
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 101
HYDROQUINONE
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1996
This is a companion volume to Environmental Health Criteria 157:
Hydroquinone
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Health and safety guide for hydroquinone
(Health and safety guide ; no. 101)
1.Hydroquinones - adverse effects 2.Dermatologic agents
3.Environmental exposure 4. I.Series
ISBN 92 4 151101 X (NLM Classification: QD 341.P5)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1996
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Environmental transport, distribution, and
transformation
2.2. Environmental levels and human exposure
2.3. Kinetics and metabolism
2.4. Effects on laboratory mammals and in vitro test systems
2.5. Effects on humans
2.6. Effects on other organisms in the laboratory and field
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.2. Recommendations
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Human health hazards, prevention and protection, first
aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.2.1. Explosion hazards
4.2.2. Fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage
4.6. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) monographs produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: hydroquinone
Molecular formula: C6H4(OH)2
Chemical structure:
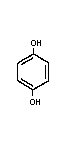 CAS chemical name: 1,4-benzenediol
Trade names: Black and White Bleaching Cream, Diak S,
Eldopoque, Eldoquin, Tecquinol, Tenox HQ
Synonyms: 1,4-benzenediol; p-benzenediol;
benzohydroquinone; benzoquinol;
1,4-dihydroxybenzene; p-dihydroxybenzene;
p-dioxobenzene; p-dioxybenzene;
hydroquinol; hydroquinole;
alpha-hydroquinone; p-hydroquinone;
p-hydroxyphenol; quinol; ß-quinol
CAS registry
number: 123-31-9
RTECS register
number: MX 3500000
1.2 Physical and Chemical Properties
Hydroquinone is a white crystalline substance when pure and is highly
soluble in water. Hydroquinone is combustible when preheated. It is a
reducing agent that is reversibly oxidized to semiquinone and quinone.
Other properties of hydroquinone are given in Table 1.
Conversion factors (at 25°C and normal atmospheric pressure)
1 ppm = 4.5 mg/m3
1 mg/m3 = 0.222 ppm
Table 1. Properties of hydroquinone
Physical state long needles
Colour white (analytical grade)
Odour odourless
Melting point 173-174°C
Boiling point 287°C
Flash point 165°C (closed cup)
Autoignition temperature 515°C
Flammability combustible when preheated
Explosion limits slight when exposed to heat; reactive at
high temperature or pressure
Vapour pressure 2.4×10-3 Pa (1.8×10-5 mmHg) at 25°C
Specific gravity 1.3 at 15°C
Vapour density 3.81
Log n-octanol/water
partition coefficient 0.59
Solubility in water (25°C) 70 g/litre
Solubility in organic
solvents (w/w at 25°C)
ethyl alcohol 57%
acetone 20%
methyl isobutyl ketone 27%
2-ethylhexanol 12%
ethyl acetate 22%
1.3 Analytical Methods
Hydroquinone in the air is sampled either by trapping in solvent or on
a mixed-cellulose-ester membrane filter.
Analysis of hydroquinone is carried out by titrimetric, colorimetric,
spectrophotometric, or, most commonly, chromatographic techniques.
1.4 Production and Uses
Hydroquinone is produced industrially in several countries. In 1979,
the total world capacity for production exceeded 40 000 tonnes, while
in 1992 it was approximately 35 000 tonnes. Hydroquinone is
extensively used as a reducing agent, as a photographic developer, as
an antioxidant for many oxidizable products, as a stabilizer or
polymerizing inhibitor for certain materials that polymerize in the
presence of free radicals, and as a chemical intermediate for the
production of antioxidants, antiozonants, agro-chemicals, and
polymers. It is a skin-lightening agent and is used in cosmetics, hair
dyes, and medical preparations.
2. SUMMARY AND EVALUATION
2.1 Environmental Transport, Distribution, and Transformation
Hydroquinone occurs in the environment as a result of man-made
processes, as well as in natural products from plants and animals.
Because of its physical and chemical properties, hydroquinone will be
distributed mainly to the water compartment when released into the
environment. It degrades as a result of both photochemical and
biological processes; consequently, it does not persist in the
environment. Bioaccumulation has not been observed.
2.2 Environmental Levels and Human Exposure
No data on hydroquinone concentrations in air, soil, or water have
been found. However, hydroquinone has been measured in mainstream
smoke from non-filter cigarettes in amounts varying from 110 to 300 µg
per cigarette, and also in sidestream smoke. Hydroquinone has been
found in plant-derived food products (e.g., wheat germ), in brewed
coffee, and in teas prepared from the leaves of some berries, where
the concentration sometimes exceeds 1%.
Amateur photographers can be exposed to hydroquinone dermally or by
inhalation. However, data on exposure levels are not available. Dermal
exposure may also result from the use of cosmetic and medical products
containing hydroquinone, such as skin lighteners. The European
Economic Community (EEC) countries have restricted its use in
cosmetics to 2% or less. In the USA, the Food and Drug Administration
has proposed concentrations between 1.5 and 2% in skin lighteners.
Concentrations up to 4% may be found in prescription drugs. In some
countries, even higher concentrations may be found in skin lighteners.
Few industrial hygiene monitoring data are available for hydroquinone.
Average concentrations in air during the manufacturing and processing
of hydroquinone have been reported to be in the range of 0.13 to
0.79 mg/m3. Occupational air exposure limits (time-weighted
average) in different countries range from 0.5 to 2 mg/m3
2.3 Kinetics and Metabolism
Hydroquinone is rapidly and extensively absorbed from the gut and
trachea of animals. Absorption via the skin is slower but may be more
rapid with vehicles such as alcohols. Hydroquinone distributes rapidly
and widely among tissues. It is metabolized to p-benzoquinone and
other oxidized products, and is detoxified by conjugation to
monoglucuronide, monosulfate, and mercapturic derivatives. The
excretion of hydroquinone and its metabolites is rapid, and occurs
primarily via the urine.
Hydroquinone and its derivatives react with different biological
components, such as macromolecules and low relative molecular mass
molecules, and have effects on cellular metabolism.
2.4 Effects on Laboratory Mammals and In Vitro Test Systems
Oral LD50 values for several animal species range between 300 and
1300 mg/kg body weight. However, LD50 values for the cat range from
42 to 86 mg/kg body weight. Acute high-level exposure to hydroquinone
causes severe effects on the central nervous system (CNS) including
hyperexcitability, tremor, convulsions, coma, and death. At sublethal
doses, these effects are reversible. The dermal LD50 value has been
estimated to be > 3800 mg/kg in rodents. Inhalation LC50 values are
not available.
