
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 32
d-PHENOTHRIN
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1989
This is a companion volume to Environmental Health Criteria 96:
d-Phenothrin
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
ISBN 92 4 154353 1
ISSN 0259
World Health Organization 1989
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city, or area
or of its authorities, or concerning the delimitation of its frontiers
or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Human exposure
2.2. Environmental fate
2.3. Kinetics and metabolism
2.4. Effects on organisms in the environment
2.5. Effects on experimental animals and in vitro test
systems
2.6. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.2. Recommendations
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1. Human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. INTERNATIONAL CHEMICAL SAFETY CARD
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging and transport
7.5. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and human health from exposure to a
chemical or combinations of chemicals, or to physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is an International Chemical Safety Card
which should be readily available, and should be clearly explained, to
all who could come into contact with the chemical. The section on
regulatory information has been extracted from the legal file of the
International Register of Potentially Toxic Chemicals (IRPTC) and from
other United Nations sources.
The target readership includes people in the occupational health
services, ministries, governmental agencies, industry, and trade
unions, who are involved in the safe use of chemicals and the
prevention of environmental health hazards, and also workers who would
like more information on this topic. An attempt has been made to use
only terms that are familiar to the user. However, sections 1 and 2
inevitably contain some technical terms. A bibliography has been
included for readers who would like to have further background
information.
Revision of the information in this Guide will take place in due
course. The eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: d-Phenothrin
Chemical structure:
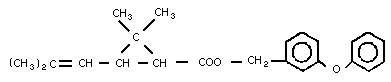 Molecular formula: C23H26O3
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-
dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)
methyl ester
CAS registry number: 26002-80-2 (racemic)
RTECS registry number: GZ 1975000 (racemic)
GZ 2002000 (d-phenothrin)
Common synonyms and trade names: Sumithrin, S-2539 Forte
Relative molecular mass: 350.49
d-Phenothrin is a synthetic pyrethroid insecticide. Racemic
phenothrin was invented in 1969. It is a mixture of four
stereoisomers. d-Phenothrin is a mixture of the two isomers that have
the highest activity and is the only technical product commercially
available. The technical grade is 92.5-94.5% pure.
1.2 Physical and Chemical Properties
Some physical and chemical properties of d-phenothrin are given in the
International Chemical Safety Card (section 6). Technical grade
d-phenothrin is a colourless or pale yellow liquid. It has a low
solubility in water, but is soluble in organic solvents such as
acetone, xylene, and hexane. It is fairly stable in air, but unstable
to light, although it is not photodegraded as rapidly as natural
pyrethrins. It is also unstable under alkaline conditions.
1.3 Analytical Methods
Residue analysis is done with high-performance liquid chromatography
with a UV detector, the minimum detectable concentration being
0.05 mg/kg. Gas chromatography with a flame ionization detector is
used to analyse technical products.
1.4 Production and Uses
d-Phenothrin has been used since 1977. It is estimated that 70 to
80 tonnes of d-phenothrin are used annually worldwide. d-Phenothrin
is used primarily to control household insects and insects endangering
public health, to protect stored grains, alone or in combination with
other insecticides and/or synergists, and to control lice in human
beings. It is formulated as an aerosol, in oil and dust formulations,
or as an emulsifiable concentrate. When used to control lice, it is
formulated as powders, shampoos, and lotions.
2. SUMMARY AND EVALUATION
2.1 Human Exposure
Air levels from conventional household aerosol spraying with
d-phenothrin are not expected to exceed 0.5 mg/m3. Residues of up
to 4 mg/kg might be present in stored wheat. In flour, these
residues decrease to 0.8 mg/kg after milling and to 0.6 mg/kg after
baking.
To control lice, d-phenothrin (32 mg/person) is usually applied to the
hair once a day every 3 days for a maximum of 9 days. No data are
available on occupational exposure to d-phenothrin.
Exposure of the general population to d-phenothrin is expected to be
very low, but precise data are not available.
2.2 Environmental Fate
Phenothrin degrades readily on plants and other surfaces and has a
half-life of <1 day. There is little translocation of d-phenothrin
and its degradation products to the untreated parts of the plants.
Limited uptake of radiolabelled products into bean plants took place
from soils treated with 14C-phenothrin. When soils are treated with
[1R, trans]-or [1R, cis]-phenothrin (1 mg/kg), both isomers
decompose rapidly having initial half-lives of 1 to 2 days. Under
flooded conditions, the degradation is much slower, with initial
half-lives of 2 to 4 weeks for [1R, trans]- phenothrin and 1 to 2
months for [1R, cis]-phenothrin. Very little movement (approximately
2%) of trans- and cis-phenothrin is observed through soil columns
when leaching is begun immediately or 14 days after treatment with the
insecticide.
In general, the degradative processes that occur in the environment
lead to less toxic products.
