
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 96
WARFARIN
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1995
This is a companion volume to Environmental Health Criteria 175:
Anticoagulant Rodenticides
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Health and safety guide for Warfarin
(Health and safety guide ; no. 96)
1.Warfarin - toxicity 2.Rodenticides 3.Anticoagulants
4.Environmental exposure I.Series
ISBN 92 4 151096 X (NLM Classification: WA 240)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1995
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Identity, physical and chemical properties, and
analytical methods
2.2. Sources of human and environmental exposure
2.3. Environmental transport, distribution, and
transformation
2.4. Environmental levels and human exposure
2.5. Kinetics and metabolism in laboratory animals and humans
2.6. Effects on laboratory mammals and in vitro test
systems
2.7. Effects on humans
2.8. Effects on other organisms in the laboratory and field
2.9. Evaluation of human health risks and effects on the
environment
2.9.1. Evaluation of human health risks
2.9.2. Evaluation of effects on the environment
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.2. Recommendations for the protection of human health and
the environment
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage
4.6. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) monographs produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: warfarin
Chemical formula: C19H16O4
Chemical structure:
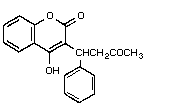 Common synonyms: coumafène, warfarine, zoocoumarin
Common trade names: Athrombine-K; Brumolin; Compound 42;
Coumadin; Coumafen; Coumarin;
Coumefene; Dethmore; Dethnel; Eastern
States Duocide; Fasco Fascrat Powder;
Frass-Ratron; Kumader; Kumadu; Kypfarin;
Maag Rattentod Cum; Mar-Frin; Maveran;
Panwarfin; Prothromadin; Rat-a-way;
Rat-b-gon; Rat-Gard; Rat-Kill; Rat-Mix;
Rat-ola; Ratro; Rats-No-More; Rodafarin;
Temus W; Warf 42; Warf Compound 42;
Warf-12; Warfarat; Warfarin +;
Warficide; Zoocoumarin
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-
2 H-1-benzopyran-2-one (9CI) (8CI) (I)
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl)
coumarin
CAS registry number: 81-81-2 (unstated stereochemistry);
5543-58-8 (R)-(+) isomer
RTECS registry number: GN4550000
1.2 Physical and Chemical Properties
Warfarin forms colourless crystals. It is practically insoluble in
water, readily soluble in acetone and dioxane and moderately soluble
in alcohols. It is acidic. The sodium salts are soluble in water but
insoluble in organic solvents.
Further physical and chemical properties of warfarin are given in the
"Summary of Chemical Safety Information" (section 6).
1.3 Analytical Methods
The determination of warfarin is mainly based on high-performance
liquid chromatography with fluorescence detection. The detection limit
in animal tissues is 0.02 mg/kg. The other methods include UV
spectrometry and reversed-phase liquid chromatography, the latter with
a detection limit in blood serum of 20 µg/litre.
1.4 Production and Uses
The anticoagulant properties of warfarin were reported in 1944, and,
in 1948, it was proposed as a rodenticide. Warfarin is used in
agriculture and urban rodent control in the form of baits containing
0.025% active ingredient. The sodium salt is available at 0.5%
concentrate for use at a final concentration of 0.05% in liquid base.
Warfarin is also used as a drug for the treatment of thromboembolic
disease in humans.
2. SUMMARY AND EVALUATION
2.1 Identity, Physical and Chemical Properties, and Analytical
Methods
Warfarin is a colourless, crystalline compound with a melting point of
151-161°C. It is practically insoluble in water, readily soluble in
acetone and dioxane, and moderately soluble in alcohols. Analytical
methods for the determination of warfarin are mainly based on high
performance liquid chromatography.
2.2 Sources of Human and Environmental Exposure
Warfarin does not occur naturally. It is used as a rodenticide as well
as a drug, and acts by disrupting the normal blood clotting mechanisms
causing an increased tendency to bleed.
2.3 Environmental Transport, Distribution, and Transformation
Warfarin is unlikely to enter the atmosphere, because of its low
volatility. It is practically insoluble in water. The rate of
degradation is relatively slow.
2.4 Environmental Levels and Human Exposure
No information is available on concentrations in air, water, and soil.
Warfarin is not intended for direct application to growing crops or
for use as a food additive. The controlled medical use of warfarin
exposes more people to higher concentations over a longer period than
would be expected to occur as a result of accidental human exposure
due to its use as a rodenticide. Accidental overexposures to warfarin
as a rodenticide are most commonly reported in children and domestic
animals.
