
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 75
METHYL PARATHION
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1992
This is a companion volume to Environmental Health Criteria
145: Methyl Parathion
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the
United Nations Environment Programme, the International Labour
Organisation, and the World Health Organization)
This report contains the collective views of an international group
of experts and does not necessarily represent the decisions or the
stated policy of the United Nations Environment Programme, the
International Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Methyl parathion : health and safety guide.
(Health and safety guide ; no. 75)
1.Methyl parathion - poisoning 2.Methyl parathion - standards
3.Methyl parathion - toxicity 4.Environmental exposure
I.Series
ISBN 92 4 151075 7 (NLM Classification: WA 240)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1992
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Environmental exposure
2.2. Uptake, metabolism, and excretion
2.3. Effects on organisms in the environment
2.4. Effects on experimental animals and in vitro
test systems
2.5. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.2. Recommendations
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.1.1 Symptoms of poisoning
4.1.1.2 Medical treatment
4.1.1.3 Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
6.1. Previous evaluations by international bodies
6.2. Exposure limit values
6.3. Specific restrictions
6.4. Labelling, packaging, and transport
6.5. Waste disposal
6.6. Other measures
BIBLIOGRAPHY
ANNEX 1
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health from exposure to
a chemical or combinations of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding
EHC. Section 4 includes advice on preventive and protective measures
and emergency action; health workers should be thoroughly familiar
with the medical information to ensure that they can act efficiently
in an emergency. The section on regulatory information has been
extracted from the legal file of the International Register of
Potentially Toxic Chemicals (IRPTC) and from other United Nations
sources.
The target readership includes occupational health services, those
in ministries, governmental agencies, industry, and trade unions who
are involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
1. PRODUCT IDENTITY AND USES
1.1 Identity
Chemical structure:
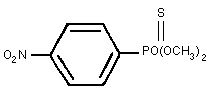 Common names: parathion-methyl, methyl parathion,
metaphos (USSR)
Molecular formula: C8H10NO5PS
IUPAC name: O,O-dimethyl O-4-nitrophenyl
phosphorothioate
CAS name: O,O-dimethyl O-(4-nitrophenyl)
phosphorothioate (9CI)
CAS registry number: 298-00-0
OMS number: 213
RTECS registry number: TG0175000
1.2 Physical and chemical properties
Pure methyl parathion is crystalline and white. It is relatively
insoluble in water, poorly soluble in petroleum ether and mineral
oils, and readily soluble in most organic solvents. Methyl parathion
is stable at pH 1-7, but undergoes rapid decomposition in alkaline
media at pH 8-9. On heating, methyl parathion readily isomerizes to
the O,S-dimethyl analogue.
Technical methyl parathion, which is at least 90% pure, is a light
to dark tan liquid.
Some physical properties of methyl parathion are given in Table 1.
Table 1. Physical properties
Relative molecular mass 263.2
Melting point (°C) 35-38
Boiling point (°C) 143
Density d 25 1.358
4
Vapour pressure (20 °C) 1.3 mPa
Water solubility (25 °C) 55-60 mg/litre
Partition coefficient (log Pow) 1.81-3.43 (reported range)
Conversion factors 1 ppm = 10.76 mg/m3
(25 °C, 1066 mbar): 1 mg/m3 = 0.0929 ppm.
1.3 Analytical methods
The active ingredient of the formulated product is determined using
gas chromatography, high pressure liquid chromatography, or
hydrolysis to 4-nitrophenol followed by colorimetric determination.
Similar methods and thin-layer chromatography are available for
residue analysis. FAO/WHO recommended methods for the analysis of
methyl parathion residues are given in FAO/WHO (1989).
1.4 Production and uses
Methyl parathion is produced throughout the world and has been
registered for use on many crops. It is a non-systemic insecticide
that controls numerous insects by contact and stomach action. It is
generally applied as a spray, mainly from the emulsifiable
concentrate formulation. It is recommended for application after
mixing at 15-25 g active ingredient (a.i.) per 100 litres. In some
countries, the dust and dispersible powder formulations are also
available. On a global basis, the available emulsifiable
concentrates contain 400, 480, and 600 g a.i./litre. The wettable
powder contains 40% a.i., while the dust formulations contain 1.5,
2, and 3% a.i.
2. SUMMARY AND EVALUATION
2.1 Environmental exposure
The distribution of methyl parathion in air, water, soil, and in
organisms in the environment is influenced by several physical,
chemical, and biological factors.
Studies using model ecosystems and mathematical modelling indicate
that methyl parathion partitions mainly to air and soil in the
environment, with lesser amounts in plants and animals. There is
virtually no movement through soil and neither the parent compound
nor its breakdown products will reach ground water. Methyl parathion
in air is mainly derived from spraying of the compound, though some
volatilization occurs with the evaporation of water from leaves and
the soil surface. Background atmospheric levels of methyl parathion
in agricultural areas range from not detectable to about 70 ng/m3.
Air concentrations after spraying declined rapidly over 3 days and
returned to background levels after about 9 days. Levels in river
water (in laboratory studies) declined to 80% of the initial
concentration after 1 h and 10% after 1 week. Methyl parathion is
retained longer in soil than in air or water, though retention is
greatly influenced by soil type; sandy soil can lose residues of the
compound more rapidly than loams. Residues on plant surfaces and
within leaves decline rapidly with half-lives of the order of a few
hours; complete loss of the methyl parathion occurs within about 6-7
days.
Animals can degrade methyl parathion and excrete the degradation
products within a very short time. However, this occurs more slowly
in lower vertebrates and invertebrates than in mammals and birds.
Bioconcentration factors are low and the accumulated methyl
parathion levels transitory.
By far the most important route for the environmental degradation of
methyl parathion is microbial degradation. Loss of the compound in
the field and in model ecosystems is more rapid than predicted from
laboratory studies. This is because of the presence of a variety of
microorganisms capable of degrading the compound in different
habitats and circumstances. When sediment or plant surfaces are
present, the microbial populations increase with a resulting
increase in the rate of breakdown of methyl parathion.
Methyl parathion can undergo oxidative degradation, to the less
stable methylparaoxon, in the presence of ultraviolet radiation
(UVR) or sunlight; sprayed films degrade under UVR with a half-life
of about 40 h. However, the contribution of photolysis to total loss
in an aquatic system was estimated to be only 4%. Hydrolysis of
methyl parathion also occurs and is more rapid under alkaline
conditions. High salinity also favours hydrolysis of the compound.
Half-lives of a few minutes were recorded in strongly reducing
sediments though methyl parathion is more stable when sorbed on
other sediments.
In towns in the centre of agricultural areas of the USA, methyl
parathion concentrations in air varied with season and peaked in
August or September; maximum levels in surveys were mainly in the
range of 100-800 ng/m3, during the growing season. Concentrations
in natural waters of agricultural areas in the USA ranged up to
0.46 µg/litre, with highest levels in summer. There is only a small
number of published reports on the residues of methyl parathion in
food throughout the world. In the USA, residues of methyl parathion
in food have generally been reported at very low levels with few
individual samples exceeding maximum residue limits (MRLs). Only
trace residue levels of methyl parathion have been detected in the
total dietary studies reported. Methyl parathion residues were
highest in leafy (up to 2 mg/kg) and root vegetables (up to 1 mg/kg)
in market basket surveys in the USA between 1966 and 1969. Food
preparation, cooking, and storage all cause decomposition of methyl
parathion residues further reducing human exposure. Raw vegetables
and fruits may contain higher residues after misuse.
The production, formulation, handling, and use of methyl parathion
as an insecticide are the main potential sources of human exposure.
Skin contact and, to a lesser degree, inhalation are the main routes
of exposure of workers. In a study of farm spray-men (with
unprotected workers and ULV hand-spray) an intake of 0.4-13 mg
methyl parathion (per 24 h) was calculated from the p-nitrophenol
excreted in the urine.
Early re-entry into treated crops is a further source of exposure.
The general population may be exposed to air-, water- and food-borne
residues of methyl parathion as a consequence of agricultural or
forestry practices, and the misuse of the agent resulting in
contamination of fields, crops, water, and air through off-target
spraying.
2.2 Uptake, metabolism, and excretion
Methyl parathion is readily absorbed via all routes of exposure
(oral, dermal, and inhalation) and is rapidly distributed to the
tissues of the body. Maximum concentrations in various organs were
detected 1-2 h after treatment. Conversion of methyl parathion to
methylparaoxon occurs within minutes following administration. In
dogs, a mean terminal half-life of 7.2 h was determined following
i.v. administration of methyl parathion. The liver is the primary
organ of metabolism and detoxification. Methyl parathion or
methylparaoxon are mainly detoxified in the liver by oxidation,
hydrolysis, and demethylation or dearylation with reduced
glutathione (GSH). The reaction products are
O-methyl- O-p-nitrophenyl phosphorothioate or dimethyl
phosphorothioic or dimethylphosphoric acids and p-nitrophenol.
Therefore, it is possible to estimate the exposure by measuring the
urinary excretion of p-nitrophenol. The urinary excretion of
p-nitrophenol was 60% within 4 h and approximately 100% within
24 h. The metabolism of methyl parathion is important for species
selective toxicity, and the development of resistance. The
elimination of methyl parathion and metabolic products occurs
primarily via the urine. Studies conducted with radiolabelled
32P-methyl parathion revealed, after 72 h, 75% of radioactivity in
the urine and up to 10% radioactivity in the faeces.
2.3 Effects on organisms in the environment
Microorganisms can use methyl parathion as a carbon source and
studies on a natural community showed that concentrations of up to
5 mg/litre increased biomass and reproductive activity. Bacteria and
actinomycetes showed a positive effect of methyl parathion while
fungi and yeasts were less able to utilize the compound. A 50%
inhibition of growth of a diatom occurred at about 5 mg/litre. Cell
growth of unicellular green algae was reduced by between 25 and
80 µg methyl parathion/litre. Populations of algae became tolerant
after exposure for several weeks.
Methyl parathion is highly toxic for aquatic invertebrates, most
LC50s ranging from <1 µg to about 40 µg/litre. A few arthropod
species are less susceptible. The no-observed-effect level for the
water flea (Daphnia magna) is 1.2 µg/litre. Molluscs are much less
susceptible with LC50s ranging between 12 and 25 mg/litre.
Most fish species in both fresh and sea water have LC50s between 6
and 25 mg/litre, a few species being substantially more or less
sensitive to methyl parathion. The acute toxicity of amphibians is
similar to that of fish.
Population effects have been seen in communities of aquatic
invertebrates in experimental ponds treated with methyl parathion.
The concentrations needed to cause these effects would occur only
with overspraying of water bodies and, even then, would last for
only a short time. Population effects are, therefore, unlikely to be
seen in the field. Kills of aquatic invertebrates would be unlikely
to lead to lasting effects.
Care should be taken to avoid the overspraying of ponds, rivers, and
lakes, when using methyl parathion. The compound should never be
sprayed in windy conditions.
Methyl parathion is a non-selective insecticide that kills
beneficial species as readily as pests. Kills of bees have been
reported following the spraying of methyl parathion. Effects on bees
in methyl parathion incidents were more severe than those of other
insecticides. Africanized honey bees are more tolerant of methyl
parathion than European strains.
Methyl parathion was moderately toxic for birds in laboratory
studies, acute oral LD50s ranging between 3 and 8 mg/kg body
weight. Dietary LC50s ranged from 70 to 680 mg/kg diet. There is
no indication that birds would be adversely affected from the
recommended usage of methyl parathion in the field.
Extreme care must be taken to time methyl parathion spraying to
avoid adverse effects on honey bees.
2.4 Effects on experimental animals and in vitro test systems
Oral LD50 values of methyl parathion in rodents range from 3 to
35 mg/kg body weight, and dermal LD50 values from 44 to 67 mg/kg
body weight.