2.3 Kinetics and Metabolism
After rats were given single or repeated oral exposure or dermal
treatment with radiolabelled phenothrin, radiolabels were rapidly and
almost completely excreted in the urine and faeces (3-7 days). The
major metabolic pathways of both trans- and cis-phenothrin in rats
were ester cleavage and oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety. Ester-cleaved
metabolites (excreted mainly in the urine) were the principal products
of the trans isomer, whereas ester-form metabolites (excreted mainly
in the faeces) were mostly formed from the cis isomer.
2.4 Effects on Organisms in the Environment
Phenothrin has been tested only on a few groups of non-target
organisms and only on a few species within each group. The range of
96-h LC50 values for racemic phenothrin and (1R)-stereoisomers in
fish ranged from 17 to 200 µg/litre. In a single study on aquatic
invertebrates, 3-h LC50 values for Daphnia pulex were 25 000
to 50 000 µg/litre for all isomers and racemic phenothrin.
A single field study in which phenothrin was applied to ponds showed
no effect on aquatic arthropods.
Toxicity to birds is low with an acute oral LD50 for bobwhite quail
of >2500 mg/kg body weight, and a dietary LC50 for mallard duck and
bobwhite quail of >5000 mg/kg diet.
Since phenothrin breaks down rapidly in sunlight and is used primarily
on stored grain, environmental exposure is expected to be very low.
Therefore, adverse effects on the environment are extremely
improbable.
2.5 Effects on Experimental Animals and In Vitro Test Systems
The acute toxicity of d-phenothrin is extremely low with LD50 values
of >5000 mg/kg body weight in rats and mice (through the oral,
subcutaneous, dermal, and intraperitoneal routes), and LC50 values
of >3760 mg/m3 in rats (through the inhalation route).
d-Phenothrin causes a poisoning syndrome which may include tremor,
ataxia, hyperexcitability, prostration, or paralysis. These symptoms,
and the results of electrophysiological studies of cockroach cercal
sensory nerves, classify d-phenothrin as a Type I pyrethroid.
When rats were exposed to d-phenothrin by inhalation at concentrations
of up to 210 mg/m3 for 4 h/day for 4 weeks, or orally for 5
consecutive days at a dose of 5000 mg/kg body weight, no adverse
toxicological effects were observed.
Several feeding studies of phenothrin (racemic or d-phenothrin, from
200 to 10 000 mg/kg diet) in rats and mice, with exposure periods from
6 months to 2 years, were performed. No-observed-effect levels (NOELs)
in these studies ranged from 300 to 1000 mg/kg diet which correspond
to approximately 40 to 160 mg/kg body weight per day. In two studies
studies in dogs in which d-phenothrin was given in doses from 100 to
3000 mg/kg diet, with exposure periods of 26 to 52 weeks, NOELs of
300 mg/kg diet, corresponding to 7-8 mg/kg body weight per day, were
obtained.
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
In 2-year studies, d-phenothrin was not oncogenic in rats and mice at
dietary levels of up to 3000 mg/kg diet.
Neither teratogenicity nor embryotoxicity was observed in fetuses of
rabbits and mice given oral d-phenothrin at up to 1000 and 3000 mg/kg
body weight, respectively. In a two-generation reproduction study in
rats, the NOEL was 1000 mg/kg diet.
Rats exposed to very high doses of d-phenothrin at a concentration of
up to 3760 mg/m3 for 4 h by inhalation, or to a dose of 5000 mg/kg
body weight per day for 5 days orally, showed no myelin degeneration
or axon disruption in the sciatic nerve.
2.6 Effects on Human Beings
Although d-phenothrin has been used for more than 10 years, there have
been no reports of poisoning in human beings.
There are no indications that d-phenothrin, when used as recommended,
will have an adverse effect on human beings.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
(a) General population: The exposure of the general population to
d-phenothrin is expected to be very low and is not likely to be a
hazard when used as recommended.
(b) Occupational exposure: With reasonable work practices, hygiene
measures, and safety precautions, d-phenothrin is unlikely to be an
occupational hazard.
(c) Environment: Since d-phenothrin breaks down rapidly in sunlight
and is used principally on stored grain, environmental exposure is
expected to be very low. Therefore, environmental effects of the
compound are extremely improbable.
3.2 Recommendations
When d-phenothrin is used as recommended, exposure levels are expected
to be very low. However, monitoring studies should be continued.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
d-Phenothrin is a synthetic pyrethroid insecticide. There have been no
cases of poisoning reported in the general population or from
occupational exposure. Experimental studies in animals suggest that
neurological signs and symptoms, tremors, and ataxia could occur after
massive over-exposure or accidental ingestion.
The human health hazards associated with certain types of exposure to
d-phenothrin, together with preventive and protective measures and
first-aid recommendations, are given in the International Chemical
Safety Card (section 6).
4.1.1 Advice to physicians
There is no specific antidote for d-phenothrin poisoning. Treat
symptomatically. Chemical pneumonitis resulting from aspiration of
the solvent into the lungs occurs when liquid formulations are
ingested.
4.1.2 Health surveillance advice
Depending on the degree of exposure, general medical examinations
should be done annually.
