
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 63
HEXACHLOROCYCLO-PENTADIENE
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1991
This is a companion volume to Environmental Health Criteria 120:
Hexachlorocyclopentadiene
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Hexachlorocyclopentadiene : health and safety guide.
(Health and safety guide ; no. 63)
1.Hydrocarbons, Chlorinated - adverse effects
2.Hydrocarbons, Chlorinated - standards
3.Hydrocarbons, Chlorinated - toxicity
4.Environmental exposure
5.Environmental pollutants I.Series
ISBN 92 4 151063 3 (NLM Classification: QV 633)
ISSN 0259-7268
(c) World Health Organization 1991
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Composition
1.4. Uses
2. SUMMARY AND EVALUATION
2.1. Exposure
2.2. Uptake, metabolism, and excretion
2.3. Effects on animals
2.4. Effects on human beings
2.5. Effects on organisms in the environment
3. CONCLUSIONS AND RECOMMENDATIONS
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 Medical advice
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.2.1. Explosion hazards
4.2.2. Fire hazards
4.2.3. Prevention
4.2.4. Extinguishing agents
4.3. Storage
4.4. Transport
4.5. Spillage
4.6. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Other measures
7.6. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: hexachlorocyclopentadiene (HEX)
Molecular formula: C5Cl6
Chemical structure:
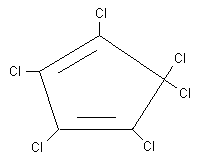 Common synonyms: perchlorocyclopentadiene (HCCPD)
Common trade names: C-56, HRS 1655, Graphlox
CAS chemical name: 1,2,3,4,5,5'-hexachloro-1,3-
cyclopentadiene
CAS registry number: 77-47-4
EINEC number: 2010293
Relative molecular mass: 272.77
Conversion factor (20°C): 1 ppm = 11.3 mg/m3
1 mg/m3 = 0.088 ppm
1.2 Physical and Chemical Properties
Pure hexachlorocyclopentadiene (HEX) is a light, lemon-yellow liquid
that has a pungent, musty odour. The odour threshold is reported to
be 0.0014-0.0016 mg/m3. HEX is highly reactive and volatile at low
temperatures. Other physical and chemical properties of HEX are given
in the Summary of Chemical Safety Information (section 6).
1.3 Composition
Commercially available HEX contains several impurities, depending on
the method of synthesis. HEX with a purity exceeding 90% is produced
by thermal dechlorination of octachlorocyclopentene. HEX with a
purity of 75% contains many lower chlorinated cyclopentadienes as well
as hexachlorobenzene and octachlorocyclopentene.
1.4 Uses
HEX is an intermediate in the manufacture of chlorinated cyclodiene
pesticides, such as heptachlor, chlordane, aldrin, dieldrin, endrin,
and mirex, and in the manufacture of resins, dyes, and pharmaceutical
products. Another major use is in the manufacture of flame
retardants, including Dechlorane Plus (R) and chlorendic anhydride.
2. SUMMARY AND EVALUATION
2.1 Exposure
Few data are available on the relative contribution of different
sources of HEX to the environment. Human exposure via air, soil, or
water is expected to be minimal, except for that of persons living in
the vicinity of manufacturing, shipping, or disposal sites.
Occupational exposure is probably the major source of human exposure.
Short-term laboratory tests have shown that HEX is highly toxic for
aquatic microorganisms, invertebrates, and fish, and less toxic for
soil micro-organisms. Information obtained under environmentally
realistic conditions is limited, but the potential hazard for the
general environment is expected to be low.
2.2 Uptake, Metabolism, and Excretion
Absorption from the gastrointestinal tract is relatively low compared
with that from the lungs or skin. HEX appears to be almost completely
metabolized in the body. The primary metabolites are described as
polar compounds, but they have not been identified. Most of the
retained HEX and its metabolites are found in the liver and kidneys,
though after inhalation exposure, the highest tissue concentrations
are found in the lung and trachea. The major routes of elimination
are the urine and faeces. Excretion in the bile has been shown to
occur with all routes of exposure.
2.3 Effects on Animals
HEX is moderately toxic for animals via the oral and dermal routes of
exposure, and extremely toxic via inhalation. The main cause of death
following acute exposure via any route is respiratory failure,
suggesting that the respiratory tract is the major target organ of
toxicity. HEX is also a primary skin irritant and causes severe eye
irritation in rabbits.
Short-term oral exposure (91 days) to HEX resulted in kidney damage.
No data have been found on effects associated with long-term oral
exposure to HEX.
The inhalation of HEX vapour at concentrations of 4.52 mg/m3
(0.4 ppm) or more, for 90 days, resulted in respiratory-tract effects,
including inflammation and respiratory distress, in mice and rats.
The inhalation of 5.65 mg/m3 (0.5 ppm) for 6 h/day, 5 days/week,
over 30 weeks, resulted in lung damage in mice and rats. No data on
the effects of long-term inhalation exposure to HEX were available.
HEX has not been shown to affect reproduction and development in
animals. There is good evidence that HEX is not mutagenic in
bacterial or mammalian cells, or in the fruit fly. Data on the
carcinogenic potential of HEX in experimental animals are not
currently available.
2.4 Effects on Human Beings
There is limited information on human health effects. Acute exposure
can result in severe irritation of the eyes, nose, throat, and
gastrointestinal tract. In a well-documented incident at a
waste-water treatment plant, exposed workers exhibited eye irritation,
headaches, nausea, and eye and throat irritation. These effects
persisted for several weeks. Exposure levels could not be estimated
accurately, but airborne concentrations in primary treatment areas,
measured 4 days after closing the facility, ranged from 3.05 to
10.96 mg/m3 (270 to 970 ppb).
No excess cancer mortality was reported in an epidemiological study of
workers employed at a chemical plant producing and processing HEX.
