
Concise International Chemical Assessment Document 10
2-Butoxyethanol
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
First draft prepared by
Ms J. Wess and Dr H. Ahlers, National Institute for Occupational
Safety and Health, Cincinnati, USA, and
Dr S. Dobson, Institute of Terrestrial Ecology, Huntingdon, United
Kingdom
Published under the joint sponsorship of the United Nations
Environment Programme, the International Labour Organisation, and the
World Health Organization, and produced within the framework of the
Inter-Organization Programme for the Sound Management of Chemicals.
World Health Organization Geneva, 1998
The International Programme on Chemical Safety (IPCS),
established in 1980, is a joint venture of the United Nations
Environment Programme (UNEP), the International Labour Organisation
(ILO), and the World Health Organization (WHO). The overall
objectives of the IPCS are to establish the scientific basis for
assessment of the risk to human health and the environment from
exposure to chemicals, through international peer review processes, as
a prerequisite for the promotion of chemical safety, and to provide
technical assistance in strengthening national capacities for the
sound management of chemicals.
The Inter-Organization Programme for the Sound Management of
Chemicals (IOMC) was established in 1995 by UNEP, ILO, the Food and
Agriculture Organization of the United Nations, WHO, the United
Nations Industrial Development Organization, the United Nations
Institute for Training and Research, and the Organisation for Economic
Co-operation and Development (Participating Organizations), following
recommendations made by the 1992 UN Conference on Environment and
Development to strengthen cooperation and increase coordination in the
field of chemical safety. The purpose of the IOMC is to promote
coordination of the policies and activities pursued by the
Participating Organizations, jointly or separately, to achieve the
sound management of chemicals in relation to human health and the
environment.
WHO Library Cataloguing in Publication Data
2-Butoxyethanol.
(Concise international chemical assessment document ; 10)
1. Ethylene glycols - adverse effects
2. Ethylene glycols - toxicity
3. Environmental exposure
4. Dose-response relationship, Drug
I. International Programme on Chemical Safety
II.Series
ISBN 92 4 153010 3 (NLM Classification: QD 341.E7)
ISSN 1020-6167
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1998
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion whatsoever
on the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city, or area
or of its authorities, or concerning the delimitation of its frontiers
or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
The Federal Ministry for the Environment, Nature Conservation and
Nuclear Safety, Germany, provided financial support for the printing
of this publication.
TABLE OF CONTENTS
FOREWORD
1. EXECUTIVE SUMMARY
2. IDENTITY AND PHYSICAL/CHEMICAL PROPERTIES
3. ANALYTICAL METHODS
4. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
5. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
6. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
6.1. Environmental levels
6.2. Human exposure
7. COMPARATIVE KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
8. EFFECTS ON LABORATORY MAMMALS AND IN VITRO TEST SYSTEMS
8.1. Single exposure
8.2. Irritation and sensitization
8.3. Short-term exposure
8.4. Long-term exposure
8.4.1. Subchronic exposure
8.4.2. Chronic exposure and carcinogenicity
8.5. Genotoxicity and related end-points
8.6. Reproductive and developmental toxicity
8.7. Immunological and neurological effects
8.8. In vitro haemolytic effects
9. EFFECTS ON HUMANS
10. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
10.1. Aquatic environment
10.2. Terrestrial environment
11. EFFECTS EVALUATION
11.1. Evaluation of health effects
11.1.1. Hazard identification and dose-response assessment
11.1.2. Criteria for setting guidance values for 2-butoxyethanol
11.1.3. Sample risk characterization
11.2. Evaluation of environmental effects
11.2.1. Aquatic environment
11.2.2. Terrestrial environment
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
13. HUMAN HEALTH PROTECTION AND EMERGENCY ACTION
13.1. Human health hazards
13.2. Advice to physicians
13.3. Health surveillance advice
13.4. Spillage
14. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
INTERNATIONAL CHEMICAL SAFETY CARD
REFERENCES
APPENDIX 1 -- SOURCE DOCUMENTS
APPENDIX 2 -- CICAD PEER REVIEW
APPENDIX 3 -- CICAD FINAL REVIEW BOARD
RÉSUMÉ D'ORIENTATION
RESUMEN DE ORIENTACION
FOREWORD
Concise International Chemical Assessment Documents (CICADs) are
the latest in a family of publications from the International
Programme on Chemical Safety (IPCS) -- a cooperative programme of the
World Health Organization (WHO), the International Labour Organisation
(ILO), and the United Nations Environment Programme (UNEP). CICADs
join the Environmental Health Criteria documents (EHCs) as
authoritative documents on the risk assessment of chemicals.
CICADs are concise documents that provide summaries of the
relevant scientific information concerning the potential effects of
chemicals upon human health and/or the environment. They are based on
selected national or regional evaluation documents or on existing
EHCs. Before acceptance for publication as CICADs by IPCS, these
documents undergo extensive peer review by internationally selected
experts to ensure their completeness, accuracy in the way in which the
original data are represented, and the validity of the conclusions
drawn.
The primary objective of CICADs is characterization of hazard and
dose-response from exposure to a chemical. CICADs are not a summary
of all available data on a particular chemical; rather, they include
only that information considered critical for characterization of the
risk posed by the chemical. The critical studies are, however,
presented in sufficient detail to support the conclusions drawn. For
additional information, the reader should consult the identified
source documents upon which the CICAD has been based.
Risks to human health and the environment will vary considerably
depending upon the type and extent of exposure. Responsible
authorities are strongly encouraged to characterize risk on the basis
of locally measured or predicted exposure scenarios. To assist the
reader, examples of exposure estimation and risk characterization are
provided in CICADs, whenever possible. These examples cannot be
considered as representing all possible exposure situations, but are
provided as guidance only. The reader is referred to EHC 1701 for
advice on the derivation of health-based guidance values.
While every effort is made to ensure that CICADs represent the
current status of knowledge, new information is being developed
constantly. Unless otherwise stated, CICADs are based on a search of
the scientific literature to the date shown in the executive summary.
In the event that a reader becomes aware of new information that would
change the conclusions drawn in a CICAD, the reader is requested to
contact IPCS to inform it of the new information.
1 International Programme on Chemical Safety (1994) Assessing
human health risks of chemicals: derivation of guidance values for
health-based exposure limits. Geneva, World Health Organization
(Environmental Health Criteria 170).
Procedures
The flow chart shows the procedures followed to produce a CICAD.
These procedures are designed to take advantage of the expertise that
exists around the world -- expertise that is required to produce the
high-quality evaluations of toxicological, exposure, and other data
that are necessary for assessing risks to human health and/or the
environment.
The first draft is based on an existing national, regional, or
international review. Authors of the first draft are usually, but not
necessarily, from the institution that developed the original review.
A standard outline has been developed to encourage consistency in
form. The first draft undergoes primary review by IPCS to ensure that
it meets the specified criteria for CICADs.
The second stage involves international peer review by scientists
known for their particular expertise and by scientists selected from
an international roster compiled by IPCS through recommendations from
IPCS national Contact Points and from IPCS Participating Institutions.
Adequate time is allowed for the selected experts to undertake a
thorough review. Authors are required to take reviewers' comments
into account and revise their draft, if necessary. The resulting
second draft is submitted to a Final Review Board together with the
reviewers' comments.
The CICAD Final Review Board has several important functions:
- to ensure that each CICAD has been subjected to an appropriate
and thorough peer review;
- to verify that the peer reviewers' comments have been addressed
appropriately;
- to provide guidance to those responsible for the preparation of
CICADs on how to resolve any remaining issues if, in the opinion
of the Board, the author has not adequately addressed all
comments of the reviewers; and
- to approve CICADs as international assessments.
Board members serve in their personal capacity, not as representatives
of any organization, government, or industry. They are selected
because of their expertise in human and environmental toxicology or
because of their experience in the regulation of chemicals. Boards
are chosen according to the range of expertise required for a meeting
and the need for balanced geographic representation.
Board members, authors, reviewers, consultants, and advisers who
participate in the preparation of a CICAD are required to declare any
real or potential conflict of interest in relation to the subjects
under discussion at any stage of the process. Representatives of
nongovernmental organizations may be invited to observe the
proceedings of the Final Review Board. Observers may participate in
Board discussions only at the invitation of the Chairperson, and they
may not participate in the final decision-making process.
 1. EXECUTIVE SUMMARY
This CICAD on 2-butoxyethanol was based upon reviews prepared by
the National Institute for Occupational Safety and Health (NIOSH,
1990) and the Agency for Toxic Substances and Disease Registry (ATSDR,
1996). Additional data were identified through an updated literature
search to May 1997, as well as during the peer review of this CICAD.
Information on the nature of the peer review and availability of the
source documents is presented in Appendix 1. Information on the peer
review of this CICAD is presented in Appendix 2. This CICAD was
approved as an international assessment at a meeting of the Final
Review Board, held in Berlin, Germany, on 26-28 November 1997.
Participants at the Final Review Board meeting are listed in Appendix
3. The International Chemical Safety Card (ICSC 0059) produced by the
International Programme on Chemical Safety (IPCS, 1993) has also been
reproduced in this document.
2-Butoxyethanol (CAS no. 111-76-2) is a high-production-volume
glycol ether. It is a colourless liquid that is miscible in water and
soluble in most organic solvents. 2-Butoxyethanol is used widely as a
solvent in surface coatings, such as spray lacquers, quick-dry
lacquers, enamels, varnishes, varnish removers, and latex paint. It
is also used in metal and household cleaners. 2-Butoxyethanol exists
in the atmosphere almost entirely as a vapour; because the chemical
has an atmospheric half-life of approximately 17 h, the risk for
transport via the atmosphere should be small. The estimated half-life
of 2-butoxyethanol in water is approximately 1-4 weeks, and the
chemical is likely readily biodegraded in aerobic soil and water. Its
potential for bioaccumulation is low. Based upon limited data,
ambient exposures in air are generally in the µg/m3 range. Indirect
exposure of the general population to 2-butoxyethanol is most likely
from inhalation and dermal absorption during the use of products
containing the chemical. Levels of airborne 2-butoxyethanol in
occupational settings are typically in the mg/m3 range.
2-Butoxyethanol is readily absorbed following inhalation, oral,
and dermal exposure. The chemical is metabolized primarily via
alcohol and aldehyde dehydrogenases, with the formation of
2-butoxyacetaldehyde and 2-butoxyacetic acid, the principal
metabolite, although other metabolic pathways have also been
identified.