Methyl parathion poisoning causes the usual organophosphate
cholinergic signs attributed to accumulation of acetylcholine at
nerve endings. Methyl parathion becomes toxic when it is metabolized
to methylparaoxon. This conversion is very rapid. No indications of
organophosphorus-induced delayed neuropathy (OPIDN) have been
observed.
Technical methyl parathion was found not to have any primary eye or
skin irritating potential.
In short-term toxicity studies using various routes of
administration on the rat, dog, and rabbit, inhibition of plasma,
red blood cells, and brain ChE and related cholinergic signs were
observed. In a 12-week feeding study on dogs, the
no-observed-adverse-effect level (NOAEL) was 5 mg/kg diet
(equivalent to 0.1 mg/kg body weight per day). In a 3-week dermal
toxicity study on rabbits, the no-observed-effect level (NOEL) was
10 mg/kg body weight per day. Inhalation exposure for 3weeks
indicated a NOEL of 0.9 mg/m3 air. At 2.6 mg/m3, only slight
inhibition of plasma ChE was observed.
Long-term toxicity/carcinogenicity studies were carried out on mice
and rats. The NOEL in rats was 0.1 mg/kg body weight per day, based
on ChE inhibition. There is no evidence of carcinogenicity in mice
and rats, following long-term exposure. In another 2-year study on
rats, however, there was evidence of a peripheral neurotoxic effect
at a dose of 50 mg/kg diet.
Methyl parathion has been reported to have DNA alkylating properties
in vitro.
Most of the results of in vitro genotoxicity studies on both
bacterial and mammalian cells were positive, while 6 in vivo
studies using 3 different test systems produced equivocal results.
In reproduction studies, at toxic dose levels (ChE inhibition),
there were no consistent effects on litter sizes, number of litters,
survival rates of pups, and lactation performance. No primary
teratogenic or embryotoxic effects were noted.
2.5 Effects on human beings
Several cases of acute methyl parathion poisoning have been
reported. Signs and symptoms are those characteristic of systemic
poisoning by cholinesterase-inhibiting organophosphorus compounds.
They include peripheral and central cholinergic nervous system
manifestations appearing as rapidly as a few minutes after exposure.
In case of dermal exposure, symptoms may increase in severity for
more than one day and may last for several days.
The results of studies on volunteers, following repeated long-term
exposures, suggest that there is a decrease in blood cholinesterase
activity without clinical manifestations.
No cases of organophosphorus-induced delayed peripheral neuropathy
(OPIDN) have been reported. Neuropsychiatric sequelae have been
reported in cases of multiple exposures to pesticides including
methyl parathion.
An increase in chromosomal aberrations has been reported in cases of
acute intoxication.
No human data were available to evaluate the teratogenic and
reproductive effects of methyl parathion.
The available epidemiological studies deal with multiple exposures
to pesticides and it has not been possible to evaluate the effects
of long-term exposure to methyl parathion.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Methyl parathion is a highly toxic organophosphorus ester
insecticide. Overexposure from handling during manufacture or use,
and/or accidental or intentional ingestion may cause severe or fatal
poisoning. Methyl parathion formulations may, or may not, be
irritating to the eyes or the skin, but they are readily absorbed.
As a consequence, hazardous exposures may occur without warning.
Methyl parathion is not persistent in the environment. It is not
bioconcentrated and is not transferred through food-chains and is
degraded rapidly by many microorganisms and other forms of wildlife.
It is only likely to cause damage to ecosystems in instances of
heavy overexposure resulting from misuse or accidental spills.
However, pollinators and other beneficial insects are at risk from
spraying with methyl parathion.
Exposure of the general population to methyl parathion residues
occurs predominantly via food. If good agricultural practice is
followed, the Acceptable Daily Intake (0-0.02 mg/kg body weight)
established by FAO/WHO will not be exceeded. Dermal exposure may
occur through accidental contact with foliar residues in sprayed
fields or in areas adjacent to spraying operations as a consequence
of off-target spraying of the chemical.
With good work practices, hygienic measures and safety precautions,
methyl parathion is unlikely to present a hazard for those
occupationally exposed.
3.2 Recommendations
* For the health and welfare of workers and the general
population, the handling and application of methyl parathion
should be entrusted only to competently supervised and
well-trained applicators, who must follow adequate safety
measures and use the chemical according to good application
practices.
* The manufacture, formulation, agricultural use, and disposal of
methyl parathion should be carefully managed to minimize
contamination of the environment.
* Regularly exposed workers should undergo appropriate monitoring
and health evaluations.
* To minimize risks for all individuals, a 48-h interval between
spraying and re-entry into any sprayed area is recommended.
* Pre-harvest intervals should be established and enforced by
national authorities.
* In view of its high toxicity, methyl parathion should not be
considered for use in hand-applied, ultra-low-volume (ULV)
spraying practices.
* Do not overspray water bodies. Choose spraying time to avoid
killing pollinating insects.
* Information on the health status of workers exposed only to
methyl parathion (i.e., in its manufacture and formulation)
should be published, in order to assist in a better evaluation
of the risks of this chemical for human health.
* More definitive studies should be conducted on the residues of
methyl parathion in fresh foods.
* A more definitive genotoxic assessment of methyl parathion
should be conducted.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human health hazards, prevention and protection, first aid
The acute oral and dermal toxicities of methyl parathion are high
and it is hazardous for human beings if incorrectly handled. With
excessive exposure, typical signs and symptoms of organophosphorus
poisoning may occur rapidly. The human health hazards associated
with certain types of exposure to methyl parathion, together with
preventive and protective measures and first aid, are listed in the
Table 2.
4.1.1 Advice to physicians
For a more complete treatise on the effects of organophosphorus
insecticides, especially their short- and long-term effects on the
nervous system, please refer to EHC 63: Organophosphorus
insecticides - a general introduction (WHO, 1986).
Methyl parathion is an indirect cholinesterase inhibitor in that the
active cholinesterase inhibitor, methyl paraoxon, is formed in the
liver. Signs and symptoms of toxicity appear rapidly.
4.1.1.1 Symptoms of poisoning
Signs and symptoms may include a feeling of exhaustion, headache,
blurred vision, weakness, and confusion. Vomiting, abdominal pain,
excessive sweating, and salivating may develop. The pupils may also
be constricted. Difficulty in breathing may be experienced, due to
congestion of the lungs and weakness of the respiratory muscles.
Arrhythmias and cardiac failure have been reported. On severe
poisoning, there will be muscle spasms, unconsciousness, and
convulsions. Breathing may stop, followed by death.
Table 2. Human health hazards, preventive and protective measures, first aid
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Cholinesterase Avoid exposure
inhibitor
SKIN: Contamination may cause Wear PVC or neoprene gloves, Wash contaminated skin with soap and
organophosphate poisoning: weakness, apron and rubber boots water; remove contaminated clothing
headache, vomiting, excessive sweating and launder before reuse; obtain
and salivation, pin-point pupils; in medical attention immediately
severe cases: convulsions,
unconsciousness and death due to
respiratory paralysis may occur
EYES: Irritation, redness Wear face shield or goggles Flush immediately with clean water
for at least 15 min; if irritation persists,
obtain medical attention immediately;
launder contaminated clothing
INHALATION: Inhalation may Avoid breathing mist or dust; use In case of signs of symptoms, remove
cause poisoning (see skin) proper (exhaust) ventilation or from contaminated area and obtain
suitable respiratory protection medical attention immediately
INGESTION: Unlikely to be a Wash hands before eating, drinking,
significant occupational hazard using the toilet, and after work;
do not keep food in areas with
potential exposure
Accidental or deliberate ingestion Obtain medical attention immediately;
may rapidly lead to severe poisoning induce vomiting, if subject is conscious,
(see skin) except in the case of an emulsifiable
concentrate; if breathing has stopped
apply artificial respiration
4.1.1.2 Medical treatment
If ingested and the formulation does not contain petroleum
distillates, induce vomiting, or preferably perform gastric lavage
using 5% sodium bicarbonate. In the case of ingestion of liquid
formulations containing hydrocarbon solvents, vomiting involves a
risk of aspiration pneumonia. Instead, the stomach should be emptied
as soon as possible by careful gastric lavage (using a cuffed
endotracheal tube). If possible, identify the solvents present in
the formulation and observe the victim for additional toxic effects.
As early as possible, administer 2 mg atropine sulfate intravenously
(i.v.) and 1000-2000 mg of pralidoxime chloride or 250 mg of
obidoxime chloride (adult dose) i.v., to patients suffering from
severe respiratory difficulties, convulsions, and unconsciousness.
Repeated doses of 2 mg of atropine sulfate should be given, as
required, based on the respiration, blood pressure, pulse frequency,
salivation, and convulsion conditions. Diazepam should be given in
all but the mildest cases in doses of 10 mg s.c. or i.v., repeated
as required.
For children, the doses are 0.04-0.08 mg atropine/kg body weight,
250 mg pralidoxime chloride per child, or 4-8 mg obidoxime chloride
per kg body weight.
Artificial respiration should be applied, if required.
Note: Contraindications: morphine, barbiturates,
phenothiazine derivatives, tranquillizers, and all
kinds of central stimulants
The diagnosis of intoxication should be confirmed as soon as
possible by determination of the cholinesterase activity in venous
blood.
For more information on the treatment of organophosphorus
insecticides see EHC 63: Organophosphorus insecticides - a general
introduction (WHO, 1986). The section from this publication on
therapy is attached as Annex 1 of this guide.
4.1.1.3 Health surveillance advice
In human beings exposed to methyl parathion, the cholinesterase
activity of the blood should be monitored regularly. Measurement of
whole blood AChE is the most widely adopted method. Because
physiological variations of blood ChE levels occur in a healthy
person and amongst populations, results should preferably be
compared with pre-exposure ChE levels.
Depressions of AChE or ChE of 20-25% are considered diagnostic of
exposure, but not necessarily indicative of hazard. Depressions of
30-50% or more are considered indicators for removal of an exposed
individual from further contact with pesticides until levels return
to normal. Work procedures and hygiene should also be checked.
Exposure can also be monitored by measuring the urinary excretion of
p-nitrophenol.
4.2 Explosion and fire hazards
Liquid formulations may be flammable. With sufficient burning or
external heat, methyl parathion will decompose, emitting toxic
fumes. Fire-fighters must wear protective clothing and
self-contained breathing apparatus. Extinguish fires with alcohol
resistant foam or powder. Confine the use of water spray to the
cooling of unaffected stock, thus avoiding polluted run-off from the
site.
4.3 Storage
Technical methyl parathion and its formulations should be stored in
the original labelled containers in locked, well-ventilated storage
areas, preferably dedicated to insecticides. Do not expose to direct
sunlight. Keep products out of reach of children and unauthorized
personnel. Do not store near food or animal feed.
4.4 Transport
Comply with any local regulations regarding movement of hazardous
goods. Do not transport with food or animal feed. Food and feed
should not be transported in vehicles that have been used for the
transport of pesticides. Make sure that containers are in good
condition and the labels are undamaged before dispatch.
4.5 Spillage and Disposal
4.5.1 Spillage
Avoid skin contamination and inhalation of vapour. Absorb spilled
liquid and cover contaminated areas with a 1:3 mixture of sodium
carbonate crystals and damp sawdust, lime, sand, or earth. Sweep up
and place it in an impervious container. Ensure that the container
is tightly closed and labelled before transfer to a safe place for
disposal.
Prevent liquid from spreading and contaminating other cargo,
vegetation, or waterways by using a barrier of the most suitable and
readily available material, e.g., earth or sand.
Empty any of the product remaining in the damaged/leaking container
into a clean empty container, which should then be tightly closed
and suitably labelled. Decontaminate emptied leaking containers with
a 10% sodium carbonate (washing soda) solution, added at a rate of
at least 1 litre/20-litre drum. Swirl round to rinse walls, empty,
and add rinsings to sawdust. Do not reuse containers for any other
purpose. Puncture and crush the container to prevent reuse.
