
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
USERS' MANUAL FOR THE IPCS HEALTH AND SAFETY GUIDES
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1996
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation
and the World Health Organization)
WORLD HEALTH ORGANIZATION, GENEVA
WHO Library Cataloguing in Publication Data
International Programme on Chemical Safety
Users' manual for the IPCS health and safety guides.
1.Occupational exposure 2.Occupational diseases
3.Hazardous substances 4.Guidelines I.Title
ISBN 92 4 154485 6 (NLM Classification: WA 465)
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions and reprints and translations already
available.
(c) World Health Organization 1996
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
PREFACE
INTRODUCTION
HOW TO USE THIS MANUAL
1. PRODUCT IDENTITY AND USES
1.1. The need to identify chemicals in the workplace and
environment
1.1.1. Basic chemistry
1.1.2. Terminology
1.2. Physical and chemical properties
1.2.1. Physical state
1.2.2. Smell and odour threshold
1.2.3. Physical properties
1.2.4. Composition
1.3. Analytical methods
1.4. Production and uses
2. EXPOSURE, TOXICITY AND RISK
2.1. Exposure
2.1.1. Inhalation
2.1.1.1 Forms
2.1.1.2 Entry into the body
2.1.2. Absorption through the skin
2.1.2.1 Forms
2.1.2.2 Entry into the body
2.1.3. Absorption through the eye
2.1.3.1 Forms
2.1.3.2 Entry into the body
2.1.4. Ingestion
2.1.4.1 Forms
2.1.4.2 Entry into the body
2.2. Processing of chemicals in the body
2.2.1. Metabolism
2.2.2. Excretion
2.2.3. Storage or accumulation
2.3. Toxic effects of chemicals
2.4. Effects on body systems
2.5. Dose, effects and response
2.6. How is the toxicity of a chemical determined?
2.6.1. Animal studies
2.6.1.1 Acute toxicity tests
2.6.1.2 Subchronic toxicity tests
2.6.1.3 Chronic (lifetime) bioassays
2.6.1.4 Short-term mutagenicity tests
2.6.1.5 Reproductive studies
2.6.1.6 Behavioural tests
2.6.2. Human evidence
2.6.2.1 Case reports
2.6.2.2 Epidemiological studies
2.6.3. Environmental assays
2.7. Assessing hazards, risks and safety
2.7.1. Hazard
2.7.2. Risk
2.7.3. Safety
3. THE PREVENTION AND CONTROL OF CHEMICAL HAZARDS: HOW TO IMPROVE
CHEMICAL SAFETY
3.1. Identification
3.2. Evaluation
3.3. Safety organization
3.4. Controlling the hazard
3.4.1. Substitution
3.4.2. Engineering controls
3.4.2.1 Total enclosure
3.4.2.2 Ventilation
3.4.3. Safe working procedures
3.4.4. Reducing the number of exposed workers
and their duration of exposure
3.4.5. Personal protective equipment
3.4.5.1 Principles
3.4.5.2 Protective clothing
3.4.5.3 Gloves
3.4.5.4 Do you need a respirator?
3.4.5.5 Types of respirators
3.4.5.6 Training and fitting
3.4.5.7 Respiratory protection programme
3.5. Monitoring the hazard
3.5.1. Environmental monitoring
3.5.2. Biological monitoring
4. PROTECTION AND EMERGENCY ACTION
4.1. First aid and emergencies
4.2. Health surveillance
4.3. Fire and explosion hazards
4.3.1. Flammable substances
4.3.2. Dust explosions
4.3.3. Sources of ignition
4.3.4. Fire-fighting
4.4. Storage
4.5. Spillage
4.6. Disposal
4.6.1. Means of disposal
4.6.2. An example of the effects of incorrect waste
disposal
5. HOW CHEMICALS CAN POISON THE ENVIRONMENT
5.1. How do chemicals get into the environment?
5.2. How do chemicals affect the environment?
5.2.1. Air
5.2.2. Water and soil
5.3. Factors affecting a cemical's environmental impact
5.4. The importance of prevention
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. REGULATIONS, GUIDELINES AND STANDARDS
7.1. Exposure limits
7.1.1. Threshold limit value
7.1.2. Maximum allowable concentration
7.1.3. Other terms for occupational exposure limits
7.1.4. Health-based exposure limits
7.1.5. Exposure limits for food products
7.2. Specific restrictions
7.3. Labelling, packaging and transport
8. GETTING FURTHER INFORMATION
ANNEX: LIST OF HEALTH AND SAFETY GUIDES
SELECTED ACRONYMS AND ABBREVIATIONS
ADI Acceptable Daily Intake
EC European Community
FAO Food and Agriculture Organization of the United Nations
HSG Health and Safety Guide
ICSC International Chemical Safety Card
ILO International Labour Organisation
IPCS International Programme on Chemical Safety
kPa kilopascals
MEK methylethylketone
mg/m3 milligram per cubic metre
MSDS material safety data sheet
PCP pentachlorophenol
ppm parts per million
UN United Nations
WHO World Health Organization
PREFACE
This manual summarizes the basic information concerning chemical
safety that is required for understanding the terms and concepts used
in the IPCS Health and Safety Guides. The first draft was prepared by
the Workers Health Centre, Lidcombe, New South Wales, Australia. It
was distributed for comments to IPCS Participating Institutions, other
agencies involved in chemical safety and nongovernmental organizations
representing trade unions, employers and consumers.
The efforts of all who provided comments and materials for the manual,
of Mr L. Strange and Ms J. Connor in coordinating the preparation of
the first draft and of Ms A. Rice and Ms M. Sheffer in undertaking the
technical editing are gratefully acknowledged. Production of the
manual was supervised for the IPCS by the WHO Office of Global and
Integrated Environmental Health. Financial support was provided by
the IPCS and the Federal Ministry for the Environment, Nature
Conservation and Nuclear Safety, Germany.
INTRODUCTION
The rapid development of chemical products during the last 100 years
has improved the quality of life. However, this has not been without
cost to the community, and particularly to those who work directly
with chemicals. It is estimated that chemical exposure at work alone
is responsible for at least 4% of all deaths from cancer, reaching as
high as 80% for certain types of cancer. Large numbers of workers may
suffer from other forms of illness caused by chemicals. The long-term
effects of chemical pollution of the environment, and of our food, are
only now beginning to be realized. In some polluted areas, major
outbreaks of poisoning have occurred; chemical food poisoning is an
increasing concern.
It is therefore essential that everyone involved in the manufacture,
use, storage, transport and disposal of chemicals is fully aware of
the dangers that many chemicals can pose to health.
The International Programme on Chemical Safety (IPCS) (jointly
sponsored by the United Nations Environment Programme, the
International Labour Organisation (ILO) and the World Health
Organization) produces a series of Health and Safety Guides (HSGs) and
one of International Chemical Safety Cards (ICSCs), which summarize
what is known about the health effects of chemicals commonly used in
the working environment.
The target audience of the HSGs includes occupational health services;
individuals working in ministries, governmental agencies, industry and
trade unions who seek to promote the safe use of chemicals and the
avoidance of environmental health hazards; and those who wish to
obtain more information on this topic. The ICSCs are intended for use
by workers on the "shop-floor" and other interested parties in
factories, agriculture, construction and other places of work.
An attempt has been made in both the HSGs and the ICSCs to use terms
that are familiar to the reader. However, some technical terms are
inevitable, so this Manual provides some background to the technical
terms and concepts used in the HSGs and ICSCs. The principal target
audience for this Manual consists of workers' health and safety
representatives and members of community organizations active on
environmental chemical issues. Nevertheless, the HSGs will have a
wider readership.
This Manual demonstrates how the information in the HSGs can be put to
practical use to improve the health and safety of workers and the
general community. The methods for improving health and safety
outlined in this Manual should thus be regarded as essential parts of
the chemical production, use and disposal process. They are not
luxuries or optional requirements. Rather, they are an integral part
of chemical use for all workers in all workplaces -- in the
factories, fields, offices and multitude of other locations that
constitute the world's workplaces -- in both developed and developing
nations. Chemical safety may be integrated relatively easily in
"fixed" workplaces, in manufacturing and the larger downstream
industries that are users of chemicals, as the resources and structure
for medical services, occupational hygienists, joint worker/employer
health and safety committees, etc. are often available. Nevertheless,
the information and principles outlined in this Manual also apply to
users of chemicals in agriculture (many HSGs and ICSCs are concerned
with pesticides) and other workplaces.
The contents of this Manual will be reviewed and revised as new HSGs
are published. You can help by letting us know about any difficulties
you have in using this Manual or the HSGs and ICSCs, and any sections
or points that could be improved.
Please address all comments to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
HOW TO USE THIS MANUAL
The format of this Manual closely follows the format of the IPCS
Health and Safety Guides. Each chapter of the Manual relates to a
corresponding chapter in the HSGs.
It is not necessary to read the Manual from cover to cover all at
once. The Manual is best used as a reference in the practical tasks of
worker protection.
You can use an HSG on a particular chemical as follows:
a) Read Chapter 1 of this Manual -- "Product Identity and Uses".
After reading this chapter, try to identify which chemicals are being
used in your workplace.
After drawing up a list of chemicals, obtain the relevant HSG (see the
list of published guides in the Annex). You may also wish to seek
further health and safety information on the product concerned (see
Chapter 8 -- "Getting Further Information").
b) Read each chapter of the HSG, followed by the corresponding
chapter of the Manual. (Although great effort has been made in this
Manual to follow the layout and general headings of the HSGs, this
has not always been possible. This is especially the case when
cross-references are made to sections of the text and numbers of some
of the figures reproduced directly from the HSGs. Moreover, the HSGs
themselves have evolved over time, so that the numbering of sections
within them has changed. However, each chapter in the Manual does
correspond to the relevant chapter in the HSGs.) After reading each
section, think about how the information in the HSG applies to your
place of work. Make a list of possible improvements that could be
made to chemical storage, handling, usage and disposal, and of
possible improvements that could be made to protect workers from
unnecessary exposure to chemicals. Also, list any questions about the
hazards of chemicals used in your workplace.
c) Discuss this list of questions and proposals with representatives
of your trade union or health and safety committee. If these channels
are not available, bring up the issues directly with your co-workers.
For more information on the role of trade unions and health and safety
committees and some suggestions on how to organize for better safety,
turn to the section entitled "Safety organization" in Chapter 3 of
this Manual. Safety questions and proposals can also be discussed
with your immediate supervisor or safety officer.
After discussions with your trade union or health and safety
committee, formally present your comments to management and ask for a
response within a specified time.
If there is an unsatisfactory or no response from management, or if
there is a positive response but no action, raise the matter again
through the agreed grievance or dispute-settling procedures of your
workplace. Always insist that reasonable time limits are set for the
resolution of the dispute.
If all of the above steps fail, contact the health and safety officer
of your trade union or some other suitable official. Send a copy of
your questions and suggestions to the Factory Inspectorate or other
government body responsible for health and safety in the working
environment. In most countries you have the right to remain anonymous
when contacting the Factory Inspectorate.
If you are a member of the public who considers that harmful pollution
of the environment by chemicals is occurring, contact your local
member of government and the local or national government body
responsible for pollution control, the environment and food safety.
Many government departments can provide information about the health
effects of chemicals in the workplace and general environment.
Moreover, there is an increasing number of nongovernmental bodies,
operated by trade union, industry and public interest groups, that
offer similar information services.
But before approaching any of these information services for further
assistance, it is always helpful to collect as much information as
possible about the names and manufacturers of chemicals used in your
workplace. Refer to Chapters 6, 7 and 8 for details and ideas on where
and how to find information on chemicals.
1. PRODUCT IDENTITY AND USES - CHAPTER 1
This chapter should be read in conjunction with Chapter 1 (Product
Identity and Uses) of the relevant HSG.
1.1 The need to identify chemicals in the workplace and environment
Everyone should have the right to know the identity and nature of any
chemicals to which he or she may be exposed in the workplace or
general environment (e.g. through contamination of air, water or
food).
Without knowledge of the health effects of the chemicals with which
they work, workers and managers are unable to protect themselves, or
others, effectively from any adverse health effects that these
chemicals might cause.
The first essential step is to determine the exact identity of the
chemicals that are present in the workplace or environment.
Unfortunately, more often than not, chemicals are labelled in the
workplace by trade name only or are known under a rare synonym. To
follow up on the potential health hazards, however, it is necessary to
know the chemical name of the product (see Fig. 1). Although the
way in which chemicals are named may seem confusing or even
intimidating, some basic knowledge of chemistry is all that is needed
to determine their nature correctly.
 1.1.1 Basic chemistry
All matter (i.e. liquids, solid substances and gases) is made of
elements. An element is the simplest sort of matter that can exist
by itself. So far, 106 different elements are known, including
oxygen, nitrogen, carbon, aluminium, copper and iron.
An atom is the smallest unit of an element that can still retain the
properties of the element. The atoms of the same element are exactly
alike, with the same mass, etc. The atoms of different elements,
however, have different masses and also differ in their ability to
combine with other atoms.
When atoms of one element combine chemically with atoms of the same
element or other elements, the result is a molecule. The process
whereby atoms combine and rearrange themselves is known as a chemical
reaction. Chemical reactions may occur slowly, taking hours or days
to complete, or they may occur suddenly or violently, as in an
explosion.
Combinations of two or more different elements are also called
chemical compounds. Substances such as water and carbon dioxide,
which can be broken down into their constituent elements -- hydrogen
and oxygen in the case of water, and carbon and oxygen in the case of
carbon dioxide -- are examples of compounds. There are nearly six
million different chemical compounds known, each with its own
particular combination of the atoms of various elements. This number
is increasing all the time. However, the number of potentially useful
chemical compounds is much less, and about 1500 chemicals account for
90% of the total volume of all chemical compounds produced.
A pure substance is a chemical that contains only a single chemical
compound or element, i.e. all the molecules are the same. The
compound that makes up the pure substance may contain the atoms of
many different types of elements, all combined chemically. A
mixture, on the other hand, is a substance that contains more than
one chemical compound or element, the separate constituents of which
will still have their own properties.
A suspension is a mixture of a liquid and tiny particles of a solid
substance (powder). A mist or aerosol is the mixture of tiny droplets
of a liquid, or tiny particles of a solid, in a gas. Although the
powder or droplets may be very fine, each particle of powder or each
droplet contains many millions of molecules of the chemical compound.
In a solution, individual molecules of one substance (the solute)
are dissolved in another substance (the solvent).
1.1.2 Terminology
Chemicals are identified in the HSGs as follows (see Fig. 2):
Common name -- This is the name that appears on the cover of the HSG
and is the commonly used name of the chemical in question. Sometimes
the common name refers to the elements that make up a chemical
compound, e.g. hydrogen sulfide contains elements of hydrogen and
sulfur.
Chemical formula -- The chemical formula uses symbols to represent
the atoms of each element contained in each molecule of the chemical
compound in question.
Table 1 gives the symbols of some of the more common elements. These
symbols come from the Latin name (Au for gold -- Latin: Aurum; Cu for
copper -- Latin: Cuprum). Sometimes the first letter is the same in
the English name (O for oxygen, C for carbon).
A formula for a molecule will also show a small number (or
subscript) below the level of the symbol, which indicates how many
atoms of that particular element are present in the molecule.
Using the example shown in Fig. 2, the chemical formula for phosphorus
trichloride -- PCl3 -- shows that it consists of one atom of
phosphorus and three of chlorine; the formula for phosphorus
oxychloride -- POCl3 -- shows that the chemical is made of one atom
of phosphorus, one of oxygen and three of chlorine.
Figure 2. Chemical identity (from HSG 35: Phosphorus Trichloride)
1.1 Identity
Phosphorus Phosphorus
Trichloride Oxychloride
Chemical formula: PCl3 POCl3
Chemical structure: Cl Cl
| |
Cl -- P Cl P == O
| |
Cl Cl
Common synonyms: Phosphorus chloride; Phosphorus
phosphorus chloride; oxytrichloride
trichlorophosphine
Abbreviations: Tri; "Pickle" Oxy; "Pockle"
CAS registry number: 7719-12-2 10025-87-3
RTECS number: TH3675000 TH4897000
UN number: 1809 1810 (II)
Conversion 1 ppm = 5.62 mg/m3 and 1 ppm = 6.27 mg/m3 and
factor: 1 mg/m3 = 0.178 ppm 1 mg/m3 = 0.159 ppm
[at 25°C and 101.3 kPa [at 25°C and 101.3 kPa
(760 mmHg, 1 bar)] 760 mmHg, 1 bar)]
Table 1. Selected elements and their symbols
Element Symbol Element Symbol
Aluminium Al Magnesium Mg
Bromine Br Mercury Hg
Calcium Ca Nitrogen N
Carbon C Oxygen O
Chlorine Cl Phosphorus P
Chromium Cr Potassium K
Copper Cu Silicon Si
Gold Au Sodium Na
Helium He Sulfur S
Hydrogen H Tin Sn
Iron Fe Uranium U
Lead Pb Zinc Zn
Chemical structure -- The chemical formula tells us how many atoms
of each element are combined in a compound, but not about how these
atoms are arranged.
The chemical structure diagram is a graphical representation of the
pattern in which atoms are combined in a chemical compound. Such
diagrams give information about the arrangement in two dimensions
only: in real life, molecules are not flat. Nevertheless, the
chemical structure can constitute important information about the
potential health effects of a chemical.
It is possible for two different chemical compounds to have the same
chemical formula (i.e. the same type and number of atoms in each
molecule) but different chemical structures (i.e. the atoms have
combined in a different arrangement). When this is the case, the
chemicals are known as isomers of each other. Isomers are often
identified by one or more numbers, letters or Greek symbols in front
of the same chemical name. For example, 1-butanol, 2-butanol,
isobutanol and tert-butanol all have the same chemical formula
(C4H10O) but different chemical structures (see Fig. 3).
There may be little or no difference between the effects of different
isomers on the health of those exposed to them. But in many cases
there are very important differences between the health effects of
isomers.
For example, 1,1,2-trichloroethane has been shown to cause cancer in
experimental animals, although the evidence is limited. On the other
hand, 1,1,1-trichloroethane has not been shown to cause cancer in
animals. The exposure limit for 1,1,1-trichloroethane in most
countries is much higher than that for 1,1,2-trichloroethane,
indicating that the former is thought to be much less hazardous. It
is easy to see that only a small change in the structure of a chemical
may make it much more toxic (or less so). The lesson from this is
that numbers and letters are an important part of some chemical names,
so these should be carefully noted when checking a substance.
Common trade names -- Manufacturers often choose to give chemical
compounds, or mixtures of chemical compounds, commercial names.
This is usually done to make advertising and selling of the product
easier (many chemical names are very long or difficult to remember) or
to distinguish a product from that of a competing manufacturer. Often
trade names refer to the name of the manufacturer or to the use of the
product (e.g. Cellosolve was originally used to dissolve cellulose in
lacquers). A trade name may also be used because it conceals
knowledge of what is in a product or how it is made. However, this
should never be accepted as a reason for concealing the identity of
chemicals from those who work with them.
Common synonyms -- Synonyms are alternative names for the same
substance. The same chemical may have a number of different names
because of the many different chemical naming systems that have
developed, e.g. phosphorus trichloride is also known as phosphorus
chloride or trichlorophosphine.
Figure 3. Identity of chemical isomers
(from HSGs on butanol isomers)
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3-CH2-CH2-CH2OH
Primary constituent: 1-butanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3-CHOH-CH2-CH3
Primary constituent: 2-butanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3
\
CH-CH2OH
/
CH3
Primary constituent: isobutanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3
|
CH3 -- C -- CH3
|
OH
Primary constituent: tert-butanol
CAS registry number -- This is a number assigned to every chemical
by the Chemical Abstracts Service, a section of the American
Chemical Society, which publishes guides to chemical research. Each
chemical is given a unique number.
The CAS number does not give any information about the properties of
the chemical itself. Its value is in overcoming the confusion caused
by the different chemical naming systems (see "Common synonyms"
above). The CAS number will often be given in product information or
literature that accompanies chemicals.
RTECS number -- RTECS stands for Registry of Toxic Effects of
Chemical Substances. This is a list of scientific articles on the
health effects of the chemicals. The Registry is operated by the
National Institute for Occupational Safety and Health, a government
body in the United States. Each chemical that is listed is given a
unique number. About 25 000 chemicals commonly used in industry are
listed.
Other numbering systems and nomenclatures are also used for
classifying chemicals. Those used in the HSGs include the IUPAC
(International Union for Pure and Applied Chemistry) name, EC numbers
assigned by the European Community and UN numbers for the transport of
dangerous goods.
Conversion factor -- The concentration or amount of chemical present
in air, water or food can be expressed in a number of different units.
The conversion factor allows these measurements to be converted from
one to another: "ppm" stands for parts of the chemical per million
parts of air and thus is a ratio of volumes; "mg/m3" stands for
milligrams of the contaminant per cubic metre of air; "kPa" stands for
kilopascals, a unit of pressure; and "mmHg" means millimetres of
mercury (760 mmHg is the normal atmospheric pressure at sea level).
1.2 Physical and chemical properties
1.2.1 Physical state
This section of the HSG includes a description of the physical form of
the chemical.
Chemicals may be gases, liquids or solids. They can change
between these "states" depending on temperature and pressure. For
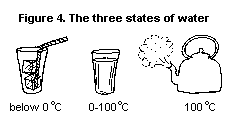
instance, water is a liquid within the temperature range 0-100 degrees
Celsius (°C). Below 0°C, water is in a solid state (ice), and above
100°C it is in a gaseous state (steam) (see Fig. 4). When the
temperature of a solid is increased, it generally turns into a liquid,
i.e. it melts. If the liquid is heated further, it will turn into a
gas or vapour and boil or evaporate. If pressure is increased without
a change in the temperature, gases may convert from the gaseous to a
liquid state.
Some chemicals can exist in only two states, passing from solid to gas
and vice versa without going through a liquid phase.
1.2.2 Smell and odour threshold
The smell of a chemical can sometimes serve as a warning of exposure.
However, it must always be remembered that smell is not a
reliable means of warning, as many chemicals have no smell
at all.
1.1.1 Basic chemistry
All matter (i.e. liquids, solid substances and gases) is made of
elements. An element is the simplest sort of matter that can exist
by itself. So far, 106 different elements are known, including
oxygen, nitrogen, carbon, aluminium, copper and iron.
An atom is the smallest unit of an element that can still retain the
properties of the element. The atoms of the same element are exactly
alike, with the same mass, etc. The atoms of different elements,
however, have different masses and also differ in their ability to
combine with other atoms.
When atoms of one element combine chemically with atoms of the same
element or other elements, the result is a molecule. The process
whereby atoms combine and rearrange themselves is known as a chemical
reaction. Chemical reactions may occur slowly, taking hours or days
to complete, or they may occur suddenly or violently, as in an
explosion.
Combinations of two or more different elements are also called
chemical compounds. Substances such as water and carbon dioxide,
which can be broken down into their constituent elements -- hydrogen
and oxygen in the case of water, and carbon and oxygen in the case of
carbon dioxide -- are examples of compounds. There are nearly six
million different chemical compounds known, each with its own
particular combination of the atoms of various elements. This number
is increasing all the time. However, the number of potentially useful
chemical compounds is much less, and about 1500 chemicals account for
90% of the total volume of all chemical compounds produced.
A pure substance is a chemical that contains only a single chemical
compound or element, i.e. all the molecules are the same. The
compound that makes up the pure substance may contain the atoms of
many different types of elements, all combined chemically. A
mixture, on the other hand, is a substance that contains more than
one chemical compound or element, the separate constituents of which
will still have their own properties.
A suspension is a mixture of a liquid and tiny particles of a solid
substance (powder). A mist or aerosol is the mixture of tiny droplets
of a liquid, or tiny particles of a solid, in a gas. Although the
powder or droplets may be very fine, each particle of powder or each
droplet contains many millions of molecules of the chemical compound.
In a solution, individual molecules of one substance (the solute)
are dissolved in another substance (the solvent).
1.1.2 Terminology
Chemicals are identified in the HSGs as follows (see Fig. 2):
Common name -- This is the name that appears on the cover of the HSG
and is the commonly used name of the chemical in question. Sometimes
the common name refers to the elements that make up a chemical
compound, e.g. hydrogen sulfide contains elements of hydrogen and
sulfur.
Chemical formula -- The chemical formula uses symbols to represent
the atoms of each element contained in each molecule of the chemical
compound in question.
Table 1 gives the symbols of some of the more common elements. These
symbols come from the Latin name (Au for gold -- Latin: Aurum; Cu for
copper -- Latin: Cuprum). Sometimes the first letter is the same in
the English name (O for oxygen, C for carbon).
A formula for a molecule will also show a small number (or
subscript) below the level of the symbol, which indicates how many
atoms of that particular element are present in the molecule.
Using the example shown in Fig. 2, the chemical formula for phosphorus
trichloride -- PCl3 -- shows that it consists of one atom of
phosphorus and three of chlorine; the formula for phosphorus
oxychloride -- POCl3 -- shows that the chemical is made of one atom
of phosphorus, one of oxygen and three of chlorine.
Figure 2. Chemical identity (from HSG 35: Phosphorus Trichloride)
1.1 Identity
Phosphorus Phosphorus
Trichloride Oxychloride
Chemical formula: PCl3 POCl3
Chemical structure: Cl Cl
| |
Cl -- P Cl P == O
| |
Cl Cl
Common synonyms: Phosphorus chloride; Phosphorus
phosphorus chloride; oxytrichloride
trichlorophosphine
Abbreviations: Tri; "Pickle" Oxy; "Pockle"
CAS registry number: 7719-12-2 10025-87-3
RTECS number: TH3675000 TH4897000
UN number: 1809 1810 (II)
Conversion 1 ppm = 5.62 mg/m3 and 1 ppm = 6.27 mg/m3 and
factor: 1 mg/m3 = 0.178 ppm 1 mg/m3 = 0.159 ppm
[at 25°C and 101.3 kPa [at 25°C and 101.3 kPa
(760 mmHg, 1 bar)] 760 mmHg, 1 bar)]
Table 1. Selected elements and their symbols
Element Symbol Element Symbol
Aluminium Al Magnesium Mg
Bromine Br Mercury Hg
Calcium Ca Nitrogen N
Carbon C Oxygen O
Chlorine Cl Phosphorus P
Chromium Cr Potassium K
Copper Cu Silicon Si
Gold Au Sodium Na
Helium He Sulfur S
Hydrogen H Tin Sn
Iron Fe Uranium U
Lead Pb Zinc Zn
Chemical structure -- The chemical formula tells us how many atoms
of each element are combined in a compound, but not about how these
atoms are arranged.
The chemical structure diagram is a graphical representation of the
pattern in which atoms are combined in a chemical compound. Such
diagrams give information about the arrangement in two dimensions
only: in real life, molecules are not flat. Nevertheless, the
chemical structure can constitute important information about the
potential health effects of a chemical.
It is possible for two different chemical compounds to have the same
chemical formula (i.e. the same type and number of atoms in each
molecule) but different chemical structures (i.e. the atoms have
combined in a different arrangement). When this is the case, the
chemicals are known as isomers of each other. Isomers are often
identified by one or more numbers, letters or Greek symbols in front
of the same chemical name. For example, 1-butanol, 2-butanol,
isobutanol and tert-butanol all have the same chemical formula
(C4H10O) but different chemical structures (see Fig. 3).
There may be little or no difference between the effects of different
isomers on the health of those exposed to them. But in many cases
there are very important differences between the health effects of
isomers.
For example, 1,1,2-trichloroethane has been shown to cause cancer in
experimental animals, although the evidence is limited. On the other
hand, 1,1,1-trichloroethane has not been shown to cause cancer in
animals. The exposure limit for 1,1,1-trichloroethane in most
countries is much higher than that for 1,1,2-trichloroethane,
indicating that the former is thought to be much less hazardous. It
is easy to see that only a small change in the structure of a chemical
may make it much more toxic (or less so). The lesson from this is
that numbers and letters are an important part of some chemical names,
so these should be carefully noted when checking a substance.
Common trade names -- Manufacturers often choose to give chemical
compounds, or mixtures of chemical compounds, commercial names.
This is usually done to make advertising and selling of the product
easier (many chemical names are very long or difficult to remember) or
to distinguish a product from that of a competing manufacturer. Often
trade names refer to the name of the manufacturer or to the use of the
product (e.g. Cellosolve was originally used to dissolve cellulose in
lacquers). A trade name may also be used because it conceals
knowledge of what is in a product or how it is made. However, this
should never be accepted as a reason for concealing the identity of
chemicals from those who work with them.
Common synonyms -- Synonyms are alternative names for the same
substance. The same chemical may have a number of different names
because of the many different chemical naming systems that have
developed, e.g. phosphorus trichloride is also known as phosphorus
chloride or trichlorophosphine.
Figure 3. Identity of chemical isomers
(from HSGs on butanol isomers)
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3-CH2-CH2-CH2OH
Primary constituent: 1-butanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3-CHOH-CH2-CH3
Primary constituent: 2-butanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3
\
CH-CH2OH
/
CH3
Primary constituent: isobutanol
1.1 Identity
Chemical formula: C4H10O
Chemical structure: CH3
|
CH3 -- C -- CH3
|
OH
Primary constituent: tert-butanol
CAS registry number -- This is a number assigned to every chemical
by the Chemical Abstracts Service, a section of the American
Chemical Society, which publishes guides to chemical research. Each
chemical is given a unique number.
The CAS number does not give any information about the properties of
the chemical itself. Its value is in overcoming the confusion caused
by the different chemical naming systems (see "Common synonyms"
above). The CAS number will often be given in product information or
literature that accompanies chemicals.
RTECS number -- RTECS stands for Registry of Toxic Effects of
Chemical Substances. This is a list of scientific articles on the
health effects of the chemicals. The Registry is operated by the
National Institute for Occupational Safety and Health, a government
body in the United States. Each chemical that is listed is given a
unique number. About 25 000 chemicals commonly used in industry are
listed.
Other numbering systems and nomenclatures are also used for
classifying chemicals. Those used in the HSGs include the IUPAC
(International Union for Pure and Applied Chemistry) name, EC numbers
assigned by the European Community and UN numbers for the transport of
dangerous goods.
Conversion factor -- The concentration or amount of chemical present
in air, water or food can be expressed in a number of different units.
The conversion factor allows these measurements to be converted from
one to another: "ppm" stands for parts of the chemical per million
parts of air and thus is a ratio of volumes; "mg/m3" stands for
milligrams of the contaminant per cubic metre of air; "kPa" stands for
kilopascals, a unit of pressure; and "mmHg" means millimetres of
mercury (760 mmHg is the normal atmospheric pressure at sea level).
1.2 Physical and chemical properties
1.2.1 Physical state
This section of the HSG includes a description of the physical form of
the chemical.
Chemicals may be gases, liquids or solids. They can change
between these "states" depending on temperature and pressure. For
instance, water is a liquid within the temperature range 0-100 degrees
Celsius (°C). Below 0°C, water is in a solid state (ice), and above
100°C it is in a gaseous state (steam) (see Fig. 4). When the
temperature of a solid is increased, it generally turns into a liquid,
i.e. it melts. If the liquid is heated further, it will turn into a
gas or vapour and boil or evaporate. If pressure is increased without
a change in the temperature, gases may convert from the gaseous to a
liquid state.
Some chemicals can exist in only two states, passing from solid to gas
and vice versa without going through a liquid phase.
1.2.2 Smell and odour threshold
The smell of a chemical can sometimes serve as a warning of exposure.
However, it must always be remembered that smell is not a
reliable means of warning, as many chemicals have no smell
at all.
 The "odour threshold" is the concentration at which humans can first
smell the substance in question. Yet many chemicals are dangerous at
concentrations well below the odour threshold. Moreover, sense of
smell varies greatly from person to person and can be affected by nose
and throat infections or head injuries. Finally, the sense of smell
can become "tired" (known as olfactory fatigue or olfactory
adaptation) if a person is exposed to the smell of a chemical for more
than a few minutes. If this happens, a large (and often dangerous)
increase in the amount of chemical present may be necessary before it
can again be smelt by those working with it. IT IS DANGEROUS TO
TRY TO IDENTIFY CHEMICALS BY SMELLING THEM.
1.2.3 Physical properties
The physical properties of each chemical are given in the
International Chemical Safety Card, or Summary of Chemical Safety
Information, contained in most HSGs (see Fig. 5). The terms used are
explained below. Further information can be obtained from the Card
Users' Guide, also produced by the IPCS to assist the user of the
ICSC.
Boiling point -- The temperature at which all the substance changes
from a liquid into a gas, e.g. water changes to steam at 100°C. It is
important to be aware of this temperature because when a chemical
substance is heated above the boiling-point, it will become a gas, and
it is much more difficult to protect against exposure to gases than to
protect against exposure to liquids or solids. Therefore, many
chemicals are more hazardous when heated. As mentioned below (see
Vapour pressure), some of the chemical will turn into a gas even at
temperatures below the boiling point. This can cause a dangerous
build-up of gas.
Melting point -- The temperature at which a substance changes from a
solid to a liquid, e.g. ice changes to water at 0°C.
Figure 5. Physical properties of a chemical (from HSG 9: Isobutanol)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Boiling point (°C) 108 Colourless liquid with
Melting point (°C) -108 characteristic odour;
Flash point (°C) 27 reacts with strong oxidants
Auto-ignition temperature (°C) 430 and alkali metals to form
Relative density (water = 1) 0.8 combustible gas (hydrogen);
Relative vapour density (air = 1) 2.6 attacks many plastics;
Vapour pressure in mbar (20°C) 12 substance may be absorbed
Solubility in water 95 into the body by inhalaltion
(g/litre at 20°C)
Explosive limits (vol. % in air) 1.2-10.9
Relative molecular mass 74.1
Flash point (open or closed cup) -- The flash point is the
temperature at which a substance gives off enough vapour to form an
ignitable mixture with air, i.e. one that burns when exposed to a
flame or spark. Open and closed cup refer to the way in which the
flash point is determined. In general, the open cup method gives a
better approximation of real-life situations. If the flash point
temperature is 20°C or lower, a spark can ignite the vapour, even at
normal room temperatures, and, in such cases, great care must be taken
to reduce the risk of accidental fire. The lower the flash point, the
greater the fire risk. Methods of reducing fire risk are discussed in
Chapter 4.
Auto-ignition temperature -- The lowest temperature at which a
substance will burn without being exposed to a flame or spark. The
closer this value is to room temperature, the greater the risk of
fire.
Relative density or specific gravity (at 20°C) -- The weight
of a specific volume of solid or liquid chemical substance compared to
the weight of the same volume of water. A substance with a specific
density greater than 1 will sink in water; if the specific density is
less than 1, the substance will float on water.
Relative vapour density -- The weight of a specific volume of a
gaseous chemical substance compared to the weight of the same volume
of air. This is also a very important safety consideration. If the
relative vapour density of a pure gas is less than 1, the gas will
rise and collect at ceiling level (if indoors) or disperse into the
atmosphere (if outdoors). On the other hand, if the relative vapour
density of a gas is greater than 1, the gas is heavier than air and
will sink and tend to collect at floor level or in depressions. Such
collection can result in unexpectedly high exposures to the substance,
can lead to a fire, or can represent an explosion hazard. In some
circumstances, gases that have a relative vapour density greater than
1 may completely replace air, including the oxygen that is essential
for life, leading to asphyxiation (suffocation). This often occurs in
tanks, pits or ship holds that are being cleaned or fumigated. This
situation can be avoided by ventilating areas in which gases or
vapours may collect. The vapours of most flammable liquids are
heavier than air. However, in workplace situations, the vapour
concentrations are generally at the level of a few hundred parts per
million (ppm), and the vapours will follow air streams and rise with
heat. In these situations, local exhaust should be as close to the
emissions as possible. Local exhaust near ground level is useful in
case large amounts of liquid leak or are spilled, which could lead to
noticeable vapour formation.
Vapour pressure (kPa) -- The pressure of the vapour produced by a
liquid or solid at room temperature. A small amount of the substance
is transformed from the solid or liquid state to the gaseous state
(vapour) at all temperatures -- evaporation (or sublimation if the
substance changes directly from a solid to a gaseous state without
going through a liquid phase). The higher the vapour pressure, the
more vapour will form above the liquid or solid, i.e. the faster it
tends to evaporate. The vapour pressure of a chemical rises as the
chemical is heated. This vapour or gas could be inhaled by workers.
Solubility in water (g/litre at 20°C) -- The amount (by weight)
of the substance that can be dissolved in 1 litre of water to form a
solution. The solubility in water may give some idea of what maximum
concentrations might occur in water bodies. A high solubility
indicates quick dissipation in surface water, with a potential for
causing acute toxic effects, e.g. to aquatic organisms in the case of
large chemical spills. For some compounds with low solubility,
aquatic organisms will not be so readily threatened by exposure to
potentially acute toxic concentrations.
Flammability (explosive) limits -- The range of concentrations
of a flammable gas or vapour in air at which it may explode in the
presence of a spark or flame. Below the lower limit (Lower Explosive
Limit, or LEL) there is not enough chemical to burn; above the upper
limit (Upper Explosive Limit, or UEL) the mixture is too rich,
i.e. too much chemical is present and not enough oxygen.
Relative molecular mass -- This is the relative weight of the
molecule of a chemical compared to the weight of one atom of hydrogen
(the lightest element).
Octanol/water partition coefficient -- This number indicates
how readily a chemical can dissolve in fats and oils. If the
octanol/water partition coefficient is high, e.g. for DDT it is about
6, then the chemical can accumulate more readily in body fat and can
be stored there for a very long time, as it may be excreted very
slowly. The octanol/water partition coefficient is therefore an
indicator of bioaccumulation. Chemicals that dissolve easily in
fats can usually be absorbed through the skin and may thereafter enter
the bloodstream. An example of a chemical that does not pass easily
through the skin is dimethyl sulfate (DMS), which has an octanol/water
partition coefficient of -4.26. However, DMS can burn the skin badly,
so a low octanol/water partition coefficient should not be seen as
evidence that a particular chemical will not have a negative health
effect if it comes into contact with the skin.
1.2.4 Composition
This section gives details of other substances that may be mixed with
the chemical in question. In some cases, these other chemicals may be
the unwanted result of the manufacturing process, in which case they
are called contaminants or impurities. In other cases, other
chemicals are added deliberately, e.g., to prevent undesirable or
unwanted chemical reactions, such as polymerization or oxidation.
It is important to know what other chemicals may be present in a
substance, as the combined health effects of the chemicals may be far
greater than would be expected by simple addition of the health
effects of the individual chemicals. This increased effect of two or
more chemicals is known as "synergism" and is discussed further in
Chapter 2 of this Manual.
1.3 Analytical methods
This section deals with methods for determining the concentration of
the chemical or its degradation products in the environment and in
foodstuffs (see Fig. 6).
Figure 6. Analytical methods (from HSG 34: Fenvalerate)
1.3 Analytical Methods
For residue and environmental analysis, gas chromatography with
electron-capture detection is used, the minimum detection level being
0.005 mg/kg. For product analysis, gas chromatography with flame
ionization detection can be used.
1.4 Production and uses
The HSGs list the common uses for chemical substances. Many chemicals
are used in a large variety of products and processes (Fig. 7). For
many chemicals it is not possible to list all possible uses. A
careful check should always be made of the chemical that will be used
for a particular type of work or task.
Figure 7. Uses of methylene chloride
(from HSG 6: Methylene Chloride)
1.4 Uses
Methylene chloride is widely used as a solvent and paint remover.
It is also used as a blowing agent for polyurethane, as a propellant
in aerosols such as insecticides, hair sprays, shampoos, and paints,
as a component in fire-extinguishing products, as an insecticidal
fumigant for grains, and as a coolant or refrigerant.
Many HSGs are concerned with the use of chemicals as pesticides and
the associated terminology may need some explaining.
The word pesticide is used as a general term for any substance, or
mixture of substances, intended for controlling the presence of
unwanted plants, animals, insects, etc. As much as possible,
pesticides are designed to control only target species of pests; their
general names then denote the type of pesticide, e.g.:
* acaricides --> spiders and ticks;
* fungicides --> fungi;
* herbicides --> weeds;
* insecticides --> insects;
* larvicides --> larvae of insects or other organisms;
* miticides --> mites;
* molluscicides --> snails; and
* rodenticides --> rats, mice and other rodents.
Pesticides kill or control pests by interfering with one or more of
their essential body systems. Most pesticides belong to a few
chemical groups, each of which has its own effect on certain systems,
e.g. organophosphorus compounds affect nerve function by inhibiting
the action of cholinesterase, an essential chemical for the passage of
impulses along nerves. Small changes in the chemical structure of
compounds in a group can result in some species being affected more by
those compounds than by others in the same group. This is why some
chemicals are more selective than others in their action on certain
pests. Pesticides that tend to kill a variety of organisms, including
both pests and others, are known as broad-spectrum pesticides.
Pesticides may act on target species in several ways: e.g. a pesticide
that kills pests by first passing through the skin or outer layer is a
contact poison; one that kills the pest by first passing into the
stomach is a stomach poison. A pesticide that kills a pest as it
flies through the air is a fumigant. Fumigants usually apply
strictly to pesticides in the form of a gas or vapour, but sometimes
when droplets or an aerosol of a pesticide acts on flying pests (i.e.
essentially a contact poison), the pesticide is said to have a
fumigant action.
A systemic pesticide is one that can be readily absorbed and
transported in plant tissue and, without affecting the plant, can
exert its action on pests feeding on the plant. It is usually applied
to the leaves or the soil around the plant so that it can pass through
the roots. Systemic herbicides are designed to kill the plant
itself if it is a weed.
Pesticides are available as technological products, i.e. the
pesticide chemical plus impurities that may be associated with its
manufacture (as opposed to purer laboratory versions). The pesticide
chemical is also known as the active ingredient. The active
ingredient is usually mixed with other chemicals to facilitate its
use. It is then known as a formulation. Some formulations may need
further dilution.
When a formulation is designed mainly to facilitate use and increase
the effectiveness of the active ingredient, it may be modified to
increase the safety of handling the pesticide. Some examples include:
* soluble powders or granules to be added to water;
* dusts or dustable powders applied as such;
* pellets or paste used as baits;
* tablets to generate smoke, gas or vapour;
* emulsifiable concentrates to be added to water;
* aerosol generators;
* pour-ons for direct application to the skin of animals; and
* shampoos for humans.
2. EXPOSURE, TOXICITY AND RISK - CHAPTER 2
This chapter should be read in conjunction with Chapter 2 (Summary and
Evaluation) of the relevant HSG (see Fig. 8).
Figure 8. Summary and evaluation (from HSG 7: tert-Butanol)
SUMMARY AND EVALUATION
2
Page
2.1 Exposure to tert-butanol 13
2.2 Uptake, metabolism, and excretion 13
2.3 Effects on organisms in the environment 13
2.4 Effects on animals 13
2.5 Effects on human beings 14
2.1 Exposure
Chemicals are found everywhere, not only in the workplace but also in
the general environment -- in the air, water and soil. The heaviest
exposures to some chemicals often occur during industrial or
agricultural activities. But significant exposure can also occur
through contact with naturally occurring ores and the surrounding
soil, from vehicle exhaust emissions, from building and insulating
materials and from various foods. (Food itself may be contaminated or
simply a medium for naturally occurring chemicals.)
Chemicals are found everywhere in the environment; many different
organisms are exposed to them -- whether they are plants exposed, for
instance, to arsenic in the soil around arsenic mines or smelter
wastes, microorganisms such as bacteria and fungi in soil, algae in
water, or fish or birds exposed to pesticides. Different species will
respond in different ways, even to similar doses of the same chemical.
What must be remembered, however, is that for a chemical to exert an
effect, there must first be an exposure. Not even the most toxic
chemicals will cause harm to an organism, including humans, if there
is no exposure. The different ways in which humans can be exposed to
chemicals are described below. Environmental exposures and the
effects of chemicals on the environment are discussed in Chapter 5,
"How Chemicals Can Poison the Environment".
There are four main exposure routes, or ways in which chemicals can
enter the body (see Fig. 9):
* inhalation (breathing in);
* absorption (through the skin or eye);
* ingestion (through the digestive system by swallowing chemicals
or eating contaminated food or drink); and
* transplacental transfer (across the placenta of the pregnant
woman to the fetus).
Most chemicals found in the workplace have the potential to be
dispersed into the air as dust, in droplets (as mist, i.e. an
aerosol) or as gas or vapour, and then inhaled. People who handle
chemicals directly are at risk of absorption via the skin or eyes.
Thus, the most important routes of exposure in the workplace leading
to systemic effects are inhalation and skin absorption (see Fig. 10).
Everyone present in a workplace is also potentially at risk of
exposure to chemicals through ingestion of contaminated food or drink.
Contaminated cigarettes are also a potential source of exposure via
ingestion.
In addition, a fetus may be exposed through transplacental transfer of
chemicals that are in the pregnant woman's bloodstream.
In the case of each exposure route, chemicals can enter the
bloodstream and thereafter be distributed to any or all of the organs
and tissues of the body. In this way, they can attack and harm organs
that are distant from the original point of entry, as well as cause
damage at the point of entry.
Information on methods of preventing exposure to chemicals in the
workplace and pollution of the environment is discussed in Chapters 3
and 4.
The "odour threshold" is the concentration at which humans can first
smell the substance in question. Yet many chemicals are dangerous at
concentrations well below the odour threshold. Moreover, sense of
smell varies greatly from person to person and can be affected by nose
and throat infections or head injuries. Finally, the sense of smell
can become "tired" (known as olfactory fatigue or olfactory
adaptation) if a person is exposed to the smell of a chemical for more
than a few minutes. If this happens, a large (and often dangerous)
increase in the amount of chemical present may be necessary before it
can again be smelt by those working with it. IT IS DANGEROUS TO
TRY TO IDENTIFY CHEMICALS BY SMELLING THEM.
1.2.3 Physical properties
The physical properties of each chemical are given in the
International Chemical Safety Card, or Summary of Chemical Safety
Information, contained in most HSGs (see Fig. 5). The terms used are
explained below. Further information can be obtained from the Card
Users' Guide, also produced by the IPCS to assist the user of the
ICSC.
Boiling point -- The temperature at which all the substance changes
from a liquid into a gas, e.g. water changes to steam at 100°C. It is
important to be aware of this temperature because when a chemical
substance is heated above the boiling-point, it will become a gas, and
it is much more difficult to protect against exposure to gases than to
protect against exposure to liquids or solids. Therefore, many
chemicals are more hazardous when heated. As mentioned below (see
Vapour pressure), some of the chemical will turn into a gas even at
temperatures below the boiling point. This can cause a dangerous
build-up of gas.
Melting point -- The temperature at which a substance changes from a
solid to a liquid, e.g. ice changes to water at 0°C.
Figure 5. Physical properties of a chemical (from HSG 9: Isobutanol)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Boiling point (°C) 108 Colourless liquid with
Melting point (°C) -108 characteristic odour;
Flash point (°C) 27 reacts with strong oxidants
Auto-ignition temperature (°C) 430 and alkali metals to form
Relative density (water = 1) 0.8 combustible gas (hydrogen);
Relative vapour density (air = 1) 2.6 attacks many plastics;
Vapour pressure in mbar (20°C) 12 substance may be absorbed
Solubility in water 95 into the body by inhalaltion
(g/litre at 20°C)
Explosive limits (vol. % in air) 1.2-10.9
Relative molecular mass 74.1
Flash point (open or closed cup) -- The flash point is the
temperature at which a substance gives off enough vapour to form an
ignitable mixture with air, i.e. one that burns when exposed to a
flame or spark. Open and closed cup refer to the way in which the
flash point is determined. In general, the open cup method gives a
better approximation of real-life situations. If the flash point
temperature is 20°C or lower, a spark can ignite the vapour, even at
normal room temperatures, and, in such cases, great care must be taken
to reduce the risk of accidental fire. The lower the flash point, the
greater the fire risk. Methods of reducing fire risk are discussed in
Chapter 4.
Auto-ignition temperature -- The lowest temperature at which a
substance will burn without being exposed to a flame or spark. The
closer this value is to room temperature, the greater the risk of
fire.
Relative density or specific gravity (at 20°C) -- The weight
of a specific volume of solid or liquid chemical substance compared to
the weight of the same volume of water. A substance with a specific
density greater than 1 will sink in water; if the specific density is
less than 1, the substance will float on water.
Relative vapour density -- The weight of a specific volume of a
gaseous chemical substance compared to the weight of the same volume
of air. This is also a very important safety consideration. If the
relative vapour density of a pure gas is less than 1, the gas will
rise and collect at ceiling level (if indoors) or disperse into the
atmosphere (if outdoors). On the other hand, if the relative vapour
density of a gas is greater than 1, the gas is heavier than air and
will sink and tend to collect at floor level or in depressions. Such
collection can result in unexpectedly high exposures to the substance,
can lead to a fire, or can represent an explosion hazard. In some
circumstances, gases that have a relative vapour density greater than
1 may completely replace air, including the oxygen that is essential
for life, leading to asphyxiation (suffocation). This often occurs in
tanks, pits or ship holds that are being cleaned or fumigated. This
situation can be avoided by ventilating areas in which gases or
vapours may collect. The vapours of most flammable liquids are
heavier than air. However, in workplace situations, the vapour
concentrations are generally at the level of a few hundred parts per
million (ppm), and the vapours will follow air streams and rise with
heat. In these situations, local exhaust should be as close to the
emissions as possible. Local exhaust near ground level is useful in
case large amounts of liquid leak or are spilled, which could lead to
noticeable vapour formation.
Vapour pressure (kPa) -- The pressure of the vapour produced by a
liquid or solid at room temperature. A small amount of the substance
is transformed from the solid or liquid state to the gaseous state
(vapour) at all temperatures -- evaporation (or sublimation if the
substance changes directly from a solid to a gaseous state without
going through a liquid phase). The higher the vapour pressure, the
more vapour will form above the liquid or solid, i.e. the faster it
tends to evaporate. The vapour pressure of a chemical rises as the
chemical is heated. This vapour or gas could be inhaled by workers.
Solubility in water (g/litre at 20°C) -- The amount (by weight)
of the substance that can be dissolved in 1 litre of water to form a
solution. The solubility in water may give some idea of what maximum
concentrations might occur in water bodies. A high solubility
indicates quick dissipation in surface water, with a potential for
causing acute toxic effects, e.g. to aquatic organisms in the case of
large chemical spills. For some compounds with low solubility,
aquatic organisms will not be so readily threatened by exposure to
potentially acute toxic concentrations.
Flammability (explosive) limits -- The range of concentrations
of a flammable gas or vapour in air at which it may explode in the
presence of a spark or flame. Below the lower limit (Lower Explosive
Limit, or LEL) there is not enough chemical to burn; above the upper
limit (Upper Explosive Limit, or UEL) the mixture is too rich,
i.e. too much chemical is present and not enough oxygen.
Relative molecular mass -- This is the relative weight of the
molecule of a chemical compared to the weight of one atom of hydrogen
(the lightest element).
Octanol/water partition coefficient -- This number indicates
how readily a chemical can dissolve in fats and oils. If the
octanol/water partition coefficient is high, e.g. for DDT it is about
6, then the chemical can accumulate more readily in body fat and can
be stored there for a very long time, as it may be excreted very
slowly. The octanol/water partition coefficient is therefore an
indicator of bioaccumulation. Chemicals that dissolve easily in
fats can usually be absorbed through the skin and may thereafter enter
the bloodstream. An example of a chemical that does not pass easily
through the skin is dimethyl sulfate (DMS), which has an octanol/water
partition coefficient of -4.26. However, DMS can burn the skin badly,
so a low octanol/water partition coefficient should not be seen as
evidence that a particular chemical will not have a negative health
effect if it comes into contact with the skin.
1.2.4 Composition
This section gives details of other substances that may be mixed with
the chemical in question. In some cases, these other chemicals may be
the unwanted result of the manufacturing process, in which case they
are called contaminants or impurities. In other cases, other
chemicals are added deliberately, e.g., to prevent undesirable or
unwanted chemical reactions, such as polymerization or oxidation.
It is important to know what other chemicals may be present in a
substance, as the combined health effects of the chemicals may be far
greater than would be expected by simple addition of the health
effects of the individual chemicals. This increased effect of two or
more chemicals is known as "synergism" and is discussed further in
Chapter 2 of this Manual.
1.3 Analytical methods
This section deals with methods for determining the concentration of
the chemical or its degradation products in the environment and in
foodstuffs (see Fig. 6).
Figure 6. Analytical methods (from HSG 34: Fenvalerate)
1.3 Analytical Methods
For residue and environmental analysis, gas chromatography with
electron-capture detection is used, the minimum detection level being
0.005 mg/kg. For product analysis, gas chromatography with flame
ionization detection can be used.
1.4 Production and uses
The HSGs list the common uses for chemical substances. Many chemicals
are used in a large variety of products and processes (Fig. 7). For
many chemicals it is not possible to list all possible uses. A
careful check should always be made of the chemical that will be used
for a particular type of work or task.
Figure 7. Uses of methylene chloride
(from HSG 6: Methylene Chloride)
1.4 Uses
Methylene chloride is widely used as a solvent and paint remover.
It is also used as a blowing agent for polyurethane, as a propellant
in aerosols such as insecticides, hair sprays, shampoos, and paints,
as a component in fire-extinguishing products, as an insecticidal
fumigant for grains, and as a coolant or refrigerant.
Many HSGs are concerned with the use of chemicals as pesticides and
the associated terminology may need some explaining.
The word pesticide is used as a general term for any substance, or
mixture of substances, intended for controlling the presence of
unwanted plants, animals, insects, etc. As much as possible,
pesticides are designed to control only target species of pests; their
general names then denote the type of pesticide, e.g.:
* acaricides --> spiders and ticks;
* fungicides --> fungi;
* herbicides --> weeds;
* insecticides --> insects;
* larvicides --> larvae of insects or other organisms;
* miticides --> mites;
* molluscicides --> snails; and
* rodenticides --> rats, mice and other rodents.
Pesticides kill or control pests by interfering with one or more of
their essential body systems. Most pesticides belong to a few
chemical groups, each of which has its own effect on certain systems,
e.g. organophosphorus compounds affect nerve function by inhibiting
the action of cholinesterase, an essential chemical for the passage of
impulses along nerves. Small changes in the chemical structure of
compounds in a group can result in some species being affected more by
those compounds than by others in the same group. This is why some
chemicals are more selective than others in their action on certain
pests. Pesticides that tend to kill a variety of organisms, including
both pests and others, are known as broad-spectrum pesticides.
Pesticides may act on target species in several ways: e.g. a pesticide
that kills pests by first passing through the skin or outer layer is a
contact poison; one that kills the pest by first passing into the
stomach is a stomach poison. A pesticide that kills a pest as it
flies through the air is a fumigant. Fumigants usually apply
strictly to pesticides in the form of a gas or vapour, but sometimes
when droplets or an aerosol of a pesticide acts on flying pests (i.e.
essentially a contact poison), the pesticide is said to have a
fumigant action.
A systemic pesticide is one that can be readily absorbed and
transported in plant tissue and, without affecting the plant, can
exert its action on pests feeding on the plant. It is usually applied
to the leaves or the soil around the plant so that it can pass through
the roots. Systemic herbicides are designed to kill the plant
itself if it is a weed.
Pesticides are available as technological products, i.e. the
pesticide chemical plus impurities that may be associated with its
manufacture (as opposed to purer laboratory versions). The pesticide
chemical is also known as the active ingredient. The active
ingredient is usually mixed with other chemicals to facilitate its
use. It is then known as a formulation. Some formulations may need
further dilution.
When a formulation is designed mainly to facilitate use and increase
the effectiveness of the active ingredient, it may be modified to
increase the safety of handling the pesticide. Some examples include:
* soluble powders or granules to be added to water;
* dusts or dustable powders applied as such;
* pellets or paste used as baits;
* tablets to generate smoke, gas or vapour;
* emulsifiable concentrates to be added to water;
* aerosol generators;
* pour-ons for direct application to the skin of animals; and
* shampoos for humans.
2. EXPOSURE, TOXICITY AND RISK - CHAPTER 2
This chapter should be read in conjunction with Chapter 2 (Summary and
Evaluation) of the relevant HSG (see Fig. 8).
Figure 8. Summary and evaluation (from HSG 7: tert-Butanol)
SUMMARY AND EVALUATION
2
Page
2.1 Exposure to tert-butanol 13
2.2 Uptake, metabolism, and excretion 13
2.3 Effects on organisms in the environment 13
2.4 Effects on animals 13
2.5 Effects on human beings 14
2.1 Exposure
Chemicals are found everywhere, not only in the workplace but also in
the general environment -- in the air, water and soil. The heaviest
exposures to some chemicals often occur during industrial or
agricultural activities. But significant exposure can also occur
through contact with naturally occurring ores and the surrounding
soil, from vehicle exhaust emissions, from building and insulating
materials and from various foods. (Food itself may be contaminated or
simply a medium for naturally occurring chemicals.)
Chemicals are found everywhere in the environment; many different
organisms are exposed to them -- whether they are plants exposed, for
instance, to arsenic in the soil around arsenic mines or smelter
wastes, microorganisms such as bacteria and fungi in soil, algae in
water, or fish or birds exposed to pesticides. Different species will
respond in different ways, even to similar doses of the same chemical.
What must be remembered, however, is that for a chemical to exert an
effect, there must first be an exposure. Not even the most toxic
chemicals will cause harm to an organism, including humans, if there
is no exposure. The different ways in which humans can be exposed to
chemicals are described below. Environmental exposures and the
effects of chemicals on the environment are discussed in Chapter 5,
"How Chemicals Can Poison the Environment".
There are four main exposure routes, or ways in which chemicals can
enter the body (see Fig. 9):
* inhalation (breathing in);
* absorption (through the skin or eye);
* ingestion (through the digestive system by swallowing chemicals
or eating contaminated food or drink); and
* transplacental transfer (across the placenta of the pregnant
woman to the fetus).
Most chemicals found in the workplace have the potential to be
dispersed into the air as dust, in droplets (as mist, i.e. an
aerosol) or as gas or vapour, and then inhaled. People who handle
chemicals directly are at risk of absorption via the skin or eyes.
Thus, the most important routes of exposure in the workplace leading
to systemic effects are inhalation and skin absorption (see Fig. 10).
Everyone present in a workplace is also potentially at risk of
exposure to chemicals through ingestion of contaminated food or drink.
Contaminated cigarettes are also a potential source of exposure via
ingestion.
In addition, a fetus may be exposed through transplacental transfer of
chemicals that are in the pregnant woman's bloodstream.
In the case of each exposure route, chemicals can enter the
bloodstream and thereafter be distributed to any or all of the organs
and tissues of the body. In this way, they can attack and harm organs
that are distant from the original point of entry, as well as cause
damage at the point of entry.
Information on methods of preventing exposure to chemicals in the
workplace and pollution of the environment is discussed in Chapters 3
and 4.
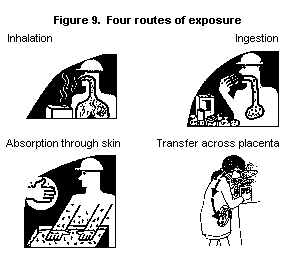 Figure 10. Occupational exposure
(from HSG 19: Pentachlorophenol)
Occupational exposure to technical PCP occurs mainly through
inhalation and skin contact. Virtually all workers exposed to
airborne concentrations take up PCP via the lungs and the skin. In
addition, workers handling treated timber or maintaining PCP-
contaminated equipment may be exposed to PCP in solution, and
may absorb from one-half (based on urinary-PCP concentrations) to
two-thirds (using serum levels) of their total PCP burden through
the skin.
2.1.1 Inhalation
2.1.1.1 Forms
* Gases and vapours -- Workplace chemicals can be emitted into the
atmosphere in a number of different ways. Evaporation is
probably the most common. Organic solvents, such as toluene,
MEK (methylethylketone) or alcohols, generally evaporate more
rapidly than water, acids or caustics. Evaporation of a
liquid produces vapours. Vapours are formed from substances that
exist as solids or liquids under normal temperature and pressure
conditions. Substances that do not exist as solids or liquids at
normal room temperatures and pressures are called gases. Gases,
as well as vapours, can contaminate the workplace air.
Chemicals with a high vapour pressure will generate more vapour
than chemicals with a low vapour pressure. The amount of vapour
released by a chemical also increases with increasing
temperature.
* Mists and aerosols -- Some industrial processes produce tiny
liquid droplets that are able to float in air. Alternatively,
mists sometimes form when a liquid breaks up, is splashed or is
atomized. Examples include acid mists from electroplating, oil
mists from cutting or grinding and paint spray mists from
painting operations.
* Dusts, fumes and smoke -- Other workplace processes can generate
tiny solid particles that are light enough to float in air; these
are referred to as dusts, fumes or smoke. Dusts are solid
particles, often generated by some mechanical or abrasive
activity. They are usually of such a weight that they settle
slowly to the ground. Fumes are solid particles formed when a
heated substance has evaporated in the air and then condensed
back to a solid form (often after combining with oxygen in the
air to form an oxide). This often occurs during welding
operations. Fume particles are extremely small and may remain
airborne indefinitely. Smoke consists of carbon or soot and is
formed during combustion. Smoke particles can settle or remain
airborne, depending on their size.
2.1.1.2 Entry into the body
Contaminated air in the workplace is inhaled through the mouth and
nose and then into the lungs (see Fig. 11). An average person will
breathe in and out about 12 times per minute. Each inhalation brings
about 500 ml of air into the lungs (corresponding to 6 litres of air
per minute at rest), together with any contaminants that this air
contains. People undertaking hard physical work will breathe harder
and take in more than 6 litres per minute. Over an eight-hour working
day, more than 2800 litres of air will pass through the lungs. In
conditions of hard physical work, up to 10 000 litres may be exchanged
during the course of an eight-hour workshift.
Figure 10. Occupational exposure
(from HSG 19: Pentachlorophenol)
Occupational exposure to technical PCP occurs mainly through
inhalation and skin contact. Virtually all workers exposed to
airborne concentrations take up PCP via the lungs and the skin. In
addition, workers handling treated timber or maintaining PCP-
contaminated equipment may be exposed to PCP in solution, and
may absorb from one-half (based on urinary-PCP concentrations) to
two-thirds (using serum levels) of their total PCP burden through
the skin.
2.1.1 Inhalation
2.1.1.1 Forms
* Gases and vapours -- Workplace chemicals can be emitted into the
atmosphere in a number of different ways. Evaporation is
probably the most common. Organic solvents, such as toluene,
MEK (methylethylketone) or alcohols, generally evaporate more
rapidly than water, acids or caustics. Evaporation of a
liquid produces vapours. Vapours are formed from substances that
exist as solids or liquids under normal temperature and pressure
conditions. Substances that do not exist as solids or liquids at
normal room temperatures and pressures are called gases. Gases,
as well as vapours, can contaminate the workplace air.
Chemicals with a high vapour pressure will generate more vapour
than chemicals with a low vapour pressure. The amount of vapour
released by a chemical also increases with increasing
temperature.
* Mists and aerosols -- Some industrial processes produce tiny
liquid droplets that are able to float in air. Alternatively,
mists sometimes form when a liquid breaks up, is splashed or is
atomized. Examples include acid mists from electroplating, oil
mists from cutting or grinding and paint spray mists from
painting operations.
* Dusts, fumes and smoke -- Other workplace processes can generate
tiny solid particles that are light enough to float in air; these
are referred to as dusts, fumes or smoke. Dusts are solid
particles, often generated by some mechanical or abrasive
activity. They are usually of such a weight that they settle
slowly to the ground. Fumes are solid particles formed when a
heated substance has evaporated in the air and then condensed
back to a solid form (often after combining with oxygen in the
air to form an oxide). This often occurs during welding
operations. Fume particles are extremely small and may remain
airborne indefinitely. Smoke consists of carbon or soot and is
formed during combustion. Smoke particles can settle or remain
airborne, depending on their size.
2.1.1.2 Entry into the body
Contaminated air in the workplace is inhaled through the mouth and
nose and then into the lungs (see Fig. 11). An average person will
breathe in and out about 12 times per minute. Each inhalation brings
about 500 ml of air into the lungs (corresponding to 6 litres of air
per minute at rest), together with any contaminants that this air
contains. People undertaking hard physical work will breathe harder
and take in more than 6 litres per minute. Over an eight-hour working
day, more than 2800 litres of air will pass through the lungs. In
conditions of hard physical work, up to 10 000 litres may be exchanged
during the course of an eight-hour workshift.
 Air breathed in through the nose is filtered by the nasal hairs;
large, solid particles in the atmosphere are therefore prevented from
passing any further into the body. Small bones and cartilage inside
the nose help the inhaled air to circulate. Large contaminating
particles may then be deposited in the nose, trapped by the moisture
of the mucous lining (see below).
Air inhaled through the nose and mouth reaches the back of the throat
and enters the pharynx. The pharynx, which is the entrance to the
airways, divides into two tubes, one called the oesophagus, which
carries food to the stomach, the other the trachea, which carries
air to the lungs. At the top of the trachea sits the larynx, where
the vocal cords are situated. Contaminated air passes into the
trachea, which itself divides into two large tubes, each called a
bronchus. Each bronchus enters a lung. Once inside the lung, each
bronchus starts to divide like the branches of a tree -- the branches
getting thinner and thinner as they spread. Eventually, the tiniest
tubes, which are called bronchioles, end in thin-walled air sacs,
each of which is called an alveolus. Collectively, they are called
alveoli, and there are hundreds of thousands of these in each lung.
The walls of the alveoli are very thin and are richly supplied with
tiny blood vessels (capillaries).
The most important function of the lung is to bring oxygen into the
body. Oxygen in inhaled breath crosses the alveolar walls and enters
the blood within the capillaries. Once oxygen has become absorbed
into the bloodstream, it is distributed throughout the body. Chemical
vapours, gases and mists that reach the alveoli in the lungs can also
pass into the blood and therefore be distributed around the body.
The airways contain a sticky, thick fluid called mucus. Tiny hairs,
known as cilia, on the inside of the tubes constantly carry the mucus
upwards towards the back of the throat from which it is either
expelled (spat out) through the mouth or swallowed and passed into the
stomach. Chemicals that are carried in air that has been inhaled can
in this way enter the stomach.
Solid particles that have reached the air sacs but which cannot pass
through the thin wall of the sacs may become lodged there. Some may
dissolve, others may be attacked and destroyed by scavenger cells
(macrophages) of the body's defence system. Some particles that
remain in the air sacs, if only in small quantities, do no apparent
harm. Other types of dust may damage the surrounding alveolar walls.
Such damage may be permanent and may result in the formation of scars,
which eventually interfere with the lung's ability to expand and pass
oxygen into the bloodstream.
Some acids, caustics or organic chemicals, when inhaled in large
amounts, can cause serious "burns" to the mouth, nose, trachea and
lungs.
Chemicals in the form of dust may settle on cigarettes or be
transferred to them from the hands. When a contaminated cigarette is
smoked, toxic chemical fumes will be inhaled. This may also occur if
cigarettes are smoked in areas where chemical vapours or gases are
present. For these reasons, cigarettes should be stored outside the
work area: no smoking should be allowed in a workplace where hazardous
chemicals are used.
2.1.2 Absorption through the skin
2.1.2.1 Forms
Chemicals that pass through the skin are nearly always in liquid form.
Dusts, gases or vapours do not generally pass through the skin unless
they are first dissolved in moisture on the skin's surface. Chemicals
that can dissolve easily in lipids (fats) will pass more readily
through the skin. The octanol/water partition coefficient (see
Chapter 1 of this Manual) can be used as a measure of how
lipid-soluble a chemical is.
There are some chemicals, however, that can pass through the skin via
other mechanisms. Some highly toxic compounds such as sarin and
parathion, for instance, penetrate the skin without causing overt
damage. Chemicals that cause local irritation or that are corrosive
may be absorbed in greater quantity owing to increased blood flow to
the skin or as a result of destruction of the outer skin barrier.
2.1.2.2 Entry into the body
The skin consists essentially of two layers: a thin, outermost layer
called the epidermis and a much thicker layer called the dermis (see
Fig. 12). The epidermis consists of several layers of flat, rather
tightly packed cells, which form a barrier against infections, water
and some chemicals. This barrier is the external part of the
epidermis. It is called the keratin layer and is largely
responsible for preventing water from escaping from the body. It can
also resist entry of water and weak acids but is much less resistant
to organic and some inorganic chemicals. The keratin layer contains
fat and fat-like substances, which readily absorb chemicals that are
solvents for fat, oil and grease.
Air breathed in through the nose is filtered by the nasal hairs;
large, solid particles in the atmosphere are therefore prevented from
passing any further into the body. Small bones and cartilage inside
the nose help the inhaled air to circulate. Large contaminating
particles may then be deposited in the nose, trapped by the moisture
of the mucous lining (see below).
Air inhaled through the nose and mouth reaches the back of the throat
and enters the pharynx. The pharynx, which is the entrance to the
airways, divides into two tubes, one called the oesophagus, which
carries food to the stomach, the other the trachea, which carries
air to the lungs. At the top of the trachea sits the larynx, where
the vocal cords are situated. Contaminated air passes into the
trachea, which itself divides into two large tubes, each called a
bronchus. Each bronchus enters a lung. Once inside the lung, each
bronchus starts to divide like the branches of a tree -- the branches
getting thinner and thinner as they spread. Eventually, the tiniest
tubes, which are called bronchioles, end in thin-walled air sacs,
each of which is called an alveolus. Collectively, they are called
alveoli, and there are hundreds of thousands of these in each lung.
The walls of the alveoli are very thin and are richly supplied with
tiny blood vessels (capillaries).
The most important function of the lung is to bring oxygen into the
body. Oxygen in inhaled breath crosses the alveolar walls and enters
the blood within the capillaries. Once oxygen has become absorbed
into the bloodstream, it is distributed throughout the body. Chemical
vapours, gases and mists that reach the alveoli in the lungs can also
pass into the blood and therefore be distributed around the body.