2.5 Kinetics and Metabolism in Laboratory Animals and Humans
Warfarin is easily absorbed through the gastrointestinal tract and
skin, and is bound to serum proteins. Warfarin is readily hydroxylated
in vitro and in vivo by rat liver microsomal enzymes to form 6-,
7-, and 8-hydroxywarfarin. It is eliminated through both the urine and
faeces.
When a single oral dose of 1.5 mg warfarin/kg was given to male and
female volunteers, maximum concentration in plasma was reached in
2-12 h. The half-life for disappearance from human plasma ranged from
15 to 58 h.
2.6 Effects on Laboratory Mammals and in vitro Test Systems
The acute oral toxicity of warfarin for rats is high. Reported LD50
values range from 11 to 323 mg/kg, females being more susceptible than
males. For classification purposes, the accepted WHO LD50 value is
10 mg/kg. Genetic resistance to warfarin among rodents and humans has
been reported. Signs of poisoning are those associated with an
increased tendency to bleed.
One study on rats showed some developmental effects. There are no data
on mutagenicity.
In feeding studies on rats, the only effect found was associated with
anticoagulant action.
2.7 Effects on Humans
Vitamin K functions as an essential element in the synthesis of
several blood coagulation factors. Warfarin inhibits this process and
consequently affects the blood coagulation mechanisms. Prolonged
inhibition of the vitamin K synthesis will lead to severe bleeding and
death, if not corrected.
Symptoms of warfarin poisoning begin a few days or weeks after
ingestion. They include nose bleeding, bleeding gums, pallor, and,
sometimes, haematomas around joints and on the buttocks, and blood in
the urine and faeces. Later, paralysis due to cerebral haemorrhage
and, finally, haemorrhagic shock and death may occur.
Poisoning incidents have been reported. Outbreaks of poisoning have
been observed in relation to warfarin-contaminated meal and also in
infants after dermal application of warfarin-contaminated talc. A case
of poisoning from prolonged skin contact during the preparation of
warfarin baits has also been reported.
Developmental effects known as "warfarin embryopathy" or "fetal
warfarin syndrome" were reported when warfarin was administered as a
therapeutic agent during pregnancy. No cases of embryopathy following
the use of warfarin as a rodenticide have been reported.
2.8 Effects on other Organisms in the Laboratory and Field
Some secondary toxicity laboratory studies on wildlife have shown that
captive predators could be intoxicated by sufficient no-choice feeding
of warfarin-poisoned rodents.
2.9 Evaluation of Human Health Risks and Effects on the Environment
2.9.1 Evaluation of human health risks
Warfarin is used as both a rodenticide and a drug. In agriculture and
urban rodent control, it is used as a tracking dust or bait containing
0.025-0.05% of active ingredient. Increased levels in air, water, and
food are unlikely. Occupational exposure may occur during manufacture,
formulation, and bait application, but data concerning the levels are
not available.
Warfarin is easily absorbed through the gastrointestinal tract and
skin, and readily hydroxylated by liver microsomal enzymes in the rat.
The half-life for disappearance of warfarin from human plasma has been
reported to be between 15 and 58 h.
Warfarin is highly toxic for mammalian species. Signs of poisoning in
all species, including humans, are associated with an increased
tendency to bleed.
Poisoning incidents after massive oral or dermal exposure have been
reported. When used therapeutically, warfarin has been given to
patients for long periods without signs of toxicity, other than
bleeding. Warfarin is known as a human teratogen. Developmental
effects have been observed following the administration of warfarin as
a therapeutic agent during pregnancy. No cases of embryopathy
following its use as a rodenticide have been reported.
The specific and potent antidote in case of poisoning is vitamin K1.
2.9.2 Evaluation of effects on the environment
Warfarin is applied to discrete sites as low-concentration baits. It
is practically insoluble in water and, when it is used as a
rodenticide, it is unlikely to be a source of environmental
contamination.
Non-target organisms are potentially at risk in two ways: from direct
consumption of baits (primary hazard) and through eating poisoned
rodents (secondary hazard). The main reason for the poisoning of
domestic animals is direct consumption of baits.
Some secondary toxicity laboratory studies have shown that predators
can be intoxicated by eating warfarin-poisoned rodents. No cases of
secondary poisoning of predators in the field following the use of
warfarin as a rodenticide have been found.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Exposure of the general population to warfarin as a rodenticide
through air, drinking-water, or food is unlikely and does not
constitute a significant health hazard. Poisoning incidents may occur
in cases of massive intentional or unintentional ingestion, massive
dermal exposure, and during manufacture and formulation. Warfarin is
teratogenic in humans at therapeutic doses. The use of warfarin as a
rodenticide is not expected to be a significant source of air, water,
soil, or food contamination. Direct or secondary poisoning of birds,
domestic and farm animals, and wildlife may occur.
