
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 100
CRESOLS
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1996
This is a companion volume to Environmental Health Criteria 168:
Cresols
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Cresols: health and safety guide.
(Health and safety guide ; no. 100)
1.Cresols - adverse effects 2.Anti-infective agents
3.Occupational exposure 4. I.Series
ISBN 92 4 151100 1 (NLM Classification: QD 341.P5)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1996
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analysis
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Environmental fate
2.2. Exposure
2.3. Uptake, metabolism, and excretion
2.4. Effects on organisms in the environment
2.5. Effects on experimental animals and in vitro test systems
2.6. Effects on humans
3. CONCLUSIONS
3.1. Human health
3.2. Environment
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Human health hazards, prevention and protection, first aid
4.1.1. Information for physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.2.1. Explosion hazards
4.2.2. Fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage
4.6. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Exposure limit values
7.2. Specific restrictions
7.3. Labelling, packaging, and transport
7.4. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) monographs produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: Cresols ( o, m, p-isomers)
Common structure:
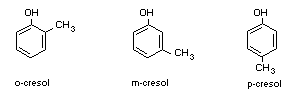 o-cresol m-cresol p-cresol
Common synonyms:
2-methyl phenol 3-methyl phenol 4-methyl phenol
2-hydroxy toluene 3-hydroxy toluene 4-hydroxyl toluene
o-cresylic acid m-cresylic acid p-cresylic acid
o-kresol (German) m-kresol (German) p-kresol(German)
Chemical formula: C7H8O
Common trade name: cresylic acid (mixture of the three isomers)
cresylic acids (mixture of phenols)
tricresol (mixture of o, m, p- isomers)
dicresol (mixture of m- and p- isomers)
lysol (mixture of 60 cresol: 40 soap)
CAS registry
number:
95-48-7 109-39-4 106-44-5
( o-cresol) ( m-cresol) ( p-cresol)
RTECS number:
G06300000 G06125000 G06475000
Relative
molecular mass: 108.14
Conversion factors: air at 25°C: 1 ppm = 4.42 mg/m3
1 mg/m3 = 0.23 ppm
water: 1 ppm = 1 mg/litre
solid or
semi-solid: 1 ppm = 1 mg/kg
1.2 Physical and Chemical Properties
Cresols are isomeric substituted phenols with a methyl substituent at
one of the o-, m-, or p- positions relative to the OH-group.
Physically, they are white crystalline solids or yellowish liquids
with a strong phenol-like odour. The compounds are highly flammable,
moderately soluble in water and soluble in ethanol, ether, acetone, or
alkali hydroxides. Chemically, these alkylphenols undergo electro-
philic substitution reactions at the vacant o- or p-positions
or undergo condensation reactions with aldehydes, ketones, or dienes.
Physical and chemical data on cresols are presented in the Summary of
Chemical Safety information (section 6).
1.3 Analysis
Cresols can be detected and quantified in a number of environmental
media and biological samples. The most commonly used methods for
determining the presence of cresols are gas chromatography-mass
spectrophotometry (GC-MS) and high-performance liquid chromatography
(HPLC). Sample detection limits are 1.33 ng/m3 for air;
4-10 µg/litre for water; 300 µg/kg for soil and 0.5-1.0 mg/litre for
urine. Air odour thresholds were reported to be 1.4-22, 0.007-1.2, and
0.002-0.04 mg/m3 for o-, m-, and p-cresol, respectively.
1.4 Production and Uses
Cresols and cresol derivatives occur naturally in the oils of various
flowering plants and trees, such as jasmine, easter lily, yucca,
conifers, oaks, and sandalwoods. p-Cresol is found in the urine of
animals and humans. Commercially, cresols are produced as by-products
in the fractional distillation of crude oil and coal tars and in coal
gasification. They are also formed as by-products during the
combustion of wood, coal, and cigarettes. The various isomers can be
manufactured by the methylation of phenols, toluene sulfonation and
alkaline hydrolysis, or the hydrolysis of 2-isopropyltoluene or
alkaline chlorotoluene. Worldwide production is not known; annual
production in the USA for 1990 was reported to be 38.3 million kg.
Cresols have a wide variety of uses as solvents, disinfectants, or
intermediates in the preparation of numerous products. They are
commonly used in the production of fragrances, antioxidants, dyes,
pesticides, and resins. In addition, p-cresol is used in the
production of lubricating oils, motor fuels, and rubber polymers,
while m-cresol is also used in the manufacture of explosives.
2. SUMMARY AND EVALUATION
2.1 Environmental Fate
Environmental transport of cresols occurs through the vapour phase of
the atmosphere and from the atmosphere to surface water and soil by
rain-scavenging. Because of their volatilization, binding to sediment,
and biodegradation, only small amounts of cresols are found in water.
