, Italy, Argentina and China.
Little information is available on the production levels of
chlordimeform. Information from the US International Trade Commission
(IARC, 1983) indicated that imports of chlordimeform to the USA
through the principal US customs districts amounted to 745 tonnes in
1979 and 198 tonnes in 1980.
In 1974, total usage of chlordimeform in the USA is estimated to
have been 590 tonnes, 77% of which was used on cotton, 15% on
deciduous fruits and nuts, and 8% on vegetables. In 1976, the US
Department of Agriculture reported that 2000 tonnes of chlordimeform
was used in the USA on major crops (IARC, 1983). In 1980, total usage
in the USA was 227 tonnes, all of which was used on cotton to control
budworm/bollworm.
Chlordimeform has been used in China throughout the 1970s and the
1980s at the rate of approximately 10 000 to 15 000 tonnes per year
(Xue, personal communication). In the Chinese province of Hu-bei, the
average annual usage during the period 1984-1988 was 3276 tonnes
(Huang et al., 1989).
3.2.2 Uses
Chlordimeform is a broad spectrum acaricide and is active mainly
against eggs and motile forms of mites and ticks and against eggs and
early instars of some Lepidoptera insects. It kills eggs, larvae and
adults not only by contact but also in the vapour phase. The major use
initially was in the control of mites on deciduous fruit.
In 1971, chlordimeform products were registered in many countries
for use on a wide variety of crops such as pome fruits, stone fruits,
cole crops, vegetables, grapes, hops, citrus, apples, pears, cherries
and strawberries. Chlordimeform also had important veterinary uses as
an acaricide. In Australia, chlordimeform was registered for use in
cattle dips for the control of cattle ticks (Boophilis mictopus), in
combination with organophosphorus acaricides (FAO/WHO, 1972).
In 1975, it was reported that the use pattern of chlordimeform
had been extended to include control of stemborers in irrigated rice,
control of Lepidoptera larvae on cotton, and control of a wide range
of Lepidoptera larvae on cabbage and tomatoes (FAO/WHO, 1976). At
this time, the control of stemborers in irrigated rice proved to be
one of the most important uses of chlordimeform. In the case of
cotton, chlordimeform became one of the most important substitutes for
DDT and other organochlorine pesticides.
Chlordimeform has had no significant usage in non-crop situations
other than on ornamentals.
In 1976, the manufacturers temporarily suspended the sale of
chlordimeform from all markets worldwide, on the basis of adverse
carcinogenicity findings in chronic mouse studies.
In 1978, having completed a number of toxicology, metabolism and
residue studies, the manufacturers re-applied in a number of countries
for registration to allow limited commercial use in cotton crops only.
The proposal was to use chlordimeform by aerial application under
supervised conditions that limited the uptake by operators and
by-standers. Chlordimeform was re-introduced for insect control in
cotton in USA, Central America, Columbia, Israel, Australia and China.
Guidelines for the handling and use of chlordimeform were set in
Australia, Columbia, Israel and USA (California). Application rates
were set to minimize the occurrence of residues in cotton fibres and
cotton seed oil. In China, extensive use of chlordimeform continued
through the 1980s on rice and cotton.
Use of chlordimeform ceased in most countries in the mid to late
1980. The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) withdrew
its temporary Acceptable Daily Intake (ADI) in 1987 and recommended
that chlordimeform should not be used where its residues, or those of
its metabolite, 4-chloro- o-toluidine, could arise in food. (FAO/WHO,
1988).
In 1988-1989, Ciba-Geigy and Schering voluntarily and finally
halted marketing of chlordimeform and decided to withdraw registration
worldwide. In China, production stopped at the end of 1992, and sales
ceased in June 1993.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1 Transport and distribution between media
4.1.1 Air
Chlordimeform has relatively high volatility, and thus when
sprayed on crops considerable evaporation would be expected from plant
surfaces as well as from the soil. Studies in plants, however,
indicate a lower rate of evaporation than expected. In bean plants,
disappearance from the surface in the first few hours was found to be
of the order of only 30-40% of the original dose applied (FAO/WHO,
1972). This result was obtained when either chlordimeform or its
hydrochloride salt was used and is considered to be due to the
buffering capacity of plant exudates with a resulting equilibrium
between the free base and salts. The low volatility from plant
surfaces was confirmed by Sen Gupta & Knowles (1969) on apple
seedlings and by Ehrhardt & Knowles (1970) on grapefruit seedlings. In
cotton plants, approximately 55% of the dose applied to leaves was
volatilized from the surface of the leaves within 2 h (Bull, 1973).
No studies are available on the volatilization of chlordimeform
from soil surfaces, but it is likely to be at least as high as from
leaf surfaces.
4.1.2 Water
While chlordimeform base has only low solubility in water, the
solubility of the hydrochloride salt is relatively high. Its stability
in water, however, is highly pH-dependent, and in the normally neutral
to slightly alkaline conditions of rivers and lakes its half-life
would be relatively short.
It also has the potential to adsorb readily to dissolved organic
matter resulting in precipitation (Maqueda et al., 1989).
The hydrolytic stability of chlordimeform is highly pH-dependent.
It slowly hydrolyses in neutral pH and is stable in strongly acid
conditions. The half-life at 10�C is about 38 days at pH 7, compared
to 8 days at pH 8. At 30�C, these values are reduced to about 3 and
0.5 days, respectively. A solution of the hydrochloride salt (pH 3-4)
showed no appreciable hydrolysis over several days (Su & Zabik, 1972).
The principal product of hydrolysis is N-formyl-4-chloro- o-
toluidine, which at room temperature is very slowly converted to
4-chloro- o-toluidine by further hydrolysis. The second step may be
accelerated by heating with strong acid or alkali.
4.1.3 Soil
Hydrolysis of chlordimeform to N-formyl-4-chloro- o-toluidine
would be expected to be significant under the slightly acid or
slightly alkaline conditions that normally prevail in soils.
Despite the reasonably high solubility of the hydrochloride salt
of chlordimeform, there appears to be little leaching from the site of
application in the soil (FAO/WHO, 1972).
In the studies by Fischer & Cassidy on the uptake of
chlordimeform from soil into cotton plants, the levels of
chlordimeform in the soil were also analysed (FAO/WHO, 1979). Soil was
treated when the cotton reached 10 weeks of maturity. Radioactivity in
the top 75-mm layer of silt loam soil accounted for 1.23 mg/kg
chlordimeform equivalents after treatment. At 7 weeks, this level had
decreased to 0.33 mg/kg and at 13 weeks to 0.20 mg/kg. Extraction of
this layer revealed partition of 32% into the organic layer and 20%
into the polar fraction, and 44% was non-extractable, indicating rapid
degradation. For all but one sample, the level of radioactivity as
chlordimeform equivalents in the lower soil levels, 75-150 mm and
150-200 mm, was less than 0.01 mg/kg, indicating that leaching did not
occur in silt loam. In later experiments with regular over-the-top
spray treatment throughout the maturation of the cotton plants, the
same rapid decrease in radioactivity (as chlordimeform equivalents)
was seen in the top 75 mm of soil. Radioactivity in deeper layers was
again equivalent to less than 0.01 mg/kg. At harvest of the cotton
plants, up to 91% of the radioactivity in the soil could be converted
to 4-chloro- o-toluidine.
The nature of the non-extractable portion of chlordimeform in
soil was investigated by Perez-Rodriguez & Hermosin (1979) and by
Hermosin & Perez-Rodriguez (1981) in experiments examining the
interaction of chlordimeform with clay minerals, montmorillonite,
kaolinite, illite and vermiculite. The earlier work indicated that the
adsorption of chlordimeform on clay is essentially a cation-exchange
reaction and that chlordimeform ions lie between the silicate layers,
thus being difficult to disperse with water or aqueous solutions of
inorganic cations. In the later study, chlordimeform adsorption to the
clay minerals montmorillonite, illite and vermiculite was found to be
an irreversible process, whereas chlordimeform adsorbed on kaolinite
is only weakly bonded and easily removed by washing with water.
The role of soil organic matter in the adsorption and degradation
of chlordimeform in soil was examined in experiments by Maqueda et al.
(1983, 1989). In the first study, the interaction of chlordimeform
with humic acid extracted from the top 20 cm of a clay soil classified
as Typic Chromozerert soil was examined. Adsorption is essentially a
cation-exchange process, although other mechanisms, such as charge
transfer, H-bonding, and van der Waals forces may contribute to the
high adsorption capacity. The variety of mechanism may make it
difficult to ascertain the long-term fate in the environment. In the
second study, the interaction of chlordimeform and other pesticides
with fulvic acids extracted from a spodosol soil was examined. Fulvic
acids are the fraction of humic substances that dissolves in both acid
and alkaline media, and thus are readily found solubilized in lakes
and rivers. The adsorption of chlordimeform was again shown to be a
cation-exchange process, together with H-bonding and charge transfer
mechanisms. Precipitation occurred upon interaction of chlordimeform
with fulvic acids. The amount of precipitate increased in a
dose-related manner up to levels of 100 mmol chlordimeform/litre.
4.1.4 Vegetation and wildlife
Benezet & Knowles (1981) examined the degradation of
chlordimeform by two algal types, Chlorella, the green alga,
and Oscillatoria, a cyanobacterium. In the presence of either
Chlorella or Oscillatoria, chlordimeform was hydrolysed to
N-formyl-4-chloro- o-toluidine, probably by a largely non-enzymatic
reaction. Further reaction formed 4-chloro- o-toluidine and some
CO2. Oxidative N-demethylation was not a major path for chlordimeform
degradation by algae.
The solubility of chlordimeform was sufficient to allow uptake
by the roots of bean and rice plants and to be transported to
plant-feeding pests, as demonstrated by the efficacy experiments of
Dittrich (1967) and Dittrich & Loncarevic (1971).
The ability of plants to take up chlordimeform from soil was
further demonstrated by the experiments of Fischer & Cassidy
(FAO/WHO, 1979), where the soil of a cotton field was treated with
[14C]-chlordimeform when the cotton was 10 weeks old. Uptake of the
radioactivity by the cotton plant was noted to occur in small
quantities, and the highest levels were found in the seeds and fibres.
Biphasic extraction showed 42% in the organic fraction and 34% in the
polar fraction, and 24% was not extractable. Thirteen weeks after
treatment, the mature cotton contained 0.09 mg/kg in the leaves.
The low level of translocation of chlordimeform in plants was
demonstrated by Sen Gupta & Knowles (1969) in experiments where
[14C]-chlordimeform was injected into the stem of apple seedlings
followed by analysis of stem and leaf radioactivity for a period of 20
days. For the first 4 days after injection, 95% of the radioactivity
was localized in the stems, predominantly as the parent compound.
After 20 days, 71.6% of the radioactivity still remained in the stem,
with 25.4% in the leaves, and only 17.9% remained as the parent
compound. The major portion of the radioactivity in the stems after 20
days was unextractable with chloroform and acetone.
In the experiments of Ehrhardt & Knowles (1970) with grapefruit
seedlings, there was no detectable movement of radioactivity into
adjacent stems and leaves 8 days after application of [14C]-
chlordimeform to two upper leaves or two lower leaves. Considerable
movement into stems and leaves was noted when [14C]-chlordimeform was
injected into the main stem, and also to the periphery of grapefruit
leaves when it is applied centrally. Thus, movement of chlordimeform
occurred mainly in the direction of the xylematic transpiration
stream.
Application of chlordimeform directly to the leaves of apple
seedlings (Sen Gupta & Knowles, 1969) or the leaves of grapefruit
seedlings (Ehrhardt & Knowles, 1970) demonstrated the limited capacity
of chlordimeform to penetrate the cuticular layers. Ercegovich et al.
(1972) reported that chlordimeform appeared to adhere to the outer
surface of fruit and did not appear to translocate readily to the
fleshy parts. The chief factors which seem to account for the decrease
of chlordimeform residues in fruit appear to be volatilization,
weathering and growth dilution.
Similarly, the application of [14C]-chlordimeform to cotton
leaves resulted in little movement of radioactivity (and none of
chlordimeform itself) into the untreated plant parts. The small amount
of translocated radioactivity consisted exclusively of polar, mainly
non-extractable substances (Gross, 1977).
In a field experiment, Fischer & Cassidy treated a cotton field
plot over-the-top with [14C]-chlordimeform at a rate of 1 kg/ha when
plants were 12-14 weeks old (FAO/WHO, 1979). Radioactivity in the
cotton plants immediately after treatment was the equivalent of
2.44 mg/kg chlordimeform. At harvest, the radioactivity calculated as
[14C]-chlordimeform was 12.91 mg/kg in the leaves, 0.99 mg/kg in
the stalks, 0.03 mg/kg in the fibre, and 0.26 mg/kg in the seed, with
0.07 mg/kg in the oil and 0.19 mg/kg in the meal. Parent chlordimeform
accounted for 31% and 45.2% in the leaves and stalks, respectively.
The data indicated that although leaf radioactivity is high, there is
still little translocation of [14C]-chlordimeform metabolites to the
seed or fibre.
Supervised residue trials to determine the residue levels in
cottonseed and cottonseed products have been conducted (FAO/WHO,
1979). In general, there is a correlation between the application rate
and the residue level but the interval between the last application
and the harvest also has a strong influence on the residue level. The
decrease of residues with time was most pronounced during the first 10
days after treatment of the cotton plants. At the maximum application
rate of 1 kg/ha, the residue level rarely exceeded 2 mg/kg in
cottonseed, seed meal or crude oil.
When used for the control of rice stem borer in Japan,
chlordimeform resulted in low levels of residues in rice grains and
straws. In rice grain after three treatments, the residue levels of
chlordimeform, demethylchlordimeform, N-formyl-4-chloro- o-
toluidine and 4-chloro- o-toluidine were 48, 0.4, 15 and 53 �g/kg,
respectively. The results indicate a low level of penetration of
chlordimeform into rice plants. The chlordimeform that entered the
plant was gradually degraded to 4-chloro- o-toluidine (Iizuka &
Masuda, 1979).
There have been no studies conducted on the uptake of
chlordimeform by wildlife. Studies with experimental animals suggest
rapid metabolism and excretion, with negligible retention.
4.1.5 Entry into food chain
Potential routes of entry of chlordimeform into the human diet
include the direct consumption of crops containing chlordimeform
residues, the consumption of processed food prepared from treated
crops, or the consumption of animal products derived from animals
treated topically with chlordimeform or raised on chlordimeform-
containing feed such as cottonseed.
Since the temporary withdrawal of the use of chlordimeform from
the market in 1976 in most countries and the later restriction to use
on cotton, dietary consumption of chlordimeform residues on crops in
these countries has virtually ceased. However, dietary consumption of
chlordimeform residues is likely to have continued at least until the
late 1980s in some areas (see section 5.2.2). The maximum residue
levels (MRLs) which were used for chlordimeform are discussed in
section 5.2.2.
4.2 Biotransformation
4.2.1 Degradation in plants
Data reviewed by JMPR (FAO/WHO, 1972) demonstrated that
chlordimeform was quite rapidly degraded in plants with a high
inherent metabolic activity (e.g., bean plants) but was only slowly
degraded in ripe fruits. Green fruits (e.g., grapes) and stems have
an intermediate rate of degradation of chlordimeform. Tentative
identification of the observed metabolites indicated that in
leaves both N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform) and N-formyl-4-chloro- o-toluidine were
major metabolites. In ripe apple and pear fruit, however, only
N'-formyl-4-chloro- o-toluidine was detected. In all tissues,
4-chloro- o-toluidine was either not detected or present in small
quantities, even when six-fold overdose treatment was used.
In the experiments of Sen Gupta & Knowles (1969), [3H]- or
[14C]-chlordimeform was applied to apple seedlings by either leaf
treatment or stem injection. The half-life of degradation was about
12-16 days, and after 20 days 40% of the radioactivity was still
unchanged chlordimeform. Organosoluble degradation products were
identified as demethylchlordimeform, N-formyl-4-chloro- o-toluidine
and 4-chloro- o-toluidine, with the last two representing less than
1% of the total radioactivity. Non-extractable radioactivity, possibly
chlordimeform degradation products complexed with polymeric cell
constituents, was observed only after stem application.
In the experiments of Ehrhardt & Knowles (1970), both
[14C]-chlordimeform and [14C]-chlordimeform hydrochloride were
applied to the leaf surface of growing grapefruit seedlings. After 20
days, only 10-20% of total radioactivity was recovered, possibly due
to evaporation from leaves, and only 1% of radioactivity was unchanged
chlordimeform. The pattern of metabolites was essentially the same as
in apple seedlings, but the levels were smaller.
Witkonton & Ercegovich (1972) examined the metabolites found in
six different fruits (apples, pears, cherries, plums, strawberries and
peaches) following treatment at varying rates with chlordimeform.
Samples of the fruit were collected at various intervals after the
last application from orchards and plants that had been treated with
aqueous sprays of chlordimeform. Of the three potential degradation
products analysed for, only one, namely, N-formyl-4-chloro- o-
toluidine, was detected, together with the parent compound. The other
potential degradation products, namely, demethylchlordimeform and
4-chloro- o-toluidine, were not detected. There was no correlation
between the amount of chlordimeform and 4-chloro- o-toluidine and the
application rate or the sampling interval. The nature of the fruit and
environmental factors were accredited as the major contributing
factors governing the formation and retention of 4-chloro- o-
toluidine. At harvest, the total residue in all crops was
approximately 1 mg/kg, except in peaches, which had approximately
2 mg/kg of total residue. The chief factors which appeared to account
for the decrease in chlordimeform residues were weathering and growth
dilution, rather than chemical or enzymatic degradation.
The potential formation of azo-derivatives of chlordimeform or
its metabolite, 4-chloro- o-toluidine, in treated fruit and
vegetables under field conditions was investigated by Geissbuhler et
al. (1971) using a sensitive gas-chromatographic residue method that
allowed the detection of 0.01 mg/kg of 2,2'-dimethyl-4,4'-
dichloroazobenzene. At 20, 30 or 40 days after a 4-fold overdose
treatment by chlordimeform to apple fruits and leaves, residues of the
azobenzene compound were either not detectable or detected at very low
levels (0.04 mg/kg) in leaves. At normal levels of treatment, residues
of azobenzene compounds would be unlikely to be detected. This result
is supported by the experiments of Witkonton (1973), who analysed
the residues on apple surfaces 60 days after treatment with
[14C]-chlordimeform. The results of these experiments do not support
the in vitro studies of Rose (1969a,b), which indicate the potential
formation of azobenzene derivatives in plants by plant peroxidases.
The metabolism of chlordimeform in cotton plants was first
examined by Bull (1973) following treatment of individual leaves with
[14C]-chlordimeform by petiole injection or by foliar application.
About 45% of the applied dose was absorbed by the leaves, and the
balance volatilized from the leaf surface within 2 h. Tentative
identification of metabolites included demethylchlordimeform,
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine. After
1 h, only 2% of the applied dose could be recovered from leaf
surfaces. The unextractable radioactivity was considered to represent
decomposition products bound to insoluble plant material.
Gross (1977) studied the metabolism of [14C]-chlordimeform in
greenhouse-grown cotton plants following treatment of leaves at a rate
equivalent to 0.6 kg a.i./ha. Metabolites were extracted into hexane,
methylene chloride and water-soluble fractions at various times up to
11 weeks after treatment. The radioactivity in the organic fractions
consisted of at least seven substances. Four were characterized by TLC
as chlordimeform, N-demethylchlordimeform, 4-chloro- o-toluidine
and N-formyl-4-chloro- o-toluidine. Fifty-six percent of the dose
was found in the plant after one week, the balance being lost by
volatilization. The main degradation pathway was hydrolysis,
demethylation only being significant at later sampling times. The loss
of chlordimeform from the surface of leaves was confirmed by
Wolfenbarger et al. (1979) who noted that 24 h after cotton leaves
were treated topically with chlordimeform, only 5% of the EC form was
recovered, whereas 25% of the HCl salt was recovered.
Fischer & Cassidy (FAO/WHO, 1979) identified the metabolites in
leaves after [14C]-chlordimeform was sprayed over-the-top on cotton
plants. At mature harvest, the radioactivity in the leaves consisted
of chlordimeform (60.3%), demethylchlordimeform (4.1%), 4-chloro-
o-toluidine ((7.6%) and N-formyl-4-chloro- o-toluidine (7%). The
results indicate that the parent chlordimeform will be the major
chemical residue in the mature cotton foliage.
Honeycutt & Cassidy (1977) investigated the metabolism of
chlordimeform in cottonseed following injection of [14C]-
chlordimeform into the stem of a growing cotton plant. Forty percent
of the radioactivity in the cottonseed was not extractable. Total
hydrolysis of the radioactivity in the cottonseed showed that a total
of 19.8% of the radioactivity could be converted to 4-chloro- o-
toluidine. The data indicated that the metabolism of chlordimeform in
cottonseed is extensive and results in conjugation to natural
products.
4.2.2 Degradation in soils
The potential for microbial degradation of chlordimeform in
the soil was first identified by Johnson & Knowles (1970), who
demonstrated the capability of several bacteria (Aerobacter
aerogenes and Serratia marcesens), actinomycetes (Streptomyces
griseus) and fungi (Fusarium moniliforme and Rhizopus nigricans)
in culture media to degrade chlordimeform extensively. The
principal metabolite of the bacterial and fungal species was
N-formyl-4-chloro- o-toluidine, while for the actinomycete,
Streptomyces griseus, the principal metabolite was 4-chloro- o-
toluidine. 4-Chloro- o-toluidine was also formed by the bacteria and
fungi. None of the microbes formed symmetrical azo-compounds.
The metabolic fate of chlordimeform in sandy loam over a one-year
period was examined by Iwan & Goller (1975). Soil samples containing
2 �Ci of either [14C- ring]- or [14C- tolyl]-chlordimeform were
prepared in an environmental chamber and methanol/benzene extracts
examined at various intervals. Extractability decreased to 50% within
7 days and was less than 2% after 360 days. In sterilized soil
samples, on the other hand, extractability decreased only slowly, and
70% was still extractable after 180 days. This result indicates that
microbial activity plays a major role in soil degradation of
chlordimeform to non-extractable components. Even though bound to
soil, degradation of chlordimeform continued, as shown by the release
of CO2 as a consequence of oxidative attack upon the tolyl group.
Little CO2 was released under anaerobic conditions and no CO2 was
released from sterile samples. The major pathway of metabolism was
through hydrolysis to 4-chloro- o-toluidine but oxidative
N-demethylation was also a significant pathway leading to
4-chloro- o-toluidine. Further hydrolysis steps followed. The azo
compound, 2,2'-dimethyl-4,4'-dichloroazobenzene, was formed in small
amounts only when the initial chlordimeform concentration was
200 mg/kg in the soil samples. Anaerobic conditions produced the same
metabolic products with the exception of oxidative products such as
demethylchlordimeform. The data suggests that even under sterile
conditions, the degradation of chlordimeform is rapid and its
half-life in non-sterile soils should not exceed one month.
In a further study, Iwan et al. (1976) isolated from
chlordimeform-treated soil four coupling products formed by one-
electron oxidation of 4-chloro- o-toluidine by soil microorganisms.
The four products, one of which is 2,2'-dimethyl-4,4'-
dichloroazobenzene, are formed only from high concentrations of
chlordimeform (70-100 mg/kg), which are at least 10 times higher than
the levels occurring after field application.
4.2.3 Bioaccumulation
There is no data to indicate that chlordimeform bioaccumulates in
plant or animal tissues. However, with a low Kow of 2.89, this
indicates a moderate potential to bioaccumulate.
4.3 Ultimate fate following use
Chlordimeform in the air and in water would be expected to
undergo photodecomposition. In water as well as in soil, chemical
hydrolysis occurs together with adsorption to organic and clay
materials. In plants, residues form complexes with polymeric cell
constituents.
Chlordimeform can be hydrolysed readily to 4-chloro- o-toluidine
by heating with alkali. For the disposal of small quantities of unused
pesticide, the following method is recommended: mix with excess lime
(CaO) or sodium hydroxide (NaOH) and sand and bury at least 0.5 m
below the surface in clay soils. Commercial formulations require
0.5-1.0 kg alkali per kg of pesticide. Alkali can be reduced by 50%
for dilute formulations, e.g., 1% solution or dust. For very
concentrated pesticides (> 50% a.i.), double the amount of alkali and
mix the pesticide with soapy water, before reaction with alkali. Test
reaction on small scale to discover whether or not it will be too
vigorous. Larger quantities should be treated in small batches or
burned in a high-temperature incinerator equipped with effluent gas
scrubbing (IRPTC, 1992).
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air and water
There are no specific data available on the monitoring of
chlordimeform levels in air and water. In neutral and alkaline
solutions, relatively rapid degradation would be expected owing to
hydrolytic instability. Under acidic conditions, slower degradation
would be expected (Su & Zabik, 1972). Adsorption to organic matter in
water would also be expected under field conditions. In both media,
there would be degradation due to photodecomposition (Knowles & Sen
Gupta, 1969).
5.1.2 Soil
Chlordimeform deposited inadvertently on soil surfaces after
spray application may be expected to dissipate by the following
processes: volatilization, chemical hydrolysis, photodecomposition and
microbial degradation. Under field conditions, chlordimeform and its
4-chloro- o-toluidine-containing metabolites are dissipated according
to first-order reactions with half-lives ranging from 20 to 40 days
(Guth & Senn, 1969; FAO/WHO, 1972). The conclusion from these
experiments is that accumulation of chlordimeform in the soil would
not be expected.
Following three applications to rice paddy fields for the control
of rice stem borer, residues of chlordimeform, demethylchlordimeform,
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
2900, 9, 190 and 68 �g/kg, respectively, in the top 5 cm of soil, and
were 150, 1, 8 and 20 �g/kg, respectively, in the 5-10 cm level of
soil. These results indicate the presence of chlordimeform and its
degradation products mainly in the upper layer with minimal movement
downward (Iizuka & Masuda, 1979).
5.2 General population exposure
5.2.1 Environmental sources
There are no longer any environmental sources for exposure of the
general population to chlordimeform. While chlordimeform was being
used on cotton, there was potential for general population exposure to
spray drift from aerial application. The persistence of residues of
chlordimeform on the leaves of cotton also raised the possibility of
exposure through contact with the leaves during the growing period or
during harvesting.
5.2.2 Residues in raw produce
Prior to the temporary suspension of its use in 1976,
chlordimeform was used on a wide variety of crops and on livestock.
The temporary maximum residue levels (MRLs) shown in Table 2 were
established at the 1971 meeting of the Joint Meeting on Pesticide
Residues (JMPR) (FAO/WHO, 1972) as a result of numerous residue trials
in various countries. Residue trials indicated that whilst there was a
sharp drop in the residue level between the day of application and the
second or third day post-treatment, thereafter the rate of decline was
remarkably slow with a half-life on apples, grapes, pears and tomatoes
exceeding 21 days.
Table 2. Temporary tolerances for chlordimeform established in 1971
(FAO/WHO, 1972)
Temporary tolerance mg/kg
Pears, peaches, prunes 5
Apples, grapes, plums, strawberries 3
Brassicas, cherries, citrus fruit, cotton seed oil
(crude and refined), cotton seed 2
Beans 0.5
Fat, meat and meat products of cattle 0.5
Milk (whole) 0.05
Butter 0.5
In 1975, the temporary MRL for pears was raised to 10 mg/kg, and
new temporary MRLs were established for tomatoes (1 mg/kg) and hulled
rice (0.1 mg/kg) (FAO/WHO, 1976). In 1978, the JMPR meeting retained
only the MRLs for cottonseed and recommended that for cottonseed oil
(edible), meat of cattle, pigs, poultry and sheep, and milk and milk
products no residues should occur at the current limit of detection
(0.05 mg/kg) (FAO/WHO, 1979).
The proportion of metabolites and parent compound in the residues
remaining on fruits at various times after application have been
determined in numerous trials. In general, the parent compound
represents the major residue (>80%), followed by N-formyl-4-chloro-
o-toluidine, N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform) and 4-chloro- o-toluidine.
In Chinese residue trials, chlordimeform residues on green
cabbage after application by direct spraying of a 800-fold dilution of
25% chlor-dimeform formulation were 20.9 mg/kg after 4 h, 11.5 mg/kg
after 2 days, 4.2 mg/kg after 7 days and 0.02 mg/kg after 14 days
(Anonymous, 1980b).
In a paper by the Chinese Special Task Group on the residues of
chlordimeform (Anonymous, 1981), the residues of chlordimeform in rice
plants during the period 1974-1980 were examined. In the period
1974-1975, after a single application of 25% aqueous chlordimeform
(9-11 litre/ha) the residue levels on rice harvested after 33-40 days
were 0.25-0.28 mg/kg. When applied at half this rate, residue levels
on rice harvested after 20-74 days were 0.17-0.71 mg/kg. In
field studies in Beijing in 1977, with the same single rate of
application, residue levels on rice harvested after 19-42 days were
0.37-0.51 mg/kg. If 2-3 applications were used, the residue levels on
rice harvested after 19-31 days were 1.3-1.8 mg/kg. The authors noted
the difficulty in meeting the requirement for a residue level of
0.1 mg/kg regardless of the pattern of application. In field studies
in Hu-bei Province in 1978 with the same application rate, the residue
levels in rice harvested after 25-42 days were 0.19-1.20 mg/kg. In
field studies in Zhe-jiang Province in 1978, residue levels in rice
when harvested after 30 days were 0.080-0.112 mg/kg, while residues in
rice harvested after 80 days were 0.039-0.100 mg/kg. In field studies
in Guang-dong Province in 1978, residues in rice harvested after 30
days were 0.042-0.149 mg/kg. In other field studies in the Guang-dong
Province in 1980, residue levels on rice harvested after 56 days were
0.185 mg/kg, but when the harvest was performed at 72 days, the
residue level was less than 0.10 mg/kg (Anonymous, 1981).
Huang et al. (1989) reported the residues of chlordimeform on
both rice and cotton plants in the Hu-bei Province of China between
1984 and 1988. With 1-3 applications to rice plants, followed by
harvest after 25-55 days, the chlordimeform residues were generally in
the range of 0.066-0.820 mg/kg for the rice, 7.70-22.30 mg/kg for the
husk, and 16.5-21.2 mg/kg for the stem. The authors noted that the
residue levels seldom met the 1975 JMPR recommended MRL of 0.10 mg/kg
for hulled rice (FAO/WHO, 1976). In further work on rice plants, it
was noted that the residue levels for late rice were generally higher
(approximately 2-fold) in late rice compared to early rice, and that
the residue levels in both the rice and the husk reduced by more than
90% when the time to harvest was increased from 26 to 72 days. With a
72-day harvest, the residue level in the rice was 0.065 mg/kg. The
residue levels in the stem (18-41 mg/kg), on the other hand, remained
relatively unchanged over the 72-day period. With 1-3 applications to
cotton plants, followed by harvest after 40 days, the chlordimeform
residues were 0.053-0.151 mg/kg in the kernel and 0.118 mg/kg in the
bracket.
Chlordimeform residues were also found in 8/15 honey samples
(Huang et al., 1989). The highest residue found was 32.2 mg/kg, and
the majority of the samples contained less than half this level. In
1994 the US FDA collected and analysed samples of honey imported from
the People's Republic of China. Of 60 samples analysed, 39 had
detectable residues, the highest being 0.058 mg/kg (Krick, 1994).
Moore (1971) summarized the results of residue trials on the use
of chlordimeform as an acaricide in cattle dips in Australia. The
residues were examined in cattle muscle, fat and liver as well as in
milk and butter from the first milking. Chlordimeform was used at
concentrations of 0.0125-0.1% in buffered cattle dips. Residues in
muscle, fat and liver did not increase greatly with increasing dose
of chlordimeform, and showed significant reductions between day 1
and day 3 post-treatment. The maximum residue levels found at day
3 post-treatment in muscle, fat and liver were 0.33, 0.51 and
0.69 mg/kg, respectively. At the first milking, the residues levels
showed a closer relationship with the concentration of chlordimeform
in the dip. The residue levels in milk and butter at a concentration
of 0.0125% were 0.01 and 0.30 mg/kg, respectively. The maximum
residue levels in milk and butter, which were found at the highest
concentration used (0.2 %), were 0.31 and 1.6 mg/kg, respectively.
In the study by Burkhard (1971), cows washed with a 0.5%
solution of chlordimeform to the hindquarters (3 treatments at 7-day
intervals), had total residue levels in milk, meat and fat below the
level of detection (0.03 mg/litre), except in milk on the day after
treatment when the levels rose to 1 mg/kg. In a further study by Voss
& Burkhard (1971), when cows were fed a concentrate containing
40-240 mg/kg chlordimeform for periods up to 42 days, the total
residues of chlordimeform and its metabolites in all milk, meat
and fat samples were below the limit of detection (0.03 mg/litre or
mg/kg). In liver and kidney samples, residues rose to a peak between
14 and 21 days (0.58 mg/kg in liver and 0.13 mg/kg in kidney), which
was followed by a slow decline.
In a study by Palmer et al. (1977), residues of chlordimeform
were determined in tissues and milk of cattle after spray application
to control cattle tick. In subcutaneous fat from animals sprayed with
0.45, 0.15 or 0.05% chlordimeform, the residue levels were 2.88, 0.46
and 0.15 mg/kg, respectively. The half-life of disappearance in all
cases was 2.46 days. Lower residue levels were found in six other
tissues, including kidney, muscle and liver. Residue levels in whole
milk of lactating cows at the three treatment levels were 1.42, 0.28
and 0.03 mg/litre, respectively. The half-life of disappearance from
milk was 0.45 days.
White Leghorn hens fed a laying mash containing chlordimeform at
levels of 0.25, 0.75 or 1.0 mg/kg were examined for residues in eggs
and tissues (breast, fat and liver) for periods of up to 28 days. No
residues were detected in breast meat. Residues were detected in fat
(0.22 mg/kg) at the 21 days only. Residues in the liver were highest
between 7 and 14 days (0.20 mg/kg) and reduced rapidly upon withdrawal
from the chlordimeform-containing feed. There were no detectable
residues of chlordimeform in eggs (FAO/WHO, 1972).
Residue trails on cotton were conducted between the years 1969
and 1978 (FAO/WHO, 1979). The application rates ranged from 0.125 to
3.6 kg/ha and resulted in mean residue levels of 0.1 to 13.1 mg/kg in
cottonseed when it was harvested immediately after application. The
final residue level was dependent on a number of factors including
application rate, number of applications, and length of waiting period
before harvest. The application rate had the largest influence.
5.2.3 Residues in processed food
Total residues of chlordimeform and its metabolites do not reduce
substantially during cooking processes, since while the proportion of
parent compound is reduced, there is an increase in the hydrolysis
product, N-formyl-4-chloro- o-toluidine. Residues of chlordimeform
itself in crops decrease through hydrolysis, but volatilization in
steam during cooking is not an important factor. The rate of
hydrolysis of chlordimeform is a function of pH and occurs much more
rapidly in weakly acid or neutral crops such as cauliflower (pH 6) or
green beans (pH 5) than in strongly acid crops such as apples (pH 2.5)
or tomatoes (pH 3). These results have been derived from studies in
different crops such as apples, grapes, tomatoes, cauliflower, beans
and sugar beet. These studies have also shown that residues of
chlordimeform and its metabolites are located in the outer parts of
crops, such as fruit peel. Excessive residues might therefore be
removed by peeling fruit (apples, citrus) or trimming the outer leaves
of leaf crops. In general, washing will remove only a small part of
the total residue (FAO/WHO, 1972).
Chlordimeform residues in whole apples reduced to approximately
40% of this level in pressed apple juice, while the level in the wet
pomace doubled (FAO/WHO, 1972) This is consistent with studies that
have shown that the residue level in the skin and outer layer is
approximately 50-fold higher than that found in the flesh (FAO/WHO,
1972).
Chlordimeform residues in tea leaves were found to be extractable
into tea prepared from these leaves to the extent of approximately 50%
of the total residues (Blass, 1972a).
Chlordimeform residues in grapes reduced to approximately 60% of
this level in grape juice (Blass, 1972b). This is consistent with
studies that have shown that the residue level in the grape skin was
between 60 and 76% of total residues (FAO/WHO, 1972). Fermentation
of the grape juice over a period of 72 days yielded a wine that
contained residue levels similar to those in grape juice (Blass,
1972c), indicating that the fermentation process does not
significantly lower the total chlordimeform residue level.
Chlordimeform residues in green hop cones, when used to prepare
beer, were found to be reduced to levels below the level of detection
(0.03 mg/kg) (Voss, 1971).
Residues associated with the processing of cottonseed have been
reported (FAO/WHO, 1979). Separation of the cottonseed oil leaves the
majority of the residues in the hulk and meal, although a significant
residue still remains in the crude oil. Additional refining processes
including bleaching, hydrogenating and deodorizing reduce the residue
level to below the level of detection. Cottonseed oil for human
consumption is subject to the bleaching and deodorizing processes and
thus residues of chlordimeform will be virtually zero.
5.3 Occupational exposure during manufacture, formulation or use
5.3.1 Exposure during manufacture and formulation
In the cases described by Folland et al. (1978) of
hospitalization of three factory workers in the USA who were exposed
to chlordimeform, the urinary levels of chlordimeform plus 4-chloro-
o-toluidine were 1.29, 6.32 and 4.85 mg/litre, respectively, three
days after exposure. This report is described in more detail in
section 8.2.2.
In a study on workers in the USA engaged in chlordimeform
production and packaging in 1976, urine was monitored in more than
100 workers. In more than 800 individual urine samples, total urinary
levels ranged from 0.05 to 50 mg/litre (personal communication by J.W.
Barnett, Ciba-Geigy Agricultural Division, Greenborough, North
Carolina, USA, to California Department of Food and Agriculture).
In China, there have been several studies in which the level of
exposure of workers to chlordimeform in chemical factories has been
examined together with a medical examination to detect any evidence of
toxicity in these workers. These are described in section 8.2.
In the study by Lu et al. (1981), the air concentrations
in 1974 in a chlordimeform-producing factory were generally below
0.036 mg/m3, with shorter periods at higher levels (0.108-
0.33 mg/m3), during specific tasks. Skin contamination on hands and
forearms was 9.1 mg/h for chemical operators and 964.2 mg/h for
packers. The urinary excretion levels for chlordimeform and
4-chloro- o-toluidine in controls were 0.015 and 0.042 mg/litre,
respectively, in chemical operators were 0.065 and 0.108 mg/litre,
respectively, and in packers were 0.263 and 0.398 mg/litre,
respectively.
In the study by Li et al. (1985b), 24 packers (9 male, 15 female)
in a chlordimeform manufacturing plant in Jiang-su Province of China,
were exposed to chlordimeform air concentrations (9 samples over 3
consecutive days) of 0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin
contamination of the hands and forearms was 110 �g/100 cm2
(S.D. 39 �g/100 cm2). Urinary chlordimeform levels were
0.20 � 0.13 mg/litre, and urinary 4-chloro- o-toluidine levels
were 0.48 � 0.29 mg/litre.
In a further study (Anonymous, 1985a) in a chlordimeform
manufacturing factory in China, packers had the highest urinary
chlordimeform and 4-chloro- o-toluidine levels at 0.39 mg/litre which
significantly correlated with skin contamination but not with air
concentration.
In the study by Tao et al. (1985), 61 employees (25 chemical
operators, 36 packers) of a pesticide factory in China were exposed to
air levels in the range 0.074 to 0.160 mg/m3. Skin contamination of
packers (2.99 mg/day) was higher than for chemical operators
(0.784 mg/day). The urinary excretion rate of chlordimeform and
4-chloro- o-toluidine in packers was also higher (0.513 mg/litre)
than for chemical operators (0.206 mg/litre) or controls
(0.055 mg/litre).
5.3.2 Exposure during use
In a company report by Kossmann (1980), summary data was provided
on the results of occupational exposure surveillance programmes on
agricultural workers associated with chlordimeform in nine countries.
Surveys of aerial pesticide applications to cotton entailed the
monitoring of about 600 airstrips in 1979 in the nine countries. Over
28 000 urine samples were analysed from workers in all phases of the
application situation. The urine was monitored and residue data
expressed as chlordimeform equivalents. In 1% of the assays,
substantial chlordimeform urinary residues indicated a significant
occupational exposure. Over 75% of the samples were at or below the
lowest level of analytical detection. This report states that, in
general, the conditions in two countries, the USA and Australia, were
indicative of favourable working conditions where only about 1% of the
samples contained a residue level indicating a higher-than-desired
level of exposure. In a subsequent report by Kenyon et al. (1993),
however, it is stated that at least 20% of the urine samples in
agricultural workers associated with chlordimeform in New South Wales,
Australia, exceeded the maximum permissible exposure level for
chlordimeform equivalents in urine, which was set at 0.2 mg/litre.
Operators who exceeded this level were required to be withdrawn from
the site until the urinary level fell below 0.1 mg/litre. The mean
sample assays for both ground rig operators and workers involved in
aerial application exceeded the set level in 1984-1985. Furthermore, a
number of workers experienced exposures that exceeded the limit on
multiple occasions. The urine monitoring programme in operation in New
South Wales, Australia, also grossly underestimated the worker
exposure levels since its protocol did not allow urine sample
collection in the first 24 h following potential exposure (Kenyon et
al., 1993). In the report by Kossman (1980), it is stated that working
conditions in some other countries (i.e., Colombia, El Salvador,
Guatemala and Honduras) were less favourable and thus exposure was
higher. However, in some areas where flagmen were unavoidably exposed,
the urinary residue levels were low, indicating that with precautions
exposure can be controlled. In New South Wales and Israel, urine
monitoring for agricultural workers was mandatory, while in the USA,
urine monitoring was conducted on a voluntary basis.
In a report by Henderson (1985), monitoring studies on operator
exposure during the 1984-1985 cotton season in NSW, Australia, were
summarized. Urine samples were examined in operators involved in
application of chlordimeform by both ground-rig (Strong & Bull, 1985a)
and aerial (Strong & Bull, 1985b) methods. Chlordimeform application
by ground-rig to 26 444 hectares involved 48 people. A total of 85
urine samples were examined; in 78.8% of samples the chlordimeform
level was below 0.20 mg/litre, and in 90.5% of samples it was below
0.50 mg/litre. The mean sample assay was 0.21 mg/litre. Chlordimeform
application by aerial spraying to 315 694 hectares involved
222 people. A total of 919 urine samples were examined and in 80.3% of
samples, the chlordimeform level was below 0.20 mg/litre, and in 89.8%
of samples was below 0.50 mg/litre. The mean sample assay was
0.24 mg/litre.
The exposure data for chlordimeform used on cotton in seven
countries (Australia, Columbia, El Salvador, Guatemala, Mexico,
Nicaragua, USA) for the period 1980-1984 has been compiled in a
company report by Limmer (1985). Urine samples indicated that in all
countries, the chlordimeform level was less than 0.3 mg/litre for
between 70 and 92% of the exposed workers, and was >5 mg/litre in
less than 2% of workers. The highest levels were recorded in the
loaders, washers and mechanics, while the lower levels were found in
the pilots and flagmen.
In a study by Jiang et al. (1985), exposure of workers engaged
in spraying chlordimeform with fine mist sprayers in both rice fields
and cotton fields was examined. The air concentration of chlordimeform
surrounding the workers during spraying was 0.80 mg/m3. Skin
contamination from spraying in a rice field was 0.777 mg/100 cm2/h
(16 samples), and from spraying in a cotton field was 0.445 mg/100
cm2/h for one group (40 samples) and 1.216 mg/100cm2/h for a
second group (40 samples). Urinary excretion of chlordimeform and
4-chloro- o-toluidine together was 0.756 mg/litre for rice workers,
and 0.490, 0.465 and 1.125 mg/litre in three separate groups (40 each)
for cotton workers. Good correlation was noted between skin
contamination and urinary excretion. It was noted that contamination
of the lower extremities of the body was significantly different
between workers with protection (0.490 mg/100 cm2 per h) and those
without (1.179 mg/100 cm2 per h).
In a study by Ling et al. (1986) and Zhang et al. (1986a),
excretion of chlordimeform and 4-chloro- o-toluidine was examined as
a measure of occupational exposure. Chlordimeform applicators (7 male,
6 female; 20-41 years) were examined during spraying of cotton for
three consecutive days for 4.7, 3.0 and 4.4 h respectively in July
1985. Protective measures included gauze mask, plastic gloves and
plastic apron, although it was noted that extensive contamination
occurred. Air levels in the breathing zone on each of the three days
were 0.011, 0.014, 0.011 mg/m3, respectively. Skin contamination on
each of the three days was estimated by the method of Zhang et al.
(1986b) to be 10.99, 4.32, and 4.45 mg/day, respectively. Urinary
chlordimeform and 4-chloro- o-toluidine together were measured over
the 3 days of exposure and for 7 days after cessation of exposure.
Urinary levels ranged from a peak of 2.408 mg/litre during exposure to
0.036 mg /litre after 7 days. Excretion of chlordimeform occurred very
rapidly and the highest level was detected in the sample collected at
the end of each shift. There was a close correlation between skin
contamination and urinary excretion. Metabolism occurred very rapidly
since 4-chloro- o-toluidine usually accounted for 70-93 % of the
total amount in the urine. The authors concluded that the level of
urinary chlordimeform plus 4-chloro- o-toluidine is an accurate index
of chlordimeform exposure.
Maddy et al. (1986) reported the results between 1982 and 1985 of
a programme of monitoring the urine of more than 200 workers who had
received training in the use of chlordimeform on cotton in California.
Protective clothing was required for all employees who handled
containers, prepared mixtures, loaded application vehicles, applied
chemical, flagged or did repair work on equipment exposed to
chlordimeform. This included cloth overalls, washable hat, waterproof
boots, waterproof gloves, and a full-face shield. Chlordimeform was
detectable in urine as early as 4 h after dermal exposure, but did not
increase during the work season. The chlordimeform concentrations
averaged about 90 �g/litre, with the highest levels found in
mixer-loaders and somewhat less in equipment washers, and the lowest
levels in pilots and flaggers. Urinary levels in the 8-10 h following
a work shift gave a good indication of exposure for the shift just
completed.
Kurtz et al. (1987) reported the results of a monitoring
programme of agricultural workers exposed to chlordimeform when used
on cotton in Imperial Valley, California, during the 1982 season. More
than 1000 urine samples were taken from 132 workers, including pilots,
mixers/loaders, flaggers and equipment maintenance workers.
Chlordimeform metabolites were detected in all workers at some time
during the study despite the use of protective clothing. The level of
urinary metabolites was positively correlated with the length of
exposure and the nature of job activity as shown in Table 3.
Mixer/loaders and maintenance workers had the highest levels.
Metabolites appeared in urine within 4 h and approximately 75% of
urinary excretion occurred within the first 24 h.
Table 3. Chlordimeform metabolite concentrations in urine (mg/litre)
of agricultural workers during an 11-week application period
(Kurtz et al., 1987)
Work group Immediately post-work Following morning
No. Mean SD No. Mean SD
All groups 535 0.12 0.41 572 0.10 0.23
Pilots 145 0.08 0.10 163 0.08 0.10
Mixers/Loaders 156 0.19a 0.71 162 0.15b 0.36
Flaggers 202 0.07 0.08 213 0.07 0.09
Others 32 0.25 0.45 34 0.21c 0.36
a Significantly greater versus flagger group (P<0.01)
b Significantly greater versus pilots (P<0.01) and flaggers
(P<0.001)
c Significantly greater versus pilots (P<0.001) and flaggers
(P<0.001)
Lemesch et al. (1987) provided the results of monitoring for
chlordimeform exposure in agricultural workers in Israel during
1980-1985. Chlordimeform was used only on cotton by aerial application
and all workers were monitored for urinary chlordimeform and its
metabolites on a weekly basis. The results indicated 86.8% of the
urine samples contained less 0.05 mg/litre, and 1.4% contained more
than 0.30 mg/litre. Overall, the loaders had the highest exposure
followed by the mechanic and then the pilots (see Table 4).
Table 4. Chlordimeform metabolite concentrations in urine (mg/litre)
of agricultural workers in Israel during 1980-1985
according to occupation (Lemesch et al., 1987)
Occupation < 0.05 0.05 - 0.30 > 0.30 Total
No. % No. % No. %
Loaders 666 79.0 157 18.6 20 2.4 843
Mechanics 383 94.8 19 4.7 2 0.5 404
Pilots 287 98.2 5 1.7 - - 292
Total 1336 86.8 181 11.8 22 1.4 1539
Balu (1989) has provided the results of monitoring field worker
exposure to chlordimeform from aerial application on cotton. During
the years 1978-1984, urine samples using a grab sample technique from
approximately 4600 field workers were examined. For mixer/loaders,
between 0.5 and 1.9% had levels >5 mg/litre, and between 2.1 and 18%
had levels of 1.0-5.0 mg/litre. The majority (46-78%) had levels in
the range <0.05-0.10 mg/litre. There was no apparent change in the
proportion of workers in the various exposure levels over the course
of the study. For the pilots, between 0.3 and 0.7% had levels
>5.0 mg/litre, while 63-90% had levels between <0.05 and
0.10 mg/litre.
The clinical signs associated with chlordimeform exposure in
these studies are described in section 8.2.2.
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption, distribution and excretion
6.1.1 Mouse and rat
The earliest investigations on the kinetics and distribution of
chlordimeform were performed in a series of studies on rats (FAO/WHO,
1972). Four male and four female rats were treated orally with 270 �g
[3H-phenyl]-chlordimeform. Over a 24 h period, 52.8% (range
41.8-59.6%) of the radioactivity was eliminated in urine and 2.5%
(range 0.13-5.30%) in faeces, while 19-23% of the dose was excreted
into the bile. Following intravenous injection of 270 �g [3H-phenyl]-
chlordimeform in rat, elimination of radioactivity over 24 h consisted
of 53.7% (range 52.0-55.6%) in urine and 1.42% (range 1.19-1.84%) in
faeces.
Oral dosing of male rats with 270 �g [3H-phenyl]-chlordimeform
resulted in residues in liver (0.78 mg/kg), kidney (0.59 mg/kg)
and lymph nodes (0.35 mg/kg) after 8 h. After 24 h, residues in
gastrointestinal tract (and contents) and liver were 0.95 and
0.35 mg/kg, respectively. All other tissues contained residue levels
of <0.16 mg/kg at 8 h, and <0.27 mg/kg at 24 h (FAO/WHO, 1972).
Oral dosing of male rats with 270 �g [3H-phenyl]-chlordimeform
for seven consecutive days resulted in excretion of 59% of the
administered label in urine and 10% in faeces during the dosing
period. Tissue residues at the termination of dosing were less than
0.03% of the administered dose (FAO/WHO, 1972).
Knowles & Sen Gupta (1970) further studied the toxicokinetics
in rats. A group of two male and two female rats was given
[14C-tolyl]-chlordimeform (3 �Ci) orally (dose unspecified). Over a
72-h period, 88% of the administered radioactivity was eliminated in
the urine, with the highest concentration occurring at 12 h, and 7.5%
was eliminated in the faeces. At sacrifice (72 h), tissue levels based
upon [14C]-label levels were 0.21 mg/kg in liver, 0.15 mg/kg in
muscle, 0.11 mg/kg in fat and less than 0.1 mg/kg in other tissues. As
part of the same study (Knowles & Sen Gupta, 1970), a similar group of
male and female rats received an oral dose of [14C-methyl]-4-chloro-
o-toluidine. Tissue levels based upon [14C]-label levels at 72 h
after dosing were 0.33 mg/kg in fat, 0.26 mg/kg in liver, 0.2 mg/kg in
kidney and oviduct, 0.1 mg/kg in brain, and less than 0.1 mg/kg in
other tissues.
In a more recent study by Watanabe & Matsumura (1987) concerning
the comparative metabolism of chlordimeform and sulfamidine in rats,
it was found that after administration of [14C]-chlordimeform as a
single oral dose (130 mg/kg), radioactivity was eliminated in the
urine (87%) and faeces (8%) within 3 days. Most of the radioactivity
was excreted within 2 days. After 5 daily doses of [14C]-
chlordimeform (26 mg/kg), 78% of the radioactivity was excreted in the
urine and 15% in the faeces. After 10 days, the residue level in all
tissues, except blood and liver, was below 1 mg/kg.
In a study by Ifflaender (1977a), groups of mice (8/sex; strain
Tif:MAG f) and rats (3/sex; strain TIF:RAI f) were administered
[14C- ring]-chlordimeform orally at a dose of 25 mg/kg body weight.
The general excretion pattern was similar for both mice and rats with
more than 70% of the [14C]-label being excreted within 24 h. Of the
excreted dose, 80-95% was excreted through the urine, while 10-15% was
excreted through the faeces. After 144 h, 95-113% of the administered
dose was recovered. Over the period of the experiment (144 h), the
levels of radioactivity in the urine were found to range from 82-97%
of the administered dose. Residues of chlordimeform were found in
liver, kidney and blood, with the highest level found to be 1 mg/kg.
Slightly higher residue levels were found to be present in females
than in males. In a subsequent study by Ifflaender (1977b) to
determine the quantitative differences between mice and rats, animals
were administered 25 mg/kg body weight [14C- ring]-chlordimeform.
Rapid urinary excretion of chlordimeform was again observed in both
mice (85%) and rats (75%) within 24 h.
In a more detailed toxicokinetic study by Kopp et al. (1977),
chlordimeform was administered orally to female mice at two dose
levels (1.2 or 120 mg/kg body weight) using either a single acute or
multiple daily administration for up to 21 days. The results again
indicated rapid excretion of chlordimeform and/or its metabolites
through the urine and did not provide any indication of
bioaccumulation at either dose level. At the high dose level, a
slightly reduced 24-h excretion pattern of the radioactivity was
observed following a single administration. This pattern returned to
normal within two to three doses in the multiple dosing regime. The
percentage of excretion was the same after a period of 21 days,
irrespective of the dose level. The authors concluded that
chlordimeform excretion was largely complete within 24 h of
discontinuation of administration. No accumulation of residues was
evident.
Knowles & Benezet (1977) studied the kinetics of chlordimeform in
mice following intraperitoneal injection of 0.6 �Ci [14C-tolyl]-
chlordimeform. Over the 96-h period, 95.5% of the administered dose
was eliminated, with 42.5% in the urine and 53% in the faeces. In the
first 3 h, 43.7% was eliminated, with 27.3% in urine and 16.4% in
faeces.
In a study in mice by Crowder & Whitson (1980), the excretion and
retention of [14C]-chlordimeform in mice was found not to be affected
by oral co-administration of toxaphene or methyl parathion. Low
residue levels of chlordimeform were evident at 196 h in all tissues
following oral administration.
6.1.2 Other species
In a study by Sen Gupta & Knowles (1970), two female dogs (18 and
20 kg) were given 10 �Ci [14C]-chlordimeform as a single oral dose
(0.3 mg/kg), and one male dog (12 kg), which had undergone cannulation
of the gall-bladder and ligation of the bile duct, was given 20 �Ci
[14C]-chlordimeform orally. Urine was collected (by catheterization)
at 1, 3, 6, 12, 24, 48 and 72 h. Faeces were collected at similar time
intervals. Of the administered [14C] label, 85% was recovered in
urine, 0.6% in faeces, and 5% in the bile within 72 h. In the same
study, two brush goats, a male (36 kg) and a lactating female (39 kg)
were administered 10 �Ci [14C]-chlordimeform orally. The male goat
eliminated 87% of the administered dose through the urine within 48 h,
while the lactating female eliminated only 67% during the same period.
Only about 0.3% of the applied dose was eliminated in the milk within
96 h.
In a review by Knowles (1970), the metabolites found in three
species, namely, rat, dog and goat, were compared. In all three
species, oral treatment with radioactive chlordimeform resulted
primarily in elimination through the urine. Cumulative percentages of
the dose excreted in the urine 24 h after treatment were 85% for rats,
70% and 80% for the two dogs, 65% for a lactating goat, and 80% for a
male goat. Rats eliminated 7.5% of the dose in the faeces by 72 h, and
only 0.6% and 1.8% of the administered radioactivity was accounted for
in dog and goat faeces, respectively. The rate of degradation of
chlordimeform was also different in the three species. By 24 h after
treatment, 25% of the radioactive material in rat urine was
organosoluble and partitioned into chloroform, but in the dog and goat
urine less than 10% was organosoluble. Levels of chlordimeform
expressed as percentages of organosoluble urinary radioactivity at 24
and 72 h post-treatment were 9.9 and 2.1% for the rat, 1.3 and 0.2%
for the dog, and 0.1 and <0.1% for the goat.
6.1.3 Human
In a study by Nixon & Neal (1983), the excretion of chlordimeform
residues was examined in eight volunteers following dermal
application. A dose of 24.75 mg chlordimeform was applied to the
forearm via a patch which was removed after 4 h and the application
site washed in propanol followed by detergent. The average absorbed
dose was calculated to be 7.95 mg. Urine was collected for 72 h
following treatment. During this period, an average of 38.3% of the
absorbed dose was recovered from the urine. The half-life for
excretion was between 5.9 and 12.1 h, with an average of 8.8 h.
A number of studies have been conducted that monitored the urine
of workers exposed to chlordimeform during use (see section 5.3.2).
The data indicate rapid metabolism of chlordimeform to 4-chloro- o-
toluidine, followed by urinary excretion. Detection in the urine was
as early as 4 h after exposure, and approximately 75% was excreted
within 24 h.
6.2 Metabolic transformation
6.2.1 Mouse and rat
In an early study (FAO/WHO, 1972), the urine from a male rat
collected over 72 h subsequent to oral administration of 1.1 mg
[3H-phenyl]-chlordimeform contained free extractables representing
22% of the [3H] label, of which 10% was in the water phase and 17%
was extractable glucuronides. The free extractable [3H]-label
comprised chlordimeform, 4-chloro- o-toluidine, N-formyl-4-chloro-
o-toluidine, and N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethylchlordimeform). Glucuronides were based on the same compounds
found as free extractables.
In a study by Knowles & Sen Gupta (1970), pairs of male and
female rats were treated orally with 3 �Ci [14C-tolyl]-chlordimeform.
Urine and faeces were collected at 3, 12, 24, 48 and 72 h after
dosing. Urinary and faecal elimination of [14C] label after 72 h
comprised 88% and 7.5% of the administered dose of [14C]-
chlordimeform, and 71 and 24.5% of the administered [14C]-4-
chloro- o-toluidine. Chloroform extraction removed 30% of the
radioactivity from the urine of [14C]-chlordimeform-treated rats, the
extract containing chlordimeform, N'-(4-chloro- o-tolyl)-
N-methylformamidine (demethylchlordimeform), N-formyl-4-chloro-
o-toluidine, and 4-chloro- o-toluidine, in addition to three
unidentified metabolites. A considerable amount of radioactivity
remained at the point of origin of the chromatograph, with the amount
remaining increasing with time, (30% at 3 h and 75% at 72 h). At 3 h,
the four identified compounds were present in approximately equal
amounts. By 12 h, the level of N'-(4-chloro- o-tolyl)- N-
methylform-amidine had decreased to approximately 25% of the level of
any of the other three compounds. By 48 h, chlordimeform levels were
half those of the other two compounds, and, by 72 h, N-formyl-
4-chloro- o-toluidine was present in the greatest proportion.
As part of the same study (Knowles & Sen Gupta, 1970), a similar
group of male and female rats received an oral dose of [14C-methyl]-
4-chloro- o-toluidine. The metabolites found in ethyl acetate-
extracted urine comprised 5-chloroanthranilic acid, and N-formyl-
5-chloroanthranilic acid increased. The level of 5-chloroanthranilic
acid remained constant. A large amount (20-50%) of the radioactivity
remained at the origin of the chromatograph. Five unidentified
compounds were noted.
The metabolic transformation of the metabolite
demethyl-chlordimeform ( N'-(4-chloro- o-tolyl)- N-
methylformamidine) in the rat was investigated by Benezet & Knowles
(1976). Eight Sprague-Dawley rats were each administered 1.5 �Ci
[14C]-demethylchlordimeform by oral intubation. Urine and faeces were
analysed over a 72-h period. The majority of the radioactivity was
eliminated through the faeces (64%) but significant amounts were also
eliminated in the urine (35%). The peak level of radioactivity
occurred in the urine between 12 and 24 h, and in the faeces between
18 and 48 h. Of the urinary radioactivity, 16-26% could be extracted
with ethyl acetate. Compounds present included demethylchlordimeform,
N'-(4-chloro- o-tolyl)formamidine, N-formyl-4-chloro- o-
toluidine, 4-chloro- o-toluidine and several unidentified compounds.
The aqueous fraction remaining after ethyl acetate extraction (74-85%
of the total radioactivity) was largely acid-labile and probably
consisted of conjugates, possible glucuronides and ethereal sulfates.
Approximately 25% of the total radioactivity of the faeces was
extractable with ethyl acetate, and similar metabolites were present.
Ifflaender (1977b) examined the quantitative differences in
urinary metabolites between mice and rats following oral
administration of [14C]-chlordimeform at a dose level of 25 mg/kg
body weight. Little quantitative difference in individual
metabolites was observed between the species. Of the total
metabolites, N'-(4-chloro- o-tolyl)- N-methyl formamidine
(demethylchlordimeform) represented 11.3% in rats and 2.4% in mice,
while 4-chloro-2-methyl-phenylurea represented 6.3% in rats and 1.2%
in mice. Sulfuric acid conjugates represented 20.8% in mice compared
to 14.0% in rats. Glucuronic acid conjugates (representing 28% of
metabolites) and all other minor metabolites were in similar amounts
in the urine of rats and mice. Acid hydrolysis of the urine released
degradation products in similar amounts in the urine of rats and mice.
Knowles & Benezet (1977) reassessed the metabolism of
chlordimeform in rat and also assessed the metabolism in mice. Ten
male rats were treated orally with 2 �Ci [14C]-chlordimeform and
urine samples collected at 12 and 24 h. Twelve male mice were injected
intraperitoneally with 0.6 �Ci [14C]-chlordimeform, and urine and
faeces samples were collected at various times up to 96 h. In rat
urine, the major organosoluble metabolites (>10%) included
3-(4-chloro- o-tolyl)urea, N-formyl-4-chloro- o-toluidine,
4-chloro- o-toluidine, and N-formyl-5-chloroanthranilic.
Demethylchlordimeform, didemethylchlordimeform, 1,1-dimethyl-3-
(4-chloro- o-tolyl)urea and 5-chloroanthranilic acid were minor
metabolites. In mouse urine, the majority of the radioactive material
was water soluble, probably consisting mainly of conjugates such as
glucuronides and ethereal sulfates (based on analogy with metabolism
in rats). The major organosoluble metabolites (>10%) were
N-formyl-4-chloro- o-toluidine, 4-chloro- o-toluidine and
N-formyl-5-chloroanthranilic acid. The minor metabolites identified
in rat urine were also present in mouse urine. The identity of the
major metabolites in rat urine were confirmed in the study of Watanabe
& Matsumura (1987).
Knowles & Benezet (1977) proposed the metabolic pathway for
chlordimeform metabolism in rats and mice shown in Fig. 1.
6.2.2 Other species
In the study of Sen Gupta & Knowles (1970) in dogs described in
section 6.1.2, chloroform extraction of the urine removed 10% of the
radioactivity. Thin-layer chromatography of the extract revealed
chlordimeform, N'-(4-chloro- o-tolyl)- N-methylformamidine
(demethyl-chlordimeform) and 4-chloro- o-toluidine in about equal
quantities, but about four times as much N-formyl-4-chloro- o-
toluidine at 1 h after treatment. The level of unchanged chlordimeform
and N'-(4-chloro- o-tolyl)- N-methylformamidine decreased steadily
with time, whereas 4-chloro- o-toluidine and N-formyl-4-chloro- o-
toluidine rose to maximum levels between 6 and 12 h prior to tapering
off. Three unidentified metabolites were present. In addition, a lot
of the radioactivity remained at the origin of the chromatograph.
Re-runs of this material in polar solvents showed 5-chloroanthranilic
acid, N-formyl-5-chloroanthranilic acid and three unidentified
compounds were present. Some radioactivity still remained at the
origin. The urinary [14C] label not extracted by chloroform was
treated with enzymes (�-glucuronidase, �-glucu-ronidase-aryl
sulfatase) to form "aglycones". About 75% of the remaining [14C]
label was extracted in this manner (hydrochloric acid released 62%),
and thin-layer chromatography showed the same compounds as found in
the chloroform extract, the major metabolite being N-formyl-
4-chloro- o-toluidine. In addition, more of one of the unidentified
metabolites was present. Again re-chromatography of the 45% of the
radioactivity remaining at the origin with more polar solvents
revealed 5-chloroanthranilic acid to be the major product. In the
bile, peak concentration of radioactivity occurred at 8 h. About 10%
of this activity could be partitioned into ether, and thin-layer
chromato-graphy of the extract indicated the same four compounds seen
in urine chloroform extract. N'-(4-chloro- o-tolyl)- N-
methylformamidine (demethylchlordimeform), N-formyl-4-chloro-
o-toluidine and an unidentified compound accounted for most of the
activity at 2 h. By 6 h, 75% of the activity was due to N-formyl-4-
chloro- o-toluidine. Incubation of extracted bile with enzymes or
acid gave the same "aglycone" compounds as found in urine. Tissue
residues of [14C] label at 72 h ranged from 72 �g/kg in liver to
30 �g/kg (kidney), 13.5 �g/kg (lung), 11.9 �g/kg (spleen and brain)
and 5 �g/kg (heart and fat and pancreas).
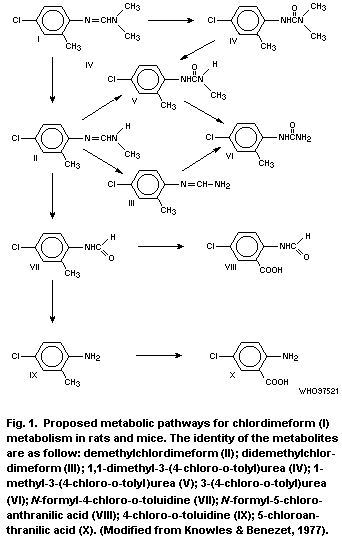 In the same study, metabolites extracted from goat urine were
analysed by thin-layer chromatography. The major urinary metabolite
was N-formyl-4-chloro- o-toluidine. The metabolites in goat urine
showed a similar pattern to those in rats, with a similar proportion
of conjugated material.
The comparative metabolic fate of chlordimeform in rats, goats
and dogs is considered in a review by Knowles (1970), which emphasizes
the similarity between these species.
6.2.3 In vitro studies
Initial studies on the in vitro metabolism of chlordimeform
were conducted with [3H-phenyl]-chlordimeform (FAO/WHO, 1972).
Incubation of [3H-phenyl]-chlordimeform for 120 min with rat liver
homogenate resulted in 24% unchanged chlordimeform, 45% 4-chloro- o-
toluidine, and 11% unidentified metabolites being formed. Rabbit liver
homogenate yielded 53, 40 and 7% of the same metabolites,
respectively. Incubation of 60 �g [3H-phenyl]-chlordimeform (30 �Ci)
with 5 ml human plasma yielded N-formyl-4-chloro- o-toluidine only.
Conversion was 25% in 5 h, and 50% in 20 h. Rose (1969a) confirmed the
rat liver homogenate studies using [14C]-chlordimeform. Three
unidentified metabolites were also observed and, in addition,
chlordimeform degradation was shown to require the presence of
nicotinamide. Spleen homogenates were inactive with regard to
chlordimeform degradation.
The metabolism of chlordimeform in vitro was first reported by
Ahmad & Knowles (1971). Incubation of [14C]-chlordimeform with
various rat liver enzyme preparations identified demethylchlordimeform
as the major metabolite, which was formed by microsomal
N-demethylase in the presence of exogenous nicotinamide. This
reaction was inhibited by mixed function oxidase inhibitor, SKF-525A.
The chlordimeform metabolites formed in vitro were qualitatively
similar to those detected in urine from chlordimeform-treated mammals.
This has been confirmed by others (Hill et al., 1979; Ghali
& Hollingworth, 1985; Kimmel et al., 1986; Watanabe & Matsumura,
1987).
Knowles & Benezet (1977) confirmed that the major in vitro
metabolite was demethylchlordimeform, but also found that
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
present in appreciable amounts.
Ahmad & Knowles (1971) also investigated the metabolism of
[14C]- N-formyl-4-chloro- o-toluidine) in the presence of rat liver
enzyme preparations. Eighty percent of this metabolite was metabolized
by an enzyme, probably a hydrolase, in the soluble fraction, with
major metabolites being 4-chloro- o-toluidine (52%) and an unknown
substance (26%).
The question of the possible formation of azo-derivatives in
animal tissues was investigated by Rose (1969a). A number of
experiments were conducted to investigate the presence or absence of
azobenzene formation from chlordimeform or 4-chloro- o-toluidine. In
the first experiment, it was demonstrated that peroxidase activity was
negligible in rat liver and spleen. Furthermore, catalase, which was
abundant in the same tissues, and which, like peroxidase, catalyses
reactions between hydroxyperoxides and many oxidizable compounds, was
shown to be unable to form symmetrical azo-derivatives from
4-chloro- o-toluidine. In the second experiment, it was demonstrated
that rat liver and spleen homogenates, which were fortified with
nicotinamide, and which degrade chlordimeform to demethylchlordimeform
and small quantities of N-formyl-4-chloro- o-toluidine and
4-chloro- o-toluidine, respectively, did not form any azobenzene
derivatives. These compounds therefore do not represent metabolites of
chlordimeform or its aromatic amine degradation products in animal
tissues.
Lin et al. (1975) have investigated the metabolism of
chlordimeform in primary embryonic lung cell cultures. In 2 h of
incubation, 97% of chlordimeform was metabolized to N-formyl-4-
chloro- o-toluidine (81.9%) and 4-chloro- o-toluidine (2.3%). The
route of metabolism, which was different to that seen in mammals,
appeared to be first demethylation followed by cleavage at the
carbon-nitrogen double bond to form N-formyl-4-chloro- o-toluidine.
The formation of the demethylchlordimeform was minute compared to that
of the N-formyl derivative. The minor metabolites observed were
demethylchlordimeform and two unknown metabolites. When incubated in
culture media without cells, chlordimeform decomposed to
N-formyl-4-chloro- o-toluidine.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 Oral
The acute oral toxicity data for chlordimeform is presented in
Table 5 and for chlordimeform hydrochloride in Table 6.
The general signs of toxicity in rats are hyperactivity,
dyspnoea, muscular weakness, tremors, "Straub's tail", spasms,
convulsions and respiratory arrest. No pathological changes were noted
in the rat following oral treatment.
In mice, signs of toxicity were similar, but some differences
were noted. Symptoms included restlessness, hyperreflexia and tremors,
particularly of the head and forelimbs, that developed to one or more
episodes of clonic convulsions. Death usually occurred within one hour
during one of the convulsive episodes. If the animal survives this
hyperexcitation and tremor, it becomes sedated, locomotion is
suppressed, and it usually recovers within 24 h.
The acute oral toxicity data for metabolites of chlordimeform is
presented in Table 7.
7.1.2 Other routes
The acute dermal toxicity data for chlordimeform in rats is
presented in Table 5 and for chlordimeform hydrochloride in Table 6.
The base, but not the hydrochloride, is readily absorbed by the skin
(Knowles, 1991). The general signs of toxicity are dyspnoea,
exophthalmos, prostration, spasms and convulsions. Pathological
changes in the rat included pale or blotchy liver, pale kidneys, and
haemorrhagic intestinal contents. No local skin irritation occurred.
In the dog, a lethal intravenous dose of chlordimeform (50 mg/kg
body weight) caused rapid and irreversible hypotension, and
respiratory arrest followed cardiovascular collapse within a few
seconds. Artificial respiration did not protect the animals against
hypotension and death, suggesting cardiovascular collapse is probably
the primary cause of death in dogs. Pathological examination following
oral administration in dogs revealed congestion of liver, kidneys and
lungs.
The acute inhalation LC50 of chlordimeform base in rats
(male and female) was 17 400 mg/m3 and for chlordimeform HCl was
>5800 mg/m3 (FAO/WHO, 1972).
The acute dermal toxicity data for metabolites of chlordimeform
is presented in Table 7.
Table 5. Acute toxicity of chlordimeform in experimental animals
Species Sex Route LD50 References
(mg/kg
body weight)
Rat male/female oral 250 FAO/WHO (1972)
male/female oral 340 Worthing & Walker (1983)
male/female oral 123 Robinson et al. (1975)
male/female oral 301 Gaines & Linder (1986)
male/female oral 178-220 FAO/WHO (1972)
male/female oral 178 FAO/WHO (1972)
female oral 170-460 FAO/WHO (1972)
female oral 265 Gaines & Linder (1986)
female oral 460 FAO/WHO (1972)
male/female dermal 640 FAO/WHO (1972)
male dermal 337 Gaines & Linder (1986)
female dermal 263 Gaines & Linder (1986)
Mouse male/female oral 290 Haddow & Shankland (1969)
male oral 267 Ghali & Hollingworth (1985)
male/female ip 110 FAO/WHO (1972)
Rabbit - oral 625 FAO/WHO (1972)
- oral 625 Worthing & Walker (1983)
Dog male oral approx.150 Hurni & Sachsse (1969)
female oral approx.100 Hurni & Sachsse (1969)
Table 6. Acute toxicity of chlordimeform hydrochloride in
experimental animals (FAO/WHO, 1972)
Species Sex Route LD50
(mg/kg body weight)
Rat male oral 305
male oral 325
female oral 330
male iv 95
- dermal approx. 4000
Mouse male/female oral 220
Rabbit - dermal >4000
Table 7. Acute toxicity of chlordimeform metabolites in the rat (FAO/WHO, 1972)
Metabolite Sex Route LD50
(mg/kg
body weight)
N-formyl-4-chloro-o-toluidine male/female oral 2900
4-chloro-o-toluidine (base) male/female oral approx.1000
4-chloro-o-toluidine-HCl male/female oral 860
N-formyl-4-chloro-o-toluidine male/female dermal (24 h) >2150
4-chloro-o-toluidine (base) male/female dermal (24 h) approx.1800
4-chloro-o-toluidine-HCl male/female dermal (24 h) >2150
7.2 Short-term exposure
7.2.1 Dietary
Dietary studies of 60 days duration have been conducted in the
mouse and rat with each of chlordimeform, N-formyl-4-chloro -o-
toluidine, and 4-chloro- o-toluidine.
7.2.1.1 Mouse
In a study with chlordimeform by Sachsse et al. (1979a), groups
of Tif:MAGf mice (30/sex/group), housed under SPF conditions, were fed
a diet containing chlordimeform at concentrations of 0, 750, 1500,
3000 or 6000 mg/kg for 60 days. This corresponded to dietary intakes
of 0, 107, 194, 717 or 1525 mg/kg body weight per day for females and
0, 110, 200, 669 or 1519 mg/kg body weight per day for males. At the
end of the 60-day period, all animals were examined for haematology,
blood chemistry and urinalysis parameters, and groups of 10 male and
10 female animals from the control and the lower three dose groups
were subjected to gross and microscopic examination of tissues and
organs. Mortality was observed in the two highest dose groups over the
course of the study. The highest dose group was terminated after two
weeks because of a poor general condition of the animals. Growth, as
shown by body weight gain, was reduced in all dietary groups. Food
consumption was reduced at all dietary levels in females only. No
clinical signs of toxicity were noted. Ophthalmological and auditory
examinations were normal. Haematological investigations showed
haemolytic anaemia in both sexes of all treated groups, which was
characterized as a reduction in haemoglobin concentration, red blood
cell count, and packed cell volume. The anaemia was associated in a
dose-related manner with an increased methaemoglobin concentration and
an increase in Heinz body formation. At 3000 mg/kg diet, there was a
slight reticulocytosis noted in both sexes. This was accompanied in
females by a shift in the differential leucocyte count noted as an
increase in the percentage of polymorphonuclear neutrophile and a
decrease in the percentage of lymphocytes. Small changes were observed
in alkaline phosphatase activity, which was slightly increased in male
mice at the highest dose level. Total protein concentration was also
slightly reduced in female mice at the highest dose level. Urinalysis
was unremarkable. In the animals that died or were killed within the
first 2-week period, all were found to be emaciated and in poor
general condition. In all treated animals dying during the test
period, congestion of the organs, especially of the liver, was
observed. At the highest dose level, atrophy of thymic tissue was
observed. There was an increased haemosiderosis at the two highest
dose levels. There were no other pathological findings associated with
the presence of chlordimeform in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980a), groups of Tif:MAGf mice (30/sex/group), housed under SPF
conditions, were fed a diet containing N-formyl-4-chloro- o-
toluidine at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg for
60 days. This corresponded to dietary intakes of 0, 138, 379, 1203,
or 3153 mg/kg body weight per day for females and 0, 140, 349, 1023,
2549 mg/kg body weight per day for males. At the end of the 60-day
period, all animals were examined for haematology, clinical chemistry
and urinalysis parameters. A group of 10 males and 10 females from
each dose level was examined for gross and microscopic pathological
changes at the conclusion of the study. Mortality was observed
predominantly at the high-dose level over the course of the study.
There were no clinical signs of toxicity, although food consumption
and growth were depressed at 1500 mg/kg and above in both sexes over
the course of the study. Ophthalmological and auditory examinations
were unremarkable. Significant haematological abnormalities were
observed at all dose levels at the conclusion of the study. Haemolytic
anaemia was observed in both males and females and was characterized
as a reduction in haemoglobin concentration, erythrocyte count
and packed cell volume. There was a dose-related increase in
methaemoglobin concentration and an increase in Heinz body formation.
Additionally, both males and females in all treated groups showed a
significant reticulocytosis, thrombocytaemia, and leucocytosis. At
higher dose levels in both males and females, the leucocytosis was
accompanied by a shift in the differential leucocyte count. There was
a slight increase in the activity of SGOT, SGPT and SAP. Urinalysis
revealed somewhat lower specific gravity and the presence of bile
pigment in animals at the two highest dietary concentrations.
Microscopic examination of tissues and organs revealed cytomegaly and
hyperplasia of the bile duct epithelium and Kupffer cells in some
animals at 750 mg/kg and in most animals at higher dose levels.
Nuclear inclusion bodies were also evident in all treated animals and,
at the highest dose level, moderate centrilobular fatty changes were
observed. Additionally, at the higher dose levels, atrophy of thymic
lymphoid tissue and of splenic white pulp was observed. Substantial
hyperplasia of the epithelium of the urinary bladder was observed in
most animals at the highest dose level and sporadically throughout the
treated groups.
In a study with 4-chloro- o-toluidine by Suter et al. (1976a),
groups of mice (30/sex/group, TIF:NMRI strain) were bred and
maintained under SPF conditions and fed a diet containing 4-chloro-
o-toluidine at concentrations of 0, 750, 1500, 3000 and 6000 mg/kg
for 60 days. Mortality of 50% was observed in the 6000 mg/kg group.
There were no clinical signs of toxicity, although food intake and
growth were retarded at the two highest dose levels. Eye examinations
did not indicate adverse ocular changes. Haemolytic anaemia occurred
in both sexes of all treated groups and was characterized by
reticulocytosis and Heinz body formation. In the male mice of all
treated groups, haemoglobin concentration, packed cell volume and
erythrocyte counts were slightly below that of controls. In addition,
leucocytosis was observed in all animals of all dosage groups with
the exception of females at the 750 mg/kg level. In both sexes at
6000 mg/kg and in the females at 3000 mg/kg total protein
concentration was reduced and blood glucose and urea nitrogen values
were increased. Plasma GPT was increased in male mice at 3000 mg/kg
and above and in females at 1500 mg/kg. Microscopic examination of
tissues and organs at the conclusion of the studies showed slight to
moderate vacuolar changes in hepatocytes, which were pronounced in
animals at the 3000 mg/kg level and above. There was also a marked
congestion of the spleen at these high dose levels. In addition, the
urinary bladder revealed hyperaemia and dilation of the capillaries in
the mucosal layer. These changes were accompanied by oedema, multiple
intra-epithelial haemorrhage and focal proliferation of the
transitional cell epithelium. On occasion, these changes in the
urinary bladder were noted at the lowest concentration.
7.2.1.2 Rat
In a study with chlordimeform by Sachsse et al. (1979b), groups
of Tif:RAIf rats (20/sex/group) were fed a diet containing
chlordimeform at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg
for 60 days. This corresponded to dietary intakes of 0, 84, 137, 222
or 462 mg/kg body weight per day for males and 0, 71, 121, 231 or
464 mg/kg body weight per day for females. Groups of 10 males and 10
females were killed at 60 days and had complete haematology, clinical
chemistry and urinalysis parameters examined. At the end of the study,
10 males and 10 females from each group were subjected to gross and
microscopic pathological examination. Animals that died during the
course of the study were similarly examined. Food intake and growth
were reduced over the course of the study at all dose levels. Slight
mortality was observed at the highest concentration. There were no
clinical signs of toxicity or adverse behaviour at any dose level.
Slight changes in several haematological parameters were noted at the
two highest levels. Methaemoglobin levels were increased in a dose-
related manner at all treatment levels. Heinz bodies were noted in
haematological examination at 1500 mg/kg and above. Slight changes
were noted in several clinical chemistry para-meters including
decreased glucose concentration, increased alkaline phosphatase
activity and increased �-glutamyl transpeptidase activity,
predominantly at the three highest dose levels. Urinalyses showed
slight changes at the two highest dose levels including a reduced
urine volume, reduced protein concentration, and reduced electrolyte
(potassium) level, predominantly at the highest dietary levels.
Terminal body weights of all animals administered chlordimeform were
significantly reduced in a dose-related fashion. Substantial changes
in growth and relative organ weights were noted in both males and
females at all dietary levels. Reductions in the weight of such organs
as the brain, heart, liver, kidneys, adrenals and thymus were reported
for both males and females. In males, reduced kidney and testes
weights were noted only at the highest dose level while reduced
ovarian weights were noted at all dose levels. Other than excessive
emaciation at the highest dose level, no gross anatomical changes were
noted in the animals killed for pathological examination. In most rats
of the highest-dose groups, haemosiderosis in the spleen was observed.
Reduced spermatogenesis was noted at the highest concentration. Focal
hyperplasia of small biliary ducts and of the transitional epithelium,
and increased vascularization in the mucous membrane of the bladder
were observed in the highest-dose group. In addition, the highest-dose
group showed thymic atrophy in several of the animals examined. No
compound-related histopathological changes were noted in rats fed
1500 mg/kg or below in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980b), groups of Tif:RAI rats (30/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of 0,
750, 1500, 3000 or 6000 mg/kg for 60 days. This corresponded to
dietary intakes of 0, 91, 176, 347 or 875 mg/kg body weight per day
for males and 0, 87, 165, 329 and 719 mg/kg body weight per day for
females. Groups of 10 males and 10 females were killed at the
conclusion of the study for complete haematological, clinical
chemistry and urinalysis examinations, and gross and microscopic
pathological examinations of tissues and organs. Extensive mortality
was observed at the high-dose level within the first few weeks of the
experiment. At the end of the third week of treatment, the highest-
dose group was terminated. There was no substantial mortality at 3000
or lower. Food intake and growth were reduced over the course of the
study in a dose-dependent fashion in all dose groups. Apart from the
mortality noted at the high dose level, no clinical signs of toxicity
or adverse behaviour were observed. Auditory and ophthalmological
examinations showed no evidence of loss of these functions in any of
the animals examined. Haematological examination indicated haemolytic
anaemia in both sexes of all treatment groups; characterized by a
reduction in haemoglobin concentration, erythrocyte count and packed
cell volume, and an increase in methaemoglobin level. Heinz bodies
were observed at 3000 mg/kg only. In addition, at 1500 mg/kg and above
there was a slight reticulocytosis and reduced partial thromboplastin
time in these dose groups. Changes in the clinical chemistry
parameters were noted at both 1500 and 3000 mg/kg. Gross examination
of certain tissues and organs showed changes in absolute weights
and relative weight ratios at all dosage levels. These reductions
appeared to follow a dose-dependent relationship. Animals administered
6000 mg/kg showed atrophy of the thymus and spleen within the first
three weeks of the test. Liver changes were noted in all dose groups
characterized as hyperplasia of the bile duct epithelium and changes
in the distribution of lipid. At the highest dose level, hyperplasia
of the urinary bladder epithelium and testes was noted. About half the
animals of both sexes in the 6000 mg/kg group showed an increase in
the mitotic index in hepatocytes.
In a study of 4-chloro- o-toluidine by Suter et al. (1976b),
groups of rats (30/sex/group; Tif/RAI strain) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 0, 750, 1500,
3000 and 6000 mg/kg for 60 days. There was no mortality over the
course of the study and clinical signs of toxicity were not observed.
Ophthalmological examinations did not suggest changes related to the
presence of 4-chloro- o-toluidine in the diet. Growth was reduced at
dietary levels of 1500 mg/kg and above. Haemolytic anaemia in both
sexes of all treated groups was characterized by a variety of
haematological changes, including reduced haemoglobin content, reduced
haematocrit content, reduced blood cell count, increased
methaemoglobin content, Heinz body formation, reticulocytosis and
polychromatophilia. In the highest-dose group, an increased number
of immature red blood cells (normoblasts) were observed. An increased
leucocyte count and prothrombin time was recorded at 3000 and
6000 mg/kg. Total protein was slightly reduced at 3000 and 6000 mg/kg
and there was a shift in the globulin content as observed by
electrophoresis. Plasma �-glutamyl transpeptidase of males and
alkaline phosphatase of females was increased at 6000 mg/kg.
Urinalysis was not significantly affected. In all treated animals, the
liver showed an increase in size accompanied by hypertrophy of the
hepatocytes. In the two highest-dose groups, the spleen was enlarged
and microscopic examination showed pronounced congestion and
haemorrhage. In the highest-dose group, slight or moderate
proliferation of the transitional cell epithelium was noted in the
urinary bladder.
7.2.1.3 Dog
In a study with chlordimeform by Blackmore (1969a), four groups
of beagle dogs were fed a dry diet containing either 0 mg/kg (10/sex),
250 mg/kg (8/sex), 500 mg/kg (8/sex) or 1000 mg/kg (10/sex) of
chlordimeform for 2 years. Two male and two female dogs were
sacrificed from each group at 26 and 52 weeks. Body weight was reduced
at 1000 mg/kg, the effect being slightly more pronounced in the
females. Total leucocyte counts were sporadically elevated in both
sexes at 1000 mg/kg and in females at 500 mg/kg. Haematocrit,
haemoglobin and erythrocyte counts tended to be depressed after 2
years in both sexes at 1000 mg/kg. Sporadic slight decreases in serum
albumin were observed, more frequently in males, at 1000 mg/kg.
Terminal spleen-to-body weight ratio was elevated in males at 500 and
1000 mg/kg, and in females at 1000 mg/kg. Histopathological
examinations revealed bile duct hyperplasia, pericholangitis and
nodular hepatocytic hyperplasia at 500 and 1000 mg/kg in both sexes,
and nodular hepatocytic hypertrophy at 1000 mg/kg in both sexes in the
liver. Kidneys showed an increased amount of pigmentation at 500 and
1000 mg/kg in both sexes.
7.2.2 Intubation
7.2.2.1 Rat
Four groups of 10 male and 10 female rats were intubated six
times weekly for one month with 5 ml/kg body weight of a 2% solution
of carboxymethylcellulose containing chlordimeform base at
concentrations such as to give dose levels of 0, 25, 50 or 100 mg/kg
(FAO/WHO, 1972). Body weight was markedly reduced in both sexes at
100 mg/kg. Hyperexcitability was observed in all test animals. At
100 mg/kg, this was apparent 20-30 min after dosing, and was followed
2 to 3 h after dosing by decreased activity and apathy. Recovery was
complete at 4 h. Similar but reduced effects were observed at 50 and
25 mg/kg, and with inconsistent frequency.
7.3 Long-term dietary exposure
7.3.1 Mouse
While there have been a number of long-term studies in mice with
chlordimeform and its metabolites, these were specifically designed to
study carcinogenic potential and are described in section 7.7.1.
7.3.2 Rat
In a study with chlordimeform by Blackmore (1969b), groups of
rats (35/sex/group) were fed a diet containing 0, 100, 250, 500 or
1000 mg/kg chlordimeform for 2 years. The 100 mg/kg group commenced
treatment 7 weeks after the other groups. This group was originally
part of the control group. Animals at that time were of similar weight
to those that had already been on test. The 1000 mg/kg group was
discontinued at 3 months due to severe growth inhibition. Growth
inhibition was observed in the males at 500 and 1000 mg/kg. In the
females, weight gain was reduced at 250 mg/kg and above. In addition,
female body weight gain was reduced at 100 mg/kg between weeks 20 and
48. Food intake was significantly reduced at 500 and 1000 mg/kg in
both sexes. Dose-related decreases in haematocrit, haemoglobin, and
erythrocyte counts, and a dose-related increase in the leucocyte count
occurred in females at 250 and 500 mg/kg up to one year. During the
second year, haematocrit only was consistently depressed in females at
500 mg/kg. Histopathological changes in the liver (nodules, and foci
of hyperplasia of hepatocytes) occurred in all groups, but the
incidence was greater at 250 and 500 mg/kg and was more severe at
500 mg/kg. Some females at 500 mg/kg showed slight hypertrophy and
vacuolation of focal groups of cells in the adrenal cortex. Terminally
organ to body weight ratios were increased in the liver (females at
250 and 500 mg/kg and males at 100 and 250 mg/kg), kidney (females at
250 and 500 mg/kg), thyroid (females at 250 and 500 mg/kg), heart
(males at 250 mg/kg and females at 500 mg/kg), adrenals (males at 100
and 250 mg/kg) and testes (100 and 500 mg/kg).
In a study with chlordimeform by Zak et al. (1973), groups of
rats (25/sex/group) were fed a diet containing chlordimeform at
concentrations of 0, 50, 75, 100, 250 and 500 mg/kg for one year. Food
consumption and weight gain data were recorded through the study.
Terminal organ weights and gross and microscopic examinations of
tissues and organs were the only other parameters reported. The
500 mg/kg group was terminated at 37 weeks after 10 males and 8
females had died. At the conclusion of the study, there was
considerable mortality noted in all groups. Food consumption was
decreased at 500 mg/kg in both sexes and was slightly reduced at
100 mg/kg and above in males only. This reduced food consumption was
not significantly reflected in the growth curves of males and females.
Gross examination did not show any compound-related abnormalities.
Organ weights and organ/body weight or organ/brain weight ratios did
not differ from control values. Histological examinations of liver and
spleen were performed on all animals. There were no significant
differences from control values with respect to fatty changes and
inflammatory changes in the liver. Slight proliferation of the bile
duct was more frequent in female rats treated with 500 mg/kg than in
the rats of other treated groups or the control group. Results of
examinations of the spleen for haemosiderosis suggested that, while
this condition was more pronounced in females, there were no
significant differences from control values.
In a study by Sachsse et al. (1980c), rats (90/sex/group;
Tif: RAIf strain) were fed a diet containing chlordimeform at
concentrations of 0, 2, 20, 100 or 500 mg/kg for 24 months. This was
equivalent to dosage levels of 0, 0.1, 1.0, 5.0 and 24 mg/kg body
weight per day for males and 0, 0.1, 1.2, 6.0, and 28 mg/kg body
weight per day for females. At the conclusion of the dietary feeding
study, all remaining rats were fed control diets for a period of time
until a survival rate of 20% per sex (10 rats) per group was attained,
at which time the animals were killed and examined. Groups of 20 male
and 20 female rats per group were examined periodically (4, 13, 26,
52, 78 and 104 weeks) for clinical laboratory investigations including
haematology, blood chemistry and urinalysis. Groups of 10 animals/sex/
group were sacrificed at 27 and 52 weeks for gross and microscopic
examination of tissues and organs. At the conclusion of the study, all
animals sacrificed (also those that died prior to the termination)
were examined for gross and microscopic pathology. There was no
mortality in the study attributable to the presence of chlordimeform.
Growth and body weight were maintained in all groups with the
exception of the 500 mg/kg group, where growth in both sexes was
slightly retarded. There were no clinical signs of toxicity or
abnormal behaviour. Ophthalmological and auditory examinations,
performed at periodic intervals, revealed no adverse effects
attributable to chlordimeform. Methaemoglobinaemia was observed at
dose levels of 20 mg/kg diet and above. At week 4, both males and
females showed a slight, but statistically significant, increase in
methaemoglobin content. At weeks 13 and 26, this condition abated but
returned at the end of one year and was significant in both sexes at
the highest dose level for the remainder of the study. Changes in
several other blood chemistry parameters were observed at the highest
dose level. Heinz body formation generally associated with
methaemoglobinaemia was not observed at week 4, but at the end of year
one and thereafter Heinz bodies were observed at the highest dose
level. A slight but significant reduction in blood glucose
concentration was noted at the higher dose levels throughout a major
part of the study. Slight changes in urinalysis parameters were
observed in the highest dose group, including a slightly reduced
urinary volume and a slightly higher specific gravity. Ketonuria and
proteinuria were observed at the high dose level at the earliest
examination periods only and were not observed at 13 weeks and
thereafter. Gross pathology and organ weight measurements (provided
for 27, 52 and 106-week sacrifice intervals) did not show any
significant dose-related responses. Microscopic histopathological
analyses of tissues and organs (performed at weeks 27 and 52 and at
the termination of the study) indicated no significant changes
attributable to chlordimeform in the diet. Although numerous benign
and malignant tumours were observed in both treated and control
animals, the frequency and type of neoplasms, reported at 12 and 24
months with pathology analyses, were not dose-related nor were they
attributable to chlordimeform in the diet. Several inherent,
degenerative or inflammatory changes were attributed to disease,
common in older animals. There was no indication of carcinogenic
potential to rats as a result of the presence of chlordimeform in the
diet. Based on the haematological occurrence of methaemoglobinaemia,
the no-observed-effect level of chlordimeform for rats was 2 mg/kg
diet, corresponding to an intake of 0.1 mg/kg body weight per day.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980d), groups of Tif:RAIf rats (90/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of
0, 2, 20, 100, or 500 mg/kg for 2 years. This corresponded to dietary
intakes of 0, 0.1, 1.0, 5 or 30 mg/kg body weight per day for females
and 0, 0.1, 1.0, 4.0 or 24 mg/kg body weight per day for males. Groups
of 10 males and 10 females were killed at periodic intervals (26 and
52 weeks) for examination of gross and microscopic pathology. Complete
haemato-logical, clinical chemistry, and urinalysis examinations were
performed at 4, 13, 26, 52 and 78 weeks on 20 males and 20 females of
each group. At 24 months, 20 males and 20 females were killed and
examined for clinical laboratory parameters and gross pathology. The
remaining animals were fed control diets for additional periods of
time until a survival rate of 20% per sex per group was attained.
At that time the remaining animals were killed and examined
microscopically for patho-logical changes, especially neoplastic and
non-neoplastic lesions. In the high-dose group, food intake and growth
were affected over the course of the study and slight growth
retardation was observed. Clinical signs of toxicity or adverse
behaviour were not observed. There was no mortality in the study
attributable to the presence of N-formyl-4-chloro- o-toluidine.
Ophthalmological examinations and auditory tests were normal. The
results of the haematological investigation showed haemoglobin
concentration to be slightly, but significantly, below that of the
controls in both male and female rats at the two highest dose levels.
In addition, slight but significant decreases in the erythrocyte count
and packed cell volume, a slight increase in reticulocytes and
somewhat higher methaemoglobin values were also seen in both male and
female rats at 500 mg/kg. With the exception of lower body weights of
the animals at the highest concentration, the most obvious change was
a significant increase in absolute and relative liver weights in both
sexes, but more pronounced in females, in the 500 mg/kg group. A
significantly increased incidence of hyperplasia of small biliary
ducts was seen in the liver of rats of the 500 mg/kg dose group. In
rats of the 500 mg/kg group that were killed after 2 years or died
after 12 months, a marked increase in the frequency of multioculated
cholangiogenic biliary cysts in the liver was noted. Both of these
finding were more pronounced and more frequent in female than in male
animals. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of neoplasms was
not treatment-related. All gross and histopathological lesions and
changes seen in both control and test animals were described as
inherent, degenerative or inflammatory in origin and were attributed
to naturally occurring diseases common in aged rats. There was no
indication of oncogenic potential in rats as a result of the presence
of N-formyl-4-chloro- o-toluidine. On the basis of the minor
haematological changes, the no-observed-effect level in this study was
20 mg/kg diet, corresponding to an intake of 1 mg/kg body weight per
day.
In a study with 4-chloro- o-toluidine by Sachsse et al. (1980e),
groups of Tif:RAIf rats (90/sex/dose level) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 or 500 mg/kg
for two years. This corresponded to dietary levels of 0, 0.1, 1.0, 5.0
or 28 mg/kg body weight per day for females, and 0, 0.1, 1.0, 4.6 or
24.6 mg/kg body weight per day for males. Groups of 10 males and 10
females were killed at periodic intervals (27 and 54 weeks) for gross
and microscopic pathological examinations. Complete haematological,
clinical chemistry and urinalysis examinations were performed at 4,
13, 26, 52, and 78 weeks on 20 females and 20 males of each group. At
24 months, 20 males and 20 females were killed and examined for
clinical laboratory parameters. Several animals were examined for
gross pathology. The remaining animals were fed control diets for
additional periods of time until a survival rate of 20% per group was
attained. At that time, the remaining animals were killed and examined
for microscopic pathology and oncogenic response. A complete
microscopic analysis was made on at least 10 rats of each sex of each
group at the termination of the experiment. All rats dying during the
course of the study were examined for tumours or neoplasms. In the
high-dose group of female rats, food intake and growth were affected
over the course of the study and slight growth retardation was
observed. There was no effect on growth in male rats at any dose
level. Clinical signs of toxicity were not observed. There was no
mortality in the study attributable to the presence of 4-chloro- o-
toluidine in the diet. Ophthalmological examinations and auditory
tests did not reveal changes that were related to the administration
of 4-chloro- o-toluidine. The results of the haematological
investigation, blood chemistry data and the urinalysis were similar
for both treated and control rats. Periodically, the haemoglobin
concentration was slightly but significantly below that of the
controls in the female rats at 100 mg/kg diet and above. Slight but
significant decreases were observed in the erythrocyte count and
packed cell volume in the female rats at 500 mg/kg. Marginal
reticulocytosis was also found to occur at 500 mg/kg in the female
rats at week 13 and in both sexes at week 26. In both male and female
rats at 500 mg/kg, the methaemoglobin level was found to be slightly
though significantly increased when compared to controls.
Periodically, this change was observed in the females of the 100 mg/kg
dose group, and, occasionally, Heinz bodies were also observed in
female rats. There were some changes to organ weights, organ-to-body
weight ratios and organ-to-brain ratios that were statistically
significant, but only the increase in absolute and relative liver
weights were dose-related. In rats from the 500 mg/kg dose group only,
a slightly but significantly increased incidence of multilobular
cholangiogenic cysts was observed in the liver. These biliary cysts
were found in 10/89 female and 3/90 male rats from the 500 mg/kg
group, compared to 4/89 female and 0/90 male rats in the control
group. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of the neoplasms
occurring in these animals was not treatment-related. Gross and
histopathological lesions and changes seen in both control and treated
animals were described as inherent, degenerative or inflammatory in
origin, and were attributed to naturally occurring diseases, common in
aged rats. There was no indication of oncogenic potential in rats as a
result of the presence of 4-chloro- o-toluidine in the diet. On the
basis of minor haematological changes, the no-observed-effect level in
this study was 20 mg/kg diet, corresponding to a dietary intake of
1 mg/kg body weight per day.
7.4 Skin and eye irritation; skin sensitization
Potential skin irritation was assessed by the application of
0.5 g chlordimeform or its hydrochloride salt to the shaved skin of
six male rabbits. When evaluated at 24 and 72 h, both compounds
produced a very slight irritation (FAO/WHO, 1972).
Potential eye irritation was assessed by application of 0.1 ml of
chlordimeform to one eye of each of nine rabbits, followed by
assessment over 7 days. All animals exhibited slight conjunctival
redness, while one showed slight chemosis. All effects had reversed
within 7 days. There was no evidence of corneal damage. Chlordimeform
may be considered a slight eye irritant (FAO/WHO, 1972).
There were no studies performed to assess the potential for
chlordimeform to cause skin sensitization.
7.5 Reproductive toxicity, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
7.5.1.1 Rat
Four groups of 10 male and 20 female rats were fed a diet
containing 0, 100, 250 and 500 mg/kg chlordimeform in corn oil during
three parental and three two-litter filial generations. Parental body
weight prior to mating tended to be reduced in all treatment groups,
especially at the highest dose level. The same tendency was apparent
with regard to food consumption. The fertility index, gestation index,
live birth index, sex ratio, mean litter size and birth weight of pups
were comparable between treatment and control groups in all
generations. At the 500 mg/kg dose level, the lactation index was
reduced in Fla, Flb and F3a litters. Weaning weight of offspring was
depressed in all high-dose litters. Gross pathological examinations
were performed on parents and pups dying during the study, and on 10
male and 10 female weanlings of the F3b generation. No compound-
related effects were noted in the pathological examination (Blackmore,
1969c).
In a study by Goldman et al. (1991), treatment of ovariectomized
Long-Evans rats with a single intraperitoneal injection of
chlordimeform at dose levels of 25 or 50 mg/kg caused a complete
suppression of luteinizing hormone surge. The observed suppression did
not persist beyond the day of treatment. In a more recent study by
Cooper et al. (1994), the effect of this delay in hormone surge on
pregnancy outcome in females was examined. Chlordimeform at a dose
level of 50 mg/kg resulted in a delay in breeding as well as a
significant reduction in litter size.
Adult male Sprague-Dawley rats were administered chlordimeform by
gavage at 200 mg/kg body weight on one day or 50 mg/kg body weight per
day for 5 days. Rats were killed on either 3 or 13 days after the last
dose. Body weights were reduced at the earlier time points only. There
were no changes in the weights of the testes or associated organs.
Caudal sperm counts were reduced after the single dose only. No other
changes were observed, including sperm motility, velocity or
morphology (Linder et al., 1992).
7.5.1.2 Hamster
Goldman et al. (1993) reported that a single intraperitoneal dose
of chlordimeform (75 mg/kg and above) is capable of delaying the
luteinizing hormone surge and altering the timing of oocyte release in
the hamster. The reproduction consequences of this effect were not
investigated.
7.5.2 Embryotoxicity and teratology
7.5.2.1 Rat
Groups of pregnant rats (25/treatment group, 30 controls) were
administered chlordimeform in carboxymethylcellulose at dose levels of
0, 10, 25 or 50 mg/kg body weight per day from days 6 to 15 of
pregnancy. Only a slight reduction in feed intake was noted at the
intermediate dose level. At the high dose level, dams showed
somnolence through the first 3 days of treatment. There was also a
reduced body weight gain and decrease in feed consumption at this dose
level. Examination of fetuses removed by caesarean section on day 21
showed there was a slight delay in growth of the fetuses at the two
highest dose levels. This effect was probably a direct result of the
toxic response in the dams. No teratogenic events were observed in the
offspring, although an increased incidence of sternal ossification
defects occurred at 25 mg/kg body weight (Fritz, 1975).
7.5.2.2 Rabbit
Three groups of 10 impregnated female New Zealand white rabbits
were administered chlordimeform by gavage on days 8 to 16 of gestation
at dose levels of 0, 7.5 or 30 mg/kg body weight per day. Five rabbits
per group were killed on day 28 of gestation. Parental mortality,
abortion rate, corpora lutea to implantation ratio, litter size,
incidence of resorption, stillbirths, fetal weight, fetal length, and
incidence of skeletal and tissue abnormalities were unaffected by the
test compound. In the remaining rabbits, which were allowed to litter
normally, gestation length, litter size and litter weights were
similar in both treated and control groups (Blackmore, 1969d).
Groups of rabbits (group size ranged from 17 to 38 dams per
group) were given chlordimeform orally from days 6 to 18 of pregnancy
at dose levels of 0, 10, 30 and 100 mg/kg body weight per day. Fetuses
were removed by caesarean section on day 28 of pregnancy. The
administration of chlordimeform at 100 mg/kg body weight produced a
distinct adverse effect on dams for 2-3 h for the first 4 days of
treatment. Examination of dams and fetuses at 28 days suggested that
the low dose had no teratogenic or embryotoxic effect. In the
intermediate and high dose groups, the implantation to corpora lutea
ratio was found to be reduced compared to controls. In the high dose
group, the number of incompletely ossified sternebrae showed a slight
increase over that observed in the controls and in the other groups.
In addition, the number of fetuses with malformations was slightly
increased at 100 mg/kg. These malformations included a median cleft
palate and exencephaly and an omphalocele. Further examination of
spontaneous malformations observed in a cumulative control of 2495
rabbit fetuses suggested that these abnormalities may be spontaneous
and not a consequence of the administration of chlordimeform (Fritz,
1971).
7.6 Mutagenicity and related endpoints
Referenced summaries of the test results with chlordimeform,
N-formyl-4-chloro- o-toluidine, and 4-chloro- o-toluidine are
given in Tables 8, 9 and 10, respectively. The important features of
these data are given below.
7.6.1 DNA damage and repair
Chlordimeform gave no evidence of unscheduled DNA synthesis in
rat hepatocytes (dose levels: 5-625 �g/ml) or in human fibroblasts
(dose levels: 2-250 �g/ml). 4-Chloro- o-toluidine, on the other hand,
gave a slight to moderate dose-related increase in the mean number of
silver grains per nucleus in rat hepatocytes over a dose range of
0.625-78.15 �g/ml, but not in human fibroblasts over the dose range of
1.25-156.25 �g/ml.
Table 8. Summary of mutagenicity and related end-point studies on chlordimeform HC1
Organism Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976a);
TA100 +/-S9 - Konopka & Heymann (1977);
TA1535 +/-S9 - Muecke et al. (1979);
TA1537 +/-S9 - Rashid et al. (1984)
TA1538 +/-S9 -
Salmonella typhimurium TA98 - Arni & M�ller (1983a)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983c)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
WP67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Insects Sex-linked recessive lethals Drosophila +a,b Kale et al. (1995)
Mammalian cells Gene mutation Mouse lymphoma L5178Y- TK+/-/ - Beilstein & M�ller (1984a)
in vitro +/-S9
Unscheduled DNA synthesis Rat hepatocytes - Puri & M�ller (1983a)
Unscheduled DNA synthesis Human fibroblasts - Puri & M�ller (1983c)
Mammalian cells Cell transformation Mouse BALB/3T3 cells +a,b Beilstein & M�ller (1983)
in vitro
Table 8. (con't)
Organism Test Test system Strain +/- References
Mammals Testicular cell chromosome Mouse spermatocytes - Hool et al. (1983)
damage Mouse spermatocytes - Arni et al. (1983a)
Micronucleus assay Chinese Hamster bone - Langauer & M�ller (1977)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & M�ller (1978)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983a)
marrow cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz (1978a)
a Chlordimeform formulation
b Not dose-related
Table 9. Summary of mutagenicity and related end-point studies on N-formyl-4-chloro-o-toluidine
Organism Test Test Systems Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976c);
TA100 +S9 + Konopka & Heymann (1977);
TA100 -S9 - Muecke et al. (1979);
TA1535 +/-S9 - Rashid et al. (1984)
TA1537 +/-S9 -
TA1538 +/-S9 -
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y +a Strasser & M�ller (1984b)
vitro
Mouse lymphoma L5178Y / -a Strasser & M�ller (1983b)
host-mediated assay
Mammals Testicular cell chromosome Mouse spermatogonia - Arni (1983b)
damage Mouse spermatocytes +b Arni & M�ller (1983e)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978a)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983f)
marrow metaphase cells
Table 9. (con't)
Organism Test Test Systems Strain +/- References
Mammals Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978b)
a No positive control
b Chromosome aberrations; not dose-related
Table 10. Summary of mutagenicity and related end-point studies on 4-chloro-o-toluidine
Organisms Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +S9 + Arni & M�ller (1976b);
TA98 +S9 - Haworth et al. (1983);
TA98 -S9 + Konopka & Heymann (1977);
Haworth et al. (1983);
TA100 +S9 - Meuke et al. (1979);
TA100 +S9 - Haworth et al. (1983)
TA100 -S9 - Rashid et al. (1984);
TA1535 +/-S9 - Haworth et al. (1983)
TA1537 +/-S9 -
TA1538 +/-S9 -
S. typhimurium TA98 - Arni & M�ller (1983b)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983d)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y/TK+/- -S9 - Beilstein & M�ller (1984b)
vitro +S9 +
Mouse lymphoma L5178Y + Strasser & M�ller (1984a)
Mouse lymphoma L5178Y - Strasser & M�ller (1983a)
/host-mediated
Table 10. (con't)
Organisms Test Test system Strain +/- References
Mammalian cells in Unscheduled DNA synthesis Rat hepatocytes + Puri & M�ller (1983b)
vitro
Human fibroblasts - Puri & M�ller (1983d)
DNA strand breakage V79 cells + Zimmer et al. (1980)
Cell transformation Mouse BALB/3T3 cells + Beilstein & M�ller (1984c)
Mammals Testicular cell chromosome Mouse spermatogonia - Hool & Arni (1983b)
damage Mouse spermatocytes - Hool & Arni (1983c)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978b)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983d)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983e)
marrow
Sister chromatid exchange Chinese hamster ovary + Galloway et al. (1987)
cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse + Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978)
N-Formyl-4-chloro- o-toluidine was not directly tested for its
ability to induce DNA damage and repair.
The macromolecular binding of 4-chloro- o-toluidine to macro-
molecules of rat and mouse liver has been investigated by several
authors. In a report by Hill et al. (1979), the binding of
4-chloro-2-[methyl-14C]-methylaniline (4-chloro- o-toluidine)
in vivo and in vitro was investigated. The major binding
in vivo was in the liver. In vitro binding was dependent on
the presence for microsomal preparations and NADPH. Two soluble
products of microsomal enzymes were identified as 5-chloro-2-
hydroxylaminotoluene and 4,4'-dichloro-2,2'-dimethylazobenzene. The
hydroxylamino compound appeared to be the more activated form of
4-chloro- o-toluidine.
4-Chloro- o-toluidine caused DNA strand breaks in Chinese
hamster V79 cells (Zimmer et al., 1980).
In studies by Bentley et al. (1986a,b), the covalent binding
of [14C- ring]-4-chloro- o-toluidine to mouse and rat liver
macromolecules was compared. After a single administration to either
species, the extent of binding decreased in the order: protein>RNA>
DNA. The level of binding to mouse liver DNA was approximately twice
as high as the binding to rat liver DNA after either single or
repeated administration, while the binding to liver RNA and protein
was greater in the rat. There was no evidence of an increased level
of incorporation of [3H]-thymidine into DNA as a result of
4-chloro- o-toluidine binding. Two major hydrophobic DNA adducts
were formed in both species, and one of these was formed to a much
greater extent (6-30 fold) in mice.
7.6.2 Mutation
The ability of chlordimeform and its metabolites to induce
mutations has been investigated in both microbial and mammalian
systems.
Chlordimeform itself gave uniformly negative results in
Salmonella typhimurium (0.1-2000 mg/ml), Saccharomyces cerevisiae
(15-400 mg/ml), and Escherichia coli (250-2000 mg/ml), with or
without S9 microsomal activation, as well as in a thymidine kinase
mutation assay in mouse lymphoma L5178Y/TK+/- cells with
(75-3000 mg/ml) or without microsomal activation (42.5-1700 mg/ml).
Kale et al. (1995) reported that a chlordimeform formulation diluted
to provide a dose level of 10 000 �g/ml is a potent sex-linked
recessive mutagen in male pre-meiotic and meiotic cells of Drosophila.
N-Formyl-4-chloro- o-toluidine was also negative in all
Salmonella typhimurium assays (0.1-1000 �g/ml) except for TA100 with
microsomal activation, in which there was a dose-related increase in
revertants. All Escherichia coli assays (250-2000 �g/ml) were
negative. In a forward mutation assay in mouse lymphoma L5178Y cells
(213 & 640 �g/ml), N-formyl-4-chloro- o-toluidine gave a positive
result in two out of three 18-h incubation experiments. In a host-
mediated experiment with mouse lymphoma L5178Y cells (300 mg/kg), the
result was negative.
4-Chloro- o-toluidine was negative in all assays with
Salmonella typhimurium (10-2000 �g/ml) except for TA100 with S9
microsomal activation and TA98 with S9 microsomal activation. Assays
with Saccharomyces cerevisiae (3.2-90 �g/ml) and with Escherichia
coli (250-2000 �g/ml) were negative. In a thymidine kinase forward
mutation assay in mouse lymphoma L5178Y/TK+/- cells, 4-chloro- o-
toluidine was negative without S9 microsomal activation (31.25-
500 �g/ml) and positive with microsomal activation (37.5-600 �g/ml).
In a separate forward mutation assay in mouse lymphoma L5178Y cells
(111 & 255 �g/ml), a positive result was obtained in one out of three
18-h incubation experiments. In a host-mediated experiment with mouse
lymphoma L5178Y cells (330 mg/kg), the result was negative.
7.6.3 Chromosome damage
Sister chromatid exchange assays in Chinese hamster bone marrow
cells were conducted following oral treatment with chlordimeform
(31-324 mg/kg) and 4-chloro- o-toluidine (100-400 mg/kg). In both
cases, the result was negative. Similarly, in an assay for chromosome
aberrations in Chinese hamster bone marrow metaphase cells, a negative
result was obtained following oral treatment with chlordimeform
(2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine (2 � 300-
1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-800 mg/kg), although
the results were somewhat erratic. A micronucleus test in Chinese
hamster bone marrow interphase cells following oral treatment with
chlordimeform (2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine
(2 � 300-1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-400 mg/kg)
was also negative.
Testicular cell chromosomal damage was assessed in mouse
spermatocytes and spermatogonia. To investigate the potential
cytogenetic effects on mouse spermatogonia, chlordimeform
(9-66 mg/kg), N-formyl-4-chloro- o-toluidine (80-320 mg/kg) or
4-chloro- o-toluidine (85-500 mg/kg) was administered orally on 5
consecutive days. The results were negative in each case. To
investigate the potential cytogenetic effects on mouse spermatocytes,
chlordimeform (18-72 mg/kg), N-formyl-4-chloro- o-toluidine
(80-320 mg/kg), or 4-chloro- o-toluidine (85-500 mg/kg) was
administered orally over 10 days on days 0, 2, 3, 5 and 9. The results
were negative in the case of both chlordimeform and 4-chloro- o-
toluidine, but non-dose-related evidence of chromosome damage was
indicated from the results with N-formyl-4-chloro- o-toluidine.
The heritable translocation assay, dominant lethal test, and
mammalian spot test, each of which might indicate minor or major
genomic changes, were conducted on all three compounds. In the
heritable translocation assay, chlordimeform (120 mg/kg/day),
N-formyl-4-chloro- o-toluidine (100 mg/kg/day) or 4-chloro- o-
toluidine (200 mg/kg/day) was administered orally for 49 days. No
induction of translocation heterozygosity was found.
In the dominant lethal assay, chlordimeform (22 or 66 mg/kg),
N-formyl-4-chloro- o-toluidine (105 or 315 mg/kg) or 4-chloro- o-
toluidine (110 or 330 mg/kg) was administered orally as a single dose.
There was no evidence of any dominant lethal effects in the progeny of
male mice.
In the mammalian spot test, chlordimeform (160 mg/kg),
N-formyl-4-chloro- o-toluidine (100 mg/kg) or 4-chloro- o-
toluidine (100 mg/kg) was administered orally on days 8-10 of
embryonic development. The results were negative except in the case of
4-chloro- o-toluidine, which induced a 3.2% incidence of spots of
genetic relevance compared to an incidence of 0.9% in controls.
7.6.4 Cell transformation
Cell transformation assays conducted with both chlordimeform and
4-chloro-toluidine in mouse BALB/3T3 cells produced an increased
incidence of transformed cell colonies with both compounds. With
chlordimeform, the experiment was conducted at dose levels up to
1 �g/ml, and increased transformation frequency was observed only at
0.0625 and 0.125 �g/ml. The transformative properties of chlordimeform
were considered weak. With 4-chloro- o-toluidine, three experiments
were conducted at dose levels up to 36 �g/ml, and a significant
increase in transformation frequency was observed. The transformative
properties of 4-chloro- o-toluidine were considered definite.
7.7 Carcinogenicity
A number of carcinogenicity studies have been conducted in mice.
With chlordimeform, there are two dietary studies and one dermal
study. With N-formyl-4-chloro- o-toluidine, there is one dietary
study. With 4-chloro- o-toluidine, there are four dietary studies.
In rats, the carcinogenic potential of chlordimeform and its
metabolites was generally investigated as part of more detailed
long-term studies, and details are provided in section 7.3.1.2. Three
studies on 4-chloro- o-toluidine that primarily investigated
carcinogenicity are reported below.
7.7.1 Mouse
The carcinogenicity of chlordimeform has been examined in two
dietary studies and in one dermal study.
In a study by Suter et al. (1978), groups of mice (50/sex/group;
Tif: MAG strain, SPF derived) were fed a diet containing chlordimeform
at concentrations of 0, 20, 100 and 500 mg/kg for 24 months. At the
conclusion of the dietary feeding interval, animals were maintained on
control diet until 90% of a group had died, at which time the
remaining animals of the group were sacrificed. There were no signs of
acute toxicity related to chlordimeform in the diet over the course of
the feeding trial. Growth and food consumption were similarly
unaffected by the presence of chlordimeform in the diet. Mortality was
significantly increased in females after 60 weeks at 500 mg/kg, and
after 90 weeks at both 100 and 500 mg/kg. In males, significantly
increased mortality was observed after 70 weeks at 500 mg/kg, and
after 110 weeks at both 100 and 500 mg/kg. However, lifespan was not
significantly affected in males at 100 mg/kg. The animals fed dietary
levels of 100 mg/kg and above displayed an increased incidence of
haemorrhagic tissue masses in subcutaneous tissues, retroperitoneum
and some internal organs (kidney, liver and spleen), which upon
examination were classified as malignant haemangioendotheliomas. These
malignancies which were reported to occur rarely in control
populations were found predominantly in the 100 and 500 mg/kg dietary
groups (see Table 11). In some animals the tumours were of multiple
origin and metastases were observed in the lungs. There were no other
types of neoplasm observed in the study that were attributable to
chlordimeform in the diet. Under the conditions of this study,
20 mg/kg in the diet appeared to be a no-observed-effect level.
In a study by Li et al. (1985a), groups of Swiss mice (50/group,
sex not stated) were fed a diet containing chlordimeform at
concentrations of 0, 20, 100, or 300 mg/kg for a period of 18 months.
A positive control group was administered 300 mg/kg of 4-chloro- o-
toluidine in the diet for 18 months. All animals were killed at the
end of the study and assessed for tumour formation. The main results
of the study are presented in Table 12. The author described the
majority of the neoplasms as angiomas, and the malignant neoplasms as
angiosarcomas. These neoplastic changes were considered to be similar
to those observed in the study by Suter et al. (1978).
In a paper by Jiang et al. (1988), the dermal carcinogenicity of
chlordimeform was investigated in mice. Groups of Swiss mice (50 per
dose level, sex not stated) were treated dermally with chlordimeform
twice per week at dose levels of 0, 100, 500, 2000 or 4000 mg/kg body
weight for a total of 17-20 treatments, together with croton oil (0.5%
in acetone). Positive controls received coal tar pitch (20 treatments)
plus croton oil (30 treatments). All animals were sacrificed after 18
months and analysed for tumour formation. Chlordimeform induced both
skin and liver tumours in this assay (see Table 13). The changes
observed in the skin consisted of epidermal hyperplasia, papillomas
and squamous cell carcinomas. The effect of croton oil application was
Table 11. Incidence of haemangioendotheliomas in mice following dietary administration
of chlordimeform, N-formyl-4-chloro-o-toluidine or 4-chloro-o-toluidine
(Suter et al., 1978; Sachsse et al., 1978a,b)
Control Dietary concentration (mg/kg)
2 20 100 500
Chlordimeform HCl
Male 1/44 (2%) - 0/44 (0%) 15/49 (30%) 39/48 (83%)
Female 1/43 (2%) - 2/46 (4%) 22/46 (50%) 35/49 (71%)
Total 2/87 (2%) - 2/90 (2%) 37/95 (41%) 74/97 (80%)
N-formyl-4-chloro-o-toluidine
Male 0/46 (2%) - 0/49 (0%) 15/48 (38%) 40/47 (89%)
Female 0/47 (0%) - 0/47 (0%) 23/43 (56%) 38/48 (79%)
Total 1/93 (1%) - 0/96 (0%) 38/91 (47%) 78/95 (84%)
4-Chloro-o-toluidine
Male 0/50 (0%) 0/47 (0%) 4/48 (8%) 23/47 (48%) 40/48 (83%)
Female 1/45 (7%) 1/45 (2%) 3/48 (6%) 30/47 (62%) 34/49 (72%)
Total 1/95 (1%) 1/92 (1%) 7/96 (19%) 53/94 (66%) 74/97 (78%)
to shorten the latent period for tumour formation and also to hasten
the malignant progression of existing neoplasms in the skin. At
500 mg/kg body weight, the time of first appearance of tumours was 483
days without croton oil and 154 days with croton oil. The latency
period also decreased with increasing dose levels of chlordimeform. In
the liver, changes consisted of enlargement, hepatocytic hyperplasia,
and hepatocytic carcinomas.
In a carcinogenicity study by Sachsse et al. (1978a), groups of
mice (50/sex/group; Tif: MAG strain) were fed N-formyl-4-chloro- o-
toluidine in the diet at concentrations of 0, 20, 100 and 500 mg/kg
for 24 months. After this time, all animals were fed a control diet
until the study was concluded when 90% of the animals in a group had
Table 12. Incidence of tumours in mice after dietary administration of chlordimeform or 4-chloro-o-toluidine (Li et al., 1985a)
Dietary Number of animals Number of animals Incidence Number of animals Incidence Days to
concentration necropsied bearing haemangiomas (%) bearing (%) appearance of
(mg/kg diet) or haemangiosarcomas haemangiosarcomas neoplasm
Chlordimeform
0 50 0 0 0 0 -
20 50 8 16 0 0 494
100 50 22 44 5 10 469
300 50 36 72 15 30 448
4-Chloro-o-toluidine
300 50 31 62 13 26 283
Table 13. Incidence of tumours in mice following dermal application of chlordimeform (Jiang et al., 1988)
Group/treatment Skin Liver
Number Carcinomas Papillomas Hyperplasia Number Carcinomas Papillomas Hyperplasia
of animals (%) (%) (%) of animals (%) (%) (%)
Water 18 0.0 0.0 6.1 17 0.0 0.0 0.0
Croton oil alone 17 0.0 0.0 17.6 24 0.0 0.0 8.3
100 mg/kg + croton oil 19 0.0 5.3 21.1 21 23.8 0.0 9.5
500 mg/kg 22 4.6 4.6 18.2 20 25.0 0.0 0.0
chlordimeform alone
500 mg/kg + croton oil 23 4.4 4.4 52.2 25 8.0 0.0 4.0
2000 mg/kg + croton oil 15 20.0 20.0 26.7 14 14.3 0.0 0.0
4000 mg/kg + croton oil 15 60.0 13.3 13.3 16 18.8 6.2 0.0
Coal tar pitch 18 88.9 11.1 0.0 19 15.8 5.3 0.0
died. There was no sign of adverse behaviour, and acute mortality was
not noted. Growth and food consumption were unaffected. There were
significant differences noted in survival after one year of age. Both
males and females showed an increased mortality at 100 and 500 mg/kg
after approximately one year of feeding. The onset of increased
mortality occurred earlier in females. The females at the 20 mg/kg
dietary level showed a slightly higher, non-significant, mortality
during the same period. Detailed gross and microscopic examination of
a variety of tissues and organs showed the presence of numerous gross
anatomical lesions. There was an increased number of haemorrhagic
masses in the subcutaneous tissues in the retroperitoneum and in some
internal organs of mice at all treatment levels. Detailed microscopic
examination confirmed that the increased incidence of haemorrhagic
masses were malignant tumours of vascular origin. These tumours were
histologically classified as malignant haemangioendotheliomas (see
Table 11). In addition to the occurrence of tumours, the time to
tumour relationship was decreased as the dietary concentration was
increased. Other neoplasms occurring in the study were not considered
to be treatment-related. A no-observed-effect-level was not
demonstrated under the condition of this experiment.