4.5.2 Disposal
Large amounts should be incinerated at high temperature in a unit
with effluent gas scrubbing or should be adsorbed on vermiculite and
disposed of in a landfill, if incineration is impossible. When no
incinerator is available, bury in an approved dump, or in an area
where there is no risk of contamination of surface or ground water.
Before burying, mix liberally with sodium carbonate (washing soda)
crystals to help neutralize the product and with soil rich in
organic matter. Comply with any local legislation.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Methyl parathion is readily degraded and non-persistent in the
environment. It is highly toxic for aquatic invertebrates, birds,
bees, and wild mammals. It is moderately toxic for fish and
non-toxic for soil microorganisms.
Do not overspray water; time spraying to avoid killing of
pollinators.
Avoid contamination of soil, water, and the atmosphere by proper
methods of use, storage, transport, handling, and waste disposal. In
case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
6.1 Previous evaluations by international bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) evaluated
methyl parathion in 1968, 1972, 1975, 1979, 1980, and 1984 (FAO/WHO,
1969, 1973, 1976, 1980, 1981, and 1985). The acceptable daily intake
for humans (ADI) was estimated at 0-0.02 mg/kg body weight in 1984.
This was based on the following levels causing no toxicological
effects:
- 2 mg/kg diet, equivalent to 0.1 mg/kg body weight in the rat;
and
- 0.3 mg/kg body weight per day in man.
The FAO/WHO Codex Alimentarius Commission (FAO/WHO, 1986)
recommended Maximum Residue Limits (MRLs) in several food
commodities, ranging from 0.05 to 0.2 mg/kg, including:
Commodity MRL (mg/kg) Commodity MRL (mg/kg)
cantaloupes 0.2 hops (dry cones) 0.05a
cole crops 0.2 melons 0.2
cottonseed oil 0.05 sugar beets 0.05a
cucumbers 0.2 tea (fermented and dried) 0.2
fruit, other 0.2 tomatoes 0.2
a Levels at or about the limit of determination.
The International Agency for Research on Cancer (IARC) classified
methyl parathion in Group 3 in 1982 and in 1987 (IARC, 1983, vol.
30, 1987, suppl. 7), and concluded that the available data do not
provide evidence that methyl parathion is carcinogenic to
experimental animals. No data on humans were available. The
available data provide no evidence that methyl parathion is likely
to present a carcinogenic risk to humans.
WHO (1990) classified technical methyl parathion as "extremely
hazardous" in normal use, on the basis of the oral LD50 in the rat
of 14 mg/kg. WHO/FAO (1975) issued a data sheet on methyl parathion
(No. 7).
6.2 Exposure limit values
Some exposure limit values are given on pp. 28-29
.
Pre-harvest intervals (the time between the last application of
methyl parathion and the harvest of the treated plants) have been
set in many countries. These intervals vary from 1 to 60 days (most
of them between 14 and 21 days), depending on the crop, the
harvesting technique, and the country, and can be verified with the
competent national authority.
6.3 Specific restrictions
In many countries where methyl parathion is approved as a pesticide,
specific uses, restrictions, and precautions are listed in national
regulatory documents, e.g., no liquid methyl parathion formulations
have been approved for registration in Finland because of their high
acute toxicity. In the USSR, the agricultural use of methyl
parathion has been restricted. In Hungary, it may only be applied in
agriculture by properly trained staff, using protective equipment.
Methyl parathion has either not been registered or has been banned
in: Bangladesh, Belgium, Bulgaria, Canada, China, Ecuador, Egypt,
Hong Kong, Ireland, Japan, Sri Lanka, and the United Kingdom.
In Japan and the USA, methyl parathion and its preparations may only
be handled by certified operators. In Germany, it may not be handled
by adolescents or pregnant and nursing women.
6.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies methyl parathion in:
- Hazard Class 6.1: poisonous substance;
- Packing Group 2: substances and preparations presenting a
serious risk of poisoning, for formulations containing
12-100% methyl parathion.
- Packing Group 3: harmful substances and preparations
presenting a serious risk of poisoning, for solid
formulations containing 3-12% active material, and liquid
formulations containing 1.2-12% active material.
Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Argentina Maximum permissible concentration
- Time-weighted average (TWA) a 0.2 mg/m3 1984
- Short-term exposure limit (STEL) 0.6 mg/m3
Finland Maximum permissible concentration
- Time-weighted average (TWA) 0.2 mg/m3 1987
- Short-term exposure limit 0.6 mg/m3
United Kingdom Occupational exposure standard (TWA) a 0.2 mg/m3 1989
- Short-term exposure limit (STEL) 0.6 mg/m3
USA (ACGIH) Threshold limit value (TLV)
- Time-weighted average (TWA) 0.2 mg/m3 b 1986
- Short-term exposure limit deleted
(STEL = 10 minutes)
USSR Maximum allowable concentration (MAC)
- Ceiling value (vapour + aerosol) 0.1 mg/m3 1977
AIR Ambient USSR Maximum allowable concentration (MAC) 0.008 mg/m3 1984
(once per day)
FOOD Uptake from FAO/WHO Acceptable daily intake (ADI) 0-0.02 mg/kg 1984
body weight
Residues FAO/WHO Maximum residue limit (MRL) 0.05-0.2 mg/kg 1986
(in specified products)
Exposure limit values (continued)
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Ambient Japan Environmental water quality standard not detectable 1981
Surface USSR Maximum acceptable concentration 0.02 mg/litre 1983
Effluent Japan Emission of total organophosphorus 1 mg/litre 1981
compounds
SOIL USSR Maximum acceptable concentration 0.1 mg/kg 1981
a TWA usually 8 h.
b Skin absorption possible.
The label should be as follows:
Packaging Group I and II:
Common names: parathion-methyl, methyl parathion,
metaphos (USSR)
Molecular formula: C8H10NO5PS
IUPAC name: O,O-dimethyl O-4-nitrophenyl
phosphorothioate
CAS name: O,O-dimethyl O-(4-nitrophenyl)
phosphorothioate (9CI)
CAS registry number: 298-00-0
OMS number: 213
RTECS registry number: TG0175000
1.2 Physical and chemical properties
Pure methyl parathion is crystalline and white. It is relatively
insoluble in water, poorly soluble in petroleum ether and mineral
oils, and readily soluble in most organic solvents. Methyl parathion
is stable at pH 1-7, but undergoes rapid decomposition in alkaline
media at pH 8-9. On heating, methyl parathion readily isomerizes to
the O,S-dimethyl analogue.
Technical methyl parathion, which is at least 90% pure, is a light
to dark tan liquid.
Some physical properties of methyl parathion are given in Table 1.
Table 1. Physical properties
Relative molecular mass 263.2
Melting point (°C) 35-38
Boiling point (°C) 143
Density d 25 1.358
4
Vapour pressure (20 °C) 1.3 mPa
Water solubility (25 °C) 55-60 mg/litre
Partition coefficient (log Pow) 1.81-3.43 (reported range)
Conversion factors 1 ppm = 10.76 mg/m3
(25 °C, 1066 mbar): 1 mg/m3 = 0.0929 ppm.
1.3 Analytical methods
The active ingredient of the formulated product is determined using
gas chromatography, high pressure liquid chromatography, or
hydrolysis to 4-nitrophenol followed by colorimetric determination.
Similar methods and thin-layer chromatography are available for
residue analysis. FAO/WHO recommended methods for the analysis of
methyl parathion residues are given in FAO/WHO (1989).
1.4 Production and uses
Methyl parathion is produced throughout the world and has been
registered for use on many crops. It is a non-systemic insecticide
that controls numerous insects by contact and stomach action. It is
generally applied as a spray, mainly from the emulsifiable
concentrate formulation. It is recommended for application after
mixing at 15-25 g active ingredient (a.i.) per 100 litres. In some
countries, the dust and dispersible powder formulations are also
available. On a global basis, the available emulsifiable
concentrates contain 400, 480, and 600 g a.i./litre. The wettable
powder contains 40% a.i., while the dust formulations contain 1.5,
2, and 3% a.i.
2. SUMMARY AND EVALUATION
2.1 Environmental exposure
The distribution of methyl parathion in air, water, soil, and in
organisms in the environment is influenced by several physical,
chemical, and biological factors.
Studies using model ecosystems and mathematical modelling indicate
that methyl parathion partitions mainly to air and soil in the
environment, with lesser amounts in plants and animals. There is
virtually no movement through soil and neither the parent compound
nor its breakdown products will reach ground water. Methyl parathion
in air is mainly derived from spraying of the compound, though some
volatilization occurs with the evaporation of water from leaves and
the soil surface. Background atmospheric levels of methyl parathion
in agricultural areas range from not detectable to about 70 ng/m3.
Air concentrations after spraying declined rapidly over 3 days and
returned to background levels after about 9 days. Levels in river
water (in laboratory studies) declined to 80% of the initial
concentration after 1 h and 10% after 1 week. Methyl parathion is
retained longer in soil than in air or water, though retention is
greatly influenced by soil type; sandy soil can lose residues of the
compound more rapidly than loams. Residues on plant surfaces and
within leaves decline rapidly with half-lives of the order of a few
hours; complete loss of the methyl parathion occurs within about 6-7
days.
Animals can degrade methyl parathion and excrete the degradation
products within a very short time. However, this occurs more slowly
in lower vertebrates and invertebrates than in mammals and birds.
Bioconcentration factors are low and the accumulated methyl
parathion levels transitory.
By far the most important route for the environmental degradation of
methyl parathion is microbial degradation. Loss of the compound in
the field and in model ecosystems is more rapid than predicted from
laboratory studies. This is because of the presence of a variety of
microorganisms capable of degrading the compound in different
habitats and circumstances. When sediment or plant surfaces are
present, the microbial populations increase with a resulting
increase in the rate of breakdown of methyl parathion.
Methyl parathion can undergo oxidative degradation, to the less
stable methylparaoxon, in the presence of ultraviolet radiation
(UVR) or sunlight; sprayed films degrade under UVR with a half-life
of about 40 h. However, the contribution of photolysis to total loss
in an aquatic system was estimated to be only 4%. Hydrolysis of
methyl parathion also occurs and is more rapid under alkaline
conditions. High salinity also favours hydrolysis of the compound.
Half-lives of a few minutes were recorded in strongly reducing
sediments though methyl parathion is more stable when sorbed on
other sediments.
In towns in the centre of agricultural areas of the USA, methyl
parathion concentrations in air varied with season and peaked in
August or September; maximum levels in surveys were mainly in the
range of 100-800 ng/m3, during the growing season. Concentrations
in natural waters of agricultural areas in the USA ranged up to
0.46 µg/litre, with highest levels in summer. There is only a small
number of published reports on the residues of methyl parathion in
food throughout the world. In the USA, residues of methyl parathion
in food have generally been reported at very low levels with few
individual samples exceeding maximum residue limits (MRLs). Only
trace residue levels of methyl parathion have been detected in the
total dietary studies reported. Methyl parathion residues were
highest in leafy (up to 2 mg/kg) and root vegetables (up to 1 mg/kg)
in market basket surveys in the USA between 1966 and 1969. Food
preparation, cooking, and storage all cause decomposition of methyl
parathion residues further reducing human exposure. Raw vegetables
and fruits may contain higher residues after misuse.
The production, formulation, handling, and use of methyl parathion
as an insecticide are the main potential sources of human exposure.
Skin contact and, to a lesser degree, inhalation are the main routes
of exposure of workers. In a study of farm spray-men (with
unprotected workers and ULV hand-spray) an intake of 0.4-13 mg
methyl parathion (per 24 h) was calculated from the p-nitrophenol
excreted in the urine.
Early re-entry into treated crops is a further source of exposure.
The general population may be exposed to air-, water- and food-borne
residues of methyl parathion as a consequence of agricultural or
forestry practices, and the misuse of the agent resulting in
contamination of fields, crops, water, and air through off-target
spraying.
