
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 10
CARBON DISULFIDE
This report contains the collective views of an
international group of experts and does not necessarily
represent the decisions or the stated policy of either
the World Health Organization or the United Nations
Environment Programme.
Published under the joint sponsorship of
the United Nations Environment Programme
and the World Health Organization
World Health Organization
Geneva, 1979
ISBN 92 4 154070 2
(c) World Health Organization 1979
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion whatsoever
on the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR CARBON DISULFIDE
1. SUMMARY AND RECOMMENDATIONS FOR FURTHER RESEARCH
1.1 Summary
1.1.1 Uses and sources of exposure
1.1.2 Populations at risk
1.1.3 Estimation of exposure
1.1.4 Metabolism
1.1.5 Mechanisms of toxic action
1.1.6 Carbon disulfide poisoning; evaluation of the
health risk to man
1.1.7 Diagnosis of carbon disulfide poisoning
1.1.8 Surveillance of exposed workers
1.2 Recommendations for further research
1.2.1 Analytical aspects
1.2.2 Studies on health effects
1.2.3 Mechanisms of toxic action
2. PROPERTIES AND ANALYTICAL METHODS
2.1 Chemical and physical properties
2.2 Analytical procedures
2.2.1 Measurement of carbon disulfide in air
2.2.2 Sampling methods
2.2.2.1 The activated charcoal tube method
2.2.2.2 The liquid absorption method
2.2.3 Methods for the determination of carbon disulfide
2.2.3.1 Direct measurement using gas detector
tubes
2.2.3.2 Photometric determination
2.2.3.3 Gas-liquid chromatographic determination
2.2.3.4 Continuous measurement using gas
analysers
2.2.3.5 Determination of metabolites in urine
3. EXPOSURE TO CARBON DISULFIDE
3.1 Occupational exposure
3.2 Community exposure
4. METABOLISM
4.1 Absorption
4.1.1 Inhalation
4.1.2 Skin absorption
4.2 Distribution and biotransformation
4.2.1 Balance of absorbed carbon disulfide
4.2.2 Transport by the bloodstream
4.2.3 Determination of carbon disulfide in blood
4.2.4 Distribution in the organism
4.2.5 Binding in blood and tissues
4.3 Elimination of carbon disulfide and metabolites
4.3.1 Elimination by breath, saliva, sweat, and faeces
4.3.2 Excretion of carbon disulfide and metabolites in
urine
5. BIOCHEMICAL EFFECTS OF CARBON DISULFIDE
5.1 Chelating effects of carbon disulfide metabolites
5.2 Effects on enzyme systems
5.3 Effects on vitamin metabolism
5.3.1 Vitamin B6
5.3.2 Nicotinic acid
5.4 Effects on catecholamine metabolism
5.5 Effects on lipid metabolism
5.6 Interaction with microsomal drug metabolism
6. CARBON DISULFIDE POISONING
6.1 Historical review
6.2 Clinical picture of carbon disulfide poisoning
6.3 Effects on organ systems
6.3.1 Dermatological effects
6.3.2 Ophthalmological effects
6.3.3 Otological effects
6.3.4 Respiratory effects
6.3.5 Gastrointestinal effects
6.3.6 Hepatic effects
6.3.7 Renal effects
6.3.8 Haematological effects
6.3.9 The endocrine system
6.3.10 Effects on the nervous system
6.3.10.1 Central nervous system
6.3.10.2 Peripheral nervous system
6.3.11 Cardiovascular effects
6.3.12 Carcinogenicity and mutagenicity
6.3.13 Teratogenic effects
6.3.14 Other effects
6.3.15 Interactions with other chemical compounds
6.4 Diagnosis
6.5 Surveillance of the health of exposed workers
6.6 Contraindications for exposure to carbon disulfide
7. EXPOSURE-EFFECT AND EXPOSURE-RESPONSE RELATIONSHIPS
7.1 Validity of exposure data
7.2 Experimental data
7.2.1 Acute animal exposure
7.2.2 Long-term animal exposure
7.3 Epidemiological data
7.3.1 Neurological and behavioural effects
7.3.2 Cardiovascular effects
7.3.3 Ophthalmological effects
7.3.4 Gonadal effects
8. CONTROL OF EXPOSURE IN THE VISCOSE INDUSTRY
REFERENCES
ANNEX I Production of viscose and its end-products
ANNEX II Maximum permissible concentrations for carbon disulfide
in different countries
NOTE TO READERS OF THE CRITERIA DOCUMENTS
While every effort has been made to present information in the
criteria documents as accurately as possible without unduly delaying
their publication, mistakes might have occurred and are likely to
occur in the future. In the interest of all users of the environmental
health criteria documents, readers are kindly requested to communicate
any errors found to the Division of Environmental Health, World Health
Organization, Geneva, Switzerland, in order that they may be included
in corrigenda which will appear in subsequent volumes.
In addition, experts in any particular field dealt with in the
criteria documents are kindly requested to make available to the WHO
Secretariat any important published information that may have
inadvertently been omitted and which may change the evaluation of
health risks from exposure to the environmental agent under
examination, so that the information may be considered in the event of
updating and re-evaluation of the conclusions contained in the
criteria documents.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR
CARBON DISULFIDE
Participants
Members
Dr G. Avilova, Institute of Hygiene and Preventive Medicine, Academy
of Medical Sciences, Moscow, USSR
Dr A. Cavalleri, Institute of Occupational Medicine, University of
Pavia, Pavia, Italy
Dr D. Djuric, Institute of Occupational and Radiological Health,
Belgrade, Yugoslavia
Professor K. J. Freundt, Institute of Pharmacology and Toxicology,
Faculty of Clinical Medicine, Mannheim, Federal Republic of
Germany
Dr S. Hernberg, Institute of Occupational Health, Helsinki, Finland
E. Lukas, Institute of Hygiene and Epidemiology, Centre of Industrial
Hygiene and Occupational Diseases, Prague, Czechoslovakia
Professor A. A. E. Massoud, Department of Preventive and Industrial
Medicine, Ein Shams University, Cairo, Egypt
Professor W. O. Phoon, Department of Social Medicine and Public
Health, Faculty of Medicine, University of Singapore, Singapore
Mr V. Rose, National Institute for Occupational Safety and Health,
Rockville, MD, USA
Dr S. Tarkowski, Department of Biochemistry, Institute of Occupational
Medicine, Lodz, Poland
Professor J. Teisinger, Institute of Hygiene and Epidemiology, Prague,
Czechoslovakia (Chairman)
Dr H. Thiele, Central Institute for Occupational Medicine, Berlin,
German Democratic Republic
Professor S. Yamaguchi, Department of Public Health, Tsukuba
University, School of Medicine, Niihari-Gun, Ibaraki-ken, Japan
Professor S. H. Zaidi, Industrial Toxicology Research Centre, Lucknow,
India
Secretariat
A. David, Institute of Hygiene and Epidemiology, Centre of Industrial
Hygiene and Occupational Diseases, Prague, Czechoslovakia
(National Coordinator and Co-Chairman)
Dr M. A. El Batawi, Chief Medical Officer, Office of Occupational
Health, World Health Organization, Geneva, Switzerland
(Secretary)
ENVIRONMENTAL HEALTH CRITERIA FOR CARBON DISULFIDE
A WHO Task Group on Environmental Health Criteria for Carbon
Disulfide met in Prague from 13 to 20 June 1977. Dr M. El Batawi,
Chief Medical Officer, Office of Occupational Health, opened the
meeting on behalf of the Director-General and expressed the
appreciation of the Organization to the Government of Czechoslovakia
for kindly acting as host to the meeting. In reply, the Group was
welcomed by Professor J. Teisinger, Institute of Hygiene and
Epidemiology, Prague. The Task Group reviewed and revised the second
draft criteria document and made an evaluation of the health risks
from exposure to carbon disulfide.
The first draft of the criteria document was prepared by
Dr. Djuric, Institute of Occupational and Radiological Health, Belgrade,
Yugoslavia, in consultation with Professor Teisinger, Dr E. Lukas,
Institute of Hygiene and Epidemiology, Prague, Czechoslovakia, and
several research workers in Belgrade and Prague. The second draft was
prepared by Dr S. Hernberg, Institute of Occupational Health,
Helsinki, Finland taking into consideration comments by Professor
K. Freundt, Institute of Toxicology and Pharmacology, Mannheim, Federal
Republic of Germany, Professor Sh. Goto, Osaka University, Japan,
Dr. I. Lancranjan, Institute of Hygiene and Public Health, Clinic of
Occupational Diseases, Bucharest, Romania, Dr J. Lieben of the
American Viscose Division, PM Corporation, Philadelphia, USA, Dr A.
Massoud, National Research Centre, Cairo University, Egypt, Dr A.M.
Seppäläinen, Institute of Occupational Health, Helsinki, Finland, and
Dr P. G. Vertin, Institute of Social Medicine, Catholic University of
Nijmegen, Netherlands.
The Secretariat wishes to acknowledge the collaboration of these
experts and, in particular, to thank Dr Djuric and Dr Hernberg for
their valuable help in all phases of the preparation of the document,
and Dr H. Nordman, Institute of Occupational Health, Helsinki,
Finland, for his assistance in the scientific editing.
This document is based primarily on original publications listed
in the reference section but much valuable information has also been
obtained from various publications reviewing the toxicity and health
aspects of carbon disulfide including those of the US National
Institute of Occupational Safety and Health (NIOSH, 1977) and Brieger
& Teisinger, ed. (1966). In addition, much useful data has been drawn
from reports of several international symposia and meetings including:
Zbornik radova o toksikologiji CS2, Yugoslavia, Loznica, 3-5 June
1965; the II International Symposium on the Toxicology of Carbon
Disulfide, Yugoslavia, Banja Kovilijaca, 25-28 May 1971; the III
International Symposium on the Toxicology of Carbon Disulfide, Egypt,
Cairo and Alexandria, 4-9 May 1974; and the IV International Symposium
on Occupational Health in the Production of Artificial Fibres,
Finland, Helsinki and Valkeakoski, 6-10 June 1977.
Details of the WHO Environmental Health Criteria Programme
including some terms frequently used in the documents may be found in
the general introduction to the Environmental Health Criteria
Programme published together with the environmental health criteria
document on mercury (Environmental Health Criteria 1, Mercury, Geneva,
World Health Organization, 1976), now also available as a reprint.
The following conversion factor has been used in this document:
carbon disulfide 1 ppm = 3.12 mg/m3
When converting values expressed in ppm to mg/m3 the numbers
have been rounded up to 2 or, exceptionally, 3 significant figures.
Where concentrations were expressed as ppm in the original
publication, this value has been given in parentheses together with
the converted value.
1. SUMMARY AND RECOMMENDATIONS FOR FURTHER RESEARCH
1.1 Summary
1.1.1 Uses and sources of exposure
By far the most important use of carbon disulfide in industry is
in the production of viscose rayon fibres. It is also used, to some
extent, as a solvent in various industrial processes including the
refining of paraffin and petroleum, and more recently in the
production of flotation agents and herbicides. However, the risk of
being exposed to high concentrationsa of carbon disulfide during
these processes is small compared with that in the viscose industry.
Viscose rayon fibres are used in the production of rayon filament
textile yarn, rayon tire yarn, rayon stable fibre and Cellophane film.
In these processes, carbon disulfide exposure occurs concomitantly
with exposure to hydrogen sulfide. The amounts of carbon disulfide and
hydrogen sulfide vapour liberated depend on the process. For every
kilogram of viscose used, about 20-30 g of carbon disulfide and 4-6 g
of hydrogen sulfide will be emitted. About 0.6-1.0 kg of viscose is
used per hour in the different processes involved in the production
of textile yarn. However, exposure to carbon disulfide is usually
highest in connection with the production of staple fibre and
Cellophane, where the equivalent amounts of viscose used are
approximately 70-100 kg and 1800-2000 kg per hour, respectively.
1.1.2 Populations at risk
Carbon disulfide is a typical industrial toxic chemical and
exposure is almost exclusively confined to occupational situations. In
theory, any worker engaged in processes using carbon disulfide may be
exposed to some degree. However, in practice, only workers in the
viscose rayon industry are exposed to concentrations high enough to
have deleterious effects on health. The exposure of the general
population living in the vicinity of carbon disulfide-emitting
industries cannot be assessed at present, because information is
inadequate.
__________
a Throughout the document the word concentration refers to mass
concentration unless otherwise stated.
1.1.3 Estimation of exposure
Exposure to carbon disulfide can be estimated either by direct
measurement of air concentrations or by the determination of carbon
disulfide metabolites in the urine of exposed individuals. Air samples
can be taken either at fixed sites, or from the breathing zone of the
workers. Sampling at fixed sites is recommended for engineering
purposes, while sampling from the breathing zone is indicated for the
assessment of personal exposure.
Monitoring at fixed sites is best done by continuous measurement
with gas analysers based on electrical conductivity or light
absorption in the infrared region. Gas detector tubes may be used for
preliminary screening, since the procedure is rapid and simple, but
their usefulness is limited because of lack of accuracy and a high
detection limit; thus, this procedure should always be complemented by
more accurate methods.
Personal exposure is best monitored by samples collected from the
breathing zone of the workers, using portable samplers. The carbon
disulfide is adsorbed on activated charcoal and later determined by
gas chromatography. Absorption in liquids is not possible, when using
portable samplers. Depending on desorption efficiency and the type of
gas chromatograph used, determination of carbon disulfide
concentrations below 1 mg/m3 is possible. Furthermore, hydrogen
sulfide does not cause interference.
The method most extensively used for the indirect assessment of
personal exposure is the iodine-azide test in which the concentration
of carbon disulfide metabolites present in the urine is measured. The
"chronometric" iodine-azide test, based on the time elapsing from
adding the iodine-azide reagent to the urine until decolorization of
the iodine solution takes place, offers a simple method to be used at
the plant level, but its rather high detection limit restricts its use
to exposure levels in excess of 50 mg/m3. Titrimetric modification of
the same test increases sensitivity and allows assessment of exposure
at levels down to 10 mg/m3.
Because of the poor correlation with carbon disulfide
concentrations in air as well as for analytical reasons, the
concentration of carbon disulfide in blood is not a useful test of
exposure.
1.1.4 Metabolism
Inhalation is the principal route of absorption of carbon
disulfide in man, equilibrium between the carbon disulfide contents of
inhaled and exhaled air being reached in about 1-2 h. At this point,
retention is about 40-50%. Absorption through the skin is a much less
important route than inhalation and other routes are negligible.
Carbon disulfide is distributed in the organism by the blood stream.
It is taken up by the erythrocytes and plasma in the blood in the
ratio of 2:1. It is readily soluble in fats and lipids and binds to
amino acids and proteins; hence, it disappears rapidly from the blood
stream and has a high affinity for all tissues and organs. Because of
the rapid elimination of carbon disulfide, the distribution pattern in
the human organism has not been fully elucidated. Ten to 30% of
absorbed carbon disulfide is exhaled, less than 1% is excreted in the
urine, and the remaining 70-90% undergoes biotransformation before
excretion in the urine in the form of metabolites.
1.1.5 Mechanisms of toxic action
The biochemical mechanisms of the adverse effects of carbon
disulfide are largely unknown. However, a number of possible
mechanisms have been suggested including:
(a) A chelating effect of the metabolites on various essential
trace metals;
(b) Inhibition of some enzymes (this may be explained, to some
extent, by chelation, but the nature of other mechanisms is not yet
known);
(c) Disturbance of the vitamin metabolism (experimental
evidence in animals has shown an impairment of vitamin B6 and
nicotinic acid metabolism);
(d) Disturbance of the catecholamine metabolism;
(e) Disturbance of the lipid metabolism;
(f) Interaction with the microsomal drug metabolizing system
(the liver toxicity may be, at least, partly explained by the
destruction of cytochrome P-450 via the oxidative desulfuration of
carbon disulfide).
1.1.6 Carbon disulfide poisoning; evaluation of the
health risk to man
Carbon disulfide can cause both acute and chronic forms of
poisoning. Massive, short-term exposure to concentrations of about
10 000 mg/m3 or more can cause "hyperacute" poisoning, characterized
by rapid falling into coma and, eventually, death. Acute and subacute
poisoning is associated with short-term exposure to concentrations of
3000-5000 mg/m3 accompanied by predominantly psychiatric and
neurological symptoms such as extreme irritability, uncontrolled
anger, rapid mood changes, euphoria, hallucinations, paranoic and
suicidal tendencies, and manic delirium. Exposure over many years may
produce the syndrome of chronic poisoning manifested by a variety of
symptoms and signs arising from manifold adverse effects on different
organ systems. Because of the lack of reliable retrospective data on
exposure levels, dose-effect and dose-response relationships are
extremely difficult to establish, and the no-observed-effect-level is
unknown for most effects.
Psychiatric signs and symptoms indicative of adverse effects on
the central nervous system following prolonged exposure to high
concentrations of carbon disulfide include restlessness, excitation,
and loss of temper with gradual development of anxiety, depression,
and paranoic tendencies. The development of chronic encephalopathy has
been associated with exposure to levels of 150 mg/m3 or more over a
period of several years. Psychological and behavioural changes have
been recorded following exposure to levels ranging from 30-120 mg/m3
for more than 6 years, and increases in the frequency and severity of
such symptoms as headache, impairment of memory, rapid mood changes,
paraesthesia, and fatigue have been noted at concentrations ranging
from 20-90 mg/m3. As poisoning progresses further, neurological
symptoms become more predominant. Both pyramidal and extrapyramidal
symptoms may develop indicating impairment of the central nervous
system.
Symmetric polyneuropathy primarily affecting the nerves of the
lower extremities and characterized by paraesthesia, dysaesthesia,
fatiguability, and diffuse pain, sometimes with hyperaesthesia or
hypersensitivity of the muscles, constitutes a well known syndrome.
Recent studies indicate that peripheral neurological dysfunction such
as the reduced conduction velocity of peripheral nerves may follow
prolonged exposure to carbon disulfide concentrations in the range of
30-90 mg/m3. Sensory polyneuropathy with increased pain threshold has
been reported following 10-15 years of exposure to concentrations as
low as 10 mg/m3.
Vascular atherosclerotic changes are also caused by long-term
exposure. Studies in Finland, Norway, and the United Kingdom have
shown that carbon disulfide promotes the development of coronary heart
disease and that exposure to levels ranging from 30 to 120 mg/m3, for
more than 10 years, appears to increase coronary mortality.
Ophthalmological changes of various types, such as increased
pressure, retrobulbar neuritis, etc. were formerly connected with
severe forms of poisoning but, under present conditions of exposure,
such findings are uncommon. However, an increased frequency of retinal
microaneurysms, related to the duration and intensity of exposure, has
been found in Japanese workers. No such abnormalities have been
diagnosed, with certainty, in European workers, in spite of
well-controlled comparative studies.
Effects on the endocrine system include a reduction in adrenal
activity attributable to reduced secretion of corticotrophine,
impairment of spermatogenesis, and disturbance of the hormonal balance
in women, evidenced by menstrual irregularities, spontaneous
abortions, and premature deliveries. Moreover, the thyroid function
may be altered, probably due to impairment of the
hypothalamic-hypophyseal system. The most sensitive endocrine changes,
i.e., depression of blood progesterone, increase of estriol, and
irregular menstruation may occur at concentrations as low as
10 mg/m3, whereas increases in spontaneous abortions and premature
births have been reported in association with an exposure level of
30 mg/m3.
Gastrointestinal symptoms including dyspeptic complaints,
gastritis, and ulcerative changes have been found in workers, heavily
exposed to carbon disulfide.
1.1.7 Diagnosis of carbon disulfide poisoning
The effects of carbon disulfide are nonspecific, making
individual diagnosis a matter of probability based on the confirmation
of exposure, the presence of symptoms and signs compatible with carbon
disulfide exposure, and the exclusion of other diseases. In workers
with ascertained exposure, carbon disulfide poisoning should be
suspected whenever subjective and neurasthenic symptoms, signs of
peripheral neuropathy, psychological disturbances, or vascular changes
are present. The diagnosis in acute forms of poisoning is
straightforward, whereas the insidious development of adverse effects
in chronic carbon disulfide poisoning makes early detection difficult.
The probability of an accurate diagnosis increases as the number of
abnormalities present increases. One recent study suggests that a
positive diagnosis can be made only if changes in the choroidal
circulation are found and provided that these occur in conjunction
with polyneuropathy, or behavioural changes, or both.
1.1.8 Surveillance of exposed workers
For the early detection of adverse effects and for the continuous
surveillance of exposed workers, medical examinations should be
carried out once or twice yearly. The following examinations are
recommended for a pre-employment check: (a) thorough medical
history; (b) clinical and neurological examination;
(c) electromyogram (EMG) examination; especially conduction velocity
measurements; (d) psychological tests; (e) measurement of the
blood pressure; (f) electrocardiography; and (g) serum cholesterol
determinations.
All or some of these examinations should be repeated regularly
during the supervision of exposed workers, whenever exposure exceeds
half the maximum permissible concentration. It is recommended that
personal exposure rather than background exposure should be measured.
For this purpose, either personal samplers, or the iodine-azide test
should be employed. The iodine-azide test should be carried out from 2
to 12 times a year depending on the level of exposure. Recommendations
for early detection and prevention of adverse effects should, of
course, be combined with technical and administrative measures for the
protection of the health of exposed workers. The adoption of a maximum
permissible concentration of carbon disulfide in the air is considered
indispensable, and it is equally important to take all measures needed
for achieving and maintaining conditions that will keep exposure below
this level.
1.2 Recommendations for Further Research
1.2.1 Analytical aspects
In the field of occupational hygiene technology there is a need
to:
(a) Improve and harmonize the methods of assessment of carbon
disulfide in the work environment with a view to facilitating the
comparability of data;
(b) to improve, use, and harmonize personal sampling techniques
in epidemiological studies; and
(c) to further investigate the relationship, if any, between
exposure as measured by personal sampling, and the iodine-azide test.
1.2.2 Studies on health effects
There is a need for internationally co-ordinated research on
exposure-response relationships using, as far as possible, harmonized,
experimental and epidemiological methods.
It is advisable to undertake comparative studies on the
relationships between carbon disulfide concentrations and coronary
artery disease both in countries with a high, and countries with a low
prevalence of the disease to find out whether or not the present
information from some industrialized countries, such as Finland, is
applicable to countries with a low prevalence of coronary artery
disease.
The possible carcinogenicity, teratogenicity, and mutagenicity of
carbon disulfide should be studied.
The effects of continuous exposure to low levels of carbon
disulfide, such as may be found in the neighbourhood of factories, are
unknown. Studies are recommended to elucidate exposure levels and any
health risks associated with such exposure, and to introduce control
measures.
1.2.3 Mechanisms of toxic action
The mechanisms of the toxic action of carbon disulfide are still
hypothetical and further studies concerning the biochemical basis of
these effects deserve high priority.
2. PROPERTIES AND ANALYTICAL METHODS
2.1 Chemical and Physical Properties
Carbon disulfide (CS2) when pure, is a colourless, mobile,
refractive solution of sweetish aromatic odour, similar to that of
chloroform. However, the crude technical product is a yellowish liquid
with a disagreeable odour of decaying radishes.
Carbon disulfide evaporates at room temperature and the vapour is
2.62 times heavier than air (one litre of vapour weighs 3.017 g).
Carbon disulfide vapour forms a highly explosive mixture with air.
Furthermore, liquid carbon disulfide may produce a static electric
charge that can initiate an explosion. Thus, it must be handled with
the greatest caution, and should never come into contact with an
electric charge or spark, a flame, or even high temperatures. Carbon
disulfide is spontaneously flammable at 130-140°C, and fire
extinguishers of the foam type must always be available, when it is
handled.
Because of its solubility in fats and lipids, carbon disulfide is
widely used as a solvent for fats, lipids, resins, rubber, sulfur
monochloride, white phosphorus, and some other substances.
Some basic physical and chemical properties of carbon disulfide
are summarized in Table 1.
Table 1. Physicochemical data on carbon disulfide.a
Synonym carbon disulphide, carbon
bisulphide
Formula CS2
Relative molecular mass 76.14
Melting point -111.53° C
Boiling point 46.3° C
Density 1 263 g/cm3 at 20° C
Water solubility 0.2 g/100 ml at 20° C
Vapour density (air = 1) 2.64
Flash point below -30° C (closed cup)
Explosive limits (% by lower 1.0%
volume in air) upper 50.0%
Vapour pressure at (28° C) 53.3 kPa (400 mmHg)
a From: Weast, R. C. (1970); Faith et al. (1965).
2.2 Analytical Procedures
2.2.1 Measurement of carbon disulfide in air
Control of exposure depends, to a great extent, on the
measurement of carbon disulfide concentrations in air.
Samples of carbon disulfide may be extracted either by the
activated charcoal tube method or by the liquid absorption method.
The following methods are recommended for the measurement of
carbon disulfide:
(a) direct measurement using gas detector tubes;
(b) photometric determination of carbon disulfide samples taken
by the liquid absorption method;
(c) gas-liquid chromatography of carbon disulfide samples taken
by the activated charcoal tube method;
(d) continuous measurement by gas analyser.
2.2.2 Sampling methods
2.2.2.1 The activated charcoal tube method
This method of sampling is preferable because the sample can be
taken from the breathing zone of the worker (see for example Truhaut
et al., 1972) and because, when combined with the biological
iodine-azide test (section 2.2.3.5), it offers the best measure of
personal exposure to carbon disulfide. The sampling device, which
consists of a charcoal tube fastened to the worker's shoulder and a
pump fastened to the belt, is small enough to be worn for the whole
working period without discomfort.
The carbon disulfide, which is absorbed by activated carbon in
the tube, is later desorbed by a solvent and determined by gas
chromatography (section 2.2.3). To determine the time-weighted average
concentration of carbon disulfide, the volume of air sampled should be
large enough to allow the determination of concentrations below the
threshold limit value (TLV). A sampling period of 15 minutes should be
used for the determination of maximum or ceiling concentrations. An
advantage of this sampling method is that the presence of hydrogen
sulfide does not impair sampling efficiency (McCammon et al., 1975).
Further information concerning possible interference with sampling
efficiency can be found in reports by McCammon et al. (1975) and NIOSH
(1977).
2.2.2.2 The liquid absorption method
The liquid absorption method can only be used for the
determination of carbon disulfide concentrations at fixed sites. The
principle of the method is that air is drawn through the absorption
liquid using two fritted bubblers in series. The carbon disulfide in
the air reacts with the liquid, which is an ethanolic solution of
copper salt and diethylamine. Hydrogen sulfide, present in the air,
must be trapped on cotton-wool treated with lead acetate before the
air enters the absorption solution (Bagon et al., 1973).
2.2.3 Methods for the determination of carbon disulfide
2.2.3.1 Direct measurement using gas detector tubes
This method of measurement is based on a reaction between the
tested gas and a specific reagent mixture. For carbon disulfide, the
indicating layer in the detector tube contains a combination of a
copper salt and an alkylamine that yields a copper-
dialkyldithiocarbamate complex with carbon disulfide. A known
volume of air is drawn through the tube. The length of the coloured
zone is a measure of the concentration. Detector tube systems provide
a rapid, inexpensive, and simple method for evaluating the level of a
contaminant in the industrial environment, the relative standard
deviation of which is about 20-30%. However, the results of this
method are only approximate and, if measurements indicate that air
contaminant levels are excessive, additional measurements should be
made by more accurate methods.
2.2.3.2 Photometric determination
The principle of this colorimetric method is that carbon
disulfide reacts in an ethanolic solution with diethylamine and a
copper salt to give a yellow-brown metallic complex of
diethyldithiocarbamate. The colour of the solution is directly
proportional to the concentration of carbon disulfide (Department of
Employment and Productivity, 1968).
The carbon disulfide concentration in the sample can be
determined using a spectrophotometer at 420 nm. Five mg of carbon
disulfide per m3 of air may be determined by this method. Hydrogen
sulfide causes interference and should be removed by the method
described in section 2.2.2.2 (Cullen, 1964).
2.2.3.3 Gas-liquid chromatographic determination
Gas chromatography in combination with the activated charcoal
sampling method (section 2.2.2.1) is widely used for the determination
of personal exposure to carbon disulfide. A method using a gas
chromatograph equipped with a flame photometric detector and a sulfur
filter has recently been described in detail (NIOSH, 1977). The assay
was validated over a range of 45.6-182.3 mg of carbon disulfide per
m3 of air, at an atmospheric temperature and pressure of 22°C and
102.1 kPa (766 mmHg), respectively, using a 6 litre sample. With this
concentration range, the coefficient of variation was 0.059
corresponding to a standard deviation of 5.6 mg/m3, at a carbon
disulfide concentration of 93 mg/m3. However, the detection of much
smaller amounts is possible using this method, if the desorption
efficiency is adequate (NIOSH, 1977). It must be emphasized that any
compound having the same retention time as the analyte may cause
interference and that, if this possibility exists, separation
conditions (column packing, temperature, etc.) should be adjusted
accordingly.
2.2.3.4 Continuous measurement using gas analysers
Some types of gas analysers are convenient for the continuous
monitoring of carbon disulfide in workroom air. The measurements can
be carried out at one or several fixed sampling sites depending on the
construction of the equipment.
Analysers suitable for continuous monitoring include:
(a) Analysers based on electrical conductivity in which an air
flow is conducted through a suitable absorbing solution. The gas to be
measured reacts with the solution and changes its electrical
conductivity according to the concentration of the gas.
In the case of carbon disulfide, the gas must first be oxidized
in a combustion oven, the determination is then based on the reaction
of carbon dioxide or sulfur dioxide with the absorbing solution.
(b) Analysers based on light absorption in the infrared region
in which the measuring effect is based on the specific radiation
absorption of heteroatomic gases in the infrared spectral range
between 2.5 and 12 µm wavelength. Absorption occurs at strictly
separated frequencies that are associated with the natural vibrations
of the molecules.
When measuring low concentrations of carbon disulfide using
infrared analysers, some other gases, especially water vapour, can
cause interference. The interference can be eliminated and the
sensitivity improved, if the carbon disulfide is first oxidized in a
combustion oven to sulfur dioxide and the latter measured by
infrared-analyser.
Numerous systems for continuous gas monitoring have been
developed; detailed information concerning the measurement of carbon
disulfide by this method can be found in Schütz (1970), Leithe (1971),
Verdin (1973), Weigman (1973).
2.2.3.5 Determination of metabolites in urine
Since there is only a poor, if any, correlation between carbon
disulfide concentrations in blood and air, and only 1% or less of
absorbed carbon disulfide is excreted unmetabolized into the urine,
there is no basis for using the determination of carbon disulfide in
either blood or urine as an exposure test (section 4.2.3 and 4.3.2).
In contrast, good results have been obtained using the concentration
of metabolites of carbon disulfide in the urine as a measure of
exposure.
(a) The iodine-azide test is based on the finding of Yoshida
(1955) that the iodine-azide reaction:
2NaN3 + I2 -> 3N2 + 2NaI
is catalysed by a metabolite present in the urine of animals exposed
to carbon disulfide. Subsequently, it was found that the C-SH and C-S
groups act as catalysts in the reaction, and a quantitative test was
developed based on the time interval between adding the iodine-azide
reagent to urine and the decolorization of the iodine solution, as
measured by a stop watch (Vasak, 1963; Vasak et al., 1963). In order
to simplify the test, the time was corrected according to the
creatinine concentration to avoid the collection of 24-h urine
samples. This time served as a basis for the calculation of the
exposure coefficient, which was indirectly proportional to the
concentration of carbon disulfide metabolites excreted in the urine.
Vasak et al. (1967) later elaborated a diagram for the evaluation of
the average concentration of carbon disulfide during the shift.
Provided that the urine is not too dilute, i.e., the creatinine
concentration is not much below 2.25 mg/ml, exposure may be considered
negligible if decolorization of the iodine-azide reagent does not take
place within 3 h. The "chronometric" iodine-azide test may be
successfully used on workers, when the average exposure is above
50 mg/m3 (Djuric et al., 1965). However, recent data from Sweden
indicate that a short decolorization time in the iodine-azide test may
occur in some workers exposed to 30-40 mg/m3, suggesting individual
differences in the reaction to carbon disulfide (Kolmodin-Hedman,
1976).
A modification of the "chronometric" test was developed by
Jakubowski (1968, 1971). The modified procedure was not based on the
time of reaction, but on measurements of the amount of iodine used for
titration of carbon disulfide metabolites catalysing the iodine-azide
reaction in 1 ml of urine and calculated for a standard creatinine
concentration of 1.5 mg/kg. With this method, it was possible to
assess exposure to levels as low as 10 mg of carbon disulfide per m3
of air with a precision of ±20%.
(b) A method for the determination of thiourea was developed by
Pergal et al. (1977a), based on the colorimetric determination of a
complex produced in a reaction between thiourea present in the urine
and potassium ferrocyanide (K4FeCN6) present as a reagent in an acid
media. Levels of thiourea excretion between 0.001 and 0.1 mg/ml could
be determined by this method. Preliminary results showed that the
amount of thiourea in the urine sampled at the end of the working
shift was not strongly correlated with the results of the iodine-azide
test. It is necessary to study the excretion dynamics of this
metabolite to establish if this method can be used as an exposure
test. So far, the results suggest that the excretion of this
metabolite reflects the rate of carbon disulfide metabolism rather
than recent exposure (Pergal et al., 1977a).
3. EXPOSURE TO CARBON DISULFIDE
3.1 Occupational Exposure
Carbon disulfide was first used as a solvent in 1851 as a
phosphorus solvent in the manufacture of matches. During the 19th
century, it was used as a solvent for fats, lacquers, and camphor, for
the refining of jelly, paraffin, and petroleum, and in the extraction
of oil from olives, palmstones, bones, and rags. In the latter half of
the century, it was used extensively in the vulcanization of rubber.
These applications still prevail to some extent and, today, it is also
used in the production of flotation agents, herbicides, rubber
accelerators, and neoprene cement, and in the fumigation of grain.
However, by far the most important use of carbon disulfide is in the
production of viscose rayon fibres.
The industrial production of viscose, which began in 1906,
quickly expanded all over the world, particularly during and after
World War I. The synthesis of other artificial fibre,; after World War
II slowed clown this expansion, but rayon fibres are still of
considerable industrial importance. As viscose rayon production is the
most important source of exposure to carbon disulfide, a more detailed
description of the technological process and the exposure hazards that
may be associated with various stages of production has been given in
Annex I. The brief account given here highlights the processes
associated with the highest risk of exposure.
Carbon disulfide is introduced into viscose production during the
so-called process of xanthation, where it is added to shredded and
oxidized alkali cellulose to form sodium cellulose xanthate. Although
exposure to carbon disulfide at this stage is mechanically controlled,
exposure to high concentrations may still occur. The sodium cellulose
xanthate is dissolved in caustic soda to produce viscose that can be
further processed either by spinning to form textile yarn, tire yarn,
or staple fibre, or by casting to form Cellophane. Carbon disulfide,
and to a lesser extent hydrogen sulfide, are evolved during spinning
and casting, and exposure to high concentrations of carbon disulfide
can occur during doting and when filaments break. Carbon disulfide is
further emitted in the cutting of rayon filaments for staple fibre,
and in the washing and drying processes. Because of the high input of
viscose, carbon disulfide emissions are highest in the production of
staple fibre and Cellophane.
3.2 Community Exposure
At the present time, very little information is available
concerning exposure to carbon disulfide outside the workplace or the
effects on the general population. Although concentrations outside the
workplace are expected to be much lower than those found inside,
special consideration must be given to the possibility that
individuals in poor health or the very young may be exposed and also
that workers, who are exposed to carbon disulfide at work may also be
exposed during non-working hours if they live close to their place of
work.
In 1976, Peyton et al. reviewed the literature concerning
environmental studies of carbon disulfide and carbonyl sulfide. Both
compounds are emitted by man-made, as well as natural sources.
Although carbon disulfide appears to be relatively stable in the
atmosphere, oxidation leads to the formation of sulfur dioxide, carbon
monoxide, and carbonyl sulfide. It has been suggested that carbonyl
sulfide itself elicits a toxic response in man because of partial
decomposition to hydrogen sulfide in the lungs and bloodstream.
From the limited data available, it appears that individuals
living close to workplaces where carbon disulfide is used can be
exposed to high enough concentrations to result in measurable uptake.
When 70 children living 400 m from a factory discharging carbon
disulfide into the atmosphere were compared with a control group of 30
children living 15 km from the factory, physical and psychological
examinations did not show any health disorders in the exposed group
even though urine concentrations of carbon disulfide indicated
increased uptake compared with the controls (Helasova, 1969).
Environmental measurements were taken for both hydrogen sulfide and
carbon disulfide. Ninety-two out of 127 measurements of carbon
disulfide concentrations in air were higher than 0.01 mg/m3.
By applying data on workplace exposure to conditions in the
general environment, Peyton et al. (1976) recommended that limiting
long-term average concentrations to 0.3 mg of carbon disulfide per m3
of air should be sufficient to protect the general population against
long-term health effects. In the USSR, the maximum allowable
concentration for carbon disulfide in the ambient air is 0.03 mg/m3
with an allowable 24-h average of 0.005 mg/m3 (Bajkov, 1963). In
addition, the USSR has also established an allowable level of carbon
disulfide in waterways (prior to treatment) of 1.0 mg per litre
(Vinogradov, 1966).
4. METABOLISM
4.1 Absorption
Inhalation and skin contact are the only significant routes of
absorption of carbon disulfide. The only way carbon disulfide may
enter the human organism through ingestion is by accidental (or
intentional) intake.
4.1.1 Inhalation
Inhalation represents the main route of carbon disulfide
absorption in occupational exposure. Data reported earlier by
Teisinger & Soucek (1952), namely that, in spite of considerable
variation between individuals, absorption seemed to be proportional to
the concentration of carbon disulfide in inhaled air, were confirmed
by Demus (1967).
Toyama & Kusano (1953) studied the absorption of carbon disulfide
through the lungs of rabbits. They found that equilibrium in the
carbon disulfide contents of inhaled and exhaled air was reached after
90-150 min of exposure, and that 70-80% was retained at equilibrium.
Inhalation studies have also been performed on human volunteers, but
the data obtained have been diverse, even controversial (Teisinger &
Soucek, 1949; Teisinger, 1954; Brieger, 1961, 1967; Djuric, 1963,
1967; Davidson & Feinleib, 1972). It was reported by Madlo & Soucek
(1953) that equilibrium in man was reached during the first 90-120 min
of exposure and that, at this stage, the retention of carbon disulfide
was about 30% of the amount present in the inhaled air. However, in a
number of Japanese studies, Tazuka (1955) found that equilibrium was
reached 30-60 min after the beginning of exposure, Toyama & Harashima
(1962), after about 180 min, and Tahara (1961), at the end of a
working shift of 8 h (480 min). The discrepancies can probably be
explained by differences in exposure conditions.
In studies by Teisinger & Soucek (1949), higher retention was
observed in volunteers exposed for the first time to carbon disulfide
than in continuously exposed workers. In volunteers, equilibrium was
reached after 120 min of exposure. An initial retention of 80% fell to
45%, when equilibrium was reached. Equilibrium in industrial workers
was already reached after 45-60 min. Harashima & Masuda (1962)
obtained similar results with exposed workers but found a retention of
65% at equilibrium. Average retentions of 41% after the first 60 min
and 48% after 240 min of exposure were reported by Petrovic & Djuric
(1966).
Thus, the majority of authors agree that, in man, an equilibrium
between the carbon disulfide concentrations in inhaled and exhaled air
is reached during the first 60 min of exposure. The percentage
retained at equilibrium appears to be about 40-50% of the amount of
carbon disulfide in the inhaled air and depends on both the
concentration of carbon disulfide in the air and the partition
coefficient between blood and tissues. This percentage is lower in
continuously exposed workers than in volunteers exposed for the first
time to carbon disulfide. This difference should be taken into account
in the planning of inhalation studies as well as in the interpretation
of the results.
4.1.2 Skin absorption
As an organic solvent, carbon disulfide can be expected to pass
through the skin and this has been confirmed in a number of studies.
Dutkiewicz & Baranowska (1967) studied absorption from an aqueous
solution through the skin of immersed hands. The solution contained
0.33-1.67 g of carbon disulfide per litre and, after 1 h, the quantity
absorbed ranged from 0.23 to 0.78 mg/cm2 of skin. The authors
calculated that immersion of a hand for 1 h in a washing bath in a
viscose rayon plant could result in the absorption of 17.5 mg of
carbon disulfide into the organism.
It is obvious that workers exposed to carbon disulfide solution
and vapour will absorb some through the skin and that, though these
amounts will be less than the quantities inhaled, they will still be
important and should be considered in the evaluation of total
exposure.
4.2 Distribution and Biotransformation
4.2.1 Balance of absorbed carbon disulfide
In animal experiments, where carbon disulfide was administered
into the gastrointestinal tract, most of it was eliminated in the
faeces and only a small part was excreted by exhalation (Soucek,
1957). However, after intraperitoneal injection, rats and guineapigs
exhaled about 55% and 70%, respectively, of the amounts administered
(Soucek, 1959, 1960a,b).
Studies in man, as summarized by Soucek (1957), show that 10-30%
of the carbon disulfide absorbed into the body is exhaled and that
less than 1% is excreted unchanged in the urine; thus, 70-90%
undergoes biotransformation and is excreted in the form of
metabolites. Demus (1964) reached similar conclusions. About 10% of
the absorbed carbon disulfide represents a body burden that is
excreted slowly in the urine, mainly in the form of metabolites. In
contrast to these studies, Dutkiewicz & Baranowska (1967) reported
that, when carbon disulfide was absorbed through the skin, only 3% was
exhaled.
4.2.2 Transport by the bloodstream
There are differences among animal species with regard to the
affinity between carbon disulfide and blood. The affinity is higher in
rats than in guineapigs (Soucek, 1959, 1960a) and this is quite in
accordance with the differences in exhalation rates after
intraperitoneal injection, referred to in section 4.2.1. The
disappearance of carbon disulfide from the circulation can be
accelerated by the administration of a mixture of fresh air and 5%
carbon dioxide; this results in a more rapid disappearance of narcotic
effects (Soucek, 1959).
In man, the carbon disulfide that is not exhaled is distributed
in the body by the bloodstream, twice as much being taken up by
erythrocytes as by the plasma (Soucek & Pavelkova, 1953). Carbon
disulfide disappears quickly from the blood because of its affinity
for lipid-rich tissues and organs. However, traces of carbon disulfide
have still been found in the blood of exposed workers 80 h after
termination of exposure (Soucek & Pavelkova, 1953).
4.2.3 Determination of carbon disulfide in blood
Bartonicek (1957, 1958, 1959) showed that the determination of
carbon disulfide in blood did not give reproducible results and that
the correlation between carbon disulfide concentrations in blood and
air was very weak or non-existent. Thus, determination of the
concentration of carbon disulfide in the blood is not a useful test of
exposure. The reasons for these discrepancies are explained by the
results of studies by Bartonicek (1957, 1958, 1959) on "free" and
"bound" carbon disulfide (section 4.2.4).
4.2.4 Distribution in the organism
Soucek (1960a) established that the partition coefficients for
carbon disulfide from air to blood and from blood to organs were 2.8
and about 100, respectively. This explains the rapid disappearance of
carbon disulfide from the blood (section 4.2.2).
The solubility in lipids and fats, and binding to amino acids and
proteins, explains the affinity of carbon disulfide for all tissues
and organs. However, at the beginning of absorption, some initial
preference for some organs seems to exist. Animal experiments have
given various results concerning the order of affinity for different
organs, but these may be explained by interspecies differences, by
differences in the mode of administration or both. These aspects have
been studied in animals only, since even postmortem studies in man are
impracticable because of the rapid elimination of carbon disulfide.
McKee (1941) first performed such experiments and results obtained up
to 1954 have been reviewed by Teisinger (1954). In studies on
guineapigs by Strittmatter et al. (1950) using labelled carbon
disulfide, initial accumulation occurred in the liver followed by
uniform distribution in the organism after some days. The following
order of initial prevalence of carbon disulfide in rats was
established by Merlevede (1951): liver, bile, kidneys, heart,
adrenals, brain. Teisinger (1954) found the largest amount of carbon
disulfide in the brain of guinea-pigs and Madlo & Soucek (1953)
demonstrated its presence in the peripheral nerves of rats.
In studies on the distribution of carbon disulfide labelled with
35S, radioactivity was retained in the brain for 2 days (Bussing et
al., 1953; Büssing & Sonnenschein, 1954).
Bartonicek (1957) found that "total" carbon disulfide accumulated
initially in the adrenals, blood, and brain of exposed rats. At the
same time, he observed the existence of both "free" and "bound" forms
in the body. "Free" carbon disulfide denotes the fraction of carbon
disulfide dissolved in body fluids and "bound" carbon disulfide, the
fraction that has reacted with amino acids to give thiocarbamates, a
reaction that is reversible. This form is acid labile. It has been
shown by De Matteis & Seawright (1973) that the sulfur released during
the process of desulfuration of carbon disulfide can form covalent
bonds with other sulfur radicals. By determining "free" and "bound"
carbon disulfide separately, Bartonicek (1957) obtained another order
of initial accumulation. "Free" carbon disulfide disappeared quite
quickly from the organs following an exponential curve and reached
very low values, 10-16 h after the termination of exposure, while the
"bound" form decreased irregularly. Thus, according to Bartonicek
(1958, 1959), "free" carbon disulfide accumulates in the liver,
muscles, spleen, blood, lungs, brain, kidneys, and heart while "bound"
carbon disulfide accumulates in the blood, spleen, liver, lungs,
heart, muscles, kidneys, and brain. Gradually more uniform
distribution takes place. The existence of 2 forms with quite
different initial affinities for blood and organs, could explain the
controversial results obtained earlier and the poor correlation
between carbon disulfide concentrations in the blood and air (section
4.2.3).
4.2.5 Binding in blood and tissues
According to Teisinger (1954), in 1910, Siegfried & Weidenhaupt
proved by in vitro experiments that carbon disulfide was bound to
glycine in blood in alkaline medium, producing glycine-dithiocarbamic
acid characterized by free -SH groups. These authors stated that
similar reactions took place with phenylalanine, sarcosine, and
asparagine. Chromatographic and spectrophotometric studies have shown
that amino acids of the blood plasma react with carbon disulfide to
form dithiocarbamic acid and a cyclic compound of the thiazolinone
type (Soucek & Madlo, 1953; Madlo, 1953; Yoshida, 1955; Cohen et al.,
1959). Bobsien (1954) demonstrated the binding of carbon disulfide to
euglobulin and albumin through -SH groups; he found that binding to
pseudoglobulin was negligible. The binding of carbon disulfide to
cysteine, methionine, and glutathione in the blood was established by
Büssing (1952), but the nature of the binding was not stated.
Using human blood, Soucek & Madlo (1953) established in vitro,
that carbon disulfide was bound to amino acids in the blood by a
first-order reaction, the half-time of which was 6.5 h. Various acids
and formaldehyde blocked this reaction, producing dithiocarbamic acid
and thiazolinone. In further in vitro studies, the same authors
(Soucek & Madlo, 1954, 1955, 1956) found that, at pH 7.3-8.3 and at a
temperature of 37°C, carbon disulfide was quantitatively bound to
albumin but not to gammaglobulin. The product formed possessed free-SH
groups that could be determined by titration with iodine chloride. The
product was very stable, not hydrolysing even at 100° C. On the other
hand, the product formed after the binding of carbon disulfide to
amino acid did not show such stability. Soucek (1957) showed that the
same processes took place in vivo.
The binding of carbon disulfide to proteolytic enzymes (trypsin,
pepsin, chymotrypsin) forming a labile compound similar to
dithiocarbamic acid was also reported by Soucek et al. (1957) and
Soucek (1959). Soucek & Madlo (1955) assumed that the formation of
dithiocarbamic acid took place in the blood and the liver, and that
the compound formed then appeared in the liver, adipose tissues,
blood, and, in small quantities, in the brain and muscles.
4.3 Elimination of Carbon Disulfide and Metabolites
4.3.1 Elimination by breath, saliva, sweat, and faeces
Some basic data on the exhalation of absorbed carbon disulfide
have already been discussed (section 4.2.1). The process takes place
in 3 phases. In the first phase, there is rapid elimination of the
carbon disulfide absorbed on the mucosa of the lungs and upper part of
the tract. In a second slower phase, exhalation of carbon disulfide
released from the blood occurs. In the third, very slow phase, carbon
disulfide released from tissues and organs is exhaled. Each phase can
be presented as a separate curve with a different angle (Soucek &
Pavelkova, 1953). In experiments on animals, De Matteis & Seawright
(1973) established that a significant part of the carbon, released
from the carbon disulfide by a desulfuration process, was exhaled as
carbon dioxide.
It was reported by Merlevede (1951) that small quantities of
carbon disulfide were excreted in the saliva and sweat. Harashima &
Masuda (1962) demonstrated the excretion of "free" carbon disulfide
through the skin of exposed workers, stating that, sometimes, the
amounts excreted by this route were as much as 3 times higher than the
amounts of unmetabolized carbon disulfide excreted in the urine.
It is generally accepted that the elimination of inhaled carbon
disulfide in the faeces is negligible.
4.3.2 Excretion of carbon disulfide and metabolites in urine
Less than 1% of absorbed carbon disulfide is excreted unchanged
in the urine but about 70-90% of retained carbon disulfide is
metabolized and excreted in the urine in the form of various
metabolites (Soucek, 1957).
A number of experimental studies on rats, dogs, and guineapigs
have shown that carbon disulfide is excreted in the form of inorganic
sulfates into the urine (Billet & Bourlier, 1944; Strittmatter et al.,
1950). Using labelled carbon disulfide, Strittmatter et al. (1950)
showed that, in guinea-pigs, 30% of intravenously injected carbon
disulfide was metabolized to form such end-products. Jakubowski (1968,
1971) isolated 3 metabolites, and Kopecky (1973) identified
2-mercapto-thiazoline-4-carbonyl acid in the urine of exposed rats.
In contrast with the results obtained in animal studies,
Merlevede (1951) did not observe any increase in the total sulfate
concentration in the urine of exposed workers and registered only a
relative increase in the ethereal fraction. This was corroborated by
Delic et al. (1966) and Djerassi & Lambroso (1968). Delic et al.
(1966) studied the urinary excretion of sulfates in 111 workers, 52 of
whom were exposed to high concentrations of carbon disulfide, i.e.,
100-1000 mg/m3, 36 to concentrations below 150 mg/m3 and 23 to
concentrations below 30 mg/m3. In the most heavily exposed group,
15.5% of the workers showed an increased excretion of total sulfates
(3.8-6 g/litre). On the other hand, an equally high percentage (15%)
of workers exposed to levels below 30 mg/m3 displayed a similar
increase (3-3.2 g/litre); 5.6% of the workers exposed to
concentrations below 150 mg/m3 also showed an increased excretion.
Thus, there was no correlation between excretion of toted sulfates and
exposure. A relative increase in the ethereal (organic) fraction of
sulfates that was evident in 60% of all the workers was also unrelated
to exposure level. The results appeared to suggest that a conjugation
process of some carbon disulfide metabolites took place rather than an
oxidation to inorganic sulfates.
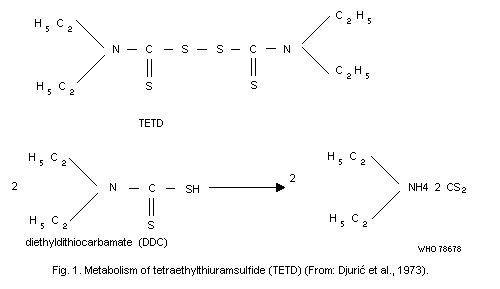 The responsibility of metabolites for the discoloration of iodine
azide remained hypothetical until Pergal et al. (1972a,b) isolated 3
metabolites from human urine and identified 2 of them as thiourea and
mercaptothiazolinone; thiourea is by far the most important of these
metabolites. Later, Pergal et al. (1977a) developed a quantitative
method for the micro-determination of thiourea in the urine of exposed
workers or of alcoholics treated with tetraethylthiuramdisulfide
(TETD, Disulfiram, Antabuse). The authors suggested that the third
metabolite was 2-mercapto-thiazoline-4-carbamic acid (Pergal et al.,
1977b). Tetraethylthiuramdisulfide is metabolized in a way that
liberates carbon disulfide (Fig. 1). Consequently, alcoholics treated
with this agent are exposed to carbon disulfide and its metabolites.
Skalicka (1967) and Novak et al. (1968) measured the iodine-azide
reaction and determined diethyldithiocarbamates (DDC) in the urine of
alcoholics treated with TETD. These results led Djuric et al. (1973)
to use TETD as a test for the evaluation of the metabolic rate of
sulfur compounds in the organism of workers, the so-called "antabuse
test".
Studies on the microsomal metabolism of carbon disulfide in the
liver of rats revealed that it was desulfurated to form
carbonylsulfide and that this was further oxidized, yielding carbon
dioxide which was exhaled (De Matteis & Seawright, 1973; De Matteis,
1974; Dalvi et al., 1974).
Data from human and animal studies on ethereal sulfate excretion
(Magos, 1973) have shown that bivalent sulfur represents a small part
of retained carbon disulfide, probably less than 5%. The major pathway
leads to the formation of sulfates that are excreted in urine.
5. BIOCHEMICAL EFFECTS OF CARBON DISULFIDE
From the chemical point of view, carbon disulfide is highly
reactive with nucleophilic reagents characterized by the presence of a
group with a free pair of electrons in the molecule. The most
important nucleophilic groups are mercapto (-SH), amino (-NH2) and
hydroxy (-OH) groups (Vasak & Kopecky 1967). However, physiological pH
values do not favour these reactions (Kopecky, 1977, private
communication).
According to the chemical structure of compounds participating in
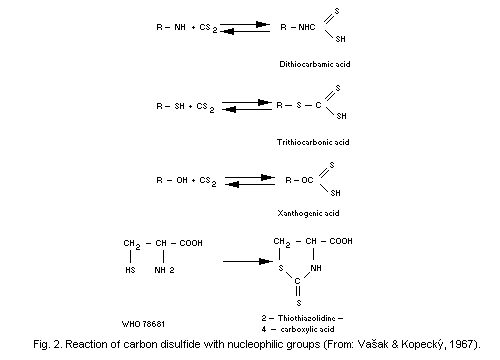
the reactions, carbon disulfide will produce dithiocarbamic,
trithiocarbonic, or xanthogenic acid. If carbon disulfide reacts with
an organic compound with 2 nucleophilic groups, a cyclic compound of
the thiazolinone type is formed (see Fig. 2).
The majority of biochemically important compounds, such as amino
acids, biogenic amines, and sugars, contain these nucleophilic groups
and, thus, may react with carbon disulfide. This is true of a large
number of substances existing in the organism.
A number of possible mechanisms of the effects of carbon
disulfide on the organism have been postulated including:
(a) the chelating effect of carbon disulfide metabolites on
various metals, essential for the functioning of enzymes;
(b) the effect of carbon disulfide on enzymatic systems;
(c) disturbances of vitamin metabolism;
(d) impairment of catecholamine metabolism;
(e) changes in lipid metabolism;
(f) interaction with microsomal drug-metabolizing enzyme
systems.
The responsibility of metabolites for the discoloration of iodine
azide remained hypothetical until Pergal et al. (1972a,b) isolated 3
metabolites from human urine and identified 2 of them as thiourea and
mercaptothiazolinone; thiourea is by far the most important of these
metabolites. Later, Pergal et al. (1977a) developed a quantitative
method for the micro-determination of thiourea in the urine of exposed
workers or of alcoholics treated with tetraethylthiuramdisulfide
(TETD, Disulfiram, Antabuse). The authors suggested that the third
metabolite was 2-mercapto-thiazoline-4-carbamic acid (Pergal et al.,
1977b). Tetraethylthiuramdisulfide is metabolized in a way that
liberates carbon disulfide (Fig. 1). Consequently, alcoholics treated
with this agent are exposed to carbon disulfide and its metabolites.
Skalicka (1967) and Novak et al. (1968) measured the iodine-azide
reaction and determined diethyldithiocarbamates (DDC) in the urine of
alcoholics treated with TETD. These results led Djuric et al. (1973)
to use TETD as a test for the evaluation of the metabolic rate of
sulfur compounds in the organism of workers, the so-called "antabuse
test".
Studies on the microsomal metabolism of carbon disulfide in the
liver of rats revealed that it was desulfurated to form
carbonylsulfide and that this was further oxidized, yielding carbon
dioxide which was exhaled (De Matteis & Seawright, 1973; De Matteis,
1974; Dalvi et al., 1974).
Data from human and animal studies on ethereal sulfate excretion
(Magos, 1973) have shown that bivalent sulfur represents a small part
of retained carbon disulfide, probably less than 5%. The major pathway
leads to the formation of sulfates that are excreted in urine.
5. BIOCHEMICAL EFFECTS OF CARBON DISULFIDE
From the chemical point of view, carbon disulfide is highly
reactive with nucleophilic reagents characterized by the presence of a
group with a free pair of electrons in the molecule. The most
important nucleophilic groups are mercapto (-SH), amino (-NH2) and
hydroxy (-OH) groups (Vasak & Kopecky 1967). However, physiological pH
values do not favour these reactions (Kopecky, 1977, private
communication).
According to the chemical structure of compounds participating in
the reactions, carbon disulfide will produce dithiocarbamic,
trithiocarbonic, or xanthogenic acid. If carbon disulfide reacts with
an organic compound with 2 nucleophilic groups, a cyclic compound of
the thiazolinone type is formed (see Fig. 2).
The majority of biochemically important compounds, such as amino
acids, biogenic amines, and sugars, contain these nucleophilic groups
and, thus, may react with carbon disulfide. This is true of a large
number of substances existing in the organism.
A number of possible mechanisms of the effects of carbon
disulfide on the organism have been postulated including:
(a) the chelating effect of carbon disulfide metabolites on
various metals, essential for the functioning of enzymes;
(b) the effect of carbon disulfide on enzymatic systems;
(c) disturbances of vitamin metabolism;
(d) impairment of catecholamine metabolism;
(e) changes in lipid metabolism;
(f) interaction with microsomal drug-metabolizing enzyme
systems.
 5.1 Chelating Effects of Carbon Disulfide Metabolites
The hypothesis of the chelating effect of carbon disulfide
metabolites was advanced by Cohen and coworkers and was based on
experiments on rabbits (Cohen et al., 1958; Paulus et al., 1957;
Scheel et al., 1960; Scheel, 1965, 1967). Considerable shifts were
found in the copper and zinc contents of various tissues, especially
in the nervous tissue, in rabbits poisoned by carbon disulfide. The
concentration of copper in the brain and spinal cord of animals killed
2 weeks after final exposure was less than half of that in the
controls. On the other hand, the zinc level in exposed rabbits was 20%
higher than that in the control animals. In general, pathological
examination of the tissues did not indicate any changes, except in the
kidneys and in the spinal cord, which showed marked degeneration of
the axis of the cylinder. The Purkinje cells of the cerebrum also
showed signs of degeneration.
The following hypothesis, based on an observation that the levels
of metal ions in tissues were altered by exposure to carbon disulfide,
was formulated by Scheel (1967):
-- carbon disulfide reacts with the amino groups of amino acids
and proteins to form thiocarbamate in blood and tissues, as was stated
by Soucek & Madlo (1956);
-- thiocarbamates, possessing sulfhydryl groups, may chelate
polyvalent inorganic ions. Because of the low dissociation of the
product, they would, thus, interfere with cellular metabolism.
-- when such interference becomes sufficiently limiting, the body
would respond by oxidizing fat and general loss in body-weight would
occur;
-- ultimately, as the metabolic limitation increases, cellular
death and loss of associated function would occur, producing signs of
tissue injury.
Since the entire hypothesis rests on chelation of metal ions, it
should be possible to prevent the occurrence of such an effect by
supplying an excess of metal ions in the diet of animals exposed to
carbon disulfide (Scheel et al., 1960; Scheel, 1967). Such a
protective effect is claimed to have been achieved by Scheel (1967).
The hypothesis of a chelating effect has been supported by the
results of other studies including those of Andreeva (1970), who
reported an increase in zinc and copper excretion in exposed rats, and
Lukas et al. (1974), who found increased copper levels in the
peripheral nervous tissue of exposed rats. A decreased level of
ceruloplasmin in rats with experimental carbon disulfide
polyneuropathy was reported by Lukas et al. (1975). This decrease was
related to the intensity and extent of the electromyographic signs of
polyneuropathy. Gadaskina & Andreeva (1969) and Cimbarevic (1970)
noticed a decrease in ceruloplasmin activity in workers exposed to
carbon disulfide for more than 10 years. However, in other studies,
the ceruloplasmin levels in exposed workers were in the normal range
(Andruszczak, 1967; Kujalova, 1973). Andruszczak (1967) found
increased ceruloplasmin levels in patients suffering from chronic
carbon disulfide poisoning.
Increased excretion of trace metals in the urine of workers
exposed to carbon disulfide was not observed in studies by Djuric et
al. (1967). Hernberg & Nordman (1969), and Hernberg et al. (1969).
However, these negative results do not necessarily exclude a chelating
effect, since exposure may have been too low. Thus, the more recent
results of El Gazzar et al. (1973) showing a temporary increase in the
zinc contents of all serum protein fractions as well as in urinary
excretion may reflect the effects of a higher exposure level than in
the previous studies.
It is known that copper and zinc ions are essential for the
prosthetic groups of many enzymes. The neurotoxic action of carbon
disulfide and its interference with the activity of many enzymes could
easily be explained by chelating effects. Zinc is required for the
activity of enzymes such as lactic acid dehydrogenase (EC 1.1.1.27)a,
carbonic anhydrase (EC 4.2.1.1), glutamate dehydrogenase (EC 1.4.1.2),
and alcohol dehydrogenase (EC 1.1.1.1). Copper, on the other hand,
represents a cofactor of pyridoxol, a form of vitamin B6.
Copper is required for the proper functioning of enzymes such as
cytochrome c oxidase (EC 1.9.3.1), the coenzyme A dehydrogenase
system, and dopamine ß hydroxylase (EC 1.14.17.1). The loss of copper
from the spinal cord is accompanied by cellular damage, producing
tissue degeneration. Disturbances of the central and peripheral
nervous systems, resulting from carbon disulfide exposure, could be
connected with the loss of copper due to chelation and consequent
inhibitory effects on enzyme systems (Scheel, 1967).
a The numbers within parentheses following the names of enzymes are
those assigned by the Enzyme Commission of the Joint IUPAC-IUB
Commission on Biochemical Nomenclature.
5.2 Effects on Enzyme Systems
Inhibition of monoamine-oxidase (EC 1.4.3.4) (MAO) activity
occurs as soon as exposure of an animal to carbon disulfide begins,
but it is reversible (Magistretti & Peirone, 1961; Lazarev et al.,
1965). The mechanism of inhibition is not yet clear, but it is known
that MAO contains a copper pyridoxal complex. Vasak & Kopecky (1967)
found a decrease in catecholamine in the urine of exposed rats. This
result suggests the possibility that carbon disulfide forms a compound
with catecholamine which cannot be split by MAO. However, Magos &
Jarvis (1970b), who also exposed rats to carbon disulfide, did not
find any inhibition of MAO. They suggest that Vasak & Kopecky's (1967)
finding could be explained by the inhibition of dopamine ß
hydroxylase.
Alkaline phosphatase (EC 3.1.3.1) activity was inhibited in the
tissues and serum of rabbits exposed for more than 22 weeks to high
concentrations of carbon disulfide, i.e., concentrations up to about
2350 mg/m3 (750 ppm) (Cohen et al., 1959). Chervenka & Wilcox (1956)
did not find any influence of carbon disulfide on derivatives of
chymotrypsinogen or on succinate dehydrogenase (EC 1.3.99.1) activity
and Minden et al. (1967) did not register any effects on glycolytic
enzymes, Kreb's cycle enzymes, and transaminases in experimental
animals.
No changes in glycolysis were found in the brain tissue of rats
after either acute or chronic exposure to carbon disulfide (Tarkowski
& Cremer, 1972; Tarkowski, 1973). Changes in the brain free amino acid
metabolism observed in rats exposed to a carbon disulfide
concentration of 2400 mg/m3 for 15 h included reductions in the
levels of glutamic delta-amino butyric acids. These effects were
accompanied by decreased activity of brain glutamate decarboxylase
(EC 4.1.1.15) (Tarkowski, 1974).
Both, acute and chronic exposures of animals to carbon disulfide
result in changes in mitochondrial respiration and oxidative
phosphorylation. Respiration of the brain mitochondria was partly
inhibited in rats exposed to carbon disulfide (Tarkowski & Sobczak,
1971); cytochrome oxidase activity was also inhibited (Tarkowski,
Wronska-Nofer, 1966). Oxidative phosphorylation in the mitochondria
was partly inhibited and partly uncoupled, and was accompanied by a
reduction in the activity of adenosinetriphosphatase (EC 3.6.1.3)
(Tarkowski & Sobczak, 1971).
Gregorczyk et al. (1975a,b) did not find any changes in liver
enzymes, proteins, and free amino acids in rats exposed to a carbon
disulfide concentration of 1300 mg/m3 for 12-26 weeks, and 5 and 10 h
daily. The authors concluded that carbon disulfide was not hepatotoxic
under such exposure conditions; this seems to be supported by the fact
that they did not find any changes in the blood serum enzymes
(Gregorczyk et al. 1975a,b).
Decreased activity of triosephosphate dehydrogenase (EC 1.2.1.9),
lactate dehydrogenase, and glycerophosphate dehydrogenase found in the
muscles of rats with developed neuropathy was accompanied by an
increase in hexokinase (EC 2.7.1.1) activity (Lukas et al., 1977).
Further studies should establish which enzymes are inhibited by
exposure of the organism to carbon disulfide, at what levels of
exposure, and the inhibition of which enzyme systems would present a
significant health hazard.
5.3 Effects on Vitamin Metabolism
5.3.1 Vitamin B6
There are 3 forms of vitamin B6, i.e., pyridoxol, pyridoxal, and
pyridoxamine, that play the role of coenzymes in various enzyme
systems. These 3 forms are in equilibrium because they are
enzymatically converted into each other (Fig. 3). The binding of one
form of vitamin B6 will block the reactions of the enzymes containing
the remaining forms. Vasak & Kopecky (1967) reported that carbon
disulfide reacted in vitro with pyridoxamine to form a salt of
pyridoxamine dithiocarbamic acid. Some authors think that this process
could also occur in vivo, causing inhibition of the enzyme systems
in which vitamin B6 is involved as a coenzyme. Disturbance of
pyridoxol metabolism during chronic intoxication was shown in
experiments on rats by Kujalova (1971). After the tryptophan load
test, excretion of xanthurenic acid increased in exposed animals while
excretion of pyridoxol acid decreased.
The temporal development and the severity of this disturbance
depended on the diet given to the animals in question (Kujalova, 1971;
Gorny, 1974). Pyridoxol deficiency due to carbon disulfide
intoxication could be eliminated in the animals by giving a diet rich
in pyridoxol (10 times the normal dietary level). However, this diet
did not prevent the development of neuropathy (Lukas, 1970) or
influence the deterioration in motor activity in rats (Frantik, 1970;
Teisinger, 1971).
5.1 Chelating Effects of Carbon Disulfide Metabolites
The hypothesis of the chelating effect of carbon disulfide
metabolites was advanced by Cohen and coworkers and was based on
experiments on rabbits (Cohen et al., 1958; Paulus et al., 1957;
Scheel et al., 1960; Scheel, 1965, 1967). Considerable shifts were
found in the copper and zinc contents of various tissues, especially
in the nervous tissue, in rabbits poisoned by carbon disulfide. The
concentration of copper in the brain and spinal cord of animals killed
2 weeks after final exposure was less than half of that in the
controls. On the other hand, the zinc level in exposed rabbits was 20%
higher than that in the control animals. In general, pathological
examination of the tissues did not indicate any changes, except in the
kidneys and in the spinal cord, which showed marked degeneration of
the axis of the cylinder. The Purkinje cells of the cerebrum also
showed signs of degeneration.
The following hypothesis, based on an observation that the levels
of metal ions in tissues were altered by exposure to carbon disulfide,
was formulated by Scheel (1967):
-- carbon disulfide reacts with the amino groups of amino acids
and proteins to form thiocarbamate in blood and tissues, as was stated
by Soucek & Madlo (1956);
-- thiocarbamates, possessing sulfhydryl groups, may chelate
polyvalent inorganic ions. Because of the low dissociation of the
product, they would, thus, interfere with cellular metabolism.
-- when such interference becomes sufficiently limiting, the body
would respond by oxidizing fat and general loss in body-weight would
occur;
-- ultimately, as the metabolic limitation increases, cellular
death and loss of associated function would occur, producing signs of
tissue injury.
Since the entire hypothesis rests on chelation of metal ions, it
should be possible to prevent the occurrence of such an effect by
supplying an excess of metal ions in the diet of animals exposed to
carbon disulfide (Scheel et al., 1960; Scheel, 1967). Such a
protective effect is claimed to have been achieved by Scheel (1967).
The hypothesis of a chelating effect has been supported by the
results of other studies including those of Andreeva (1970), who
reported an increase in zinc and copper excretion in exposed rats, and
Lukas et al. (1974), who found increased copper levels in the
peripheral nervous tissue of exposed rats. A decreased level of
ceruloplasmin in rats with experimental carbon disulfide
polyneuropathy was reported by Lukas et al. (1975). This decrease was
related to the intensity and extent of the electromyographic signs of
polyneuropathy. Gadaskina & Andreeva (1969) and Cimbarevic (1970)
noticed a decrease in ceruloplasmin activity in workers exposed to
carbon disulfide for more than 10 years. However, in other studies,
the ceruloplasmin levels in exposed workers were in the normal range
(Andruszczak, 1967; Kujalova, 1973). Andruszczak (1967) found
increased ceruloplasmin levels in patients suffering from chronic
carbon disulfide poisoning.
Increased excretion of trace metals in the urine of workers
exposed to carbon disulfide was not observed in studies by Djuric et
al. (1967). Hernberg & Nordman (1969), and Hernberg et al. (1969).
However, these negative results do not necessarily exclude a chelating
effect, since exposure may have been too low. Thus, the more recent
results of El Gazzar et al. (1973) showing a temporary increase in the
zinc contents of all serum protein fractions as well as in urinary
excretion may reflect the effects of a higher exposure level than in
the previous studies.
It is known that copper and zinc ions are essential for the
prosthetic groups of many enzymes. The neurotoxic action of carbon
disulfide and its interference with the activity of many enzymes could
easily be explained by chelating effects. Zinc is required for the
activity of enzymes such as lactic acid dehydrogenase (EC 1.1.1.27)a,
carbonic anhydrase (EC 4.2.1.1), glutamate dehydrogenase (EC 1.4.1.2),
and alcohol dehydrogenase (EC 1.1.1.1). Copper, on the other hand,
represents a cofactor of pyridoxol, a form of vitamin B6.
Copper is required for the proper functioning of enzymes such as
cytochrome c oxidase (EC 1.9.3.1), the coenzyme A dehydrogenase
system, and dopamine ß hydroxylase (EC 1.14.17.1). The loss of copper
from the spinal cord is accompanied by cellular damage, producing
tissue degeneration. Disturbances of the central and peripheral
nervous systems, resulting from carbon disulfide exposure, could be
connected with the loss of copper due to chelation and consequent
inhibitory effects on enzyme systems (Scheel, 1967).
a The numbers within parentheses following the names of enzymes are
those assigned by the Enzyme Commission of the Joint IUPAC-IUB
Commission on Biochemical Nomenclature.
5.2 Effects on Enzyme Systems
Inhibition of monoamine-oxidase (EC 1.4.3.4) (MAO) activity
occurs as soon as exposure of an animal to carbon disulfide begins,
but it is reversible (Magistretti & Peirone, 1961; Lazarev et al.,
1965). The mechanism of inhibition is not yet clear, but it is known
that MAO contains a copper pyridoxal complex. Vasak & Kopecky (1967)
found a decrease in catecholamine in the urine of exposed rats. This
result suggests the possibility that carbon disulfide forms a compound
with catecholamine which cannot be split by MAO. However, Magos &
Jarvis (1970b), who also exposed rats to carbon disulfide, did not
find any inhibition of MAO. They suggest that Vasak & Kopecky's (1967)
finding could be explained by the inhibition of dopamine ß
hydroxylase.
Alkaline phosphatase (EC 3.1.3.1) activity was inhibited in the
tissues and serum of rabbits exposed for more than 22 weeks to high
concentrations of carbon disulfide, i.e., concentrations up to about
2350 mg/m3 (750 ppm) (Cohen et al., 1959). Chervenka & Wilcox (1956)
did not find any influence of carbon disulfide on derivatives of
chymotrypsinogen or on succinate dehydrogenase (EC 1.3.99.1) activity
and Minden et al. (1967) did not register any effects on glycolytic
enzymes, Kreb's cycle enzymes, and transaminases in experimental
animals.
No changes in glycolysis were found in the brain tissue of rats
after either acute or chronic exposure to carbon disulfide (Tarkowski
& Cremer, 1972; Tarkowski, 1973). Changes in the brain free amino acid
metabolism observed in rats exposed to a carbon disulfide
concentration of 2400 mg/m3 for 15 h included reductions in the
levels of glutamic delta-amino butyric acids. These effects were
accompanied by decreased activity of brain glutamate decarboxylase
(EC 4.1.1.15) (Tarkowski, 1974).
Both, acute and chronic exposures of animals to carbon disulfide
result in changes in mitochondrial respiration and oxidative
phosphorylation. Respiration of the brain mitochondria was partly
inhibited in rats exposed to carbon disulfide (Tarkowski & Sobczak,
1971); cytochrome oxidase activity was also inhibited (Tarkowski,
Wronska-Nofer, 1966). Oxidative phosphorylation in the mitochondria
was partly inhibited and partly uncoupled, and was accompanied by a
reduction in the activity of adenosinetriphosphatase (EC 3.6.1.3)
(Tarkowski & Sobczak, 1971).
Gregorczyk et al. (1975a,b) did not find any changes in liver
enzymes, proteins, and free amino acids in rats exposed to a carbon
disulfide concentration of 1300 mg/m3 for 12-26 weeks, and 5 and 10 h
daily. The authors concluded that carbon disulfide was not hepatotoxic
under such exposure conditions; this seems to be supported by the fact
that they did not find any changes in the blood serum enzymes
(Gregorczyk et al. 1975a,b).
Decreased activity of triosephosphate dehydrogenase (EC 1.2.1.9),
lactate dehydrogenase, and glycerophosphate dehydrogenase found in the
muscles of rats with developed neuropathy was accompanied by an
increase in hexokinase (EC 2.7.1.1) activity (Lukas et al., 1977).
Further studies should establish which enzymes are inhibited by
exposure of the organism to carbon disulfide, at what levels of
exposure, and the inhibition of which enzyme systems would present a
significant health hazard.
5.3 Effects on Vitamin Metabolism
5.3.1 Vitamin B6
There are 3 forms of vitamin B6, i.e., pyridoxol, pyridoxal, and
pyridoxamine, that play the role of coenzymes in various enzyme
systems. These 3 forms are in equilibrium because they are
enzymatically converted into each other (Fig. 3). The binding of one
form of vitamin B6 will block the reactions of the enzymes containing
the remaining forms. Vasak & Kopecky (1967) reported that carbon
disulfide reacted in vitro with pyridoxamine to form a salt of
pyridoxamine dithiocarbamic acid. Some authors think that this process
could also occur in vivo, causing inhibition of the enzyme systems
in which vitamin B6 is involved as a coenzyme. Disturbance of
pyridoxol metabolism during chronic intoxication was shown in
experiments on rats by Kujalova (1971). After the tryptophan load
test, excretion of xanthurenic acid increased in exposed animals while
excretion of pyridoxol acid decreased.
The temporal development and the severity of this disturbance
depended on the diet given to the animals in question (Kujalova, 1971;
Gorny, 1974). Pyridoxol deficiency due to carbon disulfide
intoxication could be eliminated in the animals by giving a diet rich
in pyridoxol (10 times the normal dietary level). However, this diet
did not prevent the development of neuropathy (Lukas, 1970) or
influence the deterioration in motor activity in rats (Frantik, 1970;
Teisinger, 1971).
 That pyridoxol deficiency might result from carbon disulfide
intoxication was proved by Gorny (1971), who showed that the pyridoxal
phosphate concentration decreased in the serum of acutely intoxicated
rats. Furthermore, increases have been reported in nicotinamides, the
metabolites of nicotinic acid (Nofer & Wrofiska-Nofer, 1966) and in
hydroxyindolacetic acid (a metabolite of serotonine) (Abuczewicz et
al., 1971) in the urine of rats exposed to carbon disulfide. All these
substances are derived from tryptophan.
In a review on the mechanisms of chronic carbon disulfide
poisoning, Teisinger (1971) concluded that disturbance of vitamin B6
metabolism was obvious but that it did not play a major role in the
pathogenesis of nervous tissue lesions.
Transaminases are sensitive to vitamin B6 deficiency. Thus,
metabolic pathways in which transaminases are involved may be
inhibited. This is the case in tryptophan metabolism, where it is
manifested by increased excretion of xanthurenic acid in both man
(Tintera et al., 1972) and rat (Fig. 4) (Abramova, 1967).
That pyridoxol deficiency might result from carbon disulfide
intoxication was proved by Gorny (1971), who showed that the pyridoxal
phosphate concentration decreased in the serum of acutely intoxicated
rats. Furthermore, increases have been reported in nicotinamides, the
metabolites of nicotinic acid (Nofer & Wrofiska-Nofer, 1966) and in
hydroxyindolacetic acid (a metabolite of serotonine) (Abuczewicz et
al., 1971) in the urine of rats exposed to carbon disulfide. All these
substances are derived from tryptophan.
In a review on the mechanisms of chronic carbon disulfide
poisoning, Teisinger (1971) concluded that disturbance of vitamin B6
metabolism was obvious but that it did not play a major role in the
pathogenesis of nervous tissue lesions.
Transaminases are sensitive to vitamin B6 deficiency. Thus,
metabolic pathways in which transaminases are involved may be
inhibited. This is the case in tryptophan metabolism, where it is
manifested by increased excretion of xanthurenic acid in both man
(Tintera et al., 1972) and rat (Fig. 4) (Abramova, 1967).
 5.3.2 Nicotinic acid
Exposure to carbon disulfide resulted in increased excretion of
methyl nicotinamide, a nicotinic acid metabolite, in the urine of rats
(Liniecki, 1960; Wrofiska-Nofer et al., 1965). An increase in serum
lipids during the exposure of rabbits and rats to carbon disulfide was
prevented by administration of nicotinic acid (Nofer & Wrofiska-Nofer,
1966; Wrofiska-Nofer, 1970). The biochemical background of this
phenomenon is still obscure, but a hypothesis based on the
interference of carbon disulfide with pyridine nucleotide and
nicotinamide metabolism has been advanced (Nofer & Wrofiska-Nofer,
1966). However, feeding nicotinic acid to the exposed animals did not
prevent the development of neuropathies in these experiments.
As the increase in urinary excretion of nicotinamide metabolites
did not occur at the cost of the systematic pool of
nicotinamide-adenine dinucleotides, Wrofiska-Nofer et al. (1970)
suggested that it reflected an increase in the whole turnover rate.
The mechanism of the process was not clarified but it may be assumed
that an increase in the synthesis via tryptophan occurs.
5.4 Effects on Catecholamine Metabolism
Disturbance of the catecholamine metabolism can play a part in
many pathological processes. In studies on rats acutely intoxicated
with carbon disulfide, significant changes were found in the brain
catecholamine metabolism (Magos & Jarvis, 1970b; Magos, 1975). There
was a decrease in the level of noradrenaline accompanied by an
increased concentration of dopamine. Inhibition of dopamine
ß-hydroxylase, an essential enzyme in catecholamine metabolism, was
demonstrated in vivo in studies on rats by Magos (1975), and in
vitro by McKenna & Di Stefano (1975). Magos (1975) advanced a theory
concerning the central role played by catecholamine disturbances in
carbon disulfide pathology, especially where central nervous system
changes were involved, and in cardiovascular pathology. Cavalleri et
al. (1977) suggested that this could also explain the involvement of
the endocrine system.
5.5 Effects on Lipid Metabolism
Disturbance in the lipid metabolism has long since been linked
with carbon disulfide exposure. Increased levels of serum lipids, free
and total cholesterol, and ß-lipoproteins have been reported in
rabbits exposed to carbon disulfide (Paterni et al., 1958; Cohen et
al., 1959; Prerovska et al., 1961). Harashima et al. (1960) reported
elevated total and esterified cholesterol levels in the serum of
heavily exposed workers, whereas workers exposed to levels of about
15-60 mg/m3 (5-19 ppm) displayed normal serum cholesterol
concentrations. Elevated cholesterol levels were also found in exposed
workers by Manu et al. (1971) and in patients with previous exposure
to carbon disulfide (the patients had not been exposed for several
years) by Graovac-Leposavic et al. (1977). Higher levels of serum
lipids and especially cholesterol may be due to an increased rate of
synthesis in the liver and to the inhibition of the degradation of
lipids (Wrofiska-Nofer, 1969; Laurman & Wronska-Nofer, 1977). The fact
that elevated cholesterol levels have not been found consistently may
be explained by the different exposure levels in different studies
(Toyoma & Sakurai, 1967). Bittersohl & Thiele (1977) found that a
higher frequency of cholesterol values exceeding 6.72 mmol/litre
(260 mg/dl) in workers exposed to carbon disulfide was strongly
correlated with the duration of exposure.
A decreased clearing factor activity was found by Ruikka (1959)
in workers exposed to carbon disulfide and Martino et al. (1963, 1964)
reported an elevated ß-lipoprotein fraction in the serum of exposed
workers.
Changes in the lipid metabolism found in the aorta tissue may
contribute to the development of atheromatic changes in blood vessels
(Wrofiska-Nofer, 1976).
An increase in serum lipids during the exposure of rabbits to
carbon disulfide was prevented by administration of nicotinic acid
(Nofer & Wrofiska-Nofer, 1966) and in rats (Wrofiska-Nofer, 1970)
(section 5.3.2).
5.6 Interaction with Microsomal Drug Metabolism
An important feature of the liver toxicity caused by carbon
disulfide seems to be the destruction of cytochrome P-450 (Bond & De
Matteis, 1969). There is evidence that this effect is due to the
oxidative desulfuration of carbon disulfide by mixed-function oxidases
(De Matteis & Seawright, 1973). The resulting, highly reactive, sulfur
becomes covalently bound to the microsomal protein (Dalvi et al.,
1974; De Matteis, 1974; Jarvisalo et al., 1977), mainly to the
apoprotein of cytochrome P-450 (Neal et al., 1976; Jarvisalo & De
Matteis, 1977; Savolainen et al., 1977a). It is possible that the
liberated sulfur is the real toxic agent in liver toxicity arising
from carbon disulfide exposure.
Experiments on rats have shown that exposure to carbon disulfide
in concentrations up to 1250 mg/m3 (400 ppm) for 8 h is followed by
an increase in microsomal RNA content and in total protein in the
hepatic microsomal fraction and also increased incorporation of
2,4-3H-L-phenylalanine in liver microsomes (Freundt et al., 1974b).
Such changes follow the action of inducing agents, such as
phenobarbital, in the microsomal enzyme system. It has, therefore,
been suggested that carbon disulfide may have an inducing as well as
an inhibiting effect on mixed-function oxygenases. However, there is
no real evidence of such an effect at present (Freundt, 1977).
6. CARBON DISULFIDE POISONING
6.1 Historical Review
At the end of 1850, several physicians observed cases of strange
nervous and mental diseases, the origin of which remained obscure. In
1856, Delpech, reported 24 cases of carbon disulfide poisoning and
confirmed the diagnosis by animal experiments (Delpech, 1856a,b). In
1863, he reported 80 more cases of "carbon disulfide neurosis"
(Delpech, 1863). Cases of chronic poisoning were also reported in
England by Bruce (1884) and Foreman (1886). Laudenheimer (1899)
described carbon disulfide poisoning in German vulcanization shops,
stirring public opinion by drawing attention to about 50 cases of
"insanity". As further cases were reported in the USA (Jump & Cruice,
1904; Francine, 1905), the need for hygienic improvement in work
places was recognized. The first epidemic of carbon disulfide
poisoning due to the vulcanization process ended at the beginning of
the 20th century. At the same time, the viscose rayon industry started
to develop and expanded rapidly. Sporadic cases of carbon disulfide
poisoning in the viscose industry were reported between 1900 and 1930
(Quarelli, 1928) but the problem became serious in the 1930s.
Raneletti (1933), Quarelli (1934) and others described cases of
psychotic and polyneurotic disorders and extrapyramidal disturbances
(Audio-Gianotti, 1932; Teisinger, 1934).
In Japan, the viscose rayon industry was established in 1916 and,
in 1929, the first cases of carbon disulfide poisoning were reported
by Tokuhara, followed by other authors (review by Kubota, 1967). Many
cases of poisoning were also described in the USA (Hamilton, 1925,
1940; Bashore et al., 1938). The so-called Pennsylvania study by Gordy
& Trumper (1938) resulted in the establishment of the first TLV of
20 ppm adopted by the American Standards Association (1941). As the
hygienic standard in this industry improved, the incidence of severe
poisoning decreased. However, during World War II, the hygienic
situation in the expanding viscose industry deteriorated and severe
poisoning again became common.
During and after World War II, many cases of carbon disulfide
poisoning were reported, mainly from Italy (e.g., Vigliani et al.,
1944; Vigliani, 1946) but also from Belgium (Langelez, 1946; Merlevde,
1951) and Finland (Noro, 1944). After World War II, the viscose rayon
industry spread to many developing countries, where the whole sequence
of degrees of exposure was repeated. In the developed countries,
attention is now focused on slowly developing symptoms due to
long-standing exposure to relatively low concentrations.
6.2 Clinical Picture of Carbon Disulfide Poisoning
Carbon disulfide intoxication has been classified as hyperacute,
acute, subacute, and chronic.
Hyperacute poisoning occurs in extreme cases of massive exposure
for a short time to concentrations of about 10 000 mg/m3 or more. The
victim quickly falls into a coma and eventually dies. Acute and
subacute poisoning occurs with short exposure to carbon disulfide
concentrations ranging from 3000-5000 mg/m3 with predominantly
psychiatric and neurological signs and symptoms such as extreme
irritability, uncontrolled anger, rapid mood changes including maniac
delirium and hallucinations, paranoic ideas, and suicidal tendencies.
Other symptoms include memory defects, severe insomnia, nightmares,
fatigue, loss of appetite, gastrointestinal troubles, asthenia, and
interference with sexual functions, such as impotency.
The symptoms and signs of chronic carbon disulfide poisoning were
described by Vigliani (1946, 1961) and the following classification of
the different syndromes was suggested by Nesswetha & Nesswetha (1967):
-- psychoses characterized by manic and depressive symptomatology
and disorientation;
-- polyneuropathy of the lower extremities, with diminished or
completely absent Achilles and patellar tendon reflexes, sensory
disturbances in a glove-stocking distribution, diminished faradic and
galvanic excitability, and decrease of the motor and sensory
conduction velocity in the peripheral nerves;
-- disturbances of the gastrointestinal tract in the form of
chronic, hyper- and hypoacidic gastritis and duodenal ulceration;
-- myopathy of the calf muscles;
-- neurasthenic syndrome with disturbances in the autonomous
nervous system,
-- optic neuritis;
-- atherosclerotic vasculoencephalopathy; the principal forms
being bulbar-paralytic, hemiplegic, or extrapyramidal.
The typical mental deterioration has been called an organic
psycho-syndrome, which may be due to general cerebral atherosclerosis,
to direct toxic action upon the brain cells, or to both.
The pattern of carbon disulfide poisoning has changed with
improvements in hygienic standards in industry. However, when the
viscose industry is established in a country with no former
experience, there is always the risk of severe poisoning; this has
occurred many times.
6.3 Effects on Organ Systems
6.3.1 Dermatological effects
Liquid carbon disulfide represents a severe irritant for both the
skin and mucosa. Hueper (1936) found blisters in viscose rayon workers
and also in experimental animals that often resembled second and third
degree burns.
6.3.2 Ophthalmological effects
Studies on the ophthalmological effects of carbon disulfide in
animals have mostly been of a histological nature. In 1899, Koester
observed changes in the retinal ganglia. Seto (1958) found vacuolar
degeneration and tigrolysis in the retina, and atrophy of the optic
nerve in rabbits exposed to a carbon disulfide concentration of about
3700 mg/m3 for 1-3 h.
Ophthalmological examinations in man are informative because it
is possible to observe the visual capacity and to study the changes in
the vessels of the ocular fundus directly. According to the literature
(e.g., Nunziante-Cesaro et al., 1952; Savic, 1967) the following
effects have been observed:
-- changes in the motility of the eyelids;
-- changes in the sensitivity of the cornea and conjunctiva;
-- changes in the motility of the ocular bulbus;
-- changes in convergency and accommodation;
-- morphological changes in the fundus such as focal haemorrhage,
exudative changes, atrophy of the optic nerve, retrobulbar neuritis,
microaneurysms, and sclerotic changes of the blood vessels;
-- functional changes, e.g., disturbances of colour vision,
adaptation to the dark, reaction of the pupil to light, accommodation,
and decrease in visual accuracy.
Most of the effects mentioned above have resulted from heavy
exposure. Many that were observed decades ago, when the observations
often lacked comparison materials, are open to criticism. Nowadays,
under prevailing working conditions such grave changes are rarely, if
ever, seen.
Increased systolic and diastolic blood pressure in the retinal
arteries and damage to the arterial walls have been reported (Maugeri
et al., 1966d; Goto et al., 1971). Maugeri et al. (1966d, 1967)
studied the arterial pressure in 107 workers who had been exposed to
carbon disulfide at concentrations ranging from 200-500 mg/m3, with
peak concentrations of up to 900 mg/m3, for 1-9 years. Using an
ophthalmodynamographic method, the authors found an average increase
in both systolic and diastolic pressures in exposed workers of about
18.4/14.7 kPa (138/110 mmHg) compared with controls which averaged
15.3/11.6 kPa (115/87 mmHg). The increase was more marked for the
diastolic pressure than for the systolic. Measurement of the pressure
of the ophthalmic arteries may be a test that needs further evaluation
in the light of the demonstrated disturbance of the brain
catecholamine metabolism induced by carbon disulfide.
Savic (1967) studied changes in the nervous system of the eyes of
young workers exposed to carbon disulfide levels of 100-400 mg/m3.
Changes were observed but only after a long period of exposure
(>5 years) (Hotta & Savic, 1972). Retinal microaneurysms have been
reported in a high proportion of Japanese viscose rayon workers (Goto
& Hotta, 1967; Goto et al., 1971; Sugimoto et al., 1976). In one
study, the prevalences of microaneurysms as judged by fundus
photography, were 8% and 2% among exposed and unexposed male workers,
respectively (Goto & Hotta, 1967). In a subsequent study (Hotta &
Goto, 1971; Goto et al., 1971), a prevalence of 56% of microaneurysms
was found in the exposed group (241 subjects) and 15% in a control
group (30 subjects). The extremely high prevalence in the exposed
groups in these studies was due to biased selection of the subjects;
one-half of them were selected for examination because of known
microaneurysms revealed by ophthalmoscopy. Later studies by the same
team did not show such a high prevalence (Sugimoto et al., 1976), but
a statistically significant difference between exposed and unexposed
subjects still persisted. Retinaopathy was found in 89 out of 289
carbon disulfide workers (31%), but only in 2 out of 49 controls (4%).
The frequency of retinopathy was correlated with both the duration and
the intensity of exposure (Sugimoto et al., 1976). It has been
suggested that the retinal microaneurysms found in Japanese workers
were related to the diabetogenic action of carbon disulfide (Goto et
al., 1971). However, microaneurysms are generally associated with
hypoxia and, though they may occur in advanced cases of diabetes, they
are not, by any means, specific for the disease. Thus, to ascribe the
excess of aneurysms found in Japanese studies to subclinical diabetes
caused by carbon disulfide is not justified. It is more likely that
they were caused by a direct effect on the retinal vessels, possibly
in association with local hypoxia coacting with ethnic or
environmental factors.
The results of recent Finnish studies do not tally with the
Japanese findings. Out of 100 exposed men and 97 controls, only 3
exposed and 2 control subjects showed 1-5 microaneurysms, and one
exposed subject showed more than 5 (Raitta et al., 1974). Furthermore,
general narrowing of the arteries and calibre irregularity were so
common in the fundus of both the exposed and control subjects that
plain ophthalmoscopy did not have any discriminatory value in this
study. A subsequent Japanese-Finnish study showed that the differences
in values obtained in the 2 studies were true differences and were not
caused by inter-observer variation (Sugimoto et al., 1977).
The most interesting finding in a study by Raitta et al. (1974)
was the high frequency of delayed peripapillary filling that occurred
in 68 exposed and 38 unexposed men. The findings of Raitta & Tolonen
(1975), suggest that studying the microcirculation of the ocular
fundus by fluorescein-angiography and oculosphygmography would help in
the early detection of carbon disulfide effects, especially since the
vessels can be observed directly in their natural state.
Ophthalmoscopy is indicated for the examination of patients suspected
to be suffering from carbon disulfide poisoning and for following-up
such patients, but it is too prone to inter-and intra-observer
variation to be used routinely by plant physicians for regular health
checks. Fundus photography eliminates some of this variation, but its
evaluation should be standardized. The diagnosis of retinal
microaneurysms requires a fundus angioscreenography, but this method
is recommended for research purposes only.
It should be added that a mixture of carbon disulfide, hydrogen
sulfide and sulfuric acid mist from an acid bath caused
keratoconjunctivitis in exposed workers (Savic & Jovicic, 1965). This
phenomenon is called "spinner's eye".
6.3.3 Otological effects
Sulkowski & Latkowski (1969) observed that exposure to carbon
disulfide impaired hearing ability. The authors examined 60 workers
occupationally exposed to carbon disulfide for between one year and
more than 10 years. The workers were below 50 years of age.
Audiometric examinations showed impairment of hearing of the receptory
type in more than 50 workers and also a decreased ability to
distinguish sound intensity within the range 1.5-3.0 dB. This
suggested a central and supracochlear localization of the impairment.
Electronystagmographic examinations performed by these authors
disclosed reduced excitability of the vestibular apparatus, suggesting
an extralabyrinthine localization of the lesion. Loss of sensitivity
to high frequency tones has also been attributed to carbon disulfide
exposure (Zenk, 1970). Since noise levels are often very high in
viscose rayon plants, it is possible that the lesions found may be at
least partly due to this cause. Vestibular symptoms, as manifested by
vertigo and nystagmus may also be present in carbon disulfide
intoxication (Zenk, 1967, 1970).
6.3.4 Respiratory effects
Carbon disulfide is a known irritant, but few data exist about
its effects on the respiratory system (Zenk, 1967). Ranelletti (1933)
described chronic cough as a consequence of the irritant effect of
carbon disulfide, but it is necessary to take into consideration the
irritative effects of hydrogen sulfide and sulfuric acid mist and of
other irritants present in the air of viscose rayon plants. The
findings of Massoud et al. (1971) i.e., cough, phlegm, wheezing,
dyspnoea, precordial pain and palpitations, may also have been due to
such mixed exposure rather than to carbon disulfide alone.
6.3.5 Gastrointestinal effects
Gastrointestinal symptoms are common among heavily exposed
workers and patients with carbon disulfide poisoning. Bashore et al.
(1938) found a prevalence of such symptoms of 25%, Vigliani (1954),
28%, Karajovic et al. (1964), 66%, Lysina (1967), 27%, and Hass et al.
(1967), 27%. Since such symptoms occur among unexposed people too,
especially in shift work, the figures listed cannot be attributed
exclusively to carbon disulfide exposure (Knave et al., 1974). A
higher prevalence of gastrointestinal disorders and liver and bile
duct dysfunction was observed in 2 groups of workers (800 and 492
subjects, respectively) exposed to very low carbon disulfide
concentrations (4-12 mg/m3) than in a control group of 453 unexposed
workers. The prevalence in the 2 exposed groups was 4.7 and 5.6%,
respectively, and that in the control group, 2.2% (Murashko, 1975).
Later, Bittersohl & Thiele (1977) found a prevalence of 22.7% of
gastrointestinal changes in 309 exposed shift workers as opposed to
5.9% in 345 unexposed shift workers.
Based on histological studies of the gastric mucosa of 75 workers
exposed to carbon disulfide, Hassman et al. (1967) suggested
classification into 5 groups: normal gastric mucosa, superficial
gastritis, chronic gastritis with incipient atrophy, chronic atrophic
gastritis, and gastritis of undetermined type. Duodenal ulceration was
only found in 2 out of 75 subjects in this study. Of the workers
examined, 11% had dyspeptic complaints and gastritis was verified in
60% of the cases. A similar figure (66%) was found by Karajovic et al.
(1964).
6.3.6 Hepatic effects
Exposure to carbon disulfide has caused fatty degeneration and
haemorrhages of the liver in animals (Bashore et al., 1938).
Experimental studies on rats have shown that pretreatment with a drug
that stimulates liver microsome enzyme activity alters the degree of
liver damage on exposure to carbon disulfide. Thus, Bond et al. (1969)
reported necrosis in the livers of rats that had been pretreated with
phenobarbital and subsequently received a single oral dose of carbon
disulfide (1 ml/kg body weight) but not in rats treated only with
carbon disulfide. Moreover, only pretreated rats displayed hydropic
degeneration of the liver. Magos & Butler (1972) found that starvation
potentiated the effect of phenobarbital in rats subsequently treated
with carbon disulfide, and that the resulting hydropic degeneration
was reversible. Similar results were obtained by Freundt et al.
(1974a), when they administered a single oral dose of 1 ml of carbon
disulfide per kg body weight to rats pretreated with phenobarbital.
However, no degenerative changes in the liver were observed in
pretreated rats following exposure by inhalation to carbon disulfide
at about 60 mg/m3 (20 ppm) and 620 mg/m3 (200 ppm) for up to 7 days.
In experiments performed on rats with short-term (8-h) exposure to
carbon disulfide at concentrations ranging from 62-1250 mg/m3
(20-400 ppm), the energy potential of the organism was damaged mainly
because of a reversible augmentation of hepatic glycolysis (Kürzinger
& Freundt, 1969; Freundt & Kürzinger, 1975).
The possible effects of carbon disulfide on the liver have been
discussed in numerous reports of clinical observations. Some authors
deny that exposure to carbon disulfide results in toxic effects in the
liver, some claim degenerative changes of the hepatocytes, and others
state that sclerotic changes are produced under conditions of chronic
exposure. Such controversial observations can be explained by
nonuniformity in approach to the method of examination. Vidakovic et
al. (1965a) examined workers hospitalized because of chronic carbon
disulfide poisoning. The exposure of these workers had been extremely
high, ranging from 1400-2200 mg/m3 (Petrovic & Djuric, 1965).
Functional disturbances of the liver and fatty degeneration of
hepatocytes were found but no necrotic changes were observed.
Pirotskaja (1972) examined the protein-forming function of the
liver in workers (40 women and 6 men) who had been exposed to carbon
disulfide for 5 years. At first, the workers were exposed to a carbon
disulfide concentration of 90 mg/m3 but later, concentrations ranged
from 8-14 mg/m3. All exposed subjects were between 30 and 40 years of
age. The mean concentrations on cystine, thyroxine, methionine,
valine, leucine, and tryptophan in serum showed statistically
significant increases in comparison with a control group.
Statistically valid decreases occurred in the levels of glutamic acid,
aspartic acid, olanine, and lysine. The levels of histidine, arginine,
and serine did not differ from those in the control group. There was a
correlation between these changes and the clinical signs of carbon
disulfide intoxication. There was no difference in the serum contents
of total protein in the exposed and control groups.