A formulation containing 2% hydroquinone in a single-insult patch test
on rabbits resulted in an irritation score of 1.22 (on a scale of 0
to 4). Daily topical applications for three weeks of 2 or 5%
hydroquinone in an oil-water emulsion on the depilated skin of black
guinea-pigs caused depigmentation, inflammatory changes, and
thickening of the epidermis. The depigmentation was more marked at
higher concentrations, and female guinea-pigs were more sensitive than
males.
Sensitization tests on guinea-pigs have shown weak to strong
reactions, depending on the methods or vehicles used. The strongest
reactions were obtained with the guinea-pig maximization test. A
cross-sensitization of almost 100% between hydroquinone and
p-methoxyphenol was also seen in guinea-pigs, but only restricted
evidence of cross-reactions to p-phenylenediamine, sulfanilic acid
and p-benzoquinone was obtained.
A 6-week, oral toxicity study on male F-344 rats resulted in
nephropathy and renal cell proliferation. Thirteen-week oral gavage
studies on F-344 rats and B6C3F1 mice resulted in nephrotoxicity in
rats at 100 and 200 mg/kg, and tremors and convulsions in rats at
200 mg/kg; reduced body weight gain was seen in both rats and mice.
Dosing at 400 mg/kg was lethal in rats. In mice dosed for 13 weeks at
400 mg/kg, tremors, convulsions, and lesions in the gastric epithelium
were reported. Thirteen-week hydroquinone exposure of Sprague-Dawley
rats resulted in decreased body weight gain and CNS signs at
200 mg/kg. CNS signs were also observed at a dose level of 64 mg/kg
body weight, but not at 20 mg/kg.
Hydroquinone injected subcutaneously reduced fertility in male rats,
and prolonged the estrus cycle in female rats. However, the effects on
male rats were not found in oral studies (a dominant lethality study
and a two-generation study). In a developmental study in rats, oral
doses of 300 mg/kg body weight caused slight maternal toxicity and
reduced fetal body weight. In rabbits, the no-observed-effect level
(NOEL) for maternal toxicity was 25 mg/kg per day, and it was 75 mg/kg
per day for developmental toxicity. In a two-generation reproduction
study on rats, hydroquinone caused no reproductive effects at oral
doses of up to 150 mg/kg body weight per day. The no-observed-adverse-
effect level (NOAEL) for parental toxicity was determined to be
15 mg/kg per day; for reproductive effects through two generations, it
was 150 mg/kg per day.
Hydroquinone induces micronuclei in vivo and in vitro. Structural
and numerical chromosome aberrations have been observed in vitro and
after intraperitoneal administration in vivo. Furthermore, the
induction of gene mutations, sister-chromatid exchange, and DNA damage
has been demonstrated in vitro. Intraperitoneal injection of
hydroquinone caused chromosomal aberrations in male mouse germ cells
of the same order of magnitude as in mouse bone marrow cells.
Induction of germ-cell mutations could not be established in a
dominant lethal test on male rats dosed orally.
In a two-year study, oral administration of hydroquinone caused a
dose-related incidence of renal tubular cell adenomas in male F-344/N
rats. The incidence was statistically significant in the high-dose
group. In high-dose males, renal tubular cell hyperplasia was also
found. In female rats, a dose-related increased incidence of
mononuclear cell leukaemia occurred. Female B6C3F1 mice developed a
significantly increased incidence of hepatocellular adenomas. In
another study, hydroquinone (at a dietary level of 0.8%) produced a
significantly increased incidence of epithelial hyperplasia of the
renal papilla and a significant increase in renal tubular hyperplasia
and adenomas in male rats. No increased incidence of mononuclear cell
leukaemia was observed in female rats. In mice, the incidence of
squamous cell hyperplasia of the forestomach epithelium was
significantly increased in both sexes. In male mice, there was a
significantly increased incidence of hepatocellular adenomas and also
of renal tubular hyperplasia. A few renal cell adenomas were observed.
In vivo (intraperitoneal injection) and in vitro studies on mice
demonstrated that hydroquinone has a cytotoxic effect by reducing the
bone marrow and spleen cellularity and also an immunosuppressive
potential by inhibiting the maturation of B-lymphocytes and the
natural killer cell activity. Results also indicate that bone marrow
macrophages may be the primary target for hydroquinone myelotoxicity.
Myelotoxic effects were not observed in a long-term bioassay on
rodents.
In a 90-day study on rats using a functional-observational battery,
dose levels of 64 and 200 mg hydroquinone/kg produced tremors, and a
level of 200 mg/kg produced a depression in general activity. The
results of neuropathological examinations were negative.
2.5 Effects on Humans
Cases of intoxication have been reported after oral ingestion of
hydroquinone alone or of photographic developing agents containing
hydroquinone. The major signs of poisoning included dark urine,
vomiting, abdominal pain, tachycardia, tremors, convulsions, and coma.
Deaths have been reported after ingestion of photographic developing
agents containing hydroquinone. In a controlled oral study on human
volunteers, ingestion of 300-500 mg hydroquinone daily for 3-5 months
did not produce any observable pathological changes in the blood and
urine.
Dermal applications of hydroquinone at concentrations in different
bases of less than 3% caused negligible effects in male volunteers
from different human races. However, there are case reports suggesting
that skin lightening creams containing 2% hydroquinone have produced
leukoderma, as well as ochronosis. Hydroquinone (1% aqueous solution
or 5% cream) has caused irritation (erythema or staining). Allergic
contact dermatitis due to hydroquinone has been diagnosed.
Combined exposure to airborne concentrations of hydroquinone and
quinone causes eye irritation, sensitivity to light, injury of the
corneal epithelium, corneal ulcers, and visual disturbances. There
have been cases of appreciable loss of vision. Irritation has occurred
at exposure levels of 2.25 mg/m3 or more. Long-term exposure causes
staining of the conjunctiva and cornea, and also opacity. Slowly
developing inflammation and discoloration of the cornea and
conjunctiva have resulted after daily hydroquinone exposure, for at
least two years, to levels of 0.05-14.4 mg/m3; serious cases have
not occurred until after five or more years. One report described
cases of corneal damage occurring several years after the exposure to
hydroquinone had stopped.
There are no adequate epidemiological data to assess the
carcinogenicity of hydroquinone in humans.