2.2 Uptake, metabolism, and excretion
Methyl parathion is readily absorbed via all routes of exposure
(oral, dermal, and inhalation) and is rapidly distributed to the
tissues of the body. Maximum concentrations in various organs were
detected 1-2 h after treatment. Conversion of methyl parathion to
methylparaoxon occurs within minutes following administration. In
dogs, a mean terminal half-life of 7.2 h was determined following
i.v. administration of methyl parathion. The liver is the primary
organ of metabolism and detoxification. Methyl parathion or
methylparaoxon are mainly detoxified in the liver by oxidation,
hydrolysis, and demethylation or dearylation with reduced
glutathione (GSH). The reaction products are
O-methyl- O-p-nitrophenyl phosphorothioate or dimethyl
phosphorothioic or dimethylphosphoric acids and p-nitrophenol.
Therefore, it is possible to estimate the exposure by measuring the
urinary excretion of p-nitrophenol. The urinary excretion of
p-nitrophenol was 60% within 4 h and approximately 100% within
24 h. The metabolism of methyl parathion is important for species
selective toxicity, and the development of resistance. The
elimination of methyl parathion and metabolic products occurs
primarily via the urine. Studies conducted with radiolabelled
32P-methyl parathion revealed, after 72 h, 75% of radioactivity in
the urine and up to 10% radioactivity in the faeces.
2.3 Effects on organisms in the environment
Microorganisms can use methyl parathion as a carbon source and
studies on a natural community showed that concentrations of up to
5 mg/litre increased biomass and reproductive activity. Bacteria and
actinomycetes showed a positive effect of methyl parathion while
fungi and yeasts were less able to utilize the compound. A 50%
inhibition of growth of a diatom occurred at about 5 mg/litre. Cell
growth of unicellular green algae was reduced by between 25 and
80 µg methyl parathion/litre. Populations of algae became tolerant
after exposure for several weeks.
Methyl parathion is highly toxic for aquatic invertebrates, most
LC50s ranging from <1 µg to about 40 µg/litre. A few arthropod
species are less susceptible. The no-observed-effect level for the
water flea (Daphnia magna) is 1.2 µg/litre. Molluscs are much less
susceptible with LC50s ranging between 12 and 25 mg/litre.
Most fish species in both fresh and sea water have LC50s between 6
and 25 mg/litre, a few species being substantially more or less
sensitive to methyl parathion. The acute toxicity of amphibians is
similar to that of fish.
Population effects have been seen in communities of aquatic
invertebrates in experimental ponds treated with methyl parathion.
The concentrations needed to cause these effects would occur only
with overspraying of water bodies and, even then, would last for
only a short time. Population effects are, therefore, unlikely to be
seen in the field. Kills of aquatic invertebrates would be unlikely
to lead to lasting effects.
Care should be taken to avoid the overspraying of ponds, rivers, and
lakes, when using methyl parathion. The compound should never be
sprayed in windy conditions.
Methyl parathion is a non-selective insecticide that kills
beneficial species as readily as pests. Kills of bees have been
reported following the spraying of methyl parathion. Effects on bees
in methyl parathion incidents were more severe than those of other
insecticides. Africanized honey bees are more tolerant of methyl
parathion than European strains.
Methyl parathion was moderately toxic for birds in laboratory
studies, acute oral LD50s ranging between 3 and 8 mg/kg body
weight. Dietary LC50s ranged from 70 to 680 mg/kg diet. There is
no indication that birds would be adversely affected from the
recommended usage of methyl parathion in the field.
Extreme care must be taken to time methyl parathion spraying to
avoid adverse effects on honey bees.
2.4 Effects on experimental animals and in vitro test systems
Oral LD50 values of methyl parathion in rodents range from 3 to
35 mg/kg body weight, and dermal LD50 values from 44 to 67 mg/kg
body weight.
Methyl parathion poisoning causes the usual organophosphate
cholinergic signs attributed to accumulation of acetylcholine at
nerve endings. Methyl parathion becomes toxic when it is metabolized
to methylparaoxon. This conversion is very rapid. No indications of
organophosphorus-induced delayed neuropathy (OPIDN) have been
observed.
Technical methyl parathion was found not to have any primary eye or
skin irritating potential.
In short-term toxicity studies using various routes of
administration on the rat, dog, and rabbit, inhibition of plasma,
red blood cells, and brain ChE and related cholinergic signs were
observed. In a 12-week feeding study on dogs, the
no-observed-adverse-effect level (NOAEL) was 5 mg/kg diet
(equivalent to 0.1 mg/kg body weight per day). In a 3-week dermal
toxicity study on rabbits, the no-observed-effect level (NOEL) was
10 mg/kg body weight per day. Inhalation exposure for 3weeks
indicated a NOEL of 0.9 mg/m3 air. At 2.6 mg/m3, only slight
inhibition of plasma ChE was observed.
Long-term toxicity/carcinogenicity studies were carried out on mice
and rats. The NOEL in rats was 0.1 mg/kg body weight per day, based
on ChE inhibition. There is no evidence of carcinogenicity in mice
and rats, following long-term exposure. In another 2-year study on
rats, however, there was evidence of a peripheral neurotoxic effect
at a dose of 50 mg/kg diet.
Methyl parathion has been reported to have DNA alkylating properties
in vitro.
Most of the results of in vitro genotoxicity studies on both
bacterial and mammalian cells were positive, while 6 in vivo
studies using 3 different test systems produced equivocal results.
In reproduction studies, at toxic dose levels (ChE inhibition),
there were no consistent effects on litter sizes, number of litters,
survival rates of pups, and lactation performance. No primary
teratogenic or embryotoxic effects were noted.
2.5 Effects on human beings
Several cases of acute methyl parathion poisoning have been
reported. Signs and symptoms are those characteristic of systemic
poisoning by cholinesterase-inhibiting organophosphorus compounds.
They include peripheral and central cholinergic nervous system
manifestations appearing as rapidly as a few minutes after exposure.
In case of dermal exposure, symptoms may increase in severity for
more than one day and may last for several days.
The results of studies on volunteers, following repeated long-term
exposures, suggest that there is a decrease in blood cholinesterase
activity without clinical manifestations.
No cases of organophosphorus-induced delayed peripheral neuropathy
(OPIDN) have been reported. Neuropsychiatric sequelae have been
reported in cases of multiple exposures to pesticides including
methyl parathion.
An increase in chromosomal aberrations has been reported in cases of
acute intoxication.
No human data were available to evaluate the teratogenic and
reproductive effects of methyl parathion.
The available epidemiological studies deal with multiple exposures
to pesticides and it has not been possible to evaluate the effects
of long-term exposure to methyl parathion.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Methyl parathion is a highly toxic organophosphorus ester
insecticide. Overexposure from handling during manufacture or use,
and/or accidental or intentional ingestion may cause severe or fatal
poisoning. Methyl parathion formulations may, or may not, be
irritating to the eyes or the skin, but they are readily absorbed.
As a consequence, hazardous exposures may occur without warning.
Methyl parathion is not persistent in the environment. It is not
bioconcentrated and is not transferred through food-chains and is
degraded rapidly by many microorganisms and other forms of wildlife.
It is only likely to cause damage to ecosystems in instances of
heavy overexposure resulting from misuse or accidental spills.
However, pollinators and other beneficial insects are at risk from
spraying with methyl parathion.
Exposure of the general population to methyl parathion residues
occurs predominantly via food. If good agricultural practice is
followed, the Acceptable Daily Intake (0-0.02 mg/kg body weight)
established by FAO/WHO will not be exceeded. Dermal exposure may
occur through accidental contact with foliar residues in sprayed
fields or in areas adjacent to spraying operations as a consequence
of off-target spraying of the chemical.
With good work practices, hygienic measures and safety precautions,
methyl parathion is unlikely to present a hazard for those
occupationally exposed.
3.2 Recommendations
* For the health and welfare of workers and the general
population, the handling and application of methyl parathion
should be entrusted only to competently supervised and
well-trained applicators, who must follow adequate safety
measures and use the chemical according to good application
practices.
* The manufacture, formulation, agricultural use, and disposal of
methyl parathion should be carefully managed to minimize
contamination of the environment.
* Regularly exposed workers should undergo appropriate monitoring
and health evaluations.
* To minimize risks for all individuals, a 48-h interval between
spraying and re-entry into any sprayed area is recommended.
* Pre-harvest intervals should be established and enforced by
national authorities.
* In view of its high toxicity, methyl parathion should not be
considered for use in hand-applied, ultra-low-volume (ULV)
spraying practices.
* Do not overspray water bodies. Choose spraying time to avoid
killing pollinating insects.
* Information on the health status of workers exposed only to
methyl parathion (i.e., in its manufacture and formulation)
should be published, in order to assist in a better evaluation
of the risks of this chemical for human health.
* More definitive studies should be conducted on the residues of
methyl parathion in fresh foods.
* A more definitive genotoxic assessment of methyl parathion
should be conducted.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human health hazards, prevention and protection, first aid
The acute oral and dermal toxicities of methyl parathion are high
and it is hazardous for human beings if incorrectly handled. With
excessive exposure, typical signs and symptoms of organophosphorus
poisoning may occur rapidly. The human health hazards associated
with certain types of exposure to methyl parathion, together with
preventive and protective measures and first aid, are listed in the
Table 2.
4.1.1 Advice to physicians
For a more complete treatise on the effects of organophosphorus
insecticides, especially their short- and long-term effects on the
nervous system, please refer to EHC 63: Organophosphorus
insecticides - a general introduction (WHO, 1986).
Methyl parathion is an indirect cholinesterase inhibitor in that the
active cholinesterase inhibitor, methyl paraoxon, is formed in the
liver. Signs and symptoms of toxicity appear rapidly.
4.1.1.1 Symptoms of poisoning
Signs and symptoms may include a feeling of exhaustion, headache,
blurred vision, weakness, and confusion. Vomiting, abdominal pain,
excessive sweating, and salivating may develop. The pupils may also
be constricted. Difficulty in breathing may be experienced, due to
congestion of the lungs and weakness of the respiratory muscles.
Arrhythmias and cardiac failure have been reported. On severe
poisoning, there will be muscle spasms, unconsciousness, and
convulsions. Breathing may stop, followed by death.
Table 2. Human health hazards, preventive and protective measures, first aid
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Cholinesterase Avoid exposure
inhibitor
SKIN: Contamination may cause Wear PVC or neoprene gloves, Wash contaminated skin with soap and
organophosphate poisoning: weakness, apron and rubber boots water; remove contaminated clothing
headache, vomiting, excessive sweating and launder before reuse; obtain
and salivation, pin-point pupils; in medical attention immediately
severe cases: convulsions,
unconsciousness and death due to
respiratory paralysis may occur
EYES: Irritation, redness Wear face shield or goggles Flush immediately with clean water
for at least 15 min; if irritation persists,
obtain medical attention immediately;
launder contaminated clothing
INHALATION: Inhalation may Avoid breathing mist or dust; use In case of signs of symptoms, remove
cause poisoning (see skin) proper (exhaust) ventilation or from contaminated area and obtain
suitable respiratory protection medical attention immediately
INGESTION: Unlikely to be a Wash hands before eating, drinking,
significant occupational hazard using the toilet, and after work;
do not keep food in areas with
potential exposure
Accidental or deliberate ingestion Obtain medical attention immediately;
may rapidly lead to severe poisoning induce vomiting, if subject is conscious,
(see skin) except in the case of an emulsifiable
concentrate; if breathing has stopped
apply artificial respiration
4.1.1.2 Medical treatment
If ingested and the formulation does not contain petroleum
distillates, induce vomiting, or preferably perform gastric lavage
using 5% sodium bicarbonate. In the case of ingestion of liquid
formulations containing hydrocarbon solvents, vomiting involves a
risk of aspiration pneumonia. Instead, the stomach should be emptied
as soon as possible by careful gastric lavage (using a cuffed
endotracheal tube). If possible, identify the solvents present in
the formulation and observe the victim for additional toxic effects.