The airways contain a sticky, thick fluid called mucus. Tiny hairs,
known as cilia, on the inside of the tubes constantly carry the mucus
upwards towards the back of the throat from which it is either
expelled (spat out) through the mouth or swallowed and passed into the
stomach. Chemicals that are carried in air that has been inhaled can
in this way enter the stomach.
Solid particles that have reached the air sacs but which cannot pass
through the thin wall of the sacs may become lodged there. Some may
dissolve, others may be attacked and destroyed by scavenger cells
(macrophages) of the body's defence system. Some particles that
remain in the air sacs, if only in small quantities, do no apparent
harm. Other types of dust may damage the surrounding alveolar walls.
Such damage may be permanent and may result in the formation of scars,
which eventually interfere with the lung's ability to expand and pass
oxygen into the bloodstream.
Some acids, caustics or organic chemicals, when inhaled in large
amounts, can cause serious "burns" to the mouth, nose, trachea and
lungs.
Chemicals in the form of dust may settle on cigarettes or be
transferred to them from the hands. When a contaminated cigarette is
smoked, toxic chemical fumes will be inhaled. This may also occur if
cigarettes are smoked in areas where chemical vapours or gases are
present. For these reasons, cigarettes should be stored outside the
work area: no smoking should be allowed in a workplace where hazardous
chemicals are used.
2.1.2 Absorption through the skin
2.1.2.1 Forms
Chemicals that pass through the skin are nearly always in liquid form.
Dusts, gases or vapours do not generally pass through the skin unless
they are first dissolved in moisture on the skin's surface. Chemicals
that can dissolve easily in lipids (fats) will pass more readily
through the skin. The octanol/water partition coefficient (see
Chapter 1 of this Manual) can be used as a measure of how
lipid-soluble a chemical is.
There are some chemicals, however, that can pass through the skin via
other mechanisms. Some highly toxic compounds such as sarin and
parathion, for instance, penetrate the skin without causing overt
damage. Chemicals that cause local irritation or that are corrosive
may be absorbed in greater quantity owing to increased blood flow to
the skin or as a result of destruction of the outer skin barrier.
2.1.2.2 Entry into the body
The skin consists essentially of two layers: a thin, outermost layer
called the epidermis and a much thicker layer called the dermis (see
Fig. 12). The epidermis consists of several layers of flat, rather
tightly packed cells, which form a barrier against infections, water
and some chemicals. This barrier is the external part of the
epidermis. It is called the keratin layer and is largely
responsible for preventing water from escaping from the body. It can
also resist entry of water and weak acids but is much less resistant
to organic and some inorganic chemicals. The keratin layer contains
fat and fat-like substances, which readily absorb chemicals that are
solvents for fat, oil and grease.
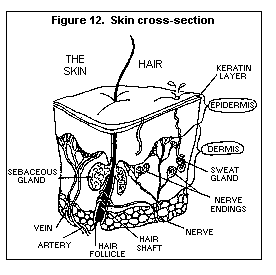 Organic and caustic (alkaline) chemicals can soften the keratin cells
and pass through this layer to the dermis, then enter the bloodstream.
Chemicals can also enter the body through cuts, punctures or scrapes
of the skin, as these constitute breaks in the protective layer.
Contact with some chemicals such as detergents or organic solvents can
cause dryness or cracking of the skin. There can also be hives, or
nettle-rash, ulcerations or skin flaking. All these conditions weaken
the protective layer of the skin, and chemicals are more likely to
enter the bloodstream.
Once in the bloodstream, the chemicals may be transported to any site
or organ of the body, where they may exert their effects.
Some chemicals are so corrosive that they may burn holes in the skin,
through which infection or entry of other chemicals can occur.
2.1.3 Absorption through the eye
2.1.3.1 Forms
Any chemical, in the form of a liquid, dust, vapour, gas, aerosol or
mist can enter the eye.
2.1.3.2 Entry into the body
Eye splashes or eye contamination due to exposure to workplace
chemicals is common. Small amounts of chemicals can enter the eye by
dissolving in the liquid surrounding the eye. The eyes are richly
supplied with blood vessels, and many chemicals can penetrate the
outer tissues and pass into the veins. The eye may be damaged during
this process, depending on the corrosive nature of the chemical and
its ability to penetrate the outer tissues. For example, the organic
solvent toluene can pass through the outer layers of the eye and
probably enter the blood. It can cause keratitis, which is an
inflammation of the outer layer of the eye (see Fig. 13). The
majority of substances that become dissolved in tears are passed to
the nose and eventually swallowed (see Fig. 14). Only in very rare
cases does absorption of chemicals through the eye cause acute
systemic effects.
2.1.4 Ingestion
2.1.4.1 Forms
All forms of chemicals, whether they exist as liquids, solids, gases,
vapours, mists, dusts, smoke or fumes, can -- directly or indirectly
-- enter the digestive system.
Organic and caustic (alkaline) chemicals can soften the keratin cells
and pass through this layer to the dermis, then enter the bloodstream.
Chemicals can also enter the body through cuts, punctures or scrapes
of the skin, as these constitute breaks in the protective layer.
Contact with some chemicals such as detergents or organic solvents can
cause dryness or cracking of the skin. There can also be hives, or
nettle-rash, ulcerations or skin flaking. All these conditions weaken
the protective layer of the skin, and chemicals are more likely to
enter the bloodstream.
Once in the bloodstream, the chemicals may be transported to any site
or organ of the body, where they may exert their effects.
Some chemicals are so corrosive that they may burn holes in the skin,
through which infection or entry of other chemicals can occur.
2.1.3 Absorption through the eye
2.1.3.1 Forms
Any chemical, in the form of a liquid, dust, vapour, gas, aerosol or
mist can enter the eye.
2.1.3.2 Entry into the body
Eye splashes or eye contamination due to exposure to workplace
chemicals is common. Small amounts of chemicals can enter the eye by
dissolving in the liquid surrounding the eye. The eyes are richly
supplied with blood vessels, and many chemicals can penetrate the
outer tissues and pass into the veins. The eye may be damaged during
this process, depending on the corrosive nature of the chemical and
its ability to penetrate the outer tissues. For example, the organic
solvent toluene can pass through the outer layers of the eye and
probably enter the blood. It can cause keratitis, which is an
inflammation of the outer layer of the eye (see Fig. 13). The
majority of substances that become dissolved in tears are passed to
the nose and eventually swallowed (see Fig. 14). Only in very rare
cases does absorption of chemicals through the eye cause acute
systemic effects.
2.1.4 Ingestion
2.1.4.1 Forms
All forms of chemicals, whether they exist as liquids, solids, gases,
vapours, mists, dusts, smoke or fumes, can -- directly or indirectly
-- enter the digestive system.
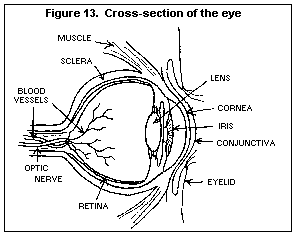
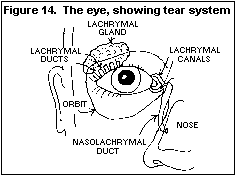 2.1.4.2 Entry into the body
Chemicals can enter the stomach following ingestion of contaminated
food or mouth contact with contaminated cigarettes (see Fig. 15).
Food and drink can become contaminated when they are produced and also
at the time of consumption by contact with unwashed hands, gloves or
clothing, or if they are left exposed in the workplace. Nail-biting
is another potential source of ingested chemicals. Some of the
chemicals that are inhaled will be trapped in the mucus of the lungs,
much of which will eventually be swallowed, thereby contributing to
the total amount of the ingested chemicals. In addition, chemicals in
liquid form may enter the body by accidental ingestion. For example,
if a liquid is transferred to a bottle and not labelled adequately, a
worker may mistake it for water or another beverage.
2.1.4.2 Entry into the body
Chemicals can enter the stomach following ingestion of contaminated
food or mouth contact with contaminated cigarettes (see Fig. 15).
Food and drink can become contaminated when they are produced and also
at the time of consumption by contact with unwashed hands, gloves or
clothing, or if they are left exposed in the workplace. Nail-biting
is another potential source of ingested chemicals. Some of the
chemicals that are inhaled will be trapped in the mucus of the lungs,
much of which will eventually be swallowed, thereby contributing to
the total amount of the ingested chemicals. In addition, chemicals in
liquid form may enter the body by accidental ingestion. For example,
if a liquid is transferred to a bottle and not labelled adequately, a
worker may mistake it for water or another beverage.
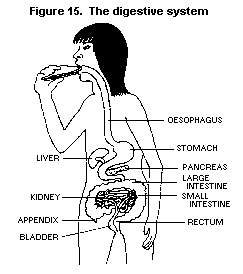 Once inside the mouth, chemicals pass down the oesophagus and into the
stomach. Food is digested with a strong acid produced by the stomach.
A few chemicals, such as alcohol, may pass across the stomach wall and
enter the veins and bloodstream there. But most chemicals move from
the stomach into the small intestine. The inside of the small
intestine has hundreds of tiny finger-like projections called villi
that have very thin walls and are filled with tiny blood vessels.
This allows the digested food to pass from the small intestine across
the walls of the villi and into the bloodstream.
Some chemicals that contaminate food or drink can also pass across the
thin walls of the villi and become absorbed into the bloodstream.
Other chemicals that are not soluble, or the basic units (molecules)
of which are too big to pass across the villi walls, will stay in the
gut and pass out through the faeces without being absorbed into the
bloodstream to a great extent. The absorption rate of a chemical
indicates the percentage of the amount swallowed that becomes absorbed
into the bloodstream. Most chemicals are only partly absorbed.
Some acids, caustics and organic chemicals may cause severe "burn"
damage to the digestive system if ingested in high concentrations.
2.2 Processing of chemicals in the body
After a chemical enters the body, it undergoes one or more of three
processes. The chemical may be metabolized by the body, stored or
accumulated in the body and/or excreted.
2.2.1 Metabolism
Metabolism is the process by which the body renders a 'foreign'
chemical more easily excretable and less toxic. The body is full of
special proteins called enzymes which initiate particular chemical
reactions. These reactions are necessary to, among other things,
convert the food we eat into a form that can be used by the tissues
and, combined with oxygen carried in the blood, to create heat and
energy. Some enzymes render foreign chemicals less toxic. They do
this by breaking down a chemical compound into simpler chemicals or by
converting a foreign chemical into a form that the body can excrete
more easily. Once a foreign chemical has been metabolized, it becomes
a different chemical, known as a metabolite. The process of
metabolism may involve many steps, and the result is many different
metabolites.
Metabolism may occur anywhere or everywhere in the body, or in just
one organ or type of tissue. Generally, this process renders a
chemical less toxic. However, some metabolites are as toxic as or
even more toxic than the original chemical. Such metabolites are
often called reactive metabolites. For most chemicals, the liver is
the main site of metabolism, but other organs such as the kidneys and
testes are also capable of metabolizing chemicals, the products of
which may be toxic.
2.2.2 Excretion
Excretion is the process by which unwanted chemicals are removed from
the body. Both foreign chemicals and those produced naturally by the
body are excreted. Most excretion occurs through the kidneys. Some
chemicals are excreted via the bile that passes from the liver into
the intestines, after which the chemicals pass out of the body in the
faeces. Other chemicals are excreted as a gas that is exhaled from
the lungs; small amounts may also be excreted in sweat.
In the kidneys, blood (which may be carrying waste or foreign
chemicals) is filtered through a set of small, twisting blood vessels
called the glomeruli. The fluid that is filtered off then passes
through tiny tubes called tubules. Here, water is reabsorbed and the
level of various salts adjusted. The combination of a glomerulus and
a tubule is called a nephron. There are hundreds of thousands of
nephrons in each kidney. The fluid that each nephron produces flows
through more tubes (the collecting ducts and the ureters) into the
bladder, from which urine is expelled from the body (see Fig. 16).
For some chemicals, it is possible to calculate the amount absorbed by
testing a urine sample. The concentration of the original chemical or
the concentration of a metabolite in the urine can also be determined.
Urine tests are often useful for determining whether absorption of a
chemical has occurred. For some chemicals, such as alcohol, exhaled
air can be used as an indication of absorption. Testing of urine,
blood or other body fluids to determine absorption is discussed
further in Chapter 3 of this Manual.
Once inside the mouth, chemicals pass down the oesophagus and into the
stomach. Food is digested with a strong acid produced by the stomach.
A few chemicals, such as alcohol, may pass across the stomach wall and
enter the veins and bloodstream there. But most chemicals move from
the stomach into the small intestine. The inside of the small
intestine has hundreds of tiny finger-like projections called villi
that have very thin walls and are filled with tiny blood vessels.
This allows the digested food to pass from the small intestine across
the walls of the villi and into the bloodstream.
Some chemicals that contaminate food or drink can also pass across the
thin walls of the villi and become absorbed into the bloodstream.
Other chemicals that are not soluble, or the basic units (molecules)
of which are too big to pass across the villi walls, will stay in the
gut and pass out through the faeces without being absorbed into the
bloodstream to a great extent. The absorption rate of a chemical
indicates the percentage of the amount swallowed that becomes absorbed
into the bloodstream. Most chemicals are only partly absorbed.
Some acids, caustics and organic chemicals may cause severe "burn"
damage to the digestive system if ingested in high concentrations.
2.2 Processing of chemicals in the body
After a chemical enters the body, it undergoes one or more of three
processes. The chemical may be metabolized by the body, stored or
accumulated in the body and/or excreted.
2.2.1 Metabolism
Metabolism is the process by which the body renders a 'foreign'
chemical more easily excretable and less toxic. The body is full of
special proteins called enzymes which initiate particular chemical
reactions. These reactions are necessary to, among other things,
convert the food we eat into a form that can be used by the tissues
and, combined with oxygen carried in the blood, to create heat and
energy. Some enzymes render foreign chemicals less toxic. They do
this by breaking down a chemical compound into simpler chemicals or by
converting a foreign chemical into a form that the body can excrete
more easily. Once a foreign chemical has been metabolized, it becomes
a different chemical, known as a metabolite. The process of
metabolism may involve many steps, and the result is many different
metabolites.
Metabolism may occur anywhere or everywhere in the body, or in just
one organ or type of tissue. Generally, this process renders a
chemical less toxic. However, some metabolites are as toxic as or
even more toxic than the original chemical. Such metabolites are
often called reactive metabolites. For most chemicals, the liver is
the main site of metabolism, but other organs such as the kidneys and
testes are also capable of metabolizing chemicals, the products of
which may be toxic.
2.2.2 Excretion
Excretion is the process by which unwanted chemicals are removed from
the body. Both foreign chemicals and those produced naturally by the
body are excreted. Most excretion occurs through the kidneys. Some
chemicals are excreted via the bile that passes from the liver into
the intestines, after which the chemicals pass out of the body in the
faeces. Other chemicals are excreted as a gas that is exhaled from
the lungs; small amounts may also be excreted in sweat.
In the kidneys, blood (which may be carrying waste or foreign
chemicals) is filtered through a set of small, twisting blood vessels
called the glomeruli. The fluid that is filtered off then passes
through tiny tubes called tubules. Here, water is reabsorbed and the
level of various salts adjusted. The combination of a glomerulus and
a tubule is called a nephron. There are hundreds of thousands of
nephrons in each kidney. The fluid that each nephron produces flows
through more tubes (the collecting ducts and the ureters) into the
bladder, from which urine is expelled from the body (see Fig. 16).
For some chemicals, it is possible to calculate the amount absorbed by
testing a urine sample. The concentration of the original chemical or
the concentration of a metabolite in the urine can also be determined.
Urine tests are often useful for determining whether absorption of a
chemical has occurred. For some chemicals, such as alcohol, exhaled
air can be used as an indication of absorption. Testing of urine,
blood or other body fluids to determine absorption is discussed
further in Chapter 3 of this Manual.
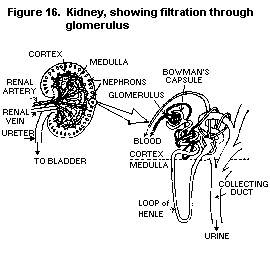 2.2.3 Storage or accumulation
Chemicals that undergo a slow metabolism or excretion are often stored
in various tissues of the body. Sustained exposure may thus increase
the amount of the chemical in tissues. Chemicals that are stored in
this way are said to accumulate.
The time taken for half of the total amount of chemical in the body to
be metabolized or excreted is called the half-time (or half-life)
of the chemical. If a chemical has a short half-time, it is
metabolized or excreted quickly by the body. Some chemicals, such as
cadmium, have a very long half-time of 15"20 years. The half-time of
a chemical varies from person to person, and this will influence how
sensitive an individual is to a chemical. An individual with a long
half-time will accumulate more of the chemical in his or her body.
The average half-time is sometimes given in information about
chemicals that are stored or that accumulate in the body.
2.3 Toxic effects of chemicals
The toxic effects, or toxicity, of a chemical can be defined as the
potential of that chemical to poison the body -- of the person
exposed, of an unborn baby (if the exposed person is pregnant), of a
future offspring of the exposed person or even of an offspring of the
exposed person's offspring.
The potential that a chemical substance has for causing negative
health effects depends principally on the toxicity of the chemical and
the degree of exposure. The toxicity is a property of the chemical
itself, while the exposure depends on how the chemical is used, for
example whether it is heated, sprayed or otherwise released into the
workplace environment. Another important concept in evaluating harm,
however, is the individual susceptibility of exposed persons. There
can be marked differences in reactions between workers who are exposed
to the same chemical, at the same worksite and in similar
concentrations. This may be due, for example, to gender (women, with
a greater relative proportion of body fat, may be more susceptible
than men to the harmful effects of solvents, for instance), age
(children and the elderly are generally more susceptible to chemical
hazards) or race (certain races may be genetically more vulnerable to
certain chemical exposures). Nutritional status may also have a
considerable effect on the action of some compounds.
Several concepts have been developed to help classify the toxic
effects of chemicals. These include:
* Acute effect -- The term acute means "of rapid onset and short
duration" and, with reference to chemicals, usually means a short
exposure with an immediate effect (see Fig. 17). While an acute
exposure can result in an acute effect, it can also result in a
chronic disease, e.g. permanent brain damage can result from
acute exposure to trialkyl tin compounds or from severe carbon
monoxide poisoning.
2.2.3 Storage or accumulation
Chemicals that undergo a slow metabolism or excretion are often stored
in various tissues of the body. Sustained exposure may thus increase
the amount of the chemical in tissues. Chemicals that are stored in
this way are said to accumulate.
The time taken for half of the total amount of chemical in the body to
be metabolized or excreted is called the half-time (or half-life)
of the chemical. If a chemical has a short half-time, it is
metabolized or excreted quickly by the body. Some chemicals, such as
cadmium, have a very long half-time of 15"20 years. The half-time of
a chemical varies from person to person, and this will influence how
sensitive an individual is to a chemical. An individual with a long
half-time will accumulate more of the chemical in his or her body.
The average half-time is sometimes given in information about
chemicals that are stored or that accumulate in the body.
2.3 Toxic effects of chemicals
The toxic effects, or toxicity, of a chemical can be defined as the
potential of that chemical to poison the body -- of the person
exposed, of an unborn baby (if the exposed person is pregnant), of a
future offspring of the exposed person or even of an offspring of the
exposed person's offspring.
The potential that a chemical substance has for causing negative
health effects depends principally on the toxicity of the chemical and
the degree of exposure. The toxicity is a property of the chemical
itself, while the exposure depends on how the chemical is used, for
example whether it is heated, sprayed or otherwise released into the
workplace environment. Another important concept in evaluating harm,
however, is the individual susceptibility of exposed persons. There
can be marked differences in reactions between workers who are exposed
to the same chemical, at the same worksite and in similar
concentrations. This may be due, for example, to gender (women, with
a greater relative proportion of body fat, may be more susceptible
than men to the harmful effects of solvents, for instance), age
(children and the elderly are generally more susceptible to chemical
hazards) or race (certain races may be genetically more vulnerable to
certain chemical exposures). Nutritional status may also have a
considerable effect on the action of some compounds.
Several concepts have been developed to help classify the toxic
effects of chemicals. These include:
* Acute effect -- The term acute means "of rapid onset and short
duration" and, with reference to chemicals, usually means a short
exposure with an immediate effect (see Fig. 17). While an acute
exposure can result in an acute effect, it can also result in a
chronic disease, e.g. permanent brain damage can result from
acute exposure to trialkyl tin compounds or from severe carbon
monoxide poisoning.
 * Chronic effect -- The term chronic means "of slow onset and long
duration" and usually refers to repeated exposure with a long
delay between the first exposure and the appearance of adverse
health effects (see Fig. 18). The delay between the time of
exposure and the onset of the health effect is called the
latency period.
* Acute and chronic effects -- A substance may have both an acute
and a chronic effect. For example, a single exposure to high
levels of carbon disulfide can result in unconsciousness (acute
effect), but repeated daily exposure for years at much lower
concentrations may result in damage to the central and peripheral
nervous system, as well as to the heart, i.e. at concentrations
that if experienced as a single exposure would not lead to
adverse effects (chronic effects).
* Chronic effect -- The term chronic means "of slow onset and long
duration" and usually refers to repeated exposure with a long
delay between the first exposure and the appearance of adverse
health effects (see Fig. 18). The delay between the time of
exposure and the onset of the health effect is called the
latency period.
* Acute and chronic effects -- A substance may have both an acute
and a chronic effect. For example, a single exposure to high
levels of carbon disulfide can result in unconsciousness (acute
effect), but repeated daily exposure for years at much lower
concentrations may result in damage to the central and peripheral
nervous system, as well as to the heart, i.e. at concentrations
that if experienced as a single exposure would not lead to
adverse effects (chronic effects).
 * Reversible (temporary) effect -- An effect that disappears if
exposure to that chemical ceases. Contact dermatitis, headaches
and nausea from exposure to solvents are examples of reversible
effects.
* Irreversible (permanent) effect -- An effect that will have a
lasting, damaging effect on the body, even if exposure to the
chemical causing that effect ceases. Cancer caused by exposure
to a chemical is an example of an irreversible effect.
* Local effect -- The harmful effect of a chemical at the point of
contact or entry to the body, e.g. burns to the skin.
* Systemic effect -- The effect of a chemical on the organs and
fluids of the body after absorption and transport from the point
of entry. Anaemia (a deficiency of haemoglobin due to a lack
of red blood cells) is a typical systemic effect. It can be
caused by a number of chemicals, including lead, beryllium,
benzene, cadmium and mercury compounds.
* Synergism -- The combined effect of exposure to more than one
chemical at one time, or to a chemical in combination with other
hazards such as heat, noise or radiation. The resultant health
effects of such exposure may be greater than the sum of the
individual effects of each hazard by itself. A mixture of sulfur
dioxide and sulfur trioxide, for example, has been shown to have
more severe effects on the functioning of respiratory passages
than the mathematical sum of their individual effects on the
respiratory system would suggest.
The toxic effects (or the toxicity) of a chemical can be classified in
the following ways:
* Corrosive -- A chemical that destroys or damages (burns) living
tissue on contact (see Fig. 19). For example, concentrated
solutions of strong acids such as sulfuric acid, or alkalis such
as caustic soda, cause chemical burns. A splash of a corrosive
liquid in the eye can result in permanent damage to eyesight.
* Irritant -- A chemical that will produce local irritation or
inflammation of the skin, eyes, nose or lung tissue.
* Sensitizer -- A chemical that causes an allergic reaction. A
person who is sensitized to a chemical will experience a
heightened reaction to it, whereas for the majority of
individuals the chemical will not be harmful at the same,
sometimes very low, dose. For a sensitized person, any
subsequent exposure -- whether through skin contact or by
inhalation -- will represent a health risk.
* Reversible (temporary) effect -- An effect that disappears if
exposure to that chemical ceases. Contact dermatitis, headaches
and nausea from exposure to solvents are examples of reversible
effects.
* Irreversible (permanent) effect -- An effect that will have a
lasting, damaging effect on the body, even if exposure to the
chemical causing that effect ceases. Cancer caused by exposure
to a chemical is an example of an irreversible effect.
* Local effect -- The harmful effect of a chemical at the point of
contact or entry to the body, e.g. burns to the skin.
* Systemic effect -- The effect of a chemical on the organs and
fluids of the body after absorption and transport from the point
of entry. Anaemia (a deficiency of haemoglobin due to a lack
of red blood cells) is a typical systemic effect. It can be
caused by a number of chemicals, including lead, beryllium,
benzene, cadmium and mercury compounds.
* Synergism -- The combined effect of exposure to more than one
chemical at one time, or to a chemical in combination with other
hazards such as heat, noise or radiation. The resultant health
effects of such exposure may be greater than the sum of the
individual effects of each hazard by itself. A mixture of sulfur
dioxide and sulfur trioxide, for example, has been shown to have
more severe effects on the functioning of respiratory passages
than the mathematical sum of their individual effects on the
respiratory system would suggest.
The toxic effects (or the toxicity) of a chemical can be classified in
the following ways:
* Corrosive -- A chemical that destroys or damages (burns) living
tissue on contact (see Fig. 19). For example, concentrated
solutions of strong acids such as sulfuric acid, or alkalis such
as caustic soda, cause chemical burns. A splash of a corrosive
liquid in the eye can result in permanent damage to eyesight.
* Irritant -- A chemical that will produce local irritation or
inflammation of the skin, eyes, nose or lung tissue.
* Sensitizer -- A chemical that causes an allergic reaction. A
person who is sensitized to a chemical will experience a
heightened reaction to it, whereas for the majority of
individuals the chemical will not be harmful at the same,
sometimes very low, dose. For a sensitized person, any
subsequent exposure -- whether through skin contact or by
inhalation -- will represent a health risk.
 * Asphyxiant -- A chemical that interferes with the oxygenation of
the body tissues. There are two types of asphyxiation: simple
asphyxiation, whereby oxygen in the air is replaced by a gas to
a level at which it cannot sustain life; and chemical
asphyxiation, whereby a direct chemical action interferes with
the body's ability to transport and use oxygen. Examples of
chemical asphyxiants include carbon monoxide and cyanides.
* Carcinogen -- A chemical that causes cancer. Cancer is a group of
diseases characterized by the manner in which abnormal cells in
the body multiply and spread out of control. The key feature of
cancer is the "malignant" or deadly way that its cells crowd out
other cells and interfere with the normal functioning of the body
(see Fig. 20).
* Mutagen -- A chemical that can cause permanent damage to the
deoxyribonucleic acid (DNA) in a cell (see Fig. 21). DNA is
a molecule that carries the genetic information controlling the
growth and function of cells. DNA damage in the human egg or
sperm may lead to reduced fertility, spontaneous abortion
(miscarriage), birth defects and heritable diseases. Thus, any
chemical known to be a mutagen must be treated with caution.
Figure 20. Carcinogenic effect (from HSG 19: Pentachlorophenol)
The results of animal studies, designed to assess the carcinogenicity
of PCP and reported to date, have been negative. Carcinogenicity
bioassays with one other chlorophenol (2,4,6-T3CP) and a mixture of
two H6CDD congeners found in PCP have been positive. Hence, the
carcinogenic effects of long-term exposure of animals to technical PCP
are not clear.
Figure 21. Mutagenic effect (from HSG 32: d-Phenothrin)
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
* Teratogen -- A chemical that, if present in the bloodstream of a
pregnant woman, crosses the placenta, affecting the developing
fetus and resulting in structural or functional congenital
abnormalities, or cancer, in the child. These effects may not be
observable until the child becomes an adult. One of the
better-known examples of a teratogen is Thalidomide, which, in
the 1960s, caused many cases of phocomelia (a reduction of the
limbs to the extent that the hands and feet are attached directly
to the body) in babies born to women who took the drug during the
early stages of pregnancy. Several industrial chemicals have been
shown to be experimental teratogens; some, such as ethylene
dichloride and vinyl chloride, have been shown to be teratogenic
in humans.
* Fetotoxicant -- A chemical that adversely affects the developing
fetus, resulting in low birth weight, symptoms of poisoning at
birth or stillbirth -- i.e. the fetus dies before it is born (see
also Fig. 22).
Figure 22. Fetotoxic effect (from HSG 19: Pentachlorophenol)
PCP is fetotoxic, delaying the development of rat embryos and
reducing litter size, neonatal body weight, neonatal survival,
and the growth of weanlings. The no-observed-adverse-effect
level for technical PCP is a maternal dose of 5 mg/kg body
weight per day during organogenesis. In one study, it was
reported that purified PCP was slightly more embryo/fetotoxic
than technical PCP, presumably because contaminants induced
enzymes that detoxified the parent compound.
2.4 Effects on body systems
Chemicals may have toxic effects on all cells in the body, or they may
affect only particular organs or body systems. Organs that are
especially sensitive to the toxic effects of a particular chemical are
known as target organs. A toxic action on one particular organ or
body system may be attributable to a particular property of the
chemical or related to the way in which a chemical enters or
circulates within the body. Toxic effects of chemicals on particular
organs and body systems include those on the:
* Lungs and the respiratory system. Short-term effects are caused
mainly by irritation (producing bronchitis or pneumonitis)
and chemical burns. Serious chemical burns inside the lungs can
result in the lungs becoming filled with fluid (pulmonary oedema)
and can be fatal. Some chemicals can sensitize or cause an
allergic reaction in the airways, leading to wheezing and
shortness of breath (asthma) (see Fig. 23).
Long-term (chronic) conditions include scarring of the lung
tissue following exposure to chemical dusts (fibrosis or
pneumoconiosis).
Figure 23. Effects on the respiratory system
(from HSG 35: Phosphorus Trichloride)
Depending on their concentration, the vapours can be irritating or
corrosive, but they may be less irritating than their hydrolysis
products. However, exposure to the vapours can cause necrosis of the
tissues of the respiratory tract. The vapours can be deeply inhaled
and may reach the lower airways where they hydrolyse to produce
hydrogen chloride and phosphorous or phosphoric acid.
Irritation of the airways causes swelling and bronchospasm resulting
in tightness of the chest, wheezing and difficulty in breathing.
Reactive secretion of mucus causes a cough with sputum and possible
bronchial obstruction and local lung collapse.
* Liver. Chemicals that can affect the liver are called
hepatotoxins. Most chemicals are metabolized in the liver, and
many therefore have the potential to damage the liver cells.
Possible short-term effects of chemicals on the liver include
inflammation of the liver cells (chemical hepatitis), necrosis
(cell death) and jaundice. Long-term effects include cirrhosis
(scarring) of the liver and liver cancer.
* Kidneys and urinary tract. Chemicals that can affect the kidneys
are called nephrotoxins. The effects of chemicals on the kidneys
include sudden failure of the kidneys (acute renal failure),
chronic renal failure and kidney or bladder cancer.
* Nervous system. Chemicals that affect the nerves are called
neurotoxins. Exposure to certain chemicals may cause a slowing
down of the function of the brain. Symptoms include sleepiness
and loss of alertness, followed by loss of consciousness (see
Fig. 24). Some of these effects are called "narcotic" effects,
based on the Greek word "narkos" meaning "sleep". Some illegal
drugs have narcotic effects and are therefore called narcotics.
Another term for these "sleep effects" is central nervous system
depression (not to be confused with depression as a mental health
problem). Other chemicals, particularly pesticides, can poison an
enzyme in the nerves going to the muscles, causing muscle
twitching and paralysis. Yet another group of chemicals may
slowly poison the nerves to the hands and feet, resulting in loss
of feeling and weakness.
Figure 24. Effects on the nervous system
(from HSG 3: 1-Butanol)
Ingestion of the liquid or inhalation of the vapour may result in
headache, drowsiness, and narcosis. The occurrence of vertigo under
conditions of severe and prolonged exposure to vapour mixtures of
1-butanol and isobutanol has been reported. From this study, it is
not possible to attribute the vertigo to a single cause. Symptoms are
reversible when exposure ceases.
* Blood and bone marrow. Some chemicals, such as arsine, damage
the red blood cells, causing haemolytic anaemia. Other chemicals
can damage the bone marrow and other organs where blood cells are
formed or can cause cancer of the blood-forming cells (e.g.
benzene causes leukaemia).
* Heart and blood vessels (cardiovascular system). Some solvents
(such as trichloroethylene) and gases can cause fatal
disturbances of the heart rhythm. Others, such as carbon
disulfide, may cause an increase in blood vessel disease, which
may lead to a heart attack.
* Skin. Many chemicals irritate the skin, causing "irritant
contact dermatitis", or they may sensitize the skin, causing
"allergic contact dermatitis". A rash results in both cases.
This rash may be very severe, widespread and disabling. Other
chemicals may cause acne (chloracne), loss of pigmentation
(vitiligo), increased sensitivity to sunlight
(photosensitization) or skin cancer.
* Reproductive system. Many chemicals are teratogens and
experimental germ cell mutagens. In addition, some chemicals can
directly affect the ovaries and testes, resulting in disruption
of menstruation and sexual function. Such chemicals are called
gonadotoxins.
* Other systems. Chemicals can also affect the immune system,
bones, muscles and particular glands, such as the thyroid gland.
2.5 Dose, effects and response
Increasing levels of exposure to or dose of a chemical will generally
lead to more severe effects. For instance, higher concentrations of
carbon monoxide in the air will progressively reduce the capacity of
an exposed person's blood to carry oxygen. The resultant lack of
oxygen in the blood leads initially to headaches. As the oxygen
levels decline further, the symptoms worsen: nausea, unconsciousness
and eventually death occur. This progression in severity of effect as
the dose increases is called the "dose-effect relationship".
In the case of a population or group of workers, increasing exposure
levels will also lead to an increasing proportion of the group
manifesting a specific effect. For example, increasing exposure to
benzidine dyes will result in a higher incidence of bladder cancers
among the exposed population. Similarly, increasing exposure to lead
will be reflected in a greater proportion of workers who undergo blood
changes (FEP, ALAD, etc.; see Fig. 25). This frequency of affected
people in an exposed population is called "the response". The
increase in response with increasing exposure level or dose is known
as the "dose"response relationship".
Another important concept is "threshold dose" or the
"no-observed-effect-level" (NOEL). This means that at low levels
of exposure to a chemical, the severity of the effect and the response
decrease, and that at a certain point there is no effect on health.
Usually this level is determined by exposing animals to lower and
lower concentrations of a chemical until a level is found at which no
effect on the animals is observed. It is impossible, however, to
examine every aspect of a test animal, and therefore effects may be
missed. Also, unless lifetime tests are performed (and they are not in
every case), long-term or chronic effects may also be missed, even
though subacute effects are noted. In addition, only limited numbers
of animals can be studied, whereas in real life many thousands of
people may be exposed to a chemical, some of whom will be much more
sensitive to its health effects than the majority. Even a very low
dose of a cancer-causing substance may increase the risk of cancer.
Studies of workers do not usually look at a sufficiently large group
to prove that exposure to low doses does, or does not, increase cancer
rates.
* Asphyxiant -- A chemical that interferes with the oxygenation of
the body tissues. There are two types of asphyxiation: simple
asphyxiation, whereby oxygen in the air is replaced by a gas to
a level at which it cannot sustain life; and chemical
asphyxiation, whereby a direct chemical action interferes with
the body's ability to transport and use oxygen. Examples of
chemical asphyxiants include carbon monoxide and cyanides.
* Carcinogen -- A chemical that causes cancer. Cancer is a group of
diseases characterized by the manner in which abnormal cells in
the body multiply and spread out of control. The key feature of
cancer is the "malignant" or deadly way that its cells crowd out
other cells and interfere with the normal functioning of the body
(see Fig. 20).
* Mutagen -- A chemical that can cause permanent damage to the
deoxyribonucleic acid (DNA) in a cell (see Fig. 21). DNA is
a molecule that carries the genetic information controlling the
growth and function of cells. DNA damage in the human egg or
sperm may lead to reduced fertility, spontaneous abortion
(miscarriage), birth defects and heritable diseases. Thus, any
chemical known to be a mutagen must be treated with caution.
Figure 20. Carcinogenic effect (from HSG 19: Pentachlorophenol)
The results of animal studies, designed to assess the carcinogenicity
of PCP and reported to date, have been negative. Carcinogenicity
bioassays with one other chlorophenol (2,4,6-T3CP) and a mixture of
two H6CDD congeners found in PCP have been positive. Hence, the
carcinogenic effects of long-term exposure of animals to technical PCP
are not clear.
Figure 21. Mutagenic effect (from HSG 32: d-Phenothrin)
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
* Teratogen -- A chemical that, if present in the bloodstream of a
pregnant woman, crosses the placenta, affecting the developing
fetus and resulting in structural or functional congenital
abnormalities, or cancer, in the child. These effects may not be
observable until the child becomes an adult. One of the
better-known examples of a teratogen is Thalidomide, which, in
the 1960s, caused many cases of phocomelia (a reduction of the
limbs to the extent that the hands and feet are attached directly
to the body) in babies born to women who took the drug during the
early stages of pregnancy. Several industrial chemicals have been
shown to be experimental teratogens; some, such as ethylene
dichloride and vinyl chloride, have been shown to be teratogenic
in humans.
* Fetotoxicant -- A chemical that adversely affects the developing
fetus, resulting in low birth weight, symptoms of poisoning at
birth or stillbirth -- i.e. the fetus dies before it is born (see
also Fig. 22).
Figure 22. Fetotoxic effect (from HSG 19: Pentachlorophenol)
PCP is fetotoxic, delaying the development of rat embryos and
reducing litter size, neonatal body weight, neonatal survival,
and the growth of weanlings. The no-observed-adverse-effect
level for technical PCP is a maternal dose of 5 mg/kg body
weight per day during organogenesis. In one study, it was
reported that purified PCP was slightly more embryo/fetotoxic
than technical PCP, presumably because contaminants induced
enzymes that detoxified the parent compound.
2.4 Effects on body systems
Chemicals may have toxic effects on all cells in the body, or they may
affect only particular organs or body systems. Organs that are
especially sensitive to the toxic effects of a particular chemical are
known as target organs. A toxic action on one particular organ or
body system may be attributable to a particular property of the
chemical or related to the way in which a chemical enters or
circulates within the body. Toxic effects of chemicals on particular
organs and body systems include those on the:
* Lungs and the respiratory system. Short-term effects are caused
mainly by irritation (producing bronchitis or pneumonitis)
and chemical burns. Serious chemical burns inside the lungs can
result in the lungs becoming filled with fluid (pulmonary oedema)
and can be fatal. Some chemicals can sensitize or cause an
allergic reaction in the airways, leading to wheezing and
shortness of breath (asthma) (see Fig. 23).
Long-term (chronic) conditions include scarring of the lung
tissue following exposure to chemical dusts (fibrosis or
pneumoconiosis).
Figure 23. Effects on the respiratory system
(from HSG 35: Phosphorus Trichloride)
Depending on their concentration, the vapours can be irritating or
corrosive, but they may be less irritating than their hydrolysis
products. However, exposure to the vapours can cause necrosis of the
tissues of the respiratory tract. The vapours can be deeply inhaled
and may reach the lower airways where they hydrolyse to produce
hydrogen chloride and phosphorous or phosphoric acid.
Irritation of the airways causes swelling and bronchospasm resulting
in tightness of the chest, wheezing and difficulty in breathing.
Reactive secretion of mucus causes a cough with sputum and possible
bronchial obstruction and local lung collapse.
* Liver. Chemicals that can affect the liver are called
hepatotoxins. Most chemicals are metabolized in the liver, and
many therefore have the potential to damage the liver cells.
Possible short-term effects of chemicals on the liver include
inflammation of the liver cells (chemical hepatitis), necrosis
(cell death) and jaundice. Long-term effects include cirrhosis
(scarring) of the liver and liver cancer.
* Kidneys and urinary tract. Chemicals that can affect the kidneys
are called nephrotoxins. The effects of chemicals on the kidneys
include sudden failure of the kidneys (acute renal failure),
chronic renal failure and kidney or bladder cancer.
* Nervous system. Chemicals that affect the nerves are called
neurotoxins. Exposure to certain chemicals may cause a slowing
down of the function of the brain. Symptoms include sleepiness
and loss of alertness, followed by loss of consciousness (see
Fig. 24). Some of these effects are called "narcotic" effects,
based on the Greek word "narkos" meaning "sleep". Some illegal
drugs have narcotic effects and are therefore called narcotics.
Another term for these "sleep effects" is central nervous system
depression (not to be confused with depression as a mental health
problem). Other chemicals, particularly pesticides, can poison an
enzyme in the nerves going to the muscles, causing muscle
twitching and paralysis. Yet another group of chemicals may
slowly poison the nerves to the hands and feet, resulting in loss
of feeling and weakness.
Figure 24. Effects on the nervous system
(from HSG 3: 1-Butanol)
Ingestion of the liquid or inhalation of the vapour may result in
headache, drowsiness, and narcosis. The occurrence of vertigo under
conditions of severe and prolonged exposure to vapour mixtures of
1-butanol and isobutanol has been reported. From this study, it is
not possible to attribute the vertigo to a single cause. Symptoms are
reversible when exposure ceases.
* Blood and bone marrow. Some chemicals, such as arsine, damage
the red blood cells, causing haemolytic anaemia. Other chemicals
can damage the bone marrow and other organs where blood cells are
formed or can cause cancer of the blood-forming cells (e.g.
benzene causes leukaemia).
* Heart and blood vessels (cardiovascular system). Some solvents
(such as trichloroethylene) and gases can cause fatal
disturbances of the heart rhythm. Others, such as carbon
disulfide, may cause an increase in blood vessel disease, which
may lead to a heart attack.
* Skin. Many chemicals irritate the skin, causing "irritant
contact dermatitis", or they may sensitize the skin, causing
"allergic contact dermatitis". A rash results in both cases.
This rash may be very severe, widespread and disabling. Other
chemicals may cause acne (chloracne), loss of pigmentation
(vitiligo), increased sensitivity to sunlight
(photosensitization) or skin cancer.
* Reproductive system. Many chemicals are teratogens and
experimental germ cell mutagens. In addition, some chemicals can
directly affect the ovaries and testes, resulting in disruption
of menstruation and sexual function. Such chemicals are called
gonadotoxins.
* Other systems. Chemicals can also affect the immune system,
bones, muscles and particular glands, such as the thyroid gland.
2.5 Dose, effects and response
Increasing levels of exposure to or dose of a chemical will generally
lead to more severe effects. For instance, higher concentrations of
carbon monoxide in the air will progressively reduce the capacity of
an exposed person's blood to carry oxygen. The resultant lack of
oxygen in the blood leads initially to headaches. As the oxygen
levels decline further, the symptoms worsen: nausea, unconsciousness
and eventually death occur. This progression in severity of effect as
the dose increases is called the "dose-effect relationship".
In the case of a population or group of workers, increasing exposure
levels will also lead to an increasing proportion of the group
manifesting a specific effect. For example, increasing exposure to
benzidine dyes will result in a higher incidence of bladder cancers
among the exposed population. Similarly, increasing exposure to lead
will be reflected in a greater proportion of workers who undergo blood
changes (FEP, ALAD, etc.; see Fig. 25). This frequency of affected
people in an exposed population is called "the response". The
increase in response with increasing exposure level or dose is known
as the "dose"response relationship".
Another important concept is "threshold dose" or the
"no-observed-effect-level" (NOEL). This means that at low levels
of exposure to a chemical, the severity of the effect and the response
decrease, and that at a certain point there is no effect on health.
Usually this level is determined by exposing animals to lower and
lower concentrations of a chemical until a level is found at which no
effect on the animals is observed. It is impossible, however, to
examine every aspect of a test animal, and therefore effects may be
missed. Also, unless lifetime tests are performed (and they are not in
every case), long-term or chronic effects may also be missed, even
though subacute effects are noted. In addition, only limited numbers
of animals can be studied, whereas in real life many thousands of
people may be exposed to a chemical, some of whom will be much more
sensitive to its health effects than the majority. Even a very low
dose of a cancer-causing substance may increase the risk of cancer.
Studies of workers do not usually look at a sufficiently large group
to prove that exposure to low doses does, or does not, increase cancer
rates.
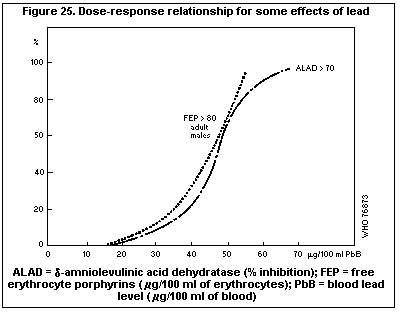 When drawing up regulations for the permissible (although not
necessarily safe) levels of chemicals in food, drinking-water and the
environment, government bodies usually divide the NOEL from animal
studies by a safety factor of 100 or 1000 (see Fig. 26). Occupational
exposure limits are usually set with a safety factor of 2-10 at the
most and even sometimes with no safety factor.
2.6 How is the toxicity of a chemical determined?
Knowledge of the toxicity of chemicals is gathered by studying the
effects of:
* exposure to the chemical in experimental animals, lower organisms
-- such as bacteria -- and laboratory cultures of cells from
mammals; and
* exposure of people to the chemical.
Figure 26. Use of safety factors (from HSG 19: Pentachlorophenol)
The US National Academy of Sciences (1977) calculated an acceptable
daily intake (ADI) for PCP of 3 µg/kg body weight per day. This ADI
is based on data from a feeding study on rats and a 1000-fold safety
factor. The results of long-term studies indicate that the
no-observed-adverse-effect level for rats is below 3 mg/kg body weight
per day. A recent human study has shown that the steady-state body
burden is 10-20 times higher than the value extrapolated from rat
pharmacokinetic data, suggesting that caution should be applied when
extrapolating directly from the rat model to humans. Furthermore, the
ADI in the USA was not based on an inhalation study, and does not
account for the possibly greater toxicity of PCP via inhalation, as
indicated by animal studies. Hence, the safety factor of 1000 used to
derive this ADI value is by no means too conservative. The intake for
a 60 kg adult exposed to concentrations of PCP at the ADI level would
be 180 µg/person per day.
2.6.1 Animal studies
2.6.1.1 Acute toxicity tests
* LD50 and LC50 tests
The standard test for acute (short-term) toxicity is to feed animals
increasing amounts of a chemical over a period of 14 days until the
animals start to die. Alternatively, the chemicals can be applied to
the animals' skin until a reaction is observed. The amount of the
chemical that kills 50% of the exposed animals is called the lethal
dose for 50%, or the LD50. The LD50 may be "oral" or "dermal",
depending on the method of exposure (see Fig. 27). Lethal doses with
respect to inhalation of chemicals in the form of a gas or aerosol can
also be tested. In this case, the concentration of gas or vapour that
kills half the animals is known as the lethal concentration for 50%,
or the LC50.
Figure 27. Effects on animals (from HSG 3: 1-Butanol)
The oral LD50 values for 1-butanol for the rat range from 0.7 to
2.1 g/kg body weight. Therefore, it is slightly toxic according to
the classification of Hodge & Sterner. It is markedly irritating to
the eyes and moderately irritating to the skin. The primary effects
from exposure to vapour for short periods are irritation of mucous
membranes, and central nervous system depression. The potency of
1-butanol for intoxication is approximately 6 times that of ethanol.
A number of investigations have shown non-specific membrane effects of
1-butanol. Effects of repeated inhalation exposure in animals include
pathological changes in the lungs, degenerative lesions in the liver
and kidneys, and narcosis. However, it is not possible to determine a
no-observed-adverse-effect level from the animal studies available.
1-Butanol has been found to be non-mutagenic. Adequate data are not
available on carcinogenicity, teratogenicity, or effects on
reproduction.
The LD50 and the LC50 are very widely used as indices of toxicity.
The following criteria are often used for purposes of classification
of acute toxic effects in animals (see Table 2):
Table 2. Classification of acute toxicity in animals
Oral LD50 Dermal LD50 Inhalation LC50
rat rat or rabbit rat
(mg/kg) (mg/kg) (mg/m3/4 h)
Harmful 200-2000 400-2000 2000-20 000
Toxic 25-200 50-400 500-2000
Very toxic < 25 < 50 < 500
The scale of Hodge & Sterner is also often used, in this case to
classify the acute toxicity of chemicals to humans (see Table 3):
Table 3. Classification of acute toxicity in humans
Toxicity rating Dosage Probable lethal
dose for average
human adult
1. Practically non-toxic > 15 g/kg > 1 litre
2. Slightly toxic 5-15 g/kg 0.5-1 litre
3. Moderately toxic 0.5-5 g/kg 30-500 ml
4. Very toxic 50-500 mg/kg 3-30 ml
5. Extremely toxic 5-50 mg/kg 7 drops-3 ml
6. Supertoxic < 5 mg/kg A taste (< 7 drops)
It is impossible to assess the health risk posed by a chemical on the
basis of its LD50 or LC50 alone. Moreover, the LD50 and LC50
give no information about the mechanism or type of toxicity of a
chemical, or its possible long-term or chronic effects.
Thus, the LD50 and LC50 are very crude indices of toxicity.
* The fixed-dose procedure
Many national and international bodies are now trying to modify or
replace the LD50 and LC50 tests by simpler methods, e.g. a
fixed-dose procedure that uses fewer animals. The fixed-dose
procedure requires only a small number of animals, and analysts can
evaluate a chemical's toxicity without the animals having to die as an
end-point. The idea is to examine how a set dose of a chemical
affects a group of animals, the dose being based on what is already
known about the physical and chemical properties of the substance
being evaluated.
* Irritation and corrosion tests
Irritation and corrosion tests provide some specific information. The
chemical being tested is applied to the test animal's skin, and the
area is examined over the next few days for signs of a rash or flared
response. Tests can also be carried out on the animal's eyes (this is
known as the "Draize" test).
2.6.1.2 Subchronic toxicity tests
Subchronic toxicity tests are normally 90-day inhalation or ingestion
studies to check for specific and obvious effects of the chemical on
the organs and biochemistry of the animal subjects. These types of
tests involve repeated exposure to the chemical being tested and are
primarily directed at detecting toxic effects that are not evident
from acute exposure. Crude studies simply call for examination of the
organs for gross abnormalities apparent to the naked eye; more
sophisticated studies involve taking tissue slices and examining them
under a microscope to check for abnormalities of the cells in the
organs. Most studies involve taking regular samples of blood or urine
from the animals for examination and analysis. These tests form the
basis for doses used in chronic bioassays (see next section).
2.6.1.3 Chronic (lifetime) bioassays
The purpose of lifetime or chronic bioassays is to determine whether
chemicals lead to any health effects that may take a long time to
develop, such as cancer, or whether long-term exposure to chemicals
leads to health effects on organs such as the kidneys (see Fig. 28).
Figure 28. Chronic bioassays (from HSG 34: Fenvalerate)
In a long-term toxicity study, microgranulomatous changes were also
observed in rats at 500 mg/kg in the diet. The NOEL for these
microgranulomatous changes was 150 mg/kg in the diet.
Fenvalerate, when fed at dietary levels up to 3000 mg/kg for 78 weeks
and 1250 mg/kg for 2 years, was not carcinogenic to mice. Nor was it
carcinogenic to rats when fed at dietary levels up to 1000 mg/kg for
2 years.
These studies are performed by exposing animals, by ingestion or
inhalation, to the chemical being tested, for most or all of the
animals' lifetime. In rats, this may be two years; in mice, a little
less. In a typical test, 50 mice or rats of each sex are exposed to a
high, but non-lethal, dose of the chemical under study. The test
animals are compared throughout their lifetime with a similar number
of "control" animals, which are similar in all respects except that
they are not exposed to the chemical. A good study exposes different
groups of animals of both sexes to different doses of the chemical.
Up to 500 animals may be used in a study of the chemical.
2.6.1.4 Short-term mutagenicity tests
Bacteria and animal cells grown in test-tubes and colonies of fruit
flies or other insects are convenient for the rapid and cheap
investigation of the potentially carcinogenic and mutagenic effects of
chemicals. The best-known and most widely used of these tests is the
Salmonella mutagenicity test (commonly known as the Ames test).
This involves growing special bacteria in the laboratory and exposing
them to the chemical in question. The test detects mutations in the
bacteria, i.e. it is a test for mutagenic effects. There are a number
of other short-term mutagenic tests or "mutagenicity assays", as they
are often called. These tests are often referred to as being in
vitro. They are thus distinguished from in vivo tests, which use
living tissue such as animals and humans (see Fig. 29).
Many, but not all, chemicals that cause cancer in animals and may
cause cancer in humans are mutagenic.
Figure 29. Short-term mutagenicity tests
(from HSG 34: Fenvalerate)
Fenvalerate did not show any mutagenic or chromosome-damaging
activities in several in vitro and in vivo assays.
2.6.1.5 Reproductive studies
Animal studies to check for the adverse effects of a chemical on
reproduction involve exposing one or both parents to the chemical
being tested, prior to mating, and then observing the effects on any
offspring. Sometimes only a pregnant animal is exposed. Reproductive
effects are classified according to whether the offspring are fewer in
number, lower in birth weight deformed or damaged in some way.
Multigeneration studies are sometimes necessary to detect effects that
may be passed on to future generations (see Fig. 30).
2.6.1.6 Behavioural tests
The effects of chemicals on the behaviour of test animals and on their
ability to learn (e.g. to find their way out of a maze) will often
indicate subtle effects of a chemical on the brain and nervous system
that are missed by other tests. Behavioural tests have been used to
show the effects of exposure to compounds such as organic solvents and
lead.
Figure 30. Reproductive effects (from HSG 34: Fenvalerate)
Fenvalerate is not teratogenic to mice or rabbits at doses up to
50 mg/kg body weight per day. It did not show any toxic effects on
reproductive parameters in a three-generation rat reproduction study
at doses up to 250 mg/kg diet.
2.6.2 Human evidence
2.6.2.1 Case reports
Case reports are accounts, usually by occupational health physicians,
of small numbers of workers or individual workers whose disease
appears to be attributable to workplace exposure. Often the part of a
process at which the chemical may have caused the problem is known,
but not the precise degree of exposure. Much information about
occupational health hazards has initially been found following
examination of case reports.
2.6.2.2 Epidemiological studies
Epidemiological studies investigate the health of a group of people to
establish whether they are affected by the chemical to which they are
exposed at work or in the general environment. These studies involve
comparing the disease outcome of an exposed group of people over time
with that of unexposed persons. Two of the more common methods of
investigation in epidemiology are case-control studies and cohort
studies.
Case-control studies are relatively simple to carry out and
increasingly used to investigate the cause of diseases - especially
rare diseases. They basically compare people with the disease, or
other outcome, under study with a suitable control group unaffected by
the disease or outcome in an attempt to identify the cause.
Cohort studies, also called follow-up or incidence studies, look
at a group of people (a cohort) who are classified into subgroups
according to exposure to a potential cause of disease or outcome.
Differences in exposure (to chemicals, for instance) are examined and
measured and the whole cohort followed up to see how the subsequent
development of the disease, or outcome, differs between the exposed
and unexposed groups (see Fig. 31).
When drawing up regulations for the permissible (although not
necessarily safe) levels of chemicals in food, drinking-water and the
environment, government bodies usually divide the NOEL from animal
studies by a safety factor of 100 or 1000 (see Fig. 26). Occupational
exposure limits are usually set with a safety factor of 2-10 at the
most and even sometimes with no safety factor.
2.6 How is the toxicity of a chemical determined?
Knowledge of the toxicity of chemicals is gathered by studying the
effects of:
* exposure to the chemical in experimental animals, lower organisms
-- such as bacteria -- and laboratory cultures of cells from
mammals; and
* exposure of people to the chemical.
Figure 26. Use of safety factors (from HSG 19: Pentachlorophenol)
The US National Academy of Sciences (1977) calculated an acceptable
daily intake (ADI) for PCP of 3 µg/kg body weight per day. This ADI
is based on data from a feeding study on rats and a 1000-fold safety
factor. The results of long-term studies indicate that the
no-observed-adverse-effect level for rats is below 3 mg/kg body weight
per day. A recent human study has shown that the steady-state body
burden is 10-20 times higher than the value extrapolated from rat
pharmacokinetic data, suggesting that caution should be applied when
extrapolating directly from the rat model to humans. Furthermore, the
ADI in the USA was not based on an inhalation study, and does not
account for the possibly greater toxicity of PCP via inhalation, as
indicated by animal studies. Hence, the safety factor of 1000 used to
derive this ADI value is by no means too conservative. The intake for
a 60 kg adult exposed to concentrations of PCP at the ADI level would
be 180 µg/person per day.
2.6.1 Animal studies
2.6.1.1 Acute toxicity tests
* LD50 and LC50 tests
The standard test for acute (short-term) toxicity is to feed animals
increasing amounts of a chemical over a period of 14 days until the
animals start to die. Alternatively, the chemicals can be applied to
the animals' skin until a reaction is observed. The amount of the
chemical that kills 50% of the exposed animals is called the lethal
dose for 50%, or the LD50. The LD50 may be "oral" or "dermal",
depending on the method of exposure (see Fig. 27). Lethal doses with
respect to inhalation of chemicals in the form of a gas or aerosol can
also be tested. In this case, the concentration of gas or vapour that
kills half the animals is known as the lethal concentration for 50%,
or the LC50.
Figure 27. Effects on animals (from HSG 3: 1-Butanol)
The oral LD50 values for 1-butanol for the rat range from 0.7 to
2.1 g/kg body weight. Therefore, it is slightly toxic according to
the classification of Hodge & Sterner. It is markedly irritating to
the eyes and moderately irritating to the skin. The primary effects
from exposure to vapour for short periods are irritation of mucous
membranes, and central nervous system depression. The potency of
1-butanol for intoxication is approximately 6 times that of ethanol.
A number of investigations have shown non-specific membrane effects of
1-butanol. Effects of repeated inhalation exposure in animals include
pathological changes in the lungs, degenerative lesions in the liver
and kidneys, and narcosis. However, it is not possible to determine a
no-observed-adverse-effect level from the animal studies available.
1-Butanol has been found to be non-mutagenic. Adequate data are not
available on carcinogenicity, teratogenicity, or effects on
reproduction.
The LD50 and the LC50 are very widely used as indices of toxicity.
The following criteria are often used for purposes of classification
of acute toxic effects in animals (see Table 2):
Table 2. Classification of acute toxicity in animals
Oral LD50 Dermal LD50 Inhalation LC50
rat rat or rabbit rat
(mg/kg) (mg/kg) (mg/m3/4 h)
Harmful 200-2000 400-2000 2000-20 000
Toxic 25-200 50-400 500-2000
Very toxic < 25 < 50 < 500
The scale of Hodge & Sterner is also often used, in this case to
classify the acute toxicity of chemicals to humans (see Table 3):
Table 3. Classification of acute toxicity in humans
Toxicity rating Dosage Probable lethal
dose for average
human adult
1. Practically non-toxic > 15 g/kg > 1 litre
2. Slightly toxic 5-15 g/kg 0.5-1 litre
3. Moderately toxic 0.5-5 g/kg 30-500 ml
4. Very toxic 50-500 mg/kg 3-30 ml
5. Extremely toxic 5-50 mg/kg 7 drops-3 ml
6. Supertoxic < 5 mg/kg A taste (< 7 drops)
It is impossible to assess the health risk posed by a chemical on the
basis of its LD50 or LC50 alone. Moreover, the LD50 and LC50
give no information about the mechanism or type of toxicity of a
chemical, or its possible long-term or chronic effects.
Thus, the LD50 and LC50 are very crude indices of toxicity.
* The fixed-dose procedure
Many national and international bodies are now trying to modify or
replace the LD50 and LC50 tests by simpler methods, e.g. a
fixed-dose procedure that uses fewer animals. The fixed-dose
procedure requires only a small number of animals, and analysts can
evaluate a chemical's toxicity without the animals having to die as an
end-point. The idea is to examine how a set dose of a chemical
affects a group of animals, the dose being based on what is already
known about the physical and chemical properties of the substance
being evaluated.
* Irritation and corrosion tests
Irritation and corrosion tests provide some specific information. The
chemical being tested is applied to the test animal's skin, and the
area is examined over the next few days for signs of a rash or flared
response. Tests can also be carried out on the animal's eyes (this is
known as the "Draize" test).
2.6.1.2 Subchronic toxicity tests
Subchronic toxicity tests are normally 90-day inhalation or ingestion
studies to check for specific and obvious effects of the chemical on
the organs and biochemistry of the animal subjects. These types of
tests involve repeated exposure to the chemical being tested and are
primarily directed at detecting toxic effects that are not evident
from acute exposure. Crude studies simply call for examination of the
organs for gross abnormalities apparent to the naked eye; more
sophisticated studies involve taking tissue slices and examining them
under a microscope to check for abnormalities of the cells in the
organs. Most studies involve taking regular samples of blood or urine
from the animals for examination and analysis. These tests form the
basis for doses used in chronic bioassays (see next section).
2.6.1.3 Chronic (lifetime) bioassays
The purpose of lifetime or chronic bioassays is to determine whether
chemicals lead to any health effects that may take a long time to
develop, such as cancer, or whether long-term exposure to chemicals
leads to health effects on organs such as the kidneys (see Fig. 28).
Figure 28. Chronic bioassays (from HSG 34: Fenvalerate)
In a long-term toxicity study, microgranulomatous changes were also
observed in rats at 500 mg/kg in the diet. The NOEL for these
microgranulomatous changes was 150 mg/kg in the diet.
Fenvalerate, when fed at dietary levels up to 3000 mg/kg for 78 weeks
and 1250 mg/kg for 2 years, was not carcinogenic to mice. Nor was it
carcinogenic to rats when fed at dietary levels up to 1000 mg/kg for
2 years.
These studies are performed by exposing animals, by ingestion or
inhalation, to the chemical being tested, for most or all of the
animals' lifetime. In rats, this may be two years; in mice, a little
less. In a typical test, 50 mice or rats of each sex are exposed to a
high, but non-lethal, dose of the chemical under study. The test
animals are compared throughout their lifetime with a similar number
of "control" animals, which are similar in all respects except that
they are not exposed to the chemical. A good study exposes different
groups of animals of both sexes to different doses of the chemical.
Up to 500 animals may be used in a study of the chemical.
2.6.1.4 Short-term mutagenicity tests
Bacteria and animal cells grown in test-tubes and colonies of fruit
flies or other insects are convenient for the rapid and cheap
investigation of the potentially carcinogenic and mutagenic effects of
chemicals. The best-known and most widely used of these tests is the
Salmonella mutagenicity test (commonly known as the Ames test).
This involves growing special bacteria in the laboratory and exposing
them to the chemical in question. The test detects mutations in the
bacteria, i.e. it is a test for mutagenic effects. There are a number
of other short-term mutagenic tests or "mutagenicity assays", as they
are often called. These tests are often referred to as being in
vitro. They are thus distinguished from in vivo tests, which use
living tissue such as animals and humans (see Fig. 29).
Many, but not all, chemicals that cause cancer in animals and may
cause cancer in humans are mutagenic.
Figure 29. Short-term mutagenicity tests
(from HSG 34: Fenvalerate)
Fenvalerate did not show any mutagenic or chromosome-damaging
activities in several in vitro and in vivo assays.
2.6.1.5 Reproductive studies
Animal studies to check for the adverse effects of a chemical on
reproduction involve exposing one or both parents to the chemical
being tested, prior to mating, and then observing the effects on any
offspring. Sometimes only a pregnant animal is exposed. Reproductive
effects are classified according to whether the offspring are fewer in
number, lower in birth weight deformed or damaged in some way.
Multigeneration studies are sometimes necessary to detect effects that
may be passed on to future generations (see Fig. 30).
2.6.1.6 Behavioural tests
The effects of chemicals on the behaviour of test animals and on their
ability to learn (e.g. to find their way out of a maze) will often
indicate subtle effects of a chemical on the brain and nervous system
that are missed by other tests. Behavioural tests have been used to
show the effects of exposure to compounds such as organic solvents and
lead.
Figure 30. Reproductive effects (from HSG 34: Fenvalerate)
Fenvalerate is not teratogenic to mice or rabbits at doses up to
50 mg/kg body weight per day. It did not show any toxic effects on
reproductive parameters in a three-generation rat reproduction study
at doses up to 250 mg/kg diet.
2.6.2 Human evidence
2.6.2.1 Case reports
Case reports are accounts, usually by occupational health physicians,
of small numbers of workers or individual workers whose disease
appears to be attributable to workplace exposure. Often the part of a
process at which the chemical may have caused the problem is known,
but not the precise degree of exposure. Much information about
occupational health hazards has initially been found following
examination of case reports.
2.6.2.2 Epidemiological studies
Epidemiological studies investigate the health of a group of people to
establish whether they are affected by the chemical to which they are
exposed at work or in the general environment. These studies involve
comparing the disease outcome of an exposed group of people over time
with that of unexposed persons. Two of the more common methods of
investigation in epidemiology are case-control studies and cohort
studies.
Case-control studies are relatively simple to carry out and
increasingly used to investigate the cause of diseases - especially
rare diseases. They basically compare people with the disease, or
other outcome, under study with a suitable control group unaffected by
the disease or outcome in an attempt to identify the cause.
Cohort studies, also called follow-up or incidence studies, look
at a group of people (a cohort) who are classified into subgroups
according to exposure to a potential cause of disease or outcome.
Differences in exposure (to chemicals, for instance) are examined and
measured and the whole cohort followed up to see how the subsequent
development of the disease, or outcome, differs between the exposed
and unexposed groups (see Fig. 31).
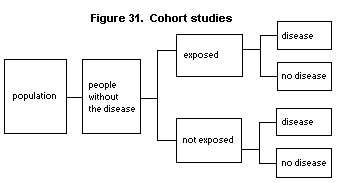 Although epidemiological investigations provide the most dependable
proof that a given chemical has an adverse effect on the health of a
population, they do have several disadvantages. Only a small group of
chemicals in use has been studied in this way because of the expense
involved and the need for good exposure records and a sufficiently
large number of exposed workers to ensure the validity of the
statistical calculations. New chemicals, of course, cannot be studied
in this manner as there will be no history of exposure. In addition,
epidemiological studies may not be able to detect rare events or
pinpoint the role of one particular chemical when workers are exposed
to mixtures of chemicals. But, above all, waiting for the results of
such studies can mean many unnecessary and largely preventable deaths
and illnesses among exposed persons. If information from epidemio-
logical studies is lacking, preventive action should be instigated
based on animal studies (see Fig. 32).
Figure 32. Human studies (from HSG 19: Pentachlorophenol)
Past use of PCP has affected workers producing or using this chemical.
Chloracne, skin irritation and rashes, respiratory disorder,
neurological changes, headaches, nausea, weakness, irritability, and
drowsiness have been documented in exposed workers.
Investigations of biochemical changes in woodworkers with long-term
exposure to PCP have failed to detect consistently significant effects
on major organs, nerves, blood, reproduction, or the immune system.
However, the statistical power of these studies has been limited as a
result of the small sample sizes used. Overall, the body of research
suggests that long-term exposure to levels of PCP encountered in the
workplace is likely to cause borderline effects on some organ systems
and biochemical processes.
Some epidemiological studies from Sweden and the USA have revealed an
association between exposure to mixtures of chlorophenols, especially
2,4,5-T3CP, and the incidences of soft-tissue sarcomas, lymphomas, and
nasal and nasopharyngeal cancers. Other studies have failed to detect
such a relationship. It was not possible to address the effects of
exposure to PCP itself in any of these studies.
The theory behind animal tests is that humans and animals such as
rats, mice or dogs share the same basic biochemistry and bodily
processes. Although there are differences between animals and humans,
the similarities in terms of the way chemicals act on the body are far
greater. Animal tests make it possible to test the toxicity of a
chemical before people are exposed to it. Such results should be used
in health and safety guidelines. This should reduce the occurrence of
unwanted effects of chemicals on human health. Exposing workers or
the general community to a chemical and waiting for human
epidemiological data to appear before warning workers and acting to
protect the general community are now unacceptable. On the other
hand, epidemiological studies will always be essential to monitor the
health of people exposed to chemicals that are already in use and to
check that preventive measures are effective.
2.6.3 Environmental assays
These tests attempt to predict whether a chemical will have unwanted
effects on animals and plants in the environment and whether chemicals
are likely to be present in water supplies and food.
2.7 Assessing hazards, risks and safety
In a general sense, the toxicity of a substance can be defined as the
substance's capacity to harm a living organism. A highly toxic
substance will harm an organism even if only very small amounts are
present in the body. Conversely, a substance of low toxicity will not
produce an effect unless the amount present in the body is very large.
The main factors that must be considered when assessing the toxicity
of a substance include:
* the quantity of the substance absorbed (the dose) by the person
exposed to that chemical;
* the route via which exposure to the chemical occurs (e.g.
inhalation, ingestion, or absorption through the skin);
* the duration of exposure to the chemical and how often that
exposure occurs;
* the type and severity of the injury caused by exposure to the
chemical; and
* whether or not that injury is permanent or reversible, e.g.
cancer is irreversible, although sometimes treatable.
Three other terms are commonly used when the toxicity of a chemical is
discussed: hazard, risk and safety.
2.7.1 Hazard
A hazard can be defined as the set of inherent properties of a
chemical, mixture of chemicals or a process that, under production,
usage or disposal conditions, has the potential to adversely affect
the environment or the organisms it contains. In other words, it is a
source of danger.
2.7.2 Risk
It is important to distinguish risk from hazard. Hazard refers to the
intrinsic properties of a chemical, whereas risk refers to the chance
or probability that the chemical will cause an adverse health effect.
If there is a high risk that a certain chemical will cause cancer in
exposed workers, then it is very likely that some of those workers
will develop cancer. If the risk is low, then it is less likely that
the workers will develop cancer. However, even if the risk of some
health effect is low, the chemical in question is still a hazard.
Depending on the circumstances, a "low risk" may be acceptable to the
people exposed. Determining the "acceptable risk" is part of the
process for setting safety standards.