The carcinogenicity of 4-chloro -o-toluidine has been examined
in four dietary studies.
An early study by Ezumi and Nakao conducted in 1974 was reviewed
by the JMPR in 1978 and considered inadequate (FAO/WHO, 1979).
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives described by Homburger et al. (1972) and Weisburger
et al. (1978), groups of CD-1 mice (25/sex/dose level) were
administered 4-chloro- o-toluidine in the diet at dose levels of 0,
750 or 1500 mg/kg for males, and 0, 2000 or 4000 mg/kg for females for
a period of 18 months. All mice were placed on a control diet for an
additional 3 months before sacrifice and complete necropsy and
histopathological examination of tissues. The incidence of
haemangioendotheliomas was increased in males at both low (12/20) and
high (13/20) dose levels compared to concurrent controls (0/14) and
historical controls (5/99), and in females at both low (18/19) and
high (12/16) dose levels compared to concurrent controls (0/15) and
historical controls (9/102).
In a study by Sachsse et al. (1978b), groups of mice
(50/sex/group; Tif: MAGf strain) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 and
500 mg/kg for 24 months. After 24 months, all animals were fed control
diets until the study was concluded when 90% of the animals in a group
had died. There were no overt signs of toxicity. Growth and food
consumption were unaffected by treatment. An adverse effect on
longevity (lifespan) was noted in both males and females at the two
highest dietary levels. At the conclusion of the study upon gross
examination there was a marked increase number of haemorrhagic masses
in subcutaneous tissue, in the retroperitoneum, and in some internal
organs. Microscopic examination revealed an increased incidence of
haemorrhagic malignant tumours of vascular origin at dose levels of
20 mg/kg and above (see Table 11). The tumour incidence in control
exceeded the incidence observed at 2 mg/kg. The tumours were
histologically classified as malignant haemangioendotheliomas and, on
occasion, metastases were observed. There was not only a significant
dose-dependent increase in the total incidence of malignant tumours
but the time to appearance of tumours occurred at a markedly earlier
date in animals at the higher concentrations than in those at the
lower concentrations. A benign variant of the haemangioma was observed
in all groups, and although without the characteristics of malignancy,
did cause local invasion. Thus, the benign and malignant tumours have
been grouped together. The vascular tumours (haemangiomas and
haemangioendotheliomas) of the type that occurred in the mice appeared
to be peculiar to this rodent species. The occurrence of other types
of neoplasms in the study was not influenced by the presence of
4-chloro- o-toluidine in the diet. Under the conditions of this
experiment, 2 mg/kg in the diet appeared to be a no-observed-effect
level.
In a study with 4-chloro- o-toluidine conducted by the National
Cancer Institute (NCI, 1979), groups of B6C3F1 mice (50/sex/dose
level) were administered 4-chloro -o-toluidine in the diet at dose
levels of 3750 or 15 000 mg/kg for males and 1250 or 5000 mg/kg for
females for 99 weeks. Control groups consisted of 20 males and 20
females. There was a dose-related increase in mortality in both sexes.
There was also a dose-related increase in the incidence of
haemangiosarcomas as shown in Table 14. The haemangiosarcomas
apparently originated in fatty tissue adjacent to the genital organs
and not in a particular organ. In some instances, they were observed
to infiltrate the abdominal muscles, uterus, ovary, prostate or
urinary bladder. The haemangiosarcomas were lethal to 89 (75%) of the
affected mice, owing to haemorrhage in the peritoneal cavity and to
the space-consuming character of the lesions. Pulmonary metastasis was
observed in only 5 (4%) of the 119 dosed animals bearing
haemangiosarcomas. Associated pathological alterations that were
recorded at necropsy were haemorrhage in the peritoneal cavity and
variable enlargement of the spleen. It was concluded that 4-chloro-
o-toluidine was carcinogenic in B6C3F1 mice.
In the study of Li et al. (1985a), a single dietary dose of
4-chloro- o-toluidine (300 mg/kg) was given to mice for 18 months as
a positive control. The incidence of tumours was similar to that seen
in mice receiving 300 mg/kg of chlordimeform, but the latency period
was considerably reduced (Table 12).
Table 14. Incidence of tumours in mice following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 3750 15 000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 20 50 50 18 49 50
Haemangiosarcomas 0 3 37 0 40 39
(0%) (6%) (74%) (0%) (82%) (78%)
Haemangioma 0 3 5 1 6 0
(0%) (6%) (10%) (6%) (12%) (0%)
7.7.2 Rat
The carcinogenicity of chlordimeform and its metabolites has
generally been considered as part of more detailed long-term studies
(see Section 7.3). In the studies below, carcinogenicity was the
primary consideration.
In a study conducted by the National Cancer Institute (NCI,
1979), groups of F344 rats (50/sex/dose level) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 1250 mg/kg or
5000 mg/kg for 107 weeks. Control groups contained 20 animals of each
sex. There was no significant dose-related trend in mortality in
either sex. There was a variety of neoplastic and non-neoplastic
changes in control and treated rats. There was a small increase in
adenomas of chromophobe cells of the pituitary gland in both male and
female rats compared to controls (see Table 15). All of these tumours
were benign, are also common in this strain of rat and have occurred
in 21% of control female rats in the NCI laboratories. The authors
concluded that on the basis of histopathological examination,
4-chloro- o-toluidine was not carcinogenic in F344 rats.
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives by Weisburger et al. (1978), groups of male Charles
River CD rats were administered 4-chloro- o-toluidine in the diet at
dose levels of 0, 2000 or 4000 mg/kg diet for the first 3 months,
which was then reduced to 0, 500 or 1000 mg/kg diet for the following
15 months. There was no statistically significant increase in tumours
in either of the treated groups.
Table 15. Incidence of tumours in rats following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 1250 5000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 19 48 47 19 48 48
Chromophobe 2 6 15 1 13 15
adenoma (11%) (13%) (32%) (5%) (27%) (31%)
Chromophobe 0 0 2 0 3 1
hyperplasia (0%) (0%) (4%) (0%) (6%) (2%)
7.8 Other special studies
7.8.1 Immunotoxicity
In a study by Wiltrout et al. (1978), the potential of various
pesticides to influence the primary humoral immune response in the
mouse with respect to both dose and time of exposure was examined.
Mice receiving a single oral dose of chlordimeform at approximately
the LD50 level (148 mg/kg body weight) experienced a significant
suppression of humoral response when the dose was administered on the
day of immunization or two days after immunization. No response was
observed at one tenth of the LD50 dose, even when administered for
8 or 28 days.
Further studies by Shopp et al. (1985) investigated the effect of
chlordimeform on both humoral and cell-mediated immunity in the mouse
following both acute and 14-day exposures by the intraperitoneal
route. There was a decrease in IgM antibody-forming (plaque-forming)
cells when measured 4 days after intraperitoneal administration at 20
or 40 mg/kg body weight per day. These dose levels did not result in
any alteration of cell-mediated immunity. When administered orally,
chlordimeform at doses as high as 120 mg/kg body weight per day did
not have any effect on the 4- or 5-day antibody response.
Immunological parameters that may be related to the carcinogenic
activity of chlordimeform in rats were investigated by Thomas et al.
(1990). These included spleen/body weight ratio, splenocyte viability,
T and B cell mitogenesis, natural killer (NK) cell and natural
cytotoxic (NC) cell activity. Chlordimeform was administered
intraperitoneally on three consecutive days at 0, 1, 10 or 75 mg/kg
body weight per day. 4-Chloro- o-toluidine was administered
intraperitoneally on three consecutive days at 0, 10, 50 or 100 mg/kg
body weight per day. Spleen/body weight changes were observed only at
the highest dose of chlordimeform. No changes were observed with
either chemical on splenocyte viability or T and B cell mitogenesis.
An inhibition of NC activity at all chlordimeform doses was observed,
and an inhibition of NK activity was observed at 10 mg/kg body weight
per day and above. The relevance of this result to the carcinogenic
activity of these chemicals is doubtful.
7.8.2 Behavioural effects
Behavioural studies of the effects of chlordimeform in rats were
first investigated by Olson et al. (1978). The effects of exposure
prenatally and post-natally were examined following a dietary intake
of 0.1 mg/kg body weight per day. Early development testing (swimming
and righting reflex) was conducted on rat pups from post-natal days 7
to 17, while motivational, learning and retention tests were conducted
on days 70 to 90. The most significant differences between control and
treated groups was in the swimming task, retarded maturation being
observed in the chlordimeform-fed group. There was no treatment-
related effect with regard to maze tests or with regard to the tests
of motivation.
Moser et al. (1988) examined the behaviour of rats using a
functional observation battery following a single oral administration
of chlordimeform at dose levels of 0, 1, 25 or 56 mg/kg body weight.
Rats were examined at 1, 5 or 24 h. Chlordimeform produced a decrease
in body weight as well as a decrease in body temperature. There was a
dose-related increase in general activity, CNS excitability and
sensory responsiveness, coupled with a decrease in rearing, gait and
arousal. Chlordimeform also produced an increase in grip strength.
Other behavioural effects observed with chlordimeform have
included appetite stimulation in rats (Pfister et al., 1978b), flavour
aversion in both rats (MacPhail & Leander, (1980) and mice (Leander et
al., 1984) and alteration in schedule-controlled performance in rats
(MacPhail & Leander, 1981), mice (Glowa, 1986) and pigeons (Leander &
MacPhail, 1980). Witkin & Leander (1982) also demonstrated that, while
causing appetite stimulation in rats, chlordimeform produced a dose-
related decrease in water consumption, in contrast to other appetite
stimulants.
7.8.3 Pharmacological and biochemical effects
The pharmacological and biochemical effects of chlordimeform in
animals have been reviewed by Knowles (1991).
The cardiovascular effects of chlordimeform treatment were
recognized from an early stage with the observation that chlordimeform
administered intraperitoneally to rabbits caused a marked decrease in
arterial blood pressure of almost 50% within 30 min of treatment
(Matsumura & Beeman, 1976). Cardiovascular changes were also noted in
the dog (Lund et al., 1979a,b; Rieger et al., 1981) but in this case
the effect was biphasic, consisting of an initial depressor response
associated with decreased cardiac contractility and vascular
resistance, and a secondary pressor response associated with increased
cardiac contractility and vascular resistance. These actions of
chlordimeform were noted to be similar to those of local anaesthetics
such as procaine and lidocaine (Pfister et al., 1978a; Lund et al.,
1979a,b,c).
In studies by Watkinson (1985, 1986a,b), the effects of
chlordimeform on cardiovascular functional parameters were examined in
post-weaning and geriatric rats following intravenous treatment at
dose levels up to 60 and 120 mg/kg body weight, respectively, or
intraperitoneal treatment of post-weaning rats at dose levels up to
60 mg/kg body weight. Chlordimeform produced profound and abrupt
decreases in heart rate and blood pressure within 3 min, together with
multiple arrhythmias and alterations in electrocardiogram waveforms
and intervals. The effects observed in post-weaning rats were less
severe than those observed in geriatric rats.
The inhibition of monoamine oxidase in rats in vivo and
in vitro by chlordimeform and/or its metabolites has been
extensively studied (Beeman & Matsumura, 1973; Maitre et al., 1978;
Benezet et al., 1978; Hollingworth et al., 1979; Kadir & Knowles,
1981; Kaloyanova et al., 1981; Bailey et al., 1982). The lack of
correlation of toxicity of chlordimeform metabolites to monoamine
oxidase inhibition and the fact that chlordimeform is a relatively
weak monoamine oxidase inhibitor suggest that monoamine oxidase
inhibition is not the primary factor involved in the acute toxicity of
chlordimeform (Neumann & Voss, 1977; Robinson & Smith, 1977;
Hollingworth et al., 1979).
Chlordimeform also has an effect on the level of biogenic amines
in brain and plasma of rats, which may in part at least be due to
the inhibition of monoamine oxidase levels. Administration of
chlordimeform to rats was found to produce an increase of 25-70% in
5-hydroxytryptamine, norepinephrine or dopamine levels in brain
(Maitre et al., 1978; Benezet et al., 1978; Bailey et al., 1982).
However, Johnson & Knowles (1983) treated rats subcutaneously with
chlordimeform (200 mg/kg body weight) and found no change in any of
the amines.
Chlordimeform and some of its metabolites have been shown to
affect platelet function, as measured by the uptake of radioactive
5-hydroxytryptamine (Knowles, 1991).
Chlordimeform also has antipyretic and anti-inflammatory actions,
as shown by its ability to reduce yeast-induced fever in rats. It also
antagonizes both early (5-hydroxytryptamine- and histamine-mediated)
and late (prostaglandin-mediated) phases of carrageenan-induced
hind-paw oedema, albumin-induced oedema, and oedema induced by direct
injection of 5-hydroxytryptamine and histamine (Yim et al., 1978).
Chlordimeform also induced mild gastric ulceration in rats after
intraperitoneal injection (20-80 mg/kg body weight) but not after oral
treatment (80-240 mg/kg body weight). The above actions may be related
to the ability of chlordimeform to inhibit prostaglandin biosynthesis
(Yim et al., 1978; Holsapple & Yim, 1981).
Chlordimeform induces hypothermia in rats (Watkinson & Gordon,
1987) and mice (Gordon et al., 1985). Watkinson et al. (1989)
examined the effect of core body temperature on both the survival
and cardiovascular functions of rats following treatment with
chlordimeform. The results indicated that at a given dose of
chlordimeform, the magnitude and duration of the observed toxic
effects are primarily a function of core body temperature. The authors
concluded that moderate hypothermia, but not extreme hypothermia, may
have a beneficial effect with respect to survival after exposure to
chlordimeform.
Chlordimeform has been shown to have an effect on both visual and
auditory functions in mammals. Intraperitoneal treatment of male rats
with acute dosages of chlordimeform (5-40 mg/kg body weight) before
testing revealed a temporary increase in both the amplitude and
latency of pattern reversal-evoked potentials and an increase only in
the latency of pattern flash-evoked potentials (Dyer & Boyes, 1983;
Boyes & Dyer, 1984). Boyes & Moser (1988) provided evidence to support
the hypothesis that these effects are evoked through actions as a
central nervous system alpha-adrenegic agonist. Janssen et al. (1983)
demonstrated effects on the brain stem auditory-evoked response after
injection of chlordimeform at a dose levels of 40 mg/kg body weight.
It has been suggested that these effects may by secondary to the
hypothermic effects induced by chlordimeform (Gordon et al., 1985).
Chlordimeform has been shown to affect the activity of hepatic
drug-metabolizing enzymes in both rats and mice. Studies have been
conducted following gastric intubation at dose levels up to 150 mg/kg
body weight per day for 7 days, and also following intraperitoneal
injections either singly (100 mg/kg body weight) or daily (75 mg/kg
body weight per day) for 4 days. Chlordimeform treatment induced
several of these hepatic drug-metabolizing enzymes with significant
species and/or sex specificity. Cytochrome P-450 content was increased
in all cases.
7.9 Factors modifying toxicity
The factors modifying the acute toxicity of chlordimeform have
been reviewed by Knowles (1991).
7.10 Mechanisms of toxicity - mode of action
7.10.1 Mechanism of acute toxicity
A large number of studies that investigated the mechanism of
action following acute poisoning with chlordimeform have been
reported.
Based on the early in vitro and in vivo studies of Aziz &
Knowles (1973) and Beeman & Matsumura (1973), it was suggested that
the increase in biogenic amines resulting from inhibition of monoamine
oxidase by chlordimeform could account for the variety of toxic signs
following acute poisoning. However, Maitre & Gfeller (1975) and
Robinson et al. (1975) demonstrated that this mechanism does not play
a significant role in the acute toxicity in rats.
A number of other studies have attempted to define the mode of
action of chlordimeform. Studies in insects have shown that
chlordimeform has little activity on cholinergic transmission although
it is an uncoupler of oxidative phosphorylation and an inhibitor of
electron transport (Abo-Khatwa & Hollingworth, 1972a). A number of
biochemical mechanisms have been postulated to explain the effects of
chlordimeform in insects, including uncoupling of respiration
(Abo-Khatwa & Hollingworth, 1972a,b), inhibition of monoamine oxidase
(Knowles & Roulston, 1972) and blockage of neuromuscular transmission
(Wang et al., 1975; Watanabe et al., 1975), and motor stimulation
through actions on central non-cholinergic synapses (Lund et al.,
1979a; Lund et al., 1979c). The latter effect is thought to be
mediated through the neurotransmitter, octopamine (Lund et al.,
1979b). Both chlordimeform and particularly demethylchlordimeform have
been shown to interact with the octopamine receptor and partially
mimic the pharmacological effects of octopamine (Evans & Gee, 1980;
Nathanson & Hunnicutt, 1981; Bokisch et al., 1985).
In mammalian systems, oxidative phosphorylation is uncoupled
(Abo-Khatwa & Hollingworth, 1972b) and RNA synthesis is inhibited by
chlordimeform, but only at very high concentrations (Murakami &
Fukami, 1974). The effects of chlordimeform on hepatic drug-
metabolizing enzymes was dependent on both sex and species and did not
show any particular pattern that would indicate a consistent mechanism
of action (Budris et al., 1983; Bentley et al., 1985; Leslie et al.,
1988).
Chlordimeform, acting as a direct depressant on cardiac and
vascular muscle, induced a hypotensive state in dogs. Chlordimeform
did not interfere with the autonomic nervous system. The mechanism of
cardiovascular depression may be related to that noted with frog nerve
preparations treated with procaine, a local anaesthetic. The
metabolite, 4-chloro- o-toluidine has been shown to interfere with
rat cardiac receptors (Wang et al., 1975; Watanabe et al., 1975;
Matsumura & Beeman, 1976; Knowles, 1976; Hollingworth, 1976; Lund et
al., 1978a).
More recent research has shown that formamidine pesticides may
exert their effects on the central nervous system by interacting
directly with adrenergic receptors, particularly the alpha-2 subtype
(Costa & Murphy, 1987; Costa et al., 1988, 1989). This interaction
appears to mediate several of the observed effects of formamidines,
such as changes in heart rate (Hsu & Kakuk, 1984, Watkinson, 1985;
1986a,b), pupil diameter (Hsu & Kakuk, 1984), visual evoked potential
(Boyes & Moser, 1988) and hormonal secretion (Goldman et al., 1990;
1991). Costa et al. (1991) demonstrated that chlordimeform
decreases the hepatic glutathione content by up to 40% in a
time- and dose-dependent manner, through an interaction with
alpha2-adrenoreceptors. Wu et al. (1990) have demonstrated that
chlordimeform inhibits the binding of the known alpha2-adrenoreceptor
blockers, clonidine and yohimbine, in rat forebrain tissue
in vitro. Furthermore, Stoker et al. (1991), in a further study on
the effects of chlordimeform on hormone release, have demonstrated in
rats, treated intraperitoneally with chlordimeform (20 or 50 mg/kg
body weight), that there is an increase in adrenocorticotropic hormone
(ACTH), circulating corticosteroid (CORT) and prolactin (PL) in a
dose-dependent manner. alpha-Adrenergic agonists specifically
inhibited these effects indicating the interference with a regulatory
signal mediated by alpha-adrenergic receptor-associated activity.
Candura et al. (1992) demonstrated that the inhibition induced by
chlordimeform in the intestinal tract is mediated by calcium channel
blockade rather than by alpha2-adrenoceptor activation. In a study by
Robinson et al. (1975), it was found that using drugs to block the
serotonergic or alpha-adrenergic receptors did not reduce the
chlordimeform-induced lethality in male rats.
7.10.2 Mechanism of carcinogenicity
Chlordimeform and its metabolites, N-formyl-4-chloro- o-
toluidine and 4-chloro -o-toluidine, have been shown to induce mouse
tumours of a vascular origin characterized histologically as
haemangioendotheliomas and haemangiosarcomas. 4-Chloro- o-toluidine
has been shown to be a more potent carcinogen than chlordimeform, both
with respect to dose-response and to a reduced latency period.
Haemangioendotheliomas and haemangiosarcomas were not induced in rats.
Cases of bladder cancer in humans associated with occupational
exposure to high levels of chlordimeform or 4-chloro- o-toluidine
have been seen in groups with high urinary levels of chlordimeform and
4-chloro- o-toluidine.
The exact mechanism of induction of these tumours is unknown but
there is evidence that a genetic mechanism involving mutations induced
by 4-chloro- o-toluidine is involved.
Metabolic studies in mice and rats indicate a similar metabolic
pathway for chlordimeform in both species. The kinetics of absorption
and elimination in mice and rats also seem to be similar. However, the
overall DNA binding was higher in mice than rats, and one DNA adduct
was formed to a 6- to 30-fold higher extent in mice.
There is considerable evidence that 4-chloro- o-toluidine causes
severe toxic effects in the human bladder leading to haemorrhagic
cystitis (see section 8). Monitoring of urinary metabolites in humans
also indicates that chlordimeform is rapidly metabolized to 4-chloro-
o-toluidine in vivo.
4-Chloro- o-toluidine also has a close structural similarity
to aromatic amines for which there is established evidence of
carcinogenicity by animal experimentation and also by human
epidemiological data (Parkes, 1984).
Taken together, the evidence strongly implicates 4-chloro- o-
toluidine as the causative agent in the induction of tumours in both
mice and humans. A proposed route of activation that may be associated
with carcinogenicity is shown in Fig. 2. However, the mechanism of the
carcinogenicity remains unclear.
In the same study, metabolites extracted from goat urine were
analysed by thin-layer chromatography. The major urinary metabolite
was N-formyl-4-chloro- o-toluidine. The metabolites in goat urine
showed a similar pattern to those in rats, with a similar proportion
of conjugated material.
The comparative metabolic fate of chlordimeform in rats, goats
and dogs is considered in a review by Knowles (1970), which emphasizes
the similarity between these species.
6.2.3 In vitro studies
Initial studies on the in vitro metabolism of chlordimeform
were conducted with [3H-phenyl]-chlordimeform (FAO/WHO, 1972).
Incubation of [3H-phenyl]-chlordimeform for 120 min with rat liver
homogenate resulted in 24% unchanged chlordimeform, 45% 4-chloro- o-
toluidine, and 11% unidentified metabolites being formed. Rabbit liver
homogenate yielded 53, 40 and 7% of the same metabolites,
respectively. Incubation of 60 �g [3H-phenyl]-chlordimeform (30 �Ci)
with 5 ml human plasma yielded N-formyl-4-chloro- o-toluidine only.
Conversion was 25% in 5 h, and 50% in 20 h. Rose (1969a) confirmed the
rat liver homogenate studies using [14C]-chlordimeform. Three
unidentified metabolites were also observed and, in addition,
chlordimeform degradation was shown to require the presence of
nicotinamide. Spleen homogenates were inactive with regard to
chlordimeform degradation.
The metabolism of chlordimeform in vitro was first reported by
Ahmad & Knowles (1971). Incubation of [14C]-chlordimeform with
various rat liver enzyme preparations identified demethylchlordimeform
as the major metabolite, which was formed by microsomal
N-demethylase in the presence of exogenous nicotinamide. This
reaction was inhibited by mixed function oxidase inhibitor, SKF-525A.
The chlordimeform metabolites formed in vitro were qualitatively
similar to those detected in urine from chlordimeform-treated mammals.
This has been confirmed by others (Hill et al., 1979; Ghali
& Hollingworth, 1985; Kimmel et al., 1986; Watanabe & Matsumura,
1987).
Knowles & Benezet (1977) confirmed that the major in vitro
metabolite was demethylchlordimeform, but also found that
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
present in appreciable amounts.
Ahmad & Knowles (1971) also investigated the metabolism of
[14C]- N-formyl-4-chloro- o-toluidine) in the presence of rat liver
enzyme preparations. Eighty percent of this metabolite was metabolized
by an enzyme, probably a hydrolase, in the soluble fraction, with
major metabolites being 4-chloro- o-toluidine (52%) and an unknown
substance (26%).
The question of the possible formation of azo-derivatives in
animal tissues was investigated by Rose (1969a). A number of
experiments were conducted to investigate the presence or absence of
azobenzene formation from chlordimeform or 4-chloro- o-toluidine. In
the first experiment, it was demonstrated that peroxidase activity was
negligible in rat liver and spleen. Furthermore, catalase, which was
abundant in the same tissues, and which, like peroxidase, catalyses
reactions between hydroxyperoxides and many oxidizable compounds, was
shown to be unable to form symmetrical azo-derivatives from
4-chloro- o-toluidine. In the second experiment, it was demonstrated
that rat liver and spleen homogenates, which were fortified with
nicotinamide, and which degrade chlordimeform to demethylchlordimeform
and small quantities of N-formyl-4-chloro- o-toluidine and
4-chloro- o-toluidine, respectively, did not form any azobenzene
derivatives. These compounds therefore do not represent metabolites of
chlordimeform or its aromatic amine degradation products in animal
tissues.
Lin et al. (1975) have investigated the metabolism of
chlordimeform in primary embryonic lung cell cultures. In 2 h of
incubation, 97% of chlordimeform was metabolized to N-formyl-4-
chloro- o-toluidine (81.9%) and 4-chloro- o-toluidine (2.3%). The
route of metabolism, which was different to that seen in mammals,
appeared to be first demethylation followed by cleavage at the
carbon-nitrogen double bond to form N-formyl-4-chloro- o-toluidine.
The formation of the demethylchlordimeform was minute compared to that
of the N-formyl derivative. The minor metabolites observed were
demethylchlordimeform and two unknown metabolites. When incubated in
culture media without cells, chlordimeform decomposed to
N-formyl-4-chloro- o-toluidine.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 Oral
The acute oral toxicity data for chlordimeform is presented in
Table 5 and for chlordimeform hydrochloride in Table 6.
The general signs of toxicity in rats are hyperactivity,
dyspnoea, muscular weakness, tremors, "Straub's tail", spasms,
convulsions and respiratory arrest. No pathological changes were noted
in the rat following oral treatment.
In mice, signs of toxicity were similar, but some differences
were noted. Symptoms included restlessness, hyperreflexia and tremors,
particularly of the head and forelimbs, that developed to one or more
episodes of clonic convulsions. Death usually occurred within one hour
during one of the convulsive episodes. If the animal survives this
hyperexcitation and tremor, it becomes sedated, locomotion is
suppressed, and it usually recovers within 24 h.
The acute oral toxicity data for metabolites of chlordimeform is
presented in Table 7.
7.1.2 Other routes
The acute dermal toxicity data for chlordimeform in rats is
presented in Table 5 and for chlordimeform hydrochloride in Table 6.
The base, but not the hydrochloride, is readily absorbed by the skin
(Knowles, 1991). The general signs of toxicity are dyspnoea,
exophthalmos, prostration, spasms and convulsions. Pathological
changes in the rat included pale or blotchy liver, pale kidneys, and
haemorrhagic intestinal contents. No local skin irritation occurred.
In the dog, a lethal intravenous dose of chlordimeform (50 mg/kg
body weight) caused rapid and irreversible hypotension, and
respiratory arrest followed cardiovascular collapse within a few
seconds. Artificial respiration did not protect the animals against
hypotension and death, suggesting cardiovascular collapse is probably
the primary cause of death in dogs. Pathological examination following
oral administration in dogs revealed congestion of liver, kidneys and
lungs.
The acute inhalation LC50 of chlordimeform base in rats
(male and female) was 17 400 mg/m3 and for chlordimeform HCl was
>5800 mg/m3 (FAO/WHO, 1972).
The acute dermal toxicity data for metabolites of chlordimeform
is presented in Table 7.
Table 5. Acute toxicity of chlordimeform in experimental animals
Species Sex Route LD50 References
(mg/kg
body weight)
Rat male/female oral 250 FAO/WHO (1972)
male/female oral 340 Worthing & Walker (1983)
male/female oral 123 Robinson et al. (1975)
male/female oral 301 Gaines & Linder (1986)
male/female oral 178-220 FAO/WHO (1972)
male/female oral 178 FAO/WHO (1972)
female oral 170-460 FAO/WHO (1972)
female oral 265 Gaines & Linder (1986)
female oral 460 FAO/WHO (1972)
male/female dermal 640 FAO/WHO (1972)
male dermal 337 Gaines & Linder (1986)
female dermal 263 Gaines & Linder (1986)
Mouse male/female oral 290 Haddow & Shankland (1969)
male oral 267 Ghali & Hollingworth (1985)
male/female ip 110 FAO/WHO (1972)
Rabbit - oral 625 FAO/WHO (1972)
- oral 625 Worthing & Walker (1983)
Dog male oral approx.150 Hurni & Sachsse (1969)
female oral approx.100 Hurni & Sachsse (1969)
Table 6. Acute toxicity of chlordimeform hydrochloride in
experimental animals (FAO/WHO, 1972)
Species Sex Route LD50
(mg/kg body weight)
Rat male oral 305
male oral 325
female oral 330
male iv 95
- dermal approx. 4000
Mouse male/female oral 220
Rabbit - dermal >4000
Table 7. Acute toxicity of chlordimeform metabolites in the rat (FAO/WHO, 1972)
Metabolite Sex Route LD50
(mg/kg
body weight)
N-formyl-4-chloro-o-toluidine male/female oral 2900
4-chloro-o-toluidine (base) male/female oral approx.1000
4-chloro-o-toluidine-HCl male/female oral 860
N-formyl-4-chloro-o-toluidine male/female dermal (24 h) >2150
4-chloro-o-toluidine (base) male/female dermal (24 h) approx.1800
4-chloro-o-toluidine-HCl male/female dermal (24 h) >2150
7.2 Short-term exposure
7.2.1 Dietary
Dietary studies of 60 days duration have been conducted in the
mouse and rat with each of chlordimeform, N-formyl-4-chloro -o-
toluidine, and 4-chloro- o-toluidine.
7.2.1.1 Mouse
In a study with chlordimeform by Sachsse et al. (1979a), groups
of Tif:MAGf mice (30/sex/group), housed under SPF conditions, were fed
a diet containing chlordimeform at concentrations of 0, 750, 1500,
3000 or 6000 mg/kg for 60 days. This corresponded to dietary intakes
of 0, 107, 194, 717 or 1525 mg/kg body weight per day for females and
0, 110, 200, 669 or 1519 mg/kg body weight per day for males. At the
end of the 60-day period, all animals were examined for haematology,
blood chemistry and urinalysis parameters, and groups of 10 male and
10 female animals from the control and the lower three dose groups
were subjected to gross and microscopic examination of tissues and
organs. Mortality was observed in the two highest dose groups over the
course of the study. The highest dose group was terminated after two
weeks because of a poor general condition of the animals. Growth, as
shown by body weight gain, was reduced in all dietary groups. Food
consumption was reduced at all dietary levels in females only. No
clinical signs of toxicity were noted. Ophthalmological and auditory
examinations were normal. Haematological investigations showed
haemolytic anaemia in both sexes of all treated groups, which was
characterized as a reduction in haemoglobin concentration, red blood
cell count, and packed cell volume. The anaemia was associated in a
dose-related manner with an increased methaemoglobin concentration and
an increase in Heinz body formation. At 3000 mg/kg diet, there was a
slight reticulocytosis noted in both sexes. This was accompanied in
females by a shift in the differential leucocyte count noted as an
increase in the percentage of polymorphonuclear neutrophile and a
decrease in the percentage of lymphocytes. Small changes were observed
in alkaline phosphatase activity, which was slightly increased in male
mice at the highest dose level. Total protein concentration was also
slightly reduced in female mice at the highest dose level. Urinalysis
was unremarkable. In the animals that died or were killed within the
first 2-week period, all were found to be emaciated and in poor
general condition. In all treated animals dying during the test
period, congestion of the organs, especially of the liver, was
observed. At the highest dose level, atrophy of thymic tissue was
observed. There was an increased haemosiderosis at the two highest
dose levels. There were no other pathological findings associated with
the presence of chlordimeform in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980a), groups of Tif:MAGf mice (30/sex/group), housed under SPF
conditions, were fed a diet containing N-formyl-4-chloro- o-
toluidine at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg for
60 days. This corresponded to dietary intakes of 0, 138, 379, 1203,
or 3153 mg/kg body weight per day for females and 0, 140, 349, 1023,
2549 mg/kg body weight per day for males. At the end of the 60-day
period, all animals were examined for haematology, clinical chemistry
and urinalysis parameters. A group of 10 males and 10 females from
each dose level was examined for gross and microscopic pathological
changes at the conclusion of the study. Mortality was observed
predominantly at the high-dose level over the course of the study.
There were no clinical signs of toxicity, although food consumption
and growth were depressed at 1500 mg/kg and above in both sexes over
the course of the study. Ophthalmological and auditory examinations
were unremarkable. Significant haematological abnormalities were
observed at all dose levels at the conclusion of the study. Haemolytic
anaemia was observed in both males and females and was characterized
as a reduction in haemoglobin concentration, erythrocyte count
and packed cell volume. There was a dose-related increase in
methaemoglobin concentration and an increase in Heinz body formation.
Additionally, both males and females in all treated groups showed a
significant reticulocytosis, thrombocytaemia, and leucocytosis. At
higher dose levels in both males and females, the leucocytosis was
accompanied by a shift in the differential leucocyte count. There was
a slight increase in the activity of SGOT, SGPT and SAP. Urinalysis
revealed somewhat lower specific gravity and the presence of bile
pigment in animals at the two highest dietary concentrations.
Microscopic examination of tissues and organs revealed cytomegaly and
hyperplasia of the bile duct epithelium and Kupffer cells in some
animals at 750 mg/kg and in most animals at higher dose levels.
Nuclear inclusion bodies were also evident in all treated animals and,
at the highest dose level, moderate centrilobular fatty changes were
observed. Additionally, at the higher dose levels, atrophy of thymic
lymphoid tissue and of splenic white pulp was observed. Substantial
hyperplasia of the epithelium of the urinary bladder was observed in
most animals at the highest dose level and sporadically throughout the
treated groups.
In a study with 4-chloro- o-toluidine by Suter et al. (1976a),
groups of mice (30/sex/group, TIF:NMRI strain) were bred and
maintained under SPF conditions and fed a diet containing 4-chloro-
o-toluidine at concentrations of 0, 750, 1500, 3000 and 6000 mg/kg
for 60 days. Mortality of 50% was observed in the 6000 mg/kg group.
There were no clinical signs of toxicity, although food intake and
growth were retarded at the two highest dose levels. Eye examinations
did not indicate adverse ocular changes. Haemolytic anaemia occurred
in both sexes of all treated groups and was characterized by
reticulocytosis and Heinz body formation. In the male mice of all
treated groups, haemoglobin concentration, packed cell volume and
erythrocyte counts were slightly below that of controls. In addition,
leucocytosis was observed in all animals of all dosage groups with
the exception of females at the 750 mg/kg level. In both sexes at
6000 mg/kg and in the females at 3000 mg/kg total protein
concentration was reduced and blood glucose and urea nitrogen values
were increased. Plasma GPT was increased in male mice at 3000 mg/kg
and above and in females at 1500 mg/kg. Microscopic examination of
tissues and organs at the conclusion of the studies showed slight to
moderate vacuolar changes in hepatocytes, which were pronounced in
animals at the 3000 mg/kg level and above. There was also a marked
congestion of the spleen at these high dose levels. In addition, the
urinary bladder revealed hyperaemia and dilation of the capillaries in
the mucosal layer. These changes were accompanied by oedema, multiple
intra-epithelial haemorrhage and focal proliferation of the
transitional cell epithelium. On occasion, these changes in the
urinary bladder were noted at the lowest concentration.
7.2.1.2 Rat
In a study with chlordimeform by Sachsse et al. (1979b), groups
of Tif:RAIf rats (20/sex/group) were fed a diet containing
chlordimeform at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg
for 60 days. This corresponded to dietary intakes of 0, 84, 137, 222
or 462 mg/kg body weight per day for males and 0, 71, 121, 231 or
464 mg/kg body weight per day for females. Groups of 10 males and 10
females were killed at 60 days and had complete haematology, clinical
chemistry and urinalysis parameters examined. At the end of the study,
10 males and 10 females from each group were subjected to gross and
microscopic pathological examination. Animals that died during the
course of the study were similarly examined. Food intake and growth
were reduced over the course of the study at all dose levels. Slight
mortality was observed at the highest concentration. There were no
clinical signs of toxicity or adverse behaviour at any dose level.
Slight changes in several haematological parameters were noted at the
two highest levels. Methaemoglobin levels were increased in a dose-
related manner at all treatment levels. Heinz bodies were noted in
haematological examination at 1500 mg/kg and above. Slight changes
were noted in several clinical chemistry para-meters including
decreased glucose concentration, increased alkaline phosphatase
activity and increased �-glutamyl transpeptidase activity,
predominantly at the three highest dose levels. Urinalyses showed
slight changes at the two highest dose levels including a reduced
urine volume, reduced protein concentration, and reduced electrolyte
(potassium) level, predominantly at the highest dietary levels.
Terminal body weights of all animals administered chlordimeform were
significantly reduced in a dose-related fashion. Substantial changes
in growth and relative organ weights were noted in both males and
females at all dietary levels. Reductions in the weight of such organs
as the brain, heart, liver, kidneys, adrenals and thymus were reported
for both males and females. In males, reduced kidney and testes
weights were noted only at the highest dose level while reduced
ovarian weights were noted at all dose levels. Other than excessive
emaciation at the highest dose level, no gross anatomical changes were
noted in the animals killed for pathological examination. In most rats
of the highest-dose groups, haemosiderosis in the spleen was observed.
Reduced spermatogenesis was noted at the highest concentration. Focal
hyperplasia of small biliary ducts and of the transitional epithelium,
and increased vascularization in the mucous membrane of the bladder
were observed in the highest-dose group. In addition, the highest-dose
group showed thymic atrophy in several of the animals examined. No
compound-related histopathological changes were noted in rats fed
1500 mg/kg or below in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980b), groups of Tif:RAI rats (30/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of 0,
750, 1500, 3000 or 6000 mg/kg for 60 days. This corresponded to
dietary intakes of 0, 91, 176, 347 or 875 mg/kg body weight per day
for males and 0, 87, 165, 329 and 719 mg/kg body weight per day for
females. Groups of 10 males and 10 females were killed at the
conclusion of the study for complete haematological, clinical
chemistry and urinalysis examinations, and gross and microscopic
pathological examinations of tissues and organs. Extensive mortality
was observed at the high-dose level within the first few weeks of the
experiment. At the end of the third week of treatment, the highest-
dose group was terminated. There was no substantial mortality at 3000
or lower. Food intake and growth were reduced over the course of the
study in a dose-dependent fashion in all dose groups. Apart from the
mortality noted at the high dose level, no clinical signs of toxicity
or adverse behaviour were observed. Auditory and ophthalmological
examinations showed no evidence of loss of these functions in any of
the animals examined. Haematological examination indicated haemolytic
anaemia in both sexes of all treatment groups; characterized by a
reduction in haemoglobin concentration, erythrocyte count and packed
cell volume, and an increase in methaemoglobin level. Heinz bodies
were observed at 3000 mg/kg only. In addition, at 1500 mg/kg and above
there was a slight reticulocytosis and reduced partial thromboplastin
time in these dose groups. Changes in the clinical chemistry
parameters were noted at both 1500 and 3000 mg/kg. Gross examination
of certain tissues and organs showed changes in absolute weights
and relative weight ratios at all dosage levels. These reductions
appeared to follow a dose-dependent relationship. Animals administered
6000 mg/kg showed atrophy of the thymus and spleen within the first
three weeks of the test. Liver changes were noted in all dose groups
characterized as hyperplasia of the bile duct epithelium and changes
in the distribution of lipid. At the highest dose level, hyperplasia
of the urinary bladder epithelium and testes was noted. About half the
animals of both sexes in the 6000 mg/kg group showed an increase in
the mitotic index in hepatocytes.
In a study of 4-chloro- o-toluidine by Suter et al. (1976b),
groups of rats (30/sex/group; Tif/RAI strain) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 0, 750, 1500,
3000 and 6000 mg/kg for 60 days. There was no mortality over the
course of the study and clinical signs of toxicity were not observed.
Ophthalmological examinations did not suggest changes related to the
presence of 4-chloro- o-toluidine in the diet. Growth was reduced at
dietary levels of 1500 mg/kg and above. Haemolytic anaemia in both
sexes of all treated groups was characterized by a variety of
haematological changes, including reduced haemoglobin content, reduced
haematocrit content, reduced blood cell count, increased
methaemoglobin content, Heinz body formation, reticulocytosis and
polychromatophilia. In the highest-dose group, an increased number
of immature red blood cells (normoblasts) were observed. An increased
leucocyte count and prothrombin time was recorded at 3000 and
6000 mg/kg. Total protein was slightly reduced at 3000 and 6000 mg/kg
and there was a shift in the globulin content as observed by
electrophoresis. Plasma �-glutamyl transpeptidase of males and
alkaline phosphatase of females was increased at 6000 mg/kg.
Urinalysis was not significantly affected. In all treated animals, the
liver showed an increase in size accompanied by hypertrophy of the
hepatocytes. In the two highest-dose groups, the spleen was enlarged
and microscopic examination showed pronounced congestion and
haemorrhage. In the highest-dose group, slight or moderate
proliferation of the transitional cell epithelium was noted in the
urinary bladder.
7.2.1.3 Dog
In a study with chlordimeform by Blackmore (1969a), four groups
of beagle dogs were fed a dry diet containing either 0 mg/kg (10/sex),
250 mg/kg (8/sex), 500 mg/kg (8/sex) or 1000 mg/kg (10/sex) of
chlordimeform for 2 years. Two male and two female dogs were
sacrificed from each group at 26 and 52 weeks. Body weight was reduced
at 1000 mg/kg, the effect being slightly more pronounced in the
females. Total leucocyte counts were sporadically elevated in both
sexes at 1000 mg/kg and in females at 500 mg/kg. Haematocrit,
haemoglobin and erythrocyte counts tended to be depressed after 2
years in both sexes at 1000 mg/kg. Sporadic slight decreases in serum
albumin were observed, more frequently in males, at 1000 mg/kg.
Terminal spleen-to-body weight ratio was elevated in males at 500 and
1000 mg/kg, and in females at 1000 mg/kg. Histopathological
examinations revealed bile duct hyperplasia, pericholangitis and
nodular hepatocytic hyperplasia at 500 and 1000 mg/kg in both sexes,
and nodular hepatocytic hypertrophy at 1000 mg/kg in both sexes in the
liver. Kidneys showed an increased amount of pigmentation at 500 and
1000 mg/kg in both sexes.
7.2.2 Intubation
7.2.2.1 Rat
Four groups of 10 male and 10 female rats were intubated six
times weekly for one month with 5 ml/kg body weight of a 2% solution
of carboxymethylcellulose containing chlordimeform base at
concentrations such as to give dose levels of 0, 25, 50 or 100 mg/kg
(FAO/WHO, 1972). Body weight was markedly reduced in both sexes at
100 mg/kg. Hyperexcitability was observed in all test animals. At
100 mg/kg, this was apparent 20-30 min after dosing, and was followed
2 to 3 h after dosing by decreased activity and apathy. Recovery was
complete at 4 h. Similar but reduced effects were observed at 50 and
25 mg/kg, and with inconsistent frequency.
7.3 Long-term dietary exposure
7.3.1 Mouse
While there have been a number of long-term studies in mice with
chlordimeform and its metabolites, these were specifically designed to
study carcinogenic potential and are described in section 7.7.1.
7.3.2 Rat
In a study with chlordimeform by Blackmore (1969b), groups of
rats (35/sex/group) were fed a diet containing 0, 100, 250, 500 or
1000 mg/kg chlordimeform for 2 years. The 100 mg/kg group commenced
treatment 7 weeks after the other groups. This group was originally
part of the control group. Animals at that time were of similar weight
to those that had already been on test. The 1000 mg/kg group was
discontinued at 3 months due to severe growth inhibition. Growth
inhibition was observed in the males at 500 and 1000 mg/kg. In the
females, weight gain was reduced at 250 mg/kg and above. In addition,
female body weight gain was reduced at 100 mg/kg between weeks 20 and
48. Food intake was significantly reduced at 500 and 1000 mg/kg in
both sexes. Dose-related decreases in haematocrit, haemoglobin, and
erythrocyte counts, and a dose-related increase in the leucocyte count
occurred in females at 250 and 500 mg/kg up to one year. During the
second year, haematocrit only was consistently depressed in females at
500 mg/kg. Histopathological changes in the liver (nodules, and foci
of hyperplasia of hepatocytes) occurred in all groups, but the
incidence was greater at 250 and 500 mg/kg and was more severe at
500 mg/kg. Some females at 500 mg/kg showed slight hypertrophy and
vacuolation of focal groups of cells in the adrenal cortex. Terminally
organ to body weight ratios were increased in the liver (females at
250 and 500 mg/kg and males at 100 and 250 mg/kg), kidney (females at
250 and 500 mg/kg), thyroid (females at 250 and 500 mg/kg), heart
(males at 250 mg/kg and females at 500 mg/kg), adrenals (males at 100
and 250 mg/kg) and testes (100 and 500 mg/kg).
In a study with chlordimeform by Zak et al. (1973), groups of
rats (25/sex/group) were fed a diet containing chlordimeform at
concentrations of 0, 50, 75, 100, 250 and 500 mg/kg for one year. Food
consumption and weight gain data were recorded through the study.
Terminal organ weights and gross and microscopic examinations of
tissues and organs were the only other parameters reported. The
500 mg/kg group was terminated at 37 weeks after 10 males and 8
females had died. At the conclusion of the study, there was
considerable mortality noted in all groups. Food consumption was
decreased at 500 mg/kg in both sexes and was slightly reduced at
100 mg/kg and above in males only. This reduced food consumption was
not significantly reflected in the growth curves of males and females.
Gross examination did not show any compound-related abnormalities.
Organ weights and organ/body weight or organ/brain weight ratios did
not differ from control values. Histological examinations of liver and
spleen were performed on all animals. There were no significant
differences from control values with respect to fatty changes and
inflammatory changes in the liver. Slight proliferation of the bile
duct was more frequent in female rats treated with 500 mg/kg than in
the rats of other treated groups or the control group. Results of
examinations of the spleen for haemosiderosis suggested that, while
this condition was more pronounced in females, there were no
significant differences from control values.
In a study by Sachsse et al. (1980c), rats (90/sex/group;
Tif: RAIf strain) were fed a diet containing chlordimeform at
concentrations of 0, 2, 20, 100 or 500 mg/kg for 24 months. This was
equivalent to dosage levels of 0, 0.1, 1.0, 5.0 and 24 mg/kg body
weight per day for males and 0, 0.1, 1.2, 6.0, and 28 mg/kg body
weight per day for females. At the conclusion of the dietary feeding
study, all remaining rats were fed control diets for a period of time
until a survival rate of 20% per sex (10 rats) per group was attained,
at which time the animals were killed and examined. Groups of 20 male
and 20 female rats per group were examined periodically (4, 13, 26,
52, 78 and 104 weeks) for clinical laboratory investigations including
haematology, blood chemistry and urinalysis. Groups of 10 animals/sex/
group were sacrificed at 27 and 52 weeks for gross and microscopic
examination of tissues and organs. At the conclusion of the study, all
animals sacrificed (also those that died prior to the termination)
were examined for gross and microscopic pathology. There was no
mortality in the study attributable to the presence of chlordimeform.
Growth and body weight were maintained in all groups with the
exception of the 500 mg/kg group, where growth in both sexes was
slightly retarded. There were no clinical signs of toxicity or
abnormal behaviour. Ophthalmological and auditory examinations,
performed at periodic intervals, revealed no adverse effects
attributable to chlordimeform. Methaemoglobinaemia was observed at
dose levels of 20 mg/kg diet and above. At week 4, both males and
females showed a slight, but statistically significant, increase in
methaemoglobin content. At weeks 13 and 26, this condition abated but
returned at the end of one year and was significant in both sexes at
the highest dose level for the remainder of the study. Changes in
several other blood chemistry parameters were observed at the highest
dose level. Heinz body formation generally associated with
methaemoglobinaemia was not observed at week 4, but at the end of year
one and thereafter Heinz bodies were observed at the highest dose
level. A slight but significant reduction in blood glucose
concentration was noted at the higher dose levels throughout a major
part of the study. Slight changes in urinalysis parameters were
observed in the highest dose group, including a slightly reduced
urinary volume and a slightly higher specific gravity. Ketonuria and
proteinuria were observed at the high dose level at the earliest
examination periods only and were not observed at 13 weeks and
thereafter. Gross pathology and organ weight measurements (provided
for 27, 52 and 106-week sacrifice intervals) did not show any
significant dose-related responses. Microscopic histopathological
analyses of tissues and organs (performed at weeks 27 and 52 and at
the termination of the study) indicated no significant changes
attributable to chlordimeform in the diet. Although numerous benign
and malignant tumours were observed in both treated and control
animals, the frequency and type of neoplasms, reported at 12 and 24
months with pathology analyses, were not dose-related nor were they
attributable to chlordimeform in the diet. Several inherent,
degenerative or inflammatory changes were attributed to disease,
common in older animals. There was no indication of carcinogenic
potential to rats as a result of the presence of chlordimeform in the
diet. Based on the haematological occurrence of methaemoglobinaemia,
the no-observed-effect level of chlordimeform for rats was 2 mg/kg
diet, corresponding to an intake of 0.1 mg/kg body weight per day.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980d), groups of Tif:RAIf rats (90/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of
0, 2, 20, 100, or 500 mg/kg for 2 years. This corresponded to dietary
intakes of 0, 0.1, 1.0, 5 or 30 mg/kg body weight per day for females
and 0, 0.1, 1.0, 4.0 or 24 mg/kg body weight per day for males. Groups
of 10 males and 10 females were killed at periodic intervals (26 and
52 weeks) for examination of gross and microscopic pathology. Complete
haemato-logical, clinical chemistry, and urinalysis examinations were
performed at 4, 13, 26, 52 and 78 weeks on 20 males and 20 females of
each group. At 24 months, 20 males and 20 females were killed and
examined for clinical laboratory parameters and gross pathology. The
remaining animals were fed control diets for additional periods of
time until a survival rate of 20% per sex per group was attained.
At that time the remaining animals were killed and examined
microscopically for patho-logical changes, especially neoplastic and
non-neoplastic lesions. In the high-dose group, food intake and growth
were affected over the course of the study and slight growth
retardation was observed. Clinical signs of toxicity or adverse
behaviour were not observed. There was no mortality in the study
attributable to the presence of N-formyl-4-chloro- o-toluidine.
Ophthalmological examinations and auditory tests were normal. The
results of the haematological investigation showed haemoglobin
concentration to be slightly, but significantly, below that of the
controls in both male and female rats at the two highest dose levels.
In addition, slight but significant decreases in the erythrocyte count
and packed cell volume, a slight increase in reticulocytes and
somewhat higher methaemoglobin values were also seen in both male and
female rats at 500 mg/kg. With the exception of lower body weights of
the animals at the highest concentration, the most obvious change was
a significant increase in absolute and relative liver weights in both
sexes, but more pronounced in females, in the 500 mg/kg group. A
significantly increased incidence of hyperplasia of small biliary
ducts was seen in the liver of rats of the 500 mg/kg dose group. In
rats of the 500 mg/kg group that were killed after 2 years or died
after 12 months, a marked increase in the frequency of multioculated
cholangiogenic biliary cysts in the liver was noted. Both of these
finding were more pronounced and more frequent in female than in male
animals. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of neoplasms was
not treatment-related. All gross and histopathological lesions and
changes seen in both control and test animals were described as
inherent, degenerative or inflammatory in origin and were attributed
to naturally occurring diseases common in aged rats. There was no
indication of oncogenic potential in rats as a result of the presence
of N-formyl-4-chloro- o-toluidine. On the basis of the minor
haematological changes, the no-observed-effect level in this study was
20 mg/kg diet, corresponding to an intake of 1 mg/kg body weight per
day.
In a study with 4-chloro- o-toluidine by Sachsse et al. (1980e),
groups of Tif:RAIf rats (90/sex/dose level) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 or 500 mg/kg
for two years. This corresponded to dietary levels of 0, 0.1, 1.0, 5.0
or 28 mg/kg body weight per day for females, and 0, 0.1, 1.0, 4.6 or
24.6 mg/kg body weight per day for males. Groups of 10 males and 10
females were killed at periodic intervals (27 and 54 weeks) for gross
and microscopic pathological examinations. Complete haematological,
clinical chemistry and urinalysis examinations were performed at 4,
13, 26, 52, and 78 weeks on 20 females and 20 males of each group. At
24 months, 20 males and 20 females were killed and examined for
clinical laboratory parameters. Several animals were examined for
gross pathology. The remaining animals were fed control diets for
additional periods of time until a survival rate of 20% per group was
attained. At that time, the remaining animals were killed and examined
for microscopic pathology and oncogenic response. A complete
microscopic analysis was made on at least 10 rats of each sex of each
group at the termination of the experiment. All rats dying during the
course of the study were examined for tumours or neoplasms. In the
high-dose group of female rats, food intake and growth were affected
over the course of the study and slight growth retardation was
observed. There was no effect on growth in male rats at any dose
level. Clinical signs of toxicity were not observed. There was no
mortality in the study attributable to the presence of 4-chloro- o-
toluidine in the diet. Ophthalmological examinations and auditory
tests did not reveal changes that were related to the administration
of 4-chloro- o-toluidine. The results of the haematological
investigation, blood chemistry data and the urinalysis were similar
for both treated and control rats. Periodically, the haemoglobin
concentration was slightly but significantly below that of the
controls in the female rats at 100 mg/kg diet and above. Slight but
significant decreases were observed in the erythrocyte count and
packed cell volume in the female rats at 500 mg/kg. Marginal
reticulocytosis was also found to occur at 500 mg/kg in the female
rats at week 13 and in both sexes at week 26. In both male and female
rats at 500 mg/kg, the methaemoglobin level was found to be slightly
though significantly increased when compared to controls.
Periodically, this change was observed in the females of the 100 mg/kg
dose group, and, occasionally, Heinz bodies were also observed in
female rats. There were some changes to organ weights, organ-to-body
weight ratios and organ-to-brain ratios that were statistically
significant, but only the increase in absolute and relative liver
weights were dose-related. In rats from the 500 mg/kg dose group only,
a slightly but significantly increased incidence of multilobular
cholangiogenic cysts was observed in the liver. These biliary cysts
were found in 10/89 female and 3/90 male rats from the 500 mg/kg
group, compared to 4/89 female and 0/90 male rats in the control
group. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of the neoplasms
occurring in these animals was not treatment-related. Gross and
histopathological lesions and changes seen in both control and treated
animals were described as inherent, degenerative or inflammatory in
origin, and were attributed to naturally occurring diseases, common in
aged rats. There was no indication of oncogenic potential in rats as a
result of the presence of 4-chloro- o-toluidine in the diet. On the
basis of minor haematological changes, the no-observed-effect level in
this study was 20 mg/kg diet, corresponding to a dietary intake of
1 mg/kg body weight per day.
7.4 Skin and eye irritation; skin sensitization
Potential skin irritation was assessed by the application of
0.5 g chlordimeform or its hydrochloride salt to the shaved skin of
six male rabbits. When evaluated at 24 and 72 h, both compounds
produced a very slight irritation (FAO/WHO, 1972).
Potential eye irritation was assessed by application of 0.1 ml of
chlordimeform to one eye of each of nine rabbits, followed by
assessment over 7 days. All animals exhibited slight conjunctival
redness, while one showed slight chemosis. All effects had reversed
within 7 days. There was no evidence of corneal damage. Chlordimeform
may be considered a slight eye irritant (FAO/WHO, 1972).
There were no studies performed to assess the potential for
chlordimeform to cause skin sensitization.
7.5 Reproductive toxicity, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
7.5.1.1 Rat
Four groups of 10 male and 20 female rats were fed a diet
containing 0, 100, 250 and 500 mg/kg chlordimeform in corn oil during
three parental and three two-litter filial generations. Parental body
weight prior to mating tended to be reduced in all treatment groups,
especially at the highest dose level. The same tendency was apparent
with regard to food consumption. The fertility index, gestation index,
live birth index, sex ratio, mean litter size and birth weight of pups
were comparable between treatment and control groups in all
generations. At the 500 mg/kg dose level, the lactation index was
reduced in Fla, Flb and F3a litters. Weaning weight of offspring was
depressed in all high-dose litters. Gross pathological examinations
were performed on parents and pups dying during the study, and on 10
male and 10 female weanlings of the F3b generation. No compound-
related effects were noted in the pathological examination (Blackmore,
1969c).
In a study by Goldman et al. (1991), treatment of ovariectomized
Long-Evans rats with a single intraperitoneal injection of
chlordimeform at dose levels of 25 or 50 mg/kg caused a complete
suppression of luteinizing hormone surge. The observed suppression did
not persist beyond the day of treatment. In a more recent study by
Cooper et al. (1994), the effect of this delay in hormone surge on
pregnancy outcome in females was examined. Chlordimeform at a dose
level of 50 mg/kg resulted in a delay in breeding as well as a
significant reduction in litter size.
Adult male Sprague-Dawley rats were administered chlordimeform by
gavage at 200 mg/kg body weight on one day or 50 mg/kg body weight per
day for 5 days. Rats were killed on either 3 or 13 days after the last
dose. Body weights were reduced at the earlier time points only. There
were no changes in the weights of the testes or associated organs.
Caudal sperm counts were reduced after the single dose only. No other
changes were observed, including sperm motility, velocity or
morphology (Linder et al., 1992).
7.5.1.2 Hamster
Goldman et al. (1993) reported that a single intraperitoneal dose
of chlordimeform (75 mg/kg and above) is capable of delaying the
luteinizing hormone surge and altering the timing of oocyte release in
the hamster. The reproduction consequences of this effect were not
investigated.
7.5.2 Embryotoxicity and teratology
7.5.2.1 Rat
Groups of pregnant rats (25/treatment group, 30 controls) were
administered chlordimeform in carboxymethylcellulose at dose levels of
0, 10, 25 or 50 mg/kg body weight per day from days 6 to 15 of
pregnancy. Only a slight reduction in feed intake was noted at the
intermediate dose level. At the high dose level, dams showed
somnolence through the first 3 days of treatment. There was also a
reduced body weight gain and decrease in feed consumption at this dose
level. Examination of fetuses removed by caesarean section on day 21
showed there was a slight delay in growth of the fetuses at the two
highest dose levels. This effect was probably a direct result of the
toxic response in the dams. No teratogenic events were observed in the
offspring, although an increased incidence of sternal ossification
defects occurred at 25 mg/kg body weight (Fritz, 1975).
7.5.2.2 Rabbit
Three groups of 10 impregnated female New Zealand white rabbits
were administered chlordimeform by gavage on days 8 to 16 of gestation
at dose levels of 0, 7.5 or 30 mg/kg body weight per day. Five rabbits
per group were killed on day 28 of gestation. Parental mortality,
abortion rate, corpora lutea to implantation ratio, litter size,
incidence of resorption, stillbirths, fetal weight, fetal length, and
incidence of skeletal and tissue abnormalities were unaffected by the
test compound. In the remaining rabbits, which were allowed to litter
normally, gestation length, litter size and litter weights were
similar in both treated and control groups (Blackmore, 1969d).
Groups of rabbits (group size ranged from 17 to 38 dams per
group) were given chlordimeform orally from days 6 to 18 of pregnancy
at dose levels of 0, 10, 30 and 100 mg/kg body weight per day. Fetuses
were removed by caesarean section on day 28 of pregnancy. The
administration of chlordimeform at 100 mg/kg body weight produced a
distinct adverse effect on dams for 2-3 h for the first 4 days of
treatment. Examination of dams and fetuses at 28 days suggested that
the low dose had no teratogenic or embryotoxic effect. In the
intermediate and high dose groups, the implantation to corpora lutea
ratio was found to be reduced compared to controls. In the high dose
group, the number of incompletely ossified sternebrae showed a slight
increase over that observed in the controls and in the other groups.
In addition, the number of fetuses with malformations was slightly
increased at 100 mg/kg. These malformations included a median cleft
palate and exencephaly and an omphalocele. Further examination of
spontaneous malformations observed in a cumulative control of 2495
rabbit fetuses suggested that these abnormalities may be spontaneous
and not a consequence of the administration of chlordimeform (Fritz,
1971).
7.6 Mutagenicity and related endpoints
Referenced summaries of the test results with chlordimeform,
N-formyl-4-chloro- o-toluidine, and 4-chloro- o-toluidine are
given in Tables 8, 9 and 10, respectively. The important features of
these data are given below.
7.6.1 DNA damage and repair
Chlordimeform gave no evidence of unscheduled DNA synthesis in
rat hepatocytes (dose levels: 5-625 �g/ml) or in human fibroblasts
(dose levels: 2-250 �g/ml). 4-Chloro- o-toluidine, on the other hand,
gave a slight to moderate dose-related increase in the mean number of
silver grains per nucleus in rat hepatocytes over a dose range of
0.625-78.15 �g/ml, but not in human fibroblasts over the dose range of
1.25-156.25 �g/ml.
Table 8. Summary of mutagenicity and related end-point studies on chlordimeform HC1
Organism Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976a);
TA100 +/-S9 - Konopka & Heymann (1977);
TA1535 +/-S9 - Muecke et al. (1979);
TA1537 +/-S9 - Rashid et al. (1984)
TA1538 +/-S9 -
Salmonella typhimurium TA98 - Arni & M�ller (1983a)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983c)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
WP67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Insects Sex-linked recessive lethals Drosophila +a,b Kale et al. (1995)
Mammalian cells Gene mutation Mouse lymphoma L5178Y- TK+/-/ - Beilstein & M�ller (1984a)
in vitro +/-S9
Unscheduled DNA synthesis Rat hepatocytes - Puri & M�ller (1983a)
Unscheduled DNA synthesis Human fibroblasts - Puri & M�ller (1983c)
Mammalian cells Cell transformation Mouse BALB/3T3 cells +a,b Beilstein & M�ller (1983)
in vitro
Table 8. (con't)
Organism Test Test system Strain +/- References
Mammals Testicular cell chromosome Mouse spermatocytes - Hool et al. (1983)
damage Mouse spermatocytes - Arni et al. (1983a)
Micronucleus assay Chinese Hamster bone - Langauer & M�ller (1977)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & M�ller (1978)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983a)
marrow cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz (1978a)
a Chlordimeform formulation
b Not dose-related
Table 9. Summary of mutagenicity and related end-point studies on N-formyl-4-chloro-o-toluidine
Organism Test Test Systems Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976c);
TA100 +S9 + Konopka & Heymann (1977);
TA100 -S9 - Muecke et al. (1979);
TA1535 +/-S9 - Rashid et al. (1984)
TA1537 +/-S9 -
TA1538 +/-S9 -
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y +a Strasser & M�ller (1984b)
vitro
Mouse lymphoma L5178Y / -a Strasser & M�ller (1983b)
host-mediated assay
Mammals Testicular cell chromosome Mouse spermatogonia - Arni (1983b)
damage Mouse spermatocytes +b Arni & M�ller (1983e)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978a)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983f)
marrow metaphase cells
Table 9. (con't)
Organism Test Test Systems Strain +/- References
Mammals Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978b)
a No positive control
b Chromosome aberrations; not dose-related
Table 10. Summary of mutagenicity and related end-point studies on 4-chloro-o-toluidine
Organisms Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +S9 + Arni & M�ller (1976b);
TA98 +S9 - Haworth et al. (1983);
TA98 -S9 + Konopka & Heymann (1977);
Haworth et al. (1983);
TA100 +S9 - Meuke et al. (1979);
TA100 +S9 - Haworth et al. (1983)
TA100 -S9 - Rashid et al. (1984);
TA1535 +/-S9 - Haworth et al. (1983)
TA1537 +/-S9 -
TA1538 +/-S9 -
S. typhimurium TA98 - Arni & M�ller (1983b)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983d)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y/TK+/- -S9 - Beilstein & M�ller (1984b)
vitro +S9 +
Mouse lymphoma L5178Y + Strasser & M�ller (1984a)
Mouse lymphoma L5178Y - Strasser & M�ller (1983a)
/host-mediated
Table 10. (con't)
Organisms Test Test system Strain +/- References
Mammalian cells in Unscheduled DNA synthesis Rat hepatocytes + Puri & M�ller (1983b)
vitro
Human fibroblasts - Puri & M�ller (1983d)
DNA strand breakage V79 cells + Zimmer et al. (1980)
Cell transformation Mouse BALB/3T3 cells + Beilstein & M�ller (1984c)
Mammals Testicular cell chromosome Mouse spermatogonia - Hool & Arni (1983b)
damage Mouse spermatocytes - Hool & Arni (1983c)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978b)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983d)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983e)
marrow
Sister chromatid exchange Chinese hamster ovary + Galloway et al. (1987)
cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse + Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978)
N-Formyl-4-chloro- o-toluidine was not directly tested for its
ability to induce DNA damage and repair.
The macromolecular binding of 4-chloro- o-toluidine to macro-
molecules of rat and mouse liver has been investigated by several
authors. In a report by Hill et al. (1979), the binding of
4-chloro-2-[methyl-14C]-methylaniline (4-chloro- o-toluidine)
in vivo and in vitro was investigated. The major binding
in vivo was in the liver. In vitro binding was dependent on
the presence for microsomal preparations and NADPH. Two soluble
products of microsomal enzymes were identified as 5-chloro-2-
hydroxylaminotoluene and 4,4'-dichloro-2,2'-dimethylazobenzene. The
hydroxylamino compound appeared to be the more activated form of
4-chloro- o-toluidine.
4-Chloro- o-toluidine caused DNA strand breaks in Chinese
hamster V79 cells (Zimmer et al., 1980).
In studies by Bentley et al. (1986a,b), the covalent binding
of [14C- ring]-4-chloro- o-toluidine to mouse and rat liver
macromolecules was compared. After a single administration to either
species, the extent of binding decreased in the order: protein>RNA>
DNA. The level of binding to mouse liver DNA was approximately twice
as high as the binding to rat liver DNA after either single or
repeated administration, while the binding to liver RNA and protein
was greater in the rat. There was no evidence of an increased level
of incorporation of [3H]-thymidine into DNA as a result of
4-chloro- o-toluidine binding. Two major hydrophobic DNA adducts
were formed in both species, and one of these was formed to a much
greater extent (6-30 fold) in mice.
7.6.2 Mutation
The ability of chlordimeform and its metabolites to induce
mutations has been investigated in both microbial and mammalian
systems.
Chlordimeform itself gave uniformly negative results in
Salmonella typhimurium (0.1-2000 mg/ml), Saccharomyces cerevisiae
(15-400 mg/ml), and Escherichia coli (250-2000 mg/ml), with or
without S9 microsomal activation, as well as in a thymidine kinase
mutation assay in mouse lymphoma L5178Y/TK+/- cells with
(75-3000 mg/ml) or without microsomal activation (42.5-1700 mg/ml).
Kale et al. (1995) reported that a chlordimeform formulation diluted
to provide a dose level of 10 000 �g/ml is a potent sex-linked
recessive mutagen in male pre-meiotic and meiotic cells of Drosophila.
N-Formyl-4-chloro- o-toluidine was also negative in all
Salmonella typhimurium assays (0.1-1000 �g/ml) except for TA100 with
microsomal activation, in which there was a dose-related increase in
revertants. All Escherichia coli assays (250-2000 �g/ml) were
negative. In a forward mutation assay in mouse lymphoma L5178Y cells
(213 & 640 �g/ml), N-formyl-4-chloro- o-toluidine gave a positive
result in two out of three 18-h incubation experiments. In a host-
mediated experiment with mouse lymphoma L5178Y cells (300 mg/kg), the
result was negative.
4-Chloro- o-toluidine was negative in all assays with
Salmonella typhimurium (10-2000 �g/ml) except for TA100 with S9
microsomal activation and TA98 with S9 microsomal activation. Assays
with Saccharomyces cerevisiae (3.2-90 �g/ml) and with Escherichia
coli (250-2000 �g/ml) were negative. In a thymidine kinase forward
mutation assay in mouse lymphoma L5178Y/TK+/- cells, 4-chloro- o-
toluidine was negative without S9 microsomal activation (31.25-
500 �g/ml) and positive with microsomal activation (37.5-600 �g/ml).
In a separate forward mutation assay in mouse lymphoma L5178Y cells
(111 & 255 �g/ml), a positive result was obtained in one out of three
18-h incubation experiments. In a host-mediated experiment with mouse
lymphoma L5178Y cells (330 mg/kg), the result was negative.
7.6.3 Chromosome damage
Sister chromatid exchange assays in Chinese hamster bone marrow
cells were conducted following oral treatment with chlordimeform
(31-324 mg/kg) and 4-chloro- o-toluidine (100-400 mg/kg). In both
cases, the result was negative. Similarly, in an assay for chromosome
aberrations in Chinese hamster bone marrow metaphase cells, a negative
result was obtained following oral treatment with chlordimeform
(2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine (2 � 300-
1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-800 mg/kg), although
the results were somewhat erratic. A micronucleus test in Chinese
hamster bone marrow interphase cells following oral treatment with
chlordimeform (2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine
(2 � 300-1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-400 mg/kg)
was also negative.
Testicular cell chromosomal damage was assessed in mouse
spermatocytes and spermatogonia. To investigate the potential
cytogenetic effects on mouse spermatogonia, chlordimeform
(9-66 mg/kg), N-formyl-4-chloro- o-toluidine (80-320 mg/kg) or
4-chloro- o-toluidine (85-500 mg/kg) was administered orally on 5
consecutive days. The results were negative in each case. To
investigate the potential cytogenetic effects on mouse spermatocytes,
chlordimeform (18-72 mg/kg), N-formyl-4-chloro- o-toluidine
(80-320 mg/kg), or 4-chloro- o-toluidine (85-500 mg/kg) was
administered orally over 10 days on days 0, 2, 3, 5 and 9. The results
were negative in the case of both chlordimeform and 4-chloro- o-
toluidine, but non-dose-related evidence of chromosome damage was
indicated from the results with N-formyl-4-chloro- o-toluidine.
The heritable translocation assay, dominant lethal test, and
mammalian spot test, each of which might indicate minor or major
genomic changes, were conducted on all three compounds. In the
heritable translocation assay, chlordimeform (120 mg/kg/day),
N-formyl-4-chloro- o-toluidine (100 mg/kg/day) or 4-chloro- o-
toluidine (200 mg/kg/day) was administered orally for 49 days. No
induction of translocation heterozygosity was found.
In the dominant lethal assay, chlordimeform (22 or 66 mg/kg),
N-formyl-4-chloro- o-toluidine (105 or 315 mg/kg) or 4-chloro- o-
toluidine (110 or 330 mg/kg) was administered orally as a single dose.
There was no evidence of any dominant lethal effects in the progeny of
male mice.
In the mammalian spot test, chlordimeform (160 mg/kg),
N-formyl-4-chloro- o-toluidine (100 mg/kg) or 4-chloro- o-
toluidine (100 mg/kg) was administered orally on days 8-10 of
embryonic development. The results were negative except in the case of
4-chloro- o-toluidine, which induced a 3.2% incidence of spots of
genetic relevance compared to an incidence of 0.9% in controls.
7.6.4 Cell transformation
Cell transformation assays conducted with both chlordimeform and
4-chloro-toluidine in mouse BALB/3T3 cells produced an increased
incidence of transformed cell colonies with both compounds. With
chlordimeform, the experiment was conducted at dose levels up to
1 �g/ml, and increased transformation frequency was observed only at
0.0625 and 0.125 �g/ml. The transformative properties of chlordimeform
were considered weak. With 4-chloro- o-toluidine, three experiments
were conducted at dose levels up to 36 �g/ml, and a significant
increase in transformation frequency was observed. The transformative
properties of 4-chloro- o-toluidine were considered definite.
7.7 Carcinogenicity
A number of carcinogenicity studies have been conducted in mice.
With chlordimeform, there are two dietary studies and one dermal
study. With N-formyl-4-chloro- o-toluidine, there is one dietary
study. With 4-chloro- o-toluidine, there are four dietary studies.
In rats, the carcinogenic potential of chlordimeform and its
metabolites was generally investigated as part of more detailed
long-term studies, and details are provided in section 7.3.1.2. Three
studies on 4-chloro- o-toluidine that primarily investigated
carcinogenicity are reported below.
7.7.1 Mouse
The carcinogenicity of chlordimeform has been examined in two
dietary studies and in one dermal study.
In a study by Suter et al. (1978), groups of mice (50/sex/group;
Tif: MAG strain, SPF derived) were fed a diet containing chlordimeform
at concentrations of 0, 20, 100 and 500 mg/kg for 24 months. At the
conclusion of the dietary feeding interval, animals were maintained on
control diet until 90% of a group had died, at which time the
remaining animals of the group were sacrificed. There were no signs of
acute toxicity related to chlordimeform in the diet over the course of
the feeding trial. Growth and food consumption were similarly
unaffected by the presence of chlordimeform in the diet. Mortality was
significantly increased in females after 60 weeks at 500 mg/kg, and
after 90 weeks at both 100 and 500 mg/kg. In males, significantly
increased mortality was observed after 70 weeks at 500 mg/kg, and
after 110 weeks at both 100 and 500 mg/kg. However, lifespan was not
significantly affected in males at 100 mg/kg. The animals fed dietary
levels of 100 mg/kg and above displayed an increased incidence of
haemorrhagic tissue masses in subcutaneous tissues, retroperitoneum
and some internal organs (kidney, liver and spleen), which upon
examination were classified as malignant haemangioendotheliomas. These
malignancies which were reported to occur rarely in control
populations were found predominantly in the 100 and 500 mg/kg dietary
groups (see Table 11). In some animals the tumours were of multiple
origin and metastases were observed in the lungs. There were no other
types of neoplasm observed in the study that were attributable to
chlordimeform in the diet. Under the conditions of this study,
20 mg/kg in the diet appeared to be a no-observed-effect level.
In a study by Li et al. (1985a), groups of Swiss mice (50/group,
sex not stated) were fed a diet containing chlordimeform at
concentrations of 0, 20, 100, or 300 mg/kg for a period of 18 months.
A positive control group was administered 300 mg/kg of 4-chloro- o-
toluidine in the diet for 18 months. All animals were killed at the
end of the study and assessed for tumour formation. The main results
of the study are presented in Table 12. The author described the
majority of the neoplasms as angiomas, and the malignant neoplasms as
angiosarcomas. These neoplastic changes were considered to be similar
to those observed in the study by Suter et al. (1978).
In a paper by Jiang et al. (1988), the dermal carcinogenicity of
chlordimeform was investigated in mice. Groups of Swiss mice (50 per
dose level, sex not stated) were treated dermally with chlordimeform
twice per week at dose levels of 0, 100, 500, 2000 or 4000 mg/kg body
weight for a total of 17-20 treatments, together with croton oil (0.5%
in acetone). Positive controls received coal tar pitch (20 treatments)
plus croton oil (30 treatments). All animals were sacrificed after 18
months and analysed for tumour formation. Chlordimeform induced both
skin and liver tumours in this assay (see Table 13). The changes
observed in the skin consisted of epidermal hyperplasia, papillomas
and squamous cell carcinomas. The effect of croton oil application was
Table 11. Incidence of haemangioendotheliomas in mice following dietary administration
of chlordimeform, N-formyl-4-chloro-o-toluidine or 4-chloro-o-toluidine
(Suter et al., 1978; Sachsse et al., 1978a,b)
Control Dietary concentration (mg/kg)
2 20 100 500
Chlordimeform HCl
Male 1/44 (2%) - 0/44 (0%) 15/49 (30%) 39/48 (83%)
Female 1/43 (2%) - 2/46 (4%) 22/46 (50%) 35/49 (71%)
Total 2/87 (2%) - 2/90 (2%) 37/95 (41%) 74/97 (80%)
N-formyl-4-chloro-o-toluidine
Male 0/46 (2%) - 0/49 (0%) 15/48 (38%) 40/47 (89%)
Female 0/47 (0%) - 0/47 (0%) 23/43 (56%) 38/48 (79%)
Total 1/93 (1%) - 0/96 (0%) 38/91 (47%) 78/95 (84%)
4-Chloro-o-toluidine
Male 0/50 (0%) 0/47 (0%) 4/48 (8%) 23/47 (48%) 40/48 (83%)
Female 1/45 (7%) 1/45 (2%) 3/48 (6%) 30/47 (62%) 34/49 (72%)
Total 1/95 (1%) 1/92 (1%) 7/96 (19%) 53/94 (66%) 74/97 (78%)
to shorten the latent period for tumour formation and also to hasten
the malignant progression of existing neoplasms in the skin. At
500 mg/kg body weight, the time of first appearance of tumours was 483
days without croton oil and 154 days with croton oil. The latency
period also decreased with increasing dose levels of chlordimeform. In
the liver, changes consisted of enlargement, hepatocytic hyperplasia,
and hepatocytic carcinomas.
In a carcinogenicity study by Sachsse et al. (1978a), groups of
mice (50/sex/group; Tif: MAG strain) were fed N-formyl-4-chloro- o-
toluidine in the diet at concentrations of 0, 20, 100 and 500 mg/kg
for 24 months. After this time, all animals were fed a control diet
until the study was concluded when 90% of the animals in a group had
Table 12. Incidence of tumours in mice after dietary administration of chlordimeform or 4-chloro-o-toluidine (Li et al., 1985a)
Dietary Number of animals Number of animals Incidence Number of animals Incidence Days to
concentration necropsied bearing haemangiomas (%) bearing (%) appearance of
(mg/kg diet) or haemangiosarcomas haemangiosarcomas neoplasm
Chlordimeform
0 50 0 0 0 0 -
20 50 8 16 0 0 494
100 50 22 44 5 10 469
300 50 36 72 15 30 448
4-Chloro-o-toluidine
300 50 31 62 13 26 283
Table 13. Incidence of tumours in mice following dermal application of chlordimeform (Jiang et al., 1988)
Group/treatment Skin Liver
Number Carcinomas Papillomas Hyperplasia Number Carcinomas Papillomas Hyperplasia
of animals (%) (%) (%) of animals (%) (%) (%)
Water 18 0.0 0.0 6.1 17 0.0 0.0 0.0
Croton oil alone 17 0.0 0.0 17.6 24 0.0 0.0 8.3
100 mg/kg + croton oil 19 0.0 5.3 21.1 21 23.8 0.0 9.5
500 mg/kg 22 4.6 4.6 18.2 20 25.0 0.0 0.0
chlordimeform alone
500 mg/kg + croton oil 23 4.4 4.4 52.2 25 8.0 0.0 4.0
2000 mg/kg + croton oil 15 20.0 20.0 26.7 14 14.3 0.0 0.0
4000 mg/kg + croton oil 15 60.0 13.3 13.3 16 18.8 6.2 0.0
Coal tar pitch 18 88.9 11.1 0.0 19 15.8 5.3 0.0
died. There was no sign of adverse behaviour, and acute mortality was
not noted. Growth and food consumption were unaffected. There were
significant differences noted in survival after one year of age. Both
males and females showed an increased mortality at 100 and 500 mg/kg
after approximately one year of feeding. The onset of increased
mortality occurred earlier in females. The females at the 20 mg/kg
dietary level showed a slightly higher, non-significant, mortality
during the same period. Detailed gross and microscopic examination of
a variety of tissues and organs showed the presence of numerous gross
anatomical lesions. There was an increased number of haemorrhagic
masses in the subcutaneous tissues in the retroperitoneum and in some
internal organs of mice at all treatment levels. Detailed microscopic
examination confirmed that the increased incidence of haemorrhagic
masses were malignant tumours of vascular origin. These tumours were
histologically classified as malignant haemangioendotheliomas (see
Table 11). In addition to the occurrence of tumours, the time to
tumour relationship was decreased as the dietary concentration was
increased. Other neoplasms occurring in the study were not considered
to be treatment-related. A no-observed-effect-level was not
demonstrated under the condition of this experiment.
The carcinogenicity of 4-chloro -o-toluidine has been examined
in four dietary studies.
An early study by Ezumi and Nakao conducted in 1974 was reviewed
by the JMPR in 1978 and considered inadequate (FAO/WHO, 1979).
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives described by Homburger et al. (1972) and Weisburger
et al. (1978), groups of CD-1 mice (25/sex/dose level) were
administered 4-chloro- o-toluidine in the diet at dose levels of 0,
750 or 1500 mg/kg for males, and 0, 2000 or 4000 mg/kg for females for
a period of 18 months. All mice were placed on a control diet for an
additional 3 months before sacrifice and complete necropsy and
histopathological examination of tissues. The incidence of
haemangioendotheliomas was increased in males at both low (12/20) and
high (13/20) dose levels compared to concurrent controls (0/14) and
historical controls (5/99), and in females at both low (18/19) and
high (12/16) dose levels compared to concurrent controls (0/15) and
historical controls (9/102).
In a study by Sachsse et al. (1978b), groups of mice
(50/sex/group; Tif: MAGf strain) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 and
500 mg/kg for 24 months. After 24 months, all animals were fed control
diets until the study was concluded when 90% of the animals in a group
had died. There were no overt signs of toxicity. Growth and food
consumption were unaffected by treatment. An adverse effect on
longevity (lifespan) was noted in both males and females at the two
highest dietary levels. At the conclusion of the study upon gross
examination there was a marked increase number of haemorrhagic masses
in subcutaneous tissue, in the retroperitoneum, and in some internal
organs. Microscopic examination revealed an increased incidence of
haemorrhagic malignant tumours of vascular origin at dose levels of
20 mg/kg and above (see Table 11). The tumour incidence in control
exceeded the incidence observed at 2 mg/kg. The tumours were
histologically classified as malignant haemangioendotheliomas and, on
occasion, metastases were observed. There was not only a significant
dose-dependent increase in the total incidence of malignant tumours
but the time to appearance of tumours occurred at a markedly earlier
date in animals at the higher concentrations than in those at the
lower concentrations. A benign variant of the haemangioma was observed
in all groups, and although without the characteristics of malignancy,
did cause local invasion. Thus, the benign and malignant tumours have
been grouped together. The vascular tumours (haemangiomas and
haemangioendotheliomas) of the type that occurred in the mice appeared
to be peculiar to this rodent species. The occurrence of other types
of neoplasms in the study was not influenced by the presence of
4-chloro- o-toluidine in the diet. Under the conditions of this
experiment, 2 mg/kg in the diet appeared to be a no-observed-effect
level.
In a study with 4-chloro- o-toluidine conducted by the National
Cancer Institute (NCI, 1979), groups of B6C3F1 mice (50/sex/dose
level) were administered 4-chloro -o-toluidine in the diet at dose
levels of 3750 or 15 000 mg/kg for males and 1250 or 5000 mg/kg for
females for 99 weeks. Control groups consisted of 20 males and 20
females. There was a dose-related increase in mortality in both sexes.
There was also a dose-related increase in the incidence of
haemangiosarcomas as shown in Table 14. The haemangiosarcomas
apparently originated in fatty tissue adjacent to the genital organs
and not in a particular organ. In some instances, they were observed
to infiltrate the abdominal muscles, uterus, ovary, prostate or
urinary bladder. The haemangiosarcomas were lethal to 89 (75%) of the
affected mice, owing to haemorrhage in the peritoneal cavity and to
the space-consuming character of the lesions. Pulmonary metastasis was
observed in only 5 (4%) of the 119 dosed animals bearing
haemangiosarcomas. Associated pathological alterations that were
recorded at necropsy were haemorrhage in the peritoneal cavity and
variable enlargement of the spleen. It was concluded that 4-chloro-
o-toluidine was carcinogenic in B6C3F1 mice.
In the study of Li et al. (1985a), a single dietary dose of
4-chloro- o-toluidine (300 mg/kg) was given to mice for 18 months as
a positive control. The incidence of tumours was similar to that seen
in mice receiving 300 mg/kg of chlordimeform, but the latency period
was considerably reduced (Table 12).
Table 14. Incidence of tumours in mice following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 3750 15 000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 20 50 50 18 49 50
Haemangiosarcomas 0 3 37 0 40 39
(0%) (6%) (74%) (0%) (82%) (78%)
Haemangioma 0 3 5 1 6 0
(0%) (6%) (10%) (6%) (12%) (0%)
7.7.2 Rat
The carcinogenicity of chlordimeform and its metabolites has
generally been considered as part of more detailed long-term studies
(see Section 7.3). In the studies below, carcinogenicity was the
primary consideration.
In a study conducted by the National Cancer Institute (NCI,
1979), groups of F344 rats (50/sex/dose level) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 1250 mg/kg or
5000 mg/kg for 107 weeks. Control groups contained 20 animals of each
sex. There was no significant dose-related trend in mortality in
either sex. There was a variety of neoplastic and non-neoplastic
changes in control and treated rats. There was a small increase in
adenomas of chromophobe cells of the pituitary gland in both male and
female rats compared to controls (see Table 15). All of these tumours
were benign, are also common in this strain of rat and have occurred
in 21% of control female rats in the NCI laboratories. The authors
concluded that on the basis of histopathological examination,
4-chloro- o-toluidine was not carcinogenic in F344 rats.
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives by Weisburger et al. (1978), groups of male Charles
River CD rats were administered 4-chloro- o-toluidine in the diet at
dose levels of 0, 2000 or 4000 mg/kg diet for the first 3 months,
which was then reduced to 0, 500 or 1000 mg/kg diet for the following
15 months. There was no statistically significant increase in tumours
in either of the treated groups.
Table 15. Incidence of tumours in rats following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 1250 5000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 19 48 47 19 48 48
Chromophobe 2 6 15 1 13 15
adenoma (11%) (13%) (32%) (5%) (27%) (31%)
Chromophobe 0 0 2 0 3 1
hyperplasia (0%) (0%) (4%) (0%) (6%) (2%)
7.8 Other special studies
7.8.1 Immunotoxicity
In a study by Wiltrout et al. (1978), the potential of various
pesticides to influence the primary humoral immune response in the
mouse with respect to both dose and time of exposure was examined.
Mice receiving a single oral dose of chlordimeform at approximately
the LD50 level (148 mg/kg body weight) experienced a significant
suppression of humoral response when the dose was administered on the
day of immunization or two days after immunization. No response was
observed at one tenth of the LD50 dose, even when administered for
8 or 28 days.
Further studies by Shopp et al. (1985) investigated the effect of
chlordimeform on both humoral and cell-mediated immunity in the mouse
following both acute and 14-day exposures by the intraperitoneal
route. There was a decrease in IgM antibody-forming (plaque-forming)
cells when measured 4 days after intraperitoneal administration at 20
or 40 mg/kg body weight per day. These dose levels did not result in
any alteration of cell-mediated immunity. When administered orally,
chlordimeform at doses as high as 120 mg/kg body weight per day did
not have any effect on the 4- or 5-day antibody response.
Immunological parameters that may be related to the carcinogenic
activity of chlordimeform in rats were investigated by Thomas et al.
(1990). These included spleen/body weight ratio, splenocyte viability,
T and B cell mitogenesis, natural killer (NK) cell and natural
cytotoxic (NC) cell activity. Chlordimeform was administered
intraperitoneally on three consecutive days at 0, 1, 10 or 75 mg/kg
body weight per day. 4-Chloro- o-toluidine was administered
intraperitoneally on three consecutive days at 0, 10, 50 or 100 mg/kg
body weight per day. Spleen/body weight changes were observed only at
the highest dose of chlordimeform. No changes were observed with
either chemical on splenocyte viability or T and B cell mitogenesis.
An inhibition of NC activity at all chlordimeform doses was observed,
and an inhibition of NK activity was observed at 10 mg/kg body weight
per day and above. The relevance of this result to the carcinogenic
activity of these chemicals is doubtful.
7.8.2 Behavioural effects
Behavioural studies of the effects of chlordimeform in rats were
first investigated by Olson et al. (1978). The effects of exposure
prenatally and post-natally were examined following a dietary intake
of 0.1 mg/kg body weight per day. Early development testing (swimming
and righting reflex) was conducted on rat pups from post-natal days 7
to 17, while motivational, learning and retention tests were conducted
on days 70 to 90. The most significant differences between control and
treated groups was in the swimming task, retarded maturation being
observed in the chlordimeform-fed group. There was no treatment-
related effect with regard to maze tests or with regard to the tests
of motivation.
Moser et al. (1988) examined the behaviour of rats using a
functional observation battery following a single oral administration
of chlordimeform at dose levels of 0, 1, 25 or 56 mg/kg body weight.
Rats were examined at 1, 5 or 24 h. Chlordimeform produced a decrease
in body weight as well as a decrease in body temperature. There was a
dose-related increase in general activity, CNS excitability and
sensory responsiveness, coupled with a decrease in rearing, gait and
arousal. Chlordimeform also produced an increase in grip strength.
Other behavioural effects observed with chlordimeform have
included appetite stimulation in rats (Pfister et al., 1978b), flavour
aversion in both rats (MacPhail & Leander, (1980) and mice (Leander et
al., 1984) and alteration in schedule-controlled performance in rats
(MacPhail & Leander, 1981), mice (Glowa, 1986) and pigeons (Leander &
MacPhail, 1980). Witkin & Leander (1982) also demonstrated that, while
causing appetite stimulation in rats, chlordimeform produced a dose-
related decrease in water consumption, in contrast to other appetite
stimulants.
7.8.3 Pharmacological and biochemical effects
The pharmacological and biochemical effects of chlordimeform in
animals have been reviewed by Knowles (1991).
The cardiovascular effects of chlordimeform treatment were
recognized from an early stage with the observation that chlordimeform
administered intraperitoneally to rabbits caused a marked decrease in
arterial blood pressure of almost 50% within 30 min of treatment
(Matsumura & Beeman, 1976). Cardiovascular changes were also noted in
the dog (Lund et al., 1979a,b; Rieger et al., 1981) but in this case
the effect was biphasic, consisting of an initial depressor response
associated with decreased cardiac contractility and vascular
resistance, and a secondary pressor response associated with increased
cardiac contractility and vascular resistance. These actions of
chlordimeform were noted to be similar to those of local anaesthetics
such as procaine and lidocaine (Pfister et al., 1978a; Lund et al.,
1979a,b,c).
In studies by Watkinson (1985, 1986a,b), the effects of
chlordimeform on cardiovascular functional parameters were examined in
post-weaning and geriatric rats following intravenous treatment at
dose levels up to 60 and 120 mg/kg body weight, respectively, or
intraperitoneal treatment of post-weaning rats at dose levels up to
60 mg/kg body weight. Chlordimeform produced profound and abrupt
decreases in heart rate and blood pressure within 3 min, together with
multiple arrhythmias and alterations in electrocardiogram waveforms
and intervals. The effects observed in post-weaning rats were less
severe than those observed in geriatric rats.
The inhibition of monoamine oxidase in rats in vivo and
in vitro by chlordimeform and/or its metabolites has been
extensively studied (Beeman & Matsumura, 1973; Maitre et al., 1978;
Benezet et al., 1978; Hollingworth et al., 1979; Kadir & Knowles,
1981; Kaloyanova et al., 1981; Bailey et al., 1982). The lack of
correlation of toxicity of chlordimeform metabolites to monoamine
oxidase inhibition and the fact that chlordimeform is a relatively
weak monoamine oxidase inhibitor suggest that monoamine oxidase
inhibition is not the primary factor involved in the acute toxicity of
chlordimeform (Neumann & Voss, 1977; Robinson & Smith, 1977;
Hollingworth et al., 1979).
Chlordimeform also has an effect on the level of biogenic amines
in brain and plasma of rats, which may in part at least be due to
the inhibition of monoamine oxidase levels. Administration of
chlordimeform to rats was found to produce an increase of 25-70% in
5-hydroxytryptamine, norepinephrine or dopamine levels in brain
(Maitre et al., 1978; Benezet et al., 1978; Bailey et al., 1982).
However, Johnson & Knowles (1983) treated rats subcutaneously with
chlordimeform (200 mg/kg body weight) and found no change in any of
the amines.
Chlordimeform and some of its metabolites have been shown to
affect platelet function, as measured by the uptake of radioactive
5-hydroxytryptamine (Knowles, 1991).
Chlordimeform also has antipyretic and anti-inflammatory actions,
as shown by its ability to reduce yeast-induced fever in rats. It also
antagonizes both early (5-hydroxytryptamine- and histamine-mediated)
and late (prostaglandin-mediated) phases of carrageenan-induced
hind-paw oedema, albumin-induced oedema, and oedema induced by direct
injection of 5-hydroxytryptamine and histamine (Yim et al., 1978).
Chlordimeform also induced mild gastric ulceration in rats after
intraperitoneal injection (20-80 mg/kg body weight) but not after oral
treatment (80-240 mg/kg body weight). The above actions may be related
to the ability of chlordimeform to inhibit prostaglandin biosynthesis
(Yim et al., 1978; Holsapple & Yim, 1981).
Chlordimeform induces hypothermia in rats (Watkinson & Gordon,
1987) and mice (Gordon et al., 1985). Watkinson et al. (1989)
examined the effect of core body temperature on both the survival
and cardiovascular functions of rats following treatment with
chlordimeform. The results indicated that at a given dose of
chlordimeform, the magnitude and duration of the observed toxic
effects are primarily a function of core body temperature. The authors
concluded that moderate hypothermia, but not extreme hypothermia, may
have a beneficial effect with respect to survival after exposure to
chlordimeform.
Chlordimeform has been shown to have an effect on both visual and
auditory functions in mammals. Intraperitoneal treatment of male rats
with acute dosages of chlordimeform (5-40 mg/kg body weight) before
testing revealed a temporary increase in both the amplitude and
latency of pattern reversal-evoked potentials and an increase only in
the latency of pattern flash-evoked potentials (Dyer & Boyes, 1983;
Boyes & Dyer, 1984). Boyes & Moser (1988) provided evidence to support
the hypothesis that these effects are evoked through actions as a
central nervous system alpha-adrenegic agonist. Janssen et al. (1983)
demonstrated effects on the brain stem auditory-evoked response after
injection of chlordimeform at a dose levels of 40 mg/kg body weight.
It has been suggested that these effects may by secondary to the
hypothermic effects induced by chlordimeform (Gordon et al., 1985).
Chlordimeform has been shown to affect the activity of hepatic
drug-metabolizing enzymes in both rats and mice. Studies have been
conducted following gastric intubation at dose levels up to 150 mg/kg
body weight per day for 7 days, and also following intraperitoneal
injections either singly (100 mg/kg body weight) or daily (75 mg/kg
body weight per day) for 4 days. Chlordimeform treatment induced
several of these hepatic drug-metabolizing enzymes with significant
species and/or sex specificity. Cytochrome P-450 content was increased
in all cases.
7.9 Factors modifying toxicity
The factors modifying the acute toxicity of chlordimeform have
been reviewed by Knowles (1991).
7.10 Mechanisms of toxicity - mode of action
7.10.1 Mechanism of acute toxicity
A large number of studies that investigated the mechanism of
action following acute poisoning with chlordimeform have been
reported.
Based on the early in vitro and in vivo studies of Aziz &
Knowles (1973) and Beeman & Matsumura (1973), it was suggested that
the increase in biogenic amines resulting from inhibition of monoamine
oxidase by chlordimeform could account for the variety of toxic signs
following acute poisoning. However, Maitre & Gfeller (1975) and
Robinson et al. (1975) demonstrated that this mechanism does not play
a significant role in the acute toxicity in rats.
A number of other studies have attempted to define the mode of
action of chlordimeform. Studies in insects have shown that
chlordimeform has little activity on cholinergic transmission although
it is an uncoupler of oxidative phosphorylation and an inhibitor of
electron transport (Abo-Khatwa & Hollingworth, 1972a). A number of
biochemical mechanisms have been postulated to explain the effects of
chlordimeform in insects, including uncoupling of respiration
(Abo-Khatwa & Hollingworth, 1972a,b), inhibition of monoamine oxidase
(Knowles & Roulston, 1972) and blockage of neuromuscular transmission
(Wang et al., 1975; Watanabe et al., 1975), and motor stimulation
through actions on central non-cholinergic synapses (Lund et al.,
1979a; Lund et al., 1979c). The latter effect is thought to be
mediated through the neurotransmitter, octopamine (Lund et al.,
1979b). Both chlordimeform and particularly demethylchlordimeform have
been shown to interact with the octopamine receptor and partially
mimic the pharmacological effects of octopamine (Evans & Gee, 1980;
Nathanson & Hunnicutt, 1981; Bokisch et al., 1985).
In mammalian systems, oxidative phosphorylation is uncoupled
(Abo-Khatwa & Hollingworth, 1972b) and RNA synthesis is inhibited by
chlordimeform, but only at very high concentrations (Murakami &
Fukami, 1974). The effects of chlordimeform on hepatic drug-
metabolizing enzymes was dependent on both sex and species and did not
show any particular pattern that would indicate a consistent mechanism
of action (Budris et al., 1983; Bentley et al., 1985; Leslie et al.,
1988).
Chlordimeform, acting as a direct depressant on cardiac and
vascular muscle, induced a hypotensive state in dogs. Chlordimeform
did not interfere with the autonomic nervous system. The mechanism of
cardiovascular depression may be related to that noted with frog nerve
preparations treated with procaine, a local anaesthetic. The
metabolite, 4-chloro- o-toluidine has been shown to interfere with
rat cardiac receptors (Wang et al., 1975; Watanabe et al., 1975;
Matsumura & Beeman, 1976; Knowles, 1976; Hollingworth, 1976; Lund et
al., 1978a).
More recent research has shown that formamidine pesticides may
exert their effects on the central nervous system by interacting
directly with adrenergic receptors, particularly the alpha-2 subtype
(Costa & Murphy, 1987; Costa et al., 1988, 1989). This interaction
appears to mediate several of the observed effects of formamidines,
such as changes in heart rate (Hsu & Kakuk, 1984, Watkinson, 1985;
1986a,b), pupil diameter (Hsu & Kakuk, 1984), visual evoked potential
(Boyes & Moser, 1988) and hormonal secretion (Goldman et al., 1990;
1991). Costa et al. (1991) demonstrated that chlordimeform
decreases the hepatic glutathione content by up to 40% in a
time- and dose-dependent manner, through an interaction with
alpha2-adrenoreceptors. Wu et al. (1990) have demonstrated that
chlordimeform inhibits the binding of the known alpha2-adrenoreceptor
blockers, clonidine and yohimbine, in rat forebrain tissue
in vitro. Furthermore, Stoker et al. (1991), in a further study on
the effects of chlordimeform on hormone release, have demonstrated in
rats, treated intraperitoneally with chlordimeform (20 or 50 mg/kg
body weight), that there is an increase in adrenocorticotropic hormone
(ACTH), circulating corticosteroid (CORT) and prolactin (PL) in a
dose-dependent manner. alpha-Adrenergic agonists specifically
inhibited these effects indicating the interference with a regulatory
signal mediated by alpha-adrenergic receptor-associated activity.
Candura et al. (1992) demonstrated that the inhibition induced by
chlordimeform in the intestinal tract is mediated by calcium channel
blockade rather than by alpha2-adrenoceptor activation. In a study by
Robinson et al. (1975), it was found that using drugs to block the
serotonergic or alpha-adrenergic receptors did not reduce the
chlordimeform-induced lethality in male rats.
7.10.2 Mechanism of carcinogenicity
Chlordimeform and its metabolites, N-formyl-4-chloro- o-
toluidine and 4-chloro -o-toluidine, have been shown to induce mouse
tumours of a vascular origin characterized histologically as
haemangioendotheliomas and haemangiosarcomas. 4-Chloro- o-toluidine
has been shown to be a more potent carcinogen than chlordimeform, both
with respect to dose-response and to a reduced latency period.
Haemangioendotheliomas and haemangiosarcomas were not induced in rats.
Cases of bladder cancer in humans associated with occupational
exposure to high levels of chlordimeform or 4-chloro- o-toluidine
have been seen in groups with high urinary levels of chlordimeform and
4-chloro- o-toluidine.
The exact mechanism of induction of these tumours is unknown but
there is evidence that a genetic mechanism involving mutations induced
by 4-chloro- o-toluidine is involved.
Metabolic studies in mice and rats indicate a similar metabolic
pathway for chlordimeform in both species. The kinetics of absorption
and elimination in mice and rats also seem to be similar. However, the
overall DNA binding was higher in mice than rats, and one DNA adduct
was formed to a 6- to 30-fold higher extent in mice.
There is considerable evidence that 4-chloro- o-toluidine causes
severe toxic effects in the human bladder leading to haemorrhagic
cystitis (see section 8). Monitoring of urinary metabolites in humans
also indicates that chlordimeform is rapidly metabolized to 4-chloro-
o-toluidine in vivo.
4-Chloro- o-toluidine also has a close structural similarity
to aromatic amines for which there is established evidence of
carcinogenicity by animal experimentation and also by human
epidemiological data (Parkes, 1984).
Taken together, the evidence strongly implicates 4-chloro- o-
toluidine as the causative agent in the induction of tumours in both
mice and humans. A proposed route of activation that may be associated
with carcinogenicity is shown in Fig. 2. However, the mechanism of the
carcinogenicity remains unclear.
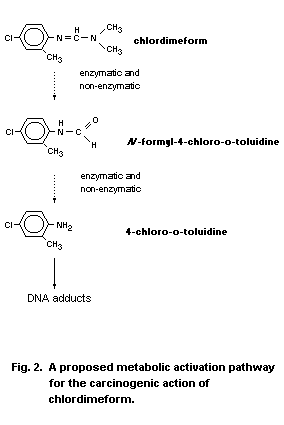 8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Acute poisoning incidents
The most comprehensive data on acute poisoning cases associated
with exposure to chlordimeform has come from China. Details of these
published poisoning cases are shown in Table 16. While many were due
to intentional ingestion, there were also cases of unintentional
poisoning as a result of consumption of contaminated food, as well as
occupational exposure to the spray. In a brief report prepared by Deng
et al. (1984) of a 1983 symposium in Hu-bei Province on chlordimeform
poisoning, which featured some 29 papers and 859 case studies, it was
stressed that the main cause of death was suppression of cardiac
contracture and dilation of blood vessels resulting in circulatory
failure.
Arima et al. (1976) described an unsuccessful suicide attempt
involving a 76-year-old male who ingested 100 g chlordimeform. He
vomited several times before arriving at hospital 50 min after
ingestion. He was lethargic with a weak pulse and cyanosis associated
with his lips, nails and skin. Methaemoglobin levels represented 17%
of total haemoglobin at 5 h but returned to normal levels by 2 days.
He regained consciousness by 50 h, although complained of headache and
blurred vision. The only treatment received was gastric lavage, which
was performed shortly after his arrival at the hospital.
8.2 Occupational exposure
8.2.1 Acute poisoning incidents
Currie (1933) reported nine cases of haematuria in workers
exposed to 4-chloro- o-toluidine (erroneously called 5-chloro- o-
toluidine) by inhalation or possibly by absorption through the skin.
All patients had difficulty urinating and had suprapubic pain. Most of
the workers were exposed to the material for only 1-2 days. Despite
efforts to control exposure to the chemical in the factory, further
cases of poisoning occurred, and manufacture was ceased. In a
follow-up study of three of the nine cases after 3 years, one patient
had no bladder trouble, one had a slight cystitis and urethritis, and
one had carcinoma of the bladder.
Jurincic et al. (1991) reported cases of acute haemorrhagic
cystitis in two men (aged 19 and 50) following involvement in cleaning
of a water-tank that had likely been used to transport chlordimeform.
Both developed abdominal pain, dysuria and haematuria in the evening
following exposure. Cystoscopy revealed haemorrhagic cystitis,
which was confirmed by bladder mucosa biopsy. Serum levels of
4-chloro- o-toluidine (referred to as 4-chloro-2-methylaniline) were
>1 mg/litre in both patients and urine levels were 16 mg/litre in the
Table 16. Case studies of acute chlordimeform poisoning in China
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
1 71 4 dermal absorption 5 ECG: 26 tachycardia; 6 bradycardia; 11 ectopic Wang & Tong (1992)
(28 male, 67 ingestion rhythm; 6 premature beat; 2 atrial fibrillation;
43 female) ventricular fibrillation; 1"Torsade de Pointes";
2 high pike P, 6 A-V block, 17 S-T depression,
3 inverse T, 1 S-T elevation ,7 Q-T elongation.
In 33 severe cases, 28 has ECG abnormalities;
38 moderate cases, 14 had abnormal ECG. Changes
in heart were found in 32 cases. Deaths were from
respiratory failure (3); ventricular fibrillation
(1) and supraventicular tachycardia (1).
2 4 ingestion(?) 0 Mild cyanosis, cystitis (2 cases occupational, Nui et al. (1990)
2 cases non-occupational; OPs also in formulation.
3 1 female ingestion 0 Jaundice on 3rd day which progressively deepened. Liu et al. (1990)
(30 years old) (150 ml) Hb 40 g/litre (70 g/litre on admission); complete
recovery, discharged on day 20.
4 52 ingestion 0 Loss of appetite (86.5%), urgency in urination He (1989)
(19 male, (20-350 ml) (84.6%), cyanosis (81.1%), coma (67.3%), miosis
33 female) (34.6%), mydriasis (15.4%),hypotension (38.5%),
tachycardia (32.7%), bradycardia (3.8%).
Impairment of liver and renal functions. 15 ECGs:
7 tachycardia, 2 bradycardia, Q-T elongation,
8 T-wave changes. Treated with methylene blue,
vitamin C, fresh blood transfusion and sopolamine.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
5 35 ingestion(?) 0 18 severe cases. Suggested use of 5-36 mg He et al. (1987)
atropine for chlordimeform poisoning and
50-128 mg for mixed pesticide poisoning.
6 1 female ingestion 0 Cyanosis, pin-point myosis. Given atropine Zhou (1987)
(30 years old) (80 ml 25% (15 mg/min) after lavage until total of 530 mg.
chlordimeform) Symptoms indicated overdose of atropine. Methylene
blue given, recovery and discharge at day 7.
7 23 4 contaminated food; 3 Mild case: nausea, vomiting, light cyanosis,
(6 male, 19 ingestion no somnolence. Moderate case: somnolence and Xu (1987)
17 female) (10-350 ml) light consciousness. Severe case: Marked
cyanosis, coma, shock. 5 ECG examined:
2 bradycardia, 1 tachycardia, A-V block,
S-T change. Mild impairment of liver renal
functions. Treated with methylene blue,
19 recovered.
8 1 female ingestion 0 Lavage and treatment led to recovery from danger. Liu & Li (1987)
(52 years old) (30 ml conc. Black stool, tachycardia occurred on 3rd day.
formulation) Complete recovery.
9 187 27 occupational spray; 13 Cyanosis (63.6%), nausea (49.2%), vomiting Ding & Huang (1987)
(66 male, 16 ingestion (20-250 ml (44.9%), mydriasis (32.1%), somnolence (33.7%),
121 female) 25% chlordimeform coma (32.1%), irritation in urination (30.5%),
formulation) hypotension. 27 ECGs: 4 tachycardia, 6 bradycardia,
4 S-T & T wave change, 2 pre-mature beat, 2
conductive blockage. 158 cases received methylene
blue and 174 recovered within 1-5 days.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
10 1 male occupational spray 0 Sprayed incorrect dilution spray. Complained of Gu et al. (1987)
(28 years old) fatigue, somnolence, loss of appetite, nausea,
vomiting, but no cyanosis, or signs of cystitis,
pulse 68, BP 128/94 (normally 120/80), MAO 25.12 U
(normally 38.87 U). Total chlordimeform in urine
on admission, 6.4 mg/ml. Recovered quickly.
11 6 (?) ? Main clinical features: drowsy, cyanosis, Chan (1985)
loss of consciousness, mydriasis, cystitis,
hypotension, bradycardia, myocarditis, shock,
methaemaglobinaemia.
12 47 ingestion 4 Symptoms: drowsy, cyanosis, cystitis, Ke (1985)
(11 male, 20-1900 ml 2 hypotension (severe case), 8 hypertension,
36 female) 10 ECG: 1 tachycardia and T-wave change.
13 25 ingestion(?) 1 Cyanosis, cystitis, hypotension, arrhythmia, Wang & Dong (1985)
S-T and T changes, Q-T elongation. Treatment
with gastric lavage, methylene blue, vitamin C
14 682 340 occupational spray; 25 279 cyanosis, 147 cystitis, 197 somnolence, Liu & Zhang (1985)
(331 male, 342 ingestion 211 coma, 81 shock, 109 tachycardia,
351 female) 64 bradycardia, 54 hypertension, 22 hypotension.
59 ECG: 8 premature beat, 4 Q-T elongation,
16 S-T and T changes.
15 358 283 ingestion 37 Somnolence, cyanosis, loss of appetite, Ding & Ru (1985)
haemorrhagic cystitis, often myocardium damage,
A-V block, cardiac failure.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
16 49 3 occupational 4 13 cases were severe. Clinical features: cyanosis Liu & Ke (1985)
46 non-occupational and cystitis with haematuria in all cases, most
with severe somnolence and a few with coma. Two
severe cases had hypothermia. Hypertension was more
common than hypotension. 10 ECGs: only one case of
T-wave change and tachycardia. Treatment with
methylene blue and lavage.
17 1 ingestion (300 ml 25% 0 Coma and cyanosis. Sudden cardiac arrest during Yang (1984)
(female, form.) lavage, rescued with mechanical respiration.
25 years old) Recovered after 14 days.
18 24 ingestion (15-150 ml 2 16 cyanosis, 14 drowsiness, 8 haematuria, Wu et al. (1983)
(11 male, 25% form.) 6 methaemoglobin, 1 cardiac arrest, which
13 female) recovered after resuscitation.
19 101 35 occupational spray, 2 89 chlordimeform alone cases: 66% cyanosis, Xie (1983)
(49 male, 66 ingestion; 32 comas, 14 cystitis, 14 hypotension, 3 cardiac
52 female) chlordimeform +Ops) failure. 8 ECGs: 6 myocardium damage (changes in
Q-T, S-T, and T waves). Treatment with methylene
blue, vitamin. C. All recovered.
12 cases with mixed pesticides (OPs and Ocs).
20 1 ingestion 1 Loss of consciousness, cyanosis, mydriasis, Wu (1982)
(female, 85 years) (30 ml) arrhythmia. ECG: bradycardia, T-wave changes.
Died on day 6.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
21 20 occupational spray 0 Farmers applied wrong dilution chlordimeform to Li et al. (1982)
(18 male, 2 female) cotton for one day. 7 drowsy, 10 loss of appetite,
4 cystitis. Symptomatic treatment. All recovered
in 2-4 days.
22 2 male ingestion (100 & 200ml) 0 Cyanosis, coma, respiratory-circulation failure, Zhang et al. (1976)
cystitis during 2nd day. Treatment with
methylene blue and atropine.
23 1 male 100 ml 0 Deep cyanosis, pulse 166. Xia & Gao (1977)
24 6 male occupational spray 0 Contamination of body surface and clothing. Su (1977)
Symptoms from day 1-4: cyanosis, haemorrhagic
cystitis, fatigue. Recovery after 18 days.
25 2 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Anonymous (1977)
no cyanosis, ECG normal. Symptomatically treated.
26 4 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Ming (1977)
cyanosis, somnolence, loss of appetite, haematuria,
RBC in urine for 20 days. Treatment: vitamin C,
antibiotics, coagulators.
51-year-old patient. Case studies of chlordimeform poisoning in China
due to occupational exposure are given in Table 17, together with a
brief account of the clinical features observed.
Table 17. Levels of urinary chlordimeform and its metabolites in hospitalized
workers (3 days following exposure) (Folland et al., 1978)
Worker Total aminesa Chlordimeform 4-Chloro-o-toluidine Conjugate
(mg/litre) (mg/litre) (mg/litre) (mg/litre)
1 11.0 1.10 3.75 6.25
2 15.2 2.16 4.16 8.67
3 2.6 0.04 1.25 1.17
a Measured following hydrolysis with 10N NaOH and 2 h at 80�C.
A brief account of the signs and symptoms of chlordimeform
poisoning and suggested interventions has been provided by Xue &
Loosli (1994).
8.2.2 Effects of long-term exposure
A report of an outbreak of haematuria in employees of a chemical
packaging plant in the USA over a 4-day period in 1975 was first
reported by Armstrong et al. (1975). Further details were described by
Folland et al. (1978). Nine of 22 workers who packaged chlordimeform
became severely ill with abdominal pain, dysuria, urgency to void, or
haematuria. In the previous year, four workers who had packaged the
chemical had similar symptoms. While six workers recovered within 7 to
18 days, three were hospitalized with symptoms which lasted from one
to two months. In these three workers, abnormalities noted were
microscopic haematuria and pyuria, proteinuria, low creatinine
clearance, elevated SGOT, prolonged BSP retention, elevated serum
amylase level, small bladder capacity, ureteral reflux and an intense
inflammatory reaction in three bladder biopsy specimens. The highest
concentrations of total amines were found in the urine of workers who
had become ill and were hospitalized. Low but measurable levels were
also found in workers who had not become ill. The major part of the
urinary amines was present as 4-chloro- o-toluidine or as conjugates.
Urinary total amines (following hydrolysis with 10 N sodium hydroxide
and 2 h at 80�C), as well as chlordimeform and 4-chloro- o-toluidine,
were measured in the hospitalized cases and are shown in Table 17.
The results of a monitoring programme on packaging workers in a
chlordimeform plant in the USA during 1976 have been described
(personal communication by J.W. Barnett, Ciba-Geigy Agricultural
Division, Greenborough, North Carolina, USA, to the California
Department of Food and Agricultural). The programme involved more than
100 workers and over 800 urine samples, monitoring for the presence of
red blood cells, for residues of chlordimeform metabolites, and for
clinical signs of toxicity in workers. Residues in urine samples were
reported to range from <0.05 to 50 mg/litre. There was no evidence of
microscopic haematuria found in the samples analysed nor of any
clinical signs of toxicity.
Four separate incidents resulting in 7 cases of frank haematuria
following industrial exposure were reported in the USA during the
period 1980-1984 (personal communication by J.W. Barnett, Ciba-Geigy
Agricultural Division, Greenborough, North Carolina, USA, to
Ciba-Geigy Ltd., Switzerland). Chemical cystitis, confirmed by
cystoscopy and biopsy, was diagnosed in one case while non-specific
bladder mucosal lesions were found in another. Six cases required
hospitalization, but all resolved after cessation of exposure.
In a study by Maddy et al. (1986), the results of a programme of
monitoring (1982-1985) the urine of more than 200 workers, who had
received training in the use of chlordimeform on cotton in California,
were described. Although urinalysis was unremarkable and no
significant cytological changes were found, a single case of bladder
cancer was detected in a pilot who had seven seasons of exposure to
chlordimeform.
By contrast, in the same period (1980-1984), no cases of
chlordimeform-induced haematuria occurred at manufacturing plants in
Switzerland and West Germany or formulation plants in Australia,
Columbia, Central America, Mexico and the USA. No cases of haematuria
reportedly resulted from application or use of chlordimeform in the
field (Anon., 1985b; personal communications by F.E. Pfister and P.
Duback (Ciba-Geigy Ltd., Agricultural Division, Switzerland) and by
N. Reckefus and K. Kossmann (Schering Aktiengesellschaft Agrochemical
Division, Berlin, Germany), 1985).
In a study by Lu et al. (1981), data on the effects of
chlordimeform exposure of factory workers in China was examined. In
this study, conducted in 1974, the air concentrations in the factory
were generally below 0.036 mg/m3, with shorter periods at higher
levels (0.108-0.33 mg/m3), during specific tasks. Skin contamination
on hands and forearms was 9.1 mg/h for chemical operators
and 964.2 mg/h for packers. The urinary excretion levels of
chlordimeform and 4-chloro- o-toluidine in controls were 0.015 and
0.042 mg/litre, respectively; in chemical operators they were
0.065 and 0.108 mg/litre, respectively; and in packers were 0.263 and
0.398 mg/litre, respectively. The health of the workers was examined
during the following 3 years (1974-1976). In 44-56 workers (equal
number of each sex) at an average age of 32 years and working period
of 2 years, the main finding were neurosis, sore throat and disorders
of the nervous system. There were no treatment-related effects on ECG,
liver function, clinical chemistry or urinalysis parameters.
In the same report (Lu et al., 1981), the effect of chlordimeform
exposure on rice field workers during 1974 was also examined. The
air concentration in the breathing space in all cases was below
0.02 mg/m3. Skin contamination was examined at the front of the
thorax, on the right forearm and on the right thigh. The applicators
applied chlordimeform for 4-5 h per day for 1-3 consecutive days,
wearing shirts and shorts with no other protection. Skin contamination
was from splash or from spray. The levels found from splash on thorax,
forearm and thigh were 0.0436, 0.0303 and 0.131 mg/100 cm2 per h,
respectively. The levels found from spray on thorax, forearm and thigh
were 0.235, 0.299 and 0.804 mg/100 cm2 per h, respectively. Medical
examination during 1974/1975 revealed complaints of light-headedness,
headache, fatigue, nausea, abdominal pain, skin itching and burning
sensation, and hypotension. There were no changes in ECG or blood
chemistry, and no reported cases of acute intoxication.
In a study by Li et al. (1985b), the health of 24 packers
(9 male, 15 female) in a chlordimeform manufacturing plant in Jiang-su
Province of China, was examined. The chlordimeform division of the
factory started manufacturing in 1975 and continued to do so at the
time of the study. The employees were working in the factory for
between 3 months and 4 years (average 1.5 years). Another 24 employees
from the kitchen and kindergarten served as controls. The air
concentration of chlordimeform (9 samples over 3 consecutive days) was
0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin contamination of the
hands and forearms was 110 �g/100cm2 (S.D. 39 �g/100 cm2). Urinary
chlordimeform levels were 0.20 � 0.13 mg/litre, and urinary
4-chloro-o-toluidine levels were 0.48 � 0.29 mg/litre. Medical
examination revealed no difference between packers and controls with
regard to symptoms, laboratory examinations including liver enzymes
and urinalysis parameters, chest X-rays, ECG, or other parameters of
cardiac function. The only symptom associated with exposure was skin
rashes and itching in 21% of exposed individuals. There was no
difference in the micronucleus counting in cultured peripheral
lymphocytes between exposed and control groups, nor were there any
positive mutagenicity results from urine samples with or without
glucuronidase or sulfatase in the medium.
In a further study in a Chinese chlordimeform manufacturing
factory, the health of employees involved in chlordimeform production
was studied for the 5-year period, 1977 to 1981 (Anon., 1985a). The
urinary chlordimeform plus 4-chloro- o-toluidine levels of packers
was the highest at 0.39 mg/litre, which significantly correlated with
skin contamination but not with air concentration. The major medical
findings were complaints of lightheadedness, disorders in sleep,
memory impairment, fatigue, loss of appetite, skin rashes and itching,
and skin spot pigmentation. There were no features of cystitis. ECG
findings in 36 employees indicated premature beats, partial A-V block,
tachycardia and bradycardia. There was no evidence of chromosome
aberrations in metaphase chromosomes of cultured peripheral
lymphocytes.
In a study by Tao et al. (1985), the health of 61 employees
(25 chemical operators, 36 packers) of a pesticide factory in China
was examined. Chlordimeform was produced in the factory for 5 months
per year. Air levels ranged from 0.074 to 0.160 mg/m3. Skin
contamination of packers (2.99 mg/day) was higher than for
hemical operators (0.784 mg/day). The urinary excretion rate of
chlordimeform plus 4-chloro- o-toluidine in packers was also higher
(0.513 mg/litre) than for chemical operators (0.206 mg/litre) or
controls (0.055 mg/litre). Symptoms of exposure noted in packers
included loss of appetite, fatigue, somnolence and skin rashes.
Hepatomegaly was observed. There was no difference in blood pressure
or heart rate. Abnormalities in ECG were noted in 10/61 exposed
employees compared to 6/76 controls.
In a study by Wang et al. (1987), the health of 16 applicators
(8 males, 8 females) spraying chlordimeform in cotton fields in
Xin-yang Farm in the Jiang-su Province of China over a 3-day period
(July 1986) was examined. Air levels in the breathing zone were
0.031 mg/m3 and the skin contamination was 4.17 mg per shift. Urinary
levels of chlordimeform plus 4-chloro- o-toluidine ranged between 1
and 3 mg/litre over the exposure period. A close correlation was noted
between the level of chlordimeform on the skin and the levels of
chlordimeform plus 4-chloro- o-toluidine in the urine. Rapid
excretion of chlordimeform plus 4-chloro- o-toluidine was noted
following exposure. There was no change noted in heart rate, blood
pressure, monoamine oxidase activity or urinalysis between exposed
individuals and controls. Mild chlordimeform exposure, however,
appeared to be related to loss of appetite and drowsiness.
In a study by Zhang et al. (1986a), conducted at the same farm
over the same period, 13 applicators (7 male, 6 female, 20-41 years of
age) were examined during spraying chlordimeform on cotton over three
consecutive days. Protective measures included gauze mask, plastic
gloves and plastic apron, although it was noted that extensive
contamination occurred. Air levels in the breath zone on each of the
three days were 0.011, 0.014 and 0.011 mg/m3, respectively. Skin
contamination on each of the three days was estimated by the method of
Zhang et al. (1986b) to be 10.99, 4.32 and 4.45 mg/person per day,
respectively. Urinary chlordimeform plus 4-chloro- o-toluidine
levels were measured over the 3 days of exposure and for 7 days
after cessation of exposure. Urinary levels ranged from a peak of
2.408 mg/litre during exposure to 0.036 mg/litre after 7 days.
Excretion of chlordimeform occurred very rapidly with the highest
level being detected in the sample collected at the end of each shift.
There was a close correlation between skin contamination and urinary
excretion. Metabolism occurred very rapidly since 4-chloro- o-
toluidine usually accounted for 70-93 % of the total amount in the
urine. Serum monoamine oxidase activity varied from 26.18 U to
19.26 U. Clinical symptoms were somnolence, headache, dizziness and
fatigue. Heart rate and blood pressure dropped on the 2nd and 3rd
days. Analysis of ECG indicated elongation of P-R, Q-T intervals. One
person complained of urgency and pain in urination, gross haematuria,
and the urinary chlordimeform plus 4-chloro- o-toluidine level was
more than 6 mg/litre. Another four subjects were found to have
microscopic haematuria. Liver function tests were normal.
In a study by Xue et al. (personal communication by S.-Z. Xue,
M. Wang, C.-M. Chu and X.-W. Zhou entitled "Effects of chlordimeform
on cardiovascular function in humans with occupational exposure",
1993), the effect of chlordimeform on cardiovascular function was
studied in exposed farm workers and in manufacturing workers in China.
