
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 136
1,1,1-TRICHLOROETHANE
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
Published under the joint sponsorship of
the United Nations Environment Programme,
the International Labour Organisation,
and the World Health Organization
First draft prepared by Dr S. Dobson,
(Institute of Terrestrial Ecology, United Kingdom)
and Dr A.A. Jensen (Danish Technological Institute)
World Health Orgnization
Geneva, 1990
The International Programme on Chemical Safety (IPCS) is a
joint venture of the United Nations Environment Programme, the
International Labour Organisation, and the World Health
Organization. The main objective of the IPCS is to carry out and
disseminate evaluations of the effects of chemicals on human health
and the quality of the environment. Supporting activities include
the development of epidemiological, experimental laboratory, and
risk-assessment methods that could produce internationally
comparable results, and the development of manpower in the field of
toxicology. Other activities carried out by the IPCS include the
development of know-how for coping with chemical accidents,
coordination of laboratory testing and epidemiological studies, and
promotion of research on the mechanisms of the biological action of
chemicals.
WHO Library Cataloguing in Publication Data
1,1,1-Trichloroethane.
(Environmental health criteria ; 136)
1.Trichloroethanes - adverse effects 2.Trichloroethanes - toxicity
3.Environmental exposure 4.Environmental pollutants
I.Series
ISBN 92 4 157136 5 (NLM Classification: QV 633)
ISSN 0250-863X
The World Health Organization welcomes requests for permission
to reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1992
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material
in this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR 1,1,1-TRICHLOROETHANE
1. SUMMARY
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES,
AND ANALYTICAL METHODS
2.1 Identity
2.2 Physical and chemical properties
2.2.1 Physical characteristics
2.2.2 Chemical reactivity
2.2.3 Commercial products
2.3 Conversion factors
2.4 Analytical methods
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Production processes
3.2 Production levels
3.3 Uses
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION,
AND TRANSFORMATION
4.1 Distribution and transport between media
4.1.1 Atmospheric transport
4.1.2 Transport in water
4.1.3 Transport in soil
4.2 Degradation and transformation
4.2.1 Abiotic degradation
4.2.1.1 In the atmosphere
4.2.1.2 In water
4.2.2 Biodegradation
4.2.2.1 Anaerobic
4.2.2.2 Aerobic
4.3 Bioaccumulation
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air
5.1.2 Water
5.1.3 Sediment and soil
5.1.4 Biota
5.2 General population exposure
5.2.1 Food
5.2.2 Drinking-water
5.2.3 Air
5.2.4 Consumer products and cosmetics
5.3 Occupational exposure
6. KINETICS AND METABOLISM IN LABORATORY
ANIMALS AND HUMANS
6.1 Absorption
6.1.1 Animal studies
6.1.1.1 Inhalation
6.1.1.2 Oral absorption
6.1.1.3 Skin absorption
6.1.2 Human studies
6.1.2.1 Inhalation
6.1.2.2 Skin contact
6.2 Distribution and retention
6.2.1 Animal studies
6.2.2 Human studies
6.3 Metabolic transformation
6.3.1 Animal studies
6.3.2 Human studies
6.3.3 Metabolic interactions
6.4 Elimination
6.4.1 Animal studies
6.4.2 Human studies
6.5 Biological monitoring
6.6 Bioaccumulation
7. EFFECTS ON LABORATORY MAMMALS AND
IN VITRO TEST SYSTEMS
7.1 Acute toxicity
7.1.1 Irritation
7.1.2 Short-term exposure
7.1.2.1 Inhalation
7.1.2.2 Oral administration
7.2 Long-term exposure
7.3 Reproductive toxicity, embryotoxicity, and
teratogenicity
7.4 Mutagenicity
7.5 Carcinogenicity
7.5.1 Oral administration
7.5.2 Inhalation
7.6 Immunotoxicity and sensitization
7.7 Interactions
7.8 Mechanisms of action
8. EFFECTS ON HUMANS
8.1 Controlled human studies
8.1.1 Single exposure period
8.1.2 Repeated exposure
8.2 Accidental exposure
8.2.1 Confined spaces at workplaces
8.2.2 Solvent abuse
8.2.3 Medical use
8.2.4 Ingestion
8.2.5 Drinking-water contamination
8.3 Effects on the skin and eyes
8.4 Long-term occupational exposure
8.5 Interactions
9. EFFECTS ON OTHER ORGANISMS IN THE
LABORATORY AND FIELD
9.1 Microorganisms
9.2 Aquatic organisms
9.3 Terrestrial organisms
10. EVALUATION OF HUMAN HEALTH RISKS AND
EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
10.2 Evaluation of effects on the environment
11. RECOMMENDATIONS FOR PROTECTION OF
HUMAN HEALTH AND THE ENVIRONMENT
12. FURTHER RESEARCH
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
RESUME
RESUMEN
WHO TASK GROUP ON ENVIRONMENTAL HEALTH
CRITERIA FOR 1,1,1-TRICHLOROETHANE
Members
Dr L.A. Albert, Consultores Ambientales Asociados, Xalapa, Veracruz,
Mexico (Vice-Chairman)
Dr A.H. El-Sebae, Faculty of Agriculture, Alexandria University,
Alexandria, Egypt
Dr S. Fairhurst, Medical Division, Health and Safety Executive,
Bootle, Merseyside, United Kingdom (Chairman)
Dr B. Gilbert, Technology Development Company (CODETEC), Cidade
Universitaria, Campinas, Brazil
Dr A.A. Jensen, Danish Technological Institute, Taastrup, Denmark
Dr T. Kawamoto, Department of Environmental Health, University of
Occupational and Environmental Health, Japan School of Medicine,
Kitakyushu City, Japan
Ms I.R. Nielsen, Environment Section, Organic Materials Division,
Building Research Establishment, Garston, Watford, United Kingdom
Dr B. Wahlstrom, Department of Science and Technology, National
Chemicals Inspectorate, Solna, Sweden
Mr R. Walentowicz, Exposure Assessment Group, US Environmental
Protection Agency, Washington, DC, USA
Mrs G. Wood, Criteria Section, Bureau of Chemical Hazards,
Environmental Health Directorate, Health Protection Branch, Health
& Welfare, Tunney's Pasture, Ottawa, Canada
Observers
Dr M.A. Collins, ICI Chemicals & Polymers, Occupational Health,
Runcorn, Cheshire, United Kingdom
Dr C. De Rooij, Solvay, Brussels, Belgium
Secretariat
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood
Experimental Station, Abbots Ripton, Huntingdon, Cambridgeshire,
United Kingdom (Rapporteur)
Dr M. Gilbert, International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland (Secretary)
Mr P.D. Howe, Institute of Terrestrial Ecology, Monks Wood
Experimental Station, Abbots Ripton, Huntingdon, Cambridgeshire,
United Kingdom (Temporary Adviser)
NOTE TO READERS OF THE CRITERIA DOCUMENTS
Every effort has been made to present information in the criteria
documents as accurately as possible without unduly delaying their
publication. In the interest of all users of the Environmental Health
Criteria documents, readers are kindly requested to communicate any
errors that may have occurred to the Director of the International
Programme on Chemical Safety, World Health Organization, Geneva,
Switzerland, in order that they may be included in corrigenda.
* * *
A detailed data profile and a legal file can be obtained from the
International Register of Potentially Toxic Chemicals, Palais des
Nations, 1211 Geneva 10, Switzerland (Telephone No. 7988400 or
7985850).
ENVIRONMENTAL HEALTH CRITERIA FOR
1,1,1-TRICHLOROETHANE
A WHO Task Group on Environmental Health Criteria for
1,1,1-Trichloroethane met at the Institute of Terrestrial Ecology
(ITE), Monks Wood, United Kingdom, from 20 to 24 May 1991. Dr M.
Roberts, Director, ITE, welcomed the participants on behalf of the
host institution and Dr M. Gilbert opened the meeting on behalf of the
three cooperating organizations of the IPCS (UNEP/ILO/WHO). The Task
Group reviewed and revised the draft criteria document and made an
evaluation of the risks for human health and the environment from
exposure to 1,1,1-trichloroethane.
The first draft of this document was prepared by Dr S. Dobson (ITE)
and Dr A.A. Jensen (Danish Technological Institute). Dr M. Gilbert and
Dr P.G. Jenkins, both members of the IPCS Central Unit, were
responsible for the technical development and editing, respectively.
The efforts of all who helped in the preparation and finalization
of the document are gratefully acknowledged.
ABBREVIATIONS
cAMP cyclic adenosine monophosphate
cGMP cyclic guanosine monophosphate
CNS central nervous system
NOAEL no-observed-adverse-effect level
NOEL no-observed-effect level
OECD Organization for Economic Cooperation and Development
ppt parts per trillion
1. SUMMARY
1,1,1-Trichloroethane is a chlorinated hydrocarbon which is
manufactured from vinyl chloride or vinylidene chloride by
chlorination. The world production was approximately 680 000 tonnes
in 1988. It is a colourless, volatile liquid with a characteristic
odour, and its vapour is more dense than air. It is mainly used in
metal degreasing and as a solvent in many industrial and consumer
products, including adhesives, spot removers, and aerosol cans. It is
also a chemical intermediate. Technical trichloroethane usually
contains 3-8% stabilizers to prevent degradation and the formation of
hydrochloric acid; this protects metal parts from corrosion. It is
non-flammable under normal conditions, but the vapour burns at high
temperatures and, during welding operations, its degradation products
include the poisonous gas phosgene. Contact with aluminium,
magnesium, and their alloys may result in very violent reactions.
1,1,1-Trichloroethane reaches the environment readily. Owing to
its long residence time in the troposphere (about 6 years) and low
biodegradability, it is now ubiquitous in the environment, even far
from industrial areas. Concentrations of up to 86 µg/m3 (16 ppb,
w/w) have been determined in air sampled near industries producing or
handling the compound.
Trichloroethane is mobile in soils and reaches the ground water.
Concentrations of up to 1600 µg/litre have been found in ground or
surface waters. This may be a source of contamination for
drinking-water supplies.
It is estimated that 15% of the annual release of
1,1,1-trichloroethane is transported to the stratosphere where it
causes ozone depletion by liberating chlorine atoms.
Acute toxic effects have been observed in bioassays with
crustaceans and fish at concentrations above 7 mg/litre. Limited
information suggests that bioaccumulation in aquatic organisms is low.
The small amount of data makes it difficult to evaluate any effects on
terrestrial organisms.
Humans are exposed to 1,1,1-trichloroethane principally by
inhalation, and the substance is then rapidly absorbed into the body.
Exposure by skin contact or ingestion may also occur. Trichloroethane
is distributed widely in body tissues and crosses the blood-brain and
placental barriers. It has also been found in human breast milk, but
is not thought to be bioaccumulated. The main route of elimination is
exhalation of unchanged compound.
The acute and chronic toxicities of 1,1,1-trichloroethane are
relatively low, but, under conditions of high exposure, there is a
risk of toxic effects. Such conditions may occur in cases of
occupational exposures, solvent abuse or accidents. Since the solvent
is volatile and the vapour is much more dense than air, unexpectedly
high and dangerous concentrations may occur in confined spaces such as
"empty" storage tanks. This has caused several fatal and near-fatal
poisonings at workplaces and elsewhere.
The critical effect in humans is on the central nervous system.
Observable effects range from slight behavioural changes (accompanied
by mild eye irritation) at 1.9 g/m3 (350 ppm) to unconsciousness and
respiratory arrest at higher concentrations. However, fatal cardiac
anomalies may also occur. Trichloroethane is less toxic to the liver
than are most other organochlorine solvents. The no-observed-effect
level (NOEL) for humans appears to be in the region of 1.35 g/m3
(250 ppm).
No adequate study of human carcinogenic effects has been
published. However, a long-term inhalation study on rats and mice
exposed to 8.1 g/m3 (1500 ppm) gave no evidence of any carcinogenic
effect. 1,1,1-Trichloroethane does not have significant genotoxic
potential.
Developmental toxicity, but not teratogenicity, has been observed
in rats and rabbits at concentrations that were toxic to the mother
animals. The limited epidemiological evidence on reproductive effects
is inconclusive.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Identity
1,1,1-Trichloroethane is an organochlorine solvent belonging to
the family of chlorinated alkanes.
Chemical structure:
Cl H
| |
Cl -- C -- C -- H
| |
Cl H
Empirical formula: C2H3Cl3
Relative molecular
mass: 133.4
IUPAC name: 1,1,1-trichloroethane
CAS name: ethane, 1,1,1-trichloro-
Some common
synonyms: methylchloroform, MC, 1,1,1-TCE
Some common
trade names: Chlorothene, Aerothene TT, Alpha-T,
Genklene, Inhibisol
CAS registry
number: 71-55-6
EEC identity
number: 602-013-00-2
RTECS number: KJ2975000
2.2 Physical and chemical properties
2.2.1 Physical characteristics
In its pure state, 1,1,1-trichloroethane is a colourless,
volatile liquid with a characteristic chloroform-like odour. The
odour threshold is reported to be around 540 mg/m3 (100 ppm)
(Stewart, 1968).
Trichloroethane has two structural isomers. The
1,1,2-trichloroethane isomer can be an impurity in the manufacture of
1,1,1-trichloroethane and it is known to have a different spectrum of
reactivity.
Some physical and chemical properties of pure 1,1,1-
trichloroethane are listed in Table 1.
2.2.2 Chemical reactivity
1,1,1-Trichloroethane has no recorded flash point and is
therefore sometimes considered as non-flammable. However, it
decomposes and/or oxidizes at high temperatures (> 300 °C) to
hydrochloric acid and dichloroethene, together with some phosgene
(Hardie, 1964). When catalysed by metal salts, especially aluminium
compounds, degradation occurs at lower temperatures (Dreher, 1989) and
decomposition in air to hydrogen chloride, carbon dioxide, and traces
of chlorine occurs slowly at ambient temperatures (Pearson &
McConnell, 1975). The formation of phosgene by the photooxidation of
1,1,1-trichloroethane during welding operations may be considerable
(Dahlberg et al., 1973). 1,1,1-Trichloroethane can burn in the vapour
state (de Nevers, 1986) and in admixture with air forms explosive
mixtures (Wrightson & Santon, 1988; Bretherick, 1989). Contact with
aluminium, magnesium, and their alloys may result in very violent
reactions (Bretherick, 1981). Trichloroethane reacts slowly with
water, but more rapidly with alkaline solutions such as an aqueous
suspension of calcium hydroxide, forming 1,1-dichloroethene
(vinylidene chloride) (Hardie, 1964). Hydrolysis, which is very slow
at 20 °C but rapid at 80 °C (see Table 7), occurs in the presence of
water or aqueous acids, yielding hydrochloric and acetic acids
(Gerkens & Franklin, 1989).
2.2.3 Commercial products
Analytical grade 1,1,1-trichloroethane has a purity of > 99.0%
and contains no added stabilizers (Fluka, 1988).
Commercially available technical and solvent grade products have
a purity of 90-95% and usually contain 3-8% stabilizers, mainly to
prevent the generation of hydrochloric acid and to avoid corrosion of
metal parts (Fluka, 1988). The stabilizers used are chemical
compounds, such as nitromethane, N-methyl pyrrole, 1,4-dioxane,
butylene oxide, 1,3-dioxolane, nitroethane, toluene, diisopropylamine,
methyl ethyl ketone, isobutyl alcohol, and 2-butanol (IARC, 1979;
Clayton & Clayton, 1981; US EPA, 1984; CEC, 1986).
Table 1. Physical and chemical properties of 1,1,1-trichloroethane
Melting point -30.4 °C Weast (1986)
Boiling point (at 760 mmHg) 74.1 °C Weast (1986)
Density 1.3390 Weast (1986)
Vapour density (air = 1) 4.6 US EPA (1984)
Vapour pressure (at 20 °C) 13.3 kPa (100 mmHg) Weast (1986)
Refractive index (at 20 °C) 1.4379 Weast (1986)
Concentration in 16.7% Clayton &
saturated air Clayton (1981)
Solubility in water 0.3 g/litre at 25 °Ca IARC (1979)
0.95 g/litre at 20 °C Archer (1979)
0.480 g/litre at 20 °C Pearson (1982)
Soluble in acetone, benzene, chloroform, Windholz (1983)
methanol, ether, ethanol, Weast (1986)
carbon disulfide, carbon CEC (1986)
tetrachloride
Partition coefficients Veith et al.
octanol/water (log Pow) 2.47 (measured) (1980)
water/air (at 20 °C) 0.71 Pearson (1982)
blood/air (at 37 °C) 3.3 US EPA (1984)
Flammability nonflammable under normal CEC (1986)
conditions, vapour burns
at high temperature
Auto-ignition temperature 537 °C Archer (1979)
Explosive limits in air 8.0-10.5 vol % Archer (1979)
at 25 °C
a US EPA (1984) reported a water solubility of 4.4 g/litre at 25 °C.
Twenty-two samples of stabilized technical 1,1,1-trichloroethane
were shown to contain potential mutagens or carcinogens such as
vinylidene chloride, dichloroethane, and 1,2-epoxybutane (Henschler et
al., 1980).
2.3 Conversion factors
1 ppm = 5.40 mg/m3
1 mg/m3 = 0.185 ppm
2.4 Analytical methods
The first step of an analytical method for routine measurements
of trichloroethane in air is sampling either on activated charcoal
(tubes or diffusion samplers) filters and extraction by a solvent
(e.g., carbon disulfide) or on a polymer trap and desorption by
heating. This is followed by determination by gas chromatography (GC)
combined with either electron capture detector (ECD), flame ionization
detector (FID) or mass spectrometry (MS). GC is also used for the
determination of trichloroethane in other types of samples (water,
sediment, blood, etc.) by, for example, headspace analysis.
Indicator tubes may be used for preliminary surveys of air
levels. The detection limit is about 270 mg/m3 (50 ppm)
(Drägerwerk, 1986).
Some examples of analytical methods are summarized in Table 2.
Table 2. Analytical methods
Medium Specification Sampling method Analytical method Detection limit Reference
Air trap on charcoal extract GC-FID 300 ng/sample Niosh (1977)
with carbon disulfide Otson & Chan (1987)
Air GC-ECD 125 mg/m3 Henschler (1978)
(10 litres air)
Air trap on Porapak-N, desorb GC-ECD 160 mg/m3 Russell & Shadoff (1977)
at 200 °C, dry over MgSO4 GC-MS (30 ppt)
Water sparge with helium, trap GC-MS Coleman et al. (1976)
on Tenax GC, desorb by heat
Water direct injection of headspace GC-ECD 0.05 µg/litre Piet et al. (1978)
gas
Water headspace air, trap on Tenax GC Pereira & Hughes (1980)
Water drinking-water trap on XAD-2 GC-MS 0.1 µg/litre Otson (1987)
Blood rat arterial headspace gas collected over GC-FID 0.5 µg (6.4 µg/g Westerberg & Larsson (1982)
75-150 µl at 75 °C, direct blood)
injection
Blood human GC-ECD 0.05 mg/litre Henschler (1978)
Blood human extraction with hexane GC-ECD < 0.07 µmol/litre Pekari & Aitio (1985)
Brain rat headspace gas collected 0.5 µg (2.5 µg/g tissue) Westerberg & Larsson (1982)
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
Appraisal
1,1,1-Trichloroethane is a man-made chemical; there are no
natural sources. In 1988, world-wide production was approximately
680 000 tonnes per year. However, the annual production level is
expected to decline progressively due to international agreements on
the control of ozone-depleting substances. It is used mainly in
metal degreasing and as a solvent in many consumer products, but also
finds use as an intermediate in the production of other chemicals.
3.1 Production processes
All 1,1,1-trichloroethane in the environment is anthropogenic in
origin, since it does not occur naturally. It is usually produced by
the hydrochlorination of vinyl chloride to 1,1-dichloroethane,
followed by thermal chlorination to 1,1,1-trichloroethane (Fishbein,
1979). 1,1-Dichloroethene (vinylidene chloride) can also be directly
hydrochlorinated to 1,1,1-trichloroethane, and direct chlorination of
ethane or chloroethane can be used. However, the latter methods may
generate other halogenated hydrocarbons as by-products (Fishbein,
1979).
3.2 Production levels
The estimated world production capacity of 1,1,1-
trichloroethane was 480 000 tonnes/year in 1973 (McConnell et al.,
1975) and 680 000 tonnes/year in 1988 (Midgley, 1989). About half the
production is in the USA and 100 000 tonnes is produced in Japan
annually. The production of 1,1,1-trichloroethane in Japan more than
doubled in the period from 1979 to 1989. The production in the
European Economic Community in 1979 was estimated to be 140 000
tonnes/year (Torslov, 1988). Production capacity is about 100 000
tonnes in Germany, while in 1973 the United Kingdom and France
produced 20 000 and 11 000 tonnes, respectively. The annual
production in eastern Europe is estimated to be less than 1000 tonnes.
The western European consumption of 1,1,1-trichloroethane in 1985
was estimated to be 173 000 tonnes, the worldwide value being about
500 000 tonnes.
Information prepared by the European Chemical Industry Federation
(CEFIC, 1986) suggested that consumption of 1,1,1-trichloroethane in
western Europe increased during the 1970s but stabilized during the
1980s.
The estimated annual worldwide production of
1,1,1-trichloroethane is shown in Table 3.
Table 3. Production and sales of 1,1,1-trichloroethane (tonnes/year)
Year Worldwide W. European W. European USA Japanese
productiona productionb salesc productiond productione
1970 155
1971 175
1972 227
1973 279 245
1974 314 92
1975 307 86
1976 407 99
1977 480 109 289
1978 524 120 292
1979 506 147 77
1980 552 144 315 86
1981 549 146 274 88
1982 514 142 89
1983 541 140 226 96
1984 600 144 111
1985 588 150 394 120
1986 609 205 138 291 128
1987 627 203 130 314 131
1988 678 218 129 340 139
1989 222 130 164
1990 229 122 338
a Midgley (1989)
b CEFIC (1991)
c CEFIC (1986)
d NIOSH (1979); Midgley (1989); CMR (1991)
e MITI (1990)
Midgley (1989) examined the annual sales of 1,1,1-trichloroethane
and grouped uses into three categories: those that result in emissions
for less than 6 months, for 1 year, and for more than one year. He
found that applications, such as solvent cleaning, that lead to rapid
emission, account for 95 to 97% of the annual reported production of
1,1,1-trichloroethane. A geographical breakdown of the emission data
into global regions revealed that during the period 1980-1988 between
90% and 94% of the total production of 1,1,1-trichloroethane was sold
to the industrial north, i.e. above latitude 30°N.
Under the agreement of the London Conference of the Montreal
Protocol (June, 1990), use of 1,1,1-trichloroethane will be
discontinued.
3.3 Uses
1,1,1-Trichloroethane is used mainly in metal cleaning/
degreasing and as a solvent in various formulations, including
adhesives, paints, varnishes, inks, dry cleaning agents, and
typewriter correction fluids. It is also utilized as a solvent in
aerosols, and it can be used as an additive to raise the flash point
of many flammable solvents. 1,1,1-Trichloroethane also finds uses as
a coolant and lubricant in metal cutting oils, as a solvent in textile
dyeing, for cleaning plastic moulds, and as a developer for printed
circuit boards.
Formerly, 1,1,1-trichloroethane was used as a solvent for various
insecticides, and, together with ethylene gas, for degreasing citrus
fruits and post-harvest fumigation of strawberries.
1,1,1-Trichloroethane is also a chemical intermediate in the
production of vinylidene chloride. In the USA, this accounts for 23%
of the total consumption of 1,1,1-trichloroethane, but this use
appears to represent only 5-10% of the production elsewhere.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
Appraisal
1,1,1-Trichloroethane has a residence time of about 6 years in
the troposphere, where it is oxidized to trichloroacetaldehyde and
trichloroacetic acid. It reaches the stratosphere in significant
amounts, which results in ozone depletion through the liberation of
reactive chlorine atoms. The ozone-depleting potential of
1,1,1-trichloroethane is ten times lower than that of
trichlorofluoromethane (CFC-11), and the global-warming potential is
about 40 times lower.
In water, 1,1,1-trichloroethane is slowly dehydrochlorinated to
1,1-dichloroethene and hydrolysed to ethanoic acid, the former
process being favoured by alkalinity. 1,1,1-Trichloroethane does not
bind to soil particles and thus leaches readily into ground water.
Biodegradation to 1,1-dichloroethane and chloroethane has been
reported to occur under anaerobic conditions.
1,1,1-Trichloroethane does not appear to bioaccumulate.
4.1 Distribution and transport between media
1,1,1-Trichloroethane enters the environment primarily via
evaporation to the atmosphere, although some is discharged in
industrial effluents. McConnell et al. (1975) reported rapid transfer
of 1,1,1-trichloroethane from water to air and concluded that,
irrespective of whether 1,1,1-trichloroethane enters the environment
via water or air, a wide distribution of the chemical is likely.
Neely (1982) used the vapour pressure, water solubility, and
relative molecular mass of 1,1,1-trichloroethane to assess its
partitioning between major environmental compartments. The
partitioning pattern was estimated to be 99.92% in air, 0.08% in
water, and zero in either benthic sediments or ground. Torslov (1988)
obtained similar results using a computer model based on the
principles of the "fugacity" model of Mackay and predicted an
environmental distribution of 99.29% in air, 0.7% in water, 0.01% in
soil, sediment and aquatic biota, and zero in suspended aquatic
material.
In 1978 it was estimated that 97.3% of the 1,1,1-trichloroethane
used in the USA was released to the environment. Of this, 86% was
released to the air, 1% to water, and about 10% was disposed of as
waste (Fischer et al., 1982). It was also estimated that only about
6% of the 1,1,1-trichloroethane produced is emitted to the air or
waste water during production, the remainder being released during
use. The global-warming potential of 1,1,1-trichloroethane, an effect
which results from its accumulation in the atmosphere, is about 40
times lower than that of trichlorofluoromethane (CFC-11) and 14 times
lower than that of carbon tetrachloride.
4.1.1 Atmospheric transport
Atmospheric transport is the major route by which
1,1,1-trichloroethane is transported in the environment. Hence, it
has been measured in air at remote locations (see section 5.1.1) and
in rainfall (see section 5.1.2).
The ratio between concentrations of 1,1,1-trichloroethane in the
northern and southern hemispheres is decreasing (Khalil & Rasmussen,
1984). Lower levels are found at mid latitudes during the summer
because of greater removal by hydroxyl radicals (see section 4.2).
In a study of a whole range of halocarbons, Edwards et al. (1982)
estimated that, of the chlorine in the troposphere, 45% originated
from industry and 55% from natural or non-industrial sources of methyl
chloride. Of the anthropogenic chlorine, 13% derived from the release
of 1,1,1-trichloroethane.
In the troposphere, 1,1,1-trichloroethane is predominantly
degraded by oxidation, but some is rained out and some is transferred
to the stratosphere. Prior to 1977 the residence time in the
troposphere was estimated to be 1 to 2 years (NRC, 1976), but the use
of more reliable estimates of globally averaged levels has led to a
calculated residence time of 5-7 years (Khalil & Rasmussen, 1984;
Prinn et al., 1987; Midgley, 1989).
1,1,1-Trichloroethane has a long enough atmospheric lifetime for
a certain portion to reach the stratosphere. In the early 1970s, it
was calculated that approximately 12% of 1,1,1-trichloroethane
reaching the troposphere would be transferred to the stratosphere.
Revised figures have calculated that nearer to 15% is transferred
(McConnell & Schiff, 1978; Singh et al., 1982).
In the stratosphere, 1,1,1-trichloroethane is degraded by
photochemical processes, forming chlorine atoms and thence chlorine
radicals that have the potential to deplete stratospheric ozone (see
section 4.2.1).
4.1.2 Transport in water
In aquatic systems, volatilization is the major route for
1,1,1-trichloroethane removal. Oxidation and hydrolysis do not appear
to play an important part in the aquatic fate of 1,1,1-trichloroethane
(Dilling et al., 1975). Dilling et al. (1975) found
1,1,1-trichloroethane to be rapidly evaporated from water. At 25 °C,
90% evaporation occurred within 60-80 min from an aqueous solution
containing 1 mg 1,1,1-trichloroethane/litre, the half-life being 20
min. The rate of disappearance was examined in the presence of
various natural and added "contaminants", such as clays, limestone,
peat, and other chemicals. None of these contaminants affected the
disappearance rate by more than a factor of 2. It was concluded that
no adsorption onto sediment or solids had taken place.
4.1.3 Transport in soil
The adsorption of 1,1,1-trichloroethane to soil is proportional
to the organic carbon content of the soil (Urano, 1985). It has a low
adsorption to silt loam (Chiou et al., 1979). These authors presented
data which show that adsorption by soil organic matter occurs via a
partitioning process rather than by physical adsorption.
In a dissertation report, Drake (1987) studied the fate in
aerobic unsaturated soils. 1,1,1-Trichloroethane was applied to
columns containing sandy or sandy loam soils (organic carbon contents
of 0.69% and 3.76%, respectively), and mass balances were carried out
to determine the fate of the compound. The processes studied were
volatilization, transport, biodegradation, and sorption. There was no
indication of biodegradation, but volatilization appeared to play a
major role in the mass balance. Approximately 90% and 45% of the
1,1,1-trichloroethane applied to the sandy and sandy loam soils,
respectively, were estimated to be lost via volatilization.
Breakthrough of 1,1,1-trichloroethane was documented in both soils,
and effluent levels of up to 10% of the 1 mg/litre influent level were
observed after 25 days (Drake,1987).
4.2 Degradation and transformation
4.2.1 Abiotic degradation
4.2.1.1 In the atmosphere
Using a concentration of 0.44 % (w/v) 1,1,1-trichloroethane in
air in an enclosed flask exposed to external solar diurnal and
climatic variations of incident radiation and temperature, Pearson &
McConnell (1975) estimated the half-life to be 26 weeks. This is
similar to the values found for dichloromethane and chloroform, and is
much slower than most other chlorohydrocarbons. The authors also
exposed 1,1,1-trichloroethane in the flasks to air in the presence of
xenon arc radiation above 290 nm (which resembles sunlight) and
monitored the degradation products. They identified carbon dioxide,
hydrochloric acid, and a trace amount of chlorine. Traces of ozone,
chlorine or nitrogen dioxide, gases that are known to occur in the
atmosphere, were found to influence the product composition. Thus, if
a minor amount of chlorine was initially present during the
photo-oxidation of 1,1,1-trichloroethane, then rather more chlorine
was found at the end of the experiment.
When 1,1,1-trichloroethane enters the troposphere, it is oxidized
by reaction with the free hydroxyl radicals produced by the action of
solar UV light to form trichloroacetaldehyde, which is further
oxidized to trichloroacetic acid. The half-life for oxidation of
1,1,1-trichloroethane has been estimated to range from 2 to 5.5 years,
corresponding to residence times of 5 to 7 years (Yung et al., 1975;
McConnell & Schiff, 1978; Pearson, 1982). Slightly wider ranges of
residence times have been recorded by other authors (Prinn et al.,
1987; Fisher et al., 1990a), but the global average value generally
used is around 6 years. As suggested in section 4.1.1, calculations
indicate that 15% of 1,1,1-trichloroethane is transported to the
stratosphere. There it is degraded by photochemical processes,
induced by shorter wavelength higher energy solar radiation which does
not occur at the tropospheric level, liberating chlorine atoms:
CH3CCl3 -> CH3CCl2 + ClÊ
Free radical chlorine atoms (ClÊ) destroy ozone and are regenerated to
repeat the process.
ClÊ + O3 -> ClOÊ + O2
ClOÊ + OÊ -> ClÊ + O2
ClOÊ + NO -> ClÊ + NO2
The ozone-depleting effect of 1,1,1-trichloroethane is estimated
to be 0.11 that of the chlorofluorocarbon CFC-11 (US EPA, 1980a; UNEP,
1989).
4.2.1.2 In water
Two parallel reactions result in the degradation of
1,1,1-trichloroethane in water: (a) dehydrochlorination to
hydrochloric acid and 1,1-dichloroethene; (b) hydrolysis to
hydrochloric and ethanoic acids (see section 2.2.2) (Gerkens &
Franklin,1989). These reactions are influenced to a different degree
by temperature and alkalinity (Pearson, 1982; CEFIC, 1986; Gerkens &
Franklin, 1989). Thus the calculated half-life at 10 °C in initially
neutral demineralized water is 9.3 years and the observed half-life at
20 °C is 1.7 years (Gerkens & Franklin, 1989). The half-life of
1,1,1-trichloroethane in water at varying temperatures is summarized
in Table 4. Pearson & McConnell (1975) determined a half-life of 39
weeks at 10 °C in sea water at pH 8. These authors stated that it is
the dehydrochlorination reaction which is very pH dependent. In
artificial sea water containing salts at a concentration of
33 g/litre, the degradation rate was identical to that in
demineralized water. At 25 °C, hydrolysis under neutral conditions
was approximately 2.7 times faster than dehydrochlorination.
Half-lives at higher temperatures have been reported by Haag & Mill
(1988) and Gerkens & Franklin (1989).
Table 4. Half-life of 1,1,1-trichloroethane in water at varying
temperaturesa
Temperature (°C) Half-life Reference
10 0.75 years Pearson & McConnell (1975)
9.3 yearsb Gerkens & Franklin (1989)
20 1.7 years Gerkens & Franklin (1989)
> 2.8 years Vogel & McCarty (1987)
25 0.5 years Dilling et al. (1975)
0.8 years Gerkens & Franklin (1989)
1 year Haag & Mill (1988)
40 24 days Haag et al. (1986)
28 days Gerkens & Franklin (1989)
55 3.6 days Gerkens & Franklin (1989)
4.1 days Mabey et al. (1983)
60 1.9 days Walraevens et al. (1974)
1.9 days Gerkens & Franklin (1989)
2.2 days Haag et al. (1986)
65 0.9 days Walraevens et al. (1974)
1 day Gerkens & Franklin (1989)
80 2.8 h Archer & Stevens (1977)
4.1 h Gerkens & Franklin (1989)
5.1 h Haag & Mill (1988)
5.3 h Mabey et al. (1983)
a From: Gerkens & Franklin (1989)
b Extrapolation outside the temperature range investigated
Since the chemical degradation rates are so slow, evaporation
turns out to be the most important mechanism of loss from water
(Dilling et al., 1975). These authors found a chemical degradation
half-life in water containing 8.3 ppm oxygen at 25 °C of 6.9 months in
natural sunlight. The same half-life was found in the dark, showing
that photodegradation is negligible at the earth's surface.
4.2.2 Biodegradation
4.2.2.1 Anaerobic
In batch bacterial cultures, under methanogenic conditions at
35 °C in the dark, 1,1,1-trichloroethane at an initial concentration
of 100 µg/litre was almost completely degraded within 8 weeks, the
final concentration being 0.3 µg/litre. No degradation occurred in the
sterile controls. In a separate experiment, 1,1,1-trichloroethane
(160 µg/litre) was added to a continuous-flow methanogenic fixed-film
laboratory-scale column containing a bacterial inoculum. As with the
batch experiment, 1,1,1-trichloroethane was almost completely degraded
within 10 weeks (Bouwer & McCarty, 1983a). The degradation products of
1,1,1-trichloroethane were not identified.
In a study by Gossett (1985), 1,1,1-trichloroethane (initial
concentration, 1.13 mg/litre) was degraded to 1,1-dichloroethane,
without a lag phase, in a bath inoculated with activated sludge under
methanogenic conditions. All had been degraded within 6 days and 40%
was degraded to 1,1-dichloroethane. The fate of the other 60% is
unknown; some leakage occurred but not enough to account for the rest
of the loss.
Bouwer & McCarty (1983b) found no degradation of
1,1,1-trichloroethane, at an initial nominal concentration of 60
µg/litre, after 8 weeks of incubation under anaerobic conditions in
the presence of batch cultures of denitrifying bacteria.
Klecka et al. (1990) reported that 1,1,1-trichloroethane was
readily degraded in both methanogenic and sulfate-reducing microbial
cultures and that there was no lag period before the onset of
degradation. Transformation products included 1,1-dichloroethane,
chloroethane, and 1,1-dichloroethene. The latter was shown to be the
product of abiotic breakdown, since it also occurred in microbial
cultures poisoned by mercuric chloride.
Parsons & Lage (1985) found that 1,1,1-trichloroethane was
biodegraded under anaerobic conditions in sediment. All of the added
1,1,1-trichloroethane (4-5 µg/ml) had disappeared within 4 to 5
months, the major degradation product being 1,1-dichloroethane.
Parsons et al. (1985) reported that 16 weeks after the addition of
1,1,1-trichloroethane at a concentration of 3.6 mg/litre, 880 µg/litre
of dichloroethane had been formed.
Wilson & White (1986) found no degradation of
1,1,1-trichloroethane (added at a concentration of 765 µg/litre) in
sand columns that were continuously supplied with propane for 21 days.
Boyer et al. (1987) demonstrated microbial degradation of
1,1,1-trichloroethane, added at a level of 5 or 20 mg/litre, in a
packed bed (soil and activated carbon) laboratory reactor, which
simulated an in situ decontamination system. The authors stated
that biodegradation occurs under anaerobic conditions if a preferred
substrate such as ethanol is present. After 43 days, no
1,1,1-trichloroethane was detectable in the reactor effluent
(< 20 µg/litre). No chlorinated metabolic intermediates were
observed.
4.2.2.2 Aerobic
Wilson et al. (1983) found no aerobic degradation of
1,1,1-trichloroethane, at a concentration of 1 mg/litre, in soil
samples collected from just above and below the groundwater table.
The samples were incubated in the dark for 9 or 27 weeks.
4.3 Bioaccumulation
A bioconcentration factor of 9, comparable to that of other
chlorinated solvents, has been reported for the bluegill sunfish
(Veith et al., 1980). This is much lower than that predicted from the
measured octanol/water partition coefficient of 2.47. The same
authors also measured the loss of 1,1,1-trichloroethane from the
bluegill sunfish following exposure for 28 days to a mean water
concentration of 73 µg/litre. The half-life of 1,1,1-trichloroethane,
as measured by the loss from the tissues of half the residue
concentration attained at equilibrium, was found to be less than 24 h.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
Appraisal
As a consequence of release during its production and use,
1,1,1-trichloroethane is found in water, soil, biota, and,
especially, in air. The background levels of 1,1,1-trichloroethane in
air have increased over recent decades, although the rate of increase
seems to be declining. Levels in industrial and urban areas tend to
be higher than those in rural areas.
For the general population, air is the major source of exposure.
In some cases, indoor air may contain higher levels than outdoor air.
Under normal conditions, food and drinking-water are minor sources.
1,1,1-Trichloroethane has been found in breast milk. The use and
abuse of consumer products and cosmetics containing 1,1,1-
trichloroethane as a solvent may lead to considerable exposure.
Workers who use 1,1,1-trichloroethane, especially for degreasing
or dry-cleaning operations, are particularly exposed to this solvent,
although some exposure is likely in its manufacture. In the
workplace, air is the major source of exposure but dermal exposure
may also occur.
5.1 Environmental levels
5.1.1 Air
Rasmussen et al. (1981) monitored atmospheric concentrations of
1,1,1-trichloroethane in the Antarctic and the north Pacific coastal
region of the USA between 1975 and 1980. They found that
trichloroethane levels increased annually by over 20% during the
period 1975-1978, then dropped below 10% during the period 1978-1980
in the Antarctic and reaching 0.55 µg/m3 (102 ppt) by 1980. In the
Pacific north-west, a level of 0.84 µg/m3 (156 ppt) was reached by
1980.
It has been estimated that the annual global increase in the rate
of emission of trichloroethane was 17% between the years 1956 and 1973
but only 8% per annum between 1975 and 1980 (Khalil & Rasmussen,
1981). This decline in turn resulted in a diminution of the
differences in atmospheric levels between the northern and southern
hemispheres (Khalil & Rasmussen, 1984).
Grimsrud & Rasmussen (1975) measured mean 1,1,1-trichloroethane
concentrations of 0.54 µg/m3 (100 ppt) in the atmosphere over the
rural north-west of the USA (Washington State) between December 1974
and February 1975, and Rasmussen & Khalil (1983) found 0.947 µg/m3
(175 ppt) in the lower troposphere over the Arctic in 1982. Average
concentrations of 1,1,1-trichloroethane in air over Hokkaido, Japan,
increased slightly between 1979 and 1988 and were of the order of
0.54-0.65 µg/m3 (100-120 ppt) (personal communication by T. Tominaga
& Y. Makiide to the IPCS, 1990).
Pearson & McConnell (1975) analysed air samples from various
locations in the United Kingdom. The highest levels of
1,1,1-trichloroethane, approximately 86 µg/m3 (16 ppb by mass), were
found near an organochlorine manufacturing plant at Runcorn, Cheshire,
and levels of 33.5-59.4 µg/m3 (6.2-11 ppb by mass) were found at
Runcorn Heath. In suburban areas of the cities of Liverpool and
Manchester, the concentrations ranged from 0.54 to 32.4 µg/m3
(< 0.1 to 6 ppb by mass).
Levels of 1,1,1-trichloroethane in air are summarized in Table 5.
Fischer et al. (1982) stated that, in relatively non-polluted
areas, average concentrations of 1,1,1-trichloroethane could be
assumed to be 0.54 µg/m3 (100 ppt) and in polluted areas levels of
2.7-5.4 µg/m3 (500-1000 ppt) could be expected. They also reported
that near to large manufacturers or consumers concentrations could be
as high as 540 µg/m3 (100 000 ppt). Lillian et al. (1975) monitored
air samples for 1,1,1-trichloroethane at various locations in the USA
and found mean levels ranging from 0.52 µg/m3 (0.097 ppb) at a rural
site in Wilmington, Ohio, to 8.6 µg/m3 (1.59 ppb) in an urban area
of Bayonne, New Jersey.
Air samples collected in the North Atlantic between 1982 and
1985, in the region of the Westerlies and the North-East trade winds,
contained 1.08 µg/m3 (200 ppt). Above the trade winds (at 1800 m
above sea level) a lower concentration of 0.84 µg/m3 (155 ppt) was
found. An equivalent concentration was found at sea level in the
region of the intertropical convergence. Baseline levels of
1,1,1-trichloroethane in 1985 in the South Atlantic were about 0.76
µg/m3 (140 ppt) (Class & Ballschmiter, 1986). Rasmussen & Khalil
(1982) calculated that average concentrations of 1,1,1-trichloroethane
in 1978 were 0.632 µg/m3 (117 ppt) in the northern hemisphere and
0.486 µg/m3 (90 ppt) in the southern hemisphere.
Table 5. 1,1,1-Trichloroethane concentrations in air
Type of Locality Year Concentration Reference
location (ppt)
Industrial Runcorn, UK 1973 approx. 16 000 Pearson & McConnell (1975)
Bayonne, NJ, USA 1973 75-14 400 Lillian et al. (1975)
Urban Los Angeles, USA 1972 130-2300 Simmonds et al. (1974)
Liverpool-Manchester, UK 1973 < 100-6000 Pearson & McConnell (1975)
New York, USA 1974 100-1600 Lillian et al. (1975)
Bochum, Germany 1978 18-1610a Bauer (1981)
Lyon, France < 840-2010 Correia et al. (1977)
Denver, USA 1980 171-2699 Singh et al. (1982)
Various sites, Japan 1982-1985 20-340 Magara & Furnichi (1986)
Rural California, USA 1972 10-50 Simmonds et al. (1974)
Various sites, UK 1973 1000-4000 Pearson & McConnell (1975)
Various sites, USA 1974 30-350 Lillian et al. (1975)
Hengelo, Netherlands 20-50 Correia et al. (1977)
Oregon, USA 1975-1985 121 (1975) - 158 (1985)b Prinn et al. (1987)
Tasmania, Australia 1978-1985 66 (1978) - 116 (1985)b Prinn et al. (1987)
Hokkaido, Japan 1979-1980 100-120 Personal communication by
T. Tominaga & Y.
Makiide to the IPCS (1990)
Oceanic Hawaii, Pacific 1980-1982 111 (1980) - 125 (1982) Khalil & Rasmussen (1984)
Samoa, Pacific 1978-1982 75 (1978) - 104 (1982) Khalil & Rasmussen (1984)
Various sites, Atlantic 1982 132-214c Class & Ballschmiter (1986)
Barbados, Atlantic 1978-1985 93 (1978) - 144 (1985) Prinn et al. (1987)
Table 5 (contd).
Type of Locality Year Concentration Reference
location (ppt)
Polar Near Barrow, Arctic 1979-1982 117 (1979) - 143 (1982) Khalil & Rasmussen (1984)
South Pole, Antarctic 1975-1980 45 (1975) - 102 (1980) Khalil & Rasmussen (1984)
a The concentrations varied with wind direction and increased in the afternoon.
b These are minimum and maximum monthly mean mixing ratios and differ slightly from the original data
published by Khalil & Rasmussen (1984).
c Northern hemisphere levels were consistently higher than southern hemisphere values.
5.1.2 Water
1,1,1-Trichloroethane has been reported to occur in ground water
in many countries. Background levels tend to be low, but high levels
of contamination are possible as a result of industrial activity and
waste disposal.
Levels of 1,1,1-trichloroethane in water are summarized in
Table 6.
Relatively high levels observed in rain water at Runcorn, United
Kingdom (Pearson & McConnell, 1975), in the river Rhine, Germany
(Fischer et al., 1982), in a canal in Modena, Italy (Aggazzotti &
Predieri, 1986), in rivers that flow through industrial or large
cities in Japan (Goto, 1979), in the inshore waters of Liverpool bay,
United Kingdom (Correira et al., 1977), in ground water in Birmingham,
United Kingdom (Rivett et al., 1990), and in Maryland, USA (Dever,
1986) all appear to be derived from nearby industrial activities where
1,1,1-trichloroethane is either manufactured or used.
Background levels in snow (Pearson, 1982) and in the open ocean
(Pearson, 1982; Fischer et al., 1982) are usually very low, although
levels up to 0.97 µg/m3 (0.18 ppb) have been found by Fischer et al.
(1982) in the eastern Atlantic ocean.
5.1.3 Sediment and soil
Gossett et al. (1983) found 1,1,1-trichloroethane levels of
< 0.5 µg/kg (dry weight) in marine sediment collected from the
vicinity of a Los Angeles waste water treatment plant, the effluent
from which contained 31 µg/litre.
Fischer et al. (1982) analysed soil samples from an industrial
area of West Germany and found that 1,1,1-trichloroethane
concentrations in soil interstitial water and soil particles were near
to or less than the detection limits (0.1 µg/litre and 0.1 µg/kg,
respectively). Samples of soil air contained 1,1,1-trichloroethane
levels ranging from 0.2 to 10 µg/m3. In the same study, soil air
samples from over 1000 bore holes in various locations were analysed.
1,1,1-Trichloroethane concentrations ranged from 1 µg/m3 in a rural
area to over 2.2 µg/m3 in agricultural and forest soils near
industrial sources and to 9 µg/m3 in urban areas.
Pearson & McConnell (1975) measured a combined concentration of
1,1,1-trichloroethane and carbon tetrachloride in sediments of
Liverpool Bay, United Kingdom, of 5.5 µg/kg (5.5 ppb).
Table 6. 1,1,1-Trichloroethane levels in water
Type of water Locality Characteristics Year Concentration Reference
(ppb)
Rain water Runcorn, UK near manufacturing site 0.09 Pearson & McConnell
(1975)
Japan urban 1975 none detected Goto (1979)
Various sites rain 0.005-0.09 Pearson (1982)
Various sites snow 0.001-0.03 Pearson (1982)
Rivers & canals Rhine, Germany river 0.01-3300 Fischer et al. (1982)
Netherlands canals 1972-1976 0.07-0.3 w/w Correia et al. (1977)
R. Durance, France river 1972-1976 none detected Correia et al. (1977)
Modena, Italy canal 10-40 Aggazzotti & Predieri
(1986)
Japan river, industrial 1975 approx. = 5.1 Goto (1979)
Japan river, large city 1975 approx. = 2.5 Goto (1979)
Japan river, smaller cities 1975 approx. = 0.1-0.81 Goto (1979)
Sea Not defined inshore waters 0.15 Pearson (1982)
Liverpool Bay, UK inshore water 1972-1976 < 0.25-3.3 Correia et al. (1977)
East Atlantic open ocean 0.05-0.18 Fischer et al. (1982)
"Typical sites" open ocean 0.01-0.03 Pearson (1982);
Fischer et al. (1982)
Wells & ground
waters in
general
Zurich, Switzerland ground water 1977 0.02-2.8 Giger et al. (1978)
Japan urban, shallow 1982 0.2-1600 Magara & Furuichi
(1986)
urban, deep 1982 0.2-70
Table 6 (contd).
Type of water Locality Characteristics Year Concentration Reference
(ppb)
Wells & ground
waters in
general
Emilia-Romagna, Italy "ground water" < 1 Aggazzotti & Predieri
(1986)
Various sites underground water 0.2 (typical), 5 (high) Pearson (1982)
Various sites, UK 1983 0.48 (average) Kenrick (1983)
Birmingham, UK industrial, ground water 1986-1988 > 0.2-780 (46% of sample) Rivett et al. (1990)
Maryland, USA near electronic plant up to 1600 Dever (1986)
Water
chlorination
plant
Emilia-Romagna, Italy chlorinated water 0.1-0.5 Aggazzotti & Predieri
(1986)
5.1.4 Biota
Table 7 gives the ranges of 1,1,1-trichloroethane concentrations
found in various marine biota collected in United Kingdom estuaries
(Liverpool Bay, Firth of Forth, and Thames Estuary) by Pearson (1982)
and Pearson & McConnell (1975).
Dickson & Riley (1976) analysed various fish species and a
mollusc species from Port Erin, Isle of Man, United Kingdom, and found
trichloroethane (isomer not stated) concentrations of 2 and 9 µg/kg
dry weight, respectively, in gill and brain tissue of the eel Conger
conger. Analysis of cod (Gadus morhua) revealed trichloroethane
levels ranging from 5 µg/kg in muscle and liver tissue to 16 µg/kg in
brain tissue. Muscle tissue of coalfish (Pollachius birens)
contained 6 µg/kg, and a level of 4 µg/kg was found in the digestive
tissue of the mollusc Modiolus modiolus.
Gossett et al. (1983) collected various marine biota from the
vicinity of the Los Angeles County, USA, waste water treatment plant.
1,1,1-Trichloroethane concentrations of 4 µg/kg wet weight were found
in whole invertebrate samples and < 0.3 µg/kg wet weight in shrimp
muscle. Levels ranged from < 0.3 to 7 µg/kg wet weight in the liver
of various fish species.
5.2 General population exposure
5.2.1 Food
A study of the average daily intake of trichloroethane in Germany
showed that 10%, i.e. 3.6 µg/day, originated from foodstuffs (Duszeln
et al., 1982). Uhler & Diachenko (1987) found levels of
1,1,1-trichloroethane in nine foodstuffs (mainly cheeses and ice
cream), sampled in the USA, at levels of between 1 and 37 µg/kg.
These levels were thought to arise from contamination either by air
contact with fugitive emission of cleaning solvent or by contact with
packaging materials containing 1,1,1-trichloroethane.
Table 7. Concentration of 1,1,1-trichloroethane in various marine
organismsa
Organism Concentration (µg/kg)
Plankton 0.03-10.7
Marine algae 10-25
Molluscs 0.05-10
Crustaceans 0.7-34
Fish, flesh 0.7-5
Fish, liver 1-15
Sea-birds, eggs 3-30
Sea-birds, liver 1-4
Seal, liver 0.2-4
Seal, blubber 8-24
a From: Pearson (1982)
Entz & Diachenko (1988) surveyed 52 margarines and spread
products from supermarkets in the Washington, D.C. metropolitan area
between 1980 and 1982 and a further 18 products in 1984. In addition,
19 margarines were examined at manufacturing plants in 1982. The
following levels were found:
Number of store Number of manufacturing
shelf samples plant samples
Not detecteda 11 1
Trace levelsa 12 1
< 50 µg/kg 35 15
50-100 µg/kg 7 2
> 100-500 µg/kg 5 -
a Trace levels of 1,1,1-trichloroethane represented < 5 µg/kg; the
detection limit was about a third of the trace level.
Miller & Uhler (1988) similarly studied 46 butter samples and
reported levels of from 10 µg/kg to more than 100 µg/kg. In one
sample containing 7500 µg/kg, the source was traced to a packing
adhesive.
Pfannhauser et al. (1988) found that levels of
1,1,1-trichloroethane were mostly below 10 µg/kg in samples of
Austrian olive oil, cheese, and chocolate; only one sample of olive
oil contained over 100 µg/kg. The authors suggested that cleaning
solvents in production areas and packaging materials were possible
sources for the contamination.
Pellizari et al. (1982) analysed breast milk for
1,1,1-trichloroethane in four urban areas of Pennsylvania, New Jersey,
and Los Angeles, USA. They sampled up to 12 women at each site, and
eight samples out of 42 were analysed manually by experienced
spectroscopists. 1,1,1-Trichloroethane was identified in all of the
samples, but no actual levels were reported. Travis et al. (1988)
also suggested contamination of human breast milk based on
pharmacokinetic modelling.
Table 8 summarizes data on the concentrations of
1,1,1-trichloroethane reported in foods.
5.2.2 Drinking-water
An investigation of drinking-water from 100 cities in Germany
showed a range of trichloroethane concentrations from < 0.1 to 1.7
µg/litre (Bauer, 1981).
A study of the daily average intake of trichloroethane in Germany
revealed that 0.6%, i.e. 0.2 µg/day, of the intake came from
drinking-water. These calculations were based on a daily intake of 1
litre of drinking-water containing 0.2 µg trichloroethane/litre of
water (Düszeln et al., 1982).
In the USA, 23 wells out of 1611 tested contained
1,1,1-trichloroethane. Another investigation in the USA showed
detectable concentrations in 835 of 1071 samples, with a maximum value
of 607.8 µg/litre and a 90th percentile of 6.1 µg/litre (Fischer et
al., 1982).
Pearson (1982) reported a value of 0.1 µg/litre as a typical
level of 1,1,1-trichloroethane in drinking-water. Fielding et al.
(1981) surveyed 14 drinking-water sources in the United Kingdom over
a period of 9 months in 1976. 1,1,1-Trichloroethane was found at 3
out of 14 sites and, although no actual levels were reported, it was
implied that concentrations were less than or equal to 1 µg/litre.
Dever (1986) reported levels of 1,1,1-trichloroethane in a
contaminated water supply in Montgomery County, Maryland, USA. Both
raw and treated potable water samples were analysed, and
1,1,1-trichloroethane levels of 180 µg/litre and 30 µg/litre,
respectively, were found.
Table 8. Concentrations of 1,1,1-trichloroethane (µg/kg) in various foodstuffsa
Foodstuff Mean content Range Reference
Dairy products 0.1-10 Pearson (1982)
0-0.6 Bauer (1981)
Meat 3-6 McConnell et al. (1975)
Pearson (1982)
Vegetable oils 0.5-10 Pearson (1982)
< 1 - > 100 Pfannhauser et al. (1988)
Margarine 45 n.d. - 500 Entz et al. (1982)
Entz & Diachenko (1988)
Butter 16 10-7500 Entz et al. (1982)
Miller & Uhler (1988)
Ice cream 2 Entz et al. (1982)
Cheese 7-9 Entz et al. (1982)
< 1 - 100 Pfannhauser et al. (1988)
Bread 2 McConnell et al. (1975)
Pearson (1982)
Potatoes 1-4 McConnell et al. (1975)
Fruit and vegetables 2-3 McConnell et al. (1975)
Pearson (1982)
Fish flesh 0.7-5 McConnell et al. (1975)
Pearson (1982)
Table 8 (contd).
Foodstuff Mean content Range Reference
Fish (cod) liver,
muscle, stomach 5-7b Dickson & Riley (1976)
Tea (packet) 7 McConnell et al. (1975)
Rolled oats 770c Daft (1988)
Popcorn 5 Daft (1988)
Pinto beans 5 Daft (1988)
Chocolate products < 1 - 100 Pfannhauser et al. (1988)
Fatty foods <1 - 10 Pfannhauser et al. (1988)
a Fresh (wet) weight, unless specified otherwise; n.d. = not detected
b Dry weight
c Possibly from fumigant contamination
Wallace et al. (1987) measured low levels of
1,1,1-trichloroethane in drinking-water collected in 3 states in the
USA during 1981/1982. The average levels in New Jersey were 0.2-0.6
ng/litre (maximum levels were 1.6-5.3 ng/litre), and in North Carolina
and North Dakota they were lower still, i.e. 0.03 ng/litre and 0.04
ng/litre, respectively.
An environmental monitoring study carried out near
1,1,1-trichloroethane production plants and user facilities in
1976/1977 yielded the following 1,1,1-trichloroethane levels in hotel
tap water (US EPA, 1977):
Freeport, Texas (industrial area): 17 µg/litre; Lake Charles,
Louisiana (industrial area): 0.3 µg/litre; Helena, Arkansas (rural
area): 0.4 µg/litre.
Concentrations of 1,1,1-trichloroethane in Japanese tap water
(Osaka City) were reported to range from 0.078 to 0.212 µg/litre
(Kajino & Yagi, 1980).
5.2.3 Air
In relatively non-polluted areas, the average concentration of
1,1,1-trichloroethane in air can be assumed to be around 540 ng/m3
(100 ppt), increasing to 2700-5400 ng/m3 (500-1000 ppt) in
industrialized areas (Bouwer & McCarty, 1983a,b). In Germany, the
average air concentration was found to be about 2 µg/m3 (Düszeln &
Thiemann, 1985).
In a monitoring study conducted in 1981 on 350 residents of New
Jersey, USA, Wallace et al. (1986) found median indoor air
concentrations of 17 µg/m3 both at night and during the day.
Concentration ranged from 0.16 to 333 000 µg/m3. The median outdoor
air level was 4.5 µg/m3, the range being 0.05 to 470 µg/m3.
Analyses conducted on the breath of these individuals showed median
levels of 6.6 µg/m3 (ranging from 0.06 to 520 µg/m3).
A study of the average daily intake of trichloroethane in Germany
estimated that 89%, i.e. 32 µg of the daily intake of 35.8 µg, came
from the air. This calculation was based on a daily inhalation of 20
m3 air containing 1.6 µg trichloroethane/m3 (300 ppt) (Düszeln et
al., 1982).
In Japan, 15 sites of Yokohama City and Kawasaki City were
sampled from July 1985 to July 1986. Mean (of 5) concentrations of
14.6, 12.4, and 8.6 µg/m3 were measured in industrial, commercial,
and residential areas, respectively (Urano et al., 1988). The
calculated average intake (µg/day) by inhalation in each area was 135,
114, and 81, respectively.
It can be concluded that, in general, inhalation is the most
important source of human exposure to trichloroethane.
5.2.4 Consumer products and cosmetics
Trichloroethane is used as a solvent in aerosol and non-aerosol
consumer products, and the concentration may be anywhere in the range
10-100% (IARC, 1979). In aerosol cans for cosmetics, a concentration
of up to 35% is allowed in countries of the Economic European
Community (EEC). Consumer use and abuse of such products may lead to
considerable exposure to trichloroethane, which can be much higher
than from other sources.
In a study by Otson et al. (1984), two fabric protectors (450 g
of each) containing 75% and 97% 1,1,1-trichloroethane, respectively,
were sprayed onto a sofa in a room with a volume of 28 m3. Initial
concentrations were as high as 1800 mg/m3. Concentrations dropped
rapidly to less than 150 mg/m3 when the room was ventilated, but
under unventilated conditions they remained above 1000 mg/m3 for at
least one hour and dropped below 500 mg/m3 only after more than 2 h.
5.3 Occupational exposure
In general, exposure levels of trichloroethane at the workplace
are much lower than established limit values for workplace air. The
8-h time-weighted average (TWA) concentrations measured in the United
Kingdom in vapour-degreasing baths were typically 10.8-270 mg/m3
(2-50 ppm) (HSE, 1990).
In addition to workers engaged in the manufacturing and
production of trichloroethane and trichloroethane-containing products,
the workers most likely to be exposed to trichloroethane are those
engaged in dry-cleaning or degreasing processes in the metallic and
electronic industries. It was estimated that about 2.23 million
workers were potentially exposed to trichloroethane in the USA (NIOSH
Survey, 1983).
During the period 1983 to 1986, the Danish Labour Inspection
Service made 476 measurements of trichloroethane at workplaces in
Denmark (AMI, 1988). In 6% of the samples the levels were found to
exceed the Occupational Limit Value of 540 mg/m3 (100 ppm).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
Appraisal
1,1,1-Trichloroethane is rapidly absorbed through the lungs.
Absorption through the gastrointestinal tract and skin also occurs,
but is of less importance than the inhalation route. In humans, the
absorption from the lungs is about 25 to 40% of the inhaled dose over
6 to 8 h. 1,1,1-Trichloroethane is distributed widely to body
tissues, especially to those with a high lipid content, e.g., brain
and adipose tissue. It crosses the blood-brain and placental
barriers. Less than 7% of an absorbed dose is metabolized.
Metabolism appears to be saturable. Excretion by exhalation of
unchanged compound accounts for > 90% of the absorbed dose. Less
than 1% of 1,1,1-trichloro-ethane remains in the human body after 9
days.
6.1 Absorption
1,1,1-Trichloroethane is rapidly absorbed through the lungs and
the gastrointestinal tract. Absorption through skin also occurs, but
is of minor importance compared to uptake via inhalation.
6.1.1 Animal studies
6.1.1.1 Inhalation
The absorption in the lungs of rats exposed to 270 or 2700 mg
1,1,1-trichloroethane/m3 (50 or 500 ppm) has been shown to be
time-dependent. The initial uptake was more than 80% of the dose.
During the first hour the absorption decreased to around 50%, the
decrease being greater in the high-dose group (Dallas et al., 1989).
Because 1,1,1-trichloroethane is poorly metabolized (see section 6.3),
absorption is expected to be low as steady state is approached.
In dogs exposed to 4.05, 8.1 or 10.8 g/m3 (750, 1500 or 2000
ppm) for one hour by inhalation, the cumulative uptake was 27, 45, and
71 mg/kg, respectively. This represented about 14% of inhaled
amounts. Concentrations in arterial and venous blood did not attain
a steady state during this period (Hobara et al., 1982).
6.1.1.2 Oral absorption
Oral toxicity data (see section 7.1, Table 10) suggest that
1,1,1-trichloroethane is readily taken up from the gastrointestinal
tract (Reitz et al., 1988).
6.1.1.3 Skin absorption
The percutaneous penetration of 1,1,1-trichloroethane was studied
in guinea-pigs by applying the solvent in a skin depot (a glass ring
attached to the skin and covered with glass). Absorption was found
to be rapid and to result in blood levels of 1.9 mg/litre during the
first 30 min. These levels were higher than those found for carbon
tetrachloride and perchloroethylene (1.1 mg/litre in both cases) in
the same study (Jakobson et al., 1980, 1982). HSE (1984) calculated
a rate of absorption through the skin of 6 µg/min per cm2 from this
study.
Compared to methylene dichloride, chloroform, carbon
tetrachloride, and 1,1,2-trichloroethane, the skin penetration of
1,1,1-trichloroethane in mice was found to be lower. The penetration
rate of these solvents increased with the degree of water solubility
(Tsuruta, 1975).
6.1.2 Human studies
The absorption of trichloroethane in humans has been studied
following exposure by inhalation and by skin contact. No studies have
so far been carried out using the oral route, but reports of
intoxication following oral ingestion indicate that the chemical can
be absorbed by this route (Stewart & Andrews, 1966, 1971).
6.1.2.1 Inhalation
The absorption of 1,1,1-trichloroethane in the lungs is lower
than that of most other chlorinated solvents. This is due to the
relatively low blood/air partition coefficient of 3.3 (US EPA, 1984).
Studies with human volunteers (males), exposed by inhalation to
1,1,1-trichloroethane at concentrations from around 200 mg/m3 (35
ppm) to 2000 mg/m3 (350 ppm) for 6-8 h, showed that about 25-40% of
the trichloroethane inhaled was absorbed by the lungs, depending on
its concentration in the inhaled air, duration of exposure, body
weight and amount of adipose tissue, blood circulation, and other
factors (Astrand et al., 1973; Humbert & Fernandez 1977; Monster et
al., 1979; Nolan et al., 1984). The amount absorbed increased with
increasing work-load, due presumably to an increase in breathing rate
(Monster et al., 1979).
6.1.2.2 Skin contact
Percutaneous absorption of 1,1,1-trichloroethane has been
measured in human subjects. Following the immersion for a few minutes
of a thumb or a hand in liquid 1,1,1-trichloroethane, it is possible
to detect the solvent in the breath (Stewart & Dodd, 1964).
The absorption of trichloroethane through the skin is slower than
for aromatic solvents and perchloroethylene, and is, in general,
considered of minor importance compared to the more rapid uptake via
the lungs (Humbert & Fernandez, 1977). Dermal application to human
volunteers of 15 ml 1,1,1-trichloroethane under occlusion resulted in
16.2-27 mg/m3 (3-5 ppm) of the solvent in the exhaled air. This
corresponded to a 2-h inhalation exposure of 54-108 mg/m3 (10-20
ppm) (Nakaaki et al., 1980).
Trichloroethane vapour can also be absorbed through the intact
skin, but the amount absorbed in the body has been estimated to be a
thousand times less than the amount absorbed by inhalation (Riihimaki
& Pfaffli, 1978).
6.2 Distribution and retention
Compared with many other chlorinated hydrocarbon solvents,
1,1,1-trichloroethane has a high lipid/blood partition coefficient
(108 at 37 °C). It would therefore be expected to distribute widely
into body tissues, especially into those, such as brain and adipose
tissue, with high lipid content (US EPA, 1984; CEC, 1986).
Blood and exhaled breath concentrations of 1,1,1-trichloroethane
in rats increased rapidly after inhalation exposure, approaching, but
not reaching, steady state after a 2-h exposure (Dallas et al., 1989).
6.2.1 Animal studies
When rats were exposed to 2700 mg/m3 (500 ppm) (6 h per day for
4 days), only trace amounts of 1,1,1-trichloroethane could be detected
in the liver, brain, and blood 17 h after the end of the exposure
period. A higher concentration remained in adipose tissue, but this
represented only about 5% of the concentration immediately after
exposure (Savolainen et al., 1977). These data indicate that
1,1,1-trichloroethane does not accumulate in the tissues. This is
supported by the observation that no significant tissue accumulation
occurred in rats exposed to 8100 mg/m3 (1500 ppm), 6 h per day, 5
days per week, for 16 months (Schumann et al., 1982).
In a study by Danielsson et al. (1986), pregnant mice were
exposed to 14C-labelled 1,1,1-trichloroethane by inhalation for 10
min on days 11, 14 or 17 of gestation. Radioactivity was detected in
the brain, lungs, liver, and kidney of the maternal mouse immediately
after exposure, the concentrations being approximately the same in all
tissues. The fetal and placental uptake was measured at all stages of
gestation studied. Although the fetal uptake was low compared with
uptake to the maternal brain, the authors stated that
1,1,1-trichloroethane passes as easily through the placental barrier
as through the blood-brain barrier. After exposure,
1,1,1-trichloroethane disappeared from both maternal and fetal bodies
within 24 h. A small amount of non-volatile radioactivity was present
in both the mother and fetus.
Concentrations of 1,1,1-trichloroethane in exhaled air from dogs
exposed to 3780, 8100 or 10 800 mg/m3 (700, 1500 or 2000 ppm)
increased rapidly for one hour, approaching steady-state levels at 80
to 90% of inhaled air concentrations. After one hour's recovery, 66
to 71% of the total uptake had been excreted through the lungs (Hobara
et al., 1982).
6.2.2 Human studies
In a fatal case of 1,1,1-trichloroethane intoxication, residues
of the solvent were determined in the bile, blood, brain, kidney,
liver, and lung. The concentration was highest in the brain, followed
by the kidney (Caplan et al., 1976). This indicates that
1,1,1-trichloroethane can also cross the blood-brain barrier in
humans.
Samples of kidney, lung, and muscle tissues taken from hospital
patients in Finland contained small amounts of 1,1,1-trichloroethane
(0.1 to 0.4 µg/kg) (Kroneld, 1989). In human samples from the German
Ruhr district, levels of 1.8 to 5.6 µg/kg (fresh weight) were found in
the same tissues. In addition, 2.1 µg/kg was found in fat tissue and
1.9 µg/kg in liver tissue (Bauer, 1981).
6.3 Metabolic transformation
1,1,1-Trichloroethane is a fairly stable molecule, which is
metabolized in mammals to a lesser degree than other trichlorinated
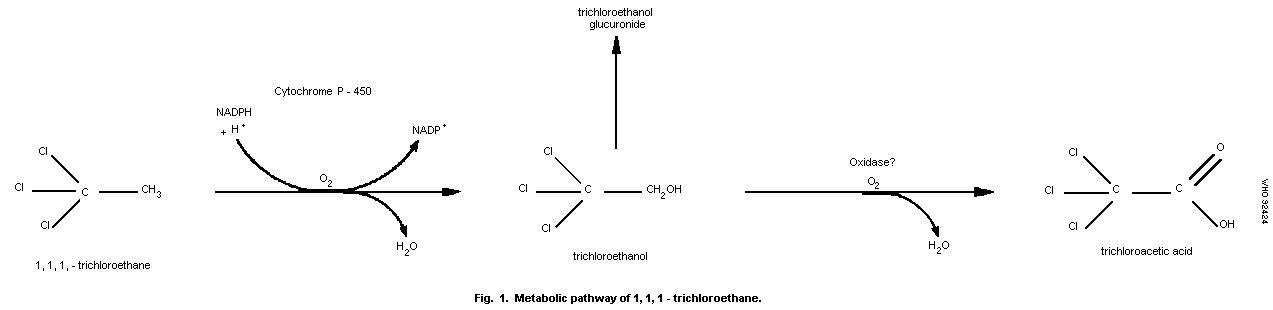
solvents (Ikeda & Ohtsuji, 1972). As shown in Fig. 1, the principal
metabolites are 2,2,2-trichloroethanol and trichloroacetic acid
(Humbert & Fernandez, 1977). These metabolites are formed in the
liver by microsomal oxidases (cytochrome P-450) (Ivanetich & Honert,
1981; US EPA, 1984). Trichloroethanol is conjugated with glucuronic
acid before excretion in the urine.
6.3.1 Animal studies
In an early study in rats, less than 3% of a single
intraperitoneal injected dose of about 700 mg 1,1,1-trichloroethane/kg
body weight was metabolized within 25 h; the rest was expired
unchanged. The metabolites identified, accounting for 1.6% of the
dose, were the glucuronide of 2,2,2-trichloroethanol in the urine
(53%) and 14CO2 in the expired air (30%) (Hake et al., 1960).
When 1,1,1-trichloroethane (143 mg/kg) was administered in the
drinking-water to rats over an 8-h period, the percentage of the dose
recovered as metabolites within 56 h was 6%, of which 37% was excreted
in the urine and 37% as 14CO2 in the expired air (Reitz et al.,
1988). Following repeated administration by gavage for 4 weeks, the
percentage of the administered dose recovered as metabolites in male
rats within 48 h was 4.2%. Under similar experimental conditions in
mice, 6.1% of the administrated dose was recovered as metabolites
(Mitoma et al., 1985). A single inhalation exposure for 4 h at a
concentration of 1188 or 2376 mg/m3 (220 or 440 ppm) resulted in,
respectively, urine metabolite concentrations of 0.58 and 0.97 mg/kg
 body weight. Both trichloroethanol and trichloroacetic acid were
identified as metabolites in the urine, trichloroethanol being
excreted (as the glucuronide) much faster than trichloroacetic acid
(Eben & Kimberle 1974).
Following a single inhalation exposure of rats to 810 and
8100 mg/m3 (150 and 1500 ppm), only a 2-4 times increase in excreted
metabolites was found between the two doses, suggesting metabolic
saturation. Repeated exposure to 8100 mg/m3 over 16 months did not
change the amount of 1,1,1-trichloroethane metabolites. Similar
results were obtained in mice. In both rats and mice, urine
metabolites accounted for 40-70% of the total amount metabolized.
Overall, mice were found to biotransform approximately 5 times more
1,1,1-trichloroethane (per kg body weight) than rats. In rats and
mice an age-related increase in the amount metabolized was observed in
aged animals as opposed to young adults (Schumann et al., 1982).
6.3.2 Human studies
The average amount of metabolites excreted in the urine of humans
(workers or volunteers) exposed to 1,1,1-trichloroethane in the air
(22-1890 mg/m3, 4-350 ppm) was variable, but was typically between
3 and 7% of the absorbed dose. The ratio of the metabolites
trichloroethanol glucuronide and trichloroacetic acid was about 2 to
1, but this increased with increasing exposure concentration (Seki et
al., 1975; Humbert & Fernandez, 1977; Monster et al., 1979; Nolan et
al., 1984).
6.3.3 Metabolic interactions
Simultaneous exposure to other solvents tends to increase the
retention and decrease the metabolism of 1,1,1-trichloroethane
(Savolainen et al., 1981). 1,1,1-Trichloroethane metabolism was
accelerated in rats pre-treated with ethanol because of the induction
of metabolic enzymes (Sato et al., 1980).
6.4 Elimination
Regardless of the route of administration, the main excretory
route for 1,1,1-trichloroethane is exhalation via the lungs. This may
be explained by the relatively low solubility in blood.
6.4.1 Animal studies
In rats and mice, 55% to 98% of the 1,1,1-trichloroethane was
excreted unchanged in expired air after oral or intraperitoneal
exposure (Hake et al., 1960; Reitz et al., 1988).
Following inhalation exposure, 94-98% of the absorbed
1,1,1-trichloroethane was excreted unchanged in the expired air of
rats during 72 h. In mice, the corresponding figure was 87-97%. The
rate of elimination was somewhat higher in mice; 85% was eliminated
during the first 3 h compared to 65% in rats (Schumann et al., 1982).
A total of about 1-8% of an absorbed dose in rodents is excreted
in the urine (Hake et al., 1960; Schumann et al., 1982).
After exposure to 1,1,1-trichloroethane vapour, the level in
blood plasma decreases rapidly in a diphasic or triphasic manner,
depending on the exposure level. Following exposure to 810 mg/m3
(150 ppm), the half-lives were 10 and 139 min, whereas with 8100
mg/m3 (1500 ppm), the half-lives were 36 and 238 min. In mice the
half-lives were a little shorter (Schumann et al., 1982).
6.4.2 Human studies
Studies with human volunteers show that over 90% of the absorbed
trichloroethane is excreted unchanged in the expired air. Only minor
parts (5-7%) of the absorbed solvent are excreted in the urine (as
trichloroethanol glucuronide and trichloroacetic acid) (Stewart et
al., 1969; Humbert & Fernadez, 1977; Nolan et al., 1984). The main
product was found to be 2,2,2-trichloroethanol glucuronide, the
excretion of which was completed within 8 days. The secondary product
was trichloroacetic acid; its excretion occurred somewhat later and
was completed within 12 days (Humbert & Fernandez, 1977).
The elimination of 1,1,1-trichloroethane, measured as plasma
concentration and concentration in expired air, in six human
volunteers exposed to 191 or 1911 mg/m3 (35 or 350 ppm) for 6 h
could be described by a three-compartment model with estimated
half-lives of 44 min, 5.7 h, and 53 h, respectively. Less than 1%
remained in the body after 9 days (Nolan et al., 1984).
In a study by Nolan et al. (1984), the trichloroethane
concentrations in blood and expired air were proportional to the
exposure concentration after 6 h of exposure and indicated that about
25% of the 1,1,1-trichloroethane inhaled during the exposure was
absorbed.
6.5 Biological monitoring
Several options exist for the biological monitoring of exposure
to 1,1,1-trichloroethane (Monster 1986). These include the
determination of:
Ê the unchanged solvent in blood or alveolar air;
Ê the metabolite trichloroethanol in blood, alveolar air or urine;
Ê the metabolite trichloroacetic acid in blood and urine.
Table 9 indicates the mean concentrations of these parameters as
a result of exposure of subjects for 8 h/day (5 days/week) to a
time-weighted average (TWA) concentration of 270 mg/m3 (50 ppm)
(Monster, 1986).
Table 9. Mean concentrations of 1,1,1-trichloroethane,
trichloroethanol, and trichloroacetic acid in blood,
alveolar air, and urinea
Time after exposure of sampling
Test End of 5-15 min 16 h 64 h
exposure
Blood (mg/litre)
1,1,1-trichloroethane 0.9 0.07
trichloroethanol 0.16 -
trichloroacetic acid 2.3 1.6
Alveolar air (mg/m3)
1,1,1-trichloroethaneb 210 (39) 13 (2.4) 8 (1.5)
trichloroethanol 0.014 0.007 -
Urine (mg/g creatinine)
trichloroacetic acid 4.9 2.5 0.9
trichloroethanol 2.5 1.8 1.5
a From: Monster (1986)
b Values in parentheses are in ppm
Trichloroethane concentrations in blood measured after work on a
Friday seem to be the best single parameter for estimation of the
TWA-week exposure (Monster, 1986). For one-day exposure, the
trichloroethane level in blood and urine is most useful (Monster,
1986).
Droz et al. (1989) suggested a physiological model for describing
variability in the biological monitoring of solvent exposure.