6.3.7 Renal effects
Some authors have drawn attention to nephrosclerosis in autopsies
of patients with carbon disulfide poisoning (Uehlinger, 1952; Yamagata
et al., 1966; Sbertoli et al., 1969), but this could be ascribed to a
general atherosclerotic process induced by carbon disulfide. For
example, in the cases studied by Uehlinger (1952), typical
glomerulosclerosis of the Kimmelstiel-Wilson type was established in 4
patients, while the fifth showed arteriosclerotic injury of the
kidney. Obviously renal involvement represents a very late consequence
of heavy, protracted carbon disulfide exposure, since
Graovac-Leposavic & Jovicic (1971) reported that only one patient with
persistent kidney insufficiency had ever been diagnosed in a large
viscose rayon plant employing several thousands of workers. In another
study of viscose rayon workers, Hemberg et al. (1971) found a slight
but statistically significant rise in the mean plasma creatinine
concentration compared with a control group. All values were within
"normal" limits, however.
6.3.8 Haematological effects
Brieger (1949) studied the bone marrow of exposed rats and found
retarded maturation of the erythrocytes. Mild anaemia, a slight
decrease in the haemoglobin concentration, slight reticulocytosis,
eosinophilia, and hypercoagulability of the blood have also been
recorded in exposed rats (Vidakovic et al., 1965b).
Ivanova (1967) found a moderate decrease in the haemoglobin
concentration and erythrocyte count in men with only slight exposure
to carbon disulfide. Such results cannot be attributed to carbon
disulfide effects, but rather to poor standardization of the study
conditions, since significant haematological changes in the peripheral
blood were not observed, even in highly-exposed workers (Vidakovic &
Andjelkovski, 1965; Fahim et al., 1973). Thus, it has not been
confirmed that carbon disulfide causes anaemia or polyglobulia. It is
probable that the anaemia mentioned in some clinical reports reflects
malnutrition or some other nonoccupational cause, perhaps even
defective study design, rather than an effect of carbon disulfide.
The effects of carbon disulfide on blood coagulation mechanisms
are discussed in section 6.3.14.
6.3.9 The endocrine system
Damage to the endocrine structures with functional alterations
was described in animals by Ranelletti as early as 1931, and by
Audio-Gianotti in 1932. Impaired sexual function in patients with
carbon disulfide poisoning was reported by Gordy & Trumper (1938),
Langelez (1946), and Vigliani (1946). Vesce et al. (1953) observed a
decrease in 17-ketosteroids excretion in the urine of exposed rabbits.
This phenomenon was confirmed in studies on exposed workers by
Fruscella (1962) and Olienacz et al. (1964). Most of these
observations were made on workers belonging to older age-groups and
the confounding effect of age was not controlled. Cavalleri & Zuccato
(1965), however, obtained the same results taking the age factor into
account.
In 1965, a joint Italian-Yugoslav group began an investigation on
young workers in a Yugoslav viscose rayon plant. The workers were
classified according to duration of exposure to carbon disulfide. The
average ages of various groups ranged from 26 to 33 years and they
were exposed to average carbon disulfide concentrations of about
200-500 mg/m3 with peak concentrations of up to 900 mg/m3. The
excretion of 17-ketosteroids in the urine showed a linear decrease
with the duration of exposure (Cavalleri et al., 1966a,b, 1967). The
decrease in excretion of 17-hydroxycorticosteroids was most marked in
workers with short exposure and did not decrease any more as exposure
continued. The excretion of androsterone appeared to decrease
progressively with duration of exposure, probably in a linear fashion;
that of etiocholanolone did not show any uniform pattern (Cavalleri et
al., 1966c,d). Urinary excretion of testosterone and gonadotropin
luteinizing hormone was also reduced in workers exposed to
concentrations of 100-400 mg/m3 for 2-12 yearsa. Lancranjan et al.
(1971) reported a decrease in 17-ketosteroid and
17-hydroxycorticosteroid excretion in the urine of exposed workers.
The thyroid function has been studied in exposed workers by
determination of serum thyroxine (Cavalleri et al., 1971; Cavalleri,
1975; Massoud et al., in press). Decreases reported in thyroxine
levels suggest a reduction of thyroid activity.
Mild hypothyroidism was reported in 8% of exposed workers in
studies by Lancranjan (1972). This effect may be primary or may be due
to inhibition of the hypothalamus-hypophysis axis. A decrease in
thyrotropin-stimulating hormone, observed in human subjects given
tetraethylthiuram, favours the hypothesis of an impairment of the
hypothalamic-hypophyseal system (Cavalleri et al., 1977).
a Exposure data submitted by Professor Cavalleri as private
communication to the Task Group.
When the thyroid function is inhibited, disturbances in the lipid
metabolism will appear. A well-known manifestation of subclinical
hypothyreosis is elevation of the serum cholesterol level, which in
turn may contribute to the vascular changes typical of carbon
disulfide poisoning. The isolation and identification of two
metabolites, thiourea and mercaptothiazoline, may help to explain the
mechanism (Pergal et al., 1972a,b). Neither of these metabolites is
directly toxic, but both may have some influence on thyroid activity.
In fact, drugs containing substances of the thiourea and
mercaptothiazolinone type are used in the treatment of
hyperthyroidism.
Lancranjan et al. (1969, 1972), reported hypospermia,
asthenospermia, and tetratospermia in the spermatic liquid of young
exposed workers, confirming the gonadal injury reported by Cavalleri
et al. (1965).
Disturbances were found in the ovarian function of 500 young
females, exposed to concentrations of carbon disulfide of around
20 mg/m3, and of 209 women exposed to a concentration of less than
10 mg/m3 (Vasiljeva, 1973). In a comparison with a group of 429
unexposed women, menstrual flow durations of more than 5 days occurred
in 18% of the first group and in 11% of the second compared with 5% in
the control group. Irregular menstruation occurred in 8% of those
exposed to 20 mg/m3 as opposed to 2% in the comparison group. Other
menstrual disorders followed a similar pattern, and were correlated
with the length of exposure. When vaginal smears were examined from
subsamples, cellular disturbances were most frequent in the group with
highest exposure. A biochemical study of the sex hormones in the urine
confirmed the vaginal-smear findings. Hence, even rather low exposure
to carbon disulfide appeared to induce hormonal and functional
disturbances in young women. Other studies corroborate these findings
(Petrun, 1967, Finkova et al., 1973).
In a study of 380 women exposed to around 30 mg/m3, Petrov
(1969) found more pregnancy complications than in a control group of
191 women. Spontaneous abortions occurred in 14% of the exposed, and
7% of the unexposed women. "Threatened pregnancy terminations" also
occurred more frequently in the exposed women than in the controls
(26% and 13% respectively). Exposed women gave birth prematurely
significantly more frequently than controls (9% and 3%, respectively).
It has been reported in several studies that carbon disulfide
produces primary damage at the hypothalamic-hypophyseal level
(Cavalleri et al., 1965; Lancranjan et al., 1971; Maugeri et al.,
1971; Cavalleri, 1972). The hypothalamus produces the releasing
factors that stimulate the hypophysis to synthesize and release
polypeptide hormones, which in turn stimulate the target glands in
question. The impairments found would, therefore, represent a
secondary consequence.
Thus, carbon disulfide causes several disturbances of the
endocrine systems that can be summarized as follows:
-- a reduction of adrenal activity that can be repaired by ACTH
administration and can therefore be ascribed to reduced secretion of
corticotrophin;
-- a reduction of the endocrine activity of the testis and
impairment in spermatogenesis possibly due to direct gonadal damage;
this finding could explain the decrease in "libido" and "potentiae"
often found in exposed workers;
-- disturbances in the hormonal balance in women, resulting in
menstrual irregularities, spontaneous abortions, and premature births;
-- impairment of thyroid function, which could be primary or due
to a deficiency in the thyrotropin-stimulating hormone (TSH) or the
TSH releasing factor or both.
6.3.10 Effects on the nervous system
6.3.10.1 Central nervous system
In experimental animals, carbon disulfide causes destruction of
the myelin sheet and axonal changes in both the central and peripheral
neurons. Degenerative changes have been observed in the cortex, basal
ganglia, the thalamus, the brain stem, and the spinal cord (Wiley et
al., 1936; Ferraro et al., 1941; Lewey et al., 1941; Fischer &
Michalova, 1956; Drogicina, 1968).
The mechanism by which carbon disulfide causes these changes has
not been elucidated, however. Mitochondrial enzymes may be inhibited
(Tarkowski & Wrofiska-Nofer, 1966; Tarkowski & Sobczak, 1971), and the
tyrosine and catecholamine metabolism may be disturbed (Magos et al.,
1974). The content of free glutamine increases as a result of carbon
disulfide exposure (Efremova & Uzbekov, 1968; Tarkowski & Cremer,
1972). Such changes were observed with acute experimental poisoning,
but chronic, repeated exposure did not produce gross changes
(Tarkowski, 1973).
Horvath & Mikiska (1957) followed the electroencephalographic
changes in rabbits exposed to carbon disulfide and found a transitory
decrease in the amplitude of basic and superimposed beta rhythms.
The results of animal experiments have to be extrapolated to man
with extreme caution; however, many data obtained from animal studies
are in good agreement with clinical observations in man. Like other
narcotic agents, carbon disulfide produces a clinical symptomatology
of irritation and excitation on one hand, and inhibited psychomotor
activity, psychological alterations, insomnia, hypersomnia, loss of
consciousness and death, on the other.
In acute poisoning, the first typical signs from the central
nervous system (CNS) are excitation, euphoria, and aggressive
behaviour. Subacute and chronic cases are first characterized by
neurotic signs of unrest, excitation, and loss of temper. Gradually
the patient becomes depressive, anxious, paranoiac and sometimes there
is a suicidal tendency. Nightmares, apathy, loss of initiative,
vegetative disturbances, and headache are common symptoms (Melissinos
& Jacobides, 1967; Mihail et al., 1968). In the further course of
chronic intoxication, neurological signs become prominent. Both
diffuse cortical and focal symptoms from the subcortical grey matter
and extrapyramidal system are typical (Teisinger, 1934). Typical
Parkinson symptomatology with bradykinesia, bradylalia, muscle
rigidity, tremor, and increased elementary postural reflexes is
similar to that in arteriosclerotic or postencephalitic parkinsonism
(Nakamura et al., 1974). Psychic, pyramidal and extrapyramidal
symptomatology, including signs from other parts of the brain
(vestibular, cerebellar) give a picture compatible with diffuse
effects, described under the term of "toxic encephalopathy".
Furthermore, nervous involvement in general may be reflected as
changes in the sensitive branch of the trigeminal nerve, in the
function of the III, IV, and VI cranial nerves, and in the sympathetic
and parasympathic nerves of the eye.
Disturbances in cerebral circulation probably explain some of the
manifestations of neurotoxicity. Vigliani et al. (1944) demonstrated
hyalinosis of the arterioles and precapillaries histologically, a
finding that fits into the picture of general atherosclerosis caused
by carbon disulfide. Thus, the vascular changes described by Vigliani
(1961) as "encephalovasculopathia sulfocarbonica", are probably
responsible for many of the manifestations of central nervous system
pathology.
On the other hand, authors of some recent publications have tried
to explain some mechanisms by direct interference of carbon disulfide
and its metabolites in the system of enzymatic processes in the brain.
Morphological studies indicate that long axons in the spinal cord are
preferentially destroyed in chronic carbon disulfide poisoning
(Szendzikowski et al., 1973).
The typical neuropathological picture includes an increase in
neurofilaments, and secondary effects on the myeline sheath (Juntunen
et al., 1974). Biochemical studies have shown that oxidative
desulfuration also take place in the brain (Savolainen et al., 1977b),
and that the highest specific binding of the liberated sulfur is
detected in spinal cord axons (Savolainen & Vainio, 1976). It appears
that most of the liberated sulfur binds to neurofilaments that are
essential to normal axon functions (Savolainen et al., 1977c). Thus,
there is evidence of a direct toxic action upon the nervous system.
The results reported by Abramova (1967), Vasak & Kopecky (1967),
and others (section 5.3.1) regarding inhibition of the vitamin B6
metabolism, might explain some changes in the peripheral myelin sheet
and even some effects on the central nervous system. The consequences
of vitamin B6 inhibition, or interference with the transamination of
glutamic acid and inactivation of pyridoxamine could, perhaps, produce
irritative cortical symptomatology, including epileptiform cramps.
Furthermore, studies showing interactions between carbon disulfide
and, tissue respiration; depression of cytochrome oxidase, succinic
dehydrogenase, alkaline phosphatase, and dopamine-ß-hydroxylase; and
influence of inhibitors of serum elastase (EC 3.4.21.11), suggest that
besides a purely vascular etiology, it is plausible to consider a
direct toxic interference of carbon disulfide with tissue metabolism
(section 5.2). In fact, direct toxic effects of carbon disulfide on
peripheral nerve cells (Szendzikowski et al., 1973), and on the
myoneural junctions (Juntunen et al., 1977) have been shown in
animals. The central nervous system may react in the same manner.
From all these considerations, it can be concluded that the
effects of carbon disulfide on the nervous system may be influenced by
many factors. The method most often used for the study of cerebral
involvement in man is electroencephalography (EEG). However, studies
on carbon disulfide poisoning are rare. Early studies emphasize the
frequent occurrence of very flat records among subjects chronically
exposed to carbon disulfide (Krolikowska & Rucinska, 1959). More
recently, Seppäläinen & Tolonen (1974) found 21 abnormal EEGs in an
exposed group comprising 54 viscose workers compared with only 6 in a
control group of 50 paper-mill workers. Thiele & Wolf (1976) found EEG
changes in 52% of a group of 139 spinning workers. The abnormalities
consisted of slight, diffuse slow-wave abnormalities, slight to
moderately severe focal slow-wave abnormalities and even spike and
wave discharges in 3 exposed men. Thus, mild brain dysfunctions were
clearly more prevalent in the exposed group than in the control. EEG
findings among 250 workers with longstanding occupational exposure to
carbon disulfide (mean exposure 11.2 years) were recently described by
Styblva (1977) and compared with the EEGs of 61 healthy controls and
47 patients suffering from cerebral arteriosclerosis. Increased
frequency of abnormal EEGs was found in 33.2% of the exposed workers,
compared with 6.6% in the control group ( P < 0.01). The most
frequent abnormalities among the exposed workers were episodic theta
activity (33%) and diffuse abnormality (30%) (it was not, however,
indicated or even deducible from the data presented, how these
percentages had been calculated, i.e., from which population they were
taken). Local abnormalities were rare. Local EEG abnormalities
predominated in patients with cerebrovascular disease. The episodic
activity in the EEG was considered to reflect the direct toxic effects
of carbon disulfide on mesodiencephalic structures. The author
proposed that diffuse abnormality could be a manifestation of
vasculoencephalopathy resulting from toxic changes in the small
diameter vessels. The frequency of alpha activity tended to be lower
among exposed workers and this was considered to indicate a cerebral
metabolic disorder due to carbon disulfide exposure.
Studies on the behavioural and psychological effects of carbon
disulfide exposure have indicated disturbances in deep subcortical
structures, namely vegetative centres, and more diffuse cortical
involvement. The methods used have included diagnostic interviews,
behavioural observations (mainly on experimental animals), and
psychological test batteries.
Both structured and unstructured interviews of human subjects
showed differences in the clinical pictures of carbon disulfide
poisoning at rather high concentrations i.e., about 950-2400 mg/m3
(300-760 ppm) and at much lower concentrations i.e., around
20-50 mg/m3 (7-30 ppm) over a long period of exposure. At high
concentrations, symptoms of headache, muscle pain, fatigue,
paraesthesia, and general weakness were consistently reported (Gordy &
Trumper, 1940; Manu et al., 1970). At lower levels of exposure, fewer
complaints of muscular pain and overall weakness were noted, the main
symptoms being headache, memory impairment, rapid mood changes,
insomnia, paraesthesia, and fatigue (Seppäläinen et al., 1972; Lilis,
1974).
Standardized batteries of tests yield results that are directly
quantifiable and comparisons can be made between groups at different
exposure levels and control groups. Using this method, Hänninen (1971)
presented 3 groups of workers with different exposures and clinical
profiles (unexposed, exposed without clinical symptoms, and poisoned).
The results suggested that syndromes of latent and manifest carbon
disulfide poisoning differ in quality as well as in intensity, i.e.,
"clinically manifested poisoning is characterized by lowered
vigilance, diminished intellectual activity, diminished rational
control, retarded speed, and motor disturbances, whereas traits
indicative of depressive mood, slight motor disturbances, and
intellectual impairment are characteristic of latent poisoning"
(Hänninen, 1971). Analysis of intercorrelations among test sources for
each of the 3 groups resulted in substantially different factor
structures across the 3 groups. This supports the contention that
behavioural differences between groups exposed to different
concentrations are qualitative as well as quantitative (Hänninen,
1974). Naturally, the extent to which such distinctions are, in fact,
true, depends on the number of false-positive diagnoses in the
poisoned group (i.e., how many patients with other diseases had been
classified as cases of carbon disulfide poisoning), and, since this
aspect could not be evaluated, it may be premature to accept the
qualitative changes found as real facts. As to the quantitative
changes, it is self-evident that their severity must be correlated
with the degree of poisoning, at least on a group basis. The
specificity of the psychological and behavioural examination increases
when other sub-clinical signs of carbon disulfide exposure, such as
EEG and EMG findings are present.
Hänninen & Mäenpää, as reviewed by Tolonen (1974), applied the
same type of tests to 97 exposed male viscose rayon workers (mean
exposure time 15 years) and 96 age-and-sex matched controls from a
paper mill with no history of exposure to carbon disulfide. The most
distinct difference was in the retardation of psychomotor speed, where
the relative risk was 2.2 ( P = 0.02). As a whole, 40% of the exposed
and 25% of the control group showed "poor" performance, as defined
according to criteria published by Tolonen (1974). These results show
a clear group difference; however, the test battery has a low
specificity for carbon disulfide, as could be seen from the high
number of "false positive" results.
Recently, Hänninen and her co-workers increased the specificity
of the test battery, reducing the number of tests on the basis of
information gathered from new analytical approaches applied to results
of 206 exposed and 152 unexposed men, examined in 1972. According to a
multiple discriminant analysis, the most specific variables for carbon
disulfide effects were those indicating retarded speed, retarded
emotionality and energy, and psychomotor disturbances (Hänninen et
al., 1978). Another approach, starting from test results classified as
abnormal, resulted in similar conclusions (Tolonen et al., 1976). Now,
the test battery can be restricted to a few rather specific tests
applicable to suspected cases of poisoning that can also be used at
periodic health examinations.
Analogous results concerning the depressing effects of carbon
disulfide on psychological performance and behaviour have been
reported by Foa et al. (1976), Gherase (1976), Schneider (1976),
Tuttle et al. (1976)a and Hakky (1977).
Foa et al. (1976) demonstrated an exposure-effect relationship by
comparing workers from 2 plants with different degrees of exposure. A
relationship between exposure time and performance was demonstrated by
Schneider (1976), but the role of age as a confounding factor could be
excluded for 2 test variables only.
a Tuttle, T. C., Wood, G. D., & Grether, C. B. (1976) Behavioral and
neurological evaluation of workers exposed to carbon disulfide
(CS2). Unpublished report submitted to NIOSH (National Institute for
Occupational Safety and Health) by Westinghouse Electric Corporation,
Behavioral Services Center, Columbia, MD, USA, 156 pp.
From these data, it can be concluded that, at rather low exposure
levels, psychological test results are almost the only way of
assessing involvement of the central nervous system. Relationships may
exist between psychological impairment and EEG abnormalities, but
further studies are needed to clarify this point.
6.3.10.2 Peripheral nervous system
Many authors have studied the neuropathy and myelopathy of
animals exposed to carbon disulfide (Fischer & Michalova, 1956;
Higashida et al., 1959; Scheel, 1967; Frantik, 1970; Luke,, 1973;
Szendzikowski et al., 1973; Wrofiska-Nofer et al., 1973; Seppäläinen &
Linnoila, 1975, 1976). Destruction of the myelin sheet has been the
most prominent finding, but axonal changes have also been observed. In
the muscle fibres, atrophy of the denervation type occurred secondary
to the polyneuropathy. Some species differences in susceptibility to
carbon disulfide have been found. Cats and rabbits tolerated only low
concentrations of carbon disulfide. On the other hand, polyneuropathy
in rats was produced only with exposure to extremely high
concentrations (1200-2400 mg/m3). The slowing of nerve conduction
velocity in the sciatic nerve in rabbits and rats preceded clinical
symptoms such as clumsiness and later on, paresis. In rabbits,
electromyographic abnormalities suggested the presence of myelopathy
together with peripheral neuropathy (Seppäläinen & Linnoila, 1975,
1976). Neuropathological studies (Linnoila et al., 1975) showed severe
morphological alterations in the lateral and anterior funiculi of the
spinal cord in addition to pronounced axonal alterations in the
peripheral nerves of rabbits.
Lukas (1970, 1973) established by electromyography (EMG) that the
development of neuropathy in the rat was influenced by dose, duration
of exposure, physical conditions, and the nutritional state of the
animal. This confirms the long-standing clinical belief that the
individual constitution bears some relationship to the quantity and
quality of the effects of carbon disulfide (Djuric, 1967, 1971; Djuric
et al., 1973; Goto et al., 1971).
The protective effects of some substances have also been studied
in animals. The addition of zinc and copper salts (Scheel, 1967;
Lukas, 1970), and of pyridoxine to the diet (Frantik, 1970; Luke,,
1970) did not change the toxic effects of carbon disulfide but
improved the biochemical consequences of the intoxication, for example
by decreasing the excretion of xanthurenic acid with urine (Kujalova &
Tintera, 1970). Addition of nicotinic acid (Wrofiska-Nofer, 1970)
provided some protection against the effects of carbon disulfide lipid
metabolism. The clinical picture of carbon disulfide intoxication in
man is mainly based on toxic polyneuropathy (Bashore et al., 1938;
Vigiliana, 1954, 1961; Lukas, 1969; Lukas & Hromadka, 1977; Milkov et
al., 1970; Seppäläinen et al., 1972; Seppäläinen & Tolonen, 1974;
Seppäläinen, 1977; Gilioli et al., 1977). The subjective complaints
include paraesthesia, dysaesthesia, and fatiguability and diffuse
pains of the lower extremities. Sometimes irritative signs are
prevalent and sometimes the patients have a feeling of local
hyperaesthesia, or hypersensitivity on palpation in the muscles of the
lower limbs.
Distribution of the complaints is mostly symmetrical, and,
objectively, there is hyporeflexia. Klimkova-Deutschova (1965) pointed
out the characteristic dissociation of the patellar reflex response,
which is normal or increased, with a decreased or absent Achilles
tendon reflex. As the neuropathy develops, changes first appear in the
sensitive fibres of the lower extremities, later reaching the upper
extremities. (Lukas, 1969; Lukas & Hromadka, 1977). Hyperaesthesia is
mostly of a "sock" character, but sometimes it is also diffuse.
Palpation of the nerve roots and sometimes of the whole muscle groups
is painful. Today, the development of symptoms seldom reaches the
stage of motor paresis and, consequently, atrophic changes
(amyotrophy) are rare. The same is true of vasomotoric changes.
Amyotrophy has been described in the earlier literature in the area of
the triceps surae and quadriceps muscles of the legs, as well as in
the small muscles of the hand, the thenat, the hypothenar, and in the
interosseal muscles.
It has also been suggested that cells in the spinal cord are
injured (Manu et al., 1970; Seppäläinen et al., 1972). On the other
hand, disturbances in myoneural function have resulted in
myastheniform signs (Seppäläinen et al., 1972).
Needle electromyography and measurement of the motor conduction
velocities of peripheral nerves have proved to be sensitive methods
for detecting early nerve damage, even in neurologically normal,
exposed workers (Lukas, 1969; Manu et al., 1970; Seppäläinen et al.,
1972; Lilis, 1974; Seppäläinen & Tolonen, 1974; Knave et al., 1974;
Thiele & Wolf 1976; Lukas et al., 1977). The EMG abnormality most
often found consists of a reduced number of motor units at maximal
contraction and an increase in the duration of motor unit potentials
and fibrillation. The maximum motor conduction velocities (MCV) are
lower, especially in the leg nerves, and the distal latencies are
prolonged. However, the most sensitive test for peripheral nervous
involvement appears to be the conduction velocity of the slower fibres
of the ulnar and deep peroneal nerves, and that of the H-reflex in the
region of tibia] nerve (Lukas, 1969, 1977; Seppäläinen & Tolonen,
1974; Gilioli et al., 1977). In a recent epidemiological study
(Seppäläinen & Tolonen, 1974) comprising 118 male viscose rayon
workers and 100 control subjects from a paper mill, the most important
findings were as follows: the conduction velocities in the nerves of
the exposed men were generally slower than those of the controls. The
most distinct differences (all P values smaller than 0.01) were
obtained for the conduction velocity of the slower fibres of the ulnar
and deep peroneal nerves, the MCV of the deep peroneal and posterior
tibial nerves, and the motor distal latency of both the median and
ulnar nerves. The exposed group in this study was predominantly
composed of clinically "well" men, exposed for a number of years
(median 15) to carbon disulfide plus hydrogen sulfide concentrationsa
of about 60-190 mg/m3 (20-60 ppm) in the 1950s, about 30-95 mg/m3
(10-30 ppm) in the 1960s, and mainly below 60 mg/m3 (20 ppm) during
the last years. A little more than half of the men were still exposed
at the time of examination, the rest were working in "clean"
departments.
Different types of nervous system symptoms and findings among 51
Swedish viscose factory workers were evaluated by Knave et al. (1974).
The iodine-azide test was used as a measure of exposure intensity. The
comparison group consisted of 51 subjects matched for sex and age.
Symptoms of peripheral nervous dysfunction (e.g., restless legs,
cramps, pain, and numbness) were over-represented among workers,
especially the combination of these symptoms. Only one neurological
sign-namely tremor-showed a higher prevalence in the exposed than in
the control group. The authors measured only the maximal MCV of the
median nerve and found minor slowing in this MCV when the duration of
employment was between 5 and 30 years, but not after shorter exposure.
The prognosis of carbon disulfide polyneuropathy has not been
considered to be too poor. According to one follow-up study of exposed
workers, electroneuromyographic abnormalities tend to ameliorate if
the exposure level decreases or exposure is stopped (Seppäläinen,
1977). A tendency for repair and restitution of lost functions has
also been demonstrated experimentally in animals (Lukas, 1970;
Seppäläinen & Linnoila, 1975, 1976).
However, it has been shown that, even after periods of 10-15
years without exposure, abnormal conduction velocity values may be
found in workers with previous carbon disulfide poisoning or previous
long-term exposure to carbon disulfide (Seppäläinen et al., 1972).
This corresponds with the experience of Gilioli et al. (1977), who
found the reduction in conduction velocity to be irreversible. In
another study, Seppäläinen (1977) described an amelioration in the EMG
findings when carbon disulfide exposure was reduced or stopped.
a Exposure levels in studies by Hernberg et al. (1970, 1973, 1976),
Raitta et al. (1974), Tolonen (1974), Tolonen & Seppäläinen (1974),
and Tolonen et al. (1975) refer to combined concentrations of carbon
disulfide and hydrogen sulfide. Hydrogen sulfide concentrations were
about 10% of the carbon disulfide concentrations (Hernberg et al.,
1970).
6.3.11 Cardiovascular effects
A number of studies have shown that carbon disulfide causes
vascular changes in various organs of experimentally exposed animals
(Lewey, 1941; Guarino & Arcello, 1954; Paterni et al., 1958). In man,
evidence of vascular changes caused by long-term exposure, has been
accumulating since World War II. Attinger (1948) was the first to
describe such changes in 5 autopsy cases. Further clinical and autopsy
studies showed clearly that carbon disulfide was a vasotropic poison
(Uehlinger, 1952; Attinger, 1954; Vigliani & Pernis, 1955; Crepet et
al., 1956; Rechenberg, 1957; Maugeri et al., 1966a,b; Prerovska &
Roth, 1968; Maugeri et al., 1971; Prerovska & Zvolsky, 1973).
According to Vigliani & Pernis (1955) the "vasculopathia
sulfocarbonica" appears in 2 forms, namely in cerebral arteries
causing encephalopathy, and in the renal arteries causing nephropathy
and hypertension. The syndromes that follow these types of vascular
damage have already been described in sections 6.2, 6.3.7, and
6.3.10.1. Autopsy studies have revealed that sclerosis is prevalent in
large and middle-size arteries and hyalinization in arterioles and
capillaries. These changes appear in the blood vessels of all organs
but they are particularly prominent in the central nervous system and
kidneys.
Vascular changes due to carbon disulfide exposure are similar to
those produced with atherosclerosis due to age. Thus, the picture in
older persons is confused by the influence of age. The carbon
disulfide induced vasculopathy is therefore best studied in young
persons with a sufficiently long exposure time. With this in mind, a
joint Italian/Yugoslav team selected 104 young workers (age around
30-35 years) with more than 10 years' exposure for closer study
(Maugeri, et al., 1966a,b; Maugeri et al., 1971; Taccola et al.,
1971). Of these, 28 were invalids due to carbon disulfide poisoning.
Organic changes in blood vessels were discovered, mostly in the
invalids, by means of cerebral rheography. The results obtained
suggested progression of such changes even after the termination of
exposure. Using peripheral rheography, plethysmography, and
oscillography, the authors established functional vasoconstriction in
the arteries of the upper and lower extremities. Organic alterations
of the sclerotic type were rare and mainly appeared after prolonged
exposure and in the lower extremities.
During the last 10 years or so, it has become increasingly
evident that long-standing exposure to carbon disulfide promotes
coronary heart disease, even under circumstances where clinical
poisoning is uncommon. Of the several attempts made to connect carbon
disulfide exposure with coronary heart disease, only a few have
complied with necessary methodological requirements. The first such
investigation was a careful mortality study in Britain published by
Tiller et al. (1968). They found a 2.5-fold excess mortality in
viscose rayon workers exposed to carbon disulfide for 10 years or
more, compared with that of other workers. The same study demonstrated
that, in the 3 plants studied, coronary mortality was proportionally
higher among workers engaged in the viscose spinning process than in
other workers, men living in the locality, and the national statistics
derived from the Registrar General's tables. Similar results have been
obtained from Norway, where a 3-fold excess mortality from coronary
heart disease was found among workers, aged 35-54 years, compared with
unexposed workers in other departments (Mowé, 1971).
A prospective study that was initiated in 1967 in Finland, showed
that the 5-year mortality in a cohort of 343 male viscose rayon
workers exposed to about 30-95 mg/m3 (10-30 ppm) was almost 5-fold
(14 deaths compared with 3) that of an unexposed comparison cohort
from a nearby paper mill. Other causes of death were evenly
distributed (Hernberg et al., 1970, 1973). The incidence of nonfatal
first myocardial infarctions was almost 3 times that in the comparison
cohort (11 compared with 4) (Tolonen et al., 1975). In a continuation
of the follow-up extending to 8 years, the cumulative incidence rate
for mortality from coronary heart disease was 5.8% in the exposed
group and 2.6% in the comparison group, giving a rate difference of
3.2% (Hernberg et al., 1976; Nurminen, 1976). Between the sixth and
ninth years of follow-up, no further excessive mortality was observed,
probably because most (81%) of the originally exposed workers were no
longer exposed to carbon disulfide and also overall concentrations of
carbon disulfide had decreased to less than 30 mg/m3 (10 ppm). This
positive trend was promising with regard to coronary heart disease:
the prognosis of exposed workers improved with improved occupational
hygienic measures and with a reduction in the length of exposure over
a lifetime.
Anamnestic angina was more prevalent both in 1967-68, and at
re-examination in 1972, whereas there were no great differences in
coronary electrocardiographic findings (Hemberg et al., 1970; Tolonen
et al., 1975) as coded by the "Minnesota Code" (Blackburn et al.,
1960). These results showed that the greatest effect of carbon
disulfide on coronary arteries was on the production of fatal
infarctions (relative risk 4.8) followed in reducing order by all
infarctions, fatal and nonfatal (relative risk 3.7), nonfatal
infarctions (relative risk 2.8), angina (relative risk 2.2), and
coronary ECGs (relative risk 1.4). In other words, the more serious
the outcome, the greater the relative risk. The interpretations may be
that exposure to carbon disulfide not only worsens the prognosis of
existing coronary heart disease but increases the incidence of new
cases. However, it is not certain that this mode of action operates in
countries where coronary heart disease is less common than in Finland.
Coronary heart disease has a multifactorial etiology, and the pattern
described above may be noticeable only when a sufficient number of
other coronary risk factors are present at the same time. In fact,
some other studies have shown a more distinct excess of coronary ECGs
(Goto & Hotta, 1967; Cirla et al., 1972). It is possible that both
dose-dependent, methodological, and even geographical factors may be
responsible for the discrepancies reported so far. For example, it may
be that the pattern of excess coronary heart disease differs in
different populations, depending on the concomitant prevalence of
other coronary risk factors such as elevated blood lipids and
hypertension.
A joint analysis of the prevalence of angina pectoris, exercise
ECGs, and blood pressure measurements in Finnish and Japanese workers
exposed to carbon disulfide and in 2 unexposed control groups
supported this view (Tolonen et al., 1976). Angina was rare and
evidence of past infarction nonexistent in both exposed and unexposed
Japanese men; in the Finnish exposed and unexposed groups the
prevalence of angina was 15 and 10%, respectively. The prevalence of
coronary ECGs was almost equal among the exposed and unexposed men in
both the Japanese and the Finnish groups. Angina plus coronary ECGs as
evidence for coronary heart disease occurred with a prevalence of nil
for both exposed and unexposed Japanese, and 5 and 2% for the exposed
and unexposed Finnish groups, respectively. The study did not yield
any evidence of an excess in any of the variables examined among the
exposed Japanese workers. However, a follow-up study of this group is
necessary to shed more light on the possibility of excess morbidity
and mortality from coronary heart disease. Vertin (1977) did not find
any significant differences in coronary heart diseases, ECG changes,
serum cholesterol concentrations, and arterial pressure in workers
exposed to carbon disulfide concentrations of about 30-95 mg/m3
(10-30 ppm) compared with a control group.
Carbon disulfide vasculopathy may be due to the combined action
of several biochemical and physiological disturbances, namely:
(a) changes in lipid metabolism (section 6.3.9);
(b) disturbances in the coagulation mechanism (section 6.3.14);
(c) elevation of blood pressure (Hemberg et al., 1970; Sakurai,
1972; Tolonen et al., 1975);
(d) subclinical hypothyroidism (Cavalleri et al., 1971);
(e) a toxic effect on the myocardium, either direct or through
interference with the catecholamine metabolism (Magos, 1972).
However, much work remains to elucidate the mechanism of carbon
disulfide-induced atherosclerosis. In particular, coronary heart
disease has a multicausal origin and is closely related to the
saturated fat intake of the population. Furthermore, its incidence is
influenced by a large number of other risk factors, such as smoking,
diabetes, and physical inactivity. A combination of 2 or more risk
factors greatly increases the incidence. Thus, it may be postulated
that carbon disulfide will only become evident as a coronary risk
factor in the presence of other risk factors.
6.3.12 Carcinogenicity and mutagenicity
No reports are available that indicate any carcinogenic or
mutagenic effects of carbon disulfide.
6.3.13 Teratogenic effects
Only a weak teratogenic effect appeared in rats following
low-level exposure to a combination of hydrogen sulfide and carbon
disulfide (Bariljak et al., 1975).
A marked impairment (increase in early embreonal mortality,
reduction in fetal weight, malformations in the brain and limbs,
behavioural deviation) in the prenatal development of 2 successive
generations was produced in rats exposed to carbon disulfide
concentrations of 100 and 200 mg/m3 throughout the period of
gestation (Tabacova et al, 1977).
6.3.14 Other effects
The literature is contradictory concerning the effects of carbon
disulfide on blood coagulation. Saita & Gattoni (1957) found a slight
decrease in factor VII and prothrombin in exposed workers. On the
other hand, Moreo & Candura (1960) and De Nicola et al. (1962)
reported hypercoagulability in the blood of experimental animals, a
finding that was later confirmed in studies of exposed workers by
Candura et al. (1962), Saita et al. (1964), and Danilova (1968). An
increase in fibrinolysis was found by De Nicola et al. (1962) in
animals suffering from acute poisoning. In contrast, Candura et al.
(1962) found decreased fibrinolysis in workers exposed to carbon
disulfide.
The coagulation mechanism in highly-exposed young workers in a
viscose factory was studied by Visconti et al. (1966a,b,c, 1967). They
found protracted coagulation, retarded production of active
thrombotastin, decreases in plasminogen and in the activity of
plasmin, and a decrease in antiheparinic activity in workers exposed
for up to 9 years. These disturbances were most pronounced during the
first years of exposure.
Visconti et al. (1966a) supposed that carbon disulfide could
affect blood coagulation either directly by interfering with the
system itself, or indirectly, by liver injury. Different routes of
entry, differences in the level and duration of exposure and in the
distribution of carbon disulfide in the organism, and also
interspecies differences are probably responsible for the confusing
results.
It has been reported that the glucose metabolism is disturbed by
carbon disulfide both in experimental animals (rabbits) (Hara, 1958)
and in exposed workers (Goto et al., 1971). There are also a few
clinical studies reporting an increased occurrence of diabetes in
patients suffering from severe carbon disulfide poisoning (Austoni &
d'Agnolo, 1957; Finulli & Ghislandi, 1959; Ferrero, 1969). Lack of
proper control groups renders the interpretation of these results
difficult.
6.3.15 Interactions with other chemical compounds
The combined effects of carbon disulfide with other chemical
compounds were first reported by Lazarev et al. (1965) who indicated
that repeated exposure to a concentration of carbon disulfide of
3900 mg/m3, for 2 h per day, over a period of 20-30 days considerably
prolonged both the hexobarbital sleeping time and alcohol retention
time in rats and mice (see also section 6.3.6).
Carbon disulfide caused reversible inhibition of the oxidative
drug metabolism in rat liver endoplasmic reticulum (Bond et al., 1969;
Freundt & Dreher, 1969; Freundt & Kuttner, 1969; Freundt & Henschler,
1971; Freundt, 1973; Sokal, 1973; Freundt et al., 1974b, 1975, 1976).
This effect was also produced in man after exposure to a carbon
disulfide concentration of about 30 mg/m3 (10 ppm) over a 6-h period
(Mack et al., 1974); the treatment led to a rapidly reversible but
significant decrease in the oxidative N-demethylation of the
analgesic, aminopyrine.
Simultaneous exposure to carbon disulfide and ethanol resulted in
an increase in blood acetaldehyde in rats and man that was considered
to be a consequence of the inhibition of aldehyde dehydrogenase by
carbon disulfide (Freundt & Netz, 1973; Freundt & Lieberwirth, 1974;
Freundt, 1974; Freundt et al., 1976). In 12 healthy male volunteers, a
50% increase in blood acetaldehyde was noted following exposure to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) for 8 h
with simultaneous ingestion of ethanol in orange juice at a
concentration that maintained the blood ethanol level at about 0.8%.
However, no signs of an "antabuse syndrome" were observed under these
experimental conditions (Freundt et al., 1976).
6.4 Diagnosis
Since all isolated effects are nonspecific, individual diagnosis
becomes a matter of probability based on: (a) the ascertainment of
exposure; (b) the demonstration of signs and symptoms of poisoning
and the combination in which they occur; and (c) the exclusion of
other diseases.
The changes due to carbon disulfide exposure described in
sections 6.2-6.3.15 affect so many different organ-systems that, in
considering the etiology of a single case, the probability of it being
caused by carbon disulfide increases in proportion to the number of
these signs and symptoms present. Although, in 1941, Lewey drew
attention to the importance of the concomitant occurrence of different
neuropsychiatric and vascular effects, as well as to their connection
with the severity of the disease, there have been few, if any, studies
systematically analysing the knowledge that has emerged since then.
For this reason it is difficult to find quantitative data. However,
carbon disulfide poisoning should always be suspected when a viscose
worker presents with subjective, neurasthenic symptoms, signs of
peripheral neuropathy, psychological disturbances, and vascular
changes in the cerebral, coronary, renal, or peripheral systems. When
exposure to toxic levels of carbon disulfide occurs, the following
chronological effects can usually be expected: the appearance of
neurasthenic symptoms accompanied by psychological behavioural
changes; signs of peripheral nervous system involvement as shown by
electromyographic changes, initially mainly in the afferent fibres but
later in the motor fibres; simultaneous disturbances in the central
nervous system may appear but may be more difficult to detect
objectively than the lesions of the peripheral nervous system. Another
early finding is a decrease in the level of serum thyroxine that is
probably due to damage of the hypothalamic-hypophyseal axis (section
6.3.9). Cardiovascular damage appears after longer periods of
exposure. The appearance and severity of the above-mentioned signs and
symptoms are related to the level of the uptake and to the duration of
exposure to carbon disulfide. The changes are also dependent on the
individual constitution and health status of exposed workers.
In the differential diagnosis of true cases, atherosclerosis due
to other causes should be considered including, postencephalitic or
atherosclerotic Parkinsonism, brain tumours, syphilis, multiple
sclerosis, and psychiatric diseases of other origins such as endogenic
depression, schizophrenia, and alcoholism. Peripheral polyneuropathy
should be distinguished from that due to alcoholism, diabetes, and
other toxic agents.
The subclinical stage is, by definition, more difficult to
identify. First, the presence of pathological symptoms and signs must
be established, which requires sensitive techniques. Second, the
causal connection between an established symptom or sign and carbon
disulfide exposure should be shown. Both aspects pose great
difficulties because of the nonspecificity of the manifestations and
their vagueness in the initial stages. So far, the only attempt to
approach this problem in a systematic way has been made by Tolonen
(1974).
He studied 97 men exposed to carbon disulfide in a viscose
factory and 96 controls of the same age from a paper mill. The mean
age was 48 years (range 33-67 years), and the mean exposure time was
15 years (range 1-27 years). The intensity of background exposure
ranged from about 60 to 125 mg/m3 (20 to 40 ppm) in the 1950s and
then from about 30-95 mg/m3 (10-30 ppm). Personal exposure data were
not available. The group comprised both completely "healthy" workers
as well as some partly incapacitated patients with past poisoning.
About half of the men were still exposed to carbon disulfide, the rest
being employed in "clean" departments. The groups underwent many
examinations including: (a) examination of the heart (positive =
history of verified myocardial infarction, and/or "Minnesota Codes"
I1-3, IV1-3, V 1-3, VIII3, VIII3 , XI1-3-5-7, and/or typical angina):
(b) psychological testing (section 6.3.10.1); (c) measurement of
the conduction velocities of 8 peripheral nerves (polyneuropathy was
considered to exist when 2 or more nerves showed reduced conduction
velocities) and (d) examination of the circulation of the ocular
fundus (the criterion for disturbed circulation was delayed
peripapillary filling -- circumferential, segmental, or both). The
occurrence of isolated and combined signs is shown in Table 2.
As many as 59% of the exposed and 29% of the unexposed men were
affected by more than one disorder under study but most combinations
of disorders occurred more frequently in the exposed group; a
combination of 3 abnormalities was 3 times more common in the exposed
group than in the controls and a combination of all 4 abnormalities
occurred only in the exposed group. Only 5% of the exposed subjects
compared with 31% of the controls were without any abnormalities.
Since disturbances in the choroidal circulation were present in all
cases of excess "morbidity" (68% of the exposed, Raitta et al., 1974),
it seems that this abnormality represents the earliest manifestation
of carbon disulfide toxicity, at least, of those considered here.
Thus, it can be postulated that a positive diagnosis of carbon
disulfide poisoning can only be made if changes in the choroidal
circulation have been observed and providing that exposure has
extended over 10 years or more, with an intensity of the magnitude of
about 30-90 mg/m3 (10-30 ppm). The estimated probability that a
combination of findings is of occupational origin is the excess
morbidity over total morbidity. The results also showed that the
etiological role of carbon disulfide could be demonstrated with
greater probability, the greater the number of abnormalities present
at the same time.
The necessity to ascertain exposure is so obvious that it should
not need to be mentioned. It should be stressed that recent experience
has shown the importance of evaluating personal, as opposed to
background exposure, since the former may be only weakly correlated
with the latter. Personal exposure can be measured using personal
samplers, or by the iodine-azide test. The latter has been claimed to
discriminate particularly sensitive workers (whose test does not
return to normal after 16 h away from exposure) (Graovac-Leposavic et
al., 1967).
Table 2. Prevalence (%) of coronary heart disease (CHD), delayed
peripapillary circulation (EYE), polyneuropathy (PN) and
behavioural symptoms (BS), their combinations in subjects
in the exposed and control groups, and the differences
between the groups.a
Exposed Group Difference =
(N = 97) control excess morbidity
(N = 96)
Free of disease 5 31
CHD only 4 9 -5
EYE only 19 19 0
PN only 6 6 0
BS only 7 6 1
CHD + EYE 4 4 0
CHD + PN 1 1 0
CHD + BS 1 2 -1
EYE + PN 12 5 7
EYE + BS 7 4 3
PN + BS 4 5 -1
CHD + EYE + PN 9 0 9
EYE + PN + BS 11 5 6
CHD + PN + BS 2 1 1
CHD + EYE + BS 2 2 0
CHD + EYE + PN + PS 6 0 6
Total 100 100 26
a From: Tolonen (1974).
6.5 Surveillance of the Health of Exposed Workers
Considering the early manifestations of chronic carbon disulfide
effects and the factors that may influence the appearance and the
severity of such effects (section 6.4), the following examinations are
recommended for health surveillance:
(a) Clinical neurological investigation;
(b) Electroneuromyographic (ENMG) examination wherever possible
(especially the conduction velocity test, which should be carried out
on several nerves);
Additional tests that may give valuable information include:
(a) Psychological and behavioural testing with a narrow battery
of well validated tests;
(b) Serum thyroxine measurement repeated at least once a year;
(c) Blood pressure measurements;
(d) Electrocardiography (preferably an exercise ECG);
(e) Fundus photography (in some countries);
(f) Blood lipid pattern estimation, if the exposure level is
high;
(g) Electroencephalography (EEG), if there are special
indications;
These tests should be performed at the pre-employment examination
and subsequently at regular intervals to detect early deviation from
baseline values. Changes observed in the same individual over a period
of time may suggest a relationship with carbon disulfide uptake.
Depending on the intensity of exposure, the iodine-azide test
should be carried out from 2-12 times a year, both immediately after
the work shift and the next morning. Those with positive tests should
be seen by the plant physician, since a pathological test in the
morning is a warning of incipient poisoning (Graovac-Leposavic et al.,
1967). In addition to this, medical examinations should be made once
or twice a year. They should comprise a thorough history and a
neurological examination.
When suspect signs of early carbon disulfide effects appear, the
worker should be removed from exposure to a "clean" job. In the light
of the new findings concerning coronary heart disease, the removal of
workers who develop coronary risk factors, such as hypertension,
hypercholesterolaemia, and ECG changes may be indicated, irrespective
of their etiology. In some plants, there is the practice of regular
periods of work in a clean atmosphere, e.g., every sixth month, or of
extra vacations in the winter.
Some authors believe in the importance of the diet of exposed
workers. However, the protective effect of various diets and
supplements for man remains unclear. Scheel (1965) claimed that
addition of zinc and copper salts to the diet of exposed rats had a
protective effect against carbon disulfide exposures. A diet poor in
proteins, especially amino acids containing sulfur, but enriched in
vitamin B6 and glutaminic acid was suggested by Agronovskij &
Goloscapov (1972). Lukas (1973) established that rats on an "optimal"
diet were more resistant to the effects of carbon disulfide than rats
on a normal diet. In the first group, the signs of intoxication
appeared after a longer time and the effects were less marked than in
the second group.
At the present time, the only recommendation that can be given in
the case of man is that workers should be provided with a diet
containing sufficient amounts of energy foods to provide the required
joules, proteins, vitamins, and trace elements. This recommendation is
of particular importance in countries where such dietary requirements
are not necessarily fulfilled normally. However, basing the preventive
programme mainly on diets or drugs should be condemned, since the
fundamental question is that of technical improvements.
6.6 Contraindications for Exposure to Carbon Disulfide
An inborn error of metabolism manifesting itself as an abnormal
iodine-azide reaction in subjects working under conditions of high or
moderate carbon disulfide exposure (less than, 50 mg/m3) should be
considered an absolute contraindication for exposure.
Other contraindications for working in an environment containing
carbon disulfide include those common to all other toxic exposures
such as youth (below 18 years), pregnancy, concomitant chronic disease
such as psychiatric neurological, cardiac, renal, and pulmonary
diseases, and any condition that prevents the use of respirators, etc.
In addition, contraindications based on the specific toxicity of
carbon disulfide include diseases of the metabolic system, such as
liver disease and endocrine disorders. Any neurological or psychiatric
disorder is also a contraindication, including vegetative dystony and
psychic lability. A history of gastritis and peptic ulcer is a
relative contraindication, depending, for instance, on whether or not
there will be shift work and on the intensity of exposure. Recently,
attention has been focused on to what extent the presence of coronary
risk factors should be regarded as a contraindication; no strict
recommendations can be given, but as a general rule the presence of
several of these in an individual, or alternatively strongly abnormal
values for one risk factor, should be regarded as contraindications
(the strongest coronary risk factors apart from age itself are
elevated serum cholesterol or triglycerides, heavy cigarette smoking,
hypertension, and diabetes). In the evaluation of these and other
contraindications, the exposure intensity and hygienic conditions
should, of course, be taken into consideration.
The employment of women in work places with carbon disulfide
exposure is a special problem.
Considering the studies referred to in section 6.3.9, it is
advisable to exclude pregnant women from carbon disulfide exposure.
The same is true for lactating women and for those with expressed
disturbances of menstruation and habitual abortion.
7. EXPOSURE-EFFECT AND EXPOSURE-RESPONSE RELATIONSHIPS
7.1 Validity of Exposure Data
In general, valid exposure data are available only from
experimental studies on animals. Almost all epidemiological studies
have failed to document the exposure levels pertinent to the effects
studied. There is a universal lack of retrospective exposure data that
usually renders the assessment of the relationship between chronic
effects and the exposure levels responsible for their occurrence
impracticable. Furthermore, even when exposure data are available,
they often consist of occasional short-time measurements from fixed
sites that are not necessarily representative of the workers'
exposure. It is, therefore, extremely difficult, if not impossible, to
establish exposure-effect or exposure-response curves for carbon
disulfide, based on epidemiological evidence. Even the relation of
isolated findings reported in the literature, to given exposure levels
is difficult. Thus, the attempts in this document, to relate the
intensity of exposure to the intensity and frequency of effects must
be regarded with caution.
7.2 Experimental Data
7.2.1 Acute animal exposure
Acute exposure to carbon disulfide in animal experiments by
inhalation or by intramuscular or intraperitoneal injection has
provided information regarding the mechanism of carbon disulfide
toxicity, but it is difficult to apply this information to the
industrial exposure of man.
7.2.2 Long-term animal exposure
Several experiments on the inhalation of carbon disulfide have
been performed on different types of animals. In most of these
studies, exposure was to carbon disulfide only, but in a few, a
combination of carbon disulfide and hydrogen sulfide was used.
The concentrations of carbon disulfide ranged from
0.1-2330 mg/m3 and those of hydrogen sulfide, from 0.1-140 mg/m3;
the exposure periods ranged from 30 min per day to 6 h per day for up
to 15 months.
The reactions to such exposures included increased mortality,
teratogenic changes, testicular lesions, aspermatogenesis, engorged
blood vessels, weakness, paralysis, lethargy, weight changes,
metabolic changes, behavioural changes; sometimes there were no
observed effects. Some of the effects resembled lesions found in
exposed workers; others did not bear any relationship to those arising
from human exposure (Wakatsuki & Higashikawa, 1959; Goldberg et al.,
1964; Minden et al., 1967; Yaroslavski, 1969; Frantik, 1970; Gondzik,
1971; Misiakiewicz et al., 1972; Szendzikowski et al., 1973; Bariljak
et al., 1975; Seppäläinen & Linnoila, 1976; Tabacova et al, 1977). The
considerable differences in ranges of concentrations and periods of
exposure used in these experiments make conclusions regarding
exposure-effect and exposure-response curves impossible. This
regrettable situation emphasizes the need for international
coordination of toxicological experiments to make comprehensive
scientific evaluation possible.
7.3 Epidemiological Data
All studies so far reported have been from the viscose rayon
industry, where exposure to carbon disulfide occurs together with
exposure to hydrogen sulfide. However, generally, the levels of carbon
disulfide are at least one order of magnitude higher than those of
hydrogen sulfide, and most authors have attributed the health effects
found to carbon disulfide rather than to hydrogen sulfide exposure.
Only some of the various effects reviewed in section 6 can be
related to exposure levels. The following section contains a brief
review of exposure-effect and exposure-response data for neurological,
cardiovascular, ocular, and gonadal effects.
7.3.1 Neurological and behavioural effects
Severe acute neurological effects such as psychosis and paralysis
occur when exposure exceeds 1500-3000 mg/m3 (500-1000 ppm) (Vigliani,
1954). Chronic encephalopathy may develop with long-standing exposure
to 150 mg/m3 (50 ppm) or more (Vigliani, 1954). More subtle
neurological changes, detectable by neurophysiological examination or
psychological testing, have been reported at lower concentrations. For
example, slowing of nerve conduction velocities and prolonged distal
latencies of several nerves have been found among viscose rayon
workers with a median exposure time of 15 years to combined
concentrations of carbon disulfide and hydrogen sulfide of
60-190 mg/m3 (20-60 ppm) in the 1950s, 30-95 mg/m3 (10-30 ppm) in
the 1960s, and mainly below 60 mg/m3 (20 ppm) during the years
preceding the examination (Seppäläinen & Tolonen, 1974). The same
workers were also studied for psychological performance (Tolonen,
1974). The most distinct difference compared with a control group was
found in the retardation of psychomotor speed. As a whole, "poor"
performance, as defined by Tolonen (1974), occurred 1.6 times more
frequently among the exposed workers. These results suggest that
concentrations around 60-90 mg/m3 (20-30 ppm) can produce
psychological disturbances. In an indirect way, Tuttle et al.a
corroborated this view by being able to relate behavioural test scores
to other indices of neurological health, thought to occur under
similar exposure conditions, but because no exposure data were
available, their findings are not directly applicable in the
exposure-effect sense.
According to Martynova et al. (1976), an excess of functional
nervous disorders was found among 108 workers exposed to carbon
disulfide concentrations about 8-11 mg/m3 (maximum approximately
20-25 mg/m3), for 10-15 years, compared with 390 control subjects.
For example, sensory polyneuritis was much more frequent and the
threshold for pain was significantly higher in the exposed group.
These studies indicate that background concentrations below about
60 mg/m3 (20 ppm) may cause neurological dysfunction.
7.3.2 Cardiovascular effects
Severe vascular effects manifesting themselves as
vasculoencephalopathy and renal atherosclerosis developed with
long-term exposure to carbon disulfide concentrations exceeding
150-300 mg/m3 (50-100 ppm) (Vigliani, 1954; Nofer et al., 1961).
Excess incidence of coronary heart disease has been reported with
longstanding exposure to concentrations averaging 30-125 mg/m3
(10-40 ppm) (Hernberg et al., 1970, 1973, Tolonen et al., 1975). The
mortality rate from coronary heart disease appeared to be most
affected while the incidence of milder manifestations such as angina
and ECG-changes was less influenced. In fact, most studies, published
so far, have failed to reveal any difference in the prevalence of
ECG-findings between exposed and unexposed workers (Tolonen et al.,
1976).
Quite recently, Vertin (1977) reported no difference between the
incidence of coronary heart disease found in control subjects and that
in workers exposed to about 30-95 mg/m3 (10-30 ppm) suggesting a
no-observed effect level for coronary heart disease in this range.
This finding does not necessarily contradict those referred to above;
possibly, the true exposure levels responsible for the excess coronary
heart disease in the Finnish studies were higher than those in
Vertin's study. It is probable that, in general, differences in actual
personal exposure levels in the studies-not necessarily those levels
measured and reported -- account for seeming discrepancies in results
and that exposure data are inadequate in most published studies.
a Tuttle et al., 1976. Unpublished report. (See footnote to section
6.3.10.1.)
Differences in blood pressure have been reported by Hernberg et
al. (1970) and Tolonen et al. (1975) between workers exposed to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) and
unexposed subjects, and by Martynova et al. (1976) in workers exposed
for 10-15 years to concentrations of about 10 mg/m3. These findings
indicate that effects upon the vascular system begin at low exposure
levels.
7.3.3 Ophthalmological effects
Among Japanese workers, the occurrence of retinal microaneurysms
was related to the duration of exposure (Sugimoto et al., 1976). The
exposure levels in earlier Japanese studies were not reported; in
present studies they are probably of the order of 60 mg/m3 (20 ppm).
The findings of retinal microaneurysms appears to be confined to
Japanese workers, since, according to a recent joint study, no such
effect could be demonstrated in Finns (Sugimoto et al., 1977). In
contrast, Finnish workers showed delayed peripapillary filling and
wider calibre of retinal arteries compared with control subjects
(Raitta et al., 1974). Exposure conditions under which these changes
occurred are described in section 6.3.2 and indicate that haemodynamic
changes in the ocular circulation are induced as early as coronary
effects.
7.3.4 Gonadal effects
The results of Vasiljeva (1973), Petrov (1969), and Finkova et
al. (1973) suggest that carbon disulfide concentrations below
10 mg/m3 (3 ppm) may cause disturbances in the ovarian function of
young females, resulting in menstrual disorders, pregnancy
complications, and spontaneous abortions. Given that the exposure
measurements reported are comparable with those of other studies, it
would seem that these would be the earliest detectable type of effects
in human beings.
A summary of the most relevant exposure-effect and exposure-
response findings is given in Table 3.
Table 3. Some exposure-effect and exposure-response relationships
for long-term exposure of man to carbon disulfide.
Concentration Exposure Symptoms & signs Reference
(mg/m3) time
(years)
10 5-10 Blood progesterone depressed, Vasiljeva (1973)
estriol increased, irregular
menstruation;
10 10-15 Sensory polyneuritis, increased Martynova et al. (1976)
pain threshold;
30 >3 Spontaneous abortions twice as Petroy (1969)
common and premature births 3
times as common as in controls;
30-120 >10 Coronary mortality about 5 times Tolonen et al. (1975)
that in controls, life expectancy Hernberg et al. (1973)
decreased 0.9-2.1 years depending Nurminen (1976)
on age; nonfatal infarctions 2.6 Hernberg et al. (1970)
times more frequent than in controls;
angina pectoris about twice as
prevalent as in controls; slightly
higher systolic and diastolic blood
pressure than in controls;
30-120 >6 Peripheral nervous and CNS Seppäläinen & Tolonen
dysfunction, conduction velocities (1974); Tolonen (1974)
slowed, psychological changes;
100-140 2-12 Decrease of testosterone and Cavalleri et al. (1970)
gonadotrophin LH in urine;
200-500 1-9 Ophthalmic pressure 18.4/14.7 Maugeri et al. (1966,
vs 15.3/11.6 kPa (138/110 vs 1967)
115/87 mmHg) in controls;
500-2500 0.5 Polyneuritis, myopathy acute Vigliani (1946)
psychoses
8. CONTROL OF EXPOSURE IN THE VISCOSE INDUSTRY
Technical and administrative measures for the protection of the
health of workers exposed to carbon disulfide in the production of
viscose rayon include:
-- exchange of production methods (substitution or elimination);
-- enclosure and local exhaust ventilation of the technological
process;
-- dilution (general) ventilation of the work room;
-- isolated and well-ventilated rooms for process control
operators and for rest periods;
-- use of personal respiratory protective equipment under certain
hazardous circumstances and,
-- rotation of workers periodically to areas free from carbon
disulfide exposure.
New developments such as the SINI viscose process make it
possible to reduce the total consumption of carbon disulfide to 70-80%
of that used in conventional processes (Sihtola, 1976). Occupational
health aspects must be taken into consideration at the planning stage
in the construction of new plants.
Enclosure of processes and machines is the second most effective
means of technical prevention. This technique should be applied in the
sulfidizing process and for spinning and washing machines. Possible
leakages in closed systems should be controlled by gas detectors. The
air removed from the processes by the exhaust ventilation should be
transported to a recovery system. The benefits of such a system are
both economic, since the plant has an interest in the recovery of
useful material, and hygienic, as the possibility of the pollution of
both the working rooms and the environment around the factory is
reduced. The air removed by exhaust ventilation is compensated with
pure air, which is brought to working sites by ventilation systems.
It is not possible to protect the workers in all situations by
ventilation systems alone. In certain circumstances, the workers may
be exposed to very high concentrations of carbon disulfide, in which
case respiratory protective devices must be used. The type of
respirator may vary from a chemical cartridge (organic vapour)
respirator, used for low concentrations, to supplied air or
self-contained breathing devices for very high concentrations. It is
also important to provide special isolated rooms with a slightly
positive pressure, compared with the work area contaminated with
carbon disulfide, for process control workers and for rest pauses.
Another factor of concern in the control of exposure and uptake
of carbon disulfide involves skin absorption resulting from direct
contact with the liquid. Where workers may come into contact with
liquid carbon disulfide, appropriate protective clothing, such as
synthetic rubber gloves and goggles or face shields, should be worn.
If carbon disulfide is spilled, potential sources of ignition
should be eliminated immediately, spark-proof ventilation provided,
and the spill cleaned up. A small spill should generally be allowed to
evaporate under conditions of good air circulation. However, large
spills should be covered with water and flushed into a retention basin
under a water layer. Carbon disulfide should not be drained into a
sewer system because of the possibility of an explosion.
In addition to these control methods, data obtained through the
biological monitoring of exposed workers can be used to determine
excessive uptake situations that require additional control measures.
The procedure for biological monitoring is discussed in section 6.5.
REFERENCES
ABRAMOVA, J. I. (1967) The questions of pathogenesis, specific
prevention and therapy of carbon disulfide intoxication. In:
Brieger, H. & Teisinger, L, ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 32-34.
ABUCEWICZ, A., SAMOCHOWIEC, L., & SZYSZKA, K. (1971) The role of
5-hydroxytryptamine in the course of experimental carbon
disulfide poisoning. Roca. Pomor. Akad. bled. Szczec, Suppl. 4,
pp. 89-96.
AGRANOVSKIJ, Z. M. & GOLOSCAPOV, O. D. (1972) [Hygienic assessment of
preventive diet for subjects exposed to carbon disulfide.] Gig.
i Sanit., 37 (II): 43-47 (in Russian).
AMERICAN STANDARDS ASSOCIATION (1941) Allowable concentration of
carbon disulfide, New York, ASA, 141, 7 pp. (ASA Z 37.3-1941).
ANDREEVA, N. B. (1970) [Experimental material for the diagnosis of
carbon disulfide poisoning, on the harmful effect of the poison
at the maximum permissible level for workroom air, and on the
passage of carbon disulfide in solution through the skin.] In:
Voprosy diagnostiki, specificeskoj profilaktiki i terapii
professional 'nyh zabolevanij, Leningrad, pp. 40-52 (in
Russian).
ANDRUSZCZAK, K. (1967) [The behaviour of ceruloplasmine level in the
blood serum in workers exposed to the carbon disulfide action.]
Med. Pr., 18:120-127 (in Polish).
ATTINGER, E. (1948) [Chronic carbon disulfide intoxication appearing
under the unusual symptoms of a heavy vascular disease.]
Schwiez. Med. Wochenscht., 78:667-669 (in German).
ATTINGER, E. (1954) [Chronic carbon disulfide intoxication and its
pathogenesis, its early detection and prevention.] Schweiz. Med.
Wochenschr., 84:912-913 (in German).
AUDIO-GIANOTTI, G. B. (1932) [Parkinsonism caused by professional
carbon disulphide poisoning.] Presse med., 40 (July-Dec.)
1289-1291 (in French).
AUSTONI, M. & D'AGNOLO, B. (1957) [Chronic carbon disulfide poisoning
and diabetes mellitus.] Minerva Med., 48:2500-2504 (in
Italian).
BAGON, D. A., CROSBY, M. T., HARDY, H. L., & SIMMONS, J. H. (1973) A
personal sampling technique for simultaneous determination of
hydrogen sulfide and carbon sulfide in air. Ann. occup. Hyg.,
16: 133-139.
BAIKOW, E. K. (1963) [Experimental data on the determination of the
maximum permissible concentration of carbon disulfide and
hydrogen disulfide in the atmosphere in their simultaneous
presence.] Gig. i Sanit., 28 (3): 3-8 (in Russian).
BARD, C. L. (1892) Cases in practice, malignant pustle and insanity
due to bisulphide of carbon. South Cal. Pract., 7: 476-485.
BARILJAK, I. R., VASILEVA, I. A., & KALINOVSKAJA, L. P. (1975) [Effect
of small concentrations of carbon disulfide and hydrogen sulfide
on intrauterine development in rats.] Arkh. Anat. Gistol.
Embriol., 58:77-81 (in Russian).
BARTONICEK, V. (1957) [The distribution of carbon disulfide in whole
blood, the brain and the suprarenal glands over a given period in
parenteral administration in white mice. Prac. Lek., 9:28-30
(in Czech).
BARTONICEK, V. (1958) [The distribution of free carbon disulphide and
bound carbon disulphide liberated by acid hydrolysis in the
organs of white rats.] Prac. Lek., 10: 334-338 (in Czech).
BARTONICEK, V. (1959) [Distribution of free carbon disulfide and bound
carbon disulfide liberated by acid hydrolysis in the organs of
white rats.] Prac. Lek., 10:504-510 (in Czech).
BASHORE, R., HAMILTON, A., LEWEY, F., ERSKINE, L., HAMMOND, J., HOUTZ,
R., BRACELAND, F., MCDONALD, R., BATSON, O., BELLET, S.,
CRESKOFF, F., EHRICH, W., & ALPERS, B. J. (1938) Survey of
carbon disulfide and hydrogen sulfide hazards in the viscose
rayon industry, Harrisburg, PA, Occupational Disease Prediction
Division, US Department of Labor and Industry p. 68 (Bull. 46).
BINET, L. & BOURLIERE, F. (1944) Sur les modifications du sang au
cours du sulfocarbonisme chronique. Arch. Mal. Prof., 6: 12-16.
BITTERSOHL, G. (1971) On relationships in action between carbon
disulfide and hydrogen sulfide. Med. Lavoro, 62: 554-556.
BITTERSOHL, G. & THIELE, H. (1977) The health of workers exposed to
carbon disulphide in the production of artificial fibres. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski (Finland), 6-10 June 1977, p. 5.
BLACKBURN, H., KEYS, A., SIMONSON, E., RAUTAHARJU, P., & PUNSAR, S.
(1960) The electrocardiogram in population studies. A
classification system. Circulation, 21: 1160-1175.
BOBSIEN, K. (1954) [New method for determination of uptake and binding
by endogenous substances.] Arch. Gewerbepath. Gewerbehyg., 13:
193-203 (in German).
BOND, E. J. & DE MATTEIS, F. (1969) Biochemical changes in rat liver
after administration of carbon disulphide, with particular
reference to microsomal changes. Biochem. Pharmacol., 18:
2531-2549.
BOND, E. J., BUTLER, W. H., DE MAITEIS, F., & BARNES, J. M. (1969)
Effects of carbon disulphide on the liver of rats. Br. J. ind.
Med, 26: 335-337.
BRIEGER, H. (1961) Chronic carbon disulfide poisoning. J. occup.
Med., 8: 302-308.
BRIEGER, H. (1967) Carbon disulphide in the living organism-Retention,
metabolism and physiological effects. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 27-31.
BRIEGER, H. & FRIEDMAN, M. H. F. (1949) In vitro effects on
digestive enzymes, theory and pathology of poisoning. J. ind.
Hyg., 31: 96-98.
BRIEGER, H. & TEISINGER, J., ed. (1966) Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 27-31.
BRUCE, A. (1884) Chronic poisoning by CS2. Edinburgh Med. J., 29:
1009.
BÜSING, K. H. (1952) [The determination of minute amounts of carbon
disulfide in blood.] Zentralbl. Arbeitsmed., 2: 115-122 (in
German).
BÜSING, K. H. & SONNENSCHEIN, W. (1954) [The binding of carbon
disulfide in the organs.] Zentralbl. Arbeitsmed. Arbeitschutz,
4:65-69 (in German).
BÜSING, K. H., SONNENSCHEIN, W., BECHER, E. W., & DREIHELLER, H.
(1953) [Experimental studies in animals on the binding of carbon
disulfide in organs using labelled carbon disulfide (C35S2).]
Z. Naturforsch., 86: 495-500 (in German).
CANDURA, F., GRAVERI, A., & STRANEO, G. (1962) [Clinical contribution
to studies on the relationship between arteriosclerosis and
carbon disulfide poisoning.] Folia Med., 45: 749-769 (in
Italian).
CAVALLERI, A. (1972) [Alterations in the endocrinological function as
an early sign of carbon disulfide poisoning.] Med. Lavoro, 63:
81-84 (in Italian).
CAVALLERI, A. (1975) Serum thyroxine in the early diagnosis of carbon
disulfide poisoning. Arch. environ. Health, 30: 85-87.
CAVALLERI, A. & ZUCCATO, F. (1965) [Endocrinological observations on
carbon disulfide poisoning. I: Adrenocortical function.] Folia
Med., 48: 519-527 (in Italian).
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1965) [Adrenocortical and
gonadal function in human carbon disulfide poisoning.] In:
Proceedings of the 28th National Congress on Occupational
Health, Naples, 1965, pp. 645-660 (in Italian).
CAVELLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1966a) Endocrinological findings in young workers
exposed to carbon disulphide. I. - Urinary excretion of total
17-ketosteroids. Med. Lavoro, 57: 566-572.
CAVALLERI, A., DJURIC, D., VISCONTI, E., REZMAN, I., MAUGERI, U., &
BRANKOVIC, D. (1966b) Endocrinological findings in young workers
exposed to carbon disulphide. II.-Urinary excretion of
17:hydroxy-corticosteroids. Med. Lavoro, 57: 573-578.
CAVALLERI, A., DJURIC, D., VISCONTI, E., BRANKOVIC, D., & MAUGERI, U.
(1966c) Androsterone and etiocholanolone in the urine of young
workers exposed to carbon disulphide. In: Proceedings of the XV
International Congress on Occupational Health, Vienna, 19-24,
1966, Vienna, Wiener Med. Akademie, Vol. II-I, pp. 347-353.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., & VISCONTI, E.
(1966d) Endocrinological findings in young workers exposed to
carbon disulphide. III.-Urinary excretion of 3alpha,
11-deoxy-17-ketosteroids. Med. Lavoro, 57: 755-760.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1967) 17-ketosteroids and 17-hydroxycorticosteroids
in the urine of young workers exposed to carbon disulphide. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 86-91.
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1970) L'excretion urinaire
de testosterone et de gonadotrophine stimulant les cellules
interstitielles (ICSH) chez des subjets exposes au sulfure de
carbone. Arch. Mal. Prof., 31: 23-30.
CAVALLERI, A., TACCOLA, A., GRAOVAC-LEPOSAVIC, LJ., MAUGERI, U., &
DJURIC, D. (1971) [Thyroinaemia in subjects exposed to carbon
disulfide.] Lav. Um., 23: 183-187 (in Italian).
CAVALLERI, A., POLATTI, F., & BOLIS, P. F. (1977) Acute effects of
tetraethylthiuram disulphide on serum levels of hypophyseal
hormones in humans. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski (Finland), 6-10
June 1977, p. 28.
CHERVENKA, C. H. & WILCOX, P. E. (1956) Chemical derivative of
chymotrypsinogen: I: Reaction with carbon disulfide. J. biol.
Chem. 222: 621-634.
CIMBAREVIC, T. K. (1970) [Ceruloplasmin activity in persons exposed to
carbon disulfide.] In: Voprosy diagnostiki, specifideskoj
profilaktiki i terapii professional 'nyh zabolevanij,
Leningrad, 65-67 (in Russian).
CIRLA, A.M., VILLA, A., & TOMASINI, M. (1972) [Study on the incidence
of coronaropathy in workers exposed to carbon disulfide in a
viscose rayon industry.] Med. Lavoro, 63: 431-441 (in Italian).
COHEN, A. E., PAULUS, H. J., KEENAN, R. G., & SCHEEL, L. D. (1958)
Skin absorption of carbon disulfide vapor in rabbits. I.
Associated changes in blood protein and zinc. Arch. ind. Health,
17: 164-169.
COHEN, A. E., SCHEEL, L. D., KOPP, J. F., STOCKELL, F. R., KEENAN, R.
G., MOUNTAIN, J. T., & PAULUS, H. J. (1959) Biochemical
mechanisms in chronic carbon disulfide poisoning. Am. Ind. Hyg.
Assoc. J., 20: 303-323.
CREPET, M., COBBATO, F., & SCANSETTI, G. (1956)] Some clinical
observations on vasculopathy resulting from chronic carbon
disulfide poisoning.] Minerva Med., 47: 379-387 (in Italian).
CULLEN, T. E. (1964) Spectrophotometric determination of
dithiocarbamates residues on food crops. Anal. Chem., 36:
221-224.
DALVI, R. R., POORE, R. E., & NEAL, R. A. (1974) Studies of the
metabolism of carbon disulfide by the rat liver microsomes. Life
Sci., 14: 1785-1796.
DANILOVA, T. N. (1968) [Data on the effect of carbon disulfide on the
coagulation system of the blood.] In: Materially 2-oj
konferencii molodyh naucnyh rabotnikov, Leningrad, pp. 102-104
(in Russian).
DAVIDSON, M. & FEINLEIB, M. (1972) Carbon disulfide poisoning: A
review. Am. Heart J., 83: 100-114.
DELIC, V., GRAOVAC-LEPOSAVIC, LJ., DJURIC, D., BERKES, I., &
DJORDJEVIC, V. (1966) The determination of sulphates in the urine
of workers exposed to carbon disulphide. Med. Lavoro, 57:
458-462.
DELPECH, A. (1856a) Notes sur les accidents que developpe, chez les
ouvriers en caoutchouc, l'inhalation du CS2 en vapeur. Gaz.
Hebd. med. Chir., 3: 40.
DELPECH, A. (1856b) Accidents que developpe, chez les ouvriers en
caoutchouc, l'inhalation du CS2 en vapeur. Union bled., 10:
265.
DELPECH, A. (1863) Industrie du caoutchou de souffle: Recherches sur
l'intoxication speciale que determine le CS2. Ann. Hyg. 2e
sér., 19: 65.
DE MATTEIS, F. (1974) Covalent binding of sulfur to microsomes and
loss of cytochrome P-450 during the oxidative desulfuration of
several chemicals. Mol. Pharmacol., 10: 849-854.
DE MATTEIS, E. & SEAWRIGHT, A. A. (1973) Oxidative metabolism of
carbon disulfide by the rat. Effect of treatments which modify
the liver toxicity of carbon disulfide. Chem. biol. Interact.,
7: 375-388.
DEMUS, H. (1964) [On the reception, chemical transformation and
excretion of carbon disulfide by the human body.] Arch.
Gewerbepath. Gewerbehyg., 20: 507-536 (in German).
DEMUS, H. (1967) The mechanism of absorption, metabolism and excretion
of carbon disulfide in the human body. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 42-49.
DE NICOLA, P., NICROSINI, F., FASOTTI, C., & DEL FAVERO, A. (1962)
[Comparative studies on the physiopathological picture of
experimental chronic carbon disulfide poisoning and
high-cholesterol diet I. Study of the lipid fractions in serum
and aorta.] G. Geront., 10: 207-220 (in Italian).
DEPARTMENT OF EMPLOYMENT AND PRODUCTIVITY (1968) Methods for
determination of toxic substances in air: Carbon disulfide
vapour. London, HMSO, 10 pp. (DEP Booklet No. 6).
DJERASSI, L. S. & LUMSROSO, R. (1968) Carbon disulphide poisoning with
increased ethereal sulphate excretion. Br. J. ind. Med., 25:
220-222.
DJURIC, D. (1963) [Metabolism of carbon disulphide.] Arh. Hig. Rada,
14: 23-31 (in Serbo-Croat).
DJURIC, D. (1967) Determination of carbon disulphide and its
metabolites in biological material. In: Brieger, H. & Teisinger,
J., ed. Toxicology of carbon disulphide, Amsterdam, Excerpta
Medica Foundation, pp. 52-61.
DJURIC, D. (1971) Some aspects of antabuse, carbon disulphide and
ethyl alcohol metabolism. Arh. Hig. Rada, 22: 171-177.
DJURIC, D., SURDUCKI, N., & BERKES, I. (1965) Iodine-azide test on
urine of persons exposed to carbon disulphide. Br. J. ind. Med.,
22: 321-323.
DJURIC, D., STOJADINOVIC, LJ., BOJOVIC, V., & REZMAN, I. (1967)
Excretion of zinc in the urine of persons exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation, pp.
118-121.
DJURIC, D., POSTIC-GRUJIN, A., GRAOVAC-LEPOSAVIC, L., & DELIC, V.
(1973) Disulfiram as an indicator of human susceptibility to
carbon disulfide excretion of diethyldithiocarbamate sodium in
the urine of workers exposed to CS2 after oral administration of
disulfiram. Arch. environ. Health, 26: 287-289.
DROGCINA, E. A. (1968) [Carbon disulfide poisoning.] In: Professional
'nye bolezni sistemy, Leningrad, Medicina, pp. 129-141 (in
Russian).
DUTKIEWICZ, T. & BARANOWSKA, B. (1967) The significance of carbon
disulphide skin resorption in the evaluation of exposure. In:
Brieger, H. & Teisinger, J., ed. Toxicology of Carbon
Disulphide, Amsterdam, Excerpta Medica Foundation, pp. 50-51.
EFREMOVA, N. F. & UZBEKOV, G. A. (1968) [Study of nitrogen metabolism
in the brains of white rats exposed to chronic carbon disulfide
poisoning.] In: Trudy Rjazanskogo Medicinskogo Instituta, 34:
163-172 (in Russian).
EHRHARDT, W. (1967) Experiences with the employment of women exposed
to carbon disulphide. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 240-244.
EL-GAZZAR, R., EL-SADIK, Y. M., & HUSSEIN, M. (1973) Changes in zinc
and serum proteins due to carbon disulphide exposure. Br. J.
ind. Med., 30: 284-288.
FAHIM, B. A., FAHMY, A.M., & KAMEL, M. Y. (1973) [Biochemical and
hematologic studies on carbon disulfide poisoning.] Z. Ges.
Hyg., 19: 538-541 (in German).
FAITH, W. L., KEYES, D. B., & CLARK, R. L. (1965) Industrial
Chemicals, 3rd ed., New York, John Wiley & Sons, pp. 226-227.
FERRERO, G. P. (1969) [Occurrence of diabetes in workers exposed to
carbon disulfide.] Med. Lavoro, 60: 38-42 (in Italian).
FERRARO, A., JARVIS, G., & FLICKER, D. (1.941) Neuropathologic changes
in experimental carbon disulfide poisoning in cats. Arch.
Pathol., 32: 732-738.
FINKOVA, A., SIMKO, A., JINDRICHOVA, J., KOVARIK, J., PREININGEROVA,
O., KLIMOVA, A., & KORISKO, F. (1973) [Gynaecological problems of
women working in an environment contaminated with carbon
disulfide.] Cesk. gynekol., 38: 535-536 (in Czech).
FINULLI, M. & GHISLANDI, E. (1959) [Occurrence of diabetes in chronic
carbon disulfide poisoning.] Med. Lavoro, 50: 189-192 (in
Italian).
FISCHER, J. & MICHALOVA, C. (1956) [Neurohistological changes caused
by carbon disulfide.] Prac. Lek., 8:16-19 (in Czech).
FOA, V., CASSITO, M. G., FORZI, M., & BULGHERONI, C. (1976) Mental
performance and personality disorders among workers exposed to
carbon disulphide: Comparison between two different rayon plants.
In: Horvarth, M., ed. Adverse effects of environmental chemicals
and psychotropic drugs, neurophysiological and behavioral tests.
Amsterdam, Oxford, New York, Elsevier, Vol. 2, pp. 173-182.
FOREMAN, W. (1886) Notes of a fatal case of poisoning by bisulphide of
carbon. Lancet, 2: 118-119.
FRANCINE, A. P. (1905) Acute carbon disulfide poisoning. Am. Med.,
9: 871.
FRANTIK, E. (1970) The development of motor disturbances in
experimental chronic carbon disulphide intoxication. Med.
Lavoro,, 61: 309-313.
FREUNDT, K. J. (1973) The inhibitory action of inhaled carbon
disulfide on microsomal enzymes as a sensitive restriction of
liver function. In: Horvath, M., ed. Adverse effects of
environmental chemical and psychotropic drugs: Quantitative
interpretation of functional tests, Amsterdam, Oxford, New
York, Elsevier, pp. 161-164.
FREUNDT, K. J. (1974) Ethanol and acetaldehyde levels in alcoholized
rats and man under carbon disulphide exposure. Abstracts of the
III International Symposium on the Toxicology of Carbon
Disulphide, Cairo-Alexandria (Egypt) 4-9 May, 1974, p. 28.
FREUNDT, K. J. (1975) [Carbon disulfide.] In: Henschler, D., ed.
Gesundheitsschadliche Arbeitstoffe, 4th ed., Federal Republic
of Germany, Verlag Chemic Weinheim, pp. 1-20.
FREUNDT, K. J. (1977) Animal experimentation studies on a possible
induction of hepatic microsomal mixed-function oxygenases (MFO)
by inhaled carbon disulphide (CS2). In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 24.
FREUNDT, K. J. & DREHER, W. (1969) Inhibition of drug metabolism by
small concentrations of carbon disulfide. Nauyn-Schmiedebergs
Arch. Pharmakol., 263: 208-209.
FREUNDT, K. J. & HENSCHLER, D. (1971) Inhibition of oxidative drug
metabolism by low doses of inhaled carbon disulfide: Kinetics and
specificity. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Bahja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 9.
FREUNDT, K. J. & KÜRZINGER, R. (1975) Energy potential and hepatic
function in rats under acute exposure to carbon disulphide. Int.
Arch. Arbeitsmed., 34: 269-282.
FREUNDT, K. J. & KUTTNER, P. (1969) Oxidative drug metabolism
following low carbon disulphide concentrations.
Nauyn-Schmiedebergs Arch. Pharmakol., 264: 232-233.
FREUNDT, K. J. & LIEBERWIRTH, H. (1974) Influence of inhaled carbon
disulfide on acetaldehyde production and liver function in
alcoholized man. Nauyn-Schmiedebergs Arch. Pharmakol., 282
(Suppl. R.21).
FREUNDT, K. J. & NETZ, H. (1973) Influence of carbon disulfide and
thiurams on ethanol elimination and acetaldehyde production.
Arch. Pharmakol. exp. Pathol., 277 (Suppl. R18).
FREUNDT, K. J., LIEBALDT, G. P., & SIEBER, K. H. M. (1974a) Effect of
barbiturates on the liver of rats exposed to carbon disulphide
vapor. Int. Arch. Arbeitsmed., 32: 297-303.
FREUNDT, K. J., SCHAUENSURG, K. J., & EICHHORN, P. (1974b) Effect of
acute exposure to carbon disulfide vapor upon some components of
the hepatic microsomal enzyme system in rats. Arch. Toxicol.,
32 (3): 233-240.
FREUNDT, K. J., SCHNAPP, E., & DREHER, W. (1975) Pharmacokinetics of
inhaled carbon disulphide in rats in relation to its inhibitory
effect on the side-chain oxidation of hexobarbital. Int. Arch.
occup. environ. Health, 35: 173-185.
FREUNDT, K. J., LIEBERWIRTH, K., NETZ, H., & POHLMANN, E. (1976) Blood
acetaldehyde in alcoholized rats and humans during inhalation of
carbon disulphide vapor. Int. Arch. occup. environ. Health, 37:
35-46.
FRUSCELLA. R. (1962) [Urinary excretion of 17-ketosteroids and
17-hydroxycorticosteroids in human subjects suffering from carbon
disulfide poisoning.] Clin. ter., 23: 749-755 (in Italian).
GADASKINA, I.D. & ANDREEVA, N. B. (1960) [Some biochemical shifts in
the organism in carbon disulfide poisoning. Free SH groups and
ceruloplasmin in the blood: Copper and zinc metabolism.] Gig.
Tr. Prof Zabol., 13: 28-32 (in Russian).
GAVRILESCU, N. & LILIS, R. (1967) Cardiovascular effects of
long-extended carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 165-167.
GHERASE, G. (1976) Psychological changes in workers exposed to
atmospheres containing carbon disulphide. In: Adverse effects of
environmental chemicals and psychotropic drugs.
Neurophysiological and behavioral tests, Amsterdam, Vol. 2,
334 pp.
GILIOLI, R., BULGHERONI, C., BERTAZZI, P. A., CIRLA, A. M., CASSITTO,
M. G., & TOMASINI, M. (1977) Endocrinological, psychological and
neurological study of carbon disulphide workers. III.
Neurological and neurophysiological findings. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), June 6-10, 1977, p. 13.
GOLDBERG, M. E., JOHNSON, H. E., POZZANI, U. C., & SMYTH, H. F. (1964)
Behavioral response of rats during inhalation of trichlorethylene
and carbon disulphide vapours. Acta Pharmacol., 21: 36-44.
GONDZIK, M. (1971) Histology and histochemistry of rat testicles as
affected by carbon disulphide. Polish med. J., 10: 133-139.
GORDY, S. T. & TRUMPER, M. (1938) CS2 poisoning with a report of six
cases. J. Am. Med. Assoc., 110: 1543.
GORDY, S. T. & TRUMPER, M. (1940) Carbon disulphide poisoning-Report
of 21 cases. Ind. Med., 9: 231-234.
GORNY, R. (1971) The level of pyridoxal phosphate (PLP) in the blood
plasma of rats exposed to carbon disulfide. Biochem. Pharmacol.,
20: 2114-2115.
GORNY, R. (1974) [Effect of prolonged exposure to carbon disulfide on
tryptophan metabolism in rats.] Med. Pr., 25: 21-27 (in
Polish).
GOTO, S. & HOTTA, R. (1967) The medical and hygienic prevention of
carbon disulphide poisoning in Japan. In: Brieger, H. &
Teisinger, J.: ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 219-230.
GOTO, S., HOTTA, R., & SUGIMOTO, K. (1971) Studies on chronic carbon
disulfide poisoning. Int. Arch. Arbeitsmed., 28: 115-126.
GOTO, S., SUGIMOTO, K., HOTTA, R., BABA, T., NAKAMURA, G., & SEKI, Y.
(1977) Regional patterns of angiopathy due to CS2. Egypt. J.
occup. Med., 5: 155-159.
GRAOVAC-LEPOSAVIC, LJ. & JOVICIC, M. (1971) Report on one case of
chronic carbon disulfide intoxication. In: Abstracts of the H
International Symposium on Carbon Disulphide Toxicology, Banjo
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 19.
GRAOVAC-LEPOSAVIC, LJ., DJURIC D., PAVLOVIC, A., & JOVICIC, M. (1967)
The use of iodine-azide test for early diagnosis of carbon
disulphide poisoning. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 62-69.
GRAOVAC-LEPOSAVIC, LJ., JOVICIC, M., SAVIC, S., DJURIC, D., &
POSTIC-GRUJIN, A. (1977) Findings in workers some years after
CS2 poisoning. Egypt. J. occup. Med., 5: 53-60.
GREGORCZYK, J., KOSMIDER, K., WIECKOWSKI, B., & WESOLOWSKA, T. (1975a)
Investigations on the extent of the liver lesion in the rat by
experimental inhalation poisoning with carbon disulfide. I.
Analysis of biochemical changes in the blood serum. Int. Arch.
Arbeitsmed., 34: 65-80.
GREGORCZYK, J., WIECKOWSKI, B., & WESOLOWSKA, T. (1975b)
Investigations on the extent of liver lesion in rats by
experimental inhalation poisoning with carbon disulfide. II.
Analysis of biochemical changes in liver homogenate and its
fractions. Int. Arch. Arbeitsmed., 34: 151-166.
GUARINO, A. & ARCIELLO, G. (1954) [Histological study on coronary
arteries in chronic experimental carbon disulfide poisoning.]
Folia Med. (Napoli) 37: 1022-1044 (in Italian).
HAKKY, U. (1977) Physiological manifestations after exposure to CS2.
Egypt. J. occup. Med., 5: 47-52.
HAMILTON, A. (1925) Industrial poisons in the United States, New
York, Macmillan Co., 590 pp.
HAMILTON, A. (1940) Occupational poisoning in the viscose rayon
industry, Washington, DC, Department of Labor, pp. 1-79 (Bull.
No. 34).
HÄNNINEN, H. (1971) Psychological picture of manifest and latent
carbon disulphide poisoning. Br. J. ind. Med., 28: 374-381.
HÄNNINEN, H. (1974) Behavioral study of the effects of carbon
disulfide. In: Xintaras, C. El, Johnson, B., De Groot, I., ed.
Behavioral toxicology, Washington, DC, Department of Health,
Education, & Welfare, pp. 72-79.
HÄNNINEN, H., NURMINEN, M., TOLONEN, M., & MARTELIN, T. (1978)
Psychological tests as indicators of excessive exposure to carbon
disulfide. Scand. J. Psychol., 19(2): 163-174.
HARA, I. (1958) [On the carbohydrate metabolism in chronic carbon
disulfide poisoning.] Q. J. labour Res. Jpn, 6: 1-20 (in
Japanese).
HARASHIMA, S. & MASUDA, Y. (1962) Quantitative determination of
absorption and elimination of carbon disulfide through different
channels in human body. Int. Arch. Gewerbepath. Gewerbehyg.,
19: 263-269.
HARASHIMA, S., TOYAMA, T., & SAKURAI, T. (1960) Serum cholesterol
level of viscose rayon workers. Keijo J. Med., 9: 81-90.
HASSMAN, P., SIMKO, A., JINDRICHOVA, J., HEROUT, V., & HRADSKY, M.
(1967) The problem of chronic gastritis in artificial fibres
industry. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation,
pp. 182-189.
HERNBERG, S. & NORDMAN, C-H. (1969) Failure to find increased
excretion of zinc or magnesium in humans exposed to carbon
disulphide. Med. Lavoro, 60: 163-168.
HERNSERG, S., MOWE, G., VIRKOLA, P., PARTANEN, T., & NORDMAS, C-H.
(1969) Magnesium and zinc values of erythrocytes and plasma for
workers exposed to carbon disulfide. Work Environ. Health, 6:
9-13.
HERNSERG, S., PARTANEN, T., NORDMAN, C-H., & SUMARI, P. (1970)
Coronary heart disease among workers exposed to carbon
disulphide. Br. J. ind. Med., 27: 313-325.
HERNBERG, S., NORDMAN, C-H., PARTANEN, T., CHRISTIANSEN, V., &
VIRKOLA, P. (1971) Blood lipids, glucose tolerance and plasma
creatinine in workers exposed to carbon disulphide. Work
Environ. Health, 8: 11-16.
HERNBERG, S., NURMINEN, M., & TOLONEN, M. (1973) Excess mortality from
coronary heart disease in viscose rayon workers exposed to carbon
disulfide. Work Environ. Health, 10: 93-99.
HERNBERG, S., TOLONEN, M., & NURMINEN, M. (1976) Eight-year follow-up
of viscose rayon workers exposed to carbon disulfide. Scand. J.
Work Environ. Health, 2: 27-30.
HIGASHIDA, T., YAMAMOTO, T., & SHINDO, S. (1959) [Electromyographic
study of the effects of carbon disulfide administration on spinal
reflex.] Brain Nerve, 11(4): 253-260 (in Japanese).
HOCKWIN, O. & SAVIC, S. (1968) [Reversibility of biochemical changes
in lease and retina of the rabbit's eye in carbon disulfide
poisoning.] Albrecht Graefs Arch. klin. exp. Ophthalmol., 175:
7-12 (in German).
HOLASOVA, P. (1969) [Observations on a group of children from an area
polluted by carbon disulfide and hydrogen sulfide exhalation
compared with a control group of children.] Cs. Hyg., 14:
260-265 (in Czech).
HORVATH, M. & MIKISKA, A. (1957) [The influence of carbon disulfide on
the CNS of rabbits as seen in the EEG.] Prat. Lek., 9: 22-28
(in Czech).
HOTTA, R. & GOTO, S. (1971) A fluoroscein angiographic study on
microangiopathia sulfocarbonica Jpn. J. Ophthal., 15: 132-139.
HOTTA, R. & SAVIC, S. (1972) Fluorescein angiographic and
ophthalmodynamometric study on chronic carbon disulfide
poisoning. Med. Lavoro, 63: 442-447.
HUEPER, M. C. (1936) Etiologic studies on the formation of skin
blisters in viscose workers. J. ind. Hyg., 18: 432-447.
IVANOVA, F. G. (1967) [Some data on the status of erythropoiesis on
exposure to low concentrations of carbon disulfide.] In: Voprosy
gigieny truda i profpatologii, Leningrad, pp. 247-248 (in
Russian).
JAKUBOWSKI, M. (1968) [Studies of the conversion of carbon disulphide
to compounds catalysing the iodine-azide reaction. I.
Chromatographic studies.] Med. Pr. 19: 244-251 (in Polish).
JAKUBOWSKI, M. ( 1971) [Studies on the carbon disulfide metabolism of
compounds catalyzing the iodine azide reaction.] Med. Pr. 22:
195-206 (in Polish).
JÄRVISALO, J. & DE MATTEIS, F. (1977) Carbon disulphide induced loss
of liver microsomal haem and cytochrome P-450. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 23.
JÄRVISALO, J., SAVOLAINEN, H., ELOVAARA, E., & VAINIO, H. (1977) The
in vivo toxicity of CS2 to liver microsomes: Binding of
labelled CS2 and changes of the microsomal enzyme activities.
Acta Pharmacol. Toxicol., 40: 329-336.
JUMP, H. D. & CRUICE, J. M. (1904) Chronic poisoning from bisulphide
of carbon. Univ. Penn. med. Bull., 17: 193-196.
JUNTUNEN, J., HALTIA, M., & LINNOILA, I. (1974) Histochemically
demonstrable non-specific cholinesterase as an indicator of
peripheral nerve lesion in carbon disulfide induced
polyneuropathy. Acta Neuropathol. (Berl.), 29: 361-366.
JUNTUNEN, J., LINNOILA, I., & HALTIA, M. (1977) Histochemical and
electron microscopic observations on the myoneural junctions of
rats with carbon disulfide induced polyneuropathy. Scand. J.
Work Environ. Health, 3: 1-15.
KARAJOVIC, D., GRAOVAC-LEPOSAVIC, LJ., & SURADNICI, V. J. (1964) [The
influence of CS2 on the health of workers in factory in Serbia.]
Arh. Hig. Rada, 15: 87-92 (in Serbo-Croat).
KLIMKOVA-DEUTSCHOVA, E. (1965) [Early diagnosis in industrial
neurology.] S.Z.N., Praha 1965, 260 pp. (in Czech).
KNAVE, B., KOLMODIN-HEDMAN, B., PERSSON, H. E., & GOLDBERG, J. M.
(1974) Chronic exposure to carbon disulfide: Effects on
occupationally exposed workers with special reference to the
nervous system. Work Environ. Health, 11: 49-58.
KOELSCHS HANDBUCH DER BERUFSERKRANKUNGEN (1972) Jena, Gustav Fischer
Verlag. Vol. II, pp. 270-275.
KOESTER, G. (1899) [Contribution to the field of chronic CS:
intoxication.] Arch. Psychiat., 32: 606 (in German).
KOLMODIN-HEDMAN, B. (1976) Iodine-azide test in a Swedish viscose
factory related to air concentration of CS2 and to health.
Arbete och Halea, No. 8. pp. 23-28.
KOMRSKA, M. (1967) L'importance de la rheoencephalographie pour le
diagnostic d'intoxication par le sulfure de carbone. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulfide,
Amsterdam, Excerpta Medica Foundation, pp. 140-144.
KOPECKY, J. (1973) [Metabolism of carbon disulfide in rats.] Acta
Hyg. Epidemiol. Microbiol. (Prague), Suppl. 12, pp. 3-6
(in Czech).
KOZIOL, A., KULCZYCKA, H., & ROZEWICKI, S. (1971) [Development of
children, whose mothers were professionally exposed to carbon
disulfide.] Med. Pr., 22: 511-514 (in Polish).
KROLIKOWSKA, W. & RUCINSKA, Z. (1959) EEG examinations in chronic
professional intoxication by carbon disulfide. Electrortceph.,
clin. Neurophysiol., 11: 180 (Abstract No. 9).
KUBOTA, J. (1967) Historical view of carbon disulfide poisoning in the
Japanese viscose rayon industry. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulfide, Amsterdam, Excerpta Medica
Foundation, pp. 192-196.
KUJALOVA, V. (1971) The effect of carbon disulfide on vitamin B6
metabolism. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Banja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 30.
KUJALOVA, V. (1973) [Changes in the pyridoxinemetabolism and in the
blood levels of ceruloplasmin in humans exposed to carbon
disulfide.] Acta Hyg. Epidemiol. Microbiol., Suppl. 12 (in
Czech).
KUJALOVA V. & TINTERA, J. (1970) The effect of carbon disulfide on
tryptophan metabolism. Med. Lavoro, 61: 227-231.
KÜRZINGER, R. & FREUNDT, K. J. (1969) Changes in hepatic functions
following low carbon disulfide concentration.
Naunyn-Schmiedebergs Arch. Pharmakol., 264: 261-262.
LANCRANJAN, I. (1972) Alterations of spermatic liquid in patients
chronically poisoned by carbon disulfide. Med. Lavoro, 63:
29-33.
LANCRANJAN, I., POPESCU, H. I., & KLEPSCH, I. (1969) Changes of the
gonadic function in chronic carbon disulfide poisoning. Med.
Lavoro, 60: 566-571.
LANCRANJAN, I., FLOREA, O., & PETRESCU, M. (1971) Corticosuprarenal
insufficiency as a feature of carbon disulfide poisoning. Med.
Lavoro, 62: 557-561.
LANCRANJAN, I., SUKMANSKY, M., STANUCA, L., ANTONESCU, C., & POPESCU,
H. I. (1972) Study of the thyroid function in chronic carbon
disulfide poisoning. Med. Lavoro, 63: 123-125.
LANGELEZ, A. (1946) Viscose et sulfure de carbone. Arch. Belg. Med.
Soc. Hyg. Rev. Pathol., 4: 67-85.
LAUDENHAIMER, R. (1899) [The carbon disulfide intoxication of rubber
workers.] Leipzig, Veit & Co., 168 pp. (in German).
LAURMAN, W. & WRONSKA-NOFER, T. (1977) [Effect of carbon disulfide on
the amount of cholesterol in the tissues of rats.] Med. Pr.,
28: 77-82 (in Polish).
LAZAREV, N.J., ABRAMOVA, ZH. I., & CERNY, Z. C. (1965) [On possible
adverse reactions following use of some drugs by persons handling
carbon disulfide.] Gig. Tr. Prof. ZaboL, 9: 24-27 (in Russian).
LEITHE, W. (1971) Determination of carbon disulfide at the work site.
The analysis of air pollutant, 2nd ed., Ann Arbor, MI, Humphrey
Science Pub., pp. 228-229.
LEWEY, F. H. (1941) Neurological, medical and biochemical signs and
symptoms indicating chronic industrial carbon disulfide
absorption. Ann. int. Med., 15: 869-883.
LEWEY, W. E., ALPERS, B. J., BELLET, S., GRESKOFF, A. J., DRABLEIN, D.