Using the criteria of the International Agency for Research on Cancer
(IARC), the available data on carcinogenicity are classified in
Group 3, i.e., because of major qualitative or quantitative
limitations, the available studies cannot be interpreted as showing
either the presence or absence of a carcinogenic effect.
2.5 Effects on Organisms in the Environment
HEX is highly toxic for aquatic organisms. There is a lack of
information on the effects of HEX on animals and vegetation.
3. CONCLUSIONS AND RECOMMENDATIONS
(i) Environmental monitoring is needed to examine the persistence
and fate of HEX in all media near production, processing, and disposal
sites, and also hazardous waste incinerators. Monitoring data are
needed for HEX in drinking-water, and surface, shower, and ground
waters.
(ii) Biomarker techniques should be developed to indicate the
possible past or current actions of HEX. Such biomarkers could be
stable metabolites derived from HEX, or the contaminants present in
the original preparation.
(iii) Research is needed on the metabolic, degradation, and reactive
products, to understand the fate of HEX in humans and the environment.
(iv) Further study of the apparent disparity between degradation
under laboratory conditions and that observed in the environment is
needed.
(v) The efficacy and safety of current disposal methods should be
evaluated, and their present and future health impacts assessed.
(vi) Developmental and reproductive studies on HEX need to be
conducted, with emphasis on the inhalation route of exposure.
(vii) Methods for the early warning of the presence of HEX should be
developed.
(viii) Occupational exposure to HEX should be minimized by the use of
closed systems. Guidelines should be followed on the disposal of HEX
and HEX wastes.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
HEX is toxic via all routes of exposure, but is particularly dangerous
via inhalation. This chemical is extremely irritating to the mucous
membranes, eyes, and skin, causing lacrimation, sneezing, and
salivation. Contact with the skin can result in discoloration,
blisters, and burns. Inhalation or ingestion can result in nausea,
vomiting, diarrhoea, headaches, lethargy, breathing difficulties, and
liver and kidney damage. Inhalation exposure may cause severe
irritation to the throat, respiratory tract, and the lungs. The human
health hazards associated with certain types of exposure, as well as
preventive and protective measures and first-aid recommendations, are
presented in the Summary of Chemical Safety Information (section 6).
4.1.1 Advice to physicians
Following the ingestion of HEX, oxygen should be administered, if
respiration is depressed. Toxic symptoms resulting from inhalation
exposure should be treated symptomatically and supportively. Oxygen
should be administered as needed. In the case of eye contact, the eyes
should be washed immediately with large amounts of water for at least
15 minutes, to remove any remaining chemical. The eyes should then be
irrigated with physiological saline solution for 30-60 minutes, or
until the pH returns to normal. Following dermal contact, the
affected area should be washed immediately with soap and large amounts
of water. If blistering develops, the skin should be covered with
loosely wrapped sterile bandages.
4.1.2 Health surveillance advice
Persons employed in the manufacture, transportation, or disposal of
HEX should undergo periodic medical examinations.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
HEX is not explosive. However, containers may explode in the heat of
fires. Furthermore, HEX may form explosive mixtures with sodium.
4.2.2 Fire hazards
HEX is not flammable. However, toxic fumes, such as hydrogen
chloride, chlorine, and phosgene, are formed in fires.
4.2.3 Prevention
Enclosed processing equipment, local exhaust, or ventilation that is
adequate to meet published exposure limits, should be provided.
4.2.4 Extinguishing agents
For fires, dry chemical powder, carbon dioxide, water spray, or
standard foam is recommended. If possible, containers should be moved
away from the fire. Water used to control fires should be contained,
or diked, for later disposal.
4.3 Storage
HEX should be stored in tightly closed containers, labelled as
corrosive, and stored in a cool, dry place, away from moisture, heat,
and strong oxidizing agents.
4.4 Transport
HEX can be transported at ambient temperatures and does not require an
inert atmosphere; it is stable under normal conditions. For shipping,
55-gallon (250-litre) drums or tank cars are recommended. Label as
corrosive.
4.5 Spillage
In the case of a spill or leak, earth, sand, or some other
noncombustible absorbent material (cement powder or soda ash) can be
used to absorb the chemical. This material is then placed in a clean,
dry, covered container for later disposal. Dry spills can be removed
using a clean shovel. For large spills, the liquid should be
contained, or diked, at a distance from the spill for later disposal.
In the case of an air release, vapours should be knocked down with a
water spray. Persons should remain upwind of the vapour cloud. Water
spills can be trapped at the bottom of deep water pockets (since HEX
is heavier than water), in excavated holding areas, or within sandbag
barriers, to avoid contamination of sewers and surface waters.
Damaged containers should be destroyed to prevent further use.
Suction hoses can then be used to remove the spilled material.
4.6 Disposal
HEX can be incinerated after mixing with a combustible fuel. This
mixture should be completely combusted to prevent the formation of
phosgene. An acid scrubber should be used to remove the halo acids
produced. HEX can be buried in specially designated chemical
landfills. However, HEX and organic solvents should not be disposed
of in the same landfill area, to prevent possible migration of HEX
from the disposal site. Dumping into sewer systems is not an
acceptable method of disposal. Deep well injection can be used.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Since environmental pollution is expected to occur as a result of
releases during the production, processing, and disposal of HEX,
contamination can be avoided by the use of proper methods of storage,
transport, handling, and waste disposal (sections 4.3, 4.4, and 4.6).
In the case of spillage, the clean-up methods described in section 4.5
should be used.
When the fate and transport of HEX in the atmosphere was modelled on
the basis of available laboratory data, a tropospheric residence time
of approximately 5 h was calculated. The atmospheric transport of HEX
has been reported from an area where waste was stored, and from wet
wells during the treatment of industrial wastes.
In water, HEX may undergo photolysis, hydrolysis, and biodegradation.
In shallow water, it has a photolytic half-life of <1 h. In deeper
water, where photolysis was precluded, the hydrolytic half-life of HEX
ranged from several days to approximately 3 months; biodegradation is
predicted to occur more slowly. HEX is known to volatilize from
surface water, and its volatilization is affected by turbulence and by
sorption on to sediments.