In soils, cresols are slightly to highly mobile, depending on the
Koc value of the soil. Cresols have been detected in groundwater;
therefore leaching does occur in soil.
2.2 Exposure
Exposure to cresols can occur through air, water, or food. The median
air concentration of o-cresols was 1.578 µg/m3 (0.359 ppb) for 32
source dominated sites in USA. In the USA, surface water
concentrations ranged from below detection limits to 77 µg/litre.
Levels of 204 µg/litre were reported in Japan in a river polluted from
industrial effluents. Concentrations as high as 2100 µg/litre for
o-cresol and 1200 µg/litre for mixed m- and p-cresols have been
detected in waste waters. Concentrations of all three isomers in
groundwater from undefined sources ranged from below detection limits
to 100 mg/litre. Rainwater concentrations ranged from 240 to
2800 ng/litre for o-cresol and 380 to 2000 ng/litre for p- and
m-cresol combined. Cresols have been detected in foods and
beverages. Concentrations in spirit beverages ranged from 0.01 to
0.2 mg/litre. The amount in tobacco smoke was 75 µg/85-mm, non-filter,
American cigarette. The general population can be exposed to cresols
through air inhalation, drinking-water, food, and beverage ingestion,
and dermal contact. In general, the lack of adequate monitoring data
makes the quantitative estimate of daily intakes of cresol from these
sources impossible. Occupational exposure levels in air as high as
5.0 mg/m3 have been reported.
2.3 Uptake, Metabolism, and Excretion
Cresols are absorbed across the respiratory and gastrointestinal
tracts and through the skin. The rate and extent of absorption of
cresols have not been studied specifically; however, studies have
shown that both gastrointestinal and dermal absorption are rapid and
extensive. Cresols are distributed to all the major organs. The
primary metabolic pathway for cresols is conjugation with glucuronic
acid and inorganic sulfate. Minor metabolic pathways for cresols
include hydroxylation of the benzene ting and side-chain oxidation.
The main route of elimination of cresols from the body is renal
elimination in the form of conjugates.
2.4 Effects on Organisms in the Environment
Observations on microorganisms, invertebrates, and fish are available
and show that cresols may represent a risk to non-mammalian organisms
at point sources with high cresol concentrations, but not in the
general environment.
2.5 Effects on Experimental Animals and In Vitro Test Systems
Acute poisoning with cresol vapours is unlikely due to the low vapour
pressure of these compounds. Mean lethal concentrations of cresols for
rats have been reported to be 29 mg/m3 for o- and p-cresols and
58 mg/m3 for m-cresols. Oral LD50 values in rats have been
reported to be 121, 207, and 242 mg/kg body weight for o-, p-, and
m-isomers, respectively. Inter-species comparisons show that all
three isomers are more toxic for mice than for rats and that toxicity
increases with concentration. Systemic toxicity and death can result
from dermal exposure. Dermal LD50 values in rabbits were 890, 2830,
300, and 2000 mg/kg body weight for o-, m-, p-, and mixed cresols,
respectively. In rats, dermal LD50 values were 620, 1100, 750, and
825 mg/kg body weight for o-, m-, p-, and dicresol, respectively.
Cresols are highly irritating to the skin and eyes of rabbits, rats,
and mice.
Short-term exposure to inhaled mixtures of o-cresol aerosol and
vapours resulted in irritation of the respiratory tract, small
haemorrhages in the lung, body weight reduction, and degeneration of
heart muscle, liver, kidney, and nerve cells. Short-term (28-days)
oral exposure to approximately 800 mg/kg body weight per day and above
resulted in reduced body weights, organ weight changes, and
histopathological changes in the respiratory and gastrointestinal
tracts of rats. In mice similarly exposed to 1500 mg/kg body weight,
more severe effects were reported and, at the highest concentrations,
death resulted from exposure to o-, and m-, and p-cresols, but
not from exposure to mixtures of the isomers.
Exposure of rats to vapours of o-, m-, or p-cresol for up to 4
months resulted in weight loss, reduced locomotor activity,
inflammation of nasal membranes and skin, and changes in the liver.
Oral exposures of up to 13 weeks of mice, rats, and hamsters resulted
in mortality, tremors, reduced body weights, haematological effects,
increased organ weights, and hyperplasia of nasal, and forestomach
epithelium.
Oral and inhalation exposures to cresol isomers have resulted in
lengthened estrus cycles, and histopathological changes in the uterus
and ovaries of rats and mice. No adverse effects were observed on
spermatogenesis in rats or mice. Mild letotoxic effects have been
reported in rats and rabbits exposed to o-, and p-cresols, but
only minor treatment-related developmental effects have been reported.
Some evidence of genotoxicity has been reported in in vitro studies
with o- and p-cresols but not m-cresol. No positive results were
obtained in in vivo studies; however, some evidence of promotive
activities in skin has been reported. No studies of carcinogenicity
have been reported for any cresol isomers.