Four separate exposure groups were studied. The first (short-term)
exposure group consisted of 16 farmers engaged in spraying
chlordimeform (0.125% solution) in a cotton field. Exposure was for a
3- to 4-h period for 3 consecutive days. The second (long-term)
exposure group consisted of 21 chlordimeform packers in a factory who
had worked for 6 months on this task. The third exposure group
consisted of 19 factory plant operators who had minimal exposure to
chlordimeform. The fourth group consisted of 9 control (non-exposed)
factory workers. Exposure was measured in the breathing zone air
(personal sampler for the working shift, usually 6 h), by dermal
contact (pooled aliquot of 10 swabs from various body sites), and by
urine measurements. In each case, chlordimeform and its major
metabolite, 4-chloro- o-toluidine, were measured. The cardiovascular
system function was determined by measurement of blood pressure, heart
rate and electrocardiography (ECG). Exposure data indicated the packer
group had a higher inhalation exposure (0.107 mg/m3) than the
sprayers (0.031 mg/m3). Dermal exposure, on the other hand, was
higher in the sprayers group (4.251 mg/m2) than in the packers group
(2.713 mg/m2). Urinary levels collected at the end of the working
shift indicated the highest level in sprayers (1.950 mg/litre)
compared to packers (1.267 mg/litre) and operators (0.097 mg/litre).
In the farmer group, analysis of cardiovascular activity indicated a
significant decrease in heart rate, and an increase in P-wave
duration, Q-T interval and amplitude of T-wave compared to the control
group. In the factory workers, the packers had significantly lower
diastolic and systolic blood pressure, and an increase in T-wave
amplitude compared to the plant operators. The heart beat of packers
was also higher than controls, but not significantly. The
cardiovascular function parameters of the plant operators were
slightly but not significantly different to those of controls.
Examination of the cardiovascular function parameters of the
packers during a month of continuous exposure indicated a relationship
between length of exposure, total urinary chlordimeform, and cardiac
function parameters (see Table 18). Analysis of the exposure-effect
correlation indicated the drop in blood pressure was the most
sensitive parameter, with the change in amplitude of the T-wave the
next most sensitive parameter. The changes of P-R interval were the
least sensitive.
Table 18: Cardiovascular function and urinary chlordimeform in factory workers
(personal communication by S.-Z. Xue, M. Wang, C.-M. Chu and X.-W.
Zhou entitled "Effects of chlordimeform on cardiovascular function
in humans with occupational exposure", 1993)
Parameter measured Duration of continuous exposure (days)
0 1 7 15 30
Total urinary 0.000 0.311 0.627 0.642 0.773
chlordimeform (mg/litre)
Systolic BP (mmHg) 111/8.6a 105/7.7b 105/12c 102/10d 102/9.6c
Diastolic BP (mmHg) 71/7.1 69/9.3 63/10c 65/8.2d 64/9.8d
Heart rate (beat/min) 64.3/9.9 69.6/8.6d 67.2/6.5 70.0/9.4 71.4/12d
Q-T interval (msec) 398/18.2 404/23.1 412/16.7d 418/22.3d 412/23.6d
P-R interval (msec) 131/215 140/178d 140/212d 141/200a 143/317d
a Figures are mean/standard deviation
b P < 0.001
c P < 0.01
d P < 0.05
The authors attributed major importance to the alteration in
cardiovascular function in relation to chlordimeform intoxication, and
in most cases considered cardiac failure to be the cause of death.
Recognition of the effects on cardiac function may have been
overlooked previously, firstly, because of the diversity of mild
changes induced by chlordimeform and, secondly, because of the
tendency to concentrate on the effects of the aniline-containing
metabolites, such as methaema-globinaemia, haematuria, and
haemorrhagic cystitis. A no-observed-effect-level (NOEL) of
0.1 mg/litre of urinary chlordimeform plus 4-chloro- o-toluidine
excretion is proposed as the threshold for effects on cardiovascular
function following long-term, exposure while 0.3 mg/litre is proposed
as the threshold for effects on cardiovascular function following
short term exposure, even as short as one day. While the
cardiovascular function parameters are unlikely to be useful as
indicators of exposure, an understanding of the mechanism of action
should assist in designing appropriate treatment.
In a post-exposure surveillance programme, the chlordimeform-
exposed group showed an increased prevalence of malignancy-associated
surface markers on exfoliated urine cells, compared to geographical
controls, but no tumours were found (Kenyon et al., 1993).
8.2.3 Epidemiological studies
8.2.3.1 4-Chloro- o-toluidine
In a retrospective epidemiological study by Ott & Langner (1983)
the mortality experience of 342 employees assigned to three aromatic
amine-based dye production areas between 1914 and 1958 was examined in
relation to duration of employment (<1 to 5 years) and interval since
entry into these areas. 4-Chloro- o-toluidine represented one of a
number of chemicals to which the workers were potentially exposed.
4-Chloro- o-toluidine and two other aromatic amines ( o-toluidine
and 4-chloro-acetyl- o-toluidine) to which the workers were exposed
have been shown to be carcinogenic in animal studies. No deaths due to
bladder cancer were observed, and no statistically significant
increases in mortality by work area or duration of exposure within
work area were found.
In a retrospective study by Stasik (1988; 1991) of 116 workers
occupationally exposed in Germany to 4-chloro- o-toluidine during
manufacture prior to 1970, eight cases of bladder cancer, diagnosed
between 1967 and 1985, were identified. This represents an incidence
more than 70-fold higher than expected. Although occupational exposure
to two other aromatic amines, o-toluidine and 6-chloro- o-
toluidine, may have occurred, analysis of the production process
indicated that exposure to 4-chloro- o-toluidine in the plant was
considerably higher than exposure to these other two chemicals. The
workers were exposed to relatively high levels (before 1970) for a
median of 14 years. In two cases, however, the exposure period was
only 1.5 and 4.0 years. No quantitative measurements of exposure were
available. Two of the patients had suffered from haemorrhagic cystitis
as a consequence of massive acute exposure to 4-chloro -o-toluidine
at 4 and 14 years, respectively, before the tumour was diagnosed. The
latency periods for these eight cases ranged from 17 to 38 years. The
significantly increased incidence of bladder cancer in this study is
remarkable.
8.2.3.2 Chlordimeform
An epidemiological study has been conducted on the incidence of
cancer deaths of employees and their relatives on Xin-Yang Farm in
Jiang-su Province of China (Gu et al., 1991). In this area,
chlordimeform has been applied aerially in large amounts since 1974,
in a relatively indiscriminate manner, with contamination of land,
ponds, creeks, and gardens of adjacent houses. The study involved 7321
people (3911 male, 3410 female and 1265 retired agricultural workers)
over the period 1 January 1971 to 30 June 1987. During this period,
there were 706 registered deaths (510 males, 196 females), of which
198 were cancer deaths (160 males, 38 females). The standardized
mortality ratio (SMR) was calculated on the basis of the specific
mortality due to cancer in the adjacent Hai-men County. Many of the
SMRs were significantly exceeded on the Xin-Yang Farm, as shown in
Table 19. The incidences of bladder cancer adjusted to the national
level were 2.65 (males) and 1.47 (females) per 100 000. The SMRs were
260 (males) and 420 (females). During the period 1 July 1987 to
30 June 1990, there were three more cases of bladder cancer (with one
death) among the cohort members. The authors concluded there is
evidence for an association between bladder cancer and exposure to
chlordimeform, but that further data is needed to strengthen this
association. It is noted that there was a high incidence of other
tumour types in this study which makes the association between bladder
cancer and exposure to chlordimeform more difficult to establish.
Table 19. Standardized mortality ratio (SMR) for workers on the
Xin-Yang farm (Gu et al., 1991)
Cause of death Adjusted mortality Standardized mortality ratio
(per 100 000) (95% C.I.)
Male Female Male Female
All deaths 785.1 610.0 134 (124-145) 139 (128-151)
All cancers 214.0 130 113 (107-120) 128 (117-139)
Oesophageal cancer 35.6 32.5 228 (208-249) 388 (352-428)
Stomach cancer 61.4 24.5 175 (161-190) 120 (110-130)
Liver cancer 31.6 6.9 40 (37-44) 27 (24-29)
Colon cancer 8.8 6.5 133 (123-145) 79 (72-86)
Lung cancer 34.2 16.9 135 (124-146) 147 (135-169)
Leukemia 3.9 5.6 144 (133-157) 260 (235-285)
Bladder cancer 4.1 3.0 197 (180-214) 750 (671-839)
Breast cancer - 16.0 - 380 (345-419)
Cervical cancer - 30.7 - 216 (198-234)
Further epidemiological data on the association between cancer
incidence and exposure to chlordimeform has been provided in papers by
Xue et al. (1990; 1991). A summary of the findings of epidemio-logical
studies between 1984 and 1988 is given in Table 20. Data from three
counties and one farm are shown. The counties are located close to one
another, with comparable environmental and socio-economic situations.
The agricultural products are mainly rice and cotton. County A acted
as a control, with little or no use of chlordimeform; County B was
the largest user of chlordimeform; and County C started using
chlordimeform at the earliest time. The results from the Xin-Yang farm
are included for comparison. A comparison between the mortality rate
in recent years (1984-1988) and the mortality rate in the years prior
to the use of chlordimeform in these counties and Xin-Yang farm is
shown in Table 21. There were excesses in the incidence of all deaths,
deaths from cancer, and urinary bladder cancer for both sexes,
although the data may not yet have reached the level of statistical
significance.
Table 21. Comparison of adjusted mortalities of urinary bladder
cancer between 1984-1988 (county) and 1973-1975
(prefecture) (Xue et al., 1990, 1991)
Item County A County B County C Xin-Yang Farm /
/ Prefecture / Prefecture Prefecturea
Male 1.52 / 1.10 0.77 / 0.77 1.12 2.65 / 1.02
SRR 1.38 1.04 1.10 2.65b
Female 0.41 / 0.35 0.46 / 0.17 0.55 1.47 / 0.35
SRR 1.17 2.71b 1.57b 4.20b
a The duration of observation was 1971-1987 (June 30)
b p < 0.05
In a retrospective study by Popp & Norpoth (1991) and Popp et al.
(1992), the exposure and incidence of bladder cancer in a German
chemical plant was examined. Chlordimeform was manufactured from
4-chloro- o-toluidine and production commenced in December 1965.
Production was not continuous, but rather was in response to orders,
so workers were subject to different periods of exposure (generally
8-12 weeks per year). Between 1965 and 1976, the exact levels of
exposure were not available because measurement of the concentration
in the air or monitoring of urine excretion was not carried out at
that time. In 1976, production was ceased in order to improve working
conditions and minimize human exposure. Production recommenced in 1980
with improved containment and monitoring of urinary excretion of
Table 20. Data on Epidemiological Studies with Chlordimeform during 1984-1988 (Xue et al., 1990, 1991)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
Year started using chlordimeform 1979 1977 1973 1973
Population (annual average) 993 549 1 076 456 736 037 8732
Average amount of chlordimeform 1.1 65.0b 29.8 89.2
used (g/Mu/year)a
All causes of mortality
Male 584.5 675.7 761.0 785.1
RR 1.2 (1.1-1.3) 1.3 (1.2-1.4) 1.3 (1.2-1.5)
Female 438.1 891.7 668.5 625.9
RR 2.0 (1.4-2.3) 1.5 (1.1-1.7) 1.4 (1.3-1.5)
Cardiovascular mortalityc
Male 143.2 167.7 221.6 -
RR 1.2 (1.1-1.3) 1.6 (1.4-1.7)
Female 138.2 280.2 234.8 -
RR 2.0 (1.9-2.2) 1.7 (1.6-1.8)
Respiratory mortalityc
Male 99.6 100.4 127.2
RR 1.0 (0.9-1.1) 1.3 (1.2-1.4)
Female 82.1 145.1 124.0
RR 1.8 (1.7-1.9) 1.5 (1.4-1.6)
Table 20. (con't)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
All cancer mortalityc
Male 188.5 246.6 232.2 214.9
RR 1.3 (1.2-1.4) 1.2 (1.1-1.3) 1.1 (1.1-1.2)
Female 101.7 227.1 145.5 130.0
RR 2.3 (2.0-2.5) 1.4 (1.3-1.6) 1.3 (1.2-1.4)
Bladder cancer mortalityc
Male 2.08 (95)d 0.90 (26) 2.10 (32) 4.10 (4)
RR 0.4 (0.39-0.47) 1.0 (0.9-1.2) 2.0 (1.8-2.2)
Female 0.40 (15) 0.20 (9) 0.90 (14) 3.00 (2)
RR 0.5 (0.46-0.55) 2.3 (2.1-2.5) 7.5 (6.7-8.4)
a The Mu is a measure of area equivalent to 1/15th acre
b Considered over the last 5 years
c All mortality figures were age-adjusted
d Figure in parentheses is the actual number of cases of bladder cancer
workers. Production finally ceased in 1986. The company identified 170
individuals who had come into contact with chlordimeform but many had
minimal exposure. The number of workers involved in the production of
chlordimeform was 49, and these comprised the study group. The period
under investigation was from the year of employment to the end of
1990. The expected incidence of bladder cancer (age- and sex-specific)
was extracted from the cancer registers of Saarland (1988), the former
German Democratic Republic (GDR) (1978-1982) and Denmark (1978-1982).
The standard incidence rate (SIR) was the ratio of the number of cases
observed to the expected number (see Table 22).
Table 22. Standard incidence rates (SIRs) of bladder carcinoma in a
group of 49 workers engaged in chlordimeform synthesis
(Popp et al., 1992)
Observed cases Expected number SIR 95% CI p value
7 0.078 (GDR) 89.7 35.6 - 168.6 0.000002
7 0.200 (Denmark) 35.0 13.9 - 65.7 0.00001
7 0.130 (Saarland) 53.8 21.3 - 101.1 0.000005
The average age for workers starting work was 30 (range 18-51), and
the exposure ranged from 3 to 956 days. By the end of 1990, an average
of 18 (10-25) years had passed since the start of exposure. Bladder
cancer was detected in 7 of the 49 subjects by the end of 1990. Of the
seven cases, six were diagnosed as transitional cell carcinoma and one
as papillary carcinoma. In five cases, the exposure period could be
determined, with an average of 575 days (range 291-766). The latency
period was an average of 19 years (range 15-23), with an average age
at diagnosis of 54 years (range 42-62). This study provides strong
evidence of an association between exposure to 4-chloro- o-toluidine
and human bladder cancer. All of the cases involved workers who were
exposed to 4-chloro- o-toluidine while synthesizing chlordimeform
before 1976. None of those workers who were handling the final
product, chlordimeform, had developed bladder cancer by the end of
1990.
In a historical cohort study (personal communication by P. Boyle
& G.J. Macfarlane to the IPCS, 1997), the bladder cancer incidence of
847 men involved in the manufacture of chlordimeform in Australia,
Switzerland, the United Kingdom and the USA was compared with that
expected on the basis of population-based cancer registry rates.
Subjects eligible to be included in the cohort were those who had been
employed in the production or formulation of chlordimeform or who had
otherwise been an integral part of a chlordimeform unit in a plant
where it had been produced or formulated for at least 6 months. The
results presented in Table 23 show an incidence rate of bladder cancer
which was significantly higher than expected. Overall, ten cases were
observed while 2.6 were expected. When the cohort was divided
according to whether members had been exposed to chlordimeform and
4-chloro- o-toluidine, or to chlordimeform alone, it was found that a
significant excess of risk of bladder cancer also occurred in those
workers thought not to have been exposed to 4-chloro- o-toluidine. In
this group of 592 men, 5 cases of bladder cancer were observed, while
1.4 cases were expected (SIR = 3.5, 95% CI (1.1, 8.3)). The authors
concluded that despite the lack of information on potentially
confounding factors in this study, the data indicated an association
between excess risk of bladder cancer in this cohort and one or more
aspects of the manufacture of chlordimeform.
Table 23. Bladder cancer risk in a cohort of men exposed to
chlordimeform (Boyle & Macfarlane, 1997)
Plant location Cohort numbers Bladder cancer cases
Observed Expected SIRa
Switzerland 273 4 0.72 5.6
USA (A)b 182 1 0.32 3.1
United Kingdom 174 3 1.06 2.8
USA (B)b 163 1 0.26 3.8
Australia 55 1 0.27 3.7
All plants 847 10 2.63 3.8
95% CIc
(1.8, 7.1)
a Standardized Incidence Ratio
b Different production sites
c Confidence Interval
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1 Laboratory experiments
9.1.1 Microorganisms
There are no data on the effects of chlordimeform on
microorganisms.
9.1.2 Aquatic organisms
9.1.2.1 Plants
There are no data on the effects of chlordimeform on aquatic
plants.
9.1.2.2 Invertebrates
There are no laboratory data on the effects of chlordimeform on
aquatic invertebrates.
9.1.2.3 Vertebrates
The toxicity of chlordimeform to some species of fish has been
determined (FAO/WHO, 1972; Mayer & Ellersieck, 1986), and is shown in
Table 24.
9.1.3 Terrestrial organisms
9.1.3.1 Plants
There are no data available for the effects of chlordimeform on
plants.
9.1.3.2 Invertebrates
Dittrich (1966, 1967) first reported studies on the efficacy of
chlordimeform as an acaricide with both ovicidal activity against
insect eggs and adulticidal activity. It killed adult spider mites
when applied as a vapour and as a spray, and penetrated plant tissues
where it was released in ovicidal quantities. Since then, its efficacy
as an insecticide has been studied in a wide range of species.
Chlordimeform not only has a direct lethal action, particularly
against eggs and early instar larvae of insects and acarines, but also
has important sublethal effects, including sterilization of eggs,
induction of hyperactivity, detachment of feeding ticks from hosts,
Table 24. Toxicity of chlordimeform to fish
Species Duration LC50 Reference
(h) (mg/litre)
Bluegill sunfish 24 1.0 FAO/WHO (1972)
48 1.0
96 1.0
Trout 24 11.7 (8.73-15.8) FAO/WHO (1972)
48 10.6 (7.80-14.50)
96 7.14 (4.70-10.80)
Cat fish 24 11.9 (8.98-15.9) FAO/WHO (1972)
48 8.72 (6.26-21.1)
96 4.54 (3.08-6.68)
Rainbow trout 24 29 Mayer & Ellersiek
96 13.2 (1986)
Channel catfish 24 20.7 Mayer & Ellersiek
96 20.2 (1986)
Carp 24 65a FAO/WHO (1972)
48 60a
96 50a
a Values are for TLm
colony dispersal behaviour in ticks and mites, anti-feeding effects
and disruption of mating and oviposition in Lepidoptera (Hollingworth,
1976).
Knowles & Shrivastava (1973) investigated its toxicity in house
flies. The LD50 was 25 �g/fly, a dose which was not at a practical
level for house-fly control, possibly due to the high rate of
metabolism in this species. Pimley (1986) investigated the toxicity of
chlordimeform to tsetse fly (Glossina morsitans). The median lethal
dose was approximately 0.4 �g/fly for unfed tsetse, and 100% mortality
was achieved with 2 �g/fly. Sublethal doses also caused a significant
depression of feeding activity.
The specificity of chlordimeform with regard to both eggs and
larval stages was examined by Streibert & Dittrich (1977). Eggs of the
three noctuid cotton pest moths, Heliothis armigera, Heliothis
virescens, and Spodoptera littoralis, when exposed to a saturated
atmosphere of 4 mg/m3, have very similar sensitivity to vaporized
chlordimeform. Agrotis ipsilon, also a noctuid, on the other
hand, is definitely less sensitive, and the coccinillid Epilachna
varivestis was the most tolerant. The larval stages of all of these
species were considerably less sensitive to chlordimeform vapour than
the egg stage, but chlordimeform does seem to cause a decrease in the
number of larvae in the field, possibly due to a repellent effect or a
behavioural change rather than a direct toxic effect. These results
with Spodoptera littoralis on the relative sensitivity of eggs and
larvae were confirmed in the studies of Salvisberg et al. (1980).
Davenport & Wright (1985) have also demonstrated the differential
susceptibility of adult and larvae of the noctuid moths, Spodoptera
littoralis and Heliothis virescens, and also highlighted the
significantly higher toxicity of the hydrochloride salt, compared to
the base, to the adults of both species.
Sparks et al. (1993) studied the effects of several insecticides
on ovicidal activity and alteration of octopamine titres in eggs of
the tobacco budworm (Heliothis virescens). Chlordimeform was highly
toxic to eggs of H. virescens. The authors reported that the
alteration in the biogenic amine octopamine titres by chlordimeform
might lead to a disruption in the ability of larvae to hatch from the
egg.
Crecelius & Knowles (1976) studied the sensitivity of the larvae
of the cabbage looper, Trichoplusia ni, to the toxic effects of
chlordimeform. Third instar larvae were more sensitive to the toxic
effects of chlordimeform than the fifth instar larvae, possible due to
slower penetration and slower metabolism of chlordimeform in the
latter instar larvae.
Bailey & Cathey (1985) demonstrated the effectiveness of
chlordimeform in reducing the percentage egg hatch of Lygus
lineolaris (Palisot de Beauvois) on pole bean ( Phaseolus
vulgaris L.) pods and cotton ( Gossypium hirsutum L.). A solution
of 0.09% chlordimeform, while not significantly reducing nymph
emergence from eggs deposited on bean pole pods, did significantly
reduce nymph emergence from eggs deposited on cotton plants.
Salvisberg et al. (1980) also demonstrated that Spodoptera
littoralis moths, when treated at doses as low as 10% of the LD50,
showed symptoms of hyperexcitation, which resulted in abnormal
patterns of egg-laying, a reduced number of eggs and lower fertility.
Further studies by Davenport & Wright (1987) on Spodoptera
littoralis have shown that chlordimeform hydrochloride significantly
reduces food consumption in fifth-instar larvae when incorporated into
the diet at a level of 0.1-10 mg/g or when topically applied. No
mortality occurred during feeding, but mortality was increased during
subsequent pupation and during emergence of the adult from the pupae.
In adult moths, egg laying was significantly decreased when
chlordimeform hydrochloride was applied topically (1 or 10 �g/moth).
Further evidence that behavioural changes may be more important
in reducing both the larval and insect populations following
chlordimeform treatment has been provided by Shimizu & Fukami (1983)
in studies of the larvae of the cabbage armyworm, Mamestra
brassicae, which showed a prolonged period of wandering behaviour in
the presence of chlordimeform. This may have caused a failure to find
or prepare a suitable site for pupation.
The behaviour-modifying effects of chlordimeform have also been
studied by Blackwell (1988a,b; 1889) in the larvae of the large
cabbage white butterfly, Pieris brassicae L. When placed on
chlordimeform-dipped leaves, the larvae become excited, in contrast to
their normal communal feeding behaviour. Locomotion was increased and
feeding was significantly reduced as a result of disaggregation
(Blackwell, 1988a). When applied directly to the larvae, chlordimeform
caused excitation and inhibition of feeding (Blackwell, 1989). Direct
application also caused developmental delays and mortality was
increased at later developmental stages (Blackwell, 1988b).
O'Brian et al. (1985) have studied the effect of insecticides on
beneficial insects, and in particular, the effect of chlordimeform on
the ecoparasitoid, Bracon mellitor, an important parasitoid of the
boll weevil (Anthonomus grandis grandis). Chlordimeform was found to
be more toxic to Bracon mellitor than to the boll weevil, and also
reduced the number of egg deposited.
The toxicity of chlordimeform hydrochloride to bees has been
examined after both ingestion and contact. Ingestion of a 0.3%
solution killed 18%, while ingestion of 0.15% killed approximately 7%.
Contact with the same solutions did not increase the mortality rate
(FAO/WHO, 1972). In a study by Johansen (1972), bees were exposed to
field-weathered residues of a range of insecticides, including
chlordimeform, on alfalfa foliage. Over a 24-h period, zero mortality
was obtained with alfalfa leafcutter bees, alkali bees and honey bees
exposed to 3-h-old residues.
9.1.3.3 Vertebrates
Fleming et al. (1985) examined the toxic and behavioural effects
of chlordimeform on the game bird, the bobwhite quail (Colinus
virginianus). When added to the diet of newborn chicks over a 7-day
period, the lethal concentration to chicks was 2835 mg/kg diet
(2169-3705 mg/kg diet). When chicks were fed a diet containing
chlordime-form at a concentration of 1000 mg/kg diet for 7 days, they
ate less, weighed less, travelled further from a fright stimulus in an
avoidance test, and had a high locomotor activity in an open-field
test than at lower dose levels. Chicks fed 100 or 1000 mg
chlordimeform/kg diet scored more highly than controls in a visual
cliff performance test. After a further 8 days on control diet, the
chicks fed 1000 mg/kg diet still scored higher than controls on the
avoidance test, but the open-field and cliff performance scores were
similar to those of controls.
In studies conducted on bobwhite quails and ducks, groups of
animals (10 per treatment group, 30 per control group) were fed
chlordimeform technical or chlordimeform 48% EC formulation in their
diets for 5 consecutive days. The dose levels were 0, 10, 31.6, 100,
316 or 1000 mg/kg diet. Both quails and ducks were tolerant of the
presence of chlordimeform in the diet. With the technical material,
one quail in each of the groups fed 100 and 316 mg/kg diet died,
while, with the formulation, one quail in each of the groups fed 316
and 1000 mg/kg diet died. All ducks survived treatment, even at the
highest dose level (FAO/WHO, 1972).
Hill et al. (1975) exposed three bird species, Japanese quail
(Coturnix japonica), ring-necked pheasant (Phasianus colchicus)
and mallard (Anas platyrhynchos), to chlordimeform. LC50 values for
Japanese quail and ring-necked pheasant were determined to be 1749 and
2608 mg/kg diet, respectively. The LC50 for mallard was determined to
be >5000 mg/kg diet; only 20% mortality was reported at the highest
exposure group, 5000 mg/kg diet. Hill & Camardese (1986) reported an
LC50 of 5079 mg/kg diet for Japanese quail exposed to chlordimeform.
9.2 Field Observations
9.2.1 Microorganisms
There are no field data on the effects of chlordimeform on
microorganisms.
9.2.2 Aquatic organisms
There are no field data on the effects of chlordimeform on
aquatic organisms.
9.2.3 Terrestrial organisms
9.2.3.1 Plants
The possibility that some insecticides might enhance the growth
of cotton plants has been suggested for some time. However, in the
case of chlordimeform, debate has continued as to whether this effect
is due to early season insect suppression (Bailey & Cathey, 1985) or
to a physiological effect (Phillips et al., 1977). Cathey & Bailey
(1987) have conducted controlled studies to examine the effects of
multiple applications of chlordimeform on the growth and development
of cotton ( Gossypium hirsutum L.) in both greenhouse and field
studies. Plants were sprayed six times with chlordimeform either alone
or in combination with fenvalerate at 5- to 7-day intervals, beginning
at the six-leaf stage of plant development. In the absence of early
season insects and when insect populations were maintained at a
relatively low level, no increases in lint yield occurred on the
chlordimeform-treated plants. However, yield increases did occur and
insect populations became lower in these treated plots when early
season insect populations in the test area were relatively high. None
of the treatments influenced the boll components, boll size, seed
index and lint percentage, or the first fibre properties, length,
strength and micronaire.
Field studies by Youngman et al. (1990) to determine the effects
of several insecticides on growth, fruiting patterns and yield of the
cotton plant, Gossypium hirsutum L., supported the conclusion that
chlordimeform does not significantly increase any plant growth
parameter when compared with the control.
9.2.3.2 Invertebrates
In a small field study conducted by Bull & House (1978), tests
were conducted in 0.05-ha plots of cotton to compare lower and more
frequent applications of chlordimeform with commercial mixtures of
insecticides against natural populations of Heliothis species. The
results indicated that the protection afforded was as good as with
commercial mixtures, probably as a result of careful observation of
the cotton to pinpoint the onset of significant egg production.
In a another small field study by Wilson (1981), the potential of
chlordimeform to control Heliothis species in cotton was tested
separately or in combination with amitraz and the microbial
insecticide, Bacillus thuringiensis. Chlordimeform was the most
efficient of the three materials and controlled Heliothis species
reasonably efficiently, but no control of the rough bollworm,
Earias hueglei was obtained. There was also good control of the
cotton looper, Anomis flava, and some indication of suppression of
mites and aphids was obtained.
The behaviour-modifying effects of chlordimeform have been
demonstrated in field studies by Uk & Dittrich (1986) on the adult
whitefly, Bemisia tabaci (Genn.), which attacks cotton in the Sudan.
At dose levels of 500-2500 g chlordimeform/ha together with 960 g
endosulfan/ha, there was evidence of irritation and mass emigration of
adults from treated cotton foliage without detectable direct
mortality.
9.2.3.3 Vertebrates
There are no field data on the effects of chlordimeform on
vertebrates.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
Production and use of chlordimeform has now ceased worldwide
and no further human exposure should occur. During the years of
chlordimeform production and use, dietary and incidental exposure to
chlordimeform occurred. Occupational exposure to chlordimeform and
4-chloro- o-toluidine (used as a starting material for synthesis)
occurred during manufacture and formulation, as well as during
application. The major route of exposure was through dermal
contamination. Application of chlordimeform occurred extensively by
aerial spraying, but it was also applied by ground-rigs as well as by
backpack spray equipment. Thus, agricultural workers were exposed
during mixing, loading, washing, and flagging operations. General
population exposure occurred through the consumption of food
containing residues of chlordimeform, and to a lesser extent through
by-stander exposure. In some cases, there was intentional ingestion of
the formulation.
Monitoring of urinary 4-chloro- o-toluidine has been found to be
a useful indicator of exposure.
10.1.2 Toxicity
In both experimental animals and humans, there is extensive
metabolism of chlordimeform, followed by rapid excretion via the
urine. A major urinary metabolite is 4-chloro- o-toluidine. In
experimental animals, symptoms of acute toxicity included neurotoxic
as well as cardiovascular effects. There was no evidence of
teratogenicity or reproductive effects. Following chronic
administration, there was a dose-related increase in
haemangioendotheliomas in mice. There was no treatment-related
increase in tumour incidence in rats. Most of the mutagenicity studies
with chlordimeform itself were negative, but there were sporadic
reports of genotoxicity with 4-chloro- o-toluidine and to a more
limited extent with N-formyl-4-chloro- o-toluidine.
In humans, chlordimeform has been shown to have both acute and
chronic effects. Acute poisoning caused fatigue, nausea and loss of
appetite, and, in more severe cases, somnolence, cyanosis, urgency in
urination, cystitis, cardiovascular effects (tachycardia, bradycardia,
ECG changes), coma and shock. The significance of the cardiovascular
effects in chlordimeform-induced mortality has only recently been
recognized. While there have been fatalities as a result of acute
chlordimeform exposure, in the majority of cases complete recovery
occurs. Symptoms of chronic exposure include those of acute exposure
as well as abdominal pain, skin itching and rashes, and gross or
microscopic haematuria.
With regard to carcinogenicity, the International Agency for
Research on Cancer (IARC) has concluded that there is limited
evidence in humans and sufficient evidence in experimental animals
for the carcinogenicity of 4-chloro- o-toluidine. The available
epidemiological data indicate an association between excess risk of
bladder cancer and exposures entailed in the manufacture of
chlordimeform. There is currently preliminary epidemiological evidence
of an association between chlordimeform use and excess risk of bladder
cancer.
10.1.3 Risk evaluation
With the withdrawal of the use of chlordimeform in agriculture
and a cessation of production worldwide, there is no longer any risk
associated with acute exposure except during the disposal of existing
stocks. The risk associated with chronic exposure, however,
particularly the risk of bladder cancer, will continue to be of
concern for many years. Human bladder cancer has a long latency
period, and establishing whether or not there is a link between
chlordimeform exposure and bladder cancer will require continued
health screening of significantly exposed individuals both from
manufacturing plants and from those rural communities where
chlordimeform was extensively used.
10.2 Evaluation of effects on the environment
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
11. CONCLUSIONS AND RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
AND THE ENVIRONMENT
11.1 Conclusions
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine, and, to a
lesser extent, chlordimeform.
Chlordimeform does not persist in the environment and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
11.2 Recommendations for protection of human health and the
environment
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
12. FURTHER RESEARCH
The following studies are needed:
1. epidemiological investigations on exposed populations.
2. studies on the dose-response relationship between exposure to
4-chloro- o-toluidine or chlordimeform and the induction of
urinary bladder cancer in humans.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Chlordimeform was considered by the International Agency for
Research on Cancer (IARC) in 1983. IARC noted that no published study
on the carcinogenicity of chlordimeform was available. However, it
considered data on the carcinogenicity of 4-chloro- o-toluidine and
concluded that the results of experiments on mice provided sufficient
evidence that 4-chloro- o-toluidine, a metabolite of chlordimeform,
is carcinogenic to experimental animals. No relevant data on humans
were available. IARC concluded the available data were inadequate to
evaluate the carcinogenicity of chlordimeform to humans (IARC, 1983).
The carcinogenicity of 4-chloro- o-toluidine, the breakdown
product and major metabolite of chlordimeform, was evaluated by IARC
in 1990 (IARC, 1990). On the basis of the available published data, it
was concluded that there is limited evidence in humans and
sufficient evidence in experimental animals for the carcinogenicity
of 4-chloro- o-toluidine. 4-chloro- o-toluidine and its strong acid
salts were classified as probably carcinogenic to humans (Group 2A).
Chlordimeform was considered at the 1971, 1975, 1978, 1979, 1980,
1985 and 1987 FAO/WHO Joint Meeting on Pesticide Residues (JMPR). In
1971, a temporary acceptable daily intake (ADI) for chlordimeform of
0-0.01 mg/kg body weight was established, and temporary maximum
residue levels (MRLs) were set for a number of crops and for the meat
and milk of cattle (FAO/WHO, 1972). In 1975, the temporary ADI was
maintained and some new temporary MRLs were established (FAO/WHO,
1976). In 1978, the temporary ADI was reduced to 0-0.0001 mg/kg body
weight, the temporary MRLs for all crops except cotton and cottonseed
were withdrawn, and the MRLs for meat and milk of cattle were set at
the level of detection (FAO/WHO, 1979). In 1979, 1980 and 1985, the
temporary ADI of 0-0.0001 mg/kg body weight was extended (FAO/WHO,
1980, 1981, 1986). In 1987, the temporary ADI for chlordimeform was
withdrawn (FAO/WHO, 1988).
REFERENCES
Abo-Khatwa N & Hollingworth RM (1972a) Chlordimeform: The relation of
the mitochondrial uncoupling to toxicity in the German cockroach. Life
Science, 11: 1181-1190.
Abo-Khatwa N & Hollingworth RM (1972b) Chlordimeform: Uncoupling
activity against rat liver mitochondria. Pest Biochem Physiol,
3: 358-369.
Ahmad S & Knowles CO (1971), Metabolism of N'-(4-chloro-o-tolyl)-
N,N-dimethyl-formamidine (chlorphenamidine) and 4'-chloro-o-
formotoluidine by rat hepatic microsomal and soluble enzymes.
Camp Gen Pharmacol, 2:189-197.
Anonymous (1971 a) Gas chromatographic method for the determination of
C 8514 residues in animal tissues, fat, milk and butter. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Anonymous (1971b) Chlordimeform: Working paper prepared for the 1971
Joint FAO/WHO Meeting on Pesticide Residues. Basel, Switzerland,
Ciba-Geigy Ltd and Berlin, Germany, Schering AG (Unpublished
document).
Anonymous (1977) [Report of two cases of chemical cystitis.] New Med,
8: 82. (in Chinese with English abstract).
Anonymous (1980a) CGA-95119: High performance liquid chromatographic
determination of urinary residues containing the 4-chloro-o-toluidine
moiety - Procedure No. 177. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Anonymous (1980b) [Study on the fate of chlordimeform residue on green
cabbage.] Shanghai, Shanghai Institute of Pesticides, Basic Metabolism
Group (in Chinese with English abstract).
Anonymous (1981) [On the residue of chlordimeform: Special Task Group
report.] In: [Selected papers on research on pesticides.] In: Chinese
Institute of Scientific Information and Bureau of Science and
Technology ed. [Selected papers on studies of pesticides.] Beijing,
Ministry of Chemistry (in Chinese with English abstract).
Anonymous (1985a) [Five years follow-up observation on the health
effect of chlordimeform on the employees in manufacturing.] Lab Med,
2: 7-10 (in Chinese with English abstract).
Anonymous (1985b) Chlordimeform: Joint submission to the WHO expert
group of the 1985 FAO/WHO Joint Meeting on Pesticide Residues. Basel,
Switzerland, Ciba-Geigy Ltd and Berlin, Germany, Schering AG
(Unpublished report).
Arima T, Morooka H, Tanigawa T, Imai M, Tsunashima T, & Kita S (1976)
Methemoglobinemia induced by chlorphenamidine. Acta Med Okayama.
30: 57-60.
Armstrong J, Graves O, Lovejoy G, Swiggart R, & Kimbrough R (1975)
Insecticide induced acute haemorrhagic cystitis. Morbid Mortal Wkly
Rep (Centres for Disease Control), 24: 374.
Arni P & M�ller D (1976a) Salmonella/mammalian-microsome mutagenicity
test with C8513 (chlordimeform HC1), Basel, Switzerland. Ciba-Geigy
Ltd (Unpublished report).
Arni P & M�ller D (1976b) Salmonella/mammalian-microsome mutagenicity
test with CGA 72647 (4-chloro-o-toluidine HCI), Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1976c) Salmonella/mammalian-microsome mutagenicity
test with CGA 72651 (N-formyl-4-chloro-o-toluidine), Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983a) C8513: Intrasanguine host-mediated assay
with S. typhimurium, Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Arni P & M�ller D (1983b) CGA 72647: Intrasanguine host-mediated assay
with S. typhimurium, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983c) C8513: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983d) CGA 72647: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983e) CGA 72651 pure: Chromosome studies in male
germinal epithelium, mouse spermatocytes. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Arni P, Hool G, & M�ller D (1983a) C8513: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P, Hool G, & M�ller D (1983b) CGA 72651: Chromosome studies in
male germinal epithelium, mouse spermatogonia. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Aziz SA & Knowles CO (1973) Inhibition of monoamine oxidase by the
pesticide chlordimeform and related compounds. Nature (Lond),
242: 417-418.
Bailey JC & Cathey GW (1985) Effect of chlordimeform on tarnished
plant bug (Heteroptera: Miridae) nymph emergence. J Econ Entomol,
78: 1485-1487.
Bailey BA, Martin RJ, & Downer RGH (1982) Monoamine oxidase inhibition
and brain catecholamine levels in the rat following treatment with
chlordimeform. Pest Biochem Physiol, 17: 293-300.
Balu K (1989) Evaluation of field worker exposure to chlordimeform by
using urine monitoring. In: Biological monitoring for pesticide
exposure: Measurement, estimation of risk reduction. Washington, DC,
American Chemical Society, pp 47-55 (ACS Symposium Series 382).
Bauer PJ & Cothren JT (1990) Growth-promoting activity of
chlordimeform. Agron J, 82: 73-75.
Beeman RW & Matsumura F (1973) Chlordimeform: A pesticide acting upon
amine regulatory mechanisms. Nature (Lond), 242: 273-274.
Beilstein P & M�ller D (1983) C8513 pure: BALB/3T3 cell transformation
assay. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Beilstein P & M�ller D (1984a) C8513 technical: L-5178Y/TK+/- mouse
lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Beilstein P & M�ller D (1984b) CGA 72647 technical: L1578Y/TK+/-
mouse lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Beilstein P & M�ller D (1984c) CGA 72647 technical: BALB/3T3 cell
transformation assay. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Benedict JH, Walmsley MH, Segers JC, & Treacy MF (1986) Yield
enhancement and insect suppression with chlordimeform (Fundal) on
dryland cotton. J Econ Etomol, 79: 238-242.
Benezet HJ & Knowles CO (1976) N'-(4-chloro-o-tolyl)-N-
methylformamidine (demethylchlordimeform) metabolism in the rat.
J Agric Food Chem, 24: 152-154.
Benezet HJ & Knowles CO (1981) Degradation of chlordimeform by algae.
Chemosphere, 10: 909-917.
Benezet HJ, Chang K, & Knowles CO (1978) Formamidine pesticides -
metabolic aspects of neurotoxicity. In: Pesticide venom neurotoxic,
pp 189-206.
Bentley P, Staubli W, Bieri F, Muecke W, & Waechter F (1985) Induction
of hepatic drug-metabolising enzymes following treatment of rats and
mice with chlordimeform. Toxicol Lett, 28: 143-149.
Bentley P, Bieri F, Muecke W, Waechter F, & Staubli W (1986a) Species
differences in toxicity of p-chloro-o-toluidine to rats and mice:
Covalent binding to hepatic macromolecules and hepatic non-parenchymal
cell DNA and an investigation of effects upon the incorporation of
[3H]-thymidine into capillary endothelial cells. Chem-Biol Interact,
57: 27-40.
Bentley P, Waechter F, Bieri F, Staubli W, & Muecke W (1986b) Species
differences in the covalent binding of p-chloro-o-toluidine to DNA.
Arch Toxicol, 9(suppl): 163-166.
Blackmore RH (1969a) Chlorphenamidine (Schering 36268): 2-year
repeated feeding study in dogs (Project No. 728-127). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969b) Chlorphenamidine (Schering 36268): 24-month
repeated feeding study in rats (Project No. 728-126). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969c) Chlorphenamidine (Schering 36268): three
generation reproduction study (Project No. 728-118). Falls Church,
Virginia, Hazleton Laboratories Inc (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969d) Chlorphenamidine (Schering 36268). Segment II:
Teratology Study -Rabbits (Project No. 728-157). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackwell A (1988a) Survival and development of Pieris brassicae
larvae upon application of sublethal doses of chlordimeform. Entomol
Exp Appl, 48: 149-156.
Blackwell A (1988b) The disruption of aggregation behaviour in Pieris
brassicae larvae by chlordimeform. Entomol Exp Appl, 48: 141-147.
Blackwell A (1989) The effects of chlordimeform on the behaviour of
Pieris brassicae larvae. Pestic Sci, 26: 11-19.
Blass W (1972a) Galecron residues in Japanese tea. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. RVA 5/72).
Blass W (1972b) Galecron residues in Swiss grapes (berries and juice).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 11/72).
Blass W (1972c) Galecron residues in Swiss wine 1971. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 16/72).
Bokisch AJ, Cox RT, & Walker RJ (1985) The actions of formamidine
pesticides on identified central neurons of Helix aspersa. Pestic:
Sci, 16: 495-503.
Boyes WK & Dyer RS (1984) Chlordimeform produces profound, selective,
and transient changes in visual evoked potentials of hooded rats. Exp
Neurol, 86: 434-447.
Boyes WK & Moser VC (1988) An alpha 2-adrenergic mode of action of
chlordimeform on rat visual function. Toxicol Appl Pharmacol,
92: 402-418.
Budris DM, Yim GK, Carlson GP, & Schnell RC (1983) Effect of acute and
repeated chlordimeform treatment on rat hepatic microsomal drug
metabolizing enzymes. Toxicol Lett, 18: 63-71.
Budris DM, Yim GK, & Schnell RC (1984) Effect of acute and repeated
chlordimeform treatment on rat hepatic microsomal drug metabolizing
enzymes. J Environ Pathol Toxicol Oncol, 5: 175-182.
Bull DL (1973) Metabolism of chlordimeform in cotton plants. Environ
Entomol, 2: 869-871.
Bull DL & House VS (1978) Effects of chlordimeform on insects
associated with cotton. Southwest Entomol, 3: 284-291.
Burkhard N (1971) Residues in milk, meat, liver and fat of a Swiss
cow. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
14/71).
Campanhola C & Plapp FW Jr (1989) Pyrethroid resistance in the tobacco
budworm (Lepidoptera: Noctuidae): Insecticide bioassays and field
monitoring. J Econ Entomol, 82: 22-28.
Candura SM, Marraccini P, Costa LG, Manzo L, Rossi A, Coccini T, &
Toninin M (1992) Calcium entry blockade as a mechanism for
chlordimeform induced inhibition of motor activity in the isolated
guinea-pig ileum. Pharmacol Toxicol, 71: 426-433.
Cathey GW & Bailey JC (1987) Evaluation of chlordimeform for cotton
yield enhancement. J Econ Entomol, 80: 670-674.
Chan Y-M (1985) [Observation and nursing of 6 cases of chlordimeform
poisoning.) Chin J Nurs, 20: 68-69 (in Chinese with English abstract).
Cheung MW, Kahrs RA, Nixon WB, Ross JA, & Tweedy BG (1989)
Determination of chlordimeform residues as 4-chloro-o-toluidine in
human urine using high-performance liquid chromatography. In:
Biological monitoring of pesticide exposure: Measuring and estimating
risk reduction. Washington, DC, American Chemical Society, pp 231-239
(ACS Symposium Series 382).
Cooper RL, Barrett MA, Goldman JM, Rehnberg GL, McElroy WK, & Stoker
TE (1994) Pregnancy alterations following xenobiotic-induced delays in
ovulation in the female rat. Fundam Appl Toxicol, 22: 474-480.
Costa LG & Murphy SD (1987) Interaction of the pesticide chlordimeform
with adrenergic receptors in mouse brain: an in vitro study. Arch
Toxicol, 59: 323-327.
Costa LG, Wu S, Olibet G, & Murphy SD (1988) Alpha2-adrenoceptors as
a target for formamidine pesticides: In vitro and in vivo studies in
mice. Toxicol Appl Pharmacol, 93: 319-328.
Costa LG, Wu S, Olibet G, & Murphy SD (1989) Formamidine pesticides
and alpha2-adrenoceptors: Studies with amitraz and chlordimeform in
rats and development of a radioreceptor binding assay. Neurotoxicol,
11: 405-411.
Costa LG, Gastel J, & Murphy SD (1991) The formamidine pesticides
chlordimeform and amitraz decrease hepatic glutathione in mice through
an interaction with alpha2-adrenoceptors. J Toxicol Environ Health,
33: 349-358.
Coye MJ, Lowe JA, & Maddy KJ (1986) Biological monitoring of
agricultural workers exposed to pesticides: II. Monitoring of intact
pesticides and their metabolites. J Occup Med, 28: 628-636.
Crecelius CS & Knowles CO (1976) Toxicity, penetration, and metabolism
of chlordimeform and its N-demethyl metabolite in cabbage looper
larvae. J Agric Food Chem, 24: 1018-1023.
Crowder LA & Whitson RS (1980) Fate of toxaphene, methyl parathion,
and chlordimeform combinations in the mouse. Bull Environ Contam
Toxicol, 24: 444-451.
Currie AN (1933) Chemical haematuria from handling 5-chloro-ortho-
toluidine. J Ind Hyg, 15: 205-213.
Davenport AP & Wright DJ (1985) Toxicity of chlordimeform and amitraz
to the Egyptian cotton leafworm (Spodoptera littoralis) and the
tobacco budworm (Heliothis virescens). Pestic Sci, 16: 81-87.
Davenport AP & Wright DJ (1987) Effect of the formamidine insecticide
chlordimeform on feeding and reproduction in the noctuid moth,
Spodoptera littoralis (Boisduval). Crop Prot, 6: 3-6.
Deng C-S, Zhang J-K, & Guo J-Y (1984) [Brief report of the symposium
on chlordimeform poisoning.] J Clin Med, 1: 51-53 (in Chinese with
English abstract).
Ding M-B & Ru X-Y (1985) [On diagnosis and treatment of chlordimeform
poisoning (analysis of 358 cases).] Jiangsu Med, 7: 30-31 (in Chinese
with English abstract).
Ding Y & Huang K-L (1987) [Analysis on clinical features of
chlordimeform poisoning.] Labor Med, 4: 8-10 (in Chinese with English
abstract).
Dittrich V (1966) N-(2-methyl-4-chlorophenyl)-N',N'-
dimethylformamidine (C-8514/Schering 36268) evaluated as an
acaricide. J Econ Entomol, 59: 889-893.
Dittrich V (1967) Systemic action of N-(2-methyl-4-chlorophenyl)-N',
N'-dimethylformamidine (C-8514/Schering 36268). J Econ Etomol,
60: 558-569.
Dittrich V & Loncarevic A (1971) New insecticides for Asiatic rice
borer control in paddy rice. J Econ Entomol, 64: 1225-1229.
Dyer RS & Boyes WK (1983) Chlordimeform differentially affects pattern
reversal-evoked and flash-evoked potentials in rats. Toxicologist,
3: 13.
Ehrhardt DA & Knowles CO (1970) Metabolism and translocation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine (chlorphenamidine) and
its hydrochloride salt in grapefruit seedlings. J Econ Entomol,
63: 1306-1314.
Ercegovich CD, Witkonton S. & Asquith DJ (1972) Disappearance of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine from six major fruit
crops. J Agric Food Chem, 20: 565-568.
Evans PD & Gee JD (1980) Action of formamidine pesticides on
octopamine receptors. Nature (Lond), 287: 60-62
Fan DF & Ge SD (1982) Gas-liquid chromatographic determination of
chlordimeform and its metabolites in cargo rice and husk. J Assoc Off
Anal Chem, 65: 1517-1520.
FAO/WHO (1972) Chlordimeform. In: 1971 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 3-45 (WHO
Pesticide Series, No. 1).
FAO/WHO (1976) Chlordimeform. In: 1975 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 85-104 (WHO
Pesticide Residues Series, No. 5).
FAO/WHO (1979) Chlordimeform. In: Pesticides residues in food - 1978:
Evaluations, Rome, Food and Agriculture Organization of the United
Nations, PP 51-78 (FAO Plant Production and Protection Paper 15 Sup.).
FAO/WHO (1980) Chlordimeform. In: Pesticide residues in food - 1979:
Evaluations. Rome, Food and Agriculture Organization of the United
Nations, pp 129-135 (FAO Plant Production and Protection Paper 20
Sup.).
FAO/WHO (1981) Chlordimeform. In: Pesticide residues in food - 1980:
Evaluations. Rome. Food and Agriculture Organization of the United
Nations, pp 89-96 (FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1986) Chlordimeform. In: Pesticide residues in food - 1985:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 17-29 (FAO Plant Production and
Protection Paper 72/2).
FAO/WHO (1988) Chlordimeform. In: Pesticide residues in food - 1987:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 25-26 (FAO Plant Production and
Protection Paper 86/2).
Fleming WJ, Heinz GH, & Schuler CA (1985) lethal and behavioral
effects of chlordimeform in bobwhite. Toxicology, 36: 37-47.
Folland DS, Kimbrough RD, Cline RE, Swiggart RC, & Schaffner (1978)
Acute hemorrhagic cystitis - Industrial exposure to the pesticide
chlordimeform. J Am Med Assoc, 239: 1052-1055.
Fritz H (1971) Reproduction study on C8514 (chlorphenamidine)
technical in rabbit - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 10071 004). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1975) Reproduction study on C8514 (chlorphenamidine)
technical in rat - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 227533). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1978a) C8513 technical: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327735).
Fritz H (1978b) CGA 72651: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327737).
Fritz H, Becker H, & M�ller D (1978) CGA 72647 technical: Dominant
lethal study (mouse). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782201).
Gaines TB & Linder RE (1986) Acute toxicity of pesticides in adult and
weanling rats. Fundam Appl Toxicol, 7: 299-308.
Gale GT Jr & Hofberg AH (1985) Gas chromatographic determination of
chlordimeform in pesticide formulations: Collaborative study. J Assoc
Off Anal Chem, 68: 589-592.
Galloway SM, Armstrong MJ, Reuben C, Colman S, Brown B, Cannon C,
Bloom AD, Nakamura F, Ahmed M, Duk S, Rimpo J, Margolin BH, Resnick
MA, Anderson B, & Zeiger E. (1987) Chromosome aberrations and sister
chromatid exchanges in Chinese hamster ovary cells: Evaluations of 108
chemicals. Environ Mol Mutagen, 10(suppl): 1-175.
Geissb�hler H, Kossmann K, Baunok I, & Boyd VF (1971) Determination
of total residues of chlorphenamidine in plant and soil material by
colorimetry of thin layer and electron capture gas chromatography.
J Agric Food Chem, 19: 365-371.
Geyer R & Fattal F (1987) HPLC determination of the metabolite
4-chloro-o-toluidine in the urine of workers occupationally exposed to
chlordimeform. J Anal Toxicol, 11: 24-26.
Ghali GZ & Hollingworth RM (1985) Influence of mixed function
oxygenase metabolism on the acute neurotoxicity of the pesticide
chlordimeform in mice. Neurotoxicology, 6: 215-238.
Gifkins MR & Jacobson RA (1980) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine (chlordimeform), C10H13CIN2.
Cryst Struct Commun, 9: 959-963.
Glowa JR (1986) Acute and sub-acute effects of deltamethrin and
chlordimeform on schedule-controlled responding in the mouse.
Neurobehav Toxicol Teratol, 8: 97-102.
Goldman JM, Cooper RL, Laws SC, Rehnberg GL, Edwards TL, McElroy WK, &
Hein JF (1990) Chlordimeform-induced alterations in endocrine
regulation within the male rat reproductive system. Toxicol Appl
Pharmacol, 104: 25-35.
Goldman JM, Cooper RL, Edwards TL, Rehnberg G, McElroy WK, & Hein JF
(1991) Suppression of the luteinizing hormone surge by chlordimeform
in ovariectomized, steroid-primed female rats. Pharmacol Toxicol,
68: 131-136.
Goldman JM, Stoker TE, Perreault SD, Cooper RL, & Crider MA (1993)
Influence of the formamidine pesticide chlordimeform on ovulation in
the female hamster - Dissociable shifts in the luteinizing hormone
surge and oocyte release. Toxicol Appl Pharmacol, 121: 279-290.
Gordon CJ & Watkinson WP (1988) Behavioral and autonomic
thermoregulation in the rat following chlordimeform administration.
Neurotoxicol Teratol, 10: 215-219.
Gordon CJ, Long MD, & Stead AG (1985) Thermoregulation in mice
following acute chlordimeform administration. Toxicol Lett, 28: 9-15.
Gross (1977) Metabolism of C 8514 (chlordimeform) in cotton. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 19/77).
Gr�bner P (1977) [Thin-layer chromatographic determination of
chlordimeform in cucumbers and apples.] Nahrung, 21: 339-345
(in German).
Gu Q, Kui N-D, Wang M & Xue S-Z (1987) [Report on a mild case of
chlordimeform poisoning.] J Environ Toxicol, 1: 61-62 (in Chinese with
English abstract).
Gu Y-E, Wang M, Gu Q, & Dai G-J (1991) [Epidemiologic study on the
cancer mortality of employees and their relatives in Xin-Yang farm.]
J Jiangsu Occup Health, 1: 5-8 (in Chinese).
Guth JA & Senn U (1969) Galecron - Dissipation in a Swiss soil 1969 as
determined by chemical analysis. Basel, Switzerland Ciba-Geigy Ltd
(Unpublished report).
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(suppl): 3-142.
He R-H (1989) [Analysis on the clinical symptoms of 52 acute
chlordimeform poisoned cases.] Occup Med, 16: 27 (in Chinese with
English abstract).
He D-Q, Peng B, & He P-L (1987) [Experience of treatment of
chlordimeform poisoning with atropine.] Hu-Nan Med, 4: 169 (in Chinese
with English abstract).
Henderson GR (1985) Report on the continuing use of chlordimeform for
cotton insect pest control by aerial and ground rig application in
1984/5 - NSW report. Pendle Hill, Ciba-Geigy Australia Ltd and
Kensington, Australia, Schering Pty Ltd (Unpublished report No. CGS
85/1).
Hermosin MC & Perez-Rodriguez JL (1981) Interaction of chlordimeform
with clay minerals. Clays Clay Miner, 29: 143-152.
Hill EF & Camardese ME (1986) Lethal dietary toxicities of
environmental contaminants and pesticides to Coturnix. Washington, DC,
US Department of the Interior, Fish and Wildlife Service, 147 pp (Fish
and Wildlife Technical Report No. 2).
Hill EF, Heath RG, Spann JW, & Williams JD (1975) Lethal dietary
toxicity of environmental pollutants to birds. Washington, DC, US
Department of the Interior, Fish and Wildlife Service, p 61 (Special
Scientific Report - Wildlife No. 191).
Hill DL, Shih T-W & Struck RF (1979) Macromolecular binding and
metabolism of the carcinogen 4-chloro-2-methylaniline. Cancer Res,
39: 2528-2531.
Hollingworth RM (1976) Chemistry, biological activity and uses of
formamidine pesticides. Environ Health Perspect, 14: 57-69.
Hollingworth RM, Leister J, & Ghali G (1979) Mode of action of
formamidine pesticides: an evaluation of monoamine oxidase as the
target. Chem-Biol Interact, 24: 35-49.
Holsapple MP & Yim GK (1981) Decreased gastrointestinal ulcerogenicity
of chlordimeform, a basic anti-inflammatory agent. Toxicol Appl
Pharmacol, 59: 107-110.
Homburger F, Friedell GH, Weisburger EK, & Weisburger JH (1972)
Carcinogenicity of simple aromatic amine derivatives in mice and Rats.
Toxicol Appl Pharmacol, 22: 280 (Abstract).
Honeycutt RC & Cassidy JE (1977) Extraction and characterization of
cottonseed metabolites from cotton injected with 14C-chlordimeform.
Greensboro, North Carolina, Ciba-Geigy Corporation (Unpublished report
No. ABR - 77087).
Hool G & Arni P (1983a) C8513 pure: Sister chromatid exchange studies
on somatic cells of Chinese hamster. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 830305).
Hool G & Arni P (1983b) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatogonia). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782902).
Hool G & Arni P (1983c) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782901).
Hool G & Arni P (1983d) CGA 72647 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 783101).
Hool G & Arni P (1983e) CGA 72647 pure: Sister chromatid exchange
studies on somatic cells of Chinese hamster. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 830312).
Hool G & Arni P (1983f) CGA 72651 pure: Chromosome studies in somatic
cells (Chinese hamster). Base], Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 774054).
Hool G & M�ller P (1978) C8513 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 40400577).
Hool G, Beilstein P, & Arni P (1983) C8513 technical: Chromosome
studies in male germinal epithelium (mouse spermatogonia). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 773062).
Hsu WH & Kakuk TJ (1984) Effect of amitraz and chlordimeform on heart
rate and pupil diameter in rats: Mediated by alpha2-adrenoreceptors.
Toxicol Appl Pharmacol, 73: 411-415.
Huang X-S, Ding S, & Chen P-H (1979) [Bioassay on the carcinogenicity
of chlordimeform.] In: Chinese Institute of Scientific Information and
Bureau of Science and Technology ed. [Selected papers on studies of
pesticides.] Beijing, Ministry of Chemical Industry, pp 789-792 (in
Chinese with English abstract).
Huang H-X, Xue K-L, & Shi G-J (1989) [Study on the status of pollution
on rice by chlordimeform.] Monograph submitted to the National
Pesticide Reviewing Committee (in Chinese with English abstract).
Hurni H & Sachsse K (1969) Report on the determination of the acute
oral LD50 to the dog of chlorfenamidin technical. Sisseln,
Switzerland, Tierfarm AG (Unpublished report).
IARC (1978) Some aromatic amines and related nitro compounds - hair
dyes, colouring agents and miscellaneous industrial chemicals. Lyon.
International Agency for Research on Cancer (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Man, Volume 16).
IARC (1983) Chlordimeform. Lyon, International Agency for Research on
Cancer, pp 61-72 (IARC Monographs on the Evaluation of Carcinogenic
Risk of Chemicals to Man, Volume 30)
IARC (1990) Some flame retardants and textile chemicals and exposures
in the textile manufacturing industry. Lyon, International Agency for
Research on Cancer, pp 123-137 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 48).
Ifflaender U (1977a) Distribution, degradation and excretion of
chlordimeform (CDF) in mice and rats. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 38/77).
Ifflaender U (1977b) Comparison of the urinary metabolite pattern of
mice and rats after oral application of "C-chlordimeform. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 39/77).
Iizuka H & Masuda T (1979) Residual fate of chlorphenamidine in rice
plant and paddy soil. Bull Environ Contam Toxicol, 22: 745-749.
IRPTC (1992) IRPTC data profile on chlordimeform. Geneva, United
Nations Environment Programme, International Register of Potentially
Toxic Chemicals.
Iwan J & Goller D (1975) Metabolic fate of chlordimeform in soil under
aerobic, anaerobic, and sterile conditions. Environ Qual Saf (Pestic),
3(suppl): 282-287.
Iwan J, Hoyer GA, Rosenberg D, & Goller D (1976) Transformations of
4-chloro-o-toluidine in soil: Generation of coupling products by
one-electron oxidation. Pestic Sci, 7: 621-631.
Janssen R, Boyes WK, & Dyer RS (1983) Effects of chlordimeform on the
brainstem auditory evoked response in rats. In: Hayes AW, Schnell RC,
& Miya TS ed. Developments in the Science and practice of toxicology,
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 533536.
Jiang J-K, Xu W-Y, Xu S-Q, Li J-L, Zhou F-D, Yang S-X, Zhong S-L, &
Zhu R-L (1985) [Survey on the hygienic status on spraying
chlordimeform with fine mist sprayer.] Labor Med, 2: 4-6 (in Chinese;
English translation).
Jiang X-Z, Li F, Xue S-Z, & Wang -L (1988) Carcinogenicity assay of
chlordimeform on dermal application in mice. Chin J Ind Hyg Occup Dis,
6: 138.
Johansen CA (1972) Toxicity of field-weathered insecticide residues to
four kinds of bees. Environ Entomol, 1: 393-394.
Johnson BT & Knowles CO (1970) Microbiological degradation of the
acaricide N-(4-chloro-o-tolyl)-N,N'-dimethylformamidine. Bull Environ
Contam Toxicol, 5: 158-170.
Johnson TL & Knowles CO (1981) Inhibition of rat platelet
5-hydroxytryptamine uptake by chlordimeform. Toxicol Lett, 9: 1-4.
Johnson TL & Knowles CO (1982) Interaction of formamidines with
the platelet 5-hydroxytryptamine uptake system. Gen Pharmacol,
13: 299-307.
Johnson TL & Knowles CO (1983) Influence of formamidines on biogenic
amine levels in rat brain and plasma. Gen Pharmacol, 14: 591-596.
Jurincic C, Horlbeck R, Metz K, & Klippel KF (1991) [Acute hemorrhagic
cystitis following chlordimeform poisoning.] Dtsch Med Wochenschr,
116: 816-818 (in German).
Kadir HA & Knowles CO (1981) Inhibition of rat brain monoamine oxidase
by insecticides, acaricides and related compounds. Gen Pharmacol,
12: 239-247.
Kale PG, Petty BT Jr, Walker S, Ford JB, Dehkordi N, Tarasia S, Tasie
BO, Kale R, & Sohni YR (1995) Mutagenicity testing of nine herbicides
and pesticides currently used in agriculture. Environ Mol Mutagen,
25: 148-153.
Kaloyanova F, Bainova A, & Zaprianov Z (1981) Inhibition of monoamine
oxidase activity after combined action of chlordimeform with the
antidepressant, nialamide. Arch Environ Contam Toxicol, 10: 1-8
Ke Z-H (1985) [Clinical features of 47 acute CDM poisonings by
swallowing.] Int Med, 11: 48-59 (in Chinese with English abstract).
Kenyon R, Garcia RJ, O'Grady HM, & Russell PJ (1993) A surveillance
programme for agricultural workers previously exposed to the suspect
bladder carcinogen chlordimeform - Proceedings of the 24th Congress of
the International Commission on Occupational Health, Nice, 26
September-1 October 1993.
Kimbrough RD (1980) Human health effects of selected pesticides,
chloroaniline derivatives. J Environ Sci Health, 15: 977-992.
Kimmel EC, Casida JE, & Ruzo LO (1986) Formamidine insecticides
and chloroacetanilide herbicides: Disubstituted anilines and
nitrosobenzenes as mammalian metabolites and bacterial mutagens.
J Agric Food Chem, 34: 157-161.
Knowles CO (1970) Metabolism of two accaricidal chemicals,
N'-(4-chloro-o-tolyl)-N, N-dimethylformamidine (chlorphenamidine)
and m-(((dimethylamino)methylene)amino)phenyl-carbamate hydrochloride
(formetanate). J Agric Food Chem, 18: 1038-1047.
Knowles CO (1976) Chemistry and toxicology of quinoxaline, organotin,
organofluorine, and formamidine acaricides. Environ Health Perspect,
14: 93-102.
Knowles CO (1991) Chlordimeform. In: Hayes WJ & Laws ER ed. Handbook
of pesticide toxicology. New York, London, San Francisco, Academic
Press Inc., vol 3. pp 1475-1486.
Knowles CO & Benezet HJ (1977) Mammalian metabolism of chlordimeform:
Formation of metabolites containing the urea moiety. J Agric Food
Chem, 25: 1022-1026.
Knowles CO & Johnson TL (1984) Effect of formamidines on
5-hydroxytryptamine uptake and biogenic amine levels in rat platelets.
Toxicology, 31: 91-98.
Knowles CO & Roulston WJ (1972) Antagonism of chlorphenaminine
toxicity to the cattle tick Boophilus microplus by piperonyl
butoxide. J Aust Entomol Soc, 11: 349-350.
Knowles CO & Sen Gupta AK (1969) Photodecomposition of the acaricide
N'-(4-chloro-o-tolyl)N,N-dimethylformamidine. J Econ Entomol,
62: 344-348.
Knowles CO & Sen Gupta AK (1970) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine-14C(Galecron) and 4-chloro-o-toluidine
14C metabolism in the white rat. J Econ Entomol, 63: 856-859.
Knowles CO & Shrivastava SP (1973) Chlordimeform and related
compounds: Toxicological studies with house flies. J Econ Entomol,
66: 75-79.
Konopka EA & Heymann H (1977) Salmonella/microsome mutagenicity test
with chlordimeform. Greensboro, North Carolina, Ciba-Geigy Corporation
(Unpublished report).
Kopp R, Humpel M, Kuhne G, Fuchs R, & Rzadkiewicz M (1977)
Pharmacokinetics of chlordimeform following single and repeated oral
administration of 0.03 and 3 mg in female mice (1.2 and 120 mg/kg).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Kossmann K (1980) Summary report on chlordimeform monitoring
programmes conducted by Ciba-Geigy and Schering at applicator sites in
1979. Berlin, Germany, Schering AG and Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report).
Kossmann K, Geissb�hler H, & Boyd VF (1971) Specific determination of
chlorphenamidine (N'(4-chloro-o-tolyl)-N,N-dimethylformamidine) in
plants and soil material by colorimetry and thin layer and electron
capture gas chromatography. J Agric Food Chem, 19: 360-364.
Krick F (1994) Method for the determination of chlordimeform in honey.
Washington, DC, US Food and Drug Administration (Laboratory
Information Bulletin 3817).
Kurtz PH, Shaw G, Kelter A, & Jackson RJ (1987) Assessment of
potential acute health effects in agricultural workers exposed during
the application of chlordimeform. J Occup Med, 29: 592-595.
Lang R (1984) The mammalian spot test and its use for testing of
mutagenic and carcinogenic potential: experience with the pesticide
chlordimeform, its principal metabolites and the drug lisuride
hydrogen maleate. Mutat Res, 135: 219-224.
Lang R & Adler ID (1982) Studies on the mutagenic potential of the
pesticide chlordimeform and its principal metabolites in the mouse
heritable translocation assay, Mutat Res, 92: 243-248.
Langauer M & M�ller D (1977) C8513 technical : Nucleus anomaly test in
somatic interphase nuclei (Chinese hamster). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 40050577).
Langauer M & M�ller D (1978a) CGA 72651 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 40550577).
Langauer M & M�ller D (1978b) CGA 72647 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 78/3002).
Leander J & MacPhail RC (1980) Effect of chlordimeform (a formamidine
pesticide) on schedule-controlled responding of pigeons. Neurobehav
Toxicol, 2: 315-321.
Leander JD, Tomlinson WT, Balster RL, & MacPhail RC (1984) Some
effects of the formamidine pesticide chlordimeform on the behaviour of
mice. Neurotoxicology, 5: 91-100.
Lemesch C, Wolf Y, & Gabai E (1987) Occupational exposure to
chlordimeform in Israel. Isr J Med Sci, 23: 1261-1262.
Leslie C, Reidy GF, Murray M, & Stacey NH (1988) Induction of
xenobiotic biotransformation by the insecticide chlordimeform, a
metabolite 4-chloro-o-toluidine and a structurally related chemical
o-toluidine. Biochem Pharmacol, 37: 2529-2535.
Li T-D, Liu B-J, & Mei C-J (1982) [Survey on 20 cases of acute
occupational chlordimeform poisoning.] Intermed Med, 3: 23-24
(in Chinese with English abstract).
Li F, Xue S-Z, Wang Y-L, Zhu S-Y, & Hu R-F (1985a) [Carcinogenic
action of chlordimeform given orally to mice in a lifetime
experiment.] Chin J Prev Med, 19: 154-156 (in Chinese with English
abstract).
Li F, Xue S-Z, Zhang R-W, Wang Y-L, Zhu Y-T, Sun S-S, Ding J-L, &
Feng B-J (1985b) (Studies on the health effect of employees in
chlordimeform production, and the mutagenic outcomes.] Labor Med,
2: 11-14 (in Chinese with English abstract).
Li F, Xue S-Z, Jiang X-Z, & Wang Y-L (1986) Report of dermal
carcinogenicity bioassay of chlordimeform in mice. J Ind Hyg Occup
Dis, 6: 76.
Limmer FB (1985) Ciba-Geigy and Schering world-wide exposure data
summary of personnel applying chlordimeform products covering the
period 1980-1984. Basel, Switzerland, Ciba-Geigy Ltd and Berlin,
Germany, Schering AG (Unpublished report).
Lin TH, North HH, & Menzer RE (1975) The metabolic fate of
chlordimeform [N-(4-chloro-o-tolyl) N'N'-dimethylformamidine] human
embryonic lung cell cultures. J Agric Food Chem, 23: 257-258.
Linder RE, Strader LF, Slott VL, & Suarez JD (1992) Endpoints of
spermatotoxicity in the rat after short duration exposures to fourteen
reproductive toxicants. Reprod Toxicol, 6: 491-505.
Ling W-Y, Zhang R-W, Gu Z-Y, Wang X-G, Zhang R-W, Jiang X-Z, Xue S-Z,
& Wang X-Z (1986) [Study on the urinary chlordimeform and its main
metabolite pCoT as index of biological monitoring in occupational
exposure.] Commun Labor Hyg Rail Syst, 8: 14-15 (in Chinese).
Lingren PD, Woffenbarger DA, Nosky JB, & Diaz M Jr (1972) Response of
Campoletis perdistinctus and Apanteles marginiventris to insecticides.
J Econ Entomol, 65: 1295-1300.
Liu Y-Z & Ke Z-H (1985) [Investigation on 53 cases of acute
chlordimeform poisoning.] Res Hyg, 14: 42-45 (in Chinese with English
abstract).
Liu X-L & Li W (1987) [Hemorrhage in upper digestive tract in
chlordimeform poisoning.] Occup Med, 14: 31 (in Chinese with English
abstract).
Liu Z & Mao Z (1980) [Studies on the analysis of metabolites of
pesticides. I. Gas chromatographic analysis of chlordimeform and its
metabolites in urine.] Hua Hsueh Pao, 38: 134-139 (in Chinese with
English Abstract).
Liu T-S & Zhang J-K (1985) [Poisoning due to chlordimeform, a new
pesticide.] Wuhan Med J, 9: 141-143 (in Chinese with English
abstract).
Liu H-G, Liu F-Q. Fang X-Z, & Wang X-M (1990) [Report on one case of
acute anemia due to chlordimeform poisoning.] PLA Med J, 15: 228
(in Chinese with English abstract).
Lu B-Q, Wang S-J, & Pong Y-F (1981) [Studies on the toxicology and
labor health of chlordimeform.] In: Chinese Institute of Scientific
Information and Bureau of Science and Technology ed. [Selected papers
on studies of pesticides.] Beijing, Ministry of Chemistry, pp 781-786
(in Chinese with English abstract).
Lund AE, Hollingworth RM, & Murdock LL (1979a) Formamidine pesticides:
A novel mechanism of action in lepidopterous larvae. Adv Pestic Sci,
3: 465-469.
Lund AE, Hollingworth RM, & Shamkland DL (1979b) Chlordimeform: Plant
protection by a sublethal, noncholinergic action on the central
nervous system. Pestic Biochem Physiol, 11: 117-128.
Lund AE, Hollingworth RM, & Yim GK (1979c) The comparative
neurotoxicity of formamidine pesticides. In: Neurotoxicology of
insecticidal pheromones, pp 119-137.
Lund AE, Shankland DL, Chinn D, & Yim GK (1978a) Similar
cardiovascular toxicity of the pesticide chlordimeform and lidocaine.
Toxicol Appl Pharmacol, 44: 357-365.
Lund AE, Yim GK, & Shankland DL (1978b) The cardiovascular toxicity of
chlordimeform: A local anesthetic-like action. In: Shankland DL,
Hollingworth RM, & Smyth T Jr ed. Pesticide and venom neurotoxicity.
New York, London, Plenum Press, pp 171-177.
Machin MV & Dingle JH (1977) Ultraviolet determination of
chlordimeform in cattle dipping baths and sprays. J Assoc Off Anal
Chem, 60: 696-698.
MacPhail RC & Leander JD (1980) Flavor aversions induced by
chlordimeform. Neurobehav Toxicol, 2: 363-365.
MacPhail RC & Leander JD (1981) Chlordimeform effects on
schedule-controlled behavior in rats. Neurobehav Toxicol Teratol,
3: 19-26.
Maddy KT, Knaak JB, & Gibbons DB (1986) Monitoring the urine of
pesticide applicators in California for residues of chlordimeform and
its metabolites, 1982-1985. Toxicol Lett, 33: 37-44.
Madhukar BV & Matsumura F (1979) Comparison of induction patterns of
rat hepatic microsomal mixed function oxidases by pesticides and
related chemicals. Pestic Biochem Physiol, 11: 301-308
Maitre L & Gfeller W (1975) Influence of compound 8514 (chlordimeform)
on the brain and liver monamineoxidase of rats. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Maitre L, Felner A, Waldmeier P, & Kehr W (1978) Monoamine oxidase
inhibition in brain and liver of rats treated with chlordimeform.
J Agric Food Chem, 26: 442-446.
Maqueda C, Perez Rodriguez JL, Martin F, & Hermosin MC (1983) A study
of the interaction between chlordimeform and humic acid from a typic
chromoxerert soil. Soil Sci, 136: 75-81.
Maqueda C, Morillo E, & Perez Rodriguez JL (1989) Interaction in
aqueous solution of certain pesticides with fulvic acids from a
spodosol soil. Soil Sci, 148: 336-345.
Matsumara F & Beeman RW (1976) Biochemical and physiological effects
of chlordimeform. Environ Health Perspect, 14: 71-82.
Mattern GC, Liu C-H, Louis JB, & Rosen JD (1991) GC/MS and LC/MS
determination of 20 pesticides for which dietary oncogenic risk has
been estimated. J Agric Food Chem, 39: 700-704.
Mayer FL & Ellersieck MR (1986) Manual of acute toxicity:
Interpretation and database for 410 chemicals and 66 species of
freshwater animals. Washington, DC, US Department of the Interior,
Fish and Wildlife Service, 505 pp (Resource Publication 160).
Ming Z-P (1977) [Report of four cases of chlordimeform poisoning,] New
Med, 8: 82-83 (in Chinese with English abstract).
Moore B (1971) Chlordimeform residues - A summary report. Pendle Hill,
Ciba-Geigy Australia Ltd (Unpublished report No. 71/11/358).
Moser VC, McCormick JP, Creason JP, & MacPhail RC (1988) Comparison of
chlordimeform and carbaryl using a functional observational battery.
Fundam Appl Toxicol, 11: 189-206.
Muecke W, Hamboeck H, & Arni P (1979) Metabolic and pharmacokinetic
aspects of chlordimeform in rats and mice. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 37/79).
Murakami M & Fukami J (1974) Effect of chlorphenamide and its
metabolites an HeLa cells. Bull Environ Contam Toxicol, 11: 184-188.
Nathanson JA & Hunnicutt EJ (1981) N-demethylchlordimeform: A potent
agonist of octopamine-sensitive adenylate cyclase. Mol Pharmacol,
20: 68-75.
NCI (1979) Bioassay of 4-chloro-o-toluidine hydrochloride for possible
carcinogenicity. Bethesda, Maryland, National Cancer Institute
(Technical Report Series No. 165).
Neumann R & Voss G (1977) MAO inhibition, an unlikely mode of action
for chlordimeform. Experientia (Basel), 33: 23-24.
Nixon WB & Neal BE (1983) Chlordimeform residues in human urine
following dermal application. Greensboro, North Carolina, Ciba-Geigy
Corporation (Unpublished report No. ABR-83017).
Nui J-H, Ma H, Muo J-T, & Wu W-L (1990) [Report on 4 cases of
poisoning due to chlordimeform and organophosphate pesticides.]
Shang-Dong Ind Hyg, 2: 35 (in Chinese with English abstract).
O'Brien PJ, Elzen GW, & Vinson SB (1985) Toxicity of azinphosmethyl
and chlordimeform to parasitoid braconmellitor (Hymenoptera:
Braxonidae): lethal and reproductive effects. Environ Entomol,
14: 891-894.
Olson KL, Boush GM, & Matsumura F (1978) Behavioral effects of
perinatal exposure of chlordimeform in rats. Bull Environ Contam
Toxicol, 20: 760-768.
Ott MG & Langner RR (1983) A mortality survey of men engaged in the
manufacture of organic dyes. J Occup Med, 25: 763-770.
Palmer WA, Dingle JHP, & Heath AB (1977) Residues of chlordimeform in
bovine tissues and milk following application by a handspray. Aust J
Exp Agric Anim Husb, 17: 380-384.
Parkes HG (1984) Epidemiology of aromatic amine cancers. In: Searle CE
ed. Chemical carcinogens. Washington, DC, American Cancer Society,
pp 277-301 (ACS Monograph No. 182).
Perez-Rodriguez JL & Hermosin MC (1979) Adsorption of chlordimeform by
montmorillonite. In: Mortland MM & Farmer VC ed. Proceedings of the
International Clay Conference, Oxford, 1978. Amsterdam, Oxford, New
York, Elsevier Science Publishers, pp 227-234.
Pfister WR, Chin C, Noland V, & Yim GK (1978a) Similar pharmacological
actions of chlordimeform and local anesthetics. Pestic Biochem
Physiol, 9: 148-156.
Pfister WR, Hollingworth RM, & Yim GW (1978b) Increased feeding in
rats treated with chlordimeform and related formamidines: A new class
of appetite stimulants. Psychopharmacology (Berlin), 60: 47-51.
Phillips JR, Herzog GA, & Nicholson WF (1977) Effect of chlordimeform
on fruiting characteristics and yield of cotton. Ark Farm Res, 26: 4.
Pimley RW (1986) Toxicity of chlordimeform to the tsetse fly Glossina
morsitans. Insect Sci Appl, 7: 663-666.
Popp W & Norpoth K (1991) [Carcinogenic effects of chlordimeform and
4-chloro-o-toluidine.] Dtsch Med Wochenschr, 116: 1454 (in German).
Popp W, Schmieding W, Speck M, Vahrenholz C, & Norpoth K (1992)
Incidence of bladder cancer in a cohort of workers exposed to
4-chloro-o-toluidine while synthesising chlordimeform. Br J Ind Med,
49: 529-531.
Puri E & M�ller D (1983a) C8513 technical: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830304).
Puri E & M�ller D (1983b) CGA 72647 pure: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830311).
Puri E & M�ller D (1983c) C8513 technical: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830306).
Puri E & M�ller D (1983d) CGA 72647 pure: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Lid
(Unpublished report No. 830313).
Rashid KA, Ercegovich CD, & Mumma RO (1984) Evaluation of
chlordimeform and degradation products for mutagenic and DNA-damaging
activity in Salmonella typhimurium and Escherichia coli. J Environ Sci
Health, B 19: 95-110.
Rieger JA, Robinson CP, Cox P, & Horst MA (1981) Cardiovascular
actions and interactions of chlordimeform in the dog. Bull Environ
Contam Toxicol, 27: 707-715.
Rieger JA, Allen LV Jr, & Fisher RC (1985) Extraction and analysis of
chlordimeform and demethylchlordimeform from human issue samples. Bull
Environ Contam Toxicol, 35: 397-405.
Robinson CP & Smith PW (1977) Lack of involvement of monoamine oxidase
inhibition in the lethality of acute poisoning by chlordimeform.
J Toxicol Environ Health, 3: 565-568.
Robinson CP, Smith PW, Zelenski JD, & Endecott BR (1975) Lack of an
effect of interference with amine mechanisms on the lethality of
chlordimeform in the rat. Toxicol Appl Pharmacol, 33: 380-383.
Rose JA (1969a) Degradation of chlorphenamidine by rat liver and
spleen homogenates with particular reference to the possible formation
of azo derivatives. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Rose JA (1969b) Formation of a symmetrical azo-derivative from
4-chloro-o-toluidine by coupled peroxidatic reactions. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Ross MSF & Leisten O (1989) Analysis of trace amounts of chlordimeform
in urine samples. Am Clin Lab, 8: 22-24.
Sachsse K, Suter P, Zak F, & Hess R (1978a) Lifespan feeding study in
mice with N-formyl-4-chloro-o-toluidine - Final report (Project No.
Siss M 4762/1. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Zak F, & Hess R (1978b) Lifespan feeding study in
mice with 4-chloro-o-toluidine-HCI - Final report (Project No. Siss M
04760/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979a)
Chlordimeform-HCL: 60-day feeding study in mice - Final report. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979b)
Chlordimeform-HC1: 60-day feeding study in rat - Final report (Project
No. Siss 6138). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980a)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in the rat - Final
report (Project No. 404078). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980b)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in mice - Final
report (Project No. 404178). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980c) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
chlordimeform-HC1 - Final report (Project No. Siss. R 05658/1). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980d) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
N-formyl-4-chloro-o-toluidine - Final report (project No. Siss
5688/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980e) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
4-chloro-o-toluidine- HC1 - Final report (project No. Siss R 05687/1).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report),
Salvisberg W, Neumann R, & Voss G (1980) Chlordimeform: Mode of toxic
action in various development stages of Spodoptera littoralis. J Econ
Entomol, 73: 193-196.
Sen Gupta AK & Knowles CO (1969) Metabolism of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine by apple seedlings.
J Agric Food Chem, 17: 595-600.
Sen Gupta AK & Knowles CO (1970) Galecron 14C(N'-(4-chloro-o-tolyl)
-N,N-dimethylformamidine) metabolism in the dog and goat. J Entomol,
63: 951-956.
Shimizu T & Fukami J (1983) Effect of chlordimeform and its analogues
on wandering behavior of the cabbage armyworm, Mamestra brassicae L.
(Lepidoptera:Noctuidae). Appl Entomol Zool, 18: 554-557.
Shopp GM Jr, McCay JA, & Holsapple MP (1985) Suppression of the
antibody response by a formamidine pesticide: dependence on the route
of exposure. J Toxicol Environ Health, 15: 293-304.
Sparks TC, Leonard BR. Schneider F. & Graves JB (1993) Ovicidal
activity and alteration of octopamine titers: Effects of selected
insecticides on eggs of the tobacco budworm (Lepidoptera: Noctuidae).
J Econ Entomol, 86: 294-300.
Stasik MJ (1988) Carcinomas of the urinary bladder in a 4-chloro-o-
toluidine cohort. Int Ach Occup Environ Health, 60: 21-24.
Stasik MJ (1991) [Bladder cancer from 4-chloro-o-toluidine.] Dtsch Med
Wochenschr, 116: 1444-1447 (in German).
Stoker TE, Goldman JM, Cooper RL, & McElroy WK (1991) Influence of
chlordimeform on alpha-adrenergic receptor-associated mechanisms of
hormonal regulation in the rat: pituitary and adrenocortical
secretion. Toxicology, 69: 257-268.
Strasser FF & M�ller D (1983a) CGA 72651 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782323).
Strasser FF & M�ller D (1983b) CGA 72647 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Lid (Unpublished report No. 782326).
Strasser FF & M�ller D (1984a) CGA 72647 technical: Point mutation
assay with mouse lymphoma cells, host-mediated assay. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 782325).
Strasser FF & M�ller D (1984b) CGA 72651: Point mutation assay with
mouse lymphoma cells. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782322)
Streibert HP & Dittrich V (1977) Toxicological response of insect eggs
and larvae to a saturated atmosphere of chlordimeform. J Econ Entomol,
70: 57-59.
Strong MB & Bull MS (1985a) Levels of chlordimeform equivalents found
in the urine of personnel involved in the ground rig application of
CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Ltd (Unpublished report
No. 85/6/1030).
Strong MB & Bull MS (1985b) Levels of chlordimeform equivalents found
in the urine of personnel involved in the aerial application of CGS
500 and CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Lid (Unpublished report
No. 85/6/1029).
Su S-T (1977) [Report of 6 cases of chlordimeform poisoning]. Intermed
Med, 6: 19 (in Chinese with English abstract).
Su GC & Zabik MJ (1972) Photochemistry of bioactive compounds:
Photolysis of arylamidine derivatives in water. J Agric Food Chem,
20: 320-323.
Suter P, Luetkemeier H, Sachsse K. Zak F, & Hess R (1976a) 60-Day
feeding study in mice with 4-chloro-o-toluidine HC1 (Project No. Siss
4760). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Suter P, Luetkemeier H, Sachsse K, Zak F, & Hess R (1976b) 60-Day
feeding study in the rat with 4-chloro-o-toluidine HC1 (Project No.
Siss 4760). Basel, Switzerland. Ciba-Geigy Lid (Unpublished report).
Suter P, Zak F, Sachsse K, & Hess R (1978) lifespan feeding study in
mice with chlordimeform-HC1 - Final report (Project No. Siss. M
04761/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Tao X-M, Sun R-M, Wang M-S, Xue S-Z, Zhang R-W, Jiang X-Z & Wang Y-L
(1985) [investigation on the hygienic status of chlordimeform
production in Cai-Qiao pesticide factory.] Chin J Ind Hyg Occup Dis,
3: 272-274 (in Chinese).
Thomas WD, Craig GK, & Stacey NH (1990) Effects of chlordimeform and
its metabolite 4-chloro-o-toluidine on rat splenic T, B and
tumoricidal effector cells. Immunopharmacology, 19: 79-86.
Uk S & Dittrich V (1986) The behavior-modifying effect of
chlordimeform and endosulfan on the adult whitefly Bemisia tabaci
(Genn.) which attacks cotton in the Sudan. Crop Prot, 5: 341-347.
Voss G (1971) Galecron residues - German hops (green cones) and beer
1970. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
42/71).
Voss G & Burkhard N (1971) Galecron residues in milk, meat, fat, liver
and kidney of a Swiss cows. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. RVA 24/7/S).
Voss G, Kossmann K, & Geissb�hler H (1973) Chlordimeform. In: Sherma J
& Zweig G ed. Analytical methods for pesticide plant growth
regulators. New York, London, Academic Press Inc., vol 7, pp 211-230.
Wang X-Z & Tong Q (1992) [Observation on 71 EKG of acute chlordimeform
poisoning cases.] Labor Prot Chem Ind, 13: 61-62 (in Chinese with
English abstract)
Wang CM, Narahashi T, & Fukami J (1975) Mechanism of neuromuscular
block by chlordimeform. Pestic Biochem Physiol, 5: 119-125.
Wang M, Zhou Z-J, Li H-L, Zhang R-W, Xue S-Z, Hong Z-R, Gu Q, Wang
X-M, & Feng Y-Q (1987) [Investigation on the hygienic status of
farmers spraying chlordimeform.] Chin J Ind Hyg Occup Dis, 5: 50-53
(in Chinese)
Wang Y-S & Dong C-R (1985) [Experience on treatment of 25 cases of
chlordimeform poisoning.] Sichuan Med, 6: 207 (in Chinese with English
abstract).
Watanabe H, Tsuda S, & Fukami J (1975) Effect of chlordimeform on
rectus abdominum muscle in frog. Pestic Biochem Physiol, 5:150-154.
Watanabe Y & Matsumura F (1987) Comparative metabolism of sulfamidine
and chlordimeform in rats. J Agric Food Chem, 35: 379-384
Watkinson WP (1985) Effects of chlordimeform on cardiovascular
functional parameters: Part 1. Lethality and arrhythmogenicity in the
geriatric rat. J Toxicol Environ Health, 15: 729-744.
Watkinson WP (1986a) Effects of chlordimeform on cardiovascular
functional parameters: Part 2. Acute and delayed effects following
intravenous administration in the postweanling rat. J Toxicol Environ
Health, 19: 195-206.
Watkinson WP (1986b), Effects of chlordimeform on cardiovascular
functional parameters: Part 3. Comparison of different routes of
administration in the postweanling rat. J Toxicol Environ Health,
19: 207-217.
Watkinson WP & Gordon CJ (1987) Effects of chlordimeform on heart rat
and body temperature of unanesthetized, unrestrained rats. Toxicol
Lett, 35: 209-216.
Watkinson WP, Highfill JW, & Gordon CJ (1989) Modulating effect of
body temperature on the toxic response produced by the pesticide
chlordimeform in rats. J Toxicol Environ Health, 28: 483-500.
Weisburger EK, Russfield AB, Homburger F, Weisburger JH. Boger E, Van
Donger CG, & Chu KC (1978) Testing of twenty-one environmental
aromatic amines for long-term toxicity or carcinogenicity. J Environ
Pathol Toxicol, 2: 325-356.
Wilson AG (1981) Field evaluation of formamidine insecticides and
Bacillus thuringiensis for selective control of Heliothis spp. on
cotton. Gen Appl Entomol, 13: 105-111.
Wiltrout RW, Ercegovich CD, & Ceglowski WS (1978) Humoral immunity in
mice following oral administration of selected pesticides. Bull
Environ Contam Toxicol, 20: 423-431.
Witkin JM & Leander JD (1982) Effects of the appetite stimulant
chlordimeform on food and water consumption of rats: Comparison with
chlordiazepoxide. J Pharmacol Exp Ther, 223: 130-134.
Witkonton S (1973) Fate of the pesticide N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine in fruit and soil, and toxicological properties
of some of its conversion products. Diss Abstr Int, 34: 3831.
Witkonton S & Ercegovich CD (1972) Degradation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine in six different
fruits. J Agric Food Chem, 20: 569-573.
Wolfenbarger DA (1974) Activity of chlordimeform HCI and chlordimeform
against arthropods attacking cotton. J Econ Etomol, 67: 445-446.
Wolfenbarger DA, Gomes ED, Robinson SH, & Parks LH (1979)
Chlordimeform residues from cotton leaves. J Econ Entomol,
72: 128-130.
Worthing CR (1979) The pesticide manual: A work compendium, 6th ed.
Croydon, United Kingdom, British Crop Protection Council, p 95.
Worthing CR & Walker SB (1983) The Pesticide manual. 7th ed. Croydon,
United Kingdom, British Crop Protection Council, p 2270.
Wu G (1982) [One case of chlordimeform poisoning.] Chin J Prev Med,
17: 275 (in Chinese with English abstract).
Wu S-Y, Liu Y-Y, Huang W-Z, Hong Q-F, Ling R-F, & Wang J-Y (1983) [On
chlordimeform poisoning - 24 case reports.] J Fujian Med Coll, 17:
42-44 (in Chinese with English abstract).
Wu D, Costa LG, & Murphy SD (1990) [The effect of pesticides-
chlordimeform and amitraz on inhibition of the binding of
3H-clonidine to alpha2-adrenoreceptor in rat forebrain tissue.]
Huaxi Yike Daxue Xuebao, 21: 17-21 (in Chinese with English abstract).
Xia S-C & Gao X-Z (1977) [Rescue after chlordimeform poisoning.]
Intermed Med, 6: 18-19 (in Chinese with English abstract).
Xie P-F (1983) [Analysis on the clinical features of chlordimeform
poisoning.] J Pract Med, 3: 145-146 (in Chinese with English
abstract).
Xu S-H (1987) [Experiences on treatment of chlordimeform poisoning.]
Hu-Nan Med, 4: 168-167 (in Chinese with English abstract).
Xue S-Z & Loosli R (1994) Formamidines. Toxicology, 91: 99-104.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J. & Wang YL (1990) The risk
assessment of occupational exposure to chlordimeform. Presented at the
23rd International Congress on Occupational Health, Montreal, Canada,
September 1990.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J, & Wang YL (1991)
[Assessment on the carcinogenic risk of chlordimeform.] Environ Chem,
10: 54-60 (in Chinese).
Yang Q-Z (1984) [Rescue of a case of cardiac arrest due to
chlordimeform poisoning.] Jiangsu Med, 10: 38 (in Chinese with English
abstract).
Yim GK, Holsapple MP, Pfister WR, & Hollingworth RM (1978)
Prostaglandin synthesis inhibited by formamidine pesticides. Life Sci.
23: 2509-2516.
Youngman RR, Leigh TF, Kerby TA, Toscano NC, & Jackson CE (1990)
Pesticides and cotton: Effect on photosynthesis, growth, and fruiting.
J Econ Entomol, 83: 1549-1557.
Zak F, Sachsse K, Bath R, & Hess R (1973) One-year feeding study on
the rat with technical C-8514 (Project No. Siss 553). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Zhang Y-J, Yuan B-Q, Jin G-L, & Liu Y-Z (1976) [Report on two severe
cases of chlordimeform poisoning and their treatment.] J Rural Doctor,
5: 19 (in Chinese with English abstract).
Zhang R-W, Lin W-Y, Wang X-G, Gu Z-Y, Qu J-Y, Xue S-Z & Wang Y-L
(1986a) [Health effects of chlordimeform on the applicators.] Chin J
Prev Med, 20: 186 (in Chinese with English abstract).
Zhang R-W, Xue S-Z, & Wang Y-L (1986b) [Measurement of total amount of
body surface contamination with regional proportional sampling.] Occup
Med, 13: 2-3 (in Chinese with English abstract).
Zhou D-J (1987) [Bitter lesson on misuse of large dose of atropine on
chlordimeform poisoning.] Hu-Nan Med, 4: 169 (in Chinese with English
abstract).
Zimmer D, Mazurek J. Petzold G. & Bhuyan BK (1980). Bacterial
mutagenicity and mammalian cell DNA damage by several substituted
anilines. Mutat Res. 77: 317-326.
R�SUM�
1. Identit�, propri�t�s physiques et chimiques et m�thodes d'analyse
Le chlordim�form est une base de force moyenne qui forme des sels
stables avec les acides forts. A l'�tat pur, le chlordim�form et son
chlorhydrate sont des solides cristallins incolores. Le point de
fusion du chlordim�form base est de 32�C, celui du chlorhydrate �tant
de 225-227�C. La base est l�g�rement soluble dans l'eau (250 mg/litre)
et facilement soluble dans les solvants organiques, tandis que le
chlorhydrate est facilement soluble dans l'eau mais plus difficilement
dans les solvants organiques. La tension de vapeur du chlordim�form
est de 48 mPa � 20�C et son coefficient de partage entre l'eau et
l'octanol (log Kow) est �gal � 2,89. On peut faire appel � de
nombreuses m�thodes d'analyse pour la recherche et le dosage du
chlordim�form dans les v�g�taux, le sol, l'eau et l'urine.
2. Sources d'exposition humaine et environnementale
Le chlordim�form n'existe pas � l'�tat naturel. On le pr�pare
industriellement par condensation du r�actif de Vilsmeier (obtenu par
r�action du dim�thylformamide sur POCl3, SOCl2 ou COCl2) soit avec
la 4-chloro- o-toluidine, soit avec la l' o-toluidine, suivie d'une
chloration du d�riv� interm�diaire obtenu. On l'utilise comme
acaricide � large spectre et il est principalement actif contre les
formes mobiles des acariens et des tiques ainsi que contre les oeufs
et les premiers stades de certains l�pidopt�res. Il agit en phase
gazeuse aussi bien que par contact. Les premiers temps de son
utilisation, on l'employait pour traiter des cultures tr�s vari�es
comme les fruits � p�pins, les drupes, les choux et autres l�gumes,
les raisins, le houblon, les agrumes, les cerises et les fraises. On
l'utilise aussi en bains contre les tiques des bovins. Ces derni�res
ann�es son usage s'est g�n�ralement limit� au coton, mais on continue
tout de m�me � l'utiliser sur le riz dans certains pays. Depuis
1988/89 il n'est plus homologu� dans la plupart des pays. En Chine, la
production a cess� en 1992, de m�me que la vente en 1993.
3. Transport, distribution et transformation dans l'environnement
Bien que sa tension de vapeur ait une valeur moyenne, le
chlordim�form ne s'�vapore pas autant qu'on le penserait des surfaces
v�g�tales. Sa stabilit� vis-�-vis de l'hydrolyse d�pend fortement du
pH; il est stable en milieu acide mais s'hydrolyse rapidement en
milieu alcalin. Le chlordim�form est capable de s'adsorber sur les
mati�res organiques dissoutes.
Dans le sol, la disparition du chlordim�form est essentiellement
imputable � l'action des microorganismes et, pour une moindre part, �
l'hydrolyse chimique. Malgr� la solubilit� du compos� dans l'eau, on
ne trouve gu�re de traces de lessivage, ce qui peut s'expliquer par
une adsorption aux mat�riaux argileux ou aux mati�res organiques du
sol ainsi que par la biod�gradation. Les principaux m�tabolites sont
la N-formyl-4-chloro- o-toluidine et la 4-chloro- o-toluidine.
Les plantes fixent le chlordim�form pr�sent dans le sol en
proportion faible mais mesurable et la concentration est suffisante
pour affecter les ravageurs qui se nourrissent � leurs d�pens. En
traitement foliaire, la p�n�tration du chlordim�form dans la cuticule
est limit�e. Le chlordim�form est rapidement d�compos� par les
v�g�taux. Les principaux m�tabolites sont le d�m�thylchlordim�form, la
N-formyl-4-chloro -o-toluidine et la 4-chloro -o-toluidine, cette
derni�re n'�tant pas produite par toutes les plantes.
Dans le sol, le chlordim�form et ses m�tabolites disparaissent
selon une cin�tique du premier ordre avec une demi-vie de 20 � 40
jours.
Les �tudes de bioaccumulation montrent que les organismes
aquatiques ne fixent qu'une petite quantit� de chlordim�form et que
celui-ci s'�limine rapidement une fois ces organismes replac�s en eau
pure.
4. Concentrations dans l'environnement et exposition humaine
On n'a pas proc�d� � des mesures de concentration dans l'air ou
l'eau. Apr�s traitement de rizi�res, on a retrouv� des r�sidus allant
jusqu'� 2900 �g/kg dans les 5 premiers centim�tres du sol et jusqu'�
150 �g/kg dans les 5 centim�tres suivants.
On a fix� des teneurs limites en r�sidus pour un grand nombre de
produits crus et dans certains cas, pour des pr�parations contenant
ces produits. Les limites maximales de r�sidus fix�es par le Codex ont
�t� supprim�es.
Il y a eu des cas d'exposition au chlordim�form au cours de la
pr�paration, de la formulation et de l'�pandage de ce produit. Depuis
quelques ann�es, on utilise la concentration urinaire totale du
chlordim�form et de ses m�tabolites pour surveiller l'exposition et il
y a d'ailleurs une bonne corr�lation entre cette concentration et le
degr� de contamination cutan�e. Dans les industries cotonni�res o�
l'on a soumis les ouvriers agricoles � une surveillance g�n�rale de la
concentration urinaire en chlordim�form, on a constat� que les plus
expos�s �taient les chargeurs, les laveurs et les m�caniciens et les
moins expos�s les signaleurs et les pilotes.
5. Cin�tique et m�tabolisme chez les animaux de laboratoire et
l'Homme
Chez les mammif�res, le chlordim�form est facilement r�sorb� au
niveau des voies digestives ainsi que par la voie transcutan�e. Il est
ensuite rapidement excr�t� � raison de 80% environ dans l'urine et de
10-15% dans les mati�res f�cales. De petites quantit�s de r�sidus sont
pr�sentes au bout de 10 jours dans tous les tissus mais rien n'indique
qu'il y ait bioaccumulation. Apr�s application cutan�e chez l'Homme,
on constate �galement une excr�tion urinaire rapide.
On retrouve dans l'urine plusieurs m�tabolites du chlordim�form
sous forme oxyd�e et conjugu�e, � savoir principalement la
N-formyl-4-chloro- o-toluidine, et la 4-chloro- o-toluidine.
In vitro, on retrouve les m�mes m�tabolites, mais avec pr�dominance
de la 4-chloro- o-toluidine.
6. Effets sur les mammif�res de laboratoire et les syst�mes d'�preuve
in vitro
Les �preuves pratiqu�es sur un certain nombre d'esp�ces montrent
que la chlordim�form pr�sente une toxicit� aigu� mod�r�e par la voie
orale et la voie transcutan�e. Chez le rat, les principaux m�tabolites
sont peu toxiques par voie orale. Chez le lapin, le chlordim�form ne
provoque qu'une l�g�re irritation oculaire et cutan�e. Apr�s
exposition de courte ou de br�ve dur�e au chlordim�form ou � ses
m�tabolites, on peut observer, au niveau des constantes
h�matologiques, des modifications qui sont imputables au traitement et
on constate, � dose �lev�e, certains signes qui d�notent une
hyperplasie de l'�pith�lium des canaux biliaires et de la vessie. Il
n'y pas d'accroissement de la fr�quence des tumeurs chez le rat. Chez
la souris, on observe, apr�s administration par voie alimentaire de
chlordim�form, de N-formyl-4-chloro -o-toluidine ou de
4-chloro- o-toluidine, une augmentation, li�e � la dose, des tumeurs
malignes h�morragiques d'origine vasculaire appartenant � la classe
des h�mangio-endoth�liomes, dont la pr�sence entra�ne un accroissement
de la mortalit� parall�le � la dose.
Le chlordim�form n'a pas d'effet ind�sirable sur les diff�rents
aspects de la fonction de reproduction et il n'a aucun pouvoir
t�ratog�ne.
Le chlordim�form a fait l'objet d'un grand nombre d'�preuves de
g�notoxicit� in vitro et in vivo. Aucune d'elles n'a donn� de
r�sultat positif, �tant entendu qu'il s'agissait de la mati�re active
et non de formulations. Par ailleurs, un certain nombre d'observations
sporadiques non confirm�es font �tat d'une activit� mutag�ne induite
par la N-formyl-4-chloro- o-toluidine et par la 4-chloro- o-
toluidine. Il n'existe qu'une seule description de transformations
cellulaires provoqu�es par le chlordim�form et par la 4-chloro -o-
toluidine. Chez des souris et des rats trait�s par le chlordim�form,
on a constat� une que le compos� se liait � l'ADN des cellules
h�patiques. A dose beaucoup plus �lev�e, il se forme chez les m�mes
animaux un important adduit hydrophobe.
Le chlordim�form provoque des effets pharmacologiques et
biochimiques divers chez l'animal, et notamment des effets
cardiovasculaires, une hypothermie, une hyperexcitabilit�, une
modification des fonctions visuelle et auditive ainsi que la
modulation des amines biogen�se et des enzymes pharmacom�tabolisantes.
7. Effets sur l'Homme
Les intoxications aigu�s se traduisent par une fatigue, des
naus�es et une perte d'app�tit, avec, dans les cas graves, somnolence,
cyanose, besoin imp�rieux d'uriner, cystite, effets cardiovasculaires
(tachy-cardie, bradycardie, anomalies de l'ECG), coma et �tat de choc.
En g�n�ral, la r�cup�ration est totale.
Apr�s une exposition de longue dur�e au chlordim�form, on peut
observer encore d'autres sympt�mes tels que des douleurs abdominales,
des d�mangeaisons et des �ruptions (en cas d'exposition cutan�e)
accompagn�s d'une h�maturie macroscopique ou micro-scopique. On a
signal� de nombreux cas d'intoxication pr�sentant des sympt�mes
d'exposition de longue dur�e parmi les ouvriers d'unit�s de production
de chlordim�form et des ouvriers agricoles.
Les donn�es �pid�miologiques obtenues � la suite de cas
d'exposition professionnelle montrent qu'il existe une forte
corr�lation entre l'exposition � la 4-chloro -o-toluidine et le
cancer de la vessie. En revanche, on n'a gu�re obtenu d'�l�ments qui
militeraient en faveur d'une association entre ce type de cancer et
l'exposition au chlordim�form.
8. Effets sur les autres �tres vivants au laboratoire et dans leur
milieu naturel
Apr�s �pandage de chlordim�form sur le sol, on n'a pas observ�
d'effets sensibles sur les populations de champignons, de bact�ries ou
d'actinomyc�tes terricoles.
Il n'existe pas de donn�es toxicologiques de laboratoire
concernant les invert�br�s dul�aquicoles. En pr�sence de
chlordim�form, il y a inhibition de la croissance des larves
d'hu�tres, avec une CE50 de 5,7 mg/litre. Pour la crevette rose,
le seul crustac� �tudi�, la CL50 � 96 h a �t� trouv�e �gale �
7,1 mg/litre et des valeurs allant de 1 � 54 mg/litre ont �t� obtenues
pour le m�me param�tre chez les poissons. On ne poss�de aucune donn�e
relative � la toxicit� chronique pour les esp�ces aquatiques.
L'ensemble des r�sultats de laboratoire et des donn�es recueillies sur
le terrain indique que le compos� est toxique pour de nombreux
arthropodes terrestres non vis�s.
Chez l'abeille, la toxicit� de contact se traduit par une DL50
de 120 �g/g, la toxicit� par voie orale correspondant � une valeur de
187 �g/g. Trois heures apr�s l'�pandage de chlordim�form sur de la
luzerne, l'exposition de certaines esp�ces d'abeilles aux r�sidus
encore pr�sents sur les plantes, n'a provoqu� aucune mortalit�.
La CL50 par voie alimentaire varie de >1000 � > 5000/kg de
nourriture pour diverses esp�ces d'oiseaux.
9. Evaluation des risques pour la sant� humaine et des effets sur
l'environnement
On a observ� des signes d'intoxication aigu� chez des
travailleurs qui, peut-�tre par suite de l'inobservation des mesures
de s�curit�, avaient �t� fortement expos�s � du chlordim�form au cours
de la pr�paration ou de l'utilisation de ce produit. Comme, � ce qu'il
semble, il n'est plus produit ni utilis� nulle part dans le monde, il
ne devrait plus y avoir de cas d'intoxication aigu�. Le risque li� �
une exposition chronique et en particulier, le risque de cancer de la
vessie, subsistera cependant pendant de nombreuses ann�es. Il faut
continuer � effectuer des contr�les sanitaires chez les sujets qui ont
subi une exposition notable pour avoir travaill� dans des ateliers de
production de chlordim�form ou avoir v�cu dans des zones rurales o� le
produit �tait largement utilis�.
Comme il s'agit d'un produit qui n'est plus en usage, on n'a pas
proc�d� � une �valuation quantitative du risque qu'il repr�sente pour
l'environnement. On ne pense pas que celui-ci puisse subir des effets
nocifs � long terme qui soient attribuables � l'utilisation ant�rieure
du produit.
10. Conclusions et recommandations
Il existe un risque non n�gligeable que le chlordim�form produise
des effets toxiques imm�diats ou � plus long terme chez les individus
expos�s. Les donn�es disponibles accr�ditent l'id�e d'une association
entre l'augmentation de l'incidence du cancer de la vessie chez
l'homme et l'exposition � la 4-chloro- o-toluidine et, dans une
moindre mesure, au chlordim�form.
Le chlordim�form ne persiste pas dans l'environnement et il ne
devrait donc pas y avoir d'effets nocifs � long terme sur celui-ci qui
r�sulteraient de l'usage ant�rieur du compos�.
Il n'est pas recommand� de reprendre la production ou l'usage du
chlordim�form dans un but commercial. Les stocks existants doivent
�tre �limin�s selon les r�gles de s�curit�.
Les personnes expos�es au chlordim�form de par leur profession
doivent �tre soumises � des examens cytologiques v�sicaux et � une
recherche syst�matique de l'h�maturie dans le cadre d'un programme
g�n�ral de d�pistage.
RESUMEN
1. Identidad, propiedades f�sicas y qu�micas y m�todos anal�ticos
El clordimeformo es una base de fuerza media que forma sales
estables con �cidos fuertes. Tanto el clordimeformo como su sal
hidroclorada en estado puro son s�lidos cristalinos incoloros. El
punto de fusi�n del clordimeformo (base) es de 32�C, mientras que el
de la sal hidroclorada es de 225-227�C. El clordimeformo (base) es
poco soluble en agua (250 mg/litro) y f�cilmente soluble en
disolventes org�nicos, mientras que la sal hidroclorada es f�cilmente
soluble en agua pero menos soluble en disolventes org�nicos. El
clordimeformo (base) tiene una presi�n de vapor de 48 mPa a 20�C y un
log Kow de 2,89. Se dispone de una amplia gama de m�todos anal�ticos
para detectar y cuantificar la presencia de clordimeformo en las
plantas, el suelo, el agua y la orina.
2. Fuentes de exposici�n humana y ambiental
El clordimeformo no existe en la naturaleza. Se produce
comercialmente mediante condensaci�n del reactivo de Vilsmeier
(obtenido por reacci�n de la dimetilformamida con POCl3, SOCl2
o COCl2) con 4-cloro- o-toluidina o bien con o-toluidina y
cloraci�n ulterior del producto intermedio resultante. Se ha utilizado
como acaricida de amplio espectro y act�a principalmente contra las
formas m�viles de �caros y garrapatas, as� como contra los huevos y
las cris�lidas en estado inicial de algunos insectos del orden
Lepid�ptera. Es activo en la fase de vapor, as� como por contacto.
Cuando comenz� a utilizarse, se aplicaba a productos de una amplia
variedad de cultivos, tales como frutas de pipas, frutas de hueso,
berzas, hortalizas, uvas, l�pulo, c�tricos, manzanas, peras, cerezas y
fresas. Tambi�n se utilizaba en ba�os antiparasitarios para combatir
las garrapatas del ganado. En los �ltimos a�os, su uso se limitaba por
lo general al algod�n, aunque en algunos pa�ses se segu�a aplicando al
arroz. En la mayor�a de los pa�ses, su registro se abandon�
voluntariamente en 1988/1989. En China dej� de producirse en 1992 y de
venderse en 1993.
3. Transporte, distribuci�n y transformaci�n en el medio ambiente
El clordimeformo tiene una presi�n de vapor moderada pero su
evaporaci�n de la superficie de las plantas es inferior a la que
cabr�a prever. La estabilidad hidrol�tica del clordimeformo depende
mucho del pH; es estable en condiciones �cidas pero se hidroliza
r�pidamente en condiciones alcalinas. El clordimeformo tiene un
potencial de adsorci�n a la materia org�nica disuelta.
Hay dispersi�n del clordimeformo en el suelo, principalmente por
acci�n microbiana y, en menor medida, por hidr�lisis qu�mica. Pese a
la solubilidad del clordimeformo en agua, hay pocos indicios de
lixiviaci�n, lo que puede deberse a su adsorci�n a minerales
arcillosos y a la materia org�nica del suelo, as� como a su
biodegradaci�n. Los principales metabolitos son la N-formil-
4-cloro- o-toluidina y la 4-cloro- o-toluidina.
La absorci�n del clordimeformo por las plantas a partir del suelo
es escasa pero detectable, y suficiente para afectar a las plagas que
se alimentan de ellas. El clordimeformo aplicado a las hojas s�lo
tiene una capacidad limitada de penetrar en las capas cuticulares. El
clordimeformo se degrada r�pidamente en las plantas. Sus principales
metabolitos son el demetilclordimeformo, la N-formil-4-cloro-
o-toluidina y la 4-cloro- o-toluidina, aunque no todas las plantas
estudiadas produjeron 4-cloro- o-toluidina.
El clordimeformo y sus metabolitos se dispersan en el suelo
conforme a una cin�tica de primer orden, con una semivida de 20-40
d�as.
Los estudios sobre bioacumulaci�n indican una escasa absorci�n
del clordimeformo por los organismos acu�ticos y una r�pida depuraci�n
de �stos despu�s de haber sido transferidos a un agua limpia.
4. Niveles medioambientales y exposici�n humana
No se han medido los niveles de clordimeformo en el aire ni en el
agua. Tras la aplicaci�n de clordimeformo a unos arrozales, en el
suelo se hallaron residuos en concentraciones de hasta 2900 �g/kg en
los 5 cm primeros de profundidad, y de 150 �g/kg en los 5 cm
siguientes.
Se establecieron niveles m�ximos de residuos aplicables a una
amplia variedad de productos sin elaborar y, en algunos casos, de
residuos trasladados a los alimentos elaborados. Los l�mites m�ximos
aplicables a los residuos de clordimeformo se han retirado del Codex
Alimentarius.
Hab�a exposici�n ocupacional al clordimeformo durante la
fabricaci�n, la formulaci�n y la aplicaci�n del producto. En los
�ltimos a�os la exposici�n se ha vigilado mediante la determinaci�n de
los niveles totales de clordimeformo y de sus metabolitos presentes en
la orina, y hay una correlaci�n positiva entre el nivel en la orina y
el grado de contaminaci�n cut�nea. Entre los trabajadores agr�colas de
los algodonales sometidos a una amplia vigilancia de la excreci�n
urinaria de clordimeformo, los niveles m�s altos de exposici�n se
hallaban en los cargadores, lavadores y mec�nicos, y los niveles m�s
bajos en los obreros se�alizadores y pilotos.
5. Cin�tica y metabolismo en animales de laboratorio y en el ser
humano
Los mam�feros absorben f�cilmente el clordimeformo por el tracto
gastrointestinal y a trav�s de la piel. Lo excretan r�pidamente,
alrededor del 80% por la orina y del 10-15% por las heces. Al cabo de
unos 10 d�as se observan niveles bajos de residuos en todos los
tejidos y no hay indicios de bioacumulaci�n. Tras la administraci�n
cut�nea a seres humanos, se observa una excreci�n r�pida semejante por
la orina.
Varios metabolitos oxidados y conjugados del clordimeformo se
excretan por la orina; los principales son el demetilclordimeformo, la
N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. En
estudios in vitro se han observado los mismos metabolitos, siendo el
principal la 4-cloro- o-toluidina.
6. Efectos en mam�feros de laboratorio y en sistemas de pruebas
in vitro
En ensayos realizados en varias especies, el clordimeformo
administrado por v�a oral y cut�nea ha mostrado tener una toxicidad
aguda moderada. Los principales metabolitos han mostrado tener una
toxicidad oral baja en ensayos realizados en ratas. El clordimeformo
provoca solamente una ligera irritaci�n cut�nea y ocular en el conejo.
Tras una exposici�n breve o prolongada de ratones y ratas al
clordimeformo o a sus metabolitos pueden observarse cambios asociados
al tratamiento en los par�metros hematol�gicos y, con dosis elevadas,
indicios de hiperplasia del epitelio de las v�as biliares y de la
vejiga. El clordimeformo no aumenta la incidencia de tumores en las
ratas. En los ratones, despu�s de administrar a trav�s de la dieta
clordimeformo N-formil-4-cloro- o-toluidina o 4-cloro- o-
toluidina, se observa, de forma relacionada con la dosis, un aumento
de los tumores malignos hemorr�gicos de origen vascular clasificados
como hemangioendoteliomas malignos, que producen un aumento de la
mortalidad asociado con la dosis.
El clordimeformo no afecta a los par�metros reproductivos ni
tiene potencial teratog�nico.
Se ha ensayado el clordimeformo en una amplia variedad de pruebas
de genotoxicidad in vitro e in vivo. No se han comunicado
reacciones positivas a ninguna de esas pruebas, en las que se ensay�
clordimeformo en estado puro. Adem�s, se han comunicado varios
informes espor�dicos y no confirmados de actividad mutag�nica inducida
por la N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. Un
informe describe una inducci�n de la transformaci�n celular por efecto
tanto del clordimeformo como de la 4-cloro- o-toluidina. En el h�gado
de los ratones y las ratas expuestos se producen enlaces con el ADN.
Se ha observado un importante aducto hidrof�bico, en los ratones en
niveles mucho mayores que en las ratas.
El clordimeformo induce diversos efectos farmacol�gicos y
bioqu�micos en los animales, tales como cambios cardiovasculares,
hipotermia, hiperexcitabilidad, efectos sobre las funciones visual
central y auditiva y modulaci�n de las aminas biog�nicas y de las
enzimas que metabolizan f�rmacos.
7. Efectos en el ser humano
La intoxicaci�n aguda causa fatiga, n�useas, p�rdida del apetito
y, en casos m�s graves, somnolencia, cianosis, micci�n imperiosa,
cistitis, efectos cardiovasculares (taquicardia, bradicardia,
alteraciones del ECG), coma y choque. En general se produce una
recuperaci�n completa de la intoxicaci�n aguda.
Otros s�ntomas asociados a la exposici�n cr�nica al
clordime-formo son dolores abdominales, prurito y exantemas
(exposici�n cut�nea), as� como hematuria macrosc�pica y microsc�pica.
Se ha comunicado un gran n�mero de casos con s�ntomas cl�nicos de
exposici�n cr�nica tanto entre los obreros de las plantas de
producci�n de clordimeformo como entre los trabajadores agr�colas.
Los indicios epidemiol�gicos relacionados con la exposici�n
ocupacional muestran una fuerte asociaci�n entre la exposici�n al
metabolito 4-cloro- o-toluidina y la incidencia de c�ncer de vejiga
en el ser humano. Actualmente se dispone de pocos indicios de
asocia-ci�n entre la exposici�n al clordimeformo y el c�ncer de vejiga
en el ser humano.
8. Efectos en otros organismos en el laboratorio y en el medio
ambiente
No se observaron efectos significativos en poblaciones de hongos
de la tierra, bacterias o actinomicetos tras la aplicaci�n de
clordime-formo al suelo.
No existen datos de laboratorio sobre la toxicidad en los
invertebrados de agua dulce. El clordimeformo inhibi� el crecimiento
de larvas de ostras, con una CE50 de 5,7 mg/litro. La CL50 a las
96-h para los camarones rosados, �nico crust�ceo estudiado, fue de
7,1 mg/litro y los valores de la CL50 a las 96-h para los peces
oscilaron entre 1 y 54 mg/litro. No se dispone de datos sobre
toxicidad acu�tica cr�nica. La combinaci�n de datos obtenidos en el
laboratorio y sobre el terreno revela que el clordimeformo es t�xico
para una amplia gama de artr�podos terrestres no combatidos.
Con respecto a las abejas, se ha comunicado una DL50 de
toxicidad por contacto de 120 �g/g y una DL50 de toxicidad oral de
187 �g/g. No se produjo mortalidad sobre el terreno tras la exposici�n
de especies de abejas a los residuos presentes en la alfalfa tres
horas despu�s del rociado.
La CL50 en la dieta de varias especies de p�jaros oscil� entre
>1000 y >5000 mg/kg de dieta.
9. Evaluaci�n de los riesgos para la salud humana y efectos en el
medio ambiente
La exposici�n intensa durante la producci�n o la utilizaci�n,
debida posiblemente a la insuficiencia de las medidas de seguridad,
dio lugar a s�ntomas de intoxicaci�n aguda en los trabajadores. Como
se ha notificado que se ha suspendido la producci�n y la utilizaci�n
de clordimeformo en todo el mundo, no deber�an producirse nuevos casos
de intoxicaci�n aguda. Sin embargo, el riesgo asociado a la exposici�n
cr�nica, en particular el riesgo de c�ncer de vejiga, seguir� siendo
preocupante durante muchos a�os. Deber�a proseguir el reconoci-miento
m�dico de las personas que han estado muy expuestas en las plantas de
producci�n y en las comunidades rurales donde se haya aplicado
extensamente el clordimeformo.
Dado que el clordimeformo ha dejado de utilizarse, no se ha
realizado ninguna evaluaci�n cuantitativa de los riesgos para el medio
ambiente. A largo plazo no se prev�n efectos perjudiciales para el
medio ambiente como consecuencia de la utilizaci�n de clordime-formo
en el pasado.
10. Conclusiones y recomendaciones
El clordimeformo tiene un potencial significativo para causar
tanto toxicidad inmediata como a largo plazo en las personas
expuestas. La informaci�n de que se dispone actualmente apunta a una
asociaci�n entre una mayor incidencia de c�ncer de vejiga en el ser
humano y la exposici�n a la 4-cloro- o-toluidina y, en menor medida,
al clordimeformo.
El clordimeformo no persiste en el medio ambiente, por lo que a
largo plazo no se prev�n efectos perjudiciales como consecuencia de su
utilizaci�n en el pasado.
Se recomienda que el clordimeformo no se produzca comercialmente
ni se utilice en el futuro. Las reservas existentes deber�an
eliminarse sin correr riesgos.
Las personas expuestas profesionalmente al clordimeformo deber�an
participar en un programa de reconocimiento m�dico que comprenda
citolog�a urinaria y detecci�n de hematuria.
8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Acute poisoning incidents
The most comprehensive data on acute poisoning cases associated
with exposure to chlordimeform has come from China. Details of these
published poisoning cases are shown in Table 16. While many were due
to intentional ingestion, there were also cases of unintentional
poisoning as a result of consumption of contaminated food, as well as
occupational exposure to the spray. In a brief report prepared by Deng
et al. (1984) of a 1983 symposium in Hu-bei Province on chlordimeform
poisoning, which featured some 29 papers and 859 case studies, it was
stressed that the main cause of death was suppression of cardiac
contracture and dilation of blood vessels resulting in circulatory
failure.
Arima et al. (1976) described an unsuccessful suicide attempt
involving a 76-year-old male who ingested 100 g chlordimeform. He
vomited several times before arriving at hospital 50 min after
ingestion. He was lethargic with a weak pulse and cyanosis associated
with his lips, nails and skin. Methaemoglobin levels represented 17%
of total haemoglobin at 5 h but returned to normal levels by 2 days.
He regained consciousness by 50 h, although complained of headache and
blurred vision. The only treatment received was gastric lavage, which
was performed shortly after his arrival at the hospital.
8.2 Occupational exposure
8.2.1 Acute poisoning incidents
Currie (1933) reported nine cases of haematuria in workers
exposed to 4-chloro- o-toluidine (erroneously called 5-chloro- o-
toluidine) by inhalation or possibly by absorption through the skin.
All patients had difficulty urinating and had suprapubic pain. Most of
the workers were exposed to the material for only 1-2 days. Despite
efforts to control exposure to the chemical in the factory, further
cases of poisoning occurred, and manufacture was ceased. In a
follow-up study of three of the nine cases after 3 years, one patient
had no bladder trouble, one had a slight cystitis and urethritis, and
one had carcinoma of the bladder.
Jurincic et al. (1991) reported cases of acute haemorrhagic
cystitis in two men (aged 19 and 50) following involvement in cleaning
of a water-tank that had likely been used to transport chlordimeform.
Both developed abdominal pain, dysuria and haematuria in the evening
following exposure. Cystoscopy revealed haemorrhagic cystitis,
which was confirmed by bladder mucosa biopsy. Serum levels of
4-chloro- o-toluidine (referred to as 4-chloro-2-methylaniline) were
>1 mg/litre in both patients and urine levels were 16 mg/litre in the
Table 16. Case studies of acute chlordimeform poisoning in China
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
1 71 4 dermal absorption 5 ECG: 26 tachycardia; 6 bradycardia; 11 ectopic Wang & Tong (1992)
(28 male, 67 ingestion rhythm; 6 premature beat; 2 atrial fibrillation;
43 female) ventricular fibrillation; 1"Torsade de Pointes";
2 high pike P, 6 A-V block, 17 S-T depression,
3 inverse T, 1 S-T elevation ,7 Q-T elongation.