2.6 Effects on Other Organisms in the Laboratory and Field
The ecotoxicological behaviour of hydroquinone has to be related to
its physical and chemical properties, which induce sensitivity to
light, pH, and dissolved oxygen. Its ecotoxicity, which is generally
high (e.g., < 1 mg/litre for aquatic organisms), varies from species
to species.
Algae, yeasts, fungi, and plants are less sensitive to hydroquinone
than the other organisms generally used for toxicity testing. However,
within the same taxonomic group, the sensitivity of different species
to hydroquinone may vary by a factor of 1000.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
The general population may be exposed to hydroquinone through
consuming plant-derived foods that contain this chemical as a natural
component, through smoking (active or passive), or through using
cosmetics and skin-lightening creams. Amateur photographers who
develop film manually may be exposed through skin contact and
inhalation.
Ingestion of large quantities may produce vomiting, convulsions, and
coma. Repeated skin contact can lead to depigmentation, allergic
contact dermatitis, and sensitization. Long-term occupational exposure
to airborne hydroquinone can result in eye irritation, sensitivity to
light, and visual disturbance.
Hydroquinone is highly toxic for most organisms in the environment,
though the toxicity varies considerably from species to species.
However, the substance is readily degraded and does not persist in the
environment.
3.2 Recommendations
a) In view of the widespread inappropriate use of skin-lightening
creams, it is recommended that over-the-counter sales of creams
containing hydroquinone be restricted. Health education programmes
should be developed to discourage the use of hydroquinone-containing
creams for whole-body skin lightening.
b) Sufficient time should be allowed for the degradation of
hydroquinone in wastewater effluent before it reaches the recipient
water.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health effects associated with certain types of exposure to
hydroquinone, together with preventive and protective measures and
first-aid recommendations, are listed in the Summary of Chemical
Safety Information (section 6).
Repeated or prolonged contact with skin may cause dermatitis and skin
sensitization. The substance may have effects on the eye and skin,
resulting in discoloration of the conjunctiva and cornea, loss of
vision, skin depigmentation, and discoloration of nails and hair.
4.1.1 Advice to physicians
At room temperature and in the presence of moisture, hydroquinone
oxidizes to quinone, which causes much worse eye irritation than
hydroquinone itself; there is a consequent risk of conjunctivitis and
corneal erosion. Lung oedema symptoms usually develop several hours
after severe inhalation exposure and are aggravated by physical
exertion; rest and hospitalization are essential. As first aid,
administration of corticosteroid spray should be considered.
In cases of dermatitis due to hydroquinone, removal from exposure will
quickly clear up the symptoms.
4.1.2 Health surveillance advice
Depending on the extent of exposure, regular medical check-ups are
advisable. Careful examination of the eyes, including visual acuity
and slit lamp examinations, should be carried out in pre-employment
and periodic examinations. The skin should also be examined.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
A hydroquinone dust cloud may explode if ignited in an enclosed area.
It is important to prevent dispersion of dust and to use a closed
system and dust explosion-proof electrical equipment and lighting.
4.2.2 Fire hazards
Hydroquinone is combustible when preheated, and forms toxic gases. It
reacts with oxidizing agents, and a violent reaction occurs with
sodium hydroxide.
Solid hydroquinone should be handled in such a way that particles do
not become airborne. In areas where it is used, there should be no
open flames and no smoking. In the event of a fire, extinguishers
containing dry chemical, alcohol-resistant foam, water, or carbon
dioxide should be used. Water used to control fires should be
contained, or diked, for subsequent disposal.
4.3 Storage
Hydroquinone should be stored in light-proof, tightly closed
containers in a cool, dark place, away from heat and oxidizing agents.
It should be labelled as corrosive.
4.4 Transport
Containers should be in good condition and labelled appropriately.
Transporters should comply with national and international
requirements regarding the transport of hazardous material.
4.5 Spillage
Rubber gloves and boots should be worn while clearing up the spillage.
The spilled substance should be swept into metal or glass fibre
containers and removed to a safe place. If available, a P2 respirator
should also be worn. Any remaining hydroquinone should be flushed away
with water, but it is important to prevent run-off entering water-
courses.
4.6 Disposal
The recommendation of the International Register of Potentially Toxic
Chemicals (IRPTC) Expert Consultation (May 1985) was:
"Incineration (1000°C, 2 seconds minimum), then scrub to remove
harmful combustion products".
The peer-review conclusions from this IRPTC Expert Consultation were:
"Oxidation produces quinone. Small amounts only: dilute to
100 mg/litre and discharge to sewer".
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Because of its physical and chemical properties, hydroquinone will be
distributed mainly to the water compartment when released into the
environment. It is, in general, highly toxic for organisms in the
environment, though the sensitivity of different species within the
same taxonomic group varies greatly. However, hydroquinone degrades as
a result of both photochemical and biological processes; consequently,
it does not persist in the environment. Bioaccumulation has not been
observed.