2-Butoxyethanol has moderate acute toxicity and is irritating to
the eyes and skin; it is not a skin sensitizer. The principal effect
exerted by 2-butoxyethanol and its metabolite 2-butoxyacetic acid is
haematotoxicity, with the rat being the most sensitive species. The
results of in vitro studies indicate that human red blood cells are
not as sensitive as rat red blood cells to the haemolytic effects of
2-butoxyethanol and 2-butoxyacetic acid and also that red blood cells
are more sensitive to haemolysis by 2-butoxyacetic acid than to
haemolysis by 2-butoxyethanol. In rats, adverse effects on the
central nervous system, kidneys, and liver occur at higher exposure
concentrations than do haemolytic effects. In animals, adverse
effects on reproduction and development have not been observed at less
than toxic doses. Although the results of in vitro tests for
mutagenicity of 2-butoxyethanol were inconsistent, the absence of
structural alerts and the negative findings from in vivo studies are
sufficiently reassuring to allow the conclusion that 2-butoxyethanol
is not mutagenic. Based on limited data from case reports and one
laboratory study, similar acute effects -- including haemolytic
effects as well as effects on the central nervous system -- are
observed in humans and rats exposed to 2-butoxyethanol, although the
effects are observed at much higher exposure concentrations in humans
than in rats. Based upon the development of haemolytic effects in
pregnant rats exposed during gestation, a sample tolerable
concentration for humans of 13.1 mg 2-butoxyethanol/m3 has been
derived.
Based upon extremely conservative assumptions, the highest
predicted concentrations of 2-butoxyethanol in surface waters in the
immediate vicinity of effluent streams may, in some cases, exceed
predicted no-observed-effect concentrations. However, more realistic
assumptions based on the available data suggest that risk to aquatic
organisms is low. Owing to the short half-life of 2-butoxyethanol in
the atmosphere, measured or predicted concentrations of this chemical
in air are considered to have no environmental significance.
2. IDENTITY AND PHYSICAL/CHEMICAL PROPERTIES
2-Butoxyethanol (CAS no. 111-76-2; C6H14O2; ethylene glycol
monobutyl ether, monobutyl glycol ether, 2-butoxy-1-ethanol,
2- n-butoxyethanol) is a synthetic glycol ether. It is a colourless
liquid with a mild ether odour; the odour threshold is approximately
0.10 ppm (0.48 mg/m3) (Amoore & Hautala, 1983). At ambient
temperature, 2-butoxyethanol is miscible in water and soluble in most
organic solvents. 2-Butoxyethanol has a boiling point of 171°C, a
vapour pressure of 0.1 kPa at 20°C, and a log octanol/water partition
coefficient of 0.83. The conversion equation for 2-butoxyethanol is
1 ppm = 4.83 mg/m3 (at 25°C, 101.3 kPa). Additional physical and
chemical properties are presented in the International Chemical Safety
Card reproduced in this document. The structural formula for
2-butoxyethanol is CH3CH2CH2CH2-O-CH2CH2OH.
3. ANALYTICAL METHODS
Laboratory analysis for 2-butoxyethanol in environmental samples
is usually by gas chromatography (GC) in combination with flame
ionization detection (FID), electron capture detection (ECD), or mass
spectrometric (MS) detection; infrared absorption spectrophotometry is
also sometimes used. The detection limits of these analytical methods
in air range from 0.031 ppm (0.15 mg/m3) for a 48-litre sample (OSHA,
1990) to 0.01-0.02 mg for 2- to 10-litre samples (NIOSH, 1994).
Multidimensional GC-MS has been used to improve the detection limit to
5-7 µg per sample (Kennedy et al., 1990).
Biological monitoring is a useful adjunct to environmental
measurements in assessing human exposure to 2-butoxyethanol, as it
accounts for both dermal and respiratory uptake. A variety of GC
methods combined with FID, ECD, or MS detection and high-performance
liquid chromatography (HPLC) methods coupled with ultraviolet or
radiochemical detection have been developed for the analysis of
2-butoxyethanol and its metabolite 2-butoxyacetic acid in the urine
and blood of exposed workers or rats.
In general, these methods are based on either extraction or
lyophilization of the blood or urine followed by derivatization and
then analysis (Smallwood et al., 1984, 1988; Groeseneken et al., 1986,
1989; Johanson et al., 1986, 1988; Rettenmeier et al., 1993; Sakai et
al., 1993, 1994; Corley et al., 1994). The detection limits range
from 0.03 to 0.1 mg 2-butoxyacetic acid/litre. 2-Butoxyethanol and
2-butoxyacetic acid in rat and human blood can be analysed by a GC-MS
derivatization method with a detection limit range of 16-18 ng/g blood
(Bormett et al., 1995). The National Institute for Occupational
Safety and Health reviewed the available data and developed guidelines
for biological monitoring of 2-butoxyacetic acid (NIOSH, 1990).
4. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
2-Butoxyethanol does not occur naturally. It is usually produced
by reacting ethylene oxide with butyl alcohol, but it may also be made
by the direct alkylation of ethylene glycol with an agent such as
dibutyl sulfate (Rowe & Wolf, 1982). Temperature, pressure, reactant
molar ratios, and catalysts are selected to give the product mix
desired.
2-Butoxyethanol is widely used as a solvent in surface coatings,
such as spray lacquers, quick-dry lacquers, enamels, varnishes,
varnish removers, and latex paint (Leaf, 1985; Sax & Lewis, 1987). In
surface coatings, it imparts blush resistance, gloss, and good
flow-out. 2-Butoxyethanol is also used as a coupling agent in metal
and household cleaners; as an intermediate in 2-butoxyethanol acetate
production; and in herbicides, automotive brake fluids, printing inks,
spot removers, and cosmetics (Leaf, 1985; ATSDR, 1996). In 1994,
176 900 tonnes of 2-butoxyethanol were produced in the USA (US ITC,
1996). Within the European Community, the total production capacity
of 2-butoxyethanol was approximately 70 000-90 000 tonnes in the same
year (ECETOC, 1994; CEFIC, 1995).
2-Butoxyethanol may be released into air or water by facilities
that manufacture, process, or use the chemical (ATSDR, 1996; US NLM,
1997). Products containing 2-butoxyethanol may also release the
substance into the air. Solvent-based building materials such as
silicone caulk will release 2-butoxyethanol to air as they dry. There
is potential for the release of 2-butoxyethanol from hazardous waste
sites, although quantitative data have not been identified. Based
upon the detection of 2-butoxyethanol in samples of groundwater and
surface water taken near municipal landfills and hazardous waste
sites, 2-butoxyethanol may be released to water in leachates from
these sites (ATSDR, 1996). Information on the total estimated release
of 2-butoxyethanol into the environment in the USA was not identified.
In Canada, emissions to the environment between 1992 and 1994 have
been reported to range from 1.4 to 3.1 tonnes per year (Canadian
Chemical Producers' Association, 1996).
5. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
In the atmosphere, 2-butoxyethanol is expected to exist in the
vapour phase. Owing to its water solubility, wet deposition is likely
to be more important than dry deposition (ATSDR, 1996). The chemical
will not persist in the atmosphere; it has an atmospheric half-life of
approximately 17 h, based on an estimated rate constant for reaction
with hydroxyl radicals (US NLM, 1997).
The miscibility of 2-butoxyethanol in water suggests that
volatilization from water, adsorption, and bioconcentration are not
important fate processes and that the chemical should not
bioconcentrate in aquatic organisms. Based upon aerobic
biodegradation rates, the half-life of 2-butoxyethanol in water is
estimated to range from 1 to 4 weeks (Howard et al., 1991).
2-Butoxyethanol is not likely to undergo direct hydrolysis in the
aquatic environment, and it is likely readily biodegraded (ATSDR,
1996). Five-day theoretical biological oxygen demand values range
from 5% (without acclimation) to 73% (with acclimation); 10-day
theoretical biological oxygen demand values range from 57% to 74%.
The maximum theoretical biological oxygen demand value reported is 88%
for 20 days (US NLM, 1997). Biodegradation is likely to be the most
important mechanism for the removal of 2-butoxyethanol from aerobic
soil and water.
6. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
6.1 Environmental levels
Limited data are available on the concentration of
2-butoxyethanol in environmental media. Reported levels of
2-butoxyethanol in samples of ambient air taken from Nepal and Europe
and from Antarctica have ranged from 0.1 to 1.59 µg/m3 and from 1.26
to 14.85 µg/m3, respectively (Ciccioli et al., 1993, 1996).
2-Butoxyethanol was detected at a concentration of 23 µg/litre in one
of seven groundwater samples collected near the Valley of Drums,
Kentucky, USA (ATSDR, 1996). Additional monitoring data on the
concentration of 2-butoxyethanol in surface waters and information on
levels in soils or sediments have not been identified. Levels below
100 µg 2-butoxyethanol/litre have been reported in samples of
industrial wastewater effluents in the USA (ATSDR, 1996). Water
samples obtained from a highly polluted site on the Hayashida River in
Japan, where effluent entered the river from the leather industry,
contained 1310 and 5680 µg 2-butoxyethanol/litre (Yasuhara et al.,
1981).
6.2 Human exposure
Quantitative information on levels of 2-butoxyethanol in
drinking-water and foodstuffs has not been identified, although the
chemical has been detected (levels not specified) in drinking-water in
six US cities, and there is the potential for the presence of
2-butoxyethanol in foods arising from labelling or packaging
materials. Data on concentrations of 2-butoxyethanol in indoor air in
the USA are limited to one report, in which the daily arithmetic mean
concentration was 0.214 ppbv (1 µg/m3) for samples obtained from 14
non-industrial offices. 2-Butoxyethanol at a concentration of 8
µg/m3 was detected in one of six samples of indoor air collected from
14 homes in northern Italy (ATSDR, 1996).
2-Butoxyethanol is present in a variety of consumer products,
including cleaning agents and surface coatings, such as paints,
lacquers, and varnishes. The average concentration of 2-butoxyethanol
in household products marketed in the USA in 1977 was 2.8%. Levels of
2-butoxyethanol in industrial and household window-cleaning agents
have been reported to range from 1% to 30% (v/v) (ATSDR, 1996). Based
upon available data, indirect exposure of the general population to
2-butoxyethanol is most likely via inhalation and dermal absorption
during the use of products containing this chemical.
Based on information from the National Occupational Exposure
Survey (NIOSH, 1983), the number of workers potentially exposed to
2-butoxyethanol in the workplace in the USA during 1981-1983 was
estimated at about 1.7 million, although it has probably increased
since then. Data on the occurrence of airborne 2-butoxyethanol in the
workplace obtained from facilities in the USA indicate that, in
general, most mean time-weighted-average exposures are below 7 ppm
(33.8 mg/m3) (NIOSH, 1990; ATSDR, 1996). Time-weighted average
2-butoxyethanol exposures have ranged from 1.1 to 5.4 ppm (5.3-26.1
mg/m3), with an average of 3.5 ppm (16.9 mg/m3), for silk screening;
average exposures of 6.8 ppm (32.8 mg/m3) for silk screeners, 2.6 ppm
(12.6 mg/m3) for silk screen spray painters, and 1.8 ppm (8.7 mg/m3)
for printing have also been reported (NIOSH, 1990; ATSDR, 1996). In a
study of various industrial operations, geometric mean atmospheric
exposures to 2-butoxyethanol ranged from 1.5 to 17.7 mg/m3 for
printing, from 3.4 to 93.6 mg/m3 for painting, and from 0.2 to 1774
mg/m3 in a mirror manufacturing plant (Veulemans et al., 1987).