L., ERICH, W. E., FRANK, J. H., JONAS, L., MCDONALD, R.,
MONTGOMERY, E. J., & REINHOLD, J. G. (1941) Experimental chronic
poisoning in dogs, clinical, biochemical and pathological study.
J. ind. Hyg., 23: 415-436.
LILIS, R. (1974) Behavioral effects of occupational carbon disulfide
exposure. In: Xintaras, C., Johnson, B. L., & de Groot, I., ed.
Behavioral toxicology, early detection of occupational hazards,
Cincinnati, US DHEW, Public Health Service, Center for Disease
Control, National Institute for Occupational Safety and Health,
pp. 51-59 (NIOSH 74-126).
LINIECKI, J. (1960) The effect of chronic carbon disulfide poisoning
on thiamine balance in rats. J. Hyg. Epidemiol. Microbiol.
Immunol. 4: 212-222.
LINNOILA, I., HALTIA, M., SEPPÄLÄINEN, A.M., & PALO, J. (1975)
Experimental carbon disulphide poisoning. Morphological and
neurophysiological studies. In: Körney, S., Tariska, S., &
Gosztonyi, G., ed. Proceedings of the VII International Congress
on Neuropathology Académiai Kiado, Budapest 1-7 September 1975,
pp. 383-386.
LUKAS, E. (1969) Conduction speed of peripheral nerves in persons
exposed to carbon disulfide (Electromyography study). Int. J.
clin. Ther. Toxicol., 2: 354-358.
LUKAS, E. (1970) Stimulation electromyography in experimental
toxicology (Carbon disulphide neuropathy in rats). Med. Lavoro,
61: 302-308.
LUKAS, E. (1973) [Some important factors in the development of
experimental carbon disulfide neuropathy in rats.] Cs. Neurol.
Neurochir., 36: 169-173 (in Czech).
LUKAS, E. (1974) Copper and zinc levels in peripheral nerve tissues of
rats with experimental carbon disulfide neuropathy. In:
Abstracts of the III International Symposium on Toxicology of
Carbon Disulphide, Cairo-Alexandria, Egypt, 4-9 May, 1974,
p. 8.
LUKAS, E. & HROMADKA, M. (1977) Conduction velocity of the ulnar and
tibial nerves in workers occupationally exposed to low
concentrations of carbon disulphide. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, June 6-10, 1977, p. 18.
LUKAS, E., KOTAS, P, & OBRUSNIK, I. (1974) Copper and zinc levels in
peripheral nerve tissues of rats with experimental carbon
disulphide neuropathy. Br. J. ind. Med., 31: 288-291.
LUKAS, E., KUSALOVA, V., & KOCINOVA, F. (1975) Activity of
ceruloplasmin in the serum of rats with experimental carbon
disulphide neuropathy. In: McCallum, R. I., ed. Abstracts of the
XVIII International Congress on Occupational Health, Brighton,
England, 14-19 September, 1975, pp. 299-300.
LUKAS, E., TEISINGER, J, Jr, & BASS, A. (1977) Activity of muscle
enzymes in rats with experimentally developed carbon disulphide
polyneuropathy. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 19.
LYSINA, G. G. (1967) Some changes in the internal organs due to the
effects of carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 179-181.
MACK, T., FREUNDT, K. J., & HENSCHLER, D. (1974) Inhibition of
oxidative N-demethylation in man by low doses of inhaled carbon
disulphide. Biochem. Pharmacol., 23: 607-614.
MADLO, Z. & SOUCEK, B. (1953) [Absorption, metabolism, and action of
carbon disulfide in the organism. VII. Inhibition of serum
cholinesterase by carbon disulfide.] Prac. Lek., 6: 312-313 (in
Czech).
MADLO, Z., PEIZKER, & SOUCEK, B. (1953) [The absorption, metabolism
and action of carbon disulphide in the organism. V. The effect of
carbon disulfide on tissue respiration.] Prac. Lek., 5: 263-266
(in Czech).
MAGISTRETTI, M. & PEIRONE, E. (1961)The action of carbon disulphide on
cerebral monoamine oxidase. Med. Lavoro, 52: 1-10.
MAGOS, L. (1972) Relevancy of bivalent sulphur excretion to carbon
disulphide exposure in different metabolic conditions. Br. J.
ind. Med., 29: 90-94.
MAGOS, L. (1975) The clinical and experimental aspects of carbon
disulphide intoxication. Rev. environ. Health, 11: 65-80.
MAGOS, L. & BUTLER, H. (1972) Effect of phenobarbitone and starvation
on hepatotoxicity in rats exposed to carbon disulfide vapor.
Br. J. ind. Med., 29: 95-98.
MAGOS, L. & JARVIS, J. A. E. (1970a) The effects of carbon disulphide
exposure on brain catecholamines in rats. J. Pharm. Pharmacol.,
29: 936-938.
MAGOS, L. & JARVIS, J. A. E. (1970b) The effects of carbon disulfide
exposure on brain catecholamines in rats. Br. J. Pharmacol.,
39: 26-33.
MAGOS, J., GREEN, A., & JARVIS, J. A. E. (1974) Half life of CS2 in
rats in relation to its effect on brain catecholamines. Int.
Arch. Arbeitsmed., 32: 289-296.
MANU, P, LANCRANJAN, I., & ZAMFIRESCU, Z. (1970) Investigation of
serum LDH and its isoenzymatic model in carbon disulphide
polyneuritis. Int. Arch. Arbeitsmed., 26: 45-49.
MANU, P., LILIS, R., NESTORESCU, B., LANCRANJAN, I., ROSENTA, A.,
IONESCU, S., & KAUFMAN, G. (1971) Effects of carbon disulfide on
cholesterol and ß-lipoprotein levels in young men. In: Abstracts
of the II International Symposium on Toxicology of Carbon
Disulfide, Banja Koviljaca, Yugoslavia, 25-28 May, 1971, pp.
28-29.
MARTINO, P., COSCIA, G. C., & PERRELLI, G. (1963) [Lipedaemia in
carbon disulfide poisoning. Preliminary observations.[ Folia
Med., 46: 1110-1114 (in Italian).
MARTINO, P., PERRELLI, G., & COSCIA, G. C. (1964) [Lipedaemia in
carbon disulfide poisoning.[ Arch. Sci. Med., 117: 124-130 (in
Italian).
MARTYNOVA, A. P., ZELENKIN, A. N., GOLJAKOVA, L. P., MILKOV, L. E.,
GRODETSKAJA, N. S, & AGADZANOVA, A. A. (1976)[Clinical hygienic
and experimental investigations of the action on the body of
small concentrations of carbon disulfide.] J. Hyg. Sanit., 5:
25-28 (in Russian).
MASSOUD, A. E. E., SADEK, S., EL HAWARY, M. F., & HASSANEIN, M. (1971)
Changes in the electrocardiogram and lung function in workers
exposed to carbon disulfide. In: Abstracts of the II
International Symposium on Toxicology of Carbon Disulfide, Banja
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 6.
MASSOUD, A. A. E., ABDEL KARIM, A. H., & FARES, R. (in press) Serum
thyroxine, serum cholesterol and its fractions in workers exposed
to carbon disulphide. Egypt. J. occup. Med.
MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., VISCONTI, E., IOVICIC, M., &
CAVALLERI, A. (1966a) [Cerebrovascular damage in young workers
exposed to carbon disulfide.] Med. Lavoro, 57: 701-708 (in
Italian).
MAUGERI, U., IOVICIC, M., CAVALLERI, A., GRAOVAC-LEPOSAVIC, LJ.,
VISCONTI, E., & REZMAN, I. (1966b) [Peripheral vascular damage in
young workers exposed to carbon disulfide.] Med. Lavoro, 57:
709-714 (in Italian).
MAUGERI, U., VISCONTI, E., & CAVALLERI, A. (1966c)] Vascular
alterations in the retina of young workers exposed to carbon
disulfide.] Med. Lavoro, 57: 741-745 (in Italian).
MAUGERI, U., CAVALLERI, A., & VISCONTI, E. (1966d)
[Ophthalmodynamography in occupational carbon disulfide
poisoning.] Med. Lavoro, 57: 730-740 (in Italian).
MAUGERI, U., CAVELLERI, A., & VISCONTI, E. (1967)
Ophthalmodynamographic study in young workers exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Excerpta Medica Foundation, Amsterdam, pp.
150-152.
MAUGERI, CAVALLERI, A., MAUGERI, U., & TACCOLA, A. (1971) New aspects
in the pathogenetic mechanism of carbon disulphide toxic action.
Med. Lavoro, 62: 398-403.
MCCAMMON, C., Jr, QUINN, M. P., & KUEPEL, R. E. (1975) A charcoal
sampling method and a gas chromatographic analytical procedure
for carbon disulfide. Am. Ind. Hyg. Assoc. J., 36: 618-625.
MCKEE, R. W. (1941) Solubility of carbon disulphide vapor in body
fluids and tissues. J. ind. Hyg., 23: 484-489.
MCKENNA, M. J. & DI SRTEFANO, V. (1975) A proposed mechanism of the
action of carbon disulfide on dopamine-ß-hydroxylase. Toxicol.
appl. Pharmacol., 33: 137.
MELISSINOS, J.-C. & JACOBIDES, G. (1967) Sur quelques formes nerveuses
d'intoxication observées en Grece chez des sujets exposeés au
sulfure de carbone. Arch. Mal. Prof., 28: 605-611.
MERLEVEDE, E. (1951) [Toxicology of carbon disulfide.] Sinte
Catharina, Brugge, 180 pp. (in Dutch).
MIHAIL, G., MIHAILA, D., BRANISTEANU, D., BODNAR, J., & CIOATA, V.
(1968) Manifestations cliniques, fonctionnelles et biochimiques
des stades precoces du sulfocarbonisme. Arch. Mal. Prof., 29:
109-120.
MILIC, S. (1965) [Some observations concerning the determination of
CS2 in the urine of exposed workers.] In: Zbornik radova o
toksikologiji CS2 (Proceedings in Yugoslavian symposium on CS2
toxicology, Loznica, 1965, p. 41-42 (in Serbo-Croat).
MILKOV, L. E., MONAENKOVA, A.M., BYALKO, N. K., GLOTOVA, K. V.,
VERETINSKAJA, A. G., ALI-ZADE, K. A., MAKARENKO, Y. L.,
NIKOLSKAJA, O. I., & KURSAKOV, A. V. (1970) [On the initial forms
of chronic carbon disulfide poisoning.] Gig. Tr. Prof. Zabol.,
14: 28-32 (in Russian).
MINDEN, H., GOHLKE, R., & ROTHE, R. (1967) [Contribution to the
pathogenesis of chronic carbon disulfide poisoning.] Z. Ges.
Hyg. Grenz., 13: 815-822 (in German).
MISIAKIEWICZ, Z., SZULINSKA, O., & CHYBA, A. (1972) [Influence of a
carbon disulfide, hydrogen sulfide mixture in air on white rats
under the conditions of permanent exposure.] Rocz. Panstw. Zakl.
Hig., 23: 465-475 (in Polish).
MOREO, S. & CANDURA, F. (1960) [The behaviour of serum-clotting
factors in animals treated with sodium arsenite, carbon
disulfide, cadmium chloride and lead acetate.] Boll. Soc. Med.
Chir., (Pavia), 74: 447-455 (in Italian).
MOWÉ, G. (1971) Coronary heart disease and occupational exposure to
carbon disulfide. In: Abstracts of the III International
symposium on toxicology of carbon disulfide, Banja Koviljaca,
Yugoslavia, 25-28 May, 1971, p. 7.
MURASKO, V. A. (1975) [The morbidity of the digestive organs through
working in contact with carbon disulfide in the viscose
industry.] Vrach. Delo, No. 4, pp. 113-117 (in Russian).
NAKAMURA, K., HARADA, M., TATETSU, S., MIYAGAWA, T., TSUKAYAMA, T.,
KAWANO, K., KABASHIMA, K., HIRATA, M., & TOYA, G. (1974) A
clinical study on chronic carbon disulfide poisoning. Psychiatr.
Neuro. Jpn., 76: 243-273.
NEAL, R. A., KATAMAKI, T., HUNTER, A. L., & CATIGNANI, G. (1976)
Monooxygenase catalyzed activation of thiono-sulfur-containing
compounds to reactive intermediates. Hoppe-Seyler's Z. physiol.
Chem., 357: 1044.
NESSWETHA, L. & NESSWETHA, W. (1967) The clinical evaluation of
compensated carbon disulphide intoxications in the German Federal
Republic since 1948. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Excerpta Medica Foundation,
Amsterdam, pp. 214-218.
NIOSH (1977) Criteria for a recommended standard...occupational
exposure to carbon disulfide, Washington, DC, US Dept Health,
Education, and Welfare, National Institute for Occupational
Safety and Health, 186 pp.
NOFER, J. & WRONSKA-NOFER, T. (1966) Elimination des certains
metabolites de niacine par rapport au témps dexposition et la
close sulfure de carbone. In: Proceedings of the IX
International Congress on Occupational Health, Vienna, 1966,
pp. 355-358 (A III 53).
NOFER, J., CHOJNOWSKI, J., KAWECKA, M., KIEC, E., WRONSKA-SZPAKOWA,
T., & WYSZOMIRSKA, Z. (1961) [Effect of occupational hazard to
carbon disulphide on the pathogenesis of arteriosclerosis.]
Med. Pr. 12: 101-118 (in Polish).
NORO, L. (1944) [Hydrogen and carbon disulphide intoxications.] Nord.
Med., 24: 2015-2020 (in Swedish).
NOVAK, L., DJURIC, D., & FRIEDMAN, V. (1968) Specificity of the
iodine-azide test for carbon disulphide exposure. I. Urinary
excretion of sulfur compounds in persons treated with disulfiram.
Arch. environ. Health, 19: 473-477.
NUNZIATE, CESARO, A., SIPALA, E., & ZUNINI, E. (1952) [Carbon
disulfide poisoning.] Folia Med., 35: 661-716 (in Italian).
NURMINEN, M. (1976) Survival experience of a cohort of carbon
disulphide exposed workers from an eight-year prospective
follow-up period. Int. J. Epidemiol., 5: 179-185.
OLIENACZ, W., PLAMIENIAKA Z., SZYMANKA, J. (1964) [Excretion of
17-ketosteroids in chronic carbon disulphide poisoning.]
Med. Pt. 15: 155-157 (in Polish).
PATERNI, L., PUSIC, G., & TEODORI, S. (1958) [Chronic carbon disulfide
poisoning and arteriosclerosis from high-cholesterol diet in
rabbits.] Folia Med., 41: 705-722 (in Italian).
PAULUS, H. J., LIPPMAN, M., & COHEN, A. E. (1957) Evaluation of
potential health hazards in fumigation of shelled corn with a
mixture of carbon disulfide and carbon tetrachloride. Am. Ind.
Hyg. Assoc. J., 18: 345-350.
PERGAL, M., VUKOJEVIC, N., GIRIN-POPOV, N., DJURIC, D., & BOJOVIC, T.
(1972a) Carbon disulfide metabolites excreted in the urine of
exposed workers. I. Isolation and identification of
2-mercapto-2-thiazolinone-5. Arch. environ. Health, 25: 38-41.
PERGAL, M., VUKOJEVIC, N., & DJURIC, D. (1972b) II. Isolation and
identification of thiocarbamide. Arch. environ. Health, 25:
42-44.
PERGAL, M. N., KOBILAROV, J., RALETIC, A., POSTICS-GRUJIN, A., &
DJURIC, D. (1977a) Determination of small quantities of
thiocarbamate in urine. Egypt. J. Decup. Med., 5: 251-257.
PERGAL, M., RALETIC, J., & DURIC, D. (1977b) Identification of the
third metabolite, giving positive iodine-azide reaction in the
urine of workers exposed to CS2. A preliminary report. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski, Finland, 6-10 June, 1977, p. 29.
PETERSON, F. (1892) Three cases of acute mania from inhaling carbon
bisulphide. Boston Med. Surg., 127: 325-326.
PETROV, M. V. (1969)] Course and termination of pregnancy in women
working in viscose industry.] Pediatr. Akush. Ginekol., 3:
50-52 (in Russian).
PETROVIC, D. & DJURIC, D. (1965) [Carbon disulfide in exhaled air of
exposed workers.] In: Zbornik radova o toksikologiji ugljen
disulfida, Loznica, pp. 35-38. (in Serbo-Croat).
PETROVIC, D. & DJURIC, D. (1966) [Carbon disulfide in the air exhaled
by exposed workers.] Arh. Hig. Rada, 17: 159-163 (in
Serbo-Croat).
PETRUN, N.M. (1967) [The effect of carbon disulfide on certain
biochemical indices of the status of the organism in its action
via the skin.] Gig. Tr. Prof. Zabol., 11: 50-53 (in Russian).
PEYTON, T. O., STEEL, R. V., MABEY, W. R., CARLIN, A., & PINES, A.
(1976) Literature review and environmental assessment of carbon
disulfide and carbonyl sulfide. Menlo Park, CA Stanford Research
Institute, pp. 1-65. (Project no EGU 3570, US Environmental
Protection Agency, Publication No. 257-947).
PIROTSKAJA, E. N. (1972) [Changes in the protein-forming function of
the liver in workers exposed to carbon disulfide.] Vrach. Delo,
9: 132-134 (in Russian).
PREROVSKA, I. & ROTH, Z. (1968) [An observation of personnel in the
carbon disulphide hazard with special reference to the
development of arteriosclerosis.] Prac. Lek., 20: 6-10 (in
Czech).
PREROVSKA, I. & ZVOLSKY, P. (1973) [The health status in workers with
chronic carbon disulphide poisoning in five to nineteen years
after ascertaining the disease.] Prac. Lek., 25: 42-46 (in
Czech).
PREROVSKA, I., VANECEK, R., & KUBAT, K. (1961) Effect of carbon
disulfide on experimental arteriosclerosis in the rabbit. Acta
Univ. Carol., 14: 177-185.
QUARELLI, G. (1928)] Occupational CS2 poisoning.] In: Bericht. d. 5.
Internat. Kongr. f. Berufskrankh., Budapest, 1928 (in Italian).
QUARELLI, G. (1934)] Occupational carbon disulfide poisoning.] Russ.
Providenza Soc., 21: 10-73 (in Italian).
RANELLETTI, A. (1931)] Occupational carbon disulfide poisoning.]
Arch. Gewerbepath. Gewerbehyg., 2: 664-675 (in German).
RANELLETTI, A. (1933)] Occupational carbon disulfide poisoning.] In:
La Grafica Moderna, Ed. Torino (in Italian).
RAITTA, C. & TOLONEN, M. (1975) Ocular pulse wave in workers exposed
to carbon disulphide. Albrecht. Graefes Arch. Ophthal., 195:
149-154.
RAITTA, C., TOLONEN, M., & NURMINEN, M. (1974) Microcirculation of
ocular fundus in viscose rayon workers exposed to carbon
disulfide. Albrecht. Graefes Arch. Ophthal., 191: 151-164.
RECHENSERG, H. K. (1957)] Carbon disulfide poisoning and the
sulfocarbotoxic vascular Late-syndrome.] Arch. Gewerbepath.
Gewerbehyg., 15: 487-530 (in German).
RUIKKA, I. (1959) Postprandial serum turbidity in chronic carbon
disulfide poisoning a comparison of viscose rayon workers with
healthy and atherosclerotic men. Ann. Med. intern. Fenn., 48:
225-237.
SAITA, G. & GATTONI, L. (1957) [The blood coagulation process in
chronic carbon disulfide poisoning.] Med. Lavoro, 48: 229-237
(in Italian).
SAITA, G., SBERTOLI, C., & FARINA, G. F. (1964) Modifications in blood
coagulation and lipid metabolism produced by carbon disulfide.
Med. Lavoro, 55: 721-734.
SAKURAI, H. (1972) Hypertension induced by the occupational exposure
to carbon disulfide. In: Abstracts of the XVII International
congress on occupational health. Buenos Aires, 11-16 September,
1972, p. 182.
SAVIC, S. M. (1967) Influence of carbon disulfide on the eye. Arch.
environ. Health, 14: 325-326.
SAVIC, S. & IOVICIC, M. (1965) [Keratoconjunctivitis (causome) in
workers employed in the production of viscose fibres.] In:
Zbornik Radova o toksikologiji CS2, Loznica, Yugoslavia 1965,
pp. 115-121 (in Serbo-Croat).
SAVOLAINEN, H. & VAINIO, H. (1976) High binding of CS2 sulphur in
spinal cord axonal fraction. Acta Neuropathol. (Berlin), 36:
251-257.
SAVOLAINEN, H., JÄRVISALO, J., & VAINIO, H. (1977a) Specific binding
of CS2 metabolites to microsomal proteins in rat liver. Acta
Pharmacol. Toxicol., 41: 94-96.
SAVOLAINEN, H., JÄRVISALO, J., ELOVAARA,. E., & VAINIO, H. (1977b) The
binding of CS2 in central nervous system of control and
phenobarbitone-pretreated rats. Toxicology, 7: 207-214.
SAVOLAINEN, H., LEHTONEN, E., & VAINIO, H. (1977c) CS2 binding to rat
spinal neurofilaments. Acta Neuropathol. (Berl.), 37: 219-223.
SBERTOLI, C., FARINA, G., & SAITA, G. (1969) [Three cases of chronic
carbon disulfide poisoning with giomerulonephrotic syndrome.[
Med. Lavoro, 60: 28-37 (in Italian).
SCHEEL, L. D. (1965) Biological changes involving metal ion shifts.
Am. ind. Hyg. J., 26: 585-592.
SCHEEL, L. D. (1967) Experimental carbon Disulfide poisoning in
rabbits-Its mechanism and similarities with human case report.
In: Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 107-115.
SCHEEL, L. D., KEENAN, R. G., MOUNTAIN,, J. T., KopP, J., HOLTZ, J., &
KILLENS, R. (1960) The effect of dietary metal ions on carbon
disulfide toxicity. In: Proceedings of the 13th International
congress on occupational health, New York, 1960, pp. 783-784.
SCHILLING, R. S. F. (1970) Coronary heart disease in viscose rayon
workers. Am. Heart J., 80: 1-2.
SCHNEIDER, H. (1976) In: Adverse effects of environmental chemicals
and psychotropic drugs. Neurophysiological and behavioural
tests. Vol. 2, Amsterdam, 1976.
SCHÜTZ, H. (1970) Equipment and systems for ambient air monitoring,
Frankfurt/Maine, Hartmann & Braun, Mess und Regeltechnik.
SEPPÄLÄINEN, A.M. (1977) Neurophysiological findings in workers
exposed to carbon disulphide-A follow-up. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, 6-10 June, 1977, p. 17.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (1975) Electrophysiological studies
of rabbits in long-term exposure to carbon disulfide. Scand. J.
Work Environ. Health, 1: 178-183.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (19745) Electrophysiological findings
in rats with experimental carbon disulphide neuropathy.
Neuropath. appl. Neurobiol., 2: 209-216.
SEPPÄLÄINEN, A.M. & TOLONEN, M. (1974) Neurotoxicity of long-term
exposure to carbon disulfide in the viscose rayon industry. A
neurophysiological study. Work Environ. Health, 11: 145-153.
SEPPÄLÄINEN, A.M., TOLONEN, M., KARLI, P., HÄNNÄINEN, H., & HERNBERG,
S. (1972) Neurophysiological findings in chronic carbon disulfide
poisoning. A descriptive study. Work Environ. Health, 9: 71-75.
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49
(in Japanese).
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49.
(in Japanese).
SIHTOLA, H. (1976). [Stages in the development of the SINI-viscose
process.] Tutkimusja tekniikka, 7/8: 63-69 (in Finnish).
SIHTOLA, H. (1977) The SINI viscose process means less chemicals and
less pollution. In; Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 43.
SKALICKA, B. (1967) [Sulfur-containing compounds after administration
of tetraethylthiuramdisulfde.] Prac. Lek., 19: 408-412 (in
Czech).
SOKAL, J. A. (1973) Effect of chronic exposure to carbon disulphide
upon some components of the electron transport system in rat
liver microsome. Biochem. Pharmacol., 22: 129-132.
SOUCEK, B. (1957) [Transformation of carbon disulfide in the
organism.] J. Hyg. Epidemiol. (Praha), 1: 10-22 (in German).
SOUCEK, B. (1959) [Absorption and elimination of carbon disulfide.]
Prac. Lek., 11: 403-407 (in Czech).
SOUCEK, B. (1960a) [Pulmonary elimination of carbon disulfide after
intraperitoneal administration.] Farmakol. Toksikol., 23:(1):
74-84 (in Russian).
SOUCEK, B. (1960b) [The effect of carbon disulphide on the serum
cholesterol and cholesterol ester level.] Prac. Lek., 12:
243-248 (in Czech).
SOUCEK, B. & MADLO, Z. (1953) [Absorption, metabolism and action of
carbon disulphide in the organism. VI. Attempt at chromatographic
proof of the reaction of carbon disulphide with aminoacids in the
blood.] Prac. Lek., 5: 309-311 (in Czech).
SOUCEK, B. & MADLO, Z. (1954) [Absorption, metabolism and action of
carbon disulphide in the organism. VIII. Reaction of carbon
trisulphide with blood in vitro.] Prac. Lek., 6: 11-17 (in
Czech).
SOUCEK, B. & MADLO, Z. (1955) Determination of sulfhydryl groups in
guineapig serum after exposure to carbon disulfide. Bull. Soc.
Chim. Biol., (Paris), 37: 1373-1378.
SOUCEK, B. & MADLO, Z. (1956) [Dithiocarbamin carbon acids as
degradation products of carbon disulfide.] Arch. Gewerbepath.
Gewerbehyg., 14: 511-521 (in German).
SOUCEK, B. & PAVELKOVA, E. (1953) [Absorption, metabolism, and effects
of carbon disulfide in the organism. IV. Long-term experiments.]
Prac. Lek., 5: 189 (in Czech).
SOUCEK, B., MADLO, Z., & HOLEYSOVSKA, H. (1957) [The influence of
carbon disulfide on proteolytic enzymes.] Arch. Gewerbepath.
Gewerbyhyg., 15: 531-538 (in German).
STRITTMATTER, C. F., PETERS, T., & MCKEE, R. W. (1950) Metabolism of
labelled carbon disulphide in guineapigs and mice. Arch. ind.
Hyg., 1: 54-64.
STYBLOCA, V. (1977) [Electroencephalography in the diagnosis of early
brain damage by carbon disulfide.] Int. Arch. occup. environ.
Health. 38: 263-282 (in German).
SUGIMOTO, K., GOTO, S., & HOTTA, R. (1976) An epidemiological study on
retinopathy due to carbon disulfide. Int. Arch. occup. environ.
Health, 37: 1-8.
SUGIMOTO, K., GOTO, S., TANIGUCHI, H., BABA, T., RAITTA, C., TOLONEN,
M., & HERNBERG, S. (1977) Ocular fundus photography of workers
exposed to carbon disulfide-A comparative epidemiological study
between Japan and Finland. Int. Arch. occup. environ.
Health, 39: 97-101.
SULKOWSKI, W. & LATKOWSKI, B. (1969) [Audiometric and
electronystagmographic examinations of workers in plants
producing synthetic fibers, who are suffering from chronic carbon
disulfide poisoning.] Pol. hyg. Lek., 24: 1563-1565 (in
Polish).
SZENDZIKOWSKI, S., STETKIEWICZ, J., WRONSKA-NOFER, T., & ZDRAJKOWSKA,
I. (1973) Structural aspects of experimental carbon disulfide
neuropathy. I. Development of neurohistological changes in
chronically intoxicated rats. Int. Arch. Arbeitsmed., 31:
135-149.
TABACOVA, S., HINKOVA, L., BALABAEVA, L., & KURCHATOVA, G. (1977).
Effects of carbon disulphide on the pre- and postnatal
development of rats. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 26.
TACCOLA, A., MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., CAVALLERI, A., &
DJURIC, D. (1971) [Vascular changes in human subjects exposed to
carbon disulfide.] Med. Lavoro, 62: 404-411 (in Italian).
TACCOLA, A., CAVALLERI, A., FRANCO, G., & MALAMANI, T. (1977) An
evaluation of the ratio of P-wave duration to P-R segment in ECG
of CS2 exposed workers. Egypt. J. occup. Med., 5: 309-312.
TAHARA, Y. (1961) [Hygienic evaluation of work environment of rayon
factories by carbon disulfide analysis of the expired air of
workers.] J. Sci. Labour (Tokyo), 37: 583-597 (in Japanese).
TARKOWSKI, S. (1973) [Glucose and free amino acid metabolism in the
brain of rats chronically poisoned with carbon disulfide.] Med.
Pr., 24: 571-575 (in Polish).
TARKOWSKI, S. (1974) Activity of glutamate decarboxylase in the brain
of rats exposed to carbon disulfide. Int. Arch. Arbeitsmed.,
33: 79-82.
TARKOWSKI, S. & CREMER, J. E. (1972) Metabolism of glucose and free
amino acids in brain, studied with 14C labelled glucose and
butyrate in rats intoxicated with carbon disulphide.
J. Neurochem., 19: 2631-2640.
TARKOWSKI, S. & SOBCZAK, H. (1971) Oxidation and phosphorylation
processes in brain mitochondria of rats exposed to carbon
disulphide. J. Neurochem., 18: 177-182.
TARKOWSKI, S. & WRONSKA-NOFER, T. (1966) [Effect of carbon disulphide
on the cytochrome oxidase in rats.] Med. Pr., 17: 375-379 (in
Polish).
TAZUKA, H. (1955) [A study on CS2 poisoning. Report II: On the
absorption of CS2.] J. Sci. Labour (Jpn), 31: 208-209 (in
Japanese).
TEISINGER, J. (1934) Mils central hemiparesis with extrapyramidal
signs in chronic poisoning. Cas. Lek. Ces., 73: 188-189.
TEISINGER, J. (1954) [Metabolism and detoxication of some industrial
poisons.] Praha, S.Z.N., p. 106 (in Czech).
TEISINGER, J. (1971) [Chronic carbon disulphide poisoning mechanism.]
Pracov. Ldk., 23: 306-308 (in Czech).
TEISINGER, J. & SOUCEK, B. (1949) Absorption and elimination of carbon
disulfide in man. J. ind. Hyg., 31: 67-73.
TEISINGER, J. & SOUCEK, B. (1952) [The importance of metabolism of
some toxic vapours considering their transmission and excretion
in man.] Cas. Ldk. Ces., 91: 1372-1375 (in Czech).
THIELE, H. & WOLF, I. (1976) [Results of complex clinical serial tests
on CS2-exposed workers.[ Z. ges. Hyg. DDR, 22: 603-608 (in
German).
TILLER, J. R., SCHILLING, R. S. F., & MORRIS, J. N. (1968)
Occupational toxic factor in mortality from coronary hear
disease. Br. med. J., 4: 407-411.
TINTERA, J., GRAOVAC-LEPOSAVIC, LJ., & MILIC, S. (1972) Excretion of
some tryptophan metabolites in man exposed to carbon disulphide.
med. Lavoro, 63: 126-133.
TOLONEN, M. (1974) Chronic subclinical carbon disulphide poisoning.
Work Environ. Health, 11: 154-161.
TOLONEN, M., HERNBERG, S., NURMINEN, M., & TIITOLA, K. (1975) A
follow-up study of coronary heart disease in viscose rayon
workers exposed to carbon disulphide. Br. J. ind. Med., 32:
1-10.
TOLONEN, M., HERNBERG, S., NORDMAN, C-H., GOTO, S., SUGIMOTO, K., &
BABA, T. (1976) Angina pectoris, electrocardiographic findings
and blood pressure in Finnish and Japanese workers exposed to
carbon disulfide. Int. Arch. occup. environ. Health, 37:
249-264.
TOLONEN, M., HANNINEN, H., & NURMINEN, M. (1978) Phychological tests
specific to individual carbon disulfide exposure. Scand. J.
Psychol., 19(3): 241-245.
TOYAMA, T. & HARASHIMA, S. (1962) {Industrial hygienic evaluation of
exposure to CS2 through analysis of biological materials.] Jpn.
J. ind. Health., 4: 81-85 (in Japanese).
TOYAMA, T. & KUSANO, H. (1953) [An experimental study on absorption
and excretion of CS2.] Jpn. J. Hyg., 8: 70 (in Japanese).
TOYAMA, T. & SAKURAI, H. (1967) Ten-year changes in exposure level and
toxicological manifestations in carbon disulphide workers. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 197-204.
TRUHAUT, R. R., BOUDENE, C., PHU-LICH, N., & BAQUET, A. (1972)
Memoires nouvelles recherches sur la mesure en continu de
l'exposition individuelle au sulfure de carbone dans l'industrie
a l'aide d'un appareil portatif. Arch. Mal. prof., 33: 341-346.
TURCEK, M. & MOLCAN, J. (1964) [Carbon disulphide psychoses.] Prac.
Lek., 16: 257-261 (in Czech).
UEHLINGER, E. (1952) [On carbon disulfide glomerulosclerosis.]
Schweiz. Z. Allg. Pathol., 15: 217-224 (in German).
VASAK, V. (1963) [Assessment of exposure of workers to carbon
disulphide vapours.] Prac. Lek., 15: 143-145 (in Czech).
VASAK, V. & KOPECKY, J. (1967) On the role of pyridoxamine in the
mechanism of the toxic action of carbon disulfide. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 35-41.
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1963) Assessment of exposure
of workers to carbon disulphide vapours. II. Application of the
iodine-azide reaction for the detection and estimation of carbon
disulphide metabolites in urine. Prac. Lék., 15: 145-149 (in
Czech).
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1969) The assessment of the
exposure of workers to carbon disulphide by the iodine-azide test
of Vasak, Vanecek and Kimmelova. (1963). In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 56-59.
VASILJEVA, I. A. (1973) [Effect of low concentrations of carbon
disulfide and hydrogen sulfide on the menstrual function in women
and on the estrous cycle in an experiment.] Gigi. Sanit., 38:
24-27 (in Russian).
VERTIN, A. (1973) In: Gas analysis instrumentation, London,
Macmillan Press Ltd.
VERTIN, P. G. (1966) [Dynamics of the elastase and the cholesterol and
phospholipid ratio with exposure to CS2.] Arch. Gewerbepath.
Gewerbyhyg., 22: 167-178 (in German).
VERTIN, P. G. (1977) An examination for cardiac/vascular diseases in
the Dutch viscose rayon industry. In: Tolonen, M., ed. Abstracts
of the IV International Symposium on Occupational Health in the
Production of Artificial Fibres, Helsinki and Valkeakoski,
Finland, 6-10 June, 1977, p. 7.
VESCE, C. A., COPPA, S., & FIMIANI, R. (1953) [Variations in urinary
17-ketosteroids in chronic poisoning from solvents.] Folia Med.
(Napoli), 36: 444-460 (in Italian).
VIDAAKOVIC, A. & ANDJELKOVSKI, A. (1965) [Haematological changes in
workers exposed to carbon disulfide.] In: Zbornik radova o
toksikologiji CS2, Loznica, 1965, p. 77-81 (in Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & ANDJELKOVSKI, A. (1965a) [A study of the
effects of carbon disulfide in the liver.] In: Zbornik radova o
toksikologiji ugljen disulfida, Loznica, 1965, pp. 71-75 (in
Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & DJORDJEVIC, V. (1956b) [Some changes in
the blood of rats in sub-acute carbon disulfide-poisoning.] In:
Zbornik radova o toksikologiji CS2 Loznica, 1965, pp. 83-87
(in Serbo-Croat).
VIGLIANI, E. C. (1946) [Chronic carbon disulfide poisoning. A report
on 100 cases.] Med. Lavoro, 37: 165-193 (in Italian).
VIGLIANI, E. C. (1954) Carbon disulphide poisoning in viscose rayon
factories. Br. J. ind. Med., 11: 235-244.
VIGLIANI, E. C. (1961) [Intoxication with carbon disulfide.] In:
Baader, E. W., ed. Handbuch der Gesamten Arbeitsmedizin, Urban
& Schwarzenberg, pp. 313-325 (in German).
VIGLIANI, E. C. & PERNIS, B. (1955) [Clinical and experimental
investigations on atherosclerosis caused by carbon disulfide.]
Arch. Gewerbepath. Gewerbehyg., 14: 190-202 (in German).
VIGLIANI, E., VISTINI, F., & MASPES, P. E. (1944) First observations
on myopathy from carbon disulfide. Med. Contemp., 10: 85-96.
VINOGRADOV, P. B. (1966) [New experimental findings for substantiating
the maximum permissible concentrations of carbon disulfide in
reservoir water.] Gig. i Sanit., 31: 13-17 (in Russian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., VISNJIC, V., MAUGERI, U.,
& REZMAN, I. (1966a) [Hematological findings in young workers
exposed to CS2. I. Fibrinolysis.] Med. Lavoro, 57: 667-676 (in
Italian).
VISCONTI, E., REZMAN, I., MAUGERI, U., VIDAKOVIC, A., & CAVALLERI, A.
(1966b) Hematochemical findings in young workers exposed to
carbon disulfide. II. The behavior of lipoproteins and the
"clearing factor". Med. Lavoro, 57: 677-683.
VISCONTI, E., VISNJIC, V., CAVALLERI, A., VIDAKOVIC, A., MAUGERI, U.,
& REZMAN, I. (1966c) [Haematological findings in young workers
exposed to carbon disulfide. III. Antiheparinic activity.] Med.
Lavoro, 57: 745-750 (in Italian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., REZMAN, I., MAUGERI, U., &
VISNJIC, V. (1967). Fibrinolytic activity in young workers
exposed to carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 128-132.
WAKATSUKI, T. & HIGASHIKAWA, n. (1959) Experimental studies on carbon
disulfide and hydrogen sulfide poisoning, histological changes in
hemopoietic organs and other internal organs. Shikoku Igaku
Zasshi, 14: 549-554.
WEAST, R. C. (1974) Handbook of Chemistry and Physics, 55th ed.,
Cleveland, CRC Press, pp. C-234-235; D-85; D-163.
WEIGMAN, H. J. (1973). [Measuring devices for automatic surveillance
of air pollution in work places.] Z. ges. Hyg., 19: 1-9 (in
German).
WILEY, F. H., HUEPER, W. C., & VON OETTINGEN, W. F. (1936) On the
toxic effects of low concentrations of carbon disulfide. J. ind.
Hyg. Toxicol., 18: 733-740.
WRONSKA-NOFER, T. (1969) The incorporation of 14C-acetate into
cholesterol in rats exposed to carbon disulphide. Biochem.
Pharmacol, 18: 925-926.
WRONSKA-NOFER, T. (1970) The influence of nicotinic acid upon the
disturbances in lipid metabolism caused by carbon disulphide in
rats. Int. Arch. Arbeitsmed., 27: 221-227.
WRONSKA-NOFER, T. (1976) Aortic phospholipids synthesis in
experimental carbon disulphide intoxication in rats. Int. Arch.
occup. environ. Health, 36: 229-234.
WRONSKA-NOFER, W., NOFER, J., & TARKOWSKI, S. (1965) [Impaired niacin
metabolite excretion in animals poisoned with carbon disulfide.]
Med. Pt., 16: 77-81 (in Polish).
WRONSKA-NOFER, T., TARKOWSKI, S., GORNY, R., SOKAL, J. A., & SZYC, M.
(1970) Accelerated turnover rate of nicotinamide adenine
dinucleotides in the liver of rats intoxicated with carbon
disulfide. Biochem. Pharmacol, 21; 2945-2950.
WRONSKA-NOFER, T., STETKIEWICZ, J., & SZENDZIKOWSKI, S. (1973)
Structural alterations and content of nicotinamine-adenine
nucleotides in skeletal muscle of rat in chronic experimental
carbon disulfi de intoxication. Int. Arch. occup. Health., 31:
125-134.
YAMAGATA, Y., YUDA, A., SUZUKI, K., NEMOTO, T., TAKAHASHI, M.,
TUCHIDA, H., SAITO, K., & KUSUNOKI, N. (1966) Carbon disulfide
nephroselerosis with special reference to the similarity to
diabetic glomerulosclerosis. J. Jpn. Diabetic. Soc., 9:
208-217.
YAROSLAVSKY, B. K. (1969) Toxic action of carbon disulfide on
reproduction and potentiation of this effect by tryptophan.
Bull. exp. Biol. Med., 68: 88-91.
YOSHIDA, K. (1955) [Fate of CS2 in the body-formation of thioketone
bonds.] J. Sci. Labour (Tokyo), 31: 209 (in Japanese).
ZEGLIO, P. (1942) Changes in gastric function due to chronic exposure
of workers in rayon industry. Med. Lavoro., 33: 217-228.
ZENK, H. (1967) The rhino-oto-vestibular symptomatoiogy of carbon
disulphide intoxication. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 190-191.
ZENK, H. (1970) [Carbon disulfide influences in the rhino-otological
functions of workers in the plastic fibers industry.] Int. Arch.
Arbeitsmed., 27: 210-220 (in German).
Annex I
PRODUCTION OF VISCOSE AND ITS END-PRODUCTS
The technological process of viscose rayon production, which is
schematically presented in Annex I Fig. 1, can briefly be described in
the following way:
(a) Cellulose pulp arrives at the factory in the form of sheets
which are placed in a hydraulic press or pulverized and steeped in a
solution of caustic soda to form alkali cellulose;
(b) the alkali cellulose is shredded to form crumbs to
facilitate aging and xanthation;
(c) after storage in aging tanks until the right level of
polymerization has been reached, the crumbs are placed in mechanical
churns;
(d) in a separate operation, sulfur is melted and poured onto
glowing charcoal where it reacts to give carbon disulfide. The liquid
carbon disulfide is stored in special tanks that are protected against
flames, static electricity, and heat;
(e) the liquid carbon disulfide, which is gradually metered
into the mechanical churns, reacts with the alkali cellulose to form
orange crumbs consisting of colourless cellulose xanthate and orange
sodium trithiocarbonate (Na2CS3);
(f) these crumbs are dissolved in caustic soda solution to give
a viscose solution;
(g) the viscose is stored in aging tanks, filtered, de-aerated,
and then pumped into the spinning tanks where it is forced through
spinnerets that are submerged in a solution of sulfuric acid (H2SO4)
and sodium and zinc sulfates (Na2SO4-ZnSO4). Filaments are formed
by coagulation of the viscose when partial decomposition of the
cellulose xanthate and the trithiocarbonate takes place, according to
the reactions:
(1) ROCS + H2SO4-ROH + NaHSO4 + CS2
SNa
(2) Na2CS3 + H2SO4 - H2S + CS2 + Na2SO4
During this process, considerable quantities of carbon disulfide
and hydrogen sulfide vapours are liberated representing a danger to
the exposed workers. It is important that reaction (2) should proceed
much more quickly than reaction (1) during spinning, so that
practically all the hydrogen sulfide is already formed in the spinning
bath or immediately over it, and the part of the carbon disulfide that
is supplied by the xanthate is mainly freed outside the spinning bath
in the acid yarn and/or in further treatments. The quantities of
carbon disulfide and hydrogen sulfide vapours that are liberated
depend on the process and the end-use of the rayon product.
The following products may be produced from viscose: rayon
filament textile yarn used in the textile industry in linings and
furniture fabrics; rayon filament tire yarn used as reinforcing
material in the carcass and belt of tires; rayon staple fibre
combined in textile fabrics with natural or synthetic fibres, such as
cotton or polyester; and Cellophane film, used for packaging.
On leaving the spinning bath, textile rayon yarn may be:
(a) wound up on acid spools on the spinning machine above the
spinning bath;
(b) formed into a cylindrical acid cake in a centrifugal pot
also incorporated in the spinning machine; or
(c) continuously transported through the regeneration, washing,
and drying treatments and then wound up as the finished product.
During spinning, the spinning machines are more or less
hermetically sealed. However, during doffing or when filament breaks
occur, the worker must open the "windows" or covers. At such times,
hydrogen sulfide and carbon disulfide vapours escape into the air. All
hydrogen sulfide is formed in the spinning bath or immediately over
it. Most of the carbon disulfide will be absorbed in the acid spool or
cake: and either be retained for a long time or be immediately freed
in a hot acid bath. Consequently, hydrogen sulfide will mainly be
found in the atmosphere of the machine and the acid baths, whereas
carbon disulfide also occurs in the departments where the acid spools
and cakes undergo further treatment (storage room, washing and drying
departments). In spool spinning, the worker has to lean over the
spinning bath to perform the doting and spinning-in operations. In
cake spinning, doffing takes place either at the front of the machine,
under conditions that are roughly the same as for spool spinning, or
at the rear of the machine, where the worker is not forced to breathe
in the machine atmosphere.
5.3.2 Nicotinic acid
Exposure to carbon disulfide resulted in increased excretion of
methyl nicotinamide, a nicotinic acid metabolite, in the urine of rats
(Liniecki, 1960; Wrofiska-Nofer et al., 1965). An increase in serum
lipids during the exposure of rabbits and rats to carbon disulfide was
prevented by administration of nicotinic acid (Nofer & Wrofiska-Nofer,
1966; Wrofiska-Nofer, 1970). The biochemical background of this
phenomenon is still obscure, but a hypothesis based on the
interference of carbon disulfide with pyridine nucleotide and
nicotinamide metabolism has been advanced (Nofer & Wrofiska-Nofer,
1966). However, feeding nicotinic acid to the exposed animals did not
prevent the development of neuropathies in these experiments.
As the increase in urinary excretion of nicotinamide metabolites
did not occur at the cost of the systematic pool of
nicotinamide-adenine dinucleotides, Wrofiska-Nofer et al. (1970)
suggested that it reflected an increase in the whole turnover rate.
The mechanism of the process was not clarified but it may be assumed
that an increase in the synthesis via tryptophan occurs.
5.4 Effects on Catecholamine Metabolism
Disturbance of the catecholamine metabolism can play a part in
many pathological processes. In studies on rats acutely intoxicated
with carbon disulfide, significant changes were found in the brain
catecholamine metabolism (Magos & Jarvis, 1970b; Magos, 1975). There
was a decrease in the level of noradrenaline accompanied by an
increased concentration of dopamine. Inhibition of dopamine
ß-hydroxylase, an essential enzyme in catecholamine metabolism, was
demonstrated in vivo in studies on rats by Magos (1975), and in
vitro by McKenna & Di Stefano (1975). Magos (1975) advanced a theory
concerning the central role played by catecholamine disturbances in
carbon disulfide pathology, especially where central nervous system
changes were involved, and in cardiovascular pathology. Cavalleri et
al. (1977) suggested that this could also explain the involvement of
the endocrine system.
5.5 Effects on Lipid Metabolism
Disturbance in the lipid metabolism has long since been linked
with carbon disulfide exposure. Increased levels of serum lipids, free
and total cholesterol, and ß-lipoproteins have been reported in
rabbits exposed to carbon disulfide (Paterni et al., 1958; Cohen et
al., 1959; Prerovska et al., 1961). Harashima et al. (1960) reported
elevated total and esterified cholesterol levels in the serum of
heavily exposed workers, whereas workers exposed to levels of about
15-60 mg/m3 (5-19 ppm) displayed normal serum cholesterol
concentrations. Elevated cholesterol levels were also found in exposed
workers by Manu et al. (1971) and in patients with previous exposure
to carbon disulfide (the patients had not been exposed for several
years) by Graovac-Leposavic et al. (1977). Higher levels of serum
lipids and especially cholesterol may be due to an increased rate of
synthesis in the liver and to the inhibition of the degradation of
lipids (Wrofiska-Nofer, 1969; Laurman & Wronska-Nofer, 1977). The fact
that elevated cholesterol levels have not been found consistently may
be explained by the different exposure levels in different studies
(Toyoma & Sakurai, 1967). Bittersohl & Thiele (1977) found that a
higher frequency of cholesterol values exceeding 6.72 mmol/litre
(260 mg/dl) in workers exposed to carbon disulfide was strongly
correlated with the duration of exposure.
A decreased clearing factor activity was found by Ruikka (1959)
in workers exposed to carbon disulfide and Martino et al. (1963, 1964)
reported an elevated ß-lipoprotein fraction in the serum of exposed
workers.
Changes in the lipid metabolism found in the aorta tissue may
contribute to the development of atheromatic changes in blood vessels
(Wrofiska-Nofer, 1976).
An increase in serum lipids during the exposure of rabbits to
carbon disulfide was prevented by administration of nicotinic acid
(Nofer & Wrofiska-Nofer, 1966) and in rats (Wrofiska-Nofer, 1970)
(section 5.3.2).
5.6 Interaction with Microsomal Drug Metabolism
An important feature of the liver toxicity caused by carbon
disulfide seems to be the destruction of cytochrome P-450 (Bond & De
Matteis, 1969). There is evidence that this effect is due to the
oxidative desulfuration of carbon disulfide by mixed-function oxidases
(De Matteis & Seawright, 1973). The resulting, highly reactive, sulfur
becomes covalently bound to the microsomal protein (Dalvi et al.,
1974; De Matteis, 1974; Jarvisalo et al., 1977), mainly to the
apoprotein of cytochrome P-450 (Neal et al., 1976; Jarvisalo & De
Matteis, 1977; Savolainen et al., 1977a). It is possible that the
liberated sulfur is the real toxic agent in liver toxicity arising
from carbon disulfide exposure.
Experiments on rats have shown that exposure to carbon disulfide
in concentrations up to 1250 mg/m3 (400 ppm) for 8 h is followed by
an increase in microsomal RNA content and in total protein in the
hepatic microsomal fraction and also increased incorporation of
2,4-3H-L-phenylalanine in liver microsomes (Freundt et al., 1974b).
Such changes follow the action of inducing agents, such as
phenobarbital, in the microsomal enzyme system. It has, therefore,
been suggested that carbon disulfide may have an inducing as well as
an inhibiting effect on mixed-function oxygenases. However, there is
no real evidence of such an effect at present (Freundt, 1977).
6. CARBON DISULFIDE POISONING
6.1 Historical Review
At the end of 1850, several physicians observed cases of strange
nervous and mental diseases, the origin of which remained obscure. In
1856, Delpech, reported 24 cases of carbon disulfide poisoning and
confirmed the diagnosis by animal experiments (Delpech, 1856a,b). In
1863, he reported 80 more cases of "carbon disulfide neurosis"
(Delpech, 1863). Cases of chronic poisoning were also reported in
England by Bruce (1884) and Foreman (1886). Laudenheimer (1899)
described carbon disulfide poisoning in German vulcanization shops,
stirring public opinion by drawing attention to about 50 cases of
"insanity". As further cases were reported in the USA (Jump & Cruice,
1904; Francine, 1905), the need for hygienic improvement in work
places was recognized. The first epidemic of carbon disulfide
poisoning due to the vulcanization process ended at the beginning of
the 20th century. At the same time, the viscose rayon industry started
to develop and expanded rapidly. Sporadic cases of carbon disulfide
poisoning in the viscose industry were reported between 1900 and 1930
(Quarelli, 1928) but the problem became serious in the 1930s.
Raneletti (1933), Quarelli (1934) and others described cases of
psychotic and polyneurotic disorders and extrapyramidal disturbances
(Audio-Gianotti, 1932; Teisinger, 1934).
In Japan, the viscose rayon industry was established in 1916 and,
in 1929, the first cases of carbon disulfide poisoning were reported
by Tokuhara, followed by other authors (review by Kubota, 1967). Many
cases of poisoning were also described in the USA (Hamilton, 1925,
1940; Bashore et al., 1938). The so-called Pennsylvania study by Gordy
& Trumper (1938) resulted in the establishment of the first TLV of
20 ppm adopted by the American Standards Association (1941). As the
hygienic standard in this industry improved, the incidence of severe
poisoning decreased. However, during World War II, the hygienic
situation in the expanding viscose industry deteriorated and severe
poisoning again became common.
During and after World War II, many cases of carbon disulfide
poisoning were reported, mainly from Italy (e.g., Vigliani et al.,
1944; Vigliani, 1946) but also from Belgium (Langelez, 1946; Merlevde,
1951) and Finland (Noro, 1944). After World War II, the viscose rayon
industry spread to many developing countries, where the whole sequence
of degrees of exposure was repeated. In the developed countries,
attention is now focused on slowly developing symptoms due to
long-standing exposure to relatively low concentrations.
6.2 Clinical Picture of Carbon Disulfide Poisoning
Carbon disulfide intoxication has been classified as hyperacute,
acute, subacute, and chronic.
Hyperacute poisoning occurs in extreme cases of massive exposure
for a short time to concentrations of about 10 000 mg/m3 or more. The
victim quickly falls into a coma and eventually dies. Acute and
subacute poisoning occurs with short exposure to carbon disulfide
concentrations ranging from 3000-5000 mg/m3 with predominantly
psychiatric and neurological signs and symptoms such as extreme
irritability, uncontrolled anger, rapid mood changes including maniac
delirium and hallucinations, paranoic ideas, and suicidal tendencies.
Other symptoms include memory defects, severe insomnia, nightmares,
fatigue, loss of appetite, gastrointestinal troubles, asthenia, and
interference with sexual functions, such as impotency.
The symptoms and signs of chronic carbon disulfide poisoning were
described by Vigliani (1946, 1961) and the following classification of
the different syndromes was suggested by Nesswetha & Nesswetha (1967):
-- psychoses characterized by manic and depressive symptomatology
and disorientation;
-- polyneuropathy of the lower extremities, with diminished or
completely absent Achilles and patellar tendon reflexes, sensory
disturbances in a glove-stocking distribution, diminished faradic and
galvanic excitability, and decrease of the motor and sensory
conduction velocity in the peripheral nerves;
-- disturbances of the gastrointestinal tract in the form of
chronic, hyper- and hypoacidic gastritis and duodenal ulceration;
-- myopathy of the calf muscles;
-- neurasthenic syndrome with disturbances in the autonomous
nervous system,
-- optic neuritis;
-- atherosclerotic vasculoencephalopathy; the principal forms
being bulbar-paralytic, hemiplegic, or extrapyramidal.
The typical mental deterioration has been called an organic
psycho-syndrome, which may be due to general cerebral atherosclerosis,
to direct toxic action upon the brain cells, or to both.
The pattern of carbon disulfide poisoning has changed with
improvements in hygienic standards in industry. However, when the
viscose industry is established in a country with no former
experience, there is always the risk of severe poisoning; this has
occurred many times.
6.3 Effects on Organ Systems
6.3.1 Dermatological effects
Liquid carbon disulfide represents a severe irritant for both the
skin and mucosa. Hueper (1936) found blisters in viscose rayon workers
and also in experimental animals that often resembled second and third
degree burns.
6.3.2 Ophthalmological effects
Studies on the ophthalmological effects of carbon disulfide in
animals have mostly been of a histological nature. In 1899, Koester
observed changes in the retinal ganglia. Seto (1958) found vacuolar
degeneration and tigrolysis in the retina, and atrophy of the optic
nerve in rabbits exposed to a carbon disulfide concentration of about
3700 mg/m3 for 1-3 h.
Ophthalmological examinations in man are informative because it
is possible to observe the visual capacity and to study the changes in
the vessels of the ocular fundus directly. According to the literature
(e.g., Nunziante-Cesaro et al., 1952; Savic, 1967) the following
effects have been observed:
-- changes in the motility of the eyelids;
-- changes in the sensitivity of the cornea and conjunctiva;
-- changes in the motility of the ocular bulbus;
-- changes in convergency and accommodation;
-- morphological changes in the fundus such as focal haemorrhage,
exudative changes, atrophy of the optic nerve, retrobulbar neuritis,
microaneurysms, and sclerotic changes of the blood vessels;
-- functional changes, e.g., disturbances of colour vision,
adaptation to the dark, reaction of the pupil to light, accommodation,
and decrease in visual accuracy.
Most of the effects mentioned above have resulted from heavy
exposure. Many that were observed decades ago, when the observations
often lacked comparison materials, are open to criticism. Nowadays,
under prevailing working conditions such grave changes are rarely, if
ever, seen.
Increased systolic and diastolic blood pressure in the retinal
arteries and damage to the arterial walls have been reported (Maugeri
et al., 1966d; Goto et al., 1971). Maugeri et al. (1966d, 1967)
studied the arterial pressure in 107 workers who had been exposed to
carbon disulfide at concentrations ranging from 200-500 mg/m3, with
peak concentrations of up to 900 mg/m3, for 1-9 years. Using an
ophthalmodynamographic method, the authors found an average increase
in both systolic and diastolic pressures in exposed workers of about
18.4/14.7 kPa (138/110 mmHg) compared with controls which averaged
15.3/11.6 kPa (115/87 mmHg). The increase was more marked for the
diastolic pressure than for the systolic. Measurement of the pressure
of the ophthalmic arteries may be a test that needs further evaluation
in the light of the demonstrated disturbance of the brain
catecholamine metabolism induced by carbon disulfide.
Savic (1967) studied changes in the nervous system of the eyes of
young workers exposed to carbon disulfide levels of 100-400 mg/m3.
Changes were observed but only after a long period of exposure
(>5 years) (Hotta & Savic, 1972). Retinal microaneurysms have been
reported in a high proportion of Japanese viscose rayon workers (Goto
& Hotta, 1967; Goto et al., 1971; Sugimoto et al., 1976). In one
study, the prevalences of microaneurysms as judged by fundus
photography, were 8% and 2% among exposed and unexposed male workers,
respectively (Goto & Hotta, 1967). In a subsequent study (Hotta &
Goto, 1971; Goto et al., 1971), a prevalence of 56% of microaneurysms
was found in the exposed group (241 subjects) and 15% in a control
group (30 subjects). The extremely high prevalence in the exposed
groups in these studies was due to biased selection of the subjects;
one-half of them were selected for examination because of known
microaneurysms revealed by ophthalmoscopy. Later studies by the same
team did not show such a high prevalence (Sugimoto et al., 1976), but
a statistically significant difference between exposed and unexposed
subjects still persisted. Retinaopathy was found in 89 out of 289
carbon disulfide workers (31%), but only in 2 out of 49 controls (4%).
The frequency of retinopathy was correlated with both the duration and
the intensity of exposure (Sugimoto et al., 1976). It has been
suggested that the retinal microaneurysms found in Japanese workers
were related to the diabetogenic action of carbon disulfide (Goto et
al., 1971). However, microaneurysms are generally associated with
hypoxia and, though they may occur in advanced cases of diabetes, they
are not, by any means, specific for the disease. Thus, to ascribe the
excess of aneurysms found in Japanese studies to subclinical diabetes
caused by carbon disulfide is not justified. It is more likely that
they were caused by a direct effect on the retinal vessels, possibly
in association with local hypoxia coacting with ethnic or
environmental factors.
The results of recent Finnish studies do not tally with the
Japanese findings. Out of 100 exposed men and 97 controls, only 3
exposed and 2 control subjects showed 1-5 microaneurysms, and one
exposed subject showed more than 5 (Raitta et al., 1974). Furthermore,
general narrowing of the arteries and calibre irregularity were so
common in the fundus of both the exposed and control subjects that
plain ophthalmoscopy did not have any discriminatory value in this
study. A subsequent Japanese-Finnish study showed that the differences
in values obtained in the 2 studies were true differences and were not
caused by inter-observer variation (Sugimoto et al., 1977).
The most interesting finding in a study by Raitta et al. (1974)
was the high frequency of delayed peripapillary filling that occurred
in 68 exposed and 38 unexposed men. The findings of Raitta & Tolonen
(1975), suggest that studying the microcirculation of the ocular
fundus by fluorescein-angiography and oculosphygmography would help in
the early detection of carbon disulfide effects, especially since the
vessels can be observed directly in their natural state.
Ophthalmoscopy is indicated for the examination of patients suspected
to be suffering from carbon disulfide poisoning and for following-up
such patients, but it is too prone to inter-and intra-observer
variation to be used routinely by plant physicians for regular health
checks. Fundus photography eliminates some of this variation, but its
evaluation should be standardized. The diagnosis of retinal
microaneurysms requires a fundus angioscreenography, but this method
is recommended for research purposes only.
It should be added that a mixture of carbon disulfide, hydrogen
sulfide and sulfuric acid mist from an acid bath caused
keratoconjunctivitis in exposed workers (Savic & Jovicic, 1965). This
phenomenon is called "spinner's eye".
6.3.3 Otological effects
Sulkowski & Latkowski (1969) observed that exposure to carbon
disulfide impaired hearing ability. The authors examined 60 workers
occupationally exposed to carbon disulfide for between one year and
more than 10 years. The workers were below 50 years of age.
Audiometric examinations showed impairment of hearing of the receptory
type in more than 50 workers and also a decreased ability to
distinguish sound intensity within the range 1.5-3.0 dB. This
suggested a central and supracochlear localization of the impairment.
Electronystagmographic examinations performed by these authors
disclosed reduced excitability of the vestibular apparatus, suggesting
an extralabyrinthine localization of the lesion. Loss of sensitivity
to high frequency tones has also been attributed to carbon disulfide
exposure (Zenk, 1970). Since noise levels are often very high in
viscose rayon plants, it is possible that the lesions found may be at
least partly due to this cause. Vestibular symptoms, as manifested by
vertigo and nystagmus may also be present in carbon disulfide
intoxication (Zenk, 1967, 1970).
6.3.4 Respiratory effects
Carbon disulfide is a known irritant, but few data exist about
its effects on the respiratory system (Zenk, 1967). Ranelletti (1933)
described chronic cough as a consequence of the irritant effect of
carbon disulfide, but it is necessary to take into consideration the
irritative effects of hydrogen sulfide and sulfuric acid mist and of
other irritants present in the air of viscose rayon plants. The
findings of Massoud et al. (1971) i.e., cough, phlegm, wheezing,
dyspnoea, precordial pain and palpitations, may also have been due to
such mixed exposure rather than to carbon disulfide alone.
6.3.5 Gastrointestinal effects
Gastrointestinal symptoms are common among heavily exposed
workers and patients with carbon disulfide poisoning. Bashore et al.
(1938) found a prevalence of such symptoms of 25%, Vigliani (1954),
28%, Karajovic et al. (1964), 66%, Lysina (1967), 27%, and Hass et al.
(1967), 27%. Since such symptoms occur among unexposed people too,
especially in shift work, the figures listed cannot be attributed
exclusively to carbon disulfide exposure (Knave et al., 1974). A
higher prevalence of gastrointestinal disorders and liver and bile
duct dysfunction was observed in 2 groups of workers (800 and 492
subjects, respectively) exposed to very low carbon disulfide
concentrations (4-12 mg/m3) than in a control group of 453 unexposed
workers. The prevalence in the 2 exposed groups was 4.7 and 5.6%,
respectively, and that in the control group, 2.2% (Murashko, 1975).
Later, Bittersohl & Thiele (1977) found a prevalence of 22.7% of
gastrointestinal changes in 309 exposed shift workers as opposed to
5.9% in 345 unexposed shift workers.
Based on histological studies of the gastric mucosa of 75 workers
exposed to carbon disulfide, Hassman et al. (1967) suggested
classification into 5 groups: normal gastric mucosa, superficial
gastritis, chronic gastritis with incipient atrophy, chronic atrophic
gastritis, and gastritis of undetermined type. Duodenal ulceration was
only found in 2 out of 75 subjects in this study. Of the workers
examined, 11% had dyspeptic complaints and gastritis was verified in
60% of the cases. A similar figure (66%) was found by Karajovic et al.
(1964).
6.3.6 Hepatic effects
Exposure to carbon disulfide has caused fatty degeneration and
haemorrhages of the liver in animals (Bashore et al., 1938).
Experimental studies on rats have shown that pretreatment with a drug
that stimulates liver microsome enzyme activity alters the degree of
liver damage on exposure to carbon disulfide. Thus, Bond et al. (1969)
reported necrosis in the livers of rats that had been pretreated with
phenobarbital and subsequently received a single oral dose of carbon
disulfide (1 ml/kg body weight) but not in rats treated only with
carbon disulfide. Moreover, only pretreated rats displayed hydropic
degeneration of the liver. Magos & Butler (1972) found that starvation
potentiated the effect of phenobarbital in rats subsequently treated
with carbon disulfide, and that the resulting hydropic degeneration
was reversible. Similar results were obtained by Freundt et al.
(1974a), when they administered a single oral dose of 1 ml of carbon
disulfide per kg body weight to rats pretreated with phenobarbital.
However, no degenerative changes in the liver were observed in
pretreated rats following exposure by inhalation to carbon disulfide
at about 60 mg/m3 (20 ppm) and 620 mg/m3 (200 ppm) for up to 7 days.
In experiments performed on rats with short-term (8-h) exposure to
carbon disulfide at concentrations ranging from 62-1250 mg/m3
(20-400 ppm), the energy potential of the organism was damaged mainly
because of a reversible augmentation of hepatic glycolysis (Kürzinger
& Freundt, 1969; Freundt & Kürzinger, 1975).
The possible effects of carbon disulfide on the liver have been
discussed in numerous reports of clinical observations. Some authors
deny that exposure to carbon disulfide results in toxic effects in the
liver, some claim degenerative changes of the hepatocytes, and others
state that sclerotic changes are produced under conditions of chronic
exposure. Such controversial observations can be explained by
nonuniformity in approach to the method of examination. Vidakovic et
al. (1965a) examined workers hospitalized because of chronic carbon
disulfide poisoning. The exposure of these workers had been extremely
high, ranging from 1400-2200 mg/m3 (Petrovic & Djuric, 1965).
Functional disturbances of the liver and fatty degeneration of
hepatocytes were found but no necrotic changes were observed.
Pirotskaja (1972) examined the protein-forming function of the
liver in workers (40 women and 6 men) who had been exposed to carbon
disulfide for 5 years. At first, the workers were exposed to a carbon
disulfide concentration of 90 mg/m3 but later, concentrations ranged
from 8-14 mg/m3. All exposed subjects were between 30 and 40 years of
age. The mean concentrations on cystine, thyroxine, methionine,
valine, leucine, and tryptophan in serum showed statistically
significant increases in comparison with a control group.
Statistically valid decreases occurred in the levels of glutamic acid,
aspartic acid, olanine, and lysine. The levels of histidine, arginine,
and serine did not differ from those in the control group. There was a
correlation between these changes and the clinical signs of carbon
disulfide intoxication. There was no difference in the serum contents
of total protein in the exposed and control groups.
6.3.7 Renal effects
Some authors have drawn attention to nephrosclerosis in autopsies
of patients with carbon disulfide poisoning (Uehlinger, 1952; Yamagata
et al., 1966; Sbertoli et al., 1969), but this could be ascribed to a
general atherosclerotic process induced by carbon disulfide. For
example, in the cases studied by Uehlinger (1952), typical
glomerulosclerosis of the Kimmelstiel-Wilson type was established in 4
patients, while the fifth showed arteriosclerotic injury of the
kidney. Obviously renal involvement represents a very late consequence
of heavy, protracted carbon disulfide exposure, since
Graovac-Leposavic & Jovicic (1971) reported that only one patient with
persistent kidney insufficiency had ever been diagnosed in a large
viscose rayon plant employing several thousands of workers. In another
study of viscose rayon workers, Hemberg et al. (1971) found a slight
but statistically significant rise in the mean plasma creatinine
concentration compared with a control group. All values were within
"normal" limits, however.
6.3.8 Haematological effects
Brieger (1949) studied the bone marrow of exposed rats and found
retarded maturation of the erythrocytes. Mild anaemia, a slight
decrease in the haemoglobin concentration, slight reticulocytosis,
eosinophilia, and hypercoagulability of the blood have also been
recorded in exposed rats (Vidakovic et al., 1965b).
Ivanova (1967) found a moderate decrease in the haemoglobin
concentration and erythrocyte count in men with only slight exposure
to carbon disulfide. Such results cannot be attributed to carbon
disulfide effects, but rather to poor standardization of the study
conditions, since significant haematological changes in the peripheral
blood were not observed, even in highly-exposed workers (Vidakovic &
Andjelkovski, 1965; Fahim et al., 1973). Thus, it has not been
confirmed that carbon disulfide causes anaemia or polyglobulia. It is
probable that the anaemia mentioned in some clinical reports reflects
malnutrition or some other nonoccupational cause, perhaps even
defective study design, rather than an effect of carbon disulfide.
The effects of carbon disulfide on blood coagulation mechanisms
are discussed in section 6.3.14.
6.3.9 The endocrine system
Damage to the endocrine structures with functional alterations
was described in animals by Ranelletti as early as 1931, and by
Audio-Gianotti in 1932. Impaired sexual function in patients with
carbon disulfide poisoning was reported by Gordy & Trumper (1938),
Langelez (1946), and Vigliani (1946). Vesce et al. (1953) observed a
decrease in 17-ketosteroids excretion in the urine of exposed rabbits.
This phenomenon was confirmed in studies on exposed workers by
Fruscella (1962) and Olienacz et al. (1964). Most of these
observations were made on workers belonging to older age-groups and
the confounding effect of age was not controlled. Cavalleri & Zuccato
(1965), however, obtained the same results taking the age factor into
account.
In 1965, a joint Italian-Yugoslav group began an investigation on
young workers in a Yugoslav viscose rayon plant. The workers were
classified according to duration of exposure to carbon disulfide. The
average ages of various groups ranged from 26 to 33 years and they
were exposed to average carbon disulfide concentrations of about
200-500 mg/m3 with peak concentrations of up to 900 mg/m3. The
excretion of 17-ketosteroids in the urine showed a linear decrease
with the duration of exposure (Cavalleri et al., 1966a,b, 1967). The
decrease in excretion of 17-hydroxycorticosteroids was most marked in
workers with short exposure and did not decrease any more as exposure
continued. The excretion of androsterone appeared to decrease
progressively with duration of exposure, probably in a linear fashion;
that of etiocholanolone did not show any uniform pattern (Cavalleri et
al., 1966c,d). Urinary excretion of testosterone and gonadotropin
luteinizing hormone was also reduced in workers exposed to
concentrations of 100-400 mg/m3 for 2-12 yearsa. Lancranjan et al.
(1971) reported a decrease in 17-ketosteroid and
17-hydroxycorticosteroid excretion in the urine of exposed workers.
The thyroid function has been studied in exposed workers by
determination of serum thyroxine (Cavalleri et al., 1971; Cavalleri,
1975; Massoud et al., in press). Decreases reported in thyroxine
levels suggest a reduction of thyroid activity.
Mild hypothyroidism was reported in 8% of exposed workers in
studies by Lancranjan (1972). This effect may be primary or may be due
to inhibition of the hypothalamus-hypophysis axis. A decrease in
thyrotropin-stimulating hormone, observed in human subjects given
tetraethylthiuram, favours the hypothesis of an impairment of the
hypothalamic-hypophyseal system (Cavalleri et al., 1977).
a Exposure data submitted by Professor Cavalleri as private
communication to the Task Group.
When the thyroid function is inhibited, disturbances in the lipid
metabolism will appear. A well-known manifestation of subclinical
hypothyreosis is elevation of the serum cholesterol level, which in
turn may contribute to the vascular changes typical of carbon
disulfide poisoning. The isolation and identification of two
metabolites, thiourea and mercaptothiazoline, may help to explain the
mechanism (Pergal et al., 1972a,b). Neither of these metabolites is
directly toxic, but both may have some influence on thyroid activity.
In fact, drugs containing substances of the thiourea and
mercaptothiazolinone type are used in the treatment of
hyperthyroidism.
Lancranjan et al. (1969, 1972), reported hypospermia,
asthenospermia, and tetratospermia in the spermatic liquid of young
exposed workers, confirming the gonadal injury reported by Cavalleri
et al. (1965).
Disturbances were found in the ovarian function of 500 young
females, exposed to concentrations of carbon disulfide of around
20 mg/m3, and of 209 women exposed to a concentration of less than
10 mg/m3 (Vasiljeva, 1973). In a comparison with a group of 429
unexposed women, menstrual flow durations of more than 5 days occurred
in 18% of the first group and in 11% of the second compared with 5% in
the control group. Irregular menstruation occurred in 8% of those
exposed to 20 mg/m3 as opposed to 2% in the comparison group. Other
menstrual disorders followed a similar pattern, and were correlated
with the length of exposure. When vaginal smears were examined from
subsamples, cellular disturbances were most frequent in the group with
highest exposure. A biochemical study of the sex hormones in the urine
confirmed the vaginal-smear findings. Hence, even rather low exposure
to carbon disulfide appeared to induce hormonal and functional
disturbances in young women. Other studies corroborate these findings
(Petrun, 1967, Finkova et al., 1973).
In a study of 380 women exposed to around 30 mg/m3, Petrov
(1969) found more pregnancy complications than in a control group of
191 women. Spontaneous abortions occurred in 14% of the exposed, and
7% of the unexposed women. "Threatened pregnancy terminations" also
occurred more frequently in the exposed women than in the controls
(26% and 13% respectively). Exposed women gave birth prematurely
significantly more frequently than controls (9% and 3%, respectively).
It has been reported in several studies that carbon disulfide
produces primary damage at the hypothalamic-hypophyseal level
(Cavalleri et al., 1965; Lancranjan et al., 1971; Maugeri et al.,
1971; Cavalleri, 1972). The hypothalamus produces the releasing
factors that stimulate the hypophysis to synthesize and release
polypeptide hormones, which in turn stimulate the target glands in
question. The impairments found would, therefore, represent a
secondary consequence.
Thus, carbon disulfide causes several disturbances of the
endocrine systems that can be summarized as follows:
-- a reduction of adrenal activity that can be repaired by ACTH
administration and can therefore be ascribed to reduced secretion of
corticotrophin;
-- a reduction of the endocrine activity of the testis and
impairment in spermatogenesis possibly due to direct gonadal damage;
this finding could explain the decrease in "libido" and "potentiae"
often found in exposed workers;
-- disturbances in the hormonal balance in women, resulting in
menstrual irregularities, spontaneous abortions, and premature births;
-- impairment of thyroid function, which could be primary or due
to a deficiency in the thyrotropin-stimulating hormone (TSH) or the
TSH releasing factor or both.
6.3.10 Effects on the nervous system
6.3.10.1 Central nervous system
In experimental animals, carbon disulfide causes destruction of
the myelin sheet and axonal changes in both the central and peripheral
neurons. Degenerative changes have been observed in the cortex, basal
ganglia, the thalamus, the brain stem, and the spinal cord (Wiley et
al., 1936; Ferraro et al., 1941; Lewey et al., 1941; Fischer &
Michalova, 1956; Drogicina, 1968).
The mechanism by which carbon disulfide causes these changes has
not been elucidated, however. Mitochondrial enzymes may be inhibited
(Tarkowski & Wrofiska-Nofer, 1966; Tarkowski & Sobczak, 1971), and the
tyrosine and catecholamine metabolism may be disturbed (Magos et al.,
1974). The content of free glutamine increases as a result of carbon
disulfide exposure (Efremova & Uzbekov, 1968; Tarkowski & Cremer,
1972). Such changes were observed with acute experimental poisoning,
but chronic, repeated exposure did not produce gross changes
(Tarkowski, 1973).
Horvath & Mikiska (1957) followed the electroencephalographic
changes in rabbits exposed to carbon disulfide and found a transitory
decrease in the amplitude of basic and superimposed beta rhythms.
The results of animal experiments have to be extrapolated to man
with extreme caution; however, many data obtained from animal studies
are in good agreement with clinical observations in man. Like other
narcotic agents, carbon disulfide produces a clinical symptomatology
of irritation and excitation on one hand, and inhibited psychomotor
activity, psychological alterations, insomnia, hypersomnia, loss of
consciousness and death, on the other.
In acute poisoning, the first typical signs from the central
nervous system (CNS) are excitation, euphoria, and aggressive
behaviour. Subacute and chronic cases are first characterized by
neurotic signs of unrest, excitation, and loss of temper. Gradually
the patient becomes depressive, anxious, paranoiac and sometimes there
is a suicidal tendency. Nightmares, apathy, loss of initiative,
vegetative disturbances, and headache are common symptoms (Melissinos
& Jacobides, 1967; Mihail et al., 1968). In the further course of
chronic intoxication, neurological signs become prominent. Both
diffuse cortical and focal symptoms from the subcortical grey matter
and extrapyramidal system are typical (Teisinger, 1934). Typical
Parkinson symptomatology with bradykinesia, bradylalia, muscle
rigidity, tremor, and increased elementary postural reflexes is
similar to that in arteriosclerotic or postencephalitic parkinsonism
(Nakamura et al., 1974). Psychic, pyramidal and extrapyramidal
symptomatology, including signs from other parts of the brain
(vestibular, cerebellar) give a picture compatible with diffuse
effects, described under the term of "toxic encephalopathy".
Furthermore, nervous involvement in general may be reflected as
changes in the sensitive branch of the trigeminal nerve, in the
function of the III, IV, and VI cranial nerves, and in the sympathetic
and parasympathic nerves of the eye.
Disturbances in cerebral circulation probably explain some of the
manifestations of neurotoxicity. Vigliani et al. (1944) demonstrated
hyalinosis of the arterioles and precapillaries histologically, a
finding that fits into the picture of general atherosclerosis caused
by carbon disulfide. Thus, the vascular changes described by Vigliani
(1961) as "encephalovasculopathia sulfocarbonica", are probably
responsible for many of the manifestations of central nervous system
pathology.
On the other hand, authors of some recent publications have tried
to explain some mechanisms by direct interference of carbon disulfide
and its metabolites in the system of enzymatic processes in the brain.
Morphological studies indicate that long axons in the spinal cord are
preferentially destroyed in chronic carbon disulfide poisoning
(Szendzikowski et al., 1973).
The typical neuropathological picture includes an increase in
neurofilaments, and secondary effects on the myeline sheath (Juntunen
et al., 1974). Biochemical studies have shown that oxidative
desulfuration also take place in the brain (Savolainen et al., 1977b),
and that the highest specific binding of the liberated sulfur is
detected in spinal cord axons (Savolainen & Vainio, 1976). It appears
that most of the liberated sulfur binds to neurofilaments that are
essential to normal axon functions (Savolainen et al., 1977c). Thus,
there is evidence of a direct toxic action upon the nervous system.
The results reported by Abramova (1967), Vasak & Kopecky (1967),
and others (section 5.3.1) regarding inhibition of the vitamin B6
metabolism, might explain some changes in the peripheral myelin sheet
and even some effects on the central nervous system. The consequences
of vitamin B6 inhibition, or interference with the transamination of
glutamic acid and inactivation of pyridoxamine could, perhaps, produce
irritative cortical symptomatology, including epileptiform cramps.
Furthermore, studies showing interactions between carbon disulfide
and, tissue respiration; depression of cytochrome oxidase, succinic
dehydrogenase, alkaline phosphatase, and dopamine-ß-hydroxylase; and
influence of inhibitors of serum elastase (EC 3.4.21.11), suggest that
besides a purely vascular etiology, it is plausible to consider a
direct toxic interference of carbon disulfide with tissue metabolism
(section 5.2). In fact, direct toxic effects of carbon disulfide on
peripheral nerve cells (Szendzikowski et al., 1973), and on the
myoneural junctions (Juntunen et al., 1977) have been shown in
animals. The central nervous system may react in the same manner.
From all these considerations, it can be concluded that the
effects of carbon disulfide on the nervous system may be influenced by
many factors. The method most often used for the study of cerebral
involvement in man is electroencephalography (EEG). However, studies
on carbon disulfide poisoning are rare. Early studies emphasize the
frequent occurrence of very flat records among subjects chronically
exposed to carbon disulfide (Krolikowska & Rucinska, 1959). More
recently, Seppäläinen & Tolonen (1974) found 21 abnormal EEGs in an
exposed group comprising 54 viscose workers compared with only 6 in a
control group of 50 paper-mill workers. Thiele & Wolf (1976) found EEG
changes in 52% of a group of 139 spinning workers. The abnormalities
consisted of slight, diffuse slow-wave abnormalities, slight to
moderately severe focal slow-wave abnormalities and even spike and
wave discharges in 3 exposed men. Thus, mild brain dysfunctions were
clearly more prevalent in the exposed group than in the control. EEG
findings among 250 workers with longstanding occupational exposure to
carbon disulfide (mean exposure 11.2 years) were recently described by
Styblva (1977) and compared with the EEGs of 61 healthy controls and
47 patients suffering from cerebral arteriosclerosis. Increased
frequency of abnormal EEGs was found in 33.2% of the exposed workers,
compared with 6.6% in the control group ( P < 0.01). The most
frequent abnormalities among the exposed workers were episodic theta
activity (33%) and diffuse abnormality (30%) (it was not, however,
indicated or even deducible from the data presented, how these
percentages had been calculated, i.e., from which population they were
taken). Local abnormalities were rare. Local EEG abnormalities
predominated in patients with cerebrovascular disease. The episodic
activity in the EEG was considered to reflect the direct toxic effects
of carbon disulfide on mesodiencephalic structures. The author
proposed that diffuse abnormality could be a manifestation of
vasculoencephalopathy resulting from toxic changes in the small
diameter vessels. The frequency of alpha activity tended to be lower
among exposed workers and this was considered to indicate a cerebral
metabolic disorder due to carbon disulfide exposure.
Studies on the behavioural and psychological effects of carbon
disulfide exposure have indicated disturbances in deep subcortical
structures, namely vegetative centres, and more diffuse cortical
involvement. The methods used have included diagnostic interviews,
behavioural observations (mainly on experimental animals), and
psychological test batteries.
Both structured and unstructured interviews of human subjects
showed differences in the clinical pictures of carbon disulfide
poisoning at rather high concentrations i.e., about 950-2400 mg/m3
(300-760 ppm) and at much lower concentrations i.e., around
20-50 mg/m3 (7-30 ppm) over a long period of exposure. At high
concentrations, symptoms of headache, muscle pain, fatigue,
paraesthesia, and general weakness were consistently reported (Gordy &
Trumper, 1940; Manu et al., 1970). At lower levels of exposure, fewer
complaints of muscular pain and overall weakness were noted, the main
symptoms being headache, memory impairment, rapid mood changes,
insomnia, paraesthesia, and fatigue (Seppäläinen et al., 1972; Lilis,
1974).
Standardized batteries of tests yield results that are directly
quantifiable and comparisons can be made between groups at different
exposure levels and control groups. Using this method, Hänninen (1971)
presented 3 groups of workers with different exposures and clinical
profiles (unexposed, exposed without clinical symptoms, and poisoned).
The results suggested that syndromes of latent and manifest carbon
disulfide poisoning differ in quality as well as in intensity, i.e.,
"clinically manifested poisoning is characterized by lowered
vigilance, diminished intellectual activity, diminished rational
control, retarded speed, and motor disturbances, whereas traits
indicative of depressive mood, slight motor disturbances, and
intellectual impairment are characteristic of latent poisoning"
(Hänninen, 1971). Analysis of intercorrelations among test sources for
each of the 3 groups resulted in substantially different factor
structures across the 3 groups. This supports the contention that
behavioural differences between groups exposed to different
concentrations are qualitative as well as quantitative (Hänninen,
1974). Naturally, the extent to which such distinctions are, in fact,
true, depends on the number of false-positive diagnoses in the
poisoned group (i.e., how many patients with other diseases had been
classified as cases of carbon disulfide poisoning), and, since this
aspect could not be evaluated, it may be premature to accept the
qualitative changes found as real facts. As to the quantitative
changes, it is self-evident that their severity must be correlated
with the degree of poisoning, at least on a group basis. The
specificity of the psychological and behavioural examination increases
when other sub-clinical signs of carbon disulfide exposure, such as
EEG and EMG findings are present.
Hänninen & Mäenpää, as reviewed by Tolonen (1974), applied the
same type of tests to 97 exposed male viscose rayon workers (mean
exposure time 15 years) and 96 age-and-sex matched controls from a
paper mill with no history of exposure to carbon disulfide. The most
distinct difference was in the retardation of psychomotor speed, where
the relative risk was 2.2 ( P = 0.02). As a whole, 40% of the exposed
and 25% of the control group showed "poor" performance, as defined
according to criteria published by Tolonen (1974). These results show
a clear group difference; however, the test battery has a low
specificity for carbon disulfide, as could be seen from the high
number of "false positive" results.
Recently, Hänninen and her co-workers increased the specificity
of the test battery, reducing the number of tests on the basis of
information gathered from new analytical approaches applied to results
of 206 exposed and 152 unexposed men, examined in 1972. According to a
multiple discriminant analysis, the most specific variables for carbon
disulfide effects were those indicating retarded speed, retarded
emotionality and energy, and psychomotor disturbances (Hänninen et
al., 1978). Another approach, starting from test results classified as
abnormal, resulted in similar conclusions (Tolonen et al., 1976). Now,
the test battery can be restricted to a few rather specific tests
applicable to suspected cases of poisoning that can also be used at
periodic health examinations.
Analogous results concerning the depressing effects of carbon
disulfide on psychological performance and behaviour have been
reported by Foa et al. (1976), Gherase (1976), Schneider (1976),
Tuttle et al. (1976)a and Hakky (1977).
Foa et al. (1976) demonstrated an exposure-effect relationship by
comparing workers from 2 plants with different degrees of exposure. A
relationship between exposure time and performance was demonstrated by
Schneider (1976), but the role of age as a confounding factor could be
excluded for 2 test variables only.
a Tuttle, T. C., Wood, G. D., & Grether, C. B. (1976) Behavioral and
neurological evaluation of workers exposed to carbon disulfide
(CS2). Unpublished report submitted to NIOSH (National Institute for
Occupational Safety and Health) by Westinghouse Electric Corporation,
Behavioral Services Center, Columbia, MD, USA, 156 pp.
From these data, it can be concluded that, at rather low exposure
levels, psychological test results are almost the only way of
assessing involvement of the central nervous system. Relationships may
exist between psychological impairment and EEG abnormalities, but
further studies are needed to clarify this point.
6.3.10.2 Peripheral nervous system
Many authors have studied the neuropathy and myelopathy of
animals exposed to carbon disulfide (Fischer & Michalova, 1956;
Higashida et al., 1959; Scheel, 1967; Frantik, 1970; Luke,, 1973;
Szendzikowski et al., 1973; Wrofiska-Nofer et al., 1973; Seppäläinen &
Linnoila, 1975, 1976). Destruction of the myelin sheet has been the
most prominent finding, but axonal changes have also been observed. In
the muscle fibres, atrophy of the denervation type occurred secondary
to the polyneuropathy. Some species differences in susceptibility to
carbon disulfide have been found. Cats and rabbits tolerated only low
concentrations of carbon disulfide. On the other hand, polyneuropathy
in rats was produced only with exposure to extremely high
concentrations (1200-2400 mg/m3). The slowing of nerve conduction
velocity in the sciatic nerve in rabbits and rats preceded clinical
symptoms such as clumsiness and later on, paresis. In rabbits,
electromyographic abnormalities suggested the presence of myelopathy
together with peripheral neuropathy (Seppäläinen & Linnoila, 1975,
1976). Neuropathological studies (Linnoila et al., 1975) showed severe
morphological alterations in the lateral and anterior funiculi of the
spinal cord in addition to pronounced axonal alterations in the
peripheral nerves of rabbits.
Lukas (1970, 1973) established by electromyography (EMG) that the
development of neuropathy in the rat was influenced by dose, duration
of exposure, physical conditions, and the nutritional state of the
animal. This confirms the long-standing clinical belief that the
individual constitution bears some relationship to the quantity and
quality of the effects of carbon disulfide (Djuric, 1967, 1971; Djuric
et al., 1973; Goto et al., 1971).
The protective effects of some substances have also been studied
in animals. The addition of zinc and copper salts (Scheel, 1967;
Lukas, 1970), and of pyridoxine to the diet (Frantik, 1970; Luke,,
1970) did not change the toxic effects of carbon disulfide but
improved the biochemical consequences of the intoxication, for example
by decreasing the excretion of xanthurenic acid with urine (Kujalova &
Tintera, 1970). Addition of nicotinic acid (Wrofiska-Nofer, 1970)
provided some protection against the effects of carbon disulfide lipid
metabolism. The clinical picture of carbon disulfide intoxication in
man is mainly based on toxic polyneuropathy (Bashore et al., 1938;
Vigiliana, 1954, 1961; Lukas, 1969; Lukas & Hromadka, 1977; Milkov et
al., 1970; Seppäläinen et al., 1972; Seppäläinen & Tolonen, 1974;
Seppäläinen, 1977; Gilioli et al., 1977). The subjective complaints
include paraesthesia, dysaesthesia, and fatiguability and diffuse
pains of the lower extremities. Sometimes irritative signs are
prevalent and sometimes the patients have a feeling of local
hyperaesthesia, or hypersensitivity on palpation in the muscles of the
lower limbs.
Distribution of the complaints is mostly symmetrical, and,
objectively, there is hyporeflexia. Klimkova-Deutschova (1965) pointed
out the characteristic dissociation of the patellar reflex response,
which is normal or increased, with a decreased or absent Achilles
tendon reflex. As the neuropathy develops, changes first appear in the
sensitive fibres of the lower extremities, later reaching the upper
extremities. (Lukas, 1969; Lukas & Hromadka, 1977). Hyperaesthesia is
mostly of a "sock" character, but sometimes it is also diffuse.
Palpation of the nerve roots and sometimes of the whole muscle groups
is painful. Today, the development of symptoms seldom reaches the
stage of motor paresis and, consequently, atrophic changes
(amyotrophy) are rare. The same is true of vasomotoric changes.
Amyotrophy has been described in the earlier literature in the area of
the triceps surae and quadriceps muscles of the legs, as well as in
the small muscles of the hand, the thenat, the hypothenar, and in the
interosseal muscles.
It has also been suggested that cells in the spinal cord are
injured (Manu et al., 1970; Seppäläinen et al., 1972). On the other
hand, disturbances in myoneural function have resulted in
myastheniform signs (Seppäläinen et al., 1972).
Needle electromyography and measurement of the motor conduction
velocities of peripheral nerves have proved to be sensitive methods
for detecting early nerve damage, even in neurologically normal,
exposed workers (Lukas, 1969; Manu et al., 1970; Seppäläinen et al.,
1972; Lilis, 1974; Seppäläinen & Tolonen, 1974; Knave et al., 1974;
Thiele & Wolf 1976; Lukas et al., 1977). The EMG abnormality most
often found consists of a reduced number of motor units at maximal
contraction and an increase in the duration of motor unit potentials
and fibrillation. The maximum motor conduction velocities (MCV) are
lower, especially in the leg nerves, and the distal latencies are
prolonged. However, the most sensitive test for peripheral nervous
involvement appears to be the conduction velocity of the slower fibres
of the ulnar and deep peroneal nerves, and that of the H-reflex in the
region of tibia] nerve (Lukas, 1969, 1977; Seppäläinen & Tolonen,
1974; Gilioli et al., 1977). In a recent epidemiological study
(Seppäläinen & Tolonen, 1974) comprising 118 male viscose rayon
workers and 100 control subjects from a paper mill, the most important
findings were as follows: the conduction velocities in the nerves of
the exposed men were generally slower than those of the controls. The
most distinct differences (all P values smaller than 0.01) were
obtained for the conduction velocity of the slower fibres of the ulnar
and deep peroneal nerves, the MCV of the deep peroneal and posterior
tibial nerves, and the motor distal latency of both the median and
ulnar nerves. The exposed group in this study was predominantly
composed of clinically "well" men, exposed for a number of years
(median 15) to carbon disulfide plus hydrogen sulfide concentrationsa
of about 60-190 mg/m3 (20-60 ppm) in the 1950s, about 30-95 mg/m3
(10-30 ppm) in the 1960s, and mainly below 60 mg/m3 (20 ppm) during
the last years. A little more than half of the men were still exposed
at the time of examination, the rest were working in "clean"
departments.
Different types of nervous system symptoms and findings among 51
Swedish viscose factory workers were evaluated by Knave et al. (1974).
The iodine-azide test was used as a measure of exposure intensity. The
comparison group consisted of 51 subjects matched for sex and age.
Symptoms of peripheral nervous dysfunction (e.g., restless legs,
cramps, pain, and numbness) were over-represented among workers,
especially the combination of these symptoms. Only one neurological
sign-namely tremor-showed a higher prevalence in the exposed than in
the control group. The authors measured only the maximal MCV of the
median nerve and found minor slowing in this MCV when the duration of
employment was between 5 and 30 years, but not after shorter exposure.
The prognosis of carbon disulfide polyneuropathy has not been
considered to be too poor. According to one follow-up study of exposed
workers, electroneuromyographic abnormalities tend to ameliorate if
the exposure level decreases or exposure is stopped (Seppäläinen,
1977). A tendency for repair and restitution of lost functions has
also been demonstrated experimentally in animals (Lukas, 1970;
Seppäläinen & Linnoila, 1975, 1976).
However, it has been shown that, even after periods of 10-15
years without exposure, abnormal conduction velocity values may be
found in workers with previous carbon disulfide poisoning or previous
long-term exposure to carbon disulfide (Seppäläinen et al., 1972).
This corresponds with the experience of Gilioli et al. (1977), who
found the reduction in conduction velocity to be irreversible. In
another study, Seppäläinen (1977) described an amelioration in the EMG
findings when carbon disulfide exposure was reduced or stopped.
a Exposure levels in studies by Hernberg et al. (1970, 1973, 1976),
Raitta et al. (1974), Tolonen (1974), Tolonen & Seppäläinen (1974),
and Tolonen et al. (1975) refer to combined concentrations of carbon
disulfide and hydrogen sulfide. Hydrogen sulfide concentrations were
about 10% of the carbon disulfide concentrations (Hernberg et al.,
1970).
6.3.11 Cardiovascular effects
A number of studies have shown that carbon disulfide causes
vascular changes in various organs of experimentally exposed animals
(Lewey, 1941; Guarino & Arcello, 1954; Paterni et al., 1958). In man,
evidence of vascular changes caused by long-term exposure, has been
accumulating since World War II. Attinger (1948) was the first to
describe such changes in 5 autopsy cases. Further clinical and autopsy
studies showed clearly that carbon disulfide was a vasotropic poison
(Uehlinger, 1952; Attinger, 1954; Vigliani & Pernis, 1955; Crepet et
al., 1956; Rechenberg, 1957; Maugeri et al., 1966a,b; Prerovska &
Roth, 1968; Maugeri et al., 1971; Prerovska & Zvolsky, 1973).
According to Vigliani & Pernis (1955) the "vasculopathia
sulfocarbonica" appears in 2 forms, namely in cerebral arteries
causing encephalopathy, and in the renal arteries causing nephropathy
and hypertension. The syndromes that follow these types of vascular
damage have already been described in sections 6.2, 6.3.7, and
6.3.10.1. Autopsy studies have revealed that sclerosis is prevalent in
large and middle-size arteries and hyalinization in arterioles and
capillaries. These changes appear in the blood vessels of all organs
but they are particularly prominent in the central nervous system and
kidneys.
Vascular changes due to carbon disulfide exposure are similar to
those produced with atherosclerosis due to age. Thus, the picture in
older persons is confused by the influence of age. The carbon
disulfide induced vasculopathy is therefore best studied in young
persons with a sufficiently long exposure time. With this in mind, a
joint Italian/Yugoslav team selected 104 young workers (age around
30-35 years) with more than 10 years' exposure for closer study
(Maugeri, et al., 1966a,b; Maugeri et al., 1971; Taccola et al.,
1971). Of these, 28 were invalids due to carbon disulfide poisoning.
Organic changes in blood vessels were discovered, mostly in the
invalids, by means of cerebral rheography. The results obtained
suggested progression of such changes even after the termination of
exposure. Using peripheral rheography, plethysmography, and
oscillography, the authors established functional vasoconstriction in
the arteries of the upper and lower extremities. Organic alterations
of the sclerotic type were rare and mainly appeared after prolonged
exposure and in the lower extremities.
During the last 10 years or so, it has become increasingly
evident that long-standing exposure to carbon disulfide promotes
coronary heart disease, even under circumstances where clinical
poisoning is uncommon. Of the several attempts made to connect carbon
disulfide exposure with coronary heart disease, only a few have
complied with necessary methodological requirements. The first such
investigation was a careful mortality study in Britain published by
Tiller et al. (1968). They found a 2.5-fold excess mortality in
viscose rayon workers exposed to carbon disulfide for 10 years or
more, compared with that of other workers. The same study demonstrated
that, in the 3 plants studied, coronary mortality was proportionally
higher among workers engaged in the viscose spinning process than in
other workers, men living in the locality, and the national statistics
derived from the Registrar General's tables. Similar results have been
obtained from Norway, where a 3-fold excess mortality from coronary
heart disease was found among workers, aged 35-54 years, compared with
unexposed workers in other departments (Mowé, 1971).
A prospective study that was initiated in 1967 in Finland, showed
that the 5-year mortality in a cohort of 343 male viscose rayon
workers exposed to about 30-95 mg/m3 (10-30 ppm) was almost 5-fold
(14 deaths compared with 3) that of an unexposed comparison cohort
from a nearby paper mill. Other causes of death were evenly
distributed (Hernberg et al., 1970, 1973). The incidence of nonfatal
first myocardial infarctions was almost 3 times that in the comparison
cohort (11 compared with 4) (Tolonen et al., 1975). In a continuation
of the follow-up extending to 8 years, the cumulative incidence rate
for mortality from coronary heart disease was 5.8% in the exposed
group and 2.6% in the comparison group, giving a rate difference of
3.2% (Hernberg et al., 1976; Nurminen, 1976). Between the sixth and
ninth years of follow-up, no further excessive mortality was observed,
probably because most (81%) of the originally exposed workers were no
longer exposed to carbon disulfide and also overall concentrations of
carbon disulfide had decreased to less than 30 mg/m3 (10 ppm). This
positive trend was promising with regard to coronary heart disease:
the prognosis of exposed workers improved with improved occupational
hygienic measures and with a reduction in the length of exposure over
a lifetime.
Anamnestic angina was more prevalent both in 1967-68, and at
re-examination in 1972, whereas there were no great differences in
coronary electrocardiographic findings (Hemberg et al., 1970; Tolonen
et al., 1975) as coded by the "Minnesota Code" (Blackburn et al.,
1960). These results showed that the greatest effect of carbon
disulfide on coronary arteries was on the production of fatal
infarctions (relative risk 4.8) followed in reducing order by all
infarctions, fatal and nonfatal (relative risk 3.7), nonfatal
infarctions (relative risk 2.8), angina (relative risk 2.2), and
coronary ECGs (relative risk 1.4). In other words, the more serious
the outcome, the greater the relative risk. The interpretations may be
that exposure to carbon disulfide not only worsens the prognosis of
existing coronary heart disease but increases the incidence of new
cases. However, it is not certain that this mode of action operates in
countries where coronary heart disease is less common than in Finland.
Coronary heart disease has a multifactorial etiology, and the pattern
described above may be noticeable only when a sufficient number of
other coronary risk factors are present at the same time. In fact,
some other studies have shown a more distinct excess of coronary ECGs
(Goto & Hotta, 1967; Cirla et al., 1972). It is possible that both
dose-dependent, methodological, and even geographical factors may be
responsible for the discrepancies reported so far. For example, it may
be that the pattern of excess coronary heart disease differs in
different populations, depending on the concomitant prevalence of
other coronary risk factors such as elevated blood lipids and
hypertension.
A joint analysis of the prevalence of angina pectoris, exercise
ECGs, and blood pressure measurements in Finnish and Japanese workers
exposed to carbon disulfide and in 2 unexposed control groups
supported this view (Tolonen et al., 1976). Angina was rare and
evidence of past infarction nonexistent in both exposed and unexposed
Japanese men; in the Finnish exposed and unexposed groups the
prevalence of angina was 15 and 10%, respectively. The prevalence of
coronary ECGs was almost equal among the exposed and unexposed men in
both the Japanese and the Finnish groups. Angina plus coronary ECGs as
evidence for coronary heart disease occurred with a prevalence of nil
for both exposed and unexposed Japanese, and 5 and 2% for the exposed
and unexposed Finnish groups, respectively. The study did not yield
any evidence of an excess in any of the variables examined among the
exposed Japanese workers. However, a follow-up study of this group is
necessary to shed more light on the possibility of excess morbidity
and mortality from coronary heart disease. Vertin (1977) did not find
any significant differences in coronary heart diseases, ECG changes,
serum cholesterol concentrations, and arterial pressure in workers
exposed to carbon disulfide concentrations of about 30-95 mg/m3
(10-30 ppm) compared with a control group.