As early as possible, administer 2 mg atropine sulfate intravenously
(i.v.) and 1000-2000 mg of pralidoxime chloride or 250 mg of
obidoxime chloride (adult dose) i.v., to patients suffering from
severe respiratory difficulties, convulsions, and unconsciousness.
Repeated doses of 2 mg of atropine sulfate should be given, as
required, based on the respiration, blood pressure, pulse frequency,
salivation, and convulsion conditions. Diazepam should be given in
all but the mildest cases in doses of 10 mg s.c. or i.v., repeated
as required.
For children, the doses are 0.04-0.08 mg atropine/kg body weight,
250 mg pralidoxime chloride per child, or 4-8 mg obidoxime chloride
per kg body weight.
Artificial respiration should be applied, if required.
Note: Contraindications: morphine, barbiturates,
phenothiazine derivatives, tranquillizers, and all
kinds of central stimulants
The diagnosis of intoxication should be confirmed as soon as
possible by determination of the cholinesterase activity in venous
blood.
For more information on the treatment of organophosphorus
insecticides see EHC 63: Organophosphorus insecticides - a general
introduction (WHO, 1986). The section from this publication on
therapy is attached as Annex 1 of this guide.
4.1.1.3 Health surveillance advice
In human beings exposed to methyl parathion, the cholinesterase
activity of the blood should be monitored regularly. Measurement of
whole blood AChE is the most widely adopted method. Because
physiological variations of blood ChE levels occur in a healthy
person and amongst populations, results should preferably be
compared with pre-exposure ChE levels.
Depressions of AChE or ChE of 20-25% are considered diagnostic of
exposure, but not necessarily indicative of hazard. Depressions of
30-50% or more are considered indicators for removal of an exposed
individual from further contact with pesticides until levels return
to normal. Work procedures and hygiene should also be checked.
Exposure can also be monitored by measuring the urinary excretion of
p-nitrophenol.
4.2 Explosion and fire hazards
Liquid formulations may be flammable. With sufficient burning or
external heat, methyl parathion will decompose, emitting toxic
fumes. Fire-fighters must wear protective clothing and
self-contained breathing apparatus. Extinguish fires with alcohol
resistant foam or powder. Confine the use of water spray to the
cooling of unaffected stock, thus avoiding polluted run-off from the
site.
4.3 Storage
Technical methyl parathion and its formulations should be stored in
the original labelled containers in locked, well-ventilated storage
areas, preferably dedicated to insecticides. Do not expose to direct
sunlight. Keep products out of reach of children and unauthorized
personnel. Do not store near food or animal feed.
4.4 Transport
Comply with any local regulations regarding movement of hazardous
goods. Do not transport with food or animal feed. Food and feed
should not be transported in vehicles that have been used for the
transport of pesticides. Make sure that containers are in good
condition and the labels are undamaged before dispatch.
4.5 Spillage and Disposal
4.5.1 Spillage
Avoid skin contamination and inhalation of vapour. Absorb spilled
liquid and cover contaminated areas with a 1:3 mixture of sodium
carbonate crystals and damp sawdust, lime, sand, or earth. Sweep up
and place it in an impervious container. Ensure that the container
is tightly closed and labelled before transfer to a safe place for
disposal.
Prevent liquid from spreading and contaminating other cargo,
vegetation, or waterways by using a barrier of the most suitable and
readily available material, e.g., earth or sand.
Empty any of the product remaining in the damaged/leaking container
into a clean empty container, which should then be tightly closed
and suitably labelled. Decontaminate emptied leaking containers with
a 10% sodium carbonate (washing soda) solution, added at a rate of
at least 1 litre/20-litre drum. Swirl round to rinse walls, empty,
and add rinsings to sawdust. Do not reuse containers for any other
purpose. Puncture and crush the container to prevent reuse.
4.5.2 Disposal
Large amounts should be incinerated at high temperature in a unit
with effluent gas scrubbing or should be adsorbed on vermiculite and
disposed of in a landfill, if incineration is impossible. When no
incinerator is available, bury in an approved dump, or in an area
where there is no risk of contamination of surface or ground water.
Before burying, mix liberally with sodium carbonate (washing soda)
crystals to help neutralize the product and with soil rich in
organic matter. Comply with any local legislation.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Methyl parathion is readily degraded and non-persistent in the
environment. It is highly toxic for aquatic invertebrates, birds,
bees, and wild mammals. It is moderately toxic for fish and
non-toxic for soil microorganisms.
Do not overspray water; time spraying to avoid killing of
pollinators.
Avoid contamination of soil, water, and the atmosphere by proper
methods of use, storage, transport, handling, and waste disposal. In
case of spillage, use the methods advised in section 4.5.1.
6. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. A full reference to the
original national document from which the information was extracted
can be obtained from IRPTC.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change
and should always be verified with the appropriate regulatory
authorities before application.
6.1 Previous evaluations by international bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) evaluated
methyl parathion in 1968, 1972, 1975, 1979, 1980, and 1984 (FAO/WHO,
1969, 1973, 1976, 1980, 1981, and 1985). The acceptable daily intake
for humans (ADI) was estimated at 0-0.02 mg/kg body weight in 1984.
This was based on the following levels causing no toxicological
effects:
- 2 mg/kg diet, equivalent to 0.1 mg/kg body weight in the rat;
and
- 0.3 mg/kg body weight per day in man.
The FAO/WHO Codex Alimentarius Commission (FAO/WHO, 1986)
recommended Maximum Residue Limits (MRLs) in several food
commodities, ranging from 0.05 to 0.2 mg/kg, including:
Commodity MRL (mg/kg) Commodity MRL (mg/kg)
cantaloupes 0.2 hops (dry cones) 0.05a
cole crops 0.2 melons 0.2
cottonseed oil 0.05 sugar beets 0.05a
cucumbers 0.2 tea (fermented and dried) 0.2
fruit, other 0.2 tomatoes 0.2
a Levels at or about the limit of determination.
The International Agency for Research on Cancer (IARC) classified
methyl parathion in Group 3 in 1982 and in 1987 (IARC, 1983, vol.
30, 1987, suppl. 7), and concluded that the available data do not
provide evidence that methyl parathion is carcinogenic to
experimental animals. No data on humans were available. The
available data provide no evidence that methyl parathion is likely
to present a carcinogenic risk to humans.
WHO (1990) classified technical methyl parathion as "extremely
hazardous" in normal use, on the basis of the oral LD50 in the rat
of 14 mg/kg. WHO/FAO (1975) issued a data sheet on methyl parathion
(No. 7).
6.2 Exposure limit values
Some exposure limit values are given on pp. 28-29
.
Pre-harvest intervals (the time between the last application of
methyl parathion and the harvest of the treated plants) have been
set in many countries. These intervals vary from 1 to 60 days (most
of them between 14 and 21 days), depending on the crop, the
harvesting technique, and the country, and can be verified with the
competent national authority.
6.3 Specific restrictions
In many countries where methyl parathion is approved as a pesticide,
specific uses, restrictions, and precautions are listed in national
regulatory documents, e.g., no liquid methyl parathion formulations
have been approved for registration in Finland because of their high
acute toxicity. In the USSR, the agricultural use of methyl
parathion has been restricted. In Hungary, it may only be applied in
agriculture by properly trained staff, using protective equipment.
Methyl parathion has either not been registered or has been banned
in: Bangladesh, Belgium, Bulgaria, Canada, China, Ecuador, Egypt,
Hong Kong, Ireland, Japan, Sri Lanka, and the United Kingdom.
In Japan and the USA, methyl parathion and its preparations may only
be handled by certified operators. In Germany, it may not be handled
by adolescents or pregnant and nursing women.
6.4 Labelling, packaging, and transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies methyl parathion in:
- Hazard Class 6.1: poisonous substance;
- Packing Group 2: substances and preparations presenting a
serious risk of poisoning, for formulations containing
12-100% methyl parathion.
- Packing Group 3: harmful substances and preparations
presenting a serious risk of poisoning, for solid
formulations containing 3-12% active material, and liquid
formulations containing 1.2-12% active material.
Exposure limit values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Workplace Argentina Maximum permissible concentration
- Time-weighted average (TWA) a 0.2 mg/m3 1984
- Short-term exposure limit (STEL) 0.6 mg/m3
Finland Maximum permissible concentration
- Time-weighted average (TWA) 0.2 mg/m3 1987
- Short-term exposure limit 0.6 mg/m3
United Kingdom Occupational exposure standard (TWA) a 0.2 mg/m3 1989
- Short-term exposure limit (STEL) 0.6 mg/m3
USA (ACGIH) Threshold limit value (TLV)
- Time-weighted average (TWA) 0.2 mg/m3 b 1986
- Short-term exposure limit deleted
(STEL = 10 minutes)
USSR Maximum allowable concentration (MAC)
- Ceiling value (vapour + aerosol) 0.1 mg/m3 1977
AIR Ambient USSR Maximum allowable concentration (MAC) 0.008 mg/m3 1984
(once per day)
FOOD Uptake from FAO/WHO Acceptable daily intake (ADI) 0-0.02 mg/kg 1984
body weight
Residues FAO/WHO Maximum residue limit (MRL) 0.05-0.2 mg/kg 1986
(in specified products)
Exposure limit values (continued)
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Ambient Japan Environmental water quality standard not detectable 1981
Surface USSR Maximum acceptable concentration 0.02 mg/litre 1983
Effluent Japan Emission of total organophosphorus 1 mg/litre 1981
compounds
SOIL USSR Maximum acceptable concentration 0.1 mg/kg 1981
a TWA usually 8 h.
b Skin absorption possible.
The label should be as follows:
Packaging Group I and II:
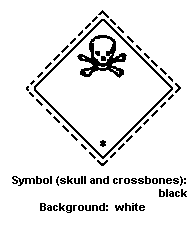 Packaging Group III:
Packaging Group III:
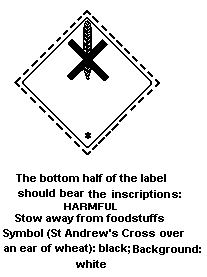 In the International Maritime Dangerous Goods (IMDG) Code, methyl
parathion is classified as a marine pollutant with a severe
pollution potential. It should bear the following mark on the label:
In the International Maritime Dangerous Goods (IMDG) Code, methyl
parathion is classified as a marine pollutant with a severe
pollution potential. It should bear the following mark on the label:
 For flammable formulations, the following subsidiary label is
required, when the flashpoint of the solution is below, or equal to,
61 °C (closed cup):
For flammable formulations, the following subsidiary label is
required, when the flashpoint of the solution is below, or equal to,
61 °C (closed cup):
 There is no WHO specification on methyl parathion as the material is
not used in public health. However, specifications for technical
material and some formulations have been agreed upon between FAO and
the manufacturers.
All packages should bear, durably and legibly marked on the
container, the following:
- Manufacturer's name
- Technical parathion to specification
- Batch or reference number, and date of test
- Net weight of contents
- Date of manufacture
and, in the case of the formulated products:
- Manufacturer's name
- Methyl parathion to specification
- Methyl parathion ... g/kg
- Batch or reference number, and date of test
- Net weight of contents
- Instructions for dilution
- Date of formulation
and the following minimum cautionary notice:
Methyl parathion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It
may be absorbed through the skin. Avoid skin contact; wear
protective gloves, clean protective clothing and a respirator
when handling the material. Wash thoroughly with soap and
water after using.
Keep the material out of the reach of children and well away
from foodstuffs and animal feed and their containers.
If poisoning occurs, call a physician. Atropine and
pralidoxime are specific antidotes, and artificial respiration
may be needed.
The methyl parathion content should be declared (minimum 90% for the
technical product) and should not differ from the declared
percentage by more than 2% for the technical product and 5-10% for
its formulations.
Containers should be suitable, clean, dry, and as specified in the
order, and should not adversely affect, or be affected by, the
product, but should adequately protect it from external conditions.
They should comply with pertinent national and international
transport and safety regulations.
Specifications for storage stability are given.
