
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 9
ISOBUTANOL
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1987
This is a companion volume to Environmental Health Criteria 65:
Butanols - Four Isomers: 1-Butanol, 2-Butanol, tert-Butanol,
Isobutanol
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
ISBN 92 4 154665 4
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1987
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
IPCS
HEALTH AND SAFETY GUIDE FOR ISOBUTANOL
INTRODUCTION
HOW TO USE THE GUIDE
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Uses
2. SUMMARY AND EVALUATION
2.1. Exposure to isobutanol
2.2. Uptake, metabolism, and excretion
2.3. Effects on organisms in the environment
2.4. Effects on animals
2.5. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
4. HEALTH HAZARDS FOR MAN, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Main hazards for man, prevention and protection, first aid
4.2. Advice to physicians
4.3. Health surveillance advice
4.4. Explosion and fire hazards
4.4.1. Explosion hazards
4.4.2. Fire hazards
4.4.3. Fire-extinguishing agents
4.5. Storage
4.6. Transport
4.7. Spillage and disposal
4.7.1. Spillage
4.7.1.1 Small spillage
4.7.1.2 Large spillage
4.7.2. Disposal
5. INTERNATIONAL CHEMICAL SAFETY CARD
6. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Exposure limit values
7.2. Specific restrictions
7.3. Labelling, packaging, and transport
INTRODUCTION
The International Programme on Chemical Safety is responsible for the
publication of a series of Environmental Health Criteria documents,
each of which assesses the existing information on the relationship
between exposure to a specific chemical, mixture of chemicals, or
combination of chemicals and physical and biological agents, and man's
health and the integrity of the environment. The documents provide
guidelines for setting exposure limits consistent with the protection
of human health and the environment.
To facilitate the application of these guidelines in national chemical
safety programmes, "Health and Safety Guides" are being prepared,
highlighting the information contained in the documents for those who
need to know the health and environmental issues involved, but not the
scientific details. The Guides include advice on preventive and
protective measures and emergency action.
Review and revision of the information in this Health and Safety Guide
will take place in due course, and the eventual aim is to use
standardized terminology. We should be grateful if you would help by
telling us of any difficulties encountered in using the information in
this guide.
Comments please, addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
HOW TO USE THE GUIDE
All people in the work-place environment should be given the relevant
written information in this book, supplemented by a clear, personal
explanation to ensure that they are fully aware of the dangers and the
current courses of protective and emergency action.
The International Chemical Safety Card should be displayed as directed
and its contents clearly explained to all working personnel. Medical
staff should be fully conversant with the medical information to
ensure they can act rapidly and efficiently in an emergency. Posters
should be used to give impact to basic safety measures.
* * *
Further copies of the Health and Safety Guide and, for those requiring
more detailed scientific information, the relevant Environmental
Health Criteria publication, are available to order.
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3
\
CH - CH2OH
/
CH3
Primary constituent: isobutanol
Common synonyms: isobutyl alcohol, isopropylcarbinol,
2-methyl-l-propanol, 2-methylpropyl
alcohol
CAS registry number: 78-83-1
1.2 Physical and Chemical Properties
Some physical and chemical properties of isobutanol are given in the
Sample International Chemical Safety Card.
1.3 Uses
The major use of isobutanol is in the manufacture of isobutyl acetate
for use in the lacquer industry. It is also used as a flavouring agent
in butter, cola, fruit, liquor, rum, and whisky. Further applications
include use as a solvent in paint and varnish removers and in the
manufacture of isobutyl esters, which serve as solvents, plasticizers,
flavourings, and perfumes. Average maximum levels at which it is used
in the USA range between 7 and 30 mg/kg.
Natural isobutanol is produced by the fermentation of carbohydrates.
It is found in brandy, cider, gin, coffee, cherries, raspberries,
blackberries, grapes, apples, hop oil, bread, and Cheddar cheese. It
can also result from decay in municipal waste plants.
2. SUMMARY AND EVALUATION
2.1 Exposure to Isobutanol
Levels of exposure of the general population to isobutanol through
food and beverages are not available, and occupational exposure levels
are inadequate.