Carbon disulfide vasculopathy may be due to the combined action
of several biochemical and physiological disturbances, namely:
(a) changes in lipid metabolism (section 6.3.9);
(b) disturbances in the coagulation mechanism (section 6.3.14);
(c) elevation of blood pressure (Hemberg et al., 1970; Sakurai,
1972; Tolonen et al., 1975);
(d) subclinical hypothyroidism (Cavalleri et al., 1971);
(e) a toxic effect on the myocardium, either direct or through
interference with the catecholamine metabolism (Magos, 1972).
However, much work remains to elucidate the mechanism of carbon
disulfide-induced atherosclerosis. In particular, coronary heart
disease has a multicausal origin and is closely related to the
saturated fat intake of the population. Furthermore, its incidence is
influenced by a large number of other risk factors, such as smoking,
diabetes, and physical inactivity. A combination of 2 or more risk
factors greatly increases the incidence. Thus, it may be postulated
that carbon disulfide will only become evident as a coronary risk
factor in the presence of other risk factors.
6.3.12 Carcinogenicity and mutagenicity
No reports are available that indicate any carcinogenic or
mutagenic effects of carbon disulfide.
6.3.13 Teratogenic effects
Only a weak teratogenic effect appeared in rats following
low-level exposure to a combination of hydrogen sulfide and carbon
disulfide (Bariljak et al., 1975).
A marked impairment (increase in early embreonal mortality,
reduction in fetal weight, malformations in the brain and limbs,
behavioural deviation) in the prenatal development of 2 successive
generations was produced in rats exposed to carbon disulfide
concentrations of 100 and 200 mg/m3 throughout the period of
gestation (Tabacova et al, 1977).
6.3.14 Other effects
The literature is contradictory concerning the effects of carbon
disulfide on blood coagulation. Saita & Gattoni (1957) found a slight
decrease in factor VII and prothrombin in exposed workers. On the
other hand, Moreo & Candura (1960) and De Nicola et al. (1962)
reported hypercoagulability in the blood of experimental animals, a
finding that was later confirmed in studies of exposed workers by
Candura et al. (1962), Saita et al. (1964), and Danilova (1968). An
increase in fibrinolysis was found by De Nicola et al. (1962) in
animals suffering from acute poisoning. In contrast, Candura et al.
(1962) found decreased fibrinolysis in workers exposed to carbon
disulfide.
The coagulation mechanism in highly-exposed young workers in a
viscose factory was studied by Visconti et al. (1966a,b,c, 1967). They
found protracted coagulation, retarded production of active
thrombotastin, decreases in plasminogen and in the activity of
plasmin, and a decrease in antiheparinic activity in workers exposed
for up to 9 years. These disturbances were most pronounced during the
first years of exposure.
Visconti et al. (1966a) supposed that carbon disulfide could
affect blood coagulation either directly by interfering with the
system itself, or indirectly, by liver injury. Different routes of
entry, differences in the level and duration of exposure and in the
distribution of carbon disulfide in the organism, and also
interspecies differences are probably responsible for the confusing
results.
It has been reported that the glucose metabolism is disturbed by
carbon disulfide both in experimental animals (rabbits) (Hara, 1958)
and in exposed workers (Goto et al., 1971). There are also a few
clinical studies reporting an increased occurrence of diabetes in
patients suffering from severe carbon disulfide poisoning (Austoni &
d'Agnolo, 1957; Finulli & Ghislandi, 1959; Ferrero, 1969). Lack of
proper control groups renders the interpretation of these results
difficult.
6.3.15 Interactions with other chemical compounds
The combined effects of carbon disulfide with other chemical
compounds were first reported by Lazarev et al. (1965) who indicated
that repeated exposure to a concentration of carbon disulfide of
3900 mg/m3, for 2 h per day, over a period of 20-30 days considerably
prolonged both the hexobarbital sleeping time and alcohol retention
time in rats and mice (see also section 6.3.6).
Carbon disulfide caused reversible inhibition of the oxidative
drug metabolism in rat liver endoplasmic reticulum (Bond et al., 1969;
Freundt & Dreher, 1969; Freundt & Kuttner, 1969; Freundt & Henschler,
1971; Freundt, 1973; Sokal, 1973; Freundt et al., 1974b, 1975, 1976).
This effect was also produced in man after exposure to a carbon
disulfide concentration of about 30 mg/m3 (10 ppm) over a 6-h period
(Mack et al., 1974); the treatment led to a rapidly reversible but
significant decrease in the oxidative N-demethylation of the
analgesic, aminopyrine.
Simultaneous exposure to carbon disulfide and ethanol resulted in
an increase in blood acetaldehyde in rats and man that was considered
to be a consequence of the inhibition of aldehyde dehydrogenase by
carbon disulfide (Freundt & Netz, 1973; Freundt & Lieberwirth, 1974;
Freundt, 1974; Freundt et al., 1976). In 12 healthy male volunteers, a
50% increase in blood acetaldehyde was noted following exposure to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) for 8 h
with simultaneous ingestion of ethanol in orange juice at a
concentration that maintained the blood ethanol level at about 0.8%.
However, no signs of an "antabuse syndrome" were observed under these
experimental conditions (Freundt et al., 1976).
6.4 Diagnosis
Since all isolated effects are nonspecific, individual diagnosis
becomes a matter of probability based on: (a) the ascertainment of
exposure; (b) the demonstration of signs and symptoms of poisoning
and the combination in which they occur; and (c) the exclusion of
other diseases.
The changes due to carbon disulfide exposure described in
sections 6.2-6.3.15 affect so many different organ-systems that, in
considering the etiology of a single case, the probability of it being
caused by carbon disulfide increases in proportion to the number of
these signs and symptoms present. Although, in 1941, Lewey drew
attention to the importance of the concomitant occurrence of different
neuropsychiatric and vascular effects, as well as to their connection
with the severity of the disease, there have been few, if any, studies
systematically analysing the knowledge that has emerged since then.
For this reason it is difficult to find quantitative data. However,
carbon disulfide poisoning should always be suspected when a viscose
worker presents with subjective, neurasthenic symptoms, signs of
peripheral neuropathy, psychological disturbances, and vascular
changes in the cerebral, coronary, renal, or peripheral systems. When
exposure to toxic levels of carbon disulfide occurs, the following
chronological effects can usually be expected: the appearance of
neurasthenic symptoms accompanied by psychological behavioural
changes; signs of peripheral nervous system involvement as shown by
electromyographic changes, initially mainly in the afferent fibres but
later in the motor fibres; simultaneous disturbances in the central
nervous system may appear but may be more difficult to detect
objectively than the lesions of the peripheral nervous system. Another
early finding is a decrease in the level of serum thyroxine that is
probably due to damage of the hypothalamic-hypophyseal axis (section
6.3.9). Cardiovascular damage appears after longer periods of
exposure. The appearance and severity of the above-mentioned signs and
symptoms are related to the level of the uptake and to the duration of
exposure to carbon disulfide. The changes are also dependent on the
individual constitution and health status of exposed workers.
In the differential diagnosis of true cases, atherosclerosis due
to other causes should be considered including, postencephalitic or
atherosclerotic Parkinsonism, brain tumours, syphilis, multiple
sclerosis, and psychiatric diseases of other origins such as endogenic
depression, schizophrenia, and alcoholism. Peripheral polyneuropathy
should be distinguished from that due to alcoholism, diabetes, and
other toxic agents.
The subclinical stage is, by definition, more difficult to
identify. First, the presence of pathological symptoms and signs must
be established, which requires sensitive techniques. Second, the
causal connection between an established symptom or sign and carbon
disulfide exposure should be shown. Both aspects pose great
difficulties because of the nonspecificity of the manifestations and
their vagueness in the initial stages. So far, the only attempt to
approach this problem in a systematic way has been made by Tolonen
(1974).
He studied 97 men exposed to carbon disulfide in a viscose
factory and 96 controls of the same age from a paper mill. The mean
age was 48 years (range 33-67 years), and the mean exposure time was
15 years (range 1-27 years). The intensity of background exposure
ranged from about 60 to 125 mg/m3 (20 to 40 ppm) in the 1950s and
then from about 30-95 mg/m3 (10-30 ppm). Personal exposure data were
not available. The group comprised both completely "healthy" workers
as well as some partly incapacitated patients with past poisoning.
About half of the men were still exposed to carbon disulfide, the rest
being employed in "clean" departments. The groups underwent many
examinations including: (a) examination of the heart (positive =
history of verified myocardial infarction, and/or "Minnesota Codes"
I1-3, IV1-3, V 1-3, VIII3, VIII3 , XI1-3-5-7, and/or typical angina):
(b) psychological testing (section 6.3.10.1); (c) measurement of
the conduction velocities of 8 peripheral nerves (polyneuropathy was
considered to exist when 2 or more nerves showed reduced conduction
velocities) and (d) examination of the circulation of the ocular
fundus (the criterion for disturbed circulation was delayed
peripapillary filling -- circumferential, segmental, or both). The
occurrence of isolated and combined signs is shown in Table 2.
As many as 59% of the exposed and 29% of the unexposed men were
affected by more than one disorder under study but most combinations
of disorders occurred more frequently in the exposed group; a
combination of 3 abnormalities was 3 times more common in the exposed
group than in the controls and a combination of all 4 abnormalities
occurred only in the exposed group. Only 5% of the exposed subjects
compared with 31% of the controls were without any abnormalities.
Since disturbances in the choroidal circulation were present in all
cases of excess "morbidity" (68% of the exposed, Raitta et al., 1974),
it seems that this abnormality represents the earliest manifestation
of carbon disulfide toxicity, at least, of those considered here.
Thus, it can be postulated that a positive diagnosis of carbon
disulfide poisoning can only be made if changes in the choroidal
circulation have been observed and providing that exposure has
extended over 10 years or more, with an intensity of the magnitude of
about 30-90 mg/m3 (10-30 ppm). The estimated probability that a
combination of findings is of occupational origin is the excess
morbidity over total morbidity. The results also showed that the
etiological role of carbon disulfide could be demonstrated with
greater probability, the greater the number of abnormalities present
at the same time.
The necessity to ascertain exposure is so obvious that it should
not need to be mentioned. It should be stressed that recent experience
has shown the importance of evaluating personal, as opposed to
background exposure, since the former may be only weakly correlated
with the latter. Personal exposure can be measured using personal
samplers, or by the iodine-azide test. The latter has been claimed to
discriminate particularly sensitive workers (whose test does not
return to normal after 16 h away from exposure) (Graovac-Leposavic et
al., 1967).
Table 2. Prevalence (%) of coronary heart disease (CHD), delayed
peripapillary circulation (EYE), polyneuropathy (PN) and
behavioural symptoms (BS), their combinations in subjects
in the exposed and control groups, and the differences
between the groups.a
Exposed Group Difference =
(N = 97) control excess morbidity
(N = 96)
Free of disease 5 31
CHD only 4 9 -5
EYE only 19 19 0
PN only 6 6 0
BS only 7 6 1
CHD + EYE 4 4 0
CHD + PN 1 1 0
CHD + BS 1 2 -1
EYE + PN 12 5 7
EYE + BS 7 4 3
PN + BS 4 5 -1
CHD + EYE + PN 9 0 9
EYE + PN + BS 11 5 6
CHD + PN + BS 2 1 1
CHD + EYE + BS 2 2 0
CHD + EYE + PN + PS 6 0 6
Total 100 100 26
a From: Tolonen (1974).
6.5 Surveillance of the Health of Exposed Workers
Considering the early manifestations of chronic carbon disulfide
effects and the factors that may influence the appearance and the
severity of such effects (section 6.4), the following examinations are
recommended for health surveillance:
(a) Clinical neurological investigation;
(b) Electroneuromyographic (ENMG) examination wherever possible
(especially the conduction velocity test, which should be carried out
on several nerves);
Additional tests that may give valuable information include:
(a) Psychological and behavioural testing with a narrow battery
of well validated tests;
(b) Serum thyroxine measurement repeated at least once a year;
(c) Blood pressure measurements;
(d) Electrocardiography (preferably an exercise ECG);
(e) Fundus photography (in some countries);
(f) Blood lipid pattern estimation, if the exposure level is
high;
(g) Electroencephalography (EEG), if there are special
indications;
These tests should be performed at the pre-employment examination
and subsequently at regular intervals to detect early deviation from
baseline values. Changes observed in the same individual over a period
of time may suggest a relationship with carbon disulfide uptake.
Depending on the intensity of exposure, the iodine-azide test
should be carried out from 2-12 times a year, both immediately after
the work shift and the next morning. Those with positive tests should
be seen by the plant physician, since a pathological test in the
morning is a warning of incipient poisoning (Graovac-Leposavic et al.,
1967). In addition to this, medical examinations should be made once
or twice a year. They should comprise a thorough history and a
neurological examination.
When suspect signs of early carbon disulfide effects appear, the
worker should be removed from exposure to a "clean" job. In the light
of the new findings concerning coronary heart disease, the removal of
workers who develop coronary risk factors, such as hypertension,
hypercholesterolaemia, and ECG changes may be indicated, irrespective
of their etiology. In some plants, there is the practice of regular
periods of work in a clean atmosphere, e.g., every sixth month, or of
extra vacations in the winter.
Some authors believe in the importance of the diet of exposed
workers. However, the protective effect of various diets and
supplements for man remains unclear. Scheel (1965) claimed that
addition of zinc and copper salts to the diet of exposed rats had a
protective effect against carbon disulfide exposures. A diet poor in
proteins, especially amino acids containing sulfur, but enriched in
vitamin B6 and glutaminic acid was suggested by Agronovskij &
Goloscapov (1972). Lukas (1973) established that rats on an "optimal"
diet were more resistant to the effects of carbon disulfide than rats
on a normal diet. In the first group, the signs of intoxication
appeared after a longer time and the effects were less marked than in
the second group.
At the present time, the only recommendation that can be given in
the case of man is that workers should be provided with a diet
containing sufficient amounts of energy foods to provide the required
joules, proteins, vitamins, and trace elements. This recommendation is
of particular importance in countries where such dietary requirements
are not necessarily fulfilled normally. However, basing the preventive
programme mainly on diets or drugs should be condemned, since the
fundamental question is that of technical improvements.
6.6 Contraindications for Exposure to Carbon Disulfide
An inborn error of metabolism manifesting itself as an abnormal
iodine-azide reaction in subjects working under conditions of high or
moderate carbon disulfide exposure (less than, 50 mg/m3) should be
considered an absolute contraindication for exposure.
Other contraindications for working in an environment containing
carbon disulfide include those common to all other toxic exposures
such as youth (below 18 years), pregnancy, concomitant chronic disease
such as psychiatric neurological, cardiac, renal, and pulmonary
diseases, and any condition that prevents the use of respirators, etc.
In addition, contraindications based on the specific toxicity of
carbon disulfide include diseases of the metabolic system, such as
liver disease and endocrine disorders. Any neurological or psychiatric
disorder is also a contraindication, including vegetative dystony and
psychic lability. A history of gastritis and peptic ulcer is a
relative contraindication, depending, for instance, on whether or not
there will be shift work and on the intensity of exposure. Recently,
attention has been focused on to what extent the presence of coronary
risk factors should be regarded as a contraindication; no strict
recommendations can be given, but as a general rule the presence of
several of these in an individual, or alternatively strongly abnormal
values for one risk factor, should be regarded as contraindications
(the strongest coronary risk factors apart from age itself are
elevated serum cholesterol or triglycerides, heavy cigarette smoking,
hypertension, and diabetes). In the evaluation of these and other
contraindications, the exposure intensity and hygienic conditions
should, of course, be taken into consideration.
The employment of women in work places with carbon disulfide
exposure is a special problem.
Considering the studies referred to in section 6.3.9, it is
advisable to exclude pregnant women from carbon disulfide exposure.
The same is true for lactating women and for those with expressed
disturbances of menstruation and habitual abortion.
7. EXPOSURE-EFFECT AND EXPOSURE-RESPONSE RELATIONSHIPS
7.1 Validity of Exposure Data
In general, valid exposure data are available only from
experimental studies on animals. Almost all epidemiological studies
have failed to document the exposure levels pertinent to the effects
studied. There is a universal lack of retrospective exposure data that
usually renders the assessment of the relationship between chronic
effects and the exposure levels responsible for their occurrence
impracticable. Furthermore, even when exposure data are available,
they often consist of occasional short-time measurements from fixed
sites that are not necessarily representative of the workers'
exposure. It is, therefore, extremely difficult, if not impossible, to
establish exposure-effect or exposure-response curves for carbon
disulfide, based on epidemiological evidence. Even the relation of
isolated findings reported in the literature, to given exposure levels
is difficult. Thus, the attempts in this document, to relate the
intensity of exposure to the intensity and frequency of effects must
be regarded with caution.
7.2 Experimental Data
7.2.1 Acute animal exposure
Acute exposure to carbon disulfide in animal experiments by
inhalation or by intramuscular or intraperitoneal injection has
provided information regarding the mechanism of carbon disulfide
toxicity, but it is difficult to apply this information to the
industrial exposure of man.
7.2.2 Long-term animal exposure
Several experiments on the inhalation of carbon disulfide have
been performed on different types of animals. In most of these
studies, exposure was to carbon disulfide only, but in a few, a
combination of carbon disulfide and hydrogen sulfide was used.
The concentrations of carbon disulfide ranged from
0.1-2330 mg/m3 and those of hydrogen sulfide, from 0.1-140 mg/m3;
the exposure periods ranged from 30 min per day to 6 h per day for up
to 15 months.
The reactions to such exposures included increased mortality,
teratogenic changes, testicular lesions, aspermatogenesis, engorged
blood vessels, weakness, paralysis, lethargy, weight changes,
metabolic changes, behavioural changes; sometimes there were no
observed effects. Some of the effects resembled lesions found in
exposed workers; others did not bear any relationship to those arising
from human exposure (Wakatsuki & Higashikawa, 1959; Goldberg et al.,
1964; Minden et al., 1967; Yaroslavski, 1969; Frantik, 1970; Gondzik,
1971; Misiakiewicz et al., 1972; Szendzikowski et al., 1973; Bariljak
et al., 1975; Seppäläinen & Linnoila, 1976; Tabacova et al, 1977). The
considerable differences in ranges of concentrations and periods of
exposure used in these experiments make conclusions regarding
exposure-effect and exposure-response curves impossible. This
regrettable situation emphasizes the need for international
coordination of toxicological experiments to make comprehensive
scientific evaluation possible.
7.3 Epidemiological Data
All studies so far reported have been from the viscose rayon
industry, where exposure to carbon disulfide occurs together with
exposure to hydrogen sulfide. However, generally, the levels of carbon
disulfide are at least one order of magnitude higher than those of
hydrogen sulfide, and most authors have attributed the health effects
found to carbon disulfide rather than to hydrogen sulfide exposure.
Only some of the various effects reviewed in section 6 can be
related to exposure levels. The following section contains a brief
review of exposure-effect and exposure-response data for neurological,
cardiovascular, ocular, and gonadal effects.
7.3.1 Neurological and behavioural effects
Severe acute neurological effects such as psychosis and paralysis
occur when exposure exceeds 1500-3000 mg/m3 (500-1000 ppm) (Vigliani,
1954). Chronic encephalopathy may develop with long-standing exposure
to 150 mg/m3 (50 ppm) or more (Vigliani, 1954). More subtle
neurological changes, detectable by neurophysiological examination or
psychological testing, have been reported at lower concentrations. For
example, slowing of nerve conduction velocities and prolonged distal
latencies of several nerves have been found among viscose rayon
workers with a median exposure time of 15 years to combined
concentrations of carbon disulfide and hydrogen sulfide of
60-190 mg/m3 (20-60 ppm) in the 1950s, 30-95 mg/m3 (10-30 ppm) in
the 1960s, and mainly below 60 mg/m3 (20 ppm) during the years
preceding the examination (Seppäläinen & Tolonen, 1974). The same
workers were also studied for psychological performance (Tolonen,
1974). The most distinct difference compared with a control group was
found in the retardation of psychomotor speed. As a whole, "poor"
performance, as defined by Tolonen (1974), occurred 1.6 times more
frequently among the exposed workers. These results suggest that
concentrations around 60-90 mg/m3 (20-30 ppm) can produce
psychological disturbances. In an indirect way, Tuttle et al.a
corroborated this view by being able to relate behavioural test scores
to other indices of neurological health, thought to occur under
similar exposure conditions, but because no exposure data were
available, their findings are not directly applicable in the
exposure-effect sense.
According to Martynova et al. (1976), an excess of functional
nervous disorders was found among 108 workers exposed to carbon
disulfide concentrations about 8-11 mg/m3 (maximum approximately
20-25 mg/m3), for 10-15 years, compared with 390 control subjects.
For example, sensory polyneuritis was much more frequent and the
threshold for pain was significantly higher in the exposed group.
These studies indicate that background concentrations below about
60 mg/m3 (20 ppm) may cause neurological dysfunction.
7.3.2 Cardiovascular effects
Severe vascular effects manifesting themselves as
vasculoencephalopathy and renal atherosclerosis developed with
long-term exposure to carbon disulfide concentrations exceeding
150-300 mg/m3 (50-100 ppm) (Vigliani, 1954; Nofer et al., 1961).
Excess incidence of coronary heart disease has been reported with
longstanding exposure to concentrations averaging 30-125 mg/m3
(10-40 ppm) (Hernberg et al., 1970, 1973, Tolonen et al., 1975). The
mortality rate from coronary heart disease appeared to be most
affected while the incidence of milder manifestations such as angina
and ECG-changes was less influenced. In fact, most studies, published
so far, have failed to reveal any difference in the prevalence of
ECG-findings between exposed and unexposed workers (Tolonen et al.,
1976).
Quite recently, Vertin (1977) reported no difference between the
incidence of coronary heart disease found in control subjects and that
in workers exposed to about 30-95 mg/m3 (10-30 ppm) suggesting a
no-observed effect level for coronary heart disease in this range.
This finding does not necessarily contradict those referred to above;
possibly, the true exposure levels responsible for the excess coronary
heart disease in the Finnish studies were higher than those in
Vertin's study. It is probable that, in general, differences in actual
personal exposure levels in the studies-not necessarily those levels
measured and reported -- account for seeming discrepancies in results
and that exposure data are inadequate in most published studies.
a Tuttle et al., 1976. Unpublished report. (See footnote to section
6.3.10.1.)
Differences in blood pressure have been reported by Hernberg et
al. (1970) and Tolonen et al. (1975) between workers exposed to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) and
unexposed subjects, and by Martynova et al. (1976) in workers exposed
for 10-15 years to concentrations of about 10 mg/m3. These findings
indicate that effects upon the vascular system begin at low exposure
levels.
7.3.3 Ophthalmological effects
Among Japanese workers, the occurrence of retinal microaneurysms
was related to the duration of exposure (Sugimoto et al., 1976). The
exposure levels in earlier Japanese studies were not reported; in
present studies they are probably of the order of 60 mg/m3 (20 ppm).
The findings of retinal microaneurysms appears to be confined to
Japanese workers, since, according to a recent joint study, no such
effect could be demonstrated in Finns (Sugimoto et al., 1977). In
contrast, Finnish workers showed delayed peripapillary filling and
wider calibre of retinal arteries compared with control subjects
(Raitta et al., 1974). Exposure conditions under which these changes
occurred are described in section 6.3.2 and indicate that haemodynamic
changes in the ocular circulation are induced as early as coronary
effects.
7.3.4 Gonadal effects
The results of Vasiljeva (1973), Petrov (1969), and Finkova et
al. (1973) suggest that carbon disulfide concentrations below
10 mg/m3 (3 ppm) may cause disturbances in the ovarian function of
young females, resulting in menstrual disorders, pregnancy
complications, and spontaneous abortions. Given that the exposure
measurements reported are comparable with those of other studies, it
would seem that these would be the earliest detectable type of effects
in human beings.
A summary of the most relevant exposure-effect and exposure-
response findings is given in Table 3.
Table 3. Some exposure-effect and exposure-response relationships
for long-term exposure of man to carbon disulfide.
Concentration Exposure Symptoms & signs Reference
(mg/m3) time
(years)
10 5-10 Blood progesterone depressed, Vasiljeva (1973)
estriol increased, irregular
menstruation;
10 10-15 Sensory polyneuritis, increased Martynova et al. (1976)
pain threshold;
30 >3 Spontaneous abortions twice as Petroy (1969)
common and premature births 3
times as common as in controls;
30-120 >10 Coronary mortality about 5 times Tolonen et al. (1975)
that in controls, life expectancy Hernberg et al. (1973)
decreased 0.9-2.1 years depending Nurminen (1976)
on age; nonfatal infarctions 2.6 Hernberg et al. (1970)
times more frequent than in controls;
angina pectoris about twice as
prevalent as in controls; slightly
higher systolic and diastolic blood
pressure than in controls;
30-120 >6 Peripheral nervous and CNS Seppäläinen & Tolonen
dysfunction, conduction velocities (1974); Tolonen (1974)
slowed, psychological changes;
100-140 2-12 Decrease of testosterone and Cavalleri et al. (1970)
gonadotrophin LH in urine;
200-500 1-9 Ophthalmic pressure 18.4/14.7 Maugeri et al. (1966,
vs 15.3/11.6 kPa (138/110 vs 1967)
115/87 mmHg) in controls;
500-2500 0.5 Polyneuritis, myopathy acute Vigliani (1946)
psychoses
8. CONTROL OF EXPOSURE IN THE VISCOSE INDUSTRY
Technical and administrative measures for the protection of the
health of workers exposed to carbon disulfide in the production of
viscose rayon include:
-- exchange of production methods (substitution or elimination);
-- enclosure and local exhaust ventilation of the technological
process;
-- dilution (general) ventilation of the work room;
-- isolated and well-ventilated rooms for process control
operators and for rest periods;
-- use of personal respiratory protective equipment under certain
hazardous circumstances and,
-- rotation of workers periodically to areas free from carbon
disulfide exposure.
New developments such as the SINI viscose process make it
possible to reduce the total consumption of carbon disulfide to 70-80%
of that used in conventional processes (Sihtola, 1976). Occupational
health aspects must be taken into consideration at the planning stage
in the construction of new plants.
Enclosure of processes and machines is the second most effective
means of technical prevention. This technique should be applied in the
sulfidizing process and for spinning and washing machines. Possible
leakages in closed systems should be controlled by gas detectors. The
air removed from the processes by the exhaust ventilation should be
transported to a recovery system. The benefits of such a system are
both economic, since the plant has an interest in the recovery of
useful material, and hygienic, as the possibility of the pollution of
both the working rooms and the environment around the factory is
reduced. The air removed by exhaust ventilation is compensated with
pure air, which is brought to working sites by ventilation systems.
It is not possible to protect the workers in all situations by
ventilation systems alone. In certain circumstances, the workers may
be exposed to very high concentrations of carbon disulfide, in which
case respiratory protective devices must be used. The type of
respirator may vary from a chemical cartridge (organic vapour)
respirator, used for low concentrations, to supplied air or
self-contained breathing devices for very high concentrations. It is
also important to provide special isolated rooms with a slightly
positive pressure, compared with the work area contaminated with
carbon disulfide, for process control workers and for rest pauses.
Another factor of concern in the control of exposure and uptake
of carbon disulfide involves skin absorption resulting from direct
contact with the liquid. Where workers may come into contact with
liquid carbon disulfide, appropriate protective clothing, such as
synthetic rubber gloves and goggles or face shields, should be worn.
If carbon disulfide is spilled, potential sources of ignition
should be eliminated immediately, spark-proof ventilation provided,
and the spill cleaned up. A small spill should generally be allowed to
evaporate under conditions of good air circulation. However, large
spills should be covered with water and flushed into a retention basin
under a water layer. Carbon disulfide should not be drained into a
sewer system because of the possibility of an explosion.
In addition to these control methods, data obtained through the
biological monitoring of exposed workers can be used to determine
excessive uptake situations that require additional control measures.
The procedure for biological monitoring is discussed in section 6.5.
REFERENCES
ABRAMOVA, J. I. (1967) The questions of pathogenesis, specific
prevention and therapy of carbon disulfide intoxication. In:
Brieger, H. & Teisinger, L, ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 32-34.
ABUCEWICZ, A., SAMOCHOWIEC, L., & SZYSZKA, K. (1971) The role of
5-hydroxytryptamine in the course of experimental carbon
disulfide poisoning. Roca. Pomor. Akad. bled. Szczec, Suppl. 4,
pp. 89-96.
AGRANOVSKIJ, Z. M. & GOLOSCAPOV, O. D. (1972) [Hygienic assessment of
preventive diet for subjects exposed to carbon disulfide.] Gig.
i Sanit., 37 (II): 43-47 (in Russian).
AMERICAN STANDARDS ASSOCIATION (1941) Allowable concentration of
carbon disulfide, New York, ASA, 141, 7 pp. (ASA Z 37.3-1941).
ANDREEVA, N. B. (1970) [Experimental material for the diagnosis of
carbon disulfide poisoning, on the harmful effect of the poison
at the maximum permissible level for workroom air, and on the
passage of carbon disulfide in solution through the skin.] In:
Voprosy diagnostiki, specificeskoj profilaktiki i terapii
professional 'nyh zabolevanij, Leningrad, pp. 40-52 (in
Russian).
ANDRUSZCZAK, K. (1967) [The behaviour of ceruloplasmine level in the
blood serum in workers exposed to the carbon disulfide action.]
Med. Pr., 18:120-127 (in Polish).
ATTINGER, E. (1948) [Chronic carbon disulfide intoxication appearing
under the unusual symptoms of a heavy vascular disease.]
Schwiez. Med. Wochenscht., 78:667-669 (in German).
ATTINGER, E. (1954) [Chronic carbon disulfide intoxication and its
pathogenesis, its early detection and prevention.] Schweiz. Med.
Wochenschr., 84:912-913 (in German).
AUDIO-GIANOTTI, G. B. (1932) [Parkinsonism caused by professional
carbon disulphide poisoning.] Presse med., 40 (July-Dec.)
1289-1291 (in French).
AUSTONI, M. & D'AGNOLO, B. (1957) [Chronic carbon disulfide poisoning
and diabetes mellitus.] Minerva Med., 48:2500-2504 (in
Italian).
BAGON, D. A., CROSBY, M. T., HARDY, H. L., & SIMMONS, J. H. (1973) A
personal sampling technique for simultaneous determination of
hydrogen sulfide and carbon sulfide in air. Ann. occup. Hyg.,
16: 133-139.
BAIKOW, E. K. (1963) [Experimental data on the determination of the
maximum permissible concentration of carbon disulfide and
hydrogen disulfide in the atmosphere in their simultaneous
presence.] Gig. i Sanit., 28 (3): 3-8 (in Russian).
BARD, C. L. (1892) Cases in practice, malignant pustle and insanity
due to bisulphide of carbon. South Cal. Pract., 7: 476-485.
BARILJAK, I. R., VASILEVA, I. A., & KALINOVSKAJA, L. P. (1975) [Effect
of small concentrations of carbon disulfide and hydrogen sulfide
on intrauterine development in rats.] Arkh. Anat. Gistol.
Embriol., 58:77-81 (in Russian).
BARTONICEK, V. (1957) [The distribution of carbon disulfide in whole
blood, the brain and the suprarenal glands over a given period in
parenteral administration in white mice. Prac. Lek., 9:28-30
(in Czech).
BARTONICEK, V. (1958) [The distribution of free carbon disulphide and
bound carbon disulphide liberated by acid hydrolysis in the
organs of white rats.] Prac. Lek., 10: 334-338 (in Czech).
BARTONICEK, V. (1959) [Distribution of free carbon disulfide and bound
carbon disulfide liberated by acid hydrolysis in the organs of
white rats.] Prac. Lek., 10:504-510 (in Czech).
BASHORE, R., HAMILTON, A., LEWEY, F., ERSKINE, L., HAMMOND, J., HOUTZ,
R., BRACELAND, F., MCDONALD, R., BATSON, O., BELLET, S.,
CRESKOFF, F., EHRICH, W., & ALPERS, B. J. (1938) Survey of
carbon disulfide and hydrogen sulfide hazards in the viscose
rayon industry, Harrisburg, PA, Occupational Disease Prediction
Division, US Department of Labor and Industry p. 68 (Bull. 46).
BINET, L. & BOURLIERE, F. (1944) Sur les modifications du sang au
cours du sulfocarbonisme chronique. Arch. Mal. Prof., 6: 12-16.
BITTERSOHL, G. (1971) On relationships in action between carbon
disulfide and hydrogen sulfide. Med. Lavoro, 62: 554-556.
BITTERSOHL, G. & THIELE, H. (1977) The health of workers exposed to
carbon disulphide in the production of artificial fibres. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski (Finland), 6-10 June 1977, p. 5.
BLACKBURN, H., KEYS, A., SIMONSON, E., RAUTAHARJU, P., & PUNSAR, S.
(1960) The electrocardiogram in population studies. A
classification system. Circulation, 21: 1160-1175.
BOBSIEN, K. (1954) [New method for determination of uptake and binding
by endogenous substances.] Arch. Gewerbepath. Gewerbehyg., 13:
193-203 (in German).
BOND, E. J. & DE MATTEIS, F. (1969) Biochemical changes in rat liver
after administration of carbon disulphide, with particular
reference to microsomal changes. Biochem. Pharmacol., 18:
2531-2549.
BOND, E. J., BUTLER, W. H., DE MAITEIS, F., & BARNES, J. M. (1969)
Effects of carbon disulphide on the liver of rats. Br. J. ind.
Med, 26: 335-337.
BRIEGER, H. (1961) Chronic carbon disulfide poisoning. J. occup.
Med., 8: 302-308.
BRIEGER, H. (1967) Carbon disulphide in the living organism-Retention,
metabolism and physiological effects. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 27-31.
BRIEGER, H. & FRIEDMAN, M. H. F. (1949) In vitro effects on
digestive enzymes, theory and pathology of poisoning. J. ind.
Hyg., 31: 96-98.
BRIEGER, H. & TEISINGER, J., ed. (1966) Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 27-31.
BRUCE, A. (1884) Chronic poisoning by CS2. Edinburgh Med. J., 29:
1009.
BÜSING, K. H. (1952) [The determination of minute amounts of carbon
disulfide in blood.] Zentralbl. Arbeitsmed., 2: 115-122 (in
German).
BÜSING, K. H. & SONNENSCHEIN, W. (1954) [The binding of carbon
disulfide in the organs.] Zentralbl. Arbeitsmed. Arbeitschutz,
4:65-69 (in German).
BÜSING, K. H., SONNENSCHEIN, W., BECHER, E. W., & DREIHELLER, H.
(1953) [Experimental studies in animals on the binding of carbon
disulfide in organs using labelled carbon disulfide (C35S2).]
Z. Naturforsch., 86: 495-500 (in German).
CANDURA, F., GRAVERI, A., & STRANEO, G. (1962) [Clinical contribution
to studies on the relationship between arteriosclerosis and
carbon disulfide poisoning.] Folia Med., 45: 749-769 (in
Italian).
CAVALLERI, A. (1972) [Alterations in the endocrinological function as
an early sign of carbon disulfide poisoning.] Med. Lavoro, 63:
81-84 (in Italian).
CAVALLERI, A. (1975) Serum thyroxine in the early diagnosis of carbon
disulfide poisoning. Arch. environ. Health, 30: 85-87.
CAVALLERI, A. & ZUCCATO, F. (1965) [Endocrinological observations on
carbon disulfide poisoning. I: Adrenocortical function.] Folia
Med., 48: 519-527 (in Italian).
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1965) [Adrenocortical and
gonadal function in human carbon disulfide poisoning.] In:
Proceedings of the 28th National Congress on Occupational
Health, Naples, 1965, pp. 645-660 (in Italian).
CAVELLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1966a) Endocrinological findings in young workers
exposed to carbon disulphide. I. - Urinary excretion of total
17-ketosteroids. Med. Lavoro, 57: 566-572.
CAVALLERI, A., DJURIC, D., VISCONTI, E., REZMAN, I., MAUGERI, U., &
BRANKOVIC, D. (1966b) Endocrinological findings in young workers
exposed to carbon disulphide. II.-Urinary excretion of
17:hydroxy-corticosteroids. Med. Lavoro, 57: 573-578.
CAVALLERI, A., DJURIC, D., VISCONTI, E., BRANKOVIC, D., & MAUGERI, U.
(1966c) Androsterone and etiocholanolone in the urine of young
workers exposed to carbon disulphide. In: Proceedings of the XV
International Congress on Occupational Health, Vienna, 19-24,
1966, Vienna, Wiener Med. Akademie, Vol. II-I, pp. 347-353.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., & VISCONTI, E.
(1966d) Endocrinological findings in young workers exposed to
carbon disulphide. III.-Urinary excretion of 3alpha,
11-deoxy-17-ketosteroids. Med. Lavoro, 57: 755-760.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1967) 17-ketosteroids and 17-hydroxycorticosteroids
in the urine of young workers exposed to carbon disulphide. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 86-91.
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1970) L'excretion urinaire
de testosterone et de gonadotrophine stimulant les cellules
interstitielles (ICSH) chez des subjets exposes au sulfure de
carbone. Arch. Mal. Prof., 31: 23-30.
CAVALLERI, A., TACCOLA, A., GRAOVAC-LEPOSAVIC, LJ., MAUGERI, U., &
DJURIC, D. (1971) [Thyroinaemia in subjects exposed to carbon
disulfide.] Lav. Um., 23: 183-187 (in Italian).
CAVALLERI, A., POLATTI, F., & BOLIS, P. F. (1977) Acute effects of
tetraethylthiuram disulphide on serum levels of hypophyseal
hormones in humans. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski (Finland), 6-10
June 1977, p. 28.
CHERVENKA, C. H. & WILCOX, P. E. (1956) Chemical derivative of
chymotrypsinogen: I: Reaction with carbon disulfide. J. biol.
Chem. 222: 621-634.
CIMBAREVIC, T. K. (1970) [Ceruloplasmin activity in persons exposed to
carbon disulfide.] In: Voprosy diagnostiki, specifideskoj
profilaktiki i terapii professional 'nyh zabolevanij,
Leningrad, 65-67 (in Russian).
CIRLA, A.M., VILLA, A., & TOMASINI, M. (1972) [Study on the incidence
of coronaropathy in workers exposed to carbon disulfide in a
viscose rayon industry.] Med. Lavoro, 63: 431-441 (in Italian).
COHEN, A. E., PAULUS, H. J., KEENAN, R. G., & SCHEEL, L. D. (1958)
Skin absorption of carbon disulfide vapor in rabbits. I.
Associated changes in blood protein and zinc. Arch. ind. Health,
17: 164-169.
COHEN, A. E., SCHEEL, L. D., KOPP, J. F., STOCKELL, F. R., KEENAN, R.
G., MOUNTAIN, J. T., & PAULUS, H. J. (1959) Biochemical
mechanisms in chronic carbon disulfide poisoning. Am. Ind. Hyg.
Assoc. J., 20: 303-323.
CREPET, M., COBBATO, F., & SCANSETTI, G. (1956)] Some clinical
observations on vasculopathy resulting from chronic carbon
disulfide poisoning.] Minerva Med., 47: 379-387 (in Italian).
CULLEN, T. E. (1964) Spectrophotometric determination of
dithiocarbamates residues on food crops. Anal. Chem., 36:
221-224.
DALVI, R. R., POORE, R. E., & NEAL, R. A. (1974) Studies of the
metabolism of carbon disulfide by the rat liver microsomes. Life
Sci., 14: 1785-1796.
DANILOVA, T. N. (1968) [Data on the effect of carbon disulfide on the
coagulation system of the blood.] In: Materially 2-oj
konferencii molodyh naucnyh rabotnikov, Leningrad, pp. 102-104
(in Russian).
DAVIDSON, M. & FEINLEIB, M. (1972) Carbon disulfide poisoning: A
review. Am. Heart J., 83: 100-114.
DELIC, V., GRAOVAC-LEPOSAVIC, LJ., DJURIC, D., BERKES, I., &
DJORDJEVIC, V. (1966) The determination of sulphates in the urine
of workers exposed to carbon disulphide. Med. Lavoro, 57:
458-462.
DELPECH, A. (1856a) Notes sur les accidents que developpe, chez les
ouvriers en caoutchouc, l'inhalation du CS2 en vapeur. Gaz.
Hebd. med. Chir., 3: 40.
DELPECH, A. (1856b) Accidents que developpe, chez les ouvriers en
caoutchouc, l'inhalation du CS2 en vapeur. Union bled., 10:
265.
DELPECH, A. (1863) Industrie du caoutchou de souffle: Recherches sur
l'intoxication speciale que determine le CS2. Ann. Hyg. 2e
sér., 19: 65.
DE MATTEIS, F. (1974) Covalent binding of sulfur to microsomes and
loss of cytochrome P-450 during the oxidative desulfuration of
several chemicals. Mol. Pharmacol., 10: 849-854.
DE MATTEIS, E. & SEAWRIGHT, A. A. (1973) Oxidative metabolism of
carbon disulfide by the rat. Effect of treatments which modify
the liver toxicity of carbon disulfide. Chem. biol. Interact.,
7: 375-388.
DEMUS, H. (1964) [On the reception, chemical transformation and
excretion of carbon disulfide by the human body.] Arch.
Gewerbepath. Gewerbehyg., 20: 507-536 (in German).
DEMUS, H. (1967) The mechanism of absorption, metabolism and excretion
of carbon disulfide in the human body. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 42-49.
DE NICOLA, P., NICROSINI, F., FASOTTI, C., & DEL FAVERO, A. (1962)
[Comparative studies on the physiopathological picture of
experimental chronic carbon disulfide poisoning and
high-cholesterol diet I. Study of the lipid fractions in serum
and aorta.] G. Geront., 10: 207-220 (in Italian).
DEPARTMENT OF EMPLOYMENT AND PRODUCTIVITY (1968) Methods for
determination of toxic substances in air: Carbon disulfide
vapour. London, HMSO, 10 pp. (DEP Booklet No. 6).
DJERASSI, L. S. & LUMSROSO, R. (1968) Carbon disulphide poisoning with
increased ethereal sulphate excretion. Br. J. ind. Med., 25:
220-222.
DJURIC, D. (1963) [Metabolism of carbon disulphide.] Arh. Hig. Rada,
14: 23-31 (in Serbo-Croat).
DJURIC, D. (1967) Determination of carbon disulphide and its
metabolites in biological material. In: Brieger, H. & Teisinger,
J., ed. Toxicology of carbon disulphide, Amsterdam, Excerpta
Medica Foundation, pp. 52-61.
DJURIC, D. (1971) Some aspects of antabuse, carbon disulphide and
ethyl alcohol metabolism. Arh. Hig. Rada, 22: 171-177.
DJURIC, D., SURDUCKI, N., & BERKES, I. (1965) Iodine-azide test on
urine of persons exposed to carbon disulphide. Br. J. ind. Med.,
22: 321-323.
DJURIC, D., STOJADINOVIC, LJ., BOJOVIC, V., & REZMAN, I. (1967)
Excretion of zinc in the urine of persons exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation, pp.
118-121.
DJURIC, D., POSTIC-GRUJIN, A., GRAOVAC-LEPOSAVIC, L., & DELIC, V.
(1973) Disulfiram as an indicator of human susceptibility to
carbon disulfide excretion of diethyldithiocarbamate sodium in
the urine of workers exposed to CS2 after oral administration of
disulfiram. Arch. environ. Health, 26: 287-289.
DROGCINA, E. A. (1968) [Carbon disulfide poisoning.] In: Professional
'nye bolezni sistemy, Leningrad, Medicina, pp. 129-141 (in
Russian).
DUTKIEWICZ, T. & BARANOWSKA, B. (1967) The significance of carbon
disulphide skin resorption in the evaluation of exposure. In:
Brieger, H. & Teisinger, J., ed. Toxicology of Carbon
Disulphide, Amsterdam, Excerpta Medica Foundation, pp. 50-51.
EFREMOVA, N. F. & UZBEKOV, G. A. (1968) [Study of nitrogen metabolism
in the brains of white rats exposed to chronic carbon disulfide
poisoning.] In: Trudy Rjazanskogo Medicinskogo Instituta, 34:
163-172 (in Russian).
EHRHARDT, W. (1967) Experiences with the employment of women exposed
to carbon disulphide. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 240-244.
EL-GAZZAR, R., EL-SADIK, Y. M., & HUSSEIN, M. (1973) Changes in zinc
and serum proteins due to carbon disulphide exposure. Br. J.
ind. Med., 30: 284-288.
FAHIM, B. A., FAHMY, A.M., & KAMEL, M. Y. (1973) [Biochemical and
hematologic studies on carbon disulfide poisoning.] Z. Ges.
Hyg., 19: 538-541 (in German).
FAITH, W. L., KEYES, D. B., & CLARK, R. L. (1965) Industrial
Chemicals, 3rd ed., New York, John Wiley & Sons, pp. 226-227.
FERRERO, G. P. (1969) [Occurrence of diabetes in workers exposed to
carbon disulfide.] Med. Lavoro, 60: 38-42 (in Italian).
FERRARO, A., JARVIS, G., & FLICKER, D. (1.941) Neuropathologic changes
in experimental carbon disulfide poisoning in cats. Arch.
Pathol., 32: 732-738.
FINKOVA, A., SIMKO, A., JINDRICHOVA, J., KOVARIK, J., PREININGEROVA,
O., KLIMOVA, A., & KORISKO, F. (1973) [Gynaecological problems of
women working in an environment contaminated with carbon
disulfide.] Cesk. gynekol., 38: 535-536 (in Czech).
FINULLI, M. & GHISLANDI, E. (1959) [Occurrence of diabetes in chronic
carbon disulfide poisoning.] Med. Lavoro, 50: 189-192 (in
Italian).
FISCHER, J. & MICHALOVA, C. (1956) [Neurohistological changes caused
by carbon disulfide.] Prac. Lek., 8:16-19 (in Czech).
FOA, V., CASSITO, M. G., FORZI, M., & BULGHERONI, C. (1976) Mental
performance and personality disorders among workers exposed to
carbon disulphide: Comparison between two different rayon plants.
In: Horvarth, M., ed. Adverse effects of environmental chemicals
and psychotropic drugs, neurophysiological and behavioral tests.
Amsterdam, Oxford, New York, Elsevier, Vol. 2, pp. 173-182.
FOREMAN, W. (1886) Notes of a fatal case of poisoning by bisulphide of
carbon. Lancet, 2: 118-119.
FRANCINE, A. P. (1905) Acute carbon disulfide poisoning. Am. Med.,
9: 871.
FRANTIK, E. (1970) The development of motor disturbances in
experimental chronic carbon disulphide intoxication. Med.
Lavoro,, 61: 309-313.
FREUNDT, K. J. (1973) The inhibitory action of inhaled carbon
disulfide on microsomal enzymes as a sensitive restriction of
liver function. In: Horvath, M., ed. Adverse effects of
environmental chemical and psychotropic drugs: Quantitative
interpretation of functional tests, Amsterdam, Oxford, New
York, Elsevier, pp. 161-164.
FREUNDT, K. J. (1974) Ethanol and acetaldehyde levels in alcoholized
rats and man under carbon disulphide exposure. Abstracts of the
III International Symposium on the Toxicology of Carbon
Disulphide, Cairo-Alexandria (Egypt) 4-9 May, 1974, p. 28.
FREUNDT, K. J. (1975) [Carbon disulfide.] In: Henschler, D., ed.
Gesundheitsschadliche Arbeitstoffe, 4th ed., Federal Republic
of Germany, Verlag Chemic Weinheim, pp. 1-20.
FREUNDT, K. J. (1977) Animal experimentation studies on a possible
induction of hepatic microsomal mixed-function oxygenases (MFO)
by inhaled carbon disulphide (CS2). In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 24.
FREUNDT, K. J. & DREHER, W. (1969) Inhibition of drug metabolism by
small concentrations of carbon disulfide. Nauyn-Schmiedebergs
Arch. Pharmakol., 263: 208-209.
FREUNDT, K. J. & HENSCHLER, D. (1971) Inhibition of oxidative drug
metabolism by low doses of inhaled carbon disulfide: Kinetics and
specificity. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Bahja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 9.
FREUNDT, K. J. & KÜRZINGER, R. (1975) Energy potential and hepatic
function in rats under acute exposure to carbon disulphide. Int.
Arch. Arbeitsmed., 34: 269-282.
FREUNDT, K. J. & KUTTNER, P. (1969) Oxidative drug metabolism
following low carbon disulphide concentrations.
Nauyn-Schmiedebergs Arch. Pharmakol., 264: 232-233.
FREUNDT, K. J. & LIEBERWIRTH, H. (1974) Influence of inhaled carbon
disulfide on acetaldehyde production and liver function in
alcoholized man. Nauyn-Schmiedebergs Arch. Pharmakol., 282
(Suppl. R.21).
FREUNDT, K. J. & NETZ, H. (1973) Influence of carbon disulfide and
thiurams on ethanol elimination and acetaldehyde production.
Arch. Pharmakol. exp. Pathol., 277 (Suppl. R18).
FREUNDT, K. J., LIEBALDT, G. P., & SIEBER, K. H. M. (1974a) Effect of
barbiturates on the liver of rats exposed to carbon disulphide
vapor. Int. Arch. Arbeitsmed., 32: 297-303.
FREUNDT, K. J., SCHAUENSURG, K. J., & EICHHORN, P. (1974b) Effect of
acute exposure to carbon disulfide vapor upon some components of
the hepatic microsomal enzyme system in rats. Arch. Toxicol.,
32 (3): 233-240.
FREUNDT, K. J., SCHNAPP, E., & DREHER, W. (1975) Pharmacokinetics of
inhaled carbon disulphide in rats in relation to its inhibitory
effect on the side-chain oxidation of hexobarbital. Int. Arch.
occup. environ. Health, 35: 173-185.
FREUNDT, K. J., LIEBERWIRTH, K., NETZ, H., & POHLMANN, E. (1976) Blood
acetaldehyde in alcoholized rats and humans during inhalation of
carbon disulphide vapor. Int. Arch. occup. environ. Health, 37:
35-46.
FRUSCELLA. R. (1962) [Urinary excretion of 17-ketosteroids and
17-hydroxycorticosteroids in human subjects suffering from carbon
disulfide poisoning.] Clin. ter., 23: 749-755 (in Italian).
GADASKINA, I.D. & ANDREEVA, N. B. (1960) [Some biochemical shifts in
the organism in carbon disulfide poisoning. Free SH groups and
ceruloplasmin in the blood: Copper and zinc metabolism.] Gig.
Tr. Prof Zabol., 13: 28-32 (in Russian).
GAVRILESCU, N. & LILIS, R. (1967) Cardiovascular effects of
long-extended carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 165-167.
GHERASE, G. (1976) Psychological changes in workers exposed to
atmospheres containing carbon disulphide. In: Adverse effects of
environmental chemicals and psychotropic drugs.
Neurophysiological and behavioral tests, Amsterdam, Vol. 2,
334 pp.
GILIOLI, R., BULGHERONI, C., BERTAZZI, P. A., CIRLA, A. M., CASSITTO,
M. G., & TOMASINI, M. (1977) Endocrinological, psychological and
neurological study of carbon disulphide workers. III.
Neurological and neurophysiological findings. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), June 6-10, 1977, p. 13.
GOLDBERG, M. E., JOHNSON, H. E., POZZANI, U. C., & SMYTH, H. F. (1964)
Behavioral response of rats during inhalation of trichlorethylene
and carbon disulphide vapours. Acta Pharmacol., 21: 36-44.
GONDZIK, M. (1971) Histology and histochemistry of rat testicles as
affected by carbon disulphide. Polish med. J., 10: 133-139.
GORDY, S. T. & TRUMPER, M. (1938) CS2 poisoning with a report of six
cases. J. Am. Med. Assoc., 110: 1543.
GORDY, S. T. & TRUMPER, M. (1940) Carbon disulphide poisoning-Report
of 21 cases. Ind. Med., 9: 231-234.
GORNY, R. (1971) The level of pyridoxal phosphate (PLP) in the blood
plasma of rats exposed to carbon disulfide. Biochem. Pharmacol.,
20: 2114-2115.
GORNY, R. (1974) [Effect of prolonged exposure to carbon disulfide on
tryptophan metabolism in rats.] Med. Pr., 25: 21-27 (in
Polish).
GOTO, S. & HOTTA, R. (1967) The medical and hygienic prevention of
carbon disulphide poisoning in Japan. In: Brieger, H. &
Teisinger, J.: ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 219-230.
GOTO, S., HOTTA, R., & SUGIMOTO, K. (1971) Studies on chronic carbon
disulfide poisoning. Int. Arch. Arbeitsmed., 28: 115-126.
GOTO, S., SUGIMOTO, K., HOTTA, R., BABA, T., NAKAMURA, G., & SEKI, Y.
(1977) Regional patterns of angiopathy due to CS2. Egypt. J.
occup. Med., 5: 155-159.
GRAOVAC-LEPOSAVIC, LJ. & JOVICIC, M. (1971) Report on one case of
chronic carbon disulfide intoxication. In: Abstracts of the H
International Symposium on Carbon Disulphide Toxicology, Banjo
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 19.
GRAOVAC-LEPOSAVIC, LJ., DJURIC D., PAVLOVIC, A., & JOVICIC, M. (1967)
The use of iodine-azide test for early diagnosis of carbon
disulphide poisoning. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 62-69.
GRAOVAC-LEPOSAVIC, LJ., JOVICIC, M., SAVIC, S., DJURIC, D., &
POSTIC-GRUJIN, A. (1977) Findings in workers some years after
CS2 poisoning. Egypt. J. occup. Med., 5: 53-60.
GREGORCZYK, J., KOSMIDER, K., WIECKOWSKI, B., & WESOLOWSKA, T. (1975a)
Investigations on the extent of the liver lesion in the rat by
experimental inhalation poisoning with carbon disulfide. I.
Analysis of biochemical changes in the blood serum. Int. Arch.
Arbeitsmed., 34: 65-80.
GREGORCZYK, J., WIECKOWSKI, B., & WESOLOWSKA, T. (1975b)
Investigations on the extent of liver lesion in rats by
experimental inhalation poisoning with carbon disulfide. II.
Analysis of biochemical changes in liver homogenate and its
fractions. Int. Arch. Arbeitsmed., 34: 151-166.
GUARINO, A. & ARCIELLO, G. (1954) [Histological study on coronary
arteries in chronic experimental carbon disulfide poisoning.]
Folia Med. (Napoli) 37: 1022-1044 (in Italian).
HAKKY, U. (1977) Physiological manifestations after exposure to CS2.
Egypt. J. occup. Med., 5: 47-52.
HAMILTON, A. (1925) Industrial poisons in the United States, New
York, Macmillan Co., 590 pp.
HAMILTON, A. (1940) Occupational poisoning in the viscose rayon
industry, Washington, DC, Department of Labor, pp. 1-79 (Bull.
No. 34).
HÄNNINEN, H. (1971) Psychological picture of manifest and latent
carbon disulphide poisoning. Br. J. ind. Med., 28: 374-381.
HÄNNINEN, H. (1974) Behavioral study of the effects of carbon
disulfide. In: Xintaras, C. El, Johnson, B., De Groot, I., ed.
Behavioral toxicology, Washington, DC, Department of Health,
Education, & Welfare, pp. 72-79.
HÄNNINEN, H., NURMINEN, M., TOLONEN, M., & MARTELIN, T. (1978)
Psychological tests as indicators of excessive exposure to carbon
disulfide. Scand. J. Psychol., 19(2): 163-174.
HARA, I. (1958) [On the carbohydrate metabolism in chronic carbon
disulfide poisoning.] Q. J. labour Res. Jpn, 6: 1-20 (in
Japanese).
HARASHIMA, S. & MASUDA, Y. (1962) Quantitative determination of
absorption and elimination of carbon disulfide through different
channels in human body. Int. Arch. Gewerbepath. Gewerbehyg.,
19: 263-269.
HARASHIMA, S., TOYAMA, T., & SAKURAI, T. (1960) Serum cholesterol
level of viscose rayon workers. Keijo J. Med., 9: 81-90.
HASSMAN, P., SIMKO, A., JINDRICHOVA, J., HEROUT, V., & HRADSKY, M.
(1967) The problem of chronic gastritis in artificial fibres
industry. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation,
pp. 182-189.
HERNBERG, S. & NORDMAN, C-H. (1969) Failure to find increased
excretion of zinc or magnesium in humans exposed to carbon
disulphide. Med. Lavoro, 60: 163-168.
HERNSERG, S., MOWE, G., VIRKOLA, P., PARTANEN, T., & NORDMAS, C-H.
(1969) Magnesium and zinc values of erythrocytes and plasma for
workers exposed to carbon disulfide. Work Environ. Health, 6:
9-13.
HERNSERG, S., PARTANEN, T., NORDMAN, C-H., & SUMARI, P. (1970)
Coronary heart disease among workers exposed to carbon
disulphide. Br. J. ind. Med., 27: 313-325.
HERNBERG, S., NORDMAN, C-H., PARTANEN, T., CHRISTIANSEN, V., &
VIRKOLA, P. (1971) Blood lipids, glucose tolerance and plasma
creatinine in workers exposed to carbon disulphide. Work
Environ. Health, 8: 11-16.
HERNBERG, S., NURMINEN, M., & TOLONEN, M. (1973) Excess mortality from
coronary heart disease in viscose rayon workers exposed to carbon
disulfide. Work Environ. Health, 10: 93-99.
HERNBERG, S., TOLONEN, M., & NURMINEN, M. (1976) Eight-year follow-up
of viscose rayon workers exposed to carbon disulfide. Scand. J.
Work Environ. Health, 2: 27-30.
HIGASHIDA, T., YAMAMOTO, T., & SHINDO, S. (1959) [Electromyographic
study of the effects of carbon disulfide administration on spinal
reflex.] Brain Nerve, 11(4): 253-260 (in Japanese).
HOCKWIN, O. & SAVIC, S. (1968) [Reversibility of biochemical changes
in lease and retina of the rabbit's eye in carbon disulfide
poisoning.] Albrecht Graefs Arch. klin. exp. Ophthalmol., 175:
7-12 (in German).
HOLASOVA, P. (1969) [Observations on a group of children from an area
polluted by carbon disulfide and hydrogen sulfide exhalation
compared with a control group of children.] Cs. Hyg., 14:
260-265 (in Czech).
HORVATH, M. & MIKISKA, A. (1957) [The influence of carbon disulfide on
the CNS of rabbits as seen in the EEG.] Prat. Lek., 9: 22-28
(in Czech).
HOTTA, R. & GOTO, S. (1971) A fluoroscein angiographic study on
microangiopathia sulfocarbonica Jpn. J. Ophthal., 15: 132-139.
HOTTA, R. & SAVIC, S. (1972) Fluorescein angiographic and
ophthalmodynamometric study on chronic carbon disulfide
poisoning. Med. Lavoro, 63: 442-447.
HUEPER, M. C. (1936) Etiologic studies on the formation of skin
blisters in viscose workers. J. ind. Hyg., 18: 432-447.
IVANOVA, F. G. (1967) [Some data on the status of erythropoiesis on
exposure to low concentrations of carbon disulfide.] In: Voprosy
gigieny truda i profpatologii, Leningrad, pp. 247-248 (in
Russian).
JAKUBOWSKI, M. (1968) [Studies of the conversion of carbon disulphide
to compounds catalysing the iodine-azide reaction. I.
Chromatographic studies.] Med. Pr. 19: 244-251 (in Polish).
JAKUBOWSKI, M. ( 1971) [Studies on the carbon disulfide metabolism of
compounds catalyzing the iodine azide reaction.] Med. Pr. 22:
195-206 (in Polish).
JÄRVISALO, J. & DE MATTEIS, F. (1977) Carbon disulphide induced loss
of liver microsomal haem and cytochrome P-450. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 23.
JÄRVISALO, J., SAVOLAINEN, H., ELOVAARA, E., & VAINIO, H. (1977) The
in vivo toxicity of CS2 to liver microsomes: Binding of
labelled CS2 and changes of the microsomal enzyme activities.
Acta Pharmacol. Toxicol., 40: 329-336.
JUMP, H. D. & CRUICE, J. M. (1904) Chronic poisoning from bisulphide
of carbon. Univ. Penn. med. Bull., 17: 193-196.
JUNTUNEN, J., HALTIA, M., & LINNOILA, I. (1974) Histochemically
demonstrable non-specific cholinesterase as an indicator of
peripheral nerve lesion in carbon disulfide induced
polyneuropathy. Acta Neuropathol. (Berl.), 29: 361-366.
JUNTUNEN, J., LINNOILA, I., & HALTIA, M. (1977) Histochemical and
electron microscopic observations on the myoneural junctions of
rats with carbon disulfide induced polyneuropathy. Scand. J.
Work Environ. Health, 3: 1-15.
KARAJOVIC, D., GRAOVAC-LEPOSAVIC, LJ., & SURADNICI, V. J. (1964) [The
influence of CS2 on the health of workers in factory in Serbia.]
Arh. Hig. Rada, 15: 87-92 (in Serbo-Croat).
KLIMKOVA-DEUTSCHOVA, E. (1965) [Early diagnosis in industrial
neurology.] S.Z.N., Praha 1965, 260 pp. (in Czech).
KNAVE, B., KOLMODIN-HEDMAN, B., PERSSON, H. E., & GOLDBERG, J. M.
(1974) Chronic exposure to carbon disulfide: Effects on
occupationally exposed workers with special reference to the
nervous system. Work Environ. Health, 11: 49-58.
KOELSCHS HANDBUCH DER BERUFSERKRANKUNGEN (1972) Jena, Gustav Fischer
Verlag. Vol. II, pp. 270-275.
KOESTER, G. (1899) [Contribution to the field of chronic CS:
intoxication.] Arch. Psychiat., 32: 606 (in German).
KOLMODIN-HEDMAN, B. (1976) Iodine-azide test in a Swedish viscose
factory related to air concentration of CS2 and to health.
Arbete och Halea, No. 8. pp. 23-28.
KOMRSKA, M. (1967) L'importance de la rheoencephalographie pour le
diagnostic d'intoxication par le sulfure de carbone. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulfide,
Amsterdam, Excerpta Medica Foundation, pp. 140-144.
KOPECKY, J. (1973) [Metabolism of carbon disulfide in rats.] Acta
Hyg. Epidemiol. Microbiol. (Prague), Suppl. 12, pp. 3-6
(in Czech).
KOZIOL, A., KULCZYCKA, H., & ROZEWICKI, S. (1971) [Development of
children, whose mothers were professionally exposed to carbon
disulfide.] Med. Pr., 22: 511-514 (in Polish).
KROLIKOWSKA, W. & RUCINSKA, Z. (1959) EEG examinations in chronic
professional intoxication by carbon disulfide. Electrortceph.,
clin. Neurophysiol., 11: 180 (Abstract No. 9).
KUBOTA, J. (1967) Historical view of carbon disulfide poisoning in the
Japanese viscose rayon industry. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulfide, Amsterdam, Excerpta Medica
Foundation, pp. 192-196.
KUJALOVA, V. (1971) The effect of carbon disulfide on vitamin B6
metabolism. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Banja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 30.
KUJALOVA, V. (1973) [Changes in the pyridoxinemetabolism and in the
blood levels of ceruloplasmin in humans exposed to carbon
disulfide.] Acta Hyg. Epidemiol. Microbiol., Suppl. 12 (in
Czech).
KUJALOVA V. & TINTERA, J. (1970) The effect of carbon disulfide on
tryptophan metabolism. Med. Lavoro, 61: 227-231.
KÜRZINGER, R. & FREUNDT, K. J. (1969) Changes in hepatic functions
following low carbon disulfide concentration.
Naunyn-Schmiedebergs Arch. Pharmakol., 264: 261-262.
LANCRANJAN, I. (1972) Alterations of spermatic liquid in patients
chronically poisoned by carbon disulfide. Med. Lavoro, 63:
29-33.
LANCRANJAN, I., POPESCU, H. I., & KLEPSCH, I. (1969) Changes of the
gonadic function in chronic carbon disulfide poisoning. Med.
Lavoro, 60: 566-571.
LANCRANJAN, I., FLOREA, O., & PETRESCU, M. (1971) Corticosuprarenal
insufficiency as a feature of carbon disulfide poisoning. Med.
Lavoro, 62: 557-561.
LANCRANJAN, I., SUKMANSKY, M., STANUCA, L., ANTONESCU, C., & POPESCU,
H. I. (1972) Study of the thyroid function in chronic carbon
disulfide poisoning. Med. Lavoro, 63: 123-125.
LANGELEZ, A. (1946) Viscose et sulfure de carbone. Arch. Belg. Med.
Soc. Hyg. Rev. Pathol., 4: 67-85.
LAUDENHAIMER, R. (1899) [The carbon disulfide intoxication of rubber
workers.] Leipzig, Veit & Co., 168 pp. (in German).
LAURMAN, W. & WRONSKA-NOFER, T. (1977) [Effect of carbon disulfide on
the amount of cholesterol in the tissues of rats.] Med. Pr.,
28: 77-82 (in Polish).
LAZAREV, N.J., ABRAMOVA, ZH. I., & CERNY, Z. C. (1965) [On possible
adverse reactions following use of some drugs by persons handling
carbon disulfide.] Gig. Tr. Prof. ZaboL, 9: 24-27 (in Russian).
LEITHE, W. (1971) Determination of carbon disulfide at the work site.
The analysis of air pollutant, 2nd ed., Ann Arbor, MI, Humphrey
Science Pub., pp. 228-229.
LEWEY, F. H. (1941) Neurological, medical and biochemical signs and
symptoms indicating chronic industrial carbon disulfide
absorption. Ann. int. Med., 15: 869-883.
LEWEY, W. E., ALPERS, B. J., BELLET, S., GRESKOFF, A. J., DRABLEIN, D.
L., ERICH, W. E., FRANK, J. H., JONAS, L., MCDONALD, R.,
MONTGOMERY, E. J., & REINHOLD, J. G. (1941) Experimental chronic
poisoning in dogs, clinical, biochemical and pathological study.
J. ind. Hyg., 23: 415-436.
LILIS, R. (1974) Behavioral effects of occupational carbon disulfide
exposure. In: Xintaras, C., Johnson, B. L., & de Groot, I., ed.
Behavioral toxicology, early detection of occupational hazards,
Cincinnati, US DHEW, Public Health Service, Center for Disease
Control, National Institute for Occupational Safety and Health,
pp. 51-59 (NIOSH 74-126).
LINIECKI, J. (1960) The effect of chronic carbon disulfide poisoning
on thiamine balance in rats. J. Hyg. Epidemiol. Microbiol.
Immunol. 4: 212-222.
LINNOILA, I., HALTIA, M., SEPPÄLÄINEN, A.M., & PALO, J. (1975)
Experimental carbon disulphide poisoning. Morphological and
neurophysiological studies. In: Körney, S., Tariska, S., &
Gosztonyi, G., ed. Proceedings of the VII International Congress
on Neuropathology Académiai Kiado, Budapest 1-7 September 1975,
pp. 383-386.
LUKAS, E. (1969) Conduction speed of peripheral nerves in persons
exposed to carbon disulfide (Electromyography study). Int. J.
clin. Ther. Toxicol., 2: 354-358.
LUKAS, E. (1970) Stimulation electromyography in experimental
toxicology (Carbon disulphide neuropathy in rats). Med. Lavoro,
61: 302-308.
LUKAS, E. (1973) [Some important factors in the development of
experimental carbon disulfide neuropathy in rats.] Cs. Neurol.
Neurochir., 36: 169-173 (in Czech).
LUKAS, E. (1974) Copper and zinc levels in peripheral nerve tissues of
rats with experimental carbon disulfide neuropathy. In:
Abstracts of the III International Symposium on Toxicology of
Carbon Disulphide, Cairo-Alexandria, Egypt, 4-9 May, 1974,
p. 8.
LUKAS, E. & HROMADKA, M. (1977) Conduction velocity of the ulnar and
tibial nerves in workers occupationally exposed to low
concentrations of carbon disulphide. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, June 6-10, 1977, p. 18.
LUKAS, E., KOTAS, P, & OBRUSNIK, I. (1974) Copper and zinc levels in
peripheral nerve tissues of rats with experimental carbon
disulphide neuropathy. Br. J. ind. Med., 31: 288-291.
LUKAS, E., KUSALOVA, V., & KOCINOVA, F. (1975) Activity of
ceruloplasmin in the serum of rats with experimental carbon
disulphide neuropathy. In: McCallum, R. I., ed. Abstracts of the
XVIII International Congress on Occupational Health, Brighton,
England, 14-19 September, 1975, pp. 299-300.
LUKAS, E., TEISINGER, J, Jr, & BASS, A. (1977) Activity of muscle
enzymes in rats with experimentally developed carbon disulphide
polyneuropathy. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 19.
LYSINA, G. G. (1967) Some changes in the internal organs due to the
effects of carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 179-181.
MACK, T., FREUNDT, K. J., & HENSCHLER, D. (1974) Inhibition of
oxidative N-demethylation in man by low doses of inhaled carbon
disulphide. Biochem. Pharmacol., 23: 607-614.
MADLO, Z. & SOUCEK, B. (1953) [Absorption, metabolism, and action of
carbon disulfide in the organism. VII. Inhibition of serum
cholinesterase by carbon disulfide.] Prac. Lek., 6: 312-313 (in
Czech).
MADLO, Z., PEIZKER, & SOUCEK, B. (1953) [The absorption, metabolism
and action of carbon disulphide in the organism. V. The effect of
carbon disulfide on tissue respiration.] Prac. Lek., 5: 263-266
(in Czech).
MAGISTRETTI, M. & PEIRONE, E. (1961)The action of carbon disulphide on
cerebral monoamine oxidase. Med. Lavoro, 52: 1-10.
MAGOS, L. (1972) Relevancy of bivalent sulphur excretion to carbon
disulphide exposure in different metabolic conditions. Br. J.
ind. Med., 29: 90-94.
MAGOS, L. (1975) The clinical and experimental aspects of carbon
disulphide intoxication. Rev. environ. Health, 11: 65-80.
MAGOS, L. & BUTLER, H. (1972) Effect of phenobarbitone and starvation
on hepatotoxicity in rats exposed to carbon disulfide vapor.
Br. J. ind. Med., 29: 95-98.
MAGOS, L. & JARVIS, J. A. E. (1970a) The effects of carbon disulphide
exposure on brain catecholamines in rats. J. Pharm. Pharmacol.,
29: 936-938.
MAGOS, L. & JARVIS, J. A. E. (1970b) The effects of carbon disulfide
exposure on brain catecholamines in rats. Br. J. Pharmacol.,
39: 26-33.
MAGOS, J., GREEN, A., & JARVIS, J. A. E. (1974) Half life of CS2 in
rats in relation to its effect on brain catecholamines. Int.
Arch. Arbeitsmed., 32: 289-296.
MANU, P, LANCRANJAN, I., & ZAMFIRESCU, Z. (1970) Investigation of
serum LDH and its isoenzymatic model in carbon disulphide
polyneuritis. Int. Arch. Arbeitsmed., 26: 45-49.
MANU, P., LILIS, R., NESTORESCU, B., LANCRANJAN, I., ROSENTA, A.,
IONESCU, S., & KAUFMAN, G. (1971) Effects of carbon disulfide on
cholesterol and ß-lipoprotein levels in young men. In: Abstracts
of the II International Symposium on Toxicology of Carbon
Disulfide, Banja Koviljaca, Yugoslavia, 25-28 May, 1971, pp.
28-29.
MARTINO, P., COSCIA, G. C., & PERRELLI, G. (1963) [Lipedaemia in
carbon disulfide poisoning. Preliminary observations.[ Folia
Med., 46: 1110-1114 (in Italian).
MARTINO, P., PERRELLI, G., & COSCIA, G. C. (1964) [Lipedaemia in
carbon disulfide poisoning.[ Arch. Sci. Med., 117: 124-130 (in
Italian).
MARTYNOVA, A. P., ZELENKIN, A. N., GOLJAKOVA, L. P., MILKOV, L. E.,
GRODETSKAJA, N. S, & AGADZANOVA, A. A. (1976)[Clinical hygienic
and experimental investigations of the action on the body of
small concentrations of carbon disulfide.] J. Hyg. Sanit., 5:
25-28 (in Russian).
MASSOUD, A. E. E., SADEK, S., EL HAWARY, M. F., & HASSANEIN, M. (1971)
Changes in the electrocardiogram and lung function in workers
exposed to carbon disulfide. In: Abstracts of the II
International Symposium on Toxicology of Carbon Disulfide, Banja
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 6.
MASSOUD, A. A. E., ABDEL KARIM, A. H., & FARES, R. (in press) Serum
thyroxine, serum cholesterol and its fractions in workers exposed
to carbon disulphide. Egypt. J. occup. Med.
MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., VISCONTI, E., IOVICIC, M., &
CAVALLERI, A. (1966a) [Cerebrovascular damage in young workers
exposed to carbon disulfide.] Med. Lavoro, 57: 701-708 (in
Italian).
MAUGERI, U., IOVICIC, M., CAVALLERI, A., GRAOVAC-LEPOSAVIC, LJ.,
VISCONTI, E., & REZMAN, I. (1966b) [Peripheral vascular damage in
young workers exposed to carbon disulfide.] Med. Lavoro, 57:
709-714 (in Italian).
MAUGERI, U., VISCONTI, E., & CAVALLERI, A. (1966c)] Vascular
alterations in the retina of young workers exposed to carbon
disulfide.] Med. Lavoro, 57: 741-745 (in Italian).
MAUGERI, U., CAVALLERI, A., & VISCONTI, E. (1966d)
[Ophthalmodynamography in occupational carbon disulfide
poisoning.] Med. Lavoro, 57: 730-740 (in Italian).
MAUGERI, U., CAVELLERI, A., & VISCONTI, E. (1967)
Ophthalmodynamographic study in young workers exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Excerpta Medica Foundation, Amsterdam, pp.
150-152.
MAUGERI, CAVALLERI, A., MAUGERI, U., & TACCOLA, A. (1971) New aspects
in the pathogenetic mechanism of carbon disulphide toxic action.
Med. Lavoro, 62: 398-403.
MCCAMMON, C., Jr, QUINN, M. P., & KUEPEL, R. E. (1975) A charcoal
sampling method and a gas chromatographic analytical procedure
for carbon disulfide. Am. Ind. Hyg. Assoc. J., 36: 618-625.
MCKEE, R. W. (1941) Solubility of carbon disulphide vapor in body
fluids and tissues. J. ind. Hyg., 23: 484-489.
MCKENNA, M. J. & DI SRTEFANO, V. (1975) A proposed mechanism of the
action of carbon disulfide on dopamine-ß-hydroxylase. Toxicol.
appl. Pharmacol., 33: 137.
MELISSINOS, J.-C. & JACOBIDES, G. (1967) Sur quelques formes nerveuses
d'intoxication observées en Grece chez des sujets exposeés au
sulfure de carbone. Arch. Mal. Prof., 28: 605-611.
MERLEVEDE, E. (1951) [Toxicology of carbon disulfide.] Sinte
Catharina, Brugge, 180 pp. (in Dutch).
MIHAIL, G., MIHAILA, D., BRANISTEANU, D., BODNAR, J., & CIOATA, V.
(1968) Manifestations cliniques, fonctionnelles et biochimiques
des stades precoces du sulfocarbonisme. Arch. Mal. Prof., 29:
109-120.
MILIC, S. (1965) [Some observations concerning the determination of
CS2 in the urine of exposed workers.] In: Zbornik radova o
toksikologiji CS2 (Proceedings in Yugoslavian symposium on CS2
toxicology, Loznica, 1965, p. 41-42 (in Serbo-Croat).
MILKOV, L. E., MONAENKOVA, A.M., BYALKO, N. K., GLOTOVA, K. V.,
VERETINSKAJA, A. G., ALI-ZADE, K. A., MAKARENKO, Y. L.,
NIKOLSKAJA, O. I., & KURSAKOV, A. V. (1970) [On the initial forms
of chronic carbon disulfide poisoning.] Gig. Tr. Prof. Zabol.,
14: 28-32 (in Russian).
MINDEN, H., GOHLKE, R., & ROTHE, R. (1967) [Contribution to the
pathogenesis of chronic carbon disulfide poisoning.] Z. Ges.
Hyg. Grenz., 13: 815-822 (in German).
MISIAKIEWICZ, Z., SZULINSKA, O., & CHYBA, A. (1972) [Influence of a
carbon disulfide, hydrogen sulfide mixture in air on white rats
under the conditions of permanent exposure.] Rocz. Panstw. Zakl.
Hig., 23: 465-475 (in Polish).
MOREO, S. & CANDURA, F. (1960) [The behaviour of serum-clotting
factors in animals treated with sodium arsenite, carbon
disulfide, cadmium chloride and lead acetate.] Boll. Soc. Med.
Chir., (Pavia), 74: 447-455 (in Italian).
MOWÉ, G. (1971) Coronary heart disease and occupational exposure to
carbon disulfide. In: Abstracts of the III International
symposium on toxicology of carbon disulfide, Banja Koviljaca,
Yugoslavia, 25-28 May, 1971, p. 7.
MURASKO, V. A. (1975) [The morbidity of the digestive organs through
working in contact with carbon disulfide in the viscose
industry.] Vrach. Delo, No. 4, pp. 113-117 (in Russian).
NAKAMURA, K., HARADA, M., TATETSU, S., MIYAGAWA, T., TSUKAYAMA, T.,
KAWANO, K., KABASHIMA, K., HIRATA, M., & TOYA, G. (1974) A
clinical study on chronic carbon disulfide poisoning. Psychiatr.
Neuro. Jpn., 76: 243-273.
NEAL, R. A., KATAMAKI, T., HUNTER, A. L., & CATIGNANI, G. (1976)
Monooxygenase catalyzed activation of thiono-sulfur-containing
compounds to reactive intermediates. Hoppe-Seyler's Z. physiol.
Chem., 357: 1044.
NESSWETHA, L. & NESSWETHA, W. (1967) The clinical evaluation of
compensated carbon disulphide intoxications in the German Federal
Republic since 1948. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Excerpta Medica Foundation,
Amsterdam, pp. 214-218.
NIOSH (1977) Criteria for a recommended standard...occupational
exposure to carbon disulfide, Washington, DC, US Dept Health,
Education, and Welfare, National Institute for Occupational
Safety and Health, 186 pp.
NOFER, J. & WRONSKA-NOFER, T. (1966) Elimination des certains
metabolites de niacine par rapport au témps dexposition et la
close sulfure de carbone. In: Proceedings of the IX
International Congress on Occupational Health, Vienna, 1966,
pp. 355-358 (A III 53).
NOFER, J., CHOJNOWSKI, J., KAWECKA, M., KIEC, E., WRONSKA-SZPAKOWA,
T., & WYSZOMIRSKA, Z. (1961) [Effect of occupational hazard to
carbon disulphide on the pathogenesis of arteriosclerosis.]
Med. Pr. 12: 101-118 (in Polish).
NORO, L. (1944) [Hydrogen and carbon disulphide intoxications.] Nord.
Med., 24: 2015-2020 (in Swedish).
NOVAK, L., DJURIC, D., & FRIEDMAN, V. (1968) Specificity of the
iodine-azide test for carbon disulphide exposure. I. Urinary
excretion of sulfur compounds in persons treated with disulfiram.
Arch. environ. Health, 19: 473-477.
NUNZIATE, CESARO, A., SIPALA, E., & ZUNINI, E. (1952) [Carbon
disulfide poisoning.] Folia Med., 35: 661-716 (in Italian).
NURMINEN, M. (1976) Survival experience of a cohort of carbon
disulphide exposed workers from an eight-year prospective
follow-up period. Int. J. Epidemiol., 5: 179-185.
OLIENACZ, W., PLAMIENIAKA Z., SZYMANKA, J. (1964) [Excretion of
17-ketosteroids in chronic carbon disulphide poisoning.]
Med. Pt. 15: 155-157 (in Polish).
PATERNI, L., PUSIC, G., & TEODORI, S. (1958) [Chronic carbon disulfide
poisoning and arteriosclerosis from high-cholesterol diet in
rabbits.] Folia Med., 41: 705-722 (in Italian).
PAULUS, H. J., LIPPMAN, M., & COHEN, A. E. (1957) Evaluation of
potential health hazards in fumigation of shelled corn with a
mixture of carbon disulfide and carbon tetrachloride. Am. Ind.
Hyg. Assoc. J., 18: 345-350.
PERGAL, M., VUKOJEVIC, N., GIRIN-POPOV, N., DJURIC, D., & BOJOVIC, T.
(1972a) Carbon disulfide metabolites excreted in the urine of
exposed workers. I. Isolation and identification of
2-mercapto-2-thiazolinone-5. Arch. environ. Health, 25: 38-41.
PERGAL, M., VUKOJEVIC, N., & DJURIC, D. (1972b) II. Isolation and
identification of thiocarbamide. Arch. environ. Health, 25:
42-44.
PERGAL, M. N., KOBILAROV, J., RALETIC, A., POSTICS-GRUJIN, A., &
DJURIC, D. (1977a) Determination of small quantities of
thiocarbamate in urine. Egypt. J. Decup. Med., 5: 251-257.
PERGAL, M., RALETIC, J., & DURIC, D. (1977b) Identification of the
third metabolite, giving positive iodine-azide reaction in the
urine of workers exposed to CS2. A preliminary report. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski, Finland, 6-10 June, 1977, p. 29.
PETERSON, F. (1892) Three cases of acute mania from inhaling carbon
bisulphide. Boston Med. Surg., 127: 325-326.
PETROV, M. V. (1969)] Course and termination of pregnancy in women
working in viscose industry.] Pediatr. Akush. Ginekol., 3:
50-52 (in Russian).
PETROVIC, D. & DJURIC, D. (1965) [Carbon disulfide in exhaled air of
exposed workers.] In: Zbornik radova o toksikologiji ugljen
disulfida, Loznica, pp. 35-38. (in Serbo-Croat).
PETROVIC, D. & DJURIC, D. (1966) [Carbon disulfide in the air exhaled
by exposed workers.] Arh. Hig. Rada, 17: 159-163 (in
Serbo-Croat).
PETRUN, N.M. (1967) [The effect of carbon disulfide on certain
biochemical indices of the status of the organism in its action
via the skin.] Gig. Tr. Prof. Zabol., 11: 50-53 (in Russian).
PEYTON, T. O., STEEL, R. V., MABEY, W. R., CARLIN, A., & PINES, A.
(1976) Literature review and environmental assessment of carbon
disulfide and carbonyl sulfide. Menlo Park, CA Stanford Research
Institute, pp. 1-65. (Project no EGU 3570, US Environmental
Protection Agency, Publication No. 257-947).
PIROTSKAJA, E. N. (1972) [Changes in the protein-forming function of
the liver in workers exposed to carbon disulfide.] Vrach. Delo,
9: 132-134 (in Russian).
PREROVSKA, I. & ROTH, Z. (1968) [An observation of personnel in the
carbon disulphide hazard with special reference to the
development of arteriosclerosis.] Prac. Lek., 20: 6-10 (in
Czech).
PREROVSKA, I. & ZVOLSKY, P. (1973) [The health status in workers with
chronic carbon disulphide poisoning in five to nineteen years
after ascertaining the disease.] Prac. Lek., 25: 42-46 (in
Czech).
PREROVSKA, I., VANECEK, R., & KUBAT, K. (1961) Effect of carbon
disulfide on experimental arteriosclerosis in the rabbit. Acta
Univ. Carol., 14: 177-185.
QUARELLI, G. (1928)] Occupational CS2 poisoning.] In: Bericht. d. 5.
Internat. Kongr. f. Berufskrankh., Budapest, 1928 (in Italian).
QUARELLI, G. (1934)] Occupational carbon disulfide poisoning.] Russ.
Providenza Soc., 21: 10-73 (in Italian).
RANELLETTI, A. (1931)] Occupational carbon disulfide poisoning.]
Arch. Gewerbepath. Gewerbehyg., 2: 664-675 (in German).
RANELLETTI, A. (1933)] Occupational carbon disulfide poisoning.] In:
La Grafica Moderna, Ed. Torino (in Italian).
RAITTA, C. & TOLONEN, M. (1975) Ocular pulse wave in workers exposed
to carbon disulphide. Albrecht. Graefes Arch. Ophthal., 195:
149-154.
RAITTA, C., TOLONEN, M., & NURMINEN, M. (1974) Microcirculation of
ocular fundus in viscose rayon workers exposed to carbon
disulfide. Albrecht. Graefes Arch. Ophthal., 191: 151-164.
RECHENSERG, H. K. (1957)] Carbon disulfide poisoning and the
sulfocarbotoxic vascular Late-syndrome.] Arch. Gewerbepath.
Gewerbehyg., 15: 487-530 (in German).
RUIKKA, I. (1959) Postprandial serum turbidity in chronic carbon
disulfide poisoning a comparison of viscose rayon workers with
healthy and atherosclerotic men. Ann. Med. intern. Fenn., 48:
225-237.
SAITA, G. & GATTONI, L. (1957) [The blood coagulation process in
chronic carbon disulfide poisoning.] Med. Lavoro, 48: 229-237
(in Italian).
SAITA, G., SBERTOLI, C., & FARINA, G. F. (1964) Modifications in blood
coagulation and lipid metabolism produced by carbon disulfide.
Med. Lavoro, 55: 721-734.
SAKURAI, H. (1972) Hypertension induced by the occupational exposure
to carbon disulfide. In: Abstracts of the XVII International
congress on occupational health. Buenos Aires, 11-16 September,
1972, p. 182.
SAVIC, S. M. (1967) Influence of carbon disulfide on the eye. Arch.
environ. Health, 14: 325-326.
SAVIC, S. & IOVICIC, M. (1965) [Keratoconjunctivitis (causome) in
workers employed in the production of viscose fibres.] In:
Zbornik Radova o toksikologiji CS2, Loznica, Yugoslavia 1965,
pp. 115-121 (in Serbo-Croat).
SAVOLAINEN, H. & VAINIO, H. (1976) High binding of CS2 sulphur in
spinal cord axonal fraction. Acta Neuropathol. (Berlin), 36:
251-257.
SAVOLAINEN, H., JÄRVISALO, J., & VAINIO, H. (1977a) Specific binding
of CS2 metabolites to microsomal proteins in rat liver. Acta
Pharmacol. Toxicol., 41: 94-96.
SAVOLAINEN, H., JÄRVISALO, J., ELOVAARA,. E., & VAINIO, H. (1977b) The
binding of CS2 in central nervous system of control and
phenobarbitone-pretreated rats. Toxicology, 7: 207-214.
SAVOLAINEN, H., LEHTONEN, E., & VAINIO, H. (1977c) CS2 binding to rat
spinal neurofilaments. Acta Neuropathol. (Berl.), 37: 219-223.
SBERTOLI, C., FARINA, G., & SAITA, G. (1969) [Three cases of chronic
carbon disulfide poisoning with giomerulonephrotic syndrome.[
Med. Lavoro, 60: 28-37 (in Italian).
SCHEEL, L. D. (1965) Biological changes involving metal ion shifts.
Am. ind. Hyg. J., 26: 585-592.
SCHEEL, L. D. (1967) Experimental carbon Disulfide poisoning in
rabbits-Its mechanism and similarities with human case report.
In: Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 107-115.
SCHEEL, L. D., KEENAN, R. G., MOUNTAIN,, J. T., KopP, J., HOLTZ, J., &
KILLENS, R. (1960) The effect of dietary metal ions on carbon
disulfide toxicity. In: Proceedings of the 13th International
congress on occupational health, New York, 1960, pp. 783-784.
SCHILLING, R. S. F. (1970) Coronary heart disease in viscose rayon
workers. Am. Heart J., 80: 1-2.
SCHNEIDER, H. (1976) In: Adverse effects of environmental chemicals
and psychotropic drugs. Neurophysiological and behavioural
tests. Vol. 2, Amsterdam, 1976.
SCHÜTZ, H. (1970) Equipment and systems for ambient air monitoring,
Frankfurt/Maine, Hartmann & Braun, Mess und Regeltechnik.
SEPPÄLÄINEN, A.M. (1977) Neurophysiological findings in workers
exposed to carbon disulphide-A follow-up. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, 6-10 June, 1977, p. 17.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (1975) Electrophysiological studies
of rabbits in long-term exposure to carbon disulfide. Scand. J.
Work Environ. Health, 1: 178-183.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (19745) Electrophysiological findings
in rats with experimental carbon disulphide neuropathy.
Neuropath. appl. Neurobiol., 2: 209-216.
SEPPÄLÄINEN, A.M. & TOLONEN, M. (1974) Neurotoxicity of long-term
exposure to carbon disulfide in the viscose rayon industry. A
neurophysiological study. Work Environ. Health, 11: 145-153.
SEPPÄLÄINEN, A.M., TOLONEN, M., KARLI, P., HÄNNÄINEN, H., & HERNBERG,
S. (1972) Neurophysiological findings in chronic carbon disulfide
poisoning. A descriptive study. Work Environ. Health, 9: 71-75.
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49
(in Japanese).
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49.
(in Japanese).
SIHTOLA, H. (1976). [Stages in the development of the SINI-viscose
process.] Tutkimusja tekniikka, 7/8: 63-69 (in Finnish).
SIHTOLA, H. (1977) The SINI viscose process means less chemicals and
less pollution. In; Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 43.
SKALICKA, B. (1967) [Sulfur-containing compounds after administration
of tetraethylthiuramdisulfde.] Prac. Lek., 19: 408-412 (in
Czech).
SOKAL, J. A. (1973) Effect of chronic exposure to carbon disulphide
upon some components of the electron transport system in rat
liver microsome. Biochem. Pharmacol., 22: 129-132.
SOUCEK, B. (1957) [Transformation of carbon disulfide in the
organism.] J. Hyg. Epidemiol. (Praha), 1: 10-22 (in German).
SOUCEK, B. (1959) [Absorption and elimination of carbon disulfide.]
Prac. Lek., 11: 403-407 (in Czech).
SOUCEK, B. (1960a) [Pulmonary elimination of carbon disulfide after
intraperitoneal administration.] Farmakol. Toksikol., 23:(1):
74-84 (in Russian).
SOUCEK, B. (1960b) [The effect of carbon disulphide on the serum
cholesterol and cholesterol ester level.] Prac. Lek., 12:
243-248 (in Czech).
SOUCEK, B. & MADLO, Z. (1953) [Absorption, metabolism and action of
carbon disulphide in the organism. VI. Attempt at chromatographic
proof of the reaction of carbon disulphide with aminoacids in the
blood.] Prac. Lek., 5: 309-311 (in Czech).
SOUCEK, B. & MADLO, Z. (1954) [Absorption, metabolism and action of
carbon disulphide in the organism. VIII. Reaction of carbon
trisulphide with blood in vitro.] Prac. Lek., 6: 11-17 (in
Czech).
SOUCEK, B. & MADLO, Z. (1955) Determination of sulfhydryl groups in
guineapig serum after exposure to carbon disulfide. Bull. Soc.
Chim. Biol., (Paris), 37: 1373-1378.
SOUCEK, B. & MADLO, Z. (1956) [Dithiocarbamin carbon acids as
degradation products of carbon disulfide.] Arch. Gewerbepath.
Gewerbehyg., 14: 511-521 (in German).
SOUCEK, B. & PAVELKOVA, E. (1953) [Absorption, metabolism, and effects
of carbon disulfide in the organism. IV. Long-term experiments.]
Prac. Lek., 5: 189 (in Czech).
SOUCEK, B., MADLO, Z., & HOLEYSOVSKA, H. (1957) [The influence of
carbon disulfide on proteolytic enzymes.] Arch. Gewerbepath.
Gewerbyhyg., 15: 531-538 (in German).
STRITTMATTER, C. F., PETERS, T., & MCKEE, R. W. (1950) Metabolism of
labelled carbon disulphide in guineapigs and mice. Arch. ind.
Hyg., 1: 54-64.
STYBLOCA, V. (1977) [Electroencephalography in the diagnosis of early
brain damage by carbon disulfide.] Int. Arch. occup. environ.
Health. 38: 263-282 (in German).
SUGIMOTO, K., GOTO, S., & HOTTA, R. (1976) An epidemiological study on
retinopathy due to carbon disulfide. Int. Arch. occup. environ.
Health, 37: 1-8.
SUGIMOTO, K., GOTO, S., TANIGUCHI, H., BABA, T., RAITTA, C., TOLONEN,
M., & HERNBERG, S. (1977) Ocular fundus photography of workers
exposed to carbon disulfide-A comparative epidemiological study
between Japan and Finland. Int. Arch. occup. environ.
Health, 39: 97-101.
SULKOWSKI, W. & LATKOWSKI, B. (1969) [Audiometric and
electronystagmographic examinations of workers in plants
producing synthetic fibers, who are suffering from chronic carbon
disulfide poisoning.] Pol. hyg. Lek., 24: 1563-1565 (in
Polish).
SZENDZIKOWSKI, S., STETKIEWICZ, J., WRONSKA-NOFER, T., & ZDRAJKOWSKA,
I. (1973) Structural aspects of experimental carbon disulfide
neuropathy. I. Development of neurohistological changes in
chronically intoxicated rats. Int. Arch. Arbeitsmed., 31:
135-149.
TABACOVA, S., HINKOVA, L., BALABAEVA, L., & KURCHATOVA, G. (1977).
Effects of carbon disulphide on the pre- and postnatal
development of rats. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 26.
TACCOLA, A., MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., CAVALLERI, A., &
DJURIC, D. (1971) [Vascular changes in human subjects exposed to
carbon disulfide.] Med. Lavoro, 62: 404-411 (in Italian).
TACCOLA, A., CAVALLERI, A., FRANCO, G., & MALAMANI, T. (1977) An
evaluation of the ratio of P-wave duration to P-R segment in ECG
of CS2 exposed workers. Egypt. J. occup. Med., 5: 309-312.
TAHARA, Y. (1961) [Hygienic evaluation of work environment of rayon
factories by carbon disulfide analysis of the expired air of
workers.] J. Sci. Labour (Tokyo), 37: 583-597 (in Japanese).
TARKOWSKI, S. (1973) [Glucose and free amino acid metabolism in the
brain of rats chronically poisoned with carbon disulfide.] Med.
Pr., 24: 571-575 (in Polish).
TARKOWSKI, S. (1974) Activity of glutamate decarboxylase in the brain
of rats exposed to carbon disulfide. Int. Arch. Arbeitsmed.,
33: 79-82.
TARKOWSKI, S. & CREMER, J. E. (1972) Metabolism of glucose and free
amino acids in brain, studied with 14C labelled glucose and
butyrate in rats intoxicated with carbon disulphide.
J. Neurochem., 19: 2631-2640.
TARKOWSKI, S. & SOBCZAK, H. (1971) Oxidation and phosphorylation
processes in brain mitochondria of rats exposed to carbon
disulphide. J. Neurochem., 18: 177-182.
TARKOWSKI, S. & WRONSKA-NOFER, T. (1966) [Effect of carbon disulphide
on the cytochrome oxidase in rats.] Med. Pr., 17: 375-379 (in
Polish).
TAZUKA, H. (1955) [A study on CS2 poisoning. Report II: On the
absorption of CS2.] J. Sci. Labour (Jpn), 31: 208-209 (in
Japanese).
TEISINGER, J. (1934) Mils central hemiparesis with extrapyramidal
signs in chronic poisoning. Cas. Lek. Ces., 73: 188-189.
TEISINGER, J. (1954) [Metabolism and detoxication of some industrial
poisons.] Praha, S.Z.N., p. 106 (in Czech).
TEISINGER, J. (1971) [Chronic carbon disulphide poisoning mechanism.]
Pracov. Ldk., 23: 306-308 (in Czech).
TEISINGER, J. & SOUCEK, B. (1949) Absorption and elimination of carbon
disulfide in man. J. ind. Hyg., 31: 67-73.
TEISINGER, J. & SOUCEK, B. (1952) [The importance of metabolism of
some toxic vapours considering their transmission and excretion
in man.] Cas. Ldk. Ces., 91: 1372-1375 (in Czech).
THIELE, H. & WOLF, I. (1976) [Results of complex clinical serial tests
on CS2-exposed workers.[ Z. ges. Hyg. DDR, 22: 603-608 (in
German).
TILLER, J. R., SCHILLING, R. S. F., & MORRIS, J. N. (1968)
Occupational toxic factor in mortality from coronary hear
disease. Br. med. J., 4: 407-411.
TINTERA, J., GRAOVAC-LEPOSAVIC, LJ., & MILIC, S. (1972) Excretion of
some tryptophan metabolites in man exposed to carbon disulphide.
med. Lavoro, 63: 126-133.
TOLONEN, M. (1974) Chronic subclinical carbon disulphide poisoning.
Work Environ. Health, 11: 154-161.
TOLONEN, M., HERNBERG, S., NURMINEN, M., & TIITOLA, K. (1975) A
follow-up study of coronary heart disease in viscose rayon
workers exposed to carbon disulphide. Br. J. ind. Med., 32:
1-10.
TOLONEN, M., HERNBERG, S., NORDMAN, C-H., GOTO, S., SUGIMOTO, K., &
BABA, T. (1976) Angina pectoris, electrocardiographic findings
and blood pressure in Finnish and Japanese workers exposed to
carbon disulfide. Int. Arch. occup. environ. Health, 37:
249-264.
TOLONEN, M., HANNINEN, H., & NURMINEN, M. (1978) Phychological tests
specific to individual carbon disulfide exposure. Scand. J.
Psychol., 19(3): 241-245.
TOYAMA, T. & HARASHIMA, S. (1962) {Industrial hygienic evaluation of
exposure to CS2 through analysis of biological materials.] Jpn.
J. ind. Health., 4: 81-85 (in Japanese).
TOYAMA, T. & KUSANO, H. (1953) [An experimental study on absorption
and excretion of CS2.] Jpn. J. Hyg., 8: 70 (in Japanese).
TOYAMA, T. & SAKURAI, H. (1967) Ten-year changes in exposure level and
toxicological manifestations in carbon disulphide workers. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 197-204.
TRUHAUT, R. R., BOUDENE, C., PHU-LICH, N., & BAQUET, A. (1972)
Memoires nouvelles recherches sur la mesure en continu de
l'exposition individuelle au sulfure de carbone dans l'industrie
a l'aide d'un appareil portatif. Arch. Mal. prof., 33: 341-346.
TURCEK, M. & MOLCAN, J. (1964) [Carbon disulphide psychoses.] Prac.
Lek., 16: 257-261 (in Czech).
UEHLINGER, E. (1952) [On carbon disulfide glomerulosclerosis.]
Schweiz. Z. Allg. Pathol., 15: 217-224 (in German).
VASAK, V. (1963) [Assessment of exposure of workers to carbon
disulphide vapours.] Prac. Lek., 15: 143-145 (in Czech).
VASAK, V. & KOPECKY, J. (1967) On the role of pyridoxamine in the
mechanism of the toxic action of carbon disulfide. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 35-41.
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1963) Assessment of exposure
of workers to carbon disulphide vapours. II. Application of the
iodine-azide reaction for the detection and estimation of carbon
disulphide metabolites in urine. Prac. Lék., 15: 145-149 (in
Czech).
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1969) The assessment of the
exposure of workers to carbon disulphide by the iodine-azide test
of Vasak, Vanecek and Kimmelova. (1963). In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 56-59.
VASILJEVA, I. A. (1973) [Effect of low concentrations of carbon
disulfide and hydrogen sulfide on the menstrual function in women
and on the estrous cycle in an experiment.] Gigi. Sanit., 38:
24-27 (in Russian).