2.6 Effects on Humans
Ingestion of cresols results in burning of the mouth and throat,
abdominal pain, and vomiting. Target organs of ingested cresols in
humans are the blood and kidneys and effects on the lungs, liver,
heart, and central nervous system have also been reported. In severe
cases, coma and death may result. Dermal exposure has been reported to
cause severe skin burns, scarring, systemic toxicity, and death.
Occupational exposure to cresols usually results from dermal contact.
Acute exposures have resulted in severe bums, anuria, coma, and death.
Very few data are available regarding the reproductive effects of
cresols and no data are available on carcinogenicity in humans.
3. CONCLUSIONS
3.1 Human Health
Cresols, at concentrations normally found in the environment, do not
pose any significant risk for the general population. However, the
potential for adverse health effects exists for specific
subpopulations and under conditions of high exposure.
No information is available regarding the effects of long-term
exposure to cresols. Thus, information to assess the carcinogenic
hazard of cresols is inadequate. On the basis of the results of short-
term studies, an NOAEL of 50 mg/kg body weight per day has been
identified for all three isomers of cresols. Applying an uncertainty
factor of 300 was recommended as follows: 10 to account for
interspecies variation; 10 to account for the lack of long-term
toxicity studies and possible genotoxic and promoting activity of
cresols; and 3 to account for intraspecies variation based on the
rapid and complete metabolism. Thus, an ADI of 0.17 mg/kg body weight
per day can be developed for cresols.
NOAEL = 50 mg/kg per day
Uncertainty factor = 300
ADI = 0.17 mg/kg body weight per day.
3.2 Environment
Observations on microorganisms, invertebrates, and fish show that
cresols may present a risk for non-mammalian organisms at point
sources with high cresol concentrations, but not in the general
environment.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure to
cresols, together with preventive and protective measures and first
aid are shown in the Summary of Chemical Safety Information
(section 6).
4.1.1 Information for physicians
High oral or dermal exposure to cresols may rapidly prove fatal.
Exposed patients should be decontaminated and treated symptomatically.
Maintenance of breathing and circulation is necessary in cases of
severe poisoning. There is no specific antidote. Treatment of local
lesions and systemic intoxication is symptomatic.
Monitor blood, liver, and kidney functions. Call the nearest Poisons
Information Centre for further advice.
4.1.2 Health surveillance advice
The blood of individuals exposed to cresols on a frequent or long-term
basis should be examined for haematological changes. Periodic medical
examinations should also include testing of kidney and liver
functions, and skin and eye sensitivity or irritation. Special
attention should be given to pregnant females and females of child-
bearing age.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
The vapours of cresols may become explosive when exposed to heat or
flames; the flash-point is approximately 80°C. Cylinders containing
liquid may explode in heat.
4.2.2 Fire hazards
Highly flammable and volatile cresols can cause severe toxic effects,
including burning and irritation of the skin, eyes, and nasal
membranes.
4.3 Storage
Cresols should be stored in well-sealed containers in cool, well-
ventilated areas, away from sources of ignition.
4.4 Transport
Transport should be in well-sealed, protective containers which should
be labelled indicating the high flammability and corrosive and
poisonous nature of the substance.
4.5 Spillage
In the case of spillage, wear protective gloves and glasses and avoid
direct contact with skin. Evacuate and ventilate the area. Absorb
leaking liquid in sand, earth, or inert material, and deposit in
sealed containers. Collect powdered material in the most convenient
and safe manner and deposit in a sealable container.
4.6 Disposal
Dispose of as hazardous waste.
5. HAZARDS FOR ENVIRONMENT AND THEIR PREVENTION
Cresols are present in the environment in the air, water, and soil,
and may undergo rapid chemical and biological transformation. It is
unlikely that cresols bioaccumulate in the environment or food chain.
Cresols are moderately toxic for aquatic bacteria, algae, and
protozoa. The acute toxicity of cresols for invertebrates is low to
moderate. Cresols have also been shown to be toxic for fish and birds.