2.2 Uptake, Metabolism, and Excretion
In animals, isobutanol is absorbed through the skin, lungs, and
gastrointestinal tract. It is metabolized by alcohol dehydrogenase to
isobutyric acid via the aldehyde and may enter the tricarboxylic acid
cycle. Small amounts of isobutanol are excreted unchanged (<0.5% of
the dose) or as the glucuronide (< 5% of the dose) in the urine. In
rabbits, metabolites found in the urine included acetaldehyde, acetic
acid, isobutylaldehyde, and isovaleric acid.
2.3 Effects on Organisms in the Environment
Few quantitative data relating to levels in the general environment
are available, but, because isobutanol is readily biodegradable,
substantial concentrations are only likely to occur locally in the
case of major spillages. It does not bioaccumulate.
At background concentrations likely to occur in the environment,
isobutanol is not directly toxic for fish, amphibia, crustacea, or
algae. Protozoa will be tolerant to levels of isobutanol likely to be
found in the environment.
Isobutanol should be managed in the environment as a slightly toxic
compound. It poses an indirect hazard for the aquatic environment,
because it is readily biodegradable, which may lead to oxygen
depletion.
2.4 Effects on Animals
Oral LD50 values (2.5 - 3.1 g/kg body weight) and inhalation LC50
values (19.2 g/m3) for the rat classify isobutanol as slightly toxic
according to the scale of Hodge & Sterner. Acute toxic effects are
alcoholic intoxication and narcosis. Isobutanol is severely irritating
for the eyes and moderately irritating for the skin. A group of rats
given a 1 mol/litre solution of isobutanol as their sole drinking
liquid for 4 months did not show any adverse effects on the liver;
another group given a 2 mol/litre solution as their sole drinking
liquid for 2 months showed a reduction in fat, glycogen, RNA content,
and overall size of the cells in the liver. Continuous inhalation
exposure of rats to a concentration of 3 mg/m3 for 4 months resulted
in depression of leg withdrawal response to electrical stimulation,
and minor changes in formed elements of the blood and in serum
enzymes. The estimated no-observed-adverse-effect level was
0.1 mg/m3.
In a life-time carcinogenicity study, groups of rats received
isobutanol subcutaneously (0.05 ml/kg body weight, twice a week) or
orally (0.2 mg/kg body weight, twice a week). The animals exhibited
toxic liver damage ranging from steatosis to cirrhosis. The total
numbers of animals showing malignant rumours were 3 in the oral group,
8 in the subcutaneous group, and 0 in the control group. The majority
of treated animals also showed hyperplasia of blood-forming tissues.
The Task Group could not determine whether isobutanol was a
genetically active compound because of a lack of mutagenicity studies.
The findings in the carcinogenicity study are a cause for concern.
Because of methodological inadequacies and the manner of reporting the
data, it could not be determined whether isobutanol should be regarded
as an animal carcinogen. Therefore, it is not possible to extrapolate
from this study to possible long-term effects in man.
From the animal studies available, it is not possible to determine a
no-observed-adverse-effect level for long-term exposure. Adequate data
are not available to assess the mutagenicity or teratogenicity of
isobutanol or its effects on reproduction.
2.5 Effects on Human Beings
Exposure of the general population to isobutanol through food and
beverages is unlikely to lead to acute toxic effects. The only
reported observations in man relate to the production of vertigo under
conditions of severe and prolonged exposure to vapour mixtures of
isobutanol and 1-butanol. It was not possible from this study to
attribute the vertigo to a single cause.
3. CONCLUSIONS AND RECOMMENDATIONS
1. On the basis of available data, the Task Group considered it
unlikely that isobutanol would pose a serious acute health risk
for the general population, under normal exposure conditions.
However, the Task Group was unable to make an assessment of the
long-term health risks of isobutanol for the general population.
It was concluded that the results of the carcinogenicity study
need verification by a bioassay of modem standards.
2. The Task Group considered that the data available were inadequate
to establish guidelines for setting occupational exposure limits.
In line with good manufacturing practice, exposure to isobutanol
should be minimized.
3. The ecotoxicological data available indicate that the impact of
background concentrations of isobutanol on the aquatic
environment can be expected to be minimal.