In 33 severe cases, 28 has ECG abnormalities;
38 moderate cases, 14 had abnormal ECG. Changes
in heart were found in 32 cases. Deaths were from
respiratory failure (3); ventricular fibrillation
(1) and supraventicular tachycardia (1).
2 4 ingestion(?) 0 Mild cyanosis, cystitis (2 cases occupational, Nui et al. (1990)
2 cases non-occupational; OPs also in formulation.
3 1 female ingestion 0 Jaundice on 3rd day which progressively deepened. Liu et al. (1990)
(30 years old) (150 ml) Hb 40 g/litre (70 g/litre on admission); complete
recovery, discharged on day 20.
4 52 ingestion 0 Loss of appetite (86.5%), urgency in urination He (1989)
(19 male, (20-350 ml) (84.6%), cyanosis (81.1%), coma (67.3%), miosis
33 female) (34.6%), mydriasis (15.4%),hypotension (38.5%),
tachycardia (32.7%), bradycardia (3.8%).
Impairment of liver and renal functions. 15 ECGs:
7 tachycardia, 2 bradycardia, Q-T elongation,
8 T-wave changes. Treated with methylene blue,
vitamin C, fresh blood transfusion and sopolamine.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
5 35 ingestion(?) 0 18 severe cases. Suggested use of 5-36 mg He et al. (1987)
atropine for chlordimeform poisoning and
50-128 mg for mixed pesticide poisoning.
6 1 female ingestion 0 Cyanosis, pin-point myosis. Given atropine Zhou (1987)
(30 years old) (80 ml 25% (15 mg/min) after lavage until total of 530 mg.
chlordimeform) Symptoms indicated overdose of atropine. Methylene
blue given, recovery and discharge at day 7.
7 23 4 contaminated food; 3 Mild case: nausea, vomiting, light cyanosis,
(6 male, 19 ingestion no somnolence. Moderate case: somnolence and Xu (1987)
17 female) (10-350 ml) light consciousness. Severe case: Marked
cyanosis, coma, shock. 5 ECG examined:
2 bradycardia, 1 tachycardia, A-V block,
S-T change. Mild impairment of liver renal
functions. Treated with methylene blue,
19 recovered.
8 1 female ingestion 0 Lavage and treatment led to recovery from danger. Liu & Li (1987)
(52 years old) (30 ml conc. Black stool, tachycardia occurred on 3rd day.
formulation) Complete recovery.
9 187 27 occupational spray; 13 Cyanosis (63.6%), nausea (49.2%), vomiting Ding & Huang (1987)
(66 male, 16 ingestion (20-250 ml (44.9%), mydriasis (32.1%), somnolence (33.7%),
121 female) 25% chlordimeform coma (32.1%), irritation in urination (30.5%),
formulation) hypotension. 27 ECGs: 4 tachycardia, 6 bradycardia,
4 S-T & T wave change, 2 pre-mature beat, 2
conductive blockage. 158 cases received methylene
blue and 174 recovered within 1-5 days.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
10 1 male occupational spray 0 Sprayed incorrect dilution spray. Complained of Gu et al. (1987)
(28 years old) fatigue, somnolence, loss of appetite, nausea,
vomiting, but no cyanosis, or signs of cystitis,
pulse 68, BP 128/94 (normally 120/80), MAO 25.12 U
(normally 38.87 U). Total chlordimeform in urine
on admission, 6.4 mg/ml. Recovered quickly.
11 6 (?) ? Main clinical features: drowsy, cyanosis, Chan (1985)
loss of consciousness, mydriasis, cystitis,
hypotension, bradycardia, myocarditis, shock,
methaemaglobinaemia.
12 47 ingestion 4 Symptoms: drowsy, cyanosis, cystitis, Ke (1985)
(11 male, 20-1900 ml 2 hypotension (severe case), 8 hypertension,
36 female) 10 ECG: 1 tachycardia and T-wave change.
13 25 ingestion(?) 1 Cyanosis, cystitis, hypotension, arrhythmia, Wang & Dong (1985)
S-T and T changes, Q-T elongation. Treatment
with gastric lavage, methylene blue, vitamin C
14 682 340 occupational spray; 25 279 cyanosis, 147 cystitis, 197 somnolence, Liu & Zhang (1985)
(331 male, 342 ingestion 211 coma, 81 shock, 109 tachycardia,
351 female) 64 bradycardia, 54 hypertension, 22 hypotension.
59 ECG: 8 premature beat, 4 Q-T elongation,
16 S-T and T changes.
15 358 283 ingestion 37 Somnolence, cyanosis, loss of appetite, Ding & Ru (1985)
haemorrhagic cystitis, often myocardium damage,
A-V block, cardiac failure.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
16 49 3 occupational 4 13 cases were severe. Clinical features: cyanosis Liu & Ke (1985)
46 non-occupational and cystitis with haematuria in all cases, most
with severe somnolence and a few with coma. Two
severe cases had hypothermia. Hypertension was more
common than hypotension. 10 ECGs: only one case of
T-wave change and tachycardia. Treatment with
methylene blue and lavage.
17 1 ingestion (300 ml 25% 0 Coma and cyanosis. Sudden cardiac arrest during Yang (1984)
(female, form.) lavage, rescued with mechanical respiration.
25 years old) Recovered after 14 days.
18 24 ingestion (15-150 ml 2 16 cyanosis, 14 drowsiness, 8 haematuria, Wu et al. (1983)
(11 male, 25% form.) 6 methaemoglobin, 1 cardiac arrest, which
13 female) recovered after resuscitation.
19 101 35 occupational spray, 2 89 chlordimeform alone cases: 66% cyanosis, Xie (1983)
(49 male, 66 ingestion; 32 comas, 14 cystitis, 14 hypotension, 3 cardiac
52 female) chlordimeform +Ops) failure. 8 ECGs: 6 myocardium damage (changes in
Q-T, S-T, and T waves). Treatment with methylene
blue, vitamin. C. All recovered.
12 cases with mixed pesticides (OPs and Ocs).
20 1 ingestion 1 Loss of consciousness, cyanosis, mydriasis, Wu (1982)
(female, 85 years) (30 ml) arrhythmia. ECG: bradycardia, T-wave changes.
Died on day 6.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
21 20 occupational spray 0 Farmers applied wrong dilution chlordimeform to Li et al. (1982)
(18 male, 2 female) cotton for one day. 7 drowsy, 10 loss of appetite,
4 cystitis. Symptomatic treatment. All recovered
in 2-4 days.
22 2 male ingestion (100 & 200ml) 0 Cyanosis, coma, respiratory-circulation failure, Zhang et al. (1976)
cystitis during 2nd day. Treatment with
methylene blue and atropine.
23 1 male 100 ml 0 Deep cyanosis, pulse 166. Xia & Gao (1977)
24 6 male occupational spray 0 Contamination of body surface and clothing. Su (1977)
Symptoms from day 1-4: cyanosis, haemorrhagic
cystitis, fatigue. Recovery after 18 days.
25 2 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Anonymous (1977)
no cyanosis, ECG normal. Symptomatically treated.
26 4 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Ming (1977)
cyanosis, somnolence, loss of appetite, haematuria,
RBC in urine for 20 days. Treatment: vitamin C,
antibiotics, coagulators.
51-year-old patient. Case studies of chlordimeform poisoning in China
due to occupational exposure are given in Table 17, together with a
brief account of the clinical features observed.
Table 17. Levels of urinary chlordimeform and its metabolites in hospitalized
workers (3 days following exposure) (Folland et al., 1978)
Worker Total aminesa Chlordimeform 4-Chloro-o-toluidine Conjugate
(mg/litre) (mg/litre) (mg/litre) (mg/litre)
1 11.0 1.10 3.75 6.25
2 15.2 2.16 4.16 8.67
3 2.6 0.04 1.25 1.17
a Measured following hydrolysis with 10N NaOH and 2 h at 80�C.
A brief account of the signs and symptoms of chlordimeform
poisoning and suggested interventions has been provided by Xue &
Loosli (1994).
8.2.2 Effects of long-term exposure
A report of an outbreak of haematuria in employees of a chemical
packaging plant in the USA over a 4-day period in 1975 was first
reported by Armstrong et al. (1975). Further details were described by
Folland et al. (1978). Nine of 22 workers who packaged chlordimeform
became severely ill with abdominal pain, dysuria, urgency to void, or
haematuria. In the previous year, four workers who had packaged the
chemical had similar symptoms. While six workers recovered within 7 to
18 days, three were hospitalized with symptoms which lasted from one
to two months. In these three workers, abnormalities noted were
microscopic haematuria and pyuria, proteinuria, low creatinine
clearance, elevated SGOT, prolonged BSP retention, elevated serum
amylase level, small bladder capacity, ureteral reflux and an intense
inflammatory reaction in three bladder biopsy specimens. The highest
concentrations of total amines were found in the urine of workers who
had become ill and were hospitalized. Low but measurable levels were
also found in workers who had not become ill. The major part of the
urinary amines was present as 4-chloro- o-toluidine or as conjugates.
Urinary total amines (following hydrolysis with 10 N sodium hydroxide
and 2 h at 80�C), as well as chlordimeform and 4-chloro- o-toluidine,
were measured in the hospitalized cases and are shown in Table 17.
The results of a monitoring programme on packaging workers in a
chlordimeform plant in the USA during 1976 have been described
(personal communication by J.W. Barnett, Ciba-Geigy Agricultural
Division, Greenborough, North Carolina, USA, to the California
Department of Food and Agricultural). The programme involved more than
100 workers and over 800 urine samples, monitoring for the presence of
red blood cells, for residues of chlordimeform metabolites, and for
clinical signs of toxicity in workers. Residues in urine samples were
reported to range from <0.05 to 50 mg/litre. There was no evidence of
microscopic haematuria found in the samples analysed nor of any
clinical signs of toxicity.
Four separate incidents resulting in 7 cases of frank haematuria
following industrial exposure were reported in the USA during the
period 1980-1984 (personal communication by J.W. Barnett, Ciba-Geigy
Agricultural Division, Greenborough, North Carolina, USA, to
Ciba-Geigy Ltd., Switzerland). Chemical cystitis, confirmed by
cystoscopy and biopsy, was diagnosed in one case while non-specific
bladder mucosal lesions were found in another. Six cases required
hospitalization, but all resolved after cessation of exposure.
In a study by Maddy et al. (1986), the results of a programme of
monitoring (1982-1985) the urine of more than 200 workers, who had
received training in the use of chlordimeform on cotton in California,
were described. Although urinalysis was unremarkable and no
significant cytological changes were found, a single case of bladder
cancer was detected in a pilot who had seven seasons of exposure to
chlordimeform.
By contrast, in the same period (1980-1984), no cases of
chlordimeform-induced haematuria occurred at manufacturing plants in
Switzerland and West Germany or formulation plants in Australia,
Columbia, Central America, Mexico and the USA. No cases of haematuria
reportedly resulted from application or use of chlordimeform in the
field (Anon., 1985b; personal communications by F.E. Pfister and P.
Duback (Ciba-Geigy Ltd., Agricultural Division, Switzerland) and by
N. Reckefus and K. Kossmann (Schering Aktiengesellschaft Agrochemical
Division, Berlin, Germany), 1985).
In a study by Lu et al. (1981), data on the effects of
chlordimeform exposure of factory workers in China was examined. In
this study, conducted in 1974, the air concentrations in the factory
were generally below 0.036 mg/m3, with shorter periods at higher
levels (0.108-0.33 mg/m3), during specific tasks. Skin contamination
on hands and forearms was 9.1 mg/h for chemical operators
and 964.2 mg/h for packers. The urinary excretion levels of
chlordimeform and 4-chloro- o-toluidine in controls were 0.015 and
0.042 mg/litre, respectively; in chemical operators they were
0.065 and 0.108 mg/litre, respectively; and in packers were 0.263 and
0.398 mg/litre, respectively. The health of the workers was examined
during the following 3 years (1974-1976). In 44-56 workers (equal
number of each sex) at an average age of 32 years and working period
of 2 years, the main finding were neurosis, sore throat and disorders
of the nervous system. There were no treatment-related effects on ECG,
liver function, clinical chemistry or urinalysis parameters.
In the same report (Lu et al., 1981), the effect of chlordimeform
exposure on rice field workers during 1974 was also examined. The
air concentration in the breathing space in all cases was below
0.02 mg/m3. Skin contamination was examined at the front of the
thorax, on the right forearm and on the right thigh. The applicators
applied chlordimeform for 4-5 h per day for 1-3 consecutive days,
wearing shirts and shorts with no other protection. Skin contamination
was from splash or from spray. The levels found from splash on thorax,
forearm and thigh were 0.0436, 0.0303 and 0.131 mg/100 cm2 per h,
respectively. The levels found from spray on thorax, forearm and thigh
were 0.235, 0.299 and 0.804 mg/100 cm2 per h, respectively. Medical
examination during 1974/1975 revealed complaints of light-headedness,
headache, fatigue, nausea, abdominal pain, skin itching and burning
sensation, and hypotension. There were no changes in ECG or blood
chemistry, and no reported cases of acute intoxication.
In a study by Li et al. (1985b), the health of 24 packers
(9 male, 15 female) in a chlordimeform manufacturing plant in Jiang-su
Province of China, was examined. The chlordimeform division of the
factory started manufacturing in 1975 and continued to do so at the
time of the study. The employees were working in the factory for
between 3 months and 4 years (average 1.5 years). Another 24 employees
from the kitchen and kindergarten served as controls. The air
concentration of chlordimeform (9 samples over 3 consecutive days) was
0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin contamination of the
hands and forearms was 110 �g/100cm2 (S.D. 39 �g/100 cm2). Urinary
chlordimeform levels were 0.20 � 0.13 mg/litre, and urinary
4-chloro-o-toluidine levels were 0.48 � 0.29 mg/litre. Medical
examination revealed no difference between packers and controls with
regard to symptoms, laboratory examinations including liver enzymes
and urinalysis parameters, chest X-rays, ECG, or other parameters of
cardiac function. The only symptom associated with exposure was skin
rashes and itching in 21% of exposed individuals. There was no
difference in the micronucleus counting in cultured peripheral
lymphocytes between exposed and control groups, nor were there any
positive mutagenicity results from urine samples with or without
glucuronidase or sulfatase in the medium.
In a further study in a Chinese chlordimeform manufacturing
factory, the health of employees involved in chlordimeform production
was studied for the 5-year period, 1977 to 1981 (Anon., 1985a). The
urinary chlordimeform plus 4-chloro- o-toluidine levels of packers
was the highest at 0.39 mg/litre, which significantly correlated with
skin contamination but not with air concentration. The major medical
findings were complaints of lightheadedness, disorders in sleep,
memory impairment, fatigue, loss of appetite, skin rashes and itching,
and skin spot pigmentation. There were no features of cystitis. ECG
findings in 36 employees indicated premature beats, partial A-V block,
tachycardia and bradycardia. There was no evidence of chromosome
aberrations in metaphase chromosomes of cultured peripheral
lymphocytes.
In a study by Tao et al. (1985), the health of 61 employees
(25 chemical operators, 36 packers) of a pesticide factory in China
was examined. Chlordimeform was produced in the factory for 5 months
per year. Air levels ranged from 0.074 to 0.160 mg/m3. Skin
contamination of packers (2.99 mg/day) was higher than for
hemical operators (0.784 mg/day). The urinary excretion rate of
chlordimeform plus 4-chloro- o-toluidine in packers was also higher
(0.513 mg/litre) than for chemical operators (0.206 mg/litre) or
controls (0.055 mg/litre). Symptoms of exposure noted in packers
included loss of appetite, fatigue, somnolence and skin rashes.
Hepatomegaly was observed. There was no difference in blood pressure
or heart rate. Abnormalities in ECG were noted in 10/61 exposed
employees compared to 6/76 controls.
In a study by Wang et al. (1987), the health of 16 applicators
(8 males, 8 females) spraying chlordimeform in cotton fields in
Xin-yang Farm in the Jiang-su Province of China over a 3-day period
(July 1986) was examined. Air levels in the breathing zone were
0.031 mg/m3 and the skin contamination was 4.17 mg per shift. Urinary
levels of chlordimeform plus 4-chloro- o-toluidine ranged between 1
and 3 mg/litre over the exposure period. A close correlation was noted
between the level of chlordimeform on the skin and the levels of
chlordimeform plus 4-chloro- o-toluidine in the urine. Rapid
excretion of chlordimeform plus 4-chloro- o-toluidine was noted
following exposure. There was no change noted in heart rate, blood
pressure, monoamine oxidase activity or urinalysis between exposed
individuals and controls. Mild chlordimeform exposure, however,
appeared to be related to loss of appetite and drowsiness.
In a study by Zhang et al. (1986a), conducted at the same farm
over the same period, 13 applicators (7 male, 6 female, 20-41 years of
age) were examined during spraying chlordimeform on cotton over three
consecutive days. Protective measures included gauze mask, plastic
gloves and plastic apron, although it was noted that extensive
contamination occurred. Air levels in the breath zone on each of the
three days were 0.011, 0.014 and 0.011 mg/m3, respectively. Skin
contamination on each of the three days was estimated by the method of
Zhang et al. (1986b) to be 10.99, 4.32 and 4.45 mg/person per day,
respectively. Urinary chlordimeform plus 4-chloro- o-toluidine
levels were measured over the 3 days of exposure and for 7 days
after cessation of exposure. Urinary levels ranged from a peak of
2.408 mg/litre during exposure to 0.036 mg/litre after 7 days.
Excretion of chlordimeform occurred very rapidly with the highest
level being detected in the sample collected at the end of each shift.
There was a close correlation between skin contamination and urinary
excretion. Metabolism occurred very rapidly since 4-chloro- o-
toluidine usually accounted for 70-93 % of the total amount in the
urine. Serum monoamine oxidase activity varied from 26.18 U to
19.26 U. Clinical symptoms were somnolence, headache, dizziness and
fatigue. Heart rate and blood pressure dropped on the 2nd and 3rd
days. Analysis of ECG indicated elongation of P-R, Q-T intervals. One
person complained of urgency and pain in urination, gross haematuria,
and the urinary chlordimeform plus 4-chloro- o-toluidine level was
more than 6 mg/litre. Another four subjects were found to have
microscopic haematuria. Liver function tests were normal.
In a study by Xue et al. (personal communication by S.-Z. Xue,
M. Wang, C.-M. Chu and X.-W. Zhou entitled "Effects of chlordimeform
on cardiovascular function in humans with occupational exposure",
1993), the effect of chlordimeform on cardiovascular function was
studied in exposed farm workers and in manufacturing workers in China.
Four separate exposure groups were studied. The first (short-term)
exposure group consisted of 16 farmers engaged in spraying
chlordimeform (0.125% solution) in a cotton field. Exposure was for a
3- to 4-h period for 3 consecutive days. The second (long-term)
exposure group consisted of 21 chlordimeform packers in a factory who
had worked for 6 months on this task. The third exposure group
consisted of 19 factory plant operators who had minimal exposure to
chlordimeform. The fourth group consisted of 9 control (non-exposed)
factory workers. Exposure was measured in the breathing zone air
(personal sampler for the working shift, usually 6 h), by dermal
contact (pooled aliquot of 10 swabs from various body sites), and by
urine measurements. In each case, chlordimeform and its major
metabolite, 4-chloro- o-toluidine, were measured. The cardiovascular
system function was determined by measurement of blood pressure, heart
rate and electrocardiography (ECG). Exposure data indicated the packer
group had a higher inhalation exposure (0.107 mg/m3) than the
sprayers (0.031 mg/m3). Dermal exposure, on the other hand, was
higher in the sprayers group (4.251 mg/m2) than in the packers group
(2.713 mg/m2). Urinary levels collected at the end of the working
shift indicated the highest level in sprayers (1.950 mg/litre)
compared to packers (1.267 mg/litre) and operators (0.097 mg/litre).
In the farmer group, analysis of cardiovascular activity indicated a
significant decrease in heart rate, and an increase in P-wave
duration, Q-T interval and amplitude of T-wave compared to the control
group. In the factory workers, the packers had significantly lower
diastolic and systolic blood pressure, and an increase in T-wave
amplitude compared to the plant operators. The heart beat of packers
was also higher than controls, but not significantly. The
cardiovascular function parameters of the plant operators were
slightly but not significantly different to those of controls.
Examination of the cardiovascular function parameters of the
packers during a month of continuous exposure indicated a relationship
between length of exposure, total urinary chlordimeform, and cardiac
function parameters (see Table 18). Analysis of the exposure-effect
correlation indicated the drop in blood pressure was the most
sensitive parameter, with the change in amplitude of the T-wave the
next most sensitive parameter. The changes of P-R interval were the
least sensitive.
Table 18: Cardiovascular function and urinary chlordimeform in factory workers
(personal communication by S.-Z. Xue, M. Wang, C.-M. Chu and X.-W.
Zhou entitled "Effects of chlordimeform on cardiovascular function
in humans with occupational exposure", 1993)
Parameter measured Duration of continuous exposure (days)
0 1 7 15 30
Total urinary 0.000 0.311 0.627 0.642 0.773
chlordimeform (mg/litre)
Systolic BP (mmHg) 111/8.6a 105/7.7b 105/12c 102/10d 102/9.6c
Diastolic BP (mmHg) 71/7.1 69/9.3 63/10c 65/8.2d 64/9.8d
Heart rate (beat/min) 64.3/9.9 69.6/8.6d 67.2/6.5 70.0/9.4 71.4/12d
Q-T interval (msec) 398/18.2 404/23.1 412/16.7d 418/22.3d 412/23.6d
P-R interval (msec) 131/215 140/178d 140/212d 141/200a 143/317d
a Figures are mean/standard deviation
b P < 0.001
c P < 0.01
d P < 0.05
The authors attributed major importance to the alteration in
cardiovascular function in relation to chlordimeform intoxication, and
in most cases considered cardiac failure to be the cause of death.
Recognition of the effects on cardiac function may have been
overlooked previously, firstly, because of the diversity of mild
changes induced by chlordimeform and, secondly, because of the
tendency to concentrate on the effects of the aniline-containing
metabolites, such as methaema-globinaemia, haematuria, and
haemorrhagic cystitis. A no-observed-effect-level (NOEL) of
0.1 mg/litre of urinary chlordimeform plus 4-chloro- o-toluidine
excretion is proposed as the threshold for effects on cardiovascular
function following long-term, exposure while 0.3 mg/litre is proposed
as the threshold for effects on cardiovascular function following
short term exposure, even as short as one day. While the
cardiovascular function parameters are unlikely to be useful as
indicators of exposure, an understanding of the mechanism of action
should assist in designing appropriate treatment.
In a post-exposure surveillance programme, the chlordimeform-
exposed group showed an increased prevalence of malignancy-associated
surface markers on exfoliated urine cells, compared to geographical
controls, but no tumours were found (Kenyon et al., 1993).
8.2.3 Epidemiological studies
8.2.3.1 4-Chloro- o-toluidine
In a retrospective epidemiological study by Ott & Langner (1983)
the mortality experience of 342 employees assigned to three aromatic
amine-based dye production areas between 1914 and 1958 was examined in
relation to duration of employment (<1 to 5 years) and interval since
entry into these areas. 4-Chloro- o-toluidine represented one of a
number of chemicals to which the workers were potentially exposed.
4-Chloro- o-toluidine and two other aromatic amines ( o-toluidine
and 4-chloro-acetyl- o-toluidine) to which the workers were exposed
have been shown to be carcinogenic in animal studies. No deaths due to
bladder cancer were observed, and no statistically significant
increases in mortality by work area or duration of exposure within
work area were found.
In a retrospective study by Stasik (1988; 1991) of 116 workers
occupationally exposed in Germany to 4-chloro- o-toluidine during
manufacture prior to 1970, eight cases of bladder cancer, diagnosed
between 1967 and 1985, were identified. This represents an incidence
more than 70-fold higher than expected. Although occupational exposure
to two other aromatic amines, o-toluidine and 6-chloro- o-
toluidine, may have occurred, analysis of the production process
indicated that exposure to 4-chloro- o-toluidine in the plant was
considerably higher than exposure to these other two chemicals. The
workers were exposed to relatively high levels (before 1970) for a
median of 14 years. In two cases, however, the exposure period was
only 1.5 and 4.0 years. No quantitative measurements of exposure were
available. Two of the patients had suffered from haemorrhagic cystitis
as a consequence of massive acute exposure to 4-chloro -o-toluidine
at 4 and 14 years, respectively, before the tumour was diagnosed. The
latency periods for these eight cases ranged from 17 to 38 years. The
significantly increased incidence of bladder cancer in this study is
remarkable.
8.2.3.2 Chlordimeform
An epidemiological study has been conducted on the incidence of
cancer deaths of employees and their relatives on Xin-Yang Farm in
Jiang-su Province of China (Gu et al., 1991). In this area,
chlordimeform has been applied aerially in large amounts since 1974,
in a relatively indiscriminate manner, with contamination of land,
ponds, creeks, and gardens of adjacent houses. The study involved 7321
people (3911 male, 3410 female and 1265 retired agricultural workers)
over the period 1 January 1971 to 30 June 1987. During this period,
there were 706 registered deaths (510 males, 196 females), of which
198 were cancer deaths (160 males, 38 females). The standardized
mortality ratio (SMR) was calculated on the basis of the specific
mortality due to cancer in the adjacent Hai-men County. Many of the
SMRs were significantly exceeded on the Xin-Yang Farm, as shown in
Table 19. The incidences of bladder cancer adjusted to the national
level were 2.65 (males) and 1.47 (females) per 100 000. The SMRs were
260 (males) and 420 (females). During the period 1 July 1987 to
30 June 1990, there were three more cases of bladder cancer (with one
death) among the cohort members. The authors concluded there is
evidence for an association between bladder cancer and exposure to
chlordimeform, but that further data is needed to strengthen this
association. It is noted that there was a high incidence of other
tumour types in this study which makes the association between bladder
cancer and exposure to chlordimeform more difficult to establish.
Table 19. Standardized mortality ratio (SMR) for workers on the
Xin-Yang farm (Gu et al., 1991)
Cause of death Adjusted mortality Standardized mortality ratio
(per 100 000) (95% C.I.)
Male Female Male Female
All deaths 785.1 610.0 134 (124-145) 139 (128-151)
All cancers 214.0 130 113 (107-120) 128 (117-139)
Oesophageal cancer 35.6 32.5 228 (208-249) 388 (352-428)
Stomach cancer 61.4 24.5 175 (161-190) 120 (110-130)
Liver cancer 31.6 6.9 40 (37-44) 27 (24-29)
Colon cancer 8.8 6.5 133 (123-145) 79 (72-86)
Lung cancer 34.2 16.9 135 (124-146) 147 (135-169)
Leukemia 3.9 5.6 144 (133-157) 260 (235-285)
Bladder cancer 4.1 3.0 197 (180-214) 750 (671-839)
Breast cancer - 16.0 - 380 (345-419)
Cervical cancer - 30.7 - 216 (198-234)
Further epidemiological data on the association between cancer
incidence and exposure to chlordimeform has been provided in papers by
Xue et al. (1990; 1991). A summary of the findings of epidemio-logical
studies between 1984 and 1988 is given in Table 20. Data from three
counties and one farm are shown. The counties are located close to one
another, with comparable environmental and socio-economic situations.
The agricultural products are mainly rice and cotton. County A acted
as a control, with little or no use of chlordimeform; County B was
the largest user of chlordimeform; and County C started using
chlordimeform at the earliest time. The results from the Xin-Yang farm
are included for comparison. A comparison between the mortality rate
in recent years (1984-1988) and the mortality rate in the years prior
to the use of chlordimeform in these counties and Xin-Yang farm is
shown in Table 21. There were excesses in the incidence of all deaths,
deaths from cancer, and urinary bladder cancer for both sexes,
although the data may not yet have reached the level of statistical
significance.
Table 21. Comparison of adjusted mortalities of urinary bladder
cancer between 1984-1988 (county) and 1973-1975
(prefecture) (Xue et al., 1990, 1991)
Item County A County B County C Xin-Yang Farm /
/ Prefecture / Prefecture Prefecturea
Male 1.52 / 1.10 0.77 / 0.77 1.12 2.65 / 1.02
SRR 1.38 1.04 1.10 2.65b
Female 0.41 / 0.35 0.46 / 0.17 0.55 1.47 / 0.35
SRR 1.17 2.71b 1.57b 4.20b
a The duration of observation was 1971-1987 (June 30)
b p < 0.05
In a retrospective study by Popp & Norpoth (1991) and Popp et al.
(1992), the exposure and incidence of bladder cancer in a German
chemical plant was examined. Chlordimeform was manufactured from
4-chloro- o-toluidine and production commenced in December 1965.
Production was not continuous, but rather was in response to orders,
so workers were subject to different periods of exposure (generally
8-12 weeks per year). Between 1965 and 1976, the exact levels of
exposure were not available because measurement of the concentration
in the air or monitoring of urine excretion was not carried out at
that time. In 1976, production was ceased in order to improve working
conditions and minimize human exposure. Production recommenced in 1980
with improved containment and monitoring of urinary excretion of
Table 20. Data on Epidemiological Studies with Chlordimeform during 1984-1988 (Xue et al., 1990, 1991)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
Year started using chlordimeform 1979 1977 1973 1973
Population (annual average) 993 549 1 076 456 736 037 8732
Average amount of chlordimeform 1.1 65.0b 29.8 89.2
used (g/Mu/year)a
All causes of mortality
Male 584.5 675.7 761.0 785.1
RR 1.2 (1.1-1.3) 1.3 (1.2-1.4) 1.3 (1.2-1.5)
Female 438.1 891.7 668.5 625.9
RR 2.0 (1.4-2.3) 1.5 (1.1-1.7) 1.4 (1.3-1.5)
Cardiovascular mortalityc
Male 143.2 167.7 221.6 -
RR 1.2 (1.1-1.3) 1.6 (1.4-1.7)
Female 138.2 280.2 234.8 -
RR 2.0 (1.9-2.2) 1.7 (1.6-1.8)
Respiratory mortalityc
Male 99.6 100.4 127.2
RR 1.0 (0.9-1.1) 1.3 (1.2-1.4)
Female 82.1 145.1 124.0
RR 1.8 (1.7-1.9) 1.5 (1.4-1.6)
Table 20. (con't)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
All cancer mortalityc
Male 188.5 246.6 232.2 214.9
RR 1.3 (1.2-1.4) 1.2 (1.1-1.3) 1.1 (1.1-1.2)
Female 101.7 227.1 145.5 130.0
RR 2.3 (2.0-2.5) 1.4 (1.3-1.6) 1.3 (1.2-1.4)
Bladder cancer mortalityc
Male 2.08 (95)d 0.90 (26) 2.10 (32) 4.10 (4)
RR 0.4 (0.39-0.47) 1.0 (0.9-1.2) 2.0 (1.8-2.2)
Female 0.40 (15) 0.20 (9) 0.90 (14) 3.00 (2)
RR 0.5 (0.46-0.55) 2.3 (2.1-2.5) 7.5 (6.7-8.4)
a The Mu is a measure of area equivalent to 1/15th acre
b Considered over the last 5 years
c All mortality figures were age-adjusted
d Figure in parentheses is the actual number of cases of bladder cancer
workers. Production finally ceased in 1986. The company identified 170
individuals who had come into contact with chlordimeform but many had
minimal exposure. The number of workers involved in the production of
chlordimeform was 49, and these comprised the study group. The period
under investigation was from the year of employment to the end of
1990. The expected incidence of bladder cancer (age- and sex-specific)
was extracted from the cancer registers of Saarland (1988), the former
German Democratic Republic (GDR) (1978-1982) and Denmark (1978-1982).
The standard incidence rate (SIR) was the ratio of the number of cases
observed to the expected number (see Table 22).
Table 22. Standard incidence rates (SIRs) of bladder carcinoma in a
group of 49 workers engaged in chlordimeform synthesis
(Popp et al., 1992)
Observed cases Expected number SIR 95% CI p value
7 0.078 (GDR) 89.7 35.6 - 168.6 0.000002
7 0.200 (Denmark) 35.0 13.9 - 65.7 0.00001
7 0.130 (Saarland) 53.8 21.3 - 101.1 0.000005
The average age for workers starting work was 30 (range 18-51), and
the exposure ranged from 3 to 956 days. By the end of 1990, an average
of 18 (10-25) years had passed since the start of exposure. Bladder
cancer was detected in 7 of the 49 subjects by the end of 1990. Of the
seven cases, six were diagnosed as transitional cell carcinoma and one
as papillary carcinoma. In five cases, the exposure period could be
determined, with an average of 575 days (range 291-766). The latency
period was an average of 19 years (range 15-23), with an average age
at diagnosis of 54 years (range 42-62). This study provides strong
evidence of an association between exposure to 4-chloro- o-toluidine
and human bladder cancer. All of the cases involved workers who were
exposed to 4-chloro- o-toluidine while synthesizing chlordimeform
before 1976. None of those workers who were handling the final
product, chlordimeform, had developed bladder cancer by the end of
1990.
In a historical cohort study (personal communication by P. Boyle
& G.J. Macfarlane to the IPCS, 1997), the bladder cancer incidence of
847 men involved in the manufacture of chlordimeform in Australia,
Switzerland, the United Kingdom and the USA was compared with that
expected on the basis of population-based cancer registry rates.
Subjects eligible to be included in the cohort were those who had been
employed in the production or formulation of chlordimeform or who had
otherwise been an integral part of a chlordimeform unit in a plant
where it had been produced or formulated for at least 6 months. The
results presented in Table 23 show an incidence rate of bladder cancer
which was significantly higher than expected. Overall, ten cases were
observed while 2.6 were expected. When the cohort was divided
according to whether members had been exposed to chlordimeform and
4-chloro- o-toluidine, or to chlordimeform alone, it was found that a
significant excess of risk of bladder cancer also occurred in those
workers thought not to have been exposed to 4-chloro- o-toluidine. In
this group of 592 men, 5 cases of bladder cancer were observed, while
1.4 cases were expected (SIR = 3.5, 95% CI (1.1, 8.3)). The authors
concluded that despite the lack of information on potentially
confounding factors in this study, the data indicated an association
between excess risk of bladder cancer in this cohort and one or more
aspects of the manufacture of chlordimeform.
Table 23. Bladder cancer risk in a cohort of men exposed to
chlordimeform (Boyle & Macfarlane, 1997)
Plant location Cohort numbers Bladder cancer cases
Observed Expected SIRa
Switzerland 273 4 0.72 5.6
USA (A)b 182 1 0.32 3.1
United Kingdom 174 3 1.06 2.8
USA (B)b 163 1 0.26 3.8
Australia 55 1 0.27 3.7
All plants 847 10 2.63 3.8
95% CIc
(1.8, 7.1)
a Standardized Incidence Ratio
b Different production sites
c Confidence Interval
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1 Laboratory experiments
9.1.1 Microorganisms
There are no data on the effects of chlordimeform on
microorganisms.
9.1.2 Aquatic organisms
9.1.2.1 Plants
There are no data on the effects of chlordimeform on aquatic
plants.
9.1.2.2 Invertebrates
There are no laboratory data on the effects of chlordimeform on
aquatic invertebrates.
9.1.2.3 Vertebrates
The toxicity of chlordimeform to some species of fish has been
determined (FAO/WHO, 1972; Mayer & Ellersieck, 1986), and is shown in
Table 24.
9.1.3 Terrestrial organisms
9.1.3.1 Plants
There are no data available for the effects of chlordimeform on
plants.
9.1.3.2 Invertebrates
Dittrich (1966, 1967) first reported studies on the efficacy of
chlordimeform as an acaricide with both ovicidal activity against
insect eggs and adulticidal activity. It killed adult spider mites
when applied as a vapour and as a spray, and penetrated plant tissues
where it was released in ovicidal quantities. Since then, its efficacy
as an insecticide has been studied in a wide range of species.
Chlordimeform not only has a direct lethal action, particularly
against eggs and early instar larvae of insects and acarines, but also
has important sublethal effects, including sterilization of eggs,
induction of hyperactivity, detachment of feeding ticks from hosts,
Table 24. Toxicity of chlordimeform to fish
Species Duration LC50 Reference
(h) (mg/litre)
Bluegill sunfish 24 1.0 FAO/WHO (1972)
48 1.0
96 1.0
Trout 24 11.7 (8.73-15.8) FAO/WHO (1972)
48 10.6 (7.80-14.50)
96 7.14 (4.70-10.80)
Cat fish 24 11.9 (8.98-15.9) FAO/WHO (1972)
48 8.72 (6.26-21.1)
96 4.54 (3.08-6.68)
Rainbow trout 24 29 Mayer & Ellersiek
96 13.2 (1986)
Channel catfish 24 20.7 Mayer & Ellersiek
96 20.2 (1986)
Carp 24 65a FAO/WHO (1972)
48 60a
96 50a
a Values are for TLm
colony dispersal behaviour in ticks and mites, anti-feeding effects
and disruption of mating and oviposition in Lepidoptera (Hollingworth,
1976).
Knowles & Shrivastava (1973) investigated its toxicity in house
flies. The LD50 was 25 �g/fly, a dose which was not at a practical
level for house-fly control, possibly due to the high rate of
metabolism in this species. Pimley (1986) investigated the toxicity of
chlordimeform to tsetse fly (Glossina morsitans). The median lethal
dose was approximately 0.4 �g/fly for unfed tsetse, and 100% mortality
was achieved with 2 �g/fly. Sublethal doses also caused a significant
depression of feeding activity.
The specificity of chlordimeform with regard to both eggs and
larval stages was examined by Streibert & Dittrich (1977). Eggs of the
three noctuid cotton pest moths, Heliothis armigera, Heliothis
virescens, and Spodoptera littoralis, when exposed to a saturated
atmosphere of 4 mg/m3, have very similar sensitivity to vaporized
chlordimeform. Agrotis ipsilon, also a noctuid, on the other
hand, is definitely less sensitive, and the coccinillid Epilachna
varivestis was the most tolerant. The larval stages of all of these
species were considerably less sensitive to chlordimeform vapour than
the egg stage, but chlordimeform does seem to cause a decrease in the
number of larvae in the field, possibly due to a repellent effect or a
behavioural change rather than a direct toxic effect. These results
with Spodoptera littoralis on the relative sensitivity of eggs and
larvae were confirmed in the studies of Salvisberg et al. (1980).
Davenport & Wright (1985) have also demonstrated the differential
susceptibility of adult and larvae of the noctuid moths, Spodoptera
littoralis and Heliothis virescens, and also highlighted the
significantly higher toxicity of the hydrochloride salt, compared to
the base, to the adults of both species.
Sparks et al. (1993) studied the effects of several insecticides
on ovicidal activity and alteration of octopamine titres in eggs of
the tobacco budworm (Heliothis virescens). Chlordimeform was highly
toxic to eggs of H. virescens. The authors reported that the
alteration in the biogenic amine octopamine titres by chlordimeform
might lead to a disruption in the ability of larvae to hatch from the
egg.
Crecelius & Knowles (1976) studied the sensitivity of the larvae
of the cabbage looper, Trichoplusia ni, to the toxic effects of
chlordimeform. Third instar larvae were more sensitive to the toxic
effects of chlordimeform than the fifth instar larvae, possible due to
slower penetration and slower metabolism of chlordimeform in the
latter instar larvae.
Bailey & Cathey (1985) demonstrated the effectiveness of
chlordimeform in reducing the percentage egg hatch of Lygus
lineolaris (Palisot de Beauvois) on pole bean ( Phaseolus
vulgaris L.) pods and cotton ( Gossypium hirsutum L.). A solution
of 0.09% chlordimeform, while not significantly reducing nymph
emergence from eggs deposited on bean pole pods, did significantly
reduce nymph emergence from eggs deposited on cotton plants.
Salvisberg et al. (1980) also demonstrated that Spodoptera
littoralis moths, when treated at doses as low as 10% of the LD50,
showed symptoms of hyperexcitation, which resulted in abnormal
patterns of egg-laying, a reduced number of eggs and lower fertility.
Further studies by Davenport & Wright (1987) on Spodoptera
littoralis have shown that chlordimeform hydrochloride significantly
reduces food consumption in fifth-instar larvae when incorporated into
the diet at a level of 0.1-10 mg/g or when topically applied. No
mortality occurred during feeding, but mortality was increased during
subsequent pupation and during emergence of the adult from the pupae.
In adult moths, egg laying was significantly decreased when
chlordimeform hydrochloride was applied topically (1 or 10 �g/moth).
Further evidence that behavioural changes may be more important
in reducing both the larval and insect populations following
chlordimeform treatment has been provided by Shimizu & Fukami (1983)
in studies of the larvae of the cabbage armyworm, Mamestra
brassicae, which showed a prolonged period of wandering behaviour in
the presence of chlordimeform. This may have caused a failure to find
or prepare a suitable site for pupation.
The behaviour-modifying effects of chlordimeform have also been
studied by Blackwell (1988a,b; 1889) in the larvae of the large
cabbage white butterfly, Pieris brassicae L. When placed on
chlordimeform-dipped leaves, the larvae become excited, in contrast to
their normal communal feeding behaviour. Locomotion was increased and
feeding was significantly reduced as a result of disaggregation
(Blackwell, 1988a). When applied directly to the larvae, chlordimeform
caused excitation and inhibition of feeding (Blackwell, 1989). Direct
application also caused developmental delays and mortality was
increased at later developmental stages (Blackwell, 1988b).
O'Brian et al. (1985) have studied the effect of insecticides on
beneficial insects, and in particular, the effect of chlordimeform on
the ecoparasitoid, Bracon mellitor, an important parasitoid of the
boll weevil (Anthonomus grandis grandis). Chlordimeform was found to
be more toxic to Bracon mellitor than to the boll weevil, and also
reduced the number of egg deposited.
The toxicity of chlordimeform hydrochloride to bees has been
examined after both ingestion and contact. Ingestion of a 0.3%
solution killed 18%, while ingestion of 0.15% killed approximately 7%.
Contact with the same solutions did not increase the mortality rate
(FAO/WHO, 1972). In a study by Johansen (1972), bees were exposed to
field-weathered residues of a range of insecticides, including
chlordimeform, on alfalfa foliage. Over a 24-h period, zero mortality
was obtained with alfalfa leafcutter bees, alkali bees and honey bees
exposed to 3-h-old residues.
9.1.3.3 Vertebrates
Fleming et al. (1985) examined the toxic and behavioural effects
of chlordimeform on the game bird, the bobwhite quail (Colinus
virginianus). When added to the diet of newborn chicks over a 7-day
period, the lethal concentration to chicks was 2835 mg/kg diet
(2169-3705 mg/kg diet). When chicks were fed a diet containing
chlordime-form at a concentration of 1000 mg/kg diet for 7 days, they
ate less, weighed less, travelled further from a fright stimulus in an
avoidance test, and had a high locomotor activity in an open-field
test than at lower dose levels. Chicks fed 100 or 1000 mg
chlordimeform/kg diet scored more highly than controls in a visual
cliff performance test. After a further 8 days on control diet, the
chicks fed 1000 mg/kg diet still scored higher than controls on the
avoidance test, but the open-field and cliff performance scores were
similar to those of controls.
In studies conducted on bobwhite quails and ducks, groups of
animals (10 per treatment group, 30 per control group) were fed
chlordimeform technical or chlordimeform 48% EC formulation in their
diets for 5 consecutive days. The dose levels were 0, 10, 31.6, 100,
316 or 1000 mg/kg diet. Both quails and ducks were tolerant of the
presence of chlordimeform in the diet. With the technical material,
one quail in each of the groups fed 100 and 316 mg/kg diet died,
while, with the formulation, one quail in each of the groups fed 316
and 1000 mg/kg diet died. All ducks survived treatment, even at the
highest dose level (FAO/WHO, 1972).
Hill et al. (1975) exposed three bird species, Japanese quail
(Coturnix japonica), ring-necked pheasant (Phasianus colchicus)
and mallard (Anas platyrhynchos), to chlordimeform. LC50 values for
Japanese quail and ring-necked pheasant were determined to be 1749 and
2608 mg/kg diet, respectively. The LC50 for mallard was determined to
be >5000 mg/kg diet; only 20% mortality was reported at the highest
exposure group, 5000 mg/kg diet. Hill & Camardese (1986) reported an
LC50 of 5079 mg/kg diet for Japanese quail exposed to chlordimeform.
9.2 Field Observations
9.2.1 Microorganisms
There are no field data on the effects of chlordimeform on
microorganisms.
9.2.2 Aquatic organisms
There are no field data on the effects of chlordimeform on
aquatic organisms.
9.2.3 Terrestrial organisms
9.2.3.1 Plants
The possibility that some insecticides might enhance the growth
of cotton plants has been suggested for some time. However, in the
case of chlordimeform, debate has continued as to whether this effect
is due to early season insect suppression (Bailey & Cathey, 1985) or
to a physiological effect (Phillips et al., 1977). Cathey & Bailey
(1987) have conducted controlled studies to examine the effects of
multiple applications of chlordimeform on the growth and development
of cotton ( Gossypium hirsutum L.) in both greenhouse and field
studies. Plants were sprayed six times with chlordimeform either alone
or in combination with fenvalerate at 5- to 7-day intervals, beginning
at the six-leaf stage of plant development. In the absence of early
season insects and when insect populations were maintained at a
relatively low level, no increases in lint yield occurred on the
chlordimeform-treated plants. However, yield increases did occur and
insect populations became lower in these treated plots when early
season insect populations in the test area were relatively high. None
of the treatments influenced the boll components, boll size, seed
index and lint percentage, or the first fibre properties, length,
strength and micronaire.
Field studies by Youngman et al. (1990) to determine the effects
of several insecticides on growth, fruiting patterns and yield of the
cotton plant, Gossypium hirsutum L., supported the conclusion that
chlordimeform does not significantly increase any plant growth
parameter when compared with the control.
9.2.3.2 Invertebrates
In a small field study conducted by Bull & House (1978), tests
were conducted in 0.05-ha plots of cotton to compare lower and more
frequent applications of chlordimeform with commercial mixtures of
insecticides against natural populations of Heliothis species. The
results indicated that the protection afforded was as good as with
commercial mixtures, probably as a result of careful observation of
the cotton to pinpoint the onset of significant egg production.
In a another small field study by Wilson (1981), the potential of
chlordimeform to control Heliothis species in cotton was tested
separately or in combination with amitraz and the microbial
insecticide, Bacillus thuringiensis. Chlordimeform was the most
efficient of the three materials and controlled Heliothis species
reasonably efficiently, but no control of the rough bollworm,
Earias hueglei was obtained. There was also good control of the
cotton looper, Anomis flava, and some indication of suppression of
mites and aphids was obtained.
The behaviour-modifying effects of chlordimeform have been
demonstrated in field studies by Uk & Dittrich (1986) on the adult
whitefly, Bemisia tabaci (Genn.), which attacks cotton in the Sudan.
At dose levels of 500-2500 g chlordimeform/ha together with 960 g
endosulfan/ha, there was evidence of irritation and mass emigration of
adults from treated cotton foliage without detectable direct
mortality.
9.2.3.3 Vertebrates
There are no field data on the effects of chlordimeform on
vertebrates.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
Production and use of chlordimeform has now ceased worldwide
and no further human exposure should occur. During the years of
chlordimeform production and use, dietary and incidental exposure to
chlordimeform occurred. Occupational exposure to chlordimeform and
4-chloro- o-toluidine (used as a starting material for synthesis)
occurred during manufacture and formulation, as well as during
application. The major route of exposure was through dermal
contamination. Application of chlordimeform occurred extensively by
aerial spraying, but it was also applied by ground-rigs as well as by
backpack spray equipment. Thus, agricultural workers were exposed
during mixing, loading, washing, and flagging operations. General
population exposure occurred through the consumption of food
containing residues of chlordimeform, and to a lesser extent through
by-stander exposure. In some cases, there was intentional ingestion of
the formulation.
Monitoring of urinary 4-chloro- o-toluidine has been found to be
a useful indicator of exposure.
10.1.2 Toxicity
In both experimental animals and humans, there is extensive
metabolism of chlordimeform, followed by rapid excretion via the
urine. A major urinary metabolite is 4-chloro- o-toluidine. In
experimental animals, symptoms of acute toxicity included neurotoxic
as well as cardiovascular effects. There was no evidence of
teratogenicity or reproductive effects. Following chronic
administration, there was a dose-related increase in
haemangioendotheliomas in mice. There was no treatment-related
increase in tumour incidence in rats. Most of the mutagenicity studies
with chlordimeform itself were negative, but there were sporadic
reports of genotoxicity with 4-chloro- o-toluidine and to a more
limited extent with N-formyl-4-chloro- o-toluidine.
In humans, chlordimeform has been shown to have both acute and
chronic effects. Acute poisoning caused fatigue, nausea and loss of
appetite, and, in more severe cases, somnolence, cyanosis, urgency in
urination, cystitis, cardiovascular effects (tachycardia, bradycardia,
ECG changes), coma and shock. The significance of the cardiovascular
effects in chlordimeform-induced mortality has only recently been
recognized. While there have been fatalities as a result of acute
chlordimeform exposure, in the majority of cases complete recovery
occurs. Symptoms of chronic exposure include those of acute exposure
as well as abdominal pain, skin itching and rashes, and gross or
microscopic haematuria.
With regard to carcinogenicity, the International Agency for
Research on Cancer (IARC) has concluded that there is limited
evidence in humans and sufficient evidence in experimental animals
for the carcinogenicity of 4-chloro- o-toluidine. The available
epidemiological data indicate an association between excess risk of
bladder cancer and exposures entailed in the manufacture of
chlordimeform. There is currently preliminary epidemiological evidence
of an association between chlordimeform use and excess risk of bladder
cancer.
10.1.3 Risk evaluation
With the withdrawal of the use of chlordimeform in agriculture
and a cessation of production worldwide, there is no longer any risk
associated with acute exposure except during the disposal of existing
stocks. The risk associated with chronic exposure, however,
particularly the risk of bladder cancer, will continue to be of
concern for many years. Human bladder cancer has a long latency
period, and establishing whether or not there is a link between
chlordimeform exposure and bladder cancer will require continued
health screening of significantly exposed individuals both from
manufacturing plants and from those rural communities where
chlordimeform was extensively used.
10.2 Evaluation of effects on the environment
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
11. CONCLUSIONS AND RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
AND THE ENVIRONMENT
11.1 Conclusions
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine, and, to a
lesser extent, chlordimeform.
Chlordimeform does not persist in the environment and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
11.2 Recommendations for protection of human health and the
environment
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
12. FURTHER RESEARCH
The following studies are needed:
1. epidemiological investigations on exposed populations.
2. studies on the dose-response relationship between exposure to
4-chloro- o-toluidine or chlordimeform and the induction of
urinary bladder cancer in humans.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Chlordimeform was considered by the International Agency for
Research on Cancer (IARC) in 1983. IARC noted that no published study
on the carcinogenicity of chlordimeform was available. However, it
considered data on the carcinogenicity of 4-chloro- o-toluidine and
concluded that the results of experiments on mice provided sufficient
evidence that 4-chloro- o-toluidine, a metabolite of chlordimeform,
is carcinogenic to experimental animals. No relevant data on humans
were available. IARC concluded the available data were inadequate to
evaluate the carcinogenicity of chlordimeform to humans (IARC, 1983).
The carcinogenicity of 4-chloro- o-toluidine, the breakdown
product and major metabolite of chlordimeform, was evaluated by IARC
in 1990 (IARC, 1990). On the basis of the available published data, it
was concluded that there is limited evidence in humans and
sufficient evidence in experimental animals for the carcinogenicity
of 4-chloro- o-toluidine. 4-chloro- o-toluidine and its strong acid
salts were classified as probably carcinogenic to humans (Group 2A).
Chlordimeform was considered at the 1971, 1975, 1978, 1979, 1980,
1985 and 1987 FAO/WHO Joint Meeting on Pesticide Residues (JMPR). In
1971, a temporary acceptable daily intake (ADI) for chlordimeform of
0-0.01 mg/kg body weight was established, and temporary maximum
residue levels (MRLs) were set for a number of crops and for the meat
and milk of cattle (FAO/WHO, 1972). In 1975, the temporary ADI was
maintained and some new temporary MRLs were established (FAO/WHO,
1976). In 1978, the temporary ADI was reduced to 0-0.0001 mg/kg body
weight, the temporary MRLs for all crops except cotton and cottonseed
were withdrawn, and the MRLs for meat and milk of cattle were set at
the level of detection (FAO/WHO, 1979). In 1979, 1980 and 1985, the
temporary ADI of 0-0.0001 mg/kg body weight was extended (FAO/WHO,
1980, 1981, 1986). In 1987, the temporary ADI for chlordimeform was
withdrawn (FAO/WHO, 1988).
REFERENCES
Abo-Khatwa N & Hollingworth RM (1972a) Chlordimeform: The relation of
the mitochondrial uncoupling to toxicity in the German cockroach. Life
Science, 11: 1181-1190.
Abo-Khatwa N & Hollingworth RM (1972b) Chlordimeform: Uncoupling
activity against rat liver mitochondria. Pest Biochem Physiol,
3: 358-369.
Ahmad S & Knowles CO (1971), Metabolism of N'-(4-chloro-o-tolyl)-
N,N-dimethyl-formamidine (chlorphenamidine) and 4'-chloro-o-
formotoluidine by rat hepatic microsomal and soluble enzymes.
Camp Gen Pharmacol, 2:189-197.
Anonymous (1971 a) Gas chromatographic method for the determination of
C 8514 residues in animal tissues, fat, milk and butter. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Anonymous (1971b) Chlordimeform: Working paper prepared for the 1971
Joint FAO/WHO Meeting on Pesticide Residues. Basel, Switzerland,
Ciba-Geigy Ltd and Berlin, Germany, Schering AG (Unpublished
document).
Anonymous (1977) [Report of two cases of chemical cystitis.] New Med,
8: 82. (in Chinese with English abstract).
Anonymous (1980a) CGA-95119: High performance liquid chromatographic
determination of urinary residues containing the 4-chloro-o-toluidine
moiety - Procedure No. 177. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Anonymous (1980b) [Study on the fate of chlordimeform residue on green
cabbage.] Shanghai, Shanghai Institute of Pesticides, Basic Metabolism
Group (in Chinese with English abstract).
Anonymous (1981) [On the residue of chlordimeform: Special Task Group
report.] In: [Selected papers on research on pesticides.] In: Chinese
Institute of Scientific Information and Bureau of Science and
Technology ed. [Selected papers on studies of pesticides.] Beijing,
Ministry of Chemistry (in Chinese with English abstract).
Anonymous (1985a) [Five years follow-up observation on the health
effect of chlordimeform on the employees in manufacturing.] Lab Med,
2: 7-10 (in Chinese with English abstract).
Anonymous (1985b) Chlordimeform: Joint submission to the WHO expert
group of the 1985 FAO/WHO Joint Meeting on Pesticide Residues. Basel,
Switzerland, Ciba-Geigy Ltd and Berlin, Germany, Schering AG
(Unpublished report).
Arima T, Morooka H, Tanigawa T, Imai M, Tsunashima T, & Kita S (1976)
Methemoglobinemia induced by chlorphenamidine. Acta Med Okayama.
30: 57-60.
Armstrong J, Graves O, Lovejoy G, Swiggart R, & Kimbrough R (1975)
Insecticide induced acute haemorrhagic cystitis. Morbid Mortal Wkly
Rep (Centres for Disease Control), 24: 374.
Arni P & M�ller D (1976a) Salmonella/mammalian-microsome mutagenicity
test with C8513 (chlordimeform HC1), Basel, Switzerland. Ciba-Geigy
Ltd (Unpublished report).
Arni P & M�ller D (1976b) Salmonella/mammalian-microsome mutagenicity
test with CGA 72647 (4-chloro-o-toluidine HCI), Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1976c) Salmonella/mammalian-microsome mutagenicity
test with CGA 72651 (N-formyl-4-chloro-o-toluidine), Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983a) C8513: Intrasanguine host-mediated assay
with S. typhimurium, Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Arni P & M�ller D (1983b) CGA 72647: Intrasanguine host-mediated assay
with S. typhimurium, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983c) C8513: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983d) CGA 72647: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983e) CGA 72651 pure: Chromosome studies in male
germinal epithelium, mouse spermatocytes. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Arni P, Hool G, & M�ller D (1983a) C8513: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P, Hool G, & M�ller D (1983b) CGA 72651: Chromosome studies in
male germinal epithelium, mouse spermatogonia. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Aziz SA & Knowles CO (1973) Inhibition of monoamine oxidase by the
pesticide chlordimeform and related compounds. Nature (Lond),
242: 417-418.
Bailey JC & Cathey GW (1985) Effect of chlordimeform on tarnished
plant bug (Heteroptera: Miridae) nymph emergence. J Econ Entomol,
78: 1485-1487.
Bailey BA, Martin RJ, & Downer RGH (1982) Monoamine oxidase inhibition
and brain catecholamine levels in the rat following treatment with
chlordimeform. Pest Biochem Physiol, 17: 293-300.
Balu K (1989) Evaluation of field worker exposure to chlordimeform by
using urine monitoring. In: Biological monitoring for pesticide
exposure: Measurement, estimation of risk reduction. Washington, DC,
American Chemical Society, pp 47-55 (ACS Symposium Series 382).
Bauer PJ & Cothren JT (1990) Growth-promoting activity of
chlordimeform. Agron J, 82: 73-75.
Beeman RW & Matsumura F (1973) Chlordimeform: A pesticide acting upon
amine regulatory mechanisms. Nature (Lond), 242: 273-274.
Beilstein P & M�ller D (1983) C8513 pure: BALB/3T3 cell transformation
assay. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Beilstein P & M�ller D (1984a) C8513 technical: L-5178Y/TK+/- mouse
lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Beilstein P & M�ller D (1984b) CGA 72647 technical: L1578Y/TK+/-
mouse lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Beilstein P & M�ller D (1984c) CGA 72647 technical: BALB/3T3 cell
transformation assay. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Benedict JH, Walmsley MH, Segers JC, & Treacy MF (1986) Yield
enhancement and insect suppression with chlordimeform (Fundal) on
dryland cotton. J Econ Etomol, 79: 238-242.
Benezet HJ & Knowles CO (1976) N'-(4-chloro-o-tolyl)-N-
methylformamidine (demethylchlordimeform) metabolism in the rat.
J Agric Food Chem, 24: 152-154.
Benezet HJ & Knowles CO (1981) Degradation of chlordimeform by algae.
Chemosphere, 10: 909-917.
Benezet HJ, Chang K, & Knowles CO (1978) Formamidine pesticides -
metabolic aspects of neurotoxicity. In: Pesticide venom neurotoxic,
pp 189-206.
Bentley P, Staubli W, Bieri F, Muecke W, & Waechter F (1985) Induction
of hepatic drug-metabolising enzymes following treatment of rats and
mice with chlordimeform. Toxicol Lett, 28: 143-149.
Bentley P, Bieri F, Muecke W, Waechter F, & Staubli W (1986a) Species
differences in toxicity of p-chloro-o-toluidine to rats and mice:
Covalent binding to hepatic macromolecules and hepatic non-parenchymal
cell DNA and an investigation of effects upon the incorporation of
[3H]-thymidine into capillary endothelial cells. Chem-Biol Interact,
57: 27-40.
Bentley P, Waechter F, Bieri F, Staubli W, & Muecke W (1986b) Species
differences in the covalent binding of p-chloro-o-toluidine to DNA.
Arch Toxicol, 9(suppl): 163-166.
Blackmore RH (1969a) Chlorphenamidine (Schering 36268): 2-year
repeated feeding study in dogs (Project No. 728-127). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969b) Chlorphenamidine (Schering 36268): 24-month
repeated feeding study in rats (Project No. 728-126). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969c) Chlorphenamidine (Schering 36268): three
generation reproduction study (Project No. 728-118). Falls Church,
Virginia, Hazleton Laboratories Inc (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969d) Chlorphenamidine (Schering 36268). Segment II:
Teratology Study -Rabbits (Project No. 728-157). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackwell A (1988a) Survival and development of Pieris brassicae
larvae upon application of sublethal doses of chlordimeform. Entomol
Exp Appl, 48: 149-156.
Blackwell A (1988b) The disruption of aggregation behaviour in Pieris
brassicae larvae by chlordimeform. Entomol Exp Appl, 48: 141-147.
Blackwell A (1989) The effects of chlordimeform on the behaviour of
Pieris brassicae larvae. Pestic Sci, 26: 11-19.
Blass W (1972a) Galecron residues in Japanese tea. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. RVA 5/72).
Blass W (1972b) Galecron residues in Swiss grapes (berries and juice).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 11/72).
Blass W (1972c) Galecron residues in Swiss wine 1971. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 16/72).
Bokisch AJ, Cox RT, & Walker RJ (1985) The actions of formamidine
pesticides on identified central neurons of Helix aspersa. Pestic:
Sci, 16: 495-503.
Boyes WK & Dyer RS (1984) Chlordimeform produces profound, selective,
and transient changes in visual evoked potentials of hooded rats. Exp
Neurol, 86: 434-447.
Boyes WK & Moser VC (1988) An alpha 2-adrenergic mode of action of
chlordimeform on rat visual function. Toxicol Appl Pharmacol,
92: 402-418.
Budris DM, Yim GK, Carlson GP, & Schnell RC (1983) Effect of acute and
repeated chlordimeform treatment on rat hepatic microsomal drug
metabolizing enzymes. Toxicol Lett, 18: 63-71.
Budris DM, Yim GK, & Schnell RC (1984) Effect of acute and repeated
chlordimeform treatment on rat hepatic microsomal drug metabolizing
enzymes. J Environ Pathol Toxicol Oncol, 5: 175-182.
Bull DL (1973) Metabolism of chlordimeform in cotton plants. Environ
Entomol, 2: 869-871.
Bull DL & House VS (1978) Effects of chlordimeform on insects
associated with cotton. Southwest Entomol, 3: 284-291.
Burkhard N (1971) Residues in milk, meat, liver and fat of a Swiss
cow. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
14/71).
Campanhola C & Plapp FW Jr (1989) Pyrethroid resistance in the tobacco
budworm (Lepidoptera: Noctuidae): Insecticide bioassays and field
monitoring. J Econ Entomol, 82: 22-28.
Candura SM, Marraccini P, Costa LG, Manzo L, Rossi A, Coccini T, &
Toninin M (1992) Calcium entry blockade as a mechanism for
chlordimeform induced inhibition of motor activity in the isolated
guinea-pig ileum. Pharmacol Toxicol, 71: 426-433.
Cathey GW & Bailey JC (1987) Evaluation of chlordimeform for cotton
yield enhancement. J Econ Entomol, 80: 670-674.
Chan Y-M (1985) [Observation and nursing of 6 cases of chlordimeform
poisoning.) Chin J Nurs, 20: 68-69 (in Chinese with English abstract).
Cheung MW, Kahrs RA, Nixon WB, Ross JA, & Tweedy BG (1989)
Determination of chlordimeform residues as 4-chloro-o-toluidine in
human urine using high-performance liquid chromatography. In:
Biological monitoring of pesticide exposure: Measuring and estimating
risk reduction. Washington, DC, American Chemical Society, pp 231-239
(ACS Symposium Series 382).
Cooper RL, Barrett MA, Goldman JM, Rehnberg GL, McElroy WK, & Stoker
TE (1994) Pregnancy alterations following xenobiotic-induced delays in
ovulation in the female rat. Fundam Appl Toxicol, 22: 474-480.
Costa LG & Murphy SD (1987) Interaction of the pesticide chlordimeform
with adrenergic receptors in mouse brain: an in vitro study. Arch
Toxicol, 59: 323-327.
Costa LG, Wu S, Olibet G, & Murphy SD (1988) Alpha2-adrenoceptors as
a target for formamidine pesticides: In vitro and in vivo studies in
mice. Toxicol Appl Pharmacol, 93: 319-328.
Costa LG, Wu S, Olibet G, & Murphy SD (1989) Formamidine pesticides
and alpha2-adrenoceptors: Studies with amitraz and chlordimeform in
rats and development of a radioreceptor binding assay. Neurotoxicol,
11: 405-411.
Costa LG, Gastel J, & Murphy SD (1991) The formamidine pesticides
chlordimeform and amitraz decrease hepatic glutathione in mice through
an interaction with alpha2-adrenoceptors. J Toxicol Environ Health,
33: 349-358.
Coye MJ, Lowe JA, & Maddy KJ (1986) Biological monitoring of
agricultural workers exposed to pesticides: II. Monitoring of intact
pesticides and their metabolites. J Occup Med, 28: 628-636.
Crecelius CS & Knowles CO (1976) Toxicity, penetration, and metabolism
of chlordimeform and its N-demethyl metabolite in cabbage looper
larvae. J Agric Food Chem, 24: 1018-1023.
Crowder LA & Whitson RS (1980) Fate of toxaphene, methyl parathion,
and chlordimeform combinations in the mouse. Bull Environ Contam
Toxicol, 24: 444-451.
Currie AN (1933) Chemical haematuria from handling 5-chloro-ortho-
toluidine. J Ind Hyg, 15: 205-213.
Davenport AP & Wright DJ (1985) Toxicity of chlordimeform and amitraz
to the Egyptian cotton leafworm (Spodoptera littoralis) and the
tobacco budworm (Heliothis virescens). Pestic Sci, 16: 81-87.
Davenport AP & Wright DJ (1987) Effect of the formamidine insecticide
chlordimeform on feeding and reproduction in the noctuid moth,
Spodoptera littoralis (Boisduval). Crop Prot, 6: 3-6.
Deng C-S, Zhang J-K, & Guo J-Y (1984) [Brief report of the symposium
on chlordimeform poisoning.] J Clin Med, 1: 51-53 (in Chinese with
English abstract).
Ding M-B & Ru X-Y (1985) [On diagnosis and treatment of chlordimeform
poisoning (analysis of 358 cases).] Jiangsu Med, 7: 30-31 (in Chinese
with English abstract).
Ding Y & Huang K-L (1987) [Analysis on clinical features of
chlordimeform poisoning.] Labor Med, 4: 8-10 (in Chinese with English
abstract).
Dittrich V (1966) N-(2-methyl-4-chlorophenyl)-N',N'-
dimethylformamidine (C-8514/Schering 36268) evaluated as an
acaricide. J Econ Entomol, 59: 889-893.
Dittrich V (1967) Systemic action of N-(2-methyl-4-chlorophenyl)-N',
N'-dimethylformamidine (C-8514/Schering 36268). J Econ Etomol,
60: 558-569.
Dittrich V & Loncarevic A (1971) New insecticides for Asiatic rice
borer control in paddy rice. J Econ Entomol, 64: 1225-1229.
Dyer RS & Boyes WK (1983) Chlordimeform differentially affects pattern
reversal-evoked and flash-evoked potentials in rats. Toxicologist,
3: 13.
Ehrhardt DA & Knowles CO (1970) Metabolism and translocation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine (chlorphenamidine) and
its hydrochloride salt in grapefruit seedlings. J Econ Entomol,
63: 1306-1314.
Ercegovich CD, Witkonton S. & Asquith DJ (1972) Disappearance of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine from six major fruit
crops. J Agric Food Chem, 20: 565-568.
Evans PD & Gee JD (1980) Action of formamidine pesticides on
octopamine receptors. Nature (Lond), 287: 60-62
Fan DF & Ge SD (1982) Gas-liquid chromatographic determination of
chlordimeform and its metabolites in cargo rice and husk. J Assoc Off
Anal Chem, 65: 1517-1520.
FAO/WHO (1972) Chlordimeform. In: 1971 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 3-45 (WHO
Pesticide Series, No. 1).
FAO/WHO (1976) Chlordimeform. In: 1975 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 85-104 (WHO
Pesticide Residues Series, No. 5).
FAO/WHO (1979) Chlordimeform. In: Pesticides residues in food - 1978:
Evaluations, Rome, Food and Agriculture Organization of the United
Nations, PP 51-78 (FAO Plant Production and Protection Paper 15 Sup.).
FAO/WHO (1980) Chlordimeform. In: Pesticide residues in food - 1979:
Evaluations. Rome, Food and Agriculture Organization of the United
Nations, pp 129-135 (FAO Plant Production and Protection Paper 20
Sup.).
FAO/WHO (1981) Chlordimeform. In: Pesticide residues in food - 1980:
Evaluations. Rome. Food and Agriculture Organization of the United
Nations, pp 89-96 (FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1986) Chlordimeform. In: Pesticide residues in food - 1985:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 17-29 (FAO Plant Production and
Protection Paper 72/2).
FAO/WHO (1988) Chlordimeform. In: Pesticide residues in food - 1987:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 25-26 (FAO Plant Production and
Protection Paper 86/2).
Fleming WJ, Heinz GH, & Schuler CA (1985) lethal and behavioral
effects of chlordimeform in bobwhite. Toxicology, 36: 37-47.
Folland DS, Kimbrough RD, Cline RE, Swiggart RC, & Schaffner (1978)
Acute hemorrhagic cystitis - Industrial exposure to the pesticide
chlordimeform. J Am Med Assoc, 239: 1052-1055.
Fritz H (1971) Reproduction study on C8514 (chlorphenamidine)
technical in rabbit - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 10071 004). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1975) Reproduction study on C8514 (chlorphenamidine)
technical in rat - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 227533). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1978a) C8513 technical: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327735).
Fritz H (1978b) CGA 72651: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327737).
Fritz H, Becker H, & M�ller D (1978) CGA 72647 technical: Dominant
lethal study (mouse). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782201).
Gaines TB & Linder RE (1986) Acute toxicity of pesticides in adult and
weanling rats. Fundam Appl Toxicol, 7: 299-308.
Gale GT Jr & Hofberg AH (1985) Gas chromatographic determination of
chlordimeform in pesticide formulations: Collaborative study. J Assoc
Off Anal Chem, 68: 589-592.
Galloway SM, Armstrong MJ, Reuben C, Colman S, Brown B, Cannon C,
Bloom AD, Nakamura F, Ahmed M, Duk S, Rimpo J, Margolin BH, Resnick
MA, Anderson B, & Zeiger E. (1987) Chromosome aberrations and sister
chromatid exchanges in Chinese hamster ovary cells: Evaluations of 108
chemicals. Environ Mol Mutagen, 10(suppl): 1-175.
Geissb�hler H, Kossmann K, Baunok I, & Boyd VF (1971) Determination
of total residues of chlorphenamidine in plant and soil material by
colorimetry of thin layer and electron capture gas chromatography.
J Agric Food Chem, 19: 365-371.
Geyer R & Fattal F (1987) HPLC determination of the metabolite
4-chloro-o-toluidine in the urine of workers occupationally exposed to
chlordimeform. J Anal Toxicol, 11: 24-26.
Ghali GZ & Hollingworth RM (1985) Influence of mixed function
oxygenase metabolism on the acute neurotoxicity of the pesticide
chlordimeform in mice. Neurotoxicology, 6: 215-238.
Gifkins MR & Jacobson RA (1980) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine (chlordimeform), C10H13CIN2.
Cryst Struct Commun, 9: 959-963.
Glowa JR (1986) Acute and sub-acute effects of deltamethrin and
chlordimeform on schedule-controlled responding in the mouse.
Neurobehav Toxicol Teratol, 8: 97-102.
Goldman JM, Cooper RL, Laws SC, Rehnberg GL, Edwards TL, McElroy WK, &
Hein JF (1990) Chlordimeform-induced alterations in endocrine
regulation within the male rat reproductive system. Toxicol Appl
Pharmacol, 104: 25-35.
Goldman JM, Cooper RL, Edwards TL, Rehnberg G, McElroy WK, & Hein JF
(1991) Suppression of the luteinizing hormone surge by chlordimeform
in ovariectomized, steroid-primed female rats. Pharmacol Toxicol,
68: 131-136.
Goldman JM, Stoker TE, Perreault SD, Cooper RL, & Crider MA (1993)
Influence of the formamidine pesticide chlordimeform on ovulation in
the female hamster - Dissociable shifts in the luteinizing hormone
surge and oocyte release. Toxicol Appl Pharmacol, 121: 279-290.
Gordon CJ & Watkinson WP (1988) Behavioral and autonomic
thermoregulation in the rat following chlordimeform administration.
Neurotoxicol Teratol, 10: 215-219.
Gordon CJ, Long MD, & Stead AG (1985) Thermoregulation in mice
following acute chlordimeform administration. Toxicol Lett, 28: 9-15.
Gross (1977) Metabolism of C 8514 (chlordimeform) in cotton. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 19/77).
Gr�bner P (1977) [Thin-layer chromatographic determination of
chlordimeform in cucumbers and apples.] Nahrung, 21: 339-345
(in German).
Gu Q, Kui N-D, Wang M & Xue S-Z (1987) [Report on a mild case of
chlordimeform poisoning.] J Environ Toxicol, 1: 61-62 (in Chinese with
English abstract).
Gu Y-E, Wang M, Gu Q, & Dai G-J (1991) [Epidemiologic study on the
cancer mortality of employees and their relatives in Xin-Yang farm.]
J Jiangsu Occup Health, 1: 5-8 (in Chinese).
Guth JA & Senn U (1969) Galecron - Dissipation in a Swiss soil 1969 as
determined by chemical analysis. Basel, Switzerland Ciba-Geigy Ltd
(Unpublished report).
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(suppl): 3-142.
He R-H (1989) [Analysis on the clinical symptoms of 52 acute
chlordimeform poisoned cases.] Occup Med, 16: 27 (in Chinese with
English abstract).
He D-Q, Peng B, & He P-L (1987) [Experience of treatment of
chlordimeform poisoning with atropine.] Hu-Nan Med, 4: 169 (in Chinese
with English abstract).
Henderson GR (1985) Report on the continuing use of chlordimeform for
cotton insect pest control by aerial and ground rig application in
1984/5 - NSW report. Pendle Hill, Ciba-Geigy Australia Ltd and
Kensington, Australia, Schering Pty Ltd (Unpublished report No. CGS
85/1).
Hermosin MC & Perez-Rodriguez JL (1981) Interaction of chlordimeform
with clay minerals. Clays Clay Miner, 29: 143-152.
Hill EF & Camardese ME (1986) Lethal dietary toxicities of
environmental contaminants and pesticides to Coturnix. Washington, DC,
US Department of the Interior, Fish and Wildlife Service, 147 pp (Fish
and Wildlife Technical Report No. 2).
Hill EF, Heath RG, Spann JW, & Williams JD (1975) Lethal dietary
toxicity of environmental pollutants to birds. Washington, DC, US
Department of the Interior, Fish and Wildlife Service, p 61 (Special
Scientific Report - Wildlife No. 191).
Hill DL, Shih T-W & Struck RF (1979) Macromolecular binding and
metabolism of the carcinogen 4-chloro-2-methylaniline. Cancer Res,
39: 2528-2531.
Hollingworth RM (1976) Chemistry, biological activity and uses of
formamidine pesticides. Environ Health Perspect, 14: 57-69.
Hollingworth RM, Leister J, & Ghali G (1979) Mode of action of
formamidine pesticides: an evaluation of monoamine oxidase as the
target. Chem-Biol Interact, 24: 35-49.
Holsapple MP & Yim GK (1981) Decreased gastrointestinal ulcerogenicity
of chlordimeform, a basic anti-inflammatory agent. Toxicol Appl
Pharmacol, 59: 107-110.
Homburger F, Friedell GH, Weisburger EK, & Weisburger JH (1972)
Carcinogenicity of simple aromatic amine derivatives in mice and Rats.
Toxicol Appl Pharmacol, 22: 280 (Abstract).
Honeycutt RC & Cassidy JE (1977) Extraction and characterization of
cottonseed metabolites from cotton injected with 14C-chlordimeform.
Greensboro, North Carolina, Ciba-Geigy Corporation (Unpublished report
No. ABR - 77087).
Hool G & Arni P (1983a) C8513 pure: Sister chromatid exchange studies
on somatic cells of Chinese hamster. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 830305).
Hool G & Arni P (1983b) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatogonia). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782902).
Hool G & Arni P (1983c) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782901).
Hool G & Arni P (1983d) CGA 72647 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 783101).
Hool G & Arni P (1983e) CGA 72647 pure: Sister chromatid exchange
studies on somatic cells of Chinese hamster. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 830312).
Hool G & Arni P (1983f) CGA 72651 pure: Chromosome studies in somatic
cells (Chinese hamster). Base], Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 774054).
Hool G & M�ller P (1978) C8513 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 40400577).
Hool G, Beilstein P, & Arni P (1983) C8513 technical: Chromosome
studies in male germinal epithelium (mouse spermatogonia). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 773062).
Hsu WH & Kakuk TJ (1984) Effect of amitraz and chlordimeform on heart
rate and pupil diameter in rats: Mediated by alpha2-adrenoreceptors.
Toxicol Appl Pharmacol, 73: 411-415.
Huang X-S, Ding S, & Chen P-H (1979) [Bioassay on the carcinogenicity
of chlordimeform.] In: Chinese Institute of Scientific Information and
Bureau of Science and Technology ed. [Selected papers on studies of
pesticides.] Beijing, Ministry of Chemical Industry, pp 789-792 (in
Chinese with English abstract).
Huang H-X, Xue K-L, & Shi G-J (1989) [Study on the status of pollution
on rice by chlordimeform.] Monograph submitted to the National
Pesticide Reviewing Committee (in Chinese with English abstract).
Hurni H & Sachsse K (1969) Report on the determination of the acute
oral LD50 to the dog of chlorfenamidin technical. Sisseln,
Switzerland, Tierfarm AG (Unpublished report).
IARC (1978) Some aromatic amines and related nitro compounds - hair
dyes, colouring agents and miscellaneous industrial chemicals. Lyon.
International Agency for Research on Cancer (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Man, Volume 16).
IARC (1983) Chlordimeform. Lyon, International Agency for Research on
Cancer, pp 61-72 (IARC Monographs on the Evaluation of Carcinogenic
Risk of Chemicals to Man, Volume 30)
IARC (1990) Some flame retardants and textile chemicals and exposures
in the textile manufacturing industry. Lyon, International Agency for
Research on Cancer, pp 123-137 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 48).
Ifflaender U (1977a) Distribution, degradation and excretion of
chlordimeform (CDF) in mice and rats. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 38/77).
Ifflaender U (1977b) Comparison of the urinary metabolite pattern of
mice and rats after oral application of "C-chlordimeform. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 39/77).
Iizuka H & Masuda T (1979) Residual fate of chlorphenamidine in rice
plant and paddy soil. Bull Environ Contam Toxicol, 22: 745-749.
IRPTC (1992) IRPTC data profile on chlordimeform. Geneva, United
Nations Environment Programme, International Register of Potentially
Toxic Chemicals.
Iwan J & Goller D (1975) Metabolic fate of chlordimeform in soil under
aerobic, anaerobic, and sterile conditions. Environ Qual Saf (Pestic),
3(suppl): 282-287.
Iwan J, Hoyer GA, Rosenberg D, & Goller D (1976) Transformations of
4-chloro-o-toluidine in soil: Generation of coupling products by
one-electron oxidation. Pestic Sci, 7: 621-631.
Janssen R, Boyes WK, & Dyer RS (1983) Effects of chlordimeform on the
brainstem auditory evoked response in rats. In: Hayes AW, Schnell RC,
& Miya TS ed. Developments in the Science and practice of toxicology,
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 533536.
Jiang J-K, Xu W-Y, Xu S-Q, Li J-L, Zhou F-D, Yang S-X, Zhong S-L, &
Zhu R-L (1985) [Survey on the hygienic status on spraying
chlordimeform with fine mist sprayer.] Labor Med, 2: 4-6 (in Chinese;
English translation).
Jiang X-Z, Li F, Xue S-Z, & Wang -L (1988) Carcinogenicity assay of
chlordimeform on dermal application in mice. Chin J Ind Hyg Occup Dis,
6: 138.
Johansen CA (1972) Toxicity of field-weathered insecticide residues to
four kinds of bees. Environ Entomol, 1: 393-394.
Johnson BT & Knowles CO (1970) Microbiological degradation of the
acaricide N-(4-chloro-o-tolyl)-N,N'-dimethylformamidine. Bull Environ
Contam Toxicol, 5: 158-170.
Johnson TL & Knowles CO (1981) Inhibition of rat platelet
5-hydroxytryptamine uptake by chlordimeform. Toxicol Lett, 9: 1-4.
Johnson TL & Knowles CO (1982) Interaction of formamidines with
the platelet 5-hydroxytryptamine uptake system. Gen Pharmacol,
13: 299-307.
Johnson TL & Knowles CO (1983) Influence of formamidines on biogenic
amine levels in rat brain and plasma. Gen Pharmacol, 14: 591-596.
Jurincic C, Horlbeck R, Metz K, & Klippel KF (1991) [Acute hemorrhagic
cystitis following chlordimeform poisoning.] Dtsch Med Wochenschr,
116: 816-818 (in German).
Kadir HA & Knowles CO (1981) Inhibition of rat brain monoamine oxidase
by insecticides, acaricides and related compounds. Gen Pharmacol,
12: 239-247.
Kale PG, Petty BT Jr, Walker S, Ford JB, Dehkordi N, Tarasia S, Tasie
BO, Kale R, & Sohni YR (1995) Mutagenicity testing of nine herbicides
and pesticides currently used in agriculture. Environ Mol Mutagen,
25: 148-153.
Kaloyanova F, Bainova A, & Zaprianov Z (1981) Inhibition of monoamine
oxidase activity after combined action of chlordimeform with the
antidepressant, nialamide. Arch Environ Contam Toxicol, 10: 1-8
Ke Z-H (1985) [Clinical features of 47 acute CDM poisonings by
swallowing.] Int Med, 11: 48-59 (in Chinese with English abstract).
Kenyon R, Garcia RJ, O'Grady HM, & Russell PJ (1993) A surveillance
programme for agricultural workers previously exposed to the suspect
bladder carcinogen chlordimeform - Proceedings of the 24th Congress of
the International Commission on Occupational Health, Nice, 26
September-1 October 1993.
Kimbrough RD (1980) Human health effects of selected pesticides,
chloroaniline derivatives. J Environ Sci Health, 15: 977-992.
Kimmel EC, Casida JE, & Ruzo LO (1986) Formamidine insecticides
and chloroacetanilide herbicides: Disubstituted anilines and
nitrosobenzenes as mammalian metabolites and bacterial mutagens.
J Agric Food Chem, 34: 157-161.
Knowles CO (1970) Metabolism of two accaricidal chemicals,
N'-(4-chloro-o-tolyl)-N, N-dimethylformamidine (chlorphenamidine)
and m-(((dimethylamino)methylene)amino)phenyl-carbamate hydrochloride
(formetanate). J Agric Food Chem, 18: 1038-1047.
Knowles CO (1976) Chemistry and toxicology of quinoxaline, organotin,
organofluorine, and formamidine acaricides. Environ Health Perspect,
14: 93-102.
Knowles CO (1991) Chlordimeform. In: Hayes WJ & Laws ER ed. Handbook
of pesticide toxicology. New York, London, San Francisco, Academic
Press Inc., vol 3. pp 1475-1486.
Knowles CO & Benezet HJ (1977) Mammalian metabolism of chlordimeform:
Formation of metabolites containing the urea moiety. J Agric Food
Chem, 25: 1022-1026.
Knowles CO & Johnson TL (1984) Effect of formamidines on
5-hydroxytryptamine uptake and biogenic amine levels in rat platelets.
Toxicology, 31: 91-98.
Knowles CO & Roulston WJ (1972) Antagonism of chlorphenaminine
toxicity to the cattle tick Boophilus microplus by piperonyl
butoxide. J Aust Entomol Soc, 11: 349-350.
Knowles CO & Sen Gupta AK (1969) Photodecomposition of the acaricide
N'-(4-chloro-o-tolyl)N,N-dimethylformamidine. J Econ Entomol,
62: 344-348.
Knowles CO & Sen Gupta AK (1970) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine-14C(Galecron) and 4-chloro-o-toluidine
14C metabolism in the white rat. J Econ Entomol, 63: 856-859.
Knowles CO & Shrivastava SP (1973) Chlordimeform and related
compounds: Toxicological studies with house flies. J Econ Entomol,
66: 75-79.
Konopka EA & Heymann H (1977) Salmonella/microsome mutagenicity test
with chlordimeform. Greensboro, North Carolina, Ciba-Geigy Corporation
(Unpublished report).
Kopp R, Humpel M, Kuhne G, Fuchs R, & Rzadkiewicz M (1977)
Pharmacokinetics of chlordimeform following single and repeated oral
administration of 0.03 and 3 mg in female mice (1.2 and 120 mg/kg).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Kossmann K (1980) Summary report on chlordimeform monitoring
programmes conducted by Ciba-Geigy and Schering at applicator sites in
1979. Berlin, Germany, Schering AG and Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report).
Kossmann K, Geissb�hler H, & Boyd VF (1971) Specific determination of
chlorphenamidine (N'(4-chloro-o-tolyl)-N,N-dimethylformamidine) in
plants and soil material by colorimetry and thin layer and electron
capture gas chromatography. J Agric Food Chem, 19: 360-364.
Krick F (1994) Method for the determination of chlordimeform in honey.
Washington, DC, US Food and Drug Administration (Laboratory
Information Bulletin 3817).
Kurtz PH, Shaw G, Kelter A, & Jackson RJ (1987) Assessment of
potential acute health effects in agricultural workers exposed during
the application of chlordimeform. J Occup Med, 29: 592-595.
Lang R (1984) The mammalian spot test and its use for testing of
mutagenic and carcinogenic potential: experience with the pesticide
chlordimeform, its principal metabolites and the drug lisuride
hydrogen maleate. Mutat Res, 135: 219-224.
Lang R & Adler ID (1982) Studies on the mutagenic potential of the
pesticide chlordimeform and its principal metabolites in the mouse
heritable translocation assay, Mutat Res, 92: 243-248.
Langauer M & M�ller D (1977) C8513 technical : Nucleus anomaly test in
somatic interphase nuclei (Chinese hamster). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 40050577).
Langauer M & M�ller D (1978a) CGA 72651 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 40550577).
Langauer M & M�ller D (1978b) CGA 72647 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 78/3002).
Leander J & MacPhail RC (1980) Effect of chlordimeform (a formamidine
pesticide) on schedule-controlled responding of pigeons. Neurobehav
Toxicol, 2: 315-321.
Leander JD, Tomlinson WT, Balster RL, & MacPhail RC (1984) Some
effects of the formamidine pesticide chlordimeform on the behaviour of
mice. Neurotoxicology, 5: 91-100.
Lemesch C, Wolf Y, & Gabai E (1987) Occupational exposure to
chlordimeform in Israel. Isr J Med Sci, 23: 1261-1262.
Leslie C, Reidy GF, Murray M, & Stacey NH (1988) Induction of
xenobiotic biotransformation by the insecticide chlordimeform, a
metabolite 4-chloro-o-toluidine and a structurally related chemical
o-toluidine. Biochem Pharmacol, 37: 2529-2535.
Li T-D, Liu B-J, & Mei C-J (1982) [Survey on 20 cases of acute
occupational chlordimeform poisoning.] Intermed Med, 3: 23-24
(in Chinese with English abstract).
Li F, Xue S-Z, Wang Y-L, Zhu S-Y, & Hu R-F (1985a) [Carcinogenic
action of chlordimeform given orally to mice in a lifetime
experiment.] Chin J Prev Med, 19: 154-156 (in Chinese with English
abstract).
Li F, Xue S-Z, Zhang R-W, Wang Y-L, Zhu Y-T, Sun S-S, Ding J-L, &
Feng B-J (1985b) (Studies on the health effect of employees in
chlordimeform production, and the mutagenic outcomes.] Labor Med,
2: 11-14 (in Chinese with English abstract).
Li F, Xue S-Z, Jiang X-Z, & Wang Y-L (1986) Report of dermal
carcinogenicity bioassay of chlordimeform in mice. J Ind Hyg Occup
Dis, 6: 76.
Limmer FB (1985) Ciba-Geigy and Schering world-wide exposure data
summary of personnel applying chlordimeform products covering the
period 1980-1984. Basel, Switzerland, Ciba-Geigy Ltd and Berlin,
Germany, Schering AG (Unpublished report).
Lin TH, North HH, & Menzer RE (1975) The metabolic fate of
chlordimeform [N-(4-chloro-o-tolyl) N'N'-dimethylformamidine] human
embryonic lung cell cultures. J Agric Food Chem, 23: 257-258.
Linder RE, Strader LF, Slott VL, & Suarez JD (1992) Endpoints of
spermatotoxicity in the rat after short duration exposures to fourteen
reproductive toxicants. Reprod Toxicol, 6: 491-505.
Ling W-Y, Zhang R-W, Gu Z-Y, Wang X-G, Zhang R-W, Jiang X-Z, Xue S-Z,
& Wang X-Z (1986) [Study on the urinary chlordimeform and its main
metabolite pCoT as index of biological monitoring in occupational
exposure.] Commun Labor Hyg Rail Syst, 8: 14-15 (in Chinese).
Lingren PD, Woffenbarger DA, Nosky JB, & Diaz M Jr (1972) Response of
Campoletis perdistinctus and Apanteles marginiventris to insecticides.
J Econ Entomol, 65: 1295-1300.
Liu Y-Z & Ke Z-H (1985) [Investigation on 53 cases of acute
chlordimeform poisoning.] Res Hyg, 14: 42-45 (in Chinese with English
abstract).
Liu X-L & Li W (1987) [Hemorrhage in upper digestive tract in
chlordimeform poisoning.] Occup Med, 14: 31 (in Chinese with English
abstract).
Liu Z & Mao Z (1980) [Studies on the analysis of metabolites of
pesticides. I. Gas chromatographic analysis of chlordimeform and its
metabolites in urine.] Hua Hsueh Pao, 38: 134-139 (in Chinese with
English Abstract).
Liu T-S & Zhang J-K (1985) [Poisoning due to chlordimeform, a new
pesticide.] Wuhan Med J, 9: 141-143 (in Chinese with English
abstract).
Liu H-G, Liu F-Q. Fang X-Z, & Wang X-M (1990) [Report on one case of
acute anemia due to chlordimeform poisoning.] PLA Med J, 15: 228
(in Chinese with English abstract).
Lu B-Q, Wang S-J, & Pong Y-F (1981) [Studies on the toxicology and
labor health of chlordimeform.] In: Chinese Institute of Scientific
Information and Bureau of Science and Technology ed. [Selected papers
on studies of pesticides.] Beijing, Ministry of Chemistry, pp 781-786
(in Chinese with English abstract).
Lund AE, Hollingworth RM, & Murdock LL (1979a) Formamidine pesticides:
A novel mechanism of action in lepidopterous larvae. Adv Pestic Sci,
3: 465-469.
Lund AE, Hollingworth RM, & Shamkland DL (1979b) Chlordimeform: Plant
protection by a sublethal, noncholinergic action on the central
nervous system. Pestic Biochem Physiol, 11: 117-128.
Lund AE, Hollingworth RM, & Yim GK (1979c) The comparative
neurotoxicity of formamidine pesticides. In: Neurotoxicology of
insecticidal pheromones, pp 119-137.
Lund AE, Shankland DL, Chinn D, & Yim GK (1978a) Similar
cardiovascular toxicity of the pesticide chlordimeform and lidocaine.
Toxicol Appl Pharmacol, 44: 357-365.
Lund AE, Yim GK, & Shankland DL (1978b) The cardiovascular toxicity of
chlordimeform: A local anesthetic-like action. In: Shankland DL,
Hollingworth RM, & Smyth T Jr ed. Pesticide and venom neurotoxicity.
New York, London, Plenum Press, pp 171-177.
Machin MV & Dingle JH (1977) Ultraviolet determination of
chlordimeform in cattle dipping baths and sprays. J Assoc Off Anal
Chem, 60: 696-698.
MacPhail RC & Leander JD (1980) Flavor aversions induced by
chlordimeform. Neurobehav Toxicol, 2: 363-365.
MacPhail RC & Leander JD (1981) Chlordimeform effects on
schedule-controlled behavior in rats. Neurobehav Toxicol Teratol,
3: 19-26.
Maddy KT, Knaak JB, & Gibbons DB (1986) Monitoring the urine of
pesticide applicators in California for residues of chlordimeform and
its metabolites, 1982-1985. Toxicol Lett, 33: 37-44.
Madhukar BV & Matsumura F (1979) Comparison of induction patterns of
rat hepatic microsomal mixed function oxidases by pesticides and
related chemicals. Pestic Biochem Physiol, 11: 301-308
Maitre L & Gfeller W (1975) Influence of compound 8514 (chlordimeform)
on the brain and liver monamineoxidase of rats. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Maitre L, Felner A, Waldmeier P, & Kehr W (1978) Monoamine oxidase
inhibition in brain and liver of rats treated with chlordimeform.
J Agric Food Chem, 26: 442-446.
Maqueda C, Perez Rodriguez JL, Martin F, & Hermosin MC (1983) A study
of the interaction between chlordimeform and humic acid from a typic
chromoxerert soil. Soil Sci, 136: 75-81.
Maqueda C, Morillo E, & Perez Rodriguez JL (1989) Interaction in
aqueous solution of certain pesticides with fulvic acids from a
spodosol soil. Soil Sci, 148: 336-345.
Matsumara F & Beeman RW (1976) Biochemical and physiological effects
of chlordimeform. Environ Health Perspect, 14: 71-82.
Mattern GC, Liu C-H, Louis JB, & Rosen JD (1991) GC/MS and LC/MS
determination of 20 pesticides for which dietary oncogenic risk has
been estimated. J Agric Food Chem, 39: 700-704.
Mayer FL & Ellersieck MR (1986) Manual of acute toxicity:
Interpretation and database for 410 chemicals and 66 species of
freshwater animals. Washington, DC, US Department of the Interior,
Fish and Wildlife Service, 505 pp (Resource Publication 160).
Ming Z-P (1977) [Report of four cases of chlordimeform poisoning,] New
Med, 8: 82-83 (in Chinese with English abstract).
Moore B (1971) Chlordimeform residues - A summary report. Pendle Hill,
Ciba-Geigy Australia Ltd (Unpublished report No. 71/11/358).
Moser VC, McCormick JP, Creason JP, & MacPhail RC (1988) Comparison of
chlordimeform and carbaryl using a functional observational battery.
Fundam Appl Toxicol, 11: 189-206.
Muecke W, Hamboeck H, & Arni P (1979) Metabolic and pharmacokinetic
aspects of chlordimeform in rats and mice. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 37/79).
Murakami M & Fukami J (1974) Effect of chlorphenamide and its
metabolites an HeLa cells. Bull Environ Contam Toxicol, 11: 184-188.
Nathanson JA & Hunnicutt EJ (1981) N-demethylchlordimeform: A potent
agonist of octopamine-sensitive adenylate cyclase. Mol Pharmacol,
20: 68-75.
NCI (1979) Bioassay of 4-chloro-o-toluidine hydrochloride for possible
carcinogenicity. Bethesda, Maryland, National Cancer Institute
(Technical Report Series No. 165).
Neumann R & Voss G (1977) MAO inhibition, an unlikely mode of action
for chlordimeform. Experientia (Basel), 33: 23-24.
Nixon WB & Neal BE (1983) Chlordimeform residues in human urine
following dermal application. Greensboro, North Carolina, Ciba-Geigy
Corporation (Unpublished report No. ABR-83017).
Nui J-H, Ma H, Muo J-T, & Wu W-L (1990) [Report on 4 cases of
poisoning due to chlordimeform and organophosphate pesticides.]
Shang-Dong Ind Hyg, 2: 35 (in Chinese with English abstract).
O'Brien PJ, Elzen GW, & Vinson SB (1985) Toxicity of azinphosmethyl
and chlordimeform to parasitoid braconmellitor (Hymenoptera:
Braxonidae): lethal and reproductive effects. Environ Entomol,
14: 891-894.
Olson KL, Boush GM, & Matsumura F (1978) Behavioral effects of
perinatal exposure of chlordimeform in rats. Bull Environ Contam
Toxicol, 20: 760-768.
Ott MG & Langner RR (1983) A mortality survey of men engaged in the
manufacture of organic dyes. J Occup Med, 25: 763-770.
Palmer WA, Dingle JHP, & Heath AB (1977) Residues of chlordimeform in
bovine tissues and milk following application by a handspray. Aust J
Exp Agric Anim Husb, 17: 380-384.
Parkes HG (1984) Epidemiology of aromatic amine cancers. In: Searle CE
ed. Chemical carcinogens. Washington, DC, American Cancer Society,
pp 277-301 (ACS Monograph No. 182).
Perez-Rodriguez JL & Hermosin MC (1979) Adsorption of chlordimeform by
montmorillonite. In: Mortland MM & Farmer VC ed. Proceedings of the
International Clay Conference, Oxford, 1978. Amsterdam, Oxford, New
York, Elsevier Science Publishers, pp 227-234.
Pfister WR, Chin C, Noland V, & Yim GK (1978a) Similar pharmacological
actions of chlordimeform and local anesthetics. Pestic Biochem
Physiol, 9: 148-156.
Pfister WR, Hollingworth RM, & Yim GW (1978b) Increased feeding in
rats treated with chlordimeform and related formamidines: A new class
of appetite stimulants. Psychopharmacology (Berlin), 60: 47-51.
Phillips JR, Herzog GA, & Nicholson WF (1977) Effect of chlordimeform
on fruiting characteristics and yield of cotton. Ark Farm Res, 26: 4.
Pimley RW (1986) Toxicity of chlordimeform to the tsetse fly Glossina
morsitans. Insect Sci Appl, 7: 663-666.
Popp W & Norpoth K (1991) [Carcinogenic effects of chlordimeform and
4-chloro-o-toluidine.] Dtsch Med Wochenschr, 116: 1454 (in German).
Popp W, Schmieding W, Speck M, Vahrenholz C, & Norpoth K (1992)
Incidence of bladder cancer in a cohort of workers exposed to
4-chloro-o-toluidine while synthesising chlordimeform. Br J Ind Med,
49: 529-531.
Puri E & M�ller D (1983a) C8513 technical: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830304).
Puri E & M�ller D (1983b) CGA 72647 pure: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830311).
Puri E & M�ller D (1983c) C8513 technical: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830306).
Puri E & M�ller D (1983d) CGA 72647 pure: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Lid
(Unpublished report No. 830313).
Rashid KA, Ercegovich CD, & Mumma RO (1984) Evaluation of
chlordimeform and degradation products for mutagenic and DNA-damaging
activity in Salmonella typhimurium and Escherichia coli. J Environ Sci
Health, B 19: 95-110.
Rieger JA, Robinson CP, Cox P, & Horst MA (1981) Cardiovascular
actions and interactions of chlordimeform in the dog. Bull Environ
Contam Toxicol, 27: 707-715.
Rieger JA, Allen LV Jr, & Fisher RC (1985) Extraction and analysis of
chlordimeform and demethylchlordimeform from human issue samples. Bull
Environ Contam Toxicol, 35: 397-405.
Robinson CP & Smith PW (1977) Lack of involvement of monoamine oxidase
inhibition in the lethality of acute poisoning by chlordimeform.
J Toxicol Environ Health, 3: 565-568.
Robinson CP, Smith PW, Zelenski JD, & Endecott BR (1975) Lack of an
effect of interference with amine mechanisms on the lethality of
chlordimeform in the rat. Toxicol Appl Pharmacol, 33: 380-383.
Rose JA (1969a) Degradation of chlorphenamidine by rat liver and
spleen homogenates with particular reference to the possible formation
of azo derivatives. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Rose JA (1969b) Formation of a symmetrical azo-derivative from
4-chloro-o-toluidine by coupled peroxidatic reactions. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Ross MSF & Leisten O (1989) Analysis of trace amounts of chlordimeform
in urine samples. Am Clin Lab, 8: 22-24.
Sachsse K, Suter P, Zak F, & Hess R (1978a) Lifespan feeding study in
mice with N-formyl-4-chloro-o-toluidine - Final report (Project No.
Siss M 4762/1. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Zak F, & Hess R (1978b) Lifespan feeding study in
mice with 4-chloro-o-toluidine-HCI - Final report (Project No. Siss M
04760/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979a)
Chlordimeform-HCL: 60-day feeding study in mice - Final report. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979b)
Chlordimeform-HC1: 60-day feeding study in rat - Final report (Project
No. Siss 6138). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980a)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in the rat - Final
report (Project No. 404078). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980b)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in mice - Final
report (Project No. 404178). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980c) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
chlordimeform-HC1 - Final report (Project No. Siss. R 05658/1). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980d) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
N-formyl-4-chloro-o-toluidine - Final report (project No. Siss
5688/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980e) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
4-chloro-o-toluidine- HC1 - Final report (project No. Siss R 05687/1).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report),
Salvisberg W, Neumann R, & Voss G (1980) Chlordimeform: Mode of toxic
action in various development stages of Spodoptera littoralis. J Econ
Entomol, 73: 193-196.
Sen Gupta AK & Knowles CO (1969) Metabolism of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine by apple seedlings.
J Agric Food Chem, 17: 595-600.
Sen Gupta AK & Knowles CO (1970) Galecron 14C(N'-(4-chloro-o-tolyl)
-N,N-dimethylformamidine) metabolism in the dog and goat. J Entomol,
63: 951-956.
Shimizu T & Fukami J (1983) Effect of chlordimeform and its analogues
on wandering behavior of the cabbage armyworm, Mamestra brassicae L.
(Lepidoptera:Noctuidae). Appl Entomol Zool, 18: 554-557.
Shopp GM Jr, McCay JA, & Holsapple MP (1985) Suppression of the
antibody response by a formamidine pesticide: dependence on the route
of exposure. J Toxicol Environ Health, 15: 293-304.
Sparks TC, Leonard BR. Schneider F. & Graves JB (1993) Ovicidal
activity and alteration of octopamine titers: Effects of selected
insecticides on eggs of the tobacco budworm (Lepidoptera: Noctuidae).
J Econ Entomol, 86: 294-300.
Stasik MJ (1988) Carcinomas of the urinary bladder in a 4-chloro-o-
toluidine cohort. Int Ach Occup Environ Health, 60: 21-24.
Stasik MJ (1991) [Bladder cancer from 4-chloro-o-toluidine.] Dtsch Med
Wochenschr, 116: 1444-1447 (in German).
Stoker TE, Goldman JM, Cooper RL, & McElroy WK (1991) Influence of
chlordimeform on alpha-adrenergic receptor-associated mechanisms of
hormonal regulation in the rat: pituitary and adrenocortical
secretion. Toxicology, 69: 257-268.
Strasser FF & M�ller D (1983a) CGA 72651 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782323).
Strasser FF & M�ller D (1983b) CGA 72647 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Lid (Unpublished report No. 782326).
Strasser FF & M�ller D (1984a) CGA 72647 technical: Point mutation
assay with mouse lymphoma cells, host-mediated assay. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 782325).
Strasser FF & M�ller D (1984b) CGA 72651: Point mutation assay with
mouse lymphoma cells. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782322)
Streibert HP & Dittrich V (1977) Toxicological response of insect eggs
and larvae to a saturated atmosphere of chlordimeform. J Econ Entomol,
70: 57-59.
Strong MB & Bull MS (1985a) Levels of chlordimeform equivalents found
in the urine of personnel involved in the ground rig application of
CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Ltd (Unpublished report
No. 85/6/1030).
Strong MB & Bull MS (1985b) Levels of chlordimeform equivalents found
in the urine of personnel involved in the aerial application of CGS
500 and CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Lid (Unpublished report
No. 85/6/1029).
Su S-T (1977) [Report of 6 cases of chlordimeform poisoning]. Intermed
Med, 6: 19 (in Chinese with English abstract).
Su GC & Zabik MJ (1972) Photochemistry of bioactive compounds:
Photolysis of arylamidine derivatives in water. J Agric Food Chem,
20: 320-323.
Suter P, Luetkemeier H, Sachsse K. Zak F, & Hess R (1976a) 60-Day
feeding study in mice with 4-chloro-o-toluidine HC1 (Project No. Siss
4760). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Suter P, Luetkemeier H, Sachsse K, Zak F, & Hess R (1976b) 60-Day
feeding study in the rat with 4-chloro-o-toluidine HC1 (Project No.
Siss 4760). Basel, Switzerland. Ciba-Geigy Lid (Unpublished report).
Suter P, Zak F, Sachsse K, & Hess R (1978) lifespan feeding study in
mice with chlordimeform-HC1 - Final report (Project No. Siss. M
04761/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Tao X-M, Sun R-M, Wang M-S, Xue S-Z, Zhang R-W, Jiang X-Z & Wang Y-L
(1985) [investigation on the hygienic status of chlordimeform
production in Cai-Qiao pesticide factory.] Chin J Ind Hyg Occup Dis,
3: 272-274 (in Chinese).
Thomas WD, Craig GK, & Stacey NH (1990) Effects of chlordimeform and
its metabolite 4-chloro-o-toluidine on rat splenic T, B and
tumoricidal effector cells. Immunopharmacology, 19: 79-86.
Uk S & Dittrich V (1986) The behavior-modifying effect of
chlordimeform and endosulfan on the adult whitefly Bemisia tabaci
(Genn.) which attacks cotton in the Sudan. Crop Prot, 5: 341-347.
Voss G (1971) Galecron residues - German hops (green cones) and beer
1970. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
42/71).
Voss G & Burkhard N (1971) Galecron residues in milk, meat, fat, liver
and kidney of a Swiss cows. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. RVA 24/7/S).
Voss G, Kossmann K, & Geissb�hler H (1973) Chlordimeform. In: Sherma J
& Zweig G ed. Analytical methods for pesticide plant growth
regulators. New York, London, Academic Press Inc., vol 7, pp 211-230.
Wang X-Z & Tong Q (1992) [Observation on 71 EKG of acute chlordimeform
poisoning cases.] Labor Prot Chem Ind, 13: 61-62 (in Chinese with
English abstract)
Wang CM, Narahashi T, & Fukami J (1975) Mechanism of neuromuscular
block by chlordimeform. Pestic Biochem Physiol, 5: 119-125.
Wang M, Zhou Z-J, Li H-L, Zhang R-W, Xue S-Z, Hong Z-R, Gu Q, Wang
X-M, & Feng Y-Q (1987) [Investigation on the hygienic status of
farmers spraying chlordimeform.] Chin J Ind Hyg Occup Dis, 5: 50-53
(in Chinese)
Wang Y-S & Dong C-R (1985) [Experience on treatment of 25 cases of
chlordimeform poisoning.] Sichuan Med, 6: 207 (in Chinese with English
abstract).
Watanabe H, Tsuda S, & Fukami J (1975) Effect of chlordimeform on
rectus abdominum muscle in frog. Pestic Biochem Physiol, 5:150-154.
Watanabe Y & Matsumura F (1987) Comparative metabolism of sulfamidine
and chlordimeform in rats. J Agric Food Chem, 35: 379-384
Watkinson WP (1985) Effects of chlordimeform on cardiovascular
functional parameters: Part 1. Lethality and arrhythmogenicity in the
geriatric rat. J Toxicol Environ Health, 15: 729-744.
Watkinson WP (1986a) Effects of chlordimeform on cardiovascular
functional parameters: Part 2. Acute and delayed effects following
intravenous administration in the postweanling rat. J Toxicol Environ
Health, 19: 195-206.
Watkinson WP (1986b), Effects of chlordimeform on cardiovascular
functional parameters: Part 3. Comparison of different routes of
administration in the postweanling rat. J Toxicol Environ Health,
19: 207-217.
Watkinson WP & Gordon CJ (1987) Effects of chlordimeform on heart rat
and body temperature of unanesthetized, unrestrained rats. Toxicol
Lett, 35: 209-216.
Watkinson WP, Highfill JW, & Gordon CJ (1989) Modulating effect of
body temperature on the toxic response produced by the pesticide
chlordimeform in rats. J Toxicol Environ Health, 28: 483-500.
Weisburger EK, Russfield AB, Homburger F, Weisburger JH. Boger E, Van
Donger CG, & Chu KC (1978) Testing of twenty-one environmental
aromatic amines for long-term toxicity or carcinogenicity. J Environ
Pathol Toxicol, 2: 325-356.
Wilson AG (1981) Field evaluation of formamidine insecticides and
Bacillus thuringiensis for selective control of Heliothis spp. on
cotton. Gen Appl Entomol, 13: 105-111.
Wiltrout RW, Ercegovich CD, & Ceglowski WS (1978) Humoral immunity in
mice following oral administration of selected pesticides. Bull
Environ Contam Toxicol, 20: 423-431.
Witkin JM & Leander JD (1982) Effects of the appetite stimulant
chlordimeform on food and water consumption of rats: Comparison with
chlordiazepoxide. J Pharmacol Exp Ther, 223: 130-134.
Witkonton S (1973) Fate of the pesticide N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine in fruit and soil, and toxicological properties
of some of its conversion products. Diss Abstr Int, 34: 3831.
Witkonton S & Ercegovich CD (1972) Degradation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine in six different
fruits. J Agric Food Chem, 20: 569-573.
Wolfenbarger DA (1974) Activity of chlordimeform HCI and chlordimeform
against arthropods attacking cotton. J Econ Etomol, 67: 445-446.
Wolfenbarger DA, Gomes ED, Robinson SH, & Parks LH (1979)
Chlordimeform residues from cotton leaves. J Econ Entomol,
72: 128-130.
Worthing CR (1979) The pesticide manual: A work compendium, 6th ed.
Croydon, United Kingdom, British Crop Protection Council, p 95.
Worthing CR & Walker SB (1983) The Pesticide manual. 7th ed. Croydon,
United Kingdom, British Crop Protection Council, p 2270.
Wu G (1982) [One case of chlordimeform poisoning.] Chin J Prev Med,
17: 275 (in Chinese with English abstract).
Wu S-Y, Liu Y-Y, Huang W-Z, Hong Q-F, Ling R-F, & Wang J-Y (1983) [On
chlordimeform poisoning - 24 case reports.] J Fujian Med Coll, 17:
42-44 (in Chinese with English abstract).
Wu D, Costa LG, & Murphy SD (1990) [The effect of pesticides-
chlordimeform and amitraz on inhibition of the binding of
3H-clonidine to alpha2-adrenoreceptor in rat forebrain tissue.]
Huaxi Yike Daxue Xuebao, 21: 17-21 (in Chinese with English abstract).
Xia S-C & Gao X-Z (1977) [Rescue after chlordimeform poisoning.]
Intermed Med, 6: 18-19 (in Chinese with English abstract).
Xie P-F (1983) [Analysis on the clinical features of chlordimeform
poisoning.] J Pract Med, 3: 145-146 (in Chinese with English
abstract).
Xu S-H (1987) [Experiences on treatment of chlordimeform poisoning.]
Hu-Nan Med, 4: 168-167 (in Chinese with English abstract).
Xue S-Z & Loosli R (1994) Formamidines. Toxicology, 91: 99-104.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J. & Wang YL (1990) The risk
assessment of occupational exposure to chlordimeform. Presented at the
23rd International Congress on Occupational Health, Montreal, Canada,
September 1990.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J, & Wang YL (1991)
[Assessment on the carcinogenic risk of chlordimeform.] Environ Chem,
10: 54-60 (in Chinese).
Yang Q-Z (1984) [Rescue of a case of cardiac arrest due to
chlordimeform poisoning.] Jiangsu Med, 10: 38 (in Chinese with English
abstract).
Yim GK, Holsapple MP, Pfister WR, & Hollingworth RM (1978)
Prostaglandin synthesis inhibited by formamidine pesticides. Life Sci.
23: 2509-2516.
Youngman RR, Leigh TF, Kerby TA, Toscano NC, & Jackson CE (1990)
Pesticides and cotton: Effect on photosynthesis, growth, and fruiting.
J Econ Entomol, 83: 1549-1557.
Zak F, Sachsse K, Bath R, & Hess R (1973) One-year feeding study on
the rat with technical C-8514 (Project No. Siss 553). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Zhang Y-J, Yuan B-Q, Jin G-L, & Liu Y-Z (1976) [Report on two severe
cases of chlordimeform poisoning and their treatment.] J Rural Doctor,
5: 19 (in Chinese with English abstract).
Zhang R-W, Lin W-Y, Wang X-G, Gu Z-Y, Qu J-Y, Xue S-Z & Wang Y-L
(1986a) [Health effects of chlordimeform on the applicators.] Chin J
Prev Med, 20: 186 (in Chinese with English abstract).
Zhang R-W, Xue S-Z, & Wang Y-L (1986b) [Measurement of total amount of
body surface contamination with regional proportional sampling.] Occup
Med, 13: 2-3 (in Chinese with English abstract).
Zhou D-J (1987) [Bitter lesson on misuse of large dose of atropine on
chlordimeform poisoning.] Hu-Nan Med, 4: 169 (in Chinese with English
abstract).
Zimmer D, Mazurek J. Petzold G. & Bhuyan BK (1980). Bacterial
mutagenicity and mammalian cell DNA damage by several substituted
anilines. Mutat Res. 77: 317-326.
R�SUM�
1. Identit�, propri�t�s physiques et chimiques et m�thodes d'analyse
Le chlordim�form est une base de force moyenne qui forme des sels
stables avec les acides forts. A l'�tat pur, le chlordim�form et son
chlorhydrate sont des solides cristallins incolores. Le point de
fusion du chlordim�form base est de 32�C, celui du chlorhydrate �tant
de 225-227�C. La base est l�g�rement soluble dans l'eau (250 mg/litre)
et facilement soluble dans les solvants organiques, tandis que le
chlorhydrate est facilement soluble dans l'eau mais plus difficilement
dans les solvants organiques. La tension de vapeur du chlordim�form
est de 48 mPa � 20�C et son coefficient de partage entre l'eau et
l'octanol (log Kow) est �gal � 2,89. On peut faire appel � de
nombreuses m�thodes d'analyse pour la recherche et le dosage du
chlordim�form dans les v�g�taux, le sol, l'eau et l'urine.
2. Sources d'exposition humaine et environnementale
Le chlordim�form n'existe pas � l'�tat naturel. On le pr�pare
industriellement par condensation du r�actif de Vilsmeier (obtenu par
r�action du dim�thylformamide sur POCl3, SOCl2 ou COCl2) soit avec
la 4-chloro- o-toluidine, soit avec la l' o-toluidine, suivie d'une
chloration du d�riv� interm�diaire obtenu. On l'utilise comme
acaricide � large spectre et il est principalement actif contre les
formes mobiles des acariens et des tiques ainsi que contre les oeufs
et les premiers stades de certains l�pidopt�res. Il agit en phase
gazeuse aussi bien que par contact. Les premiers temps de son
utilisation, on l'employait pour traiter des cultures tr�s vari�es
comme les fruits � p�pins, les drupes, les choux et autres l�gumes,
les raisins, le houblon, les agrumes, les cerises et les fraises. On
l'utilise aussi en bains contre les tiques des bovins. Ces derni�res
ann�es son usage s'est g�n�ralement limit� au coton, mais on continue
tout de m�me � l'utiliser sur le riz dans certains pays. Depuis
1988/89 il n'est plus homologu� dans la plupart des pays. En Chine, la
production a cess� en 1992, de m�me que la vente en 1993.
3. Transport, distribution et transformation dans l'environnement
Bien que sa tension de vapeur ait une valeur moyenne, le
chlordim�form ne s'�vapore pas autant qu'on le penserait des surfaces
v�g�tales. Sa stabilit� vis-�-vis de l'hydrolyse d�pend fortement du
pH; il est stable en milieu acide mais s'hydrolyse rapidement en
milieu alcalin. Le chlordim�form est capable de s'adsorber sur les
mati�res organiques dissoutes.
Dans le sol, la disparition du chlordim�form est essentiellement
imputable � l'action des microorganismes et, pour une moindre part, �
l'hydrolyse chimique. Malgr� la solubilit� du compos� dans l'eau, on
ne trouve gu�re de traces de lessivage, ce qui peut s'expliquer par
une adsorption aux mat�riaux argileux ou aux mati�res organiques du
sol ainsi que par la biod�gradation. Les principaux m�tabolites sont
la N-formyl-4-chloro- o-toluidine et la 4-chloro- o-toluidine.
Les plantes fixent le chlordim�form pr�sent dans le sol en
proportion faible mais mesurable et la concentration est suffisante
pour affecter les ravageurs qui se nourrissent � leurs d�pens. En
traitement foliaire, la p�n�tration du chlordim�form dans la cuticule
est limit�e. Le chlordim�form est rapidement d�compos� par les
v�g�taux. Les principaux m�tabolites sont le d�m�thylchlordim�form, la
N-formyl-4-chloro -o-toluidine et la 4-chloro -o-toluidine, cette
derni�re n'�tant pas produite par toutes les plantes.
Dans le sol, le chlordim�form et ses m�tabolites disparaissent
selon une cin�tique du premier ordre avec une demi-vie de 20 � 40
jours.
Les �tudes de bioaccumulation montrent que les organismes
aquatiques ne fixent qu'une petite quantit� de chlordim�form et que
celui-ci s'�limine rapidement une fois ces organismes replac�s en eau
pure.
4. Concentrations dans l'environnement et exposition humaine
On n'a pas proc�d� � des mesures de concentration dans l'air ou
l'eau. Apr�s traitement de rizi�res, on a retrouv� des r�sidus allant
jusqu'� 2900 �g/kg dans les 5 premiers centim�tres du sol et jusqu'�
150 �g/kg dans les 5 centim�tres suivants.
On a fix� des teneurs limites en r�sidus pour un grand nombre de
produits crus et dans certains cas, pour des pr�parations contenant
ces produits. Les limites maximales de r�sidus fix�es par le Codex ont
�t� supprim�es.
Il y a eu des cas d'exposition au chlordim�form au cours de la
pr�paration, de la formulation et de l'�pandage de ce produit. Depuis
quelques ann�es, on utilise la concentration urinaire totale du
chlordim�form et de ses m�tabolites pour surveiller l'exposition et il
y a d'ailleurs une bonne corr�lation entre cette concentration et le
degr� de contamination cutan�e. Dans les industries cotonni�res o�
l'on a soumis les ouvriers agricoles � une surveillance g�n�rale de la
concentration urinaire en chlordim�form, on a constat� que les plus
expos�s �taient les chargeurs, les laveurs et les m�caniciens et les
moins expos�s les signaleurs et les pilotes.
5. Cin�tique et m�tabolisme chez les animaux de laboratoire et
l'Homme
Chez les mammif�res, le chlordim�form est facilement r�sorb� au
niveau des voies digestives ainsi que par la voie transcutan�e. Il est
ensuite rapidement excr�t� � raison de 80% environ dans l'urine et de
10-15% dans les mati�res f�cales. De petites quantit�s de r�sidus sont
pr�sentes au bout de 10 jours dans tous les tissus mais rien n'indique
qu'il y ait bioaccumulation. Apr�s application cutan�e chez l'Homme,
on constate �galement une excr�tion urinaire rapide.
On retrouve dans l'urine plusieurs m�tabolites du chlordim�form
sous forme oxyd�e et conjugu�e, � savoir principalement la
N-formyl-4-chloro- o-toluidine, et la 4-chloro- o-toluidine.
In vitro, on retrouve les m�mes m�tabolites, mais avec pr�dominance
de la 4-chloro- o-toluidine.
6. Effets sur les mammif�res de laboratoire et les syst�mes d'�preuve
in vitro
Les �preuves pratiqu�es sur un certain nombre d'esp�ces montrent
que la chlordim�form pr�sente une toxicit� aigu� mod�r�e par la voie
orale et la voie transcutan�e. Chez le rat, les principaux m�tabolites
sont peu toxiques par voie orale. Chez le lapin, le chlordim�form ne
provoque qu'une l�g�re irritation oculaire et cutan�e. Apr�s
exposition de courte ou de br�ve dur�e au chlordim�form ou � ses
m�tabolites, on peut observer, au niveau des constantes
h�matologiques, des modifications qui sont imputables au traitement et
on constate, � dose �lev�e, certains signes qui d�notent une
hyperplasie de l'�pith�lium des canaux biliaires et de la vessie. Il
n'y pas d'accroissement de la fr�quence des tumeurs chez le rat. Chez
la souris, on observe, apr�s administration par voie alimentaire de
chlordim�form, de N-formyl-4-chloro -o-toluidine ou de
4-chloro- o-toluidine, une augmentation, li�e � la dose, des tumeurs
malignes h�morragiques d'origine vasculaire appartenant � la classe
des h�mangio-endoth�liomes, dont la pr�sence entra�ne un accroissement
de la mortalit� parall�le � la dose.
Le chlordim�form n'a pas d'effet ind�sirable sur les diff�rents
aspects de la fonction de reproduction et il n'a aucun pouvoir
t�ratog�ne.
Le chlordim�form a fait l'objet d'un grand nombre d'�preuves de
g�notoxicit� in vitro et in vivo. Aucune d'elles n'a donn� de
r�sultat positif, �tant entendu qu'il s'agissait de la mati�re active
et non de formulations. Par ailleurs, un certain nombre d'observations
sporadiques non confirm�es font �tat d'une activit� mutag�ne induite
par la N-formyl-4-chloro- o-toluidine et par la 4-chloro- o-
toluidine. Il n'existe qu'une seule description de transformations
cellulaires provoqu�es par le chlordim�form et par la 4-chloro -o-
toluidine. Chez des souris et des rats trait�s par le chlordim�form,
on a constat� une que le compos� se liait � l'ADN des cellules
h�patiques. A dose beaucoup plus �lev�e, il se forme chez les m�mes
animaux un important adduit hydrophobe.
Le chlordim�form provoque des effets pharmacologiques et
biochimiques divers chez l'animal, et notamment des effets
cardiovasculaires, une hypothermie, une hyperexcitabilit�, une
modification des fonctions visuelle et auditive ainsi que la
modulation des amines biogen�se et des enzymes pharmacom�tabolisantes.
7. Effets sur l'Homme
Les intoxications aigu�s se traduisent par une fatigue, des
naus�es et une perte d'app�tit, avec, dans les cas graves, somnolence,
cyanose, besoin imp�rieux d'uriner, cystite, effets cardiovasculaires
(tachy-cardie, bradycardie, anomalies de l'ECG), coma et �tat de choc.
En g�n�ral, la r�cup�ration est totale.
Apr�s une exposition de longue dur�e au chlordim�form, on peut
observer encore d'autres sympt�mes tels que des douleurs abdominales,
des d�mangeaisons et des �ruptions (en cas d'exposition cutan�e)
accompagn�s d'une h�maturie macroscopique ou micro-scopique. On a
signal� de nombreux cas d'intoxication pr�sentant des sympt�mes
d'exposition de longue dur�e parmi les ouvriers d'unit�s de production
de chlordim�form et des ouvriers agricoles.
Les donn�es �pid�miologiques obtenues � la suite de cas
d'exposition professionnelle montrent qu'il existe une forte
corr�lation entre l'exposition � la 4-chloro -o-toluidine et le
cancer de la vessie. En revanche, on n'a gu�re obtenu d'�l�ments qui
militeraient en faveur d'une association entre ce type de cancer et
l'exposition au chlordim�form.
8. Effets sur les autres �tres vivants au laboratoire et dans leur
milieu naturel
Apr�s �pandage de chlordim�form sur le sol, on n'a pas observ�
d'effets sensibles sur les populations de champignons, de bact�ries ou
d'actinomyc�tes terricoles.