Standard statistical distributions are used to simulate variability in
exposure concentration, physical workload, body fluid, liver function,
and renal clearance. For groups of workers exposed daily, the model
calculates air monitoring indicators and biological monitoring
results, including levels in expired air, blood, and urine. The
calculated results obtained are discussed and compared with measured
data for physiological and toxicokinetic parameters for six solvents
including 1,1,1-trichloroethane and their metabolites. It is
suggested that such mathematical models are applicable for prediction
and management studies.
6.6 Bioaccumulation
Inhalation exposure of rats to 2700 mg/m3 (500 ppm) for 4 days,
6 h per day, led to an accumulation of 1,1,1-trichloroethane in the
fat 17 h after the last exposure. Further exposure on the 5th day
increased brain, liver, lung, and blood levels (Savolainen et al.,
1981).
A study by Travis et al. (1988) indicated that bioaccumulation of
organic chemicals, including 1,1,1-trichloroethane, perchloroethane,
trichloroethylene, and dichloromethane, in human adipose tissues was
positively correlated with their octanol-water partition coefficients.
The information presented, however, was based on data from a human
pharmacokinetic model.
7. EFFECTS ON LABORATORY MAMMALS AND IN VITRO TEST SYSTEMS
Appraisal
1,1,1-Trichloroethane administered by various routes has low
acute toxicity for laboratory animals. The acute toxicity pattern is
characterised by central nervous system depression and respiratory
arrest at lethal levels. 1,1,1-Trichloroethane is a moderate skin
and mild eye irritant.
Short-term inhalation exposure of rats to 4320-5400 mg/m3
(800-1000 ppm) or more produced an increase in liver weight. At
similar concentrations in mice, the liver changes were more marked
and included fatty infiltration and single cell necrosis. Minor
cytoplasmic alterations were seen in mice exposed to 1350 mg/m3
(250 ppm). Studies on Mongolian gerbils using 1,1,1-trichloroethane
concentrations down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several regions of
the brain. The significance of these findings is uncertain. The
no-observed-effect level (NOEL) for rats is about 2700 mg/m3 (500
ppm).
In long-term toxicity studies, 1,1,1-trichloroethane
administration led to reduced weight gain in both rats and mice.
Slight microscopic hepatic effects were seen, but only at the highest
concentration of 8100 mg/m3 (1500 ppm). There was no evidence of
carcinogenic potential in oral and inhalation studies on rats and
mice. One study reported increased incidence of leukaemia but this
study is considered inadequate.
1,1,1-Trichloroethane has been shown to have low genotoxic
potential in a range of in vitro and in vivo studies.
In a multigeneration study on mice, there was no evidence of
adverse reproductive effects (including effects on fertility) caused
by 1,1,1-trichloroethane in drinking-water at concentrations up to
1000 mg/kg body weight. Inhalation studies on female rats and mice
at 4590 mg/m3 (850 ppm) showed no evidence of teratogenic or
fetotoxic effects. However, in rats exposed to 11 340 mg/m3 (2100
ppm), there was some evidence of fetotoxicity.
The data available are inadequate to assess the immunotoxic
potential of 1,1,1-trichloroethane.
Enhanced toxicity of 1,1,1-trichloroethane was observed when
exposure was combined with exposure to ethanol.
7.1 Acute toxicity
1,1,1-Trichloroethane has a very low acute toxicity in laboratory
animals dosed by various routes of administration. Selected LD50
and LC50 values are given in Tables 10 and 11.
The toxicological pattern of acute trichloroethane poisoning is
central nervous system (CNS) depression, eventually culminating in
respiratory arrest or cardiac failure at lethal exposure levels
(Stewart, 1968).
The findings of rapid and shallow breathing in a dog following
inhalation of 0.9% (by volume) 1,1,1-trichloroethane (corresponding to
48 600 mg/m3 or 9000 ppm) may be related to an increase in the
activity of the lung stretch receptor in the vagus nerve (Kobayashi et
al., 1986). A level of 1.3% increased the heart rate but 2.8%
decreased it (Kobayashi et al., 1987).
Non-lethal acute toxicity has also been investigated (see Table
12). In a study on rats, 50% of the experimental group showed loss of
coordination (ataxia) after inhalation of 20 400 mg/m3 (3780 ppm)
for 4 h, and loss of righting-reflex was observed at 45 800 mg/m3
(8480 ppm). Tremors and death were observed at 64 800 mg/m3 (12 000
ppm) within one hour of exposure (Mullin & Krivanek, 1982).
Cardiac sensitization to adrenaline was reported in dogs exposed
to 37 800 mg/m3 (range: 21 600 to 59 400 mg/m3) for 5 min (Clark
& Tinston, 1982). An increase in concentration (range: 37 300 to 373
000 mg/m3) and inhalation period (0.5 to 20 min) was accompanied by
a decrease in the amount of adrenaline that induced arrhythmia
(Kobayashi et al., 1982).
Reinhardt et al. (1973) reported that 1,1,1-trichloroethane
caused cardiac sensitization to adrenaline in dogs at and above the
0.5% (v/v) level. The marked response was associated with ventricular
fibrillation following the challenge dose of adrenaline. At this
level, the solvent was also associated with excitement and struggling
in the animals. Histopathological examination of samples from the
dogs that developed fatal arrhythmias did not show any gross or
microscopic abnormalities.
Table 10. Acute toxicity (LD50) of 1,1,1-trichloroethane in experimental animals
Species Sex Route LD50 (mg/kg Reference
body weight)
Rat male oral 14 300 Torkelson et al. (1958)
Rat female oral 11 000 Torkelson et al. (1958)
Mouse female oral 9700 Torkelson et al. (1958)
Guinea-pig male & female oral 8600 Torkelson et al. (1958)
Rabbit male & female oral 10 500 Torkelson et al. (1958)
Rabbit male & female skin 15 800 Torkelson et al. (1958)
Mouse female intraperitoneal 3700 Gradiski et al. (1974)
Mouse male intraperitoneal 5080 Klaassen & Plaa (1966)
Dog male intraperitoneal 4140 Klaassen & Plaa (1967)
Table 11. Acute toxicity by inhalation (LC50) of 1,1,1-trichloroethane
in experimental animals
Species Sex LC50 Exposure Reference
(g/m3) (ppm) duration
Rat male & female 97.2 18 000 3 h Adams et al. (1950)
Rat male & female 76.9 14 250 7 h Adams et al. (1950)
Rat male 99.4 18 400 4 h Siegel et al. (1971)
Rat male 55.6 10 300 6 h Bonnet et al. (1981)
Rat male & female 205 38 000 15 min Clark & Tinston (1982)
Mouse male 120 22 240 30 min Woolverton & Balster (1981)
Mouse female 72.4 13 410 6 h Gradiski et al. (1978)
Mouse male 21.1 3910 2 h Horiguchi & Horiguchi (1971)
Mouse male 99.1 18 358 1 h Moser & Balster (1985)
Mouse male 111 20 616 30 min Moser & Balster (1985)
Mouse male 159 29 492 10 min Moser & Balster (1985)
Table 12. Non-lethal acute toxicity in experimental animals
exposed to 1,1,1-trichloroethane by inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 45 800 4 h loss of righting Mullin & Krivanek (1982)
reflex (EC50)
Rat 20 400 4 h loss of coordination Mullin & Krivanek (1982)
(EC50)
Mouse 31 000 1 h inverted screen test Moser & Balster (1985)
performance (EC50)
Dog 37 800 5 min cardiac sensitization Clark & Tinston (1982)
to adrenaline
Dog 21 600 a few min decrease in blood Kobayashi et al. (1983)
pressure
The effect of 1,1,1-trichloroethane on liver enzyme activity in
the serum of experimental animals has been investigated. An almost
lethal intraperitoneal dose of 1,1,1-trichloroethane (0.87 ml/kg body
weight) caused a significant elevation of ALAT (alanine
aminotransferase) in dogs (Klaassen & Plaa, 1967), and one eighth of
a lethal intraperitoneal dose caused a significant elevation in SDH
(sorbitol dehydrogenase) (Lundberg et al., 1986). In both studies,
the hepatotoxicity of 1,1,1-trichloroethane was relatively small
compared to that of other organic solvents, e.g., chloroform, carbon
tetrachloride, dimethylformamide (Lundberg et al., 1986),
dichloromethane, 1,1,2-trichloroethane, and trichloroethylene
(Klaassen & Plaa, 1967).
7.1.1 Irritation
Trichloroethane has been shown to produce "mild" skin irritation
in rabbits when repeatedly applied topically under an occlusive
dressing. Redness was noted and the skin became scaly, but the effect
was transient, the skin rapidly returned to normal (Torkelson et al.,
1958).
After guinea-pigs had been exposed to 1 ml of
1,1,1-trichloroethane via a skin depot (a glass ring attached to the
skin and covered with glass), for 15 min, oedema was observed.
Prolonged exposure for several hours led to more severe inflammatory
reactions in the upper part of the dermis, and histological
examination at 15 min and 1, 4, and 16 h showed a number of changes to
the epidermis. The extent of these changes increased with duration of
exposure (Kronevi et al., 1981).
Repeated topical application of trichloroethane to abraded and
non-abraded rabbit skin for up to 90 days resulted in slight,
reversible irritation (Torkelson et al., 1958). The same study showed
that when trichloroethane was applied soaked in a cotton wool pad and
bandaged to the shaven belly of a rabbit slight reddening and
scaliness occurred, but this only increased slightly with repeated
applications.
The application of 0.5 ml trichloroethane to the shaven skin of
rabbits under an occlusive dressing for 24 h, resulted in moderate
skin irritation (Duprat et al., 1976). In a recent study of OECD
methods (4-h exposure under semi-occlusive dressing) trichloro-ethane
was reported to be a skin irritant (van Beek, 1990).
Trichloroethane is a "mild" eye irritant. Slight to moderate
pain and slight conjunctival irritation, but no corneal damage, was
reported following a single application of 100 µl trichloroethane to
the eyes of rabbits (Torkelson et al., 1958). Duprat et al. (1976)
also found that instillation of 0.1 ml trichloroethane to the eyes of
rabbits produced slight irritation.
7.1.2 Short-term exposure
7.1.2.1 Inhalation
When groups of rats, guinea-pigs, rabbits, dogs, and squirrel
monkeys were exposed to 1,1,1-trichloroethane (11 880 mg/m3, 2200
ppm) for 8 h/day, 5 days/week for 6 weeks, the only sign of toxicity
was reduced body weight gain in rabbits and dogs. There were no
effects on haematological parameters or serum urea nitrogen and no
histopathological changes were observed (Prendergast et al., 1967).
In a study by Adams et al. (1950), rats were exposed to 0 or 27
000 mg/m3 (0 or 5000 ppm) for 7 h/day on 32 out of 45 days and
guinea-pigs received 20 to 65 7-h exposures to 0, 3510, 8100, 16 200
or 27 000 mg/m3 (0, 650, 1500, 3000 or 5000 ppm) for a period of 1-3
months. Body weight gain was reduced in both species at all exposure
levels used, but there were no other signs of toxicity and no effects
were observed on blood urea nitrogen. The only 1,1,1-trichloroethane-
related effect observed on histopathological examination was fatty
degeneration, without necrosis, in the liver of guinea-pigs at 16 200
and 27 000 mg/m3.
Rats exposed to 2700 mg/m3 (500 ppm), 6 h/day for 5 days,
showed no behavioural effects. However, there was a slight decrease
in brain RNA content relative to controls (Savolainen et al., 1977).
Inhalation of 1750 mg/m3 (320 ppm) for 30 days had no effects on the
composition of brain lipids in rats (Kyrklund et al., 1988).
When rats were given 4320 mg/m3 (800 ppm) by inhalation 6
h/day, 5 days/week for 4 weeks, absolute and relative liver weights
were increased but there was no induction of liver microsomal
cytochrome P-450 (Toftgaard et al., 1981). A 1-h daily exposure to 54
000 mg/m3 (10 000 ppm) for 3 months resulted in a narcotic effect
(sedation and transient sleep) and in an increase of relative liver
weight in rats, but there was no evidence of organ damage (Torkelson
et al., 1958).
When rats were exposed continuously for 100 days to 1350 or 5400
mg/m3 (250 or 1000 ppm), no effects were observed in the low-dose
group but an increase in relative liver weight was seen in the
high-dose group (McEwen & Vernot, 1974). No toxicity was observed in
similar experiments with dogs and monkeys (McEwen & Vernot 1974).
Exposure of mice to 5400 mg/m3 (1000 ppm) continuously for 14
weeks resulted in marked liver changes (elevated relative liver
weight, moderate liver triglyceride accumulation, and necrosis of
individual hepatocytes). Electron microscopy showed extensive
cytoplasmic modifications consisting of vesiculation of the rough
endoplasmic reticulum with loss of attached polyribosomes, increased
smooth endoplasmic reticulum, microbodies (peroxisomes), and
triglyceride droplets. The observed toxic effects were similar to,
but much less severe than, those produced by carbon tetrachloride.
Only minor cytoplasmic alterations were seen in mice exposed to 1350
mg/m3 (250 ppm) (McNutt et al., 1975).
No signs of toxicity were observed in rats, rabbits, guinea-pigs,
dogs or monkeys exposed to 2730 mg/m3 (500 ppm) 7 h/day, 5 days/week
for 6 months (Torkelson et al., 1958).
In a study by Prendergast et al. (1967), rats, rabbits,
guinea-pigs, dogs, and monkeys were exposed continuously to 754 or
2059 mg/m3 for 90 days. At the higher dose level, there were no
deaths in any species after 90 days, and the authors stated that no
visible toxic signs were observed. However, at the lower dose level,
some deaths occurred (2 out of 15 rats and 1 out of 3 rabbits).
Varying degrees of lung congestion were noted in the surviving
animals. In view of this and the deaths at the lowest dose tested,
the authors stated that no positive conclusion could be drawn as to
whether the effects were associated with the exposure.
No adverse effects were seen in male Wistar rats exposed to 1100
mg/m3 (204 ppm) 8 h/day, 5 days/week for 14 weeks (Eben & Kimmerle,
1974).
When young adult Mongolian gerbils (Meriones ungiculatus) were
continuously exposed to 378, 1134 or 5400 mg/m3 (70, 210 or 1000
ppm) for 3 months, followed by a 4-month period without exposure,
increased glial fibrillary acidic protein (GFA) was found in the
cerebral cortex at the two highest exposure levels, indicating
astrogliosis in this region of the brain (Rosengren et al., 1985). In
a similar study by Karlsson et al. (1987), the DNA concentrations in
several brain regions were decreased in animals exposed to 378 mg/m3
(70 ppm), the only exposure concentration used.
Table 13 summarizes data from short-term inhalation studies.
7.1.2.2 Oral administration
When rats were dosed orally with 1,1,1-trichloroethane in corn
oil, 5 days/week for 6 weeks, a dosage of 3.2 g/kg body weight per day
had no adverse effects. However, total doses of 5.6 g/kg body weight
in females and 10 g/kg body weight in males induced 40% mortality and
decreased the body weight of survivors (NCI, 1977). In mice no
mortality was seen with a dose of 5.6 g/kg body weight. Thus,
trichloroethane has low toxicity in rats and mice following repeated
exposure by the oral route.
Table 13. Short-term toxicity (exposure-effect relationships) in
experimental animals exposed to 1,1,1-trichloroethane by
inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 5400 100 days increased relative McEwen & Vernot
liver weight (1974)
Rat 1100 14 weeks, NOAEL Eben & Kimberle
8 h/day, (1974)
5 days/week
Gerbil 5400 3 months reduced brain weight Rosengren et al.
(1985)
Gerbil 380 3 months biochemical changes Karlsson et al.
in the brain (1987)
Mouse 5400 14 weeks liver necrosis McNutt et al.
(1975)
Mouse 1350 14 weeks minor liver effects McNutt et al.
(1975)
Rat, dog, 2700 6 months, NOAEL Torkelson et al.
rabbit, 7 h/day, (1958)
monkey, 5 days/week
guinea-pig
Rat, dog, 2000 90 days NOAEL Prendergast et al.
rabbit, (1967)
monkey,
guinea-pig
7.2 Long-term exposure
In the NCI (1977) carcinogenesis study (see section 7.5.1 for
details of doses), reduced weight gain was noted in both rats and mice
during the exposure period at all dose levels. Furthermore, bloody
discharge and crusting around the eyes of some rats was observed
during the second year (NCI, 1977).
In an oncogenicity inhalation study, female rats exposed to the
highest concentration (8100 mg/m3, 1500 ppm) of 1,1,1-
trichloroethane exhibited a significant decrease in body weight, and
both females and males showed very slight microscopic hepatic effects
at 6, 12, and 18 months. At lower concentrations and in male rats and
mice of both sex, no toxicity was observable (Quast et al., 1988).
7.3 Reproductive toxicity, embryotoxicity, and teratogenicity
Inhalation studies in female rats and mice gave no evidence of
any teratogenic or fetotoxic effects followed exposure to
1,1,1-trichloroethane at a concentration of 4700 mg/m3 (875 ppm) for
7 h/day on days 6-15 of gestation (Leong et al., 1975). No signs of
maternal toxicity were noted.
In a study of female rats exposed to a higher
1,1,1-trichloroethane concentration (11 340 mg/m3, 2100 ppm, 6
h/day, 5 days/week) for 2 weeks before mating and/or during 20 days of
gestation (6 h/day, 7 days/week), there was some evidence of
fetotoxicity. This consisted of reduced fetal weight in the group
exposed during pregnancy only and minor visceral and skeletal
abnormalities, such as delayed ossification, in the group exposed
before and during pregnancy (York et al., 1982). No signs of maternal
toxicity were observed.
In a two-generation fertility study incorporating teratogenicity
and dominant lethal elements, mice received 0, 100, 300 or 1000 mg
1,1,1-trichloroethane/kg daily in the drinking-water (Lane et al.,
1982). Two or three litters were produced from each of the two
generations, and the parental animals and offspring were used
variously to investigate fertility, developmental toxicity, and
dominant lethal effects. No adverse effects on any aspect of
reproduction were evident in this study. The level of
1,1,1-trichloroethane in the drinking-water in this study was reported
to be up to 6000 mg/litre but was not actually measured. In a further
study, rats exhibited slight aversion to 30 mg/litre in drinking-water
(George et al. 1989).
Rats (Sprague-Dawley) and rabbits (New Zealand White) were
exposed on days 6-15 and 6-18 of gestation, respectively, to 0, 5400,
16 200 or 32 400 mg/m3 (0, 1000, 3000 or 6000 ppm) for 6 h/day.
1,1,1-Trichloroethane was found to be developmentally toxic to both
rats and rabbits at the highest dose level, producing an increase in
unossified and poorly ossified cervical centra, decreased female fetal
body weight, an increase in non-viable implantations in rats, and an
increase in the frequency of bilateral extra 13th rib in rabbits.
Both rats and rabbits showed a NOEL of 16 200 mg/m3 for
developmental toxicity. For both species, the developmental toxicity
was seen in the presence of maternal tox-icity (Personal communication
by E.Z. Francis to S. Ells, US EPA, Test Rules Development Branch,
Existing Chemical Assessment Division. Review of Developmental
Toxicity Studies on 1,1,1-Trichloroethane (TCE). OHEA, ORD. Sept. 28,
1988).
Dapson et al. (1984) reported that exposure to 10 mg
1,1,1-trichloroethane per litre drinking-water caused cardiac
anomalies in developing Sprague-Dawley rats. The repeatability of
this preliminary study was investigated by George et al. (1989), who
exposed male and female CD rats to 3, 10 or 30 mg of 97% pure
1,1,1-trichloroethane per litre drinking-water using 0.05% Tween 80 as
an emulsifying agent. The animals were exposed for a period of 14
days before and at least 13 days after co-habitation. Sperm-positive
females continued to be exposed during pregnancy and lactation until
postnatal day 21. No significant effects were noted concerning the
reproductive competence of the parental animals or the postnatal
growth and development of the offspring (fertility, length of
gestation period, litter size, pup body weight, and pup survival were
all investigated). In addition, there was no increase in the
incidence of any cardiac malformations or other anomalies. Thus the
finding of Dapson et al. (1984) was not confirmed using a different
strain of rats and a 3-fold higher concentration of
1,1,1-trichloroethane.
7.4 Mutagenicity
Appraisal
1,1,1-Trichloroethane has been tested extensively for
mutagenicity, both in vitro and in vivo, and the principal studies
are summarized in Table 14. The results of most of the studies were
clearly negative. The positive results seen in some in vitro tests
give rise to some concern, but may well have been due to the presence
of stabilizers and/or impurities. Some of the studies yielding
negative results were of insufficient sensitivity. All of the in
vivo tests gave negative results. Overall, it appears that
1,1,1-trichloroethane does not have significant mutagenic potential.
Table 14. Mutagenicity studies
Test method Organism Test conditions Result References
In vitro studies on microorganisms
Baterial Salmonella typhimurium many studies, using mostly negative; some Simmon et al. (1977), Snow et al. (1979),
mutagenicity or Escherichia coli liquid or vapour, positive results attributed Nestmann et al. (1980), Gocke et al. (1981),
with and without S9 to stabilizers or impurities Rowland & Severn (1981), McDonald (1981),
activation Nagao & Takahashi (1981), Richold & Jones
(1981), Simmon & Shepherd (1981),
Ichinotsubo et al. (1981), de Serres &
Ashby (1981), HSE (1984), Shimada et al.
(1985)
Bacterial DNA E. coli or Bacillus several studies mainly negative; Kada (1981), Ichinotsubo et al. (1981),
damage/repair subtilis occasionally equivocal Green (1981), Tweats (1981), Rosenkranz
tests results et al. (1981), de Serres & Ashby (1981)
Yeasts, Saccharomyces numerous studies negative HSE (1984), de Serres & Ashby (1981),
gene mutation, cerevisiae
gene conversion Schizosaccharomyces
pombe
S. cerevisiae and mice 5000 mg/kg negative Loprieno et al. (1979)
(host-mediated assay) intraperitoneal
Table 14 (contd).
Test method Organism Test conditions Result References
In vitro mammalian cell studies
Chromosome CHO cells ± S9 activation clear positive without S9; Galloway et al. (1987)
aberrations equivocal with S9
Gene mutation L5178Y mouse ± S9 equivocal with S9; Myhr & Caspary (1988)
lymphoma cells negative without S9
Unscheduled rat, mouse hepatocytes; ± S9 negative Althaus et al. (1982), Martin &
DNA synthesis HeLa cells McDermid (1981)
DNA repair rat hepatocytes ± S9 mainly negative; some Williams (1983), Shimada et al. (1985)
positive results attributed
to presence of stabilizers
or impurities
Sister chromatid CHO cells ± S9 negative Perry & Thomson (1981)
exchange equivocal Galloway et al. (1987)
Cell various cell types ± S9 both positive and Daniel & Dehnel (1981), Styles (1981),
transformation negative results Price et al. (1978), Hatch et al. (1983)
Tu et al. (1985), HSE (1984)
Drosophila studies
Sex-linked 25 nM negative Gocke et al. (1981)
recessive
lethals
Table 14 (contd).
Test method Organism Test conditions Result References
In vivo mammalian studies
Chromosome rats 875 or 1750 ppm negative Quast et al. (1978)
aberrations 6 h/day, 52 weeks
(bone marrow)
Micronucleus mice 34-67 mg/kg negative Salamone et al. (1981)
intraperitoneal
Micronucleus mice 11-42 mg/kg negative Tsuchimoto & Matter (1981)
intraperitoneal
Micronucleus mice 266-2000 mg/kg negative Gocke et al. (1981)
intraperitoneal
Dominant mice 100-1000 mg/kg per negative Lane et al. (1982)
lethal day oral
Sperm mice 130-2680 mg/kg per negative Topham (1980)
morphology day intraperitoneal
DNA binding rats and mice low binding Prodi et al. (1988)
(liver)
7.5 Carcinogenicity
1,1,1-Trichloroethane has been investigated for carcinogenic
effects in life-time bioassays on both rats and mice by oral
administration or inhalation.
7.5.1 Oral administration
In a US National Cancer Institute (NCI) assay, 7 week-old
Osborne-Mendel rats were fed by gavage a 75% solution of technical
grade 1,1,1-trichloroethane in corn oil, 5 days/week for 78 weeks.
The daily doses were calculated to be 750 or 1500 mg/kg body weight,
and the study was terminated after 110 weeks. The number of animals
in each treated group was 50 of each sex, and the control groups
consisted of 20 animals of each sex, which were not vehicle dosed. No
increased tumour incidence was found (NCI, 1977). However, because of
an unusually high mortality both among treated and control animals
(only 6 out of 240 animals survived the 110 weeks to the end of the
experiment), this study was considered insufficient by US EPA (1984)
and placed in class D (not applicable to assess human
carcinogenicity).
In another NCI study with 5-week-old B6C3F1 mice, technical grade
trichloroethane containing 3% 1,4-dioxane was given by gavage as a
40-60% solution in corn oil at two dose levels (average daily doses:
2807 and 5615 mg/kg body weight) 5 days/week for 78 weeks. The same
number of animals was used as in the NCI rat study reported above.
All surviving animals were killed after 90 weeks of study. A variety
of neoplasms was observed in the treated groups, but the incidence was
not statistically significantly different from that of the control
groups (NCI, 1977). Again the mortality rate was very high in both
treated and control animals, although it was a little lower than in
the rat study. Nevertheless this study was also considered
insufficient (US EPA 1984).
In a study by Maltoni et al. (1986), groups of male and female
rats were given 1,1,1-trichloroethane (500 mg/kg body weight) in olive
oil by gavage (4 to 5 days/week for 104 weeks). The
1,1,1-trichloroethane contained about 4% 1,4-dioxane and 1% of other
impurities. An increased incidence of "leukaemias" was detected in
the exposed group. However, data were not evaluated statistically nor
were any data for historical controls supplied.
7.5.2 Inhalation
In an inhalation study carried out by the Dow Chemical Co.,
1-month-old Sprague-Dawley rats were exposed to technical
1,1,1-trichloroethane (6 h/day, 5 days/week for 12 months) and
observed 18 months later, before the survivors were killed. Each dose
group consisted of 96 rats of each sex. The unexposed control group
consisted of the same number of rats as the two exposed groups
together. The two exposure concentrations used were 4778 mg/m3 (875
ppm) and 9555 mg/m3 (1750 ppm). The treatment had no significant
effect on mortality, and the tumour incidence in the treated animals
was similar to that of the controls (Rampy et al., 1977). The purity
of the 1,1,1-trichloroethane used was 96%, and the stabilizers were 3%
1,4-dioxane, 0.4% nitromethane, and 0.5% butylene oxide. In the
validation of this study, it was pointed out that the exposure period
was only 12 months and not lifelong, that no subchronic range-finding
test had been done, and that the exposure concentration could have
been too low (US EPA, 1984).
A more recent long-term inhalation study, this time on both rats
and mice, has been carried out by the Dow Chemical Co. (Quast et al.,
1988). Groups of male and female 4- to 6-week-old Fischer-344 rats
and 5- to 6-week-old B6C3F1 mice (80 of each sex per group) were
exposed to 1,1,1-trichloroethane (technical formulation) vapour
concentrations of 0 mg/m3, 820 mg/m3 (150 ppm), 2730 mg/m3 (500
ppm), 8190 mg/m3 (1500 ppm) 6 h/day, 5 days/week for 2 years. Ten
animals of each sex from each group were predesignated for interim
sacrifices after 6, 12, and 18 months of exposure. The purity of the
formulation was 94% (by volume) 1,1,1-trichloroethane, 5% stabilizers
(butylene oxide, tert-amyl alcohol, methyl butynol, nitroethane, and
nitromethane) and < 1% minor impurities. In none of the treated
animal groups, did the tumour incidence differ significantly from that
of the controls.
The early studies with mice and rats were evaluated at IARC
(International Agency for Research on Cancer) and considered
inadequate (IARC, 1979; IARC, 1987).
7.6 Immunotoxicity and sensitization
Shmuter (1977) exposed rabbits to 1,1,1-trichloroethane
concentrations of 2 mg/m3 (0.4 ppm), 10 mg/m3 (1.8 ppm), and 100
mg/m3 (18 ppm) by inhalation 3 h/day, 6 days/week for 8-10 months.
At week 6, Salmonella typhimurium was injected subcutaneously to
investigate the immune response over an 8-month period. The antibody
response was depressed at both 10 mg/m3 and 100 mg/m3. This
reduction in immune response was followed by an increased
electrophoretic mobility of the antibodies toward alpha- and
ß-globulin fractions and also by a significant drop in the level of
normal haemolysins to the Forsman's antigen of the sheep erythrocytes
occuring during treatment.
7.7 Interactions
Interaction between 1,1,1-trichloroethane and ethanol was studied
in mice given ethanol (0-2 g/kg body weight) by gavage 30 min before
exposure to up to 120 000 mg trichloroethane/m3 (22 200 ppm) by
inhalation. Different combinations of exposure to the solvents
enhanced the toxicity of trichloroethane in the animals previous
exposed to ethanol. The observed behavioural and lethal effects were
dose related for each compound and additive (Woolverton & Balster,
1981).
When dogs anaesthetized with either pentobarbital sodium or
chloralose plus pentobarbital sodium were acutely exposed to a
commercial spot remover containing 1,1,1-trichloroethane, there was a
dose-dependent biphasic decline in arterial pressure with an initial
increase in the heart rate. A similar effect was obtained when
1,1,1-trichloroethane (99.5% by volume) was used instead of the
commercial spot remover. It was found that the type of anaesthetic
contributed in part to the effect. The authors reported that, in
addition to the peripheral vascular effects, significant alterations
in myocardial function occurred, and they concluded that the solvent
acts by inhibiting myocardial contractility (Herd et al., 1974).
7.8 Mechanisms of action
1,1,1-Trichloroethane given to mice intraperitoneally or by short
inhalation exposure has been shown to reduce the cyclic guanosine
monophosphate (cGMP) content in the cerebellum, cerebral cortex, and
brain stem. This was attributed to an increased rate of cGMP
hydrolysis (Nilsson, 1986a). The cyclic adenosine monophosphate
(cAMP) content in the brain stem of mice was increased in a
dose-related and time-dependant manner. This might be mediated by
adrenoceptor interaction stimulating the catalytic unit of adenylate
cyclase (Nilsson, 1986b).
A study on isolated mouse brain synaptosomes exposed in vivo
and in vitro showed that 1,1,1-trichloroethane increases calcium
influx in the brain stem but not in the cerebellum and cerebral
cortex. The data indicated that 1,1,1-trichloroethane may influence
voltage-dependant calcium channels in brain stem nerve endings
(Nilsson, 1987).
1,1,1-Trichloroethane suppressed the uptake of ovabain
taurocholate and 2-aminoisobutyric acid, but not of cadmium chloride
or 3- O-methyl-D-glucose, into isolated rat hepatocytes. Both the
cellular ATP level and ATP-ase activities in isolated membrane
fractions were also decreased. It was suggested that energy-dependant
transport functions are disrupted, owing to decreased ATP levels
and/or inhibition of membrane ATP-ases (Kukongviriyapan et al., 1990).
8. EFFECTS ON HUMANS
Appraisal
Both acute and long-term inhalation exposure to
1,1,1-trichloroethane can produce CNS effects ranging from slight
behavioural changes to unconsciousness. Death may follow from
respiratory failure or cardiac arrest. There is also the possibility
of hepatic alterations and cardiac damage.
Ingestion and dermal exposure do not appear to be serious
concerns, although skin irritation may occur.
1,1,1-Trichloroethane has properties (see chapter 2) that make it
a dangerous compound in poorly ventilated areas and confined spaces
such as tanks and vaults. The vapour is five times more dense than
air, which may result in non-uniform distribution, and very high
levels can occur in certain areas such as the bottom of empty tanks
and container vessels. The combination of these properties has been
the cause of some serious accidents, including fatalities. Solvent
abuse has also produced a large number of fatalities.
There are insufficient studies to permit any conclusion
concerning the reproductive and carcinogenic effects of this compound
on humans.
Exposure to 1,1,1-trichloroethane may increase sensitivity to
other compounds, e.g., anaesthetics.
The NOEL for humans appears to be in the region of 1350 mg/m3
(250 ppm).
Table 15 summarizes the toxic effects of 1,1,1-trichloroethane
after inhalation exposure.
8.1 Controlled human studies
Several studies on volunteers under controlled conditions have
investigated the effects of 1,1,1-trichloroethane on the CNS as
measured by performance in certain behavioural tests. These studies
have usually been conducted using the technical grade of
1,1,1-trichloroethane containing stabilizers, since this is the type
of exposure that occurs most frequently.
Table 15. Toxic effects in humans related to inhalation exposure to 1,1,1-trichloroethane
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
378 000 70 000 a few min death Northfield (1981)
54 000 10 000 2 min general anaesthesia when given Dornette & Jones (1960)
together with nitrous oxide (N2O)
> 27 000 > 5000 10 min death Kleinfeld & Feiner (1966)
27 000 5000 5 min marked incoordination Stewart (1968)
10 260 1900 5 min disturbance of equilibrium Torkelson et al. (1958)
5400 1000 short-term mild eye and nasal discomfort, Stewart (1968)
impairment of coordination
5000 920 70-75 min CNS disturbances (lightheadedness), Torkelson et al. (1958)
slight eye irritation
4860 900 73 min impaired performance in Romberg's Stewart et al. (1961)
test, mild eye irritation
2700 500 6-7 h/day for impaired performance in Romberg's Stewart et al. (1969)
5 days test after 1 h of exposure, mild
eye irritation, drowsiness, headaches
2700 500 90-450 min no CNS disturbances Torkelson et al. (1958)
2430 450 30 min dizziness, mild eye irritation Salvini et al. (1971)
2430 450 30 min reduced reaction time and Gamberale & Hultengren (1973)
perceptual speed
Table 15 (contd).
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
1890 350 30 min reduced perceptual speed Gamberale & Hultengren (1973)
1900 350 3.5 h reduced psychomotor performance Mackay et al. (1987)
< 1860 < 345 6.7 years NOEL Maroni et al. (1977)
< 1350 < 250 1-6 years NOEL Kramer et al. (1978)
1350 250 30 min NOEL Gamberale & Hultengren (1973)
950 175 3.5 h reduced reaction time MacKay et al. (1987)
540 100 odour threshold Stewart (1968)
8.1.1 Single exposure period
In a study by Torkelson et al. (1958), four volunteers were
exposed in an inhalation chamber to average trichloroethane levels of
4968 mg/m3 (920 ppm) for 70-75 min. Symptoms of slight CNS
disturbances (lightheadedness) were noted in three of the four
subjects and slight eye irritation was noted in one individual. Two
volunteers reported a strong odour during the experiment.
Concentrations in the range of 2700-2970 mg/m3 (500-550 ppm) for
90-450 min apparently produced no symptoms of CNS disturbance, but
exposure to 10 260 mg/m3 (1900 ppm) for 5 min produced a very
noticeable odour and obvious disturbances in equilibrium.
Unfortunately, full details of this study are not available.
Exposure to about 2700 mg/m3 (500 ppm) trichloroethane for
78-186 min did not result in CNS toxicity or other effects (Stewart et
al., 1961). However, exposure to about 4860 mg/m3 (900 ppm) for up
to 73 min resulted in CNS disturbances (light-headedness and impaired
Romberg test performance) in approximately half of the subjects. Mild
eye irritation was also observed. In another experiment in this
series, the volunteers were exposed to increasing concentrations in
the range of 0-14 310 mg/m3 (0-2650 ppm) over a 15-min period.
Signs of mild eye irritation were noted in six out of seven subjects
when the concentration reached about 5400 mg/m3 (1000 ppm), and
there was a rapid increase in CNS symptoms, principally dizziness.
Throat irritation was experienced by six out of seven subjects at
about 10 800 mg/m3 (2000 ppm). Severe CNS disturbances (imbalance,
light-headedness) occurred at the end of the exposure period, when the
concentration had reached 14 310 mg/m3. In addition, minor effects
on liver and kidney function were seen. The authors considered that
all the effects were reversible.
The effect on young men of exposure to a mean trichloroethane
level of 2430 mg/m3 (450 ppm) for two periods of 4 h (separated by
a lunch break of 1´ h) was investigated by Salvini et al. (1971). The
volunteers experienced dizziness and slight excitation, but only
during the first 30 min of exposure, and mild eye irritation was
noted. No significant change in performance in any of the behavioural
tests was seen.
In a study by Gamberale & Hultengren (1973), 12 male volunteers
were exposed to 1350 mg/m3 (250 ppm) for 30 min, followed by 1890
mg/m3 (350 ppm) for the next 30 min, and 2430 mg/m3 (450 ppm) and
2970 mg/m3 (550 ppm) for subsequent 30-min periods. At 2430
mg/m3, the reaction time, perceptual speed, and manual dexterity
were reduced, and at 1890 mg/m3 the perceptual speed was reduced.
The NOEL was 1350 mg/m3.
Mackay et al. (1987) reported the results of an exposure chamber
study in which volunteers were exposed to 950 mg/m3 (175 ppm) and
1990 mg/m3 (350 ppm) for 3 to 5 h. Several human psychomotor
behavioural tests were employed, including two new ones. One was
concerned with distraction of attention and concentration (the Stroop
test) and the other with analysing grammatical statements (the
syntactic reasoning test). Performance deficits in the psychomotor
behavioural tests were recorded in volunteers exposed to
1,1,1-trichloroethane; these depended on the time of exposure and the
blood levels. The performance changes were rapid and occurred in some
cases within 20 min. However, different effects were found. In the
Stroop test, enhanced performance was observed following exposure,
whereas the syntactic reasoning test was found not to respond to
1,1,1-trichloroethane exposure effects. Measures of short-term
subjective well-being were not affected by exposure. Other
parameters, such as simple reaction time and four-choice reaction time
were increased.
The effect of exposure to trichloroethane on performance in a
very extensive range of behavioural tests was investigated by
Savolainen et al. (1981, 1982a, 1982b). Nine healthy male students
(aged 20-25) were exposed in an inhalation chamber to 1080 and 2160 mg
trichloroethane/m3 (200 and 400 ppm) for 4 h, with a 6-day interval
between exposure at the two levels. There was no clear indication of
any effects on the CNS, although some minor changes were noted in
body-sway measurements when the subjects had their eyes closed.
8.1.2 Repeated exposure
The effect of repeated exposure of male volunteers to 2700
mg/m3 (500 ppm), 7 h/day for 5 days, was studied in an inhalation
chamber (Stewart et al., 1969). Several subjective symptoms were
noted, e.g., drowsiness, headaches, lightheadedness, and eye and nose
irritation, but the absence of a control group made it difficult to
assess the significance. No performance impairment in behavioural
tests (modified Romberg test) was noted, apart from the inability of
two subjects to perform the balance test on two occasions.
8.2 Accidental exposure
Respiratory system symptoms, such as cough, breathlessness, and
chest tightness, are common in cases of acute poisoning (Boyer et al.,
1987).
Since 1,1,1-trichloroethane is a very volatile solvent with a
saturated vapour concentration of 864 g/m3 (160 000 ppm) at 25 °C,
there is a potential for very high air concentrations in confined
spaces, such as tanks and vaults. Furthermore, its vapour is five
times more dense than air. This may result in non-uniform
distribution in the workplace air and very high levels in certain
areas such as just above degreasing vessels and at the bottom of
empty tanks, even though levels elsewhere are low and safe. This
property has been the cause of some serious accidents and fatalities.
A near-fatal childhood intoxication was described by Gerace
(1981). A 4-year-old boy was playing under his bed covers with a
flower-making kit which contained a 30-cm3 container of
1,1,1-trichloroethane. Because of some unusual noises heard by a
sibling, the child was quickly discovered. However, he was not
breathing at this point. He was resuscitated on the way to the
hospital but remained comatose for about 12 min after arrival. The
author suggested that the child was rendered unconscious by the
1,1,1-trichloroethane and that the noises heard were seizures caused
by hypoxia. The child was released 48 h after admission. According
to the author, clinical investigation during the child's hospital stay
demonstrated some hepatic abnormalities with alteration in liver
bilirubin.
Caplan et al. (1976) reported the death of a woman due to
accidental exposure while cleaning a paint spill using a paint thinner
in a poorly ventilated room. Autopsy results showed "mild fatty
changes" in the liver and acute oedema and congestion of the lungs.
The concentrations of 1,1,1-trichloroethane in the brain, kidney,
liver, and blood were 36, 12, 5, and 2 mg/100 ml, respectively.
8.2.1 Confined spaces at workplaces
A man who had spent 10 min in a vault, where the concentration of
1,1,1-trichloroethane was estimated to be well in excess of 27 g/m3
(5000 ppm), collapsed and died shortly after leaving the vault
(Kleinfeld & Feiner, 1966).
Seven fatalities from using trichloroethane to clean equipment on
naval ships or aircraft tanks have been reported (Stahl et al., 1969;
Hatfield & Maykoski, 1970). The exposure level in one of these cases
was estimated to be in the order of 270 g/m3 (50 000 ppm). In all
cases the most significant pathological finding was pulmonary oedema.
Liver damage was not seen.
Other case histories with accidental death due to
1,1,1-trichloroethane exposure in confined spaces have been reported
(Stewart 1968; Bonventre et al., 1977).
During the period 1961-1980, the Factory Inspectorate in the
United Kingdom revealed 52 incidents due to trichloroethane (McCarthy
& Jones, 1983). Loss of consciousness was reported in 26 cases and
death in three cases, all involving very young workers. In one case
the worker died after leaning over an open tank of trichloroethane,
apparently to wash his hands in the solvent. He was found unconscious
slumped over the side of the tank, and the solvent concentration in
his breathing zone was later estimated to be about 378 g/m3 (70 000
ppm) (Northfield, 1981). In a second case, a worker was using
trichloroethane for cleaning purposes in a closed room (Jones &
Winter, 1983). He was found on the floor with chemical burns on the
head and neck, which was consistent with prolonged skin absorption
from spills of solvent on the floor. The third case was a teenage
worker who cleaned the interior of a car with a cloth soaked in the
solvent (Jones & Winter, 1983). Death was presumed to be from
1,1,1-tri-chloroethane intoxication and simultaneous inhalation of
vomit.
8.2.2 Solvent abuse
During the 1960s, 110 cases of sudden death from solvent abuse
were reported in the USA. Of these, spot remover containing
1,1,1-trichloroethane was identified as responsible for 29 cases
(Bass, 1970). Death frequently occurred immediately following
stressful activity. Where autopsies were performed, no abnormalities
could be detected, and it was suggested that deaths was due to cardiac
arrhythmia.
Over the period 1971-1981, 140 deaths from solvent abuse were
reported in the United Kingdom; 20 of them were believed to be due to
1,1,1-trichloroethane, mainly in dry-cleaning fluid, domestic cleaners
or plaster remover (Anderson et al., 1982).
In the case of a fatality due to the abuse of cleaning solvent
containing 1,1,1-trichloroethane, very minor hepatic changes were seen
(Hall & Hine, 1966). In four cases of deaths caused by inhalation of
typewriter correction fluid containing 1,1,1-trichloroethane and
trichloroethylene, the autopsies showed no evidence of kidney or liver
damage (King et al., 1985).
According to Marjot & McLeod (1989), toluene and the chlorinated
hydrocarbons 1,1,1-trichloroethane and trichloroethylene can cause
permanent damage to the kidney, liver, heart, and lung in certain
volatile substance abusers.
8.2.3 Medical use
Trichloroethane has been used as an anaesthetic on an
experimental basis. General anaesthesia in humans has been rapidly
induced by 2 min of exposure to 54 000 mg/m3 (10 000 ppm) given with
a mixture of nitrous oxide (N2O) and oxygen (80:20). Cardiovascular
side effects (slight to severe hypotension, cardiac arrhythmias, and,
in one case, cardiac arrest) were observed in several of the patients
during anaesthesia. In view of these effects, the experimental use of
1,1,1-trichloroethane as an anaesthetic was discontinued (Dornette &
Jones, 1960).
In the USA, after aerosol drug products containing
1,1,1-trichloroethane had been implicated in 21 deaths from their
abuse or misuse, the Food and Drug Administration (FDA) removed the
products from the market (US Food and Drug Administration, 1973). The
medical products were mainly used as cough suppressants and contained
1,1,1-trichloroethane as a solvent for the active ingredients and to
reduce the vapour pressure of the propellants. The FDA found lack of
general recognition by qualified experts of the safety or
effectiveness of 1,1,1-trichloroethane in aerosol drug products
intended for inhalation.
Under 1977 rules from the FDA, it was stated that
1,1,1-trichloroethane is potentially toxic to the cardiovascular
system by sensitizing the heart to adrenaline (Food and Drug
Administration, 1977).
8.2.4 Ingestion
In a case of accidental ingestion of about 600 mg
trichloroethane/kg, signs of severe gastrointestinal irritation
(vomiting, diarrhoea) were evident shortly after the ingestion. There
was no evidence of any CNS disturbance or any liver or kidney
dysfunction (Stewart & Andrews, 1966).
Ingestion of about 30 ml trichloroethane has been found to cause
severe vomiting and diarrhoea (CEC, 1986).
8.2.5 Drinking-water contamination
In December 1981, a water well in Santa Clara County, California,
USA, was removed from service after contamination with
1,1,1-trichloroethane and other solvents was detected. A
1,1,1-trichloroethane level of 1.7 mg/litre was detected at the well
head, but the level of the other contaminants (dichloroethylene,
isopropyl alcohol, and freon) was not reported. Subsequently
suspicion of a cluster of spontaneous abortions and congenital heart
anomalies from pregnancies conceived during 1980-1981 was reported by
the local community. Epidemiological studies indicated that a cluster
did exist, that the odds ratio for spontaneous abortion was 2.3 and
the relative risk for congenital malformations was 3.1, but they were
unable to draw a direct link between the ingestion of the contaminated
water and these effects (Goldberg et al., 1990).
8.3 Effects on the skin and eyes
Prolonged or repeated skin contact with 1,1,1-trichloroethane may
lead to dermatitis, due to its defatting action. However, despite its
wide use only a few cases of skin irritancy have been reported
(Torkelson et al., 1958).
Immersion of a hand in liquid 1,1,1-trichloroethane for 30 min
resulted in mild erythema, which persisted for one hour (Stewart &
Dodd, 1964).
Splashed in the eye, the compound may produce conjunctivitis
(Stewart, 1968). However, accidental eye contact has been reported to
produce only transient irritation (Savolainen et al., 1982a,b).
Exposure to a vapour concentration of 2700 mg/m3 (500 ppm) may
produce mild eye irritation (see section 8.1).
8.4 Long-term occupational exposure
A case study described a patient with liver cirrhosis after
several years of heavy exposure to trichloroethylene followed by 3
months of work that frequently involved using aerosolized degreaser
containing trichloroethane (Thiele et al., 1982).
An investigation of the neurotoxicity of trichloroethane was
carried out on a group of 22 female workers exposed to the solvent for
an average period of 6.7 years. The group was subdivided into three
exposure groups (mean levels: 594 mg/m3 (110 ppm), 756-864 mg/m3
(140-160 ppm), and 1080-1863 mg/m3 (200-345 ppm)). Each woman had
a general physical examination, and information about symptoms was
obtained from a questionnaire. The measurement of nerve conduction
velocity and psychometric functions indicated no differences between
the exposed workers and an unexposed control group of 7 workers
(Maroni et al., 1977).
Hodgson et al. (1989) reported that four out of ten clinical
cases of fatty liver disease were associated with occupational
exposure to 1,1,1-trichloroethane.
A matching-pair study of 151 pairs of workers at textile plants,
with and without daily exposure to trichloroethane levels of up to
1365 mg/m3 (250 ppm), found no toxic effect among the exposed
workers, the majority of whom were exposed to less than 810 mg/m3
(150 ppm) for a period of 1-6 years (Kramer et al., 1978).
In a case-control study of the effect of occupational exposure to
various solvents on spontaneous abortion rates in Finland (1973-1983),
a statistically significant elevation in relative risk was found for
exposure to organic solvents in general. However, there were very few
cases related specifically to 1,1,1-trichloroethane. The low
statistical power, therefore, precludes any conclusion as to whether
the data show an effect or not for 1,1,1-trichloroethane (Lindbohm et
al., 1990)
There are no adequate epidemiological studies available
concerning long-term exposure to 1,1,1-trichloroethane and
carcinogenicity.
8.5 Interactions
Chronic cardiac arrhythmia developed in two cases, after routine
halothane anaesthesia, in subjects with previous repeated long-term
exposure to 1,1,1-trichloroethane (in one case, occupational and in
the other, due to solvent abuse). The effects involved myocardiac
damage with either ventricular arrhythmia or deterioration of left
ventricular function. In one of the cases, the pathologist suggested
a subacute myocarditis superimposed on long-standing damage. The
authors postulated that the interaction between the two chemicals may
have caused the cardiac deterioration and concluded that cardiac
damage sustained after long-term exposure to 1,1,1-trichloroethane may
be severe, life-threatening, and irreversible (McLeod et al.,1987)
Marjot & McLeod (1989) reported two cases, a 14-year-old boy
exposed to 1,1,1-trichloroethane via solvent abuse and a 54-year-old
man exposed occupationally, where the cardiac toxicity of
1,1,1-trichloroethane may have been increased by exposure to halothane
(2-bromo-2-chloro-1,1,1-trifluoroethane) as an anaesthetic.
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
Appraisal
1,1,1-Trichloroethane is not likely to be a significant hazard to
non-target organisms in the environment. For example, 96-h LC50
values for fish ranged from 33 mg/litre for dabs to 105 mg/litre for
fathead minnows. The 48-h LC50 for Daphnia was around 60 mg/litre.
The EC50 for carbon dioxide incorporation into algae was 5
mg/litre.
9.1 Microorganisms
A toxic threshold of 93 mg 1,1,1-trichloroethane/litre has been
estimated for the inhibition of cell multiplication in the bacterium
Pseudomonas putida (Bringmann & Kuhn, 1977). The 3-h EC50 values,
for inhibition of photosynthesis (measured by monitoring the uptake of
14CO2), for the green algae Chlamydomonas angulosa (at a cell
concentration of 5 x 104 cells/ml) and Chlorella vulgaris (at a
cell concentration of 20 x 104 cells/ml) were 280 mg/litre and 153
mg/litre, respectively, under static conditions (Hutchinson et al.,
1980). Bringmann & Kuhn (1978) calculated toxic thresholds of 350
mg/litre and 430 mg/litre, respectively, for inhibition of cell
multiplication, over an 8-day period, of the cyano bacterium
(blue-green alga) Microcystis aeruginosa and the green alga
Scenedesmus quadricauda. The 96-h EC50 for chlorophyll a and cell
number in the green alga Selenastrum capricornutum was > 669
mg/litre (US EPA, 1980a,b). Pearson & McConnell (1975) assessed the
toxicity of 1,1,1-trichloroethane to the unicellular alga
Phaeodactylum tricornutum by measuring changes in the uptake of
carbon, from 14CO2, during photosynthesis, and obtained an EC50
of 5 mg/litre.
9.2 Aquatic organisms
The acute toxicity of 1,1,1-trichloroethane to aquatic organisms
is summarized in Table 16.
Flow-through tests gave much lower LC50 values than static
tests, suggesting that some loss via evaporation may be occurring.
The 48-h LC50 for aquatic invertebrates ranges from 7.5 mg/litre for
barnacles in a flow-through test to > 530 mg/litre for daphnids in a
static test. The 96-h LC50 for fish ranges from 33 mg/litre for the
dab (Limanda limanda) in flow-through tests to 105 mg/litre for the
fathead minnow (Pimephales promelas) in static tests.
Table 16. Acute toxicity of 1,1,1-trichloroethane to aquatic organisms
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Invertebrates
Mysid shrimp stat 96 31.2 n US EPA (1980b)
(Mysidopsis bahia)
Barnacle nauplii stat 48 7.5 n Pearson & McConnell (1975)
(Eliminius modestus)
Water flea < 24 h stat 21-23 72 6.7-8.1 48 > 530 n LeBlanc (1980)
(Daphnia magna) 4-6 day stat 21-25 48 59.6 Abernethy et al. (1986)
4-6 day stat 48 57.6 Bobra et al. (1984)
Fish
Dab 15-20 cm flow 96 33 m Pearson & McConnell (1975)
(Limanda limanda)
Sheepshead minnow 8-15 mm stat 25-31 10-31d 96 71 (60-81) n Heitmuller et al. (1981)
(Cyprinodon variegatus)
Bluegill 0.32-1.2 g stat 21-23 32-48 6.7-7.8 24 110 n Buccafusco et al. (1981)
(Lepomis macrochirus) 0.32-1.2 g stat 21-23 32-48 6.7-7.8 96 72 (57-90) n Buccafusco et al. (1981)
stat 96 69.7 n US EPA (1980b)
Fathead minnow 1 g stat 12 7.8-8.0 96 105 (91-126) n Alexander et al. (1978)
(Pimephales promelas) 1 g flow 12 7.8-8.0 96 52.8 (43.7-77.7) m Alexander et al. (1978)
Table 16 (contd).
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Fish (contd)
Guppy 2-3 month statb 168 133 Konemann (1981)
(Poecilia reticulata)
a stat = static conditions (water unchanged for the duration of the test) unless stated otherwise; flow = flow-through conditions
(1,1,1-trichloroethane concentration continuously maintained)
b static conditions but water renewed daily
c hardness expressed as mg CaCO3 per litre
d value refers to salinity (%), not hardness
e n = nominal; m = measured
LeBlanc (1980) exposed the water flea Daphnia magna to
1,1,1-trichloroethane for 48 h under static conditions. At the
highest concentration of 530 mg/litre, no discernible effect was
observed. A no-observed-effect level (NOEL) of 43 mg/litre was
estimated by Heitmuller et al. (1981) after exposing the sheepshead
minnow (Cyprinodon variegatus) to 1,1,1-trichloroethane for 96 h.
When Thompson & Carmichael (1989) exposed the water flea Daphnia
magna to 1,1,1-trichloroethane concentrations of between 1.3 and 23
mg/litre for 17 days under semistatic conditions (water renewed every
2 days), no effect on mortality or reproduction (as measured by number
of offspring per parent) was observed at 1.3 mg/litre. However, the
mortality increased with increasing concentration of
1,1,1-trichloroethane from 30% at 2.4 mg/litre to 100% at 23 mg/litre.
The number of offspring per parent was significantly reduced at
concentrations of 2.4 mg/litre or more.
Thompson & Carmichael (1989) also exposed the mirror carp
Cyprinus carpio to 1,1,1-trichloroethane concentrations ranging from
1.4 to 30 mg/litre for 14 days under flow-through conditions. There
was no mortality at any of the concentrations, but at the highest
concentration the fish showed surfacing behaviour, loss of balance and
coughing, and their mean weight decreased. The authors considered,
therefore, that the NOEL was the next highest concentration, i.e. 7.7
mg/litre.
Alexander et al. (1978) observed fathead minnows (Pimephales
promelas), during exposure to 1,1,1-trichloroethane for up to 96 h,
for the following effects: loss of equilibrium, melanization,
narcosis, and swollen, haemorrhaging gills. The concentration
producing one or more of these observable effects in 50% of the fish
(EC50) was 11.1 mg/litre.
9.3 Terrestrial organisms
Thompson & Carmichael (1989) exposed emergent seedlings of crop
sorghum (Sorghum bicolor) and oil seed rape (Brassica napus) to
1,1,1-trichloroethane in the gaseous phase at nominal concentrations
of between 3.2 and 320 mg/litre (g/m3) for a period of 14 days (the
test material was replenished after 7 days in a sealed vessel). Mean
measured concentrations ranged from 58% to 75% of the nominal
concentration, reflecting the decline of 1,1,1-trichloroethane in the
gas phase concentration during the 7-day periods between toxicant
renewal. In the case of Sorghum bicolor, the 14-day EC50 (based
on mean plant weight and a mean measured 1,1,1-trichloroethane
concentration) was 48 mg/litre; the NOEL being 19 mg/litre. For
Brassica napus, the 14-day EC50 was 19 mg/litre and the NOEL 6.9
mg/litre, both values being based on mean measured concentrations.
Rajendran (1990) exposed 1- to 2-day-old pupae of the red flour
beetle (Tribolium castaneum) to 1,1,1-trichloroethane in fumigation
chambers. An LC50 of 208.4 mg/litre was estimated after a 24-h
exposure and a 20-day post-fumigation observation period. When pupae
were exposed to a mixture of 1,1,1-trichloroethane and methyl bromide
at a concentration of 2.5 mg/litre or 3.0 mg/litre, the toxicity was
similar to that of the individual components.
In a contact toxicity test, red earthworms (Eisenia foetida)
were exposed to 1,1,1-trichloroethane via filter paper in glass vials.
An LC50 of 83 µg/cm2 was estimated (Neuhauser et al., 1986).
Elovaara et al. (1979) injected 1,1,1-trichloroethane, at
concentrations ranging from 5 to 100 µmol/egg, into the air space of
fertilized chicken eggs at 3 or 6 days of incubation, and the
embryotoxicity was evaluated after 14 days of incubation. A clear
dose-response relationship with respect to survival was found,
irrespective of whether the eggs were injected after 3 or 6 days of
incubation. The approximate LD50 for 1,1,1-trichloroethane was
between 50 and 100 µmol/egg.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
Humans are exposed to 1,1,1-trichloroethane principally by
inhalation, where the substance is rapidly absorbed into the body.
Exposure by skin absorption or ingestion may also occur but, compared
to inhalation, is of lesser concern. 1,1,1-Trichloroethane is
considered not to bioaccumulate. The acute and chronic toxicities are
fairly low. Under conditions of high exposure there is a risk of
toxic effects; such conditions may occur in occupational exposures,
solvent abuse or accidents. Since 1,1,1-trichloroethane is volatile
and the vapour is much more dense than air, toxic and explosive
concentrations may occur unexpectedly in confined spaces. This has
caused several fatal and near-fatal accident at workplaces and
elsewhere. The critical effect in humans relates to the central
nervous system, leading to unconsciousness and respiratory arrest at
high concentrations. 1,1,1-Trichloroethane is less toxic to the liver
than most other organochlorine solvents. The no-observed-effect level
for humans appears to be in the region of 1350 mg/m3 (250 ppm).
Although one study suggested effects at a lower level, the
interpretation of the results was unclear.
Studies on Mongolian gerbils involving 1,1,1-trichloroethane
levels down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several brain regions.
If these findings are validated and confirmed, this may be of
significant concern.
No adequate study of carcinogenic effects on humans has been
published. A long-term inhalation study on rats and mice exposed to
8100 mg/m3 (1500 ppm) gave no evidence of any carcinogenic effects.
One study recorded increases numbers of "leukaemias", but it was
performed under non-standard protocol. It gave no statistical
evaluation and is, therefore, considered inadequate. It is unlikely
that 1,1,1-trichloroethane itself presents a significant risk of
genotoxic or carcinogenic effects in humans.
1,1,1-Trichloroethane is not teratogenic and elicits no
developmental effects at doses that are not maternally toxic. The
limited epidemiological evidence on reproductive effects is
inconclusive due to the low statistical power of the study.
10.2 Evaluation of effects on the environment
1,1,1-Trichloroethane is released in large quantities to the
environment. Due to its volatility and general stability it is a
ubiquitous environmental contaminant.
Although the ozone-depleting and global-warming potentials of
1,1,1-trichloroethane are lower than those of many
chlorofluorocarbons, the large-scale release of the compound into the
atmosphere merits serious concern in these respects.
As a result of its widespread use and disposal and its mobility
in soil, persistent 1,1,1-trichloroethane contamination of ground
water, including deep aquifers, occurs.
Conditions under which 1,1,1-trichloroethane causes
ecotoxicological effects are not found in the environment except after
spills.
11. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH AND
THE ENVIRONMENT
a) Emissions of 1,1,1-trichloroethane should be reduced as far as
is practicable in order to minimize exposure of workers and of
the general population.
b) The release of 1,1,1-trichloroethane into the environment must
be reduced to the greatest extent possible in order to avoid
damage to the ozone layer.
c) Contamination of ground water by 1,1,1-trichloroethane from
spills and waste disposal should be avoided.
d) Safe substitutes for 1,1,1-trichloroethane should be identified.
12. FURTHER RESEARCH
a) More study is needed to clarify the possible effects of
1,1,1-trichloroethane on the biochemistry of the central nervous
system and their significance.
b) More study is necessary to clarify the possible interaction
between 1,1,1-trichloroethane and ethanol in humans.
c) An adequate subchronic or long-term oral study is needed to
assess the hepatotoxicity of 1,1,1-trichloroethane.
d) Further epidemiological studies on reproductive effects are
desirable.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
IARC (International Agency for Research on Cancer) working groups
have evaluated 1,1,1-trichloroethane and concluded that the evidence
for genotoxicity is limited (IARC, 1979, 1987).
REFERENCES
ABERNETHY, S., BOBRA, A.M., SHIU, W.Y., WELLS, P.G., & MACKAY, D.
(1986) Acute lethal toxicity of hydrocarbons and chlorinated
hydrocarbons to two planktonic crustaceans: The key role of
organism-water partitioning. Aquat. Toxicol., 8: 163-174.
ADAMS, E.M., SPENCER, H.C., ROWE, V.K., & IRISH, D.D. (1950) Vapor
toxicity of 1,1,1-trichloroethane (methyl chloroform) determined by
experiments on laboratory animals. Arch. ind. Hyg., 1: 225-236.
AGGAZZOTTI, G. & PREDIERI, G. (1986) Survey of volatile halogenated
organics (VHO) in Italy. Water Res., 20: 959-963.
ALEXANDER, H.C., MCCARTY, W.M., & BARTLETT, E.A. (1978) Toxicity of
perchloroethylene, trichloroethylene, 1,1,1-trichloroethane, and
methylene chloride to fathead minnow. Bull. environ. Contam. Toxicol.,
20: 344-352.
ALTHAUS, F.R., LAWRENCE, S.D., SATTLER, G.L., LONGFELLOW, D.G., &
PITOT, H.C. (1982) Chemical quantification of unscheduled DNA
synthesis in cultured hepatocytes as an assay for the rapid screening
of potential chemical carcinogens. Cancer Res., 42(8): 3010-3015.
AMI (1988) [The Factories Inspectorate's data base for workplace
measurements, ATABAS, stock-taking for the period January 1983 -
December 1987], Copenhagen, Institute for the Working Environment (in
Danish).
ANDERSON, H.R., DICK, B., MACNAIR, R.S., PALMER, J.C., & RAMSEY, J.D.
(1982) An investigation of 140 deaths associated with volatile
substance abuse in the United Kingdom (1971-1981). Hum. Toxicol., 1:
207-221.
ARCHER, W.L. (1979) 1,1,1-Trichloroethane. In: Kirk-Othmer
encyclopedia of chemical technology, 3rd ed., New York, Chichester,
John Wiley and Sons, pp. 728-731.
ARCHER, W.L. & STEVENS, V.L. (1977) Comparison of chlorinated,
aliphatic, aromatic, and oxygenated hydrocarbons as solvents. Ind.
Eng. Chem. Prod. Res. Dev., 16: 319-325.
ASTRAND, I., KILBOM, A., WAHLBERG, I., & OVRUM, P. (1973) Methyl
chloroform exposure. I. Concentration in alveolar air and blood at
rest and during exercise. Scand. J. Work Environ. Health, 10: 69-81.
BASS (1970) Sudden sniffing death. J. Am. Med. Assoc., 212: 2075-2079.
BAUER, U. (1981) [Burden on man caused by harmful substances in the
environment.] Zbl. Bakteriol. Hyg. I Abt. Orig. B, 174: 200-237 (in
German).
BERODE, M., BOILLAT, M.-A., GUILLEMIN, M.P., WU, M.W., & SAVOLAINEN,
H. (1990) Demethylation pathways in caffeine metabolism as indicators
of variability in 1,1,1-trichloroethane oxidation in man. Pharmacol.
Toxicol., 67(1): 41-46.
BOBRA, A.M., SHIU, W.Y., & MACKAY, D. (1984) Structure-activity
relationships for toxicity of hydrocarbons, chlorinated hydrocarbons
and oils to Daphnia magna. In: Kaiser, K.L.E., ed. Quantitative
structure activity relations in environmental toxicology, Dordrecht,
Germany, D. Reidel Publishing Co., pp. 3-16.
BONNET, P., FRANCIN, J.-M., GRADISKI, D., RAOULT, G., & ZISSU, D.
(1981) Determination of the median lethal concentration of the
principal chlorinated aliphatic hydrocarbons in the rat. Arch. Mal.
prof. Méd. Trav. Sécur. soc., 41: 317-321.
BONVENTRE, J., BRENNAN, O., JASON, D., HENDERSON, A., & BASTOS, M.L.
(1977) Two deaths following accidental inhalation of dichloromethane
and 1,1,1-trichloroethane. J. anal. Toxicol., 4: 158-160.
BOUWER, E.J. & MCCARTY, P.L. (1983a) Transformations of 1- and
2-carbon halogenated aliphatic organic compounds under methanogenic
conditions. Appl. environ. Microbiol., 45: 1286-1294.
BOUWER, E.J. & MCCARTY, P.L. (1983b) Transformations of halogenated
organic compounds under denitrification conditions. Appl. environ.
Microbiol., 45: 1295-1299.
BOYER, J.D, AHLERT, R.C., & KOSSON, D.S. (1987) Degradation of
1,1,1-trichloroethane in bench-scale bioreactors. Hazardous Waste
hazardous Matter, 4: 241-260.
BRETHERICK, L., ed. (1981) Hazards in the chemical laboratory, 3rd
ed., London, The Royal Society of Chemistry.
BRETHERICK, L. (1989) 1,1,1-Trichloroethane. Chem. Eng. News, Dec. 12:
2.
BRINGMANN, G. & KUHN, R. (1977) [Limit values for the harmful effect
of water-endangering substances on bacteria (Psuedomonas putida) and
green algae (Scenedesmus quadricauda) in the cell division
inhibition test.] Z. Wasser Abwasser Forsch., 10: 87-98 (in German).
BRINGMANN, G. & KUHN, R. (1978) [Limit values for the harmful effect
of water endangering substances on blue algae (Microcystis
aeroginosa) and green algae (Scenedesmus quadricauda) in the cell
division inhibition test.] Vom Wasser, 50: 45-60 (in German).
BUCCAFUSCO, R.J., ELLS, S.J., & LEBLANC, G.A. (1981) Acute toxicity of
priority pollutants to bluegill (Lepomis macrochirus). Bull.
environ. Contam. Toxicol., 26: 446-452.
CAPLAN, Y.H., BACKER, R.C., & WHITAKER, J.Q. (1976)
1,1,1-Trichloroethane: report of a fatal intoxication. Clin. Toxicol.,
9: 69-74.
CEC (Commission of the European Communities) (1986) Organochlorine
solvents: Health risks to workers, Cambridge, Royal Society of
Chemistry, pp. 57-58.
CEFIC (1986) The occurrence of chlorinated solvents in the
environment. Prepared by a workshop of the European Council of
Chemical Industry Federation (CEFIC). Chem. Ind., 1986: 861-869.
CHIOU, C.T., PETERS, L.J., & FREED, V.H. (1979) A physical concept of
soil-water equilibria for nonionic organic compounds. Science, 206:
831-832.
CLARK, D.G. & TINSTON, D.J. (1982) Acute inhalation toxicity of some
halogenated and non-halogenated hydrocarbons. Hum. Toxicol., 1:
239-247.
CLASS, T. & BALLSCHMITER, K. (1986) Chemistry of organic traces in air
VI: Distribution of chlorinated C1 - C4 hydrocarbons in air over the
Northern and Southern Atlantic ocean. Chemosphere, 15: 413-427.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology, 3rd revised ed., New York, Chichester, John Wiley and
Sons, Vol. 2B.
COLEMAN, W.E., LINGG, R.D., MELTON, R.G., & KOPFLER, F.C. (1976) The
occurrence of volatile organics in five drinking water supplies using
gas chromatography/mass spectrometry. In: Keith, L.H., ed.
Identification and analysis of organic pollutants in water, Ann Arbor,
Michigan, Ann Arbor Science Publishers, Inc., pp. 305-327.
CORREIA, Y., MARTENS, G.J., VAN MESCH, F.H., & WHIM, B.P. (1977) The
occurrence of trichloroethylene, tetrachloroethylene and
1,1,1-trichloroethane in western Europe in air and water. Atmos.
Environ., 11: 1113-1116.
DAFT, J.L. (1988) Fumigant contamination during large-scale food
sampling for analysis. Arch. environ. Contam. Toxicol., 17: 177-181.
DAHLBERG, J.A., CHRISTIANSEN, V.O., & ERIKSEN, E.A. (1973) On the
formation of phosgene by photooxidation of methyl chloroform in
welding. Ann. occup. Hyg., 16: 41-46.
DALLAS, C.E., RAMANATHAN, R., MURALIDHARA, S., GALLO, J.M., &
BRUCKNER, J.V. (1989) The uptake and elimination of
1,1,1-trichloroethane during and following inhalation in rats.
Toxicol. appl. Pharmacol., 98: 385-397.
DANIEL, M.R. & DEHNEL, J.M. (1981) Cell transformation test with baby
hamster kidney cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 626-637 (Progress in Mutation Research, Vol. 1).
DANIELSSON, B.R.G., GHANTOUS, H., & DENCKER, L. (1986) Distribution of
chloroform and methylchloroform and their metabolites in pregnant
mice. Biol. Res. Pregnancy, 7: 77-83.
DAPSON, S.C., HUTCHEON, D.E., & LEHR, D. (1984) Effect of methyl
chloroform on cardiovascular development in rats. Teratology, 29: 25A.
DE NEVERS, N. (1986) A fatal fire with "non-flammable" methyl
chloroform. Arch. environ. Health, 41(5): 279-287.
DE SERRES, F.J. & ASHBY, J., ed. (1981) Evaluation of short term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland (Progress in
Mutation Research, Vol. 1).
DEVER, R.J. (1986) Responding to industrial contamination of
groundwater: a case study. J. Am. Water Works Assoc., 78: 82-86.
DICKSON, A.G. & RILEY, J.P. (1976) The distribution of short-chain
halogenated aliphatic hydrocarbons in some marine organisms. Mar.
Pollut. Bull., 7: 167-169.
DILLING, W.L., TERERTILLER, N.B., & KALLOS, G.J. (1975) Evaporation
rates and reactivities of methylene chloride, chloroform,
1,1,1-trichloroethane, trichloroethylene, tetrachloroethylene, and
other chlorinated compounds in dilute aqueous solutions. Environ. Sci.
Technol., 9: 833-838.
DORNETTE, W.H.L. & JONES, J.P. (1960) Clinical experience with
1,1,1-trichloroethane. Anesthesiol. Analg., 39: 249-253.
DRAKE, R.J. (1987) Fate of methylene chloride and
1,1,1-trichloroethane in unsaturated, biologically active soils,
University of Massachusetts (Dissertation).
DREHER, E.-L. (1989) Chlorinated hydrocarbons. In: Ullmann's
encyclopedia of chemical technology, New York, Weinheim, VCH
Publishers, Inc., Vol. A6, pp. 271-272.
DROZ, P.O., WU, M.M., CUMBERLAND, W.G., & BERUDE, M. (1989)
Variability in biological monitoring of solvent exposure. I
Development of a population physiological model. Br. J. ind. Med., 46:
447-460.
DUPRAT, P., DELSAUT, L., & GRADISKI, D. (1976) Pouvoir irritant des
principaux solvants chlorés aliphatiques sur la peau et les muqueuses
oculaires du lapin. Eur. J. Toxicol. environ. Hyg., 9: 171-177.
DÜSZELN, J.V., VON, LAHL, U., BÄTZER, K., CETINKAYA, M., STACHEL, B.,
& THIEMANN, W. (1982) [Volatile halogenated hydrocarbons in air, water
and food in the Federal Republic of Germany.] Dtsch. Lebensm.
Rundsch., 78: 352-356 (in German).
DÜSZELN, J.V., VON & THIEMANN, W. (1985) Volatile chlorinated
hydrocarbons in a costal urban atmosphere. Sci. total Environ., 41:
187-194.
EBEN, A. & KIMMERLE, G. (1974) Metabolism, excretion and toxicology of
methylchloroform in acute and subacute exposed rats. Arch. Toxicol.,
31: 233-242.
EDWARDS, P.R., CAMPBELL, I., & MILNE, G.S. (1982) The impact of
chloromethanes on the environment. Part 1. The atmospheric chlorine
cycle. Chem. Ind., 16: 574-578.
ELOVAARA, E., HEMMINKI, K., & VAINIO, H. (1979) Effects of methylene
chloride, trichloroethane, trichloroethylene and toluene on the
development of chick embryos. Toxicology, 12: 111-119.
ENTZ, R.C. & DIACHENKO, G.W. (1988) Residues of volatile halocarbons
in margarines. Food Addit. Contam., 5: 267-276.
ENTZ, R.C., THOMAS, K.W., & DIACHENKO, G.W. (1982) Residues of
volatile halocarbons in foods using headspace gas chromatography. J.
agric. food Chem., 30: 846-849.
FDA (1973) Trichloroethane. Aerosol drug products for human use. Fed.
Reg., 38: 21935-21936.
FDA (1977) Trichloroethane: status as a new drug in aerosolized drug
products intended for inhalation. FDA Final Rule. Fed. Reg., 42:
63386-63387.
FIELDING, M., GIBSON, T.M., JAMES, H.A., MCLOUGHLIN, K., & STEEL, C.P.
(1981) Organic micropollutants in drinking water, Medmenham, United
Kingdom, Water Research Centre (Technical Report No. TR 159).
FISCHER, M., FRIESEL, P., KOCH, G., MILDE, G., & ROSSKAMP, E. (1982)
Evaluation of water pollution caused by chlorinated and brominated
ethanes (Unpublished report submitted to the Commission of the
European Communities under Contract No. U71/516).
FISHBEIN, L. (1979) Potential halogenated industrial carcinogenic and
mutagenic chemicals. II. Halogenated saturated hydrocarbons. Sci.
total Environ., 11: 163-195.
FISHER, D.A., HALES, C.H., FILKIN, D.L., KO, M.K.W., SZE, N.D.,
CONNELL, P.S., WUEBBLES, D.J., ISAKSEN, I.S.A., & STORDAL, F. (1990a)
Model calculations of the relative effects of CFCs and their
replacement on stratospheric ozone. Nature (Lond.), 344: 508-512.
FISHER, D.A., HALES, C.H., WANG, W.-C., KO, M.K.W., & SZE, N.D.
(1990b) Model calculations of the relative effects of CFCs and their
replacement on global warming. Nature (Lond.), 344: 513-516.
FLUKA (1988) Catalogue 1988/89: Fluka Chemika-BioChemika, Buchs,
Switzerland, Fluka Chemie AG.
GALLOWAY, S.M., ARMSTRONG, M.J., REUBEN, C., COLMAN, S., BROWN, B.,
CANNON, C., BLOOM, A.D., NAKAMURA, F., AHMED, M., DUK, S. ET AL.
(1987) Chromosome aberrations and sister chromatid exchanges in
Chinese hamster ovary cells: Evaluations of 108 chemicals. Environ.
mol. Mutagen., 10: 1-175.
GAMBERALE, F. & HULTENGREN, M. (1973) Methylchloroform exposure II.
Psychophysiological functions. Scand. J. Work Environ. Health, 10:
82-92.
GEORGE, J.D., PRICE, C.J., MARR, M.C., SADLER, B.M., SCHWETZ, B.A.,
BIRNBAUM, L.S., & MORRISEY, R.E. (1989) Developmental toxicity of
1,1,1-trichloroethane in CD rats. Fundam. appl. Toxicol., 13, 641-651.
GERACE, R.V. (1981) Near-fatal intoxication by 1,1,1-trichloroethane.
Ann. Emergency Med., 10: 533-534.
GERKENS, R.R. & FRANKLIN, J.A. (1989) The rate of degradation of
1,1,1-trichloroethane in water by hydrolysis and dehydrochlorination.
Chemosphere, 19: 1919-1937.
GIGER, W., MOLNAR-KUBICA, E., & WAKEHAM, S. (1978) Volatile
chlorinated hydrocarbons in ground and lake waters. In: Aquatic
pollutants: Transformation and biological effects. Proceedings of the
2nd International Symposium on Aquatic Pollutants, Oxford, New York,
Pergamon Press, pp. 101-123 (Pergamon Series on Environmental Science
No. 1).
GOCKE, E., KING, M.-T., ECKHARDT, K., & WILD, D. (1981) Mutagenicity
of cosmetics ingredients licensed by the European Communities. Mutat.
Res., 90: 91-109.
GOLDBERG, S.J., LEBOWITZ, MD., GRAVER, E.J., & HICKS, S. (1990) An
association of human congenital cardiac malformations and
drinking-water contaminants. J. Am. Coll. Cardiol., 16: 155-164.
GOSSETT, J.M. (1985) Anaerobic degradation of C1 and C2 chlorinated
hydrocarbons, Tyndall AFB, Florida, US Air Force Engineering Services
Center (Technical Report No. ESL-TR-85-38).
GOSSETT, R.W., BROWN, D.A., & YOUNG, D.R. (1983) Predicting the
bioaccumulation of organic compounds in marine organisms using
octanol/water partition coefficients. Mar. Pollut. Bull., 14: 387-392.
GOTO, M. (1979) Monitoring environmental materials and specimen
banking. State-of-the-art of Japanese experience and knowledge. In:
Monitoring environmental materials and specimen banking, Proc. Int.
Workshop, Berlin (West) 23-28 October 1978, The Hague, Nijhoff
Publishers, pp. 271-288.
