
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 65
FENITROTHION
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1991
This is a companion volume to Environmental Health Criteria 133:
Fenitrothion
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Fenitrothion : health and safety guide.
(Health and safety guide ; no. 65)
1.Fenitrothion - adverse effects 2.Fenitrothion - poisoning
3.Fenitrothion - standards 4.Environmental exposure I.Series
ISBN 92 4 151065 X (NLM Classification: WA 240)
ISSN 0259-7268
(c) World Health Organization 1991
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Exposure
2.2. Uptake, metabolism, and excretion
2.3. Effects on organisms in the environment
2.4. Effects on experimental animals and in vitro test systems
2.5. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 Medical treatment
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
6.1. Previous evaluations by international bodies
6.2. Exposure limit values
6.3. Specific restrictions
6.4. Labelling, packaging, and transport
6.5. Waste disposal
BIBLIOGRAPHY
ANNEX 1. TREATMENT OF ORGANOPHOSPHATE INSECTICIDE POISONING IN MAN
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a table of human health hazards,
preventive and protective measures, and first aid, which should be
readily available, and should be clearly explained, to all who could
come into contact with the chemical. The section on regulatory
information has been extracted from the legal file of the
International Register of Potentially Toxic Chemicals (IRPTC) and from
other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
(a) Primary constituent
Common name: fenitrothion
Chemical formula: C9H12NO5PS
Chemical structure:
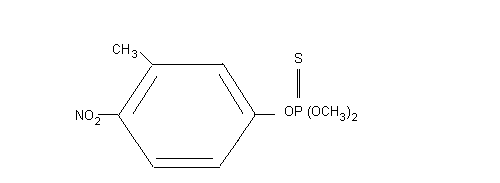 Relative molecular mass: 277.25
CAS chemical name: O,O-dimethyl- O-(3-methyl-4-nitrophenyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
thiophosphate
CAS Registry number: 122-14-5
RTECS Registry number: TG0350000
Synonyms: Bayer 41831, Bayer S 5660, Sumitomo 1102A
(b) Technical product
Major trade names: Aceothion, Agrothion, Cytel, Cyfen, Dybar,
Fenitox, Folithion, MEP, Metathion,
Novathion, Nuvanol, Sumithion
Purity: 93% (Sumithion)
Impurities: O,O-dimethyl- O-3-nitro- m-tolyl-
phosphorothioate <1.5%
O-methyl O,O-bis(4-nitro- m-tolyl)-
phosphorothioate <2.5%
O-methyl S-methyl- O-(4-nitro- m-tolyl)-
phosphorothioate ( S-isomer) <0.8%
O,O-dimethyl- O-2-nitro- m-tolyl-
phosphorothioate <3.0%
O,O-dimethyl- O-6-nitro- m-tolyl-
phosphorothioate <2.5%
O,O-dimethyl- O-2,4-dinitro- m-tolyl-
phosphorothioate <2.0%
O,O-dimethyl- O-4,6-dinitro- m-tolyl-
phosphorothioate <1.5%
3-methyl-4-nitrophenol <0.5%
Isomeric composition: S-isomer <0.8%
1.2 Physical and Chemical Properties
Fenitrothion is a yellow-brown liquid with a "chemical" odour. It is
poorly soluble in water, but highly soluble in alcohols, esters,
ketones, and aromatic hydrocarbons. It is stable at room temperature,
but decomposes at temperatures exceeding 145°C. It is hydrolysed by
alkali: the half-time in 0.01N NaOH at 30°C is 4 h.
Some physical properties are given in Table 1.
1.3 Analytical Methods
The common analytical procedure for the determination of residues in
foods and environmental media consists of : (1) extraction, (2)
partition, (3) chromatographic separation (clean-up), and (4)
qualitative and quantitative analysis using chromatographic methods.
Fenitrothion levels in technical products and formulations are mainly
determined using : the diazo method, colorimetry, or gas-liquid
chromatography. The usual procedure consists of: (1) dissolution of
the extract, (2) separation of impurities, and (3) determination.
Table 1. Physical properties of fenitrothion
Melting point 0.3°C
Boiling point 140-145°C (decomp.) /0.1 mmHg
Flash point 157°C
Explosion limit not available
Vapour pressure 18 mPa at 20°C; 6 x 10-6 mmHg at 20°C
25 25
Density d 1.32-1.34; d 1.3227
25 4
Vapour density not available
n-Octanol/water partition
coefficient (log Pow) 3.16
Solubility in water (30°C) 14 mg/litre
Solubility in organic highly soluble in alcohols, esters,
solvents ketones, and aromatic hydrocarbons:
1000 g/kg in dichloromethane, methanol,
xylene; 193 g/kg in propan-2-ol;
42 g/kg in hexane (20-25°C)
Conversion factors (20°C) 1 ppm = 11.5 mg/m3
1 mg/m3 = 0.087 ppm
1.4 Production and Uses
Fenitrothion is an organophosphorous insecticide that has been in use
since 1959. It is used in agriculture to control insects on rice,
cereals, fruits, vegetables, stored grains, and cotton. It is also
used to control insects in forests and for fly, mosquito, and
cockroach control in public health programmes. Fenitrothion is
formulated as an emulsifiable concentrate, ultra-low-volume
concentrate, powder, granules, dust, oil-based spray, and in
combination with other pesticides. Between 15 000 and 20 000 tonnes
of fenitrothion are produced per year.
2. SUMMARY AND EVALUATION
2.1 Exposure
Fenitrothion enters the air through volatilization from contaminated
surfaces and may drift beyond the intended target area during
spraying. It leaches very slowly from most soils, but some run-off
can occur.
Fenitrothion is degraded by photolysis and hydrolysis. In the
presence of ultraviolet radiation (UVR) or sunlight, the half-life of
fenitrothion in water is less than 24 h. The presence of microflora
may also accelerate degradation. Thus, fenitrothion is stable in
water only in the absence of sunlight or microbial contamination. In
soil, biodegradation is the primary route of degradation, though
photolysis may also play a role.
Airborne concentrations of fenitrothion can be as high as 5 µg/m3,
directly after spraying, but decrease markedly with time and with
distance from the site of application. Levels in water may be as high
as 20 µg/litre, but decrease rapidly.
Bioconcentration factors for fenitrothion with continuing exposure
have been estimated to range from 20 to 450 for a number of different
aquatic species.
Levels of fenitrothion residues in fruits, vegetables, and cereal
grains may range from 0.001 to 9.5 mg/kg immediately after treatment,
but decline rapidly with a half-life of 1-2 days.
2.2 Uptake, Metabolism, and Excretion
Fenitrothion is rapidly absorbed from the intestinal tract of
experimental animals and distributed to various body tissues. The
half-life for the dermal absorption of fenitrothion in the monkey was
found to be 15-17 h. Fenitrothion has been shown to be metabolized
through the major pathways of O-demethylation and by cleavage of the
the P-O-aryl bond. The nitro group of fenitrothion is reduced by
intestinal microorganisms, in ruminants only. The major route of
excretion is via the urine, most of the metabolites being excreted
within 2-4 days in rats, guinea-pigs, mice, and dogs.
The major metabolites that have been reported include demethyl
fenitrothion, demethyl fenitrooxon, dimethylphosphorothioic acid,
dimethyl phosphoric acid, and 3-methyl-4-nitrophenol and its
conjugates. Differences in the composition of the metabolites found
among laboratory animals and between sexes of the same species appear
to be mainly quantitative in nature. Only rabbits appear to excrete
fenitrooxon and aminofenitrooxon in small, though quantifiable,
amounts in the urine.
Evidence from rabbits and dogs showed the preferential deposition of
fenitrothion in adipose tissue.
Residues found in the milk of cows following exposure to fenitrothion
were not detected two days later.
Though fenitrothion is readily absorbed via the oral route, it is
rapidly metabolized and excreted, and is unlikely to remain in the
body for any prolonged period.
2.3 Effects on Organisms in the Environment
At concentrations likely to be found in the environment, fenitrothion
has no effect on microorganisms in soil or water.
Fenitrothion is highly toxic for aquatic invertebrates in both
freshwater and seawater with LC50 values of a few µg/litre for most
species tested. The no-observed-effect level (NOEL) for Daphnia in
48-h tests was <2 µg/litre; in life-cycle tests, a maximum acceptable
toxicant concentration (MATC) of 0.14 µg/litre was established. Field
observations and studies on experimental ponds showed effects on
populations of invertebrates. However, most of the changes observed
were temporary, even at concentrations considerably higher than those
likely to occur after recommended usage.
Fish are less sensitive to fenitrothion than invertebrates and show
96-h LC50 values in the range of 1.7-10 mg/litre. The early larval
stage is the most sensitive. Long-term studies have established a
MATC at, or above, 0.1 mg/litre for 3 species of freshwater fish. In
field studies, in which fenitrothion was applied in forests, no
effects were observed on wild populations of fish or on the survival
of caged test fish with measured water concentrations of fenitrothion
of up to 0.018 mg/litre. Repeated applications of fenitrothion in
forests did not have any effects on fish populations.
Amphibian tadpoles showed LC50 values in the range of
1.2-15 mg/litre in laboratory tests. No field effects were seen after
forest spraying at 140 g/ha.
Fenitrothion is highly toxic for bees (topical LD50 at
0.03-0.04 µg/bee). Large numbers of honey bees and other species have
been reported killed locally in the field. However, the total numbers
of honey bees killed represented only a small percentage of the hive
population.
The acute oral LD50 values for birds ranged between 25 and
1190 mg/kg body weight and most 5-day dietary LC50s exceeded
5000 mg/kg diet. NOEL values for reproduction were 10 mg/kg body
weight for the quail and 100 mg/kg body weight for the mallard.