Workers employed in varnish production facilities have been reported
to have individual exposures ranging from <0.1 to 8.1 ppm
(<0.5-39.1 mg/m3) (Angerer et al., 1990; Sohnlein et al., 1993). In
a study of automobile cleaners using products containing
2-butoxyethanol, time-weighted-average personal exposures ranged from
<0.1 to 7.33 ppm (<0.5-35.4 mg/m3) (Vincent et al., 1993).
7. COMPARATIVE KINETICS AND METABOLISM IN LABORATORY ANIMALS AND
HUMANS
Results of animal and human studies (most of the available data
are from studies conducted with rats) indicate that 2-butoxyethanol is
readily absorbed following inhalation, oral, and dermal exposure and
is oxidized to 2-butoxyacetic acid (Jonsson & Steen, 1978).
2-Butoxyethanol is metabolized primarily via alcohol and aldehyde
dehydrogenases, with the formation of 2-butoxyacetaldehyde and
2-butoxyacetic acid, the principal metabolite (Ghanayem et al., 1987b;
Medinsky et al., 1990). This is the favoured metabolic pathway for
lower systemic doses of 2-butoxyethanol. Alternative pathways include
O-dealkylation to ethylene glycol and conjugation to 2-butoxyethanol
glucuronide and/or 2-butoxyethanol sulfate (Medinsky et al., 1990).
In the study conducted by Medinsky et al. (1990), higher relative
concentrations of 2-butoxyacetic acid and ethylene glycol were
obtained at lower vapour concentrations of 2-butoxyethanol; higher
2-butoxyethanol glucuronide levels were observed at the high exposures
to 2-butoxyethanol, possibly owing to saturation of the pathways
leading to the formation of 2-butoxyacetic acid and ethylene glycol.
In human but not animal studies, the amino acid conjugate of
2-butoxyethanol, N-butoxyacetylglutamine, has been identified as a
metabolite (Rettenmeier et al., 1993).
In general, the metabolism of 2-butoxyethanol to 2-butoxyacetic
acid is linearly related to exposure concentration up to levels
causing mortality. In one study, after inhalation exposure in rats,
2-butoxyethanol and 2-butoxyacetic acid were analysed in blood,
muscle, liver, and testes. The kinetic profile of 2-butoxyacetic acid
tissue concentrations was similar to that of 2-butoxyethanol tissue
concentrations. Sixty-four per cent of the inhaled dose of
2-butoxyacetic acid was eliminated in urine, and the rate of urinary
excretion of 2-butoxyacetic acid was dose-dependent (Johanson, 1994).
In humans exposed to 20 ppm (96.6 mg/m3) 2-butoxyethanol for 2 h
via inhalation, the concentration of 2-butoxyethanol in the blood
reached a plateau of 7.4 µmol/litre within 1-2 h, and the chemical
could no longer be detected in the blood 2-4 h after exposure. The
mean elimination half-time was 40 min. Less than 0.03% of the total
uptake of 2-butoxyethanol was excreted in the urine, whereas urinary
excretion as 2-butoxyacetic acid ranged from 17% to 55% (Johanson et
al., 1986). Similarly, after percutaneous uptake of 2-butoxyethanol,
the urinary excretion of 2-butoxyacetic acid peaked 3 h after exposure
and subsequently declined, with an average half-life of 3.1 h. The
accumulated excretion of 2-butoxyacetic acid ranged from 8.7 to 313
µmol, corresponding to 2.5-39% of uptake (Johanson et al., 1988).
Several physiologically based pharmacokinetic (PBPK) models of
2-butoxyethanol absorption, metabolism, disposition, and excretion
have been developed. One model examined human inhalation exposures
during rest and exercise (Johanson et al., 1986; Johanson & Boman,
1991), whereas another addressed high-to-low-dose extrapolation and
route of administration extrapolation based on animal data (Shyr et
al., 1993). In the Shyr et al. (1993) model, 2-butoxyethanol is
metabolized to 2-butoxyacetic acid and ethylene glycol. An additional
model combined aspects of the preceding models and addressed the
disposition of 2-butoxyacetic acid in rats and humans (Corley et al.,
1994). The Corley et al. (1994) PBPK model describes the uptake,
distribution, metabolism, and elimination of 2-butoxyethanol and its
metabolite 2-butoxyacetic acid. It was developed by expanding a
previous inhalation model for 2-butoxyethanol (Johanson et al., 1986)
and is composed of two separate models for 2-butoxyethanol and
2-butoxyacetic acid that are joined through metabolism in the liver.
Both the 2-butoxyethanol and 2-butoxyacetic acid models have the same
eight compartments, with an additional kidney compartment in the
2-butoxyacetic acid model. Unlike the original model of Johanson et
al. (1986), the muscle and skin compartments have been separated.
Corley et al. (1994) also incorporated protein binding and saturable
elimination of 2-butoxyacetic acid by the kidneys. Equations for
additional routes of exposure (oral, dermal, and intravenous infusion)
were also added. Physiological and biochemical parameters were
allometrically scaled rather than using standard values for a 70-kg
human. This allows simulations to be conducted for specific data
sets. A rat version of the model was also developed.
The Corley et al. (1994) model accurately predicted animal data
at dose levels that did not cause haemolysis, the principal effect
exerted by 2-butoxyethanol (see below). At dose levels causing
haemolysis, the model overpredicted the amount of 2-butoxyacetic acid
excreted in the urine. This overprediction is assumed to be caused by
toxicity in the kidneys that is secondary to haemolysis. The model
does not accommodate toxicity in the kidneys and assumes that the
kidneys will continue to function as normal, thereby leading to the
overprediction of 2-butoxyacetic acid levels in the urine. The
results of the Johanson & Boman (1991) study indicated that during
whole-body exposure to 2-butoxyethanol vapour, dermal uptake accounted
for approximately 75% of the total uptake of the chemical. The Corley
et al. (1994) model was able to accurately predict the Johanson &
Boman (1991) human whole-body exposure blood data when it was assumed
that the sampled blood did not represent systemic venous blood but
instead represented venous blood draining from the skin compartment.
This blood had not yet been diluted by the venous blood pool. Corley
et al. (1994, 1997) suggested that the blood samples collected by
Johanson & Boman (1991) were not representative of systemic blood
concentrations and that dermal uptake is approximately 21% of the
total, rather than the 75% suggested by Johanson & Boman (1991). An
additional study further addresses dermal uptake in humans from the
vapour phase but does not address direct skin contact with liquid
containing 2-butoxyethanol (Corley et al., 1997).
8. EFFECTS ON LABORATORY MAMMALS AND IN Vitro TEST SYSTEMS
8.1 Single exposure
Many acute toxicity studies of 2-butoxyethanol have led to the
establishment of LC50s or LD50s in a variety of species by
inhalation, oral, and dermal exposure. Inhalation LC50s for
2-butoxyethanol of 486 ppm (2347 mg/m3) (male rats, 4 h), 450 ppm
(2174 mg/m3) (female rats, 4 h), 700 ppm (3381 mg/m3) (mice, 7 h),
and >650 ppm (3140 mg/m3) (guinea-pigs, 1 h) have been reported.
Oral LD50s for rats (2500 mg/kg body weight), mice (1400 mg/kg body
weight), guinea-pigs (1200 mg/kg body weight), and rabbits (320 mg/kg
body weight) have also been reported. Dermal LD50s of 404-502 and
2000 mg/kg body weight have been reported for rabbits and guinea-pigs,
respectively. Effects observed in rats, mice, and guinea-pigs exposed
by inhalation to the LC50 or by ingestion to the LD50 include loss of
coordination, ataxia, sluggishness, muscular flaccidity, enlarged
kidney, blood in the bladder, haemoglobinuria, splenic lesions, and
pulmonary congestion (Werner et al., 1943a; Carpenter et al., 1956;
Dodd et al., 1983; Gingell et al., 1997). Inhalation exposures of
female rats to 62 ppm (299 mg/m3) 2-butoxyethanol for 4-h periods
resulted in increased osmotic fragility of erythrocytes (Carpenter et
al., 1956).
Ghanayem et al. (1987a) indicated that the haemolytic activity of
2-butoxyethanol in rats is age-dependent, with older rats being more
susceptible than younger animals. In their study, 2-butoxyethanol (0,
125, or 500 mg/kg body weight) was administered orally to young (4- to
5-week-old) and adult (9- to 13-week-old) male F344 rats, and
haematotoxicity was assessed from 2 to 48 h later. A decrease in red
blood cells, haemoglobin, and haematocrit was accompanied by a
significant ( p < 0.05) dose-dependent increase in free haemoglobin
plasma levels in both age groups administered 500 mg
2-butoxyethanol/kg body weight; in both groups, there was a gradual
recovery after 48 h. Although no significant haematotoxic effects
were observed in the younger rats administered 125 mg
2-butoxyethanol/kg body weight, effects in older animals administered
this dose included a significant decrease ( p < 0.05) in the
number of red blood cells, haematocrit, and haemoglobin. Free
haemoglobin plasma levels were significantly increased ( p < 0.05)
in adult rats 8 h after oral administration of 125 mg/kg body weight;
there was no effect on free haemoglobin plasma levels in young
animals. Histopathological evaluation of tissues collected 24 h after
2-butoxyethanol administration to rats of various ages revealed dose-
and age-dependent liver and kidney changes. These histopathological
changes exhibited signs of regression when examined 48 h following
exposure. Severe acute haemolytic anaemia was evidenced by a decrease
in circulating red blood cells, an increase in the concentration of
free haemoglobin in plasma, and the development of haemoglobinuria.
Using a laser-based haematology analyser, Ghanayem et al. (1987a)
indicated that the acute haemolysis in 2-butoxyethanol-exposed rats
was caused by a time- and dose-dependent decrease in the number of red
blood cells, in haemoglobin concentrations, and in haematocrit, with
little or no change in mean cell volume. In a follow-up study in
which the authors used both a laser-based haematology analyser and an
impedance-based analyser, haematology profiles from the
impedance-based haematology analyser revealed a time- and
dose-dependent increase in haematocrit and mean cell volume; the
laser-based analyser was unable to detect early increases in
haematocrit and mean cell volume in the exposed animals. Based on
these data, Ghanayem et al. (1990) concluded that 2-butoxyethanol
causes spherical swelling of red blood cells followed by haemolysis.
To investigate the induction of tolerance, Ghanayem et al. (1992)
assessed haematological parameters in naive or previously bled rats
administered a single dose of 125 or 250 mg 2-butoxyethanol/kg body
weight. The bled/recovered rats were less sensitive to
2-butoxyethanol than the naive animals. In vitro incubations with
2-butoxyacetic acid revealed that red blood cells from the
bled/recovered rats were less sensitive than those cells from naive
animals. Ghanayem et al. (1992) concluded that young red blood cells
formed during the regeneration process were less sensitive to
2-butoxyacetic acid than older red blood cells. Chronic exposure to
2-butoxyethanol would be expected to result in tolerance to
2-butoxyethanol-induced haemolytic anaemia. The mechanism is probably
related to the greater susceptibility of older cells to 2-butoxyacetic
acid; haemolysis of these cells during the initial exposure followed
by their replacement with less susceptible younger cells may account
for the development of tolerance.