GRADISKI, D., MAGADUR, J.-L., BAILLOT, M., DANIERE, M.C., & SCHUH,
M.B. (1974) Toxicité comparée des principaux solvants chlorés
aliphatiques. J. eur. Toxicol., 7: 247-254.
GRADISKI, D., BONNET, P., RAOULT, G., MAGADUR, J., & FRANCIN, J.M.
(1978) Compared acute pulmonary toxicity of the main chlorinated
aliphatic solvents. Arch. Mal. prof. Méd. Trav. Sécur. soc., 39:
249-257.
GREEN, M.H. (1981) Differential killing test using an improved
repair-deficient strain of Escherichia coli. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
Oxford, Elsevier/North Holland, pp. 183-194 (Progress in Mutation
Resarch, Vol. 1).
GRIMSRUD, E.P. & RASMUSSEN, R.A. (1975) Survey and analysis of
halocarbons in the atmosphere by gas chromatography-mass spectrometry.
Atmos. Environ., 9: 1014-1017.
HAAG, W.R. & MILL, T. (1988) Effect of a subsurface sediment on
hydrolysis of haloalkanes and epoxides. Environ. Sci. Technol., 22:
658-663.
HAAG, W.R., MILL, T., & RICHARDSON, A. (1986) Extended abstracts of
the 192nd National Meeting of the American Chemical Society, Anaheim,
Washington, DC, American Chemical Society, Division of Environmental
Chemistry, Vol. 26, pp. 248-253.
HAKE, C.L., WAGGONER, T.B., ROBERTSON, D.N., & ROWE, V.K. (1960) The
metabolism of 1,1,1-trichloroethane by the rat. Arch. environ. Health,
1: 101-105.
HALL, F.B. & HINE, C.H. (1966) Trichloroethane intoxication: a report
of two cases. J. Forensic Sci., 11: 404-413.
HARDIE, D.W.F. (1964) 1,1,1-Trichloroethane. In: Kirk, R.E. & Othmer,
D.F., ed. Encyclopedia of chemical technology, 2nd ed., New York,
Chichester, Wiley and Sons, Vol. 5, pp. 154-157, 170.
HATCH, G.G., MAMAY, P.P., AYER, M.L., CASTO, B.C., & NESNOW, S. (1983)
Chemical enhancement of viral transformation in Syrian hamster embryo
cells by gaseous volatile chlorinated methanes and ethanes. Cancer
Res., 43: 1945-1950.
HATFIELD, T.R. & MAYKOSKI, R.T. (1970) A fatal methyl chloroform
(trichloroethane) poisoning. Arch. environ. Health, 20: 279-281.
HEITMULLER, P.T., HOLLISTER, T.A., & PARRISH, P.R. (1981) Acute
toxicity of 54 industrial chemicals to sheepshead minnows (Cyprinodon
variegatus) Bull. environ. Contam. Toxicol., 27: 596-604.
HENSCHLER, D., ed. (1978) [German Association for the Encouragement of
Research: Analytical methods for the testing of materials harmful to
health. I. Air analyses], Weinheim, Verlag Chemie (in German).
HENSCHLER, D., REICHERT, D., & METZLER, M. (1980) Identification of
potential carcinogens in technical grade 1,1,1-trichloroethane. Int.
Arch. occup. environ. Health, 47: 263-268.
HERD, P.A., LIPSKY, M., & MARTIN, H.F. (1974) Cardiovascular effects
of 1,1,1-trichloroethane. Arch. environ. Health, 28: 227-233.
HOBARA, T., KOBAYASHI, H., HIGASHIHARA, E., IWAMOTO, S., KAWAMOTO, T.,
SAKAI, T., & TSUBOTA, N. (1982) [Experimental examinations and
toxicokinetic analysis of the absorption and excretion of
1,1,1-trichloroethane by the lung.] Jpn. J. ind. Health, 24: 599-607
(in Japanese).
HODGSON, M.J., HEYL, A.E., & VAN THIEL, D.H. (1989) Liver disease
associated with exposure to 1,1,1-trichloroethane. Arch. intern. Med.,
149: 1791-1798.
HORIGUCHI, S. & HORIGUCHI, K. (1971) An experiment of
1,1,1-trichloroethane vapour exposure to mice. Sangyo Igaku, 13:
226-227.
HSE (1984) 1,1,1-Trichloroethane, London, Health and Safety Executive.
HSE (1990) Guidance note EH40/90. Occupational exposure limits 1990,
London, Health and Safety Executive.
HUMBERT, B.E. & FERNANDEZ, J.G. (1977) Exposition au
1,1,1-trichloroéthane: Contribution à l'étude de l'absorption, de
l'excrétion et du métabolisme sur des sujets humains. Arch. Mal.
prof., 38: 415-425.
HUTCHINSON, T.C., HELLEBUST, J.A., TAM, D., MACKAY, D., MASCARENHAS,
R.A., & SHIU, W.Y. (1980) The correlation of the toxicity to algae of
hydrocarbons and halogenated hydrocarbons with their physical-chemical
properties. Environ. Sci. Res., 1: 577-586.
IARC (1979) 1,1,1-Trichloroethane. In: Some halogenated hydrocarbons,
Lyon, International Agency for Research on Cancer, pp. 515-532 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Vol. 20).
IARC (1987) Overall evaluations of carcinogenicity: An updating of
IARC Monographs, Volumes 1 to 42, Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluations of Carcinogenic
Risk to Humans, Suppl. 7).
ICHINOTSUBO, D., MOWER, H., & MANDEL, M. (1981) Mutagen testing of a
series of paired compounds with the Ames Salmonella testing system.
In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland, pp. 298-301
(Progress in Mutation Research. Vol. 1).
IKEDA, M. & OHTSUJI, H. (1972) A comparative study of the excretion of
Fujiwara reaction-positive substances in urine of humans and rodents
given trichloro- or tetrachloro-derivatives of ethane and ethylene.
Brit. J. ind. Med., 29: 99-104.
IVANETICH, K.M. & HONERT, L.H., VAN DEN (1981) Chloroethanes: their
metabolism by hepatic cytochrome P-450. Carcinogenesis, 2: 697-702
JAKOBSON, M., LIEPINS, M., & ERINS, P. (1980) [Effect of changes in
the reticular structure of a lignocarbohydrate matrix on the
properties of the wood. 2. Effect of cross-linking on the
hydrophilicity of birchwood.] Khim. Drev., 1: 37-46 (in Russian).
JAKOBSON, I., WAHLBERG, J.E., HOLMBERG, B., & JOHANSSON, G. (1982)
Uptake via the blood and elimination of 10 organic solvents following
epicutaneous exposure of anesthetized guinea-pigs. Toxicol. appl.
Pharmacol., 63: 181-187.
JONES, R.D. & WINTER, D.P. (1983) Two case reports of deaths on
industrial premises attributed to 1,1,1-trichloroethane. Arch.
environ. Health, 38: 59-61.
KADA, T. (1981) DNA-damaging activity of 42 coded compounds in the
rec-assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 175-182 (Progress in Mutation Research, Vol. 1).
KAJINO, M. & YAGI, M. (1980) Formation of trihalomethanes during
chlorination and determination of halogenated hydrocarbons in drinking
water. In: Hydrocarbons and halogenated hydrocarbons in the aquatic
environment, New York, London, Plenum Press.
KARLSSON, J.-E., ROSENGREN, L.E., KJELLSTRAND, P., & HAGILD K.G.
(1987) Effect of low-dose inhalation of three chlorinated aliphatic
organic solvents on DNA in gerbil brain. Scand. J. Work Environ.
Health, 13: 453-458.
KHALIL, M.A. & RASMUSSEN, R.A. (1981) Decline in the atmospheric
accumulation rates of CCl3F(F-11), CCl2F2(F-12) and CH3CCl3.
J. Air Pollut. Control Assoc., 31: 1274-1275.
KHALIL, M.A. & RASMUSSEN, R.A. (1984) Methylchloroform: Global
distribution, seasonal cycles and anthropogenic chlorine. Chemosphere,
13: 789-800.
KING, G.S., SMIALEK, J.E., & TROUTMAN, W.G. (1985) Sudden death in
adolescent resulting from the inhalation of typewriter correction
fluid. J. Am. Med. Assoc., 253: 1604-1606.
KLAASSEN, C.D. & PLAA, G.L. (1966) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in mice.
Toxicol. appl. Pharmacol., 9: 139-151.
KLAASSEN, C.D. & PLAA G.L. (1967) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in dogs.
Toxicol. appl. Pharmacol., 10: 119-131.
KLECKA, G.M., GONSIOR, S.J., & MARKHAM, D.A. (1990) Biological
transformations of 1,1,1-trichloroethane in subsurface soils and
ground water. Environ. Toxicol. Chem., 9: 1437-1451.
KLEINFELD, M. & FEINER, B. (1966) Health hazard associated with work
in confined spaces. J. occup. Med., 8: 358-364.
KOBAYASHI, H., HOBARA, T., HIROTA, H., HIGASHIHARA, E., IWAMOTO, S.,
& SAKAI, T. (1982) Sensitization of dog myocardium to epinephrine by
1,1,1-trichloroethane. Jpn. J. ind. Health, 24: 450-454.
KOBAYASHI, H., HOBARA, T., HIROTA, H., & SAKAI, T. (1983) Neural
control of blood pressure following 1,1,1-trichloroethane inhalation:
A role of sympathetic nervous system. Arch. environ. Health, 38:
93-98.
KOBAYASHI, H., HOBARA, T., SATOH, T., & SAKAI, T. (1986) Respiratory
disorders following 1,1,1-trichloroethane inhalation: A role of reflex
mechanism arising from lungs. Arch. environ. Health, 41: 149-154.
KOBAYASHI, H., HOBARA, T., KAWAMOTO, T., & SAKAI, T. (1987) Effect of
1,1,1-trichloroethane inhalation on heart rate and its mechanism: A
role of autonomic nervous system. Arch. environ. Health, 42: 140-143.
KONEMANN, H. (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part I. Relationship for 50 industrial
pollutants. Toxicology, 19: 209-221.
KRAMER, C.G., OTT, M.G., FULKERSON, J.E., & HICKS, N. (1978) Health of
workers exposed to 1,1,1-trichloroethane: A matched-pair study. Arch.
environ. Health, 33: 331-342.
KRONELD, R. (1989) Volatile pollutants in the environment and human
tissues. Bull. environ. Contam. Toxicol., 42: 873-877.
KRONEVI, T., WAHLBERG, J.E., & HOLMBERG, B. (1981) Skin pathology
following epicutaneous exposure to seven organic solvents. Int. J.
Tissue React., 3: 21-30.
KUKONGVIRIYAPAN, V., KUKONGVIRIYAPAN, U., & STACEY, N.H. (1990)
Interference with hepatocellular substrate uptake by
1,1,1-trichloroethane and tetrachloroethylene. Toxicol. appl.
Pharmacol., 102: 80-90.
KYRKLUND, T., KJELLSTRAND, P., & HAGLID, K.G. (1988) Effects of
exposure to Freon 11, 1,1,1-trichloroethane or perchloroethylene on
the lipid and fatty-acid composition in rat cerebral cortex. Scand. J.
Work Environ. Health, 14: 91-94.
LANE, R.W., RIDDLE, B.L., & BORZELLECA, J.F. (1982) Effects of
1,2-dichloroethane and 1,1,1-trichloroethane in drinking water on
reproduction and development in mice. Toxicol. appl. Pharmacol., 63:
409-421.
LEBLANC, G.A. (1980) Acute toxicity of priority pollutants to water
flea (Daphnia magna). Bull environ. Contam. Toxicol., 24: 684-691.
LEONG, B.K.J., SCHWETZ, B.A., & GEHRING, P.J. (1975) Embryo- and
fetotoxicity of inhaled trichloroethylene, perchloroethylene,
methylchloroform, and methylene chloride in mice and rats. Toxicol.
appl. Pharmacol., 33: 136 (Abstract 34).
LILLIAN, D., SINGH, H.B., APPLEBY, A., LOBBAN, L., ARNTS, R., GUMPERT,
R., HAGUE, R., TOOMEY, J., KAZASIS, J., ANTELL, M., HANSEN, D., &
SCOTT, B. (1975) Atmospheric fates of halogenated compounds. Environ.
Sci. Technol., 9: 1043-1048.
LINDBOHM, M-L., TASKINEN, H., SALLMEN, M., & HEMMINKI, K. (1990)
Spontaneous abortions among women exposed to organic solvents. Am. J.
ind. Med., 17: 449-463.
LOPRIENO, N., BARATE, R., ROSSI, A.M., FUMERO, S., MERIGGI, G.,
MONDINO, A., & SILVEST S. (1979) In vivo mutagenicity studies with
trichloroethylene and other solvents, Ivrea, Italy, Istituto di
Ricerche Biomedicale
LUNDBERG, I., EKDAHL, M., KRONEVI, T., LIDUMS, V., & LUNDBERG, S.
(1986) Relative hepatotoxicity of some industrial solvents after
intraperitoneal injection or inhalation exposure in rats. Environ.
Res., 40: 411-420.
MABEY, W., BARICH, V., & MILL, T. (1983) Extended abstracts of the
186th Meeting of the American Chemical Society, Washington, DC,
American Chemical Society, Division of Environmental Chemistry, Vol.
23, pp. 359-361.
MCCARTHY, T.B. & JONES, R.D. (1983) Industrial gassing poisonings due
to trichloroethylene, perchlorethylene, and 1,1,1-trichloroethane,
1961-80. Br. J. ind. Med., 40: 450-455.
MCCONNELL, G., FERGUSON, D.M., & PEARSON, C.R. (1975) Chlorinated
hydrocarbons and the environment. Endeavour, 34: 13-18.
MCCONNELL, J.C. & SCHIFF, H.I. (1978) Methyl chloroform: Impact on
stratospheric ozone. Science, 199: 174-177.
MCDONALD, D.J. (1981) Salmonella/microsome tests on 42 coded
chemicals. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 285-297 (Progress in Mutation Research, Vol. 1).
MCEWEN, J.D. & VERNOT, E.H. (1974) The biological effect of continuous
inhalation exposure of 1,1,1-trichloroethane (methyl chloroform) on
animals. In: Toxic hazard research unit: Annual technical report 1974,
Ohio, Wright-Patterson Air-Force Base, Aerospace Medical Research
Laboratory, pp. 81-90 (AMRL-TR-74-78).
MACKAY, C.J., CAMPBELL, L., SAMUEL, A.M., ALDERMAN, K.J., IDZIKOWSKI,
C., WILSON, H.K., & COMPERTZ, D. (1987) Behavioral changes during
exposure to 1,1,1-trichloroethane: time-course and relationship to
blood solvents levels. Am. J. ind. Med., 11: 223-239.
MCLEOD, A.A., MARJOT, A., MONAGHAN, M.J., HUGH-JONES, P., & JACKSON,
G. (1987) Chronic cardiac toxicity after inhalation of
1,1,1-trichloroethane. Br. med. J., 214: 727-729.
MCNUTT, N.S., AMSTER, R.L., MCCONNELL, E.F., & MORRIS, F (1975)
Hepatic lesions in mice after continuous inhalation exposure to
1,1,1-trichloroethane. Lab. Invest., 5: 642-654.
MAGARA, Y. & FURUICHI, T. (1986) Environmental pollution by
trichloroethylene and tetrachloroethylene: a nationwide survey. In:
Chambers, P.L., Gehring, P., & Sakai, F., ed. New concepts and
developments in toxicology, Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp. 231-243.
MALTONI, C., COTTI, G., & PATELLA, V. (1986) Results of long-term
carcinogenicity bioassays on Sprague-Dawley rats of methyl chloroform,
administered by ingestion. Acta oncol., 7: 101-117.
MARONI, M.C., BULGGHERONI, C., CASSITTO, M.G., MERLUZZI, F., GILIOLO,
R., & FOA, V. (1977) A clinical, neurophysiological and behavioral
study of female workers exposed to 1,1,1-trichloroethane. Scand. J.
Work Environ. Health, 3: 16-22.
MARJOT, R. & MCLEOD, A.A. (1989) Chronic non-neurological toxicity
from volatile substance abuse. Hum. Toxicol., 8: 301-306.
MARTIN, C.N. & MCDERMID, A.C. (1981) Testing of 42 coded compounds for
their ability to induce unscheduled DNA repair synthesis in HeLa
cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term
tests for carcinogens. Report of the international collaborative
programme, New York, Amsterdam, Oxford, Elsevier/North Holland, pp.
533-537 (Progress in Mutation Research, Vol. 1).
MIDGLEY, P.M. (1989) The production and release to the atmosphere of
1,1,1-trichloroethane (methyl chloroform). Atmos. Environ., 23:
2663-2665.
MILLER, L.J. & UHLER, A.D. (1988) Volatile halocarbons in butter:
Elevated tetrachloroethylene levels in samples obtained in close
proximity to dry-cleaning establishments. Bull. environ. Contam.
Toxicol., 41: 469-474.
MITI (1990) The yearbook of chemical trade and industries statistics,
Tokyo, Ministry of International Trade and Industry.
MITOMA, C., STEEGER, T., JACKSON, S.E., WHEELER, K.P., ROGERS, J.H.,
& MILMAN, H.A. (1985) Metabolic disposition study of chlorinated
hydrocarbons in rats and mice. Drug chem. Toxicol., 8(3): 183-194.
MONSTER, A.C. (1979) Differences in uptake, elimination and metabolism
in exposure to trichloroethylene, 1,1,1-trichloroethane and
tetrachloroethylene. Int. Arch. occup. environ. Health, 42: 311-317.
MONSTER, A.C. (1986) Biological monitoring of chlorinated hydrocarbon
solvents. J. occup. Med., 28: 583-588.
MONSTER, A.C. & HOUTKOPPER, J.M. (1979) Estimation of individual
uptake of trichloroethylene, 1,1,1-trichloroethane, and
tetrachloroethylene from biological parameters. Int. Arch. occup.
environ. Health, 42: 319-323.
MONSTER, A..C., BOERSMA, G., & STEENWEG, H. (1979) Kinetics of
1,1,1-trichloroethane in volunteers: influence of exposure
concentration and work load. Int. Arch. occup. environ. Health, 42:
293-301.
MOSER, V.C. & BALSTER, R.L. (1985) Acute motor and lethal effects of
inhaled toluene, 1,1,1-trichloroethane, halothane, and ethanol in
mice: effects of exposure duration. Toxicol. appl. Pharmacol., 77:
285-291.
MULLIN, L.S. & KRIVANEK, N.D. (1982) Comparison of unconditioned
reflex and conditioned avoidance tests in rats exposed by inhalation
to carbon monoxide, 1,1,1-trichloroethane, toluene or ethanol.
Neurotoxicology, 3: 126-137.
MYHR, B.C. & CASPARY, W.J. (1988) Evaluation of the L5178Y mouse
lymphoma cell mutagenesis assay: Intralaboratory results for
sixty-three coded chemicals tested at Litton Bionetics, Inc. Environ.
mol. Mutagen., 13: 103-194.
NAGAO, M. & TAKAHASHI, Y. (1981) Mutagenic activity of 42 coded
compounds in the salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 302-313 (Progress in Mutation
Research, Vol. 1).
NAKAAKI, K., FUKABORI, S., & TADA, O. (1980) An experimental study on
percutaneous absorption of some organic solvents. J. Sci. Labour, 56:
1-9.
NCI (1977) Bioassay of 1,1,1-trichloroethane for possible
carcinogenicity, Bethesda, Maryland, National Cancer Institute
(Carcinogenesis Technical Report Series No. 3).
NEELY, W.B. (1982) Organizing data for environmental studies. Environ.
Toxicol. Chem., 1: 259-266.
NESTMANN, E.R., LEE, E.G.H., MATULA, T.I. DOUGLAS, G.R., & MUELLER,
J.C. (1980) Mutagenicity of constituents identified in pulp and paper
mill effluents using the Salmonella/mammalian-microsome assay.
Mutat. Res., 79: 203-212.
NEUHAUSER, E.F., LOEHR, R.C., & MALECKI, M.R. (1986) Contact and
artificial soil tests using earthworms to evaluate the impact of
wastes in soil. In: Petros, J.K., Lacy, W.J., & Conway, R.A., ed.
Hazardous and industrial solid waste testing: Fourth Symposium.
Philadelphia, American Society for Testing and Materials, pp. 192-203
(ASTM STP 886).
NIEF, L. & MERCIER, J. (1981) Skin pathology following epicutaneous
exposure to seven organic solvents. Int. J. Tissue React., 111: 21-30.
NILSSON, K.B. (1986a) Effects of 1,1,1-trichloroethane on the cGMP
metabolism in mouse brain. Acta pharmacol. toxicol., 58: 318-326.
NILSSON, K.B. (1986b) Actions of 1,1,1-trichloroethane on the cAMP
metabolism in mouse brain. Acta pharmacol. toxicol., 59: 362-369.
NILSSON, K.B. (1987) Effects of 1,1,1-trichloroethane on synaptosomal
calcium accumulation in mouse brain. Pharmacol. Toxicol., 61: 215-219.
NIOSH (1976) Criteria for a recommended standard: occupational
exposure to 1,1,1-trichloroethane (methyl chloroform), Cincinnati,
Ohio, National Institute for Occupational Safety and Health
(Publication DHEW (NIOSH) No. 76-184).
NIOSH (1977) NIOSH manual of analytical methods. Part II: Standards
completion program validated methods (Method 5328), 2nd ed.,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, pp. 5328/1-5328/9 (DHEW (NIOSH) Publication No. 77-157-B).
NIOSH (1979) Occupational exposure to 1,1,1-trichloroethane,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health.
NIOSH (1983) National occupational exposure survey, 1981-1983,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, Division of Surveillance, Hazard Evaluations and Field
Studies, Surveillance Branch, Hazard Section (Unpublished data).
NOLAN, R.J., FRESHOUR, N.L., RICH, D.L., MCCARTY, L.P., & SAUNDERS,
J.H. (1984) Kinetics and metabolism of inhaled methyl chloroform
(1,1,1-trichloroethane) in male volunteers. Fundam. appl. Toxicol., 4:
654-662.
NORTHFIELD, R.R. (1981) Avoidable deaths due to acute exposure to
1,1,1-trichloroethane. J. Soc. Occup. Med., 31: 164-166.
NRC (1976) Halocarbons. Effects on stratospheric ozone, Washington,
DC, National Research Council, Panel on Atmospheric Chemistry. OTSON,
R. (1987) Purgeable organics in Great Lakes raw and treated water.
Int. J. environ. anal. Chem., 31: 41-53.
OTSON, R. & CHAN, C. (1987) Sample handling and analysis for 51
volatile organics by an adapted purge and trap GC-MS technique. Int.
J. environ. anal. Chem., 30: 275-287.
OTSON, R., WILLIAMS, D.T., & BOTHWELL, P.D. (1984) Fabric protectors.
Part II - Propane, 1,1,1-trichloroethane and petroleum distillates in
air after application of fabric protectors. Am. Ind. Hyg. Assoc. J.,
45: 28-33.
PARSONS, F. & LAGE, G.B. (1985) Chlorinated organics in simulated
groundwater environments. J. Am. Water Works Assoc., 77: 52-59.
PARSONS, F., LAGE, G.B., & RICE, R. (1985) Biotransformation of
chlorinated organic solvents in static microcosms. Environ. Toxicol.
Chem., 4: 739-742.
PEARSON, C.R. (1982) C1 and C2 halocarbons. In: Hutzinger, O., ed.
The handbook of environmental chemistry, Berlin, Heidelberg, New York,
Springer-Verlag, Vol. 3, Part B, pp. 69-88.
PEARSON, C.R. & MCCONNELL, G. (1975) Chlorinated C1 and C2
hydrocarbons in the marine environment. Proc. R. Soc. Lond., B189:
305-332.
PEKARI, K. & AITIO, A. (1985) Method 26. Determination of
1,1,1-trichloroethane in blood. In: Fishbein, L. & O'Neil, I.K., ed.
Environmental carcinogens selected methods of analysis. Volume 7 -
Some volatile halogenated hydrocarbons, Lyon, International Agency for
Research on Cancer, pp. 445-450 (IARC Scientific Publications No. 68).
PELLIZARI, E.D., HARTWELL, T.D., HARRIS, B.S.H., WADDELL, R.D.,
WHITAKER, D.A., & ERICKSON, M.D. (1982) Purgeable organic compounds in
mothers' milk. Bull. environ. Contam. Toxicol., 28: 322-328.
PEREIRA, W.E. & HUGHES, B.A. (1980) Determination of selected volatile
organic priority pollutants in water by computerized gas
chromatography - quadruple mass spectrometry. J. Am. Water Assoc., 72:
220-230.
PERRY, P.E. & THOMSON, E.J. (1981) Evaluation of the sister-chromatid
exchange method in mammalian cells as a screening system for
carcinogens. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 560-569 (Progress in Mutation Research, Vol. 1).
PFANNHAUSER, W., GOMBOS, J., & THALLER, A. (1988) [Halogenated
aliphatic hydrocarbons in food: Investigations on enrichment and
content in fat containing food.] Ernährung, 12: 611-615 (in German).
PIET, G.J., SLINGERLAND, P., DE GRUNT, F.E., HEUVEL, V.D., & ZOETEMAN,
B.C.J. (1978) Determination of very volatile halogenated organic
compounds in water by means of direct head-space analysis. Anal.
Lett., 11: 437-448.
PRENDERGAST, J.A., JONES, R.A., JENKINS, L.J., & SIEGEL, J. (1967)
Effects on experimental animals of long-term inhalation of
trichloroethylene, carbon tetrachloride, 1,1,1-trichloroethane,
dichlorodifluoromethane, and 1,1-dichloroethylene. Toxicol. appl.
Pharmacol., 10: 270-289.
PRICE, P.J., HASSETT, C.M., & MANSFIELD, J.I. (1978) Transforming
activities of trichloroethylene and proposed industrial alternatives.
14: 290-293.
PRINN, R., CUNNOLD, D., RASMUSSEN, R., SIMMONDS, P., ALYEA, F.,
CRAWFORD, A., FRASER, P., & ROSEN, R. (1987) Atmospheric trends in
methylchloroform and the global average for the hydroxyl radical.
Science, 238: 945-950.
PRODI, G., COLLACCI, A., GRILLI, S., LATTANZI, G., MAZZULO, M., &
TURINA P. (1988) Comparison of the covalent binding of various
chloroethanes with nucleic acids. In: Feo, F., ed. Chemical
carcinogenesis (models and mechanisms), New York, Plenum Press, pp.
93-102.
QUAST, J.F., RAMPY, L.W., BALMER, M.F., LEONG, B.D.J., & GEHRING P.J.
(1978) Toxicological and carcinogenic evaluation of a
1,1,1-trichloroethane formulation by chronic inhalation in rats,
Midland, Michigan, Dow Chemical Co.
QUAST, J.F., CALHOUN, L.L., & FRAUSON, L.E. (1988)
1,1,1-Trichloroethane formulation: a chronic inhalation toxicity and
oncogenicity study in Fischer 344 rats and B6C3F1 mice. Fundam. appl.
Toxicol., 11: 611-625.
RAJENDRAN, S. (1990) The toxicity of phosphine, methyl bromide,
1,1,1-trichloroethane and carbon dioxide alone and as mixtures to the
pupae of red flour beetle, Tribolium castaneum, Herbst. Pestic.
Sci., 29: 75-83.
RAMPY, L.W., QUAST, J.F., LEONG, B.K.J., & GEHRING, P.J. (1977)
Results of long-term inhalation studies on rats of
1,1,1-trichloroethane and perchloroethylene formulations. In: Plaa,
G.L. & Duncan, W.A.M., ed. Proceedings of the 1st International
Congress on Toxicology, Toronto, New York, London, Academic Press, p.
27 (Abstract).
RASMUSSEN, R.A. & KHALIL, M.A.K. (1982) Latitudinal distributions of
trace gases in and above the boundary layer. Chemosphere, 11: 117-235.
RASMUSSEN, R.A. & KHALIL, M.A.K. (1983) Natural and anthropogenic
trace gases in and above the boundary layer. Chemosphere, 12: 371-375.
RASMUSSEN, R.A., KHALIL, M.A.K., & DALLAGUE, R.W. (1981) Atmospheric
trace gases in Antarctica. Science, 211: 185-287.
REINHARDT, C.F., MULLIN, L.S., & MAXFIELD, M.E. (1973)
Epinephrine-induced cardiac arrhythmia potential of some common
industrial solvents. J. occup. Med., 15: 953-955.
REITZ, R.H., MCDOUGAL, J.N., HIMMELSTEIN, M.W., NOLAN, R.J., &
SCHUMANN, A.M. (1988) Physiologically based pharmacokinetic modelling
with methylchloroform: Implications for interspecies, high dose/low
dose and dose route extrapolations. Toxicol. appl. Pharmacol., 95:
185-199.
RENNICK, R.B., MALTON, S.D., MOW, G.K., & SEEVERS, M.H. (1949)
Induction of idioventricular rhythms by 1,1,1-trichloroethane and
epinephrine. J. Pharmacol. exp. Ther., 94: 327.
RICHOLD, M. & JONES, E. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluations of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 314-322 (Progress in Mutation
Research, Vol. 1).
RIIHIMAKI, V. & PFAFFLI, P (1978) Percutaneous absorption of solvent
vapor in man. Scand. J. Work Environ. Health, 4: 73-85.
RIVETT, M.O., LERNER, D.N., & LLOYD, J.W. (1990) Chlorinated solvents
in U.K. aquifers. J. Inst. Water Environ. Manage., 4: 242-250.
ROSENGREN, L.E., AURELL, A., KJELLSTRAND, P., & HAGLID, K.G. (1985)
Astrogliosis in the cerebral cortex of gerbils after long-term
exposure to 1,1,1-trichloroethane. Scand. J. Work Environ. Health, 11:
447-455.
ROSENKRANZ, H.S., HYMAN, J., & LEIFER, Z. (1981) DNA polymerase
deficient assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 210-218 (Progress in Mutation Research, Vol. 1).
ROWLAND, I. & SEVERN, B. (1981) Mutagenicity of carcinogens and
noncarcinogens in the Salmonella/microsome test. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 323-332 (Progress in
Mutation Research, Vol. 1).
RUSSELL, J.W. & SHADOFF, L.A. (1977) The sampling and determination of
halocarbons in ambient air using concentration on porous polymer. J.
Chromatogr., 134: 375-384.
SALAMONE, M.F., HEDDLE, J.A., & KATZ M. (1981) Mutagenic activity of
41 compounds in the in vivo micronucleus assay. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 686-697 (Progress in
Mutation Research, Vol. 1).
SALVINI, M., BINASCHI, S., & RIVA, M. (1971) Evaluation of the
psychophysiological functions in humans exposed to the "threshold
limit value" of 1,1,1-trichloroethane. Br. J. ind. Med., 28: 286-292.
SATO, A., NAKAJIMA, T., & KOYAMA, Y. (1980) Effects of chronic ethanol
consumption on hepatic metabolism of aromatic and chlorinated
hydrocarbons in rats. Br. J. ind. Med., 37: 382-386.
SAVOLAINEN, H., PFAFFLI, P., TENGEN, M., & VAINIO, H. (1977)
Trichloroethylene and 1,1,1-trichloroethane: effects on brain and
liver activity after five days intermittent inhalation. Arch.
Toxicol., 38: 229-237.
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1981)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Int. Arch. occup. environ. Health, 49: 89-98.
SAVOLAINEN, K., RIIHIMÄKI, V., & LAINE A. (1982a) Biphasic effects of
inhaled solvents on human equilibrium. Acta pharmacol. toxicol., 51:
237-242
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1982b)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Arch. Toxicol., Suppl. 5: 96-99.
SCHUMANN, A.M., FOX, T.R., & WATANABE, P.G. (1982) [14C]-Methyl
chloroform (1,1,1-trichloroethane): Pharmacokinetics in rats and mice
following inhalation exposure. Toxicol. appl. Pharmacol., 62: 390-401.
SEKI, Y., URASHIMA, Y., AIKAWA, H., MATSUMURA, H., ICHIKAWA, Y.,
HIRATSUKA, F., YOSHIOKA, Y., SHIMBO, S., & IKEDA, M. (1975)
Trichloro-compounds in the urine of humans exposed to methyl
chloroform at sub-threshold levels. Int. Arch. Arbeitsmed., 34: 39-49.
SHIMADA, T., SWANSON, A.F., LEBER, P., & WILLIAMS, G.M. (1985)
Activities of chlorinated ethane and ethylene compounds in the
Salmonella/rat microsome mutagenesis and rat hepatocyte/DNA repair
assays under vapor phase exposure conditions. Cell Biol. Toxicol., 1:
159-179.
SHMUTER, L.M. (1977) [The effect of chronic exposure to low
concentrations of chlorinated ethane series hydrocarbons on specific
and non-specific immunological response in animals under experimental
conditions.] Gig. Tr. prof. Zabol., 8: 38-43 (in Russian).
SIEGEL, J., JONES, R.A., COON, R.A., & LYON J.P. (1971) Effects on
experimental animals of acute, repeated and continuous inhalation
exposures to dichloroacetylene mixtures. Toxicol. appl. Pharmacol.,
18: 168-174.
SIMMON, V.F. & SHEPHERD, G.F. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 333-342 (Progress in Mutation
Research, Vol. 1).
SIMMON, V.F., KAUHANEN, K., & TARDIFF, R.G. (1977) Mutagenic activity
of chemicals identified in drinking water. In: Scott, D., Bridges,
B.A., & Sobels, F.H., ed. Progress in genetic toxicology, Amsterdam,
Elsevier/North Holland, pp. 249-258.
SIMMONDS, P.G., KERRIN, S.L., LOVELOCK, J.E., & SHAIR, F.H. (1974)
Distribution of atmospheric halocarbons in the air over the Los
Angeles basin. Atmos. Environ., 8: 209-216.
SINGH, H.B., SALAS, L.J., & STILES, R.E. (1982) Distribution of
selected gaseous organic mutagens and suspect carcinogens in ambient
air. Environ. Sci. Technol., 16: 982-880.
SNOW, L.P., MCNAIR, B.C., & CASTRO B.C. (1979) Mutagenesis testing of
1,1,1-trichloroethane in Salmonella strains TA100 and TA98, Research
Triangle Park, North Carolina, Northrop Services Inc. (Unpublished
report prepared for the US Environmental Protection Agency).
STAHL, C.J. FATTEH, A.V., & DOMINGUEZ, A.M. (1969) Trichloroethane
poisoning: observations on the pathology and toxicology in six fatal
cases. J. Forensic. Sci., 14: 393-397.
STEWART, R.D. & ANDREWS J.T. (1966) Acute intoxication with methyl
chloroform. J. Am. Med. Assoc., 195: 904-906.
STEWART, R.D. (1968) The toxicology of 1,1,1-trichloroethane. Ann.
occup. Hyg., 11: 71-79.
STEWART, R.D. (1971) Methyl chloroform intoxication. J. Am. Med.
Assoc., 215: 1789-1792.
STEWART, R.D. & DODD, H.D. (1964) Absorption of carbon tetrachloride,
trichloroethylene, tetrachloroethylene, methylene chloride and
1,1,1-trichloroethane through the human skin. Am. Ind. Hyg. Assoc. J.,
25: 439-446.
STEWART, R.D., GAY, H.H., ERLEY, D.S., HAKE, C.L., & SCHAFFER, A.W.
(1961) Human exposure to 1,1,1-trichloroethane vapour: relationship of
expired air and blood concentrations. Am. Ind. Hyg. Assoc. J., 22:
252-262.
STEWART, R.D., GAY, H.H., SCHAFFER, A.W., ERLEY, D.S., & ROWE, V.K.
(1969) Experimental human exposure to methyl chloroform vapor. Arch.
environ. Health, 19: 467-472.
STYLES, J.A. (1981) Activity of 42 coded compounds in the BHK-21 cell
transformation test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 638-646 (Progress in Mutation Research, Vol. 1).
THIELE, D.L., EIGENBRODT, E.H., & WARE, A.J. (1982) Cirrhosis after
repeated trichloroethylene and 1,1,1-trichloroethane exposure.
Gastroenterology, 83: 926-929.
THOMPSON, R.S. & CARMICHAEL, N.G. (1989) 1,1,1-Trichloroethane:
Medium-term toxicity to carp, daphnids, and higher plants. Ecotoxicol.
environ. Saf., 17: 172-182.
TOFTGAARD, R., NILSEN, O.G., & GUSTAFSSON, J.A. (1981) Changes in rat
liver microsomal cytochrome P-450 and enzymatic activities after the
inhalation of n-hexane, xylene, methyl ethyl ketone and
methylchloroform for four weeks. Scand. J. Work Environ. Health, 7:
31-37.
TOPHAM, J.C. (1980) Do induced sperm-head abnormalities in mice
specifically identify mammalian mutagens rather than carcinogens?
Mutat. Res., 74: 379-387.
TORKELSON, T.R., OYEN, F., MCCOLLISTER, D.D., & ROWE V.K. (1958)
Toxicity of 1,1,1-trichloroethane as determined on laboratory animals
and human subjects. Am. Ind. Hyg. Assoc. J., 19: 353-362.
TORSLOV, J. (1988) Hazard assessment of 1,1,1-trichloroethane,
Copenhagen, Denmark, Ministry of the Environment, National Agency of
Environmental Protection, 67 pp (Miljoproject, No. 10).
TRAVIS, C.C., HATTEMER-FREY, H.A., & ARMS, A.D. (1988) Relationship
between dietary intake of organic chemicals and their concentrations
in human adipose tissue and breast milk. Arch. environ. Contam.
Toxicol., 17: 473-478.
TSUCHIMOTO, T. & MATTER, B.E. (1981) Activity of coded compounds in
the micronucleus test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 705-711 (Progress in Mutation Research, Vol. 1). TSURUTA,
H. (1975) Percutaneous absorption of organic solvents. I. Comparative
study of the in vivo percutaneous absorption of chlorinated solvents
in mice. Ind. Health, 13: 227-236.
TU, A.S., MURRAY, T.A., HATCH, K.M., SIVAK, A., & MILMAN, H.A. (1985)
In vitro transformation of balb/c-3T3 cells by chlorinated ethanes
and ethylenes. Cancer Lett., 28: 85-92.
TWEATS, D.J. (1981) Activity of 42 coded compounds in a differential
killing test using Escherichia coli strains WP2, WP67 (uvrA polA),
and CM871 (uvrA lexA recA). In: de Serres, F.J. & Ashby, J., ed.
Evaluation of short-term tests for carcinogens. Report of the
international collaborative programme, New York, Amsterdam, London,
Elsevier/North Holland, pp. 199-209 (Progress in Mutation Research,
Vol. 1).
UHLER, A.D. & DIACHENKO, G.W. (1987) Volatile halocarbon compounds in
process water and processed foods. Bull. environ. Contam. Toxicol.,
39: 601-607.
UNEP (1989) Technical progress on protecting the ozone layer. Report
of the Technology Review Panel, Nairobi, United Nations Environment
Programme, 103 pp.
URANO, K. (1985) Usage and pollution control of chlorinated organic
compounds. Jpn. J. Water Pollut. Res., 8: 269-273.
URANO, K., KAWAMOTO, K., ABE, Y., & OTAKE, M. (1988) Chlorinated
organic compounds in urban air in Japan. Sci. total Environ., 74:
121-131.
US EPA (1977) Environmental monitoring near industrial areas -
Methylchloroform, Washington, DC, US Environmental Protection Agency
(EPA-560/6-77-025).
US EPA (1980a) Proceedings of the Conference on Methyl Chloroform and
Other Halocarbon Pollutants, Washington, DC, US Environmental
Protection Agency (EPA-600/9-80-003).
US EPA (1980b) Ambient water quality criteria for chlorinated ethanes,
Washington, DC, US Environmental Protection Agency (EPA-440/5-80-029).
US EPA (1984) Health assessment document for 1,1,1-trichloroethane
(methyl chloroform), Washington, DC, US Environmental Protection
Agency (EPA-600/8-82-003F).
VAN BEEK, L. (1990) Investigation of a possibility to reduce the
number of rabbits in skin irritation tests; experiments with
dichloromethane, trichloromethylene, tetrachloroethylene and
1,1,1-trichloroethane (Project No. B 86-0927) Apeldoorn, The
Netherlands, Organization for Applied Scientific Research (TNO Report
No. V 89.265).
VEITH, G.D., MACEK, K.J. PETROCELLI, S.R., & CARROLL, J. (1980) An
evaluation of using partition coefficients and water solubility to
estimate bioconcentration factors for organic chemicals in fish. In:
Eaton, J.G., Parrish, P., & Hendricks, A.C., ed. Aquatic toxicology.
Proceedings of the Third Annual Symposium on Aquatic Toxicology,
Philadelphia, American Society for Testing and Materials, pp. 116-129
(ASTM STP No. 707).
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., WHITMORE, R.
SPARACINO, C., & ZELON, H. (1986) Total exposure assessment
methodology (TEAM) study: personal exposures, indoor-outdoor
relationships, and breath levels of volatile organic compounds in New
Jersey. Environ. Int., 12: 369-387.
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., SPARACINO, C.,
WHITMORE, R., SHELDON, L., ZELON, H., & PERRITT, R. (1987) The TEAM
study: Personal exposure to substances in air, drinking water, and
breath of 400 residents of New Jersey, North Carolina and North
Dakota. Environ. Res., 43: 290-307.
WALRAEVENS, R., TROUILLET, P., & DEVOS, A. (1974) Basic elimination of
HCI from chlorinated ethanes. Int. J. chem. Kinet., VI: 777-786.
WEAST, R.C. (1986) CRC handbook of chemistry and physics, 67th ed,
Boca Raton, Florida, CRC Press.
WESTERBERG, E. & LARSSON, L. (1982) Use of automated head-space gas
chromatography for determination of 1,1,1-trichloroethane in rat blood
and brain tissue. Int. J. environ. anal. Chem., 12: 233-239.
WILLIAMS, G.M. (1983) DNA repair tests of 11 chlorinated hydrocarbon
analogs, Washington, DC, US Environmental Protection Agency (Draft
Final Report TR-507-18).
WILSON, B.H. & WHITE, M.V. (1986) A fixed-film bioreactor to treat
trichloroethylene-laden waters from interdiction wells. In:
Proceedings of the 1986 National Symposium and Exposition on Aquifer
Restoration and Ground Water Monitoring, Columbus, Ohio, 19-22 May,
Dublin, Ohio, National Well Water Association, pp. 425-436.
WILSON, J.T., MCNABB, J.F., WILSON, B.H., & NOONAN, M.J. (1983)
Biotransformation of selected organic pollutants in ground water. Dev.
ind. Microbiol., 24: 125-233.
WHO (in preparation) Health and Safety Guide for
1,1,1-trichloroethane, Geneva, World Health Organization.
WINDHOLZ, M. ed. (1983) The Merck index: An encyclopedia of chemicals,
drugs and biologicals, 10th ed., Rahway, New Jersey, Merck and Co.,
Inc.
WMO (1982) Global ozone research and monitoring project: Report No.
11. The stratosphere 1981: Theory and measurements, Geneva,
Switzerland, World Meteorological Organization.
WOOLVERTON, W.L. & BALSTER, R.L. (1981) Behavioral and lethal effects
of combinations of oral ethanol and inhaled 1,1,1-trichloroethane in
mice. Toxicol. appl. Pharmacol., 59: 1-7.
WRIGHT, M.F. & STROBL, D.J. (1984) 1,1,1-Trichloroethane cardiac
toxicity: Report of a case. J. Am. Osteopath. Assoc., 84: 285-288.
WRIGHTSON, I. & SANTON, R.C. (1988) An explosion involving
1,1,1-trichloroethane. Loss Prev. Bull., 083: 21-23.
YORK, R.G., SOWRY, B.M., HASTINGS, L., & MANSON, J.M. (1982)
Evaluation of teratogenicity and neurotoxicity with maternal
inhalation exposure to methylchloroform. J. Toxicol. environ. Health,
9: 251-266.
YUNG, Y.L., MCELROY, M.B., & WOFSY, S.C. (1975) Atmospheric
halocarbons: A discussion with emphasis on chloroform. Geophys. Res.
Lett., 2: 397-399.
RESUME
Le 1,1,1-trichloréthane est un hydrocarbure chloré produit par
chloration du chlorure de vinyle ou du chlorure de vinylidène. En
1988, la production mondiale était d'environ 680 000 tonnes. C'est un
liquide incolore et volatil à l'odeur caractéristique dont la vapeur
est plus dense que l'air. On l'utilise essentiellement pour le
dégraissage des surfaces métalliques et comme solvant dans un grand
nombre de produits industriels et de produits de consom-mation,
notamment des adhésifs, des détachants et des bombes aérosols. Il
sert également d'intermédiaire en synthèse chimique. Le
trichloréthane technique contient généralement 3 à 8% de stabilisants
destinés à empêcher sa décomposition et la formation d'acide
chlorhydrique; les parties métalliques sont ainsi protégées de la
corrosion. Il est ininflammable dans les conditions normales mais la
vapeur s'enflamme à haute température et, au cours du soudage, elle
peut se décomposer en libérant du phosgène, un gaz toxique. Des
réactions extrêmement violentes peuvent se produire en cas de contact
avec de l'aluminium, du magnésium ou leurs alliages.
Le 1,1,1-trichloréthane pénètre facilement dans l'environne-ment.
Comme il séjourne longtemps dans la troposphère (environ six ans) et
qu'il n'est que lentement biodégradable, on le retrouve maintenant
partout dans l'environnement, même à grande distance des zones
industrielles. A proximité d'installations industrielles où ce
composé est produit ou manipulé, on a trouvé du 1,1,1-trichloréthane
à des concentrations atteignant 86 µg/m3 (16 000 ppm, p/p) dans des
échantillons d'air.
Le trichloréthane se déplace dans le sol et parvient jusqu'aux
eaux souterraines. On en a trouvé à des concentrations atteignant
1600 µg/litre dans les eaux souterraines ou les eaux de surface. Ce
phénomène peut être à l'origine de la contamination des réserves d'eau
potable.
On estime que 15% des émissions annuelles de
1,1,1-trichloré-thane sont entraînées dans la stratosphère où elles
peuvent endommager la couche d'ozone par libération d'atomes de
chlore.
Des essais biologiques sur des crustacés et des poissons à des
concentrations supérieures à 7 mg/litre ont révélé des effets toxiques
aigus. On possède quelques données selon lesquelles le composé ne
s'accumulerait que faiblement chez les organismes aquatiques. En
raison du caractère limité des données disponibles, il est difficile
d'évaluer les effets qu'il peut avoir sur les organismes terrestres.
L'exposition humaine au 1,1,1-trichloréthane est due
principalement à l'inhalation de ce composé qui est ensuite rapidement
résorbé par l'organisme. Il peut y avoir également exposition par
contact cutané ou ingestion. Le trichloréthane se répartit largement
dans les tissus de l'organisme et il traverse la barrière
hématoencéphalique et placentaire. On le retrouve également dans le
lait humain mais il ne semble pas qu'il s'accumule dans les tissus.
Sa principale voie d'élimination est l'exhalation du composé non
métabolisé.
La toxicité aiguë et chronique du 1,1,1-trichloréthane est
relativement faible mais en cas de forte exposition, le risque
d'intoxication est réel. Cela peut se produire en cas d'exposition
professionnelle, d'accidents ou encore de toxicomanie aux solvants.
Comme il s'agit d'un solvant volatil dont la vapeur est beaucoup plus
dense que l'air, il peut se trouver à des concentrations
inhabituellement fortes et dangereuses dans des espaces confinés comme
les réservoirs "vides". Ce genre de situation a donné lieu à
plusieurs intoxications mortelles ou qui auraient pu l'être sur les
lieux de travail ou ailleurs.
Chez l'homme, l'effet critique s'exerce au niveau du système
nerveux central. On observe des effets qui vont de légers troubles du
comportement (accompagnés d'une légère irritation occulaire) à la dose
de 1,9 g/m3 (350 ppm), jusqu'à l'inconscience et à l'arrêt
respiratoire lorsque la concentration est élevée. Dans certains cas,
des anomalies cardiaques mortelles peuvent se produire. Le
trichloréthane est moins toxique pour le foie que la plupart des
autres solvants organiques chlorés. La dose sans effet observable
pour l'homme se situe autour de 1,35 g/m3 (250 ppm).
Aucune étude satisfaisante n'a été publiée sur les effets
cancérogènes de ce solvant chez l'homme. Toutefois une étude par
inhalation de longue durée sur des rats et des souris exposés à 8,1
g/m3 (1500 ppm) n'a pas révélé le moindre effet cancérogène. Le
1,1,1-trichloréthane ne présente pas d'activité génotoxique
significative.
Chez des rats et des lapins exposés au trichloréthane à des
concentrations qui étaient toxiques pour les femelles gestantes, on
n'a pas noté d'effets tératogènes, malgré la présence d'effets
toxiques sur le développement. Les données épidémiologiques relatives
à ces effets sur la reproduction sont trop limitées pour permettre
d'en tirer une conclusion.
RESUMEN
El 1,1,1-tricloroetano es un hidrocarburo clorado que se fabrica
a partir del cloruro de vinilo o del cloruro de vinilideno por
cloración. En 1988 la producción mundial fue de unas 680 000
toneladas. Es un líquido volátil e incoloro con un olor
característico, y su vapor es más denso que el aire. Se utiliza
principalmente en el desengrasado de metales y como disolvente en
muchos productos industriales y de consumo, como adhesivos,
quitamanchas y envases de aerosoles. Es también un producto químico
intermediario. El tricloroetano de calidad técnica suele contener del
3% al 8% de estabilizadores que impiden la degradación y la formación
de ácido clorhídrico a fin de proteger las partes metálicas de la
corrosión. En condiciones normales no es inflamable, pero su vapor
arde a altas temperaturas y, durante las operaciones de soldadura, su
degradación produce fosgeno, un gas tóxico. Cuando entra en contacto
con el aluminio, el magnesio y sus aleaciones puede provocar
reacciones muy violentas.
El 1,1,1-tricloroetano llega fácilmente al medio ambiente. Su
prolongada permanencia en la troposfera (unos seis años) y baja
biodegradabilidad son razón de su actual omnipresencia en el medio
ambiente, incluso lejos de las zonas industriales. En muestras de
aire de zonas próximas a industrias que lo fabrican o lo manipulan se
han detectado concentraciones de hasta 86 µg/m3 (16 ppb, peso/peso).
El tricloroetano se desplaza en los suelos y alcanza las aguas
subterráneas. Se han encontrado concentraciones de hasta 1600
µg/litro en aguas subterráneas o superficiales. Esta puede ser una
fuente de contaminación para suministros de agua potable.
Se estima que el 15% del volumen anual liberado llega a la
estratosfera, donde libera átomos de cloro que contribuyen a la
destrucción del ozono.
En bioensayos con crustáceos y peces se han observado efectos
tóxicos agudos con concentraciones superiores a 7 mg/litro. Limitada
información limitada disponible parece indicar que la bioacumulación
en los organismos acuáticos es baja. La escasez de datos dificulta la
evaluación de los posibles efectos en los organismos terrestres.
El ser humano está expuesto al 1,1,1-tricloroetano principalmente
por inhalación, y luego el organismo absorbe rápidamente la sustancia.
También se puede dar la exposición por contacto cutáneo o ingestión.
El tricloroetano se distribuye ampliamente por los tejidos y atraviesa
las barreras hematoencefálica y placentaria. Aunque también se ha
encontrado en la leche humana, no parece que se produzca
bioacumulación. La principal vía de eliminación es la exhalación del
compuesto inalterado.
Las toxicidades aguda y crónica del 1,1,1-tricloroetano son
relativamente bajas, pero en condiciones de exposición intensa pueden
producirse efectos tóxicos. Estas condiciones se pueden presentar en
casos de exposición profesional, de abuso del disolvente o de
accidentes. Dado que el disolvente es volátil y el vapor es mucho más
denso que el aire, se pueden producir concentraciones inesperadamente
elevadas y peligrosas en espacios reducidos, como depósitos de
almacenamiento "vacíos". Así, se han producido algunas intoxicaciones
mortales y casi mortales en lugares de trabajo y otros.
El efecto más importante en el ser humano se produce en el
sistema nervioso central. Los efectos observables van desde ligeros
cambios de comportamiento (acompañados de una leve irritación ocular)
con 1,9 g/m3 (350 ppm) hasta la pérdida del conocimiento y el paro
respiratorio con concentraciones más elevadas. Sin embargo, también
se pueden producir anomalías cardíacas fatales. La toxicidad hepática
del tricloroetano es inferior a la de la mayoría de los disolventes
organoclorados. El nivel sin efecto observado (NOEL) para la especie
humana parece ser del orden de 1,35 g/m3 (250 ppm).
No se han publicado estudios adecuados sobre los efectos
carcinogénicos en la especie humana. Sin embargo, en los estudios de
inhalación prolongada en ratas y ratones expuestos a 8,1 g/m3 (1500
ppm) no se obtuvieron pruebas de ningún efecto carcinogénico. El
1,1,1-tricloroetano no tiene un potencial genotóxico importante.
En ratas y conejos se ha observado toxicidad en la fase de
crecimiento, pero no teratogenicidad, con concentraciones que fueron
tóxicas para las madres. Las escasas pruebas epidemiológicas en
relación con sus efectos sobre la reproducción no son concluyentes.
body weight. Both trichloroethanol and trichloroacetic acid were
identified as metabolites in the urine, trichloroethanol being
excreted (as the glucuronide) much faster than trichloroacetic acid
(Eben & Kimberle 1974).
Following a single inhalation exposure of rats to 810 and
8100 mg/m3 (150 and 1500 ppm), only a 2-4 times increase in excreted
metabolites was found between the two doses, suggesting metabolic
saturation. Repeated exposure to 8100 mg/m3 over 16 months did not
change the amount of 1,1,1-trichloroethane metabolites. Similar
results were obtained in mice. In both rats and mice, urine
metabolites accounted for 40-70% of the total amount metabolized.
Overall, mice were found to biotransform approximately 5 times more
1,1,1-trichloroethane (per kg body weight) than rats. In rats and
mice an age-related increase in the amount metabolized was observed in
aged animals as opposed to young adults (Schumann et al., 1982).
6.3.2 Human studies
The average amount of metabolites excreted in the urine of humans
(workers or volunteers) exposed to 1,1,1-trichloroethane in the air
(22-1890 mg/m3, 4-350 ppm) was variable, but was typically between
3 and 7% of the absorbed dose. The ratio of the metabolites
trichloroethanol glucuronide and trichloroacetic acid was about 2 to
1, but this increased with increasing exposure concentration (Seki et
al., 1975; Humbert & Fernandez, 1977; Monster et al., 1979; Nolan et
al., 1984).
6.3.3 Metabolic interactions
Simultaneous exposure to other solvents tends to increase the
retention and decrease the metabolism of 1,1,1-trichloroethane
(Savolainen et al., 1981). 1,1,1-Trichloroethane metabolism was
accelerated in rats pre-treated with ethanol because of the induction
of metabolic enzymes (Sato et al., 1980).
6.4 Elimination
Regardless of the route of administration, the main excretory
route for 1,1,1-trichloroethane is exhalation via the lungs. This may
be explained by the relatively low solubility in blood.
6.4.1 Animal studies
In rats and mice, 55% to 98% of the 1,1,1-trichloroethane was
excreted unchanged in expired air after oral or intraperitoneal
exposure (Hake et al., 1960; Reitz et al., 1988).
Following inhalation exposure, 94-98% of the absorbed
1,1,1-trichloroethane was excreted unchanged in the expired air of
rats during 72 h. In mice, the corresponding figure was 87-97%. The
rate of elimination was somewhat higher in mice; 85% was eliminated
during the first 3 h compared to 65% in rats (Schumann et al., 1982).
A total of about 1-8% of an absorbed dose in rodents is excreted
in the urine (Hake et al., 1960; Schumann et al., 1982).
After exposure to 1,1,1-trichloroethane vapour, the level in
blood plasma decreases rapidly in a diphasic or triphasic manner,
depending on the exposure level. Following exposure to 810 mg/m3
(150 ppm), the half-lives were 10 and 139 min, whereas with 8100
mg/m3 (1500 ppm), the half-lives were 36 and 238 min. In mice the
half-lives were a little shorter (Schumann et al., 1982).
6.4.2 Human studies
Studies with human volunteers show that over 90% of the absorbed
trichloroethane is excreted unchanged in the expired air. Only minor
parts (5-7%) of the absorbed solvent are excreted in the urine (as
trichloroethanol glucuronide and trichloroacetic acid) (Stewart et
al., 1969; Humbert & Fernadez, 1977; Nolan et al., 1984). The main
product was found to be 2,2,2-trichloroethanol glucuronide, the
excretion of which was completed within 8 days. The secondary product
was trichloroacetic acid; its excretion occurred somewhat later and
was completed within 12 days (Humbert & Fernandez, 1977).
The elimination of 1,1,1-trichloroethane, measured as plasma
concentration and concentration in expired air, in six human
volunteers exposed to 191 or 1911 mg/m3 (35 or 350 ppm) for 6 h
could be described by a three-compartment model with estimated
half-lives of 44 min, 5.7 h, and 53 h, respectively. Less than 1%
remained in the body after 9 days (Nolan et al., 1984).
In a study by Nolan et al. (1984), the trichloroethane
concentrations in blood and expired air were proportional to the
exposure concentration after 6 h of exposure and indicated that about
25% of the 1,1,1-trichloroethane inhaled during the exposure was
absorbed.
6.5 Biological monitoring
Several options exist for the biological monitoring of exposure
to 1,1,1-trichloroethane (Monster 1986). These include the
determination of:
Ê the unchanged solvent in blood or alveolar air;
Ê the metabolite trichloroethanol in blood, alveolar air or urine;
Ê the metabolite trichloroacetic acid in blood and urine.
Table 9 indicates the mean concentrations of these parameters as
a result of exposure of subjects for 8 h/day (5 days/week) to a
time-weighted average (TWA) concentration of 270 mg/m3 (50 ppm)
(Monster, 1986).
Table 9. Mean concentrations of 1,1,1-trichloroethane,
trichloroethanol, and trichloroacetic acid in blood,
alveolar air, and urinea
Time after exposure of sampling
Test End of 5-15 min 16 h 64 h
exposure
Blood (mg/litre)
1,1,1-trichloroethane 0.9 0.07
trichloroethanol 0.16 -
trichloroacetic acid 2.3 1.6
Alveolar air (mg/m3)
1,1,1-trichloroethaneb 210 (39) 13 (2.4) 8 (1.5)
trichloroethanol 0.014 0.007 -
Urine (mg/g creatinine)
trichloroacetic acid 4.9 2.5 0.9
trichloroethanol 2.5 1.8 1.5
a From: Monster (1986)
b Values in parentheses are in ppm
Trichloroethane concentrations in blood measured after work on a
Friday seem to be the best single parameter for estimation of the
TWA-week exposure (Monster, 1986). For one-day exposure, the
trichloroethane level in blood and urine is most useful (Monster,
1986).
Droz et al. (1989) suggested a physiological model for describing
variability in the biological monitoring of solvent exposure.
Standard statistical distributions are used to simulate variability in
exposure concentration, physical workload, body fluid, liver function,
and renal clearance. For groups of workers exposed daily, the model
calculates air monitoring indicators and biological monitoring
results, including levels in expired air, blood, and urine. The
calculated results obtained are discussed and compared with measured
data for physiological and toxicokinetic parameters for six solvents
including 1,1,1-trichloroethane and their metabolites. It is
suggested that such mathematical models are applicable for prediction
and management studies.
6.6 Bioaccumulation
Inhalation exposure of rats to 2700 mg/m3 (500 ppm) for 4 days,
6 h per day, led to an accumulation of 1,1,1-trichloroethane in the
fat 17 h after the last exposure. Further exposure on the 5th day
increased brain, liver, lung, and blood levels (Savolainen et al.,
1981).
A study by Travis et al. (1988) indicated that bioaccumulation of
organic chemicals, including 1,1,1-trichloroethane, perchloroethane,
trichloroethylene, and dichloromethane, in human adipose tissues was
positively correlated with their octanol-water partition coefficients.
The information presented, however, was based on data from a human
pharmacokinetic model.
7. EFFECTS ON LABORATORY MAMMALS AND IN VITRO TEST SYSTEMS
Appraisal
1,1,1-Trichloroethane administered by various routes has low
acute toxicity for laboratory animals. The acute toxicity pattern is
characterised by central nervous system depression and respiratory
arrest at lethal levels. 1,1,1-Trichloroethane is a moderate skin
and mild eye irritant.
Short-term inhalation exposure of rats to 4320-5400 mg/m3
(800-1000 ppm) or more produced an increase in liver weight. At
similar concentrations in mice, the liver changes were more marked
and included fatty infiltration and single cell necrosis. Minor
cytoplasmic alterations were seen in mice exposed to 1350 mg/m3
(250 ppm). Studies on Mongolian gerbils using 1,1,1-trichloroethane
concentrations down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several regions of
the brain. The significance of these findings is uncertain. The
no-observed-effect level (NOEL) for rats is about 2700 mg/m3 (500
ppm).
In long-term toxicity studies, 1,1,1-trichloroethane
administration led to reduced weight gain in both rats and mice.
Slight microscopic hepatic effects were seen, but only at the highest
concentration of 8100 mg/m3 (1500 ppm). There was no evidence of
carcinogenic potential in oral and inhalation studies on rats and
mice. One study reported increased incidence of leukaemia but this
study is considered inadequate.
1,1,1-Trichloroethane has been shown to have low genotoxic
potential in a range of in vitro and in vivo studies.
In a multigeneration study on mice, there was no evidence of
adverse reproductive effects (including effects on fertility) caused
by 1,1,1-trichloroethane in drinking-water at concentrations up to
1000 mg/kg body weight. Inhalation studies on female rats and mice
at 4590 mg/m3 (850 ppm) showed no evidence of teratogenic or
fetotoxic effects. However, in rats exposed to 11 340 mg/m3 (2100
ppm), there was some evidence of fetotoxicity.
The data available are inadequate to assess the immunotoxic
potential of 1,1,1-trichloroethane.
Enhanced toxicity of 1,1,1-trichloroethane was observed when
exposure was combined with exposure to ethanol.
7.1 Acute toxicity
1,1,1-Trichloroethane has a very low acute toxicity in laboratory
animals dosed by various routes of administration. Selected LD50
and LC50 values are given in Tables 10 and 11.
The toxicological pattern of acute trichloroethane poisoning is
central nervous system (CNS) depression, eventually culminating in
respiratory arrest or cardiac failure at lethal exposure levels
(Stewart, 1968).
The findings of rapid and shallow breathing in a dog following
inhalation of 0.9% (by volume) 1,1,1-trichloroethane (corresponding to
48 600 mg/m3 or 9000 ppm) may be related to an increase in the
activity of the lung stretch receptor in the vagus nerve (Kobayashi et
al., 1986). A level of 1.3% increased the heart rate but 2.8%
decreased it (Kobayashi et al., 1987).
Non-lethal acute toxicity has also been investigated (see Table
12). In a study on rats, 50% of the experimental group showed loss of
coordination (ataxia) after inhalation of 20 400 mg/m3 (3780 ppm)
for 4 h, and loss of righting-reflex was observed at 45 800 mg/m3
(8480 ppm). Tremors and death were observed at 64 800 mg/m3 (12 000
ppm) within one hour of exposure (Mullin & Krivanek, 1982).
Cardiac sensitization to adrenaline was reported in dogs exposed
to 37 800 mg/m3 (range: 21 600 to 59 400 mg/m3) for 5 min (Clark
& Tinston, 1982). An increase in concentration (range: 37 300 to 373
000 mg/m3) and inhalation period (0.5 to 20 min) was accompanied by
a decrease in the amount of adrenaline that induced arrhythmia
(Kobayashi et al., 1982).
Reinhardt et al. (1973) reported that 1,1,1-trichloroethane
caused cardiac sensitization to adrenaline in dogs at and above the
0.5% (v/v) level. The marked response was associated with ventricular
fibrillation following the challenge dose of adrenaline. At this
level, the solvent was also associated with excitement and struggling
in the animals. Histopathological examination of samples from the
dogs that developed fatal arrhythmias did not show any gross or
microscopic abnormalities.
Table 10. Acute toxicity (LD50) of 1,1,1-trichloroethane in experimental animals
Species Sex Route LD50 (mg/kg Reference
body weight)
Rat male oral 14 300 Torkelson et al. (1958)
Rat female oral 11 000 Torkelson et al. (1958)
Mouse female oral 9700 Torkelson et al. (1958)
Guinea-pig male & female oral 8600 Torkelson et al. (1958)
Rabbit male & female oral 10 500 Torkelson et al. (1958)
Rabbit male & female skin 15 800 Torkelson et al. (1958)
Mouse female intraperitoneal 3700 Gradiski et al. (1974)
Mouse male intraperitoneal 5080 Klaassen & Plaa (1966)
Dog male intraperitoneal 4140 Klaassen & Plaa (1967)
Table 11. Acute toxicity by inhalation (LC50) of 1,1,1-trichloroethane
in experimental animals
Species Sex LC50 Exposure Reference
(g/m3) (ppm) duration
Rat male & female 97.2 18 000 3 h Adams et al. (1950)
Rat male & female 76.9 14 250 7 h Adams et al. (1950)
Rat male 99.4 18 400 4 h Siegel et al. (1971)
Rat male 55.6 10 300 6 h Bonnet et al. (1981)
Rat male & female 205 38 000 15 min Clark & Tinston (1982)
Mouse male 120 22 240 30 min Woolverton & Balster (1981)
Mouse female 72.4 13 410 6 h Gradiski et al. (1978)
Mouse male 21.1 3910 2 h Horiguchi & Horiguchi (1971)
Mouse male 99.1 18 358 1 h Moser & Balster (1985)
Mouse male 111 20 616 30 min Moser & Balster (1985)
Mouse male 159 29 492 10 min Moser & Balster (1985)
Table 12. Non-lethal acute toxicity in experimental animals
exposed to 1,1,1-trichloroethane by inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 45 800 4 h loss of righting Mullin & Krivanek (1982)
reflex (EC50)
Rat 20 400 4 h loss of coordination Mullin & Krivanek (1982)
(EC50)
Mouse 31 000 1 h inverted screen test Moser & Balster (1985)
performance (EC50)
Dog 37 800 5 min cardiac sensitization Clark & Tinston (1982)
to adrenaline
Dog 21 600 a few min decrease in blood Kobayashi et al. (1983)
pressure
The effect of 1,1,1-trichloroethane on liver enzyme activity in
the serum of experimental animals has been investigated. An almost
lethal intraperitoneal dose of 1,1,1-trichloroethane (0.87 ml/kg body
weight) caused a significant elevation of ALAT (alanine
aminotransferase) in dogs (Klaassen & Plaa, 1967), and one eighth of
a lethal intraperitoneal dose caused a significant elevation in SDH
(sorbitol dehydrogenase) (Lundberg et al., 1986). In both studies,
the hepatotoxicity of 1,1,1-trichloroethane was relatively small
compared to that of other organic solvents, e.g., chloroform, carbon
tetrachloride, dimethylformamide (Lundberg et al., 1986),
dichloromethane, 1,1,2-trichloroethane, and trichloroethylene
(Klaassen & Plaa, 1967).
7.1.1 Irritation
Trichloroethane has been shown to produce "mild" skin irritation
in rabbits when repeatedly applied topically under an occlusive
dressing. Redness was noted and the skin became scaly, but the effect
was transient, the skin rapidly returned to normal (Torkelson et al.,
1958).
After guinea-pigs had been exposed to 1 ml of
1,1,1-trichloroethane via a skin depot (a glass ring attached to the
skin and covered with glass), for 15 min, oedema was observed.
Prolonged exposure for several hours led to more severe inflammatory
reactions in the upper part of the dermis, and histological
examination at 15 min and 1, 4, and 16 h showed a number of changes to
the epidermis. The extent of these changes increased with duration of
exposure (Kronevi et al., 1981).
Repeated topical application of trichloroethane to abraded and
non-abraded rabbit skin for up to 90 days resulted in slight,
reversible irritation (Torkelson et al., 1958). The same study showed
that when trichloroethane was applied soaked in a cotton wool pad and
bandaged to the shaven belly of a rabbit slight reddening and
scaliness occurred, but this only increased slightly with repeated
applications.
The application of 0.5 ml trichloroethane to the shaven skin of
rabbits under an occlusive dressing for 24 h, resulted in moderate
skin irritation (Duprat et al., 1976). In a recent study of OECD
methods (4-h exposure under semi-occlusive dressing) trichloro-ethane
was reported to be a skin irritant (van Beek, 1990).
Trichloroethane is a "mild" eye irritant. Slight to moderate
pain and slight conjunctival irritation, but no corneal damage, was
reported following a single application of 100 µl trichloroethane to
the eyes of rabbits (Torkelson et al., 1958). Duprat et al. (1976)
also found that instillation of 0.1 ml trichloroethane to the eyes of
rabbits produced slight irritation.
7.1.2 Short-term exposure
7.1.2.1 Inhalation
When groups of rats, guinea-pigs, rabbits, dogs, and squirrel
monkeys were exposed to 1,1,1-trichloroethane (11 880 mg/m3, 2200
ppm) for 8 h/day, 5 days/week for 6 weeks, the only sign of toxicity
was reduced body weight gain in rabbits and dogs. There were no
effects on haematological parameters or serum urea nitrogen and no
histopathological changes were observed (Prendergast et al., 1967).
In a study by Adams et al. (1950), rats were exposed to 0 or 27
000 mg/m3 (0 or 5000 ppm) for 7 h/day on 32 out of 45 days and
guinea-pigs received 20 to 65 7-h exposures to 0, 3510, 8100, 16 200
or 27 000 mg/m3 (0, 650, 1500, 3000 or 5000 ppm) for a period of 1-3
months. Body weight gain was reduced in both species at all exposure
levels used, but there were no other signs of toxicity and no effects
were observed on blood urea nitrogen. The only 1,1,1-trichloroethane-
related effect observed on histopathological examination was fatty
degeneration, without necrosis, in the liver of guinea-pigs at 16 200
and 27 000 mg/m3.
Rats exposed to 2700 mg/m3 (500 ppm), 6 h/day for 5 days,
showed no behavioural effects. However, there was a slight decrease
in brain RNA content relative to controls (Savolainen et al., 1977).
Inhalation of 1750 mg/m3 (320 ppm) for 30 days had no effects on the
composition of brain lipids in rats (Kyrklund et al., 1988).
When rats were given 4320 mg/m3 (800 ppm) by inhalation 6
h/day, 5 days/week for 4 weeks, absolute and relative liver weights
were increased but there was no induction of liver microsomal
cytochrome P-450 (Toftgaard et al., 1981). A 1-h daily exposure to 54
000 mg/m3 (10 000 ppm) for 3 months resulted in a narcotic effect
(sedation and transient sleep) and in an increase of relative liver
weight in rats, but there was no evidence of organ damage (Torkelson
et al., 1958).
When rats were exposed continuously for 100 days to 1350 or 5400
mg/m3 (250 or 1000 ppm), no effects were observed in the low-dose
group but an increase in relative liver weight was seen in the
high-dose group (McEwen & Vernot, 1974). No toxicity was observed in
similar experiments with dogs and monkeys (McEwen & Vernot 1974).
Exposure of mice to 5400 mg/m3 (1000 ppm) continuously for 14
weeks resulted in marked liver changes (elevated relative liver
weight, moderate liver triglyceride accumulation, and necrosis of
individual hepatocytes). Electron microscopy showed extensive
cytoplasmic modifications consisting of vesiculation of the rough
endoplasmic reticulum with loss of attached polyribosomes, increased
smooth endoplasmic reticulum, microbodies (peroxisomes), and
triglyceride droplets. The observed toxic effects were similar to,
but much less severe than, those produced by carbon tetrachloride.
Only minor cytoplasmic alterations were seen in mice exposed to 1350
mg/m3 (250 ppm) (McNutt et al., 1975).
No signs of toxicity were observed in rats, rabbits, guinea-pigs,
dogs or monkeys exposed to 2730 mg/m3 (500 ppm) 7 h/day, 5 days/week
for 6 months (Torkelson et al., 1958).
In a study by Prendergast et al. (1967), rats, rabbits,
guinea-pigs, dogs, and monkeys were exposed continuously to 754 or
2059 mg/m3 for 90 days. At the higher dose level, there were no
deaths in any species after 90 days, and the authors stated that no
visible toxic signs were observed. However, at the lower dose level,
some deaths occurred (2 out of 15 rats and 1 out of 3 rabbits).
Varying degrees of lung congestion were noted in the surviving
animals. In view of this and the deaths at the lowest dose tested,
the authors stated that no positive conclusion could be drawn as to
whether the effects were associated with the exposure.
No adverse effects were seen in male Wistar rats exposed to 1100
mg/m3 (204 ppm) 8 h/day, 5 days/week for 14 weeks (Eben & Kimmerle,
1974).
When young adult Mongolian gerbils (Meriones ungiculatus) were
continuously exposed to 378, 1134 or 5400 mg/m3 (70, 210 or 1000
ppm) for 3 months, followed by a 4-month period without exposure,
increased glial fibrillary acidic protein (GFA) was found in the
cerebral cortex at the two highest exposure levels, indicating
astrogliosis in this region of the brain (Rosengren et al., 1985). In
a similar study by Karlsson et al. (1987), the DNA concentrations in
several brain regions were decreased in animals exposed to 378 mg/m3
(70 ppm), the only exposure concentration used.
Table 13 summarizes data from short-term inhalation studies.
7.1.2.2 Oral administration
When rats were dosed orally with 1,1,1-trichloroethane in corn
oil, 5 days/week for 6 weeks, a dosage of 3.2 g/kg body weight per day
had no adverse effects. However, total doses of 5.6 g/kg body weight
in females and 10 g/kg body weight in males induced 40% mortality and
decreased the body weight of survivors (NCI, 1977). In mice no
mortality was seen with a dose of 5.6 g/kg body weight. Thus,
trichloroethane has low toxicity in rats and mice following repeated
exposure by the oral route.
Table 13. Short-term toxicity (exposure-effect relationships) in
experimental animals exposed to 1,1,1-trichloroethane by
inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 5400 100 days increased relative McEwen & Vernot
liver weight (1974)
Rat 1100 14 weeks, NOAEL Eben & Kimberle
8 h/day, (1974)
5 days/week
Gerbil 5400 3 months reduced brain weight Rosengren et al.
(1985)
Gerbil 380 3 months biochemical changes Karlsson et al.
in the brain (1987)
Mouse 5400 14 weeks liver necrosis McNutt et al.
(1975)
Mouse 1350 14 weeks minor liver effects McNutt et al.
(1975)
Rat, dog, 2700 6 months, NOAEL Torkelson et al.
rabbit, 7 h/day, (1958)
monkey, 5 days/week
guinea-pig
Rat, dog, 2000 90 days NOAEL Prendergast et al.
rabbit, (1967)
monkey,
guinea-pig
7.2 Long-term exposure
In the NCI (1977) carcinogenesis study (see section 7.5.1 for
details of doses), reduced weight gain was noted in both rats and mice
during the exposure period at all dose levels. Furthermore, bloody
discharge and crusting around the eyes of some rats was observed
during the second year (NCI, 1977).
In an oncogenicity inhalation study, female rats exposed to the
highest concentration (8100 mg/m3, 1500 ppm) of 1,1,1-
trichloroethane exhibited a significant decrease in body weight, and
both females and males showed very slight microscopic hepatic effects
at 6, 12, and 18 months. At lower concentrations and in male rats and
mice of both sex, no toxicity was observable (Quast et al., 1988).
7.3 Reproductive toxicity, embryotoxicity, and teratogenicity
Inhalation studies in female rats and mice gave no evidence of
any teratogenic or fetotoxic effects followed exposure to
1,1,1-trichloroethane at a concentration of 4700 mg/m3 (875 ppm) for
7 h/day on days 6-15 of gestation (Leong et al., 1975). No signs of
maternal toxicity were noted.
In a study of female rats exposed to a higher
1,1,1-trichloroethane concentration (11 340 mg/m3, 2100 ppm, 6
h/day, 5 days/week) for 2 weeks before mating and/or during 20 days of
gestation (6 h/day, 7 days/week), there was some evidence of
fetotoxicity. This consisted of reduced fetal weight in the group
exposed during pregnancy only and minor visceral and skeletal
abnormalities, such as delayed ossification, in the group exposed
before and during pregnancy (York et al., 1982). No signs of maternal
toxicity were observed.
In a two-generation fertility study incorporating teratogenicity
and dominant lethal elements, mice received 0, 100, 300 or 1000 mg
1,1,1-trichloroethane/kg daily in the drinking-water (Lane et al.,
1982). Two or three litters were produced from each of the two
generations, and the parental animals and offspring were used
variously to investigate fertility, developmental toxicity, and
dominant lethal effects. No adverse effects on any aspect of
reproduction were evident in this study. The level of
1,1,1-trichloroethane in the drinking-water in this study was reported
to be up to 6000 mg/litre but was not actually measured. In a further
study, rats exhibited slight aversion to 30 mg/litre in drinking-water
(George et al. 1989).
Rats (Sprague-Dawley) and rabbits (New Zealand White) were
exposed on days 6-15 and 6-18 of gestation, respectively, to 0, 5400,
16 200 or 32 400 mg/m3 (0, 1000, 3000 or 6000 ppm) for 6 h/day.
1,1,1-Trichloroethane was found to be developmentally toxic to both
rats and rabbits at the highest dose level, producing an increase in
unossified and poorly ossified cervical centra, decreased female fetal
body weight, an increase in non-viable implantations in rats, and an
increase in the frequency of bilateral extra 13th rib in rabbits.