Song-bird deaths, which occurred soon after the application of
fenitrothion at a rate of 280 g/ha, were markedly higher at 560 g/ha,
for species living in the forest canopy. After spraying at 420 g/ha
followed by 210 g/ha a few days later, the reproductive success of
white-throated sparrows was reduced. In many studies, song-birds
showed inhibition of ChE, soon after the spraying of forests with
fenitrothion.
Field observations did not reveal any effects of fenitrothion on
populations of small, wild mammals.
2.4 Effects on Experimental Animals and In Vitro Test Systems
Fenitrothion is an organophosphate and causes cholinesterase activity
depression in plasma, red blood cells, and brain and liver tissues.
It is metabolized to fenitrooxon, which is more acutely toxic. Its
toxicity may be potentiated by some other organophosphate compounds.
Fenitrothion is an insecticide of moderate toxicity with oral LD50
values in rats and mice ranging from 330 to 2400 mg/kg body weight.
The acute dermal toxicity in rodents ranged from 890 mg/kg body weight
to more than 2500 mg/kg body weight.
Fenitrothion is only minimally irritating to the eyes and is
nonirritating to the skin. The dermal sensitizing potential of the
chemical was shown in one of two studies on guinea-pigs.
Fenitrothion has been tested in short-term studies on rats, dogs, and
rabbits and in long-term carcinogenicity studies on rats and mice. In
short-term studies on rats and dogs, the NOAELs, based on brain ChE
activity, were 10 mg/kg diet and 50 mg/kg diet, respectively.
Long-term studies on rats and mice indicated a NOAEL, based on brain
ChE activity, of 10 mg/kg diet.
No carcinogenic effects were found in any of the long-term studies
reported.
Fenitrothion was not mutagenic in in vivo tests, with the exception
of two in which near lethal dosages were used.
Fenitrothion was found not to be teratogenic at levels of up to
30 mg/kg body weight in rabbits and up to 25 mg/kg body weight in
rats. Dose levels exceeding 8 mg/kg body weight were maternally toxic
in the rat.
Developing young rats exhibited behavioural deficits post-natally
following in utero exposure. A NOEL for this effect was established
at 5 mg/kg body weight per day.
Multigeneration reproduction studies in rats did not indicate any
morphological effects. NOAELs of 10 and 120 mg/kg diet, based on
reproductive parameters, were demonstrated in different studies.
Delayed neurotoxicity has not been reported from exposure to
fenitrothion.
2.5 Effects on Human Beings
Administration of fenitrothion to human volunteers in the form of a
single oral dose of 0.042-0.33 mg/kg body weight or repeated doses of
0.04-0.08 mg/kg body weight did not cause inhibition of plasma- and
erythrocyte-ChE. The urinary excretion of a metabolite, 3-methyl-
4-nitrophenol, was complete within 24 h.
Several cases of poisoning have occurred. The signs and symptoms were
those of parasympathic stimulation. In a few cases, the onset of
toxic manifestations was delayed and they recurred for up to a few
months. It has been suggested that the slow release of the
insecticide from the adipose tissue can give rise to a protracted
clinical course or late symptoms of intoxication. In some cases,
contact dermatitis has been attributed to exposure to this
insecticide.
There is no evidence of delayed neurotoxicity or of an association
with Reye's syndrome following exposure to fenitrothion.
In some programmes of the World Health Organization, indoor residual
spraying of fenitrothion has been used in a few countries for malaria
control (application dose 2.0 g active ingredient/m2). No evidence
of toxicity was noted among the thousands of inhabitants observed,
except in one study in which less than 2% of the inhabitants reported
mild complaints. However, approximately 30% of the spray applicators
showed up to 50% inhibition of whole-blood ChE activity. Following
the aerial application of a 50% EC formulation, some workers developed
symptoms of poisoning and decreased whole-blood ChE activity, within
48 h. Occupational exposure of male workers in a production plant for
a period of over 5 years and female workers in the packaging unit
produced clinical signs and symptoms of poisoning in 15% of the male
and 33% of the female workers. The air concentrations of fenitrothion
measured in the workplace ranged between 0.028 and 0.118 mg/m3.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Fenitrothion is a moderately toxic organophosphorus ester insecticide.
However, overexposure through handling during manufacture or use, or
through accidental or intentional ingestion, may cause serious
poisoning.
Exposure of the general population, mainly through agricultural and
forestry practices and public health programmes, should not constitute
a health hazard.
With good work practices, hygienic measures, and safety precautions,
fenitrothion is unlikely to present a hazard to those occupationally
exposed.
Despite its high toxicity for non-target arthropods, fenitrothion has
been extensively used for pest control with few, or no, adverse
effects on populations in the environment.
3.2 Recommendations
For the health and welfare of workers and the general population, the
handling and application of fenitrothion should only be entrusted to
competently supervised and well-trained operators, who will follow
adequate safety measures and use fenitrothion according to good
application practices.
The manufacture, formulation, use, and disposal of fenitrothion should
be carefully managed to minimize the contamination of the environment,
particularly of surface waters.
Regularly exposed workers should undergo periodic health evaluations.
The rates of application of fenitrothion should be limited, to avoid
effects on non-target arthropods. The insecticide should never be
sprayed over water bodies or streams.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
Fenitrothion is an organophosphorus insecticide. Technical
fenitrothion and concentrated formulations are moderately toxic for
animals (acute oral LD50 for the rat, approximately 503 mg/kg), but
can be hazardous for human beings, if incorrectly handled.
Fenitrothion is hazardous through ingestion and skin contact, because
of fast absorption. Typical signs and symptoms of organophosphorus
poisoning may occur rapidly with over-exposure.
The human health hazards associated with certain types of exposure to
fenitrothion, together with preventive and protective measures and
first aid recommendations are listed in Table 2.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
Fenitrothion is an indirect inhibitor of cholinesterase. In the body,
it has first to be transformed into the active esterase inhibitor
fenitrooxon. As a result, signs and symptoms of overexposure develop
after a latent period and may continue to increase after exposure has
been discontinued. Initially, there may be feelings of exhaustion,
headache, weakness, muscle fasciculations in the face, and confusion.
Nausea, vomiting, abdominal pain, excessive sweating, pinpoint pupils,
and salivation may develop. Difficulty in breathing may be
experienced, due to either congestion of the lungs or weakness of the
respiratory muscles. In severe cases of poisoning, muscle spasms,
unconsciousness, and convulsions may develop. Death may result from
respiratory failure.
For a more complete treatise on the effects of organophosphorus
insecticides, especially their short- and long-term effects on the
nervous system, refer to Environmental Health Criteria 63:
Organophosphorus insecticides - a general introduction.
Table 2
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: readily absorbed via Avoid exposure
skin, ingestion, and inhalation;
may cause organophosphate
poisoning: weakness, headache,
vomiting, excessive sweating and
salivation, pinpoint pupils; in
severe cases: convulsions,
unconsciousness, and death due
to respiratory paralysis
SKIN: irritation; redness; extensive Wear PVC or neoprene gloves and Remove and wash contaminated
contamination may cause poisoning apron; rubber boots clothing; wash contaminated skin
with water and soap; obtain medical
attention immediately
EYES: irritation; redness Wear safety goggles or face shield Flush eyes with clean water for at
least 15 min; if irritation persists,
obtain medical attention immediately
INHALATION: overexposure may Avoid breathing the vapour; use In case of signs and symptoms,
cause poisoning proper (exhaust) ventilation or remove from contaminated area and
suitable respiratory protection obtain medical attention immediately
INGESTION: an unlikely Wash hands before eating, drinking,
occupational hazard using the toilet, and after work
Accidental or intentional ingestion Obtain medical attention immediately;
may rapidly lead to severe poisoning if breathing has stopped, apply
artificial respiration; induce
vomiting, except in the case of
an emulsifiable concentrate
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
REPEATED EXPOSURE VIA As above As above
INHALATION OR INGESTION,
OR THROUGH SKIN may gradually
lead to signs and symptoms of
inhibition of cholinesterase activity
4.1.1.2 Medical treatment
If fenitrothion has been ingested and the formulation did not contain
petroleum distillates, induce vomiting, or preferably perform gastric
lavage using 5% sodium bicarbonate. In the case of ingestion of
liquid formulations containing hydrocarbon solvents, vomiting involves
a risk of aspiration pneumonia. Instead, the stomach should be
emptied as soon as possible by careful gastric lavage (with a cuffed
endotracheal tube already in place). If possible, identify the
solvents present in the formulation and observe the victim for
additional toxic effects. As early as possible, administer 2 mg of
atropine sulfate iv and 1000-2000 mg pralidoxime chloride or 250 mg
obidoxime chloride (adult dose) im or iv to patients suffering from
severe respiratory difficulties, convulsions, or unconsciousness.
Repeated doses of 2 mg atropine sulfate should be given, as required,
based on the respiration, blood pressure, pulse frequency, salivation,
and convulsion conditions. For children, the doses are 0.04-0.08 mg
atropine/kg body weight, 250 mg pralidoxime chloride per child, or
4-8 mg obidoxime chloride/kg body weight.
Artificial respiration should be applied, if required.
Morphine, barbiturates, phenothiazine derivatives, tranquillizers, and
all kinds of central stimulants are contraindicated.
The diagnosis of intoxication should be confirmed, as soon as
possible, by the determination of the cholinisterase (ChE) activity in
venous blood.
For more information on the treatment of poisoning with
organophosphorus insecticides see Environmental Health Criteria 63:
Organophosphorus insecticides - a general introduction. The section
on therapy from this publication is attached as Annex I of this guide.