The European Economic Community legislation requires labelling as
dangerous substance using the symbol:
There is no WHO specification on methyl parathion as the material is
not used in public health. However, specifications for technical
material and some formulations have been agreed upon between FAO and
the manufacturers.
All packages should bear, durably and legibly marked on the
container, the following:
- Manufacturer's name
- Technical parathion to specification
- Batch or reference number, and date of test
- Net weight of contents
- Date of manufacture
and, in the case of the formulated products:
- Manufacturer's name
- Methyl parathion to specification
- Methyl parathion ... g/kg
- Batch or reference number, and date of test
- Net weight of contents
- Instructions for dilution
- Date of formulation
and the following minimum cautionary notice:
Methyl parathion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It
may be absorbed through the skin. Avoid skin contact; wear
protective gloves, clean protective clothing and a respirator
when handling the material. Wash thoroughly with soap and
water after using.
Keep the material out of the reach of children and well away
from foodstuffs and animal feed and their containers.
If poisoning occurs, call a physician. Atropine and
pralidoxime are specific antidotes, and artificial respiration
may be needed.
The methyl parathion content should be declared (minimum 90% for the
technical product) and should not differ from the declared
percentage by more than 2% for the technical product and 5-10% for
its formulations.
Containers should be suitable, clean, dry, and as specified in the
order, and should not adversely affect, or be affected by, the
product, but should adequately protect it from external conditions.
They should comply with pertinent national and international
transport and safety regulations.
Specifications for storage stability are given.
The European Economic Community legislation requires labelling as
dangerous substance using the symbol:
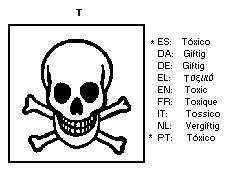 The label must read:
Very toxic by inhalation, in contact with skin and if
swallowed; keep locked up; keep away from food, drink and
animal feeding stuffs; after contact with skin, wash
immediately with plenty of ____ - (to be specified by the
manufacturer); in case of accident or if you feel unwell, seek
medical advice immediately (show the label where possible)
The European Economic Community legislation on labelling of
pesticide preparations classifies pesticide preparations that
contain methyl parathion in Class 1A, as toxic at concentrations
> 1% and as harmful at > 0.05-1%. Member States should ensure that
pesticides cannot be placed on the market unless their packaging,
fastenings, and labels comply with the requirements laid down.
6.5 Waste disposal
In the USA, any non-domestic waste containing methyl parathion is
considered a hazardous waste and the competent authority should be
notified. Permits are required for its handling, transport,
treatment, storage, or disposal. Waste incinerators must achieve
99.99% destruction and removal of this substance.
6.6 Other measures
The European Economic Community legislation has listed methyl
parathion as a dangerous substance at quantities >100 kg in the
directive on the major accident hazards of certain industrial
activities. Any person in charge of an industrial activity
involving, or possibly involving, one or more dangerous substances
is obliged to take all the measures necessary to prevent major
accidents, to limit their consequences for man and the environment,
and to notify the competent authority about the industrial activity.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - classification and
labelling in the European Communities. Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
FAO (1971) Parathion-methyl - FAO specifications for plant
protection products. Rome, Food and Agriculture Organization of the
United Nations, pp 22.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome. Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluations of pesticide residues in food,
Rome. Food and Agriculture Organization of the United Nations.
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. 3rd ed.
Rome, Codex Alimentarius Commission, Food and Agriculture
Organization of the United Nations (CAC/Vol XIII).
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. Codex
Alimentarius Commission CAC/Vol. XIII. 3rd ed. Rome, Food and
Agriculture Organization of the United Nations.
FAO/WHO (1989) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 4th ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
Hayes WJJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology. 3
Vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IARC (1983) IARC monographs on the evaluation of carcinogenic risk
of chemicals to man. Vol. 30. Miscellaneous chemicals. Lyon,
International Agency for Research on Cancer.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished document No. VBC/84.889).
Sax NI (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme, Industry and Environment Office, 80 pp.
UNITED NATIONS (1989a) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 2nd ed. New York, United
Nations.
UNITED NATIONS (1989b) Recommendations on the transport of dangerous
goods. 6th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH)
01-123).
WHO (1986) EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 181 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification, 1992-93. Geneva, World
Health Organization, 66 pp. (Unpublished document, WHO/PCS/92.14).
WHO/FAO (1975-87) Methyl parathion: Data sheets on pesticides, No.
7. Geneva, World Health Organization (Unpublished documents).
WHO (in preparation) EHC: 145 Methyl parathion. Geneva, World Health
Organization.
Worthing CR & Hance RJ (1991) The pesticide manual. 9th ed. Unwin
Brothers Ltd., Surrey, United Kingdom, British Crop Protection
Council.
ANNEX 1. TREATMENT OF ORGANOPHOSPHATE INSECTICIDE
POISONING IN MANa
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be
included in the IPCS Health and Safety Guides to be prepared for
selected organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in cleaning the skin area where venepuncture is
performed. Blood might be contaminated with direct-acting
organophosphorus esters and, therefore, inaccurate measures of ChE
inhibition might result. Extensive eye irrigation with water or
saline should also be performed. In the case of ingestion, vomiting
might be induced, if the patient is conscious, by the administration
of ipecacuanha syrup (10-30 ml) followed by 200 ml water. This
treatment is, however, contraindicated in the case of pesticides
dissolved in hydrocarbon solvents. Gastric lavage (with addition of
bicarbonate solution or activated charcoal) can also be performed,
particularly in unconscious patients, taking care to prevent
aspiration of fluids into the lungs (i.e., only after a tracheal
tube has been put into place).
The volume of fluid introduced into the stomach should be recorded
and samples of gastric lavage frozen and stored for subsequent
chemical analysis. If the formulation of the pesticide involved is
available, it should also be stored for further analysis (i.e.,
detection of toxicologically relevant impurities). A purgative can
be administered to remove the ingested compound.
a From EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 1986.
I.2 General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific pharmacological treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine
treatment varies from case to case, but should maintain the patient
fully atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases
and total daily doses up to several hundred mg may be necessary
during the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have
beneficial effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and
drugs that may depress respiration are not recommended in the
absence of artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one
time may cause toxic side-effects. Pulse-rate should not exceed
120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be
taken into, and then released from, fat depots over a period of many
days. It is therefore quite incorrect to abandon oxime treatment
after 1-2 days on the supposition that all inhibited enzyme will be
aged. Ecobichon et al. (1977) noted prompt improvement in both
condition and blood-ChEs in response to pralidoxime given on the
11th-15th days after major symptoms of poisoning appeared due to
extended exposure to fenitrothion (a dimethyl phosphate with a short
half-life for aging of inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in
error, while drunk. Therapeutic dosages were progressively increased
up to 6 mg atropine iv every 15 min together with continuous iv
infusion of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to
6 after intoxication. After considerable improvement, the patient
relapsed and further aggressive therapy was given at a declining
rate from days 10 to 16 (atropine) and to day 23 (oxime),
respectively. In total, 92 g of pralidoxime chloride and 3912 mg of
atropine were given and the patient was discharged on the
thirty-third day with no apparent sequelae.
References to Annex 1
Ecobichon DJ, Ozere RL, Reid E, & Crocker JFS (1977) Acute
fenitrothion poisoning. Can Med Assoc J, 116: 377-379.
Kagan Ju S (1977) [Toxicology of organophosphorus pesticides],
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning, Geneva, World Health Organization
(Unpublished document VBC/84.889).
Taylor P (1980) Anticholinesterase agents. In: Goodman LS & Gilman
A, ed. The pharmacological basis of therapeutics, 6th ed., New York,
Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of
medical practitioners, London, United Kingdom Department of Health
and Social Security, pp. 41-47.
Vale JA & Scott GW (1974) Organophosphorus poisoning. Guy's Hosp
Rep, 123: 13-25.
Warriner RA III, Nies AS, & Hayes WJ Jr (1977) Severe
organophosphate poisoning complicated by alcohol and turpentine
ingestion. Arch environ Health, 32: 203-205.
The label must read:
Very toxic by inhalation, in contact with skin and if
swallowed; keep locked up; keep away from food, drink and
animal feeding stuffs; after contact with skin, wash
immediately with plenty of ____ - (to be specified by the
manufacturer); in case of accident or if you feel unwell, seek
medical advice immediately (show the label where possible)
The European Economic Community legislation on labelling of
pesticide preparations classifies pesticide preparations that
contain methyl parathion in Class 1A, as toxic at concentrations
> 1% and as harmful at > 0.05-1%. Member States should ensure that
pesticides cannot be placed on the market unless their packaging,
fastenings, and labels comply with the requirements laid down.
6.5 Waste disposal
In the USA, any non-domestic waste containing methyl parathion is
considered a hazardous waste and the competent authority should be
notified. Permits are required for its handling, transport,
treatment, storage, or disposal. Waste incinerators must achieve
99.99% destruction and removal of this substance.
6.6 Other measures
The European Economic Community legislation has listed methyl
parathion as a dangerous substance at quantities >100 kg in the
directive on the major accident hazards of certain industrial
activities. Any person in charge of an industrial activity
involving, or possibly involving, one or more dangerous substances
is obliged to take all the measures necessary to prevent major
accidents, to limit their consequences for man and the environment,
and to notify the competent authority about the industrial activity.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - classification and
labelling in the European Communities. Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
FAO (1971) Parathion-methyl - FAO specifications for plant
protection products. Rome, Food and Agriculture Organization of the
United Nations, pp 22.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome. Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluations of pesticide residues in food,
Rome. Food and Agriculture Organization of the United Nations.
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. 3rd ed.
Rome, Codex Alimentarius Commission, Food and Agriculture
Organization of the United Nations (CAC/Vol XIII).
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. Codex
Alimentarius Commission CAC/Vol. XIII. 3rd ed. Rome, Food and
Agriculture Organization of the United Nations.
FAO/WHO (1989) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 4th ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
Hayes WJJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology. 3
Vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IARC (1983) IARC monographs on the evaluation of carcinogenic risk
of chemicals to man. Vol. 30. Miscellaneous chemicals. Lyon,
International Agency for Research on Cancer.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished document No. VBC/84.889).
Sax NI (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme, Industry and Environment Office, 80 pp.
UNITED NATIONS (1989a) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 2nd ed. New York, United
Nations.
UNITED NATIONS (1989b) Recommendations on the transport of dangerous
goods. 6th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH)
01-123).
WHO (1986) EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 181 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification, 1992-93. Geneva, World
Health Organization, 66 pp. (Unpublished document, WHO/PCS/92.14).
WHO/FAO (1975-87) Methyl parathion: Data sheets on pesticides, No.
7. Geneva, World Health Organization (Unpublished documents).
WHO (in preparation) EHC: 145 Methyl parathion. Geneva, World Health
Organization.
Worthing CR & Hance RJ (1991) The pesticide manual. 9th ed. Unwin
Brothers Ltd., Surrey, United Kingdom, British Crop Protection
Council.
ANNEX 1. TREATMENT OF ORGANOPHOSPHATE INSECTICIDE
POISONING IN MANa
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be
included in the IPCS Health and Safety Guides to be prepared for
selected organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in cleaning the skin area where venepuncture is
performed. Blood might be contaminated with direct-acting
organophosphorus esters and, therefore, inaccurate measures of ChE
inhibition might result. Extensive eye irrigation with water or
saline should also be performed. In the case of ingestion, vomiting
might be induced, if the patient is conscious, by the administration
of ipecacuanha syrup (10-30 ml) followed by 200 ml water. This
treatment is, however, contraindicated in the case of pesticides
dissolved in hydrocarbon solvents. Gastric lavage (with addition of
bicarbonate solution or activated charcoal) can also be performed,
particularly in unconscious patients, taking care to prevent
aspiration of fluids into the lungs (i.e., only after a tracheal
tube has been put into place).