Because of its low solubility in water, HEX should be relatively
immobile in soil; however, HEX has been found in ground water.
Volatilization, which is likely to occur primarily at the soil
surface, is inversely related to the levels of organic matter in the
soil. From laboratory studies, chemical hydrolysis and microbial
metabolism, both aerobic and anaerobic, would be expected to reduce
HEX levels in soils.
Low concentrations of HEX have been shown to be toxic for aquatic
life. Lethality in acute exposures (48-96 h) was observed in both
freshwater and saltwater crustaceans and fish at nominal
concentrations of 32-180 µg/litre in static exposure systems where the
water was not renewed during the test. As the photolytic half-life is
<1 h, the concentration of the HEX would have decreased substantially
during the 48-96 h of exposure.
In aqueous media, HEX is toxic for many other microorganisms at
nominal concentrations of 0.2-10 mg/litre, or levels substantially
higher than those needed to kill most aquatic animals or plants. HEX
appears to be less toxic for microorganisms in the soil than in
aquatic media, probably because of the sorption of HEX on to the soil
matrix.
Although exposure is expected to be low, data currently available are
insufficient to determine the effects of HEX exposure on terrestrial
vegetation or wildlife.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hexachlorocyclopentadiene. It should be
displayed at, or near, entrances to areas where there is potential
exposure to hexachlorocyclopentadiene, and on processing equipment
and containers. The summary should be translated into the
appropriate language(s). All persons potentially exposed to the
chemical should also have the instructions in the summary clearly
explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
HEXACHLOROCYCLOPENTADIENE (C5Cl6)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 272.77 Reacts slowly with water to form
Appearance lemon-yellow liquid hydrochloric acid; may form explosive
Odour threshold 0.00019 mg/m3 mixture with sodium; corrosive to iron
Odour pungent, musty and other metals, in the presence of
Melting point (°C) -9.6 moisture
Boiling point (°C) 239
Solubility in water (25°C) 1.03-1.25 mg/litre
Relative vapour density (air =1) 9.42
Density (25°C) 1.702
Vapour pressure (25°C) 0.08 mmHg
Electronegativity ND
Flame colouration test ND
Octanol/water partition
coefficient (Kow) 1.1 x 105
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Severe irritation; burns, Avoid exposure; wear protective Remove contaminated clothing and shoes;
blisters; readily absorbed through clothing and rubber gloves and flush skin with plenty of water; cover burns
intact skin shoes with sterile bandages, loosely wrapped
EYES: Severe irritation; Wear a face-shield if there is a Flush the eyes with plenty of water for
redness; pain possibility of eye contact 15 minutes, making sure to remove any
remaining chemical; seek medical attention
INHALATION: Severe irritation Avoid exposure by using Fresh air, rest; if breathing has
to mucous membranes and upper ventilation, local exhaust, or stopped, apply artificial respiration;
respiratory tract; sore throat, respiratory protection, such as a administer oxygen, if required; seek
coughing, shortness of breath, self-contained breathing medical attention immediately
headache, vomiting, lacrimation, apparatus and a face-shield
pulmonary oedema, degenerative
changes in lung, liver, and kidney;
death may occur from respiratory
failure
INGESTION: Salivation, Do not eat, drink, or smoke when Rinse mouth; seek medical
diarrhoea, lethargy, shortness of handling the compound attention
breath or difficulty in breathing,
degenerative changes in the lung,
liver, kidney, adrenal glands,
heart, and brain
HEX is highly toxic to aquatic Do not contaminate surface
environments waters or sewers
SPILLAGE STORAGE FIRE AND EXPLOSION
Do not touch spilled material; Store in a cool, dry place in Negligible fire hazard; hydrogen
for dry spills, using clean shovel tightly closed containers chloride, chlorine, and phosgene
place material into a clean, dry may be formed in the heat of
container; for small liquid spills, fires; closed containers may
take up with sand or other explode in the heat of fires
absorbent material and place into
containers for disposal; for large
liquid spills, dike far ahead of
spill for later disposal
WASTE DISPOSAL NATIONAL INFORMATION
Incinerate after mixing with National Occupational Exposure Code of Federal Regulations:
combustible fuel; take care to Limit: Poison, Class B
ensure complete combustion;
apply acid scrubbers; can also be National Poison Control Centre:
disposed of in specially designated
chemical landfill; care should be Local trade names:
taken to avoid disposal together
with organic solvents
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
7.1 Previous Evaluations by International Bodies
Data are insufficient to assess the carcinogenic potential of HEX in
humans. No previous evaluations have been carried out by
international bodies.
7.2 Exposure Limit Values
See table on pages 24-25.
7.3 Specific Restrictions
No specific restrictions were found.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transport of Dangerous
Goods classifies HEX as a poisonous substance (Hazard Class 6.1), and
as a very dangerous substance (Packing Group I) with regard to
packing.
Country Type Media Level/remarka Date
Australia recommendation air/occupational TLV/TWA = 0.1 mg/m3 (0.01 ppm) 1983
STEL = 0.3 mg/m3 (0.03 ppm)
Belgium recommendation air/occupational TLV/TWA = 0.1 mg/m3 (0.01 ppm) 1988
Canada regulation air/occupational TLV/TWA = 0.01 ppm (0.1 mg/m3) 1980
Canada regulation transport specific transportation regulations 1987
Finland recommendation air/occupational TWA = 1.0 mg/m3 (0.1 ppm) 1989
skin STEL = 3.0 mg/m3 (0.3 ppm) 1989
Germany, regulation waste "toxic waste" subject to specific handling, 1981
Federal transport, treatment, storage, and disposal
Republic of regulation/permits
Netherlands recommendation air/occupational TWA/OCC = 0.11 mg/m3 (0.01 ppm) 1986
Switzerland regulation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1987
USA regulation water 1 µg/litre ambient 1980
water quality criteria (organoleptic)
USA (ACGIH) recommendation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1980
USA regulation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1989
USA regulation water/land notification of spill of 1 lb (0.454 kg) 1983
in 24-h period
USA regulation waste/transport "toxic waste" subject to specific handling, 1980
transport, treatment, storage, and disposal
regulation/permits
Country Type Media Level/remarka Date
USA draft
recommendation drinking-water 7 µg/kg per day (lifetime) 1990
USSR regulation air/occupational TLV = 0.01 mg/m3 (0.01 ppm vapour) 1989
USSR regulation water MAC = 0.001 mg/ml (organoleptic) 1985
USSR regulation air/ambient STEL = 0.001 mg/m3 1987
Yugoslavia regulation air/occupational TWA = 0.1 mg/m3 (skin) 1985
a TLV = threshold limit value; TWA = time-weighted average; STEL = short-term exposure level; OCC = occupational;
MAC = maximum allowable concentration.