4.1.2 Health surveillance advice
Occupational exposure to organophosphorus insecticides can be
monitored by measurement of erythrocyte- and whole blood-ChE activity.
Simple monitoring kits are available for this purpose. Physiological
variations in erythrocyte- and blood-ChE values occur in healthy
persons.
Inhibition of acetyl-cholinesterase (AChE) or ChE activity of less
than 20-25% is considered diagnostic of exposure, but not necessarily
indicative of hazard. However, work procedures and hygiene should be
checked. Inhibition of 30-50% or more is considered an indication
that an exposed individual should be removed from further contact with
ChE-inhibiting pesticides, until values return to normal. Work
procedures and hygiene should also be checked.
4.2 Explosion and Fire Hazards
Liquid formulations may be flammable. Inform the fire service that
skin contamination and the breathing of fumes must be avoided.
Protective clothing and self-contained breathing apparatus must be
worn.
Extinguish fires with alcohol-resistant foam or powder. The use of
water spray should be confined to the cooling of unaffected stock, to
avoid polluted runoff from the site.
4.3 Storage
Technical fenitrothion and its formulations should be stored in
locked, well-ventilated buildings, preferably specifically used for
insecticide storage. Do not expose to direct sunlight. Keep products
out of reach of children and unauthorized personnel. Do not store
near animal feed or foodstuffs.
4.4 Transport
Comply with any local regulations regarding the movement of hazardous
goods. Do not load with animal feed or foodstuffs. Check that
containers are sound and labels undamaged before despatch.
4.5 Spillage and Disposal
4.5.1 Spillage
Stay upwind, avoid skin contamination and inhalation of vapour.
Absorb spilled liquid and cover contaminated areas with a 1:3 mixture
of sodium carbonate crystals and damp sawdust, lime, sand, or earth.
Sweep up and place in a closeable impervious container. Ensure that
the container is tightly closed and suitably labelled before transfer
to a safe place for disposal.
Prevent liquid from spreading and contaminating other cargo,
vegetation, or waterways by constructing a barrier of the most
suitable material available, e.g., earth or sand. If spillage into a
waterway occurs and the fenitrothion-containing material is immiscible
with water and sinks, dam the waterway to stop the flow and to retard
dissipation by water movement. Use a bottom pump, dredging, or
underwater vacuum equipment to remove undissolved material.
Empty any of the product remaining in the damaged/leaking container
into a clean empty container, which should then be tightly closed and
suitably labelled.
Decontaminate emptied containers with a 10% sodium carbonate solution
added at the rate of at least 1 litre per 20-litre drum. Swirl round
to rinse walls, empty, and add rinsings to absorbent, such as sawdust,
sand, or earth. Puncture empty containers to prevent re-use.
4.5.2 Disposal
Contaminated absorbents, containers, and surplus product, should be
burnt in a proper incinerator at high temperatures in a unit with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, or in an area where there is no risk of contamination
of surface or ground water. Before burying, mix liberally with sodium
carbonate (washing soda) crystals, to help neutralize the product, and
mix with soil rich in organic matter. Comply with any local
legislation.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Fenitrothion is moderately toxic for birds and fish, but highly toxic
for arthropods. It does not bioaccumulate and breaks down rapidly in
the environment.
Avoid contamination of the soil, water, and the atmosphere by proper
methods of storage, transport, handling, and waste disposal.
In case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
6.1 Previous Evaluations by International Bodies
Fenitrothion was evaluated by the Joint FAO/WHO Meeting on Pesticide
Residues (JMPR) in 1969, 1974, 1976, 1977, 1979, 1982, 1983, 1984,
1986, 1987, 1988, and 1989. In 1988, the JMPR established an
Acceptable Daily Intake (ADI) for man of 0-0.005 mg/kg body weight.
For Codex Maximum Residue Limits (MRLs) see Table 3.
The International Programme on Chemical Safety (WHO) has classified
technical fenitrothion as "moderately hazardous" (Class II). WHO has
issued a data sheet on fenitrothion (No. 30).
6.2 Exposure Limit Values
Some exposure limit values are given in Table 3.
6.3 Specific Restrictions
Fenitrothion has been approved for use as a pesticide in many
countries. In some countries, specific uses are defined as well as
limitations and precautions.
6.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
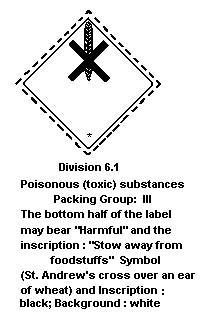
Dangerous Goods classifies fenitrothion in:
Hazard Class 6.1: poisonous substance;
Packing Group III: a substance presenting a relatively low risk
of poisoning in transport, for material
containing 48% (liquid).
The label should be as follows:
Relative molecular mass: 277.25
CAS chemical name: O,O-dimethyl- O-(3-methyl-4-nitrophenyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
thiophosphate
CAS Registry number: 122-14-5
RTECS Registry number: TG0350000
Synonyms: Bayer 41831, Bayer S 5660, Sumitomo 1102A
(b) Technical product
Major trade names: Aceothion, Agrothion, Cytel, Cyfen, Dybar,
Fenitox, Folithion, MEP, Metathion,
Novathion, Nuvanol, Sumithion
Purity: 93% (Sumithion)
Impurities: O,O-dimethyl- O-3-nitro- m-tolyl-
phosphorothioate <1.5%
O-methyl O,O-bis(4-nitro- m-tolyl)-
phosphorothioate <2.5%
O-methyl S-methyl- O-(4-nitro- m-tolyl)-
phosphorothioate ( S-isomer) <0.8%
O,O-dimethyl- O-2-nitro- m-tolyl-
phosphorothioate <3.0%
O,O-dimethyl- O-6-nitro- m-tolyl-
phosphorothioate <2.5%
O,O-dimethyl- O-2,4-dinitro- m-tolyl-
phosphorothioate <2.0%
O,O-dimethyl- O-4,6-dinitro- m-tolyl-
phosphorothioate <1.5%
3-methyl-4-nitrophenol <0.5%
Isomeric composition: S-isomer <0.8%
1.2 Physical and Chemical Properties
Fenitrothion is a yellow-brown liquid with a "chemical" odour. It is
poorly soluble in water, but highly soluble in alcohols, esters,
ketones, and aromatic hydrocarbons. It is stable at room temperature,
but decomposes at temperatures exceeding 145°C. It is hydrolysed by
alkali: the half-time in 0.01N NaOH at 30°C is 4 h.
Some physical properties are given in Table 1.
1.3 Analytical Methods
The common analytical procedure for the determination of residues in
foods and environmental media consists of : (1) extraction, (2)
partition, (3) chromatographic separation (clean-up), and (4)
qualitative and quantitative analysis using chromatographic methods.
Fenitrothion levels in technical products and formulations are mainly
determined using : the diazo method, colorimetry, or gas-liquid
chromatography. The usual procedure consists of: (1) dissolution of
the extract, (2) separation of impurities, and (3) determination.
Table 1. Physical properties of fenitrothion
Melting point 0.3°C
Boiling point 140-145°C (decomp.) /0.1 mmHg
Flash point 157°C
Explosion limit not available
Vapour pressure 18 mPa at 20°C; 6 x 10-6 mmHg at 20°C
25 25
Density d 1.32-1.34; d 1.3227
25 4
Vapour density not available
n-Octanol/water partition
coefficient (log Pow) 3.16
Solubility in water (30°C) 14 mg/litre
Solubility in organic highly soluble in alcohols, esters,
solvents ketones, and aromatic hydrocarbons:
1000 g/kg in dichloromethane, methanol,
xylene; 193 g/kg in propan-2-ol;
42 g/kg in hexane (20-25°C)
Conversion factors (20°C) 1 ppm = 11.5 mg/m3
1 mg/m3 = 0.087 ppm
1.4 Production and Uses
Fenitrothion is an organophosphorous insecticide that has been in use
since 1959. It is used in agriculture to control insects on rice,
cereals, fruits, vegetables, stored grains, and cotton. It is also
used to control insects in forests and for fly, mosquito, and
cockroach control in public health programmes. Fenitrothion is
formulated as an emulsifiable concentrate, ultra-low-volume
concentrate, powder, granules, dust, oil-based spray, and in
combination with other pesticides. Between 15 000 and 20 000 tonnes
of fenitrothion are produced per year.
2. SUMMARY AND EVALUATION
2.1 Exposure
Fenitrothion enters the air through volatilization from contaminated
surfaces and may drift beyond the intended target area during
spraying. It leaches very slowly from most soils, but some run-off
can occur.
Fenitrothion is degraded by photolysis and hydrolysis. In the
presence of ultraviolet radiation (UVR) or sunlight, the half-life of
fenitrothion in water is less than 24 h. The presence of microflora
may also accelerate degradation. Thus, fenitrothion is stable in
water only in the absence of sunlight or microbial contamination. In
soil, biodegradation is the primary route of degradation, though
photolysis may also play a role.
Airborne concentrations of fenitrothion can be as high as 5 µg/m3,
directly after spraying, but decrease markedly with time and with
distance from the site of application. Levels in water may be as high
as 20 µg/litre, but decrease rapidly.
Bioconcentration factors for fenitrothion with continuing exposure
have been estimated to range from 20 to 450 for a number of different
aquatic species.
Levels of fenitrothion residues in fruits, vegetables, and cereal
grains may range from 0.001 to 9.5 mg/kg immediately after treatment,
but decline rapidly with a half-life of 1-2 days.