* Risk assessment involves identification of the hazard
(the chemical of concern, for instance, and its adverse effects,
target populations and conditions of exposure); characterizing
the risk; assessing exposure (by measuring and monitoring); and
estimating the risk. Thus, it consists of identification and
quantification of the risk resulting from a specific use or
occurrence of a chemical, taking into account the possible
harmful effects on individuals of using the chemical in the
manner and amount proposed, and all possible routes of exposure.
* Risk management covers the whole range of actions taken to
prevent, minimize or otherwise control specific risks posed by a
certain chemical or situation. It is based on concepts of safety
and therefore contains elements of policy relating to political,
social and economic factors, as well as engineering and process
control.
2.7.3 Safety
Safety is even more difficult to define than risk or hazard. The
safety of a chemical, in the context of human health, is the extent to
which a chemical may be used in the amount necessary for the intended
purpose, with a minimum risk of adverse health effects. It can also
be defined as a "socially acceptable" level of risk. But it is
usually unclear which part of society is judging the risk. Workers
that are exposed to the risk are likely to be more concerned about the
safety of a chemical than others. Therefore, it is very important to
question statements such as "this chemical is safe" or "there is a
high level of safety when using this chemical". Safety is a
subjective concept.
3. THE PREVENTION AND CONTROL OF CHEMICAL HAZARDS: HOW TO IMPROVE
CHEMICAL SAFETY - CHAPTER 3
This chapter corresponds to Chapter 3 of each HSG (Conclusions and
recommendations), which evaluates the degree of hazard of a chemical
and recommends control measures to limit exposure (see Fig. 33).
Figure 33. Conclusions and recommendations
(from HSG 35: Phosphorus Trichloride)
3.1 Conclusions
Phosphorus trichloride and phosphorus oxychloride are highly reactive
and hazardous corrosive chemicals. Intense exposure to their vapours
may leave a residual restrictive defect in the lungs. Repeated minor
overexposures may result in progressive impairment of lung function
and can be fatal. Apart from spillages that destroy plant and animal
life in the immediate area, the impact of these chemicals on the
environment is negligible.
3.2 Recommendations
Phosphorus trichloride and phosphorus oxychloride should only be used
and handled under the careful supervision of managers who fully
understand the hazards and the good handling and manufacturing
practices necessary to control them. These managers should train
operators, maintenance personnel and contractors about the hazards and
safety procedures.
There are four steps in the prevention and control of workplace
chemical hazards:
* identification of the hazard;
* evaluation of the hazard and risk;
* organization to prevent, control or eliminate the risk; and
* controlling the hazard through specific actions.
Although epidemiological investigations provide the most dependable
proof that a given chemical has an adverse effect on the health of a
population, they do have several disadvantages. Only a small group of
chemicals in use has been studied in this way because of the expense
involved and the need for good exposure records and a sufficiently
large number of exposed workers to ensure the validity of the
statistical calculations. New chemicals, of course, cannot be studied
in this manner as there will be no history of exposure. In addition,
epidemiological studies may not be able to detect rare events or
pinpoint the role of one particular chemical when workers are exposed
to mixtures of chemicals. But, above all, waiting for the results of
such studies can mean many unnecessary and largely preventable deaths
and illnesses among exposed persons. If information from epidemio-
logical studies is lacking, preventive action should be instigated
based on animal studies (see Fig. 32).
Figure 32. Human studies (from HSG 19: Pentachlorophenol)
Past use of PCP has affected workers producing or using this chemical.
Chloracne, skin irritation and rashes, respiratory disorder,
neurological changes, headaches, nausea, weakness, irritability, and
drowsiness have been documented in exposed workers.
Investigations of biochemical changes in woodworkers with long-term
exposure to PCP have failed to detect consistently significant effects
on major organs, nerves, blood, reproduction, or the immune system.
However, the statistical power of these studies has been limited as a
result of the small sample sizes used. Overall, the body of research
suggests that long-term exposure to levels of PCP encountered in the
workplace is likely to cause borderline effects on some organ systems
and biochemical processes.
Some epidemiological studies from Sweden and the USA have revealed an
association between exposure to mixtures of chlorophenols, especially
2,4,5-T3CP, and the incidences of soft-tissue sarcomas, lymphomas, and
nasal and nasopharyngeal cancers. Other studies have failed to detect
such a relationship. It was not possible to address the effects of
exposure to PCP itself in any of these studies.
The theory behind animal tests is that humans and animals such as
rats, mice or dogs share the same basic biochemistry and bodily
processes. Although there are differences between animals and humans,
the similarities in terms of the way chemicals act on the body are far
greater. Animal tests make it possible to test the toxicity of a
chemical before people are exposed to it. Such results should be used
in health and safety guidelines. This should reduce the occurrence of
unwanted effects of chemicals on human health. Exposing workers or
the general community to a chemical and waiting for human
epidemiological data to appear before warning workers and acting to
protect the general community are now unacceptable. On the other
hand, epidemiological studies will always be essential to monitor the
health of people exposed to chemicals that are already in use and to
check that preventive measures are effective.
2.6.3 Environmental assays
These tests attempt to predict whether a chemical will have unwanted
effects on animals and plants in the environment and whether chemicals
are likely to be present in water supplies and food.
2.7 Assessing hazards, risks and safety
In a general sense, the toxicity of a substance can be defined as the
substance's capacity to harm a living organism. A highly toxic
substance will harm an organism even if only very small amounts are
present in the body. Conversely, a substance of low toxicity will not
produce an effect unless the amount present in the body is very large.
The main factors that must be considered when assessing the toxicity
of a substance include:
* the quantity of the substance absorbed (the dose) by the person
exposed to that chemical;
* the route via which exposure to the chemical occurs (e.g.
inhalation, ingestion, or absorption through the skin);
* the duration of exposure to the chemical and how often that
exposure occurs;
* the type and severity of the injury caused by exposure to the
chemical; and
* whether or not that injury is permanent or reversible, e.g.
cancer is irreversible, although sometimes treatable.
Three other terms are commonly used when the toxicity of a chemical is
discussed: hazard, risk and safety.
2.7.1 Hazard
A hazard can be defined as the set of inherent properties of a
chemical, mixture of chemicals or a process that, under production,
usage or disposal conditions, has the potential to adversely affect
the environment or the organisms it contains. In other words, it is a
source of danger.
2.7.2 Risk
It is important to distinguish risk from hazard. Hazard refers to the
intrinsic properties of a chemical, whereas risk refers to the chance
or probability that the chemical will cause an adverse health effect.
If there is a high risk that a certain chemical will cause cancer in
exposed workers, then it is very likely that some of those workers
will develop cancer. If the risk is low, then it is less likely that
the workers will develop cancer. However, even if the risk of some
health effect is low, the chemical in question is still a hazard.
Depending on the circumstances, a "low risk" may be acceptable to the
people exposed. Determining the "acceptable risk" is part of the
process for setting safety standards.
* Risk assessment involves identification of the hazard
(the chemical of concern, for instance, and its adverse effects,
target populations and conditions of exposure); characterizing
the risk; assessing exposure (by measuring and monitoring); and
estimating the risk. Thus, it consists of identification and
quantification of the risk resulting from a specific use or
occurrence of a chemical, taking into account the possible
harmful effects on individuals of using the chemical in the
manner and amount proposed, and all possible routes of exposure.
* Risk management covers the whole range of actions taken to
prevent, minimize or otherwise control specific risks posed by a
certain chemical or situation. It is based on concepts of safety
and therefore contains elements of policy relating to political,
social and economic factors, as well as engineering and process
control.
2.7.3 Safety
Safety is even more difficult to define than risk or hazard. The
safety of a chemical, in the context of human health, is the extent to
which a chemical may be used in the amount necessary for the intended
purpose, with a minimum risk of adverse health effects. It can also
be defined as a "socially acceptable" level of risk. But it is
usually unclear which part of society is judging the risk. Workers
that are exposed to the risk are likely to be more concerned about the
safety of a chemical than others. Therefore, it is very important to
question statements such as "this chemical is safe" or "there is a
high level of safety when using this chemical". Safety is a
subjective concept.
3. THE PREVENTION AND CONTROL OF CHEMICAL HAZARDS: HOW TO IMPROVE
CHEMICAL SAFETY - CHAPTER 3
This chapter corresponds to Chapter 3 of each HSG (Conclusions and
recommendations), which evaluates the degree of hazard of a chemical
and recommends control measures to limit exposure (see Fig. 33).
Figure 33. Conclusions and recommendations
(from HSG 35: Phosphorus Trichloride)
3.1 Conclusions
Phosphorus trichloride and phosphorus oxychloride are highly reactive
and hazardous corrosive chemicals. Intense exposure to their vapours
may leave a residual restrictive defect in the lungs. Repeated minor
overexposures may result in progressive impairment of lung function
and can be fatal. Apart from spillages that destroy plant and animal
life in the immediate area, the impact of these chemicals on the
environment is negligible.
3.2 Recommendations
Phosphorus trichloride and phosphorus oxychloride should only be used
and handled under the careful supervision of managers who fully
understand the hazards and the good handling and manufacturing
practices necessary to control them. These managers should train
operators, maintenance personnel and contractors about the hazards and
safety procedures.
There are four steps in the prevention and control of workplace
chemical hazards:
* identification of the hazard;
* evaluation of the hazard and risk;
* organization to prevent, control or eliminate the risk; and
* controlling the hazard through specific actions.
 3.1 Identification
All workers have the right to know the possible effects of their work
on their health and safety. This includes the right of access to
information about the health effects of chemicals, other substances
and work processes and about procedures for healthy and safe systems
of work.
In some countries, the right to know this information is backed up by
special laws. Under these laws, employers and manufacturers,
suppliers and importers of chemicals must provide clear, detailed
information about the particular chemical, substance or product, its
possible health effects, including the results of animal tests and
surveys of exposed workers (see Chapter 2), and means of protecting
workers from any harmful effects.
These legal rights may apply to each worker or to elected health and
safety representatives or committees. Find out from your employer,
trade union or government occupational health agency if you are
covered by such a law.
3.2 Evaluation
If "right-to-know" laws operate, employers, manufacturers and
suppliers of chemical products are required to:
* produce Material Safety Data Sheets (MSDSs) for all chemicals
used in the workplace;
* label chemical products clearly to indicate their (potentially)
harmful effects, and provide guidance on how to use the products
as safely as possible; and
* instruct workers in the meaning of labels and MSDSs.
Development of a workplace chemical register is one means of
evaluating hazards. This is simply a list of every chemical used in
the workplace. An MSDS should be available for every chemical on the
Register.
Workers should have the right to refuse to work with chemicals for
which full health and safety information is not available. If you
experience difficulty obtaining full information from your employer,
contact your government health or labour ministry, trade union or a
workers' health or environmental organization for help.
Most HSGs available from the IPCS also contain an ICSC or Summary of
Chemical Safety Information for the chemical concerned. Each ICSC is
a brief, internationally reviewed summary of the properties, hazards,
preventive methods and emergency treatment relating to the chemical in
question.
Workers' representatives should be given the opportunity to review
complete health and safety information before any new chemical is
introduced into the workplace. Arrangements for the safe use of new
chemicals should be finalized and put into practice before a chemical
is introduced.
3.3 Safety organization
All workplaces should implement effective safety procedures for
protection against chemical hazards agreed jointly by employer and
workers. In some countries, these agreements will be negotiated as
collective bargaining agreements or health and safety agreements
between management and workers. Sometimes these agreements are
additional to the minimum obligations imposed on employers by
workplace health and safety laws.
The employer and elected worker health and safety representatives
and/or committees in each workplace should participate in the
identification and control of chemical hazards through:
* regular inspections with standard checklists for particular
chemicals and chemical processes;
* investigation of workers' complaints;
* use of accident and sickness records;
* regular surveys of workers' health;
* environmental and biological monitoring;
* assessment of government inspectors'/consultants' reports;
* investigation of the causes of accidents and their prevention;
and
* development of a workplace chemical register.
3.4 Controlling the hazard
The prevention of adverse health effects arising from occupational
exposure to chemicals requires a comprehensive control strategy.
Ideally, exposure should be prevented altogether, i.e. at the source,
through substitution or enclosed processes, for example. If this
cannot be achieved, the level of exposure should be reduced as much as
possible, i.e. during the transmission stage, through ventilation and
the use of protective clothing -- and, certainly, to levels at which
neither health effects nor irritation occurs. A third strategy
comprises measures to counteract the effects of exposure through early
diagnosis of any disease and attempts to prevent the progression of
existing disease, through regular medical monitoring.
The following controls may be used, in descending order of priority:
* substitution of hazardous chemicals or processes with less
hazardous ones;
* engineering controls, e.g. improved ventilation;
* development of safe working procedures;
* reduction of the number of exposed workers;
* reduction of the duration and/or frequency of exposure of
workers;
* use of personal protective equipment, e.g. respirators, goggles;
and
* regular environmental and biological (medical) monitoring or
surveillance to check that the above control methods are proving
effective.
3.4.1 Substitution
The most effective control measure for any hazardous chemical is to
remove it entirely from the workplace and replace it with a less
hazardous chemical. This is crucial for very toxic chemicals,
carcinogens, chemicals that can damage the reproductive system and
sensitizing agents. This approach should, of course, be applied to all
chemical hazards.
An example of substitution is the replacement of the solvents
2-methoxy- and 2-ethoxy-ethanol (commonly used in paints and lacquers)
with the solvent 2-butoxy-ethanol. Both 2-methoxy- and
2-ethoxy-ethanol cause reproductive health effects (including
shrinking of the testicles and birth defects) in animals at low levels
of exposure; 2-butoxy-ethanol has not been found to cause these
effects. In terms of solvent properties, there is no significant
difference between any of these three solvents.
Care must be taken to obtain all available information on proposed
alternative chemicals. Substitutes may turn out to be just as
hazardous as or even more hazardous than the materials they replace.
It is also often possible to substitute safer processes. In this
connection, a process is the sequence of steps involved in the
manufacture or use of a chemical. The manufacture of chemicals
usually entails a series of intermediate stages. The chemicals
produced during these stages (called intermediates) are sometimes
more toxic than the starting or final materials. Whenever possible,
dangerous processes should be substituted to avoid the production of
toxic intermediates.
3.4.2 Engineering controls
3.4.2.1 Total enclosure
If a chemical hazard cannot be removed from the workplace by
substitution, then the next best solution is to physically enclose the
hazard to prevent it from coming into contact with either workers or
the environment. This is known as total enclosure or containment of
a process (see Fig. 35). For example, open tanks from which chemical
vapours can escape into the workplace air can be replaced with closed
tanks with inlet and outlet ports for filling and emptying. Liquids
such as solvents can be transferred by being pumped through sealed
pipes rather than poured in the open air.
3.1 Identification
All workers have the right to know the possible effects of their work
on their health and safety. This includes the right of access to
information about the health effects of chemicals, other substances
and work processes and about procedures for healthy and safe systems
of work.
In some countries, the right to know this information is backed up by
special laws. Under these laws, employers and manufacturers,
suppliers and importers of chemicals must provide clear, detailed
information about the particular chemical, substance or product, its
possible health effects, including the results of animal tests and
surveys of exposed workers (see Chapter 2), and means of protecting
workers from any harmful effects.
These legal rights may apply to each worker or to elected health and
safety representatives or committees. Find out from your employer,
trade union or government occupational health agency if you are
covered by such a law.
3.2 Evaluation
If "right-to-know" laws operate, employers, manufacturers and
suppliers of chemical products are required to:
* produce Material Safety Data Sheets (MSDSs) for all chemicals
used in the workplace;
* label chemical products clearly to indicate their (potentially)
harmful effects, and provide guidance on how to use the products
as safely as possible; and
* instruct workers in the meaning of labels and MSDSs.
Development of a workplace chemical register is one means of
evaluating hazards. This is simply a list of every chemical used in
the workplace. An MSDS should be available for every chemical on the
Register.
Workers should have the right to refuse to work with chemicals for
which full health and safety information is not available. If you
experience difficulty obtaining full information from your employer,
contact your government health or labour ministry, trade union or a
workers' health or environmental organization for help.
Most HSGs available from the IPCS also contain an ICSC or Summary of
Chemical Safety Information for the chemical concerned. Each ICSC is
a brief, internationally reviewed summary of the properties, hazards,
preventive methods and emergency treatment relating to the chemical in
question.
Workers' representatives should be given the opportunity to review
complete health and safety information before any new chemical is
introduced into the workplace. Arrangements for the safe use of new
chemicals should be finalized and put into practice before a chemical
is introduced.
3.3 Safety organization
All workplaces should implement effective safety procedures for
protection against chemical hazards agreed jointly by employer and
workers. In some countries, these agreements will be negotiated as
collective bargaining agreements or health and safety agreements
between management and workers. Sometimes these agreements are
additional to the minimum obligations imposed on employers by
workplace health and safety laws.
The employer and elected worker health and safety representatives
and/or committees in each workplace should participate in the
identification and control of chemical hazards through:
* regular inspections with standard checklists for particular
chemicals and chemical processes;
* investigation of workers' complaints;
* use of accident and sickness records;
* regular surveys of workers' health;
* environmental and biological monitoring;
* assessment of government inspectors'/consultants' reports;
* investigation of the causes of accidents and their prevention;
and
* development of a workplace chemical register.
3.4 Controlling the hazard
The prevention of adverse health effects arising from occupational
exposure to chemicals requires a comprehensive control strategy.
Ideally, exposure should be prevented altogether, i.e. at the source,
through substitution or enclosed processes, for example. If this
cannot be achieved, the level of exposure should be reduced as much as
possible, i.e. during the transmission stage, through ventilation and
the use of protective clothing -- and, certainly, to levels at which
neither health effects nor irritation occurs. A third strategy
comprises measures to counteract the effects of exposure through early
diagnosis of any disease and attempts to prevent the progression of
existing disease, through regular medical monitoring.
The following controls may be used, in descending order of priority:
* substitution of hazardous chemicals or processes with less
hazardous ones;
* engineering controls, e.g. improved ventilation;
* development of safe working procedures;
* reduction of the number of exposed workers;
* reduction of the duration and/or frequency of exposure of
workers;
* use of personal protective equipment, e.g. respirators, goggles;
and
* regular environmental and biological (medical) monitoring or
surveillance to check that the above control methods are proving
effective.
3.4.1 Substitution
The most effective control measure for any hazardous chemical is to
remove it entirely from the workplace and replace it with a less
hazardous chemical. This is crucial for very toxic chemicals,
carcinogens, chemicals that can damage the reproductive system and
sensitizing agents. This approach should, of course, be applied to all
chemical hazards.
An example of substitution is the replacement of the solvents
2-methoxy- and 2-ethoxy-ethanol (commonly used in paints and lacquers)
with the solvent 2-butoxy-ethanol. Both 2-methoxy- and
2-ethoxy-ethanol cause reproductive health effects (including
shrinking of the testicles and birth defects) in animals at low levels
of exposure; 2-butoxy-ethanol has not been found to cause these
effects. In terms of solvent properties, there is no significant
difference between any of these three solvents.
Care must be taken to obtain all available information on proposed
alternative chemicals. Substitutes may turn out to be just as
hazardous as or even more hazardous than the materials they replace.
It is also often possible to substitute safer processes. In this
connection, a process is the sequence of steps involved in the
manufacture or use of a chemical. The manufacture of chemicals
usually entails a series of intermediate stages. The chemicals
produced during these stages (called intermediates) are sometimes
more toxic than the starting or final materials. Whenever possible,
dangerous processes should be substituted to avoid the production of
toxic intermediates.
3.4.2 Engineering controls
3.4.2.1 Total enclosure
If a chemical hazard cannot be removed from the workplace by
substitution, then the next best solution is to physically enclose the
hazard to prevent it from coming into contact with either workers or
the environment. This is known as total enclosure or containment of
a process (see Fig. 35). For example, open tanks from which chemical
vapours can escape into the workplace air can be replaced with closed
tanks with inlet and outlet ports for filling and emptying. Liquids
such as solvents can be transferred by being pumped through sealed
pipes rather than poured in the open air.
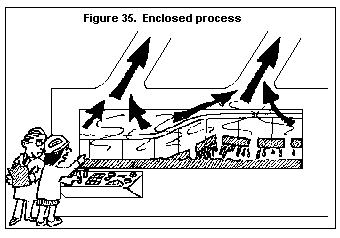 3.4.2.2 Ventilation
Ventilation systems are one means of removing contaminated air from
the workplace. There are two general types of ventilation:
* dilution or general ventilation; and
* local exhaust ventilation.
Dilution ventilation is simply the process whereby clean air is mixed
with contaminated air. The concentration of the airborne
contaminant is thus reduced, although the workplace air will still
contain some of the contaminant.
A simple dilution ventilation system consists of two major components:
a source of clean air and an exhaust fan for removing the contaminated
air. The ventilation system can be totally passive, which means that
the exhaust is a chimney or open vent in the ceiling from which the
dirty air is expelled, and the source of clean air an open inlet in
one of the workplace walls (see Fig. 36). This is the most basic, i.e.
least effective, system. An improvement on this system would be a fan
at the exhaust forcing the dirty air out. An even better system would
have both an exhaust fan and an inlet fan.
Dilution ventilation is rarely an adequate means of safeguarding
health in the workplace, as:
* it does not prevent the release of a contaminant into the
workplace air;
* it is ineffective against dusts, metal particles and metal fumes
that are too heavy to be removed by gentle air movement;
* it does not provide sufficient protection against highly toxic
chemical vapours and gases, carcinogens or chemicals causing
reproductive effects;
* it is ineffective against "surges" of gases and vapours or
irregular emissions of contaminants; and
* workers very close to a contaminant source may still experience
very high levels of exposure.
3.4.2.2 Ventilation
Ventilation systems are one means of removing contaminated air from
the workplace. There are two general types of ventilation:
* dilution or general ventilation; and
* local exhaust ventilation.
Dilution ventilation is simply the process whereby clean air is mixed
with contaminated air. The concentration of the airborne
contaminant is thus reduced, although the workplace air will still
contain some of the contaminant.
A simple dilution ventilation system consists of two major components:
a source of clean air and an exhaust fan for removing the contaminated
air. The ventilation system can be totally passive, which means that
the exhaust is a chimney or open vent in the ceiling from which the
dirty air is expelled, and the source of clean air an open inlet in
one of the workplace walls (see Fig. 36). This is the most basic, i.e.
least effective, system. An improvement on this system would be a fan
at the exhaust forcing the dirty air out. An even better system would
have both an exhaust fan and an inlet fan.
Dilution ventilation is rarely an adequate means of safeguarding
health in the workplace, as:
* it does not prevent the release of a contaminant into the
workplace air;
* it is ineffective against dusts, metal particles and metal fumes
that are too heavy to be removed by gentle air movement;
* it does not provide sufficient protection against highly toxic
chemical vapours and gases, carcinogens or chemicals causing
reproductive effects;
* it is ineffective against "surges" of gases and vapours or
irregular emissions of contaminants; and
* workers very close to a contaminant source may still experience
very high levels of exposure.
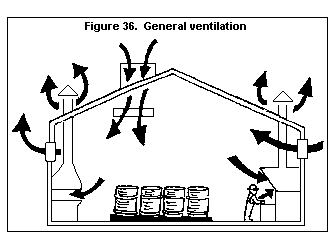 Local exhaust ventilation systems remove airborne contaminants near
their source. Such systems must be properly designed and maintained
and must work effectively so that workers will not inhale contaminated
air. For any work process that uses or produces moderately toxic or
highly toxic chemicals that cannot be effectively enclosed, a local
exhaust system is essential (see Fig. 37).
A local exhaust ventilation system contains four major parts: a hood,
ducts, an air cleaner and a fan. Contaminants are drawn in
through the hood, transported inside the ducts, removed by an air
cleaner and sucked through the system by a fan. A local exhaust
system always needs a suitable inlet of clean replacement air.
Some of the advantages of local exhaust ventilation include:
* the capture of hazardous contaminants at their source, and the
removal of contaminants from the workplace;
* the capacity to handle all types of contaminants, including
dusts, metal particles and metal fumes; and
* the capacity to protect workers close to the contaminant's
source.
To be effective, however, a local exhaust ventilation system must be
carefully designed for the specific process, and the appropriate
parts selected. The hood should be close enough to capture all
contamination and pull it away from, not past, the worker's breathing
zone.
Moreover, local exhaust systems require regular cleaning, inspection
and maintenance (e.g., regular filter changing, checking of the
airflow rate with an anemometer, checking for leaks or holes in the
ducting and checking for blockages). It should be noted that exhaust
air will be contaminated and may endanger the health of people outside
the workplace.
3.4.3 Safe working procedures
Workers involved in the manufacture or use of chemicals are usually
given a list of instructions by their employer on how to weigh,
measure and combine the various chemicals used in their workplace.
Often these instructions tell the worker what to do to make the
product but carry no information on the hazards or risks involved or
on ways to protect against the chemical hazards present, e.g. how to
operate ventilation systems or when to change filters on breathing
masks.
Local exhaust ventilation systems remove airborne contaminants near
their source. Such systems must be properly designed and maintained
and must work effectively so that workers will not inhale contaminated
air. For any work process that uses or produces moderately toxic or
highly toxic chemicals that cannot be effectively enclosed, a local
exhaust system is essential (see Fig. 37).
A local exhaust ventilation system contains four major parts: a hood,
ducts, an air cleaner and a fan. Contaminants are drawn in
through the hood, transported inside the ducts, removed by an air
cleaner and sucked through the system by a fan. A local exhaust
system always needs a suitable inlet of clean replacement air.
Some of the advantages of local exhaust ventilation include:
* the capture of hazardous contaminants at their source, and the
removal of contaminants from the workplace;
* the capacity to handle all types of contaminants, including
dusts, metal particles and metal fumes; and
* the capacity to protect workers close to the contaminant's
source.
To be effective, however, a local exhaust ventilation system must be
carefully designed for the specific process, and the appropriate
parts selected. The hood should be close enough to capture all
contamination and pull it away from, not past, the worker's breathing
zone.
Moreover, local exhaust systems require regular cleaning, inspection
and maintenance (e.g., regular filter changing, checking of the
airflow rate with an anemometer, checking for leaks or holes in the
ducting and checking for blockages). It should be noted that exhaust
air will be contaminated and may endanger the health of people outside
the workplace.
3.4.3 Safe working procedures
Workers involved in the manufacture or use of chemicals are usually
given a list of instructions by their employer on how to weigh,
measure and combine the various chemicals used in their workplace.
Often these instructions tell the worker what to do to make the
product but carry no information on the hazards or risks involved or
on ways to protect against the chemical hazards present, e.g. how to
operate ventilation systems or when to change filters on breathing
masks.
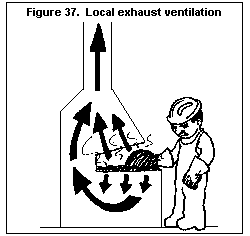 A safe working procedure should include instructions for each step in
a process, as well as the steps necessary for workers to protect
themselves from the health effects of the chemicals used in that
process.
A "Code of Practice" is a more general description of safety measures
to be adhered to during a particular type of process or by a
particular industry (see Fig. 38). The International Labour
Organisation (ILO) has developed codes of practice for some processes,
and some countries have national codes of practice. For shop-floor
use, a relevant code of practice should include:
* a list of all chemicals used in that process;
* a summary of the health effects of those chemicals and the way in
which exposure to, and absorption of, the chemicals may occur;
* an outline of the equipment necessary to complete a task
correctly and safely;
* the procedures for operating that equipment correctly. For
example, if a chemical has to be heated, information should be
included on the maximum temperature, the consequences of heating
the chemical too much (the risk of explosion or fire) and the
ventilation system required to protect workers and the general
public against fumes or vapours of the heated chemical;
* a description of any personal protective equipment that may be
necessary, such as face masks or gloves, together with the
specifications and standards these must meet and information on
exactly how and when the equipment should be used; and
* details of how often and what environmental and biological
monitoring should be performed and of procedures for action if
"trigger" levels (see below) are exceeded. For example, carbon
monoxide is a highly toxic but colourless and odourless gas
produced by many industrial processes that involve heating or
burning (e.g. blast furnaces and coke ovens). The carbon
monoxide level in the workplace air should be monitored
continuously while the process is in operation. When the carbon
monoxide level reaches a set percentage of the exposure limit
(see section 7.1 of this manual), an alarm should sound and
workers should leave the workplace until the carbon monoxide
level has fallen to a safe level. This "action" or "trigger"
level must be a small fraction of the exposure limit (around 10%)
to allow room for error in measurement and delay in alerting
workers to the hazard.
A safe working procedure should include instructions for each step in
a process, as well as the steps necessary for workers to protect
themselves from the health effects of the chemicals used in that
process.
A "Code of Practice" is a more general description of safety measures
to be adhered to during a particular type of process or by a
particular industry (see Fig. 38). The International Labour
Organisation (ILO) has developed codes of practice for some processes,
and some countries have national codes of practice. For shop-floor
use, a relevant code of practice should include:
* a list of all chemicals used in that process;
* a summary of the health effects of those chemicals and the way in
which exposure to, and absorption of, the chemicals may occur;
* an outline of the equipment necessary to complete a task
correctly and safely;
* the procedures for operating that equipment correctly. For
example, if a chemical has to be heated, information should be
included on the maximum temperature, the consequences of heating
the chemical too much (the risk of explosion or fire) and the
ventilation system required to protect workers and the general
public against fumes or vapours of the heated chemical;
* a description of any personal protective equipment that may be
necessary, such as face masks or gloves, together with the
specifications and standards these must meet and information on
exactly how and when the equipment should be used; and
* details of how often and what environmental and biological
monitoring should be performed and of procedures for action if
"trigger" levels (see below) are exceeded. For example, carbon
monoxide is a highly toxic but colourless and odourless gas
produced by many industrial processes that involve heating or
burning (e.g. blast furnaces and coke ovens). The carbon
monoxide level in the workplace air should be monitored
continuously while the process is in operation. When the carbon
monoxide level reaches a set percentage of the exposure limit
(see section 7.1 of this manual), an alarm should sound and
workers should leave the workplace until the carbon monoxide
level has fallen to a safe level. This "action" or "trigger"
level must be a small fraction of the exposure limit (around 10%)
to allow room for error in measurement and delay in alerting
workers to the hazard.
 Some work procedures require clearance or a "permit-to-work"
certification. The latter is essentially a document that sets out the
work to be done, the hazards involved and the precautions to be taken,
such as testing the working area for chemicals before entering. It
predetermines a safe drill and is a record that all foreseeable
hazards have been considered in advance and precautions taken. It is
of particular importance with respect to maintenance workers, for the
repair of chemical-bearing lines, for work in confined spaces and so
on.
A permit-to-work system does not in itself make a job safe but is
designed to protect the workers involved. Workers should therefore be
well trained in the accompanying procedures.
3.4.4 Reducing the number of exposed workers and their duration
of exposure
Only those directly involved with a job or chemical process should be
exposed to any chemical hazards present. For example, if chemicals
that give off vapours are being heated, then only those workers needed
for the job should be in the area. Maintenance workers, electricians,
cleaners or other workers should do their work when the chemical
hazard is not present.
The risk to maintenance workers is often ignored or seriously
underestimated when planning chemical control measures. Maintenance
workers may be more highly exposed because normal control procedures
are not geared to their work; these procedures may not be operational
at times when they do their work; and they may be in closer proximity
to hazardous plants and processes than other workers when they do
repair or maintenance work. Specific provisions for the protection of
maintenance workers must be included in any chemical safety procedure.
In certain circumstances, the reduction of the duration or the
frequency of exposure of workers may be achieved by job rotation.
However, it is never acceptable to simply expose more workers less
often to unacceptably high levels as an alternative to reducing
exposure levels.
3.4.5 Personal protective equipment
3.4.5.1 Principles
Whereas engineering controls place a barrier around a hazardous
process or chemical, personal protective equipment is often used to
create a "barrier" around a worker to prevent chemicals already
released into the workplace air from reaching the worker's lungs,
skin, etc. The use of personal protective equipment should be
resorted to only after the methods outlined above -- substitution and
engineering controls -- have first been considered and acted upon.
Personal protective equipment is rated as the least effective method
of protection and is often uncomfortable or difficult to work with.
Personal protective equipment against chemicals includes:
* face shields, goggles and safety glasses;
* gloves;
* rubber boots;
* plastic or rubber overalls and aprons;
* hard hats;
* respirators; and
* dust masks.
A personal protective equipment programme requires the following steps
and resources:
* the correct equipment -- e.g. a respirator designed to protect
against dust is useless if the hazardous chemical is present as a
gas; and many solvents can rapidly penetrate natural rubber
gloves;
* a thorough training programme for workers who are required to use
the equipment, with follow-up training at regular intervals;
* tests to ensure that equipment fits correctly. This is
particularly important for face masks and respirators;
* a regular equipment maintenance programme. This includes regular
cleaning of equipment, inspection to ensure that it is operating
correctly and regular replacement of items such as gloves or
disposable parts such as respirator filters. Respirator filters
should be replaced at regular time intervals rather than only
when they have become clogged; and
* a personal set of equipment for each worker and a secure and
clean place in which to store it.
In some situations, the use of personal protective equipment is
unavoidable. This applies particularly to eye goggles, face shields,
boots and hard hats. Because these items are designed to protect the
worker against accidents and unexpected exposures, they must be worn
at all times. For some jobs, such as pesticide spraying by hand, no
other means of protection is possible, in which case protective
clothing, gloves and respirator masks must be worn.
Other tasks that may be necessary from time to time, such as the
cleaning of a ventilation system, will often require the wearing of
personal protective equipment such as respirators. When the chemicals
are particularly dangerous or concentrations very high, air-supplied
respirators are required.
Chapter 4 of each HSG -- and also the ICSC or Summary of Chemical
Safety Information -- provides general advice on types of personal
protective equipment that are suitable for the chemical being used.
Workers should also seek information from the manufacturer of the
chemical about suitable protective equipment. Often this information
is included in the chemical manufacturer's MSDS (see Chapter 8 on
"Getting Further Information").
3.4.5.2 Protective clothing
Not all protective clothing protects its wearer from all chemicals.
Gloves, aprons and overalls, for instance, must be made from a
material that is not penetrable by the chemical in use at the time.
Otherwise, chemical solvents may pass unnoticed through the material,
emerging in an invisible but hazardous form inside gloves or other
protective clothing. If this occurs, the chemical will come into
contact with the skin and could prove as dangerous as would be the
case if the individual were working without any protective clothing.
3.4.5.3 Gloves
General advice about choosing protective gloves includes the
following:
* For maximum protection against skin absorption, gloves should be
of suitable material, fit properly and be in good condition.
* It is important to get information from the suppliers of
protective equipment about what equipment has been tested and
found to be resistant to the particular chemicals you are using.
Ask specifically about the chemical's breakthrough time, i.e. how
long you can wear the gloves before the chemical penetrates
through to your hands. Some gloves, especially PVA (polyvinyl
alcohol) gloves, actually disintegrate on contact with water.
* Only use gloves once when working with chemicals that have a
quick breakthrough time. Remember, the chemical may still be
penetrating the gloves after you have removed them.
* Cotton flock or fabric lining in gloves helps absorb perspiration
and also partly insulates the hands against heat or cold.
* Double gloving is recommended if little is known about the
chemical in use.
* Unless gloves are thoroughly cleaned when they are taken off, any
chemical picked up on the glove lining could come into contact
with the skin.
* Dusts from recently activated resins are almost as irritating and
harmful to the skin as the original liquid; gloves should be worn
even when handling the resultant laminate.
* Some chemicals in combination will penetrate gloves more quickly
than they would separately, e.g. pentane and trichloroethylene.
* Solvents may also carry other substances through the gloves; e.g.
xylene carries organophosphates through PVC (polyvinyl chloride)
gloves.
* Search for a listing of gloves available in your country and
information on their suitability for different jobs. The local
safety inspectorate may have such a list.
3.4.5.4 Do you need a respirator?
There are many different types of respirators available. This section
will help you to ask the right questions to make sure that you are
being provided with the correct type of respirator for your work
process.
* What are you exposed to?
This will depend on the type of hazard, i.e. whether the contaminant
is a gas, vapour, dust, mist or fume; whether there is a potential for
oxygen deficiency (in the USA, if the oxygen level is likely to fall
below 17%, or 19.5% in some countries, fresh air must be provided); or
whether the atmosphere is immediately dangerous to life or health,
e.g. high concentrations of cyanide fumes.
* What concentration of the chemical are you exposed to?
It is important to know how much of the chemical is present in the air
you are breathing, because different types of respirators have
different levels of efficiency. You should know what concentration of
the chemical you are exposed to and the concentration that is
classified as acceptable (see section 7.1 of this Manual on "exposure
limits" for reasons why even these limits may not be safe inside a
respirator).
Once you know these two concentrations of the chemical, you will be
able to work out the amount of protection you need from a respirator.
For example, if the acceptable level of exposure for Solvent B is 10
ppm and the concentration in the workplace is 100 ppm, you will need
an air filter with a "nominal protection factor" of 100/10 = 10.
* Does the chemical have any warning signs?
You will need to know how long you can wear the respirator before you
have to change the filter or cartridge/canister or before the air
supply runs out in a self-contained breathing apparatus. You will
also need to know whether the chemical has a particular smell or is
irritating (either will serve as a warning if the chemical begins to
break through the absorbent material in the respirator). This is
particularly important when air-purifying respirators are used against
gases and vapours.
* Is there any other chemical present that you also need protection
against?
If a mixture of gases or vapours or particulates is present, this will
influence the choice of respirator.
* Can the chemical be absorbed through the skin?
If the answer is "Yes", then the rest of the body, including the face,
must be protected and a full-face respirator worn.
* Can the chemical irritate the eyes at the expected concentration?
If "Yes", then the eyes must be protected -- either separately through
the use of goggles or by use of a full-face respirator.
A SUITABLE RESPIRATOR CAN BE CHOSEN ONLY AFTER THESE QUESTIONS HAVE
BEEN ANSWERED.
3.4.5.5 Types of respirators
I Air-purifying respirators -- The air is drawn in through an
absorbent material in the case of gaseous contaminants or through a
filter in the case of particulate contaminants.
a) Air-purifying respirators for gaseous contaminants
These consist of a face-piece with a cartridge or canister to remove
the gas or vapour. The face-piece can be quarter-mask, covering the
nose and mouth but not fitting under the chin; half-mask, covering the
nose and mouth and fitting under the chin; or full-face mask, covering
the face from the hairline to under the chin. The full-face mask
offers greater protection than half- or quarter-masks, as the mask
will fit the face more closely, giving a better seal.
The cartridge or canister containing the material that removes the gas
or vapour is fitted to the front of the face-piece and air drawn
through it (see Fig. 39). A cartridge offers less protection than a
canister. A cartridge can be used to protect against gases or vapours
of relatively low toxicity. A canister will remove limited
concentrations of certain toxic gases or vapours for a specified time,
after which the canister must be replaced.
Some work procedures require clearance or a "permit-to-work"
certification. The latter is essentially a document that sets out the
work to be done, the hazards involved and the precautions to be taken,
such as testing the working area for chemicals before entering. It
predetermines a safe drill and is a record that all foreseeable
hazards have been considered in advance and precautions taken. It is
of particular importance with respect to maintenance workers, for the
repair of chemical-bearing lines, for work in confined spaces and so
on.
A permit-to-work system does not in itself make a job safe but is
designed to protect the workers involved. Workers should therefore be
well trained in the accompanying procedures.
3.4.4 Reducing the number of exposed workers and their duration
of exposure
Only those directly involved with a job or chemical process should be
exposed to any chemical hazards present. For example, if chemicals
that give off vapours are being heated, then only those workers needed
for the job should be in the area. Maintenance workers, electricians,
cleaners or other workers should do their work when the chemical
hazard is not present.
The risk to maintenance workers is often ignored or seriously
underestimated when planning chemical control measures. Maintenance
workers may be more highly exposed because normal control procedures
are not geared to their work; these procedures may not be operational
at times when they do their work; and they may be in closer proximity
to hazardous plants and processes than other workers when they do
repair or maintenance work. Specific provisions for the protection of
maintenance workers must be included in any chemical safety procedure.
In certain circumstances, the reduction of the duration or the
frequency of exposure of workers may be achieved by job rotation.
However, it is never acceptable to simply expose more workers less
often to unacceptably high levels as an alternative to reducing
exposure levels.
3.4.5 Personal protective equipment
3.4.5.1 Principles
Whereas engineering controls place a barrier around a hazardous
process or chemical, personal protective equipment is often used to
create a "barrier" around a worker to prevent chemicals already
released into the workplace air from reaching the worker's lungs,
skin, etc. The use of personal protective equipment should be
resorted to only after the methods outlined above -- substitution and
engineering controls -- have first been considered and acted upon.
Personal protective equipment is rated as the least effective method
of protection and is often uncomfortable or difficult to work with.
Personal protective equipment against chemicals includes:
* face shields, goggles and safety glasses;
* gloves;
* rubber boots;
* plastic or rubber overalls and aprons;
* hard hats;
* respirators; and
* dust masks.
A personal protective equipment programme requires the following steps
and resources:
* the correct equipment -- e.g. a respirator designed to protect
against dust is useless if the hazardous chemical is present as a
gas; and many solvents can rapidly penetrate natural rubber
gloves;
* a thorough training programme for workers who are required to use
the equipment, with follow-up training at regular intervals;
* tests to ensure that equipment fits correctly. This is
particularly important for face masks and respirators;
* a regular equipment maintenance programme. This includes regular
cleaning of equipment, inspection to ensure that it is operating
correctly and regular replacement of items such as gloves or
disposable parts such as respirator filters. Respirator filters
should be replaced at regular time intervals rather than only
when they have become clogged; and
* a personal set of equipment for each worker and a secure and
clean place in which to store it.
In some situations, the use of personal protective equipment is
unavoidable. This applies particularly to eye goggles, face shields,
boots and hard hats. Because these items are designed to protect the
worker against accidents and unexpected exposures, they must be worn
at all times. For some jobs, such as pesticide spraying by hand, no
other means of protection is possible, in which case protective
clothing, gloves and respirator masks must be worn.
Other tasks that may be necessary from time to time, such as the
cleaning of a ventilation system, will often require the wearing of
personal protective equipment such as respirators. When the chemicals
are particularly dangerous or concentrations very high, air-supplied
respirators are required.
Chapter 4 of each HSG -- and also the ICSC or Summary of Chemical
Safety Information -- provides general advice on types of personal
protective equipment that are suitable for the chemical being used.
Workers should also seek information from the manufacturer of the
chemical about suitable protective equipment. Often this information
is included in the chemical manufacturer's MSDS (see Chapter 8 on
"Getting Further Information").
3.4.5.2 Protective clothing
Not all protective clothing protects its wearer from all chemicals.
Gloves, aprons and overalls, for instance, must be made from a
material that is not penetrable by the chemical in use at the time.
Otherwise, chemical solvents may pass unnoticed through the material,
emerging in an invisible but hazardous form inside gloves or other
protective clothing. If this occurs, the chemical will come into
contact with the skin and could prove as dangerous as would be the
case if the individual were working without any protective clothing.
3.4.5.3 Gloves
General advice about choosing protective gloves includes the
following:
* For maximum protection against skin absorption, gloves should be
of suitable material, fit properly and be in good condition.
* It is important to get information from the suppliers of
protective equipment about what equipment has been tested and
found to be resistant to the particular chemicals you are using.
Ask specifically about the chemical's breakthrough time, i.e. how
long you can wear the gloves before the chemical penetrates
through to your hands. Some gloves, especially PVA (polyvinyl
alcohol) gloves, actually disintegrate on contact with water.
* Only use gloves once when working with chemicals that have a
quick breakthrough time. Remember, the chemical may still be
penetrating the gloves after you have removed them.
* Cotton flock or fabric lining in gloves helps absorb perspiration
and also partly insulates the hands against heat or cold.
* Double gloving is recommended if little is known about the
chemical in use.
* Unless gloves are thoroughly cleaned when they are taken off, any
chemical picked up on the glove lining could come into contact
with the skin.
* Dusts from recently activated resins are almost as irritating and
harmful to the skin as the original liquid; gloves should be worn
even when handling the resultant laminate.
* Some chemicals in combination will penetrate gloves more quickly
than they would separately, e.g. pentane and trichloroethylene.
* Solvents may also carry other substances through the gloves; e.g.
xylene carries organophosphates through PVC (polyvinyl chloride)
gloves.
* Search for a listing of gloves available in your country and
information on their suitability for different jobs. The local
safety inspectorate may have such a list.
3.4.5.4 Do you need a respirator?
There are many different types of respirators available. This section
will help you to ask the right questions to make sure that you are
being provided with the correct type of respirator for your work
process.
* What are you exposed to?
This will depend on the type of hazard, i.e. whether the contaminant
is a gas, vapour, dust, mist or fume; whether there is a potential for
oxygen deficiency (in the USA, if the oxygen level is likely to fall
below 17%, or 19.5% in some countries, fresh air must be provided); or
whether the atmosphere is immediately dangerous to life or health,
e.g. high concentrations of cyanide fumes.
* What concentration of the chemical are you exposed to?
It is important to know how much of the chemical is present in the air
you are breathing, because different types of respirators have
different levels of efficiency. You should know what concentration of
the chemical you are exposed to and the concentration that is
classified as acceptable (see section 7.1 of this Manual on "exposure
limits" for reasons why even these limits may not be safe inside a
respirator).
Once you know these two concentrations of the chemical, you will be
able to work out the amount of protection you need from a respirator.
For example, if the acceptable level of exposure for Solvent B is 10
ppm and the concentration in the workplace is 100 ppm, you will need
an air filter with a "nominal protection factor" of 100/10 = 10.
* Does the chemical have any warning signs?
You will need to know how long you can wear the respirator before you
have to change the filter or cartridge/canister or before the air
supply runs out in a self-contained breathing apparatus. You will
also need to know whether the chemical has a particular smell or is
irritating (either will serve as a warning if the chemical begins to
break through the absorbent material in the respirator). This is
particularly important when air-purifying respirators are used against
gases and vapours.
* Is there any other chemical present that you also need protection
against?
If a mixture of gases or vapours or particulates is present, this will
influence the choice of respirator.
* Can the chemical be absorbed through the skin?
If the answer is "Yes", then the rest of the body, including the face,
must be protected and a full-face respirator worn.
* Can the chemical irritate the eyes at the expected concentration?
If "Yes", then the eyes must be protected -- either separately through
the use of goggles or by use of a full-face respirator.
A SUITABLE RESPIRATOR CAN BE CHOSEN ONLY AFTER THESE QUESTIONS HAVE
BEEN ANSWERED.
3.4.5.5 Types of respirators
I Air-purifying respirators -- The air is drawn in through an
absorbent material in the case of gaseous contaminants or through a
filter in the case of particulate contaminants.
a) Air-purifying respirators for gaseous contaminants
These consist of a face-piece with a cartridge or canister to remove
the gas or vapour. The face-piece can be quarter-mask, covering the
nose and mouth but not fitting under the chin; half-mask, covering the
nose and mouth and fitting under the chin; or full-face mask, covering
the face from the hairline to under the chin. The full-face mask
offers greater protection than half- or quarter-masks, as the mask
will fit the face more closely, giving a better seal.
The cartridge or canister containing the material that removes the gas
or vapour is fitted to the front of the face-piece and air drawn
through it (see Fig. 39). A cartridge offers less protection than a
canister. A cartridge can be used to protect against gases or vapours
of relatively low toxicity. A canister will remove limited
concentrations of certain toxic gases or vapours for a specified time,
after which the canister must be replaced.
 b) Air-purifying respirators for particulate contaminants
These consist of a face-piece and a filter. The face-piece can be a
quarter-, half- or full-face mask. The filters are classified as:
* low efficiency -- used against dusts and mists of low toxicity,
in low concentrations;
* medium efficiency -- used against dust and fume concentrations up
to 10 times the exposure standard, except where the dust or fume
is highly toxic, e.g. hexavalent chromium; and
* high efficiency -- used, with a full face-piece respirator, where
toxicity or concentration of a chemical is high.
A combined gas and dust respirator should be used where both gases/
vapours and dusts are present.
c) Air-purifying respirators for both gaseous and particulate
contaminants
These respirators can be used where a combined hazard is present, e.g.
paint spraying or spraying with pesticides. Some particulate filters
will also absorb some gases/vapours, but you must insist that evidence
is provided that they will protect against both. The filters should
be marked to show what they protect against.
The length of time a cartridge or canister can be used depends on the
concentration of gas, vapour or particulate and the activity of the
wearer. If you are doing hard physical work, you will breathe more
deeply and the canister will become saturated sooner.
Air-purifying respirators should not be used in a confined space, or
there might be a shortage of oxygen.
Face masks that do not fit the worker or that are worn out or blocked
by particulates can result in serious injury, as the wearer believes
him/herself protected and is therefore unaware that he or she is
exposed. The presence of facial hair can also mean that the face mask
does not fit perfectly, with the result that contaminated air could
enter between the mask and the skin.
II Supplied air units -- These respirators supply clean air directly
into the respirator, rather than filtering out the chemical from the
air in the work environment (see Fig. 40). There are two types:
a) Supplied air respirator -- Air is supplied through an air supply
line or hose. In the case of an air supply line, the air comes
from a compressed air source through a high-pressure hose. The air
can be supplied to a half- or full-face mask, helmet, hood or complete
suit. These respirators can be used against particulates, gases or
vapours. They provide a high degree of protection but cannot be used
in atmospheres immediately hazardous to life or health, as any fault
with the air supply or air line could render the worker highly
vulnerable.
For hose masks, air is supplied from a clean-air area through a
large-diameter hose to the face-piece. Compressed air is not used.
The air can be either pumped in by a hand- or motor-operated blower or
simply drawn in by the wearer. These respirators always have a
full-face mask, which must fit the face well. They should also not be
used in atmospheres immediately dangerous to life or health.
b) Air-purifying respirators for particulate contaminants
These consist of a face-piece and a filter. The face-piece can be a
quarter-, half- or full-face mask. The filters are classified as:
* low efficiency -- used against dusts and mists of low toxicity,
in low concentrations;
* medium efficiency -- used against dust and fume concentrations up
to 10 times the exposure standard, except where the dust or fume
is highly toxic, e.g. hexavalent chromium; and
* high efficiency -- used, with a full face-piece respirator, where
toxicity or concentration of a chemical is high.
A combined gas and dust respirator should be used where both gases/
vapours and dusts are present.
c) Air-purifying respirators for both gaseous and particulate
contaminants
These respirators can be used where a combined hazard is present, e.g.
paint spraying or spraying with pesticides. Some particulate filters
will also absorb some gases/vapours, but you must insist that evidence
is provided that they will protect against both. The filters should
be marked to show what they protect against.
The length of time a cartridge or canister can be used depends on the
concentration of gas, vapour or particulate and the activity of the
wearer. If you are doing hard physical work, you will breathe more
deeply and the canister will become saturated sooner.
Air-purifying respirators should not be used in a confined space, or
there might be a shortage of oxygen.
Face masks that do not fit the worker or that are worn out or blocked
by particulates can result in serious injury, as the wearer believes
him/herself protected and is therefore unaware that he or she is
exposed. The presence of facial hair can also mean that the face mask
does not fit perfectly, with the result that contaminated air could
enter between the mask and the skin.
II Supplied air units -- These respirators supply clean air directly
into the respirator, rather than filtering out the chemical from the
air in the work environment (see Fig. 40). There are two types:
a) Supplied air respirator -- Air is supplied through an air supply
line or hose. In the case of an air supply line, the air comes
from a compressed air source through a high-pressure hose. The air
can be supplied to a half- or full-face mask, helmet, hood or complete
suit. These respirators can be used against particulates, gases or
vapours. They provide a high degree of protection but cannot be used
in atmospheres immediately hazardous to life or health, as any fault
with the air supply or air line could render the worker highly
vulnerable.
For hose masks, air is supplied from a clean-air area through a
large-diameter hose to the face-piece. Compressed air is not used.
The air can be either pumped in by a hand- or motor-operated blower or
simply drawn in by the wearer. These respirators always have a
full-face mask, which must fit the face well. They should also not be
used in atmospheres immediately dangerous to life or health.
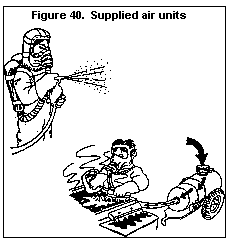 b) Self-contained breathing apparatus -- This allows the wearer to
carry a respirable breathing supply, which may last from a few minutes
to four hours, depending on type. With some, a negative pressure is
created inside the face-piece when the wearer breathes in, so the air
is drawn into the face-piece only when the wearer breathes in, instead
of being supplied to the face-piece before the wearer breathes in. If
the face seal is not good and a negative pressure is created inside
the face-piece, contaminated air may be sucked into the respirator
through the gaps between the face and the face-piece. These
negative-pressure types of breathing apparatus should not be used in
atmospheres immediately hazardous to life or health.
3.4.5.6 Training and fitting
Workers must be trained as to why respirators must be worn and how to
choose, fit and use them before they are expected to use them.
Respirators are uncomfortable to wear, and workers will often not use
them if they do not fully understand the health risks they face.
3.4.5.7 Respiratory protection programme
If respirators are to be used correctly and effectively, a respiratory
protection programme administered by a trained person should be
established. The programme should include written standard operating
procedures and specifications for:
* respirators (selected on the basis of hazards);
* instruction and training of the user;
* cleaning and disinfection;
* storage;
* inspection;
* surveillance of work area conditions;
* evaluation of the respiratory protection programme;
* use of certified respirators; and
* medical review.
3.5 Monitoring the hazard
Monitoring should be performed to check the efficiency of control
methods in the workplace. There are two types of monitoring --
environmental monitoring to measure workers' exposures to air
contaminants, and biological monitoring of individual workers to
test for degree of exposure, whether dermal, respiratory, via
ingestion, etc. The results can be compared with safety standards for
workplace air or biological materials.
3.5.1 Environmental monitoring
Monitoring of the workplace environment for contaminants may be
undertaken in two ways: through integrated sampling or, for some
substances, use of direct reading instruments. Direct reading
instruments give an immediate result, but for many substances such
devices are not available. In addition, they must be carefully
calibrated. Integrated sampling, on the other hand, can be used for a
wider range of substances, although the result is available only
following laboratory analysis. Integrated sampling involves measuring
the concentrations or levels of dusts, mists or gases in the workplace
air, by sampling known amounts of air containing the dust, mist or gas
with special pumps and filters and then chemically analysing the
results. This monitoring may be technically complicated and must
usually be carried out by an industrial hygienist; in some cases, it
is less complex and can be done by trained workers.
There are two main types of integrated sampling:
* Static monitoring -- in which an air pump is set up at a fixed
point. This is useful for chemicals that are expected to spread
evenly throughout the entire workplace. Static monitoring may
over- or underestimate the exposure individual workers are
receiving, depending on where measurements are made.
* Personal sampling -- in which air is sampled from individual
workers' breathing zones by a portable pump attached to the
workers' clothing. This method gives a much better indication of
an individual worker's exposure to a chemical and, of the two air
sampling techniques, is the method of choice. The advantage over
static or fixed-point monitoring is that a reading for exposure
is obtained that better reflects contamination in the worker's
breathing zone. With static or fixed-point monitoring,
concentrations in the worker's breathing zone will not be
measured if the measurement is taken at too great a distance from
the source.
The following questions should be considered in any environmental
monitoring programme:
* Are the samples being taken at the right place, i.e. near the
breathing zones of operators?
* Is the sample being taken at the time when pollution levels are
highest? The aim of environmental monitoring should always be to
determine the highest level of exposure, not the average or usual
level of exposure.
* Are all contaminants being measured?
* Have workers been involved in the planning of monitoring of their
workplace?
* Are the test results being made available to safety
representatives and safety committees regularly, and are the
results being discussed?
Environmental monitoring should be performed before decisions are made
regarding methods for controlling chemical exposure. In this way,
control methods can be designed that cope with the highest levels of
chemicals measured, not the average levels. In other words, control
measures must be able to cope with the "worst case".
After methods to control exposure (e.g. exhaust ventilation) have been
introduced, environmental monitoring should be repeated regularly to
ensure that the control methods are operating satisfactorily.
3.5.2 Biological monitoring
Biological monitoring of chemical exposure aims to measure either the
amount of a chemical that has been absorbed by a worker or the effect
of that absorbed chemical on the worker (as opposed to environmental
monitoring, which measures the level of exposure).
Biological monitoring involves taking a sample of body fluid, usually
blood or urine, and measuring the level of the chemical or its
metabolite (see Chapter 2). Alternatively, an effect of that chemical
on the body may be determined by measuring the level of an enzyme or
other chemical in the blood or urine.
A good example is biological monitoring for lead absorption. All
workers regularly exposed to lead or to significant levels of lead
should have blood samples taken regularly. The level of lead in the
blood can then be measured directly, and this will give an accurate
estimate of how much lead that worker has recently absorbed. One of
the effects of lead on the body is the poisoning of red blood cells.
By measuring the level of protoporphyrin -- a chemical in the blood
produced by poisoned red blood cells -- an estimate of the effects of
lead absorption can be obtained (see Fig. 41).
Many chemicals can be monitored biologically, but the results do not
always reflect the level of absorption. Each chemical should be
considered separately when deciding whether to perform biological
monitoring. Lists of those chemicals for which biological monitoring
is recommended are available, and some such chemicals may have a value
-- i.e. a biological exposure index -- that should not be exceeded.
Figure 41. Biological monitoring (from EHC 3: Lead)
Pb levels at which no more than 5% of the population will
show the indicated intensity of effect
Biochemical PbB
effect Intensity of effect Population (µg/100 ml)
ALAD inhibition perceptible inhibition adult, children 10
in blood >40% inhibition adults 15-20
>70% inhibition adults 30
>70% inhibition children 25-30
ALA in urine perceptible increase adults, children 40
>10 mg/litre adults, children 50
FEP in blood perceptible increase adult males 30
adult females 25
children 20
ALA = delta-aminolevulinic acid
ALAD = delta-aminolevulinic acid dehydratase
FEP = free erythrocyte porphyrins
PbB = blood lead level
4. PROTECTION AND EMERGENCY ACTION - CHAPTER 4
This chapter should be read in conjunction with chapter 4 of the
relevant HSG, which outlines the main hazards of the chemical
concerned (including a description of the fire and explosion risks),
provides medical advice in case of exposure and outlines storage,
transport and disposal considerations.
Good safety organization, ventilation and engineering controls,
adequate provision of information on the health hazards of chemicals
and training of workers can reduce and control chemical exposures in a
workplace.
Nevertheless, poisonings may still occur, and it is important that
training and proper equipment are available to workers so that
emergency situations can be handled satisfactorily (see Fig. 42).
4.1 First aid and emergencies
The following general principles should be employed when designing a
first-aid programme for a workplace where chemicals are used:
* There should be at least one worker present on every shift who
has received approved first-aid training. Follow-up or refresher
courses should be offered regularly.
* All workers should receive training in the technique of cardio-
pulmonary resuscitation (CPR). CPR is used when a person's heart
has stopped beating or when he or she has stopped breathing.
* It is essential that any workers affected or injured by chemicals
are removed from the area where the chemicals are present.
Rescuers should make sure of their own safety in any attempt at
rescue and/or resuscitation. Self-contained breathing apparatus
(a respirator mask with its own air supply) may be necessary so
that those attempting to rescue an injured or affected worker do
not become poisoned themselves.
* Emergency showers and eye-baths should be situated close to the
site of any potentially hazardous work processes.
b) Self-contained breathing apparatus -- This allows the wearer to
carry a respirable breathing supply, which may last from a few minutes
to four hours, depending on type. With some, a negative pressure is
created inside the face-piece when the wearer breathes in, so the air
is drawn into the face-piece only when the wearer breathes in, instead
of being supplied to the face-piece before the wearer breathes in. If
the face seal is not good and a negative pressure is created inside
the face-piece, contaminated air may be sucked into the respirator
through the gaps between the face and the face-piece. These
negative-pressure types of breathing apparatus should not be used in
atmospheres immediately hazardous to life or health.
3.4.5.6 Training and fitting
Workers must be trained as to why respirators must be worn and how to
choose, fit and use them before they are expected to use them.
Respirators are uncomfortable to wear, and workers will often not use
them if they do not fully understand the health risks they face.
3.4.5.7 Respiratory protection programme
If respirators are to be used correctly and effectively, a respiratory
protection programme administered by a trained person should be
established. The programme should include written standard operating
procedures and specifications for:
* respirators (selected on the basis of hazards);
* instruction and training of the user;
* cleaning and disinfection;
* storage;
* inspection;
* surveillance of work area conditions;
* evaluation of the respiratory protection programme;
* use of certified respirators; and
* medical review.
3.5 Monitoring the hazard
Monitoring should be performed to check the efficiency of control
methods in the workplace. There are two types of monitoring --
environmental monitoring to measure workers' exposures to air
contaminants, and biological monitoring of individual workers to
test for degree of exposure, whether dermal, respiratory, via
ingestion, etc. The results can be compared with safety standards for
workplace air or biological materials.
3.5.1 Environmental monitoring
Monitoring of the workplace environment for contaminants may be
undertaken in two ways: through integrated sampling or, for some
substances, use of direct reading instruments. Direct reading
instruments give an immediate result, but for many substances such
devices are not available. In addition, they must be carefully
calibrated. Integrated sampling, on the other hand, can be used for a
wider range of substances, although the result is available only
following laboratory analysis. Integrated sampling involves measuring
the concentrations or levels of dusts, mists or gases in the workplace
air, by sampling known amounts of air containing the dust, mist or gas
with special pumps and filters and then chemically analysing the
results. This monitoring may be technically complicated and must
usually be carried out by an industrial hygienist; in some cases, it
is less complex and can be done by trained workers.
There are two main types of integrated sampling:
* Static monitoring -- in which an air pump is set up at a fixed
point. This is useful for chemicals that are expected to spread
evenly throughout the entire workplace. Static monitoring may
over- or underestimate the exposure individual workers are
receiving, depending on where measurements are made.
* Personal sampling -- in which air is sampled from individual
workers' breathing zones by a portable pump attached to the
workers' clothing. This method gives a much better indication of
an individual worker's exposure to a chemical and, of the two air
sampling techniques, is the method of choice. The advantage over
static or fixed-point monitoring is that a reading for exposure
is obtained that better reflects contamination in the worker's
breathing zone. With static or fixed-point monitoring,
concentrations in the worker's breathing zone will not be
measured if the measurement is taken at too great a distance from
the source.
The following questions should be considered in any environmental
monitoring programme:
* Are the samples being taken at the right place, i.e. near the
breathing zones of operators?
* Is the sample being taken at the time when pollution levels are
highest? The aim of environmental monitoring should always be to
determine the highest level of exposure, not the average or usual
level of exposure.
* Are all contaminants being measured?
* Have workers been involved in the planning of monitoring of their
workplace?
* Are the test results being made available to safety
representatives and safety committees regularly, and are the
results being discussed?
Environmental monitoring should be performed before decisions are made
regarding methods for controlling chemical exposure. In this way,
control methods can be designed that cope with the highest levels of
chemicals measured, not the average levels. In other words, control
measures must be able to cope with the "worst case".
After methods to control exposure (e.g. exhaust ventilation) have been
introduced, environmental monitoring should be repeated regularly to
ensure that the control methods are operating satisfactorily.
3.5.2 Biological monitoring
Biological monitoring of chemical exposure aims to measure either the
amount of a chemical that has been absorbed by a worker or the effect
of that absorbed chemical on the worker (as opposed to environmental
monitoring, which measures the level of exposure).
Biological monitoring involves taking a sample of body fluid, usually
blood or urine, and measuring the level of the chemical or its
metabolite (see Chapter 2). Alternatively, an effect of that chemical
on the body may be determined by measuring the level of an enzyme or
other chemical in the blood or urine.
A good example is biological monitoring for lead absorption. All
workers regularly exposed to lead or to significant levels of lead
should have blood samples taken regularly. The level of lead in the
blood can then be measured directly, and this will give an accurate
estimate of how much lead that worker has recently absorbed. One of
the effects of lead on the body is the poisoning of red blood cells.
By measuring the level of protoporphyrin -- a chemical in the blood
produced by poisoned red blood cells -- an estimate of the effects of
lead absorption can be obtained (see Fig. 41).
Many chemicals can be monitored biologically, but the results do not
always reflect the level of absorption. Each chemical should be
considered separately when deciding whether to perform biological
monitoring. Lists of those chemicals for which biological monitoring
is recommended are available, and some such chemicals may have a value
-- i.e. a biological exposure index -- that should not be exceeded.
Figure 41. Biological monitoring (from EHC 3: Lead)
Pb levels at which no more than 5% of the population will
show the indicated intensity of effect
Biochemical PbB
effect Intensity of effect Population (µg/100 ml)
ALAD inhibition perceptible inhibition adult, children 10
in blood >40% inhibition adults 15-20
>70% inhibition adults 30
>70% inhibition children 25-30
ALA in urine perceptible increase adults, children 40
>10 mg/litre adults, children 50
FEP in blood perceptible increase adult males 30
adult females 25
children 20
ALA = delta-aminolevulinic acid
ALAD = delta-aminolevulinic acid dehydratase
FEP = free erythrocyte porphyrins
PbB = blood lead level
4. PROTECTION AND EMERGENCY ACTION - CHAPTER 4
This chapter should be read in conjunction with chapter 4 of the
relevant HSG, which outlines the main hazards of the chemical
concerned (including a description of the fire and explosion risks),
provides medical advice in case of exposure and outlines storage,
transport and disposal considerations.
Good safety organization, ventilation and engineering controls,
adequate provision of information on the health hazards of chemicals
and training of workers can reduce and control chemical exposures in a
workplace.
Nevertheless, poisonings may still occur, and it is important that
training and proper equipment are available to workers so that
emergency situations can be handled satisfactorily (see Fig. 42).
4.1 First aid and emergencies
The following general principles should be employed when designing a
first-aid programme for a workplace where chemicals are used:
* There should be at least one worker present on every shift who
has received approved first-aid training. Follow-up or refresher
courses should be offered regularly.
* All workers should receive training in the technique of cardio-
pulmonary resuscitation (CPR). CPR is used when a person's heart
has stopped beating or when he or she has stopped breathing.
* It is essential that any workers affected or injured by chemicals
are removed from the area where the chemicals are present.
Rescuers should make sure of their own safety in any attempt at
rescue and/or resuscitation. Self-contained breathing apparatus
(a respirator mask with its own air supply) may be necessary so
that those attempting to rescue an injured or affected worker do
not become poisoned themselves.
* Emergency showers and eye-baths should be situated close to the
site of any potentially hazardous work processes.
 * First-aid boxes should be provided in all work areas and workers
trained in emergency first-aid procedures for any accident or
chemical exposure that may occur.
* Telephone numbers for medical assistance and ambulances should be
prominently displayed in the workplace. A telephone must always
be available for use in an emergency.
* There should be an "emergency response plan" in which individuals
are assigned to perform certain tasks (such as summon help) while
all other personnel quickly leave the workplace. The plan should
be written down and prominently displayed in the workplace and
should be practised regularly.
* A system should operate whereby all accidents and injuries, no
matter how slight, are reported in writing to the management or
employer. These accident records should be made available to
workers and their representatives and analysed to determine how
to prevent the same, or similar, accidents from occurring again.
In many countries, occupational accidents and injuries must be
reported to government bodies.
* If an injured worker is sent to hospital or to see a doctor or
nurse, it is essential that information on the chemicals involved
in the accident be sent with the worker. The manufacturer's MSDS
and the ICSC or Summary of Chemical Safety Information contained
in HSGs (see Chapter 3) are usually suitable for this purpose.
4.2 Health surveillance
Health surveillance or medical monitoring consists of periodic
examination of a worker by a doctor, nurse or other health worker in
order to detect any health effects of a chemical, to monitor the
progression or reversal of symptoms or to see if treatment or
preventive measures are effective. Tests of lung, heart, liver and
kidney functions, for instance, may be performed. Although in some
cases -- e.g. chemicals that cause asthma -- such testing is useful,
it should always be remembered that medical monitoring can detect
damage to health only after it has occurred (see Fig. 43). Further
damage can then be prevented, but medical monitoring does not actually
prevent the occurrence of the initial health effects.
Figure 43. Health surveillance (from HSG 6: Methylene Chloride)
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically. Administer
oxygen if needed. Do not administer fats, oils, alcohol, epinephrine,
or ephedrine. Pay attention to signs of anoxaemia, impending
pulmonary oedema, and ischaemia of the heart. Monitor ECG. Treat
skin as for a thermal burn.
4.3 Health Surveillance Advice
Persons handling methylene chloride should frequently undergo
periodic medical examination with emphasis on the functioning
of the central nervous system and irritation of the skin and
eyes. Monitoring of blood-carboxyhaemoglobin concentrations can
give an indication of the extent of exposure.
Medical monitoring is often unable to detect the long-term or chronic
health effects of chemicals until they are quite advanced. This
applies particularly to:
* cancer caused by carcinogenic chemicals;
* effects on the brain and nervous system; and
* reproductive effects.
Medical monitoring is useful for assessing the effectiveness of
measures implemented to control chemical exposure, but it is not a
substitute for environmental monitoring and control methods.
4.3 Fire and explosion hazards
Many chemicals used in the workplace will burn or explode easily.
Accidental fires or explosions of flammable or explosive chemicals
represent major hazards. Special precautions must be taken when
handling, storing or transporting these chemicals to prevent fires or
explosions (see Fig. 44).
Figure 44. Explosion and fire hazards
(from HSG 6: Methylene Chloride)
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
Methylene chloride may react explosively with light metals (lithium,
sodium, potassium, aluminium, magnesium) as well as with alloys of
sodium. Explosive mixtures are formed with dinitrogen tetroxide
(N2O4) dinitrogen pentoxide (N2O5), nitric acid, and
compressed oxygen.