Contamination of the environment can be reduced by controlling
emissions from industrial point sources. In addition, proper methods
for the storage, transport, and waste disposal of cresols are
recommended to reduce the incidence of accidental releases of cresols
into the environment.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, cresols. It should be displayed at, or
near, entrances to areas where there is potential exposure to
cresols, and on processing equipment and containers. The summary
should be translated into the appropriate language(s). All persons
potentially exposed to the chemical should also have the instructions
in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
Cresols ( o, m, p isomers)
C7H8O
Physical properties o-cresol m-cresol p-cresol Other characteristics
Relative molecular mass 108.14 108.14 108.14 White crystalline solid or yellowish liquid;
Melting point (°C) 30.94 12.22 34.74 soluble in ethanol, ethyl ether, acetone,
Boiling point (°C) 191.0 202.32 201.94 benzene, aqueous alkali hydroxides; pbenolic-
Water solubility (25°C) 25.95 22.70 21.52 like odour, highly corrosive, and strong
Relative density (25°C) 1.135 1.030 1.154 irritant; highly reactive with metal (steel,
Vapour density 3.7 3.72 3.72 chromium, aluminum) and their alloys;
Vapour pressure (25°C) 0.31 0.143 0.13 soluble in water
Flash point (°C) 81 86 86
Ignition temperature (°C) 558 558 558
Hazard/symptom Prevention/protection First aid
Skin: Irritant, corrosive; severe Protective clothing, gloves, and shoes Wash immediately with large amount
burns and scarring of water; remove contaminated clothing;
obtain medical care
Eyes: Corrosive, strong irritant; Safety glasses Wash with water continuously for 30 min;
lacrimation, redness obtain medical care immediately
Inhalation: Irritation and burning of Adequate ventilation, use of respirators Remove from exposure; provide fresh air
nasal membrane; burning in nose and adequate ventilation; obtain medical
and throat care
Ingestion: Irritation and burning of Store in properly sealed containers; Rinse mouth; obtain immediate medical care
mouth, throat, and gastrointestinal avoid contact with food or beverages
tract; vomiting
Environment: Toxic for aquatic and Avoid environmental releases from
terrestrial organisms industrial processes and accidental spills
Cresols ( o, m, p isomers) (cont'd)
Spillage Storage Fire and explosion
Evacuate and ventilate area; collect Store away from food and incompatible Flash-point 81-86°C; may be explosive in
leaking liquid in sealed containers; materials (metals) in well-labelled reactions with metals; do not use near open
absorb liquid in earth, sand, or inert containers and well-ventilated areas; fires; poisonous gas may be produced
material; wear protective clothing sources of ignition/open flames, and in a fire
smoking, must be prohibited
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information in the following table has been extracted from the
International Register of Potentially Toxic chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should be verified with the appropriate regulatory authorities before
application.
7.1 Exposure Limit Values
Values for exposure limits are given in the following table.
7.2 Specific Restrictions
Approved in the USA for use as a pesticide.
7.3 Labelling, Packaging, and Transport
In the USA, cresols (all isomers) have been designated as a hazardous
substance R1987 and must be managed according to federal and/or state
hazardous waste regulations.
7.4 Waste Disposal
In the USA, any sealed waste must be listed as hazardous waste,
subject to handling, transport, storage, and disposal requirements,
specified by CERLA, and the Clean Waste Act (effective date 1991).
These regulations apply to all discharges.
As a commercial chemical, cresols must be identified as "toxic waste",
subject to restrictions and disposal regulations, permitting, and
notification (effective date 1989).
Any discharge into surface waters requires a permit under the Clean
Water Act (effective date 1987).
Exposure limit values
Medium Specification Country/ Exposure limit Valueb Effective
organization descriptiona date
Air occupational Argentina 8-h TWA 22 mg/m3 May 1991
Czech Republic TWA 20 mg/m3 1985
CLV 40 mg/m3 1985
Germany 8-h TWA 22 mg/m3 1991
5-min STEL 44 mg/m3
Japan TWA 22 mg/m3 1991
Russian TWA 0.5 mg/m3 1989
Federation CLV 1.5 mg/m3 1989
United Kingdom 8-h TWA 22 mg/m3 1992
USA TWA 22 mg/m3 1989
(ACGIH)
Air ambient Russian 0.02 mg/m3( m-, p-cresols) 1983
Federation 0.028 mg/m3 ( o-cresol)
a TWA: time-weighted average;
CLV: ceiling value;
STEL: short-term exposure limit.
b Refers to o-, m-, p-cresols unless otherwise specified.
BIBLIOGRAPHY
ATSDR (1992) Toxicological profile for cresols. US Department of
Health & Human Services, Agency for Toxic Substances and Disease
Registry.
CEFIC (1979) Transport emergency cards for individual products.
Chemical Industries Association Ltd.
IPCS (1995) Environmental Health Criteria 168: Cresols. Geneva,
World Health Organization.
IRPTC (1994) Data profile (legal file) on cresols. Geneva,
International Register of Potentially Toxic Chemicals.
Sax NI & Lewis RJ (1989) Dangerous properties of industrial
materials, 7th ed. New York, Van Nostrand Reinhold Company.
US NTP (1992) Toxicity studies of cresols (CAS nos. 95-48-7,
108-39-4, 106-44-5) in F344/N rats and B6C3F1 mice (feed studies).