4.2 Explosion and Fire Hazards
Some solvents in pyrethroid formulations are highly flammable. DO NOT
USE WATER to extinguish fires. Use dry powder, carbon dioxide, or
alcohol-resistant foam, sand, or earth. Cool nearby drums with water
spray.
If pyrethroid products are involved in a major fire, advise the fire
service to wear protective clothing and breathing apparatus. Inform
the fire service and other relevant authorities that pyrethroids are
toxic for fish and that water should be used only to cool unaffected
stock. In this way, the accumulation of polluted run-off from the site
is prevented.
4.3 Storage
Store technical material and formulations away from heat, in a locked,
well-ventilated area, preferably without drains, designated for
insecticide storage only. Keep out of reach of children, unauthorized
personnel, and away from animals.
Store away from food and animal feed.
4.4 Transport
For transport purposes, pyrethroids are classified as "harmful" or as
"low hazard". Formulations made with flammable solvents may be
subject to local transport controls. Before transport, ensure that
containers are sound and that labels are securely fixed and not
damaged. Comply with local transport regulations.
Do not transport in compartments that contain food and animal feed.
4.5 Spillage and Disposal
4.5.1 Spillage
Keep spectators away from leaking or spilled product. Prohibit
smoking and the use of naked flames in the immediate vicinity.
Avoid exposure by wearing appropriate protective clothing and masks.
Transfer any product remaining in damaged or leaking containers into a
clean, empty drum, and label the drum.
Absorb spillage and cover contaminated area with lime, damp sawdust,
sand, or earth, and place in a secure container for safe disposal (see
section 4.5.2). Contain a large spillage by building a barrier of
earth or sandbags.
Prevent liquid from spreading to other cargo, vegetation, or waterways
by containing it with the most readily available barrier material,
e.g., earth or sand.
Decontaminate empty, damaged, or leaking containers with a 10% sodium
carbonate solution added at the rate of at least 1 litre per 20-litre
drum. Puncture containers to prevent reuse.
4.5.2 Disposal
Waste that contains d-phenothrin should be burnt in an appropriate
high-temperature incinerator with effluent scrubbing. If no
incinerator is available, contaminated absorbents or surplus products
should be decomposed by hydrolysis at pH 12 or above. Contact with a
suitable hydrolysing agent is required to ensure degradation of the
active ingredient to a safe level.
For emulsifiable material, use 5% sodium hydroxide (caustic soda)
solution or saturated (7-10%) sodium carbonate (washing soda)
solution.
For non-emulsifiable material, use a 1:1 mixture (by volume) of
caustic soda or washing soda (as above) and a water/oil soluble
solvent such as denatured alcohol, monoethylene glycol, hexylene
glycol, or isopropylalcohol.
Cover the material with a hydrolysing agent and let it stand for 7
days. Before disposal, the waste must be analysed to ensure that the
active ingredient has been degraded to a safe level.
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams,
water-courses, open waterways, ditches, fields with drainage systems,
or to the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
When used as recommended, it is very unlikely that either
d-phenothrin or its degradation products will reach levels of
environmental significance. d-Phenothrin is toxic for fish, but due
to the very low exposure levels that could occur, it will only cause a
problem if spilled.
Do not dispose of d-phenothrin or its containers in ponds, waterways,
or ditches.
6. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers concerned
with, and users of, d-phenothrin. It should be displayed at, or near,
entrances to areas where there is potential exposure to d-phenothrin,
and on processing equipment and containers. The card should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions on the
chemical safety card clearly explained.
Space is available on the card for insertion of the National
Occupational Exposure Limit, the address and telephone number of the
National Poison Control Centre, and for local trade names.
d-PHENOTHRIN
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)methyl ester
CAS registry no: 26002-80-2
Molecular formula: C23H26O3
RTECS registry no: GZ2002000
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state liquid Phenothrin is a mixture of four stereo-isomers;
Colour pale yellow to yellow brown d-Phenothrin is a mixture of the
Relative molecular mass 350.49 two most active isomers. The technical
Water solubility (25 °C) 2 mg/litre grade is 92.5-94.5% pure. d-Phenothrin is
Solubility in organic solvents solublea fairly stable in air but unstable to light
25
Density d25 1.058-1.061 and under alkaline conditions. It is a synthetic
pyrethroid, mainly used alone or in
Vapour pressure (20 °C) 0.16 mPa combination as a household insecticide and
for public health. It is used mostly in
aerosol form.