Toxic effects in the kidneys have been observed in rabbits
exposed percutaneously to 2-butoxyethanol (Carpenter et al., 1956).
Necropsy of rabbits exposed for 24 h to undiluted 2-butoxyethanol
(0.48-0.64 ml/kg body weight) revealed congestion of the kidneys,
haemoglobinuria, pale livers, and engorged spleens (Carpenter et al.,
1956).
When 2-butoxyethanol (200, 260, 320, 375, or 500 mg/kg body
weight) was applied to the shaved dorsal skin of groups of female
rats, increased mean cell volume, a lowered erythrocyte count and
haemoglobin level, and haemoglobinuria were observed within 6 h of
exposure to the highest dose; no haemolytic effects were observed at
the lowest dose tested (Bartnik et al., 1987). 2-Butoxyethanol at
doses of 260, 320, and 375 mg/kg body weight produced similar effects
in at least some animals in each group; however, there was no
discernible dose-response relationship, which was attributed to the
inherent biological variation in percutaneous absorption and
haemolytic susceptibility and to the small number of animals ( n = 3)
in these dose groups.
8.2 Irritation and sensitization
2-Butoxyethanol is irritating to the eyes and skin. In rabbits,
instillation of an unspecified amount of undiluted 2-butoxyethanol
caused severe eye irritation, including conjunctival hyperaemia and
oedema (von Oettingen & Jirouche, 1931). More recent ocular tests in
rabbits revealed that 30% and 70% concentrations of 2-butoxyethanol
were moderately irritating (Kennah et al., 1989). When applied to the
skin of rabbits for 4 h, 2-butoxyethanol caused mild irritation;
extending the period of contact increased the severity of irritation
(Tyler, 1984). 2-Butoxyethanol was classified as a severe cutaneous
irritant when the Draize method was used (Zissu, 1995).
2-Butoxyethanol did not induce skin sensitization in guinea-pigs
(Unilever, 1989, as cited in ECETOC, 1994; Zissu, 1995).
8.3 Short-term exposure
In older studies, haematotoxic effects (e.g. increased osmotic
fragility, decreased haemoglobin, decreased numbers of red blood
cells) have been observed in rats (54-320 ppm; 261-1546 mg/m3), dogs
(200-385 ppm; 966-1860 mg/m3), and monkeys (210 ppm; 1014 mg/m3)
exposed repeatedly via inhalation to 2-butoxyethanol for up to
approximately 30-35 days (Werner et al., 1943b; Carpenter et al.,
1956).
Dodd et al. (1983) exposed Fischer 344 rats of both sexes to 0,
20, 86, or 245 ppm (0, 97, 415, or 1183 mg/m3) 2-butoxyethanol, 6
h/day for 9 days in total (5 consecutive days of exposure, followed by
2 days of no exposure, then 4 additional consecutive days of
exposure). In both sexes, exposure to 245 ppm (1183 mg/m3) was
associated with a significant reduction in red blood cell counts
( p < 0.001), haemoglobin levels ( p < 0.001), and mean cell
haemoglobin concentration ( p < 0.01), as well as a significant
increase ( p < 0.001 in all cases) in mean cell volume, nucleated
red blood cells, and reticulocytes. Fourteen days post-exposure, a
substantial recovery of the affected erythroid parameters was
observed; however, statistically significant differences from controls
were still observed for the males (i.e. red blood cell count
[ p < 0.01], mean cell volume [ p < 0.001], and mean cell
haemoglobin [ p < 0.001]). Exposure of both sexes to 86 ppm (415
mg/m3) 2-butoxyethanol was associated with a significant but less
profound effect on erythroid parameters. The no-observed-adverse-
effect level (NOAEL) in this study is 20 ppm (97 mg/m3).
In a study designed primarily to assess developmental effects,
Tyl et al. (1984) exposed pregnant Fischer 344 rats (36 per group) and
New Zealand white rabbits (24 per group) to 2-butoxyethanol (0, 25,
50, 100, or 200 ppm; 0, 121, 242, 483, or 966 mg/m3) for 6 h/day on
days 6-15 of gestation for the rats and on days 6-18 of gestation for
the rabbits. In rats, there were significant reductions in red blood
cell count and significant increases in haemoglobin and haematocrit at
200 ppm (966 mg/m3) ( p < 0.001); the red blood cell count was also
reduced at 100 ppm (483 mg/m3) ( p < 0.001). In dams exposed to
100 or 200 ppm (483 or 966 mg/m3) 2-butoxyethanol, mean cell volume
and mean cell haemoglobin were significantly increased relative to
controls; the mean cell haemoglobin concentration was significantly
reduced at 100 ppm (483 mg/m3) 2-butoxyethanol ( p < 0.01) and 200
ppm (966 mg/m3) 2-butoxyethanol ( p < 0.001), relative to controls.
In the rabbits, statistically significant increases in haemoglobin
content and haematocrit were observed at 100 ppm (483 mg/m3)
( p < 0.01) but not at 200 ppm (966 mg/m3) 2-butoxyethanol. The
results of this study indicate that rats are more sensitive than
rabbits to the haemolytic effects of 2-butoxyethanol. The NOAEL in
this study is 50 ppm (242 mg/m3) 2-butoxyethanol.
The oral administration of 500 or 1000 mg 2-butoxyethanol/kg body
weight per day for 4 consecutive days to male F344 rats produced a
pronounced dose-dependent effect on circulating red and white blood
cells (Grant et al., 1985); however, some effects were reversible
following the end of exposure. Reduced erythrocyte counts,
haematocrit, haemoglobin levels, and leukocyte counts and elevated
mean cell volume, reticulocyte counts, and mean cell haemoglobin
concentration ( p < 0.001) were observed in animals in the high-dose
group. Similar, although less severe, effects were observed in the
low-dose group.
To assess the development of tolerance to the haemolytic effects
of 2-butoxyethanol exposure in laboratory animals, male F344 rats were
administered (by gavage) 125 mg 2-butoxyethanol/kg body weight per day
for 0, 1, 2, 3, 6, and 12 days, and haematological parameters (red
blood cell counts, haemoglobin content, haematocrit) were determined
after exposure (Ghanayem et al., 1987a). Administration of
2-butoxyethanol for 2 and 3 days caused significant haemolysis of red
blood cells, although after the third day there was a gradual increase
in the number of red blood cells and haemoglobin content. After 12
days, red blood cells and haemoglobin approached pre-exposure levels,
indicative of the development of tolerance to the haemolytic effects
of 2-butoxyethanol. In a follow-up study, Ghanayem et al. (1992)
assessed the haemolytic effects of 2-butoxyethanol (administered as a
single dose of 0, 125, or 250 mg/kg body weight) in untreated
(control) or 2-butoxyethanol-pretreated male F344 rats. The
pretreated animals were administered (by gavage) 125 mg
2-butoxyethanol/kg body weight per day for 3 days and then allowed to
recover for 7 days prior to study. The pretreated animals were less
sensitive to the haemolytic effects of subsequent exposure to
2-butoxyethanol than the untreated controls. In vitro incubations
with 2-butoxyacetic acid revealed that red blood cells from the
2-butoxyethanol-pretreated group were less sensitive than cells from
the untreated controls. The authors suggested that the development of
tolerance to the haemolytic effects of 2-butoxyethanol might be due in
part to the reduced sensitivity of young erythrocytes formed during
the blood regeneration process.
In mice orally administered 500 or 1000 mg 2-butoxyethanol/kg
body weight per day, 5 days/week for 5 weeks, no effect upon white
blood cell counts, mean cell volume, or haemoglobin levels was
observed; however, red blood cell counts were reduced at both doses
(Nagano et al., 1979). The oral administration of 222, 443, or 885 mg
2-butoxyethanol/kg body weight per day, 5 days/week for 6 weeks, to
male rats principally affected red blood cells, whereas white blood
cell counts were unaffected (Krasavage, 1986).
In a study in which F344/N rats and B6C3F1 mice were
administered 2-butoxyethanol in drinking-water daily for 2 weeks,
estimates of 2-butoxyethanol intake by rats and mice ranged from 70 to
300 mg/kg body weight per day and from 90 to 1400 mg/kg body weight
per day, respectively (NTP, 1993). Survival of both species was not
affected by exposure to 2-butoxyethanol. Statistically significant
decreases ( p < 0.05) in relative and absolute thymus weights were
noted in male mice receiving 400 or 650 mg 2-butoxyethanol/kg body
weight per day. No haematological tests were conducted in this study.
8.4 Long-term exposure
8.4.1 Subchronic exposure
In older studies, haematotoxic effects (e.g. increased osmotic
fragility, decreased haemoglobin, decreased red blood cell numbers)
have been observed in mice (100-400 ppm; 483-1932 mg/m3), dogs (415
ppm; 2004 mg/m3), and monkeys (100 ppm; 483 mg/m3) exposed
repeatedly by inhalation to 2-butoxyethanol for up to approximately 90
days (Werner et al., 1943c; Carpenter et al., 1956). More recent
studies on effects associated with the subchronic exposure of
laboratory animals to 2-butoxyethanol are limited.
Dodd et al. (1983) exposed Fischer 344 rats of both sexes (16 per
group) to 0, 5, 25, or 77 ppm (0, 24, 121, or 372 mg/m3)
2-butoxyethanol by inhalation, 6 h/day, 5 days/week, for 13 weeks.
After 6 weeks, animals exposed to 77 ppm (372 mg/m3) 2-butoxyethanol
had a slight but statistically significant decrease in red blood cell
counts ( p < 0.01) and haemoglobin level (statistics not reported),
accompanied by an 11% increase in mean cell haemoglobin concentration
( p < 0.001). At the end of the study, these effects had either
lessened or returned to the ranges of control values. The only
significant haemolytic effect for male rats in the 77 ppm (372 mg/m3)
2-butoxyethanol exposure group was a 5% decrease in red blood cell
count after 66 exposures to 2-butoxyethanol (statistics not provided).
The NOAEL in this study is 25 ppm (121 mg/m3).
Groups of F344/N rats and B6C3F1 mice (10 per sex per
concentration) were administered 2-butoxyethanol in drinking-water (0,
750, 1500, 3000, 4500, or 6000 mg/litre) daily for 13 weeks; estimated
intakes by rats and mice ranged from 70 to 500 mg/kg body weight per
day and from 100 to 1300 mg/kg body weight per day, respectively (NTP,
1993). Effects observed in both species included decreased body
weight gain and water consumption. In rats, reduced red blood cell
counts and histopathological lesions in the liver, spleen, and bone
marrow were observed in males and females (at concentrations of
3000-6000 mg/litre and 750-6000 mg/litre, respectively). Reduced
thymus weights (at concentrations of 4500 and 6000 mg/litre in males
and females, respectively), diminished uterine size (at 4500 and 6000
mg/litre in females), and diminished sperm concentration (750-6000
mg/litre in males) were also noted. A NOAEL could not be identified
owing to a mild to moderate anaemia present in most dose groups of
rats. In mice, the only effect observed was reduced body weight gain
in males and females at concentrations of 3000-6000 mg/litre.