Research Triangle Park, North Carolina, National Toxicology Program.
o-cresol m-cresol p-cresol
Common synonyms:
2-methyl phenol 3-methyl phenol 4-methyl phenol
2-hydroxy toluene 3-hydroxy toluene 4-hydroxyl toluene
o-cresylic acid m-cresylic acid p-cresylic acid
o-kresol (German) m-kresol (German) p-kresol(German)
Chemical formula: C7H8O
Common trade name: cresylic acid (mixture of the three isomers)
cresylic acids (mixture of phenols)
tricresol (mixture of o, m, p- isomers)
dicresol (mixture of m- and p- isomers)
lysol (mixture of 60 cresol: 40 soap)
CAS registry
number:
95-48-7 109-39-4 106-44-5
( o-cresol) ( m-cresol) ( p-cresol)
RTECS number:
G06300000 G06125000 G06475000
Relative
molecular mass: 108.14
Conversion factors: air at 25°C: 1 ppm = 4.42 mg/m3
1 mg/m3 = 0.23 ppm
water: 1 ppm = 1 mg/litre
solid or
semi-solid: 1 ppm = 1 mg/kg
1.2 Physical and Chemical Properties
Cresols are isomeric substituted phenols with a methyl substituent at
one of the o-, m-, or p- positions relative to the OH-group.
Physically, they are white crystalline solids or yellowish liquids
with a strong phenol-like odour. The compounds are highly flammable,
moderately soluble in water and soluble in ethanol, ether, acetone, or
alkali hydroxides. Chemically, these alkylphenols undergo electro-
philic substitution reactions at the vacant o- or p-positions
or undergo condensation reactions with aldehydes, ketones, or dienes.
Physical and chemical data on cresols are presented in the Summary of
Chemical Safety information (section 6).
1.3 Analysis
Cresols can be detected and quantified in a number of environmental
media and biological samples. The most commonly used methods for
determining the presence of cresols are gas chromatography-mass
spectrophotometry (GC-MS) and high-performance liquid chromatography
(HPLC). Sample detection limits are 1.33 ng/m3 for air;
4-10 µg/litre for water; 300 µg/kg for soil and 0.5-1.0 mg/litre for
urine. Air odour thresholds were reported to be 1.4-22, 0.007-1.2, and
0.002-0.04 mg/m3 for o-, m-, and p-cresol, respectively.
1.4 Production and Uses
Cresols and cresol derivatives occur naturally in the oils of various
flowering plants and trees, such as jasmine, easter lily, yucca,
conifers, oaks, and sandalwoods. p-Cresol is found in the urine of
animals and humans. Commercially, cresols are produced as by-products
in the fractional distillation of crude oil and coal tars and in coal
gasification. They are also formed as by-products during the
combustion of wood, coal, and cigarettes. The various isomers can be
manufactured by the methylation of phenols, toluene sulfonation and
alkaline hydrolysis, or the hydrolysis of 2-isopropyltoluene or
alkaline chlorotoluene. Worldwide production is not known; annual
production in the USA for 1990 was reported to be 38.3 million kg.
Cresols have a wide variety of uses as solvents, disinfectants, or
intermediates in the preparation of numerous products. They are
commonly used in the production of fragrances, antioxidants, dyes,
pesticides, and resins. In addition, p-cresol is used in the
production of lubricating oils, motor fuels, and rubber polymers,
while m-cresol is also used in the manufacture of explosives.
2. SUMMARY AND EVALUATION
2.1 Environmental Fate
Environmental transport of cresols occurs through the vapour phase of
the atmosphere and from the atmosphere to surface water and soil by
rain-scavenging. Because of their volatilization, binding to sediment,
and biodegradation, only small amounts of cresols are found in water.
In soils, cresols are slightly to highly mobile, depending on the
Koc value of the soil. Cresols have been detected in groundwater;
therefore leaching does occur in soil.
2.2 Exposure
Exposure to cresols can occur through air, water, or food. The median
air concentration of o-cresols was 1.578 µg/m3 (0.359 ppb) for 32
source dominated sites in USA. In the USA, surface water
concentrations ranged from below detection limits to 77 µg/litre.
Levels of 204 µg/litre were reported in Japan in a river polluted from
industrial effluents. Concentrations as high as 2100 µg/litre for
o-cresol and 1200 µg/litre for mixed m- and p-cresols have been
detected in waste waters. Concentrations of all three isomers in
groundwater from undefined sources ranged from below detection limits
to 100 mg/litre. Rainwater concentrations ranged from 240 to
2800 ng/litre for o-cresol and 380 to 2000 ng/litre for p- and
m-cresol combined. Cresols have been detected in foods and
beverages. Concentrations in spirit beverages ranged from 0.01 to
0.2 mg/litre. The amount in tobacco smoke was 75 µg/85-mm, non-filter,
American cigarette. The general population can be exposed to cresols
through air inhalation, drinking-water, food, and beverage ingestion,
and dermal contact. In general, the lack of adequate monitoring data
makes the quantitative estimate of daily intakes of cresol from these
sources impossible. Occupational exposure levels in air as high as
5.0 mg/m3 have been reported.