2.2 Uptake, Metabolism, and Excretion
Fenitrothion is rapidly absorbed from the intestinal tract of
experimental animals and distributed to various body tissues. The
half-life for the dermal absorption of fenitrothion in the monkey was
found to be 15-17 h. Fenitrothion has been shown to be metabolized
through the major pathways of O-demethylation and by cleavage of the
the P-O-aryl bond. The nitro group of fenitrothion is reduced by
intestinal microorganisms, in ruminants only. The major route of
excretion is via the urine, most of the metabolites being excreted
within 2-4 days in rats, guinea-pigs, mice, and dogs.
The major metabolites that have been reported include demethyl
fenitrothion, demethyl fenitrooxon, dimethylphosphorothioic acid,
dimethyl phosphoric acid, and 3-methyl-4-nitrophenol and its
conjugates. Differences in the composition of the metabolites found
among laboratory animals and between sexes of the same species appear
to be mainly quantitative in nature. Only rabbits appear to excrete
fenitrooxon and aminofenitrooxon in small, though quantifiable,
amounts in the urine.
Evidence from rabbits and dogs showed the preferential deposition of
fenitrothion in adipose tissue.
Residues found in the milk of cows following exposure to fenitrothion
were not detected two days later.
Though fenitrothion is readily absorbed via the oral route, it is
rapidly metabolized and excreted, and is unlikely to remain in the
body for any prolonged period.
2.3 Effects on Organisms in the Environment
At concentrations likely to be found in the environment, fenitrothion
has no effect on microorganisms in soil or water.
Fenitrothion is highly toxic for aquatic invertebrates in both
freshwater and seawater with LC50 values of a few µg/litre for most
species tested. The no-observed-effect level (NOEL) for Daphnia in
48-h tests was <2 µg/litre; in life-cycle tests, a maximum acceptable
toxicant concentration (MATC) of 0.14 µg/litre was established. Field
observations and studies on experimental ponds showed effects on
populations of invertebrates. However, most of the changes observed
were temporary, even at concentrations considerably higher than those
likely to occur after recommended usage.
Fish are less sensitive to fenitrothion than invertebrates and show
96-h LC50 values in the range of 1.7-10 mg/litre. The early larval
stage is the most sensitive. Long-term studies have established a
MATC at, or above, 0.1 mg/litre for 3 species of freshwater fish. In
field studies, in which fenitrothion was applied in forests, no
effects were observed on wild populations of fish or on the survival
of caged test fish with measured water concentrations of fenitrothion
of up to 0.018 mg/litre. Repeated applications of fenitrothion in
forests did not have any effects on fish populations.
Amphibian tadpoles showed LC50 values in the range of
1.2-15 mg/litre in laboratory tests. No field effects were seen after
forest spraying at 140 g/ha.
Fenitrothion is highly toxic for bees (topical LD50 at
0.03-0.04 µg/bee). Large numbers of honey bees and other species have
been reported killed locally in the field. However, the total numbers
of honey bees killed represented only a small percentage of the hive
population.
The acute oral LD50 values for birds ranged between 25 and
1190 mg/kg body weight and most 5-day dietary LC50s exceeded
5000 mg/kg diet. NOEL values for reproduction were 10 mg/kg body
weight for the quail and 100 mg/kg body weight for the mallard.
Song-bird deaths, which occurred soon after the application of
fenitrothion at a rate of 280 g/ha, were markedly higher at 560 g/ha,
for species living in the forest canopy. After spraying at 420 g/ha
followed by 210 g/ha a few days later, the reproductive success of
white-throated sparrows was reduced. In many studies, song-birds
showed inhibition of ChE, soon after the spraying of forests with
fenitrothion.
Field observations did not reveal any effects of fenitrothion on
populations of small, wild mammals.
2.4 Effects on Experimental Animals and In Vitro Test Systems
Fenitrothion is an organophosphate and causes cholinesterase activity
depression in plasma, red blood cells, and brain and liver tissues.
It is metabolized to fenitrooxon, which is more acutely toxic. Its
toxicity may be potentiated by some other organophosphate compounds.
Fenitrothion is an insecticide of moderate toxicity with oral LD50
values in rats and mice ranging from 330 to 2400 mg/kg body weight.
The acute dermal toxicity in rodents ranged from 890 mg/kg body weight
to more than 2500 mg/kg body weight.
Fenitrothion is only minimally irritating to the eyes and is
nonirritating to the skin. The dermal sensitizing potential of the
chemical was shown in one of two studies on guinea-pigs.
Fenitrothion has been tested in short-term studies on rats, dogs, and
rabbits and in long-term carcinogenicity studies on rats and mice. In
short-term studies on rats and dogs, the NOAELs, based on brain ChE
activity, were 10 mg/kg diet and 50 mg/kg diet, respectively.
Long-term studies on rats and mice indicated a NOAEL, based on brain
ChE activity, of 10 mg/kg diet.
No carcinogenic effects were found in any of the long-term studies
reported.
Fenitrothion was not mutagenic in in vivo tests, with the exception
of two in which near lethal dosages were used.
Fenitrothion was found not to be teratogenic at levels of up to
30 mg/kg body weight in rabbits and up to 25 mg/kg body weight in
rats. Dose levels exceeding 8 mg/kg body weight were maternally toxic
in the rat.
Developing young rats exhibited behavioural deficits post-natally
following in utero exposure. A NOEL for this effect was established
at 5 mg/kg body weight per day.
Multigeneration reproduction studies in rats did not indicate any
morphological effects. NOAELs of 10 and 120 mg/kg diet, based on
reproductive parameters, were demonstrated in different studies.
Delayed neurotoxicity has not been reported from exposure to
fenitrothion.
2.5 Effects on Human Beings
Administration of fenitrothion to human volunteers in the form of a
single oral dose of 0.042-0.33 mg/kg body weight or repeated doses of
0.04-0.08 mg/kg body weight did not cause inhibition of plasma- and
erythrocyte-ChE. The urinary excretion of a metabolite, 3-methyl-
4-nitrophenol, was complete within 24 h.
Several cases of poisoning have occurred. The signs and symptoms were
those of parasympathic stimulation. In a few cases, the onset of
toxic manifestations was delayed and they recurred for up to a few
months. It has been suggested that the slow release of the
insecticide from the adipose tissue can give rise to a protracted
clinical course or late symptoms of intoxication. In some cases,
contact dermatitis has been attributed to exposure to this
insecticide.
There is no evidence of delayed neurotoxicity or of an association
with Reye's syndrome following exposure to fenitrothion.
In some programmes of the World Health Organization, indoor residual
spraying of fenitrothion has been used in a few countries for malaria
control (application dose 2.0 g active ingredient/m2). No evidence
of toxicity was noted among the thousands of inhabitants observed,
except in one study in which less than 2% of the inhabitants reported
mild complaints. However, approximately 30% of the spray applicators
showed up to 50% inhibition of whole-blood ChE activity. Following
the aerial application of a 50% EC formulation, some workers developed
symptoms of poisoning and decreased whole-blood ChE activity, within
48 h. Occupational exposure of male workers in a production plant for
a period of over 5 years and female workers in the packaging unit
produced clinical signs and symptoms of poisoning in 15% of the male
and 33% of the female workers. The air concentrations of fenitrothion
measured in the workplace ranged between 0.028 and 0.118 mg/m3.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Fenitrothion is a moderately toxic organophosphorus ester insecticide.
However, overexposure through handling during manufacture or use, or
through accidental or intentional ingestion, may cause serious
poisoning.
Exposure of the general population, mainly through agricultural and
forestry practices and public health programmes, should not constitute
a health hazard.
With good work practices, hygienic measures, and safety precautions,
fenitrothion is unlikely to present a hazard to those occupationally
exposed.
Despite its high toxicity for non-target arthropods, fenitrothion has
been extensively used for pest control with few, or no, adverse
effects on populations in the environment.
3.2 Recommendations
For the health and welfare of workers and the general population, the
handling and application of fenitrothion should only be entrusted to
competently supervised and well-trained operators, who will follow
adequate safety measures and use fenitrothion according to good
application practices.
The manufacture, formulation, use, and disposal of fenitrothion should
be carefully managed to minimize the contamination of the environment,
particularly of surface waters.
Regularly exposed workers should undergo periodic health evaluations.
The rates of application of fenitrothion should be limited, to avoid
effects on non-target arthropods. The insecticide should never be
sprayed over water bodies or streams.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
Fenitrothion is an organophosphorus insecticide. Technical
fenitrothion and concentrated formulations are moderately toxic for
animals (acute oral LD50 for the rat, approximately 503 mg/kg), but
can be hazardous for human beings, if incorrectly handled.
Fenitrothion is hazardous through ingestion and skin contact, because
of fast absorption. Typical signs and symptoms of organophosphorus
poisoning may occur rapidly with over-exposure.
The human health hazards associated with certain types of exposure to
fenitrothion, together with preventive and protective measures and
first aid recommendations are listed in Table 2.
4.1.1 Advice to physicians
4.1.1.1 Symptoms of poisoning
Fenitrothion is an indirect inhibitor of cholinesterase. In the body,
it has first to be transformed into the active esterase inhibitor
fenitrooxon. As a result, signs and symptoms of overexposure develop
after a latent period and may continue to increase after exposure has
been discontinued. Initially, there may be feelings of exhaustion,
headache, weakness, muscle fasciculations in the face, and confusion.
Nausea, vomiting, abdominal pain, excessive sweating, pinpoint pupils,
and salivation may develop. Difficulty in breathing may be
experienced, due to either congestion of the lungs or weakness of the
respiratory muscles. In severe cases of poisoning, muscle spasms,
unconsciousness, and convulsions may develop. Death may result from
respiratory failure.
For a more complete treatise on the effects of organophosphorus
insecticides, especially their short- and long-term effects on the
nervous system, refer to Environmental Health Criteria 63:
Organophosphorus insecticides - a general introduction.