Both rats and rabbits showed a NOEL of 16 200 mg/m3 for
developmental toxicity. For both species, the developmental toxicity
was seen in the presence of maternal tox-icity (Personal communication
by E.Z. Francis to S. Ells, US EPA, Test Rules Development Branch,
Existing Chemical Assessment Division. Review of Developmental
Toxicity Studies on 1,1,1-Trichloroethane (TCE). OHEA, ORD. Sept. 28,
1988).
Dapson et al. (1984) reported that exposure to 10 mg
1,1,1-trichloroethane per litre drinking-water caused cardiac
anomalies in developing Sprague-Dawley rats. The repeatability of
this preliminary study was investigated by George et al. (1989), who
exposed male and female CD rats to 3, 10 or 30 mg of 97% pure
1,1,1-trichloroethane per litre drinking-water using 0.05% Tween 80 as
an emulsifying agent. The animals were exposed for a period of 14
days before and at least 13 days after co-habitation. Sperm-positive
females continued to be exposed during pregnancy and lactation until
postnatal day 21. No significant effects were noted concerning the
reproductive competence of the parental animals or the postnatal
growth and development of the offspring (fertility, length of
gestation period, litter size, pup body weight, and pup survival were
all investigated). In addition, there was no increase in the
incidence of any cardiac malformations or other anomalies. Thus the
finding of Dapson et al. (1984) was not confirmed using a different
strain of rats and a 3-fold higher concentration of
1,1,1-trichloroethane.
7.4 Mutagenicity
Appraisal
1,1,1-Trichloroethane has been tested extensively for
mutagenicity, both in vitro and in vivo, and the principal studies
are summarized in Table 14. The results of most of the studies were
clearly negative. The positive results seen in some in vitro tests
give rise to some concern, but may well have been due to the presence
of stabilizers and/or impurities. Some of the studies yielding
negative results were of insufficient sensitivity. All of the in
vivo tests gave negative results. Overall, it appears that
1,1,1-trichloroethane does not have significant mutagenic potential.
Table 14. Mutagenicity studies
Test method Organism Test conditions Result References
In vitro studies on microorganisms
Baterial Salmonella typhimurium many studies, using mostly negative; some Simmon et al. (1977), Snow et al. (1979),
mutagenicity or Escherichia coli liquid or vapour, positive results attributed Nestmann et al. (1980), Gocke et al. (1981),
with and without S9 to stabilizers or impurities Rowland & Severn (1981), McDonald (1981),
activation Nagao & Takahashi (1981), Richold & Jones
(1981), Simmon & Shepherd (1981),
Ichinotsubo et al. (1981), de Serres &
Ashby (1981), HSE (1984), Shimada et al.
(1985)
Bacterial DNA E. coli or Bacillus several studies mainly negative; Kada (1981), Ichinotsubo et al. (1981),
damage/repair subtilis occasionally equivocal Green (1981), Tweats (1981), Rosenkranz
tests results et al. (1981), de Serres & Ashby (1981)
Yeasts, Saccharomyces numerous studies negative HSE (1984), de Serres & Ashby (1981),
gene mutation, cerevisiae
gene conversion Schizosaccharomyces
pombe
S. cerevisiae and mice 5000 mg/kg negative Loprieno et al. (1979)
(host-mediated assay) intraperitoneal
Table 14 (contd).
Test method Organism Test conditions Result References
In vitro mammalian cell studies
Chromosome CHO cells ± S9 activation clear positive without S9; Galloway et al. (1987)
aberrations equivocal with S9
Gene mutation L5178Y mouse ± S9 equivocal with S9; Myhr & Caspary (1988)
lymphoma cells negative without S9
Unscheduled rat, mouse hepatocytes; ± S9 negative Althaus et al. (1982), Martin &
DNA synthesis HeLa cells McDermid (1981)
DNA repair rat hepatocytes ± S9 mainly negative; some Williams (1983), Shimada et al. (1985)
positive results attributed
to presence of stabilizers
or impurities
Sister chromatid CHO cells ± S9 negative Perry & Thomson (1981)
exchange equivocal Galloway et al. (1987)
Cell various cell types ± S9 both positive and Daniel & Dehnel (1981), Styles (1981),
transformation negative results Price et al. (1978), Hatch et al. (1983)
Tu et al. (1985), HSE (1984)
Drosophila studies
Sex-linked 25 nM negative Gocke et al. (1981)
recessive
lethals
Table 14 (contd).
Test method Organism Test conditions Result References
In vivo mammalian studies
Chromosome rats 875 or 1750 ppm negative Quast et al. (1978)
aberrations 6 h/day, 52 weeks
(bone marrow)
Micronucleus mice 34-67 mg/kg negative Salamone et al. (1981)
intraperitoneal
Micronucleus mice 11-42 mg/kg negative Tsuchimoto & Matter (1981)
intraperitoneal
Micronucleus mice 266-2000 mg/kg negative Gocke et al. (1981)
intraperitoneal
Dominant mice 100-1000 mg/kg per negative Lane et al. (1982)
lethal day oral
Sperm mice 130-2680 mg/kg per negative Topham (1980)
morphology day intraperitoneal
DNA binding rats and mice low binding Prodi et al. (1988)
(liver)
7.5 Carcinogenicity
1,1,1-Trichloroethane has been investigated for carcinogenic
effects in life-time bioassays on both rats and mice by oral
administration or inhalation.
7.5.1 Oral administration
In a US National Cancer Institute (NCI) assay, 7 week-old
Osborne-Mendel rats were fed by gavage a 75% solution of technical
grade 1,1,1-trichloroethane in corn oil, 5 days/week for 78 weeks.
The daily doses were calculated to be 750 or 1500 mg/kg body weight,
and the study was terminated after 110 weeks. The number of animals
in each treated group was 50 of each sex, and the control groups
consisted of 20 animals of each sex, which were not vehicle dosed. No
increased tumour incidence was found (NCI, 1977). However, because of
an unusually high mortality both among treated and control animals
(only 6 out of 240 animals survived the 110 weeks to the end of the
experiment), this study was considered insufficient by US EPA (1984)
and placed in class D (not applicable to assess human
carcinogenicity).
In another NCI study with 5-week-old B6C3F1 mice, technical grade
trichloroethane containing 3% 1,4-dioxane was given by gavage as a
40-60% solution in corn oil at two dose levels (average daily doses:
2807 and 5615 mg/kg body weight) 5 days/week for 78 weeks. The same
number of animals was used as in the NCI rat study reported above.
All surviving animals were killed after 90 weeks of study. A variety
of neoplasms was observed in the treated groups, but the incidence was
not statistically significantly different from that of the control
groups (NCI, 1977). Again the mortality rate was very high in both
treated and control animals, although it was a little lower than in
the rat study. Nevertheless this study was also considered
insufficient (US EPA 1984).
In a study by Maltoni et al. (1986), groups of male and female
rats were given 1,1,1-trichloroethane (500 mg/kg body weight) in olive
oil by gavage (4 to 5 days/week for 104 weeks). The
1,1,1-trichloroethane contained about 4% 1,4-dioxane and 1% of other
impurities. An increased incidence of "leukaemias" was detected in
the exposed group. However, data were not evaluated statistically nor
were any data for historical controls supplied.
7.5.2 Inhalation
In an inhalation study carried out by the Dow Chemical Co.,
1-month-old Sprague-Dawley rats were exposed to technical
1,1,1-trichloroethane (6 h/day, 5 days/week for 12 months) and
observed 18 months later, before the survivors were killed. Each dose
group consisted of 96 rats of each sex. The unexposed control group
consisted of the same number of rats as the two exposed groups
together. The two exposure concentrations used were 4778 mg/m3 (875
ppm) and 9555 mg/m3 (1750 ppm). The treatment had no significant
effect on mortality, and the tumour incidence in the treated animals
was similar to that of the controls (Rampy et al., 1977). The purity
of the 1,1,1-trichloroethane used was 96%, and the stabilizers were 3%
1,4-dioxane, 0.4% nitromethane, and 0.5% butylene oxide. In the
validation of this study, it was pointed out that the exposure period
was only 12 months and not lifelong, that no subchronic range-finding
test had been done, and that the exposure concentration could have
been too low (US EPA, 1984).
A more recent long-term inhalation study, this time on both rats
and mice, has been carried out by the Dow Chemical Co. (Quast et al.,
1988). Groups of male and female 4- to 6-week-old Fischer-344 rats
and 5- to 6-week-old B6C3F1 mice (80 of each sex per group) were
exposed to 1,1,1-trichloroethane (technical formulation) vapour
concentrations of 0 mg/m3, 820 mg/m3 (150 ppm), 2730 mg/m3 (500
ppm), 8190 mg/m3 (1500 ppm) 6 h/day, 5 days/week for 2 years. Ten
animals of each sex from each group were predesignated for interim
sacrifices after 6, 12, and 18 months of exposure. The purity of the
formulation was 94% (by volume) 1,1,1-trichloroethane, 5% stabilizers
(butylene oxide, tert-amyl alcohol, methyl butynol, nitroethane, and
nitromethane) and < 1% minor impurities. In none of the treated
animal groups, did the tumour incidence differ significantly from that
of the controls.
The early studies with mice and rats were evaluated at IARC
(International Agency for Research on Cancer) and considered
inadequate (IARC, 1979; IARC, 1987).
7.6 Immunotoxicity and sensitization
Shmuter (1977) exposed rabbits to 1,1,1-trichloroethane
concentrations of 2 mg/m3 (0.4 ppm), 10 mg/m3 (1.8 ppm), and 100
mg/m3 (18 ppm) by inhalation 3 h/day, 6 days/week for 8-10 months.
At week 6, Salmonella typhimurium was injected subcutaneously to
investigate the immune response over an 8-month period. The antibody
response was depressed at both 10 mg/m3 and 100 mg/m3. This
reduction in immune response was followed by an increased
electrophoretic mobility of the antibodies toward alpha- and
ß-globulin fractions and also by a significant drop in the level of
normal haemolysins to the Forsman's antigen of the sheep erythrocytes
occuring during treatment.
7.7 Interactions
Interaction between 1,1,1-trichloroethane and ethanol was studied
in mice given ethanol (0-2 g/kg body weight) by gavage 30 min before
exposure to up to 120 000 mg trichloroethane/m3 (22 200 ppm) by
inhalation. Different combinations of exposure to the solvents
enhanced the toxicity of trichloroethane in the animals previous
exposed to ethanol. The observed behavioural and lethal effects were
dose related for each compound and additive (Woolverton & Balster,
1981).
When dogs anaesthetized with either pentobarbital sodium or
chloralose plus pentobarbital sodium were acutely exposed to a
commercial spot remover containing 1,1,1-trichloroethane, there was a
dose-dependent biphasic decline in arterial pressure with an initial
increase in the heart rate. A similar effect was obtained when
1,1,1-trichloroethane (99.5% by volume) was used instead of the
commercial spot remover. It was found that the type of anaesthetic
contributed in part to the effect. The authors reported that, in
addition to the peripheral vascular effects, significant alterations
in myocardial function occurred, and they concluded that the solvent
acts by inhibiting myocardial contractility (Herd et al., 1974).
7.8 Mechanisms of action
1,1,1-Trichloroethane given to mice intraperitoneally or by short
inhalation exposure has been shown to reduce the cyclic guanosine
monophosphate (cGMP) content in the cerebellum, cerebral cortex, and
brain stem. This was attributed to an increased rate of cGMP
hydrolysis (Nilsson, 1986a). The cyclic adenosine monophosphate
(cAMP) content in the brain stem of mice was increased in a
dose-related and time-dependant manner. This might be mediated by
adrenoceptor interaction stimulating the catalytic unit of adenylate
cyclase (Nilsson, 1986b).
A study on isolated mouse brain synaptosomes exposed in vivo
and in vitro showed that 1,1,1-trichloroethane increases calcium
influx in the brain stem but not in the cerebellum and cerebral
cortex. The data indicated that 1,1,1-trichloroethane may influence
voltage-dependant calcium channels in brain stem nerve endings
(Nilsson, 1987).
1,1,1-Trichloroethane suppressed the uptake of ovabain
taurocholate and 2-aminoisobutyric acid, but not of cadmium chloride
or 3- O-methyl-D-glucose, into isolated rat hepatocytes. Both the
cellular ATP level and ATP-ase activities in isolated membrane
fractions were also decreased. It was suggested that energy-dependant
transport functions are disrupted, owing to decreased ATP levels
and/or inhibition of membrane ATP-ases (Kukongviriyapan et al., 1990).
8. EFFECTS ON HUMANS
Appraisal
Both acute and long-term inhalation exposure to
1,1,1-trichloroethane can produce CNS effects ranging from slight
behavioural changes to unconsciousness. Death may follow from
respiratory failure or cardiac arrest. There is also the possibility
of hepatic alterations and cardiac damage.
Ingestion and dermal exposure do not appear to be serious
concerns, although skin irritation may occur.
1,1,1-Trichloroethane has properties (see chapter 2) that make it
a dangerous compound in poorly ventilated areas and confined spaces
such as tanks and vaults. The vapour is five times more dense than
air, which may result in non-uniform distribution, and very high
levels can occur in certain areas such as the bottom of empty tanks
and container vessels. The combination of these properties has been
the cause of some serious accidents, including fatalities. Solvent
abuse has also produced a large number of fatalities.
There are insufficient studies to permit any conclusion
concerning the reproductive and carcinogenic effects of this compound
on humans.
Exposure to 1,1,1-trichloroethane may increase sensitivity to
other compounds, e.g., anaesthetics.
The NOEL for humans appears to be in the region of 1350 mg/m3
(250 ppm).
Table 15 summarizes the toxic effects of 1,1,1-trichloroethane
after inhalation exposure.
8.1 Controlled human studies
Several studies on volunteers under controlled conditions have
investigated the effects of 1,1,1-trichloroethane on the CNS as
measured by performance in certain behavioural tests. These studies
have usually been conducted using the technical grade of
1,1,1-trichloroethane containing stabilizers, since this is the type
of exposure that occurs most frequently.
Table 15. Toxic effects in humans related to inhalation exposure to 1,1,1-trichloroethane
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
378 000 70 000 a few min death Northfield (1981)
54 000 10 000 2 min general anaesthesia when given Dornette & Jones (1960)
together with nitrous oxide (N2O)
> 27 000 > 5000 10 min death Kleinfeld & Feiner (1966)
27 000 5000 5 min marked incoordination Stewart (1968)
10 260 1900 5 min disturbance of equilibrium Torkelson et al. (1958)
5400 1000 short-term mild eye and nasal discomfort, Stewart (1968)
impairment of coordination
5000 920 70-75 min CNS disturbances (lightheadedness), Torkelson et al. (1958)
slight eye irritation
4860 900 73 min impaired performance in Romberg's Stewart et al. (1961)
test, mild eye irritation
2700 500 6-7 h/day for impaired performance in Romberg's Stewart et al. (1969)
5 days test after 1 h of exposure, mild
eye irritation, drowsiness, headaches
2700 500 90-450 min no CNS disturbances Torkelson et al. (1958)
2430 450 30 min dizziness, mild eye irritation Salvini et al. (1971)
2430 450 30 min reduced reaction time and Gamberale & Hultengren (1973)
perceptual speed
Table 15 (contd).
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
1890 350 30 min reduced perceptual speed Gamberale & Hultengren (1973)
1900 350 3.5 h reduced psychomotor performance Mackay et al. (1987)
< 1860 < 345 6.7 years NOEL Maroni et al. (1977)
< 1350 < 250 1-6 years NOEL Kramer et al. (1978)
1350 250 30 min NOEL Gamberale & Hultengren (1973)
950 175 3.5 h reduced reaction time MacKay et al. (1987)
540 100 odour threshold Stewart (1968)
8.1.1 Single exposure period
In a study by Torkelson et al. (1958), four volunteers were
exposed in an inhalation chamber to average trichloroethane levels of
4968 mg/m3 (920 ppm) for 70-75 min. Symptoms of slight CNS
disturbances (lightheadedness) were noted in three of the four
subjects and slight eye irritation was noted in one individual. Two
volunteers reported a strong odour during the experiment.
Concentrations in the range of 2700-2970 mg/m3 (500-550 ppm) for
90-450 min apparently produced no symptoms of CNS disturbance, but
exposure to 10 260 mg/m3 (1900 ppm) for 5 min produced a very
noticeable odour and obvious disturbances in equilibrium.
Unfortunately, full details of this study are not available.
Exposure to about 2700 mg/m3 (500 ppm) trichloroethane for
78-186 min did not result in CNS toxicity or other effects (Stewart et
al., 1961). However, exposure to about 4860 mg/m3 (900 ppm) for up
to 73 min resulted in CNS disturbances (light-headedness and impaired
Romberg test performance) in approximately half of the subjects. Mild
eye irritation was also observed. In another experiment in this
series, the volunteers were exposed to increasing concentrations in
the range of 0-14 310 mg/m3 (0-2650 ppm) over a 15-min period.
Signs of mild eye irritation were noted in six out of seven subjects
when the concentration reached about 5400 mg/m3 (1000 ppm), and
there was a rapid increase in CNS symptoms, principally dizziness.
Throat irritation was experienced by six out of seven subjects at
about 10 800 mg/m3 (2000 ppm). Severe CNS disturbances (imbalance,
light-headedness) occurred at the end of the exposure period, when the
concentration had reached 14 310 mg/m3. In addition, minor effects
on liver and kidney function were seen. The authors considered that
all the effects were reversible.
The effect on young men of exposure to a mean trichloroethane
level of 2430 mg/m3 (450 ppm) for two periods of 4 h (separated by
a lunch break of 1´ h) was investigated by Salvini et al. (1971). The
volunteers experienced dizziness and slight excitation, but only
during the first 30 min of exposure, and mild eye irritation was
noted. No significant change in performance in any of the behavioural
tests was seen.
In a study by Gamberale & Hultengren (1973), 12 male volunteers
were exposed to 1350 mg/m3 (250 ppm) for 30 min, followed by 1890
mg/m3 (350 ppm) for the next 30 min, and 2430 mg/m3 (450 ppm) and
2970 mg/m3 (550 ppm) for subsequent 30-min periods. At 2430
mg/m3, the reaction time, perceptual speed, and manual dexterity
were reduced, and at 1890 mg/m3 the perceptual speed was reduced.
The NOEL was 1350 mg/m3.
Mackay et al. (1987) reported the results of an exposure chamber
study in which volunteers were exposed to 950 mg/m3 (175 ppm) and
1990 mg/m3 (350 ppm) for 3 to 5 h. Several human psychomotor
behavioural tests were employed, including two new ones. One was
concerned with distraction of attention and concentration (the Stroop
test) and the other with analysing grammatical statements (the
syntactic reasoning test). Performance deficits in the psychomotor
behavioural tests were recorded in volunteers exposed to
1,1,1-trichloroethane; these depended on the time of exposure and the
blood levels. The performance changes were rapid and occurred in some
cases within 20 min. However, different effects were found. In the
Stroop test, enhanced performance was observed following exposure,
whereas the syntactic reasoning test was found not to respond to
1,1,1-trichloroethane exposure effects. Measures of short-term
subjective well-being were not affected by exposure. Other
parameters, such as simple reaction time and four-choice reaction time
were increased.
The effect of exposure to trichloroethane on performance in a
very extensive range of behavioural tests was investigated by
Savolainen et al. (1981, 1982a, 1982b). Nine healthy male students
(aged 20-25) were exposed in an inhalation chamber to 1080 and 2160 mg
trichloroethane/m3 (200 and 400 ppm) for 4 h, with a 6-day interval
between exposure at the two levels. There was no clear indication of
any effects on the CNS, although some minor changes were noted in
body-sway measurements when the subjects had their eyes closed.
8.1.2 Repeated exposure
The effect of repeated exposure of male volunteers to 2700
mg/m3 (500 ppm), 7 h/day for 5 days, was studied in an inhalation
chamber (Stewart et al., 1969). Several subjective symptoms were
noted, e.g., drowsiness, headaches, lightheadedness, and eye and nose
irritation, but the absence of a control group made it difficult to
assess the significance. No performance impairment in behavioural
tests (modified Romberg test) was noted, apart from the inability of
two subjects to perform the balance test on two occasions.
8.2 Accidental exposure
Respiratory system symptoms, such as cough, breathlessness, and
chest tightness, are common in cases of acute poisoning (Boyer et al.,
1987).
Since 1,1,1-trichloroethane is a very volatile solvent with a
saturated vapour concentration of 864 g/m3 (160 000 ppm) at 25 °C,
there is a potential for very high air concentrations in confined
spaces, such as tanks and vaults. Furthermore, its vapour is five
times more dense than air. This may result in non-uniform
distribution in the workplace air and very high levels in certain
areas such as just above degreasing vessels and at the bottom of
empty tanks, even though levels elsewhere are low and safe. This
property has been the cause of some serious accidents and fatalities.
A near-fatal childhood intoxication was described by Gerace
(1981). A 4-year-old boy was playing under his bed covers with a
flower-making kit which contained a 30-cm3 container of
1,1,1-trichloroethane. Because of some unusual noises heard by a
sibling, the child was quickly discovered. However, he was not
breathing at this point. He was resuscitated on the way to the
hospital but remained comatose for about 12 min after arrival. The
author suggested that the child was rendered unconscious by the
1,1,1-trichloroethane and that the noises heard were seizures caused
by hypoxia. The child was released 48 h after admission. According
to the author, clinical investigation during the child's hospital stay
demonstrated some hepatic abnormalities with alteration in liver
bilirubin.
Caplan et al. (1976) reported the death of a woman due to
accidental exposure while cleaning a paint spill using a paint thinner
in a poorly ventilated room. Autopsy results showed "mild fatty
changes" in the liver and acute oedema and congestion of the lungs.
The concentrations of 1,1,1-trichloroethane in the brain, kidney,
liver, and blood were 36, 12, 5, and 2 mg/100 ml, respectively.
8.2.1 Confined spaces at workplaces
A man who had spent 10 min in a vault, where the concentration of
1,1,1-trichloroethane was estimated to be well in excess of 27 g/m3
(5000 ppm), collapsed and died shortly after leaving the vault
(Kleinfeld & Feiner, 1966).
Seven fatalities from using trichloroethane to clean equipment on
naval ships or aircraft tanks have been reported (Stahl et al., 1969;
Hatfield & Maykoski, 1970). The exposure level in one of these cases
was estimated to be in the order of 270 g/m3 (50 000 ppm). In all
cases the most significant pathological finding was pulmonary oedema.
Liver damage was not seen.
Other case histories with accidental death due to
1,1,1-trichloroethane exposure in confined spaces have been reported
(Stewart 1968; Bonventre et al., 1977).
During the period 1961-1980, the Factory Inspectorate in the
United Kingdom revealed 52 incidents due to trichloroethane (McCarthy
& Jones, 1983). Loss of consciousness was reported in 26 cases and
death in three cases, all involving very young workers. In one case
the worker died after leaning over an open tank of trichloroethane,
apparently to wash his hands in the solvent. He was found unconscious
slumped over the side of the tank, and the solvent concentration in
his breathing zone was later estimated to be about 378 g/m3 (70 000
ppm) (Northfield, 1981). In a second case, a worker was using
trichloroethane for cleaning purposes in a closed room (Jones &
Winter, 1983). He was found on the floor with chemical burns on the
head and neck, which was consistent with prolonged skin absorption
from spills of solvent on the floor. The third case was a teenage
worker who cleaned the interior of a car with a cloth soaked in the
solvent (Jones & Winter, 1983). Death was presumed to be from
1,1,1-tri-chloroethane intoxication and simultaneous inhalation of
vomit.
8.2.2 Solvent abuse
During the 1960s, 110 cases of sudden death from solvent abuse
were reported in the USA. Of these, spot remover containing
1,1,1-trichloroethane was identified as responsible for 29 cases
(Bass, 1970). Death frequently occurred immediately following
stressful activity. Where autopsies were performed, no abnormalities
could be detected, and it was suggested that deaths was due to cardiac
arrhythmia.
Over the period 1971-1981, 140 deaths from solvent abuse were
reported in the United Kingdom; 20 of them were believed to be due to
1,1,1-trichloroethane, mainly in dry-cleaning fluid, domestic cleaners
or plaster remover (Anderson et al., 1982).
In the case of a fatality due to the abuse of cleaning solvent
containing 1,1,1-trichloroethane, very minor hepatic changes were seen
(Hall & Hine, 1966). In four cases of deaths caused by inhalation of
typewriter correction fluid containing 1,1,1-trichloroethane and
trichloroethylene, the autopsies showed no evidence of kidney or liver
damage (King et al., 1985).
According to Marjot & McLeod (1989), toluene and the chlorinated
hydrocarbons 1,1,1-trichloroethane and trichloroethylene can cause
permanent damage to the kidney, liver, heart, and lung in certain
volatile substance abusers.
8.2.3 Medical use
Trichloroethane has been used as an anaesthetic on an
experimental basis. General anaesthesia in humans has been rapidly
induced by 2 min of exposure to 54 000 mg/m3 (10 000 ppm) given with
a mixture of nitrous oxide (N2O) and oxygen (80:20). Cardiovascular
side effects (slight to severe hypotension, cardiac arrhythmias, and,
in one case, cardiac arrest) were observed in several of the patients
during anaesthesia. In view of these effects, the experimental use of
1,1,1-trichloroethane as an anaesthetic was discontinued (Dornette &
Jones, 1960).
In the USA, after aerosol drug products containing
1,1,1-trichloroethane had been implicated in 21 deaths from their
abuse or misuse, the Food and Drug Administration (FDA) removed the
products from the market (US Food and Drug Administration, 1973). The
medical products were mainly used as cough suppressants and contained
1,1,1-trichloroethane as a solvent for the active ingredients and to
reduce the vapour pressure of the propellants. The FDA found lack of
general recognition by qualified experts of the safety or
effectiveness of 1,1,1-trichloroethane in aerosol drug products
intended for inhalation.
Under 1977 rules from the FDA, it was stated that
1,1,1-trichloroethane is potentially toxic to the cardiovascular
system by sensitizing the heart to adrenaline (Food and Drug
Administration, 1977).
8.2.4 Ingestion
In a case of accidental ingestion of about 600 mg
trichloroethane/kg, signs of severe gastrointestinal irritation
(vomiting, diarrhoea) were evident shortly after the ingestion. There
was no evidence of any CNS disturbance or any liver or kidney
dysfunction (Stewart & Andrews, 1966).
Ingestion of about 30 ml trichloroethane has been found to cause
severe vomiting and diarrhoea (CEC, 1986).
8.2.5 Drinking-water contamination
In December 1981, a water well in Santa Clara County, California,
USA, was removed from service after contamination with
1,1,1-trichloroethane and other solvents was detected. A
1,1,1-trichloroethane level of 1.7 mg/litre was detected at the well
head, but the level of the other contaminants (dichloroethylene,
isopropyl alcohol, and freon) was not reported. Subsequently
suspicion of a cluster of spontaneous abortions and congenital heart
anomalies from pregnancies conceived during 1980-1981 was reported by
the local community. Epidemiological studies indicated that a cluster
did exist, that the odds ratio for spontaneous abortion was 2.3 and
the relative risk for congenital malformations was 3.1, but they were
unable to draw a direct link between the ingestion of the contaminated
water and these effects (Goldberg et al., 1990).
8.3 Effects on the skin and eyes
Prolonged or repeated skin contact with 1,1,1-trichloroethane may
lead to dermatitis, due to its defatting action. However, despite its
wide use only a few cases of skin irritancy have been reported
(Torkelson et al., 1958).
Immersion of a hand in liquid 1,1,1-trichloroethane for 30 min
resulted in mild erythema, which persisted for one hour (Stewart &
Dodd, 1964).
Splashed in the eye, the compound may produce conjunctivitis
(Stewart, 1968). However, accidental eye contact has been reported to
produce only transient irritation (Savolainen et al., 1982a,b).
Exposure to a vapour concentration of 2700 mg/m3 (500 ppm) may
produce mild eye irritation (see section 8.1).
8.4 Long-term occupational exposure
A case study described a patient with liver cirrhosis after
several years of heavy exposure to trichloroethylene followed by 3
months of work that frequently involved using aerosolized degreaser
containing trichloroethane (Thiele et al., 1982).
An investigation of the neurotoxicity of trichloroethane was
carried out on a group of 22 female workers exposed to the solvent for
an average period of 6.7 years. The group was subdivided into three
exposure groups (mean levels: 594 mg/m3 (110 ppm), 756-864 mg/m3
(140-160 ppm), and 1080-1863 mg/m3 (200-345 ppm)). Each woman had
a general physical examination, and information about symptoms was
obtained from a questionnaire. The measurement of nerve conduction
velocity and psychometric functions indicated no differences between
the exposed workers and an unexposed control group of 7 workers
(Maroni et al., 1977).
Hodgson et al. (1989) reported that four out of ten clinical
cases of fatty liver disease were associated with occupational
exposure to 1,1,1-trichloroethane.
A matching-pair study of 151 pairs of workers at textile plants,
with and without daily exposure to trichloroethane levels of up to
1365 mg/m3 (250 ppm), found no toxic effect among the exposed
workers, the majority of whom were exposed to less than 810 mg/m3
(150 ppm) for a period of 1-6 years (Kramer et al., 1978).
In a case-control study of the effect of occupational exposure to
various solvents on spontaneous abortion rates in Finland (1973-1983),
a statistically significant elevation in relative risk was found for
exposure to organic solvents in general. However, there were very few
cases related specifically to 1,1,1-trichloroethane. The low
statistical power, therefore, precludes any conclusion as to whether
the data show an effect or not for 1,1,1-trichloroethane (Lindbohm et
al., 1990)
There are no adequate epidemiological studies available
concerning long-term exposure to 1,1,1-trichloroethane and
carcinogenicity.
8.5 Interactions
Chronic cardiac arrhythmia developed in two cases, after routine
halothane anaesthesia, in subjects with previous repeated long-term
exposure to 1,1,1-trichloroethane (in one case, occupational and in
the other, due to solvent abuse). The effects involved myocardiac
damage with either ventricular arrhythmia or deterioration of left
ventricular function. In one of the cases, the pathologist suggested
a subacute myocarditis superimposed on long-standing damage. The
authors postulated that the interaction between the two chemicals may
have caused the cardiac deterioration and concluded that cardiac
damage sustained after long-term exposure to 1,1,1-trichloroethane may
be severe, life-threatening, and irreversible (McLeod et al.,1987)
Marjot & McLeod (1989) reported two cases, a 14-year-old boy
exposed to 1,1,1-trichloroethane via solvent abuse and a 54-year-old
man exposed occupationally, where the cardiac toxicity of
1,1,1-trichloroethane may have been increased by exposure to halothane
(2-bromo-2-chloro-1,1,1-trifluoroethane) as an anaesthetic.
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
Appraisal
1,1,1-Trichloroethane is not likely to be a significant hazard to
non-target organisms in the environment. For example, 96-h LC50
values for fish ranged from 33 mg/litre for dabs to 105 mg/litre for
fathead minnows. The 48-h LC50 for Daphnia was around 60 mg/litre.
The EC50 for carbon dioxide incorporation into algae was 5
mg/litre.
9.1 Microorganisms
A toxic threshold of 93 mg 1,1,1-trichloroethane/litre has been
estimated for the inhibition of cell multiplication in the bacterium
Pseudomonas putida (Bringmann & Kuhn, 1977). The 3-h EC50 values,
for inhibition of photosynthesis (measured by monitoring the uptake of
14CO2), for the green algae Chlamydomonas angulosa (at a cell
concentration of 5 x 104 cells/ml) and Chlorella vulgaris (at a
cell concentration of 20 x 104 cells/ml) were 280 mg/litre and 153
mg/litre, respectively, under static conditions (Hutchinson et al.,
1980). Bringmann & Kuhn (1978) calculated toxic thresholds of 350
mg/litre and 430 mg/litre, respectively, for inhibition of cell
multiplication, over an 8-day period, of the cyano bacterium
(blue-green alga) Microcystis aeruginosa and the green alga
Scenedesmus quadricauda. The 96-h EC50 for chlorophyll a and cell
number in the green alga Selenastrum capricornutum was > 669
mg/litre (US EPA, 1980a,b). Pearson & McConnell (1975) assessed the
toxicity of 1,1,1-trichloroethane to the unicellular alga
Phaeodactylum tricornutum by measuring changes in the uptake of
carbon, from 14CO2, during photosynthesis, and obtained an EC50
of 5 mg/litre.
9.2 Aquatic organisms
The acute toxicity of 1,1,1-trichloroethane to aquatic organisms
is summarized in Table 16.
Flow-through tests gave much lower LC50 values than static
tests, suggesting that some loss via evaporation may be occurring.
The 48-h LC50 for aquatic invertebrates ranges from 7.5 mg/litre for
barnacles in a flow-through test to > 530 mg/litre for daphnids in a
static test. The 96-h LC50 for fish ranges from 33 mg/litre for the
dab (Limanda limanda) in flow-through tests to 105 mg/litre for the
fathead minnow (Pimephales promelas) in static tests.
Table 16. Acute toxicity of 1,1,1-trichloroethane to aquatic organisms
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Invertebrates
Mysid shrimp stat 96 31.2 n US EPA (1980b)
(Mysidopsis bahia)
Barnacle nauplii stat 48 7.5 n Pearson & McConnell (1975)
(Eliminius modestus)
Water flea < 24 h stat 21-23 72 6.7-8.1 48 > 530 n LeBlanc (1980)
(Daphnia magna) 4-6 day stat 21-25 48 59.6 Abernethy et al. (1986)
4-6 day stat 48 57.6 Bobra et al. (1984)
Fish
Dab 15-20 cm flow 96 33 m Pearson & McConnell (1975)
(Limanda limanda)
Sheepshead minnow 8-15 mm stat 25-31 10-31d 96 71 (60-81) n Heitmuller et al. (1981)
(Cyprinodon variegatus)
Bluegill 0.32-1.2 g stat 21-23 32-48 6.7-7.8 24 110 n Buccafusco et al. (1981)
(Lepomis macrochirus) 0.32-1.2 g stat 21-23 32-48 6.7-7.8 96 72 (57-90) n Buccafusco et al. (1981)
stat 96 69.7 n US EPA (1980b)
Fathead minnow 1 g stat 12 7.8-8.0 96 105 (91-126) n Alexander et al. (1978)
(Pimephales promelas) 1 g flow 12 7.8-8.0 96 52.8 (43.7-77.7) m Alexander et al. (1978)
Table 16 (contd).
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Fish (contd)
Guppy 2-3 month statb 168 133 Konemann (1981)
(Poecilia reticulata)
a stat = static conditions (water unchanged for the duration of the test) unless stated otherwise; flow = flow-through conditions
(1,1,1-trichloroethane concentration continuously maintained)
b static conditions but water renewed daily
c hardness expressed as mg CaCO3 per litre
d value refers to salinity (%), not hardness
e n = nominal; m = measured
LeBlanc (1980) exposed the water flea Daphnia magna to
1,1,1-trichloroethane for 48 h under static conditions. At the
highest concentration of 530 mg/litre, no discernible effect was
observed. A no-observed-effect level (NOEL) of 43 mg/litre was
estimated by Heitmuller et al. (1981) after exposing the sheepshead
minnow (Cyprinodon variegatus) to 1,1,1-trichloroethane for 96 h.
When Thompson & Carmichael (1989) exposed the water flea Daphnia
magna to 1,1,1-trichloroethane concentrations of between 1.3 and 23
mg/litre for 17 days under semistatic conditions (water renewed every
2 days), no effect on mortality or reproduction (as measured by number
of offspring per parent) was observed at 1.3 mg/litre. However, the
mortality increased with increasing concentration of
1,1,1-trichloroethane from 30% at 2.4 mg/litre to 100% at 23 mg/litre.
The number of offspring per parent was significantly reduced at
concentrations of 2.4 mg/litre or more.
Thompson & Carmichael (1989) also exposed the mirror carp
Cyprinus carpio to 1,1,1-trichloroethane concentrations ranging from
1.4 to 30 mg/litre for 14 days under flow-through conditions. There
was no mortality at any of the concentrations, but at the highest
concentration the fish showed surfacing behaviour, loss of balance and
coughing, and their mean weight decreased. The authors considered,
therefore, that the NOEL was the next highest concentration, i.e. 7.7
mg/litre.
Alexander et al. (1978) observed fathead minnows (Pimephales
promelas), during exposure to 1,1,1-trichloroethane for up to 96 h,
for the following effects: loss of equilibrium, melanization,
narcosis, and swollen, haemorrhaging gills. The concentration
producing one or more of these observable effects in 50% of the fish
(EC50) was 11.1 mg/litre.
9.3 Terrestrial organisms
Thompson & Carmichael (1989) exposed emergent seedlings of crop
sorghum (Sorghum bicolor) and oil seed rape (Brassica napus) to
1,1,1-trichloroethane in the gaseous phase at nominal concentrations
of between 3.2 and 320 mg/litre (g/m3) for a period of 14 days (the
test material was replenished after 7 days in a sealed vessel). Mean
measured concentrations ranged from 58% to 75% of the nominal
concentration, reflecting the decline of 1,1,1-trichloroethane in the
gas phase concentration during the 7-day periods between toxicant
renewal. In the case of Sorghum bicolor, the 14-day EC50 (based
on mean plant weight and a mean measured 1,1,1-trichloroethane
concentration) was 48 mg/litre; the NOEL being 19 mg/litre. For
Brassica napus, the 14-day EC50 was 19 mg/litre and the NOEL 6.9
mg/litre, both values being based on mean measured concentrations.
Rajendran (1990) exposed 1- to 2-day-old pupae of the red flour
beetle (Tribolium castaneum) to 1,1,1-trichloroethane in fumigation
chambers. An LC50 of 208.4 mg/litre was estimated after a 24-h
exposure and a 20-day post-fumigation observation period. When pupae
were exposed to a mixture of 1,1,1-trichloroethane and methyl bromide
at a concentration of 2.5 mg/litre or 3.0 mg/litre, the toxicity was
similar to that of the individual components.
In a contact toxicity test, red earthworms (Eisenia foetida)
were exposed to 1,1,1-trichloroethane via filter paper in glass vials.
An LC50 of 83 µg/cm2 was estimated (Neuhauser et al., 1986).
Elovaara et al. (1979) injected 1,1,1-trichloroethane, at
concentrations ranging from 5 to 100 µmol/egg, into the air space of
fertilized chicken eggs at 3 or 6 days of incubation, and the
embryotoxicity was evaluated after 14 days of incubation. A clear
dose-response relationship with respect to survival was found,
irrespective of whether the eggs were injected after 3 or 6 days of
incubation. The approximate LD50 for 1,1,1-trichloroethane was
between 50 and 100 µmol/egg.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
Humans are exposed to 1,1,1-trichloroethane principally by
inhalation, where the substance is rapidly absorbed into the body.
Exposure by skin absorption or ingestion may also occur but, compared
to inhalation, is of lesser concern. 1,1,1-Trichloroethane is
considered not to bioaccumulate. The acute and chronic toxicities are
fairly low. Under conditions of high exposure there is a risk of
toxic effects; such conditions may occur in occupational exposures,
solvent abuse or accidents. Since 1,1,1-trichloroethane is volatile
and the vapour is much more dense than air, toxic and explosive
concentrations may occur unexpectedly in confined spaces. This has
caused several fatal and near-fatal accident at workplaces and
elsewhere. The critical effect in humans relates to the central
nervous system, leading to unconsciousness and respiratory arrest at
high concentrations. 1,1,1-Trichloroethane is less toxic to the liver
than most other organochlorine solvents. The no-observed-effect level
for humans appears to be in the region of 1350 mg/m3 (250 ppm).
Although one study suggested effects at a lower level, the
interpretation of the results was unclear.
Studies on Mongolian gerbils involving 1,1,1-trichloroethane
levels down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several brain regions.
If these findings are validated and confirmed, this may be of
significant concern.
No adequate study of carcinogenic effects on humans has been
published. A long-term inhalation study on rats and mice exposed to
8100 mg/m3 (1500 ppm) gave no evidence of any carcinogenic effects.
One study recorded increases numbers of "leukaemias", but it was
performed under non-standard protocol. It gave no statistical
evaluation and is, therefore, considered inadequate. It is unlikely
that 1,1,1-trichloroethane itself presents a significant risk of
genotoxic or carcinogenic effects in humans.
1,1,1-Trichloroethane is not teratogenic and elicits no
developmental effects at doses that are not maternally toxic. The
limited epidemiological evidence on reproductive effects is
inconclusive due to the low statistical power of the study.
10.2 Evaluation of effects on the environment
1,1,1-Trichloroethane is released in large quantities to the
environment. Due to its volatility and general stability it is a
ubiquitous environmental contaminant.
Although the ozone-depleting and global-warming potentials of
1,1,1-trichloroethane are lower than those of many
chlorofluorocarbons, the large-scale release of the compound into the
atmosphere merits serious concern in these respects.
As a result of its widespread use and disposal and its mobility
in soil, persistent 1,1,1-trichloroethane contamination of ground
water, including deep aquifers, occurs.
Conditions under which 1,1,1-trichloroethane causes
ecotoxicological effects are not found in the environment except after
spills.
11. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH AND
THE ENVIRONMENT
a) Emissions of 1,1,1-trichloroethane should be reduced as far as
is practicable in order to minimize exposure of workers and of
the general population.
b) The release of 1,1,1-trichloroethane into the environment must
be reduced to the greatest extent possible in order to avoid
damage to the ozone layer.
c) Contamination of ground water by 1,1,1-trichloroethane from
spills and waste disposal should be avoided.
d) Safe substitutes for 1,1,1-trichloroethane should be identified.
12. FURTHER RESEARCH
a) More study is needed to clarify the possible effects of
1,1,1-trichloroethane on the biochemistry of the central nervous
system and their significance.
b) More study is necessary to clarify the possible interaction
between 1,1,1-trichloroethane and ethanol in humans.
c) An adequate subchronic or long-term oral study is needed to
assess the hepatotoxicity of 1,1,1-trichloroethane.
d) Further epidemiological studies on reproductive effects are
desirable.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
IARC (International Agency for Research on Cancer) working groups
have evaluated 1,1,1-trichloroethane and concluded that the evidence
for genotoxicity is limited (IARC, 1979, 1987).
REFERENCES
ABERNETHY, S., BOBRA, A.M., SHIU, W.Y., WELLS, P.G., & MACKAY, D.
(1986) Acute lethal toxicity of hydrocarbons and chlorinated
hydrocarbons to two planktonic crustaceans: The key role of
organism-water partitioning. Aquat. Toxicol., 8: 163-174.
ADAMS, E.M., SPENCER, H.C., ROWE, V.K., & IRISH, D.D. (1950) Vapor
toxicity of 1,1,1-trichloroethane (methyl chloroform) determined by
experiments on laboratory animals. Arch. ind. Hyg., 1: 225-236.
AGGAZZOTTI, G. & PREDIERI, G. (1986) Survey of volatile halogenated
organics (VHO) in Italy. Water Res., 20: 959-963.
ALEXANDER, H.C., MCCARTY, W.M., & BARTLETT, E.A. (1978) Toxicity of
perchloroethylene, trichloroethylene, 1,1,1-trichloroethane, and
methylene chloride to fathead minnow. Bull. environ. Contam. Toxicol.,
20: 344-352.
ALTHAUS, F.R., LAWRENCE, S.D., SATTLER, G.L., LONGFELLOW, D.G., &
PITOT, H.C. (1982) Chemical quantification of unscheduled DNA
synthesis in cultured hepatocytes as an assay for the rapid screening
of potential chemical carcinogens. Cancer Res., 42(8): 3010-3015.
AMI (1988) [The Factories Inspectorate's data base for workplace
measurements, ATABAS, stock-taking for the period January 1983 -
December 1987], Copenhagen, Institute for the Working Environment (in
Danish).
ANDERSON, H.R., DICK, B., MACNAIR, R.S., PALMER, J.C., & RAMSEY, J.D.
(1982) An investigation of 140 deaths associated with volatile
substance abuse in the United Kingdom (1971-1981). Hum. Toxicol., 1:
207-221.
ARCHER, W.L. (1979) 1,1,1-Trichloroethane. In: Kirk-Othmer
encyclopedia of chemical technology, 3rd ed., New York, Chichester,
John Wiley and Sons, pp. 728-731.
ARCHER, W.L. & STEVENS, V.L. (1977) Comparison of chlorinated,
aliphatic, aromatic, and oxygenated hydrocarbons as solvents. Ind.
Eng. Chem. Prod. Res. Dev., 16: 319-325.
ASTRAND, I., KILBOM, A., WAHLBERG, I., & OVRUM, P. (1973) Methyl
chloroform exposure. I. Concentration in alveolar air and blood at
rest and during exercise. Scand. J. Work Environ. Health, 10: 69-81.
BASS (1970) Sudden sniffing death. J. Am. Med. Assoc., 212: 2075-2079.
BAUER, U. (1981) [Burden on man caused by harmful substances in the
environment.] Zbl. Bakteriol. Hyg. I Abt. Orig. B, 174: 200-237 (in
German).
BERODE, M., BOILLAT, M.-A., GUILLEMIN, M.P., WU, M.W., & SAVOLAINEN,
H. (1990) Demethylation pathways in caffeine metabolism as indicators
of variability in 1,1,1-trichloroethane oxidation in man. Pharmacol.
Toxicol., 67(1): 41-46.
BOBRA, A.M., SHIU, W.Y., & MACKAY, D. (1984) Structure-activity
relationships for toxicity of hydrocarbons, chlorinated hydrocarbons
and oils to Daphnia magna. In: Kaiser, K.L.E., ed. Quantitative
structure activity relations in environmental toxicology, Dordrecht,
Germany, D. Reidel Publishing Co., pp. 3-16.
BONNET, P., FRANCIN, J.-M., GRADISKI, D., RAOULT, G., & ZISSU, D.
(1981) Determination of the median lethal concentration of the
principal chlorinated aliphatic hydrocarbons in the rat. Arch. Mal.
prof. Méd. Trav. Sécur. soc., 41: 317-321.
BONVENTRE, J., BRENNAN, O., JASON, D., HENDERSON, A., & BASTOS, M.L.
(1977) Two deaths following accidental inhalation of dichloromethane
and 1,1,1-trichloroethane. J. anal. Toxicol., 4: 158-160.
BOUWER, E.J. & MCCARTY, P.L. (1983a) Transformations of 1- and
2-carbon halogenated aliphatic organic compounds under methanogenic
conditions. Appl. environ. Microbiol., 45: 1286-1294.
BOUWER, E.J. & MCCARTY, P.L. (1983b) Transformations of halogenated
organic compounds under denitrification conditions. Appl. environ.
Microbiol., 45: 1295-1299.
BOYER, J.D, AHLERT, R.C., & KOSSON, D.S. (1987) Degradation of
1,1,1-trichloroethane in bench-scale bioreactors. Hazardous Waste
hazardous Matter, 4: 241-260.
BRETHERICK, L., ed. (1981) Hazards in the chemical laboratory, 3rd
ed., London, The Royal Society of Chemistry.
BRETHERICK, L. (1989) 1,1,1-Trichloroethane. Chem. Eng. News, Dec. 12:
2.
BRINGMANN, G. & KUHN, R. (1977) [Limit values for the harmful effect
of water-endangering substances on bacteria (Psuedomonas putida) and
green algae (Scenedesmus quadricauda) in the cell division
inhibition test.] Z. Wasser Abwasser Forsch., 10: 87-98 (in German).
BRINGMANN, G. & KUHN, R. (1978) [Limit values for the harmful effect
of water endangering substances on blue algae (Microcystis
aeroginosa) and green algae (Scenedesmus quadricauda) in the cell
division inhibition test.] Vom Wasser, 50: 45-60 (in German).
BUCCAFUSCO, R.J., ELLS, S.J., & LEBLANC, G.A. (1981) Acute toxicity of
priority pollutants to bluegill (Lepomis macrochirus). Bull.
environ. Contam. Toxicol., 26: 446-452.
CAPLAN, Y.H., BACKER, R.C., & WHITAKER, J.Q. (1976)
1,1,1-Trichloroethane: report of a fatal intoxication. Clin. Toxicol.,
9: 69-74.
CEC (Commission of the European Communities) (1986) Organochlorine
solvents: Health risks to workers, Cambridge, Royal Society of
Chemistry, pp. 57-58.
CEFIC (1986) The occurrence of chlorinated solvents in the
environment. Prepared by a workshop of the European Council of
Chemical Industry Federation (CEFIC). Chem. Ind., 1986: 861-869.
CHIOU, C.T., PETERS, L.J., & FREED, V.H. (1979) A physical concept of
soil-water equilibria for nonionic organic compounds. Science, 206:
831-832.
CLARK, D.G. & TINSTON, D.J. (1982) Acute inhalation toxicity of some
halogenated and non-halogenated hydrocarbons. Hum. Toxicol., 1:
239-247.
CLASS, T. & BALLSCHMITER, K. (1986) Chemistry of organic traces in air
VI: Distribution of chlorinated C1 - C4 hydrocarbons in air over the
Northern and Southern Atlantic ocean. Chemosphere, 15: 413-427.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology, 3rd revised ed., New York, Chichester, John Wiley and
Sons, Vol. 2B.
COLEMAN, W.E., LINGG, R.D., MELTON, R.G., & KOPFLER, F.C. (1976) The
occurrence of volatile organics in five drinking water supplies using
gas chromatography/mass spectrometry. In: Keith, L.H., ed.
Identification and analysis of organic pollutants in water, Ann Arbor,
Michigan, Ann Arbor Science Publishers, Inc., pp. 305-327.
CORREIA, Y., MARTENS, G.J., VAN MESCH, F.H., & WHIM, B.P. (1977) The
occurrence of trichloroethylene, tetrachloroethylene and
1,1,1-trichloroethane in western Europe in air and water. Atmos.
Environ., 11: 1113-1116.
DAFT, J.L. (1988) Fumigant contamination during large-scale food
sampling for analysis. Arch. environ. Contam. Toxicol., 17: 177-181.
DAHLBERG, J.A., CHRISTIANSEN, V.O., & ERIKSEN, E.A. (1973) On the
formation of phosgene by photooxidation of methyl chloroform in
welding. Ann. occup. Hyg., 16: 41-46.
DALLAS, C.E., RAMANATHAN, R., MURALIDHARA, S., GALLO, J.M., &
BRUCKNER, J.V. (1989) The uptake and elimination of
1,1,1-trichloroethane during and following inhalation in rats.
Toxicol. appl. Pharmacol., 98: 385-397.
DANIEL, M.R. & DEHNEL, J.M. (1981) Cell transformation test with baby
hamster kidney cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 626-637 (Progress in Mutation Research, Vol. 1).
DANIELSSON, B.R.G., GHANTOUS, H., & DENCKER, L. (1986) Distribution of
chloroform and methylchloroform and their metabolites in pregnant
mice. Biol. Res. Pregnancy, 7: 77-83.
DAPSON, S.C., HUTCHEON, D.E., & LEHR, D. (1984) Effect of methyl
chloroform on cardiovascular development in rats. Teratology, 29: 25A.
DE NEVERS, N. (1986) A fatal fire with "non-flammable" methyl
chloroform. Arch. environ. Health, 41(5): 279-287.
DE SERRES, F.J. & ASHBY, J., ed. (1981) Evaluation of short term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland (Progress in
Mutation Research, Vol. 1).
DEVER, R.J. (1986) Responding to industrial contamination of
groundwater: a case study. J. Am. Water Works Assoc., 78: 82-86.
DICKSON, A.G. & RILEY, J.P. (1976) The distribution of short-chain
halogenated aliphatic hydrocarbons in some marine organisms. Mar.
Pollut. Bull., 7: 167-169.
DILLING, W.L., TERERTILLER, N.B., & KALLOS, G.J. (1975) Evaporation
rates and reactivities of methylene chloride, chloroform,
1,1,1-trichloroethane, trichloroethylene, tetrachloroethylene, and
other chlorinated compounds in dilute aqueous solutions. Environ. Sci.
Technol., 9: 833-838.
DORNETTE, W.H.L. & JONES, J.P. (1960) Clinical experience with
1,1,1-trichloroethane. Anesthesiol. Analg., 39: 249-253.
DRAKE, R.J. (1987) Fate of methylene chloride and
1,1,1-trichloroethane in unsaturated, biologically active soils,
University of Massachusetts (Dissertation).
DREHER, E.-L. (1989) Chlorinated hydrocarbons. In: Ullmann's
encyclopedia of chemical technology, New York, Weinheim, VCH
Publishers, Inc., Vol. A6, pp. 271-272.
DROZ, P.O., WU, M.M., CUMBERLAND, W.G., & BERUDE, M. (1989)
Variability in biological monitoring of solvent exposure. I
Development of a population physiological model. Br. J. ind. Med., 46:
447-460.
DUPRAT, P., DELSAUT, L., & GRADISKI, D. (1976) Pouvoir irritant des
principaux solvants chlorés aliphatiques sur la peau et les muqueuses
oculaires du lapin. Eur. J. Toxicol. environ. Hyg., 9: 171-177.
DÜSZELN, J.V., VON, LAHL, U., BÄTZER, K., CETINKAYA, M., STACHEL, B.,
& THIEMANN, W. (1982) [Volatile halogenated hydrocarbons in air, water
and food in the Federal Republic of Germany.] Dtsch. Lebensm.
Rundsch., 78: 352-356 (in German).
DÜSZELN, J.V., VON & THIEMANN, W. (1985) Volatile chlorinated
hydrocarbons in a costal urban atmosphere. Sci. total Environ., 41:
187-194.
EBEN, A. & KIMMERLE, G. (1974) Metabolism, excretion and toxicology of
methylchloroform in acute and subacute exposed rats. Arch. Toxicol.,
31: 233-242.
EDWARDS, P.R., CAMPBELL, I., & MILNE, G.S. (1982) The impact of
chloromethanes on the environment. Part 1. The atmospheric chlorine
cycle. Chem. Ind., 16: 574-578.
ELOVAARA, E., HEMMINKI, K., & VAINIO, H. (1979) Effects of methylene
chloride, trichloroethane, trichloroethylene and toluene on the
development of chick embryos. Toxicology, 12: 111-119.
ENTZ, R.C. & DIACHENKO, G.W. (1988) Residues of volatile halocarbons
in margarines. Food Addit. Contam., 5: 267-276.
ENTZ, R.C., THOMAS, K.W., & DIACHENKO, G.W. (1982) Residues of
volatile halocarbons in foods using headspace gas chromatography. J.
agric. food Chem., 30: 846-849.
FDA (1973) Trichloroethane. Aerosol drug products for human use. Fed.
Reg., 38: 21935-21936.
FDA (1977) Trichloroethane: status as a new drug in aerosolized drug
products intended for inhalation. FDA Final Rule. Fed. Reg., 42:
63386-63387.
FIELDING, M., GIBSON, T.M., JAMES, H.A., MCLOUGHLIN, K., & STEEL, C.P.
(1981) Organic micropollutants in drinking water, Medmenham, United
Kingdom, Water Research Centre (Technical Report No. TR 159).
FISCHER, M., FRIESEL, P., KOCH, G., MILDE, G., & ROSSKAMP, E. (1982)
Evaluation of water pollution caused by chlorinated and brominated
ethanes (Unpublished report submitted to the Commission of the
European Communities under Contract No. U71/516).
FISHBEIN, L. (1979) Potential halogenated industrial carcinogenic and
mutagenic chemicals. II. Halogenated saturated hydrocarbons. Sci.
total Environ., 11: 163-195.
FISHER, D.A., HALES, C.H., FILKIN, D.L., KO, M.K.W., SZE, N.D.,
CONNELL, P.S., WUEBBLES, D.J., ISAKSEN, I.S.A., & STORDAL, F. (1990a)
Model calculations of the relative effects of CFCs and their
replacement on stratospheric ozone. Nature (Lond.), 344: 508-512.
FISHER, D.A., HALES, C.H., WANG, W.-C., KO, M.K.W., & SZE, N.D.
(1990b) Model calculations of the relative effects of CFCs and their
replacement on global warming. Nature (Lond.), 344: 513-516.
FLUKA (1988) Catalogue 1988/89: Fluka Chemika-BioChemika, Buchs,
Switzerland, Fluka Chemie AG.
GALLOWAY, S.M., ARMSTRONG, M.J., REUBEN, C., COLMAN, S., BROWN, B.,
CANNON, C., BLOOM, A.D., NAKAMURA, F., AHMED, M., DUK, S. ET AL.
(1987) Chromosome aberrations and sister chromatid exchanges in
Chinese hamster ovary cells: Evaluations of 108 chemicals. Environ.
mol. Mutagen., 10: 1-175.
GAMBERALE, F. & HULTENGREN, M. (1973) Methylchloroform exposure II.
Psychophysiological functions. Scand. J. Work Environ. Health, 10:
82-92.
GEORGE, J.D., PRICE, C.J., MARR, M.C., SADLER, B.M., SCHWETZ, B.A.,
BIRNBAUM, L.S., & MORRISEY, R.E. (1989) Developmental toxicity of
1,1,1-trichloroethane in CD rats. Fundam. appl. Toxicol., 13, 641-651.
GERACE, R.V. (1981) Near-fatal intoxication by 1,1,1-trichloroethane.
Ann. Emergency Med., 10: 533-534.
GERKENS, R.R. & FRANKLIN, J.A. (1989) The rate of degradation of
1,1,1-trichloroethane in water by hydrolysis and dehydrochlorination.
Chemosphere, 19: 1919-1937.
GIGER, W., MOLNAR-KUBICA, E., & WAKEHAM, S. (1978) Volatile
chlorinated hydrocarbons in ground and lake waters. In: Aquatic
pollutants: Transformation and biological effects. Proceedings of the
2nd International Symposium on Aquatic Pollutants, Oxford, New York,
Pergamon Press, pp. 101-123 (Pergamon Series on Environmental Science
No. 1).
GOCKE, E., KING, M.-T., ECKHARDT, K., & WILD, D. (1981) Mutagenicity
of cosmetics ingredients licensed by the European Communities. Mutat.
Res., 90: 91-109.
GOLDBERG, S.J., LEBOWITZ, MD., GRAVER, E.J., & HICKS, S. (1990) An
association of human congenital cardiac malformations and
drinking-water contaminants. J. Am. Coll. Cardiol., 16: 155-164.
GOSSETT, J.M. (1985) Anaerobic degradation of C1 and C2 chlorinated
hydrocarbons, Tyndall AFB, Florida, US Air Force Engineering Services
Center (Technical Report No. ESL-TR-85-38).
GOSSETT, R.W., BROWN, D.A., & YOUNG, D.R. (1983) Predicting the
bioaccumulation of organic compounds in marine organisms using
octanol/water partition coefficients. Mar. Pollut. Bull., 14: 387-392.
GOTO, M. (1979) Monitoring environmental materials and specimen
banking. State-of-the-art of Japanese experience and knowledge. In:
Monitoring environmental materials and specimen banking, Proc. Int.
Workshop, Berlin (West) 23-28 October 1978, The Hague, Nijhoff
Publishers, pp. 271-288.
GRADISKI, D., MAGADUR, J.-L., BAILLOT, M., DANIERE, M.C., & SCHUH,
M.B. (1974) Toxicité comparée des principaux solvants chlorés
aliphatiques. J. eur. Toxicol., 7: 247-254.
GRADISKI, D., BONNET, P., RAOULT, G., MAGADUR, J., & FRANCIN, J.M.
(1978) Compared acute pulmonary toxicity of the main chlorinated
aliphatic solvents. Arch. Mal. prof. Méd. Trav. Sécur. soc., 39:
249-257.
GREEN, M.H. (1981) Differential killing test using an improved
repair-deficient strain of Escherichia coli. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
Oxford, Elsevier/North Holland, pp. 183-194 (Progress in Mutation
Resarch, Vol. 1).
GRIMSRUD, E.P. & RASMUSSEN, R.A. (1975) Survey and analysis of
halocarbons in the atmosphere by gas chromatography-mass spectrometry.
Atmos. Environ., 9: 1014-1017.
HAAG, W.R. & MILL, T. (1988) Effect of a subsurface sediment on
hydrolysis of haloalkanes and epoxides. Environ. Sci. Technol., 22:
658-663.
HAAG, W.R., MILL, T., & RICHARDSON, A. (1986) Extended abstracts of
the 192nd National Meeting of the American Chemical Society, Anaheim,
Washington, DC, American Chemical Society, Division of Environmental
Chemistry, Vol. 26, pp. 248-253.
HAKE, C.L., WAGGONER, T.B., ROBERTSON, D.N., & ROWE, V.K. (1960) The
metabolism of 1,1,1-trichloroethane by the rat. Arch. environ. Health,
1: 101-105.
HALL, F.B. & HINE, C.H. (1966) Trichloroethane intoxication: a report
of two cases. J. Forensic Sci., 11: 404-413.
HARDIE, D.W.F. (1964) 1,1,1-Trichloroethane. In: Kirk, R.E. & Othmer,
D.F., ed. Encyclopedia of chemical technology, 2nd ed., New York,
Chichester, Wiley and Sons, Vol. 5, pp. 154-157, 170.
HATCH, G.G., MAMAY, P.P., AYER, M.L., CASTO, B.C., & NESNOW, S. (1983)
Chemical enhancement of viral transformation in Syrian hamster embryo
cells by gaseous volatile chlorinated methanes and ethanes. Cancer
Res., 43: 1945-1950.
HATFIELD, T.R. & MAYKOSKI, R.T. (1970) A fatal methyl chloroform
(trichloroethane) poisoning. Arch. environ. Health, 20: 279-281.
HEITMULLER, P.T., HOLLISTER, T.A., & PARRISH, P.R. (1981) Acute
toxicity of 54 industrial chemicals to sheepshead minnows (Cyprinodon
variegatus) Bull. environ. Contam. Toxicol., 27: 596-604.
HENSCHLER, D., ed. (1978) [German Association for the Encouragement of
Research: Analytical methods for the testing of materials harmful to
health. I. Air analyses], Weinheim, Verlag Chemie (in German).
HENSCHLER, D., REICHERT, D., & METZLER, M. (1980) Identification of
potential carcinogens in technical grade 1,1,1-trichloroethane. Int.
Arch. occup. environ. Health, 47: 263-268.
HERD, P.A., LIPSKY, M., & MARTIN, H.F. (1974) Cardiovascular effects
of 1,1,1-trichloroethane. Arch. environ. Health, 28: 227-233.
HOBARA, T., KOBAYASHI, H., HIGASHIHARA, E., IWAMOTO, S., KAWAMOTO, T.,
SAKAI, T., & TSUBOTA, N. (1982) [Experimental examinations and
toxicokinetic analysis of the absorption and excretion of
1,1,1-trichloroethane by the lung.] Jpn. J. ind. Health, 24: 599-607
(in Japanese).
HODGSON, M.J., HEYL, A.E., & VAN THIEL, D.H. (1989) Liver disease
associated with exposure to 1,1,1-trichloroethane. Arch. intern. Med.,
149: 1791-1798.
HORIGUCHI, S. & HORIGUCHI, K. (1971) An experiment of
1,1,1-trichloroethane vapour exposure to mice. Sangyo Igaku, 13:
226-227.
HSE (1984) 1,1,1-Trichloroethane, London, Health and Safety Executive.
HSE (1990) Guidance note EH40/90. Occupational exposure limits 1990,
London, Health and Safety Executive.
HUMBERT, B.E. & FERNANDEZ, J.G. (1977) Exposition au
1,1,1-trichloroéthane: Contribution à l'étude de l'absorption, de
l'excrétion et du métabolisme sur des sujets humains. Arch. Mal.
prof., 38: 415-425.
HUTCHINSON, T.C., HELLEBUST, J.A., TAM, D., MACKAY, D., MASCARENHAS,
R.A., & SHIU, W.Y. (1980) The correlation of the toxicity to algae of
hydrocarbons and halogenated hydrocarbons with their physical-chemical
properties. Environ. Sci. Res., 1: 577-586.
IARC (1979) 1,1,1-Trichloroethane. In: Some halogenated hydrocarbons,
Lyon, International Agency for Research on Cancer, pp. 515-532 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Vol. 20).
IARC (1987) Overall evaluations of carcinogenicity: An updating of
IARC Monographs, Volumes 1 to 42, Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluations of Carcinogenic
Risk to Humans, Suppl. 7).
ICHINOTSUBO, D., MOWER, H., & MANDEL, M. (1981) Mutagen testing of a
series of paired compounds with the Ames Salmonella testing system.
In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland, pp. 298-301
(Progress in Mutation Research. Vol. 1).
IKEDA, M. & OHTSUJI, H. (1972) A comparative study of the excretion of
Fujiwara reaction-positive substances in urine of humans and rodents
given trichloro- or tetrachloro-derivatives of ethane and ethylene.
Brit. J. ind. Med., 29: 99-104.
IVANETICH, K.M. & HONERT, L.H., VAN DEN (1981) Chloroethanes: their
metabolism by hepatic cytochrome P-450. Carcinogenesis, 2: 697-702
JAKOBSON, M., LIEPINS, M., & ERINS, P. (1980) [Effect of changes in
the reticular structure of a lignocarbohydrate matrix on the
properties of the wood. 2. Effect of cross-linking on the
hydrophilicity of birchwood.] Khim. Drev., 1: 37-46 (in Russian).
JAKOBSON, I., WAHLBERG, J.E., HOLMBERG, B., & JOHANSSON, G. (1982)
Uptake via the blood and elimination of 10 organic solvents following
epicutaneous exposure of anesthetized guinea-pigs. Toxicol. appl.
Pharmacol., 63: 181-187.
JONES, R.D. & WINTER, D.P. (1983) Two case reports of deaths on
industrial premises attributed to 1,1,1-trichloroethane. Arch.
environ. Health, 38: 59-61.
KADA, T. (1981) DNA-damaging activity of 42 coded compounds in the
rec-assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 175-182 (Progress in Mutation Research, Vol. 1).
KAJINO, M. & YAGI, M. (1980) Formation of trihalomethanes during
chlorination and determination of halogenated hydrocarbons in drinking
water. In: Hydrocarbons and halogenated hydrocarbons in the aquatic
environment, New York, London, Plenum Press.
KARLSSON, J.-E., ROSENGREN, L.E., KJELLSTRAND, P., & HAGILD K.G.
(1987) Effect of low-dose inhalation of three chlorinated aliphatic
organic solvents on DNA in gerbil brain. Scand. J. Work Environ.
Health, 13: 453-458.
KHALIL, M.A. & RASMUSSEN, R.A. (1981) Decline in the atmospheric
accumulation rates of CCl3F(F-11), CCl2F2(F-12) and CH3CCl3.
J. Air Pollut. Control Assoc., 31: 1274-1275.
KHALIL, M.A. & RASMUSSEN, R.A. (1984) Methylchloroform: Global
distribution, seasonal cycles and anthropogenic chlorine. Chemosphere,
13: 789-800.
KING, G.S., SMIALEK, J.E., & TROUTMAN, W.G. (1985) Sudden death in
adolescent resulting from the inhalation of typewriter correction
fluid. J. Am. Med. Assoc., 253: 1604-1606.
KLAASSEN, C.D. & PLAA, G.L. (1966) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in mice.
Toxicol. appl. Pharmacol., 9: 139-151.
KLAASSEN, C.D. & PLAA G.L. (1967) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in dogs.
Toxicol. appl. Pharmacol., 10: 119-131.
KLECKA, G.M., GONSIOR, S.J., & MARKHAM, D.A. (1990) Biological
transformations of 1,1,1-trichloroethane in subsurface soils and
ground water. Environ. Toxicol. Chem., 9: 1437-1451.
KLEINFELD, M. & FEINER, B. (1966) Health hazard associated with work
in confined spaces. J. occup. Med., 8: 358-364.
KOBAYASHI, H., HOBARA, T., HIROTA, H., HIGASHIHARA, E., IWAMOTO, S.,
& SAKAI, T. (1982) Sensitization of dog myocardium to epinephrine by
1,1,1-trichloroethane. Jpn. J. ind. Health, 24: 450-454.
KOBAYASHI, H., HOBARA, T., HIROTA, H., & SAKAI, T. (1983) Neural
control of blood pressure following 1,1,1-trichloroethane inhalation:
A role of sympathetic nervous system. Arch. environ. Health, 38:
93-98.
KOBAYASHI, H., HOBARA, T., SATOH, T., & SAKAI, T. (1986) Respiratory
disorders following 1,1,1-trichloroethane inhalation: A role of reflex
mechanism arising from lungs. Arch. environ. Health, 41: 149-154.
KOBAYASHI, H., HOBARA, T., KAWAMOTO, T., & SAKAI, T. (1987) Effect of
1,1,1-trichloroethane inhalation on heart rate and its mechanism: A
role of autonomic nervous system. Arch. environ. Health, 42: 140-143.
KONEMANN, H. (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part I. Relationship for 50 industrial
pollutants. Toxicology, 19: 209-221.
KRAMER, C.G., OTT, M.G., FULKERSON, J.E., & HICKS, N. (1978) Health of
workers exposed to 1,1,1-trichloroethane: A matched-pair study. Arch.
environ. Health, 33: 331-342.
KRONELD, R. (1989) Volatile pollutants in the environment and human
tissues. Bull. environ. Contam. Toxicol., 42: 873-877.
KRONEVI, T., WAHLBERG, J.E., & HOLMBERG, B. (1981) Skin pathology
following epicutaneous exposure to seven organic solvents. Int. J.
Tissue React., 3: 21-30.
KUKONGVIRIYAPAN, V., KUKONGVIRIYAPAN, U., & STACEY, N.H. (1990)
Interference with hepatocellular substrate uptake by
1,1,1-trichloroethane and tetrachloroethylene. Toxicol. appl.
Pharmacol., 102: 80-90.
KYRKLUND, T., KJELLSTRAND, P., & HAGLID, K.G. (1988) Effects of
exposure to Freon 11, 1,1,1-trichloroethane or perchloroethylene on
the lipid and fatty-acid composition in rat cerebral cortex. Scand. J.
Work Environ. Health, 14: 91-94.
LANE, R.W., RIDDLE, B.L., & BORZELLECA, J.F. (1982) Effects of
1,2-dichloroethane and 1,1,1-trichloroethane in drinking water on
reproduction and development in mice. Toxicol. appl. Pharmacol., 63:
409-421.
LEBLANC, G.A. (1980) Acute toxicity of priority pollutants to water
flea (Daphnia magna). Bull environ. Contam. Toxicol., 24: 684-691.
LEONG, B.K.J., SCHWETZ, B.A., & GEHRING, P.J. (1975) Embryo- and
fetotoxicity of inhaled trichloroethylene, perchloroethylene,
methylchloroform, and methylene chloride in mice and rats. Toxicol.
appl. Pharmacol., 33: 136 (Abstract 34).
LILLIAN, D., SINGH, H.B., APPLEBY, A., LOBBAN, L., ARNTS, R., GUMPERT,
R., HAGUE, R., TOOMEY, J., KAZASIS, J., ANTELL, M., HANSEN, D., &
SCOTT, B. (1975) Atmospheric fates of halogenated compounds. Environ.
Sci. Technol., 9: 1043-1048.
LINDBOHM, M-L., TASKINEN, H., SALLMEN, M., & HEMMINKI, K. (1990)
Spontaneous abortions among women exposed to organic solvents. Am. J.
ind. Med., 17: 449-463.
LOPRIENO, N., BARATE, R., ROSSI, A.M., FUMERO, S., MERIGGI, G.,
MONDINO, A., & SILVEST S. (1979) In vivo mutagenicity studies with
trichloroethylene and other solvents, Ivrea, Italy, Istituto di
Ricerche Biomedicale
LUNDBERG, I., EKDAHL, M., KRONEVI, T., LIDUMS, V., & LUNDBERG, S.
(1986) Relative hepatotoxicity of some industrial solvents after
intraperitoneal injection or inhalation exposure in rats. Environ.
Res., 40: 411-420.
MABEY, W., BARICH, V., & MILL, T. (1983) Extended abstracts of the
186th Meeting of the American Chemical Society, Washington, DC,
American Chemical Society, Division of Environmental Chemistry, Vol.
23, pp. 359-361.
MCCARTHY, T.B. & JONES, R.D. (1983) Industrial gassing poisonings due
to trichloroethylene, perchlorethylene, and 1,1,1-trichloroethane,
1961-80. Br. J. ind. Med., 40: 450-455.
MCCONNELL, G., FERGUSON, D.M., & PEARSON, C.R. (1975) Chlorinated
hydrocarbons and the environment. Endeavour, 34: 13-18.
MCCONNELL, J.C. & SCHIFF, H.I. (1978) Methyl chloroform: Impact on
stratospheric ozone. Science, 199: 174-177.
MCDONALD, D.J. (1981) Salmonella/microsome tests on 42 coded
chemicals. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 285-297 (Progress in Mutation Research, Vol. 1).
MCEWEN, J.D. & VERNOT, E.H. (1974) The biological effect of continuous
inhalation exposure of 1,1,1-trichloroethane (methyl chloroform) on
animals. In: Toxic hazard research unit: Annual technical report 1974,
Ohio, Wright-Patterson Air-Force Base, Aerospace Medical Research
Laboratory, pp. 81-90 (AMRL-TR-74-78).
MACKAY, C.J., CAMPBELL, L., SAMUEL, A.M., ALDERMAN, K.J., IDZIKOWSKI,
C., WILSON, H.K., & COMPERTZ, D. (1987) Behavioral changes during
exposure to 1,1,1-trichloroethane: time-course and relationship to
blood solvents levels. Am. J. ind. Med., 11: 223-239.
MCLEOD, A.A., MARJOT, A., MONAGHAN, M.J., HUGH-JONES, P., & JACKSON,
G. (1987) Chronic cardiac toxicity after inhalation of
1,1,1-trichloroethane. Br. med. J., 214: 727-729.
MCNUTT, N.S., AMSTER, R.L., MCCONNELL, E.F., & MORRIS, F (1975)
Hepatic lesions in mice after continuous inhalation exposure to
1,1,1-trichloroethane. Lab. Invest., 5: 642-654.
MAGARA, Y. & FURUICHI, T. (1986) Environmental pollution by
trichloroethylene and tetrachloroethylene: a nationwide survey. In:
Chambers, P.L., Gehring, P., & Sakai, F., ed. New concepts and
developments in toxicology, Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp. 231-243.
MALTONI, C., COTTI, G., & PATELLA, V. (1986) Results of long-term
carcinogenicity bioassays on Sprague-Dawley rats of methyl chloroform,
administered by ingestion. Acta oncol., 7: 101-117.
MARONI, M.C., BULGGHERONI, C., CASSITTO, M.G., MERLUZZI, F., GILIOLO,
R., & FOA, V. (1977) A clinical, neurophysiological and behavioral
study of female workers exposed to 1,1,1-trichloroethane. Scand. J.
Work Environ. Health, 3: 16-22.
MARJOT, R. & MCLEOD, A.A. (1989) Chronic non-neurological toxicity
from volatile substance abuse. Hum. Toxicol., 8: 301-306.
MARTIN, C.N. & MCDERMID, A.C. (1981) Testing of 42 coded compounds for
their ability to induce unscheduled DNA repair synthesis in HeLa
cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term
tests for carcinogens. Report of the international collaborative
programme, New York, Amsterdam, Oxford, Elsevier/North Holland, pp.
533-537 (Progress in Mutation Research, Vol. 1).
MIDGLEY, P.M. (1989) The production and release to the atmosphere of
1,1,1-trichloroethane (methyl chloroform). Atmos. Environ., 23:
2663-2665.
MILLER, L.J. & UHLER, A.D. (1988) Volatile halocarbons in butter:
Elevated tetrachloroethylene levels in samples obtained in close
proximity to dry-cleaning establishments. Bull. environ. Contam.
Toxicol., 41: 469-474.
MITI (1990) The yearbook of chemical trade and industries statistics,
Tokyo, Ministry of International Trade and Industry.
MITOMA, C., STEEGER, T., JACKSON, S.E., WHEELER, K.P., ROGERS, J.H.,
& MILMAN, H.A. (1985) Metabolic disposition study of chlorinated
hydrocarbons in rats and mice. Drug chem. Toxicol., 8(3): 183-194.
MONSTER, A.C. (1979) Differences in uptake, elimination and metabolism
in exposure to trichloroethylene, 1,1,1-trichloroethane and
tetrachloroethylene. Int. Arch. occup. environ. Health, 42: 311-317.
MONSTER, A.C. (1986) Biological monitoring of chlorinated hydrocarbon
solvents. J. occup. Med., 28: 583-588.
MONSTER, A.C. & HOUTKOPPER, J.M. (1979) Estimation of individual
uptake of trichloroethylene, 1,1,1-trichloroethane, and
tetrachloroethylene from biological parameters. Int. Arch. occup.
environ. Health, 42: 319-323.
MONSTER, A..C., BOERSMA, G., & STEENWEG, H. (1979) Kinetics of
1,1,1-trichloroethane in volunteers: influence of exposure
concentration and work load. Int. Arch. occup. environ. Health, 42:
293-301.
MOSER, V.C. & BALSTER, R.L. (1985) Acute motor and lethal effects of
inhaled toluene, 1,1,1-trichloroethane, halothane, and ethanol in
mice: effects of exposure duration. Toxicol. appl. Pharmacol., 77:
285-291.
MULLIN, L.S. & KRIVANEK, N.D. (1982) Comparison of unconditioned
reflex and conditioned avoidance tests in rats exposed by inhalation
to carbon monoxide, 1,1,1-trichloroethane, toluene or ethanol.