Contamination of the environment can be avoided by the use of suitable
methods of storage, transport, handling, and waste disposal (see
sections 4.3, 4.4, and 4.6). Sufficient time should be allowed for
hydroquinone in wastewater effluent, e.g., from photographic
processing, to degrade before it reaches the recipient water. In the
case of spillage, the clean-up methods described in section 4.5 should
be used.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hydroquinone. It should be displayed
at, or near, entrances to areas where there is potential exposure to
hydroquinone, and on processing equipment and containers. The
summary should be translated into the appropriate language(s). All
persons potentially exposed to the chemical should also have the
instructions in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
HYDROQUINONE
1,4-benzenediol
C6H4(OH)2
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 110.11 Light tan, light grey, or colourless crystals;
Melting point (°C) 173-174 the vapour mixes readily with air;
Boiling point (°C) 287 can enter the body by inhalation or ingestion or,
Flash point (closed cup) (°C) 165 to a a limited extent, through the skin;
Autoignition temperature (°C) 515 corrosive to the eyes, skin, and respiratory tract;
Relative density (15°C) 1.332 prolonged exposure to fumes, dust, or vapour can
Relative vapour density 3.81 cause lung disorders
Vapour pressure (Pa) (25°C) 2.4 × 10-3
Solubility in water (g/litre, 25°C) 70
Log P n-octanol/water 0.59
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
EYES: Corrosive; redness, pain, Wear safety goggles or face Rinse with plenty of water for at least
blurred vision shield 15 min (remove contact lenses);
obtain medical attention immediately
SKIN: Corrosive; redness, pain, Avoid skin contact; wear Remove contaminated clothing immediately;
serious burns, allergic dermatitis, protective clothing and gloves wash skin with soap and plenty of water;
sensitization obtain medical attention
INHALATION: Corrosive; Apply local exhaust or breathing Remove victim to fresh air and place in half-
coughing, breathing difficulties, protection; avoid inhalation of sitting position; obtain medical attention
headache, dizziness, nausea, vapour, particularly when immediately
diarrhoea liberated at high temperature
HYDROQUINONE (cont'd)
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
INGESTION: Corrosive; blue skin, Do not eat, drink, chew, or smoke Rinse mouth; give water to drink (ONLY IN
confusion, dizziness, headache, during work; keep out of reach of CONSCIOUS PERSONS!); obtain medical
vomiting, unconsciousness, children advice immediately
haemolytic anaemia, liver effects
ENVIRONMENT: Presents a risk Contamination of water and soil
for aquatic and soil organisms should be avoided by proper methods
of storage, transport, and waste
disposal
SPILLAGE STORAGE FIRE AND EXPLOSION
Wear rubber gloves and boots; Store in a cool, dark place in Solid hydroquinone is combustible and dust
clean up spilled substance and place light-proof, tightly-closed explosions are possible; in case of fire, keep
in metal or glass fibre containers; containers containers cool with water spray; evacuate
flush away any remainder with personnel to a safe area; use powder; water,
water (additional individual alcohol-resistant foam, or carbon dioxide to
protection: P2 respirator) extinguish fire
WASTE DISPOSAL LABELLING
Incinerate (1000°C, 2 seconds National occupational exposure limit:
minimum) then scrub to remove
harmful combustion products; United Nations No. 2662
sufficient time should be allowed
for hydroquinone in wastewater Hazard Class 6.1
effluent to degrade before reaching
recipient water Packing Class III
National Poison Control Centre:
Local trade names:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals,
taken in a certain country, can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
7.1 Previous Evaluations by International Bodies
In 1977, the International Agency for Research on Cancer (IARC)
Working Group concluded that the available data on hydroquinone did
not allow an evaluation of its carcinogenicity.
Hydroquinone was evaluated by a Nordic Expert Group for Documentation
of Occupational Exposure Limits in 1989. It was recommended that its
genotoxic effects should be given attention and also its possible
effects on the immune system, bone marrow, skin, and mucous membranes.
7.2 Exposure Limit Values
Some exposure limit values are given in the following table.
7.3 Specific Restrictions
In the European Economic Community countries, hydroquinone is
restricted for use in cosmetics to 2% or less. The US Food and Drug
Administration has issued a Notice of Proposed Rule-making for the use
of hydroquinone as a skin lightener in over-the-counter drugs at
concentrations below 1.5-2.0%.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transport of Dangerous
Goods classifies hydroquinone as a toxic substance (Hazard Class 6.1),
and, with regard to packing, as a substance presenting minor danger
(Packing Group III).
Exposure Limit Values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum permissible concentration (MPC) 1991
- Time-weighted average (TWA) 2 mg/m3
Canada Threshold limit value (TLV) 1990
- Time-weighted average (TWA) 2 mg/m3
Germany Maximum worksite concentration (MAK) 1992(r)
- Time-weighted average (TWA) 2 mg/m3
- Short-term exposure limit (STEL) 4 mg/m3
(5-min)
Russian Maximum allowable concentration (MAC) 1989
Federation - Ceiling value (aerosol) 1 mg/m3
Sweden Hygienic limit value (HLV) 1991
- Time-weighted average (TWA) 0.5 mg/m3
- Short-term exposure limit (STEL) 1.5 mg/m3
(15-min)
United Kingdom Occupational exposure standard (OES) 1992
- Time-weighted average (TWA) 2 mg/m3
- Short-term exposure limit (STEL) 4 mg/m3
(10-min TWA)
USA (ACGIH) - Threshold limit value (TLV) 1989
- Time-weighted average (TWA) 2 mg/m3
USA (NIOSH) Recommended exposure limit (REL) 1990(r)
- Ceiling value 2 mg/m3
Exposure Limit Values (cont'd)
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational USA (OSHA) Permissible exposure limit (PEL) 1990(r)
- Time-weighted average (TWA) 2 mg/m3
AIR Ambient Russian Preliminary safety level (PSL) 0.02 mg/m3 1983
Federation
WATER Surface Russian Maximum allowable concentration 1989
Federation (MAC) 0.2 mg/litre
European Economic Community legislation requires labelling as a
harmful substance using the symbol Xn.
CAS chemical name: 1,4-benzenediol
Trade names: Black and White Bleaching Cream, Diak S,
Eldopoque, Eldoquin, Tecquinol, Tenox HQ
Synonyms: 1,4-benzenediol; p-benzenediol;
benzohydroquinone; benzoquinol;
1,4-dihydroxybenzene; p-dihydroxybenzene;
p-dioxobenzene; p-dioxybenzene;
hydroquinol; hydroquinole;
alpha-hydroquinone; p-hydroquinone;
p-hydroxyphenol; quinol; ß-quinol
CAS registry
number: 123-31-9
RTECS register
number: MX 3500000
1.2 Physical and Chemical Properties
Hydroquinone is a white crystalline substance when pure and is highly
soluble in water. Hydroquinone is combustible when preheated. It is a
reducing agent that is reversibly oxidized to semiquinone and quinone.
Other properties of hydroquinone are given in Table 1.
Conversion factors (at 25°C and normal atmospheric pressure)
1 ppm = 4.5 mg/m3
1 mg/m3 = 0.222 ppm
Table 1. Properties of hydroquinone
Physical state long needles
Colour white (analytical grade)
Odour odourless
Melting point 173-174°C
Boiling point 287°C
Flash point 165°C (closed cup)
Autoignition temperature 515°C
Flammability combustible when preheated
Explosion limits slight when exposed to heat; reactive at
high temperature or pressure
Vapour pressure 2.4×10-3 Pa (1.8×10-5 mmHg) at 25°C
Specific gravity 1.3 at 15°C
Vapour density 3.81
Log n-octanol/water
partition coefficient 0.59
Solubility in water (25°C) 70 g/litre
Solubility in organic
solvents (w/w at 25°C)
ethyl alcohol 57%
acetone 20%
methyl isobutyl ketone 27%
2-ethylhexanol 12%
ethyl acetate 22%
1.3 Analytical Methods
Hydroquinone in the air is sampled either by trapping in solvent or on
a mixed-cellulose-ester membrane filter.