Table 2
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: readily absorbed via Avoid exposure
skin, ingestion, and inhalation;
may cause organophosphate
poisoning: weakness, headache,
vomiting, excessive sweating and
salivation, pinpoint pupils; in
severe cases: convulsions,
unconsciousness, and death due
to respiratory paralysis
SKIN: irritation; redness; extensive Wear PVC or neoprene gloves and Remove and wash contaminated
contamination may cause poisoning apron; rubber boots clothing; wash contaminated skin
with water and soap; obtain medical
attention immediately
EYES: irritation; redness Wear safety goggles or face shield Flush eyes with clean water for at
least 15 min; if irritation persists,
obtain medical attention immediately
INHALATION: overexposure may Avoid breathing the vapour; use In case of signs and symptoms,
cause poisoning proper (exhaust) ventilation or remove from contaminated area and
suitable respiratory protection obtain medical attention immediately
INGESTION: an unlikely Wash hands before eating, drinking,
occupational hazard using the toilet, and after work
Accidental or intentional ingestion Obtain medical attention immediately;
may rapidly lead to severe poisoning if breathing has stopped, apply
artificial respiration; induce
vomiting, except in the case of
an emulsifiable concentrate
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
REPEATED EXPOSURE VIA As above As above
INHALATION OR INGESTION,
OR THROUGH SKIN may gradually
lead to signs and symptoms of
inhibition of cholinesterase activity
4.1.1.2 Medical treatment
If fenitrothion has been ingested and the formulation did not contain
petroleum distillates, induce vomiting, or preferably perform gastric
lavage using 5% sodium bicarbonate. In the case of ingestion of
liquid formulations containing hydrocarbon solvents, vomiting involves
a risk of aspiration pneumonia. Instead, the stomach should be
emptied as soon as possible by careful gastric lavage (with a cuffed
endotracheal tube already in place). If possible, identify the
solvents present in the formulation and observe the victim for
additional toxic effects. As early as possible, administer 2 mg of
atropine sulfate iv and 1000-2000 mg pralidoxime chloride or 250 mg
obidoxime chloride (adult dose) im or iv to patients suffering from
severe respiratory difficulties, convulsions, or unconsciousness.
Repeated doses of 2 mg atropine sulfate should be given, as required,
based on the respiration, blood pressure, pulse frequency, salivation,
and convulsion conditions. For children, the doses are 0.04-0.08 mg
atropine/kg body weight, 250 mg pralidoxime chloride per child, or
4-8 mg obidoxime chloride/kg body weight.
Artificial respiration should be applied, if required.
Morphine, barbiturates, phenothiazine derivatives, tranquillizers, and
all kinds of central stimulants are contraindicated.
The diagnosis of intoxication should be confirmed, as soon as
possible, by the determination of the cholinisterase (ChE) activity in
venous blood.
For more information on the treatment of poisoning with
organophosphorus insecticides see Environmental Health Criteria 63:
Organophosphorus insecticides - a general introduction. The section
on therapy from this publication is attached as Annex I of this guide.
4.1.2 Health surveillance advice
Occupational exposure to organophosphorus insecticides can be
monitored by measurement of erythrocyte- and whole blood-ChE activity.
Simple monitoring kits are available for this purpose. Physiological
variations in erythrocyte- and blood-ChE values occur in healthy
persons.
Inhibition of acetyl-cholinesterase (AChE) or ChE activity of less
than 20-25% is considered diagnostic of exposure, but not necessarily
indicative of hazard. However, work procedures and hygiene should be
checked. Inhibition of 30-50% or more is considered an indication
that an exposed individual should be removed from further contact with
ChE-inhibiting pesticides, until values return to normal. Work
procedures and hygiene should also be checked.
4.2 Explosion and Fire Hazards
Liquid formulations may be flammable. Inform the fire service that
skin contamination and the breathing of fumes must be avoided.
Protective clothing and self-contained breathing apparatus must be
worn.
Extinguish fires with alcohol-resistant foam or powder. The use of
water spray should be confined to the cooling of unaffected stock, to
avoid polluted runoff from the site.
4.3 Storage
Technical fenitrothion and its formulations should be stored in
locked, well-ventilated buildings, preferably specifically used for
insecticide storage. Do not expose to direct sunlight. Keep products
out of reach of children and unauthorized personnel. Do not store
near animal feed or foodstuffs.
4.4 Transport
Comply with any local regulations regarding the movement of hazardous
goods. Do not load with animal feed or foodstuffs. Check that
containers are sound and labels undamaged before despatch.
4.5 Spillage and Disposal
4.5.1 Spillage
Stay upwind, avoid skin contamination and inhalation of vapour.
Absorb spilled liquid and cover contaminated areas with a 1:3 mixture
of sodium carbonate crystals and damp sawdust, lime, sand, or earth.
Sweep up and place in a closeable impervious container. Ensure that
the container is tightly closed and suitably labelled before transfer
to a safe place for disposal.
Prevent liquid from spreading and contaminating other cargo,
vegetation, or waterways by constructing a barrier of the most
suitable material available, e.g., earth or sand. If spillage into a
waterway occurs and the fenitrothion-containing material is immiscible
with water and sinks, dam the waterway to stop the flow and to retard
dissipation by water movement. Use a bottom pump, dredging, or
underwater vacuum equipment to remove undissolved material.
Empty any of the product remaining in the damaged/leaking container
into a clean empty container, which should then be tightly closed and
suitably labelled.
Decontaminate emptied containers with a 10% sodium carbonate solution
added at the rate of at least 1 litre per 20-litre drum. Swirl round
to rinse walls, empty, and add rinsings to absorbent, such as sawdust,
sand, or earth. Puncture empty containers to prevent re-use.
4.5.2 Disposal
Contaminated absorbents, containers, and surplus product, should be
burnt in a proper incinerator at high temperatures in a unit with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, or in an area where there is no risk of contamination
of surface or ground water. Before burying, mix liberally with sodium
carbonate (washing soda) crystals, to help neutralize the product, and
mix with soil rich in organic matter. Comply with any local
legislation.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Fenitrothion is moderately toxic for birds and fish, but highly toxic
for arthropods. It does not bioaccumulate and breaks down rapidly in
the environment.
Avoid contamination of the soil, water, and the atmosphere by proper
methods of storage, transport, handling, and waste disposal.
In case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
6.1 Previous Evaluations by International Bodies
Fenitrothion was evaluated by the Joint FAO/WHO Meeting on Pesticide
Residues (JMPR) in 1969, 1974, 1976, 1977, 1979, 1982, 1983, 1984,
1986, 1987, 1988, and 1989. In 1988, the JMPR established an
Acceptable Daily Intake (ADI) for man of 0-0.005 mg/kg body weight.
For Codex Maximum Residue Limits (MRLs) see Table 3.
The International Programme on Chemical Safety (WHO) has classified
technical fenitrothion as "moderately hazardous" (Class II). WHO has
issued a data sheet on fenitrothion (No. 30).
6.2 Exposure Limit Values
Some exposure limit values are given in Table 3.
6.3 Specific Restrictions
Fenitrothion has been approved for use as a pesticide in many
countries. In some countries, specific uses are defined as well as
limitations and precautions.
6.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies fenitrothion in:
Hazard Class 6.1: poisonous substance;
Packing Group III: a substance presenting a relatively low risk
of poisoning in transport, for material
containing 48% (liquid).
The label should be as follows:
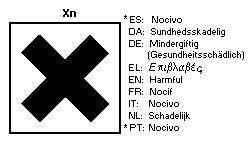
 The European Economic Community legislation requires labelling of
fenitrothion as dangerous substance using the symbol:
The European Economic Community legislation requires labelling of
fenitrothion as dangerous substance using the symbol:
 The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink and
animal feeding stuffs.
The European Economic Community legislation on the labelling of
pesticide preparations classifies fenitrothion in Class IIB for the
purpose of determining the label.
WHO gives the following product specification for technical
fenitrothion for use in public health:
"The material shall consist of fenitrothion together with related
manufacturing compounds and shall be in the form of a yellow to brown
liquid free from extraneous impurities or added modifying agents."
It should contain at least 930 g of fenitrothion per kg. The
specification gives the required acidity and water content and also
analytical methods for checking The specification continues:
"The technical fenitrothion shall be packed in suitable clean
containers, as specified in the order."
"All packages shall bear, durably and legibly marked on the
container, the following:
Manufacturer's name
Technical fenitrothion to specification WHO/SIT/17.R2
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
"Fenitrothion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It may be
absorbed through the skin. Avoid skin contact; wear protective
gloves, clean protective clothing, and a respirator when handling the
material. Wash thoroughly with soap and water after using."
"Keep out of the reach of children and well away from foodstuffs and
animal feed and their containers."
"If poisoning occurs, call a physician. Atropine and pralidoxime are
specific antidotes and artificial respiration may be needed."
Similar specifications and instructions are given for fenitrothion
water-dispersible powder and emulsified concentrate.
FAO gives similar product specifications for fenitrothion for its use
in plant protection. In this case, the technical material should
contain at least 95% active material, and containers must comply with
pertinent national and international transport and safety regulations.