Il n'existe pas de donn�es toxicologiques de laboratoire
concernant les invert�br�s dul�aquicoles. En pr�sence de
chlordim�form, il y a inhibition de la croissance des larves
d'hu�tres, avec une CE50 de 5,7 mg/litre. Pour la crevette rose,
le seul crustac� �tudi�, la CL50 � 96 h a �t� trouv�e �gale �
7,1 mg/litre et des valeurs allant de 1 � 54 mg/litre ont �t� obtenues
pour le m�me param�tre chez les poissons. On ne poss�de aucune donn�e
relative � la toxicit� chronique pour les esp�ces aquatiques.
L'ensemble des r�sultats de laboratoire et des donn�es recueillies sur
le terrain indique que le compos� est toxique pour de nombreux
arthropodes terrestres non vis�s.
Chez l'abeille, la toxicit� de contact se traduit par une DL50
de 120 �g/g, la toxicit� par voie orale correspondant � une valeur de
187 �g/g. Trois heures apr�s l'�pandage de chlordim�form sur de la
luzerne, l'exposition de certaines esp�ces d'abeilles aux r�sidus
encore pr�sents sur les plantes, n'a provoqu� aucune mortalit�.
La CL50 par voie alimentaire varie de >1000 � > 5000/kg de
nourriture pour diverses esp�ces d'oiseaux.
9. Evaluation des risques pour la sant� humaine et des effets sur
l'environnement
On a observ� des signes d'intoxication aigu� chez des
travailleurs qui, peut-�tre par suite de l'inobservation des mesures
de s�curit�, avaient �t� fortement expos�s � du chlordim�form au cours
de la pr�paration ou de l'utilisation de ce produit. Comme, � ce qu'il
semble, il n'est plus produit ni utilis� nulle part dans le monde, il
ne devrait plus y avoir de cas d'intoxication aigu�. Le risque li� �
une exposition chronique et en particulier, le risque de cancer de la
vessie, subsistera cependant pendant de nombreuses ann�es. Il faut
continuer � effectuer des contr�les sanitaires chez les sujets qui ont
subi une exposition notable pour avoir travaill� dans des ateliers de
production de chlordim�form ou avoir v�cu dans des zones rurales o� le
produit �tait largement utilis�.
Comme il s'agit d'un produit qui n'est plus en usage, on n'a pas
proc�d� � une �valuation quantitative du risque qu'il repr�sente pour
l'environnement. On ne pense pas que celui-ci puisse subir des effets
nocifs � long terme qui soient attribuables � l'utilisation ant�rieure
du produit.
10. Conclusions et recommandations
Il existe un risque non n�gligeable que le chlordim�form produise
des effets toxiques imm�diats ou � plus long terme chez les individus
expos�s. Les donn�es disponibles accr�ditent l'id�e d'une association
entre l'augmentation de l'incidence du cancer de la vessie chez
l'homme et l'exposition � la 4-chloro- o-toluidine et, dans une
moindre mesure, au chlordim�form.
Le chlordim�form ne persiste pas dans l'environnement et il ne
devrait donc pas y avoir d'effets nocifs � long terme sur celui-ci qui
r�sulteraient de l'usage ant�rieur du compos�.
Il n'est pas recommand� de reprendre la production ou l'usage du
chlordim�form dans un but commercial. Les stocks existants doivent
�tre �limin�s selon les r�gles de s�curit�.
Les personnes expos�es au chlordim�form de par leur profession
doivent �tre soumises � des examens cytologiques v�sicaux et � une
recherche syst�matique de l'h�maturie dans le cadre d'un programme
g�n�ral de d�pistage.
RESUMEN
1. Identidad, propiedades f�sicas y qu�micas y m�todos anal�ticos
El clordimeformo es una base de fuerza media que forma sales
estables con �cidos fuertes. Tanto el clordimeformo como su sal
hidroclorada en estado puro son s�lidos cristalinos incoloros. El
punto de fusi�n del clordimeformo (base) es de 32�C, mientras que el
de la sal hidroclorada es de 225-227�C. El clordimeformo (base) es
poco soluble en agua (250 mg/litro) y f�cilmente soluble en
disolventes org�nicos, mientras que la sal hidroclorada es f�cilmente
soluble en agua pero menos soluble en disolventes org�nicos. El
clordimeformo (base) tiene una presi�n de vapor de 48 mPa a 20�C y un
log Kow de 2,89. Se dispone de una amplia gama de m�todos anal�ticos
para detectar y cuantificar la presencia de clordimeformo en las
plantas, el suelo, el agua y la orina.
2. Fuentes de exposici�n humana y ambiental
El clordimeformo no existe en la naturaleza. Se produce
comercialmente mediante condensaci�n del reactivo de Vilsmeier
(obtenido por reacci�n de la dimetilformamida con POCl3, SOCl2
o COCl2) con 4-cloro- o-toluidina o bien con o-toluidina y
cloraci�n ulterior del producto intermedio resultante. Se ha utilizado
como acaricida de amplio espectro y act�a principalmente contra las
formas m�viles de �caros y garrapatas, as� como contra los huevos y
las cris�lidas en estado inicial de algunos insectos del orden
Lepid�ptera. Es activo en la fase de vapor, as� como por contacto.
Cuando comenz� a utilizarse, se aplicaba a productos de una amplia
variedad de cultivos, tales como frutas de pipas, frutas de hueso,
berzas, hortalizas, uvas, l�pulo, c�tricos, manzanas, peras, cerezas y
fresas. Tambi�n se utilizaba en ba�os antiparasitarios para combatir
las garrapatas del ganado. En los �ltimos a�os, su uso se limitaba por
lo general al algod�n, aunque en algunos pa�ses se segu�a aplicando al
arroz. En la mayor�a de los pa�ses, su registro se abandon�
voluntariamente en 1988/1989. En China dej� de producirse en 1992 y de
venderse en 1993.
3. Transporte, distribuci�n y transformaci�n en el medio ambiente
El clordimeformo tiene una presi�n de vapor moderada pero su
evaporaci�n de la superficie de las plantas es inferior a la que
cabr�a prever. La estabilidad hidrol�tica del clordimeformo depende
mucho del pH; es estable en condiciones �cidas pero se hidroliza
r�pidamente en condiciones alcalinas. El clordimeformo tiene un
potencial de adsorci�n a la materia org�nica disuelta.
Hay dispersi�n del clordimeformo en el suelo, principalmente por
acci�n microbiana y, en menor medida, por hidr�lisis qu�mica. Pese a
la solubilidad del clordimeformo en agua, hay pocos indicios de
lixiviaci�n, lo que puede deberse a su adsorci�n a minerales
arcillosos y a la materia org�nica del suelo, as� como a su
biodegradaci�n. Los principales metabolitos son la N-formil-
4-cloro- o-toluidina y la 4-cloro- o-toluidina.
La absorci�n del clordimeformo por las plantas a partir del suelo
es escasa pero detectable, y suficiente para afectar a las plagas que
se alimentan de ellas. El clordimeformo aplicado a las hojas s�lo
tiene una capacidad limitada de penetrar en las capas cuticulares. El
clordimeformo se degrada r�pidamente en las plantas. Sus principales
metabolitos son el demetilclordimeformo, la N-formil-4-cloro-
o-toluidina y la 4-cloro- o-toluidina, aunque no todas las plantas
estudiadas produjeron 4-cloro- o-toluidina.
El clordimeformo y sus metabolitos se dispersan en el suelo
conforme a una cin�tica de primer orden, con una semivida de 20-40
d�as.
Los estudios sobre bioacumulaci�n indican una escasa absorci�n
del clordimeformo por los organismos acu�ticos y una r�pida depuraci�n
de �stos despu�s de haber sido transferidos a un agua limpia.
4. Niveles medioambientales y exposici�n humana
No se han medido los niveles de clordimeformo en el aire ni en el
agua. Tras la aplicaci�n de clordimeformo a unos arrozales, en el
suelo se hallaron residuos en concentraciones de hasta 2900 �g/kg en
los 5 cm primeros de profundidad, y de 150 �g/kg en los 5 cm
siguientes.
Se establecieron niveles m�ximos de residuos aplicables a una
amplia variedad de productos sin elaborar y, en algunos casos, de
residuos trasladados a los alimentos elaborados. Los l�mites m�ximos
aplicables a los residuos de clordimeformo se han retirado del Codex
Alimentarius.
Hab�a exposici�n ocupacional al clordimeformo durante la
fabricaci�n, la formulaci�n y la aplicaci�n del producto. En los
�ltimos a�os la exposici�n se ha vigilado mediante la determinaci�n de
los niveles totales de clordimeformo y de sus metabolitos presentes en
la orina, y hay una correlaci�n positiva entre el nivel en la orina y
el grado de contaminaci�n cut�nea. Entre los trabajadores agr�colas de
los algodonales sometidos a una amplia vigilancia de la excreci�n
urinaria de clordimeformo, los niveles m�s altos de exposici�n se
hallaban en los cargadores, lavadores y mec�nicos, y los niveles m�s
bajos en los obreros se�alizadores y pilotos.
5. Cin�tica y metabolismo en animales de laboratorio y en el ser
humano
Los mam�feros absorben f�cilmente el clordimeformo por el tracto
gastrointestinal y a trav�s de la piel. Lo excretan r�pidamente,
alrededor del 80% por la orina y del 10-15% por las heces. Al cabo de
unos 10 d�as se observan niveles bajos de residuos en todos los
tejidos y no hay indicios de bioacumulaci�n. Tras la administraci�n
cut�nea a seres humanos, se observa una excreci�n r�pida semejante por
la orina.
Varios metabolitos oxidados y conjugados del clordimeformo se
excretan por la orina; los principales son el demetilclordimeformo, la
N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. En
estudios in vitro se han observado los mismos metabolitos, siendo el
principal la 4-cloro- o-toluidina.
6. Efectos en mam�feros de laboratorio y en sistemas de pruebas
in vitro
En ensayos realizados en varias especies, el clordimeformo
administrado por v�a oral y cut�nea ha mostrado tener una toxicidad
aguda moderada. Los principales metabolitos han mostrado tener una
toxicidad oral baja en ensayos realizados en ratas. El clordimeformo
provoca solamente una ligera irritaci�n cut�nea y ocular en el conejo.
Tras una exposici�n breve o prolongada de ratones y ratas al
clordimeformo o a sus metabolitos pueden observarse cambios asociados
al tratamiento en los par�metros hematol�gicos y, con dosis elevadas,
indicios de hiperplasia del epitelio de las v�as biliares y de la
vejiga. El clordimeformo no aumenta la incidencia de tumores en las
ratas. En los ratones, despu�s de administrar a trav�s de la dieta
clordimeformo N-formil-4-cloro- o-toluidina o 4-cloro- o-
toluidina, se observa, de forma relacionada con la dosis, un aumento
de los tumores malignos hemorr�gicos de origen vascular clasificados
como hemangioendoteliomas malignos, que producen un aumento de la
mortalidad asociado con la dosis.
El clordimeformo no afecta a los par�metros reproductivos ni
tiene potencial teratog�nico.
Se ha ensayado el clordimeformo en una amplia variedad de pruebas
de genotoxicidad in vitro e in vivo. No se han comunicado
reacciones positivas a ninguna de esas pruebas, en las que se ensay�
clordimeformo en estado puro. Adem�s, se han comunicado varios
informes espor�dicos y no confirmados de actividad mutag�nica inducida
por la N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. Un
informe describe una inducci�n de la transformaci�n celular por efecto
tanto del clordimeformo como de la 4-cloro- o-toluidina. En el h�gado
de los ratones y las ratas expuestos se producen enlaces con el ADN.
Se ha observado un importante aducto hidrof�bico, en los ratones en
niveles mucho mayores que en las ratas.
El clordimeformo induce diversos efectos farmacol�gicos y
bioqu�micos en los animales, tales como cambios cardiovasculares,
hipotermia, hiperexcitabilidad, efectos sobre las funciones visual
central y auditiva y modulaci�n de las aminas biog�nicas y de las
enzimas que metabolizan f�rmacos.
7. Efectos en el ser humano
La intoxicaci�n aguda causa fatiga, n�useas, p�rdida del apetito
y, en casos m�s graves, somnolencia, cianosis, micci�n imperiosa,
cistitis, efectos cardiovasculares (taquicardia, bradicardia,
alteraciones del ECG), coma y choque. En general se produce una
recuperaci�n completa de la intoxicaci�n aguda.
Otros s�ntomas asociados a la exposici�n cr�nica al
clordime-formo son dolores abdominales, prurito y exantemas
(exposici�n cut�nea), as� como hematuria macrosc�pica y microsc�pica.
Se ha comunicado un gran n�mero de casos con s�ntomas cl�nicos de
exposici�n cr�nica tanto entre los obreros de las plantas de
producci�n de clordimeformo como entre los trabajadores agr�colas.
Los indicios epidemiol�gicos relacionados con la exposici�n
ocupacional muestran una fuerte asociaci�n entre la exposici�n al
metabolito 4-cloro- o-toluidina y la incidencia de c�ncer de vejiga
en el ser humano. Actualmente se dispone de pocos indicios de
asocia-ci�n entre la exposici�n al clordimeformo y el c�ncer de vejiga
en el ser humano.
8. Efectos en otros organismos en el laboratorio y en el medio
ambiente
No se observaron efectos significativos en poblaciones de hongos
de la tierra, bacterias o actinomicetos tras la aplicaci�n de
clordime-formo al suelo.
No existen datos de laboratorio sobre la toxicidad en los
invertebrados de agua dulce. El clordimeformo inhibi� el crecimiento
de larvas de ostras, con una CE50 de 5,7 mg/litro. La CL50 a las
96-h para los camarones rosados, �nico crust�ceo estudiado, fue de
7,1 mg/litro y los valores de la CL50 a las 96-h para los peces
oscilaron entre 1 y 54 mg/litro. No se dispone de datos sobre
toxicidad acu�tica cr�nica. La combinaci�n de datos obtenidos en el
laboratorio y sobre el terreno revela que el clordimeformo es t�xico
para una amplia gama de artr�podos terrestres no combatidos.
Con respecto a las abejas, se ha comunicado una DL50 de
toxicidad por contacto de 120 �g/g y una DL50 de toxicidad oral de
187 �g/g. No se produjo mortalidad sobre el terreno tras la exposici�n
de especies de abejas a los residuos presentes en la alfalfa tres
horas despu�s del rociado.
La CL50 en la dieta de varias especies de p�jaros oscil� entre
>1000 y >5000 mg/kg de dieta.
9. Evaluaci�n de los riesgos para la salud humana y efectos en el
medio ambiente
La exposici�n intensa durante la producci�n o la utilizaci�n,
debida posiblemente a la insuficiencia de las medidas de seguridad,
dio lugar a s�ntomas de intoxicaci�n aguda en los trabajadores. Como
se ha notificado que se ha suspendido la producci�n y la utilizaci�n
de clordimeformo en todo el mundo, no deber�an producirse nuevos casos
de intoxicaci�n aguda. Sin embargo, el riesgo asociado a la exposici�n
cr�nica, en particular el riesgo de c�ncer de vejiga, seguir� siendo
preocupante durante muchos a�os. Deber�a proseguir el reconoci-miento
m�dico de las personas que han estado muy expuestas en las plantas de
producci�n y en las comunidades rurales donde se haya aplicado
extensamente el clordimeformo.
Dado que el clordimeformo ha dejado de utilizarse, no se ha
realizado ninguna evaluaci�n cuantitativa de los riesgos para el medio
ambiente. A largo plazo no se prev�n efectos perjudiciales para el
medio ambiente como consecuencia de la utilizaci�n de clordime-formo
en el pasado.
10. Conclusiones y recomendaciones
El clordimeformo tiene un potencial significativo para causar
tanto toxicidad inmediata como a largo plazo en las personas
expuestas. La informaci�n de que se dispone actualmente apunta a una
asociaci�n entre una mayor incidencia de c�ncer de vejiga en el ser
humano y la exposici�n a la 4-cloro- o-toluidina y, en menor medida,
al clordimeformo.
El clordimeformo no persiste en el medio ambiente, por lo que a
largo plazo no se prev�n efectos perjudiciales como consecuencia de su
utilizaci�n en el pasado.
Se recomienda que el clordimeformo no se produzca comercialmente
ni se utilice en el futuro. Las reservas existentes deber�an
eliminarse sin correr riesgos.
Las personas expuestas profesionalmente al clordimeformo deber�an
participar en un programa de reconocimiento m�dico que comprenda
citolog�a urinaria y detecci�n de hematuria.
 It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
Members
Dr P.J. Abbott, Australia and New Zealand Food Authority
(ANZFA), Canberra, Australia
Dr K. Barab�s, Department of Public Health, Albert Szent-Gyorgyi,
University Medical School, Szeged, Hungary
Dr A.L. Black, Woden, ACT, Australia
Professor J.F. Borzelleca, Pharmacology and Toxicology,
Richmond, Virginia, USA
Dr P.J. Campbell, Pesticides Safety Directorate, Ministry of
Agriculture, Fisheries and Food, Kings Pool, York,
United Kingdom
Professor L.G. Costa, Department of Environmental Health,
University of Washington, Seattle, USA
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood,
Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Dr I. Dewhurst, Mammalian Toxicology Branch, Pesticides Safety
Directorate, Ministry of Agriculture, Fisheries and Food,
Kings Pool, York, United Kingdom
Dr V. Drevenkar, Institute for Medical Research and Occupational
Health, Zagreb, Croatia
Dr W. Erickson, Environmental Fate and Effects Division,
US Environmental Protection Agency, Washington, D.C., USA
Dr A. Finizio, Group of Ecotoxicology, Institute of Agricultural
Entomology, University of Milan, Milan, Italy
Mr K. Garvey, Office of Pesticide Programs (7501C),
US Environmental Protection Agency, Washington, D.C., USA
Dr A.B. Kocialski, Health Effects Division, Office of Pesticide
Programs, US Environmental Protection Agency,
Washington, D.C., USA
Dr A. Moretto, Institute of Occupational Medicine, University of
Padua, Padua, Italy
Professor O. Pelkonen, Department of Pharmacology and
Toxicology, University of Oulu, Oulu, Finland
Dr D. Ray, Medical Research Council Toxicology Unit, University
of Leicester, Leicester, United Kingdom
Dr J.H.M. Temmink, Department of Toxicology, Wageningen
Agricultural University, Wageningen, The Netherlands
Observers
Dr J.W. Adcock, AgrEvo UK Limited, Chesterford Park, Saffron,
Waldon, Essex, United Kingdom
Mr D. Arnold, Environmental Sciences, AgrEvo UK Ltd.,
Chesterford Park, Saffron Waldon, Essex, United Kingdom
Dr E. Bellet, CCII, Overland Park, Kansas, USA
Mr Jan Chart, AMVAC Chemical Corporation, Newport Beach,
California, USA
Dr H. Egli, Novartis Crop Protection AG, Basel, Switzerland
Dr P. Harvey, AgrEvo UK Ltd., Chesterford Park, Saffron Walden,
Essex, United Kingdom
Dr G. Krinke, Novartis Crop Protection AG, Basel, Switzerland
Dr A. McReath, DowElanco Limited, Letcombe Regis, Wantage,
Oxford, United Kingdom
Dr H. Scheffler, Novartis Crop Protection AG, Basel, Switzerland
Dr A.E. Smith, Novartis Crop Protection AG, Basel, Switzerland
Secretariat
Dr L. Harrison, Health and Safety Executive, Bootle, Merseyside,
United Kingdom
Dr J.L. Herrman, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr P.G. Jenkins, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr D. McGregor, Unit of Carcinogen Identification and Evaluation,
International Agency for Research on Cancer, Lyon, France
Dr R. Plestina, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr E. Smith, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
Dr P. Toft, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
IPCS TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
The Core Assessment Group (CAG) of the Joint Meeting on
Pesticides (JMP) met at the Institute for Environment and Health,
Leicester, United Kingdom, from 3 to 8 March 1997. Dr L.L. Smith
welcomed the participants on behalf of the Institute, and
Dr R. Plestina on behalf of the three IPCS cooperating organizations
(UNEP/ILO/WHO). The CAG reviewed and revised the draft monograph and
made an evaluation of the risks for human health and the environment
from exposure to chlordimeform.
The first draft of the monograph was prepared by Dr P. Abbott,
Canberra, Australia. Extensive scientific comments were received
following circulation of the first draft to the IPCS contact points
for Environmental Health Criteria monographs and these comments were
incorporated into the second draft by the Secretariat.
Dr R. Plestina and Dr P.G. Jenkins, both members of the IPCS
Central Unit, were responsible for the overall scientific content and
technical editing, respectively. The efforts of all who helped in the
preparation and finalization of the monograph are gratefully
acknowledged.
ABBREVIATIONS
ACTH adrenocorticotropic hormone
ADI acceptable daily intake
a.i. active ingredient
BSP bromosulfophthalein
CIMS chemical ionization mass spectrometry
CNS central nervous system
CORT corticosteroid
DNA deoxyribonucleic acid
EC emulsifiable concentrate
ECG electrocardiography
GC gas chromatography
HPLC high performance liquid chromatography
IgM immunoglobulin M
JMPR Joint FAO/WHO Meeting on Pesticide Residues
MRL maximum residue limit
Mu Chinese measure of an area equivalent to 1/15 acre
or 1/60 ha or 166 m2
MS mass spectroscopy
NADPH reduced nicotinamide adenine dinucleotide
NC cell activity natural cytotoxic cell activity
NK cell activity natural killer cell activity
NOEL no-observable-effect level
PL prolactin
SAP serum alkaline phosphatase
SGOT serum glutamate-oxalate transaminase
SGPT serum glutamate-pyruvate transaminase
SIR standard incidence rate
SMR standardized mortality ratio
SPF specific pathogen free
TLC thin layer chromatography
TLm median tolerance limit
UV ultraviolet
1. SUMMARY
1.1 Identity, physical and chemical properties, and analytical
methods
Chlordimeform is a base of medium strength and forms stable salts
with strong acids. Both chlordimeform and its hydrochloride salt in
the pure state are colourless crystalline solids. Chlordimeform base
has a melting point of 32�C, while the hydrochloride salt has a
melting point of 225-227�C. Chlordimeform base is sparingly soluble in
water (250 mg/litre) and readily soluble in organic solvents, whereas
the hydrochloride salt is readily soluble in water but less soluble in
organic solvents. Chlordimeform base has a vapour pressure at 20�C of
48 mPa and a log Kow of 2.89. A wide range of analytical methods are
available for detection and quantification of chlordimeform in plants,
soil, water and urine.
1.2 Sources of human and environmental exposure
Chlordimeform does not occur naturally. It is manufactured
commercially by condensation of the Vilsmeier reagent (obtained by
reaction of dimethylformamide with POCl3, SOCl2 or COCl2) either
with 4-chloro- o-toluidine or with o-toluidine and subsequent
chlorination of the resulting intermediate . It has been used as a
broad spectrum acaricide and is active mainly against motile forms of
mites and ticks and against eggs and early instars of some
Lepidoptera insects. It is active in the vapour phase as well as by
contact. In the early period of its use, it was used on a wide variety
of crops such as pome fruits, stone fruits, cole crops, vegetables,
grapes, hops, citrus fruits, apples, pears, cherries and strawberries.
It was also used in cattle dips for the control of cattle ticks. In
the latter years, its use was generally restricted to cotton, although
in some countries, there was continued use on rice. Its registration
was voluntarily withdrawn in 1988/1989 in most countries. In China,
production stopped in 1992 and sales ceased in 1993.
1.3 Environmental transport, distribution and transformation
Chlordimeform has a moderate vapour pressure but its evaporation
from plant surfaces is less than would be expected. The hydrolytic
stability of chlordimeform is strongly pH-dependent; it is stable in
acid conditions but rapidly hydrolysed in alkaline conditions.
Chlordimeform has the potential to adsorb to dissolved organic matter.
In soils, chlordimeform is primarily dissipated by microbial
action with some contribution by chemical hydrolysis. There is little
evidence of leaching despite its water solubility, which may be
due to its adsorption to clay minerals, soil organic matter and
biodegradation. The principal metabolites are N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine.
There is a low but measurable uptake of chlordimeform into plants
from soil, sufficient to affect plant-feeding pests. When applied to
the leaves, chlordimeform has only limited capacity to penetrate the
cuticular layers. Chlordimeform is degraded rapidly in plants. The
principal metabolites are demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine, though not all plants
studied produced the 4-chloro- o-toluidine.
In soils, chlordimeform and its metabolites are dissipated
according to first-order kinetics with a half-life of 20-40 days.
Bioaccumulation studies have demonstrated low uptake of
chlordimeform by aquatic organisms and rapid depuration on transfer to
clean water.
1.4 Environmental levels and human exposure
Levels have not been measured in air and water. Following
applications to paddy fields residues of up to 2900 �g/kg in the top
5 cm of soil and 150 �g/kg in the next 5 cm have been found.
Maximum residue levels were set for a wide range of raw produce
and, in some cases, the residues carried over into processed food. The
Codex maximum residue limits for chlordimeform have been withdrawn.
Occupational exposure to chlordimeform has taken place during
manufacture, formulation and application. In recent years, total
urinary levels of chlordimeform and its metabolites have been used as
a monitor for exposure, and the urine level correlates well with the
degree of skin contamination. Where agricultural workers in the cotton
industry have undergone extensively surveillance for urinary excretion
of chlordimeform, the highest exposure levels were in loaders, washers
and mechanics, with lower levels in flagmen and pilots.
1.5 Kinetics and metabolism in laboratory animals and humans
Chlordimeform is readily absorbed from the gastrointestinal tract
and through the skin of mammals. Rapid excretion follows, with
approximately 80% in the urine and 10-15% in faeces. Low residue
levels are evident in all tissues after approximately 10 days, and
there is no evidence of bioaccumulation. Following dermal
administration in humans, similar rapid excretion through the urine is
observed.
Several oxidized and conjugated metabolites of chlordimeform are
excreted in the urine, demethylchlordimeform, N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine being the major metabolites.
In in vitro studies, the same metabolites are formed,
4-chloro- o-toluidine being the major metabolite.
1.6 Effects on laboratory mammals and in vitro test systems
Chlordimeform has moderate acute toxicity when tested in several
species by oral and dermal routes of administration. The major
metabolites have low oral toxicity when tested in rats. Chlordimeform
causes only slight skin and eye irritation in rabbits. Following
either short- or long-term exposure in both mice and rats with either
chlordimeform or its metabolites, treatment-related changes can be
observed in haematological parameters, and there is some evidence of
hyperplasia of the epithelium of the bile duct and urinary bladder at
the high dose levels. Chlordimeform does not cause an increase in
tumour incidence in rats. In mice, following dietary administration
of either chlordimeform, N-formyl-4-chloro- o-toluidine or
4-chloro- o-toluidine, there is a dose-related increase in
haemorrhagic malignant tumours of vascular origin classified as
malignant haemangioendotheliomas, which cause a dose-related increase
in mortality.
Chlordimeform does not affect reproductive parameters, nor does
it have any teratogenic potential.
Chlordimeform has been tested in a broad range of in vitro and
in vivo genotoxicity assays. No positive responses have been
reported with any of these tests in which unformulated chlordimeform
was tested. In addition, there have been several sporadic and
unconfirmed reports of mutagenic activity induced by N-formyl-
4-chloro- o-toluidine and 4-chloro- o-toluidine. A single report
describes cell transformation induction by both chlordimeform and
4-chloro- o-toluidine. Binding to DNA occurs in the liver of dosed
mice and rats. One major hydrophobic adduct is found at a much higher
level in mice than in rats.
Chlordimeform induces a variety of pharmacological and
biochemical effects in animals, including cardiovascular changes,
hypothermia, hyperexcitability, effects on central visual and auditory
functions, and modulation of biogenic amines and drug-metabolizing
enzymes.
1.7 Effects on humans
Acute poisoning causes fatigue, nausea and loss of appetite, and,
in more severe cases, somnolence, cyanosis, urgency in urination,
cystitis, cardiovascular effects (tachycardia, bradycardia, ECG
changes), coma and shock. Generally, there is complete recovery from
acute intoxication.
Following chronic exposure to chlordimeform, additional symptoms
include abdominal pain, skin itching and rashes (dermal exposure), and
gross and microscopic haematuria. A large number of cases with
clinical symptoms of chronic exposure have been reported in both
chlordimeform-manufacturing plants as well as in agricultural workers.
Following occupational exposure, epidemiological evidence has
provided a strong association between exposure to the metabolite
4-chloro- o-toluidine and the incidence of human urinary bladder
cancer. There is currently only weak evidence for an association
between exposure to chlordimeform and human bladder cancer.
1.8 Effects on other organisms in the laboratory and field
There were no significant effects on populations of soil fungi,
bacteria or actinomycetes following application of chlordimeform to
soil.
There are no laboratory toxicity data on freshwater
invertebrates. Growth of larval oysters was inhibited by chlordimeform
with an EC50 of 5.7 mg/litre. The 96-h LC50 for pink shrimp, the only
crustacean studied, was 7.1 mg/litre and the 96-h LC50 values for
fish ranged from 1 to 54 mg/litre. There are no chronic aquatic
toxicity data available. A mixture of laboratory and field data shows
that chlordimeform is toxic to a wide range of terrestrial non-target
arthropods.
The contact toxicity LD50 for bees has been reported to be
120 �g/g and that for oral toxicity 187 �g/g. There was no mortality
in the field following exposure of species of bees to residues on
alfalfa 3 h after spraying.
The dietary LC50 for various birds species ranged from >1000 to
>5000 mg/kg diet.
1.9 Evaluation of human health risks and effects on the environment
Heavy exposure during manufacture or use, possibly resulting from
inadequate safety precautions, has led to signs of acute poisoning in
workers. Since both production and use are reported to have ceased
worldwide, acute poisoning should no longer occur. The risk associated
with chronic exposure, however, particularly the risk of bladder
cancer, will continue to be of concern for many years. Health
screening of significantly exposed individuals from manufacturing
plants from those rural communities where chlordimeform was
extensively used should be continued.
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
1.10 Conclusions and recommendations
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine and, to a lesser
extent, chlordimeform.
Chlordimeform does not persist in the environment, and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Identity
Common name: Chlordimeform
Chemical structure:
It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
Members
Dr P.J. Abbott, Australia and New Zealand Food Authority
(ANZFA), Canberra, Australia
Dr K. Barab�s, Department of Public Health, Albert Szent-Gyorgyi,
University Medical School, Szeged, Hungary
Dr A.L. Black, Woden, ACT, Australia
Professor J.F. Borzelleca, Pharmacology and Toxicology,
Richmond, Virginia, USA
Dr P.J. Campbell, Pesticides Safety Directorate, Ministry of
Agriculture, Fisheries and Food, Kings Pool, York,
United Kingdom
Professor L.G. Costa, Department of Environmental Health,
University of Washington, Seattle, USA
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood,
Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Dr I. Dewhurst, Mammalian Toxicology Branch, Pesticides Safety
Directorate, Ministry of Agriculture, Fisheries and Food,
Kings Pool, York, United Kingdom
Dr V. Drevenkar, Institute for Medical Research and Occupational
Health, Zagreb, Croatia
Dr W. Erickson, Environmental Fate and Effects Division,
US Environmental Protection Agency, Washington, D.C., USA
Dr A. Finizio, Group of Ecotoxicology, Institute of Agricultural
Entomology, University of Milan, Milan, Italy
Mr K. Garvey, Office of Pesticide Programs (7501C),
US Environmental Protection Agency, Washington, D.C., USA
Dr A.B. Kocialski, Health Effects Division, Office of Pesticide
Programs, US Environmental Protection Agency,
Washington, D.C., USA
Dr A. Moretto, Institute of Occupational Medicine, University of
Padua, Padua, Italy
Professor O. Pelkonen, Department of Pharmacology and
Toxicology, University of Oulu, Oulu, Finland
Dr D. Ray, Medical Research Council Toxicology Unit, University
of Leicester, Leicester, United Kingdom
Dr J.H.M. Temmink, Department of Toxicology, Wageningen
Agricultural University, Wageningen, The Netherlands
Observers
Dr J.W. Adcock, AgrEvo UK Limited, Chesterford Park, Saffron,
Waldon, Essex, United Kingdom
Mr D. Arnold, Environmental Sciences, AgrEvo UK Ltd.,
Chesterford Park, Saffron Waldon, Essex, United Kingdom
Dr E. Bellet, CCII, Overland Park, Kansas, USA
Mr Jan Chart, AMVAC Chemical Corporation, Newport Beach,
California, USA
Dr H. Egli, Novartis Crop Protection AG, Basel, Switzerland
Dr P. Harvey, AgrEvo UK Ltd., Chesterford Park, Saffron Walden,
Essex, United Kingdom
Dr G. Krinke, Novartis Crop Protection AG, Basel, Switzerland
Dr A. McReath, DowElanco Limited, Letcombe Regis, Wantage,
Oxford, United Kingdom
Dr H. Scheffler, Novartis Crop Protection AG, Basel, Switzerland
Dr A.E. Smith, Novartis Crop Protection AG, Basel, Switzerland
Secretariat
Dr L. Harrison, Health and Safety Executive, Bootle, Merseyside,
United Kingdom
Dr J.L. Herrman, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr P.G. Jenkins, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr D. McGregor, Unit of Carcinogen Identification and Evaluation,
International Agency for Research on Cancer, Lyon, France
Dr R. Plestina, International Programme on Chemical Safety,
World Health Organization, Geneva, Switzerland
Dr E. Smith, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
Dr P. Toft, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland
IPCS TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR CHLORDIMEFORM
The Core Assessment Group (CAG) of the Joint Meeting on
Pesticides (JMP) met at the Institute for Environment and Health,
Leicester, United Kingdom, from 3 to 8 March 1997. Dr L.L. Smith
welcomed the participants on behalf of the Institute, and
Dr R. Plestina on behalf of the three IPCS cooperating organizations
(UNEP/ILO/WHO). The CAG reviewed and revised the draft monograph and
made an evaluation of the risks for human health and the environment
from exposure to chlordimeform.
The first draft of the monograph was prepared by Dr P. Abbott,
Canberra, Australia. Extensive scientific comments were received
following circulation of the first draft to the IPCS contact points
for Environmental Health Criteria monographs and these comments were
incorporated into the second draft by the Secretariat.
Dr R. Plestina and Dr P.G. Jenkins, both members of the IPCS
Central Unit, were responsible for the overall scientific content and
technical editing, respectively. The efforts of all who helped in the
preparation and finalization of the monograph are gratefully
acknowledged.
ABBREVIATIONS
ACTH adrenocorticotropic hormone
ADI acceptable daily intake
a.i. active ingredient
BSP bromosulfophthalein
CIMS chemical ionization mass spectrometry
CNS central nervous system
CORT corticosteroid
DNA deoxyribonucleic acid
EC emulsifiable concentrate
ECG electrocardiography
GC gas chromatography
HPLC high performance liquid chromatography
IgM immunoglobulin M
JMPR Joint FAO/WHO Meeting on Pesticide Residues
MRL maximum residue limit
Mu Chinese measure of an area equivalent to 1/15 acre
or 1/60 ha or 166 m2
MS mass spectroscopy
NADPH reduced nicotinamide adenine dinucleotide
NC cell activity natural cytotoxic cell activity
NK cell activity natural killer cell activity
NOEL no-observable-effect level
PL prolactin
SAP serum alkaline phosphatase
SGOT serum glutamate-oxalate transaminase
SGPT serum glutamate-pyruvate transaminase
SIR standard incidence rate
SMR standardized mortality ratio
SPF specific pathogen free
TLC thin layer chromatography
TLm median tolerance limit
UV ultraviolet
1. SUMMARY
1.1 Identity, physical and chemical properties, and analytical
methods
Chlordimeform is a base of medium strength and forms stable salts
with strong acids. Both chlordimeform and its hydrochloride salt in
the pure state are colourless crystalline solids. Chlordimeform base
has a melting point of 32�C, while the hydrochloride salt has a
melting point of 225-227�C. Chlordimeform base is sparingly soluble in
water (250 mg/litre) and readily soluble in organic solvents, whereas
the hydrochloride salt is readily soluble in water but less soluble in
organic solvents. Chlordimeform base has a vapour pressure at 20�C of
48 mPa and a log Kow of 2.89. A wide range of analytical methods are
available for detection and quantification of chlordimeform in plants,
soil, water and urine.
1.2 Sources of human and environmental exposure
Chlordimeform does not occur naturally. It is manufactured
commercially by condensation of the Vilsmeier reagent (obtained by
reaction of dimethylformamide with POCl3, SOCl2 or COCl2) either
with 4-chloro- o-toluidine or with o-toluidine and subsequent
chlorination of the resulting intermediate . It has been used as a
broad spectrum acaricide and is active mainly against motile forms of
mites and ticks and against eggs and early instars of some
Lepidoptera insects. It is active in the vapour phase as well as by
contact. In the early period of its use, it was used on a wide variety
of crops such as pome fruits, stone fruits, cole crops, vegetables,
grapes, hops, citrus fruits, apples, pears, cherries and strawberries.
It was also used in cattle dips for the control of cattle ticks. In
the latter years, its use was generally restricted to cotton, although
in some countries, there was continued use on rice. Its registration
was voluntarily withdrawn in 1988/1989 in most countries. In China,
production stopped in 1992 and sales ceased in 1993.
1.3 Environmental transport, distribution and transformation
Chlordimeform has a moderate vapour pressure but its evaporation
from plant surfaces is less than would be expected. The hydrolytic
stability of chlordimeform is strongly pH-dependent; it is stable in
acid conditions but rapidly hydrolysed in alkaline conditions.
Chlordimeform has the potential to adsorb to dissolved organic matter.
In soils, chlordimeform is primarily dissipated by microbial
action with some contribution by chemical hydrolysis. There is little
evidence of leaching despite its water solubility, which may be
due to its adsorption to clay minerals, soil organic matter and
biodegradation. The principal metabolites are N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine.
There is a low but measurable uptake of chlordimeform into plants
from soil, sufficient to affect plant-feeding pests. When applied to
the leaves, chlordimeform has only limited capacity to penetrate the
cuticular layers. Chlordimeform is degraded rapidly in plants. The
principal metabolites are demethylchlordimeform, N-formyl-4-
chloro- o-toluidine and 4-chloro- o-toluidine, though not all plants
studied produced the 4-chloro- o-toluidine.
In soils, chlordimeform and its metabolites are dissipated
according to first-order kinetics with a half-life of 20-40 days.
Bioaccumulation studies have demonstrated low uptake of
chlordimeform by aquatic organisms and rapid depuration on transfer to
clean water.
1.4 Environmental levels and human exposure
Levels have not been measured in air and water. Following
applications to paddy fields residues of up to 2900 �g/kg in the top
5 cm of soil and 150 �g/kg in the next 5 cm have been found.
Maximum residue levels were set for a wide range of raw produce
and, in some cases, the residues carried over into processed food. The
Codex maximum residue limits for chlordimeform have been withdrawn.
Occupational exposure to chlordimeform has taken place during
manufacture, formulation and application. In recent years, total
urinary levels of chlordimeform and its metabolites have been used as
a monitor for exposure, and the urine level correlates well with the
degree of skin contamination. Where agricultural workers in the cotton
industry have undergone extensively surveillance for urinary excretion
of chlordimeform, the highest exposure levels were in loaders, washers
and mechanics, with lower levels in flagmen and pilots.
1.5 Kinetics and metabolism in laboratory animals and humans
Chlordimeform is readily absorbed from the gastrointestinal tract
and through the skin of mammals. Rapid excretion follows, with
approximately 80% in the urine and 10-15% in faeces. Low residue
levels are evident in all tissues after approximately 10 days, and
there is no evidence of bioaccumulation. Following dermal
administration in humans, similar rapid excretion through the urine is
observed.
Several oxidized and conjugated metabolites of chlordimeform are
excreted in the urine, demethylchlordimeform, N-formyl-4-chloro-
o-toluidine and 4-chloro- o-toluidine being the major metabolites.
In in vitro studies, the same metabolites are formed,
4-chloro- o-toluidine being the major metabolite.
1.6 Effects on laboratory mammals and in vitro test systems
Chlordimeform has moderate acute toxicity when tested in several
species by oral and dermal routes of administration. The major
metabolites have low oral toxicity when tested in rats. Chlordimeform
causes only slight skin and eye irritation in rabbits. Following
either short- or long-term exposure in both mice and rats with either
chlordimeform or its metabolites, treatment-related changes can be
observed in haematological parameters, and there is some evidence of
hyperplasia of the epithelium of the bile duct and urinary bladder at
the high dose levels. Chlordimeform does not cause an increase in
tumour incidence in rats. In mice, following dietary administration
of either chlordimeform, N-formyl-4-chloro- o-toluidine or
4-chloro- o-toluidine, there is a dose-related increase in
haemorrhagic malignant tumours of vascular origin classified as
malignant haemangioendotheliomas, which cause a dose-related increase
in mortality.
Chlordimeform does not affect reproductive parameters, nor does
it have any teratogenic potential.
Chlordimeform has been tested in a broad range of in vitro and
in vivo genotoxicity assays. No positive responses have been
reported with any of these tests in which unformulated chlordimeform
was tested. In addition, there have been several sporadic and
unconfirmed reports of mutagenic activity induced by N-formyl-
4-chloro- o-toluidine and 4-chloro- o-toluidine. A single report
describes cell transformation induction by both chlordimeform and
4-chloro- o-toluidine. Binding to DNA occurs in the liver of dosed
mice and rats. One major hydrophobic adduct is found at a much higher
level in mice than in rats.
Chlordimeform induces a variety of pharmacological and
biochemical effects in animals, including cardiovascular changes,
hypothermia, hyperexcitability, effects on central visual and auditory
functions, and modulation of biogenic amines and drug-metabolizing
enzymes.
1.7 Effects on humans
Acute poisoning causes fatigue, nausea and loss of appetite, and,
in more severe cases, somnolence, cyanosis, urgency in urination,
cystitis, cardiovascular effects (tachycardia, bradycardia, ECG
changes), coma and shock. Generally, there is complete recovery from
acute intoxication.
Following chronic exposure to chlordimeform, additional symptoms
include abdominal pain, skin itching and rashes (dermal exposure), and
gross and microscopic haematuria. A large number of cases with
clinical symptoms of chronic exposure have been reported in both
chlordimeform-manufacturing plants as well as in agricultural workers.
Following occupational exposure, epidemiological evidence has
provided a strong association between exposure to the metabolite
4-chloro- o-toluidine and the incidence of human urinary bladder
cancer. There is currently only weak evidence for an association
between exposure to chlordimeform and human bladder cancer.
1.8 Effects on other organisms in the laboratory and field
There were no significant effects on populations of soil fungi,
bacteria or actinomycetes following application of chlordimeform to
soil.
There are no laboratory toxicity data on freshwater
invertebrates. Growth of larval oysters was inhibited by chlordimeform
with an EC50 of 5.7 mg/litre. The 96-h LC50 for pink shrimp, the only
crustacean studied, was 7.1 mg/litre and the 96-h LC50 values for
fish ranged from 1 to 54 mg/litre. There are no chronic aquatic
toxicity data available. A mixture of laboratory and field data shows
that chlordimeform is toxic to a wide range of terrestrial non-target
arthropods.
The contact toxicity LD50 for bees has been reported to be
120 �g/g and that for oral toxicity 187 �g/g. There was no mortality
in the field following exposure of species of bees to residues on
alfalfa 3 h after spraying.
The dietary LC50 for various birds species ranged from >1000 to
>5000 mg/kg diet.
1.9 Evaluation of human health risks and effects on the environment
Heavy exposure during manufacture or use, possibly resulting from
inadequate safety precautions, has led to signs of acute poisoning in
workers. Since both production and use are reported to have ceased
worldwide, acute poisoning should no longer occur. The risk associated
with chronic exposure, however, particularly the risk of bladder
cancer, will continue to be of concern for many years. Health
screening of significantly exposed individuals from manufacturing
plants from those rural communities where chlordimeform was
extensively used should be continued.
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
1.10 Conclusions and recommendations
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine and, to a lesser
extent, chlordimeform.
Chlordimeform does not persist in the environment, and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Identity
Common name: Chlordimeform
Chemical structure:
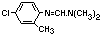 Chemical formula: C10H13ClN2
Relative molecular mass: 196.7
CAS name: N'(4-chloro-2-methylphenyl)-
N, N-dimethyl-methanimidamide
IUPAC name: N2-(4-Chloro- o-tolyl)-
N1, N1-dimethylformamidine
CAS registry number:
Chemical formula: C10H13ClN2
Relative molecular mass: 196.7
CAS name: N'(4-chloro-2-methylphenyl)-
N, N-dimethyl-methanimidamide
IUPAC name: N2-(4-Chloro- o-tolyl)-
N1, N1-dimethylformamidine
CAS registry number:  In the same study, metabolites extracted from goat urine were
analysed by thin-layer chromatography. The major urinary metabolite
was N-formyl-4-chloro- o-toluidine. The metabolites in goat urine
showed a similar pattern to those in rats, with a similar proportion
of conjugated material.
The comparative metabolic fate of chlordimeform in rats, goats
and dogs is considered in a review by Knowles (1970), which emphasizes
the similarity between these species.
6.2.3 In vitro studies
Initial studies on the in vitro metabolism of chlordimeform
were conducted with [3H-phenyl]-chlordimeform (FAO/WHO, 1972).
Incubation of [3H-phenyl]-chlordimeform for 120 min with rat liver
homogenate resulted in 24% unchanged chlordimeform, 45% 4-chloro- o-
toluidine, and 11% unidentified metabolites being formed. Rabbit liver
homogenate yielded 53, 40 and 7% of the same metabolites,
respectively. Incubation of 60 �g [3H-phenyl]-chlordimeform (30 �Ci)
with 5 ml human plasma yielded N-formyl-4-chloro- o-toluidine only.
Conversion was 25% in 5 h, and 50% in 20 h. Rose (1969a) confirmed the
rat liver homogenate studies using [14C]-chlordimeform. Three
unidentified metabolites were also observed and, in addition,
chlordimeform degradation was shown to require the presence of
nicotinamide. Spleen homogenates were inactive with regard to
chlordimeform degradation.
The metabolism of chlordimeform in vitro was first reported by
Ahmad & Knowles (1971). Incubation of [14C]-chlordimeform with
various rat liver enzyme preparations identified demethylchlordimeform
as the major metabolite, which was formed by microsomal
N-demethylase in the presence of exogenous nicotinamide. This
reaction was inhibited by mixed function oxidase inhibitor, SKF-525A.
The chlordimeform metabolites formed in vitro were qualitatively
similar to those detected in urine from chlordimeform-treated mammals.
This has been confirmed by others (Hill et al., 1979; Ghali
& Hollingworth, 1985; Kimmel et al., 1986; Watanabe & Matsumura,
1987).
Knowles & Benezet (1977) confirmed that the major in vitro
metabolite was demethylchlordimeform, but also found that
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
present in appreciable amounts.
Ahmad & Knowles (1971) also investigated the metabolism of
[14C]- N-formyl-4-chloro- o-toluidine) in the presence of rat liver
enzyme preparations. Eighty percent of this metabolite was metabolized
by an enzyme, probably a hydrolase, in the soluble fraction, with
major metabolites being 4-chloro- o-toluidine (52%) and an unknown
substance (26%).
The question of the possible formation of azo-derivatives in
animal tissues was investigated by Rose (1969a). A number of
experiments were conducted to investigate the presence or absence of
azobenzene formation from chlordimeform or 4-chloro- o-toluidine. In
the first experiment, it was demonstrated that peroxidase activity was
negligible in rat liver and spleen. Furthermore, catalase, which was
abundant in the same tissues, and which, like peroxidase, catalyses
reactions between hydroxyperoxides and many oxidizable compounds, was
shown to be unable to form symmetrical azo-derivatives from
4-chloro- o-toluidine. In the second experiment, it was demonstrated
that rat liver and spleen homogenates, which were fortified with
nicotinamide, and which degrade chlordimeform to demethylchlordimeform
and small quantities of N-formyl-4-chloro- o-toluidine and
4-chloro- o-toluidine, respectively, did not form any azobenzene
derivatives. These compounds therefore do not represent metabolites of
chlordimeform or its aromatic amine degradation products in animal
tissues.
Lin et al. (1975) have investigated the metabolism of
chlordimeform in primary embryonic lung cell cultures. In 2 h of
incubation, 97% of chlordimeform was metabolized to N-formyl-4-
chloro- o-toluidine (81.9%) and 4-chloro- o-toluidine (2.3%). The
route of metabolism, which was different to that seen in mammals,
appeared to be first demethylation followed by cleavage at the
carbon-nitrogen double bond to form N-formyl-4-chloro- o-toluidine.
The formation of the demethylchlordimeform was minute compared to that
of the N-formyl derivative. The minor metabolites observed were
demethylchlordimeform and two unknown metabolites. When incubated in
culture media without cells, chlordimeform decomposed to
N-formyl-4-chloro- o-toluidine.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 Oral
The acute oral toxicity data for chlordimeform is presented in
Table 5 and for chlordimeform hydrochloride in Table 6.
The general signs of toxicity in rats are hyperactivity,
dyspnoea, muscular weakness, tremors, "Straub's tail", spasms,
convulsions and respiratory arrest. No pathological changes were noted
in the rat following oral treatment.
In mice, signs of toxicity were similar, but some differences
were noted. Symptoms included restlessness, hyperreflexia and tremors,
particularly of the head and forelimbs, that developed to one or more
episodes of clonic convulsions. Death usually occurred within one hour
during one of the convulsive episodes. If the animal survives this
hyperexcitation and tremor, it becomes sedated, locomotion is
suppressed, and it usually recovers within 24 h.
The acute oral toxicity data for metabolites of chlordimeform is
presented in Table 7.
7.1.2 Other routes
The acute dermal toxicity data for chlordimeform in rats is
presented in Table 5 and for chlordimeform hydrochloride in Table 6.
The base, but not the hydrochloride, is readily absorbed by the skin
(Knowles, 1991). The general signs of toxicity are dyspnoea,
exophthalmos, prostration, spasms and convulsions. Pathological
changes in the rat included pale or blotchy liver, pale kidneys, and
haemorrhagic intestinal contents. No local skin irritation occurred.
In the dog, a lethal intravenous dose of chlordimeform (50 mg/kg
body weight) caused rapid and irreversible hypotension, and
respiratory arrest followed cardiovascular collapse within a few
seconds. Artificial respiration did not protect the animals against
hypotension and death, suggesting cardiovascular collapse is probably
the primary cause of death in dogs. Pathological examination following
oral administration in dogs revealed congestion of liver, kidneys and
lungs.
The acute inhalation LC50 of chlordimeform base in rats
(male and female) was 17 400 mg/m3 and for chlordimeform HCl was
>5800 mg/m3 (FAO/WHO, 1972).
The acute dermal toxicity data for metabolites of chlordimeform
is presented in Table 7.
Table 5. Acute toxicity of chlordimeform in experimental animals
Species Sex Route LD50 References
(mg/kg
body weight)
Rat male/female oral 250 FAO/WHO (1972)
male/female oral 340 Worthing & Walker (1983)
male/female oral 123 Robinson et al. (1975)
male/female oral 301 Gaines & Linder (1986)
male/female oral 178-220 FAO/WHO (1972)
male/female oral 178 FAO/WHO (1972)
female oral 170-460 FAO/WHO (1972)
female oral 265 Gaines & Linder (1986)
female oral 460 FAO/WHO (1972)
male/female dermal 640 FAO/WHO (1972)
male dermal 337 Gaines & Linder (1986)
female dermal 263 Gaines & Linder (1986)
Mouse male/female oral 290 Haddow & Shankland (1969)
male oral 267 Ghali & Hollingworth (1985)
male/female ip 110 FAO/WHO (1972)
Rabbit - oral 625 FAO/WHO (1972)
- oral 625 Worthing & Walker (1983)
Dog male oral approx.150 Hurni & Sachsse (1969)
female oral approx.100 Hurni & Sachsse (1969)
Table 6. Acute toxicity of chlordimeform hydrochloride in
experimental animals (FAO/WHO, 1972)
Species Sex Route LD50
(mg/kg body weight)
Rat male oral 305
male oral 325
female oral 330
male iv 95
- dermal approx. 4000
Mouse male/female oral 220
Rabbit - dermal >4000
Table 7. Acute toxicity of chlordimeform metabolites in the rat (FAO/WHO, 1972)
Metabolite Sex Route LD50
(mg/kg
body weight)
N-formyl-4-chloro-o-toluidine male/female oral 2900
4-chloro-o-toluidine (base) male/female oral approx.1000
4-chloro-o-toluidine-HCl male/female oral 860
N-formyl-4-chloro-o-toluidine male/female dermal (24 h) >2150
4-chloro-o-toluidine (base) male/female dermal (24 h) approx.1800
4-chloro-o-toluidine-HCl male/female dermal (24 h) >2150
7.2 Short-term exposure
7.2.1 Dietary
Dietary studies of 60 days duration have been conducted in the
mouse and rat with each of chlordimeform, N-formyl-4-chloro -o-
toluidine, and 4-chloro- o-toluidine.
7.2.1.1 Mouse
In a study with chlordimeform by Sachsse et al. (1979a), groups
of Tif:MAGf mice (30/sex/group), housed under SPF conditions, were fed
a diet containing chlordimeform at concentrations of 0, 750, 1500,
3000 or 6000 mg/kg for 60 days. This corresponded to dietary intakes
of 0, 107, 194, 717 or 1525 mg/kg body weight per day for females and
0, 110, 200, 669 or 1519 mg/kg body weight per day for males. At the
end of the 60-day period, all animals were examined for haematology,
blood chemistry and urinalysis parameters, and groups of 10 male and
10 female animals from the control and the lower three dose groups
were subjected to gross and microscopic examination of tissues and
organs. Mortality was observed in the two highest dose groups over the
course of the study. The highest dose group was terminated after two
weeks because of a poor general condition of the animals. Growth, as
shown by body weight gain, was reduced in all dietary groups. Food
consumption was reduced at all dietary levels in females only. No
clinical signs of toxicity were noted. Ophthalmological and auditory
examinations were normal. Haematological investigations showed
haemolytic anaemia in both sexes of all treated groups, which was
characterized as a reduction in haemoglobin concentration, red blood
cell count, and packed cell volume. The anaemia was associated in a
dose-related manner with an increased methaemoglobin concentration and
an increase in Heinz body formation. At 3000 mg/kg diet, there was a
slight reticulocytosis noted in both sexes. This was accompanied in
females by a shift in the differential leucocyte count noted as an
increase in the percentage of polymorphonuclear neutrophile and a
decrease in the percentage of lymphocytes. Small changes were observed
in alkaline phosphatase activity, which was slightly increased in male
mice at the highest dose level. Total protein concentration was also
slightly reduced in female mice at the highest dose level. Urinalysis
was unremarkable. In the animals that died or were killed within the
first 2-week period, all were found to be emaciated and in poor
general condition. In all treated animals dying during the test
period, congestion of the organs, especially of the liver, was
observed. At the highest dose level, atrophy of thymic tissue was
observed. There was an increased haemosiderosis at the two highest
dose levels. There were no other pathological findings associated with
the presence of chlordimeform in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980a), groups of Tif:MAGf mice (30/sex/group), housed under SPF
conditions, were fed a diet containing N-formyl-4-chloro- o-
toluidine at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg for
60 days. This corresponded to dietary intakes of 0, 138, 379, 1203,
or 3153 mg/kg body weight per day for females and 0, 140, 349, 1023,
2549 mg/kg body weight per day for males. At the end of the 60-day
period, all animals were examined for haematology, clinical chemistry
and urinalysis parameters. A group of 10 males and 10 females from
each dose level was examined for gross and microscopic pathological
changes at the conclusion of the study. Mortality was observed
predominantly at the high-dose level over the course of the study.
There were no clinical signs of toxicity, although food consumption
and growth were depressed at 1500 mg/kg and above in both sexes over
the course of the study. Ophthalmological and auditory examinations
were unremarkable. Significant haematological abnormalities were
observed at all dose levels at the conclusion of the study. Haemolytic
anaemia was observed in both males and females and was characterized
as a reduction in haemoglobin concentration, erythrocyte count
and packed cell volume. There was a dose-related increase in
methaemoglobin concentration and an increase in Heinz body formation.
Additionally, both males and females in all treated groups showed a
significant reticulocytosis, thrombocytaemia, and leucocytosis. At
higher dose levels in both males and females, the leucocytosis was
accompanied by a shift in the differential leucocyte count. There was
a slight increase in the activity of SGOT, SGPT and SAP. Urinalysis
revealed somewhat lower specific gravity and the presence of bile
pigment in animals at the two highest dietary concentrations.
Microscopic examination of tissues and organs revealed cytomegaly and
hyperplasia of the bile duct epithelium and Kupffer cells in some
animals at 750 mg/kg and in most animals at higher dose levels.
Nuclear inclusion bodies were also evident in all treated animals and,
at the highest dose level, moderate centrilobular fatty changes were
observed. Additionally, at the higher dose levels, atrophy of thymic
lymphoid tissue and of splenic white pulp was observed. Substantial
hyperplasia of the epithelium of the urinary bladder was observed in
most animals at the highest dose level and sporadically throughout the
treated groups.
In a study with 4-chloro- o-toluidine by Suter et al. (1976a),
groups of mice (30/sex/group, TIF:NMRI strain) were bred and
maintained under SPF conditions and fed a diet containing 4-chloro-
o-toluidine at concentrations of 0, 750, 1500, 3000 and 6000 mg/kg
for 60 days. Mortality of 50% was observed in the 6000 mg/kg group.
There were no clinical signs of toxicity, although food intake and
growth were retarded at the two highest dose levels. Eye examinations
did not indicate adverse ocular changes. Haemolytic anaemia occurred
in both sexes of all treated groups and was characterized by
reticulocytosis and Heinz body formation. In the male mice of all
treated groups, haemoglobin concentration, packed cell volume and
erythrocyte counts were slightly below that of controls. In addition,
leucocytosis was observed in all animals of all dosage groups with
the exception of females at the 750 mg/kg level. In both sexes at
6000 mg/kg and in the females at 3000 mg/kg total protein
concentration was reduced and blood glucose and urea nitrogen values
were increased. Plasma GPT was increased in male mice at 3000 mg/kg
and above and in females at 1500 mg/kg. Microscopic examination of
tissues and organs at the conclusion of the studies showed slight to
moderate vacuolar changes in hepatocytes, which were pronounced in
animals at the 3000 mg/kg level and above. There was also a marked
congestion of the spleen at these high dose levels. In addition, the
urinary bladder revealed hyperaemia and dilation of the capillaries in
the mucosal layer. These changes were accompanied by oedema, multiple
intra-epithelial haemorrhage and focal proliferation of the
transitional cell epithelium. On occasion, these changes in the
urinary bladder were noted at the lowest concentration.
7.2.1.2 Rat
In a study with chlordimeform by Sachsse et al. (1979b), groups
of Tif:RAIf rats (20/sex/group) were fed a diet containing
chlordimeform at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg
for 60 days. This corresponded to dietary intakes of 0, 84, 137, 222
or 462 mg/kg body weight per day for males and 0, 71, 121, 231 or
464 mg/kg body weight per day for females. Groups of 10 males and 10
females were killed at 60 days and had complete haematology, clinical
chemistry and urinalysis parameters examined. At the end of the study,
10 males and 10 females from each group were subjected to gross and
microscopic pathological examination. Animals that died during the
course of the study were similarly examined. Food intake and growth
were reduced over the course of the study at all dose levels. Slight
mortality was observed at the highest concentration. There were no
clinical signs of toxicity or adverse behaviour at any dose level.
Slight changes in several haematological parameters were noted at the
two highest levels. Methaemoglobin levels were increased in a dose-
related manner at all treatment levels. Heinz bodies were noted in
haematological examination at 1500 mg/kg and above. Slight changes
were noted in several clinical chemistry para-meters including
decreased glucose concentration, increased alkaline phosphatase
activity and increased �-glutamyl transpeptidase activity,
predominantly at the three highest dose levels. Urinalyses showed
slight changes at the two highest dose levels including a reduced
urine volume, reduced protein concentration, and reduced electrolyte
(potassium) level, predominantly at the highest dietary levels.
Terminal body weights of all animals administered chlordimeform were
significantly reduced in a dose-related fashion. Substantial changes
in growth and relative organ weights were noted in both males and
females at all dietary levels. Reductions in the weight of such organs
as the brain, heart, liver, kidneys, adrenals and thymus were reported
for both males and females. In males, reduced kidney and testes
weights were noted only at the highest dose level while reduced
ovarian weights were noted at all dose levels. Other than excessive
emaciation at the highest dose level, no gross anatomical changes were
noted in the animals killed for pathological examination. In most rats
of the highest-dose groups, haemosiderosis in the spleen was observed.
Reduced spermatogenesis was noted at the highest concentration. Focal
hyperplasia of small biliary ducts and of the transitional epithelium,
and increased vascularization in the mucous membrane of the bladder
were observed in the highest-dose group. In addition, the highest-dose
group showed thymic atrophy in several of the animals examined. No
compound-related histopathological changes were noted in rats fed
1500 mg/kg or below in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980b), groups of Tif:RAI rats (30/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of 0,
750, 1500, 3000 or 6000 mg/kg for 60 days. This corresponded to
dietary intakes of 0, 91, 176, 347 or 875 mg/kg body weight per day
for males and 0, 87, 165, 329 and 719 mg/kg body weight per day for
females. Groups of 10 males and 10 females were killed at the
conclusion of the study for complete haematological, clinical
chemistry and urinalysis examinations, and gross and microscopic
pathological examinations of tissues and organs. Extensive mortality
was observed at the high-dose level within the first few weeks of the
experiment. At the end of the third week of treatment, the highest-
dose group was terminated. There was no substantial mortality at 3000
or lower. Food intake and growth were reduced over the course of the
study in a dose-dependent fashion in all dose groups. Apart from the
mortality noted at the high dose level, no clinical signs of toxicity
or adverse behaviour were observed. Auditory and ophthalmological
examinations showed no evidence of loss of these functions in any of
the animals examined. Haematological examination indicated haemolytic
anaemia in both sexes of all treatment groups; characterized by a
reduction in haemoglobin concentration, erythrocyte count and packed
cell volume, and an increase in methaemoglobin level. Heinz bodies
were observed at 3000 mg/kg only. In addition, at 1500 mg/kg and above
there was a slight reticulocytosis and reduced partial thromboplastin
time in these dose groups. Changes in the clinical chemistry
parameters were noted at both 1500 and 3000 mg/kg. Gross examination
of certain tissues and organs showed changes in absolute weights
and relative weight ratios at all dosage levels. These reductions
appeared to follow a dose-dependent relationship. Animals administered
6000 mg/kg showed atrophy of the thymus and spleen within the first
three weeks of the test. Liver changes were noted in all dose groups
characterized as hyperplasia of the bile duct epithelium and changes
in the distribution of lipid. At the highest dose level, hyperplasia
of the urinary bladder epithelium and testes was noted. About half the
animals of both sexes in the 6000 mg/kg group showed an increase in
the mitotic index in hepatocytes.
In a study of 4-chloro- o-toluidine by Suter et al. (1976b),
groups of rats (30/sex/group; Tif/RAI strain) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 0, 750, 1500,
3000 and 6000 mg/kg for 60 days. There was no mortality over the
course of the study and clinical signs of toxicity were not observed.
Ophthalmological examinations did not suggest changes related to the
presence of 4-chloro- o-toluidine in the diet. Growth was reduced at
dietary levels of 1500 mg/kg and above. Haemolytic anaemia in both
sexes of all treated groups was characterized by a variety of
haematological changes, including reduced haemoglobin content, reduced
haematocrit content, reduced blood cell count, increased
methaemoglobin content, Heinz body formation, reticulocytosis and
polychromatophilia. In the highest-dose group, an increased number
of immature red blood cells (normoblasts) were observed. An increased
leucocyte count and prothrombin time was recorded at 3000 and
6000 mg/kg. Total protein was slightly reduced at 3000 and 6000 mg/kg
and there was a shift in the globulin content as observed by
electrophoresis. Plasma �-glutamyl transpeptidase of males and
alkaline phosphatase of females was increased at 6000 mg/kg.
Urinalysis was not significantly affected. In all treated animals, the
liver showed an increase in size accompanied by hypertrophy of the
hepatocytes. In the two highest-dose groups, the spleen was enlarged
and microscopic examination showed pronounced congestion and
haemorrhage. In the highest-dose group, slight or moderate
proliferation of the transitional cell epithelium was noted in the
urinary bladder.
7.2.1.3 Dog
In a study with chlordimeform by Blackmore (1969a), four groups
of beagle dogs were fed a dry diet containing either 0 mg/kg (10/sex),
250 mg/kg (8/sex), 500 mg/kg (8/sex) or 1000 mg/kg (10/sex) of
chlordimeform for 2 years. Two male and two female dogs were
sacrificed from each group at 26 and 52 weeks. Body weight was reduced
at 1000 mg/kg, the effect being slightly more pronounced in the
females. Total leucocyte counts were sporadically elevated in both
sexes at 1000 mg/kg and in females at 500 mg/kg. Haematocrit,
haemoglobin and erythrocyte counts tended to be depressed after 2
years in both sexes at 1000 mg/kg. Sporadic slight decreases in serum
albumin were observed, more frequently in males, at 1000 mg/kg.
Terminal spleen-to-body weight ratio was elevated in males at 500 and
1000 mg/kg, and in females at 1000 mg/kg. Histopathological
examinations revealed bile duct hyperplasia, pericholangitis and
nodular hepatocytic hyperplasia at 500 and 1000 mg/kg in both sexes,
and nodular hepatocytic hypertrophy at 1000 mg/kg in both sexes in the
liver. Kidneys showed an increased amount of pigmentation at 500 and
1000 mg/kg in both sexes.
7.2.2 Intubation
7.2.2.1 Rat
Four groups of 10 male and 10 female rats were intubated six
times weekly for one month with 5 ml/kg body weight of a 2% solution
of carboxymethylcellulose containing chlordimeform base at
concentrations such as to give dose levels of 0, 25, 50 or 100 mg/kg
(FAO/WHO, 1972). Body weight was markedly reduced in both sexes at
100 mg/kg. Hyperexcitability was observed in all test animals. At
100 mg/kg, this was apparent 20-30 min after dosing, and was followed
2 to 3 h after dosing by decreased activity and apathy. Recovery was
complete at 4 h. Similar but reduced effects were observed at 50 and
25 mg/kg, and with inconsistent frequency.
7.3 Long-term dietary exposure
7.3.1 Mouse
While there have been a number of long-term studies in mice with
chlordimeform and its metabolites, these were specifically designed to
study carcinogenic potential and are described in section 7.7.1.
7.3.2 Rat
In a study with chlordimeform by Blackmore (1969b), groups of
rats (35/sex/group) were fed a diet containing 0, 100, 250, 500 or
1000 mg/kg chlordimeform for 2 years. The 100 mg/kg group commenced
treatment 7 weeks after the other groups. This group was originally
part of the control group. Animals at that time were of similar weight
to those that had already been on test. The 1000 mg/kg group was
discontinued at 3 months due to severe growth inhibition. Growth
inhibition was observed in the males at 500 and 1000 mg/kg. In the
females, weight gain was reduced at 250 mg/kg and above. In addition,
female body weight gain was reduced at 100 mg/kg between weeks 20 and
48. Food intake was significantly reduced at 500 and 1000 mg/kg in
both sexes. Dose-related decreases in haematocrit, haemoglobin, and
erythrocyte counts, and a dose-related increase in the leucocyte count
occurred in females at 250 and 500 mg/kg up to one year. During the
second year, haematocrit only was consistently depressed in females at
500 mg/kg. Histopathological changes in the liver (nodules, and foci
of hyperplasia of hepatocytes) occurred in all groups, but the
incidence was greater at 250 and 500 mg/kg and was more severe at
500 mg/kg. Some females at 500 mg/kg showed slight hypertrophy and
vacuolation of focal groups of cells in the adrenal cortex. Terminally
organ to body weight ratios were increased in the liver (females at
250 and 500 mg/kg and males at 100 and 250 mg/kg), kidney (females at
250 and 500 mg/kg), thyroid (females at 250 and 500 mg/kg), heart
(males at 250 mg/kg and females at 500 mg/kg), adrenals (males at 100
and 250 mg/kg) and testes (100 and 500 mg/kg).
In a study with chlordimeform by Zak et al. (1973), groups of
rats (25/sex/group) were fed a diet containing chlordimeform at
concentrations of 0, 50, 75, 100, 250 and 500 mg/kg for one year. Food
consumption and weight gain data were recorded through the study.
Terminal organ weights and gross and microscopic examinations of
tissues and organs were the only other parameters reported. The
500 mg/kg group was terminated at 37 weeks after 10 males and 8
females had died. At the conclusion of the study, there was
considerable mortality noted in all groups. Food consumption was
decreased at 500 mg/kg in both sexes and was slightly reduced at
100 mg/kg and above in males only. This reduced food consumption was
not significantly reflected in the growth curves of males and females.
Gross examination did not show any compound-related abnormalities.
Organ weights and organ/body weight or organ/brain weight ratios did
not differ from control values. Histological examinations of liver and
spleen were performed on all animals. There were no significant
differences from control values with respect to fatty changes and
inflammatory changes in the liver. Slight proliferation of the bile
duct was more frequent in female rats treated with 500 mg/kg than in
the rats of other treated groups or the control group. Results of
examinations of the spleen for haemosiderosis suggested that, while
this condition was more pronounced in females, there were no
significant differences from control values.
In a study by Sachsse et al. (1980c), rats (90/sex/group;
Tif: RAIf strain) were fed a diet containing chlordimeform at
concentrations of 0, 2, 20, 100 or 500 mg/kg for 24 months. This was
equivalent to dosage levels of 0, 0.1, 1.0, 5.0 and 24 mg/kg body
weight per day for males and 0, 0.1, 1.2, 6.0, and 28 mg/kg body
weight per day for females. At the conclusion of the dietary feeding
study, all remaining rats were fed control diets for a period of time
until a survival rate of 20% per sex (10 rats) per group was attained,
at which time the animals were killed and examined. Groups of 20 male
and 20 female rats per group were examined periodically (4, 13, 26,
52, 78 and 104 weeks) for clinical laboratory investigations including
haematology, blood chemistry and urinalysis. Groups of 10 animals/sex/
group were sacrificed at 27 and 52 weeks for gross and microscopic
examination of tissues and organs. At the conclusion of the study, all
animals sacrificed (also those that died prior to the termination)
were examined for gross and microscopic pathology. There was no
mortality in the study attributable to the presence of chlordimeform.
Growth and body weight were maintained in all groups with the
exception of the 500 mg/kg group, where growth in both sexes was
slightly retarded. There were no clinical signs of toxicity or
abnormal behaviour. Ophthalmological and auditory examinations,
performed at periodic intervals, revealed no adverse effects
attributable to chlordimeform. Methaemoglobinaemia was observed at
dose levels of 20 mg/kg diet and above. At week 4, both males and
females showed a slight, but statistically significant, increase in
methaemoglobin content. At weeks 13 and 26, this condition abated but
returned at the end of one year and was significant in both sexes at
the highest dose level for the remainder of the study. Changes in
several other blood chemistry parameters were observed at the highest
dose level. Heinz body formation generally associated with
methaemoglobinaemia was not observed at week 4, but at the end of year
one and thereafter Heinz bodies were observed at the highest dose
level. A slight but significant reduction in blood glucose
concentration was noted at the higher dose levels throughout a major
part of the study. Slight changes in urinalysis parameters were
observed in the highest dose group, including a slightly reduced
urinary volume and a slightly higher specific gravity. Ketonuria and
proteinuria were observed at the high dose level at the earliest
examination periods only and were not observed at 13 weeks and
thereafter. Gross pathology and organ weight measurements (provided
for 27, 52 and 106-week sacrifice intervals) did not show any
significant dose-related responses. Microscopic histopathological
analyses of tissues and organs (performed at weeks 27 and 52 and at
the termination of the study) indicated no significant changes
attributable to chlordimeform in the diet. Although numerous benign
and malignant tumours were observed in both treated and control
animals, the frequency and type of neoplasms, reported at 12 and 24
months with pathology analyses, were not dose-related nor were they
attributable to chlordimeform in the diet. Several inherent,
degenerative or inflammatory changes were attributed to disease,
common in older animals. There was no indication of carcinogenic
potential to rats as a result of the presence of chlordimeform in the
diet. Based on the haematological occurrence of methaemoglobinaemia,
the no-observed-effect level of chlordimeform for rats was 2 mg/kg
diet, corresponding to an intake of 0.1 mg/kg body weight per day.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980d), groups of Tif:RAIf rats (90/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of
0, 2, 20, 100, or 500 mg/kg for 2 years. This corresponded to dietary
intakes of 0, 0.1, 1.0, 5 or 30 mg/kg body weight per day for females
and 0, 0.1, 1.0, 4.0 or 24 mg/kg body weight per day for males. Groups
of 10 males and 10 females were killed at periodic intervals (26 and
52 weeks) for examination of gross and microscopic pathology. Complete
haemato-logical, clinical chemistry, and urinalysis examinations were
performed at 4, 13, 26, 52 and 78 weeks on 20 males and 20 females of
each group. At 24 months, 20 males and 20 females were killed and
examined for clinical laboratory parameters and gross pathology. The
remaining animals were fed control diets for additional periods of
time until a survival rate of 20% per sex per group was attained.
At that time the remaining animals were killed and examined
microscopically for patho-logical changes, especially neoplastic and
non-neoplastic lesions. In the high-dose group, food intake and growth
were affected over the course of the study and slight growth
retardation was observed. Clinical signs of toxicity or adverse
behaviour were not observed. There was no mortality in the study
attributable to the presence of N-formyl-4-chloro- o-toluidine.
Ophthalmological examinations and auditory tests were normal. The
results of the haematological investigation showed haemoglobin
concentration to be slightly, but significantly, below that of the
controls in both male and female rats at the two highest dose levels.
In addition, slight but significant decreases in the erythrocyte count
and packed cell volume, a slight increase in reticulocytes and
somewhat higher methaemoglobin values were also seen in both male and
female rats at 500 mg/kg. With the exception of lower body weights of
the animals at the highest concentration, the most obvious change was
a significant increase in absolute and relative liver weights in both
sexes, but more pronounced in females, in the 500 mg/kg group. A
significantly increased incidence of hyperplasia of small biliary
ducts was seen in the liver of rats of the 500 mg/kg dose group. In
rats of the 500 mg/kg group that were killed after 2 years or died
after 12 months, a marked increase in the frequency of multioculated
cholangiogenic biliary cysts in the liver was noted. Both of these
finding were more pronounced and more frequent in female than in male
animals. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of neoplasms was
not treatment-related. All gross and histopathological lesions and
changes seen in both control and test animals were described as
inherent, degenerative or inflammatory in origin and were attributed
to naturally occurring diseases common in aged rats. There was no
indication of oncogenic potential in rats as a result of the presence
of N-formyl-4-chloro- o-toluidine. On the basis of the minor
haematological changes, the no-observed-effect level in this study was
20 mg/kg diet, corresponding to an intake of 1 mg/kg body weight per
day.
In a study with 4-chloro- o-toluidine by Sachsse et al. (1980e),
groups of Tif:RAIf rats (90/sex/dose level) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 or 500 mg/kg
for two years. This corresponded to dietary levels of 0, 0.1, 1.0, 5.0
or 28 mg/kg body weight per day for females, and 0, 0.1, 1.0, 4.6 or
24.6 mg/kg body weight per day for males. Groups of 10 males and 10
females were killed at periodic intervals (27 and 54 weeks) for gross
and microscopic pathological examinations. Complete haematological,
clinical chemistry and urinalysis examinations were performed at 4,
13, 26, 52, and 78 weeks on 20 females and 20 males of each group. At
24 months, 20 males and 20 females were killed and examined for
clinical laboratory parameters. Several animals were examined for
gross pathology. The remaining animals were fed control diets for
additional periods of time until a survival rate of 20% per group was
attained. At that time, the remaining animals were killed and examined
for microscopic pathology and oncogenic response. A complete
microscopic analysis was made on at least 10 rats of each sex of each
group at the termination of the experiment. All rats dying during the
course of the study were examined for tumours or neoplasms. In the
high-dose group of female rats, food intake and growth were affected
over the course of the study and slight growth retardation was
observed. There was no effect on growth in male rats at any dose
level. Clinical signs of toxicity were not observed. There was no
mortality in the study attributable to the presence of 4-chloro- o-
toluidine in the diet. Ophthalmological examinations and auditory
tests did not reveal changes that were related to the administration
of 4-chloro- o-toluidine. The results of the haematological
investigation, blood chemistry data and the urinalysis were similar
for both treated and control rats. Periodically, the haemoglobin
concentration was slightly but significantly below that of the
controls in the female rats at 100 mg/kg diet and above. Slight but
significant decreases were observed in the erythrocyte count and
packed cell volume in the female rats at 500 mg/kg. Marginal
reticulocytosis was also found to occur at 500 mg/kg in the female
rats at week 13 and in both sexes at week 26. In both male and female
rats at 500 mg/kg, the methaemoglobin level was found to be slightly
though significantly increased when compared to controls.
Periodically, this change was observed in the females of the 100 mg/kg
dose group, and, occasionally, Heinz bodies were also observed in
female rats. There were some changes to organ weights, organ-to-body
weight ratios and organ-to-brain ratios that were statistically
significant, but only the increase in absolute and relative liver
weights were dose-related. In rats from the 500 mg/kg dose group only,
a slightly but significantly increased incidence of multilobular
cholangiogenic cysts was observed in the liver. These biliary cysts
were found in 10/89 female and 3/90 male rats from the 500 mg/kg
group, compared to 4/89 female and 0/90 male rats in the control
group. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of the neoplasms
occurring in these animals was not treatment-related. Gross and
histopathological lesions and changes seen in both control and treated
animals were described as inherent, degenerative or inflammatory in
origin, and were attributed to naturally occurring diseases, common in
aged rats. There was no indication of oncogenic potential in rats as a
result of the presence of 4-chloro- o-toluidine in the diet. On the
basis of minor haematological changes, the no-observed-effect level in
this study was 20 mg/kg diet, corresponding to a dietary intake of
1 mg/kg body weight per day.
7.4 Skin and eye irritation; skin sensitization
Potential skin irritation was assessed by the application of
0.5 g chlordimeform or its hydrochloride salt to the shaved skin of
six male rabbits. When evaluated at 24 and 72 h, both compounds
produced a very slight irritation (FAO/WHO, 1972).
Potential eye irritation was assessed by application of 0.1 ml of
chlordimeform to one eye of each of nine rabbits, followed by
assessment over 7 days. All animals exhibited slight conjunctival
redness, while one showed slight chemosis. All effects had reversed
within 7 days. There was no evidence of corneal damage. Chlordimeform
may be considered a slight eye irritant (FAO/WHO, 1972).
There were no studies performed to assess the potential for
chlordimeform to cause skin sensitization.
7.5 Reproductive toxicity, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
7.5.1.1 Rat
Four groups of 10 male and 20 female rats were fed a diet
containing 0, 100, 250 and 500 mg/kg chlordimeform in corn oil during
three parental and three two-litter filial generations. Parental body
weight prior to mating tended to be reduced in all treatment groups,
especially at the highest dose level. The same tendency was apparent
with regard to food consumption. The fertility index, gestation index,
live birth index, sex ratio, mean litter size and birth weight of pups
were comparable between treatment and control groups in all
generations. At the 500 mg/kg dose level, the lactation index was
reduced in Fla, Flb and F3a litters. Weaning weight of offspring was
depressed in all high-dose litters. Gross pathological examinations
were performed on parents and pups dying during the study, and on 10
male and 10 female weanlings of the F3b generation. No compound-
related effects were noted in the pathological examination (Blackmore,
1969c).
In a study by Goldman et al. (1991), treatment of ovariectomized
Long-Evans rats with a single intraperitoneal injection of
chlordimeform at dose levels of 25 or 50 mg/kg caused a complete
suppression of luteinizing hormone surge. The observed suppression did
not persist beyond the day of treatment. In a more recent study by
Cooper et al. (1994), the effect of this delay in hormone surge on
pregnancy outcome in females was examined. Chlordimeform at a dose
level of 50 mg/kg resulted in a delay in breeding as well as a
significant reduction in litter size.
Adult male Sprague-Dawley rats were administered chlordimeform by
gavage at 200 mg/kg body weight on one day or 50 mg/kg body weight per
day for 5 days. Rats were killed on either 3 or 13 days after the last
dose. Body weights were reduced at the earlier time points only. There
were no changes in the weights of the testes or associated organs.
Caudal sperm counts were reduced after the single dose only. No other
changes were observed, including sperm motility, velocity or
morphology (Linder et al., 1992).
7.5.1.2 Hamster
Goldman et al. (1993) reported that a single intraperitoneal dose
of chlordimeform (75 mg/kg and above) is capable of delaying the
luteinizing hormone surge and altering the timing of oocyte release in
the hamster. The reproduction consequences of this effect were not
investigated.
7.5.2 Embryotoxicity and teratology
7.5.2.1 Rat
Groups of pregnant rats (25/treatment group, 30 controls) were
administered chlordimeform in carboxymethylcellulose at dose levels of
0, 10, 25 or 50 mg/kg body weight per day from days 6 to 15 of
pregnancy. Only a slight reduction in feed intake was noted at the
intermediate dose level. At the high dose level, dams showed
somnolence through the first 3 days of treatment. There was also a
reduced body weight gain and decrease in feed consumption at this dose
level. Examination of fetuses removed by caesarean section on day 21
showed there was a slight delay in growth of the fetuses at the two
highest dose levels. This effect was probably a direct result of the
toxic response in the dams. No teratogenic events were observed in the
offspring, although an increased incidence of sternal ossification
defects occurred at 25 mg/kg body weight (Fritz, 1975).
7.5.2.2 Rabbit
Three groups of 10 impregnated female New Zealand white rabbits
were administered chlordimeform by gavage on days 8 to 16 of gestation
at dose levels of 0, 7.5 or 30 mg/kg body weight per day. Five rabbits
per group were killed on day 28 of gestation. Parental mortality,
abortion rate, corpora lutea to implantation ratio, litter size,
incidence of resorption, stillbirths, fetal weight, fetal length, and
incidence of skeletal and tissue abnormalities were unaffected by the
test compound. In the remaining rabbits, which were allowed to litter
normally, gestation length, litter size and litter weights were
similar in both treated and control groups (Blackmore, 1969d).
Groups of rabbits (group size ranged from 17 to 38 dams per
group) were given chlordimeform orally from days 6 to 18 of pregnancy
at dose levels of 0, 10, 30 and 100 mg/kg body weight per day. Fetuses
were removed by caesarean section on day 28 of pregnancy. The
administration of chlordimeform at 100 mg/kg body weight produced a
distinct adverse effect on dams for 2-3 h for the first 4 days of
treatment. Examination of dams and fetuses at 28 days suggested that
the low dose had no teratogenic or embryotoxic effect. In the
intermediate and high dose groups, the implantation to corpora lutea
ratio was found to be reduced compared to controls. In the high dose
group, the number of incompletely ossified sternebrae showed a slight
increase over that observed in the controls and in the other groups.
In addition, the number of fetuses with malformations was slightly
increased at 100 mg/kg. These malformations included a median cleft
palate and exencephaly and an omphalocele. Further examination of
spontaneous malformations observed in a cumulative control of 2495
rabbit fetuses suggested that these abnormalities may be spontaneous
and not a consequence of the administration of chlordimeform (Fritz,
1971).
7.6 Mutagenicity and related endpoints
Referenced summaries of the test results with chlordimeform,
N-formyl-4-chloro- o-toluidine, and 4-chloro- o-toluidine are
given in Tables 8, 9 and 10, respectively. The important features of
these data are given below.
7.6.1 DNA damage and repair
Chlordimeform gave no evidence of unscheduled DNA synthesis in
rat hepatocytes (dose levels: 5-625 �g/ml) or in human fibroblasts
(dose levels: 2-250 �g/ml). 4-Chloro- o-toluidine, on the other hand,
gave a slight to moderate dose-related increase in the mean number of
silver grains per nucleus in rat hepatocytes over a dose range of
0.625-78.15 �g/ml, but not in human fibroblasts over the dose range of
1.25-156.25 �g/ml.
Table 8. Summary of mutagenicity and related end-point studies on chlordimeform HC1
Organism Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976a);
TA100 +/-S9 - Konopka & Heymann (1977);
TA1535 +/-S9 - Muecke et al. (1979);
TA1537 +/-S9 - Rashid et al. (1984)
TA1538 +/-S9 -
Salmonella typhimurium TA98 - Arni & M�ller (1983a)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983c)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
WP67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Insects Sex-linked recessive lethals Drosophila +a,b Kale et al. (1995)
Mammalian cells Gene mutation Mouse lymphoma L5178Y- TK+/-/ - Beilstein & M�ller (1984a)
in vitro +/-S9
Unscheduled DNA synthesis Rat hepatocytes - Puri & M�ller (1983a)
Unscheduled DNA synthesis Human fibroblasts - Puri & M�ller (1983c)
Mammalian cells Cell transformation Mouse BALB/3T3 cells +a,b Beilstein & M�ller (1983)
in vitro
Table 8. (con't)
Organism Test Test system Strain +/- References
Mammals Testicular cell chromosome Mouse spermatocytes - Hool et al. (1983)
damage Mouse spermatocytes - Arni et al. (1983a)
Micronucleus assay Chinese Hamster bone - Langauer & M�ller (1977)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & M�ller (1978)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983a)
marrow cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz (1978a)
a Chlordimeform formulation
b Not dose-related
Table 9. Summary of mutagenicity and related end-point studies on N-formyl-4-chloro-o-toluidine
Organism Test Test Systems Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976c);
TA100 +S9 + Konopka & Heymann (1977);
TA100 -S9 - Muecke et al. (1979);
TA1535 +/-S9 - Rashid et al. (1984)
TA1537 +/-S9 -
TA1538 +/-S9 -
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y +a Strasser & M�ller (1984b)
vitro
Mouse lymphoma L5178Y / -a Strasser & M�ller (1983b)
host-mediated assay
Mammals Testicular cell chromosome Mouse spermatogonia - Arni (1983b)
damage Mouse spermatocytes +b Arni & M�ller (1983e)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978a)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983f)
marrow metaphase cells
Table 9. (con't)
Organism Test Test Systems Strain +/- References
Mammals Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978b)
a No positive control
b Chromosome aberrations; not dose-related
Table 10. Summary of mutagenicity and related end-point studies on 4-chloro-o-toluidine
Organisms Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +S9 + Arni & M�ller (1976b);
TA98 +S9 - Haworth et al. (1983);
TA98 -S9 + Konopka & Heymann (1977);
Haworth et al. (1983);
TA100 +S9 - Meuke et al. (1979);
TA100 +S9 - Haworth et al. (1983)
TA100 -S9 - Rashid et al. (1984);
TA1535 +/-S9 - Haworth et al. (1983)
TA1537 +/-S9 -
TA1538 +/-S9 -
S. typhimurium TA98 - Arni & M�ller (1983b)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983d)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y/TK+/- -S9 - Beilstein & M�ller (1984b)
vitro +S9 +
Mouse lymphoma L5178Y + Strasser & M�ller (1984a)
Mouse lymphoma L5178Y - Strasser & M�ller (1983a)
/host-mediated
Table 10. (con't)
Organisms Test Test system Strain +/- References
Mammalian cells in Unscheduled DNA synthesis Rat hepatocytes + Puri & M�ller (1983b)
vitro
Human fibroblasts - Puri & M�ller (1983d)
DNA strand breakage V79 cells + Zimmer et al. (1980)
Cell transformation Mouse BALB/3T3 cells + Beilstein & M�ller (1984c)
Mammals Testicular cell chromosome Mouse spermatogonia - Hool & Arni (1983b)
damage Mouse spermatocytes - Hool & Arni (1983c)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978b)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983d)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983e)
marrow
Sister chromatid exchange Chinese hamster ovary + Galloway et al. (1987)
cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse + Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978)
N-Formyl-4-chloro- o-toluidine was not directly tested for its
ability to induce DNA damage and repair.
The macromolecular binding of 4-chloro- o-toluidine to macro-
molecules of rat and mouse liver has been investigated by several
authors. In a report by Hill et al. (1979), the binding of
4-chloro-2-[methyl-14C]-methylaniline (4-chloro- o-toluidine)
in vivo and in vitro was investigated. The major binding
in vivo was in the liver. In vitro binding was dependent on
the presence for microsomal preparations and NADPH. Two soluble
products of microsomal enzymes were identified as 5-chloro-2-
hydroxylaminotoluene and 4,4'-dichloro-2,2'-dimethylazobenzene. The
hydroxylamino compound appeared to be the more activated form of
4-chloro- o-toluidine.
4-Chloro- o-toluidine caused DNA strand breaks in Chinese
hamster V79 cells (Zimmer et al., 1980).
In studies by Bentley et al. (1986a,b), the covalent binding
of [14C- ring]-4-chloro- o-toluidine to mouse and rat liver
macromolecules was compared. After a single administration to either
species, the extent of binding decreased in the order: protein>RNA>
DNA. The level of binding to mouse liver DNA was approximately twice
as high as the binding to rat liver DNA after either single or
repeated administration, while the binding to liver RNA and protein
was greater in the rat. There was no evidence of an increased level
of incorporation of [3H]-thymidine into DNA as a result of
4-chloro- o-toluidine binding. Two major hydrophobic DNA adducts
were formed in both species, and one of these was formed to a much
greater extent (6-30 fold) in mice.
7.6.2 Mutation
The ability of chlordimeform and its metabolites to induce
mutations has been investigated in both microbial and mammalian
systems.
Chlordimeform itself gave uniformly negative results in
Salmonella typhimurium (0.1-2000 mg/ml), Saccharomyces cerevisiae
(15-400 mg/ml), and Escherichia coli (250-2000 mg/ml), with or
without S9 microsomal activation, as well as in a thymidine kinase
mutation assay in mouse lymphoma L5178Y/TK+/- cells with
(75-3000 mg/ml) or without microsomal activation (42.5-1700 mg/ml).
Kale et al. (1995) reported that a chlordimeform formulation diluted
to provide a dose level of 10 000 �g/ml is a potent sex-linked
recessive mutagen in male pre-meiotic and meiotic cells of Drosophila.
N-Formyl-4-chloro- o-toluidine was also negative in all
Salmonella typhimurium assays (0.1-1000 �g/ml) except for TA100 with
microsomal activation, in which there was a dose-related increase in
revertants. All Escherichia coli assays (250-2000 �g/ml) were
negative. In a forward mutation assay in mouse lymphoma L5178Y cells
(213 & 640 �g/ml), N-formyl-4-chloro- o-toluidine gave a positive
result in two out of three 18-h incubation experiments. In a host-
mediated experiment with mouse lymphoma L5178Y cells (300 mg/kg), the
result was negative.
4-Chloro- o-toluidine was negative in all assays with
Salmonella typhimurium (10-2000 �g/ml) except for TA100 with S9
microsomal activation and TA98 with S9 microsomal activation. Assays
with Saccharomyces cerevisiae (3.2-90 �g/ml) and with Escherichia
coli (250-2000 �g/ml) were negative. In a thymidine kinase forward
mutation assay in mouse lymphoma L5178Y/TK+/- cells, 4-chloro- o-
toluidine was negative without S9 microsomal activation (31.25-
500 �g/ml) and positive with microsomal activation (37.5-600 �g/ml).
In a separate forward mutation assay in mouse lymphoma L5178Y cells
(111 & 255 �g/ml), a positive result was obtained in one out of three
18-h incubation experiments. In a host-mediated experiment with mouse
lymphoma L5178Y cells (330 mg/kg), the result was negative.
7.6.3 Chromosome damage
Sister chromatid exchange assays in Chinese hamster bone marrow
cells were conducted following oral treatment with chlordimeform
(31-324 mg/kg) and 4-chloro- o-toluidine (100-400 mg/kg). In both
cases, the result was negative. Similarly, in an assay for chromosome
aberrations in Chinese hamster bone marrow metaphase cells, a negative
result was obtained following oral treatment with chlordimeform
(2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine (2 � 300-
1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-800 mg/kg), although
the results were somewhat erratic. A micronucleus test in Chinese
hamster bone marrow interphase cells following oral treatment with
chlordimeform (2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine
(2 � 300-1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-400 mg/kg)
was also negative.
Testicular cell chromosomal damage was assessed in mouse
spermatocytes and spermatogonia. To investigate the potential
cytogenetic effects on mouse spermatogonia, chlordimeform
(9-66 mg/kg), N-formyl-4-chloro- o-toluidine (80-320 mg/kg) or
4-chloro- o-toluidine (85-500 mg/kg) was administered orally on 5
consecutive days. The results were negative in each case. To
investigate the potential cytogenetic effects on mouse spermatocytes,
chlordimeform (18-72 mg/kg), N-formyl-4-chloro- o-toluidine
(80-320 mg/kg), or 4-chloro- o-toluidine (85-500 mg/kg) was
administered orally over 10 days on days 0, 2, 3, 5 and 9. The results
were negative in the case of both chlordimeform and 4-chloro- o-
toluidine, but non-dose-related evidence of chromosome damage was
indicated from the results with N-formyl-4-chloro- o-toluidine.
The heritable translocation assay, dominant lethal test, and
mammalian spot test, each of which might indicate minor or major
genomic changes, were conducted on all three compounds. In the
heritable translocation assay, chlordimeform (120 mg/kg/day),
N-formyl-4-chloro- o-toluidine (100 mg/kg/day) or 4-chloro- o-
toluidine (200 mg/kg/day) was administered orally for 49 days. No
induction of translocation heterozygosity was found.
In the dominant lethal assay, chlordimeform (22 or 66 mg/kg),
N-formyl-4-chloro- o-toluidine (105 or 315 mg/kg) or 4-chloro- o-
toluidine (110 or 330 mg/kg) was administered orally as a single dose.
There was no evidence of any dominant lethal effects in the progeny of
male mice.
In the mammalian spot test, chlordimeform (160 mg/kg),
N-formyl-4-chloro- o-toluidine (100 mg/kg) or 4-chloro- o-
toluidine (100 mg/kg) was administered orally on days 8-10 of
embryonic development. The results were negative except in the case of
4-chloro- o-toluidine, which induced a 3.2% incidence of spots of
genetic relevance compared to an incidence of 0.9% in controls.
7.6.4 Cell transformation
Cell transformation assays conducted with both chlordimeform and
4-chloro-toluidine in mouse BALB/3T3 cells produced an increased
incidence of transformed cell colonies with both compounds. With
chlordimeform, the experiment was conducted at dose levels up to
1 �g/ml, and increased transformation frequency was observed only at
0.0625 and 0.125 �g/ml. The transformative properties of chlordimeform
were considered weak. With 4-chloro- o-toluidine, three experiments
were conducted at dose levels up to 36 �g/ml, and a significant
increase in transformation frequency was observed. The transformative
properties of 4-chloro- o-toluidine were considered definite.
7.7 Carcinogenicity
A number of carcinogenicity studies have been conducted in mice.
With chlordimeform, there are two dietary studies and one dermal
study. With N-formyl-4-chloro- o-toluidine, there is one dietary
study. With 4-chloro- o-toluidine, there are four dietary studies.
In rats, the carcinogenic potential of chlordimeform and its
metabolites was generally investigated as part of more detailed
long-term studies, and details are provided in section 7.3.1.2. Three
studies on 4-chloro- o-toluidine that primarily investigated
carcinogenicity are reported below.
7.7.1 Mouse
The carcinogenicity of chlordimeform has been examined in two
dietary studies and in one dermal study.
In a study by Suter et al. (1978), groups of mice (50/sex/group;
Tif: MAG strain, SPF derived) were fed a diet containing chlordimeform
at concentrations of 0, 20, 100 and 500 mg/kg for 24 months. At the
conclusion of the dietary feeding interval, animals were maintained on
control diet until 90% of a group had died, at which time the
remaining animals of the group were sacrificed. There were no signs of
acute toxicity related to chlordimeform in the diet over the course of
the feeding trial. Growth and food consumption were similarly
unaffected by the presence of chlordimeform in the diet. Mortality was
significantly increased in females after 60 weeks at 500 mg/kg, and
after 90 weeks at both 100 and 500 mg/kg. In males, significantly
increased mortality was observed after 70 weeks at 500 mg/kg, and
after 110 weeks at both 100 and 500 mg/kg. However, lifespan was not
significantly affected in males at 100 mg/kg. The animals fed dietary
levels of 100 mg/kg and above displayed an increased incidence of
haemorrhagic tissue masses in subcutaneous tissues, retroperitoneum
and some internal organs (kidney, liver and spleen), which upon
examination were classified as malignant haemangioendotheliomas. These
malignancies which were reported to occur rarely in control
populations were found predominantly in the 100 and 500 mg/kg dietary
groups (see Table 11). In some animals the tumours were of multiple
origin and metastases were observed in the lungs. There were no other
types of neoplasm observed in the study that were attributable to
chlordimeform in the diet. Under the conditions of this study,
20 mg/kg in the diet appeared to be a no-observed-effect level.
In a study by Li et al. (1985a), groups of Swiss mice (50/group,
sex not stated) were fed a diet containing chlordimeform at
concentrations of 0, 20, 100, or 300 mg/kg for a period of 18 months.
A positive control group was administered 300 mg/kg of 4-chloro- o-
toluidine in the diet for 18 months. All animals were killed at the
end of the study and assessed for tumour formation. The main results
of the study are presented in Table 12. The author described the
majority of the neoplasms as angiomas, and the malignant neoplasms as
angiosarcomas. These neoplastic changes were considered to be similar
to those observed in the study by Suter et al. (1978).
In a paper by Jiang et al. (1988), the dermal carcinogenicity of
chlordimeform was investigated in mice. Groups of Swiss mice (50 per
dose level, sex not stated) were treated dermally with chlordimeform
twice per week at dose levels of 0, 100, 500, 2000 or 4000 mg/kg body
weight for a total of 17-20 treatments, together with croton oil (0.5%
in acetone). Positive controls received coal tar pitch (20 treatments)
plus croton oil (30 treatments). All animals were sacrificed after 18
months and analysed for tumour formation. Chlordimeform induced both
skin and liver tumours in this assay (see Table 13). The changes
observed in the skin consisted of epidermal hyperplasia, papillomas
and squamous cell carcinomas. The effect of croton oil application was
Table 11. Incidence of haemangioendotheliomas in mice following dietary administration
of chlordimeform, N-formyl-4-chloro-o-toluidine or 4-chloro-o-toluidine
(Suter et al., 1978; Sachsse et al., 1978a,b)
Control Dietary concentration (mg/kg)
2 20 100 500
Chlordimeform HCl
Male 1/44 (2%) - 0/44 (0%) 15/49 (30%) 39/48 (83%)
Female 1/43 (2%) - 2/46 (4%) 22/46 (50%) 35/49 (71%)
Total 2/87 (2%) - 2/90 (2%) 37/95 (41%) 74/97 (80%)
N-formyl-4-chloro-o-toluidine
Male 0/46 (2%) - 0/49 (0%) 15/48 (38%) 40/47 (89%)
Female 0/47 (0%) - 0/47 (0%) 23/43 (56%) 38/48 (79%)
Total 1/93 (1%) - 0/96 (0%) 38/91 (47%) 78/95 (84%)
4-Chloro-o-toluidine
Male 0/50 (0%) 0/47 (0%) 4/48 (8%) 23/47 (48%) 40/48 (83%)
Female 1/45 (7%) 1/45 (2%) 3/48 (6%) 30/47 (62%) 34/49 (72%)
Total 1/95 (1%) 1/92 (1%) 7/96 (19%) 53/94 (66%) 74/97 (78%)
to shorten the latent period for tumour formation and also to hasten
the malignant progression of existing neoplasms in the skin. At
500 mg/kg body weight, the time of first appearance of tumours was 483
days without croton oil and 154 days with croton oil. The latency
period also decreased with increasing dose levels of chlordimeform. In
the liver, changes consisted of enlargement, hepatocytic hyperplasia,
and hepatocytic carcinomas.
In a carcinogenicity study by Sachsse et al. (1978a), groups of
mice (50/sex/group; Tif: MAG strain) were fed N-formyl-4-chloro- o-
toluidine in the diet at concentrations of 0, 20, 100 and 500 mg/kg
for 24 months. After this time, all animals were fed a control diet
until the study was concluded when 90% of the animals in a group had
Table 12. Incidence of tumours in mice after dietary administration of chlordimeform or 4-chloro-o-toluidine (Li et al., 1985a)
Dietary Number of animals Number of animals Incidence Number of animals Incidence Days to
concentration necropsied bearing haemangiomas (%) bearing (%) appearance of
(mg/kg diet) or haemangiosarcomas haemangiosarcomas neoplasm
Chlordimeform
0 50 0 0 0 0 -
20 50 8 16 0 0 494
100 50 22 44 5 10 469
300 50 36 72 15 30 448
4-Chloro-o-toluidine
300 50 31 62 13 26 283
Table 13. Incidence of tumours in mice following dermal application of chlordimeform (Jiang et al., 1988)
Group/treatment Skin Liver
Number Carcinomas Papillomas Hyperplasia Number Carcinomas Papillomas Hyperplasia
of animals (%) (%) (%) of animals (%) (%) (%)
Water 18 0.0 0.0 6.1 17 0.0 0.0 0.0
Croton oil alone 17 0.0 0.0 17.6 24 0.0 0.0 8.3
100 mg/kg + croton oil 19 0.0 5.3 21.1 21 23.8 0.0 9.5
500 mg/kg 22 4.6 4.6 18.2 20 25.0 0.0 0.0
chlordimeform alone
500 mg/kg + croton oil 23 4.4 4.4 52.2 25 8.0 0.0 4.0
2000 mg/kg + croton oil 15 20.0 20.0 26.7 14 14.3 0.0 0.0
4000 mg/kg + croton oil 15 60.0 13.3 13.3 16 18.8 6.2 0.0
Coal tar pitch 18 88.9 11.1 0.0 19 15.8 5.3 0.0
died. There was no sign of adverse behaviour, and acute mortality was
not noted. Growth and food consumption were unaffected. There were
significant differences noted in survival after one year of age. Both
males and females showed an increased mortality at 100 and 500 mg/kg
after approximately one year of feeding. The onset of increased
mortality occurred earlier in females. The females at the 20 mg/kg
dietary level showed a slightly higher, non-significant, mortality
during the same period. Detailed gross and microscopic examination of
a variety of tissues and organs showed the presence of numerous gross
anatomical lesions. There was an increased number of haemorrhagic
masses in the subcutaneous tissues in the retroperitoneum and in some
internal organs of mice at all treatment levels. Detailed microscopic
examination confirmed that the increased incidence of haemorrhagic
masses were malignant tumours of vascular origin. These tumours were
histologically classified as malignant haemangioendotheliomas (see
Table 11). In addition to the occurrence of tumours, the time to
tumour relationship was decreased as the dietary concentration was
increased. Other neoplasms occurring in the study were not considered
to be treatment-related. A no-observed-effect-level was not
demonstrated under the condition of this experiment.
The carcinogenicity of 4-chloro -o-toluidine has been examined
in four dietary studies.
An early study by Ezumi and Nakao conducted in 1974 was reviewed
by the JMPR in 1978 and considered inadequate (FAO/WHO, 1979).
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives described by Homburger et al. (1972) and Weisburger
et al. (1978), groups of CD-1 mice (25/sex/dose level) were
administered 4-chloro- o-toluidine in the diet at dose levels of 0,
750 or 1500 mg/kg for males, and 0, 2000 or 4000 mg/kg for females for
a period of 18 months. All mice were placed on a control diet for an
additional 3 months before sacrifice and complete necropsy and
histopathological examination of tissues. The incidence of
haemangioendotheliomas was increased in males at both low (12/20) and
high (13/20) dose levels compared to concurrent controls (0/14) and
historical controls (5/99), and in females at both low (18/19) and
high (12/16) dose levels compared to concurrent controls (0/15) and
historical controls (9/102).
In a study by Sachsse et al. (1978b), groups of mice
(50/sex/group; Tif: MAGf strain) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 and
500 mg/kg for 24 months. After 24 months, all animals were fed control
diets until the study was concluded when 90% of the animals in a group
had died. There were no overt signs of toxicity. Growth and food
consumption were unaffected by treatment. An adverse effect on
longevity (lifespan) was noted in both males and females at the two
highest dietary levels. At the conclusion of the study upon gross
examination there was a marked increase number of haemorrhagic masses
in subcutaneous tissue, in the retroperitoneum, and in some internal
organs. Microscopic examination revealed an increased incidence of
haemorrhagic malignant tumours of vascular origin at dose levels of
20 mg/kg and above (see Table 11). The tumour incidence in control
exceeded the incidence observed at 2 mg/kg. The tumours were
histologically classified as malignant haemangioendotheliomas and, on
occasion, metastases were observed. There was not only a significant
dose-dependent increase in the total incidence of malignant tumours
but the time to appearance of tumours occurred at a markedly earlier
date in animals at the higher concentrations than in those at the
lower concentrations. A benign variant of the haemangioma was observed
in all groups, and although without the characteristics of malignancy,
did cause local invasion. Thus, the benign and malignant tumours have
been grouped together. The vascular tumours (haemangiomas and
haemangioendotheliomas) of the type that occurred in the mice appeared
to be peculiar to this rodent species. The occurrence of other types
of neoplasms in the study was not influenced by the presence of
4-chloro- o-toluidine in the diet. Under the conditions of this
experiment, 2 mg/kg in the diet appeared to be a no-observed-effect
level.
In a study with 4-chloro- o-toluidine conducted by the National
Cancer Institute (NCI, 1979), groups of B6C3F1 mice (50/sex/dose
level) were administered 4-chloro -o-toluidine in the diet at dose
levels of 3750 or 15 000 mg/kg for males and 1250 or 5000 mg/kg for
females for 99 weeks. Control groups consisted of 20 males and 20
females. There was a dose-related increase in mortality in both sexes.
There was also a dose-related increase in the incidence of
haemangiosarcomas as shown in Table 14. The haemangiosarcomas
apparently originated in fatty tissue adjacent to the genital organs
and not in a particular organ. In some instances, they were observed
to infiltrate the abdominal muscles, uterus, ovary, prostate or
urinary bladder. The haemangiosarcomas were lethal to 89 (75%) of the
affected mice, owing to haemorrhage in the peritoneal cavity and to
the space-consuming character of the lesions. Pulmonary metastasis was
observed in only 5 (4%) of the 119 dosed animals bearing
haemangiosarcomas. Associated pathological alterations that were
recorded at necropsy were haemorrhage in the peritoneal cavity and
variable enlargement of the spleen. It was concluded that 4-chloro-
o-toluidine was carcinogenic in B6C3F1 mice.
In the study of Li et al. (1985a), a single dietary dose of
4-chloro- o-toluidine (300 mg/kg) was given to mice for 18 months as
a positive control. The incidence of tumours was similar to that seen
in mice receiving 300 mg/kg of chlordimeform, but the latency period
was considerably reduced (Table 12).
Table 14. Incidence of tumours in mice following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 3750 15 000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 20 50 50 18 49 50
Haemangiosarcomas 0 3 37 0 40 39
(0%) (6%) (74%) (0%) (82%) (78%)
Haemangioma 0 3 5 1 6 0
(0%) (6%) (10%) (6%) (12%) (0%)
7.7.2 Rat
The carcinogenicity of chlordimeform and its metabolites has
generally been considered as part of more detailed long-term studies
(see Section 7.3). In the studies below, carcinogenicity was the
primary consideration.
In a study conducted by the National Cancer Institute (NCI,
1979), groups of F344 rats (50/sex/dose level) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 1250 mg/kg or
5000 mg/kg for 107 weeks. Control groups contained 20 animals of each
sex. There was no significant dose-related trend in mortality in
either sex. There was a variety of neoplastic and non-neoplastic
changes in control and treated rats. There was a small increase in
adenomas of chromophobe cells of the pituitary gland in both male and
female rats compared to controls (see Table 15). All of these tumours
were benign, are also common in this strain of rat and have occurred
in 21% of control female rats in the NCI laboratories. The authors
concluded that on the basis of histopathological examination,
4-chloro- o-toluidine was not carcinogenic in F344 rats.
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives by Weisburger et al. (1978), groups of male Charles
River CD rats were administered 4-chloro- o-toluidine in the diet at
dose levels of 0, 2000 or 4000 mg/kg diet for the first 3 months,
which was then reduced to 0, 500 or 1000 mg/kg diet for the following
15 months. There was no statistically significant increase in tumours
in either of the treated groups.
Table 15. Incidence of tumours in rats following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 1250 5000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 19 48 47 19 48 48
Chromophobe 2 6 15 1 13 15
adenoma (11%) (13%) (32%) (5%) (27%) (31%)
Chromophobe 0 0 2 0 3 1
hyperplasia (0%) (0%) (4%) (0%) (6%) (2%)
7.8 Other special studies
7.8.1 Immunotoxicity
In a study by Wiltrout et al. (1978), the potential of various
pesticides to influence the primary humoral immune response in the
mouse with respect to both dose and time of exposure was examined.
Mice receiving a single oral dose of chlordimeform at approximately
the LD50 level (148 mg/kg body weight) experienced a significant
suppression of humoral response when the dose was administered on the
day of immunization or two days after immunization. No response was
observed at one tenth of the LD50 dose, even when administered for
8 or 28 days.
Further studies by Shopp et al. (1985) investigated the effect of
chlordimeform on both humoral and cell-mediated immunity in the mouse
following both acute and 14-day exposures by the intraperitoneal
route. There was a decrease in IgM antibody-forming (plaque-forming)
cells when measured 4 days after intraperitoneal administration at 20
or 40 mg/kg body weight per day. These dose levels did not result in
any alteration of cell-mediated immunity. When administered orally,
chlordimeform at doses as high as 120 mg/kg body weight per day did
not have any effect on the 4- or 5-day antibody response.
Immunological parameters that may be related to the carcinogenic
activity of chlordimeform in rats were investigated by Thomas et al.
(1990). These included spleen/body weight ratio, splenocyte viability,
T and B cell mitogenesis, natural killer (NK) cell and natural
cytotoxic (NC) cell activity. Chlordimeform was administered
intraperitoneally on three consecutive days at 0, 1, 10 or 75 mg/kg
body weight per day. 4-Chloro- o-toluidine was administered
intraperitoneally on three consecutive days at 0, 10, 50 or 100 mg/kg
body weight per day. Spleen/body weight changes were observed only at
the highest dose of chlordimeform. No changes were observed with
either chemical on splenocyte viability or T and B cell mitogenesis.
An inhibition of NC activity at all chlordimeform doses was observed,
and an inhibition of NK activity was observed at 10 mg/kg body weight
per day and above. The relevance of this result to the carcinogenic
activity of these chemicals is doubtful.
7.8.2 Behavioural effects
Behavioural studies of the effects of chlordimeform in rats were
first investigated by Olson et al. (1978). The effects of exposure
prenatally and post-natally were examined following a dietary intake
of 0.1 mg/kg body weight per day. Early development testing (swimming
and righting reflex) was conducted on rat pups from post-natal days 7
to 17, while motivational, learning and retention tests were conducted
on days 70 to 90. The most significant differences between control and
treated groups was in the swimming task, retarded maturation being
observed in the chlordimeform-fed group. There was no treatment-
related effect with regard to maze tests or with regard to the tests
of motivation.
Moser et al. (1988) examined the behaviour of rats using a
functional observation battery following a single oral administration
of chlordimeform at dose levels of 0, 1, 25 or 56 mg/kg body weight.
Rats were examined at 1, 5 or 24 h. Chlordimeform produced a decrease
in body weight as well as a decrease in body temperature. There was a
dose-related increase in general activity, CNS excitability and
sensory responsiveness, coupled with a decrease in rearing, gait and
arousal. Chlordimeform also produced an increase in grip strength.
Other behavioural effects observed with chlordimeform have
included appetite stimulation in rats (Pfister et al., 1978b), flavour
aversion in both rats (MacPhail & Leander, (1980) and mice (Leander et
al., 1984) and alteration in schedule-controlled performance in rats
(MacPhail & Leander, 1981), mice (Glowa, 1986) and pigeons (Leander &
MacPhail, 1980). Witkin & Leander (1982) also demonstrated that, while
causing appetite stimulation in rats, chlordimeform produced a dose-
related decrease in water consumption, in contrast to other appetite
stimulants.
7.8.3 Pharmacological and biochemical effects
The pharmacological and biochemical effects of chlordimeform in
animals have been reviewed by Knowles (1991).
The cardiovascular effects of chlordimeform treatment were
recognized from an early stage with the observation that chlordimeform
administered intraperitoneally to rabbits caused a marked decrease in
arterial blood pressure of almost 50% within 30 min of treatment
(Matsumura & Beeman, 1976). Cardiovascular changes were also noted in
the dog (Lund et al., 1979a,b; Rieger et al., 1981) but in this case
the effect was biphasic, consisting of an initial depressor response
associated with decreased cardiac contractility and vascular
resistance, and a secondary pressor response associated with increased
cardiac contractility and vascular resistance. These actions of
chlordimeform were noted to be similar to those of local anaesthetics
such as procaine and lidocaine (Pfister et al., 1978a; Lund et al.,
1979a,b,c).
In studies by Watkinson (1985, 1986a,b), the effects of
chlordimeform on cardiovascular functional parameters were examined in
post-weaning and geriatric rats following intravenous treatment at
dose levels up to 60 and 120 mg/kg body weight, respectively, or
intraperitoneal treatment of post-weaning rats at dose levels up to
60 mg/kg body weight. Chlordimeform produced profound and abrupt
decreases in heart rate and blood pressure within 3 min, together with
multiple arrhythmias and alterations in electrocardiogram waveforms
and intervals. The effects observed in post-weaning rats were less
severe than those observed in geriatric rats.
The inhibition of monoamine oxidase in rats in vivo and
in vitro by chlordimeform and/or its metabolites has been
extensively studied (Beeman & Matsumura, 1973; Maitre et al., 1978;
Benezet et al., 1978; Hollingworth et al., 1979; Kadir & Knowles,
1981; Kaloyanova et al., 1981; Bailey et al., 1982). The lack of
correlation of toxicity of chlordimeform metabolites to monoamine
oxidase inhibition and the fact that chlordimeform is a relatively
weak monoamine oxidase inhibitor suggest that monoamine oxidase
inhibition is not the primary factor involved in the acute toxicity of
chlordimeform (Neumann & Voss, 1977; Robinson & Smith, 1977;
Hollingworth et al., 1979).
Chlordimeform also has an effect on the level of biogenic amines
in brain and plasma of rats, which may in part at least be due to
the inhibition of monoamine oxidase levels. Administration of
chlordimeform to rats was found to produce an increase of 25-70% in
5-hydroxytryptamine, norepinephrine or dopamine levels in brain
(Maitre et al., 1978; Benezet et al., 1978; Bailey et al., 1982).
However, Johnson & Knowles (1983) treated rats subcutaneously with
chlordimeform (200 mg/kg body weight) and found no change in any of
the amines.
Chlordimeform and some of its metabolites have been shown to
affect platelet function, as measured by the uptake of radioactive
5-hydroxytryptamine (Knowles, 1991).
Chlordimeform also has antipyretic and anti-inflammatory actions,
as shown by its ability to reduce yeast-induced fever in rats. It also
antagonizes both early (5-hydroxytryptamine- and histamine-mediated)
and late (prostaglandin-mediated) phases of carrageenan-induced
hind-paw oedema, albumin-induced oedema, and oedema induced by direct
injection of 5-hydroxytryptamine and histamine (Yim et al., 1978).
Chlordimeform also induced mild gastric ulceration in rats after
intraperitoneal injection (20-80 mg/kg body weight) but not after oral
treatment (80-240 mg/kg body weight). The above actions may be related
to the ability of chlordimeform to inhibit prostaglandin biosynthesis
(Yim et al., 1978; Holsapple & Yim, 1981).
Chlordimeform induces hypothermia in rats (Watkinson & Gordon,
1987) and mice (Gordon et al., 1985). Watkinson et al. (1989)
examined the effect of core body temperature on both the survival
and cardiovascular functions of rats following treatment with
chlordimeform. The results indicated that at a given dose of
chlordimeform, the magnitude and duration of the observed toxic
effects are primarily a function of core body temperature. The authors
concluded that moderate hypothermia, but not extreme hypothermia, may
have a beneficial effect with respect to survival after exposure to
chlordimeform.
Chlordimeform has been shown to have an effect on both visual and
auditory functions in mammals. Intraperitoneal treatment of male rats
with acute dosages of chlordimeform (5-40 mg/kg body weight) before
testing revealed a temporary increase in both the amplitude and
latency of pattern reversal-evoked potentials and an increase only in
the latency of pattern flash-evoked potentials (Dyer & Boyes, 1983;
Boyes & Dyer, 1984). Boyes & Moser (1988) provided evidence to support
the hypothesis that these effects are evoked through actions as a
central nervous system alpha-adrenegic agonist. Janssen et al. (1983)
demonstrated effects on the brain stem auditory-evoked response after
injection of chlordimeform at a dose levels of 40 mg/kg body weight.
It has been suggested that these effects may by secondary to the
hypothermic effects induced by chlordimeform (Gordon et al., 1985).
Chlordimeform has been shown to affect the activity of hepatic
drug-metabolizing enzymes in both rats and mice. Studies have been
conducted following gastric intubation at dose levels up to 150 mg/kg
body weight per day for 7 days, and also following intraperitoneal
injections either singly (100 mg/kg body weight) or daily (75 mg/kg
body weight per day) for 4 days. Chlordimeform treatment induced
several of these hepatic drug-metabolizing enzymes with significant
species and/or sex specificity. Cytochrome P-450 content was increased
in all cases.
7.9 Factors modifying toxicity
The factors modifying the acute toxicity of chlordimeform have
been reviewed by Knowles (1991).
7.10 Mechanisms of toxicity - mode of action
7.10.1 Mechanism of acute toxicity
A large number of studies that investigated the mechanism of
action following acute poisoning with chlordimeform have been
reported.
Based on the early in vitro and in vivo studies of Aziz &
Knowles (1973) and Beeman & Matsumura (1973), it was suggested that
the increase in biogenic amines resulting from inhibition of monoamine
oxidase by chlordimeform could account for the variety of toxic signs
following acute poisoning. However, Maitre & Gfeller (1975) and
Robinson et al. (1975) demonstrated that this mechanism does not play
a significant role in the acute toxicity in rats.
A number of other studies have attempted to define the mode of
action of chlordimeform. Studies in insects have shown that
chlordimeform has little activity on cholinergic transmission although
it is an uncoupler of oxidative phosphorylation and an inhibitor of
electron transport (Abo-Khatwa & Hollingworth, 1972a). A number of
biochemical mechanisms have been postulated to explain the effects of
chlordimeform in insects, including uncoupling of respiration
(Abo-Khatwa & Hollingworth, 1972a,b), inhibition of monoamine oxidase
(Knowles & Roulston, 1972) and blockage of neuromuscular transmission
(Wang et al., 1975; Watanabe et al., 1975), and motor stimulation
through actions on central non-cholinergic synapses (Lund et al.,
1979a; Lund et al., 1979c). The latter effect is thought to be
mediated through the neurotransmitter, octopamine (Lund et al.,
1979b). Both chlordimeform and particularly demethylchlordimeform have
been shown to interact with the octopamine receptor and partially
mimic the pharmacological effects of octopamine (Evans & Gee, 1980;
Nathanson & Hunnicutt, 1981; Bokisch et al., 1985).
In mammalian systems, oxidative phosphorylation is uncoupled
(Abo-Khatwa & Hollingworth, 1972b) and RNA synthesis is inhibited by
chlordimeform, but only at very high concentrations (Murakami &
Fukami, 1974). The effects of chlordimeform on hepatic drug-
metabolizing enzymes was dependent on both sex and species and did not
show any particular pattern that would indicate a consistent mechanism
of action (Budris et al., 1983; Bentley et al., 1985; Leslie et al.,
1988).
Chlordimeform, acting as a direct depressant on cardiac and
vascular muscle, induced a hypotensive state in dogs. Chlordimeform
did not interfere with the autonomic nervous system. The mechanism of
cardiovascular depression may be related to that noted with frog nerve
preparations treated with procaine, a local anaesthetic. The
metabolite, 4-chloro- o-toluidine has been shown to interfere with
rat cardiac receptors (Wang et al., 1975; Watanabe et al., 1975;
Matsumura & Beeman, 1976; Knowles, 1976; Hollingworth, 1976; Lund et
al., 1978a).
More recent research has shown that formamidine pesticides may
exert their effects on the central nervous system by interacting
directly with adrenergic receptors, particularly the alpha-2 subtype
(Costa & Murphy, 1987; Costa et al., 1988, 1989). This interaction
appears to mediate several of the observed effects of formamidines,
such as changes in heart rate (Hsu & Kakuk, 1984, Watkinson, 1985;
1986a,b), pupil diameter (Hsu & Kakuk, 1984), visual evoked potential
(Boyes & Moser, 1988) and hormonal secretion (Goldman et al., 1990;
1991). Costa et al. (1991) demonstrated that chlordimeform
decreases the hepatic glutathione content by up to 40% in a
time- and dose-dependent manner, through an interaction with
alpha2-adrenoreceptors. Wu et al. (1990) have demonstrated that
chlordimeform inhibits the binding of the known alpha2-adrenoreceptor
blockers, clonidine and yohimbine, in rat forebrain tissue
in vitro. Furthermore, Stoker et al. (1991), in a further study on
the effects of chlordimeform on hormone release, have demonstrated in
rats, treated intraperitoneally with chlordimeform (20 or 50 mg/kg
body weight), that there is an increase in adrenocorticotropic hormone
(ACTH), circulating corticosteroid (CORT) and prolactin (PL) in a
dose-dependent manner. alpha-Adrenergic agonists specifically
inhibited these effects indicating the interference with a regulatory
signal mediated by alpha-adrenergic receptor-associated activity.
Candura et al. (1992) demonstrated that the inhibition induced by
chlordimeform in the intestinal tract is mediated by calcium channel
blockade rather than by alpha2-adrenoceptor activation. In a study by
Robinson et al. (1975), it was found that using drugs to block the
serotonergic or alpha-adrenergic receptors did not reduce the
chlordimeform-induced lethality in male rats.
7.10.2 Mechanism of carcinogenicity
Chlordimeform and its metabolites, N-formyl-4-chloro- o-
toluidine and 4-chloro -o-toluidine, have been shown to induce mouse
tumours of a vascular origin characterized histologically as
haemangioendotheliomas and haemangiosarcomas. 4-Chloro- o-toluidine
has been shown to be a more potent carcinogen than chlordimeform, both
with respect to dose-response and to a reduced latency period.
Haemangioendotheliomas and haemangiosarcomas were not induced in rats.
Cases of bladder cancer in humans associated with occupational
exposure to high levels of chlordimeform or 4-chloro- o-toluidine
have been seen in groups with high urinary levels of chlordimeform and
4-chloro- o-toluidine.
The exact mechanism of induction of these tumours is unknown but
there is evidence that a genetic mechanism involving mutations induced
by 4-chloro- o-toluidine is involved.
Metabolic studies in mice and rats indicate a similar metabolic
pathway for chlordimeform in both species. The kinetics of absorption
and elimination in mice and rats also seem to be similar. However, the
overall DNA binding was higher in mice than rats, and one DNA adduct
was formed to a 6- to 30-fold higher extent in mice.