Analysis of hydroquinone is carried out by titrimetric, colorimetric,
spectrophotometric, or, most commonly, chromatographic techniques.
1.4 Production and Uses
Hydroquinone is produced industrially in several countries. In 1979,
the total world capacity for production exceeded 40 000 tonnes, while
in 1992 it was approximately 35 000 tonnes. Hydroquinone is
extensively used as a reducing agent, as a photographic developer, as
an antioxidant for many oxidizable products, as a stabilizer or
polymerizing inhibitor for certain materials that polymerize in the
presence of free radicals, and as a chemical intermediate for the
production of antioxidants, antiozonants, agro-chemicals, and
polymers. It is a skin-lightening agent and is used in cosmetics, hair
dyes, and medical preparations.
2. SUMMARY AND EVALUATION
2.1 Environmental Transport, Distribution, and Transformation
Hydroquinone occurs in the environment as a result of man-made
processes, as well as in natural products from plants and animals.
Because of its physical and chemical properties, hydroquinone will be
distributed mainly to the water compartment when released into the
environment. It degrades as a result of both photochemical and
biological processes; consequently, it does not persist in the
environment. Bioaccumulation has not been observed.
2.2 Environmental Levels and Human Exposure
No data on hydroquinone concentrations in air, soil, or water have
been found. However, hydroquinone has been measured in mainstream
smoke from non-filter cigarettes in amounts varying from 110 to 300 µg
per cigarette, and also in sidestream smoke. Hydroquinone has been
found in plant-derived food products (e.g., wheat germ), in brewed
coffee, and in teas prepared from the leaves of some berries, where
the concentration sometimes exceeds 1%.
Amateur photographers can be exposed to hydroquinone dermally or by
inhalation. However, data on exposure levels are not available. Dermal
exposure may also result from the use of cosmetic and medical products
containing hydroquinone, such as skin lighteners. The European
Economic Community (EEC) countries have restricted its use in
cosmetics to 2% or less. In the USA, the Food and Drug Administration
has proposed concentrations between 1.5 and 2% in skin lighteners.
Concentrations up to 4% may be found in prescription drugs. In some
countries, even higher concentrations may be found in skin lighteners.
Few industrial hygiene monitoring data are available for hydroquinone.
Average concentrations in air during the manufacturing and processing
of hydroquinone have been reported to be in the range of 0.13 to
0.79 mg/m3. Occupational air exposure limits (time-weighted
average) in different countries range from 0.5 to 2 mg/m3
2.3 Kinetics and Metabolism
Hydroquinone is rapidly and extensively absorbed from the gut and
trachea of animals. Absorption via the skin is slower but may be more
rapid with vehicles such as alcohols. Hydroquinone distributes rapidly
and widely among tissues. It is metabolized to p-benzoquinone and
other oxidized products, and is detoxified by conjugation to
monoglucuronide, monosulfate, and mercapturic derivatives. The
excretion of hydroquinone and its metabolites is rapid, and occurs
primarily via the urine.
Hydroquinone and its derivatives react with different biological
components, such as macromolecules and low relative molecular mass
molecules, and have effects on cellular metabolism.
2.4 Effects on Laboratory Mammals and In Vitro Test Systems
Oral LD50 values for several animal species range between 300 and
1300 mg/kg body weight. However, LD50 values for the cat range from
42 to 86 mg/kg body weight. Acute high-level exposure to hydroquinone
causes severe effects on the central nervous system (CNS) including
hyperexcitability, tremor, convulsions, coma, and death. At sublethal
doses, these effects are reversible. The dermal LD50 value has been
estimated to be > 3800 mg/kg in rodents. Inhalation LC50 values are
not available.
A formulation containing 2% hydroquinone in a single-insult patch test
on rabbits resulted in an irritation score of 1.22 (on a scale of 0
to 4). Daily topical applications for three weeks of 2 or 5%
hydroquinone in an oil-water emulsion on the depilated skin of black
guinea-pigs caused depigmentation, inflammatory changes, and
thickening of the epidermis. The depigmentation was more marked at
higher concentrations, and female guinea-pigs were more sensitive than
males.
Sensitization tests on guinea-pigs have shown weak to strong
reactions, depending on the methods or vehicles used. The strongest
reactions were obtained with the guinea-pig maximization test. A
cross-sensitization of almost 100% between hydroquinone and
p-methoxyphenol was also seen in guinea-pigs, but only restricted
evidence of cross-reactions to p-phenylenediamine, sulfanilic acid
and p-benzoquinone was obtained.
A 6-week, oral toxicity study on male F-344 rats resulted in
nephropathy and renal cell proliferation. Thirteen-week oral gavage
studies on F-344 rats and B6C3F1 mice resulted in nephrotoxicity in
rats at 100 and 200 mg/kg, and tremors and convulsions in rats at
200 mg/kg; reduced body weight gain was seen in both rats and mice.
Dosing at 400 mg/kg was lethal in rats. In mice dosed for 13 weeks at
400 mg/kg, tremors, convulsions, and lesions in the gastric epithelium
were reported. Thirteen-week hydroquinone exposure of Sprague-Dawley
rats resulted in decreased body weight gain and CNS signs at
200 mg/kg. CNS signs were also observed at a dose level of 64 mg/kg
body weight, but not at 20 mg/kg.
Hydroquinone injected subcutaneously reduced fertility in male rats,
and prolonged the estrus cycle in female rats. However, the effects on
male rats were not found in oral studies (a dominant lethality study
and a two-generation study). In a developmental study in rats, oral
doses of 300 mg/kg body weight caused slight maternal toxicity and
reduced fetal body weight. In rabbits, the no-observed-effect level
(NOEL) for maternal toxicity was 25 mg/kg per day, and it was 75 mg/kg
per day for developmental toxicity. In a two-generation reproduction
study on rats, hydroquinone caused no reproductive effects at oral
doses of up to 150 mg/kg body weight per day. The no-observed-adverse-
effect level (NOAEL) for parental toxicity was determined to be
15 mg/kg per day; for reproductive effects through two generations, it
was 150 mg/kg per day.