8.4.2 Chronic exposure and carcinogenicity
Published information on effects associated with the chronic
exposure of laboratory animals to 2-butoxyethanol was not
identified.1
8.5 Genotoxicity and related end-points
2-Butoxyethanol has been tested for genotoxicity in a range of
in vitro and in vivo assays (see Elliott & Ashby, 1997, for a
recent review). In standard tests in bacteria, 2-butoxyethanol was
not mutagenic in Salmonella typhimurium strains TA1535, TA1537,
TA97, TA98, TA100, and TA102 (Zeiger et al., 1992; Hoflack et al.,
1995; Gollapudi et al., 1996). However, the results for strain TA98a
were inconsistent, with one report of mutagenicity observed in both
the presence and absence of metabolic activation (Hoflack et al.,
1995) and another report of no mutagenicity (Gollapudi et al., 1996).
2-Butoxyethanol was not mutagenic at the HPRT locus in Chinese
hamster ovary cells in either the presence or absence of metabolic
activation (McGregor, 1984; Chiewchanwit & Au, 1995). However, there
was evidence that it caused gene mutations at the HPRT locus in
Chinese hamster lung (V79) cells (Elias et al., 1996). An in vitro
assay for unscheduled DNA synthesis in rat hepatocytes yielded
equivocal results (Elliott & Ashby, 1997). 2-Butoxyethanol produced
sister chromatid exchanges in human peripheral lymphocytes but not in
Chinese hamster lung (V79) or ovary cells. In vitro cytogenetic
assays conducted with human peripheral lymphocytes, Chinese hamster
lung (V79) cells, and Chinese hamster ovary cells revealed no
induction of chromosomal aberrations. An in vitro micronucleus
assay in Chinese hamster lung (V79) cells, which incorporated a test
for aneuploidy, yielded equivocal results (Elliott & Ashby, 1997).
In vivo mutagenicity tests have yielded uniformly negative
results for 2-butoxyethanol. These assays have included three bone
marrow micronucleus tests utilizing intraperitoneal injection in rats
and mice (Elias et al., 1996; Elliott & Ashby, 1997); a
[32P]post-labelling assay for DNA adducts in the brain, kidney,
liver, spleen, and testes of orally dosed rats (Keith et al., 1996);
an assay for DNA methylation in the brain, kidney, liver, spleen, and
testes of rats and in FVB/N transgenic mice carrying the v-Ha- ras
1 Results of a US National Toxicology Program 2-year carcinogenesis
bioassay completed in July 1995 were not available at the time this
CICAD was prepared.
oncogene (Keith et al., 1996); as well as a test for tumour formation
in FVB/N transgenic mice (Keith et al., 1996). Although the results
of in vitro tests for mutagenicity of 2-butoxyethanol are
inconsistent, the absence of structural alerts as well as the negative
results from in vivo studies are sufficiently reassuring to allow
the conclusion that 2-butoxyethanol is not mutagenic.
Mutagenicity studies have also been performed on two metabolites
of 2-butoxyethanol -- 2-butoxyacetic acid and 2-butoxyacetaldehyde.
2-Butoxyacetic acid was not mutagenic in a series of in vitro
assays, in addition to an in vivo micronucleus assay in mice
administered the chemical by intraperitoneal injection (Hoflack et
al., 1995; Elias et al., 1996; Elliott & Ashby, 1997).
2-Butoxyacetaldehyde exhibited mutagenic potential in several
in vitro studies (including tests for HPRT gene mutation,
chromosomal aberrations, micronuclei, aneuploidy, and sister chromatid
exchange); however, in the absence of data from in vivo studies, it
is not possible to reach a final conclusion concerning the possible
mutagenic hazard of this metabolite (Chiewchanwit & Au, 1995; Hoflack
et al., 1995; Elias et al., 1996; Elliott & Ashby, 1997).
8.6 Reproductive and developmental toxicity
Effects on the testes were not observed in studies in which
Alpk/Ap (Wistar-derived) rats were exposed by inhalation to 800 ppm
(3864 mg/m3) 2-butoxyethanol for 3 h (Doe, 1984), JCL-ICR mice were
orally administered 2-butoxyethanol at doses ranging from 500 to 2000
mg/kg body weight per day, 5 days/week, for 5 weeks (Nagano et al.,
1979), or rats were administered 2-butoxyethanol (by gavage) at doses
ranging from 222 to 885 mg/kg body weight per day, 5 days/week, for 6
weeks (Krasavage, 1986). Testicular damage was not observed in groups
of Alpk/Ap (Wistar-derived) rats administered a single oral dose of
174, 434, or 868 mg 2-butoxyacetic acid/kg body weight (Foster et al.,
1987).
No adverse effects were observed in either the dams or pups
(number of resorptions, fetal weights, and incidence of malformations)
in a study in which Sprague-Dawley rats were exposed by inhalation for
7 h/day on days 7-15 of gestation to 150 or 200 ppm (725 or 966
mg/m3) 2-butoxyethanol (Nelson et al., 1984); exposure to 250 or 500
ppm (1208 or 2415 mg/m3) 2-butoxyethanol caused death in the dams.
Tyl et al. (1984) exposed pregnant Fischer 344 rats (36 per
group) and New Zealand white rabbits (24 per group) to 0, 25, 50, 100,
or 200 ppm (0, 121, 242, 483, or 966 mg/m3) 2-butoxyethanol for 6
h/day on days 6-15 of gestation for the rats and on days 6-18 of
gestation for the rabbits. No adverse reproductive or developmental
effects were observed in rats or rabbits exposed to 25 ppm or 50 ppm
(121 or 242 mg/m3) 2-butoxyethanol. In rats, exposure to 200 ppm
(966 mg/m3) 2-butoxyethanol was associated with a reduction in
maternal weight gain, a significant ( p < 0.01) increase in the
number of totally resorbed litters, and a reduction in the number of
viable implants ( p < 0.001) and in the percentage of live fetuses
( p < 0.01) per litter. However, there were no statistically
significant increases in incidences of external, visceral, skeletal,
or total malformations associated with exposure to 2-butoxyethanol.
Exposure to 200 ppm (966 mg/m3) 2-butoxyethanol was also associated
with a significant increase ( p < 0.05) in the number of litters
with one or more fetuses with unossified skeletal elements and poorly
ossified skeletal elements. There was a decreased incidence of
bilobed cervical centrum 5, bilobed thoracic centra 9 and 13, as well
as poorly ossified proximal phalanges of the hindlimb. Following
maternal exposure to 100 ppm (483 mg/m3) 2-butoxyethanol, skeletal
ossification in the fetuses was retarded, with a significant
( p < 0.05) decreased (primarily because at these exposure
concentrations this skeletal element was largely unossified) incidence
of bilobed cervical centrum 5 and an increased incidence ( p < 0.05)
of unossified cervical centrum 6. In rabbits, exposure to 200 ppm
(966 mg/m3) 2-butoxyethanol produced a significant reduction in
maternal body weight, gravid uterine weight, and numbers of total
implants and viable implants. No significant increases in the number
of fetuses or litters with malformations were observed in any
treatment group; however, exposure to 200 ppm (966 mg/m3)
2-butoxyethanol was associated with a significant ( p < 0.05)
reduction in unossified sternebra 6 and in rudimentary rib at the
first lumbar rib. The occurrence of unossified skeletal elements in
both rats and rabbits was an indication of delayed development in rats
and rabbits exposed to 2-butoxyethanol under maternally toxic
conditions (Tyl et al., 1984).
Maternal deaths and a reduction in the number of viable litters
were observed in a study in which CD-1 mice were orally administered
4000 mg 2-butoxyethanol/kg body weight per day on days 7-14 of
gestation (Schuler et al., 1984).
Heindel et al. (1990) used a continuous breeding protocol
(Heindel et al., 1989) to assess the reproductive toxicity of
2-butoxyethanol. Male and female Swiss CD-1 mice were administered
2-butoxyethanol in drinking-water (0, 0.5, 1.0, or 2.0%; equivalent to
0, 0.7, 1.3, and 2.1 g/kg body weight per day) 7 days prior to and
during a 98-day cohabitation period (20 pairs of mice per dose).
Exposure to 1.0% or 2.0% 2-butoxyethanol in drinking-water was
associated with increased mortality in the females and a significant
reduction ( p < 0.05) in the number of live pups per litter, the
proportion of pups born alive, and the live pup weights (both absolute
and adjusted). The authors noted that these effects occurred in the
presence of maternal toxicity, as evidenced by decreased body weight,
decreased water consumption, and increased kidney weight in the female
mice. Necropsy revealed that testes and epididymis weights were
normal, as were sperm number and motility. The reproductive toxicity
of 2-butoxyethanol was evident only in female mice, at doses that also
elicited general toxicity (Heindel et al., 1990).
No maternal, embryotoxic, fetotoxic, or teratogenic effects were
detected when 2-butoxyethanol (106 mg) was applied to the shaved
interscapular skin of female Sprague-Dawley rats, four times daily on
days 7-14 of gestation (Hardin et al., 1984).
8.7 Immunological and neurological effects
Effects on the immune system were examined in two studies in
which 2-butoxyethanol was administered orally, by drinking-water or
gavage. In the first study, Sprague-Dawley rats were administered
2-butoxyethanol at 0, 2000, or 6000 mg/litre (males) or 0, 1600, or
4800 mg/litre (females) in drinking-water for 21 consecutive days.
Exposure to 2-butoxyethanol had no effect on antibody production,
delayed-type hypersensitivity reactions, and interferon or
interleukin-2 production. However, natural killer cell cytotoxicity
responses were enhanced ( p < 0.05) in rats receiving the lowest
concentrations of 2-butoxyethanol (Exon et al., 1991). In the second
study, male Fischer rats were administered (by gavage) 0, 50, 100,
200, or 400 mg 2-butoxyethanol/kg body weight per day for 2
consecutive days, following immunization with
trinitrophenyl-lipopolysaccharide. A reduction ( p < 0.05) in the
serum haemagglutination titre was observed 3 days later in rats
administered 200 mg 2-butoxyethanol/kg body weight per day. All
animals in the highest dose group died (Smialowicz et al., 1992).
No specific investigations on potential neurological effects
associated with exposure to 2-butoxyethanol were identified. However,
adverse effects on the central nervous system associated with exposure
to 2-butoxyethanol have been observed. These included loss of
coordination, sluggishness, narcosis, muscular flaccidity, and ataxia
(Carpenter et al., 1956; Dodd et al., 1983; Hardin et al., 1984;
Krasavage, 1986).