The US Code of Federal Regulations lists HEX as a Class B poison and
recommends that the label presented below be placed on packaging.
Label as a corrosive.
Common synonyms: perchlorocyclopentadiene (HCCPD)
Common trade names: C-56, HRS 1655, Graphlox
CAS chemical name: 1,2,3,4,5,5'-hexachloro-1,3-
cyclopentadiene
CAS registry number: 77-47-4
EINEC number: 2010293
Relative molecular mass: 272.77
Conversion factor (20°C): 1 ppm = 11.3 mg/m3
1 mg/m3 = 0.088 ppm
1.2 Physical and Chemical Properties
Pure hexachlorocyclopentadiene (HEX) is a light, lemon-yellow liquid
that has a pungent, musty odour. The odour threshold is reported to
be 0.0014-0.0016 mg/m3. HEX is highly reactive and volatile at low
temperatures. Other physical and chemical properties of HEX are given
in the Summary of Chemical Safety Information (section 6).
1.3 Composition
Commercially available HEX contains several impurities, depending on
the method of synthesis. HEX with a purity exceeding 90% is produced
by thermal dechlorination of octachlorocyclopentene. HEX with a
purity of 75% contains many lower chlorinated cyclopentadienes as well
as hexachlorobenzene and octachlorocyclopentene.
1.4 Uses
HEX is an intermediate in the manufacture of chlorinated cyclodiene
pesticides, such as heptachlor, chlordane, aldrin, dieldrin, endrin,
and mirex, and in the manufacture of resins, dyes, and pharmaceutical
products. Another major use is in the manufacture of flame
retardants, including Dechlorane Plus (R) and chlorendic anhydride.
2. SUMMARY AND EVALUATION
2.1 Exposure
Few data are available on the relative contribution of different
sources of HEX to the environment. Human exposure via air, soil, or
water is expected to be minimal, except for that of persons living in
the vicinity of manufacturing, shipping, or disposal sites.
Occupational exposure is probably the major source of human exposure.
Short-term laboratory tests have shown that HEX is highly toxic for
aquatic microorganisms, invertebrates, and fish, and less toxic for
soil micro-organisms. Information obtained under environmentally
realistic conditions is limited, but the potential hazard for the
general environment is expected to be low.
2.2 Uptake, Metabolism, and Excretion
Absorption from the gastrointestinal tract is relatively low compared
with that from the lungs or skin. HEX appears to be almost completely
metabolized in the body. The primary metabolites are described as
polar compounds, but they have not been identified. Most of the
retained HEX and its metabolites are found in the liver and kidneys,
though after inhalation exposure, the highest tissue concentrations
are found in the lung and trachea. The major routes of elimination
are the urine and faeces. Excretion in the bile has been shown to
occur with all routes of exposure.
2.3 Effects on Animals
HEX is moderately toxic for animals via the oral and dermal routes of
exposure, and extremely toxic via inhalation. The main cause of death
following acute exposure via any route is respiratory failure,
suggesting that the respiratory tract is the major target organ of
toxicity. HEX is also a primary skin irritant and causes severe eye
irritation in rabbits.
Short-term oral exposure (91 days) to HEX resulted in kidney damage.
No data have been found on effects associated with long-term oral
exposure to HEX.
The inhalation of HEX vapour at concentrations of 4.52 mg/m3
(0.4 ppm) or more, for 90 days, resulted in respiratory-tract effects,
including inflammation and respiratory distress, in mice and rats.
The inhalation of 5.65 mg/m3 (0.5 ppm) for 6 h/day, 5 days/week,
over 30 weeks, resulted in lung damage in mice and rats. No data on
the effects of long-term inhalation exposure to HEX were available.
HEX has not been shown to affect reproduction and development in
animals. There is good evidence that HEX is not mutagenic in
bacterial or mammalian cells, or in the fruit fly. Data on the
carcinogenic potential of HEX in experimental animals are not
currently available.
2.4 Effects on Human Beings
There is limited information on human health effects. Acute exposure
can result in severe irritation of the eyes, nose, throat, and
gastrointestinal tract. In a well-documented incident at a
waste-water treatment plant, exposed workers exhibited eye irritation,
headaches, nausea, and eye and throat irritation. These effects
persisted for several weeks. Exposure levels could not be estimated
accurately, but airborne concentrations in primary treatment areas,
measured 4 days after closing the facility, ranged from 3.05 to
10.96 mg/m3 (270 to 970 ppb).
No excess cancer mortality was reported in an epidemiological study of
workers employed at a chemical plant producing and processing HEX.
Using the criteria of the International Agency for Research on Cancer
(IARC), the available data on carcinogenicity are classified in
Group 3, i.e., because of major qualitative or quantitative
limitations, the available studies cannot be interpreted as showing
either the presence or absence of a carcinogenic effect.
2.5 Effects on Organisms in the Environment
HEX is highly toxic for aquatic organisms. There is a lack of
information on the effects of HEX on animals and vegetation.