3.2 Recommendations for the Protection of Human Health and the
Environment
Potentially exposed workers should receive appropriate biomonitoring
and health evaluation.
Exposure of pregnant women to concentrate material must be kept to a
minimum. Uneaten baits as well as killed rodents should be burned or
buried to reduce the risk of primary or secondary poisonings in
non-target organisms.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
Warfarin is highly toxic for mammalian species, including humans. The
probable lethal oral dose in humans is believed to be between 50 and
500 mg/kg. A cumulative total dose of about 1000 mg of warfarin
consumed in 13 days has been reported to be fatal for an adult man,
equivalent to about 1.1 mg/kg per day.
The main features of warfarin poisoning in less severe cases are
excessive bruising, nose and gum bleeding, and blood in the urine and
faeces. Bleeding from several organs within the body, leading to shock
and possibly death, occurs in the more severe cases. The onset of the
signs of poisoning may not be evident until a few days after exposure.
When handling technical material or powder concentrates, full air-fed
protection and an impervious suit suitable for wash-down are
necessary. Operations with liquid concentrates require the use of PVC
or nitrile-rubber gloves, armlets, and an apron, with a face shield
and rubber boots.
All persons who are bleeding, or suspected to be heavily exposed, must
receive medical attention.
4.1.1 Advice to physicians
If poisoning is recent (within a few hours), gastric lavage is
recommended. Administration of active charcoal in repeated doses is
also recommended.
Take a venous blood sample for measurement of the haemoglobin level,
prothrombin time, blood grouping, and cross-matching.
In less severe cases of poisoning give vitamin K1 (phytomenadione)
(5-10 mg), by slow intravenous infusion, 3 times daily on the first
day. After initial intravenous administration, oral treatment can be
continued until normalization of prothrombin tests.
In more severe cases, transfusion of matched whole blood or fresh,
frozen plasma should also be given and repeated daily until
prothrombin tests have returned to normal. Administration of factor
concentrate may be considered to prevent a volume overload.
Prothrombin tests should be made at least twice daily until a return
to normal is clearly established.
Keep the patient in hospital until the prothrombin time has remained
normal for three days.
4.1.2 Health surveillance advice
Workers handling concentrates should undergo periodic determinations
of blood prothrombin time. Persons with a history of blood disorders
with bleeding tendencies would be expected to be at increased risk in
case of exposure.
4.2 Explosion and Fire Hazards
Flash point and flammable limits have not been found. Contact with
strong oxidizers may cause fires and explosions. Recommended
extinguishers are foam, carbon dioxide, or dry chemicals.
High temperature decomposition or burning may lead to the formation of
toxic gases and vapours. Wear full protective clothing and
self-contained breathing apparatus in fire-fighting.
Run-off water from the fire should be prevented from entering
surface-water drains or water courses.
4.3 Storage
Technical material and formulations should be stored in sealed
containers in locked, well-ventilated, dry areas away from frost,
direct sunlight, and sources of heat and ignition. Keep products out
of reach of children, unauthorized personnel, and domestic animals. Do
not store near food or animal feed.
4.4 Transport
Comply with any local regulations regarding the movement of hazardous
goods. Before despatch ensure that the containers are sound and that
labels are securely fixed and undamaged.
4.5 Spillage
During decontamination, the operator must wear protective clothing,
PVC or nitrile gloves, a face shield, and rubber boots.
Small spills should be collected and disposed of as toxic waste
according to local legislation.
Large quantities may be destroyed by dissolving in a flammable solvent
(e.g., alcohol) and atomizing in a combustion chamber.
Contaminated areas should be washed down with cold water containing
surfactant or detergent; the washings must be prevented from entering
surface water and drains.
4.6 Disposal
No specific data are available.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Warfarin is practically insoluble in water. It is unlikely to be a
source of environmental contamination.