a Hexane (>1 mg/kg), acetone, methanol, xylene.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Some formulations may Use proper application techniques Remove contaminated clothing; wash
cause skin irritation and proper skin protection skin with soap and water
EYES: Splashes may cause Wear face shield or goggles Flush immediately with clean water
irritation for at least 15 min
INHALATION: Dust or droplets Do not inhale fine dust and mist Fresh air
may cause irritation
INGESTION: Unlikely Do not eat, drink, or smoke during
occupational hazard work; wash hands before eating,
drinking, or smoking
Accidental or deliberate Obtain medical attention; if breathing
ingestion could lead to neurological has stopped, apply artificial
signs and symptoms such as tremor respiration
and ataxia
A hazard of ingested liquid Do not induce vomiting
formulations is aspiration
into lungs
ENVIRONMENT: Toxic for Do not contaminate ponds, waterways,
fish or ditches with product or used
containers
SPILLAGE STORAGE FIRE AND EXPLOSION
Absorb spillage with lime, damp Store in locked, well-ventilated DO NOT USE WATER; some liquid
sawdust, sand, or earth; sweep storeroom, out of reach of children formulations may be highly flammable;
up, place in closed container, and unauthorized personnel, and use dry powder, carbon dioxide, or
and dispose of safely; do not away from food and animal feed. alcohol-resistant foam; cool nearby
contaminate personnel, ponds, drums with water spray
or waterways
WASTE DISPOSAL NATIONAL INFORMATION
Burn in high-temperature National Occupational Exposure Limit:
incinerator with effluent scrubbing
Or, treat with 5% caustic soda National Poison Control Centre:
as a hydrolysing agent; comply
with local regulation Local trade names:
Molecular formula: C23H26O3
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-
dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)
methyl ester
CAS registry number: 26002-80-2 (racemic)
RTECS registry number: GZ 1975000 (racemic)
GZ 2002000 (d-phenothrin)
Common synonyms and trade names: Sumithrin, S-2539 Forte
Relative molecular mass: 350.49
d-Phenothrin is a synthetic pyrethroid insecticide. Racemic
phenothrin was invented in 1969. It is a mixture of four
stereoisomers. d-Phenothrin is a mixture of the two isomers that have
the highest activity and is the only technical product commercially
available. The technical grade is 92.5-94.5% pure.
1.2 Physical and Chemical Properties
Some physical and chemical properties of d-phenothrin are given in the
International Chemical Safety Card (section 6). Technical grade
d-phenothrin is a colourless or pale yellow liquid. It has a low
solubility in water, but is soluble in organic solvents such as
acetone, xylene, and hexane. It is fairly stable in air, but unstable
to light, although it is not photodegraded as rapidly as natural
pyrethrins. It is also unstable under alkaline conditions.
1.3 Analytical Methods
Residue analysis is done with high-performance liquid chromatography
with a UV detector, the minimum detectable concentration being
0.05 mg/kg. Gas chromatography with a flame ionization detector is
used to analyse technical products.
1.4 Production and Uses
d-Phenothrin has been used since 1977. It is estimated that 70 to
80 tonnes of d-phenothrin are used annually worldwide. d-Phenothrin
is used primarily to control household insects and insects endangering
public health, to protect stored grains, alone or in combination with
other insecticides and/or synergists, and to control lice in human
beings. It is formulated as an aerosol, in oil and dust formulations,
or as an emulsifiable concentrate. When used to control lice, it is
formulated as powders, shampoos, and lotions.
2. SUMMARY AND EVALUATION
2.1 Human Exposure
Air levels from conventional household aerosol spraying with
d-phenothrin are not expected to exceed 0.5 mg/m3. Residues of up
to 4 mg/kg might be present in stored wheat. In flour, these
residues decrease to 0.8 mg/kg after milling and to 0.6 mg/kg after
baking.
To control lice, d-phenothrin (32 mg/person) is usually applied to the
hair once a day every 3 days for a maximum of 9 days. No data are
available on occupational exposure to d-phenothrin.
Exposure of the general population to d-phenothrin is expected to be
very low, but precise data are not available.
2.2 Environmental Fate
Phenothrin degrades readily on plants and other surfaces and has a
half-life of <1 day. There is little translocation of d-phenothrin
and its degradation products to the untreated parts of the plants.
Limited uptake of radiolabelled products into bean plants took place
from soils treated with 14C-phenothrin. When soils are treated with
[1R, trans]-or [1R, cis]-phenothrin (1 mg/kg), both isomers
decompose rapidly having initial half-lives of 1 to 2 days. Under
flooded conditions, the degradation is much slower, with initial
half-lives of 2 to 4 weeks for [1R, trans]- phenothrin and 1 to 2
months for [1R, cis]-phenothrin. Very little movement (approximately
2%) of trans- and cis-phenothrin is observed through soil columns
when leaching is begun immediately or 14 days after treatment with the
insecticide.
In general, the degradative processes that occur in the environment
lead to less toxic products.
2.3 Kinetics and Metabolism
After rats were given single or repeated oral exposure or dermal
treatment with radiolabelled phenothrin, radiolabels were rapidly and
almost completely excreted in the urine and faeces (3-7 days). The
major metabolic pathways of both trans- and cis-phenothrin in rats
were ester cleavage and oxidation at the 4'-position of the alcohol
moiety or the isobutenyl group of the acid moiety. Ester-cleaved
metabolites (excreted mainly in the urine) were the principal products
of the trans isomer, whereas ester-form metabolites (excreted mainly
in the faeces) were mostly formed from the cis isomer.