Neurotoxicology, 3: 126-137.
MYHR, B.C. & CASPARY, W.J. (1988) Evaluation of the L5178Y mouse
lymphoma cell mutagenesis assay: Intralaboratory results for
sixty-three coded chemicals tested at Litton Bionetics, Inc. Environ.
mol. Mutagen., 13: 103-194.
NAGAO, M. & TAKAHASHI, Y. (1981) Mutagenic activity of 42 coded
compounds in the salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 302-313 (Progress in Mutation
Research, Vol. 1).
NAKAAKI, K., FUKABORI, S., & TADA, O. (1980) An experimental study on
percutaneous absorption of some organic solvents. J. Sci. Labour, 56:
1-9.
NCI (1977) Bioassay of 1,1,1-trichloroethane for possible
carcinogenicity, Bethesda, Maryland, National Cancer Institute
(Carcinogenesis Technical Report Series No. 3).
NEELY, W.B. (1982) Organizing data for environmental studies. Environ.
Toxicol. Chem., 1: 259-266.
NESTMANN, E.R., LEE, E.G.H., MATULA, T.I. DOUGLAS, G.R., & MUELLER,
J.C. (1980) Mutagenicity of constituents identified in pulp and paper
mill effluents using the Salmonella/mammalian-microsome assay.
Mutat. Res., 79: 203-212.
NEUHAUSER, E.F., LOEHR, R.C., & MALECKI, M.R. (1986) Contact and
artificial soil tests using earthworms to evaluate the impact of
wastes in soil. In: Petros, J.K., Lacy, W.J., & Conway, R.A., ed.
Hazardous and industrial solid waste testing: Fourth Symposium.
Philadelphia, American Society for Testing and Materials, pp. 192-203
(ASTM STP 886).
NIEF, L. & MERCIER, J. (1981) Skin pathology following epicutaneous
exposure to seven organic solvents. Int. J. Tissue React., 111: 21-30.
NILSSON, K.B. (1986a) Effects of 1,1,1-trichloroethane on the cGMP
metabolism in mouse brain. Acta pharmacol. toxicol., 58: 318-326.
NILSSON, K.B. (1986b) Actions of 1,1,1-trichloroethane on the cAMP
metabolism in mouse brain. Acta pharmacol. toxicol., 59: 362-369.
NILSSON, K.B. (1987) Effects of 1,1,1-trichloroethane on synaptosomal
calcium accumulation in mouse brain. Pharmacol. Toxicol., 61: 215-219.
NIOSH (1976) Criteria for a recommended standard: occupational
exposure to 1,1,1-trichloroethane (methyl chloroform), Cincinnati,
Ohio, National Institute for Occupational Safety and Health
(Publication DHEW (NIOSH) No. 76-184).
NIOSH (1977) NIOSH manual of analytical methods. Part II: Standards
completion program validated methods (Method 5328), 2nd ed.,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, pp. 5328/1-5328/9 (DHEW (NIOSH) Publication No. 77-157-B).
NIOSH (1979) Occupational exposure to 1,1,1-trichloroethane,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health.
NIOSH (1983) National occupational exposure survey, 1981-1983,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, Division of Surveillance, Hazard Evaluations and Field
Studies, Surveillance Branch, Hazard Section (Unpublished data).
NOLAN, R.J., FRESHOUR, N.L., RICH, D.L., MCCARTY, L.P., & SAUNDERS,
J.H. (1984) Kinetics and metabolism of inhaled methyl chloroform
(1,1,1-trichloroethane) in male volunteers. Fundam. appl. Toxicol., 4:
654-662.
NORTHFIELD, R.R. (1981) Avoidable deaths due to acute exposure to
1,1,1-trichloroethane. J. Soc. Occup. Med., 31: 164-166.
NRC (1976) Halocarbons. Effects on stratospheric ozone, Washington,
DC, National Research Council, Panel on Atmospheric Chemistry. OTSON,
R. (1987) Purgeable organics in Great Lakes raw and treated water.
Int. J. environ. anal. Chem., 31: 41-53.
OTSON, R. & CHAN, C. (1987) Sample handling and analysis for 51
volatile organics by an adapted purge and trap GC-MS technique. Int.
J. environ. anal. Chem., 30: 275-287.
OTSON, R., WILLIAMS, D.T., & BOTHWELL, P.D. (1984) Fabric protectors.
Part II - Propane, 1,1,1-trichloroethane and petroleum distillates in
air after application of fabric protectors. Am. Ind. Hyg. Assoc. J.,
45: 28-33.
PARSONS, F. & LAGE, G.B. (1985) Chlorinated organics in simulated
groundwater environments. J. Am. Water Works Assoc., 77: 52-59.
PARSONS, F., LAGE, G.B., & RICE, R. (1985) Biotransformation of
chlorinated organic solvents in static microcosms. Environ. Toxicol.
Chem., 4: 739-742.
PEARSON, C.R. (1982) C1 and C2 halocarbons. In: Hutzinger, O., ed.
The handbook of environmental chemistry, Berlin, Heidelberg, New York,
Springer-Verlag, Vol. 3, Part B, pp. 69-88.
PEARSON, C.R. & MCCONNELL, G. (1975) Chlorinated C1 and C2
hydrocarbons in the marine environment. Proc. R. Soc. Lond., B189:
305-332.
PEKARI, K. & AITIO, A. (1985) Method 26. Determination of
1,1,1-trichloroethane in blood. In: Fishbein, L. & O'Neil, I.K., ed.
Environmental carcinogens selected methods of analysis. Volume 7 -
Some volatile halogenated hydrocarbons, Lyon, International Agency for
Research on Cancer, pp. 445-450 (IARC Scientific Publications No. 68).
PELLIZARI, E.D., HARTWELL, T.D., HARRIS, B.S.H., WADDELL, R.D.,
WHITAKER, D.A., & ERICKSON, M.D. (1982) Purgeable organic compounds in
mothers' milk. Bull. environ. Contam. Toxicol., 28: 322-328.
PEREIRA, W.E. & HUGHES, B.A. (1980) Determination of selected volatile
organic priority pollutants in water by computerized gas
chromatography - quadruple mass spectrometry. J. Am. Water Assoc., 72:
220-230.
PERRY, P.E. & THOMSON, E.J. (1981) Evaluation of the sister-chromatid
exchange method in mammalian cells as a screening system for
carcinogens. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 560-569 (Progress in Mutation Research, Vol. 1).
PFANNHAUSER, W., GOMBOS, J., & THALLER, A. (1988) [Halogenated
aliphatic hydrocarbons in food: Investigations on enrichment and
content in fat containing food.] Ernährung, 12: 611-615 (in German).
PIET, G.J., SLINGERLAND, P., DE GRUNT, F.E., HEUVEL, V.D., & ZOETEMAN,
B.C.J. (1978) Determination of very volatile halogenated organic
compounds in water by means of direct head-space analysis. Anal.
Lett., 11: 437-448.
PRENDERGAST, J.A., JONES, R.A., JENKINS, L.J., & SIEGEL, J. (1967)
Effects on experimental animals of long-term inhalation of
trichloroethylene, carbon tetrachloride, 1,1,1-trichloroethane,
dichlorodifluoromethane, and 1,1-dichloroethylene. Toxicol. appl.
Pharmacol., 10: 270-289.
PRICE, P.J., HASSETT, C.M., & MANSFIELD, J.I. (1978) Transforming
activities of trichloroethylene and proposed industrial alternatives.
14: 290-293.
PRINN, R., CUNNOLD, D., RASMUSSEN, R., SIMMONDS, P., ALYEA, F.,
CRAWFORD, A., FRASER, P., & ROSEN, R. (1987) Atmospheric trends in
methylchloroform and the global average for the hydroxyl radical.
Science, 238: 945-950.
PRODI, G., COLLACCI, A., GRILLI, S., LATTANZI, G., MAZZULO, M., &
TURINA P. (1988) Comparison of the covalent binding of various
chloroethanes with nucleic acids. In: Feo, F., ed. Chemical
carcinogenesis (models and mechanisms), New York, Plenum Press, pp.
93-102.
QUAST, J.F., RAMPY, L.W., BALMER, M.F., LEONG, B.D.J., & GEHRING P.J.
(1978) Toxicological and carcinogenic evaluation of a
1,1,1-trichloroethane formulation by chronic inhalation in rats,
Midland, Michigan, Dow Chemical Co.
QUAST, J.F., CALHOUN, L.L., & FRAUSON, L.E. (1988)
1,1,1-Trichloroethane formulation: a chronic inhalation toxicity and
oncogenicity study in Fischer 344 rats and B6C3F1 mice. Fundam. appl.
Toxicol., 11: 611-625.
RAJENDRAN, S. (1990) The toxicity of phosphine, methyl bromide,
1,1,1-trichloroethane and carbon dioxide alone and as mixtures to the
pupae of red flour beetle, Tribolium castaneum, Herbst. Pestic.
Sci., 29: 75-83.
RAMPY, L.W., QUAST, J.F., LEONG, B.K.J., & GEHRING, P.J. (1977)
Results of long-term inhalation studies on rats of
1,1,1-trichloroethane and perchloroethylene formulations. In: Plaa,
G.L. & Duncan, W.A.M., ed. Proceedings of the 1st International
Congress on Toxicology, Toronto, New York, London, Academic Press, p.
27 (Abstract).
RASMUSSEN, R.A. & KHALIL, M.A.K. (1982) Latitudinal distributions of
trace gases in and above the boundary layer. Chemosphere, 11: 117-235.
RASMUSSEN, R.A. & KHALIL, M.A.K. (1983) Natural and anthropogenic
trace gases in and above the boundary layer. Chemosphere, 12: 371-375.
RASMUSSEN, R.A., KHALIL, M.A.K., & DALLAGUE, R.W. (1981) Atmospheric
trace gases in Antarctica. Science, 211: 185-287.
REINHARDT, C.F., MULLIN, L.S., & MAXFIELD, M.E. (1973)
Epinephrine-induced cardiac arrhythmia potential of some common
industrial solvents. J. occup. Med., 15: 953-955.
REITZ, R.H., MCDOUGAL, J.N., HIMMELSTEIN, M.W., NOLAN, R.J., &
SCHUMANN, A.M. (1988) Physiologically based pharmacokinetic modelling
with methylchloroform: Implications for interspecies, high dose/low
dose and dose route extrapolations. Toxicol. appl. Pharmacol., 95:
185-199.
RENNICK, R.B., MALTON, S.D., MOW, G.K., & SEEVERS, M.H. (1949)
Induction of idioventricular rhythms by 1,1,1-trichloroethane and
epinephrine. J. Pharmacol. exp. Ther., 94: 327.
RICHOLD, M. & JONES, E. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluations of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 314-322 (Progress in Mutation
Research, Vol. 1).
RIIHIMAKI, V. & PFAFFLI, P (1978) Percutaneous absorption of solvent
vapor in man. Scand. J. Work Environ. Health, 4: 73-85.
RIVETT, M.O., LERNER, D.N., & LLOYD, J.W. (1990) Chlorinated solvents
in U.K. aquifers. J. Inst. Water Environ. Manage., 4: 242-250.
ROSENGREN, L.E., AURELL, A., KJELLSTRAND, P., & HAGLID, K.G. (1985)
Astrogliosis in the cerebral cortex of gerbils after long-term
exposure to 1,1,1-trichloroethane. Scand. J. Work Environ. Health, 11:
447-455.
ROSENKRANZ, H.S., HYMAN, J., & LEIFER, Z. (1981) DNA polymerase
deficient assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 210-218 (Progress in Mutation Research, Vol. 1).
ROWLAND, I. & SEVERN, B. (1981) Mutagenicity of carcinogens and
noncarcinogens in the Salmonella/microsome test. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 323-332 (Progress in
Mutation Research, Vol. 1).
RUSSELL, J.W. & SHADOFF, L.A. (1977) The sampling and determination of
halocarbons in ambient air using concentration on porous polymer. J.
Chromatogr., 134: 375-384.
SALAMONE, M.F., HEDDLE, J.A., & KATZ M. (1981) Mutagenic activity of
41 compounds in the in vivo micronucleus assay. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 686-697 (Progress in
Mutation Research, Vol. 1).
SALVINI, M., BINASCHI, S., & RIVA, M. (1971) Evaluation of the
psychophysiological functions in humans exposed to the "threshold
limit value" of 1,1,1-trichloroethane. Br. J. ind. Med., 28: 286-292.
SATO, A., NAKAJIMA, T., & KOYAMA, Y. (1980) Effects of chronic ethanol
consumption on hepatic metabolism of aromatic and chlorinated
hydrocarbons in rats. Br. J. ind. Med., 37: 382-386.
SAVOLAINEN, H., PFAFFLI, P., TENGEN, M., & VAINIO, H. (1977)
Trichloroethylene and 1,1,1-trichloroethane: effects on brain and
liver activity after five days intermittent inhalation. Arch.
Toxicol., 38: 229-237.
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1981)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Int. Arch. occup. environ. Health, 49: 89-98.
SAVOLAINEN, K., RIIHIMÄKI, V., & LAINE A. (1982a) Biphasic effects of
inhaled solvents on human equilibrium. Acta pharmacol. toxicol., 51:
237-242
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1982b)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Arch. Toxicol., Suppl. 5: 96-99.
SCHUMANN, A.M., FOX, T.R., & WATANABE, P.G. (1982) [14C]-Methyl
chloroform (1,1,1-trichloroethane): Pharmacokinetics in rats and mice
following inhalation exposure. Toxicol. appl. Pharmacol., 62: 390-401.
SEKI, Y., URASHIMA, Y., AIKAWA, H., MATSUMURA, H., ICHIKAWA, Y.,
HIRATSUKA, F., YOSHIOKA, Y., SHIMBO, S., & IKEDA, M. (1975)
Trichloro-compounds in the urine of humans exposed to methyl
chloroform at sub-threshold levels. Int. Arch. Arbeitsmed., 34: 39-49.
SHIMADA, T., SWANSON, A.F., LEBER, P., & WILLIAMS, G.M. (1985)
Activities of chlorinated ethane and ethylene compounds in the
Salmonella/rat microsome mutagenesis and rat hepatocyte/DNA repair
assays under vapor phase exposure conditions. Cell Biol. Toxicol., 1:
159-179.
SHMUTER, L.M. (1977) [The effect of chronic exposure to low
concentrations of chlorinated ethane series hydrocarbons on specific
and non-specific immunological response in animals under experimental
conditions.] Gig. Tr. prof. Zabol., 8: 38-43 (in Russian).
SIEGEL, J., JONES, R.A., COON, R.A., & LYON J.P. (1971) Effects on
experimental animals of acute, repeated and continuous inhalation
exposures to dichloroacetylene mixtures. Toxicol. appl. Pharmacol.,
18: 168-174.
SIMMON, V.F. & SHEPHERD, G.F. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 333-342 (Progress in Mutation
Research, Vol. 1).
SIMMON, V.F., KAUHANEN, K., & TARDIFF, R.G. (1977) Mutagenic activity
of chemicals identified in drinking water. In: Scott, D., Bridges,
B.A., & Sobels, F.H., ed. Progress in genetic toxicology, Amsterdam,
Elsevier/North Holland, pp. 249-258.
SIMMONDS, P.G., KERRIN, S.L., LOVELOCK, J.E., & SHAIR, F.H. (1974)
Distribution of atmospheric halocarbons in the air over the Los
Angeles basin. Atmos. Environ., 8: 209-216.
SINGH, H.B., SALAS, L.J., & STILES, R.E. (1982) Distribution of
selected gaseous organic mutagens and suspect carcinogens in ambient
air. Environ. Sci. Technol., 16: 982-880.
SNOW, L.P., MCNAIR, B.C., & CASTRO B.C. (1979) Mutagenesis testing of
1,1,1-trichloroethane in Salmonella strains TA100 and TA98, Research
Triangle Park, North Carolina, Northrop Services Inc. (Unpublished
report prepared for the US Environmental Protection Agency).
STAHL, C.J. FATTEH, A.V., & DOMINGUEZ, A.M. (1969) Trichloroethane
poisoning: observations on the pathology and toxicology in six fatal
cases. J. Forensic. Sci., 14: 393-397.
STEWART, R.D. & ANDREWS J.T. (1966) Acute intoxication with methyl
chloroform. J. Am. Med. Assoc., 195: 904-906.
STEWART, R.D. (1968) The toxicology of 1,1,1-trichloroethane. Ann.
occup. Hyg., 11: 71-79.
STEWART, R.D. (1971) Methyl chloroform intoxication. J. Am. Med.
Assoc., 215: 1789-1792.
STEWART, R.D. & DODD, H.D. (1964) Absorption of carbon tetrachloride,
trichloroethylene, tetrachloroethylene, methylene chloride and
1,1,1-trichloroethane through the human skin. Am. Ind. Hyg. Assoc. J.,
25: 439-446.
STEWART, R.D., GAY, H.H., ERLEY, D.S., HAKE, C.L., & SCHAFFER, A.W.
(1961) Human exposure to 1,1,1-trichloroethane vapour: relationship of
expired air and blood concentrations. Am. Ind. Hyg. Assoc. J., 22:
252-262.
STEWART, R.D., GAY, H.H., SCHAFFER, A.W., ERLEY, D.S., & ROWE, V.K.
(1969) Experimental human exposure to methyl chloroform vapor. Arch.
environ. Health, 19: 467-472.
STYLES, J.A. (1981) Activity of 42 coded compounds in the BHK-21 cell
transformation test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 638-646 (Progress in Mutation Research, Vol. 1).
THIELE, D.L., EIGENBRODT, E.H., & WARE, A.J. (1982) Cirrhosis after
repeated trichloroethylene and 1,1,1-trichloroethane exposure.
Gastroenterology, 83: 926-929.
THOMPSON, R.S. & CARMICHAEL, N.G. (1989) 1,1,1-Trichloroethane:
Medium-term toxicity to carp, daphnids, and higher plants. Ecotoxicol.
environ. Saf., 17: 172-182.
TOFTGAARD, R., NILSEN, O.G., & GUSTAFSSON, J.A. (1981) Changes in rat
liver microsomal cytochrome P-450 and enzymatic activities after the
inhalation of n-hexane, xylene, methyl ethyl ketone and
methylchloroform for four weeks. Scand. J. Work Environ. Health, 7:
31-37.
TOPHAM, J.C. (1980) Do induced sperm-head abnormalities in mice
specifically identify mammalian mutagens rather than carcinogens?
Mutat. Res., 74: 379-387.
TORKELSON, T.R., OYEN, F., MCCOLLISTER, D.D., & ROWE V.K. (1958)
Toxicity of 1,1,1-trichloroethane as determined on laboratory animals
and human subjects. Am. Ind. Hyg. Assoc. J., 19: 353-362.
TORSLOV, J. (1988) Hazard assessment of 1,1,1-trichloroethane,
Copenhagen, Denmark, Ministry of the Environment, National Agency of
Environmental Protection, 67 pp (Miljoproject, No. 10).
TRAVIS, C.C., HATTEMER-FREY, H.A., & ARMS, A.D. (1988) Relationship
between dietary intake of organic chemicals and their concentrations
in human adipose tissue and breast milk. Arch. environ. Contam.
Toxicol., 17: 473-478.
TSUCHIMOTO, T. & MATTER, B.E. (1981) Activity of coded compounds in
the micronucleus test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 705-711 (Progress in Mutation Research, Vol. 1). TSURUTA,
H. (1975) Percutaneous absorption of organic solvents. I. Comparative
study of the in vivo percutaneous absorption of chlorinated solvents
in mice. Ind. Health, 13: 227-236.
TU, A.S., MURRAY, T.A., HATCH, K.M., SIVAK, A., & MILMAN, H.A. (1985)
In vitro transformation of balb/c-3T3 cells by chlorinated ethanes
and ethylenes. Cancer Lett., 28: 85-92.
TWEATS, D.J. (1981) Activity of 42 coded compounds in a differential
killing test using Escherichia coli strains WP2, WP67 (uvrA polA),
and CM871 (uvrA lexA recA). In: de Serres, F.J. & Ashby, J., ed.
Evaluation of short-term tests for carcinogens. Report of the
international collaborative programme, New York, Amsterdam, London,
Elsevier/North Holland, pp. 199-209 (Progress in Mutation Research,
Vol. 1).
UHLER, A.D. & DIACHENKO, G.W. (1987) Volatile halocarbon compounds in
process water and processed foods. Bull. environ. Contam. Toxicol.,
39: 601-607.
UNEP (1989) Technical progress on protecting the ozone layer. Report
of the Technology Review Panel, Nairobi, United Nations Environment
Programme, 103 pp.
URANO, K. (1985) Usage and pollution control of chlorinated organic
compounds. Jpn. J. Water Pollut. Res., 8: 269-273.
URANO, K., KAWAMOTO, K., ABE, Y., & OTAKE, M. (1988) Chlorinated
organic compounds in urban air in Japan. Sci. total Environ., 74:
121-131.
US EPA (1977) Environmental monitoring near industrial areas -
Methylchloroform, Washington, DC, US Environmental Protection Agency
(EPA-560/6-77-025).
US EPA (1980a) Proceedings of the Conference on Methyl Chloroform and
Other Halocarbon Pollutants, Washington, DC, US Environmental
Protection Agency (EPA-600/9-80-003).
US EPA (1980b) Ambient water quality criteria for chlorinated ethanes,
Washington, DC, US Environmental Protection Agency (EPA-440/5-80-029).
US EPA (1984) Health assessment document for 1,1,1-trichloroethane
(methyl chloroform), Washington, DC, US Environmental Protection
Agency (EPA-600/8-82-003F).
VAN BEEK, L. (1990) Investigation of a possibility to reduce the
number of rabbits in skin irritation tests; experiments with
dichloromethane, trichloromethylene, tetrachloroethylene and
1,1,1-trichloroethane (Project No. B 86-0927) Apeldoorn, The
Netherlands, Organization for Applied Scientific Research (TNO Report
No. V 89.265).
VEITH, G.D., MACEK, K.J. PETROCELLI, S.R., & CARROLL, J. (1980) An
evaluation of using partition coefficients and water solubility to
estimate bioconcentration factors for organic chemicals in fish. In:
Eaton, J.G., Parrish, P., & Hendricks, A.C., ed. Aquatic toxicology.
Proceedings of the Third Annual Symposium on Aquatic Toxicology,
Philadelphia, American Society for Testing and Materials, pp. 116-129
(ASTM STP No. 707).
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., WHITMORE, R.
SPARACINO, C., & ZELON, H. (1986) Total exposure assessment
methodology (TEAM) study: personal exposures, indoor-outdoor
relationships, and breath levels of volatile organic compounds in New
Jersey. Environ. Int., 12: 369-387.
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., SPARACINO, C.,
WHITMORE, R., SHELDON, L., ZELON, H., & PERRITT, R. (1987) The TEAM
study: Personal exposure to substances in air, drinking water, and
breath of 400 residents of New Jersey, North Carolina and North
Dakota. Environ. Res., 43: 290-307.
WALRAEVENS, R., TROUILLET, P., & DEVOS, A. (1974) Basic elimination of
HCI from chlorinated ethanes. Int. J. chem. Kinet., VI: 777-786.
WEAST, R.C. (1986) CRC handbook of chemistry and physics, 67th ed,
Boca Raton, Florida, CRC Press.
WESTERBERG, E. & LARSSON, L. (1982) Use of automated head-space gas
chromatography for determination of 1,1,1-trichloroethane in rat blood
and brain tissue. Int. J. environ. anal. Chem., 12: 233-239.
WILLIAMS, G.M. (1983) DNA repair tests of 11 chlorinated hydrocarbon
analogs, Washington, DC, US Environmental Protection Agency (Draft
Final Report TR-507-18).
WILSON, B.H. & WHITE, M.V. (1986) A fixed-film bioreactor to treat
trichloroethylene-laden waters from interdiction wells. In:
Proceedings of the 1986 National Symposium and Exposition on Aquifer
Restoration and Ground Water Monitoring, Columbus, Ohio, 19-22 May,
Dublin, Ohio, National Well Water Association, pp. 425-436.
WILSON, J.T., MCNABB, J.F., WILSON, B.H., & NOONAN, M.J. (1983)
Biotransformation of selected organic pollutants in ground water. Dev.
ind. Microbiol., 24: 125-233.
WHO (in preparation) Health and Safety Guide for
1,1,1-trichloroethane, Geneva, World Health Organization.
WINDHOLZ, M. ed. (1983) The Merck index: An encyclopedia of chemicals,
drugs and biologicals, 10th ed., Rahway, New Jersey, Merck and Co.,
Inc.
WMO (1982) Global ozone research and monitoring project: Report No.
11. The stratosphere 1981: Theory and measurements, Geneva,
Switzerland, World Meteorological Organization.
WOOLVERTON, W.L. & BALSTER, R.L. (1981) Behavioral and lethal effects
of combinations of oral ethanol and inhaled 1,1,1-trichloroethane in
mice. Toxicol. appl. Pharmacol., 59: 1-7.
WRIGHT, M.F. & STROBL, D.J. (1984) 1,1,1-Trichloroethane cardiac
toxicity: Report of a case. J. Am. Osteopath. Assoc., 84: 285-288.
WRIGHTSON, I. & SANTON, R.C. (1988) An explosion involving
1,1,1-trichloroethane. Loss Prev. Bull., 083: 21-23.
YORK, R.G., SOWRY, B.M., HASTINGS, L., & MANSON, J.M. (1982)
Evaluation of teratogenicity and neurotoxicity with maternal
inhalation exposure to methylchloroform. J. Toxicol. environ. Health,
9: 251-266.
YUNG, Y.L., MCELROY, M.B., & WOFSY, S.C. (1975) Atmospheric
halocarbons: A discussion with emphasis on chloroform. Geophys. Res.
Lett., 2: 397-399.
RESUME
Le 1,1,1-trichloréthane est un hydrocarbure chloré produit par
chloration du chlorure de vinyle ou du chlorure de vinylidène. En
1988, la production mondiale était d'environ 680 000 tonnes. C'est un
liquide incolore et volatil à l'odeur caractéristique dont la vapeur
est plus dense que l'air. On l'utilise essentiellement pour le
dégraissage des surfaces métalliques et comme solvant dans un grand
nombre de produits industriels et de produits de consom-mation,
notamment des adhésifs, des détachants et des bombes aérosols. Il
sert également d'intermédiaire en synthèse chimique. Le
trichloréthane technique contient généralement 3 à 8% de stabilisants
destinés à empêcher sa décomposition et la formation d'acide
chlorhydrique; les parties métalliques sont ainsi protégées de la
corrosion. Il est ininflammable dans les conditions normales mais la
vapeur s'enflamme à haute température et, au cours du soudage, elle
peut se décomposer en libérant du phosgène, un gaz toxique. Des
réactions extrêmement violentes peuvent se produire en cas de contact
avec de l'aluminium, du magnésium ou leurs alliages.
Le 1,1,1-trichloréthane pénètre facilement dans l'environne-ment.
Comme il séjourne longtemps dans la troposphère (environ six ans) et
qu'il n'est que lentement biodégradable, on le retrouve maintenant
partout dans l'environnement, même à grande distance des zones
industrielles. A proximité d'installations industrielles où ce
composé est produit ou manipulé, on a trouvé du 1,1,1-trichloréthane
à des concentrations atteignant 86 µg/m3 (16 000 ppm, p/p) dans des
échantillons d'air.
Le trichloréthane se déplace dans le sol et parvient jusqu'aux
eaux souterraines. On en a trouvé à des concentrations atteignant
1600 µg/litre dans les eaux souterraines ou les eaux de surface. Ce
phénomène peut être à l'origine de la contamination des réserves d'eau
potable.
On estime que 15% des émissions annuelles de
1,1,1-trichloré-thane sont entraînées dans la stratosphère où elles
peuvent endommager la couche d'ozone par libération d'atomes de
chlore.
Des essais biologiques sur des crustacés et des poissons à des
concentrations supérieures à 7 mg/litre ont révélé des effets toxiques
aigus. On possède quelques données selon lesquelles le composé ne
s'accumulerait que faiblement chez les organismes aquatiques. En
raison du caractère limité des données disponibles, il est difficile
d'évaluer les effets qu'il peut avoir sur les organismes terrestres.
L'exposition humaine au 1,1,1-trichloréthane est due
principalement à l'inhalation de ce composé qui est ensuite rapidement
résorbé par l'organisme. Il peut y avoir également exposition par
contact cutané ou ingestion. Le trichloréthane se répartit largement
dans les tissus de l'organisme et il traverse la barrière
hématoencéphalique et placentaire. On le retrouve également dans le
lait humain mais il ne semble pas qu'il s'accumule dans les tissus.
Sa principale voie d'élimination est l'exhalation du composé non
métabolisé.
La toxicité aiguë et chronique du 1,1,1-trichloréthane est
relativement faible mais en cas de forte exposition, le risque
d'intoxication est réel. Cela peut se produire en cas d'exposition
professionnelle, d'accidents ou encore de toxicomanie aux solvants.
Comme il s'agit d'un solvant volatil dont la vapeur est beaucoup plus
dense que l'air, il peut se trouver à des concentrations
inhabituellement fortes et dangereuses dans des espaces confinés comme
les réservoirs "vides". Ce genre de situation a donné lieu à
plusieurs intoxications mortelles ou qui auraient pu l'être sur les
lieux de travail ou ailleurs.
Chez l'homme, l'effet critique s'exerce au niveau du système
nerveux central. On observe des effets qui vont de légers troubles du
comportement (accompagnés d'une légère irritation occulaire) à la dose
de 1,9 g/m3 (350 ppm), jusqu'à l'inconscience et à l'arrêt
respiratoire lorsque la concentration est élevée. Dans certains cas,
des anomalies cardiaques mortelles peuvent se produire. Le
trichloréthane est moins toxique pour le foie que la plupart des
autres solvants organiques chlorés. La dose sans effet observable
pour l'homme se situe autour de 1,35 g/m3 (250 ppm).
Aucune étude satisfaisante n'a été publiée sur les effets
cancérogènes de ce solvant chez l'homme. Toutefois une étude par
inhalation de longue durée sur des rats et des souris exposés à 8,1
g/m3 (1500 ppm) n'a pas révélé le moindre effet cancérogène. Le
1,1,1-trichloréthane ne présente pas d'activité génotoxique
significative.
Chez des rats et des lapins exposés au trichloréthane à des
concentrations qui étaient toxiques pour les femelles gestantes, on
n'a pas noté d'effets tératogènes, malgré la présence d'effets
toxiques sur le développement. Les données épidémiologiques relatives
à ces effets sur la reproduction sont trop limitées pour permettre
d'en tirer une conclusion.
RESUMEN
El 1,1,1-tricloroetano es un hidrocarburo clorado que se fabrica
a partir del cloruro de vinilo o del cloruro de vinilideno por
cloración. En 1988 la producción mundial fue de unas 680 000
toneladas. Es un líquido volátil e incoloro con un olor
característico, y su vapor es más denso que el aire. Se utiliza
principalmente en el desengrasado de metales y como disolvente en
muchos productos industriales y de consumo, como adhesivos,
quitamanchas y envases de aerosoles. Es también un producto químico
intermediario. El tricloroetano de calidad técnica suele contener del
3% al 8% de estabilizadores que impiden la degradación y la formación
de ácido clorhídrico a fin de proteger las partes metálicas de la
corrosión. En condiciones normales no es inflamable, pero su vapor
arde a altas temperaturas y, durante las operaciones de soldadura, su
degradación produce fosgeno, un gas tóxico. Cuando entra en contacto
con el aluminio, el magnesio y sus aleaciones puede provocar
reacciones muy violentas.
El 1,1,1-tricloroetano llega fácilmente al medio ambiente. Su
prolongada permanencia en la troposfera (unos seis años) y baja
biodegradabilidad son razón de su actual omnipresencia en el medio
ambiente, incluso lejos de las zonas industriales. En muestras de
aire de zonas próximas a industrias que lo fabrican o lo manipulan se
han detectado concentraciones de hasta 86 µg/m3 (16 ppb, peso/peso).
El tricloroetano se desplaza en los suelos y alcanza las aguas
subterráneas. Se han encontrado concentraciones de hasta 1600
µg/litro en aguas subterráneas o superficiales. Esta puede ser una
fuente de contaminación para suministros de agua potable.
Se estima que el 15% del volumen anual liberado llega a la
estratosfera, donde libera átomos de cloro que contribuyen a la
destrucción del ozono.
En bioensayos con crustáceos y peces se han observado efectos
tóxicos agudos con concentraciones superiores a 7 mg/litro. Limitada
información limitada disponible parece indicar que la bioacumulación
en los organismos acuáticos es baja. La escasez de datos dificulta la
evaluación de los posibles efectos en los organismos terrestres.
El ser humano está expuesto al 1,1,1-tricloroetano principalmente
por inhalación, y luego el organismo absorbe rápidamente la sustancia.
También se puede dar la exposición por contacto cutáneo o ingestión.
El tricloroetano se distribuye ampliamente por los tejidos y atraviesa
las barreras hematoencefálica y placentaria. Aunque también se ha
encontrado en la leche humana, no parece que se produzca
bioacumulación. La principal vía de eliminación es la exhalación del
compuesto inalterado.
Las toxicidades aguda y crónica del 1,1,1-tricloroetano son
relativamente bajas, pero en condiciones de exposición intensa pueden
producirse efectos tóxicos. Estas condiciones se pueden presentar en
casos de exposición profesional, de abuso del disolvente o de
accidentes. Dado que el disolvente es volátil y el vapor es mucho más
denso que el aire, se pueden producir concentraciones inesperadamente
elevadas y peligrosas en espacios reducidos, como depósitos de
almacenamiento "vacíos". Así, se han producido algunas intoxicaciones
mortales y casi mortales en lugares de trabajo y otros.
El efecto más importante en el ser humano se produce en el
sistema nervioso central. Los efectos observables van desde ligeros
cambios de comportamiento (acompañados de una leve irritación ocular)
con 1,9 g/m3 (350 ppm) hasta la pérdida del conocimiento y el paro
respiratorio con concentraciones más elevadas. Sin embargo, también
se pueden producir anomalías cardíacas fatales. La toxicidad hepática
del tricloroetano es inferior a la de la mayoría de los disolventes
organoclorados. El nivel sin efecto observado (NOEL) para la especie
humana parece ser del orden de 1,35 g/m3 (250 ppm).
No se han publicado estudios adecuados sobre los efectos
carcinogénicos en la especie humana. Sin embargo, en los estudios de
inhalación prolongada en ratas y ratones expuestos a 8,1 g/m3 (1500
ppm) no se obtuvieron pruebas de ningún efecto carcinogénico. El
1,1,1-tricloroetano no tiene un potencial genotóxico importante.
En ratas y conejos se ha observado toxicidad en la fase de
crecimiento, pero no teratogenicidad, con concentraciones que fueron
tóxicas para las madres. Las escasas pruebas epidemiológicas en
relación con sus efectos sobre la reproducción no son concluyentes.
 body weight. Both trichloroethanol and trichloroacetic acid were
identified as metabolites in the urine, trichloroethanol being
excreted (as the glucuronide) much faster than trichloroacetic acid
(Eben & Kimberle 1974).
Following a single inhalation exposure of rats to 810 and
8100 mg/m3 (150 and 1500 ppm), only a 2-4 times increase in excreted
metabolites was found between the two doses, suggesting metabolic
saturation. Repeated exposure to 8100 mg/m3 over 16 months did not
change the amount of 1,1,1-trichloroethane metabolites. Similar
results were obtained in mice. In both rats and mice, urine
metabolites accounted for 40-70% of the total amount metabolized.
Overall, mice were found to biotransform approximately 5 times more
1,1,1-trichloroethane (per kg body weight) than rats. In rats and
mice an age-related increase in the amount metabolized was observed in
aged animals as opposed to young adults (Schumann et al., 1982).
6.3.2 Human studies
The average amount of metabolites excreted in the urine of humans
(workers or volunteers) exposed to 1,1,1-trichloroethane in the air
(22-1890 mg/m3, 4-350 ppm) was variable, but was typically between
3 and 7% of the absorbed dose. The ratio of the metabolites
trichloroethanol glucuronide and trichloroacetic acid was about 2 to
1, but this increased with increasing exposure concentration (Seki et
al., 1975; Humbert & Fernandez, 1977; Monster et al., 1979; Nolan et
al., 1984).
6.3.3 Metabolic interactions
Simultaneous exposure to other solvents tends to increase the
retention and decrease the metabolism of 1,1,1-trichloroethane
(Savolainen et al., 1981). 1,1,1-Trichloroethane metabolism was
accelerated in rats pre-treated with ethanol because of the induction
of metabolic enzymes (Sato et al., 1980).
6.4 Elimination
Regardless of the route of administration, the main excretory
route for 1,1,1-trichloroethane is exhalation via the lungs. This may
be explained by the relatively low solubility in blood.
6.4.1 Animal studies
In rats and mice, 55% to 98% of the 1,1,1-trichloroethane was
excreted unchanged in expired air after oral or intraperitoneal
exposure (Hake et al., 1960; Reitz et al., 1988).
Following inhalation exposure, 94-98% of the absorbed
1,1,1-trichloroethane was excreted unchanged in the expired air of
rats during 72 h. In mice, the corresponding figure was 87-97%. The
rate of elimination was somewhat higher in mice; 85% was eliminated
during the first 3 h compared to 65% in rats (Schumann et al., 1982).
A total of about 1-8% of an absorbed dose in rodents is excreted
in the urine (Hake et al., 1960; Schumann et al., 1982).
After exposure to 1,1,1-trichloroethane vapour, the level in
blood plasma decreases rapidly in a diphasic or triphasic manner,
depending on the exposure level. Following exposure to 810 mg/m3
(150 ppm), the half-lives were 10 and 139 min, whereas with 8100
mg/m3 (1500 ppm), the half-lives were 36 and 238 min. In mice the
half-lives were a little shorter (Schumann et al., 1982).
6.4.2 Human studies
Studies with human volunteers show that over 90% of the absorbed
trichloroethane is excreted unchanged in the expired air. Only minor
parts (5-7%) of the absorbed solvent are excreted in the urine (as
trichloroethanol glucuronide and trichloroacetic acid) (Stewart et
al., 1969; Humbert & Fernadez, 1977; Nolan et al., 1984). The main
product was found to be 2,2,2-trichloroethanol glucuronide, the
excretion of which was completed within 8 days. The secondary product
was trichloroacetic acid; its excretion occurred somewhat later and
was completed within 12 days (Humbert & Fernandez, 1977).
The elimination of 1,1,1-trichloroethane, measured as plasma
concentration and concentration in expired air, in six human
volunteers exposed to 191 or 1911 mg/m3 (35 or 350 ppm) for 6 h
could be described by a three-compartment model with estimated
half-lives of 44 min, 5.7 h, and 53 h, respectively. Less than 1%
remained in the body after 9 days (Nolan et al., 1984).
In a study by Nolan et al. (1984), the trichloroethane
concentrations in blood and expired air were proportional to the
exposure concentration after 6 h of exposure and indicated that about
25% of the 1,1,1-trichloroethane inhaled during the exposure was
absorbed.
6.5 Biological monitoring
Several options exist for the biological monitoring of exposure
to 1,1,1-trichloroethane (Monster 1986). These include the
determination of:
Ê the unchanged solvent in blood or alveolar air;
Ê the metabolite trichloroethanol in blood, alveolar air or urine;
Ê the metabolite trichloroacetic acid in blood and urine.
Table 9 indicates the mean concentrations of these parameters as
a result of exposure of subjects for 8 h/day (5 days/week) to a
time-weighted average (TWA) concentration of 270 mg/m3 (50 ppm)
(Monster, 1986).
Table 9. Mean concentrations of 1,1,1-trichloroethane,
trichloroethanol, and trichloroacetic acid in blood,
alveolar air, and urinea
Time after exposure of sampling
Test End of 5-15 min 16 h 64 h
exposure
Blood (mg/litre)
1,1,1-trichloroethane 0.9 0.07
trichloroethanol 0.16 -
trichloroacetic acid 2.3 1.6
Alveolar air (mg/m3)
1,1,1-trichloroethaneb 210 (39) 13 (2.4) 8 (1.5)
trichloroethanol 0.014 0.007 -
Urine (mg/g creatinine)
trichloroacetic acid 4.9 2.5 0.9
trichloroethanol 2.5 1.8 1.5
a From: Monster (1986)
b Values in parentheses are in ppm
Trichloroethane concentrations in blood measured after work on a
Friday seem to be the best single parameter for estimation of the
TWA-week exposure (Monster, 1986). For one-day exposure, the
trichloroethane level in blood and urine is most useful (Monster,
1986).
Droz et al. (1989) suggested a physiological model for describing
variability in the biological monitoring of solvent exposure.
Standard statistical distributions are used to simulate variability in
exposure concentration, physical workload, body fluid, liver function,
and renal clearance. For groups of workers exposed daily, the model
calculates air monitoring indicators and biological monitoring
results, including levels in expired air, blood, and urine. The
calculated results obtained are discussed and compared with measured
data for physiological and toxicokinetic parameters for six solvents
including 1,1,1-trichloroethane and their metabolites. It is
suggested that such mathematical models are applicable for prediction
and management studies.
6.6 Bioaccumulation
Inhalation exposure of rats to 2700 mg/m3 (500 ppm) for 4 days,
6 h per day, led to an accumulation of 1,1,1-trichloroethane in the
fat 17 h after the last exposure. Further exposure on the 5th day
increased brain, liver, lung, and blood levels (Savolainen et al.,
1981).
A study by Travis et al. (1988) indicated that bioaccumulation of
organic chemicals, including 1,1,1-trichloroethane, perchloroethane,
trichloroethylene, and dichloromethane, in human adipose tissues was
positively correlated with their octanol-water partition coefficients.
The information presented, however, was based on data from a human
pharmacokinetic model.
7. EFFECTS ON LABORATORY MAMMALS AND IN VITRO TEST SYSTEMS
Appraisal
1,1,1-Trichloroethane administered by various routes has low
acute toxicity for laboratory animals. The acute toxicity pattern is
characterised by central nervous system depression and respiratory
arrest at lethal levels. 1,1,1-Trichloroethane is a moderate skin
and mild eye irritant.
Short-term inhalation exposure of rats to 4320-5400 mg/m3
(800-1000 ppm) or more produced an increase in liver weight. At
similar concentrations in mice, the liver changes were more marked
and included fatty infiltration and single cell necrosis. Minor
cytoplasmic alterations were seen in mice exposed to 1350 mg/m3
(250 ppm). Studies on Mongolian gerbils using 1,1,1-trichloroethane
concentrations down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several regions of
the brain. The significance of these findings is uncertain. The
no-observed-effect level (NOEL) for rats is about 2700 mg/m3 (500
ppm).
In long-term toxicity studies, 1,1,1-trichloroethane
administration led to reduced weight gain in both rats and mice.
Slight microscopic hepatic effects were seen, but only at the highest
concentration of 8100 mg/m3 (1500 ppm). There was no evidence of
carcinogenic potential in oral and inhalation studies on rats and
mice. One study reported increased incidence of leukaemia but this
study is considered inadequate.
1,1,1-Trichloroethane has been shown to have low genotoxic
potential in a range of in vitro and in vivo studies.
In a multigeneration study on mice, there was no evidence of
adverse reproductive effects (including effects on fertility) caused
by 1,1,1-trichloroethane in drinking-water at concentrations up to
1000 mg/kg body weight. Inhalation studies on female rats and mice
at 4590 mg/m3 (850 ppm) showed no evidence of teratogenic or
fetotoxic effects. However, in rats exposed to 11 340 mg/m3 (2100
ppm), there was some evidence of fetotoxicity.
The data available are inadequate to assess the immunotoxic
potential of 1,1,1-trichloroethane.
Enhanced toxicity of 1,1,1-trichloroethane was observed when
exposure was combined with exposure to ethanol.
7.1 Acute toxicity
1,1,1-Trichloroethane has a very low acute toxicity in laboratory
animals dosed by various routes of administration. Selected LD50
and LC50 values are given in Tables 10 and 11.
The toxicological pattern of acute trichloroethane poisoning is
central nervous system (CNS) depression, eventually culminating in
respiratory arrest or cardiac failure at lethal exposure levels
(Stewart, 1968).
The findings of rapid and shallow breathing in a dog following
inhalation of 0.9% (by volume) 1,1,1-trichloroethane (corresponding to
48 600 mg/m3 or 9000 ppm) may be related to an increase in the
activity of the lung stretch receptor in the vagus nerve (Kobayashi et
al., 1986). A level of 1.3% increased the heart rate but 2.8%
decreased it (Kobayashi et al., 1987).
Non-lethal acute toxicity has also been investigated (see Table
12). In a study on rats, 50% of the experimental group showed loss of
coordination (ataxia) after inhalation of 20 400 mg/m3 (3780 ppm)
for 4 h, and loss of righting-reflex was observed at 45 800 mg/m3
(8480 ppm). Tremors and death were observed at 64 800 mg/m3 (12 000
ppm) within one hour of exposure (Mullin & Krivanek, 1982).
Cardiac sensitization to adrenaline was reported in dogs exposed
to 37 800 mg/m3 (range: 21 600 to 59 400 mg/m3) for 5 min (Clark
& Tinston, 1982). An increase in concentration (range: 37 300 to 373
000 mg/m3) and inhalation period (0.5 to 20 min) was accompanied by
a decrease in the amount of adrenaline that induced arrhythmia
(Kobayashi et al., 1982).
Reinhardt et al. (1973) reported that 1,1,1-trichloroethane
caused cardiac sensitization to adrenaline in dogs at and above the
0.5% (v/v) level. The marked response was associated with ventricular
fibrillation following the challenge dose of adrenaline. At this
level, the solvent was also associated with excitement and struggling
in the animals. Histopathological examination of samples from the
dogs that developed fatal arrhythmias did not show any gross or
microscopic abnormalities.
Table 10. Acute toxicity (LD50) of 1,1,1-trichloroethane in experimental animals
Species Sex Route LD50 (mg/kg Reference
body weight)
Rat male oral 14 300 Torkelson et al. (1958)
Rat female oral 11 000 Torkelson et al. (1958)
Mouse female oral 9700 Torkelson et al. (1958)
Guinea-pig male & female oral 8600 Torkelson et al. (1958)
Rabbit male & female oral 10 500 Torkelson et al. (1958)
Rabbit male & female skin 15 800 Torkelson et al. (1958)
Mouse female intraperitoneal 3700 Gradiski et al. (1974)
Mouse male intraperitoneal 5080 Klaassen & Plaa (1966)
Dog male intraperitoneal 4140 Klaassen & Plaa (1967)
Table 11. Acute toxicity by inhalation (LC50) of 1,1,1-trichloroethane
in experimental animals
Species Sex LC50 Exposure Reference
(g/m3) (ppm) duration
Rat male & female 97.2 18 000 3 h Adams et al. (1950)
Rat male & female 76.9 14 250 7 h Adams et al. (1950)
Rat male 99.4 18 400 4 h Siegel et al. (1971)
Rat male 55.6 10 300 6 h Bonnet et al. (1981)
Rat male & female 205 38 000 15 min Clark & Tinston (1982)
Mouse male 120 22 240 30 min Woolverton & Balster (1981)
Mouse female 72.4 13 410 6 h Gradiski et al. (1978)
Mouse male 21.1 3910 2 h Horiguchi & Horiguchi (1971)
Mouse male 99.1 18 358 1 h Moser & Balster (1985)
Mouse male 111 20 616 30 min Moser & Balster (1985)
Mouse male 159 29 492 10 min Moser & Balster (1985)
Table 12. Non-lethal acute toxicity in experimental animals
exposed to 1,1,1-trichloroethane by inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 45 800 4 h loss of righting Mullin & Krivanek (1982)
reflex (EC50)
Rat 20 400 4 h loss of coordination Mullin & Krivanek (1982)
(EC50)
Mouse 31 000 1 h inverted screen test Moser & Balster (1985)
performance (EC50)
Dog 37 800 5 min cardiac sensitization Clark & Tinston (1982)
to adrenaline
Dog 21 600 a few min decrease in blood Kobayashi et al. (1983)
pressure
The effect of 1,1,1-trichloroethane on liver enzyme activity in
the serum of experimental animals has been investigated. An almost
lethal intraperitoneal dose of 1,1,1-trichloroethane (0.87 ml/kg body
weight) caused a significant elevation of ALAT (alanine
aminotransferase) in dogs (Klaassen & Plaa, 1967), and one eighth of
a lethal intraperitoneal dose caused a significant elevation in SDH
(sorbitol dehydrogenase) (Lundberg et al., 1986). In both studies,
the hepatotoxicity of 1,1,1-trichloroethane was relatively small
compared to that of other organic solvents, e.g., chloroform, carbon
tetrachloride, dimethylformamide (Lundberg et al., 1986),
dichloromethane, 1,1,2-trichloroethane, and trichloroethylene
(Klaassen & Plaa, 1967).
7.1.1 Irritation
Trichloroethane has been shown to produce "mild" skin irritation
in rabbits when repeatedly applied topically under an occlusive
dressing. Redness was noted and the skin became scaly, but the effect
was transient, the skin rapidly returned to normal (Torkelson et al.,
1958).
After guinea-pigs had been exposed to 1 ml of
1,1,1-trichloroethane via a skin depot (a glass ring attached to the
skin and covered with glass), for 15 min, oedema was observed.
Prolonged exposure for several hours led to more severe inflammatory
reactions in the upper part of the dermis, and histological
examination at 15 min and 1, 4, and 16 h showed a number of changes to
the epidermis. The extent of these changes increased with duration of
exposure (Kronevi et al., 1981).
Repeated topical application of trichloroethane to abraded and
non-abraded rabbit skin for up to 90 days resulted in slight,
reversible irritation (Torkelson et al., 1958). The same study showed
that when trichloroethane was applied soaked in a cotton wool pad and
bandaged to the shaven belly of a rabbit slight reddening and
scaliness occurred, but this only increased slightly with repeated
applications.
The application of 0.5 ml trichloroethane to the shaven skin of
rabbits under an occlusive dressing for 24 h, resulted in moderate
skin irritation (Duprat et al., 1976). In a recent study of OECD
methods (4-h exposure under semi-occlusive dressing) trichloro-ethane
was reported to be a skin irritant (van Beek, 1990).
Trichloroethane is a "mild" eye irritant. Slight to moderate
pain and slight conjunctival irritation, but no corneal damage, was
reported following a single application of 100 µl trichloroethane to
the eyes of rabbits (Torkelson et al., 1958). Duprat et al. (1976)
also found that instillation of 0.1 ml trichloroethane to the eyes of
rabbits produced slight irritation.
7.1.2 Short-term exposure
7.1.2.1 Inhalation
When groups of rats, guinea-pigs, rabbits, dogs, and squirrel
monkeys were exposed to 1,1,1-trichloroethane (11 880 mg/m3, 2200
ppm) for 8 h/day, 5 days/week for 6 weeks, the only sign of toxicity
was reduced body weight gain in rabbits and dogs. There were no
effects on haematological parameters or serum urea nitrogen and no
histopathological changes were observed (Prendergast et al., 1967).
In a study by Adams et al. (1950), rats were exposed to 0 or 27
000 mg/m3 (0 or 5000 ppm) for 7 h/day on 32 out of 45 days and
guinea-pigs received 20 to 65 7-h exposures to 0, 3510, 8100, 16 200
or 27 000 mg/m3 (0, 650, 1500, 3000 or 5000 ppm) for a period of 1-3
months. Body weight gain was reduced in both species at all exposure
levels used, but there were no other signs of toxicity and no effects
were observed on blood urea nitrogen. The only 1,1,1-trichloroethane-
related effect observed on histopathological examination was fatty
degeneration, without necrosis, in the liver of guinea-pigs at 16 200
and 27 000 mg/m3.
Rats exposed to 2700 mg/m3 (500 ppm), 6 h/day for 5 days,
showed no behavioural effects. However, there was a slight decrease
in brain RNA content relative to controls (Savolainen et al., 1977).
Inhalation of 1750 mg/m3 (320 ppm) for 30 days had no effects on the
composition of brain lipids in rats (Kyrklund et al., 1988).
When rats were given 4320 mg/m3 (800 ppm) by inhalation 6
h/day, 5 days/week for 4 weeks, absolute and relative liver weights
were increased but there was no induction of liver microsomal
cytochrome P-450 (Toftgaard et al., 1981). A 1-h daily exposure to 54
000 mg/m3 (10 000 ppm) for 3 months resulted in a narcotic effect
(sedation and transient sleep) and in an increase of relative liver
weight in rats, but there was no evidence of organ damage (Torkelson
et al., 1958).
When rats were exposed continuously for 100 days to 1350 or 5400
mg/m3 (250 or 1000 ppm), no effects were observed in the low-dose
group but an increase in relative liver weight was seen in the
high-dose group (McEwen & Vernot, 1974). No toxicity was observed in
similar experiments with dogs and monkeys (McEwen & Vernot 1974).
Exposure of mice to 5400 mg/m3 (1000 ppm) continuously for 14
weeks resulted in marked liver changes (elevated relative liver
weight, moderate liver triglyceride accumulation, and necrosis of
individual hepatocytes). Electron microscopy showed extensive
cytoplasmic modifications consisting of vesiculation of the rough
endoplasmic reticulum with loss of attached polyribosomes, increased
smooth endoplasmic reticulum, microbodies (peroxisomes), and
triglyceride droplets. The observed toxic effects were similar to,
but much less severe than, those produced by carbon tetrachloride.
Only minor cytoplasmic alterations were seen in mice exposed to 1350
mg/m3 (250 ppm) (McNutt et al., 1975).
No signs of toxicity were observed in rats, rabbits, guinea-pigs,
dogs or monkeys exposed to 2730 mg/m3 (500 ppm) 7 h/day, 5 days/week
for 6 months (Torkelson et al., 1958).
In a study by Prendergast et al. (1967), rats, rabbits,
guinea-pigs, dogs, and monkeys were exposed continuously to 754 or
2059 mg/m3 for 90 days. At the higher dose level, there were no
deaths in any species after 90 days, and the authors stated that no
visible toxic signs were observed. However, at the lower dose level,
some deaths occurred (2 out of 15 rats and 1 out of 3 rabbits).
Varying degrees of lung congestion were noted in the surviving
animals. In view of this and the deaths at the lowest dose tested,
the authors stated that no positive conclusion could be drawn as to
whether the effects were associated with the exposure.
No adverse effects were seen in male Wistar rats exposed to 1100
mg/m3 (204 ppm) 8 h/day, 5 days/week for 14 weeks (Eben & Kimmerle,
1974).
When young adult Mongolian gerbils (Meriones ungiculatus) were
continuously exposed to 378, 1134 or 5400 mg/m3 (70, 210 or 1000
ppm) for 3 months, followed by a 4-month period without exposure,
increased glial fibrillary acidic protein (GFA) was found in the
cerebral cortex at the two highest exposure levels, indicating
astrogliosis in this region of the brain (Rosengren et al., 1985). In
a similar study by Karlsson et al. (1987), the DNA concentrations in
several brain regions were decreased in animals exposed to 378 mg/m3
(70 ppm), the only exposure concentration used.
Table 13 summarizes data from short-term inhalation studies.
7.1.2.2 Oral administration
When rats were dosed orally with 1,1,1-trichloroethane in corn
oil, 5 days/week for 6 weeks, a dosage of 3.2 g/kg body weight per day
had no adverse effects. However, total doses of 5.6 g/kg body weight
in females and 10 g/kg body weight in males induced 40% mortality and
decreased the body weight of survivors (NCI, 1977). In mice no
mortality was seen with a dose of 5.6 g/kg body weight. Thus,
trichloroethane has low toxicity in rats and mice following repeated
exposure by the oral route.
Table 13. Short-term toxicity (exposure-effect relationships) in
experimental animals exposed to 1,1,1-trichloroethane by
inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 5400 100 days increased relative McEwen & Vernot
liver weight (1974)
Rat 1100 14 weeks, NOAEL Eben & Kimberle
8 h/day, (1974)
5 days/week
Gerbil 5400 3 months reduced brain weight Rosengren et al.
(1985)
Gerbil 380 3 months biochemical changes Karlsson et al.
in the brain (1987)
Mouse 5400 14 weeks liver necrosis McNutt et al.
(1975)
Mouse 1350 14 weeks minor liver effects McNutt et al.
(1975)
Rat, dog, 2700 6 months, NOAEL Torkelson et al.
rabbit, 7 h/day, (1958)
monkey, 5 days/week
guinea-pig
Rat, dog, 2000 90 days NOAEL Prendergast et al.
rabbit, (1967)
monkey,
guinea-pig
7.2 Long-term exposure
In the NCI (1977) carcinogenesis study (see section 7.5.1 for
details of doses), reduced weight gain was noted in both rats and mice
during the exposure period at all dose levels. Furthermore, bloody
discharge and crusting around the eyes of some rats was observed
during the second year (NCI, 1977).
In an oncogenicity inhalation study, female rats exposed to the
highest concentration (8100 mg/m3, 1500 ppm) of 1,1,1-
trichloroethane exhibited a significant decrease in body weight, and
both females and males showed very slight microscopic hepatic effects
at 6, 12, and 18 months. At lower concentrations and in male rats and
mice of both sex, no toxicity was observable (Quast et al., 1988).
7.3 Reproductive toxicity, embryotoxicity, and teratogenicity
Inhalation studies in female rats and mice gave no evidence of
any teratogenic or fetotoxic effects followed exposure to
1,1,1-trichloroethane at a concentration of 4700 mg/m3 (875 ppm) for
7 h/day on days 6-15 of gestation (Leong et al., 1975). No signs of
maternal toxicity were noted.
In a study of female rats exposed to a higher
1,1,1-trichloroethane concentration (11 340 mg/m3, 2100 ppm, 6
h/day, 5 days/week) for 2 weeks before mating and/or during 20 days of
gestation (6 h/day, 7 days/week), there was some evidence of
fetotoxicity. This consisted of reduced fetal weight in the group
exposed during pregnancy only and minor visceral and skeletal
abnormalities, such as delayed ossification, in the group exposed
before and during pregnancy (York et al., 1982). No signs of maternal
toxicity were observed.
In a two-generation fertility study incorporating teratogenicity
and dominant lethal elements, mice received 0, 100, 300 or 1000 mg
1,1,1-trichloroethane/kg daily in the drinking-water (Lane et al.,
1982). Two or three litters were produced from each of the two
generations, and the parental animals and offspring were used
variously to investigate fertility, developmental toxicity, and
dominant lethal effects. No adverse effects on any aspect of
reproduction were evident in this study. The level of
1,1,1-trichloroethane in the drinking-water in this study was reported
to be up to 6000 mg/litre but was not actually measured. In a further
study, rats exhibited slight aversion to 30 mg/litre in drinking-water
(George et al. 1989).
Rats (Sprague-Dawley) and rabbits (New Zealand White) were
exposed on days 6-15 and 6-18 of gestation, respectively, to 0, 5400,
16 200 or 32 400 mg/m3 (0, 1000, 3000 or 6000 ppm) for 6 h/day.
1,1,1-Trichloroethane was found to be developmentally toxic to both
rats and rabbits at the highest dose level, producing an increase in
unossified and poorly ossified cervical centra, decreased female fetal
body weight, an increase in non-viable implantations in rats, and an
increase in the frequency of bilateral extra 13th rib in rabbits.
Both rats and rabbits showed a NOEL of 16 200 mg/m3 for
developmental toxicity. For both species, the developmental toxicity
was seen in the presence of maternal tox-icity (Personal communication
by E.Z. Francis to S. Ells, US EPA, Test Rules Development Branch,
Existing Chemical Assessment Division. Review of Developmental
Toxicity Studies on 1,1,1-Trichloroethane (TCE). OHEA, ORD. Sept. 28,
1988).
Dapson et al. (1984) reported that exposure to 10 mg
1,1,1-trichloroethane per litre drinking-water caused cardiac
anomalies in developing Sprague-Dawley rats. The repeatability of
this preliminary study was investigated by George et al. (1989), who
exposed male and female CD rats to 3, 10 or 30 mg of 97% pure
1,1,1-trichloroethane per litre drinking-water using 0.05% Tween 80 as
an emulsifying agent. The animals were exposed for a period of 14
days before and at least 13 days after co-habitation. Sperm-positive
females continued to be exposed during pregnancy and lactation until
postnatal day 21. No significant effects were noted concerning the
reproductive competence of the parental animals or the postnatal
growth and development of the offspring (fertility, length of
gestation period, litter size, pup body weight, and pup survival were
all investigated). In addition, there was no increase in the
incidence of any cardiac malformations or other anomalies. Thus the
finding of Dapson et al. (1984) was not confirmed using a different
strain of rats and a 3-fold higher concentration of
1,1,1-trichloroethane.
7.4 Mutagenicity
Appraisal
1,1,1-Trichloroethane has been tested extensively for
mutagenicity, both in vitro and in vivo, and the principal studies
are summarized in Table 14. The results of most of the studies were
clearly negative. The positive results seen in some in vitro tests
give rise to some concern, but may well have been due to the presence
of stabilizers and/or impurities. Some of the studies yielding
negative results were of insufficient sensitivity. All of the in
vivo tests gave negative results. Overall, it appears that
1,1,1-trichloroethane does not have significant mutagenic potential.
Table 14. Mutagenicity studies
Test method Organism Test conditions Result References
In vitro studies on microorganisms
Baterial Salmonella typhimurium many studies, using mostly negative; some Simmon et al. (1977), Snow et al. (1979),
mutagenicity or Escherichia coli liquid or vapour, positive results attributed Nestmann et al. (1980), Gocke et al. (1981),
with and without S9 to stabilizers or impurities Rowland & Severn (1981), McDonald (1981),
activation Nagao & Takahashi (1981), Richold & Jones
(1981), Simmon & Shepherd (1981),
Ichinotsubo et al. (1981), de Serres &
Ashby (1981), HSE (1984), Shimada et al.
(1985)
Bacterial DNA E. coli or Bacillus several studies mainly negative; Kada (1981), Ichinotsubo et al. (1981),
damage/repair subtilis occasionally equivocal Green (1981), Tweats (1981), Rosenkranz
tests results et al. (1981), de Serres & Ashby (1981)
Yeasts, Saccharomyces numerous studies negative HSE (1984), de Serres & Ashby (1981),
gene mutation, cerevisiae
gene conversion Schizosaccharomyces
pombe
S. cerevisiae and mice 5000 mg/kg negative Loprieno et al. (1979)
(host-mediated assay) intraperitoneal
Table 14 (contd).
Test method Organism Test conditions Result References
In vitro mammalian cell studies
Chromosome CHO cells ± S9 activation clear positive without S9; Galloway et al. (1987)
aberrations equivocal with S9
Gene mutation L5178Y mouse ± S9 equivocal with S9; Myhr & Caspary (1988)
lymphoma cells negative without S9
Unscheduled rat, mouse hepatocytes; ± S9 negative Althaus et al. (1982), Martin &
DNA synthesis HeLa cells McDermid (1981)
DNA repair rat hepatocytes ± S9 mainly negative; some Williams (1983), Shimada et al. (1985)
positive results attributed
to presence of stabilizers
or impurities
Sister chromatid CHO cells ± S9 negative Perry & Thomson (1981)
exchange equivocal Galloway et al. (1987)
Cell various cell types ± S9 both positive and Daniel & Dehnel (1981), Styles (1981),
transformation negative results Price et al. (1978), Hatch et al. (1983)
Tu et al. (1985), HSE (1984)
Drosophila studies
Sex-linked 25 nM negative Gocke et al. (1981)
recessive
lethals
Table 14 (contd).
Test method Organism Test conditions Result References
In vivo mammalian studies
Chromosome rats 875 or 1750 ppm negative Quast et al. (1978)
aberrations 6 h/day, 52 weeks
(bone marrow)
Micronucleus mice 34-67 mg/kg negative Salamone et al. (1981)
intraperitoneal
Micronucleus mice 11-42 mg/kg negative Tsuchimoto & Matter (1981)
intraperitoneal
Micronucleus mice 266-2000 mg/kg negative Gocke et al. (1981)
intraperitoneal
Dominant mice 100-1000 mg/kg per negative Lane et al. (1982)
lethal day oral
Sperm mice 130-2680 mg/kg per negative Topham (1980)
morphology day intraperitoneal
DNA binding rats and mice low binding Prodi et al. (1988)
(liver)
7.5 Carcinogenicity
1,1,1-Trichloroethane has been investigated for carcinogenic
effects in life-time bioassays on both rats and mice by oral
administration or inhalation.
7.5.1 Oral administration
In a US National Cancer Institute (NCI) assay, 7 week-old
Osborne-Mendel rats were fed by gavage a 75% solution of technical
grade 1,1,1-trichloroethane in corn oil, 5 days/week for 78 weeks.
The daily doses were calculated to be 750 or 1500 mg/kg body weight,
and the study was terminated after 110 weeks. The number of animals
in each treated group was 50 of each sex, and the control groups
consisted of 20 animals of each sex, which were not vehicle dosed. No
increased tumour incidence was found (NCI, 1977). However, because of
an unusually high mortality both among treated and control animals
(only 6 out of 240 animals survived the 110 weeks to the end of the
experiment), this study was considered insufficient by US EPA (1984)
and placed in class D (not applicable to assess human
carcinogenicity).
In another NCI study with 5-week-old B6C3F1 mice, technical grade
trichloroethane containing 3% 1,4-dioxane was given by gavage as a
40-60% solution in corn oil at two dose levels (average daily doses:
2807 and 5615 mg/kg body weight) 5 days/week for 78 weeks. The same
number of animals was used as in the NCI rat study reported above.
All surviving animals were killed after 90 weeks of study. A variety
of neoplasms was observed in the treated groups, but the incidence was
not statistically significantly different from that of the control
groups (NCI, 1977). Again the mortality rate was very high in both
treated and control animals, although it was a little lower than in
the rat study. Nevertheless this study was also considered
insufficient (US EPA 1984).
In a study by Maltoni et al. (1986), groups of male and female
rats were given 1,1,1-trichloroethane (500 mg/kg body weight) in olive
oil by gavage (4 to 5 days/week for 104 weeks). The
1,1,1-trichloroethane contained about 4% 1,4-dioxane and 1% of other
impurities. An increased incidence of "leukaemias" was detected in
the exposed group. However, data were not evaluated statistically nor
were any data for historical controls supplied.
7.5.2 Inhalation
In an inhalation study carried out by the Dow Chemical Co.,
1-month-old Sprague-Dawley rats were exposed to technical
1,1,1-trichloroethane (6 h/day, 5 days/week for 12 months) and
observed 18 months later, before the survivors were killed. Each dose
group consisted of 96 rats of each sex. The unexposed control group
consisted of the same number of rats as the two exposed groups
together. The two exposure concentrations used were 4778 mg/m3 (875
ppm) and 9555 mg/m3 (1750 ppm). The treatment had no significant
effect on mortality, and the tumour incidence in the treated animals
was similar to that of the controls (Rampy et al., 1977). The purity
of the 1,1,1-trichloroethane used was 96%, and the stabilizers were 3%
1,4-dioxane, 0.4% nitromethane, and 0.5% butylene oxide. In the
validation of this study, it was pointed out that the exposure period
was only 12 months and not lifelong, that no subchronic range-finding
test had been done, and that the exposure concentration could have
been too low (US EPA, 1984).
A more recent long-term inhalation study, this time on both rats
and mice, has been carried out by the Dow Chemical Co. (Quast et al.,
1988). Groups of male and female 4- to 6-week-old Fischer-344 rats
and 5- to 6-week-old B6C3F1 mice (80 of each sex per group) were
exposed to 1,1,1-trichloroethane (technical formulation) vapour
concentrations of 0 mg/m3, 820 mg/m3 (150 ppm), 2730 mg/m3 (500
ppm), 8190 mg/m3 (1500 ppm) 6 h/day, 5 days/week for 2 years. Ten
animals of each sex from each group were predesignated for interim
sacrifices after 6, 12, and 18 months of exposure. The purity of the
formulation was 94% (by volume) 1,1,1-trichloroethane, 5% stabilizers
(butylene oxide, tert-amyl alcohol, methyl butynol, nitroethane, and
nitromethane) and < 1% minor impurities. In none of the treated
animal groups, did the tumour incidence differ significantly from that
of the controls.
The early studies with mice and rats were evaluated at IARC
(International Agency for Research on Cancer) and considered
inadequate (IARC, 1979; IARC, 1987).
7.6 Immunotoxicity and sensitization
Shmuter (1977) exposed rabbits to 1,1,1-trichloroethane
concentrations of 2 mg/m3 (0.4 ppm), 10 mg/m3 (1.8 ppm), and 100
mg/m3 (18 ppm) by inhalation 3 h/day, 6 days/week for 8-10 months.
At week 6, Salmonella typhimurium was injected subcutaneously to
investigate the immune response over an 8-month period. The antibody
response was depressed at both 10 mg/m3 and 100 mg/m3. This
reduction in immune response was followed by an increased
electrophoretic mobility of the antibodies toward alpha- and
ß-globulin fractions and also by a significant drop in the level of
normal haemolysins to the Forsman's antigen of the sheep erythrocytes
occuring during treatment.
7.7 Interactions
Interaction between 1,1,1-trichloroethane and ethanol was studied
in mice given ethanol (0-2 g/kg body weight) by gavage 30 min before
exposure to up to 120 000 mg trichloroethane/m3 (22 200 ppm) by
inhalation. Different combinations of exposure to the solvents
enhanced the toxicity of trichloroethane in the animals previous
exposed to ethanol. The observed behavioural and lethal effects were
dose related for each compound and additive (Woolverton & Balster,
1981).
When dogs anaesthetized with either pentobarbital sodium or
chloralose plus pentobarbital sodium were acutely exposed to a
commercial spot remover containing 1,1,1-trichloroethane, there was a
dose-dependent biphasic decline in arterial pressure with an initial
increase in the heart rate. A similar effect was obtained when
1,1,1-trichloroethane (99.5% by volume) was used instead of the
commercial spot remover. It was found that the type of anaesthetic
contributed in part to the effect. The authors reported that, in
addition to the peripheral vascular effects, significant alterations
in myocardial function occurred, and they concluded that the solvent
acts by inhibiting myocardial contractility (Herd et al., 1974).
7.8 Mechanisms of action
1,1,1-Trichloroethane given to mice intraperitoneally or by short
inhalation exposure has been shown to reduce the cyclic guanosine
monophosphate (cGMP) content in the cerebellum, cerebral cortex, and
brain stem. This was attributed to an increased rate of cGMP
hydrolysis (Nilsson, 1986a). The cyclic adenosine monophosphate
(cAMP) content in the brain stem of mice was increased in a
dose-related and time-dependant manner. This might be mediated by
adrenoceptor interaction stimulating the catalytic unit of adenylate
cyclase (Nilsson, 1986b).
A study on isolated mouse brain synaptosomes exposed in vivo
and in vitro showed that 1,1,1-trichloroethane increases calcium
influx in the brain stem but not in the cerebellum and cerebral
cortex. The data indicated that 1,1,1-trichloroethane may influence
voltage-dependant calcium channels in brain stem nerve endings
(Nilsson, 1987).
1,1,1-Trichloroethane suppressed the uptake of ovabain
taurocholate and 2-aminoisobutyric acid, but not of cadmium chloride
or 3- O-methyl-D-glucose, into isolated rat hepatocytes. Both the
cellular ATP level and ATP-ase activities in isolated membrane
fractions were also decreased. It was suggested that energy-dependant
transport functions are disrupted, owing to decreased ATP levels
and/or inhibition of membrane ATP-ases (Kukongviriyapan et al., 1990).
8. EFFECTS ON HUMANS
Appraisal
Both acute and long-term inhalation exposure to
1,1,1-trichloroethane can produce CNS effects ranging from slight
behavioural changes to unconsciousness. Death may follow from
respiratory failure or cardiac arrest. There is also the possibility
of hepatic alterations and cardiac damage.
Ingestion and dermal exposure do not appear to be serious
concerns, although skin irritation may occur.
1,1,1-Trichloroethane has properties (see chapter 2) that make it
a dangerous compound in poorly ventilated areas and confined spaces
such as tanks and vaults. The vapour is five times more dense than
air, which may result in non-uniform distribution, and very high
levels can occur in certain areas such as the bottom of empty tanks
and container vessels. The combination of these properties has been
the cause of some serious accidents, including fatalities. Solvent
abuse has also produced a large number of fatalities.
There are insufficient studies to permit any conclusion
concerning the reproductive and carcinogenic effects of this compound
on humans.
Exposure to 1,1,1-trichloroethane may increase sensitivity to
other compounds, e.g., anaesthetics.
The NOEL for humans appears to be in the region of 1350 mg/m3
(250 ppm).
Table 15 summarizes the toxic effects of 1,1,1-trichloroethane
after inhalation exposure.
8.1 Controlled human studies
Several studies on volunteers under controlled conditions have
investigated the effects of 1,1,1-trichloroethane on the CNS as
measured by performance in certain behavioural tests. These studies
have usually been conducted using the technical grade of
1,1,1-trichloroethane containing stabilizers, since this is the type
of exposure that occurs most frequently.
Table 15. Toxic effects in humans related to inhalation exposure to 1,1,1-trichloroethane
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
378 000 70 000 a few min death Northfield (1981)
54 000 10 000 2 min general anaesthesia when given Dornette & Jones (1960)
together with nitrous oxide (N2O)
> 27 000 > 5000 10 min death Kleinfeld & Feiner (1966)
27 000 5000 5 min marked incoordination Stewart (1968)
10 260 1900 5 min disturbance of equilibrium Torkelson et al. (1958)
5400 1000 short-term mild eye and nasal discomfort, Stewart (1968)
impairment of coordination
5000 920 70-75 min CNS disturbances (lightheadedness), Torkelson et al. (1958)
slight eye irritation
4860 900 73 min impaired performance in Romberg's Stewart et al. (1961)
test, mild eye irritation
2700 500 6-7 h/day for impaired performance in Romberg's Stewart et al. (1969)
5 days test after 1 h of exposure, mild
eye irritation, drowsiness, headaches
2700 500 90-450 min no CNS disturbances Torkelson et al. (1958)
2430 450 30 min dizziness, mild eye irritation Salvini et al. (1971)
2430 450 30 min reduced reaction time and Gamberale & Hultengren (1973)
perceptual speed
Table 15 (contd).
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
1890 350 30 min reduced perceptual speed Gamberale & Hultengren (1973)
1900 350 3.5 h reduced psychomotor performance Mackay et al. (1987)
< 1860 < 345 6.7 years NOEL Maroni et al. (1977)
< 1350 < 250 1-6 years NOEL Kramer et al. (1978)
1350 250 30 min NOEL Gamberale & Hultengren (1973)
950 175 3.5 h reduced reaction time MacKay et al. (1987)
540 100 odour threshold Stewart (1968)
8.1.1 Single exposure period
In a study by Torkelson et al. (1958), four volunteers were
exposed in an inhalation chamber to average trichloroethane levels of
4968 mg/m3 (920 ppm) for 70-75 min. Symptoms of slight CNS
disturbances (lightheadedness) were noted in three of the four
subjects and slight eye irritation was noted in one individual. Two
volunteers reported a strong odour during the experiment.
Concentrations in the range of 2700-2970 mg/m3 (500-550 ppm) for
90-450 min apparently produced no symptoms of CNS disturbance, but
exposure to 10 260 mg/m3 (1900 ppm) for 5 min produced a very
noticeable odour and obvious disturbances in equilibrium.
Unfortunately, full details of this study are not available.
Exposure to about 2700 mg/m3 (500 ppm) trichloroethane for
78-186 min did not result in CNS toxicity or other effects (Stewart et
al., 1961). However, exposure to about 4860 mg/m3 (900 ppm) for up
to 73 min resulted in CNS disturbances (light-headedness and impaired
Romberg test performance) in approximately half of the subjects. Mild
eye irritation was also observed. In another experiment in this
series, the volunteers were exposed to increasing concentrations in
the range of 0-14 310 mg/m3 (0-2650 ppm) over a 15-min period.
Signs of mild eye irritation were noted in six out of seven subjects
when the concentration reached about 5400 mg/m3 (1000 ppm), and
there was a rapid increase in CNS symptoms, principally dizziness.
Throat irritation was experienced by six out of seven subjects at
about 10 800 mg/m3 (2000 ppm). Severe CNS disturbances (imbalance,
light-headedness) occurred at the end of the exposure period, when the
concentration had reached 14 310 mg/m3. In addition, minor effects
on liver and kidney function were seen. The authors considered that
all the effects were reversible.
The effect on young men of exposure to a mean trichloroethane
level of 2430 mg/m3 (450 ppm) for two periods of 4 h (separated by
a lunch break of 1´ h) was investigated by Salvini et al. (1971). The
volunteers experienced dizziness and slight excitation, but only
during the first 30 min of exposure, and mild eye irritation was
noted. No significant change in performance in any of the behavioural
tests was seen.
In a study by Gamberale & Hultengren (1973), 12 male volunteers
were exposed to 1350 mg/m3 (250 ppm) for 30 min, followed by 1890
mg/m3 (350 ppm) for the next 30 min, and 2430 mg/m3 (450 ppm) and
2970 mg/m3 (550 ppm) for subsequent 30-min periods. At 2430
mg/m3, the reaction time, perceptual speed, and manual dexterity
were reduced, and at 1890 mg/m3 the perceptual speed was reduced.
The NOEL was 1350 mg/m3.