Do not place baits where domestic or farm animals and birds can reach
them. Burn or bury any uneaten bait. Do not dump it in water. Look for
dead rats and mice and burn or bury them.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, warfarin. It should be displayed at, or
near, entrances to areas where there is potential exposure to
warfarin, and on processing equipment and containers. The summary
should be translated into the appropriate language(s). All persons
potentially exposed to the chemical should also have the instructions
in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
WARFARIN
Chemical formula: C19H16O4
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-2 H-1-benzopyran-2-one
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl) coumarin
CAS registry number: 81-81-2 (unstated stereochemistry)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state crystalline When used as a rodenticide it is formulated as
Colour colourless baits containing 250-1000 mg active ingredient/kg
Relative molecular mass 308.6
Melting point (°C) 159-161
Solubility in water insoluble
Solubility in
alcohols moderate
acetone readily
dioxane readily
HAZARD SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Easily absorbed Avoid exposure Obtain medical attention; antidote - vitamin K1
following ingestion and through
the skin; if absorbed, may cause
increased bleeding tendency
to massive haemorrhage
SKIN: Absorption may occur Wear gloves when handling Wash with soap and water; seek medical
concentrate material attention
EYES: Use a face shield when handling Flush eyes with water for at least 15 min
concentrate material
WARFARIN (cont'd)
HAZARD SYMPTOMS PREVENTION AND PROTECTION FIRST AID
INHALATION: Significant Avoid breathing aerosols or dust Move patient to fresh air; call emergency
hazard medical care
INGESTION: An unlikely Wash hands before eating, drinking,
occupational hazard or smoking
Accidental or intentional ingestion Keep out of reach of children and Transfer to hospital immediately; rinse out
may lead to poisoning in several under lock and key the mouth with water
hours
SPILLAGE STORAGE FIRE/EXPLOSION
Wear protective clothing during Store in sealed containers in a Burning in air may lead to the formation of
decontamination; dry spillage - collect dry, ventilated and locked toxic gases and vapours; use alcohol foam,
by suction and dispose of as toxic storeroom, away from children and carbon dioxide, or dry chemicals; keep
waste; large spillage - dissolve in unauthorized persons, food, and containers cool by spraying with water
a flammable solvent for atomizing animal feed
in a combustion chamber; do not
contaminate surface-water drains
WASTE DISPOSAL NATIONAL INFORMATION
Proper incineration
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1 Previous Evaluations by International Bodies
Warfarin has been classified by WHO in Class Ib - Highly Hazardous,
based on an acute oral LD50 of 10 mg/kg for rats.
7.2 Exposure Limit Values
ACGIH TLV, TWA 0.1 mg/m3
STEL 0.3 mg/m3
OSHA PEL, TWA 0.1 mg/m3
7.3 Specific Restrictions
Warfarin has been approved for use as a rodenticide in many countries.
In some countries, specific uses are defined, as well as limitations
and precautions.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies warfarin in:
Hazard Class: 6.1 -- poisonous substance.
Packing Group: I -- very dangerous substance when active
ingredient = 100-60%.
II -- substance presenting medium danger when
active ingredient = 60-6%.
III -- substance presenting minor danger when
active ingredient 6-1.5% (solid) or 6-0.6%
(liquid).
The European Economic Community legislation requires labelling of
warfarin as a dangerous substance using the symbol T - toxic.
R 26/27/28 Very toxic by inhalation, in contact with skin, and if
swallowed.
7.5 Waste Disposal
In the USA, warfarin waste is identified as an "acute hazardous
waste".
BIBLIOGRAPHY
CEC (1987) Legislation in dangerous substances - Classification and
labelling in the European Communities - Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
Hayes WJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology,
Vol. 3, New York, Academic Press.
IPCS (1992) Poisons information monograph - Warfarin,
IPCS (1995) Environmental Health Criteria 175: Anticoagulant
rodenticides, Geneva, World Health Organization.
IPCS-CEC (1993) International Chemical Safety Card No. 0821, Warfarin.
IRPTC (1987) IRPTC legal file 1986, Geneva, International Register for
Potentially Toxic Chemicals, United Nations Environment Programme.
IPCS/INTOX Project, Geneva, World Health Organization (unpublished
document IPCS/INTOX/PIM.563).
United Nations (1989) Recommendations on the transport of dangerous
goods, Sixth edition, New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards, 3 vol., Washington DC, US Department of Health and Human
Services. US Department of Labour (Publication No. DHHS (NIOSH)
01-12).
WHO (1994) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1994-1995, Geneva, World Health
Organization (unpublished document WHO/PCS/94.2).
Worthing CR & Hance RJ, ed. (1991) The pesticide manual, 9th ed.,
Farnham, United Kingdom, The British Crop Protection Council.