There is considerable evidence that 4-chloro- o-toluidine causes
severe toxic effects in the human bladder leading to haemorrhagic
cystitis (see section 8). Monitoring of urinary metabolites in humans
also indicates that chlordimeform is rapidly metabolized to 4-chloro-
o-toluidine in vivo.
4-Chloro- o-toluidine also has a close structural similarity
to aromatic amines for which there is established evidence of
carcinogenicity by animal experimentation and also by human
epidemiological data (Parkes, 1984).
Taken together, the evidence strongly implicates 4-chloro- o-
toluidine as the causative agent in the induction of tumours in both
mice and humans. A proposed route of activation that may be associated
with carcinogenicity is shown in Fig. 2. However, the mechanism of the
carcinogenicity remains unclear.
In the same study, metabolites extracted from goat urine were
analysed by thin-layer chromatography. The major urinary metabolite
was N-formyl-4-chloro- o-toluidine. The metabolites in goat urine
showed a similar pattern to those in rats, with a similar proportion
of conjugated material.
The comparative metabolic fate of chlordimeform in rats, goats
and dogs is considered in a review by Knowles (1970), which emphasizes
the similarity between these species.
6.2.3 In vitro studies
Initial studies on the in vitro metabolism of chlordimeform
were conducted with [3H-phenyl]-chlordimeform (FAO/WHO, 1972).
Incubation of [3H-phenyl]-chlordimeform for 120 min with rat liver
homogenate resulted in 24% unchanged chlordimeform, 45% 4-chloro- o-
toluidine, and 11% unidentified metabolites being formed. Rabbit liver
homogenate yielded 53, 40 and 7% of the same metabolites,
respectively. Incubation of 60 �g [3H-phenyl]-chlordimeform (30 �Ci)
with 5 ml human plasma yielded N-formyl-4-chloro- o-toluidine only.
Conversion was 25% in 5 h, and 50% in 20 h. Rose (1969a) confirmed the
rat liver homogenate studies using [14C]-chlordimeform. Three
unidentified metabolites were also observed and, in addition,
chlordimeform degradation was shown to require the presence of
nicotinamide. Spleen homogenates were inactive with regard to
chlordimeform degradation.
The metabolism of chlordimeform in vitro was first reported by
Ahmad & Knowles (1971). Incubation of [14C]-chlordimeform with
various rat liver enzyme preparations identified demethylchlordimeform
as the major metabolite, which was formed by microsomal
N-demethylase in the presence of exogenous nicotinamide. This
reaction was inhibited by mixed function oxidase inhibitor, SKF-525A.
The chlordimeform metabolites formed in vitro were qualitatively
similar to those detected in urine from chlordimeform-treated mammals.
This has been confirmed by others (Hill et al., 1979; Ghali
& Hollingworth, 1985; Kimmel et al., 1986; Watanabe & Matsumura,
1987).
Knowles & Benezet (1977) confirmed that the major in vitro
metabolite was demethylchlordimeform, but also found that
N-formyl-4-chloro- o-toluidine and 4-chloro- o-toluidine were
present in appreciable amounts.
Ahmad & Knowles (1971) also investigated the metabolism of
[14C]- N-formyl-4-chloro- o-toluidine) in the presence of rat liver
enzyme preparations. Eighty percent of this metabolite was metabolized
by an enzyme, probably a hydrolase, in the soluble fraction, with
major metabolites being 4-chloro- o-toluidine (52%) and an unknown
substance (26%).
The question of the possible formation of azo-derivatives in
animal tissues was investigated by Rose (1969a). A number of
experiments were conducted to investigate the presence or absence of
azobenzene formation from chlordimeform or 4-chloro- o-toluidine. In
the first experiment, it was demonstrated that peroxidase activity was
negligible in rat liver and spleen. Furthermore, catalase, which was
abundant in the same tissues, and which, like peroxidase, catalyses
reactions between hydroxyperoxides and many oxidizable compounds, was
shown to be unable to form symmetrical azo-derivatives from
4-chloro- o-toluidine. In the second experiment, it was demonstrated
that rat liver and spleen homogenates, which were fortified with
nicotinamide, and which degrade chlordimeform to demethylchlordimeform
and small quantities of N-formyl-4-chloro- o-toluidine and
4-chloro- o-toluidine, respectively, did not form any azobenzene
derivatives. These compounds therefore do not represent metabolites of
chlordimeform or its aromatic amine degradation products in animal
tissues.
Lin et al. (1975) have investigated the metabolism of
chlordimeform in primary embryonic lung cell cultures. In 2 h of
incubation, 97% of chlordimeform was metabolized to N-formyl-4-
chloro- o-toluidine (81.9%) and 4-chloro- o-toluidine (2.3%). The
route of metabolism, which was different to that seen in mammals,
appeared to be first demethylation followed by cleavage at the
carbon-nitrogen double bond to form N-formyl-4-chloro- o-toluidine.
The formation of the demethylchlordimeform was minute compared to that
of the N-formyl derivative. The minor metabolites observed were
demethylchlordimeform and two unknown metabolites. When incubated in
culture media without cells, chlordimeform decomposed to
N-formyl-4-chloro- o-toluidine.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single exposure
7.1.1 Oral
The acute oral toxicity data for chlordimeform is presented in
Table 5 and for chlordimeform hydrochloride in Table 6.
The general signs of toxicity in rats are hyperactivity,
dyspnoea, muscular weakness, tremors, "Straub's tail", spasms,
convulsions and respiratory arrest. No pathological changes were noted
in the rat following oral treatment.
In mice, signs of toxicity were similar, but some differences
were noted. Symptoms included restlessness, hyperreflexia and tremors,
particularly of the head and forelimbs, that developed to one or more
episodes of clonic convulsions. Death usually occurred within one hour
during one of the convulsive episodes. If the animal survives this
hyperexcitation and tremor, it becomes sedated, locomotion is
suppressed, and it usually recovers within 24 h.
The acute oral toxicity data for metabolites of chlordimeform is
presented in Table 7.
7.1.2 Other routes
The acute dermal toxicity data for chlordimeform in rats is
presented in Table 5 and for chlordimeform hydrochloride in Table 6.
The base, but not the hydrochloride, is readily absorbed by the skin
(Knowles, 1991). The general signs of toxicity are dyspnoea,
exophthalmos, prostration, spasms and convulsions. Pathological
changes in the rat included pale or blotchy liver, pale kidneys, and
haemorrhagic intestinal contents. No local skin irritation occurred.
In the dog, a lethal intravenous dose of chlordimeform (50 mg/kg
body weight) caused rapid and irreversible hypotension, and
respiratory arrest followed cardiovascular collapse within a few
seconds. Artificial respiration did not protect the animals against
hypotension and death, suggesting cardiovascular collapse is probably
the primary cause of death in dogs. Pathological examination following
oral administration in dogs revealed congestion of liver, kidneys and
lungs.
The acute inhalation LC50 of chlordimeform base in rats
(male and female) was 17 400 mg/m3 and for chlordimeform HCl was
>5800 mg/m3 (FAO/WHO, 1972).
The acute dermal toxicity data for metabolites of chlordimeform
is presented in Table 7.
Table 5. Acute toxicity of chlordimeform in experimental animals
Species Sex Route LD50 References
(mg/kg
body weight)
Rat male/female oral 250 FAO/WHO (1972)
male/female oral 340 Worthing & Walker (1983)
male/female oral 123 Robinson et al. (1975)
male/female oral 301 Gaines & Linder (1986)
male/female oral 178-220 FAO/WHO (1972)
male/female oral 178 FAO/WHO (1972)
female oral 170-460 FAO/WHO (1972)
female oral 265 Gaines & Linder (1986)
female oral 460 FAO/WHO (1972)
male/female dermal 640 FAO/WHO (1972)
male dermal 337 Gaines & Linder (1986)
female dermal 263 Gaines & Linder (1986)
Mouse male/female oral 290 Haddow & Shankland (1969)
male oral 267 Ghali & Hollingworth (1985)
male/female ip 110 FAO/WHO (1972)
Rabbit - oral 625 FAO/WHO (1972)
- oral 625 Worthing & Walker (1983)
Dog male oral approx.150 Hurni & Sachsse (1969)
female oral approx.100 Hurni & Sachsse (1969)
Table 6. Acute toxicity of chlordimeform hydrochloride in
experimental animals (FAO/WHO, 1972)
Species Sex Route LD50
(mg/kg body weight)
Rat male oral 305
male oral 325
female oral 330
male iv 95
- dermal approx. 4000
Mouse male/female oral 220
Rabbit - dermal >4000
Table 7. Acute toxicity of chlordimeform metabolites in the rat (FAO/WHO, 1972)
Metabolite Sex Route LD50
(mg/kg
body weight)
N-formyl-4-chloro-o-toluidine male/female oral 2900
4-chloro-o-toluidine (base) male/female oral approx.1000
4-chloro-o-toluidine-HCl male/female oral 860
N-formyl-4-chloro-o-toluidine male/female dermal (24 h) >2150
4-chloro-o-toluidine (base) male/female dermal (24 h) approx.1800
4-chloro-o-toluidine-HCl male/female dermal (24 h) >2150
7.2 Short-term exposure
7.2.1 Dietary
Dietary studies of 60 days duration have been conducted in the
mouse and rat with each of chlordimeform, N-formyl-4-chloro -o-
toluidine, and 4-chloro- o-toluidine.
7.2.1.1 Mouse
In a study with chlordimeform by Sachsse et al. (1979a), groups
of Tif:MAGf mice (30/sex/group), housed under SPF conditions, were fed
a diet containing chlordimeform at concentrations of 0, 750, 1500,
3000 or 6000 mg/kg for 60 days. This corresponded to dietary intakes
of 0, 107, 194, 717 or 1525 mg/kg body weight per day for females and
0, 110, 200, 669 or 1519 mg/kg body weight per day for males. At the
end of the 60-day period, all animals were examined for haematology,
blood chemistry and urinalysis parameters, and groups of 10 male and
10 female animals from the control and the lower three dose groups
were subjected to gross and microscopic examination of tissues and
organs. Mortality was observed in the two highest dose groups over the
course of the study. The highest dose group was terminated after two
weeks because of a poor general condition of the animals. Growth, as
shown by body weight gain, was reduced in all dietary groups. Food
consumption was reduced at all dietary levels in females only. No
clinical signs of toxicity were noted. Ophthalmological and auditory
examinations were normal. Haematological investigations showed
haemolytic anaemia in both sexes of all treated groups, which was
characterized as a reduction in haemoglobin concentration, red blood
cell count, and packed cell volume. The anaemia was associated in a
dose-related manner with an increased methaemoglobin concentration and
an increase in Heinz body formation. At 3000 mg/kg diet, there was a
slight reticulocytosis noted in both sexes. This was accompanied in
females by a shift in the differential leucocyte count noted as an
increase in the percentage of polymorphonuclear neutrophile and a
decrease in the percentage of lymphocytes. Small changes were observed
in alkaline phosphatase activity, which was slightly increased in male
mice at the highest dose level. Total protein concentration was also
slightly reduced in female mice at the highest dose level. Urinalysis
was unremarkable. In the animals that died or were killed within the
first 2-week period, all were found to be emaciated and in poor
general condition. In all treated animals dying during the test
period, congestion of the organs, especially of the liver, was
observed. At the highest dose level, atrophy of thymic tissue was
observed. There was an increased haemosiderosis at the two highest
dose levels. There were no other pathological findings associated with
the presence of chlordimeform in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980a), groups of Tif:MAGf mice (30/sex/group), housed under SPF
conditions, were fed a diet containing N-formyl-4-chloro- o-
toluidine at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg for
60 days. This corresponded to dietary intakes of 0, 138, 379, 1203,
or 3153 mg/kg body weight per day for females and 0, 140, 349, 1023,
2549 mg/kg body weight per day for males. At the end of the 60-day
period, all animals were examined for haematology, clinical chemistry
and urinalysis parameters. A group of 10 males and 10 females from
each dose level was examined for gross and microscopic pathological
changes at the conclusion of the study. Mortality was observed
predominantly at the high-dose level over the course of the study.
There were no clinical signs of toxicity, although food consumption
and growth were depressed at 1500 mg/kg and above in both sexes over
the course of the study. Ophthalmological and auditory examinations
were unremarkable. Significant haematological abnormalities were
observed at all dose levels at the conclusion of the study. Haemolytic
anaemia was observed in both males and females and was characterized
as a reduction in haemoglobin concentration, erythrocyte count
and packed cell volume. There was a dose-related increase in
methaemoglobin concentration and an increase in Heinz body formation.
Additionally, both males and females in all treated groups showed a
significant reticulocytosis, thrombocytaemia, and leucocytosis. At
higher dose levels in both males and females, the leucocytosis was
accompanied by a shift in the differential leucocyte count. There was
a slight increase in the activity of SGOT, SGPT and SAP. Urinalysis
revealed somewhat lower specific gravity and the presence of bile
pigment in animals at the two highest dietary concentrations.
Microscopic examination of tissues and organs revealed cytomegaly and
hyperplasia of the bile duct epithelium and Kupffer cells in some
animals at 750 mg/kg and in most animals at higher dose levels.
Nuclear inclusion bodies were also evident in all treated animals and,
at the highest dose level, moderate centrilobular fatty changes were
observed. Additionally, at the higher dose levels, atrophy of thymic
lymphoid tissue and of splenic white pulp was observed. Substantial
hyperplasia of the epithelium of the urinary bladder was observed in
most animals at the highest dose level and sporadically throughout the
treated groups.
In a study with 4-chloro- o-toluidine by Suter et al. (1976a),
groups of mice (30/sex/group, TIF:NMRI strain) were bred and
maintained under SPF conditions and fed a diet containing 4-chloro-
o-toluidine at concentrations of 0, 750, 1500, 3000 and 6000 mg/kg
for 60 days. Mortality of 50% was observed in the 6000 mg/kg group.
There were no clinical signs of toxicity, although food intake and
growth were retarded at the two highest dose levels. Eye examinations
did not indicate adverse ocular changes. Haemolytic anaemia occurred
in both sexes of all treated groups and was characterized by
reticulocytosis and Heinz body formation. In the male mice of all
treated groups, haemoglobin concentration, packed cell volume and
erythrocyte counts were slightly below that of controls. In addition,
leucocytosis was observed in all animals of all dosage groups with
the exception of females at the 750 mg/kg level. In both sexes at
6000 mg/kg and in the females at 3000 mg/kg total protein
concentration was reduced and blood glucose and urea nitrogen values
were increased. Plasma GPT was increased in male mice at 3000 mg/kg
and above and in females at 1500 mg/kg. Microscopic examination of
tissues and organs at the conclusion of the studies showed slight to
moderate vacuolar changes in hepatocytes, which were pronounced in
animals at the 3000 mg/kg level and above. There was also a marked
congestion of the spleen at these high dose levels. In addition, the
urinary bladder revealed hyperaemia and dilation of the capillaries in
the mucosal layer. These changes were accompanied by oedema, multiple
intra-epithelial haemorrhage and focal proliferation of the
transitional cell epithelium. On occasion, these changes in the
urinary bladder were noted at the lowest concentration.
7.2.1.2 Rat
In a study with chlordimeform by Sachsse et al. (1979b), groups
of Tif:RAIf rats (20/sex/group) were fed a diet containing
chlordimeform at concentrations of 0, 750, 1500, 3000 or 6000 mg/kg
for 60 days. This corresponded to dietary intakes of 0, 84, 137, 222
or 462 mg/kg body weight per day for males and 0, 71, 121, 231 or
464 mg/kg body weight per day for females. Groups of 10 males and 10
females were killed at 60 days and had complete haematology, clinical
chemistry and urinalysis parameters examined. At the end of the study,
10 males and 10 females from each group were subjected to gross and
microscopic pathological examination. Animals that died during the
course of the study were similarly examined. Food intake and growth
were reduced over the course of the study at all dose levels. Slight
mortality was observed at the highest concentration. There were no
clinical signs of toxicity or adverse behaviour at any dose level.
Slight changes in several haematological parameters were noted at the
two highest levels. Methaemoglobin levels were increased in a dose-
related manner at all treatment levels. Heinz bodies were noted in
haematological examination at 1500 mg/kg and above. Slight changes
were noted in several clinical chemistry para-meters including
decreased glucose concentration, increased alkaline phosphatase
activity and increased �-glutamyl transpeptidase activity,
predominantly at the three highest dose levels. Urinalyses showed
slight changes at the two highest dose levels including a reduced
urine volume, reduced protein concentration, and reduced electrolyte
(potassium) level, predominantly at the highest dietary levels.
Terminal body weights of all animals administered chlordimeform were
significantly reduced in a dose-related fashion. Substantial changes
in growth and relative organ weights were noted in both males and
females at all dietary levels. Reductions in the weight of such organs
as the brain, heart, liver, kidneys, adrenals and thymus were reported
for both males and females. In males, reduced kidney and testes
weights were noted only at the highest dose level while reduced
ovarian weights were noted at all dose levels. Other than excessive
emaciation at the highest dose level, no gross anatomical changes were
noted in the animals killed for pathological examination. In most rats
of the highest-dose groups, haemosiderosis in the spleen was observed.
Reduced spermatogenesis was noted at the highest concentration. Focal
hyperplasia of small biliary ducts and of the transitional epithelium,
and increased vascularization in the mucous membrane of the bladder
were observed in the highest-dose group. In addition, the highest-dose
group showed thymic atrophy in several of the animals examined. No
compound-related histopathological changes were noted in rats fed
1500 mg/kg or below in the diet.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980b), groups of Tif:RAI rats (30/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of 0,
750, 1500, 3000 or 6000 mg/kg for 60 days. This corresponded to
dietary intakes of 0, 91, 176, 347 or 875 mg/kg body weight per day
for males and 0, 87, 165, 329 and 719 mg/kg body weight per day for
females. Groups of 10 males and 10 females were killed at the
conclusion of the study for complete haematological, clinical
chemistry and urinalysis examinations, and gross and microscopic
pathological examinations of tissues and organs. Extensive mortality
was observed at the high-dose level within the first few weeks of the
experiment. At the end of the third week of treatment, the highest-
dose group was terminated. There was no substantial mortality at 3000
or lower. Food intake and growth were reduced over the course of the
study in a dose-dependent fashion in all dose groups. Apart from the
mortality noted at the high dose level, no clinical signs of toxicity
or adverse behaviour were observed. Auditory and ophthalmological
examinations showed no evidence of loss of these functions in any of
the animals examined. Haematological examination indicated haemolytic
anaemia in both sexes of all treatment groups; characterized by a
reduction in haemoglobin concentration, erythrocyte count and packed
cell volume, and an increase in methaemoglobin level. Heinz bodies
were observed at 3000 mg/kg only. In addition, at 1500 mg/kg and above
there was a slight reticulocytosis and reduced partial thromboplastin
time in these dose groups. Changes in the clinical chemistry
parameters were noted at both 1500 and 3000 mg/kg. Gross examination
of certain tissues and organs showed changes in absolute weights
and relative weight ratios at all dosage levels. These reductions
appeared to follow a dose-dependent relationship. Animals administered
6000 mg/kg showed atrophy of the thymus and spleen within the first
three weeks of the test. Liver changes were noted in all dose groups
characterized as hyperplasia of the bile duct epithelium and changes
in the distribution of lipid. At the highest dose level, hyperplasia
of the urinary bladder epithelium and testes was noted. About half the
animals of both sexes in the 6000 mg/kg group showed an increase in
the mitotic index in hepatocytes.
In a study of 4-chloro- o-toluidine by Suter et al. (1976b),
groups of rats (30/sex/group; Tif/RAI strain) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 0, 750, 1500,
3000 and 6000 mg/kg for 60 days. There was no mortality over the
course of the study and clinical signs of toxicity were not observed.
Ophthalmological examinations did not suggest changes related to the
presence of 4-chloro- o-toluidine in the diet. Growth was reduced at
dietary levels of 1500 mg/kg and above. Haemolytic anaemia in both
sexes of all treated groups was characterized by a variety of
haematological changes, including reduced haemoglobin content, reduced
haematocrit content, reduced blood cell count, increased
methaemoglobin content, Heinz body formation, reticulocytosis and
polychromatophilia. In the highest-dose group, an increased number
of immature red blood cells (normoblasts) were observed. An increased
leucocyte count and prothrombin time was recorded at 3000 and
6000 mg/kg. Total protein was slightly reduced at 3000 and 6000 mg/kg
and there was a shift in the globulin content as observed by
electrophoresis. Plasma �-glutamyl transpeptidase of males and
alkaline phosphatase of females was increased at 6000 mg/kg.
Urinalysis was not significantly affected. In all treated animals, the
liver showed an increase in size accompanied by hypertrophy of the
hepatocytes. In the two highest-dose groups, the spleen was enlarged
and microscopic examination showed pronounced congestion and
haemorrhage. In the highest-dose group, slight or moderate
proliferation of the transitional cell epithelium was noted in the
urinary bladder.
7.2.1.3 Dog
In a study with chlordimeform by Blackmore (1969a), four groups
of beagle dogs were fed a dry diet containing either 0 mg/kg (10/sex),
250 mg/kg (8/sex), 500 mg/kg (8/sex) or 1000 mg/kg (10/sex) of
chlordimeform for 2 years. Two male and two female dogs were
sacrificed from each group at 26 and 52 weeks. Body weight was reduced
at 1000 mg/kg, the effect being slightly more pronounced in the
females. Total leucocyte counts were sporadically elevated in both
sexes at 1000 mg/kg and in females at 500 mg/kg. Haematocrit,
haemoglobin and erythrocyte counts tended to be depressed after 2
years in both sexes at 1000 mg/kg. Sporadic slight decreases in serum
albumin were observed, more frequently in males, at 1000 mg/kg.
Terminal spleen-to-body weight ratio was elevated in males at 500 and
1000 mg/kg, and in females at 1000 mg/kg. Histopathological
examinations revealed bile duct hyperplasia, pericholangitis and
nodular hepatocytic hyperplasia at 500 and 1000 mg/kg in both sexes,
and nodular hepatocytic hypertrophy at 1000 mg/kg in both sexes in the
liver. Kidneys showed an increased amount of pigmentation at 500 and
1000 mg/kg in both sexes.
7.2.2 Intubation
7.2.2.1 Rat
Four groups of 10 male and 10 female rats were intubated six
times weekly for one month with 5 ml/kg body weight of a 2% solution
of carboxymethylcellulose containing chlordimeform base at
concentrations such as to give dose levels of 0, 25, 50 or 100 mg/kg
(FAO/WHO, 1972). Body weight was markedly reduced in both sexes at
100 mg/kg. Hyperexcitability was observed in all test animals. At
100 mg/kg, this was apparent 20-30 min after dosing, and was followed
2 to 3 h after dosing by decreased activity and apathy. Recovery was
complete at 4 h. Similar but reduced effects were observed at 50 and
25 mg/kg, and with inconsistent frequency.
7.3 Long-term dietary exposure
7.3.1 Mouse
While there have been a number of long-term studies in mice with
chlordimeform and its metabolites, these were specifically designed to
study carcinogenic potential and are described in section 7.7.1.
7.3.2 Rat
In a study with chlordimeform by Blackmore (1969b), groups of
rats (35/sex/group) were fed a diet containing 0, 100, 250, 500 or
1000 mg/kg chlordimeform for 2 years. The 100 mg/kg group commenced
treatment 7 weeks after the other groups. This group was originally
part of the control group. Animals at that time were of similar weight
to those that had already been on test. The 1000 mg/kg group was
discontinued at 3 months due to severe growth inhibition. Growth
inhibition was observed in the males at 500 and 1000 mg/kg. In the
females, weight gain was reduced at 250 mg/kg and above. In addition,
female body weight gain was reduced at 100 mg/kg between weeks 20 and
48. Food intake was significantly reduced at 500 and 1000 mg/kg in
both sexes. Dose-related decreases in haematocrit, haemoglobin, and
erythrocyte counts, and a dose-related increase in the leucocyte count
occurred in females at 250 and 500 mg/kg up to one year. During the
second year, haematocrit only was consistently depressed in females at
500 mg/kg. Histopathological changes in the liver (nodules, and foci
of hyperplasia of hepatocytes) occurred in all groups, but the
incidence was greater at 250 and 500 mg/kg and was more severe at
500 mg/kg. Some females at 500 mg/kg showed slight hypertrophy and
vacuolation of focal groups of cells in the adrenal cortex. Terminally
organ to body weight ratios were increased in the liver (females at
250 and 500 mg/kg and males at 100 and 250 mg/kg), kidney (females at
250 and 500 mg/kg), thyroid (females at 250 and 500 mg/kg), heart
(males at 250 mg/kg and females at 500 mg/kg), adrenals (males at 100
and 250 mg/kg) and testes (100 and 500 mg/kg).
In a study with chlordimeform by Zak et al. (1973), groups of
rats (25/sex/group) were fed a diet containing chlordimeform at
concentrations of 0, 50, 75, 100, 250 and 500 mg/kg for one year. Food
consumption and weight gain data were recorded through the study.
Terminal organ weights and gross and microscopic examinations of
tissues and organs were the only other parameters reported. The
500 mg/kg group was terminated at 37 weeks after 10 males and 8
females had died. At the conclusion of the study, there was
considerable mortality noted in all groups. Food consumption was
decreased at 500 mg/kg in both sexes and was slightly reduced at
100 mg/kg and above in males only. This reduced food consumption was
not significantly reflected in the growth curves of males and females.
Gross examination did not show any compound-related abnormalities.
Organ weights and organ/body weight or organ/brain weight ratios did
not differ from control values. Histological examinations of liver and
spleen were performed on all animals. There were no significant
differences from control values with respect to fatty changes and
inflammatory changes in the liver. Slight proliferation of the bile
duct was more frequent in female rats treated with 500 mg/kg than in
the rats of other treated groups or the control group. Results of
examinations of the spleen for haemosiderosis suggested that, while
this condition was more pronounced in females, there were no
significant differences from control values.
In a study by Sachsse et al. (1980c), rats (90/sex/group;
Tif: RAIf strain) were fed a diet containing chlordimeform at
concentrations of 0, 2, 20, 100 or 500 mg/kg for 24 months. This was
equivalent to dosage levels of 0, 0.1, 1.0, 5.0 and 24 mg/kg body
weight per day for males and 0, 0.1, 1.2, 6.0, and 28 mg/kg body
weight per day for females. At the conclusion of the dietary feeding
study, all remaining rats were fed control diets for a period of time
until a survival rate of 20% per sex (10 rats) per group was attained,
at which time the animals were killed and examined. Groups of 20 male
and 20 female rats per group were examined periodically (4, 13, 26,
52, 78 and 104 weeks) for clinical laboratory investigations including
haematology, blood chemistry and urinalysis. Groups of 10 animals/sex/
group were sacrificed at 27 and 52 weeks for gross and microscopic
examination of tissues and organs. At the conclusion of the study, all
animals sacrificed (also those that died prior to the termination)
were examined for gross and microscopic pathology. There was no
mortality in the study attributable to the presence of chlordimeform.
Growth and body weight were maintained in all groups with the
exception of the 500 mg/kg group, where growth in both sexes was
slightly retarded. There were no clinical signs of toxicity or
abnormal behaviour. Ophthalmological and auditory examinations,
performed at periodic intervals, revealed no adverse effects
attributable to chlordimeform. Methaemoglobinaemia was observed at
dose levels of 20 mg/kg diet and above. At week 4, both males and
females showed a slight, but statistically significant, increase in
methaemoglobin content. At weeks 13 and 26, this condition abated but
returned at the end of one year and was significant in both sexes at
the highest dose level for the remainder of the study. Changes in
several other blood chemistry parameters were observed at the highest
dose level. Heinz body formation generally associated with
methaemoglobinaemia was not observed at week 4, but at the end of year
one and thereafter Heinz bodies were observed at the highest dose
level. A slight but significant reduction in blood glucose
concentration was noted at the higher dose levels throughout a major
part of the study. Slight changes in urinalysis parameters were
observed in the highest dose group, including a slightly reduced
urinary volume and a slightly higher specific gravity. Ketonuria and
proteinuria were observed at the high dose level at the earliest
examination periods only and were not observed at 13 weeks and
thereafter. Gross pathology and organ weight measurements (provided
for 27, 52 and 106-week sacrifice intervals) did not show any
significant dose-related responses. Microscopic histopathological
analyses of tissues and organs (performed at weeks 27 and 52 and at
the termination of the study) indicated no significant changes
attributable to chlordimeform in the diet. Although numerous benign
and malignant tumours were observed in both treated and control
animals, the frequency and type of neoplasms, reported at 12 and 24
months with pathology analyses, were not dose-related nor were they
attributable to chlordimeform in the diet. Several inherent,
degenerative or inflammatory changes were attributed to disease,
common in older animals. There was no indication of carcinogenic
potential to rats as a result of the presence of chlordimeform in the
diet. Based on the haematological occurrence of methaemoglobinaemia,
the no-observed-effect level of chlordimeform for rats was 2 mg/kg
diet, corresponding to an intake of 0.1 mg/kg body weight per day.
In a study with N-formyl-4-chloro- o-toluidine by Sachsse et
al. (1980d), groups of Tif:RAIf rats (90/sex/group) were fed a diet
containing N-formyl-4-chloro- o-toluidine at concentrations of
0, 2, 20, 100, or 500 mg/kg for 2 years. This corresponded to dietary
intakes of 0, 0.1, 1.0, 5 or 30 mg/kg body weight per day for females
and 0, 0.1, 1.0, 4.0 or 24 mg/kg body weight per day for males. Groups
of 10 males and 10 females were killed at periodic intervals (26 and
52 weeks) for examination of gross and microscopic pathology. Complete
haemato-logical, clinical chemistry, and urinalysis examinations were
performed at 4, 13, 26, 52 and 78 weeks on 20 males and 20 females of
each group. At 24 months, 20 males and 20 females were killed and
examined for clinical laboratory parameters and gross pathology. The
remaining animals were fed control diets for additional periods of
time until a survival rate of 20% per sex per group was attained.
At that time the remaining animals were killed and examined
microscopically for patho-logical changes, especially neoplastic and
non-neoplastic lesions. In the high-dose group, food intake and growth
were affected over the course of the study and slight growth
retardation was observed. Clinical signs of toxicity or adverse
behaviour were not observed. There was no mortality in the study
attributable to the presence of N-formyl-4-chloro- o-toluidine.
Ophthalmological examinations and auditory tests were normal. The
results of the haematological investigation showed haemoglobin
concentration to be slightly, but significantly, below that of the
controls in both male and female rats at the two highest dose levels.
In addition, slight but significant decreases in the erythrocyte count
and packed cell volume, a slight increase in reticulocytes and
somewhat higher methaemoglobin values were also seen in both male and
female rats at 500 mg/kg. With the exception of lower body weights of
the animals at the highest concentration, the most obvious change was
a significant increase in absolute and relative liver weights in both
sexes, but more pronounced in females, in the 500 mg/kg group. A
significantly increased incidence of hyperplasia of small biliary
ducts was seen in the liver of rats of the 500 mg/kg dose group. In
rats of the 500 mg/kg group that were killed after 2 years or died
after 12 months, a marked increase in the frequency of multioculated
cholangiogenic biliary cysts in the liver was noted. Both of these
finding were more pronounced and more frequent in female than in male
animals. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of neoplasms was
not treatment-related. All gross and histopathological lesions and
changes seen in both control and test animals were described as
inherent, degenerative or inflammatory in origin and were attributed
to naturally occurring diseases common in aged rats. There was no
indication of oncogenic potential in rats as a result of the presence
of N-formyl-4-chloro- o-toluidine. On the basis of the minor
haematological changes, the no-observed-effect level in this study was
20 mg/kg diet, corresponding to an intake of 1 mg/kg body weight per
day.
In a study with 4-chloro- o-toluidine by Sachsse et al. (1980e),
groups of Tif:RAIf rats (90/sex/dose level) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 or 500 mg/kg
for two years. This corresponded to dietary levels of 0, 0.1, 1.0, 5.0
or 28 mg/kg body weight per day for females, and 0, 0.1, 1.0, 4.6 or
24.6 mg/kg body weight per day for males. Groups of 10 males and 10
females were killed at periodic intervals (27 and 54 weeks) for gross
and microscopic pathological examinations. Complete haematological,
clinical chemistry and urinalysis examinations were performed at 4,
13, 26, 52, and 78 weeks on 20 females and 20 males of each group. At
24 months, 20 males and 20 females were killed and examined for
clinical laboratory parameters. Several animals were examined for
gross pathology. The remaining animals were fed control diets for
additional periods of time until a survival rate of 20% per group was
attained. At that time, the remaining animals were killed and examined
for microscopic pathology and oncogenic response. A complete
microscopic analysis was made on at least 10 rats of each sex of each
group at the termination of the experiment. All rats dying during the
course of the study were examined for tumours or neoplasms. In the
high-dose group of female rats, food intake and growth were affected
over the course of the study and slight growth retardation was
observed. There was no effect on growth in male rats at any dose
level. Clinical signs of toxicity were not observed. There was no
mortality in the study attributable to the presence of 4-chloro- o-
toluidine in the diet. Ophthalmological examinations and auditory
tests did not reveal changes that were related to the administration
of 4-chloro- o-toluidine. The results of the haematological
investigation, blood chemistry data and the urinalysis were similar
for both treated and control rats. Periodically, the haemoglobin
concentration was slightly but significantly below that of the
controls in the female rats at 100 mg/kg diet and above. Slight but
significant decreases were observed in the erythrocyte count and
packed cell volume in the female rats at 500 mg/kg. Marginal
reticulocytosis was also found to occur at 500 mg/kg in the female
rats at week 13 and in both sexes at week 26. In both male and female
rats at 500 mg/kg, the methaemoglobin level was found to be slightly
though significantly increased when compared to controls.
Periodically, this change was observed in the females of the 100 mg/kg
dose group, and, occasionally, Heinz bodies were also observed in
female rats. There were some changes to organ weights, organ-to-body
weight ratios and organ-to-brain ratios that were statistically
significant, but only the increase in absolute and relative liver
weights were dose-related. In rats from the 500 mg/kg dose group only,
a slightly but significantly increased incidence of multilobular
cholangiogenic cysts was observed in the liver. These biliary cysts
were found in 10/89 female and 3/90 male rats from the 500 mg/kg
group, compared to 4/89 female and 0/90 male rats in the control
group. Numerous benign and malignant tumours were observed in both
control and treated rats, but the frequency and types of the neoplasms
occurring in these animals was not treatment-related. Gross and
histopathological lesions and changes seen in both control and treated
animals were described as inherent, degenerative or inflammatory in
origin, and were attributed to naturally occurring diseases, common in
aged rats. There was no indication of oncogenic potential in rats as a
result of the presence of 4-chloro- o-toluidine in the diet. On the
basis of minor haematological changes, the no-observed-effect level in
this study was 20 mg/kg diet, corresponding to a dietary intake of
1 mg/kg body weight per day.
7.4 Skin and eye irritation; skin sensitization
Potential skin irritation was assessed by the application of
0.5 g chlordimeform or its hydrochloride salt to the shaved skin of
six male rabbits. When evaluated at 24 and 72 h, both compounds
produced a very slight irritation (FAO/WHO, 1972).
Potential eye irritation was assessed by application of 0.1 ml of
chlordimeform to one eye of each of nine rabbits, followed by
assessment over 7 days. All animals exhibited slight conjunctival
redness, while one showed slight chemosis. All effects had reversed
within 7 days. There was no evidence of corneal damage. Chlordimeform
may be considered a slight eye irritant (FAO/WHO, 1972).
There were no studies performed to assess the potential for
chlordimeform to cause skin sensitization.
7.5 Reproductive toxicity, embryotoxicity and teratogenicity
7.5.1 Reproductive toxicity
7.5.1.1 Rat
Four groups of 10 male and 20 female rats were fed a diet
containing 0, 100, 250 and 500 mg/kg chlordimeform in corn oil during
three parental and three two-litter filial generations. Parental body
weight prior to mating tended to be reduced in all treatment groups,
especially at the highest dose level. The same tendency was apparent
with regard to food consumption. The fertility index, gestation index,
live birth index, sex ratio, mean litter size and birth weight of pups
were comparable between treatment and control groups in all
generations. At the 500 mg/kg dose level, the lactation index was
reduced in Fla, Flb and F3a litters. Weaning weight of offspring was
depressed in all high-dose litters. Gross pathological examinations
were performed on parents and pups dying during the study, and on 10
male and 10 female weanlings of the F3b generation. No compound-
related effects were noted in the pathological examination (Blackmore,
1969c).
In a study by Goldman et al. (1991), treatment of ovariectomized
Long-Evans rats with a single intraperitoneal injection of
chlordimeform at dose levels of 25 or 50 mg/kg caused a complete
suppression of luteinizing hormone surge. The observed suppression did
not persist beyond the day of treatment. In a more recent study by
Cooper et al. (1994), the effect of this delay in hormone surge on
pregnancy outcome in females was examined. Chlordimeform at a dose
level of 50 mg/kg resulted in a delay in breeding as well as a
significant reduction in litter size.
Adult male Sprague-Dawley rats were administered chlordimeform by
gavage at 200 mg/kg body weight on one day or 50 mg/kg body weight per
day for 5 days. Rats were killed on either 3 or 13 days after the last
dose. Body weights were reduced at the earlier time points only. There
were no changes in the weights of the testes or associated organs.
Caudal sperm counts were reduced after the single dose only. No other
changes were observed, including sperm motility, velocity or
morphology (Linder et al., 1992).
7.5.1.2 Hamster
Goldman et al. (1993) reported that a single intraperitoneal dose
of chlordimeform (75 mg/kg and above) is capable of delaying the
luteinizing hormone surge and altering the timing of oocyte release in
the hamster. The reproduction consequences of this effect were not
investigated.
7.5.2 Embryotoxicity and teratology
7.5.2.1 Rat
Groups of pregnant rats (25/treatment group, 30 controls) were
administered chlordimeform in carboxymethylcellulose at dose levels of
0, 10, 25 or 50 mg/kg body weight per day from days 6 to 15 of
pregnancy. Only a slight reduction in feed intake was noted at the
intermediate dose level. At the high dose level, dams showed
somnolence through the first 3 days of treatment. There was also a
reduced body weight gain and decrease in feed consumption at this dose
level. Examination of fetuses removed by caesarean section on day 21
showed there was a slight delay in growth of the fetuses at the two
highest dose levels. This effect was probably a direct result of the
toxic response in the dams. No teratogenic events were observed in the
offspring, although an increased incidence of sternal ossification
defects occurred at 25 mg/kg body weight (Fritz, 1975).
7.5.2.2 Rabbit
Three groups of 10 impregnated female New Zealand white rabbits
were administered chlordimeform by gavage on days 8 to 16 of gestation
at dose levels of 0, 7.5 or 30 mg/kg body weight per day. Five rabbits
per group were killed on day 28 of gestation. Parental mortality,
abortion rate, corpora lutea to implantation ratio, litter size,
incidence of resorption, stillbirths, fetal weight, fetal length, and
incidence of skeletal and tissue abnormalities were unaffected by the
test compound. In the remaining rabbits, which were allowed to litter
normally, gestation length, litter size and litter weights were
similar in both treated and control groups (Blackmore, 1969d).
Groups of rabbits (group size ranged from 17 to 38 dams per
group) were given chlordimeform orally from days 6 to 18 of pregnancy
at dose levels of 0, 10, 30 and 100 mg/kg body weight per day. Fetuses
were removed by caesarean section on day 28 of pregnancy. The
administration of chlordimeform at 100 mg/kg body weight produced a
distinct adverse effect on dams for 2-3 h for the first 4 days of
treatment. Examination of dams and fetuses at 28 days suggested that
the low dose had no teratogenic or embryotoxic effect. In the
intermediate and high dose groups, the implantation to corpora lutea
ratio was found to be reduced compared to controls. In the high dose
group, the number of incompletely ossified sternebrae showed a slight
increase over that observed in the controls and in the other groups.
In addition, the number of fetuses with malformations was slightly
increased at 100 mg/kg. These malformations included a median cleft
palate and exencephaly and an omphalocele. Further examination of
spontaneous malformations observed in a cumulative control of 2495
rabbit fetuses suggested that these abnormalities may be spontaneous
and not a consequence of the administration of chlordimeform (Fritz,
1971).
7.6 Mutagenicity and related endpoints
Referenced summaries of the test results with chlordimeform,
N-formyl-4-chloro- o-toluidine, and 4-chloro- o-toluidine are
given in Tables 8, 9 and 10, respectively. The important features of
these data are given below.
7.6.1 DNA damage and repair
Chlordimeform gave no evidence of unscheduled DNA synthesis in
rat hepatocytes (dose levels: 5-625 �g/ml) or in human fibroblasts
(dose levels: 2-250 �g/ml). 4-Chloro- o-toluidine, on the other hand,
gave a slight to moderate dose-related increase in the mean number of
silver grains per nucleus in rat hepatocytes over a dose range of
0.625-78.15 �g/ml, but not in human fibroblasts over the dose range of
1.25-156.25 �g/ml.
Table 8. Summary of mutagenicity and related end-point studies on chlordimeform HC1
Organism Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976a);
TA100 +/-S9 - Konopka & Heymann (1977);
TA1535 +/-S9 - Muecke et al. (1979);
TA1537 +/-S9 - Rashid et al. (1984)
TA1538 +/-S9 -
Salmonella typhimurium TA98 - Arni & M�ller (1983a)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983c)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
WP67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Insects Sex-linked recessive lethals Drosophila +a,b Kale et al. (1995)
Mammalian cells Gene mutation Mouse lymphoma L5178Y- TK+/-/ - Beilstein & M�ller (1984a)
in vitro +/-S9
Unscheduled DNA synthesis Rat hepatocytes - Puri & M�ller (1983a)
Unscheduled DNA synthesis Human fibroblasts - Puri & M�ller (1983c)
Mammalian cells Cell transformation Mouse BALB/3T3 cells +a,b Beilstein & M�ller (1983)
in vitro
Table 8. (con't)
Organism Test Test system Strain +/- References
Mammals Testicular cell chromosome Mouse spermatocytes - Hool et al. (1983)
damage Mouse spermatocytes - Arni et al. (1983a)
Micronucleus assay Chinese Hamster bone - Langauer & M�ller (1977)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & M�ller (1978)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983a)
marrow cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz (1978a)
a Chlordimeform formulation
b Not dose-related
Table 9. Summary of mutagenicity and related end-point studies on N-formyl-4-chloro-o-toluidine
Organism Test Test Systems Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +/-S9 - Arni & M�ller (1976c);
TA100 +S9 + Konopka & Heymann (1977);
TA100 -S9 - Muecke et al. (1979);
TA1535 +/-S9 - Rashid et al. (1984)
TA1537 +/-S9 -
TA1538 +/-S9 -
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y +a Strasser & M�ller (1984b)
vitro
Mouse lymphoma L5178Y / -a Strasser & M�ller (1983b)
host-mediated assay
Mammals Testicular cell chromosome Mouse spermatogonia - Arni (1983b)
damage Mouse spermatocytes +b Arni & M�ller (1983e)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978a)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983f)
marrow metaphase cells
Table 9. (con't)
Organism Test Test Systems Strain +/- References
Mammals Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse - Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978b)
a No positive control
b Chromosome aberrations; not dose-related
Table 10. Summary of mutagenicity and related end-point studies on 4-chloro-o-toluidine
Organisms Test Test system Strain +/- References
Microorganisms Point mutation Salmonella typhimurium TA98 +S9 + Arni & M�ller (1976b);
TA98 +S9 - Haworth et al. (1983);
TA98 -S9 + Konopka & Heymann (1977);
Haworth et al. (1983);
TA100 +S9 - Meuke et al. (1979);
TA100 +S9 - Haworth et al. (1983)
TA100 -S9 - Rashid et al. (1984);
TA1535 +/-S9 - Haworth et al. (1983)
TA1537 +/-S9 -
TA1538 +/-S9 -
S. typhimurium TA98 - Arni & M�ller (1983b)
Intrasanguine host- TA100 -
mediated assay TA1535 -
Saccharomyces cerevisiae D7 +/-S9 - Arni & M�ller (1983d)
Escherichia coli WP2 +/-S9 - Rashid et al. (1984)
WP2uvrA +/-S9 -
Wp67 +/-S9 -
CM611 +/-S9 -
CM571 +/-S9 -
Mammalian cells in Gene mutation Mouse lymphoma L5178Y/TK+/- -S9 - Beilstein & M�ller (1984b)
vitro +S9 +
Mouse lymphoma L5178Y + Strasser & M�ller (1984a)
Mouse lymphoma L5178Y - Strasser & M�ller (1983a)
/host-mediated
Table 10. (con't)
Organisms Test Test system Strain +/- References
Mammalian cells in Unscheduled DNA synthesis Rat hepatocytes + Puri & M�ller (1983b)
vitro
Human fibroblasts - Puri & M�ller (1983d)
DNA strand breakage V79 cells + Zimmer et al. (1980)
Cell transformation Mouse BALB/3T3 cells + Beilstein & M�ller (1984c)
Mammals Testicular cell chromosome Mouse spermatogonia - Hool & Arni (1983b)
damage Mouse spermatocytes - Hool & Arni (1983c)
Micronucleus assay Chinese hamster bone - Langauer & M�ller (1978b)
marrow interphase cells
Chromosome aberrations Chinese hamster bone - Hool & Arni (1983d)
marrow metaphase cells
Sister chromatid exchange Chinese hamster bone - Hool & Arni (1983e)
marrow
Sister chromatid exchange Chinese hamster ovary + Galloway et al. (1987)
cells
Heritable translocation Mouse - Lang & Adler (1982)
Mammalian spot test Mouse + Lang (1984)
Dominant lethal Mouse - Fritz et al. (1978)
N-Formyl-4-chloro- o-toluidine was not directly tested for its
ability to induce DNA damage and repair.
The macromolecular binding of 4-chloro- o-toluidine to macro-
molecules of rat and mouse liver has been investigated by several
authors. In a report by Hill et al. (1979), the binding of
4-chloro-2-[methyl-14C]-methylaniline (4-chloro- o-toluidine)
in vivo and in vitro was investigated. The major binding
in vivo was in the liver. In vitro binding was dependent on
the presence for microsomal preparations and NADPH. Two soluble
products of microsomal enzymes were identified as 5-chloro-2-
hydroxylaminotoluene and 4,4'-dichloro-2,2'-dimethylazobenzene. The
hydroxylamino compound appeared to be the more activated form of
4-chloro- o-toluidine.
4-Chloro- o-toluidine caused DNA strand breaks in Chinese
hamster V79 cells (Zimmer et al., 1980).
In studies by Bentley et al. (1986a,b), the covalent binding
of [14C- ring]-4-chloro- o-toluidine to mouse and rat liver
macromolecules was compared. After a single administration to either
species, the extent of binding decreased in the order: protein>RNA>
DNA. The level of binding to mouse liver DNA was approximately twice
as high as the binding to rat liver DNA after either single or
repeated administration, while the binding to liver RNA and protein
was greater in the rat. There was no evidence of an increased level
of incorporation of [3H]-thymidine into DNA as a result of
4-chloro- o-toluidine binding. Two major hydrophobic DNA adducts
were formed in both species, and one of these was formed to a much
greater extent (6-30 fold) in mice.
7.6.2 Mutation
The ability of chlordimeform and its metabolites to induce
mutations has been investigated in both microbial and mammalian
systems.
Chlordimeform itself gave uniformly negative results in
Salmonella typhimurium (0.1-2000 mg/ml), Saccharomyces cerevisiae
(15-400 mg/ml), and Escherichia coli (250-2000 mg/ml), with or
without S9 microsomal activation, as well as in a thymidine kinase
mutation assay in mouse lymphoma L5178Y/TK+/- cells with
(75-3000 mg/ml) or without microsomal activation (42.5-1700 mg/ml).
Kale et al. (1995) reported that a chlordimeform formulation diluted
to provide a dose level of 10 000 �g/ml is a potent sex-linked
recessive mutagen in male pre-meiotic and meiotic cells of Drosophila.
N-Formyl-4-chloro- o-toluidine was also negative in all
Salmonella typhimurium assays (0.1-1000 �g/ml) except for TA100 with
microsomal activation, in which there was a dose-related increase in
revertants. All Escherichia coli assays (250-2000 �g/ml) were
negative. In a forward mutation assay in mouse lymphoma L5178Y cells
(213 & 640 �g/ml), N-formyl-4-chloro- o-toluidine gave a positive
result in two out of three 18-h incubation experiments. In a host-
mediated experiment with mouse lymphoma L5178Y cells (300 mg/kg), the
result was negative.
4-Chloro- o-toluidine was negative in all assays with
Salmonella typhimurium (10-2000 �g/ml) except for TA100 with S9
microsomal activation and TA98 with S9 microsomal activation. Assays
with Saccharomyces cerevisiae (3.2-90 �g/ml) and with Escherichia
coli (250-2000 �g/ml) were negative. In a thymidine kinase forward
mutation assay in mouse lymphoma L5178Y/TK+/- cells, 4-chloro- o-
toluidine was negative without S9 microsomal activation (31.25-
500 �g/ml) and positive with microsomal activation (37.5-600 �g/ml).
In a separate forward mutation assay in mouse lymphoma L5178Y cells
(111 & 255 �g/ml), a positive result was obtained in one out of three
18-h incubation experiments. In a host-mediated experiment with mouse
lymphoma L5178Y cells (330 mg/kg), the result was negative.
7.6.3 Chromosome damage
Sister chromatid exchange assays in Chinese hamster bone marrow
cells were conducted following oral treatment with chlordimeform
(31-324 mg/kg) and 4-chloro- o-toluidine (100-400 mg/kg). In both
cases, the result was negative. Similarly, in an assay for chromosome
aberrations in Chinese hamster bone marrow metaphase cells, a negative
result was obtained following oral treatment with chlordimeform
(2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine (2 � 300-
1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-800 mg/kg), although
the results were somewhat erratic. A micronucleus test in Chinese
hamster bone marrow interphase cells following oral treatment with
chlordimeform (2 � 60-240 mg/kg), N-formyl-4-chloro- o-toluidine
(2 � 300-1200 mg/kg) and 4-chloro- o-toluidine (2 � 100-400 mg/kg)
was also negative.
Testicular cell chromosomal damage was assessed in mouse
spermatocytes and spermatogonia. To investigate the potential
cytogenetic effects on mouse spermatogonia, chlordimeform
(9-66 mg/kg), N-formyl-4-chloro- o-toluidine (80-320 mg/kg) or
4-chloro- o-toluidine (85-500 mg/kg) was administered orally on 5
consecutive days. The results were negative in each case. To
investigate the potential cytogenetic effects on mouse spermatocytes,
chlordimeform (18-72 mg/kg), N-formyl-4-chloro- o-toluidine
(80-320 mg/kg), or 4-chloro- o-toluidine (85-500 mg/kg) was
administered orally over 10 days on days 0, 2, 3, 5 and 9. The results
were negative in the case of both chlordimeform and 4-chloro- o-
toluidine, but non-dose-related evidence of chromosome damage was
indicated from the results with N-formyl-4-chloro- o-toluidine.
The heritable translocation assay, dominant lethal test, and
mammalian spot test, each of which might indicate minor or major
genomic changes, were conducted on all three compounds. In the
heritable translocation assay, chlordimeform (120 mg/kg/day),
N-formyl-4-chloro- o-toluidine (100 mg/kg/day) or 4-chloro- o-
toluidine (200 mg/kg/day) was administered orally for 49 days. No
induction of translocation heterozygosity was found.
In the dominant lethal assay, chlordimeform (22 or 66 mg/kg),
N-formyl-4-chloro- o-toluidine (105 or 315 mg/kg) or 4-chloro- o-
toluidine (110 or 330 mg/kg) was administered orally as a single dose.
There was no evidence of any dominant lethal effects in the progeny of
male mice.
In the mammalian spot test, chlordimeform (160 mg/kg),
N-formyl-4-chloro- o-toluidine (100 mg/kg) or 4-chloro- o-
toluidine (100 mg/kg) was administered orally on days 8-10 of
embryonic development. The results were negative except in the case of
4-chloro- o-toluidine, which induced a 3.2% incidence of spots of
genetic relevance compared to an incidence of 0.9% in controls.
7.6.4 Cell transformation
Cell transformation assays conducted with both chlordimeform and
4-chloro-toluidine in mouse BALB/3T3 cells produced an increased
incidence of transformed cell colonies with both compounds. With
chlordimeform, the experiment was conducted at dose levels up to
1 �g/ml, and increased transformation frequency was observed only at
0.0625 and 0.125 �g/ml. The transformative properties of chlordimeform
were considered weak. With 4-chloro- o-toluidine, three experiments
were conducted at dose levels up to 36 �g/ml, and a significant
increase in transformation frequency was observed. The transformative
properties of 4-chloro- o-toluidine were considered definite.
7.7 Carcinogenicity
A number of carcinogenicity studies have been conducted in mice.
With chlordimeform, there are two dietary studies and one dermal
study. With N-formyl-4-chloro- o-toluidine, there is one dietary
study. With 4-chloro- o-toluidine, there are four dietary studies.
In rats, the carcinogenic potential of chlordimeform and its
metabolites was generally investigated as part of more detailed
long-term studies, and details are provided in section 7.3.1.2. Three
studies on 4-chloro- o-toluidine that primarily investigated
carcinogenicity are reported below.
7.7.1 Mouse
The carcinogenicity of chlordimeform has been examined in two
dietary studies and in one dermal study.
In a study by Suter et al. (1978), groups of mice (50/sex/group;
Tif: MAG strain, SPF derived) were fed a diet containing chlordimeform
at concentrations of 0, 20, 100 and 500 mg/kg for 24 months. At the
conclusion of the dietary feeding interval, animals were maintained on
control diet until 90% of a group had died, at which time the
remaining animals of the group were sacrificed. There were no signs of
acute toxicity related to chlordimeform in the diet over the course of
the feeding trial. Growth and food consumption were similarly
unaffected by the presence of chlordimeform in the diet. Mortality was
significantly increased in females after 60 weeks at 500 mg/kg, and
after 90 weeks at both 100 and 500 mg/kg. In males, significantly
increased mortality was observed after 70 weeks at 500 mg/kg, and
after 110 weeks at both 100 and 500 mg/kg. However, lifespan was not
significantly affected in males at 100 mg/kg. The animals fed dietary
levels of 100 mg/kg and above displayed an increased incidence of
haemorrhagic tissue masses in subcutaneous tissues, retroperitoneum
and some internal organs (kidney, liver and spleen), which upon
examination were classified as malignant haemangioendotheliomas. These
malignancies which were reported to occur rarely in control
populations were found predominantly in the 100 and 500 mg/kg dietary
groups (see Table 11). In some animals the tumours were of multiple
origin and metastases were observed in the lungs. There were no other
types of neoplasm observed in the study that were attributable to
chlordimeform in the diet. Under the conditions of this study,
20 mg/kg in the diet appeared to be a no-observed-effect level.
In a study by Li et al. (1985a), groups of Swiss mice (50/group,
sex not stated) were fed a diet containing chlordimeform at
concentrations of 0, 20, 100, or 300 mg/kg for a period of 18 months.
A positive control group was administered 300 mg/kg of 4-chloro- o-
toluidine in the diet for 18 months. All animals were killed at the
end of the study and assessed for tumour formation. The main results
of the study are presented in Table 12. The author described the
majority of the neoplasms as angiomas, and the malignant neoplasms as
angiosarcomas. These neoplastic changes were considered to be similar
to those observed in the study by Suter et al. (1978).
In a paper by Jiang et al. (1988), the dermal carcinogenicity of
chlordimeform was investigated in mice. Groups of Swiss mice (50 per
dose level, sex not stated) were treated dermally with chlordimeform
twice per week at dose levels of 0, 100, 500, 2000 or 4000 mg/kg body
weight for a total of 17-20 treatments, together with croton oil (0.5%
in acetone). Positive controls received coal tar pitch (20 treatments)
plus croton oil (30 treatments). All animals were sacrificed after 18
months and analysed for tumour formation. Chlordimeform induced both
skin and liver tumours in this assay (see Table 13). The changes
observed in the skin consisted of epidermal hyperplasia, papillomas
and squamous cell carcinomas. The effect of croton oil application was
Table 11. Incidence of haemangioendotheliomas in mice following dietary administration
of chlordimeform, N-formyl-4-chloro-o-toluidine or 4-chloro-o-toluidine
(Suter et al., 1978; Sachsse et al., 1978a,b)
Control Dietary concentration (mg/kg)
2 20 100 500
Chlordimeform HCl
Male 1/44 (2%) - 0/44 (0%) 15/49 (30%) 39/48 (83%)
Female 1/43 (2%) - 2/46 (4%) 22/46 (50%) 35/49 (71%)
Total 2/87 (2%) - 2/90 (2%) 37/95 (41%) 74/97 (80%)
N-formyl-4-chloro-o-toluidine
Male 0/46 (2%) - 0/49 (0%) 15/48 (38%) 40/47 (89%)
Female 0/47 (0%) - 0/47 (0%) 23/43 (56%) 38/48 (79%)
Total 1/93 (1%) - 0/96 (0%) 38/91 (47%) 78/95 (84%)
4-Chloro-o-toluidine
Male 0/50 (0%) 0/47 (0%) 4/48 (8%) 23/47 (48%) 40/48 (83%)
Female 1/45 (7%) 1/45 (2%) 3/48 (6%) 30/47 (62%) 34/49 (72%)
Total 1/95 (1%) 1/92 (1%) 7/96 (19%) 53/94 (66%) 74/97 (78%)
to shorten the latent period for tumour formation and also to hasten
the malignant progression of existing neoplasms in the skin. At
500 mg/kg body weight, the time of first appearance of tumours was 483
days without croton oil and 154 days with croton oil. The latency
period also decreased with increasing dose levels of chlordimeform. In
the liver, changes consisted of enlargement, hepatocytic hyperplasia,
and hepatocytic carcinomas.
In a carcinogenicity study by Sachsse et al. (1978a), groups of
mice (50/sex/group; Tif: MAG strain) were fed N-formyl-4-chloro- o-
toluidine in the diet at concentrations of 0, 20, 100 and 500 mg/kg
for 24 months. After this time, all animals were fed a control diet
until the study was concluded when 90% of the animals in a group had
Table 12. Incidence of tumours in mice after dietary administration of chlordimeform or 4-chloro-o-toluidine (Li et al., 1985a)
Dietary Number of animals Number of animals Incidence Number of animals Incidence Days to
concentration necropsied bearing haemangiomas (%) bearing (%) appearance of
(mg/kg diet) or haemangiosarcomas haemangiosarcomas neoplasm
Chlordimeform
0 50 0 0 0 0 -
20 50 8 16 0 0 494
100 50 22 44 5 10 469
300 50 36 72 15 30 448
4-Chloro-o-toluidine
300 50 31 62 13 26 283
Table 13. Incidence of tumours in mice following dermal application of chlordimeform (Jiang et al., 1988)
Group/treatment Skin Liver
Number Carcinomas Papillomas Hyperplasia Number Carcinomas Papillomas Hyperplasia
of animals (%) (%) (%) of animals (%) (%) (%)
Water 18 0.0 0.0 6.1 17 0.0 0.0 0.0
Croton oil alone 17 0.0 0.0 17.6 24 0.0 0.0 8.3
100 mg/kg + croton oil 19 0.0 5.3 21.1 21 23.8 0.0 9.5
500 mg/kg 22 4.6 4.6 18.2 20 25.0 0.0 0.0
chlordimeform alone
500 mg/kg + croton oil 23 4.4 4.4 52.2 25 8.0 0.0 4.0
2000 mg/kg + croton oil 15 20.0 20.0 26.7 14 14.3 0.0 0.0
4000 mg/kg + croton oil 15 60.0 13.3 13.3 16 18.8 6.2 0.0
Coal tar pitch 18 88.9 11.1 0.0 19 15.8 5.3 0.0
died. There was no sign of adverse behaviour, and acute mortality was
not noted. Growth and food consumption were unaffected. There were
significant differences noted in survival after one year of age. Both
males and females showed an increased mortality at 100 and 500 mg/kg
after approximately one year of feeding. The onset of increased
mortality occurred earlier in females. The females at the 20 mg/kg
dietary level showed a slightly higher, non-significant, mortality
during the same period. Detailed gross and microscopic examination of
a variety of tissues and organs showed the presence of numerous gross
anatomical lesions. There was an increased number of haemorrhagic
masses in the subcutaneous tissues in the retroperitoneum and in some
internal organs of mice at all treatment levels. Detailed microscopic
examination confirmed that the increased incidence of haemorrhagic
masses were malignant tumours of vascular origin. These tumours were
histologically classified as malignant haemangioendotheliomas (see
Table 11). In addition to the occurrence of tumours, the time to
tumour relationship was decreased as the dietary concentration was
increased. Other neoplasms occurring in the study were not considered
to be treatment-related. A no-observed-effect-level was not
demonstrated under the condition of this experiment.
The carcinogenicity of 4-chloro -o-toluidine has been examined
in four dietary studies.
An early study by Ezumi and Nakao conducted in 1974 was reviewed
by the JMPR in 1978 and considered inadequate (FAO/WHO, 1979).
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives described by Homburger et al. (1972) and Weisburger
et al. (1978), groups of CD-1 mice (25/sex/dose level) were
administered 4-chloro- o-toluidine in the diet at dose levels of 0,
750 or 1500 mg/kg for males, and 0, 2000 or 4000 mg/kg for females for
a period of 18 months. All mice were placed on a control diet for an
additional 3 months before sacrifice and complete necropsy and
histopathological examination of tissues. The incidence of
haemangioendotheliomas was increased in males at both low (12/20) and
high (13/20) dose levels compared to concurrent controls (0/14) and
historical controls (5/99), and in females at both low (18/19) and
high (12/16) dose levels compared to concurrent controls (0/15) and
historical controls (9/102).
In a study by Sachsse et al. (1978b), groups of mice
(50/sex/group; Tif: MAGf strain) were fed a diet containing
4-chloro- o-toluidine at concentrations of 0, 2, 20, 100 and
500 mg/kg for 24 months. After 24 months, all animals were fed control
diets until the study was concluded when 90% of the animals in a group
had died. There were no overt signs of toxicity. Growth and food
consumption were unaffected by treatment. An adverse effect on
longevity (lifespan) was noted in both males and females at the two
highest dietary levels. At the conclusion of the study upon gross
examination there was a marked increase number of haemorrhagic masses
in subcutaneous tissue, in the retroperitoneum, and in some internal
organs. Microscopic examination revealed an increased incidence of
haemorrhagic malignant tumours of vascular origin at dose levels of
20 mg/kg and above (see Table 11). The tumour incidence in control
exceeded the incidence observed at 2 mg/kg. The tumours were
histologically classified as malignant haemangioendotheliomas and, on
occasion, metastases were observed. There was not only a significant
dose-dependent increase in the total incidence of malignant tumours
but the time to appearance of tumours occurred at a markedly earlier
date in animals at the higher concentrations than in those at the
lower concentrations. A benign variant of the haemangioma was observed
in all groups, and although without the characteristics of malignancy,
did cause local invasion. Thus, the benign and malignant tumours have
been grouped together. The vascular tumours (haemangiomas and
haemangioendotheliomas) of the type that occurred in the mice appeared
to be peculiar to this rodent species. The occurrence of other types
of neoplasms in the study was not influenced by the presence of
4-chloro- o-toluidine in the diet. Under the conditions of this
experiment, 2 mg/kg in the diet appeared to be a no-observed-effect
level.
In a study with 4-chloro- o-toluidine conducted by the National
Cancer Institute (NCI, 1979), groups of B6C3F1 mice (50/sex/dose
level) were administered 4-chloro -o-toluidine in the diet at dose
levels of 3750 or 15 000 mg/kg for males and 1250 or 5000 mg/kg for
females for 99 weeks. Control groups consisted of 20 males and 20
females. There was a dose-related increase in mortality in both sexes.
There was also a dose-related increase in the incidence of
haemangiosarcomas as shown in Table 14. The haemangiosarcomas
apparently originated in fatty tissue adjacent to the genital organs
and not in a particular organ. In some instances, they were observed
to infiltrate the abdominal muscles, uterus, ovary, prostate or
urinary bladder. The haemangiosarcomas were lethal to 89 (75%) of the
affected mice, owing to haemorrhage in the peritoneal cavity and to
the space-consuming character of the lesions. Pulmonary metastasis was
observed in only 5 (4%) of the 119 dosed animals bearing
haemangiosarcomas. Associated pathological alterations that were
recorded at necropsy were haemorrhage in the peritoneal cavity and
variable enlargement of the spleen. It was concluded that 4-chloro-
o-toluidine was carcinogenic in B6C3F1 mice.
In the study of Li et al. (1985a), a single dietary dose of
4-chloro- o-toluidine (300 mg/kg) was given to mice for 18 months as
a positive control. The incidence of tumours was similar to that seen
in mice receiving 300 mg/kg of chlordimeform, but the latency period
was considerably reduced (Table 12).
Table 14. Incidence of tumours in mice following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 3750 15 000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 20 50 50 18 49 50
Haemangiosarcomas 0 3 37 0 40 39
(0%) (6%) (74%) (0%) (82%) (78%)
Haemangioma 0 3 5 1 6 0
(0%) (6%) (10%) (6%) (12%) (0%)
7.7.2 Rat
The carcinogenicity of chlordimeform and its metabolites has
generally been considered as part of more detailed long-term studies
(see Section 7.3). In the studies below, carcinogenicity was the
primary consideration.
In a study conducted by the National Cancer Institute (NCI,
1979), groups of F344 rats (50/sex/dose level) were fed a diet
containing 4-chloro- o-toluidine at concentrations of 1250 mg/kg or
5000 mg/kg for 107 weeks. Control groups contained 20 animals of each
sex. There was no significant dose-related trend in mortality in
either sex. There was a variety of neoplastic and non-neoplastic
changes in control and treated rats. There was a small increase in
adenomas of chromophobe cells of the pituitary gland in both male and
female rats compared to controls (see Table 15). All of these tumours
were benign, are also common in this strain of rat and have occurred
in 21% of control female rats in the NCI laboratories. The authors
concluded that on the basis of histopathological examination,
4-chloro- o-toluidine was not carcinogenic in F344 rats.
In a large study on the carcinogenicity of 21 aromatic amines and
their derivatives by Weisburger et al. (1978), groups of male Charles
River CD rats were administered 4-chloro- o-toluidine in the diet at
dose levels of 0, 2000 or 4000 mg/kg diet for the first 3 months,
which was then reduced to 0, 500 or 1000 mg/kg diet for the following
15 months. There was no statistically significant increase in tumours
in either of the treated groups.
Table 15. Incidence of tumours in rats following dietary
administration of 4-chloro-o-toluidine (NCI, 1979)
Male Female
Control 1250 5000 Control 1250 5000
mg/kg mg/kg mg/kg mg/kg
Number of animals 19 48 47 19 48 48
Chromophobe 2 6 15 1 13 15
adenoma (11%) (13%) (32%) (5%) (27%) (31%)
Chromophobe 0 0 2 0 3 1
hyperplasia (0%) (0%) (4%) (0%) (6%) (2%)
7.8 Other special studies
7.8.1 Immunotoxicity
In a study by Wiltrout et al. (1978), the potential of various
pesticides to influence the primary humoral immune response in the
mouse with respect to both dose and time of exposure was examined.
Mice receiving a single oral dose of chlordimeform at approximately
the LD50 level (148 mg/kg body weight) experienced a significant
suppression of humoral response when the dose was administered on the
day of immunization or two days after immunization. No response was
observed at one tenth of the LD50 dose, even when administered for
8 or 28 days.
Further studies by Shopp et al. (1985) investigated the effect of
chlordimeform on both humoral and cell-mediated immunity in the mouse
following both acute and 14-day exposures by the intraperitoneal
route. There was a decrease in IgM antibody-forming (plaque-forming)
cells when measured 4 days after intraperitoneal administration at 20
or 40 mg/kg body weight per day. These dose levels did not result in
any alteration of cell-mediated immunity. When administered orally,
chlordimeform at doses as high as 120 mg/kg body weight per day did
not have any effect on the 4- or 5-day antibody response.
Immunological parameters that may be related to the carcinogenic
activity of chlordimeform in rats were investigated by Thomas et al.
(1990). These included spleen/body weight ratio, splenocyte viability,
T and B cell mitogenesis, natural killer (NK) cell and natural
cytotoxic (NC) cell activity. Chlordimeform was administered
intraperitoneally on three consecutive days at 0, 1, 10 or 75 mg/kg
body weight per day. 4-Chloro- o-toluidine was administered
intraperitoneally on three consecutive days at 0, 10, 50 or 100 mg/kg
body weight per day. Spleen/body weight changes were observed only at
the highest dose of chlordimeform. No changes were observed with
either chemical on splenocyte viability or T and B cell mitogenesis.
An inhibition of NC activity at all chlordimeform doses was observed,
and an inhibition of NK activity was observed at 10 mg/kg body weight
per day and above. The relevance of this result to the carcinogenic
activity of these chemicals is doubtful.
7.8.2 Behavioural effects
Behavioural studies of the effects of chlordimeform in rats were
first investigated by Olson et al. (1978). The effects of exposure
prenatally and post-natally were examined following a dietary intake
of 0.1 mg/kg body weight per day. Early development testing (swimming
and righting reflex) was conducted on rat pups from post-natal days 7
to 17, while motivational, learning and retention tests were conducted
on days 70 to 90. The most significant differences between control and
treated groups was in the swimming task, retarded maturation being
observed in the chlordimeform-fed group. There was no treatment-
related effect with regard to maze tests or with regard to the tests
of motivation.
Moser et al. (1988) examined the behaviour of rats using a
functional observation battery following a single oral administration
of chlordimeform at dose levels of 0, 1, 25 or 56 mg/kg body weight.
Rats were examined at 1, 5 or 24 h. Chlordimeform produced a decrease
in body weight as well as a decrease in body temperature. There was a
dose-related increase in general activity, CNS excitability and
sensory responsiveness, coupled with a decrease in rearing, gait and
arousal. Chlordimeform also produced an increase in grip strength.
Other behavioural effects observed with chlordimeform have
included appetite stimulation in rats (Pfister et al., 1978b), flavour
aversion in both rats (MacPhail & Leander, (1980) and mice (Leander et
al., 1984) and alteration in schedule-controlled performance in rats
(MacPhail & Leander, 1981), mice (Glowa, 1986) and pigeons (Leander &
MacPhail, 1980). Witkin & Leander (1982) also demonstrated that, while
causing appetite stimulation in rats, chlordimeform produced a dose-
related decrease in water consumption, in contrast to other appetite
stimulants.
7.8.3 Pharmacological and biochemical effects
The pharmacological and biochemical effects of chlordimeform in
animals have been reviewed by Knowles (1991).
The cardiovascular effects of chlordimeform treatment were
recognized from an early stage with the observation that chlordimeform
administered intraperitoneally to rabbits caused a marked decrease in
arterial blood pressure of almost 50% within 30 min of treatment
(Matsumura & Beeman, 1976). Cardiovascular changes were also noted in
the dog (Lund et al., 1979a,b; Rieger et al., 1981) but in this case
the effect was biphasic, consisting of an initial depressor response
associated with decreased cardiac contractility and vascular
resistance, and a secondary pressor response associated with increased
cardiac contractility and vascular resistance. These actions of
chlordimeform were noted to be similar to those of local anaesthetics
such as procaine and lidocaine (Pfister et al., 1978a; Lund et al.,
1979a,b,c).
In studies by Watkinson (1985, 1986a,b), the effects of
chlordimeform on cardiovascular functional parameters were examined in
post-weaning and geriatric rats following intravenous treatment at
dose levels up to 60 and 120 mg/kg body weight, respectively, or
intraperitoneal treatment of post-weaning rats at dose levels up to
60 mg/kg body weight. Chlordimeform produced profound and abrupt
decreases in heart rate and blood pressure within 3 min, together with
multiple arrhythmias and alterations in electrocardiogram waveforms
and intervals. The effects observed in post-weaning rats were less
severe than those observed in geriatric rats.
The inhibition of monoamine oxidase in rats in vivo and
in vitro by chlordimeform and/or its metabolites has been
extensively studied (Beeman & Matsumura, 1973; Maitre et al., 1978;
Benezet et al., 1978; Hollingworth et al., 1979; Kadir & Knowles,
1981; Kaloyanova et al., 1981; Bailey et al., 1982). The lack of
correlation of toxicity of chlordimeform metabolites to monoamine
oxidase inhibition and the fact that chlordimeform is a relatively
weak monoamine oxidase inhibitor suggest that monoamine oxidase
inhibition is not the primary factor involved in the acute toxicity of
chlordimeform (Neumann & Voss, 1977; Robinson & Smith, 1977;
Hollingworth et al., 1979).
Chlordimeform also has an effect on the level of biogenic amines
in brain and plasma of rats, which may in part at least be due to
the inhibition of monoamine oxidase levels. Administration of
chlordimeform to rats was found to produce an increase of 25-70% in
5-hydroxytryptamine, norepinephrine or dopamine levels in brain
(Maitre et al., 1978; Benezet et al., 1978; Bailey et al., 1982).
However, Johnson & Knowles (1983) treated rats subcutaneously with
chlordimeform (200 mg/kg body weight) and found no change in any of
the amines.
Chlordimeform and some of its metabolites have been shown to
affect platelet function, as measured by the uptake of radioactive
5-hydroxytryptamine (Knowles, 1991).
Chlordimeform also has antipyretic and anti-inflammatory actions,
as shown by its ability to reduce yeast-induced fever in rats. It also
antagonizes both early (5-hydroxytryptamine- and histamine-mediated)
and late (prostaglandin-mediated) phases of carrageenan-induced
hind-paw oedema, albumin-induced oedema, and oedema induced by direct
injection of 5-hydroxytryptamine and histamine (Yim et al., 1978).
Chlordimeform also induced mild gastric ulceration in rats after
intraperitoneal injection (20-80 mg/kg body weight) but not after oral
treatment (80-240 mg/kg body weight). The above actions may be related
to the ability of chlordimeform to inhibit prostaglandin biosynthesis
(Yim et al., 1978; Holsapple & Yim, 1981).
Chlordimeform induces hypothermia in rats (Watkinson & Gordon,
1987) and mice (Gordon et al., 1985). Watkinson et al. (1989)
examined the effect of core body temperature on both the survival
and cardiovascular functions of rats following treatment with
chlordimeform. The results indicated that at a given dose of
chlordimeform, the magnitude and duration of the observed toxic
effects are primarily a function of core body temperature. The authors
concluded that moderate hypothermia, but not extreme hypothermia, may
have a beneficial effect with respect to survival after exposure to
chlordimeform.
Chlordimeform has been shown to have an effect on both visual and
auditory functions in mammals. Intraperitoneal treatment of male rats
with acute dosages of chlordimeform (5-40 mg/kg body weight) before
testing revealed a temporary increase in both the amplitude and
latency of pattern reversal-evoked potentials and an increase only in
the latency of pattern flash-evoked potentials (Dyer & Boyes, 1983;
Boyes & Dyer, 1984). Boyes & Moser (1988) provided evidence to support
the hypothesis that these effects are evoked through actions as a
central nervous system alpha-adrenegic agonist. Janssen et al. (1983)
demonstrated effects on the brain stem auditory-evoked response after
injection of chlordimeform at a dose levels of 40 mg/kg body weight.
It has been suggested that these effects may by secondary to the
hypothermic effects induced by chlordimeform (Gordon et al., 1985).
Chlordimeform has been shown to affect the activity of hepatic
drug-metabolizing enzymes in both rats and mice. Studies have been
conducted following gastric intubation at dose levels up to 150 mg/kg
body weight per day for 7 days, and also following intraperitoneal
injections either singly (100 mg/kg body weight) or daily (75 mg/kg
body weight per day) for 4 days. Chlordimeform treatment induced
several of these hepatic drug-metabolizing enzymes with significant
species and/or sex specificity. Cytochrome P-450 content was increased
in all cases.
7.9 Factors modifying toxicity
The factors modifying the acute toxicity of chlordimeform have
been reviewed by Knowles (1991).
7.10 Mechanisms of toxicity - mode of action
7.10.1 Mechanism of acute toxicity
A large number of studies that investigated the mechanism of
action following acute poisoning with chlordimeform have been
reported.
Based on the early in vitro and in vivo studies of Aziz &
Knowles (1973) and Beeman & Matsumura (1973), it was suggested that
the increase in biogenic amines resulting from inhibition of monoamine
oxidase by chlordimeform could account for the variety of toxic signs
following acute poisoning. However, Maitre & Gfeller (1975) and
Robinson et al. (1975) demonstrated that this mechanism does not play
a significant role in the acute toxicity in rats.
A number of other studies have attempted to define the mode of
action of chlordimeform. Studies in insects have shown that
chlordimeform has little activity on cholinergic transmission although
it is an uncoupler of oxidative phosphorylation and an inhibitor of
electron transport (Abo-Khatwa & Hollingworth, 1972a). A number of
biochemical mechanisms have been postulated to explain the effects of
chlordimeform in insects, including uncoupling of respiration
(Abo-Khatwa & Hollingworth, 1972a,b), inhibition of monoamine oxidase
(Knowles & Roulston, 1972) and blockage of neuromuscular transmission
(Wang et al., 1975; Watanabe et al., 1975), and motor stimulation
through actions on central non-cholinergic synapses (Lund et al.,
1979a; Lund et al., 1979c). The latter effect is thought to be
mediated through the neurotransmitter, octopamine (Lund et al.,
1979b). Both chlordimeform and particularly demethylchlordimeform have
been shown to interact with the octopamine receptor and partially
mimic the pharmacological effects of octopamine (Evans & Gee, 1980;
Nathanson & Hunnicutt, 1981; Bokisch et al., 1985).
In mammalian systems, oxidative phosphorylation is uncoupled
(Abo-Khatwa & Hollingworth, 1972b) and RNA synthesis is inhibited by
chlordimeform, but only at very high concentrations (Murakami &
Fukami, 1974). The effects of chlordimeform on hepatic drug-
metabolizing enzymes was dependent on both sex and species and did not
show any particular pattern that would indicate a consistent mechanism
of action (Budris et al., 1983; Bentley et al., 1985; Leslie et al.,
1988).
Chlordimeform, acting as a direct depressant on cardiac and
vascular muscle, induced a hypotensive state in dogs. Chlordimeform
did not interfere with the autonomic nervous system. The mechanism of
cardiovascular depression may be related to that noted with frog nerve
preparations treated with procaine, a local anaesthetic. The
metabolite, 4-chloro- o-toluidine has been shown to interfere with
rat cardiac receptors (Wang et al., 1975; Watanabe et al., 1975;
Matsumura & Beeman, 1976; Knowles, 1976; Hollingworth, 1976; Lund et
al., 1978a).
More recent research has shown that formamidine pesticides may
exert their effects on the central nervous system by interacting
directly with adrenergic receptors, particularly the alpha-2 subtype
(Costa & Murphy, 1987; Costa et al., 1988, 1989). This interaction
appears to mediate several of the observed effects of formamidines,
such as changes in heart rate (Hsu & Kakuk, 1984, Watkinson, 1985;
1986a,b), pupil diameter (Hsu & Kakuk, 1984), visual evoked potential
(Boyes & Moser, 1988) and hormonal secretion (Goldman et al., 1990;
1991). Costa et al. (1991) demonstrated that chlordimeform
decreases the hepatic glutathione content by up to 40% in a
time- and dose-dependent manner, through an interaction with
alpha2-adrenoreceptors. Wu et al. (1990) have demonstrated that
chlordimeform inhibits the binding of the known alpha2-adrenoreceptor
blockers, clonidine and yohimbine, in rat forebrain tissue
in vitro. Furthermore, Stoker et al. (1991), in a further study on
the effects of chlordimeform on hormone release, have demonstrated in
rats, treated intraperitoneally with chlordimeform (20 or 50 mg/kg
body weight), that there is an increase in adrenocorticotropic hormone
(ACTH), circulating corticosteroid (CORT) and prolactin (PL) in a
dose-dependent manner. alpha-Adrenergic agonists specifically
inhibited these effects indicating the interference with a regulatory
signal mediated by alpha-adrenergic receptor-associated activity.
Candura et al. (1992) demonstrated that the inhibition induced by
chlordimeform in the intestinal tract is mediated by calcium channel
blockade rather than by alpha2-adrenoceptor activation. In a study by
Robinson et al. (1975), it was found that using drugs to block the
serotonergic or alpha-adrenergic receptors did not reduce the
chlordimeform-induced lethality in male rats.
7.10.2 Mechanism of carcinogenicity
Chlordimeform and its metabolites, N-formyl-4-chloro- o-
toluidine and 4-chloro -o-toluidine, have been shown to induce mouse
tumours of a vascular origin characterized histologically as
haemangioendotheliomas and haemangiosarcomas. 4-Chloro- o-toluidine
has been shown to be a more potent carcinogen than chlordimeform, both
with respect to dose-response and to a reduced latency period.
Haemangioendotheliomas and haemangiosarcomas were not induced in rats.
Cases of bladder cancer in humans associated with occupational
exposure to high levels of chlordimeform or 4-chloro- o-toluidine
have been seen in groups with high urinary levels of chlordimeform and
4-chloro- o-toluidine.
The exact mechanism of induction of these tumours is unknown but
there is evidence that a genetic mechanism involving mutations induced
by 4-chloro- o-toluidine is involved.
Metabolic studies in mice and rats indicate a similar metabolic
pathway for chlordimeform in both species. The kinetics of absorption
and elimination in mice and rats also seem to be similar. However, the
overall DNA binding was higher in mice than rats, and one DNA adduct
was formed to a 6- to 30-fold higher extent in mice.
There is considerable evidence that 4-chloro- o-toluidine causes
severe toxic effects in the human bladder leading to haemorrhagic
cystitis (see section 8). Monitoring of urinary metabolites in humans
also indicates that chlordimeform is rapidly metabolized to 4-chloro-
o-toluidine in vivo.
4-Chloro- o-toluidine also has a close structural similarity
to aromatic amines for which there is established evidence of
carcinogenicity by animal experimentation and also by human
epidemiological data (Parkes, 1984).
Taken together, the evidence strongly implicates 4-chloro- o-
toluidine as the causative agent in the induction of tumours in both
mice and humans. A proposed route of activation that may be associated
with carcinogenicity is shown in Fig. 2. However, the mechanism of the
carcinogenicity remains unclear.
 8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Acute poisoning incidents
The most comprehensive data on acute poisoning cases associated
with exposure to chlordimeform has come from China. Details of these
published poisoning cases are shown in Table 16. While many were due
to intentional ingestion, there were also cases of unintentional
poisoning as a result of consumption of contaminated food, as well as
occupational exposure to the spray. In a brief report prepared by Deng
et al. (1984) of a 1983 symposium in Hu-bei Province on chlordimeform
poisoning, which featured some 29 papers and 859 case studies, it was
stressed that the main cause of death was suppression of cardiac
contracture and dilation of blood vessels resulting in circulatory
failure.
Arima et al. (1976) described an unsuccessful suicide attempt
involving a 76-year-old male who ingested 100 g chlordimeform. He
vomited several times before arriving at hospital 50 min after
ingestion. He was lethargic with a weak pulse and cyanosis associated
with his lips, nails and skin. Methaemoglobin levels represented 17%
of total haemoglobin at 5 h but returned to normal levels by 2 days.
He regained consciousness by 50 h, although complained of headache and
blurred vision. The only treatment received was gastric lavage, which
was performed shortly after his arrival at the hospital.
8.2 Occupational exposure
8.2.1 Acute poisoning incidents
Currie (1933) reported nine cases of haematuria in workers
exposed to 4-chloro- o-toluidine (erroneously called 5-chloro- o-
toluidine) by inhalation or possibly by absorption through the skin.
All patients had difficulty urinating and had suprapubic pain. Most of
the workers were exposed to the material for only 1-2 days. Despite
efforts to control exposure to the chemical in the factory, further
cases of poisoning occurred, and manufacture was ceased. In a
follow-up study of three of the nine cases after 3 years, one patient
had no bladder trouble, one had a slight cystitis and urethritis, and
one had carcinoma of the bladder.
Jurincic et al. (1991) reported cases of acute haemorrhagic
cystitis in two men (aged 19 and 50) following involvement in cleaning
of a water-tank that had likely been used to transport chlordimeform.
Both developed abdominal pain, dysuria and haematuria in the evening
following exposure. Cystoscopy revealed haemorrhagic cystitis,
which was confirmed by bladder mucosa biopsy. Serum levels of
4-chloro- o-toluidine (referred to as 4-chloro-2-methylaniline) were
>1 mg/litre in both patients and urine levels were 16 mg/litre in the
Table 16. Case studies of acute chlordimeform poisoning in China
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
1 71 4 dermal absorption 5 ECG: 26 tachycardia; 6 bradycardia; 11 ectopic Wang & Tong (1992)
(28 male, 67 ingestion rhythm; 6 premature beat; 2 atrial fibrillation;
43 female) ventricular fibrillation; 1"Torsade de Pointes";
2 high pike P, 6 A-V block, 17 S-T depression,
3 inverse T, 1 S-T elevation ,7 Q-T elongation.
In 33 severe cases, 28 has ECG abnormalities;
38 moderate cases, 14 had abnormal ECG. Changes
in heart were found in 32 cases. Deaths were from
respiratory failure (3); ventricular fibrillation
(1) and supraventicular tachycardia (1).
2 4 ingestion(?) 0 Mild cyanosis, cystitis (2 cases occupational, Nui et al. (1990)
2 cases non-occupational; OPs also in formulation.
3 1 female ingestion 0 Jaundice on 3rd day which progressively deepened. Liu et al. (1990)
(30 years old) (150 ml) Hb 40 g/litre (70 g/litre on admission); complete
recovery, discharged on day 20.
4 52 ingestion 0 Loss of appetite (86.5%), urgency in urination He (1989)
(19 male, (20-350 ml) (84.6%), cyanosis (81.1%), coma (67.3%), miosis
33 female) (34.6%), mydriasis (15.4%),hypotension (38.5%),
tachycardia (32.7%), bradycardia (3.8%).
Impairment of liver and renal functions. 15 ECGs:
7 tachycardia, 2 bradycardia, Q-T elongation,
8 T-wave changes. Treated with methylene blue,
vitamin C, fresh blood transfusion and sopolamine.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
5 35 ingestion(?) 0 18 severe cases. Suggested use of 5-36 mg He et al. (1987)
atropine for chlordimeform poisoning and
50-128 mg for mixed pesticide poisoning.
6 1 female ingestion 0 Cyanosis, pin-point myosis. Given atropine Zhou (1987)
(30 years old) (80 ml 25% (15 mg/min) after lavage until total of 530 mg.
chlordimeform) Symptoms indicated overdose of atropine. Methylene
blue given, recovery and discharge at day 7.
7 23 4 contaminated food; 3 Mild case: nausea, vomiting, light cyanosis,
(6 male, 19 ingestion no somnolence. Moderate case: somnolence and Xu (1987)
17 female) (10-350 ml) light consciousness. Severe case: Marked
cyanosis, coma, shock. 5 ECG examined:
2 bradycardia, 1 tachycardia, A-V block,
S-T change. Mild impairment of liver renal
functions. Treated with methylene blue,
19 recovered.
8 1 female ingestion 0 Lavage and treatment led to recovery from danger. Liu & Li (1987)
(52 years old) (30 ml conc. Black stool, tachycardia occurred on 3rd day.
formulation) Complete recovery.
9 187 27 occupational spray; 13 Cyanosis (63.6%), nausea (49.2%), vomiting Ding & Huang (1987)
(66 male, 16 ingestion (20-250 ml (44.9%), mydriasis (32.1%), somnolence (33.7%),
121 female) 25% chlordimeform coma (32.1%), irritation in urination (30.5%),
formulation) hypotension. 27 ECGs: 4 tachycardia, 6 bradycardia,
4 S-T & T wave change, 2 pre-mature beat, 2
conductive blockage. 158 cases received methylene
blue and 174 recovered within 1-5 days.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
10 1 male occupational spray 0 Sprayed incorrect dilution spray. Complained of Gu et al. (1987)
(28 years old) fatigue, somnolence, loss of appetite, nausea,
vomiting, but no cyanosis, or signs of cystitis,
pulse 68, BP 128/94 (normally 120/80), MAO 25.12 U
(normally 38.87 U). Total chlordimeform in urine
on admission, 6.4 mg/ml. Recovered quickly.
11 6 (?) ? Main clinical features: drowsy, cyanosis, Chan (1985)
loss of consciousness, mydriasis, cystitis,
hypotension, bradycardia, myocarditis, shock,
methaemaglobinaemia.
12 47 ingestion 4 Symptoms: drowsy, cyanosis, cystitis, Ke (1985)
(11 male, 20-1900 ml 2 hypotension (severe case), 8 hypertension,
36 female) 10 ECG: 1 tachycardia and T-wave change.
13 25 ingestion(?) 1 Cyanosis, cystitis, hypotension, arrhythmia, Wang & Dong (1985)
S-T and T changes, Q-T elongation. Treatment
with gastric lavage, methylene blue, vitamin C
14 682 340 occupational spray; 25 279 cyanosis, 147 cystitis, 197 somnolence, Liu & Zhang (1985)
(331 male, 342 ingestion 211 coma, 81 shock, 109 tachycardia,
351 female) 64 bradycardia, 54 hypertension, 22 hypotension.
59 ECG: 8 premature beat, 4 Q-T elongation,
16 S-T and T changes.
15 358 283 ingestion 37 Somnolence, cyanosis, loss of appetite, Ding & Ru (1985)
haemorrhagic cystitis, often myocardium damage,
A-V block, cardiac failure.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
16 49 3 occupational 4 13 cases were severe. Clinical features: cyanosis Liu & Ke (1985)
46 non-occupational and cystitis with haematuria in all cases, most
with severe somnolence and a few with coma. Two
severe cases had hypothermia. Hypertension was more
common than hypotension. 10 ECGs: only one case of
T-wave change and tachycardia. Treatment with
methylene blue and lavage.
17 1 ingestion (300 ml 25% 0 Coma and cyanosis. Sudden cardiac arrest during Yang (1984)
(female, form.) lavage, rescued with mechanical respiration.
25 years old) Recovered after 14 days.
18 24 ingestion (15-150 ml 2 16 cyanosis, 14 drowsiness, 8 haematuria, Wu et al. (1983)
(11 male, 25% form.) 6 methaemoglobin, 1 cardiac arrest, which
13 female) recovered after resuscitation.
19 101 35 occupational spray, 2 89 chlordimeform alone cases: 66% cyanosis, Xie (1983)
(49 male, 66 ingestion; 32 comas, 14 cystitis, 14 hypotension, 3 cardiac
52 female) chlordimeform +Ops) failure. 8 ECGs: 6 myocardium damage (changes in
Q-T, S-T, and T waves). Treatment with methylene
blue, vitamin. C. All recovered.
12 cases with mixed pesticides (OPs and Ocs).
20 1 ingestion 1 Loss of consciousness, cyanosis, mydriasis, Wu (1982)
(female, 85 years) (30 ml) arrhythmia. ECG: bradycardia, T-wave changes.
Died on day 6.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
21 20 occupational spray 0 Farmers applied wrong dilution chlordimeform to Li et al. (1982)
(18 male, 2 female) cotton for one day. 7 drowsy, 10 loss of appetite,
4 cystitis. Symptomatic treatment. All recovered
in 2-4 days.
22 2 male ingestion (100 & 200ml) 0 Cyanosis, coma, respiratory-circulation failure, Zhang et al. (1976)
cystitis during 2nd day. Treatment with
methylene blue and atropine.
23 1 male 100 ml 0 Deep cyanosis, pulse 166. Xia & Gao (1977)
24 6 male occupational spray 0 Contamination of body surface and clothing. Su (1977)
Symptoms from day 1-4: cyanosis, haemorrhagic
cystitis, fatigue. Recovery after 18 days.
25 2 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Anonymous (1977)
no cyanosis, ECG normal. Symptomatically treated.
26 4 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Ming (1977)
cyanosis, somnolence, loss of appetite, haematuria,
RBC in urine for 20 days. Treatment: vitamin C,
antibiotics, coagulators.
51-year-old patient. Case studies of chlordimeform poisoning in China
due to occupational exposure are given in Table 17, together with a
brief account of the clinical features observed.
Table 17. Levels of urinary chlordimeform and its metabolites in hospitalized
workers (3 days following exposure) (Folland et al., 1978)
Worker Total aminesa Chlordimeform 4-Chloro-o-toluidine Conjugate
(mg/litre) (mg/litre) (mg/litre) (mg/litre)
1 11.0 1.10 3.75 6.25
2 15.2 2.16 4.16 8.67
3 2.6 0.04 1.25 1.17
a Measured following hydrolysis with 10N NaOH and 2 h at 80�C.
A brief account of the signs and symptoms of chlordimeform
poisoning and suggested interventions has been provided by Xue &
Loosli (1994).
8.2.2 Effects of long-term exposure
A report of an outbreak of haematuria in employees of a chemical
packaging plant in the USA over a 4-day period in 1975 was first
reported by Armstrong et al. (1975). Further details were described by
Folland et al. (1978). Nine of 22 workers who packaged chlordimeform
became severely ill with abdominal pain, dysuria, urgency to void, or
haematuria. In the previous year, four workers who had packaged the
chemical had similar symptoms. While six workers recovered within 7 to
18 days, three were hospitalized with symptoms which lasted from one
to two months. In these three workers, abnormalities noted were
microscopic haematuria and pyuria, proteinuria, low creatinine
clearance, elevated SGOT, prolonged BSP retention, elevated serum
amylase level, small bladder capacity, ureteral reflux and an intense
inflammatory reaction in three bladder biopsy specimens. The highest
concentrations of total amines were found in the urine of workers who
had become ill and were hospitalized. Low but measurable levels were
also found in workers who had not become ill. The major part of the
urinary amines was present as 4-chloro- o-toluidine or as conjugates.
Urinary total amines (following hydrolysis with 10 N sodium hydroxide
and 2 h at 80�C), as well as chlordimeform and 4-chloro- o-toluidine,
were measured in the hospitalized cases and are shown in Table 17.
The results of a monitoring programme on packaging workers in a
chlordimeform plant in the USA during 1976 have been described
(personal communication by J.W. Barnett, Ciba-Geigy Agricultural
Division, Greenborough, North Carolina, USA, to the California
Department of Food and Agricultural). The programme involved more than
100 workers and over 800 urine samples, monitoring for the presence of
red blood cells, for residues of chlordimeform metabolites, and for
clinical signs of toxicity in workers. Residues in urine samples were
reported to range from <0.05 to 50 mg/litre. There was no evidence of
microscopic haematuria found in the samples analysed nor of any
clinical signs of toxicity.
Four separate incidents resulting in 7 cases of frank haematuria
following industrial exposure were reported in the USA during the
period 1980-1984 (personal communication by J.W. Barnett, Ciba-Geigy
Agricultural Division, Greenborough, North Carolina, USA, to
Ciba-Geigy Ltd., Switzerland). Chemical cystitis, confirmed by
cystoscopy and biopsy, was diagnosed in one case while non-specific
bladder mucosal lesions were found in another. Six cases required
hospitalization, but all resolved after cessation of exposure.
In a study by Maddy et al. (1986), the results of a programme of
monitoring (1982-1985) the urine of more than 200 workers, who had
received training in the use of chlordimeform on cotton in California,
were described. Although urinalysis was unremarkable and no
significant cytological changes were found, a single case of bladder
cancer was detected in a pilot who had seven seasons of exposure to
chlordimeform.
By contrast, in the same period (1980-1984), no cases of
chlordimeform-induced haematuria occurred at manufacturing plants in
Switzerland and West Germany or formulation plants in Australia,
Columbia, Central America, Mexico and the USA. No cases of haematuria
reportedly resulted from application or use of chlordimeform in the
field (Anon., 1985b; personal communications by F.E. Pfister and P.
Duback (Ciba-Geigy Ltd., Agricultural Division, Switzerland) and by
N. Reckefus and K. Kossmann (Schering Aktiengesellschaft Agrochemical
Division, Berlin, Germany), 1985).
In a study by Lu et al. (1981), data on the effects of
chlordimeform exposure of factory workers in China was examined. In
this study, conducted in 1974, the air concentrations in the factory
were generally below 0.036 mg/m3, with shorter periods at higher
levels (0.108-0.33 mg/m3), during specific tasks. Skin contamination
on hands and forearms was 9.1 mg/h for chemical operators
and 964.2 mg/h for packers. The urinary excretion levels of
chlordimeform and 4-chloro- o-toluidine in controls were 0.015 and
0.042 mg/litre, respectively; in chemical operators they were
0.065 and 0.108 mg/litre, respectively; and in packers were 0.263 and
0.398 mg/litre, respectively. The health of the workers was examined
during the following 3 years (1974-1976). In 44-56 workers (equal
number of each sex) at an average age of 32 years and working period
of 2 years, the main finding were neurosis, sore throat and disorders
of the nervous system. There were no treatment-related effects on ECG,
liver function, clinical chemistry or urinalysis parameters.
In the same report (Lu et al., 1981), the effect of chlordimeform
exposure on rice field workers during 1974 was also examined. The
air concentration in the breathing space in all cases was below
0.02 mg/m3. Skin contamination was examined at the front of the
thorax, on the right forearm and on the right thigh. The applicators
applied chlordimeform for 4-5 h per day for 1-3 consecutive days,
wearing shirts and shorts with no other protection. Skin contamination
was from splash or from spray. The levels found from splash on thorax,
forearm and thigh were 0.0436, 0.0303 and 0.131 mg/100 cm2 per h,
respectively. The levels found from spray on thorax, forearm and thigh
were 0.235, 0.299 and 0.804 mg/100 cm2 per h, respectively. Medical
examination during 1974/1975 revealed complaints of light-headedness,
headache, fatigue, nausea, abdominal pain, skin itching and burning
sensation, and hypotension. There were no changes in ECG or blood
chemistry, and no reported cases of acute intoxication.
In a study by Li et al. (1985b), the health of 24 packers
(9 male, 15 female) in a chlordimeform manufacturing plant in Jiang-su
Province of China, was examined. The chlordimeform division of the
factory started manufacturing in 1975 and continued to do so at the
time of the study. The employees were working in the factory for
between 3 months and 4 years (average 1.5 years). Another 24 employees
from the kitchen and kindergarten served as controls. The air
concentration of chlordimeform (9 samples over 3 consecutive days) was
0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin contamination of the
hands and forearms was 110 �g/100cm2 (S.D. 39 �g/100 cm2). Urinary
chlordimeform levels were 0.20 � 0.13 mg/litre, and urinary
4-chloro-o-toluidine levels were 0.48 � 0.29 mg/litre. Medical
examination revealed no difference between packers and controls with
regard to symptoms, laboratory examinations including liver enzymes
and urinalysis parameters, chest X-rays, ECG, or other parameters of
cardiac function. The only symptom associated with exposure was skin
rashes and itching in 21% of exposed individuals. There was no
difference in the micronucleus counting in cultured peripheral
lymphocytes between exposed and control groups, nor were there any
positive mutagenicity results from urine samples with or without
glucuronidase or sulfatase in the medium.
In a further study in a Chinese chlordimeform manufacturing
factory, the health of employees involved in chlordimeform production
was studied for the 5-year period, 1977 to 1981 (Anon., 1985a). The
urinary chlordimeform plus 4-chloro- o-toluidine levels of packers
was the highest at 0.39 mg/litre, which significantly correlated with
skin contamination but not with air concentration. The major medical
findings were complaints of lightheadedness, disorders in sleep,
memory impairment, fatigue, loss of appetite, skin rashes and itching,
and skin spot pigmentation. There were no features of cystitis. ECG
findings in 36 employees indicated premature beats, partial A-V block,
tachycardia and bradycardia. There was no evidence of chromosome
aberrations in metaphase chromosomes of cultured peripheral
lymphocytes.
In a study by Tao et al. (1985), the health of 61 employees
(25 chemical operators, 36 packers) of a pesticide factory in China
was examined. Chlordimeform was produced in the factory for 5 months
per year. Air levels ranged from 0.074 to 0.160 mg/m3. Skin
contamination of packers (2.99 mg/day) was higher than for
hemical operators (0.784 mg/day). The urinary excretion rate of
chlordimeform plus 4-chloro- o-toluidine in packers was also higher
(0.513 mg/litre) than for chemical operators (0.206 mg/litre) or
controls (0.055 mg/litre). Symptoms of exposure noted in packers
included loss of appetite, fatigue, somnolence and skin rashes.
Hepatomegaly was observed. There was no difference in blood pressure
or heart rate. Abnormalities in ECG were noted in 10/61 exposed
employees compared to 6/76 controls.
In a study by Wang et al. (1987), the health of 16 applicators
(8 males, 8 females) spraying chlordimeform in cotton fields in
Xin-yang Farm in the Jiang-su Province of China over a 3-day period
(July 1986) was examined. Air levels in the breathing zone were
0.031 mg/m3 and the skin contamination was 4.17 mg per shift. Urinary
levels of chlordimeform plus 4-chloro- o-toluidine ranged between 1
and 3 mg/litre over the exposure period. A close correlation was noted
between the level of chlordimeform on the skin and the levels of
chlordimeform plus 4-chloro- o-toluidine in the urine. Rapid
excretion of chlordimeform plus 4-chloro- o-toluidine was noted
following exposure. There was no change noted in heart rate, blood
pressure, monoamine oxidase activity or urinalysis between exposed
individuals and controls. Mild chlordimeform exposure, however,
appeared to be related to loss of appetite and drowsiness.
In a study by Zhang et al. (1986a), conducted at the same farm
over the same period, 13 applicators (7 male, 6 female, 20-41 years of
age) were examined during spraying chlordimeform on cotton over three
consecutive days. Protective measures included gauze mask, plastic
gloves and plastic apron, although it was noted that extensive
contamination occurred. Air levels in the breath zone on each of the
three days were 0.011, 0.014 and 0.011 mg/m3, respectively. Skin
contamination on each of the three days was estimated by the method of
Zhang et al. (1986b) to be 10.99, 4.32 and 4.45 mg/person per day,
respectively. Urinary chlordimeform plus 4-chloro- o-toluidine
levels were measured over the 3 days of exposure and for 7 days
after cessation of exposure. Urinary levels ranged from a peak of
2.408 mg/litre during exposure to 0.036 mg/litre after 7 days.
Excretion of chlordimeform occurred very rapidly with the highest
level being detected in the sample collected at the end of each shift.
There was a close correlation between skin contamination and urinary
excretion. Metabolism occurred very rapidly since 4-chloro- o-
toluidine usually accounted for 70-93 % of the total amount in the
urine. Serum monoamine oxidase activity varied from 26.18 U to
19.26 U. Clinical symptoms were somnolence, headache, dizziness and
fatigue. Heart rate and blood pressure dropped on the 2nd and 3rd
days. Analysis of ECG indicated elongation of P-R, Q-T intervals. One
person complained of urgency and pain in urination, gross haematuria,
and the urinary chlordimeform plus 4-chloro- o-toluidine level was
more than 6 mg/litre. Another four subjects were found to have
microscopic haematuria. Liver function tests were normal.
In a study by Xue et al. (personal communication by S.-Z. Xue,
M. Wang, C.-M. Chu and X.-W. Zhou entitled "Effects of chlordimeform
on cardiovascular function in humans with occupational exposure",
1993), the effect of chlordimeform on cardiovascular function was
studied in exposed farm workers and in manufacturing workers in China.
Four separate exposure groups were studied. The first (short-term)
exposure group consisted of 16 farmers engaged in spraying
chlordimeform (0.125% solution) in a cotton field. Exposure was for a
3- to 4-h period for 3 consecutive days. The second (long-term)
exposure group consisted of 21 chlordimeform packers in a factory who
had worked for 6 months on this task. The third exposure group
consisted of 19 factory plant operators who had minimal exposure to
chlordimeform. The fourth group consisted of 9 control (non-exposed)
factory workers. Exposure was measured in the breathing zone air
(personal sampler for the working shift, usually 6 h), by dermal
contact (pooled aliquot of 10 swabs from various body sites), and by
urine measurements. In each case, chlordimeform and its major
metabolite, 4-chloro- o-toluidine, were measured. The cardiovascular
system function was determined by measurement of blood pressure, heart
rate and electrocardiography (ECG). Exposure data indicated the packer
group had a higher inhalation exposure (0.107 mg/m3) than the
sprayers (0.031 mg/m3). Dermal exposure, on the other hand, was
higher in the sprayers group (4.251 mg/m2) than in the packers group
(2.713 mg/m2). Urinary levels collected at the end of the working
shift indicated the highest level in sprayers (1.950 mg/litre)
compared to packers (1.267 mg/litre) and operators (0.097 mg/litre).
In the farmer group, analysis of cardiovascular activity indicated a
significant decrease in heart rate, and an increase in P-wave
duration, Q-T interval and amplitude of T-wave compared to the control
group. In the factory workers, the packers had significantly lower
diastolic and systolic blood pressure, and an increase in T-wave
amplitude compared to the plant operators. The heart beat of packers
was also higher than controls, but not significantly. The
cardiovascular function parameters of the plant operators were
slightly but not significantly different to those of controls.
Examination of the cardiovascular function parameters of the
packers during a month of continuous exposure indicated a relationship
between length of exposure, total urinary chlordimeform, and cardiac
function parameters (see Table 18). Analysis of the exposure-effect
correlation indicated the drop in blood pressure was the most
sensitive parameter, with the change in amplitude of the T-wave the
next most sensitive parameter. The changes of P-R interval were the
least sensitive.
Table 18: Cardiovascular function and urinary chlordimeform in factory workers
(personal communication by S.-Z. Xue, M. Wang, C.-M. Chu and X.-W.
Zhou entitled "Effects of chlordimeform on cardiovascular function
in humans with occupational exposure", 1993)
Parameter measured Duration of continuous exposure (days)
0 1 7 15 30
Total urinary 0.000 0.311 0.627 0.642 0.773
chlordimeform (mg/litre)
Systolic BP (mmHg) 111/8.6a 105/7.7b 105/12c 102/10d 102/9.6c
Diastolic BP (mmHg) 71/7.1 69/9.3 63/10c 65/8.2d 64/9.8d
Heart rate (beat/min) 64.3/9.9 69.6/8.6d 67.2/6.5 70.0/9.4 71.4/12d
Q-T interval (msec) 398/18.2 404/23.1 412/16.7d 418/22.3d 412/23.6d
P-R interval (msec) 131/215 140/178d 140/212d 141/200a 143/317d
a Figures are mean/standard deviation
b P < 0.001
c P < 0.01
d P < 0.05
The authors attributed major importance to the alteration in
cardiovascular function in relation to chlordimeform intoxication, and
in most cases considered cardiac failure to be the cause of death.
Recognition of the effects on cardiac function may have been
overlooked previously, firstly, because of the diversity of mild
changes induced by chlordimeform and, secondly, because of the
tendency to concentrate on the effects of the aniline-containing
metabolites, such as methaema-globinaemia, haematuria, and
haemorrhagic cystitis. A no-observed-effect-level (NOEL) of
0.1 mg/litre of urinary chlordimeform plus 4-chloro- o-toluidine
excretion is proposed as the threshold for effects on cardiovascular
function following long-term, exposure while 0.3 mg/litre is proposed
as the threshold for effects on cardiovascular function following
short term exposure, even as short as one day. While the
cardiovascular function parameters are unlikely to be useful as
indicators of exposure, an understanding of the mechanism of action
should assist in designing appropriate treatment.
In a post-exposure surveillance programme, the chlordimeform-
exposed group showed an increased prevalence of malignancy-associated
surface markers on exfoliated urine cells, compared to geographical
controls, but no tumours were found (Kenyon et al., 1993).
8.2.3 Epidemiological studies
8.2.3.1 4-Chloro- o-toluidine
In a retrospective epidemiological study by Ott & Langner (1983)
the mortality experience of 342 employees assigned to three aromatic
amine-based dye production areas between 1914 and 1958 was examined in
relation to duration of employment (<1 to 5 years) and interval since
entry into these areas. 4-Chloro- o-toluidine represented one of a
number of chemicals to which the workers were potentially exposed.
4-Chloro- o-toluidine and two other aromatic amines ( o-toluidine
and 4-chloro-acetyl- o-toluidine) to which the workers were exposed
have been shown to be carcinogenic in animal studies. No deaths due to
bladder cancer were observed, and no statistically significant
increases in mortality by work area or duration of exposure within
work area were found.
In a retrospective study by Stasik (1988; 1991) of 116 workers
occupationally exposed in Germany to 4-chloro- o-toluidine during
manufacture prior to 1970, eight cases of bladder cancer, diagnosed
between 1967 and 1985, were identified. This represents an incidence
more than 70-fold higher than expected. Although occupational exposure
to two other aromatic amines, o-toluidine and 6-chloro- o-
toluidine, may have occurred, analysis of the production process
indicated that exposure to 4-chloro- o-toluidine in the plant was
considerably higher than exposure to these other two chemicals. The
workers were exposed to relatively high levels (before 1970) for a
median of 14 years. In two cases, however, the exposure period was
only 1.5 and 4.0 years. No quantitative measurements of exposure were
available. Two of the patients had suffered from haemorrhagic cystitis
as a consequence of massive acute exposure to 4-chloro -o-toluidine
at 4 and 14 years, respectively, before the tumour was diagnosed. The
latency periods for these eight cases ranged from 17 to 38 years. The
significantly increased incidence of bladder cancer in this study is
remarkable.
8.2.3.2 Chlordimeform
An epidemiological study has been conducted on the incidence of
cancer deaths of employees and their relatives on Xin-Yang Farm in
Jiang-su Province of China (Gu et al., 1991). In this area,
chlordimeform has been applied aerially in large amounts since 1974,
in a relatively indiscriminate manner, with contamination of land,
ponds, creeks, and gardens of adjacent houses. The study involved 7321
people (3911 male, 3410 female and 1265 retired agricultural workers)
over the period 1 January 1971 to 30 June 1987. During this period,
there were 706 registered deaths (510 males, 196 females), of which
198 were cancer deaths (160 males, 38 females). The standardized
mortality ratio (SMR) was calculated on the basis of the specific
mortality due to cancer in the adjacent Hai-men County. Many of the
SMRs were significantly exceeded on the Xin-Yang Farm, as shown in
Table 19. The incidences of bladder cancer adjusted to the national
level were 2.65 (males) and 1.47 (females) per 100 000. The SMRs were
260 (males) and 420 (females). During the period 1 July 1987 to
30 June 1990, there were three more cases of bladder cancer (with one
death) among the cohort members. The authors concluded there is
evidence for an association between bladder cancer and exposure to
chlordimeform, but that further data is needed to strengthen this
association. It is noted that there was a high incidence of other
tumour types in this study which makes the association between bladder
cancer and exposure to chlordimeform more difficult to establish.
Table 19. Standardized mortality ratio (SMR) for workers on the
Xin-Yang farm (Gu et al., 1991)
Cause of death Adjusted mortality Standardized mortality ratio
(per 100 000) (95% C.I.)
Male Female Male Female
All deaths 785.1 610.0 134 (124-145) 139 (128-151)
All cancers 214.0 130 113 (107-120) 128 (117-139)
Oesophageal cancer 35.6 32.5 228 (208-249) 388 (352-428)
Stomach cancer 61.4 24.5 175 (161-190) 120 (110-130)
Liver cancer 31.6 6.9 40 (37-44) 27 (24-29)
Colon cancer 8.8 6.5 133 (123-145) 79 (72-86)
Lung cancer 34.2 16.9 135 (124-146) 147 (135-169)
Leukemia 3.9 5.6 144 (133-157) 260 (235-285)
Bladder cancer 4.1 3.0 197 (180-214) 750 (671-839)
Breast cancer - 16.0 - 380 (345-419)
Cervical cancer - 30.7 - 216 (198-234)
Further epidemiological data on the association between cancer
incidence and exposure to chlordimeform has been provided in papers by
Xue et al. (1990; 1991). A summary of the findings of epidemio-logical
studies between 1984 and 1988 is given in Table 20. Data from three
counties and one farm are shown. The counties are located close to one
another, with comparable environmental and socio-economic situations.
The agricultural products are mainly rice and cotton. County A acted
as a control, with little or no use of chlordimeform; County B was
the largest user of chlordimeform; and County C started using
chlordimeform at the earliest time. The results from the Xin-Yang farm
are included for comparison. A comparison between the mortality rate
in recent years (1984-1988) and the mortality rate in the years prior
to the use of chlordimeform in these counties and Xin-Yang farm is
shown in Table 21. There were excesses in the incidence of all deaths,
deaths from cancer, and urinary bladder cancer for both sexes,
although the data may not yet have reached the level of statistical
significance.
Table 21. Comparison of adjusted mortalities of urinary bladder
cancer between 1984-1988 (county) and 1973-1975
(prefecture) (Xue et al., 1990, 1991)
Item County A County B County C Xin-Yang Farm /
/ Prefecture / Prefecture Prefecturea
Male 1.52 / 1.10 0.77 / 0.77 1.12 2.65 / 1.02
SRR 1.38 1.04 1.10 2.65b
Female 0.41 / 0.35 0.46 / 0.17 0.55 1.47 / 0.35
SRR 1.17 2.71b 1.57b 4.20b
a The duration of observation was 1971-1987 (June 30)
b p < 0.05
In a retrospective study by Popp & Norpoth (1991) and Popp et al.
(1992), the exposure and incidence of bladder cancer in a German
chemical plant was examined. Chlordimeform was manufactured from
4-chloro- o-toluidine and production commenced in December 1965.
Production was not continuous, but rather was in response to orders,
so workers were subject to different periods of exposure (generally
8-12 weeks per year). Between 1965 and 1976, the exact levels of
exposure were not available because measurement of the concentration
in the air or monitoring of urine excretion was not carried out at
that time. In 1976, production was ceased in order to improve working
conditions and minimize human exposure. Production recommenced in 1980
with improved containment and monitoring of urinary excretion of
Table 20. Data on Epidemiological Studies with Chlordimeform during 1984-1988 (Xue et al., 1990, 1991)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
Year started using chlordimeform 1979 1977 1973 1973
Population (annual average) 993 549 1 076 456 736 037 8732
Average amount of chlordimeform 1.1 65.0b 29.8 89.2
used (g/Mu/year)a
All causes of mortality
Male 584.5 675.7 761.0 785.1
RR 1.2 (1.1-1.3) 1.3 (1.2-1.4) 1.3 (1.2-1.5)
Female 438.1 891.7 668.5 625.9
RR 2.0 (1.4-2.3) 1.5 (1.1-1.7) 1.4 (1.3-1.5)
Cardiovascular mortalityc
Male 143.2 167.7 221.6 -
RR 1.2 (1.1-1.3) 1.6 (1.4-1.7)
Female 138.2 280.2 234.8 -
RR 2.0 (1.9-2.2) 1.7 (1.6-1.8)
Respiratory mortalityc
Male 99.6 100.4 127.2
RR 1.0 (0.9-1.1) 1.3 (1.2-1.4)
Female 82.1 145.1 124.0
RR 1.8 (1.7-1.9) 1.5 (1.4-1.6)
Table 20. (con't)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
All cancer mortalityc
Male 188.5 246.6 232.2 214.9
RR 1.3 (1.2-1.4) 1.2 (1.1-1.3) 1.1 (1.1-1.2)
Female 101.7 227.1 145.5 130.0
RR 2.3 (2.0-2.5) 1.4 (1.3-1.6) 1.3 (1.2-1.4)
Bladder cancer mortalityc
Male 2.08 (95)d 0.90 (26) 2.10 (32) 4.10 (4)
RR 0.4 (0.39-0.47) 1.0 (0.9-1.2) 2.0 (1.8-2.2)
Female 0.40 (15) 0.20 (9) 0.90 (14) 3.00 (2)
RR 0.5 (0.46-0.55) 2.3 (2.1-2.5) 7.5 (6.7-8.4)
a The Mu is a measure of area equivalent to 1/15th acre
b Considered over the last 5 years
c All mortality figures were age-adjusted
d Figure in parentheses is the actual number of cases of bladder cancer
workers. Production finally ceased in 1986. The company identified 170
individuals who had come into contact with chlordimeform but many had
minimal exposure. The number of workers involved in the production of
chlordimeform was 49, and these comprised the study group. The period
under investigation was from the year of employment to the end of
1990. The expected incidence of bladder cancer (age- and sex-specific)
was extracted from the cancer registers of Saarland (1988), the former
German Democratic Republic (GDR) (1978-1982) and Denmark (1978-1982).
The standard incidence rate (SIR) was the ratio of the number of cases
observed to the expected number (see Table 22).
Table 22. Standard incidence rates (SIRs) of bladder carcinoma in a
group of 49 workers engaged in chlordimeform synthesis
(Popp et al., 1992)
Observed cases Expected number SIR 95% CI p value
7 0.078 (GDR) 89.7 35.6 - 168.6 0.000002
7 0.200 (Denmark) 35.0 13.9 - 65.7 0.00001
7 0.130 (Saarland) 53.8 21.3 - 101.1 0.000005
The average age for workers starting work was 30 (range 18-51), and
the exposure ranged from 3 to 956 days. By the end of 1990, an average
of 18 (10-25) years had passed since the start of exposure. Bladder
cancer was detected in 7 of the 49 subjects by the end of 1990. Of the
seven cases, six were diagnosed as transitional cell carcinoma and one
as papillary carcinoma. In five cases, the exposure period could be
determined, with an average of 575 days (range 291-766). The latency
period was an average of 19 years (range 15-23), with an average age
at diagnosis of 54 years (range 42-62). This study provides strong
evidence of an association between exposure to 4-chloro- o-toluidine
and human bladder cancer. All of the cases involved workers who were
exposed to 4-chloro- o-toluidine while synthesizing chlordimeform
before 1976. None of those workers who were handling the final
product, chlordimeform, had developed bladder cancer by the end of
1990.
In a historical cohort study (personal communication by P. Boyle
& G.J. Macfarlane to the IPCS, 1997), the bladder cancer incidence of
847 men involved in the manufacture of chlordimeform in Australia,
Switzerland, the United Kingdom and the USA was compared with that
expected on the basis of population-based cancer registry rates.
Subjects eligible to be included in the cohort were those who had been
employed in the production or formulation of chlordimeform or who had
otherwise been an integral part of a chlordimeform unit in a plant
where it had been produced or formulated for at least 6 months. The
results presented in Table 23 show an incidence rate of bladder cancer
which was significantly higher than expected. Overall, ten cases were
observed while 2.6 were expected. When the cohort was divided
according to whether members had been exposed to chlordimeform and
4-chloro- o-toluidine, or to chlordimeform alone, it was found that a
significant excess of risk of bladder cancer also occurred in those
workers thought not to have been exposed to 4-chloro- o-toluidine. In
this group of 592 men, 5 cases of bladder cancer were observed, while
1.4 cases were expected (SIR = 3.5, 95% CI (1.1, 8.3)). The authors
concluded that despite the lack of information on potentially
confounding factors in this study, the data indicated an association
between excess risk of bladder cancer in this cohort and one or more
aspects of the manufacture of chlordimeform.
Table 23. Bladder cancer risk in a cohort of men exposed to
chlordimeform (Boyle & Macfarlane, 1997)
Plant location Cohort numbers Bladder cancer cases
Observed Expected SIRa
Switzerland 273 4 0.72 5.6
USA (A)b 182 1 0.32 3.1
United Kingdom 174 3 1.06 2.8
USA (B)b 163 1 0.26 3.8
Australia 55 1 0.27 3.7
All plants 847 10 2.63 3.8
95% CIc
(1.8, 7.1)
a Standardized Incidence Ratio
b Different production sites
c Confidence Interval
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1 Laboratory experiments
9.1.1 Microorganisms
There are no data on the effects of chlordimeform on
microorganisms.
9.1.2 Aquatic organisms
9.1.2.1 Plants
There are no data on the effects of chlordimeform on aquatic
plants.
9.1.2.2 Invertebrates
There are no laboratory data on the effects of chlordimeform on
aquatic invertebrates.
9.1.2.3 Vertebrates
The toxicity of chlordimeform to some species of fish has been
determined (FAO/WHO, 1972; Mayer & Ellersieck, 1986), and is shown in
Table 24.
9.1.3 Terrestrial organisms
9.1.3.1 Plants
There are no data available for the effects of chlordimeform on
plants.
9.1.3.2 Invertebrates
Dittrich (1966, 1967) first reported studies on the efficacy of
chlordimeform as an acaricide with both ovicidal activity against
insect eggs and adulticidal activity. It killed adult spider mites
when applied as a vapour and as a spray, and penetrated plant tissues
where it was released in ovicidal quantities. Since then, its efficacy
as an insecticide has been studied in a wide range of species.
Chlordimeform not only has a direct lethal action, particularly
against eggs and early instar larvae of insects and acarines, but also
has important sublethal effects, including sterilization of eggs,
induction of hyperactivity, detachment of feeding ticks from hosts,
Table 24. Toxicity of chlordimeform to fish
Species Duration LC50 Reference
(h) (mg/litre)
Bluegill sunfish 24 1.0 FAO/WHO (1972)
48 1.0
96 1.0
Trout 24 11.7 (8.73-15.8) FAO/WHO (1972)
48 10.6 (7.80-14.50)
96 7.14 (4.70-10.80)
Cat fish 24 11.9 (8.98-15.9) FAO/WHO (1972)
48 8.72 (6.26-21.1)
96 4.54 (3.08-6.68)
Rainbow trout 24 29 Mayer & Ellersiek
96 13.2 (1986)
Channel catfish 24 20.7 Mayer & Ellersiek
96 20.2 (1986)
Carp 24 65a FAO/WHO (1972)
48 60a
96 50a
a Values are for TLm
colony dispersal behaviour in ticks and mites, anti-feeding effects
and disruption of mating and oviposition in Lepidoptera (Hollingworth,
1976).
Knowles & Shrivastava (1973) investigated its toxicity in house
flies. The LD50 was 25 �g/fly, a dose which was not at a practical
level for house-fly control, possibly due to the high rate of
metabolism in this species. Pimley (1986) investigated the toxicity of
chlordimeform to tsetse fly (Glossina morsitans). The median lethal
dose was approximately 0.4 �g/fly for unfed tsetse, and 100% mortality
was achieved with 2 �g/fly. Sublethal doses also caused a significant
depression of feeding activity.
The specificity of chlordimeform with regard to both eggs and
larval stages was examined by Streibert & Dittrich (1977). Eggs of the
three noctuid cotton pest moths, Heliothis armigera, Heliothis
virescens, and Spodoptera littoralis, when exposed to a saturated
atmosphere of 4 mg/m3, have very similar sensitivity to vaporized
chlordimeform. Agrotis ipsilon, also a noctuid, on the other
hand, is definitely less sensitive, and the coccinillid Epilachna
varivestis was the most tolerant. The larval stages of all of these
species were considerably less sensitive to chlordimeform vapour than
the egg stage, but chlordimeform does seem to cause a decrease in the
number of larvae in the field, possibly due to a repellent effect or a
behavioural change rather than a direct toxic effect. These results
with Spodoptera littoralis on the relative sensitivity of eggs and
larvae were confirmed in the studies of Salvisberg et al. (1980).
Davenport & Wright (1985) have also demonstrated the differential
susceptibility of adult and larvae of the noctuid moths, Spodoptera
littoralis and Heliothis virescens, and also highlighted the
significantly higher toxicity of the hydrochloride salt, compared to
the base, to the adults of both species.