Mackay et al. (1987) reported the results of an exposure chamber
study in which volunteers were exposed to 950 mg/m3 (175 ppm) and
1990 mg/m3 (350 ppm) for 3 to 5 h. Several human psychomotor
behavioural tests were employed, including two new ones. One was
concerned with distraction of attention and concentration (the Stroop
test) and the other with analysing grammatical statements (the
syntactic reasoning test). Performance deficits in the psychomotor
behavioural tests were recorded in volunteers exposed to
1,1,1-trichloroethane; these depended on the time of exposure and the
blood levels. The performance changes were rapid and occurred in some
cases within 20 min. However, different effects were found. In the
Stroop test, enhanced performance was observed following exposure,
whereas the syntactic reasoning test was found not to respond to
1,1,1-trichloroethane exposure effects. Measures of short-term
subjective well-being were not affected by exposure. Other
parameters, such as simple reaction time and four-choice reaction time
were increased.
The effect of exposure to trichloroethane on performance in a
very extensive range of behavioural tests was investigated by
Savolainen et al. (1981, 1982a, 1982b). Nine healthy male students
(aged 20-25) were exposed in an inhalation chamber to 1080 and 2160 mg
trichloroethane/m3 (200 and 400 ppm) for 4 h, with a 6-day interval
between exposure at the two levels. There was no clear indication of
any effects on the CNS, although some minor changes were noted in
body-sway measurements when the subjects had their eyes closed.
8.1.2 Repeated exposure
The effect of repeated exposure of male volunteers to 2700
mg/m3 (500 ppm), 7 h/day for 5 days, was studied in an inhalation
chamber (Stewart et al., 1969). Several subjective symptoms were
noted, e.g., drowsiness, headaches, lightheadedness, and eye and nose
irritation, but the absence of a control group made it difficult to
assess the significance. No performance impairment in behavioural
tests (modified Romberg test) was noted, apart from the inability of
two subjects to perform the balance test on two occasions.
8.2 Accidental exposure
Respiratory system symptoms, such as cough, breathlessness, and
chest tightness, are common in cases of acute poisoning (Boyer et al.,
1987).
Since 1,1,1-trichloroethane is a very volatile solvent with a
saturated vapour concentration of 864 g/m3 (160 000 ppm) at 25 °C,
there is a potential for very high air concentrations in confined
spaces, such as tanks and vaults. Furthermore, its vapour is five
times more dense than air. This may result in non-uniform
distribution in the workplace air and very high levels in certain
areas such as just above degreasing vessels and at the bottom of
empty tanks, even though levels elsewhere are low and safe. This
property has been the cause of some serious accidents and fatalities.
A near-fatal childhood intoxication was described by Gerace
(1981). A 4-year-old boy was playing under his bed covers with a
flower-making kit which contained a 30-cm3 container of
1,1,1-trichloroethane. Because of some unusual noises heard by a
sibling, the child was quickly discovered. However, he was not
breathing at this point. He was resuscitated on the way to the
hospital but remained comatose for about 12 min after arrival. The
author suggested that the child was rendered unconscious by the
1,1,1-trichloroethane and that the noises heard were seizures caused
by hypoxia. The child was released 48 h after admission. According
to the author, clinical investigation during the child's hospital stay
demonstrated some hepatic abnormalities with alteration in liver
bilirubin.
Caplan et al. (1976) reported the death of a woman due to
accidental exposure while cleaning a paint spill using a paint thinner
in a poorly ventilated room. Autopsy results showed "mild fatty
changes" in the liver and acute oedema and congestion of the lungs.
The concentrations of 1,1,1-trichloroethane in the brain, kidney,
liver, and blood were 36, 12, 5, and 2 mg/100 ml, respectively.
8.2.1 Confined spaces at workplaces
A man who had spent 10 min in a vault, where the concentration of
1,1,1-trichloroethane was estimated to be well in excess of 27 g/m3
(5000 ppm), collapsed and died shortly after leaving the vault
(Kleinfeld & Feiner, 1966).
Seven fatalities from using trichloroethane to clean equipment on
naval ships or aircraft tanks have been reported (Stahl et al., 1969;
Hatfield & Maykoski, 1970). The exposure level in one of these cases
was estimated to be in the order of 270 g/m3 (50 000 ppm). In all
cases the most significant pathological finding was pulmonary oedema.
Liver damage was not seen.
Other case histories with accidental death due to
1,1,1-trichloroethane exposure in confined spaces have been reported
(Stewart 1968; Bonventre et al., 1977).
During the period 1961-1980, the Factory Inspectorate in the
United Kingdom revealed 52 incidents due to trichloroethane (McCarthy
& Jones, 1983). Loss of consciousness was reported in 26 cases and
death in three cases, all involving very young workers. In one case
the worker died after leaning over an open tank of trichloroethane,
apparently to wash his hands in the solvent. He was found unconscious
slumped over the side of the tank, and the solvent concentration in
his breathing zone was later estimated to be about 378 g/m3 (70 000
ppm) (Northfield, 1981). In a second case, a worker was using
trichloroethane for cleaning purposes in a closed room (Jones &
Winter, 1983). He was found on the floor with chemical burns on the
head and neck, which was consistent with prolonged skin absorption
from spills of solvent on the floor. The third case was a teenage
worker who cleaned the interior of a car with a cloth soaked in the
solvent (Jones & Winter, 1983). Death was presumed to be from
1,1,1-tri-chloroethane intoxication and simultaneous inhalation of
vomit.
8.2.2 Solvent abuse
During the 1960s, 110 cases of sudden death from solvent abuse
were reported in the USA. Of these, spot remover containing
1,1,1-trichloroethane was identified as responsible for 29 cases
(Bass, 1970). Death frequently occurred immediately following
stressful activity. Where autopsies were performed, no abnormalities
could be detected, and it was suggested that deaths was due to cardiac
arrhythmia.
Over the period 1971-1981, 140 deaths from solvent abuse were
reported in the United Kingdom; 20 of them were believed to be due to
1,1,1-trichloroethane, mainly in dry-cleaning fluid, domestic cleaners
or plaster remover (Anderson et al., 1982).
In the case of a fatality due to the abuse of cleaning solvent
containing 1,1,1-trichloroethane, very minor hepatic changes were seen
(Hall & Hine, 1966). In four cases of deaths caused by inhalation of
typewriter correction fluid containing 1,1,1-trichloroethane and
trichloroethylene, the autopsies showed no evidence of kidney or liver
damage (King et al., 1985).
According to Marjot & McLeod (1989), toluene and the chlorinated
hydrocarbons 1,1,1-trichloroethane and trichloroethylene can cause
permanent damage to the kidney, liver, heart, and lung in certain
volatile substance abusers.
8.2.3 Medical use
Trichloroethane has been used as an anaesthetic on an
experimental basis. General anaesthesia in humans has been rapidly
induced by 2 min of exposure to 54 000 mg/m3 (10 000 ppm) given with
a mixture of nitrous oxide (N2O) and oxygen (80:20). Cardiovascular
side effects (slight to severe hypotension, cardiac arrhythmias, and,
in one case, cardiac arrest) were observed in several of the patients
during anaesthesia. In view of these effects, the experimental use of
1,1,1-trichloroethane as an anaesthetic was discontinued (Dornette &
Jones, 1960).
In the USA, after aerosol drug products containing
1,1,1-trichloroethane had been implicated in 21 deaths from their
abuse or misuse, the Food and Drug Administration (FDA) removed the
products from the market (US Food and Drug Administration, 1973). The
medical products were mainly used as cough suppressants and contained
1,1,1-trichloroethane as a solvent for the active ingredients and to
reduce the vapour pressure of the propellants. The FDA found lack of
general recognition by qualified experts of the safety or
effectiveness of 1,1,1-trichloroethane in aerosol drug products
intended for inhalation.
Under 1977 rules from the FDA, it was stated that
1,1,1-trichloroethane is potentially toxic to the cardiovascular
system by sensitizing the heart to adrenaline (Food and Drug
Administration, 1977).
8.2.4 Ingestion
In a case of accidental ingestion of about 600 mg
trichloroethane/kg, signs of severe gastrointestinal irritation
(vomiting, diarrhoea) were evident shortly after the ingestion. There
was no evidence of any CNS disturbance or any liver or kidney
dysfunction (Stewart & Andrews, 1966).
Ingestion of about 30 ml trichloroethane has been found to cause
severe vomiting and diarrhoea (CEC, 1986).
8.2.5 Drinking-water contamination
In December 1981, a water well in Santa Clara County, California,
USA, was removed from service after contamination with
1,1,1-trichloroethane and other solvents was detected. A
1,1,1-trichloroethane level of 1.7 mg/litre was detected at the well
head, but the level of the other contaminants (dichloroethylene,
isopropyl alcohol, and freon) was not reported. Subsequently
suspicion of a cluster of spontaneous abortions and congenital heart
anomalies from pregnancies conceived during 1980-1981 was reported by
the local community. Epidemiological studies indicated that a cluster
did exist, that the odds ratio for spontaneous abortion was 2.3 and
the relative risk for congenital malformations was 3.1, but they were
unable to draw a direct link between the ingestion of the contaminated
water and these effects (Goldberg et al., 1990).
8.3 Effects on the skin and eyes
Prolonged or repeated skin contact with 1,1,1-trichloroethane may
lead to dermatitis, due to its defatting action. However, despite its
wide use only a few cases of skin irritancy have been reported
(Torkelson et al., 1958).
Immersion of a hand in liquid 1,1,1-trichloroethane for 30 min
resulted in mild erythema, which persisted for one hour (Stewart &
Dodd, 1964).
Splashed in the eye, the compound may produce conjunctivitis
(Stewart, 1968). However, accidental eye contact has been reported to
produce only transient irritation (Savolainen et al., 1982a,b).
Exposure to a vapour concentration of 2700 mg/m3 (500 ppm) may
produce mild eye irritation (see section 8.1).
8.4 Long-term occupational exposure
A case study described a patient with liver cirrhosis after
several years of heavy exposure to trichloroethylene followed by 3
months of work that frequently involved using aerosolized degreaser
containing trichloroethane (Thiele et al., 1982).
An investigation of the neurotoxicity of trichloroethane was
carried out on a group of 22 female workers exposed to the solvent for
an average period of 6.7 years. The group was subdivided into three
exposure groups (mean levels: 594 mg/m3 (110 ppm), 756-864 mg/m3
(140-160 ppm), and 1080-1863 mg/m3 (200-345 ppm)). Each woman had
a general physical examination, and information about symptoms was
obtained from a questionnaire. The measurement of nerve conduction
velocity and psychometric functions indicated no differences between
the exposed workers and an unexposed control group of 7 workers
(Maroni et al., 1977).
Hodgson et al. (1989) reported that four out of ten clinical
cases of fatty liver disease were associated with occupational
exposure to 1,1,1-trichloroethane.
A matching-pair study of 151 pairs of workers at textile plants,
with and without daily exposure to trichloroethane levels of up to
1365 mg/m3 (250 ppm), found no toxic effect among the exposed
workers, the majority of whom were exposed to less than 810 mg/m3
(150 ppm) for a period of 1-6 years (Kramer et al., 1978).
In a case-control study of the effect of occupational exposure to
various solvents on spontaneous abortion rates in Finland (1973-1983),
a statistically significant elevation in relative risk was found for
exposure to organic solvents in general. However, there were very few
cases related specifically to 1,1,1-trichloroethane. The low
statistical power, therefore, precludes any conclusion as to whether
the data show an effect or not for 1,1,1-trichloroethane (Lindbohm et
al., 1990)
There are no adequate epidemiological studies available
concerning long-term exposure to 1,1,1-trichloroethane and
carcinogenicity.
8.5 Interactions
Chronic cardiac arrhythmia developed in two cases, after routine
halothane anaesthesia, in subjects with previous repeated long-term
exposure to 1,1,1-trichloroethane (in one case, occupational and in
the other, due to solvent abuse). The effects involved myocardiac
damage with either ventricular arrhythmia or deterioration of left
ventricular function. In one of the cases, the pathologist suggested
a subacute myocarditis superimposed on long-standing damage. The
authors postulated that the interaction between the two chemicals may
have caused the cardiac deterioration and concluded that cardiac
damage sustained after long-term exposure to 1,1,1-trichloroethane may
be severe, life-threatening, and irreversible (McLeod et al.,1987)
Marjot & McLeod (1989) reported two cases, a 14-year-old boy
exposed to 1,1,1-trichloroethane via solvent abuse and a 54-year-old
man exposed occupationally, where the cardiac toxicity of
1,1,1-trichloroethane may have been increased by exposure to halothane
(2-bromo-2-chloro-1,1,1-trifluoroethane) as an anaesthetic.
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
Appraisal
1,1,1-Trichloroethane is not likely to be a significant hazard to
non-target organisms in the environment. For example, 96-h LC50
values for fish ranged from 33 mg/litre for dabs to 105 mg/litre for
fathead minnows. The 48-h LC50 for Daphnia was around 60 mg/litre.
The EC50 for carbon dioxide incorporation into algae was 5
mg/litre.
9.1 Microorganisms
A toxic threshold of 93 mg 1,1,1-trichloroethane/litre has been
estimated for the inhibition of cell multiplication in the bacterium
Pseudomonas putida (Bringmann & Kuhn, 1977). The 3-h EC50 values,
for inhibition of photosynthesis (measured by monitoring the uptake of
14CO2), for the green algae Chlamydomonas angulosa (at a cell
concentration of 5 x 104 cells/ml) and Chlorella vulgaris (at a
cell concentration of 20 x 104 cells/ml) were 280 mg/litre and 153
mg/litre, respectively, under static conditions (Hutchinson et al.,
1980). Bringmann & Kuhn (1978) calculated toxic thresholds of 350
mg/litre and 430 mg/litre, respectively, for inhibition of cell
multiplication, over an 8-day period, of the cyano bacterium
(blue-green alga) Microcystis aeruginosa and the green alga
Scenedesmus quadricauda. The 96-h EC50 for chlorophyll a and cell
number in the green alga Selenastrum capricornutum was > 669
mg/litre (US EPA, 1980a,b). Pearson & McConnell (1975) assessed the
toxicity of 1,1,1-trichloroethane to the unicellular alga
Phaeodactylum tricornutum by measuring changes in the uptake of
carbon, from 14CO2, during photosynthesis, and obtained an EC50
of 5 mg/litre.
9.2 Aquatic organisms
The acute toxicity of 1,1,1-trichloroethane to aquatic organisms
is summarized in Table 16.
Flow-through tests gave much lower LC50 values than static
tests, suggesting that some loss via evaporation may be occurring.
The 48-h LC50 for aquatic invertebrates ranges from 7.5 mg/litre for
barnacles in a flow-through test to > 530 mg/litre for daphnids in a
static test. The 96-h LC50 for fish ranges from 33 mg/litre for the
dab (Limanda limanda) in flow-through tests to 105 mg/litre for the
fathead minnow (Pimephales promelas) in static tests.
Table 16. Acute toxicity of 1,1,1-trichloroethane to aquatic organisms
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Invertebrates
Mysid shrimp stat 96 31.2 n US EPA (1980b)
(Mysidopsis bahia)
Barnacle nauplii stat 48 7.5 n Pearson & McConnell (1975)
(Eliminius modestus)
Water flea < 24 h stat 21-23 72 6.7-8.1 48 > 530 n LeBlanc (1980)
(Daphnia magna) 4-6 day stat 21-25 48 59.6 Abernethy et al. (1986)
4-6 day stat 48 57.6 Bobra et al. (1984)
Fish
Dab 15-20 cm flow 96 33 m Pearson & McConnell (1975)
(Limanda limanda)
Sheepshead minnow 8-15 mm stat 25-31 10-31d 96 71 (60-81) n Heitmuller et al. (1981)
(Cyprinodon variegatus)
Bluegill 0.32-1.2 g stat 21-23 32-48 6.7-7.8 24 110 n Buccafusco et al. (1981)
(Lepomis macrochirus) 0.32-1.2 g stat 21-23 32-48 6.7-7.8 96 72 (57-90) n Buccafusco et al. (1981)
stat 96 69.7 n US EPA (1980b)
Fathead minnow 1 g stat 12 7.8-8.0 96 105 (91-126) n Alexander et al. (1978)
(Pimephales promelas) 1 g flow 12 7.8-8.0 96 52.8 (43.7-77.7) m Alexander et al. (1978)
Table 16 (contd).
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Fish (contd)
Guppy 2-3 month statb 168 133 Konemann (1981)
(Poecilia reticulata)
a stat = static conditions (water unchanged for the duration of the test) unless stated otherwise; flow = flow-through conditions
(1,1,1-trichloroethane concentration continuously maintained)
b static conditions but water renewed daily
c hardness expressed as mg CaCO3 per litre
d value refers to salinity (%), not hardness
e n = nominal; m = measured
LeBlanc (1980) exposed the water flea Daphnia magna to
1,1,1-trichloroethane for 48 h under static conditions. At the
highest concentration of 530 mg/litre, no discernible effect was
observed. A no-observed-effect level (NOEL) of 43 mg/litre was
estimated by Heitmuller et al. (1981) after exposing the sheepshead
minnow (Cyprinodon variegatus) to 1,1,1-trichloroethane for 96 h.
When Thompson & Carmichael (1989) exposed the water flea Daphnia
magna to 1,1,1-trichloroethane concentrations of between 1.3 and 23
mg/litre for 17 days under semistatic conditions (water renewed every
2 days), no effect on mortality or reproduction (as measured by number
of offspring per parent) was observed at 1.3 mg/litre. However, the
mortality increased with increasing concentration of
1,1,1-trichloroethane from 30% at 2.4 mg/litre to 100% at 23 mg/litre.
The number of offspring per parent was significantly reduced at
concentrations of 2.4 mg/litre or more.
Thompson & Carmichael (1989) also exposed the mirror carp
Cyprinus carpio to 1,1,1-trichloroethane concentrations ranging from
1.4 to 30 mg/litre for 14 days under flow-through conditions. There
was no mortality at any of the concentrations, but at the highest
concentration the fish showed surfacing behaviour, loss of balance and
coughing, and their mean weight decreased. The authors considered,
therefore, that the NOEL was the next highest concentration, i.e. 7.7
mg/litre.
Alexander et al. (1978) observed fathead minnows (Pimephales
promelas), during exposure to 1,1,1-trichloroethane for up to 96 h,
for the following effects: loss of equilibrium, melanization,
narcosis, and swollen, haemorrhaging gills. The concentration
producing one or more of these observable effects in 50% of the fish
(EC50) was 11.1 mg/litre.
9.3 Terrestrial organisms
Thompson & Carmichael (1989) exposed emergent seedlings of crop
sorghum (Sorghum bicolor) and oil seed rape (Brassica napus) to
1,1,1-trichloroethane in the gaseous phase at nominal concentrations
of between 3.2 and 320 mg/litre (g/m3) for a period of 14 days (the
test material was replenished after 7 days in a sealed vessel). Mean
measured concentrations ranged from 58% to 75% of the nominal
concentration, reflecting the decline of 1,1,1-trichloroethane in the
gas phase concentration during the 7-day periods between toxicant
renewal. In the case of Sorghum bicolor, the 14-day EC50 (based
on mean plant weight and a mean measured 1,1,1-trichloroethane
concentration) was 48 mg/litre; the NOEL being 19 mg/litre. For
Brassica napus, the 14-day EC50 was 19 mg/litre and the NOEL 6.9
mg/litre, both values being based on mean measured concentrations.
Rajendran (1990) exposed 1- to 2-day-old pupae of the red flour
beetle (Tribolium castaneum) to 1,1,1-trichloroethane in fumigation
chambers. An LC50 of 208.4 mg/litre was estimated after a 24-h
exposure and a 20-day post-fumigation observation period. When pupae
were exposed to a mixture of 1,1,1-trichloroethane and methyl bromide
at a concentration of 2.5 mg/litre or 3.0 mg/litre, the toxicity was
similar to that of the individual components.
In a contact toxicity test, red earthworms (Eisenia foetida)
were exposed to 1,1,1-trichloroethane via filter paper in glass vials.
An LC50 of 83 µg/cm2 was estimated (Neuhauser et al., 1986).
Elovaara et al. (1979) injected 1,1,1-trichloroethane, at
concentrations ranging from 5 to 100 µmol/egg, into the air space of
fertilized chicken eggs at 3 or 6 days of incubation, and the
embryotoxicity was evaluated after 14 days of incubation. A clear
dose-response relationship with respect to survival was found,
irrespective of whether the eggs were injected after 3 or 6 days of
incubation. The approximate LD50 for 1,1,1-trichloroethane was
between 50 and 100 µmol/egg.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
Humans are exposed to 1,1,1-trichloroethane principally by
inhalation, where the substance is rapidly absorbed into the body.
Exposure by skin absorption or ingestion may also occur but, compared
to inhalation, is of lesser concern. 1,1,1-Trichloroethane is
considered not to bioaccumulate. The acute and chronic toxicities are
fairly low. Under conditions of high exposure there is a risk of
toxic effects; such conditions may occur in occupational exposures,
solvent abuse or accidents. Since 1,1,1-trichloroethane is volatile
and the vapour is much more dense than air, toxic and explosive
concentrations may occur unexpectedly in confined spaces. This has
caused several fatal and near-fatal accident at workplaces and
elsewhere. The critical effect in humans relates to the central
nervous system, leading to unconsciousness and respiratory arrest at
high concentrations. 1,1,1-Trichloroethane is less toxic to the liver
than most other organochlorine solvents. The no-observed-effect level
for humans appears to be in the region of 1350 mg/m3 (250 ppm).
Although one study suggested effects at a lower level, the
interpretation of the results was unclear.
Studies on Mongolian gerbils involving 1,1,1-trichloroethane
levels down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several brain regions.
If these findings are validated and confirmed, this may be of
significant concern.
No adequate study of carcinogenic effects on humans has been
published. A long-term inhalation study on rats and mice exposed to
8100 mg/m3 (1500 ppm) gave no evidence of any carcinogenic effects.
One study recorded increases numbers of "leukaemias", but it was
performed under non-standard protocol. It gave no statistical
evaluation and is, therefore, considered inadequate. It is unlikely
that 1,1,1-trichloroethane itself presents a significant risk of
genotoxic or carcinogenic effects in humans.
1,1,1-Trichloroethane is not teratogenic and elicits no
developmental effects at doses that are not maternally toxic. The
limited epidemiological evidence on reproductive effects is
inconclusive due to the low statistical power of the study.
10.2 Evaluation of effects on the environment
1,1,1-Trichloroethane is released in large quantities to the
environment. Due to its volatility and general stability it is a
ubiquitous environmental contaminant.
Although the ozone-depleting and global-warming potentials of
1,1,1-trichloroethane are lower than those of many
chlorofluorocarbons, the large-scale release of the compound into the
atmosphere merits serious concern in these respects.
As a result of its widespread use and disposal and its mobility
in soil, persistent 1,1,1-trichloroethane contamination of ground
water, including deep aquifers, occurs.
Conditions under which 1,1,1-trichloroethane causes
ecotoxicological effects are not found in the environment except after
spills.
11. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH AND
THE ENVIRONMENT
a) Emissions of 1,1,1-trichloroethane should be reduced as far as
is practicable in order to minimize exposure of workers and of
the general population.
b) The release of 1,1,1-trichloroethane into the environment must
be reduced to the greatest extent possible in order to avoid
damage to the ozone layer.
c) Contamination of ground water by 1,1,1-trichloroethane from
spills and waste disposal should be avoided.
d) Safe substitutes for 1,1,1-trichloroethane should be identified.
12. FURTHER RESEARCH
a) More study is needed to clarify the possible effects of
1,1,1-trichloroethane on the biochemistry of the central nervous
system and their significance.
b) More study is necessary to clarify the possible interaction
between 1,1,1-trichloroethane and ethanol in humans.
c) An adequate subchronic or long-term oral study is needed to
assess the hepatotoxicity of 1,1,1-trichloroethane.
d) Further epidemiological studies on reproductive effects are
desirable.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
IARC (International Agency for Research on Cancer) working groups
have evaluated 1,1,1-trichloroethane and concluded that the evidence
for genotoxicity is limited (IARC, 1979, 1987).
REFERENCES
ABERNETHY, S., BOBRA, A.M., SHIU, W.Y., WELLS, P.G., & MACKAY, D.
(1986) Acute lethal toxicity of hydrocarbons and chlorinated
hydrocarbons to two planktonic crustaceans: The key role of
organism-water partitioning. Aquat. Toxicol., 8: 163-174.
ADAMS, E.M., SPENCER, H.C., ROWE, V.K., & IRISH, D.D. (1950) Vapor
toxicity of 1,1,1-trichloroethane (methyl chloroform) determined by
experiments on laboratory animals. Arch. ind. Hyg., 1: 225-236.
AGGAZZOTTI, G. & PREDIERI, G. (1986) Survey of volatile halogenated
organics (VHO) in Italy. Water Res., 20: 959-963.
ALEXANDER, H.C., MCCARTY, W.M., & BARTLETT, E.A. (1978) Toxicity of
perchloroethylene, trichloroethylene, 1,1,1-trichloroethane, and
methylene chloride to fathead minnow. Bull. environ. Contam. Toxicol.,
20: 344-352.
ALTHAUS, F.R., LAWRENCE, S.D., SATTLER, G.L., LONGFELLOW, D.G., &
PITOT, H.C. (1982) Chemical quantification of unscheduled DNA
synthesis in cultured hepatocytes as an assay for the rapid screening
of potential chemical carcinogens. Cancer Res., 42(8): 3010-3015.
AMI (1988) [The Factories Inspectorate's data base for workplace
measurements, ATABAS, stock-taking for the period January 1983 -
December 1987], Copenhagen, Institute for the Working Environment (in
Danish).
ANDERSON, H.R., DICK, B., MACNAIR, R.S., PALMER, J.C., & RAMSEY, J.D.
(1982) An investigation of 140 deaths associated with volatile
substance abuse in the United Kingdom (1971-1981). Hum. Toxicol., 1:
207-221.
ARCHER, W.L. (1979) 1,1,1-Trichloroethane. In: Kirk-Othmer
encyclopedia of chemical technology, 3rd ed., New York, Chichester,
John Wiley and Sons, pp. 728-731.
ARCHER, W.L. & STEVENS, V.L. (1977) Comparison of chlorinated,
aliphatic, aromatic, and oxygenated hydrocarbons as solvents. Ind.
Eng. Chem. Prod. Res. Dev., 16: 319-325.
ASTRAND, I., KILBOM, A., WAHLBERG, I., & OVRUM, P. (1973) Methyl
chloroform exposure. I. Concentration in alveolar air and blood at
rest and during exercise. Scand. J. Work Environ. Health, 10: 69-81.
BASS (1970) Sudden sniffing death. J. Am. Med. Assoc., 212: 2075-2079.
BAUER, U. (1981) [Burden on man caused by harmful substances in the
environment.] Zbl. Bakteriol. Hyg. I Abt. Orig. B, 174: 200-237 (in
German).
BERODE, M., BOILLAT, M.-A., GUILLEMIN, M.P., WU, M.W., & SAVOLAINEN,
H. (1990) Demethylation pathways in caffeine metabolism as indicators
of variability in 1,1,1-trichloroethane oxidation in man. Pharmacol.
Toxicol., 67(1): 41-46.
BOBRA, A.M., SHIU, W.Y., & MACKAY, D. (1984) Structure-activity
relationships for toxicity of hydrocarbons, chlorinated hydrocarbons
and oils to Daphnia magna. In: Kaiser, K.L.E., ed. Quantitative
structure activity relations in environmental toxicology, Dordrecht,
Germany, D. Reidel Publishing Co., pp. 3-16.
BONNET, P., FRANCIN, J.-M., GRADISKI, D., RAOULT, G., & ZISSU, D.
(1981) Determination of the median lethal concentration of the
principal chlorinated aliphatic hydrocarbons in the rat. Arch. Mal.
prof. Méd. Trav. Sécur. soc., 41: 317-321.
BONVENTRE, J., BRENNAN, O., JASON, D., HENDERSON, A., & BASTOS, M.L.
(1977) Two deaths following accidental inhalation of dichloromethane
and 1,1,1-trichloroethane. J. anal. Toxicol., 4: 158-160.
BOUWER, E.J. & MCCARTY, P.L. (1983a) Transformations of 1- and
2-carbon halogenated aliphatic organic compounds under methanogenic
conditions. Appl. environ. Microbiol., 45: 1286-1294.
BOUWER, E.J. & MCCARTY, P.L. (1983b) Transformations of halogenated
organic compounds under denitrification conditions. Appl. environ.
Microbiol., 45: 1295-1299.
BOYER, J.D, AHLERT, R.C., & KOSSON, D.S. (1987) Degradation of
1,1,1-trichloroethane in bench-scale bioreactors. Hazardous Waste
hazardous Matter, 4: 241-260.
BRETHERICK, L., ed. (1981) Hazards in the chemical laboratory, 3rd
ed., London, The Royal Society of Chemistry.
BRETHERICK, L. (1989) 1,1,1-Trichloroethane. Chem. Eng. News, Dec. 12:
2.
BRINGMANN, G. & KUHN, R. (1977) [Limit values for the harmful effect
of water-endangering substances on bacteria (Psuedomonas putida) and
green algae (Scenedesmus quadricauda) in the cell division
inhibition test.] Z. Wasser Abwasser Forsch., 10: 87-98 (in German).
BRINGMANN, G. & KUHN, R. (1978) [Limit values for the harmful effect
of water endangering substances on blue algae (Microcystis
aeroginosa) and green algae (Scenedesmus quadricauda) in the cell
division inhibition test.] Vom Wasser, 50: 45-60 (in German).
BUCCAFUSCO, R.J., ELLS, S.J., & LEBLANC, G.A. (1981) Acute toxicity of
priority pollutants to bluegill (Lepomis macrochirus). Bull.
environ. Contam. Toxicol., 26: 446-452.
CAPLAN, Y.H., BACKER, R.C., & WHITAKER, J.Q. (1976)
1,1,1-Trichloroethane: report of a fatal intoxication. Clin. Toxicol.,
9: 69-74.
CEC (Commission of the European Communities) (1986) Organochlorine
solvents: Health risks to workers, Cambridge, Royal Society of
Chemistry, pp. 57-58.
CEFIC (1986) The occurrence of chlorinated solvents in the
environment. Prepared by a workshop of the European Council of
Chemical Industry Federation (CEFIC). Chem. Ind., 1986: 861-869.
CHIOU, C.T., PETERS, L.J., & FREED, V.H. (1979) A physical concept of
soil-water equilibria for nonionic organic compounds. Science, 206:
831-832.
CLARK, D.G. & TINSTON, D.J. (1982) Acute inhalation toxicity of some
halogenated and non-halogenated hydrocarbons. Hum. Toxicol., 1:
239-247.
CLASS, T. & BALLSCHMITER, K. (1986) Chemistry of organic traces in air
VI: Distribution of chlorinated C1 - C4 hydrocarbons in air over the
Northern and Southern Atlantic ocean. Chemosphere, 15: 413-427.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology, 3rd revised ed., New York, Chichester, John Wiley and
Sons, Vol. 2B.
COLEMAN, W.E., LINGG, R.D., MELTON, R.G., & KOPFLER, F.C. (1976) The
occurrence of volatile organics in five drinking water supplies using
gas chromatography/mass spectrometry. In: Keith, L.H., ed.
Identification and analysis of organic pollutants in water, Ann Arbor,
Michigan, Ann Arbor Science Publishers, Inc., pp. 305-327.
CORREIA, Y., MARTENS, G.J., VAN MESCH, F.H., & WHIM, B.P. (1977) The
occurrence of trichloroethylene, tetrachloroethylene and
1,1,1-trichloroethane in western Europe in air and water. Atmos.
Environ., 11: 1113-1116.
DAFT, J.L. (1988) Fumigant contamination during large-scale food
sampling for analysis. Arch. environ. Contam. Toxicol., 17: 177-181.
DAHLBERG, J.A., CHRISTIANSEN, V.O., & ERIKSEN, E.A. (1973) On the
formation of phosgene by photooxidation of methyl chloroform in
welding. Ann. occup. Hyg., 16: 41-46.
DALLAS, C.E., RAMANATHAN, R., MURALIDHARA, S., GALLO, J.M., &
BRUCKNER, J.V. (1989) The uptake and elimination of
1,1,1-trichloroethane during and following inhalation in rats.
Toxicol. appl. Pharmacol., 98: 385-397.
DANIEL, M.R. & DEHNEL, J.M. (1981) Cell transformation test with baby
hamster kidney cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 626-637 (Progress in Mutation Research, Vol. 1).
DANIELSSON, B.R.G., GHANTOUS, H., & DENCKER, L. (1986) Distribution of
chloroform and methylchloroform and their metabolites in pregnant
mice. Biol. Res. Pregnancy, 7: 77-83.
DAPSON, S.C., HUTCHEON, D.E., & LEHR, D. (1984) Effect of methyl
chloroform on cardiovascular development in rats. Teratology, 29: 25A.
DE NEVERS, N. (1986) A fatal fire with "non-flammable" methyl
chloroform. Arch. environ. Health, 41(5): 279-287.
DE SERRES, F.J. & ASHBY, J., ed. (1981) Evaluation of short term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland (Progress in
Mutation Research, Vol. 1).
DEVER, R.J. (1986) Responding to industrial contamination of
groundwater: a case study. J. Am. Water Works Assoc., 78: 82-86.
DICKSON, A.G. & RILEY, J.P. (1976) The distribution of short-chain
halogenated aliphatic hydrocarbons in some marine organisms. Mar.
Pollut. Bull., 7: 167-169.
DILLING, W.L., TERERTILLER, N.B., & KALLOS, G.J. (1975) Evaporation
rates and reactivities of methylene chloride, chloroform,
1,1,1-trichloroethane, trichloroethylene, tetrachloroethylene, and
other chlorinated compounds in dilute aqueous solutions. Environ. Sci.
Technol., 9: 833-838.
DORNETTE, W.H.L. & JONES, J.P. (1960) Clinical experience with
1,1,1-trichloroethane. Anesthesiol. Analg., 39: 249-253.
DRAKE, R.J. (1987) Fate of methylene chloride and
1,1,1-trichloroethane in unsaturated, biologically active soils,
University of Massachusetts (Dissertation).
DREHER, E.-L. (1989) Chlorinated hydrocarbons. In: Ullmann's
encyclopedia of chemical technology, New York, Weinheim, VCH
Publishers, Inc., Vol. A6, pp. 271-272.
DROZ, P.O., WU, M.M., CUMBERLAND, W.G., & BERUDE, M. (1989)
Variability in biological monitoring of solvent exposure. I
Development of a population physiological model. Br. J. ind. Med., 46:
447-460.
DUPRAT, P., DELSAUT, L., & GRADISKI, D. (1976) Pouvoir irritant des
principaux solvants chlorés aliphatiques sur la peau et les muqueuses
oculaires du lapin. Eur. J. Toxicol. environ. Hyg., 9: 171-177.
DÜSZELN, J.V., VON, LAHL, U., BÄTZER, K., CETINKAYA, M., STACHEL, B.,
& THIEMANN, W. (1982) [Volatile halogenated hydrocarbons in air, water
and food in the Federal Republic of Germany.] Dtsch. Lebensm.
Rundsch., 78: 352-356 (in German).
DÜSZELN, J.V., VON & THIEMANN, W. (1985) Volatile chlorinated
hydrocarbons in a costal urban atmosphere. Sci. total Environ., 41:
187-194.
EBEN, A. & KIMMERLE, G. (1974) Metabolism, excretion and toxicology of
methylchloroform in acute and subacute exposed rats. Arch. Toxicol.,
31: 233-242.
EDWARDS, P.R., CAMPBELL, I., & MILNE, G.S. (1982) The impact of
chloromethanes on the environment. Part 1. The atmospheric chlorine
cycle. Chem. Ind., 16: 574-578.
ELOVAARA, E., HEMMINKI, K., & VAINIO, H. (1979) Effects of methylene
chloride, trichloroethane, trichloroethylene and toluene on the
development of chick embryos. Toxicology, 12: 111-119.
ENTZ, R.C. & DIACHENKO, G.W. (1988) Residues of volatile halocarbons
in margarines. Food Addit. Contam., 5: 267-276.
ENTZ, R.C., THOMAS, K.W., & DIACHENKO, G.W. (1982) Residues of
volatile halocarbons in foods using headspace gas chromatography. J.
agric. food Chem., 30: 846-849.
FDA (1973) Trichloroethane. Aerosol drug products for human use. Fed.
Reg., 38: 21935-21936.
FDA (1977) Trichloroethane: status as a new drug in aerosolized drug
products intended for inhalation. FDA Final Rule. Fed. Reg., 42:
63386-63387.
FIELDING, M., GIBSON, T.M., JAMES, H.A., MCLOUGHLIN, K., & STEEL, C.P.
(1981) Organic micropollutants in drinking water, Medmenham, United
Kingdom, Water Research Centre (Technical Report No. TR 159).
FISCHER, M., FRIESEL, P., KOCH, G., MILDE, G., & ROSSKAMP, E. (1982)
Evaluation of water pollution caused by chlorinated and brominated
ethanes (Unpublished report submitted to the Commission of the
European Communities under Contract No. U71/516).
FISHBEIN, L. (1979) Potential halogenated industrial carcinogenic and
mutagenic chemicals. II. Halogenated saturated hydrocarbons. Sci.
total Environ., 11: 163-195.
FISHER, D.A., HALES, C.H., FILKIN, D.L., KO, M.K.W., SZE, N.D.,
CONNELL, P.S., WUEBBLES, D.J., ISAKSEN, I.S.A., & STORDAL, F. (1990a)
Model calculations of the relative effects of CFCs and their
replacement on stratospheric ozone. Nature (Lond.), 344: 508-512.
FISHER, D.A., HALES, C.H., WANG, W.-C., KO, M.K.W., & SZE, N.D.
(1990b) Model calculations of the relative effects of CFCs and their
replacement on global warming. Nature (Lond.), 344: 513-516.
FLUKA (1988) Catalogue 1988/89: Fluka Chemika-BioChemika, Buchs,
Switzerland, Fluka Chemie AG.
GALLOWAY, S.M., ARMSTRONG, M.J., REUBEN, C., COLMAN, S., BROWN, B.,
CANNON, C., BLOOM, A.D., NAKAMURA, F., AHMED, M., DUK, S. ET AL.
(1987) Chromosome aberrations and sister chromatid exchanges in
Chinese hamster ovary cells: Evaluations of 108 chemicals. Environ.
mol. Mutagen., 10: 1-175.
GAMBERALE, F. & HULTENGREN, M. (1973) Methylchloroform exposure II.
Psychophysiological functions. Scand. J. Work Environ. Health, 10:
82-92.
GEORGE, J.D., PRICE, C.J., MARR, M.C., SADLER, B.M., SCHWETZ, B.A.,
BIRNBAUM, L.S., & MORRISEY, R.E. (1989) Developmental toxicity of
1,1,1-trichloroethane in CD rats. Fundam. appl. Toxicol., 13, 641-651.
GERACE, R.V. (1981) Near-fatal intoxication by 1,1,1-trichloroethane.
Ann. Emergency Med., 10: 533-534.
GERKENS, R.R. & FRANKLIN, J.A. (1989) The rate of degradation of
1,1,1-trichloroethane in water by hydrolysis and dehydrochlorination.
Chemosphere, 19: 1919-1937.
GIGER, W., MOLNAR-KUBICA, E., & WAKEHAM, S. (1978) Volatile
chlorinated hydrocarbons in ground and lake waters. In: Aquatic
pollutants: Transformation and biological effects. Proceedings of the
2nd International Symposium on Aquatic Pollutants, Oxford, New York,
Pergamon Press, pp. 101-123 (Pergamon Series on Environmental Science
No. 1).
GOCKE, E., KING, M.-T., ECKHARDT, K., & WILD, D. (1981) Mutagenicity
of cosmetics ingredients licensed by the European Communities. Mutat.
Res., 90: 91-109.
GOLDBERG, S.J., LEBOWITZ, MD., GRAVER, E.J., & HICKS, S. (1990) An
association of human congenital cardiac malformations and
drinking-water contaminants. J. Am. Coll. Cardiol., 16: 155-164.
GOSSETT, J.M. (1985) Anaerobic degradation of C1 and C2 chlorinated
hydrocarbons, Tyndall AFB, Florida, US Air Force Engineering Services
Center (Technical Report No. ESL-TR-85-38).
GOSSETT, R.W., BROWN, D.A., & YOUNG, D.R. (1983) Predicting the
bioaccumulation of organic compounds in marine organisms using
octanol/water partition coefficients. Mar. Pollut. Bull., 14: 387-392.
GOTO, M. (1979) Monitoring environmental materials and specimen
banking. State-of-the-art of Japanese experience and knowledge. In:
Monitoring environmental materials and specimen banking, Proc. Int.
Workshop, Berlin (West) 23-28 October 1978, The Hague, Nijhoff
Publishers, pp. 271-288.
GRADISKI, D., MAGADUR, J.-L., BAILLOT, M., DANIERE, M.C., & SCHUH,
M.B. (1974) Toxicité comparée des principaux solvants chlorés
aliphatiques. J. eur. Toxicol., 7: 247-254.
GRADISKI, D., BONNET, P., RAOULT, G., MAGADUR, J., & FRANCIN, J.M.
(1978) Compared acute pulmonary toxicity of the main chlorinated
aliphatic solvents. Arch. Mal. prof. Méd. Trav. Sécur. soc., 39:
249-257.
GREEN, M.H. (1981) Differential killing test using an improved
repair-deficient strain of Escherichia coli. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
Oxford, Elsevier/North Holland, pp. 183-194 (Progress in Mutation
Resarch, Vol. 1).
GRIMSRUD, E.P. & RASMUSSEN, R.A. (1975) Survey and analysis of
halocarbons in the atmosphere by gas chromatography-mass spectrometry.
Atmos. Environ., 9: 1014-1017.
HAAG, W.R. & MILL, T. (1988) Effect of a subsurface sediment on
hydrolysis of haloalkanes and epoxides. Environ. Sci. Technol., 22:
658-663.
HAAG, W.R., MILL, T., & RICHARDSON, A. (1986) Extended abstracts of
the 192nd National Meeting of the American Chemical Society, Anaheim,
Washington, DC, American Chemical Society, Division of Environmental
Chemistry, Vol. 26, pp. 248-253.
HAKE, C.L., WAGGONER, T.B., ROBERTSON, D.N., & ROWE, V.K. (1960) The
metabolism of 1,1,1-trichloroethane by the rat. Arch. environ. Health,
1: 101-105.
HALL, F.B. & HINE, C.H. (1966) Trichloroethane intoxication: a report
of two cases. J. Forensic Sci., 11: 404-413.
HARDIE, D.W.F. (1964) 1,1,1-Trichloroethane. In: Kirk, R.E. & Othmer,
D.F., ed. Encyclopedia of chemical technology, 2nd ed., New York,
Chichester, Wiley and Sons, Vol. 5, pp. 154-157, 170.
HATCH, G.G., MAMAY, P.P., AYER, M.L., CASTO, B.C., & NESNOW, S. (1983)
Chemical enhancement of viral transformation in Syrian hamster embryo
cells by gaseous volatile chlorinated methanes and ethanes. Cancer
Res., 43: 1945-1950.
HATFIELD, T.R. & MAYKOSKI, R.T. (1970) A fatal methyl chloroform
(trichloroethane) poisoning. Arch. environ. Health, 20: 279-281.
HEITMULLER, P.T., HOLLISTER, T.A., & PARRISH, P.R. (1981) Acute
toxicity of 54 industrial chemicals to sheepshead minnows (Cyprinodon
variegatus) Bull. environ. Contam. Toxicol., 27: 596-604.
HENSCHLER, D., ed. (1978) [German Association for the Encouragement of
Research: Analytical methods for the testing of materials harmful to
health. I. Air analyses], Weinheim, Verlag Chemie (in German).
HENSCHLER, D., REICHERT, D., & METZLER, M. (1980) Identification of
potential carcinogens in technical grade 1,1,1-trichloroethane. Int.
Arch. occup. environ. Health, 47: 263-268.
HERD, P.A., LIPSKY, M., & MARTIN, H.F. (1974) Cardiovascular effects
of 1,1,1-trichloroethane. Arch. environ. Health, 28: 227-233.
HOBARA, T., KOBAYASHI, H., HIGASHIHARA, E., IWAMOTO, S., KAWAMOTO, T.,
SAKAI, T., & TSUBOTA, N. (1982) [Experimental examinations and
toxicokinetic analysis of the absorption and excretion of
1,1,1-trichloroethane by the lung.] Jpn. J. ind. Health, 24: 599-607
(in Japanese).
HODGSON, M.J., HEYL, A.E., & VAN THIEL, D.H. (1989) Liver disease
associated with exposure to 1,1,1-trichloroethane. Arch. intern. Med.,
149: 1791-1798.
HORIGUCHI, S. & HORIGUCHI, K. (1971) An experiment of
1,1,1-trichloroethane vapour exposure to mice. Sangyo Igaku, 13:
226-227.
HSE (1984) 1,1,1-Trichloroethane, London, Health and Safety Executive.
HSE (1990) Guidance note EH40/90. Occupational exposure limits 1990,
London, Health and Safety Executive.
HUMBERT, B.E. & FERNANDEZ, J.G. (1977) Exposition au
1,1,1-trichloroéthane: Contribution à l'étude de l'absorption, de
l'excrétion et du métabolisme sur des sujets humains. Arch. Mal.
prof., 38: 415-425.
HUTCHINSON, T.C., HELLEBUST, J.A., TAM, D., MACKAY, D., MASCARENHAS,
R.A., & SHIU, W.Y. (1980) The correlation of the toxicity to algae of
hydrocarbons and halogenated hydrocarbons with their physical-chemical
properties. Environ. Sci. Res., 1: 577-586.
IARC (1979) 1,1,1-Trichloroethane. In: Some halogenated hydrocarbons,
Lyon, International Agency for Research on Cancer, pp. 515-532 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Vol. 20).
IARC (1987) Overall evaluations of carcinogenicity: An updating of
IARC Monographs, Volumes 1 to 42, Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluations of Carcinogenic
Risk to Humans, Suppl. 7).
ICHINOTSUBO, D., MOWER, H., & MANDEL, M. (1981) Mutagen testing of a
series of paired compounds with the Ames Salmonella testing system.
In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland, pp. 298-301
(Progress in Mutation Research. Vol. 1).
IKEDA, M. & OHTSUJI, H. (1972) A comparative study of the excretion of
Fujiwara reaction-positive substances in urine of humans and rodents
given trichloro- or tetrachloro-derivatives of ethane and ethylene.
Brit. J. ind. Med., 29: 99-104.
IVANETICH, K.M. & HONERT, L.H., VAN DEN (1981) Chloroethanes: their
metabolism by hepatic cytochrome P-450. Carcinogenesis, 2: 697-702
JAKOBSON, M., LIEPINS, M., & ERINS, P. (1980) [Effect of changes in
the reticular structure of a lignocarbohydrate matrix on the
properties of the wood. 2. Effect of cross-linking on the
hydrophilicity of birchwood.] Khim. Drev., 1: 37-46 (in Russian).
JAKOBSON, I., WAHLBERG, J.E., HOLMBERG, B., & JOHANSSON, G. (1982)
Uptake via the blood and elimination of 10 organic solvents following
epicutaneous exposure of anesthetized guinea-pigs. Toxicol. appl.
Pharmacol., 63: 181-187.
JONES, R.D. & WINTER, D.P. (1983) Two case reports of deaths on
industrial premises attributed to 1,1,1-trichloroethane. Arch.
environ. Health, 38: 59-61.
KADA, T. (1981) DNA-damaging activity of 42 coded compounds in the
rec-assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 175-182 (Progress in Mutation Research, Vol. 1).
KAJINO, M. & YAGI, M. (1980) Formation of trihalomethanes during
chlorination and determination of halogenated hydrocarbons in drinking
water. In: Hydrocarbons and halogenated hydrocarbons in the aquatic
environment, New York, London, Plenum Press.
KARLSSON, J.-E., ROSENGREN, L.E., KJELLSTRAND, P., & HAGILD K.G.
(1987) Effect of low-dose inhalation of three chlorinated aliphatic
organic solvents on DNA in gerbil brain. Scand. J. Work Environ.
Health, 13: 453-458.
KHALIL, M.A. & RASMUSSEN, R.A. (1981) Decline in the atmospheric
accumulation rates of CCl3F(F-11), CCl2F2(F-12) and CH3CCl3.
J. Air Pollut. Control Assoc., 31: 1274-1275.
KHALIL, M.A. & RASMUSSEN, R.A. (1984) Methylchloroform: Global
distribution, seasonal cycles and anthropogenic chlorine. Chemosphere,
13: 789-800.
KING, G.S., SMIALEK, J.E., & TROUTMAN, W.G. (1985) Sudden death in
adolescent resulting from the inhalation of typewriter correction
fluid. J. Am. Med. Assoc., 253: 1604-1606.
KLAASSEN, C.D. & PLAA, G.L. (1966) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in mice.
Toxicol. appl. Pharmacol., 9: 139-151.
KLAASSEN, C.D. & PLAA G.L. (1967) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in dogs.
Toxicol. appl. Pharmacol., 10: 119-131.
KLECKA, G.M., GONSIOR, S.J., & MARKHAM, D.A. (1990) Biological
transformations of 1,1,1-trichloroethane in subsurface soils and
ground water. Environ. Toxicol. Chem., 9: 1437-1451.
KLEINFELD, M. & FEINER, B. (1966) Health hazard associated with work
in confined spaces. J. occup. Med., 8: 358-364.
KOBAYASHI, H., HOBARA, T., HIROTA, H., HIGASHIHARA, E., IWAMOTO, S.,
& SAKAI, T. (1982) Sensitization of dog myocardium to epinephrine by
1,1,1-trichloroethane. Jpn. J. ind. Health, 24: 450-454.
KOBAYASHI, H., HOBARA, T., HIROTA, H., & SAKAI, T. (1983) Neural
control of blood pressure following 1,1,1-trichloroethane inhalation:
A role of sympathetic nervous system. Arch. environ. Health, 38:
93-98.
KOBAYASHI, H., HOBARA, T., SATOH, T., & SAKAI, T. (1986) Respiratory
disorders following 1,1,1-trichloroethane inhalation: A role of reflex
mechanism arising from lungs. Arch. environ. Health, 41: 149-154.
KOBAYASHI, H., HOBARA, T., KAWAMOTO, T., & SAKAI, T. (1987) Effect of
1,1,1-trichloroethane inhalation on heart rate and its mechanism: A
role of autonomic nervous system. Arch. environ. Health, 42: 140-143.
KONEMANN, H. (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part I. Relationship for 50 industrial
pollutants. Toxicology, 19: 209-221.
KRAMER, C.G., OTT, M.G., FULKERSON, J.E., & HICKS, N. (1978) Health of
workers exposed to 1,1,1-trichloroethane: A matched-pair study. Arch.
environ. Health, 33: 331-342.
KRONELD, R. (1989) Volatile pollutants in the environment and human
tissues. Bull. environ. Contam. Toxicol., 42: 873-877.
KRONEVI, T., WAHLBERG, J.E., & HOLMBERG, B. (1981) Skin pathology
following epicutaneous exposure to seven organic solvents. Int. J.
Tissue React., 3: 21-30.
KUKONGVIRIYAPAN, V., KUKONGVIRIYAPAN, U., & STACEY, N.H. (1990)
Interference with hepatocellular substrate uptake by
1,1,1-trichloroethane and tetrachloroethylene. Toxicol. appl.
Pharmacol., 102: 80-90.
KYRKLUND, T., KJELLSTRAND, P., & HAGLID, K.G. (1988) Effects of
exposure to Freon 11, 1,1,1-trichloroethane or perchloroethylene on
the lipid and fatty-acid composition in rat cerebral cortex. Scand. J.
Work Environ. Health, 14: 91-94.
LANE, R.W., RIDDLE, B.L., & BORZELLECA, J.F. (1982) Effects of
1,2-dichloroethane and 1,1,1-trichloroethane in drinking water on
reproduction and development in mice. Toxicol. appl. Pharmacol., 63:
409-421.
LEBLANC, G.A. (1980) Acute toxicity of priority pollutants to water
flea (Daphnia magna). Bull environ. Contam. Toxicol., 24: 684-691.
LEONG, B.K.J., SCHWETZ, B.A., & GEHRING, P.J. (1975) Embryo- and
fetotoxicity of inhaled trichloroethylene, perchloroethylene,
methylchloroform, and methylene chloride in mice and rats. Toxicol.
appl. Pharmacol., 33: 136 (Abstract 34).
LILLIAN, D., SINGH, H.B., APPLEBY, A., LOBBAN, L., ARNTS, R., GUMPERT,
R., HAGUE, R., TOOMEY, J., KAZASIS, J., ANTELL, M., HANSEN, D., &
SCOTT, B. (1975) Atmospheric fates of halogenated compounds. Environ.
Sci. Technol., 9: 1043-1048.
LINDBOHM, M-L., TASKINEN, H., SALLMEN, M., & HEMMINKI, K. (1990)
Spontaneous abortions among women exposed to organic solvents. Am. J.
ind. Med., 17: 449-463.
LOPRIENO, N., BARATE, R., ROSSI, A.M., FUMERO, S., MERIGGI, G.,
MONDINO, A., & SILVEST S. (1979) In vivo mutagenicity studies with
trichloroethylene and other solvents, Ivrea, Italy, Istituto di
Ricerche Biomedicale
LUNDBERG, I., EKDAHL, M., KRONEVI, T., LIDUMS, V., & LUNDBERG, S.
(1986) Relative hepatotoxicity of some industrial solvents after
intraperitoneal injection or inhalation exposure in rats. Environ.
Res., 40: 411-420.
MABEY, W., BARICH, V., & MILL, T. (1983) Extended abstracts of the
186th Meeting of the American Chemical Society, Washington, DC,
American Chemical Society, Division of Environmental Chemistry, Vol.
23, pp. 359-361.
MCCARTHY, T.B. & JONES, R.D. (1983) Industrial gassing poisonings due
to trichloroethylene, perchlorethylene, and 1,1,1-trichloroethane,
1961-80. Br. J. ind. Med., 40: 450-455.
MCCONNELL, G., FERGUSON, D.M., & PEARSON, C.R. (1975) Chlorinated
hydrocarbons and the environment. Endeavour, 34: 13-18.
MCCONNELL, J.C. & SCHIFF, H.I. (1978) Methyl chloroform: Impact on
stratospheric ozone. Science, 199: 174-177.
MCDONALD, D.J. (1981) Salmonella/microsome tests on 42 coded
chemicals. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 285-297 (Progress in Mutation Research, Vol. 1).
MCEWEN, J.D. & VERNOT, E.H. (1974) The biological effect of continuous
inhalation exposure of 1,1,1-trichloroethane (methyl chloroform) on
animals. In: Toxic hazard research unit: Annual technical report 1974,
Ohio, Wright-Patterson Air-Force Base, Aerospace Medical Research
Laboratory, pp. 81-90 (AMRL-TR-74-78).
MACKAY, C.J., CAMPBELL, L., SAMUEL, A.M., ALDERMAN, K.J., IDZIKOWSKI,
C., WILSON, H.K., & COMPERTZ, D. (1987) Behavioral changes during
exposure to 1,1,1-trichloroethane: time-course and relationship to
blood solvents levels. Am. J. ind. Med., 11: 223-239.
MCLEOD, A.A., MARJOT, A., MONAGHAN, M.J., HUGH-JONES, P., & JACKSON,
G. (1987) Chronic cardiac toxicity after inhalation of
1,1,1-trichloroethane. Br. med. J., 214: 727-729.
MCNUTT, N.S., AMSTER, R.L., MCCONNELL, E.F., & MORRIS, F (1975)
Hepatic lesions in mice after continuous inhalation exposure to
1,1,1-trichloroethane. Lab. Invest., 5: 642-654.
MAGARA, Y. & FURUICHI, T. (1986) Environmental pollution by
trichloroethylene and tetrachloroethylene: a nationwide survey. In:
Chambers, P.L., Gehring, P., & Sakai, F., ed. New concepts and
developments in toxicology, Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp. 231-243.
MALTONI, C., COTTI, G., & PATELLA, V. (1986) Results of long-term
carcinogenicity bioassays on Sprague-Dawley rats of methyl chloroform,
administered by ingestion. Acta oncol., 7: 101-117.
MARONI, M.C., BULGGHERONI, C., CASSITTO, M.G., MERLUZZI, F., GILIOLO,
R., & FOA, V. (1977) A clinical, neurophysiological and behavioral
study of female workers exposed to 1,1,1-trichloroethane. Scand. J.
Work Environ. Health, 3: 16-22.
MARJOT, R. & MCLEOD, A.A. (1989) Chronic non-neurological toxicity
from volatile substance abuse. Hum. Toxicol., 8: 301-306.
MARTIN, C.N. & MCDERMID, A.C. (1981) Testing of 42 coded compounds for
their ability to induce unscheduled DNA repair synthesis in HeLa
cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term
tests for carcinogens. Report of the international collaborative
programme, New York, Amsterdam, Oxford, Elsevier/North Holland, pp.
533-537 (Progress in Mutation Research, Vol. 1).
MIDGLEY, P.M. (1989) The production and release to the atmosphere of
1,1,1-trichloroethane (methyl chloroform). Atmos. Environ., 23:
2663-2665.
MILLER, L.J. & UHLER, A.D. (1988) Volatile halocarbons in butter:
Elevated tetrachloroethylene levels in samples obtained in close
proximity to dry-cleaning establishments. Bull. environ. Contam.
Toxicol., 41: 469-474.
MITI (1990) The yearbook of chemical trade and industries statistics,
Tokyo, Ministry of International Trade and Industry.
MITOMA, C., STEEGER, T., JACKSON, S.E., WHEELER, K.P., ROGERS, J.H.,
& MILMAN, H.A. (1985) Metabolic disposition study of chlorinated
hydrocarbons in rats and mice. Drug chem. Toxicol., 8(3): 183-194.
MONSTER, A.C. (1979) Differences in uptake, elimination and metabolism
in exposure to trichloroethylene, 1,1,1-trichloroethane and
tetrachloroethylene. Int. Arch. occup. environ. Health, 42: 311-317.
MONSTER, A.C. (1986) Biological monitoring of chlorinated hydrocarbon
solvents. J. occup. Med., 28: 583-588.
MONSTER, A.C. & HOUTKOPPER, J.M. (1979) Estimation of individual
uptake of trichloroethylene, 1,1,1-trichloroethane, and
tetrachloroethylene from biological parameters. Int. Arch. occup.
environ. Health, 42: 319-323.
MONSTER, A..C., BOERSMA, G., & STEENWEG, H. (1979) Kinetics of
1,1,1-trichloroethane in volunteers: influence of exposure
concentration and work load. Int. Arch. occup. environ. Health, 42:
293-301.
MOSER, V.C. & BALSTER, R.L. (1985) Acute motor and lethal effects of
inhaled toluene, 1,1,1-trichloroethane, halothane, and ethanol in
mice: effects of exposure duration. Toxicol. appl. Pharmacol., 77:
285-291.
MULLIN, L.S. & KRIVANEK, N.D. (1982) Comparison of unconditioned
reflex and conditioned avoidance tests in rats exposed by inhalation
to carbon monoxide, 1,1,1-trichloroethane, toluene or ethanol.
Neurotoxicology, 3: 126-137.
MYHR, B.C. & CASPARY, W.J. (1988) Evaluation of the L5178Y mouse
lymphoma cell mutagenesis assay: Intralaboratory results for
sixty-three coded chemicals tested at Litton Bionetics, Inc. Environ.
mol. Mutagen., 13: 103-194.
NAGAO, M. & TAKAHASHI, Y. (1981) Mutagenic activity of 42 coded
compounds in the salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 302-313 (Progress in Mutation
Research, Vol. 1).
NAKAAKI, K., FUKABORI, S., & TADA, O. (1980) An experimental study on
percutaneous absorption of some organic solvents. J. Sci. Labour, 56:
1-9.
NCI (1977) Bioassay of 1,1,1-trichloroethane for possible
carcinogenicity, Bethesda, Maryland, National Cancer Institute
(Carcinogenesis Technical Report Series No. 3).
NEELY, W.B. (1982) Organizing data for environmental studies. Environ.
Toxicol. Chem., 1: 259-266.
NESTMANN, E.R., LEE, E.G.H., MATULA, T.I. DOUGLAS, G.R., & MUELLER,
J.C. (1980) Mutagenicity of constituents identified in pulp and paper
mill effluents using the Salmonella/mammalian-microsome assay.
Mutat. Res., 79: 203-212.
NEUHAUSER, E.F., LOEHR, R.C., & MALECKI, M.R. (1986) Contact and
artificial soil tests using earthworms to evaluate the impact of
wastes in soil. In: Petros, J.K., Lacy, W.J., & Conway, R.A., ed.
Hazardous and industrial solid waste testing: Fourth Symposium.
Philadelphia, American Society for Testing and Materials, pp. 192-203
(ASTM STP 886).
NIEF, L. & MERCIER, J. (1981) Skin pathology following epicutaneous
exposure to seven organic solvents. Int. J. Tissue React., 111: 21-30.
NILSSON, K.B. (1986a) Effects of 1,1,1-trichloroethane on the cGMP
metabolism in mouse brain. Acta pharmacol. toxicol., 58: 318-326.
NILSSON, K.B. (1986b) Actions of 1,1,1-trichloroethane on the cAMP
metabolism in mouse brain. Acta pharmacol. toxicol., 59: 362-369.
NILSSON, K.B. (1987) Effects of 1,1,1-trichloroethane on synaptosomal
calcium accumulation in mouse brain. Pharmacol. Toxicol., 61: 215-219.
NIOSH (1976) Criteria for a recommended standard: occupational
exposure to 1,1,1-trichloroethane (methyl chloroform), Cincinnati,
Ohio, National Institute for Occupational Safety and Health
(Publication DHEW (NIOSH) No. 76-184).
NIOSH (1977) NIOSH manual of analytical methods. Part II: Standards
completion program validated methods (Method 5328), 2nd ed.,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, pp. 5328/1-5328/9 (DHEW (NIOSH) Publication No. 77-157-B).
NIOSH (1979) Occupational exposure to 1,1,1-trichloroethane,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health.
NIOSH (1983) National occupational exposure survey, 1981-1983,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, Division of Surveillance, Hazard Evaluations and Field
Studies, Surveillance Branch, Hazard Section (Unpublished data).
NOLAN, R.J., FRESHOUR, N.L., RICH, D.L., MCCARTY, L.P., & SAUNDERS,
J.H. (1984) Kinetics and metabolism of inhaled methyl chloroform
(1,1,1-trichloroethane) in male volunteers. Fundam. appl. Toxicol., 4:
654-662.
NORTHFIELD, R.R. (1981) Avoidable deaths due to acute exposure to
1,1,1-trichloroethane. J. Soc. Occup. Med., 31: 164-166.
NRC (1976) Halocarbons. Effects on stratospheric ozone, Washington,
DC, National Research Council, Panel on Atmospheric Chemistry. OTSON,
R. (1987) Purgeable organics in Great Lakes raw and treated water.
Int. J. environ. anal. Chem., 31: 41-53.
OTSON, R. & CHAN, C. (1987) Sample handling and analysis for 51
volatile organics by an adapted purge and trap GC-MS technique. Int.
J. environ. anal. Chem., 30: 275-287.
OTSON, R., WILLIAMS, D.T., & BOTHWELL, P.D. (1984) Fabric protectors.
Part II - Propane, 1,1,1-trichloroethane and petroleum distillates in
air after application of fabric protectors. Am. Ind. Hyg. Assoc. J.,
45: 28-33.
PARSONS, F. & LAGE, G.B. (1985) Chlorinated organics in simulated
groundwater environments. J. Am. Water Works Assoc., 77: 52-59.
PARSONS, F., LAGE, G.B., & RICE, R. (1985) Biotransformation of
chlorinated organic solvents in static microcosms. Environ. Toxicol.
Chem., 4: 739-742.
PEARSON, C.R. (1982) C1 and C2 halocarbons. In: Hutzinger, O., ed.
The handbook of environmental chemistry, Berlin, Heidelberg, New York,
Springer-Verlag, Vol. 3, Part B, pp. 69-88.
PEARSON, C.R. & MCCONNELL, G. (1975) Chlorinated C1 and C2
hydrocarbons in the marine environment. Proc. R. Soc. Lond., B189:
305-332.
PEKARI, K. & AITIO, A. (1985) Method 26. Determination of
1,1,1-trichloroethane in blood. In: Fishbein, L. & O'Neil, I.K., ed.
Environmental carcinogens selected methods of analysis. Volume 7 -
Some volatile halogenated hydrocarbons, Lyon, International Agency for
Research on Cancer, pp. 445-450 (IARC Scientific Publications No. 68).
PELLIZARI, E.D., HARTWELL, T.D., HARRIS, B.S.H., WADDELL, R.D.,
WHITAKER, D.A., & ERICKSON, M.D. (1982) Purgeable organic compounds in
mothers' milk. Bull. environ. Contam. Toxicol., 28: 322-328.
PEREIRA, W.E. & HUGHES, B.A. (1980) Determination of selected volatile
organic priority pollutants in water by computerized gas
chromatography - quadruple mass spectrometry. J. Am. Water Assoc., 72:
220-230.
PERRY, P.E. & THOMSON, E.J. (1981) Evaluation of the sister-chromatid
exchange method in mammalian cells as a screening system for
carcinogens. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 560-569 (Progress in Mutation Research, Vol. 1).
PFANNHAUSER, W., GOMBOS, J., & THALLER, A. (1988) [Halogenated
aliphatic hydrocarbons in food: Investigations on enrichment and
content in fat containing food.] Ernährung, 12: 611-615 (in German).
PIET, G.J., SLINGERLAND, P., DE GRUNT, F.E., HEUVEL, V.D., & ZOETEMAN,
B.C.J. (1978) Determination of very volatile halogenated organic
compounds in water by means of direct head-space analysis. Anal.
Lett., 11: 437-448.
PRENDERGAST, J.A., JONES, R.A., JENKINS, L.J., & SIEGEL, J. (1967)
Effects on experimental animals of long-term inhalation of
trichloroethylene, carbon tetrachloride, 1,1,1-trichloroethane,
dichlorodifluoromethane, and 1,1-dichloroethylene. Toxicol. appl.
Pharmacol., 10: 270-289.
PRICE, P.J., HASSETT, C.M., & MANSFIELD, J.I. (1978) Transforming
activities of trichloroethylene and proposed industrial alternatives.
14: 290-293.
PRINN, R., CUNNOLD, D., RASMUSSEN, R., SIMMONDS, P., ALYEA, F.,
CRAWFORD, A., FRASER, P., & ROSEN, R. (1987) Atmospheric trends in
methylchloroform and the global average for the hydroxyl radical.
Science, 238: 945-950.
PRODI, G., COLLACCI, A., GRILLI, S., LATTANZI, G., MAZZULO, M., &
TURINA P. (1988) Comparison of the covalent binding of various
chloroethanes with nucleic acids. In: Feo, F., ed. Chemical
carcinogenesis (models and mechanisms), New York, Plenum Press, pp.
93-102.
QUAST, J.F., RAMPY, L.W., BALMER, M.F., LEONG, B.D.J., & GEHRING P.J.
(1978) Toxicological and carcinogenic evaluation of a
1,1,1-trichloroethane formulation by chronic inhalation in rats,
Midland, Michigan, Dow Chemical Co.
QUAST, J.F., CALHOUN, L.L., & FRAUSON, L.E. (1988)
1,1,1-Trichloroethane formulation: a chronic inhalation toxicity and
oncogenicity study in Fischer 344 rats and B6C3F1 mice. Fundam. appl.
Toxicol., 11: 611-625.
RAJENDRAN, S. (1990) The toxicity of phosphine, methyl bromide,
1,1,1-trichloroethane and carbon dioxide alone and as mixtures to the
pupae of red flour beetle, Tribolium castaneum, Herbst. Pestic.
Sci., 29: 75-83.
RAMPY, L.W., QUAST, J.F., LEONG, B.K.J., & GEHRING, P.J. (1977)
Results of long-term inhalation studies on rats of
1,1,1-trichloroethane and perchloroethylene formulations. In: Plaa,
G.L. & Duncan, W.A.M., ed. Proceedings of the 1st International
Congress on Toxicology, Toronto, New York, London, Academic Press, p.
27 (Abstract).
RASMUSSEN, R.A. & KHALIL, M.A.K. (1982) Latitudinal distributions of
trace gases in and above the boundary layer. Chemosphere, 11: 117-235.
RASMUSSEN, R.A. & KHALIL, M.A.K. (1983) Natural and anthropogenic
trace gases in and above the boundary layer. Chemosphere, 12: 371-375.
RASMUSSEN, R.A., KHALIL, M.A.K., & DALLAGUE, R.W. (1981) Atmospheric
trace gases in Antarctica. Science, 211: 185-287.
REINHARDT, C.F., MULLIN, L.S., & MAXFIELD, M.E. (1973)
Epinephrine-induced cardiac arrhythmia potential of some common
industrial solvents. J. occup. Med., 15: 953-955.
REITZ, R.H., MCDOUGAL, J.N., HIMMELSTEIN, M.W., NOLAN, R.J., &
SCHUMANN, A.M. (1988) Physiologically based pharmacokinetic modelling
with methylchloroform: Implications for interspecies, high dose/low
dose and dose route extrapolations. Toxicol. appl. Pharmacol., 95:
185-199.
RENNICK, R.B., MALTON, S.D., MOW, G.K., & SEEVERS, M.H. (1949)
Induction of idioventricular rhythms by 1,1,1-trichloroethane and
epinephrine. J. Pharmacol. exp. Ther., 94: 327.
RICHOLD, M. & JONES, E. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluations of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 314-322 (Progress in Mutation
Research, Vol. 1).
RIIHIMAKI, V. & PFAFFLI, P (1978) Percutaneous absorption of solvent
vapor in man. Scand. J. Work Environ. Health, 4: 73-85.
RIVETT, M.O., LERNER, D.N., & LLOYD, J.W. (1990) Chlorinated solvents
in U.K. aquifers. J. Inst. Water Environ. Manage., 4: 242-250.
ROSENGREN, L.E., AURELL, A., KJELLSTRAND, P., & HAGLID, K.G. (1985)
Astrogliosis in the cerebral cortex of gerbils after long-term
exposure to 1,1,1-trichloroethane. Scand. J. Work Environ. Health, 11:
447-455.
ROSENKRANZ, H.S., HYMAN, J., & LEIFER, Z. (1981) DNA polymerase
deficient assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 210-218 (Progress in Mutation Research, Vol. 1).
ROWLAND, I. & SEVERN, B. (1981) Mutagenicity of carcinogens and
noncarcinogens in the Salmonella/microsome test. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 323-332 (Progress in
Mutation Research, Vol. 1).
RUSSELL, J.W. & SHADOFF, L.A. (1977) The sampling and determination of
halocarbons in ambient air using concentration on porous polymer. J.
Chromatogr., 134: 375-384.
SALAMONE, M.F., HEDDLE, J.A., & KATZ M. (1981) Mutagenic activity of
41 compounds in the in vivo micronucleus assay. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 686-697 (Progress in
Mutation Research, Vol. 1).
SALVINI, M., BINASCHI, S., & RIVA, M. (1971) Evaluation of the
psychophysiological functions in humans exposed to the "threshold
limit value" of 1,1,1-trichloroethane. Br. J. ind. Med., 28: 286-292.
SATO, A., NAKAJIMA, T., & KOYAMA, Y. (1980) Effects of chronic ethanol
consumption on hepatic metabolism of aromatic and chlorinated
hydrocarbons in rats. Br. J. ind. Med., 37: 382-386.
SAVOLAINEN, H., PFAFFLI, P., TENGEN, M., & VAINIO, H. (1977)
Trichloroethylene and 1,1,1-trichloroethane: effects on brain and
liver activity after five days intermittent inhalation. Arch.
Toxicol., 38: 229-237.
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1981)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Int. Arch. occup. environ. Health, 49: 89-98.
SAVOLAINEN, K., RIIHIMÄKI, V., & LAINE A. (1982a) Biphasic effects of
inhaled solvents on human equilibrium. Acta pharmacol. toxicol., 51:
237-242
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1982b)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Arch. Toxicol., Suppl. 5: 96-99.
SCHUMANN, A.M., FOX, T.R., & WATANABE, P.G. (1982) [14C]-Methyl
chloroform (1,1,1-trichloroethane): Pharmacokinetics in rats and mice
following inhalation exposure. Toxicol. appl. Pharmacol., 62: 390-401.
SEKI, Y., URASHIMA, Y., AIKAWA, H., MATSUMURA, H., ICHIKAWA, Y.,
HIRATSUKA, F., YOSHIOKA, Y., SHIMBO, S., & IKEDA, M. (1975)
Trichloro-compounds in the urine of humans exposed to methyl
chloroform at sub-threshold levels. Int. Arch. Arbeitsmed., 34: 39-49.
SHIMADA, T., SWANSON, A.F., LEBER, P., & WILLIAMS, G.M. (1985)
Activities of chlorinated ethane and ethylene compounds in the
Salmonella/rat microsome mutagenesis and rat hepatocyte/DNA repair
assays under vapor phase exposure conditions. Cell Biol. Toxicol., 1:
159-179.
SHMUTER, L.M. (1977) [The effect of chronic exposure to low
concentrations of chlorinated ethane series hydrocarbons on specific
and non-specific immunological response in animals under experimental
conditions.] Gig. Tr. prof. Zabol., 8: 38-43 (in Russian).
SIEGEL, J., JONES, R.A., COON, R.A., & LYON J.P. (1971) Effects on
experimental animals of acute, repeated and continuous inhalation
exposures to dichloroacetylene mixtures. Toxicol. appl. Pharmacol.,
18: 168-174.
SIMMON, V.F. & SHEPHERD, G.F. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 333-342 (Progress in Mutation
Research, Vol. 1).
SIMMON, V.F., KAUHANEN, K., & TARDIFF, R.G. (1977) Mutagenic activity
of chemicals identified in drinking water. In: Scott, D., Bridges,
B.A., & Sobels, F.H., ed. Progress in genetic toxicology, Amsterdam,
Elsevier/North Holland, pp. 249-258.
SIMMONDS, P.G., KERRIN, S.L., LOVELOCK, J.E., & SHAIR, F.H. (1974)
Distribution of atmospheric halocarbons in the air over the Los
Angeles basin. Atmos. Environ., 8: 209-216.
SINGH, H.B., SALAS, L.J., & STILES, R.E. (1982) Distribution of
selected gaseous organic mutagens and suspect carcinogens in ambient
air. Environ. Sci. Technol., 16: 982-880.
SNOW, L.P., MCNAIR, B.C., & CASTRO B.C. (1979) Mutagenesis testing of
1,1,1-trichloroethane in Salmonella strains TA100 and TA98, Research
Triangle Park, North Carolina, Northrop Services Inc. (Unpublished
report prepared for the US Environmental Protection Agency).
STAHL, C.J. FATTEH, A.V., & DOMINGUEZ, A.M. (1969) Trichloroethane
poisoning: observations on the pathology and toxicology in six fatal
cases. J. Forensic. Sci., 14: 393-397.
STEWART, R.D. & ANDREWS J.T. (1966) Acute intoxication with methyl
chloroform. J. Am. Med. Assoc., 195: 904-906.
STEWART, R.D. (1968) The toxicology of 1,1,1-trichloroethane. Ann.
occup. Hyg., 11: 71-79.
STEWART, R.D. (1971) Methyl chloroform intoxication. J. Am. Med.
Assoc., 215: 1789-1792.
STEWART, R.D. & DODD, H.D. (1964) Absorption of carbon tetrachloride,
trichloroethylene, tetrachloroethylene, methylene chloride and
1,1,1-trichloroethane through the human skin. Am. Ind. Hyg. Assoc. J.,
25: 439-446.
STEWART, R.D., GAY, H.H., ERLEY, D.S., HAKE, C.L., & SCHAFFER, A.W.
(1961) Human exposure to 1,1,1-trichloroethane vapour: relationship of
expired air and blood concentrations. Am. Ind. Hyg. Assoc. J., 22:
252-262.
STEWART, R.D., GAY, H.H., SCHAFFER, A.W., ERLEY, D.S., & ROWE, V.K.
(1969) Experimental human exposure to methyl chloroform vapor. Arch.
environ. Health, 19: 467-472.
STYLES, J.A. (1981) Activity of 42 coded compounds in the BHK-21 cell
transformation test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 638-646 (Progress in Mutation Research, Vol. 1).
THIELE, D.L., EIGENBRODT, E.H., & WARE, A.J. (1982) Cirrhosis after
repeated trichloroethylene and 1,1,1-trichloroethane exposure.
Gastroenterology, 83: 926-929.
THOMPSON, R.S. & CARMICHAEL, N.G. (1989) 1,1,1-Trichloroethane:
Medium-term toxicity to carp, daphnids, and higher plants. Ecotoxicol.
environ. Saf., 17: 172-182.
TOFTGAARD, R., NILSEN, O.G., & GUSTAFSSON, J.A. (1981) Changes in rat
liver microsomal cytochrome P-450 and enzymatic activities after the
inhalation of n-hexane, xylene, methyl ethyl ketone and
methylchloroform for four weeks. Scand. J. Work Environ. Health, 7:
31-37.
TOPHAM, J.C. (1980) Do induced sperm-head abnormalities in mice
specifically identify mammalian mutagens rather than carcinogens?
Mutat. Res., 74: 379-387.