Common synonyms: coumafène, warfarine, zoocoumarin
Common trade names: Athrombine-K; Brumolin; Compound 42;
Coumadin; Coumafen; Coumarin;
Coumefene; Dethmore; Dethnel; Eastern
States Duocide; Fasco Fascrat Powder;
Frass-Ratron; Kumader; Kumadu; Kypfarin;
Maag Rattentod Cum; Mar-Frin; Maveran;
Panwarfin; Prothromadin; Rat-a-way;
Rat-b-gon; Rat-Gard; Rat-Kill; Rat-Mix;
Rat-ola; Ratro; Rats-No-More; Rodafarin;
Temus W; Warf 42; Warf Compound 42;
Warf-12; Warfarat; Warfarin +;
Warficide; Zoocoumarin
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-
2 H-1-benzopyran-2-one (9CI) (8CI) (I)
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl)
coumarin
CAS registry number: 81-81-2 (unstated stereochemistry);
5543-58-8 (R)-(+) isomer
RTECS registry number: GN4550000
1.2 Physical and Chemical Properties
Warfarin forms colourless crystals. It is practically insoluble in
water, readily soluble in acetone and dioxane and moderately soluble
in alcohols. It is acidic. The sodium salts are soluble in water but
insoluble in organic solvents.
Further physical and chemical properties of warfarin are given in the
"Summary of Chemical Safety Information" (section 6).
1.3 Analytical Methods
The determination of warfarin is mainly based on high-performance
liquid chromatography with fluorescence detection. The detection limit
in animal tissues is 0.02 mg/kg. The other methods include UV
spectrometry and reversed-phase liquid chromatography, the latter with
a detection limit in blood serum of 20 µg/litre.
1.4 Production and Uses
The anticoagulant properties of warfarin were reported in 1944, and,
in 1948, it was proposed as a rodenticide. Warfarin is used in
agriculture and urban rodent control in the form of baits containing
0.025% active ingredient. The sodium salt is available at 0.5%
concentrate for use at a final concentration of 0.05% in liquid base.
Warfarin is also used as a drug for the treatment of thromboembolic
disease in humans.
2. SUMMARY AND EVALUATION
2.1 Identity, Physical and Chemical Properties, and Analytical
Methods
Warfarin is a colourless, crystalline compound with a melting point of
151-161°C. It is practically insoluble in water, readily soluble in
acetone and dioxane, and moderately soluble in alcohols. Analytical
methods for the determination of warfarin are mainly based on high
performance liquid chromatography.
2.2 Sources of Human and Environmental Exposure
Warfarin does not occur naturally. It is used as a rodenticide as well
as a drug, and acts by disrupting the normal blood clotting mechanisms
causing an increased tendency to bleed.
2.3 Environmental Transport, Distribution, and Transformation
Warfarin is unlikely to enter the atmosphere, because of its low
volatility. It is practically insoluble in water. The rate of
degradation is relatively slow.
2.4 Environmental Levels and Human Exposure
No information is available on concentrations in air, water, and soil.
Warfarin is not intended for direct application to growing crops or
for use as a food additive. The controlled medical use of warfarin
exposes more people to higher concentations over a longer period than
would be expected to occur as a result of accidental human exposure
due to its use as a rodenticide. Accidental overexposures to warfarin
as a rodenticide are most commonly reported in children and domestic
animals.
2.5 Kinetics and Metabolism in Laboratory Animals and Humans
Warfarin is easily absorbed through the gastrointestinal tract and
skin, and is bound to serum proteins. Warfarin is readily hydroxylated
in vitro and in vivo by rat liver microsomal enzymes to form 6-,
7-, and 8-hydroxywarfarin. It is eliminated through both the urine and
faeces.
When a single oral dose of 1.5 mg warfarin/kg was given to male and
female volunteers, maximum concentration in plasma was reached in
2-12 h. The half-life for disappearance from human plasma ranged from
15 to 58 h.
2.6 Effects on Laboratory Mammals and in vitro Test Systems
The acute oral toxicity of warfarin for rats is high. Reported LD50
values range from 11 to 323 mg/kg, females being more susceptible than
males. For classification purposes, the accepted WHO LD50 value is
10 mg/kg. Genetic resistance to warfarin among rodents and humans has
been reported. Signs of poisoning are those associated with an
increased tendency to bleed.
One study on rats showed some developmental effects. There are no data
on mutagenicity.
In feeding studies on rats, the only effect found was associated with
anticoagulant action.
2.7 Effects on Humans
Vitamin K functions as an essential element in the synthesis of
several blood coagulation factors. Warfarin inhibits this process and
consequently affects the blood coagulation mechanisms. Prolonged
inhibition of the vitamin K synthesis will lead to severe bleeding and
death, if not corrected.
Symptoms of warfarin poisoning begin a few days or weeks after
ingestion. They include nose bleeding, bleeding gums, pallor, and,
sometimes, haematomas around joints and on the buttocks, and blood in
the urine and faeces. Later, paralysis due to cerebral haemorrhage
and, finally, haemorrhagic shock and death may occur.