8.8 In vitro haemolytic effects
Bartnik et al. (1987) examined the effects of 2-butoxyethanol and
2-butoxyacetic acid on human (from healthy males) and rat (four male
Wistar) erythrocytes in vitro. Under these conditions 175, 200, 225,
and 250 mmol 2-butoxyethanol/litre induced complete lysis of rat
erythrocytes, whereas only 200, 225, and 250 mmol
2-butoxyethanol/litre induced complete lysis of human erythrocytes.
Although 3.75-7.5 mmol 2-butoxyacetic acid/litre caused complete lysis
of rat erythrocytes, lysis of human erythrocytes was not observed at
these concentrations. These results indicate that rats may be more
susceptible than humans to the haemolytic effects of 2-butoxyethanol
and its metabolite 2-butoxyacetic acid (Bartnik et al., 1987).
Ghanayem (1989) examined the effect of 2-butoxyethanol and
2-butoxyacetic acid on blood collected by cardiac puncture from male
F344 rats. The addition of 2-butoxyethanol to whole blood to
concentrations of 5 or 10 mmol/litre had no effect on haematocrit,
whereas a concentration of 20 mmol/litre caused significant haemolysis
( p < 0.05). The addition of 2-butoxyacetic acid to rat
erythrocytes to concentrations of 0.5 or 1 mmol/litre caused a time-
and concentration-dependent increase in haematocrit followed by
haemolysis. Incubation with 2 mmol 2-butoxyacetic acid/litre caused a
faster time-dependent increase in haematocrit, with the haematocrit
reaching a maximum after 2 h, followed by nearly complete haemolysis
after 4 h. Also examined was the effect of 2-butoxyacetic acid (0.5,
1, 2, 4, or 8 mmol/litre) on human blood obtained from healthy young
male and female volunteers (Ghanayem, 1989). No significant changes
in haematocrit or haemolysis were observed at 2-butoxyacetic acid
concentrations of 4 mmol/litre or less; at 8 mmol 2-butoxyacetic
acid/litre, there was a slight but significant increase in haematocrit
( p < 0.05), followed by a slight but significant haemolysis
( p < 0.05) of erythrocytes.
In a subsequent study, Ghanayem & Sullivan (1993) assessed the
haemolytic activity of 2-butoxyacetic acid (1 or 2 mmol/litre) in
blood collected from a variety of species (i.e. rats, mice, hamsters,
baboons, rabbits, pigs, guinea-pigs, dogs, cats, and humans).
2-Butoxyacetic acid caused a time- and concentration-dependent
increase in mean cell volume and haematocrit of blood from rats,
rabbits, hamsters, mice, and baboons. However, no or minimal effects
were observed on blood from humans, guinea-pigs, dogs, cats, and pigs
(Ghanayem & Sullivan, 1993), demonstrating the sensitivity of rat
erythrocytes and the relative insensitivity of human erythrocytes to
the haemolytic effects of 2-butoxyacetic acid.
The effect of 2-butoxyacetic acid on red blood cells from healthy
young and older individuals (Udden & Patton, 1994) and individuals
with a possible susceptibility to 2-butoxyethanol-induced haemolysis
(i.e. sickle cell and spherocytosis patients) (Udden, 1994, 1996) has
also been examined. Along with haemolysis, 0.2 and 2 mmol
2-butoxyacetic acid/litre caused decreased red blood cell
deformability and increased mean cellular volume in rat red blood
cells (Udden & Patton, 1994). However, none of the human blood
samples exhibited prehaemolytic changes (i.e. decreased deformability
and increased mean cellular volume) or haemolysis after treatment with
2 mmol 2-butoxyacetic acid/litre for up to 4 h (Udden, 1994, 1996;
Udden & Patton, 1994). The results of these in vitro studies
provide further evidence that rat erythrocytes are more susceptible
than human erythrocytes to 2-butoxyacetic acid-induced haemolysis.
9. EFFECTS ON HUMANS
Information on human health effects associated with exposure to
2-butoxyethanol are limited to a few case reports and one laboratory
investigation; epidemiological studies have not been identified. The
principal human health effects attributed to 2-butoxyethanol exposure
have involved the central nervous system, the blood, and the kidneys
(ATSDR, 1996).
In one report involving a number of small studies, the exposure
of two males to 113 ppm (546 mg/m3) 2-butoxyethanol for 4 h produced
nose and eye irritation as well as disturbed taste, but there was no
evidence of haemolytic effects. Similar effects were observed in a
second study in which two males and one female were exposed to 195 ppm
(942 mg/m3) 2-butoxyethanol for two 4-h periods, separated by a
30-min period of no exposure. When two males and two females were
exposed to 100 ppm (483 mg/m3) 2-butoxyethanol for 8 h, the effects
included vomiting and headache. No clinical signs of haemolysis were
observed in any of the subjects; however, following exposure to 195
ppm (942 mg/m3) 2-butoxyethanol, increased osmotic fragility of
erythrocytes was observed in vitro (Carpenter et al., 1956).
Haemoglobinuria, erythropenia, and hypotension (Rambourg-Schepens
et al., 1988; Gijsenbergh et al., 1989), metabolic acidosis, shock,
non-cardiogenic pulmonary oedema, and albuminuria (Bauer et al.,
1992), and metabolic acidosis, hepatic laboratory abnormalities, and
haematuria (Gualtieri et al., 1995) have been reported in case-studies
of individuals who had attempted suicide by ingesting 2-butoxyethanol-
containing cleaning solutions (involving an estimated ingestion of
25-60 g 2-butoxyethanol). In two of the cases, haemodialysis was
employed, and all patients recovered fully with appropriate treatment.
A survey of paediatric poisonings identified 24 children who had
ingested 5-300 ml of glass cleaners containing 2-butoxyethanol (Dean &
Krenzelok, 1992). The two children with the highest intake exhibited
no evidence of haemolytic effects. 2-Butoxyethanol is reportedly not
a skin sensitizer in humans (Greenspan et al., 1995).
10. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
10.1 Aquatic environment
Results of acute and long-term studies on toxicity to aquatic
organisms are summarized in Table 1. Long-term studies are restricted
to microorganisms and unicellular algae, for which 72 h is the cut-off
point for the designation of acute/long-term studies.
10.2 Terrestrial environment
Information on the toxicological effects of 2-butoxyethanol on
terrestrial organisms was not identified.
Table 1: Acute and long-term studies on toxicity to aquatic organisms.
Species End-pointa Concentration Reference
(mg/litre)
Freshwater
Bacterium (Pseudomonas putida) 16-h LOEC (growth) 700 Bringmann & Kuhn, 1980a
Sewage sludge bacteria 16-h IC50 >1000 Union Carbide, 1989
Protozoan (Entosiphon sulcatum) 72-h LOEC (growth) 91 Bringmann & Kuhn, 1980a
Protozoan (Chilomonas paramecium) 48-h EC5 (growth) 911 Bringmann & Kuhn, 1980b
Protozoan (Uronema parduczi) 48-h EC5 (growth) 463 Bringmann & Kuhn, 1980b
Cyanobacterium (Microcystis aeruginosa) 8-day LOEC (growth) 35 Bringmann & Kuhn, 1980a
Green alga (Scenedesmus quadricaudata) 7-day LOEC (growth) 900 Bringmann & Kuhn, 1980a
Green alga (Selenastrum capricornutum) 7-day NOEC 125 Dow, 1988
7-day EC50 >1000
Water flea (Daphnia magna) 24-h LC50 1720 Bringmann & Kuhn, 1977
24-h LC50 1698-1940 Bringmann & Kuhn, 1982
24-h LC50 5000 CMA, 1994
48-h LC50 835 Dow, 1979
Guppy (Poecilia reticulata) 7-day LC50 982 Koenemann, 1981
Golden ide (Leuciscus idus melanotus) 48-h LC50 165-186 Junke & Ludemann, 1978
48-h LC50 1880 CMA, 1994
Bluegill sunfish (Lepomis macrochirus) 96-h LC50 1490 Dawson et al., 1977
Goldfish (Carassius auratus) 24-h LC50 1700 Bridie, 1979
24-h LC50 1650 Verschueren, 1983
Fathead minnow (Pimephales promelas) 96-h LC50 2137 Dow, 1979
Emerald shiner (Notropus atherinoides) 72-h LC50 >500 Dill, 1995
Rainbow trout (Oncorhynchus mykiss) 96-h LC50 >1000 Environment Canada, 1997
Estuarine/marine
Oyster (Crassostrea virginica) 96-h LC50 89 US EPA, 1984
White shrimp (Penaeus setiferus) 96-h LC50 130 OECD, 1997
Grass shrimp (Palaemonetes pugio) 96-h LC50 5.4 Environment Canada, 1997
Brown shrimp (Crangon crangon) 48-h LC50 600-1000 Verschueren, 1983
96-h LC50 550-950
Table 1: (continued)
Species End-pointa Concentration Reference
(mg/litre)
Brine shrimp (Artemia salina) 24-h LC50 1000 Price et al., 1974
Inland silverside (Menidia beryllina) 96-h LC50 1250 Dawson et al., 1977
Sheepshead minnow (Cyprinodon variegatus) 96-h LC50 116 OECD, 1997
a NOEC = no-observed-effect concentration; LOEC = lowest-observed-effect concentration.
11. EFFECTS EVALUATION
11.1 Evaluation of health effects
11.1.1 Hazard identification and dose-response assessment
In general, effects associated with exposure to 2-butoxyethanol
have been identified from studies in animals. 2-Butoxyethanol has
moderate acute toxicity following inhalation, ingestion, or dermal
exposure. It is an eye and skin irritant, but it is not a skin
sensitizer. 2-Butoxyethanol is readily absorbed via inhalation,
dermal exposure, and ingestion. The pharmacokinetic models of Corley
et al. (1994, 1997) and Johanson & Boman (1991) consider dermal
absorption from the vapour phase to account for approximately 21-75%
of the total uptake. Pathways for the metabolism of 2-butoxyethanol
are similar in animals and humans; the principal metabolite is
2-butoxyacetic acid.
The principal effect exerted by 2-butoxyethanol and its
metabolite 2-butoxyacetic acid is haematotoxicity, with rats being the
most sensitive species. Older rats are more sensitive than younger
animals to the haemolytic effects of 2-butoxyethanol and
2-butoxyacetic acid. Critical effects observed in inhalation studies
conducted with rats were decreased haemoglobin and mean cell
haemoglobin; increased haematocrit and mean cell volume (NOAEL = 20
ppm [97 mg/m3]; lowest-observed-adverse-effect level [LOAEL] = 86 ppm
[415 mg/m3] in animals exposed for 9 days; Dodd et al., 1983);
decreased red blood cells and haemoglobin (NOAEL = 25 ppm [121
mg/m3]; LOAEL = 77 ppm [372 mg/m3]) in animals exposed
subchronically (Dodd et al., 1983); and decreased red blood cells and
increased mean cell volume (NOAEL = 50 ppm [242 mg/m3]; LOAEL = 100
ppm [483 mg/m3]) in pregnant animals exposed on days 6-15 of
gestation (Tyl et al., 1984). The results of in vitro studies
indicate that human red blood cells are not as sensitive as rat red
blood cells to the haemolytic effects of 2-butoxyethanol and
2-butoxyacetic acid and also that red blood cells are more sensitive
to haemolysis by 2-butoxyacetic acid than to haemolysis by
2-butoxyethanol (Bartnik et al., 1987; Ghanayem, 1989; Ghanayem &
Sullivan, 1993; Udden, 1994; Udden & Patton, 1994).