4.3.2 Fire hazards
Methylene chloride is flammable in an atmosphere with an increased
oxygen content. In pure oxygen, the flammable limits (explosive
limits) are 15.5% and 66.4%. Methylene chloride is also flammable
if small amounts of combustible material are added. The compound
decomposes in contact with open flames and glowing surfaces with the
formation of harmful gases, such as hydrogen chloride, which forms
mists of hydrochloric acid with moisture, and phosgene.
4.3.3 Prevention
Do not use methylene chloride in the vicinity of a fire, a hot
surface (e.g. a portable heating unit), or during welding. Do
not smoke. Do not add any combustible material to methylene
chloride. Avoid using compressed oxygen when handling
methylene chloride. Avoid contact between the compound and
light metals, alloys of light metals, dinitrogen tetroxide,
dinitrogen pentoxide, nitric acid, or liquid oxygen. In case
of fire, keep drums cool by spraying with water.
Fire (or combustion) is the violent combination of oxygen in the air
with a combustible chemical while producing heat (an exothermic
reaction).
In fires involving chemicals or chemical products, the chemicals
usually become broken down into combustion by-products. Many of
these combustion by-products are highly toxic gases and fumes. This
may be the case even if the original chemical was not particularly
toxic. For example, the gas Freon 12, or dichlorodifluoromethane, is
widely used as a refrigerant. It is toxic only at very high
concentrations. However, the thermal decomposition products of Freon
12 are very toxic gases, including phosgene, which causes potentially
fatal pulmonary oedema (accumulation of water in the lungs), even at
very low levels of exposure. Similarly, polyurethane foam, which is
commonly used as insulation in industry, has relatively low toxicity
in the cured state. However, when polyurethane foam burns, large
amounts of highly toxic gases, including cyanide and hydrogen
chloride, are released.
4.3.1 Flammable substances
A flammable substance is any chemical or material that will continue
to burn after having been ignited. A substance of high flammability
is one that will easily ignite at room temperature on contact with
low-energy ignition sources such as a spark. A substance of low
flammability will ignite only after heating or prolonged contact with
a high-energy ignition source. The flammability of a substance is
often ranked according to its ability to catch fire at certain
temperatures:
temperature
extremely flammable < 0°C
highly flammable 0-21°C
flammable 21-55°C
combustible > 55°C
Other important information about the flammability of a substance can
be gathered by looking at the flash point and the flammability
(explosive) limits (see Chapter 1) for that substance. This
information is given in the Physical Properties section of the ICSC
and usually appears on the MSDS supplied by the chemical manufacturer.
4.3.2 Dust explosions
Any solid material that is capable of burning in a finely divided form
can give rise to a dust explosion hazard. All that is necessary is
for enough dust to be in the air and for a source of ignition to be
present. Flame from the source of ignition will spread in all
directions through the cloud of dust, burning the dust and causing
expansion, thus increasing pressure on the surroundings. In a grain
store, for example, this pressure could be great enough to cause the
roof or walls to be blown off. The first explosion can whip dust that
is lying on window sills, pipework and equipment into the air, and a
second explosion can result.
Many dusts, including natural materials such as starch, sugar, flour,
coal and wood, plastics and organic chemicals, and light metals such
as aluminium, magnesium and titanium, can explode in this way.
Accordingly, dust explosion must be guarded against in agriculture,
chemical, metallurgical and process industries, as well as coal
mining.
4.3.3 Sources of ignition
Flammable substances or dust clouds may be ignited in one of the
following ways:
* Direct flames -- matches, cigarettes, soldering, welding or
cutting torches.
* Heat radiation -- all sources of heat emit heat (thermal)
radiation, which may ignite a flammable material at a distance.
* Electrical sparks -- produced by electric current in a number of
ways, including during electric arc welding; by power supplies
such as sockets and switches and sliding contacts; from electric
motors in power tools or the ignition systems of internal
combustion engines; by lightning; and as a result of static
electricity build-up between two non-conducting materials such as
belts on a pulley, rubber tyres on the ground or liquids pumped
or poured through pipes.
* Mechanically produced sparks -- two hard materials rubbing
against each other at high speed may produce sparks capable of
igniting flammable chemicals, e.g. when grinding.
* Chemical reactions -- many chemical reactions produce heat. The
design of the chemical process should take this into account and
make provision for the removal of excess heat. However, excess
heat may develop if the incorrect or wrong proportions of
chemicals are used. This may start a fire or trigger an
explosion.
* Spontaneous combustion -- some substances oxidize (combine with
oxygen) as soon as they are exposed to air. Usually this occurs
slowly, but with some chemicals it may happen rapidly and produce
considerable heat in the process. These chemicals must be stored
in tanks or containers that allow all air to be removed and
replaced with a gas that will not combine with the chemical
(usually nitrogen). If this mechanism fails and air enters the
container, a fire or explosion may result.
Safety information about fire hazards is given in chapter 4 of the
HSGs and is also contained in the ICSCs. Information on fire and
explosion hazards should always be included in chemical manufacturers'
MSDS.
4.3.4 Fire-fighting
Equipment used to extinguish and control fires is of two types: fixed
and portable. Fixed systems include water equipment, automatic
sprinklers, hydrants and special pipe systems for dry chemicals,
carbon dioxide, foam, etc. Special pipe systems are applicable to
areas of high fire potential where water may not be effective, such as
tanks for storage of flammable liquids.
Fixed systems must be supplemented by portable fire extinguishers. As
well as providing a means of rapidly extinguishing a fire, they can
often be used to prevent a small fire from spreading.
To be effective, portable extinguishers must be appropriate for the
type of fire; the HSGs provide information to this effect. For
example, water-based extinguishers can be used on ordinary
combustibles such as wood, paper, textiles, etc., where a
quenching/cooling effect is required. Dry chemical or carbon dioxide
extinguishers are used on flammable liquid and gas fires, such as oil,
gasoline, paint and grease, where an oxygen-exclusion or
flame-interrupting effect is necessary. Dry powder agents are
necessary to combat metal fires.
In addition, portable extinguishers must be available in sufficient
quantity to protect against potential fires in any area. They should
be located where they are readily accessible for immediate use,
maintained in good operating condition, inspected frequently and
recharged as necessary. Workers should be trained to use them
effectively.
If a fire or explosion warrants intervention by outside fire service
personnel, these personnel should be advised of the nature of the
chemicals and the worksite so that they can evaluate what is needed
for their own safety.
4.4 Storage
The HSGs give some advice on storage (see Fig. 45), but the
manufacturers' or suppliers' MSDSs should give specific instructions
on the storage of each chemical. These conditions must be strictly
adhered to, as storage requirements vary according to the nature of
the chemical being stored. Incorrect storage can have disastrous
results, e.g. fire, explosion or release of toxic chemicals.
The MSDS should include the following information:
* Need for isolation -- Certain chemicals must not be stored
together, as any vapours or leaks may, if they intermingle, lead
to an explosion.
Figure 45. Storage (from HSG 19: Pentachlorophenol)
4.3 Storage
Technical PCP is a solid. The formulated product is usually a
solution in oil or organic solvent, or an emulsifiable concentrate.
All PCP products should be stored in secure, well-ventilated
buildings under cool and dry conditions, and out of reach of
children and unauthorized persons. Keep away from food, drink,
and animal feed.
* Flash point -- Chemicals must be stored at a temperature below
their flash points. (Refer to Chapter 2 of this Manual for a
discussion of flash point.) Chemicals with flash points below
34°C are especially dangerous.
* Upper and lower flammable limits -- Information on flammable
limits can be found in Chapter 2. MSDSs often specify a
"well-ventilated" store-room for particular chemicals, and it is
essential to comply with this requirement. More specific
guidance on the amount of ventilation required can be obtained
from the manufacturers of the chemicals and guidance can be
checked by an industrial hygienist or ventilation engineer.
* Auto-ignition temperature -- The storage temperature must
obviously be below the auto-ignition temperature (see Chapter 2).
* Type of container -- Chemicals may react with the material from
which containers are made; the type of container required must be
specified on the MSDS. This is especially important if chemicals
may be transferred from one container to another. Also, there
may be other requirements, such as pressure relief valves, which
are relevant to the storage of particular chemicals.
* Type of flooring -- This should be specified, as the flooring
must be resistant to, and not potentially reactive with, the
chemical being stored.
* Dykes (berms or bunding) -- These are low walls or embankments
that are constructed around the storage area to contain any leaks
from storage containers. The height of the dyke should be
sufficient to contain any leaks that may occur and any water or
foam sprayed in the event of a fire.
* Alarms -- Alarms should be fitted in areas where potentially
dangerous chemicals are stored to give early warning of releases
of those chemicals.
4.5 Spillage
Many spills can be prevented by thorough planning of work, provision
of suitable equipment for carrying out the work, regular preventive
maintenance and good training of workers. Any spillages that do
happen should be thoroughly investigated and remedial action taken to
prevent their recurrence.
Employers should ensure that they have the necessary plans and
equipment to deal with spillages, that the workforce and their
representatives have been consulted about all such planning and that
the necessary training is carried out regularly.
When a spillage occurs, suitable precautions must first be taken to
protect workers from the dangers of the chemical (fumes, burns, etc.)
before steps are taken to deal with the spillage (see Fig. 46).
Some general measures that may apply include the following:
* use self-contained breathing apparatus and full protective
clothing, when applicable;
* remove ignition sources;
* do not smoke;
* evacuate area;
* collect leaking liquid in sealable containers;
* prevent liquid from spreading or contaminating other areas,
vegetation, waterways and cargo, with a barrier of the most
suitable available material, e.g. soil or sand;
* in some cases (e.g. hydrazine), a foam can be applied to slow
down vaporization;
* absorb spills in sand, soil, moist sawdust or other inert
material and transfer to a suitable container; then remove to a
safe place, with disposal in accordance with local regulations;
* sweep up solid products, and transfer to a suitable container;
and
* depending on the chemical, do not allow run-off into sewers: it
may cause an explosion, kill wildlife or affect water supplies.
Figure 46. Spillage (from HSG 35: Phosphorus Trichloride)
4.7.1 Spillage
Liquid spillage should be handled only by trained personnel wearing
protective clothing and full-face mask/positive-pressure breathing
apparatus. The spillage should be absorbed in dry sand or in an
inert absorbent material and shovelled into sealable polyethylene-
lined containers for disposal.
Vapour drifting towards people can be "knocked-down" with water
spray. Do not wet the spillage itself and never use a water jet on
a spillage. When the absorbent medium has been cleared from the
ground, flush the contaminated area with water until the fumes
disappear; residues may be neutralized with soda ash (sodium
carbonate or lime).
If phosphorus trichloride or phosphorus oxychloride enters a
watercourse or sewer, or contaminates soil or vegetation, warn the
police and public authorities immediately.
The actual steps taken will depend on the chemical in question, and
information on this must already have been collected from the MSDS and
from consultations with the manufacturers and/or suppliers and
emergency services (e.g. fire authorities) and incorporated in the
employer's written statement on spills.
4.6 Disposal
4.6.1 Means of disposal
Given that enormous volumes of waste are generated in the production
and use of chemicals, waste disposal has become a key issue in
environmental protection. As with protection of the workplace, a
hierarchy of controls should be applied when dealing with chemical
waste, as follows:
* reduction of waste at the source;
* segregation of waste;
* recovery and recycling;
* waste exchange;
* incineration;
* immobilization of intractable wastes;
* landfill;
* discharge into sewer; and
* storage.
The volume of hazardous waste can be reduced by modifying a process or
improving process control to produce less waste or to render the waste
less hazardous.
Waste can be segregated to assist analysis, control and treatment --
thereby reducing disposal costs.
* Recycling -- The best-known form of recycling is the recovery of
useful fractions for reuse. These processes are usually carried
out by specialist recovery operators off-site and involve
recovery of materials such as oils and solvents, as well as other
valuable materials, such as the silver in photographic waste.
Not all wastes are suitable for recycling. Even when valuable
materials such as mercury or lead are present, full recovery
might not be economically feasible owing to such factors as high
processing costs, small volumes and irregular composition. As
extraction techniques improve, more waste products are likely to
become eligible for recycling.
* Waste exchanges -- A considerable amount of waste is suitable for
exchange. The aim of the exchange is to put potential users of
waste materials in touch with industries producing the waste, and
vice versa. Waste exchanges reduce the volume of wastes requiring
landfill or incineration and therefore reduce the load on
disposal facilities and the environment, as well as the cost of
disposal.
* Incineration -- This process, which involves burning in special
high-temperature (1200°C) incinerators, is the only
environmentally acceptable means of disposing of some wastes,
such as chlorinated hydrocarbons, polychlorinated biphenyls
(PCBs) and dioxins (see Fig 47). These chemicals are highly
toxic, persistent and non-biodegradable. Incineration effectively
destroys many organic wastes, and the energy in solvent and fuel
waste can be exploited in the process. On the other hand,
inorganic chemical dyes in plastics may cause pollution problems
when the plastics are incinerated, and dangerous dioxins may be
formed if some organic materials are incinerated at too low
temperatures.
Figure 47. Disposal (from HSG 19: Pentachlorophenol)
4.5.2 Disposal
Surplus product, contaminated absorbents and containers should be
burned at a high temperature in an appropriate incinerator with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, in an area where there is no risk of contamination of
surface water or groundwater. Comply with any local legislation.
* Immobilization of intractable wastes -- This is used to isolate a
hazard physically and/or chemically from its surroundings.
* Chemical fixation -- Waste is incorporated into an inert matrix
(framework) to limit or prevent its migration into the
environment. Should the waste block be fractured, the waste
remains bound to the matrix, and leaching is prevented.
* Encapsulation -- The waste is sealed within a stable, inert
material to prevent contact with the environment and prevent
movement in the environment (migration). Should the
encapsulating jacket be broken, the waste could leach away.
Encapsulation is more suited to those wastes that pose a handling
hazard but are relatively inert once buried (e.g. asbestos).
* Landfill -- Because of the limited amount of suitable land
available for waste disposal, it is increasingly desirable that
liquid wastes be treated by chemical and physical methods,
followed by biological oxidation (digestion by special bacteria
or algae). After treatment, most of the liquid will be suitable
for discharge to sewers. The remaining liquid can be recycled or
incinerated. Landfills could then be used for most residual
solids or pastes because they are of smaller volume and less
likely to migrate through the soil. A number of solid hazardous
wastes are not regarded as suitable for disposal to normal
chemical landfill facilities. These wastes require the higher
degree of safety afforded by secure landfill, whereby waste is
poured into small cells lined with an impermeable clay or
synthetic material and subsequently buried under a layer of soil.
However, it is highly unlikely that such a system would function
without some leakage through the clay or synthetic material,
without any damage to the cell and with 100% collection of any
leaks. Added to this are the potential problems of infiltration
by rainwater and ensuring permanent maintenance of the cell if
the company moves away or ceases to operate.
* Disposal of less hazardous waste to the sewer -- This is
generally not recommended (see Fig. 48). Most sewage authorities
make regulatory provisions for the control of the kind and
quantity of (less) hazardous waste that may be disposed of
through the sewer. Improper disposal to the sewer can disrupt
the biological treatment of sewage and represent a hazard at
sewer outfalls. In addition, toxic chemicals (e.g. heavy metals)
may accumulate in the sewage sludge and create hazards when they
are disposed of.
Figure 48. "Avoid run-off into sewers"
(from HSG 34: Fenvalerate)
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams, water
courses, open waterways, ditches, fields with drainage systems, or to
the catchment areas of boreholes, wells, springs, or ponds.
* Storage of intractable wastes -- A large volume of hazardous
waste is currently stored -- usually in steel drums on industrial
sites, awaiting the development of satisfactory disposal methods
-- because it is too toxic to be disposed of legally into air,
water or landfill sites. Most drums are stored in the open, and
many contain corrosive materials. There is an added risk of
hazard from fires, structural damage and vandalism of such toxic
waste stores. It is likely that some of these drums will corrode
and leak. In some countries, such stores must be registered with
authorities who, in turn, have a duty to inspect them.
4.6.2 An example of the effects of incorrect waste disposal
For years, the residents of Love Canal, a residential community near
Niagara Falls, New York, USA, complained of noxious vapours in their
basements, which they believed originated from a disused toxic waste
dump beneath their housing estate. Following prolonged rains and
heavy snows in the winter of 1977, water seeped into the dump-site,
causing it to overflow and forcing the chemicals out into the soil.
Two hundred and thirty-five families were evacuated, and Love Canal
was declared a federal disaster area, the first instance of a national
emergency being caused by chemical pollution. Later, more families
had to be moved when it was discovered that the toxic waste was
migrating further afield via underground streams.
Almost 22 000 tonnes of chemicals had been buried in Love Canal; the
canal had been capped with clay and covered with soil in 1953. The
clay cap was apparently damaged during later building work on the
site. Approximately 300 different chemical compounds were buried
there, although no records were kept of what, or how much, was dumped.
Studies now reveal that the Love Canal area experienced three times
the normal rate of miscarriages and three-and-a-half times the normal
rate of birth defects, most notably cleft palate, mental retardation
and club foot.
In addition, US$30 million have been spent on cleaning up the area,
and US$4.5 million on rehousing residents.
5. HOW CHEMICALS CAN POISON THE ENVIRONMENT - CHAPTER 5
This chapter is a background to chapter 5 of the HSG: "Hazards for the
Environment and their Prevention".
Almost any industrial or agricultural activity in which chemicals are
used will, by its very nature, have an impact on the environment (see
Fig. 49).
Figure 49. How chemicals can affect the environment
(from HSG 35: Phosphorus Trichloride)
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Phosphorus trichloride and phosphorus oxychloride hydrolyse in water
to form hydrochloric and phosphorous or phosphoric acids. The toxic
effects on organisms in the environment depend on the extent to which
the hydrogen ion is diluted and buffered; organisms die if they come
in contact with undiluted material.
In general, an environmental hazard will occur only in the immediate
area of a spillage or if disposed of improperly. Recovery of soil
and surface waters is rapid, and there are no residual effects on
the environment.
5.1 How do chemicals get into the environment?
Chemicals are released from the workplace into the environment in the
form of liquid, dust, fumes or gas. Such releases can be planned
(part of the work process, e.g. smokestack emissions, exhaust
ventilation, waste water into rivers, etc.) or unplanned (accidental).
Chemicals can also enter the environment during transport (e.g. from
the site of manufacture to the site of use), during their intended use
(e.g. pesticide spraying) or through disposal in landfills and
waterways. Chemicals also enter the environment through
non-occupational activities. Examples include exhaust fumes from motor
vehicles. Additionally, naturally occurring chemicals such as metals
in ores may be released by natural processes.
Chemicals released as smoke and dust from factory chimneys will
eventually fall to the earth's surface as dust and in rain. Wind
speed, air currents and other climatic conditions will determine when
and where the chemicals will be deposited. Some will settle on water
in lakes, rivers and streams, eventually passing into sediments and
nearby soils. Others may be transported over long distances and may
even affect the chemical and physical processes of the atmosphere and
climate. For example, the effects of sulfur and nitrogen oxides
released in industrial areas have contributed to acid rain, often
falling in countries other than those in which they were emitted.
Emissions of carbon dioxide, methane, ozone, etc. are thought to
contribute to global warming.
Once chemicals are released into the environment in this way, humans
and animals will be exposed to them through breathing contaminated
air, from direct contact with contaminated soil and water, through
eating plants grown on contaminated soils, by drinking contaminated
water supplies or by consuming food from contaminated animals (see
Fig. 50).
* First-aid boxes should be provided in all work areas and workers
trained in emergency first-aid procedures for any accident or
chemical exposure that may occur.
* Telephone numbers for medical assistance and ambulances should be
prominently displayed in the workplace. A telephone must always
be available for use in an emergency.
* There should be an "emergency response plan" in which individuals
are assigned to perform certain tasks (such as summon help) while
all other personnel quickly leave the workplace. The plan should
be written down and prominently displayed in the workplace and
should be practised regularly.
* A system should operate whereby all accidents and injuries, no
matter how slight, are reported in writing to the management or
employer. These accident records should be made available to
workers and their representatives and analysed to determine how
to prevent the same, or similar, accidents from occurring again.
In many countries, occupational accidents and injuries must be
reported to government bodies.
* If an injured worker is sent to hospital or to see a doctor or
nurse, it is essential that information on the chemicals involved
in the accident be sent with the worker. The manufacturer's MSDS
and the ICSC or Summary of Chemical Safety Information contained
in HSGs (see Chapter 3) are usually suitable for this purpose.
4.2 Health surveillance
Health surveillance or medical monitoring consists of periodic
examination of a worker by a doctor, nurse or other health worker in
order to detect any health effects of a chemical, to monitor the
progression or reversal of symptoms or to see if treatment or
preventive measures are effective. Tests of lung, heart, liver and
kidney functions, for instance, may be performed. Although in some
cases -- e.g. chemicals that cause asthma -- such testing is useful,
it should always be remembered that medical monitoring can detect
damage to health only after it has occurred (see Fig. 43). Further
damage can then be prevented, but medical monitoring does not actually
prevent the occurrence of the initial health effects.
Figure 43. Health surveillance (from HSG 6: Methylene Chloride)
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically. Administer
oxygen if needed. Do not administer fats, oils, alcohol, epinephrine,
or ephedrine. Pay attention to signs of anoxaemia, impending
pulmonary oedema, and ischaemia of the heart. Monitor ECG. Treat
skin as for a thermal burn.
4.3 Health Surveillance Advice
Persons handling methylene chloride should frequently undergo
periodic medical examination with emphasis on the functioning
of the central nervous system and irritation of the skin and
eyes. Monitoring of blood-carboxyhaemoglobin concentrations can
give an indication of the extent of exposure.
Medical monitoring is often unable to detect the long-term or chronic
health effects of chemicals until they are quite advanced. This
applies particularly to:
* cancer caused by carcinogenic chemicals;
* effects on the brain and nervous system; and
* reproductive effects.
Medical monitoring is useful for assessing the effectiveness of
measures implemented to control chemical exposure, but it is not a
substitute for environmental monitoring and control methods.
4.3 Fire and explosion hazards
Many chemicals used in the workplace will burn or explode easily.
Accidental fires or explosions of flammable or explosive chemicals
represent major hazards. Special precautions must be taken when
handling, storing or transporting these chemicals to prevent fires or
explosions (see Fig. 44).
Figure 44. Explosion and fire hazards
(from HSG 6: Methylene Chloride)
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
Methylene chloride may react explosively with light metals (lithium,
sodium, potassium, aluminium, magnesium) as well as with alloys of
sodium. Explosive mixtures are formed with dinitrogen tetroxide
(N2O4) dinitrogen pentoxide (N2O5), nitric acid, and
compressed oxygen.
4.3.2 Fire hazards
Methylene chloride is flammable in an atmosphere with an increased
oxygen content. In pure oxygen, the flammable limits (explosive
limits) are 15.5% and 66.4%. Methylene chloride is also flammable
if small amounts of combustible material are added. The compound
decomposes in contact with open flames and glowing surfaces with the
formation of harmful gases, such as hydrogen chloride, which forms
mists of hydrochloric acid with moisture, and phosgene.
4.3.3 Prevention
Do not use methylene chloride in the vicinity of a fire, a hot
surface (e.g. a portable heating unit), or during welding. Do
not smoke. Do not add any combustible material to methylene
chloride. Avoid using compressed oxygen when handling
methylene chloride. Avoid contact between the compound and
light metals, alloys of light metals, dinitrogen tetroxide,
dinitrogen pentoxide, nitric acid, or liquid oxygen. In case
of fire, keep drums cool by spraying with water.
Fire (or combustion) is the violent combination of oxygen in the air
with a combustible chemical while producing heat (an exothermic
reaction).
In fires involving chemicals or chemical products, the chemicals
usually become broken down into combustion by-products. Many of
these combustion by-products are highly toxic gases and fumes. This
may be the case even if the original chemical was not particularly
toxic. For example, the gas Freon 12, or dichlorodifluoromethane, is
widely used as a refrigerant. It is toxic only at very high
concentrations. However, the thermal decomposition products of Freon
12 are very toxic gases, including phosgene, which causes potentially
fatal pulmonary oedema (accumulation of water in the lungs), even at
very low levels of exposure. Similarly, polyurethane foam, which is
commonly used as insulation in industry, has relatively low toxicity
in the cured state. However, when polyurethane foam burns, large
amounts of highly toxic gases, including cyanide and hydrogen
chloride, are released.
4.3.1 Flammable substances
A flammable substance is any chemical or material that will continue
to burn after having been ignited. A substance of high flammability
is one that will easily ignite at room temperature on contact with
low-energy ignition sources such as a spark. A substance of low
flammability will ignite only after heating or prolonged contact with
a high-energy ignition source. The flammability of a substance is
often ranked according to its ability to catch fire at certain
temperatures:
temperature
extremely flammable < 0°C
highly flammable 0-21°C
flammable 21-55°C
combustible > 55°C
Other important information about the flammability of a substance can
be gathered by looking at the flash point and the flammability
(explosive) limits (see Chapter 1) for that substance. This
information is given in the Physical Properties section of the ICSC
and usually appears on the MSDS supplied by the chemical manufacturer.
4.3.2 Dust explosions
Any solid material that is capable of burning in a finely divided form
can give rise to a dust explosion hazard. All that is necessary is
for enough dust to be in the air and for a source of ignition to be
present. Flame from the source of ignition will spread in all
directions through the cloud of dust, burning the dust and causing
expansion, thus increasing pressure on the surroundings. In a grain
store, for example, this pressure could be great enough to cause the
roof or walls to be blown off. The first explosion can whip dust that
is lying on window sills, pipework and equipment into the air, and a
second explosion can result.
Many dusts, including natural materials such as starch, sugar, flour,
coal and wood, plastics and organic chemicals, and light metals such
as aluminium, magnesium and titanium, can explode in this way.
Accordingly, dust explosion must be guarded against in agriculture,
chemical, metallurgical and process industries, as well as coal
mining.
4.3.3 Sources of ignition
Flammable substances or dust clouds may be ignited in one of the
following ways:
* Direct flames -- matches, cigarettes, soldering, welding or
cutting torches.
* Heat radiation -- all sources of heat emit heat (thermal)
radiation, which may ignite a flammable material at a distance.
* Electrical sparks -- produced by electric current in a number of
ways, including during electric arc welding; by power supplies
such as sockets and switches and sliding contacts; from electric
motors in power tools or the ignition systems of internal
combustion engines; by lightning; and as a result of static
electricity build-up between two non-conducting materials such as
belts on a pulley, rubber tyres on the ground or liquids pumped
or poured through pipes.
* Mechanically produced sparks -- two hard materials rubbing
against each other at high speed may produce sparks capable of
igniting flammable chemicals, e.g. when grinding.
* Chemical reactions -- many chemical reactions produce heat. The
design of the chemical process should take this into account and
make provision for the removal of excess heat. However, excess
heat may develop if the incorrect or wrong proportions of
chemicals are used. This may start a fire or trigger an
explosion.
* Spontaneous combustion -- some substances oxidize (combine with
oxygen) as soon as they are exposed to air. Usually this occurs
slowly, but with some chemicals it may happen rapidly and produce
considerable heat in the process. These chemicals must be stored
in tanks or containers that allow all air to be removed and
replaced with a gas that will not combine with the chemical
(usually nitrogen). If this mechanism fails and air enters the
container, a fire or explosion may result.
Safety information about fire hazards is given in chapter 4 of the
HSGs and is also contained in the ICSCs. Information on fire and
explosion hazards should always be included in chemical manufacturers'
MSDS.
4.3.4 Fire-fighting
Equipment used to extinguish and control fires is of two types: fixed
and portable. Fixed systems include water equipment, automatic
sprinklers, hydrants and special pipe systems for dry chemicals,
carbon dioxide, foam, etc. Special pipe systems are applicable to
areas of high fire potential where water may not be effective, such as
tanks for storage of flammable liquids.
Fixed systems must be supplemented by portable fire extinguishers. As
well as providing a means of rapidly extinguishing a fire, they can
often be used to prevent a small fire from spreading.
To be effective, portable extinguishers must be appropriate for the
type of fire; the HSGs provide information to this effect. For
example, water-based extinguishers can be used on ordinary
combustibles such as wood, paper, textiles, etc., where a
quenching/cooling effect is required. Dry chemical or carbon dioxide
extinguishers are used on flammable liquid and gas fires, such as oil,
gasoline, paint and grease, where an oxygen-exclusion or
flame-interrupting effect is necessary. Dry powder agents are
necessary to combat metal fires.
In addition, portable extinguishers must be available in sufficient
quantity to protect against potential fires in any area. They should
be located where they are readily accessible for immediate use,
maintained in good operating condition, inspected frequently and
recharged as necessary. Workers should be trained to use them
effectively.
If a fire or explosion warrants intervention by outside fire service
personnel, these personnel should be advised of the nature of the
chemicals and the worksite so that they can evaluate what is needed
for their own safety.
4.4 Storage
The HSGs give some advice on storage (see Fig. 45), but the
manufacturers' or suppliers' MSDSs should give specific instructions
on the storage of each chemical. These conditions must be strictly
adhered to, as storage requirements vary according to the nature of
the chemical being stored. Incorrect storage can have disastrous
results, e.g. fire, explosion or release of toxic chemicals.
The MSDS should include the following information:
* Need for isolation -- Certain chemicals must not be stored
together, as any vapours or leaks may, if they intermingle, lead
to an explosion.
Figure 45. Storage (from HSG 19: Pentachlorophenol)
4.3 Storage
Technical PCP is a solid. The formulated product is usually a
solution in oil or organic solvent, or an emulsifiable concentrate.
All PCP products should be stored in secure, well-ventilated
buildings under cool and dry conditions, and out of reach of
children and unauthorized persons. Keep away from food, drink,
and animal feed.
* Flash point -- Chemicals must be stored at a temperature below
their flash points. (Refer to Chapter 2 of this Manual for a
discussion of flash point.) Chemicals with flash points below
34°C are especially dangerous.
* Upper and lower flammable limits -- Information on flammable
limits can be found in Chapter 2. MSDSs often specify a
"well-ventilated" store-room for particular chemicals, and it is
essential to comply with this requirement. More specific
guidance on the amount of ventilation required can be obtained
from the manufacturers of the chemicals and guidance can be
checked by an industrial hygienist or ventilation engineer.
* Auto-ignition temperature -- The storage temperature must
obviously be below the auto-ignition temperature (see Chapter 2).
* Type of container -- Chemicals may react with the material from
which containers are made; the type of container required must be
specified on the MSDS. This is especially important if chemicals
may be transferred from one container to another. Also, there
may be other requirements, such as pressure relief valves, which
are relevant to the storage of particular chemicals.
* Type of flooring -- This should be specified, as the flooring
must be resistant to, and not potentially reactive with, the
chemical being stored.
* Dykes (berms or bunding) -- These are low walls or embankments
that are constructed around the storage area to contain any leaks
from storage containers. The height of the dyke should be
sufficient to contain any leaks that may occur and any water or
foam sprayed in the event of a fire.
* Alarms -- Alarms should be fitted in areas where potentially
dangerous chemicals are stored to give early warning of releases
of those chemicals.
4.5 Spillage
Many spills can be prevented by thorough planning of work, provision
of suitable equipment for carrying out the work, regular preventive
maintenance and good training of workers. Any spillages that do
happen should be thoroughly investigated and remedial action taken to
prevent their recurrence.
Employers should ensure that they have the necessary plans and
equipment to deal with spillages, that the workforce and their
representatives have been consulted about all such planning and that
the necessary training is carried out regularly.
When a spillage occurs, suitable precautions must first be taken to
protect workers from the dangers of the chemical (fumes, burns, etc.)
before steps are taken to deal with the spillage (see Fig. 46).
Some general measures that may apply include the following:
* use self-contained breathing apparatus and full protective
clothing, when applicable;
* remove ignition sources;
* do not smoke;
* evacuate area;
* collect leaking liquid in sealable containers;
* prevent liquid from spreading or contaminating other areas,
vegetation, waterways and cargo, with a barrier of the most
suitable available material, e.g. soil or sand;
* in some cases (e.g. hydrazine), a foam can be applied to slow
down vaporization;
* absorb spills in sand, soil, moist sawdust or other inert
material and transfer to a suitable container; then remove to a
safe place, with disposal in accordance with local regulations;
* sweep up solid products, and transfer to a suitable container;
and
* depending on the chemical, do not allow run-off into sewers: it
may cause an explosion, kill wildlife or affect water supplies.
Figure 46. Spillage (from HSG 35: Phosphorus Trichloride)
4.7.1 Spillage
Liquid spillage should be handled only by trained personnel wearing
protective clothing and full-face mask/positive-pressure breathing
apparatus. The spillage should be absorbed in dry sand or in an
inert absorbent material and shovelled into sealable polyethylene-
lined containers for disposal.
Vapour drifting towards people can be "knocked-down" with water
spray. Do not wet the spillage itself and never use a water jet on
a spillage. When the absorbent medium has been cleared from the
ground, flush the contaminated area with water until the fumes
disappear; residues may be neutralized with soda ash (sodium
carbonate or lime).
If phosphorus trichloride or phosphorus oxychloride enters a
watercourse or sewer, or contaminates soil or vegetation, warn the
police and public authorities immediately.
The actual steps taken will depend on the chemical in question, and
information on this must already have been collected from the MSDS and
from consultations with the manufacturers and/or suppliers and
emergency services (e.g. fire authorities) and incorporated in the
employer's written statement on spills.
4.6 Disposal
4.6.1 Means of disposal
Given that enormous volumes of waste are generated in the production
and use of chemicals, waste disposal has become a key issue in
environmental protection. As with protection of the workplace, a
hierarchy of controls should be applied when dealing with chemical
waste, as follows:
* reduction of waste at the source;
* segregation of waste;
* recovery and recycling;
* waste exchange;
* incineration;
* immobilization of intractable wastes;
* landfill;
* discharge into sewer; and
* storage.
The volume of hazardous waste can be reduced by modifying a process or
improving process control to produce less waste or to render the waste
less hazardous.
Waste can be segregated to assist analysis, control and treatment --
thereby reducing disposal costs.
* Recycling -- The best-known form of recycling is the recovery of
useful fractions for reuse. These processes are usually carried
out by specialist recovery operators off-site and involve
recovery of materials such as oils and solvents, as well as other
valuable materials, such as the silver in photographic waste.
Not all wastes are suitable for recycling. Even when valuable
materials such as mercury or lead are present, full recovery
might not be economically feasible owing to such factors as high
processing costs, small volumes and irregular composition. As
extraction techniques improve, more waste products are likely to
become eligible for recycling.
* Waste exchanges -- A considerable amount of waste is suitable for
exchange. The aim of the exchange is to put potential users of
waste materials in touch with industries producing the waste, and
vice versa. Waste exchanges reduce the volume of wastes requiring
landfill or incineration and therefore reduce the load on
disposal facilities and the environment, as well as the cost of
disposal.
* Incineration -- This process, which involves burning in special
high-temperature (1200°C) incinerators, is the only
environmentally acceptable means of disposing of some wastes,
such as chlorinated hydrocarbons, polychlorinated biphenyls
(PCBs) and dioxins (see Fig 47). These chemicals are highly
toxic, persistent and non-biodegradable. Incineration effectively
destroys many organic wastes, and the energy in solvent and fuel
waste can be exploited in the process. On the other hand,
inorganic chemical dyes in plastics may cause pollution problems
when the plastics are incinerated, and dangerous dioxins may be
formed if some organic materials are incinerated at too low
temperatures.
Figure 47. Disposal (from HSG 19: Pentachlorophenol)
4.5.2 Disposal
Surplus product, contaminated absorbents and containers should be
burned at a high temperature in an appropriate incinerator with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, in an area where there is no risk of contamination of
surface water or groundwater. Comply with any local legislation.
* Immobilization of intractable wastes -- This is used to isolate a
hazard physically and/or chemically from its surroundings.
* Chemical fixation -- Waste is incorporated into an inert matrix
(framework) to limit or prevent its migration into the
environment. Should the waste block be fractured, the waste
remains bound to the matrix, and leaching is prevented.
* Encapsulation -- The waste is sealed within a stable, inert
material to prevent contact with the environment and prevent
movement in the environment (migration). Should the
encapsulating jacket be broken, the waste could leach away.
Encapsulation is more suited to those wastes that pose a handling
hazard but are relatively inert once buried (e.g. asbestos).
* Landfill -- Because of the limited amount of suitable land
available for waste disposal, it is increasingly desirable that
liquid wastes be treated by chemical and physical methods,
followed by biological oxidation (digestion by special bacteria
or algae). After treatment, most of the liquid will be suitable
for discharge to sewers. The remaining liquid can be recycled or
incinerated. Landfills could then be used for most residual
solids or pastes because they are of smaller volume and less
likely to migrate through the soil. A number of solid hazardous
wastes are not regarded as suitable for disposal to normal
chemical landfill facilities. These wastes require the higher
degree of safety afforded by secure landfill, whereby waste is
poured into small cells lined with an impermeable clay or
synthetic material and subsequently buried under a layer of soil.
However, it is highly unlikely that such a system would function
without some leakage through the clay or synthetic material,
without any damage to the cell and with 100% collection of any
leaks. Added to this are the potential problems of infiltration
by rainwater and ensuring permanent maintenance of the cell if
the company moves away or ceases to operate.
* Disposal of less hazardous waste to the sewer -- This is
generally not recommended (see Fig. 48). Most sewage authorities
make regulatory provisions for the control of the kind and
quantity of (less) hazardous waste that may be disposed of
through the sewer. Improper disposal to the sewer can disrupt
the biological treatment of sewage and represent a hazard at
sewer outfalls. In addition, toxic chemicals (e.g. heavy metals)
may accumulate in the sewage sludge and create hazards when they
are disposed of.
Figure 48. "Avoid run-off into sewers"
(from HSG 34: Fenvalerate)
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams, water
courses, open waterways, ditches, fields with drainage systems, or to
the catchment areas of boreholes, wells, springs, or ponds.
* Storage of intractable wastes -- A large volume of hazardous
waste is currently stored -- usually in steel drums on industrial
sites, awaiting the development of satisfactory disposal methods
-- because it is too toxic to be disposed of legally into air,
water or landfill sites. Most drums are stored in the open, and
many contain corrosive materials. There is an added risk of
hazard from fires, structural damage and vandalism of such toxic
waste stores. It is likely that some of these drums will corrode
and leak. In some countries, such stores must be registered with
authorities who, in turn, have a duty to inspect them.
4.6.2 An example of the effects of incorrect waste disposal
For years, the residents of Love Canal, a residential community near
Niagara Falls, New York, USA, complained of noxious vapours in their
basements, which they believed originated from a disused toxic waste
dump beneath their housing estate. Following prolonged rains and
heavy snows in the winter of 1977, water seeped into the dump-site,
causing it to overflow and forcing the chemicals out into the soil.
Two hundred and thirty-five families were evacuated, and Love Canal
was declared a federal disaster area, the first instance of a national
emergency being caused by chemical pollution. Later, more families
had to be moved when it was discovered that the toxic waste was
migrating further afield via underground streams.
Almost 22 000 tonnes of chemicals had been buried in Love Canal; the
canal had been capped with clay and covered with soil in 1953. The
clay cap was apparently damaged during later building work on the
site. Approximately 300 different chemical compounds were buried
there, although no records were kept of what, or how much, was dumped.
Studies now reveal that the Love Canal area experienced three times
the normal rate of miscarriages and three-and-a-half times the normal
rate of birth defects, most notably cleft palate, mental retardation
and club foot.
In addition, US$30 million have been spent on cleaning up the area,
and US$4.5 million on rehousing residents.
5. HOW CHEMICALS CAN POISON THE ENVIRONMENT - CHAPTER 5
This chapter is a background to chapter 5 of the HSG: "Hazards for the
Environment and their Prevention".
Almost any industrial or agricultural activity in which chemicals are
used will, by its very nature, have an impact on the environment (see
Fig. 49).
Figure 49. How chemicals can affect the environment
(from HSG 35: Phosphorus Trichloride)
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Phosphorus trichloride and phosphorus oxychloride hydrolyse in water
to form hydrochloric and phosphorous or phosphoric acids. The toxic
effects on organisms in the environment depend on the extent to which
the hydrogen ion is diluted and buffered; organisms die if they come
in contact with undiluted material.
In general, an environmental hazard will occur only in the immediate
area of a spillage or if disposed of improperly. Recovery of soil
and surface waters is rapid, and there are no residual effects on
the environment.
5.1 How do chemicals get into the environment?
Chemicals are released from the workplace into the environment in the
form of liquid, dust, fumes or gas. Such releases can be planned
(part of the work process, e.g. smokestack emissions, exhaust
ventilation, waste water into rivers, etc.) or unplanned (accidental).
Chemicals can also enter the environment during transport (e.g. from
the site of manufacture to the site of use), during their intended use
(e.g. pesticide spraying) or through disposal in landfills and
waterways. Chemicals also enter the environment through
non-occupational activities. Examples include exhaust fumes from motor
vehicles. Additionally, naturally occurring chemicals such as metals
in ores may be released by natural processes.
Chemicals released as smoke and dust from factory chimneys will
eventually fall to the earth's surface as dust and in rain. Wind
speed, air currents and other climatic conditions will determine when
and where the chemicals will be deposited. Some will settle on water
in lakes, rivers and streams, eventually passing into sediments and
nearby soils. Others may be transported over long distances and may
even affect the chemical and physical processes of the atmosphere and
climate. For example, the effects of sulfur and nitrogen oxides
released in industrial areas have contributed to acid rain, often
falling in countries other than those in which they were emitted.
Emissions of carbon dioxide, methane, ozone, etc. are thought to
contribute to global warming.
Once chemicals are released into the environment in this way, humans
and animals will be exposed to them through breathing contaminated
air, from direct contact with contaminated soil and water, through
eating plants grown on contaminated soils, by drinking contaminated
water supplies or by consuming food from contaminated animals (see
Fig. 50).
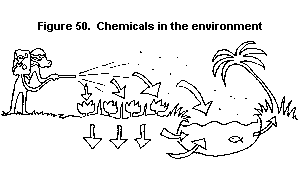 5.2 How do chemicals affect the environment?
Just as humans and animals can be affected by exposure to toxic
chemicals (as outlined in Chapter 2), chemicals can also have an
impact on the environment and the organisms living in the different
environmental media -- the air, water and soil.
5.2.1 Air
Many chemical pollutants in the air begin to have an effect on plants
and animals at extremely low concentrations, at even less than 1 µg
(or one-millionth of a gram) of chemical per cubic metre of air.
Plants and animals vary widely in their sensitivity to chemicals in
the air.
Fluorides released from aluminium smelters are an example of chronic
air pollutants that have caused great damage to livestock over long
periods of time. Animals exposed to excessive fluoride pollution
develop tooth, bone and joint diseases; they become unable to chew
their food or move. As a result, they lose weight and eventually die.
Fluoride builds up in the teeth, bones and tissues of all grazing
animals and has been found in large amounts in deer, rabbits and birds
near aluminium smelters. Humans can also be affected.
Fluorides also affect plants, causing leaves to die, damage to fruit,
a slowing of growth and the failure of seed to sprout.
Plants that are sensitive to fluoride pollution, such as grape vines,
begin to suffer significant damage if fluoride concentrations in air
exceed 0.5 µg/m3.
Environmental contamination by chemicals can also occur following
massive accidental releases, with large-scale immediate and long-term
consequences. For example, at least 2500 people died and 200 000
people were injured when methyl isocyanate gas was released from the
Union Carbide pesticide plant in Bhopal, India, on 3 December 1984.
According to an International Trade Union report, the disaster was
attributable to poor process design, dangerous operating procedures,
lack of proper maintenance, faulty equipment and major cuts in
staffing levels, crew sizes, worker training programmes and skilled
supervision. Smaller releases of toxic chemicals had occurred,
leading to one death and numerous injuries at the Union Carbide plant
at Bhopal before the 1984 disaster, but little or nothing was done to
correct these problems, despite vigorous protests by the union
representing Union Carbide workers.
Apart from the immediate deaths, reports indicate that other long-term
health effects are being suffered by the survivors of the disaster.
These include lung problems and reproductive effects such as an
increased rate of miscarriages and stillbirths.
5.2.2 Water and soil
As with chemicals in the air, very low concentrations of heavy metals
and other chemicals in water can have an enormous impact, particularly
on fish and other aquatic species.
The effect a chemical pollutant has in water depends on the nature of
the pollutant and on factors such as acidity (pH), temperature, water
hardness, the presence of organic materials such as algae and weeds
and the oxygen content of the water. For example, the toxicity of
certain (heavy) metals tends to increase as the acidity of the water
increases.
5.3 Factors affecting a chemical's environmental impact
The impact of a chemical on organisms and ecological systems will
depend on the chemical's environmental transport, persistence,
bioaccumulation and toxicity.
* Environmental transport, or mobility, refers to the ease
with which a substance moves from one environmental medium to
another (air, water, soil, sediment, etc.) by such processes as
evaporation, leaching, weathering, etc.
* Environmental persistence is one aspect of a chemical's
environmental fate, i.e. what happens to it when it enters the
environment. Once in the environment, a substance can degrade,
in which case it is generally assumed to be non-hazardous to the
environment, at least on a large scale, even though there may be
local effects in the short term. On the other hand, if a
chemical is not readily degradable, it can persist in the
environment for long periods and become widely dispersed.
Environmental persistence is therefore the opposite of
degradation and can be measured in terms of half-life or
half-time of a chemical in an environmental medium, i.e. the time
required for the chemical to be reduced to half its original
amount. Local conditions may affect a chemical's persistence.
For instance, a chemical may persist for longer periods in colder
climates, whereas it may be readily broken down in sunlight and
at higher temperatures.
The organochlorine pesticides are a group of chemicals well known for
their persistence. A study found that after only one application of
the pesticide aldrin to soil, more than 34% was found to be present in
the soil five years later. Most of the remaining 66% must therefore
have remained in water, air or soil, either as unchanged aldrin or in
the form of other closely related chemicals formed by the
decomposition of aldrin. The process by which a chemical is modified
by a living organism is called biotransformation.
Bioaccumulation is the accumulation of a chemical by an organism to
a concentration that exceeds that of the surrounding environment. For
example, the concentration of aldrin in the tissues of experimental
snails has been found to be nearly 5000 times the concentration of
aldrin in the water in which the snails live. The more a chemical
persists, the greater the potential for bioaccumulation. Chemicals in
the environment can become increasingly concentrated in the tissues of
animals higher up in the food-chain, a process known as
biomagnification.
The octanol/water partition coefficient of a chemical (see Chapter 1
for a discussion of this value) is a good indicator of its
bioaccumulation potential: the higher the coefficient, the more a
chemical will tend to bioaccumulate.
* Toxicity. The impact of a chemical on the environment,
resulting from its toxicity, is more complex. Chemical
pollutants affect some environmental media more than others,
whether these are air, surface water, groundwater, soil or
sediments. As explained in Chapter 2, different species will
react in varying ways and to different degrees to the same
chemical. What may be highly toxic to aquatic life may not be
toxic to birds, for instance, even at the same dose. Toxicity
forms the basis of use of pesticides. Pesticides are applied in
order to damage or kill a target organism (i.e. the organism that
is subject to the action of the pesticide). But it is seldom or
never possible to limit the effects to target organisms.
Pesticides are therefore often perceived as a particular threat
to health and the environment.
By determining the effects of a chemical on different species,
toxicity studies are able to evaluate the environmental impact of that
chemical. The effects to be determined should include acute lethality
(oral, dermal, inhalation LD50 and LC50); sublethal effects on
non-mammals (soil organisms, bees, birds, etc.), mammals and plants
(usually expressed as the percentage of growth reduction as a result
of exposure to the chemical in water, air or soil); mutagenicity/
genotoxicity; teratogenicity; and carcinogenicity.
5.4 The importance of prevention
Once chemicals have been released into the environment, little or
nothing can be done to remove them. Many chemicals will continue to
damage animals and plants and poison food and water for years or
decades. Therefore, it is essential that chemical pollution of the
environment be prevented at its source -- i.e., wherever chemicals are
manufactured or used. Good occupational hygiene principles should
always be applied when using or handling any chemicals at work.
Chemicals must be controlled not simply in order to ensure compliance
with government standards (exposure limits, etc.; see Chapter 7), but
to prevent chemical contamination -- to the maximum extent possible --
of the general environment. In the first instance, this means
judicious use of less toxic and/or more readily degradable chemicals
and the use of more selective pesticides. Chemical processes should
be enclosed as far as possible to prevent release of chemicals as
dust, fumes or gas. Dust, fumes and gas that are generated during a
process such as grinding or welding should be captured by local
exhaust ventilation. Contaminated air from such processes, together
with contaminated air from ventilation of closed systems and general
ventilation, should not be allowed to escape to the atmosphere inside
the workplace, or outside, without first having had the contamination
removed. Some methods and principles for the prevention of
environmental chemical pollution are given in Chapter 4 of this Manual
(see sections on Storage, Spillage and Disposal).
6. SUMMARY OF CHEMICAL SAFETY INFORMATION - CHAPTER 6
Most HSGs contain a Summary of Chemical Safety Information or an ICSC
that summarizes information and advice on the relevant chemical. It
is designed for easy, quick reference for health workers concerned
with, and users of, chemicals. It should be displayed near entrances
to areas where exposure to the chemical in question could potentially
occur and, if possible, on processing equipment and containers.
A sample is given below; the terms used are defined in Chapters 1 and
2 of this Manual. All persons potentially exposed to chemicals should
have the instructions on the relevant ICSCs clearly explained to them.
1,4-DIOXANE ICSC: 0041
1,4-DIOXANE
1,4-Diethylene dioxide
1,4-Diethyleneoxide
C4H8O2
Molecular mass: 88.1
5.2 How do chemicals affect the environment?
Just as humans and animals can be affected by exposure to toxic
chemicals (as outlined in Chapter 2), chemicals can also have an
impact on the environment and the organisms living in the different
environmental media -- the air, water and soil.
5.2.1 Air
Many chemical pollutants in the air begin to have an effect on plants
and animals at extremely low concentrations, at even less than 1 µg
(or one-millionth of a gram) of chemical per cubic metre of air.
Plants and animals vary widely in their sensitivity to chemicals in
the air.
Fluorides released from aluminium smelters are an example of chronic
air pollutants that have caused great damage to livestock over long
periods of time. Animals exposed to excessive fluoride pollution
develop tooth, bone and joint diseases; they become unable to chew
their food or move. As a result, they lose weight and eventually die.
Fluoride builds up in the teeth, bones and tissues of all grazing
animals and has been found in large amounts in deer, rabbits and birds
near aluminium smelters. Humans can also be affected.
Fluorides also affect plants, causing leaves to die, damage to fruit,
a slowing of growth and the failure of seed to sprout.
Plants that are sensitive to fluoride pollution, such as grape vines,
begin to suffer significant damage if fluoride concentrations in air
exceed 0.5 µg/m3.
Environmental contamination by chemicals can also occur following
massive accidental releases, with large-scale immediate and long-term
consequences. For example, at least 2500 people died and 200 000
people were injured when methyl isocyanate gas was released from the
Union Carbide pesticide plant in Bhopal, India, on 3 December 1984.
According to an International Trade Union report, the disaster was
attributable to poor process design, dangerous operating procedures,
lack of proper maintenance, faulty equipment and major cuts in
staffing levels, crew sizes, worker training programmes and skilled
supervision. Smaller releases of toxic chemicals had occurred,
leading to one death and numerous injuries at the Union Carbide plant
at Bhopal before the 1984 disaster, but little or nothing was done to
correct these problems, despite vigorous protests by the union
representing Union Carbide workers.
Apart from the immediate deaths, reports indicate that other long-term
health effects are being suffered by the survivors of the disaster.
These include lung problems and reproductive effects such as an
increased rate of miscarriages and stillbirths.
5.2.2 Water and soil
As with chemicals in the air, very low concentrations of heavy metals
and other chemicals in water can have an enormous impact, particularly
on fish and other aquatic species.
The effect a chemical pollutant has in water depends on the nature of
the pollutant and on factors such as acidity (pH), temperature, water
hardness, the presence of organic materials such as algae and weeds
and the oxygen content of the water. For example, the toxicity of
certain (heavy) metals tends to increase as the acidity of the water
increases.
5.3 Factors affecting a chemical's environmental impact
The impact of a chemical on organisms and ecological systems will
depend on the chemical's environmental transport, persistence,
bioaccumulation and toxicity.
* Environmental transport, or mobility, refers to the ease
with which a substance moves from one environmental medium to
another (air, water, soil, sediment, etc.) by such processes as
evaporation, leaching, weathering, etc.
* Environmental persistence is one aspect of a chemical's
environmental fate, i.e. what happens to it when it enters the
environment. Once in the environment, a substance can degrade,
in which case it is generally assumed to be non-hazardous to the
environment, at least on a large scale, even though there may be
local effects in the short term. On the other hand, if a
chemical is not readily degradable, it can persist in the
environment for long periods and become widely dispersed.
Environmental persistence is therefore the opposite of
degradation and can be measured in terms of half-life or
half-time of a chemical in an environmental medium, i.e. the time
required for the chemical to be reduced to half its original
amount. Local conditions may affect a chemical's persistence.
For instance, a chemical may persist for longer periods in colder
climates, whereas it may be readily broken down in sunlight and
at higher temperatures.
The organochlorine pesticides are a group of chemicals well known for
their persistence. A study found that after only one application of
the pesticide aldrin to soil, more than 34% was found to be present in
the soil five years later. Most of the remaining 66% must therefore
have remained in water, air or soil, either as unchanged aldrin or in
the form of other closely related chemicals formed by the
decomposition of aldrin. The process by which a chemical is modified
by a living organism is called biotransformation.
Bioaccumulation is the accumulation of a chemical by an organism to
a concentration that exceeds that of the surrounding environment. For
example, the concentration of aldrin in the tissues of experimental
snails has been found to be nearly 5000 times the concentration of
aldrin in the water in which the snails live. The more a chemical
persists, the greater the potential for bioaccumulation. Chemicals in
the environment can become increasingly concentrated in the tissues of
animals higher up in the food-chain, a process known as
biomagnification.
The octanol/water partition coefficient of a chemical (see Chapter 1
for a discussion of this value) is a good indicator of its
bioaccumulation potential: the higher the coefficient, the more a
chemical will tend to bioaccumulate.
* Toxicity. The impact of a chemical on the environment,
resulting from its toxicity, is more complex. Chemical
pollutants affect some environmental media more than others,
whether these are air, surface water, groundwater, soil or
sediments. As explained in Chapter 2, different species will
react in varying ways and to different degrees to the same
chemical. What may be highly toxic to aquatic life may not be
toxic to birds, for instance, even at the same dose. Toxicity
forms the basis of use of pesticides. Pesticides are applied in
order to damage or kill a target organism (i.e. the organism that
is subject to the action of the pesticide). But it is seldom or
never possible to limit the effects to target organisms.
Pesticides are therefore often perceived as a particular threat
to health and the environment.
By determining the effects of a chemical on different species,
toxicity studies are able to evaluate the environmental impact of that
chemical. The effects to be determined should include acute lethality
(oral, dermal, inhalation LD50 and LC50); sublethal effects on
non-mammals (soil organisms, bees, birds, etc.), mammals and plants
(usually expressed as the percentage of growth reduction as a result
of exposure to the chemical in water, air or soil); mutagenicity/
genotoxicity; teratogenicity; and carcinogenicity.
5.4 The importance of prevention
Once chemicals have been released into the environment, little or
nothing can be done to remove them. Many chemicals will continue to
damage animals and plants and poison food and water for years or
decades. Therefore, it is essential that chemical pollution of the
environment be prevented at its source -- i.e., wherever chemicals are
manufactured or used. Good occupational hygiene principles should
always be applied when using or handling any chemicals at work.
Chemicals must be controlled not simply in order to ensure compliance
with government standards (exposure limits, etc.; see Chapter 7), but
to prevent chemical contamination -- to the maximum extent possible --
of the general environment. In the first instance, this means
judicious use of less toxic and/or more readily degradable chemicals
and the use of more selective pesticides. Chemical processes should
be enclosed as far as possible to prevent release of chemicals as
dust, fumes or gas. Dust, fumes and gas that are generated during a
process such as grinding or welding should be captured by local
exhaust ventilation. Contaminated air from such processes, together
with contaminated air from ventilation of closed systems and general
ventilation, should not be allowed to escape to the atmosphere inside
the workplace, or outside, without first having had the contamination
removed. Some methods and principles for the prevention of
environmental chemical pollution are given in Chapter 4 of this Manual
(see sections on Storage, Spillage and Disposal).
6. SUMMARY OF CHEMICAL SAFETY INFORMATION - CHAPTER 6
Most HSGs contain a Summary of Chemical Safety Information or an ICSC
that summarizes information and advice on the relevant chemical. It
is designed for easy, quick reference for health workers concerned
with, and users of, chemicals. It should be displayed near entrances
to areas where exposure to the chemical in question could potentially
occur and, if possible, on processing equipment and containers.
A sample is given below; the terms used are defined in Chapters 1 and
2 of this Manual. All persons potentially exposed to chemicals should
have the instructions on the relevant ICSCs clearly explained to them.
1,4-DIOXANE ICSC: 0041
1,4-DIOXANE
1,4-Diethylene dioxide
1,4-Diethyleneoxide
C4H8O2
Molecular mass: 88.1
 CAS # 123-91-1
RTECS # JG8225000
ICSC # 0041
ON # 1165
EC # 603-024-00-5
TYPES OF
HAZARDS/ ACTIVE HAZARDS/ FIRST AID/
EXPOSURE SYMPTOMS PREVENTION FIRE FIGHTING
FIRE Highly flammable. NO open flames, NO sparks, Powder, alcohol-resistant
and NO smoking. NO contact foam, water spray, carbon
with strong oxidants. NO dioxide.
contact with hot surfaces.
EXPLOSION Vapour/air mixtures are Closed system, ventilation, In case of fire: keep drums,
explosive. Risk of fire and explosion-proof electrical etc., cool by spraying with
explosion on contact with equipment and lighting. water.
incompatible materials: see Prevent build-up of
Chemical Dangers. electrostatic charges (e.g.,
by grounding) (see Notes).
EXPOSURE AVOID ALL CONTACT!
INHALATION Headache. Nausea. Cough. Sore Ventilation, local exhaust, Fresh air, rest. Refer for
throat. Abdominal pain. or breathing protection. medical attention.
Dizziness. Drowsiness.
Vomiting. Unconsciousness.
SKIN MAY BE ABSORBED! Redness. Protective gloves. Protective Remove contaminated clothes.
Clothing. Rinse skin with plenty of
water or shower.
EYES Redness. Pain. Watery eyes. Face shield or eye protection First rinse with plenty of
in combination with breathing water for several minutes
protection. (remove contact lenses if
easily possible), then take
to a doctor.
INGESTION (further see Inhalation). Do not eat, drink, or smoke Rinse mouth. Refer for
during work. medical attention.
SPILLAGE DISPOSAL STORAGE PACKAGING & LABELLING
Collect Leaking and spilled liquid in Fireproof. Separated from strong Airtight.
sealable containers as far as oxidants, strong acids. Cool. Dry.
possible. Wash away reminder with Keep in the dark. Store only if F symbol
plenty of water (extra personal stabilized. Xn symbol
protection: complete protective
clothing including self-contained R: 11-19-36/37-40
breathing apparatus). S: 16-36/37
UN Hazard Class: 3
UN Packing Group: II
SEE IMPORTANT INFORMATION ON BACK
ICSC: 0041 1.1 Prepared in the context of cooperation between the International Programme on
Chemical Safety % the Commission of the European Communities (C) IPCS CEC 1993
PHYSICAL STATE; APPEARANCE: INHALATION RISK:
COLOURLESS LIQUID, WITH CHARACTERISTIC ODOUR. A harmful contamination of the air can be
reached rather quickly on evaporation of this
IMPORTANT PHYSICAL DANGERS: substance at 20°C on spraying much faster.
DATA
The vapour is heavier than air and may travel EFFECTS OF SHORT-TERM EXPOSURE:
along the ground; distant ignition possible.
As a result of flow, agitation. etc., The substance irritates the eyes and the
electrostatic charges can be generated. respiratory tract. The substance may cause
effects on the central nervous system, Liver
CHEMICAL DANGERS: and kidneys. Exposure to high vapour
concentrations my result in unconsciousness.
The substance can form explosive peroxides.
Reacts vigorously with strong oxidants and EFFECTS OF LONG-TERN OR REPEATED EXPOSURE:
concentrated strong acids. Reacts explosively
with some catalysts (e.g., Raney-nickel above The Liquid defats the skin. This substance is
210°C). Attacks many plastics. probably carcinogenic to humans.
OCCUPATIONAL EXPOSURE LIMITS (OELs):
TLV: 25 ppm; 90 mg/m3 (as TWA) (skin)
(ACGIH 1992-1993).
ROUTES OF EXPOSURE:
The substance can be absorbed into the body by
inhalation of this vapour and through the skin.
PHYSICAL Boiling point: 101°C Flash point: 12°C
PROPERTIES Melting point: 12°C Auto-ignition temperature: 180°C
Relative density (water = 1): 1.03 Explosive limits, vol% in air: 2-22.5
Solubility in water: Miscible Octanol/water partition coefficient
Vapour pressure, kPa at 20°C: 4.1 as log Pow: -0.42
Relative vapour density (air = 1): 3.0
Relative density of the vapour/
air-mixture at 20°C (air = 1): 1.08
ENVIRONMENTAL
DATA
NOTES
Use of alcoholic beverages enhances the harmful effect.
Depending on the degree of exposure, periodic medical examination is indicated.
The odour warning when the exposure limit value is exceeded is insufficient.
Check for peroxides prior to distillation; render harmless if positive.
EXPLOSION/PREVENTION: Do NOT use compressed air for filling, discharging, or handling. Use non-sparking handtools.
Transport Emergency Card: TEC (R)-546
NFPA Code: H2; F3; R1
ADDITIONAL INFORMATION
ICSC: 0041 1.1 (C) IPCS, CEC, 1993 1,4-DIOXANE
7. REGULATIONS, GUIDELINES AND STANDARDS - CHAPTER 7
Chapter 7 of the HSG outlines the regulations pertaining to particular
chemicals that apply in certain countries. Restrictions and safety
precautions may operate in some of the countries where a certain
chemical is registered. A number of standards may be listed in the
HSGs, as requirements sometimes vary between countries.
One of the standards that most workers will come across in the
workplace is the exposure limit, so this term is discussed in detail
below.
7.1 Exposure limits
The term exposure limit was introduced by the ILO in 1977. It is a
general expression that embraces the various words currently used to
refer to air quality limit values in the workplace and which are
referred to in the HSG (see Fig. 51).
7.1.1 Threshold limit value
The term threshold limit value (TLV), used by the American Conference
of Governmental Industrial Hygienists, refers to the amount of a
substance found in workplace air to which it is believed nearly all
workers may be repeatedly exposed day after day without adverse
effect. Because of wide variation in individual susceptibility,
however, a small percentage of workers may experience discomfort from
some substances at or below the threshold limit value. A smaller
percentage may be more seriously affected by aggravation of a
pre-existing condition or by development of an occupational illness.
Threshold limit values may be expressed as a Time-weighted Average
(TLV-TWA), i.e. an average concentration for a normal eight-hour
working day and a 40-hour working week; or a Short-term Exposure
Limit (TLV-STEL), which is a 15-minute time-weighted average
exposure that should not be exceeded at any time during a working day
even if the eight-hour time-weighted average is within the TLV.
Exposures at the STEL should not exceed 15 minutes in duration and
should not be repeated more than four times each day; there should be
at least 60 minutes between successive exposures at the STEL. The
STEL values are useful for protecting against the health effects of
fast-acting substances such as solvents. A ceiling value -- i.e. an
air concentration of a chemical that should not be exceeded for even
an instant -- is often given for chemicals that affect health after
only a very short exposure. This applies to chemicals that are
capable of producing severe irritation, lung oedema (fluid in the
lungs) or an allergic sensitization.
CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Australia Threshold limit value (TWA) 19 mg/m3 1983
Germany, Federal Maximum worksite concentration (TWA) 19 mg/m3 1985
Republic of Short-term exposure limit (5-min) 38 mg/m3 1985
Hungary Maximum allowable concentration (TWA) 5 mg/m3 1978
Short-term exposure limit (30-min) 10 mg/m3 1978
Japan Maximum allowable concentration (TWA) 19 mg/m3 1981
Sweden Highest limit value (TWA) 4 mg/m3 b 1985
Short-term exposure limit (15-min TWA) 8 mg/m3 1985
USA (OSHA) Permissible exposure level (TWA) 19 mg/m3 1974
USA (ACGIH) Threshold limit value (TWA) 19 mg/m3 1984
Short-term exposure limit (TWA) 38 mg/m3 1984
USSR Ceiling value 0.3 mg/m3 1977
AIR Ambient Czechoslovakia Maximum allowable concentration
average per day 0.01 mg/m3 1981
average per 0.5 h 0.01 mg/m3 1981
USSR Maximum allowable concentration
average per day 0.003 mg/m3 1984
once per day 0.01 mg/m3 1984
Figure 51. Exposure limit values (from HSG 88: Phenol)
7.1.2 Maximum allowable concentration
The term maximum allowable concentration (MAC) is used widely in the
former Soviet Union and Central and Eastern European countries, the
Netherlands and Germany. In the former Soviet Union, MAC is defined
as that concentration of a harmful substance in the air of the
workplace which, in the case of daily exposure at work for eight
hours, throughout a worker's entire working life, will not cause any
disease or deviation from a normal state of health to that worker or
his or her offspring, detectable by current methods of investigation,
whether during the work itself or in the long term.
7.1.3 Other terms for occupational exposure limits
Just as the regulations and guidelines of countries are subject to
change, so too is the terminology concerning exposure limits.
Although TLV and MAC are terms that are well known because of their
wide usage over a long time, some countries have developed their own
terminology or evolved their own standards, criteria and methods for
determining exposure limits. These vary in practice between the
stringent concept of MAC and the more elastic approach of TLV, which
makes allowances for reversible clinical changes.
Some of the expressions currently used in addition to TLV and MAC and
that are used in the HSGs include:
* Maximum permissible concentration (MPC), used in Argentina,
Finland and Poland, among others; this term is also used in
relation to a chemical's concentration in drinking-water in
several countries, including Japan, Germany and the USA.
* Permissible exposure limit (PEL), used in the United Kingdom
and by the US Occupational Safety and Health Administration, etc.
* Technical guiding concentration (TRK) is a term applied in
Germany.
Information on the methodology, scientific basis and any other aspects
related to the establishment of exposure limits should be requested
from the relevant national bodies of your country.
While the terms used above may give the impression that exposure to
chemicals below their exposure limit is harmless and without adverse
health effects, it should be stated that exposure limits are
approximate guidelines only as to what is safe and what is dangerous.
They are not necessarily safe limits.
Factors that must be taken into account when applying exposure limits
and other environmental control values include:
* Length of work shift and working week -- Exposure limit values
assume that workers will be exposed to chemicals in the workplace
only for eight hours per day, five days per week. If each shift
is longer than eight hours, or if more than 40 hours are worked
in each week, the exposure limit values must be lowered. If
workers live near their workplace, environmental pollution may
add to exposure levels. This should be taken into account when
assessing exposure risk.
* Environmental conditions -- Environmental or physical factors
such as heat, ultraviolet and ionizing radiation and humidity may
place added stress on the body. The effects of exposure at a
threshold limit may therefore be altered. Most of these stresses
act adversely to increase the toxic response of a substance. The
safety factors for most substances do not permit significant
deviation.
* Combined exposures -- The combined health effects of exposure to
two or more chemicals that produce similar effects are often much
greater than the additive effects of separate exposure to each of
the chemicals. This is known as synergism (see Chapter 2).
Synergism may also occur if workers are taking prescribed or
non-prescribed drugs. Many prescribed drugs such as
contraceptive pills or sleeping tablets can exacerbate the
negative health effects of workplace chemicals. Similarly,
alcohol can worsen the effects of solvents. A well-known example
of synergism is the relationship between tobacco smoking and
asbestos exposure. Smokers who work with asbestos have been shown
to have a probability of dying from lung cancer that is greater
than the sum of the probabilities arising from smoking and from
working with asbestos.
Chemicals that are handled in the workplace may not be pure, and
the contaminants may pose a greater health hazard than the
chemical itself. This is often overlooked when exposure limits
are set.
* Workload -- Heavy workloads may increase the absorption of
chemicals present in the workplace air. When performing heavy
physical work, a worker will breathe faster and more deeply.
Therefore, more air (containing chemicals) will pass through the
worker's lungs. It has been estimated that for equal
concentrations of a substance, the amount taken in by the lungs
could vary from one work situation to another by as much as
five-fold.
* Reproductive effects -- Although greater efforts have been made
recently to set exposure limits sufficiently low to protect
workers and their offspring from reproductive health effects (see
Chapter 2), knowledge in this area is still quite limited, and
most exposure limits do not take reproductive hazards into
account.
* Route of entry -- Although inhalation of airborne dust or
droplets is the most common way in which chemicals enter the
body, some chemicals for which exposure limits have been set may
also enter by other routes such as the skin. In these cases, a
warning of this hazard appears in the HSG.
* Carcinogens -- Exposure to carcinogens should be kept as "low as
is technically feasible". This means that all available control
methods (i.e. substitution, enclosure, ventilation and personal
protective equipment when absolutely essential) should be used to
keep worker exposure to the carcinogenic chemical to an absolute
minimum.
* Average worker -- Exposure limits are set to protect the average
worker from the effects of chemicals. As most exposure limits
have been developed in the industrialized countries, this average
worker is often presumed to be a healthy, well-nourished, white
male. The clearest example of the average worker problem is that
of the female worker, who may react quite differently to exposure
to chemicals, such as solvents, because of her greater degree of
body fat. Women's higher risk of reproductive complications
should also be taken into account when exposure limits are set.