6.5 Waste Disposal
In the USA, any waste containing fenitrothion is considered a
hazardous waste and permits are required for its handling, transport,
treatment, storage or discharge and disposal.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Japan Maximum acceptable concentration (MAC)
- Time-weighted average (TWA) 1 mg/m3a 1986
USSR Maximum allowable concentration (MAC)
- Ceiling value (CLV) (vapour and aerosol) 0.1 mg/m3 1982
Ambient USSR Maximum allowable concentration (MAC)
- average per day 0.001 mg/m3 1982
- 1 × /day 0.005 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.005 mg/kg 1988
body weight
Residue FAO/WHO Maximum residue limit (MRL) 0.002-20 mg/kg 1989
(with its oxygen analogue expressed
as fenitrothion); products specified as follows:
- milks 0.002 mg/kg
- cucumbers, meat, onions, potatoes 0.05 mg/kg
- cauliflower, cocoa beans, egg plants,
peppers, soybeans (dry) 0.1 mg/kg
- bread (white) leeks, radishes 0.2 mg/kg
- apples, cabbage, cabbage red, cherries,
grapes, lettuce, pear, peas,
strawberries, tea (dried, green),
tomatoes 0.5 mg/kg
- peach, rice (polished) 1 mg/kg
- citrus fruit, wheat flour (white),b
processed wheat bran 2 mg/kg
- wheat flour (wholemeal) 5 mg/kg
- cereal grains 10 mg/kg
- raw wheat bran, rice bran unprocessed 20 mg/kg
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Surface USSR Maximum allowable concentration (MAC) 0.25 mg/litre 1983
Fishing USSR Maximum allowable concentration (MAC) 0.00 mg/litre 1983
SOIL USSR Maximum allowable concentration (MAC) 1.0 mg/kg 1984
a Notice : skin absorption.
b Codex 1990.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986a) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in food.
Rome, Food and Agriculture Organization of the United Nations.
FAO/WHO (1986b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues. 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1986c) Codex maximum limits for pesticide residues.
CAC/Vol.XIII - ed. 2, Rome, Codex Alimentarius Commission, Food and
Agriculture Organization of the United Nations, (+ Annex 2 (1990)).
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides,
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) Environmental Health Criteria 63: Organophosphorus
insecticides - a general introduction. Geneva, World Health
Organization, 181 pp.
WHO (1990) The WHO recommended classification of pesticides by hazard
and guidelines to classification, 1990-91. Geneva, World Health
Organization (Unpublished document WHO/PCS/90.1).
WHO (1990) Environmental Health Criteria 133: Fenitrothion. Geneva,
World Health Organization.
WHO/FAO (1977) Fenitrothion. Data sheets on pesticides, No. 30.
Geneva, World Health Organization (Unpublished documents VBC,
DS,77.30).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
ANNEX I
TREATMENT OF ORGANOPHOSPHATE INSECTICIDE POISONING IN MAN
(From EHC 63: Organophosphorus Insecticides - A General Introduction)
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be included
in the IPCS Health and Safety Guides to be prepared for selected
organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the Absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with alkaline
soap or with a sodium bicarbonate solution. Particular care should be
taken in cleaning the skin area where venupuncture is performed.
Blood might be contaminated with direct-acting organophosphorus esters
and, therefore, inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting might be induced, if
the patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml water. This treatment is, however,
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with addition of bicarbonate solution or
activated charcoal) can also be performed, particularly in unconscious
patients, taking care to prevent aspiration of fluids into the lungs
(i.e., only after a tracheal tube has been put in place).
The volume of fluid introduced into the stomach should be recorded and
samples of gastric lavage frozen and stored for subsequent chemical
analysis. If the formulation of the pesticide involved is available,
it should also be stored for further analysis (i.e., detection of
toxicologically relevant impurities). A purgative can be administered
to remove the ingested compound.
I.2 General Supportive Treatment
Artificial respiration (via a tracheal tube) should be started at the
first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general,
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific Pharmacological Treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine treatment
varies from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases and
total daily doses of up to several hundred mg may be necessary during
the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision on whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have beneficial
effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms, which are not affected by atropine.
Doses of 10 mg s.c. or i.v. are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and drugs
that may depress respiration are not recommended in the absence of
artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one time
may cause toxic side-effects. Pulse-rate should not exceed 120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be taken
into, and then released from, fat depots over a period of many days.
It is therefore quite incorrect to abandon oxime treatment after
1-2 days on the supposition that all inhibited enzyme will be aged.
Ecobichon et al. (1977) noted prompt improvement in both condition and
blood-ChEs in response to pralidoxime given on the 11th-15th days
after major symptoms of poisoning appeared due to extended exposure to
fenitrothion (a dimethyl phosphate with a short half-life for aging of
inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in error,
while drunk. Therapeutic dosages were progressively increased up to
6 mg atropine i.v. every 15 min together with continuous i.v. infusion
of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to 6 after
intoxication. After considerable improvement, the patient relapsed
and further aggressive therapy was given at a declining rate from days
10 to 16 (atropine) and to day 23 (oxime), respectively. In total,
92 g of pralidoxime chloride and 3912 mg of atropine were given and
the patient was discharged on the thirty-third day with no apparent
sequelae.
References to Annex I
ECOBICHON, D.J., OZERE, R.L., REID, E., & CROCKER, J.F.S (1977)
Acute fenitrothion poisoning. Can. Med. Assoc. J., 116: 377-379.
KAGAN, JU.S. (1977) [Toxicology of organophosphorus pesticides.]
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
WHO document. VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed. New
York, Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of medical
practitioners, London, United Kingdom Department of Health and Social
Security, pp. 41-47.
VALE, J.A. & SCOTT, G.W. (1974) Organophosphorus poisoning. Guy's
Hosp. Rep., 123: 13-25.
WARRINER, R.A., III, NIES, A.S., & HAYES, W.J., Jr (1977) Severe
organophosphate poisoning complicated by alcohol and terpentine
ingestion. Arch. environ. Health, 32: 203-205.
The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink and
animal feeding stuffs.
The European Economic Community legislation on the labelling of
pesticide preparations classifies fenitrothion in Class IIB for the
purpose of determining the label.
WHO gives the following product specification for technical
fenitrothion for use in public health:
"The material shall consist of fenitrothion together with related
manufacturing compounds and shall be in the form of a yellow to brown
liquid free from extraneous impurities or added modifying agents."
It should contain at least 930 g of fenitrothion per kg. The
specification gives the required acidity and water content and also
analytical methods for checking The specification continues:
"The technical fenitrothion shall be packed in suitable clean
containers, as specified in the order."
"All packages shall bear, durably and legibly marked on the
container, the following:
Manufacturer's name
Technical fenitrothion to specification WHO/SIT/17.R2
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
"Fenitrothion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It may be
absorbed through the skin. Avoid skin contact; wear protective
gloves, clean protective clothing, and a respirator when handling the
material. Wash thoroughly with soap and water after using."
"Keep out of the reach of children and well away from foodstuffs and
animal feed and their containers."
"If poisoning occurs, call a physician. Atropine and pralidoxime are
specific antidotes and artificial respiration may be needed."
Similar specifications and instructions are given for fenitrothion
water-dispersible powder and emulsified concentrate.
FAO gives similar product specifications for fenitrothion for its use
in plant protection. In this case, the technical material should
contain at least 95% active material, and containers must comply with
pertinent national and international transport and safety regulations.
6.5 Waste Disposal
In the USA, any waste containing fenitrothion is considered a
hazardous waste and permits are required for its handling, transport,
treatment, storage or discharge and disposal.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Japan Maximum acceptable concentration (MAC)
- Time-weighted average (TWA) 1 mg/m3a 1986
USSR Maximum allowable concentration (MAC)
- Ceiling value (CLV) (vapour and aerosol) 0.1 mg/m3 1982
Ambient USSR Maximum allowable concentration (MAC)
- average per day 0.001 mg/m3 1982
- 1 × /day 0.005 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.005 mg/kg 1988
body weight
Residue FAO/WHO Maximum residue limit (MRL) 0.002-20 mg/kg 1989
(with its oxygen analogue expressed
as fenitrothion); products specified as follows:
- milks 0.002 mg/kg
- cucumbers, meat, onions, potatoes 0.05 mg/kg
- cauliflower, cocoa beans, egg plants,
peppers, soybeans (dry) 0.1 mg/kg
- bread (white) leeks, radishes 0.2 mg/kg
- apples, cabbage, cabbage red, cherries,
grapes, lettuce, pear, peas,
strawberries, tea (dried, green),
tomatoes 0.5 mg/kg
- peach, rice (polished) 1 mg/kg
- citrus fruit, wheat flour (white),b
processed wheat bran 2 mg/kg
- wheat flour (wholemeal) 5 mg/kg
- cereal grains 10 mg/kg
- raw wheat bran, rice bran unprocessed 20 mg/kg
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Surface USSR Maximum allowable concentration (MAC) 0.25 mg/litre 1983
Fishing USSR Maximum allowable concentration (MAC) 0.00 mg/litre 1983
SOIL USSR Maximum allowable concentration (MAC) 1.0 mg/kg 1984
a Notice : skin absorption.
b Codex 1990.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986a) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in food.
Rome, Food and Agriculture Organization of the United Nations.
FAO/WHO (1986b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues. 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1986c) Codex maximum limits for pesticide residues.
CAC/Vol.XIII - ed. 2, Rome, Codex Alimentarius Commission, Food and
Agriculture Organization of the United Nations, (+ Annex 2 (1990)).
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides,
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) Environmental Health Criteria 63: Organophosphorus
insecticides - a general introduction. Geneva, World Health
Organization, 181 pp.
WHO (1990) The WHO recommended classification of pesticides by hazard
and guidelines to classification, 1990-91. Geneva, World Health
Organization (Unpublished document WHO/PCS/90.1).
WHO (1990) Environmental Health Criteria 133: Fenitrothion. Geneva,
World Health Organization.
WHO/FAO (1977) Fenitrothion. Data sheets on pesticides, No. 30.