TORKELSON, T.R., OYEN, F., MCCOLLISTER, D.D., & ROWE V.K. (1958)
Toxicity of 1,1,1-trichloroethane as determined on laboratory animals
and human subjects. Am. Ind. Hyg. Assoc. J., 19: 353-362.
TORSLOV, J. (1988) Hazard assessment of 1,1,1-trichloroethane,
Copenhagen, Denmark, Ministry of the Environment, National Agency of
Environmental Protection, 67 pp (Miljoproject, No. 10).
TRAVIS, C.C., HATTEMER-FREY, H.A., & ARMS, A.D. (1988) Relationship
between dietary intake of organic chemicals and their concentrations
in human adipose tissue and breast milk. Arch. environ. Contam.
Toxicol., 17: 473-478.
TSUCHIMOTO, T. & MATTER, B.E. (1981) Activity of coded compounds in
the micronucleus test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 705-711 (Progress in Mutation Research, Vol. 1). TSURUTA,
H. (1975) Percutaneous absorption of organic solvents. I. Comparative
study of the in vivo percutaneous absorption of chlorinated solvents
in mice. Ind. Health, 13: 227-236.
TU, A.S., MURRAY, T.A., HATCH, K.M., SIVAK, A., & MILMAN, H.A. (1985)
In vitro transformation of balb/c-3T3 cells by chlorinated ethanes
and ethylenes. Cancer Lett., 28: 85-92.
TWEATS, D.J. (1981) Activity of 42 coded compounds in a differential
killing test using Escherichia coli strains WP2, WP67 (uvrA polA),
and CM871 (uvrA lexA recA). In: de Serres, F.J. & Ashby, J., ed.
Evaluation of short-term tests for carcinogens. Report of the
international collaborative programme, New York, Amsterdam, London,
Elsevier/North Holland, pp. 199-209 (Progress in Mutation Research,
Vol. 1).
UHLER, A.D. & DIACHENKO, G.W. (1987) Volatile halocarbon compounds in
process water and processed foods. Bull. environ. Contam. Toxicol.,
39: 601-607.
UNEP (1989) Technical progress on protecting the ozone layer. Report
of the Technology Review Panel, Nairobi, United Nations Environment
Programme, 103 pp.
URANO, K. (1985) Usage and pollution control of chlorinated organic
compounds. Jpn. J. Water Pollut. Res., 8: 269-273.
URANO, K., KAWAMOTO, K., ABE, Y., & OTAKE, M. (1988) Chlorinated
organic compounds in urban air in Japan. Sci. total Environ., 74:
121-131.
US EPA (1977) Environmental monitoring near industrial areas -
Methylchloroform, Washington, DC, US Environmental Protection Agency
(EPA-560/6-77-025).
US EPA (1980a) Proceedings of the Conference on Methyl Chloroform and
Other Halocarbon Pollutants, Washington, DC, US Environmental
Protection Agency (EPA-600/9-80-003).
US EPA (1980b) Ambient water quality criteria for chlorinated ethanes,
Washington, DC, US Environmental Protection Agency (EPA-440/5-80-029).
US EPA (1984) Health assessment document for 1,1,1-trichloroethane
(methyl chloroform), Washington, DC, US Environmental Protection
Agency (EPA-600/8-82-003F).
VAN BEEK, L. (1990) Investigation of a possibility to reduce the
number of rabbits in skin irritation tests; experiments with
dichloromethane, trichloromethylene, tetrachloroethylene and
1,1,1-trichloroethane (Project No. B 86-0927) Apeldoorn, The
Netherlands, Organization for Applied Scientific Research (TNO Report
No. V 89.265).
VEITH, G.D., MACEK, K.J. PETROCELLI, S.R., & CARROLL, J. (1980) An
evaluation of using partition coefficients and water solubility to
estimate bioconcentration factors for organic chemicals in fish. In:
Eaton, J.G., Parrish, P., & Hendricks, A.C., ed. Aquatic toxicology.
Proceedings of the Third Annual Symposium on Aquatic Toxicology,
Philadelphia, American Society for Testing and Materials, pp. 116-129
(ASTM STP No. 707).
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., WHITMORE, R.
SPARACINO, C., & ZELON, H. (1986) Total exposure assessment
methodology (TEAM) study: personal exposures, indoor-outdoor
relationships, and breath levels of volatile organic compounds in New
Jersey. Environ. Int., 12: 369-387.
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., SPARACINO, C.,
WHITMORE, R., SHELDON, L., ZELON, H., & PERRITT, R. (1987) The TEAM
study: Personal exposure to substances in air, drinking water, and
breath of 400 residents of New Jersey, North Carolina and North
Dakota. Environ. Res., 43: 290-307.
WALRAEVENS, R., TROUILLET, P., & DEVOS, A. (1974) Basic elimination of
HCI from chlorinated ethanes. Int. J. chem. Kinet., VI: 777-786.
WEAST, R.C. (1986) CRC handbook of chemistry and physics, 67th ed,
Boca Raton, Florida, CRC Press.
WESTERBERG, E. & LARSSON, L. (1982) Use of automated head-space gas
chromatography for determination of 1,1,1-trichloroethane in rat blood
and brain tissue. Int. J. environ. anal. Chem., 12: 233-239.
WILLIAMS, G.M. (1983) DNA repair tests of 11 chlorinated hydrocarbon
analogs, Washington, DC, US Environmental Protection Agency (Draft
Final Report TR-507-18).
WILSON, B.H. & WHITE, M.V. (1986) A fixed-film bioreactor to treat
trichloroethylene-laden waters from interdiction wells. In:
Proceedings of the 1986 National Symposium and Exposition on Aquifer
Restoration and Ground Water Monitoring, Columbus, Ohio, 19-22 May,
Dublin, Ohio, National Well Water Association, pp. 425-436.
WILSON, J.T., MCNABB, J.F., WILSON, B.H., & NOONAN, M.J. (1983)
Biotransformation of selected organic pollutants in ground water. Dev.
ind. Microbiol., 24: 125-233.
WHO (in preparation) Health and Safety Guide for
1,1,1-trichloroethane, Geneva, World Health Organization.
WINDHOLZ, M. ed. (1983) The Merck index: An encyclopedia of chemicals,
drugs and biologicals, 10th ed., Rahway, New Jersey, Merck and Co.,
Inc.
WMO (1982) Global ozone research and monitoring project: Report No.
11. The stratosphere 1981: Theory and measurements, Geneva,
Switzerland, World Meteorological Organization.
WOOLVERTON, W.L. & BALSTER, R.L. (1981) Behavioral and lethal effects
of combinations of oral ethanol and inhaled 1,1,1-trichloroethane in
mice. Toxicol. appl. Pharmacol., 59: 1-7.
WRIGHT, M.F. & STROBL, D.J. (1984) 1,1,1-Trichloroethane cardiac
toxicity: Report of a case. J. Am. Osteopath. Assoc., 84: 285-288.
WRIGHTSON, I. & SANTON, R.C. (1988) An explosion involving
1,1,1-trichloroethane. Loss Prev. Bull., 083: 21-23.
YORK, R.G., SOWRY, B.M., HASTINGS, L., & MANSON, J.M. (1982)
Evaluation of teratogenicity and neurotoxicity with maternal
inhalation exposure to methylchloroform. J. Toxicol. environ. Health,
9: 251-266.
YUNG, Y.L., MCELROY, M.B., & WOFSY, S.C. (1975) Atmospheric
halocarbons: A discussion with emphasis on chloroform. Geophys. Res.
Lett., 2: 397-399.
RESUME
Le 1,1,1-trichloréthane est un hydrocarbure chloré produit par
chloration du chlorure de vinyle ou du chlorure de vinylidène. En
1988, la production mondiale était d'environ 680 000 tonnes. C'est un
liquide incolore et volatil à l'odeur caractéristique dont la vapeur
est plus dense que l'air. On l'utilise essentiellement pour le
dégraissage des surfaces métalliques et comme solvant dans un grand
nombre de produits industriels et de produits de consom-mation,
notamment des adhésifs, des détachants et des bombes aérosols. Il
sert également d'intermédiaire en synthèse chimique. Le
trichloréthane technique contient généralement 3 à 8% de stabilisants
destinés à empêcher sa décomposition et la formation d'acide
chlorhydrique; les parties métalliques sont ainsi protégées de la
corrosion. Il est ininflammable dans les conditions normales mais la
vapeur s'enflamme à haute température et, au cours du soudage, elle
peut se décomposer en libérant du phosgène, un gaz toxique. Des
réactions extrêmement violentes peuvent se produire en cas de contact
avec de l'aluminium, du magnésium ou leurs alliages.
Le 1,1,1-trichloréthane pénètre facilement dans l'environne-ment.
Comme il séjourne longtemps dans la troposphère (environ six ans) et
qu'il n'est que lentement biodégradable, on le retrouve maintenant
partout dans l'environnement, même à grande distance des zones
industrielles. A proximité d'installations industrielles où ce
composé est produit ou manipulé, on a trouvé du 1,1,1-trichloréthane
à des concentrations atteignant 86 µg/m3 (16 000 ppm, p/p) dans des
échantillons d'air.
Le trichloréthane se déplace dans le sol et parvient jusqu'aux
eaux souterraines. On en a trouvé à des concentrations atteignant
1600 µg/litre dans les eaux souterraines ou les eaux de surface. Ce
phénomène peut être à l'origine de la contamination des réserves d'eau
potable.
On estime que 15% des émissions annuelles de
1,1,1-trichloré-thane sont entraînées dans la stratosphère où elles
peuvent endommager la couche d'ozone par libération d'atomes de
chlore.
Des essais biologiques sur des crustacés et des poissons à des
concentrations supérieures à 7 mg/litre ont révélé des effets toxiques
aigus. On possède quelques données selon lesquelles le composé ne
s'accumulerait que faiblement chez les organismes aquatiques. En
raison du caractère limité des données disponibles, il est difficile
d'évaluer les effets qu'il peut avoir sur les organismes terrestres.
L'exposition humaine au 1,1,1-trichloréthane est due
principalement à l'inhalation de ce composé qui est ensuite rapidement
résorbé par l'organisme. Il peut y avoir également exposition par
contact cutané ou ingestion. Le trichloréthane se répartit largement
dans les tissus de l'organisme et il traverse la barrière
hématoencéphalique et placentaire. On le retrouve également dans le
lait humain mais il ne semble pas qu'il s'accumule dans les tissus.
Sa principale voie d'élimination est l'exhalation du composé non
métabolisé.
La toxicité aiguë et chronique du 1,1,1-trichloréthane est
relativement faible mais en cas de forte exposition, le risque
d'intoxication est réel. Cela peut se produire en cas d'exposition
professionnelle, d'accidents ou encore de toxicomanie aux solvants.
Comme il s'agit d'un solvant volatil dont la vapeur est beaucoup plus
dense que l'air, il peut se trouver à des concentrations
inhabituellement fortes et dangereuses dans des espaces confinés comme
les réservoirs "vides". Ce genre de situation a donné lieu à
plusieurs intoxications mortelles ou qui auraient pu l'être sur les
lieux de travail ou ailleurs.
Chez l'homme, l'effet critique s'exerce au niveau du système
nerveux central. On observe des effets qui vont de légers troubles du
comportement (accompagnés d'une légère irritation occulaire) à la dose
de 1,9 g/m3 (350 ppm), jusqu'à l'inconscience et à l'arrêt
respiratoire lorsque la concentration est élevée. Dans certains cas,
des anomalies cardiaques mortelles peuvent se produire. Le
trichloréthane est moins toxique pour le foie que la plupart des
autres solvants organiques chlorés. La dose sans effet observable
pour l'homme se situe autour de 1,35 g/m3 (250 ppm).
Aucune étude satisfaisante n'a été publiée sur les effets
cancérogènes de ce solvant chez l'homme. Toutefois une étude par
inhalation de longue durée sur des rats et des souris exposés à 8,1
g/m3 (1500 ppm) n'a pas révélé le moindre effet cancérogène. Le
1,1,1-trichloréthane ne présente pas d'activité génotoxique
significative.
Chez des rats et des lapins exposés au trichloréthane à des
concentrations qui étaient toxiques pour les femelles gestantes, on
n'a pas noté d'effets tératogènes, malgré la présence d'effets
toxiques sur le développement. Les données épidémiologiques relatives
à ces effets sur la reproduction sont trop limitées pour permettre
d'en tirer une conclusion.
RESUMEN
El 1,1,1-tricloroetano es un hidrocarburo clorado que se fabrica
a partir del cloruro de vinilo o del cloruro de vinilideno por
cloración. En 1988 la producción mundial fue de unas 680 000
toneladas. Es un líquido volátil e incoloro con un olor
característico, y su vapor es más denso que el aire. Se utiliza
principalmente en el desengrasado de metales y como disolvente en
muchos productos industriales y de consumo, como adhesivos,
quitamanchas y envases de aerosoles. Es también un producto químico
intermediario. El tricloroetano de calidad técnica suele contener del
3% al 8% de estabilizadores que impiden la degradación y la formación
de ácido clorhídrico a fin de proteger las partes metálicas de la
corrosión. En condiciones normales no es inflamable, pero su vapor
arde a altas temperaturas y, durante las operaciones de soldadura, su
degradación produce fosgeno, un gas tóxico. Cuando entra en contacto
con el aluminio, el magnesio y sus aleaciones puede provocar
reacciones muy violentas.
El 1,1,1-tricloroetano llega fácilmente al medio ambiente. Su
prolongada permanencia en la troposfera (unos seis años) y baja
biodegradabilidad son razón de su actual omnipresencia en el medio
ambiente, incluso lejos de las zonas industriales. En muestras de
aire de zonas próximas a industrias que lo fabrican o lo manipulan se
han detectado concentraciones de hasta 86 µg/m3 (16 ppb, peso/peso).
El tricloroetano se desplaza en los suelos y alcanza las aguas
subterráneas. Se han encontrado concentraciones de hasta 1600
µg/litro en aguas subterráneas o superficiales. Esta puede ser una
fuente de contaminación para suministros de agua potable.
Se estima que el 15% del volumen anual liberado llega a la
estratosfera, donde libera átomos de cloro que contribuyen a la
destrucción del ozono.
En bioensayos con crustáceos y peces se han observado efectos
tóxicos agudos con concentraciones superiores a 7 mg/litro. Limitada
información limitada disponible parece indicar que la bioacumulación
en los organismos acuáticos es baja. La escasez de datos dificulta la
evaluación de los posibles efectos en los organismos terrestres.
El ser humano está expuesto al 1,1,1-tricloroetano principalmente
por inhalación, y luego el organismo absorbe rápidamente la sustancia.
También se puede dar la exposición por contacto cutáneo o ingestión.
El tricloroetano se distribuye ampliamente por los tejidos y atraviesa
las barreras hematoencefálica y placentaria. Aunque también se ha
encontrado en la leche humana, no parece que se produzca
bioacumulación. La principal vía de eliminación es la exhalación del
compuesto inalterado.
Las toxicidades aguda y crónica del 1,1,1-tricloroetano son
relativamente bajas, pero en condiciones de exposición intensa pueden
producirse efectos tóxicos. Estas condiciones se pueden presentar en
casos de exposición profesional, de abuso del disolvente o de
accidentes. Dado que el disolvente es volátil y el vapor es mucho más
denso que el aire, se pueden producir concentraciones inesperadamente
elevadas y peligrosas en espacios reducidos, como depósitos de
almacenamiento "vacíos". Así, se han producido algunas intoxicaciones
mortales y casi mortales en lugares de trabajo y otros.
El efecto más importante en el ser humano se produce en el
sistema nervioso central. Los efectos observables van desde ligeros
cambios de comportamiento (acompañados de una leve irritación ocular)
con 1,9 g/m3 (350 ppm) hasta la pérdida del conocimiento y el paro
respiratorio con concentraciones más elevadas. Sin embargo, también
se pueden producir anomalías cardíacas fatales. La toxicidad hepática
del tricloroetano es inferior a la de la mayoría de los disolventes
organoclorados. El nivel sin efecto observado (NOEL) para la especie
humana parece ser del orden de 1,35 g/m3 (250 ppm).
No se han publicado estudios adecuados sobre los efectos
carcinogénicos en la especie humana. Sin embargo, en los estudios de
inhalación prolongada en ratas y ratones expuestos a 8,1 g/m3 (1500
ppm) no se obtuvieron pruebas de ningún efecto carcinogénico. El
1,1,1-tricloroetano no tiene un potencial genotóxico importante.
En ratas y conejos se ha observado toxicidad en la fase de
crecimiento, pero no teratogenicidad, con concentraciones que fueron
tóxicas para las madres. Las escasas pruebas epidemiológicas en
relación con sus efectos sobre la reproducción no son concluyentes.
body weight. Both trichloroethanol and trichloroacetic acid were
identified as metabolites in the urine, trichloroethanol being
excreted (as the glucuronide) much faster than trichloroacetic acid
(Eben & Kimberle 1974).
Following a single inhalation exposure of rats to 810 and
8100 mg/m3 (150 and 1500 ppm), only a 2-4 times increase in excreted
metabolites was found between the two doses, suggesting metabolic
saturation. Repeated exposure to 8100 mg/m3 over 16 months did not
change the amount of 1,1,1-trichloroethane metabolites. Similar
results were obtained in mice. In both rats and mice, urine
metabolites accounted for 40-70% of the total amount metabolized.
Overall, mice were found to biotransform approximately 5 times more
1,1,1-trichloroethane (per kg body weight) than rats. In rats and
mice an age-related increase in the amount metabolized was observed in
aged animals as opposed to young adults (Schumann et al., 1982).
6.3.2 Human studies
The average amount of metabolites excreted in the urine of humans
(workers or volunteers) exposed to 1,1,1-trichloroethane in the air
(22-1890 mg/m3, 4-350 ppm) was variable, but was typically between
3 and 7% of the absorbed dose. The ratio of the metabolites
trichloroethanol glucuronide and trichloroacetic acid was about 2 to
1, but this increased with increasing exposure concentration (Seki et
al., 1975; Humbert & Fernandez, 1977; Monster et al., 1979; Nolan et
al., 1984).
6.3.3 Metabolic interactions
Simultaneous exposure to other solvents tends to increase the
retention and decrease the metabolism of 1,1,1-trichloroethane
(Savolainen et al., 1981). 1,1,1-Trichloroethane metabolism was
accelerated in rats pre-treated with ethanol because of the induction
of metabolic enzymes (Sato et al., 1980).
6.4 Elimination
Regardless of the route of administration, the main excretory
route for 1,1,1-trichloroethane is exhalation via the lungs. This may
be explained by the relatively low solubility in blood.
6.4.1 Animal studies
In rats and mice, 55% to 98% of the 1,1,1-trichloroethane was
excreted unchanged in expired air after oral or intraperitoneal
exposure (Hake et al., 1960; Reitz et al., 1988).
Following inhalation exposure, 94-98% of the absorbed
1,1,1-trichloroethane was excreted unchanged in the expired air of
rats during 72 h. In mice, the corresponding figure was 87-97%. The
rate of elimination was somewhat higher in mice; 85% was eliminated
during the first 3 h compared to 65% in rats (Schumann et al., 1982).
A total of about 1-8% of an absorbed dose in rodents is excreted
in the urine (Hake et al., 1960; Schumann et al., 1982).
After exposure to 1,1,1-trichloroethane vapour, the level in
blood plasma decreases rapidly in a diphasic or triphasic manner,
depending on the exposure level. Following exposure to 810 mg/m3
(150 ppm), the half-lives were 10 and 139 min, whereas with 8100
mg/m3 (1500 ppm), the half-lives were 36 and 238 min. In mice the
half-lives were a little shorter (Schumann et al., 1982).
6.4.2 Human studies
Studies with human volunteers show that over 90% of the absorbed
trichloroethane is excreted unchanged in the expired air. Only minor
parts (5-7%) of the absorbed solvent are excreted in the urine (as
trichloroethanol glucuronide and trichloroacetic acid) (Stewart et
al., 1969; Humbert & Fernadez, 1977; Nolan et al., 1984). The main
product was found to be 2,2,2-trichloroethanol glucuronide, the
excretion of which was completed within 8 days. The secondary product
was trichloroacetic acid; its excretion occurred somewhat later and
was completed within 12 days (Humbert & Fernandez, 1977).
The elimination of 1,1,1-trichloroethane, measured as plasma
concentration and concentration in expired air, in six human
volunteers exposed to 191 or 1911 mg/m3 (35 or 350 ppm) for 6 h
could be described by a three-compartment model with estimated
half-lives of 44 min, 5.7 h, and 53 h, respectively. Less than 1%
remained in the body after 9 days (Nolan et al., 1984).
In a study by Nolan et al. (1984), the trichloroethane
concentrations in blood and expired air were proportional to the
exposure concentration after 6 h of exposure and indicated that about
25% of the 1,1,1-trichloroethane inhaled during the exposure was
absorbed.
6.5 Biological monitoring
Several options exist for the biological monitoring of exposure
to 1,1,1-trichloroethane (Monster 1986). These include the
determination of:
Ê the unchanged solvent in blood or alveolar air;
Ê the metabolite trichloroethanol in blood, alveolar air or urine;
Ê the metabolite trichloroacetic acid in blood and urine.
Table 9 indicates the mean concentrations of these parameters as
a result of exposure of subjects for 8 h/day (5 days/week) to a
time-weighted average (TWA) concentration of 270 mg/m3 (50 ppm)
(Monster, 1986).
Table 9. Mean concentrations of 1,1,1-trichloroethane,
trichloroethanol, and trichloroacetic acid in blood,
alveolar air, and urinea
Time after exposure of sampling
Test End of 5-15 min 16 h 64 h
exposure
Blood (mg/litre)
1,1,1-trichloroethane 0.9 0.07
trichloroethanol 0.16 -
trichloroacetic acid 2.3 1.6
Alveolar air (mg/m3)
1,1,1-trichloroethaneb 210 (39) 13 (2.4) 8 (1.5)
trichloroethanol 0.014 0.007 -
Urine (mg/g creatinine)
trichloroacetic acid 4.9 2.5 0.9
trichloroethanol 2.5 1.8 1.5
a From: Monster (1986)
b Values in parentheses are in ppm
Trichloroethane concentrations in blood measured after work on a
Friday seem to be the best single parameter for estimation of the
TWA-week exposure (Monster, 1986). For one-day exposure, the
trichloroethane level in blood and urine is most useful (Monster,
1986).
Droz et al. (1989) suggested a physiological model for describing
variability in the biological monitoring of solvent exposure.
Standard statistical distributions are used to simulate variability in
exposure concentration, physical workload, body fluid, liver function,
and renal clearance. For groups of workers exposed daily, the model
calculates air monitoring indicators and biological monitoring
results, including levels in expired air, blood, and urine. The
calculated results obtained are discussed and compared with measured
data for physiological and toxicokinetic parameters for six solvents
including 1,1,1-trichloroethane and their metabolites. It is
suggested that such mathematical models are applicable for prediction
and management studies.
6.6 Bioaccumulation
Inhalation exposure of rats to 2700 mg/m3 (500 ppm) for 4 days,
6 h per day, led to an accumulation of 1,1,1-trichloroethane in the
fat 17 h after the last exposure. Further exposure on the 5th day
increased brain, liver, lung, and blood levels (Savolainen et al.,
1981).
A study by Travis et al. (1988) indicated that bioaccumulation of
organic chemicals, including 1,1,1-trichloroethane, perchloroethane,
trichloroethylene, and dichloromethane, in human adipose tissues was
positively correlated with their octanol-water partition coefficients.
The information presented, however, was based on data from a human
pharmacokinetic model.
7. EFFECTS ON LABORATORY MAMMALS AND IN VITRO TEST SYSTEMS
Appraisal
1,1,1-Trichloroethane administered by various routes has low
acute toxicity for laboratory animals. The acute toxicity pattern is
characterised by central nervous system depression and respiratory
arrest at lethal levels. 1,1,1-Trichloroethane is a moderate skin
and mild eye irritant.
Short-term inhalation exposure of rats to 4320-5400 mg/m3
(800-1000 ppm) or more produced an increase in liver weight. At
similar concentrations in mice, the liver changes were more marked
and included fatty infiltration and single cell necrosis. Minor
cytoplasmic alterations were seen in mice exposed to 1350 mg/m3
(250 ppm). Studies on Mongolian gerbils using 1,1,1-trichloroethane
concentrations down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several regions of
the brain. The significance of these findings is uncertain. The
no-observed-effect level (NOEL) for rats is about 2700 mg/m3 (500
ppm).
In long-term toxicity studies, 1,1,1-trichloroethane
administration led to reduced weight gain in both rats and mice.
Slight microscopic hepatic effects were seen, but only at the highest
concentration of 8100 mg/m3 (1500 ppm). There was no evidence of
carcinogenic potential in oral and inhalation studies on rats and
mice. One study reported increased incidence of leukaemia but this
study is considered inadequate.
1,1,1-Trichloroethane has been shown to have low genotoxic
potential in a range of in vitro and in vivo studies.
In a multigeneration study on mice, there was no evidence of
adverse reproductive effects (including effects on fertility) caused
by 1,1,1-trichloroethane in drinking-water at concentrations up to
1000 mg/kg body weight. Inhalation studies on female rats and mice
at 4590 mg/m3 (850 ppm) showed no evidence of teratogenic or
fetotoxic effects. However, in rats exposed to 11 340 mg/m3 (2100
ppm), there was some evidence of fetotoxicity.
The data available are inadequate to assess the immunotoxic
potential of 1,1,1-trichloroethane.
Enhanced toxicity of 1,1,1-trichloroethane was observed when
exposure was combined with exposure to ethanol.
7.1 Acute toxicity
1,1,1-Trichloroethane has a very low acute toxicity in laboratory
animals dosed by various routes of administration. Selected LD50
and LC50 values are given in Tables 10 and 11.
The toxicological pattern of acute trichloroethane poisoning is
central nervous system (CNS) depression, eventually culminating in
respiratory arrest or cardiac failure at lethal exposure levels
(Stewart, 1968).
The findings of rapid and shallow breathing in a dog following
inhalation of 0.9% (by volume) 1,1,1-trichloroethane (corresponding to
48 600 mg/m3 or 9000 ppm) may be related to an increase in the
activity of the lung stretch receptor in the vagus nerve (Kobayashi et
al., 1986). A level of 1.3% increased the heart rate but 2.8%
decreased it (Kobayashi et al., 1987).
Non-lethal acute toxicity has also been investigated (see Table
12). In a study on rats, 50% of the experimental group showed loss of
coordination (ataxia) after inhalation of 20 400 mg/m3 (3780 ppm)
for 4 h, and loss of righting-reflex was observed at 45 800 mg/m3
(8480 ppm). Tremors and death were observed at 64 800 mg/m3 (12 000
ppm) within one hour of exposure (Mullin & Krivanek, 1982).
Cardiac sensitization to adrenaline was reported in dogs exposed
to 37 800 mg/m3 (range: 21 600 to 59 400 mg/m3) for 5 min (Clark
& Tinston, 1982). An increase in concentration (range: 37 300 to 373
000 mg/m3) and inhalation period (0.5 to 20 min) was accompanied by
a decrease in the amount of adrenaline that induced arrhythmia
(Kobayashi et al., 1982).
Reinhardt et al. (1973) reported that 1,1,1-trichloroethane
caused cardiac sensitization to adrenaline in dogs at and above the
0.5% (v/v) level. The marked response was associated with ventricular
fibrillation following the challenge dose of adrenaline. At this
level, the solvent was also associated with excitement and struggling
in the animals. Histopathological examination of samples from the
dogs that developed fatal arrhythmias did not show any gross or
microscopic abnormalities.
Table 10. Acute toxicity (LD50) of 1,1,1-trichloroethane in experimental animals
Species Sex Route LD50 (mg/kg Reference
body weight)
Rat male oral 14 300 Torkelson et al. (1958)
Rat female oral 11 000 Torkelson et al. (1958)
Mouse female oral 9700 Torkelson et al. (1958)
Guinea-pig male & female oral 8600 Torkelson et al. (1958)
Rabbit male & female oral 10 500 Torkelson et al. (1958)
Rabbit male & female skin 15 800 Torkelson et al. (1958)
Mouse female intraperitoneal 3700 Gradiski et al. (1974)
Mouse male intraperitoneal 5080 Klaassen & Plaa (1966)
Dog male intraperitoneal 4140 Klaassen & Plaa (1967)
Table 11. Acute toxicity by inhalation (LC50) of 1,1,1-trichloroethane
in experimental animals
Species Sex LC50 Exposure Reference
(g/m3) (ppm) duration
Rat male & female 97.2 18 000 3 h Adams et al. (1950)
Rat male & female 76.9 14 250 7 h Adams et al. (1950)
Rat male 99.4 18 400 4 h Siegel et al. (1971)
Rat male 55.6 10 300 6 h Bonnet et al. (1981)
Rat male & female 205 38 000 15 min Clark & Tinston (1982)
Mouse male 120 22 240 30 min Woolverton & Balster (1981)
Mouse female 72.4 13 410 6 h Gradiski et al. (1978)
Mouse male 21.1 3910 2 h Horiguchi & Horiguchi (1971)
Mouse male 99.1 18 358 1 h Moser & Balster (1985)
Mouse male 111 20 616 30 min Moser & Balster (1985)
Mouse male 159 29 492 10 min Moser & Balster (1985)
Table 12. Non-lethal acute toxicity in experimental animals
exposed to 1,1,1-trichloroethane by inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 45 800 4 h loss of righting Mullin & Krivanek (1982)
reflex (EC50)
Rat 20 400 4 h loss of coordination Mullin & Krivanek (1982)
(EC50)
Mouse 31 000 1 h inverted screen test Moser & Balster (1985)
performance (EC50)
Dog 37 800 5 min cardiac sensitization Clark & Tinston (1982)
to adrenaline
Dog 21 600 a few min decrease in blood Kobayashi et al. (1983)
pressure
The effect of 1,1,1-trichloroethane on liver enzyme activity in
the serum of experimental animals has been investigated. An almost
lethal intraperitoneal dose of 1,1,1-trichloroethane (0.87 ml/kg body
weight) caused a significant elevation of ALAT (alanine
aminotransferase) in dogs (Klaassen & Plaa, 1967), and one eighth of
a lethal intraperitoneal dose caused a significant elevation in SDH
(sorbitol dehydrogenase) (Lundberg et al., 1986). In both studies,
the hepatotoxicity of 1,1,1-trichloroethane was relatively small
compared to that of other organic solvents, e.g., chloroform, carbon
tetrachloride, dimethylformamide (Lundberg et al., 1986),
dichloromethane, 1,1,2-trichloroethane, and trichloroethylene
(Klaassen & Plaa, 1967).
7.1.1 Irritation
Trichloroethane has been shown to produce "mild" skin irritation
in rabbits when repeatedly applied topically under an occlusive
dressing. Redness was noted and the skin became scaly, but the effect
was transient, the skin rapidly returned to normal (Torkelson et al.,
1958).
After guinea-pigs had been exposed to 1 ml of
1,1,1-trichloroethane via a skin depot (a glass ring attached to the
skin and covered with glass), for 15 min, oedema was observed.
Prolonged exposure for several hours led to more severe inflammatory
reactions in the upper part of the dermis, and histological
examination at 15 min and 1, 4, and 16 h showed a number of changes to
the epidermis. The extent of these changes increased with duration of
exposure (Kronevi et al., 1981).
Repeated topical application of trichloroethane to abraded and
non-abraded rabbit skin for up to 90 days resulted in slight,
reversible irritation (Torkelson et al., 1958). The same study showed
that when trichloroethane was applied soaked in a cotton wool pad and
bandaged to the shaven belly of a rabbit slight reddening and
scaliness occurred, but this only increased slightly with repeated
applications.
The application of 0.5 ml trichloroethane to the shaven skin of
rabbits under an occlusive dressing for 24 h, resulted in moderate
skin irritation (Duprat et al., 1976). In a recent study of OECD
methods (4-h exposure under semi-occlusive dressing) trichloro-ethane
was reported to be a skin irritant (van Beek, 1990).
Trichloroethane is a "mild" eye irritant. Slight to moderate
pain and slight conjunctival irritation, but no corneal damage, was
reported following a single application of 100 µl trichloroethane to
the eyes of rabbits (Torkelson et al., 1958). Duprat et al. (1976)
also found that instillation of 0.1 ml trichloroethane to the eyes of
rabbits produced slight irritation.
7.1.2 Short-term exposure
7.1.2.1 Inhalation
When groups of rats, guinea-pigs, rabbits, dogs, and squirrel
monkeys were exposed to 1,1,1-trichloroethane (11 880 mg/m3, 2200
ppm) for 8 h/day, 5 days/week for 6 weeks, the only sign of toxicity
was reduced body weight gain in rabbits and dogs. There were no
effects on haematological parameters or serum urea nitrogen and no
histopathological changes were observed (Prendergast et al., 1967).
In a study by Adams et al. (1950), rats were exposed to 0 or 27
000 mg/m3 (0 or 5000 ppm) for 7 h/day on 32 out of 45 days and
guinea-pigs received 20 to 65 7-h exposures to 0, 3510, 8100, 16 200
or 27 000 mg/m3 (0, 650, 1500, 3000 or 5000 ppm) for a period of 1-3
months. Body weight gain was reduced in both species at all exposure
levels used, but there were no other signs of toxicity and no effects
were observed on blood urea nitrogen. The only 1,1,1-trichloroethane-
related effect observed on histopathological examination was fatty
degeneration, without necrosis, in the liver of guinea-pigs at 16 200
and 27 000 mg/m3.
Rats exposed to 2700 mg/m3 (500 ppm), 6 h/day for 5 days,
showed no behavioural effects. However, there was a slight decrease
in brain RNA content relative to controls (Savolainen et al., 1977).
Inhalation of 1750 mg/m3 (320 ppm) for 30 days had no effects on the
composition of brain lipids in rats (Kyrklund et al., 1988).
When rats were given 4320 mg/m3 (800 ppm) by inhalation 6
h/day, 5 days/week for 4 weeks, absolute and relative liver weights
were increased but there was no induction of liver microsomal
cytochrome P-450 (Toftgaard et al., 1981). A 1-h daily exposure to 54
000 mg/m3 (10 000 ppm) for 3 months resulted in a narcotic effect
(sedation and transient sleep) and in an increase of relative liver
weight in rats, but there was no evidence of organ damage (Torkelson
et al., 1958).
When rats were exposed continuously for 100 days to 1350 or 5400
mg/m3 (250 or 1000 ppm), no effects were observed in the low-dose
group but an increase in relative liver weight was seen in the
high-dose group (McEwen & Vernot, 1974). No toxicity was observed in
similar experiments with dogs and monkeys (McEwen & Vernot 1974).
Exposure of mice to 5400 mg/m3 (1000 ppm) continuously for 14
weeks resulted in marked liver changes (elevated relative liver
weight, moderate liver triglyceride accumulation, and necrosis of
individual hepatocytes). Electron microscopy showed extensive
cytoplasmic modifications consisting of vesiculation of the rough
endoplasmic reticulum with loss of attached polyribosomes, increased
smooth endoplasmic reticulum, microbodies (peroxisomes), and
triglyceride droplets. The observed toxic effects were similar to,
but much less severe than, those produced by carbon tetrachloride.
Only minor cytoplasmic alterations were seen in mice exposed to 1350
mg/m3 (250 ppm) (McNutt et al., 1975).
No signs of toxicity were observed in rats, rabbits, guinea-pigs,
dogs or monkeys exposed to 2730 mg/m3 (500 ppm) 7 h/day, 5 days/week
for 6 months (Torkelson et al., 1958).
In a study by Prendergast et al. (1967), rats, rabbits,
guinea-pigs, dogs, and monkeys were exposed continuously to 754 or
2059 mg/m3 for 90 days. At the higher dose level, there were no
deaths in any species after 90 days, and the authors stated that no
visible toxic signs were observed. However, at the lower dose level,
some deaths occurred (2 out of 15 rats and 1 out of 3 rabbits).
Varying degrees of lung congestion were noted in the surviving
animals. In view of this and the deaths at the lowest dose tested,
the authors stated that no positive conclusion could be drawn as to
whether the effects were associated with the exposure.
No adverse effects were seen in male Wistar rats exposed to 1100
mg/m3 (204 ppm) 8 h/day, 5 days/week for 14 weeks (Eben & Kimmerle,
1974).
When young adult Mongolian gerbils (Meriones ungiculatus) were
continuously exposed to 378, 1134 or 5400 mg/m3 (70, 210 or 1000
ppm) for 3 months, followed by a 4-month period without exposure,
increased glial fibrillary acidic protein (GFA) was found in the
cerebral cortex at the two highest exposure levels, indicating
astrogliosis in this region of the brain (Rosengren et al., 1985). In
a similar study by Karlsson et al. (1987), the DNA concentrations in
several brain regions were decreased in animals exposed to 378 mg/m3
(70 ppm), the only exposure concentration used.
Table 13 summarizes data from short-term inhalation studies.
7.1.2.2 Oral administration
When rats were dosed orally with 1,1,1-trichloroethane in corn
oil, 5 days/week for 6 weeks, a dosage of 3.2 g/kg body weight per day
had no adverse effects. However, total doses of 5.6 g/kg body weight
in females and 10 g/kg body weight in males induced 40% mortality and
decreased the body weight of survivors (NCI, 1977). In mice no
mortality was seen with a dose of 5.6 g/kg body weight. Thus,
trichloroethane has low toxicity in rats and mice following repeated
exposure by the oral route.
Table 13. Short-term toxicity (exposure-effect relationships) in
experimental animals exposed to 1,1,1-trichloroethane by
inhalation
Species Exposure Exposure Effects Reference
level duration
(mg/m3)
Rat 5400 100 days increased relative McEwen & Vernot
liver weight (1974)
Rat 1100 14 weeks, NOAEL Eben & Kimberle
8 h/day, (1974)
5 days/week
Gerbil 5400 3 months reduced brain weight Rosengren et al.
(1985)
Gerbil 380 3 months biochemical changes Karlsson et al.
in the brain (1987)
Mouse 5400 14 weeks liver necrosis McNutt et al.
(1975)
Mouse 1350 14 weeks minor liver effects McNutt et al.
(1975)
Rat, dog, 2700 6 months, NOAEL Torkelson et al.
rabbit, 7 h/day, (1958)
monkey, 5 days/week
guinea-pig
Rat, dog, 2000 90 days NOAEL Prendergast et al.
rabbit, (1967)
monkey,
guinea-pig
7.2 Long-term exposure
In the NCI (1977) carcinogenesis study (see section 7.5.1 for
details of doses), reduced weight gain was noted in both rats and mice
during the exposure period at all dose levels. Furthermore, bloody
discharge and crusting around the eyes of some rats was observed
during the second year (NCI, 1977).
In an oncogenicity inhalation study, female rats exposed to the
highest concentration (8100 mg/m3, 1500 ppm) of 1,1,1-
trichloroethane exhibited a significant decrease in body weight, and
both females and males showed very slight microscopic hepatic effects
at 6, 12, and 18 months. At lower concentrations and in male rats and
mice of both sex, no toxicity was observable (Quast et al., 1988).
7.3 Reproductive toxicity, embryotoxicity, and teratogenicity
Inhalation studies in female rats and mice gave no evidence of
any teratogenic or fetotoxic effects followed exposure to
1,1,1-trichloroethane at a concentration of 4700 mg/m3 (875 ppm) for
7 h/day on days 6-15 of gestation (Leong et al., 1975). No signs of
maternal toxicity were noted.
In a study of female rats exposed to a higher
1,1,1-trichloroethane concentration (11 340 mg/m3, 2100 ppm, 6
h/day, 5 days/week) for 2 weeks before mating and/or during 20 days of
gestation (6 h/day, 7 days/week), there was some evidence of
fetotoxicity. This consisted of reduced fetal weight in the group
exposed during pregnancy only and minor visceral and skeletal
abnormalities, such as delayed ossification, in the group exposed
before and during pregnancy (York et al., 1982). No signs of maternal
toxicity were observed.
In a two-generation fertility study incorporating teratogenicity
and dominant lethal elements, mice received 0, 100, 300 or 1000 mg
1,1,1-trichloroethane/kg daily in the drinking-water (Lane et al.,
1982). Two or three litters were produced from each of the two
generations, and the parental animals and offspring were used
variously to investigate fertility, developmental toxicity, and
dominant lethal effects. No adverse effects on any aspect of
reproduction were evident in this study. The level of
1,1,1-trichloroethane in the drinking-water in this study was reported
to be up to 6000 mg/litre but was not actually measured. In a further
study, rats exhibited slight aversion to 30 mg/litre in drinking-water
(George et al. 1989).
Rats (Sprague-Dawley) and rabbits (New Zealand White) were
exposed on days 6-15 and 6-18 of gestation, respectively, to 0, 5400,
16 200 or 32 400 mg/m3 (0, 1000, 3000 or 6000 ppm) for 6 h/day.
1,1,1-Trichloroethane was found to be developmentally toxic to both
rats and rabbits at the highest dose level, producing an increase in
unossified and poorly ossified cervical centra, decreased female fetal
body weight, an increase in non-viable implantations in rats, and an
increase in the frequency of bilateral extra 13th rib in rabbits.
Both rats and rabbits showed a NOEL of 16 200 mg/m3 for
developmental toxicity. For both species, the developmental toxicity
was seen in the presence of maternal tox-icity (Personal communication
by E.Z. Francis to S. Ells, US EPA, Test Rules Development Branch,
Existing Chemical Assessment Division. Review of Developmental
Toxicity Studies on 1,1,1-Trichloroethane (TCE). OHEA, ORD. Sept. 28,
1988).
Dapson et al. (1984) reported that exposure to 10 mg
1,1,1-trichloroethane per litre drinking-water caused cardiac
anomalies in developing Sprague-Dawley rats. The repeatability of
this preliminary study was investigated by George et al. (1989), who
exposed male and female CD rats to 3, 10 or 30 mg of 97% pure
1,1,1-trichloroethane per litre drinking-water using 0.05% Tween 80 as
an emulsifying agent. The animals were exposed for a period of 14
days before and at least 13 days after co-habitation. Sperm-positive
females continued to be exposed during pregnancy and lactation until
postnatal day 21. No significant effects were noted concerning the
reproductive competence of the parental animals or the postnatal
growth and development of the offspring (fertility, length of
gestation period, litter size, pup body weight, and pup survival were
all investigated). In addition, there was no increase in the
incidence of any cardiac malformations or other anomalies. Thus the
finding of Dapson et al. (1984) was not confirmed using a different
strain of rats and a 3-fold higher concentration of
1,1,1-trichloroethane.
7.4 Mutagenicity
Appraisal
1,1,1-Trichloroethane has been tested extensively for
mutagenicity, both in vitro and in vivo, and the principal studies
are summarized in Table 14. The results of most of the studies were
clearly negative. The positive results seen in some in vitro tests
give rise to some concern, but may well have been due to the presence
of stabilizers and/or impurities. Some of the studies yielding
negative results were of insufficient sensitivity. All of the in
vivo tests gave negative results. Overall, it appears that
1,1,1-trichloroethane does not have significant mutagenic potential.
Table 14. Mutagenicity studies
Test method Organism Test conditions Result References
In vitro studies on microorganisms
Baterial Salmonella typhimurium many studies, using mostly negative; some Simmon et al. (1977), Snow et al. (1979),
mutagenicity or Escherichia coli liquid or vapour, positive results attributed Nestmann et al. (1980), Gocke et al. (1981),
with and without S9 to stabilizers or impurities Rowland & Severn (1981), McDonald (1981),
activation Nagao & Takahashi (1981), Richold & Jones
(1981), Simmon & Shepherd (1981),
Ichinotsubo et al. (1981), de Serres &
Ashby (1981), HSE (1984), Shimada et al.
(1985)
Bacterial DNA E. coli or Bacillus several studies mainly negative; Kada (1981), Ichinotsubo et al. (1981),
damage/repair subtilis occasionally equivocal Green (1981), Tweats (1981), Rosenkranz
tests results et al. (1981), de Serres & Ashby (1981)
Yeasts, Saccharomyces numerous studies negative HSE (1984), de Serres & Ashby (1981),
gene mutation, cerevisiae
gene conversion Schizosaccharomyces
pombe
S. cerevisiae and mice 5000 mg/kg negative Loprieno et al. (1979)
(host-mediated assay) intraperitoneal
Table 14 (contd).
Test method Organism Test conditions Result References
In vitro mammalian cell studies
Chromosome CHO cells ± S9 activation clear positive without S9; Galloway et al. (1987)
aberrations equivocal with S9
Gene mutation L5178Y mouse ± S9 equivocal with S9; Myhr & Caspary (1988)
lymphoma cells negative without S9
Unscheduled rat, mouse hepatocytes; ± S9 negative Althaus et al. (1982), Martin &
DNA synthesis HeLa cells McDermid (1981)
DNA repair rat hepatocytes ± S9 mainly negative; some Williams (1983), Shimada et al. (1985)
positive results attributed
to presence of stabilizers
or impurities
Sister chromatid CHO cells ± S9 negative Perry & Thomson (1981)
exchange equivocal Galloway et al. (1987)
Cell various cell types ± S9 both positive and Daniel & Dehnel (1981), Styles (1981),
transformation negative results Price et al. (1978), Hatch et al. (1983)
Tu et al. (1985), HSE (1984)
Drosophila studies
Sex-linked 25 nM negative Gocke et al. (1981)
recessive
lethals
Table 14 (contd).
Test method Organism Test conditions Result References
In vivo mammalian studies
Chromosome rats 875 or 1750 ppm negative Quast et al. (1978)
aberrations 6 h/day, 52 weeks
(bone marrow)
Micronucleus mice 34-67 mg/kg negative Salamone et al. (1981)
intraperitoneal
Micronucleus mice 11-42 mg/kg negative Tsuchimoto & Matter (1981)
intraperitoneal
Micronucleus mice 266-2000 mg/kg negative Gocke et al. (1981)
intraperitoneal
Dominant mice 100-1000 mg/kg per negative Lane et al. (1982)
lethal day oral
Sperm mice 130-2680 mg/kg per negative Topham (1980)
morphology day intraperitoneal
DNA binding rats and mice low binding Prodi et al. (1988)
(liver)
7.5 Carcinogenicity
1,1,1-Trichloroethane has been investigated for carcinogenic
effects in life-time bioassays on both rats and mice by oral
administration or inhalation.
7.5.1 Oral administration
In a US National Cancer Institute (NCI) assay, 7 week-old
Osborne-Mendel rats were fed by gavage a 75% solution of technical
grade 1,1,1-trichloroethane in corn oil, 5 days/week for 78 weeks.
The daily doses were calculated to be 750 or 1500 mg/kg body weight,
and the study was terminated after 110 weeks. The number of animals
in each treated group was 50 of each sex, and the control groups
consisted of 20 animals of each sex, which were not vehicle dosed. No
increased tumour incidence was found (NCI, 1977). However, because of
an unusually high mortality both among treated and control animals
(only 6 out of 240 animals survived the 110 weeks to the end of the
experiment), this study was considered insufficient by US EPA (1984)
and placed in class D (not applicable to assess human
carcinogenicity).
In another NCI study with 5-week-old B6C3F1 mice, technical grade
trichloroethane containing 3% 1,4-dioxane was given by gavage as a
40-60% solution in corn oil at two dose levels (average daily doses:
2807 and 5615 mg/kg body weight) 5 days/week for 78 weeks. The same
number of animals was used as in the NCI rat study reported above.
All surviving animals were killed after 90 weeks of study. A variety
of neoplasms was observed in the treated groups, but the incidence was
not statistically significantly different from that of the control
groups (NCI, 1977). Again the mortality rate was very high in both
treated and control animals, although it was a little lower than in
the rat study. Nevertheless this study was also considered
insufficient (US EPA 1984).
In a study by Maltoni et al. (1986), groups of male and female
rats were given 1,1,1-trichloroethane (500 mg/kg body weight) in olive
oil by gavage (4 to 5 days/week for 104 weeks). The
1,1,1-trichloroethane contained about 4% 1,4-dioxane and 1% of other
impurities. An increased incidence of "leukaemias" was detected in
the exposed group. However, data were not evaluated statistically nor
were any data for historical controls supplied.
7.5.2 Inhalation
In an inhalation study carried out by the Dow Chemical Co.,
1-month-old Sprague-Dawley rats were exposed to technical
1,1,1-trichloroethane (6 h/day, 5 days/week for 12 months) and
observed 18 months later, before the survivors were killed. Each dose
group consisted of 96 rats of each sex. The unexposed control group
consisted of the same number of rats as the two exposed groups
together. The two exposure concentrations used were 4778 mg/m3 (875
ppm) and 9555 mg/m3 (1750 ppm). The treatment had no significant
effect on mortality, and the tumour incidence in the treated animals
was similar to that of the controls (Rampy et al., 1977). The purity
of the 1,1,1-trichloroethane used was 96%, and the stabilizers were 3%
1,4-dioxane, 0.4% nitromethane, and 0.5% butylene oxide. In the
validation of this study, it was pointed out that the exposure period
was only 12 months and not lifelong, that no subchronic range-finding
test had been done, and that the exposure concentration could have
been too low (US EPA, 1984).
A more recent long-term inhalation study, this time on both rats
and mice, has been carried out by the Dow Chemical Co. (Quast et al.,
1988). Groups of male and female 4- to 6-week-old Fischer-344 rats
and 5- to 6-week-old B6C3F1 mice (80 of each sex per group) were
exposed to 1,1,1-trichloroethane (technical formulation) vapour
concentrations of 0 mg/m3, 820 mg/m3 (150 ppm), 2730 mg/m3 (500
ppm), 8190 mg/m3 (1500 ppm) 6 h/day, 5 days/week for 2 years. Ten
animals of each sex from each group were predesignated for interim
sacrifices after 6, 12, and 18 months of exposure. The purity of the
formulation was 94% (by volume) 1,1,1-trichloroethane, 5% stabilizers
(butylene oxide, tert-amyl alcohol, methyl butynol, nitroethane, and
nitromethane) and < 1% minor impurities. In none of the treated
animal groups, did the tumour incidence differ significantly from that
of the controls.
The early studies with mice and rats were evaluated at IARC
(International Agency for Research on Cancer) and considered
inadequate (IARC, 1979; IARC, 1987).
7.6 Immunotoxicity and sensitization
Shmuter (1977) exposed rabbits to 1,1,1-trichloroethane
concentrations of 2 mg/m3 (0.4 ppm), 10 mg/m3 (1.8 ppm), and 100
mg/m3 (18 ppm) by inhalation 3 h/day, 6 days/week for 8-10 months.
At week 6, Salmonella typhimurium was injected subcutaneously to
investigate the immune response over an 8-month period. The antibody
response was depressed at both 10 mg/m3 and 100 mg/m3. This
reduction in immune response was followed by an increased
electrophoretic mobility of the antibodies toward alpha- and
ß-globulin fractions and also by a significant drop in the level of
normal haemolysins to the Forsman's antigen of the sheep erythrocytes
occuring during treatment.
7.7 Interactions
Interaction between 1,1,1-trichloroethane and ethanol was studied
in mice given ethanol (0-2 g/kg body weight) by gavage 30 min before
exposure to up to 120 000 mg trichloroethane/m3 (22 200 ppm) by
inhalation. Different combinations of exposure to the solvents
enhanced the toxicity of trichloroethane in the animals previous
exposed to ethanol. The observed behavioural and lethal effects were
dose related for each compound and additive (Woolverton & Balster,
1981).
When dogs anaesthetized with either pentobarbital sodium or
chloralose plus pentobarbital sodium were acutely exposed to a
commercial spot remover containing 1,1,1-trichloroethane, there was a
dose-dependent biphasic decline in arterial pressure with an initial
increase in the heart rate. A similar effect was obtained when
1,1,1-trichloroethane (99.5% by volume) was used instead of the
commercial spot remover. It was found that the type of anaesthetic
contributed in part to the effect. The authors reported that, in
addition to the peripheral vascular effects, significant alterations
in myocardial function occurred, and they concluded that the solvent
acts by inhibiting myocardial contractility (Herd et al., 1974).
7.8 Mechanisms of action
1,1,1-Trichloroethane given to mice intraperitoneally or by short
inhalation exposure has been shown to reduce the cyclic guanosine
monophosphate (cGMP) content in the cerebellum, cerebral cortex, and
brain stem. This was attributed to an increased rate of cGMP
hydrolysis (Nilsson, 1986a). The cyclic adenosine monophosphate
(cAMP) content in the brain stem of mice was increased in a
dose-related and time-dependant manner. This might be mediated by
adrenoceptor interaction stimulating the catalytic unit of adenylate
cyclase (Nilsson, 1986b).
A study on isolated mouse brain synaptosomes exposed in vivo
and in vitro showed that 1,1,1-trichloroethane increases calcium
influx in the brain stem but not in the cerebellum and cerebral
cortex. The data indicated that 1,1,1-trichloroethane may influence
voltage-dependant calcium channels in brain stem nerve endings
(Nilsson, 1987).
1,1,1-Trichloroethane suppressed the uptake of ovabain
taurocholate and 2-aminoisobutyric acid, but not of cadmium chloride
or 3- O-methyl-D-glucose, into isolated rat hepatocytes. Both the
cellular ATP level and ATP-ase activities in isolated membrane
fractions were also decreased. It was suggested that energy-dependant
transport functions are disrupted, owing to decreased ATP levels
and/or inhibition of membrane ATP-ases (Kukongviriyapan et al., 1990).
8. EFFECTS ON HUMANS
Appraisal
Both acute and long-term inhalation exposure to
1,1,1-trichloroethane can produce CNS effects ranging from slight
behavioural changes to unconsciousness. Death may follow from
respiratory failure or cardiac arrest. There is also the possibility
of hepatic alterations and cardiac damage.
Ingestion and dermal exposure do not appear to be serious
concerns, although skin irritation may occur.
1,1,1-Trichloroethane has properties (see chapter 2) that make it
a dangerous compound in poorly ventilated areas and confined spaces
such as tanks and vaults. The vapour is five times more dense than
air, which may result in non-uniform distribution, and very high
levels can occur in certain areas such as the bottom of empty tanks
and container vessels. The combination of these properties has been
the cause of some serious accidents, including fatalities. Solvent
abuse has also produced a large number of fatalities.
There are insufficient studies to permit any conclusion
concerning the reproductive and carcinogenic effects of this compound
on humans.
Exposure to 1,1,1-trichloroethane may increase sensitivity to
other compounds, e.g., anaesthetics.
The NOEL for humans appears to be in the region of 1350 mg/m3
(250 ppm).
Table 15 summarizes the toxic effects of 1,1,1-trichloroethane
after inhalation exposure.
8.1 Controlled human studies
Several studies on volunteers under controlled conditions have
investigated the effects of 1,1,1-trichloroethane on the CNS as
measured by performance in certain behavioural tests. These studies
have usually been conducted using the technical grade of
1,1,1-trichloroethane containing stabilizers, since this is the type
of exposure that occurs most frequently.
Table 15. Toxic effects in humans related to inhalation exposure to 1,1,1-trichloroethane
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
378 000 70 000 a few min death Northfield (1981)
54 000 10 000 2 min general anaesthesia when given Dornette & Jones (1960)
together with nitrous oxide (N2O)
> 27 000 > 5000 10 min death Kleinfeld & Feiner (1966)
27 000 5000 5 min marked incoordination Stewart (1968)
10 260 1900 5 min disturbance of equilibrium Torkelson et al. (1958)
5400 1000 short-term mild eye and nasal discomfort, Stewart (1968)
impairment of coordination
5000 920 70-75 min CNS disturbances (lightheadedness), Torkelson et al. (1958)
slight eye irritation
4860 900 73 min impaired performance in Romberg's Stewart et al. (1961)
test, mild eye irritation
2700 500 6-7 h/day for impaired performance in Romberg's Stewart et al. (1969)
5 days test after 1 h of exposure, mild
eye irritation, drowsiness, headaches
2700 500 90-450 min no CNS disturbances Torkelson et al. (1958)
2430 450 30 min dizziness, mild eye irritation Salvini et al. (1971)
2430 450 30 min reduced reaction time and Gamberale & Hultengren (1973)
perceptual speed
Table 15 (contd).
Exposure concentration Duration of exposure Effect Reference
(mg/m3) (ppm)
1890 350 30 min reduced perceptual speed Gamberale & Hultengren (1973)
1900 350 3.5 h reduced psychomotor performance Mackay et al. (1987)
< 1860 < 345 6.7 years NOEL Maroni et al. (1977)
< 1350 < 250 1-6 years NOEL Kramer et al. (1978)
1350 250 30 min NOEL Gamberale & Hultengren (1973)
950 175 3.5 h reduced reaction time MacKay et al. (1987)
540 100 odour threshold Stewart (1968)
8.1.1 Single exposure period
In a study by Torkelson et al. (1958), four volunteers were
exposed in an inhalation chamber to average trichloroethane levels of
4968 mg/m3 (920 ppm) for 70-75 min. Symptoms of slight CNS
disturbances (lightheadedness) were noted in three of the four
subjects and slight eye irritation was noted in one individual. Two
volunteers reported a strong odour during the experiment.
Concentrations in the range of 2700-2970 mg/m3 (500-550 ppm) for
90-450 min apparently produced no symptoms of CNS disturbance, but
exposure to 10 260 mg/m3 (1900 ppm) for 5 min produced a very
noticeable odour and obvious disturbances in equilibrium.
Unfortunately, full details of this study are not available.
Exposure to about 2700 mg/m3 (500 ppm) trichloroethane for
78-186 min did not result in CNS toxicity or other effects (Stewart et
al., 1961). However, exposure to about 4860 mg/m3 (900 ppm) for up
to 73 min resulted in CNS disturbances (light-headedness and impaired
Romberg test performance) in approximately half of the subjects. Mild
eye irritation was also observed. In another experiment in this
series, the volunteers were exposed to increasing concentrations in
the range of 0-14 310 mg/m3 (0-2650 ppm) over a 15-min period.
Signs of mild eye irritation were noted in six out of seven subjects
when the concentration reached about 5400 mg/m3 (1000 ppm), and
there was a rapid increase in CNS symptoms, principally dizziness.
Throat irritation was experienced by six out of seven subjects at
about 10 800 mg/m3 (2000 ppm). Severe CNS disturbances (imbalance,
light-headedness) occurred at the end of the exposure period, when the
concentration had reached 14 310 mg/m3. In addition, minor effects
on liver and kidney function were seen. The authors considered that
all the effects were reversible.
The effect on young men of exposure to a mean trichloroethane
level of 2430 mg/m3 (450 ppm) for two periods of 4 h (separated by
a lunch break of 1´ h) was investigated by Salvini et al. (1971). The
volunteers experienced dizziness and slight excitation, but only
during the first 30 min of exposure, and mild eye irritation was
noted. No significant change in performance in any of the behavioural
tests was seen.
In a study by Gamberale & Hultengren (1973), 12 male volunteers
were exposed to 1350 mg/m3 (250 ppm) for 30 min, followed by 1890
mg/m3 (350 ppm) for the next 30 min, and 2430 mg/m3 (450 ppm) and
2970 mg/m3 (550 ppm) for subsequent 30-min periods. At 2430
mg/m3, the reaction time, perceptual speed, and manual dexterity
were reduced, and at 1890 mg/m3 the perceptual speed was reduced.
The NOEL was 1350 mg/m3.
Mackay et al. (1987) reported the results of an exposure chamber
study in which volunteers were exposed to 950 mg/m3 (175 ppm) and
1990 mg/m3 (350 ppm) for 3 to 5 h. Several human psychomotor
behavioural tests were employed, including two new ones. One was
concerned with distraction of attention and concentration (the Stroop
test) and the other with analysing grammatical statements (the
syntactic reasoning test). Performance deficits in the psychomotor
behavioural tests were recorded in volunteers exposed to
1,1,1-trichloroethane; these depended on the time of exposure and the
blood levels. The performance changes were rapid and occurred in some
cases within 20 min. However, different effects were found. In the
Stroop test, enhanced performance was observed following exposure,
whereas the syntactic reasoning test was found not to respond to
1,1,1-trichloroethane exposure effects. Measures of short-term
subjective well-being were not affected by exposure. Other
parameters, such as simple reaction time and four-choice reaction time
were increased.
The effect of exposure to trichloroethane on performance in a
very extensive range of behavioural tests was investigated by
Savolainen et al. (1981, 1982a, 1982b). Nine healthy male students
(aged 20-25) were exposed in an inhalation chamber to 1080 and 2160 mg
trichloroethane/m3 (200 and 400 ppm) for 4 h, with a 6-day interval
between exposure at the two levels. There was no clear indication of
any effects on the CNS, although some minor changes were noted in
body-sway measurements when the subjects had their eyes closed.
8.1.2 Repeated exposure
The effect of repeated exposure of male volunteers to 2700
mg/m3 (500 ppm), 7 h/day for 5 days, was studied in an inhalation
chamber (Stewart et al., 1969). Several subjective symptoms were
noted, e.g., drowsiness, headaches, lightheadedness, and eye and nose
irritation, but the absence of a control group made it difficult to
assess the significance. No performance impairment in behavioural
tests (modified Romberg test) was noted, apart from the inability of
two subjects to perform the balance test on two occasions.
8.2 Accidental exposure
Respiratory system symptoms, such as cough, breathlessness, and
chest tightness, are common in cases of acute poisoning (Boyer et al.,
1987).
Since 1,1,1-trichloroethane is a very volatile solvent with a
saturated vapour concentration of 864 g/m3 (160 000 ppm) at 25 °C,
there is a potential for very high air concentrations in confined
spaces, such as tanks and vaults. Furthermore, its vapour is five
times more dense than air. This may result in non-uniform
distribution in the workplace air and very high levels in certain
areas such as just above degreasing vessels and at the bottom of
empty tanks, even though levels elsewhere are low and safe. This
property has been the cause of some serious accidents and fatalities.
A near-fatal childhood intoxication was described by Gerace
(1981). A 4-year-old boy was playing under his bed covers with a
flower-making kit which contained a 30-cm3 container of
1,1,1-trichloroethane. Because of some unusual noises heard by a
sibling, the child was quickly discovered. However, he was not
breathing at this point. He was resuscitated on the way to the
hospital but remained comatose for about 12 min after arrival. The
author suggested that the child was rendered unconscious by the
1,1,1-trichloroethane and that the noises heard were seizures caused
by hypoxia. The child was released 48 h after admission. According
to the author, clinical investigation during the child's hospital stay
demonstrated some hepatic abnormalities with alteration in liver
bilirubin.
Caplan et al. (1976) reported the death of a woman due to
accidental exposure while cleaning a paint spill using a paint thinner
in a poorly ventilated room. Autopsy results showed "mild fatty
changes" in the liver and acute oedema and congestion of the lungs.
The concentrations of 1,1,1-trichloroethane in the brain, kidney,
liver, and blood were 36, 12, 5, and 2 mg/100 ml, respectively.
8.2.1 Confined spaces at workplaces
A man who had spent 10 min in a vault, where the concentration of
1,1,1-trichloroethane was estimated to be well in excess of 27 g/m3
(5000 ppm), collapsed and died shortly after leaving the vault
(Kleinfeld & Feiner, 1966).
Seven fatalities from using trichloroethane to clean equipment on
naval ships or aircraft tanks have been reported (Stahl et al., 1969;
Hatfield & Maykoski, 1970). The exposure level in one of these cases
was estimated to be in the order of 270 g/m3 (50 000 ppm). In all
cases the most significant pathological finding was pulmonary oedema.
Liver damage was not seen.
Other case histories with accidental death due to
1,1,1-trichloroethane exposure in confined spaces have been reported
(Stewart 1968; Bonventre et al., 1977).
During the period 1961-1980, the Factory Inspectorate in the
United Kingdom revealed 52 incidents due to trichloroethane (McCarthy
& Jones, 1983). Loss of consciousness was reported in 26 cases and
death in three cases, all involving very young workers. In one case
the worker died after leaning over an open tank of trichloroethane,
apparently to wash his hands in the solvent. He was found unconscious
slumped over the side of the tank, and the solvent concentration in
his breathing zone was later estimated to be about 378 g/m3 (70 000
ppm) (Northfield, 1981). In a second case, a worker was using
trichloroethane for cleaning purposes in a closed room (Jones &
Winter, 1983). He was found on the floor with chemical burns on the
head and neck, which was consistent with prolonged skin absorption
from spills of solvent on the floor. The third case was a teenage
worker who cleaned the interior of a car with a cloth soaked in the
solvent (Jones & Winter, 1983). Death was presumed to be from
1,1,1-tri-chloroethane intoxication and simultaneous inhalation of
vomit.
8.2.2 Solvent abuse
During the 1960s, 110 cases of sudden death from solvent abuse
were reported in the USA. Of these, spot remover containing
1,1,1-trichloroethane was identified as responsible for 29 cases
(Bass, 1970). Death frequently occurred immediately following
stressful activity. Where autopsies were performed, no abnormalities
could be detected, and it was suggested that deaths was due to cardiac
arrhythmia.
Over the period 1971-1981, 140 deaths from solvent abuse were
reported in the United Kingdom; 20 of them were believed to be due to
1,1,1-trichloroethane, mainly in dry-cleaning fluid, domestic cleaners
or plaster remover (Anderson et al., 1982).
In the case of a fatality due to the abuse of cleaning solvent
containing 1,1,1-trichloroethane, very minor hepatic changes were seen
(Hall & Hine, 1966). In four cases of deaths caused by inhalation of
typewriter correction fluid containing 1,1,1-trichloroethane and
trichloroethylene, the autopsies showed no evidence of kidney or liver
damage (King et al., 1985).
According to Marjot & McLeod (1989), toluene and the chlorinated
hydrocarbons 1,1,1-trichloroethane and trichloroethylene can cause
permanent damage to the kidney, liver, heart, and lung in certain
volatile substance abusers.
8.2.3 Medical use
Trichloroethane has been used as an anaesthetic on an
experimental basis. General anaesthesia in humans has been rapidly
induced by 2 min of exposure to 54 000 mg/m3 (10 000 ppm) given with
a mixture of nitrous oxide (N2O) and oxygen (80:20). Cardiovascular
side effects (slight to severe hypotension, cardiac arrhythmias, and,
in one case, cardiac arrest) were observed in several of the patients
during anaesthesia. In view of these effects, the experimental use of
1,1,1-trichloroethane as an anaesthetic was discontinued (Dornette &
Jones, 1960).
In the USA, after aerosol drug products containing
1,1,1-trichloroethane had been implicated in 21 deaths from their
abuse or misuse, the Food and Drug Administration (FDA) removed the
products from the market (US Food and Drug Administration, 1973). The
medical products were mainly used as cough suppressants and contained
1,1,1-trichloroethane as a solvent for the active ingredients and to
reduce the vapour pressure of the propellants. The FDA found lack of
general recognition by qualified experts of the safety or
effectiveness of 1,1,1-trichloroethane in aerosol drug products
intended for inhalation.
Under 1977 rules from the FDA, it was stated that
1,1,1-trichloroethane is potentially toxic to the cardiovascular
system by sensitizing the heart to adrenaline (Food and Drug
Administration, 1977).
8.2.4 Ingestion
In a case of accidental ingestion of about 600 mg
trichloroethane/kg, signs of severe gastrointestinal irritation
(vomiting, diarrhoea) were evident shortly after the ingestion. There
was no evidence of any CNS disturbance or any liver or kidney
dysfunction (Stewart & Andrews, 1966).
Ingestion of about 30 ml trichloroethane has been found to cause
severe vomiting and diarrhoea (CEC, 1986).
8.2.5 Drinking-water contamination
In December 1981, a water well in Santa Clara County, California,
USA, was removed from service after contamination with
1,1,1-trichloroethane and other solvents was detected. A
1,1,1-trichloroethane level of 1.7 mg/litre was detected at the well
head, but the level of the other contaminants (dichloroethylene,
isopropyl alcohol, and freon) was not reported. Subsequently
suspicion of a cluster of spontaneous abortions and congenital heart
anomalies from pregnancies conceived during 1980-1981 was reported by
the local community. Epidemiological studies indicated that a cluster
did exist, that the odds ratio for spontaneous abortion was 2.3 and
the relative risk for congenital malformations was 3.1, but they were
unable to draw a direct link between the ingestion of the contaminated
water and these effects (Goldberg et al., 1990).
8.3 Effects on the skin and eyes
Prolonged or repeated skin contact with 1,1,1-trichloroethane may
lead to dermatitis, due to its defatting action. However, despite its
wide use only a few cases of skin irritancy have been reported
(Torkelson et al., 1958).
Immersion of a hand in liquid 1,1,1-trichloroethane for 30 min
resulted in mild erythema, which persisted for one hour (Stewart &
Dodd, 1964).
Splashed in the eye, the compound may produce conjunctivitis
(Stewart, 1968). However, accidental eye contact has been reported to
produce only transient irritation (Savolainen et al., 1982a,b).
Exposure to a vapour concentration of 2700 mg/m3 (500 ppm) may
produce mild eye irritation (see section 8.1).
8.4 Long-term occupational exposure
A case study described a patient with liver cirrhosis after
several years of heavy exposure to trichloroethylene followed by 3
months of work that frequently involved using aerosolized degreaser
containing trichloroethane (Thiele et al., 1982).
An investigation of the neurotoxicity of trichloroethane was
carried out on a group of 22 female workers exposed to the solvent for
an average period of 6.7 years. The group was subdivided into three
exposure groups (mean levels: 594 mg/m3 (110 ppm), 756-864 mg/m3
(140-160 ppm), and 1080-1863 mg/m3 (200-345 ppm)). Each woman had
a general physical examination, and information about symptoms was
obtained from a questionnaire. The measurement of nerve conduction
velocity and psychometric functions indicated no differences between
the exposed workers and an unexposed control group of 7 workers
(Maroni et al., 1977).
Hodgson et al. (1989) reported that four out of ten clinical
cases of fatty liver disease were associated with occupational
exposure to 1,1,1-trichloroethane.
A matching-pair study of 151 pairs of workers at textile plants,
with and without daily exposure to trichloroethane levels of up to
1365 mg/m3 (250 ppm), found no toxic effect among the exposed
workers, the majority of whom were exposed to less than 810 mg/m3
(150 ppm) for a period of 1-6 years (Kramer et al., 1978).
In a case-control study of the effect of occupational exposure to
various solvents on spontaneous abortion rates in Finland (1973-1983),
a statistically significant elevation in relative risk was found for
exposure to organic solvents in general. However, there were very few
cases related specifically to 1,1,1-trichloroethane. The low
statistical power, therefore, precludes any conclusion as to whether
the data show an effect or not for 1,1,1-trichloroethane (Lindbohm et
al., 1990)
There are no adequate epidemiological studies available
concerning long-term exposure to 1,1,1-trichloroethane and
carcinogenicity.
8.5 Interactions
Chronic cardiac arrhythmia developed in two cases, after routine
halothane anaesthesia, in subjects with previous repeated long-term
exposure to 1,1,1-trichloroethane (in one case, occupational and in
the other, due to solvent abuse). The effects involved myocardiac
damage with either ventricular arrhythmia or deterioration of left
ventricular function. In one of the cases, the pathologist suggested
a subacute myocarditis superimposed on long-standing damage. The
authors postulated that the interaction between the two chemicals may
have caused the cardiac deterioration and concluded that cardiac
damage sustained after long-term exposure to 1,1,1-trichloroethane may
be severe, life-threatening, and irreversible (McLeod et al.,1987)
Marjot & McLeod (1989) reported two cases, a 14-year-old boy
exposed to 1,1,1-trichloroethane via solvent abuse and a 54-year-old
man exposed occupationally, where the cardiac toxicity of
1,1,1-trichloroethane may have been increased by exposure to halothane
(2-bromo-2-chloro-1,1,1-trifluoroethane) as an anaesthetic.
9. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
Appraisal
1,1,1-Trichloroethane is not likely to be a significant hazard to
non-target organisms in the environment. For example, 96-h LC50
values for fish ranged from 33 mg/litre for dabs to 105 mg/litre for
fathead minnows. The 48-h LC50 for Daphnia was around 60 mg/litre.
The EC50 for carbon dioxide incorporation into algae was 5
mg/litre.
9.1 Microorganisms
A toxic threshold of 93 mg 1,1,1-trichloroethane/litre has been
estimated for the inhibition of cell multiplication in the bacterium
Pseudomonas putida (Bringmann & Kuhn, 1977). The 3-h EC50 values,
for inhibition of photosynthesis (measured by monitoring the uptake of
14CO2), for the green algae Chlamydomonas angulosa (at a cell
concentration of 5 x 104 cells/ml) and Chlorella vulgaris (at a
cell concentration of 20 x 104 cells/ml) were 280 mg/litre and 153
mg/litre, respectively, under static conditions (Hutchinson et al.,
1980). Bringmann & Kuhn (1978) calculated toxic thresholds of 350
mg/litre and 430 mg/litre, respectively, for inhibition of cell
multiplication, over an 8-day period, of the cyano bacterium
(blue-green alga) Microcystis aeruginosa and the green alga
Scenedesmus quadricauda. The 96-h EC50 for chlorophyll a and cell
number in the green alga Selenastrum capricornutum was > 669
mg/litre (US EPA, 1980a,b). Pearson & McConnell (1975) assessed the
toxicity of 1,1,1-trichloroethane to the unicellular alga
Phaeodactylum tricornutum by measuring changes in the uptake of
carbon, from 14CO2, during photosynthesis, and obtained an EC50
of 5 mg/litre.
9.2 Aquatic organisms
The acute toxicity of 1,1,1-trichloroethane to aquatic organisms
is summarized in Table 16.
Flow-through tests gave much lower LC50 values than static
tests, suggesting that some loss via evaporation may be occurring.
The 48-h LC50 for aquatic invertebrates ranges from 7.5 mg/litre for
barnacles in a flow-through test to > 530 mg/litre for daphnids in a
static test. The 96-h LC50 for fish ranges from 33 mg/litre for the
dab (Limanda limanda) in flow-through tests to 105 mg/litre for the
fathead minnow (Pimephales promelas) in static tests.
Table 16. Acute toxicity of 1,1,1-trichloroethane to aquatic organisms
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Invertebrates
Mysid shrimp stat 96 31.2 n US EPA (1980b)
(Mysidopsis bahia)
Barnacle nauplii stat 48 7.5 n Pearson & McConnell (1975)
(Eliminius modestus)
Water flea < 24 h stat 21-23 72 6.7-8.1 48 > 530 n LeBlanc (1980)
(Daphnia magna) 4-6 day stat 21-25 48 59.6 Abernethy et al. (1986)
4-6 day stat 48 57.6 Bobra et al. (1984)
Fish
Dab 15-20 cm flow 96 33 m Pearson & McConnell (1975)
(Limanda limanda)
Sheepshead minnow 8-15 mm stat 25-31 10-31d 96 71 (60-81) n Heitmuller et al. (1981)
(Cyprinodon variegatus)
Bluegill 0.32-1.2 g stat 21-23 32-48 6.7-7.8 24 110 n Buccafusco et al. (1981)
(Lepomis macrochirus) 0.32-1.2 g stat 21-23 32-48 6.7-7.8 96 72 (57-90) n Buccafusco et al. (1981)
stat 96 69.7 n US EPA (1980b)
Fathead minnow 1 g stat 12 7.8-8.0 96 105 (91-126) n Alexander et al. (1978)
(Pimephales promelas) 1 g flow 12 7.8-8.0 96 52.8 (43.7-77.7) m Alexander et al. (1978)
Table 16 (contd).
Organism Age/ Stat/ Temperature Hardnessc pH Duration LC50e Reference
size flowa (°C) (h) (mg/litre)
Fish (contd)
Guppy 2-3 month statb 168 133 Konemann (1981)
(Poecilia reticulata)
a stat = static conditions (water unchanged for the duration of the test) unless stated otherwise; flow = flow-through conditions
(1,1,1-trichloroethane concentration continuously maintained)
b static conditions but water renewed daily
c hardness expressed as mg CaCO3 per litre
d value refers to salinity (%), not hardness
e n = nominal; m = measured
LeBlanc (1980) exposed the water flea Daphnia magna to
1,1,1-trichloroethane for 48 h under static conditions. At the
highest concentration of 530 mg/litre, no discernible effect was
observed. A no-observed-effect level (NOEL) of 43 mg/litre was
estimated by Heitmuller et al. (1981) after exposing the sheepshead
minnow (Cyprinodon variegatus) to 1,1,1-trichloroethane for 96 h.
When Thompson & Carmichael (1989) exposed the water flea Daphnia
magna to 1,1,1-trichloroethane concentrations of between 1.3 and 23
mg/litre for 17 days under semistatic conditions (water renewed every
2 days), no effect on mortality or reproduction (as measured by number
of offspring per parent) was observed at 1.3 mg/litre. However, the
mortality increased with increasing concentration of
1,1,1-trichloroethane from 30% at 2.4 mg/litre to 100% at 23 mg/litre.
The number of offspring per parent was significantly reduced at
concentrations of 2.4 mg/litre or more.
Thompson & Carmichael (1989) also exposed the mirror carp
Cyprinus carpio to 1,1,1-trichloroethane concentrations ranging from
1.4 to 30 mg/litre for 14 days under flow-through conditions. There
was no mortality at any of the concentrations, but at the highest
concentration the fish showed surfacing behaviour, loss of balance and
coughing, and their mean weight decreased. The authors considered,
therefore, that the NOEL was the next highest concentration, i.e. 7.7
mg/litre.