4. The Task Group noted that animal data were inadequate to
determine a no-observed-adverse-effect level. Relevant studies
should be conducted so that this can be achieved.
5. The Task Group considered that adequate studies should be
conducted to assess the mutagenicity and carcinogenicity of
isobutanol.
6. Epidemiological studies, including precise exposure data, would
enable a better assessment of the occupational hazards of
isobutanol to be made.
7. Additional information on environmental pathways (notably
emission and leaching) and residues are desirable.
From: Environmental Health Criteria 65: Butanols - Four Isomers:
1-Butanol, 2-Butanol, tert-Butanol, Isobutanol
4. HEALTH HAZARDS FOR MAN, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Main Hazards for Man, Prevention and Protection, First Aid
Liquid isobutanol is moderately irritating to the skin and severely
irritating to the eyes. It is irritating to the mucous membranes of
the respiratory tract. At high concentrations, its vapour can cause
narcosis. It has been shown to be hepatoxic in both human beings and
animals and is a questionable carcinogen.
The human health hazards associated with certain types of exposure to
isobutanol, together with preventive and protective measures and first
aid recommendations are listed in the following table.
GOLDEN RULES
1. Do not smoke, drink, or eat in the work-place.
2. In case of overexposure, the victim should leave, or be removed
from, the contaminated area to fresh air as rapidly as possible.
3. Remove contaminated clothing and shoes and wash with plenty of
water and soap.
4. Flush affected eye(s) with water for at least 15 minutes.
4.2 Advice to Physicians
Treat by observation and supportive measures as indicated by the
patient's condition.
4.3 Health Surveillance Advice
In view of its hepatotoxicity and its irritation potential, regular
medical surveillance may be advisable at the discretion of a medical
adviser after taking into account the likely extent of exposure.
4.4 Explosion and Fire Hazards
4.4.1 Explosion hazards
Explosive vapour-air mixtures are formed at temperatures exceeding
27°C.
4.4.2 Fire hazards
Isobutanol is flammable.
ROUTE HEALTH HAZARDS PREVENTION AND PROTECTION FIRST AID
SINGLE EXPOSURE
SKIN Moderately irritating Minimize exposure, Remove contaminated clothing;
wear protective clothing rinse skin with water; if skin
irritation persists, seek medical
attention
EYES Potentially moderately Minimize exposure, Flush eyes immediately with
irritating wear goggles or face shield water for at least 15 minutes;
seek medical attention
INHALATION Irritant; high vapour Minimize exposure, Remove patient to fresh air;
concentrations may cause use adequate ventilation if breathing has stopped, apply
narcosis or suitable respiratory artificial respiration; seek medical
protection attention immediately
INGESTION Unlikely occupational hazard; Apply normal hygienic Do not induce vomiting; seek
it may be absorbed and cause practices medical attention immediately
systemic effects such as alcohol
poisoning and hepatoxicity
(cont'd)
REPEATED EXPOSURE
INHALATION As for single exposure Minimize exposure,
apply normal hygienic practices
INGESTION Long-term consumption Apply normal hygienic practices;
(present in most alcoholic moderation
beverages) may result in liver
damage
4.4.3 Fire-extinguishing agents
Dry chemical powder extinguishers are recommended. These are
particularly useful when fires involve "live" electrical equipment,
because the powder is non-conducting.
For a small fire, use carbon dioxide, dry chemical powder, alcohol-
resistant foam, sand, earth, or water spray. Do not use water in a
jet. For a large fire, use alcohol-resistant foam, or water fog.
4.5 Storage
Store drums away from sources of heat and out of direct sunlight.
4.6 Transport
Treat as for flammable liquids; otherwise, no special measures
indicated.
4.7 Spillage and Disposal
4.7.1 Spillage
Extinguish naked flames. Do not smoke. Avoid sparks. Avoid contact
with skin, eyes, and clothing. Wear gloves, goggles or face shield,
and boots. Avoid breathing the vapour.
4.7.1.1 Small spillage
Absorb the spilt liquid with sand, earth, sawdust, or other suitable
absorbant material. Shovel up and remove all material to a safe area
for subsequent disposal by burning or burying. Flush the contaminated
area with plenty of water.
4.7.1.2 Large spillage
Prevent spilt liquid from spreading by the use of sand or earth. If
possible, transfer the liquid to a salvage tank. Otherwise, treat as
for a small spillage. Inform the local authorities (particularly the
fire service) at once, if the spilt liquid enters the surface drains,
since a potential explosive hazard will be created.
4.7.2 Disposal
Incineration is the recommended method. Wastes should be sprayed into
the furnace. Incineration is more efficient if the wastes are mixed
with a more flammable liquid.
5. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers concerned
with, and users of, isobutanol. It should be displayed at, or near,
entrances to areas where there is potential exposure to isobutanol,
and on processing equipment and containers.
The card should be translated into the appropriate language(s). All
persons potentially exposed to the chemical should also have the
instructions on the chemical safety card clearly explained.
ISOBUTANOL
(2-methyl-1-propanol, isopropylcarbinol, isobutyl alcohol) [(CH3)2CHCH2OH]
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Boiling point (°C) 108 Colourless liquid with characteristic odour; reacts
Melting point (°C) -108 with strong oxidants and alkali metals to form
Flash point (°C) 27 combustible gas (hydrogen); attacks many plastics
Autoignition temperature (°C) 430 substance may be absorbed into the body by inhalation
Relative density (water = 1) 0.8
Relative vapour density 2.6
(air = 1)
Vapour pressure in mbar (20°C) 12
Solubility in water 95
(g/litre at 20°C)
Explosive limits (vol. % in air) 1.2-10.9
Relative molecular mass 74.1
HAZARDS/SYMPTOMS PREVENTION FIRST AID
INHALATION: Sore throat, coughing, Minimize exposure, Fresh air; rest in half upright position;
shortness of breath, dullness, headache use ventilation, local exhaust seek medical attention, if necessary
or breathing protection
SKIN: May be absorbed; redness Minimize exposure, Remove contaminated clothing; rinse skin
wear protective gloves with plenty of water or shower
EYES: Redness, pain, blurred vision Minimize exposure, First rinse with plenty of water; then seek
wear safety goggles medical attention
INGESTION: Abdominal pain, vomiting, Rinse mouth; give plenty of water to drink;
dullness seek medical attention or transport to hospital
GENERAL: Alcoholic beverages may
enhance the toxic effects
(cont'd)
SPILLAGE STORAGE FIRE AND EXPLOSION
Collect leaking liquid in sealable Fireproof Flammable; above 27°C, explosive vapour-air mixtures
containers; absorb spilt liquid may be formed; no open flames, no sparks, and no
in sand or inert absorbent and smoking; above 27°C, use closed systems, ventilation,
remove to safe place explosion-proof electrical equipment; in case of fire,
keep drums cool by spraying with water; extinguish
fire with powder, AFFF, foam, halons, or carbon
dioxide
WASTE DISPOSAL
National Occupational Exposure UN: 1212
Limit:
National Poison Control Centre:
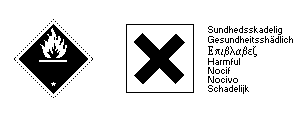 Adapted from: Handling Chemicals Safely (1980) Published by the Dutch Association of Safety Experts, the Dutch Chemical Industry
Association, and the Dutch Safety Institute, The Hague.
6. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Isobutanol should be managed in the environment as a compound that is,
at most, slightly toxic, but that poses an indirect hazard for the
aquatic environment, because of ready biodegradation, which may lead
to oxygen depletion. Thus, in the case of spillage in surface water,
consider dilution or (artificial) re aeration.
Minimize contamination of water. Avoid gross contamination of soil.
Spilled material should be contained and removed as far as possible.
Final traces can be dispersed with water.
Disposal should be acceptable within the requirements of operative
legislation; if necessary, consult local authorities.
7. REGIONS, GUIDELINES, AND STANDARDS
The information given in this paragraph has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. A full reference to the
original national document from which the information was extracted
can be obtained from the IRPTC.a
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is shown, indicated by
(r).
7.1 Exposure Limit Values
For some exposure limit values, see the following table.
7.2 Specific Restrictions
Isobutanol can be used in the USA as a component of adhesives in
articles intended for use in packaging, transporting, or holding of
food.
7.3 Labelling, Packaging, and Transport
Isobutanol is classified as a flammable liquid (Hazard Class 3) by the
United Nations Committee of Experts on the Transport of Dangerous
Goods, as "a toxic waste" in the USA, and as a flammable liquid by the
International Maritime Organization (Hazard Class 3.2). The following
symbol should be used:
Background: red
Adapted from: Handling Chemicals Safely (1980) Published by the Dutch Association of Safety Experts, the Dutch Chemical Industry
Association, and the Dutch Safety Institute, The Hague.
6. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Isobutanol should be managed in the environment as a compound that is,
at most, slightly toxic, but that poses an indirect hazard for the
aquatic environment, because of ready biodegradation, which may lead
to oxygen depletion. Thus, in the case of spillage in surface water,
consider dilution or (artificial) re aeration.
Minimize contamination of water. Avoid gross contamination of soil.
Spilled material should be contained and removed as far as possible.
Final traces can be dispersed with water.
Disposal should be acceptable within the requirements of operative
legislation; if necessary, consult local authorities.
7. REGIONS, GUIDELINES, AND STANDARDS
The information given in this paragraph has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. A full reference to the
original national document from which the information was extracted
can be obtained from the IRPTC.a
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is shown, indicated by
(r).
7.1 Exposure Limit Values
For some exposure limit values, see the following table.
7.2 Specific Restrictions
Isobutanol can be used in the USA as a component of adhesives in
articles intended for use in packaging, transporting, or holding of
food.
7.3 Labelling, Packaging, and Transport
Isobutanol is classified as a flammable liquid (Hazard Class 3) by the
United Nations Committee of Experts on the Transport of Dangerous
Goods, as "a toxic waste" in the USA, and as a flammable liquid by the
International Maritime Organization (Hazard Class 3.2). The following
symbol should be used:
Background: red  a International Register of Potentially Toxic Chemicals, Palais des
Nations, 1211 Geneva 10, Switzerland (Telephone No. 988400-985850).
The European Community legislation requires labelling as a dangerous
substance using the symbol:
a International Register of Potentially Toxic Chemicals, Palais des
Nations, 1211 Geneva 10, Switzerland (Telephone No. 988400-985850).
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read: flammable - harmful by inhalation; keep away from
sources of ignition - no smoking.
The European Community legislation on labelling of solvant
preparations classifies isobutanol in class II d for the purpose of
determining the label for preparations containing isobutanol and other
active ingredients (1980).
SOME EXPOSURE LIMIT VALUES
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
AIR Occupational Argentina Maximum permissible concentration (MPC) 1979
- Time-weighted average 150 mg/m3
- Short-term exposure level (STEL) 225 mg/m3
Australia Threshold limit value (TLV) 1983 (r)
- Time-weighted average 300 mg/m3
Belgium Threshold limit value (TLV) 150 mg/m3
Finland Maximum permissible concentration (MPC) 1982 (r)
- Time-weighted average 300 mg/m3
Germany, Federal Republic of Maximum allowable concentration (MAC) 1985 (r)
- 8-h time-weighted average 300 mg/m3
- Short-term exposure limit (STEL) 600 mg/m3
(30 min, 4 x per shift)
(average value)
Italy Threshold limit value (TLV) 150-225 mg/m3
Netherlands Maximum limit 1985 (r)
- Time-weighted average 150 mg/m3
Romania Maximum permissible concentration (MPC) 1975 (r)
- Time-weighted average 200 mg/m3
- Ceiling value 300 mg/m3
Sweden Maximum tolerable concentration (MTC) 1984 (r)
- One-day time-weighted average 700 mg/m3
- Short-term exposure limit (STEL) 1000 mg/m3
(15-min time-weighted average)
Switzerland Maximum work-site concentration (MAK) 1984 (r)
- Time-weighted average 150 mg/m3
United Kingdom Recommended limit 1985 (r)
- 8-h time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
(10-min time-weighted average)
(cont'd)
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
USA (ACGIH)b Threshold limit value (TLV) 1984 (r)
- Time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
USA (OSHA) Permissible exposure limits (PEL) 1981 (r)
- Time-weighted average 300 mg/m3
USSR Maximum allowable concentration (MAC) 1980 (r)
- Ceiling value for vapour 10 mg/m3
Yugoslavia Maximum allowable concentration (MAC) 1971 (r)
- Time-weighted average 200 mg/m3
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- One time per day 0.1 mg/m3
- Average per day 0.1 mg/m3
FOOD Plant USA Exempted from tolerance requirements 1981 (r)
in plant products when used according
to good agricultural practice as inert
ingredient of pesticides for some
specific use
WATER Surface USSR Maximum allowable concentration (MAC) 0.15 mg/litre 1983
a Unless otherwise defined, time-weighted average is over one working day (usually 8 h).
b Value recommended by the American Conference of Government Industrial Hygienists.
The label must read: flammable - harmful by inhalation; keep away from
sources of ignition - no smoking.
The European Community legislation on labelling of solvant
preparations classifies isobutanol in class II d for the purpose of
determining the label for preparations containing isobutanol and other
active ingredients (1980).
SOME EXPOSURE LIMIT VALUES
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
AIR Occupational Argentina Maximum permissible concentration (MPC) 1979
- Time-weighted average 150 mg/m3
- Short-term exposure level (STEL) 225 mg/m3
Australia Threshold limit value (TLV) 1983 (r)
- Time-weighted average 300 mg/m3
Belgium Threshold limit value (TLV) 150 mg/m3
Finland Maximum permissible concentration (MPC) 1982 (r)
- Time-weighted average 300 mg/m3
Germany, Federal Republic of Maximum allowable concentration (MAC) 1985 (r)
- 8-h time-weighted average 300 mg/m3
- Short-term exposure limit (STEL) 600 mg/m3
(30 min, 4 x per shift)
(average value)
Italy Threshold limit value (TLV) 150-225 mg/m3
Netherlands Maximum limit 1985 (r)
- Time-weighted average 150 mg/m3
Romania Maximum permissible concentration (MPC) 1975 (r)
- Time-weighted average 200 mg/m3
- Ceiling value 300 mg/m3
Sweden Maximum tolerable concentration (MTC) 1984 (r)
- One-day time-weighted average 700 mg/m3
- Short-term exposure limit (STEL) 1000 mg/m3
(15-min time-weighted average)
Switzerland Maximum work-site concentration (MAK) 1984 (r)
- Time-weighted average 150 mg/m3
United Kingdom Recommended limit 1985 (r)
- 8-h time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
(10-min time-weighted average)
(cont'd)
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
USA (ACGIH)b Threshold limit value (TLV) 1984 (r)
- Time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
USA (OSHA) Permissible exposure limits (PEL) 1981 (r)
- Time-weighted average 300 mg/m3
USSR Maximum allowable concentration (MAC) 1980 (r)
- Ceiling value for vapour 10 mg/m3
Yugoslavia Maximum allowable concentration (MAC) 1971 (r)
- Time-weighted average 200 mg/m3
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- One time per day 0.1 mg/m3
- Average per day 0.1 mg/m3
FOOD Plant USA Exempted from tolerance requirements 1981 (r)
in plant products when used according
to good agricultural practice as inert
ingredient of pesticides for some
specific use
WATER Surface USSR Maximum allowable concentration (MAC) 0.15 mg/litre 1983
a Unless otherwise defined, time-weighted average is over one working day (usually 8 h).
b Value recommended by the American Conference of Government Industrial Hygienists.
 Adapted from: Handling Chemicals Safely (1980) Published by the Dutch Association of Safety Experts, the Dutch Chemical Industry
Association, and the Dutch Safety Institute, The Hague.
6. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Isobutanol should be managed in the environment as a compound that is,
at most, slightly toxic, but that poses an indirect hazard for the
aquatic environment, because of ready biodegradation, which may lead
to oxygen depletion. Thus, in the case of spillage in surface water,
consider dilution or (artificial) re aeration.
Minimize contamination of water. Avoid gross contamination of soil.
Spilled material should be contained and removed as far as possible.
Final traces can be dispersed with water.
Disposal should be acceptable within the requirements of operative
legislation; if necessary, consult local authorities.
7. REGIONS, GUIDELINES, AND STANDARDS
The information given in this paragraph has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. A full reference to the
original national document from which the information was extracted
can be obtained from the IRPTC.a
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is shown, indicated by
(r).
7.1 Exposure Limit Values
For some exposure limit values, see the following table.
7.2 Specific Restrictions
Isobutanol can be used in the USA as a component of adhesives in
articles intended for use in packaging, transporting, or holding of
food.
7.3 Labelling, Packaging, and Transport
Isobutanol is classified as a flammable liquid (Hazard Class 3) by the
United Nations Committee of Experts on the Transport of Dangerous
Goods, as "a toxic waste" in the USA, and as a flammable liquid by the
International Maritime Organization (Hazard Class 3.2). The following
symbol should be used:
Background: red
Adapted from: Handling Chemicals Safely (1980) Published by the Dutch Association of Safety Experts, the Dutch Chemical Industry
Association, and the Dutch Safety Institute, The Hague.
6. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Isobutanol should be managed in the environment as a compound that is,
at most, slightly toxic, but that poses an indirect hazard for the
aquatic environment, because of ready biodegradation, which may lead
to oxygen depletion. Thus, in the case of spillage in surface water,
consider dilution or (artificial) re aeration.
Minimize contamination of water. Avoid gross contamination of soil.
Spilled material should be contained and removed as far as possible.
Final traces can be dispersed with water.
Disposal should be acceptable within the requirements of operative
legislation; if necessary, consult local authorities.
7. REGIONS, GUIDELINES, AND STANDARDS
The information given in this paragraph has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. A full reference to the
original national document from which the information was extracted
can be obtained from the IRPTC.a
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is shown, indicated by
(r).
7.1 Exposure Limit Values
For some exposure limit values, see the following table.
7.2 Specific Restrictions
Isobutanol can be used in the USA as a component of adhesives in
articles intended for use in packaging, transporting, or holding of
food.
7.3 Labelling, Packaging, and Transport
Isobutanol is classified as a flammable liquid (Hazard Class 3) by the
United Nations Committee of Experts on the Transport of Dangerous
Goods, as "a toxic waste" in the USA, and as a flammable liquid by the
International Maritime Organization (Hazard Class 3.2). The following
symbol should be used:
Background: red  a International Register of Potentially Toxic Chemicals, Palais des
Nations, 1211 Geneva 10, Switzerland (Telephone No. 988400-985850).
The European Community legislation requires labelling as a dangerous
substance using the symbol:
a International Register of Potentially Toxic Chemicals, Palais des
Nations, 1211 Geneva 10, Switzerland (Telephone No. 988400-985850).
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read: flammable - harmful by inhalation; keep away from
sources of ignition - no smoking.
The European Community legislation on labelling of solvant
preparations classifies isobutanol in class II d for the purpose of
determining the label for preparations containing isobutanol and other
active ingredients (1980).
SOME EXPOSURE LIMIT VALUES
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
AIR Occupational Argentina Maximum permissible concentration (MPC) 1979
- Time-weighted average 150 mg/m3
- Short-term exposure level (STEL) 225 mg/m3
Australia Threshold limit value (TLV) 1983 (r)
- Time-weighted average 300 mg/m3
Belgium Threshold limit value (TLV) 150 mg/m3
Finland Maximum permissible concentration (MPC) 1982 (r)
- Time-weighted average 300 mg/m3
Germany, Federal Republic of Maximum allowable concentration (MAC) 1985 (r)
- 8-h time-weighted average 300 mg/m3
- Short-term exposure limit (STEL) 600 mg/m3
(30 min, 4 x per shift)
(average value)
Italy Threshold limit value (TLV) 150-225 mg/m3
Netherlands Maximum limit 1985 (r)
- Time-weighted average 150 mg/m3
Romania Maximum permissible concentration (MPC) 1975 (r)
- Time-weighted average 200 mg/m3
- Ceiling value 300 mg/m3
Sweden Maximum tolerable concentration (MTC) 1984 (r)
- One-day time-weighted average 700 mg/m3
- Short-term exposure limit (STEL) 1000 mg/m3
(15-min time-weighted average)
Switzerland Maximum work-site concentration (MAK) 1984 (r)
- Time-weighted average 150 mg/m3
United Kingdom Recommended limit 1985 (r)
- 8-h time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
(10-min time-weighted average)
(cont'd)
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
USA (ACGIH)b Threshold limit value (TLV) 1984 (r)
- Time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
USA (OSHA) Permissible exposure limits (PEL) 1981 (r)
- Time-weighted average 300 mg/m3
USSR Maximum allowable concentration (MAC) 1980 (r)
- Ceiling value for vapour 10 mg/m3
Yugoslavia Maximum allowable concentration (MAC) 1971 (r)
- Time-weighted average 200 mg/m3
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- One time per day 0.1 mg/m3
- Average per day 0.1 mg/m3
FOOD Plant USA Exempted from tolerance requirements 1981 (r)
in plant products when used according
to good agricultural practice as inert
ingredient of pesticides for some
specific use
WATER Surface USSR Maximum allowable concentration (MAC) 0.15 mg/litre 1983
a Unless otherwise defined, time-weighted average is over one working day (usually 8 h).
b Value recommended by the American Conference of Government Industrial Hygienists.
The label must read: flammable - harmful by inhalation; keep away from
sources of ignition - no smoking.
The European Community legislation on labelling of solvant
preparations classifies isobutanol in class II d for the purpose of
determining the label for preparations containing isobutanol and other
active ingredients (1980).
SOME EXPOSURE LIMIT VALUES
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
AIR Occupational Argentina Maximum permissible concentration (MPC) 1979
- Time-weighted average 150 mg/m3
- Short-term exposure level (STEL) 225 mg/m3
Australia Threshold limit value (TLV) 1983 (r)
- Time-weighted average 300 mg/m3
Belgium Threshold limit value (TLV) 150 mg/m3
Finland Maximum permissible concentration (MPC) 1982 (r)
- Time-weighted average 300 mg/m3
Germany, Federal Republic of Maximum allowable concentration (MAC) 1985 (r)
- 8-h time-weighted average 300 mg/m3
- Short-term exposure limit (STEL) 600 mg/m3
(30 min, 4 x per shift)
(average value)
Italy Threshold limit value (TLV) 150-225 mg/m3
Netherlands Maximum limit 1985 (r)
- Time-weighted average 150 mg/m3
Romania Maximum permissible concentration (MPC) 1975 (r)
- Time-weighted average 200 mg/m3
- Ceiling value 300 mg/m3
Sweden Maximum tolerable concentration (MTC) 1984 (r)
- One-day time-weighted average 700 mg/m3
- Short-term exposure limit (STEL) 1000 mg/m3
(15-min time-weighted average)
Switzerland Maximum work-site concentration (MAK) 1984 (r)
- Time-weighted average 150 mg/m3
United Kingdom Recommended limit 1985 (r)
- 8-h time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
(10-min time-weighted average)
(cont'd)
MEDIUM SPECIFICATION COUNTRY/ EXPOSURE LIMIT DESCRIPTIONa VALUE EFFECTIVE
ORGANIZATION DATE
USA (ACGIH)b Threshold limit value (TLV) 1984 (r)
- Time-weighted average 150 mg/m3
- Short-term exposure limit (STEL) 225 mg/m3
USA (OSHA) Permissible exposure limits (PEL) 1981 (r)
- Time-weighted average 300 mg/m3
USSR Maximum allowable concentration (MAC) 1980 (r)
- Ceiling value for vapour 10 mg/m3
Yugoslavia Maximum allowable concentration (MAC) 1971 (r)
- Time-weighted average 200 mg/m3
AIR Ambient USSR Maximum allowable concentration (MAC) 1984
- One time per day 0.1 mg/m3
- Average per day 0.1 mg/m3
FOOD Plant USA Exempted from tolerance requirements 1981 (r)
in plant products when used according
to good agricultural practice as inert
ingredient of pesticides for some
specific use
WATER Surface USSR Maximum allowable concentration (MAC) 0.15 mg/litre 1983
a Unless otherwise defined, time-weighted average is over one working day (usually 8 h).
b Value recommended by the American Conference of Government Industrial Hygienists.