Geneva, World Health Organization (Unpublished documents VBC,
DS,77.30).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
ANNEX I
TREATMENT OF ORGANOPHOSPHATE INSECTICIDE POISONING IN MAN
(From EHC 63: Organophosphorus Insecticides - A General Introduction)
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be included
in the IPCS Health and Safety Guides to be prepared for selected
organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the Absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with alkaline
soap or with a sodium bicarbonate solution. Particular care should be
taken in cleaning the skin area where venupuncture is performed.
Blood might be contaminated with direct-acting organophosphorus esters
and, therefore, inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting might be induced, if
the patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml water. This treatment is, however,
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with addition of bicarbonate solution or
activated charcoal) can also be performed, particularly in unconscious
patients, taking care to prevent aspiration of fluids into the lungs
(i.e., only after a tracheal tube has been put in place).
The volume of fluid introduced into the stomach should be recorded and
samples of gastric lavage frozen and stored for subsequent chemical
analysis. If the formulation of the pesticide involved is available,
it should also be stored for further analysis (i.e., detection of
toxicologically relevant impurities). A purgative can be administered
to remove the ingested compound.
I.2 General Supportive Treatment
Artificial respiration (via a tracheal tube) should be started at the
first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general,
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific Pharmacological Treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine treatment
varies from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases and
total daily doses of up to several hundred mg may be necessary during
the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision on whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have beneficial
effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms, which are not affected by atropine.
Doses of 10 mg s.c. or i.v. are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and drugs
that may depress respiration are not recommended in the absence of
artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one time
may cause toxic side-effects. Pulse-rate should not exceed 120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be taken
into, and then released from, fat depots over a period of many days.
It is therefore quite incorrect to abandon oxime treatment after
1-2 days on the supposition that all inhibited enzyme will be aged.
Ecobichon et al. (1977) noted prompt improvement in both condition and
blood-ChEs in response to pralidoxime given on the 11th-15th days
after major symptoms of poisoning appeared due to extended exposure to
fenitrothion (a dimethyl phosphate with a short half-life for aging of
inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in error,
while drunk. Therapeutic dosages were progressively increased up to
6 mg atropine i.v. every 15 min together with continuous i.v. infusion
of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to 6 after
intoxication. After considerable improvement, the patient relapsed
and further aggressive therapy was given at a declining rate from days
10 to 16 (atropine) and to day 23 (oxime), respectively. In total,
92 g of pralidoxime chloride and 3912 mg of atropine were given and
the patient was discharged on the thirty-third day with no apparent
sequelae.
References to Annex I
ECOBICHON, D.J., OZERE, R.L., REID, E., & CROCKER, J.F.S (1977)
Acute fenitrothion poisoning. Can. Med. Assoc. J., 116: 377-379.
KAGAN, JU.S. (1977) [Toxicology of organophosphorus pesticides.]
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
WHO document. VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed. New
York, Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of medical
practitioners, London, United Kingdom Department of Health and Social
Security, pp. 41-47.
VALE, J.A. & SCOTT, G.W. (1974) Organophosphorus poisoning. Guy's
Hosp. Rep., 123: 13-25.
WARRINER, R.A., III, NIES, A.S., & HAYES, W.J., Jr (1977) Severe
organophosphate poisoning complicated by alcohol and terpentine
ingestion. Arch. environ. Health, 32: 203-205.
 Relative molecular mass: 277.25
CAS chemical name: O,O-dimethyl- O-(3-methyl-4-nitrophenyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
thiophosphate
CAS Registry number:
Relative molecular mass: 277.25
CAS chemical name: O,O-dimethyl- O-(3-methyl-4-nitrophenyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
phosphorothioate
O,O-dimethyl- O-(4-nitro- m-tolyl)-
thiophosphate
CAS Registry number:  The European Economic Community legislation requires labelling of
fenitrothion as dangerous substance using the symbol:
The European Economic Community legislation requires labelling of
fenitrothion as dangerous substance using the symbol:
 The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink and
animal feeding stuffs.
The European Economic Community legislation on the labelling of
pesticide preparations classifies fenitrothion in Class IIB for the
purpose of determining the label.
WHO gives the following product specification for technical
fenitrothion for use in public health:
"The material shall consist of fenitrothion together with related
manufacturing compounds and shall be in the form of a yellow to brown
liquid free from extraneous impurities or added modifying agents."
It should contain at least 930 g of fenitrothion per kg. The
specification gives the required acidity and water content and also
analytical methods for checking The specification continues:
"The technical fenitrothion shall be packed in suitable clean
containers, as specified in the order."
"All packages shall bear, durably and legibly marked on the
container, the following:
Manufacturer's name
Technical fenitrothion to specification WHO/SIT/17.R2
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
"Fenitrothion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It may be
absorbed through the skin. Avoid skin contact; wear protective
gloves, clean protective clothing, and a respirator when handling the
material. Wash thoroughly with soap and water after using."
"Keep out of the reach of children and well away from foodstuffs and
animal feed and their containers."
"If poisoning occurs, call a physician. Atropine and pralidoxime are
specific antidotes and artificial respiration may be needed."
Similar specifications and instructions are given for fenitrothion
water-dispersible powder and emulsified concentrate.
FAO gives similar product specifications for fenitrothion for its use
in plant protection. In this case, the technical material should
contain at least 95% active material, and containers must comply with
pertinent national and international transport and safety regulations.
6.5 Waste Disposal
In the USA, any waste containing fenitrothion is considered a
hazardous waste and permits are required for its handling, transport,
treatment, storage or discharge and disposal.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Japan Maximum acceptable concentration (MAC)
- Time-weighted average (TWA) 1 mg/m3a 1986
USSR Maximum allowable concentration (MAC)
- Ceiling value (CLV) (vapour and aerosol) 0.1 mg/m3 1982
Ambient USSR Maximum allowable concentration (MAC)
- average per day 0.001 mg/m3 1982
- 1 × /day 0.005 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.005 mg/kg 1988
body weight
Residue FAO/WHO Maximum residue limit (MRL) 0.002-20 mg/kg 1989
(with its oxygen analogue expressed
as fenitrothion); products specified as follows:
- milks 0.002 mg/kg
- cucumbers, meat, onions, potatoes 0.05 mg/kg
- cauliflower, cocoa beans, egg plants,
peppers, soybeans (dry) 0.1 mg/kg
- bread (white) leeks, radishes 0.2 mg/kg
- apples, cabbage, cabbage red, cherries,
grapes, lettuce, pear, peas,
strawberries, tea (dried, green),
tomatoes 0.5 mg/kg
- peach, rice (polished) 1 mg/kg
- citrus fruit, wheat flour (white),b
processed wheat bran 2 mg/kg
- wheat flour (wholemeal) 5 mg/kg
- cereal grains 10 mg/kg
- raw wheat bran, rice bran unprocessed 20 mg/kg
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Surface USSR Maximum allowable concentration (MAC) 0.25 mg/litre 1983
Fishing USSR Maximum allowable concentration (MAC) 0.00 mg/litre 1983
SOIL USSR Maximum allowable concentration (MAC) 1.0 mg/kg 1984
a Notice : skin absorption.
b Codex 1990.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986a) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in food.
Rome, Food and Agriculture Organization of the United Nations.
FAO/WHO (1986b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues. 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1986c) Codex maximum limits for pesticide residues.
CAC/Vol.XIII - ed. 2, Rome, Codex Alimentarius Commission, Food and
Agriculture Organization of the United Nations, (+ Annex 2 (1990)).
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides,
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) Environmental Health Criteria 63: Organophosphorus
insecticides - a general introduction. Geneva, World Health
Organization, 181 pp.
WHO (1990) The WHO recommended classification of pesticides by hazard
and guidelines to classification, 1990-91. Geneva, World Health
Organization (Unpublished document WHO/PCS/90.1).
WHO (1990) Environmental Health Criteria 133: Fenitrothion. Geneva,
World Health Organization.
WHO/FAO (1977) Fenitrothion. Data sheets on pesticides, No. 30.
Geneva, World Health Organization (Unpublished documents VBC,
DS,77.30).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
ANNEX I
TREATMENT OF ORGANOPHOSPHATE INSECTICIDE POISONING IN MAN
(From EHC 63: Organophosphorus Insecticides - A General Introduction)
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be included
in the IPCS Health and Safety Guides to be prepared for selected
organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the Absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with alkaline
soap or with a sodium bicarbonate solution. Particular care should be
taken in cleaning the skin area where venupuncture is performed.
Blood might be contaminated with direct-acting organophosphorus esters
and, therefore, inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting might be induced, if
the patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml water. This treatment is, however,
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with addition of bicarbonate solution or
activated charcoal) can also be performed, particularly in unconscious
patients, taking care to prevent aspiration of fluids into the lungs
(i.e., only after a tracheal tube has been put in place).
The volume of fluid introduced into the stomach should be recorded and
samples of gastric lavage frozen and stored for subsequent chemical
analysis. If the formulation of the pesticide involved is available,
it should also be stored for further analysis (i.e., detection of
toxicologically relevant impurities). A purgative can be administered
to remove the ingested compound.
I.2 General Supportive Treatment
Artificial respiration (via a tracheal tube) should be started at the
first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general,
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific Pharmacological Treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine treatment
varies from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases and
total daily doses of up to several hundred mg may be necessary during
the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision on whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have beneficial
effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms, which are not affected by atropine.
Doses of 10 mg s.c. or i.v. are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and drugs
that may depress respiration are not recommended in the absence of
artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one time
may cause toxic side-effects. Pulse-rate should not exceed 120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be taken
into, and then released from, fat depots over a period of many days.
It is therefore quite incorrect to abandon oxime treatment after
1-2 days on the supposition that all inhibited enzyme will be aged.