* Insufficient and sometimes biased information -- Many exposure
limits have been set without sufficient information. For
example, very few chemicals have been adequately tested over a
long period of time for their contribution to illnesses such as
heart disease and cancer or to genetic effects. Moreover, some
exposure limits are based largely on information developed and
evaluated by the chemical industry. In general, workers and
their representatives have little or no input into setting
exposure limits, even though they are most at risk.
7.1.4 Health-based exposure limits
If the protection of workers were the only consideration, most
exposure limits would be much more stringent. This is the case with
the WHO's "Recommended health-based occupational exposure limits",
which are based on toxicity data and their relevance to the protection
of workers' health. Recommended health-based occupational exposure
limits should not be seen as absolute levels above which effects will
occur and below which they will not occur. They are levels above
which the probability that adverse health effects will occur starts to
increase, and below which this probability is very low. Recommended
health-based exposure limits already exist for some metals, solvents,
pesticides, vegetable dusts and irritant gases (published in the WHO
Technical Report Series). These WHO health-based limits are generally
stricter and more protective than national safety standards. This is
because they are based on health considerations only. They may be
seen as the "ultimate goal" for standards. In each country they can
be used by representatives of government, employers and workers to set
their own limits, taking into account other factors such as
socioeconomic and technological constraints.
7.1.5 Exposure limits for food products
Many of the HSGs are for pesticide chemicals. In addition to
occupational exposure during manufacture and use of these products,
exposure may occur in the form of residues in and on food products.
The issue of whether any such residues pose a threat to health is
therefore of some concern.
Many governments and international bodies such as WHO and the Food and
Agriculture Organization of the United Nations (FAO) have developed
different standards and/or legally established limits to control
exposure to and ingestion of residues (see Fig. 52).
* Maximum residue limit (MRL) is the maximum concentration of a
pesticide residue (expressed in mg/kg) that is legally permitted
or recognized as acceptable in or on a food, an agricultural
commodity or animal feed.
* Acceptable daily intake (ADI) is the daily intake of a chemical
(expressed in mg/kg of body weight) that, during an entire
lifetime, appears to be without appreciable risk to the health of
the consumer on the basis of all known facts at the time of the
evaluation of the chemical. ADIs are established internationally
by the Joint FAO/WHO Meeting on Pesticide Residues (JMPR) and, in
the case of many countries, by the national regulatory
authorities (see Fig. 52).
7.2 Specific restrictions
A chemical may have special restrictions imposed on its use in any
country, mainly as a result of concern about its effects on health or
the environment. However, regulatory decisions taken in a certain
country can only be fully understood in the framework of the
legislation of that country. It does not therefore follow that a
banned or restricted chemical in one country will be banned, or have
the same restrictions imposed on it in other countries.
Figure 52. Exposure limits for food products in some countries
(from HSG 73: Rotenone)
7. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
7.1 Exposure limit values
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has not
reviewed the compound to establish an ADI. A threshold limit value,
time weighted average (TWA) for rotenone of 5 mg/m3 has been
recommended by the US ACGIH and adopted by several countries.
Some exposure limits for food products are given in Table 1.
Table 1. Exposure limits for food products in some countries
Country Food product Exposure Value Effective
limit (mg/kg) date
description
Austria All products 0.05 1988
Germany All products Maximum 0.1 1990
of plant origin residue limit
France Fruits and 0.05 1989
vegetables
Italy Specified 0.04 1985
fruits and
vegetables
Netherlands All products 0.05 1988
Asbestos, for instance, is a known human carcinogen -- it causes lung
cancer -- and has been effectively banned in Sweden and Denmark, but
its use is still permitted elsewhere. Some countries may have
restricted the use of certain types of asbestos (e.g. crocidolite or
blue asbestos) or certain processes (e.g. spraying of asbestos). In
other countries the use of asbestos is actively encouraged,
particularly for building and for piping. In all countries, its
health effects will be the same, but for various reasons -- mainly
economic or political -- some countries see fit to allow its continued
use.
If you suspect that you may be working with an imported dangerous
chemical, it would be worthwhile finding out what restrictions are
imposed on the chemical in the country of origin or in other
countries. The HSGs outline such restrictions (see Fig. 53).
7.3 Labelling, packaging and transport
Labelling of a chemical to warn of potential health hazards is based
on classification. Classification is concerned with both acute and
long-term effects of substances, not only with respect to systemic
toxicity (carcinogenicity, mutagenicity and teratogenicity), but also
with respect to allergic qualities and local effects on the skin and
eyes, as well as fire and explosion risks.
Some of the more commonly used terms for the purposes of
classification include harmful, toxic, very toxic, irritant,
corrosive, oxidizing material, etc. For substances that have
carcinogenic, mutagenic, allergenic or other subacute or chronic
effects, the classification of very toxic, toxic or harmful should be
used according to the magnitude of these effects.
Appropriate symbols are used to provide immediate visual
identification of the general nature and degree of hazard (see
Fig. 54). Label sizes, shapes, colours and borders will be specified,
and the HSG outlines the appropriate specifications (see Fig. 55).
This is especially important if chemicals are to be transported.
Figure 53. Restrictions imposed on chemical use in various countries
(from HSG 19: Pentachlorophenol)
Specific restrictions
The use of pentachlorophenol has been more and more restricted during
the last few years as a result of the increasing concern about the
potential health and environmental hazards of PCP and its impurities.
To mention a few:
* Sweden banned all use of PCP in 1977 and the Federal Republic of
Germany banned all use in 1987;
* the USA cancelled its registration for herbicidal and
anti-microbial use and for the preservation of wood in contact
with food, feed, domestic animals, and livestock. The sale and
use of PCP are restricted to certified applicators;
* the agricultural use of PCP has been suspended or restricted in,
among others, Canada, Denmark, German Democratic Republic, and
Japan;
* Canada and the Netherlands have suspended its use for indoor
wood treatment.
In addition to warning symbols, standard risk and safety phrases
should also be used to describe in detail the nature of the risk and
appropriate safety measures. Thus, a complete label should give the
name of the substance (including the internationally recognized
chemical name), the name and address of the manufacturer, distributor
or importer and whatever symbols and risk and safety phrases that may
be appropriate.
CAS # 123-91-1
RTECS # JG8225000
ICSC # 0041
ON # 1165
EC # 603-024-00-5
TYPES OF
HAZARDS/ ACTIVE HAZARDS/ FIRST AID/
EXPOSURE SYMPTOMS PREVENTION FIRE FIGHTING
FIRE Highly flammable. NO open flames, NO sparks, Powder, alcohol-resistant
and NO smoking. NO contact foam, water spray, carbon
with strong oxidants. NO dioxide.
contact with hot surfaces.
EXPLOSION Vapour/air mixtures are Closed system, ventilation, In case of fire: keep drums,
explosive. Risk of fire and explosion-proof electrical etc., cool by spraying with
explosion on contact with equipment and lighting. water.
incompatible materials: see Prevent build-up of
Chemical Dangers. electrostatic charges (e.g.,
by grounding) (see Notes).
EXPOSURE AVOID ALL CONTACT!
INHALATION Headache. Nausea. Cough. Sore Ventilation, local exhaust, Fresh air, rest. Refer for
throat. Abdominal pain. or breathing protection. medical attention.
Dizziness. Drowsiness.
Vomiting. Unconsciousness.
SKIN MAY BE ABSORBED! Redness. Protective gloves. Protective Remove contaminated clothes.
Clothing. Rinse skin with plenty of
water or shower.
EYES Redness. Pain. Watery eyes. Face shield or eye protection First rinse with plenty of
in combination with breathing water for several minutes
protection. (remove contact lenses if
easily possible), then take
to a doctor.
INGESTION (further see Inhalation). Do not eat, drink, or smoke Rinse mouth. Refer for
during work. medical attention.
SPILLAGE DISPOSAL STORAGE PACKAGING & LABELLING
Collect Leaking and spilled liquid in Fireproof. Separated from strong Airtight.
sealable containers as far as oxidants, strong acids. Cool. Dry.
possible. Wash away reminder with Keep in the dark. Store only if F symbol
plenty of water (extra personal stabilized. Xn symbol
protection: complete protective
clothing including self-contained R: 11-19-36/37-40
breathing apparatus). S: 16-36/37
UN Hazard Class: 3
UN Packing Group: II
SEE IMPORTANT INFORMATION ON BACK
ICSC: 0041 1.1 Prepared in the context of cooperation between the International Programme on
Chemical Safety % the Commission of the European Communities (C) IPCS CEC 1993
PHYSICAL STATE; APPEARANCE: INHALATION RISK:
COLOURLESS LIQUID, WITH CHARACTERISTIC ODOUR. A harmful contamination of the air can be
reached rather quickly on evaporation of this
IMPORTANT PHYSICAL DANGERS: substance at 20°C on spraying much faster.
DATA
The vapour is heavier than air and may travel EFFECTS OF SHORT-TERM EXPOSURE:
along the ground; distant ignition possible.
As a result of flow, agitation. etc., The substance irritates the eyes and the
electrostatic charges can be generated. respiratory tract. The substance may cause
effects on the central nervous system, Liver
CHEMICAL DANGERS: and kidneys. Exposure to high vapour
concentrations my result in unconsciousness.
The substance can form explosive peroxides.
Reacts vigorously with strong oxidants and EFFECTS OF LONG-TERN OR REPEATED EXPOSURE:
concentrated strong acids. Reacts explosively
with some catalysts (e.g., Raney-nickel above The Liquid defats the skin. This substance is
210°C). Attacks many plastics. probably carcinogenic to humans.
OCCUPATIONAL EXPOSURE LIMITS (OELs):
TLV: 25 ppm; 90 mg/m3 (as TWA) (skin)
(ACGIH 1992-1993).
ROUTES OF EXPOSURE:
The substance can be absorbed into the body by
inhalation of this vapour and through the skin.
PHYSICAL Boiling point: 101°C Flash point: 12°C
PROPERTIES Melting point: 12°C Auto-ignition temperature: 180°C
Relative density (water = 1): 1.03 Explosive limits, vol% in air: 2-22.5
Solubility in water: Miscible Octanol/water partition coefficient
Vapour pressure, kPa at 20°C: 4.1 as log Pow: -0.42
Relative vapour density (air = 1): 3.0
Relative density of the vapour/
air-mixture at 20°C (air = 1): 1.08
ENVIRONMENTAL
DATA
NOTES
Use of alcoholic beverages enhances the harmful effect.
Depending on the degree of exposure, periodic medical examination is indicated.
The odour warning when the exposure limit value is exceeded is insufficient.
Check for peroxides prior to distillation; render harmless if positive.
EXPLOSION/PREVENTION: Do NOT use compressed air for filling, discharging, or handling. Use non-sparking handtools.
Transport Emergency Card: TEC (R)-546
NFPA Code: H2; F3; R1
ADDITIONAL INFORMATION
ICSC: 0041 1.1 (C) IPCS, CEC, 1993 1,4-DIOXANE
7. REGULATIONS, GUIDELINES AND STANDARDS - CHAPTER 7
Chapter 7 of the HSG outlines the regulations pertaining to particular
chemicals that apply in certain countries. Restrictions and safety
precautions may operate in some of the countries where a certain
chemical is registered. A number of standards may be listed in the
HSGs, as requirements sometimes vary between countries.
One of the standards that most workers will come across in the
workplace is the exposure limit, so this term is discussed in detail
below.
7.1 Exposure limits
The term exposure limit was introduced by the ILO in 1977. It is a
general expression that embraces the various words currently used to
refer to air quality limit values in the workplace and which are
referred to in the HSG (see Fig. 51).
7.1.1 Threshold limit value
The term threshold limit value (TLV), used by the American Conference
of Governmental Industrial Hygienists, refers to the amount of a
substance found in workplace air to which it is believed nearly all
workers may be repeatedly exposed day after day without adverse
effect. Because of wide variation in individual susceptibility,
however, a small percentage of workers may experience discomfort from
some substances at or below the threshold limit value. A smaller
percentage may be more seriously affected by aggravation of a
pre-existing condition or by development of an occupational illness.
Threshold limit values may be expressed as a Time-weighted Average
(TLV-TWA), i.e. an average concentration for a normal eight-hour
working day and a 40-hour working week; or a Short-term Exposure
Limit (TLV-STEL), which is a 15-minute time-weighted average
exposure that should not be exceeded at any time during a working day
even if the eight-hour time-weighted average is within the TLV.
Exposures at the STEL should not exceed 15 minutes in duration and
should not be repeated more than four times each day; there should be
at least 60 minutes between successive exposures at the STEL. The
STEL values are useful for protecting against the health effects of
fast-acting substances such as solvents. A ceiling value -- i.e. an
air concentration of a chemical that should not be exceeded for even
an instant -- is often given for chemicals that affect health after
only a very short exposure. This applies to chemicals that are
capable of producing severe irritation, lung oedema (fluid in the
lungs) or an allergic sensitization.
CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Australia Threshold limit value (TWA) 19 mg/m3 1983
Germany, Federal Maximum worksite concentration (TWA) 19 mg/m3 1985
Republic of Short-term exposure limit (5-min) 38 mg/m3 1985
Hungary Maximum allowable concentration (TWA) 5 mg/m3 1978
Short-term exposure limit (30-min) 10 mg/m3 1978
Japan Maximum allowable concentration (TWA) 19 mg/m3 1981
Sweden Highest limit value (TWA) 4 mg/m3 b 1985
Short-term exposure limit (15-min TWA) 8 mg/m3 1985
USA (OSHA) Permissible exposure level (TWA) 19 mg/m3 1974
USA (ACGIH) Threshold limit value (TWA) 19 mg/m3 1984
Short-term exposure limit (TWA) 38 mg/m3 1984
USSR Ceiling value 0.3 mg/m3 1977
AIR Ambient Czechoslovakia Maximum allowable concentration
average per day 0.01 mg/m3 1981
average per 0.5 h 0.01 mg/m3 1981
USSR Maximum allowable concentration
average per day 0.003 mg/m3 1984
once per day 0.01 mg/m3 1984
Figure 51. Exposure limit values (from HSG 88: Phenol)
7.1.2 Maximum allowable concentration
The term maximum allowable concentration (MAC) is used widely in the
former Soviet Union and Central and Eastern European countries, the
Netherlands and Germany. In the former Soviet Union, MAC is defined
as that concentration of a harmful substance in the air of the
workplace which, in the case of daily exposure at work for eight
hours, throughout a worker's entire working life, will not cause any
disease or deviation from a normal state of health to that worker or
his or her offspring, detectable by current methods of investigation,
whether during the work itself or in the long term.
7.1.3 Other terms for occupational exposure limits
Just as the regulations and guidelines of countries are subject to
change, so too is the terminology concerning exposure limits.
Although TLV and MAC are terms that are well known because of their
wide usage over a long time, some countries have developed their own
terminology or evolved their own standards, criteria and methods for
determining exposure limits. These vary in practice between the
stringent concept of MAC and the more elastic approach of TLV, which
makes allowances for reversible clinical changes.
Some of the expressions currently used in addition to TLV and MAC and
that are used in the HSGs include:
* Maximum permissible concentration (MPC), used in Argentina,
Finland and Poland, among others; this term is also used in
relation to a chemical's concentration in drinking-water in
several countries, including Japan, Germany and the USA.
* Permissible exposure limit (PEL), used in the United Kingdom
and by the US Occupational Safety and Health Administration, etc.
* Technical guiding concentration (TRK) is a term applied in
Germany.
Information on the methodology, scientific basis and any other aspects
related to the establishment of exposure limits should be requested
from the relevant national bodies of your country.
While the terms used above may give the impression that exposure to
chemicals below their exposure limit is harmless and without adverse
health effects, it should be stated that exposure limits are
approximate guidelines only as to what is safe and what is dangerous.
They are not necessarily safe limits.
Factors that must be taken into account when applying exposure limits
and other environmental control values include:
* Length of work shift and working week -- Exposure limit values
assume that workers will be exposed to chemicals in the workplace
only for eight hours per day, five days per week. If each shift
is longer than eight hours, or if more than 40 hours are worked
in each week, the exposure limit values must be lowered. If
workers live near their workplace, environmental pollution may
add to exposure levels. This should be taken into account when
assessing exposure risk.
* Environmental conditions -- Environmental or physical factors
such as heat, ultraviolet and ionizing radiation and humidity may
place added stress on the body. The effects of exposure at a
threshold limit may therefore be altered. Most of these stresses
act adversely to increase the toxic response of a substance. The
safety factors for most substances do not permit significant
deviation.
* Combined exposures -- The combined health effects of exposure to
two or more chemicals that produce similar effects are often much
greater than the additive effects of separate exposure to each of
the chemicals. This is known as synergism (see Chapter 2).
Synergism may also occur if workers are taking prescribed or
non-prescribed drugs. Many prescribed drugs such as
contraceptive pills or sleeping tablets can exacerbate the
negative health effects of workplace chemicals. Similarly,
alcohol can worsen the effects of solvents. A well-known example
of synergism is the relationship between tobacco smoking and
asbestos exposure. Smokers who work with asbestos have been shown
to have a probability of dying from lung cancer that is greater
than the sum of the probabilities arising from smoking and from
working with asbestos.
Chemicals that are handled in the workplace may not be pure, and
the contaminants may pose a greater health hazard than the
chemical itself. This is often overlooked when exposure limits
are set.
* Workload -- Heavy workloads may increase the absorption of
chemicals present in the workplace air. When performing heavy
physical work, a worker will breathe faster and more deeply.
Therefore, more air (containing chemicals) will pass through the
worker's lungs. It has been estimated that for equal
concentrations of a substance, the amount taken in by the lungs
could vary from one work situation to another by as much as
five-fold.
* Reproductive effects -- Although greater efforts have been made
recently to set exposure limits sufficiently low to protect
workers and their offspring from reproductive health effects (see
Chapter 2), knowledge in this area is still quite limited, and
most exposure limits do not take reproductive hazards into
account.
* Route of entry -- Although inhalation of airborne dust or
droplets is the most common way in which chemicals enter the
body, some chemicals for which exposure limits have been set may
also enter by other routes such as the skin. In these cases, a
warning of this hazard appears in the HSG.
* Carcinogens -- Exposure to carcinogens should be kept as "low as
is technically feasible". This means that all available control
methods (i.e. substitution, enclosure, ventilation and personal
protective equipment when absolutely essential) should be used to
keep worker exposure to the carcinogenic chemical to an absolute
minimum.
* Average worker -- Exposure limits are set to protect the average
worker from the effects of chemicals. As most exposure limits
have been developed in the industrialized countries, this average
worker is often presumed to be a healthy, well-nourished, white
male. The clearest example of the average worker problem is that
of the female worker, who may react quite differently to exposure
to chemicals, such as solvents, because of her greater degree of
body fat. Women's higher risk of reproductive complications
should also be taken into account when exposure limits are set.
* Insufficient and sometimes biased information -- Many exposure
limits have been set without sufficient information. For
example, very few chemicals have been adequately tested over a
long period of time for their contribution to illnesses such as
heart disease and cancer or to genetic effects. Moreover, some
exposure limits are based largely on information developed and
evaluated by the chemical industry. In general, workers and
their representatives have little or no input into setting
exposure limits, even though they are most at risk.
7.1.4 Health-based exposure limits
If the protection of workers were the only consideration, most
exposure limits would be much more stringent. This is the case with
the WHO's "Recommended health-based occupational exposure limits",
which are based on toxicity data and their relevance to the protection
of workers' health. Recommended health-based occupational exposure
limits should not be seen as absolute levels above which effects will
occur and below which they will not occur. They are levels above
which the probability that adverse health effects will occur starts to
increase, and below which this probability is very low. Recommended
health-based exposure limits already exist for some metals, solvents,
pesticides, vegetable dusts and irritant gases (published in the WHO
Technical Report Series). These WHO health-based limits are generally
stricter and more protective than national safety standards. This is
because they are based on health considerations only. They may be
seen as the "ultimate goal" for standards. In each country they can
be used by representatives of government, employers and workers to set
their own limits, taking into account other factors such as
socioeconomic and technological constraints.
7.1.5 Exposure limits for food products
Many of the HSGs are for pesticide chemicals. In addition to
occupational exposure during manufacture and use of these products,
exposure may occur in the form of residues in and on food products.
The issue of whether any such residues pose a threat to health is
therefore of some concern.
Many governments and international bodies such as WHO and the Food and
Agriculture Organization of the United Nations (FAO) have developed
different standards and/or legally established limits to control
exposure to and ingestion of residues (see Fig. 52).
* Maximum residue limit (MRL) is the maximum concentration of a
pesticide residue (expressed in mg/kg) that is legally permitted
or recognized as acceptable in or on a food, an agricultural
commodity or animal feed.
* Acceptable daily intake (ADI) is the daily intake of a chemical
(expressed in mg/kg of body weight) that, during an entire
lifetime, appears to be without appreciable risk to the health of
the consumer on the basis of all known facts at the time of the
evaluation of the chemical. ADIs are established internationally
by the Joint FAO/WHO Meeting on Pesticide Residues (JMPR) and, in
the case of many countries, by the national regulatory
authorities (see Fig. 52).
7.2 Specific restrictions
A chemical may have special restrictions imposed on its use in any
country, mainly as a result of concern about its effects on health or
the environment. However, regulatory decisions taken in a certain
country can only be fully understood in the framework of the
legislation of that country. It does not therefore follow that a
banned or restricted chemical in one country will be banned, or have
the same restrictions imposed on it in other countries.
Figure 52. Exposure limits for food products in some countries
(from HSG 73: Rotenone)
7. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
7.1 Exposure limit values
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) has not
reviewed the compound to establish an ADI. A threshold limit value,
time weighted average (TWA) for rotenone of 5 mg/m3 has been
recommended by the US ACGIH and adopted by several countries.
Some exposure limits for food products are given in Table 1.
Table 1. Exposure limits for food products in some countries
Country Food product Exposure Value Effective
limit (mg/kg) date
description
Austria All products 0.05 1988
Germany All products Maximum 0.1 1990
of plant origin residue limit
France Fruits and 0.05 1989
vegetables
Italy Specified 0.04 1985
fruits and
vegetables
Netherlands All products 0.05 1988
Asbestos, for instance, is a known human carcinogen -- it causes lung
cancer -- and has been effectively banned in Sweden and Denmark, but
its use is still permitted elsewhere. Some countries may have
restricted the use of certain types of asbestos (e.g. crocidolite or
blue asbestos) or certain processes (e.g. spraying of asbestos). In
other countries the use of asbestos is actively encouraged,
particularly for building and for piping. In all countries, its
health effects will be the same, but for various reasons -- mainly
economic or political -- some countries see fit to allow its continued
use.
If you suspect that you may be working with an imported dangerous
chemical, it would be worthwhile finding out what restrictions are
imposed on the chemical in the country of origin or in other
countries. The HSGs outline such restrictions (see Fig. 53).
7.3 Labelling, packaging and transport
Labelling of a chemical to warn of potential health hazards is based
on classification. Classification is concerned with both acute and
long-term effects of substances, not only with respect to systemic
toxicity (carcinogenicity, mutagenicity and teratogenicity), but also
with respect to allergic qualities and local effects on the skin and
eyes, as well as fire and explosion risks.
Some of the more commonly used terms for the purposes of
classification include harmful, toxic, very toxic, irritant,
corrosive, oxidizing material, etc. For substances that have
carcinogenic, mutagenic, allergenic or other subacute or chronic
effects, the classification of very toxic, toxic or harmful should be
used according to the magnitude of these effects.
Appropriate symbols are used to provide immediate visual
identification of the general nature and degree of hazard (see
Fig. 54). Label sizes, shapes, colours and borders will be specified,
and the HSG outlines the appropriate specifications (see Fig. 55).
This is especially important if chemicals are to be transported.
Figure 53. Restrictions imposed on chemical use in various countries
(from HSG 19: Pentachlorophenol)
Specific restrictions
The use of pentachlorophenol has been more and more restricted during
the last few years as a result of the increasing concern about the
potential health and environmental hazards of PCP and its impurities.
To mention a few:
* Sweden banned all use of PCP in 1977 and the Federal Republic of
Germany banned all use in 1987;
* the USA cancelled its registration for herbicidal and
anti-microbial use and for the preservation of wood in contact
with food, feed, domestic animals, and livestock. The sale and
use of PCP are restricted to certified applicators;
* the agricultural use of PCP has been suspended or restricted in,
among others, Canada, Denmark, German Democratic Republic, and
Japan;
* Canada and the Netherlands have suspended its use for indoor
wood treatment.
In addition to warning symbols, standard risk and safety phrases
should also be used to describe in detail the nature of the risk and
appropriate safety measures. Thus, a complete label should give the
name of the substance (including the internationally recognized
chemical name), the name and address of the manufacturer, distributor
or importer and whatever symbols and risk and safety phrases that may
be appropriate.
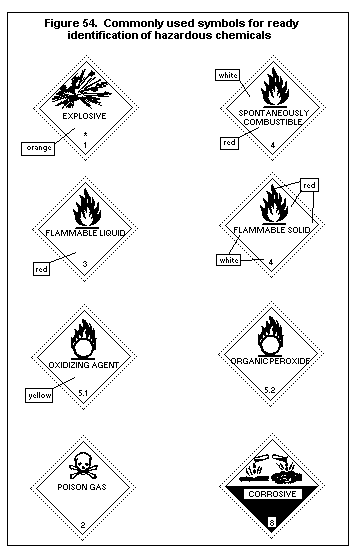 Figure 55. Labelling requirements (from HSG 9: Isobutanol)
Figure 55. Labelling requirements (from HSG 9: Isobutanol)
 The European Community legislation requires labelling as a
dangerous substance using the symbol:
The European Community legislation requires labelling as a
dangerous substance using the symbol:
 The label must read: flammable - harmful by inhalation; keep
away from sources of ignition - no smoking.
The European Community legislation on labelling of solvent
preparations classifies isobutanol in class II d for the
purpose of determining the label for preparations containing
isobutanol and other active ingredients (1980).
The criteria for classification are too complex for inclusion here.
In addition, there are many different classification and labelling
systems that depend on the user, i.e. the manufacturer, the shippers
or the employers, all of whom will have different areas of interest --
consumer products, shipping or environmental regulations or employee
protection. All of these are regulated by different national and
international agencies (national departments of transportation,
environmental protection agencies), local ordinances, the United
Nations Committee of Experts on the Transport of Dangerous Goods and
the International Maritime Organization.
It is generally agreed that proper application -- and therefore proper
interpretation -- of labelling of chemicals is often a difficult and
frustrating task. End-users are therefore advised to contact their
trade union or appropriate national or international association for
basic information on specific labelling.
8. GETTING FURTHER INFORMATION - CHAPTER 8
The information available to workers (and the general public) about
the health and safety effects of chemicals is often inadequate. Some
suggestions for obtaining further information are given below.
I. Identify and list the chemicals used in your workplace
II. Approach your employer for full information
It is the employer's responsibility to provide employees with full
health and safety information for all chemicals and chemical processes
used in a workplace. You should always ask your employer for health
and safety information as a first step.
You may wish to do this through your trade union or through the
workplace health and safety committee, if one exists. Always put your
requests in writing. If the employer's reply is inadequate, you
should inform your trade union.
III. Contact the chemical manufacturer or supplier
If your employer is unwilling or unable to provide the requested
information, it will be necessary to write to the chemical
manufacturer or supplier. Its name and address usually appear on the
label of the chemical container.
The manufacturer's or supplier's reply should include the following
information:
* the manufacturer's trade name for the chemical;
* any code numbers that appear on the label;
* the proper chemical or generic names of the chemical, if
available; and
* a brief description of how the chemical is used.
IV. Contact your government's Department of Occupational Health
and Safety
The government department responsible for occupational health and
safety may provide an information service that can assist you in
obtaining further information about particular chemicals.
V. Contact your trade union
There may be services concerned with the safety of chemicals run by
public interest groups or the trade union movement.
VI. Search for the information yourself
Many large public libraries and most university or technical college
libraries hold reference books on occupational health and safety and
the hazards of chemicals. Ask the librarian for help. The following
books may be particularly useful:
* IPCS Health and Safety Guides;
* IPCS Environmental Health Criteria (more detailed scientific
information on each chemical);
* ILO Encyclopaedia of Occupational Health and Safety;
* Reports from the International Register of Potentially Toxic
Chemicals (IRPTC/UNEP);
* Dangerous Properties of Industrial Materials -- Author: I. Sax;
* Patty's Industrial Hygiene and Toxicology;
* UN "Consolidated list of products whose consumption and/or sale
have been banned, withdrawn, severely restricted or not approved
by governments". Fourth ed. New York, United Nations Department
of International Economic and Social Affairs, 1991;
* ILO Convention No. 170 and Recommendation No. 177 on Safety in
the Use of Chemicals at Work;
* ILO Convention No. 174 and Recommendation No. 181 on Prevention
of Major Industrial Accidents;
* ILO Codes of Practice on "Safety in the Use of Chemicals at Work"
and "Prevention of Major Industrial Accidents"; and
* ILO. Safety and health in the use of chemicals at work: a
training manual.
Information on toxic effects of chemicals can also be obtained from
several databases, some of which are available on electronic media
such as CD-ROM. Many national and international trade unions have
access to such electronic information and can readily respond to
requests from affiliated unions and members.
The label must read: flammable - harmful by inhalation; keep
away from sources of ignition - no smoking.
The European Community legislation on labelling of solvent
preparations classifies isobutanol in class II d for the
purpose of determining the label for preparations containing
isobutanol and other active ingredients (1980).
The criteria for classification are too complex for inclusion here.
In addition, there are many different classification and labelling
systems that depend on the user, i.e. the manufacturer, the shippers
or the employers, all of whom will have different areas of interest --
consumer products, shipping or environmental regulations or employee
protection. All of these are regulated by different national and
international agencies (national departments of transportation,
environmental protection agencies), local ordinances, the United
Nations Committee of Experts on the Transport of Dangerous Goods and
the International Maritime Organization.
It is generally agreed that proper application -- and therefore proper
interpretation -- of labelling of chemicals is often a difficult and
frustrating task. End-users are therefore advised to contact their
trade union or appropriate national or international association for
basic information on specific labelling.
8. GETTING FURTHER INFORMATION - CHAPTER 8
The information available to workers (and the general public) about
the health and safety effects of chemicals is often inadequate. Some
suggestions for obtaining further information are given below.
I. Identify and list the chemicals used in your workplace
II. Approach your employer for full information
It is the employer's responsibility to provide employees with full
health and safety information for all chemicals and chemical processes
used in a workplace. You should always ask your employer for health
and safety information as a first step.
You may wish to do this through your trade union or through the
workplace health and safety committee, if one exists. Always put your
requests in writing. If the employer's reply is inadequate, you
should inform your trade union.
III. Contact the chemical manufacturer or supplier
If your employer is unwilling or unable to provide the requested
information, it will be necessary to write to the chemical
manufacturer or supplier. Its name and address usually appear on the
label of the chemical container.
The manufacturer's or supplier's reply should include the following
information:
* the manufacturer's trade name for the chemical;
* any code numbers that appear on the label;
* the proper chemical or generic names of the chemical, if
available; and
* a brief description of how the chemical is used.
IV. Contact your government's Department of Occupational Health
and Safety
The government department responsible for occupational health and
safety may provide an information service that can assist you in
obtaining further information about particular chemicals.
V. Contact your trade union
There may be services concerned with the safety of chemicals run by
public interest groups or the trade union movement.
VI. Search for the information yourself
Many large public libraries and most university or technical college
libraries hold reference books on occupational health and safety and
the hazards of chemicals. Ask the librarian for help. The following
books may be particularly useful:
* IPCS Health and Safety Guides;
* IPCS Environmental Health Criteria (more detailed scientific
information on each chemical);
* ILO Encyclopaedia of Occupational Health and Safety;
* Reports from the International Register of Potentially Toxic
Chemicals (IRPTC/UNEP);
* Dangerous Properties of Industrial Materials -- Author: I. Sax;
* Patty's Industrial Hygiene and Toxicology;
* UN "Consolidated list of products whose consumption and/or sale
have been banned, withdrawn, severely restricted or not approved
by governments". Fourth ed. New York, United Nations Department
of International Economic and Social Affairs, 1991;
* ILO Convention No. 170 and Recommendation No. 177 on Safety in
the Use of Chemicals at Work;
* ILO Convention No. 174 and Recommendation No. 181 on Prevention
of Major Industrial Accidents;
* ILO Codes of Practice on "Safety in the Use of Chemicals at Work"
and "Prevention of Major Industrial Accidents"; and
* ILO. Safety and health in the use of chemicals at work: a
training manual.
Information on toxic effects of chemicals can also be obtained from
several databases, some of which are available on electronic media
such as CD-ROM. Many national and international trade unions have
access to such electronic information and can readily respond to
requests from affiliated unions and members.
 1.1.1 Basic chemistry
All matter (i.e. liquids, solid substances and gases) is made of
elements. An element is the simplest sort of matter that can exist
by itself. So far, 106 different elements are known, including
oxygen, nitrogen, carbon, aluminium, copper and iron.
An atom is the smallest unit of an element that can still retain the
properties of the element. The atoms of the same element are exactly
alike, with the same mass, etc. The atoms of different elements,
however, have different masses and also differ in their ability to
combine with other atoms.
When atoms of one element combine chemically with atoms of the same
element or other elements, the result is a molecule. The process
whereby atoms combine and rearrange themselves is known as a chemical
reaction. Chemical reactions may occur slowly, taking hours or days
to complete, or they may occur suddenly or violently, as in an
explosion.
Combinations of two or more different elements are also called
chemical compounds. Substances such as water and carbon dioxide,
which can be broken down into their constituent elements -- hydrogen
and oxygen in the case of water, and carbon and oxygen in the case of
carbon dioxide -- are examples of compounds. There are nearly six
million different chemical compounds known, each with its own
particular combination of the atoms of various elements. This number
is increasing all the time. However, the number of potentially useful
chemical compounds is much less, and about 1500 chemicals account for
90% of the total volume of all chemical compounds produced.
A pure substance is a chemical that contains only a single chemical
compound or element, i.e. all the molecules are the same. The
compound that makes up the pure substance may contain the atoms of
many different types of elements, all combined chemically. A
mixture, on the other hand, is a substance that contains more than
one chemical compound or element, the separate constituents of which
will still have their own properties.
A suspension is a mixture of a liquid and tiny particles of a solid
substance (powder). A mist or aerosol is the mixture of tiny droplets
of a liquid, or tiny particles of a solid, in a gas. Although the
powder or droplets may be very fine, each particle of powder or each
droplet contains many millions of molecules of the chemical compound.
In a solution, individual molecules of one substance (the solute)
are dissolved in another substance (the solvent).
1.1.2 Terminology
Chemicals are identified in the HSGs as follows (see Fig. 2):
Common name -- This is the name that appears on the cover of the HSG
and is the commonly used name of the chemical in question. Sometimes
the common name refers to the elements that make up a chemical
compound, e.g. hydrogen sulfide contains elements of hydrogen and
sulfur.
Chemical formula -- The chemical formula uses symbols to represent
the atoms of each element contained in each molecule of the chemical
compound in question.
Table 1 gives the symbols of some of the more common elements. These
symbols come from the Latin name (Au for gold -- Latin: Aurum; Cu for
copper -- Latin: Cuprum). Sometimes the first letter is the same in
the English name (O for oxygen, C for carbon).
A formula for a molecule will also show a small number (or
subscript) below the level of the symbol, which indicates how many
atoms of that particular element are present in the molecule.
Using the example shown in Fig. 2, the chemical formula for phosphorus
trichloride -- PCl3 -- shows that it consists of one atom of
phosphorus and three of chlorine; the formula for phosphorus
oxychloride -- POCl3 -- shows that the chemical is made of one atom
of phosphorus, one of oxygen and three of chlorine.
Figure 2. Chemical identity (from HSG 35: Phosphorus Trichloride)
1.1 Identity
Phosphorus Phosphorus
Trichloride Oxychloride
Chemical formula: PCl3 POCl3
Chemical structure: Cl Cl
| |
Cl -- P Cl P == O
| |
Cl Cl
Common synonyms: Phosphorus chloride; Phosphorus
phosphorus chloride; oxytrichloride
trichlorophosphine
Abbreviations: Tri; "Pickle" Oxy; "Pockle"
CAS registry number:
1.1.1 Basic chemistry
All matter (i.e. liquids, solid substances and gases) is made of
elements. An element is the simplest sort of matter that can exist
by itself. So far, 106 different elements are known, including
oxygen, nitrogen, carbon, aluminium, copper and iron.
An atom is the smallest unit of an element that can still retain the
properties of the element. The atoms of the same element are exactly
alike, with the same mass, etc. The atoms of different elements,
however, have different masses and also differ in their ability to
combine with other atoms.
When atoms of one element combine chemically with atoms of the same
element or other elements, the result is a molecule. The process
whereby atoms combine and rearrange themselves is known as a chemical
reaction. Chemical reactions may occur slowly, taking hours or days
to complete, or they may occur suddenly or violently, as in an
explosion.
Combinations of two or more different elements are also called
chemical compounds. Substances such as water and carbon dioxide,
which can be broken down into their constituent elements -- hydrogen
and oxygen in the case of water, and carbon and oxygen in the case of
carbon dioxide -- are examples of compounds. There are nearly six
million different chemical compounds known, each with its own
particular combination of the atoms of various elements. This number
is increasing all the time. However, the number of potentially useful
chemical compounds is much less, and about 1500 chemicals account for
90% of the total volume of all chemical compounds produced.
A pure substance is a chemical that contains only a single chemical
compound or element, i.e. all the molecules are the same. The
compound that makes up the pure substance may contain the atoms of
many different types of elements, all combined chemically. A
mixture, on the other hand, is a substance that contains more than
one chemical compound or element, the separate constituents of which
will still have their own properties.
A suspension is a mixture of a liquid and tiny particles of a solid
substance (powder). A mist or aerosol is the mixture of tiny droplets
of a liquid, or tiny particles of a solid, in a gas. Although the
powder or droplets may be very fine, each particle of powder or each
droplet contains many millions of molecules of the chemical compound.
In a solution, individual molecules of one substance (the solute)
are dissolved in another substance (the solvent).
1.1.2 Terminology
Chemicals are identified in the HSGs as follows (see Fig. 2):
Common name -- This is the name that appears on the cover of the HSG
and is the commonly used name of the chemical in question. Sometimes
the common name refers to the elements that make up a chemical
compound, e.g. hydrogen sulfide contains elements of hydrogen and
sulfur.
Chemical formula -- The chemical formula uses symbols to represent
the atoms of each element contained in each molecule of the chemical
compound in question.
Table 1 gives the symbols of some of the more common elements. These
symbols come from the Latin name (Au for gold -- Latin: Aurum; Cu for
copper -- Latin: Cuprum). Sometimes the first letter is the same in
the English name (O for oxygen, C for carbon).
A formula for a molecule will also show a small number (or
subscript) below the level of the symbol, which indicates how many
atoms of that particular element are present in the molecule.
Using the example shown in Fig. 2, the chemical formula for phosphorus
trichloride -- PCl3 -- shows that it consists of one atom of
phosphorus and three of chlorine; the formula for phosphorus
oxychloride -- POCl3 -- shows that the chemical is made of one atom
of phosphorus, one of oxygen and three of chlorine.
Figure 2. Chemical identity (from HSG 35: Phosphorus Trichloride)
1.1 Identity
Phosphorus Phosphorus
Trichloride Oxychloride
Chemical formula: PCl3 POCl3
Chemical structure: Cl Cl
| |
Cl -- P Cl P == O
| |
Cl Cl
Common synonyms: Phosphorus chloride; Phosphorus
phosphorus chloride; oxytrichloride
trichlorophosphine
Abbreviations: Tri; "Pickle" Oxy; "Pockle"
CAS registry number:  The "odour threshold" is the concentration at which humans can first
smell the substance in question. Yet many chemicals are dangerous at
concentrations well below the odour threshold. Moreover, sense of
smell varies greatly from person to person and can be affected by nose
and throat infections or head injuries. Finally, the sense of smell
can become "tired" (known as olfactory fatigue or olfactory
adaptation) if a person is exposed to the smell of a chemical for more
than a few minutes. If this happens, a large (and often dangerous)
increase in the amount of chemical present may be necessary before it
can again be smelt by those working with it. IT IS DANGEROUS TO
TRY TO IDENTIFY CHEMICALS BY SMELLING THEM.
1.2.3 Physical properties
The physical properties of each chemical are given in the
International Chemical Safety Card, or Summary of Chemical Safety
Information, contained in most HSGs (see Fig. 5). The terms used are
explained below. Further information can be obtained from the Card
Users' Guide, also produced by the IPCS to assist the user of the
ICSC.
Boiling point -- The temperature at which all the substance changes
from a liquid into a gas, e.g. water changes to steam at 100°C. It is
important to be aware of this temperature because when a chemical
substance is heated above the boiling-point, it will become a gas, and
it is much more difficult to protect against exposure to gases than to
protect against exposure to liquids or solids. Therefore, many
chemicals are more hazardous when heated. As mentioned below (see
Vapour pressure), some of the chemical will turn into a gas even at
temperatures below the boiling point. This can cause a dangerous
build-up of gas.
Melting point -- The temperature at which a substance changes from a
solid to a liquid, e.g. ice changes to water at 0°C.
Figure 5. Physical properties of a chemical (from HSG 9: Isobutanol)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Boiling point (°C) 108 Colourless liquid with
Melting point (°C) -108 characteristic odour;
Flash point (°C) 27 reacts with strong oxidants
Auto-ignition temperature (°C) 430 and alkali metals to form
Relative density (water = 1) 0.8 combustible gas (hydrogen);
Relative vapour density (air = 1) 2.6 attacks many plastics;
Vapour pressure in mbar (20°C) 12 substance may be absorbed
Solubility in water 95 into the body by inhalaltion
(g/litre at 20°C)
Explosive limits (vol. % in air) 1.2-10.9
Relative molecular mass 74.1
Flash point (open or closed cup) -- The flash point is the
temperature at which a substance gives off enough vapour to form an
ignitable mixture with air, i.e. one that burns when exposed to a
flame or spark. Open and closed cup refer to the way in which the
flash point is determined. In general, the open cup method gives a
better approximation of real-life situations. If the flash point
temperature is 20°C or lower, a spark can ignite the vapour, even at
normal room temperatures, and, in such cases, great care must be taken
to reduce the risk of accidental fire. The lower the flash point, the
greater the fire risk. Methods of reducing fire risk are discussed in
Chapter 4.
Auto-ignition temperature -- The lowest temperature at which a
substance will burn without being exposed to a flame or spark. The
closer this value is to room temperature, the greater the risk of
fire.
Relative density or specific gravity (at 20°C) -- The weight
of a specific volume of solid or liquid chemical substance compared to
the weight of the same volume of water. A substance with a specific
density greater than 1 will sink in water; if the specific density is
less than 1, the substance will float on water.
Relative vapour density -- The weight of a specific volume of a
gaseous chemical substance compared to the weight of the same volume
of air. This is also a very important safety consideration. If the
relative vapour density of a pure gas is less than 1, the gas will
rise and collect at ceiling level (if indoors) or disperse into the
atmosphere (if outdoors). On the other hand, if the relative vapour
density of a gas is greater than 1, the gas is heavier than air and
will sink and tend to collect at floor level or in depressions. Such
collection can result in unexpectedly high exposures to the substance,
can lead to a fire, or can represent an explosion hazard. In some
circumstances, gases that have a relative vapour density greater than
1 may completely replace air, including the oxygen that is essential
for life, leading to asphyxiation (suffocation). This often occurs in
tanks, pits or ship holds that are being cleaned or fumigated. This
situation can be avoided by ventilating areas in which gases or
vapours may collect. The vapours of most flammable liquids are
heavier than air. However, in workplace situations, the vapour
concentrations are generally at the level of a few hundred parts per
million (ppm), and the vapours will follow air streams and rise with
heat. In these situations, local exhaust should be as close to the
emissions as possible. Local exhaust near ground level is useful in
case large amounts of liquid leak or are spilled, which could lead to
noticeable vapour formation.
Vapour pressure (kPa) -- The pressure of the vapour produced by a
liquid or solid at room temperature. A small amount of the substance
is transformed from the solid or liquid state to the gaseous state
(vapour) at all temperatures -- evaporation (or sublimation if the
substance changes directly from a solid to a gaseous state without
going through a liquid phase). The higher the vapour pressure, the
more vapour will form above the liquid or solid, i.e. the faster it
tends to evaporate. The vapour pressure of a chemical rises as the
chemical is heated. This vapour or gas could be inhaled by workers.
Solubility in water (g/litre at 20°C) -- The amount (by weight)
of the substance that can be dissolved in 1 litre of water to form a
solution. The solubility in water may give some idea of what maximum
concentrations might occur in water bodies. A high solubility
indicates quick dissipation in surface water, with a potential for
causing acute toxic effects, e.g. to aquatic organisms in the case of
large chemical spills. For some compounds with low solubility,
aquatic organisms will not be so readily threatened by exposure to
potentially acute toxic concentrations.
Flammability (explosive) limits -- The range of concentrations
of a flammable gas or vapour in air at which it may explode in the
presence of a spark or flame. Below the lower limit (Lower Explosive
Limit, or LEL) there is not enough chemical to burn; above the upper
limit (Upper Explosive Limit, or UEL) the mixture is too rich,
i.e. too much chemical is present and not enough oxygen.
Relative molecular mass -- This is the relative weight of the
molecule of a chemical compared to the weight of one atom of hydrogen
(the lightest element).
Octanol/water partition coefficient -- This number indicates
how readily a chemical can dissolve in fats and oils. If the
octanol/water partition coefficient is high, e.g. for DDT it is about
6, then the chemical can accumulate more readily in body fat and can
be stored there for a very long time, as it may be excreted very
slowly. The octanol/water partition coefficient is therefore an
indicator of bioaccumulation. Chemicals that dissolve easily in
fats can usually be absorbed through the skin and may thereafter enter
the bloodstream. An example of a chemical that does not pass easily
through the skin is dimethyl sulfate (DMS), which has an octanol/water
partition coefficient of -4.26. However, DMS can burn the skin badly,
so a low octanol/water partition coefficient should not be seen as
evidence that a particular chemical will not have a negative health
effect if it comes into contact with the skin.
1.2.4 Composition
This section gives details of other substances that may be mixed with
the chemical in question. In some cases, these other chemicals may be
the unwanted result of the manufacturing process, in which case they
are called contaminants or impurities. In other cases, other
chemicals are added deliberately, e.g., to prevent undesirable or
unwanted chemical reactions, such as polymerization or oxidation.
It is important to know what other chemicals may be present in a
substance, as the combined health effects of the chemicals may be far
greater than would be expected by simple addition of the health
effects of the individual chemicals. This increased effect of two or
more chemicals is known as "synergism" and is discussed further in
Chapter 2 of this Manual.
1.3 Analytical methods
This section deals with methods for determining the concentration of
the chemical or its degradation products in the environment and in
foodstuffs (see Fig. 6).
Figure 6. Analytical methods (from HSG 34: Fenvalerate)
1.3 Analytical Methods
For residue and environmental analysis, gas chromatography with
electron-capture detection is used, the minimum detection level being
0.005 mg/kg. For product analysis, gas chromatography with flame
ionization detection can be used.
1.4 Production and uses
The HSGs list the common uses for chemical substances. Many chemicals
are used in a large variety of products and processes (Fig. 7). For
many chemicals it is not possible to list all possible uses. A
careful check should always be made of the chemical that will be used
for a particular type of work or task.
Figure 7. Uses of methylene chloride
(from HSG 6: Methylene Chloride)
1.4 Uses
Methylene chloride is widely used as a solvent and paint remover.
It is also used as a blowing agent for polyurethane, as a propellant
in aerosols such as insecticides, hair sprays, shampoos, and paints,
as a component in fire-extinguishing products, as an insecticidal
fumigant for grains, and as a coolant or refrigerant.
Many HSGs are concerned with the use of chemicals as pesticides and
the associated terminology may need some explaining.
The word pesticide is used as a general term for any substance, or
mixture of substances, intended for controlling the presence of
unwanted plants, animals, insects, etc. As much as possible,
pesticides are designed to control only target species of pests; their
general names then denote the type of pesticide, e.g.:
* acaricides --> spiders and ticks;
* fungicides --> fungi;
* herbicides --> weeds;
* insecticides --> insects;
* larvicides --> larvae of insects or other organisms;
* miticides --> mites;
* molluscicides --> snails; and
* rodenticides --> rats, mice and other rodents.
Pesticides kill or control pests by interfering with one or more of
their essential body systems. Most pesticides belong to a few
chemical groups, each of which has its own effect on certain systems,
e.g. organophosphorus compounds affect nerve function by inhibiting
the action of cholinesterase, an essential chemical for the passage of
impulses along nerves. Small changes in the chemical structure of
compounds in a group can result in some species being affected more by
those compounds than by others in the same group. This is why some
chemicals are more selective than others in their action on certain
pests. Pesticides that tend to kill a variety of organisms, including
both pests and others, are known as broad-spectrum pesticides.
Pesticides may act on target species in several ways: e.g. a pesticide
that kills pests by first passing through the skin or outer layer is a
contact poison; one that kills the pest by first passing into the
stomach is a stomach poison. A pesticide that kills a pest as it
flies through the air is a fumigant. Fumigants usually apply
strictly to pesticides in the form of a gas or vapour, but sometimes
when droplets or an aerosol of a pesticide acts on flying pests (i.e.
essentially a contact poison), the pesticide is said to have a
fumigant action.
A systemic pesticide is one that can be readily absorbed and
transported in plant tissue and, without affecting the plant, can
exert its action on pests feeding on the plant. It is usually applied
to the leaves or the soil around the plant so that it can pass through
the roots. Systemic herbicides are designed to kill the plant
itself if it is a weed.
Pesticides are available as technological products, i.e. the
pesticide chemical plus impurities that may be associated with its
manufacture (as opposed to purer laboratory versions). The pesticide
chemical is also known as the active ingredient. The active
ingredient is usually mixed with other chemicals to facilitate its
use. It is then known as a formulation. Some formulations may need
further dilution.
When a formulation is designed mainly to facilitate use and increase
the effectiveness of the active ingredient, it may be modified to
increase the safety of handling the pesticide. Some examples include:
* soluble powders or granules to be added to water;
* dusts or dustable powders applied as such;
* pellets or paste used as baits;
* tablets to generate smoke, gas or vapour;
* emulsifiable concentrates to be added to water;
* aerosol generators;
* pour-ons for direct application to the skin of animals; and
* shampoos for humans.
2. EXPOSURE, TOXICITY AND RISK - CHAPTER 2
This chapter should be read in conjunction with Chapter 2 (Summary and
Evaluation) of the relevant HSG (see Fig. 8).
Figure 8. Summary and evaluation (from HSG 7: tert-Butanol)
SUMMARY AND EVALUATION
2
Page
2.1 Exposure to tert-butanol 13
2.2 Uptake, metabolism, and excretion 13
2.3 Effects on organisms in the environment 13
2.4 Effects on animals 13
2.5 Effects on human beings 14
2.1 Exposure
Chemicals are found everywhere, not only in the workplace but also in
the general environment -- in the air, water and soil. The heaviest
exposures to some chemicals often occur during industrial or
agricultural activities. But significant exposure can also occur
through contact with naturally occurring ores and the surrounding
soil, from vehicle exhaust emissions, from building and insulating
materials and from various foods. (Food itself may be contaminated or
simply a medium for naturally occurring chemicals.)
Chemicals are found everywhere in the environment; many different
organisms are exposed to them -- whether they are plants exposed, for
instance, to arsenic in the soil around arsenic mines or smelter
wastes, microorganisms such as bacteria and fungi in soil, algae in
water, or fish or birds exposed to pesticides. Different species will
respond in different ways, even to similar doses of the same chemical.
What must be remembered, however, is that for a chemical to exert an
effect, there must first be an exposure. Not even the most toxic
chemicals will cause harm to an organism, including humans, if there
is no exposure. The different ways in which humans can be exposed to
chemicals are described below. Environmental exposures and the
effects of chemicals on the environment are discussed in Chapter 5,
"How Chemicals Can Poison the Environment".
There are four main exposure routes, or ways in which chemicals can
enter the body (see Fig. 9):
* inhalation (breathing in);
* absorption (through the skin or eye);
* ingestion (through the digestive system by swallowing chemicals
or eating contaminated food or drink); and
* transplacental transfer (across the placenta of the pregnant
woman to the fetus).
Most chemicals found in the workplace have the potential to be
dispersed into the air as dust, in droplets (as mist, i.e. an
aerosol) or as gas or vapour, and then inhaled. People who handle
chemicals directly are at risk of absorption via the skin or eyes.
Thus, the most important routes of exposure in the workplace leading
to systemic effects are inhalation and skin absorption (see Fig. 10).
Everyone present in a workplace is also potentially at risk of
exposure to chemicals through ingestion of contaminated food or drink.
Contaminated cigarettes are also a potential source of exposure via
ingestion.
In addition, a fetus may be exposed through transplacental transfer of
chemicals that are in the pregnant woman's bloodstream.
In the case of each exposure route, chemicals can enter the
bloodstream and thereafter be distributed to any or all of the organs
and tissues of the body. In this way, they can attack and harm organs
that are distant from the original point of entry, as well as cause
damage at the point of entry.
Information on methods of preventing exposure to chemicals in the
workplace and pollution of the environment is discussed in Chapters 3
and 4.
The "odour threshold" is the concentration at which humans can first
smell the substance in question. Yet many chemicals are dangerous at
concentrations well below the odour threshold. Moreover, sense of
smell varies greatly from person to person and can be affected by nose
and throat infections or head injuries. Finally, the sense of smell
can become "tired" (known as olfactory fatigue or olfactory
adaptation) if a person is exposed to the smell of a chemical for more
than a few minutes. If this happens, a large (and often dangerous)
increase in the amount of chemical present may be necessary before it
can again be smelt by those working with it. IT IS DANGEROUS TO
TRY TO IDENTIFY CHEMICALS BY SMELLING THEM.
1.2.3 Physical properties
The physical properties of each chemical are given in the
International Chemical Safety Card, or Summary of Chemical Safety
Information, contained in most HSGs (see Fig. 5). The terms used are
explained below. Further information can be obtained from the Card
Users' Guide, also produced by the IPCS to assist the user of the
ICSC.
Boiling point -- The temperature at which all the substance changes
from a liquid into a gas, e.g. water changes to steam at 100°C. It is
important to be aware of this temperature because when a chemical
substance is heated above the boiling-point, it will become a gas, and
it is much more difficult to protect against exposure to gases than to
protect against exposure to liquids or solids. Therefore, many
chemicals are more hazardous when heated. As mentioned below (see
Vapour pressure), some of the chemical will turn into a gas even at
temperatures below the boiling point. This can cause a dangerous
build-up of gas.
Melting point -- The temperature at which a substance changes from a
solid to a liquid, e.g. ice changes to water at 0°C.
Figure 5. Physical properties of a chemical (from HSG 9: Isobutanol)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Boiling point (°C) 108 Colourless liquid with
Melting point (°C) -108 characteristic odour;
Flash point (°C) 27 reacts with strong oxidants
Auto-ignition temperature (°C) 430 and alkali metals to form
Relative density (water = 1) 0.8 combustible gas (hydrogen);
Relative vapour density (air = 1) 2.6 attacks many plastics;
Vapour pressure in mbar (20°C) 12 substance may be absorbed
Solubility in water 95 into the body by inhalaltion
(g/litre at 20°C)
Explosive limits (vol. % in air) 1.2-10.9
Relative molecular mass 74.1
Flash point (open or closed cup) -- The flash point is the
temperature at which a substance gives off enough vapour to form an
ignitable mixture with air, i.e. one that burns when exposed to a
flame or spark. Open and closed cup refer to the way in which the
flash point is determined. In general, the open cup method gives a
better approximation of real-life situations. If the flash point
temperature is 20°C or lower, a spark can ignite the vapour, even at
normal room temperatures, and, in such cases, great care must be taken
to reduce the risk of accidental fire. The lower the flash point, the
greater the fire risk. Methods of reducing fire risk are discussed in
Chapter 4.
Auto-ignition temperature -- The lowest temperature at which a
substance will burn without being exposed to a flame or spark. The
closer this value is to room temperature, the greater the risk of
fire.
Relative density or specific gravity (at 20°C) -- The weight
of a specific volume of solid or liquid chemical substance compared to
the weight of the same volume of water. A substance with a specific
density greater than 1 will sink in water; if the specific density is
less than 1, the substance will float on water.
Relative vapour density -- The weight of a specific volume of a
gaseous chemical substance compared to the weight of the same volume
of air. This is also a very important safety consideration. If the
relative vapour density of a pure gas is less than 1, the gas will
rise and collect at ceiling level (if indoors) or disperse into the
atmosphere (if outdoors). On the other hand, if the relative vapour
density of a gas is greater than 1, the gas is heavier than air and
will sink and tend to collect at floor level or in depressions. Such
collection can result in unexpectedly high exposures to the substance,
can lead to a fire, or can represent an explosion hazard. In some
circumstances, gases that have a relative vapour density greater than
1 may completely replace air, including the oxygen that is essential
for life, leading to asphyxiation (suffocation). This often occurs in
tanks, pits or ship holds that are being cleaned or fumigated. This
situation can be avoided by ventilating areas in which gases or
vapours may collect. The vapours of most flammable liquids are
heavier than air. However, in workplace situations, the vapour
concentrations are generally at the level of a few hundred parts per
million (ppm), and the vapours will follow air streams and rise with
heat. In these situations, local exhaust should be as close to the
emissions as possible. Local exhaust near ground level is useful in
case large amounts of liquid leak or are spilled, which could lead to
noticeable vapour formation.
Vapour pressure (kPa) -- The pressure of the vapour produced by a
liquid or solid at room temperature. A small amount of the substance
is transformed from the solid or liquid state to the gaseous state
(vapour) at all temperatures -- evaporation (or sublimation if the
substance changes directly from a solid to a gaseous state without
going through a liquid phase). The higher the vapour pressure, the
more vapour will form above the liquid or solid, i.e. the faster it
tends to evaporate. The vapour pressure of a chemical rises as the
chemical is heated. This vapour or gas could be inhaled by workers.
Solubility in water (g/litre at 20°C) -- The amount (by weight)
of the substance that can be dissolved in 1 litre of water to form a
solution. The solubility in water may give some idea of what maximum
concentrations might occur in water bodies. A high solubility
indicates quick dissipation in surface water, with a potential for
causing acute toxic effects, e.g. to aquatic organisms in the case of
large chemical spills. For some compounds with low solubility,
aquatic organisms will not be so readily threatened by exposure to
potentially acute toxic concentrations.
Flammability (explosive) limits -- The range of concentrations
of a flammable gas or vapour in air at which it may explode in the
presence of a spark or flame. Below the lower limit (Lower Explosive
Limit, or LEL) there is not enough chemical to burn; above the upper
limit (Upper Explosive Limit, or UEL) the mixture is too rich,
i.e. too much chemical is present and not enough oxygen.
Relative molecular mass -- This is the relative weight of the
molecule of a chemical compared to the weight of one atom of hydrogen
(the lightest element).
Octanol/water partition coefficient -- This number indicates
how readily a chemical can dissolve in fats and oils. If the
octanol/water partition coefficient is high, e.g. for DDT it is about
6, then the chemical can accumulate more readily in body fat and can
be stored there for a very long time, as it may be excreted very
slowly. The octanol/water partition coefficient is therefore an
indicator of bioaccumulation. Chemicals that dissolve easily in
fats can usually be absorbed through the skin and may thereafter enter
the bloodstream. An example of a chemical that does not pass easily
through the skin is dimethyl sulfate (DMS), which has an octanol/water
partition coefficient of -4.26. However, DMS can burn the skin badly,
so a low octanol/water partition coefficient should not be seen as
evidence that a particular chemical will not have a negative health
effect if it comes into contact with the skin.
1.2.4 Composition
This section gives details of other substances that may be mixed with
the chemical in question. In some cases, these other chemicals may be
the unwanted result of the manufacturing process, in which case they
are called contaminants or impurities. In other cases, other
chemicals are added deliberately, e.g., to prevent undesirable or
unwanted chemical reactions, such as polymerization or oxidation.
It is important to know what other chemicals may be present in a
substance, as the combined health effects of the chemicals may be far
greater than would be expected by simple addition of the health
effects of the individual chemicals. This increased effect of two or
more chemicals is known as "synergism" and is discussed further in
Chapter 2 of this Manual.
1.3 Analytical methods
This section deals with methods for determining the concentration of
the chemical or its degradation products in the environment and in
foodstuffs (see Fig. 6).
Figure 6. Analytical methods (from HSG 34: Fenvalerate)
1.3 Analytical Methods
For residue and environmental analysis, gas chromatography with
electron-capture detection is used, the minimum detection level being
0.005 mg/kg. For product analysis, gas chromatography with flame
ionization detection can be used.
1.4 Production and uses
The HSGs list the common uses for chemical substances. Many chemicals
are used in a large variety of products and processes (Fig. 7). For
many chemicals it is not possible to list all possible uses. A
careful check should always be made of the chemical that will be used
for a particular type of work or task.
Figure 7. Uses of methylene chloride
(from HSG 6: Methylene Chloride)
1.4 Uses
Methylene chloride is widely used as a solvent and paint remover.
It is also used as a blowing agent for polyurethane, as a propellant
in aerosols such as insecticides, hair sprays, shampoos, and paints,
as a component in fire-extinguishing products, as an insecticidal
fumigant for grains, and as a coolant or refrigerant.
Many HSGs are concerned with the use of chemicals as pesticides and
the associated terminology may need some explaining.
The word pesticide is used as a general term for any substance, or
mixture of substances, intended for controlling the presence of
unwanted plants, animals, insects, etc. As much as possible,
pesticides are designed to control only target species of pests; their
general names then denote the type of pesticide, e.g.:
* acaricides --> spiders and ticks;
* fungicides --> fungi;
* herbicides --> weeds;
* insecticides --> insects;
* larvicides --> larvae of insects or other organisms;
* miticides --> mites;
* molluscicides --> snails; and
* rodenticides --> rats, mice and other rodents.
Pesticides kill or control pests by interfering with one or more of
their essential body systems. Most pesticides belong to a few
chemical groups, each of which has its own effect on certain systems,
e.g. organophosphorus compounds affect nerve function by inhibiting
the action of cholinesterase, an essential chemical for the passage of
impulses along nerves. Small changes in the chemical structure of
compounds in a group can result in some species being affected more by
those compounds than by others in the same group. This is why some
chemicals are more selective than others in their action on certain
pests. Pesticides that tend to kill a variety of organisms, including
both pests and others, are known as broad-spectrum pesticides.
Pesticides may act on target species in several ways: e.g. a pesticide
that kills pests by first passing through the skin or outer layer is a
contact poison; one that kills the pest by first passing into the
stomach is a stomach poison. A pesticide that kills a pest as it
flies through the air is a fumigant. Fumigants usually apply
strictly to pesticides in the form of a gas or vapour, but sometimes
when droplets or an aerosol of a pesticide acts on flying pests (i.e.
essentially a contact poison), the pesticide is said to have a
fumigant action.
A systemic pesticide is one that can be readily absorbed and
transported in plant tissue and, without affecting the plant, can
exert its action on pests feeding on the plant. It is usually applied
to the leaves or the soil around the plant so that it can pass through
the roots. Systemic herbicides are designed to kill the plant
itself if it is a weed.
Pesticides are available as technological products, i.e. the
pesticide chemical plus impurities that may be associated with its
manufacture (as opposed to purer laboratory versions). The pesticide
chemical is also known as the active ingredient. The active
ingredient is usually mixed with other chemicals to facilitate its
use. It is then known as a formulation. Some formulations may need
further dilution.
When a formulation is designed mainly to facilitate use and increase
the effectiveness of the active ingredient, it may be modified to
increase the safety of handling the pesticide. Some examples include:
* soluble powders or granules to be added to water;
* dusts or dustable powders applied as such;
* pellets or paste used as baits;
* tablets to generate smoke, gas or vapour;
* emulsifiable concentrates to be added to water;
* aerosol generators;
* pour-ons for direct application to the skin of animals; and
* shampoos for humans.
2. EXPOSURE, TOXICITY AND RISK - CHAPTER 2
This chapter should be read in conjunction with Chapter 2 (Summary and
Evaluation) of the relevant HSG (see Fig. 8).
Figure 8. Summary and evaluation (from HSG 7: tert-Butanol)
SUMMARY AND EVALUATION
2
Page
2.1 Exposure to tert-butanol 13
2.2 Uptake, metabolism, and excretion 13
2.3 Effects on organisms in the environment 13
2.4 Effects on animals 13
2.5 Effects on human beings 14
2.1 Exposure
Chemicals are found everywhere, not only in the workplace but also in
the general environment -- in the air, water and soil. The heaviest
exposures to some chemicals often occur during industrial or
agricultural activities. But significant exposure can also occur
through contact with naturally occurring ores and the surrounding
soil, from vehicle exhaust emissions, from building and insulating
materials and from various foods. (Food itself may be contaminated or
simply a medium for naturally occurring chemicals.)
Chemicals are found everywhere in the environment; many different
organisms are exposed to them -- whether they are plants exposed, for
instance, to arsenic in the soil around arsenic mines or smelter
wastes, microorganisms such as bacteria and fungi in soil, algae in
water, or fish or birds exposed to pesticides. Different species will
respond in different ways, even to similar doses of the same chemical.
What must be remembered, however, is that for a chemical to exert an
effect, there must first be an exposure. Not even the most toxic
chemicals will cause harm to an organism, including humans, if there
is no exposure. The different ways in which humans can be exposed to
chemicals are described below. Environmental exposures and the
effects of chemicals on the environment are discussed in Chapter 5,
"How Chemicals Can Poison the Environment".
There are four main exposure routes, or ways in which chemicals can
enter the body (see Fig. 9):
* inhalation (breathing in);
* absorption (through the skin or eye);
* ingestion (through the digestive system by swallowing chemicals
or eating contaminated food or drink); and
* transplacental transfer (across the placenta of the pregnant
woman to the fetus).
Most chemicals found in the workplace have the potential to be
dispersed into the air as dust, in droplets (as mist, i.e. an
aerosol) or as gas or vapour, and then inhaled. People who handle
chemicals directly are at risk of absorption via the skin or eyes.
Thus, the most important routes of exposure in the workplace leading
to systemic effects are inhalation and skin absorption (see Fig. 10).
Everyone present in a workplace is also potentially at risk of
exposure to chemicals through ingestion of contaminated food or drink.
Contaminated cigarettes are also a potential source of exposure via
ingestion.
In addition, a fetus may be exposed through transplacental transfer of
chemicals that are in the pregnant woman's bloodstream.
In the case of each exposure route, chemicals can enter the
bloodstream and thereafter be distributed to any or all of the organs
and tissues of the body. In this way, they can attack and harm organs
that are distant from the original point of entry, as well as cause
damage at the point of entry.
Information on methods of preventing exposure to chemicals in the
workplace and pollution of the environment is discussed in Chapters 3
and 4.
 Figure 10. Occupational exposure
(from HSG 19: Pentachlorophenol)
Occupational exposure to technical PCP occurs mainly through
inhalation and skin contact. Virtually all workers exposed to
airborne concentrations take up PCP via the lungs and the skin. In
addition, workers handling treated timber or maintaining PCP-
contaminated equipment may be exposed to PCP in solution, and
may absorb from one-half (based on urinary-PCP concentrations) to
two-thirds (using serum levels) of their total PCP burden through
the skin.
2.1.1 Inhalation
2.1.1.1 Forms
* Gases and vapours -- Workplace chemicals can be emitted into the
atmosphere in a number of different ways. Evaporation is
probably the most common. Organic solvents, such as toluene,
MEK (methylethylketone) or alcohols, generally evaporate more
rapidly than water, acids or caustics. Evaporation of a
liquid produces vapours. Vapours are formed from substances that
exist as solids or liquids under normal temperature and pressure
conditions. Substances that do not exist as solids or liquids at
normal room temperatures and pressures are called gases. Gases,
as well as vapours, can contaminate the workplace air.
Chemicals with a high vapour pressure will generate more vapour
than chemicals with a low vapour pressure. The amount of vapour
released by a chemical also increases with increasing
temperature.
* Mists and aerosols -- Some industrial processes produce tiny
liquid droplets that are able to float in air. Alternatively,
mists sometimes form when a liquid breaks up, is splashed or is
atomized. Examples include acid mists from electroplating, oil
mists from cutting or grinding and paint spray mists from
painting operations.
* Dusts, fumes and smoke -- Other workplace processes can generate
tiny solid particles that are light enough to float in air; these
are referred to as dusts, fumes or smoke. Dusts are solid
particles, often generated by some mechanical or abrasive
activity. They are usually of such a weight that they settle
slowly to the ground. Fumes are solid particles formed when a
heated substance has evaporated in the air and then condensed
back to a solid form (often after combining with oxygen in the
air to form an oxide). This often occurs during welding
operations. Fume particles are extremely small and may remain
airborne indefinitely. Smoke consists of carbon or soot and is
formed during combustion. Smoke particles can settle or remain
airborne, depending on their size.
2.1.1.2 Entry into the body
Contaminated air in the workplace is inhaled through the mouth and
nose and then into the lungs (see Fig. 11). An average person will
breathe in and out about 12 times per minute. Each inhalation brings
about 500 ml of air into the lungs (corresponding to 6 litres of air
per minute at rest), together with any contaminants that this air
contains. People undertaking hard physical work will breathe harder
and take in more than 6 litres per minute. Over an eight-hour working
day, more than 2800 litres of air will pass through the lungs. In
conditions of hard physical work, up to 10 000 litres may be exchanged
during the course of an eight-hour workshift.