VERTIN, A. (1973) In: Gas analysis instrumentation, London,
Macmillan Press Ltd.
VERTIN, P. G. (1966) [Dynamics of the elastase and the cholesterol and
phospholipid ratio with exposure to CS2.] Arch. Gewerbepath.
Gewerbyhyg., 22: 167-178 (in German).
VERTIN, P. G. (1977) An examination for cardiac/vascular diseases in
the Dutch viscose rayon industry. In: Tolonen, M., ed. Abstracts
of the IV International Symposium on Occupational Health in the
Production of Artificial Fibres, Helsinki and Valkeakoski,
Finland, 6-10 June, 1977, p. 7.
VESCE, C. A., COPPA, S., & FIMIANI, R. (1953) [Variations in urinary
17-ketosteroids in chronic poisoning from solvents.] Folia Med.
(Napoli), 36: 444-460 (in Italian).
VIDAAKOVIC, A. & ANDJELKOVSKI, A. (1965) [Haematological changes in
workers exposed to carbon disulfide.] In: Zbornik radova o
toksikologiji CS2, Loznica, 1965, p. 77-81 (in Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & ANDJELKOVSKI, A. (1965a) [A study of the
effects of carbon disulfide in the liver.] In: Zbornik radova o
toksikologiji ugljen disulfida, Loznica, 1965, pp. 71-75 (in
Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & DJORDJEVIC, V. (1956b) [Some changes in
the blood of rats in sub-acute carbon disulfide-poisoning.] In:
Zbornik radova o toksikologiji CS2 Loznica, 1965, pp. 83-87
(in Serbo-Croat).
VIGLIANI, E. C. (1946) [Chronic carbon disulfide poisoning. A report
on 100 cases.] Med. Lavoro, 37: 165-193 (in Italian).
VIGLIANI, E. C. (1954) Carbon disulphide poisoning in viscose rayon
factories. Br. J. ind. Med., 11: 235-244.
VIGLIANI, E. C. (1961) [Intoxication with carbon disulfide.] In:
Baader, E. W., ed. Handbuch der Gesamten Arbeitsmedizin, Urban
& Schwarzenberg, pp. 313-325 (in German).
VIGLIANI, E. C. & PERNIS, B. (1955) [Clinical and experimental
investigations on atherosclerosis caused by carbon disulfide.]
Arch. Gewerbepath. Gewerbehyg., 14: 190-202 (in German).
VIGLIANI, E., VISTINI, F., & MASPES, P. E. (1944) First observations
on myopathy from carbon disulfide. Med. Contemp., 10: 85-96.
VINOGRADOV, P. B. (1966) [New experimental findings for substantiating
the maximum permissible concentrations of carbon disulfide in
reservoir water.] Gig. i Sanit., 31: 13-17 (in Russian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., VISNJIC, V., MAUGERI, U.,
& REZMAN, I. (1966a) [Hematological findings in young workers
exposed to CS2. I. Fibrinolysis.] Med. Lavoro, 57: 667-676 (in
Italian).
VISCONTI, E., REZMAN, I., MAUGERI, U., VIDAKOVIC, A., & CAVALLERI, A.
(1966b) Hematochemical findings in young workers exposed to
carbon disulfide. II. The behavior of lipoproteins and the
"clearing factor". Med. Lavoro, 57: 677-683.
VISCONTI, E., VISNJIC, V., CAVALLERI, A., VIDAKOVIC, A., MAUGERI, U.,
& REZMAN, I. (1966c) [Haematological findings in young workers
exposed to carbon disulfide. III. Antiheparinic activity.] Med.
Lavoro, 57: 745-750 (in Italian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., REZMAN, I., MAUGERI, U., &
VISNJIC, V. (1967). Fibrinolytic activity in young workers
exposed to carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 128-132.
WAKATSUKI, T. & HIGASHIKAWA, n. (1959) Experimental studies on carbon
disulfide and hydrogen sulfide poisoning, histological changes in
hemopoietic organs and other internal organs. Shikoku Igaku
Zasshi, 14: 549-554.
WEAST, R. C. (1974) Handbook of Chemistry and Physics, 55th ed.,
Cleveland, CRC Press, pp. C-234-235; D-85; D-163.
WEIGMAN, H. J. (1973). [Measuring devices for automatic surveillance
of air pollution in work places.] Z. ges. Hyg., 19: 1-9 (in
German).
WILEY, F. H., HUEPER, W. C., & VON OETTINGEN, W. F. (1936) On the
toxic effects of low concentrations of carbon disulfide. J. ind.
Hyg. Toxicol., 18: 733-740.
WRONSKA-NOFER, T. (1969) The incorporation of 14C-acetate into
cholesterol in rats exposed to carbon disulphide. Biochem.
Pharmacol, 18: 925-926.
WRONSKA-NOFER, T. (1970) The influence of nicotinic acid upon the
disturbances in lipid metabolism caused by carbon disulphide in
rats. Int. Arch. Arbeitsmed., 27: 221-227.
WRONSKA-NOFER, T. (1976) Aortic phospholipids synthesis in
experimental carbon disulphide intoxication in rats. Int. Arch.
occup. environ. Health, 36: 229-234.
WRONSKA-NOFER, W., NOFER, J., & TARKOWSKI, S. (1965) [Impaired niacin
metabolite excretion in animals poisoned with carbon disulfide.]
Med. Pt., 16: 77-81 (in Polish).
WRONSKA-NOFER, T., TARKOWSKI, S., GORNY, R., SOKAL, J. A., & SZYC, M.
(1970) Accelerated turnover rate of nicotinamide adenine
dinucleotides in the liver of rats intoxicated with carbon
disulfide. Biochem. Pharmacol, 21; 2945-2950.
WRONSKA-NOFER, T., STETKIEWICZ, J., & SZENDZIKOWSKI, S. (1973)
Structural alterations and content of nicotinamine-adenine
nucleotides in skeletal muscle of rat in chronic experimental
carbon disulfi de intoxication. Int. Arch. occup. Health., 31:
125-134.
YAMAGATA, Y., YUDA, A., SUZUKI, K., NEMOTO, T., TAKAHASHI, M.,
TUCHIDA, H., SAITO, K., & KUSUNOKI, N. (1966) Carbon disulfide
nephroselerosis with special reference to the similarity to
diabetic glomerulosclerosis. J. Jpn. Diabetic. Soc., 9:
208-217.
YAROSLAVSKY, B. K. (1969) Toxic action of carbon disulfide on
reproduction and potentiation of this effect by tryptophan.
Bull. exp. Biol. Med., 68: 88-91.
YOSHIDA, K. (1955) [Fate of CS2 in the body-formation of thioketone
bonds.] J. Sci. Labour (Tokyo), 31: 209 (in Japanese).
ZEGLIO, P. (1942) Changes in gastric function due to chronic exposure
of workers in rayon industry. Med. Lavoro., 33: 217-228.
ZENK, H. (1967) The rhino-oto-vestibular symptomatoiogy of carbon
disulphide intoxication. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 190-191.
ZENK, H. (1970) [Carbon disulfide influences in the rhino-otological
functions of workers in the plastic fibers industry.] Int. Arch.
Arbeitsmed., 27: 210-220 (in German).
Annex I
PRODUCTION OF VISCOSE AND ITS END-PRODUCTS
The technological process of viscose rayon production, which is
schematically presented in Annex I Fig. 1, can briefly be described in
the following way:
(a) Cellulose pulp arrives at the factory in the form of sheets
which are placed in a hydraulic press or pulverized and steeped in a
solution of caustic soda to form alkali cellulose;
(b) the alkali cellulose is shredded to form crumbs to
facilitate aging and xanthation;
(c) after storage in aging tanks until the right level of
polymerization has been reached, the crumbs are placed in mechanical
churns;
(d) in a separate operation, sulfur is melted and poured onto
glowing charcoal where it reacts to give carbon disulfide. The liquid
carbon disulfide is stored in special tanks that are protected against
flames, static electricity, and heat;
(e) the liquid carbon disulfide, which is gradually metered
into the mechanical churns, reacts with the alkali cellulose to form
orange crumbs consisting of colourless cellulose xanthate and orange
sodium trithiocarbonate (Na2CS3);
(f) these crumbs are dissolved in caustic soda solution to give
a viscose solution;
(g) the viscose is stored in aging tanks, filtered, de-aerated,
and then pumped into the spinning tanks where it is forced through
spinnerets that are submerged in a solution of sulfuric acid (H2SO4)
and sodium and zinc sulfates (Na2SO4-ZnSO4). Filaments are formed
by coagulation of the viscose when partial decomposition of the
cellulose xanthate and the trithiocarbonate takes place, according to
the reactions:
(1) ROCS + H2SO4-ROH + NaHSO4 + CS2
SNa
(2) Na2CS3 + H2SO4 - H2S + CS2 + Na2SO4
During this process, considerable quantities of carbon disulfide
and hydrogen sulfide vapours are liberated representing a danger to
the exposed workers. It is important that reaction (2) should proceed
much more quickly than reaction (1) during spinning, so that
practically all the hydrogen sulfide is already formed in the spinning
bath or immediately over it, and the part of the carbon disulfide that
is supplied by the xanthate is mainly freed outside the spinning bath
in the acid yarn and/or in further treatments. The quantities of
carbon disulfide and hydrogen sulfide vapours that are liberated
depend on the process and the end-use of the rayon product.
The following products may be produced from viscose: rayon
filament textile yarn used in the textile industry in linings and
furniture fabrics; rayon filament tire yarn used as reinforcing
material in the carcass and belt of tires; rayon staple fibre
combined in textile fabrics with natural or synthetic fibres, such as
cotton or polyester; and Cellophane film, used for packaging.
On leaving the spinning bath, textile rayon yarn may be:
(a) wound up on acid spools on the spinning machine above the
spinning bath;
(b) formed into a cylindrical acid cake in a centrifugal pot
also incorporated in the spinning machine; or
(c) continuously transported through the regeneration, washing,
and drying treatments and then wound up as the finished product.
During spinning, the spinning machines are more or less
hermetically sealed. However, during doffing or when filament breaks
occur, the worker must open the "windows" or covers. At such times,
hydrogen sulfide and carbon disulfide vapours escape into the air. All
hydrogen sulfide is formed in the spinning bath or immediately over
it. Most of the carbon disulfide will be absorbed in the acid spool or
cake: and either be retained for a long time or be immediately freed
in a hot acid bath. Consequently, hydrogen sulfide will mainly be
found in the atmosphere of the machine and the acid baths, whereas
carbon disulfide also occurs in the departments where the acid spools
and cakes undergo further treatment (storage room, washing and drying
departments). In spool spinning, the worker has to lean over the
spinning bath to perform the doting and spinning-in operations. In
cake spinning, doffing takes place either at the front of the machine,
under conditions that are roughly the same as for spool spinning, or
at the rear of the machine, where the worker is not forced to breathe
in the machine atmosphere.
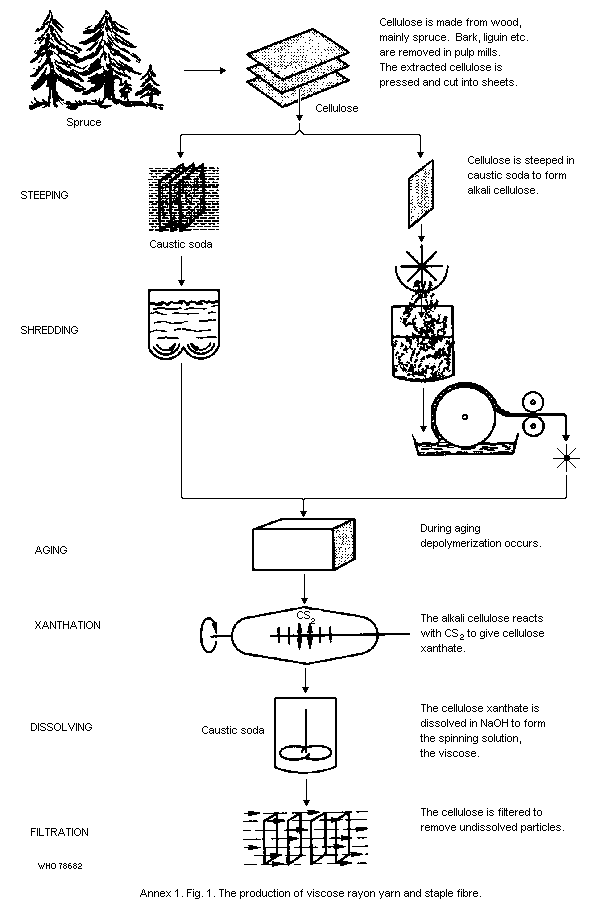 Approximately 50% of the quantity of carbon disulfide that was
originally added is present in the spool and is recovered by steaming.
The cake contains less carbon disulfide because the centrifugal,
motion of the pot removes a certain amount.
Tire yarn is mostly spun in glass tubes, where the viscose is
in direct contact with the spinning bath. The bath acid drains to the
sealed spinning tanks via return pipes and the yarn is run through a
hot acid bath, where the xanthate is largely decomposed. This second
hot acid bath may either be incorporated in the spinning machine or
installed outside it. In both cases the concentration of carbon
disulfide over this bath is high and, when a yarn breaks, the worker
may be exposed to the vapours.
After leaving the hot acid bath, the yarn is either (a) wound
on large acid spools; (b) made into cakes in centrifugal pots or
(c) run through washing and drying sections.
Staple fibre, like textile filament yarn, is mostly spun in an
open bath. However, hydrogen sulfide and carbon disulfide
concentrations are much higher, because of the large amounts of
viscose used per unit time and per spinning position. The thick tow is
treated in a separate sealed hot-acid bath (as in tire yarn spinning)
for further regeneration and large amounts of carbon disulfide are
freed in the process. After this stage, the cable is cut and more
carbon disulfide is emitted. The staple fibre is then washed and dried
continuously and carbon disulfide emissions are much lower.
Cellophane casting takes place on a continuous machine. The
sheet is cast in an open casting bath, like the spinning bath in
textile filament spinning. The Cellophane sheet then passes through a
number of baths in which decomposition, washing, bleaching, and drying
take place. In Cellophane casting, large quantities of viscose are
pressed into relatively small acid and second baths. As a result,
hydrogen sulfide and carbon disulfide emissions are very high over a
relatively small surface area.
The following data and Annex 1 Table 1 give some insight into the
emissions of hydrogen sulfide and carbon disulfide in the different
processes:
For every kg of yarn, staple fibre, or Cellophane, some 10-12 kg
of viscose is fed into the spinning or casting bath.
1 kg of viscose will give approximately: 20-30 g CS2
4-6 g H2S
(i.e., about 5 times more CS2 than H2S)
Viscose inputs in the spinning or casting machine per hour per
position are:
textile yarn 0.6-1.0 kg
tire yarn 10-12 kg
staple fibre 70/100 kg
Cellophane 1800-2000 kg
Total amounts of vapours and gases (in grams) that can be liberated
per hour per position are:
CS2 H2S
textile yarn 20-25 3-5
tire yarn 225-250 40-50
staple fibre 1000-1500 200-300
Cellophane 38 000-42 000 9000-10 000
Annex 1. Table 1. Percentage emissions of carbon disulfide and hydrogen
sulfide during the formation of viscose products.
Acid bath Spinning machine In the spool, cake,
tanks over first and tow, or Cellophane
second baths after the baths
textile yarn 35 55 10 H2S
25 65 CS2
tire yarn 15 5-55 15 H2S
25 CS2
staple fibre 45 40 15 H2S
8 12-45 35 CS2
Cellophane 15 70 15 H2S
25 55 20 CS2
Annex II
MAXIMUM PERMISSIBLE CONCENTRATIONS FOR CARBON DISULFIDE IN
DIFFERENT COUNTRIES
Chronic manifest poisoning can be prevented in the viscose rayon
industry by maintaining the carbon disulfide concentrations below
60 mg/m3 (20 ppm). However, recent studies have shown that
gynaecological cardiovascular, neurological, and neurophysiological
effects can still be detected under such conditions (sections 6.3.9,
6.3.10, 6.3.11, 7.3). It appears that a time-weighted average (TWA) of
30 mg/m3 (10 ppm) is the lowest level at which most of these effects
can be found. However, it has been reported in some studies from the
USSR that gynaecological, cardiovascular, and neurological disorders
may occur at even lower levels. It seems possible that most toxic
effects could be prevented by keeping carbon disulfide levels,
expressed as background TWAs, below 30 mg/m3 (10 ppm). The levels for
personal exposure may prove to be different but too few data are
available, at present, to make any definite statements.
If the effect is severe, as in the case of coronary deaths, it is
prudent to apply a safety margin for that effect. The need for such a
safety margin must be considered separately in each, case. Both, the
new NIOSH recommendation (NIOSH, 1977) and the USSR maximum allowable
concentration (MAC) value include safety margins. Most other countries
have not yet considered it necessary or practically feasible to adapt
safety margins. Table 1 in this annex presents permissible exposure
levels for a number of countries.
Based on results of studies on man and animals, the USSR has
adopted an MAC value of 1 mg/m3. Measurements of carbon disulfide
concentrations were made during the last 5 years of exposure on
workers exposed to 10 mg/m3 for 10-15 years. No significant changes
in the work environment had reportedly occurred during the entire
exposure period. The workers showed neurological, cardiovascular and
gynaecological disorders. (Petrov, 1969; Vasiljeva, 1973; Martynova et
al., 1976). Animals exposed to 100 and 10 mg/m3, respectively, showed
several adverse effects while only those exposed to 1 mg/m3 were free
from symptoms.
The Federal Republic of Germany has adopted a level of 30 mg/m3
"because at 60 mg/m3 level the liver oxygenesis was inhibited and
between 30-60 mg/m3 of longstanding exposure, coronary heart
diseases, hypertension, psychological and neurophysiological
disturbances can occur" (Freundt, 1975).
In the USA, NIOSH (1977) has recommended a standard of 3 mg/m3
(1 ppm) as a 10-h TWA concentration during a 40-h working week. A
level of 30 mg/m3 (10 ppm) for a 15-minute sampling period is
recommended to avoid acute toxicity. NIOSH based its choice of levels
on the probability of excess coronary deaths, and applied a safety
factor of 10 to the lowest concentration thought to be associated with
such cardiovascular disorders.
It should be stressed that the background concentration does not
reflect personal exposure, which may be considerably higher.
Therefore, the use of a personal sampler is recommended for the
assessment of actual uptake of carbon disulfide by individual workers,
accompanied by the use of the iodine-azide test. As already suggested,
it may well be that such experience will create a need for expressing
maximum permissible concentrations in terms of personal exposure
instead of background exposure.
Annex II. Table 1 Maximum permissible concentrations in
various countries. If not otherwise stated,
the figures represent TWAs.
Country ppm mg/3
Czechoslovakia (1963) 30
Egypt (1974) 30
Finland (1972) 30
German Democratic Republic (1971) 50
Germany, Federal Republic of (1975) 10 30
Hungary 20
Japan (1974) 10 30
Poland (1976) 25
Sweden (1975) 10 30
Switzerland (1971) 10 30
United Kingdom 10 30
USA (1973) 20 60
100 (MAC) 300 (MAC)
USSR (1976) 1 (MAC)
Yugoslavia (1971) 15 50 (MAC)
Approximately 50% of the quantity of carbon disulfide that was
originally added is present in the spool and is recovered by steaming.
The cake contains less carbon disulfide because the centrifugal,
motion of the pot removes a certain amount.
Tire yarn is mostly spun in glass tubes, where the viscose is
in direct contact with the spinning bath. The bath acid drains to the
sealed spinning tanks via return pipes and the yarn is run through a
hot acid bath, where the xanthate is largely decomposed. This second
hot acid bath may either be incorporated in the spinning machine or
installed outside it. In both cases the concentration of carbon
disulfide over this bath is high and, when a yarn breaks, the worker
may be exposed to the vapours.
After leaving the hot acid bath, the yarn is either (a) wound
on large acid spools; (b) made into cakes in centrifugal pots or
(c) run through washing and drying sections.
Staple fibre, like textile filament yarn, is mostly spun in an
open bath. However, hydrogen sulfide and carbon disulfide
concentrations are much higher, because of the large amounts of
viscose used per unit time and per spinning position. The thick tow is
treated in a separate sealed hot-acid bath (as in tire yarn spinning)
for further regeneration and large amounts of carbon disulfide are
freed in the process. After this stage, the cable is cut and more
carbon disulfide is emitted. The staple fibre is then washed and dried
continuously and carbon disulfide emissions are much lower.
Cellophane casting takes place on a continuous machine. The
sheet is cast in an open casting bath, like the spinning bath in
textile filament spinning. The Cellophane sheet then passes through a
number of baths in which decomposition, washing, bleaching, and drying
take place. In Cellophane casting, large quantities of viscose are
pressed into relatively small acid and second baths. As a result,
hydrogen sulfide and carbon disulfide emissions are very high over a
relatively small surface area.
The following data and Annex 1 Table 1 give some insight into the
emissions of hydrogen sulfide and carbon disulfide in the different
processes:
For every kg of yarn, staple fibre, or Cellophane, some 10-12 kg
of viscose is fed into the spinning or casting bath.
1 kg of viscose will give approximately: 20-30 g CS2
4-6 g H2S
(i.e., about 5 times more CS2 than H2S)
Viscose inputs in the spinning or casting machine per hour per
position are:
textile yarn 0.6-1.0 kg
tire yarn 10-12 kg
staple fibre 70/100 kg
Cellophane 1800-2000 kg
Total amounts of vapours and gases (in grams) that can be liberated
per hour per position are:
CS2 H2S
textile yarn 20-25 3-5
tire yarn 225-250 40-50
staple fibre 1000-1500 200-300
Cellophane 38 000-42 000 9000-10 000
Annex 1. Table 1. Percentage emissions of carbon disulfide and hydrogen
sulfide during the formation of viscose products.
Acid bath Spinning machine In the spool, cake,
tanks over first and tow, or Cellophane
second baths after the baths
textile yarn 35 55 10 H2S
25 65 CS2
tire yarn 15 5-55 15 H2S
25 CS2
staple fibre 45 40 15 H2S
8 12-45 35 CS2
Cellophane 15 70 15 H2S
25 55 20 CS2
Annex II
MAXIMUM PERMISSIBLE CONCENTRATIONS FOR CARBON DISULFIDE IN
DIFFERENT COUNTRIES
Chronic manifest poisoning can be prevented in the viscose rayon
industry by maintaining the carbon disulfide concentrations below
60 mg/m3 (20 ppm). However, recent studies have shown that
gynaecological cardiovascular, neurological, and neurophysiological
effects can still be detected under such conditions (sections 6.3.9,
6.3.10, 6.3.11, 7.3). It appears that a time-weighted average (TWA) of
30 mg/m3 (10 ppm) is the lowest level at which most of these effects
can be found. However, it has been reported in some studies from the
USSR that gynaecological, cardiovascular, and neurological disorders
may occur at even lower levels. It seems possible that most toxic
effects could be prevented by keeping carbon disulfide levels,
expressed as background TWAs, below 30 mg/m3 (10 ppm). The levels for
personal exposure may prove to be different but too few data are
available, at present, to make any definite statements.
If the effect is severe, as in the case of coronary deaths, it is
prudent to apply a safety margin for that effect. The need for such a
safety margin must be considered separately in each, case. Both, the
new NIOSH recommendation (NIOSH, 1977) and the USSR maximum allowable
concentration (MAC) value include safety margins. Most other countries
have not yet considered it necessary or practically feasible to adapt
safety margins. Table 1 in this annex presents permissible exposure
levels for a number of countries.
Based on results of studies on man and animals, the USSR has
adopted an MAC value of 1 mg/m3. Measurements of carbon disulfide
concentrations were made during the last 5 years of exposure on
workers exposed to 10 mg/m3 for 10-15 years. No significant changes
in the work environment had reportedly occurred during the entire
exposure period. The workers showed neurological, cardiovascular and
gynaecological disorders. (Petrov, 1969; Vasiljeva, 1973; Martynova et
al., 1976). Animals exposed to 100 and 10 mg/m3, respectively, showed
several adverse effects while only those exposed to 1 mg/m3 were free
from symptoms.
The Federal Republic of Germany has adopted a level of 30 mg/m3
"because at 60 mg/m3 level the liver oxygenesis was inhibited and
between 30-60 mg/m3 of longstanding exposure, coronary heart
diseases, hypertension, psychological and neurophysiological
disturbances can occur" (Freundt, 1975).
In the USA, NIOSH (1977) has recommended a standard of 3 mg/m3
(1 ppm) as a 10-h TWA concentration during a 40-h working week. A
level of 30 mg/m3 (10 ppm) for a 15-minute sampling period is
recommended to avoid acute toxicity. NIOSH based its choice of levels
on the probability of excess coronary deaths, and applied a safety
factor of 10 to the lowest concentration thought to be associated with
such cardiovascular disorders.
It should be stressed that the background concentration does not
reflect personal exposure, which may be considerably higher.
Therefore, the use of a personal sampler is recommended for the
assessment of actual uptake of carbon disulfide by individual workers,
accompanied by the use of the iodine-azide test. As already suggested,
it may well be that such experience will create a need for expressing
maximum permissible concentrations in terms of personal exposure
instead of background exposure.
Annex II. Table 1 Maximum permissible concentrations in
various countries. If not otherwise stated,
the figures represent TWAs.
Country ppm mg/3
Czechoslovakia (1963) 30
Egypt (1974) 30
Finland (1972) 30
German Democratic Republic (1971) 50
Germany, Federal Republic of (1975) 10 30
Hungary 20
Japan (1974) 10 30
Poland (1976) 25
Sweden (1975) 10 30
Switzerland (1971) 10 30
United Kingdom 10 30
USA (1973) 20 60
100 (MAC) 300 (MAC)
USSR (1976) 1 (MAC)
Yugoslavia (1971) 15 50 (MAC)
 The responsibility of metabolites for the discoloration of iodine
azide remained hypothetical until Pergal et al. (1972a,b) isolated 3
metabolites from human urine and identified 2 of them as thiourea and
mercaptothiazolinone; thiourea is by far the most important of these
metabolites. Later, Pergal et al. (1977a) developed a quantitative
method for the micro-determination of thiourea in the urine of exposed
workers or of alcoholics treated with tetraethylthiuramdisulfide
(TETD, Disulfiram, Antabuse). The authors suggested that the third
metabolite was 2-mercapto-thiazoline-4-carbamic acid (Pergal et al.,
1977b). Tetraethylthiuramdisulfide is metabolized in a way that
liberates carbon disulfide (Fig. 1). Consequently, alcoholics treated
with this agent are exposed to carbon disulfide and its metabolites.
Skalicka (1967) and Novak et al. (1968) measured the iodine-azide
reaction and determined diethyldithiocarbamates (DDC) in the urine of
alcoholics treated with TETD. These results led Djuric et al. (1973)
to use TETD as a test for the evaluation of the metabolic rate of
sulfur compounds in the organism of workers, the so-called "antabuse
test".
Studies on the microsomal metabolism of carbon disulfide in the
liver of rats revealed that it was desulfurated to form
carbonylsulfide and that this was further oxidized, yielding carbon
dioxide which was exhaled (De Matteis & Seawright, 1973; De Matteis,
1974; Dalvi et al., 1974).
Data from human and animal studies on ethereal sulfate excretion
(Magos, 1973) have shown that bivalent sulfur represents a small part
of retained carbon disulfide, probably less than 5%. The major pathway
leads to the formation of sulfates that are excreted in urine.
5. BIOCHEMICAL EFFECTS OF CARBON DISULFIDE
From the chemical point of view, carbon disulfide is highly
reactive with nucleophilic reagents characterized by the presence of a
group with a free pair of electrons in the molecule. The most
important nucleophilic groups are mercapto (-SH), amino (-NH2) and
hydroxy (-OH) groups (Vasak & Kopecky 1967). However, physiological pH
values do not favour these reactions (Kopecky, 1977, private
communication).
According to the chemical structure of compounds participating in
the reactions, carbon disulfide will produce dithiocarbamic,
trithiocarbonic, or xanthogenic acid. If carbon disulfide reacts with
an organic compound with 2 nucleophilic groups, a cyclic compound of
the thiazolinone type is formed (see Fig. 2).
The majority of biochemically important compounds, such as amino
acids, biogenic amines, and sugars, contain these nucleophilic groups
and, thus, may react with carbon disulfide. This is true of a large
number of substances existing in the organism.
A number of possible mechanisms of the effects of carbon
disulfide on the organism have been postulated including:
(a) the chelating effect of carbon disulfide metabolites on
various metals, essential for the functioning of enzymes;
(b) the effect of carbon disulfide on enzymatic systems;
(c) disturbances of vitamin metabolism;
(d) impairment of catecholamine metabolism;
(e) changes in lipid metabolism;
(f) interaction with microsomal drug-metabolizing enzyme
systems.
The responsibility of metabolites for the discoloration of iodine
azide remained hypothetical until Pergal et al. (1972a,b) isolated 3
metabolites from human urine and identified 2 of them as thiourea and
mercaptothiazolinone; thiourea is by far the most important of these
metabolites. Later, Pergal et al. (1977a) developed a quantitative
method for the micro-determination of thiourea in the urine of exposed
workers or of alcoholics treated with tetraethylthiuramdisulfide
(TETD, Disulfiram, Antabuse). The authors suggested that the third
metabolite was 2-mercapto-thiazoline-4-carbamic acid (Pergal et al.,
1977b). Tetraethylthiuramdisulfide is metabolized in a way that
liberates carbon disulfide (Fig. 1). Consequently, alcoholics treated
with this agent are exposed to carbon disulfide and its metabolites.
Skalicka (1967) and Novak et al. (1968) measured the iodine-azide
reaction and determined diethyldithiocarbamates (DDC) in the urine of
alcoholics treated with TETD. These results led Djuric et al. (1973)
to use TETD as a test for the evaluation of the metabolic rate of
sulfur compounds in the organism of workers, the so-called "antabuse
test".
Studies on the microsomal metabolism of carbon disulfide in the
liver of rats revealed that it was desulfurated to form
carbonylsulfide and that this was further oxidized, yielding carbon
dioxide which was exhaled (De Matteis & Seawright, 1973; De Matteis,
1974; Dalvi et al., 1974).
Data from human and animal studies on ethereal sulfate excretion
(Magos, 1973) have shown that bivalent sulfur represents a small part
of retained carbon disulfide, probably less than 5%. The major pathway
leads to the formation of sulfates that are excreted in urine.
5. BIOCHEMICAL EFFECTS OF CARBON DISULFIDE
From the chemical point of view, carbon disulfide is highly
reactive with nucleophilic reagents characterized by the presence of a
group with a free pair of electrons in the molecule. The most
important nucleophilic groups are mercapto (-SH), amino (-NH2) and
hydroxy (-OH) groups (Vasak & Kopecky 1967). However, physiological pH
values do not favour these reactions (Kopecky, 1977, private
communication).
According to the chemical structure of compounds participating in
the reactions, carbon disulfide will produce dithiocarbamic,
trithiocarbonic, or xanthogenic acid. If carbon disulfide reacts with
an organic compound with 2 nucleophilic groups, a cyclic compound of
the thiazolinone type is formed (see Fig. 2).
The majority of biochemically important compounds, such as amino
acids, biogenic amines, and sugars, contain these nucleophilic groups
and, thus, may react with carbon disulfide. This is true of a large
number of substances existing in the organism.
A number of possible mechanisms of the effects of carbon
disulfide on the organism have been postulated including:
(a) the chelating effect of carbon disulfide metabolites on
various metals, essential for the functioning of enzymes;
(b) the effect of carbon disulfide on enzymatic systems;
(c) disturbances of vitamin metabolism;
(d) impairment of catecholamine metabolism;
(e) changes in lipid metabolism;
(f) interaction with microsomal drug-metabolizing enzyme
systems.
 5.1 Chelating Effects of Carbon Disulfide Metabolites
The hypothesis of the chelating effect of carbon disulfide
metabolites was advanced by Cohen and coworkers and was based on
experiments on rabbits (Cohen et al., 1958; Paulus et al., 1957;
Scheel et al., 1960; Scheel, 1965, 1967). Considerable shifts were
found in the copper and zinc contents of various tissues, especially
in the nervous tissue, in rabbits poisoned by carbon disulfide. The
concentration of copper in the brain and spinal cord of animals killed
2 weeks after final exposure was less than half of that in the
controls. On the other hand, the zinc level in exposed rabbits was 20%
higher than that in the control animals. In general, pathological
examination of the tissues did not indicate any changes, except in the
kidneys and in the spinal cord, which showed marked degeneration of
the axis of the cylinder. The Purkinje cells of the cerebrum also
showed signs of degeneration.
The following hypothesis, based on an observation that the levels
of metal ions in tissues were altered by exposure to carbon disulfide,
was formulated by Scheel (1967):
-- carbon disulfide reacts with the amino groups of amino acids
and proteins to form thiocarbamate in blood and tissues, as was stated
by Soucek & Madlo (1956);
-- thiocarbamates, possessing sulfhydryl groups, may chelate
polyvalent inorganic ions. Because of the low dissociation of the
product, they would, thus, interfere with cellular metabolism.
-- when such interference becomes sufficiently limiting, the body
would respond by oxidizing fat and general loss in body-weight would
occur;
-- ultimately, as the metabolic limitation increases, cellular
death and loss of associated function would occur, producing signs of
tissue injury.
Since the entire hypothesis rests on chelation of metal ions, it
should be possible to prevent the occurrence of such an effect by
supplying an excess of metal ions in the diet of animals exposed to
carbon disulfide (Scheel et al., 1960; Scheel, 1967). Such a
protective effect is claimed to have been achieved by Scheel (1967).
The hypothesis of a chelating effect has been supported by the
results of other studies including those of Andreeva (1970), who
reported an increase in zinc and copper excretion in exposed rats, and
Lukas et al. (1974), who found increased copper levels in the
peripheral nervous tissue of exposed rats. A decreased level of
ceruloplasmin in rats with experimental carbon disulfide
polyneuropathy was reported by Lukas et al. (1975). This decrease was
related to the intensity and extent of the electromyographic signs of
polyneuropathy. Gadaskina & Andreeva (1969) and Cimbarevic (1970)
noticed a decrease in ceruloplasmin activity in workers exposed to
carbon disulfide for more than 10 years. However, in other studies,
the ceruloplasmin levels in exposed workers were in the normal range
(Andruszczak, 1967; Kujalova, 1973). Andruszczak (1967) found
increased ceruloplasmin levels in patients suffering from chronic
carbon disulfide poisoning.
Increased excretion of trace metals in the urine of workers
exposed to carbon disulfide was not observed in studies by Djuric et
al. (1967). Hernberg & Nordman (1969), and Hernberg et al. (1969).
However, these negative results do not necessarily exclude a chelating
effect, since exposure may have been too low. Thus, the more recent
results of El Gazzar et al. (1973) showing a temporary increase in the
zinc contents of all serum protein fractions as well as in urinary
excretion may reflect the effects of a higher exposure level than in
the previous studies.
It is known that copper and zinc ions are essential for the
prosthetic groups of many enzymes. The neurotoxic action of carbon
disulfide and its interference with the activity of many enzymes could
easily be explained by chelating effects. Zinc is required for the
activity of enzymes such as lactic acid dehydrogenase (EC 1.1.1.27)a,
carbonic anhydrase (EC 4.2.1.1), glutamate dehydrogenase (EC 1.4.1.2),
and alcohol dehydrogenase (EC 1.1.1.1). Copper, on the other hand,
represents a cofactor of pyridoxol, a form of vitamin B6.
Copper is required for the proper functioning of enzymes such as
cytochrome c oxidase (EC 1.9.3.1), the coenzyme A dehydrogenase
system, and dopamine ß hydroxylase (EC 1.14.17.1). The loss of copper
from the spinal cord is accompanied by cellular damage, producing
tissue degeneration. Disturbances of the central and peripheral
nervous systems, resulting from carbon disulfide exposure, could be
connected with the loss of copper due to chelation and consequent
inhibitory effects on enzyme systems (Scheel, 1967).
a The numbers within parentheses following the names of enzymes are
those assigned by the Enzyme Commission of the Joint IUPAC-IUB
Commission on Biochemical Nomenclature.
5.2 Effects on Enzyme Systems
Inhibition of monoamine-oxidase (EC 1.4.3.4) (MAO) activity
occurs as soon as exposure of an animal to carbon disulfide begins,
but it is reversible (Magistretti & Peirone, 1961; Lazarev et al.,
1965). The mechanism of inhibition is not yet clear, but it is known
that MAO contains a copper pyridoxal complex. Vasak & Kopecky (1967)
found a decrease in catecholamine in the urine of exposed rats. This
result suggests the possibility that carbon disulfide forms a compound
with catecholamine which cannot be split by MAO. However, Magos &
Jarvis (1970b), who also exposed rats to carbon disulfide, did not
find any inhibition of MAO. They suggest that Vasak & Kopecky's (1967)
finding could be explained by the inhibition of dopamine ß
hydroxylase.
Alkaline phosphatase (EC 3.1.3.1) activity was inhibited in the
tissues and serum of rabbits exposed for more than 22 weeks to high
concentrations of carbon disulfide, i.e., concentrations up to about
2350 mg/m3 (750 ppm) (Cohen et al., 1959). Chervenka & Wilcox (1956)
did not find any influence of carbon disulfide on derivatives of
chymotrypsinogen or on succinate dehydrogenase (EC 1.3.99.1) activity
and Minden et al. (1967) did not register any effects on glycolytic
enzymes, Kreb's cycle enzymes, and transaminases in experimental
animals.
No changes in glycolysis were found in the brain tissue of rats
after either acute or chronic exposure to carbon disulfide (Tarkowski
& Cremer, 1972; Tarkowski, 1973). Changes in the brain free amino acid
metabolism observed in rats exposed to a carbon disulfide
concentration of 2400 mg/m3 for 15 h included reductions in the
levels of glutamic delta-amino butyric acids. These effects were
accompanied by decreased activity of brain glutamate decarboxylase
(EC 4.1.1.15) (Tarkowski, 1974).
Both, acute and chronic exposures of animals to carbon disulfide
result in changes in mitochondrial respiration and oxidative
phosphorylation. Respiration of the brain mitochondria was partly
inhibited in rats exposed to carbon disulfide (Tarkowski & Sobczak,
1971); cytochrome oxidase activity was also inhibited (Tarkowski,
Wronska-Nofer, 1966). Oxidative phosphorylation in the mitochondria
was partly inhibited and partly uncoupled, and was accompanied by a
reduction in the activity of adenosinetriphosphatase (EC 3.6.1.3)
(Tarkowski & Sobczak, 1971).
Gregorczyk et al. (1975a,b) did not find any changes in liver
enzymes, proteins, and free amino acids in rats exposed to a carbon
disulfide concentration of 1300 mg/m3 for 12-26 weeks, and 5 and 10 h
daily. The authors concluded that carbon disulfide was not hepatotoxic
under such exposure conditions; this seems to be supported by the fact
that they did not find any changes in the blood serum enzymes
(Gregorczyk et al. 1975a,b).
Decreased activity of triosephosphate dehydrogenase (EC 1.2.1.9),
lactate dehydrogenase, and glycerophosphate dehydrogenase found in the
muscles of rats with developed neuropathy was accompanied by an
increase in hexokinase (EC 2.7.1.1) activity (Lukas et al., 1977).
Further studies should establish which enzymes are inhibited by
exposure of the organism to carbon disulfide, at what levels of
exposure, and the inhibition of which enzyme systems would present a
significant health hazard.
5.3 Effects on Vitamin Metabolism
5.3.1 Vitamin B6
There are 3 forms of vitamin B6, i.e., pyridoxol, pyridoxal, and
pyridoxamine, that play the role of coenzymes in various enzyme
systems. These 3 forms are in equilibrium because they are
enzymatically converted into each other (Fig. 3). The binding of one
form of vitamin B6 will block the reactions of the enzymes containing
the remaining forms. Vasak & Kopecky (1967) reported that carbon
disulfide reacted in vitro with pyridoxamine to form a salt of
pyridoxamine dithiocarbamic acid. Some authors think that this process
could also occur in vivo, causing inhibition of the enzyme systems
in which vitamin B6 is involved as a coenzyme. Disturbance of
pyridoxol metabolism during chronic intoxication was shown in
experiments on rats by Kujalova (1971). After the tryptophan load
test, excretion of xanthurenic acid increased in exposed animals while
excretion of pyridoxol acid decreased.
The temporal development and the severity of this disturbance
depended on the diet given to the animals in question (Kujalova, 1971;
Gorny, 1974). Pyridoxol deficiency due to carbon disulfide
intoxication could be eliminated in the animals by giving a diet rich
in pyridoxol (10 times the normal dietary level). However, this diet
did not prevent the development of neuropathy (Lukas, 1970) or
influence the deterioration in motor activity in rats (Frantik, 1970;
Teisinger, 1971).
5.1 Chelating Effects of Carbon Disulfide Metabolites
The hypothesis of the chelating effect of carbon disulfide
metabolites was advanced by Cohen and coworkers and was based on
experiments on rabbits (Cohen et al., 1958; Paulus et al., 1957;
Scheel et al., 1960; Scheel, 1965, 1967). Considerable shifts were
found in the copper and zinc contents of various tissues, especially
in the nervous tissue, in rabbits poisoned by carbon disulfide. The
concentration of copper in the brain and spinal cord of animals killed
2 weeks after final exposure was less than half of that in the
controls. On the other hand, the zinc level in exposed rabbits was 20%
higher than that in the control animals. In general, pathological
examination of the tissues did not indicate any changes, except in the
kidneys and in the spinal cord, which showed marked degeneration of
the axis of the cylinder. The Purkinje cells of the cerebrum also
showed signs of degeneration.
The following hypothesis, based on an observation that the levels
of metal ions in tissues were altered by exposure to carbon disulfide,
was formulated by Scheel (1967):
-- carbon disulfide reacts with the amino groups of amino acids
and proteins to form thiocarbamate in blood and tissues, as was stated
by Soucek & Madlo (1956);
-- thiocarbamates, possessing sulfhydryl groups, may chelate
polyvalent inorganic ions. Because of the low dissociation of the
product, they would, thus, interfere with cellular metabolism.
-- when such interference becomes sufficiently limiting, the body
would respond by oxidizing fat and general loss in body-weight would
occur;
-- ultimately, as the metabolic limitation increases, cellular
death and loss of associated function would occur, producing signs of
tissue injury.
Since the entire hypothesis rests on chelation of metal ions, it
should be possible to prevent the occurrence of such an effect by
supplying an excess of metal ions in the diet of animals exposed to
carbon disulfide (Scheel et al., 1960; Scheel, 1967). Such a
protective effect is claimed to have been achieved by Scheel (1967).
The hypothesis of a chelating effect has been supported by the
results of other studies including those of Andreeva (1970), who
reported an increase in zinc and copper excretion in exposed rats, and
Lukas et al. (1974), who found increased copper levels in the
peripheral nervous tissue of exposed rats. A decreased level of
ceruloplasmin in rats with experimental carbon disulfide
polyneuropathy was reported by Lukas et al. (1975). This decrease was
related to the intensity and extent of the electromyographic signs of
polyneuropathy. Gadaskina & Andreeva (1969) and Cimbarevic (1970)
noticed a decrease in ceruloplasmin activity in workers exposed to
carbon disulfide for more than 10 years. However, in other studies,
the ceruloplasmin levels in exposed workers were in the normal range
(Andruszczak, 1967; Kujalova, 1973). Andruszczak (1967) found
increased ceruloplasmin levels in patients suffering from chronic
carbon disulfide poisoning.
Increased excretion of trace metals in the urine of workers
exposed to carbon disulfide was not observed in studies by Djuric et
al. (1967). Hernberg & Nordman (1969), and Hernberg et al. (1969).
However, these negative results do not necessarily exclude a chelating
effect, since exposure may have been too low. Thus, the more recent
results of El Gazzar et al. (1973) showing a temporary increase in the
zinc contents of all serum protein fractions as well as in urinary
excretion may reflect the effects of a higher exposure level than in
the previous studies.
It is known that copper and zinc ions are essential for the
prosthetic groups of many enzymes. The neurotoxic action of carbon
disulfide and its interference with the activity of many enzymes could
easily be explained by chelating effects. Zinc is required for the
activity of enzymes such as lactic acid dehydrogenase (EC 1.1.1.27)a,
carbonic anhydrase (EC 4.2.1.1), glutamate dehydrogenase (EC 1.4.1.2),
and alcohol dehydrogenase (EC 1.1.1.1). Copper, on the other hand,
represents a cofactor of pyridoxol, a form of vitamin B6.
Copper is required for the proper functioning of enzymes such as
cytochrome c oxidase (EC 1.9.3.1), the coenzyme A dehydrogenase
system, and dopamine ß hydroxylase (EC 1.14.17.1). The loss of copper
from the spinal cord is accompanied by cellular damage, producing
tissue degeneration. Disturbances of the central and peripheral
nervous systems, resulting from carbon disulfide exposure, could be
connected with the loss of copper due to chelation and consequent
inhibitory effects on enzyme systems (Scheel, 1967).
a The numbers within parentheses following the names of enzymes are
those assigned by the Enzyme Commission of the Joint IUPAC-IUB
Commission on Biochemical Nomenclature.
5.2 Effects on Enzyme Systems
Inhibition of monoamine-oxidase (EC 1.4.3.4) (MAO) activity
occurs as soon as exposure of an animal to carbon disulfide begins,
but it is reversible (Magistretti & Peirone, 1961; Lazarev et al.,
1965). The mechanism of inhibition is not yet clear, but it is known
that MAO contains a copper pyridoxal complex. Vasak & Kopecky (1967)
found a decrease in catecholamine in the urine of exposed rats. This
result suggests the possibility that carbon disulfide forms a compound
with catecholamine which cannot be split by MAO. However, Magos &
Jarvis (1970b), who also exposed rats to carbon disulfide, did not
find any inhibition of MAO. They suggest that Vasak & Kopecky's (1967)
finding could be explained by the inhibition of dopamine ß
hydroxylase.
Alkaline phosphatase (EC 3.1.3.1) activity was inhibited in the
tissues and serum of rabbits exposed for more than 22 weeks to high
concentrations of carbon disulfide, i.e., concentrations up to about
2350 mg/m3 (750 ppm) (Cohen et al., 1959). Chervenka & Wilcox (1956)
did not find any influence of carbon disulfide on derivatives of
chymotrypsinogen or on succinate dehydrogenase (EC 1.3.99.1) activity
and Minden et al. (1967) did not register any effects on glycolytic
enzymes, Kreb's cycle enzymes, and transaminases in experimental
animals.
No changes in glycolysis were found in the brain tissue of rats
after either acute or chronic exposure to carbon disulfide (Tarkowski
& Cremer, 1972; Tarkowski, 1973). Changes in the brain free amino acid
metabolism observed in rats exposed to a carbon disulfide
concentration of 2400 mg/m3 for 15 h included reductions in the
levels of glutamic delta-amino butyric acids. These effects were
accompanied by decreased activity of brain glutamate decarboxylase
(EC 4.1.1.15) (Tarkowski, 1974).
Both, acute and chronic exposures of animals to carbon disulfide
result in changes in mitochondrial respiration and oxidative
phosphorylation. Respiration of the brain mitochondria was partly
inhibited in rats exposed to carbon disulfide (Tarkowski & Sobczak,
1971); cytochrome oxidase activity was also inhibited (Tarkowski,
Wronska-Nofer, 1966). Oxidative phosphorylation in the mitochondria
was partly inhibited and partly uncoupled, and was accompanied by a
reduction in the activity of adenosinetriphosphatase (EC 3.6.1.3)
(Tarkowski & Sobczak, 1971).
Gregorczyk et al. (1975a,b) did not find any changes in liver
enzymes, proteins, and free amino acids in rats exposed to a carbon
disulfide concentration of 1300 mg/m3 for 12-26 weeks, and 5 and 10 h
daily. The authors concluded that carbon disulfide was not hepatotoxic
under such exposure conditions; this seems to be supported by the fact
that they did not find any changes in the blood serum enzymes
(Gregorczyk et al. 1975a,b).
Decreased activity of triosephosphate dehydrogenase (EC 1.2.1.9),
lactate dehydrogenase, and glycerophosphate dehydrogenase found in the
muscles of rats with developed neuropathy was accompanied by an
increase in hexokinase (EC 2.7.1.1) activity (Lukas et al., 1977).
Further studies should establish which enzymes are inhibited by
exposure of the organism to carbon disulfide, at what levels of
exposure, and the inhibition of which enzyme systems would present a
significant health hazard.
5.3 Effects on Vitamin Metabolism
5.3.1 Vitamin B6
There are 3 forms of vitamin B6, i.e., pyridoxol, pyridoxal, and
pyridoxamine, that play the role of coenzymes in various enzyme
systems. These 3 forms are in equilibrium because they are
enzymatically converted into each other (Fig. 3). The binding of one
form of vitamin B6 will block the reactions of the enzymes containing
the remaining forms. Vasak & Kopecky (1967) reported that carbon
disulfide reacted in vitro with pyridoxamine to form a salt of
pyridoxamine dithiocarbamic acid. Some authors think that this process
could also occur in vivo, causing inhibition of the enzyme systems
in which vitamin B6 is involved as a coenzyme. Disturbance of
pyridoxol metabolism during chronic intoxication was shown in
experiments on rats by Kujalova (1971). After the tryptophan load
test, excretion of xanthurenic acid increased in exposed animals while
excretion of pyridoxol acid decreased.
The temporal development and the severity of this disturbance
depended on the diet given to the animals in question (Kujalova, 1971;
Gorny, 1974). Pyridoxol deficiency due to carbon disulfide
intoxication could be eliminated in the animals by giving a diet rich
in pyridoxol (10 times the normal dietary level). However, this diet
did not prevent the development of neuropathy (Lukas, 1970) or
influence the deterioration in motor activity in rats (Frantik, 1970;
Teisinger, 1971).
 That pyridoxol deficiency might result from carbon disulfide
intoxication was proved by Gorny (1971), who showed that the pyridoxal
phosphate concentration decreased in the serum of acutely intoxicated
rats. Furthermore, increases have been reported in nicotinamides, the
metabolites of nicotinic acid (Nofer & Wrofiska-Nofer, 1966) and in
hydroxyindolacetic acid (a metabolite of serotonine) (Abuczewicz et
al., 1971) in the urine of rats exposed to carbon disulfide. All these
substances are derived from tryptophan.
In a review on the mechanisms of chronic carbon disulfide
poisoning, Teisinger (1971) concluded that disturbance of vitamin B6
metabolism was obvious but that it did not play a major role in the
pathogenesis of nervous tissue lesions.
Transaminases are sensitive to vitamin B6 deficiency. Thus,
metabolic pathways in which transaminases are involved may be
inhibited. This is the case in tryptophan metabolism, where it is
manifested by increased excretion of xanthurenic acid in both man
(Tintera et al., 1972) and rat (Fig. 4) (Abramova, 1967).
That pyridoxol deficiency might result from carbon disulfide
intoxication was proved by Gorny (1971), who showed that the pyridoxal
phosphate concentration decreased in the serum of acutely intoxicated
rats. Furthermore, increases have been reported in nicotinamides, the
metabolites of nicotinic acid (Nofer & Wrofiska-Nofer, 1966) and in
hydroxyindolacetic acid (a metabolite of serotonine) (Abuczewicz et
al., 1971) in the urine of rats exposed to carbon disulfide. All these
substances are derived from tryptophan.
In a review on the mechanisms of chronic carbon disulfide
poisoning, Teisinger (1971) concluded that disturbance of vitamin B6
metabolism was obvious but that it did not play a major role in the
pathogenesis of nervous tissue lesions.
Transaminases are sensitive to vitamin B6 deficiency. Thus,
metabolic pathways in which transaminases are involved may be
inhibited. This is the case in tryptophan metabolism, where it is
manifested by increased excretion of xanthurenic acid in both man
(Tintera et al., 1972) and rat (Fig. 4) (Abramova, 1967).
 5.3.2 Nicotinic acid
Exposure to carbon disulfide resulted in increased excretion of
methyl nicotinamide, a nicotinic acid metabolite, in the urine of rats
(Liniecki, 1960; Wrofiska-Nofer et al., 1965). An increase in serum
lipids during the exposure of rabbits and rats to carbon disulfide was
prevented by administration of nicotinic acid (Nofer & Wrofiska-Nofer,
1966; Wrofiska-Nofer, 1970). The biochemical background of this
phenomenon is still obscure, but a hypothesis based on the
interference of carbon disulfide with pyridine nucleotide and
nicotinamide metabolism has been advanced (Nofer & Wrofiska-Nofer,
1966). However, feeding nicotinic acid to the exposed animals did not
prevent the development of neuropathies in these experiments.
As the increase in urinary excretion of nicotinamide metabolites
did not occur at the cost of the systematic pool of
nicotinamide-adenine dinucleotides, Wrofiska-Nofer et al. (1970)
suggested that it reflected an increase in the whole turnover rate.
The mechanism of the process was not clarified but it may be assumed
that an increase in the synthesis via tryptophan occurs.
5.4 Effects on Catecholamine Metabolism
Disturbance of the catecholamine metabolism can play a part in
many pathological processes. In studies on rats acutely intoxicated
with carbon disulfide, significant changes were found in the brain
catecholamine metabolism (Magos & Jarvis, 1970b; Magos, 1975). There
was a decrease in the level of noradrenaline accompanied by an
increased concentration of dopamine. Inhibition of dopamine
ß-hydroxylase, an essential enzyme in catecholamine metabolism, was
demonstrated in vivo in studies on rats by Magos (1975), and in
vitro by McKenna & Di Stefano (1975). Magos (1975) advanced a theory
concerning the central role played by catecholamine disturbances in
carbon disulfide pathology, especially where central nervous system
changes were involved, and in cardiovascular pathology. Cavalleri et
al. (1977) suggested that this could also explain the involvement of
the endocrine system.
5.5 Effects on Lipid Metabolism
Disturbance in the lipid metabolism has long since been linked
with carbon disulfide exposure. Increased levels of serum lipids, free
and total cholesterol, and ß-lipoproteins have been reported in
rabbits exposed to carbon disulfide (Paterni et al., 1958; Cohen et
al., 1959; Prerovska et al., 1961). Harashima et al. (1960) reported
elevated total and esterified cholesterol levels in the serum of
heavily exposed workers, whereas workers exposed to levels of about
15-60 mg/m3 (5-19 ppm) displayed normal serum cholesterol
concentrations. Elevated cholesterol levels were also found in exposed
workers by Manu et al. (1971) and in patients with previous exposure
to carbon disulfide (the patients had not been exposed for several
years) by Graovac-Leposavic et al. (1977). Higher levels of serum
lipids and especially cholesterol may be due to an increased rate of
synthesis in the liver and to the inhibition of the degradation of
lipids (Wrofiska-Nofer, 1969; Laurman & Wronska-Nofer, 1977). The fact
that elevated cholesterol levels have not been found consistently may
be explained by the different exposure levels in different studies
(Toyoma & Sakurai, 1967). Bittersohl & Thiele (1977) found that a
higher frequency of cholesterol values exceeding 6.72 mmol/litre
(260 mg/dl) in workers exposed to carbon disulfide was strongly
correlated with the duration of exposure.
A decreased clearing factor activity was found by Ruikka (1959)
in workers exposed to carbon disulfide and Martino et al. (1963, 1964)
reported an elevated ß-lipoprotein fraction in the serum of exposed
workers.
Changes in the lipid metabolism found in the aorta tissue may
contribute to the development of atheromatic changes in blood vessels
(Wrofiska-Nofer, 1976).
An increase in serum lipids during the exposure of rabbits to
carbon disulfide was prevented by administration of nicotinic acid
(Nofer & Wrofiska-Nofer, 1966) and in rats (Wrofiska-Nofer, 1970)
(section 5.3.2).
5.6 Interaction with Microsomal Drug Metabolism
An important feature of the liver toxicity caused by carbon
disulfide seems to be the destruction of cytochrome P-450 (Bond & De
Matteis, 1969). There is evidence that this effect is due to the
oxidative desulfuration of carbon disulfide by mixed-function oxidases
(De Matteis & Seawright, 1973). The resulting, highly reactive, sulfur
becomes covalently bound to the microsomal protein (Dalvi et al.,
1974; De Matteis, 1974; Jarvisalo et al., 1977), mainly to the
apoprotein of cytochrome P-450 (Neal et al., 1976; Jarvisalo & De
Matteis, 1977; Savolainen et al., 1977a). It is possible that the
liberated sulfur is the real toxic agent in liver toxicity arising
from carbon disulfide exposure.
Experiments on rats have shown that exposure to carbon disulfide
in concentrations up to 1250 mg/m3 (400 ppm) for 8 h is followed by
an increase in microsomal RNA content and in total protein in the
hepatic microsomal fraction and also increased incorporation of
2,4-3H-L-phenylalanine in liver microsomes (Freundt et al., 1974b).
Such changes follow the action of inducing agents, such as
phenobarbital, in the microsomal enzyme system. It has, therefore,
been suggested that carbon disulfide may have an inducing as well as
an inhibiting effect on mixed-function oxygenases. However, there is
no real evidence of such an effect at present (Freundt, 1977).
6. CARBON DISULFIDE POISONING
6.1 Historical Review
At the end of 1850, several physicians observed cases of strange
nervous and mental diseases, the origin of which remained obscure. In
1856, Delpech, reported 24 cases of carbon disulfide poisoning and
confirmed the diagnosis by animal experiments (Delpech, 1856a,b). In
1863, he reported 80 more cases of "carbon disulfide neurosis"
(Delpech, 1863). Cases of chronic poisoning were also reported in
England by Bruce (1884) and Foreman (1886). Laudenheimer (1899)
described carbon disulfide poisoning in German vulcanization shops,
stirring public opinion by drawing attention to about 50 cases of
"insanity". As further cases were reported in the USA (Jump & Cruice,
1904; Francine, 1905), the need for hygienic improvement in work
places was recognized. The first epidemic of carbon disulfide
poisoning due to the vulcanization process ended at the beginning of
the 20th century. At the same time, the viscose rayon industry started
to develop and expanded rapidly. Sporadic cases of carbon disulfide
poisoning in the viscose industry were reported between 1900 and 1930
(Quarelli, 1928) but the problem became serious in the 1930s.
Raneletti (1933), Quarelli (1934) and others described cases of
psychotic and polyneurotic disorders and extrapyramidal disturbances
(Audio-Gianotti, 1932; Teisinger, 1934).
In Japan, the viscose rayon industry was established in 1916 and,
in 1929, the first cases of carbon disulfide poisoning were reported
by Tokuhara, followed by other authors (review by Kubota, 1967). Many
cases of poisoning were also described in the USA (Hamilton, 1925,
1940; Bashore et al., 1938). The so-called Pennsylvania study by Gordy
& Trumper (1938) resulted in the establishment of the first TLV of
20 ppm adopted by the American Standards Association (1941). As the
hygienic standard in this industry improved, the incidence of severe
poisoning decreased. However, during World War II, the hygienic
situation in the expanding viscose industry deteriorated and severe
poisoning again became common.
During and after World War II, many cases of carbon disulfide
poisoning were reported, mainly from Italy (e.g., Vigliani et al.,
1944; Vigliani, 1946) but also from Belgium (Langelez, 1946; Merlevde,
1951) and Finland (Noro, 1944). After World War II, the viscose rayon
industry spread to many developing countries, where the whole sequence
of degrees of exposure was repeated. In the developed countries,
attention is now focused on slowly developing symptoms due to
long-standing exposure to relatively low concentrations.
6.2 Clinical Picture of Carbon Disulfide Poisoning
Carbon disulfide intoxication has been classified as hyperacute,
acute, subacute, and chronic.
Hyperacute poisoning occurs in extreme cases of massive exposure
for a short time to concentrations of about 10 000 mg/m3 or more. The
victim quickly falls into a coma and eventually dies. Acute and
subacute poisoning occurs with short exposure to carbon disulfide
concentrations ranging from 3000-5000 mg/m3 with predominantly
psychiatric and neurological signs and symptoms such as extreme
irritability, uncontrolled anger, rapid mood changes including maniac
delirium and hallucinations, paranoic ideas, and suicidal tendencies.
Other symptoms include memory defects, severe insomnia, nightmares,
fatigue, loss of appetite, gastrointestinal troubles, asthenia, and
interference with sexual functions, such as impotency.
The symptoms and signs of chronic carbon disulfide poisoning were
described by Vigliani (1946, 1961) and the following classification of
the different syndromes was suggested by Nesswetha & Nesswetha (1967):
-- psychoses characterized by manic and depressive symptomatology
and disorientation;
-- polyneuropathy of the lower extremities, with diminished or
completely absent Achilles and patellar tendon reflexes, sensory
disturbances in a glove-stocking distribution, diminished faradic and
galvanic excitability, and decrease of the motor and sensory
conduction velocity in the peripheral nerves;
-- disturbances of the gastrointestinal tract in the form of
chronic, hyper- and hypoacidic gastritis and duodenal ulceration;
-- myopathy of the calf muscles;
-- neurasthenic syndrome with disturbances in the autonomous
nervous system,
-- optic neuritis;
-- atherosclerotic vasculoencephalopathy; the principal forms
being bulbar-paralytic, hemiplegic, or extrapyramidal.
The typical mental deterioration has been called an organic
psycho-syndrome, which may be due to general cerebral atherosclerosis,
to direct toxic action upon the brain cells, or to both.
The pattern of carbon disulfide poisoning has changed with
improvements in hygienic standards in industry. However, when the
viscose industry is established in a country with no former
experience, there is always the risk of severe poisoning; this has
occurred many times.
6.3 Effects on Organ Systems
6.3.1 Dermatological effects
Liquid carbon disulfide represents a severe irritant for both the
skin and mucosa. Hueper (1936) found blisters in viscose rayon workers
and also in experimental animals that often resembled second and third
degree burns.
6.3.2 Ophthalmological effects
Studies on the ophthalmological effects of carbon disulfide in
animals have mostly been of a histological nature. In 1899, Koester
observed changes in the retinal ganglia. Seto (1958) found vacuolar
degeneration and tigrolysis in the retina, and atrophy of the optic
nerve in rabbits exposed to a carbon disulfide concentration of about
3700 mg/m3 for 1-3 h.
Ophthalmological examinations in man are informative because it
is possible to observe the visual capacity and to study the changes in
the vessels of the ocular fundus directly. According to the literature
(e.g., Nunziante-Cesaro et al., 1952; Savic, 1967) the following
effects have been observed:
-- changes in the motility of the eyelids;
-- changes in the sensitivity of the cornea and conjunctiva;
-- changes in the motility of the ocular bulbus;
-- changes in convergency and accommodation;
-- morphological changes in the fundus such as focal haemorrhage,
exudative changes, atrophy of the optic nerve, retrobulbar neuritis,
microaneurysms, and sclerotic changes of the blood vessels;
-- functional changes, e.g., disturbances of colour vision,
adaptation to the dark, reaction of the pupil to light, accommodation,
and decrease in visual accuracy.
Most of the effects mentioned above have resulted from heavy
exposure. Many that were observed decades ago, when the observations
often lacked comparison materials, are open to criticism. Nowadays,
under prevailing working conditions such grave changes are rarely, if
ever, seen.
Increased systolic and diastolic blood pressure in the retinal
arteries and damage to the arterial walls have been reported (Maugeri
et al., 1966d; Goto et al., 1971). Maugeri et al. (1966d, 1967)
studied the arterial pressure in 107 workers who had been exposed to
carbon disulfide at concentrations ranging from 200-500 mg/m3, with
peak concentrations of up to 900 mg/m3, for 1-9 years. Using an
ophthalmodynamographic method, the authors found an average increase
in both systolic and diastolic pressures in exposed workers of about
18.4/14.7 kPa (138/110 mmHg) compared with controls which averaged
15.3/11.6 kPa (115/87 mmHg). The increase was more marked for the
diastolic pressure than for the systolic. Measurement of the pressure
of the ophthalmic arteries may be a test that needs further evaluation
in the light of the demonstrated disturbance of the brain
catecholamine metabolism induced by carbon disulfide.
Savic (1967) studied changes in the nervous system of the eyes of
young workers exposed to carbon disulfide levels of 100-400 mg/m3.
Changes were observed but only after a long period of exposure
(>5 years) (Hotta & Savic, 1972). Retinal microaneurysms have been
reported in a high proportion of Japanese viscose rayon workers (Goto
& Hotta, 1967; Goto et al., 1971; Sugimoto et al., 1976). In one
study, the prevalences of microaneurysms as judged by fundus
photography, were 8% and 2% among exposed and unexposed male workers,
respectively (Goto & Hotta, 1967). In a subsequent study (Hotta &
Goto, 1971; Goto et al., 1971), a prevalence of 56% of microaneurysms
was found in the exposed group (241 subjects) and 15% in a control
group (30 subjects). The extremely high prevalence in the exposed
groups in these studies was due to biased selection of the subjects;
one-half of them were selected for examination because of known
microaneurysms revealed by ophthalmoscopy. Later studies by the same
team did not show such a high prevalence (Sugimoto et al., 1976), but
a statistically significant difference between exposed and unexposed
subjects still persisted. Retinaopathy was found in 89 out of 289
carbon disulfide workers (31%), but only in 2 out of 49 controls (4%).
The frequency of retinopathy was correlated with both the duration and
the intensity of exposure (Sugimoto et al., 1976). It has been
suggested that the retinal microaneurysms found in Japanese workers
were related to the diabetogenic action of carbon disulfide (Goto et
al., 1971). However, microaneurysms are generally associated with
hypoxia and, though they may occur in advanced cases of diabetes, they
are not, by any means, specific for the disease. Thus, to ascribe the
excess of aneurysms found in Japanese studies to subclinical diabetes
caused by carbon disulfide is not justified. It is more likely that
they were caused by a direct effect on the retinal vessels, possibly
in association with local hypoxia coacting with ethnic or
environmental factors.
The results of recent Finnish studies do not tally with the
Japanese findings. Out of 100 exposed men and 97 controls, only 3
exposed and 2 control subjects showed 1-5 microaneurysms, and one
exposed subject showed more than 5 (Raitta et al., 1974). Furthermore,
general narrowing of the arteries and calibre irregularity were so
common in the fundus of both the exposed and control subjects that
plain ophthalmoscopy did not have any discriminatory value in this
study. A subsequent Japanese-Finnish study showed that the differences
in values obtained in the 2 studies were true differences and were not
caused by inter-observer variation (Sugimoto et al., 1977).
The most interesting finding in a study by Raitta et al. (1974)
was the high frequency of delayed peripapillary filling that occurred
in 68 exposed and 38 unexposed men. The findings of Raitta & Tolonen
(1975), suggest that studying the microcirculation of the ocular
fundus by fluorescein-angiography and oculosphygmography would help in
the early detection of carbon disulfide effects, especially since the
vessels can be observed directly in their natural state.
Ophthalmoscopy is indicated for the examination of patients suspected
to be suffering from carbon disulfide poisoning and for following-up
such patients, but it is too prone to inter-and intra-observer
variation to be used routinely by plant physicians for regular health
checks. Fundus photography eliminates some of this variation, but its
evaluation should be standardized. The diagnosis of retinal
microaneurysms requires a fundus angioscreenography, but this method
is recommended for research purposes only.
It should be added that a mixture of carbon disulfide, hydrogen
sulfide and sulfuric acid mist from an acid bath caused
keratoconjunctivitis in exposed workers (Savic & Jovicic, 1965). This
phenomenon is called "spinner's eye".
6.3.3 Otological effects
Sulkowski & Latkowski (1969) observed that exposure to carbon
disulfide impaired hearing ability. The authors examined 60 workers
occupationally exposed to carbon disulfide for between one year and
more than 10 years. The workers were below 50 years of age.
Audiometric examinations showed impairment of hearing of the receptory
type in more than 50 workers and also a decreased ability to
distinguish sound intensity within the range 1.5-3.0 dB. This
suggested a central and supracochlear localization of the impairment.
Electronystagmographic examinations performed by these authors
disclosed reduced excitability of the vestibular apparatus, suggesting
an extralabyrinthine localization of the lesion. Loss of sensitivity
to high frequency tones has also been attributed to carbon disulfide
exposure (Zenk, 1970). Since noise levels are often very high in
viscose rayon plants, it is possible that the lesions found may be at
least partly due to this cause. Vestibular symptoms, as manifested by
vertigo and nystagmus may also be present in carbon disulfide
intoxication (Zenk, 1967, 1970).
6.3.4 Respiratory effects
Carbon disulfide is a known irritant, but few data exist about
its effects on the respiratory system (Zenk, 1967). Ranelletti (1933)
described chronic cough as a consequence of the irritant effect of
carbon disulfide, but it is necessary to take into consideration the
irritative effects of hydrogen sulfide and sulfuric acid mist and of
other irritants present in the air of viscose rayon plants. The
findings of Massoud et al. (1971) i.e., cough, phlegm, wheezing,
dyspnoea, precordial pain and palpitations, may also have been due to
such mixed exposure rather than to carbon disulfide alone.
6.3.5 Gastrointestinal effects
Gastrointestinal symptoms are common among heavily exposed
workers and patients with carbon disulfide poisoning. Bashore et al.
(1938) found a prevalence of such symptoms of 25%, Vigliani (1954),
28%, Karajovic et al. (1964), 66%, Lysina (1967), 27%, and Hass et al.
(1967), 27%. Since such symptoms occur among unexposed people too,
especially in shift work, the figures listed cannot be attributed
exclusively to carbon disulfide exposure (Knave et al., 1974). A
higher prevalence of gastrointestinal disorders and liver and bile
duct dysfunction was observed in 2 groups of workers (800 and 492
subjects, respectively) exposed to very low carbon disulfide
concentrations (4-12 mg/m3) than in a control group of 453 unexposed
workers. The prevalence in the 2 exposed groups was 4.7 and 5.6%,
respectively, and that in the control group, 2.2% (Murashko, 1975).
Later, Bittersohl & Thiele (1977) found a prevalence of 22.7% of
gastrointestinal changes in 309 exposed shift workers as opposed to
5.9% in 345 unexposed shift workers.
Based on histological studies of the gastric mucosa of 75 workers
exposed to carbon disulfide, Hassman et al. (1967) suggested
classification into 5 groups: normal gastric mucosa, superficial
gastritis, chronic gastritis with incipient atrophy, chronic atrophic
gastritis, and gastritis of undetermined type. Duodenal ulceration was
only found in 2 out of 75 subjects in this study. Of the workers
examined, 11% had dyspeptic complaints and gastritis was verified in
60% of the cases. A similar figure (66%) was found by Karajovic et al.
(1964).
6.3.6 Hepatic effects
Exposure to carbon disulfide has caused fatty degeneration and
haemorrhages of the liver in animals (Bashore et al., 1938).
Experimental studies on rats have shown that pretreatment with a drug
that stimulates liver microsome enzyme activity alters the degree of
liver damage on exposure to carbon disulfide. Thus, Bond et al. (1969)
reported necrosis in the livers of rats that had been pretreated with
phenobarbital and subsequently received a single oral dose of carbon
disulfide (1 ml/kg body weight) but not in rats treated only with
carbon disulfide. Moreover, only pretreated rats displayed hydropic
degeneration of the liver. Magos & Butler (1972) found that starvation
potentiated the effect of phenobarbital in rats subsequently treated
with carbon disulfide, and that the resulting hydropic degeneration
was reversible. Similar results were obtained by Freundt et al.
(1974a), when they administered a single oral dose of 1 ml of carbon
disulfide per kg body weight to rats pretreated with phenobarbital.
However, no degenerative changes in the liver were observed in
pretreated rats following exposure by inhalation to carbon disulfide
at about 60 mg/m3 (20 ppm) and 620 mg/m3 (200 ppm) for up to 7 days.
In experiments performed on rats with short-term (8-h) exposure to
carbon disulfide at concentrations ranging from 62-1250 mg/m3
(20-400 ppm), the energy potential of the organism was damaged mainly
because of a reversible augmentation of hepatic glycolysis (Kürzinger
& Freundt, 1969; Freundt & Kürzinger, 1975).
The possible effects of carbon disulfide on the liver have been
discussed in numerous reports of clinical observations. Some authors
deny that exposure to carbon disulfide results in toxic effects in the
liver, some claim degenerative changes of the hepatocytes, and others
state that sclerotic changes are produced under conditions of chronic
exposure. Such controversial observations can be explained by
nonuniformity in approach to the method of examination. Vidakovic et
al. (1965a) examined workers hospitalized because of chronic carbon
disulfide poisoning. The exposure of these workers had been extremely
high, ranging from 1400-2200 mg/m3 (Petrovic & Djuric, 1965).
Functional disturbances of the liver and fatty degeneration of
hepatocytes were found but no necrotic changes were observed.
Pirotskaja (1972) examined the protein-forming function of the
liver in workers (40 women and 6 men) who had been exposed to carbon
disulfide for 5 years. At first, the workers were exposed to a carbon
disulfide concentration of 90 mg/m3 but later, concentrations ranged
from 8-14 mg/m3. All exposed subjects were between 30 and 40 years of
age. The mean concentrations on cystine, thyroxine, methionine,
valine, leucine, and tryptophan in serum showed statistically
significant increases in comparison with a control group.
Statistically valid decreases occurred in the levels of glutamic acid,
aspartic acid, olanine, and lysine. The levels of histidine, arginine,
and serine did not differ from those in the control group. There was a
correlation between these changes and the clinical signs of carbon
disulfide intoxication. There was no difference in the serum contents
of total protein in the exposed and control groups.
6.3.7 Renal effects
Some authors have drawn attention to nephrosclerosis in autopsies
of patients with carbon disulfide poisoning (Uehlinger, 1952; Yamagata
et al., 1966; Sbertoli et al., 1969), but this could be ascribed to a
general atherosclerotic process induced by carbon disulfide. For
example, in the cases studied by Uehlinger (1952), typical
glomerulosclerosis of the Kimmelstiel-Wilson type was established in 4
patients, while the fifth showed arteriosclerotic injury of the
kidney. Obviously renal involvement represents a very late consequence
of heavy, protracted carbon disulfide exposure, since
Graovac-Leposavic & Jovicic (1971) reported that only one patient with
persistent kidney insufficiency had ever been diagnosed in a large
viscose rayon plant employing several thousands of workers. In another
study of viscose rayon workers, Hemberg et al. (1971) found a slight
but statistically significant rise in the mean plasma creatinine
concentration compared with a control group. All values were within
"normal" limits, however.
6.3.8 Haematological effects
Brieger (1949) studied the bone marrow of exposed rats and found
retarded maturation of the erythrocytes. Mild anaemia, a slight
decrease in the haemoglobin concentration, slight reticulocytosis,
eosinophilia, and hypercoagulability of the blood have also been
recorded in exposed rats (Vidakovic et al., 1965b).
Ivanova (1967) found a moderate decrease in the haemoglobin
concentration and erythrocyte count in men with only slight exposure
to carbon disulfide. Such results cannot be attributed to carbon
disulfide effects, but rather to poor standardization of the study
conditions, since significant haematological changes in the peripheral
blood were not observed, even in highly-exposed workers (Vidakovic &
Andjelkovski, 1965; Fahim et al., 1973). Thus, it has not been
confirmed that carbon disulfide causes anaemia or polyglobulia. It is
probable that the anaemia mentioned in some clinical reports reflects
malnutrition or some other nonoccupational cause, perhaps even
defective study design, rather than an effect of carbon disulfide.
The effects of carbon disulfide on blood coagulation mechanisms
are discussed in section 6.3.14.
6.3.9 The endocrine system
Damage to the endocrine structures with functional alterations
was described in animals by Ranelletti as early as 1931, and by
Audio-Gianotti in 1932. Impaired sexual function in patients with
carbon disulfide poisoning was reported by Gordy & Trumper (1938),
Langelez (1946), and Vigliani (1946). Vesce et al. (1953) observed a
decrease in 17-ketosteroids excretion in the urine of exposed rabbits.
This phenomenon was confirmed in studies on exposed workers by
Fruscella (1962) and Olienacz et al. (1964). Most of these
observations were made on workers belonging to older age-groups and
the confounding effect of age was not controlled. Cavalleri & Zuccato
(1965), however, obtained the same results taking the age factor into
account.
In 1965, a joint Italian-Yugoslav group began an investigation on
young workers in a Yugoslav viscose rayon plant. The workers were
classified according to duration of exposure to carbon disulfide. The
average ages of various groups ranged from 26 to 33 years and they
were exposed to average carbon disulfide concentrations of about
200-500 mg/m3 with peak concentrations of up to 900 mg/m3. The
excretion of 17-ketosteroids in the urine showed a linear decrease
with the duration of exposure (Cavalleri et al., 1966a,b, 1967). The
decrease in excretion of 17-hydroxycorticosteroids was most marked in
workers with short exposure and did not decrease any more as exposure
continued. The excretion of androsterone appeared to decrease
progressively with duration of exposure, probably in a linear fashion;
that of etiocholanolone did not show any uniform pattern (Cavalleri et
al., 1966c,d). Urinary excretion of testosterone and gonadotropin
luteinizing hormone was also reduced in workers exposed to
concentrations of 100-400 mg/m3 for 2-12 yearsa. Lancranjan et al.
(1971) reported a decrease in 17-ketosteroid and
17-hydroxycorticosteroid excretion in the urine of exposed workers.
The thyroid function has been studied in exposed workers by
determination of serum thyroxine (Cavalleri et al., 1971; Cavalleri,
1975; Massoud et al., in press). Decreases reported in thyroxine
levels suggest a reduction of thyroid activity.
Mild hypothyroidism was reported in 8% of exposed workers in
studies by Lancranjan (1972). This effect may be primary or may be due
to inhibition of the hypothalamus-hypophysis axis. A decrease in
thyrotropin-stimulating hormone, observed in human subjects given
tetraethylthiuram, favours the hypothesis of an impairment of the
hypothalamic-hypophyseal system (Cavalleri et al., 1977).
a Exposure data submitted by Professor Cavalleri as private
communication to the Task Group.
When the thyroid function is inhibited, disturbances in the lipid
metabolism will appear. A well-known manifestation of subclinical
hypothyreosis is elevation of the serum cholesterol level, which in
turn may contribute to the vascular changes typical of carbon
disulfide poisoning. The isolation and identification of two
metabolites, thiourea and mercaptothiazoline, may help to explain the
mechanism (Pergal et al., 1972a,b). Neither of these metabolites is
directly toxic, but both may have some influence on thyroid activity.
In fact, drugs containing substances of the thiourea and
mercaptothiazolinone type are used in the treatment of
hyperthyroidism.
Lancranjan et al. (1969, 1972), reported hypospermia,
asthenospermia, and tetratospermia in the spermatic liquid of young
exposed workers, confirming the gonadal injury reported by Cavalleri
et al. (1965).
Disturbances were found in the ovarian function of 500 young
females, exposed to concentrations of carbon disulfide of around
20 mg/m3, and of 209 women exposed to a concentration of less than
10 mg/m3 (Vasiljeva, 1973). In a comparison with a group of 429
unexposed women, menstrual flow durations of more than 5 days occurred
in 18% of the first group and in 11% of the second compared with 5% in
the control group. Irregular menstruation occurred in 8% of those
exposed to 20 mg/m3 as opposed to 2% in the comparison group. Other
menstrual disorders followed a similar pattern, and were correlated
with the length of exposure. When vaginal smears were examined from
subsamples, cellular disturbances were most frequent in the group with
highest exposure. A biochemical study of the sex hormones in the urine
confirmed the vaginal-smear findings. Hence, even rather low exposure
to carbon disulfide appeared to induce hormonal and functional
disturbances in young women. Other studies corroborate these findings
(Petrun, 1967, Finkova et al., 1973).
In a study of 380 women exposed to around 30 mg/m3, Petrov
(1969) found more pregnancy complications than in a control group of
191 women. Spontaneous abortions occurred in 14% of the exposed, and
7% of the unexposed women. "Threatened pregnancy terminations" also
occurred more frequently in the exposed women than in the controls
(26% and 13% respectively). Exposed women gave birth prematurely
significantly more frequently than controls (9% and 3%, respectively).
It has been reported in several studies that carbon disulfide
produces primary damage at the hypothalamic-hypophyseal level
(Cavalleri et al., 1965; Lancranjan et al., 1971; Maugeri et al.,
1971; Cavalleri, 1972). The hypothalamus produces the releasing
factors that stimulate the hypophysis to synthesize and release
polypeptide hormones, which in turn stimulate the target glands in
question. The impairments found would, therefore, represent a
secondary consequence.
Thus, carbon disulfide causes several disturbances of the
endocrine systems that can be summarized as follows:
-- a reduction of adrenal activity that can be repaired by ACTH
administration and can therefore be ascribed to reduced secretion of
corticotrophin;
-- a reduction of the endocrine activity of the testis and
impairment in spermatogenesis possibly due to direct gonadal damage;
this finding could explain the decrease in "libido" and "potentiae"
often found in exposed workers;
-- disturbances in the hormonal balance in women, resulting in
menstrual irregularities, spontaneous abortions, and premature births;
-- impairment of thyroid function, which could be primary or due
to a deficiency in the thyrotropin-stimulating hormone (TSH) or the
TSH releasing factor or both.
6.3.10 Effects on the nervous system
6.3.10.1 Central nervous system
In experimental animals, carbon disulfide causes destruction of
the myelin sheet and axonal changes in both the central and peripheral
neurons. Degenerative changes have been observed in the cortex, basal
ganglia, the thalamus, the brain stem, and the spinal cord (Wiley et
al., 1936; Ferraro et al., 1941; Lewey et al., 1941; Fischer &
Michalova, 1956; Drogicina, 1968).
The mechanism by which carbon disulfide causes these changes has
not been elucidated, however. Mitochondrial enzymes may be inhibited
(Tarkowski & Wrofiska-Nofer, 1966; Tarkowski & Sobczak, 1971), and the
tyrosine and catecholamine metabolism may be disturbed (Magos et al.,
1974). The content of free glutamine increases as a result of carbon
disulfide exposure (Efremova & Uzbekov, 1968; Tarkowski & Cremer,
1972). Such changes were observed with acute experimental poisoning,
but chronic, repeated exposure did not produce gross changes
(Tarkowski, 1973).
Horvath & Mikiska (1957) followed the electroencephalographic
changes in rabbits exposed to carbon disulfide and found a transitory
decrease in the amplitude of basic and superimposed beta rhythms.
The results of animal experiments have to be extrapolated to man
with extreme caution; however, many data obtained from animal studies
are in good agreement with clinical observations in man. Like other
narcotic agents, carbon disulfide produces a clinical symptomatology
of irritation and excitation on one hand, and inhibited psychomotor
activity, psychological alterations, insomnia, hypersomnia, loss of
consciousness and death, on the other.
In acute poisoning, the first typical signs from the central
nervous system (CNS) are excitation, euphoria, and aggressive
behaviour. Subacute and chronic cases are first characterized by
neurotic signs of unrest, excitation, and loss of temper. Gradually
the patient becomes depressive, anxious, paranoiac and sometimes there
is a suicidal tendency. Nightmares, apathy, loss of initiative,
vegetative disturbances, and headache are common symptoms (Melissinos
& Jacobides, 1967; Mihail et al., 1968). In the further course of
chronic intoxication, neurological signs become prominent. Both
diffuse cortical and focal symptoms from the subcortical grey matter
and extrapyramidal system are typical (Teisinger, 1934). Typical
Parkinson symptomatology with bradykinesia, bradylalia, muscle
rigidity, tremor, and increased elementary postural reflexes is
similar to that in arteriosclerotic or postencephalitic parkinsonism
(Nakamura et al., 1974). Psychic, pyramidal and extrapyramidal
symptomatology, including signs from other parts of the brain
(vestibular, cerebellar) give a picture compatible with diffuse
effects, described under the term of "toxic encephalopathy".
Furthermore, nervous involvement in general may be reflected as
changes in the sensitive branch of the trigeminal nerve, in the
function of the III, IV, and VI cranial nerves, and in the sympathetic
and parasympathic nerves of the eye.
Disturbances in cerebral circulation probably explain some of the
manifestations of neurotoxicity. Vigliani et al. (1944) demonstrated
hyalinosis of the arterioles and precapillaries histologically, a
finding that fits into the picture of general atherosclerosis caused
by carbon disulfide. Thus, the vascular changes described by Vigliani
(1961) as "encephalovasculopathia sulfocarbonica", are probably
responsible for many of the manifestations of central nervous system
pathology.
On the other hand, authors of some recent publications have tried
to explain some mechanisms by direct interference of carbon disulfide
and its metabolites in the system of enzymatic processes in the brain.
Morphological studies indicate that long axons in the spinal cord are
preferentially destroyed in chronic carbon disulfide poisoning
(Szendzikowski et al., 1973).
The typical neuropathological picture includes an increase in
neurofilaments, and secondary effects on the myeline sheath (Juntunen
et al., 1974). Biochemical studies have shown that oxidative
desulfuration also take place in the brain (Savolainen et al., 1977b),
and that the highest specific binding of the liberated sulfur is
detected in spinal cord axons (Savolainen & Vainio, 1976). It appears
that most of the liberated sulfur binds to neurofilaments that are
essential to normal axon functions (Savolainen et al., 1977c). Thus,
there is evidence of a direct toxic action upon the nervous system.
The results reported by Abramova (1967), Vasak & Kopecky (1967),
and others (section 5.3.1) regarding inhibition of the vitamin B6
metabolism, might explain some changes in the peripheral myelin sheet
and even some effects on the central nervous system. The consequences
of vitamin B6 inhibition, or interference with the transamination of
glutamic acid and inactivation of pyridoxamine could, perhaps, produce
irritative cortical symptomatology, including epileptiform cramps.
Furthermore, studies showing interactions between carbon disulfide
and, tissue respiration; depression of cytochrome oxidase, succinic
dehydrogenase, alkaline phosphatase, and dopamine-ß-hydroxylase; and
influence of inhibitors of serum elastase (EC 3.4.21.11), suggest that
besides a purely vascular etiology, it is plausible to consider a
direct toxic interference of carbon disulfide with tissue metabolism
(section 5.2). In fact, direct toxic effects of carbon disulfide on
peripheral nerve cells (Szendzikowski et al., 1973), and on the
myoneural junctions (Juntunen et al., 1977) have been shown in
animals. The central nervous system may react in the same manner.
From all these considerations, it can be concluded that the
effects of carbon disulfide on the nervous system may be influenced by
many factors. The method most often used for the study of cerebral
involvement in man is electroencephalography (EEG). However, studies
on carbon disulfide poisoning are rare. Early studies emphasize the
frequent occurrence of very flat records among subjects chronically
exposed to carbon disulfide (Krolikowska & Rucinska, 1959). More
recently, Seppäläinen & Tolonen (1974) found 21 abnormal EEGs in an
exposed group comprising 54 viscose workers compared with only 6 in a
control group of 50 paper-mill workers. Thiele & Wolf (1976) found EEG
changes in 52% of a group of 139 spinning workers. The abnormalities
consisted of slight, diffuse slow-wave abnormalities, slight to
moderately severe focal slow-wave abnormalities and even spike and
wave discharges in 3 exposed men. Thus, mild brain dysfunctions were
clearly more prevalent in the exposed group than in the control. EEG
findings among 250 workers with longstanding occupational exposure to
carbon disulfide (mean exposure 11.2 years) were recently described by
Styblva (1977) and compared with the EEGs of 61 healthy controls and
47 patients suffering from cerebral arteriosclerosis. Increased
frequency of abnormal EEGs was found in 33.2% of the exposed workers,
compared with 6.6% in the control group ( P < 0.01). The most
frequent abnormalities among the exposed workers were episodic theta
activity (33%) and diffuse abnormality (30%) (it was not, however,
indicated or even deducible from the data presented, how these
percentages had been calculated, i.e., from which population they were
taken). Local abnormalities were rare. Local EEG abnormalities
predominated in patients with cerebrovascular disease. The episodic
activity in the EEG was considered to reflect the direct toxic effects
of carbon disulfide on mesodiencephalic structures. The author
proposed that diffuse abnormality could be a manifestation of
vasculoencephalopathy resulting from toxic changes in the small
diameter vessels. The frequency of alpha activity tended to be lower
among exposed workers and this was considered to indicate a cerebral
metabolic disorder due to carbon disulfide exposure.
Studies on the behavioural and psychological effects of carbon
disulfide exposure have indicated disturbances in deep subcortical
structures, namely vegetative centres, and more diffuse cortical
involvement. The methods used have included diagnostic interviews,
behavioural observations (mainly on experimental animals), and
psychological test batteries.
Both structured and unstructured interviews of human subjects
showed differences in the clinical pictures of carbon disulfide
poisoning at rather high concentrations i.e., about 950-2400 mg/m3
(300-760 ppm) and at much lower concentrations i.e., around
20-50 mg/m3 (7-30 ppm) over a long period of exposure. At high
concentrations, symptoms of headache, muscle pain, fatigue,
paraesthesia, and general weakness were consistently reported (Gordy &
Trumper, 1940; Manu et al., 1970). At lower levels of exposure, fewer
complaints of muscular pain and overall weakness were noted, the main
symptoms being headache, memory impairment, rapid mood changes,
insomnia, paraesthesia, and fatigue (Seppäläinen et al., 1972; Lilis,
1974).
Standardized batteries of tests yield results that are directly
quantifiable and comparisons can be made between groups at different
exposure levels and control groups. Using this method, Hänninen (1971)
presented 3 groups of workers with different exposures and clinical
profiles (unexposed, exposed without clinical symptoms, and poisoned).
The results suggested that syndromes of latent and manifest carbon
disulfide poisoning differ in quality as well as in intensity, i.e.,
"clinically manifested poisoning is characterized by lowered
vigilance, diminished intellectual activity, diminished rational
control, retarded speed, and motor disturbances, whereas traits
indicative of depressive mood, slight motor disturbances, and
intellectual impairment are characteristic of latent poisoning"
(Hänninen, 1971). Analysis of intercorrelations among test sources for
each of the 3 groups resulted in substantially different factor
structures across the 3 groups. This supports the contention that
behavioural differences between groups exposed to different
concentrations are qualitative as well as quantitative (Hänninen,
1974). Naturally, the extent to which such distinctions are, in fact,
true, depends on the number of false-positive diagnoses in the
poisoned group (i.e., how many patients with other diseases had been
classified as cases of carbon disulfide poisoning), and, since this
aspect could not be evaluated, it may be premature to accept the
qualitative changes found as real facts. As to the quantitative
changes, it is self-evident that their severity must be correlated
with the degree of poisoning, at least on a group basis. The
specificity of the psychological and behavioural examination increases
when other sub-clinical signs of carbon disulfide exposure, such as
EEG and EMG findings are present.
Hänninen & Mäenpää, as reviewed by Tolonen (1974), applied the
same type of tests to 97 exposed male viscose rayon workers (mean
exposure time 15 years) and 96 age-and-sex matched controls from a
paper mill with no history of exposure to carbon disulfide. The most
distinct difference was in the retardation of psychomotor speed, where
the relative risk was 2.2 ( P = 0.02). As a whole, 40% of the exposed
and 25% of the control group showed "poor" performance, as defined
according to criteria published by Tolonen (1974). These results show
a clear group difference; however, the test battery has a low
specificity for carbon disulfide, as could be seen from the high
number of "false positive" results.
Recently, Hänninen and her co-workers increased the specificity
of the test battery, reducing the number of tests on the basis of
information gathered from new analytical approaches applied to results
of 206 exposed and 152 unexposed men, examined in 1972. According to a
multiple discriminant analysis, the most specific variables for carbon
disulfide effects were those indicating retarded speed, retarded
emotionality and energy, and psychomotor disturbances (Hänninen et
al., 1978). Another approach, starting from test results classified as
abnormal, resulted in similar conclusions (Tolonen et al., 1976). Now,
the test battery can be restricted to a few rather specific tests
applicable to suspected cases of poisoning that can also be used at
periodic health examinations.
Analogous results concerning the depressing effects of carbon
disulfide on psychological performance and behaviour have been
reported by Foa et al. (1976), Gherase (1976), Schneider (1976),
Tuttle et al. (1976)a and Hakky (1977).
Foa et al. (1976) demonstrated an exposure-effect relationship by
comparing workers from 2 plants with different degrees of exposure. A
relationship between exposure time and performance was demonstrated by
Schneider (1976), but the role of age as a confounding factor could be
excluded for 2 test variables only.
a Tuttle, T. C., Wood, G. D., & Grether, C. B. (1976) Behavioral and
neurological evaluation of workers exposed to carbon disulfide
(CS2). Unpublished report submitted to NIOSH (National Institute for
Occupational Safety and Health) by Westinghouse Electric Corporation,
Behavioral Services Center, Columbia, MD, USA, 156 pp.
From these data, it can be concluded that, at rather low exposure
levels, psychological test results are almost the only way of
assessing involvement of the central nervous system. Relationships may
exist between psychological impairment and EEG abnormalities, but
further studies are needed to clarify this point.
6.3.10.2 Peripheral nervous system
Many authors have studied the neuropathy and myelopathy of
animals exposed to carbon disulfide (Fischer & Michalova, 1956;
Higashida et al., 1959; Scheel, 1967; Frantik, 1970; Luke,, 1973;
Szendzikowski et al., 1973; Wrofiska-Nofer et al., 1973; Seppäläinen &
Linnoila, 1975, 1976). Destruction of the myelin sheet has been the
most prominent finding, but axonal changes have also been observed. In
the muscle fibres, atrophy of the denervation type occurred secondary
to the polyneuropathy. Some species differences in susceptibility to
carbon disulfide have been found. Cats and rabbits tolerated only low
concentrations of carbon disulfide. On the other hand, polyneuropathy
in rats was produced only with exposure to extremely high
concentrations (1200-2400 mg/m3). The slowing of nerve conduction
velocity in the sciatic nerve in rabbits and rats preceded clinical
symptoms such as clumsiness and later on, paresis. In rabbits,
electromyographic abnormalities suggested the presence of myelopathy
together with peripheral neuropathy (Seppäläinen & Linnoila, 1975,
1976). Neuropathological studies (Linnoila et al., 1975) showed severe
morphological alterations in the lateral and anterior funiculi of the
spinal cord in addition to pronounced axonal alterations in the
peripheral nerves of rabbits.
Lukas (1970, 1973) established by electromyography (EMG) that the
development of neuropathy in the rat was influenced by dose, duration
of exposure, physical conditions, and the nutritional state of the
animal. This confirms the long-standing clinical belief that the
individual constitution bears some relationship to the quantity and
quality of the effects of carbon disulfide (Djuric, 1967, 1971; Djuric
et al., 1973; Goto et al., 1971).
The protective effects of some substances have also been studied
in animals. The addition of zinc and copper salts (Scheel, 1967;
Lukas, 1970), and of pyridoxine to the diet (Frantik, 1970; Luke,,
1970) did not change the toxic effects of carbon disulfide but
improved the biochemical consequences of the intoxication, for example
by decreasing the excretion of xanthurenic acid with urine (Kujalova &
Tintera, 1970). Addition of nicotinic acid (Wrofiska-Nofer, 1970)
provided some protection against the effects of carbon disulfide lipid
metabolism. The clinical picture of carbon disulfide intoxication in
man is mainly based on toxic polyneuropathy (Bashore et al., 1938;
Vigiliana, 1954, 1961; Lukas, 1969; Lukas & Hromadka, 1977; Milkov et
al., 1970; Seppäläinen et al., 1972; Seppäläinen & Tolonen, 1974;
Seppäläinen, 1977; Gilioli et al., 1977). The subjective complaints
include paraesthesia, dysaesthesia, and fatiguability and diffuse
pains of the lower extremities. Sometimes irritative signs are
prevalent and sometimes the patients have a feeling of local
hyperaesthesia, or hypersensitivity on palpation in the muscles of the
lower limbs.
Distribution of the complaints is mostly symmetrical, and,
objectively, there is hyporeflexia. Klimkova-Deutschova (1965) pointed
out the characteristic dissociation of the patellar reflex response,
which is normal or increased, with a decreased or absent Achilles
tendon reflex. As the neuropathy develops, changes first appear in the
sensitive fibres of the lower extremities, later reaching the upper
extremities. (Lukas, 1969; Lukas & Hromadka, 1977). Hyperaesthesia is
mostly of a "sock" character, but sometimes it is also diffuse.
Palpation of the nerve roots and sometimes of the whole muscle groups
is painful. Today, the development of symptoms seldom reaches the
stage of motor paresis and, consequently, atrophic changes
(amyotrophy) are rare. The same is true of vasomotoric changes.
Amyotrophy has been described in the earlier literature in the area of
the triceps surae and quadriceps muscles of the legs, as well as in
the small muscles of the hand, the thenat, the hypothenar, and in the
interosseal muscles.
It has also been suggested that cells in the spinal cord are
injured (Manu et al., 1970; Seppäläinen et al., 1972). On the other
hand, disturbances in myoneural function have resulted in
myastheniform signs (Seppäläinen et al., 1972).
Needle electromyography and measurement of the motor conduction
velocities of peripheral nerves have proved to be sensitive methods
for detecting early nerve damage, even in neurologically normal,
exposed workers (Lukas, 1969; Manu et al., 1970; Seppäläinen et al.,
1972; Lilis, 1974; Seppäläinen & Tolonen, 1974; Knave et al., 1974;
Thiele & Wolf 1976; Lukas et al., 1977). The EMG abnormality most
often found consists of a reduced number of motor units at maximal
contraction and an increase in the duration of motor unit potentials
and fibrillation. The maximum motor conduction velocities (MCV) are
lower, especially in the leg nerves, and the distal latencies are
prolonged. However, the most sensitive test for peripheral nervous
involvement appears to be the conduction velocity of the slower fibres
of the ulnar and deep peroneal nerves, and that of the H-reflex in the
region of tibia] nerve (Lukas, 1969, 1977; Seppäläinen & Tolonen,
1974; Gilioli et al., 1977). In a recent epidemiological study
(Seppäläinen & Tolonen, 1974) comprising 118 male viscose rayon
workers and 100 control subjects from a paper mill, the most important
findings were as follows: the conduction velocities in the nerves of
the exposed men were generally slower than those of the controls. The
most distinct differences (all P values smaller than 0.01) were
obtained for the conduction velocity of the slower fibres of the ulnar
and deep peroneal nerves, the MCV of the deep peroneal and posterior
tibial nerves, and the motor distal latency of both the median and
ulnar nerves. The exposed group in this study was predominantly
composed of clinically "well" men, exposed for a number of years
(median 15) to carbon disulfide plus hydrogen sulfide concentrationsa
of about 60-190 mg/m3 (20-60 ppm) in the 1950s, about 30-95 mg/m3
(10-30 ppm) in the 1960s, and mainly below 60 mg/m3 (20 ppm) during
the last years. A little more than half of the men were still exposed
at the time of examination, the rest were working in "clean"
departments.
Different types of nervous system symptoms and findings among 51
Swedish viscose factory workers were evaluated by Knave et al. (1974).
The iodine-azide test was used as a measure of exposure intensity. The
comparison group consisted of 51 subjects matched for sex and age.
Symptoms of peripheral nervous dysfunction (e.g., restless legs,
cramps, pain, and numbness) were over-represented among workers,
especially the combination of these symptoms. Only one neurological
sign-namely tremor-showed a higher prevalence in the exposed than in
the control group. The authors measured only the maximal MCV of the
median nerve and found minor slowing in this MCV when the duration of
employment was between 5 and 30 years, but not after shorter exposure.
The prognosis of carbon disulfide polyneuropathy has not been
considered to be too poor. According to one follow-up study of exposed
workers, electroneuromyographic abnormalities tend to ameliorate if
the exposure level decreases or exposure is stopped (Seppäläinen,
1977). A tendency for repair and restitution of lost functions has
also been demonstrated experimentally in animals (Lukas, 1970;
Seppäläinen & Linnoila, 1975, 1976).