In rats, adverse effects on the central nervous system, kidneys,
and liver occur at higher exposure concentrations than do haemolytic
effects. 2-Butoxyethanol (and, in one study, 2-butoxyacetic acid) did
not cause adverse reproductive or developmental effects in either sex
at less than toxic doses (Nagano et al., 1979; Doe, 1984; Hardin et
al., 1984; Nelson et al., 1984; Tyl et al., 1984; Foster et al., 1987;
Heindel et al., 1990). Although sperm concentration was reduced in
rats administered drinking-water containing 2-butoxyethanol (NTP,
1993), the reduction was not dose-dependent, and no change in sperm
cell morphology was observed. Although the results of in vitro
tests for mutagenicity of 2-butoxyethanol were inconsistent, the
absence of structural alerts and the negative findings from in vivo
studies are sufficiently reassuring to allow the conclusion that
2-butoxyethanol is not mutagenic. 2-Butoxyethanol has not been found
to have an adverse effect on the immune system (Exon et al., 1991;
Smialowicz et al., 1992).
Based on limited data from case reports and one laboratory study,
similar acute effects -- including haemolytic effects as well as
effects on the central nervous system -- are observed in humans and
rats exposed to 2-butoxyethanol, although the effects are observed at
much higher exposure concentrations in humans than in rats.
11.1.2 Criteria for setting guidance values for 2-butoxyethanol
The following guidance is provided as a possible basis for
derivation of limits of exposure and for judgement of the quality of
environmental media by relevant authorities. Available data indicate
that the haematotoxicity associated with exposure to 2-butoxyethanol
is similar in laboratory animals and humans, although available data
do not permit quantification of dose-response for the latter. The
guidance value provided here is derived, therefore, on the basis of
studies conducted in animals. Based on limited data in humans, the
rat is likely more sensitive to the haemolytic effects of
2-butoxyethanol exposure (Carpenter et al., 1956; Bartnik et al.,
1987).
The dose-response for haematotoxicity in rats has been consistent
in an inhalation study of developmental toxicity, in which pregnant
animals were exposed to 2-butoxyethanol on days 6-15 of gestation (Tyl
et al., 1984), and in a subchronic inhalation toxicity study (Dodd et
al., 1983). In the developmental study, the NOAEL and LOAEL in the
dams were 50 ppm (242 mg/m3) and 100 ppm (483 mg/m3), respectively
(Tyl et al., 1984). In the subchronic inhalation study, the NOAEL and
LOAEL were 25 ppm (121 mg/m3) and 77 ppm (372 mg/m3), respectively
(Dodd et al., 1983). A tolerable concentration (TC) has been derived
as follows:
TC = [(242 mg/m3)/10] × [6/24] × [(0.16 m3 per day/0.215
kg)/(22 m3 per day/64 kg)]
= 13.1 mg/m3
where:
* 242 mg/m3 (50 ppm) is the NOAEL from the study (Tyl et al.,
1984) providing the best bounding of dose-response in the most
sensitive species;
* 10 is the uncertainty factor to account for intraspecies
variability in humans. No additional factor was incorporated to
address interspecies variability on the basis of limited data in
humans and several in vitro studies that indicate that rat
erythrocytes are far more sensitive than human erythrocytes to
the haemolytic effects associated with exposure to
2-butoxyethanol (and its metabolite 2-butoxyacetic acid). No
additional factor was incorporated to account for the short
duration of exposure in the critical study, as there is no
indication that effect levels vary with increased exposure
duration;
* 6/24 is the conversion from 6 h/day to continuous exposure; and
* [(0.16 m3 per day/0.215 kg)/(22 m3 per day/64 kg)] is the
scaling factor from rats to humans, based on the assumed
inhalation volume and body weight for rats (0.16 m3 per day and
0.215 kg, respectively) and humans (22 m3 per day and 64 kg,
respectively). The PBPK model of Corley et al. (1994, 1997)
would not result in an appreciably different TC at this level of
exposure.
It should be noted that the TC was based on a study involving the
whole-body exposure of rats and an assumption that 100% of the inhaled
2-butoxyethanol was retained. The extent of skin absorption has not
been formally taken into account in the development of this TC and may
be greater than intake via inhalation.
11.1.3 Sample risk characterization
The extremely limited nature of the available data to serve as a
basis for estimation of exposure should be borne in mind in
interpreting the comparisons presented here for indirect exposure of
the general population to 2-butoxyethanol. The concentration of 8 µg
2-butoxyethanol/m3 measured in a sample of indoor air collected in
northern Italy is approximately 1600-fold lower than the TC developed
in the preceding section. Levels of 2-butoxyethanol are considerably
higher in some occupational settings. In addition, skin absorption
may be of greater importance than respiratory absorption.
11.2 Evaluation of environmental effects
11.2.1 Aquatic environment
Data on measured levels of 2-butoxyethanol in surface waters are
insufficient for risk characterization. However, a sample risk
characterization for the aquatic environment is presented in which the
ratio between a predicted (local) environmental concentration
(PEClocal) and a predicted no-effect concentration (PNEC) is
calculated.
PEClocals for surface waters have been derived based upon data
from Australia (OECD, 1997) as well as information on all reported
releases to the environment in 1993 from individual industrial plants
in the USA (Staples et al., 1998). Calculations of expected surface
water concentration were based on worst-case scenarios for local river
flows identified from a US Geological Survey database. Site-specific
estimates were made for 36 industrial plants, of which 26 discharged
through sewage treatment plants and 10 discharged directly to rivers.
Both studies relied on fugacity modelling to predict the environmental
distribution of 2-butoxyethanol, yielding slightly different results.
However, both approaches indicated that most (84-96%) of the chemical
will partition to water, with almost all of the remainder volatilizing
to air. There is negligible binding of 2-butoxyethanol to
particulates, and no bioconcentration in organisms is expected. In
addition, 2-butoxyethanol is readily degraded by microorganisms.
A PEC for surface water in Sydney, Australia, based on the
assumption that all local usage passes through a single sewage
treatment plant and releases at a point source to a river, was
calculated as follows:
PEClocal (water) = Ceffluent/[(1 + Kp(susp) × C(susp)) × D]
= 50.4 µg/litre
where:
* Ceffluent is the concentration (g/litre) of the chemical in the
sewage treatment plant effluent, calculated as Ceffluent = W ×
(100 × P)/(100 × Q)
where:
W = emission rate: 1400 kg/day (OECD, 1997)
P = % removal by biodegradation in the sewage
treatment plant (modelled as 91% using the
SIMPLETREAT model)
Q = volume of wastewater: 250 000 m3/day (OECD, 1997)
* Kp(susp) is the suspended matter/water adsorption coefficient),
calculated as Kp(susp) = Foc(susp) × Koc
where:
Foc(susp)= the fraction of organic carbon in suspended
matter (0.01)
Koc = 0.411 × Kow
where:
Kow = the octanol/water partition coefficient
(6.76)
* C(susp) is the concentration of suspended matter in river water
(default value = 15mg/litre)
* D is the dilution factor for river flow (default value = 10)
As degradation in the sewage treatment plant is a large component
of the assumptions, and as it cannot be assumed that this level of
sewage treatment occurs in all countries globally, this calculation
can be revised assuming no sewage treatment (i.e. P = 0), yielding a
PEC of 560 µg/litre. This value assumes that all local release is
diluted with general wastewater from the urban centre. No values were
available for individual industrial plants in Sydney, Australia, and
therefore concentrations released directly to rivers cannot easily be
calculated.
Using the other approach of site-specific estimation (Staples et
al., 1998), 36 industrial plants in the USA were selected from 814
reporting emissions, on the basis of availability of river flow values
and worst-case releases. Calculations were based on local stream
flows, taking a value for the lowest flow expected over any single
7-day period once in 10 years. For plants emitting via a sewage
treatment system, degradation rates of 90% were assumed. Calculated
concentrations are "instantaneous," assuming no dilution by the
receiving stream, no degradation in the receiving waters, and no
distribution to media other than water. These are conservative
assumptions. Calculated in-stream concentrations ranged from 0.0002
to 21.7 mg/litre for emissions via sewage treatment (annual release
ranged from 18 000 to 974 000 kg for the 26 plants with sewage
treatment) and from 0.000 01 to 4.66 mg/litre for untreated emissions
(annual release ranged from 1870 to 35 000 kg for the 10 plants with
no sewage treatment). The highest reported concentration of
2-butoxyethanol in surface waters was 5.7 mg/litre following release
by the leather industry into the Hayashida River in Japan, before
treatment was introduced (Yasuhara et al., 1981). These measured and
estimated surface water concentrations are summarized in Table 2.
Table 2: PEC/PNEC ratios.
Location Sewage Highest PEC/PNEC
treatment concentration ratioa
(mg/litre)
Australiab Yes 0.05 0.3
(Sydney) No 0.56 3.4
USA (site Yes 21.7 131.5
specific)c No 4.66 28.2
Japand No 5.7 34.5
a Based on a PNEC of 165 µg/litre (see text).
b Modelled.
c Modelled, but based on known annual release for each site.
d Measured.
As a guide for those wishing to perform similar calculations
using local use/release figures, the Staples et al. (1998) study
estimates that the annual release of total glycol ethers (assuming
that 50% of released compounds would be 2-butoxyethanol) leading to
instantaneous 2-butoxyethanol concentrations in surface waters of 1
mg/litre would be 18 000 kg with sewage treatment and 1800 kg without
sewage treatment for streams with very low flow at 0.03 m3/s
(equivalent to 2.5 million litres/day).
A PNEC for surface waters may be calculated as follows:
PNEC = (165 mg/litre)/1000
= 165 µg/litre
where:
* 165 mg/litre is the lowest reported effect level for a lethality
end-point in aquatic species (48-h LC50 in the golden ide
[ Leuciscus idus melanotus], a freshwater fish); and
* 1000 is the uncertainty factor. The range of organisms tested in
short-term tests would justify application of an uncertainty
factor of 100, yielding a PNEC of 1.65 mg/litre, based on the
lowest reported LC50 in fish. However, there is some indication
that estuarine species may be more sensitive, although the lowest
reported LC50 for the grass shrimp ( Palaemonetes pugio) (96-h
LC50 = 5.4 mg/litre) is such an extreme outlier compared with
the range of other data that it is difficult to justify its use
as the basis for the PNEC calculation. Application of an
uncertainty factor of 1000 to the lowest freshwater value would
be protective for both freshwater and estuarine environments,
yielding margins relative to the 96-h LC50s for the grass shrimp
(5.4 mg/litre) and the oyster ( Crassostrea virginica) (89
mg/litre), the most sensitive of the estuarine invertebrates, of
33 and 540, respectively. For freshwater organisms, the
threshold concentration for inhibition of growth in algae
(long-term effect) cannot be justified as the basis for
application of uncertainty factors to establish a PNEC.