Ecobichon et al. (1977) noted prompt improvement in both condition and
blood-ChEs in response to pralidoxime given on the 11th-15th days
after major symptoms of poisoning appeared due to extended exposure to
fenitrothion (a dimethyl phosphate with a short half-life for aging of
inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in error,
while drunk. Therapeutic dosages were progressively increased up to
6 mg atropine i.v. every 15 min together with continuous i.v. infusion
of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to 6 after
intoxication. After considerable improvement, the patient relapsed
and further aggressive therapy was given at a declining rate from days
10 to 16 (atropine) and to day 23 (oxime), respectively. In total,
92 g of pralidoxime chloride and 3912 mg of atropine were given and
the patient was discharged on the thirty-third day with no apparent
sequelae.
References to Annex I
ECOBICHON, D.J., OZERE, R.L., REID, E., & CROCKER, J.F.S (1977)
Acute fenitrothion poisoning. Can. Med. Assoc. J., 116: 377-379.
KAGAN, JU.S. (1977) [Toxicology of organophosphorus pesticides.]
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
WHO document. VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed. New
York, Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of medical
practitioners, London, United Kingdom Department of Health and Social
Security, pp. 41-47.
VALE, J.A. & SCOTT, G.W. (1974) Organophosphorus poisoning. Guy's
Hosp. Rep., 123: 13-25.
WARRINER, R.A., III, NIES, A.S., & HAYES, W.J., Jr (1977) Severe
organophosphate poisoning complicated by alcohol and terpentine
ingestion. Arch. environ. Health, 32: 203-205.
The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink and
animal feeding stuffs.
The European Economic Community legislation on the labelling of
pesticide preparations classifies fenitrothion in Class IIB for the
purpose of determining the label.
WHO gives the following product specification for technical
fenitrothion for use in public health:
"The material shall consist of fenitrothion together with related
manufacturing compounds and shall be in the form of a yellow to brown
liquid free from extraneous impurities or added modifying agents."
It should contain at least 930 g of fenitrothion per kg. The
specification gives the required acidity and water content and also
analytical methods for checking The specification continues:
"The technical fenitrothion shall be packed in suitable clean
containers, as specified in the order."
"All packages shall bear, durably and legibly marked on the
container, the following:
Manufacturer's name
Technical fenitrothion to specification WHO/SIT/17.R2
Batch or reference number, and date of test
Net weight of contents
Date of manufacture
and the following minimum cautionary notice:
"Fenitrothion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It may be
absorbed through the skin. Avoid skin contact; wear protective
gloves, clean protective clothing, and a respirator when handling the
material. Wash thoroughly with soap and water after using."
"Keep out of the reach of children and well away from foodstuffs and
animal feed and their containers."
"If poisoning occurs, call a physician. Atropine and pralidoxime are
specific antidotes and artificial respiration may be needed."
Similar specifications and instructions are given for fenitrothion
water-dispersible powder and emulsified concentrate.
FAO gives similar product specifications for fenitrothion for its use
in plant protection. In this case, the technical material should
contain at least 95% active material, and containers must comply with
pertinent national and international transport and safety regulations.
6.5 Waste Disposal
In the USA, any waste containing fenitrothion is considered a
hazardous waste and permits are required for its handling, transport,
treatment, storage or discharge and disposal.
Table 3. Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Japan Maximum acceptable concentration (MAC)
- Time-weighted average (TWA) 1 mg/m3a 1986
USSR Maximum allowable concentration (MAC)
- Ceiling value (CLV) (vapour and aerosol) 0.1 mg/m3 1982
Ambient USSR Maximum allowable concentration (MAC)
- average per day 0.001 mg/m3 1982
- 1 × /day 0.005 mg/m3
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.005 mg/kg 1988
body weight
Residue FAO/WHO Maximum residue limit (MRL) 0.002-20 mg/kg 1989
(with its oxygen analogue expressed
as fenitrothion); products specified as follows:
- milks 0.002 mg/kg
- cucumbers, meat, onions, potatoes 0.05 mg/kg
- cauliflower, cocoa beans, egg plants,
peppers, soybeans (dry) 0.1 mg/kg
- bread (white) leeks, radishes 0.2 mg/kg
- apples, cabbage, cabbage red, cherries,
grapes, lettuce, pear, peas,
strawberries, tea (dried, green),
tomatoes 0.5 mg/kg
- peach, rice (polished) 1 mg/kg
- citrus fruit, wheat flour (white),b
processed wheat bran 2 mg/kg
- wheat flour (wholemeal) 5 mg/kg
- cereal grains 10 mg/kg
- raw wheat bran, rice bran unprocessed 20 mg/kg
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Surface USSR Maximum allowable concentration (MAC) 0.25 mg/litre 1983
Fishing USSR Maximum allowable concentration (MAC) 0.00 mg/litre 1983
SOIL USSR Maximum allowable concentration (MAC) 1.0 mg/kg 1984
a Notice : skin absorption.
b Codex 1990.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986a) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluation of some pesticide residues in food.
Rome, Food and Agriculture Organization of the United Nations.
FAO/WHO (1986b) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of pesticide
residues. 3rd ed., Rome, Codex Committee on Pesticide Residues.
FAO/WHO (1986c) Codex maximum limits for pesticide residues.
CAC/Vol.XIII - ed. 2, Rome, Codex Alimentarius Commission, Food and
Agriculture Organization of the United Nations, (+ Annex 2 (1990)).
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides,
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
document VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols, Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS (NIOSH)
01-123).
WHO (1986) Environmental Health Criteria 63: Organophosphorus
insecticides - a general introduction. Geneva, World Health
Organization, 181 pp.
WHO (1990) The WHO recommended classification of pesticides by hazard
and guidelines to classification, 1990-91. Geneva, World Health
Organization (Unpublished document WHO/PCS/90.1).
WHO (1990) Environmental Health Criteria 133: Fenitrothion. Geneva,
World Health Organization.
WHO/FAO (1977) Fenitrothion. Data sheets on pesticides, No. 30.
Geneva, World Health Organization (Unpublished documents VBC,
DS,77.30).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
ANNEX I
TREATMENT OF ORGANOPHOSPHATE INSECTICIDE POISONING IN MAN
(From EHC 63: Organophosphorus Insecticides - A General Introduction)
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be included
in the IPCS Health and Safety Guides to be prepared for selected
organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the Absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with alkaline
soap or with a sodium bicarbonate solution. Particular care should be
taken in cleaning the skin area where venupuncture is performed.
Blood might be contaminated with direct-acting organophosphorus esters
and, therefore, inaccurate measures of ChE inhibition might result.
Extensive eye irrigation with water or saline should also be
performed. In the case of ingestion, vomiting might be induced, if
the patient is conscious, by the administration of ipecacuanha syrup
(10-30 ml) followed by 200 ml water. This treatment is, however,
contraindicated in the case of pesticides dissolved in hydrocarbon
solvents. Gastric lavage (with addition of bicarbonate solution or
activated charcoal) can also be performed, particularly in unconscious
patients, taking care to prevent aspiration of fluids into the lungs
(i.e., only after a tracheal tube has been put in place).
The volume of fluid introduced into the stomach should be recorded and
samples of gastric lavage frozen and stored for subsequent chemical
analysis. If the formulation of the pesticide involved is available,
it should also be stored for further analysis (i.e., detection of
toxicologically relevant impurities). A purgative can be administered
to remove the ingested compound.
I.2 General Supportive Treatment
Artificial respiration (via a tracheal tube) should be started at the
first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general,
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific Pharmacological Treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine treatment
varies from case to case, but should maintain the patient fully
atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases and
total daily doses of up to several hundred mg may be necessary during
the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision on whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have beneficial
effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms, which are not affected by atropine.
Doses of 10 mg s.c. or i.v. are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and drugs
that may depress respiration are not recommended in the absence of
artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one time
may cause toxic side-effects. Pulse-rate should not exceed 120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be taken
into, and then released from, fat depots over a period of many days.
It is therefore quite incorrect to abandon oxime treatment after
1-2 days on the supposition that all inhibited enzyme will be aged.
Ecobichon et al. (1977) noted prompt improvement in both condition and
blood-ChEs in response to pralidoxime given on the 11th-15th days
after major symptoms of poisoning appeared due to extended exposure to
fenitrothion (a dimethyl phosphate with a short half-life for aging of
inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in error,
while drunk. Therapeutic dosages were progressively increased up to
6 mg atropine i.v. every 15 min together with continuous i.v. infusion
of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to 6 after
intoxication. After considerable improvement, the patient relapsed
and further aggressive therapy was given at a declining rate from days
10 to 16 (atropine) and to day 23 (oxime), respectively. In total,
92 g of pralidoxime chloride and 3912 mg of atropine were given and
the patient was discharged on the thirty-third day with no apparent
sequelae.
References to Annex I
ECOBICHON, D.J., OZERE, R.L., REID, E., & CROCKER, J.F.S (1977)
Acute fenitrothion poisoning. Can. Med. Assoc. J., 116: 377-379.
KAGAN, JU.S. (1977) [Toxicology of organophosphorus pesticides.]
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization (Unpublished
WHO document. VBC/84.889).
TAYLOR, P. (1980) Anticholinesterase agents. In: Goodman, L.S. &
Gilman, A., ed. The pharmacological basis of therapeutics. 6th ed. New
York, Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of medical
practitioners, London, United Kingdom Department of Health and Social
Security, pp. 41-47.
VALE, J.A. & SCOTT, G.W. (1974) Organophosphorus poisoning. Guy's
Hosp. Rep., 123: 13-25.
WARRINER, R.A., III, NIES, A.S., & HAYES, W.J., Jr (1977) Severe
organophosphate poisoning complicated by alcohol and terpentine
ingestion. Arch. environ. Health, 32: 203-205.