However, it has been shown that, even after periods of 10-15
years without exposure, abnormal conduction velocity values may be
found in workers with previous carbon disulfide poisoning or previous
long-term exposure to carbon disulfide (Seppäläinen et al., 1972).
This corresponds with the experience of Gilioli et al. (1977), who
found the reduction in conduction velocity to be irreversible. In
another study, Seppäläinen (1977) described an amelioration in the EMG
findings when carbon disulfide exposure was reduced or stopped.
a Exposure levels in studies by Hernberg et al. (1970, 1973, 1976),
Raitta et al. (1974), Tolonen (1974), Tolonen & Seppäläinen (1974),
and Tolonen et al. (1975) refer to combined concentrations of carbon
disulfide and hydrogen sulfide. Hydrogen sulfide concentrations were
about 10% of the carbon disulfide concentrations (Hernberg et al.,
1970).
6.3.11 Cardiovascular effects
A number of studies have shown that carbon disulfide causes
vascular changes in various organs of experimentally exposed animals
(Lewey, 1941; Guarino & Arcello, 1954; Paterni et al., 1958). In man,
evidence of vascular changes caused by long-term exposure, has been
accumulating since World War II. Attinger (1948) was the first to
describe such changes in 5 autopsy cases. Further clinical and autopsy
studies showed clearly that carbon disulfide was a vasotropic poison
(Uehlinger, 1952; Attinger, 1954; Vigliani & Pernis, 1955; Crepet et
al., 1956; Rechenberg, 1957; Maugeri et al., 1966a,b; Prerovska &
Roth, 1968; Maugeri et al., 1971; Prerovska & Zvolsky, 1973).
According to Vigliani & Pernis (1955) the "vasculopathia
sulfocarbonica" appears in 2 forms, namely in cerebral arteries
causing encephalopathy, and in the renal arteries causing nephropathy
and hypertension. The syndromes that follow these types of vascular
damage have already been described in sections 6.2, 6.3.7, and
6.3.10.1. Autopsy studies have revealed that sclerosis is prevalent in
large and middle-size arteries and hyalinization in arterioles and
capillaries. These changes appear in the blood vessels of all organs
but they are particularly prominent in the central nervous system and
kidneys.
Vascular changes due to carbon disulfide exposure are similar to
those produced with atherosclerosis due to age. Thus, the picture in
older persons is confused by the influence of age. The carbon
disulfide induced vasculopathy is therefore best studied in young
persons with a sufficiently long exposure time. With this in mind, a
joint Italian/Yugoslav team selected 104 young workers (age around
30-35 years) with more than 10 years' exposure for closer study
(Maugeri, et al., 1966a,b; Maugeri et al., 1971; Taccola et al.,
1971). Of these, 28 were invalids due to carbon disulfide poisoning.
Organic changes in blood vessels were discovered, mostly in the
invalids, by means of cerebral rheography. The results obtained
suggested progression of such changes even after the termination of
exposure. Using peripheral rheography, plethysmography, and
oscillography, the authors established functional vasoconstriction in
the arteries of the upper and lower extremities. Organic alterations
of the sclerotic type were rare and mainly appeared after prolonged
exposure and in the lower extremities.
During the last 10 years or so, it has become increasingly
evident that long-standing exposure to carbon disulfide promotes
coronary heart disease, even under circumstances where clinical
poisoning is uncommon. Of the several attempts made to connect carbon
disulfide exposure with coronary heart disease, only a few have
complied with necessary methodological requirements. The first such
investigation was a careful mortality study in Britain published by
Tiller et al. (1968). They found a 2.5-fold excess mortality in
viscose rayon workers exposed to carbon disulfide for 10 years or
more, compared with that of other workers. The same study demonstrated
that, in the 3 plants studied, coronary mortality was proportionally
higher among workers engaged in the viscose spinning process than in
other workers, men living in the locality, and the national statistics
derived from the Registrar General's tables. Similar results have been
obtained from Norway, where a 3-fold excess mortality from coronary
heart disease was found among workers, aged 35-54 years, compared with
unexposed workers in other departments (Mowé, 1971).
A prospective study that was initiated in 1967 in Finland, showed
that the 5-year mortality in a cohort of 343 male viscose rayon
workers exposed to about 30-95 mg/m3 (10-30 ppm) was almost 5-fold
(14 deaths compared with 3) that of an unexposed comparison cohort
from a nearby paper mill. Other causes of death were evenly
distributed (Hernberg et al., 1970, 1973). The incidence of nonfatal
first myocardial infarctions was almost 3 times that in the comparison
cohort (11 compared with 4) (Tolonen et al., 1975). In a continuation
of the follow-up extending to 8 years, the cumulative incidence rate
for mortality from coronary heart disease was 5.8% in the exposed
group and 2.6% in the comparison group, giving a rate difference of
3.2% (Hernberg et al., 1976; Nurminen, 1976). Between the sixth and
ninth years of follow-up, no further excessive mortality was observed,
probably because most (81%) of the originally exposed workers were no
longer exposed to carbon disulfide and also overall concentrations of
carbon disulfide had decreased to less than 30 mg/m3 (10 ppm). This
positive trend was promising with regard to coronary heart disease:
the prognosis of exposed workers improved with improved occupational
hygienic measures and with a reduction in the length of exposure over
a lifetime.
Anamnestic angina was more prevalent both in 1967-68, and at
re-examination in 1972, whereas there were no great differences in
coronary electrocardiographic findings (Hemberg et al., 1970; Tolonen
et al., 1975) as coded by the "Minnesota Code" (Blackburn et al.,
1960). These results showed that the greatest effect of carbon
disulfide on coronary arteries was on the production of fatal
infarctions (relative risk 4.8) followed in reducing order by all
infarctions, fatal and nonfatal (relative risk 3.7), nonfatal
infarctions (relative risk 2.8), angina (relative risk 2.2), and
coronary ECGs (relative risk 1.4). In other words, the more serious
the outcome, the greater the relative risk. The interpretations may be
that exposure to carbon disulfide not only worsens the prognosis of
existing coronary heart disease but increases the incidence of new
cases. However, it is not certain that this mode of action operates in
countries where coronary heart disease is less common than in Finland.
Coronary heart disease has a multifactorial etiology, and the pattern
described above may be noticeable only when a sufficient number of
other coronary risk factors are present at the same time. In fact,
some other studies have shown a more distinct excess of coronary ECGs
(Goto & Hotta, 1967; Cirla et al., 1972). It is possible that both
dose-dependent, methodological, and even geographical factors may be
responsible for the discrepancies reported so far. For example, it may
be that the pattern of excess coronary heart disease differs in
different populations, depending on the concomitant prevalence of
other coronary risk factors such as elevated blood lipids and
hypertension.
A joint analysis of the prevalence of angina pectoris, exercise
ECGs, and blood pressure measurements in Finnish and Japanese workers
exposed to carbon disulfide and in 2 unexposed control groups
supported this view (Tolonen et al., 1976). Angina was rare and
evidence of past infarction nonexistent in both exposed and unexposed
Japanese men; in the Finnish exposed and unexposed groups the
prevalence of angina was 15 and 10%, respectively. The prevalence of
coronary ECGs was almost equal among the exposed and unexposed men in
both the Japanese and the Finnish groups. Angina plus coronary ECGs as
evidence for coronary heart disease occurred with a prevalence of nil
for both exposed and unexposed Japanese, and 5 and 2% for the exposed
and unexposed Finnish groups, respectively. The study did not yield
any evidence of an excess in any of the variables examined among the
exposed Japanese workers. However, a follow-up study of this group is
necessary to shed more light on the possibility of excess morbidity
and mortality from coronary heart disease. Vertin (1977) did not find
any significant differences in coronary heart diseases, ECG changes,
serum cholesterol concentrations, and arterial pressure in workers
exposed to carbon disulfide concentrations of about 30-95 mg/m3
(10-30 ppm) compared with a control group.
Carbon disulfide vasculopathy may be due to the combined action
of several biochemical and physiological disturbances, namely:
(a) changes in lipid metabolism (section 6.3.9);
(b) disturbances in the coagulation mechanism (section 6.3.14);
(c) elevation of blood pressure (Hemberg et al., 1970; Sakurai,
1972; Tolonen et al., 1975);
(d) subclinical hypothyroidism (Cavalleri et al., 1971);
(e) a toxic effect on the myocardium, either direct or through
interference with the catecholamine metabolism (Magos, 1972).
However, much work remains to elucidate the mechanism of carbon
disulfide-induced atherosclerosis. In particular, coronary heart
disease has a multicausal origin and is closely related to the
saturated fat intake of the population. Furthermore, its incidence is
influenced by a large number of other risk factors, such as smoking,
diabetes, and physical inactivity. A combination of 2 or more risk
factors greatly increases the incidence. Thus, it may be postulated
that carbon disulfide will only become evident as a coronary risk
factor in the presence of other risk factors.
6.3.12 Carcinogenicity and mutagenicity
No reports are available that indicate any carcinogenic or
mutagenic effects of carbon disulfide.
6.3.13 Teratogenic effects
Only a weak teratogenic effect appeared in rats following
low-level exposure to a combination of hydrogen sulfide and carbon
disulfide (Bariljak et al., 1975).
A marked impairment (increase in early embreonal mortality,
reduction in fetal weight, malformations in the brain and limbs,
behavioural deviation) in the prenatal development of 2 successive
generations was produced in rats exposed to carbon disulfide
concentrations of 100 and 200 mg/m3 throughout the period of
gestation (Tabacova et al, 1977).
6.3.14 Other effects
The literature is contradictory concerning the effects of carbon
disulfide on blood coagulation. Saita & Gattoni (1957) found a slight
decrease in factor VII and prothrombin in exposed workers. On the
other hand, Moreo & Candura (1960) and De Nicola et al. (1962)
reported hypercoagulability in the blood of experimental animals, a
finding that was later confirmed in studies of exposed workers by
Candura et al. (1962), Saita et al. (1964), and Danilova (1968). An
increase in fibrinolysis was found by De Nicola et al. (1962) in
animals suffering from acute poisoning. In contrast, Candura et al.
(1962) found decreased fibrinolysis in workers exposed to carbon
disulfide.
The coagulation mechanism in highly-exposed young workers in a
viscose factory was studied by Visconti et al. (1966a,b,c, 1967). They
found protracted coagulation, retarded production of active
thrombotastin, decreases in plasminogen and in the activity of
plasmin, and a decrease in antiheparinic activity in workers exposed
for up to 9 years. These disturbances were most pronounced during the
first years of exposure.
Visconti et al. (1966a) supposed that carbon disulfide could
affect blood coagulation either directly by interfering with the
system itself, or indirectly, by liver injury. Different routes of
entry, differences in the level and duration of exposure and in the
distribution of carbon disulfide in the organism, and also
interspecies differences are probably responsible for the confusing
results.
It has been reported that the glucose metabolism is disturbed by
carbon disulfide both in experimental animals (rabbits) (Hara, 1958)
and in exposed workers (Goto et al., 1971). There are also a few
clinical studies reporting an increased occurrence of diabetes in
patients suffering from severe carbon disulfide poisoning (Austoni &
d'Agnolo, 1957; Finulli & Ghislandi, 1959; Ferrero, 1969). Lack of
proper control groups renders the interpretation of these results
difficult.
6.3.15 Interactions with other chemical compounds
The combined effects of carbon disulfide with other chemical
compounds were first reported by Lazarev et al. (1965) who indicated
that repeated exposure to a concentration of carbon disulfide of
3900 mg/m3, for 2 h per day, over a period of 20-30 days considerably
prolonged both the hexobarbital sleeping time and alcohol retention
time in rats and mice (see also section 6.3.6).
Carbon disulfide caused reversible inhibition of the oxidative
drug metabolism in rat liver endoplasmic reticulum (Bond et al., 1969;
Freundt & Dreher, 1969; Freundt & Kuttner, 1969; Freundt & Henschler,
1971; Freundt, 1973; Sokal, 1973; Freundt et al., 1974b, 1975, 1976).
This effect was also produced in man after exposure to a carbon
disulfide concentration of about 30 mg/m3 (10 ppm) over a 6-h period
(Mack et al., 1974); the treatment led to a rapidly reversible but
significant decrease in the oxidative N-demethylation of the
analgesic, aminopyrine.
Simultaneous exposure to carbon disulfide and ethanol resulted in
an increase in blood acetaldehyde in rats and man that was considered
to be a consequence of the inhibition of aldehyde dehydrogenase by
carbon disulfide (Freundt & Netz, 1973; Freundt & Lieberwirth, 1974;
Freundt, 1974; Freundt et al., 1976). In 12 healthy male volunteers, a
50% increase in blood acetaldehyde was noted following exposure to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) for 8 h
with simultaneous ingestion of ethanol in orange juice at a
concentration that maintained the blood ethanol level at about 0.8%.
However, no signs of an "antabuse syndrome" were observed under these
experimental conditions (Freundt et al., 1976).
6.4 Diagnosis
Since all isolated effects are nonspecific, individual diagnosis
becomes a matter of probability based on: (a) the ascertainment of
exposure; (b) the demonstration of signs and symptoms of poisoning
and the combination in which they occur; and (c) the exclusion of
other diseases.
The changes due to carbon disulfide exposure described in
sections 6.2-6.3.15 affect so many different organ-systems that, in
considering the etiology of a single case, the probability of it being
caused by carbon disulfide increases in proportion to the number of
these signs and symptoms present. Although, in 1941, Lewey drew
attention to the importance of the concomitant occurrence of different
neuropsychiatric and vascular effects, as well as to their connection
with the severity of the disease, there have been few, if any, studies
systematically analysing the knowledge that has emerged since then.
For this reason it is difficult to find quantitative data. However,
carbon disulfide poisoning should always be suspected when a viscose
worker presents with subjective, neurasthenic symptoms, signs of
peripheral neuropathy, psychological disturbances, and vascular
changes in the cerebral, coronary, renal, or peripheral systems. When
exposure to toxic levels of carbon disulfide occurs, the following
chronological effects can usually be expected: the appearance of
neurasthenic symptoms accompanied by psychological behavioural
changes; signs of peripheral nervous system involvement as shown by
electromyographic changes, initially mainly in the afferent fibres but
later in the motor fibres; simultaneous disturbances in the central
nervous system may appear but may be more difficult to detect
objectively than the lesions of the peripheral nervous system. Another
early finding is a decrease in the level of serum thyroxine that is
probably due to damage of the hypothalamic-hypophyseal axis (section
6.3.9). Cardiovascular damage appears after longer periods of
exposure. The appearance and severity of the above-mentioned signs and
symptoms are related to the level of the uptake and to the duration of
exposure to carbon disulfide. The changes are also dependent on the
individual constitution and health status of exposed workers.
In the differential diagnosis of true cases, atherosclerosis due
to other causes should be considered including, postencephalitic or
atherosclerotic Parkinsonism, brain tumours, syphilis, multiple
sclerosis, and psychiatric diseases of other origins such as endogenic
depression, schizophrenia, and alcoholism. Peripheral polyneuropathy
should be distinguished from that due to alcoholism, diabetes, and
other toxic agents.
The subclinical stage is, by definition, more difficult to
identify. First, the presence of pathological symptoms and signs must
be established, which requires sensitive techniques. Second, the
causal connection between an established symptom or sign and carbon
disulfide exposure should be shown. Both aspects pose great
difficulties because of the nonspecificity of the manifestations and
their vagueness in the initial stages. So far, the only attempt to
approach this problem in a systematic way has been made by Tolonen
(1974).
He studied 97 men exposed to carbon disulfide in a viscose
factory and 96 controls of the same age from a paper mill. The mean
age was 48 years (range 33-67 years), and the mean exposure time was
15 years (range 1-27 years). The intensity of background exposure
ranged from about 60 to 125 mg/m3 (20 to 40 ppm) in the 1950s and
then from about 30-95 mg/m3 (10-30 ppm). Personal exposure data were
not available. The group comprised both completely "healthy" workers
as well as some partly incapacitated patients with past poisoning.
About half of the men were still exposed to carbon disulfide, the rest
being employed in "clean" departments. The groups underwent many
examinations including: (a) examination of the heart (positive =
history of verified myocardial infarction, and/or "Minnesota Codes"
I1-3, IV1-3, V 1-3, VIII3, VIII3 , XI1-3-5-7, and/or typical angina):
(b) psychological testing (section 6.3.10.1); (c) measurement of
the conduction velocities of 8 peripheral nerves (polyneuropathy was
considered to exist when 2 or more nerves showed reduced conduction
velocities) and (d) examination of the circulation of the ocular
fundus (the criterion for disturbed circulation was delayed
peripapillary filling -- circumferential, segmental, or both). The
occurrence of isolated and combined signs is shown in Table 2.
As many as 59% of the exposed and 29% of the unexposed men were
affected by more than one disorder under study but most combinations
of disorders occurred more frequently in the exposed group; a
combination of 3 abnormalities was 3 times more common in the exposed
group than in the controls and a combination of all 4 abnormalities
occurred only in the exposed group. Only 5% of the exposed subjects
compared with 31% of the controls were without any abnormalities.
Since disturbances in the choroidal circulation were present in all
cases of excess "morbidity" (68% of the exposed, Raitta et al., 1974),
it seems that this abnormality represents the earliest manifestation
of carbon disulfide toxicity, at least, of those considered here.
Thus, it can be postulated that a positive diagnosis of carbon
disulfide poisoning can only be made if changes in the choroidal
circulation have been observed and providing that exposure has
extended over 10 years or more, with an intensity of the magnitude of
about 30-90 mg/m3 (10-30 ppm). The estimated probability that a
combination of findings is of occupational origin is the excess
morbidity over total morbidity. The results also showed that the
etiological role of carbon disulfide could be demonstrated with
greater probability, the greater the number of abnormalities present
at the same time.
The necessity to ascertain exposure is so obvious that it should
not need to be mentioned. It should be stressed that recent experience
has shown the importance of evaluating personal, as opposed to
background exposure, since the former may be only weakly correlated
with the latter. Personal exposure can be measured using personal
samplers, or by the iodine-azide test. The latter has been claimed to
discriminate particularly sensitive workers (whose test does not
return to normal after 16 h away from exposure) (Graovac-Leposavic et
al., 1967).
Table 2. Prevalence (%) of coronary heart disease (CHD), delayed
peripapillary circulation (EYE), polyneuropathy (PN) and
behavioural symptoms (BS), their combinations in subjects
in the exposed and control groups, and the differences
between the groups.a
Exposed Group Difference =
(N = 97) control excess morbidity
(N = 96)
Free of disease 5 31
CHD only 4 9 -5
EYE only 19 19 0
PN only 6 6 0
BS only 7 6 1
CHD + EYE 4 4 0
CHD + PN 1 1 0
CHD + BS 1 2 -1
EYE + PN 12 5 7
EYE + BS 7 4 3
PN + BS 4 5 -1
CHD + EYE + PN 9 0 9
EYE + PN + BS 11 5 6
CHD + PN + BS 2 1 1
CHD + EYE + BS 2 2 0
CHD + EYE + PN + PS 6 0 6
Total 100 100 26
a From: Tolonen (1974).
6.5 Surveillance of the Health of Exposed Workers
Considering the early manifestations of chronic carbon disulfide
effects and the factors that may influence the appearance and the
severity of such effects (section 6.4), the following examinations are
recommended for health surveillance:
(a) Clinical neurological investigation;
(b) Electroneuromyographic (ENMG) examination wherever possible
(especially the conduction velocity test, which should be carried out
on several nerves);
Additional tests that may give valuable information include:
(a) Psychological and behavioural testing with a narrow battery
of well validated tests;
(b) Serum thyroxine measurement repeated at least once a year;
(c) Blood pressure measurements;
(d) Electrocardiography (preferably an exercise ECG);
(e) Fundus photography (in some countries);
(f) Blood lipid pattern estimation, if the exposure level is
high;
(g) Electroencephalography (EEG), if there are special
indications;
These tests should be performed at the pre-employment examination
and subsequently at regular intervals to detect early deviation from
baseline values. Changes observed in the same individual over a period
of time may suggest a relationship with carbon disulfide uptake.
Depending on the intensity of exposure, the iodine-azide test
should be carried out from 2-12 times a year, both immediately after
the work shift and the next morning. Those with positive tests should
be seen by the plant physician, since a pathological test in the
morning is a warning of incipient poisoning (Graovac-Leposavic et al.,
1967). In addition to this, medical examinations should be made once
or twice a year. They should comprise a thorough history and a
neurological examination.
When suspect signs of early carbon disulfide effects appear, the
worker should be removed from exposure to a "clean" job. In the light
of the new findings concerning coronary heart disease, the removal of
workers who develop coronary risk factors, such as hypertension,
hypercholesterolaemia, and ECG changes may be indicated, irrespective
of their etiology. In some plants, there is the practice of regular
periods of work in a clean atmosphere, e.g., every sixth month, or of
extra vacations in the winter.
Some authors believe in the importance of the diet of exposed
workers. However, the protective effect of various diets and
supplements for man remains unclear. Scheel (1965) claimed that
addition of zinc and copper salts to the diet of exposed rats had a
protective effect against carbon disulfide exposures. A diet poor in
proteins, especially amino acids containing sulfur, but enriched in
vitamin B6 and glutaminic acid was suggested by Agronovskij &
Goloscapov (1972). Lukas (1973) established that rats on an "optimal"
diet were more resistant to the effects of carbon disulfide than rats
on a normal diet. In the first group, the signs of intoxication
appeared after a longer time and the effects were less marked than in
the second group.
At the present time, the only recommendation that can be given in
the case of man is that workers should be provided with a diet
containing sufficient amounts of energy foods to provide the required
joules, proteins, vitamins, and trace elements. This recommendation is
of particular importance in countries where such dietary requirements
are not necessarily fulfilled normally. However, basing the preventive
programme mainly on diets or drugs should be condemned, since the
fundamental question is that of technical improvements.
6.6 Contraindications for Exposure to Carbon Disulfide
An inborn error of metabolism manifesting itself as an abnormal
iodine-azide reaction in subjects working under conditions of high or
moderate carbon disulfide exposure (less than, 50 mg/m3) should be
considered an absolute contraindication for exposure.
Other contraindications for working in an environment containing
carbon disulfide include those common to all other toxic exposures
such as youth (below 18 years), pregnancy, concomitant chronic disease
such as psychiatric neurological, cardiac, renal, and pulmonary
diseases, and any condition that prevents the use of respirators, etc.
In addition, contraindications based on the specific toxicity of
carbon disulfide include diseases of the metabolic system, such as
liver disease and endocrine disorders. Any neurological or psychiatric
disorder is also a contraindication, including vegetative dystony and
psychic lability. A history of gastritis and peptic ulcer is a
relative contraindication, depending, for instance, on whether or not
there will be shift work and on the intensity of exposure. Recently,
attention has been focused on to what extent the presence of coronary
risk factors should be regarded as a contraindication; no strict
recommendations can be given, but as a general rule the presence of
several of these in an individual, or alternatively strongly abnormal
values for one risk factor, should be regarded as contraindications
(the strongest coronary risk factors apart from age itself are
elevated serum cholesterol or triglycerides, heavy cigarette smoking,
hypertension, and diabetes). In the evaluation of these and other
contraindications, the exposure intensity and hygienic conditions
should, of course, be taken into consideration.
The employment of women in work places with carbon disulfide
exposure is a special problem.
Considering the studies referred to in section 6.3.9, it is
advisable to exclude pregnant women from carbon disulfide exposure.
The same is true for lactating women and for those with expressed
disturbances of menstruation and habitual abortion.
7. EXPOSURE-EFFECT AND EXPOSURE-RESPONSE RELATIONSHIPS
7.1 Validity of Exposure Data
In general, valid exposure data are available only from
experimental studies on animals. Almost all epidemiological studies
have failed to document the exposure levels pertinent to the effects
studied. There is a universal lack of retrospective exposure data that
usually renders the assessment of the relationship between chronic
effects and the exposure levels responsible for their occurrence
impracticable. Furthermore, even when exposure data are available,
they often consist of occasional short-time measurements from fixed
sites that are not necessarily representative of the workers'
exposure. It is, therefore, extremely difficult, if not impossible, to
establish exposure-effect or exposure-response curves for carbon
disulfide, based on epidemiological evidence. Even the relation of
isolated findings reported in the literature, to given exposure levels
is difficult. Thus, the attempts in this document, to relate the
intensity of exposure to the intensity and frequency of effects must
be regarded with caution.
7.2 Experimental Data
7.2.1 Acute animal exposure
Acute exposure to carbon disulfide in animal experiments by
inhalation or by intramuscular or intraperitoneal injection has
provided information regarding the mechanism of carbon disulfide
toxicity, but it is difficult to apply this information to the
industrial exposure of man.
7.2.2 Long-term animal exposure
Several experiments on the inhalation of carbon disulfide have
been performed on different types of animals. In most of these
studies, exposure was to carbon disulfide only, but in a few, a
combination of carbon disulfide and hydrogen sulfide was used.
The concentrations of carbon disulfide ranged from
0.1-2330 mg/m3 and those of hydrogen sulfide, from 0.1-140 mg/m3;
the exposure periods ranged from 30 min per day to 6 h per day for up
to 15 months.
The reactions to such exposures included increased mortality,
teratogenic changes, testicular lesions, aspermatogenesis, engorged
blood vessels, weakness, paralysis, lethargy, weight changes,
metabolic changes, behavioural changes; sometimes there were no
observed effects. Some of the effects resembled lesions found in
exposed workers; others did not bear any relationship to those arising
from human exposure (Wakatsuki & Higashikawa, 1959; Goldberg et al.,
1964; Minden et al., 1967; Yaroslavski, 1969; Frantik, 1970; Gondzik,
1971; Misiakiewicz et al., 1972; Szendzikowski et al., 1973; Bariljak
et al., 1975; Seppäläinen & Linnoila, 1976; Tabacova et al, 1977). The
considerable differences in ranges of concentrations and periods of
exposure used in these experiments make conclusions regarding
exposure-effect and exposure-response curves impossible. This
regrettable situation emphasizes the need for international
coordination of toxicological experiments to make comprehensive
scientific evaluation possible.
7.3 Epidemiological Data
All studies so far reported have been from the viscose rayon
industry, where exposure to carbon disulfide occurs together with
exposure to hydrogen sulfide. However, generally, the levels of carbon
disulfide are at least one order of magnitude higher than those of
hydrogen sulfide, and most authors have attributed the health effects
found to carbon disulfide rather than to hydrogen sulfide exposure.
Only some of the various effects reviewed in section 6 can be
related to exposure levels. The following section contains a brief
review of exposure-effect and exposure-response data for neurological,
cardiovascular, ocular, and gonadal effects.
7.3.1 Neurological and behavioural effects
Severe acute neurological effects such as psychosis and paralysis
occur when exposure exceeds 1500-3000 mg/m3 (500-1000 ppm) (Vigliani,
1954). Chronic encephalopathy may develop with long-standing exposure
to 150 mg/m3 (50 ppm) or more (Vigliani, 1954). More subtle
neurological changes, detectable by neurophysiological examination or
psychological testing, have been reported at lower concentrations. For
example, slowing of nerve conduction velocities and prolonged distal
latencies of several nerves have been found among viscose rayon
workers with a median exposure time of 15 years to combined
concentrations of carbon disulfide and hydrogen sulfide of
60-190 mg/m3 (20-60 ppm) in the 1950s, 30-95 mg/m3 (10-30 ppm) in
the 1960s, and mainly below 60 mg/m3 (20 ppm) during the years
preceding the examination (Seppäläinen & Tolonen, 1974). The same
workers were also studied for psychological performance (Tolonen,
1974). The most distinct difference compared with a control group was
found in the retardation of psychomotor speed. As a whole, "poor"
performance, as defined by Tolonen (1974), occurred 1.6 times more
frequently among the exposed workers. These results suggest that
concentrations around 60-90 mg/m3 (20-30 ppm) can produce
psychological disturbances. In an indirect way, Tuttle et al.a
corroborated this view by being able to relate behavioural test scores
to other indices of neurological health, thought to occur under
similar exposure conditions, but because no exposure data were
available, their findings are not directly applicable in the
exposure-effect sense.
According to Martynova et al. (1976), an excess of functional
nervous disorders was found among 108 workers exposed to carbon
disulfide concentrations about 8-11 mg/m3 (maximum approximately
20-25 mg/m3), for 10-15 years, compared with 390 control subjects.
For example, sensory polyneuritis was much more frequent and the
threshold for pain was significantly higher in the exposed group.
These studies indicate that background concentrations below about
60 mg/m3 (20 ppm) may cause neurological dysfunction.
7.3.2 Cardiovascular effects
Severe vascular effects manifesting themselves as
vasculoencephalopathy and renal atherosclerosis developed with
long-term exposure to carbon disulfide concentrations exceeding
150-300 mg/m3 (50-100 ppm) (Vigliani, 1954; Nofer et al., 1961).
Excess incidence of coronary heart disease has been reported with
longstanding exposure to concentrations averaging 30-125 mg/m3
(10-40 ppm) (Hernberg et al., 1970, 1973, Tolonen et al., 1975). The
mortality rate from coronary heart disease appeared to be most
affected while the incidence of milder manifestations such as angina
and ECG-changes was less influenced. In fact, most studies, published
so far, have failed to reveal any difference in the prevalence of
ECG-findings between exposed and unexposed workers (Tolonen et al.,
1976).
Quite recently, Vertin (1977) reported no difference between the
incidence of coronary heart disease found in control subjects and that
in workers exposed to about 30-95 mg/m3 (10-30 ppm) suggesting a
no-observed effect level for coronary heart disease in this range.
This finding does not necessarily contradict those referred to above;
possibly, the true exposure levels responsible for the excess coronary
heart disease in the Finnish studies were higher than those in
Vertin's study. It is probable that, in general, differences in actual
personal exposure levels in the studies-not necessarily those levels
measured and reported -- account for seeming discrepancies in results
and that exposure data are inadequate in most published studies.
a Tuttle et al., 1976. Unpublished report. (See footnote to section
6.3.10.1.)
Differences in blood pressure have been reported by Hernberg et
al. (1970) and Tolonen et al. (1975) between workers exposed to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) and
unexposed subjects, and by Martynova et al. (1976) in workers exposed
for 10-15 years to concentrations of about 10 mg/m3. These findings
indicate that effects upon the vascular system begin at low exposure
levels.
7.3.3 Ophthalmological effects
Among Japanese workers, the occurrence of retinal microaneurysms
was related to the duration of exposure (Sugimoto et al., 1976). The
exposure levels in earlier Japanese studies were not reported; in
present studies they are probably of the order of 60 mg/m3 (20 ppm).
The findings of retinal microaneurysms appears to be confined to
Japanese workers, since, according to a recent joint study, no such
effect could be demonstrated in Finns (Sugimoto et al., 1977). In
contrast, Finnish workers showed delayed peripapillary filling and
wider calibre of retinal arteries compared with control subjects
(Raitta et al., 1974). Exposure conditions under which these changes
occurred are described in section 6.3.2 and indicate that haemodynamic
changes in the ocular circulation are induced as early as coronary
effects.
7.3.4 Gonadal effects
The results of Vasiljeva (1973), Petrov (1969), and Finkova et
al. (1973) suggest that carbon disulfide concentrations below
10 mg/m3 (3 ppm) may cause disturbances in the ovarian function of
young females, resulting in menstrual disorders, pregnancy
complications, and spontaneous abortions. Given that the exposure
measurements reported are comparable with those of other studies, it
would seem that these would be the earliest detectable type of effects
in human beings.
A summary of the most relevant exposure-effect and exposure-
response findings is given in Table 3.
Table 3. Some exposure-effect and exposure-response relationships
for long-term exposure of man to carbon disulfide.
Concentration Exposure Symptoms & signs Reference
(mg/m3) time
(years)
10 5-10 Blood progesterone depressed, Vasiljeva (1973)
estriol increased, irregular
menstruation;
10 10-15 Sensory polyneuritis, increased Martynova et al. (1976)
pain threshold;
30 >3 Spontaneous abortions twice as Petroy (1969)
common and premature births 3
times as common as in controls;
30-120 >10 Coronary mortality about 5 times Tolonen et al. (1975)
that in controls, life expectancy Hernberg et al. (1973)
decreased 0.9-2.1 years depending Nurminen (1976)
on age; nonfatal infarctions 2.6 Hernberg et al. (1970)
times more frequent than in controls;
angina pectoris about twice as
prevalent as in controls; slightly
higher systolic and diastolic blood
pressure than in controls;
30-120 >6 Peripheral nervous and CNS Seppäläinen & Tolonen
dysfunction, conduction velocities (1974); Tolonen (1974)
slowed, psychological changes;
100-140 2-12 Decrease of testosterone and Cavalleri et al. (1970)
gonadotrophin LH in urine;
200-500 1-9 Ophthalmic pressure 18.4/14.7 Maugeri et al. (1966,
vs 15.3/11.6 kPa (138/110 vs 1967)
115/87 mmHg) in controls;
500-2500 0.5 Polyneuritis, myopathy acute Vigliani (1946)
psychoses
8. CONTROL OF EXPOSURE IN THE VISCOSE INDUSTRY
Technical and administrative measures for the protection of the
health of workers exposed to carbon disulfide in the production of
viscose rayon include:
-- exchange of production methods (substitution or elimination);
-- enclosure and local exhaust ventilation of the technological
process;
-- dilution (general) ventilation of the work room;
-- isolated and well-ventilated rooms for process control
operators and for rest periods;
-- use of personal respiratory protective equipment under certain
hazardous circumstances and,
-- rotation of workers periodically to areas free from carbon
disulfide exposure.
New developments such as the SINI viscose process make it
possible to reduce the total consumption of carbon disulfide to 70-80%
of that used in conventional processes (Sihtola, 1976). Occupational
health aspects must be taken into consideration at the planning stage
in the construction of new plants.
Enclosure of processes and machines is the second most effective
means of technical prevention. This technique should be applied in the
sulfidizing process and for spinning and washing machines. Possible
leakages in closed systems should be controlled by gas detectors. The
air removed from the processes by the exhaust ventilation should be
transported to a recovery system. The benefits of such a system are
both economic, since the plant has an interest in the recovery of
useful material, and hygienic, as the possibility of the pollution of
both the working rooms and the environment around the factory is
reduced. The air removed by exhaust ventilation is compensated with
pure air, which is brought to working sites by ventilation systems.
It is not possible to protect the workers in all situations by
ventilation systems alone. In certain circumstances, the workers may
be exposed to very high concentrations of carbon disulfide, in which
case respiratory protective devices must be used. The type of
respirator may vary from a chemical cartridge (organic vapour)
respirator, used for low concentrations, to supplied air or
self-contained breathing devices for very high concentrations. It is
also important to provide special isolated rooms with a slightly
positive pressure, compared with the work area contaminated with
carbon disulfide, for process control workers and for rest pauses.
Another factor of concern in the control of exposure and uptake
of carbon disulfide involves skin absorption resulting from direct
contact with the liquid. Where workers may come into contact with
liquid carbon disulfide, appropriate protective clothing, such as
synthetic rubber gloves and goggles or face shields, should be worn.
If carbon disulfide is spilled, potential sources of ignition
should be eliminated immediately, spark-proof ventilation provided,
and the spill cleaned up. A small spill should generally be allowed to
evaporate under conditions of good air circulation. However, large
spills should be covered with water and flushed into a retention basin
under a water layer. Carbon disulfide should not be drained into a
sewer system because of the possibility of an explosion.
In addition to these control methods, data obtained through the
biological monitoring of exposed workers can be used to determine
excessive uptake situations that require additional control measures.
The procedure for biological monitoring is discussed in section 6.5.
REFERENCES
ABRAMOVA, J. I. (1967) The questions of pathogenesis, specific
prevention and therapy of carbon disulfide intoxication. In:
Brieger, H. & Teisinger, L, ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 32-34.
ABUCEWICZ, A., SAMOCHOWIEC, L., & SZYSZKA, K. (1971) The role of
5-hydroxytryptamine in the course of experimental carbon
disulfide poisoning. Roca. Pomor. Akad. bled. Szczec, Suppl. 4,
pp. 89-96.
AGRANOVSKIJ, Z. M. & GOLOSCAPOV, O. D. (1972) [Hygienic assessment of
preventive diet for subjects exposed to carbon disulfide.] Gig.
i Sanit., 37 (II): 43-47 (in Russian).
AMERICAN STANDARDS ASSOCIATION (1941) Allowable concentration of
carbon disulfide, New York, ASA, 141, 7 pp. (ASA Z 37.3-1941).
ANDREEVA, N. B. (1970) [Experimental material for the diagnosis of
carbon disulfide poisoning, on the harmful effect of the poison
at the maximum permissible level for workroom air, and on the
passage of carbon disulfide in solution through the skin.] In:
Voprosy diagnostiki, specificeskoj profilaktiki i terapii
professional 'nyh zabolevanij, Leningrad, pp. 40-52 (in
Russian).
ANDRUSZCZAK, K. (1967) [The behaviour of ceruloplasmine level in the
blood serum in workers exposed to the carbon disulfide action.]
Med. Pr., 18:120-127 (in Polish).
ATTINGER, E. (1948) [Chronic carbon disulfide intoxication appearing
under the unusual symptoms of a heavy vascular disease.]
Schwiez. Med. Wochenscht., 78:667-669 (in German).
ATTINGER, E. (1954) [Chronic carbon disulfide intoxication and its
pathogenesis, its early detection and prevention.] Schweiz. Med.
Wochenschr., 84:912-913 (in German).
AUDIO-GIANOTTI, G. B. (1932) [Parkinsonism caused by professional
carbon disulphide poisoning.] Presse med., 40 (July-Dec.)
1289-1291 (in French).
AUSTONI, M. & D'AGNOLO, B. (1957) [Chronic carbon disulfide poisoning
and diabetes mellitus.] Minerva Med., 48:2500-2504 (in
Italian).
BAGON, D. A., CROSBY, M. T., HARDY, H. L., & SIMMONS, J. H. (1973) A
personal sampling technique for simultaneous determination of
hydrogen sulfide and carbon sulfide in air. Ann. occup. Hyg.,
16: 133-139.
BAIKOW, E. K. (1963) [Experimental data on the determination of the
maximum permissible concentration of carbon disulfide and
hydrogen disulfide in the atmosphere in their simultaneous
presence.] Gig. i Sanit., 28 (3): 3-8 (in Russian).
BARD, C. L. (1892) Cases in practice, malignant pustle and insanity
due to bisulphide of carbon. South Cal. Pract., 7: 476-485.
BARILJAK, I. R., VASILEVA, I. A., & KALINOVSKAJA, L. P. (1975) [Effect
of small concentrations of carbon disulfide and hydrogen sulfide
on intrauterine development in rats.] Arkh. Anat. Gistol.
Embriol., 58:77-81 (in Russian).
BARTONICEK, V. (1957) [The distribution of carbon disulfide in whole
blood, the brain and the suprarenal glands over a given period in
parenteral administration in white mice. Prac. Lek., 9:28-30
(in Czech).
BARTONICEK, V. (1958) [The distribution of free carbon disulphide and
bound carbon disulphide liberated by acid hydrolysis in the
organs of white rats.] Prac. Lek., 10: 334-338 (in Czech).
BARTONICEK, V. (1959) [Distribution of free carbon disulfide and bound
carbon disulfide liberated by acid hydrolysis in the organs of
white rats.] Prac. Lek., 10:504-510 (in Czech).
BASHORE, R., HAMILTON, A., LEWEY, F., ERSKINE, L., HAMMOND, J., HOUTZ,
R., BRACELAND, F., MCDONALD, R., BATSON, O., BELLET, S.,
CRESKOFF, F., EHRICH, W., & ALPERS, B. J. (1938) Survey of
carbon disulfide and hydrogen sulfide hazards in the viscose
rayon industry, Harrisburg, PA, Occupational Disease Prediction
Division, US Department of Labor and Industry p. 68 (Bull. 46).
BINET, L. & BOURLIERE, F. (1944) Sur les modifications du sang au
cours du sulfocarbonisme chronique. Arch. Mal. Prof., 6: 12-16.
BITTERSOHL, G. (1971) On relationships in action between carbon
disulfide and hydrogen sulfide. Med. Lavoro, 62: 554-556.
BITTERSOHL, G. & THIELE, H. (1977) The health of workers exposed to
carbon disulphide in the production of artificial fibres. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski (Finland), 6-10 June 1977, p. 5.
BLACKBURN, H., KEYS, A., SIMONSON, E., RAUTAHARJU, P., & PUNSAR, S.
(1960) The electrocardiogram in population studies. A
classification system. Circulation, 21: 1160-1175.
BOBSIEN, K. (1954) [New method for determination of uptake and binding
by endogenous substances.] Arch. Gewerbepath. Gewerbehyg., 13:
193-203 (in German).
BOND, E. J. & DE MATTEIS, F. (1969) Biochemical changes in rat liver
after administration of carbon disulphide, with particular
reference to microsomal changes. Biochem. Pharmacol., 18:
2531-2549.
BOND, E. J., BUTLER, W. H., DE MAITEIS, F., & BARNES, J. M. (1969)
Effects of carbon disulphide on the liver of rats. Br. J. ind.
Med, 26: 335-337.
BRIEGER, H. (1961) Chronic carbon disulfide poisoning. J. occup.
Med., 8: 302-308.
BRIEGER, H. (1967) Carbon disulphide in the living organism-Retention,
metabolism and physiological effects. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 27-31.
BRIEGER, H. & FRIEDMAN, M. H. F. (1949) In vitro effects on
digestive enzymes, theory and pathology of poisoning. J. ind.
Hyg., 31: 96-98.
BRIEGER, H. & TEISINGER, J., ed. (1966) Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 27-31.
BRUCE, A. (1884) Chronic poisoning by CS2. Edinburgh Med. J., 29:
1009.
BÜSING, K. H. (1952) [The determination of minute amounts of carbon
disulfide in blood.] Zentralbl. Arbeitsmed., 2: 115-122 (in
German).
BÜSING, K. H. & SONNENSCHEIN, W. (1954) [The binding of carbon
disulfide in the organs.] Zentralbl. Arbeitsmed. Arbeitschutz,
4:65-69 (in German).
BÜSING, K. H., SONNENSCHEIN, W., BECHER, E. W., & DREIHELLER, H.
(1953) [Experimental studies in animals on the binding of carbon
disulfide in organs using labelled carbon disulfide (C35S2).]
Z. Naturforsch., 86: 495-500 (in German).
CANDURA, F., GRAVERI, A., & STRANEO, G. (1962) [Clinical contribution
to studies on the relationship between arteriosclerosis and
carbon disulfide poisoning.] Folia Med., 45: 749-769 (in
Italian).
CAVALLERI, A. (1972) [Alterations in the endocrinological function as
an early sign of carbon disulfide poisoning.] Med. Lavoro, 63:
81-84 (in Italian).
CAVALLERI, A. (1975) Serum thyroxine in the early diagnosis of carbon
disulfide poisoning. Arch. environ. Health, 30: 85-87.
CAVALLERI, A. & ZUCCATO, F. (1965) [Endocrinological observations on
carbon disulfide poisoning. I: Adrenocortical function.] Folia
Med., 48: 519-527 (in Italian).
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1965) [Adrenocortical and
gonadal function in human carbon disulfide poisoning.] In:
Proceedings of the 28th National Congress on Occupational
Health, Naples, 1965, pp. 645-660 (in Italian).
CAVELLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1966a) Endocrinological findings in young workers
exposed to carbon disulphide. I. - Urinary excretion of total
17-ketosteroids. Med. Lavoro, 57: 566-572.
CAVALLERI, A., DJURIC, D., VISCONTI, E., REZMAN, I., MAUGERI, U., &
BRANKOVIC, D. (1966b) Endocrinological findings in young workers
exposed to carbon disulphide. II.-Urinary excretion of
17:hydroxy-corticosteroids. Med. Lavoro, 57: 573-578.
CAVALLERI, A., DJURIC, D., VISCONTI, E., BRANKOVIC, D., & MAUGERI, U.
(1966c) Androsterone and etiocholanolone in the urine of young
workers exposed to carbon disulphide. In: Proceedings of the XV
International Congress on Occupational Health, Vienna, 19-24,
1966, Vienna, Wiener Med. Akademie, Vol. II-I, pp. 347-353.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., & VISCONTI, E.
(1966d) Endocrinological findings in young workers exposed to
carbon disulphide. III.-Urinary excretion of 3alpha,
11-deoxy-17-ketosteroids. Med. Lavoro, 57: 755-760.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1967) 17-ketosteroids and 17-hydroxycorticosteroids
in the urine of young workers exposed to carbon disulphide. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 86-91.
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1970) L'excretion urinaire
de testosterone et de gonadotrophine stimulant les cellules
interstitielles (ICSH) chez des subjets exposes au sulfure de
carbone. Arch. Mal. Prof., 31: 23-30.
CAVALLERI, A., TACCOLA, A., GRAOVAC-LEPOSAVIC, LJ., MAUGERI, U., &
DJURIC, D. (1971) [Thyroinaemia in subjects exposed to carbon
disulfide.] Lav. Um., 23: 183-187 (in Italian).
CAVALLERI, A., POLATTI, F., & BOLIS, P. F. (1977) Acute effects of
tetraethylthiuram disulphide on serum levels of hypophyseal
hormones in humans. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski (Finland), 6-10
June 1977, p. 28.
CHERVENKA, C. H. & WILCOX, P. E. (1956) Chemical derivative of
chymotrypsinogen: I: Reaction with carbon disulfide. J. biol.
Chem. 222: 621-634.
CIMBAREVIC, T. K. (1970) [Ceruloplasmin activity in persons exposed to
carbon disulfide.] In: Voprosy diagnostiki, specifideskoj
profilaktiki i terapii professional 'nyh zabolevanij,
Leningrad, 65-67 (in Russian).
CIRLA, A.M., VILLA, A., & TOMASINI, M. (1972) [Study on the incidence
of coronaropathy in workers exposed to carbon disulfide in a
viscose rayon industry.] Med. Lavoro, 63: 431-441 (in Italian).
COHEN, A. E., PAULUS, H. J., KEENAN, R. G., & SCHEEL, L. D. (1958)
Skin absorption of carbon disulfide vapor in rabbits. I.
Associated changes in blood protein and zinc. Arch. ind. Health,
17: 164-169.
COHEN, A. E., SCHEEL, L. D., KOPP, J. F., STOCKELL, F. R., KEENAN, R.
G., MOUNTAIN, J. T., & PAULUS, H. J. (1959) Biochemical
mechanisms in chronic carbon disulfide poisoning. Am. Ind. Hyg.
Assoc. J., 20: 303-323.
CREPET, M., COBBATO, F., & SCANSETTI, G. (1956)] Some clinical
observations on vasculopathy resulting from chronic carbon
disulfide poisoning.] Minerva Med., 47: 379-387 (in Italian).
CULLEN, T. E. (1964) Spectrophotometric determination of
dithiocarbamates residues on food crops. Anal. Chem., 36:
221-224.
DALVI, R. R., POORE, R. E., & NEAL, R. A. (1974) Studies of the
metabolism of carbon disulfide by the rat liver microsomes. Life
Sci., 14: 1785-1796.
DANILOVA, T. N. (1968) [Data on the effect of carbon disulfide on the
coagulation system of the blood.] In: Materially 2-oj
konferencii molodyh naucnyh rabotnikov, Leningrad, pp. 102-104
(in Russian).
DAVIDSON, M. & FEINLEIB, M. (1972) Carbon disulfide poisoning: A
review. Am. Heart J., 83: 100-114.
DELIC, V., GRAOVAC-LEPOSAVIC, LJ., DJURIC, D., BERKES, I., &
DJORDJEVIC, V. (1966) The determination of sulphates in the urine
of workers exposed to carbon disulphide. Med. Lavoro, 57:
458-462.
DELPECH, A. (1856a) Notes sur les accidents que developpe, chez les
ouvriers en caoutchouc, l'inhalation du CS2 en vapeur. Gaz.
Hebd. med. Chir., 3: 40.
DELPECH, A. (1856b) Accidents que developpe, chez les ouvriers en
caoutchouc, l'inhalation du CS2 en vapeur. Union bled., 10:
265.
DELPECH, A. (1863) Industrie du caoutchou de souffle: Recherches sur
l'intoxication speciale que determine le CS2. Ann. Hyg. 2e
sér., 19: 65.
DE MATTEIS, F. (1974) Covalent binding of sulfur to microsomes and
loss of cytochrome P-450 during the oxidative desulfuration of
several chemicals. Mol. Pharmacol., 10: 849-854.
DE MATTEIS, E. & SEAWRIGHT, A. A. (1973) Oxidative metabolism of
carbon disulfide by the rat. Effect of treatments which modify
the liver toxicity of carbon disulfide. Chem. biol. Interact.,
7: 375-388.
DEMUS, H. (1964) [On the reception, chemical transformation and
excretion of carbon disulfide by the human body.] Arch.
Gewerbepath. Gewerbehyg., 20: 507-536 (in German).
DEMUS, H. (1967) The mechanism of absorption, metabolism and excretion
of carbon disulfide in the human body. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 42-49.
DE NICOLA, P., NICROSINI, F., FASOTTI, C., & DEL FAVERO, A. (1962)
[Comparative studies on the physiopathological picture of
experimental chronic carbon disulfide poisoning and
high-cholesterol diet I. Study of the lipid fractions in serum
and aorta.] G. Geront., 10: 207-220 (in Italian).
DEPARTMENT OF EMPLOYMENT AND PRODUCTIVITY (1968) Methods for
determination of toxic substances in air: Carbon disulfide
vapour. London, HMSO, 10 pp. (DEP Booklet No. 6).
DJERASSI, L. S. & LUMSROSO, R. (1968) Carbon disulphide poisoning with
increased ethereal sulphate excretion. Br. J. ind. Med., 25:
220-222.
DJURIC, D. (1963) [Metabolism of carbon disulphide.] Arh. Hig. Rada,
14: 23-31 (in Serbo-Croat).
DJURIC, D. (1967) Determination of carbon disulphide and its
metabolites in biological material. In: Brieger, H. & Teisinger,
J., ed. Toxicology of carbon disulphide, Amsterdam, Excerpta
Medica Foundation, pp. 52-61.
DJURIC, D. (1971) Some aspects of antabuse, carbon disulphide and
ethyl alcohol metabolism. Arh. Hig. Rada, 22: 171-177.
DJURIC, D., SURDUCKI, N., & BERKES, I. (1965) Iodine-azide test on
urine of persons exposed to carbon disulphide. Br. J. ind. Med.,
22: 321-323.
DJURIC, D., STOJADINOVIC, LJ., BOJOVIC, V., & REZMAN, I. (1967)
Excretion of zinc in the urine of persons exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation, pp.
118-121.
DJURIC, D., POSTIC-GRUJIN, A., GRAOVAC-LEPOSAVIC, L., & DELIC, V.
(1973) Disulfiram as an indicator of human susceptibility to
carbon disulfide excretion of diethyldithiocarbamate sodium in
the urine of workers exposed to CS2 after oral administration of
disulfiram. Arch. environ. Health, 26: 287-289.
DROGCINA, E. A. (1968) [Carbon disulfide poisoning.] In: Professional
'nye bolezni sistemy, Leningrad, Medicina, pp. 129-141 (in
Russian).
DUTKIEWICZ, T. & BARANOWSKA, B. (1967) The significance of carbon
disulphide skin resorption in the evaluation of exposure. In:
Brieger, H. & Teisinger, J., ed. Toxicology of Carbon
Disulphide, Amsterdam, Excerpta Medica Foundation, pp. 50-51.
EFREMOVA, N. F. & UZBEKOV, G. A. (1968) [Study of nitrogen metabolism
in the brains of white rats exposed to chronic carbon disulfide
poisoning.] In: Trudy Rjazanskogo Medicinskogo Instituta, 34:
163-172 (in Russian).
EHRHARDT, W. (1967) Experiences with the employment of women exposed
to carbon disulphide. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 240-244.
EL-GAZZAR, R., EL-SADIK, Y. M., & HUSSEIN, M. (1973) Changes in zinc
and serum proteins due to carbon disulphide exposure. Br. J.
ind. Med., 30: 284-288.
FAHIM, B. A., FAHMY, A.M., & KAMEL, M. Y. (1973) [Biochemical and
hematologic studies on carbon disulfide poisoning.] Z. Ges.
Hyg., 19: 538-541 (in German).
FAITH, W. L., KEYES, D. B., & CLARK, R. L. (1965) Industrial
Chemicals, 3rd ed., New York, John Wiley & Sons, pp. 226-227.
FERRERO, G. P. (1969) [Occurrence of diabetes in workers exposed to
carbon disulfide.] Med. Lavoro, 60: 38-42 (in Italian).
FERRARO, A., JARVIS, G., & FLICKER, D. (1.941) Neuropathologic changes
in experimental carbon disulfide poisoning in cats. Arch.
Pathol., 32: 732-738.
FINKOVA, A., SIMKO, A., JINDRICHOVA, J., KOVARIK, J., PREININGEROVA,
O., KLIMOVA, A., & KORISKO, F. (1973) [Gynaecological problems of
women working in an environment contaminated with carbon
disulfide.] Cesk. gynekol., 38: 535-536 (in Czech).
FINULLI, M. & GHISLANDI, E. (1959) [Occurrence of diabetes in chronic
carbon disulfide poisoning.] Med. Lavoro, 50: 189-192 (in
Italian).
FISCHER, J. & MICHALOVA, C. (1956) [Neurohistological changes caused
by carbon disulfide.] Prac. Lek., 8:16-19 (in Czech).
FOA, V., CASSITO, M. G., FORZI, M., & BULGHERONI, C. (1976) Mental
performance and personality disorders among workers exposed to
carbon disulphide: Comparison between two different rayon plants.
In: Horvarth, M., ed. Adverse effects of environmental chemicals
and psychotropic drugs, neurophysiological and behavioral tests.
Amsterdam, Oxford, New York, Elsevier, Vol. 2, pp. 173-182.
FOREMAN, W. (1886) Notes of a fatal case of poisoning by bisulphide of
carbon. Lancet, 2: 118-119.
FRANCINE, A. P. (1905) Acute carbon disulfide poisoning. Am. Med.,
9: 871.
FRANTIK, E. (1970) The development of motor disturbances in
experimental chronic carbon disulphide intoxication. Med.
Lavoro,, 61: 309-313.
FREUNDT, K. J. (1973) The inhibitory action of inhaled carbon
disulfide on microsomal enzymes as a sensitive restriction of
liver function. In: Horvath, M., ed. Adverse effects of
environmental chemical and psychotropic drugs: Quantitative
interpretation of functional tests, Amsterdam, Oxford, New
York, Elsevier, pp. 161-164.
FREUNDT, K. J. (1974) Ethanol and acetaldehyde levels in alcoholized
rats and man under carbon disulphide exposure. Abstracts of the
III International Symposium on the Toxicology of Carbon
Disulphide, Cairo-Alexandria (Egypt) 4-9 May, 1974, p. 28.
FREUNDT, K. J. (1975) [Carbon disulfide.] In: Henschler, D., ed.
Gesundheitsschadliche Arbeitstoffe, 4th ed., Federal Republic
of Germany, Verlag Chemic Weinheim, pp. 1-20.
FREUNDT, K. J. (1977) Animal experimentation studies on a possible
induction of hepatic microsomal mixed-function oxygenases (MFO)
by inhaled carbon disulphide (CS2). In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 24.
FREUNDT, K. J. & DREHER, W. (1969) Inhibition of drug metabolism by
small concentrations of carbon disulfide. Nauyn-Schmiedebergs
Arch. Pharmakol., 263: 208-209.
FREUNDT, K. J. & HENSCHLER, D. (1971) Inhibition of oxidative drug
metabolism by low doses of inhaled carbon disulfide: Kinetics and
specificity. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Bahja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 9.
FREUNDT, K. J. & KÜRZINGER, R. (1975) Energy potential and hepatic
function in rats under acute exposure to carbon disulphide. Int.
Arch. Arbeitsmed., 34: 269-282.
FREUNDT, K. J. & KUTTNER, P. (1969) Oxidative drug metabolism
following low carbon disulphide concentrations.
Nauyn-Schmiedebergs Arch. Pharmakol., 264: 232-233.
FREUNDT, K. J. & LIEBERWIRTH, H. (1974) Influence of inhaled carbon
disulfide on acetaldehyde production and liver function in
alcoholized man. Nauyn-Schmiedebergs Arch. Pharmakol., 282
(Suppl. R.21).
FREUNDT, K. J. & NETZ, H. (1973) Influence of carbon disulfide and
thiurams on ethanol elimination and acetaldehyde production.
Arch. Pharmakol. exp. Pathol., 277 (Suppl. R18).
FREUNDT, K. J., LIEBALDT, G. P., & SIEBER, K. H. M. (1974a) Effect of
barbiturates on the liver of rats exposed to carbon disulphide
vapor. Int. Arch. Arbeitsmed., 32: 297-303.
FREUNDT, K. J., SCHAUENSURG, K. J., & EICHHORN, P. (1974b) Effect of
acute exposure to carbon disulfide vapor upon some components of
the hepatic microsomal enzyme system in rats. Arch. Toxicol.,
32 (3): 233-240.
FREUNDT, K. J., SCHNAPP, E., & DREHER, W. (1975) Pharmacokinetics of
inhaled carbon disulphide in rats in relation to its inhibitory
effect on the side-chain oxidation of hexobarbital. Int. Arch.
occup. environ. Health, 35: 173-185.
FREUNDT, K. J., LIEBERWIRTH, K., NETZ, H., & POHLMANN, E. (1976) Blood
acetaldehyde in alcoholized rats and humans during inhalation of
carbon disulphide vapor. Int. Arch. occup. environ. Health, 37:
35-46.
FRUSCELLA. R. (1962) [Urinary excretion of 17-ketosteroids and
17-hydroxycorticosteroids in human subjects suffering from carbon
disulfide poisoning.] Clin. ter., 23: 749-755 (in Italian).
GADASKINA, I.D. & ANDREEVA, N. B. (1960) [Some biochemical shifts in
the organism in carbon disulfide poisoning. Free SH groups and
ceruloplasmin in the blood: Copper and zinc metabolism.] Gig.
Tr. Prof Zabol., 13: 28-32 (in Russian).
GAVRILESCU, N. & LILIS, R. (1967) Cardiovascular effects of
long-extended carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 165-167.
GHERASE, G. (1976) Psychological changes in workers exposed to
atmospheres containing carbon disulphide. In: Adverse effects of
environmental chemicals and psychotropic drugs.
Neurophysiological and behavioral tests, Amsterdam, Vol. 2,
334 pp.
GILIOLI, R., BULGHERONI, C., BERTAZZI, P. A., CIRLA, A. M., CASSITTO,
M. G., & TOMASINI, M. (1977) Endocrinological, psychological and
neurological study of carbon disulphide workers. III.
Neurological and neurophysiological findings. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), June 6-10, 1977, p. 13.
GOLDBERG, M. E., JOHNSON, H. E., POZZANI, U. C., & SMYTH, H. F. (1964)
Behavioral response of rats during inhalation of trichlorethylene
and carbon disulphide vapours. Acta Pharmacol., 21: 36-44.
GONDZIK, M. (1971) Histology and histochemistry of rat testicles as
affected by carbon disulphide. Polish med. J., 10: 133-139.
GORDY, S. T. & TRUMPER, M. (1938) CS2 poisoning with a report of six
cases. J. Am. Med. Assoc., 110: 1543.
GORDY, S. T. & TRUMPER, M. (1940) Carbon disulphide poisoning-Report
of 21 cases. Ind. Med., 9: 231-234.
GORNY, R. (1971) The level of pyridoxal phosphate (PLP) in the blood
plasma of rats exposed to carbon disulfide. Biochem. Pharmacol.,
20: 2114-2115.
GORNY, R. (1974) [Effect of prolonged exposure to carbon disulfide on
tryptophan metabolism in rats.] Med. Pr., 25: 21-27 (in
Polish).
GOTO, S. & HOTTA, R. (1967) The medical and hygienic prevention of
carbon disulphide poisoning in Japan. In: Brieger, H. &
Teisinger, J.: ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 219-230.
GOTO, S., HOTTA, R., & SUGIMOTO, K. (1971) Studies on chronic carbon
disulfide poisoning. Int. Arch. Arbeitsmed., 28: 115-126.
GOTO, S., SUGIMOTO, K., HOTTA, R., BABA, T., NAKAMURA, G., & SEKI, Y.
(1977) Regional patterns of angiopathy due to CS2. Egypt. J.
occup. Med., 5: 155-159.
GRAOVAC-LEPOSAVIC, LJ. & JOVICIC, M. (1971) Report on one case of
chronic carbon disulfide intoxication. In: Abstracts of the H
International Symposium on Carbon Disulphide Toxicology, Banjo
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 19.
GRAOVAC-LEPOSAVIC, LJ., DJURIC D., PAVLOVIC, A., & JOVICIC, M. (1967)
The use of iodine-azide test for early diagnosis of carbon
disulphide poisoning. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 62-69.
GRAOVAC-LEPOSAVIC, LJ., JOVICIC, M., SAVIC, S., DJURIC, D., &
POSTIC-GRUJIN, A. (1977) Findings in workers some years after
CS2 poisoning. Egypt. J. occup. Med., 5: 53-60.
GREGORCZYK, J., KOSMIDER, K., WIECKOWSKI, B., & WESOLOWSKA, T. (1975a)
Investigations on the extent of the liver lesion in the rat by
experimental inhalation poisoning with carbon disulfide. I.
Analysis of biochemical changes in the blood serum. Int. Arch.
Arbeitsmed., 34: 65-80.
GREGORCZYK, J., WIECKOWSKI, B., & WESOLOWSKA, T. (1975b)
Investigations on the extent of liver lesion in rats by
experimental inhalation poisoning with carbon disulfide. II.
Analysis of biochemical changes in liver homogenate and its
fractions. Int. Arch. Arbeitsmed., 34: 151-166.
GUARINO, A. & ARCIELLO, G. (1954) [Histological study on coronary
arteries in chronic experimental carbon disulfide poisoning.]
Folia Med. (Napoli) 37: 1022-1044 (in Italian).
HAKKY, U. (1977) Physiological manifestations after exposure to CS2.
Egypt. J. occup. Med., 5: 47-52.
HAMILTON, A. (1925) Industrial poisons in the United States, New
York, Macmillan Co., 590 pp.
HAMILTON, A. (1940) Occupational poisoning in the viscose rayon
industry, Washington, DC, Department of Labor, pp. 1-79 (Bull.
No. 34).
HÄNNINEN, H. (1971) Psychological picture of manifest and latent
carbon disulphide poisoning. Br. J. ind. Med., 28: 374-381.
HÄNNINEN, H. (1974) Behavioral study of the effects of carbon
disulfide. In: Xintaras, C. El, Johnson, B., De Groot, I., ed.
Behavioral toxicology, Washington, DC, Department of Health,
Education, & Welfare, pp. 72-79.
HÄNNINEN, H., NURMINEN, M., TOLONEN, M., & MARTELIN, T. (1978)
Psychological tests as indicators of excessive exposure to carbon
disulfide. Scand. J. Psychol., 19(2): 163-174.
HARA, I. (1958) [On the carbohydrate metabolism in chronic carbon
disulfide poisoning.] Q. J. labour Res. Jpn, 6: 1-20 (in
Japanese).
HARASHIMA, S. & MASUDA, Y. (1962) Quantitative determination of
absorption and elimination of carbon disulfide through different
channels in human body. Int. Arch. Gewerbepath. Gewerbehyg.,
19: 263-269.
HARASHIMA, S., TOYAMA, T., & SAKURAI, T. (1960) Serum cholesterol
level of viscose rayon workers. Keijo J. Med., 9: 81-90.
HASSMAN, P., SIMKO, A., JINDRICHOVA, J., HEROUT, V., & HRADSKY, M.
(1967) The problem of chronic gastritis in artificial fibres
industry. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation,
pp. 182-189.
HERNBERG, S. & NORDMAN, C-H. (1969) Failure to find increased
excretion of zinc or magnesium in humans exposed to carbon
disulphide. Med. Lavoro, 60: 163-168.
HERNSERG, S., MOWE, G., VIRKOLA, P., PARTANEN, T., & NORDMAS, C-H.
(1969) Magnesium and zinc values of erythrocytes and plasma for
workers exposed to carbon disulfide. Work Environ. Health, 6:
9-13.
HERNSERG, S., PARTANEN, T., NORDMAN, C-H., & SUMARI, P. (1970)
Coronary heart disease among workers exposed to carbon
disulphide. Br. J. ind. Med., 27: 313-325.
HERNBERG, S., NORDMAN, C-H., PARTANEN, T., CHRISTIANSEN, V., &
VIRKOLA, P. (1971) Blood lipids, glucose tolerance and plasma
creatinine in workers exposed to carbon disulphide. Work
Environ. Health, 8: 11-16.
HERNBERG, S., NURMINEN, M., & TOLONEN, M. (1973) Excess mortality from
coronary heart disease in viscose rayon workers exposed to carbon
disulfide. Work Environ. Health, 10: 93-99.
HERNBERG, S., TOLONEN, M., & NURMINEN, M. (1976) Eight-year follow-up
of viscose rayon workers exposed to carbon disulfide. Scand. J.
Work Environ. Health, 2: 27-30.
HIGASHIDA, T., YAMAMOTO, T., & SHINDO, S. (1959) [Electromyographic
study of the effects of carbon disulfide administration on spinal
reflex.] Brain Nerve, 11(4): 253-260 (in Japanese).
HOCKWIN, O. & SAVIC, S. (1968) [Reversibility of biochemical changes
in lease and retina of the rabbit's eye in carbon disulfide
poisoning.] Albrecht Graefs Arch. klin. exp. Ophthalmol., 175:
7-12 (in German).
HOLASOVA, P. (1969) [Observations on a group of children from an area
polluted by carbon disulfide and hydrogen sulfide exhalation
compared with a control group of children.] Cs. Hyg., 14:
260-265 (in Czech).
HORVATH, M. & MIKISKA, A. (1957) [The influence of carbon disulfide on
the CNS of rabbits as seen in the EEG.] Prat. Lek., 9: 22-28
(in Czech).
HOTTA, R. & GOTO, S. (1971) A fluoroscein angiographic study on
microangiopathia sulfocarbonica Jpn. J. Ophthal., 15: 132-139.
HOTTA, R. & SAVIC, S. (1972) Fluorescein angiographic and
ophthalmodynamometric study on chronic carbon disulfide
poisoning. Med. Lavoro, 63: 442-447.
HUEPER, M. C. (1936) Etiologic studies on the formation of skin
blisters in viscose workers. J. ind. Hyg., 18: 432-447.
IVANOVA, F. G. (1967) [Some data on the status of erythropoiesis on
exposure to low concentrations of carbon disulfide.] In: Voprosy
gigieny truda i profpatologii, Leningrad, pp. 247-248 (in
Russian).
JAKUBOWSKI, M. (1968) [Studies of the conversion of carbon disulphide
to compounds catalysing the iodine-azide reaction. I.
Chromatographic studies.] Med. Pr. 19: 244-251 (in Polish).
JAKUBOWSKI, M. ( 1971) [Studies on the carbon disulfide metabolism of
compounds catalyzing the iodine azide reaction.] Med. Pr. 22:
195-206 (in Polish).
JÄRVISALO, J. & DE MATTEIS, F. (1977) Carbon disulphide induced loss
of liver microsomal haem and cytochrome P-450. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 23.
JÄRVISALO, J., SAVOLAINEN, H., ELOVAARA, E., & VAINIO, H. (1977) The
in vivo toxicity of CS2 to liver microsomes: Binding of
labelled CS2 and changes of the microsomal enzyme activities.
Acta Pharmacol. Toxicol., 40: 329-336.
JUMP, H. D. & CRUICE, J. M. (1904) Chronic poisoning from bisulphide
of carbon. Univ. Penn. med. Bull., 17: 193-196.
JUNTUNEN, J., HALTIA, M., & LINNOILA, I. (1974) Histochemically
demonstrable non-specific cholinesterase as an indicator of
peripheral nerve lesion in carbon disulfide induced
polyneuropathy. Acta Neuropathol. (Berl.), 29: 361-366.
JUNTUNEN, J., LINNOILA, I., & HALTIA, M. (1977) Histochemical and
electron microscopic observations on the myoneural junctions of
rats with carbon disulfide induced polyneuropathy. Scand. J.
Work Environ. Health, 3: 1-15.
KARAJOVIC, D., GRAOVAC-LEPOSAVIC, LJ., & SURADNICI, V. J. (1964) [The
influence of CS2 on the health of workers in factory in Serbia.]
Arh. Hig. Rada, 15: 87-92 (in Serbo-Croat).
KLIMKOVA-DEUTSCHOVA, E. (1965) [Early diagnosis in industrial
neurology.] S.Z.N., Praha 1965, 260 pp. (in Czech).
KNAVE, B., KOLMODIN-HEDMAN, B., PERSSON, H. E., & GOLDBERG, J. M.
(1974) Chronic exposure to carbon disulfide: Effects on
occupationally exposed workers with special reference to the
nervous system. Work Environ. Health, 11: 49-58.
KOELSCHS HANDBUCH DER BERUFSERKRANKUNGEN (1972) Jena, Gustav Fischer
Verlag. Vol. II, pp. 270-275.
KOESTER, G. (1899) [Contribution to the field of chronic CS:
intoxication.] Arch. Psychiat., 32: 606 (in German).
KOLMODIN-HEDMAN, B. (1976) Iodine-azide test in a Swedish viscose
factory related to air concentration of CS2 and to health.
Arbete och Halea, No. 8. pp. 23-28.
KOMRSKA, M. (1967) L'importance de la rheoencephalographie pour le
diagnostic d'intoxication par le sulfure de carbone. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulfide,
Amsterdam, Excerpta Medica Foundation, pp. 140-144.
KOPECKY, J. (1973) [Metabolism of carbon disulfide in rats.] Acta
Hyg. Epidemiol. Microbiol. (Prague), Suppl. 12, pp. 3-6
(in Czech).
KOZIOL, A., KULCZYCKA, H., & ROZEWICKI, S. (1971) [Development of
children, whose mothers were professionally exposed to carbon
disulfide.] Med. Pr., 22: 511-514 (in Polish).
KROLIKOWSKA, W. & RUCINSKA, Z. (1959) EEG examinations in chronic
professional intoxication by carbon disulfide. Electrortceph.,
clin. Neurophysiol., 11: 180 (Abstract No. 9).
KUBOTA, J. (1967) Historical view of carbon disulfide poisoning in the
Japanese viscose rayon industry. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulfide, Amsterdam, Excerpta Medica
Foundation, pp. 192-196.
KUJALOVA, V. (1971) The effect of carbon disulfide on vitamin B6
metabolism. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Banja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 30.
KUJALOVA, V. (1973) [Changes in the pyridoxinemetabolism and in the
blood levels of ceruloplasmin in humans exposed to carbon
disulfide.] Acta Hyg. Epidemiol. Microbiol., Suppl. 12 (in
Czech).
KUJALOVA V. & TINTERA, J. (1970) The effect of carbon disulfide on
tryptophan metabolism. Med. Lavoro, 61: 227-231.
KÜRZINGER, R. & FREUNDT, K. J. (1969) Changes in hepatic functions
following low carbon disulfide concentration.
Naunyn-Schmiedebergs Arch. Pharmakol., 264: 261-262.
LANCRANJAN, I. (1972) Alterations of spermatic liquid in patients
chronically poisoned by carbon disulfide. Med. Lavoro, 63:
29-33.
LANCRANJAN, I., POPESCU, H. I., & KLEPSCH, I. (1969) Changes of the
gonadic function in chronic carbon disulfide poisoning. Med.
Lavoro, 60: 566-571.
LANCRANJAN, I., FLOREA, O., & PETRESCU, M. (1971) Corticosuprarenal
insufficiency as a feature of carbon disulfide poisoning. Med.
Lavoro, 62: 557-561.
LANCRANJAN, I., SUKMANSKY, M., STANUCA, L., ANTONESCU, C., & POPESCU,
H. I. (1972) Study of the thyroid function in chronic carbon
disulfide poisoning. Med. Lavoro, 63: 123-125.
LANGELEZ, A. (1946) Viscose et sulfure de carbone. Arch. Belg. Med.
Soc. Hyg. Rev. Pathol., 4: 67-85.
LAUDENHAIMER, R. (1899) [The carbon disulfide intoxication of rubber
workers.] Leipzig, Veit & Co., 168 pp. (in German).
LAURMAN, W. & WRONSKA-NOFER, T. (1977) [Effect of carbon disulfide on
the amount of cholesterol in the tissues of rats.] Med. Pr.,
28: 77-82 (in Polish).
LAZAREV, N.J., ABRAMOVA, ZH. I., & CERNY, Z. C. (1965) [On possible
adverse reactions following use of some drugs by persons handling
carbon disulfide.] Gig. Tr. Prof. ZaboL, 9: 24-27 (in Russian).
LEITHE, W. (1971) Determination of carbon disulfide at the work site.
The analysis of air pollutant, 2nd ed., Ann Arbor, MI, Humphrey
Science Pub., pp. 228-229.
LEWEY, F. H. (1941) Neurological, medical and biochemical signs and
symptoms indicating chronic industrial carbon disulfide
absorption. Ann. int. Med., 15: 869-883.
LEWEY, W. E., ALPERS, B. J., BELLET, S., GRESKOFF, A. J., DRABLEIN, D.
L., ERICH, W. E., FRANK, J. H., JONAS, L., MCDONALD, R.,
MONTGOMERY, E. J., & REINHOLD, J. G. (1941) Experimental chronic
poisoning in dogs, clinical, biochemical and pathological study.
J. ind. Hyg., 23: 415-436.
LILIS, R. (1974) Behavioral effects of occupational carbon disulfide
exposure. In: Xintaras, C., Johnson, B. L., & de Groot, I., ed.
Behavioral toxicology, early detection of occupational hazards,
Cincinnati, US DHEW, Public Health Service, Center for Disease
Control, National Institute for Occupational Safety and Health,
pp. 51-59 (NIOSH 74-126).
LINIECKI, J. (1960) The effect of chronic carbon disulfide poisoning
on thiamine balance in rats. J. Hyg. Epidemiol. Microbiol.
Immunol. 4: 212-222.
LINNOILA, I., HALTIA, M., SEPPÄLÄINEN, A.M., & PALO, J. (1975)
Experimental carbon disulphide poisoning. Morphological and
neurophysiological studies. In: Körney, S., Tariska, S., &
Gosztonyi, G., ed. Proceedings of the VII International Congress
on Neuropathology Académiai Kiado, Budapest 1-7 September 1975,
pp. 383-386.
LUKAS, E. (1969) Conduction speed of peripheral nerves in persons
exposed to carbon disulfide (Electromyography study). Int. J.
clin. Ther. Toxicol., 2: 354-358.
LUKAS, E. (1970) Stimulation electromyography in experimental
toxicology (Carbon disulphide neuropathy in rats). Med. Lavoro,
61: 302-308.
LUKAS, E. (1973) [Some important factors in the development of
experimental carbon disulfide neuropathy in rats.] Cs. Neurol.
Neurochir., 36: 169-173 (in Czech).
LUKAS, E. (1974) Copper and zinc levels in peripheral nerve tissues of
rats with experimental carbon disulfide neuropathy. In:
Abstracts of the III International Symposium on Toxicology of
Carbon Disulphide, Cairo-Alexandria, Egypt, 4-9 May, 1974,
p. 8.
LUKAS, E. & HROMADKA, M. (1977) Conduction velocity of the ulnar and
tibial nerves in workers occupationally exposed to low
concentrations of carbon disulphide. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, June 6-10, 1977, p. 18.
LUKAS, E., KOTAS, P, & OBRUSNIK, I. (1974) Copper and zinc levels in
peripheral nerve tissues of rats with experimental carbon
disulphide neuropathy. Br. J. ind. Med., 31: 288-291.
LUKAS, E., KUSALOVA, V., & KOCINOVA, F. (1975) Activity of
ceruloplasmin in the serum of rats with experimental carbon
disulphide neuropathy. In: McCallum, R. I., ed. Abstracts of the
XVIII International Congress on Occupational Health, Brighton,
England, 14-19 September, 1975, pp. 299-300.
LUKAS, E., TEISINGER, J, Jr, & BASS, A. (1977) Activity of muscle
enzymes in rats with experimentally developed carbon disulphide
polyneuropathy. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 19.
LYSINA, G. G. (1967) Some changes in the internal organs due to the
effects of carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 179-181.
MACK, T., FREUNDT, K. J., & HENSCHLER, D. (1974) Inhibition of
oxidative N-demethylation in man by low doses of inhaled carbon
disulphide. Biochem. Pharmacol., 23: 607-614.
MADLO, Z. & SOUCEK, B. (1953) [Absorption, metabolism, and action of
carbon disulfide in the organism. VII. Inhibition of serum
cholinesterase by carbon disulfide.] Prac. Lek., 6: 312-313 (in
Czech).
MADLO, Z., PEIZKER, & SOUCEK, B. (1953) [The absorption, metabolism
and action of carbon disulphide in the organism. V. The effect of
carbon disulfide on tissue respiration.] Prac. Lek., 5: 263-266
(in Czech).
MAGISTRETTI, M. & PEIRONE, E. (1961)The action of carbon disulphide on
cerebral monoamine oxidase. Med. Lavoro, 52: 1-10.
MAGOS, L. (1972) Relevancy of bivalent sulphur excretion to carbon
disulphide exposure in different metabolic conditions. Br. J.
ind. Med., 29: 90-94.
MAGOS, L. (1975) The clinical and experimental aspects of carbon
disulphide intoxication. Rev. environ. Health, 11: 65-80.
MAGOS, L. & BUTLER, H. (1972) Effect of phenobarbitone and starvation
on hepatotoxicity in rats exposed to carbon disulfide vapor.
Br. J. ind. Med., 29: 95-98.
MAGOS, L. & JARVIS, J. A. E. (1970a) The effects of carbon disulphide
exposure on brain catecholamines in rats. J. Pharm. Pharmacol.,
29: 936-938.
MAGOS, L. & JARVIS, J. A. E. (1970b) The effects of carbon disulfide
exposure on brain catecholamines in rats. Br. J. Pharmacol.,
39: 26-33.
MAGOS, J., GREEN, A., & JARVIS, J. A. E. (1974) Half life of CS2 in
rats in relation to its effect on brain catecholamines. Int.
Arch. Arbeitsmed., 32: 289-296.
MANU, P, LANCRANJAN, I., & ZAMFIRESCU, Z. (1970) Investigation of
serum LDH and its isoenzymatic model in carbon disulphide
polyneuritis. Int. Arch. Arbeitsmed., 26: 45-49.
MANU, P., LILIS, R., NESTORESCU, B., LANCRANJAN, I., ROSENTA, A.,
IONESCU, S., & KAUFMAN, G. (1971) Effects of carbon disulfide on
cholesterol and ß-lipoprotein levels in young men. In: Abstracts
of the II International Symposium on Toxicology of Carbon
Disulfide, Banja Koviljaca, Yugoslavia, 25-28 May, 1971, pp.
28-29.
MARTINO, P., COSCIA, G. C., & PERRELLI, G. (1963) [Lipedaemia in
carbon disulfide poisoning. Preliminary observations.[ Folia
Med., 46: 1110-1114 (in Italian).
MARTINO, P., PERRELLI, G., & COSCIA, G. C. (1964) [Lipedaemia in
carbon disulfide poisoning.[ Arch. Sci. Med., 117: 124-130 (in
Italian).
MARTYNOVA, A. P., ZELENKIN, A. N., GOLJAKOVA, L. P., MILKOV, L. E.,
GRODETSKAJA, N. S, & AGADZANOVA, A. A. (1976)[Clinical hygienic
and experimental investigations of the action on the body of
small concentrations of carbon disulfide.] J. Hyg. Sanit., 5:
25-28 (in Russian).
MASSOUD, A. E. E., SADEK, S., EL HAWARY, M. F., & HASSANEIN, M. (1971)
Changes in the electrocardiogram and lung function in workers
exposed to carbon disulfide. In: Abstracts of the II
International Symposium on Toxicology of Carbon Disulfide, Banja
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 6.
MASSOUD, A. A. E., ABDEL KARIM, A. H., & FARES, R. (in press) Serum
thyroxine, serum cholesterol and its fractions in workers exposed
to carbon disulphide. Egypt. J. occup. Med.
MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., VISCONTI, E., IOVICIC, M., &
CAVALLERI, A. (1966a) [Cerebrovascular damage in young workers
exposed to carbon disulfide.] Med. Lavoro, 57: 701-708 (in
Italian).
MAUGERI, U., IOVICIC, M., CAVALLERI, A., GRAOVAC-LEPOSAVIC, LJ.,
VISCONTI, E., & REZMAN, I. (1966b) [Peripheral vascular damage in
young workers exposed to carbon disulfide.] Med. Lavoro, 57:
709-714 (in Italian).
MAUGERI, U., VISCONTI, E., & CAVALLERI, A. (1966c)] Vascular
alterations in the retina of young workers exposed to carbon
disulfide.] Med. Lavoro, 57: 741-745 (in Italian).
MAUGERI, U., CAVALLERI, A., & VISCONTI, E. (1966d)
[Ophthalmodynamography in occupational carbon disulfide
poisoning.] Med. Lavoro, 57: 730-740 (in Italian).
MAUGERI, U., CAVELLERI, A., & VISCONTI, E. (1967)
Ophthalmodynamographic study in young workers exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Excerpta Medica Foundation, Amsterdam, pp.
150-152.
MAUGERI, CAVALLERI, A., MAUGERI, U., & TACCOLA, A. (1971) New aspects
in the pathogenetic mechanism of carbon disulphide toxic action.
Med. Lavoro, 62: 398-403.
MCCAMMON, C., Jr, QUINN, M. P., & KUEPEL, R. E. (1975) A charcoal
sampling method and a gas chromatographic analytical procedure
for carbon disulfide. Am. Ind. Hyg. Assoc. J., 36: 618-625.
MCKEE, R. W. (1941) Solubility of carbon disulphide vapor in body
fluids and tissues. J. ind. Hyg., 23: 484-489.
MCKENNA, M. J. & DI SRTEFANO, V. (1975) A proposed mechanism of the
action of carbon disulfide on dopamine-ß-hydroxylase. Toxicol.
appl. Pharmacol., 33: 137.
MELISSINOS, J.-C. & JACOBIDES, G. (1967) Sur quelques formes nerveuses
d'intoxication observées en Grece chez des sujets exposeés au
sulfure de carbone. Arch. Mal. Prof., 28: 605-611.
MERLEVEDE, E. (1951) [Toxicology of carbon disulfide.] Sinte
Catharina, Brugge, 180 pp. (in Dutch).
MIHAIL, G., MIHAILA, D., BRANISTEANU, D., BODNAR, J., & CIOATA, V.
(1968) Manifestations cliniques, fonctionnelles et biochimiques
des stades precoces du sulfocarbonisme. Arch. Mal. Prof., 29:
109-120.
MILIC, S. (1965) [Some observations concerning the determination of
CS2 in the urine of exposed workers.] In: Zbornik radova o
toksikologiji CS2 (Proceedings in Yugoslavian symposium on CS2
toxicology, Loznica, 1965, p. 41-42 (in Serbo-Croat).
MILKOV, L. E., MONAENKOVA, A.M., BYALKO, N. K., GLOTOVA, K. V.,
VERETINSKAJA, A. G., ALI-ZADE, K. A., MAKARENKO, Y. L.,
NIKOLSKAJA, O. I., & KURSAKOV, A. V. (1970) [On the initial forms
of chronic carbon disulfide poisoning.] Gig. Tr. Prof. Zabol.,
14: 28-32 (in Russian).
MINDEN, H., GOHLKE, R., & ROTHE, R. (1967) [Contribution to the
pathogenesis of chronic carbon disulfide poisoning.] Z. Ges.
Hyg. Grenz., 13: 815-822 (in German).
MISIAKIEWICZ, Z., SZULINSKA, O., & CHYBA, A. (1972) [Influence of a
carbon disulfide, hydrogen sulfide mixture in air on white rats
under the conditions of permanent exposure.] Rocz. Panstw. Zakl.
Hig., 23: 465-475 (in Polish).
MOREO, S. & CANDURA, F. (1960) [The behaviour of serum-clotting
factors in animals treated with sodium arsenite, carbon
disulfide, cadmium chloride and lead acetate.] Boll. Soc. Med.
Chir., (Pavia), 74: 447-455 (in Italian).
MOWÉ, G. (1971) Coronary heart disease and occupational exposure to
carbon disulfide. In: Abstracts of the III International
symposium on toxicology of carbon disulfide, Banja Koviljaca,
Yugoslavia, 25-28 May, 1971, p. 7.
MURASKO, V. A. (1975) [The morbidity of the digestive organs through
working in contact with carbon disulfide in the viscose
industry.] Vrach. Delo, No. 4, pp. 113-117 (in Russian).
NAKAMURA, K., HARADA, M., TATETSU, S., MIYAGAWA, T., TSUKAYAMA, T.,
KAWANO, K., KABASHIMA, K., HIRATA, M., & TOYA, G. (1974) A
clinical study on chronic carbon disulfide poisoning. Psychiatr.
Neuro. Jpn., 76: 243-273.
NEAL, R. A., KATAMAKI, T., HUNTER, A. L., & CATIGNANI, G. (1976)
Monooxygenase catalyzed activation of thiono-sulfur-containing
compounds to reactive intermediates. Hoppe-Seyler's Z. physiol.
Chem., 357: 1044.
NESSWETHA, L. & NESSWETHA, W. (1967) The clinical evaluation of
compensated carbon disulphide intoxications in the German Federal
Republic since 1948. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Excerpta Medica Foundation,
Amsterdam, pp. 214-218.
NIOSH (1977) Criteria for a recommended standard...occupational
exposure to carbon disulfide, Washington, DC, US Dept Health,
Education, and Welfare, National Institute for Occupational
Safety and Health, 186 pp.
NOFER, J. & WRONSKA-NOFER, T. (1966) Elimination des certains
metabolites de niacine par rapport au témps dexposition et la
close sulfure de carbone. In: Proceedings of the IX
International Congress on Occupational Health, Vienna, 1966,
pp. 355-358 (A III 53).
NOFER, J., CHOJNOWSKI, J., KAWECKA, M., KIEC, E., WRONSKA-SZPAKOWA,
T., & WYSZOMIRSKA, Z. (1961) [Effect of occupational hazard to
carbon disulphide on the pathogenesis of arteriosclerosis.]
Med. Pr. 12: 101-118 (in Polish).
NORO, L. (1944) [Hydrogen and carbon disulphide intoxications.] Nord.
Med., 24: 2015-2020 (in Swedish).
NOVAK, L., DJURIC, D., & FRIEDMAN, V. (1968) Specificity of the
iodine-azide test for carbon disulphide exposure. I. Urinary
excretion of sulfur compounds in persons treated with disulfiram.
Arch. environ. Health, 19: 473-477.
NUNZIATE, CESARO, A., SIPALA, E., & ZUNINI, E. (1952) [Carbon
disulfide poisoning.] Folia Med., 35: 661-716 (in Italian).
NURMINEN, M. (1976) Survival experience of a cohort of carbon
disulphide exposed workers from an eight-year prospective
follow-up period. Int. J. Epidemiol., 5: 179-185.
OLIENACZ, W., PLAMIENIAKA Z., SZYMANKA, J. (1964) [Excretion of
17-ketosteroids in chronic carbon disulphide poisoning.]
Med. Pt. 15: 155-157 (in Polish).
PATERNI, L., PUSIC, G., & TEODORI, S. (1958) [Chronic carbon disulfide
poisoning and arteriosclerosis from high-cholesterol diet in
rabbits.] Folia Med., 41: 705-722 (in Italian).
PAULUS, H. J., LIPPMAN, M., & COHEN, A. E. (1957) Evaluation of
potential health hazards in fumigation of shelled corn with a
mixture of carbon disulfide and carbon tetrachloride. Am. Ind.
Hyg. Assoc. J., 18: 345-350.
PERGAL, M., VUKOJEVIC, N., GIRIN-POPOV, N., DJURIC, D., & BOJOVIC, T.
(1972a) Carbon disulfide metabolites excreted in the urine of
exposed workers. I. Isolation and identification of
2-mercapto-2-thiazolinone-5. Arch. environ. Health, 25: 38-41.
PERGAL, M., VUKOJEVIC, N., & DJURIC, D. (1972b) II. Isolation and
identification of thiocarbamide. Arch. environ. Health, 25:
42-44.
PERGAL, M. N., KOBILAROV, J., RALETIC, A., POSTICS-GRUJIN, A., &
DJURIC, D. (1977a) Determination of small quantities of
thiocarbamate in urine. Egypt. J. Decup. Med., 5: 251-257.
PERGAL, M., RALETIC, J., & DURIC, D. (1977b) Identification of the
third metabolite, giving positive iodine-azide reaction in the
urine of workers exposed to CS2. A preliminary report. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski, Finland, 6-10 June, 1977, p. 29.
PETERSON, F. (1892) Three cases of acute mania from inhaling carbon
bisulphide. Boston Med. Surg., 127: 325-326.
PETROV, M. V. (1969)] Course and termination of pregnancy in women
working in viscose industry.] Pediatr. Akush. Ginekol., 3:
50-52 (in Russian).
PETROVIC, D. & DJURIC, D. (1965) [Carbon disulfide in exhaled air of
exposed workers.] In: Zbornik radova o toksikologiji ugljen
disulfida, Loznica, pp. 35-38. (in Serbo-Croat).
PETROVIC, D. & DJURIC, D. (1966) [Carbon disulfide in the air exhaled
by exposed workers.] Arh. Hig. Rada, 17: 159-163 (in
Serbo-Croat).
PETRUN, N.M. (1967) [The effect of carbon disulfide on certain
biochemical indices of the status of the organism in its action
via the skin.] Gig. Tr. Prof. Zabol., 11: 50-53 (in Russian).
PEYTON, T. O., STEEL, R. V., MABEY, W. R., CARLIN, A., & PINES, A.
(1976) Literature review and environmental assessment of carbon
disulfide and carbonyl sulfide. Menlo Park, CA Stanford Research
Institute, pp. 1-65. (Project no EGU 3570, US Environmental
Protection Agency, Publication No. 257-947).
PIROTSKAJA, E. N. (1972) [Changes in the protein-forming function of
the liver in workers exposed to carbon disulfide.] Vrach. Delo,
9: 132-134 (in Russian).
PREROVSKA, I. & ROTH, Z. (1968) [An observation of personnel in the
carbon disulphide hazard with special reference to the
development of arteriosclerosis.] Prac. Lek., 20: 6-10 (in
Czech).
PREROVSKA, I. & ZVOLSKY, P. (1973) [The health status in workers with
chronic carbon disulphide poisoning in five to nineteen years
after ascertaining the disease.] Prac. Lek., 25: 42-46 (in
Czech).
PREROVSKA, I., VANECEK, R., & KUBAT, K. (1961) Effect of carbon
disulfide on experimental arteriosclerosis in the rabbit. Acta
Univ. Carol., 14: 177-185.
QUARELLI, G. (1928)] Occupational CS2 poisoning.] In: Bericht. d. 5.
Internat. Kongr. f. Berufskrankh., Budapest, 1928 (in Italian).
QUARELLI, G. (1934)] Occupational carbon disulfide poisoning.] Russ.
Providenza Soc., 21: 10-73 (in Italian).
RANELLETTI, A. (1931)] Occupational carbon disulfide poisoning.]
Arch. Gewerbepath. Gewerbehyg., 2: 664-675 (in German).
RANELLETTI, A. (1933)] Occupational carbon disulfide poisoning.] In:
La Grafica Moderna, Ed. Torino (in Italian).
RAITTA, C. & TOLONEN, M. (1975) Ocular pulse wave in workers exposed
to carbon disulphide. Albrecht. Graefes Arch. Ophthal., 195:
149-154.
RAITTA, C., TOLONEN, M., & NURMINEN, M. (1974) Microcirculation of
ocular fundus in viscose rayon workers exposed to carbon
disulfide. Albrecht. Graefes Arch. Ophthal., 191: 151-164.
RECHENSERG, H. K. (1957)] Carbon disulfide poisoning and the
sulfocarbotoxic vascular Late-syndrome.] Arch. Gewerbepath.
Gewerbehyg., 15: 487-530 (in German).
RUIKKA, I. (1959) Postprandial serum turbidity in chronic carbon
disulfide poisoning a comparison of viscose rayon workers with
healthy and atherosclerotic men. Ann. Med. intern. Fenn., 48:
225-237.
SAITA, G. & GATTONI, L. (1957) [The blood coagulation process in
chronic carbon disulfide poisoning.] Med. Lavoro, 48: 229-237
(in Italian).
SAITA, G., SBERTOLI, C., & FARINA, G. F. (1964) Modifications in blood
coagulation and lipid metabolism produced by carbon disulfide.
Med. Lavoro, 55: 721-734.
SAKURAI, H. (1972) Hypertension induced by the occupational exposure
to carbon disulfide. In: Abstracts of the XVII International
congress on occupational health. Buenos Aires, 11-16 September,
1972, p. 182.
SAVIC, S. M. (1967) Influence of carbon disulfide on the eye. Arch.
environ. Health, 14: 325-326.
SAVIC, S. & IOVICIC, M. (1965) [Keratoconjunctivitis (causome) in
workers employed in the production of viscose fibres.] In:
Zbornik Radova o toksikologiji CS2, Loznica, Yugoslavia 1965,
pp. 115-121 (in Serbo-Croat).
SAVOLAINEN, H. & VAINIO, H. (1976) High binding of CS2 sulphur in
spinal cord axonal fraction. Acta Neuropathol. (Berlin), 36:
251-257.
SAVOLAINEN, H., JÄRVISALO, J., & VAINIO, H. (1977a) Specific binding
of CS2 metabolites to microsomal proteins in rat liver. Acta
Pharmacol. Toxicol., 41: 94-96.
SAVOLAINEN, H., JÄRVISALO, J., ELOVAARA,. E., & VAINIO, H. (1977b) The
binding of CS2 in central nervous system of control and
phenobarbitone-pretreated rats. Toxicology, 7: 207-214.
SAVOLAINEN, H., LEHTONEN, E., & VAINIO, H. (1977c) CS2 binding to rat
spinal neurofilaments. Acta Neuropathol. (Berl.), 37: 219-223.
SBERTOLI, C., FARINA, G., & SAITA, G. (1969) [Three cases of chronic
carbon disulfide poisoning with giomerulonephrotic syndrome.[
Med. Lavoro, 60: 28-37 (in Italian).
SCHEEL, L. D. (1965) Biological changes involving metal ion shifts.
Am. ind. Hyg. J., 26: 585-592.
SCHEEL, L. D. (1967) Experimental carbon Disulfide poisoning in
rabbits-Its mechanism and similarities with human case report.
In: Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 107-115.
SCHEEL, L. D., KEENAN, R. G., MOUNTAIN,, J. T., KopP, J., HOLTZ, J., &
KILLENS, R. (1960) The effect of dietary metal ions on carbon
disulfide toxicity. In: Proceedings of the 13th International
congress on occupational health, New York, 1960, pp. 783-784.
SCHILLING, R. S. F. (1970) Coronary heart disease in viscose rayon
workers. Am. Heart J., 80: 1-2.
SCHNEIDER, H. (1976) In: Adverse effects of environmental chemicals
and psychotropic drugs. Neurophysiological and behavioural
tests. Vol. 2, Amsterdam, 1976.
SCHÜTZ, H. (1970) Equipment and systems for ambient air monitoring,
Frankfurt/Maine, Hartmann & Braun, Mess und Regeltechnik.
SEPPÄLÄINEN, A.M. (1977) Neurophysiological findings in workers
exposed to carbon disulphide-A follow-up. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, 6-10 June, 1977, p. 17.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (1975) Electrophysiological studies
of rabbits in long-term exposure to carbon disulfide. Scand. J.
Work Environ. Health, 1: 178-183.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (19745) Electrophysiological findings
in rats with experimental carbon disulphide neuropathy.
Neuropath. appl. Neurobiol., 2: 209-216.
SEPPÄLÄINEN, A.M. & TOLONEN, M. (1974) Neurotoxicity of long-term
exposure to carbon disulfide in the viscose rayon industry. A
neurophysiological study. Work Environ. Health, 11: 145-153.
SEPPÄLÄINEN, A.M., TOLONEN, M., KARLI, P., HÄNNÄINEN, H., & HERNBERG,
S. (1972) Neurophysiological findings in chronic carbon disulfide
poisoning. A descriptive study. Work Environ. Health, 9: 71-75.
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49
(in Japanese).
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49.
(in Japanese).
SIHTOLA, H. (1976). [Stages in the development of the SINI-viscose
process.] Tutkimusja tekniikka, 7/8: 63-69 (in Finnish).
SIHTOLA, H. (1977) The SINI viscose process means less chemicals and
less pollution. In; Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 43.
SKALICKA, B. (1967) [Sulfur-containing compounds after administration
of tetraethylthiuramdisulfde.] Prac. Lek., 19: 408-412 (in
Czech).
SOKAL, J. A. (1973) Effect of chronic exposure to carbon disulphide
upon some components of the electron transport system in rat
liver microsome. Biochem. Pharmacol., 22: 129-132.
SOUCEK, B. (1957) [Transformation of carbon disulfide in the
organism.] J. Hyg. Epidemiol. (Praha), 1: 10-22 (in German).
SOUCEK, B. (1959) [Absorption and elimination of carbon disulfide.]
Prac. Lek., 11: 403-407 (in Czech).
SOUCEK, B. (1960a) [Pulmonary elimination of carbon disulfide after
intraperitoneal administration.] Farmakol. Toksikol., 23:(1):
74-84 (in Russian).
SOUCEK, B. (1960b) [The effect of carbon disulphide on the serum
cholesterol and cholesterol ester level.] Prac. Lek., 12:
243-248 (in Czech).
SOUCEK, B. & MADLO, Z. (1953) [Absorption, metabolism and action of
carbon disulphide in the organism. VI. Attempt at chromatographic
proof of the reaction of carbon disulphide with aminoacids in the
blood.] Prac. Lek., 5: 309-311 (in Czech).
SOUCEK, B. & MADLO, Z. (1954) [Absorption, metabolism and action of
carbon disulphide in the organism. VIII. Reaction of carbon
trisulphide with blood in vitro.] Prac. Lek., 6: 11-17 (in
Czech).
SOUCEK, B. & MADLO, Z. (1955) Determination of sulfhydryl groups in
guineapig serum after exposure to carbon disulfide. Bull. Soc.
Chim. Biol., (Paris), 37: 1373-1378.
SOUCEK, B. & MADLO, Z. (1956) [Dithiocarbamin carbon acids as
degradation products of carbon disulfide.] Arch. Gewerbepath.
Gewerbehyg., 14: 511-521 (in German).
SOUCEK, B. & PAVELKOVA, E. (1953) [Absorption, metabolism, and effects
of carbon disulfide in the organism. IV. Long-term experiments.]
Prac. Lek., 5: 189 (in Czech).
SOUCEK, B., MADLO, Z., & HOLEYSOVSKA, H. (1957) [The influence of
carbon disulfide on proteolytic enzymes.] Arch. Gewerbepath.
Gewerbyhyg., 15: 531-538 (in German).
STRITTMATTER, C. F., PETERS, T., & MCKEE, R. W. (1950) Metabolism of
labelled carbon disulphide in guineapigs and mice. Arch. ind.
Hyg., 1: 54-64.
STYBLOCA, V. (1977) [Electroencephalography in the diagnosis of early
brain damage by carbon disulfide.] Int. Arch. occup. environ.
Health. 38: 263-282 (in German).