3. CONCLUSIONS AND RECOMMENDATIONS
(i) Environmental monitoring is needed to examine the persistence
and fate of HEX in all media near production, processing, and disposal
sites, and also hazardous waste incinerators. Monitoring data are
needed for HEX in drinking-water, and surface, shower, and ground
waters.
(ii) Biomarker techniques should be developed to indicate the
possible past or current actions of HEX. Such biomarkers could be
stable metabolites derived from HEX, or the contaminants present in
the original preparation.
(iii) Research is needed on the metabolic, degradation, and reactive
products, to understand the fate of HEX in humans and the environment.
(iv) Further study of the apparent disparity between degradation
under laboratory conditions and that observed in the environment is
needed.
(v) The efficacy and safety of current disposal methods should be
evaluated, and their present and future health impacts assessed.
(vi) Developmental and reproductive studies on HEX need to be
conducted, with emphasis on the inhalation route of exposure.
(vii) Methods for the early warning of the presence of HEX should be
developed.
(viii) Occupational exposure to HEX should be minimized by the use of
closed systems. Guidelines should be followed on the disposal of HEX
and HEX wastes.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
HEX is toxic via all routes of exposure, but is particularly dangerous
via inhalation. This chemical is extremely irritating to the mucous
membranes, eyes, and skin, causing lacrimation, sneezing, and
salivation. Contact with the skin can result in discoloration,
blisters, and burns. Inhalation or ingestion can result in nausea,
vomiting, diarrhoea, headaches, lethargy, breathing difficulties, and
liver and kidney damage. Inhalation exposure may cause severe
irritation to the throat, respiratory tract, and the lungs. The human
health hazards associated with certain types of exposure, as well as
preventive and protective measures and first-aid recommendations, are
presented in the Summary of Chemical Safety Information (section 6).
4.1.1 Advice to physicians
Following the ingestion of HEX, oxygen should be administered, if
respiration is depressed. Toxic symptoms resulting from inhalation
exposure should be treated symptomatically and supportively. Oxygen
should be administered as needed. In the case of eye contact, the eyes
should be washed immediately with large amounts of water for at least
15 minutes, to remove any remaining chemical. The eyes should then be
irrigated with physiological saline solution for 30-60 minutes, or
until the pH returns to normal. Following dermal contact, the
affected area should be washed immediately with soap and large amounts
of water. If blistering develops, the skin should be covered with
loosely wrapped sterile bandages.
4.1.2 Health surveillance advice
Persons employed in the manufacture, transportation, or disposal of
HEX should undergo periodic medical examinations.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
HEX is not explosive. However, containers may explode in the heat of
fires. Furthermore, HEX may form explosive mixtures with sodium.
4.2.2 Fire hazards
HEX is not flammable. However, toxic fumes, such as hydrogen
chloride, chlorine, and phosgene, are formed in fires.
4.2.3 Prevention
Enclosed processing equipment, local exhaust, or ventilation that is
adequate to meet published exposure limits, should be provided.
4.2.4 Extinguishing agents
For fires, dry chemical powder, carbon dioxide, water spray, or
standard foam is recommended. If possible, containers should be moved
away from the fire. Water used to control fires should be contained,
or diked, for later disposal.
4.3 Storage
HEX should be stored in tightly closed containers, labelled as
corrosive, and stored in a cool, dry place, away from moisture, heat,
and strong oxidizing agents.
4.4 Transport
HEX can be transported at ambient temperatures and does not require an
inert atmosphere; it is stable under normal conditions. For shipping,
55-gallon (250-litre) drums or tank cars are recommended. Label as
corrosive.
4.5 Spillage
In the case of a spill or leak, earth, sand, or some other
noncombustible absorbent material (cement powder or soda ash) can be
used to absorb the chemical. This material is then placed in a clean,
dry, covered container for later disposal. Dry spills can be removed
using a clean shovel. For large spills, the liquid should be
contained, or diked, at a distance from the spill for later disposal.
In the case of an air release, vapours should be knocked down with a
water spray. Persons should remain upwind of the vapour cloud. Water
spills can be trapped at the bottom of deep water pockets (since HEX
is heavier than water), in excavated holding areas, or within sandbag
barriers, to avoid contamination of sewers and surface waters.
Damaged containers should be destroyed to prevent further use.
Suction hoses can then be used to remove the spilled material.
4.6 Disposal
HEX can be incinerated after mixing with a combustible fuel. This
mixture should be completely combusted to prevent the formation of
phosgene. An acid scrubber should be used to remove the halo acids
produced. HEX can be buried in specially designated chemical
landfills. However, HEX and organic solvents should not be disposed
of in the same landfill area, to prevent possible migration of HEX
from the disposal site. Dumping into sewer systems is not an
acceptable method of disposal. Deep well injection can be used.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Since environmental pollution is expected to occur as a result of
releases during the production, processing, and disposal of HEX,
contamination can be avoided by the use of proper methods of storage,
transport, handling, and waste disposal (sections 4.3, 4.4, and 4.6).
In the case of spillage, the clean-up methods described in section 4.5
should be used.
When the fate and transport of HEX in the atmosphere was modelled on
the basis of available laboratory data, a tropospheric residence time
of approximately 5 h was calculated. The atmospheric transport of HEX
has been reported from an area where waste was stored, and from wet
wells during the treatment of industrial wastes.
In water, HEX may undergo photolysis, hydrolysis, and biodegradation.
In shallow water, it has a photolytic half-life of <1 h. In deeper
water, where photolysis was precluded, the hydrolytic half-life of HEX
ranged from several days to approximately 3 months; biodegradation is
predicted to occur more slowly. HEX is known to volatilize from
surface water, and its volatilization is affected by turbulence and by
sorption on to sediments.
Because of its low solubility in water, HEX should be relatively
immobile in soil; however, HEX has been found in ground water.
Volatilization, which is likely to occur primarily at the soil
surface, is inversely related to the levels of organic matter in the
soil. From laboratory studies, chemical hydrolysis and microbial
metabolism, both aerobic and anaerobic, would be expected to reduce
HEX levels in soils.