Sparks et al. (1993) studied the effects of several insecticides
on ovicidal activity and alteration of octopamine titres in eggs of
the tobacco budworm (Heliothis virescens). Chlordimeform was highly
toxic to eggs of H. virescens. The authors reported that the
alteration in the biogenic amine octopamine titres by chlordimeform
might lead to a disruption in the ability of larvae to hatch from the
egg.
Crecelius & Knowles (1976) studied the sensitivity of the larvae
of the cabbage looper, Trichoplusia ni, to the toxic effects of
chlordimeform. Third instar larvae were more sensitive to the toxic
effects of chlordimeform than the fifth instar larvae, possible due to
slower penetration and slower metabolism of chlordimeform in the
latter instar larvae.
Bailey & Cathey (1985) demonstrated the effectiveness of
chlordimeform in reducing the percentage egg hatch of Lygus
lineolaris (Palisot de Beauvois) on pole bean ( Phaseolus
vulgaris L.) pods and cotton ( Gossypium hirsutum L.). A solution
of 0.09% chlordimeform, while not significantly reducing nymph
emergence from eggs deposited on bean pole pods, did significantly
reduce nymph emergence from eggs deposited on cotton plants.
Salvisberg et al. (1980) also demonstrated that Spodoptera
littoralis moths, when treated at doses as low as 10% of the LD50,
showed symptoms of hyperexcitation, which resulted in abnormal
patterns of egg-laying, a reduced number of eggs and lower fertility.
Further studies by Davenport & Wright (1987) on Spodoptera
littoralis have shown that chlordimeform hydrochloride significantly
reduces food consumption in fifth-instar larvae when incorporated into
the diet at a level of 0.1-10 mg/g or when topically applied. No
mortality occurred during feeding, but mortality was increased during
subsequent pupation and during emergence of the adult from the pupae.
In adult moths, egg laying was significantly decreased when
chlordimeform hydrochloride was applied topically (1 or 10 �g/moth).
Further evidence that behavioural changes may be more important
in reducing both the larval and insect populations following
chlordimeform treatment has been provided by Shimizu & Fukami (1983)
in studies of the larvae of the cabbage armyworm, Mamestra
brassicae, which showed a prolonged period of wandering behaviour in
the presence of chlordimeform. This may have caused a failure to find
or prepare a suitable site for pupation.
The behaviour-modifying effects of chlordimeform have also been
studied by Blackwell (1988a,b; 1889) in the larvae of the large
cabbage white butterfly, Pieris brassicae L. When placed on
chlordimeform-dipped leaves, the larvae become excited, in contrast to
their normal communal feeding behaviour. Locomotion was increased and
feeding was significantly reduced as a result of disaggregation
(Blackwell, 1988a). When applied directly to the larvae, chlordimeform
caused excitation and inhibition of feeding (Blackwell, 1989). Direct
application also caused developmental delays and mortality was
increased at later developmental stages (Blackwell, 1988b).
O'Brian et al. (1985) have studied the effect of insecticides on
beneficial insects, and in particular, the effect of chlordimeform on
the ecoparasitoid, Bracon mellitor, an important parasitoid of the
boll weevil (Anthonomus grandis grandis). Chlordimeform was found to
be more toxic to Bracon mellitor than to the boll weevil, and also
reduced the number of egg deposited.
The toxicity of chlordimeform hydrochloride to bees has been
examined after both ingestion and contact. Ingestion of a 0.3%
solution killed 18%, while ingestion of 0.15% killed approximately 7%.
Contact with the same solutions did not increase the mortality rate
(FAO/WHO, 1972). In a study by Johansen (1972), bees were exposed to
field-weathered residues of a range of insecticides, including
chlordimeform, on alfalfa foliage. Over a 24-h period, zero mortality
was obtained with alfalfa leafcutter bees, alkali bees and honey bees
exposed to 3-h-old residues.
9.1.3.3 Vertebrates
Fleming et al. (1985) examined the toxic and behavioural effects
of chlordimeform on the game bird, the bobwhite quail (Colinus
virginianus). When added to the diet of newborn chicks over a 7-day
period, the lethal concentration to chicks was 2835 mg/kg diet
(2169-3705 mg/kg diet). When chicks were fed a diet containing
chlordime-form at a concentration of 1000 mg/kg diet for 7 days, they
ate less, weighed less, travelled further from a fright stimulus in an
avoidance test, and had a high locomotor activity in an open-field
test than at lower dose levels. Chicks fed 100 or 1000 mg
chlordimeform/kg diet scored more highly than controls in a visual
cliff performance test. After a further 8 days on control diet, the
chicks fed 1000 mg/kg diet still scored higher than controls on the
avoidance test, but the open-field and cliff performance scores were
similar to those of controls.
In studies conducted on bobwhite quails and ducks, groups of
animals (10 per treatment group, 30 per control group) were fed
chlordimeform technical or chlordimeform 48% EC formulation in their
diets for 5 consecutive days. The dose levels were 0, 10, 31.6, 100,
316 or 1000 mg/kg diet. Both quails and ducks were tolerant of the
presence of chlordimeform in the diet. With the technical material,
one quail in each of the groups fed 100 and 316 mg/kg diet died,
while, with the formulation, one quail in each of the groups fed 316
and 1000 mg/kg diet died. All ducks survived treatment, even at the
highest dose level (FAO/WHO, 1972).
Hill et al. (1975) exposed three bird species, Japanese quail
(Coturnix japonica), ring-necked pheasant (Phasianus colchicus)
and mallard (Anas platyrhynchos), to chlordimeform. LC50 values for
Japanese quail and ring-necked pheasant were determined to be 1749 and
2608 mg/kg diet, respectively. The LC50 for mallard was determined to
be >5000 mg/kg diet; only 20% mortality was reported at the highest
exposure group, 5000 mg/kg diet. Hill & Camardese (1986) reported an
LC50 of 5079 mg/kg diet for Japanese quail exposed to chlordimeform.
9.2 Field Observations
9.2.1 Microorganisms
There are no field data on the effects of chlordimeform on
microorganisms.
9.2.2 Aquatic organisms
There are no field data on the effects of chlordimeform on
aquatic organisms.
9.2.3 Terrestrial organisms
9.2.3.1 Plants
The possibility that some insecticides might enhance the growth
of cotton plants has been suggested for some time. However, in the
case of chlordimeform, debate has continued as to whether this effect
is due to early season insect suppression (Bailey & Cathey, 1985) or
to a physiological effect (Phillips et al., 1977). Cathey & Bailey
(1987) have conducted controlled studies to examine the effects of
multiple applications of chlordimeform on the growth and development
of cotton ( Gossypium hirsutum L.) in both greenhouse and field
studies. Plants were sprayed six times with chlordimeform either alone
or in combination with fenvalerate at 5- to 7-day intervals, beginning
at the six-leaf stage of plant development. In the absence of early
season insects and when insect populations were maintained at a
relatively low level, no increases in lint yield occurred on the
chlordimeform-treated plants. However, yield increases did occur and
insect populations became lower in these treated plots when early
season insect populations in the test area were relatively high. None
of the treatments influenced the boll components, boll size, seed
index and lint percentage, or the first fibre properties, length,
strength and micronaire.
Field studies by Youngman et al. (1990) to determine the effects
of several insecticides on growth, fruiting patterns and yield of the
cotton plant, Gossypium hirsutum L., supported the conclusion that
chlordimeform does not significantly increase any plant growth
parameter when compared with the control.
9.2.3.2 Invertebrates
In a small field study conducted by Bull & House (1978), tests
were conducted in 0.05-ha plots of cotton to compare lower and more
frequent applications of chlordimeform with commercial mixtures of
insecticides against natural populations of Heliothis species. The
results indicated that the protection afforded was as good as with
commercial mixtures, probably as a result of careful observation of
the cotton to pinpoint the onset of significant egg production.
In a another small field study by Wilson (1981), the potential of
chlordimeform to control Heliothis species in cotton was tested
separately or in combination with amitraz and the microbial
insecticide, Bacillus thuringiensis. Chlordimeform was the most
efficient of the three materials and controlled Heliothis species
reasonably efficiently, but no control of the rough bollworm,
Earias hueglei was obtained. There was also good control of the
cotton looper, Anomis flava, and some indication of suppression of
mites and aphids was obtained.
The behaviour-modifying effects of chlordimeform have been
demonstrated in field studies by Uk & Dittrich (1986) on the adult
whitefly, Bemisia tabaci (Genn.), which attacks cotton in the Sudan.
At dose levels of 500-2500 g chlordimeform/ha together with 960 g
endosulfan/ha, there was evidence of irritation and mass emigration of
adults from treated cotton foliage without detectable direct
mortality.
9.2.3.3 Vertebrates
There are no field data on the effects of chlordimeform on
vertebrates.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
Production and use of chlordimeform has now ceased worldwide
and no further human exposure should occur. During the years of
chlordimeform production and use, dietary and incidental exposure to
chlordimeform occurred. Occupational exposure to chlordimeform and
4-chloro- o-toluidine (used as a starting material for synthesis)
occurred during manufacture and formulation, as well as during
application. The major route of exposure was through dermal
contamination. Application of chlordimeform occurred extensively by
aerial spraying, but it was also applied by ground-rigs as well as by
backpack spray equipment. Thus, agricultural workers were exposed
during mixing, loading, washing, and flagging operations. General
population exposure occurred through the consumption of food
containing residues of chlordimeform, and to a lesser extent through
by-stander exposure. In some cases, there was intentional ingestion of
the formulation.
Monitoring of urinary 4-chloro- o-toluidine has been found to be
a useful indicator of exposure.
10.1.2 Toxicity
In both experimental animals and humans, there is extensive
metabolism of chlordimeform, followed by rapid excretion via the
urine. A major urinary metabolite is 4-chloro- o-toluidine. In
experimental animals, symptoms of acute toxicity included neurotoxic
as well as cardiovascular effects. There was no evidence of
teratogenicity or reproductive effects. Following chronic
administration, there was a dose-related increase in
haemangioendotheliomas in mice. There was no treatment-related
increase in tumour incidence in rats. Most of the mutagenicity studies
with chlordimeform itself were negative, but there were sporadic
reports of genotoxicity with 4-chloro- o-toluidine and to a more
limited extent with N-formyl-4-chloro- o-toluidine.
In humans, chlordimeform has been shown to have both acute and
chronic effects. Acute poisoning caused fatigue, nausea and loss of
appetite, and, in more severe cases, somnolence, cyanosis, urgency in
urination, cystitis, cardiovascular effects (tachycardia, bradycardia,
ECG changes), coma and shock. The significance of the cardiovascular
effects in chlordimeform-induced mortality has only recently been
recognized. While there have been fatalities as a result of acute
chlordimeform exposure, in the majority of cases complete recovery
occurs. Symptoms of chronic exposure include those of acute exposure
as well as abdominal pain, skin itching and rashes, and gross or
microscopic haematuria.
With regard to carcinogenicity, the International Agency for
Research on Cancer (IARC) has concluded that there is limited
evidence in humans and sufficient evidence in experimental animals
for the carcinogenicity of 4-chloro- o-toluidine. The available
epidemiological data indicate an association between excess risk of
bladder cancer and exposures entailed in the manufacture of
chlordimeform. There is currently preliminary epidemiological evidence
of an association between chlordimeform use and excess risk of bladder
cancer.
10.1.3 Risk evaluation
With the withdrawal of the use of chlordimeform in agriculture
and a cessation of production worldwide, there is no longer any risk
associated with acute exposure except during the disposal of existing
stocks. The risk associated with chronic exposure, however,
particularly the risk of bladder cancer, will continue to be of
concern for many years. Human bladder cancer has a long latency
period, and establishing whether or not there is a link between
chlordimeform exposure and bladder cancer will require continued
health screening of significantly exposed individuals both from
manufacturing plants and from those rural communities where
chlordimeform was extensively used.
10.2 Evaluation of effects on the environment
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
11. CONCLUSIONS AND RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
AND THE ENVIRONMENT
11.1 Conclusions
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine, and, to a
lesser extent, chlordimeform.
Chlordimeform does not persist in the environment and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
11.2 Recommendations for protection of human health and the
environment
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
12. FURTHER RESEARCH
The following studies are needed:
1. epidemiological investigations on exposed populations.
2. studies on the dose-response relationship between exposure to
4-chloro- o-toluidine or chlordimeform and the induction of
urinary bladder cancer in humans.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Chlordimeform was considered by the International Agency for
Research on Cancer (IARC) in 1983. IARC noted that no published study
on the carcinogenicity of chlordimeform was available. However, it
considered data on the carcinogenicity of 4-chloro- o-toluidine and
concluded that the results of experiments on mice provided sufficient
evidence that 4-chloro- o-toluidine, a metabolite of chlordimeform,
is carcinogenic to experimental animals. No relevant data on humans
were available. IARC concluded the available data were inadequate to
evaluate the carcinogenicity of chlordimeform to humans (IARC, 1983).
The carcinogenicity of 4-chloro- o-toluidine, the breakdown
product and major metabolite of chlordimeform, was evaluated by IARC
in 1990 (IARC, 1990). On the basis of the available published data, it
was concluded that there is limited evidence in humans and
sufficient evidence in experimental animals for the carcinogenicity
of 4-chloro- o-toluidine. 4-chloro- o-toluidine and its strong acid
salts were classified as probably carcinogenic to humans (Group 2A).
Chlordimeform was considered at the 1971, 1975, 1978, 1979, 1980,
1985 and 1987 FAO/WHO Joint Meeting on Pesticide Residues (JMPR). In
1971, a temporary acceptable daily intake (ADI) for chlordimeform of
0-0.01 mg/kg body weight was established, and temporary maximum
residue levels (MRLs) were set for a number of crops and for the meat
and milk of cattle (FAO/WHO, 1972). In 1975, the temporary ADI was
maintained and some new temporary MRLs were established (FAO/WHO,
1976). In 1978, the temporary ADI was reduced to 0-0.0001 mg/kg body
weight, the temporary MRLs for all crops except cotton and cottonseed
were withdrawn, and the MRLs for meat and milk of cattle were set at
the level of detection (FAO/WHO, 1979). In 1979, 1980 and 1985, the
temporary ADI of 0-0.0001 mg/kg body weight was extended (FAO/WHO,
1980, 1981, 1986). In 1987, the temporary ADI for chlordimeform was
withdrawn (FAO/WHO, 1988).
REFERENCES
Abo-Khatwa N & Hollingworth RM (1972a) Chlordimeform: The relation of
the mitochondrial uncoupling to toxicity in the German cockroach. Life
Science, 11: 1181-1190.
Abo-Khatwa N & Hollingworth RM (1972b) Chlordimeform: Uncoupling
activity against rat liver mitochondria. Pest Biochem Physiol,
3: 358-369.
Ahmad S & Knowles CO (1971), Metabolism of N'-(4-chloro-o-tolyl)-
N,N-dimethyl-formamidine (chlorphenamidine) and 4'-chloro-o-
formotoluidine by rat hepatic microsomal and soluble enzymes.
Camp Gen Pharmacol, 2:189-197.
Anonymous (1971 a) Gas chromatographic method for the determination of
C 8514 residues in animal tissues, fat, milk and butter. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Anonymous (1971b) Chlordimeform: Working paper prepared for the 1971
Joint FAO/WHO Meeting on Pesticide Residues. Basel, Switzerland,
Ciba-Geigy Ltd and Berlin, Germany, Schering AG (Unpublished
document).
Anonymous (1977) [Report of two cases of chemical cystitis.] New Med,
8: 82. (in Chinese with English abstract).
Anonymous (1980a) CGA-95119: High performance liquid chromatographic
determination of urinary residues containing the 4-chloro-o-toluidine
moiety - Procedure No. 177. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Anonymous (1980b) [Study on the fate of chlordimeform residue on green
cabbage.] Shanghai, Shanghai Institute of Pesticides, Basic Metabolism
Group (in Chinese with English abstract).
Anonymous (1981) [On the residue of chlordimeform: Special Task Group
report.] In: [Selected papers on research on pesticides.] In: Chinese
Institute of Scientific Information and Bureau of Science and
Technology ed. [Selected papers on studies of pesticides.] Beijing,
Ministry of Chemistry (in Chinese with English abstract).
Anonymous (1985a) [Five years follow-up observation on the health
effect of chlordimeform on the employees in manufacturing.] Lab Med,
2: 7-10 (in Chinese with English abstract).
Anonymous (1985b) Chlordimeform: Joint submission to the WHO expert
group of the 1985 FAO/WHO Joint Meeting on Pesticide Residues. Basel,
Switzerland, Ciba-Geigy Ltd and Berlin, Germany, Schering AG
(Unpublished report).
Arima T, Morooka H, Tanigawa T, Imai M, Tsunashima T, & Kita S (1976)
Methemoglobinemia induced by chlorphenamidine. Acta Med Okayama.
30: 57-60.
Armstrong J, Graves O, Lovejoy G, Swiggart R, & Kimbrough R (1975)
Insecticide induced acute haemorrhagic cystitis. Morbid Mortal Wkly
Rep (Centres for Disease Control), 24: 374.
Arni P & M�ller D (1976a) Salmonella/mammalian-microsome mutagenicity
test with C8513 (chlordimeform HC1), Basel, Switzerland. Ciba-Geigy
Ltd (Unpublished report).
Arni P & M�ller D (1976b) Salmonella/mammalian-microsome mutagenicity
test with CGA 72647 (4-chloro-o-toluidine HCI), Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1976c) Salmonella/mammalian-microsome mutagenicity
test with CGA 72651 (N-formyl-4-chloro-o-toluidine), Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983a) C8513: Intrasanguine host-mediated assay
with S. typhimurium, Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Arni P & M�ller D (1983b) CGA 72647: Intrasanguine host-mediated assay
with S. typhimurium, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983c) C8513: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983d) CGA 72647: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983e) CGA 72651 pure: Chromosome studies in male
germinal epithelium, mouse spermatocytes. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Arni P, Hool G, & M�ller D (1983a) C8513: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P, Hool G, & M�ller D (1983b) CGA 72651: Chromosome studies in
male germinal epithelium, mouse spermatogonia. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Aziz SA & Knowles CO (1973) Inhibition of monoamine oxidase by the
pesticide chlordimeform and related compounds. Nature (Lond),
242: 417-418.
Bailey JC & Cathey GW (1985) Effect of chlordimeform on tarnished
plant bug (Heteroptera: Miridae) nymph emergence. J Econ Entomol,
78: 1485-1487.
Bailey BA, Martin RJ, & Downer RGH (1982) Monoamine oxidase inhibition
and brain catecholamine levels in the rat following treatment with
chlordimeform. Pest Biochem Physiol, 17: 293-300.
Balu K (1989) Evaluation of field worker exposure to chlordimeform by
using urine monitoring. In: Biological monitoring for pesticide
exposure: Measurement, estimation of risk reduction. Washington, DC,
American Chemical Society, pp 47-55 (ACS Symposium Series 382).
Bauer PJ & Cothren JT (1990) Growth-promoting activity of
chlordimeform. Agron J, 82: 73-75.
Beeman RW & Matsumura F (1973) Chlordimeform: A pesticide acting upon
amine regulatory mechanisms. Nature (Lond), 242: 273-274.
Beilstein P & M�ller D (1983) C8513 pure: BALB/3T3 cell transformation
assay. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Beilstein P & M�ller D (1984a) C8513 technical: L-5178Y/TK+/- mouse
lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Beilstein P & M�ller D (1984b) CGA 72647 technical: L1578Y/TK+/-
mouse lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Beilstein P & M�ller D (1984c) CGA 72647 technical: BALB/3T3 cell
transformation assay. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Benedict JH, Walmsley MH, Segers JC, & Treacy MF (1986) Yield
enhancement and insect suppression with chlordimeform (Fundal) on
dryland cotton. J Econ Etomol, 79: 238-242.
Benezet HJ & Knowles CO (1976) N'-(4-chloro-o-tolyl)-N-
methylformamidine (demethylchlordimeform) metabolism in the rat.
J Agric Food Chem, 24: 152-154.
Benezet HJ & Knowles CO (1981) Degradation of chlordimeform by algae.
Chemosphere, 10: 909-917.
Benezet HJ, Chang K, & Knowles CO (1978) Formamidine pesticides -
metabolic aspects of neurotoxicity. In: Pesticide venom neurotoxic,
pp 189-206.
Bentley P, Staubli W, Bieri F, Muecke W, & Waechter F (1985) Induction
of hepatic drug-metabolising enzymes following treatment of rats and
mice with chlordimeform. Toxicol Lett, 28: 143-149.
Bentley P, Bieri F, Muecke W, Waechter F, & Staubli W (1986a) Species
differences in toxicity of p-chloro-o-toluidine to rats and mice:
Covalent binding to hepatic macromolecules and hepatic non-parenchymal
cell DNA and an investigation of effects upon the incorporation of
[3H]-thymidine into capillary endothelial cells. Chem-Biol Interact,
57: 27-40.
Bentley P, Waechter F, Bieri F, Staubli W, & Muecke W (1986b) Species
differences in the covalent binding of p-chloro-o-toluidine to DNA.
Arch Toxicol, 9(suppl): 163-166.
Blackmore RH (1969a) Chlorphenamidine (Schering 36268): 2-year
repeated feeding study in dogs (Project No. 728-127). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969b) Chlorphenamidine (Schering 36268): 24-month
repeated feeding study in rats (Project No. 728-126). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969c) Chlorphenamidine (Schering 36268): three
generation reproduction study (Project No. 728-118). Falls Church,
Virginia, Hazleton Laboratories Inc (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969d) Chlorphenamidine (Schering 36268). Segment II:
Teratology Study -Rabbits (Project No. 728-157). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackwell A (1988a) Survival and development of Pieris brassicae
larvae upon application of sublethal doses of chlordimeform. Entomol
Exp Appl, 48: 149-156.
Blackwell A (1988b) The disruption of aggregation behaviour in Pieris
brassicae larvae by chlordimeform. Entomol Exp Appl, 48: 141-147.
Blackwell A (1989) The effects of chlordimeform on the behaviour of
Pieris brassicae larvae. Pestic Sci, 26: 11-19.
Blass W (1972a) Galecron residues in Japanese tea. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. RVA 5/72).
Blass W (1972b) Galecron residues in Swiss grapes (berries and juice).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 11/72).
Blass W (1972c) Galecron residues in Swiss wine 1971. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 16/72).
Bokisch AJ, Cox RT, & Walker RJ (1985) The actions of formamidine
pesticides on identified central neurons of Helix aspersa. Pestic:
Sci, 16: 495-503.
Boyes WK & Dyer RS (1984) Chlordimeform produces profound, selective,
and transient changes in visual evoked potentials of hooded rats. Exp
Neurol, 86: 434-447.
Boyes WK & Moser VC (1988) An alpha 2-adrenergic mode of action of
chlordimeform on rat visual function. Toxicol Appl Pharmacol,
92: 402-418.
Budris DM, Yim GK, Carlson GP, & Schnell RC (1983) Effect of acute and
repeated chlordimeform treatment on rat hepatic microsomal drug
metabolizing enzymes. Toxicol Lett, 18: 63-71.
Budris DM, Yim GK, & Schnell RC (1984) Effect of acute and repeated
chlordimeform treatment on rat hepatic microsomal drug metabolizing
enzymes. J Environ Pathol Toxicol Oncol, 5: 175-182.
Bull DL (1973) Metabolism of chlordimeform in cotton plants. Environ
Entomol, 2: 869-871.
Bull DL & House VS (1978) Effects of chlordimeform on insects
associated with cotton. Southwest Entomol, 3: 284-291.
Burkhard N (1971) Residues in milk, meat, liver and fat of a Swiss
cow. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
14/71).
Campanhola C & Plapp FW Jr (1989) Pyrethroid resistance in the tobacco
budworm (Lepidoptera: Noctuidae): Insecticide bioassays and field
monitoring. J Econ Entomol, 82: 22-28.
Candura SM, Marraccini P, Costa LG, Manzo L, Rossi A, Coccini T, &
Toninin M (1992) Calcium entry blockade as a mechanism for
chlordimeform induced inhibition of motor activity in the isolated
guinea-pig ileum. Pharmacol Toxicol, 71: 426-433.
Cathey GW & Bailey JC (1987) Evaluation of chlordimeform for cotton
yield enhancement. J Econ Entomol, 80: 670-674.
Chan Y-M (1985) [Observation and nursing of 6 cases of chlordimeform
poisoning.) Chin J Nurs, 20: 68-69 (in Chinese with English abstract).
Cheung MW, Kahrs RA, Nixon WB, Ross JA, & Tweedy BG (1989)
Determination of chlordimeform residues as 4-chloro-o-toluidine in
human urine using high-performance liquid chromatography. In:
Biological monitoring of pesticide exposure: Measuring and estimating
risk reduction. Washington, DC, American Chemical Society, pp 231-239
(ACS Symposium Series 382).
Cooper RL, Barrett MA, Goldman JM, Rehnberg GL, McElroy WK, & Stoker
TE (1994) Pregnancy alterations following xenobiotic-induced delays in
ovulation in the female rat. Fundam Appl Toxicol, 22: 474-480.
Costa LG & Murphy SD (1987) Interaction of the pesticide chlordimeform
with adrenergic receptors in mouse brain: an in vitro study. Arch
Toxicol, 59: 323-327.
Costa LG, Wu S, Olibet G, & Murphy SD (1988) Alpha2-adrenoceptors as
a target for formamidine pesticides: In vitro and in vivo studies in
mice. Toxicol Appl Pharmacol, 93: 319-328.
Costa LG, Wu S, Olibet G, & Murphy SD (1989) Formamidine pesticides
and alpha2-adrenoceptors: Studies with amitraz and chlordimeform in
rats and development of a radioreceptor binding assay. Neurotoxicol,
11: 405-411.
Costa LG, Gastel J, & Murphy SD (1991) The formamidine pesticides
chlordimeform and amitraz decrease hepatic glutathione in mice through
an interaction with alpha2-adrenoceptors. J Toxicol Environ Health,
33: 349-358.
Coye MJ, Lowe JA, & Maddy KJ (1986) Biological monitoring of
agricultural workers exposed to pesticides: II. Monitoring of intact
pesticides and their metabolites. J Occup Med, 28: 628-636.
Crecelius CS & Knowles CO (1976) Toxicity, penetration, and metabolism
of chlordimeform and its N-demethyl metabolite in cabbage looper
larvae. J Agric Food Chem, 24: 1018-1023.
Crowder LA & Whitson RS (1980) Fate of toxaphene, methyl parathion,
and chlordimeform combinations in the mouse. Bull Environ Contam
Toxicol, 24: 444-451.
Currie AN (1933) Chemical haematuria from handling 5-chloro-ortho-
toluidine. J Ind Hyg, 15: 205-213.
Davenport AP & Wright DJ (1985) Toxicity of chlordimeform and amitraz
to the Egyptian cotton leafworm (Spodoptera littoralis) and the
tobacco budworm (Heliothis virescens). Pestic Sci, 16: 81-87.
Davenport AP & Wright DJ (1987) Effect of the formamidine insecticide
chlordimeform on feeding and reproduction in the noctuid moth,
Spodoptera littoralis (Boisduval). Crop Prot, 6: 3-6.
Deng C-S, Zhang J-K, & Guo J-Y (1984) [Brief report of the symposium
on chlordimeform poisoning.] J Clin Med, 1: 51-53 (in Chinese with
English abstract).
Ding M-B & Ru X-Y (1985) [On diagnosis and treatment of chlordimeform
poisoning (analysis of 358 cases).] Jiangsu Med, 7: 30-31 (in Chinese
with English abstract).
Ding Y & Huang K-L (1987) [Analysis on clinical features of
chlordimeform poisoning.] Labor Med, 4: 8-10 (in Chinese with English
abstract).
Dittrich V (1966) N-(2-methyl-4-chlorophenyl)-N',N'-
dimethylformamidine (C-8514/Schering 36268) evaluated as an
acaricide. J Econ Entomol, 59: 889-893.
Dittrich V (1967) Systemic action of N-(2-methyl-4-chlorophenyl)-N',
N'-dimethylformamidine (C-8514/Schering 36268). J Econ Etomol,
60: 558-569.
Dittrich V & Loncarevic A (1971) New insecticides for Asiatic rice
borer control in paddy rice. J Econ Entomol, 64: 1225-1229.
Dyer RS & Boyes WK (1983) Chlordimeform differentially affects pattern
reversal-evoked and flash-evoked potentials in rats. Toxicologist,
3: 13.
Ehrhardt DA & Knowles CO (1970) Metabolism and translocation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine (chlorphenamidine) and
its hydrochloride salt in grapefruit seedlings. J Econ Entomol,
63: 1306-1314.
Ercegovich CD, Witkonton S. & Asquith DJ (1972) Disappearance of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine from six major fruit
crops. J Agric Food Chem, 20: 565-568.
Evans PD & Gee JD (1980) Action of formamidine pesticides on
octopamine receptors. Nature (Lond), 287: 60-62
Fan DF & Ge SD (1982) Gas-liquid chromatographic determination of
chlordimeform and its metabolites in cargo rice and husk. J Assoc Off
Anal Chem, 65: 1517-1520.
FAO/WHO (1972) Chlordimeform. In: 1971 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 3-45 (WHO
Pesticide Series, No. 1).
FAO/WHO (1976) Chlordimeform. In: 1975 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 85-104 (WHO
Pesticide Residues Series, No. 5).
FAO/WHO (1979) Chlordimeform. In: Pesticides residues in food - 1978:
Evaluations, Rome, Food and Agriculture Organization of the United
Nations, PP 51-78 (FAO Plant Production and Protection Paper 15 Sup.).
FAO/WHO (1980) Chlordimeform. In: Pesticide residues in food - 1979:
Evaluations. Rome, Food and Agriculture Organization of the United
Nations, pp 129-135 (FAO Plant Production and Protection Paper 20
Sup.).
FAO/WHO (1981) Chlordimeform. In: Pesticide residues in food - 1980:
Evaluations. Rome. Food and Agriculture Organization of the United
Nations, pp 89-96 (FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1986) Chlordimeform. In: Pesticide residues in food - 1985:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 17-29 (FAO Plant Production and
Protection Paper 72/2).
FAO/WHO (1988) Chlordimeform. In: Pesticide residues in food - 1987:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 25-26 (FAO Plant Production and
Protection Paper 86/2).
Fleming WJ, Heinz GH, & Schuler CA (1985) lethal and behavioral
effects of chlordimeform in bobwhite. Toxicology, 36: 37-47.
Folland DS, Kimbrough RD, Cline RE, Swiggart RC, & Schaffner (1978)
Acute hemorrhagic cystitis - Industrial exposure to the pesticide
chlordimeform. J Am Med Assoc, 239: 1052-1055.
Fritz H (1971) Reproduction study on C8514 (chlorphenamidine)
technical in rabbit - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 10071 004). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1975) Reproduction study on C8514 (chlorphenamidine)
technical in rat - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 227533). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1978a) C8513 technical: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327735).
Fritz H (1978b) CGA 72651: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327737).
Fritz H, Becker H, & M�ller D (1978) CGA 72647 technical: Dominant
lethal study (mouse). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782201).
Gaines TB & Linder RE (1986) Acute toxicity of pesticides in adult and
weanling rats. Fundam Appl Toxicol, 7: 299-308.
Gale GT Jr & Hofberg AH (1985) Gas chromatographic determination of
chlordimeform in pesticide formulations: Collaborative study. J Assoc
Off Anal Chem, 68: 589-592.
Galloway SM, Armstrong MJ, Reuben C, Colman S, Brown B, Cannon C,
Bloom AD, Nakamura F, Ahmed M, Duk S, Rimpo J, Margolin BH, Resnick
MA, Anderson B, & Zeiger E. (1987) Chromosome aberrations and sister
chromatid exchanges in Chinese hamster ovary cells: Evaluations of 108
chemicals. Environ Mol Mutagen, 10(suppl): 1-175.
Geissb�hler H, Kossmann K, Baunok I, & Boyd VF (1971) Determination
of total residues of chlorphenamidine in plant and soil material by
colorimetry of thin layer and electron capture gas chromatography.
J Agric Food Chem, 19: 365-371.
Geyer R & Fattal F (1987) HPLC determination of the metabolite
4-chloro-o-toluidine in the urine of workers occupationally exposed to
chlordimeform. J Anal Toxicol, 11: 24-26.
Ghali GZ & Hollingworth RM (1985) Influence of mixed function
oxygenase metabolism on the acute neurotoxicity of the pesticide
chlordimeform in mice. Neurotoxicology, 6: 215-238.
Gifkins MR & Jacobson RA (1980) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine (chlordimeform), C10H13CIN2.
Cryst Struct Commun, 9: 959-963.
Glowa JR (1986) Acute and sub-acute effects of deltamethrin and
chlordimeform on schedule-controlled responding in the mouse.
Neurobehav Toxicol Teratol, 8: 97-102.
Goldman JM, Cooper RL, Laws SC, Rehnberg GL, Edwards TL, McElroy WK, &
Hein JF (1990) Chlordimeform-induced alterations in endocrine
regulation within the male rat reproductive system. Toxicol Appl
Pharmacol, 104: 25-35.
Goldman JM, Cooper RL, Edwards TL, Rehnberg G, McElroy WK, & Hein JF
(1991) Suppression of the luteinizing hormone surge by chlordimeform
in ovariectomized, steroid-primed female rats. Pharmacol Toxicol,
68: 131-136.
Goldman JM, Stoker TE, Perreault SD, Cooper RL, & Crider MA (1993)
Influence of the formamidine pesticide chlordimeform on ovulation in
the female hamster - Dissociable shifts in the luteinizing hormone
surge and oocyte release. Toxicol Appl Pharmacol, 121: 279-290.
Gordon CJ & Watkinson WP (1988) Behavioral and autonomic
thermoregulation in the rat following chlordimeform administration.
Neurotoxicol Teratol, 10: 215-219.
Gordon CJ, Long MD, & Stead AG (1985) Thermoregulation in mice
following acute chlordimeform administration. Toxicol Lett, 28: 9-15.
Gross (1977) Metabolism of C 8514 (chlordimeform) in cotton. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 19/77).
Gr�bner P (1977) [Thin-layer chromatographic determination of
chlordimeform in cucumbers and apples.] Nahrung, 21: 339-345
(in German).
Gu Q, Kui N-D, Wang M & Xue S-Z (1987) [Report on a mild case of
chlordimeform poisoning.] J Environ Toxicol, 1: 61-62 (in Chinese with
English abstract).
Gu Y-E, Wang M, Gu Q, & Dai G-J (1991) [Epidemiologic study on the
cancer mortality of employees and their relatives in Xin-Yang farm.]
J Jiangsu Occup Health, 1: 5-8 (in Chinese).
Guth JA & Senn U (1969) Galecron - Dissipation in a Swiss soil 1969 as
determined by chemical analysis. Basel, Switzerland Ciba-Geigy Ltd
(Unpublished report).
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(suppl): 3-142.
He R-H (1989) [Analysis on the clinical symptoms of 52 acute
chlordimeform poisoned cases.] Occup Med, 16: 27 (in Chinese with
English abstract).
He D-Q, Peng B, & He P-L (1987) [Experience of treatment of
chlordimeform poisoning with atropine.] Hu-Nan Med, 4: 169 (in Chinese
with English abstract).
Henderson GR (1985) Report on the continuing use of chlordimeform for
cotton insect pest control by aerial and ground rig application in
1984/5 - NSW report. Pendle Hill, Ciba-Geigy Australia Ltd and
Kensington, Australia, Schering Pty Ltd (Unpublished report No. CGS
85/1).
Hermosin MC & Perez-Rodriguez JL (1981) Interaction of chlordimeform
with clay minerals. Clays Clay Miner, 29: 143-152.
Hill EF & Camardese ME (1986) Lethal dietary toxicities of
environmental contaminants and pesticides to Coturnix. Washington, DC,
US Department of the Interior, Fish and Wildlife Service, 147 pp (Fish
and Wildlife Technical Report No. 2).
Hill EF, Heath RG, Spann JW, & Williams JD (1975) Lethal dietary
toxicity of environmental pollutants to birds. Washington, DC, US
Department of the Interior, Fish and Wildlife Service, p 61 (Special
Scientific Report - Wildlife No. 191).
Hill DL, Shih T-W & Struck RF (1979) Macromolecular binding and
metabolism of the carcinogen 4-chloro-2-methylaniline. Cancer Res,
39: 2528-2531.
Hollingworth RM (1976) Chemistry, biological activity and uses of
formamidine pesticides. Environ Health Perspect, 14: 57-69.
Hollingworth RM, Leister J, & Ghali G (1979) Mode of action of
formamidine pesticides: an evaluation of monoamine oxidase as the
target. Chem-Biol Interact, 24: 35-49.
Holsapple MP & Yim GK (1981) Decreased gastrointestinal ulcerogenicity
of chlordimeform, a basic anti-inflammatory agent. Toxicol Appl
Pharmacol, 59: 107-110.
Homburger F, Friedell GH, Weisburger EK, & Weisburger JH (1972)
Carcinogenicity of simple aromatic amine derivatives in mice and Rats.
Toxicol Appl Pharmacol, 22: 280 (Abstract).
Honeycutt RC & Cassidy JE (1977) Extraction and characterization of
cottonseed metabolites from cotton injected with 14C-chlordimeform.
Greensboro, North Carolina, Ciba-Geigy Corporation (Unpublished report
No. ABR - 77087).
Hool G & Arni P (1983a) C8513 pure: Sister chromatid exchange studies
on somatic cells of Chinese hamster. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 830305).
Hool G & Arni P (1983b) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatogonia). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782902).
Hool G & Arni P (1983c) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782901).
Hool G & Arni P (1983d) CGA 72647 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 783101).
Hool G & Arni P (1983e) CGA 72647 pure: Sister chromatid exchange
studies on somatic cells of Chinese hamster. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 830312).
Hool G & Arni P (1983f) CGA 72651 pure: Chromosome studies in somatic
cells (Chinese hamster). Base], Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 774054).
Hool G & M�ller P (1978) C8513 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 40400577).
Hool G, Beilstein P, & Arni P (1983) C8513 technical: Chromosome
studies in male germinal epithelium (mouse spermatogonia). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 773062).
Hsu WH & Kakuk TJ (1984) Effect of amitraz and chlordimeform on heart
rate and pupil diameter in rats: Mediated by alpha2-adrenoreceptors.
Toxicol Appl Pharmacol, 73: 411-415.
Huang X-S, Ding S, & Chen P-H (1979) [Bioassay on the carcinogenicity
of chlordimeform.] In: Chinese Institute of Scientific Information and
Bureau of Science and Technology ed. [Selected papers on studies of
pesticides.] Beijing, Ministry of Chemical Industry, pp 789-792 (in
Chinese with English abstract).
Huang H-X, Xue K-L, & Shi G-J (1989) [Study on the status of pollution
on rice by chlordimeform.] Monograph submitted to the National
Pesticide Reviewing Committee (in Chinese with English abstract).
Hurni H & Sachsse K (1969) Report on the determination of the acute
oral LD50 to the dog of chlorfenamidin technical. Sisseln,
Switzerland, Tierfarm AG (Unpublished report).
IARC (1978) Some aromatic amines and related nitro compounds - hair
dyes, colouring agents and miscellaneous industrial chemicals. Lyon.
International Agency for Research on Cancer (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Man, Volume 16).
IARC (1983) Chlordimeform. Lyon, International Agency for Research on
Cancer, pp 61-72 (IARC Monographs on the Evaluation of Carcinogenic
Risk of Chemicals to Man, Volume 30)
IARC (1990) Some flame retardants and textile chemicals and exposures
in the textile manufacturing industry. Lyon, International Agency for
Research on Cancer, pp 123-137 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 48).
Ifflaender U (1977a) Distribution, degradation and excretion of
chlordimeform (CDF) in mice and rats. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 38/77).
Ifflaender U (1977b) Comparison of the urinary metabolite pattern of
mice and rats after oral application of "C-chlordimeform. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 39/77).
Iizuka H & Masuda T (1979) Residual fate of chlorphenamidine in rice
plant and paddy soil. Bull Environ Contam Toxicol, 22: 745-749.
IRPTC (1992) IRPTC data profile on chlordimeform. Geneva, United
Nations Environment Programme, International Register of Potentially
Toxic Chemicals.
Iwan J & Goller D (1975) Metabolic fate of chlordimeform in soil under
aerobic, anaerobic, and sterile conditions. Environ Qual Saf (Pestic),
3(suppl): 282-287.
Iwan J, Hoyer GA, Rosenberg D, & Goller D (1976) Transformations of
4-chloro-o-toluidine in soil: Generation of coupling products by
one-electron oxidation. Pestic Sci, 7: 621-631.
Janssen R, Boyes WK, & Dyer RS (1983) Effects of chlordimeform on the
brainstem auditory evoked response in rats. In: Hayes AW, Schnell RC,
& Miya TS ed. Developments in the Science and practice of toxicology,
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 533536.
Jiang J-K, Xu W-Y, Xu S-Q, Li J-L, Zhou F-D, Yang S-X, Zhong S-L, &
Zhu R-L (1985) [Survey on the hygienic status on spraying
chlordimeform with fine mist sprayer.] Labor Med, 2: 4-6 (in Chinese;
English translation).
Jiang X-Z, Li F, Xue S-Z, & Wang -L (1988) Carcinogenicity assay of
chlordimeform on dermal application in mice. Chin J Ind Hyg Occup Dis,
6: 138.
Johansen CA (1972) Toxicity of field-weathered insecticide residues to
four kinds of bees. Environ Entomol, 1: 393-394.
Johnson BT & Knowles CO (1970) Microbiological degradation of the
acaricide N-(4-chloro-o-tolyl)-N,N'-dimethylformamidine. Bull Environ
Contam Toxicol, 5: 158-170.
Johnson TL & Knowles CO (1981) Inhibition of rat platelet
5-hydroxytryptamine uptake by chlordimeform. Toxicol Lett, 9: 1-4.
Johnson TL & Knowles CO (1982) Interaction of formamidines with
the platelet 5-hydroxytryptamine uptake system. Gen Pharmacol,
13: 299-307.
Johnson TL & Knowles CO (1983) Influence of formamidines on biogenic
amine levels in rat brain and plasma. Gen Pharmacol, 14: 591-596.
Jurincic C, Horlbeck R, Metz K, & Klippel KF (1991) [Acute hemorrhagic
cystitis following chlordimeform poisoning.] Dtsch Med Wochenschr,
116: 816-818 (in German).
Kadir HA & Knowles CO (1981) Inhibition of rat brain monoamine oxidase
by insecticides, acaricides and related compounds. Gen Pharmacol,
12: 239-247.
Kale PG, Petty BT Jr, Walker S, Ford JB, Dehkordi N, Tarasia S, Tasie
BO, Kale R, & Sohni YR (1995) Mutagenicity testing of nine herbicides
and pesticides currently used in agriculture. Environ Mol Mutagen,
25: 148-153.
Kaloyanova F, Bainova A, & Zaprianov Z (1981) Inhibition of monoamine
oxidase activity after combined action of chlordimeform with the
antidepressant, nialamide. Arch Environ Contam Toxicol, 10: 1-8
Ke Z-H (1985) [Clinical features of 47 acute CDM poisonings by
swallowing.] Int Med, 11: 48-59 (in Chinese with English abstract).
Kenyon R, Garcia RJ, O'Grady HM, & Russell PJ (1993) A surveillance
programme for agricultural workers previously exposed to the suspect
bladder carcinogen chlordimeform - Proceedings of the 24th Congress of
the International Commission on Occupational Health, Nice, 26
September-1 October 1993.
Kimbrough RD (1980) Human health effects of selected pesticides,
chloroaniline derivatives. J Environ Sci Health, 15: 977-992.
Kimmel EC, Casida JE, & Ruzo LO (1986) Formamidine insecticides
and chloroacetanilide herbicides: Disubstituted anilines and
nitrosobenzenes as mammalian metabolites and bacterial mutagens.
J Agric Food Chem, 34: 157-161.
Knowles CO (1970) Metabolism of two accaricidal chemicals,
N'-(4-chloro-o-tolyl)-N, N-dimethylformamidine (chlorphenamidine)
and m-(((dimethylamino)methylene)amino)phenyl-carbamate hydrochloride
(formetanate). J Agric Food Chem, 18: 1038-1047.
Knowles CO (1976) Chemistry and toxicology of quinoxaline, organotin,
organofluorine, and formamidine acaricides. Environ Health Perspect,
14: 93-102.
Knowles CO (1991) Chlordimeform. In: Hayes WJ & Laws ER ed. Handbook
of pesticide toxicology. New York, London, San Francisco, Academic
Press Inc., vol 3. pp 1475-1486.
Knowles CO & Benezet HJ (1977) Mammalian metabolism of chlordimeform:
Formation of metabolites containing the urea moiety. J Agric Food
Chem, 25: 1022-1026.
Knowles CO & Johnson TL (1984) Effect of formamidines on
5-hydroxytryptamine uptake and biogenic amine levels in rat platelets.
Toxicology, 31: 91-98.
Knowles CO & Roulston WJ (1972) Antagonism of chlorphenaminine
toxicity to the cattle tick Boophilus microplus by piperonyl
butoxide. J Aust Entomol Soc, 11: 349-350.
Knowles CO & Sen Gupta AK (1969) Photodecomposition of the acaricide
N'-(4-chloro-o-tolyl)N,N-dimethylformamidine. J Econ Entomol,
62: 344-348.
Knowles CO & Sen Gupta AK (1970) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine-14C(Galecron) and 4-chloro-o-toluidine
14C metabolism in the white rat. J Econ Entomol, 63: 856-859.
Knowles CO & Shrivastava SP (1973) Chlordimeform and related
compounds: Toxicological studies with house flies. J Econ Entomol,
66: 75-79.
Konopka EA & Heymann H (1977) Salmonella/microsome mutagenicity test
with chlordimeform. Greensboro, North Carolina, Ciba-Geigy Corporation
(Unpublished report).
Kopp R, Humpel M, Kuhne G, Fuchs R, & Rzadkiewicz M (1977)
Pharmacokinetics of chlordimeform following single and repeated oral
administration of 0.03 and 3 mg in female mice (1.2 and 120 mg/kg).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Kossmann K (1980) Summary report on chlordimeform monitoring
programmes conducted by Ciba-Geigy and Schering at applicator sites in
1979. Berlin, Germany, Schering AG and Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report).
Kossmann K, Geissb�hler H, & Boyd VF (1971) Specific determination of
chlorphenamidine (N'(4-chloro-o-tolyl)-N,N-dimethylformamidine) in
plants and soil material by colorimetry and thin layer and electron
capture gas chromatography. J Agric Food Chem, 19: 360-364.
Krick F (1994) Method for the determination of chlordimeform in honey.
Washington, DC, US Food and Drug Administration (Laboratory
Information Bulletin 3817).
Kurtz PH, Shaw G, Kelter A, & Jackson RJ (1987) Assessment of
potential acute health effects in agricultural workers exposed during
the application of chlordimeform. J Occup Med, 29: 592-595.
Lang R (1984) The mammalian spot test and its use for testing of
mutagenic and carcinogenic potential: experience with the pesticide
chlordimeform, its principal metabolites and the drug lisuride
hydrogen maleate. Mutat Res, 135: 219-224.
Lang R & Adler ID (1982) Studies on the mutagenic potential of the
pesticide chlordimeform and its principal metabolites in the mouse
heritable translocation assay, Mutat Res, 92: 243-248.
Langauer M & M�ller D (1977) C8513 technical : Nucleus anomaly test in
somatic interphase nuclei (Chinese hamster). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 40050577).
Langauer M & M�ller D (1978a) CGA 72651 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 40550577).
Langauer M & M�ller D (1978b) CGA 72647 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 78/3002).
Leander J & MacPhail RC (1980) Effect of chlordimeform (a formamidine
pesticide) on schedule-controlled responding of pigeons. Neurobehav
Toxicol, 2: 315-321.
Leander JD, Tomlinson WT, Balster RL, & MacPhail RC (1984) Some
effects of the formamidine pesticide chlordimeform on the behaviour of
mice. Neurotoxicology, 5: 91-100.
Lemesch C, Wolf Y, & Gabai E (1987) Occupational exposure to
chlordimeform in Israel. Isr J Med Sci, 23: 1261-1262.
Leslie C, Reidy GF, Murray M, & Stacey NH (1988) Induction of
xenobiotic biotransformation by the insecticide chlordimeform, a
metabolite 4-chloro-o-toluidine and a structurally related chemical
o-toluidine. Biochem Pharmacol, 37: 2529-2535.
Li T-D, Liu B-J, & Mei C-J (1982) [Survey on 20 cases of acute
occupational chlordimeform poisoning.] Intermed Med, 3: 23-24
(in Chinese with English abstract).
Li F, Xue S-Z, Wang Y-L, Zhu S-Y, & Hu R-F (1985a) [Carcinogenic
action of chlordimeform given orally to mice in a lifetime
experiment.] Chin J Prev Med, 19: 154-156 (in Chinese with English
abstract).
Li F, Xue S-Z, Zhang R-W, Wang Y-L, Zhu Y-T, Sun S-S, Ding J-L, &
Feng B-J (1985b) (Studies on the health effect of employees in
chlordimeform production, and the mutagenic outcomes.] Labor Med,
2: 11-14 (in Chinese with English abstract).
Li F, Xue S-Z, Jiang X-Z, & Wang Y-L (1986) Report of dermal
carcinogenicity bioassay of chlordimeform in mice. J Ind Hyg Occup
Dis, 6: 76.
Limmer FB (1985) Ciba-Geigy and Schering world-wide exposure data
summary of personnel applying chlordimeform products covering the
period 1980-1984. Basel, Switzerland, Ciba-Geigy Ltd and Berlin,
Germany, Schering AG (Unpublished report).
Lin TH, North HH, & Menzer RE (1975) The metabolic fate of
chlordimeform [N-(4-chloro-o-tolyl) N'N'-dimethylformamidine] human
embryonic lung cell cultures. J Agric Food Chem, 23: 257-258.
Linder RE, Strader LF, Slott VL, & Suarez JD (1992) Endpoints of
spermatotoxicity in the rat after short duration exposures to fourteen
reproductive toxicants. Reprod Toxicol, 6: 491-505.
Ling W-Y, Zhang R-W, Gu Z-Y, Wang X-G, Zhang R-W, Jiang X-Z, Xue S-Z,
& Wang X-Z (1986) [Study on the urinary chlordimeform and its main
metabolite pCoT as index of biological monitoring in occupational
exposure.] Commun Labor Hyg Rail Syst, 8: 14-15 (in Chinese).
Lingren PD, Woffenbarger DA, Nosky JB, & Diaz M Jr (1972) Response of
Campoletis perdistinctus and Apanteles marginiventris to insecticides.
J Econ Entomol, 65: 1295-1300.
Liu Y-Z & Ke Z-H (1985) [Investigation on 53 cases of acute
chlordimeform poisoning.] Res Hyg, 14: 42-45 (in Chinese with English
abstract).
Liu X-L & Li W (1987) [Hemorrhage in upper digestive tract in
chlordimeform poisoning.] Occup Med, 14: 31 (in Chinese with English
abstract).
Liu Z & Mao Z (1980) [Studies on the analysis of metabolites of
pesticides. I. Gas chromatographic analysis of chlordimeform and its
metabolites in urine.] Hua Hsueh Pao, 38: 134-139 (in Chinese with
English Abstract).
Liu T-S & Zhang J-K (1985) [Poisoning due to chlordimeform, a new
pesticide.] Wuhan Med J, 9: 141-143 (in Chinese with English
abstract).
Liu H-G, Liu F-Q. Fang X-Z, & Wang X-M (1990) [Report on one case of
acute anemia due to chlordimeform poisoning.] PLA Med J, 15: 228
(in Chinese with English abstract).
Lu B-Q, Wang S-J, & Pong Y-F (1981) [Studies on the toxicology and
labor health of chlordimeform.] In: Chinese Institute of Scientific
Information and Bureau of Science and Technology ed. [Selected papers
on studies of pesticides.] Beijing, Ministry of Chemistry, pp 781-786
(in Chinese with English abstract).
Lund AE, Hollingworth RM, & Murdock LL (1979a) Formamidine pesticides:
A novel mechanism of action in lepidopterous larvae. Adv Pestic Sci,
3: 465-469.
Lund AE, Hollingworth RM, & Shamkland DL (1979b) Chlordimeform: Plant
protection by a sublethal, noncholinergic action on the central
nervous system. Pestic Biochem Physiol, 11: 117-128.
Lund AE, Hollingworth RM, & Yim GK (1979c) The comparative
neurotoxicity of formamidine pesticides. In: Neurotoxicology of
insecticidal pheromones, pp 119-137.
Lund AE, Shankland DL, Chinn D, & Yim GK (1978a) Similar
cardiovascular toxicity of the pesticide chlordimeform and lidocaine.
Toxicol Appl Pharmacol, 44: 357-365.
Lund AE, Yim GK, & Shankland DL (1978b) The cardiovascular toxicity of
chlordimeform: A local anesthetic-like action. In: Shankland DL,
Hollingworth RM, & Smyth T Jr ed. Pesticide and venom neurotoxicity.
New York, London, Plenum Press, pp 171-177.
Machin MV & Dingle JH (1977) Ultraviolet determination of
chlordimeform in cattle dipping baths and sprays. J Assoc Off Anal
Chem, 60: 696-698.
MacPhail RC & Leander JD (1980) Flavor aversions induced by
chlordimeform. Neurobehav Toxicol, 2: 363-365.
MacPhail RC & Leander JD (1981) Chlordimeform effects on
schedule-controlled behavior in rats. Neurobehav Toxicol Teratol,
3: 19-26.
Maddy KT, Knaak JB, & Gibbons DB (1986) Monitoring the urine of
pesticide applicators in California for residues of chlordimeform and
its metabolites, 1982-1985. Toxicol Lett, 33: 37-44.
Madhukar BV & Matsumura F (1979) Comparison of induction patterns of
rat hepatic microsomal mixed function oxidases by pesticides and
related chemicals. Pestic Biochem Physiol, 11: 301-308
Maitre L & Gfeller W (1975) Influence of compound 8514 (chlordimeform)
on the brain and liver monamineoxidase of rats. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Maitre L, Felner A, Waldmeier P, & Kehr W (1978) Monoamine oxidase
inhibition in brain and liver of rats treated with chlordimeform.
J Agric Food Chem, 26: 442-446.
Maqueda C, Perez Rodriguez JL, Martin F, & Hermosin MC (1983) A study
of the interaction between chlordimeform and humic acid from a typic
chromoxerert soil. Soil Sci, 136: 75-81.
Maqueda C, Morillo E, & Perez Rodriguez JL (1989) Interaction in
aqueous solution of certain pesticides with fulvic acids from a
spodosol soil. Soil Sci, 148: 336-345.
Matsumara F & Beeman RW (1976) Biochemical and physiological effects
of chlordimeform. Environ Health Perspect, 14: 71-82.
Mattern GC, Liu C-H, Louis JB, & Rosen JD (1991) GC/MS and LC/MS
determination of 20 pesticides for which dietary oncogenic risk has
been estimated. J Agric Food Chem, 39: 700-704.
Mayer FL & Ellersieck MR (1986) Manual of acute toxicity:
Interpretation and database for 410 chemicals and 66 species of
freshwater animals. Washington, DC, US Department of the Interior,
Fish and Wildlife Service, 505 pp (Resource Publication 160).
Ming Z-P (1977) [Report of four cases of chlordimeform poisoning,] New
Med, 8: 82-83 (in Chinese with English abstract).
Moore B (1971) Chlordimeform residues - A summary report. Pendle Hill,
Ciba-Geigy Australia Ltd (Unpublished report No. 71/11/358).
Moser VC, McCormick JP, Creason JP, & MacPhail RC (1988) Comparison of
chlordimeform and carbaryl using a functional observational battery.
Fundam Appl Toxicol, 11: 189-206.
Muecke W, Hamboeck H, & Arni P (1979) Metabolic and pharmacokinetic
aspects of chlordimeform in rats and mice. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 37/79).
Murakami M & Fukami J (1974) Effect of chlorphenamide and its
metabolites an HeLa cells. Bull Environ Contam Toxicol, 11: 184-188.
Nathanson JA & Hunnicutt EJ (1981) N-demethylchlordimeform: A potent
agonist of octopamine-sensitive adenylate cyclase. Mol Pharmacol,
20: 68-75.
NCI (1979) Bioassay of 4-chloro-o-toluidine hydrochloride for possible
carcinogenicity. Bethesda, Maryland, National Cancer Institute
(Technical Report Series No. 165).
Neumann R & Voss G (1977) MAO inhibition, an unlikely mode of action
for chlordimeform. Experientia (Basel), 33: 23-24.
Nixon WB & Neal BE (1983) Chlordimeform residues in human urine
following dermal application. Greensboro, North Carolina, Ciba-Geigy
Corporation (Unpublished report No. ABR-83017).
Nui J-H, Ma H, Muo J-T, & Wu W-L (1990) [Report on 4 cases of
poisoning due to chlordimeform and organophosphate pesticides.]
Shang-Dong Ind Hyg, 2: 35 (in Chinese with English abstract).
O'Brien PJ, Elzen GW, & Vinson SB (1985) Toxicity of azinphosmethyl
and chlordimeform to parasitoid braconmellitor (Hymenoptera:
Braxonidae): lethal and reproductive effects. Environ Entomol,
14: 891-894.
Olson KL, Boush GM, & Matsumura F (1978) Behavioral effects of
perinatal exposure of chlordimeform in rats. Bull Environ Contam
Toxicol, 20: 760-768.
Ott MG & Langner RR (1983) A mortality survey of men engaged in the
manufacture of organic dyes. J Occup Med, 25: 763-770.
Palmer WA, Dingle JHP, & Heath AB (1977) Residues of chlordimeform in
bovine tissues and milk following application by a handspray. Aust J
Exp Agric Anim Husb, 17: 380-384.
Parkes HG (1984) Epidemiology of aromatic amine cancers. In: Searle CE
ed. Chemical carcinogens. Washington, DC, American Cancer Society,
pp 277-301 (ACS Monograph No. 182).
Perez-Rodriguez JL & Hermosin MC (1979) Adsorption of chlordimeform by
montmorillonite. In: Mortland MM & Farmer VC ed. Proceedings of the
International Clay Conference, Oxford, 1978. Amsterdam, Oxford, New
York, Elsevier Science Publishers, pp 227-234.
Pfister WR, Chin C, Noland V, & Yim GK (1978a) Similar pharmacological
actions of chlordimeform and local anesthetics. Pestic Biochem
Physiol, 9: 148-156.
Pfister WR, Hollingworth RM, & Yim GW (1978b) Increased feeding in
rats treated with chlordimeform and related formamidines: A new class
of appetite stimulants. Psychopharmacology (Berlin), 60: 47-51.
Phillips JR, Herzog GA, & Nicholson WF (1977) Effect of chlordimeform
on fruiting characteristics and yield of cotton. Ark Farm Res, 26: 4.
Pimley RW (1986) Toxicity of chlordimeform to the tsetse fly Glossina
morsitans. Insect Sci Appl, 7: 663-666.
Popp W & Norpoth K (1991) [Carcinogenic effects of chlordimeform and
4-chloro-o-toluidine.] Dtsch Med Wochenschr, 116: 1454 (in German).
Popp W, Schmieding W, Speck M, Vahrenholz C, & Norpoth K (1992)
Incidence of bladder cancer in a cohort of workers exposed to
4-chloro-o-toluidine while synthesising chlordimeform. Br J Ind Med,
49: 529-531.
Puri E & M�ller D (1983a) C8513 technical: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830304).
Puri E & M�ller D (1983b) CGA 72647 pure: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830311).
Puri E & M�ller D (1983c) C8513 technical: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830306).
Puri E & M�ller D (1983d) CGA 72647 pure: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Lid
(Unpublished report No. 830313).
Rashid KA, Ercegovich CD, & Mumma RO (1984) Evaluation of
chlordimeform and degradation products for mutagenic and DNA-damaging
activity in Salmonella typhimurium and Escherichia coli. J Environ Sci
Health, B 19: 95-110.
Rieger JA, Robinson CP, Cox P, & Horst MA (1981) Cardiovascular
actions and interactions of chlordimeform in the dog. Bull Environ
Contam Toxicol, 27: 707-715.
Rieger JA, Allen LV Jr, & Fisher RC (1985) Extraction and analysis of
chlordimeform and demethylchlordimeform from human issue samples. Bull
Environ Contam Toxicol, 35: 397-405.
Robinson CP & Smith PW (1977) Lack of involvement of monoamine oxidase
inhibition in the lethality of acute poisoning by chlordimeform.
J Toxicol Environ Health, 3: 565-568.
Robinson CP, Smith PW, Zelenski JD, & Endecott BR (1975) Lack of an
effect of interference with amine mechanisms on the lethality of
chlordimeform in the rat. Toxicol Appl Pharmacol, 33: 380-383.
Rose JA (1969a) Degradation of chlorphenamidine by rat liver and
spleen homogenates with particular reference to the possible formation
of azo derivatives. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Rose JA (1969b) Formation of a symmetrical azo-derivative from
4-chloro-o-toluidine by coupled peroxidatic reactions. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Ross MSF & Leisten O (1989) Analysis of trace amounts of chlordimeform
in urine samples. Am Clin Lab, 8: 22-24.
Sachsse K, Suter P, Zak F, & Hess R (1978a) Lifespan feeding study in
mice with N-formyl-4-chloro-o-toluidine - Final report (Project No.
Siss M 4762/1. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Zak F, & Hess R (1978b) Lifespan feeding study in
mice with 4-chloro-o-toluidine-HCI - Final report (Project No. Siss M
04760/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979a)
Chlordimeform-HCL: 60-day feeding study in mice - Final report. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979b)
Chlordimeform-HC1: 60-day feeding study in rat - Final report (Project
No. Siss 6138). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980a)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in the rat - Final
report (Project No. 404078). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980b)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in mice - Final
report (Project No. 404178). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980c) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
chlordimeform-HC1 - Final report (Project No. Siss. R 05658/1). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980d) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
N-formyl-4-chloro-o-toluidine - Final report (project No. Siss
5688/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980e) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
4-chloro-o-toluidine- HC1 - Final report (project No. Siss R 05687/1).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report),
Salvisberg W, Neumann R, & Voss G (1980) Chlordimeform: Mode of toxic
action in various development stages of Spodoptera littoralis. J Econ
Entomol, 73: 193-196.
Sen Gupta AK & Knowles CO (1969) Metabolism of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine by apple seedlings.
J Agric Food Chem, 17: 595-600.
Sen Gupta AK & Knowles CO (1970) Galecron 14C(N'-(4-chloro-o-tolyl)
-N,N-dimethylformamidine) metabolism in the dog and goat. J Entomol,
63: 951-956.
Shimizu T & Fukami J (1983) Effect of chlordimeform and its analogues
on wandering behavior of the cabbage armyworm, Mamestra brassicae L.
(Lepidoptera:Noctuidae). Appl Entomol Zool, 18: 554-557.
Shopp GM Jr, McCay JA, & Holsapple MP (1985) Suppression of the
antibody response by a formamidine pesticide: dependence on the route
of exposure. J Toxicol Environ Health, 15: 293-304.
Sparks TC, Leonard BR. Schneider F. & Graves JB (1993) Ovicidal
activity and alteration of octopamine titers: Effects of selected
insecticides on eggs of the tobacco budworm (Lepidoptera: Noctuidae).
J Econ Entomol, 86: 294-300.
Stasik MJ (1988) Carcinomas of the urinary bladder in a 4-chloro-o-
toluidine cohort. Int Ach Occup Environ Health, 60: 21-24.
Stasik MJ (1991) [Bladder cancer from 4-chloro-o-toluidine.] Dtsch Med
Wochenschr, 116: 1444-1447 (in German).
Stoker TE, Goldman JM, Cooper RL, & McElroy WK (1991) Influence of
chlordimeform on alpha-adrenergic receptor-associated mechanisms of
hormonal regulation in the rat: pituitary and adrenocortical
secretion. Toxicology, 69: 257-268.
Strasser FF & M�ller D (1983a) CGA 72651 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782323).
Strasser FF & M�ller D (1983b) CGA 72647 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Lid (Unpublished report No. 782326).
Strasser FF & M�ller D (1984a) CGA 72647 technical: Point mutation
assay with mouse lymphoma cells, host-mediated assay. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 782325).
Strasser FF & M�ller D (1984b) CGA 72651: Point mutation assay with
mouse lymphoma cells. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782322)
Streibert HP & Dittrich V (1977) Toxicological response of insect eggs
and larvae to a saturated atmosphere of chlordimeform. J Econ Entomol,
70: 57-59.
Strong MB & Bull MS (1985a) Levels of chlordimeform equivalents found
in the urine of personnel involved in the ground rig application of
CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Ltd (Unpublished report
No. 85/6/1030).
Strong MB & Bull MS (1985b) Levels of chlordimeform equivalents found
in the urine of personnel involved in the aerial application of CGS
500 and CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Lid (Unpublished report
No. 85/6/1029).
Su S-T (1977) [Report of 6 cases of chlordimeform poisoning]. Intermed
Med, 6: 19 (in Chinese with English abstract).
Su GC & Zabik MJ (1972) Photochemistry of bioactive compounds:
Photolysis of arylamidine derivatives in water. J Agric Food Chem,
20: 320-323.
Suter P, Luetkemeier H, Sachsse K. Zak F, & Hess R (1976a) 60-Day
feeding study in mice with 4-chloro-o-toluidine HC1 (Project No. Siss
4760). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Suter P, Luetkemeier H, Sachsse K, Zak F, & Hess R (1976b) 60-Day
feeding study in the rat with 4-chloro-o-toluidine HC1 (Project No.
Siss 4760). Basel, Switzerland. Ciba-Geigy Lid (Unpublished report).
Suter P, Zak F, Sachsse K, & Hess R (1978) lifespan feeding study in
mice with chlordimeform-HC1 - Final report (Project No. Siss. M
04761/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Tao X-M, Sun R-M, Wang M-S, Xue S-Z, Zhang R-W, Jiang X-Z & Wang Y-L
(1985) [investigation on the hygienic status of chlordimeform
production in Cai-Qiao pesticide factory.] Chin J Ind Hyg Occup Dis,
3: 272-274 (in Chinese).
Thomas WD, Craig GK, & Stacey NH (1990) Effects of chlordimeform and
its metabolite 4-chloro-o-toluidine on rat splenic T, B and
tumoricidal effector cells. Immunopharmacology, 19: 79-86.
Uk S & Dittrich V (1986) The behavior-modifying effect of
chlordimeform and endosulfan on the adult whitefly Bemisia tabaci
(Genn.) which attacks cotton in the Sudan. Crop Prot, 5: 341-347.
Voss G (1971) Galecron residues - German hops (green cones) and beer
1970. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
42/71).
Voss G & Burkhard N (1971) Galecron residues in milk, meat, fat, liver
and kidney of a Swiss cows. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. RVA 24/7/S).
Voss G, Kossmann K, & Geissb�hler H (1973) Chlordimeform. In: Sherma J
& Zweig G ed. Analytical methods for pesticide plant growth
regulators. New York, London, Academic Press Inc., vol 7, pp 211-230.
Wang X-Z & Tong Q (1992) [Observation on 71 EKG of acute chlordimeform
poisoning cases.] Labor Prot Chem Ind, 13: 61-62 (in Chinese with
English abstract)
Wang CM, Narahashi T, & Fukami J (1975) Mechanism of neuromuscular
block by chlordimeform. Pestic Biochem Physiol, 5: 119-125.
Wang M, Zhou Z-J, Li H-L, Zhang R-W, Xue S-Z, Hong Z-R, Gu Q, Wang
X-M, & Feng Y-Q (1987) [Investigation on the hygienic status of
farmers spraying chlordimeform.] Chin J Ind Hyg Occup Dis, 5: 50-53
(in Chinese)
Wang Y-S & Dong C-R (1985) [Experience on treatment of 25 cases of
chlordimeform poisoning.] Sichuan Med, 6: 207 (in Chinese with English
abstract).
Watanabe H, Tsuda S, & Fukami J (1975) Effect of chlordimeform on
rectus abdominum muscle in frog. Pestic Biochem Physiol, 5:150-154.
Watanabe Y & Matsumura F (1987) Comparative metabolism of sulfamidine
and chlordimeform in rats. J Agric Food Chem, 35: 379-384
Watkinson WP (1985) Effects of chlordimeform on cardiovascular
functional parameters: Part 1. Lethality and arrhythmogenicity in the
geriatric rat. J Toxicol Environ Health, 15: 729-744.
Watkinson WP (1986a) Effects of chlordimeform on cardiovascular
functional parameters: Part 2. Acute and delayed effects following
intravenous administration in the postweanling rat. J Toxicol Environ
Health, 19: 195-206.
Watkinson WP (1986b), Effects of chlordimeform on cardiovascular
functional parameters: Part 3. Comparison of different routes of
administration in the postweanling rat. J Toxicol Environ Health,
19: 207-217.
Watkinson WP & Gordon CJ (1987) Effects of chlordimeform on heart rat
and body temperature of unanesthetized, unrestrained rats. Toxicol
Lett, 35: 209-216.
Watkinson WP, Highfill JW, & Gordon CJ (1989) Modulating effect of
body temperature on the toxic response produced by the pesticide
chlordimeform in rats. J Toxicol Environ Health, 28: 483-500.
Weisburger EK, Russfield AB, Homburger F, Weisburger JH. Boger E, Van
Donger CG, & Chu KC (1978) Testing of twenty-one environmental
aromatic amines for long-term toxicity or carcinogenicity. J Environ
Pathol Toxicol, 2: 325-356.
Wilson AG (1981) Field evaluation of formamidine insecticides and
Bacillus thuringiensis for selective control of Heliothis spp. on
cotton. Gen Appl Entomol, 13: 105-111.
Wiltrout RW, Ercegovich CD, & Ceglowski WS (1978) Humoral immunity in
mice following oral administration of selected pesticides. Bull
Environ Contam Toxicol, 20: 423-431.
Witkin JM & Leander JD (1982) Effects of the appetite stimulant
chlordimeform on food and water consumption of rats: Comparison with
chlordiazepoxide. J Pharmacol Exp Ther, 223: 130-134.
Witkonton S (1973) Fate of the pesticide N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine in fruit and soil, and toxicological properties
of some of its conversion products. Diss Abstr Int, 34: 3831.
Witkonton S & Ercegovich CD (1972) Degradation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine in six different
fruits. J Agric Food Chem, 20: 569-573.
Wolfenbarger DA (1974) Activity of chlordimeform HCI and chlordimeform
against arthropods attacking cotton. J Econ Etomol, 67: 445-446.
Wolfenbarger DA, Gomes ED, Robinson SH, & Parks LH (1979)
Chlordimeform residues from cotton leaves. J Econ Entomol,
72: 128-130.
Worthing CR (1979) The pesticide manual: A work compendium, 6th ed.
Croydon, United Kingdom, British Crop Protection Council, p 95.
Worthing CR & Walker SB (1983) The Pesticide manual. 7th ed. Croydon,
United Kingdom, British Crop Protection Council, p 2270.
Wu G (1982) [One case of chlordimeform poisoning.] Chin J Prev Med,
17: 275 (in Chinese with English abstract).
Wu S-Y, Liu Y-Y, Huang W-Z, Hong Q-F, Ling R-F, & Wang J-Y (1983) [On
chlordimeform poisoning - 24 case reports.] J Fujian Med Coll, 17:
42-44 (in Chinese with English abstract).
Wu D, Costa LG, & Murphy SD (1990) [The effect of pesticides-
chlordimeform and amitraz on inhibition of the binding of
3H-clonidine to alpha2-adrenoreceptor in rat forebrain tissue.]
Huaxi Yike Daxue Xuebao, 21: 17-21 (in Chinese with English abstract).
Xia S-C & Gao X-Z (1977) [Rescue after chlordimeform poisoning.]
Intermed Med, 6: 18-19 (in Chinese with English abstract).
Xie P-F (1983) [Analysis on the clinical features of chlordimeform
poisoning.] J Pract Med, 3: 145-146 (in Chinese with English
abstract).
Xu S-H (1987) [Experiences on treatment of chlordimeform poisoning.]
Hu-Nan Med, 4: 168-167 (in Chinese with English abstract).
Xue S-Z & Loosli R (1994) Formamidines. Toxicology, 91: 99-104.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J. & Wang YL (1990) The risk
assessment of occupational exposure to chlordimeform. Presented at the
23rd International Congress on Occupational Health, Montreal, Canada,
September 1990.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J, & Wang YL (1991)
[Assessment on the carcinogenic risk of chlordimeform.] Environ Chem,
10: 54-60 (in Chinese).
Yang Q-Z (1984) [Rescue of a case of cardiac arrest due to
chlordimeform poisoning.] Jiangsu Med, 10: 38 (in Chinese with English
abstract).
Yim GK, Holsapple MP, Pfister WR, & Hollingworth RM (1978)
Prostaglandin synthesis inhibited by formamidine pesticides. Life Sci.
23: 2509-2516.
Youngman RR, Leigh TF, Kerby TA, Toscano NC, & Jackson CE (1990)
Pesticides and cotton: Effect on photosynthesis, growth, and fruiting.
J Econ Entomol, 83: 1549-1557.
Zak F, Sachsse K, Bath R, & Hess R (1973) One-year feeding study on
the rat with technical C-8514 (Project No. Siss 553). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Zhang Y-J, Yuan B-Q, Jin G-L, & Liu Y-Z (1976) [Report on two severe
cases of chlordimeform poisoning and their treatment.] J Rural Doctor,
5: 19 (in Chinese with English abstract).
Zhang R-W, Lin W-Y, Wang X-G, Gu Z-Y, Qu J-Y, Xue S-Z & Wang Y-L
(1986a) [Health effects of chlordimeform on the applicators.] Chin J
Prev Med, 20: 186 (in Chinese with English abstract).
Zhang R-W, Xue S-Z, & Wang Y-L (1986b) [Measurement of total amount of
body surface contamination with regional proportional sampling.] Occup
Med, 13: 2-3 (in Chinese with English abstract).
Zhou D-J (1987) [Bitter lesson on misuse of large dose of atropine on
chlordimeform poisoning.] Hu-Nan Med, 4: 169 (in Chinese with English
abstract).
Zimmer D, Mazurek J. Petzold G. & Bhuyan BK (1980). Bacterial
mutagenicity and mammalian cell DNA damage by several substituted
anilines. Mutat Res. 77: 317-326.
R�SUM�
1. Identit�, propri�t�s physiques et chimiques et m�thodes d'analyse
Le chlordim�form est une base de force moyenne qui forme des sels
stables avec les acides forts. A l'�tat pur, le chlordim�form et son
chlorhydrate sont des solides cristallins incolores. Le point de
fusion du chlordim�form base est de 32�C, celui du chlorhydrate �tant
de 225-227�C. La base est l�g�rement soluble dans l'eau (250 mg/litre)
et facilement soluble dans les solvants organiques, tandis que le
chlorhydrate est facilement soluble dans l'eau mais plus difficilement
dans les solvants organiques. La tension de vapeur du chlordim�form
est de 48 mPa � 20�C et son coefficient de partage entre l'eau et
l'octanol (log Kow) est �gal � 2,89. On peut faire appel � de
nombreuses m�thodes d'analyse pour la recherche et le dosage du
chlordim�form dans les v�g�taux, le sol, l'eau et l'urine.
2. Sources d'exposition humaine et environnementale
Le chlordim�form n'existe pas � l'�tat naturel. On le pr�pare
industriellement par condensation du r�actif de Vilsmeier (obtenu par
r�action du dim�thylformamide sur POCl3, SOCl2 ou COCl2) soit avec
la 4-chloro- o-toluidine, soit avec la l' o-toluidine, suivie d'une
chloration du d�riv� interm�diaire obtenu. On l'utilise comme
acaricide � large spectre et il est principalement actif contre les
formes mobiles des acariens et des tiques ainsi que contre les oeufs
et les premiers stades de certains l�pidopt�res. Il agit en phase
gazeuse aussi bien que par contact. Les premiers temps de son
utilisation, on l'employait pour traiter des cultures tr�s vari�es
comme les fruits � p�pins, les drupes, les choux et autres l�gumes,
les raisins, le houblon, les agrumes, les cerises et les fraises. On
l'utilise aussi en bains contre les tiques des bovins. Ces derni�res
ann�es son usage s'est g�n�ralement limit� au coton, mais on continue
tout de m�me � l'utiliser sur le riz dans certains pays. Depuis
1988/89 il n'est plus homologu� dans la plupart des pays. En Chine, la
production a cess� en 1992, de m�me que la vente en 1993.
3. Transport, distribution et transformation dans l'environnement
Bien que sa tension de vapeur ait une valeur moyenne, le
chlordim�form ne s'�vapore pas autant qu'on le penserait des surfaces
v�g�tales. Sa stabilit� vis-�-vis de l'hydrolyse d�pend fortement du
pH; il est stable en milieu acide mais s'hydrolyse rapidement en
milieu alcalin. Le chlordim�form est capable de s'adsorber sur les
mati�res organiques dissoutes.
Dans le sol, la disparition du chlordim�form est essentiellement
imputable � l'action des microorganismes et, pour une moindre part, �
l'hydrolyse chimique. Malgr� la solubilit� du compos� dans l'eau, on
ne trouve gu�re de traces de lessivage, ce qui peut s'expliquer par
une adsorption aux mat�riaux argileux ou aux mati�res organiques du
sol ainsi que par la biod�gradation. Les principaux m�tabolites sont
la N-formyl-4-chloro- o-toluidine et la 4-chloro- o-toluidine.
Les plantes fixent le chlordim�form pr�sent dans le sol en
proportion faible mais mesurable et la concentration est suffisante
pour affecter les ravageurs qui se nourrissent � leurs d�pens. En
traitement foliaire, la p�n�tration du chlordim�form dans la cuticule
est limit�e. Le chlordim�form est rapidement d�compos� par les
v�g�taux. Les principaux m�tabolites sont le d�m�thylchlordim�form, la
N-formyl-4-chloro -o-toluidine et la 4-chloro -o-toluidine, cette
derni�re n'�tant pas produite par toutes les plantes.
Dans le sol, le chlordim�form et ses m�tabolites disparaissent
selon une cin�tique du premier ordre avec une demi-vie de 20 � 40
jours.
Les �tudes de bioaccumulation montrent que les organismes
aquatiques ne fixent qu'une petite quantit� de chlordim�form et que
celui-ci s'�limine rapidement une fois ces organismes replac�s en eau
pure.
4. Concentrations dans l'environnement et exposition humaine
On n'a pas proc�d� � des mesures de concentration dans l'air ou
l'eau. Apr�s traitement de rizi�res, on a retrouv� des r�sidus allant
jusqu'� 2900 �g/kg dans les 5 premiers centim�tres du sol et jusqu'�
150 �g/kg dans les 5 centim�tres suivants.
On a fix� des teneurs limites en r�sidus pour un grand nombre de
produits crus et dans certains cas, pour des pr�parations contenant
ces produits. Les limites maximales de r�sidus fix�es par le Codex ont
�t� supprim�es.
Il y a eu des cas d'exposition au chlordim�form au cours de la
pr�paration, de la formulation et de l'�pandage de ce produit. Depuis
quelques ann�es, on utilise la concentration urinaire totale du
chlordim�form et de ses m�tabolites pour surveiller l'exposition et il
y a d'ailleurs une bonne corr�lation entre cette concentration et le
degr� de contamination cutan�e. Dans les industries cotonni�res o�
l'on a soumis les ouvriers agricoles � une surveillance g�n�rale de la
concentration urinaire en chlordim�form, on a constat� que les plus
expos�s �taient les chargeurs, les laveurs et les m�caniciens et les
moins expos�s les signaleurs et les pilotes.
5. Cin�tique et m�tabolisme chez les animaux de laboratoire et
l'Homme
Chez les mammif�res, le chlordim�form est facilement r�sorb� au
niveau des voies digestives ainsi que par la voie transcutan�e. Il est
ensuite rapidement excr�t� � raison de 80% environ dans l'urine et de
10-15% dans les mati�res f�cales. De petites quantit�s de r�sidus sont
pr�sentes au bout de 10 jours dans tous les tissus mais rien n'indique
qu'il y ait bioaccumulation. Apr�s application cutan�e chez l'Homme,
on constate �galement une excr�tion urinaire rapide.
On retrouve dans l'urine plusieurs m�tabolites du chlordim�form
sous forme oxyd�e et conjugu�e, � savoir principalement la
N-formyl-4-chloro- o-toluidine, et la 4-chloro- o-toluidine.
In vitro, on retrouve les m�mes m�tabolites, mais avec pr�dominance
de la 4-chloro- o-toluidine.
6. Effets sur les mammif�res de laboratoire et les syst�mes d'�preuve
in vitro
Les �preuves pratiqu�es sur un certain nombre d'esp�ces montrent
que la chlordim�form pr�sente une toxicit� aigu� mod�r�e par la voie
orale et la voie transcutan�e. Chez le rat, les principaux m�tabolites
sont peu toxiques par voie orale. Chez le lapin, le chlordim�form ne
provoque qu'une l�g�re irritation oculaire et cutan�e. Apr�s
exposition de courte ou de br�ve dur�e au chlordim�form ou � ses
m�tabolites, on peut observer, au niveau des constantes
h�matologiques, des modifications qui sont imputables au traitement et
on constate, � dose �lev�e, certains signes qui d�notent une
hyperplasie de l'�pith�lium des canaux biliaires et de la vessie. Il
n'y pas d'accroissement de la fr�quence des tumeurs chez le rat. Chez
la souris, on observe, apr�s administration par voie alimentaire de
chlordim�form, de N-formyl-4-chloro -o-toluidine ou de
4-chloro- o-toluidine, une augmentation, li�e � la dose, des tumeurs
malignes h�morragiques d'origine vasculaire appartenant � la classe
des h�mangio-endoth�liomes, dont la pr�sence entra�ne un accroissement
de la mortalit� parall�le � la dose.
Le chlordim�form n'a pas d'effet ind�sirable sur les diff�rents
aspects de la fonction de reproduction et il n'a aucun pouvoir
t�ratog�ne.
Le chlordim�form a fait l'objet d'un grand nombre d'�preuves de
g�notoxicit� in vitro et in vivo. Aucune d'elles n'a donn� de
r�sultat positif, �tant entendu qu'il s'agissait de la mati�re active
et non de formulations. Par ailleurs, un certain nombre d'observations
sporadiques non confirm�es font �tat d'une activit� mutag�ne induite
par la N-formyl-4-chloro- o-toluidine et par la 4-chloro- o-
toluidine. Il n'existe qu'une seule description de transformations
cellulaires provoqu�es par le chlordim�form et par la 4-chloro -o-
toluidine. Chez des souris et des rats trait�s par le chlordim�form,
on a constat� une que le compos� se liait � l'ADN des cellules
h�patiques. A dose beaucoup plus �lev�e, il se forme chez les m�mes
animaux un important adduit hydrophobe.
Le chlordim�form provoque des effets pharmacologiques et
biochimiques divers chez l'animal, et notamment des effets
cardiovasculaires, une hypothermie, une hyperexcitabilit�, une
modification des fonctions visuelle et auditive ainsi que la
modulation des amines biogen�se et des enzymes pharmacom�tabolisantes.
7. Effets sur l'Homme
Les intoxications aigu�s se traduisent par une fatigue, des
naus�es et une perte d'app�tit, avec, dans les cas graves, somnolence,
cyanose, besoin imp�rieux d'uriner, cystite, effets cardiovasculaires
(tachy-cardie, bradycardie, anomalies de l'ECG), coma et �tat de choc.
En g�n�ral, la r�cup�ration est totale.
Apr�s une exposition de longue dur�e au chlordim�form, on peut
observer encore d'autres sympt�mes tels que des douleurs abdominales,
des d�mangeaisons et des �ruptions (en cas d'exposition cutan�e)
accompagn�s d'une h�maturie macroscopique ou micro-scopique. On a
signal� de nombreux cas d'intoxication pr�sentant des sympt�mes
d'exposition de longue dur�e parmi les ouvriers d'unit�s de production
de chlordim�form et des ouvriers agricoles.
Les donn�es �pid�miologiques obtenues � la suite de cas
d'exposition professionnelle montrent qu'il existe une forte
corr�lation entre l'exposition � la 4-chloro -o-toluidine et le
cancer de la vessie. En revanche, on n'a gu�re obtenu d'�l�ments qui
militeraient en faveur d'une association entre ce type de cancer et
l'exposition au chlordim�form.
8. Effets sur les autres �tres vivants au laboratoire et dans leur
milieu naturel
Apr�s �pandage de chlordim�form sur le sol, on n'a pas observ�
d'effets sensibles sur les populations de champignons, de bact�ries ou
d'actinomyc�tes terricoles.
Il n'existe pas de donn�es toxicologiques de laboratoire
concernant les invert�br�s dul�aquicoles. En pr�sence de
chlordim�form, il y a inhibition de la croissance des larves
d'hu�tres, avec une CE50 de 5,7 mg/litre. Pour la crevette rose,
le seul crustac� �tudi�, la CL50 � 96 h a �t� trouv�e �gale �
7,1 mg/litre et des valeurs allant de 1 � 54 mg/litre ont �t� obtenues
pour le m�me param�tre chez les poissons. On ne poss�de aucune donn�e
relative � la toxicit� chronique pour les esp�ces aquatiques.
L'ensemble des r�sultats de laboratoire et des donn�es recueillies sur
le terrain indique que le compos� est toxique pour de nombreux
arthropodes terrestres non vis�s.
Chez l'abeille, la toxicit� de contact se traduit par une DL50
de 120 �g/g, la toxicit� par voie orale correspondant � une valeur de
187 �g/g. Trois heures apr�s l'�pandage de chlordim�form sur de la
luzerne, l'exposition de certaines esp�ces d'abeilles aux r�sidus
encore pr�sents sur les plantes, n'a provoqu� aucune mortalit�.
La CL50 par voie alimentaire varie de >1000 � > 5000/kg de
nourriture pour diverses esp�ces d'oiseaux.
9. Evaluation des risques pour la sant� humaine et des effets sur
l'environnement
On a observ� des signes d'intoxication aigu� chez des
travailleurs qui, peut-�tre par suite de l'inobservation des mesures
de s�curit�, avaient �t� fortement expos�s � du chlordim�form au cours
de la pr�paration ou de l'utilisation de ce produit. Comme, � ce qu'il
semble, il n'est plus produit ni utilis� nulle part dans le monde, il
ne devrait plus y avoir de cas d'intoxication aigu�. Le risque li� �
une exposition chronique et en particulier, le risque de cancer de la
vessie, subsistera cependant pendant de nombreuses ann�es. Il faut
continuer � effectuer des contr�les sanitaires chez les sujets qui ont
subi une exposition notable pour avoir travaill� dans des ateliers de
production de chlordim�form ou avoir v�cu dans des zones rurales o� le
produit �tait largement utilis�.
Comme il s'agit d'un produit qui n'est plus en usage, on n'a pas
proc�d� � une �valuation quantitative du risque qu'il repr�sente pour
l'environnement. On ne pense pas que celui-ci puisse subir des effets
nocifs � long terme qui soient attribuables � l'utilisation ant�rieure
du produit.
10. Conclusions et recommandations
Il existe un risque non n�gligeable que le chlordim�form produise
des effets toxiques imm�diats ou � plus long terme chez les individus
expos�s. Les donn�es disponibles accr�ditent l'id�e d'une association
entre l'augmentation de l'incidence du cancer de la vessie chez
l'homme et l'exposition � la 4-chloro- o-toluidine et, dans une
moindre mesure, au chlordim�form.
Le chlordim�form ne persiste pas dans l'environnement et il ne
devrait donc pas y avoir d'effets nocifs � long terme sur celui-ci qui
r�sulteraient de l'usage ant�rieur du compos�.
Il n'est pas recommand� de reprendre la production ou l'usage du
chlordim�form dans un but commercial. Les stocks existants doivent
�tre �limin�s selon les r�gles de s�curit�.
Les personnes expos�es au chlordim�form de par leur profession
doivent �tre soumises � des examens cytologiques v�sicaux et � une
recherche syst�matique de l'h�maturie dans le cadre d'un programme
g�n�ral de d�pistage.
RESUMEN
1. Identidad, propiedades f�sicas y qu�micas y m�todos anal�ticos
El clordimeformo es una base de fuerza media que forma sales
estables con �cidos fuertes. Tanto el clordimeformo como su sal
hidroclorada en estado puro son s�lidos cristalinos incoloros. El
punto de fusi�n del clordimeformo (base) es de 32�C, mientras que el
de la sal hidroclorada es de 225-227�C. El clordimeformo (base) es
poco soluble en agua (250 mg/litro) y f�cilmente soluble en
disolventes org�nicos, mientras que la sal hidroclorada es f�cilmente
soluble en agua pero menos soluble en disolventes org�nicos. El
clordimeformo (base) tiene una presi�n de vapor de 48 mPa a 20�C y un
log Kow de 2,89. Se dispone de una amplia gama de m�todos anal�ticos
para detectar y cuantificar la presencia de clordimeformo en las
plantas, el suelo, el agua y la orina.
2. Fuentes de exposici�n humana y ambiental
El clordimeformo no existe en la naturaleza. Se produce
comercialmente mediante condensaci�n del reactivo de Vilsmeier
(obtenido por reacci�n de la dimetilformamida con POCl3, SOCl2
o COCl2) con 4-cloro- o-toluidina o bien con o-toluidina y
cloraci�n ulterior del producto intermedio resultante. Se ha utilizado
como acaricida de amplio espectro y act�a principalmente contra las
formas m�viles de �caros y garrapatas, as� como contra los huevos y
las cris�lidas en estado inicial de algunos insectos del orden
Lepid�ptera. Es activo en la fase de vapor, as� como por contacto.
Cuando comenz� a utilizarse, se aplicaba a productos de una amplia
variedad de cultivos, tales como frutas de pipas, frutas de hueso,
berzas, hortalizas, uvas, l�pulo, c�tricos, manzanas, peras, cerezas y
fresas. Tambi�n se utilizaba en ba�os antiparasitarios para combatir
las garrapatas del ganado. En los �ltimos a�os, su uso se limitaba por
lo general al algod�n, aunque en algunos pa�ses se segu�a aplicando al
arroz. En la mayor�a de los pa�ses, su registro se abandon�
voluntariamente en 1988/1989. En China dej� de producirse en 1992 y de
venderse en 1993.
3. Transporte, distribuci�n y transformaci�n en el medio ambiente
El clordimeformo tiene una presi�n de vapor moderada pero su
evaporaci�n de la superficie de las plantas es inferior a la que
cabr�a prever. La estabilidad hidrol�tica del clordimeformo depende
mucho del pH; es estable en condiciones �cidas pero se hidroliza
r�pidamente en condiciones alcalinas. El clordimeformo tiene un
potencial de adsorci�n a la materia org�nica disuelta.
Hay dispersi�n del clordimeformo en el suelo, principalmente por
acci�n microbiana y, en menor medida, por hidr�lisis qu�mica. Pese a
la solubilidad del clordimeformo en agua, hay pocos indicios de
lixiviaci�n, lo que puede deberse a su adsorci�n a minerales
arcillosos y a la materia org�nica del suelo, as� como a su
biodegradaci�n. Los principales metabolitos son la N-formil-
4-cloro- o-toluidina y la 4-cloro- o-toluidina.
La absorci�n del clordimeformo por las plantas a partir del suelo
es escasa pero detectable, y suficiente para afectar a las plagas que
se alimentan de ellas. El clordimeformo aplicado a las hojas s�lo
tiene una capacidad limitada de penetrar en las capas cuticulares. El
clordimeformo se degrada r�pidamente en las plantas. Sus principales
metabolitos son el demetilclordimeformo, la N-formil-4-cloro-
o-toluidina y la 4-cloro- o-toluidina, aunque no todas las plantas
estudiadas produjeron 4-cloro- o-toluidina.
El clordimeformo y sus metabolitos se dispersan en el suelo
conforme a una cin�tica de primer orden, con una semivida de 20-40
d�as.
Los estudios sobre bioacumulaci�n indican una escasa absorci�n
del clordimeformo por los organismos acu�ticos y una r�pida depuraci�n
de �stos despu�s de haber sido transferidos a un agua limpia.
4. Niveles medioambientales y exposici�n humana
No se han medido los niveles de clordimeformo en el aire ni en el
agua. Tras la aplicaci�n de clordimeformo a unos arrozales, en el
suelo se hallaron residuos en concentraciones de hasta 2900 �g/kg en
los 5 cm primeros de profundidad, y de 150 �g/kg en los 5 cm
siguientes.
Se establecieron niveles m�ximos de residuos aplicables a una
amplia variedad de productos sin elaborar y, en algunos casos, de
residuos trasladados a los alimentos elaborados. Los l�mites m�ximos
aplicables a los residuos de clordimeformo se han retirado del Codex
Alimentarius.
Hab�a exposici�n ocupacional al clordimeformo durante la
fabricaci�n, la formulaci�n y la aplicaci�n del producto. En los
�ltimos a�os la exposici�n se ha vigilado mediante la determinaci�n de
los niveles totales de clordimeformo y de sus metabolitos presentes en
la orina, y hay una correlaci�n positiva entre el nivel en la orina y
el grado de contaminaci�n cut�nea. Entre los trabajadores agr�colas de
los algodonales sometidos a una amplia vigilancia de la excreci�n
urinaria de clordimeformo, los niveles m�s altos de exposici�n se
hallaban en los cargadores, lavadores y mec�nicos, y los niveles m�s
bajos en los obreros se�alizadores y pilotos.
5. Cin�tica y metabolismo en animales de laboratorio y en el ser
humano
Los mam�feros absorben f�cilmente el clordimeformo por el tracto
gastrointestinal y a trav�s de la piel. Lo excretan r�pidamente,
alrededor del 80% por la orina y del 10-15% por las heces. Al cabo de
unos 10 d�as se observan niveles bajos de residuos en todos los
tejidos y no hay indicios de bioacumulaci�n. Tras la administraci�n
cut�nea a seres humanos, se observa una excreci�n r�pida semejante por
la orina.
Varios metabolitos oxidados y conjugados del clordimeformo se
excretan por la orina; los principales son el demetilclordimeformo, la
N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. En
estudios in vitro se han observado los mismos metabolitos, siendo el
principal la 4-cloro- o-toluidina.
6. Efectos en mam�feros de laboratorio y en sistemas de pruebas
in vitro
En ensayos realizados en varias especies, el clordimeformo
administrado por v�a oral y cut�nea ha mostrado tener una toxicidad
aguda moderada. Los principales metabolitos han mostrado tener una
toxicidad oral baja en ensayos realizados en ratas. El clordimeformo
provoca solamente una ligera irritaci�n cut�nea y ocular en el conejo.
Tras una exposici�n breve o prolongada de ratones y ratas al
clordimeformo o a sus metabolitos pueden observarse cambios asociados
al tratamiento en los par�metros hematol�gicos y, con dosis elevadas,
indicios de hiperplasia del epitelio de las v�as biliares y de la
vejiga. El clordimeformo no aumenta la incidencia de tumores en las
ratas. En los ratones, despu�s de administrar a trav�s de la dieta
clordimeformo N-formil-4-cloro- o-toluidina o 4-cloro- o-
toluidina, se observa, de forma relacionada con la dosis, un aumento
de los tumores malignos hemorr�gicos de origen vascular clasificados
como hemangioendoteliomas malignos, que producen un aumento de la
mortalidad asociado con la dosis.
El clordimeformo no afecta a los par�metros reproductivos ni
tiene potencial teratog�nico.
Se ha ensayado el clordimeformo en una amplia variedad de pruebas
de genotoxicidad in vitro e in vivo. No se han comunicado
reacciones positivas a ninguna de esas pruebas, en las que se ensay�
clordimeformo en estado puro. Adem�s, se han comunicado varios
informes espor�dicos y no confirmados de actividad mutag�nica inducida
por la N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. Un
informe describe una inducci�n de la transformaci�n celular por efecto
tanto del clordimeformo como de la 4-cloro- o-toluidina. En el h�gado
de los ratones y las ratas expuestos se producen enlaces con el ADN.
Se ha observado un importante aducto hidrof�bico, en los ratones en
niveles mucho mayores que en las ratas.
El clordimeformo induce diversos efectos farmacol�gicos y
bioqu�micos en los animales, tales como cambios cardiovasculares,
hipotermia, hiperexcitabilidad, efectos sobre las funciones visual
central y auditiva y modulaci�n de las aminas biog�nicas y de las
enzimas que metabolizan f�rmacos.
7. Efectos en el ser humano
La intoxicaci�n aguda causa fatiga, n�useas, p�rdida del apetito
y, en casos m�s graves, somnolencia, cianosis, micci�n imperiosa,
cistitis, efectos cardiovasculares (taquicardia, bradicardia,
alteraciones del ECG), coma y choque. En general se produce una
recuperaci�n completa de la intoxicaci�n aguda.
Otros s�ntomas asociados a la exposici�n cr�nica al
clordime-formo son dolores abdominales, prurito y exantemas
(exposici�n cut�nea), as� como hematuria macrosc�pica y microsc�pica.
Se ha comunicado un gran n�mero de casos con s�ntomas cl�nicos de
exposici�n cr�nica tanto entre los obreros de las plantas de
producci�n de clordimeformo como entre los trabajadores agr�colas.
Los indicios epidemiol�gicos relacionados con la exposici�n
ocupacional muestran una fuerte asociaci�n entre la exposici�n al
metabolito 4-cloro- o-toluidina y la incidencia de c�ncer de vejiga
en el ser humano. Actualmente se dispone de pocos indicios de
asocia-ci�n entre la exposici�n al clordimeformo y el c�ncer de vejiga
en el ser humano.
8. Efectos en otros organismos en el laboratorio y en el medio
ambiente
No se observaron efectos significativos en poblaciones de hongos
de la tierra, bacterias o actinomicetos tras la aplicaci�n de
clordime-formo al suelo.
No existen datos de laboratorio sobre la toxicidad en los
invertebrados de agua dulce. El clordimeformo inhibi� el crecimiento
de larvas de ostras, con una CE50 de 5,7 mg/litro. La CL50 a las
96-h para los camarones rosados, �nico crust�ceo estudiado, fue de
7,1 mg/litro y los valores de la CL50 a las 96-h para los peces
oscilaron entre 1 y 54 mg/litro. No se dispone de datos sobre
toxicidad acu�tica cr�nica. La combinaci�n de datos obtenidos en el
laboratorio y sobre el terreno revela que el clordimeformo es t�xico
para una amplia gama de artr�podos terrestres no combatidos.
Con respecto a las abejas, se ha comunicado una DL50 de
toxicidad por contacto de 120 �g/g y una DL50 de toxicidad oral de
187 �g/g. No se produjo mortalidad sobre el terreno tras la exposici�n
de especies de abejas a los residuos presentes en la alfalfa tres
horas despu�s del rociado.
La CL50 en la dieta de varias especies de p�jaros oscil� entre
>1000 y >5000 mg/kg de dieta.
9. Evaluaci�n de los riesgos para la salud humana y efectos en el
medio ambiente
La exposici�n intensa durante la producci�n o la utilizaci�n,
debida posiblemente a la insuficiencia de las medidas de seguridad,
dio lugar a s�ntomas de intoxicaci�n aguda en los trabajadores. Como
se ha notificado que se ha suspendido la producci�n y la utilizaci�n
de clordimeformo en todo el mundo, no deber�an producirse nuevos casos
de intoxicaci�n aguda. Sin embargo, el riesgo asociado a la exposici�n
cr�nica, en particular el riesgo de c�ncer de vejiga, seguir� siendo
preocupante durante muchos a�os. Deber�a proseguir el reconoci-miento
m�dico de las personas que han estado muy expuestas en las plantas de
producci�n y en las comunidades rurales donde se haya aplicado
extensamente el clordimeformo.
Dado que el clordimeformo ha dejado de utilizarse, no se ha
realizado ninguna evaluaci�n cuantitativa de los riesgos para el medio
ambiente. A largo plazo no se prev�n efectos perjudiciales para el
medio ambiente como consecuencia de la utilizaci�n de clordime-formo
en el pasado.
10. Conclusiones y recomendaciones
El clordimeformo tiene un potencial significativo para causar
tanto toxicidad inmediata como a largo plazo en las personas
expuestas. La informaci�n de que se dispone actualmente apunta a una
asociaci�n entre una mayor incidencia de c�ncer de vejiga en el ser
humano y la exposici�n a la 4-cloro- o-toluidina y, en menor medida,
al clordimeformo.
El clordimeformo no persiste en el medio ambiente, por lo que a
largo plazo no se prev�n efectos perjudiciales como consecuencia de su
utilizaci�n en el pasado.
Se recomienda que el clordimeformo no se produzca comercialmente
ni se utilice en el futuro. Las reservas existentes deber�an
eliminarse sin correr riesgos.
Las personas expuestas profesionalmente al clordimeformo deber�an
participar en un programa de reconocimiento m�dico que comprenda
citolog�a urinaria y detecci�n de hematuria.
8. EFFECTS ON HUMANS
8.1 General population exposure
8.1.1 Acute poisoning incidents
The most comprehensive data on acute poisoning cases associated
with exposure to chlordimeform has come from China. Details of these
published poisoning cases are shown in Table 16. While many were due
to intentional ingestion, there were also cases of unintentional
poisoning as a result of consumption of contaminated food, as well as
occupational exposure to the spray. In a brief report prepared by Deng
et al. (1984) of a 1983 symposium in Hu-bei Province on chlordimeform
poisoning, which featured some 29 papers and 859 case studies, it was
stressed that the main cause of death was suppression of cardiac
contracture and dilation of blood vessels resulting in circulatory
failure.
Arima et al. (1976) described an unsuccessful suicide attempt
involving a 76-year-old male who ingested 100 g chlordimeform. He
vomited several times before arriving at hospital 50 min after
ingestion. He was lethargic with a weak pulse and cyanosis associated
with his lips, nails and skin. Methaemoglobin levels represented 17%
of total haemoglobin at 5 h but returned to normal levels by 2 days.
He regained consciousness by 50 h, although complained of headache and
blurred vision. The only treatment received was gastric lavage, which
was performed shortly after his arrival at the hospital.
8.2 Occupational exposure
8.2.1 Acute poisoning incidents
Currie (1933) reported nine cases of haematuria in workers
exposed to 4-chloro- o-toluidine (erroneously called 5-chloro- o-
toluidine) by inhalation or possibly by absorption through the skin.
All patients had difficulty urinating and had suprapubic pain. Most of
the workers were exposed to the material for only 1-2 days. Despite
efforts to control exposure to the chemical in the factory, further
cases of poisoning occurred, and manufacture was ceased. In a
follow-up study of three of the nine cases after 3 years, one patient
had no bladder trouble, one had a slight cystitis and urethritis, and
one had carcinoma of the bladder.
Jurincic et al. (1991) reported cases of acute haemorrhagic
cystitis in two men (aged 19 and 50) following involvement in cleaning
of a water-tank that had likely been used to transport chlordimeform.
Both developed abdominal pain, dysuria and haematuria in the evening
following exposure. Cystoscopy revealed haemorrhagic cystitis,
which was confirmed by bladder mucosa biopsy. Serum levels of
4-chloro- o-toluidine (referred to as 4-chloro-2-methylaniline) were
>1 mg/litre in both patients and urine levels were 16 mg/litre in the
Table 16. Case studies of acute chlordimeform poisoning in China
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
1 71 4 dermal absorption 5 ECG: 26 tachycardia; 6 bradycardia; 11 ectopic Wang & Tong (1992)
(28 male, 67 ingestion rhythm; 6 premature beat; 2 atrial fibrillation;
43 female) ventricular fibrillation; 1"Torsade de Pointes";
2 high pike P, 6 A-V block, 17 S-T depression,
3 inverse T, 1 S-T elevation ,7 Q-T elongation.
In 33 severe cases, 28 has ECG abnormalities;
38 moderate cases, 14 had abnormal ECG. Changes
in heart were found in 32 cases. Deaths were from
respiratory failure (3); ventricular fibrillation
(1) and supraventicular tachycardia (1).
2 4 ingestion(?) 0 Mild cyanosis, cystitis (2 cases occupational, Nui et al. (1990)
2 cases non-occupational; OPs also in formulation.
3 1 female ingestion 0 Jaundice on 3rd day which progressively deepened. Liu et al. (1990)
(30 years old) (150 ml) Hb 40 g/litre (70 g/litre on admission); complete
recovery, discharged on day 20.
4 52 ingestion 0 Loss of appetite (86.5%), urgency in urination He (1989)
(19 male, (20-350 ml) (84.6%), cyanosis (81.1%), coma (67.3%), miosis
33 female) (34.6%), mydriasis (15.4%),hypotension (38.5%),
tachycardia (32.7%), bradycardia (3.8%).
Impairment of liver and renal functions. 15 ECGs:
7 tachycardia, 2 bradycardia, Q-T elongation,
8 T-wave changes. Treated with methylene blue,
vitamin C, fresh blood transfusion and sopolamine.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
5 35 ingestion(?) 0 18 severe cases. Suggested use of 5-36 mg He et al. (1987)
atropine for chlordimeform poisoning and
50-128 mg for mixed pesticide poisoning.
6 1 female ingestion 0 Cyanosis, pin-point myosis. Given atropine Zhou (1987)
(30 years old) (80 ml 25% (15 mg/min) after lavage until total of 530 mg.
chlordimeform) Symptoms indicated overdose of atropine. Methylene
blue given, recovery and discharge at day 7.
7 23 4 contaminated food; 3 Mild case: nausea, vomiting, light cyanosis,
(6 male, 19 ingestion no somnolence. Moderate case: somnolence and Xu (1987)
17 female) (10-350 ml) light consciousness. Severe case: Marked
cyanosis, coma, shock. 5 ECG examined:
2 bradycardia, 1 tachycardia, A-V block,
S-T change. Mild impairment of liver renal
functions. Treated with methylene blue,
19 recovered.
8 1 female ingestion 0 Lavage and treatment led to recovery from danger. Liu & Li (1987)
(52 years old) (30 ml conc. Black stool, tachycardia occurred on 3rd day.
formulation) Complete recovery.
9 187 27 occupational spray; 13 Cyanosis (63.6%), nausea (49.2%), vomiting Ding & Huang (1987)
(66 male, 16 ingestion (20-250 ml (44.9%), mydriasis (32.1%), somnolence (33.7%),
121 female) 25% chlordimeform coma (32.1%), irritation in urination (30.5%),
formulation) hypotension. 27 ECGs: 4 tachycardia, 6 bradycardia,
4 S-T & T wave change, 2 pre-mature beat, 2
conductive blockage. 158 cases received methylene
blue and 174 recovered within 1-5 days.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
10 1 male occupational spray 0 Sprayed incorrect dilution spray. Complained of Gu et al. (1987)
(28 years old) fatigue, somnolence, loss of appetite, nausea,
vomiting, but no cyanosis, or signs of cystitis,
pulse 68, BP 128/94 (normally 120/80), MAO 25.12 U
(normally 38.87 U). Total chlordimeform in urine
on admission, 6.4 mg/ml. Recovered quickly.
11 6 (?) ? Main clinical features: drowsy, cyanosis, Chan (1985)
loss of consciousness, mydriasis, cystitis,
hypotension, bradycardia, myocarditis, shock,
methaemaglobinaemia.
12 47 ingestion 4 Symptoms: drowsy, cyanosis, cystitis, Ke (1985)
(11 male, 20-1900 ml 2 hypotension (severe case), 8 hypertension,
36 female) 10 ECG: 1 tachycardia and T-wave change.
13 25 ingestion(?) 1 Cyanosis, cystitis, hypotension, arrhythmia, Wang & Dong (1985)
S-T and T changes, Q-T elongation. Treatment
with gastric lavage, methylene blue, vitamin C
14 682 340 occupational spray; 25 279 cyanosis, 147 cystitis, 197 somnolence, Liu & Zhang (1985)
(331 male, 342 ingestion 211 coma, 81 shock, 109 tachycardia,
351 female) 64 bradycardia, 54 hypertension, 22 hypotension.
59 ECG: 8 premature beat, 4 Q-T elongation,
16 S-T and T changes.
15 358 283 ingestion 37 Somnolence, cyanosis, loss of appetite, Ding & Ru (1985)
haemorrhagic cystitis, often myocardium damage,
A-V block, cardiac failure.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
16 49 3 occupational 4 13 cases were severe. Clinical features: cyanosis Liu & Ke (1985)
46 non-occupational and cystitis with haematuria in all cases, most
with severe somnolence and a few with coma. Two
severe cases had hypothermia. Hypertension was more
common than hypotension. 10 ECGs: only one case of
T-wave change and tachycardia. Treatment with
methylene blue and lavage.
17 1 ingestion (300 ml 25% 0 Coma and cyanosis. Sudden cardiac arrest during Yang (1984)
(female, form.) lavage, rescued with mechanical respiration.
25 years old) Recovered after 14 days.
18 24 ingestion (15-150 ml 2 16 cyanosis, 14 drowsiness, 8 haematuria, Wu et al. (1983)
(11 male, 25% form.) 6 methaemoglobin, 1 cardiac arrest, which
13 female) recovered after resuscitation.
19 101 35 occupational spray, 2 89 chlordimeform alone cases: 66% cyanosis, Xie (1983)
(49 male, 66 ingestion; 32 comas, 14 cystitis, 14 hypotension, 3 cardiac
52 female) chlordimeform +Ops) failure. 8 ECGs: 6 myocardium damage (changes in
Q-T, S-T, and T waves). Treatment with methylene
blue, vitamin. C. All recovered.
12 cases with mixed pesticides (OPs and Ocs).
20 1 ingestion 1 Loss of consciousness, cyanosis, mydriasis, Wu (1982)
(female, 85 years) (30 ml) arrhythmia. ECG: bradycardia, T-wave changes.
Died on day 6.
Table 16. (con't)
Study Number of patients Route of exposure Number Clinical features Reference
number (sex and/or age) of deaths
21 20 occupational spray 0 Farmers applied wrong dilution chlordimeform to Li et al. (1982)
(18 male, 2 female) cotton for one day. 7 drowsy, 10 loss of appetite,
4 cystitis. Symptomatic treatment. All recovered
in 2-4 days.
22 2 male ingestion (100 & 200ml) 0 Cyanosis, coma, respiratory-circulation failure, Zhang et al. (1976)
cystitis during 2nd day. Treatment with
methylene blue and atropine.
23 1 male 100 ml 0 Deep cyanosis, pulse 166. Xia & Gao (1977)
24 6 male occupational spray 0 Contamination of body surface and clothing. Su (1977)
Symptoms from day 1-4: cyanosis, haemorrhagic
cystitis, fatigue. Recovery after 18 days.
25 2 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Anonymous (1977)
no cyanosis, ECG normal. Symptomatically treated.
26 4 male occupational spray 0 Clothing contaminated. Haemorrhagic cystitis, Ming (1977)
cyanosis, somnolence, loss of appetite, haematuria,
RBC in urine for 20 days. Treatment: vitamin C,
antibiotics, coagulators.
51-year-old patient. Case studies of chlordimeform poisoning in China
due to occupational exposure are given in Table 17, together with a
brief account of the clinical features observed.
Table 17. Levels of urinary chlordimeform and its metabolites in hospitalized
workers (3 days following exposure) (Folland et al., 1978)
Worker Total aminesa Chlordimeform 4-Chloro-o-toluidine Conjugate
(mg/litre) (mg/litre) (mg/litre) (mg/litre)
1 11.0 1.10 3.75 6.25
2 15.2 2.16 4.16 8.67
3 2.6 0.04 1.25 1.17
a Measured following hydrolysis with 10N NaOH and 2 h at 80�C.
A brief account of the signs and symptoms of chlordimeform
poisoning and suggested interventions has been provided by Xue &
Loosli (1994).
8.2.2 Effects of long-term exposure
A report of an outbreak of haematuria in employees of a chemical
packaging plant in the USA over a 4-day period in 1975 was first
reported by Armstrong et al. (1975). Further details were described by
Folland et al. (1978). Nine of 22 workers who packaged chlordimeform
became severely ill with abdominal pain, dysuria, urgency to void, or
haematuria. In the previous year, four workers who had packaged the
chemical had similar symptoms. While six workers recovered within 7 to
18 days, three were hospitalized with symptoms which lasted from one
to two months. In these three workers, abnormalities noted were
microscopic haematuria and pyuria, proteinuria, low creatinine
clearance, elevated SGOT, prolonged BSP retention, elevated serum
amylase level, small bladder capacity, ureteral reflux and an intense
inflammatory reaction in three bladder biopsy specimens. The highest
concentrations of total amines were found in the urine of workers who
had become ill and were hospitalized. Low but measurable levels were
also found in workers who had not become ill. The major part of the
urinary amines was present as 4-chloro- o-toluidine or as conjugates.
Urinary total amines (following hydrolysis with 10 N sodium hydroxide
and 2 h at 80�C), as well as chlordimeform and 4-chloro- o-toluidine,
were measured in the hospitalized cases and are shown in Table 17.
The results of a monitoring programme on packaging workers in a
chlordimeform plant in the USA during 1976 have been described
(personal communication by J.W. Barnett, Ciba-Geigy Agricultural
Division, Greenborough, North Carolina, USA, to the California
Department of Food and Agricultural). The programme involved more than
100 workers and over 800 urine samples, monitoring for the presence of
red blood cells, for residues of chlordimeform metabolites, and for
clinical signs of toxicity in workers. Residues in urine samples were
reported to range from <0.05 to 50 mg/litre. There was no evidence of
microscopic haematuria found in the samples analysed nor of any
clinical signs of toxicity.
Four separate incidents resulting in 7 cases of frank haematuria
following industrial exposure were reported in the USA during the
period 1980-1984 (personal communication by J.W. Barnett, Ciba-Geigy
Agricultural Division, Greenborough, North Carolina, USA, to
Ciba-Geigy Ltd., Switzerland). Chemical cystitis, confirmed by
cystoscopy and biopsy, was diagnosed in one case while non-specific
bladder mucosal lesions were found in another. Six cases required
hospitalization, but all resolved after cessation of exposure.
In a study by Maddy et al. (1986), the results of a programme of
monitoring (1982-1985) the urine of more than 200 workers, who had
received training in the use of chlordimeform on cotton in California,
were described. Although urinalysis was unremarkable and no
significant cytological changes were found, a single case of bladder
cancer was detected in a pilot who had seven seasons of exposure to
chlordimeform.
By contrast, in the same period (1980-1984), no cases of
chlordimeform-induced haematuria occurred at manufacturing plants in
Switzerland and West Germany or formulation plants in Australia,
Columbia, Central America, Mexico and the USA. No cases of haematuria
reportedly resulted from application or use of chlordimeform in the
field (Anon., 1985b; personal communications by F.E. Pfister and P.
Duback (Ciba-Geigy Ltd., Agricultural Division, Switzerland) and by
N. Reckefus and K. Kossmann (Schering Aktiengesellschaft Agrochemical
Division, Berlin, Germany), 1985).
In a study by Lu et al. (1981), data on the effects of
chlordimeform exposure of factory workers in China was examined. In
this study, conducted in 1974, the air concentrations in the factory
were generally below 0.036 mg/m3, with shorter periods at higher
levels (0.108-0.33 mg/m3), during specific tasks. Skin contamination
on hands and forearms was 9.1 mg/h for chemical operators
and 964.2 mg/h for packers. The urinary excretion levels of
chlordimeform and 4-chloro- o-toluidine in controls were 0.015 and
0.042 mg/litre, respectively; in chemical operators they were
0.065 and 0.108 mg/litre, respectively; and in packers were 0.263 and
0.398 mg/litre, respectively. The health of the workers was examined
during the following 3 years (1974-1976). In 44-56 workers (equal
number of each sex) at an average age of 32 years and working period
of 2 years, the main finding were neurosis, sore throat and disorders
of the nervous system. There were no treatment-related effects on ECG,
liver function, clinical chemistry or urinalysis parameters.
In the same report (Lu et al., 1981), the effect of chlordimeform
exposure on rice field workers during 1974 was also examined. The
air concentration in the breathing space in all cases was below
0.02 mg/m3. Skin contamination was examined at the front of the
thorax, on the right forearm and on the right thigh. The applicators
applied chlordimeform for 4-5 h per day for 1-3 consecutive days,
wearing shirts and shorts with no other protection. Skin contamination
was from splash or from spray. The levels found from splash on thorax,
forearm and thigh were 0.0436, 0.0303 and 0.131 mg/100 cm2 per h,
respectively. The levels found from spray on thorax, forearm and thigh
were 0.235, 0.299 and 0.804 mg/100 cm2 per h, respectively. Medical
examination during 1974/1975 revealed complaints of light-headedness,
headache, fatigue, nausea, abdominal pain, skin itching and burning
sensation, and hypotension. There were no changes in ECG or blood
chemistry, and no reported cases of acute intoxication.
In a study by Li et al. (1985b), the health of 24 packers
(9 male, 15 female) in a chlordimeform manufacturing plant in Jiang-su
Province of China, was examined. The chlordimeform division of the
factory started manufacturing in 1975 and continued to do so at the
time of the study. The employees were working in the factory for
between 3 months and 4 years (average 1.5 years). Another 24 employees
from the kitchen and kindergarten served as controls. The air
concentration of chlordimeform (9 samples over 3 consecutive days) was
0.066 mg/m3 (range 0.017-0.121 mg/m3). Skin contamination of the
hands and forearms was 110 �g/100cm2 (S.D. 39 �g/100 cm2). Urinary
chlordimeform levels were 0.20 � 0.13 mg/litre, and urinary
4-chloro-o-toluidine levels were 0.48 � 0.29 mg/litre. Medical
examination revealed no difference between packers and controls with
regard to symptoms, laboratory examinations including liver enzymes
and urinalysis parameters, chest X-rays, ECG, or other parameters of
cardiac function. The only symptom associated with exposure was skin
rashes and itching in 21% of exposed individuals. There was no
difference in the micronucleus counting in cultured peripheral
lymphocytes between exposed and control groups, nor were there any
positive mutagenicity results from urine samples with or without
glucuronidase or sulfatase in the medium.
In a further study in a Chinese chlordimeform manufacturing
factory, the health of employees involved in chlordimeform production
was studied for the 5-year period, 1977 to 1981 (Anon., 1985a). The
urinary chlordimeform plus 4-chloro- o-toluidine levels of packers
was the highest at 0.39 mg/litre, which significantly correlated with
skin contamination but not with air concentration. The major medical
findings were complaints of lightheadedness, disorders in sleep,
memory impairment, fatigue, loss of appetite, skin rashes and itching,
and skin spot pigmentation. There were no features of cystitis. ECG
findings in 36 employees indicated premature beats, partial A-V block,
tachycardia and bradycardia. There was no evidence of chromosome
aberrations in metaphase chromosomes of cultured peripheral
lymphocytes.
In a study by Tao et al. (1985), the health of 61 employees
(25 chemical operators, 36 packers) of a pesticide factory in China
was examined. Chlordimeform was produced in the factory for 5 months
per year. Air levels ranged from 0.074 to 0.160 mg/m3. Skin
contamination of packers (2.99 mg/day) was higher than for
hemical operators (0.784 mg/day). The urinary excretion rate of
chlordimeform plus 4-chloro- o-toluidine in packers was also higher
(0.513 mg/litre) than for chemical operators (0.206 mg/litre) or
controls (0.055 mg/litre). Symptoms of exposure noted in packers
included loss of appetite, fatigue, somnolence and skin rashes.
Hepatomegaly was observed. There was no difference in blood pressure
or heart rate. Abnormalities in ECG were noted in 10/61 exposed
employees compared to 6/76 controls.
In a study by Wang et al. (1987), the health of 16 applicators
(8 males, 8 females) spraying chlordimeform in cotton fields in
Xin-yang Farm in the Jiang-su Province of China over a 3-day period
(July 1986) was examined. Air levels in the breathing zone were
0.031 mg/m3 and the skin contamination was 4.17 mg per shift. Urinary
levels of chlordimeform plus 4-chloro- o-toluidine ranged between 1
and 3 mg/litre over the exposure period. A close correlation was noted
between the level of chlordimeform on the skin and the levels of
chlordimeform plus 4-chloro- o-toluidine in the urine. Rapid
excretion of chlordimeform plus 4-chloro- o-toluidine was noted
following exposure. There was no change noted in heart rate, blood
pressure, monoamine oxidase activity or urinalysis between exposed
individuals and controls. Mild chlordimeform exposure, however,
appeared to be related to loss of appetite and drowsiness.
In a study by Zhang et al. (1986a), conducted at the same farm
over the same period, 13 applicators (7 male, 6 female, 20-41 years of
age) were examined during spraying chlordimeform on cotton over three
consecutive days. Protective measures included gauze mask, plastic
gloves and plastic apron, although it was noted that extensive
contamination occurred. Air levels in the breath zone on each of the
three days were 0.011, 0.014 and 0.011 mg/m3, respectively. Skin
contamination on each of the three days was estimated by the method of
Zhang et al. (1986b) to be 10.99, 4.32 and 4.45 mg/person per day,
respectively. Urinary chlordimeform plus 4-chloro- o-toluidine
levels were measured over the 3 days of exposure and for 7 days
after cessation of exposure. Urinary levels ranged from a peak of
2.408 mg/litre during exposure to 0.036 mg/litre after 7 days.
Excretion of chlordimeform occurred very rapidly with the highest
level being detected in the sample collected at the end of each shift.
There was a close correlation between skin contamination and urinary
excretion. Metabolism occurred very rapidly since 4-chloro- o-
toluidine usually accounted for 70-93 % of the total amount in the
urine. Serum monoamine oxidase activity varied from 26.18 U to
19.26 U. Clinical symptoms were somnolence, headache, dizziness and
fatigue. Heart rate and blood pressure dropped on the 2nd and 3rd
days. Analysis of ECG indicated elongation of P-R, Q-T intervals. One
person complained of urgency and pain in urination, gross haematuria,
and the urinary chlordimeform plus 4-chloro- o-toluidine level was
more than 6 mg/litre. Another four subjects were found to have
microscopic haematuria. Liver function tests were normal.
In a study by Xue et al. (personal communication by S.-Z. Xue,
M. Wang, C.-M. Chu and X.-W. Zhou entitled "Effects of chlordimeform
on cardiovascular function in humans with occupational exposure",
1993), the effect of chlordimeform on cardiovascular function was
studied in exposed farm workers and in manufacturing workers in China.
Four separate exposure groups were studied. The first (short-term)
exposure group consisted of 16 farmers engaged in spraying
chlordimeform (0.125% solution) in a cotton field. Exposure was for a
3- to 4-h period for 3 consecutive days. The second (long-term)
exposure group consisted of 21 chlordimeform packers in a factory who
had worked for 6 months on this task. The third exposure group
consisted of 19 factory plant operators who had minimal exposure to
chlordimeform. The fourth group consisted of 9 control (non-exposed)
factory workers. Exposure was measured in the breathing zone air
(personal sampler for the working shift, usually 6 h), by dermal
contact (pooled aliquot of 10 swabs from various body sites), and by
urine measurements. In each case, chlordimeform and its major
metabolite, 4-chloro- o-toluidine, were measured. The cardiovascular
system function was determined by measurement of blood pressure, heart
rate and electrocardiography (ECG). Exposure data indicated the packer
group had a higher inhalation exposure (0.107 mg/m3) than the
sprayers (0.031 mg/m3). Dermal exposure, on the other hand, was
higher in the sprayers group (4.251 mg/m2) than in the packers group
(2.713 mg/m2). Urinary levels collected at the end of the working
shift indicated the highest level in sprayers (1.950 mg/litre)
compared to packers (1.267 mg/litre) and operators (0.097 mg/litre).