Hydroquinone induces micronuclei in vivo and in vitro. Structural
and numerical chromosome aberrations have been observed in vitro and
after intraperitoneal administration in vivo. Furthermore, the
induction of gene mutations, sister-chromatid exchange, and DNA damage
has been demonstrated in vitro. Intraperitoneal injection of
hydroquinone caused chromosomal aberrations in male mouse germ cells
of the same order of magnitude as in mouse bone marrow cells.
Induction of germ-cell mutations could not be established in a
dominant lethal test on male rats dosed orally.
In a two-year study, oral administration of hydroquinone caused a
dose-related incidence of renal tubular cell adenomas in male F-344/N
rats. The incidence was statistically significant in the high-dose
group. In high-dose males, renal tubular cell hyperplasia was also
found. In female rats, a dose-related increased incidence of
mononuclear cell leukaemia occurred. Female B6C3F1 mice developed a
significantly increased incidence of hepatocellular adenomas. In
another study, hydroquinone (at a dietary level of 0.8%) produced a
significantly increased incidence of epithelial hyperplasia of the
renal papilla and a significant increase in renal tubular hyperplasia
and adenomas in male rats. No increased incidence of mononuclear cell
leukaemia was observed in female rats. In mice, the incidence of
squamous cell hyperplasia of the forestomach epithelium was
significantly increased in both sexes. In male mice, there was a
significantly increased incidence of hepatocellular adenomas and also
of renal tubular hyperplasia. A few renal cell adenomas were observed.
In vivo (intraperitoneal injection) and in vitro studies on mice
demonstrated that hydroquinone has a cytotoxic effect by reducing the
bone marrow and spleen cellularity and also an immunosuppressive
potential by inhibiting the maturation of B-lymphocytes and the
natural killer cell activity. Results also indicate that bone marrow
macrophages may be the primary target for hydroquinone myelotoxicity.
Myelotoxic effects were not observed in a long-term bioassay on
rodents.
In a 90-day study on rats using a functional-observational battery,
dose levels of 64 and 200 mg hydroquinone/kg produced tremors, and a
level of 200 mg/kg produced a depression in general activity. The
results of neuropathological examinations were negative.
2.5 Effects on Humans
Cases of intoxication have been reported after oral ingestion of
hydroquinone alone or of photographic developing agents containing
hydroquinone. The major signs of poisoning included dark urine,
vomiting, abdominal pain, tachycardia, tremors, convulsions, and coma.
Deaths have been reported after ingestion of photographic developing
agents containing hydroquinone. In a controlled oral study on human
volunteers, ingestion of 300-500 mg hydroquinone daily for 3-5 months
did not produce any observable pathological changes in the blood and
urine.
Dermal applications of hydroquinone at concentrations in different
bases of less than 3% caused negligible effects in male volunteers
from different human races. However, there are case reports suggesting
that skin lightening creams containing 2% hydroquinone have produced
leukoderma, as well as ochronosis. Hydroquinone (1% aqueous solution
or 5% cream) has caused irritation (erythema or staining). Allergic
contact dermatitis due to hydroquinone has been diagnosed.
Combined exposure to airborne concentrations of hydroquinone and
quinone causes eye irritation, sensitivity to light, injury of the
corneal epithelium, corneal ulcers, and visual disturbances. There
have been cases of appreciable loss of vision. Irritation has occurred
at exposure levels of 2.25 mg/m3 or more. Long-term exposure causes
staining of the conjunctiva and cornea, and also opacity. Slowly
developing inflammation and discoloration of the cornea and
conjunctiva have resulted after daily hydroquinone exposure, for at
least two years, to levels of 0.05-14.4 mg/m3; serious cases have
not occurred until after five or more years. One report described
cases of corneal damage occurring several years after the exposure to
hydroquinone had stopped.
There are no adequate epidemiological data to assess the
carcinogenicity of hydroquinone in humans.
2.6 Effects on Other Organisms in the Laboratory and Field
The ecotoxicological behaviour of hydroquinone has to be related to
its physical and chemical properties, which induce sensitivity to
light, pH, and dissolved oxygen. Its ecotoxicity, which is generally
high (e.g., < 1 mg/litre for aquatic organisms), varies from species
to species.
Algae, yeasts, fungi, and plants are less sensitive to hydroquinone
than the other organisms generally used for toxicity testing. However,
within the same taxonomic group, the sensitivity of different species
to hydroquinone may vary by a factor of 1000.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
The general population may be exposed to hydroquinone through
consuming plant-derived foods that contain this chemical as a natural
component, through smoking (active or passive), or through using
cosmetics and skin-lightening creams. Amateur photographers who
develop film manually may be exposed through skin contact and
inhalation.
Ingestion of large quantities may produce vomiting, convulsions, and
coma. Repeated skin contact can lead to depigmentation, allergic
contact dermatitis, and sensitization. Long-term occupational exposure
to airborne hydroquinone can result in eye irritation, sensitivity to
light, and visual disturbance.
Hydroquinone is highly toxic for most organisms in the environment,
though the toxicity varies considerably from species to species.
However, the substance is readily degraded and does not persist in the
environment.
3.2 Recommendations
a) In view of the widespread inappropriate use of skin-lightening
creams, it is recommended that over-the-counter sales of creams
containing hydroquinone be restricted. Health education programmes
should be developed to discourage the use of hydroquinone-containing
creams for whole-body skin lightening.
b) Sufficient time should be allowed for the degradation of
hydroquinone in wastewater effluent before it reaches the recipient
water.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health effects associated with certain types of exposure to
hydroquinone, together with preventive and protective measures and
first-aid recommendations, are listed in the Summary of Chemical
Safety Information (section 6).
Repeated or prolonged contact with skin may cause dermatitis and skin
sensitization. The substance may have effects on the eye and skin,
resulting in discoloration of the conjunctiva and cornea, loss of
vision, skin depigmentation, and discoloration of nails and hair.
4.1.1 Advice to physicians
At room temperature and in the presence of moisture, hydroquinone
oxidizes to quinone, which causes much worse eye irritation than
hydroquinone itself; there is a consequent risk of conjunctivitis and
corneal erosion. Lung oedema symptoms usually develop several hours
after severe inhalation exposure and are aggravated by physical
exertion; rest and hospitalization are essential. As first aid,
administration of corticosteroid spray should be considered.
In cases of dermatitis due to hydroquinone, removal from exposure will
quickly clear up the symptoms.
4.1.2 Health surveillance advice
Depending on the extent of exposure, regular medical check-ups are
advisable. Careful examination of the eyes, including visual acuity
and slit lamp examinations, should be carried out in pre-employment
and periodic examinations. The skin should also be examined.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
A hydroquinone dust cloud may explode if ignited in an enclosed area.