SUGIMOTO, K., GOTO, S., & HOTTA, R. (1976) An epidemiological study on
retinopathy due to carbon disulfide. Int. Arch. occup. environ.
Health, 37: 1-8.
SUGIMOTO, K., GOTO, S., TANIGUCHI, H., BABA, T., RAITTA, C., TOLONEN,
M., & HERNBERG, S. (1977) Ocular fundus photography of workers
exposed to carbon disulfide-A comparative epidemiological study
between Japan and Finland. Int. Arch. occup. environ.
Health, 39: 97-101.
SULKOWSKI, W. & LATKOWSKI, B. (1969) [Audiometric and
electronystagmographic examinations of workers in plants
producing synthetic fibers, who are suffering from chronic carbon
disulfide poisoning.] Pol. hyg. Lek., 24: 1563-1565 (in
Polish).
SZENDZIKOWSKI, S., STETKIEWICZ, J., WRONSKA-NOFER, T., & ZDRAJKOWSKA,
I. (1973) Structural aspects of experimental carbon disulfide
neuropathy. I. Development of neurohistological changes in
chronically intoxicated rats. Int. Arch. Arbeitsmed., 31:
135-149.
TABACOVA, S., HINKOVA, L., BALABAEVA, L., & KURCHATOVA, G. (1977).
Effects of carbon disulphide on the pre- and postnatal
development of rats. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 26.
TACCOLA, A., MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., CAVALLERI, A., &
DJURIC, D. (1971) [Vascular changes in human subjects exposed to
carbon disulfide.] Med. Lavoro, 62: 404-411 (in Italian).
TACCOLA, A., CAVALLERI, A., FRANCO, G., & MALAMANI, T. (1977) An
evaluation of the ratio of P-wave duration to P-R segment in ECG
of CS2 exposed workers. Egypt. J. occup. Med., 5: 309-312.
TAHARA, Y. (1961) [Hygienic evaluation of work environment of rayon
factories by carbon disulfide analysis of the expired air of
workers.] J. Sci. Labour (Tokyo), 37: 583-597 (in Japanese).
TARKOWSKI, S. (1973) [Glucose and free amino acid metabolism in the
brain of rats chronically poisoned with carbon disulfide.] Med.
Pr., 24: 571-575 (in Polish).
TARKOWSKI, S. (1974) Activity of glutamate decarboxylase in the brain
of rats exposed to carbon disulfide. Int. Arch. Arbeitsmed.,
33: 79-82.
TARKOWSKI, S. & CREMER, J. E. (1972) Metabolism of glucose and free
amino acids in brain, studied with 14C labelled glucose and
butyrate in rats intoxicated with carbon disulphide.
J. Neurochem., 19: 2631-2640.
TARKOWSKI, S. & SOBCZAK, H. (1971) Oxidation and phosphorylation
processes in brain mitochondria of rats exposed to carbon
disulphide. J. Neurochem., 18: 177-182.
TARKOWSKI, S. & WRONSKA-NOFER, T. (1966) [Effect of carbon disulphide
on the cytochrome oxidase in rats.] Med. Pr., 17: 375-379 (in
Polish).
TAZUKA, H. (1955) [A study on CS2 poisoning. Report II: On the
absorption of CS2.] J. Sci. Labour (Jpn), 31: 208-209 (in
Japanese).
TEISINGER, J. (1934) Mils central hemiparesis with extrapyramidal
signs in chronic poisoning. Cas. Lek. Ces., 73: 188-189.
TEISINGER, J. (1954) [Metabolism and detoxication of some industrial
poisons.] Praha, S.Z.N., p. 106 (in Czech).
TEISINGER, J. (1971) [Chronic carbon disulphide poisoning mechanism.]
Pracov. Ldk., 23: 306-308 (in Czech).
TEISINGER, J. & SOUCEK, B. (1949) Absorption and elimination of carbon
disulfide in man. J. ind. Hyg., 31: 67-73.
TEISINGER, J. & SOUCEK, B. (1952) [The importance of metabolism of
some toxic vapours considering their transmission and excretion
in man.] Cas. Ldk. Ces., 91: 1372-1375 (in Czech).
THIELE, H. & WOLF, I. (1976) [Results of complex clinical serial tests
on CS2-exposed workers.[ Z. ges. Hyg. DDR, 22: 603-608 (in
German).
TILLER, J. R., SCHILLING, R. S. F., & MORRIS, J. N. (1968)
Occupational toxic factor in mortality from coronary hear
disease. Br. med. J., 4: 407-411.
TINTERA, J., GRAOVAC-LEPOSAVIC, LJ., & MILIC, S. (1972) Excretion of
some tryptophan metabolites in man exposed to carbon disulphide.
med. Lavoro, 63: 126-133.
TOLONEN, M. (1974) Chronic subclinical carbon disulphide poisoning.
Work Environ. Health, 11: 154-161.
TOLONEN, M., HERNBERG, S., NURMINEN, M., & TIITOLA, K. (1975) A
follow-up study of coronary heart disease in viscose rayon
workers exposed to carbon disulphide. Br. J. ind. Med., 32:
1-10.
TOLONEN, M., HERNBERG, S., NORDMAN, C-H., GOTO, S., SUGIMOTO, K., &
BABA, T. (1976) Angina pectoris, electrocardiographic findings
and blood pressure in Finnish and Japanese workers exposed to
carbon disulfide. Int. Arch. occup. environ. Health, 37:
249-264.
TOLONEN, M., HANNINEN, H., & NURMINEN, M. (1978) Phychological tests
specific to individual carbon disulfide exposure. Scand. J.
Psychol., 19(3): 241-245.
TOYAMA, T. & HARASHIMA, S. (1962) {Industrial hygienic evaluation of
exposure to CS2 through analysis of biological materials.] Jpn.
J. ind. Health., 4: 81-85 (in Japanese).
TOYAMA, T. & KUSANO, H. (1953) [An experimental study on absorption
and excretion of CS2.] Jpn. J. Hyg., 8: 70 (in Japanese).
TOYAMA, T. & SAKURAI, H. (1967) Ten-year changes in exposure level and
toxicological manifestations in carbon disulphide workers. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 197-204.
TRUHAUT, R. R., BOUDENE, C., PHU-LICH, N., & BAQUET, A. (1972)
Memoires nouvelles recherches sur la mesure en continu de
l'exposition individuelle au sulfure de carbone dans l'industrie
a l'aide d'un appareil portatif. Arch. Mal. prof., 33: 341-346.
TURCEK, M. & MOLCAN, J. (1964) [Carbon disulphide psychoses.] Prac.
Lek., 16: 257-261 (in Czech).
UEHLINGER, E. (1952) [On carbon disulfide glomerulosclerosis.]
Schweiz. Z. Allg. Pathol., 15: 217-224 (in German).
VASAK, V. (1963) [Assessment of exposure of workers to carbon
disulphide vapours.] Prac. Lek., 15: 143-145 (in Czech).
VASAK, V. & KOPECKY, J. (1967) On the role of pyridoxamine in the
mechanism of the toxic action of carbon disulfide. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 35-41.
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1963) Assessment of exposure
of workers to carbon disulphide vapours. II. Application of the
iodine-azide reaction for the detection and estimation of carbon
disulphide metabolites in urine. Prac. Lék., 15: 145-149 (in
Czech).
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1969) The assessment of the
exposure of workers to carbon disulphide by the iodine-azide test
of Vasak, Vanecek and Kimmelova. (1963). In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 56-59.
VASILJEVA, I. A. (1973) [Effect of low concentrations of carbon
disulfide and hydrogen sulfide on the menstrual function in women
and on the estrous cycle in an experiment.] Gigi. Sanit., 38:
24-27 (in Russian).
VERTIN, A. (1973) In: Gas analysis instrumentation, London,
Macmillan Press Ltd.
VERTIN, P. G. (1966) [Dynamics of the elastase and the cholesterol and
phospholipid ratio with exposure to CS2.] Arch. Gewerbepath.
Gewerbyhyg., 22: 167-178 (in German).
VERTIN, P. G. (1977) An examination for cardiac/vascular diseases in
the Dutch viscose rayon industry. In: Tolonen, M., ed. Abstracts
of the IV International Symposium on Occupational Health in the
Production of Artificial Fibres, Helsinki and Valkeakoski,
Finland, 6-10 June, 1977, p. 7.
VESCE, C. A., COPPA, S., & FIMIANI, R. (1953) [Variations in urinary
17-ketosteroids in chronic poisoning from solvents.] Folia Med.
(Napoli), 36: 444-460 (in Italian).
VIDAAKOVIC, A. & ANDJELKOVSKI, A. (1965) [Haematological changes in
workers exposed to carbon disulfide.] In: Zbornik radova o
toksikologiji CS2, Loznica, 1965, p. 77-81 (in Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & ANDJELKOVSKI, A. (1965a) [A study of the
effects of carbon disulfide in the liver.] In: Zbornik radova o
toksikologiji ugljen disulfida, Loznica, 1965, pp. 71-75 (in
Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & DJORDJEVIC, V. (1956b) [Some changes in
the blood of rats in sub-acute carbon disulfide-poisoning.] In:
Zbornik radova o toksikologiji CS2 Loznica, 1965, pp. 83-87
(in Serbo-Croat).
VIGLIANI, E. C. (1946) [Chronic carbon disulfide poisoning. A report
on 100 cases.] Med. Lavoro, 37: 165-193 (in Italian).
VIGLIANI, E. C. (1954) Carbon disulphide poisoning in viscose rayon
factories. Br. J. ind. Med., 11: 235-244.
VIGLIANI, E. C. (1961) [Intoxication with carbon disulfide.] In:
Baader, E. W., ed. Handbuch der Gesamten Arbeitsmedizin, Urban
& Schwarzenberg, pp. 313-325 (in German).
VIGLIANI, E. C. & PERNIS, B. (1955) [Clinical and experimental
investigations on atherosclerosis caused by carbon disulfide.]
Arch. Gewerbepath. Gewerbehyg., 14: 190-202 (in German).
VIGLIANI, E., VISTINI, F., & MASPES, P. E. (1944) First observations
on myopathy from carbon disulfide. Med. Contemp., 10: 85-96.
VINOGRADOV, P. B. (1966) [New experimental findings for substantiating
the maximum permissible concentrations of carbon disulfide in
reservoir water.] Gig. i Sanit., 31: 13-17 (in Russian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., VISNJIC, V., MAUGERI, U.,
& REZMAN, I. (1966a) [Hematological findings in young workers
exposed to CS2. I. Fibrinolysis.] Med. Lavoro, 57: 667-676 (in
Italian).
VISCONTI, E., REZMAN, I., MAUGERI, U., VIDAKOVIC, A., & CAVALLERI, A.
(1966b) Hematochemical findings in young workers exposed to
carbon disulfide. II. The behavior of lipoproteins and the
"clearing factor". Med. Lavoro, 57: 677-683.
VISCONTI, E., VISNJIC, V., CAVALLERI, A., VIDAKOVIC, A., MAUGERI, U.,
& REZMAN, I. (1966c) [Haematological findings in young workers
exposed to carbon disulfide. III. Antiheparinic activity.] Med.
Lavoro, 57: 745-750 (in Italian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., REZMAN, I., MAUGERI, U., &
VISNJIC, V. (1967). Fibrinolytic activity in young workers
exposed to carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 128-132.
WAKATSUKI, T. & HIGASHIKAWA, n. (1959) Experimental studies on carbon
disulfide and hydrogen sulfide poisoning, histological changes in
hemopoietic organs and other internal organs. Shikoku Igaku
Zasshi, 14: 549-554.
WEAST, R. C. (1974) Handbook of Chemistry and Physics, 55th ed.,
Cleveland, CRC Press, pp. C-234-235; D-85; D-163.
WEIGMAN, H. J. (1973). [Measuring devices for automatic surveillance
of air pollution in work places.] Z. ges. Hyg., 19: 1-9 (in
German).
WILEY, F. H., HUEPER, W. C., & VON OETTINGEN, W. F. (1936) On the
toxic effects of low concentrations of carbon disulfide. J. ind.
Hyg. Toxicol., 18: 733-740.
WRONSKA-NOFER, T. (1969) The incorporation of 14C-acetate into
cholesterol in rats exposed to carbon disulphide. Biochem.
Pharmacol, 18: 925-926.
WRONSKA-NOFER, T. (1970) The influence of nicotinic acid upon the
disturbances in lipid metabolism caused by carbon disulphide in
rats. Int. Arch. Arbeitsmed., 27: 221-227.
WRONSKA-NOFER, T. (1976) Aortic phospholipids synthesis in
experimental carbon disulphide intoxication in rats. Int. Arch.
occup. environ. Health, 36: 229-234.
WRONSKA-NOFER, W., NOFER, J., & TARKOWSKI, S. (1965) [Impaired niacin
metabolite excretion in animals poisoned with carbon disulfide.]
Med. Pt., 16: 77-81 (in Polish).
WRONSKA-NOFER, T., TARKOWSKI, S., GORNY, R., SOKAL, J. A., & SZYC, M.
(1970) Accelerated turnover rate of nicotinamide adenine
dinucleotides in the liver of rats intoxicated with carbon
disulfide. Biochem. Pharmacol, 21; 2945-2950.
WRONSKA-NOFER, T., STETKIEWICZ, J., & SZENDZIKOWSKI, S. (1973)
Structural alterations and content of nicotinamine-adenine
nucleotides in skeletal muscle of rat in chronic experimental
carbon disulfi de intoxication. Int. Arch. occup. Health., 31:
125-134.
YAMAGATA, Y., YUDA, A., SUZUKI, K., NEMOTO, T., TAKAHASHI, M.,
TUCHIDA, H., SAITO, K., & KUSUNOKI, N. (1966) Carbon disulfide
nephroselerosis with special reference to the similarity to
diabetic glomerulosclerosis. J. Jpn. Diabetic. Soc., 9:
208-217.
YAROSLAVSKY, B. K. (1969) Toxic action of carbon disulfide on
reproduction and potentiation of this effect by tryptophan.
Bull. exp. Biol. Med., 68: 88-91.
YOSHIDA, K. (1955) [Fate of CS2 in the body-formation of thioketone
bonds.] J. Sci. Labour (Tokyo), 31: 209 (in Japanese).
ZEGLIO, P. (1942) Changes in gastric function due to chronic exposure
of workers in rayon industry. Med. Lavoro., 33: 217-228.
ZENK, H. (1967) The rhino-oto-vestibular symptomatoiogy of carbon
disulphide intoxication. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 190-191.
ZENK, H. (1970) [Carbon disulfide influences in the rhino-otological
functions of workers in the plastic fibers industry.] Int. Arch.
Arbeitsmed., 27: 210-220 (in German).
Annex I
PRODUCTION OF VISCOSE AND ITS END-PRODUCTS
The technological process of viscose rayon production, which is
schematically presented in Annex I Fig. 1, can briefly be described in
the following way:
(a) Cellulose pulp arrives at the factory in the form of sheets
which are placed in a hydraulic press or pulverized and steeped in a
solution of caustic soda to form alkali cellulose;
(b) the alkali cellulose is shredded to form crumbs to
facilitate aging and xanthation;
(c) after storage in aging tanks until the right level of
polymerization has been reached, the crumbs are placed in mechanical
churns;
(d) in a separate operation, sulfur is melted and poured onto
glowing charcoal where it reacts to give carbon disulfide. The liquid
carbon disulfide is stored in special tanks that are protected against
flames, static electricity, and heat;
(e) the liquid carbon disulfide, which is gradually metered
into the mechanical churns, reacts with the alkali cellulose to form
orange crumbs consisting of colourless cellulose xanthate and orange
sodium trithiocarbonate (Na2CS3);
(f) these crumbs are dissolved in caustic soda solution to give
a viscose solution;
(g) the viscose is stored in aging tanks, filtered, de-aerated,
and then pumped into the spinning tanks where it is forced through
spinnerets that are submerged in a solution of sulfuric acid (H2SO4)
and sodium and zinc sulfates (Na2SO4-ZnSO4). Filaments are formed
by coagulation of the viscose when partial decomposition of the
cellulose xanthate and the trithiocarbonate takes place, according to
the reactions:
(1) ROCS + H2SO4-ROH + NaHSO4 + CS2
SNa
(2) Na2CS3 + H2SO4 - H2S + CS2 + Na2SO4
During this process, considerable quantities of carbon disulfide
and hydrogen sulfide vapours are liberated representing a danger to
the exposed workers. It is important that reaction (2) should proceed
much more quickly than reaction (1) during spinning, so that
practically all the hydrogen sulfide is already formed in the spinning
bath or immediately over it, and the part of the carbon disulfide that
is supplied by the xanthate is mainly freed outside the spinning bath
in the acid yarn and/or in further treatments. The quantities of
carbon disulfide and hydrogen sulfide vapours that are liberated
depend on the process and the end-use of the rayon product.
The following products may be produced from viscose: rayon
filament textile yarn used in the textile industry in linings and
furniture fabrics; rayon filament tire yarn used as reinforcing
material in the carcass and belt of tires; rayon staple fibre
combined in textile fabrics with natural or synthetic fibres, such as
cotton or polyester; and Cellophane film, used for packaging.
On leaving the spinning bath, textile rayon yarn may be:
(a) wound up on acid spools on the spinning machine above the
spinning bath;
(b) formed into a cylindrical acid cake in a centrifugal pot
also incorporated in the spinning machine; or
(c) continuously transported through the regeneration, washing,
and drying treatments and then wound up as the finished product.
During spinning, the spinning machines are more or less
hermetically sealed. However, during doffing or when filament breaks
occur, the worker must open the "windows" or covers. At such times,
hydrogen sulfide and carbon disulfide vapours escape into the air. All
hydrogen sulfide is formed in the spinning bath or immediately over
it. Most of the carbon disulfide will be absorbed in the acid spool or
cake: and either be retained for a long time or be immediately freed
in a hot acid bath. Consequently, hydrogen sulfide will mainly be
found in the atmosphere of the machine and the acid baths, whereas
carbon disulfide also occurs in the departments where the acid spools
and cakes undergo further treatment (storage room, washing and drying
departments). In spool spinning, the worker has to lean over the
spinning bath to perform the doting and spinning-in operations. In
cake spinning, doffing takes place either at the front of the machine,
under conditions that are roughly the same as for spool spinning, or
at the rear of the machine, where the worker is not forced to breathe
in the machine atmosphere.
5.3.2 Nicotinic acid
Exposure to carbon disulfide resulted in increased excretion of
methyl nicotinamide, a nicotinic acid metabolite, in the urine of rats
(Liniecki, 1960; Wrofiska-Nofer et al., 1965). An increase in serum
lipids during the exposure of rabbits and rats to carbon disulfide was
prevented by administration of nicotinic acid (Nofer & Wrofiska-Nofer,
1966; Wrofiska-Nofer, 1970). The biochemical background of this
phenomenon is still obscure, but a hypothesis based on the
interference of carbon disulfide with pyridine nucleotide and
nicotinamide metabolism has been advanced (Nofer & Wrofiska-Nofer,
1966). However, feeding nicotinic acid to the exposed animals did not
prevent the development of neuropathies in these experiments.
As the increase in urinary excretion of nicotinamide metabolites
did not occur at the cost of the systematic pool of
nicotinamide-adenine dinucleotides, Wrofiska-Nofer et al. (1970)
suggested that it reflected an increase in the whole turnover rate.
The mechanism of the process was not clarified but it may be assumed
that an increase in the synthesis via tryptophan occurs.
5.4 Effects on Catecholamine Metabolism
Disturbance of the catecholamine metabolism can play a part in
many pathological processes. In studies on rats acutely intoxicated
with carbon disulfide, significant changes were found in the brain
catecholamine metabolism (Magos & Jarvis, 1970b; Magos, 1975). There
was a decrease in the level of noradrenaline accompanied by an
increased concentration of dopamine. Inhibition of dopamine
ß-hydroxylase, an essential enzyme in catecholamine metabolism, was
demonstrated in vivo in studies on rats by Magos (1975), and in
vitro by McKenna & Di Stefano (1975). Magos (1975) advanced a theory
concerning the central role played by catecholamine disturbances in
carbon disulfide pathology, especially where central nervous system
changes were involved, and in cardiovascular pathology. Cavalleri et
al. (1977) suggested that this could also explain the involvement of
the endocrine system.
5.5 Effects on Lipid Metabolism
Disturbance in the lipid metabolism has long since been linked
with carbon disulfide exposure. Increased levels of serum lipids, free
and total cholesterol, and ß-lipoproteins have been reported in
rabbits exposed to carbon disulfide (Paterni et al., 1958; Cohen et
al., 1959; Prerovska et al., 1961). Harashima et al. (1960) reported
elevated total and esterified cholesterol levels in the serum of
heavily exposed workers, whereas workers exposed to levels of about
15-60 mg/m3 (5-19 ppm) displayed normal serum cholesterol
concentrations. Elevated cholesterol levels were also found in exposed
workers by Manu et al. (1971) and in patients with previous exposure
to carbon disulfide (the patients had not been exposed for several
years) by Graovac-Leposavic et al. (1977). Higher levels of serum
lipids and especially cholesterol may be due to an increased rate of
synthesis in the liver and to the inhibition of the degradation of
lipids (Wrofiska-Nofer, 1969; Laurman & Wronska-Nofer, 1977). The fact
that elevated cholesterol levels have not been found consistently may
be explained by the different exposure levels in different studies
(Toyoma & Sakurai, 1967). Bittersohl & Thiele (1977) found that a
higher frequency of cholesterol values exceeding 6.72 mmol/litre
(260 mg/dl) in workers exposed to carbon disulfide was strongly
correlated with the duration of exposure.
A decreased clearing factor activity was found by Ruikka (1959)
in workers exposed to carbon disulfide and Martino et al. (1963, 1964)
reported an elevated ß-lipoprotein fraction in the serum of exposed
workers.
Changes in the lipid metabolism found in the aorta tissue may
contribute to the development of atheromatic changes in blood vessels
(Wrofiska-Nofer, 1976).
An increase in serum lipids during the exposure of rabbits to
carbon disulfide was prevented by administration of nicotinic acid
(Nofer & Wrofiska-Nofer, 1966) and in rats (Wrofiska-Nofer, 1970)
(section 5.3.2).
5.6 Interaction with Microsomal Drug Metabolism
An important feature of the liver toxicity caused by carbon
disulfide seems to be the destruction of cytochrome P-450 (Bond & De
Matteis, 1969). There is evidence that this effect is due to the
oxidative desulfuration of carbon disulfide by mixed-function oxidases
(De Matteis & Seawright, 1973). The resulting, highly reactive, sulfur
becomes covalently bound to the microsomal protein (Dalvi et al.,
1974; De Matteis, 1974; Jarvisalo et al., 1977), mainly to the
apoprotein of cytochrome P-450 (Neal et al., 1976; Jarvisalo & De
Matteis, 1977; Savolainen et al., 1977a). It is possible that the
liberated sulfur is the real toxic agent in liver toxicity arising
from carbon disulfide exposure.
Experiments on rats have shown that exposure to carbon disulfide
in concentrations up to 1250 mg/m3 (400 ppm) for 8 h is followed by
an increase in microsomal RNA content and in total protein in the
hepatic microsomal fraction and also increased incorporation of
2,4-3H-L-phenylalanine in liver microsomes (Freundt et al., 1974b).
Such changes follow the action of inducing agents, such as
phenobarbital, in the microsomal enzyme system. It has, therefore,
been suggested that carbon disulfide may have an inducing as well as
an inhibiting effect on mixed-function oxygenases. However, there is
no real evidence of such an effect at present (Freundt, 1977).
6. CARBON DISULFIDE POISONING
6.1 Historical Review
At the end of 1850, several physicians observed cases of strange
nervous and mental diseases, the origin of which remained obscure. In
1856, Delpech, reported 24 cases of carbon disulfide poisoning and
confirmed the diagnosis by animal experiments (Delpech, 1856a,b). In
1863, he reported 80 more cases of "carbon disulfide neurosis"
(Delpech, 1863). Cases of chronic poisoning were also reported in
England by Bruce (1884) and Foreman (1886). Laudenheimer (1899)
described carbon disulfide poisoning in German vulcanization shops,
stirring public opinion by drawing attention to about 50 cases of
"insanity". As further cases were reported in the USA (Jump & Cruice,
1904; Francine, 1905), the need for hygienic improvement in work
places was recognized. The first epidemic of carbon disulfide
poisoning due to the vulcanization process ended at the beginning of
the 20th century. At the same time, the viscose rayon industry started
to develop and expanded rapidly. Sporadic cases of carbon disulfide
poisoning in the viscose industry were reported between 1900 and 1930
(Quarelli, 1928) but the problem became serious in the 1930s.
Raneletti (1933), Quarelli (1934) and others described cases of
psychotic and polyneurotic disorders and extrapyramidal disturbances
(Audio-Gianotti, 1932; Teisinger, 1934).
In Japan, the viscose rayon industry was established in 1916 and,
in 1929, the first cases of carbon disulfide poisoning were reported
by Tokuhara, followed by other authors (review by Kubota, 1967). Many
cases of poisoning were also described in the USA (Hamilton, 1925,
1940; Bashore et al., 1938). The so-called Pennsylvania study by Gordy
& Trumper (1938) resulted in the establishment of the first TLV of
20 ppm adopted by the American Standards Association (1941). As the
hygienic standard in this industry improved, the incidence of severe
poisoning decreased. However, during World War II, the hygienic
situation in the expanding viscose industry deteriorated and severe
poisoning again became common.
During and after World War II, many cases of carbon disulfide
poisoning were reported, mainly from Italy (e.g., Vigliani et al.,
1944; Vigliani, 1946) but also from Belgium (Langelez, 1946; Merlevde,
1951) and Finland (Noro, 1944). After World War II, the viscose rayon
industry spread to many developing countries, where the whole sequence
of degrees of exposure was repeated. In the developed countries,
attention is now focused on slowly developing symptoms due to
long-standing exposure to relatively low concentrations.
6.2 Clinical Picture of Carbon Disulfide Poisoning
Carbon disulfide intoxication has been classified as hyperacute,
acute, subacute, and chronic.
Hyperacute poisoning occurs in extreme cases of massive exposure
for a short time to concentrations of about 10 000 mg/m3 or more. The
victim quickly falls into a coma and eventually dies. Acute and
subacute poisoning occurs with short exposure to carbon disulfide
concentrations ranging from 3000-5000 mg/m3 with predominantly
psychiatric and neurological signs and symptoms such as extreme
irritability, uncontrolled anger, rapid mood changes including maniac
delirium and hallucinations, paranoic ideas, and suicidal tendencies.
Other symptoms include memory defects, severe insomnia, nightmares,
fatigue, loss of appetite, gastrointestinal troubles, asthenia, and
interference with sexual functions, such as impotency.
The symptoms and signs of chronic carbon disulfide poisoning were
described by Vigliani (1946, 1961) and the following classification of
the different syndromes was suggested by Nesswetha & Nesswetha (1967):
-- psychoses characterized by manic and depressive symptomatology
and disorientation;
-- polyneuropathy of the lower extremities, with diminished or
completely absent Achilles and patellar tendon reflexes, sensory
disturbances in a glove-stocking distribution, diminished faradic and
galvanic excitability, and decrease of the motor and sensory
conduction velocity in the peripheral nerves;
-- disturbances of the gastrointestinal tract in the form of
chronic, hyper- and hypoacidic gastritis and duodenal ulceration;
-- myopathy of the calf muscles;
-- neurasthenic syndrome with disturbances in the autonomous
nervous system,
-- optic neuritis;
-- atherosclerotic vasculoencephalopathy; the principal forms
being bulbar-paralytic, hemiplegic, or extrapyramidal.
The typical mental deterioration has been called an organic
psycho-syndrome, which may be due to general cerebral atherosclerosis,
to direct toxic action upon the brain cells, or to both.
The pattern of carbon disulfide poisoning has changed with
improvements in hygienic standards in industry. However, when the
viscose industry is established in a country with no former
experience, there is always the risk of severe poisoning; this has
occurred many times.
6.3 Effects on Organ Systems
6.3.1 Dermatological effects
Liquid carbon disulfide represents a severe irritant for both the
skin and mucosa. Hueper (1936) found blisters in viscose rayon workers
and also in experimental animals that often resembled second and third
degree burns.
6.3.2 Ophthalmological effects
Studies on the ophthalmological effects of carbon disulfide in
animals have mostly been of a histological nature. In 1899, Koester
observed changes in the retinal ganglia. Seto (1958) found vacuolar
degeneration and tigrolysis in the retina, and atrophy of the optic
nerve in rabbits exposed to a carbon disulfide concentration of about
3700 mg/m3 for 1-3 h.
Ophthalmological examinations in man are informative because it
is possible to observe the visual capacity and to study the changes in
the vessels of the ocular fundus directly. According to the literature
(e.g., Nunziante-Cesaro et al., 1952; Savic, 1967) the following
effects have been observed:
-- changes in the motility of the eyelids;
-- changes in the sensitivity of the cornea and conjunctiva;
-- changes in the motility of the ocular bulbus;
-- changes in convergency and accommodation;
-- morphological changes in the fundus such as focal haemorrhage,
exudative changes, atrophy of the optic nerve, retrobulbar neuritis,
microaneurysms, and sclerotic changes of the blood vessels;
-- functional changes, e.g., disturbances of colour vision,
adaptation to the dark, reaction of the pupil to light, accommodation,
and decrease in visual accuracy.
Most of the effects mentioned above have resulted from heavy
exposure. Many that were observed decades ago, when the observations
often lacked comparison materials, are open to criticism. Nowadays,
under prevailing working conditions such grave changes are rarely, if
ever, seen.
Increased systolic and diastolic blood pressure in the retinal
arteries and damage to the arterial walls have been reported (Maugeri
et al., 1966d; Goto et al., 1971). Maugeri et al. (1966d, 1967)
studied the arterial pressure in 107 workers who had been exposed to
carbon disulfide at concentrations ranging from 200-500 mg/m3, with
peak concentrations of up to 900 mg/m3, for 1-9 years. Using an
ophthalmodynamographic method, the authors found an average increase
in both systolic and diastolic pressures in exposed workers of about
18.4/14.7 kPa (138/110 mmHg) compared with controls which averaged
15.3/11.6 kPa (115/87 mmHg). The increase was more marked for the
diastolic pressure than for the systolic. Measurement of the pressure
of the ophthalmic arteries may be a test that needs further evaluation
in the light of the demonstrated disturbance of the brain
catecholamine metabolism induced by carbon disulfide.
Savic (1967) studied changes in the nervous system of the eyes of
young workers exposed to carbon disulfide levels of 100-400 mg/m3.
Changes were observed but only after a long period of exposure
(>5 years) (Hotta & Savic, 1972). Retinal microaneurysms have been
reported in a high proportion of Japanese viscose rayon workers (Goto
& Hotta, 1967; Goto et al., 1971; Sugimoto et al., 1976). In one
study, the prevalences of microaneurysms as judged by fundus
photography, were 8% and 2% among exposed and unexposed male workers,
respectively (Goto & Hotta, 1967). In a subsequent study (Hotta &
Goto, 1971; Goto et al., 1971), a prevalence of 56% of microaneurysms
was found in the exposed group (241 subjects) and 15% in a control
group (30 subjects). The extremely high prevalence in the exposed
groups in these studies was due to biased selection of the subjects;
one-half of them were selected for examination because of known
microaneurysms revealed by ophthalmoscopy. Later studies by the same
team did not show such a high prevalence (Sugimoto et al., 1976), but
a statistically significant difference between exposed and unexposed
subjects still persisted. Retinaopathy was found in 89 out of 289
carbon disulfide workers (31%), but only in 2 out of 49 controls (4%).
The frequency of retinopathy was correlated with both the duration and
the intensity of exposure (Sugimoto et al., 1976). It has been
suggested that the retinal microaneurysms found in Japanese workers
were related to the diabetogenic action of carbon disulfide (Goto et
al., 1971). However, microaneurysms are generally associated with
hypoxia and, though they may occur in advanced cases of diabetes, they
are not, by any means, specific for the disease. Thus, to ascribe the
excess of aneurysms found in Japanese studies to subclinical diabetes
caused by carbon disulfide is not justified. It is more likely that
they were caused by a direct effect on the retinal vessels, possibly
in association with local hypoxia coacting with ethnic or
environmental factors.
The results of recent Finnish studies do not tally with the
Japanese findings. Out of 100 exposed men and 97 controls, only 3
exposed and 2 control subjects showed 1-5 microaneurysms, and one
exposed subject showed more than 5 (Raitta et al., 1974). Furthermore,
general narrowing of the arteries and calibre irregularity were so
common in the fundus of both the exposed and control subjects that
plain ophthalmoscopy did not have any discriminatory value in this
study. A subsequent Japanese-Finnish study showed that the differences
in values obtained in the 2 studies were true differences and were not
caused by inter-observer variation (Sugimoto et al., 1977).
The most interesting finding in a study by Raitta et al. (1974)
was the high frequency of delayed peripapillary filling that occurred
in 68 exposed and 38 unexposed men. The findings of Raitta & Tolonen
(1975), suggest that studying the microcirculation of the ocular
fundus by fluorescein-angiography and oculosphygmography would help in
the early detection of carbon disulfide effects, especially since the
vessels can be observed directly in their natural state.
Ophthalmoscopy is indicated for the examination of patients suspected
to be suffering from carbon disulfide poisoning and for following-up
such patients, but it is too prone to inter-and intra-observer
variation to be used routinely by plant physicians for regular health
checks. Fundus photography eliminates some of this variation, but its
evaluation should be standardized. The diagnosis of retinal
microaneurysms requires a fundus angioscreenography, but this method
is recommended for research purposes only.
It should be added that a mixture of carbon disulfide, hydrogen
sulfide and sulfuric acid mist from an acid bath caused
keratoconjunctivitis in exposed workers (Savic & Jovicic, 1965). This
phenomenon is called "spinner's eye".
6.3.3 Otological effects
Sulkowski & Latkowski (1969) observed that exposure to carbon
disulfide impaired hearing ability. The authors examined 60 workers
occupationally exposed to carbon disulfide for between one year and
more than 10 years. The workers were below 50 years of age.
Audiometric examinations showed impairment of hearing of the receptory
type in more than 50 workers and also a decreased ability to
distinguish sound intensity within the range 1.5-3.0 dB. This
suggested a central and supracochlear localization of the impairment.
Electronystagmographic examinations performed by these authors
disclosed reduced excitability of the vestibular apparatus, suggesting
an extralabyrinthine localization of the lesion. Loss of sensitivity
to high frequency tones has also been attributed to carbon disulfide
exposure (Zenk, 1970). Since noise levels are often very high in
viscose rayon plants, it is possible that the lesions found may be at
least partly due to this cause. Vestibular symptoms, as manifested by
vertigo and nystagmus may also be present in carbon disulfide
intoxication (Zenk, 1967, 1970).
6.3.4 Respiratory effects
Carbon disulfide is a known irritant, but few data exist about
its effects on the respiratory system (Zenk, 1967). Ranelletti (1933)
described chronic cough as a consequence of the irritant effect of
carbon disulfide, but it is necessary to take into consideration the
irritative effects of hydrogen sulfide and sulfuric acid mist and of
other irritants present in the air of viscose rayon plants. The
findings of Massoud et al. (1971) i.e., cough, phlegm, wheezing,
dyspnoea, precordial pain and palpitations, may also have been due to
such mixed exposure rather than to carbon disulfide alone.
6.3.5 Gastrointestinal effects
Gastrointestinal symptoms are common among heavily exposed
workers and patients with carbon disulfide poisoning. Bashore et al.
(1938) found a prevalence of such symptoms of 25%, Vigliani (1954),
28%, Karajovic et al. (1964), 66%, Lysina (1967), 27%, and Hass et al.
(1967), 27%. Since such symptoms occur among unexposed people too,
especially in shift work, the figures listed cannot be attributed
exclusively to carbon disulfide exposure (Knave et al., 1974). A
higher prevalence of gastrointestinal disorders and liver and bile
duct dysfunction was observed in 2 groups of workers (800 and 492
subjects, respectively) exposed to very low carbon disulfide
concentrations (4-12 mg/m3) than in a control group of 453 unexposed
workers. The prevalence in the 2 exposed groups was 4.7 and 5.6%,
respectively, and that in the control group, 2.2% (Murashko, 1975).
Later, Bittersohl & Thiele (1977) found a prevalence of 22.7% of
gastrointestinal changes in 309 exposed shift workers as opposed to
5.9% in 345 unexposed shift workers.
Based on histological studies of the gastric mucosa of 75 workers
exposed to carbon disulfide, Hassman et al. (1967) suggested
classification into 5 groups: normal gastric mucosa, superficial
gastritis, chronic gastritis with incipient atrophy, chronic atrophic
gastritis, and gastritis of undetermined type. Duodenal ulceration was
only found in 2 out of 75 subjects in this study. Of the workers
examined, 11% had dyspeptic complaints and gastritis was verified in
60% of the cases. A similar figure (66%) was found by Karajovic et al.
(1964).
6.3.6 Hepatic effects
Exposure to carbon disulfide has caused fatty degeneration and
haemorrhages of the liver in animals (Bashore et al., 1938).
Experimental studies on rats have shown that pretreatment with a drug
that stimulates liver microsome enzyme activity alters the degree of
liver damage on exposure to carbon disulfide. Thus, Bond et al. (1969)
reported necrosis in the livers of rats that had been pretreated with
phenobarbital and subsequently received a single oral dose of carbon
disulfide (1 ml/kg body weight) but not in rats treated only with
carbon disulfide. Moreover, only pretreated rats displayed hydropic
degeneration of the liver. Magos & Butler (1972) found that starvation
potentiated the effect of phenobarbital in rats subsequently treated
with carbon disulfide, and that the resulting hydropic degeneration
was reversible. Similar results were obtained by Freundt et al.
(1974a), when they administered a single oral dose of 1 ml of carbon
disulfide per kg body weight to rats pretreated with phenobarbital.
However, no degenerative changes in the liver were observed in
pretreated rats following exposure by inhalation to carbon disulfide
at about 60 mg/m3 (20 ppm) and 620 mg/m3 (200 ppm) for up to 7 days.
In experiments performed on rats with short-term (8-h) exposure to
carbon disulfide at concentrations ranging from 62-1250 mg/m3
(20-400 ppm), the energy potential of the organism was damaged mainly
because of a reversible augmentation of hepatic glycolysis (Kürzinger
& Freundt, 1969; Freundt & Kürzinger, 1975).
The possible effects of carbon disulfide on the liver have been
discussed in numerous reports of clinical observations. Some authors
deny that exposure to carbon disulfide results in toxic effects in the
liver, some claim degenerative changes of the hepatocytes, and others
state that sclerotic changes are produced under conditions of chronic
exposure. Such controversial observations can be explained by
nonuniformity in approach to the method of examination. Vidakovic et
al. (1965a) examined workers hospitalized because of chronic carbon
disulfide poisoning. The exposure of these workers had been extremely
high, ranging from 1400-2200 mg/m3 (Petrovic & Djuric, 1965).
Functional disturbances of the liver and fatty degeneration of
hepatocytes were found but no necrotic changes were observed.
Pirotskaja (1972) examined the protein-forming function of the
liver in workers (40 women and 6 men) who had been exposed to carbon
disulfide for 5 years. At first, the workers were exposed to a carbon
disulfide concentration of 90 mg/m3 but later, concentrations ranged
from 8-14 mg/m3. All exposed subjects were between 30 and 40 years of
age. The mean concentrations on cystine, thyroxine, methionine,
valine, leucine, and tryptophan in serum showed statistically
significant increases in comparison with a control group.
Statistically valid decreases occurred in the levels of glutamic acid,
aspartic acid, olanine, and lysine. The levels of histidine, arginine,
and serine did not differ from those in the control group. There was a
correlation between these changes and the clinical signs of carbon
disulfide intoxication. There was no difference in the serum contents
of total protein in the exposed and control groups.
6.3.7 Renal effects
Some authors have drawn attention to nephrosclerosis in autopsies
of patients with carbon disulfide poisoning (Uehlinger, 1952; Yamagata
et al., 1966; Sbertoli et al., 1969), but this could be ascribed to a
general atherosclerotic process induced by carbon disulfide. For
example, in the cases studied by Uehlinger (1952), typical
glomerulosclerosis of the Kimmelstiel-Wilson type was established in 4
patients, while the fifth showed arteriosclerotic injury of the
kidney. Obviously renal involvement represents a very late consequence
of heavy, protracted carbon disulfide exposure, since
Graovac-Leposavic & Jovicic (1971) reported that only one patient with
persistent kidney insufficiency had ever been diagnosed in a large
viscose rayon plant employing several thousands of workers. In another
study of viscose rayon workers, Hemberg et al. (1971) found a slight
but statistically significant rise in the mean plasma creatinine
concentration compared with a control group. All values were within
"normal" limits, however.
6.3.8 Haematological effects
Brieger (1949) studied the bone marrow of exposed rats and found
retarded maturation of the erythrocytes. Mild anaemia, a slight
decrease in the haemoglobin concentration, slight reticulocytosis,
eosinophilia, and hypercoagulability of the blood have also been
recorded in exposed rats (Vidakovic et al., 1965b).
Ivanova (1967) found a moderate decrease in the haemoglobin
concentration and erythrocyte count in men with only slight exposure
to carbon disulfide. Such results cannot be attributed to carbon
disulfide effects, but rather to poor standardization of the study
conditions, since significant haematological changes in the peripheral
blood were not observed, even in highly-exposed workers (Vidakovic &
Andjelkovski, 1965; Fahim et al., 1973). Thus, it has not been
confirmed that carbon disulfide causes anaemia or polyglobulia. It is
probable that the anaemia mentioned in some clinical reports reflects
malnutrition or some other nonoccupational cause, perhaps even
defective study design, rather than an effect of carbon disulfide.
The effects of carbon disulfide on blood coagulation mechanisms
are discussed in section 6.3.14.
6.3.9 The endocrine system
Damage to the endocrine structures with functional alterations
was described in animals by Ranelletti as early as 1931, and by
Audio-Gianotti in 1932. Impaired sexual function in patients with
carbon disulfide poisoning was reported by Gordy & Trumper (1938),
Langelez (1946), and Vigliani (1946). Vesce et al. (1953) observed a
decrease in 17-ketosteroids excretion in the urine of exposed rabbits.
This phenomenon was confirmed in studies on exposed workers by
Fruscella (1962) and Olienacz et al. (1964). Most of these
observations were made on workers belonging to older age-groups and
the confounding effect of age was not controlled. Cavalleri & Zuccato
(1965), however, obtained the same results taking the age factor into
account.
In 1965, a joint Italian-Yugoslav group began an investigation on
young workers in a Yugoslav viscose rayon plant. The workers were
classified according to duration of exposure to carbon disulfide. The
average ages of various groups ranged from 26 to 33 years and they
were exposed to average carbon disulfide concentrations of about
200-500 mg/m3 with peak concentrations of up to 900 mg/m3. The
excretion of 17-ketosteroids in the urine showed a linear decrease
with the duration of exposure (Cavalleri et al., 1966a,b, 1967). The
decrease in excretion of 17-hydroxycorticosteroids was most marked in
workers with short exposure and did not decrease any more as exposure
continued. The excretion of androsterone appeared to decrease
progressively with duration of exposure, probably in a linear fashion;
that of etiocholanolone did not show any uniform pattern (Cavalleri et
al., 1966c,d). Urinary excretion of testosterone and gonadotropin
luteinizing hormone was also reduced in workers exposed to
concentrations of 100-400 mg/m3 for 2-12 yearsa. Lancranjan et al.
(1971) reported a decrease in 17-ketosteroid and
17-hydroxycorticosteroid excretion in the urine of exposed workers.
The thyroid function has been studied in exposed workers by
determination of serum thyroxine (Cavalleri et al., 1971; Cavalleri,
1975; Massoud et al., in press). Decreases reported in thyroxine
levels suggest a reduction of thyroid activity.
Mild hypothyroidism was reported in 8% of exposed workers in
studies by Lancranjan (1972). This effect may be primary or may be due
to inhibition of the hypothalamus-hypophysis axis. A decrease in
thyrotropin-stimulating hormone, observed in human subjects given
tetraethylthiuram, favours the hypothesis of an impairment of the
hypothalamic-hypophyseal system (Cavalleri et al., 1977).
a Exposure data submitted by Professor Cavalleri as private
communication to the Task Group.
When the thyroid function is inhibited, disturbances in the lipid
metabolism will appear. A well-known manifestation of subclinical
hypothyreosis is elevation of the serum cholesterol level, which in
turn may contribute to the vascular changes typical of carbon
disulfide poisoning. The isolation and identification of two
metabolites, thiourea and mercaptothiazoline, may help to explain the
mechanism (Pergal et al., 1972a,b). Neither of these metabolites is
directly toxic, but both may have some influence on thyroid activity.
In fact, drugs containing substances of the thiourea and
mercaptothiazolinone type are used in the treatment of
hyperthyroidism.
Lancranjan et al. (1969, 1972), reported hypospermia,
asthenospermia, and tetratospermia in the spermatic liquid of young
exposed workers, confirming the gonadal injury reported by Cavalleri
et al. (1965).
Disturbances were found in the ovarian function of 500 young
females, exposed to concentrations of carbon disulfide of around
20 mg/m3, and of 209 women exposed to a concentration of less than
10 mg/m3 (Vasiljeva, 1973). In a comparison with a group of 429
unexposed women, menstrual flow durations of more than 5 days occurred
in 18% of the first group and in 11% of the second compared with 5% in
the control group. Irregular menstruation occurred in 8% of those
exposed to 20 mg/m3 as opposed to 2% in the comparison group. Other
menstrual disorders followed a similar pattern, and were correlated
with the length of exposure. When vaginal smears were examined from
subsamples, cellular disturbances were most frequent in the group with
highest exposure. A biochemical study of the sex hormones in the urine
confirmed the vaginal-smear findings. Hence, even rather low exposure
to carbon disulfide appeared to induce hormonal and functional
disturbances in young women. Other studies corroborate these findings
(Petrun, 1967, Finkova et al., 1973).
In a study of 380 women exposed to around 30 mg/m3, Petrov
(1969) found more pregnancy complications than in a control group of
191 women. Spontaneous abortions occurred in 14% of the exposed, and
7% of the unexposed women. "Threatened pregnancy terminations" also
occurred more frequently in the exposed women than in the controls
(26% and 13% respectively). Exposed women gave birth prematurely
significantly more frequently than controls (9% and 3%, respectively).
It has been reported in several studies that carbon disulfide
produces primary damage at the hypothalamic-hypophyseal level
(Cavalleri et al., 1965; Lancranjan et al., 1971; Maugeri et al.,
1971; Cavalleri, 1972). The hypothalamus produces the releasing
factors that stimulate the hypophysis to synthesize and release
polypeptide hormones, which in turn stimulate the target glands in
question. The impairments found would, therefore, represent a
secondary consequence.
Thus, carbon disulfide causes several disturbances of the
endocrine systems that can be summarized as follows:
-- a reduction of adrenal activity that can be repaired by ACTH
administration and can therefore be ascribed to reduced secretion of
corticotrophin;
-- a reduction of the endocrine activity of the testis and
impairment in spermatogenesis possibly due to direct gonadal damage;
this finding could explain the decrease in "libido" and "potentiae"
often found in exposed workers;
-- disturbances in the hormonal balance in women, resulting in
menstrual irregularities, spontaneous abortions, and premature births;
-- impairment of thyroid function, which could be primary or due
to a deficiency in the thyrotropin-stimulating hormone (TSH) or the
TSH releasing factor or both.
6.3.10 Effects on the nervous system
6.3.10.1 Central nervous system
In experimental animals, carbon disulfide causes destruction of
the myelin sheet and axonal changes in both the central and peripheral
neurons. Degenerative changes have been observed in the cortex, basal
ganglia, the thalamus, the brain stem, and the spinal cord (Wiley et
al., 1936; Ferraro et al., 1941; Lewey et al., 1941; Fischer &
Michalova, 1956; Drogicina, 1968).
The mechanism by which carbon disulfide causes these changes has
not been elucidated, however. Mitochondrial enzymes may be inhibited
(Tarkowski & Wrofiska-Nofer, 1966; Tarkowski & Sobczak, 1971), and the
tyrosine and catecholamine metabolism may be disturbed (Magos et al.,
1974). The content of free glutamine increases as a result of carbon
disulfide exposure (Efremova & Uzbekov, 1968; Tarkowski & Cremer,
1972). Such changes were observed with acute experimental poisoning,
but chronic, repeated exposure did not produce gross changes
(Tarkowski, 1973).
Horvath & Mikiska (1957) followed the electroencephalographic
changes in rabbits exposed to carbon disulfide and found a transitory
decrease in the amplitude of basic and superimposed beta rhythms.
The results of animal experiments have to be extrapolated to man
with extreme caution; however, many data obtained from animal studies
are in good agreement with clinical observations in man. Like other
narcotic agents, carbon disulfide produces a clinical symptomatology
of irritation and excitation on one hand, and inhibited psychomotor
activity, psychological alterations, insomnia, hypersomnia, loss of
consciousness and death, on the other.
In acute poisoning, the first typical signs from the central
nervous system (CNS) are excitation, euphoria, and aggressive
behaviour. Subacute and chronic cases are first characterized by
neurotic signs of unrest, excitation, and loss of temper. Gradually
the patient becomes depressive, anxious, paranoiac and sometimes there
is a suicidal tendency. Nightmares, apathy, loss of initiative,
vegetative disturbances, and headache are common symptoms (Melissinos
& Jacobides, 1967; Mihail et al., 1968). In the further course of
chronic intoxication, neurological signs become prominent. Both
diffuse cortical and focal symptoms from the subcortical grey matter
and extrapyramidal system are typical (Teisinger, 1934). Typical
Parkinson symptomatology with bradykinesia, bradylalia, muscle
rigidity, tremor, and increased elementary postural reflexes is
similar to that in arteriosclerotic or postencephalitic parkinsonism
(Nakamura et al., 1974). Psychic, pyramidal and extrapyramidal
symptomatology, including signs from other parts of the brain
(vestibular, cerebellar) give a picture compatible with diffuse
effects, described under the term of "toxic encephalopathy".
Furthermore, nervous involvement in general may be reflected as
changes in the sensitive branch of the trigeminal nerve, in the
function of the III, IV, and VI cranial nerves, and in the sympathetic
and parasympathic nerves of the eye.
Disturbances in cerebral circulation probably explain some of the
manifestations of neurotoxicity. Vigliani et al. (1944) demonstrated
hyalinosis of the arterioles and precapillaries histologically, a
finding that fits into the picture of general atherosclerosis caused
by carbon disulfide. Thus, the vascular changes described by Vigliani
(1961) as "encephalovasculopathia sulfocarbonica", are probably
responsible for many of the manifestations of central nervous system
pathology.
On the other hand, authors of some recent publications have tried
to explain some mechanisms by direct interference of carbon disulfide
and its metabolites in the system of enzymatic processes in the brain.
Morphological studies indicate that long axons in the spinal cord are
preferentially destroyed in chronic carbon disulfide poisoning
(Szendzikowski et al., 1973).
The typical neuropathological picture includes an increase in
neurofilaments, and secondary effects on the myeline sheath (Juntunen
et al., 1974). Biochemical studies have shown that oxidative
desulfuration also take place in the brain (Savolainen et al., 1977b),
and that the highest specific binding of the liberated sulfur is
detected in spinal cord axons (Savolainen & Vainio, 1976). It appears
that most of the liberated sulfur binds to neurofilaments that are
essential to normal axon functions (Savolainen et al., 1977c). Thus,
there is evidence of a direct toxic action upon the nervous system.
The results reported by Abramova (1967), Vasak & Kopecky (1967),
and others (section 5.3.1) regarding inhibition of the vitamin B6
metabolism, might explain some changes in the peripheral myelin sheet
and even some effects on the central nervous system. The consequences
of vitamin B6 inhibition, or interference with the transamination of
glutamic acid and inactivation of pyridoxamine could, perhaps, produce
irritative cortical symptomatology, including epileptiform cramps.
Furthermore, studies showing interactions between carbon disulfide
and, tissue respiration; depression of cytochrome oxidase, succinic
dehydrogenase, alkaline phosphatase, and dopamine-ß-hydroxylase; and
influence of inhibitors of serum elastase (EC 3.4.21.11), suggest that
besides a purely vascular etiology, it is plausible to consider a
direct toxic interference of carbon disulfide with tissue metabolism
(section 5.2). In fact, direct toxic effects of carbon disulfide on
peripheral nerve cells (Szendzikowski et al., 1973), and on the
myoneural junctions (Juntunen et al., 1977) have been shown in
animals. The central nervous system may react in the same manner.
From all these considerations, it can be concluded that the
effects of carbon disulfide on the nervous system may be influenced by
many factors. The method most often used for the study of cerebral
involvement in man is electroencephalography (EEG). However, studies
on carbon disulfide poisoning are rare. Early studies emphasize the
frequent occurrence of very flat records among subjects chronically
exposed to carbon disulfide (Krolikowska & Rucinska, 1959). More
recently, Seppäläinen & Tolonen (1974) found 21 abnormal EEGs in an
exposed group comprising 54 viscose workers compared with only 6 in a
control group of 50 paper-mill workers. Thiele & Wolf (1976) found EEG
changes in 52% of a group of 139 spinning workers. The abnormalities
consisted of slight, diffuse slow-wave abnormalities, slight to
moderately severe focal slow-wave abnormalities and even spike and
wave discharges in 3 exposed men. Thus, mild brain dysfunctions were
clearly more prevalent in the exposed group than in the control. EEG
findings among 250 workers with longstanding occupational exposure to
carbon disulfide (mean exposure 11.2 years) were recently described by
Styblva (1977) and compared with the EEGs of 61 healthy controls and
47 patients suffering from cerebral arteriosclerosis. Increased
frequency of abnormal EEGs was found in 33.2% of the exposed workers,
compared with 6.6% in the control group ( P < 0.01). The most
frequent abnormalities among the exposed workers were episodic theta
activity (33%) and diffuse abnormality (30%) (it was not, however,
indicated or even deducible from the data presented, how these
percentages had been calculated, i.e., from which population they were
taken). Local abnormalities were rare. Local EEG abnormalities
predominated in patients with cerebrovascular disease. The episodic
activity in the EEG was considered to reflect the direct toxic effects
of carbon disulfide on mesodiencephalic structures. The author
proposed that diffuse abnormality could be a manifestation of
vasculoencephalopathy resulting from toxic changes in the small
diameter vessels. The frequency of alpha activity tended to be lower
among exposed workers and this was considered to indicate a cerebral
metabolic disorder due to carbon disulfide exposure.
Studies on the behavioural and psychological effects of carbon
disulfide exposure have indicated disturbances in deep subcortical
structures, namely vegetative centres, and more diffuse cortical
involvement. The methods used have included diagnostic interviews,
behavioural observations (mainly on experimental animals), and
psychological test batteries.
Both structured and unstructured interviews of human subjects
showed differences in the clinical pictures of carbon disulfide
poisoning at rather high concentrations i.e., about 950-2400 mg/m3
(300-760 ppm) and at much lower concentrations i.e., around
20-50 mg/m3 (7-30 ppm) over a long period of exposure. At high
concentrations, symptoms of headache, muscle pain, fatigue,
paraesthesia, and general weakness were consistently reported (Gordy &
Trumper, 1940; Manu et al., 1970). At lower levels of exposure, fewer
complaints of muscular pain and overall weakness were noted, the main
symptoms being headache, memory impairment, rapid mood changes,
insomnia, paraesthesia, and fatigue (Seppäläinen et al., 1972; Lilis,
1974).
Standardized batteries of tests yield results that are directly
quantifiable and comparisons can be made between groups at different
exposure levels and control groups. Using this method, Hänninen (1971)
presented 3 groups of workers with different exposures and clinical
profiles (unexposed, exposed without clinical symptoms, and poisoned).
The results suggested that syndromes of latent and manifest carbon
disulfide poisoning differ in quality as well as in intensity, i.e.,
"clinically manifested poisoning is characterized by lowered
vigilance, diminished intellectual activity, diminished rational
control, retarded speed, and motor disturbances, whereas traits
indicative of depressive mood, slight motor disturbances, and
intellectual impairment are characteristic of latent poisoning"
(Hänninen, 1971). Analysis of intercorrelations among test sources for
each of the 3 groups resulted in substantially different factor
structures across the 3 groups. This supports the contention that
behavioural differences between groups exposed to different
concentrations are qualitative as well as quantitative (Hänninen,
1974). Naturally, the extent to which such distinctions are, in fact,
true, depends on the number of false-positive diagnoses in the
poisoned group (i.e., how many patients with other diseases had been
classified as cases of carbon disulfide poisoning), and, since this
aspect could not be evaluated, it may be premature to accept the
qualitative changes found as real facts. As to the quantitative
changes, it is self-evident that their severity must be correlated
with the degree of poisoning, at least on a group basis. The
specificity of the psychological and behavioural examination increases
when other sub-clinical signs of carbon disulfide exposure, such as
EEG and EMG findings are present.
Hänninen & Mäenpää, as reviewed by Tolonen (1974), applied the
same type of tests to 97 exposed male viscose rayon workers (mean
exposure time 15 years) and 96 age-and-sex matched controls from a
paper mill with no history of exposure to carbon disulfide. The most
distinct difference was in the retardation of psychomotor speed, where
the relative risk was 2.2 ( P = 0.02). As a whole, 40% of the exposed
and 25% of the control group showed "poor" performance, as defined
according to criteria published by Tolonen (1974). These results show
a clear group difference; however, the test battery has a low
specificity for carbon disulfide, as could be seen from the high
number of "false positive" results.
Recently, Hänninen and her co-workers increased the specificity
of the test battery, reducing the number of tests on the basis of
information gathered from new analytical approaches applied to results
of 206 exposed and 152 unexposed men, examined in 1972. According to a
multiple discriminant analysis, the most specific variables for carbon
disulfide effects were those indicating retarded speed, retarded
emotionality and energy, and psychomotor disturbances (Hänninen et
al., 1978). Another approach, starting from test results classified as
abnormal, resulted in similar conclusions (Tolonen et al., 1976). Now,
the test battery can be restricted to a few rather specific tests
applicable to suspected cases of poisoning that can also be used at
periodic health examinations.
Analogous results concerning the depressing effects of carbon
disulfide on psychological performance and behaviour have been
reported by Foa et al. (1976), Gherase (1976), Schneider (1976),
Tuttle et al. (1976)a and Hakky (1977).
Foa et al. (1976) demonstrated an exposure-effect relationship by
comparing workers from 2 plants with different degrees of exposure. A
relationship between exposure time and performance was demonstrated by
Schneider (1976), but the role of age as a confounding factor could be
excluded for 2 test variables only.
a Tuttle, T. C., Wood, G. D., & Grether, C. B. (1976) Behavioral and
neurological evaluation of workers exposed to carbon disulfide
(CS2). Unpublished report submitted to NIOSH (National Institute for
Occupational Safety and Health) by Westinghouse Electric Corporation,
Behavioral Services Center, Columbia, MD, USA, 156 pp.
From these data, it can be concluded that, at rather low exposure
levels, psychological test results are almost the only way of
assessing involvement of the central nervous system. Relationships may
exist between psychological impairment and EEG abnormalities, but
further studies are needed to clarify this point.
6.3.10.2 Peripheral nervous system
Many authors have studied the neuropathy and myelopathy of
animals exposed to carbon disulfide (Fischer & Michalova, 1956;
Higashida et al., 1959; Scheel, 1967; Frantik, 1970; Luke,, 1973;
Szendzikowski et al., 1973; Wrofiska-Nofer et al., 1973; Seppäläinen &
Linnoila, 1975, 1976). Destruction of the myelin sheet has been the
most prominent finding, but axonal changes have also been observed. In
the muscle fibres, atrophy of the denervation type occurred secondary
to the polyneuropathy. Some species differences in susceptibility to
carbon disulfide have been found. Cats and rabbits tolerated only low
concentrations of carbon disulfide. On the other hand, polyneuropathy
in rats was produced only with exposure to extremely high
concentrations (1200-2400 mg/m3). The slowing of nerve conduction
velocity in the sciatic nerve in rabbits and rats preceded clinical
symptoms such as clumsiness and later on, paresis. In rabbits,
electromyographic abnormalities suggested the presence of myelopathy
together with peripheral neuropathy (Seppäläinen & Linnoila, 1975,
1976). Neuropathological studies (Linnoila et al., 1975) showed severe
morphological alterations in the lateral and anterior funiculi of the
spinal cord in addition to pronounced axonal alterations in the
peripheral nerves of rabbits.
Lukas (1970, 1973) established by electromyography (EMG) that the
development of neuropathy in the rat was influenced by dose, duration
of exposure, physical conditions, and the nutritional state of the
animal. This confirms the long-standing clinical belief that the
individual constitution bears some relationship to the quantity and
quality of the effects of carbon disulfide (Djuric, 1967, 1971; Djuric
et al., 1973; Goto et al., 1971).
The protective effects of some substances have also been studied
in animals. The addition of zinc and copper salts (Scheel, 1967;
Lukas, 1970), and of pyridoxine to the diet (Frantik, 1970; Luke,,
1970) did not change the toxic effects of carbon disulfide but
improved the biochemical consequences of the intoxication, for example
by decreasing the excretion of xanthurenic acid with urine (Kujalova &
Tintera, 1970). Addition of nicotinic acid (Wrofiska-Nofer, 1970)
provided some protection against the effects of carbon disulfide lipid
metabolism. The clinical picture of carbon disulfide intoxication in
man is mainly based on toxic polyneuropathy (Bashore et al., 1938;
Vigiliana, 1954, 1961; Lukas, 1969; Lukas & Hromadka, 1977; Milkov et
al., 1970; Seppäläinen et al., 1972; Seppäläinen & Tolonen, 1974;
Seppäläinen, 1977; Gilioli et al., 1977). The subjective complaints
include paraesthesia, dysaesthesia, and fatiguability and diffuse
pains of the lower extremities. Sometimes irritative signs are
prevalent and sometimes the patients have a feeling of local
hyperaesthesia, or hypersensitivity on palpation in the muscles of the
lower limbs.
Distribution of the complaints is mostly symmetrical, and,
objectively, there is hyporeflexia. Klimkova-Deutschova (1965) pointed
out the characteristic dissociation of the patellar reflex response,
which is normal or increased, with a decreased or absent Achilles
tendon reflex. As the neuropathy develops, changes first appear in the
sensitive fibres of the lower extremities, later reaching the upper
extremities. (Lukas, 1969; Lukas & Hromadka, 1977). Hyperaesthesia is
mostly of a "sock" character, but sometimes it is also diffuse.
Palpation of the nerve roots and sometimes of the whole muscle groups
is painful. Today, the development of symptoms seldom reaches the
stage of motor paresis and, consequently, atrophic changes
(amyotrophy) are rare. The same is true of vasomotoric changes.
Amyotrophy has been described in the earlier literature in the area of
the triceps surae and quadriceps muscles of the legs, as well as in
the small muscles of the hand, the thenat, the hypothenar, and in the
interosseal muscles.
It has also been suggested that cells in the spinal cord are
injured (Manu et al., 1970; Seppäläinen et al., 1972). On the other
hand, disturbances in myoneural function have resulted in
myastheniform signs (Seppäläinen et al., 1972).
Needle electromyography and measurement of the motor conduction
velocities of peripheral nerves have proved to be sensitive methods
for detecting early nerve damage, even in neurologically normal,
exposed workers (Lukas, 1969; Manu et al., 1970; Seppäläinen et al.,
1972; Lilis, 1974; Seppäläinen & Tolonen, 1974; Knave et al., 1974;
Thiele & Wolf 1976; Lukas et al., 1977). The EMG abnormality most
often found consists of a reduced number of motor units at maximal
contraction and an increase in the duration of motor unit potentials
and fibrillation. The maximum motor conduction velocities (MCV) are
lower, especially in the leg nerves, and the distal latencies are
prolonged. However, the most sensitive test for peripheral nervous
involvement appears to be the conduction velocity of the slower fibres
of the ulnar and deep peroneal nerves, and that of the H-reflex in the
region of tibia] nerve (Lukas, 1969, 1977; Seppäläinen & Tolonen,
1974; Gilioli et al., 1977). In a recent epidemiological study
(Seppäläinen & Tolonen, 1974) comprising 118 male viscose rayon
workers and 100 control subjects from a paper mill, the most important
findings were as follows: the conduction velocities in the nerves of
the exposed men were generally slower than those of the controls. The
most distinct differences (all P values smaller than 0.01) were
obtained for the conduction velocity of the slower fibres of the ulnar
and deep peroneal nerves, the MCV of the deep peroneal and posterior
tibial nerves, and the motor distal latency of both the median and
ulnar nerves. The exposed group in this study was predominantly
composed of clinically "well" men, exposed for a number of years
(median 15) to carbon disulfide plus hydrogen sulfide concentrationsa
of about 60-190 mg/m3 (20-60 ppm) in the 1950s, about 30-95 mg/m3
(10-30 ppm) in the 1960s, and mainly below 60 mg/m3 (20 ppm) during
the last years. A little more than half of the men were still exposed
at the time of examination, the rest were working in "clean"
departments.
Different types of nervous system symptoms and findings among 51
Swedish viscose factory workers were evaluated by Knave et al. (1974).
The iodine-azide test was used as a measure of exposure intensity. The
comparison group consisted of 51 subjects matched for sex and age.
Symptoms of peripheral nervous dysfunction (e.g., restless legs,
cramps, pain, and numbness) were over-represented among workers,
especially the combination of these symptoms. Only one neurological
sign-namely tremor-showed a higher prevalence in the exposed than in
the control group. The authors measured only the maximal MCV of the
median nerve and found minor slowing in this MCV when the duration of
employment was between 5 and 30 years, but not after shorter exposure.
The prognosis of carbon disulfide polyneuropathy has not been
considered to be too poor. According to one follow-up study of exposed
workers, electroneuromyographic abnormalities tend to ameliorate if
the exposure level decreases or exposure is stopped (Seppäläinen,
1977). A tendency for repair and restitution of lost functions has
also been demonstrated experimentally in animals (Lukas, 1970;
Seppäläinen & Linnoila, 1975, 1976).
However, it has been shown that, even after periods of 10-15
years without exposure, abnormal conduction velocity values may be
found in workers with previous carbon disulfide poisoning or previous
long-term exposure to carbon disulfide (Seppäläinen et al., 1972).
This corresponds with the experience of Gilioli et al. (1977), who
found the reduction in conduction velocity to be irreversible. In
another study, Seppäläinen (1977) described an amelioration in the EMG
findings when carbon disulfide exposure was reduced or stopped.
a Exposure levels in studies by Hernberg et al. (1970, 1973, 1976),
Raitta et al. (1974), Tolonen (1974), Tolonen & Seppäläinen (1974),
and Tolonen et al. (1975) refer to combined concentrations of carbon
disulfide and hydrogen sulfide. Hydrogen sulfide concentrations were
about 10% of the carbon disulfide concentrations (Hernberg et al.,
1970).
6.3.11 Cardiovascular effects
A number of studies have shown that carbon disulfide causes
vascular changes in various organs of experimentally exposed animals
(Lewey, 1941; Guarino & Arcello, 1954; Paterni et al., 1958). In man,
evidence of vascular changes caused by long-term exposure, has been
accumulating since World War II. Attinger (1948) was the first to
describe such changes in 5 autopsy cases. Further clinical and autopsy
studies showed clearly that carbon disulfide was a vasotropic poison
(Uehlinger, 1952; Attinger, 1954; Vigliani & Pernis, 1955; Crepet et
al., 1956; Rechenberg, 1957; Maugeri et al., 1966a,b; Prerovska &
Roth, 1968; Maugeri et al., 1971; Prerovska & Zvolsky, 1973).
According to Vigliani & Pernis (1955) the "vasculopathia
sulfocarbonica" appears in 2 forms, namely in cerebral arteries
causing encephalopathy, and in the renal arteries causing nephropathy
and hypertension. The syndromes that follow these types of vascular
damage have already been described in sections 6.2, 6.3.7, and
6.3.10.1. Autopsy studies have revealed that sclerosis is prevalent in
large and middle-size arteries and hyalinization in arterioles and
capillaries. These changes appear in the blood vessels of all organs
but they are particularly prominent in the central nervous system and
kidneys.
Vascular changes due to carbon disulfide exposure are similar to
those produced with atherosclerosis due to age. Thus, the picture in
older persons is confused by the influence of age. The carbon
disulfide induced vasculopathy is therefore best studied in young
persons with a sufficiently long exposure time. With this in mind, a
joint Italian/Yugoslav team selected 104 young workers (age around
30-35 years) with more than 10 years' exposure for closer study
(Maugeri, et al., 1966a,b; Maugeri et al., 1971; Taccola et al.,
1971). Of these, 28 were invalids due to carbon disulfide poisoning.
Organic changes in blood vessels were discovered, mostly in the
invalids, by means of cerebral rheography. The results obtained
suggested progression of such changes even after the termination of
exposure. Using peripheral rheography, plethysmography, and
oscillography, the authors established functional vasoconstriction in
the arteries of the upper and lower extremities. Organic alterations
of the sclerotic type were rare and mainly appeared after prolonged
exposure and in the lower extremities.
During the last 10 years or so, it has become increasingly
evident that long-standing exposure to carbon disulfide promotes
coronary heart disease, even under circumstances where clinical
poisoning is uncommon. Of the several attempts made to connect carbon
disulfide exposure with coronary heart disease, only a few have
complied with necessary methodological requirements. The first such
investigation was a careful mortality study in Britain published by
Tiller et al. (1968). They found a 2.5-fold excess mortality in
viscose rayon workers exposed to carbon disulfide for 10 years or
more, compared with that of other workers. The same study demonstrated
that, in the 3 plants studied, coronary mortality was proportionally
higher among workers engaged in the viscose spinning process than in
other workers, men living in the locality, and the national statistics
derived from the Registrar General's tables. Similar results have been
obtained from Norway, where a 3-fold excess mortality from coronary
heart disease was found among workers, aged 35-54 years, compared with
unexposed workers in other departments (Mowé, 1971).
A prospective study that was initiated in 1967 in Finland, showed
that the 5-year mortality in a cohort of 343 male viscose rayon
workers exposed to about 30-95 mg/m3 (10-30 ppm) was almost 5-fold
(14 deaths compared with 3) that of an unexposed comparison cohort
from a nearby paper mill. Other causes of death were evenly
distributed (Hernberg et al., 1970, 1973). The incidence of nonfatal
first myocardial infarctions was almost 3 times that in the comparison
cohort (11 compared with 4) (Tolonen et al., 1975). In a continuation
of the follow-up extending to 8 years, the cumulative incidence rate
for mortality from coronary heart disease was 5.8% in the exposed
group and 2.6% in the comparison group, giving a rate difference of
3.2% (Hernberg et al., 1976; Nurminen, 1976). Between the sixth and
ninth years of follow-up, no further excessive mortality was observed,
probably because most (81%) of the originally exposed workers were no
longer exposed to carbon disulfide and also overall concentrations of
carbon disulfide had decreased to less than 30 mg/m3 (10 ppm). This
positive trend was promising with regard to coronary heart disease:
the prognosis of exposed workers improved with improved occupational
hygienic measures and with a reduction in the length of exposure over
a lifetime.
Anamnestic angina was more prevalent both in 1967-68, and at
re-examination in 1972, whereas there were no great differences in
coronary electrocardiographic findings (Hemberg et al., 1970; Tolonen
et al., 1975) as coded by the "Minnesota Code" (Blackburn et al.,
1960). These results showed that the greatest effect of carbon
disulfide on coronary arteries was on the production of fatal
infarctions (relative risk 4.8) followed in reducing order by all
infarctions, fatal and nonfatal (relative risk 3.7), nonfatal
infarctions (relative risk 2.8), angina (relative risk 2.2), and
coronary ECGs (relative risk 1.4). In other words, the more serious
the outcome, the greater the relative risk. The interpretations may be
that exposure to carbon disulfide not only worsens the prognosis of
existing coronary heart disease but increases the incidence of new
cases. However, it is not certain that this mode of action operates in
countries where coronary heart disease is less common than in Finland.
Coronary heart disease has a multifactorial etiology, and the pattern
described above may be noticeable only when a sufficient number of
other coronary risk factors are present at the same time. In fact,
some other studies have shown a more distinct excess of coronary ECGs
(Goto & Hotta, 1967; Cirla et al., 1972). It is possible that both
dose-dependent, methodological, and even geographical factors may be
responsible for the discrepancies reported so far. For example, it may
be that the pattern of excess coronary heart disease differs in
different populations, depending on the concomitant prevalence of
other coronary risk factors such as elevated blood lipids and
hypertension.
A joint analysis of the prevalence of angina pectoris, exercise
ECGs, and blood pressure measurements in Finnish and Japanese workers
exposed to carbon disulfide and in 2 unexposed control groups
supported this view (Tolonen et al., 1976). Angina was rare and
evidence of past infarction nonexistent in both exposed and unexposed
Japanese men; in the Finnish exposed and unexposed groups the
prevalence of angina was 15 and 10%, respectively. The prevalence of
coronary ECGs was almost equal among the exposed and unexposed men in
both the Japanese and the Finnish groups. Angina plus coronary ECGs as
evidence for coronary heart disease occurred with a prevalence of nil
for both exposed and unexposed Japanese, and 5 and 2% for the exposed
and unexposed Finnish groups, respectively. The study did not yield
any evidence of an excess in any of the variables examined among the
exposed Japanese workers. However, a follow-up study of this group is
necessary to shed more light on the possibility of excess morbidity
and mortality from coronary heart disease. Vertin (1977) did not find
any significant differences in coronary heart diseases, ECG changes,
serum cholesterol concentrations, and arterial pressure in workers
exposed to carbon disulfide concentrations of about 30-95 mg/m3
(10-30 ppm) compared with a control group.
Carbon disulfide vasculopathy may be due to the combined action
of several biochemical and physiological disturbances, namely:
(a) changes in lipid metabolism (section 6.3.9);
(b) disturbances in the coagulation mechanism (section 6.3.14);
(c) elevation of blood pressure (Hemberg et al., 1970; Sakurai,
1972; Tolonen et al., 1975);
(d) subclinical hypothyroidism (Cavalleri et al., 1971);
(e) a toxic effect on the myocardium, either direct or through
interference with the catecholamine metabolism (Magos, 1972).
However, much work remains to elucidate the mechanism of carbon
disulfide-induced atherosclerosis. In particular, coronary heart
disease has a multicausal origin and is closely related to the
saturated fat intake of the population. Furthermore, its incidence is
influenced by a large number of other risk factors, such as smoking,
diabetes, and physical inactivity. A combination of 2 or more risk
factors greatly increases the incidence. Thus, it may be postulated
that carbon disulfide will only become evident as a coronary risk
factor in the presence of other risk factors.
6.3.12 Carcinogenicity and mutagenicity
No reports are available that indicate any carcinogenic or
mutagenic effects of carbon disulfide.
6.3.13 Teratogenic effects
Only a weak teratogenic effect appeared in rats following
low-level exposure to a combination of hydrogen sulfide and carbon
disulfide (Bariljak et al., 1975).
A marked impairment (increase in early embreonal mortality,
reduction in fetal weight, malformations in the brain and limbs,
behavioural deviation) in the prenatal development of 2 successive
generations was produced in rats exposed to carbon disulfide
concentrations of 100 and 200 mg/m3 throughout the period of
gestation (Tabacova et al, 1977).
6.3.14 Other effects
The literature is contradictory concerning the effects of carbon
disulfide on blood coagulation. Saita & Gattoni (1957) found a slight
decrease in factor VII and prothrombin in exposed workers. On the
other hand, Moreo & Candura (1960) and De Nicola et al. (1962)
reported hypercoagulability in the blood of experimental animals, a
finding that was later confirmed in studies of exposed workers by
Candura et al. (1962), Saita et al. (1964), and Danilova (1968). An
increase in fibrinolysis was found by De Nicola et al. (1962) in
animals suffering from acute poisoning. In contrast, Candura et al.
(1962) found decreased fibrinolysis in workers exposed to carbon
disulfide.
The coagulation mechanism in highly-exposed young workers in a
viscose factory was studied by Visconti et al. (1966a,b,c, 1967). They
found protracted coagulation, retarded production of active
thrombotastin, decreases in plasminogen and in the activity of
plasmin, and a decrease in antiheparinic activity in workers exposed
for up to 9 years. These disturbances were most pronounced during the
first years of exposure.
Visconti et al. (1966a) supposed that carbon disulfide could
affect blood coagulation either directly by interfering with the
system itself, or indirectly, by liver injury. Different routes of
entry, differences in the level and duration of exposure and in the
distribution of carbon disulfide in the organism, and also
interspecies differences are probably responsible for the confusing
results.
It has been reported that the glucose metabolism is disturbed by
carbon disulfide both in experimental animals (rabbits) (Hara, 1958)
and in exposed workers (Goto et al., 1971). There are also a few
clinical studies reporting an increased occurrence of diabetes in
patients suffering from severe carbon disulfide poisoning (Austoni &
d'Agnolo, 1957; Finulli & Ghislandi, 1959; Ferrero, 1969). Lack of
proper control groups renders the interpretation of these results
difficult.
6.3.15 Interactions with other chemical compounds
The combined effects of carbon disulfide with other chemical
compounds were first reported by Lazarev et al. (1965) who indicated
that repeated exposure to a concentration of carbon disulfide of
3900 mg/m3, for 2 h per day, over a period of 20-30 days considerably
prolonged both the hexobarbital sleeping time and alcohol retention
time in rats and mice (see also section 6.3.6).
Carbon disulfide caused reversible inhibition of the oxidative
drug metabolism in rat liver endoplasmic reticulum (Bond et al., 1969;
Freundt & Dreher, 1969; Freundt & Kuttner, 1969; Freundt & Henschler,
1971; Freundt, 1973; Sokal, 1973; Freundt et al., 1974b, 1975, 1976).
This effect was also produced in man after exposure to a carbon
disulfide concentration of about 30 mg/m3 (10 ppm) over a 6-h period
(Mack et al., 1974); the treatment led to a rapidly reversible but
significant decrease in the oxidative N-demethylation of the
analgesic, aminopyrine.
Simultaneous exposure to carbon disulfide and ethanol resulted in
an increase in blood acetaldehyde in rats and man that was considered
to be a consequence of the inhibition of aldehyde dehydrogenase by
carbon disulfide (Freundt & Netz, 1973; Freundt & Lieberwirth, 1974;
Freundt, 1974; Freundt et al., 1976). In 12 healthy male volunteers, a
50% increase in blood acetaldehyde was noted following exposure to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) for 8 h
with simultaneous ingestion of ethanol in orange juice at a
concentration that maintained the blood ethanol level at about 0.8%.
However, no signs of an "antabuse syndrome" were observed under these
experimental conditions (Freundt et al., 1976).
6.4 Diagnosis
Since all isolated effects are nonspecific, individual diagnosis
becomes a matter of probability based on: (a) the ascertainment of
exposure; (b) the demonstration of signs and symptoms of poisoning
and the combination in which they occur; and (c) the exclusion of
other diseases.
The changes due to carbon disulfide exposure described in
sections 6.2-6.3.15 affect so many different organ-systems that, in
considering the etiology of a single case, the probability of it being
caused by carbon disulfide increases in proportion to the number of
these signs and symptoms present. Although, in 1941, Lewey drew
attention to the importance of the concomitant occurrence of different
neuropsychiatric and vascular effects, as well as to their connection
with the severity of the disease, there have been few, if any, studies
systematically analysing the knowledge that has emerged since then.
For this reason it is difficult to find quantitative data. However,
carbon disulfide poisoning should always be suspected when a viscose
worker presents with subjective, neurasthenic symptoms, signs of
peripheral neuropathy, psychological disturbances, and vascular
changes in the cerebral, coronary, renal, or peripheral systems. When
exposure to toxic levels of carbon disulfide occurs, the following
chronological effects can usually be expected: the appearance of
neurasthenic symptoms accompanied by psychological behavioural
changes; signs of peripheral nervous system involvement as shown by
electromyographic changes, initially mainly in the afferent fibres but
later in the motor fibres; simultaneous disturbances in the central
nervous system may appear but may be more difficult to detect
objectively than the lesions of the peripheral nervous system. Another
early finding is a decrease in the level of serum thyroxine that is
probably due to damage of the hypothalamic-hypophyseal axis (section
6.3.9). Cardiovascular damage appears after longer periods of
exposure. The appearance and severity of the above-mentioned signs and
symptoms are related to the level of the uptake and to the duration of
exposure to carbon disulfide. The changes are also dependent on the
individual constitution and health status of exposed workers.
In the differential diagnosis of true cases, atherosclerosis due
to other causes should be considered including, postencephalitic or
atherosclerotic Parkinsonism, brain tumours, syphilis, multiple
sclerosis, and psychiatric diseases of other origins such as endogenic
depression, schizophrenia, and alcoholism. Peripheral polyneuropathy
should be distinguished from that due to alcoholism, diabetes, and
other toxic agents.
The subclinical stage is, by definition, more difficult to
identify. First, the presence of pathological symptoms and signs must
be established, which requires sensitive techniques. Second, the
causal connection between an established symptom or sign and carbon
disulfide exposure should be shown. Both aspects pose great
difficulties because of the nonspecificity of the manifestations and
their vagueness in the initial stages. So far, the only attempt to
approach this problem in a systematic way has been made by Tolonen
(1974).
He studied 97 men exposed to carbon disulfide in a viscose
factory and 96 controls of the same age from a paper mill. The mean
age was 48 years (range 33-67 years), and the mean exposure time was
15 years (range 1-27 years). The intensity of background exposure
ranged from about 60 to 125 mg/m3 (20 to 40 ppm) in the 1950s and
then from about 30-95 mg/m3 (10-30 ppm). Personal exposure data were
not available. The group comprised both completely "healthy" workers
as well as some partly incapacitated patients with past poisoning.
About half of the men were still exposed to carbon disulfide, the rest
being employed in "clean" departments. The groups underwent many
examinations including: (a) examination of the heart (positive =
history of verified myocardial infarction, and/or "Minnesota Codes"
I1-3, IV1-3, V 1-3, VIII3, VIII3 , XI1-3-5-7, and/or typical angina):
(b) psychological testing (section 6.3.10.1); (c) measurement of
the conduction velocities of 8 peripheral nerves (polyneuropathy was
considered to exist when 2 or more nerves showed reduced conduction
velocities) and (d) examination of the circulation of the ocular
fundus (the criterion for disturbed circulation was delayed
peripapillary filling -- circumferential, segmental, or both). The
occurrence of isolated and combined signs is shown in Table 2.
As many as 59% of the exposed and 29% of the unexposed men were
affected by more than one disorder under study but most combinations
of disorders occurred more frequently in the exposed group; a
combination of 3 abnormalities was 3 times more common in the exposed
group than in the controls and a combination of all 4 abnormalities
occurred only in the exposed group. Only 5% of the exposed subjects
compared with 31% of the controls were without any abnormalities.
Since disturbances in the choroidal circulation were present in all
cases of excess "morbidity" (68% of the exposed, Raitta et al., 1974),
it seems that this abnormality represents the earliest manifestation
of carbon disulfide toxicity, at least, of those considered here.
Thus, it can be postulated that a positive diagnosis of carbon
disulfide poisoning can only be made if changes in the choroidal
circulation have been observed and providing that exposure has
extended over 10 years or more, with an intensity of the magnitude of
about 30-90 mg/m3 (10-30 ppm). The estimated probability that a
combination of findings is of occupational origin is the excess
morbidity over total morbidity. The results also showed that the
etiological role of carbon disulfide could be demonstrated with
greater probability, the greater the number of abnormalities present
at the same time.
The necessity to ascertain exposure is so obvious that it should
not need to be mentioned. It should be stressed that recent experience
has shown the importance of evaluating personal, as opposed to
background exposure, since the former may be only weakly correlated
with the latter. Personal exposure can be measured using personal
samplers, or by the iodine-azide test. The latter has been claimed to
discriminate particularly sensitive workers (whose test does not
return to normal after 16 h away from exposure) (Graovac-Leposavic et
al., 1967).
Table 2. Prevalence (%) of coronary heart disease (CHD), delayed
peripapillary circulation (EYE), polyneuropathy (PN) and
behavioural symptoms (BS), their combinations in subjects
in the exposed and control groups, and the differences
between the groups.a
Exposed Group Difference =
(N = 97) control excess morbidity
(N = 96)
Free of disease 5 31
CHD only 4 9 -5
EYE only 19 19 0
PN only 6 6 0
BS only 7 6 1
CHD + EYE 4 4 0
CHD + PN 1 1 0
CHD + BS 1 2 -1
EYE + PN 12 5 7
EYE + BS 7 4 3
PN + BS 4 5 -1
CHD + EYE + PN 9 0 9
EYE + PN + BS 11 5 6
CHD + PN + BS 2 1 1
CHD + EYE + BS 2 2 0
CHD + EYE + PN + PS 6 0 6
Total 100 100 26
a From: Tolonen (1974).
6.5 Surveillance of the Health of Exposed Workers
Considering the early manifestations of chronic carbon disulfide
effects and the factors that may influence the appearance and the
severity of such effects (section 6.4), the following examinations are
recommended for health surveillance:
(a) Clinical neurological investigation;
(b) Electroneuromyographic (ENMG) examination wherever possible
(especially the conduction velocity test, which should be carried out
on several nerves);
Additional tests that may give valuable information include:
(a) Psychological and behavioural testing with a narrow battery
of well validated tests;
(b) Serum thyroxine measurement repeated at least once a year;
(c) Blood pressure measurements;
(d) Electrocardiography (preferably an exercise ECG);
(e) Fundus photography (in some countries);
(f) Blood lipid pattern estimation, if the exposure level is
high;
(g) Electroencephalography (EEG), if there are special
indications;
These tests should be performed at the pre-employment examination
and subsequently at regular intervals to detect early deviation from
baseline values. Changes observed in the same individual over a period
of time may suggest a relationship with carbon disulfide uptake.
Depending on the intensity of exposure, the iodine-azide test
should be carried out from 2-12 times a year, both immediately after
the work shift and the next morning. Those with positive tests should
be seen by the plant physician, since a pathological test in the
morning is a warning of incipient poisoning (Graovac-Leposavic et al.,
1967). In addition to this, medical examinations should be made once
or twice a year. They should comprise a thorough history and a
neurological examination.
When suspect signs of early carbon disulfide effects appear, the
worker should be removed from exposure to a "clean" job. In the light
of the new findings concerning coronary heart disease, the removal of
workers who develop coronary risk factors, such as hypertension,
hypercholesterolaemia, and ECG changes may be indicated, irrespective
of their etiology. In some plants, there is the practice of regular
periods of work in a clean atmosphere, e.g., every sixth month, or of
extra vacations in the winter.
Some authors believe in the importance of the diet of exposed
workers. However, the protective effect of various diets and
supplements for man remains unclear. Scheel (1965) claimed that
addition of zinc and copper salts to the diet of exposed rats had a
protective effect against carbon disulfide exposures. A diet poor in
proteins, especially amino acids containing sulfur, but enriched in
vitamin B6 and glutaminic acid was suggested by Agronovskij &
Goloscapov (1972). Lukas (1973) established that rats on an "optimal"
diet were more resistant to the effects of carbon disulfide than rats
on a normal diet. In the first group, the signs of intoxication
appeared after a longer time and the effects were less marked than in
the second group.
At the present time, the only recommendation that can be given in
the case of man is that workers should be provided with a diet
containing sufficient amounts of energy foods to provide the required
joules, proteins, vitamins, and trace elements. This recommendation is
of particular importance in countries where such dietary requirements
are not necessarily fulfilled normally. However, basing the preventive
programme mainly on diets or drugs should be condemned, since the
fundamental question is that of technical improvements.
6.6 Contraindications for Exposure to Carbon Disulfide
An inborn error of metabolism manifesting itself as an abnormal
iodine-azide reaction in subjects working under conditions of high or
moderate carbon disulfide exposure (less than, 50 mg/m3) should be
considered an absolute contraindication for exposure.
Other contraindications for working in an environment containing
carbon disulfide include those common to all other toxic exposures
such as youth (below 18 years), pregnancy, concomitant chronic disease
such as psychiatric neurological, cardiac, renal, and pulmonary
diseases, and any condition that prevents the use of respirators, etc.
In addition, contraindications based on the specific toxicity of
carbon disulfide include diseases of the metabolic system, such as
liver disease and endocrine disorders. Any neurological or psychiatric
disorder is also a contraindication, including vegetative dystony and
psychic lability. A history of gastritis and peptic ulcer is a
relative contraindication, depending, for instance, on whether or not
there will be shift work and on the intensity of exposure. Recently,
attention has been focused on to what extent the presence of coronary
risk factors should be regarded as a contraindication; no strict
recommendations can be given, but as a general rule the presence of
several of these in an individual, or alternatively strongly abnormal
values for one risk factor, should be regarded as contraindications
(the strongest coronary risk factors apart from age itself are
elevated serum cholesterol or triglycerides, heavy cigarette smoking,
hypertension, and diabetes). In the evaluation of these and other
contraindications, the exposure intensity and hygienic conditions
should, of course, be taken into consideration.
The employment of women in work places with carbon disulfide
exposure is a special problem.
Considering the studies referred to in section 6.3.9, it is
advisable to exclude pregnant women from carbon disulfide exposure.
The same is true for lactating women and for those with expressed
disturbances of menstruation and habitual abortion.
7. EXPOSURE-EFFECT AND EXPOSURE-RESPONSE RELATIONSHIPS
7.1 Validity of Exposure Data
In general, valid exposure data are available only from
experimental studies on animals. Almost all epidemiological studies
have failed to document the exposure levels pertinent to the effects
studied. There is a universal lack of retrospective exposure data that
usually renders the assessment of the relationship between chronic
effects and the exposure levels responsible for their occurrence
impracticable. Furthermore, even when exposure data are available,
they often consist of occasional short-time measurements from fixed
sites that are not necessarily representative of the workers'
exposure. It is, therefore, extremely difficult, if not impossible, to
establish exposure-effect or exposure-response curves for carbon
disulfide, based on epidemiological evidence. Even the relation of
isolated findings reported in the literature, to given exposure levels
is difficult. Thus, the attempts in this document, to relate the
intensity of exposure to the intensity and frequency of effects must
be regarded with caution.
7.2 Experimental Data
7.2.1 Acute animal exposure
Acute exposure to carbon disulfide in animal experiments by
inhalation or by intramuscular or intraperitoneal injection has
provided information regarding the mechanism of carbon disulfide
toxicity, but it is difficult to apply this information to the
industrial exposure of man.
7.2.2 Long-term animal exposure
Several experiments on the inhalation of carbon disulfide have
been performed on different types of animals. In most of these
studies, exposure was to carbon disulfide only, but in a few, a
combination of carbon disulfide and hydrogen sulfide was used.
The concentrations of carbon disulfide ranged from
0.1-2330 mg/m3 and those of hydrogen sulfide, from 0.1-140 mg/m3;
the exposure periods ranged from 30 min per day to 6 h per day for up
to 15 months.
The reactions to such exposures included increased mortality,
teratogenic changes, testicular lesions, aspermatogenesis, engorged
blood vessels, weakness, paralysis, lethargy, weight changes,
metabolic changes, behavioural changes; sometimes there were no
observed effects. Some of the effects resembled lesions found in
exposed workers; others did not bear any relationship to those arising
from human exposure (Wakatsuki & Higashikawa, 1959; Goldberg et al.,
1964; Minden et al., 1967; Yaroslavski, 1969; Frantik, 1970; Gondzik,
1971; Misiakiewicz et al., 1972; Szendzikowski et al., 1973; Bariljak
et al., 1975; Seppäläinen & Linnoila, 1976; Tabacova et al, 1977). The
considerable differences in ranges of concentrations and periods of
exposure used in these experiments make conclusions regarding
exposure-effect and exposure-response curves impossible. This
regrettable situation emphasizes the need for international
coordination of toxicological experiments to make comprehensive
scientific evaluation possible.
7.3 Epidemiological Data
All studies so far reported have been from the viscose rayon
industry, where exposure to carbon disulfide occurs together with
exposure to hydrogen sulfide. However, generally, the levels of carbon
disulfide are at least one order of magnitude higher than those of
hydrogen sulfide, and most authors have attributed the health effects
found to carbon disulfide rather than to hydrogen sulfide exposure.
Only some of the various effects reviewed in section 6 can be
related to exposure levels. The following section contains a brief
review of exposure-effect and exposure-response data for neurological,
cardiovascular, ocular, and gonadal effects.
7.3.1 Neurological and behavioural effects
Severe acute neurological effects such as psychosis and paralysis
occur when exposure exceeds 1500-3000 mg/m3 (500-1000 ppm) (Vigliani,
1954). Chronic encephalopathy may develop with long-standing exposure
to 150 mg/m3 (50 ppm) or more (Vigliani, 1954). More subtle
neurological changes, detectable by neurophysiological examination or
psychological testing, have been reported at lower concentrations. For
example, slowing of nerve conduction velocities and prolonged distal
latencies of several nerves have been found among viscose rayon
workers with a median exposure time of 15 years to combined
concentrations of carbon disulfide and hydrogen sulfide of
60-190 mg/m3 (20-60 ppm) in the 1950s, 30-95 mg/m3 (10-30 ppm) in
the 1960s, and mainly below 60 mg/m3 (20 ppm) during the years
preceding the examination (Seppäläinen & Tolonen, 1974). The same
workers were also studied for psychological performance (Tolonen,
1974). The most distinct difference compared with a control group was
found in the retardation of psychomotor speed. As a whole, "poor"
performance, as defined by Tolonen (1974), occurred 1.6 times more
frequently among the exposed workers. These results suggest that
concentrations around 60-90 mg/m3 (20-30 ppm) can produce
psychological disturbances. In an indirect way, Tuttle et al.a
corroborated this view by being able to relate behavioural test scores
to other indices of neurological health, thought to occur under
similar exposure conditions, but because no exposure data were
available, their findings are not directly applicable in the
exposure-effect sense.
According to Martynova et al. (1976), an excess of functional
nervous disorders was found among 108 workers exposed to carbon
disulfide concentrations about 8-11 mg/m3 (maximum approximately
20-25 mg/m3), for 10-15 years, compared with 390 control subjects.
For example, sensory polyneuritis was much more frequent and the
threshold for pain was significantly higher in the exposed group.
These studies indicate that background concentrations below about
60 mg/m3 (20 ppm) may cause neurological dysfunction.
7.3.2 Cardiovascular effects
Severe vascular effects manifesting themselves as
vasculoencephalopathy and renal atherosclerosis developed with
long-term exposure to carbon disulfide concentrations exceeding
150-300 mg/m3 (50-100 ppm) (Vigliani, 1954; Nofer et al., 1961).
Excess incidence of coronary heart disease has been reported with
longstanding exposure to concentrations averaging 30-125 mg/m3
(10-40 ppm) (Hernberg et al., 1970, 1973, Tolonen et al., 1975). The
mortality rate from coronary heart disease appeared to be most
affected while the incidence of milder manifestations such as angina
and ECG-changes was less influenced. In fact, most studies, published
so far, have failed to reveal any difference in the prevalence of
ECG-findings between exposed and unexposed workers (Tolonen et al.,
1976).
Quite recently, Vertin (1977) reported no difference between the
incidence of coronary heart disease found in control subjects and that
in workers exposed to about 30-95 mg/m3 (10-30 ppm) suggesting a
no-observed effect level for coronary heart disease in this range.
This finding does not necessarily contradict those referred to above;
possibly, the true exposure levels responsible for the excess coronary
heart disease in the Finnish studies were higher than those in
Vertin's study. It is probable that, in general, differences in actual
personal exposure levels in the studies-not necessarily those levels
measured and reported -- account for seeming discrepancies in results
and that exposure data are inadequate in most published studies.
a Tuttle et al., 1976. Unpublished report. (See footnote to section
6.3.10.1.)
Differences in blood pressure have been reported by Hernberg et
al. (1970) and Tolonen et al. (1975) between workers exposed to a
carbon disulfide concentration of about 60 mg/m3 (20 ppm) and
unexposed subjects, and by Martynova et al. (1976) in workers exposed
for 10-15 years to concentrations of about 10 mg/m3. These findings
indicate that effects upon the vascular system begin at low exposure
levels.
7.3.3 Ophthalmological effects
Among Japanese workers, the occurrence of retinal microaneurysms
was related to the duration of exposure (Sugimoto et al., 1976). The
exposure levels in earlier Japanese studies were not reported; in
present studies they are probably of the order of 60 mg/m3 (20 ppm).
The findings of retinal microaneurysms appears to be confined to
Japanese workers, since, according to a recent joint study, no such
effect could be demonstrated in Finns (Sugimoto et al., 1977). In
contrast, Finnish workers showed delayed peripapillary filling and
wider calibre of retinal arteries compared with control subjects
(Raitta et al., 1974). Exposure conditions under which these changes
occurred are described in section 6.3.2 and indicate that haemodynamic
changes in the ocular circulation are induced as early as coronary
effects.
7.3.4 Gonadal effects
The results of Vasiljeva (1973), Petrov (1969), and Finkova et
al. (1973) suggest that carbon disulfide concentrations below
10 mg/m3 (3 ppm) may cause disturbances in the ovarian function of
young females, resulting in menstrual disorders, pregnancy
complications, and spontaneous abortions. Given that the exposure
measurements reported are comparable with those of other studies, it
would seem that these would be the earliest detectable type of effects
in human beings.
A summary of the most relevant exposure-effect and exposure-
response findings is given in Table 3.
Table 3. Some exposure-effect and exposure-response relationships
for long-term exposure of man to carbon disulfide.
Concentration Exposure Symptoms & signs Reference
(mg/m3) time
(years)
10 5-10 Blood progesterone depressed, Vasiljeva (1973)
estriol increased, irregular
menstruation;
10 10-15 Sensory polyneuritis, increased Martynova et al. (1976)
pain threshold;
30 >3 Spontaneous abortions twice as Petroy (1969)
common and premature births 3
times as common as in controls;
30-120 >10 Coronary mortality about 5 times Tolonen et al. (1975)
that in controls, life expectancy Hernberg et al. (1973)
decreased 0.9-2.1 years depending Nurminen (1976)
on age; nonfatal infarctions 2.6 Hernberg et al. (1970)
times more frequent than in controls;
angina pectoris about twice as
prevalent as in controls; slightly
higher systolic and diastolic blood
pressure than in controls;
30-120 >6 Peripheral nervous and CNS Seppäläinen & Tolonen
dysfunction, conduction velocities (1974); Tolonen (1974)
slowed, psychological changes;
100-140 2-12 Decrease of testosterone and Cavalleri et al. (1970)
gonadotrophin LH in urine;
200-500 1-9 Ophthalmic pressure 18.4/14.7 Maugeri et al. (1966,
vs 15.3/11.6 kPa (138/110 vs 1967)
115/87 mmHg) in controls;
500-2500 0.5 Polyneuritis, myopathy acute Vigliani (1946)
psychoses
8. CONTROL OF EXPOSURE IN THE VISCOSE INDUSTRY
Technical and administrative measures for the protection of the
health of workers exposed to carbon disulfide in the production of
viscose rayon include:
-- exchange of production methods (substitution or elimination);
-- enclosure and local exhaust ventilation of the technological
process;
-- dilution (general) ventilation of the work room;
-- isolated and well-ventilated rooms for process control
operators and for rest periods;
-- use of personal respiratory protective equipment under certain
hazardous circumstances and,
-- rotation of workers periodically to areas free from carbon
disulfide exposure.
New developments such as the SINI viscose process make it
possible to reduce the total consumption of carbon disulfide to 70-80%
of that used in conventional processes (Sihtola, 1976). Occupational
health aspects must be taken into consideration at the planning stage
in the construction of new plants.