2.4 Effects on Organisms in the Environment
Phenothrin has been tested only on a few groups of non-target
organisms and only on a few species within each group. The range of
96-h LC50 values for racemic phenothrin and (1R)-stereoisomers in
fish ranged from 17 to 200 µg/litre. In a single study on aquatic
invertebrates, 3-h LC50 values for Daphnia pulex were 25 000
to 50 000 µg/litre for all isomers and racemic phenothrin.
A single field study in which phenothrin was applied to ponds showed
no effect on aquatic arthropods.
Toxicity to birds is low with an acute oral LD50 for bobwhite quail
of >2500 mg/kg body weight, and a dietary LC50 for mallard duck and
bobwhite quail of >5000 mg/kg diet.
Since phenothrin breaks down rapidly in sunlight and is used primarily
on stored grain, environmental exposure is expected to be very low.
Therefore, adverse effects on the environment are extremely
improbable.
2.5 Effects on Experimental Animals and In Vitro Test Systems
The acute toxicity of d-phenothrin is extremely low with LD50 values
of >5000 mg/kg body weight in rats and mice (through the oral,
subcutaneous, dermal, and intraperitoneal routes), and LC50 values
of >3760 mg/m3 in rats (through the inhalation route).
d-Phenothrin causes a poisoning syndrome which may include tremor,
ataxia, hyperexcitability, prostration, or paralysis. These symptoms,
and the results of electrophysiological studies of cockroach cercal
sensory nerves, classify d-phenothrin as a Type I pyrethroid.
When rats were exposed to d-phenothrin by inhalation at concentrations
of up to 210 mg/m3 for 4 h/day for 4 weeks, or orally for 5
consecutive days at a dose of 5000 mg/kg body weight, no adverse
toxicological effects were observed.
Several feeding studies of phenothrin (racemic or d-phenothrin, from
200 to 10 000 mg/kg diet) in rats and mice, with exposure periods from
6 months to 2 years, were performed. No-observed-effect levels (NOELs)
in these studies ranged from 300 to 1000 mg/kg diet which correspond
to approximately 40 to 160 mg/kg body weight per day. In two studies
studies in dogs in which d-phenothrin was given in doses from 100 to
3000 mg/kg diet, with exposure periods of 26 to 52 weeks, NOELs of
300 mg/kg diet, corresponding to 7-8 mg/kg body weight per day, were
obtained.
d-Phenothrin is not mutagenic in a variety of in vivo and in vitro
test systems that studied gene mutations, DNA damage, DNA repair, and
chromosomal effects.
In 2-year studies, d-phenothrin was not oncogenic in rats and mice at
dietary levels of up to 3000 mg/kg diet.
Neither teratogenicity nor embryotoxicity was observed in fetuses of
rabbits and mice given oral d-phenothrin at up to 1000 and 3000 mg/kg
body weight, respectively. In a two-generation reproduction study in
rats, the NOEL was 1000 mg/kg diet.
Rats exposed to very high doses of d-phenothrin at a concentration of
up to 3760 mg/m3 for 4 h by inhalation, or to a dose of 5000 mg/kg
body weight per day for 5 days orally, showed no myelin degeneration
or axon disruption in the sciatic nerve.
2.6 Effects on Human Beings
Although d-phenothrin has been used for more than 10 years, there have
been no reports of poisoning in human beings.
There are no indications that d-phenothrin, when used as recommended,
will have an adverse effect on human beings.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
(a) General population: The exposure of the general population to
d-phenothrin is expected to be very low and is not likely to be a
hazard when used as recommended.
(b) Occupational exposure: With reasonable work practices, hygiene
measures, and safety precautions, d-phenothrin is unlikely to be an
occupational hazard.
(c) Environment: Since d-phenothrin breaks down rapidly in sunlight
and is used principally on stored grain, environmental exposure is
expected to be very low. Therefore, environmental effects of the
compound are extremely improbable.
3.2 Recommendations
When d-phenothrin is used as recommended, exposure levels are expected
to be very low. However, monitoring studies should be continued.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY
ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
d-Phenothrin is a synthetic pyrethroid insecticide. There have been no
cases of poisoning reported in the general population or from
occupational exposure. Experimental studies in animals suggest that
neurological signs and symptoms, tremors, and ataxia could occur after
massive over-exposure or accidental ingestion.
The human health hazards associated with certain types of exposure to
d-phenothrin, together with preventive and protective measures and
first-aid recommendations, are given in the International Chemical
Safety Card (section 6).
4.1.1 Advice to physicians
There is no specific antidote for d-phenothrin poisoning. Treat
symptomatically. Chemical pneumonitis resulting from aspiration of
the solvent into the lungs occurs when liquid formulations are
ingested.
4.1.2 Health surveillance advice
Depending on the degree of exposure, general medical examinations
should be done annually.