Low concentrations of HEX have been shown to be toxic for aquatic
life. Lethality in acute exposures (48-96 h) was observed in both
freshwater and saltwater crustaceans and fish at nominal
concentrations of 32-180 µg/litre in static exposure systems where the
water was not renewed during the test. As the photolytic half-life is
<1 h, the concentration of the HEX would have decreased substantially
during the 48-96 h of exposure.
In aqueous media, HEX is toxic for many other microorganisms at
nominal concentrations of 0.2-10 mg/litre, or levels substantially
higher than those needed to kill most aquatic animals or plants. HEX
appears to be less toxic for microorganisms in the soil than in
aquatic media, probably because of the sorption of HEX on to the soil
matrix.
Although exposure is expected to be low, data currently available are
insufficient to determine the effects of HEX exposure on terrestrial
vegetation or wildlife.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hexachlorocyclopentadiene. It should be
displayed at, or near, entrances to areas where there is potential
exposure to hexachlorocyclopentadiene, and on processing equipment
and containers. The summary should be translated into the
appropriate language(s). All persons potentially exposed to the
chemical should also have the instructions in the summary clearly
explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
HEXACHLOROCYCLOPENTADIENE (C5Cl6)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 272.77 Reacts slowly with water to form
Appearance lemon-yellow liquid hydrochloric acid; may form explosive
Odour threshold 0.00019 mg/m3 mixture with sodium; corrosive to iron
Odour pungent, musty and other metals, in the presence of
Melting point (°C) -9.6 moisture
Boiling point (°C) 239
Solubility in water (25°C) 1.03-1.25 mg/litre
Relative vapour density (air =1) 9.42
Density (25°C) 1.702
Vapour pressure (25°C) 0.08 mmHg
Electronegativity ND
Flame colouration test ND
Octanol/water partition
coefficient (Kow) 1.1 x 105
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Severe irritation; burns, Avoid exposure; wear protective Remove contaminated clothing and shoes;
blisters; readily absorbed through clothing and rubber gloves and flush skin with plenty of water; cover burns
intact skin shoes with sterile bandages, loosely wrapped
EYES: Severe irritation; Wear a face-shield if there is a Flush the eyes with plenty of water for
redness; pain possibility of eye contact 15 minutes, making sure to remove any
remaining chemical; seek medical attention
INHALATION: Severe irritation Avoid exposure by using Fresh air, rest; if breathing has
to mucous membranes and upper ventilation, local exhaust, or stopped, apply artificial respiration;
respiratory tract; sore throat, respiratory protection, such as a administer oxygen, if required; seek
coughing, shortness of breath, self-contained breathing medical attention immediately
headache, vomiting, lacrimation, apparatus and a face-shield
pulmonary oedema, degenerative
changes in lung, liver, and kidney;
death may occur from respiratory
failure
INGESTION: Salivation, Do not eat, drink, or smoke when Rinse mouth; seek medical
diarrhoea, lethargy, shortness of handling the compound attention
breath or difficulty in breathing,
degenerative changes in the lung,
liver, kidney, adrenal glands,
heart, and brain
HEX is highly toxic to aquatic Do not contaminate surface
environments waters or sewers
SPILLAGE STORAGE FIRE AND EXPLOSION
Do not touch spilled material; Store in a cool, dry place in Negligible fire hazard; hydrogen
for dry spills, using clean shovel tightly closed containers chloride, chlorine, and phosgene
place material into a clean, dry may be formed in the heat of
container; for small liquid spills, fires; closed containers may
take up with sand or other explode in the heat of fires
absorbent material and place into
containers for disposal; for large
liquid spills, dike far ahead of
spill for later disposal
WASTE DISPOSAL NATIONAL INFORMATION
Incinerate after mixing with National Occupational Exposure Code of Federal Regulations:
combustible fuel; take care to Limit: Poison, Class B
ensure complete combustion;
apply acid scrubbers; can also be National Poison Control Centre:
disposed of in specially designated
chemical landfill; care should be Local trade names:
taken to avoid disposal together
with organic solvents
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
7.1 Previous Evaluations by International Bodies
Data are insufficient to assess the carcinogenic potential of HEX in
humans. No previous evaluations have been carried out by
international bodies.
7.2 Exposure Limit Values
See table on pages 24-25.
7.3 Specific Restrictions
No specific restrictions were found.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transport of Dangerous
Goods classifies HEX as a poisonous substance (Hazard Class 6.1), and
as a very dangerous substance (Packing Group I) with regard to
packing.
Country Type Media Level/remarka Date
Australia recommendation air/occupational TLV/TWA = 0.1 mg/m3 (0.01 ppm) 1983
STEL = 0.3 mg/m3 (0.03 ppm)
Belgium recommendation air/occupational TLV/TWA = 0.1 mg/m3 (0.01 ppm) 1988
Canada regulation air/occupational TLV/TWA = 0.01 ppm (0.1 mg/m3) 1980
Canada regulation transport specific transportation regulations 1987
Finland recommendation air/occupational TWA = 1.0 mg/m3 (0.1 ppm) 1989
skin STEL = 3.0 mg/m3 (0.3 ppm) 1989
Germany, regulation waste "toxic waste" subject to specific handling, 1981
Federal transport, treatment, storage, and disposal
Republic of regulation/permits
Netherlands recommendation air/occupational TWA/OCC = 0.11 mg/m3 (0.01 ppm) 1986
Switzerland regulation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1987
USA regulation water 1 µg/litre ambient 1980
water quality criteria (organoleptic)
USA (ACGIH) recommendation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1980
USA regulation air/occupational TWA = 0.1 mg/m3 (0.01 ppm) 1989
USA regulation water/land notification of spill of 1 lb (0.454 kg) 1983
in 24-h period
USA regulation waste/transport "toxic waste" subject to specific handling, 1980
transport, treatment, storage, and disposal
regulation/permits
Country Type Media Level/remarka Date
USA draft
recommendation drinking-water 7 µg/kg per day (lifetime) 1990
USSR regulation air/occupational TLV = 0.01 mg/m3 (0.01 ppm vapour) 1989
USSR regulation water MAC = 0.001 mg/ml (organoleptic) 1985
USSR regulation air/ambient STEL = 0.001 mg/m3 1987
Yugoslavia regulation air/occupational TWA = 0.1 mg/m3 (skin) 1985
a TLV = threshold limit value; TWA = time-weighted average; STEL = short-term exposure level; OCC = occupational;
MAC = maximum allowable concentration.