It is important to prevent dispersion of dust and to use a closed
system and dust explosion-proof electrical equipment and lighting.
4.2.2 Fire hazards
Hydroquinone is combustible when preheated, and forms toxic gases. It
reacts with oxidizing agents, and a violent reaction occurs with
sodium hydroxide.
Solid hydroquinone should be handled in such a way that particles do
not become airborne. In areas where it is used, there should be no
open flames and no smoking. In the event of a fire, extinguishers
containing dry chemical, alcohol-resistant foam, water, or carbon
dioxide should be used. Water used to control fires should be
contained, or diked, for subsequent disposal.
4.3 Storage
Hydroquinone should be stored in light-proof, tightly closed
containers in a cool, dark place, away from heat and oxidizing agents.
It should be labelled as corrosive.
4.4 Transport
Containers should be in good condition and labelled appropriately.
Transporters should comply with national and international
requirements regarding the transport of hazardous material.
4.5 Spillage
Rubber gloves and boots should be worn while clearing up the spillage.
The spilled substance should be swept into metal or glass fibre
containers and removed to a safe place. If available, a P2 respirator
should also be worn. Any remaining hydroquinone should be flushed away
with water, but it is important to prevent run-off entering water-
courses.
4.6 Disposal
The recommendation of the International Register of Potentially Toxic
Chemicals (IRPTC) Expert Consultation (May 1985) was:
"Incineration (1000°C, 2 seconds minimum), then scrub to remove
harmful combustion products".
The peer-review conclusions from this IRPTC Expert Consultation were:
"Oxidation produces quinone. Small amounts only: dilute to
100 mg/litre and discharge to sewer".
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Because of its physical and chemical properties, hydroquinone will be
distributed mainly to the water compartment when released into the
environment. It is, in general, highly toxic for organisms in the
environment, though the sensitivity of different species within the
same taxonomic group varies greatly. However, hydroquinone degrades as
a result of both photochemical and biological processes; consequently,
it does not persist in the environment. Bioaccumulation has not been
observed.
Contamination of the environment can be avoided by the use of suitable
methods of storage, transport, handling, and waste disposal (see
sections 4.3, 4.4, and 4.6). Sufficient time should be allowed for
hydroquinone in wastewater effluent, e.g., from photographic
processing, to degrade before it reaches the recipient water. In the
case of spillage, the clean-up methods described in section 4.5 should
be used.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hydroquinone. It should be displayed
at, or near, entrances to areas where there is potential exposure to
hydroquinone, and on processing equipment and containers. The
summary should be translated into the appropriate language(s). All
persons potentially exposed to the chemical should also have the
instructions in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
HYDROQUINONE
1,4-benzenediol
C6H4(OH)2
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 110.11 Light tan, light grey, or colourless crystals;
Melting point (°C) 173-174 the vapour mixes readily with air;
Boiling point (°C) 287 can enter the body by inhalation or ingestion or,
Flash point (closed cup) (°C) 165 to a a limited extent, through the skin;
Autoignition temperature (°C) 515 corrosive to the eyes, skin, and respiratory tract;
Relative density (15°C) 1.332 prolonged exposure to fumes, dust, or vapour can
Relative vapour density 3.81 cause lung disorders
Vapour pressure (Pa) (25°C) 2.4 × 10-3
Solubility in water (g/litre, 25°C) 70
Log P n-octanol/water 0.59
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
EYES: Corrosive; redness, pain, Wear safety goggles or face Rinse with plenty of water for at least
blurred vision shield 15 min (remove contact lenses);
obtain medical attention immediately
SKIN: Corrosive; redness, pain, Avoid skin contact; wear Remove contaminated clothing immediately;
serious burns, allergic dermatitis, protective clothing and gloves wash skin with soap and plenty of water;
sensitization obtain medical attention
INHALATION: Corrosive; Apply local exhaust or breathing Remove victim to fresh air and place in half-
coughing, breathing difficulties, protection; avoid inhalation of sitting position; obtain medical attention
headache, dizziness, nausea, vapour, particularly when immediately
diarrhoea liberated at high temperature
HYDROQUINONE (cont'd)
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
INGESTION: Corrosive; blue skin, Do not eat, drink, chew, or smoke Rinse mouth; give water to drink (ONLY IN
confusion, dizziness, headache, during work; keep out of reach of CONSCIOUS PERSONS!); obtain medical
vomiting, unconsciousness, children advice immediately
haemolytic anaemia, liver effects
ENVIRONMENT: Presents a risk Contamination of water and soil
for aquatic and soil organisms should be avoided by proper methods
of storage, transport, and waste
disposal
SPILLAGE STORAGE FIRE AND EXPLOSION
Wear rubber gloves and boots; Store in a cool, dark place in Solid hydroquinone is combustible and dust
clean up spilled substance and place light-proof, tightly-closed explosions are possible; in case of fire, keep
in metal or glass fibre containers; containers containers cool with water spray; evacuate
flush away any remainder with personnel to a safe area; use powder; water,
water (additional individual alcohol-resistant foam, or carbon dioxide to
protection: P2 respirator) extinguish fire
WASTE DISPOSAL LABELLING
Incinerate (1000°C, 2 seconds National occupational exposure limit:
minimum) then scrub to remove
harmful combustion products; United Nations No. 2662
sufficient time should be allowed
for hydroquinone in wastewater Hazard Class 6.1
effluent to degrade before reaching
recipient water Packing Class III
National Poison Control Centre:
Local trade names:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals,
taken in a certain country, can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
7.1 Previous Evaluations by International Bodies
In 1977, the International Agency for Research on Cancer (IARC)
Working Group concluded that the available data on hydroquinone did
not allow an evaluation of its carcinogenicity.
Hydroquinone was evaluated by a Nordic Expert Group for Documentation
of Occupational Exposure Limits in 1989. It was recommended that its
genotoxic effects should be given attention and also its possible
effects on the immune system, bone marrow, skin, and mucous membranes.
7.2 Exposure Limit Values
Some exposure limit values are given in the following table.
7.3 Specific Restrictions
In the European Economic Community countries, hydroquinone is
restricted for use in cosmetics to 2% or less. The US Food and Drug
Administration has issued a Notice of Proposed Rule-making for the use
of hydroquinone as a skin lightener in over-the-counter drugs at
concentrations below 1.5-2.0%.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transport of Dangerous
Goods classifies hydroquinone as a toxic substance (Hazard Class 6.1),
and, with regard to packing, as a substance presenting minor danger
(Packing Group III).