4.2 Explosion and Fire Hazards
Some solvents in pyrethroid formulations are highly flammable. DO NOT
USE WATER to extinguish fires. Use dry powder, carbon dioxide, or
alcohol-resistant foam, sand, or earth. Cool nearby drums with water
spray.
If pyrethroid products are involved in a major fire, advise the fire
service to wear protective clothing and breathing apparatus. Inform
the fire service and other relevant authorities that pyrethroids are
toxic for fish and that water should be used only to cool unaffected
stock. In this way, the accumulation of polluted run-off from the site
is prevented.
4.3 Storage
Store technical material and formulations away from heat, in a locked,
well-ventilated area, preferably without drains, designated for
insecticide storage only. Keep out of reach of children, unauthorized
personnel, and away from animals.
Store away from food and animal feed.
4.4 Transport
For transport purposes, pyrethroids are classified as "harmful" or as
"low hazard". Formulations made with flammable solvents may be
subject to local transport controls. Before transport, ensure that
containers are sound and that labels are securely fixed and not
damaged. Comply with local transport regulations.
Do not transport in compartments that contain food and animal feed.
4.5 Spillage and Disposal
4.5.1 Spillage
Keep spectators away from leaking or spilled product. Prohibit
smoking and the use of naked flames in the immediate vicinity.
Avoid exposure by wearing appropriate protective clothing and masks.
Transfer any product remaining in damaged or leaking containers into a
clean, empty drum, and label the drum.
Absorb spillage and cover contaminated area with lime, damp sawdust,
sand, or earth, and place in a secure container for safe disposal (see
section 4.5.2). Contain a large spillage by building a barrier of
earth or sandbags.
Prevent liquid from spreading to other cargo, vegetation, or waterways
by containing it with the most readily available barrier material,
e.g., earth or sand.
Decontaminate empty, damaged, or leaking containers with a 10% sodium
carbonate solution added at the rate of at least 1 litre per 20-litre
drum. Puncture containers to prevent reuse.
4.5.2 Disposal
Waste that contains d-phenothrin should be burnt in an appropriate
high-temperature incinerator with effluent scrubbing. If no
incinerator is available, contaminated absorbents or surplus products
should be decomposed by hydrolysis at pH 12 or above. Contact with a
suitable hydrolysing agent is required to ensure degradation of the
active ingredient to a safe level.
For emulsifiable material, use 5% sodium hydroxide (caustic soda)
solution or saturated (7-10%) sodium carbonate (washing soda)
solution.
For non-emulsifiable material, use a 1:1 mixture (by volume) of
caustic soda or washing soda (as above) and a water/oil soluble
solvent such as denatured alcohol, monoethylene glycol, hexylene
glycol, or isopropylalcohol.
Cover the material with a hydrolysing agent and let it stand for 7
days. Before disposal, the waste must be analysed to ensure that the
active ingredient has been degraded to a safe level.
Never pour untreated waste or surplus products into public sewers or
where there is any danger of run-off or seepage to streams,
water-courses, open waterways, ditches, fields with drainage systems,
or to the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
When used as recommended, it is very unlikely that either
d-phenothrin or its degradation products will reach levels of
environmental significance. d-Phenothrin is toxic for fish, but due
to the very low exposure levels that could occur, it will only cause a
problem if spilled.
Do not dispose of d-phenothrin or its containers in ponds, waterways,
or ditches.
6. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers concerned
with, and users of, d-phenothrin. It should be displayed at, or near,
entrances to areas where there is potential exposure to d-phenothrin,
and on processing equipment and containers. The card should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions on the
chemical safety card clearly explained.
Space is available on the card for insertion of the National
Occupational Exposure Limit, the address and telephone number of the
National Poison Control Centre, and for local trade names.
d-PHENOTHRIN
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)methyl ester
CAS registry no: 26002-80-2
Molecular formula: C23H26O3
RTECS registry no: GZ2002000
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state liquid Phenothrin is a mixture of four stereo-isomers;
Colour pale yellow to yellow brown d-Phenothrin is a mixture of the
Relative molecular mass 350.49 two most active isomers. The technical
Water solubility (25 °C) 2 mg/litre grade is 92.5-94.5% pure. d-Phenothrin is
Solubility in organic solvents solublea fairly stable in air but unstable to light
25
Density d25 1.058-1.061 and under alkaline conditions. It is a synthetic
pyrethroid, mainly used alone or in
Vapour pressure (20 °C) 0.16 mPa combination as a household insecticide and
for public health. It is used mostly in
aerosol form.