The US Code of Federal Regulations lists HEX as a Class B poison and
recommends that the label presented below be placed on packaging.
Label as a corrosive.
 7.5 Other Measures
None found.
7.6 Waste Disposal
The US EPA classifies HEX as a toxic pollutant and is required to set
effluent limitations and pretreatment standards for 21 major
industries. Permits are required for the discharge of any pollutant
from any point source into US waters. Applicants who have reason to
believe that HEX is contained in any of the outfalls produced, or
applicants using specific industrial processes listed in the Code of
Federal Regulations (40 CFR 122.53; 1981), must report quantitative
data for HEX. Inspecting, monitoring, and reporting requirements are
specified after the permit is issued. Discharge of the substance
must be reported, regardless of permit requirements, if levels exceed
10 µg/litre, five times the maximum concentration reported in the
application, or the level established by the US EPA administrator.
The US EPA further classifies certain specified industrial solid
wastes containing HEX as hazardous substances.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed. Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, pp. 300.
BRANSFORD, J., ed. (1989) Material safety data sheet (MSDS). CD-ROM.
New York, Occupational Health Services, Inc..
BRETHERICK, L. (1985) Handbook of reactive chemical hazards. 3rd ed.
London, England, Butterworths, pp. 491, 1317.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, England, London Royal Society of Chemistry, p. 351.
IRPTC (1985) Treatment and disposal methods for waste chemicals.
Geneva, Switzerland, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1989) Data profile: Hexachlorocyclopentadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
New Jersey, Sigma-Aldrich Chemical Corporation, p. 1014.
National Library of Medicine (1989) Hazardous substances data bank
(HSDB). Online. Washington, DC, National Institutes of Health,
MEDLARS Management Section.
NFPA (1986) 43D. Code for storage of pesticides in portable
containers. Boston, Massachusetts, National Fire Protection
Association, pp. A-2 - A-11.
NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Cincinnati, Ohio, National Institute for Occupational
Safety and Health, US Department of Health and Human Services, Public
Health Service, Center for Disease Control.
SAX, N.I., ed. (1986) Hazardous chemicals information annual. New
York, Van Nostrand Reinhold Company Inc., pp. 119-121.
US EPA (1988) Risk screening guide. Attachment: SARA Section 313
ROADMAPS information hexachlorocyclopentadiene. Washington, DC, Office
of Toxic Substances.
US EPA (1989) Integrated risk information system (IRIS). Online.
Cincinnati, Ohio, Office of Health and Environmental Assessment,
Environmental Criteria and Assessment Office.
WEISS, G., ed. (1980) Hazardous chemicals data book. Park Ridge,
New Jersey, Noyes Data Corporation, p. 498.
WHO (1991) Environmental Health Criteria 120: Hexachloro
cyclopentadiene. Geneva, World Health Organization.
7.5 Other Measures
None found.
7.6 Waste Disposal
The US EPA classifies HEX as a toxic pollutant and is required to set
effluent limitations and pretreatment standards for 21 major
industries. Permits are required for the discharge of any pollutant
from any point source into US waters. Applicants who have reason to
believe that HEX is contained in any of the outfalls produced, or
applicants using specific industrial processes listed in the Code of
Federal Regulations (40 CFR 122.53; 1981), must report quantitative
data for HEX. Inspecting, monitoring, and reporting requirements are
specified after the permit is issued. Discharge of the substance
must be reported, regardless of permit requirements, if levels exceed
10 µg/litre, five times the maximum concentration reported in the
application, or the level established by the US EPA administrator.
The US EPA further classifies certain specified industrial solid
wastes containing HEX as hazardous substances.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed. Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, pp. 300.
BRANSFORD, J., ed. (1989) Material safety data sheet (MSDS). CD-ROM.
New York, Occupational Health Services, Inc..
BRETHERICK, L. (1985) Handbook of reactive chemical hazards. 3rd ed.
London, England, Butterworths, pp. 491, 1317.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, England, London Royal Society of Chemistry, p. 351.
IRPTC (1985) Treatment and disposal methods for waste chemicals.
Geneva, Switzerland, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1989) Data profile: Hexachlorocyclopentadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
New Jersey, Sigma-Aldrich Chemical Corporation, p. 1014.
National Library of Medicine (1989) Hazardous substances data bank
(HSDB). Online. Washington, DC, National Institutes of Health,
MEDLARS Management Section.
NFPA (1986) 43D. Code for storage of pesticides in portable
containers. Boston, Massachusetts, National Fire Protection
Association, pp. A-2 - A-11.
NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Cincinnati, Ohio, National Institute for Occupational
Safety and Health, US Department of Health and Human Services, Public
Health Service, Center for Disease Control.
SAX, N.I., ed. (1986) Hazardous chemicals information annual. New
York, Van Nostrand Reinhold Company Inc., pp. 119-121.
US EPA (1988) Risk screening guide. Attachment: SARA Section 313
ROADMAPS information hexachlorocyclopentadiene. Washington, DC, Office
of Toxic Substances.
US EPA (1989) Integrated risk information system (IRIS). Online.
Cincinnati, Ohio, Office of Health and Environmental Assessment,
Environmental Criteria and Assessment Office.
WEISS, G., ed. (1980) Hazardous chemicals data book. Park Ridge,
New Jersey, Noyes Data Corporation, p. 498.