Figure 10. Occupational exposure
(from HSG 19: Pentachlorophenol)
Occupational exposure to technical PCP occurs mainly through
inhalation and skin contact. Virtually all workers exposed to
airborne concentrations take up PCP via the lungs and the skin. In
addition, workers handling treated timber or maintaining PCP-
contaminated equipment may be exposed to PCP in solution, and
may absorb from one-half (based on urinary-PCP concentrations) to
two-thirds (using serum levels) of their total PCP burden through
the skin.
2.1.1 Inhalation
2.1.1.1 Forms
* Gases and vapours -- Workplace chemicals can be emitted into the
atmosphere in a number of different ways. Evaporation is
probably the most common. Organic solvents, such as toluene,
MEK (methylethylketone) or alcohols, generally evaporate more
rapidly than water, acids or caustics. Evaporation of a
liquid produces vapours. Vapours are formed from substances that
exist as solids or liquids under normal temperature and pressure
conditions. Substances that do not exist as solids or liquids at
normal room temperatures and pressures are called gases. Gases,
as well as vapours, can contaminate the workplace air.
Chemicals with a high vapour pressure will generate more vapour
than chemicals with a low vapour pressure. The amount of vapour
released by a chemical also increases with increasing
temperature.
* Mists and aerosols -- Some industrial processes produce tiny
liquid droplets that are able to float in air. Alternatively,
mists sometimes form when a liquid breaks up, is splashed or is
atomized. Examples include acid mists from electroplating, oil
mists from cutting or grinding and paint spray mists from
painting operations.
* Dusts, fumes and smoke -- Other workplace processes can generate
tiny solid particles that are light enough to float in air; these
are referred to as dusts, fumes or smoke. Dusts are solid
particles, often generated by some mechanical or abrasive
activity. They are usually of such a weight that they settle
slowly to the ground. Fumes are solid particles formed when a
heated substance has evaporated in the air and then condensed
back to a solid form (often after combining with oxygen in the
air to form an oxide). This often occurs during welding
operations. Fume particles are extremely small and may remain
airborne indefinitely. Smoke consists of carbon or soot and is
formed during combustion. Smoke particles can settle or remain
airborne, depending on their size.
2.1.1.2 Entry into the body
Contaminated air in the workplace is inhaled through the mouth and
nose and then into the lungs (see Fig. 11). An average person will
breathe in and out about 12 times per minute. Each inhalation brings
about 500 ml of air into the lungs (corresponding to 6 litres of air
per minute at rest), together with any contaminants that this air
contains. People undertaking hard physical work will breathe harder
and take in more than 6 litres per minute. Over an eight-hour working
day, more than 2800 litres of air will pass through the lungs. In
conditions of hard physical work, up to 10 000 litres may be exchanged
during the course of an eight-hour workshift.
 Air breathed in through the nose is filtered by the nasal hairs;
large, solid particles in the atmosphere are therefore prevented from
passing any further into the body. Small bones and cartilage inside
the nose help the inhaled air to circulate. Large contaminating
particles may then be deposited in the nose, trapped by the moisture
of the mucous lining (see below).
Air inhaled through the nose and mouth reaches the back of the throat
and enters the pharynx. The pharynx, which is the entrance to the
airways, divides into two tubes, one called the oesophagus, which
carries food to the stomach, the other the trachea, which carries
air to the lungs. At the top of the trachea sits the larynx, where
the vocal cords are situated. Contaminated air passes into the
trachea, which itself divides into two large tubes, each called a
bronchus. Each bronchus enters a lung. Once inside the lung, each
bronchus starts to divide like the branches of a tree -- the branches
getting thinner and thinner as they spread. Eventually, the tiniest
tubes, which are called bronchioles, end in thin-walled air sacs,
each of which is called an alveolus. Collectively, they are called
alveoli, and there are hundreds of thousands of these in each lung.
The walls of the alveoli are very thin and are richly supplied with
tiny blood vessels (capillaries).
The most important function of the lung is to bring oxygen into the
body. Oxygen in inhaled breath crosses the alveolar walls and enters
the blood within the capillaries. Once oxygen has become absorbed
into the bloodstream, it is distributed throughout the body. Chemical
vapours, gases and mists that reach the alveoli in the lungs can also
pass into the blood and therefore be distributed around the body.
The airways contain a sticky, thick fluid called mucus. Tiny hairs,
known as cilia, on the inside of the tubes constantly carry the mucus
upwards towards the back of the throat from which it is either
expelled (spat out) through the mouth or swallowed and passed into the
stomach. Chemicals that are carried in air that has been inhaled can
in this way enter the stomach.
Solid particles that have reached the air sacs but which cannot pass
through the thin wall of the sacs may become lodged there. Some may
dissolve, others may be attacked and destroyed by scavenger cells
(macrophages) of the body's defence system. Some particles that
remain in the air sacs, if only in small quantities, do no apparent
harm. Other types of dust may damage the surrounding alveolar walls.
Such damage may be permanent and may result in the formation of scars,
which eventually interfere with the lung's ability to expand and pass
oxygen into the bloodstream.
Some acids, caustics or organic chemicals, when inhaled in large
amounts, can cause serious "burns" to the mouth, nose, trachea and
lungs.
Chemicals in the form of dust may settle on cigarettes or be
transferred to them from the hands. When a contaminated cigarette is
smoked, toxic chemical fumes will be inhaled. This may also occur if
cigarettes are smoked in areas where chemical vapours or gases are
present. For these reasons, cigarettes should be stored outside the
work area: no smoking should be allowed in a workplace where hazardous
chemicals are used.
2.1.2 Absorption through the skin
2.1.2.1 Forms
Chemicals that pass through the skin are nearly always in liquid form.
Dusts, gases or vapours do not generally pass through the skin unless
they are first dissolved in moisture on the skin's surface. Chemicals
that can dissolve easily in lipids (fats) will pass more readily
through the skin. The octanol/water partition coefficient (see
Chapter 1 of this Manual) can be used as a measure of how
lipid-soluble a chemical is.
There are some chemicals, however, that can pass through the skin via
other mechanisms. Some highly toxic compounds such as sarin and
parathion, for instance, penetrate the skin without causing overt
damage. Chemicals that cause local irritation or that are corrosive
may be absorbed in greater quantity owing to increased blood flow to
the skin or as a result of destruction of the outer skin barrier.
2.1.2.2 Entry into the body
The skin consists essentially of two layers: a thin, outermost layer
called the epidermis and a much thicker layer called the dermis (see
Fig. 12). The epidermis consists of several layers of flat, rather
tightly packed cells, which form a barrier against infections, water
and some chemicals. This barrier is the external part of the
epidermis. It is called the keratin layer and is largely
responsible for preventing water from escaping from the body. It can
also resist entry of water and weak acids but is much less resistant
to organic and some inorganic chemicals. The keratin layer contains
fat and fat-like substances, which readily absorb chemicals that are
solvents for fat, oil and grease.
Air breathed in through the nose is filtered by the nasal hairs;
large, solid particles in the atmosphere are therefore prevented from
passing any further into the body. Small bones and cartilage inside
the nose help the inhaled air to circulate. Large contaminating
particles may then be deposited in the nose, trapped by the moisture
of the mucous lining (see below).
Air inhaled through the nose and mouth reaches the back of the throat
and enters the pharynx. The pharynx, which is the entrance to the
airways, divides into two tubes, one called the oesophagus, which
carries food to the stomach, the other the trachea, which carries
air to the lungs. At the top of the trachea sits the larynx, where
the vocal cords are situated. Contaminated air passes into the
trachea, which itself divides into two large tubes, each called a
bronchus. Each bronchus enters a lung. Once inside the lung, each
bronchus starts to divide like the branches of a tree -- the branches
getting thinner and thinner as they spread. Eventually, the tiniest
tubes, which are called bronchioles, end in thin-walled air sacs,
each of which is called an alveolus. Collectively, they are called
alveoli, and there are hundreds of thousands of these in each lung.
The walls of the alveoli are very thin and are richly supplied with
tiny blood vessels (capillaries).
The most important function of the lung is to bring oxygen into the
body. Oxygen in inhaled breath crosses the alveolar walls and enters
the blood within the capillaries. Once oxygen has become absorbed
into the bloodstream, it is distributed throughout the body. Chemical
vapours, gases and mists that reach the alveoli in the lungs can also
pass into the blood and therefore be distributed around the body.
The airways contain a sticky, thick fluid called mucus. Tiny hairs,
known as cilia, on the inside of the tubes constantly carry the mucus
upwards towards the back of the throat from which it is either
expelled (spat out) through the mouth or swallowed and passed into the
stomach. Chemicals that are carried in air that has been inhaled can
in this way enter the stomach.
Solid particles that have reached the air sacs but which cannot pass
through the thin wall of the sacs may become lodged there. Some may
dissolve, others may be attacked and destroyed by scavenger cells
(macrophages) of the body's defence system. Some particles that
remain in the air sacs, if only in small quantities, do no apparent
harm. Other types of dust may damage the surrounding alveolar walls.
Such damage may be permanent and may result in the formation of scars,
which eventually interfere with the lung's ability to expand and pass
oxygen into the bloodstream.
Some acids, caustics or organic chemicals, when inhaled in large
amounts, can cause serious "burns" to the mouth, nose, trachea and
lungs.
Chemicals in the form of dust may settle on cigarettes or be
transferred to them from the hands. When a contaminated cigarette is
smoked, toxic chemical fumes will be inhaled. This may also occur if
cigarettes are smoked in areas where chemical vapours or gases are
present. For these reasons, cigarettes should be stored outside the
work area: no smoking should be allowed in a workplace where hazardous
chemicals are used.
2.1.2 Absorption through the skin
2.1.2.1 Forms
Chemicals that pass through the skin are nearly always in liquid form.
Dusts, gases or vapours do not generally pass through the skin unless
they are first dissolved in moisture on the skin's surface. Chemicals
that can dissolve easily in lipids (fats) will pass more readily
through the skin. The octanol/water partition coefficient (see
Chapter 1 of this Manual) can be used as a measure of how
lipid-soluble a chemical is.
There are some chemicals, however, that can pass through the skin via
other mechanisms. Some highly toxic compounds such as sarin and
parathion, for instance, penetrate the skin without causing overt
damage. Chemicals that cause local irritation or that are corrosive
may be absorbed in greater quantity owing to increased blood flow to
the skin or as a result of destruction of the outer skin barrier.
2.1.2.2 Entry into the body
The skin consists essentially of two layers: a thin, outermost layer
called the epidermis and a much thicker layer called the dermis (see
Fig. 12). The epidermis consists of several layers of flat, rather
tightly packed cells, which form a barrier against infections, water
and some chemicals. This barrier is the external part of the
epidermis. It is called the keratin layer and is largely
responsible for preventing water from escaping from the body. It can
also resist entry of water and weak acids but is much less resistant
to organic and some inorganic chemicals. The keratin layer contains
fat and fat-like substances, which readily absorb chemicals that are
solvents for fat, oil and grease.
 Organic and caustic (alkaline) chemicals can soften the keratin cells
and pass through this layer to the dermis, then enter the bloodstream.
Chemicals can also enter the body through cuts, punctures or scrapes
of the skin, as these constitute breaks in the protective layer.
Contact with some chemicals such as detergents or organic solvents can
cause dryness or cracking of the skin. There can also be hives, or
nettle-rash, ulcerations or skin flaking. All these conditions weaken
the protective layer of the skin, and chemicals are more likely to
enter the bloodstream.
Once in the bloodstream, the chemicals may be transported to any site
or organ of the body, where they may exert their effects.
Some chemicals are so corrosive that they may burn holes in the skin,
through which infection or entry of other chemicals can occur.
2.1.3 Absorption through the eye
2.1.3.1 Forms
Any chemical, in the form of a liquid, dust, vapour, gas, aerosol or
mist can enter the eye.
2.1.3.2 Entry into the body
Eye splashes or eye contamination due to exposure to workplace
chemicals is common. Small amounts of chemicals can enter the eye by
dissolving in the liquid surrounding the eye. The eyes are richly
supplied with blood vessels, and many chemicals can penetrate the
outer tissues and pass into the veins. The eye may be damaged during
this process, depending on the corrosive nature of the chemical and
its ability to penetrate the outer tissues. For example, the organic
solvent toluene can pass through the outer layers of the eye and
probably enter the blood. It can cause keratitis, which is an
inflammation of the outer layer of the eye (see Fig. 13). The
majority of substances that become dissolved in tears are passed to
the nose and eventually swallowed (see Fig. 14). Only in very rare
cases does absorption of chemicals through the eye cause acute
systemic effects.
2.1.4 Ingestion
2.1.4.1 Forms
All forms of chemicals, whether they exist as liquids, solids, gases,
vapours, mists, dusts, smoke or fumes, can -- directly or indirectly
-- enter the digestive system.
Organic and caustic (alkaline) chemicals can soften the keratin cells
and pass through this layer to the dermis, then enter the bloodstream.
Chemicals can also enter the body through cuts, punctures or scrapes
of the skin, as these constitute breaks in the protective layer.
Contact with some chemicals such as detergents or organic solvents can
cause dryness or cracking of the skin. There can also be hives, or
nettle-rash, ulcerations or skin flaking. All these conditions weaken
the protective layer of the skin, and chemicals are more likely to
enter the bloodstream.
Once in the bloodstream, the chemicals may be transported to any site
or organ of the body, where they may exert their effects.
Some chemicals are so corrosive that they may burn holes in the skin,
through which infection or entry of other chemicals can occur.
2.1.3 Absorption through the eye
2.1.3.1 Forms
Any chemical, in the form of a liquid, dust, vapour, gas, aerosol or
mist can enter the eye.
2.1.3.2 Entry into the body
Eye splashes or eye contamination due to exposure to workplace
chemicals is common. Small amounts of chemicals can enter the eye by
dissolving in the liquid surrounding the eye. The eyes are richly
supplied with blood vessels, and many chemicals can penetrate the
outer tissues and pass into the veins. The eye may be damaged during
this process, depending on the corrosive nature of the chemical and
its ability to penetrate the outer tissues. For example, the organic
solvent toluene can pass through the outer layers of the eye and
probably enter the blood. It can cause keratitis, which is an
inflammation of the outer layer of the eye (see Fig. 13). The
majority of substances that become dissolved in tears are passed to
the nose and eventually swallowed (see Fig. 14). Only in very rare
cases does absorption of chemicals through the eye cause acute
systemic effects.
2.1.4 Ingestion
2.1.4.1 Forms
All forms of chemicals, whether they exist as liquids, solids, gases,
vapours, mists, dusts, smoke or fumes, can -- directly or indirectly
-- enter the digestive system.

 2.1.4.2 Entry into the body
Chemicals can enter the stomach following ingestion of contaminated
food or mouth contact with contaminated cigarettes (see Fig. 15).
Food and drink can become contaminated when they are produced and also
at the time of consumption by contact with unwashed hands, gloves or
clothing, or if they are left exposed in the workplace. Nail-biting
is another potential source of ingested chemicals. Some of the
chemicals that are inhaled will be trapped in the mucus of the lungs,
much of which will eventually be swallowed, thereby contributing to
the total amount of the ingested chemicals. In addition, chemicals in
liquid form may enter the body by accidental ingestion. For example,
if a liquid is transferred to a bottle and not labelled adequately, a
worker may mistake it for water or another beverage.
2.1.4.2 Entry into the body
Chemicals can enter the stomach following ingestion of contaminated
food or mouth contact with contaminated cigarettes (see Fig. 15).
Food and drink can become contaminated when they are produced and also
at the time of consumption by contact with unwashed hands, gloves or
clothing, or if they are left exposed in the workplace. Nail-biting
is another potential source of ingested chemicals. Some of the
chemicals that are inhaled will be trapped in the mucus of the lungs,
much of which will eventually be swallowed, thereby contributing to
the total amount of the ingested chemicals. In addition, chemicals in
liquid form may enter the body by accidental ingestion. For example,
if a liquid is transferred to a bottle and not labelled adequately, a
worker may mistake it for water or another beverage.
 Once inside the mouth, chemicals pass down the oesophagus and into the
stomach. Food is digested with a strong acid produced by the stomach.
A few chemicals, such as alcohol, may pass across the stomach wall and
enter the veins and bloodstream there. But most chemicals move from
the stomach into the small intestine. The inside of the small
intestine has hundreds of tiny finger-like projections called villi
that have very thin walls and are filled with tiny blood vessels.
This allows the digested food to pass from the small intestine across
the walls of the villi and into the bloodstream.
Some chemicals that contaminate food or drink can also pass across the
thin walls of the villi and become absorbed into the bloodstream.
Other chemicals that are not soluble, or the basic units (molecules)
of which are too big to pass across the villi walls, will stay in the
gut and pass out through the faeces without being absorbed into the
bloodstream to a great extent. The absorption rate of a chemical
indicates the percentage of the amount swallowed that becomes absorbed
into the bloodstream. Most chemicals are only partly absorbed.
Some acids, caustics and organic chemicals may cause severe "burn"
damage to the digestive system if ingested in high concentrations.
2.2 Processing of chemicals in the body
After a chemical enters the body, it undergoes one or more of three
processes. The chemical may be metabolized by the body, stored or
accumulated in the body and/or excreted.
2.2.1 Metabolism
Metabolism is the process by which the body renders a 'foreign'
chemical more easily excretable and less toxic. The body is full of
special proteins called enzymes which initiate particular chemical
reactions. These reactions are necessary to, among other things,
convert the food we eat into a form that can be used by the tissues
and, combined with oxygen carried in the blood, to create heat and
energy. Some enzymes render foreign chemicals less toxic. They do
this by breaking down a chemical compound into simpler chemicals or by
converting a foreign chemical into a form that the body can excrete
more easily. Once a foreign chemical has been metabolized, it becomes
a different chemical, known as a metabolite. The process of
metabolism may involve many steps, and the result is many different
metabolites.
Metabolism may occur anywhere or everywhere in the body, or in just
one organ or type of tissue. Generally, this process renders a
chemical less toxic. However, some metabolites are as toxic as or
even more toxic than the original chemical. Such metabolites are
often called reactive metabolites. For most chemicals, the liver is
the main site of metabolism, but other organs such as the kidneys and
testes are also capable of metabolizing chemicals, the products of
which may be toxic.
2.2.2 Excretion
Excretion is the process by which unwanted chemicals are removed from
the body. Both foreign chemicals and those produced naturally by the
body are excreted. Most excretion occurs through the kidneys. Some
chemicals are excreted via the bile that passes from the liver into
the intestines, after which the chemicals pass out of the body in the
faeces. Other chemicals are excreted as a gas that is exhaled from
the lungs; small amounts may also be excreted in sweat.
In the kidneys, blood (which may be carrying waste or foreign
chemicals) is filtered through a set of small, twisting blood vessels
called the glomeruli. The fluid that is filtered off then passes
through tiny tubes called tubules. Here, water is reabsorbed and the
level of various salts adjusted. The combination of a glomerulus and
a tubule is called a nephron. There are hundreds of thousands of
nephrons in each kidney. The fluid that each nephron produces flows
through more tubes (the collecting ducts and the ureters) into the
bladder, from which urine is expelled from the body (see Fig. 16).
For some chemicals, it is possible to calculate the amount absorbed by
testing a urine sample. The concentration of the original chemical or
the concentration of a metabolite in the urine can also be determined.
Urine tests are often useful for determining whether absorption of a
chemical has occurred. For some chemicals, such as alcohol, exhaled
air can be used as an indication of absorption. Testing of urine,
blood or other body fluids to determine absorption is discussed
further in Chapter 3 of this Manual.
Once inside the mouth, chemicals pass down the oesophagus and into the
stomach. Food is digested with a strong acid produced by the stomach.
A few chemicals, such as alcohol, may pass across the stomach wall and
enter the veins and bloodstream there. But most chemicals move from
the stomach into the small intestine. The inside of the small
intestine has hundreds of tiny finger-like projections called villi
that have very thin walls and are filled with tiny blood vessels.
This allows the digested food to pass from the small intestine across
the walls of the villi and into the bloodstream.
Some chemicals that contaminate food or drink can also pass across the
thin walls of the villi and become absorbed into the bloodstream.
Other chemicals that are not soluble, or the basic units (molecules)
of which are too big to pass across the villi walls, will stay in the
gut and pass out through the faeces without being absorbed into the
bloodstream to a great extent. The absorption rate of a chemical
indicates the percentage of the amount swallowed that becomes absorbed
into the bloodstream. Most chemicals are only partly absorbed.
Some acids, caustics and organic chemicals may cause severe "burn"
damage to the digestive system if ingested in high concentrations.
2.2 Processing of chemicals in the body
After a chemical enters the body, it undergoes one or more of three
processes. The chemical may be metabolized by the body, stored or
accumulated in the body and/or excreted.
2.2.1 Metabolism
Metabolism is the process by which the body renders a 'foreign'
chemical more easily excretable and less toxic. The body is full of
special proteins called enzymes which initiate particular chemical
reactions. These reactions are necessary to, among other things,
convert the food we eat into a form that can be used by the tissues
and, combined with oxygen carried in the blood, to create heat and
energy. Some enzymes render foreign chemicals less toxic. They do
this by breaking down a chemical compound into simpler chemicals or by
converting a foreign chemical into a form that the body can excrete
more easily. Once a foreign chemical has been metabolized, it becomes
a different chemical, known as a metabolite. The process of
metabolism may involve many steps, and the result is many different
metabolites.
Metabolism may occur anywhere or everywhere in the body, or in just
one organ or type of tissue. Generally, this process renders a
chemical less toxic. However, some metabolites are as toxic as or
even more toxic than the original chemical. Such metabolites are
often called reactive metabolites. For most chemicals, the liver is
the main site of metabolism, but other organs such as the kidneys and
testes are also capable of metabolizing chemicals, the products of
which may be toxic.
2.2.2 Excretion
Excretion is the process by which unwanted chemicals are removed from
the body. Both foreign chemicals and those produced naturally by the
body are excreted. Most excretion occurs through the kidneys. Some
chemicals are excreted via the bile that passes from the liver into
the intestines, after which the chemicals pass out of the body in the
faeces. Other chemicals are excreted as a gas that is exhaled from
the lungs; small amounts may also be excreted in sweat.
In the kidneys, blood (which may be carrying waste or foreign
chemicals) is filtered through a set of small, twisting blood vessels
called the glomeruli. The fluid that is filtered off then passes
through tiny tubes called tubules. Here, water is reabsorbed and the
level of various salts adjusted. The combination of a glomerulus and
a tubule is called a nephron. There are hundreds of thousands of
nephrons in each kidney. The fluid that each nephron produces flows
through more tubes (the collecting ducts and the ureters) into the
bladder, from which urine is expelled from the body (see Fig. 16).
For some chemicals, it is possible to calculate the amount absorbed by
testing a urine sample. The concentration of the original chemical or
the concentration of a metabolite in the urine can also be determined.
Urine tests are often useful for determining whether absorption of a
chemical has occurred. For some chemicals, such as alcohol, exhaled
air can be used as an indication of absorption. Testing of urine,
blood or other body fluids to determine absorption is discussed
further in Chapter 3 of this Manual.
 2.2.3 Storage or accumulation
Chemicals that undergo a slow metabolism or excretion are often stored
in various tissues of the body. Sustained exposure may thus increase
the amount of the chemical in tissues. Chemicals that are stored in
this way are said to accumulate.
The time taken for half of the total amount of chemical in the body to
be metabolized or excreted is called the half-time (or half-life)
of the chemical. If a chemical has a short half-time, it is
metabolized or excreted quickly by the body. Some chemicals, such as
cadmium, have a very long half-time of 15"20 years. The half-time of
a chemical varies from person to person, and this will influence how
sensitive an individual is to a chemical. An individual with a long
half-time will accumulate more of the chemical in his or her body.
The average half-time is sometimes given in information about
chemicals that are stored or that accumulate in the body.
2.3 Toxic effects of chemicals
The toxic effects, or toxicity, of a chemical can be defined as the
potential of that chemical to poison the body -- of the person
exposed, of an unborn baby (if the exposed person is pregnant), of a
future offspring of the exposed person or even of an offspring of the
exposed person's offspring.
The potential that a chemical substance has for causing negative
health effects depends principally on the toxicity of the chemical and
the degree of exposure. The toxicity is a property of the chemical
itself, while the exposure depends on how the chemical is used, for
example whether it is heated, sprayed or otherwise released into the
workplace environment. Another important concept in evaluating harm,
however, is the individual susceptibility of exposed persons. There
can be marked differences in reactions between workers who are exposed
to the same chemical, at the same worksite and in similar
concentrations. This may be due, for example, to gender (women, with
a greater relative proportion of body fat, may be more susceptible
than men to the harmful effects of solvents, for instance), age
(children and the elderly are generally more susceptible to chemical
hazards) or race (certain races may be genetically more vulnerable to
certain chemical exposures). Nutritional status may also have a
considerable effect on the action of some compounds.
Several concepts have been developed to help classify the toxic
effects of chemicals. These include:
* Acute effect -- The term acute means "of rapid onset and short
duration" and, with reference to chemicals, usually means a short
exposure with an immediate effect (see Fig. 17). While an acute
exposure can result in an acute effect, it can also result in a
chronic disease, e.g. permanent brain damage can result from
acute exposure to trialkyl tin compounds or from severe carbon
monoxide poisoning.
2.2.3 Storage or accumulation
Chemicals that undergo a slow metabolism or excretion are often stored
in various tissues of the body. Sustained exposure may thus increase
the amount of the chemical in tissues. Chemicals that are stored in
this way are said to accumulate.
The time taken for half of the total amount of chemical in the body to
be metabolized or excreted is called the half-time (or half-life)
of the chemical. If a chemical has a short half-time, it is
metabolized or excreted quickly by the body. Some chemicals, such as
cadmium, have a very long half-time of 15"20 years. The half-time of
a chemical varies from person to person, and this will influence how
sensitive an individual is to a chemical. An individual with a long
half-time will accumulate more of the chemical in his or her body.
The average half-time is sometimes given in information about
chemicals that are stored or that accumulate in the body.
2.3 Toxic effects of chemicals
The toxic effects, or toxicity, of a chemical can be defined as the
potential of that chemical to poison the body -- of the person
exposed, of an unborn baby (if the exposed person is pregnant), of a
future offspring of the exposed person or even of an offspring of the
exposed person's offspring.
The potential that a chemical substance has for causing negative
health effects depends principally on the toxicity of the chemical and
the degree of exposure. The toxicity is a property of the chemical
itself, while the exposure depends on how the chemical is used, for
example whether it is heated, sprayed or otherwise released into the
workplace environment. Another important concept in evaluating harm,
however, is the individual susceptibility of exposed persons. There
can be marked differences in reactions between workers who are exposed
to the same chemical, at the same worksite and in similar
concentrations. This may be due, for example, to gender (women, with
a greater relative proportion of body fat, may be more susceptible
than men to the harmful effects of solvents, for instance), age
(children and the elderly are generally more susceptible to chemical
hazards) or race (certain races may be genetically more vulnerable to
certain chemical exposures). Nutritional status may also have a
considerable effect on the action of some compounds.
Several concepts have been developed to help classify the toxic
effects of chemicals. These include:
* Acute effect -- The term acute means "of rapid onset and short
duration" and, with reference to chemicals, usually means a short
exposure with an immediate effect (see Fig. 17). While an acute
exposure can result in an acute effect, it can also result in a
chronic disease, e.g. permanent brain damage can result from
acute exposure to trialkyl tin compounds or from severe carbon
monoxide poisoning.
 * Chronic effect -- The term chronic means "of slow onset and long
duration" and usually refers to repeated exposure with a long
delay between the first exposure and the appearance of adverse
health effects (see Fig. 18). The delay between the time of
exposure and the onset of the health effect is called the
latency period.
* Acute and chronic effects -- A substance may have both an acute
and a chronic effect. For example, a single exposure to high
levels of carbon disulfide can result in unconsciousness (acute
effect), but repeated daily exposure for years at much lower
concentrations may result in damage to the central and peripheral
nervous system, as well as to the heart, i.e. at concentrations
that if experienced as a single exposure would not lead to
adverse effects (chronic effects).
* Chronic effect -- The term chronic means "of slow onset and long
duration" and usually refers to repeated exposure with a long
delay between the first exposure and the appearance of adverse
health effects (see Fig. 18). The delay between the time of
exposure and the onset of the health effect is called the
latency period.
* Acute and chronic effects -- A substance may have both an acute
and a chronic effect. For example, a single exposure to high
levels of carbon disulfide can result in unconsciousness (acute
effect), but repeated daily exposure for years at much lower
concentrations may result in damage to the central and peripheral
nervous system, as well as to the heart, i.e. at concentrations
that if experienced as a single exposure would not lead to
adverse effects (chronic effects).
 * Reversible (temporary) effect -- An effect that disappears if
exposure to that chemical ceases. Contact dermatitis, headaches
and nausea from exposure to solvents are examples of reversible
effects.
* Irreversible (permanent) effect -- An effect that will have a
lasting, damaging effect on the body, even if exposure to the
chemical causing that effect ceases. Cancer caused by exposure
to a chemical is an example of an irreversible effect.
* Local effect -- The harmful effect of a chemical at the point of
contact or entry to the body, e.g. burns to the skin.
* Systemic effect -- The effect of a chemical on the organs and
fluids of the body after absorption and transport from the point
of entry. Anaemia (a deficiency of haemoglobin due to a lack
of red blood cells) is a typical systemic effect. It can be
caused by a number of chemicals, including lead, beryllium,
benzene, cadmium and mercury compounds.
* Synergism -- The combined effect of exposure to more than one
chemical at one time, or to a chemical in combination with other
hazards such as heat, noise or radiation. The resultant health
effects of such exposure may be greater than the sum of the
individual effects of each hazard by itself. A mixture of sulfur
dioxide and sulfur trioxide, for example, has been shown to have
more severe effects on the functioning of respiratory passages
than the mathematical sum of their individual effects on the
respiratory system would suggest.
The toxic effects (or the toxicity) of a chemical can be classified in
the following ways:
* Corrosive -- A chemical that destroys or damages (burns) living
tissue on contact (see Fig. 19). For example, concentrated
solutions of strong acids such as sulfuric acid, or alkalis such
as caustic soda, cause chemical burns. A splash of a corrosive
liquid in the eye can result in permanent damage to eyesight.
* Irritant -- A chemical that will produce local irritation or
inflammation of the skin, eyes, nose or lung tissue.
* Sensitizer -- A chemical that causes an allergic reaction. A
person who is sensitized to a chemical will experience a
heightened reaction to it, whereas for the majority of
individuals the chemical will not be harmful at the same,
sometimes very low, dose. For a sensitized person, any
subsequent exposure -- whether through skin contact or by
inhalation -- will represent a health risk.
* Reversible (temporary) effect -- An effect that disappears if
exposure to that chemical ceases. Contact dermatitis, headaches
and nausea from exposure to solvents are examples of reversible
effects.
* Irreversible (permanent) effect -- An effect that will have a
lasting, damaging effect on the body, even if exposure to the
chemical causing that effect ceases. Cancer caused by exposure
to a chemical is an example of an irreversible effect.
* Local effect -- The harmful effect of a chemical at the point of
contact or entry to the body, e.g. burns to the skin.
* Systemic effect -- The effect of a chemical on the organs and
fluids of the body after absorption and transport from the point
of entry. Anaemia (a deficiency of haemoglobin due to a lack
of red blood cells) is a typical systemic effect. It can be
caused by a number of chemicals, including lead, beryllium,
benzene, cadmium and mercury compounds.
* Synergism -- The combined effect of exposure to more than one
chemical at one time, or to a chemical in combination with other
hazards such as heat, noise or radiation. The resultant health
effects of such exposure may be greater than the sum of the
individual effects of each hazard by itself. A mixture of sulfur
dioxide and sulfur trioxide, for example, has been shown to have
more severe effects on the functioning of respiratory passages
than the mathematical sum of their individual effects on the
respiratory system would suggest.
The toxic effects (or the toxicity) of a chemical can be classified in
the following ways:
* Corrosive -- A chemical that destroys or damages (burns) living
tissue on contact (see Fig. 19). For example, concentrated
solutions of strong acids such as sulfuric acid, or alkalis such
as caustic soda, cause chemical burns. A splash of a corrosive
liquid in the eye can result in permanent damage to eyesight.
* Irritant -- A chemical that will produce local irritation or
inflammation of the skin, eyes, nose or lung tissue.
* Sensitizer -- A chemical that causes an allergic reaction. A
person who is sensitized to a chemical will experience a
heightened reaction to it, whereas for the majority of
individuals the chemical will not be harmful at the same,
sometimes very low, dose. For a sensitized person, any
subsequent exposure -- whether through skin contact or by
inhalation -- will represent a health risk.
 * Asphyxiant -- A chemical that interferes with the oxygenation of
the body tissues. There are two types of asphyxiation: simple
asphyxiation, whereby oxygen in the air is replaced by a gas to
a level at which it cannot sustain life; and chemical
asphyxiation, whereby a direct chemical action interferes with
the body's ability to transport and use oxygen. Examples of
chemical asphyxiants include carbon monoxide and cyanides.
* Carcinogen -- A chemical that causes cancer. Cancer is a group of
diseases characterized by the manner in which abnormal cells in
the body multiply and spread out of control. The key feature of
cancer is the "malignant" or deadly way that its cells crowd out
other cells and interfere with the normal functioning of the body
(see Fig. 20).
* Mutagen -- A chemical that can cause permanent damage to the
deoxyribonucleic acid (DNA) in a cell (see Fig. 21). DNA is
a molecule that carries the genetic information controlling the
growth and function of cells. DNA damage in the human egg or
sperm may lead to reduced fertility, spontaneous abortion
(miscarriage), birth defects and heritable diseases. Thus, any
chemical known to be a mutagen must be treated with caution.
Figure 20. Carcinogenic effect (from HSG 19: Pentachlorophenol)
The results of animal studies, designed to assess the carcinogenicity
of PCP and reported to date, have been negative. Carcinogenicity
bioassays with one other chlorophenol (2,4,6-T3CP) and a mixture of
two H6CDD congeners found in PCP have been positive. Hence, the
carcinogenic effects of long-term exposure of animals to technical PCP
are not clear.
Figure 21. Mutagenic effect (from HSG 32: d-Phenothrin)
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
* Teratogen -- A chemical that, if present in the bloodstream of a
pregnant woman, crosses the placenta, affecting the developing
fetus and resulting in structural or functional congenital
abnormalities, or cancer, in the child. These effects may not be
observable until the child becomes an adult. One of the
better-known examples of a teratogen is Thalidomide, which, in
the 1960s, caused many cases of phocomelia (a reduction of the
limbs to the extent that the hands and feet are attached directly
to the body) in babies born to women who took the drug during the
early stages of pregnancy. Several industrial chemicals have been
shown to be experimental teratogens; some, such as ethylene
dichloride and vinyl chloride, have been shown to be teratogenic
in humans.
* Fetotoxicant -- A chemical that adversely affects the developing
fetus, resulting in low birth weight, symptoms of poisoning at
birth or stillbirth -- i.e. the fetus dies before it is born (see
also Fig. 22).
Figure 22. Fetotoxic effect (from HSG 19: Pentachlorophenol)
PCP is fetotoxic, delaying the development of rat embryos and
reducing litter size, neonatal body weight, neonatal survival,
and the growth of weanlings. The no-observed-adverse-effect
level for technical PCP is a maternal dose of 5 mg/kg body
weight per day during organogenesis. In one study, it was
reported that purified PCP was slightly more embryo/fetotoxic
than technical PCP, presumably because contaminants induced
enzymes that detoxified the parent compound.
2.4 Effects on body systems
Chemicals may have toxic effects on all cells in the body, or they may
affect only particular organs or body systems. Organs that are
especially sensitive to the toxic effects of a particular chemical are
known as target organs. A toxic action on one particular organ or
body system may be attributable to a particular property of the
chemical or related to the way in which a chemical enters or
circulates within the body. Toxic effects of chemicals on particular
organs and body systems include those on the:
* Lungs and the respiratory system. Short-term effects are caused
mainly by irritation (producing bronchitis or pneumonitis)
and chemical burns. Serious chemical burns inside the lungs can
result in the lungs becoming filled with fluid (pulmonary oedema)
and can be fatal. Some chemicals can sensitize or cause an
allergic reaction in the airways, leading to wheezing and
shortness of breath (asthma) (see Fig. 23).
Long-term (chronic) conditions include scarring of the lung
tissue following exposure to chemical dusts (fibrosis or
pneumoconiosis).
Figure 23. Effects on the respiratory system
(from HSG 35: Phosphorus Trichloride)
Depending on their concentration, the vapours can be irritating or
corrosive, but they may be less irritating than their hydrolysis
products. However, exposure to the vapours can cause necrosis of the
tissues of the respiratory tract. The vapours can be deeply inhaled
and may reach the lower airways where they hydrolyse to produce
hydrogen chloride and phosphorous or phosphoric acid.
Irritation of the airways causes swelling and bronchospasm resulting
in tightness of the chest, wheezing and difficulty in breathing.
Reactive secretion of mucus causes a cough with sputum and possible
bronchial obstruction and local lung collapse.
* Liver. Chemicals that can affect the liver are called
hepatotoxins. Most chemicals are metabolized in the liver, and
many therefore have the potential to damage the liver cells.
Possible short-term effects of chemicals on the liver include
inflammation of the liver cells (chemical hepatitis), necrosis
(cell death) and jaundice. Long-term effects include cirrhosis
(scarring) of the liver and liver cancer.
* Kidneys and urinary tract. Chemicals that can affect the kidneys
are called nephrotoxins. The effects of chemicals on the kidneys
include sudden failure of the kidneys (acute renal failure),
chronic renal failure and kidney or bladder cancer.
* Nervous system. Chemicals that affect the nerves are called
neurotoxins. Exposure to certain chemicals may cause a slowing
down of the function of the brain. Symptoms include sleepiness
and loss of alertness, followed by loss of consciousness (see
Fig. 24). Some of these effects are called "narcotic" effects,
based on the Greek word "narkos" meaning "sleep". Some illegal
drugs have narcotic effects and are therefore called narcotics.
Another term for these "sleep effects" is central nervous system
depression (not to be confused with depression as a mental health
problem). Other chemicals, particularly pesticides, can poison an
enzyme in the nerves going to the muscles, causing muscle
twitching and paralysis. Yet another group of chemicals may
slowly poison the nerves to the hands and feet, resulting in loss
of feeling and weakness.
Figure 24. Effects on the nervous system
(from HSG 3: 1-Butanol)
Ingestion of the liquid or inhalation of the vapour may result in
headache, drowsiness, and narcosis. The occurrence of vertigo under
conditions of severe and prolonged exposure to vapour mixtures of
1-butanol and isobutanol has been reported. From this study, it is
not possible to attribute the vertigo to a single cause. Symptoms are
reversible when exposure ceases.
* Blood and bone marrow. Some chemicals, such as arsine, damage
the red blood cells, causing haemolytic anaemia. Other chemicals
can damage the bone marrow and other organs where blood cells are
formed or can cause cancer of the blood-forming cells (e.g.
benzene causes leukaemia).
* Heart and blood vessels (cardiovascular system). Some solvents
(such as trichloroethylene) and gases can cause fatal
disturbances of the heart rhythm. Others, such as carbon
disulfide, may cause an increase in blood vessel disease, which
may lead to a heart attack.
* Skin. Many chemicals irritate the skin, causing "irritant
contact dermatitis", or they may sensitize the skin, causing
"allergic contact dermatitis". A rash results in both cases.
This rash may be very severe, widespread and disabling. Other
chemicals may cause acne (chloracne), loss of pigmentation
(vitiligo), increased sensitivity to sunlight
(photosensitization) or skin cancer.
* Reproductive system. Many chemicals are teratogens and
experimental germ cell mutagens. In addition, some chemicals can
directly affect the ovaries and testes, resulting in disruption
of menstruation and sexual function. Such chemicals are called
gonadotoxins.
* Other systems. Chemicals can also affect the immune system,
bones, muscles and particular glands, such as the thyroid gland.
2.5 Dose, effects and response
Increasing levels of exposure to or dose of a chemical will generally
lead to more severe effects. For instance, higher concentrations of
carbon monoxide in the air will progressively reduce the capacity of
an exposed person's blood to carry oxygen. The resultant lack of
oxygen in the blood leads initially to headaches. As the oxygen
levels decline further, the symptoms worsen: nausea, unconsciousness
and eventually death occur. This progression in severity of effect as
the dose increases is called the "dose-effect relationship".
In the case of a population or group of workers, increasing exposure
levels will also lead to an increasing proportion of the group
manifesting a specific effect. For example, increasing exposure to
benzidine dyes will result in a higher incidence of bladder cancers
among the exposed population. Similarly, increasing exposure to lead
will be reflected in a greater proportion of workers who undergo blood
changes (FEP, ALAD, etc.; see Fig. 25). This frequency of affected
people in an exposed population is called "the response". The
increase in response with increasing exposure level or dose is known
as the "dose"response relationship".
Another important concept is "threshold dose" or the
"no-observed-effect-level" (NOEL). This means that at low levels
of exposure to a chemical, the severity of the effect and the response
decrease, and that at a certain point there is no effect on health.
Usually this level is determined by exposing animals to lower and
lower concentrations of a chemical until a level is found at which no
effect on the animals is observed. It is impossible, however, to
examine every aspect of a test animal, and therefore effects may be
missed. Also, unless lifetime tests are performed (and they are not in
every case), long-term or chronic effects may also be missed, even
though subacute effects are noted. In addition, only limited numbers
of animals can be studied, whereas in real life many thousands of
people may be exposed to a chemical, some of whom will be much more
sensitive to its health effects than the majority. Even a very low
dose of a cancer-causing substance may increase the risk of cancer.
Studies of workers do not usually look at a sufficiently large group
to prove that exposure to low doses does, or does not, increase cancer
rates.
* Asphyxiant -- A chemical that interferes with the oxygenation of
the body tissues. There are two types of asphyxiation: simple
asphyxiation, whereby oxygen in the air is replaced by a gas to
a level at which it cannot sustain life; and chemical
asphyxiation, whereby a direct chemical action interferes with
the body's ability to transport and use oxygen. Examples of
chemical asphyxiants include carbon monoxide and cyanides.
* Carcinogen -- A chemical that causes cancer. Cancer is a group of
diseases characterized by the manner in which abnormal cells in
the body multiply and spread out of control. The key feature of
cancer is the "malignant" or deadly way that its cells crowd out
other cells and interfere with the normal functioning of the body
(see Fig. 20).
* Mutagen -- A chemical that can cause permanent damage to the
deoxyribonucleic acid (DNA) in a cell (see Fig. 21). DNA is
a molecule that carries the genetic information controlling the
growth and function of cells. DNA damage in the human egg or
sperm may lead to reduced fertility, spontaneous abortion
(miscarriage), birth defects and heritable diseases. Thus, any
chemical known to be a mutagen must be treated with caution.
Figure 20. Carcinogenic effect (from HSG 19: Pentachlorophenol)
The results of animal studies, designed to assess the carcinogenicity
of PCP and reported to date, have been negative. Carcinogenicity
bioassays with one other chlorophenol (2,4,6-T3CP) and a mixture of
two H6CDD congeners found in PCP have been positive. Hence, the
carcinogenic effects of long-term exposure of animals to technical PCP
are not clear.
Figure 21. Mutagenic effect (from HSG 32: d-Phenothrin)
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
* Teratogen -- A chemical that, if present in the bloodstream of a
pregnant woman, crosses the placenta, affecting the developing
fetus and resulting in structural or functional congenital
abnormalities, or cancer, in the child. These effects may not be
observable until the child becomes an adult. One of the
better-known examples of a teratogen is Thalidomide, which, in
the 1960s, caused many cases of phocomelia (a reduction of the
limbs to the extent that the hands and feet are attached directly
to the body) in babies born to women who took the drug during the
early stages of pregnancy. Several industrial chemicals have been
shown to be experimental teratogens; some, such as ethylene
dichloride and vinyl chloride, have been shown to be teratogenic
in humans.
* Fetotoxicant -- A chemical that adversely affects the developing
fetus, resulting in low birth weight, symptoms of poisoning at
birth or stillbirth -- i.e. the fetus dies before it is born (see
also Fig. 22).
Figure 22. Fetotoxic effect (from HSG 19: Pentachlorophenol)
PCP is fetotoxic, delaying the development of rat embryos and
reducing litter size, neonatal body weight, neonatal survival,
and the growth of weanlings. The no-observed-adverse-effect
level for technical PCP is a maternal dose of 5 mg/kg body
weight per day during organogenesis. In one study, it was
reported that purified PCP was slightly more embryo/fetotoxic
than technical PCP, presumably because contaminants induced
enzymes that detoxified the parent compound.
2.4 Effects on body systems
Chemicals may have toxic effects on all cells in the body, or they may
affect only particular organs or body systems. Organs that are
especially sensitive to the toxic effects of a particular chemical are
known as target organs. A toxic action on one particular organ or
body system may be attributable to a particular property of the
chemical or related to the way in which a chemical enters or
circulates within the body. Toxic effects of chemicals on particular
organs and body systems include those on the:
* Lungs and the respiratory system. Short-term effects are caused
mainly by irritation (producing bronchitis or pneumonitis)
and chemical burns. Serious chemical burns inside the lungs can
result in the lungs becoming filled with fluid (pulmonary oedema)
and can be fatal. Some chemicals can sensitize or cause an
allergic reaction in the airways, leading to wheezing and
shortness of breath (asthma) (see Fig. 23).
Long-term (chronic) conditions include scarring of the lung
tissue following exposure to chemical dusts (fibrosis or
pneumoconiosis).
Figure 23. Effects on the respiratory system
(from HSG 35: Phosphorus Trichloride)
Depending on their concentration, the vapours can be irritating or
corrosive, but they may be less irritating than their hydrolysis
products. However, exposure to the vapours can cause necrosis of the
tissues of the respiratory tract. The vapours can be deeply inhaled
and may reach the lower airways where they hydrolyse to produce
hydrogen chloride and phosphorous or phosphoric acid.
Irritation of the airways causes swelling and bronchospasm resulting
in tightness of the chest, wheezing and difficulty in breathing.
Reactive secretion of mucus causes a cough with sputum and possible
bronchial obstruction and local lung collapse.
* Liver. Chemicals that can affect the liver are called
hepatotoxins. Most chemicals are metabolized in the liver, and
many therefore have the potential to damage the liver cells.
Possible short-term effects of chemicals on the liver include
inflammation of the liver cells (chemical hepatitis), necrosis
(cell death) and jaundice. Long-term effects include cirrhosis
(scarring) of the liver and liver cancer.
* Kidneys and urinary tract. Chemicals that can affect the kidneys
are called nephrotoxins. The effects of chemicals on the kidneys
include sudden failure of the kidneys (acute renal failure),
chronic renal failure and kidney or bladder cancer.
* Nervous system. Chemicals that affect the nerves are called
neurotoxins. Exposure to certain chemicals may cause a slowing
down of the function of the brain. Symptoms include sleepiness
and loss of alertness, followed by loss of consciousness (see
Fig. 24). Some of these effects are called "narcotic" effects,
based on the Greek word "narkos" meaning "sleep". Some illegal
drugs have narcotic effects and are therefore called narcotics.
Another term for these "sleep effects" is central nervous system
depression (not to be confused with depression as a mental health
problem). Other chemicals, particularly pesticides, can poison an
enzyme in the nerves going to the muscles, causing muscle
twitching and paralysis. Yet another group of chemicals may
slowly poison the nerves to the hands and feet, resulting in loss
of feeling and weakness.
Figure 24. Effects on the nervous system
(from HSG 3: 1-Butanol)
Ingestion of the liquid or inhalation of the vapour may result in
headache, drowsiness, and narcosis. The occurrence of vertigo under
conditions of severe and prolonged exposure to vapour mixtures of
1-butanol and isobutanol has been reported. From this study, it is
not possible to attribute the vertigo to a single cause. Symptoms are
reversible when exposure ceases.
* Blood and bone marrow. Some chemicals, such as arsine, damage
the red blood cells, causing haemolytic anaemia. Other chemicals
can damage the bone marrow and other organs where blood cells are
formed or can cause cancer of the blood-forming cells (e.g.
benzene causes leukaemia).
* Heart and blood vessels (cardiovascular system). Some solvents
(such as trichloroethylene) and gases can cause fatal
disturbances of the heart rhythm. Others, such as carbon
disulfide, may cause an increase in blood vessel disease, which
may lead to a heart attack.
* Skin. Many chemicals irritate the skin, causing "irritant
contact dermatitis", or they may sensitize the skin, causing
"allergic contact dermatitis". A rash results in both cases.
This rash may be very severe, widespread and disabling. Other
chemicals may cause acne (chloracne), loss of pigmentation
(vitiligo), increased sensitivity to sunlight
(photosensitization) or skin cancer.
* Reproductive system. Many chemicals are teratogens and
experimental germ cell mutagens. In addition, some chemicals can
directly affect the ovaries and testes, resulting in disruption
of menstruation and sexual function. Such chemicals are called
gonadotoxins.
* Other systems. Chemicals can also affect the immune system,
bones, muscles and particular glands, such as the thyroid gland.
2.5 Dose, effects and response
Increasing levels of exposure to or dose of a chemical will generally
lead to more severe effects. For instance, higher concentrations of
carbon monoxide in the air will progressively reduce the capacity of
an exposed person's blood to carry oxygen. The resultant lack of
oxygen in the blood leads initially to headaches. As the oxygen
levels decline further, the symptoms worsen: nausea, unconsciousness
and eventually death occur. This progression in severity of effect as
the dose increases is called the "dose-effect relationship".
In the case of a population or group of workers, increasing exposure
levels will also lead to an increasing proportion of the group
manifesting a specific effect. For example, increasing exposure to
benzidine dyes will result in a higher incidence of bladder cancers
among the exposed population. Similarly, increasing exposure to lead
will be reflected in a greater proportion of workers who undergo blood
changes (FEP, ALAD, etc.; see Fig. 25). This frequency of affected
people in an exposed population is called "the response". The
increase in response with increasing exposure level or dose is known
as the "dose"response relationship".
Another important concept is "threshold dose" or the
"no-observed-effect-level" (NOEL). This means that at low levels
of exposure to a chemical, the severity of the effect and the response
decrease, and that at a certain point there is no effect on health.
Usually this level is determined by exposing animals to lower and
lower concentrations of a chemical until a level is found at which no
effect on the animals is observed. It is impossible, however, to
examine every aspect of a test animal, and therefore effects may be
missed. Also, unless lifetime tests are performed (and they are not in
every case), long-term or chronic effects may also be missed, even
though subacute effects are noted. In addition, only limited numbers
of animals can be studied, whereas in real life many thousands of
people may be exposed to a chemical, some of whom will be much more
sensitive to its health effects than the majority. Even a very low
dose of a cancer-causing substance may increase the risk of cancer.
Studies of workers do not usually look at a sufficiently large group
to prove that exposure to low doses does, or does not, increase cancer
rates.
 When drawing up regulations for the permissible (although not
necessarily safe) levels of chemicals in food, drinking-water and the
environment, government bodies usually divide the NOEL from animal
studies by a safety factor of 100 or 1000 (see Fig. 26). Occupational
exposure limits are usually set with a safety factor of 2-10 at the
most and even sometimes with no safety factor.
2.6 How is the toxicity of a chemical determined?
Knowledge of the toxicity of chemicals is gathered by studying the
effects of:
* exposure to the chemical in experimental animals, lower organisms
-- such as bacteria -- and laboratory cultures of cells from
mammals; and
* exposure of people to the chemical.
Figure 26. Use of safety factors (from HSG 19: Pentachlorophenol)
The US National Academy of Sciences (1977) calculated an acceptable
daily intake (ADI) for PCP of 3 µg/kg body weight per day. This ADI
is based on data from a feeding study on rats and a 1000-fold safety
factor. The results of long-term studies indicate that the
no-observed-adverse-effect level for rats is below 3 mg/kg body weight
per day. A recent human study has shown that the steady-state body
burden is 10-20 times higher than the value extrapolated from rat
pharmacokinetic data, suggesting that caution should be applied when
extrapolating directly from the rat model to humans. Furthermore, the
ADI in the USA was not based on an inhalation study, and does not
account for the possibly greater toxicity of PCP via inhalation, as
indicated by animal studies. Hence, the safety factor of 1000 used to
derive this ADI value is by no means too conservative. The intake for
a 60 kg adult exposed to concentrations of PCP at the ADI level would
be 180 µg/person per day.
2.6.1 Animal studies
2.6.1.1 Acute toxicity tests
* LD50 and LC50 tests
The standard test for acute (short-term) toxicity is to feed animals
increasing amounts of a chemical over a period of 14 days until the
animals start to die. Alternatively, the chemicals can be applied to
the animals' skin until a reaction is observed. The amount of the
chemical that kills 50% of the exposed animals is called the lethal
dose for 50%, or the LD50. The LD50 may be "oral" or "dermal",
depending on the method of exposure (see Fig. 27). Lethal doses with
respect to inhalation of chemicals in the form of a gas or aerosol can
also be tested. In this case, the concentration of gas or vapour that
kills half the animals is known as the lethal concentration for 50%,
or the LC50.
Figure 27. Effects on animals (from HSG 3: 1-Butanol)
The oral LD50 values for 1-butanol for the rat range from 0.7 to
2.1 g/kg body weight. Therefore, it is slightly toxic according to
the classification of Hodge & Sterner. It is markedly irritating to
the eyes and moderately irritating to the skin. The primary effects
from exposure to vapour for short periods are irritation of mucous
membranes, and central nervous system depression. The potency of
1-butanol for intoxication is approximately 6 times that of ethanol.
A number of investigations have shown non-specific membrane effects of
1-butanol. Effects of repeated inhalation exposure in animals include
pathological changes in the lungs, degenerative lesions in the liver
and kidneys, and narcosis. However, it is not possible to determine a
no-observed-adverse-effect level from the animal studies available.
1-Butanol has been found to be non-mutagenic. Adequate data are not
available on carcinogenicity, teratogenicity, or effects on
reproduction.
The LD50 and the LC50 are very widely used as indices of toxicity.
The following criteria are often used for purposes of classification
of acute toxic effects in animals (see Table 2):
Table 2. Classification of acute toxicity in animals
Oral LD50 Dermal LD50 Inhalation LC50
rat rat or rabbit rat
(mg/kg) (mg/kg) (mg/m3/4 h)
Harmful 200-2000 400-2000 2000-20 000
Toxic 25-200 50-400 500-2000
Very toxic < 25 < 50 < 500
The scale of Hodge & Sterner is also often used, in this case to
classify the acute toxicity of chemicals to humans (see Table 3):
Table 3. Classification of acute toxicity in humans
Toxicity rating Dosage Probable lethal
dose for average
human adult
1. Practically non-toxic > 15 g/kg > 1 litre
2. Slightly toxic 5-15 g/kg 0.5-1 litre
3. Moderately toxic 0.5-5 g/kg 30-500 ml
4. Very toxic 50-500 mg/kg 3-30 ml
5. Extremely toxic 5-50 mg/kg 7 drops-3 ml
6. Supertoxic < 5 mg/kg A taste (< 7 drops)
It is impossible to assess the health risk posed by a chemical on the
basis of its LD50 or LC50 alone. Moreover, the LD50 and LC50
give no information about the mechanism or type of toxicity of a
chemical, or its possible long-term or chronic effects.
Thus, the LD50 and LC50 are very crude indices of toxicity.
* The fixed-dose procedure
Many national and international bodies are now trying to modify or
replace the LD50 and LC50 tests by simpler methods, e.g. a
fixed-dose procedure that uses fewer animals. The fixed-dose
procedure requires only a small number of animals, and analysts can
evaluate a chemical's toxicity without the animals having to die as an
end-point. The idea is to examine how a set dose of a chemical
affects a group of animals, the dose being based on what is already
known about the physical and chemical properties of the substance
being evaluated.
* Irritation and corrosion tests
Irritation and corrosion tests provide some specific information. The
chemical being tested is applied to the test animal's skin, and the
area is examined over the next few days for signs of a rash or flared
response. Tests can also be carried out on the animal's eyes (this is
known as the "Draize" test).
2.6.1.2 Subchronic toxicity tests
Subchronic toxicity tests are normally 90-day inhalation or ingestion
studies to check for specific and obvious effects of the chemical on
the organs and biochemistry of the animal subjects. These types of
tests involve repeated exposure to the chemical being tested and are
primarily directed at detecting toxic effects that are not evident
from acute exposure. Crude studies simply call for examination of the
organs for gross abnormalities apparent to the naked eye; more
sophisticated studies involve taking tissue slices and examining them
under a microscope to check for abnormalities of the cells in the
organs. Most studies involve taking regular samples of blood or urine
from the animals for examination and analysis. These tests form the
basis for doses used in chronic bioassays (see next section).
2.6.1.3 Chronic (lifetime) bioassays
The purpose of lifetime or chronic bioassays is to determine whether
chemicals lead to any health effects that may take a long time to
develop, such as cancer, or whether long-term exposure to chemicals
leads to health effects on organs such as the kidneys (see Fig. 28).
Figure 28. Chronic bioassays (from HSG 34: Fenvalerate)
In a long-term toxicity study, microgranulomatous changes were also
observed in rats at 500 mg/kg in the diet. The NOEL for these
microgranulomatous changes was 150 mg/kg in the diet.
Fenvalerate, when fed at dietary levels up to 3000 mg/kg for 78 weeks
and 1250 mg/kg for 2 years, was not carcinogenic to mice. Nor was it
carcinogenic to rats when fed at dietary levels up to 1000 mg/kg for
2 years.
These studies are performed by exposing animals, by ingestion or
inhalation, to the chemical being tested, for most or all of the
animals' lifetime. In rats, this may be two years; in mice, a little
less. In a typical test, 50 mice or rats of each sex are exposed to a
high, but non-lethal, dose of the chemical under study. The test
animals are compared throughout their lifetime with a similar number
of "control" animals, which are similar in all respects except that
they are not exposed to the chemical. A good study exposes different
groups of animals of both sexes to different doses of the chemical.
Up to 500 animals may be used in a study of the chemical.
2.6.1.4 Short-term mutagenicity tests
Bacteria and animal cells grown in test-tubes and colonies of fruit
flies or other insects are convenient for the rapid and cheap
investigation of the potentially carcinogenic and mutagenic effects of
chemicals. The best-known and most widely used of these tests is the
Salmonella mutagenicity test (commonly known as the Ames test).
This involves growing special bacteria in the laboratory and exposing
them to the chemical in question. The test detects mutations in the
bacteria, i.e. it is a test for mutagenic effects. There are a number
of other short-term mutagenic tests or "mutagenicity assays", as they
are often called. These tests are often referred to as being in
vitro. They are thus distinguished from in vivo tests, which use
living tissue such as animals and humans (see Fig. 29).
Many, but not all, chemicals that cause cancer in animals and may
cause cancer in humans are mutagenic.
Figure 29. Short-term mutagenicity tests
(from HSG 34: Fenvalerate)
Fenvalerate did not show any mutagenic or chromosome-damaging
activities in several in vitro and in vivo assays.
2.6.1.5 Reproductive studies
Animal studies to check for the adverse effects of a chemical on
reproduction involve exposing one or both parents to the chemical
being tested, prior to mating, and then observing the effects on any
offspring. Sometimes only a pregnant animal is exposed. Reproductive
effects are classified according to whether the offspring are fewer in
number, lower in birth weight deformed or damaged in some way.
Multigeneration studies are sometimes necessary to detect effects that
may be passed on to future generations (see Fig. 30).
2.6.1.6 Behavioural tests
The effects of chemicals on the behaviour of test animals and on their
ability to learn (e.g. to find their way out of a maze) will often
indicate subtle effects of a chemical on the brain and nervous system
that are missed by other tests. Behavioural tests have been used to
show the effects of exposure to compounds such as organic solvents and
lead.
Figure 30. Reproductive effects (from HSG 34: Fenvalerate)
Fenvalerate is not teratogenic to mice or rabbits at doses up to
50 mg/kg body weight per day. It did not show any toxic effects on
reproductive parameters in a three-generation rat reproduction study
at doses up to 250 mg/kg diet.
2.6.2 Human evidence
2.6.2.1 Case reports
Case reports are accounts, usually by occupational health physicians,
of small numbers of workers or individual workers whose disease
appears to be attributable to workplace exposure. Often the part of a
process at which the chemical may have caused the problem is known,
but not the precise degree of exposure. Much information about
occupational health hazards has initially been found following
examination of case reports.
2.6.2.2 Epidemiological studies
Epidemiological studies investigate the health of a group of people to
establish whether they are affected by the chemical to which they are
exposed at work or in the general environment. These studies involve
comparing the disease outcome of an exposed group of people over time
with that of unexposed persons. Two of the more common methods of
investigation in epidemiology are case-control studies and cohort
studies.
Case-control studies are relatively simple to carry out and
increasingly used to investigate the cause of diseases - especially
rare diseases. They basically compare people with the disease, or
other outcome, under study with a suitable control group unaffected by
the disease or outcome in an attempt to identify the cause.
Cohort studies, also called follow-up or incidence studies, look
at a group of people (a cohort) who are classified into subgroups
according to exposure to a potential cause of disease or outcome.
Differences in exposure (to chemicals, for instance) are examined and
measured and the whole cohort followed up to see how the subsequent
development of the disease, or outcome, differs between the exposed
and unexposed groups (see Fig. 31).
When drawing up regulations for the permissible (although not
necessarily safe) levels of chemicals in food, drinking-water and the
environment, government bodies usually divide the NOEL from animal
studies by a safety factor of 100 or 1000 (see Fig. 26). Occupational
exposure limits are usually set with a safety factor of 2-10 at the
most and even sometimes with no safety factor.
2.6 How is the toxicity of a chemical determined?
Knowledge of the toxicity of chemicals is gathered by studying the
effects of:
* exposure to the chemical in experimental animals, lower organisms
-- such as bacteria -- and laboratory cultures of cells from
mammals; and
* exposure of people to the chemical.
Figure 26. Use of safety factors (from HSG 19: Pentachlorophenol)
The US National Academy of Sciences (1977) calculated an acceptable
daily intake (ADI) for PCP of 3 µg/kg body weight per day. This ADI
is based on data from a feeding study on rats and a 1000-fold safety
factor. The results of long-term studies indicate that the
no-observed-adverse-effect level for rats is below 3 mg/kg body weight
per day. A recent human study has shown that the steady-state body
burden is 10-20 times higher than the value extrapolated from rat
pharmacokinetic data, suggesting that caution should be applied when
extrapolating directly from the rat model to humans. Furthermore, the
ADI in the USA was not based on an inhalation study, and does not
account for the possibly greater toxicity of PCP via inhalation, as
indicated by animal studies. Hence, the safety factor of 1000 used to
derive this ADI value is by no means too conservative. The intake for
a 60 kg adult exposed to concentrations of PCP at the ADI level would
be 180 µg/person per day.
2.6.1 Animal studies
2.6.1.1 Acute toxicity tests
* LD50 and LC50 tests
The standard test for acute (short-term) toxicity is to feed animals
increasing amounts of a chemical over a period of 14 days until the
animals start to die. Alternatively, the chemicals can be applied to
the animals' skin until a reaction is observed. The amount of the
chemical that kills 50% of the exposed animals is called the lethal
dose for 50%, or the LD50. The LD50 may be "oral" or "dermal",
depending on the method of exposure (see Fig. 27). Lethal doses with
respect to inhalation of chemicals in the form of a gas or aerosol can
also be tested. In this case, the concentration of gas or vapour that
kills half the animals is known as the lethal concentration for 50%,
or the LC50.
Figure 27. Effects on animals (from HSG 3: 1-Butanol)
The oral LD50 values for 1-butanol for the rat range from 0.7 to
2.1 g/kg body weight. Therefore, it is slightly toxic according to
the classification of Hodge & Sterner. It is markedly irritating to
the eyes and moderately irritating to the skin. The primary effects
from exposure to vapour for short periods are irritation of mucous
membranes, and central nervous system depression. The potency of
1-butanol for intoxication is approximately 6 times that of ethanol.
A number of investigations have shown non-specific membrane effects of
1-butanol. Effects of repeated inhalation exposure in animals include
pathological changes in the lungs, degenerative lesions in the liver
and kidneys, and narcosis. However, it is not possible to determine a
no-observed-adverse-effect level from the animal studies available.
1-Butanol has been found to be non-mutagenic. Adequate data are not
available on carcinogenicity, teratogenicity, or effects on
reproduction.
The LD50 and the LC50 are very widely used as indices of toxicity.
The following criteria are often used for purposes of classification
of acute toxic effects in animals (see Table 2):
Table 2. Classification of acute toxicity in animals
Oral LD50 Dermal LD50 Inhalation LC50
rat rat or rabbit rat
(mg/kg) (mg/kg) (mg/m3/4 h)
Harmful 200-2000 400-2000 2000-20 000
Toxic 25-200 50-400 500-2000
Very toxic < 25 < 50 < 500
The scale of Hodge & Sterner is also often used, in this case to
classify the acute toxicity of chemicals to humans (see Table 3):
Table 3. Classification of acute toxicity in humans
Toxicity rating Dosage Probable lethal
dose for average
human adult
1. Practically non-toxic > 15 g/kg > 1 litre
2. Slightly toxic 5-15 g/kg 0.5-1 litre
3. Moderately toxic 0.5-5 g/kg 30-500 ml
4. Very toxic 50-500 mg/kg 3-30 ml
5. Extremely toxic 5-50 mg/kg 7 drops-3 ml
6. Supertoxic < 5 mg/kg A taste (< 7 drops)
It is impossible to assess the health risk posed by a chemical on the
basis of its LD50 or LC50 alone. Moreover, the LD50 and LC50
give no information about the mechanism or type of toxicity of a
chemical, or its possible long-term or chronic effects.
Thus, the LD50 and LC50 are very crude indices of toxicity.
* The fixed-dose procedure
Many national and international bodies are now trying to modify or
replace the LD50 and LC50 tests by simpler methods, e.g. a
fixed-dose procedure that uses fewer animals. The fixed-dose
procedure requires only a small number of animals, and analysts can
evaluate a chemical's toxicity without the animals having to die as an
end-point. The idea is to examine how a set dose of a chemical
affects a group of animals, the dose being based on what is already
known about the physical and chemical properties of the substance
being evaluated.
* Irritation and corrosion tests
Irritation and corrosion tests provide some specific information. The
chemical being tested is applied to the test animal's skin, and the
area is examined over the next few days for signs of a rash or flared
response. Tests can also be carried out on the animal's eyes (this is
known as the "Draize" test).
2.6.1.2 Subchronic toxicity tests
Subchronic toxicity tests are normally 90-day inhalation or ingestion
studies to check for specific and obvious effects of the chemical on
the organs and biochemistry of the animal subjects. These types of
tests involve repeated exposure to the chemical being tested and are
primarily directed at detecting toxic effects that are not evident
from acute exposure. Crude studies simply call for examination of the
organs for gross abnormalities apparent to the naked eye; more
sophisticated studies involve taking tissue slices and examining them
under a microscope to check for abnormalities of the cells in the
organs. Most studies involve taking regular samples of blood or urine
from the animals for examination and analysis. These tests form the
basis for doses used in chronic bioassays (see next section).
2.6.1.3 Chronic (lifetime) bioassays
The purpose of lifetime or chronic bioassays is to determine whether
chemicals lead to any health effects that may take a long time to
develop, such as cancer, or whether long-term exposure to chemicals
leads to health effects on organs such as the kidneys (see Fig. 28).
Figure 28. Chronic bioassays (from HSG 34: Fenvalerate)
In a long-term toxicity study, microgranulomatous changes were also
observed in rats at 500 mg/kg in the diet. The NOEL for these
microgranulomatous changes was 150 mg/kg in the diet.
Fenvalerate, when fed at dietary levels up to 3000 mg/kg for 78 weeks
and 1250 mg/kg for 2 years, was not carcinogenic to mice. Nor was it
carcinogenic to rats when fed at dietary levels up to 1000 mg/kg for
2 years.
These studies are performed by exposing animals, by ingestion or
inhalation, to the chemical being tested, for most or all of the
animals' lifetime. In rats, this may be two years; in mice, a little
less. In a typical test, 50 mice or rats of each sex are exposed to a
high, but non-lethal, dose of the chemical under study. The test
animals are compared throughout their lifetime with a similar number
of "control" animals, which are similar in all respects except that
they are not exposed to the chemical. A good study exposes different
groups of animals of both sexes to different doses of the chemical.
Up to 500 animals may be used in a study of the chemical.
2.6.1.4 Short-term mutagenicity tests
Bacteria and animal cells grown in test-tubes and colonies of fruit
flies or other insects are convenient for the rapid and cheap
investigation of the potentially carcinogenic and mutagenic effects of
chemicals. The best-known and most widely used of these tests is the
Salmonella mutagenicity test (commonly known as the Ames test).
This involves growing special bacteria in the laboratory and exposing
them to the chemical in question. The test detects mutations in the
bacteria, i.e. it is a test for mutagenic effects. There are a number
of other short-term mutagenic tests or "mutagenicity assays", as they
are often called. These tests are often referred to as being in
vitro. They are thus distinguished from in vivo tests, which use
living tissue such as animals and humans (see Fig. 29).
Many, but not all, chemicals that cause cancer in animals and may
cause cancer in humans are mutagenic.
Figure 29. Short-term mutagenicity tests
(from HSG 34: Fenvalerate)
Fenvalerate did not show any mutagenic or chromosome-damaging
activities in several in vitro and in vivo assays.
2.6.1.5 Reproductive studies
Animal studies to check for the adverse effects of a chemical on
reproduction involve exposing one or both parents to the chemical
being tested, prior to mating, and then observing the effects on any
offspring. Sometimes only a pregnant animal is exposed. Reproductive
effects are classified according to whether the offspring are fewer in
number, lower in birth weight deformed or damaged in some way.
Multigeneration studies are sometimes necessary to detect effects that
may be passed on to future generations (see Fig. 30).