a Hexane (>1 mg/kg), acetone, methanol, xylene.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Some formulations may Use proper application techniques Remove contaminated clothing; wash
cause skin irritation and proper skin protection skin with soap and water
EYES: Splashes may cause Wear face shield or goggles Flush immediately with clean water
irritation for at least 15 min
INHALATION: Dust or droplets Do not inhale fine dust and mist Fresh air
may cause irritation
INGESTION: Unlikely Do not eat, drink, or smoke during
occupational hazard work; wash hands before eating,
drinking, or smoking
Accidental or deliberate Obtain medical attention; if breathing
ingestion could lead to neurological has stopped, apply artificial
signs and symptoms such as tremor respiration
and ataxia
A hazard of ingested liquid Do not induce vomiting
formulations is aspiration
into lungs
ENVIRONMENT: Toxic for Do not contaminate ponds, waterways,
fish or ditches with product or used
containers
SPILLAGE STORAGE FIRE AND EXPLOSION
Absorb spillage with lime, damp Store in locked, well-ventilated DO NOT USE WATER; some liquid
sawdust, sand, or earth; sweep storeroom, out of reach of children formulations may be highly flammable;
up, place in closed container, and unauthorized personnel, and use dry powder, carbon dioxide, or
and dispose of safely; do not away from food and animal feed. alcohol-resistant foam; cool nearby
contaminate personnel, ponds, drums with water spray
or waterways
WASTE DISPOSAL NATIONAL INFORMATION
Burn in high-temperature National Occupational Exposure Limit:
incinerator with effluent scrubbing
Or, treat with 5% caustic soda National Poison Control Centre:
as a hydrolysing agent; comply
with local regulation Local trade names:
 7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. It is a representative but
non-exhaustive overview of current regulations, guidelines, and
standards.
Regulations and guidelines about chemicals can be fully understood
only within the framework of a country's legislation, and are always
subject to change. Therefore, they should always be verified with the
appropriate authorities.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Expert Committee on Pesticide Residues (JMPR)
evaluated d-phenothrin at its meetings in 1979, 1980, 1984, and 1988.
In 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg body weight
was established.
The Division of Vector Biology and Control, World Health Organization,
classified phenothrin as a technical product that is not likely to
present an acute hazard when used as recommended (WHO, 1988).
7.2 Exposure Limit Values
Some exposure limit values are given in the table that follows.
7.3 Specific Restrictions
No information is available.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies pyrethroids in:
- hazard class 6.1: poisonous substance
- packing group III: a substance with a relatively low risk of
poisoning during transport.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD Intake from FAO/WHO Temporary acceptable daily intake 0.04 mg/kg 1985
(TADI) (mg/kg body weight for
d-phenothrin)
FOOD Plant residues FAO/WHO Temporary maximum residue limit
(TMRL)
- Wheat bran (unprocessed) 15 mg/kg 1980
FAO/WHO Maximum residue limit (MRL) 5 mg/kg 1980
(cereal grains)
The label should be as follows:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. It is a representative but
non-exhaustive overview of current regulations, guidelines, and
standards.
Regulations and guidelines about chemicals can be fully understood
only within the framework of a country's legislation, and are always
subject to change. Therefore, they should always be verified with the
appropriate authorities.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Expert Committee on Pesticide Residues (JMPR)
evaluated d-phenothrin at its meetings in 1979, 1980, 1984, and 1988.
In 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg body weight
was established.
The Division of Vector Biology and Control, World Health Organization,
classified phenothrin as a technical product that is not likely to
present an acute hazard when used as recommended (WHO, 1988).
7.2 Exposure Limit Values
Some exposure limit values are given in the table that follows.
7.3 Specific Restrictions
No information is available.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies pyrethroids in:
- hazard class 6.1: poisonous substance
- packing group III: a substance with a relatively low risk of
poisoning during transport.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD Intake from FAO/WHO Temporary acceptable daily intake 0.04 mg/kg 1985
(TADI) (mg/kg body weight for
d-phenothrin)
FOOD Plant residues FAO/WHO Temporary maximum residue limit
(TMRL)
- Wheat bran (unprocessed) 15 mg/kg 1980
FAO/WHO Maximum residue limit (MRL) 5 mg/kg 1980
(cereal grains)
The label should be as follows:
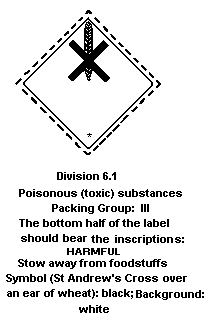 The European Community legislation requires labelling as a dangerous
substance using the symbol:
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink, and
animal feeding stuff.
7.5 Waste Disposal
In some countries, permits are required to empty pyrethroids into
water.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed., Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of carcinogenic
risk of chemicals to man. Lyons, International Agency for Research
on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols., Washington DC, US Department of Health and Human
Services, US Departmnent of Labor (Publication No. DHSS (NIOSH)
01-123).
WHO (In press) Environmental Health Criteria No. 96: d-Phenothrin.
Geneva, World Health Organization.
WHO (1988) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1988-89. Geneva, World Health
Organization (unpublished document WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink, and
animal feeding stuff.
7.5 Waste Disposal
In some countries, permits are required to empty pyrethroids into
water.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed., Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of carcinogenic
risk of chemicals to man. Lyons, International Agency for Research
on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols., Washington DC, US Department of Health and Human
Services, US Departmnent of Labor (Publication No. DHSS (NIOSH)
01-123).