WHO (1991) Environmental Health Criteria 120: Hexachloro
cyclopentadiene. Geneva, World Health Organization.
 Common synonyms: perchlorocyclopentadiene (HCCPD)
Common trade names: C-56, HRS 1655, Graphlox
CAS chemical name: 1,2,3,4,5,5'-hexachloro-1,3-
cyclopentadiene
CAS registry number:
Common synonyms: perchlorocyclopentadiene (HCCPD)
Common trade names: C-56, HRS 1655, Graphlox
CAS chemical name: 1,2,3,4,5,5'-hexachloro-1,3-
cyclopentadiene
CAS registry number:  7.5 Other Measures
None found.
7.6 Waste Disposal
The US EPA classifies HEX as a toxic pollutant and is required to set
effluent limitations and pretreatment standards for 21 major
industries. Permits are required for the discharge of any pollutant
from any point source into US waters. Applicants who have reason to
believe that HEX is contained in any of the outfalls produced, or
applicants using specific industrial processes listed in the Code of
Federal Regulations (40 CFR 122.53; 1981), must report quantitative
data for HEX. Inspecting, monitoring, and reporting requirements are
specified after the permit is issued. Discharge of the substance
must be reported, regardless of permit requirements, if levels exceed
10 µg/litre, five times the maximum concentration reported in the
application, or the level established by the US EPA administrator.
The US EPA further classifies certain specified industrial solid
wastes containing HEX as hazardous substances.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed. Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, pp. 300.
BRANSFORD, J., ed. (1989) Material safety data sheet (MSDS). CD-ROM.
New York, Occupational Health Services, Inc..
BRETHERICK, L. (1985) Handbook of reactive chemical hazards. 3rd ed.
London, England, Butterworths, pp. 491, 1317.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, England, London Royal Society of Chemistry, p. 351.
IRPTC (1985) Treatment and disposal methods for waste chemicals.
Geneva, Switzerland, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1989) Data profile: Hexachlorocyclopentadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
New Jersey, Sigma-Aldrich Chemical Corporation, p. 1014.
National Library of Medicine (1989) Hazardous substances data bank
(HSDB). Online. Washington, DC, National Institutes of Health,
MEDLARS Management Section.
NFPA (1986) 43D. Code for storage of pesticides in portable
containers. Boston, Massachusetts, National Fire Protection
Association, pp. A-2 - A-11.
NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Cincinnati, Ohio, National Institute for Occupational
Safety and Health, US Department of Health and Human Services, Public
Health Service, Center for Disease Control.
SAX, N.I., ed. (1986) Hazardous chemicals information annual. New
York, Van Nostrand Reinhold Company Inc., pp. 119-121.
US EPA (1988) Risk screening guide. Attachment: SARA Section 313
ROADMAPS information hexachlorocyclopentadiene. Washington, DC, Office
of Toxic Substances.
US EPA (1989) Integrated risk information system (IRIS). Online.
Cincinnati, Ohio, Office of Health and Environmental Assessment,
Environmental Criteria and Assessment Office.
WEISS, G., ed. (1980) Hazardous chemicals data book. Park Ridge,
New Jersey, Noyes Data Corporation, p. 498.
WHO (1991) Environmental Health Criteria 120: Hexachloro
cyclopentadiene. Geneva, World Health Organization.
7.5 Other Measures
None found.
7.6 Waste Disposal
The US EPA classifies HEX as a toxic pollutant and is required to set
effluent limitations and pretreatment standards for 21 major
industries. Permits are required for the discharge of any pollutant
from any point source into US waters. Applicants who have reason to
believe that HEX is contained in any of the outfalls produced, or
applicants using specific industrial processes listed in the Code of
Federal Regulations (40 CFR 122.53; 1981), must report quantitative
data for HEX. Inspecting, monitoring, and reporting requirements are
specified after the permit is issued. Discharge of the substance
must be reported, regardless of permit requirements, if levels exceed
10 µg/litre, five times the maximum concentration reported in the
application, or the level established by the US EPA administrator.
The US EPA further classifies certain specified industrial solid
wastes containing HEX as hazardous substances.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed. Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, pp. 300.
BRANSFORD, J., ed. (1989) Material safety data sheet (MSDS). CD-ROM.
New York, Occupational Health Services, Inc..
BRETHERICK, L. (1985) Handbook of reactive chemical hazards. 3rd ed.
London, England, Butterworths, pp. 491, 1317.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, England, London Royal Society of Chemistry, p. 351.
IRPTC (1985) Treatment and disposal methods for waste chemicals.
Geneva, Switzerland, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1989) Data profile: Hexachlorocyclopentadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
New Jersey, Sigma-Aldrich Chemical Corporation, p. 1014.
National Library of Medicine (1989) Hazardous substances data bank
(HSDB). Online. Washington, DC, National Institutes of Health,
MEDLARS Management Section.
NFPA (1986) 43D. Code for storage of pesticides in portable
containers. Boston, Massachusetts, National Fire Protection
Association, pp. A-2 - A-11.
NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Cincinnati, Ohio, National Institute for Occupational
Safety and Health, US Department of Health and Human Services, Public
Health Service, Center for Disease Control.
SAX, N.I., ed. (1986) Hazardous chemicals information annual. New
York, Van Nostrand Reinhold Company Inc., pp. 119-121.
US EPA (1988) Risk screening guide. Attachment: SARA Section 313
ROADMAPS information hexachlorocyclopentadiene. Washington, DC, Office
of Toxic Substances.
US EPA (1989) Integrated risk information system (IRIS). Online.
Cincinnati, Ohio, Office of Health and Environmental Assessment,
Environmental Criteria and Assessment Office.
WEISS, G., ed. (1980) Hazardous chemicals data book. Park Ridge,
New Jersey, Noyes Data Corporation, p. 498.
WHO (1991) Environmental Health Criteria 120: Hexachloro
cyclopentadiene. Geneva, World Health Organization.