2.3 Uptake, Metabolism, and Excretion
Cresols are absorbed across the respiratory and gastrointestinal
tracts and through the skin. The rate and extent of absorption of
cresols have not been studied specifically; however, studies have
shown that both gastrointestinal and dermal absorption are rapid and
extensive. Cresols are distributed to all the major organs. The
primary metabolic pathway for cresols is conjugation with glucuronic
acid and inorganic sulfate. Minor metabolic pathways for cresols
include hydroxylation of the benzene ting and side-chain oxidation.
The main route of elimination of cresols from the body is renal
elimination in the form of conjugates.
2.4 Effects on Organisms in the Environment
Observations on microorganisms, invertebrates, and fish are available
and show that cresols may represent a risk to non-mammalian organisms
at point sources with high cresol concentrations, but not in the
general environment.
2.5 Effects on Experimental Animals and In Vitro Test Systems
Acute poisoning with cresol vapours is unlikely due to the low vapour
pressure of these compounds. Mean lethal concentrations of cresols for
rats have been reported to be 29 mg/m3 for o- and p-cresols and
58 mg/m3 for m-cresols. Oral LD50 values in rats have been
reported to be 121, 207, and 242 mg/kg body weight for o-, p-, and
m-isomers, respectively. Inter-species comparisons show that all
three isomers are more toxic for mice than for rats and that toxicity
increases with concentration. Systemic toxicity and death can result
from dermal exposure. Dermal LD50 values in rabbits were 890, 2830,
300, and 2000 mg/kg body weight for o-, m-, p-, and mixed cresols,
respectively. In rats, dermal LD50 values were 620, 1100, 750, and
825 mg/kg body weight for o-, m-, p-, and dicresol, respectively.
Cresols are highly irritating to the skin and eyes of rabbits, rats,
and mice.
Short-term exposure to inhaled mixtures of o-cresol aerosol and
vapours resulted in irritation of the respiratory tract, small
haemorrhages in the lung, body weight reduction, and degeneration of
heart muscle, liver, kidney, and nerve cells. Short-term (28-days)
oral exposure to approximately 800 mg/kg body weight per day and above
resulted in reduced body weights, organ weight changes, and
histopathological changes in the respiratory and gastrointestinal
tracts of rats. In mice similarly exposed to 1500 mg/kg body weight,
more severe effects were reported and, at the highest concentrations,
death resulted from exposure to o-, and m-, and p-cresols, but
not from exposure to mixtures of the isomers.
Exposure of rats to vapours of o-, m-, or p-cresol for up to 4
months resulted in weight loss, reduced locomotor activity,
inflammation of nasal membranes and skin, and changes in the liver.
Oral exposures of up to 13 weeks of mice, rats, and hamsters resulted
in mortality, tremors, reduced body weights, haematological effects,
increased organ weights, and hyperplasia of nasal, and forestomach
epithelium.
Oral and inhalation exposures to cresol isomers have resulted in
lengthened estrus cycles, and histopathological changes in the uterus
and ovaries of rats and mice. No adverse effects were observed on
spermatogenesis in rats or mice. Mild letotoxic effects have been
reported in rats and rabbits exposed to o-, and p-cresols, but
only minor treatment-related developmental effects have been reported.
Some evidence of genotoxicity has been reported in in vitro studies
with o- and p-cresols but not m-cresol. No positive results were
obtained in in vivo studies; however, some evidence of promotive
activities in skin has been reported. No studies of carcinogenicity
have been reported for any cresol isomers.
2.6 Effects on Humans
Ingestion of cresols results in burning of the mouth and throat,
abdominal pain, and vomiting. Target organs of ingested cresols in
humans are the blood and kidneys and effects on the lungs, liver,
heart, and central nervous system have also been reported. In severe
cases, coma and death may result. Dermal exposure has been reported to
cause severe skin burns, scarring, systemic toxicity, and death.
Occupational exposure to cresols usually results from dermal contact.
Acute exposures have resulted in severe bums, anuria, coma, and death.
Very few data are available regarding the reproductive effects of
cresols and no data are available on carcinogenicity in humans.
3. CONCLUSIONS
3.1 Human Health
Cresols, at concentrations normally found in the environment, do not
pose any significant risk for the general population. However, the
potential for adverse health effects exists for specific
subpopulations and under conditions of high exposure.