Poisoning incidents have been reported. Outbreaks of poisoning have
been observed in relation to warfarin-contaminated meal and also in
infants after dermal application of warfarin-contaminated talc. A case
of poisoning from prolonged skin contact during the preparation of
warfarin baits has also been reported.
Developmental effects known as "warfarin embryopathy" or "fetal
warfarin syndrome" were reported when warfarin was administered as a
therapeutic agent during pregnancy. No cases of embryopathy following
the use of warfarin as a rodenticide have been reported.
2.8 Effects on other Organisms in the Laboratory and Field
Some secondary toxicity laboratory studies on wildlife have shown that
captive predators could be intoxicated by sufficient no-choice feeding
of warfarin-poisoned rodents.
2.9 Evaluation of Human Health Risks and Effects on the Environment
2.9.1 Evaluation of human health risks
Warfarin is used as both a rodenticide and a drug. In agriculture and
urban rodent control, it is used as a tracking dust or bait containing
0.025-0.05% of active ingredient. Increased levels in air, water, and
food are unlikely. Occupational exposure may occur during manufacture,
formulation, and bait application, but data concerning the levels are
not available.
Warfarin is easily absorbed through the gastrointestinal tract and
skin, and readily hydroxylated by liver microsomal enzymes in the rat.
The half-life for disappearance of warfarin from human plasma has been
reported to be between 15 and 58 h.
Warfarin is highly toxic for mammalian species. Signs of poisoning in
all species, including humans, are associated with an increased
tendency to bleed.
Poisoning incidents after massive oral or dermal exposure have been
reported. When used therapeutically, warfarin has been given to
patients for long periods without signs of toxicity, other than
bleeding. Warfarin is known as a human teratogen. Developmental
effects have been observed following the administration of warfarin as
a therapeutic agent during pregnancy. No cases of embryopathy
following its use as a rodenticide have been reported.
The specific and potent antidote in case of poisoning is vitamin K1.
2.9.2 Evaluation of effects on the environment
Warfarin is applied to discrete sites as low-concentration baits. It
is practically insoluble in water and, when it is used as a
rodenticide, it is unlikely to be a source of environmental
contamination.
Non-target organisms are potentially at risk in two ways: from direct
consumption of baits (primary hazard) and through eating poisoned
rodents (secondary hazard). The main reason for the poisoning of
domestic animals is direct consumption of baits.
Some secondary toxicity laboratory studies have shown that predators
can be intoxicated by eating warfarin-poisoned rodents. No cases of
secondary poisoning of predators in the field following the use of
warfarin as a rodenticide have been found.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
Exposure of the general population to warfarin as a rodenticide
through air, drinking-water, or food is unlikely and does not
constitute a significant health hazard. Poisoning incidents may occur
in cases of massive intentional or unintentional ingestion, massive
dermal exposure, and during manufacture and formulation. Warfarin is
teratogenic in humans at therapeutic doses. The use of warfarin as a
rodenticide is not expected to be a significant source of air, water,
soil, or food contamination. Direct or secondary poisoning of birds,
domestic and farm animals, and wildlife may occur.
3.2 Recommendations for the Protection of Human Health and the
Environment
Potentially exposed workers should receive appropriate biomonitoring
and health evaluation.
Exposure of pregnant women to concentrate material must be kept to a
minimum. Uneaten baits as well as killed rodents should be burned or
buried to reduce the risk of primary or secondary poisonings in
non-target organisms.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
Warfarin is highly toxic for mammalian species, including humans. The
probable lethal oral dose in humans is believed to be between 50 and
500 mg/kg. A cumulative total dose of about 1000 mg of warfarin
consumed in 13 days has been reported to be fatal for an adult man,
equivalent to about 1.1 mg/kg per day.
The main features of warfarin poisoning in less severe cases are
excessive bruising, nose and gum bleeding, and blood in the urine and
faeces. Bleeding from several organs within the body, leading to shock
and possibly death, occurs in the more severe cases. The onset of the
signs of poisoning may not be evident until a few days after exposure.
When handling technical material or powder concentrates, full air-fed
protection and an impervious suit suitable for wash-down are
necessary. Operations with liquid concentrates require the use of PVC
or nitrile-rubber gloves, armlets, and an apron, with a face shield
and rubber boots.
All persons who are bleeding, or suspected to be heavily exposed, must
receive medical attention.
4.1.1 Advice to physicians
If poisoning is recent (within a few hours), gastric lavage is
recommended. Administration of active charcoal in repeated doses is
also recommended.
Take a venous blood sample for measurement of the haemoglobin level,
prothrombin time, blood grouping, and cross-matching.