The volume of fluid introduced into the stomach should be recorded
and samples of gastric lavage frozen and stored for subsequent
chemical analysis. If the formulation of the pesticide involved is
available, it should also be stored for further analysis (i.e.,
detection of toxicologically relevant impurities). A purgative can
be administered to remove the ingested compound.
a From EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 1986.
I.2 General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific pharmacological treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine
treatment varies from case to case, but should maintain the patient
fully atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases
and total daily doses up to several hundred mg may be necessary
during the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have
beneficial effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and
drugs that may depress respiration are not recommended in the
absence of artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one
time may cause toxic side-effects. Pulse-rate should not exceed
120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be
taken into, and then released from, fat depots over a period of many
days. It is therefore quite incorrect to abandon oxime treatment
after 1-2 days on the supposition that all inhibited enzyme will be
aged. Ecobichon et al. (1977) noted prompt improvement in both
condition and blood-ChEs in response to pralidoxime given on the
11th-15th days after major symptoms of poisoning appeared due to
extended exposure to fenitrothion (a dimethyl phosphate with a short
half-life for aging of inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in
error, while drunk. Therapeutic dosages were progressively increased
up to 6 mg atropine iv every 15 min together with continuous iv
infusion of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to
6 after intoxication. After considerable improvement, the patient
relapsed and further aggressive therapy was given at a declining
rate from days 10 to 16 (atropine) and to day 23 (oxime),
respectively. In total, 92 g of pralidoxime chloride and 3912 mg of
atropine were given and the patient was discharged on the
thirty-third day with no apparent sequelae.
References to Annex 1
Ecobichon DJ, Ozere RL, Reid E, & Crocker JFS (1977) Acute
fenitrothion poisoning. Can Med Assoc J, 116: 377-379.
Kagan Ju S (1977) [Toxicology of organophosphorus pesticides],
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning, Geneva, World Health Organization
(Unpublished document VBC/84.889).
Taylor P (1980) Anticholinesterase agents. In: Goodman LS & Gilman
A, ed. The pharmacological basis of therapeutics, 6th ed., New York,
Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of
medical practitioners, London, United Kingdom Department of Health
and Social Security, pp. 41-47.
Vale JA & Scott GW (1974) Organophosphorus poisoning. Guy's Hosp
Rep, 123: 13-25.
Warriner RA III, Nies AS, & Hayes WJ Jr (1977) Severe
organophosphate poisoning complicated by alcohol and turpentine
ingestion. Arch environ Health, 32: 203-205.
 Common names: parathion-methyl, methyl parathion,
metaphos (USSR)
Molecular formula: C8H10NO5PS
IUPAC name: O,O-dimethyl O-4-nitrophenyl
phosphorothioate
CAS name: O,O-dimethyl O-(4-nitrophenyl)
phosphorothioate (9CI)
CAS registry number:
Common names: parathion-methyl, methyl parathion,
metaphos (USSR)
Molecular formula: C8H10NO5PS
IUPAC name: O,O-dimethyl O-4-nitrophenyl
phosphorothioate
CAS name: O,O-dimethyl O-(4-nitrophenyl)
phosphorothioate (9CI)
CAS registry number:  Packaging Group III:
Packaging Group III:
 In the International Maritime Dangerous Goods (IMDG) Code, methyl
parathion is classified as a marine pollutant with a severe
pollution potential. It should bear the following mark on the label:
In the International Maritime Dangerous Goods (IMDG) Code, methyl
parathion is classified as a marine pollutant with a severe
pollution potential. It should bear the following mark on the label:
 For flammable formulations, the following subsidiary label is
required, when the flashpoint of the solution is below, or equal to,
61 °C (closed cup):
For flammable formulations, the following subsidiary label is
required, when the flashpoint of the solution is below, or equal to,
61 °C (closed cup):
 There is no WHO specification on methyl parathion as the material is
not used in public health. However, specifications for technical
material and some formulations have been agreed upon between FAO and
the manufacturers.
All packages should bear, durably and legibly marked on the
container, the following:
- Manufacturer's name
- Technical parathion to specification
- Batch or reference number, and date of test
- Net weight of contents
- Date of manufacture
and, in the case of the formulated products:
- Manufacturer's name
- Methyl parathion to specification
- Methyl parathion ... g/kg
- Batch or reference number, and date of test
- Net weight of contents
- Instructions for dilution
- Date of formulation
and the following minimum cautionary notice:
Methyl parathion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It
may be absorbed through the skin. Avoid skin contact; wear
protective gloves, clean protective clothing and a respirator
when handling the material. Wash thoroughly with soap and
water after using.
Keep the material out of the reach of children and well away
from foodstuffs and animal feed and their containers.
If poisoning occurs, call a physician. Atropine and
pralidoxime are specific antidotes, and artificial respiration
may be needed.
The methyl parathion content should be declared (minimum 90% for the
technical product) and should not differ from the declared
percentage by more than 2% for the technical product and 5-10% for
its formulations.
Containers should be suitable, clean, dry, and as specified in the
order, and should not adversely affect, or be affected by, the
product, but should adequately protect it from external conditions.
They should comply with pertinent national and international
transport and safety regulations.
Specifications for storage stability are given.
The European Economic Community legislation requires labelling as
dangerous substance using the symbol:
There is no WHO specification on methyl parathion as the material is
not used in public health. However, specifications for technical
material and some formulations have been agreed upon between FAO and
the manufacturers.
All packages should bear, durably and legibly marked on the
container, the following:
- Manufacturer's name
- Technical parathion to specification
- Batch or reference number, and date of test
- Net weight of contents
- Date of manufacture
and, in the case of the formulated products:
- Manufacturer's name
- Methyl parathion to specification
- Methyl parathion ... g/kg
- Batch or reference number, and date of test
- Net weight of contents
- Instructions for dilution
- Date of formulation
and the following minimum cautionary notice:
Methyl parathion is an organophosphorus compound that inhibits
cholinesterase. It is poisonous if swallowed or inhaled. It
may be absorbed through the skin. Avoid skin contact; wear
protective gloves, clean protective clothing and a respirator
when handling the material. Wash thoroughly with soap and
water after using.
Keep the material out of the reach of children and well away
from foodstuffs and animal feed and their containers.
If poisoning occurs, call a physician. Atropine and
pralidoxime are specific antidotes, and artificial respiration
may be needed.
The methyl parathion content should be declared (minimum 90% for the
technical product) and should not differ from the declared
percentage by more than 2% for the technical product and 5-10% for
its formulations.
Containers should be suitable, clean, dry, and as specified in the
order, and should not adversely affect, or be affected by, the
product, but should adequately protect it from external conditions.
They should comply with pertinent national and international
transport and safety regulations.
Specifications for storage stability are given.
The European Economic Community legislation requires labelling as
dangerous substance using the symbol:
 The label must read:
Very toxic by inhalation, in contact with skin and if
swallowed; keep locked up; keep away from food, drink and
animal feeding stuffs; after contact with skin, wash
immediately with plenty of ____ - (to be specified by the
manufacturer); in case of accident or if you feel unwell, seek
medical advice immediately (show the label where possible)
The European Economic Community legislation on labelling of
pesticide preparations classifies pesticide preparations that
contain methyl parathion in Class 1A, as toxic at concentrations
> 1% and as harmful at > 0.05-1%. Member States should ensure that
pesticides cannot be placed on the market unless their packaging,
fastenings, and labels comply with the requirements laid down.
6.5 Waste disposal
In the USA, any non-domestic waste containing methyl parathion is
considered a hazardous waste and the competent authority should be
notified. Permits are required for its handling, transport,
treatment, storage, or disposal. Waste incinerators must achieve
99.99% destruction and removal of this substance.
6.6 Other measures
The European Economic Community legislation has listed methyl
parathion as a dangerous substance at quantities >100 kg in the
directive on the major accident hazards of certain industrial
activities. Any person in charge of an industrial activity
involving, or possibly involving, one or more dangerous substances
is obliged to take all the measures necessary to prevent major
accidents, to limit their consequences for man and the environment,
and to notify the competent authority about the industrial activity.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - classification and
labelling in the European Communities. Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
FAO (1971) Parathion-methyl - FAO specifications for plant
protection products. Rome, Food and Agriculture Organization of the
United Nations, pp 22.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome. Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluations of pesticide residues in food,
Rome. Food and Agriculture Organization of the United Nations.
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. 3rd ed.
Rome, Codex Alimentarius Commission, Food and Agriculture
Organization of the United Nations (CAC/Vol XIII).
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. Codex
Alimentarius Commission CAC/Vol. XIII. 3rd ed. Rome, Food and
Agriculture Organization of the United Nations.
FAO/WHO (1989) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 4th ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
Hayes WJJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology. 3
Vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IARC (1983) IARC monographs on the evaluation of carcinogenic risk
of chemicals to man. Vol. 30. Miscellaneous chemicals. Lyon,
International Agency for Research on Cancer.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished document No. VBC/84.889).
Sax NI (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme, Industry and Environment Office, 80 pp.
UNITED NATIONS (1989a) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 2nd ed. New York, United
Nations.
UNITED NATIONS (1989b) Recommendations on the transport of dangerous
goods. 6th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH)
01-123).
WHO (1986) EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 181 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification, 1992-93. Geneva, World
Health Organization, 66 pp. (Unpublished document, WHO/PCS/92.14).
WHO/FAO (1975-87) Methyl parathion: Data sheets on pesticides, No.
7. Geneva, World Health Organization (Unpublished documents).
WHO (in preparation) EHC: 145 Methyl parathion. Geneva, World Health
Organization.
Worthing CR & Hance RJ (1991) The pesticide manual. 9th ed. Unwin
Brothers Ltd., Surrey, United Kingdom, British Crop Protection
Council.
ANNEX 1. TREATMENT OF ORGANOPHOSPHATE INSECTICIDE
POISONING IN MANa
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be
included in the IPCS Health and Safety Guides to be prepared for
selected organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in cleaning the skin area where venepuncture is
performed. Blood might be contaminated with direct-acting
organophosphorus esters and, therefore, inaccurate measures of ChE
inhibition might result. Extensive eye irrigation with water or
saline should also be performed. In the case of ingestion, vomiting
might be induced, if the patient is conscious, by the administration
of ipecacuanha syrup (10-30 ml) followed by 200 ml water. This
treatment is, however, contraindicated in the case of pesticides
dissolved in hydrocarbon solvents. Gastric lavage (with addition of
bicarbonate solution or activated charcoal) can also be performed,
particularly in unconscious patients, taking care to prevent
aspiration of fluids into the lungs (i.e., only after a tracheal
tube has been put into place).
The volume of fluid introduced into the stomach should be recorded
and samples of gastric lavage frozen and stored for subsequent
chemical analysis. If the formulation of the pesticide involved is
available, it should also be stored for further analysis (i.e.,
detection of toxicologically relevant impurities). A purgative can
be administered to remove the ingested compound.
a From EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 1986.
I.2 General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific pharmacological treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine
treatment varies from case to case, but should maintain the patient
fully atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases
and total daily doses up to several hundred mg may be necessary
during the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have
beneficial effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and
drugs that may depress respiration are not recommended in the
absence of artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one
time may cause toxic side-effects. Pulse-rate should not exceed
120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be
taken into, and then released from, fat depots over a period of many
days. It is therefore quite incorrect to abandon oxime treatment
after 1-2 days on the supposition that all inhibited enzyme will be
aged. Ecobichon et al. (1977) noted prompt improvement in both
condition and blood-ChEs in response to pralidoxime given on the
11th-15th days after major symptoms of poisoning appeared due to
extended exposure to fenitrothion (a dimethyl phosphate with a short
half-life for aging of inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in
error, while drunk. Therapeutic dosages were progressively increased
up to 6 mg atropine iv every 15 min together with continuous iv
infusion of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to
6 after intoxication. After considerable improvement, the patient
relapsed and further aggressive therapy was given at a declining
rate from days 10 to 16 (atropine) and to day 23 (oxime),
respectively. In total, 92 g of pralidoxime chloride and 3912 mg of
atropine were given and the patient was discharged on the
thirty-third day with no apparent sequelae.