No information is available regarding the effects of long-term
exposure to cresols. Thus, information to assess the carcinogenic
hazard of cresols is inadequate. On the basis of the results of short-
term studies, an NOAEL of 50 mg/kg body weight per day has been
identified for all three isomers of cresols. Applying an uncertainty
factor of 300 was recommended as follows: 10 to account for
interspecies variation; 10 to account for the lack of long-term
toxicity studies and possible genotoxic and promoting activity of
cresols; and 3 to account for intraspecies variation based on the
rapid and complete metabolism. Thus, an ADI of 0.17 mg/kg body weight
per day can be developed for cresols.
NOAEL = 50 mg/kg per day
Uncertainty factor = 300
ADI = 0.17 mg/kg body weight per day.
3.2 Environment
Observations on microorganisms, invertebrates, and fish show that
cresols may present a risk for non-mammalian organisms at point
sources with high cresol concentrations, but not in the general
environment.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure to
cresols, together with preventive and protective measures and first
aid are shown in the Summary of Chemical Safety Information
(section 6).
4.1.1 Information for physicians
High oral or dermal exposure to cresols may rapidly prove fatal.
Exposed patients should be decontaminated and treated symptomatically.
Maintenance of breathing and circulation is necessary in cases of
severe poisoning. There is no specific antidote. Treatment of local
lesions and systemic intoxication is symptomatic.
Monitor blood, liver, and kidney functions. Call the nearest Poisons
Information Centre for further advice.
4.1.2 Health surveillance advice
The blood of individuals exposed to cresols on a frequent or long-term
basis should be examined for haematological changes. Periodic medical
examinations should also include testing of kidney and liver
functions, and skin and eye sensitivity or irritation. Special
attention should be given to pregnant females and females of child-
bearing age.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
The vapours of cresols may become explosive when exposed to heat or
flames; the flash-point is approximately 80°C. Cylinders containing
liquid may explode in heat.
4.2.2 Fire hazards
Highly flammable and volatile cresols can cause severe toxic effects,
including burning and irritation of the skin, eyes, and nasal
membranes.
4.3 Storage
Cresols should be stored in well-sealed containers in cool, well-
ventilated areas, away from sources of ignition.
4.4 Transport
Transport should be in well-sealed, protective containers which should
be labelled indicating the high flammability and corrosive and
poisonous nature of the substance.
4.5 Spillage
In the case of spillage, wear protective gloves and glasses and avoid
direct contact with skin. Evacuate and ventilate the area. Absorb
leaking liquid in sand, earth, or inert material, and deposit in
sealed containers. Collect powdered material in the most convenient
and safe manner and deposit in a sealable container.
4.6 Disposal
Dispose of as hazardous waste.
5. HAZARDS FOR ENVIRONMENT AND THEIR PREVENTION
Cresols are present in the environment in the air, water, and soil,
and may undergo rapid chemical and biological transformation. It is
unlikely that cresols bioaccumulate in the environment or food chain.
Cresols are moderately toxic for aquatic bacteria, algae, and
protozoa. The acute toxicity of cresols for invertebrates is low to
moderate. Cresols have also been shown to be toxic for fish and birds.
Contamination of the environment can be reduced by controlling
emissions from industrial point sources. In addition, proper methods
for the storage, transport, and waste disposal of cresols are
recommended to reduce the incidence of accidental releases of cresols
into the environment.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, cresols. It should be displayed at, or
near, entrances to areas where there is potential exposure to
cresols, and on processing equipment and containers. The summary
should be translated into the appropriate language(s). All persons
potentially exposed to the chemical should also have the instructions
in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
Cresols ( o, m, p isomers)
C7H8O
Physical properties o-cresol m-cresol p-cresol Other characteristics
Relative molecular mass 108.14 108.14 108.14 White crystalline solid or yellowish liquid;
Melting point (°C) 30.94 12.22 34.74 soluble in ethanol, ethyl ether, acetone,
Boiling point (°C) 191.0 202.32 201.94 benzene, aqueous alkali hydroxides; pbenolic-
Water solubility (25°C) 25.95 22.70 21.52 like odour, highly corrosive, and strong
Relative density (25°C) 1.135 1.030 1.154 irritant; highly reactive with metal (steel,
Vapour density 3.7 3.72 3.72 chromium, aluminum) and their alloys;
Vapour pressure (25°C) 0.31 0.143 0.13 soluble in water
Flash point (°C) 81 86 86
Ignition temperature (°C) 558 558 558
Hazard/symptom Prevention/protection First aid
Skin: Irritant, corrosive; severe Protective clothing, gloves, and shoes Wash immediately with large amount
burns and scarring of water; remove contaminated clothing;
obtain medical care
Eyes: Corrosive, strong irritant; Safety glasses Wash with water continuously for 30 min;
lacrimation, redness obtain medical care immediately
Inhalation: Irritation and burning of Adequate ventilation, use of respirators Remove from exposure; provide fresh air
nasal membrane; burning in nose and adequate ventilation; obtain medical
and throat care
Ingestion: Irritation and burning of Store in properly sealed containers; Rinse mouth; obtain immediate medical care
mouth, throat, and gastrointestinal avoid contact with food or beverages
tract; vomiting
Environment: Toxic for aquatic and Avoid environmental releases from
terrestrial organisms industrial processes and accidental spills
Cresols ( o, m, p isomers) (cont'd)
Spillage Storage Fire and explosion
Evacuate and ventilate area; collect Store away from food and incompatible Flash-point 81-86°C; may be explosive in
leaking liquid in sealed containers; materials (metals) in well-labelled reactions with metals; do not use near open
absorb liquid in earth, sand, or inert containers and well-ventilated areas; fires; poisonous gas may be produced
material; wear protective clothing sources of ignition/open flames, and in a fire
smoking, must be prohibited
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information in the following table has been extracted from the
International Register of Potentially Toxic chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should be verified with the appropriate regulatory authorities before
application.