Enclosure of processes and machines is the second most effective
means of technical prevention. This technique should be applied in the
sulfidizing process and for spinning and washing machines. Possible
leakages in closed systems should be controlled by gas detectors. The
air removed from the processes by the exhaust ventilation should be
transported to a recovery system. The benefits of such a system are
both economic, since the plant has an interest in the recovery of
useful material, and hygienic, as the possibility of the pollution of
both the working rooms and the environment around the factory is
reduced. The air removed by exhaust ventilation is compensated with
pure air, which is brought to working sites by ventilation systems.
It is not possible to protect the workers in all situations by
ventilation systems alone. In certain circumstances, the workers may
be exposed to very high concentrations of carbon disulfide, in which
case respiratory protective devices must be used. The type of
respirator may vary from a chemical cartridge (organic vapour)
respirator, used for low concentrations, to supplied air or
self-contained breathing devices for very high concentrations. It is
also important to provide special isolated rooms with a slightly
positive pressure, compared with the work area contaminated with
carbon disulfide, for process control workers and for rest pauses.
Another factor of concern in the control of exposure and uptake
of carbon disulfide involves skin absorption resulting from direct
contact with the liquid. Where workers may come into contact with
liquid carbon disulfide, appropriate protective clothing, such as
synthetic rubber gloves and goggles or face shields, should be worn.
If carbon disulfide is spilled, potential sources of ignition
should be eliminated immediately, spark-proof ventilation provided,
and the spill cleaned up. A small spill should generally be allowed to
evaporate under conditions of good air circulation. However, large
spills should be covered with water and flushed into a retention basin
under a water layer. Carbon disulfide should not be drained into a
sewer system because of the possibility of an explosion.
In addition to these control methods, data obtained through the
biological monitoring of exposed workers can be used to determine
excessive uptake situations that require additional control measures.
The procedure for biological monitoring is discussed in section 6.5.
REFERENCES
ABRAMOVA, J. I. (1967) The questions of pathogenesis, specific
prevention and therapy of carbon disulfide intoxication. In:
Brieger, H. & Teisinger, L, ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 32-34.
ABUCEWICZ, A., SAMOCHOWIEC, L., & SZYSZKA, K. (1971) The role of
5-hydroxytryptamine in the course of experimental carbon
disulfide poisoning. Roca. Pomor. Akad. bled. Szczec, Suppl. 4,
pp. 89-96.
AGRANOVSKIJ, Z. M. & GOLOSCAPOV, O. D. (1972) [Hygienic assessment of
preventive diet for subjects exposed to carbon disulfide.] Gig.
i Sanit., 37 (II): 43-47 (in Russian).
AMERICAN STANDARDS ASSOCIATION (1941) Allowable concentration of
carbon disulfide, New York, ASA, 141, 7 pp. (ASA Z 37.3-1941).
ANDREEVA, N. B. (1970) [Experimental material for the diagnosis of
carbon disulfide poisoning, on the harmful effect of the poison
at the maximum permissible level for workroom air, and on the
passage of carbon disulfide in solution through the skin.] In:
Voprosy diagnostiki, specificeskoj profilaktiki i terapii
professional 'nyh zabolevanij, Leningrad, pp. 40-52 (in
Russian).
ANDRUSZCZAK, K. (1967) [The behaviour of ceruloplasmine level in the
blood serum in workers exposed to the carbon disulfide action.]
Med. Pr., 18:120-127 (in Polish).
ATTINGER, E. (1948) [Chronic carbon disulfide intoxication appearing
under the unusual symptoms of a heavy vascular disease.]
Schwiez. Med. Wochenscht., 78:667-669 (in German).
ATTINGER, E. (1954) [Chronic carbon disulfide intoxication and its
pathogenesis, its early detection and prevention.] Schweiz. Med.
Wochenschr., 84:912-913 (in German).
AUDIO-GIANOTTI, G. B. (1932) [Parkinsonism caused by professional
carbon disulphide poisoning.] Presse med., 40 (July-Dec.)
1289-1291 (in French).
AUSTONI, M. & D'AGNOLO, B. (1957) [Chronic carbon disulfide poisoning
and diabetes mellitus.] Minerva Med., 48:2500-2504 (in
Italian).
BAGON, D. A., CROSBY, M. T., HARDY, H. L., & SIMMONS, J. H. (1973) A
personal sampling technique for simultaneous determination of
hydrogen sulfide and carbon sulfide in air. Ann. occup. Hyg.,
16: 133-139.
BAIKOW, E. K. (1963) [Experimental data on the determination of the
maximum permissible concentration of carbon disulfide and
hydrogen disulfide in the atmosphere in their simultaneous
presence.] Gig. i Sanit., 28 (3): 3-8 (in Russian).
BARD, C. L. (1892) Cases in practice, malignant pustle and insanity
due to bisulphide of carbon. South Cal. Pract., 7: 476-485.
BARILJAK, I. R., VASILEVA, I. A., & KALINOVSKAJA, L. P. (1975) [Effect
of small concentrations of carbon disulfide and hydrogen sulfide
on intrauterine development in rats.] Arkh. Anat. Gistol.
Embriol., 58:77-81 (in Russian).
BARTONICEK, V. (1957) [The distribution of carbon disulfide in whole
blood, the brain and the suprarenal glands over a given period in
parenteral administration in white mice. Prac. Lek., 9:28-30
(in Czech).
BARTONICEK, V. (1958) [The distribution of free carbon disulphide and
bound carbon disulphide liberated by acid hydrolysis in the
organs of white rats.] Prac. Lek., 10: 334-338 (in Czech).
BARTONICEK, V. (1959) [Distribution of free carbon disulfide and bound
carbon disulfide liberated by acid hydrolysis in the organs of
white rats.] Prac. Lek., 10:504-510 (in Czech).
BASHORE, R., HAMILTON, A., LEWEY, F., ERSKINE, L., HAMMOND, J., HOUTZ,
R., BRACELAND, F., MCDONALD, R., BATSON, O., BELLET, S.,
CRESKOFF, F., EHRICH, W., & ALPERS, B. J. (1938) Survey of
carbon disulfide and hydrogen sulfide hazards in the viscose
rayon industry, Harrisburg, PA, Occupational Disease Prediction
Division, US Department of Labor and Industry p. 68 (Bull. 46).
BINET, L. & BOURLIERE, F. (1944) Sur les modifications du sang au
cours du sulfocarbonisme chronique. Arch. Mal. Prof., 6: 12-16.
BITTERSOHL, G. (1971) On relationships in action between carbon
disulfide and hydrogen sulfide. Med. Lavoro, 62: 554-556.
BITTERSOHL, G. & THIELE, H. (1977) The health of workers exposed to
carbon disulphide in the production of artificial fibres. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski (Finland), 6-10 June 1977, p. 5.
BLACKBURN, H., KEYS, A., SIMONSON, E., RAUTAHARJU, P., & PUNSAR, S.
(1960) The electrocardiogram in population studies. A
classification system. Circulation, 21: 1160-1175.
BOBSIEN, K. (1954) [New method for determination of uptake and binding
by endogenous substances.] Arch. Gewerbepath. Gewerbehyg., 13:
193-203 (in German).
BOND, E. J. & DE MATTEIS, F. (1969) Biochemical changes in rat liver
after administration of carbon disulphide, with particular
reference to microsomal changes. Biochem. Pharmacol., 18:
2531-2549.
BOND, E. J., BUTLER, W. H., DE MAITEIS, F., & BARNES, J. M. (1969)
Effects of carbon disulphide on the liver of rats. Br. J. ind.
Med, 26: 335-337.
BRIEGER, H. (1961) Chronic carbon disulfide poisoning. J. occup.
Med., 8: 302-308.
BRIEGER, H. (1967) Carbon disulphide in the living organism-Retention,
metabolism and physiological effects. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 27-31.
BRIEGER, H. & FRIEDMAN, M. H. F. (1949) In vitro effects on
digestive enzymes, theory and pathology of poisoning. J. ind.
Hyg., 31: 96-98.
BRIEGER, H. & TEISINGER, J., ed. (1966) Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 27-31.
BRUCE, A. (1884) Chronic poisoning by CS2. Edinburgh Med. J., 29:
1009.
BÜSING, K. H. (1952) [The determination of minute amounts of carbon
disulfide in blood.] Zentralbl. Arbeitsmed., 2: 115-122 (in
German).
BÜSING, K. H. & SONNENSCHEIN, W. (1954) [The binding of carbon
disulfide in the organs.] Zentralbl. Arbeitsmed. Arbeitschutz,
4:65-69 (in German).
BÜSING, K. H., SONNENSCHEIN, W., BECHER, E. W., & DREIHELLER, H.
(1953) [Experimental studies in animals on the binding of carbon
disulfide in organs using labelled carbon disulfide (C35S2).]
Z. Naturforsch., 86: 495-500 (in German).
CANDURA, F., GRAVERI, A., & STRANEO, G. (1962) [Clinical contribution
to studies on the relationship between arteriosclerosis and
carbon disulfide poisoning.] Folia Med., 45: 749-769 (in
Italian).
CAVALLERI, A. (1972) [Alterations in the endocrinological function as
an early sign of carbon disulfide poisoning.] Med. Lavoro, 63:
81-84 (in Italian).
CAVALLERI, A. (1975) Serum thyroxine in the early diagnosis of carbon
disulfide poisoning. Arch. environ. Health, 30: 85-87.
CAVALLERI, A. & ZUCCATO, F. (1965) [Endocrinological observations on
carbon disulfide poisoning. I: Adrenocortical function.] Folia
Med., 48: 519-527 (in Italian).
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1965) [Adrenocortical and
gonadal function in human carbon disulfide poisoning.] In:
Proceedings of the 28th National Congress on Occupational
Health, Naples, 1965, pp. 645-660 (in Italian).
CAVELLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1966a) Endocrinological findings in young workers
exposed to carbon disulphide. I. - Urinary excretion of total
17-ketosteroids. Med. Lavoro, 57: 566-572.
CAVALLERI, A., DJURIC, D., VISCONTI, E., REZMAN, I., MAUGERI, U., &
BRANKOVIC, D. (1966b) Endocrinological findings in young workers
exposed to carbon disulphide. II.-Urinary excretion of
17:hydroxy-corticosteroids. Med. Lavoro, 57: 573-578.
CAVALLERI, A., DJURIC, D., VISCONTI, E., BRANKOVIC, D., & MAUGERI, U.
(1966c) Androsterone and etiocholanolone in the urine of young
workers exposed to carbon disulphide. In: Proceedings of the XV
International Congress on Occupational Health, Vienna, 19-24,
1966, Vienna, Wiener Med. Akademie, Vol. II-I, pp. 347-353.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., & VISCONTI, E.
(1966d) Endocrinological findings in young workers exposed to
carbon disulphide. III.-Urinary excretion of 3alpha,
11-deoxy-17-ketosteroids. Med. Lavoro, 57: 755-760.
CAVALLERI, A., DJURIC, D., MAUGERI, U., BRANKOVIC, D., VISCONTI, E., &
REZMAN, I. (1967) 17-ketosteroids and 17-hydroxycorticosteroids
in the urine of young workers exposed to carbon disulphide. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 86-91.
CAVALLERI, A., MAUGERI, U., & VISCONTI, E. (1970) L'excretion urinaire
de testosterone et de gonadotrophine stimulant les cellules
interstitielles (ICSH) chez des subjets exposes au sulfure de
carbone. Arch. Mal. Prof., 31: 23-30.
CAVALLERI, A., TACCOLA, A., GRAOVAC-LEPOSAVIC, LJ., MAUGERI, U., &
DJURIC, D. (1971) [Thyroinaemia in subjects exposed to carbon
disulfide.] Lav. Um., 23: 183-187 (in Italian).
CAVALLERI, A., POLATTI, F., & BOLIS, P. F. (1977) Acute effects of
tetraethylthiuram disulphide on serum levels of hypophyseal
hormones in humans. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski (Finland), 6-10
June 1977, p. 28.
CHERVENKA, C. H. & WILCOX, P. E. (1956) Chemical derivative of
chymotrypsinogen: I: Reaction with carbon disulfide. J. biol.
Chem. 222: 621-634.
CIMBAREVIC, T. K. (1970) [Ceruloplasmin activity in persons exposed to
carbon disulfide.] In: Voprosy diagnostiki, specifideskoj
profilaktiki i terapii professional 'nyh zabolevanij,
Leningrad, 65-67 (in Russian).
CIRLA, A.M., VILLA, A., & TOMASINI, M. (1972) [Study on the incidence
of coronaropathy in workers exposed to carbon disulfide in a
viscose rayon industry.] Med. Lavoro, 63: 431-441 (in Italian).
COHEN, A. E., PAULUS, H. J., KEENAN, R. G., & SCHEEL, L. D. (1958)
Skin absorption of carbon disulfide vapor in rabbits. I.
Associated changes in blood protein and zinc. Arch. ind. Health,
17: 164-169.
COHEN, A. E., SCHEEL, L. D., KOPP, J. F., STOCKELL, F. R., KEENAN, R.
G., MOUNTAIN, J. T., & PAULUS, H. J. (1959) Biochemical
mechanisms in chronic carbon disulfide poisoning. Am. Ind. Hyg.
Assoc. J., 20: 303-323.
CREPET, M., COBBATO, F., & SCANSETTI, G. (1956)] Some clinical
observations on vasculopathy resulting from chronic carbon
disulfide poisoning.] Minerva Med., 47: 379-387 (in Italian).
CULLEN, T. E. (1964) Spectrophotometric determination of
dithiocarbamates residues on food crops. Anal. Chem., 36:
221-224.
DALVI, R. R., POORE, R. E., & NEAL, R. A. (1974) Studies of the
metabolism of carbon disulfide by the rat liver microsomes. Life
Sci., 14: 1785-1796.
DANILOVA, T. N. (1968) [Data on the effect of carbon disulfide on the
coagulation system of the blood.] In: Materially 2-oj
konferencii molodyh naucnyh rabotnikov, Leningrad, pp. 102-104
(in Russian).
DAVIDSON, M. & FEINLEIB, M. (1972) Carbon disulfide poisoning: A
review. Am. Heart J., 83: 100-114.
DELIC, V., GRAOVAC-LEPOSAVIC, LJ., DJURIC, D., BERKES, I., &
DJORDJEVIC, V. (1966) The determination of sulphates in the urine
of workers exposed to carbon disulphide. Med. Lavoro, 57:
458-462.
DELPECH, A. (1856a) Notes sur les accidents que developpe, chez les
ouvriers en caoutchouc, l'inhalation du CS2 en vapeur. Gaz.
Hebd. med. Chir., 3: 40.
DELPECH, A. (1856b) Accidents que developpe, chez les ouvriers en
caoutchouc, l'inhalation du CS2 en vapeur. Union bled., 10:
265.
DELPECH, A. (1863) Industrie du caoutchou de souffle: Recherches sur
l'intoxication speciale que determine le CS2. Ann. Hyg. 2e
sér., 19: 65.
DE MATTEIS, F. (1974) Covalent binding of sulfur to microsomes and
loss of cytochrome P-450 during the oxidative desulfuration of
several chemicals. Mol. Pharmacol., 10: 849-854.
DE MATTEIS, E. & SEAWRIGHT, A. A. (1973) Oxidative metabolism of
carbon disulfide by the rat. Effect of treatments which modify
the liver toxicity of carbon disulfide. Chem. biol. Interact.,
7: 375-388.
DEMUS, H. (1964) [On the reception, chemical transformation and
excretion of carbon disulfide by the human body.] Arch.
Gewerbepath. Gewerbehyg., 20: 507-536 (in German).
DEMUS, H. (1967) The mechanism of absorption, metabolism and excretion
of carbon disulfide in the human body. In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 42-49.
DE NICOLA, P., NICROSINI, F., FASOTTI, C., & DEL FAVERO, A. (1962)
[Comparative studies on the physiopathological picture of
experimental chronic carbon disulfide poisoning and
high-cholesterol diet I. Study of the lipid fractions in serum
and aorta.] G. Geront., 10: 207-220 (in Italian).
DEPARTMENT OF EMPLOYMENT AND PRODUCTIVITY (1968) Methods for
determination of toxic substances in air: Carbon disulfide
vapour. London, HMSO, 10 pp. (DEP Booklet No. 6).
DJERASSI, L. S. & LUMSROSO, R. (1968) Carbon disulphide poisoning with
increased ethereal sulphate excretion. Br. J. ind. Med., 25:
220-222.
DJURIC, D. (1963) [Metabolism of carbon disulphide.] Arh. Hig. Rada,
14: 23-31 (in Serbo-Croat).
DJURIC, D. (1967) Determination of carbon disulphide and its
metabolites in biological material. In: Brieger, H. & Teisinger,
J., ed. Toxicology of carbon disulphide, Amsterdam, Excerpta
Medica Foundation, pp. 52-61.
DJURIC, D. (1971) Some aspects of antabuse, carbon disulphide and
ethyl alcohol metabolism. Arh. Hig. Rada, 22: 171-177.
DJURIC, D., SURDUCKI, N., & BERKES, I. (1965) Iodine-azide test on
urine of persons exposed to carbon disulphide. Br. J. ind. Med.,
22: 321-323.
DJURIC, D., STOJADINOVIC, LJ., BOJOVIC, V., & REZMAN, I. (1967)
Excretion of zinc in the urine of persons exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation, pp.
118-121.
DJURIC, D., POSTIC-GRUJIN, A., GRAOVAC-LEPOSAVIC, L., & DELIC, V.
(1973) Disulfiram as an indicator of human susceptibility to
carbon disulfide excretion of diethyldithiocarbamate sodium in
the urine of workers exposed to CS2 after oral administration of
disulfiram. Arch. environ. Health, 26: 287-289.
DROGCINA, E. A. (1968) [Carbon disulfide poisoning.] In: Professional
'nye bolezni sistemy, Leningrad, Medicina, pp. 129-141 (in
Russian).
DUTKIEWICZ, T. & BARANOWSKA, B. (1967) The significance of carbon
disulphide skin resorption in the evaluation of exposure. In:
Brieger, H. & Teisinger, J., ed. Toxicology of Carbon
Disulphide, Amsterdam, Excerpta Medica Foundation, pp. 50-51.
EFREMOVA, N. F. & UZBEKOV, G. A. (1968) [Study of nitrogen metabolism
in the brains of white rats exposed to chronic carbon disulfide
poisoning.] In: Trudy Rjazanskogo Medicinskogo Instituta, 34:
163-172 (in Russian).
EHRHARDT, W. (1967) Experiences with the employment of women exposed
to carbon disulphide. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 240-244.
EL-GAZZAR, R., EL-SADIK, Y. M., & HUSSEIN, M. (1973) Changes in zinc
and serum proteins due to carbon disulphide exposure. Br. J.
ind. Med., 30: 284-288.
FAHIM, B. A., FAHMY, A.M., & KAMEL, M. Y. (1973) [Biochemical and
hematologic studies on carbon disulfide poisoning.] Z. Ges.
Hyg., 19: 538-541 (in German).
FAITH, W. L., KEYES, D. B., & CLARK, R. L. (1965) Industrial
Chemicals, 3rd ed., New York, John Wiley & Sons, pp. 226-227.
FERRERO, G. P. (1969) [Occurrence of diabetes in workers exposed to
carbon disulfide.] Med. Lavoro, 60: 38-42 (in Italian).
FERRARO, A., JARVIS, G., & FLICKER, D. (1.941) Neuropathologic changes
in experimental carbon disulfide poisoning in cats. Arch.
Pathol., 32: 732-738.
FINKOVA, A., SIMKO, A., JINDRICHOVA, J., KOVARIK, J., PREININGEROVA,
O., KLIMOVA, A., & KORISKO, F. (1973) [Gynaecological problems of
women working in an environment contaminated with carbon
disulfide.] Cesk. gynekol., 38: 535-536 (in Czech).
FINULLI, M. & GHISLANDI, E. (1959) [Occurrence of diabetes in chronic
carbon disulfide poisoning.] Med. Lavoro, 50: 189-192 (in
Italian).
FISCHER, J. & MICHALOVA, C. (1956) [Neurohistological changes caused
by carbon disulfide.] Prac. Lek., 8:16-19 (in Czech).
FOA, V., CASSITO, M. G., FORZI, M., & BULGHERONI, C. (1976) Mental
performance and personality disorders among workers exposed to
carbon disulphide: Comparison between two different rayon plants.
In: Horvarth, M., ed. Adverse effects of environmental chemicals
and psychotropic drugs, neurophysiological and behavioral tests.
Amsterdam, Oxford, New York, Elsevier, Vol. 2, pp. 173-182.
FOREMAN, W. (1886) Notes of a fatal case of poisoning by bisulphide of
carbon. Lancet, 2: 118-119.
FRANCINE, A. P. (1905) Acute carbon disulfide poisoning. Am. Med.,
9: 871.
FRANTIK, E. (1970) The development of motor disturbances in
experimental chronic carbon disulphide intoxication. Med.
Lavoro,, 61: 309-313.
FREUNDT, K. J. (1973) The inhibitory action of inhaled carbon
disulfide on microsomal enzymes as a sensitive restriction of
liver function. In: Horvath, M., ed. Adverse effects of
environmental chemical and psychotropic drugs: Quantitative
interpretation of functional tests, Amsterdam, Oxford, New
York, Elsevier, pp. 161-164.
FREUNDT, K. J. (1974) Ethanol and acetaldehyde levels in alcoholized
rats and man under carbon disulphide exposure. Abstracts of the
III International Symposium on the Toxicology of Carbon
Disulphide, Cairo-Alexandria (Egypt) 4-9 May, 1974, p. 28.
FREUNDT, K. J. (1975) [Carbon disulfide.] In: Henschler, D., ed.
Gesundheitsschadliche Arbeitstoffe, 4th ed., Federal Republic
of Germany, Verlag Chemic Weinheim, pp. 1-20.
FREUNDT, K. J. (1977) Animal experimentation studies on a possible
induction of hepatic microsomal mixed-function oxygenases (MFO)
by inhaled carbon disulphide (CS2). In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 24.
FREUNDT, K. J. & DREHER, W. (1969) Inhibition of drug metabolism by
small concentrations of carbon disulfide. Nauyn-Schmiedebergs
Arch. Pharmakol., 263: 208-209.
FREUNDT, K. J. & HENSCHLER, D. (1971) Inhibition of oxidative drug
metabolism by low doses of inhaled carbon disulfide: Kinetics and
specificity. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Bahja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 9.
FREUNDT, K. J. & KÜRZINGER, R. (1975) Energy potential and hepatic
function in rats under acute exposure to carbon disulphide. Int.
Arch. Arbeitsmed., 34: 269-282.
FREUNDT, K. J. & KUTTNER, P. (1969) Oxidative drug metabolism
following low carbon disulphide concentrations.
Nauyn-Schmiedebergs Arch. Pharmakol., 264: 232-233.
FREUNDT, K. J. & LIEBERWIRTH, H. (1974) Influence of inhaled carbon
disulfide on acetaldehyde production and liver function in
alcoholized man. Nauyn-Schmiedebergs Arch. Pharmakol., 282
(Suppl. R.21).
FREUNDT, K. J. & NETZ, H. (1973) Influence of carbon disulfide and
thiurams on ethanol elimination and acetaldehyde production.
Arch. Pharmakol. exp. Pathol., 277 (Suppl. R18).
FREUNDT, K. J., LIEBALDT, G. P., & SIEBER, K. H. M. (1974a) Effect of
barbiturates on the liver of rats exposed to carbon disulphide
vapor. Int. Arch. Arbeitsmed., 32: 297-303.
FREUNDT, K. J., SCHAUENSURG, K. J., & EICHHORN, P. (1974b) Effect of
acute exposure to carbon disulfide vapor upon some components of
the hepatic microsomal enzyme system in rats. Arch. Toxicol.,
32 (3): 233-240.
FREUNDT, K. J., SCHNAPP, E., & DREHER, W. (1975) Pharmacokinetics of
inhaled carbon disulphide in rats in relation to its inhibitory
effect on the side-chain oxidation of hexobarbital. Int. Arch.
occup. environ. Health, 35: 173-185.
FREUNDT, K. J., LIEBERWIRTH, K., NETZ, H., & POHLMANN, E. (1976) Blood
acetaldehyde in alcoholized rats and humans during inhalation of
carbon disulphide vapor. Int. Arch. occup. environ. Health, 37:
35-46.
FRUSCELLA. R. (1962) [Urinary excretion of 17-ketosteroids and
17-hydroxycorticosteroids in human subjects suffering from carbon
disulfide poisoning.] Clin. ter., 23: 749-755 (in Italian).
GADASKINA, I.D. & ANDREEVA, N. B. (1960) [Some biochemical shifts in
the organism in carbon disulfide poisoning. Free SH groups and
ceruloplasmin in the blood: Copper and zinc metabolism.] Gig.
Tr. Prof Zabol., 13: 28-32 (in Russian).
GAVRILESCU, N. & LILIS, R. (1967) Cardiovascular effects of
long-extended carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 165-167.
GHERASE, G. (1976) Psychological changes in workers exposed to
atmospheres containing carbon disulphide. In: Adverse effects of
environmental chemicals and psychotropic drugs.
Neurophysiological and behavioral tests, Amsterdam, Vol. 2,
334 pp.
GILIOLI, R., BULGHERONI, C., BERTAZZI, P. A., CIRLA, A. M., CASSITTO,
M. G., & TOMASINI, M. (1977) Endocrinological, psychological and
neurological study of carbon disulphide workers. III.
Neurological and neurophysiological findings. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), June 6-10, 1977, p. 13.
GOLDBERG, M. E., JOHNSON, H. E., POZZANI, U. C., & SMYTH, H. F. (1964)
Behavioral response of rats during inhalation of trichlorethylene
and carbon disulphide vapours. Acta Pharmacol., 21: 36-44.
GONDZIK, M. (1971) Histology and histochemistry of rat testicles as
affected by carbon disulphide. Polish med. J., 10: 133-139.
GORDY, S. T. & TRUMPER, M. (1938) CS2 poisoning with a report of six
cases. J. Am. Med. Assoc., 110: 1543.
GORDY, S. T. & TRUMPER, M. (1940) Carbon disulphide poisoning-Report
of 21 cases. Ind. Med., 9: 231-234.
GORNY, R. (1971) The level of pyridoxal phosphate (PLP) in the blood
plasma of rats exposed to carbon disulfide. Biochem. Pharmacol.,
20: 2114-2115.
GORNY, R. (1974) [Effect of prolonged exposure to carbon disulfide on
tryptophan metabolism in rats.] Med. Pr., 25: 21-27 (in
Polish).
GOTO, S. & HOTTA, R. (1967) The medical and hygienic prevention of
carbon disulphide poisoning in Japan. In: Brieger, H. &
Teisinger, J.: ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 219-230.
GOTO, S., HOTTA, R., & SUGIMOTO, K. (1971) Studies on chronic carbon
disulfide poisoning. Int. Arch. Arbeitsmed., 28: 115-126.
GOTO, S., SUGIMOTO, K., HOTTA, R., BABA, T., NAKAMURA, G., & SEKI, Y.
(1977) Regional patterns of angiopathy due to CS2. Egypt. J.
occup. Med., 5: 155-159.
GRAOVAC-LEPOSAVIC, LJ. & JOVICIC, M. (1971) Report on one case of
chronic carbon disulfide intoxication. In: Abstracts of the H
International Symposium on Carbon Disulphide Toxicology, Banjo
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 19.
GRAOVAC-LEPOSAVIC, LJ., DJURIC D., PAVLOVIC, A., & JOVICIC, M. (1967)
The use of iodine-azide test for early diagnosis of carbon
disulphide poisoning. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 62-69.
GRAOVAC-LEPOSAVIC, LJ., JOVICIC, M., SAVIC, S., DJURIC, D., &
POSTIC-GRUJIN, A. (1977) Findings in workers some years after
CS2 poisoning. Egypt. J. occup. Med., 5: 53-60.
GREGORCZYK, J., KOSMIDER, K., WIECKOWSKI, B., & WESOLOWSKA, T. (1975a)
Investigations on the extent of the liver lesion in the rat by
experimental inhalation poisoning with carbon disulfide. I.
Analysis of biochemical changes in the blood serum. Int. Arch.
Arbeitsmed., 34: 65-80.
GREGORCZYK, J., WIECKOWSKI, B., & WESOLOWSKA, T. (1975b)
Investigations on the extent of liver lesion in rats by
experimental inhalation poisoning with carbon disulfide. II.
Analysis of biochemical changes in liver homogenate and its
fractions. Int. Arch. Arbeitsmed., 34: 151-166.
GUARINO, A. & ARCIELLO, G. (1954) [Histological study on coronary
arteries in chronic experimental carbon disulfide poisoning.]
Folia Med. (Napoli) 37: 1022-1044 (in Italian).
HAKKY, U. (1977) Physiological manifestations after exposure to CS2.
Egypt. J. occup. Med., 5: 47-52.
HAMILTON, A. (1925) Industrial poisons in the United States, New
York, Macmillan Co., 590 pp.
HAMILTON, A. (1940) Occupational poisoning in the viscose rayon
industry, Washington, DC, Department of Labor, pp. 1-79 (Bull.
No. 34).
HÄNNINEN, H. (1971) Psychological picture of manifest and latent
carbon disulphide poisoning. Br. J. ind. Med., 28: 374-381.
HÄNNINEN, H. (1974) Behavioral study of the effects of carbon
disulfide. In: Xintaras, C. El, Johnson, B., De Groot, I., ed.
Behavioral toxicology, Washington, DC, Department of Health,
Education, & Welfare, pp. 72-79.
HÄNNINEN, H., NURMINEN, M., TOLONEN, M., & MARTELIN, T. (1978)
Psychological tests as indicators of excessive exposure to carbon
disulfide. Scand. J. Psychol., 19(2): 163-174.
HARA, I. (1958) [On the carbohydrate metabolism in chronic carbon
disulfide poisoning.] Q. J. labour Res. Jpn, 6: 1-20 (in
Japanese).
HARASHIMA, S. & MASUDA, Y. (1962) Quantitative determination of
absorption and elimination of carbon disulfide through different
channels in human body. Int. Arch. Gewerbepath. Gewerbehyg.,
19: 263-269.
HARASHIMA, S., TOYAMA, T., & SAKURAI, T. (1960) Serum cholesterol
level of viscose rayon workers. Keijo J. Med., 9: 81-90.
HASSMAN, P., SIMKO, A., JINDRICHOVA, J., HEROUT, V., & HRADSKY, M.
(1967) The problem of chronic gastritis in artificial fibres
industry. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Amsterdam, Excerpta Medica Foundation,
pp. 182-189.
HERNBERG, S. & NORDMAN, C-H. (1969) Failure to find increased
excretion of zinc or magnesium in humans exposed to carbon
disulphide. Med. Lavoro, 60: 163-168.
HERNSERG, S., MOWE, G., VIRKOLA, P., PARTANEN, T., & NORDMAS, C-H.
(1969) Magnesium and zinc values of erythrocytes and plasma for
workers exposed to carbon disulfide. Work Environ. Health, 6:
9-13.
HERNSERG, S., PARTANEN, T., NORDMAN, C-H., & SUMARI, P. (1970)
Coronary heart disease among workers exposed to carbon
disulphide. Br. J. ind. Med., 27: 313-325.
HERNBERG, S., NORDMAN, C-H., PARTANEN, T., CHRISTIANSEN, V., &
VIRKOLA, P. (1971) Blood lipids, glucose tolerance and plasma
creatinine in workers exposed to carbon disulphide. Work
Environ. Health, 8: 11-16.
HERNBERG, S., NURMINEN, M., & TOLONEN, M. (1973) Excess mortality from
coronary heart disease in viscose rayon workers exposed to carbon
disulfide. Work Environ. Health, 10: 93-99.
HERNBERG, S., TOLONEN, M., & NURMINEN, M. (1976) Eight-year follow-up
of viscose rayon workers exposed to carbon disulfide. Scand. J.
Work Environ. Health, 2: 27-30.
HIGASHIDA, T., YAMAMOTO, T., & SHINDO, S. (1959) [Electromyographic
study of the effects of carbon disulfide administration on spinal
reflex.] Brain Nerve, 11(4): 253-260 (in Japanese).
HOCKWIN, O. & SAVIC, S. (1968) [Reversibility of biochemical changes
in lease and retina of the rabbit's eye in carbon disulfide
poisoning.] Albrecht Graefs Arch. klin. exp. Ophthalmol., 175:
7-12 (in German).
HOLASOVA, P. (1969) [Observations on a group of children from an area
polluted by carbon disulfide and hydrogen sulfide exhalation
compared with a control group of children.] Cs. Hyg., 14:
260-265 (in Czech).
HORVATH, M. & MIKISKA, A. (1957) [The influence of carbon disulfide on
the CNS of rabbits as seen in the EEG.] Prat. Lek., 9: 22-28
(in Czech).
HOTTA, R. & GOTO, S. (1971) A fluoroscein angiographic study on
microangiopathia sulfocarbonica Jpn. J. Ophthal., 15: 132-139.
HOTTA, R. & SAVIC, S. (1972) Fluorescein angiographic and
ophthalmodynamometric study on chronic carbon disulfide
poisoning. Med. Lavoro, 63: 442-447.
HUEPER, M. C. (1936) Etiologic studies on the formation of skin
blisters in viscose workers. J. ind. Hyg., 18: 432-447.
IVANOVA, F. G. (1967) [Some data on the status of erythropoiesis on
exposure to low concentrations of carbon disulfide.] In: Voprosy
gigieny truda i profpatologii, Leningrad, pp. 247-248 (in
Russian).
JAKUBOWSKI, M. (1968) [Studies of the conversion of carbon disulphide
to compounds catalysing the iodine-azide reaction. I.
Chromatographic studies.] Med. Pr. 19: 244-251 (in Polish).
JAKUBOWSKI, M. ( 1971) [Studies on the carbon disulfide metabolism of
compounds catalyzing the iodine azide reaction.] Med. Pr. 22:
195-206 (in Polish).
JÄRVISALO, J. & DE MATTEIS, F. (1977) Carbon disulphide induced loss
of liver microsomal haem and cytochrome P-450. In: Tolonen, M.,
ed. Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski (Finland), 6-10 June, 1977, p. 23.
JÄRVISALO, J., SAVOLAINEN, H., ELOVAARA, E., & VAINIO, H. (1977) The
in vivo toxicity of CS2 to liver microsomes: Binding of
labelled CS2 and changes of the microsomal enzyme activities.
Acta Pharmacol. Toxicol., 40: 329-336.
JUMP, H. D. & CRUICE, J. M. (1904) Chronic poisoning from bisulphide
of carbon. Univ. Penn. med. Bull., 17: 193-196.
JUNTUNEN, J., HALTIA, M., & LINNOILA, I. (1974) Histochemically
demonstrable non-specific cholinesterase as an indicator of
peripheral nerve lesion in carbon disulfide induced
polyneuropathy. Acta Neuropathol. (Berl.), 29: 361-366.
JUNTUNEN, J., LINNOILA, I., & HALTIA, M. (1977) Histochemical and
electron microscopic observations on the myoneural junctions of
rats with carbon disulfide induced polyneuropathy. Scand. J.
Work Environ. Health, 3: 1-15.
KARAJOVIC, D., GRAOVAC-LEPOSAVIC, LJ., & SURADNICI, V. J. (1964) [The
influence of CS2 on the health of workers in factory in Serbia.]
Arh. Hig. Rada, 15: 87-92 (in Serbo-Croat).
KLIMKOVA-DEUTSCHOVA, E. (1965) [Early diagnosis in industrial
neurology.] S.Z.N., Praha 1965, 260 pp. (in Czech).
KNAVE, B., KOLMODIN-HEDMAN, B., PERSSON, H. E., & GOLDBERG, J. M.
(1974) Chronic exposure to carbon disulfide: Effects on
occupationally exposed workers with special reference to the
nervous system. Work Environ. Health, 11: 49-58.
KOELSCHS HANDBUCH DER BERUFSERKRANKUNGEN (1972) Jena, Gustav Fischer
Verlag. Vol. II, pp. 270-275.
KOESTER, G. (1899) [Contribution to the field of chronic CS:
intoxication.] Arch. Psychiat., 32: 606 (in German).
KOLMODIN-HEDMAN, B. (1976) Iodine-azide test in a Swedish viscose
factory related to air concentration of CS2 and to health.
Arbete och Halea, No. 8. pp. 23-28.
KOMRSKA, M. (1967) L'importance de la rheoencephalographie pour le
diagnostic d'intoxication par le sulfure de carbone. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulfide,
Amsterdam, Excerpta Medica Foundation, pp. 140-144.
KOPECKY, J. (1973) [Metabolism of carbon disulfide in rats.] Acta
Hyg. Epidemiol. Microbiol. (Prague), Suppl. 12, pp. 3-6
(in Czech).
KOZIOL, A., KULCZYCKA, H., & ROZEWICKI, S. (1971) [Development of
children, whose mothers were professionally exposed to carbon
disulfide.] Med. Pr., 22: 511-514 (in Polish).
KROLIKOWSKA, W. & RUCINSKA, Z. (1959) EEG examinations in chronic
professional intoxication by carbon disulfide. Electrortceph.,
clin. Neurophysiol., 11: 180 (Abstract No. 9).
KUBOTA, J. (1967) Historical view of carbon disulfide poisoning in the
Japanese viscose rayon industry. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulfide, Amsterdam, Excerpta Medica
Foundation, pp. 192-196.
KUJALOVA, V. (1971) The effect of carbon disulfide on vitamin B6
metabolism. In: Abstracts of the II International Symposium on
Toxicology of Carbon Disulfide, Banja Koviljaca (Yugoslavia),
25-28 May, 1971, p. 30.
KUJALOVA, V. (1973) [Changes in the pyridoxinemetabolism and in the
blood levels of ceruloplasmin in humans exposed to carbon
disulfide.] Acta Hyg. Epidemiol. Microbiol., Suppl. 12 (in
Czech).
KUJALOVA V. & TINTERA, J. (1970) The effect of carbon disulfide on
tryptophan metabolism. Med. Lavoro, 61: 227-231.
KÜRZINGER, R. & FREUNDT, K. J. (1969) Changes in hepatic functions
following low carbon disulfide concentration.
Naunyn-Schmiedebergs Arch. Pharmakol., 264: 261-262.
LANCRANJAN, I. (1972) Alterations of spermatic liquid in patients
chronically poisoned by carbon disulfide. Med. Lavoro, 63:
29-33.
LANCRANJAN, I., POPESCU, H. I., & KLEPSCH, I. (1969) Changes of the
gonadic function in chronic carbon disulfide poisoning. Med.
Lavoro, 60: 566-571.
LANCRANJAN, I., FLOREA, O., & PETRESCU, M. (1971) Corticosuprarenal
insufficiency as a feature of carbon disulfide poisoning. Med.
Lavoro, 62: 557-561.
LANCRANJAN, I., SUKMANSKY, M., STANUCA, L., ANTONESCU, C., & POPESCU,
H. I. (1972) Study of the thyroid function in chronic carbon
disulfide poisoning. Med. Lavoro, 63: 123-125.
LANGELEZ, A. (1946) Viscose et sulfure de carbone. Arch. Belg. Med.
Soc. Hyg. Rev. Pathol., 4: 67-85.
LAUDENHAIMER, R. (1899) [The carbon disulfide intoxication of rubber
workers.] Leipzig, Veit & Co., 168 pp. (in German).
LAURMAN, W. & WRONSKA-NOFER, T. (1977) [Effect of carbon disulfide on
the amount of cholesterol in the tissues of rats.] Med. Pr.,
28: 77-82 (in Polish).
LAZAREV, N.J., ABRAMOVA, ZH. I., & CERNY, Z. C. (1965) [On possible
adverse reactions following use of some drugs by persons handling
carbon disulfide.] Gig. Tr. Prof. ZaboL, 9: 24-27 (in Russian).
LEITHE, W. (1971) Determination of carbon disulfide at the work site.
The analysis of air pollutant, 2nd ed., Ann Arbor, MI, Humphrey
Science Pub., pp. 228-229.
LEWEY, F. H. (1941) Neurological, medical and biochemical signs and
symptoms indicating chronic industrial carbon disulfide
absorption. Ann. int. Med., 15: 869-883.
LEWEY, W. E., ALPERS, B. J., BELLET, S., GRESKOFF, A. J., DRABLEIN, D.
L., ERICH, W. E., FRANK, J. H., JONAS, L., MCDONALD, R.,
MONTGOMERY, E. J., & REINHOLD, J. G. (1941) Experimental chronic
poisoning in dogs, clinical, biochemical and pathological study.
J. ind. Hyg., 23: 415-436.
LILIS, R. (1974) Behavioral effects of occupational carbon disulfide
exposure. In: Xintaras, C., Johnson, B. L., & de Groot, I., ed.
Behavioral toxicology, early detection of occupational hazards,
Cincinnati, US DHEW, Public Health Service, Center for Disease
Control, National Institute for Occupational Safety and Health,
pp. 51-59 (NIOSH 74-126).
LINIECKI, J. (1960) The effect of chronic carbon disulfide poisoning
on thiamine balance in rats. J. Hyg. Epidemiol. Microbiol.
Immunol. 4: 212-222.
LINNOILA, I., HALTIA, M., SEPPÄLÄINEN, A.M., & PALO, J. (1975)
Experimental carbon disulphide poisoning. Morphological and
neurophysiological studies. In: Körney, S., Tariska, S., &
Gosztonyi, G., ed. Proceedings of the VII International Congress
on Neuropathology Académiai Kiado, Budapest 1-7 September 1975,
pp. 383-386.
LUKAS, E. (1969) Conduction speed of peripheral nerves in persons
exposed to carbon disulfide (Electromyography study). Int. J.
clin. Ther. Toxicol., 2: 354-358.
LUKAS, E. (1970) Stimulation electromyography in experimental
toxicology (Carbon disulphide neuropathy in rats). Med. Lavoro,
61: 302-308.
LUKAS, E. (1973) [Some important factors in the development of
experimental carbon disulfide neuropathy in rats.] Cs. Neurol.
Neurochir., 36: 169-173 (in Czech).
LUKAS, E. (1974) Copper and zinc levels in peripheral nerve tissues of
rats with experimental carbon disulfide neuropathy. In:
Abstracts of the III International Symposium on Toxicology of
Carbon Disulphide, Cairo-Alexandria, Egypt, 4-9 May, 1974,
p. 8.
LUKAS, E. & HROMADKA, M. (1977) Conduction velocity of the ulnar and
tibial nerves in workers occupationally exposed to low
concentrations of carbon disulphide. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, June 6-10, 1977, p. 18.
LUKAS, E., KOTAS, P, & OBRUSNIK, I. (1974) Copper and zinc levels in
peripheral nerve tissues of rats with experimental carbon
disulphide neuropathy. Br. J. ind. Med., 31: 288-291.
LUKAS, E., KUSALOVA, V., & KOCINOVA, F. (1975) Activity of
ceruloplasmin in the serum of rats with experimental carbon
disulphide neuropathy. In: McCallum, R. I., ed. Abstracts of the
XVIII International Congress on Occupational Health, Brighton,
England, 14-19 September, 1975, pp. 299-300.
LUKAS, E., TEISINGER, J, Jr, & BASS, A. (1977) Activity of muscle
enzymes in rats with experimentally developed carbon disulphide
polyneuropathy. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 19.
LYSINA, G. G. (1967) Some changes in the internal organs due to the
effects of carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 179-181.
MACK, T., FREUNDT, K. J., & HENSCHLER, D. (1974) Inhibition of
oxidative N-demethylation in man by low doses of inhaled carbon
disulphide. Biochem. Pharmacol., 23: 607-614.
MADLO, Z. & SOUCEK, B. (1953) [Absorption, metabolism, and action of
carbon disulfide in the organism. VII. Inhibition of serum
cholinesterase by carbon disulfide.] Prac. Lek., 6: 312-313 (in
Czech).
MADLO, Z., PEIZKER, & SOUCEK, B. (1953) [The absorption, metabolism
and action of carbon disulphide in the organism. V. The effect of
carbon disulfide on tissue respiration.] Prac. Lek., 5: 263-266
(in Czech).
MAGISTRETTI, M. & PEIRONE, E. (1961)The action of carbon disulphide on
cerebral monoamine oxidase. Med. Lavoro, 52: 1-10.
MAGOS, L. (1972) Relevancy of bivalent sulphur excretion to carbon
disulphide exposure in different metabolic conditions. Br. J.
ind. Med., 29: 90-94.
MAGOS, L. (1975) The clinical and experimental aspects of carbon
disulphide intoxication. Rev. environ. Health, 11: 65-80.
MAGOS, L. & BUTLER, H. (1972) Effect of phenobarbitone and starvation
on hepatotoxicity in rats exposed to carbon disulfide vapor.
Br. J. ind. Med., 29: 95-98.
MAGOS, L. & JARVIS, J. A. E. (1970a) The effects of carbon disulphide
exposure on brain catecholamines in rats. J. Pharm. Pharmacol.,
29: 936-938.
MAGOS, L. & JARVIS, J. A. E. (1970b) The effects of carbon disulfide
exposure on brain catecholamines in rats. Br. J. Pharmacol.,
39: 26-33.
MAGOS, J., GREEN, A., & JARVIS, J. A. E. (1974) Half life of CS2 in
rats in relation to its effect on brain catecholamines. Int.
Arch. Arbeitsmed., 32: 289-296.
MANU, P, LANCRANJAN, I., & ZAMFIRESCU, Z. (1970) Investigation of
serum LDH and its isoenzymatic model in carbon disulphide
polyneuritis. Int. Arch. Arbeitsmed., 26: 45-49.
MANU, P., LILIS, R., NESTORESCU, B., LANCRANJAN, I., ROSENTA, A.,
IONESCU, S., & KAUFMAN, G. (1971) Effects of carbon disulfide on
cholesterol and ß-lipoprotein levels in young men. In: Abstracts
of the II International Symposium on Toxicology of Carbon
Disulfide, Banja Koviljaca, Yugoslavia, 25-28 May, 1971, pp.
28-29.
MARTINO, P., COSCIA, G. C., & PERRELLI, G. (1963) [Lipedaemia in
carbon disulfide poisoning. Preliminary observations.[ Folia
Med., 46: 1110-1114 (in Italian).
MARTINO, P., PERRELLI, G., & COSCIA, G. C. (1964) [Lipedaemia in
carbon disulfide poisoning.[ Arch. Sci. Med., 117: 124-130 (in
Italian).
MARTYNOVA, A. P., ZELENKIN, A. N., GOLJAKOVA, L. P., MILKOV, L. E.,
GRODETSKAJA, N. S, & AGADZANOVA, A. A. (1976)[Clinical hygienic
and experimental investigations of the action on the body of
small concentrations of carbon disulfide.] J. Hyg. Sanit., 5:
25-28 (in Russian).
MASSOUD, A. E. E., SADEK, S., EL HAWARY, M. F., & HASSANEIN, M. (1971)
Changes in the electrocardiogram and lung function in workers
exposed to carbon disulfide. In: Abstracts of the II
International Symposium on Toxicology of Carbon Disulfide, Banja
Koviljaca, Yugoslavia, 25-28 May, 1971, p. 6.
MASSOUD, A. A. E., ABDEL KARIM, A. H., & FARES, R. (in press) Serum
thyroxine, serum cholesterol and its fractions in workers exposed
to carbon disulphide. Egypt. J. occup. Med.
MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., VISCONTI, E., IOVICIC, M., &
CAVALLERI, A. (1966a) [Cerebrovascular damage in young workers
exposed to carbon disulfide.] Med. Lavoro, 57: 701-708 (in
Italian).
MAUGERI, U., IOVICIC, M., CAVALLERI, A., GRAOVAC-LEPOSAVIC, LJ.,
VISCONTI, E., & REZMAN, I. (1966b) [Peripheral vascular damage in
young workers exposed to carbon disulfide.] Med. Lavoro, 57:
709-714 (in Italian).
MAUGERI, U., VISCONTI, E., & CAVALLERI, A. (1966c)] Vascular
alterations in the retina of young workers exposed to carbon
disulfide.] Med. Lavoro, 57: 741-745 (in Italian).
MAUGERI, U., CAVALLERI, A., & VISCONTI, E. (1966d)
[Ophthalmodynamography in occupational carbon disulfide
poisoning.] Med. Lavoro, 57: 730-740 (in Italian).
MAUGERI, U., CAVELLERI, A., & VISCONTI, E. (1967)
Ophthalmodynamographic study in young workers exposed to carbon
disulphide. In: Brieger, H. & Teisinger, J., ed. Toxicology of
carbon disulphide, Excerpta Medica Foundation, Amsterdam, pp.
150-152.
MAUGERI, CAVALLERI, A., MAUGERI, U., & TACCOLA, A. (1971) New aspects
in the pathogenetic mechanism of carbon disulphide toxic action.
Med. Lavoro, 62: 398-403.
MCCAMMON, C., Jr, QUINN, M. P., & KUEPEL, R. E. (1975) A charcoal
sampling method and a gas chromatographic analytical procedure
for carbon disulfide. Am. Ind. Hyg. Assoc. J., 36: 618-625.
MCKEE, R. W. (1941) Solubility of carbon disulphide vapor in body
fluids and tissues. J. ind. Hyg., 23: 484-489.
MCKENNA, M. J. & DI SRTEFANO, V. (1975) A proposed mechanism of the
action of carbon disulfide on dopamine-ß-hydroxylase. Toxicol.
appl. Pharmacol., 33: 137.
MELISSINOS, J.-C. & JACOBIDES, G. (1967) Sur quelques formes nerveuses
d'intoxication observées en Grece chez des sujets exposeés au
sulfure de carbone. Arch. Mal. Prof., 28: 605-611.
MERLEVEDE, E. (1951) [Toxicology of carbon disulfide.] Sinte
Catharina, Brugge, 180 pp. (in Dutch).
MIHAIL, G., MIHAILA, D., BRANISTEANU, D., BODNAR, J., & CIOATA, V.
(1968) Manifestations cliniques, fonctionnelles et biochimiques
des stades precoces du sulfocarbonisme. Arch. Mal. Prof., 29:
109-120.
MILIC, S. (1965) [Some observations concerning the determination of
CS2 in the urine of exposed workers.] In: Zbornik radova o
toksikologiji CS2 (Proceedings in Yugoslavian symposium on CS2
toxicology, Loznica, 1965, p. 41-42 (in Serbo-Croat).
MILKOV, L. E., MONAENKOVA, A.M., BYALKO, N. K., GLOTOVA, K. V.,
VERETINSKAJA, A. G., ALI-ZADE, K. A., MAKARENKO, Y. L.,
NIKOLSKAJA, O. I., & KURSAKOV, A. V. (1970) [On the initial forms
of chronic carbon disulfide poisoning.] Gig. Tr. Prof. Zabol.,
14: 28-32 (in Russian).
MINDEN, H., GOHLKE, R., & ROTHE, R. (1967) [Contribution to the
pathogenesis of chronic carbon disulfide poisoning.] Z. Ges.
Hyg. Grenz., 13: 815-822 (in German).
MISIAKIEWICZ, Z., SZULINSKA, O., & CHYBA, A. (1972) [Influence of a
carbon disulfide, hydrogen sulfide mixture in air on white rats
under the conditions of permanent exposure.] Rocz. Panstw. Zakl.
Hig., 23: 465-475 (in Polish).
MOREO, S. & CANDURA, F. (1960) [The behaviour of serum-clotting
factors in animals treated with sodium arsenite, carbon
disulfide, cadmium chloride and lead acetate.] Boll. Soc. Med.
Chir., (Pavia), 74: 447-455 (in Italian).
MOWÉ, G. (1971) Coronary heart disease and occupational exposure to
carbon disulfide. In: Abstracts of the III International
symposium on toxicology of carbon disulfide, Banja Koviljaca,
Yugoslavia, 25-28 May, 1971, p. 7.
MURASKO, V. A. (1975) [The morbidity of the digestive organs through
working in contact with carbon disulfide in the viscose
industry.] Vrach. Delo, No. 4, pp. 113-117 (in Russian).
NAKAMURA, K., HARADA, M., TATETSU, S., MIYAGAWA, T., TSUKAYAMA, T.,
KAWANO, K., KABASHIMA, K., HIRATA, M., & TOYA, G. (1974) A
clinical study on chronic carbon disulfide poisoning. Psychiatr.
Neuro. Jpn., 76: 243-273.
NEAL, R. A., KATAMAKI, T., HUNTER, A. L., & CATIGNANI, G. (1976)
Monooxygenase catalyzed activation of thiono-sulfur-containing
compounds to reactive intermediates. Hoppe-Seyler's Z. physiol.
Chem., 357: 1044.
NESSWETHA, L. & NESSWETHA, W. (1967) The clinical evaluation of
compensated carbon disulphide intoxications in the German Federal
Republic since 1948. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Excerpta Medica Foundation,
Amsterdam, pp. 214-218.
NIOSH (1977) Criteria for a recommended standard...occupational
exposure to carbon disulfide, Washington, DC, US Dept Health,
Education, and Welfare, National Institute for Occupational
Safety and Health, 186 pp.
NOFER, J. & WRONSKA-NOFER, T. (1966) Elimination des certains
metabolites de niacine par rapport au témps dexposition et la
close sulfure de carbone. In: Proceedings of the IX
International Congress on Occupational Health, Vienna, 1966,
pp. 355-358 (A III 53).
NOFER, J., CHOJNOWSKI, J., KAWECKA, M., KIEC, E., WRONSKA-SZPAKOWA,
T., & WYSZOMIRSKA, Z. (1961) [Effect of occupational hazard to
carbon disulphide on the pathogenesis of arteriosclerosis.]
Med. Pr. 12: 101-118 (in Polish).
NORO, L. (1944) [Hydrogen and carbon disulphide intoxications.] Nord.
Med., 24: 2015-2020 (in Swedish).
NOVAK, L., DJURIC, D., & FRIEDMAN, V. (1968) Specificity of the
iodine-azide test for carbon disulphide exposure. I. Urinary
excretion of sulfur compounds in persons treated with disulfiram.
Arch. environ. Health, 19: 473-477.
NUNZIATE, CESARO, A., SIPALA, E., & ZUNINI, E. (1952) [Carbon
disulfide poisoning.] Folia Med., 35: 661-716 (in Italian).
NURMINEN, M. (1976) Survival experience of a cohort of carbon
disulphide exposed workers from an eight-year prospective
follow-up period. Int. J. Epidemiol., 5: 179-185.
OLIENACZ, W., PLAMIENIAKA Z., SZYMANKA, J. (1964) [Excretion of
17-ketosteroids in chronic carbon disulphide poisoning.]
Med. Pt. 15: 155-157 (in Polish).
PATERNI, L., PUSIC, G., & TEODORI, S. (1958) [Chronic carbon disulfide
poisoning and arteriosclerosis from high-cholesterol diet in
rabbits.] Folia Med., 41: 705-722 (in Italian).
PAULUS, H. J., LIPPMAN, M., & COHEN, A. E. (1957) Evaluation of
potential health hazards in fumigation of shelled corn with a
mixture of carbon disulfide and carbon tetrachloride. Am. Ind.
Hyg. Assoc. J., 18: 345-350.
PERGAL, M., VUKOJEVIC, N., GIRIN-POPOV, N., DJURIC, D., & BOJOVIC, T.
(1972a) Carbon disulfide metabolites excreted in the urine of
exposed workers. I. Isolation and identification of
2-mercapto-2-thiazolinone-5. Arch. environ. Health, 25: 38-41.
PERGAL, M., VUKOJEVIC, N., & DJURIC, D. (1972b) II. Isolation and
identification of thiocarbamide. Arch. environ. Health, 25:
42-44.
PERGAL, M. N., KOBILAROV, J., RALETIC, A., POSTICS-GRUJIN, A., &
DJURIC, D. (1977a) Determination of small quantities of
thiocarbamate in urine. Egypt. J. Decup. Med., 5: 251-257.
PERGAL, M., RALETIC, J., & DURIC, D. (1977b) Identification of the
third metabolite, giving positive iodine-azide reaction in the
urine of workers exposed to CS2. A preliminary report. In:
Tolonen, M., ed. Abstracts of the IV International Symposium on
Occupational Health in the Production of Artificial Fibres,
Helsinki and Valkeakoski, Finland, 6-10 June, 1977, p. 29.
PETERSON, F. (1892) Three cases of acute mania from inhaling carbon
bisulphide. Boston Med. Surg., 127: 325-326.
PETROV, M. V. (1969)] Course and termination of pregnancy in women
working in viscose industry.] Pediatr. Akush. Ginekol., 3:
50-52 (in Russian).
PETROVIC, D. & DJURIC, D. (1965) [Carbon disulfide in exhaled air of
exposed workers.] In: Zbornik radova o toksikologiji ugljen
disulfida, Loznica, pp. 35-38. (in Serbo-Croat).
PETROVIC, D. & DJURIC, D. (1966) [Carbon disulfide in the air exhaled
by exposed workers.] Arh. Hig. Rada, 17: 159-163 (in
Serbo-Croat).
PETRUN, N.M. (1967) [The effect of carbon disulfide on certain
biochemical indices of the status of the organism in its action
via the skin.] Gig. Tr. Prof. Zabol., 11: 50-53 (in Russian).
PEYTON, T. O., STEEL, R. V., MABEY, W. R., CARLIN, A., & PINES, A.
(1976) Literature review and environmental assessment of carbon
disulfide and carbonyl sulfide. Menlo Park, CA Stanford Research
Institute, pp. 1-65. (Project no EGU 3570, US Environmental
Protection Agency, Publication No. 257-947).
PIROTSKAJA, E. N. (1972) [Changes in the protein-forming function of
the liver in workers exposed to carbon disulfide.] Vrach. Delo,
9: 132-134 (in Russian).
PREROVSKA, I. & ROTH, Z. (1968) [An observation of personnel in the
carbon disulphide hazard with special reference to the
development of arteriosclerosis.] Prac. Lek., 20: 6-10 (in
Czech).
PREROVSKA, I. & ZVOLSKY, P. (1973) [The health status in workers with
chronic carbon disulphide poisoning in five to nineteen years
after ascertaining the disease.] Prac. Lek., 25: 42-46 (in
Czech).
PREROVSKA, I., VANECEK, R., & KUBAT, K. (1961) Effect of carbon
disulfide on experimental arteriosclerosis in the rabbit. Acta
Univ. Carol., 14: 177-185.
QUARELLI, G. (1928)] Occupational CS2 poisoning.] In: Bericht. d. 5.
Internat. Kongr. f. Berufskrankh., Budapest, 1928 (in Italian).
QUARELLI, G. (1934)] Occupational carbon disulfide poisoning.] Russ.
Providenza Soc., 21: 10-73 (in Italian).
RANELLETTI, A. (1931)] Occupational carbon disulfide poisoning.]
Arch. Gewerbepath. Gewerbehyg., 2: 664-675 (in German).
RANELLETTI, A. (1933)] Occupational carbon disulfide poisoning.] In:
La Grafica Moderna, Ed. Torino (in Italian).
RAITTA, C. & TOLONEN, M. (1975) Ocular pulse wave in workers exposed
to carbon disulphide. Albrecht. Graefes Arch. Ophthal., 195:
149-154.
RAITTA, C., TOLONEN, M., & NURMINEN, M. (1974) Microcirculation of
ocular fundus in viscose rayon workers exposed to carbon
disulfide. Albrecht. Graefes Arch. Ophthal., 191: 151-164.
RECHENSERG, H. K. (1957)] Carbon disulfide poisoning and the
sulfocarbotoxic vascular Late-syndrome.] Arch. Gewerbepath.
Gewerbehyg., 15: 487-530 (in German).
RUIKKA, I. (1959) Postprandial serum turbidity in chronic carbon
disulfide poisoning a comparison of viscose rayon workers with
healthy and atherosclerotic men. Ann. Med. intern. Fenn., 48:
225-237.
SAITA, G. & GATTONI, L. (1957) [The blood coagulation process in
chronic carbon disulfide poisoning.] Med. Lavoro, 48: 229-237
(in Italian).
SAITA, G., SBERTOLI, C., & FARINA, G. F. (1964) Modifications in blood
coagulation and lipid metabolism produced by carbon disulfide.
Med. Lavoro, 55: 721-734.
SAKURAI, H. (1972) Hypertension induced by the occupational exposure
to carbon disulfide. In: Abstracts of the XVII International
congress on occupational health. Buenos Aires, 11-16 September,
1972, p. 182.
SAVIC, S. M. (1967) Influence of carbon disulfide on the eye. Arch.
environ. Health, 14: 325-326.
SAVIC, S. & IOVICIC, M. (1965) [Keratoconjunctivitis (causome) in
workers employed in the production of viscose fibres.] In:
Zbornik Radova o toksikologiji CS2, Loznica, Yugoslavia 1965,
pp. 115-121 (in Serbo-Croat).
SAVOLAINEN, H. & VAINIO, H. (1976) High binding of CS2 sulphur in
spinal cord axonal fraction. Acta Neuropathol. (Berlin), 36:
251-257.
SAVOLAINEN, H., JÄRVISALO, J., & VAINIO, H. (1977a) Specific binding
of CS2 metabolites to microsomal proteins in rat liver. Acta
Pharmacol. Toxicol., 41: 94-96.
SAVOLAINEN, H., JÄRVISALO, J., ELOVAARA,. E., & VAINIO, H. (1977b) The
binding of CS2 in central nervous system of control and
phenobarbitone-pretreated rats. Toxicology, 7: 207-214.
SAVOLAINEN, H., LEHTONEN, E., & VAINIO, H. (1977c) CS2 binding to rat
spinal neurofilaments. Acta Neuropathol. (Berl.), 37: 219-223.
SBERTOLI, C., FARINA, G., & SAITA, G. (1969) [Three cases of chronic
carbon disulfide poisoning with giomerulonephrotic syndrome.[
Med. Lavoro, 60: 28-37 (in Italian).
SCHEEL, L. D. (1965) Biological changes involving metal ion shifts.
Am. ind. Hyg. J., 26: 585-592.
SCHEEL, L. D. (1967) Experimental carbon Disulfide poisoning in
rabbits-Its mechanism and similarities with human case report.
In: Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulfide, Amsterdam, Excerpta Medica Foundation, pp. 107-115.
SCHEEL, L. D., KEENAN, R. G., MOUNTAIN,, J. T., KopP, J., HOLTZ, J., &
KILLENS, R. (1960) The effect of dietary metal ions on carbon
disulfide toxicity. In: Proceedings of the 13th International
congress on occupational health, New York, 1960, pp. 783-784.
SCHILLING, R. S. F. (1970) Coronary heart disease in viscose rayon
workers. Am. Heart J., 80: 1-2.
SCHNEIDER, H. (1976) In: Adverse effects of environmental chemicals
and psychotropic drugs. Neurophysiological and behavioural
tests. Vol. 2, Amsterdam, 1976.
SCHÜTZ, H. (1970) Equipment and systems for ambient air monitoring,
Frankfurt/Maine, Hartmann & Braun, Mess und Regeltechnik.
SEPPÄLÄINEN, A.M. (1977) Neurophysiological findings in workers
exposed to carbon disulphide-A follow-up. In: Tolonen, M., ed.
Abstracts of the IV International Symposium on Occupational
Health in the Production of Artificial Fibres, Helsinki and
Valkeakoski, Finland, 6-10 June, 1977, p. 17.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (1975) Electrophysiological studies
of rabbits in long-term exposure to carbon disulfide. Scand. J.
Work Environ. Health, 1: 178-183.
SEPPÄLÄINEN, A.M. & LINNOILA, I. (19745) Electrophysiological findings
in rats with experimental carbon disulphide neuropathy.
Neuropath. appl. Neurobiol., 2: 209-216.
SEPPÄLÄINEN, A.M. & TOLONEN, M. (1974) Neurotoxicity of long-term
exposure to carbon disulfide in the viscose rayon industry. A
neurophysiological study. Work Environ. Health, 11: 145-153.
SEPPÄLÄINEN, A.M., TOLONEN, M., KARLI, P., HÄNNÄINEN, H., & HERNBERG,
S. (1972) Neurophysiological findings in chronic carbon disulfide
poisoning. A descriptive study. Work Environ. Health, 9: 71-75.
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49
(in Japanese).
SETO, Y. (1958) Experimental studies on the influences of carbon
disulfide on the electroretinogram. Report II: the ERG on the
rabbits of acute exposure. Jpn. J. Ophthalmol., 62(8): 39-49.
(in Japanese).
SIHTOLA, H. (1976). [Stages in the development of the SINI-viscose
process.] Tutkimusja tekniikka, 7/8: 63-69 (in Finnish).
SIHTOLA, H. (1977) The SINI viscose process means less chemicals and
less pollution. In; Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 43.
SKALICKA, B. (1967) [Sulfur-containing compounds after administration
of tetraethylthiuramdisulfde.] Prac. Lek., 19: 408-412 (in
Czech).
SOKAL, J. A. (1973) Effect of chronic exposure to carbon disulphide
upon some components of the electron transport system in rat
liver microsome. Biochem. Pharmacol., 22: 129-132.
SOUCEK, B. (1957) [Transformation of carbon disulfide in the
organism.] J. Hyg. Epidemiol. (Praha), 1: 10-22 (in German).
SOUCEK, B. (1959) [Absorption and elimination of carbon disulfide.]
Prac. Lek., 11: 403-407 (in Czech).
SOUCEK, B. (1960a) [Pulmonary elimination of carbon disulfide after
intraperitoneal administration.] Farmakol. Toksikol., 23:(1):
74-84 (in Russian).
SOUCEK, B. (1960b) [The effect of carbon disulphide on the serum
cholesterol and cholesterol ester level.] Prac. Lek., 12:
243-248 (in Czech).
SOUCEK, B. & MADLO, Z. (1953) [Absorption, metabolism and action of
carbon disulphide in the organism. VI. Attempt at chromatographic
proof of the reaction of carbon disulphide with aminoacids in the
blood.] Prac. Lek., 5: 309-311 (in Czech).
SOUCEK, B. & MADLO, Z. (1954) [Absorption, metabolism and action of
carbon disulphide in the organism. VIII. Reaction of carbon
trisulphide with blood in vitro.] Prac. Lek., 6: 11-17 (in
Czech).
SOUCEK, B. & MADLO, Z. (1955) Determination of sulfhydryl groups in
guineapig serum after exposure to carbon disulfide. Bull. Soc.
Chim. Biol., (Paris), 37: 1373-1378.
SOUCEK, B. & MADLO, Z. (1956) [Dithiocarbamin carbon acids as
degradation products of carbon disulfide.] Arch. Gewerbepath.
Gewerbehyg., 14: 511-521 (in German).
SOUCEK, B. & PAVELKOVA, E. (1953) [Absorption, metabolism, and effects
of carbon disulfide in the organism. IV. Long-term experiments.]
Prac. Lek., 5: 189 (in Czech).
SOUCEK, B., MADLO, Z., & HOLEYSOVSKA, H. (1957) [The influence of
carbon disulfide on proteolytic enzymes.] Arch. Gewerbepath.
Gewerbyhyg., 15: 531-538 (in German).
STRITTMATTER, C. F., PETERS, T., & MCKEE, R. W. (1950) Metabolism of
labelled carbon disulphide in guineapigs and mice. Arch. ind.
Hyg., 1: 54-64.
STYBLOCA, V. (1977) [Electroencephalography in the diagnosis of early
brain damage by carbon disulfide.] Int. Arch. occup. environ.
Health. 38: 263-282 (in German).
SUGIMOTO, K., GOTO, S., & HOTTA, R. (1976) An epidemiological study on
retinopathy due to carbon disulfide. Int. Arch. occup. environ.
Health, 37: 1-8.
SUGIMOTO, K., GOTO, S., TANIGUCHI, H., BABA, T., RAITTA, C., TOLONEN,
M., & HERNBERG, S. (1977) Ocular fundus photography of workers
exposed to carbon disulfide-A comparative epidemiological study
between Japan and Finland. Int. Arch. occup. environ.
Health, 39: 97-101.
SULKOWSKI, W. & LATKOWSKI, B. (1969) [Audiometric and
electronystagmographic examinations of workers in plants
producing synthetic fibers, who are suffering from chronic carbon
disulfide poisoning.] Pol. hyg. Lek., 24: 1563-1565 (in
Polish).
SZENDZIKOWSKI, S., STETKIEWICZ, J., WRONSKA-NOFER, T., & ZDRAJKOWSKA,
I. (1973) Structural aspects of experimental carbon disulfide
neuropathy. I. Development of neurohistological changes in
chronically intoxicated rats. Int. Arch. Arbeitsmed., 31:
135-149.
TABACOVA, S., HINKOVA, L., BALABAEVA, L., & KURCHATOVA, G. (1977).
Effects of carbon disulphide on the pre- and postnatal
development of rats. In: Tolonen, M., ed. Abstracts of the IV
International Symposium on Occupational Health in the Production
of Artificial Fibres, Helsinki and Valkeakoski, Finland, 6-10
June, 1977, p. 26.
TACCOLA, A., MAUGERI, U., GRAOVAC-LEPOSAVIC, LJ., CAVALLERI, A., &
DJURIC, D. (1971) [Vascular changes in human subjects exposed to
carbon disulfide.] Med. Lavoro, 62: 404-411 (in Italian).
TACCOLA, A., CAVALLERI, A., FRANCO, G., & MALAMANI, T. (1977) An
evaluation of the ratio of P-wave duration to P-R segment in ECG
of CS2 exposed workers. Egypt. J. occup. Med., 5: 309-312.
TAHARA, Y. (1961) [Hygienic evaluation of work environment of rayon
factories by carbon disulfide analysis of the expired air of
workers.] J. Sci. Labour (Tokyo), 37: 583-597 (in Japanese).
TARKOWSKI, S. (1973) [Glucose and free amino acid metabolism in the
brain of rats chronically poisoned with carbon disulfide.] Med.
Pr., 24: 571-575 (in Polish).
TARKOWSKI, S. (1974) Activity of glutamate decarboxylase in the brain
of rats exposed to carbon disulfide. Int. Arch. Arbeitsmed.,
33: 79-82.
TARKOWSKI, S. & CREMER, J. E. (1972) Metabolism of glucose and free
amino acids in brain, studied with 14C labelled glucose and
butyrate in rats intoxicated with carbon disulphide.
J. Neurochem., 19: 2631-2640.
TARKOWSKI, S. & SOBCZAK, H. (1971) Oxidation and phosphorylation
processes in brain mitochondria of rats exposed to carbon
disulphide. J. Neurochem., 18: 177-182.
TARKOWSKI, S. & WRONSKA-NOFER, T. (1966) [Effect of carbon disulphide
on the cytochrome oxidase in rats.] Med. Pr., 17: 375-379 (in
Polish).
TAZUKA, H. (1955) [A study on CS2 poisoning. Report II: On the
absorption of CS2.] J. Sci. Labour (Jpn), 31: 208-209 (in
Japanese).
TEISINGER, J. (1934) Mils central hemiparesis with extrapyramidal
signs in chronic poisoning. Cas. Lek. Ces., 73: 188-189.
TEISINGER, J. (1954) [Metabolism and detoxication of some industrial
poisons.] Praha, S.Z.N., p. 106 (in Czech).
TEISINGER, J. (1971) [Chronic carbon disulphide poisoning mechanism.]
Pracov. Ldk., 23: 306-308 (in Czech).
TEISINGER, J. & SOUCEK, B. (1949) Absorption and elimination of carbon
disulfide in man. J. ind. Hyg., 31: 67-73.
TEISINGER, J. & SOUCEK, B. (1952) [The importance of metabolism of
some toxic vapours considering their transmission and excretion
in man.] Cas. Ldk. Ces., 91: 1372-1375 (in Czech).
THIELE, H. & WOLF, I. (1976) [Results of complex clinical serial tests
on CS2-exposed workers.[ Z. ges. Hyg. DDR, 22: 603-608 (in
German).
TILLER, J. R., SCHILLING, R. S. F., & MORRIS, J. N. (1968)
Occupational toxic factor in mortality from coronary hear
disease. Br. med. J., 4: 407-411.
TINTERA, J., GRAOVAC-LEPOSAVIC, LJ., & MILIC, S. (1972) Excretion of
some tryptophan metabolites in man exposed to carbon disulphide.
med. Lavoro, 63: 126-133.
TOLONEN, M. (1974) Chronic subclinical carbon disulphide poisoning.
Work Environ. Health, 11: 154-161.
TOLONEN, M., HERNBERG, S., NURMINEN, M., & TIITOLA, K. (1975) A
follow-up study of coronary heart disease in viscose rayon
workers exposed to carbon disulphide. Br. J. ind. Med., 32:
1-10.
TOLONEN, M., HERNBERG, S., NORDMAN, C-H., GOTO, S., SUGIMOTO, K., &
BABA, T. (1976) Angina pectoris, electrocardiographic findings
and blood pressure in Finnish and Japanese workers exposed to
carbon disulfide. Int. Arch. occup. environ. Health, 37:
249-264.
TOLONEN, M., HANNINEN, H., & NURMINEN, M. (1978) Phychological tests
specific to individual carbon disulfide exposure. Scand. J.
Psychol., 19(3): 241-245.
TOYAMA, T. & HARASHIMA, S. (1962) {Industrial hygienic evaluation of
exposure to CS2 through analysis of biological materials.] Jpn.
J. ind. Health., 4: 81-85 (in Japanese).
TOYAMA, T. & KUSANO, H. (1953) [An experimental study on absorption
and excretion of CS2.] Jpn. J. Hyg., 8: 70 (in Japanese).
TOYAMA, T. & SAKURAI, H. (1967) Ten-year changes in exposure level and
toxicological manifestations in carbon disulphide workers. In:
Brieger, H. & Teisinger, J., ed. Toxicology of carbon
disulphide, Amsterdam, Excerpta Medica Foundation, pp. 197-204.
TRUHAUT, R. R., BOUDENE, C., PHU-LICH, N., & BAQUET, A. (1972)
Memoires nouvelles recherches sur la mesure en continu de
l'exposition individuelle au sulfure de carbone dans l'industrie
a l'aide d'un appareil portatif. Arch. Mal. prof., 33: 341-346.
TURCEK, M. & MOLCAN, J. (1964) [Carbon disulphide psychoses.] Prac.
Lek., 16: 257-261 (in Czech).
UEHLINGER, E. (1952) [On carbon disulfide glomerulosclerosis.]
Schweiz. Z. Allg. Pathol., 15: 217-224 (in German).
VASAK, V. (1963) [Assessment of exposure of workers to carbon
disulphide vapours.] Prac. Lek., 15: 143-145 (in Czech).
VASAK, V. & KOPECKY, J. (1967) On the role of pyridoxamine in the
mechanism of the toxic action of carbon disulfide. In: Brieger,
H. & Teisinger, J., ed. Toxicology of carbon disulphide,
Amsterdam, Excerpta Medica Foundation, pp. 35-41.
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1963) Assessment of exposure
of workers to carbon disulphide vapours. II. Application of the
iodine-azide reaction for the detection and estimation of carbon
disulphide metabolites in urine. Prac. Lék., 15: 145-149 (in
Czech).
VASAK, V., VANECEK, M., & KIMMELOVA, B. (1969) The assessment of the
exposure of workers to carbon disulphide by the iodine-azide test
of Vasak, Vanecek and Kimmelova. (1963). In: Brieger, H. &
Teisinger, J., ed. Toxicology of carbon disulphide, Amsterdam,
Excerpta Medica Foundation, pp. 56-59.
VASILJEVA, I. A. (1973) [Effect of low concentrations of carbon
disulfide and hydrogen sulfide on the menstrual function in women
and on the estrous cycle in an experiment.] Gigi. Sanit., 38:
24-27 (in Russian).
VERTIN, A. (1973) In: Gas analysis instrumentation, London,
Macmillan Press Ltd.
VERTIN, P. G. (1966) [Dynamics of the elastase and the cholesterol and
phospholipid ratio with exposure to CS2.] Arch. Gewerbepath.
Gewerbyhyg., 22: 167-178 (in German).
VERTIN, P. G. (1977) An examination for cardiac/vascular diseases in
the Dutch viscose rayon industry. In: Tolonen, M., ed. Abstracts
of the IV International Symposium on Occupational Health in the
Production of Artificial Fibres, Helsinki and Valkeakoski,
Finland, 6-10 June, 1977, p. 7.
VESCE, C. A., COPPA, S., & FIMIANI, R. (1953) [Variations in urinary
17-ketosteroids in chronic poisoning from solvents.] Folia Med.
(Napoli), 36: 444-460 (in Italian).
VIDAAKOVIC, A. & ANDJELKOVSKI, A. (1965) [Haematological changes in
workers exposed to carbon disulfide.] In: Zbornik radova o
toksikologiji CS2, Loznica, 1965, p. 77-81 (in Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & ANDJELKOVSKI, A. (1965a) [A study of the
effects of carbon disulfide in the liver.] In: Zbornik radova o
toksikologiji ugljen disulfida, Loznica, 1965, pp. 71-75 (in
Serbo-Croat).
VIDAKOVIC, A., VISNJIC, V., & DJORDJEVIC, V. (1956b) [Some changes in
the blood of rats in sub-acute carbon disulfide-poisoning.] In:
Zbornik radova o toksikologiji CS2 Loznica, 1965, pp. 83-87
(in Serbo-Croat).
VIGLIANI, E. C. (1946) [Chronic carbon disulfide poisoning. A report
on 100 cases.] Med. Lavoro, 37: 165-193 (in Italian).
VIGLIANI, E. C. (1954) Carbon disulphide poisoning in viscose rayon
factories. Br. J. ind. Med., 11: 235-244.
VIGLIANI, E. C. (1961) [Intoxication with carbon disulfide.] In:
Baader, E. W., ed. Handbuch der Gesamten Arbeitsmedizin, Urban
& Schwarzenberg, pp. 313-325 (in German).
VIGLIANI, E. C. & PERNIS, B. (1955) [Clinical and experimental
investigations on atherosclerosis caused by carbon disulfide.]
Arch. Gewerbepath. Gewerbehyg., 14: 190-202 (in German).
VIGLIANI, E., VISTINI, F., & MASPES, P. E. (1944) First observations
on myopathy from carbon disulfide. Med. Contemp., 10: 85-96.
VINOGRADOV, P. B. (1966) [New experimental findings for substantiating
the maximum permissible concentrations of carbon disulfide in
reservoir water.] Gig. i Sanit., 31: 13-17 (in Russian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., VISNJIC, V., MAUGERI, U.,
& REZMAN, I. (1966a) [Hematological findings in young workers
exposed to CS2. I. Fibrinolysis.] Med. Lavoro, 57: 667-676 (in
Italian).
VISCONTI, E., REZMAN, I., MAUGERI, U., VIDAKOVIC, A., & CAVALLERI, A.
(1966b) Hematochemical findings in young workers exposed to
carbon disulfide. II. The behavior of lipoproteins and the
"clearing factor". Med. Lavoro, 57: 677-683.
VISCONTI, E., VISNJIC, V., CAVALLERI, A., VIDAKOVIC, A., MAUGERI, U.,
& REZMAN, I. (1966c) [Haematological findings in young workers
exposed to carbon disulfide. III. Antiheparinic activity.] Med.
Lavoro, 57: 745-750 (in Italian).
VISCONTI, E., VIDAKOVIC, A., CAVALLERI, A., REZMAN, I., MAUGERI, U., &
VISNJIC, V. (1967). Fibrinolytic activity in young workers
exposed to carbon disulphide. In: Brieger, H. & Teisinger, J.,
ed. Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 128-132.
WAKATSUKI, T. & HIGASHIKAWA, n. (1959) Experimental studies on carbon
disulfide and hydrogen sulfide poisoning, histological changes in
hemopoietic organs and other internal organs. Shikoku Igaku
Zasshi, 14: 549-554.
WEAST, R. C. (1974) Handbook of Chemistry and Physics, 55th ed.,
Cleveland, CRC Press, pp. C-234-235; D-85; D-163.
WEIGMAN, H. J. (1973). [Measuring devices for automatic surveillance
of air pollution in work places.] Z. ges. Hyg., 19: 1-9 (in
German).
WILEY, F. H., HUEPER, W. C., & VON OETTINGEN, W. F. (1936) On the
toxic effects of low concentrations of carbon disulfide. J. ind.
Hyg. Toxicol., 18: 733-740.
WRONSKA-NOFER, T. (1969) The incorporation of 14C-acetate into
cholesterol in rats exposed to carbon disulphide. Biochem.
Pharmacol, 18: 925-926.
WRONSKA-NOFER, T. (1970) The influence of nicotinic acid upon the
disturbances in lipid metabolism caused by carbon disulphide in
rats. Int. Arch. Arbeitsmed., 27: 221-227.
WRONSKA-NOFER, T. (1976) Aortic phospholipids synthesis in
experimental carbon disulphide intoxication in rats. Int. Arch.
occup. environ. Health, 36: 229-234.
WRONSKA-NOFER, W., NOFER, J., & TARKOWSKI, S. (1965) [Impaired niacin
metabolite excretion in animals poisoned with carbon disulfide.]
Med. Pt., 16: 77-81 (in Polish).
WRONSKA-NOFER, T., TARKOWSKI, S., GORNY, R., SOKAL, J. A., & SZYC, M.
(1970) Accelerated turnover rate of nicotinamide adenine
dinucleotides in the liver of rats intoxicated with carbon
disulfide. Biochem. Pharmacol, 21; 2945-2950.
WRONSKA-NOFER, T., STETKIEWICZ, J., & SZENDZIKOWSKI, S. (1973)
Structural alterations and content of nicotinamine-adenine
nucleotides in skeletal muscle of rat in chronic experimental
carbon disulfi de intoxication. Int. Arch. occup. Health., 31:
125-134.
YAMAGATA, Y., YUDA, A., SUZUKI, K., NEMOTO, T., TAKAHASHI, M.,
TUCHIDA, H., SAITO, K., & KUSUNOKI, N. (1966) Carbon disulfide
nephroselerosis with special reference to the similarity to
diabetic glomerulosclerosis. J. Jpn. Diabetic. Soc., 9:
208-217.
YAROSLAVSKY, B. K. (1969) Toxic action of carbon disulfide on
reproduction and potentiation of this effect by tryptophan.
Bull. exp. Biol. Med., 68: 88-91.
YOSHIDA, K. (1955) [Fate of CS2 in the body-formation of thioketone
bonds.] J. Sci. Labour (Tokyo), 31: 209 (in Japanese).
ZEGLIO, P. (1942) Changes in gastric function due to chronic exposure
of workers in rayon industry. Med. Lavoro., 33: 217-228.
ZENK, H. (1967) The rhino-oto-vestibular symptomatoiogy of carbon
disulphide intoxication. In: Brieger, H. & Teisinger, J., ed.
Toxicology of carbon disulphide, Amsterdam, Excerpta Medica
Foundation, pp. 190-191.
ZENK, H. (1970) [Carbon disulfide influences in the rhino-otological
functions of workers in the plastic fibers industry.] Int. Arch.
Arbeitsmed., 27: 210-220 (in German).
Annex I
PRODUCTION OF VISCOSE AND ITS END-PRODUCTS
The technological process of viscose rayon production, which is
schematically presented in Annex I Fig. 1, can briefly be described in
the following way:
(a) Cellulose pulp arrives at the factory in the form of sheets
which are placed in a hydraulic press or pulverized and steeped in a
solution of caustic soda to form alkali cellulose;
(b) the alkali cellulose is shredded to form crumbs to
facilitate aging and xanthation;
(c) after storage in aging tanks until the right level of
polymerization has been reached, the crumbs are placed in mechanical
churns;
(d) in a separate operation, sulfur is melted and poured onto
glowing charcoal where it reacts to give carbon disulfide. The liquid
carbon disulfide is stored in special tanks that are protected against
flames, static electricity, and heat;
(e) the liquid carbon disulfide, which is gradually metered
into the mechanical churns, reacts with the alkali cellulose to form
orange crumbs consisting of colourless cellulose xanthate and orange
sodium trithiocarbonate (Na2CS3);
(f) these crumbs are dissolved in caustic soda solution to give
a viscose solution;
(g) the viscose is stored in aging tanks, filtered, de-aerated,
and then pumped into the spinning tanks where it is forced through
spinnerets that are submerged in a solution of sulfuric acid (H2SO4)
and sodium and zinc sulfates (Na2SO4-ZnSO4). Filaments are formed
by coagulation of the viscose when partial decomposition of the
cellulose xanthate and the trithiocarbonate takes place, according to
the reactions:
(1) ROCS + H2SO4-ROH + NaHSO4 + CS2
SNa
(2) Na2CS3 + H2SO4 - H2S + CS2 + Na2SO4
During this process, considerable quantities of carbon disulfide
and hydrogen sulfide vapours are liberated representing a danger to
the exposed workers. It is important that reaction (2) should proceed
much more quickly than reaction (1) during spinning, so that
practically all the hydrogen sulfide is already formed in the spinning
bath or immediately over it, and the part of the carbon disulfide that
is supplied by the xanthate is mainly freed outside the spinning bath
in the acid yarn and/or in further treatments. The quantities of
carbon disulfide and hydrogen sulfide vapours that are liberated
depend on the process and the end-use of the rayon product.
The following products may be produced from viscose: rayon
filament textile yarn used in the textile industry in linings and
furniture fabrics; rayon filament tire yarn used as reinforcing
material in the carcass and belt of tires; rayon staple fibre
combined in textile fabrics with natural or synthetic fibres, such as
cotton or polyester; and Cellophane film, used for packaging.
On leaving the spinning bath, textile rayon yarn may be:
(a) wound up on acid spools on the spinning machine above the
spinning bath;
(b) formed into a cylindrical acid cake in a centrifugal pot
also incorporated in the spinning machine; or
(c) continuously transported through the regeneration, washing,
and drying treatments and then wound up as the finished product.
During spinning, the spinning machines are more or less
hermetically sealed. However, during doffing or when filament breaks
occur, the worker must open the "windows" or covers. At such times,
hydrogen sulfide and carbon disulfide vapours escape into the air. All
hydrogen sulfide is formed in the spinning bath or immediately over
it. Most of the carbon disulfide will be absorbed in the acid spool or
cake: and either be retained for a long time or be immediately freed
in a hot acid bath. Consequently, hydrogen sulfide will mainly be
found in the atmosphere of the machine and the acid baths, whereas
carbon disulfide also occurs in the departments where the acid spools
and cakes undergo further treatment (storage room, washing and drying
departments). In spool spinning, the worker has to lean over the
spinning bath to perform the doting and spinning-in operations. In
cake spinning, doffing takes place either at the front of the machine,
under conditions that are roughly the same as for spool spinning, or
at the rear of the machine, where the worker is not forced to breathe
in the machine atmosphere.
 Approximately 50% of the quantity of carbon disulfide that was
originally added is present in the spool and is recovered by steaming.
The cake contains less carbon disulfide because the centrifugal,
motion of the pot removes a certain amount.
Tire yarn is mostly spun in glass tubes, where the viscose is
in direct contact with the spinning bath. The bath acid drains to the
sealed spinning tanks via return pipes and the yarn is run through a
hot acid bath, where the xanthate is largely decomposed. This second
hot acid bath may either be incorporated in the spinning machine or
installed outside it. In both cases the concentration of carbon
disulfide over this bath is high and, when a yarn breaks, the worker
may be exposed to the vapours.
After leaving the hot acid bath, the yarn is either (a) wound
on large acid spools; (b) made into cakes in centrifugal pots or
(c) run through washing and drying sections.
Staple fibre, like textile filament yarn, is mostly spun in an
open bath. However, hydrogen sulfide and carbon disulfide
concentrations are much higher, because of the large amounts of
viscose used per unit time and per spinning position. The thick tow is
treated in a separate sealed hot-acid bath (as in tire yarn spinning)
for further regeneration and large amounts of carbon disulfide are
freed in the process. After this stage, the cable is cut and more
carbon disulfide is emitted. The staple fibre is then washed and dried
continuously and carbon disulfide emissions are much lower.
Cellophane casting takes place on a continuous machine. The
sheet is cast in an open casting bath, like the spinning bath in
textile filament spinning. The Cellophane sheet then passes through a
number of baths in which decomposition, washing, bleaching, and drying
take place. In Cellophane casting, large quantities of viscose are
pressed into relatively small acid and second baths. As a result,
hydrogen sulfide and carbon disulfide emissions are very high over a
relatively small surface area.
The following data and Annex 1 Table 1 give some insight into the
emissions of hydrogen sulfide and carbon disulfide in the different
processes:
For every kg of yarn, staple fibre, or Cellophane, some 10-12 kg
of viscose is fed into the spinning or casting bath.
1 kg of viscose will give approximately: 20-30 g CS2
4-6 g H2S
(i.e., about 5 times more CS2 than H2S)
Viscose inputs in the spinning or casting machine per hour per
position are:
textile yarn 0.6-1.0 kg
tire yarn 10-12 kg
staple fibre 70/100 kg
Cellophane 1800-2000 kg
Total amounts of vapours and gases (in grams) that can be liberated
per hour per position are:
CS2 H2S
textile yarn 20-25 3-5
tire yarn 225-250 40-50
staple fibre 1000-1500 200-300
Cellophane 38 000-42 000 9000-10 000
Annex 1. Table 1. Percentage emissions of carbon disulfide and hydrogen
sulfide during the formation of viscose products.
Acid bath Spinning machine In the spool, cake,
tanks over first and tow, or Cellophane
second baths after the baths
textile yarn 35 55 10 H2S
25 65 CS2
tire yarn 15 5-55 15 H2S
25 CS2
staple fibre 45 40 15 H2S
8 12-45 35 CS2
Cellophane 15 70 15 H2S
25 55 20 CS2
Annex II
MAXIMUM PERMISSIBLE CONCENTRATIONS FOR CARBON DISULFIDE IN
DIFFERENT COUNTRIES
Chronic manifest poisoning can be prevented in the viscose rayon
industry by maintaining the carbon disulfide concentrations below
60 mg/m3 (20 ppm). However, recent studies have shown that
gynaecological cardiovascular, neurological, and neurophysiological
effects can still be detected under such conditions (sections 6.3.9,
6.3.10, 6.3.11, 7.3). It appears that a time-weighted average (TWA) of
30 mg/m3 (10 ppm) is the lowest level at which most of these effects
can be found. However, it has been reported in some studies from the
USSR that gynaecological, cardiovascular, and neurological disorders
may occur at even lower levels. It seems possible that most toxic
effects could be prevented by keeping carbon disulfide levels,
expressed as background TWAs, below 30 mg/m3 (10 ppm). The levels for
personal exposure may prove to be different but too few data are
available, at present, to make any definite statements.
If the effect is severe, as in the case of coronary deaths, it is
prudent to apply a safety margin for that effect. The need for such a
safety margin must be considered separately in each, case. Both, the
new NIOSH recommendation (NIOSH, 1977) and the USSR maximum allowable
concentration (MAC) value include safety margins. Most other countries
have not yet considered it necessary or practically feasible to adapt
safety margins. Table 1 in this annex presents permissible exposure
levels for a number of countries.
Based on results of studies on man and animals, the USSR has
adopted an MAC value of 1 mg/m3. Measurements of carbon disulfide
concentrations were made during the last 5 years of exposure on
workers exposed to 10 mg/m3 for 10-15 years. No significant changes
in the work environment had reportedly occurred during the entire
exposure period. The workers showed neurological, cardiovascular and
gynaecological disorders. (Petrov, 1969; Vasiljeva, 1973; Martynova et
al., 1976). Animals exposed to 100 and 10 mg/m3, respectively, showed
several adverse effects while only those exposed to 1 mg/m3 were free
from symptoms.
The Federal Republic of Germany has adopted a level of 30 mg/m3
"because at 60 mg/m3 level the liver oxygenesis was inhibited and
between 30-60 mg/m3 of longstanding exposure, coronary heart
diseases, hypertension, psychological and neurophysiological
disturbances can occur" (Freundt, 1975).
In the USA, NIOSH (1977) has recommended a standard of 3 mg/m3
(1 ppm) as a 10-h TWA concentration during a 40-h working week. A
level of 30 mg/m3 (10 ppm) for a 15-minute sampling period is
recommended to avoid acute toxicity. NIOSH based its choice of levels
on the probability of excess coronary deaths, and applied a safety
factor of 10 to the lowest concentration thought to be associated with
such cardiovascular disorders.
It should be stressed that the background concentration does not
reflect personal exposure, which may be considerably higher.
Therefore, the use of a personal sampler is recommended for the
assessment of actual uptake of carbon disulfide by individual workers,
accompanied by the use of the iodine-azide test. As already suggested,
it may well be that such experience will create a need for expressing
maximum permissible concentrations in terms of personal exposure
instead of background exposure.
Annex II. Table 1 Maximum permissible concentrations in
various countries. If not otherwise stated,
the figures represent TWAs.
Country ppm mg/3
Czechoslovakia (1963) 30
Egypt (1974) 30
Finland (1972) 30
German Democratic Republic (1971) 50
Germany, Federal Republic of (1975) 10 30
Hungary 20
Japan (1974) 10 30
Poland (1976) 25
Sweden (1975) 10 30
Switzerland (1971) 10 30
United Kingdom 10 30
USA (1973) 20 60
100 (MAC) 300 (MAC)
USSR (1976) 1 (MAC)
Yugoslavia (1971) 15 50 (MAC)
Approximately 50% of the quantity of carbon disulfide that was
originally added is present in the spool and is recovered by steaming.
The cake contains less carbon disulfide because the centrifugal,
motion of the pot removes a certain amount.
Tire yarn is mostly spun in glass tubes, where the viscose is
in direct contact with the spinning bath. The bath acid drains to the
sealed spinning tanks via return pipes and the yarn is run through a
hot acid bath, where the xanthate is largely decomposed. This second
hot acid bath may either be incorporated in the spinning machine or
installed outside it. In both cases the concentration of carbon
disulfide over this bath is high and, when a yarn breaks, the worker
may be exposed to the vapours.
After leaving the hot acid bath, the yarn is either (a) wound
on large acid spools; (b) made into cakes in centrifugal pots or
(c) run through washing and drying sections.
Staple fibre, like textile filament yarn, is mostly spun in an
open bath. However, hydrogen sulfide and carbon disulfide
concentrations are much higher, because of the large amounts of
viscose used per unit time and per spinning position. The thick tow is
treated in a separate sealed hot-acid bath (as in tire yarn spinning)
for further regeneration and large amounts of carbon disulfide are
freed in the process. After this stage, the cable is cut and more
carbon disulfide is emitted. The staple fibre is then washed and dried
continuously and carbon disulfide emissions are much lower.
Cellophane casting takes place on a continuous machine. The
sheet is cast in an open casting bath, like the spinning bath in
textile filament spinning. The Cellophane sheet then passes through a
number of baths in which decomposition, washing, bleaching, and drying
take place. In Cellophane casting, large quantities of viscose are
pressed into relatively small acid and second baths. As a result,
hydrogen sulfide and carbon disulfide emissions are very high over a
relatively small surface area.
The following data and Annex 1 Table 1 give some insight into the
emissions of hydrogen sulfide and carbon disulfide in the different
processes:
For every kg of yarn, staple fibre, or Cellophane, some 10-12 kg
of viscose is fed into the spinning or casting bath.
1 kg of viscose will give approximately: 20-30 g CS2
4-6 g H2S
(i.e., about 5 times more CS2 than H2S)
Viscose inputs in the spinning or casting machine per hour per
position are:
textile yarn 0.6-1.0 kg
tire yarn 10-12 kg
staple fibre 70/100 kg
Cellophane 1800-2000 kg
Total amounts of vapours and gases (in grams) that can be liberated
per hour per position are:
CS2 H2S
textile yarn 20-25 3-5
tire yarn 225-250 40-50
staple fibre 1000-1500 200-300
Cellophane 38 000-42 000 9000-10 000
Annex 1. Table 1. Percentage emissions of carbon disulfide and hydrogen
sulfide during the formation of viscose products.
Acid bath Spinning machine In the spool, cake,
tanks over first and tow, or Cellophane
second baths after the baths
textile yarn 35 55 10 H2S
25 65 CS2
tire yarn 15 5-55 15 H2S
25 CS2
staple fibre 45 40 15 H2S
8 12-45 35 CS2
Cellophane 15 70 15 H2S
25 55 20 CS2
Annex II
MAXIMUM PERMISSIBLE CONCENTRATIONS FOR CARBON DISULFIDE IN
DIFFERENT COUNTRIES
Chronic manifest poisoning can be prevented in the viscose rayon
industry by maintaining the carbon disulfide concentrations below
60 mg/m3 (20 ppm). However, recent studies have shown that
gynaecological cardiovascular, neurological, and neurophysiological
effects can still be detected under such conditions (sections 6.3.9,
6.3.10, 6.3.11, 7.3). It appears that a time-weighted average (TWA) of
30 mg/m3 (10 ppm) is the lowest level at which most of these effects
can be found. However, it has been reported in some studies from the
USSR that gynaecological, cardiovascular, and neurological disorders
may occur at even lower levels. It seems possible that most toxic
effects could be prevented by keeping carbon disulfide levels,
expressed as background TWAs, below 30 mg/m3 (10 ppm). The levels for
personal exposure may prove to be different but too few data are
available, at present, to make any definite statements.
If the effect is severe, as in the case of coronary deaths, it is
prudent to apply a safety margin for that effect. The need for such a
safety margin must be considered separately in each, case. Both, the
new NIOSH recommendation (NIOSH, 1977) and the USSR maximum allowable
concentration (MAC) value include safety margins. Most other countries
have not yet considered it necessary or practically feasible to adapt
safety margins. Table 1 in this annex presents permissible exposure
levels for a number of countries.
Based on results of studies on man and animals, the USSR has
adopted an MAC value of 1 mg/m3. Measurements of carbon disulfide
concentrations were made during the last 5 years of exposure on
workers exposed to 10 mg/m3 for 10-15 years. No significant changes
in the work environment had reportedly occurred during the entire
exposure period. The workers showed neurological, cardiovascular and
gynaecological disorders. (Petrov, 1969; Vasiljeva, 1973; Martynova et
al., 1976). Animals exposed to 100 and 10 mg/m3, respectively, showed
several adverse effects while only those exposed to 1 mg/m3 were free
from symptoms.
The Federal Republic of Germany has adopted a level of 30 mg/m3
"because at 60 mg/m3 level the liver oxygenesis was inhibited and
between 30-60 mg/m3 of longstanding exposure, coronary heart
diseases, hypertension, psychological and neurophysiological
disturbances can occur" (Freundt, 1975).
In the USA, NIOSH (1977) has recommended a standard of 3 mg/m3
(1 ppm) as a 10-h TWA concentration during a 40-h working week. A
level of 30 mg/m3 (10 ppm) for a 15-minute sampling period is
recommended to avoid acute toxicity. NIOSH based its choice of levels
on the probability of excess coronary deaths, and applied a safety
factor of 10 to the lowest concentration thought to be associated with
such cardiovascular disorders.
It should be stressed that the background concentration does not
reflect personal exposure, which may be considerably higher.
Therefore, the use of a personal sampler is recommended for the
assessment of actual uptake of carbon disulfide by individual workers,
accompanied by the use of the iodine-azide test. As already suggested,
it may well be that such experience will create a need for expressing
maximum permissible concentrations in terms of personal exposure
instead of background exposure.
Annex II. Table 1 Maximum permissible concentrations in
various countries. If not otherwise stated,
the figures represent TWAs.
Country ppm mg/3
Czechoslovakia (1963) 30
Egypt (1974) 30
Finland (1972) 30
German Democratic Republic (1971) 50
Germany, Federal Republic of (1975) 10 30
Hungary 20
Japan (1974) 10 30
Poland (1976) 25
Sweden (1975) 10 30
Switzerland (1971) 10 30
United Kingdom 10 30
USA (1973) 20 60
100 (MAC) 300 (MAC)
USSR (1976) 1 (MAC)
Yugoslavia (1971) 15 50 (MAC)