In less severe cases of poisoning give vitamin K1 (phytomenadione)
(5-10 mg), by slow intravenous infusion, 3 times daily on the first
day. After initial intravenous administration, oral treatment can be
continued until normalization of prothrombin tests.
In more severe cases, transfusion of matched whole blood or fresh,
frozen plasma should also be given and repeated daily until
prothrombin tests have returned to normal. Administration of factor
concentrate may be considered to prevent a volume overload.
Prothrombin tests should be made at least twice daily until a return
to normal is clearly established.
Keep the patient in hospital until the prothrombin time has remained
normal for three days.
4.1.2 Health surveillance advice
Workers handling concentrates should undergo periodic determinations
of blood prothrombin time. Persons with a history of blood disorders
with bleeding tendencies would be expected to be at increased risk in
case of exposure.
4.2 Explosion and Fire Hazards
Flash point and flammable limits have not been found. Contact with
strong oxidizers may cause fires and explosions. Recommended
extinguishers are foam, carbon dioxide, or dry chemicals.
High temperature decomposition or burning may lead to the formation of
toxic gases and vapours. Wear full protective clothing and
self-contained breathing apparatus in fire-fighting.
Run-off water from the fire should be prevented from entering
surface-water drains or water courses.
4.3 Storage
Technical material and formulations should be stored in sealed
containers in locked, well-ventilated, dry areas away from frost,
direct sunlight, and sources of heat and ignition. Keep products out
of reach of children, unauthorized personnel, and domestic animals. Do
not store near food or animal feed.
4.4 Transport
Comply with any local regulations regarding the movement of hazardous
goods. Before despatch ensure that the containers are sound and that
labels are securely fixed and undamaged.
4.5 Spillage
During decontamination, the operator must wear protective clothing,
PVC or nitrile gloves, a face shield, and rubber boots.
Small spills should be collected and disposed of as toxic waste
according to local legislation.
Large quantities may be destroyed by dissolving in a flammable solvent
(e.g., alcohol) and atomizing in a combustion chamber.
Contaminated areas should be washed down with cold water containing
surfactant or detergent; the washings must be prevented from entering
surface water and drains.
4.6 Disposal
No specific data are available.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Warfarin is practically insoluble in water. It is unlikely to be a
source of environmental contamination.
Do not place baits where domestic or farm animals and birds can reach
them. Burn or bury any uneaten bait. Do not dump it in water. Look for
dead rats and mice and burn or bury them.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, warfarin. It should be displayed at, or
near, entrances to areas where there is potential exposure to
warfarin, and on processing equipment and containers. The summary
should be translated into the appropriate language(s). All persons
potentially exposed to the chemical should also have the instructions
in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
WARFARIN
Chemical formula: C19H16O4
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-2 H-1-benzopyran-2-one
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl) coumarin
CAS registry number: 81-81-2 (unstated stereochemistry)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state crystalline When used as a rodenticide it is formulated as
Colour colourless baits containing 250-1000 mg active ingredient/kg
Relative molecular mass 308.6
Melting point (°C) 159-161
Solubility in water insoluble
Solubility in
alcohols moderate
acetone readily
dioxane readily
HAZARD SYMPTOMS PREVENTION AND PROTECTION FIRST AID
GENERAL: Easily absorbed Avoid exposure Obtain medical attention; antidote - vitamin K1
following ingestion and through
the skin; if absorbed, may cause
increased bleeding tendency
to massive haemorrhage
SKIN: Absorption may occur Wear gloves when handling Wash with soap and water; seek medical
concentrate material attention
EYES: Use a face shield when handling Flush eyes with water for at least 15 min
concentrate material
WARFARIN (cont'd)
HAZARD SYMPTOMS PREVENTION AND PROTECTION FIRST AID
INHALATION: Significant Avoid breathing aerosols or dust Move patient to fresh air; call emergency
hazard medical care
INGESTION: An unlikely Wash hands before eating, drinking,
occupational hazard or smoking
Accidental or intentional ingestion Keep out of reach of children and Transfer to hospital immediately; rinse out
may lead to poisoning in several under lock and key the mouth with water
hours
SPILLAGE STORAGE FIRE/EXPLOSION
Wear protective clothing during Store in sealed containers in a Burning in air may lead to the formation of
decontamination; dry spillage - collect dry, ventilated and locked toxic gases and vapours; use alcohol foam,
by suction and dispose of as toxic storeroom, away from children and carbon dioxide, or dry chemicals; keep
waste; large spillage - dissolve in unauthorized persons, food, and containers cool by spraying with water
a flammable solvent for atomizing animal feed
in a combustion chamber; do not
contaminate surface-water drains
WASTE DISPOSAL NATIONAL INFORMATION
Proper incineration
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1 Previous Evaluations by International Bodies
Warfarin has been classified by WHO in Class Ib - Highly Hazardous,
based on an acute oral LD50 of 10 mg/kg for rats.
7.2 Exposure Limit Values
ACGIH TLV, TWA 0.1 mg/m3
STEL 0.3 mg/m3
OSHA PEL, TWA 0.1 mg/m3
7.3 Specific Restrictions
Warfarin has been approved for use as a rodenticide in many countries.
In some countries, specific uses are defined, as well as limitations
and precautions.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies warfarin in:
Hazard Class: 6.1 -- poisonous substance.
Packing Group: I -- very dangerous substance when active
ingredient = 100-60%.
II -- substance presenting medium danger when
active ingredient = 60-6%.
III -- substance presenting minor danger when
active ingredient 6-1.5% (solid) or 6-0.6%
(liquid).
The European Economic Community legislation requires labelling of
warfarin as a dangerous substance using the symbol T - toxic.
R 26/27/28 Very toxic by inhalation, in contact with skin, and if
swallowed.
7.5 Waste Disposal
In the USA, warfarin waste is identified as an "acute hazardous
waste".
BIBLIOGRAPHY
CEC (1987) Legislation in dangerous substances - Classification and
labelling in the European Communities - Vol. 1 & 2. Commission of the
European Communities, London, Graham & Trotman, Ltd.
Hayes WJ Jr & Laws ER Jr (1991) Handbook of pesticide toxicology,
Vol. 3, New York, Academic Press.
IPCS (1992) Poisons information monograph - Warfarin,
IPCS (1995) Environmental Health Criteria 175: Anticoagulant
rodenticides, Geneva, World Health Organization.
IPCS-CEC (1993) International Chemical Safety Card No. 0821, Warfarin.
IRPTC (1987) IRPTC legal file 1986, Geneva, International Register for
Potentially Toxic Chemicals, United Nations Environment Programme.
IPCS/INTOX Project, Geneva, World Health Organization (unpublished
document IPCS/INTOX/PIM.563).
United Nations (1989) Recommendations on the transport of dangerous
goods, Sixth edition, New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards, 3 vol., Washington DC, US Department of Health and Human
Services. US Department of Labour (Publication No. DHHS (NIOSH)
01-12).
WHO (1994) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1994-1995, Geneva, World Health
Organization (unpublished document WHO/PCS/94.2).
Worthing CR & Hance RJ, ed. (1991) The pesticide manual, 9th ed.,
Farnham, United Kingdom, The British Crop Protection Council.
 Common synonyms: coumafène, warfarine, zoocoumarin
Common trade names: Athrombine-K; Brumolin; Compound 42;
Coumadin; Coumafen; Coumarin;
Coumefene; Dethmore; Dethnel; Eastern
States Duocide; Fasco Fascrat Powder;
Frass-Ratron; Kumader; Kumadu; Kypfarin;
Maag Rattentod Cum; Mar-Frin; Maveran;
Panwarfin; Prothromadin; Rat-a-way;
Rat-b-gon; Rat-Gard; Rat-Kill; Rat-Mix;
Rat-ola; Ratro; Rats-No-More; Rodafarin;
Temus W; Warf 42; Warf Compound 42;
Warf-12; Warfarat; Warfarin +;
Warficide; Zoocoumarin
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-
2 H-1-benzopyran-2-one (9CI) (8CI) (I)
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl)
coumarin
CAS registry number:
Common synonyms: coumafène, warfarine, zoocoumarin
Common trade names: Athrombine-K; Brumolin; Compound 42;
Coumadin; Coumafen; Coumarin;
Coumefene; Dethmore; Dethnel; Eastern
States Duocide; Fasco Fascrat Powder;
Frass-Ratron; Kumader; Kumadu; Kypfarin;
Maag Rattentod Cum; Mar-Frin; Maveran;
Panwarfin; Prothromadin; Rat-a-way;
Rat-b-gon; Rat-Gard; Rat-Kill; Rat-Mix;
Rat-ola; Ratro; Rats-No-More; Rodafarin;
Temus W; Warf 42; Warf Compound 42;
Warf-12; Warfarat; Warfarin +;
Warficide; Zoocoumarin
CAS chemical name: 4-hydroxy-3-(3-oxo-1-phenylbutyl)-
2 H-1-benzopyran-2-one (9CI) (8CI) (I)
IUPAC chemical name: (RS) 4-hydroxy-3-(3-oxo-1-phenylbutyl)
coumarin
CAS registry number: 