WHO (In press) Environmental Health Criteria No. 96: d-Phenothrin.
Geneva, World Health Organization.
WHO (1988) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1988-89. Geneva, World Health
Organization (unpublished document WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
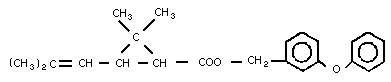 Molecular formula: C23H26O3
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-
dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)
methyl ester
CAS registry number:
Molecular formula: C23H26O3
CAS chemical name: Cyclopropanecarboxylic acid, 2,2-
dimethyl-3-(2-methyl-
1-propenyl)-(3-phenoxyphenyl)
methyl ester
CAS registry number:  7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. It is a representative but
non-exhaustive overview of current regulations, guidelines, and
standards.
Regulations and guidelines about chemicals can be fully understood
only within the framework of a country's legislation, and are always
subject to change. Therefore, they should always be verified with the
appropriate authorities.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Expert Committee on Pesticide Residues (JMPR)
evaluated d-phenothrin at its meetings in 1979, 1980, 1984, and 1988.
In 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg body weight
was established.
The Division of Vector Biology and Control, World Health Organization,
classified phenothrin as a technical product that is not likely to
present an acute hazard when used as recommended (WHO, 1988).
7.2 Exposure Limit Values
Some exposure limit values are given in the table that follows.
7.3 Specific Restrictions
No information is available.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies pyrethroids in:
- hazard class 6.1: poisonous substance
- packing group III: a substance with a relatively low risk of
poisoning during transport.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD Intake from FAO/WHO Temporary acceptable daily intake 0.04 mg/kg 1985
(TADI) (mg/kg body weight for
d-phenothrin)
FOOD Plant residues FAO/WHO Temporary maximum residue limit
(TMRL)
- Wheat bran (unprocessed) 15 mg/kg 1980
FAO/WHO Maximum residue limit (MRL) 5 mg/kg 1980
(cereal grains)
The label should be as follows:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. It is a representative but
non-exhaustive overview of current regulations, guidelines, and
standards.
Regulations and guidelines about chemicals can be fully understood
only within the framework of a country's legislation, and are always
subject to change. Therefore, they should always be verified with the
appropriate authorities.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Expert Committee on Pesticide Residues (JMPR)
evaluated d-phenothrin at its meetings in 1979, 1980, 1984, and 1988.
In 1988, an acceptable daily intake (ADI) of 0-0.07 mg/kg body weight
was established.
The Division of Vector Biology and Control, World Health Organization,
classified phenothrin as a technical product that is not likely to
present an acute hazard when used as recommended (WHO, 1988).
7.2 Exposure Limit Values
Some exposure limit values are given in the table that follows.
7.3 Specific Restrictions
No information is available.
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies pyrethroids in:
- hazard class 6.1: poisonous substance
- packing group III: a substance with a relatively low risk of
poisoning during transport.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD Intake from FAO/WHO Temporary acceptable daily intake 0.04 mg/kg 1985
(TADI) (mg/kg body weight for
d-phenothrin)
FOOD Plant residues FAO/WHO Temporary maximum residue limit
(TMRL)
- Wheat bran (unprocessed) 15 mg/kg 1980
FAO/WHO Maximum residue limit (MRL) 5 mg/kg 1980
(cereal grains)
The label should be as follows:
 The European Community legislation requires labelling as a dangerous
substance using the symbol:
The European Community legislation requires labelling as a dangerous
substance using the symbol:
 The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink, and
animal feeding stuff.
7.5 Waste Disposal
In some countries, permits are required to empty pyrethroids into
water.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed., Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of carcinogenic
risk of chemicals to man. Lyons, International Agency for Research
on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols., Washington DC, US Department of Health and Human
Services, US Departmnent of Labor (Publication No. DHSS (NIOSH)
01-123).
WHO (In press) Environmental Health Criteria No. 96: d-Phenothrin.
Geneva, World Health Organization.
WHO (1988) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1988-89. Geneva, World Health
Organization (unpublished document WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
The label must read:
Harmful by inhalation, in contact with skin and if swallowed;
keep out of reach of children; keep away from food, drink, and
animal feeding stuff.
7.5 Waste Disposal
In some countries, permits are required to empty pyrethroids into
water.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed., Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of carcinogenic
risk of chemicals to man. Lyons, International Agency for Research
on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1983. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vols., Washington DC, US Department of Health and Human
Services, US Departmnent of Labor (Publication No. DHSS (NIOSH)
01-123).
WHO (In press) Environmental Health Criteria No. 96: d-Phenothrin.
Geneva, World Health Organization.
WHO (1988) The WHO recommended classification of pesticides by
hazard. Guidelines to classification 1988-89. Geneva, World Health
Organization (unpublished document WHO/VBC/88.953).
WORTHING, C.R. & WALKER, S.B. (1983) The pesticide manual. 7th ed.,
Lavenham, Lavenham Press Limited, British Crop Protection Council.