7.1 Exposure Limit Values
Values for exposure limits are given in the following table.
7.2 Specific Restrictions
Approved in the USA for use as a pesticide.
7.3 Labelling, Packaging, and Transport
In the USA, cresols (all isomers) have been designated as a hazardous
substance R1987 and must be managed according to federal and/or state
hazardous waste regulations.
7.4 Waste Disposal
In the USA, any sealed waste must be listed as hazardous waste,
subject to handling, transport, storage, and disposal requirements,
specified by CERLA, and the Clean Waste Act (effective date 1991).
These regulations apply to all discharges.
As a commercial chemical, cresols must be identified as "toxic waste",
subject to restrictions and disposal regulations, permitting, and
notification (effective date 1989).
Any discharge into surface waters requires a permit under the Clean
Water Act (effective date 1987).
Exposure limit values
Medium Specification Country/ Exposure limit Valueb Effective
organization descriptiona date
Air occupational Argentina 8-h TWA 22 mg/m3 May 1991
Czech Republic TWA 20 mg/m3 1985
CLV 40 mg/m3 1985
Germany 8-h TWA 22 mg/m3 1991
5-min STEL 44 mg/m3
Japan TWA 22 mg/m3 1991
Russian TWA 0.5 mg/m3 1989
Federation CLV 1.5 mg/m3 1989
United Kingdom 8-h TWA 22 mg/m3 1992
USA TWA 22 mg/m3 1989
(ACGIH)
Air ambient Russian 0.02 mg/m3( m-, p-cresols) 1983
Federation 0.028 mg/m3 ( o-cresol)
a TWA: time-weighted average;
CLV: ceiling value;
STEL: short-term exposure limit.
b Refers to o-, m-, p-cresols unless otherwise specified.
BIBLIOGRAPHY
ATSDR (1992) Toxicological profile for cresols. US Department of
Health & Human Services, Agency for Toxic Substances and Disease
Registry.
CEFIC (1979) Transport emergency cards for individual products.
Chemical Industries Association Ltd.
IPCS (1995) Environmental Health Criteria 168: Cresols. Geneva,
World Health Organization.
IRPTC (1994) Data profile (legal file) on cresols. Geneva,
International Register of Potentially Toxic Chemicals.
Sax NI & Lewis RJ (1989) Dangerous properties of industrial
materials, 7th ed. New York, Van Nostrand Reinhold Company.
US NTP (1992) Toxicity studies of cresols (CAS nos. 95-48-7,
108-39-4, 106-44-5) in F344/N rats and B6C3F1 mice (feed studies).
Research Triangle Park, North Carolina, National Toxicology Program.
 o-cresol m-cresol p-cresol
Common synonyms:
2-methyl phenol 3-methyl phenol 4-methyl phenol
2-hydroxy toluene 3-hydroxy toluene 4-hydroxyl toluene
o-cresylic acid m-cresylic acid p-cresylic acid
o-kresol (German) m-kresol (German) p-kresol(German)
Chemical formula: C7H8O
Common trade name: cresylic acid (mixture of the three isomers)
cresylic acids (mixture of phenols)
tricresol (mixture of o, m, p- isomers)
dicresol (mixture of m- and p- isomers)
lysol (mixture of 60 cresol: 40 soap)
CAS registry
number:
o-cresol m-cresol p-cresol
Common synonyms:
2-methyl phenol 3-methyl phenol 4-methyl phenol
2-hydroxy toluene 3-hydroxy toluene 4-hydroxyl toluene
o-cresylic acid m-cresylic acid p-cresylic acid
o-kresol (German) m-kresol (German) p-kresol(German)
Chemical formula: C7H8O
Common trade name: cresylic acid (mixture of the three isomers)
cresylic acids (mixture of phenols)
tricresol (mixture of o, m, p- isomers)
dicresol (mixture of m- and p- isomers)
lysol (mixture of 60 cresol: 40 soap)
CAS registry
number:
