
WHO FOOD ADDITIVES SERIES: 52
INTRODUCTION
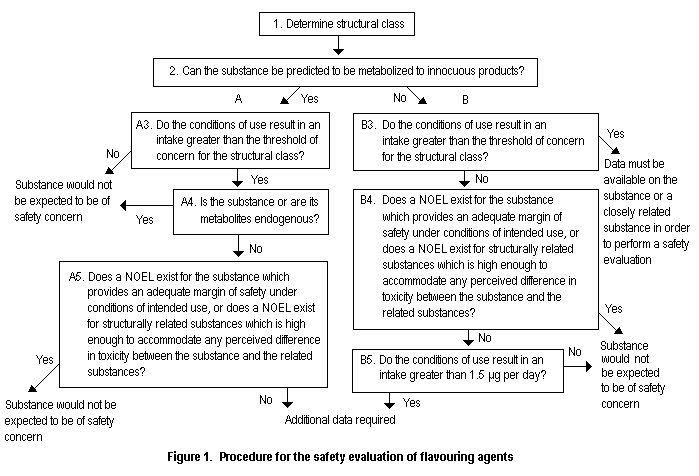
Four groups of flavouring agents and additional compounds for two previously published groups were evaluated using the Procedure for the Safety Evaluation of Flavouring Agents as outlined in Figure 1 (Annex 1, references 116, 122, 131, 137, 143, 149, 154 and 160). In applying the Procedure, the chemical is first assigned to a structural class as identified by the Committee at its forty-sixth meeting (Annex 1, reference 122). The structural classes are as follows:
A key element of the Procedure involves determining whether a flavouring agent and the product(s) of its metabolism are innocuous and/or endogenous substances. For the purpose of the evaluations, the Committee used the following definitions, adapted from the report of its fortysixth meeting:
Innocuous metabolic products are defined as products that are known or readily predicted to be harmless to humans at the estimated intake of the flavouring agent.
Endogenous substances are intermediary metabolites normally present in human tissues and fluids, whether free or conjugated; hormones and other substances with biochemical or physiological regulatory functions are not included. The estimated intake of a flavouring agent that is, or is metabolized to, an endogenous substance should be judged not to give rise to perturbations outside the physiological range.
INTAKE
Estimates of the intake of flavouring agents by populations typically involve the acquisition of data on the amounts used in food. These data were derived from surveys in Europe and the USA. In Europe, a survey was conducted in 1995 by the International Organization of the Flavour Industry, in which flavour manufacturers reported the total amount of each flavouring agent incorporated into food sold in the European Union during the previous year. Manufacturers were requested to exclude use of flavouring agents in pharmaceutical, tobacco or cosmetic products.

Figure 1. Procedure for the safety evaluation of flavouring agents
In the USA, a series of surveys was conducted between 1970 and 1987 by the National Academy of Sciences National Research Council (under contract to the Food & Drug Administration) in which information was obtained from ingredient manufacturers and food processors on the amount of each substance destined for addition to the food supply and on the usual and maximal levels at which each substance was added in a number of broad food categories.
In using the data from these surveys to estimate intakes of flavouring agents, it was assumed that only 60% of the total amount used is reported in Europe and 80% of the amount used is reported in the USA and that the total amount used in food is consumed by only 10% of the population.

The population of consumers was assumed to be 32 × 106 in Europe and 26 × 106 in the USA.
Several of the flavouring agents that were evaluated at the present meeting were not included in the above surveys or were placed on the market after the surveys were conducted. Intakes of these flavouring agents were estimated on the basis of anticipated use by the manufacturer in the USA, and the standard formula was applied.
At its present meeting, the Committee also considered the evaluation of natural flavouring complexes and decided to apply, and expand if necessary, the decision-tree approach developed for individual flavouring substances (see The safety evaluation of natural flavouring complexes, p 435). The Committee also decided to test this approach on a few selected compounds at its next meeting.
See Also:
Toxicological Abbreviations