2.6.1.6 Behavioural tests
The effects of chemicals on the behaviour of test animals and on their
ability to learn (e.g. to find their way out of a maze) will often
indicate subtle effects of a chemical on the brain and nervous system
that are missed by other tests. Behavioural tests have been used to
show the effects of exposure to compounds such as organic solvents and
lead.
Figure 30. Reproductive effects (from HSG 34: Fenvalerate)
Fenvalerate is not teratogenic to mice or rabbits at doses up to
50 mg/kg body weight per day. It did not show any toxic effects on
reproductive parameters in a three-generation rat reproduction study
at doses up to 250 mg/kg diet.
2.6.2 Human evidence
2.6.2.1 Case reports
Case reports are accounts, usually by occupational health physicians,
of small numbers of workers or individual workers whose disease
appears to be attributable to workplace exposure. Often the part of a
process at which the chemical may have caused the problem is known,
but not the precise degree of exposure. Much information about
occupational health hazards has initially been found following
examination of case reports.
2.6.2.2 Epidemiological studies
Epidemiological studies investigate the health of a group of people to
establish whether they are affected by the chemical to which they are
exposed at work or in the general environment. These studies involve
comparing the disease outcome of an exposed group of people over time
with that of unexposed persons. Two of the more common methods of
investigation in epidemiology are case-control studies and cohort
studies.
Case-control studies are relatively simple to carry out and
increasingly used to investigate the cause of diseases - especially
rare diseases. They basically compare people with the disease, or
other outcome, under study with a suitable control group unaffected by
the disease or outcome in an attempt to identify the cause.
Cohort studies, also called follow-up or incidence studies, look
at a group of people (a cohort) who are classified into subgroups
according to exposure to a potential cause of disease or outcome.
Differences in exposure (to chemicals, for instance) are examined and
measured and the whole cohort followed up to see how the subsequent
development of the disease, or outcome, differs between the exposed
and unexposed groups (see Fig. 31).
 Although epidemiological investigations provide the most dependable
proof that a given chemical has an adverse effect on the health of a
population, they do have several disadvantages. Only a small group of
chemicals in use has been studied in this way because of the expense
involved and the need for good exposure records and a sufficiently
large number of exposed workers to ensure the validity of the
statistical calculations. New chemicals, of course, cannot be studied
in this manner as there will be no history of exposure. In addition,
epidemiological studies may not be able to detect rare events or
pinpoint the role of one particular chemical when workers are exposed
to mixtures of chemicals. But, above all, waiting for the results of
such studies can mean many unnecessary and largely preventable deaths
and illnesses among exposed persons. If information from epidemio-
logical studies is lacking, preventive action should be instigated
based on animal studies (see Fig. 32).
Figure 32. Human studies (from HSG 19: Pentachlorophenol)
Past use of PCP has affected workers producing or using this chemical.
Chloracne, skin irritation and rashes, respiratory disorder,
neurological changes, headaches, nausea, weakness, irritability, and
drowsiness have been documented in exposed workers.
Investigations of biochemical changes in woodworkers with long-term
exposure to PCP have failed to detect consistently significant effects
on major organs, nerves, blood, reproduction, or the immune system.
However, the statistical power of these studies has been limited as a
result of the small sample sizes used. Overall, the body of research
suggests that long-term exposure to levels of PCP encountered in the
workplace is likely to cause borderline effects on some organ systems
and biochemical processes.
Some epidemiological studies from Sweden and the USA have revealed an
association between exposure to mixtures of chlorophenols, especially
2,4,5-T3CP, and the incidences of soft-tissue sarcomas, lymphomas, and
nasal and nasopharyngeal cancers. Other studies have failed to detect
such a relationship. It was not possible to address the effects of
exposure to PCP itself in any of these studies.
The theory behind animal tests is that humans and animals such as
rats, mice or dogs share the same basic biochemistry and bodily
processes. Although there are differences between animals and humans,
the similarities in terms of the way chemicals act on the body are far
greater. Animal tests make it possible to test the toxicity of a
chemical before people are exposed to it. Such results should be used
in health and safety guidelines. This should reduce the occurrence of
unwanted effects of chemicals on human health. Exposing workers or
the general community to a chemical and waiting for human
epidemiological data to appear before warning workers and acting to
protect the general community are now unacceptable. On the other
hand, epidemiological studies will always be essential to monitor the
health of people exposed to chemicals that are already in use and to
check that preventive measures are effective.
2.6.3 Environmental assays
These tests attempt to predict whether a chemical will have unwanted
effects on animals and plants in the environment and whether chemicals
are likely to be present in water supplies and food.
2.7 Assessing hazards, risks and safety
In a general sense, the toxicity of a substance can be defined as the
substance's capacity to harm a living organism. A highly toxic
substance will harm an organism even if only very small amounts are
present in the body. Conversely, a substance of low toxicity will not
produce an effect unless the amount present in the body is very large.
The main factors that must be considered when assessing the toxicity
of a substance include:
* the quantity of the substance absorbed (the dose) by the person
exposed to that chemical;
* the route via which exposure to the chemical occurs (e.g.
inhalation, ingestion, or absorption through the skin);
* the duration of exposure to the chemical and how often that
exposure occurs;
* the type and severity of the injury caused by exposure to the
chemical; and
* whether or not that injury is permanent or reversible, e.g.
cancer is irreversible, although sometimes treatable.
Three other terms are commonly used when the toxicity of a chemical is
discussed: hazard, risk and safety.
2.7.1 Hazard
A hazard can be defined as the set of inherent properties of a
chemical, mixture of chemicals or a process that, under production,
usage or disposal conditions, has the potential to adversely affect
the environment or the organisms it contains. In other words, it is a
source of danger.
2.7.2 Risk
It is important to distinguish risk from hazard. Hazard refers to the
intrinsic properties of a chemical, whereas risk refers to the chance
or probability that the chemical will cause an adverse health effect.
If there is a high risk that a certain chemical will cause cancer in
exposed workers, then it is very likely that some of those workers
will develop cancer. If the risk is low, then it is less likely that
the workers will develop cancer. However, even if the risk of some
health effect is low, the chemical in question is still a hazard.
Depending on the circumstances, a "low risk" may be acceptable to the
people exposed. Determining the "acceptable risk" is part of the
process for setting safety standards.
* Risk assessment involves identification of the hazard
(the chemical of concern, for instance, and its adverse effects,
target populations and conditions of exposure); characterizing
the risk; assessing exposure (by measuring and monitoring); and
estimating the risk. Thus, it consists of identification and
quantification of the risk resulting from a specific use or
occurrence of a chemical, taking into account the possible
harmful effects on individuals of using the chemical in the
manner and amount proposed, and all possible routes of exposure.
* Risk management covers the whole range of actions taken to
prevent, minimize or otherwise control specific risks posed by a
certain chemical or situation. It is based on concepts of safety
and therefore contains elements of policy relating to political,
social and economic factors, as well as engineering and process
control.
2.7.3 Safety
Safety is even more difficult to define than risk or hazard. The
safety of a chemical, in the context of human health, is the extent to
which a chemical may be used in the amount necessary for the intended
purpose, with a minimum risk of adverse health effects. It can also
be defined as a "socially acceptable" level of risk. But it is
usually unclear which part of society is judging the risk. Workers
that are exposed to the risk are likely to be more concerned about the
safety of a chemical than others. Therefore, it is very important to
question statements such as "this chemical is safe" or "there is a
high level of safety when using this chemical". Safety is a
subjective concept.
3. THE PREVENTION AND CONTROL OF CHEMICAL HAZARDS: HOW TO IMPROVE
CHEMICAL SAFETY - CHAPTER 3
This chapter corresponds to Chapter 3 of each HSG (Conclusions and
recommendations), which evaluates the degree of hazard of a chemical
and recommends control measures to limit exposure (see Fig. 33).
Figure 33. Conclusions and recommendations
(from HSG 35: Phosphorus Trichloride)
3.1 Conclusions
Phosphorus trichloride and phosphorus oxychloride are highly reactive
and hazardous corrosive chemicals. Intense exposure to their vapours
may leave a residual restrictive defect in the lungs. Repeated minor
overexposures may result in progressive impairment of lung function
and can be fatal. Apart from spillages that destroy plant and animal
life in the immediate area, the impact of these chemicals on the
environment is negligible.
3.2 Recommendations
Phosphorus trichloride and phosphorus oxychloride should only be used
and handled under the careful supervision of managers who fully
understand the hazards and the good handling and manufacturing
practices necessary to control them. These managers should train
operators, maintenance personnel and contractors about the hazards and
safety procedures.
There are four steps in the prevention and control of workplace
chemical hazards:
* identification of the hazard;
* evaluation of the hazard and risk;
* organization to prevent, control or eliminate the risk; and
* controlling the hazard through specific actions.
Although epidemiological investigations provide the most dependable
proof that a given chemical has an adverse effect on the health of a
population, they do have several disadvantages. Only a small group of
chemicals in use has been studied in this way because of the expense
involved and the need for good exposure records and a sufficiently
large number of exposed workers to ensure the validity of the
statistical calculations. New chemicals, of course, cannot be studied
in this manner as there will be no history of exposure. In addition,
epidemiological studies may not be able to detect rare events or
pinpoint the role of one particular chemical when workers are exposed
to mixtures of chemicals. But, above all, waiting for the results of
such studies can mean many unnecessary and largely preventable deaths
and illnesses among exposed persons. If information from epidemio-
logical studies is lacking, preventive action should be instigated
based on animal studies (see Fig. 32).
Figure 32. Human studies (from HSG 19: Pentachlorophenol)
Past use of PCP has affected workers producing or using this chemical.
Chloracne, skin irritation and rashes, respiratory disorder,
neurological changes, headaches, nausea, weakness, irritability, and
drowsiness have been documented in exposed workers.
Investigations of biochemical changes in woodworkers with long-term
exposure to PCP have failed to detect consistently significant effects
on major organs, nerves, blood, reproduction, or the immune system.
However, the statistical power of these studies has been limited as a
result of the small sample sizes used. Overall, the body of research
suggests that long-term exposure to levels of PCP encountered in the
workplace is likely to cause borderline effects on some organ systems
and biochemical processes.
Some epidemiological studies from Sweden and the USA have revealed an
association between exposure to mixtures of chlorophenols, especially
2,4,5-T3CP, and the incidences of soft-tissue sarcomas, lymphomas, and
nasal and nasopharyngeal cancers. Other studies have failed to detect
such a relationship. It was not possible to address the effects of
exposure to PCP itself in any of these studies.
The theory behind animal tests is that humans and animals such as
rats, mice or dogs share the same basic biochemistry and bodily
processes. Although there are differences between animals and humans,
the similarities in terms of the way chemicals act on the body are far
greater. Animal tests make it possible to test the toxicity of a
chemical before people are exposed to it. Such results should be used
in health and safety guidelines. This should reduce the occurrence of
unwanted effects of chemicals on human health. Exposing workers or
the general community to a chemical and waiting for human
epidemiological data to appear before warning workers and acting to
protect the general community are now unacceptable. On the other
hand, epidemiological studies will always be essential to monitor the
health of people exposed to chemicals that are already in use and to
check that preventive measures are effective.
2.6.3 Environmental assays
These tests attempt to predict whether a chemical will have unwanted
effects on animals and plants in the environment and whether chemicals
are likely to be present in water supplies and food.
2.7 Assessing hazards, risks and safety
In a general sense, the toxicity of a substance can be defined as the
substance's capacity to harm a living organism. A highly toxic
substance will harm an organism even if only very small amounts are
present in the body. Conversely, a substance of low toxicity will not
produce an effect unless the amount present in the body is very large.
The main factors that must be considered when assessing the toxicity
of a substance include:
* the quantity of the substance absorbed (the dose) by the person
exposed to that chemical;
* the route via which exposure to the chemical occurs (e.g.
inhalation, ingestion, or absorption through the skin);
* the duration of exposure to the chemical and how often that
exposure occurs;
* the type and severity of the injury caused by exposure to the
chemical; and
* whether or not that injury is permanent or reversible, e.g.
cancer is irreversible, although sometimes treatable.
Three other terms are commonly used when the toxicity of a chemical is
discussed: hazard, risk and safety.
2.7.1 Hazard
A hazard can be defined as the set of inherent properties of a
chemical, mixture of chemicals or a process that, under production,
usage or disposal conditions, has the potential to adversely affect
the environment or the organisms it contains. In other words, it is a
source of danger.
2.7.2 Risk
It is important to distinguish risk from hazard. Hazard refers to the
intrinsic properties of a chemical, whereas risk refers to the chance
or probability that the chemical will cause an adverse health effect.
If there is a high risk that a certain chemical will cause cancer in
exposed workers, then it is very likely that some of those workers
will develop cancer. If the risk is low, then it is less likely that
the workers will develop cancer. However, even if the risk of some
health effect is low, the chemical in question is still a hazard.
Depending on the circumstances, a "low risk" may be acceptable to the
people exposed. Determining the "acceptable risk" is part of the
process for setting safety standards.
* Risk assessment involves identification of the hazard
(the chemical of concern, for instance, and its adverse effects,
target populations and conditions of exposure); characterizing
the risk; assessing exposure (by measuring and monitoring); and
estimating the risk. Thus, it consists of identification and
quantification of the risk resulting from a specific use or
occurrence of a chemical, taking into account the possible
harmful effects on individuals of using the chemical in the
manner and amount proposed, and all possible routes of exposure.
* Risk management covers the whole range of actions taken to
prevent, minimize or otherwise control specific risks posed by a
certain chemical or situation. It is based on concepts of safety
and therefore contains elements of policy relating to political,
social and economic factors, as well as engineering and process
control.
2.7.3 Safety
Safety is even more difficult to define than risk or hazard. The
safety of a chemical, in the context of human health, is the extent to
which a chemical may be used in the amount necessary for the intended
purpose, with a minimum risk of adverse health effects. It can also
be defined as a "socially acceptable" level of risk. But it is
usually unclear which part of society is judging the risk. Workers
that are exposed to the risk are likely to be more concerned about the
safety of a chemical than others. Therefore, it is very important to
question statements such as "this chemical is safe" or "there is a
high level of safety when using this chemical". Safety is a
subjective concept.
3. THE PREVENTION AND CONTROL OF CHEMICAL HAZARDS: HOW TO IMPROVE
CHEMICAL SAFETY - CHAPTER 3
This chapter corresponds to Chapter 3 of each HSG (Conclusions and
recommendations), which evaluates the degree of hazard of a chemical
and recommends control measures to limit exposure (see Fig. 33).
Figure 33. Conclusions and recommendations
(from HSG 35: Phosphorus Trichloride)
3.1 Conclusions
Phosphorus trichloride and phosphorus oxychloride are highly reactive
and hazardous corrosive chemicals. Intense exposure to their vapours
may leave a residual restrictive defect in the lungs. Repeated minor
overexposures may result in progressive impairment of lung function
and can be fatal. Apart from spillages that destroy plant and animal
life in the immediate area, the impact of these chemicals on the
environment is negligible.
3.2 Recommendations
Phosphorus trichloride and phosphorus oxychloride should only be used
and handled under the careful supervision of managers who fully
understand the hazards and the good handling and manufacturing
practices necessary to control them. These managers should train
operators, maintenance personnel and contractors about the hazards and
safety procedures.
There are four steps in the prevention and control of workplace
chemical hazards:
* identification of the hazard;
* evaluation of the hazard and risk;
* organization to prevent, control or eliminate the risk; and
* controlling the hazard through specific actions.
 3.1 Identification
All workers have the right to know the possible effects of their work
on their health and safety. This includes the right of access to
information about the health effects of chemicals, other substances
and work processes and about procedures for healthy and safe systems
of work.
In some countries, the right to know this information is backed up by
special laws. Under these laws, employers and manufacturers,
suppliers and importers of chemicals must provide clear, detailed
information about the particular chemical, substance or product, its
possible health effects, including the results of animal tests and
surveys of exposed workers (see Chapter 2), and means of protecting
workers from any harmful effects.
These legal rights may apply to each worker or to elected health and
safety representatives or committees. Find out from your employer,
trade union or government occupational health agency if you are
covered by such a law.
3.2 Evaluation
If "right-to-know" laws operate, employers, manufacturers and
suppliers of chemical products are required to:
* produce Material Safety Data Sheets (MSDSs) for all chemicals
used in the workplace;
* label chemical products clearly to indicate their (potentially)
harmful effects, and provide guidance on how to use the products
as safely as possible; and
* instruct workers in the meaning of labels and MSDSs.
Development of a workplace chemical register is one means of
evaluating hazards. This is simply a list of every chemical used in
the workplace. An MSDS should be available for every chemical on the
Register.
Workers should have the right to refuse to work with chemicals for
which full health and safety information is not available. If you
experience difficulty obtaining full information from your employer,
contact your government health or labour ministry, trade union or a
workers' health or environmental organization for help.
Most HSGs available from the IPCS also contain an ICSC or Summary of
Chemical Safety Information for the chemical concerned. Each ICSC is
a brief, internationally reviewed summary of the properties, hazards,
preventive methods and emergency treatment relating to the chemical in
question.
Workers' representatives should be given the opportunity to review
complete health and safety information before any new chemical is
introduced into the workplace. Arrangements for the safe use of new
chemicals should be finalized and put into practice before a chemical
is introduced.
3.3 Safety organization
All workplaces should implement effective safety procedures for
protection against chemical hazards agreed jointly by employer and
workers. In some countries, these agreements will be negotiated as
collective bargaining agreements or health and safety agreements
between management and workers. Sometimes these agreements are
additional to the minimum obligations imposed on employers by
workplace health and safety laws.
The employer and elected worker health and safety representatives
and/or committees in each workplace should participate in the
identification and control of chemical hazards through:
* regular inspections with standard checklists for particular
chemicals and chemical processes;
* investigation of workers' complaints;
* use of accident and sickness records;
* regular surveys of workers' health;
* environmental and biological monitoring;
* assessment of government inspectors'/consultants' reports;
* investigation of the causes of accidents and their prevention;
and
* development of a workplace chemical register.
3.4 Controlling the hazard
The prevention of adverse health effects arising from occupational
exposure to chemicals requires a comprehensive control strategy.
Ideally, exposure should be prevented altogether, i.e. at the source,
through substitution or enclosed processes, for example. If this
cannot be achieved, the level of exposure should be reduced as much as
possible, i.e. during the transmission stage, through ventilation and
the use of protective clothing -- and, certainly, to levels at which
neither health effects nor irritation occurs. A third strategy
comprises measures to counteract the effects of exposure through early
diagnosis of any disease and attempts to prevent the progression of
existing disease, through regular medical monitoring.
The following controls may be used, in descending order of priority:
* substitution of hazardous chemicals or processes with less
hazardous ones;
* engineering controls, e.g. improved ventilation;
* development of safe working procedures;
* reduction of the number of exposed workers;
* reduction of the duration and/or frequency of exposure of
workers;
* use of personal protective equipment, e.g. respirators, goggles;
and
* regular environmental and biological (medical) monitoring or
surveillance to check that the above control methods are proving
effective.
3.4.1 Substitution
The most effective control measure for any hazardous chemical is to
remove it entirely from the workplace and replace it with a less
hazardous chemical. This is crucial for very toxic chemicals,
carcinogens, chemicals that can damage the reproductive system and
sensitizing agents. This approach should, of course, be applied to all
chemical hazards.
An example of substitution is the replacement of the solvents
2-methoxy- and 2-ethoxy-ethanol (commonly used in paints and lacquers)
with the solvent 2-butoxy-ethanol. Both 2-methoxy- and
2-ethoxy-ethanol cause reproductive health effects (including
shrinking of the testicles and birth defects) in animals at low levels
of exposure; 2-butoxy-ethanol has not been found to cause these
effects. In terms of solvent properties, there is no significant
difference between any of these three solvents.
Care must be taken to obtain all available information on proposed
alternative chemicals. Substitutes may turn out to be just as
hazardous as or even more hazardous than the materials they replace.
It is also often possible to substitute safer processes. In this
connection, a process is the sequence of steps involved in the
manufacture or use of a chemical. The manufacture of chemicals
usually entails a series of intermediate stages. The chemicals
produced during these stages (called intermediates) are sometimes
more toxic than the starting or final materials. Whenever possible,
dangerous processes should be substituted to avoid the production of
toxic intermediates.
3.4.2 Engineering controls
3.4.2.1 Total enclosure
If a chemical hazard cannot be removed from the workplace by
substitution, then the next best solution is to physically enclose the
hazard to prevent it from coming into contact with either workers or
the environment. This is known as total enclosure or containment of
a process (see Fig. 35). For example, open tanks from which chemical
vapours can escape into the workplace air can be replaced with closed
tanks with inlet and outlet ports for filling and emptying. Liquids
such as solvents can be transferred by being pumped through sealed
pipes rather than poured in the open air.
3.1 Identification
All workers have the right to know the possible effects of their work
on their health and safety. This includes the right of access to
information about the health effects of chemicals, other substances
and work processes and about procedures for healthy and safe systems
of work.
In some countries, the right to know this information is backed up by
special laws. Under these laws, employers and manufacturers,
suppliers and importers of chemicals must provide clear, detailed
information about the particular chemical, substance or product, its
possible health effects, including the results of animal tests and
surveys of exposed workers (see Chapter 2), and means of protecting
workers from any harmful effects.
These legal rights may apply to each worker or to elected health and
safety representatives or committees. Find out from your employer,
trade union or government occupational health agency if you are
covered by such a law.
3.2 Evaluation
If "right-to-know" laws operate, employers, manufacturers and
suppliers of chemical products are required to:
* produce Material Safety Data Sheets (MSDSs) for all chemicals
used in the workplace;
* label chemical products clearly to indicate their (potentially)
harmful effects, and provide guidance on how to use the products
as safely as possible; and
* instruct workers in the meaning of labels and MSDSs.
Development of a workplace chemical register is one means of
evaluating hazards. This is simply a list of every chemical used in
the workplace. An MSDS should be available for every chemical on the
Register.
Workers should have the right to refuse to work with chemicals for
which full health and safety information is not available. If you
experience difficulty obtaining full information from your employer,
contact your government health or labour ministry, trade union or a
workers' health or environmental organization for help.
Most HSGs available from the IPCS also contain an ICSC or Summary of
Chemical Safety Information for the chemical concerned. Each ICSC is
a brief, internationally reviewed summary of the properties, hazards,
preventive methods and emergency treatment relating to the chemical in
question.
Workers' representatives should be given the opportunity to review
complete health and safety information before any new chemical is
introduced into the workplace. Arrangements for the safe use of new
chemicals should be finalized and put into practice before a chemical
is introduced.
3.3 Safety organization
All workplaces should implement effective safety procedures for
protection against chemical hazards agreed jointly by employer and
workers. In some countries, these agreements will be negotiated as
collective bargaining agreements or health and safety agreements
between management and workers. Sometimes these agreements are
additional to the minimum obligations imposed on employers by
workplace health and safety laws.
The employer and elected worker health and safety representatives
and/or committees in each workplace should participate in the
identification and control of chemical hazards through:
* regular inspections with standard checklists for particular
chemicals and chemical processes;
* investigation of workers' complaints;
* use of accident and sickness records;
* regular surveys of workers' health;
* environmental and biological monitoring;
* assessment of government inspectors'/consultants' reports;
* investigation of the causes of accidents and their prevention;
and
* development of a workplace chemical register.
3.4 Controlling the hazard
The prevention of adverse health effects arising from occupational
exposure to chemicals requires a comprehensive control strategy.
Ideally, exposure should be prevented altogether, i.e. at the source,
through substitution or enclosed processes, for example. If this
cannot be achieved, the level of exposure should be reduced as much as
possible, i.e. during the transmission stage, through ventilation and
the use of protective clothing -- and, certainly, to levels at which
neither health effects nor irritation occurs. A third strategy
comprises measures to counteract the effects of exposure through early
diagnosis of any disease and attempts to prevent the progression of
existing disease, through regular medical monitoring.
The following controls may be used, in descending order of priority:
* substitution of hazardous chemicals or processes with less
hazardous ones;
* engineering controls, e.g. improved ventilation;
* development of safe working procedures;
* reduction of the number of exposed workers;
* reduction of the duration and/or frequency of exposure of
workers;
* use of personal protective equipment, e.g. respirators, goggles;
and
* regular environmental and biological (medical) monitoring or
surveillance to check that the above control methods are proving
effective.
3.4.1 Substitution
The most effective control measure for any hazardous chemical is to
remove it entirely from the workplace and replace it with a less
hazardous chemical. This is crucial for very toxic chemicals,
carcinogens, chemicals that can damage the reproductive system and
sensitizing agents. This approach should, of course, be applied to all
chemical hazards.
An example of substitution is the replacement of the solvents
2-methoxy- and 2-ethoxy-ethanol (commonly used in paints and lacquers)
with the solvent 2-butoxy-ethanol. Both 2-methoxy- and
2-ethoxy-ethanol cause reproductive health effects (including
shrinking of the testicles and birth defects) in animals at low levels
of exposure; 2-butoxy-ethanol has not been found to cause these
effects. In terms of solvent properties, there is no significant
difference between any of these three solvents.
Care must be taken to obtain all available information on proposed
alternative chemicals. Substitutes may turn out to be just as
hazardous as or even more hazardous than the materials they replace.
It is also often possible to substitute safer processes. In this
connection, a process is the sequence of steps involved in the
manufacture or use of a chemical. The manufacture of chemicals
usually entails a series of intermediate stages. The chemicals
produced during these stages (called intermediates) are sometimes
more toxic than the starting or final materials. Whenever possible,
dangerous processes should be substituted to avoid the production of
toxic intermediates.
3.4.2 Engineering controls
3.4.2.1 Total enclosure
If a chemical hazard cannot be removed from the workplace by
substitution, then the next best solution is to physically enclose the
hazard to prevent it from coming into contact with either workers or
the environment. This is known as total enclosure or containment of
a process (see Fig. 35). For example, open tanks from which chemical
vapours can escape into the workplace air can be replaced with closed
tanks with inlet and outlet ports for filling and emptying. Liquids
such as solvents can be transferred by being pumped through sealed
pipes rather than poured in the open air.
 3.4.2.2 Ventilation
Ventilation systems are one means of removing contaminated air from
the workplace. There are two general types of ventilation:
* dilution or general ventilation; and
* local exhaust ventilation.
Dilution ventilation is simply the process whereby clean air is mixed
with contaminated air. The concentration of the airborne
contaminant is thus reduced, although the workplace air will still
contain some of the contaminant.
A simple dilution ventilation system consists of two major components:
a source of clean air and an exhaust fan for removing the contaminated
air. The ventilation system can be totally passive, which means that
the exhaust is a chimney or open vent in the ceiling from which the
dirty air is expelled, and the source of clean air an open inlet in
one of the workplace walls (see Fig. 36). This is the most basic, i.e.
least effective, system. An improvement on this system would be a fan
at the exhaust forcing the dirty air out. An even better system would
have both an exhaust fan and an inlet fan.
Dilution ventilation is rarely an adequate means of safeguarding
health in the workplace, as:
* it does not prevent the release of a contaminant into the
workplace air;
* it is ineffective against dusts, metal particles and metal fumes
that are too heavy to be removed by gentle air movement;
* it does not provide sufficient protection against highly toxic
chemical vapours and gases, carcinogens or chemicals causing
reproductive effects;
* it is ineffective against "surges" of gases and vapours or
irregular emissions of contaminants; and
* workers very close to a contaminant source may still experience
very high levels of exposure.
3.4.2.2 Ventilation
Ventilation systems are one means of removing contaminated air from
the workplace. There are two general types of ventilation:
* dilution or general ventilation; and
* local exhaust ventilation.
Dilution ventilation is simply the process whereby clean air is mixed
with contaminated air. The concentration of the airborne
contaminant is thus reduced, although the workplace air will still
contain some of the contaminant.
A simple dilution ventilation system consists of two major components:
a source of clean air and an exhaust fan for removing the contaminated
air. The ventilation system can be totally passive, which means that
the exhaust is a chimney or open vent in the ceiling from which the
dirty air is expelled, and the source of clean air an open inlet in
one of the workplace walls (see Fig. 36). This is the most basic, i.e.
least effective, system. An improvement on this system would be a fan
at the exhaust forcing the dirty air out. An even better system would
have both an exhaust fan and an inlet fan.
Dilution ventilation is rarely an adequate means of safeguarding
health in the workplace, as:
* it does not prevent the release of a contaminant into the
workplace air;
* it is ineffective against dusts, metal particles and metal fumes
that are too heavy to be removed by gentle air movement;
* it does not provide sufficient protection against highly toxic
chemical vapours and gases, carcinogens or chemicals causing
reproductive effects;
* it is ineffective against "surges" of gases and vapours or
irregular emissions of contaminants; and
* workers very close to a contaminant source may still experience
very high levels of exposure.
 Local exhaust ventilation systems remove airborne contaminants near
their source. Such systems must be properly designed and maintained
and must work effectively so that workers will not inhale contaminated
air. For any work process that uses or produces moderately toxic or
highly toxic chemicals that cannot be effectively enclosed, a local
exhaust system is essential (see Fig. 37).
A local exhaust ventilation system contains four major parts: a hood,
ducts, an air cleaner and a fan. Contaminants are drawn in
through the hood, transported inside the ducts, removed by an air
cleaner and sucked through the system by a fan. A local exhaust
system always needs a suitable inlet of clean replacement air.
Some of the advantages of local exhaust ventilation include:
* the capture of hazardous contaminants at their source, and the
removal of contaminants from the workplace;
* the capacity to handle all types of contaminants, including
dusts, metal particles and metal fumes; and
* the capacity to protect workers close to the contaminant's
source.
To be effective, however, a local exhaust ventilation system must be
carefully designed for the specific process, and the appropriate
parts selected. The hood should be close enough to capture all
contamination and pull it away from, not past, the worker's breathing
zone.
Moreover, local exhaust systems require regular cleaning, inspection
and maintenance (e.g., regular filter changing, checking of the
airflow rate with an anemometer, checking for leaks or holes in the
ducting and checking for blockages). It should be noted that exhaust
air will be contaminated and may endanger the health of people outside
the workplace.
3.4.3 Safe working procedures
Workers involved in the manufacture or use of chemicals are usually
given a list of instructions by their employer on how to weigh,
measure and combine the various chemicals used in their workplace.
Often these instructions tell the worker what to do to make the
product but carry no information on the hazards or risks involved or
on ways to protect against the chemical hazards present, e.g. how to
operate ventilation systems or when to change filters on breathing
masks.
Local exhaust ventilation systems remove airborne contaminants near
their source. Such systems must be properly designed and maintained
and must work effectively so that workers will not inhale contaminated
air. For any work process that uses or produces moderately toxic or
highly toxic chemicals that cannot be effectively enclosed, a local
exhaust system is essential (see Fig. 37).
A local exhaust ventilation system contains four major parts: a hood,
ducts, an air cleaner and a fan. Contaminants are drawn in
through the hood, transported inside the ducts, removed by an air
cleaner and sucked through the system by a fan. A local exhaust
system always needs a suitable inlet of clean replacement air.
Some of the advantages of local exhaust ventilation include:
* the capture of hazardous contaminants at their source, and the
removal of contaminants from the workplace;
* the capacity to handle all types of contaminants, including
dusts, metal particles and metal fumes; and
* the capacity to protect workers close to the contaminant's
source.
To be effective, however, a local exhaust ventilation system must be
carefully designed for the specific process, and the appropriate
parts selected. The hood should be close enough to capture all
contamination and pull it away from, not past, the worker's breathing
zone.
Moreover, local exhaust systems require regular cleaning, inspection
and maintenance (e.g., regular filter changing, checking of the
airflow rate with an anemometer, checking for leaks or holes in the
ducting and checking for blockages). It should be noted that exhaust
air will be contaminated and may endanger the health of people outside
the workplace.
3.4.3 Safe working procedures
Workers involved in the manufacture or use of chemicals are usually
given a list of instructions by their employer on how to weigh,
measure and combine the various chemicals used in their workplace.
Often these instructions tell the worker what to do to make the
product but carry no information on the hazards or risks involved or
on ways to protect against the chemical hazards present, e.g. how to
operate ventilation systems or when to change filters on breathing
masks.
 A safe working procedure should include instructions for each step in
a process, as well as the steps necessary for workers to protect
themselves from the health effects of the chemicals used in that
process.
A "Code of Practice" is a more general description of safety measures
to be adhered to during a particular type of process or by a
particular industry (see Fig. 38). The International Labour
Organisation (ILO) has developed codes of practice for some processes,
and some countries have national codes of practice. For shop-floor
use, a relevant code of practice should include:
* a list of all chemicals used in that process;
* a summary of the health effects of those chemicals and the way in
which exposure to, and absorption of, the chemicals may occur;
* an outline of the equipment necessary to complete a task
correctly and safely;
* the procedures for operating that equipment correctly. For
example, if a chemical has to be heated, information should be
included on the maximum temperature, the consequences of heating
the chemical too much (the risk of explosion or fire) and the
ventilation system required to protect workers and the general
public against fumes or vapours of the heated chemical;
* a description of any personal protective equipment that may be
necessary, such as face masks or gloves, together with the
specifications and standards these must meet and information on
exactly how and when the equipment should be used; and
* details of how often and what environmental and biological
monitoring should be performed and of procedures for action if
"trigger" levels (see below) are exceeded. For example, carbon
monoxide is a highly toxic but colourless and odourless gas
produced by many industrial processes that involve heating or
burning (e.g. blast furnaces and coke ovens). The carbon
monoxide level in the workplace air should be monitored
continuously while the process is in operation. When the carbon
monoxide level reaches a set percentage of the exposure limit
(see section 7.1 of this manual), an alarm should sound and
workers should leave the workplace until the carbon monoxide
level has fallen to a safe level. This "action" or "trigger"
level must be a small fraction of the exposure limit (around 10%)
to allow room for error in measurement and delay in alerting
workers to the hazard.
A safe working procedure should include instructions for each step in
a process, as well as the steps necessary for workers to protect
themselves from the health effects of the chemicals used in that
process.
A "Code of Practice" is a more general description of safety measures
to be adhered to during a particular type of process or by a
particular industry (see Fig. 38). The International Labour
Organisation (ILO) has developed codes of practice for some processes,
and some countries have national codes of practice. For shop-floor
use, a relevant code of practice should include:
* a list of all chemicals used in that process;
* a summary of the health effects of those chemicals and the way in
which exposure to, and absorption of, the chemicals may occur;
* an outline of the equipment necessary to complete a task
correctly and safely;
* the procedures for operating that equipment correctly. For
example, if a chemical has to be heated, information should be
included on the maximum temperature, the consequences of heating
the chemical too much (the risk of explosion or fire) and the
ventilation system required to protect workers and the general
public against fumes or vapours of the heated chemical;
* a description of any personal protective equipment that may be
necessary, such as face masks or gloves, together with the
specifications and standards these must meet and information on
exactly how and when the equipment should be used; and
* details of how often and what environmental and biological
monitoring should be performed and of procedures for action if
"trigger" levels (see below) are exceeded. For example, carbon
monoxide is a highly toxic but colourless and odourless gas
produced by many industrial processes that involve heating or
burning (e.g. blast furnaces and coke ovens). The carbon
monoxide level in the workplace air should be monitored
continuously while the process is in operation. When the carbon
monoxide level reaches a set percentage of the exposure limit
(see section 7.1 of this manual), an alarm should sound and
workers should leave the workplace until the carbon monoxide
level has fallen to a safe level. This "action" or "trigger"
level must be a small fraction of the exposure limit (around 10%)
to allow room for error in measurement and delay in alerting
workers to the hazard.
 Some work procedures require clearance or a "permit-to-work"
certification. The latter is essentially a document that sets out the
work to be done, the hazards involved and the precautions to be taken,
such as testing the working area for chemicals before entering. It
predetermines a safe drill and is a record that all foreseeable
hazards have been considered in advance and precautions taken. It is
of particular importance with respect to maintenance workers, for the
repair of chemical-bearing lines, for work in confined spaces and so
on.
A permit-to-work system does not in itself make a job safe but is
designed to protect the workers involved. Workers should therefore be
well trained in the accompanying procedures.
3.4.4 Reducing the number of exposed workers and their duration
of exposure
Only those directly involved with a job or chemical process should be
exposed to any chemical hazards present. For example, if chemicals
that give off vapours are being heated, then only those workers needed
for the job should be in the area. Maintenance workers, electricians,
cleaners or other workers should do their work when the chemical
hazard is not present.
The risk to maintenance workers is often ignored or seriously
underestimated when planning chemical control measures. Maintenance
workers may be more highly exposed because normal control procedures
are not geared to their work; these procedures may not be operational
at times when they do their work; and they may be in closer proximity
to hazardous plants and processes than other workers when they do
repair or maintenance work. Specific provisions for the protection of
maintenance workers must be included in any chemical safety procedure.
In certain circumstances, the reduction of the duration or the
frequency of exposure of workers may be achieved by job rotation.
However, it is never acceptable to simply expose more workers less
often to unacceptably high levels as an alternative to reducing
exposure levels.
3.4.5 Personal protective equipment
3.4.5.1 Principles
Whereas engineering controls place a barrier around a hazardous
process or chemical, personal protective equipment is often used to
create a "barrier" around a worker to prevent chemicals already
released into the workplace air from reaching the worker's lungs,
skin, etc. The use of personal protective equipment should be
resorted to only after the methods outlined above -- substitution and
engineering controls -- have first been considered and acted upon.
Personal protective equipment is rated as the least effective method
of protection and is often uncomfortable or difficult to work with.
Personal protective equipment against chemicals includes:
* face shields, goggles and safety glasses;
* gloves;
* rubber boots;
* plastic or rubber overalls and aprons;
* hard hats;
* respirators; and
* dust masks.
A personal protective equipment programme requires the following steps
and resources:
* the correct equipment -- e.g. a respirator designed to protect
against dust is useless if the hazardous chemical is present as a
gas; and many solvents can rapidly penetrate natural rubber
gloves;
* a thorough training programme for workers who are required to use
the equipment, with follow-up training at regular intervals;
* tests to ensure that equipment fits correctly. This is
particularly important for face masks and respirators;
* a regular equipment maintenance programme. This includes regular
cleaning of equipment, inspection to ensure that it is operating
correctly and regular replacement of items such as gloves or
disposable parts such as respirator filters. Respirator filters
should be replaced at regular time intervals rather than only
when they have become clogged; and
* a personal set of equipment for each worker and a secure and
clean place in which to store it.
In some situations, the use of personal protective equipment is
unavoidable. This applies particularly to eye goggles, face shields,
boots and hard hats. Because these items are designed to protect the
worker against accidents and unexpected exposures, they must be worn
at all times. For some jobs, such as pesticide spraying by hand, no
other means of protection is possible, in which case protective
clothing, gloves and respirator masks must be worn.
Other tasks that may be necessary from time to time, such as the
cleaning of a ventilation system, will often require the wearing of
personal protective equipment such as respirators. When the chemicals
are particularly dangerous or concentrations very high, air-supplied
respirators are required.
Chapter 4 of each HSG -- and also the ICSC or Summary of Chemical
Safety Information -- provides general advice on types of personal
protective equipment that are suitable for the chemical being used.
Workers should also seek information from the manufacturer of the
chemical about suitable protective equipment. Often this information
is included in the chemical manufacturer's MSDS (see Chapter 8 on
"Getting Further Information").
3.4.5.2 Protective clothing
Not all protective clothing protects its wearer from all chemicals.
Gloves, aprons and overalls, for instance, must be made from a
material that is not penetrable by the chemical in use at the time.
Otherwise, chemical solvents may pass unnoticed through the material,
emerging in an invisible but hazardous form inside gloves or other
protective clothing. If this occurs, the chemical will come into
contact with the skin and could prove as dangerous as would be the
case if the individual were working without any protective clothing.
3.4.5.3 Gloves
General advice about choosing protective gloves includes the
following:
* For maximum protection against skin absorption, gloves should be
of suitable material, fit properly and be in good condition.
* It is important to get information from the suppliers of
protective equipment about what equipment has been tested and
found to be resistant to the particular chemicals you are using.
Ask specifically about the chemical's breakthrough time, i.e. how
long you can wear the gloves before the chemical penetrates
through to your hands. Some gloves, especially PVA (polyvinyl
alcohol) gloves, actually disintegrate on contact with water.
* Only use gloves once when working with chemicals that have a
quick breakthrough time. Remember, the chemical may still be
penetrating the gloves after you have removed them.
* Cotton flock or fabric lining in gloves helps absorb perspiration
and also partly insulates the hands against heat or cold.
* Double gloving is recommended if little is known about the
chemical in use.
* Unless gloves are thoroughly cleaned when they are taken off, any
chemical picked up on the glove lining could come into contact
with the skin.
* Dusts from recently activated resins are almost as irritating and
harmful to the skin as the original liquid; gloves should be worn
even when handling the resultant laminate.
* Some chemicals in combination will penetrate gloves more quickly
than they would separately, e.g. pentane and trichloroethylene.
* Solvents may also carry other substances through the gloves; e.g.
xylene carries organophosphates through PVC (polyvinyl chloride)
gloves.
* Search for a listing of gloves available in your country and
information on their suitability for different jobs. The local
safety inspectorate may have such a list.
3.4.5.4 Do you need a respirator?
There are many different types of respirators available. This section
will help you to ask the right questions to make sure that you are
being provided with the correct type of respirator for your work
process.
* What are you exposed to?
This will depend on the type of hazard, i.e. whether the contaminant
is a gas, vapour, dust, mist or fume; whether there is a potential for
oxygen deficiency (in the USA, if the oxygen level is likely to fall
below 17%, or 19.5% in some countries, fresh air must be provided); or
whether the atmosphere is immediately dangerous to life or health,
e.g. high concentrations of cyanide fumes.
* What concentration of the chemical are you exposed to?
It is important to know how much of the chemical is present in the air
you are breathing, because different types of respirators have
different levels of efficiency. You should know what concentration of
the chemical you are exposed to and the concentration that is
classified as acceptable (see section 7.1 of this Manual on "exposure
limits" for reasons why even these limits may not be safe inside a
respirator).
Once you know these two concentrations of the chemical, you will be
able to work out the amount of protection you need from a respirator.
For example, if the acceptable level of exposure for Solvent B is 10
ppm and the concentration in the workplace is 100 ppm, you will need
an air filter with a "nominal protection factor" of 100/10 = 10.
* Does the chemical have any warning signs?
You will need to know how long you can wear the respirator before you
have to change the filter or cartridge/canister or before the air
supply runs out in a self-contained breathing apparatus. You will
also need to know whether the chemical has a particular smell or is
irritating (either will serve as a warning if the chemical begins to
break through the absorbent material in the respirator). This is
particularly important when air-purifying respirators are used against
gases and vapours.
* Is there any other chemical present that you also need protection
against?
If a mixture of gases or vapours or particulates is present, this will
influence the choice of respirator.
* Can the chemical be absorbed through the skin?
If the answer is "Yes", then the rest of the body, including the face,
must be protected and a full-face respirator worn.
* Can the chemical irritate the eyes at the expected concentration?
If "Yes", then the eyes must be protected -- either separately through
the use of goggles or by use of a full-face respirator.
A SUITABLE RESPIRATOR CAN BE CHOSEN ONLY AFTER THESE QUESTIONS HAVE
BEEN ANSWERED.
3.4.5.5 Types of respirators
I Air-purifying respirators -- The air is drawn in through an
absorbent material in the case of gaseous contaminants or through a
filter in the case of particulate contaminants.
a) Air-purifying respirators for gaseous contaminants
These consist of a face-piece with a cartridge or canister to remove
the gas or vapour. The face-piece can be quarter-mask, covering the
nose and mouth but not fitting under the chin; half-mask, covering the
nose and mouth and fitting under the chin; or full-face mask, covering
the face from the hairline to under the chin. The full-face mask
offers greater protection than half- or quarter-masks, as the mask
will fit the face more closely, giving a better seal.
The cartridge or canister containing the material that removes the gas
or vapour is fitted to the front of the face-piece and air drawn
through it (see Fig. 39). A cartridge offers less protection than a
canister. A cartridge can be used to protect against gases or vapours
of relatively low toxicity. A canister will remove limited
concentrations of certain toxic gases or vapours for a specified time,
after which the canister must be replaced.
Some work procedures require clearance or a "permit-to-work"
certification. The latter is essentially a document that sets out the
work to be done, the hazards involved and the precautions to be taken,
such as testing the working area for chemicals before entering. It
predetermines a safe drill and is a record that all foreseeable
hazards have been considered in advance and precautions taken. It is
of particular importance with respect to maintenance workers, for the
repair of chemical-bearing lines, for work in confined spaces and so
on.
A permit-to-work system does not in itself make a job safe but is
designed to protect the workers involved. Workers should therefore be
well trained in the accompanying procedures.
3.4.4 Reducing the number of exposed workers and their duration
of exposure
Only those directly involved with a job or chemical process should be
exposed to any chemical hazards present. For example, if chemicals
that give off vapours are being heated, then only those workers needed
for the job should be in the area. Maintenance workers, electricians,
cleaners or other workers should do their work when the chemical
hazard is not present.
The risk to maintenance workers is often ignored or seriously
underestimated when planning chemical control measures. Maintenance
workers may be more highly exposed because normal control procedures
are not geared to their work; these procedures may not be operational
at times when they do their work; and they may be in closer proximity
to hazardous plants and processes than other workers when they do
repair or maintenance work. Specific provisions for the protection of
maintenance workers must be included in any chemical safety procedure.
In certain circumstances, the reduction of the duration or the
frequency of exposure of workers may be achieved by job rotation.
However, it is never acceptable to simply expose more workers less
often to unacceptably high levels as an alternative to reducing
exposure levels.
3.4.5 Personal protective equipment
3.4.5.1 Principles
Whereas engineering controls place a barrier around a hazardous
process or chemical, personal protective equipment is often used to
create a "barrier" around a worker to prevent chemicals already
released into the workplace air from reaching the worker's lungs,
skin, etc. The use of personal protective equipment should be
resorted to only after the methods outlined above -- substitution and
engineering controls -- have first been considered and acted upon.
Personal protective equipment is rated as the least effective method
of protection and is often uncomfortable or difficult to work with.
Personal protective equipment against chemicals includes:
* face shields, goggles and safety glasses;
* gloves;
* rubber boots;
* plastic or rubber overalls and aprons;
* hard hats;
* respirators; and
* dust masks.
A personal protective equipment programme requires the following steps
and resources:
* the correct equipment -- e.g. a respirator designed to protect
against dust is useless if the hazardous chemical is present as a
gas; and many solvents can rapidly penetrate natural rubber
gloves;
* a thorough training programme for workers who are required to use
the equipment, with follow-up training at regular intervals;
* tests to ensure that equipment fits correctly. This is
particularly important for face masks and respirators;
* a regular equipment maintenance programme. This includes regular
cleaning of equipment, inspection to ensure that it is operating
correctly and regular replacement of items such as gloves or
disposable parts such as respirator filters. Respirator filters
should be replaced at regular time intervals rather than only
when they have become clogged; and
* a personal set of equipment for each worker and a secure and
clean place in which to store it.
In some situations, the use of personal protective equipment is
unavoidable. This applies particularly to eye goggles, face shields,
boots and hard hats. Because these items are designed to protect the
worker against accidents and unexpected exposures, they must be worn
at all times. For some jobs, such as pesticide spraying by hand, no
other means of protection is possible, in which case protective
clothing, gloves and respirator masks must be worn.
Other tasks that may be necessary from time to time, such as the
cleaning of a ventilation system, will often require the wearing of
personal protective equipment such as respirators. When the chemicals
are particularly dangerous or concentrations very high, air-supplied
respirators are required.
Chapter 4 of each HSG -- and also the ICSC or Summary of Chemical
Safety Information -- provides general advice on types of personal
protective equipment that are suitable for the chemical being used.
Workers should also seek information from the manufacturer of the
chemical about suitable protective equipment. Often this information
is included in the chemical manufacturer's MSDS (see Chapter 8 on
"Getting Further Information").
3.4.5.2 Protective clothing
Not all protective clothing protects its wearer from all chemicals.
Gloves, aprons and overalls, for instance, must be made from a
material that is not penetrable by the chemical in use at the time.
Otherwise, chemical solvents may pass unnoticed through the material,
emerging in an invisible but hazardous form inside gloves or other
protective clothing. If this occurs, the chemical will come into
contact with the skin and could prove as dangerous as would be the
case if the individual were working without any protective clothing.
3.4.5.3 Gloves
General advice about choosing protective gloves includes the
following:
* For maximum protection against skin absorption, gloves should be
of suitable material, fit properly and be in good condition.
* It is important to get information from the suppliers of
protective equipment about what equipment has been tested and
found to be resistant to the particular chemicals you are using.
Ask specifically about the chemical's breakthrough time, i.e. how
long you can wear the gloves before the chemical penetrates
through to your hands. Some gloves, especially PVA (polyvinyl
alcohol) gloves, actually disintegrate on contact with water.
* Only use gloves once when working with chemicals that have a
quick breakthrough time. Remember, the chemical may still be
penetrating the gloves after you have removed them.
* Cotton flock or fabric lining in gloves helps absorb perspiration
and also partly insulates the hands against heat or cold.
* Double gloving is recommended if little is known about the
chemical in use.
* Unless gloves are thoroughly cleaned when they are taken off, any
chemical picked up on the glove lining could come into contact
with the skin.
* Dusts from recently activated resins are almost as irritating and
harmful to the skin as the original liquid; gloves should be worn
even when handling the resultant laminate.
* Some chemicals in combination will penetrate gloves more quickly
than they would separately, e.g. pentane and trichloroethylene.
* Solvents may also carry other substances through the gloves; e.g.
xylene carries organophosphates through PVC (polyvinyl chloride)
gloves.
* Search for a listing of gloves available in your country and
information on their suitability for different jobs. The local
safety inspectorate may have such a list.
3.4.5.4 Do you need a respirator?
There are many different types of respirators available. This section
will help you to ask the right questions to make sure that you are
being provided with the correct type of respirator for your work
process.
* What are you exposed to?
This will depend on the type of hazard, i.e. whether the contaminant
is a gas, vapour, dust, mist or fume; whether there is a potential for
oxygen deficiency (in the USA, if the oxygen level is likely to fall
below 17%, or 19.5% in some countries, fresh air must be provided); or
whether the atmosphere is immediately dangerous to life or health,
e.g. high concentrations of cyanide fumes.
* What concentration of the chemical are you exposed to?
It is important to know how much of the chemical is present in the air
you are breathing, because different types of respirators have
different levels of efficiency. You should know what concentration of
the chemical you are exposed to and the concentration that is
classified as acceptable (see section 7.1 of this Manual on "exposure
limits" for reasons why even these limits may not be safe inside a
respirator).
Once you know these two concentrations of the chemical, you will be
able to work out the amount of protection you need from a respirator.
For example, if the acceptable level of exposure for Solvent B is 10
ppm and the concentration in the workplace is 100 ppm, you will need
an air filter with a "nominal protection factor" of 100/10 = 10.
* Does the chemical have any warning signs?
You will need to know how long you can wear the respirator before you
have to change the filter or cartridge/canister or before the air
supply runs out in a self-contained breathing apparatus. You will
also need to know whether the chemical has a particular smell or is
irritating (either will serve as a warning if the chemical begins to
break through the absorbent material in the respirator). This is
particularly important when air-purifying respirators are used against
gases and vapours.
* Is there any other chemical present that you also need protection
against?
If a mixture of gases or vapours or particulates is present, this will
influence the choice of respirator.
* Can the chemical be absorbed through the skin?
If the answer is "Yes", then the rest of the body, including the face,
must be protected and a full-face respirator worn.
* Can the chemical irritate the eyes at the expected concentration?
If "Yes", then the eyes must be protected -- either separately through
the use of goggles or by use of a full-face respirator.
A SUITABLE RESPIRATOR CAN BE CHOSEN ONLY AFTER THESE QUESTIONS HAVE
BEEN ANSWERED.
3.4.5.5 Types of respirators
I Air-purifying respirators -- The air is drawn in through an
absorbent material in the case of gaseous contaminants or through a
filter in the case of particulate contaminants.
a) Air-purifying respirators for gaseous contaminants
These consist of a face-piece with a cartridge or canister to remove
the gas or vapour. The face-piece can be quarter-mask, covering the
nose and mouth but not fitting under the chin; half-mask, covering the
nose and mouth and fitting under the chin; or full-face mask, covering
the face from the hairline to under the chin. The full-face mask
offers greater protection than half- or quarter-masks, as the mask
will fit the face more closely, giving a better seal.
The cartridge or canister containing the material that removes the gas
or vapour is fitted to the front of the face-piece and air drawn
through it (see Fig. 39). A cartridge offers less protection than a
canister. A cartridge can be used to protect against gases or vapours
of relatively low toxicity. A canister will remove limited
concentrations of certain toxic gases or vapours for a specified time,
after which the canister must be replaced.
 b) Air-purifying respirators for particulate contaminants
These consist of a face-piece and a filter. The face-piece can be a
quarter-, half- or full-face mask. The filters are classified as:
* low efficiency -- used against dusts and mists of low toxicity,
in low concentrations;
* medium efficiency -- used against dust and fume concentrations up
to 10 times the exposure standard, except where the dust or fume
is highly toxic, e.g. hexavalent chromium; and
* high efficiency -- used, with a full face-piece respirator, where
toxicity or concentration of a chemical is high.
A combined gas and dust respirator should be used where both gases/
vapours and dusts are present.
c) Air-purifying respirators for both gaseous and particulate
contaminants
These respirators can be used where a combined hazard is present, e.g.
paint spraying or spraying with pesticides. Some particulate filters
will also absorb some gases/vapours, but you must insist that evidence
is provided that they will protect against both. The filters should
be marked to show what they protect against.
The length of time a cartridge or canister can be used depends on the
concentration of gas, vapour or particulate and the activity of the
wearer. If you are doing hard physical work, you will breathe more
deeply and the canister will become saturated sooner.
Air-purifying respirators should not be used in a confined space, or
there might be a shortage of oxygen.
Face masks that do not fit the worker or that are worn out or blocked
by particulates can result in serious injury, as the wearer believes
him/herself protected and is therefore unaware that he or she is
exposed. The presence of facial hair can also mean that the face mask
does not fit perfectly, with the result that contaminated air could
enter between the mask and the skin.
II Supplied air units -- These respirators supply clean air directly
into the respirator, rather than filtering out the chemical from the
air in the work environment (see Fig. 40). There are two types:
a) Supplied air respirator -- Air is supplied through an air supply
line or hose. In the case of an air supply line, the air comes
from a compressed air source through a high-pressure hose. The air
can be supplied to a half- or full-face mask, helmet, hood or complete
suit. These respirators can be used against particulates, gases or
vapours. They provide a high degree of protection but cannot be used
in atmospheres immediately hazardous to life or health, as any fault
with the air supply or air line could render the worker highly
vulnerable.
For hose masks, air is supplied from a clean-air area through a
large-diameter hose to the face-piece. Compressed air is not used.
The air can be either pumped in by a hand- or motor-operated blower or
simply drawn in by the wearer. These respirators always have a
full-face mask, which must fit the face well. They should also not be
used in atmospheres immediately dangerous to life or health.
b) Air-purifying respirators for particulate contaminants
These consist of a face-piece and a filter. The face-piece can be a
quarter-, half- or full-face mask. The filters are classified as:
* low efficiency -- used against dusts and mists of low toxicity,
in low concentrations;
* medium efficiency -- used against dust and fume concentrations up
to 10 times the exposure standard, except where the dust or fume
is highly toxic, e.g. hexavalent chromium; and
* high efficiency -- used, with a full face-piece respirator, where
toxicity or concentration of a chemical is high.
A combined gas and dust respirator should be used where both gases/
vapours and dusts are present.
c) Air-purifying respirators for both gaseous and particulate
contaminants
These respirators can be used where a combined hazard is present, e.g.
paint spraying or spraying with pesticides. Some particulate filters
will also absorb some gases/vapours, but you must insist that evidence
is provided that they will protect against both. The filters should
be marked to show what they protect against.
The length of time a cartridge or canister can be used depends on the
concentration of gas, vapour or particulate and the activity of the
wearer. If you are doing hard physical work, you will breathe more
deeply and the canister will become saturated sooner.
Air-purifying respirators should not be used in a confined space, or
there might be a shortage of oxygen.
Face masks that do not fit the worker or that are worn out or blocked
by particulates can result in serious injury, as the wearer believes
him/herself protected and is therefore unaware that he or she is
exposed. The presence of facial hair can also mean that the face mask
does not fit perfectly, with the result that contaminated air could
enter between the mask and the skin.
II Supplied air units -- These respirators supply clean air directly
into the respirator, rather than filtering out the chemical from the
air in the work environment (see Fig. 40). There are two types:
a) Supplied air respirator -- Air is supplied through an air supply
line or hose. In the case of an air supply line, the air comes
from a compressed air source through a high-pressure hose. The air
can be supplied to a half- or full-face mask, helmet, hood or complete
suit. These respirators can be used against particulates, gases or
vapours. They provide a high degree of protection but cannot be used
in atmospheres immediately hazardous to life or health, as any fault
with the air supply or air line could render the worker highly
vulnerable.
For hose masks, air is supplied from a clean-air area through a
large-diameter hose to the face-piece. Compressed air is not used.
The air can be either pumped in by a hand- or motor-operated blower or
simply drawn in by the wearer. These respirators always have a
full-face mask, which must fit the face well. They should also not be
used in atmospheres immediately dangerous to life or health.
 b) Self-contained breathing apparatus -- This allows the wearer to
carry a respirable breathing supply, which may last from a few minutes
to four hours, depending on type. With some, a negative pressure is
created inside the face-piece when the wearer breathes in, so the air
is drawn into the face-piece only when the wearer breathes in, instead
of being supplied to the face-piece before the wearer breathes in. If
the face seal is not good and a negative pressure is created inside
the face-piece, contaminated air may be sucked into the respirator
through the gaps between the face and the face-piece. These
negative-pressure types of breathing apparatus should not be used in
atmospheres immediately hazardous to life or health.
3.4.5.6 Training and fitting
Workers must be trained as to why respirators must be worn and how to
choose, fit and use them before they are expected to use them.
Respirators are uncomfortable to wear, and workers will often not use
them if they do not fully understand the health risks they face.
3.4.5.7 Respiratory protection programme
If respirators are to be used correctly and effectively, a respiratory
protection programme administered by a trained person should be
established. The programme should include written standard operating
procedures and specifications for:
* respirators (selected on the basis of hazards);
* instruction and training of the user;
* cleaning and disinfection;
* storage;
* inspection;
* surveillance of work area conditions;
* evaluation of the respiratory protection programme;
* use of certified respirators; and
* medical review.
3.5 Monitoring the hazard
Monitoring should be performed to check the efficiency of control
methods in the workplace. There are two types of monitoring --
environmental monitoring to measure workers' exposures to air
contaminants, and biological monitoring of individual workers to
test for degree of exposure, whether dermal, respiratory, via
ingestion, etc. The results can be compared with safety standards for
workplace air or biological materials.
3.5.1 Environmental monitoring
Monitoring of the workplace environment for contaminants may be
undertaken in two ways: through integrated sampling or, for some
substances, use of direct reading instruments. Direct reading
instruments give an immediate result, but for many substances such
devices are not available. In addition, they must be carefully
calibrated. Integrated sampling, on the other hand, can be used for a
wider range of substances, although the result is available only
following laboratory analysis. Integrated sampling involves measuring
the concentrations or levels of dusts, mists or gases in the workplace
air, by sampling known amounts of air containing the dust, mist or gas
with special pumps and filters and then chemically analysing the
results. This monitoring may be technically complicated and must
usually be carried out by an industrial hygienist; in some cases, it
is less complex and can be done by trained workers.
There are two main types of integrated sampling:
* Static monitoring -- in which an air pump is set up at a fixed
point. This is useful for chemicals that are expected to spread
evenly throughout the entire workplace. Static monitoring may
over- or underestimate the exposure individual workers are
receiving, depending on where measurements are made.
* Personal sampling -- in which air is sampled from individual
workers' breathing zones by a portable pump attached to the
workers' clothing. This method gives a much better indication of
an individual worker's exposure to a chemical and, of the two air
sampling techniques, is the method of choice. The advantage over
static or fixed-point monitoring is that a reading for exposure
is obtained that better reflects contamination in the worker's
breathing zone. With static or fixed-point monitoring,
concentrations in the worker's breathing zone will not be
measured if the measurement is taken at too great a distance from
the source.
The following questions should be considered in any environmental
monitoring programme:
* Are the samples being taken at the right place, i.e. near the
breathing zones of operators?
* Is the sample being taken at the time when pollution levels are
highest? The aim of environmental monitoring should always be to
determine the highest level of exposure, not the average or usual
level of exposure.
* Are all contaminants being measured?
* Have workers been involved in the planning of monitoring of their
workplace?
* Are the test results being made available to safety
representatives and safety committees regularly, and are the
results being discussed?
Environmental monitoring should be performed before decisions are made
regarding methods for controlling chemical exposure. In this way,
control methods can be designed that cope with the highest levels of
chemicals measured, not the average levels. In other words, control
measures must be able to cope with the "worst case".
After methods to control exposure (e.g. exhaust ventilation) have been
introduced, environmental monitoring should be repeated regularly to
ensure that the control methods are operating satisfactorily.
3.5.2 Biological monitoring
Biological monitoring of chemical exposure aims to measure either the
amount of a chemical that has been absorbed by a worker or the effect
of that absorbed chemical on the worker (as opposed to environmental
monitoring, which measures the level of exposure).
Biological monitoring involves taking a sample of body fluid, usually
blood or urine, and measuring the level of the chemical or its
metabolite (see Chapter 2). Alternatively, an effect of that chemical
on the body may be determined by measuring the level of an enzyme or
other chemical in the blood or urine.
A good example is biological monitoring for lead absorption. All
workers regularly exposed to lead or to significant levels of lead
should have blood samples taken regularly. The level of lead in the
blood can then be measured directly, and this will give an accurate
estimate of how much lead that worker has recently absorbed. One of
the effects of lead on the body is the poisoning of red blood cells.
By measuring the level of protoporphyrin -- a chemical in the blood
produced by poisoned red blood cells -- an estimate of the effects of
lead absorption can be obtained (see Fig. 41).
Many chemicals can be monitored biologically, but the results do not
always reflect the level of absorption. Each chemical should be
considered separately when deciding whether to perform biological
monitoring. Lists of those chemicals for which biological monitoring
is recommended are available, and some such chemicals may have a value
-- i.e. a biological exposure index -- that should not be exceeded.
Figure 41. Biological monitoring (from EHC 3: Lead)
Pb levels at which no more than 5% of the population will
show the indicated intensity of effect
Biochemical PbB
effect Intensity of effect Population (µg/100 ml)
ALAD inhibition perceptible inhibition adult, children 10
in blood >40% inhibition adults 15-20
>70% inhibition adults 30
>70% inhibition children 25-30
ALA in urine perceptible increase adults, children 40
>10 mg/litre adults, children 50
FEP in blood perceptible increase adult males 30
adult females 25
children 20
ALA = delta-aminolevulinic acid
ALAD = delta-aminolevulinic acid dehydratase
FEP = free erythrocyte porphyrins
PbB = blood lead level
4. PROTECTION AND EMERGENCY ACTION - CHAPTER 4
This chapter should be read in conjunction with chapter 4 of the
relevant HSG, which outlines the main hazards of the chemical
concerned (including a description of the fire and explosion risks),
provides medical advice in case of exposure and outlines storage,
transport and disposal considerations.
Good safety organization, ventilation and engineering controls,
adequate provision of information on the health hazards of chemicals
and training of workers can reduce and control chemical exposures in a
workplace.
Nevertheless, poisonings may still occur, and it is important that
training and proper equipment are available to workers so that
emergency situations can be handled satisfactorily (see Fig. 42).
4.1 First aid and emergencies
The following general principles should be employed when designing a
first-aid programme for a workplace where chemicals are used:
* There should be at least one worker present on every shift who
has received approved first-aid training. Follow-up or refresher
courses should be offered regularly.
* All workers should receive training in the technique of cardio-
pulmonary resuscitation (CPR). CPR is used when a person's heart
has stopped beating or when he or she has stopped breathing.
* It is essential that any workers affected or injured by chemicals
are removed from the area where the chemicals are present.
Rescuers should make sure of their own safety in any attempt at
rescue and/or resuscitation. Self-contained breathing apparatus
(a respirator mask with its own air supply) may be necessary so
that those attempting to rescue an injured or affected worker do
not become poisoned themselves.
* Emergency showers and eye-baths should be situated close to the
site of any potentially hazardous work processes.
b) Self-contained breathing apparatus -- This allows the wearer to
carry a respirable breathing supply, which may last from a few minutes
to four hours, depending on type. With some, a negative pressure is
created inside the face-piece when the wearer breathes in, so the air
is drawn into the face-piece only when the wearer breathes in, instead
of being supplied to the face-piece before the wearer breathes in. If
the face seal is not good and a negative pressure is created inside
the face-piece, contaminated air may be sucked into the respirator
through the gaps between the face and the face-piece. These
negative-pressure types of breathing apparatus should not be used in
atmospheres immediately hazardous to life or health.
3.4.5.6 Training and fitting
Workers must be trained as to why respirators must be worn and how to
choose, fit and use them before they are expected to use them.
Respirators are uncomfortable to wear, and workers will often not use
them if they do not fully understand the health risks they face.
3.4.5.7 Respiratory protection programme
If respirators are to be used correctly and effectively, a respiratory
protection programme administered by a trained person should be
established. The programme should include written standard operating
procedures and specifications for:
* respirators (selected on the basis of hazards);
* instruction and training of the user;
* cleaning and disinfection;
* storage;
* inspection;
* surveillance of work area conditions;
* evaluation of the respiratory protection programme;
* use of certified respirators; and
* medical review.
3.5 Monitoring the hazard
Monitoring should be performed to check the efficiency of control
methods in the workplace. There are two types of monitoring --
environmental monitoring to measure workers' exposures to air
contaminants, and biological monitoring of individual workers to
test for degree of exposure, whether dermal, respiratory, via
ingestion, etc. The results can be compared with safety standards for
workplace air or biological materials.
3.5.1 Environmental monitoring
Monitoring of the workplace environment for contaminants may be
undertaken in two ways: through integrated sampling or, for some
substances, use of direct reading instruments. Direct reading
instruments give an immediate result, but for many substances such
devices are not available. In addition, they must be carefully
calibrated. Integrated sampling, on the other hand, can be used for a
wider range of substances, although the result is available only
following laboratory analysis. Integrated sampling involves measuring
the concentrations or levels of dusts, mists or gases in the workplace
air, by sampling known amounts of air containing the dust, mist or gas
with special pumps and filters and then chemically analysing the
results. This monitoring may be technically complicated and must
usually be carried out by an industrial hygienist; in some cases, it
is less complex and can be done by trained workers.
There are two main types of integrated sampling:
* Static monitoring -- in which an air pump is set up at a fixed
point. This is useful for chemicals that are expected to spread
evenly throughout the entire workplace. Static monitoring may
over- or underestimate the exposure individual workers are
receiving, depending on where measurements are made.
* Personal sampling -- in which air is sampled from individual
workers' breathing zones by a portable pump attached to the
workers' clothing. This method gives a much better indication of
an individual worker's exposure to a chemical and, of the two air
sampling techniques, is the method of choice. The advantage over
static or fixed-point monitoring is that a reading for exposure
is obtained that better reflects contamination in the worker's
breathing zone. With static or fixed-point monitoring,
concentrations in the worker's breathing zone will not be
measured if the measurement is taken at too great a distance from
the source.
The following questions should be considered in any environmental
monitoring programme:
* Are the samples being taken at the right place, i.e. near the
breathing zones of operators?
* Is the sample being taken at the time when pollution levels are
highest? The aim of environmental monitoring should always be to
determine the highest level of exposure, not the average or usual
level of exposure.
* Are all contaminants being measured?
* Have workers been involved in the planning of monitoring of their
workplace?
* Are the test results being made available to safety
representatives and safety committees regularly, and are the
results being discussed?
Environmental monitoring should be performed before decisions are made
regarding methods for controlling chemical exposure. In this way,
control methods can be designed that cope with the highest levels of
chemicals measured, not the average levels. In other words, control
measures must be able to cope with the "worst case".
After methods to control exposure (e.g. exhaust ventilation) have been
introduced, environmental monitoring should be repeated regularly to
ensure that the control methods are operating satisfactorily.
3.5.2 Biological monitoring
Biological monitoring of chemical exposure aims to measure either the
amount of a chemical that has been absorbed by a worker or the effect
of that absorbed chemical on the worker (as opposed to environmental
monitoring, which measures the level of exposure).
Biological monitoring involves taking a sample of body fluid, usually
blood or urine, and measuring the level of the chemical or its
metabolite (see Chapter 2). Alternatively, an effect of that chemical
on the body may be determined by measuring the level of an enzyme or
other chemical in the blood or urine.
A good example is biological monitoring for lead absorption. All
workers regularly exposed to lead or to significant levels of lead
should have blood samples taken regularly. The level of lead in the
blood can then be measured directly, and this will give an accurate
estimate of how much lead that worker has recently absorbed. One of
the effects of lead on the body is the poisoning of red blood cells.
By measuring the level of protoporphyrin -- a chemical in the blood
produced by poisoned red blood cells -- an estimate of the effects of
lead absorption can be obtained (see Fig. 41).
Many chemicals can be monitored biologically, but the results do not
always reflect the level of absorption. Each chemical should be
considered separately when deciding whether to perform biological
monitoring. Lists of those chemicals for which biological monitoring
is recommended are available, and some such chemicals may have a value
-- i.e. a biological exposure index -- that should not be exceeded.