Alexander et al. (1978) observed fathead minnows (Pimephales
promelas), during exposure to 1,1,1-trichloroethane for up to 96 h,
for the following effects: loss of equilibrium, melanization,
narcosis, and swollen, haemorrhaging gills. The concentration
producing one or more of these observable effects in 50% of the fish
(EC50) was 11.1 mg/litre.
9.3 Terrestrial organisms
Thompson & Carmichael (1989) exposed emergent seedlings of crop
sorghum (Sorghum bicolor) and oil seed rape (Brassica napus) to
1,1,1-trichloroethane in the gaseous phase at nominal concentrations
of between 3.2 and 320 mg/litre (g/m3) for a period of 14 days (the
test material was replenished after 7 days in a sealed vessel). Mean
measured concentrations ranged from 58% to 75% of the nominal
concentration, reflecting the decline of 1,1,1-trichloroethane in the
gas phase concentration during the 7-day periods between toxicant
renewal. In the case of Sorghum bicolor, the 14-day EC50 (based
on mean plant weight and a mean measured 1,1,1-trichloroethane
concentration) was 48 mg/litre; the NOEL being 19 mg/litre. For
Brassica napus, the 14-day EC50 was 19 mg/litre and the NOEL 6.9
mg/litre, both values being based on mean measured concentrations.
Rajendran (1990) exposed 1- to 2-day-old pupae of the red flour
beetle (Tribolium castaneum) to 1,1,1-trichloroethane in fumigation
chambers. An LC50 of 208.4 mg/litre was estimated after a 24-h
exposure and a 20-day post-fumigation observation period. When pupae
were exposed to a mixture of 1,1,1-trichloroethane and methyl bromide
at a concentration of 2.5 mg/litre or 3.0 mg/litre, the toxicity was
similar to that of the individual components.
In a contact toxicity test, red earthworms (Eisenia foetida)
were exposed to 1,1,1-trichloroethane via filter paper in glass vials.
An LC50 of 83 µg/cm2 was estimated (Neuhauser et al., 1986).
Elovaara et al. (1979) injected 1,1,1-trichloroethane, at
concentrations ranging from 5 to 100 µmol/egg, into the air space of
fertilized chicken eggs at 3 or 6 days of incubation, and the
embryotoxicity was evaluated after 14 days of incubation. A clear
dose-response relationship with respect to survival was found,
irrespective of whether the eggs were injected after 3 or 6 days of
incubation. The approximate LD50 for 1,1,1-trichloroethane was
between 50 and 100 µmol/egg.
10. EVALUATION OF HUMAN HEALTH RISKS AND EFFECTS ON THE ENVIRONMENT
10.1 Evaluation of human health risks
Humans are exposed to 1,1,1-trichloroethane principally by
inhalation, where the substance is rapidly absorbed into the body.
Exposure by skin absorption or ingestion may also occur but, compared
to inhalation, is of lesser concern. 1,1,1-Trichloroethane is
considered not to bioaccumulate. The acute and chronic toxicities are
fairly low. Under conditions of high exposure there is a risk of
toxic effects; such conditions may occur in occupational exposures,
solvent abuse or accidents. Since 1,1,1-trichloroethane is volatile
and the vapour is much more dense than air, toxic and explosive
concentrations may occur unexpectedly in confined spaces. This has
caused several fatal and near-fatal accident at workplaces and
elsewhere. The critical effect in humans relates to the central
nervous system, leading to unconsciousness and respiratory arrest at
high concentrations. 1,1,1-Trichloroethane is less toxic to the liver
than most other organochlorine solvents. The no-observed-effect level
for humans appears to be in the region of 1350 mg/m3 (250 ppm).
Although one study suggested effects at a lower level, the
interpretation of the results was unclear.
Studies on Mongolian gerbils involving 1,1,1-trichloroethane
levels down to 378 mg/m3 (70 ppm) revealed effects on DNA
concentrations and other biochemical effects in several brain regions.
If these findings are validated and confirmed, this may be of
significant concern.
No adequate study of carcinogenic effects on humans has been
published. A long-term inhalation study on rats and mice exposed to
8100 mg/m3 (1500 ppm) gave no evidence of any carcinogenic effects.
One study recorded increases numbers of "leukaemias", but it was
performed under non-standard protocol. It gave no statistical
evaluation and is, therefore, considered inadequate. It is unlikely
that 1,1,1-trichloroethane itself presents a significant risk of
genotoxic or carcinogenic effects in humans.
1,1,1-Trichloroethane is not teratogenic and elicits no
developmental effects at doses that are not maternally toxic. The
limited epidemiological evidence on reproductive effects is
inconclusive due to the low statistical power of the study.
10.2 Evaluation of effects on the environment
1,1,1-Trichloroethane is released in large quantities to the
environment. Due to its volatility and general stability it is a
ubiquitous environmental contaminant.
Although the ozone-depleting and global-warming potentials of
1,1,1-trichloroethane are lower than those of many
chlorofluorocarbons, the large-scale release of the compound into the
atmosphere merits serious concern in these respects.
As a result of its widespread use and disposal and its mobility
in soil, persistent 1,1,1-trichloroethane contamination of ground
water, including deep aquifers, occurs.
Conditions under which 1,1,1-trichloroethane causes
ecotoxicological effects are not found in the environment except after
spills.
11. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH AND
THE ENVIRONMENT
a) Emissions of 1,1,1-trichloroethane should be reduced as far as
is practicable in order to minimize exposure of workers and of
the general population.
b) The release of 1,1,1-trichloroethane into the environment must
be reduced to the greatest extent possible in order to avoid
damage to the ozone layer.
c) Contamination of ground water by 1,1,1-trichloroethane from
spills and waste disposal should be avoided.
d) Safe substitutes for 1,1,1-trichloroethane should be identified.
12. FURTHER RESEARCH
a) More study is needed to clarify the possible effects of
1,1,1-trichloroethane on the biochemistry of the central nervous
system and their significance.
b) More study is necessary to clarify the possible interaction
between 1,1,1-trichloroethane and ethanol in humans.
c) An adequate subchronic or long-term oral study is needed to
assess the hepatotoxicity of 1,1,1-trichloroethane.
d) Further epidemiological studies on reproductive effects are
desirable.
13. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
IARC (International Agency for Research on Cancer) working groups
have evaluated 1,1,1-trichloroethane and concluded that the evidence
for genotoxicity is limited (IARC, 1979, 1987).
REFERENCES
ABERNETHY, S., BOBRA, A.M., SHIU, W.Y., WELLS, P.G., & MACKAY, D.
(1986) Acute lethal toxicity of hydrocarbons and chlorinated
hydrocarbons to two planktonic crustaceans: The key role of
organism-water partitioning. Aquat. Toxicol., 8: 163-174.
ADAMS, E.M., SPENCER, H.C., ROWE, V.K., & IRISH, D.D. (1950) Vapor
toxicity of 1,1,1-trichloroethane (methyl chloroform) determined by
experiments on laboratory animals. Arch. ind. Hyg., 1: 225-236.
AGGAZZOTTI, G. & PREDIERI, G. (1986) Survey of volatile halogenated
organics (VHO) in Italy. Water Res., 20: 959-963.
ALEXANDER, H.C., MCCARTY, W.M., & BARTLETT, E.A. (1978) Toxicity of
perchloroethylene, trichloroethylene, 1,1,1-trichloroethane, and
methylene chloride to fathead minnow. Bull. environ. Contam. Toxicol.,
20: 344-352.
ALTHAUS, F.R., LAWRENCE, S.D., SATTLER, G.L., LONGFELLOW, D.G., &
PITOT, H.C. (1982) Chemical quantification of unscheduled DNA
synthesis in cultured hepatocytes as an assay for the rapid screening
of potential chemical carcinogens. Cancer Res., 42(8): 3010-3015.
AMI (1988) [The Factories Inspectorate's data base for workplace
measurements, ATABAS, stock-taking for the period January 1983 -
December 1987], Copenhagen, Institute for the Working Environment (in
Danish).
ANDERSON, H.R., DICK, B., MACNAIR, R.S., PALMER, J.C., & RAMSEY, J.D.
(1982) An investigation of 140 deaths associated with volatile
substance abuse in the United Kingdom (1971-1981). Hum. Toxicol., 1:
207-221.
ARCHER, W.L. (1979) 1,1,1-Trichloroethane. In: Kirk-Othmer
encyclopedia of chemical technology, 3rd ed., New York, Chichester,
John Wiley and Sons, pp. 728-731.
ARCHER, W.L. & STEVENS, V.L. (1977) Comparison of chlorinated,
aliphatic, aromatic, and oxygenated hydrocarbons as solvents. Ind.
Eng. Chem. Prod. Res. Dev., 16: 319-325.
ASTRAND, I., KILBOM, A., WAHLBERG, I., & OVRUM, P. (1973) Methyl
chloroform exposure. I. Concentration in alveolar air and blood at
rest and during exercise. Scand. J. Work Environ. Health, 10: 69-81.
BASS (1970) Sudden sniffing death. J. Am. Med. Assoc., 212: 2075-2079.
BAUER, U. (1981) [Burden on man caused by harmful substances in the
environment.] Zbl. Bakteriol. Hyg. I Abt. Orig. B, 174: 200-237 (in
German).
BERODE, M., BOILLAT, M.-A., GUILLEMIN, M.P., WU, M.W., & SAVOLAINEN,
H. (1990) Demethylation pathways in caffeine metabolism as indicators
of variability in 1,1,1-trichloroethane oxidation in man. Pharmacol.
Toxicol., 67(1): 41-46.
BOBRA, A.M., SHIU, W.Y., & MACKAY, D. (1984) Structure-activity
relationships for toxicity of hydrocarbons, chlorinated hydrocarbons
and oils to Daphnia magna. In: Kaiser, K.L.E., ed. Quantitative
structure activity relations in environmental toxicology, Dordrecht,
Germany, D. Reidel Publishing Co., pp. 3-16.
BONNET, P., FRANCIN, J.-M., GRADISKI, D., RAOULT, G., & ZISSU, D.
(1981) Determination of the median lethal concentration of the
principal chlorinated aliphatic hydrocarbons in the rat. Arch. Mal.
prof. Méd. Trav. Sécur. soc., 41: 317-321.
BONVENTRE, J., BRENNAN, O., JASON, D., HENDERSON, A., & BASTOS, M.L.
(1977) Two deaths following accidental inhalation of dichloromethane
and 1,1,1-trichloroethane. J. anal. Toxicol., 4: 158-160.
BOUWER, E.J. & MCCARTY, P.L. (1983a) Transformations of 1- and
2-carbon halogenated aliphatic organic compounds under methanogenic
conditions. Appl. environ. Microbiol., 45: 1286-1294.
BOUWER, E.J. & MCCARTY, P.L. (1983b) Transformations of halogenated
organic compounds under denitrification conditions. Appl. environ.
Microbiol., 45: 1295-1299.
BOYER, J.D, AHLERT, R.C., & KOSSON, D.S. (1987) Degradation of
1,1,1-trichloroethane in bench-scale bioreactors. Hazardous Waste
hazardous Matter, 4: 241-260.
BRETHERICK, L., ed. (1981) Hazards in the chemical laboratory, 3rd
ed., London, The Royal Society of Chemistry.
BRETHERICK, L. (1989) 1,1,1-Trichloroethane. Chem. Eng. News, Dec. 12:
2.
BRINGMANN, G. & KUHN, R. (1977) [Limit values for the harmful effect
of water-endangering substances on bacteria (Psuedomonas putida) and
green algae (Scenedesmus quadricauda) in the cell division
inhibition test.] Z. Wasser Abwasser Forsch., 10: 87-98 (in German).
BRINGMANN, G. & KUHN, R. (1978) [Limit values for the harmful effect
of water endangering substances on blue algae (Microcystis
aeroginosa) and green algae (Scenedesmus quadricauda) in the cell
division inhibition test.] Vom Wasser, 50: 45-60 (in German).
BUCCAFUSCO, R.J., ELLS, S.J., & LEBLANC, G.A. (1981) Acute toxicity of
priority pollutants to bluegill (Lepomis macrochirus). Bull.
environ. Contam. Toxicol., 26: 446-452.
CAPLAN, Y.H., BACKER, R.C., & WHITAKER, J.Q. (1976)
1,1,1-Trichloroethane: report of a fatal intoxication. Clin. Toxicol.,
9: 69-74.
CEC (Commission of the European Communities) (1986) Organochlorine
solvents: Health risks to workers, Cambridge, Royal Society of
Chemistry, pp. 57-58.
CEFIC (1986) The occurrence of chlorinated solvents in the
environment. Prepared by a workshop of the European Council of
Chemical Industry Federation (CEFIC). Chem. Ind., 1986: 861-869.
CHIOU, C.T., PETERS, L.J., & FREED, V.H. (1979) A physical concept of
soil-water equilibria for nonionic organic compounds. Science, 206:
831-832.
CLARK, D.G. & TINSTON, D.J. (1982) Acute inhalation toxicity of some
halogenated and non-halogenated hydrocarbons. Hum. Toxicol., 1:
239-247.
CLASS, T. & BALLSCHMITER, K. (1986) Chemistry of organic traces in air
VI: Distribution of chlorinated C1 - C4 hydrocarbons in air over the
Northern and Southern Atlantic ocean. Chemosphere, 15: 413-427.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology, 3rd revised ed., New York, Chichester, John Wiley and
Sons, Vol. 2B.
COLEMAN, W.E., LINGG, R.D., MELTON, R.G., & KOPFLER, F.C. (1976) The
occurrence of volatile organics in five drinking water supplies using
gas chromatography/mass spectrometry. In: Keith, L.H., ed.
Identification and analysis of organic pollutants in water, Ann Arbor,
Michigan, Ann Arbor Science Publishers, Inc., pp. 305-327.
CORREIA, Y., MARTENS, G.J., VAN MESCH, F.H., & WHIM, B.P. (1977) The
occurrence of trichloroethylene, tetrachloroethylene and
1,1,1-trichloroethane in western Europe in air and water. Atmos.
Environ., 11: 1113-1116.
DAFT, J.L. (1988) Fumigant contamination during large-scale food
sampling for analysis. Arch. environ. Contam. Toxicol., 17: 177-181.
DAHLBERG, J.A., CHRISTIANSEN, V.O., & ERIKSEN, E.A. (1973) On the
formation of phosgene by photooxidation of methyl chloroform in
welding. Ann. occup. Hyg., 16: 41-46.
DALLAS, C.E., RAMANATHAN, R., MURALIDHARA, S., GALLO, J.M., &
BRUCKNER, J.V. (1989) The uptake and elimination of
1,1,1-trichloroethane during and following inhalation in rats.
Toxicol. appl. Pharmacol., 98: 385-397.
DANIEL, M.R. & DEHNEL, J.M. (1981) Cell transformation test with baby
hamster kidney cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 626-637 (Progress in Mutation Research, Vol. 1).
DANIELSSON, B.R.G., GHANTOUS, H., & DENCKER, L. (1986) Distribution of
chloroform and methylchloroform and their metabolites in pregnant
mice. Biol. Res. Pregnancy, 7: 77-83.
DAPSON, S.C., HUTCHEON, D.E., & LEHR, D. (1984) Effect of methyl
chloroform on cardiovascular development in rats. Teratology, 29: 25A.
DE NEVERS, N. (1986) A fatal fire with "non-flammable" methyl
chloroform. Arch. environ. Health, 41(5): 279-287.
DE SERRES, F.J. & ASHBY, J., ed. (1981) Evaluation of short term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland (Progress in
Mutation Research, Vol. 1).
DEVER, R.J. (1986) Responding to industrial contamination of
groundwater: a case study. J. Am. Water Works Assoc., 78: 82-86.
DICKSON, A.G. & RILEY, J.P. (1976) The distribution of short-chain
halogenated aliphatic hydrocarbons in some marine organisms. Mar.
Pollut. Bull., 7: 167-169.
DILLING, W.L., TERERTILLER, N.B., & KALLOS, G.J. (1975) Evaporation
rates and reactivities of methylene chloride, chloroform,
1,1,1-trichloroethane, trichloroethylene, tetrachloroethylene, and
other chlorinated compounds in dilute aqueous solutions. Environ. Sci.
Technol., 9: 833-838.
DORNETTE, W.H.L. & JONES, J.P. (1960) Clinical experience with
1,1,1-trichloroethane. Anesthesiol. Analg., 39: 249-253.
DRAKE, R.J. (1987) Fate of methylene chloride and
1,1,1-trichloroethane in unsaturated, biologically active soils,
University of Massachusetts (Dissertation).
DREHER, E.-L. (1989) Chlorinated hydrocarbons. In: Ullmann's
encyclopedia of chemical technology, New York, Weinheim, VCH
Publishers, Inc., Vol. A6, pp. 271-272.
DROZ, P.O., WU, M.M., CUMBERLAND, W.G., & BERUDE, M. (1989)
Variability in biological monitoring of solvent exposure. I
Development of a population physiological model. Br. J. ind. Med., 46:
447-460.
DUPRAT, P., DELSAUT, L., & GRADISKI, D. (1976) Pouvoir irritant des
principaux solvants chlorés aliphatiques sur la peau et les muqueuses
oculaires du lapin. Eur. J. Toxicol. environ. Hyg., 9: 171-177.
DÜSZELN, J.V., VON, LAHL, U., BÄTZER, K., CETINKAYA, M., STACHEL, B.,
& THIEMANN, W. (1982) [Volatile halogenated hydrocarbons in air, water
and food in the Federal Republic of Germany.] Dtsch. Lebensm.
Rundsch., 78: 352-356 (in German).
DÜSZELN, J.V., VON & THIEMANN, W. (1985) Volatile chlorinated
hydrocarbons in a costal urban atmosphere. Sci. total Environ., 41:
187-194.
EBEN, A. & KIMMERLE, G. (1974) Metabolism, excretion and toxicology of
methylchloroform in acute and subacute exposed rats. Arch. Toxicol.,
31: 233-242.
EDWARDS, P.R., CAMPBELL, I., & MILNE, G.S. (1982) The impact of
chloromethanes on the environment. Part 1. The atmospheric chlorine
cycle. Chem. Ind., 16: 574-578.
ELOVAARA, E., HEMMINKI, K., & VAINIO, H. (1979) Effects of methylene
chloride, trichloroethane, trichloroethylene and toluene on the
development of chick embryos. Toxicology, 12: 111-119.
ENTZ, R.C. & DIACHENKO, G.W. (1988) Residues of volatile halocarbons
in margarines. Food Addit. Contam., 5: 267-276.
ENTZ, R.C., THOMAS, K.W., & DIACHENKO, G.W. (1982) Residues of
volatile halocarbons in foods using headspace gas chromatography. J.
agric. food Chem., 30: 846-849.
FDA (1973) Trichloroethane. Aerosol drug products for human use. Fed.
Reg., 38: 21935-21936.
FDA (1977) Trichloroethane: status as a new drug in aerosolized drug
products intended for inhalation. FDA Final Rule. Fed. Reg., 42:
63386-63387.
FIELDING, M., GIBSON, T.M., JAMES, H.A., MCLOUGHLIN, K., & STEEL, C.P.
(1981) Organic micropollutants in drinking water, Medmenham, United
Kingdom, Water Research Centre (Technical Report No. TR 159).
FISCHER, M., FRIESEL, P., KOCH, G., MILDE, G., & ROSSKAMP, E. (1982)
Evaluation of water pollution caused by chlorinated and brominated
ethanes (Unpublished report submitted to the Commission of the
European Communities under Contract No. U71/516).
FISHBEIN, L. (1979) Potential halogenated industrial carcinogenic and
mutagenic chemicals. II. Halogenated saturated hydrocarbons. Sci.
total Environ., 11: 163-195.
FISHER, D.A., HALES, C.H., FILKIN, D.L., KO, M.K.W., SZE, N.D.,
CONNELL, P.S., WUEBBLES, D.J., ISAKSEN, I.S.A., & STORDAL, F. (1990a)
Model calculations of the relative effects of CFCs and their
replacement on stratospheric ozone. Nature (Lond.), 344: 508-512.
FISHER, D.A., HALES, C.H., WANG, W.-C., KO, M.K.W., & SZE, N.D.
(1990b) Model calculations of the relative effects of CFCs and their
replacement on global warming. Nature (Lond.), 344: 513-516.
FLUKA (1988) Catalogue 1988/89: Fluka Chemika-BioChemika, Buchs,
Switzerland, Fluka Chemie AG.
GALLOWAY, S.M., ARMSTRONG, M.J., REUBEN, C., COLMAN, S., BROWN, B.,
CANNON, C., BLOOM, A.D., NAKAMURA, F., AHMED, M., DUK, S. ET AL.
(1987) Chromosome aberrations and sister chromatid exchanges in
Chinese hamster ovary cells: Evaluations of 108 chemicals. Environ.
mol. Mutagen., 10: 1-175.
GAMBERALE, F. & HULTENGREN, M. (1973) Methylchloroform exposure II.
Psychophysiological functions. Scand. J. Work Environ. Health, 10:
82-92.
GEORGE, J.D., PRICE, C.J., MARR, M.C., SADLER, B.M., SCHWETZ, B.A.,
BIRNBAUM, L.S., & MORRISEY, R.E. (1989) Developmental toxicity of
1,1,1-trichloroethane in CD rats. Fundam. appl. Toxicol., 13, 641-651.
GERACE, R.V. (1981) Near-fatal intoxication by 1,1,1-trichloroethane.
Ann. Emergency Med., 10: 533-534.
GERKENS, R.R. & FRANKLIN, J.A. (1989) The rate of degradation of
1,1,1-trichloroethane in water by hydrolysis and dehydrochlorination.
Chemosphere, 19: 1919-1937.
GIGER, W., MOLNAR-KUBICA, E., & WAKEHAM, S. (1978) Volatile
chlorinated hydrocarbons in ground and lake waters. In: Aquatic
pollutants: Transformation and biological effects. Proceedings of the
2nd International Symposium on Aquatic Pollutants, Oxford, New York,
Pergamon Press, pp. 101-123 (Pergamon Series on Environmental Science
No. 1).
GOCKE, E., KING, M.-T., ECKHARDT, K., & WILD, D. (1981) Mutagenicity
of cosmetics ingredients licensed by the European Communities. Mutat.
Res., 90: 91-109.
GOLDBERG, S.J., LEBOWITZ, MD., GRAVER, E.J., & HICKS, S. (1990) An
association of human congenital cardiac malformations and
drinking-water contaminants. J. Am. Coll. Cardiol., 16: 155-164.
GOSSETT, J.M. (1985) Anaerobic degradation of C1 and C2 chlorinated
hydrocarbons, Tyndall AFB, Florida, US Air Force Engineering Services
Center (Technical Report No. ESL-TR-85-38).
GOSSETT, R.W., BROWN, D.A., & YOUNG, D.R. (1983) Predicting the
bioaccumulation of organic compounds in marine organisms using
octanol/water partition coefficients. Mar. Pollut. Bull., 14: 387-392.
GOTO, M. (1979) Monitoring environmental materials and specimen
banking. State-of-the-art of Japanese experience and knowledge. In:
Monitoring environmental materials and specimen banking, Proc. Int.
Workshop, Berlin (West) 23-28 October 1978, The Hague, Nijhoff
Publishers, pp. 271-288.
GRADISKI, D., MAGADUR, J.-L., BAILLOT, M., DANIERE, M.C., & SCHUH,
M.B. (1974) Toxicité comparée des principaux solvants chlorés
aliphatiques. J. eur. Toxicol., 7: 247-254.
GRADISKI, D., BONNET, P., RAOULT, G., MAGADUR, J., & FRANCIN, J.M.
(1978) Compared acute pulmonary toxicity of the main chlorinated
aliphatic solvents. Arch. Mal. prof. Méd. Trav. Sécur. soc., 39:
249-257.
GREEN, M.H. (1981) Differential killing test using an improved
repair-deficient strain of Escherichia coli. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
Oxford, Elsevier/North Holland, pp. 183-194 (Progress in Mutation
Resarch, Vol. 1).
GRIMSRUD, E.P. & RASMUSSEN, R.A. (1975) Survey and analysis of
halocarbons in the atmosphere by gas chromatography-mass spectrometry.
Atmos. Environ., 9: 1014-1017.
HAAG, W.R. & MILL, T. (1988) Effect of a subsurface sediment on
hydrolysis of haloalkanes and epoxides. Environ. Sci. Technol., 22:
658-663.
HAAG, W.R., MILL, T., & RICHARDSON, A. (1986) Extended abstracts of
the 192nd National Meeting of the American Chemical Society, Anaheim,
Washington, DC, American Chemical Society, Division of Environmental
Chemistry, Vol. 26, pp. 248-253.
HAKE, C.L., WAGGONER, T.B., ROBERTSON, D.N., & ROWE, V.K. (1960) The
metabolism of 1,1,1-trichloroethane by the rat. Arch. environ. Health,
1: 101-105.
HALL, F.B. & HINE, C.H. (1966) Trichloroethane intoxication: a report
of two cases. J. Forensic Sci., 11: 404-413.
HARDIE, D.W.F. (1964) 1,1,1-Trichloroethane. In: Kirk, R.E. & Othmer,
D.F., ed. Encyclopedia of chemical technology, 2nd ed., New York,
Chichester, Wiley and Sons, Vol. 5, pp. 154-157, 170.
HATCH, G.G., MAMAY, P.P., AYER, M.L., CASTO, B.C., & NESNOW, S. (1983)
Chemical enhancement of viral transformation in Syrian hamster embryo
cells by gaseous volatile chlorinated methanes and ethanes. Cancer
Res., 43: 1945-1950.
HATFIELD, T.R. & MAYKOSKI, R.T. (1970) A fatal methyl chloroform
(trichloroethane) poisoning. Arch. environ. Health, 20: 279-281.
HEITMULLER, P.T., HOLLISTER, T.A., & PARRISH, P.R. (1981) Acute
toxicity of 54 industrial chemicals to sheepshead minnows (Cyprinodon
variegatus) Bull. environ. Contam. Toxicol., 27: 596-604.
HENSCHLER, D., ed. (1978) [German Association for the Encouragement of
Research: Analytical methods for the testing of materials harmful to
health. I. Air analyses], Weinheim, Verlag Chemie (in German).
HENSCHLER, D., REICHERT, D., & METZLER, M. (1980) Identification of
potential carcinogens in technical grade 1,1,1-trichloroethane. Int.
Arch. occup. environ. Health, 47: 263-268.
HERD, P.A., LIPSKY, M., & MARTIN, H.F. (1974) Cardiovascular effects
of 1,1,1-trichloroethane. Arch. environ. Health, 28: 227-233.
HOBARA, T., KOBAYASHI, H., HIGASHIHARA, E., IWAMOTO, S., KAWAMOTO, T.,
SAKAI, T., & TSUBOTA, N. (1982) [Experimental examinations and
toxicokinetic analysis of the absorption and excretion of
1,1,1-trichloroethane by the lung.] Jpn. J. ind. Health, 24: 599-607
(in Japanese).
HODGSON, M.J., HEYL, A.E., & VAN THIEL, D.H. (1989) Liver disease
associated with exposure to 1,1,1-trichloroethane. Arch. intern. Med.,
149: 1791-1798.
HORIGUCHI, S. & HORIGUCHI, K. (1971) An experiment of
1,1,1-trichloroethane vapour exposure to mice. Sangyo Igaku, 13:
226-227.
HSE (1984) 1,1,1-Trichloroethane, London, Health and Safety Executive.
HSE (1990) Guidance note EH40/90. Occupational exposure limits 1990,
London, Health and Safety Executive.
HUMBERT, B.E. & FERNANDEZ, J.G. (1977) Exposition au
1,1,1-trichloroéthane: Contribution à l'étude de l'absorption, de
l'excrétion et du métabolisme sur des sujets humains. Arch. Mal.
prof., 38: 415-425.
HUTCHINSON, T.C., HELLEBUST, J.A., TAM, D., MACKAY, D., MASCARENHAS,
R.A., & SHIU, W.Y. (1980) The correlation of the toxicity to algae of
hydrocarbons and halogenated hydrocarbons with their physical-chemical
properties. Environ. Sci. Res., 1: 577-586.
IARC (1979) 1,1,1-Trichloroethane. In: Some halogenated hydrocarbons,
Lyon, International Agency for Research on Cancer, pp. 515-532 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Vol. 20).
IARC (1987) Overall evaluations of carcinogenicity: An updating of
IARC Monographs, Volumes 1 to 42, Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluations of Carcinogenic
Risk to Humans, Suppl. 7).
ICHINOTSUBO, D., MOWER, H., & MANDEL, M. (1981) Mutagen testing of a
series of paired compounds with the Ames Salmonella testing system.
In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term tests
for carcinogens. Report of the international collaborative programme,
New York, Amsterdam, London, Elsevier/North Holland, pp. 298-301
(Progress in Mutation Research. Vol. 1).
IKEDA, M. & OHTSUJI, H. (1972) A comparative study of the excretion of
Fujiwara reaction-positive substances in urine of humans and rodents
given trichloro- or tetrachloro-derivatives of ethane and ethylene.
Brit. J. ind. Med., 29: 99-104.
IVANETICH, K.M. & HONERT, L.H., VAN DEN (1981) Chloroethanes: their
metabolism by hepatic cytochrome P-450. Carcinogenesis, 2: 697-702
JAKOBSON, M., LIEPINS, M., & ERINS, P. (1980) [Effect of changes in
the reticular structure of a lignocarbohydrate matrix on the
properties of the wood. 2. Effect of cross-linking on the
hydrophilicity of birchwood.] Khim. Drev., 1: 37-46 (in Russian).
JAKOBSON, I., WAHLBERG, J.E., HOLMBERG, B., & JOHANSSON, G. (1982)
Uptake via the blood and elimination of 10 organic solvents following
epicutaneous exposure of anesthetized guinea-pigs. Toxicol. appl.
Pharmacol., 63: 181-187.
JONES, R.D. & WINTER, D.P. (1983) Two case reports of deaths on
industrial premises attributed to 1,1,1-trichloroethane. Arch.
environ. Health, 38: 59-61.
KADA, T. (1981) DNA-damaging activity of 42 coded compounds in the
rec-assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 175-182 (Progress in Mutation Research, Vol. 1).
KAJINO, M. & YAGI, M. (1980) Formation of trihalomethanes during
chlorination and determination of halogenated hydrocarbons in drinking
water. In: Hydrocarbons and halogenated hydrocarbons in the aquatic
environment, New York, London, Plenum Press.
KARLSSON, J.-E., ROSENGREN, L.E., KJELLSTRAND, P., & HAGILD K.G.
(1987) Effect of low-dose inhalation of three chlorinated aliphatic
organic solvents on DNA in gerbil brain. Scand. J. Work Environ.
Health, 13: 453-458.
KHALIL, M.A. & RASMUSSEN, R.A. (1981) Decline in the atmospheric
accumulation rates of CCl3F(F-11), CCl2F2(F-12) and CH3CCl3.
J. Air Pollut. Control Assoc., 31: 1274-1275.
KHALIL, M.A. & RASMUSSEN, R.A. (1984) Methylchloroform: Global
distribution, seasonal cycles and anthropogenic chlorine. Chemosphere,
13: 789-800.
KING, G.S., SMIALEK, J.E., & TROUTMAN, W.G. (1985) Sudden death in
adolescent resulting from the inhalation of typewriter correction
fluid. J. Am. Med. Assoc., 253: 1604-1606.
KLAASSEN, C.D. & PLAA, G.L. (1966) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in mice.
Toxicol. appl. Pharmacol., 9: 139-151.
KLAASSEN, C.D. & PLAA G.L. (1967) Relative effects of various
chlorinated hydrocarbons on liver and kidney function in dogs.
Toxicol. appl. Pharmacol., 10: 119-131.
KLECKA, G.M., GONSIOR, S.J., & MARKHAM, D.A. (1990) Biological
transformations of 1,1,1-trichloroethane in subsurface soils and
ground water. Environ. Toxicol. Chem., 9: 1437-1451.
KLEINFELD, M. & FEINER, B. (1966) Health hazard associated with work
in confined spaces. J. occup. Med., 8: 358-364.
KOBAYASHI, H., HOBARA, T., HIROTA, H., HIGASHIHARA, E., IWAMOTO, S.,
& SAKAI, T. (1982) Sensitization of dog myocardium to epinephrine by
1,1,1-trichloroethane. Jpn. J. ind. Health, 24: 450-454.
KOBAYASHI, H., HOBARA, T., HIROTA, H., & SAKAI, T. (1983) Neural
control of blood pressure following 1,1,1-trichloroethane inhalation:
A role of sympathetic nervous system. Arch. environ. Health, 38:
93-98.
KOBAYASHI, H., HOBARA, T., SATOH, T., & SAKAI, T. (1986) Respiratory
disorders following 1,1,1-trichloroethane inhalation: A role of reflex
mechanism arising from lungs. Arch. environ. Health, 41: 149-154.
KOBAYASHI, H., HOBARA, T., KAWAMOTO, T., & SAKAI, T. (1987) Effect of
1,1,1-trichloroethane inhalation on heart rate and its mechanism: A
role of autonomic nervous system. Arch. environ. Health, 42: 140-143.
KONEMANN, H. (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part I. Relationship for 50 industrial
pollutants. Toxicology, 19: 209-221.
KRAMER, C.G., OTT, M.G., FULKERSON, J.E., & HICKS, N. (1978) Health of
workers exposed to 1,1,1-trichloroethane: A matched-pair study. Arch.
environ. Health, 33: 331-342.
KRONELD, R. (1989) Volatile pollutants in the environment and human
tissues. Bull. environ. Contam. Toxicol., 42: 873-877.
KRONEVI, T., WAHLBERG, J.E., & HOLMBERG, B. (1981) Skin pathology
following epicutaneous exposure to seven organic solvents. Int. J.
Tissue React., 3: 21-30.
KUKONGVIRIYAPAN, V., KUKONGVIRIYAPAN, U., & STACEY, N.H. (1990)
Interference with hepatocellular substrate uptake by
1,1,1-trichloroethane and tetrachloroethylene. Toxicol. appl.
Pharmacol., 102: 80-90.
KYRKLUND, T., KJELLSTRAND, P., & HAGLID, K.G. (1988) Effects of
exposure to Freon 11, 1,1,1-trichloroethane or perchloroethylene on
the lipid and fatty-acid composition in rat cerebral cortex. Scand. J.
Work Environ. Health, 14: 91-94.
LANE, R.W., RIDDLE, B.L., & BORZELLECA, J.F. (1982) Effects of
1,2-dichloroethane and 1,1,1-trichloroethane in drinking water on
reproduction and development in mice. Toxicol. appl. Pharmacol., 63:
409-421.
LEBLANC, G.A. (1980) Acute toxicity of priority pollutants to water
flea (Daphnia magna). Bull environ. Contam. Toxicol., 24: 684-691.
LEONG, B.K.J., SCHWETZ, B.A., & GEHRING, P.J. (1975) Embryo- and
fetotoxicity of inhaled trichloroethylene, perchloroethylene,
methylchloroform, and methylene chloride in mice and rats. Toxicol.
appl. Pharmacol., 33: 136 (Abstract 34).
LILLIAN, D., SINGH, H.B., APPLEBY, A., LOBBAN, L., ARNTS, R., GUMPERT,
R., HAGUE, R., TOOMEY, J., KAZASIS, J., ANTELL, M., HANSEN, D., &
SCOTT, B. (1975) Atmospheric fates of halogenated compounds. Environ.
Sci. Technol., 9: 1043-1048.
LINDBOHM, M-L., TASKINEN, H., SALLMEN, M., & HEMMINKI, K. (1990)
Spontaneous abortions among women exposed to organic solvents. Am. J.
ind. Med., 17: 449-463.
LOPRIENO, N., BARATE, R., ROSSI, A.M., FUMERO, S., MERIGGI, G.,
MONDINO, A., & SILVEST S. (1979) In vivo mutagenicity studies with
trichloroethylene and other solvents, Ivrea, Italy, Istituto di
Ricerche Biomedicale
LUNDBERG, I., EKDAHL, M., KRONEVI, T., LIDUMS, V., & LUNDBERG, S.
(1986) Relative hepatotoxicity of some industrial solvents after
intraperitoneal injection or inhalation exposure in rats. Environ.
Res., 40: 411-420.
MABEY, W., BARICH, V., & MILL, T. (1983) Extended abstracts of the
186th Meeting of the American Chemical Society, Washington, DC,
American Chemical Society, Division of Environmental Chemistry, Vol.
23, pp. 359-361.
MCCARTHY, T.B. & JONES, R.D. (1983) Industrial gassing poisonings due
to trichloroethylene, perchlorethylene, and 1,1,1-trichloroethane,
1961-80. Br. J. ind. Med., 40: 450-455.
MCCONNELL, G., FERGUSON, D.M., & PEARSON, C.R. (1975) Chlorinated
hydrocarbons and the environment. Endeavour, 34: 13-18.
MCCONNELL, J.C. & SCHIFF, H.I. (1978) Methyl chloroform: Impact on
stratospheric ozone. Science, 199: 174-177.
MCDONALD, D.J. (1981) Salmonella/microsome tests on 42 coded
chemicals. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 285-297 (Progress in Mutation Research, Vol. 1).
MCEWEN, J.D. & VERNOT, E.H. (1974) The biological effect of continuous
inhalation exposure of 1,1,1-trichloroethane (methyl chloroform) on
animals. In: Toxic hazard research unit: Annual technical report 1974,
Ohio, Wright-Patterson Air-Force Base, Aerospace Medical Research
Laboratory, pp. 81-90 (AMRL-TR-74-78).
MACKAY, C.J., CAMPBELL, L., SAMUEL, A.M., ALDERMAN, K.J., IDZIKOWSKI,
C., WILSON, H.K., & COMPERTZ, D. (1987) Behavioral changes during
exposure to 1,1,1-trichloroethane: time-course and relationship to
blood solvents levels. Am. J. ind. Med., 11: 223-239.
MCLEOD, A.A., MARJOT, A., MONAGHAN, M.J., HUGH-JONES, P., & JACKSON,
G. (1987) Chronic cardiac toxicity after inhalation of
1,1,1-trichloroethane. Br. med. J., 214: 727-729.
MCNUTT, N.S., AMSTER, R.L., MCCONNELL, E.F., & MORRIS, F (1975)
Hepatic lesions in mice after continuous inhalation exposure to
1,1,1-trichloroethane. Lab. Invest., 5: 642-654.
MAGARA, Y. & FURUICHI, T. (1986) Environmental pollution by
trichloroethylene and tetrachloroethylene: a nationwide survey. In:
Chambers, P.L., Gehring, P., & Sakai, F., ed. New concepts and
developments in toxicology, Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp. 231-243.
MALTONI, C., COTTI, G., & PATELLA, V. (1986) Results of long-term
carcinogenicity bioassays on Sprague-Dawley rats of methyl chloroform,
administered by ingestion. Acta oncol., 7: 101-117.
MARONI, M.C., BULGGHERONI, C., CASSITTO, M.G., MERLUZZI, F., GILIOLO,
R., & FOA, V. (1977) A clinical, neurophysiological and behavioral
study of female workers exposed to 1,1,1-trichloroethane. Scand. J.
Work Environ. Health, 3: 16-22.
MARJOT, R. & MCLEOD, A.A. (1989) Chronic non-neurological toxicity
from volatile substance abuse. Hum. Toxicol., 8: 301-306.
MARTIN, C.N. & MCDERMID, A.C. (1981) Testing of 42 coded compounds for
their ability to induce unscheduled DNA repair synthesis in HeLa
cells. In: de Serres, F.J. & Ashby, J., ed. Evaluation of short-term
tests for carcinogens. Report of the international collaborative
programme, New York, Amsterdam, Oxford, Elsevier/North Holland, pp.
533-537 (Progress in Mutation Research, Vol. 1).
MIDGLEY, P.M. (1989) The production and release to the atmosphere of
1,1,1-trichloroethane (methyl chloroform). Atmos. Environ., 23:
2663-2665.
MILLER, L.J. & UHLER, A.D. (1988) Volatile halocarbons in butter:
Elevated tetrachloroethylene levels in samples obtained in close
proximity to dry-cleaning establishments. Bull. environ. Contam.
Toxicol., 41: 469-474.
MITI (1990) The yearbook of chemical trade and industries statistics,
Tokyo, Ministry of International Trade and Industry.
MITOMA, C., STEEGER, T., JACKSON, S.E., WHEELER, K.P., ROGERS, J.H.,
& MILMAN, H.A. (1985) Metabolic disposition study of chlorinated
hydrocarbons in rats and mice. Drug chem. Toxicol., 8(3): 183-194.
MONSTER, A.C. (1979) Differences in uptake, elimination and metabolism
in exposure to trichloroethylene, 1,1,1-trichloroethane and
tetrachloroethylene. Int. Arch. occup. environ. Health, 42: 311-317.
MONSTER, A.C. (1986) Biological monitoring of chlorinated hydrocarbon
solvents. J. occup. Med., 28: 583-588.
MONSTER, A.C. & HOUTKOPPER, J.M. (1979) Estimation of individual
uptake of trichloroethylene, 1,1,1-trichloroethane, and
tetrachloroethylene from biological parameters. Int. Arch. occup.
environ. Health, 42: 319-323.
MONSTER, A..C., BOERSMA, G., & STEENWEG, H. (1979) Kinetics of
1,1,1-trichloroethane in volunteers: influence of exposure
concentration and work load. Int. Arch. occup. environ. Health, 42:
293-301.
MOSER, V.C. & BALSTER, R.L. (1985) Acute motor and lethal effects of
inhaled toluene, 1,1,1-trichloroethane, halothane, and ethanol in
mice: effects of exposure duration. Toxicol. appl. Pharmacol., 77:
285-291.
MULLIN, L.S. & KRIVANEK, N.D. (1982) Comparison of unconditioned
reflex and conditioned avoidance tests in rats exposed by inhalation
to carbon monoxide, 1,1,1-trichloroethane, toluene or ethanol.
Neurotoxicology, 3: 126-137.
MYHR, B.C. & CASPARY, W.J. (1988) Evaluation of the L5178Y mouse
lymphoma cell mutagenesis assay: Intralaboratory results for
sixty-three coded chemicals tested at Litton Bionetics, Inc. Environ.
mol. Mutagen., 13: 103-194.
NAGAO, M. & TAKAHASHI, Y. (1981) Mutagenic activity of 42 coded
compounds in the salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 302-313 (Progress in Mutation
Research, Vol. 1).
NAKAAKI, K., FUKABORI, S., & TADA, O. (1980) An experimental study on
percutaneous absorption of some organic solvents. J. Sci. Labour, 56:
1-9.
NCI (1977) Bioassay of 1,1,1-trichloroethane for possible
carcinogenicity, Bethesda, Maryland, National Cancer Institute
(Carcinogenesis Technical Report Series No. 3).
NEELY, W.B. (1982) Organizing data for environmental studies. Environ.
Toxicol. Chem., 1: 259-266.
NESTMANN, E.R., LEE, E.G.H., MATULA, T.I. DOUGLAS, G.R., & MUELLER,
J.C. (1980) Mutagenicity of constituents identified in pulp and paper
mill effluents using the Salmonella/mammalian-microsome assay.
Mutat. Res., 79: 203-212.
NEUHAUSER, E.F., LOEHR, R.C., & MALECKI, M.R. (1986) Contact and
artificial soil tests using earthworms to evaluate the impact of
wastes in soil. In: Petros, J.K., Lacy, W.J., & Conway, R.A., ed.
Hazardous and industrial solid waste testing: Fourth Symposium.
Philadelphia, American Society for Testing and Materials, pp. 192-203
(ASTM STP 886).
NIEF, L. & MERCIER, J. (1981) Skin pathology following epicutaneous
exposure to seven organic solvents. Int. J. Tissue React., 111: 21-30.
NILSSON, K.B. (1986a) Effects of 1,1,1-trichloroethane on the cGMP
metabolism in mouse brain. Acta pharmacol. toxicol., 58: 318-326.
NILSSON, K.B. (1986b) Actions of 1,1,1-trichloroethane on the cAMP
metabolism in mouse brain. Acta pharmacol. toxicol., 59: 362-369.
NILSSON, K.B. (1987) Effects of 1,1,1-trichloroethane on synaptosomal
calcium accumulation in mouse brain. Pharmacol. Toxicol., 61: 215-219.
NIOSH (1976) Criteria for a recommended standard: occupational
exposure to 1,1,1-trichloroethane (methyl chloroform), Cincinnati,
Ohio, National Institute for Occupational Safety and Health
(Publication DHEW (NIOSH) No. 76-184).
NIOSH (1977) NIOSH manual of analytical methods. Part II: Standards
completion program validated methods (Method 5328), 2nd ed.,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, pp. 5328/1-5328/9 (DHEW (NIOSH) Publication No. 77-157-B).
NIOSH (1979) Occupational exposure to 1,1,1-trichloroethane,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health.
NIOSH (1983) National occupational exposure survey, 1981-1983,
Cincinnati, Ohio, National Institute for Occupational Safety and
Health, Division of Surveillance, Hazard Evaluations and Field
Studies, Surveillance Branch, Hazard Section (Unpublished data).
NOLAN, R.J., FRESHOUR, N.L., RICH, D.L., MCCARTY, L.P., & SAUNDERS,
J.H. (1984) Kinetics and metabolism of inhaled methyl chloroform
(1,1,1-trichloroethane) in male volunteers. Fundam. appl. Toxicol., 4:
654-662.
NORTHFIELD, R.R. (1981) Avoidable deaths due to acute exposure to
1,1,1-trichloroethane. J. Soc. Occup. Med., 31: 164-166.
NRC (1976) Halocarbons. Effects on stratospheric ozone, Washington,
DC, National Research Council, Panel on Atmospheric Chemistry. OTSON,
R. (1987) Purgeable organics in Great Lakes raw and treated water.
Int. J. environ. anal. Chem., 31: 41-53.
OTSON, R. & CHAN, C. (1987) Sample handling and analysis for 51
volatile organics by an adapted purge and trap GC-MS technique. Int.
J. environ. anal. Chem., 30: 275-287.
OTSON, R., WILLIAMS, D.T., & BOTHWELL, P.D. (1984) Fabric protectors.
Part II - Propane, 1,1,1-trichloroethane and petroleum distillates in
air after application of fabric protectors. Am. Ind. Hyg. Assoc. J.,
45: 28-33.
PARSONS, F. & LAGE, G.B. (1985) Chlorinated organics in simulated
groundwater environments. J. Am. Water Works Assoc., 77: 52-59.
PARSONS, F., LAGE, G.B., & RICE, R. (1985) Biotransformation of
chlorinated organic solvents in static microcosms. Environ. Toxicol.
Chem., 4: 739-742.
PEARSON, C.R. (1982) C1 and C2 halocarbons. In: Hutzinger, O., ed.
The handbook of environmental chemistry, Berlin, Heidelberg, New York,
Springer-Verlag, Vol. 3, Part B, pp. 69-88.
PEARSON, C.R. & MCCONNELL, G. (1975) Chlorinated C1 and C2
hydrocarbons in the marine environment. Proc. R. Soc. Lond., B189:
305-332.
PEKARI, K. & AITIO, A. (1985) Method 26. Determination of
1,1,1-trichloroethane in blood. In: Fishbein, L. & O'Neil, I.K., ed.
Environmental carcinogens selected methods of analysis. Volume 7 -
Some volatile halogenated hydrocarbons, Lyon, International Agency for
Research on Cancer, pp. 445-450 (IARC Scientific Publications No. 68).
PELLIZARI, E.D., HARTWELL, T.D., HARRIS, B.S.H., WADDELL, R.D.,
WHITAKER, D.A., & ERICKSON, M.D. (1982) Purgeable organic compounds in
mothers' milk. Bull. environ. Contam. Toxicol., 28: 322-328.
PEREIRA, W.E. & HUGHES, B.A. (1980) Determination of selected volatile
organic priority pollutants in water by computerized gas
chromatography - quadruple mass spectrometry. J. Am. Water Assoc., 72:
220-230.
PERRY, P.E. & THOMSON, E.J. (1981) Evaluation of the sister-chromatid
exchange method in mammalian cells as a screening system for
carcinogens. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 560-569 (Progress in Mutation Research, Vol. 1).
PFANNHAUSER, W., GOMBOS, J., & THALLER, A. (1988) [Halogenated
aliphatic hydrocarbons in food: Investigations on enrichment and
content in fat containing food.] Ernährung, 12: 611-615 (in German).
PIET, G.J., SLINGERLAND, P., DE GRUNT, F.E., HEUVEL, V.D., & ZOETEMAN,
B.C.J. (1978) Determination of very volatile halogenated organic
compounds in water by means of direct head-space analysis. Anal.
Lett., 11: 437-448.
PRENDERGAST, J.A., JONES, R.A., JENKINS, L.J., & SIEGEL, J. (1967)
Effects on experimental animals of long-term inhalation of
trichloroethylene, carbon tetrachloride, 1,1,1-trichloroethane,
dichlorodifluoromethane, and 1,1-dichloroethylene. Toxicol. appl.
Pharmacol., 10: 270-289.
PRICE, P.J., HASSETT, C.M., & MANSFIELD, J.I. (1978) Transforming
activities of trichloroethylene and proposed industrial alternatives.
14: 290-293.
PRINN, R., CUNNOLD, D., RASMUSSEN, R., SIMMONDS, P., ALYEA, F.,
CRAWFORD, A., FRASER, P., & ROSEN, R. (1987) Atmospheric trends in
methylchloroform and the global average for the hydroxyl radical.
Science, 238: 945-950.
PRODI, G., COLLACCI, A., GRILLI, S., LATTANZI, G., MAZZULO, M., &
TURINA P. (1988) Comparison of the covalent binding of various
chloroethanes with nucleic acids. In: Feo, F., ed. Chemical
carcinogenesis (models and mechanisms), New York, Plenum Press, pp.
93-102.
QUAST, J.F., RAMPY, L.W., BALMER, M.F., LEONG, B.D.J., & GEHRING P.J.
(1978) Toxicological and carcinogenic evaluation of a
1,1,1-trichloroethane formulation by chronic inhalation in rats,
Midland, Michigan, Dow Chemical Co.
QUAST, J.F., CALHOUN, L.L., & FRAUSON, L.E. (1988)
1,1,1-Trichloroethane formulation: a chronic inhalation toxicity and
oncogenicity study in Fischer 344 rats and B6C3F1 mice. Fundam. appl.
Toxicol., 11: 611-625.
RAJENDRAN, S. (1990) The toxicity of phosphine, methyl bromide,
1,1,1-trichloroethane and carbon dioxide alone and as mixtures to the
pupae of red flour beetle, Tribolium castaneum, Herbst. Pestic.
Sci., 29: 75-83.
RAMPY, L.W., QUAST, J.F., LEONG, B.K.J., & GEHRING, P.J. (1977)
Results of long-term inhalation studies on rats of
1,1,1-trichloroethane and perchloroethylene formulations. In: Plaa,
G.L. & Duncan, W.A.M., ed. Proceedings of the 1st International
Congress on Toxicology, Toronto, New York, London, Academic Press, p.
27 (Abstract).
RASMUSSEN, R.A. & KHALIL, M.A.K. (1982) Latitudinal distributions of
trace gases in and above the boundary layer. Chemosphere, 11: 117-235.
RASMUSSEN, R.A. & KHALIL, M.A.K. (1983) Natural and anthropogenic
trace gases in and above the boundary layer. Chemosphere, 12: 371-375.
RASMUSSEN, R.A., KHALIL, M.A.K., & DALLAGUE, R.W. (1981) Atmospheric
trace gases in Antarctica. Science, 211: 185-287.
REINHARDT, C.F., MULLIN, L.S., & MAXFIELD, M.E. (1973)
Epinephrine-induced cardiac arrhythmia potential of some common
industrial solvents. J. occup. Med., 15: 953-955.
REITZ, R.H., MCDOUGAL, J.N., HIMMELSTEIN, M.W., NOLAN, R.J., &
SCHUMANN, A.M. (1988) Physiologically based pharmacokinetic modelling
with methylchloroform: Implications for interspecies, high dose/low
dose and dose route extrapolations. Toxicol. appl. Pharmacol., 95:
185-199.
RENNICK, R.B., MALTON, S.D., MOW, G.K., & SEEVERS, M.H. (1949)
Induction of idioventricular rhythms by 1,1,1-trichloroethane and
epinephrine. J. Pharmacol. exp. Ther., 94: 327.
RICHOLD, M. & JONES, E. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluations of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 314-322 (Progress in Mutation
Research, Vol. 1).
RIIHIMAKI, V. & PFAFFLI, P (1978) Percutaneous absorption of solvent
vapor in man. Scand. J. Work Environ. Health, 4: 73-85.
RIVETT, M.O., LERNER, D.N., & LLOYD, J.W. (1990) Chlorinated solvents
in U.K. aquifers. J. Inst. Water Environ. Manage., 4: 242-250.
ROSENGREN, L.E., AURELL, A., KJELLSTRAND, P., & HAGLID, K.G. (1985)
Astrogliosis in the cerebral cortex of gerbils after long-term
exposure to 1,1,1-trichloroethane. Scand. J. Work Environ. Health, 11:
447-455.
ROSENKRANZ, H.S., HYMAN, J., & LEIFER, Z. (1981) DNA polymerase
deficient assay. In: de Serres, F.J. & Ashby, J., ed. Evaluation of
short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 210-218 (Progress in Mutation Research, Vol. 1).
ROWLAND, I. & SEVERN, B. (1981) Mutagenicity of carcinogens and
noncarcinogens in the Salmonella/microsome test. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 323-332 (Progress in
Mutation Research, Vol. 1).
RUSSELL, J.W. & SHADOFF, L.A. (1977) The sampling and determination of
halocarbons in ambient air using concentration on porous polymer. J.
Chromatogr., 134: 375-384.
SALAMONE, M.F., HEDDLE, J.A., & KATZ M. (1981) Mutagenic activity of
41 compounds in the in vivo micronucleus assay. In: de Serres, F.J.
& Ashby, J., ed. Evaluation of short-term tests for carcinogens.
Report of the international collaborative programme, New York,
Amsterdam, London, Elsevier/North Holland, pp. 686-697 (Progress in
Mutation Research, Vol. 1).
SALVINI, M., BINASCHI, S., & RIVA, M. (1971) Evaluation of the
psychophysiological functions in humans exposed to the "threshold
limit value" of 1,1,1-trichloroethane. Br. J. ind. Med., 28: 286-292.
SATO, A., NAKAJIMA, T., & KOYAMA, Y. (1980) Effects of chronic ethanol
consumption on hepatic metabolism of aromatic and chlorinated
hydrocarbons in rats. Br. J. ind. Med., 37: 382-386.
SAVOLAINEN, H., PFAFFLI, P., TENGEN, M., & VAINIO, H. (1977)
Trichloroethylene and 1,1,1-trichloroethane: effects on brain and
liver activity after five days intermittent inhalation. Arch.
Toxicol., 38: 229-237.
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1981)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Int. Arch. occup. environ. Health, 49: 89-98.
SAVOLAINEN, K., RIIHIMÄKI, V., & LAINE A. (1982a) Biphasic effects of
inhaled solvents on human equilibrium. Acta pharmacol. toxicol., 51:
237-242
SAVOLAINEN, K., RIIHIMÄKI, V., LAINE, A., & KEKONI, J. (1982b)
Short-term exposure of human subjects to m-xylene and
1,1,1-trichloroethane. Arch. Toxicol., Suppl. 5: 96-99.
SCHUMANN, A.M., FOX, T.R., & WATANABE, P.G. (1982) [14C]-Methyl
chloroform (1,1,1-trichloroethane): Pharmacokinetics in rats and mice
following inhalation exposure. Toxicol. appl. Pharmacol., 62: 390-401.
SEKI, Y., URASHIMA, Y., AIKAWA, H., MATSUMURA, H., ICHIKAWA, Y.,
HIRATSUKA, F., YOSHIOKA, Y., SHIMBO, S., & IKEDA, M. (1975)
Trichloro-compounds in the urine of humans exposed to methyl
chloroform at sub-threshold levels. Int. Arch. Arbeitsmed., 34: 39-49.
SHIMADA, T., SWANSON, A.F., LEBER, P., & WILLIAMS, G.M. (1985)
Activities of chlorinated ethane and ethylene compounds in the
Salmonella/rat microsome mutagenesis and rat hepatocyte/DNA repair
assays under vapor phase exposure conditions. Cell Biol. Toxicol., 1:
159-179.
SHMUTER, L.M. (1977) [The effect of chronic exposure to low
concentrations of chlorinated ethane series hydrocarbons on specific
and non-specific immunological response in animals under experimental
conditions.] Gig. Tr. prof. Zabol., 8: 38-43 (in Russian).
SIEGEL, J., JONES, R.A., COON, R.A., & LYON J.P. (1971) Effects on
experimental animals of acute, repeated and continuous inhalation
exposures to dichloroacetylene mixtures. Toxicol. appl. Pharmacol.,
18: 168-174.
SIMMON, V.F. & SHEPHERD, G.F. (1981) Mutagenic activity of 42 coded
compounds in the Salmonella/microsome assay. In: de Serres, F.J. &
Ashby, J., ed. Evaluation of short-term tests for carcinogens. Report
of the international collaborative programme, New York, Amsterdam,
London, Elsevier/North Holland, pp. 333-342 (Progress in Mutation
Research, Vol. 1).
SIMMON, V.F., KAUHANEN, K., & TARDIFF, R.G. (1977) Mutagenic activity
of chemicals identified in drinking water. In: Scott, D., Bridges,
B.A., & Sobels, F.H., ed. Progress in genetic toxicology, Amsterdam,
Elsevier/North Holland, pp. 249-258.
SIMMONDS, P.G., KERRIN, S.L., LOVELOCK, J.E., & SHAIR, F.H. (1974)
Distribution of atmospheric halocarbons in the air over the Los
Angeles basin. Atmos. Environ., 8: 209-216.
SINGH, H.B., SALAS, L.J., & STILES, R.E. (1982) Distribution of
selected gaseous organic mutagens and suspect carcinogens in ambient
air. Environ. Sci. Technol., 16: 982-880.
SNOW, L.P., MCNAIR, B.C., & CASTRO B.C. (1979) Mutagenesis testing of
1,1,1-trichloroethane in Salmonella strains TA100 and TA98, Research
Triangle Park, North Carolina, Northrop Services Inc. (Unpublished
report prepared for the US Environmental Protection Agency).
STAHL, C.J. FATTEH, A.V., & DOMINGUEZ, A.M. (1969) Trichloroethane
poisoning: observations on the pathology and toxicology in six fatal
cases. J. Forensic. Sci., 14: 393-397.
STEWART, R.D. & ANDREWS J.T. (1966) Acute intoxication with methyl
chloroform. J. Am. Med. Assoc., 195: 904-906.
STEWART, R.D. (1968) The toxicology of 1,1,1-trichloroethane. Ann.
occup. Hyg., 11: 71-79.
STEWART, R.D. (1971) Methyl chloroform intoxication. J. Am. Med.
Assoc., 215: 1789-1792.
STEWART, R.D. & DODD, H.D. (1964) Absorption of carbon tetrachloride,
trichloroethylene, tetrachloroethylene, methylene chloride and
1,1,1-trichloroethane through the human skin. Am. Ind. Hyg. Assoc. J.,
25: 439-446.
STEWART, R.D., GAY, H.H., ERLEY, D.S., HAKE, C.L., & SCHAFFER, A.W.
(1961) Human exposure to 1,1,1-trichloroethane vapour: relationship of
expired air and blood concentrations. Am. Ind. Hyg. Assoc. J., 22:
252-262.
STEWART, R.D., GAY, H.H., SCHAFFER, A.W., ERLEY, D.S., & ROWE, V.K.
(1969) Experimental human exposure to methyl chloroform vapor. Arch.
environ. Health, 19: 467-472.
STYLES, J.A. (1981) Activity of 42 coded compounds in the BHK-21 cell
transformation test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 638-646 (Progress in Mutation Research, Vol. 1).
THIELE, D.L., EIGENBRODT, E.H., & WARE, A.J. (1982) Cirrhosis after
repeated trichloroethylene and 1,1,1-trichloroethane exposure.
Gastroenterology, 83: 926-929.
THOMPSON, R.S. & CARMICHAEL, N.G. (1989) 1,1,1-Trichloroethane:
Medium-term toxicity to carp, daphnids, and higher plants. Ecotoxicol.
environ. Saf., 17: 172-182.
TOFTGAARD, R., NILSEN, O.G., & GUSTAFSSON, J.A. (1981) Changes in rat
liver microsomal cytochrome P-450 and enzymatic activities after the
inhalation of n-hexane, xylene, methyl ethyl ketone and
methylchloroform for four weeks. Scand. J. Work Environ. Health, 7:
31-37.
TOPHAM, J.C. (1980) Do induced sperm-head abnormalities in mice
specifically identify mammalian mutagens rather than carcinogens?
Mutat. Res., 74: 379-387.
TORKELSON, T.R., OYEN, F., MCCOLLISTER, D.D., & ROWE V.K. (1958)
Toxicity of 1,1,1-trichloroethane as determined on laboratory animals
and human subjects. Am. Ind. Hyg. Assoc. J., 19: 353-362.
TORSLOV, J. (1988) Hazard assessment of 1,1,1-trichloroethane,
Copenhagen, Denmark, Ministry of the Environment, National Agency of
Environmental Protection, 67 pp (Miljoproject, No. 10).
TRAVIS, C.C., HATTEMER-FREY, H.A., & ARMS, A.D. (1988) Relationship
between dietary intake of organic chemicals and their concentrations
in human adipose tissue and breast milk. Arch. environ. Contam.
Toxicol., 17: 473-478.
TSUCHIMOTO, T. & MATTER, B.E. (1981) Activity of coded compounds in
the micronucleus test. In: de Serres, F.J. & Ashby, J., ed. Evaluation
of short-term tests for carcinogens. Report of the international
collaborative programme, New York, Amsterdam, London, Elsevier/North
Holland, pp. 705-711 (Progress in Mutation Research, Vol. 1). TSURUTA,
H. (1975) Percutaneous absorption of organic solvents. I. Comparative
study of the in vivo percutaneous absorption of chlorinated solvents
in mice. Ind. Health, 13: 227-236.
TU, A.S., MURRAY, T.A., HATCH, K.M., SIVAK, A., & MILMAN, H.A. (1985)
In vitro transformation of balb/c-3T3 cells by chlorinated ethanes
and ethylenes. Cancer Lett., 28: 85-92.
TWEATS, D.J. (1981) Activity of 42 coded compounds in a differential
killing test using Escherichia coli strains WP2, WP67 (uvrA polA),
and CM871 (uvrA lexA recA). In: de Serres, F.J. & Ashby, J., ed.
Evaluation of short-term tests for carcinogens. Report of the
international collaborative programme, New York, Amsterdam, London,
Elsevier/North Holland, pp. 199-209 (Progress in Mutation Research,
Vol. 1).
UHLER, A.D. & DIACHENKO, G.W. (1987) Volatile halocarbon compounds in
process water and processed foods. Bull. environ. Contam. Toxicol.,
39: 601-607.
UNEP (1989) Technical progress on protecting the ozone layer. Report
of the Technology Review Panel, Nairobi, United Nations Environment
Programme, 103 pp.
URANO, K. (1985) Usage and pollution control of chlorinated organic
compounds. Jpn. J. Water Pollut. Res., 8: 269-273.
URANO, K., KAWAMOTO, K., ABE, Y., & OTAKE, M. (1988) Chlorinated
organic compounds in urban air in Japan. Sci. total Environ., 74:
121-131.
US EPA (1977) Environmental monitoring near industrial areas -
Methylchloroform, Washington, DC, US Environmental Protection Agency
(EPA-560/6-77-025).
US EPA (1980a) Proceedings of the Conference on Methyl Chloroform and
Other Halocarbon Pollutants, Washington, DC, US Environmental
Protection Agency (EPA-600/9-80-003).
US EPA (1980b) Ambient water quality criteria for chlorinated ethanes,
Washington, DC, US Environmental Protection Agency (EPA-440/5-80-029).
US EPA (1984) Health assessment document for 1,1,1-trichloroethane
(methyl chloroform), Washington, DC, US Environmental Protection
Agency (EPA-600/8-82-003F).
VAN BEEK, L. (1990) Investigation of a possibility to reduce the
number of rabbits in skin irritation tests; experiments with
dichloromethane, trichloromethylene, tetrachloroethylene and
1,1,1-trichloroethane (Project No. B 86-0927) Apeldoorn, The
Netherlands, Organization for Applied Scientific Research (TNO Report
No. V 89.265).
VEITH, G.D., MACEK, K.J. PETROCELLI, S.R., & CARROLL, J. (1980) An
evaluation of using partition coefficients and water solubility to
estimate bioconcentration factors for organic chemicals in fish. In:
Eaton, J.G., Parrish, P., & Hendricks, A.C., ed. Aquatic toxicology.
Proceedings of the Third Annual Symposium on Aquatic Toxicology,
Philadelphia, American Society for Testing and Materials, pp. 116-129
(ASTM STP No. 707).
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., WHITMORE, R.
SPARACINO, C., & ZELON, H. (1986) Total exposure assessment
methodology (TEAM) study: personal exposures, indoor-outdoor
relationships, and breath levels of volatile organic compounds in New
Jersey. Environ. Int., 12: 369-387.
WALLACE, L.A., PELLIZZARI, E.D., HARTWELL, T.D., SPARACINO, C.,
WHITMORE, R., SHELDON, L., ZELON, H., & PERRITT, R. (1987) The TEAM
study: Personal exposure to substances in air, drinking water, and
breath of 400 residents of New Jersey, North Carolina and North
Dakota. Environ. Res., 43: 290-307.
WALRAEVENS, R., TROUILLET, P., & DEVOS, A. (1974) Basic elimination of
HCI from chlorinated ethanes. Int. J. chem. Kinet., VI: 777-786.
WEAST, R.C. (1986) CRC handbook of chemistry and physics, 67th ed,
Boca Raton, Florida, CRC Press.
WESTERBERG, E. & LARSSON, L. (1982) Use of automated head-space gas
chromatography for determination of 1,1,1-trichloroethane in rat blood
and brain tissue. Int. J. environ. anal. Chem., 12: 233-239.
WILLIAMS, G.M. (1983) DNA repair tests of 11 chlorinated hydrocarbon
analogs, Washington, DC, US Environmental Protection Agency (Draft
Final Report TR-507-18).
WILSON, B.H. & WHITE, M.V. (1986) A fixed-film bioreactor to treat
trichloroethylene-laden waters from interdiction wells. In:
Proceedings of the 1986 National Symposium and Exposition on Aquifer
Restoration and Ground Water Monitoring, Columbus, Ohio, 19-22 May,
Dublin, Ohio, National Well Water Association, pp. 425-436.
WILSON, J.T., MCNABB, J.F., WILSON, B.H., & NOONAN, M.J. (1983)
Biotransformation of selected organic pollutants in ground water. Dev.
ind. Microbiol., 24: 125-233.
WHO (in preparation) Health and Safety Guide for
1,1,1-trichloroethane, Geneva, World Health Organization.
WINDHOLZ, M. ed. (1983) The Merck index: An encyclopedia of chemicals,
drugs and biologicals, 10th ed., Rahway, New Jersey, Merck and Co.,
Inc.
WMO (1982) Global ozone research and monitoring project: Report No.
11. The stratosphere 1981: Theory and measurements, Geneva,
Switzerland, World Meteorological Organization.
WOOLVERTON, W.L. & BALSTER, R.L. (1981) Behavioral and lethal effects
of combinations of oral ethanol and inhaled 1,1,1-trichloroethane in
mice. Toxicol. appl. Pharmacol., 59: 1-7.
WRIGHT, M.F. & STROBL, D.J. (1984) 1,1,1-Trichloroethane cardiac
toxicity: Report of a case. J. Am. Osteopath. Assoc., 84: 285-288.
WRIGHTSON, I. & SANTON, R.C. (1988) An explosion involving
1,1,1-trichloroethane. Loss Prev. Bull., 083: 21-23.
YORK, R.G., SOWRY, B.M., HASTINGS, L., & MANSON, J.M. (1982)
Evaluation of teratogenicity and neurotoxicity with maternal
inhalation exposure to methylchloroform. J. Toxicol. environ. Health,
9: 251-266.
YUNG, Y.L., MCELROY, M.B., & WOFSY, S.C. (1975) Atmospheric
halocarbons: A discussion with emphasis on chloroform. Geophys. Res.
Lett., 2: 397-399.
RESUME
Le 1,1,1-trichloréthane est un hydrocarbure chloré produit par
chloration du chlorure de vinyle ou du chlorure de vinylidène. En
1988, la production mondiale était d'environ 680 000 tonnes. C'est un
liquide incolore et volatil à l'odeur caractéristique dont la vapeur
est plus dense que l'air. On l'utilise essentiellement pour le
dégraissage des surfaces métalliques et comme solvant dans un grand
nombre de produits industriels et de produits de consom-mation,
notamment des adhésifs, des détachants et des bombes aérosols. Il
sert également d'intermédiaire en synthèse chimique. Le
trichloréthane technique contient généralement 3 à 8% de stabilisants
destinés à empêcher sa décomposition et la formation d'acide
chlorhydrique; les parties métalliques sont ainsi protégées de la
corrosion. Il est ininflammable dans les conditions normales mais la
vapeur s'enflamme à haute température et, au cours du soudage, elle
peut se décomposer en libérant du phosgène, un gaz toxique. Des
réactions extrêmement violentes peuvent se produire en cas de contact
avec de l'aluminium, du magnésium ou leurs alliages.
Le 1,1,1-trichloréthane pénètre facilement dans l'environne-ment.
Comme il séjourne longtemps dans la troposphère (environ six ans) et
qu'il n'est que lentement biodégradable, on le retrouve maintenant
partout dans l'environnement, même à grande distance des zones
industrielles. A proximité d'installations industrielles où ce
composé est produit ou manipulé, on a trouvé du 1,1,1-trichloréthane
à des concentrations atteignant 86 µg/m3 (16 000 ppm, p/p) dans des
échantillons d'air.
Le trichloréthane se déplace dans le sol et parvient jusqu'aux
eaux souterraines. On en a trouvé à des concentrations atteignant
1600 µg/litre dans les eaux souterraines ou les eaux de surface. Ce
phénomène peut être à l'origine de la contamination des réserves d'eau
potable.
On estime que 15% des émissions annuelles de
1,1,1-trichloré-thane sont entraînées dans la stratosphère où elles
peuvent endommager la couche d'ozone par libération d'atomes de
chlore.
Des essais biologiques sur des crustacés et des poissons à des
concentrations supérieures à 7 mg/litre ont révélé des effets toxiques
aigus. On possède quelques données selon lesquelles le composé ne
s'accumulerait que faiblement chez les organismes aquatiques. En
raison du caractère limité des données disponibles, il est difficile
d'évaluer les effets qu'il peut avoir sur les organismes terrestres.
L'exposition humaine au 1,1,1-trichloréthane est due
principalement à l'inhalation de ce composé qui est ensuite rapidement
résorbé par l'organisme. Il peut y avoir également exposition par
contact cutané ou ingestion. Le trichloréthane se répartit largement
dans les tissus de l'organisme et il traverse la barrière
hématoencéphalique et placentaire. On le retrouve également dans le
lait humain mais il ne semble pas qu'il s'accumule dans les tissus.
Sa principale voie d'élimination est l'exhalation du composé non
métabolisé.
La toxicité aiguë et chronique du 1,1,1-trichloréthane est
relativement faible mais en cas de forte exposition, le risque
d'intoxication est réel. Cela peut se produire en cas d'exposition
professionnelle, d'accidents ou encore de toxicomanie aux solvants.
Comme il s'agit d'un solvant volatil dont la vapeur est beaucoup plus
dense que l'air, il peut se trouver à des concentrations
inhabituellement fortes et dangereuses dans des espaces confinés comme
les réservoirs "vides". Ce genre de situation a donné lieu à
plusieurs intoxications mortelles ou qui auraient pu l'être sur les
lieux de travail ou ailleurs.
Chez l'homme, l'effet critique s'exerce au niveau du système
nerveux central. On observe des effets qui vont de légers troubles du
comportement (accompagnés d'une légère irritation occulaire) à la dose
de 1,9 g/m3 (350 ppm), jusqu'à l'inconscience et à l'arrêt
respiratoire lorsque la concentration est élevée. Dans certains cas,
des anomalies cardiaques mortelles peuvent se produire. Le
trichloréthane est moins toxique pour le foie que la plupart des
autres solvants organiques chlorés. La dose sans effet observable
pour l'homme se situe autour de 1,35 g/m3 (250 ppm).
Aucune étude satisfaisante n'a été publiée sur les effets
cancérogènes de ce solvant chez l'homme. Toutefois une étude par
inhalation de longue durée sur des rats et des souris exposés à 8,1
g/m3 (1500 ppm) n'a pas révélé le moindre effet cancérogène. Le
1,1,1-trichloréthane ne présente pas d'activité génotoxique
significative.
Chez des rats et des lapins exposés au trichloréthane à des
concentrations qui étaient toxiques pour les femelles gestantes, on
n'a pas noté d'effets tératogènes, malgré la présence d'effets
toxiques sur le développement. Les données épidémiologiques relatives
à ces effets sur la reproduction sont trop limitées pour permettre
d'en tirer une conclusion.
RESUMEN
El 1,1,1-tricloroetano es un hidrocarburo clorado que se fabrica
a partir del cloruro de vinilo o del cloruro de vinilideno por
cloración. En 1988 la producción mundial fue de unas 680 000
toneladas. Es un líquido volátil e incoloro con un olor
característico, y su vapor es más denso que el aire. Se utiliza
principalmente en el desengrasado de metales y como disolvente en
muchos productos industriales y de consumo, como adhesivos,
quitamanchas y envases de aerosoles. Es también un producto químico
intermediario. El tricloroetano de calidad técnica suele contener del
3% al 8% de estabilizadores que impiden la degradación y la formación
de ácido clorhídrico a fin de proteger las partes metálicas de la
corrosión. En condiciones normales no es inflamable, pero su vapor
arde a altas temperaturas y, durante las operaciones de soldadura, su
degradación produce fosgeno, un gas tóxico. Cuando entra en contacto
con el aluminio, el magnesio y sus aleaciones puede provocar
reacciones muy violentas.
El 1,1,1-tricloroetano llega fácilmente al medio ambiente. Su
prolongada permanencia en la troposfera (unos seis años) y baja
biodegradabilidad son razón de su actual omnipresencia en el medio
ambiente, incluso lejos de las zonas industriales. En muestras de
aire de zonas próximas a industrias que lo fabrican o lo manipulan se
han detectado concentraciones de hasta 86 µg/m3 (16 ppb, peso/peso).
El tricloroetano se desplaza en los suelos y alcanza las aguas
subterráneas. Se han encontrado concentraciones de hasta 1600
µg/litro en aguas subterráneas o superficiales. Esta puede ser una
fuente de contaminación para suministros de agua potable.
Se estima que el 15% del volumen anual liberado llega a la
estratosfera, donde libera átomos de cloro que contribuyen a la
destrucción del ozono.
En bioensayos con crustáceos y peces se han observado efectos
tóxicos agudos con concentraciones superiores a 7 mg/litro. Limitada
información limitada disponible parece indicar que la bioacumulación
en los organismos acuáticos es baja. La escasez de datos dificulta la
evaluación de los posibles efectos en los organismos terrestres.
El ser humano está expuesto al 1,1,1-tricloroetano principalmente
por inhalación, y luego el organismo absorbe rápidamente la sustancia.
También se puede dar la exposición por contacto cutáneo o ingestión.
El tricloroetano se distribuye ampliamente por los tejidos y atraviesa
las barreras hematoencefálica y placentaria. Aunque también se ha
encontrado en la leche humana, no parece que se produzca
bioacumulación. La principal vía de eliminación es la exhalación del
compuesto inalterado.
Las toxicidades aguda y crónica del 1,1,1-tricloroetano son
relativamente bajas, pero en condiciones de exposición intensa pueden
producirse efectos tóxicos. Estas condiciones se pueden presentar en
casos de exposición profesional, de abuso del disolvente o de
accidentes. Dado que el disolvente es volátil y el vapor es mucho más
denso que el aire, se pueden producir concentraciones inesperadamente
elevadas y peligrosas en espacios reducidos, como depósitos de
almacenamiento "vacíos". Así, se han producido algunas intoxicaciones
mortales y casi mortales en lugares de trabajo y otros.
El efecto más importante en el ser humano se produce en el
sistema nervioso central. Los efectos observables van desde ligeros
cambios de comportamiento (acompañados de una leve irritación ocular)
con 1,9 g/m3 (350 ppm) hasta la pérdida del conocimiento y el paro
respiratorio con concentraciones más elevadas. Sin embargo, también
se pueden producir anomalías cardíacas fatales. La toxicidad hepática
del tricloroetano es inferior a la de la mayoría de los disolventes
organoclorados. El nivel sin efecto observado (NOEL) para la especie
humana parece ser del orden de 1,35 g/m3 (250 ppm).
No se han publicado estudios adecuados sobre los efectos
carcinogénicos en la especie humana. Sin embargo, en los estudios de
inhalación prolongada en ratas y ratones expuestos a 8,1 g/m3 (1500
ppm) no se obtuvieron pruebas de ningún efecto carcinogénico. El
1,1,1-tricloroetano no tiene un potencial genotóxico importante.
En ratas y conejos se ha observado toxicidad en la fase de
crecimiento, pero no teratogenicidad, con concentraciones que fueron
tóxicas para las madres. Las escasas pruebas epidemiológicas en
relación con sus efectos sobre la reproducción no son concluyentes.