Exposure Limit Values
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum permissible concentration (MPC) 1991
- Time-weighted average (TWA) 2 mg/m3
Canada Threshold limit value (TLV) 1990
- Time-weighted average (TWA) 2 mg/m3
Germany Maximum worksite concentration (MAK) 1992(r)
- Time-weighted average (TWA) 2 mg/m3
- Short-term exposure limit (STEL) 4 mg/m3
(5-min)
Russian Maximum allowable concentration (MAC) 1989
Federation - Ceiling value (aerosol) 1 mg/m3
Sweden Hygienic limit value (HLV) 1991
- Time-weighted average (TWA) 0.5 mg/m3
- Short-term exposure limit (STEL) 1.5 mg/m3
(15-min)
United Kingdom Occupational exposure standard (OES) 1992
- Time-weighted average (TWA) 2 mg/m3
- Short-term exposure limit (STEL) 4 mg/m3
(10-min TWA)
USA (ACGIH) - Threshold limit value (TLV) 1989
- Time-weighted average (TWA) 2 mg/m3
USA (NIOSH) Recommended exposure limit (REL) 1990(r)
- Ceiling value 2 mg/m3
Exposure Limit Values (cont'd)
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational USA (OSHA) Permissible exposure limit (PEL) 1990(r)
- Time-weighted average (TWA) 2 mg/m3
AIR Ambient Russian Preliminary safety level (PSL) 0.02 mg/m3 1983
Federation
WATER Surface Russian Maximum allowable concentration 1989
Federation (MAC) 0.2 mg/litre
European Economic Community legislation requires labelling as a
harmful substance using the symbol Xn.
 The following label statements are required:
R 20/22 Harmful by inhalation and if swallowed
S 2 Keep out of reach of children
S 24/25 Avoid contact with skin and eyes
S 39 Wear eye/face protection
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CEC/IPCS (1991) International Chemical Safety Card 166: Hydroquinone.
Luxembourg, Commission of the European Communities.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2B. New York, John Wiley & Sons.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1991) Chemical safety sheets.
Kluwer Academic Publishers, Samson Chemical Publishers, Dutch
Institute for the Working Environment, Dutch Chemical Industry
Association.
GOSSELIN, R.E., HODGE, H.C., SMITH, R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
IPCS (1994) Environmental Health Criteria 157: Hydroquinone. Geneva,
World Health Organization.
IRPTC (1992-1993) Legal file. Geneva, International Register of
Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Health Administration.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labour (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication
No. 85.114).
The following label statements are required:
R 20/22 Harmful by inhalation and if swallowed
S 2 Keep out of reach of children
S 24/25 Avoid contact with skin and eyes
S 39 Wear eye/face protection
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CEC/IPCS (1991) International Chemical Safety Card 166: Hydroquinone.
Luxembourg, Commission of the European Communities.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2B. New York, John Wiley & Sons.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1991) Chemical safety sheets.
Kluwer Academic Publishers, Samson Chemical Publishers, Dutch
Institute for the Working Environment, Dutch Chemical Industry
Association.
GOSSELIN, R.E., HODGE, H.C., SMITH, R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
IPCS (1994) Environmental Health Criteria 157: Hydroquinone. Geneva,
World Health Organization.
IRPTC (1992-1993) Legal file. Geneva, International Register of
Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Health Administration.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labour (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication
No. 85.114).
 CAS chemical name: 1,4-benzenediol
Trade names: Black and White Bleaching Cream, Diak S,
Eldopoque, Eldoquin, Tecquinol, Tenox HQ
Synonyms: 1,4-benzenediol; p-benzenediol;
benzohydroquinone; benzoquinol;
1,4-dihydroxybenzene; p-dihydroxybenzene;
p-dioxobenzene; p-dioxybenzene;
hydroquinol; hydroquinole;
alpha-hydroquinone; p-hydroquinone;
p-hydroxyphenol; quinol; ß-quinol
CAS registry
number:
CAS chemical name: 1,4-benzenediol
Trade names: Black and White Bleaching Cream, Diak S,
Eldopoque, Eldoquin, Tecquinol, Tenox HQ
Synonyms: 1,4-benzenediol; p-benzenediol;
benzohydroquinone; benzoquinol;
1,4-dihydroxybenzene; p-dihydroxybenzene;
p-dioxobenzene; p-dioxybenzene;
hydroquinol; hydroquinole;
alpha-hydroquinone; p-hydroquinone;
p-hydroxyphenol; quinol; ß-quinol
CAS registry
number:  The following label statements are required:
R 20/22 Harmful by inhalation and if swallowed
S 2 Keep out of reach of children
S 24/25 Avoid contact with skin and eyes
S 39 Wear eye/face protection
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CEC/IPCS (1991) International Chemical Safety Card 166: Hydroquinone.
Luxembourg, Commission of the European Communities.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2B. New York, John Wiley & Sons.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1991) Chemical safety sheets.
Kluwer Academic Publishers, Samson Chemical Publishers, Dutch
Institute for the Working Environment, Dutch Chemical Industry
Association.
GOSSELIN, R.E., HODGE, H.C., SMITH, R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
IPCS (1994) Environmental Health Criteria 157: Hydroquinone. Geneva,
World Health Organization.
IRPTC (1992-1993) Legal file. Geneva, International Register of
Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Health Administration.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labour (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication
No. 85.114).
The following label statements are required:
R 20/22 Harmful by inhalation and if swallowed
S 2 Keep out of reach of children
S 24/25 Avoid contact with skin and eyes
S 39 Wear eye/face protection
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CEC/IPCS (1991) International Chemical Safety Card 166: Hydroquinone.
Luxembourg, Commission of the European Communities.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2B. New York, John Wiley & Sons.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1991) Chemical safety sheets.
Kluwer Academic Publishers, Samson Chemical Publishers, Dutch
Institute for the Working Environment, Dutch Chemical Industry
Association.
GOSSELIN, R.E., HODGE, H.C., SMITH, R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
IPCS (1994) Environmental Health Criteria 157: Hydroquinone. Geneva,
World Health Organization.
IRPTC (1992-1993) Legal file. Geneva, International Register of
Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Health Administration.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labour (Publication No. DHHS (NIOSH)
01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Health Administration (Publication
No. 85.114).