Figure 41. Biological monitoring (from EHC 3: Lead)
Pb levels at which no more than 5% of the population will
show the indicated intensity of effect
Biochemical PbB
effect Intensity of effect Population (µg/100 ml)
ALAD inhibition perceptible inhibition adult, children 10
in blood >40% inhibition adults 15-20
>70% inhibition adults 30
>70% inhibition children 25-30
ALA in urine perceptible increase adults, children 40
>10 mg/litre adults, children 50
FEP in blood perceptible increase adult males 30
adult females 25
children 20
ALA = delta-aminolevulinic acid
ALAD = delta-aminolevulinic acid dehydratase
FEP = free erythrocyte porphyrins
PbB = blood lead level
4. PROTECTION AND EMERGENCY ACTION - CHAPTER 4
This chapter should be read in conjunction with chapter 4 of the
relevant HSG, which outlines the main hazards of the chemical
concerned (including a description of the fire and explosion risks),
provides medical advice in case of exposure and outlines storage,
transport and disposal considerations.
Good safety organization, ventilation and engineering controls,
adequate provision of information on the health hazards of chemicals
and training of workers can reduce and control chemical exposures in a
workplace.
Nevertheless, poisonings may still occur, and it is important that
training and proper equipment are available to workers so that
emergency situations can be handled satisfactorily (see Fig. 42).
4.1 First aid and emergencies
The following general principles should be employed when designing a
first-aid programme for a workplace where chemicals are used:
* There should be at least one worker present on every shift who
has received approved first-aid training. Follow-up or refresher
courses should be offered regularly.
* All workers should receive training in the technique of cardio-
pulmonary resuscitation (CPR). CPR is used when a person's heart
has stopped beating or when he or she has stopped breathing.
* It is essential that any workers affected or injured by chemicals
are removed from the area where the chemicals are present.
Rescuers should make sure of their own safety in any attempt at
rescue and/or resuscitation. Self-contained breathing apparatus
(a respirator mask with its own air supply) may be necessary so
that those attempting to rescue an injured or affected worker do
not become poisoned themselves.
* Emergency showers and eye-baths should be situated close to the
site of any potentially hazardous work processes.
 * First-aid boxes should be provided in all work areas and workers
trained in emergency first-aid procedures for any accident or
chemical exposure that may occur.
* Telephone numbers for medical assistance and ambulances should be
prominently displayed in the workplace. A telephone must always
be available for use in an emergency.
* There should be an "emergency response plan" in which individuals
are assigned to perform certain tasks (such as summon help) while
all other personnel quickly leave the workplace. The plan should
be written down and prominently displayed in the workplace and
should be practised regularly.
* A system should operate whereby all accidents and injuries, no
matter how slight, are reported in writing to the management or
employer. These accident records should be made available to
workers and their representatives and analysed to determine how
to prevent the same, or similar, accidents from occurring again.
In many countries, occupational accidents and injuries must be
reported to government bodies.
* If an injured worker is sent to hospital or to see a doctor or
nurse, it is essential that information on the chemicals involved
in the accident be sent with the worker. The manufacturer's MSDS
and the ICSC or Summary of Chemical Safety Information contained
in HSGs (see Chapter 3) are usually suitable for this purpose.
4.2 Health surveillance
Health surveillance or medical monitoring consists of periodic
examination of a worker by a doctor, nurse or other health worker in
order to detect any health effects of a chemical, to monitor the
progression or reversal of symptoms or to see if treatment or
preventive measures are effective. Tests of lung, heart, liver and
kidney functions, for instance, may be performed. Although in some
cases -- e.g. chemicals that cause asthma -- such testing is useful,
it should always be remembered that medical monitoring can detect
damage to health only after it has occurred (see Fig. 43). Further
damage can then be prevented, but medical monitoring does not actually
prevent the occurrence of the initial health effects.
Figure 43. Health surveillance (from HSG 6: Methylene Chloride)
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically. Administer
oxygen if needed. Do not administer fats, oils, alcohol, epinephrine,
or ephedrine. Pay attention to signs of anoxaemia, impending
pulmonary oedema, and ischaemia of the heart. Monitor ECG. Treat
skin as for a thermal burn.
4.3 Health Surveillance Advice
Persons handling methylene chloride should frequently undergo
periodic medical examination with emphasis on the functioning
of the central nervous system and irritation of the skin and
eyes. Monitoring of blood-carboxyhaemoglobin concentrations can
give an indication of the extent of exposure.
Medical monitoring is often unable to detect the long-term or chronic
health effects of chemicals until they are quite advanced. This
applies particularly to:
* cancer caused by carcinogenic chemicals;
* effects on the brain and nervous system; and
* reproductive effects.
Medical monitoring is useful for assessing the effectiveness of
measures implemented to control chemical exposure, but it is not a
substitute for environmental monitoring and control methods.
4.3 Fire and explosion hazards
Many chemicals used in the workplace will burn or explode easily.
Accidental fires or explosions of flammable or explosive chemicals
represent major hazards. Special precautions must be taken when
handling, storing or transporting these chemicals to prevent fires or
explosions (see Fig. 44).
Figure 44. Explosion and fire hazards
(from HSG 6: Methylene Chloride)
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
Methylene chloride may react explosively with light metals (lithium,
sodium, potassium, aluminium, magnesium) as well as with alloys of
sodium. Explosive mixtures are formed with dinitrogen tetroxide
(N2O4) dinitrogen pentoxide (N2O5), nitric acid, and
compressed oxygen.
4.3.2 Fire hazards
Methylene chloride is flammable in an atmosphere with an increased
oxygen content. In pure oxygen, the flammable limits (explosive
limits) are 15.5% and 66.4%. Methylene chloride is also flammable
if small amounts of combustible material are added. The compound
decomposes in contact with open flames and glowing surfaces with the
formation of harmful gases, such as hydrogen chloride, which forms
mists of hydrochloric acid with moisture, and phosgene.
4.3.3 Prevention
Do not use methylene chloride in the vicinity of a fire, a hot
surface (e.g. a portable heating unit), or during welding. Do
not smoke. Do not add any combustible material to methylene
chloride. Avoid using compressed oxygen when handling
methylene chloride. Avoid contact between the compound and
light metals, alloys of light metals, dinitrogen tetroxide,
dinitrogen pentoxide, nitric acid, or liquid oxygen. In case
of fire, keep drums cool by spraying with water.
Fire (or combustion) is the violent combination of oxygen in the air
with a combustible chemical while producing heat (an exothermic
reaction).
In fires involving chemicals or chemical products, the chemicals
usually become broken down into combustion by-products. Many of
these combustion by-products are highly toxic gases and fumes. This
may be the case even if the original chemical was not particularly
toxic. For example, the gas Freon 12, or dichlorodifluoromethane, is
widely used as a refrigerant. It is toxic only at very high
concentrations. However, the thermal decomposition products of Freon
12 are very toxic gases, including phosgene, which causes potentially
fatal pulmonary oedema (accumulation of water in the lungs), even at
very low levels of exposure. Similarly, polyurethane foam, which is
commonly used as insulation in industry, has relatively low toxicity
in the cured state. However, when polyurethane foam burns, large
amounts of highly toxic gases, including cyanide and hydrogen
chloride, are released.
4.3.1 Flammable substances
A flammable substance is any chemical or material that will continue
to burn after having been ignited. A substance of high flammability
is one that will easily ignite at room temperature on contact with
low-energy ignition sources such as a spark. A substance of low
flammability will ignite only after heating or prolonged contact with
a high-energy ignition source. The flammability of a substance is
often ranked according to its ability to catch fire at certain
temperatures:
temperature
extremely flammable < 0°C
highly flammable 0-21°C
flammable 21-55°C
combustible > 55°C
Other important information about the flammability of a substance can
be gathered by looking at the flash point and the flammability
(explosive) limits (see Chapter 1) for that substance. This
information is given in the Physical Properties section of the ICSC
and usually appears on the MSDS supplied by the chemical manufacturer.
4.3.2 Dust explosions
Any solid material that is capable of burning in a finely divided form
can give rise to a dust explosion hazard. All that is necessary is
for enough dust to be in the air and for a source of ignition to be
present. Flame from the source of ignition will spread in all
directions through the cloud of dust, burning the dust and causing
expansion, thus increasing pressure on the surroundings. In a grain
store, for example, this pressure could be great enough to cause the
roof or walls to be blown off. The first explosion can whip dust that
is lying on window sills, pipework and equipment into the air, and a
second explosion can result.
Many dusts, including natural materials such as starch, sugar, flour,
coal and wood, plastics and organic chemicals, and light metals such
as aluminium, magnesium and titanium, can explode in this way.
Accordingly, dust explosion must be guarded against in agriculture,
chemical, metallurgical and process industries, as well as coal
mining.
4.3.3 Sources of ignition
Flammable substances or dust clouds may be ignited in one of the
following ways:
* Direct flames -- matches, cigarettes, soldering, welding or
cutting torches.
* Heat radiation -- all sources of heat emit heat (thermal)
radiation, which may ignite a flammable material at a distance.
* Electrical sparks -- produced by electric current in a number of
ways, including during electric arc welding; by power supplies
such as sockets and switches and sliding contacts; from electric
motors in power tools or the ignition systems of internal
combustion engines; by lightning; and as a result of static
electricity build-up between two non-conducting materials such as
belts on a pulley, rubber tyres on the ground or liquids pumped
or poured through pipes.
* Mechanically produced sparks -- two hard materials rubbing
against each other at high speed may produce sparks capable of
igniting flammable chemicals, e.g. when grinding.
* Chemical reactions -- many chemical reactions produce heat. The
design of the chemical process should take this into account and
make provision for the removal of excess heat. However, excess
heat may develop if the incorrect or wrong proportions of
chemicals are used. This may start a fire or trigger an
explosion.
* Spontaneous combustion -- some substances oxidize (combine with
oxygen) as soon as they are exposed to air. Usually this occurs
slowly, but with some chemicals it may happen rapidly and produce
considerable heat in the process. These chemicals must be stored
in tanks or containers that allow all air to be removed and
replaced with a gas that will not combine with the chemical
(usually nitrogen). If this mechanism fails and air enters the
container, a fire or explosion may result.
Safety information about fire hazards is given in chapter 4 of the
HSGs and is also contained in the ICSCs. Information on fire and
explosion hazards should always be included in chemical manufacturers'
MSDS.
4.3.4 Fire-fighting
Equipment used to extinguish and control fires is of two types: fixed
and portable. Fixed systems include water equipment, automatic
sprinklers, hydrants and special pipe systems for dry chemicals,
carbon dioxide, foam, etc. Special pipe systems are applicable to
areas of high fire potential where water may not be effective, such as
tanks for storage of flammable liquids.
Fixed systems must be supplemented by portable fire extinguishers. As
well as providing a means of rapidly extinguishing a fire, they can
often be used to prevent a small fire from spreading.
To be effective, portable extinguishers must be appropriate for the
type of fire; the HSGs provide information to this effect. For
example, water-based extinguishers can be used on ordinary
combustibles such as wood, paper, textiles, etc., where a
quenching/cooling effect is required. Dry chemical or carbon dioxide
extinguishers are used on flammable liquid and gas fires, such as oil,
gasoline, paint and grease, where an oxygen-exclusion or
flame-interrupting effect is necessary. Dry powder agents are
necessary to combat metal fires.
In addition, portable extinguishers must be available in sufficient
quantity to protect against potential fires in any area. They should
be located where they are readily accessible for immediate use,
maintained in good operating condition, inspected frequently and
recharged as necessary. Workers should be trained to use them
effectively.
If a fire or explosion warrants intervention by outside fire service
personnel, these personnel should be advised of the nature of the
chemicals and the worksite so that they can evaluate what is needed
for their own safety.
4.4 Storage
The HSGs give some advice on storage (see Fig. 45), but the
manufacturers' or suppliers' MSDSs should give specific instructions
on the storage of each chemical. These conditions must be strictly
adhered to, as storage requirements vary according to the nature of
the chemical being stored. Incorrect storage can have disastrous
results, e.g. fire, explosion or release of toxic chemicals.
The MSDS should include the following information:
* Need for isolation -- Certain chemicals must not be stored
together, as any vapours or leaks may, if they intermingle, lead
to an explosion.
Figure 45. Storage (from HSG 19: Pentachlorophenol)
4.3 Storage
Technical PCP is a solid. The formulated product is usually a
solution in oil or organic solvent, or an emulsifiable concentrate.
All PCP products should be stored in secure, well-ventilated
buildings under cool and dry conditions, and out of reach of
children and unauthorized persons. Keep away from food, drink,
and animal feed.
* Flash point -- Chemicals must be stored at a temperature below
their flash points. (Refer to Chapter 2 of this Manual for a
discussion of flash point.) Chemicals with flash points below
34°C are especially dangerous.
* Upper and lower flammable limits -- Information on flammable
limits can be found in Chapter 2. MSDSs often specify a
"well-ventilated" store-room for particular chemicals, and it is
essential to comply with this requirement. More specific
guidance on the amount of ventilation required can be obtained
from the manufacturers of the chemicals and guidance can be
checked by an industrial hygienist or ventilation engineer.
* Auto-ignition temperature -- The storage temperature must
obviously be below the auto-ignition temperature (see Chapter 2).
* Type of container -- Chemicals may react with the material from
which containers are made; the type of container required must be
specified on the MSDS. This is especially important if chemicals
may be transferred from one container to another. Also, there
may be other requirements, such as pressure relief valves, which
are relevant to the storage of particular chemicals.
* Type of flooring -- This should be specified, as the flooring
must be resistant to, and not potentially reactive with, the
chemical being stored.
* Dykes (berms or bunding) -- These are low walls or embankments
that are constructed around the storage area to contain any leaks
from storage containers. The height of the dyke should be
sufficient to contain any leaks that may occur and any water or
foam sprayed in the event of a fire.
* Alarms -- Alarms should be fitted in areas where potentially
dangerous chemicals are stored to give early warning of releases
of those chemicals.
4.5 Spillage
Many spills can be prevented by thorough planning of work, provision
of suitable equipment for carrying out the work, regular preventive
maintenance and good training of workers. Any spillages that do
happen should be thoroughly investigated and remedial action taken to
prevent their recurrence.
Employers should ensure that they have the necessary plans and
equipment to deal with spillages, that the workforce and their
representatives have been consulted about all such planning and that
the necessary training is carried out regularly.
When a spillage occurs, suitable precautions must first be taken to
protect workers from the dangers of the chemical (fumes, burns, etc.)
before steps are taken to deal with the spillage (see Fig. 46).
Some general measures that may apply include the following:
* use self-contained breathing apparatus and full protective
clothing, when applicable;
* remove ignition sources;
* do not smoke;
* evacuate area;
* collect leaking liquid in sealable containers;
* prevent liquid from spreading or contaminating other areas,
vegetation, waterways and cargo, with a barrier of the most
suitable available material, e.g. soil or sand;
* in some cases (e.g. hydrazine), a foam can be applied to slow
down vaporization;
* absorb spills in sand, soil, moist sawdust or other inert
material and transfer to a suitable container; then remove to a
safe place, with disposal in accordance with local regulations;
* sweep up solid products, and transfer to a suitable container;
and
* depending on the chemical, do not allow run-off into sewers: it
may cause an explosion, kill wildlife or affect water supplies.
Figure 46. Spillage (from HSG 35: Phosphorus Trichloride)
4.7.1 Spillage
Liquid spillage should be handled only by trained personnel wearing
protective clothing and full-face mask/positive-pressure breathing
apparatus. The spillage should be absorbed in dry sand or in an
inert absorbent material and shovelled into sealable polyethylene-
lined containers for disposal.
Vapour drifting towards people can be "knocked-down" with water
spray. Do not wet the spillage itself and never use a water jet on
a spillage. When the absorbent medium has been cleared from the
ground, flush the contaminated area with water until the fumes
disappear; residues may be neutralized with soda ash (sodium
carbonate or lime).
If phosphorus trichloride or phosphorus oxychloride enters a
watercourse or sewer, or contaminates soil or vegetation, warn the
police and public authorities immediately.
The actual steps taken will depend on the chemical in question, and
information on this must already have been collected from the MSDS and
from consultations with the manufacturers and/or suppliers and
emergency services (e.g. fire authorities) and incorporated in the
employer's written statement on spills.
4.6 Disposal
4.6.1 Means of disposal
Given that enormous volumes of waste are generated in the production
and use of chemicals, waste disposal has become a key issue in
environmental protection. As with protection of the workplace, a
hierarchy of controls should be applied when dealing with chemical
waste, as follows:
* reduction of waste at the source;
* segregation of waste;
* recovery and recycling;
* waste exchange;
* incineration;
* immobilization of intractable wastes;
* landfill;
* discharge into sewer; and
* storage.
The volume of hazardous waste can be reduced by modifying a process or
improving process control to produce less waste or to render the waste
less hazardous.
Waste can be segregated to assist analysis, control and treatment --
thereby reducing disposal costs.
* Recycling -- The best-known form of recycling is the recovery of
useful fractions for reuse. These processes are usually carried
out by specialist recovery operators off-site and involve
recovery of materials such as oils and solvents, as well as other
valuable materials, such as the silver in photographic waste.
Not all wastes are suitable for recycling. Even when valuable
materials such as mercury or lead are present, full recovery
might not be economically feasible owing to such factors as high
processing costs, small volumes and irregular composition. As
extraction techniques improve, more waste products are likely to
become eligible for recycling.
* Waste exchanges -- A considerable amount of waste is suitable for
exchange. The aim of the exchange is to put potential users of
waste materials in touch with industries producing the waste, and
vice versa. Waste exchanges reduce the volume of wastes requiring
landfill or incineration and therefore reduce the load on
disposal facilities and the environment, as well as the cost of
disposal.
* Incineration -- This process, which involves burning in special
high-temperature (1200°C) incinerators, is the only
environmentally acceptable means of disposing of some wastes,
such as chlorinated hydrocarbons, polychlorinated biphenyls
(PCBs) and dioxins (see Fig 47). These chemicals are highly
toxic, persistent and non-biodegradable. Incineration effectively
destroys many organic wastes, and the energy in solvent and fuel
waste can be exploited in the process. On the other hand,
inorganic chemical dyes in plastics may cause pollution problems
when the plastics are incinerated, and dangerous dioxins may be
formed if some organic materials are incinerated at too low
temperatures.
Figure 47. Disposal (from HSG 19: Pentachlorophenol)
4.5.2 Disposal
Surplus product, contaminated absorbents and containers should be
burned at a high temperature in an appropriate incinerator with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, in an area where there is no risk of contamination of
surface water or groundwater. Comply with any local legislation.
* Immobilization of intractable wastes -- This is used to isolate a
hazard physically and/or chemically from its surroundings.
* Chemical fixation -- Waste is incorporated into an inert matrix
(framework) to limit or prevent its migration into the
environment. Should the waste block be fractured, the waste
remains bound to the matrix, and leaching is prevented.
* Encapsulation -- The waste is sealed within a stable, inert
material to prevent contact with the environment and prevent
movement in the environment (migration). Should the
encapsulating jacket be broken, the waste could leach away.
Encapsulation is more suited to those wastes that pose a handling
hazard but are relatively inert once buried (e.g. asbestos).
* Landfill -- Because of the limited amount of suitable land
available for waste disposal, it is increasingly desirable that
liquid wastes be treated by chemical and physical methods,
followed by biological oxidation (digestion by special bacteria
or algae). After treatment, most of the liquid will be suitable
for discharge to sewers. The remaining liquid can be recycled or
incinerated. Landfills could then be used for most residual
solids or pastes because they are of smaller volume and less
likely to migrate through the soil. A number of solid hazardous
wastes are not regarded as suitable for disposal to normal
chemical landfill facilities. These wastes require the higher
degree of safety afforded by secure landfill, whereby waste is
poured into small cells lined with an impermeable clay or
synthetic material and subsequently buried under a layer of soil.
However, it is highly unlikely that such a system would function
without some leakage through the clay or synthetic material,
without any damage to the cell and with 100% collection of any
leaks. Added to this are the potential problems of infiltration
by rainwater and ensuring permanent maintenance of the cell if
the company moves away or ceases to operate.
* Disposal of less hazardous waste to the sewer -- This is
generally not recommended (see Fig. 48). Most sewage authorities
make regulatory provisions for the control of the kind and
quantity of (less) hazardous waste that may be disposed of
through the sewer. Improper disposal to the sewer can disrupt
the biological treatment of sewage and represent a hazard at
sewer outfalls. In addition, toxic chemicals (e.g. heavy metals)
may accumulate in the sewage sludge and create hazards when they
are disposed of.
Figure 48. "Avoid run-off into sewers"
(from HSG 34: Fenvalerate)
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams, water
courses, open waterways, ditches, fields with drainage systems, or to
the catchment areas of boreholes, wells, springs, or ponds.
* Storage of intractable wastes -- A large volume of hazardous
waste is currently stored -- usually in steel drums on industrial
sites, awaiting the development of satisfactory disposal methods
-- because it is too toxic to be disposed of legally into air,
water or landfill sites. Most drums are stored in the open, and
many contain corrosive materials. There is an added risk of
hazard from fires, structural damage and vandalism of such toxic
waste stores. It is likely that some of these drums will corrode
and leak. In some countries, such stores must be registered with
authorities who, in turn, have a duty to inspect them.
4.6.2 An example of the effects of incorrect waste disposal
For years, the residents of Love Canal, a residential community near
Niagara Falls, New York, USA, complained of noxious vapours in their
basements, which they believed originated from a disused toxic waste
dump beneath their housing estate. Following prolonged rains and
heavy snows in the winter of 1977, water seeped into the dump-site,
causing it to overflow and forcing the chemicals out into the soil.
Two hundred and thirty-five families were evacuated, and Love Canal
was declared a federal disaster area, the first instance of a national
emergency being caused by chemical pollution. Later, more families
had to be moved when it was discovered that the toxic waste was
migrating further afield via underground streams.
Almost 22 000 tonnes of chemicals had been buried in Love Canal; the
canal had been capped with clay and covered with soil in 1953. The
clay cap was apparently damaged during later building work on the
site. Approximately 300 different chemical compounds were buried
there, although no records were kept of what, or how much, was dumped.
Studies now reveal that the Love Canal area experienced three times
the normal rate of miscarriages and three-and-a-half times the normal
rate of birth defects, most notably cleft palate, mental retardation
and club foot.
In addition, US$30 million have been spent on cleaning up the area,
and US$4.5 million on rehousing residents.
5. HOW CHEMICALS CAN POISON THE ENVIRONMENT - CHAPTER 5
This chapter is a background to chapter 5 of the HSG: "Hazards for the
Environment and their Prevention".
Almost any industrial or agricultural activity in which chemicals are
used will, by its very nature, have an impact on the environment (see
Fig. 49).
Figure 49. How chemicals can affect the environment
(from HSG 35: Phosphorus Trichloride)
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Phosphorus trichloride and phosphorus oxychloride hydrolyse in water
to form hydrochloric and phosphorous or phosphoric acids. The toxic
effects on organisms in the environment depend on the extent to which
the hydrogen ion is diluted and buffered; organisms die if they come
in contact with undiluted material.
In general, an environmental hazard will occur only in the immediate
area of a spillage or if disposed of improperly. Recovery of soil
and surface waters is rapid, and there are no residual effects on
the environment.
5.1 How do chemicals get into the environment?
Chemicals are released from the workplace into the environment in the
form of liquid, dust, fumes or gas. Such releases can be planned
(part of the work process, e.g. smokestack emissions, exhaust
ventilation, waste water into rivers, etc.) or unplanned (accidental).
Chemicals can also enter the environment during transport (e.g. from
the site of manufacture to the site of use), during their intended use
(e.g. pesticide spraying) or through disposal in landfills and
waterways. Chemicals also enter the environment through
non-occupational activities. Examples include exhaust fumes from motor
vehicles. Additionally, naturally occurring chemicals such as metals
in ores may be released by natural processes.
Chemicals released as smoke and dust from factory chimneys will
eventually fall to the earth's surface as dust and in rain. Wind
speed, air currents and other climatic conditions will determine when
and where the chemicals will be deposited. Some will settle on water
in lakes, rivers and streams, eventually passing into sediments and
nearby soils. Others may be transported over long distances and may
even affect the chemical and physical processes of the atmosphere and
climate. For example, the effects of sulfur and nitrogen oxides
released in industrial areas have contributed to acid rain, often
falling in countries other than those in which they were emitted.
Emissions of carbon dioxide, methane, ozone, etc. are thought to
contribute to global warming.
Once chemicals are released into the environment in this way, humans
and animals will be exposed to them through breathing contaminated
air, from direct contact with contaminated soil and water, through
eating plants grown on contaminated soils, by drinking contaminated
water supplies or by consuming food from contaminated animals (see
Fig. 50).
* First-aid boxes should be provided in all work areas and workers
trained in emergency first-aid procedures for any accident or
chemical exposure that may occur.
* Telephone numbers for medical assistance and ambulances should be
prominently displayed in the workplace. A telephone must always
be available for use in an emergency.
* There should be an "emergency response plan" in which individuals
are assigned to perform certain tasks (such as summon help) while
all other personnel quickly leave the workplace. The plan should
be written down and prominently displayed in the workplace and
should be practised regularly.
* A system should operate whereby all accidents and injuries, no
matter how slight, are reported in writing to the management or
employer. These accident records should be made available to
workers and their representatives and analysed to determine how
to prevent the same, or similar, accidents from occurring again.
In many countries, occupational accidents and injuries must be
reported to government bodies.
* If an injured worker is sent to hospital or to see a doctor or
nurse, it is essential that information on the chemicals involved
in the accident be sent with the worker. The manufacturer's MSDS
and the ICSC or Summary of Chemical Safety Information contained
in HSGs (see Chapter 3) are usually suitable for this purpose.
4.2 Health surveillance
Health surveillance or medical monitoring consists of periodic
examination of a worker by a doctor, nurse or other health worker in
order to detect any health effects of a chemical, to monitor the
progression or reversal of symptoms or to see if treatment or
preventive measures are effective. Tests of lung, heart, liver and
kidney functions, for instance, may be performed. Although in some
cases -- e.g. chemicals that cause asthma -- such testing is useful,
it should always be remembered that medical monitoring can detect
damage to health only after it has occurred (see Fig. 43). Further
damage can then be prevented, but medical monitoring does not actually
prevent the occurrence of the initial health effects.
Figure 43. Health surveillance (from HSG 6: Methylene Chloride)
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically. Administer
oxygen if needed. Do not administer fats, oils, alcohol, epinephrine,
or ephedrine. Pay attention to signs of anoxaemia, impending
pulmonary oedema, and ischaemia of the heart. Monitor ECG. Treat
skin as for a thermal burn.
4.3 Health Surveillance Advice
Persons handling methylene chloride should frequently undergo
periodic medical examination with emphasis on the functioning
of the central nervous system and irritation of the skin and
eyes. Monitoring of blood-carboxyhaemoglobin concentrations can
give an indication of the extent of exposure.
Medical monitoring is often unable to detect the long-term or chronic
health effects of chemicals until they are quite advanced. This
applies particularly to:
* cancer caused by carcinogenic chemicals;
* effects on the brain and nervous system; and
* reproductive effects.
Medical monitoring is useful for assessing the effectiveness of
measures implemented to control chemical exposure, but it is not a
substitute for environmental monitoring and control methods.
4.3 Fire and explosion hazards
Many chemicals used in the workplace will burn or explode easily.
Accidental fires or explosions of flammable or explosive chemicals
represent major hazards. Special precautions must be taken when
handling, storing or transporting these chemicals to prevent fires or
explosions (see Fig. 44).
Figure 44. Explosion and fire hazards
(from HSG 6: Methylene Chloride)
4.3 Explosion and Fire Hazards
4.3.1 Explosion hazards
Methylene chloride may react explosively with light metals (lithium,
sodium, potassium, aluminium, magnesium) as well as with alloys of
sodium. Explosive mixtures are formed with dinitrogen tetroxide
(N2O4) dinitrogen pentoxide (N2O5), nitric acid, and
compressed oxygen.
4.3.2 Fire hazards
Methylene chloride is flammable in an atmosphere with an increased
oxygen content. In pure oxygen, the flammable limits (explosive
limits) are 15.5% and 66.4%. Methylene chloride is also flammable
if small amounts of combustible material are added. The compound
decomposes in contact with open flames and glowing surfaces with the
formation of harmful gases, such as hydrogen chloride, which forms
mists of hydrochloric acid with moisture, and phosgene.
4.3.3 Prevention
Do not use methylene chloride in the vicinity of a fire, a hot
surface (e.g. a portable heating unit), or during welding. Do
not smoke. Do not add any combustible material to methylene
chloride. Avoid using compressed oxygen when handling
methylene chloride. Avoid contact between the compound and
light metals, alloys of light metals, dinitrogen tetroxide,
dinitrogen pentoxide, nitric acid, or liquid oxygen. In case
of fire, keep drums cool by spraying with water.
Fire (or combustion) is the violent combination of oxygen in the air
with a combustible chemical while producing heat (an exothermic
reaction).
In fires involving chemicals or chemical products, the chemicals
usually become broken down into combustion by-products. Many of
these combustion by-products are highly toxic gases and fumes. This
may be the case even if the original chemical was not particularly
toxic. For example, the gas Freon 12, or dichlorodifluoromethane, is
widely used as a refrigerant. It is toxic only at very high
concentrations. However, the thermal decomposition products of Freon
12 are very toxic gases, including phosgene, which causes potentially
fatal pulmonary oedema (accumulation of water in the lungs), even at
very low levels of exposure. Similarly, polyurethane foam, which is
commonly used as insulation in industry, has relatively low toxicity
in the cured state. However, when polyurethane foam burns, large
amounts of highly toxic gases, including cyanide and hydrogen
chloride, are released.
4.3.1 Flammable substances
A flammable substance is any chemical or material that will continue
to burn after having been ignited. A substance of high flammability
is one that will easily ignite at room temperature on contact with
low-energy ignition sources such as a spark. A substance of low
flammability will ignite only after heating or prolonged contact with
a high-energy ignition source. The flammability of a substance is
often ranked according to its ability to catch fire at certain
temperatures:
temperature
extremely flammable < 0°C
highly flammable 0-21°C
flammable 21-55°C
combustible > 55°C
Other important information about the flammability of a substance can
be gathered by looking at the flash point and the flammability
(explosive) limits (see Chapter 1) for that substance. This
information is given in the Physical Properties section of the ICSC
and usually appears on the MSDS supplied by the chemical manufacturer.
4.3.2 Dust explosions
Any solid material that is capable of burning in a finely divided form
can give rise to a dust explosion hazard. All that is necessary is
for enough dust to be in the air and for a source of ignition to be
present. Flame from the source of ignition will spread in all
directions through the cloud of dust, burning the dust and causing
expansion, thus increasing pressure on the surroundings. In a grain
store, for example, this pressure could be great enough to cause the
roof or walls to be blown off. The first explosion can whip dust that
is lying on window sills, pipework and equipment into the air, and a
second explosion can result.
Many dusts, including natural materials such as starch, sugar, flour,
coal and wood, plastics and organic chemicals, and light metals such
as aluminium, magnesium and titanium, can explode in this way.
Accordingly, dust explosion must be guarded against in agriculture,
chemical, metallurgical and process industries, as well as coal
mining.
4.3.3 Sources of ignition
Flammable substances or dust clouds may be ignited in one of the
following ways:
* Direct flames -- matches, cigarettes, soldering, welding or
cutting torches.
* Heat radiation -- all sources of heat emit heat (thermal)
radiation, which may ignite a flammable material at a distance.
* Electrical sparks -- produced by electric current in a number of
ways, including during electric arc welding; by power supplies
such as sockets and switches and sliding contacts; from electric
motors in power tools or the ignition systems of internal
combustion engines; by lightning; and as a result of static
electricity build-up between two non-conducting materials such as
belts on a pulley, rubber tyres on the ground or liquids pumped
or poured through pipes.
* Mechanically produced sparks -- two hard materials rubbing
against each other at high speed may produce sparks capable of
igniting flammable chemicals, e.g. when grinding.
* Chemical reactions -- many chemical reactions produce heat. The
design of the chemical process should take this into account and
make provision for the removal of excess heat. However, excess
heat may develop if the incorrect or wrong proportions of
chemicals are used. This may start a fire or trigger an
explosion.
* Spontaneous combustion -- some substances oxidize (combine with
oxygen) as soon as they are exposed to air. Usually this occurs
slowly, but with some chemicals it may happen rapidly and produce
considerable heat in the process. These chemicals must be stored
in tanks or containers that allow all air to be removed and
replaced with a gas that will not combine with the chemical
(usually nitrogen). If this mechanism fails and air enters the
container, a fire or explosion may result.
Safety information about fire hazards is given in chapter 4 of the
HSGs and is also contained in the ICSCs. Information on fire and
explosion hazards should always be included in chemical manufacturers'
MSDS.
4.3.4 Fire-fighting
Equipment used to extinguish and control fires is of two types: fixed
and portable. Fixed systems include water equipment, automatic
sprinklers, hydrants and special pipe systems for dry chemicals,
carbon dioxide, foam, etc. Special pipe systems are applicable to
areas of high fire potential where water may not be effective, such as
tanks for storage of flammable liquids.
Fixed systems must be supplemented by portable fire extinguishers. As
well as providing a means of rapidly extinguishing a fire, they can
often be used to prevent a small fire from spreading.
To be effective, portable extinguishers must be appropriate for the
type of fire; the HSGs provide information to this effect. For
example, water-based extinguishers can be used on ordinary
combustibles such as wood, paper, textiles, etc., where a
quenching/cooling effect is required. Dry chemical or carbon dioxide
extinguishers are used on flammable liquid and gas fires, such as oil,
gasoline, paint and grease, where an oxygen-exclusion or
flame-interrupting effect is necessary. Dry powder agents are
necessary to combat metal fires.
In addition, portable extinguishers must be available in sufficient
quantity to protect against potential fires in any area. They should
be located where they are readily accessible for immediate use,
maintained in good operating condition, inspected frequently and
recharged as necessary. Workers should be trained to use them
effectively.
If a fire or explosion warrants intervention by outside fire service
personnel, these personnel should be advised of the nature of the
chemicals and the worksite so that they can evaluate what is needed
for their own safety.
4.4 Storage
The HSGs give some advice on storage (see Fig. 45), but the
manufacturers' or suppliers' MSDSs should give specific instructions
on the storage of each chemical. These conditions must be strictly
adhered to, as storage requirements vary according to the nature of
the chemical being stored. Incorrect storage can have disastrous
results, e.g. fire, explosion or release of toxic chemicals.
The MSDS should include the following information:
* Need for isolation -- Certain chemicals must not be stored
together, as any vapours or leaks may, if they intermingle, lead
to an explosion.
Figure 45. Storage (from HSG 19: Pentachlorophenol)
4.3 Storage
Technical PCP is a solid. The formulated product is usually a
solution in oil or organic solvent, or an emulsifiable concentrate.
All PCP products should be stored in secure, well-ventilated
buildings under cool and dry conditions, and out of reach of
children and unauthorized persons. Keep away from food, drink,
and animal feed.
* Flash point -- Chemicals must be stored at a temperature below
their flash points. (Refer to Chapter 2 of this Manual for a
discussion of flash point.) Chemicals with flash points below
34°C are especially dangerous.
* Upper and lower flammable limits -- Information on flammable
limits can be found in Chapter 2. MSDSs often specify a
"well-ventilated" store-room for particular chemicals, and it is
essential to comply with this requirement. More specific
guidance on the amount of ventilation required can be obtained
from the manufacturers of the chemicals and guidance can be
checked by an industrial hygienist or ventilation engineer.
* Auto-ignition temperature -- The storage temperature must
obviously be below the auto-ignition temperature (see Chapter 2).
* Type of container -- Chemicals may react with the material from
which containers are made; the type of container required must be
specified on the MSDS. This is especially important if chemicals
may be transferred from one container to another. Also, there
may be other requirements, such as pressure relief valves, which
are relevant to the storage of particular chemicals.
* Type of flooring -- This should be specified, as the flooring
must be resistant to, and not potentially reactive with, the
chemical being stored.
* Dykes (berms or bunding) -- These are low walls or embankments
that are constructed around the storage area to contain any leaks
from storage containers. The height of the dyke should be
sufficient to contain any leaks that may occur and any water or
foam sprayed in the event of a fire.
* Alarms -- Alarms should be fitted in areas where potentially
dangerous chemicals are stored to give early warning of releases
of those chemicals.
4.5 Spillage
Many spills can be prevented by thorough planning of work, provision
of suitable equipment for carrying out the work, regular preventive
maintenance and good training of workers. Any spillages that do
happen should be thoroughly investigated and remedial action taken to
prevent their recurrence.
Employers should ensure that they have the necessary plans and
equipment to deal with spillages, that the workforce and their
representatives have been consulted about all such planning and that
the necessary training is carried out regularly.
When a spillage occurs, suitable precautions must first be taken to
protect workers from the dangers of the chemical (fumes, burns, etc.)
before steps are taken to deal with the spillage (see Fig. 46).
Some general measures that may apply include the following:
* use self-contained breathing apparatus and full protective
clothing, when applicable;
* remove ignition sources;
* do not smoke;
* evacuate area;
* collect leaking liquid in sealable containers;
* prevent liquid from spreading or contaminating other areas,
vegetation, waterways and cargo, with a barrier of the most
suitable available material, e.g. soil or sand;
* in some cases (e.g. hydrazine), a foam can be applied to slow
down vaporization;
* absorb spills in sand, soil, moist sawdust or other inert
material and transfer to a suitable container; then remove to a
safe place, with disposal in accordance with local regulations;
* sweep up solid products, and transfer to a suitable container;
and
* depending on the chemical, do not allow run-off into sewers: it
may cause an explosion, kill wildlife or affect water supplies.
Figure 46. Spillage (from HSG 35: Phosphorus Trichloride)
4.7.1 Spillage
Liquid spillage should be handled only by trained personnel wearing
protective clothing and full-face mask/positive-pressure breathing
apparatus. The spillage should be absorbed in dry sand or in an
inert absorbent material and shovelled into sealable polyethylene-
lined containers for disposal.
Vapour drifting towards people can be "knocked-down" with water
spray. Do not wet the spillage itself and never use a water jet on
a spillage. When the absorbent medium has been cleared from the
ground, flush the contaminated area with water until the fumes
disappear; residues may be neutralized with soda ash (sodium
carbonate or lime).
If phosphorus trichloride or phosphorus oxychloride enters a
watercourse or sewer, or contaminates soil or vegetation, warn the
police and public authorities immediately.
The actual steps taken will depend on the chemical in question, and
information on this must already have been collected from the MSDS and
from consultations with the manufacturers and/or suppliers and
emergency services (e.g. fire authorities) and incorporated in the
employer's written statement on spills.
4.6 Disposal
4.6.1 Means of disposal
Given that enormous volumes of waste are generated in the production
and use of chemicals, waste disposal has become a key issue in
environmental protection. As with protection of the workplace, a
hierarchy of controls should be applied when dealing with chemical
waste, as follows:
* reduction of waste at the source;
* segregation of waste;
* recovery and recycling;
* waste exchange;
* incineration;
* immobilization of intractable wastes;
* landfill;
* discharge into sewer; and
* storage.
The volume of hazardous waste can be reduced by modifying a process or
improving process control to produce less waste or to render the waste
less hazardous.
Waste can be segregated to assist analysis, control and treatment --
thereby reducing disposal costs.
* Recycling -- The best-known form of recycling is the recovery of
useful fractions for reuse. These processes are usually carried
out by specialist recovery operators off-site and involve
recovery of materials such as oils and solvents, as well as other
valuable materials, such as the silver in photographic waste.
Not all wastes are suitable for recycling. Even when valuable
materials such as mercury or lead are present, full recovery
might not be economically feasible owing to such factors as high
processing costs, small volumes and irregular composition. As
extraction techniques improve, more waste products are likely to
become eligible for recycling.
* Waste exchanges -- A considerable amount of waste is suitable for
exchange. The aim of the exchange is to put potential users of
waste materials in touch with industries producing the waste, and
vice versa. Waste exchanges reduce the volume of wastes requiring
landfill or incineration and therefore reduce the load on
disposal facilities and the environment, as well as the cost of
disposal.
* Incineration -- This process, which involves burning in special
high-temperature (1200°C) incinerators, is the only
environmentally acceptable means of disposing of some wastes,
such as chlorinated hydrocarbons, polychlorinated biphenyls
(PCBs) and dioxins (see Fig 47). These chemicals are highly
toxic, persistent and non-biodegradable. Incineration effectively
destroys many organic wastes, and the energy in solvent and fuel
waste can be exploited in the process. On the other hand,
inorganic chemical dyes in plastics may cause pollution problems
when the plastics are incinerated, and dangerous dioxins may be
formed if some organic materials are incinerated at too low
temperatures.
Figure 47. Disposal (from HSG 19: Pentachlorophenol)
4.5.2 Disposal
Surplus product, contaminated absorbents and containers should be
burned at a high temperature in an appropriate incinerator with
effluent gas scrubbing. When no incinerator is available, bury in an
approved dump, in an area where there is no risk of contamination of
surface water or groundwater. Comply with any local legislation.
* Immobilization of intractable wastes -- This is used to isolate a
hazard physically and/or chemically from its surroundings.
* Chemical fixation -- Waste is incorporated into an inert matrix
(framework) to limit or prevent its migration into the
environment. Should the waste block be fractured, the waste
remains bound to the matrix, and leaching is prevented.
* Encapsulation -- The waste is sealed within a stable, inert
material to prevent contact with the environment and prevent
movement in the environment (migration). Should the
encapsulating jacket be broken, the waste could leach away.
Encapsulation is more suited to those wastes that pose a handling
hazard but are relatively inert once buried (e.g. asbestos).
* Landfill -- Because of the limited amount of suitable land
available for waste disposal, it is increasingly desirable that
liquid wastes be treated by chemical and physical methods,
followed by biological oxidation (digestion by special bacteria
or algae). After treatment, most of the liquid will be suitable
for discharge to sewers. The remaining liquid can be recycled or
incinerated. Landfills could then be used for most residual
solids or pastes because they are of smaller volume and less
likely to migrate through the soil. A number of solid hazardous
wastes are not regarded as suitable for disposal to normal
chemical landfill facilities. These wastes require the higher
degree of safety afforded by secure landfill, whereby waste is
poured into small cells lined with an impermeable clay or
synthetic material and subsequently buried under a layer of soil.
However, it is highly unlikely that such a system would function
without some leakage through the clay or synthetic material,
without any damage to the cell and with 100% collection of any
leaks. Added to this are the potential problems of infiltration
by rainwater and ensuring permanent maintenance of the cell if
the company moves away or ceases to operate.
* Disposal of less hazardous waste to the sewer -- This is
generally not recommended (see Fig. 48). Most sewage authorities
make regulatory provisions for the control of the kind and
quantity of (less) hazardous waste that may be disposed of
through the sewer. Improper disposal to the sewer can disrupt
the biological treatment of sewage and represent a hazard at
sewer outfalls. In addition, toxic chemicals (e.g. heavy metals)
may accumulate in the sewage sludge and create hazards when they
are disposed of.
Figure 48. "Avoid run-off into sewers"
(from HSG 34: Fenvalerate)
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams, water
courses, open waterways, ditches, fields with drainage systems, or to
the catchment areas of boreholes, wells, springs, or ponds.
* Storage of intractable wastes -- A large volume of hazardous
waste is currently stored -- usually in steel drums on industrial
sites, awaiting the development of satisfactory disposal methods
-- because it is too toxic to be disposed of legally into air,
water or landfill sites. Most drums are stored in the open, and
many contain corrosive materials. There is an added risk of
hazard from fires, structural damage and vandalism of such toxic
waste stores. It is likely that some of these drums will corrode
and leak. In some countries, such stores must be registered with
authorities who, in turn, have a duty to inspect them.
4.6.2 An example of the effects of incorrect waste disposal
For years, the residents of Love Canal, a residential community near
Niagara Falls, New York, USA, complained of noxious vapours in their
basements, which they believed originated from a disused toxic waste
dump beneath their housing estate. Following prolonged rains and
heavy snows in the winter of 1977, water seeped into the dump-site,
causing it to overflow and forcing the chemicals out into the soil.
Two hundred and thirty-five families were evacuated, and Love Canal
was declared a federal disaster area, the first instance of a national
emergency being caused by chemical pollution. Later, more families
had to be moved when it was discovered that the toxic waste was
migrating further afield via underground streams.
Almost 22 000 tonnes of chemicals had been buried in Love Canal; the
canal had been capped with clay and covered with soil in 1953. The
clay cap was apparently damaged during later building work on the
site. Approximately 300 different chemical compounds were buried
there, although no records were kept of what, or how much, was dumped.
Studies now reveal that the Love Canal area experienced three times
the normal rate of miscarriages and three-and-a-half times the normal
rate of birth defects, most notably cleft palate, mental retardation
and club foot.
In addition, US$30 million have been spent on cleaning up the area,
and US$4.5 million on rehousing residents.
5. HOW CHEMICALS CAN POISON THE ENVIRONMENT - CHAPTER 5
This chapter is a background to chapter 5 of the HSG: "Hazards for the
Environment and their Prevention".
Almost any industrial or agricultural activity in which chemicals are
used will, by its very nature, have an impact on the environment (see
Fig. 49).
Figure 49. How chemicals can affect the environment
(from HSG 35: Phosphorus Trichloride)
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Phosphorus trichloride and phosphorus oxychloride hydrolyse in water
to form hydrochloric and phosphorous or phosphoric acids. The toxic
effects on organisms in the environment depend on the extent to which
the hydrogen ion is diluted and buffered; organisms die if they come
in contact with undiluted material.
In general, an environmental hazard will occur only in the immediate
area of a spillage or if disposed of improperly. Recovery of soil
and surface waters is rapid, and there are no residual effects on
the environment.
5.1 How do chemicals get into the environment?
Chemicals are released from the workplace into the environment in the
form of liquid, dust, fumes or gas. Such releases can be planned
(part of the work process, e.g. smokestack emissions, exhaust
ventilation, waste water into rivers, etc.) or unplanned (accidental).
Chemicals can also enter the environment during transport (e.g. from
the site of manufacture to the site of use), during their intended use
(e.g. pesticide spraying) or through disposal in landfills and
waterways. Chemicals also enter the environment through
non-occupational activities. Examples include exhaust fumes from motor
vehicles. Additionally, naturally occurring chemicals such as metals
in ores may be released by natural processes.
Chemicals released as smoke and dust from factory chimneys will
eventually fall to the earth's surface as dust and in rain. Wind
speed, air currents and other climatic conditions will determine when
and where the chemicals will be deposited. Some will settle on water
in lakes, rivers and streams, eventually passing into sediments and
nearby soils. Others may be transported over long distances and may
even affect the chemical and physical processes of the atmosphere and
climate. For example, the effects of sulfur and nitrogen oxides
released in industrial areas have contributed to acid rain, often
falling in countries other than those in which they were emitted.
Emissions of carbon dioxide, methane, ozone, etc. are thought to
contribute to global warming.
Once chemicals are released into the environment in this way, humans
and animals will be exposed to them through breathing contaminated
air, from direct contact with contaminated soil and water, through
eating plants grown on contaminated soils, by drinking contaminated
water supplies or by consuming food from contaminated animals (see
Fig. 50).
 5.2 How do chemicals affect the environment?
Just as humans and animals can be affected by exposure to toxic
chemicals (as outlined in Chapter 2), chemicals can also have an
impact on the environment and the organisms living in the different
environmental media -- the air, water and soil.
5.2.1 Air
Many chemical pollutants in the air begin to have an effect on plants
and animals at extremely low concentrations, at even less than 1 µg
(or one-millionth of a gram) of chemical per cubic metre of air.
Plants and animals vary widely in their sensitivity to chemicals in
the air.
Fluorides released from aluminium smelters are an example of chronic
air pollutants that have caused great damage to livestock over long
periods of time. Animals exposed to excessive fluoride pollution
develop tooth, bone and joint diseases; they become unable to chew
their food or move. As a result, they lose weight and eventually die.
Fluoride builds up in the teeth, bones and tissues of all grazing
animals and has been found in large amounts in deer, rabbits and birds
near aluminium smelters. Humans can also be affected.
Fluorides also affect plants, causing leaves to die, damage to fruit,
a slowing of growth and the failure of seed to sprout.
Plants that are sensitive to fluoride pollution, such as grape vines,
begin to suffer significant damage if fluoride concentrations in air
exceed 0.5 µg/m3.
Environmental contamination by chemicals can also occur following
massive accidental releases, with large-scale immediate and long-term
consequences. For example, at least 2500 people died and 200 000
people were injured when methyl isocyanate gas was released from the
Union Carbide pesticide plant in Bhopal, India, on 3 December 1984.
According to an International Trade Union report, the disaster was
attributable to poor process design, dangerous operating procedures,
lack of proper maintenance, faulty equipment and major cuts in
staffing levels, crew sizes, worker training programmes and skilled
supervision. Smaller releases of toxic chemicals had occurred,
leading to one death and numerous injuries at the Union Carbide plant
at Bhopal before the 1984 disaster, but little or nothing was done to
correct these problems, despite vigorous protests by the union
representing Union Carbide workers.
Apart from the immediate deaths, reports indicate that other long-term
health effects are being suffered by the survivors of the disaster.
These include lung problems and reproductive effects such as an
increased rate of miscarriages and stillbirths.
5.2.2 Water and soil
As with chemicals in the air, very low concentrations of heavy metals
and other chemicals in water can have an enormous impact, particularly
on fish and other aquatic species.
The effect a chemical pollutant has in water depends on the nature of
the pollutant and on factors such as acidity (pH), temperature, water
hardness, the presence of organic materials such as algae and weeds
and the oxygen content of the water. For example, the toxicity of
certain (heavy) metals tends to increase as the acidity of the water
increases.
5.3 Factors affecting a chemical's environmental impact
The impact of a chemical on organisms and ecological systems will
depend on the chemical's environmental transport, persistence,
bioaccumulation and toxicity.
* Environmental transport, or mobility, refers to the ease
with which a substance moves from one environmental medium to
another (air, water, soil, sediment, etc.) by such processes as
evaporation, leaching, weathering, etc.
* Environmental persistence is one aspect of a chemical's
environmental fate, i.e. what happens to it when it enters the
environment. Once in the environment, a substance can degrade,
in which case it is generally assumed to be non-hazardous to the
environment, at least on a large scale, even though there may be
local effects in the short term. On the other hand, if a
chemical is not readily degradable, it can persist in the
environment for long periods and become widely dispersed.
Environmental persistence is therefore the opposite of
degradation and can be measured in terms of half-life or
half-time of a chemical in an environmental medium, i.e. the time
required for the chemical to be reduced to half its original
amount. Local conditions may affect a chemical's persistence.
For instance, a chemical may persist for longer periods in colder
climates, whereas it may be readily broken down in sunlight and
at higher temperatures.
The organochlorine pesticides are a group of chemicals well known for
their persistence. A study found that after only one application of
the pesticide aldrin to soil, more than 34% was found to be present in
the soil five years later. Most of the remaining 66% must therefore
have remained in water, air or soil, either as unchanged aldrin or in
the form of other closely related chemicals formed by the
decomposition of aldrin. The process by which a chemical is modified
by a living organism is called biotransformation.
Bioaccumulation is the accumulation of a chemical by an organism to
a concentration that exceeds that of the surrounding environment. For
example, the concentration of aldrin in the tissues of experimental
snails has been found to be nearly 5000 times the concentration of
aldrin in the water in which the snails live. The more a chemical
persists, the greater the potential for bioaccumulation. Chemicals in
the environment can become increasingly concentrated in the tissues of
animals higher up in the food-chain, a process known as
biomagnification.
The octanol/water partition coefficient of a chemical (see Chapter 1
for a discussion of this value) is a good indicator of its
bioaccumulation potential: the higher the coefficient, the more a
chemical will tend to bioaccumulate.
* Toxicity. The impact of a chemical on the environment,
resulting from its toxicity, is more complex. Chemical
pollutants affect some environmental media more than others,
whether these are air, surface water, groundwater, soil or
sediments. As explained in Chapter 2, different species will
react in varying ways and to different degrees to the same
chemical. What may be highly toxic to aquatic life may not be
toxic to birds, for instance, even at the same dose. Toxicity
forms the basis of use of pesticides. Pesticides are applied in
order to damage or kill a target organism (i.e. the organism that
is subject to the action of the pesticide). But it is seldom or
never possible to limit the effects to target organisms.
Pesticides are therefore often perceived as a particular threat
to health and the environment.
By determining the effects of a chemical on different species,
toxicity studies are able to evaluate the environmental impact of that
chemical. The effects to be determined should include acute lethality
(oral, dermal, inhalation LD50 and LC50); sublethal effects on
non-mammals (soil organisms, bees, birds, etc.), mammals and plants
(usually expressed as the percentage of growth reduction as a result
of exposure to the chemical in water, air or soil); mutagenicity/
genotoxicity; teratogenicity; and carcinogenicity.
5.4 The importance of prevention
Once chemicals have been released into the environment, little or
nothing can be done to remove them. Many chemicals will continue to
damage animals and plants and poison food and water for years or
decades. Therefore, it is essential that chemical pollution of the
environment be prevented at its source -- i.e., wherever chemicals are
manufactured or used. Good occupational hygiene principles should
always be applied when using or handling any chemicals at work.
Chemicals must be controlled not simply in order to ensure compliance
with government standards (exposure limits, etc.; see Chapter 7), but
to prevent chemical contamination -- to the maximum extent possible --
of the general environment. In the first instance, this means
judicious use of less toxic and/or more readily degradable chemicals
and the use of more selective pesticides. Chemical processes should
be enclosed as far as possible to prevent release of chemicals as
dust, fumes or gas. Dust, fumes and gas that are generated during a
process such as grinding or welding should be captured by local
exhaust ventilation. Contaminated air from such processes, together
with contaminated air from ventilation of closed systems and general
ventilation, should not be allowed to escape to the atmosphere inside
the workplace, or outside, without first having had the contamination
removed. Some methods and principles for the prevention of
environmental chemical pollution are given in Chapter 4 of this Manual
(see sections on Storage, Spillage and Disposal).
6. SUMMARY OF CHEMICAL SAFETY INFORMATION - CHAPTER 6
Most HSGs contain a Summary of Chemical Safety Information or an ICSC
that summarizes information and advice on the relevant chemical. It
is designed for easy, quick reference for health workers concerned
with, and users of, chemicals. It should be displayed near entrances
to areas where exposure to the chemical in question could potentially
occur and, if possible, on processing equipment and containers.
A sample is given below; the terms used are defined in Chapters 1 and
2 of this Manual. All persons potentially exposed to chemicals should
have the instructions on the relevant ICSCs clearly explained to them.
1,4-DIOXANE ICSC: 0041
1,4-DIOXANE
1,4-Diethylene dioxide
1,4-Diethyleneoxide
C4H8O2
Molecular mass: 88.1
5.2 How do chemicals affect the environment?
Just as humans and animals can be affected by exposure to toxic
chemicals (as outlined in Chapter 2), chemicals can also have an
impact on the environment and the organisms living in the different
environmental media -- the air, water and soil.
5.2.1 Air
Many chemical pollutants in the air begin to have an effect on plants
and animals at extremely low concentrations, at even less than 1 µg
(or one-millionth of a gram) of chemical per cubic metre of air.
Plants and animals vary widely in their sensitivity to chemicals in
the air.
Fluorides released from aluminium smelters are an example of chronic
air pollutants that have caused great damage to livestock over long
periods of time. Animals exposed to excessive fluoride pollution
develop tooth, bone and joint diseases; they become unable to chew
their food or move. As a result, they lose weight and eventually die.
Fluoride builds up in the teeth, bones and tissues of all grazing
animals and has been found in large amounts in deer, rabbits and birds
near aluminium smelters. Humans can also be affected.
Fluorides also affect plants, causing leaves to die, damage to fruit,
a slowing of growth and the failure of seed to sprout.
Plants that are sensitive to fluoride pollution, such as grape vines,
begin to suffer significant damage if fluoride concentrations in air
exceed 0.5 µg/m3.
Environmental contamination by chemicals can also occur following
massive accidental releases, with large-scale immediate and long-term
consequences. For example, at least 2500 people died and 200 000
people were injured when methyl isocyanate gas was released from the
Union Carbide pesticide plant in Bhopal, India, on 3 December 1984.
According to an International Trade Union report, the disaster was
attributable to poor process design, dangerous operating procedures,
lack of proper maintenance, faulty equipment and major cuts in
staffing levels, crew sizes, worker training programmes and skilled
supervision. Smaller releases of toxic chemicals had occurred,
leading to one death and numerous injuries at the Union Carbide plant
at Bhopal before the 1984 disaster, but little or nothing was done to
correct these problems, despite vigorous protests by the union
representing Union Carbide workers.
Apart from the immediate deaths, reports indicate that other long-term
health effects are being suffered by the survivors of the disaster.
These include lung problems and reproductive effects such as an
increased rate of miscarriages and stillbirths.
5.2.2 Water and soil
As with chemicals in the air, very low concentrations of heavy metals
and other chemicals in water can have an enormous impact, particularly
on fish and other aquatic species.
The effect a chemical pollutant has in water depends on the nature of
the pollutant and on factors such as acidity (pH), temperature, water
hardness, the presence of organic materials such as algae and weeds
and the oxygen content of the water. For example, the toxicity of
certain (heavy) metals tends to increase as the acidity of the water
increases.
5.3 Factors affecting a chemical's environmental impact
The impact of a chemical on organisms and ecological systems will
depend on the chemical's environmental transport, persistence,
bioaccumulation and toxicity.
* Environmental transport, or mobility, refers to the ease
with which a substance moves from one environmental medium to
another (air, water, soil, sediment, etc.) by such processes as
evaporation, leaching, weathering, etc.
* Environmental persistence is one aspect of a chemical's
environmental fate, i.e. what happens to it when it enters the
environment. Once in the environment, a substance can degrade,
in which case it is generally assumed to be non-hazardous to the
environment, at least on a large scale, even though there may be
local effects in the short term. On the other hand, if a
chemical is not readily degradable, it can persist in the
environment for long periods and become widely dispersed.
Environmental persistence is therefore the opposite of
degradation and can be measured in terms of half-life or
half-time of a chemical in an environmental medium, i.e. the time
required for the chemical to be reduced to half its original
amount. Local conditions may affect a chemical's persistence.
For instance, a chemical may persist for longer periods in colder
climates, whereas it may be readily broken down in sunlight and
at higher temperatures.
The organochlorine pesticides are a group of chemicals well known for
their persistence. A study found that after only one application of
the pesticide aldrin to soil, more than 34% was found to be present in
the soil five years later. Most of the remaining 66% must therefore
have remained in water, air or soil, either as unchanged aldrin or in
the form of other closely related chemicals formed by the
decomposition of aldrin. The process by which a chemical is modified
by a living organism is called biotransformation.
Bioaccumulation is the accumulation of a chemical by an organism to
a concentration that exceeds that of the surrounding environment. For
example, the concentration of aldrin in the tissues of experimental
snails has been found to be nearly 5000 times the concentration of
aldrin in the water in which the snails live. The more a chemical
persists, the greater the potential for bioaccumulation. Chemicals in
the environment can become increasingly concentrated in the tissues of
animals higher up in the food-chain, a process known as
biomagnification.
The octanol/water partition coefficient of a chemical (see Chapter 1
for a discussion of this value) is a good indicator of its
bioaccumulation potential: the higher the coefficient, the more a
chemical will tend to bioaccumulate.
* Toxicity. The impact of a chemical on the environment,
resulting from its toxicity, is more complex. Chemical
pollutants affect some environmental media more than others,
whether these are air, surface water, groundwater, soil or
sediments. As explained in Chapter 2, different species will
react in varying ways and to different degrees to the same
chemical. What may be highly toxic to aquatic life may not be
toxic to birds, for instance, even at the same dose. Toxicity
forms the basis of use of pesticides. Pesticides are applied in
order to damage or kill a target organism (i.e. the organism that
is subject to the action of the pesticide). But it is seldom or
never possible to limit the effects to target organisms.
Pesticides are therefore often perceived as a particular threat
to health and the environment.
By determining the effects of a chemical on different species,
toxicity studies are able to evaluate the environmental impact of that
chemical. The effects to be determined should include acute lethality
(oral, dermal, inhalation LD50 and LC50); sublethal effects on
non-mammals (soil organisms, bees, birds, etc.), mammals and plants
(usually expressed as the percentage of growth reduction as a result
of exposure to the chemical in water, air or soil); mutagenicity/
genotoxicity; teratogenicity; and carcinogenicity.
5.4 The importance of prevention
Once chemicals have been released into the environment, little or
nothing can be done to remove them. Many chemicals will continue to
damage animals and plants and poison food and water for years or
decades. Therefore, it is essential that chemical pollution of the
environment be prevented at its source -- i.e., wherever chemicals are
manufactured or used. Good occupational hygiene principles should
always be applied when using or handling any chemicals at work.
Chemicals must be controlled not simply in order to ensure compliance
with government standards (exposure limits, etc.; see Chapter 7), but
to prevent chemical contamination -- to the maximum extent possible --
of the general environment. In the first instance, this means
judicious use of less toxic and/or more readily degradable chemicals
and the use of more selective pesticides. Chemical processes should
be enclosed as far as possible to prevent release of chemicals as
dust, fumes or gas. Dust, fumes and gas that are generated during a
process such as grinding or welding should be captured by local
exhaust ventilation. Contaminated air from such processes, together
with contaminated air from ventilation of closed systems and general
ventilation, should not be allowed to escape to the atmosphere inside
the workplace, or outside, without first having had the contamination
removed. Some methods and principles for the prevention of
environmental chemical pollution are given in Chapter 4 of this Manual
(see sections on Storage, Spillage and Disposal).
6. SUMMARY OF CHEMICAL SAFETY INFORMATION - CHAPTER 6
Most HSGs contain a Summary of Chemical Safety Information or an ICSC
that summarizes information and advice on the relevant chemical. It
is designed for easy, quick reference for health workers concerned
with, and users of, chemicals. It should be displayed near entrances
to areas where exposure to the chemical in question could potentially
occur and, if possible, on processing equipment and containers.
A sample is given below; the terms used are defined in Chapters 1 and
2 of this Manual. All persons potentially exposed to chemicals should
have the instructions on the relevant ICSCs clearly explained to them.
1,4-DIOXANE ICSC: 0041
1,4-DIOXANE
1,4-Diethylene dioxide
1,4-Diethyleneoxide
C4H8O2
Molecular mass: 88.1
 CAS #
CAS #  Figure 55. Labelling requirements (from HSG 9: Isobutanol)
Figure 55. Labelling requirements (from HSG 9: Isobutanol)
 The European Community legislation requires labelling as a
dangerous substance using the symbol:
The European Community legislation requires labelling as a
dangerous substance using the symbol:
 The label must read: flammable - harmful by inhalation; keep
away from sources of ignition - no smoking.
The European Community legislation on labelling of solvent
preparations classifies isobutanol in class II d for the
purpose of determining the label for preparations containing
isobutanol and other active ingredients (1980).
The criteria for classification are too complex for inclusion here.
In addition, there are many different classification and labelling
systems that depend on the user, i.e. the manufacturer, the shippers
or the employers, all of whom will have different areas of interest --
consumer products, shipping or environmental regulations or employee
protection. All of these are regulated by different national and
international agencies (national departments of transportation,
environmental protection agencies), local ordinances, the United
Nations Committee of Experts on the Transport of Dangerous Goods and
the International Maritime Organization.
It is generally agreed that proper application -- and therefore proper
interpretation -- of labelling of chemicals is often a difficult and
frustrating task. End-users are therefore advised to contact their
trade union or appropriate national or international association for
basic information on specific labelling.
8. GETTING FURTHER INFORMATION - CHAPTER 8
The information available to workers (and the general public) about
the health and safety effects of chemicals is often inadequate. Some
suggestions for obtaining further information are given below.
I. Identify and list the chemicals used in your workplace
II. Approach your employer for full information
It is the employer's responsibility to provide employees with full
health and safety information for all chemicals and chemical processes
used in a workplace. You should always ask your employer for health
and safety information as a first step.
You may wish to do this through your trade union or through the
workplace health and safety committee, if one exists. Always put your
requests in writing. If the employer's reply is inadequate, you
should inform your trade union.
III. Contact the chemical manufacturer or supplier
If your employer is unwilling or unable to provide the requested
information, it will be necessary to write to the chemical
manufacturer or supplier. Its name and address usually appear on the
label of the chemical container.
The manufacturer's or supplier's reply should include the following
information:
* the manufacturer's trade name for the chemical;
* any code numbers that appear on the label;
* the proper chemical or generic names of the chemical, if
available; and
* a brief description of how the chemical is used.
IV. Contact your government's Department of Occupational Health
and Safety
The government department responsible for occupational health and
safety may provide an information service that can assist you in
obtaining further information about particular chemicals.
V. Contact your trade union
There may be services concerned with the safety of chemicals run by
public interest groups or the trade union movement.
VI. Search for the information yourself
Many large public libraries and most university or technical college
libraries hold reference books on occupational health and safety and
the hazards of chemicals. Ask the librarian for help. The following
books may be particularly useful:
* IPCS Health and Safety Guides;
* IPCS Environmental Health Criteria (more detailed scientific
information on each chemical);
* ILO Encyclopaedia of Occupational Health and Safety;
* Reports from the International Register of Potentially Toxic
Chemicals (IRPTC/UNEP);
* Dangerous Properties of Industrial Materials -- Author: I. Sax;
* Patty's Industrial Hygiene and Toxicology;
* UN "Consolidated list of products whose consumption and/or sale
have been banned, withdrawn, severely restricted or not approved
by governments". Fourth ed. New York, United Nations Department
of International Economic and Social Affairs, 1991;
* ILO Convention No. 170 and Recommendation No. 177 on Safety in
the Use of Chemicals at Work;
* ILO Convention No. 174 and Recommendation No. 181 on Prevention
of Major Industrial Accidents;
* ILO Codes of Practice on "Safety in the Use of Chemicals at Work"
and "Prevention of Major Industrial Accidents"; and
* ILO. Safety and health in the use of chemicals at work: a
training manual.
Information on toxic effects of chemicals can also be obtained from
several databases, some of which are available on electronic media
such as CD-ROM. Many national and international trade unions have
access to such electronic information and can readily respond to
requests from affiliated unions and members.
The label must read: flammable - harmful by inhalation; keep
away from sources of ignition - no smoking.
The European Community legislation on labelling of solvent
preparations classifies isobutanol in class II d for the
purpose of determining the label for preparations containing
isobutanol and other active ingredients (1980).
The criteria for classification are too complex for inclusion here.
In addition, there are many different classification and labelling
systems that depend on the user, i.e. the manufacturer, the shippers
or the employers, all of whom will have different areas of interest --
consumer products, shipping or environmental regulations or employee
protection. All of these are regulated by different national and
international agencies (national departments of transportation,
environmental protection agencies), local ordinances, the United
Nations Committee of Experts on the Transport of Dangerous Goods and
the International Maritime Organization.
It is generally agreed that proper application -- and therefore proper
interpretation -- of labelling of chemicals is often a difficult and
frustrating task. End-users are therefore advised to contact their
trade union or appropriate national or international association for
basic information on specific labelling.
8. GETTING FURTHER INFORMATION - CHAPTER 8
The information available to workers (and the general public) about
the health and safety effects of chemicals is often inadequate. Some
suggestions for obtaining further information are given below.
I. Identify and list the chemicals used in your workplace
II. Approach your employer for full information
It is the employer's responsibility to provide employees with full
health and safety information for all chemicals and chemical processes
used in a workplace. You should always ask your employer for health
and safety information as a first step.
You may wish to do this through your trade union or through the
workplace health and safety committee, if one exists. Always put your
requests in writing. If the employer's reply is inadequate, you
should inform your trade union.
III. Contact the chemical manufacturer or supplier
If your employer is unwilling or unable to provide the requested
information, it will be necessary to write to the chemical
manufacturer or supplier. Its name and address usually appear on the
label of the chemical container.
The manufacturer's or supplier's reply should include the following
information:
* the manufacturer's trade name for the chemical;
* any code numbers that appear on the label;
* the proper chemical or generic names of the chemical, if
available; and
* a brief description of how the chemical is used.
IV. Contact your government's Department of Occupational Health
and Safety
The government department responsible for occupational health and
safety may provide an information service that can assist you in
obtaining further information about particular chemicals.
V. Contact your trade union
There may be services concerned with the safety of chemicals run by
public interest groups or the trade union movement.
VI. Search for the information yourself
Many large public libraries and most university or technical college
libraries hold reference books on occupational health and safety and
the hazards of chemicals. Ask the librarian for help. The following
books may be particularly useful:
* IPCS Health and Safety Guides;
* IPCS Environmental Health Criteria (more detailed scientific
information on each chemical);
* ILO Encyclopaedia of Occupational Health and Safety;
* Reports from the International Register of Potentially Toxic
Chemicals (IRPTC/UNEP);
* Dangerous Properties of Industrial Materials -- Author: I. Sax;
* Patty's Industrial Hygiene and Toxicology;
* UN "Consolidated list of products whose consumption and/or sale
have been banned, withdrawn, severely restricted or not approved
by governments". Fourth ed. New York, United Nations Department
of International Economic and Social Affairs, 1991;
* ILO Convention No. 170 and Recommendation No. 177 on Safety in
the Use of Chemicals at Work;
* ILO Convention No. 174 and Recommendation No. 181 on Prevention
of Major Industrial Accidents;
* ILO Codes of Practice on "Safety in the Use of Chemicals at Work"
and "Prevention of Major Industrial Accidents"; and
* ILO. Safety and health in the use of chemicals at work: a
training manual.
Information on toxic effects of chemicals can also be obtained from
several databases, some of which are available on electronic media
such as CD-ROM. Many national and international trade unions have
access to such electronic information and can readily respond to
requests from affiliated unions and members.