In the farmer group, analysis of cardiovascular activity indicated a
significant decrease in heart rate, and an increase in P-wave
duration, Q-T interval and amplitude of T-wave compared to the control
group. In the factory workers, the packers had significantly lower
diastolic and systolic blood pressure, and an increase in T-wave
amplitude compared to the plant operators. The heart beat of packers
was also higher than controls, but not significantly. The
cardiovascular function parameters of the plant operators were
slightly but not significantly different to those of controls.
Examination of the cardiovascular function parameters of the
packers during a month of continuous exposure indicated a relationship
between length of exposure, total urinary chlordimeform, and cardiac
function parameters (see Table 18). Analysis of the exposure-effect
correlation indicated the drop in blood pressure was the most
sensitive parameter, with the change in amplitude of the T-wave the
next most sensitive parameter. The changes of P-R interval were the
least sensitive.
Table 18: Cardiovascular function and urinary chlordimeform in factory workers
(personal communication by S.-Z. Xue, M. Wang, C.-M. Chu and X.-W.
Zhou entitled "Effects of chlordimeform on cardiovascular function
in humans with occupational exposure", 1993)
Parameter measured Duration of continuous exposure (days)
0 1 7 15 30
Total urinary 0.000 0.311 0.627 0.642 0.773
chlordimeform (mg/litre)
Systolic BP (mmHg) 111/8.6a 105/7.7b 105/12c 102/10d 102/9.6c
Diastolic BP (mmHg) 71/7.1 69/9.3 63/10c 65/8.2d 64/9.8d
Heart rate (beat/min) 64.3/9.9 69.6/8.6d 67.2/6.5 70.0/9.4 71.4/12d
Q-T interval (msec) 398/18.2 404/23.1 412/16.7d 418/22.3d 412/23.6d
P-R interval (msec) 131/215 140/178d 140/212d 141/200a 143/317d
a Figures are mean/standard deviation
b P < 0.001
c P < 0.01
d P < 0.05
The authors attributed major importance to the alteration in
cardiovascular function in relation to chlordimeform intoxication, and
in most cases considered cardiac failure to be the cause of death.
Recognition of the effects on cardiac function may have been
overlooked previously, firstly, because of the diversity of mild
changes induced by chlordimeform and, secondly, because of the
tendency to concentrate on the effects of the aniline-containing
metabolites, such as methaema-globinaemia, haematuria, and
haemorrhagic cystitis. A no-observed-effect-level (NOEL) of
0.1 mg/litre of urinary chlordimeform plus 4-chloro- o-toluidine
excretion is proposed as the threshold for effects on cardiovascular
function following long-term, exposure while 0.3 mg/litre is proposed
as the threshold for effects on cardiovascular function following
short term exposure, even as short as one day. While the
cardiovascular function parameters are unlikely to be useful as
indicators of exposure, an understanding of the mechanism of action
should assist in designing appropriate treatment.
In a post-exposure surveillance programme, the chlordimeform-
exposed group showed an increased prevalence of malignancy-associated
surface markers on exfoliated urine cells, compared to geographical
controls, but no tumours were found (Kenyon et al., 1993).
8.2.3 Epidemiological studies
8.2.3.1 4-Chloro- o-toluidine
In a retrospective epidemiological study by Ott & Langner (1983)
the mortality experience of 342 employees assigned to three aromatic
amine-based dye production areas between 1914 and 1958 was examined in
relation to duration of employment (<1 to 5 years) and interval since
entry into these areas. 4-Chloro- o-toluidine represented one of a
number of chemicals to which the workers were potentially exposed.
4-Chloro- o-toluidine and two other aromatic amines ( o-toluidine
and 4-chloro-acetyl- o-toluidine) to which the workers were exposed
have been shown to be carcinogenic in animal studies. No deaths due to
bladder cancer were observed, and no statistically significant
increases in mortality by work area or duration of exposure within
work area were found.
In a retrospective study by Stasik (1988; 1991) of 116 workers
occupationally exposed in Germany to 4-chloro- o-toluidine during
manufacture prior to 1970, eight cases of bladder cancer, diagnosed
between 1967 and 1985, were identified. This represents an incidence
more than 70-fold higher than expected. Although occupational exposure
to two other aromatic amines, o-toluidine and 6-chloro- o-
toluidine, may have occurred, analysis of the production process
indicated that exposure to 4-chloro- o-toluidine in the plant was
considerably higher than exposure to these other two chemicals. The
workers were exposed to relatively high levels (before 1970) for a
median of 14 years. In two cases, however, the exposure period was
only 1.5 and 4.0 years. No quantitative measurements of exposure were
available. Two of the patients had suffered from haemorrhagic cystitis
as a consequence of massive acute exposure to 4-chloro -o-toluidine
at 4 and 14 years, respectively, before the tumour was diagnosed. The
latency periods for these eight cases ranged from 17 to 38 years. The
significantly increased incidence of bladder cancer in this study is
remarkable.
8.2.3.2 Chlordimeform
An epidemiological study has been conducted on the incidence of
cancer deaths of employees and their relatives on Xin-Yang Farm in
Jiang-su Province of China (Gu et al., 1991). In this area,
chlordimeform has been applied aerially in large amounts since 1974,
in a relatively indiscriminate manner, with contamination of land,
ponds, creeks, and gardens of adjacent houses. The study involved 7321
people (3911 male, 3410 female and 1265 retired agricultural workers)
over the period 1 January 1971 to 30 June 1987. During this period,
there were 706 registered deaths (510 males, 196 females), of which
198 were cancer deaths (160 males, 38 females). The standardized
mortality ratio (SMR) was calculated on the basis of the specific
mortality due to cancer in the adjacent Hai-men County. Many of the
SMRs were significantly exceeded on the Xin-Yang Farm, as shown in
Table 19. The incidences of bladder cancer adjusted to the national
level were 2.65 (males) and 1.47 (females) per 100 000. The SMRs were
260 (males) and 420 (females). During the period 1 July 1987 to
30 June 1990, there were three more cases of bladder cancer (with one
death) among the cohort members. The authors concluded there is
evidence for an association between bladder cancer and exposure to
chlordimeform, but that further data is needed to strengthen this
association. It is noted that there was a high incidence of other
tumour types in this study which makes the association between bladder
cancer and exposure to chlordimeform more difficult to establish.
Table 19. Standardized mortality ratio (SMR) for workers on the
Xin-Yang farm (Gu et al., 1991)
Cause of death Adjusted mortality Standardized mortality ratio
(per 100 000) (95% C.I.)
Male Female Male Female
All deaths 785.1 610.0 134 (124-145) 139 (128-151)
All cancers 214.0 130 113 (107-120) 128 (117-139)
Oesophageal cancer 35.6 32.5 228 (208-249) 388 (352-428)
Stomach cancer 61.4 24.5 175 (161-190) 120 (110-130)
Liver cancer 31.6 6.9 40 (37-44) 27 (24-29)
Colon cancer 8.8 6.5 133 (123-145) 79 (72-86)
Lung cancer 34.2 16.9 135 (124-146) 147 (135-169)
Leukemia 3.9 5.6 144 (133-157) 260 (235-285)
Bladder cancer 4.1 3.0 197 (180-214) 750 (671-839)
Breast cancer - 16.0 - 380 (345-419)
Cervical cancer - 30.7 - 216 (198-234)
Further epidemiological data on the association between cancer
incidence and exposure to chlordimeform has been provided in papers by
Xue et al. (1990; 1991). A summary of the findings of epidemio-logical
studies between 1984 and 1988 is given in Table 20. Data from three
counties and one farm are shown. The counties are located close to one
another, with comparable environmental and socio-economic situations.
The agricultural products are mainly rice and cotton. County A acted
as a control, with little or no use of chlordimeform; County B was
the largest user of chlordimeform; and County C started using
chlordimeform at the earliest time. The results from the Xin-Yang farm
are included for comparison. A comparison between the mortality rate
in recent years (1984-1988) and the mortality rate in the years prior
to the use of chlordimeform in these counties and Xin-Yang farm is
shown in Table 21. There were excesses in the incidence of all deaths,
deaths from cancer, and urinary bladder cancer for both sexes,
although the data may not yet have reached the level of statistical
significance.
Table 21. Comparison of adjusted mortalities of urinary bladder
cancer between 1984-1988 (county) and 1973-1975
(prefecture) (Xue et al., 1990, 1991)
Item County A County B County C Xin-Yang Farm /
/ Prefecture / Prefecture Prefecturea
Male 1.52 / 1.10 0.77 / 0.77 1.12 2.65 / 1.02
SRR 1.38 1.04 1.10 2.65b
Female 0.41 / 0.35 0.46 / 0.17 0.55 1.47 / 0.35
SRR 1.17 2.71b 1.57b 4.20b
a The duration of observation was 1971-1987 (June 30)
b p < 0.05
In a retrospective study by Popp & Norpoth (1991) and Popp et al.
(1992), the exposure and incidence of bladder cancer in a German
chemical plant was examined. Chlordimeform was manufactured from
4-chloro- o-toluidine and production commenced in December 1965.
Production was not continuous, but rather was in response to orders,
so workers were subject to different periods of exposure (generally
8-12 weeks per year). Between 1965 and 1976, the exact levels of
exposure were not available because measurement of the concentration
in the air or monitoring of urine excretion was not carried out at
that time. In 1976, production was ceased in order to improve working
conditions and minimize human exposure. Production recommenced in 1980
with improved containment and monitoring of urinary excretion of
Table 20. Data on Epidemiological Studies with Chlordimeform during 1984-1988 (Xue et al., 1990, 1991)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
Year started using chlordimeform 1979 1977 1973 1973
Population (annual average) 993 549 1 076 456 736 037 8732
Average amount of chlordimeform 1.1 65.0b 29.8 89.2
used (g/Mu/year)a
All causes of mortality
Male 584.5 675.7 761.0 785.1
RR 1.2 (1.1-1.3) 1.3 (1.2-1.4) 1.3 (1.2-1.5)
Female 438.1 891.7 668.5 625.9
RR 2.0 (1.4-2.3) 1.5 (1.1-1.7) 1.4 (1.3-1.5)
Cardiovascular mortalityc
Male 143.2 167.7 221.6 -
RR 1.2 (1.1-1.3) 1.6 (1.4-1.7)
Female 138.2 280.2 234.8 -
RR 2.0 (1.9-2.2) 1.7 (1.6-1.8)
Respiratory mortalityc
Male 99.6 100.4 127.2
RR 1.0 (0.9-1.1) 1.3 (1.2-1.4)
Female 82.1 145.1 124.0
RR 1.8 (1.7-1.9) 1.5 (1.4-1.6)
Table 20. (con't)
Items County A County B County C Xin-Yang Farm
(control) (largest amount) (earliest in using)
All cancer mortalityc
Male 188.5 246.6 232.2 214.9
RR 1.3 (1.2-1.4) 1.2 (1.1-1.3) 1.1 (1.1-1.2)
Female 101.7 227.1 145.5 130.0
RR 2.3 (2.0-2.5) 1.4 (1.3-1.6) 1.3 (1.2-1.4)
Bladder cancer mortalityc
Male 2.08 (95)d 0.90 (26) 2.10 (32) 4.10 (4)
RR 0.4 (0.39-0.47) 1.0 (0.9-1.2) 2.0 (1.8-2.2)
Female 0.40 (15) 0.20 (9) 0.90 (14) 3.00 (2)
RR 0.5 (0.46-0.55) 2.3 (2.1-2.5) 7.5 (6.7-8.4)
a The Mu is a measure of area equivalent to 1/15th acre
b Considered over the last 5 years
c All mortality figures were age-adjusted
d Figure in parentheses is the actual number of cases of bladder cancer
workers. Production finally ceased in 1986. The company identified 170
individuals who had come into contact with chlordimeform but many had
minimal exposure. The number of workers involved in the production of
chlordimeform was 49, and these comprised the study group. The period
under investigation was from the year of employment to the end of
1990. The expected incidence of bladder cancer (age- and sex-specific)
was extracted from the cancer registers of Saarland (1988), the former
German Democratic Republic (GDR) (1978-1982) and Denmark (1978-1982).
The standard incidence rate (SIR) was the ratio of the number of cases
observed to the expected number (see Table 22).
Table 22. Standard incidence rates (SIRs) of bladder carcinoma in a
group of 49 workers engaged in chlordimeform synthesis
(Popp et al., 1992)
Observed cases Expected number SIR 95% CI p value
7 0.078 (GDR) 89.7 35.6 - 168.6 0.000002
7 0.200 (Denmark) 35.0 13.9 - 65.7 0.00001
7 0.130 (Saarland) 53.8 21.3 - 101.1 0.000005
The average age for workers starting work was 30 (range 18-51), and
the exposure ranged from 3 to 956 days. By the end of 1990, an average
of 18 (10-25) years had passed since the start of exposure. Bladder
cancer was detected in 7 of the 49 subjects by the end of 1990. Of the
seven cases, six were diagnosed as transitional cell carcinoma and one
as papillary carcinoma. In five cases, the exposure period could be
determined, with an average of 575 days (range 291-766). The latency
period was an average of 19 years (range 15-23), with an average age
at diagnosis of 54 years (range 42-62). This study provides strong
evidence of an association between exposure to 4-chloro- o-toluidine
and human bladder cancer. All of the cases involved workers who were
exposed to 4-chloro- o-toluidine while synthesizing chlordimeform
before 1976. None of those workers who were handling the final
product, chlordimeform, had developed bladder cancer by the end of
1990.
In a historical cohort study (personal communication by P. Boyle
& G.J. Macfarlane to the IPCS, 1997), the bladder cancer incidence of
847 men involved in the manufacture of chlordimeform in Australia,
Switzerland, the United Kingdom and the USA was compared with that
expected on the basis of population-based cancer registry rates.
Subjects eligible to be included in the cohort were those who had been
employed in the production or formulation of chlordimeform or who had
otherwise been an integral part of a chlordimeform unit in a plant
where it had been produced or formulated for at least 6 months. The
results presented in Table 23 show an incidence rate of bladder cancer
which was significantly higher than expected. Overall, ten cases were
observed while 2.6 were expected. When the cohort was divided
according to whether members had been exposed to chlordimeform and
4-chloro- o-toluidine, or to chlordimeform alone, it was found that a
significant excess of risk of bladder cancer also occurred in those
workers thought not to have been exposed to 4-chloro- o-toluidine. In
this group of 592 men, 5 cases of bladder cancer were observed, while
1.4 cases were expected (SIR = 3.5, 95% CI (1.1, 8.3)). The authors
concluded that despite the lack of information on potentially
confounding factors in this study, the data indicated an association
between excess risk of bladder cancer in this cohort and one or more
aspects of the manufacture of chlordimeform.
Table 23. Bladder cancer risk in a cohort of men exposed to
chlordimeform (Boyle & Macfarlane, 1997)
Plant location Cohort numbers Bladder cancer cases
Observed Expected SIRa
Switzerland 273 4 0.72 5.6
USA (A)b 182 1 0.32 3.1
United Kingdom 174 3 1.06 2.8
USA (B)b 163 1 0.26 3.8
Australia 55 1 0.27 3.7
All plants 847 10 2.63 3.8
95% CIc
(1.8, 7.1)
a Standardized Incidence Ratio
b Different production sites
c Confidence Interval
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
9.1 Laboratory experiments
9.1.1 Microorganisms
There are no data on the effects of chlordimeform on
microorganisms.
9.1.2 Aquatic organisms
9.1.2.1 Plants
There are no data on the effects of chlordimeform on aquatic
plants.
9.1.2.2 Invertebrates
There are no laboratory data on the effects of chlordimeform on
aquatic invertebrates.
9.1.2.3 Vertebrates
The toxicity of chlordimeform to some species of fish has been
determined (FAO/WHO, 1972; Mayer & Ellersieck, 1986), and is shown in
Table 24.
9.1.3 Terrestrial organisms
9.1.3.1 Plants
There are no data available for the effects of chlordimeform on
plants.
9.1.3.2 Invertebrates
Dittrich (1966, 1967) first reported studies on the efficacy of
chlordimeform as an acaricide with both ovicidal activity against
insect eggs and adulticidal activity. It killed adult spider mites
when applied as a vapour and as a spray, and penetrated plant tissues
where it was released in ovicidal quantities. Since then, its efficacy
as an insecticide has been studied in a wide range of species.
Chlordimeform not only has a direct lethal action, particularly
against eggs and early instar larvae of insects and acarines, but also
has important sublethal effects, including sterilization of eggs,
induction of hyperactivity, detachment of feeding ticks from hosts,
Table 24. Toxicity of chlordimeform to fish
Species Duration LC50 Reference
(h) (mg/litre)
Bluegill sunfish 24 1.0 FAO/WHO (1972)
48 1.0
96 1.0
Trout 24 11.7 (8.73-15.8) FAO/WHO (1972)
48 10.6 (7.80-14.50)
96 7.14 (4.70-10.80)
Cat fish 24 11.9 (8.98-15.9) FAO/WHO (1972)
48 8.72 (6.26-21.1)
96 4.54 (3.08-6.68)
Rainbow trout 24 29 Mayer & Ellersiek
96 13.2 (1986)
Channel catfish 24 20.7 Mayer & Ellersiek
96 20.2 (1986)
Carp 24 65a FAO/WHO (1972)
48 60a
96 50a
a Values are for TLm
colony dispersal behaviour in ticks and mites, anti-feeding effects
and disruption of mating and oviposition in Lepidoptera (Hollingworth,
1976).
Knowles & Shrivastava (1973) investigated its toxicity in house
flies. The LD50 was 25 �g/fly, a dose which was not at a practical
level for house-fly control, possibly due to the high rate of
metabolism in this species. Pimley (1986) investigated the toxicity of
chlordimeform to tsetse fly (Glossina morsitans). The median lethal
dose was approximately 0.4 �g/fly for unfed tsetse, and 100% mortality
was achieved with 2 �g/fly. Sublethal doses also caused a significant
depression of feeding activity.
The specificity of chlordimeform with regard to both eggs and
larval stages was examined by Streibert & Dittrich (1977). Eggs of the
three noctuid cotton pest moths, Heliothis armigera, Heliothis
virescens, and Spodoptera littoralis, when exposed to a saturated
atmosphere of 4 mg/m3, have very similar sensitivity to vaporized
chlordimeform. Agrotis ipsilon, also a noctuid, on the other
hand, is definitely less sensitive, and the coccinillid Epilachna
varivestis was the most tolerant. The larval stages of all of these
species were considerably less sensitive to chlordimeform vapour than
the egg stage, but chlordimeform does seem to cause a decrease in the
number of larvae in the field, possibly due to a repellent effect or a
behavioural change rather than a direct toxic effect. These results
with Spodoptera littoralis on the relative sensitivity of eggs and
larvae were confirmed in the studies of Salvisberg et al. (1980).
Davenport & Wright (1985) have also demonstrated the differential
susceptibility of adult and larvae of the noctuid moths, Spodoptera
littoralis and Heliothis virescens, and also highlighted the
significantly higher toxicity of the hydrochloride salt, compared to
the base, to the adults of both species.
Sparks et al. (1993) studied the effects of several insecticides
on ovicidal activity and alteration of octopamine titres in eggs of
the tobacco budworm (Heliothis virescens). Chlordimeform was highly
toxic to eggs of H. virescens. The authors reported that the
alteration in the biogenic amine octopamine titres by chlordimeform
might lead to a disruption in the ability of larvae to hatch from the
egg.
Crecelius & Knowles (1976) studied the sensitivity of the larvae
of the cabbage looper, Trichoplusia ni, to the toxic effects of
chlordimeform. Third instar larvae were more sensitive to the toxic
effects of chlordimeform than the fifth instar larvae, possible due to
slower penetration and slower metabolism of chlordimeform in the
latter instar larvae.
Bailey & Cathey (1985) demonstrated the effectiveness of
chlordimeform in reducing the percentage egg hatch of Lygus
lineolaris (Palisot de Beauvois) on pole bean ( Phaseolus
vulgaris L.) pods and cotton ( Gossypium hirsutum L.). A solution
of 0.09% chlordimeform, while not significantly reducing nymph
emergence from eggs deposited on bean pole pods, did significantly
reduce nymph emergence from eggs deposited on cotton plants.
Salvisberg et al. (1980) also demonstrated that Spodoptera
littoralis moths, when treated at doses as low as 10% of the LD50,
showed symptoms of hyperexcitation, which resulted in abnormal
patterns of egg-laying, a reduced number of eggs and lower fertility.
Further studies by Davenport & Wright (1987) on Spodoptera
littoralis have shown that chlordimeform hydrochloride significantly
reduces food consumption in fifth-instar larvae when incorporated into
the diet at a level of 0.1-10 mg/g or when topically applied. No
mortality occurred during feeding, but mortality was increased during
subsequent pupation and during emergence of the adult from the pupae.
In adult moths, egg laying was significantly decreased when
chlordimeform hydrochloride was applied topically (1 or 10 �g/moth).
Further evidence that behavioural changes may be more important
in reducing both the larval and insect populations following
chlordimeform treatment has been provided by Shimizu & Fukami (1983)
in studies of the larvae of the cabbage armyworm, Mamestra
brassicae, which showed a prolonged period of wandering behaviour in
the presence of chlordimeform. This may have caused a failure to find
or prepare a suitable site for pupation.
The behaviour-modifying effects of chlordimeform have also been
studied by Blackwell (1988a,b; 1889) in the larvae of the large
cabbage white butterfly, Pieris brassicae L. When placed on
chlordimeform-dipped leaves, the larvae become excited, in contrast to
their normal communal feeding behaviour. Locomotion was increased and
feeding was significantly reduced as a result of disaggregation
(Blackwell, 1988a). When applied directly to the larvae, chlordimeform
caused excitation and inhibition of feeding (Blackwell, 1989). Direct
application also caused developmental delays and mortality was
increased at later developmental stages (Blackwell, 1988b).
O'Brian et al. (1985) have studied the effect of insecticides on
beneficial insects, and in particular, the effect of chlordimeform on
the ecoparasitoid, Bracon mellitor, an important parasitoid of the
boll weevil (Anthonomus grandis grandis). Chlordimeform was found to
be more toxic to Bracon mellitor than to the boll weevil, and also
reduced the number of egg deposited.
The toxicity of chlordimeform hydrochloride to bees has been
examined after both ingestion and contact. Ingestion of a 0.3%
solution killed 18%, while ingestion of 0.15% killed approximately 7%.
Contact with the same solutions did not increase the mortality rate
(FAO/WHO, 1972). In a study by Johansen (1972), bees were exposed to
field-weathered residues of a range of insecticides, including
chlordimeform, on alfalfa foliage. Over a 24-h period, zero mortality
was obtained with alfalfa leafcutter bees, alkali bees and honey bees
exposed to 3-h-old residues.
9.1.3.3 Vertebrates
Fleming et al. (1985) examined the toxic and behavioural effects
of chlordimeform on the game bird, the bobwhite quail (Colinus
virginianus). When added to the diet of newborn chicks over a 7-day
period, the lethal concentration to chicks was 2835 mg/kg diet
(2169-3705 mg/kg diet). When chicks were fed a diet containing
chlordime-form at a concentration of 1000 mg/kg diet for 7 days, they
ate less, weighed less, travelled further from a fright stimulus in an
avoidance test, and had a high locomotor activity in an open-field
test than at lower dose levels. Chicks fed 100 or 1000 mg
chlordimeform/kg diet scored more highly than controls in a visual
cliff performance test. After a further 8 days on control diet, the
chicks fed 1000 mg/kg diet still scored higher than controls on the
avoidance test, but the open-field and cliff performance scores were
similar to those of controls.
In studies conducted on bobwhite quails and ducks, groups of
animals (10 per treatment group, 30 per control group) were fed
chlordimeform technical or chlordimeform 48% EC formulation in their
diets for 5 consecutive days. The dose levels were 0, 10, 31.6, 100,
316 or 1000 mg/kg diet. Both quails and ducks were tolerant of the
presence of chlordimeform in the diet. With the technical material,
one quail in each of the groups fed 100 and 316 mg/kg diet died,
while, with the formulation, one quail in each of the groups fed 316
and 1000 mg/kg diet died. All ducks survived treatment, even at the
highest dose level (FAO/WHO, 1972).
Hill et al. (1975) exposed three bird species, Japanese quail
(Coturnix japonica), ring-necked pheasant (Phasianus colchicus)
and mallard (Anas platyrhynchos), to chlordimeform. LC50 values for
Japanese quail and ring-necked pheasant were determined to be 1749 and
2608 mg/kg diet, respectively. The LC50 for mallard was determined to
be >5000 mg/kg diet; only 20% mortality was reported at the highest
exposure group, 5000 mg/kg diet. Hill & Camardese (1986) reported an
LC50 of 5079 mg/kg diet for Japanese quail exposed to chlordimeform.
9.2 Field Observations
9.2.1 Microorganisms
There are no field data on the effects of chlordimeform on
microorganisms.
9.2.2 Aquatic organisms
There are no field data on the effects of chlordimeform on
aquatic organisms.
9.2.3 Terrestrial organisms
9.2.3.1 Plants
The possibility that some insecticides might enhance the growth
of cotton plants has been suggested for some time. However, in the
case of chlordimeform, debate has continued as to whether this effect
is due to early season insect suppression (Bailey & Cathey, 1985) or
to a physiological effect (Phillips et al., 1977). Cathey & Bailey
(1987) have conducted controlled studies to examine the effects of
multiple applications of chlordimeform on the growth and development
of cotton ( Gossypium hirsutum L.) in both greenhouse and field
studies. Plants were sprayed six times with chlordimeform either alone
or in combination with fenvalerate at 5- to 7-day intervals, beginning
at the six-leaf stage of plant development. In the absence of early
season insects and when insect populations were maintained at a
relatively low level, no increases in lint yield occurred on the
chlordimeform-treated plants. However, yield increases did occur and
insect populations became lower in these treated plots when early
season insect populations in the test area were relatively high. None
of the treatments influenced the boll components, boll size, seed
index and lint percentage, or the first fibre properties, length,
strength and micronaire.
Field studies by Youngman et al. (1990) to determine the effects
of several insecticides on growth, fruiting patterns and yield of the
cotton plant, Gossypium hirsutum L., supported the conclusion that
chlordimeform does not significantly increase any plant growth
parameter when compared with the control.
9.2.3.2 Invertebrates
In a small field study conducted by Bull & House (1978), tests
were conducted in 0.05-ha plots of cotton to compare lower and more
frequent applications of chlordimeform with commercial mixtures of
insecticides against natural populations of Heliothis species. The
results indicated that the protection afforded was as good as with
commercial mixtures, probably as a result of careful observation of
the cotton to pinpoint the onset of significant egg production.
In a another small field study by Wilson (1981), the potential of
chlordimeform to control Heliothis species in cotton was tested
separately or in combination with amitraz and the microbial
insecticide, Bacillus thuringiensis. Chlordimeform was the most
efficient of the three materials and controlled Heliothis species
reasonably efficiently, but no control of the rough bollworm,
Earias hueglei was obtained. There was also good control of the
cotton looper, Anomis flava, and some indication of suppression of
mites and aphids was obtained.
The behaviour-modifying effects of chlordimeform have been
demonstrated in field studies by Uk & Dittrich (1986) on the adult
whitefly, Bemisia tabaci (Genn.), which attacks cotton in the Sudan.
At dose levels of 500-2500 g chlordimeform/ha together with 960 g
endosulfan/ha, there was evidence of irritation and mass emigration of
adults from treated cotton foliage without detectable direct
mortality.
9.2.3.3 Vertebrates
There are no field data on the effects of chlordimeform on
vertebrates.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.1.1 Exposure
Production and use of chlordimeform has now ceased worldwide
and no further human exposure should occur. During the years of
chlordimeform production and use, dietary and incidental exposure to
chlordimeform occurred. Occupational exposure to chlordimeform and
4-chloro- o-toluidine (used as a starting material for synthesis)
occurred during manufacture and formulation, as well as during
application. The major route of exposure was through dermal
contamination. Application of chlordimeform occurred extensively by
aerial spraying, but it was also applied by ground-rigs as well as by
backpack spray equipment. Thus, agricultural workers were exposed
during mixing, loading, washing, and flagging operations. General
population exposure occurred through the consumption of food
containing residues of chlordimeform, and to a lesser extent through
by-stander exposure. In some cases, there was intentional ingestion of
the formulation.
Monitoring of urinary 4-chloro- o-toluidine has been found to be
a useful indicator of exposure.
10.1.2 Toxicity
In both experimental animals and humans, there is extensive
metabolism of chlordimeform, followed by rapid excretion via the
urine. A major urinary metabolite is 4-chloro- o-toluidine. In
experimental animals, symptoms of acute toxicity included neurotoxic
as well as cardiovascular effects. There was no evidence of
teratogenicity or reproductive effects. Following chronic
administration, there was a dose-related increase in
haemangioendotheliomas in mice. There was no treatment-related
increase in tumour incidence in rats. Most of the mutagenicity studies
with chlordimeform itself were negative, but there were sporadic
reports of genotoxicity with 4-chloro- o-toluidine and to a more
limited extent with N-formyl-4-chloro- o-toluidine.
In humans, chlordimeform has been shown to have both acute and
chronic effects. Acute poisoning caused fatigue, nausea and loss of
appetite, and, in more severe cases, somnolence, cyanosis, urgency in
urination, cystitis, cardiovascular effects (tachycardia, bradycardia,
ECG changes), coma and shock. The significance of the cardiovascular
effects in chlordimeform-induced mortality has only recently been
recognized. While there have been fatalities as a result of acute
chlordimeform exposure, in the majority of cases complete recovery
occurs. Symptoms of chronic exposure include those of acute exposure
as well as abdominal pain, skin itching and rashes, and gross or
microscopic haematuria.
With regard to carcinogenicity, the International Agency for
Research on Cancer (IARC) has concluded that there is limited
evidence in humans and sufficient evidence in experimental animals
for the carcinogenicity of 4-chloro- o-toluidine. The available
epidemiological data indicate an association between excess risk of
bladder cancer and exposures entailed in the manufacture of
chlordimeform. There is currently preliminary epidemiological evidence
of an association between chlordimeform use and excess risk of bladder
cancer.
10.1.3 Risk evaluation
With the withdrawal of the use of chlordimeform in agriculture
and a cessation of production worldwide, there is no longer any risk
associated with acute exposure except during the disposal of existing
stocks. The risk associated with chronic exposure, however,
particularly the risk of bladder cancer, will continue to be of
concern for many years. Human bladder cancer has a long latency
period, and establishing whether or not there is a link between
chlordimeform exposure and bladder cancer will require continued
health screening of significantly exposed individuals both from
manufacturing plants and from those rural communities where
chlordimeform was extensively used.
10.2 Evaluation of effects on the environment
Since chlordimeform is no longer used, no quantitative risk
assessment for the environment has been performed. There are not
expected to be any long-term detrimental effects on the environment as
a result of past use of chlordimeform.
11. CONCLUSIONS AND RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
AND THE ENVIRONMENT
11.1 Conclusions
Chlordimeform has significant potential to cause both immediate
and long-term toxicity in exposed individuals. Current information
supports an association between an increased incidence of human
bladder cancer and exposure to 4-chloro- o-toluidine, and, to a
lesser extent, chlordimeform.
Chlordimeform does not persist in the environment and therefore
there are not expected to be any long-term detrimental effects on the
environment as a result of past use.
11.2 Recommendations for protection of human health and the
environment
Future commercial production or use of chlordimeform is not
recommended. Existing stocks should be disposed of safely.
Those with occupational exposure to chlordimeform should
participate in a health screening programme that includes urinary
cytology and the detection of haematuria.
12. FURTHER RESEARCH
The following studies are needed:
1. epidemiological investigations on exposed populations.
2. studies on the dose-response relationship between exposure to
4-chloro- o-toluidine or chlordimeform and the induction of
urinary bladder cancer in humans.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Chlordimeform was considered by the International Agency for
Research on Cancer (IARC) in 1983. IARC noted that no published study
on the carcinogenicity of chlordimeform was available. However, it
considered data on the carcinogenicity of 4-chloro- o-toluidine and
concluded that the results of experiments on mice provided sufficient
evidence that 4-chloro- o-toluidine, a metabolite of chlordimeform,
is carcinogenic to experimental animals. No relevant data on humans
were available. IARC concluded the available data were inadequate to
evaluate the carcinogenicity of chlordimeform to humans (IARC, 1983).
The carcinogenicity of 4-chloro- o-toluidine, the breakdown
product and major metabolite of chlordimeform, was evaluated by IARC
in 1990 (IARC, 1990). On the basis of the available published data, it
was concluded that there is limited evidence in humans and
sufficient evidence in experimental animals for the carcinogenicity
of 4-chloro- o-toluidine. 4-chloro- o-toluidine and its strong acid
salts were classified as probably carcinogenic to humans (Group 2A).
Chlordimeform was considered at the 1971, 1975, 1978, 1979, 1980,
1985 and 1987 FAO/WHO Joint Meeting on Pesticide Residues (JMPR). In
1971, a temporary acceptable daily intake (ADI) for chlordimeform of
0-0.01 mg/kg body weight was established, and temporary maximum
residue levels (MRLs) were set for a number of crops and for the meat
and milk of cattle (FAO/WHO, 1972). In 1975, the temporary ADI was
maintained and some new temporary MRLs were established (FAO/WHO,
1976). In 1978, the temporary ADI was reduced to 0-0.0001 mg/kg body
weight, the temporary MRLs for all crops except cotton and cottonseed
were withdrawn, and the MRLs for meat and milk of cattle were set at
the level of detection (FAO/WHO, 1979). In 1979, 1980 and 1985, the
temporary ADI of 0-0.0001 mg/kg body weight was extended (FAO/WHO,
1980, 1981, 1986). In 1987, the temporary ADI for chlordimeform was
withdrawn (FAO/WHO, 1988).
REFERENCES
Abo-Khatwa N & Hollingworth RM (1972a) Chlordimeform: The relation of
the mitochondrial uncoupling to toxicity in the German cockroach. Life
Science, 11: 1181-1190.
Abo-Khatwa N & Hollingworth RM (1972b) Chlordimeform: Uncoupling
activity against rat liver mitochondria. Pest Biochem Physiol,
3: 358-369.
Ahmad S & Knowles CO (1971), Metabolism of N'-(4-chloro-o-tolyl)-
N,N-dimethyl-formamidine (chlorphenamidine) and 4'-chloro-o-
formotoluidine by rat hepatic microsomal and soluble enzymes.
Camp Gen Pharmacol, 2:189-197.
Anonymous (1971 a) Gas chromatographic method for the determination of
C 8514 residues in animal tissues, fat, milk and butter. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Anonymous (1971b) Chlordimeform: Working paper prepared for the 1971
Joint FAO/WHO Meeting on Pesticide Residues. Basel, Switzerland,
Ciba-Geigy Ltd and Berlin, Germany, Schering AG (Unpublished
document).
Anonymous (1977) [Report of two cases of chemical cystitis.] New Med,
8: 82. (in Chinese with English abstract).
Anonymous (1980a) CGA-95119: High performance liquid chromatographic
determination of urinary residues containing the 4-chloro-o-toluidine
moiety - Procedure No. 177. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Anonymous (1980b) [Study on the fate of chlordimeform residue on green
cabbage.] Shanghai, Shanghai Institute of Pesticides, Basic Metabolism
Group (in Chinese with English abstract).
Anonymous (1981) [On the residue of chlordimeform: Special Task Group
report.] In: [Selected papers on research on pesticides.] In: Chinese
Institute of Scientific Information and Bureau of Science and
Technology ed. [Selected papers on studies of pesticides.] Beijing,
Ministry of Chemistry (in Chinese with English abstract).
Anonymous (1985a) [Five years follow-up observation on the health
effect of chlordimeform on the employees in manufacturing.] Lab Med,
2: 7-10 (in Chinese with English abstract).
Anonymous (1985b) Chlordimeform: Joint submission to the WHO expert
group of the 1985 FAO/WHO Joint Meeting on Pesticide Residues. Basel,
Switzerland, Ciba-Geigy Ltd and Berlin, Germany, Schering AG
(Unpublished report).
Arima T, Morooka H, Tanigawa T, Imai M, Tsunashima T, & Kita S (1976)
Methemoglobinemia induced by chlorphenamidine. Acta Med Okayama.
30: 57-60.
Armstrong J, Graves O, Lovejoy G, Swiggart R, & Kimbrough R (1975)
Insecticide induced acute haemorrhagic cystitis. Morbid Mortal Wkly
Rep (Centres for Disease Control), 24: 374.
Arni P & M�ller D (1976a) Salmonella/mammalian-microsome mutagenicity
test with C8513 (chlordimeform HC1), Basel, Switzerland. Ciba-Geigy
Ltd (Unpublished report).
Arni P & M�ller D (1976b) Salmonella/mammalian-microsome mutagenicity
test with CGA 72647 (4-chloro-o-toluidine HCI), Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1976c) Salmonella/mammalian-microsome mutagenicity
test with CGA 72651 (N-formyl-4-chloro-o-toluidine), Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983a) C8513: Intrasanguine host-mediated assay
with S. typhimurium, Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Arni P & M�ller D (1983b) CGA 72647: Intrasanguine host-mediated assay
with S. typhimurium, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983c) C8513: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983d) CGA 72647: Saccharomyces cerevisiae
D7/mammalian-microsome mutagenicity test in vitro. Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P & M�ller D (1983e) CGA 72651 pure: Chromosome studies in male
germinal epithelium, mouse spermatocytes. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Arni P, Hool G, & M�ller D (1983a) C8513: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Arni P, Hool G, & M�ller D (1983b) CGA 72651: Chromosome studies in
male germinal epithelium, mouse spermatogonia. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Aziz SA & Knowles CO (1973) Inhibition of monoamine oxidase by the
pesticide chlordimeform and related compounds. Nature (Lond),
242: 417-418.
Bailey JC & Cathey GW (1985) Effect of chlordimeform on tarnished
plant bug (Heteroptera: Miridae) nymph emergence. J Econ Entomol,
78: 1485-1487.
Bailey BA, Martin RJ, & Downer RGH (1982) Monoamine oxidase inhibition
and brain catecholamine levels in the rat following treatment with
chlordimeform. Pest Biochem Physiol, 17: 293-300.
Balu K (1989) Evaluation of field worker exposure to chlordimeform by
using urine monitoring. In: Biological monitoring for pesticide
exposure: Measurement, estimation of risk reduction. Washington, DC,
American Chemical Society, pp 47-55 (ACS Symposium Series 382).
Bauer PJ & Cothren JT (1990) Growth-promoting activity of
chlordimeform. Agron J, 82: 73-75.
Beeman RW & Matsumura F (1973) Chlordimeform: A pesticide acting upon
amine regulatory mechanisms. Nature (Lond), 242: 273-274.
Beilstein P & M�ller D (1983) C8513 pure: BALB/3T3 cell transformation
assay. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Beilstein P & M�ller D (1984a) C8513 technical: L-5178Y/TK+/- mouse
lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Beilstein P & M�ller D (1984b) CGA 72647 technical: L1578Y/TK+/-
mouse lymphoma mutagenicity test. Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Beilstein P & M�ller D (1984c) CGA 72647 technical: BALB/3T3 cell
transformation assay. Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Benedict JH, Walmsley MH, Segers JC, & Treacy MF (1986) Yield
enhancement and insect suppression with chlordimeform (Fundal) on
dryland cotton. J Econ Etomol, 79: 238-242.
Benezet HJ & Knowles CO (1976) N'-(4-chloro-o-tolyl)-N-
methylformamidine (demethylchlordimeform) metabolism in the rat.
J Agric Food Chem, 24: 152-154.
Benezet HJ & Knowles CO (1981) Degradation of chlordimeform by algae.
Chemosphere, 10: 909-917.
Benezet HJ, Chang K, & Knowles CO (1978) Formamidine pesticides -
metabolic aspects of neurotoxicity. In: Pesticide venom neurotoxic,
pp 189-206.
Bentley P, Staubli W, Bieri F, Muecke W, & Waechter F (1985) Induction
of hepatic drug-metabolising enzymes following treatment of rats and
mice with chlordimeform. Toxicol Lett, 28: 143-149.
Bentley P, Bieri F, Muecke W, Waechter F, & Staubli W (1986a) Species
differences in toxicity of p-chloro-o-toluidine to rats and mice:
Covalent binding to hepatic macromolecules and hepatic non-parenchymal
cell DNA and an investigation of effects upon the incorporation of
[3H]-thymidine into capillary endothelial cells. Chem-Biol Interact,
57: 27-40.
Bentley P, Waechter F, Bieri F, Staubli W, & Muecke W (1986b) Species
differences in the covalent binding of p-chloro-o-toluidine to DNA.
Arch Toxicol, 9(suppl): 163-166.
Blackmore RH (1969a) Chlorphenamidine (Schering 36268): 2-year
repeated feeding study in dogs (Project No. 728-127). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969b) Chlorphenamidine (Schering 36268): 24-month
repeated feeding study in rats (Project No. 728-126). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969c) Chlorphenamidine (Schering 36268): three
generation reproduction study (Project No. 728-118). Falls Church,
Virginia, Hazleton Laboratories Inc (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackmore RH (1969d) Chlorphenamidine (Schering 36268). Segment II:
Teratology Study -Rabbits (Project No. 728-157). Falls Church,
Virginia, Hazleton Laboratories Inc. (Unpublished report submitted to
the 1971 Joint FAO/WHO Meeting on Pesticide Residues).
Blackwell A (1988a) Survival and development of Pieris brassicae
larvae upon application of sublethal doses of chlordimeform. Entomol
Exp Appl, 48: 149-156.
Blackwell A (1988b) The disruption of aggregation behaviour in Pieris
brassicae larvae by chlordimeform. Entomol Exp Appl, 48: 141-147.
Blackwell A (1989) The effects of chlordimeform on the behaviour of
Pieris brassicae larvae. Pestic Sci, 26: 11-19.
Blass W (1972a) Galecron residues in Japanese tea. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. RVA 5/72).
Blass W (1972b) Galecron residues in Swiss grapes (berries and juice).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 11/72).
Blass W (1972c) Galecron residues in Swiss wine 1971. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA 16/72).
Bokisch AJ, Cox RT, & Walker RJ (1985) The actions of formamidine
pesticides on identified central neurons of Helix aspersa. Pestic:
Sci, 16: 495-503.
Boyes WK & Dyer RS (1984) Chlordimeform produces profound, selective,
and transient changes in visual evoked potentials of hooded rats. Exp
Neurol, 86: 434-447.
Boyes WK & Moser VC (1988) An alpha 2-adrenergic mode of action of
chlordimeform on rat visual function. Toxicol Appl Pharmacol,
92: 402-418.
Budris DM, Yim GK, Carlson GP, & Schnell RC (1983) Effect of acute and
repeated chlordimeform treatment on rat hepatic microsomal drug
metabolizing enzymes. Toxicol Lett, 18: 63-71.
Budris DM, Yim GK, & Schnell RC (1984) Effect of acute and repeated
chlordimeform treatment on rat hepatic microsomal drug metabolizing
enzymes. J Environ Pathol Toxicol Oncol, 5: 175-182.
Bull DL (1973) Metabolism of chlordimeform in cotton plants. Environ
Entomol, 2: 869-871.
Bull DL & House VS (1978) Effects of chlordimeform on insects
associated with cotton. Southwest Entomol, 3: 284-291.
Burkhard N (1971) Residues in milk, meat, liver and fat of a Swiss
cow. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
14/71).
Campanhola C & Plapp FW Jr (1989) Pyrethroid resistance in the tobacco
budworm (Lepidoptera: Noctuidae): Insecticide bioassays and field
monitoring. J Econ Entomol, 82: 22-28.
Candura SM, Marraccini P, Costa LG, Manzo L, Rossi A, Coccini T, &
Toninin M (1992) Calcium entry blockade as a mechanism for
chlordimeform induced inhibition of motor activity in the isolated
guinea-pig ileum. Pharmacol Toxicol, 71: 426-433.
Cathey GW & Bailey JC (1987) Evaluation of chlordimeform for cotton
yield enhancement. J Econ Entomol, 80: 670-674.
Chan Y-M (1985) [Observation and nursing of 6 cases of chlordimeform
poisoning.) Chin J Nurs, 20: 68-69 (in Chinese with English abstract).
Cheung MW, Kahrs RA, Nixon WB, Ross JA, & Tweedy BG (1989)
Determination of chlordimeform residues as 4-chloro-o-toluidine in
human urine using high-performance liquid chromatography. In:
Biological monitoring of pesticide exposure: Measuring and estimating
risk reduction. Washington, DC, American Chemical Society, pp 231-239
(ACS Symposium Series 382).
Cooper RL, Barrett MA, Goldman JM, Rehnberg GL, McElroy WK, & Stoker
TE (1994) Pregnancy alterations following xenobiotic-induced delays in
ovulation in the female rat. Fundam Appl Toxicol, 22: 474-480.
Costa LG & Murphy SD (1987) Interaction of the pesticide chlordimeform
with adrenergic receptors in mouse brain: an in vitro study. Arch
Toxicol, 59: 323-327.
Costa LG, Wu S, Olibet G, & Murphy SD (1988) Alpha2-adrenoceptors as
a target for formamidine pesticides: In vitro and in vivo studies in
mice. Toxicol Appl Pharmacol, 93: 319-328.
Costa LG, Wu S, Olibet G, & Murphy SD (1989) Formamidine pesticides
and alpha2-adrenoceptors: Studies with amitraz and chlordimeform in
rats and development of a radioreceptor binding assay. Neurotoxicol,
11: 405-411.
Costa LG, Gastel J, & Murphy SD (1991) The formamidine pesticides
chlordimeform and amitraz decrease hepatic glutathione in mice through
an interaction with alpha2-adrenoceptors. J Toxicol Environ Health,
33: 349-358.
Coye MJ, Lowe JA, & Maddy KJ (1986) Biological monitoring of
agricultural workers exposed to pesticides: II. Monitoring of intact
pesticides and their metabolites. J Occup Med, 28: 628-636.
Crecelius CS & Knowles CO (1976) Toxicity, penetration, and metabolism
of chlordimeform and its N-demethyl metabolite in cabbage looper
larvae. J Agric Food Chem, 24: 1018-1023.
Crowder LA & Whitson RS (1980) Fate of toxaphene, methyl parathion,
and chlordimeform combinations in the mouse. Bull Environ Contam
Toxicol, 24: 444-451.
Currie AN (1933) Chemical haematuria from handling 5-chloro-ortho-
toluidine. J Ind Hyg, 15: 205-213.
Davenport AP & Wright DJ (1985) Toxicity of chlordimeform and amitraz
to the Egyptian cotton leafworm (Spodoptera littoralis) and the
tobacco budworm (Heliothis virescens). Pestic Sci, 16: 81-87.
Davenport AP & Wright DJ (1987) Effect of the formamidine insecticide
chlordimeform on feeding and reproduction in the noctuid moth,
Spodoptera littoralis (Boisduval). Crop Prot, 6: 3-6.
Deng C-S, Zhang J-K, & Guo J-Y (1984) [Brief report of the symposium
on chlordimeform poisoning.] J Clin Med, 1: 51-53 (in Chinese with
English abstract).
Ding M-B & Ru X-Y (1985) [On diagnosis and treatment of chlordimeform
poisoning (analysis of 358 cases).] Jiangsu Med, 7: 30-31 (in Chinese
with English abstract).
Ding Y & Huang K-L (1987) [Analysis on clinical features of
chlordimeform poisoning.] Labor Med, 4: 8-10 (in Chinese with English
abstract).
Dittrich V (1966) N-(2-methyl-4-chlorophenyl)-N',N'-
dimethylformamidine (C-8514/Schering 36268) evaluated as an
acaricide. J Econ Entomol, 59: 889-893.
Dittrich V (1967) Systemic action of N-(2-methyl-4-chlorophenyl)-N',
N'-dimethylformamidine (C-8514/Schering 36268). J Econ Etomol,
60: 558-569.
Dittrich V & Loncarevic A (1971) New insecticides for Asiatic rice
borer control in paddy rice. J Econ Entomol, 64: 1225-1229.
Dyer RS & Boyes WK (1983) Chlordimeform differentially affects pattern
reversal-evoked and flash-evoked potentials in rats. Toxicologist,
3: 13.
Ehrhardt DA & Knowles CO (1970) Metabolism and translocation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine (chlorphenamidine) and
its hydrochloride salt in grapefruit seedlings. J Econ Entomol,
63: 1306-1314.
Ercegovich CD, Witkonton S. & Asquith DJ (1972) Disappearance of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine from six major fruit
crops. J Agric Food Chem, 20: 565-568.
Evans PD & Gee JD (1980) Action of formamidine pesticides on
octopamine receptors. Nature (Lond), 287: 60-62
Fan DF & Ge SD (1982) Gas-liquid chromatographic determination of
chlordimeform and its metabolites in cargo rice and husk. J Assoc Off
Anal Chem, 65: 1517-1520.
FAO/WHO (1972) Chlordimeform. In: 1971 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 3-45 (WHO
Pesticide Series, No. 1).
FAO/WHO (1976) Chlordimeform. In: 1975 Evaluation of some pesticide
residues in food. Geneva, World Health Organization, pp 85-104 (WHO
Pesticide Residues Series, No. 5).
FAO/WHO (1979) Chlordimeform. In: Pesticides residues in food - 1978:
Evaluations, Rome, Food and Agriculture Organization of the United
Nations, PP 51-78 (FAO Plant Production and Protection Paper 15 Sup.).
FAO/WHO (1980) Chlordimeform. In: Pesticide residues in food - 1979:
Evaluations. Rome, Food and Agriculture Organization of the United
Nations, pp 129-135 (FAO Plant Production and Protection Paper 20
Sup.).
FAO/WHO (1981) Chlordimeform. In: Pesticide residues in food - 1980:
Evaluations. Rome. Food and Agriculture Organization of the United
Nations, pp 89-96 (FAO Plant Production and Protection Paper 26 Sup.).
FAO/WHO (1986) Chlordimeform. In: Pesticide residues in food - 1985:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 17-29 (FAO Plant Production and
Protection Paper 72/2).
FAO/WHO (1988) Chlordimeform. In: Pesticide residues in food - 1987:
Evaluations, Part II - Toxicology. Rome, Food and Agriculture
Organization of the United Nations, pp 25-26 (FAO Plant Production and
Protection Paper 86/2).
Fleming WJ, Heinz GH, & Schuler CA (1985) lethal and behavioral
effects of chlordimeform in bobwhite. Toxicology, 36: 37-47.
Folland DS, Kimbrough RD, Cline RE, Swiggart RC, & Schaffner (1978)
Acute hemorrhagic cystitis - Industrial exposure to the pesticide
chlordimeform. J Am Med Assoc, 239: 1052-1055.
Fritz H (1971) Reproduction study on C8514 (chlorphenamidine)
technical in rabbit - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 10071 004). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1975) Reproduction study on C8514 (chlorphenamidine)
technical in rat - Segment II: Test for teratogenic and embryotoxic
effects (Experiment No. 227533). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Fritz H (1978a) C8513 technical: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327735).
Fritz H (1978b) CGA 72651: Dominant lethal study (mouse). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 327737).
Fritz H, Becker H, & M�ller D (1978) CGA 72647 technical: Dominant
lethal study (mouse). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782201).
Gaines TB & Linder RE (1986) Acute toxicity of pesticides in adult and
weanling rats. Fundam Appl Toxicol, 7: 299-308.
Gale GT Jr & Hofberg AH (1985) Gas chromatographic determination of
chlordimeform in pesticide formulations: Collaborative study. J Assoc
Off Anal Chem, 68: 589-592.
Galloway SM, Armstrong MJ, Reuben C, Colman S, Brown B, Cannon C,
Bloom AD, Nakamura F, Ahmed M, Duk S, Rimpo J, Margolin BH, Resnick
MA, Anderson B, & Zeiger E. (1987) Chromosome aberrations and sister
chromatid exchanges in Chinese hamster ovary cells: Evaluations of 108
chemicals. Environ Mol Mutagen, 10(suppl): 1-175.
Geissb�hler H, Kossmann K, Baunok I, & Boyd VF (1971) Determination
of total residues of chlorphenamidine in plant and soil material by
colorimetry of thin layer and electron capture gas chromatography.
J Agric Food Chem, 19: 365-371.
Geyer R & Fattal F (1987) HPLC determination of the metabolite
4-chloro-o-toluidine in the urine of workers occupationally exposed to
chlordimeform. J Anal Toxicol, 11: 24-26.
Ghali GZ & Hollingworth RM (1985) Influence of mixed function
oxygenase metabolism on the acute neurotoxicity of the pesticide
chlordimeform in mice. Neurotoxicology, 6: 215-238.
Gifkins MR & Jacobson RA (1980) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine (chlordimeform), C10H13CIN2.
Cryst Struct Commun, 9: 959-963.
Glowa JR (1986) Acute and sub-acute effects of deltamethrin and
chlordimeform on schedule-controlled responding in the mouse.
Neurobehav Toxicol Teratol, 8: 97-102.
Goldman JM, Cooper RL, Laws SC, Rehnberg GL, Edwards TL, McElroy WK, &
Hein JF (1990) Chlordimeform-induced alterations in endocrine
regulation within the male rat reproductive system. Toxicol Appl
Pharmacol, 104: 25-35.
Goldman JM, Cooper RL, Edwards TL, Rehnberg G, McElroy WK, & Hein JF
(1991) Suppression of the luteinizing hormone surge by chlordimeform
in ovariectomized, steroid-primed female rats. Pharmacol Toxicol,
68: 131-136.
Goldman JM, Stoker TE, Perreault SD, Cooper RL, & Crider MA (1993)
Influence of the formamidine pesticide chlordimeform on ovulation in
the female hamster - Dissociable shifts in the luteinizing hormone
surge and oocyte release. Toxicol Appl Pharmacol, 121: 279-290.
Gordon CJ & Watkinson WP (1988) Behavioral and autonomic
thermoregulation in the rat following chlordimeform administration.
Neurotoxicol Teratol, 10: 215-219.
Gordon CJ, Long MD, & Stead AG (1985) Thermoregulation in mice
following acute chlordimeform administration. Toxicol Lett, 28: 9-15.
Gross (1977) Metabolism of C 8514 (chlordimeform) in cotton. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 19/77).
Gr�bner P (1977) [Thin-layer chromatographic determination of
chlordimeform in cucumbers and apples.] Nahrung, 21: 339-345
(in German).
Gu Q, Kui N-D, Wang M & Xue S-Z (1987) [Report on a mild case of
chlordimeform poisoning.] J Environ Toxicol, 1: 61-62 (in Chinese with
English abstract).
Gu Y-E, Wang M, Gu Q, & Dai G-J (1991) [Epidemiologic study on the
cancer mortality of employees and their relatives in Xin-Yang farm.]
J Jiangsu Occup Health, 1: 5-8 (in Chinese).
Guth JA & Senn U (1969) Galecron - Dissipation in a Swiss soil 1969 as
determined by chemical analysis. Basel, Switzerland Ciba-Geigy Ltd
(Unpublished report).
Haworth S, Lawlor T, Mortelmans K, Speck W, & Zeiger E (1983)
Salmonella mutagenicity test results for 250 chemicals. Environ
Mutagen, 1(suppl): 3-142.
He R-H (1989) [Analysis on the clinical symptoms of 52 acute
chlordimeform poisoned cases.] Occup Med, 16: 27 (in Chinese with
English abstract).
He D-Q, Peng B, & He P-L (1987) [Experience of treatment of
chlordimeform poisoning with atropine.] Hu-Nan Med, 4: 169 (in Chinese
with English abstract).
Henderson GR (1985) Report on the continuing use of chlordimeform for
cotton insect pest control by aerial and ground rig application in
1984/5 - NSW report. Pendle Hill, Ciba-Geigy Australia Ltd and
Kensington, Australia, Schering Pty Ltd (Unpublished report No. CGS
85/1).
Hermosin MC & Perez-Rodriguez JL (1981) Interaction of chlordimeform
with clay minerals. Clays Clay Miner, 29: 143-152.
Hill EF & Camardese ME (1986) Lethal dietary toxicities of
environmental contaminants and pesticides to Coturnix. Washington, DC,
US Department of the Interior, Fish and Wildlife Service, 147 pp (Fish
and Wildlife Technical Report No. 2).
Hill EF, Heath RG, Spann JW, & Williams JD (1975) Lethal dietary
toxicity of environmental pollutants to birds. Washington, DC, US
Department of the Interior, Fish and Wildlife Service, p 61 (Special
Scientific Report - Wildlife No. 191).
Hill DL, Shih T-W & Struck RF (1979) Macromolecular binding and
metabolism of the carcinogen 4-chloro-2-methylaniline. Cancer Res,
39: 2528-2531.
Hollingworth RM (1976) Chemistry, biological activity and uses of
formamidine pesticides. Environ Health Perspect, 14: 57-69.
Hollingworth RM, Leister J, & Ghali G (1979) Mode of action of
formamidine pesticides: an evaluation of monoamine oxidase as the
target. Chem-Biol Interact, 24: 35-49.
Holsapple MP & Yim GK (1981) Decreased gastrointestinal ulcerogenicity
of chlordimeform, a basic anti-inflammatory agent. Toxicol Appl
Pharmacol, 59: 107-110.
Homburger F, Friedell GH, Weisburger EK, & Weisburger JH (1972)
Carcinogenicity of simple aromatic amine derivatives in mice and Rats.
Toxicol Appl Pharmacol, 22: 280 (Abstract).
Honeycutt RC & Cassidy JE (1977) Extraction and characterization of
cottonseed metabolites from cotton injected with 14C-chlordimeform.
Greensboro, North Carolina, Ciba-Geigy Corporation (Unpublished report
No. ABR - 77087).
Hool G & Arni P (1983a) C8513 pure: Sister chromatid exchange studies
on somatic cells of Chinese hamster. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 830305).
Hool G & Arni P (1983b) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatogonia). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782902).
Hool G & Arni P (1983c) CGA 72647 pure: Chromosome studies in male
germinal epithelium (mouse spermatocytes). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782901).
Hool G & Arni P (1983d) CGA 72647 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 783101).
Hool G & Arni P (1983e) CGA 72647 pure: Sister chromatid exchange
studies on somatic cells of Chinese hamster. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 830312).
Hool G & Arni P (1983f) CGA 72651 pure: Chromosome studies in somatic
cells (Chinese hamster). Base], Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 774054).
Hool G & M�ller P (1978) C8513 technical: Chromosome studies in
somatic cells (Chinese hamster). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 40400577).
Hool G, Beilstein P, & Arni P (1983) C8513 technical: Chromosome
studies in male germinal epithelium (mouse spermatogonia). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 773062).
Hsu WH & Kakuk TJ (1984) Effect of amitraz and chlordimeform on heart
rate and pupil diameter in rats: Mediated by alpha2-adrenoreceptors.
Toxicol Appl Pharmacol, 73: 411-415.
Huang X-S, Ding S, & Chen P-H (1979) [Bioassay on the carcinogenicity
of chlordimeform.] In: Chinese Institute of Scientific Information and
Bureau of Science and Technology ed. [Selected papers on studies of
pesticides.] Beijing, Ministry of Chemical Industry, pp 789-792 (in
Chinese with English abstract).
Huang H-X, Xue K-L, & Shi G-J (1989) [Study on the status of pollution
on rice by chlordimeform.] Monograph submitted to the National
Pesticide Reviewing Committee (in Chinese with English abstract).
Hurni H & Sachsse K (1969) Report on the determination of the acute
oral LD50 to the dog of chlorfenamidin technical. Sisseln,
Switzerland, Tierfarm AG (Unpublished report).
IARC (1978) Some aromatic amines and related nitro compounds - hair
dyes, colouring agents and miscellaneous industrial chemicals. Lyon.
International Agency for Research on Cancer (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Man, Volume 16).
IARC (1983) Chlordimeform. Lyon, International Agency for Research on
Cancer, pp 61-72 (IARC Monographs on the Evaluation of Carcinogenic
Risk of Chemicals to Man, Volume 30)
IARC (1990) Some flame retardants and textile chemicals and exposures
in the textile manufacturing industry. Lyon, International Agency for
Research on Cancer, pp 123-137 (IARC Monographs on the Evaluation of
Carcinogenic Risks to Humans, Volume 48).
Ifflaender U (1977a) Distribution, degradation and excretion of
chlordimeform (CDF) in mice and rats. Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report No. 38/77).
Ifflaender U (1977b) Comparison of the urinary metabolite pattern of
mice and rats after oral application of "C-chlordimeform. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 39/77).
Iizuka H & Masuda T (1979) Residual fate of chlorphenamidine in rice
plant and paddy soil. Bull Environ Contam Toxicol, 22: 745-749.
IRPTC (1992) IRPTC data profile on chlordimeform. Geneva, United
Nations Environment Programme, International Register of Potentially
Toxic Chemicals.
Iwan J & Goller D (1975) Metabolic fate of chlordimeform in soil under
aerobic, anaerobic, and sterile conditions. Environ Qual Saf (Pestic),
3(suppl): 282-287.
Iwan J, Hoyer GA, Rosenberg D, & Goller D (1976) Transformations of
4-chloro-o-toluidine in soil: Generation of coupling products by
one-electron oxidation. Pestic Sci, 7: 621-631.
Janssen R, Boyes WK, & Dyer RS (1983) Effects of chlordimeform on the
brainstem auditory evoked response in rats. In: Hayes AW, Schnell RC,
& Miya TS ed. Developments in the Science and practice of toxicology,
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 533536.
Jiang J-K, Xu W-Y, Xu S-Q, Li J-L, Zhou F-D, Yang S-X, Zhong S-L, &
Zhu R-L (1985) [Survey on the hygienic status on spraying
chlordimeform with fine mist sprayer.] Labor Med, 2: 4-6 (in Chinese;
English translation).
Jiang X-Z, Li F, Xue S-Z, & Wang -L (1988) Carcinogenicity assay of
chlordimeform on dermal application in mice. Chin J Ind Hyg Occup Dis,
6: 138.
Johansen CA (1972) Toxicity of field-weathered insecticide residues to
four kinds of bees. Environ Entomol, 1: 393-394.
Johnson BT & Knowles CO (1970) Microbiological degradation of the
acaricide N-(4-chloro-o-tolyl)-N,N'-dimethylformamidine. Bull Environ
Contam Toxicol, 5: 158-170.
Johnson TL & Knowles CO (1981) Inhibition of rat platelet
5-hydroxytryptamine uptake by chlordimeform. Toxicol Lett, 9: 1-4.
Johnson TL & Knowles CO (1982) Interaction of formamidines with
the platelet 5-hydroxytryptamine uptake system. Gen Pharmacol,
13: 299-307.
Johnson TL & Knowles CO (1983) Influence of formamidines on biogenic
amine levels in rat brain and plasma. Gen Pharmacol, 14: 591-596.
Jurincic C, Horlbeck R, Metz K, & Klippel KF (1991) [Acute hemorrhagic
cystitis following chlordimeform poisoning.] Dtsch Med Wochenschr,
116: 816-818 (in German).
Kadir HA & Knowles CO (1981) Inhibition of rat brain monoamine oxidase
by insecticides, acaricides and related compounds. Gen Pharmacol,
12: 239-247.
Kale PG, Petty BT Jr, Walker S, Ford JB, Dehkordi N, Tarasia S, Tasie
BO, Kale R, & Sohni YR (1995) Mutagenicity testing of nine herbicides
and pesticides currently used in agriculture. Environ Mol Mutagen,
25: 148-153.
Kaloyanova F, Bainova A, & Zaprianov Z (1981) Inhibition of monoamine
oxidase activity after combined action of chlordimeform with the
antidepressant, nialamide. Arch Environ Contam Toxicol, 10: 1-8
Ke Z-H (1985) [Clinical features of 47 acute CDM poisonings by
swallowing.] Int Med, 11: 48-59 (in Chinese with English abstract).
Kenyon R, Garcia RJ, O'Grady HM, & Russell PJ (1993) A surveillance
programme for agricultural workers previously exposed to the suspect
bladder carcinogen chlordimeform - Proceedings of the 24th Congress of
the International Commission on Occupational Health, Nice, 26
September-1 October 1993.
Kimbrough RD (1980) Human health effects of selected pesticides,
chloroaniline derivatives. J Environ Sci Health, 15: 977-992.
Kimmel EC, Casida JE, & Ruzo LO (1986) Formamidine insecticides
and chloroacetanilide herbicides: Disubstituted anilines and
nitrosobenzenes as mammalian metabolites and bacterial mutagens.
J Agric Food Chem, 34: 157-161.
Knowles CO (1970) Metabolism of two accaricidal chemicals,
N'-(4-chloro-o-tolyl)-N, N-dimethylformamidine (chlorphenamidine)
and m-(((dimethylamino)methylene)amino)phenyl-carbamate hydrochloride
(formetanate). J Agric Food Chem, 18: 1038-1047.
Knowles CO (1976) Chemistry and toxicology of quinoxaline, organotin,
organofluorine, and formamidine acaricides. Environ Health Perspect,
14: 93-102.
Knowles CO (1991) Chlordimeform. In: Hayes WJ & Laws ER ed. Handbook
of pesticide toxicology. New York, London, San Francisco, Academic
Press Inc., vol 3. pp 1475-1486.
Knowles CO & Benezet HJ (1977) Mammalian metabolism of chlordimeform:
Formation of metabolites containing the urea moiety. J Agric Food
Chem, 25: 1022-1026.
Knowles CO & Johnson TL (1984) Effect of formamidines on
5-hydroxytryptamine uptake and biogenic amine levels in rat platelets.
Toxicology, 31: 91-98.
Knowles CO & Roulston WJ (1972) Antagonism of chlorphenaminine
toxicity to the cattle tick Boophilus microplus by piperonyl
butoxide. J Aust Entomol Soc, 11: 349-350.
Knowles CO & Sen Gupta AK (1969) Photodecomposition of the acaricide
N'-(4-chloro-o-tolyl)N,N-dimethylformamidine. J Econ Entomol,
62: 344-348.
Knowles CO & Sen Gupta AK (1970) N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine-14C(Galecron) and 4-chloro-o-toluidine
14C metabolism in the white rat. J Econ Entomol, 63: 856-859.
Knowles CO & Shrivastava SP (1973) Chlordimeform and related
compounds: Toxicological studies with house flies. J Econ Entomol,
66: 75-79.
Konopka EA & Heymann H (1977) Salmonella/microsome mutagenicity test
with chlordimeform. Greensboro, North Carolina, Ciba-Geigy Corporation
(Unpublished report).
Kopp R, Humpel M, Kuhne G, Fuchs R, & Rzadkiewicz M (1977)
Pharmacokinetics of chlordimeform following single and repeated oral
administration of 0.03 and 3 mg in female mice (1.2 and 120 mg/kg).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Kossmann K (1980) Summary report on chlordimeform monitoring
programmes conducted by Ciba-Geigy and Schering at applicator sites in
1979. Berlin, Germany, Schering AG and Basel, Switzerland, Ciba-Geigy
Ltd (Unpublished report).
Kossmann K, Geissb�hler H, & Boyd VF (1971) Specific determination of
chlorphenamidine (N'(4-chloro-o-tolyl)-N,N-dimethylformamidine) in
plants and soil material by colorimetry and thin layer and electron
capture gas chromatography. J Agric Food Chem, 19: 360-364.
Krick F (1994) Method for the determination of chlordimeform in honey.
Washington, DC, US Food and Drug Administration (Laboratory
Information Bulletin 3817).
Kurtz PH, Shaw G, Kelter A, & Jackson RJ (1987) Assessment of
potential acute health effects in agricultural workers exposed during
the application of chlordimeform. J Occup Med, 29: 592-595.
Lang R (1984) The mammalian spot test and its use for testing of
mutagenic and carcinogenic potential: experience with the pesticide
chlordimeform, its principal metabolites and the drug lisuride
hydrogen maleate. Mutat Res, 135: 219-224.
Lang R & Adler ID (1982) Studies on the mutagenic potential of the
pesticide chlordimeform and its principal metabolites in the mouse
heritable translocation assay, Mutat Res, 92: 243-248.
Langauer M & M�ller D (1977) C8513 technical : Nucleus anomaly test in
somatic interphase nuclei (Chinese hamster). Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 40050577).
Langauer M & M�ller D (1978a) CGA 72651 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 40550577).
Langauer M & M�ller D (1978b) CGA 72647 technical: Nucleus anomaly
test in somatic interphase nuclei (Chinese hamster). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 78/3002).
Leander J & MacPhail RC (1980) Effect of chlordimeform (a formamidine
pesticide) on schedule-controlled responding of pigeons. Neurobehav
Toxicol, 2: 315-321.
Leander JD, Tomlinson WT, Balster RL, & MacPhail RC (1984) Some
effects of the formamidine pesticide chlordimeform on the behaviour of
mice. Neurotoxicology, 5: 91-100.
Lemesch C, Wolf Y, & Gabai E (1987) Occupational exposure to
chlordimeform in Israel. Isr J Med Sci, 23: 1261-1262.
Leslie C, Reidy GF, Murray M, & Stacey NH (1988) Induction of
xenobiotic biotransformation by the insecticide chlordimeform, a
metabolite 4-chloro-o-toluidine and a structurally related chemical
o-toluidine. Biochem Pharmacol, 37: 2529-2535.
Li T-D, Liu B-J, & Mei C-J (1982) [Survey on 20 cases of acute
occupational chlordimeform poisoning.] Intermed Med, 3: 23-24
(in Chinese with English abstract).
Li F, Xue S-Z, Wang Y-L, Zhu S-Y, & Hu R-F (1985a) [Carcinogenic
action of chlordimeform given orally to mice in a lifetime
experiment.] Chin J Prev Med, 19: 154-156 (in Chinese with English
abstract).
Li F, Xue S-Z, Zhang R-W, Wang Y-L, Zhu Y-T, Sun S-S, Ding J-L, &
Feng B-J (1985b) (Studies on the health effect of employees in
chlordimeform production, and the mutagenic outcomes.] Labor Med,
2: 11-14 (in Chinese with English abstract).
Li F, Xue S-Z, Jiang X-Z, & Wang Y-L (1986) Report of dermal
carcinogenicity bioassay of chlordimeform in mice. J Ind Hyg Occup
Dis, 6: 76.
Limmer FB (1985) Ciba-Geigy and Schering world-wide exposure data
summary of personnel applying chlordimeform products covering the
period 1980-1984. Basel, Switzerland, Ciba-Geigy Ltd and Berlin,
Germany, Schering AG (Unpublished report).
Lin TH, North HH, & Menzer RE (1975) The metabolic fate of
chlordimeform [N-(4-chloro-o-tolyl) N'N'-dimethylformamidine] human
embryonic lung cell cultures. J Agric Food Chem, 23: 257-258.
Linder RE, Strader LF, Slott VL, & Suarez JD (1992) Endpoints of
spermatotoxicity in the rat after short duration exposures to fourteen
reproductive toxicants. Reprod Toxicol, 6: 491-505.
Ling W-Y, Zhang R-W, Gu Z-Y, Wang X-G, Zhang R-W, Jiang X-Z, Xue S-Z,
& Wang X-Z (1986) [Study on the urinary chlordimeform and its main
metabolite pCoT as index of biological monitoring in occupational
exposure.] Commun Labor Hyg Rail Syst, 8: 14-15 (in Chinese).
Lingren PD, Woffenbarger DA, Nosky JB, & Diaz M Jr (1972) Response of
Campoletis perdistinctus and Apanteles marginiventris to insecticides.
J Econ Entomol, 65: 1295-1300.
Liu Y-Z & Ke Z-H (1985) [Investigation on 53 cases of acute
chlordimeform poisoning.] Res Hyg, 14: 42-45 (in Chinese with English
abstract).
Liu X-L & Li W (1987) [Hemorrhage in upper digestive tract in
chlordimeform poisoning.] Occup Med, 14: 31 (in Chinese with English
abstract).
Liu Z & Mao Z (1980) [Studies on the analysis of metabolites of
pesticides. I. Gas chromatographic analysis of chlordimeform and its
metabolites in urine.] Hua Hsueh Pao, 38: 134-139 (in Chinese with
English Abstract).
Liu T-S & Zhang J-K (1985) [Poisoning due to chlordimeform, a new
pesticide.] Wuhan Med J, 9: 141-143 (in Chinese with English
abstract).
Liu H-G, Liu F-Q. Fang X-Z, & Wang X-M (1990) [Report on one case of
acute anemia due to chlordimeform poisoning.] PLA Med J, 15: 228
(in Chinese with English abstract).
Lu B-Q, Wang S-J, & Pong Y-F (1981) [Studies on the toxicology and
labor health of chlordimeform.] In: Chinese Institute of Scientific
Information and Bureau of Science and Technology ed. [Selected papers
on studies of pesticides.] Beijing, Ministry of Chemistry, pp 781-786
(in Chinese with English abstract).
Lund AE, Hollingworth RM, & Murdock LL (1979a) Formamidine pesticides:
A novel mechanism of action in lepidopterous larvae. Adv Pestic Sci,
3: 465-469.
Lund AE, Hollingworth RM, & Shamkland DL (1979b) Chlordimeform: Plant
protection by a sublethal, noncholinergic action on the central
nervous system. Pestic Biochem Physiol, 11: 117-128.
Lund AE, Hollingworth RM, & Yim GK (1979c) The comparative
neurotoxicity of formamidine pesticides. In: Neurotoxicology of
insecticidal pheromones, pp 119-137.
Lund AE, Shankland DL, Chinn D, & Yim GK (1978a) Similar
cardiovascular toxicity of the pesticide chlordimeform and lidocaine.
Toxicol Appl Pharmacol, 44: 357-365.
Lund AE, Yim GK, & Shankland DL (1978b) The cardiovascular toxicity of
chlordimeform: A local anesthetic-like action. In: Shankland DL,
Hollingworth RM, & Smyth T Jr ed. Pesticide and venom neurotoxicity.
New York, London, Plenum Press, pp 171-177.
Machin MV & Dingle JH (1977) Ultraviolet determination of
chlordimeform in cattle dipping baths and sprays. J Assoc Off Anal
Chem, 60: 696-698.
MacPhail RC & Leander JD (1980) Flavor aversions induced by
chlordimeform. Neurobehav Toxicol, 2: 363-365.
MacPhail RC & Leander JD (1981) Chlordimeform effects on
schedule-controlled behavior in rats. Neurobehav Toxicol Teratol,
3: 19-26.
Maddy KT, Knaak JB, & Gibbons DB (1986) Monitoring the urine of
pesticide applicators in California for residues of chlordimeform and
its metabolites, 1982-1985. Toxicol Lett, 33: 37-44.
Madhukar BV & Matsumura F (1979) Comparison of induction patterns of
rat hepatic microsomal mixed function oxidases by pesticides and
related chemicals. Pestic Biochem Physiol, 11: 301-308
Maitre L & Gfeller W (1975) Influence of compound 8514 (chlordimeform)
on the brain and liver monamineoxidase of rats. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report).
Maitre L, Felner A, Waldmeier P, & Kehr W (1978) Monoamine oxidase
inhibition in brain and liver of rats treated with chlordimeform.
J Agric Food Chem, 26: 442-446.
Maqueda C, Perez Rodriguez JL, Martin F, & Hermosin MC (1983) A study
of the interaction between chlordimeform and humic acid from a typic
chromoxerert soil. Soil Sci, 136: 75-81.
Maqueda C, Morillo E, & Perez Rodriguez JL (1989) Interaction in
aqueous solution of certain pesticides with fulvic acids from a
spodosol soil. Soil Sci, 148: 336-345.
Matsumara F & Beeman RW (1976) Biochemical and physiological effects
of chlordimeform. Environ Health Perspect, 14: 71-82.
Mattern GC, Liu C-H, Louis JB, & Rosen JD (1991) GC/MS and LC/MS
determination of 20 pesticides for which dietary oncogenic risk has
been estimated. J Agric Food Chem, 39: 700-704.
Mayer FL & Ellersieck MR (1986) Manual of acute toxicity:
Interpretation and database for 410 chemicals and 66 species of
freshwater animals. Washington, DC, US Department of the Interior,
Fish and Wildlife Service, 505 pp (Resource Publication 160).
Ming Z-P (1977) [Report of four cases of chlordimeform poisoning,] New
Med, 8: 82-83 (in Chinese with English abstract).
Moore B (1971) Chlordimeform residues - A summary report. Pendle Hill,
Ciba-Geigy Australia Ltd (Unpublished report No. 71/11/358).
Moser VC, McCormick JP, Creason JP, & MacPhail RC (1988) Comparison of
chlordimeform and carbaryl using a functional observational battery.
Fundam Appl Toxicol, 11: 189-206.
Muecke W, Hamboeck H, & Arni P (1979) Metabolic and pharmacokinetic
aspects of chlordimeform in rats and mice. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 37/79).
Murakami M & Fukami J (1974) Effect of chlorphenamide and its
metabolites an HeLa cells. Bull Environ Contam Toxicol, 11: 184-188.
Nathanson JA & Hunnicutt EJ (1981) N-demethylchlordimeform: A potent
agonist of octopamine-sensitive adenylate cyclase. Mol Pharmacol,
20: 68-75.
NCI (1979) Bioassay of 4-chloro-o-toluidine hydrochloride for possible
carcinogenicity. Bethesda, Maryland, National Cancer Institute
(Technical Report Series No. 165).
Neumann R & Voss G (1977) MAO inhibition, an unlikely mode of action
for chlordimeform. Experientia (Basel), 33: 23-24.
Nixon WB & Neal BE (1983) Chlordimeform residues in human urine
following dermal application. Greensboro, North Carolina, Ciba-Geigy
Corporation (Unpublished report No. ABR-83017).
Nui J-H, Ma H, Muo J-T, & Wu W-L (1990) [Report on 4 cases of
poisoning due to chlordimeform and organophosphate pesticides.]
Shang-Dong Ind Hyg, 2: 35 (in Chinese with English abstract).
O'Brien PJ, Elzen GW, & Vinson SB (1985) Toxicity of azinphosmethyl
and chlordimeform to parasitoid braconmellitor (Hymenoptera:
Braxonidae): lethal and reproductive effects. Environ Entomol,
14: 891-894.
Olson KL, Boush GM, & Matsumura F (1978) Behavioral effects of
perinatal exposure of chlordimeform in rats. Bull Environ Contam
Toxicol, 20: 760-768.
Ott MG & Langner RR (1983) A mortality survey of men engaged in the
manufacture of organic dyes. J Occup Med, 25: 763-770.
Palmer WA, Dingle JHP, & Heath AB (1977) Residues of chlordimeform in
bovine tissues and milk following application by a handspray. Aust J
Exp Agric Anim Husb, 17: 380-384.
Parkes HG (1984) Epidemiology of aromatic amine cancers. In: Searle CE
ed. Chemical carcinogens. Washington, DC, American Cancer Society,
pp 277-301 (ACS Monograph No. 182).
Perez-Rodriguez JL & Hermosin MC (1979) Adsorption of chlordimeform by
montmorillonite. In: Mortland MM & Farmer VC ed. Proceedings of the
International Clay Conference, Oxford, 1978. Amsterdam, Oxford, New
York, Elsevier Science Publishers, pp 227-234.
Pfister WR, Chin C, Noland V, & Yim GK (1978a) Similar pharmacological
actions of chlordimeform and local anesthetics. Pestic Biochem
Physiol, 9: 148-156.
Pfister WR, Hollingworth RM, & Yim GW (1978b) Increased feeding in
rats treated with chlordimeform and related formamidines: A new class
of appetite stimulants. Psychopharmacology (Berlin), 60: 47-51.
Phillips JR, Herzog GA, & Nicholson WF (1977) Effect of chlordimeform
on fruiting characteristics and yield of cotton. Ark Farm Res, 26: 4.
Pimley RW (1986) Toxicity of chlordimeform to the tsetse fly Glossina
morsitans. Insect Sci Appl, 7: 663-666.
Popp W & Norpoth K (1991) [Carcinogenic effects of chlordimeform and
4-chloro-o-toluidine.] Dtsch Med Wochenschr, 116: 1454 (in German).
Popp W, Schmieding W, Speck M, Vahrenholz C, & Norpoth K (1992)
Incidence of bladder cancer in a cohort of workers exposed to
4-chloro-o-toluidine while synthesising chlordimeform. Br J Ind Med,
49: 529-531.
Puri E & M�ller D (1983a) C8513 technical: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830304).
Puri E & M�ller D (1983b) CGA 72647 pure: Autoradiographic DNA repair
test on rat hepatocytes. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830311).
Puri E & M�ller D (1983c) C8513 technical: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. 830306).
Puri E & M�ller D (1983d) CGA 72647 pure: Autoradiographic DNA repair
test on human fibroblasts. Basel, Switzerland, Ciba-Geigy Lid
(Unpublished report No. 830313).
Rashid KA, Ercegovich CD, & Mumma RO (1984) Evaluation of
chlordimeform and degradation products for mutagenic and DNA-damaging
activity in Salmonella typhimurium and Escherichia coli. J Environ Sci
Health, B 19: 95-110.
Rieger JA, Robinson CP, Cox P, & Horst MA (1981) Cardiovascular
actions and interactions of chlordimeform in the dog. Bull Environ
Contam Toxicol, 27: 707-715.
Rieger JA, Allen LV Jr, & Fisher RC (1985) Extraction and analysis of
chlordimeform and demethylchlordimeform from human issue samples. Bull
Environ Contam Toxicol, 35: 397-405.
Robinson CP & Smith PW (1977) Lack of involvement of monoamine oxidase
inhibition in the lethality of acute poisoning by chlordimeform.
J Toxicol Environ Health, 3: 565-568.
Robinson CP, Smith PW, Zelenski JD, & Endecott BR (1975) Lack of an
effect of interference with amine mechanisms on the lethality of
chlordimeform in the rat. Toxicol Appl Pharmacol, 33: 380-383.
Rose JA (1969a) Degradation of chlorphenamidine by rat liver and
spleen homogenates with particular reference to the possible formation
of azo derivatives. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Rose JA (1969b) Formation of a symmetrical azo-derivative from
4-chloro-o-toluidine by coupled peroxidatic reactions. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Ross MSF & Leisten O (1989) Analysis of trace amounts of chlordimeform
in urine samples. Am Clin Lab, 8: 22-24.
Sachsse K, Suter P, Zak F, & Hess R (1978a) Lifespan feeding study in
mice with N-formyl-4-chloro-o-toluidine - Final report (Project No.
Siss M 4762/1. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Zak F, & Hess R (1978b) Lifespan feeding study in
mice with 4-chloro-o-toluidine-HCI - Final report (Project No. Siss M
04760/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979a)
Chlordimeform-HCL: 60-day feeding study in mice - Final report. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1979b)
Chlordimeform-HC1: 60-day feeding study in rat - Final report (Project
No. Siss 6138). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980a)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in the rat - Final
report (Project No. 404078). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980b)
N-formyl-4-chloro-o-toluidine: 60-day feeding study in mice - Final
report (Project No. 404178). Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980c) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
chlordimeform-HC1 - Final report (Project No. Siss. R 05658/1). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980d) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
N-formyl-4-chloro-o-toluidine - Final report (project No. Siss
5688/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Sachsse K, Suter P, Luetkemeier H, Zak F, & Hess R (1980e) Lifespan
(chronic toxicity and carcinogenicity) feeding study in rats with
4-chloro-o-toluidine- HC1 - Final report (project No. Siss R 05687/1).
Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report),
Salvisberg W, Neumann R, & Voss G (1980) Chlordimeform: Mode of toxic
action in various development stages of Spodoptera littoralis. J Econ
Entomol, 73: 193-196.
Sen Gupta AK & Knowles CO (1969) Metabolism of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine by apple seedlings.
J Agric Food Chem, 17: 595-600.
Sen Gupta AK & Knowles CO (1970) Galecron 14C(N'-(4-chloro-o-tolyl)
-N,N-dimethylformamidine) metabolism in the dog and goat. J Entomol,
63: 951-956.
Shimizu T & Fukami J (1983) Effect of chlordimeform and its analogues
on wandering behavior of the cabbage armyworm, Mamestra brassicae L.
(Lepidoptera:Noctuidae). Appl Entomol Zool, 18: 554-557.
Shopp GM Jr, McCay JA, & Holsapple MP (1985) Suppression of the
antibody response by a formamidine pesticide: dependence on the route
of exposure. J Toxicol Environ Health, 15: 293-304.
Sparks TC, Leonard BR. Schneider F. & Graves JB (1993) Ovicidal
activity and alteration of octopamine titers: Effects of selected
insecticides on eggs of the tobacco budworm (Lepidoptera: Noctuidae).
J Econ Entomol, 86: 294-300.
Stasik MJ (1988) Carcinomas of the urinary bladder in a 4-chloro-o-
toluidine cohort. Int Ach Occup Environ Health, 60: 21-24.
Stasik MJ (1991) [Bladder cancer from 4-chloro-o-toluidine.] Dtsch Med
Wochenschr, 116: 1444-1447 (in German).
Stoker TE, Goldman JM, Cooper RL, & McElroy WK (1991) Influence of
chlordimeform on alpha-adrenergic receptor-associated mechanisms of
hormonal regulation in the rat: pituitary and adrenocortical
secretion. Toxicology, 69: 257-268.
Strasser FF & M�ller D (1983a) CGA 72651 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Ltd (Unpublished report No. 782323).
Strasser FF & M�ller D (1983b) CGA 72647 pure: Point mutation assay
with mouse lymphoma cells, host-mediated assay. Basel, Switzerland,
Ciba-Geigy Lid (Unpublished report No. 782326).
Strasser FF & M�ller D (1984a) CGA 72647 technical: Point mutation
assay with mouse lymphoma cells, host-mediated assay. Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report No. 782325).
Strasser FF & M�ller D (1984b) CGA 72651: Point mutation assay with
mouse lymphoma cells. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished
report No. 782322)
Streibert HP & Dittrich V (1977) Toxicological response of insect eggs
and larvae to a saturated atmosphere of chlordimeform. J Econ Entomol,
70: 57-59.
Strong MB & Bull MS (1985a) Levels of chlordimeform equivalents found
in the urine of personnel involved in the ground rig application of
CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Ltd (Unpublished report
No. 85/6/1030).
Strong MB & Bull MS (1985b) Levels of chlordimeform equivalents found
in the urine of personnel involved in the aerial application of CGS
500 and CGS 800 in New South Wales during the 1984/1985 cotton growing
season. Pendle Hill, Ciba-Geigy Australia Lid (Unpublished report
No. 85/6/1029).
Su S-T (1977) [Report of 6 cases of chlordimeform poisoning]. Intermed
Med, 6: 19 (in Chinese with English abstract).
Su GC & Zabik MJ (1972) Photochemistry of bioactive compounds:
Photolysis of arylamidine derivatives in water. J Agric Food Chem,
20: 320-323.
Suter P, Luetkemeier H, Sachsse K. Zak F, & Hess R (1976a) 60-Day
feeding study in mice with 4-chloro-o-toluidine HC1 (Project No. Siss
4760). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Suter P, Luetkemeier H, Sachsse K, Zak F, & Hess R (1976b) 60-Day
feeding study in the rat with 4-chloro-o-toluidine HC1 (Project No.
Siss 4760). Basel, Switzerland. Ciba-Geigy Lid (Unpublished report).
Suter P, Zak F, Sachsse K, & Hess R (1978) lifespan feeding study in
mice with chlordimeform-HC1 - Final report (Project No. Siss. M
04761/1). Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report).
Tao X-M, Sun R-M, Wang M-S, Xue S-Z, Zhang R-W, Jiang X-Z & Wang Y-L
(1985) [investigation on the hygienic status of chlordimeform
production in Cai-Qiao pesticide factory.] Chin J Ind Hyg Occup Dis,
3: 272-274 (in Chinese).
Thomas WD, Craig GK, & Stacey NH (1990) Effects of chlordimeform and
its metabolite 4-chloro-o-toluidine on rat splenic T, B and
tumoricidal effector cells. Immunopharmacology, 19: 79-86.
Uk S & Dittrich V (1986) The behavior-modifying effect of
chlordimeform and endosulfan on the adult whitefly Bemisia tabaci
(Genn.) which attacks cotton in the Sudan. Crop Prot, 5: 341-347.
Voss G (1971) Galecron residues - German hops (green cones) and beer
1970. Basel, Switzerland, Ciba-Geigy Ltd (Unpublished report No. RVA
42/71).
Voss G & Burkhard N (1971) Galecron residues in milk, meat, fat, liver
and kidney of a Swiss cows. Basel, Switzerland, Ciba-Geigy Ltd
(Unpublished report No. RVA 24/7/S).
Voss G, Kossmann K, & Geissb�hler H (1973) Chlordimeform. In: Sherma J
& Zweig G ed. Analytical methods for pesticide plant growth
regulators. New York, London, Academic Press Inc., vol 7, pp 211-230.
Wang X-Z & Tong Q (1992) [Observation on 71 EKG of acute chlordimeform
poisoning cases.] Labor Prot Chem Ind, 13: 61-62 (in Chinese with
English abstract)
Wang CM, Narahashi T, & Fukami J (1975) Mechanism of neuromuscular
block by chlordimeform. Pestic Biochem Physiol, 5: 119-125.
Wang M, Zhou Z-J, Li H-L, Zhang R-W, Xue S-Z, Hong Z-R, Gu Q, Wang
X-M, & Feng Y-Q (1987) [Investigation on the hygienic status of
farmers spraying chlordimeform.] Chin J Ind Hyg Occup Dis, 5: 50-53
(in Chinese)
Wang Y-S & Dong C-R (1985) [Experience on treatment of 25 cases of
chlordimeform poisoning.] Sichuan Med, 6: 207 (in Chinese with English
abstract).
Watanabe H, Tsuda S, & Fukami J (1975) Effect of chlordimeform on
rectus abdominum muscle in frog. Pestic Biochem Physiol, 5:150-154.
Watanabe Y & Matsumura F (1987) Comparative metabolism of sulfamidine
and chlordimeform in rats. J Agric Food Chem, 35: 379-384
Watkinson WP (1985) Effects of chlordimeform on cardiovascular
functional parameters: Part 1. Lethality and arrhythmogenicity in the
geriatric rat. J Toxicol Environ Health, 15: 729-744.
Watkinson WP (1986a) Effects of chlordimeform on cardiovascular
functional parameters: Part 2. Acute and delayed effects following
intravenous administration in the postweanling rat. J Toxicol Environ
Health, 19: 195-206.
Watkinson WP (1986b), Effects of chlordimeform on cardiovascular
functional parameters: Part 3. Comparison of different routes of
administration in the postweanling rat. J Toxicol Environ Health,
19: 207-217.
Watkinson WP & Gordon CJ (1987) Effects of chlordimeform on heart rat
and body temperature of unanesthetized, unrestrained rats. Toxicol
Lett, 35: 209-216.
Watkinson WP, Highfill JW, & Gordon CJ (1989) Modulating effect of
body temperature on the toxic response produced by the pesticide
chlordimeform in rats. J Toxicol Environ Health, 28: 483-500.
Weisburger EK, Russfield AB, Homburger F, Weisburger JH. Boger E, Van
Donger CG, & Chu KC (1978) Testing of twenty-one environmental
aromatic amines for long-term toxicity or carcinogenicity. J Environ
Pathol Toxicol, 2: 325-356.
Wilson AG (1981) Field evaluation of formamidine insecticides and
Bacillus thuringiensis for selective control of Heliothis spp. on
cotton. Gen Appl Entomol, 13: 105-111.
Wiltrout RW, Ercegovich CD, & Ceglowski WS (1978) Humoral immunity in
mice following oral administration of selected pesticides. Bull
Environ Contam Toxicol, 20: 423-431.
Witkin JM & Leander JD (1982) Effects of the appetite stimulant
chlordimeform on food and water consumption of rats: Comparison with
chlordiazepoxide. J Pharmacol Exp Ther, 223: 130-134.
Witkonton S (1973) Fate of the pesticide N'-(4-chloro-o-tolyl)-N,
N-dimethylformamidine in fruit and soil, and toxicological properties
of some of its conversion products. Diss Abstr Int, 34: 3831.
Witkonton S & Ercegovich CD (1972) Degradation of
N'-(4-chloro-o-tolyl)-N,N-dimethylformamidine in six different
fruits. J Agric Food Chem, 20: 569-573.
Wolfenbarger DA (1974) Activity of chlordimeform HCI and chlordimeform
against arthropods attacking cotton. J Econ Etomol, 67: 445-446.
Wolfenbarger DA, Gomes ED, Robinson SH, & Parks LH (1979)
Chlordimeform residues from cotton leaves. J Econ Entomol,
72: 128-130.
Worthing CR (1979) The pesticide manual: A work compendium, 6th ed.
Croydon, United Kingdom, British Crop Protection Council, p 95.
Worthing CR & Walker SB (1983) The Pesticide manual. 7th ed. Croydon,
United Kingdom, British Crop Protection Council, p 2270.
Wu G (1982) [One case of chlordimeform poisoning.] Chin J Prev Med,
17: 275 (in Chinese with English abstract).
Wu S-Y, Liu Y-Y, Huang W-Z, Hong Q-F, Ling R-F, & Wang J-Y (1983) [On
chlordimeform poisoning - 24 case reports.] J Fujian Med Coll, 17:
42-44 (in Chinese with English abstract).
Wu D, Costa LG, & Murphy SD (1990) [The effect of pesticides-
chlordimeform and amitraz on inhibition of the binding of
3H-clonidine to alpha2-adrenoreceptor in rat forebrain tissue.]
Huaxi Yike Daxue Xuebao, 21: 17-21 (in Chinese with English abstract).
Xia S-C & Gao X-Z (1977) [Rescue after chlordimeform poisoning.]
Intermed Med, 6: 18-19 (in Chinese with English abstract).
Xie P-F (1983) [Analysis on the clinical features of chlordimeform
poisoning.] J Pract Med, 3: 145-146 (in Chinese with English
abstract).
Xu S-H (1987) [Experiences on treatment of chlordimeform poisoning.]
Hu-Nan Med, 4: 168-167 (in Chinese with English abstract).
Xue S-Z & Loosli R (1994) Formamidines. Toxicology, 91: 99-104.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J. & Wang YL (1990) The risk
assessment of occupational exposure to chlordimeform. Presented at the
23rd International Congress on Occupational Health, Montreal, Canada,
September 1990.
Xue S-Z, Wang M, Li F, Jiang X-Z, Zhou Z-J, & Wang YL (1991)
[Assessment on the carcinogenic risk of chlordimeform.] Environ Chem,
10: 54-60 (in Chinese).
Yang Q-Z (1984) [Rescue of a case of cardiac arrest due to
chlordimeform poisoning.] Jiangsu Med, 10: 38 (in Chinese with English
abstract).
Yim GK, Holsapple MP, Pfister WR, & Hollingworth RM (1978)
Prostaglandin synthesis inhibited by formamidine pesticides. Life Sci.
23: 2509-2516.
Youngman RR, Leigh TF, Kerby TA, Toscano NC, & Jackson CE (1990)
Pesticides and cotton: Effect on photosynthesis, growth, and fruiting.
J Econ Entomol, 83: 1549-1557.
Zak F, Sachsse K, Bath R, & Hess R (1973) One-year feeding study on
the rat with technical C-8514 (Project No. Siss 553). Basel,
Switzerland, Ciba-Geigy Ltd (Unpublished report).
Zhang Y-J, Yuan B-Q, Jin G-L, & Liu Y-Z (1976) [Report on two severe
cases of chlordimeform poisoning and their treatment.] J Rural Doctor,
5: 19 (in Chinese with English abstract).
Zhang R-W, Lin W-Y, Wang X-G, Gu Z-Y, Qu J-Y, Xue S-Z & Wang Y-L
(1986a) [Health effects of chlordimeform on the applicators.] Chin J
Prev Med, 20: 186 (in Chinese with English abstract).
Zhang R-W, Xue S-Z, & Wang Y-L (1986b) [Measurement of total amount of
body surface contamination with regional proportional sampling.] Occup
Med, 13: 2-3 (in Chinese with English abstract).
Zhou D-J (1987) [Bitter lesson on misuse of large dose of atropine on
chlordimeform poisoning.] Hu-Nan Med, 4: 169 (in Chinese with English
abstract).
Zimmer D, Mazurek J. Petzold G. & Bhuyan BK (1980). Bacterial
mutagenicity and mammalian cell DNA damage by several substituted
anilines. Mutat Res. 77: 317-326.
R�SUM�
1. Identit�, propri�t�s physiques et chimiques et m�thodes d'analyse
Le chlordim�form est une base de force moyenne qui forme des sels
stables avec les acides forts. A l'�tat pur, le chlordim�form et son
chlorhydrate sont des solides cristallins incolores. Le point de
fusion du chlordim�form base est de 32�C, celui du chlorhydrate �tant
de 225-227�C. La base est l�g�rement soluble dans l'eau (250 mg/litre)
et facilement soluble dans les solvants organiques, tandis que le
chlorhydrate est facilement soluble dans l'eau mais plus difficilement
dans les solvants organiques. La tension de vapeur du chlordim�form
est de 48 mPa � 20�C et son coefficient de partage entre l'eau et
l'octanol (log Kow) est �gal � 2,89. On peut faire appel � de
nombreuses m�thodes d'analyse pour la recherche et le dosage du
chlordim�form dans les v�g�taux, le sol, l'eau et l'urine.
2. Sources d'exposition humaine et environnementale
Le chlordim�form n'existe pas � l'�tat naturel. On le pr�pare
industriellement par condensation du r�actif de Vilsmeier (obtenu par
r�action du dim�thylformamide sur POCl3, SOCl2 ou COCl2) soit avec
la 4-chloro- o-toluidine, soit avec la l' o-toluidine, suivie d'une
chloration du d�riv� interm�diaire obtenu. On l'utilise comme
acaricide � large spectre et il est principalement actif contre les
formes mobiles des acariens et des tiques ainsi que contre les oeufs
et les premiers stades de certains l�pidopt�res. Il agit en phase
gazeuse aussi bien que par contact. Les premiers temps de son
utilisation, on l'employait pour traiter des cultures tr�s vari�es
comme les fruits � p�pins, les drupes, les choux et autres l�gumes,
les raisins, le houblon, les agrumes, les cerises et les fraises. On
l'utilise aussi en bains contre les tiques des bovins. Ces derni�res
ann�es son usage s'est g�n�ralement limit� au coton, mais on continue
tout de m�me � l'utiliser sur le riz dans certains pays. Depuis
1988/89 il n'est plus homologu� dans la plupart des pays. En Chine, la
production a cess� en 1992, de m�me que la vente en 1993.
3. Transport, distribution et transformation dans l'environnement
Bien que sa tension de vapeur ait une valeur moyenne, le
chlordim�form ne s'�vapore pas autant qu'on le penserait des surfaces
v�g�tales. Sa stabilit� vis-�-vis de l'hydrolyse d�pend fortement du
pH; il est stable en milieu acide mais s'hydrolyse rapidement en
milieu alcalin. Le chlordim�form est capable de s'adsorber sur les
mati�res organiques dissoutes.
Dans le sol, la disparition du chlordim�form est essentiellement
imputable � l'action des microorganismes et, pour une moindre part, �
l'hydrolyse chimique. Malgr� la solubilit� du compos� dans l'eau, on
ne trouve gu�re de traces de lessivage, ce qui peut s'expliquer par
une adsorption aux mat�riaux argileux ou aux mati�res organiques du
sol ainsi que par la biod�gradation. Les principaux m�tabolites sont
la N-formyl-4-chloro- o-toluidine et la 4-chloro- o-toluidine.
Les plantes fixent le chlordim�form pr�sent dans le sol en
proportion faible mais mesurable et la concentration est suffisante
pour affecter les ravageurs qui se nourrissent � leurs d�pens. En
traitement foliaire, la p�n�tration du chlordim�form dans la cuticule
est limit�e. Le chlordim�form est rapidement d�compos� par les
v�g�taux. Les principaux m�tabolites sont le d�m�thylchlordim�form, la
N-formyl-4-chloro -o-toluidine et la 4-chloro -o-toluidine, cette
derni�re n'�tant pas produite par toutes les plantes.
Dans le sol, le chlordim�form et ses m�tabolites disparaissent
selon une cin�tique du premier ordre avec une demi-vie de 20 � 40
jours.
Les �tudes de bioaccumulation montrent que les organismes
aquatiques ne fixent qu'une petite quantit� de chlordim�form et que
celui-ci s'�limine rapidement une fois ces organismes replac�s en eau
pure.
4. Concentrations dans l'environnement et exposition humaine
On n'a pas proc�d� � des mesures de concentration dans l'air ou
l'eau. Apr�s traitement de rizi�res, on a retrouv� des r�sidus allant
jusqu'� 2900 �g/kg dans les 5 premiers centim�tres du sol et jusqu'�
150 �g/kg dans les 5 centim�tres suivants.
On a fix� des teneurs limites en r�sidus pour un grand nombre de
produits crus et dans certains cas, pour des pr�parations contenant
ces produits. Les limites maximales de r�sidus fix�es par le Codex ont
�t� supprim�es.
Il y a eu des cas d'exposition au chlordim�form au cours de la
pr�paration, de la formulation et de l'�pandage de ce produit. Depuis
quelques ann�es, on utilise la concentration urinaire totale du
chlordim�form et de ses m�tabolites pour surveiller l'exposition et il
y a d'ailleurs une bonne corr�lation entre cette concentration et le
degr� de contamination cutan�e. Dans les industries cotonni�res o�
l'on a soumis les ouvriers agricoles � une surveillance g�n�rale de la
concentration urinaire en chlordim�form, on a constat� que les plus
expos�s �taient les chargeurs, les laveurs et les m�caniciens et les
moins expos�s les signaleurs et les pilotes.
5. Cin�tique et m�tabolisme chez les animaux de laboratoire et
l'Homme
Chez les mammif�res, le chlordim�form est facilement r�sorb� au
niveau des voies digestives ainsi que par la voie transcutan�e. Il est
ensuite rapidement excr�t� � raison de 80% environ dans l'urine et de
10-15% dans les mati�res f�cales. De petites quantit�s de r�sidus sont
pr�sentes au bout de 10 jours dans tous les tissus mais rien n'indique
qu'il y ait bioaccumulation. Apr�s application cutan�e chez l'Homme,
on constate �galement une excr�tion urinaire rapide.
On retrouve dans l'urine plusieurs m�tabolites du chlordim�form
sous forme oxyd�e et conjugu�e, � savoir principalement la
N-formyl-4-chloro- o-toluidine, et la 4-chloro- o-toluidine.
In vitro, on retrouve les m�mes m�tabolites, mais avec pr�dominance
de la 4-chloro- o-toluidine.
6. Effets sur les mammif�res de laboratoire et les syst�mes d'�preuve
in vitro
Les �preuves pratiqu�es sur un certain nombre d'esp�ces montrent
que la chlordim�form pr�sente une toxicit� aigu� mod�r�e par la voie
orale et la voie transcutan�e. Chez le rat, les principaux m�tabolites
sont peu toxiques par voie orale. Chez le lapin, le chlordim�form ne
provoque qu'une l�g�re irritation oculaire et cutan�e. Apr�s
exposition de courte ou de br�ve dur�e au chlordim�form ou � ses
m�tabolites, on peut observer, au niveau des constantes
h�matologiques, des modifications qui sont imputables au traitement et
on constate, � dose �lev�e, certains signes qui d�notent une
hyperplasie de l'�pith�lium des canaux biliaires et de la vessie. Il
n'y pas d'accroissement de la fr�quence des tumeurs chez le rat. Chez
la souris, on observe, apr�s administration par voie alimentaire de
chlordim�form, de N-formyl-4-chloro -o-toluidine ou de
4-chloro- o-toluidine, une augmentation, li�e � la dose, des tumeurs
malignes h�morragiques d'origine vasculaire appartenant � la classe
des h�mangio-endoth�liomes, dont la pr�sence entra�ne un accroissement
de la mortalit� parall�le � la dose.
Le chlordim�form n'a pas d'effet ind�sirable sur les diff�rents
aspects de la fonction de reproduction et il n'a aucun pouvoir
t�ratog�ne.
Le chlordim�form a fait l'objet d'un grand nombre d'�preuves de
g�notoxicit� in vitro et in vivo. Aucune d'elles n'a donn� de
r�sultat positif, �tant entendu qu'il s'agissait de la mati�re active
et non de formulations. Par ailleurs, un certain nombre d'observations
sporadiques non confirm�es font �tat d'une activit� mutag�ne induite
par la N-formyl-4-chloro- o-toluidine et par la 4-chloro- o-
toluidine. Il n'existe qu'une seule description de transformations
cellulaires provoqu�es par le chlordim�form et par la 4-chloro -o-
toluidine. Chez des souris et des rats trait�s par le chlordim�form,
on a constat� une que le compos� se liait � l'ADN des cellules
h�patiques. A dose beaucoup plus �lev�e, il se forme chez les m�mes
animaux un important adduit hydrophobe.
Le chlordim�form provoque des effets pharmacologiques et
biochimiques divers chez l'animal, et notamment des effets
cardiovasculaires, une hypothermie, une hyperexcitabilit�, une
modification des fonctions visuelle et auditive ainsi que la
modulation des amines biogen�se et des enzymes pharmacom�tabolisantes.
7. Effets sur l'Homme
Les intoxications aigu�s se traduisent par une fatigue, des
naus�es et une perte d'app�tit, avec, dans les cas graves, somnolence,
cyanose, besoin imp�rieux d'uriner, cystite, effets cardiovasculaires
(tachy-cardie, bradycardie, anomalies de l'ECG), coma et �tat de choc.
En g�n�ral, la r�cup�ration est totale.
Apr�s une exposition de longue dur�e au chlordim�form, on peut
observer encore d'autres sympt�mes tels que des douleurs abdominales,
des d�mangeaisons et des �ruptions (en cas d'exposition cutan�e)
accompagn�s d'une h�maturie macroscopique ou micro-scopique. On a
signal� de nombreux cas d'intoxication pr�sentant des sympt�mes
d'exposition de longue dur�e parmi les ouvriers d'unit�s de production
de chlordim�form et des ouvriers agricoles.
Les donn�es �pid�miologiques obtenues � la suite de cas
d'exposition professionnelle montrent qu'il existe une forte
corr�lation entre l'exposition � la 4-chloro -o-toluidine et le
cancer de la vessie. En revanche, on n'a gu�re obtenu d'�l�ments qui
militeraient en faveur d'une association entre ce type de cancer et
l'exposition au chlordim�form.
8. Effets sur les autres �tres vivants au laboratoire et dans leur
milieu naturel
Apr�s �pandage de chlordim�form sur le sol, on n'a pas observ�
d'effets sensibles sur les populations de champignons, de bact�ries ou
d'actinomyc�tes terricoles.
Il n'existe pas de donn�es toxicologiques de laboratoire
concernant les invert�br�s dul�aquicoles. En pr�sence de
chlordim�form, il y a inhibition de la croissance des larves
d'hu�tres, avec une CE50 de 5,7 mg/litre. Pour la crevette rose,
le seul crustac� �tudi�, la CL50 � 96 h a �t� trouv�e �gale �
7,1 mg/litre et des valeurs allant de 1 � 54 mg/litre ont �t� obtenues
pour le m�me param�tre chez les poissons. On ne poss�de aucune donn�e
relative � la toxicit� chronique pour les esp�ces aquatiques.
L'ensemble des r�sultats de laboratoire et des donn�es recueillies sur
le terrain indique que le compos� est toxique pour de nombreux
arthropodes terrestres non vis�s.
Chez l'abeille, la toxicit� de contact se traduit par une DL50
de 120 �g/g, la toxicit� par voie orale correspondant � une valeur de
187 �g/g. Trois heures apr�s l'�pandage de chlordim�form sur de la
luzerne, l'exposition de certaines esp�ces d'abeilles aux r�sidus
encore pr�sents sur les plantes, n'a provoqu� aucune mortalit�.
La CL50 par voie alimentaire varie de >1000 � > 5000/kg de
nourriture pour diverses esp�ces d'oiseaux.
9. Evaluation des risques pour la sant� humaine et des effets sur
l'environnement
On a observ� des signes d'intoxication aigu� chez des
travailleurs qui, peut-�tre par suite de l'inobservation des mesures
de s�curit�, avaient �t� fortement expos�s � du chlordim�form au cours
de la pr�paration ou de l'utilisation de ce produit. Comme, � ce qu'il
semble, il n'est plus produit ni utilis� nulle part dans le monde, il
ne devrait plus y avoir de cas d'intoxication aigu�. Le risque li� �
une exposition chronique et en particulier, le risque de cancer de la
vessie, subsistera cependant pendant de nombreuses ann�es. Il faut
continuer � effectuer des contr�les sanitaires chez les sujets qui ont
subi une exposition notable pour avoir travaill� dans des ateliers de
production de chlordim�form ou avoir v�cu dans des zones rurales o� le
produit �tait largement utilis�.
Comme il s'agit d'un produit qui n'est plus en usage, on n'a pas
proc�d� � une �valuation quantitative du risque qu'il repr�sente pour
l'environnement. On ne pense pas que celui-ci puisse subir des effets
nocifs � long terme qui soient attribuables � l'utilisation ant�rieure
du produit.
10. Conclusions et recommandations
Il existe un risque non n�gligeable que le chlordim�form produise
des effets toxiques imm�diats ou � plus long terme chez les individus
expos�s. Les donn�es disponibles accr�ditent l'id�e d'une association
entre l'augmentation de l'incidence du cancer de la vessie chez
l'homme et l'exposition � la 4-chloro- o-toluidine et, dans une
moindre mesure, au chlordim�form.
Le chlordim�form ne persiste pas dans l'environnement et il ne
devrait donc pas y avoir d'effets nocifs � long terme sur celui-ci qui
r�sulteraient de l'usage ant�rieur du compos�.
Il n'est pas recommand� de reprendre la production ou l'usage du
chlordim�form dans un but commercial. Les stocks existants doivent
�tre �limin�s selon les r�gles de s�curit�.
Les personnes expos�es au chlordim�form de par leur profession
doivent �tre soumises � des examens cytologiques v�sicaux et � une
recherche syst�matique de l'h�maturie dans le cadre d'un programme
g�n�ral de d�pistage.
RESUMEN
1. Identidad, propiedades f�sicas y qu�micas y m�todos anal�ticos
El clordimeformo es una base de fuerza media que forma sales
estables con �cidos fuertes. Tanto el clordimeformo como su sal
hidroclorada en estado puro son s�lidos cristalinos incoloros. El
punto de fusi�n del clordimeformo (base) es de 32�C, mientras que el
de la sal hidroclorada es de 225-227�C. El clordimeformo (base) es
poco soluble en agua (250 mg/litro) y f�cilmente soluble en
disolventes org�nicos, mientras que la sal hidroclorada es f�cilmente
soluble en agua pero menos soluble en disolventes org�nicos. El
clordimeformo (base) tiene una presi�n de vapor de 48 mPa a 20�C y un
log Kow de 2,89. Se dispone de una amplia gama de m�todos anal�ticos
para detectar y cuantificar la presencia de clordimeformo en las
plantas, el suelo, el agua y la orina.
2. Fuentes de exposici�n humana y ambiental
El clordimeformo no existe en la naturaleza. Se produce
comercialmente mediante condensaci�n del reactivo de Vilsmeier
(obtenido por reacci�n de la dimetilformamida con POCl3, SOCl2
o COCl2) con 4-cloro- o-toluidina o bien con o-toluidina y
cloraci�n ulterior del producto intermedio resultante. Se ha utilizado
como acaricida de amplio espectro y act�a principalmente contra las
formas m�viles de �caros y garrapatas, as� como contra los huevos y
las cris�lidas en estado inicial de algunos insectos del orden
Lepid�ptera. Es activo en la fase de vapor, as� como por contacto.
Cuando comenz� a utilizarse, se aplicaba a productos de una amplia
variedad de cultivos, tales como frutas de pipas, frutas de hueso,
berzas, hortalizas, uvas, l�pulo, c�tricos, manzanas, peras, cerezas y
fresas. Tambi�n se utilizaba en ba�os antiparasitarios para combatir
las garrapatas del ganado. En los �ltimos a�os, su uso se limitaba por
lo general al algod�n, aunque en algunos pa�ses se segu�a aplicando al
arroz. En la mayor�a de los pa�ses, su registro se abandon�
voluntariamente en 1988/1989. En China dej� de producirse en 1992 y de
venderse en 1993.
3. Transporte, distribuci�n y transformaci�n en el medio ambiente
El clordimeformo tiene una presi�n de vapor moderada pero su
evaporaci�n de la superficie de las plantas es inferior a la que
cabr�a prever. La estabilidad hidrol�tica del clordimeformo depende
mucho del pH; es estable en condiciones �cidas pero se hidroliza
r�pidamente en condiciones alcalinas. El clordimeformo tiene un
potencial de adsorci�n a la materia org�nica disuelta.
Hay dispersi�n del clordimeformo en el suelo, principalmente por
acci�n microbiana y, en menor medida, por hidr�lisis qu�mica. Pese a
la solubilidad del clordimeformo en agua, hay pocos indicios de
lixiviaci�n, lo que puede deberse a su adsorci�n a minerales
arcillosos y a la materia org�nica del suelo, as� como a su
biodegradaci�n. Los principales metabolitos son la N-formil-
4-cloro- o-toluidina y la 4-cloro- o-toluidina.
La absorci�n del clordimeformo por las plantas a partir del suelo
es escasa pero detectable, y suficiente para afectar a las plagas que
se alimentan de ellas. El clordimeformo aplicado a las hojas s�lo
tiene una capacidad limitada de penetrar en las capas cuticulares. El
clordimeformo se degrada r�pidamente en las plantas. Sus principales
metabolitos son el demetilclordimeformo, la N-formil-4-cloro-
o-toluidina y la 4-cloro- o-toluidina, aunque no todas las plantas
estudiadas produjeron 4-cloro- o-toluidina.
El clordimeformo y sus metabolitos se dispersan en el suelo
conforme a una cin�tica de primer orden, con una semivida de 20-40
d�as.
Los estudios sobre bioacumulaci�n indican una escasa absorci�n
del clordimeformo por los organismos acu�ticos y una r�pida depuraci�n
de �stos despu�s de haber sido transferidos a un agua limpia.
4. Niveles medioambientales y exposici�n humana
No se han medido los niveles de clordimeformo en el aire ni en el
agua. Tras la aplicaci�n de clordimeformo a unos arrozales, en el
suelo se hallaron residuos en concentraciones de hasta 2900 �g/kg en
los 5 cm primeros de profundidad, y de 150 �g/kg en los 5 cm
siguientes.
Se establecieron niveles m�ximos de residuos aplicables a una
amplia variedad de productos sin elaborar y, en algunos casos, de
residuos trasladados a los alimentos elaborados. Los l�mites m�ximos
aplicables a los residuos de clordimeformo se han retirado del Codex
Alimentarius.
Hab�a exposici�n ocupacional al clordimeformo durante la
fabricaci�n, la formulaci�n y la aplicaci�n del producto. En los
�ltimos a�os la exposici�n se ha vigilado mediante la determinaci�n de
los niveles totales de clordimeformo y de sus metabolitos presentes en
la orina, y hay una correlaci�n positiva entre el nivel en la orina y
el grado de contaminaci�n cut�nea. Entre los trabajadores agr�colas de
los algodonales sometidos a una amplia vigilancia de la excreci�n
urinaria de clordimeformo, los niveles m�s altos de exposici�n se
hallaban en los cargadores, lavadores y mec�nicos, y los niveles m�s
bajos en los obreros se�alizadores y pilotos.
5. Cin�tica y metabolismo en animales de laboratorio y en el ser
humano
Los mam�feros absorben f�cilmente el clordimeformo por el tracto
gastrointestinal y a trav�s de la piel. Lo excretan r�pidamente,
alrededor del 80% por la orina y del 10-15% por las heces. Al cabo de
unos 10 d�as se observan niveles bajos de residuos en todos los
tejidos y no hay indicios de bioacumulaci�n. Tras la administraci�n
cut�nea a seres humanos, se observa una excreci�n r�pida semejante por
la orina.
Varios metabolitos oxidados y conjugados del clordimeformo se
excretan por la orina; los principales son el demetilclordimeformo, la
N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. En
estudios in vitro se han observado los mismos metabolitos, siendo el
principal la 4-cloro- o-toluidina.
6. Efectos en mam�feros de laboratorio y en sistemas de pruebas
in vitro
En ensayos realizados en varias especies, el clordimeformo
administrado por v�a oral y cut�nea ha mostrado tener una toxicidad
aguda moderada. Los principales metabolitos han mostrado tener una
toxicidad oral baja en ensayos realizados en ratas. El clordimeformo
provoca solamente una ligera irritaci�n cut�nea y ocular en el conejo.
Tras una exposici�n breve o prolongada de ratones y ratas al
clordimeformo o a sus metabolitos pueden observarse cambios asociados
al tratamiento en los par�metros hematol�gicos y, con dosis elevadas,
indicios de hiperplasia del epitelio de las v�as biliares y de la
vejiga. El clordimeformo no aumenta la incidencia de tumores en las
ratas. En los ratones, despu�s de administrar a trav�s de la dieta
clordimeformo N-formil-4-cloro- o-toluidina o 4-cloro- o-
toluidina, se observa, de forma relacionada con la dosis, un aumento
de los tumores malignos hemorr�gicos de origen vascular clasificados
como hemangioendoteliomas malignos, que producen un aumento de la
mortalidad asociado con la dosis.
El clordimeformo no afecta a los par�metros reproductivos ni
tiene potencial teratog�nico.
Se ha ensayado el clordimeformo en una amplia variedad de pruebas
de genotoxicidad in vitro e in vivo. No se han comunicado
reacciones positivas a ninguna de esas pruebas, en las que se ensay�
clordimeformo en estado puro. Adem�s, se han comunicado varios
informes espor�dicos y no confirmados de actividad mutag�nica inducida
por la N-formil-4-cloro- o-toluidina y la 4-cloro- o-toluidina. Un
informe describe una inducci�n de la transformaci�n celular por efecto
tanto del clordimeformo como de la 4-cloro- o-toluidina. En el h�gado
de los ratones y las ratas expuestos se producen enlaces con el ADN.
Se ha observado un importante aducto hidrof�bico, en los ratones en
niveles mucho mayores que en las ratas.
El clordimeformo induce diversos efectos farmacol�gicos y
bioqu�micos en los animales, tales como cambios cardiovasculares,
hipotermia, hiperexcitabilidad, efectos sobre las funciones visual
central y auditiva y modulaci�n de las aminas biog�nicas y de las
enzimas que metabolizan f�rmacos.
7. Efectos en el ser humano
La intoxicaci�n aguda causa fatiga, n�useas, p�rdida del apetito
y, en casos m�s graves, somnolencia, cianosis, micci�n imperiosa,
cistitis, efectos cardiovasculares (taquicardia, bradicardia,
alteraciones del ECG), coma y choque. En general se produce una
recuperaci�n completa de la intoxicaci�n aguda.
Otros s�ntomas asociados a la exposici�n cr�nica al
clordime-formo son dolores abdominales, prurito y exantemas
(exposici�n cut�nea), as� como hematuria macrosc�pica y microsc�pica.
Se ha comunicado un gran n�mero de casos con s�ntomas cl�nicos de
exposici�n cr�nica tanto entre los obreros de las plantas de
producci�n de clordimeformo como entre los trabajadores agr�colas.
Los indicios epidemiol�gicos relacionados con la exposici�n
ocupacional muestran una fuerte asociaci�n entre la exposici�n al
metabolito 4-cloro- o-toluidina y la incidencia de c�ncer de vejiga
en el ser humano. Actualmente se dispone de pocos indicios de
asocia-ci�n entre la exposici�n al clordimeformo y el c�ncer de vejiga
en el ser humano.
8. Efectos en otros organismos en el laboratorio y en el medio
ambiente
No se observaron efectos significativos en poblaciones de hongos
de la tierra, bacterias o actinomicetos tras la aplicaci�n de
clordime-formo al suelo.
No existen datos de laboratorio sobre la toxicidad en los
invertebrados de agua dulce. El clordimeformo inhibi� el crecimiento
de larvas de ostras, con una CE50 de 5,7 mg/litro. La CL50 a las
96-h para los camarones rosados, �nico crust�ceo estudiado, fue de
7,1 mg/litro y los valores de la CL50 a las 96-h para los peces
oscilaron entre 1 y 54 mg/litro. No se dispone de datos sobre
toxicidad acu�tica cr�nica. La combinaci�n de datos obtenidos en el
laboratorio y sobre el terreno revela que el clordimeformo es t�xico
para una amplia gama de artr�podos terrestres no combatidos.
Con respecto a las abejas, se ha comunicado una DL50 de
toxicidad por contacto de 120 �g/g y una DL50 de toxicidad oral de
187 �g/g. No se produjo mortalidad sobre el terreno tras la exposici�n
de especies de abejas a los residuos presentes en la alfalfa tres
horas despu�s del rociado.
La CL50 en la dieta de varias especies de p�jaros oscil� entre
>1000 y >5000 mg/kg de dieta.
9. Evaluaci�n de los riesgos para la salud humana y efectos en el
medio ambiente
La exposici�n intensa durante la producci�n o la utilizaci�n,
debida posiblemente a la insuficiencia de las medidas de seguridad,
dio lugar a s�ntomas de intoxicaci�n aguda en los trabajadores. Como
se ha notificado que se ha suspendido la producci�n y la utilizaci�n
de clordimeformo en todo el mundo, no deber�an producirse nuevos casos
de intoxicaci�n aguda. Sin embargo, el riesgo asociado a la exposici�n
cr�nica, en particular el riesgo de c�ncer de vejiga, seguir� siendo
preocupante durante muchos a�os. Deber�a proseguir el reconoci-miento
m�dico de las personas que han estado muy expuestas en las plantas de
producci�n y en las comunidades rurales donde se haya aplicado
extensamente el clordimeformo.
Dado que el clordimeformo ha dejado de utilizarse, no se ha
realizado ninguna evaluaci�n cuantitativa de los riesgos para el medio
ambiente. A largo plazo no se prev�n efectos perjudiciales para el
medio ambiente como consecuencia de la utilizaci�n de clordime-formo
en el pasado.
10. Conclusiones y recomendaciones
El clordimeformo tiene un potencial significativo para causar
tanto toxicidad inmediata como a largo plazo en las personas
expuestas. La informaci�n de que se dispone actualmente apunta a una
asociaci�n entre una mayor incidencia de c�ncer de vejiga en el ser
humano y la exposici�n a la 4-cloro- o-toluidina y, en menor medida,
al clordimeformo.
El clordimeformo no persiste en el medio ambiente, por lo que a
largo plazo no se prev�n efectos perjudiciales como consecuencia de su
utilizaci�n en el pasado.
Se recomienda que el clordimeformo no se produzca comercialmente
ni se utilice en el futuro. Las reservas existentes deber�an
eliminarse sin correr riesgos.
Las personas expuestas profesionalmente al clordimeformo deber�an
participar en un programa de reconocimiento m�dico que comprenda
citolog�a urinaria y detecci�n de hematuria.