References to Annex 1
Ecobichon DJ, Ozere RL, Reid E, & Crocker JFS (1977) Acute
fenitrothion poisoning. Can Med Assoc J, 116: 377-379.
Kagan Ju S (1977) [Toxicology of organophosphorus pesticides],
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning, Geneva, World Health Organization
(Unpublished document VBC/84.889).
Taylor P (1980) Anticholinesterase agents. In: Goodman LS & Gilman
A, ed. The pharmacological basis of therapeutics, 6th ed., New York,
Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of
medical practitioners, London, United Kingdom Department of Health
and Social Security, pp. 41-47.
Vale JA & Scott GW (1974) Organophosphorus poisoning. Guy's Hosp
Rep, 123: 13-25.
Warriner RA III, Nies AS, & Hayes WJ Jr (1977) Severe
organophosphate poisoning complicated by alcohol and turpentine
ingestion. Arch environ Health, 32: 203-205.
The label must read:
Very toxic by inhalation, in contact with skin and if
swallowed; keep locked up; keep away from food, drink and
animal feeding stuffs; after contact with skin, wash
immediately with plenty of ____ - (to be specified by the
manufacturer); in case of accident or if you feel unwell, seek
medical advice immediately (show the label where possible)
The European Economic Community legislation on labelling of
pesticide preparations classifies pesticide preparations that
contain methyl parathion in Class 1A, as toxic at concentrations
> 1% and as harmful at > 0.05-1%. Member States should ensure that
pesticides cannot be placed on the market unless their packaging,
fastenings, and labels comply with the requirements laid down.
6.5 Waste disposal
In the USA, any non-domestic waste containing methyl parathion is
considered a hazardous waste and the competent authority should be
notified. Permits are required for its handling, transport,
treatment, storage, or disposal. Waste incinerators must achieve
99.99% destruction and removal of this substance.
6.6 Other measures
The European Economic Community legislation has listed methyl
parathion as a dangerous substance at quantities >100 kg in the
directive on the major accident hazards of certain industrial
activities. Any person in charge of an industrial activity
involving, or possibly involving, one or more dangerous substances
is obliged to take all the measures necessary to prevent major
accidents, to limit their consequences for man and the environment,
and to notify the competent authority about the industrial activity.
BIBLIOGRAPHY
CEC (1987) Legislation on dangerous substances - classification and
labelling in the European Communities. Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
FAO (1971) Parathion-methyl - FAO specifications for plant
protection products. Rome, Food and Agriculture Organization of the
United Nations, pp 22.
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome. Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1964-present) Evaluations of pesticide residues in food,
Rome. Food and Agriculture Organization of the United Nations.
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. 3rd ed.
Rome, Codex Alimentarius Commission, Food and Agriculture
Organization of the United Nations (CAC/Vol XIII).
FAO/WHO (1986) Codex Maximum Limits for pesticide residues. Codex
Alimentarius Commission CAC/Vol. XIII. 3rd ed. Rome, Food and
Agriculture Organization of the United Nations.
FAO/WHO (1989) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues. 4th ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants
de Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
Hayes WJJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology. 3
Vol. New York, Academic Press.
IARC (1972-present) IARC Monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IARC (1983) IARC monographs on the evaluation of carcinogenic risk
of chemicals to man. Vol. 30. Miscellaneous chemicals. Lyon,
International Agency for Research on Cancer.
ILO (1991) Safety and health in the use of agro-chemicals - a guide.
Geneva, International Labour Office.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register of Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(Unpublished document No. VBC/84.889).
Sax NI (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNEP/IEO (1990) Storage of hazardous materials: a technical guide
for safe warehousing of hazardous materials. Paris, United Nations
Environment Programme, Industry and Environment Office, 80 pp.
UNITED NATIONS (1989a) Consolidated list of products whose
consumption and/or sale have been banned, withdrawn, severely
restricted or not approved by governments. 2nd ed. New York, United
Nations.
UNITED NATIONS (1989b) Recommendations on the transport of dangerous
goods. 6th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH)
01-123).
WHO (1986) EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 181 pp.
WHO (1992) The WHO recommended classification of pesticides by
hazard and guidelines to classification, 1992-93. Geneva, World
Health Organization, 66 pp. (Unpublished document, WHO/PCS/92.14).
WHO/FAO (1975-87) Methyl parathion: Data sheets on pesticides, No.
7. Geneva, World Health Organization (Unpublished documents).
WHO (in preparation) EHC: 145 Methyl parathion. Geneva, World Health
Organization.
Worthing CR & Hance RJ (1991) The pesticide manual. 9th ed. Unwin
Brothers Ltd., Surrey, United Kingdom, British Crop Protection
Council.
ANNEX 1. TREATMENT OF ORGANOPHOSPHATE INSECTICIDE
POISONING IN MANa
All cases of organophosphorus poisoning should be dealt with as an
emergency and the patient sent to hospital as quickly as possible.
Although symptoms may develop rapidly, delay in onset or a steady
increase in severity may be seen up to 48 h after ingestion of some
formulated organophosphorus insecticides.
Extensive descriptions of treatment of poisoning by organophosphorus
insecticides are given in several major references (Kagan, 1977;
Taylor, 1980; UK DHSS, 1983; Plestina, 1984) and will also be
included in the IPCS Health and Safety Guides to be prepared for
selected organophosphorus insecticides.
The treatment is based on:
(a) minimizing the absorption;
(b) general supportive treatment; and
(c) specific pharmacological treatment.
I.1 Minimizing the absorption
When dermal exposure occurs, decontamination procedures include
removal of contaminated clothes and washing of the skin with
alkaline soap or with a sodium bicarbonate solution. Particular care
should be taken in cleaning the skin area where venepuncture is
performed. Blood might be contaminated with direct-acting
organophosphorus esters and, therefore, inaccurate measures of ChE
inhibition might result. Extensive eye irrigation with water or
saline should also be performed. In the case of ingestion, vomiting
might be induced, if the patient is conscious, by the administration
of ipecacuanha syrup (10-30 ml) followed by 200 ml water. This
treatment is, however, contraindicated in the case of pesticides
dissolved in hydrocarbon solvents. Gastric lavage (with addition of
bicarbonate solution or activated charcoal) can also be performed,
particularly in unconscious patients, taking care to prevent
aspiration of fluids into the lungs (i.e., only after a tracheal
tube has been put into place).
The volume of fluid introduced into the stomach should be recorded
and samples of gastric lavage frozen and stored for subsequent
chemical analysis. If the formulation of the pesticide involved is
available, it should also be stored for further analysis (i.e.,
detection of toxicologically relevant impurities). A purgative can
be administered to remove the ingested compound.
a From EHC 63: Organophosphorus insecticides - a general
introduction. Geneva, World Health Organization, 1986.
I.2 General supportive treatment
Artificial respiration (via a tracheal tube) should be started at
the first sign of respiratory failure and maintained for as long as
necessary.
Cautious administration of fluids is advised, as well as general
supportive and symptomatic pharmacological treatment and absolute
rest.
I.3 Specific pharmacological treatment
I.3.1 Atropine
Atropine should be given, beginning with 2 mg iv and given at
15-30-min intervals. The dose and the frequency of atropine
treatment varies from case to case, but should maintain the patient
fully atropinized (dilated pupils, dry mouth, skin flushing, etc.).
Continuous infusion of atropine may be necessary in extreme cases
and total daily doses up to several hundred mg may be necessary
during the first few days of treatment.
I.3.2 Oxime reactivators
Cholinesterase reactivators (e.g., pralidoxime, obidoxime)
specifically restore AChE activity inhibited by organophosphates.
This is not the case with enzymes inhibited by carbamates. The
treatment should begin as soon as possible, because oximes are not
effective on "aged" phosphorylated ChEs. However, if absorption,
distribution, and metabolism are thought to be delayed for any
reasons, oximes can be administered for several days after
intoxication. Effective treatment with oximes reduces the required
dose of atropine. Pralidoxime is the most widely available oxime. A
dose of 1 g pralidoxime can be given either im or iv and repeated
2-3 times per day or, in extreme cases, more often. If possible,
blood samples should be taken for AChE determinations before and
during treatment. Skin should be carefully cleansed before sampling.
Results of the assays should influence the decision whether to
continue oxime therapy after the first 2 days.
There are indications that oxime therapy may possibly have
beneficial effects on CNS-derived symptoms.
I.3.3 Diazepam
Diazepam should be included in the therapy of all but the mildest
cases. Besides relieving anxiety, it appears to counteract some
aspects of CNS-derived symptoms that are not affected by atropine.
Doses of 10 mg sc or iv are appropriate and may be repeated as
required (Vale & Scott, 1974). Other centrally acting drugs and
drugs that may depress respiration are not recommended in the
absence of artificial respiration procedures.
I.3.4 Notes on the recommended treatment
I.3.4.1 Effects of atropine and oxime
The combined effect far exceeds the benefit of either drug singly.
I.3.4.2 Response to atropine
The response of the eye pupil may be unreliable in cases of
organophosphorus poisoning. A flushed skin and drying of secretions
are the best guide to the effectiveness of atropinization. Although
repeated dosing may well be necessary, excessive doses at any one
time may cause toxic side-effects. Pulse-rate should not exceed
120/min.
I.3.4.3 Persistence of treatment
Some organophosphorus pesticides are very lipophilic and may be
taken into, and then released from, fat depots over a period of many
days. It is therefore quite incorrect to abandon oxime treatment
after 1-2 days on the supposition that all inhibited enzyme will be
aged. Ecobichon et al. (1977) noted prompt improvement in both
condition and blood-ChEs in response to pralidoxime given on the
11th-15th days after major symptoms of poisoning appeared due to
extended exposure to fenitrothion (a dimethyl phosphate with a short
half-life for aging of inhibited AChE).
I.3.4.4 Dosage of atropine and oxime
The recommended doses above pertain to exposures, usually for an
occupational setting, but, in the case of very severe exposure or
massive ingestion (accidental or deliberate), the therapeutic doses
may be extended considerably. Warriner et al. (1977) reported the
case of a patient who drank a large quantity of dicrotophos, in
error, while drunk. Therapeutic dosages were progressively increased
up to 6 mg atropine iv every 15 min together with continuous iv
infusion of pralidoxime chloride at 0.5 g/h for 72 h, from days 3 to
6 after intoxication. After considerable improvement, the patient
relapsed and further aggressive therapy was given at a declining
rate from days 10 to 16 (atropine) and to day 23 (oxime),
respectively. In total, 92 g of pralidoxime chloride and 3912 mg of
atropine were given and the patient was discharged on the
thirty-third day with no apparent sequelae.
References to Annex 1
Ecobichon DJ, Ozere RL, Reid E, & Crocker JFS (1977) Acute
fenitrothion poisoning. Can Med Assoc J, 116: 377-379.
Kagan Ju S (1977) [Toxicology of organophosphorus pesticides],
Moscow, Meditsina, pp. 111-121, 219-233, 260-269 (in Russian).
Plestina R (1984) Prevention, diagnosis, and treatment of
insecticide poisoning, Geneva, World Health Organization
(Unpublished document VBC/84.889).
Taylor P (1980) Anticholinesterase agents. In: Goodman LS & Gilman
A, ed. The pharmacological basis of therapeutics, 6th ed., New York,
Macmillan Publishing Company, pp. 100-119.
UK DHSS (1983) Pesticide poisoning: notes for the guidance of
medical practitioners, London, United Kingdom Department of Health
and Social Security, pp. 41-47.
Vale JA & Scott GW (1974) Organophosphorus poisoning. Guy's Hosp
Rep, 123: 13-25.
Warriner RA III, Nies AS, & Hayes WJ Jr (1977) Severe
organophosphate poisoning complicated by alcohol and turpentine
ingestion. Arch environ Health, 32: 203-205.