As the highest measured concentration in surface waters (at 5.7
mg/litre) is almost identical to the lowest reported LC50
concentration (at 5.4 mg/litre for the grass shrimp), it is not
surprising that high risk factors are generated. High-volume usage
and emissions to surface waters in a range of industries would lead to
locally high concentrations, principally where sewage treatment was
not in operation and river flow was low. It can be expected that
concentrations would exceed those likely to produce effects in some
aquatic species under these circumstances. However, the majority of
reported acute toxicity effect levels are 100 mg/litre or higher, and
most exceed 800 mg/litre. Four of 38 estimated surface water
concentrations exceed 2 mg/litre, with the remainder less than, and
usually substantially less than, 1 mg/litre (Figure 1). Most of these
estimates also fail to account for dilution in rivers. Using an
uncertainty factor of 100, justified by the range of toxicity data, on
the lowest reported freshwater LC50 and typical estimates of water
concentrations yields PEC/PNEC ratios of <1. Therefore, for most
releases to surface waters, the risk is considered to be low. It is
also unlikely that 2-butoxyethanol would be toxic to sewage treatment
plant bacteria, as the only reported effect level for bacteria is an
IC50 of >1000 mg/litre (Union Carbide, 1989).
1. EXECUTIVE SUMMARY
This CICAD on 2-butoxyethanol was based upon reviews prepared by
the National Institute for Occupational Safety and Health (NIOSH,
1990) and the Agency for Toxic Substances and Disease Registry (ATSDR,
1996). Additional data were identified through an updated literature
search to May 1997, as well as during the peer review of this CICAD.
Information on the nature of the peer review and availability of the
source documents is presented in Appendix 1. Information on the peer
review of this CICAD is presented in Appendix 2. This CICAD was
approved as an international assessment at a meeting of the Final
Review Board, held in Berlin, Germany, on 26-28 November 1997.
Participants at the Final Review Board meeting are listed in Appendix
3. The International Chemical Safety Card (ICSC 0059) produced by the
International Programme on Chemical Safety (IPCS, 1993) has also been
reproduced in this document.
2-Butoxyethanol (CAS no. 111-76-2) is a high-production-volume
glycol ether. It is a colourless liquid that is miscible in water and
soluble in most organic solvents. 2-Butoxyethanol is used widely as a
solvent in surface coatings, such as spray lacquers, quick-dry
lacquers, enamels, varnishes, varnish removers, and latex paint. It
is also used in metal and household cleaners. 2-Butoxyethanol exists
in the atmosphere almost entirely as a vapour; because the chemical
has an atmospheric half-life of approximately 17 h, the risk for
transport via the atmosphere should be small. The estimated half-life
of 2-butoxyethanol in water is approximately 1-4 weeks, and the
chemical is likely readily biodegraded in aerobic soil and water. Its
potential for bioaccumulation is low. Based upon limited data,
ambient exposures in air are generally in the µg/m3 range. Indirect
exposure of the general population to 2-butoxyethanol is most likely
from inhalation and dermal absorption during the use of products
containing the chemical. Levels of airborne 2-butoxyethanol in
occupational settings are typically in the mg/m3 range.
2-Butoxyethanol is readily absorbed following inhalation, oral,
and dermal exposure. The chemical is metabolized primarily via
alcohol and aldehyde dehydrogenases, with the formation of
2-butoxyacetaldehyde and 2-butoxyacetic acid, the principal
metabolite, although other metabolic pathways have also been
identified.
2-Butoxyethanol has moderate acute toxicity and is irritating to
the eyes and skin; it is not a skin sensitizer. The principal effect
exerted by 2-butoxyethanol and its metabolite 2-butoxyacetic acid is
haematotoxicity, with the rat being the most sensitive species. The
results of in vitro studies indicate that human red blood cells are
not as sensitive as rat red blood cells to the haemolytic effects of
2-butoxyethanol and 2-butoxyacetic acid and also that red blood cells
are more sensitive to haemolysis by 2-butoxyacetic acid than to
haemolysis by 2-butoxyethanol. In rats, adverse effects on the
central nervous system, kidneys, and liver occur at higher exposure
concentrations than do haemolytic effects. In animals, adverse
effects on reproduction and development have not been observed at less
than toxic doses. Although the results of in vitro tests for
mutagenicity of 2-butoxyethanol were inconsistent, the absence of
structural alerts and the negative findings from in vivo studies are
sufficiently reassuring to allow the conclusion that 2-butoxyethanol
is not mutagenic. Based on limited data from case reports and one
laboratory study, similar acute effects -- including haemolytic
effects as well as effects on the central nervous system -- are
observed in humans and rats exposed to 2-butoxyethanol, although the
effects are observed at much higher exposure concentrations in humans
than in rats. Based upon the development of haemolytic effects in
pregnant rats exposed during gestation, a sample tolerable
concentration for humans of 13.1 mg 2-butoxyethanol/m3 has been
derived.
Based upon extremely conservative assumptions, the highest
predicted concentrations of 2-butoxyethanol in surface waters in the
immediate vicinity of effluent streams may, in some cases, exceed
predicted no-observed-effect concentrations. However, more realistic
assumptions based on the available data suggest that risk to aquatic
organisms is low. Owing to the short half-life of 2-butoxyethanol in
the atmosphere, measured or predicted concentrations of this chemical
in air are considered to have no environmental significance.
2. IDENTITY AND PHYSICAL/CHEMICAL PROPERTIES
2-Butoxyethanol (CAS no. 111-76-2; C6H14O2; ethylene glycol
monobutyl ether, monobutyl glycol ether, 2-butoxy-1-ethanol,
2- n-butoxyethanol) is a synthetic glycol ether. It is a colourless
liquid with a mild ether odour; the odour threshold is approximately
0.10 ppm (0.48 mg/m3) (Amoore & Hautala, 1983). At ambient
temperature, 2-butoxyethanol is miscible in water and soluble in most
organic solvents. 2-Butoxyethanol has a boiling point of 171°C, a
vapour pressure of 0.1 kPa at 20°C, and a log octanol/water partition
coefficient of 0.83. The conversion equation for 2-butoxyethanol is
1 ppm = 4.83 mg/m3 (at 25°C, 101.3 kPa). Additional physical and
chemical properties are presented in the International Chemical Safety
Card reproduced in this document. The structural formula for
2-butoxyethanol is CH3CH2CH2CH2-O-CH2CH2OH.
3. ANALYTICAL METHODS
Laboratory analysis for 2-butoxyethanol in environmental samples
is usually by gas chromatography (GC) in combination with flame
ionization detection (FID), electron capture detection (ECD), or mass
spectrometric (MS) detection; infrared absorption spectrophotometry is
also sometimes used. The detection limits of these analytical methods
in air range from 0.031 ppm (0.15 mg/m3) for a 48-litre sample (OSHA,
1990) to 0.01-0.02 mg for 2- to 10-litre samples (NIOSH, 1994).
Multidimensional GC-MS has been used to improve the detection limit to
5-7 µg per sample (Kennedy et al., 1990).
Biological monitoring is a useful adjunct to environmental
measurements in assessing human exposure to 2-butoxyethanol, as it
accounts for both dermal and respiratory uptake. A variety of GC
methods combined with FID, ECD, or MS detection and high-performance
liquid chromatography (HPLC) methods coupled with ultraviolet or
radiochemical detection have been developed for the analysis of
2-butoxyethanol and its metabolite 2-butoxyacetic acid in the urine
and blood of exposed workers or rats.
In general, these methods are based on either extraction or
lyophilization of the blood or urine followed by derivatization and
then analysis (Smallwood et al., 1984, 1988; Groeseneken et al., 1986,
1989; Johanson et al., 1986, 1988; Rettenmeier et al., 1993; Sakai et
al., 1993, 1994; Corley et al., 1994). The detection limits range
from 0.03 to 0.1 mg 2-butoxyacetic acid/litre. 2-Butoxyethanol and
2-butoxyacetic acid in rat and human blood can be analysed by a GC-MS
derivatization method with a detection limit range of 16-18 ng/g blood
(Bormett et al., 1995). The National Institute for Occupational
Safety and Health reviewed the available data and developed guidelines
for biological monitoring of 2-butoxyacetic acid (NIOSH, 1990).
4. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
2-Butoxyethanol does not occur naturally. It is usually produced
by reacting ethylene oxide with butyl alcohol, but it may also be made
by the direct alkylation of ethylene glycol with an agent such as
dibutyl sulfate (Rowe & Wolf, 1982). Temperature, pressure, reactant
molar ratios, and catalysts are selected to give the product mix
desired.
2-Butoxyethanol is widely used as a solvent in surface coatings,
such as spray lacquers, quick-dry lacquers, enamels, varnishes,
varnish removers, and latex paint (Leaf, 1985; Sax & Lewis, 1987). In
surface coatings, it imparts blush resistance, gloss, and good
flow-out. 2-Butoxyethanol is also used as a coupling agent in metal
and household cleaners; as an intermediate in 2-butoxyethanol acetate
production; and in herbicides, automotive brake fluids, printing inks,
spot removers, and cosmetics (Leaf, 1985; ATSDR, 1996). In 1994,
176 900 tonnes of 2-butoxyethanol were produced in the USA (US ITC,
1996). Within the European Community, the total production capacity
of 2-butoxyethanol was approximately 70 000-90 000 tonnes in the same
year (ECETOC, 1994; CEFIC, 1995).
2-Butoxyethanol may be released into air or water by facilities
that manufacture, process, or use the chemical (ATSDR, 1996; US NLM,
1997). Products containing 2-butoxyethanol may also release the
substance into the air. Solvent-based building materials such as
silicone caulk will release 2-butoxyethanol to air as they dry. There
is potential for the release of 2-butoxyethanol from hazardous waste
sites, although quantitative data have not been identified. Based
upon the detection of 2-butoxyethanol in samples of groundwater and
surface water taken near municipal landfills and hazardous waste
sites, 2-butoxyethanol may be released to water in leachates from
these sites (ATSDR, 1996). Information on the total estimated release
of 2-butoxyethanol into the environment in the USA was not identified.
In Canada, emissions to the environment between 1992 and 1994 have
been reported to range from 1.4 to 3.1 tonnes per year (Canadian
Chemical Producers' Association, 1996).
5. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
In the atmosphere, 2-butoxyethanol is expected to exist in the
vapour phase. Owing to its water solubility, wet deposition is likely
to be more important than dry deposition (ATSDR, 1996). The chemical
will not persist in the atmosphere; it has an atmospheric half-life of
approximately 17 h, based on an estimated rate constant for reaction
with hydroxyl radicals (US NLM, 1997).
The miscibility of 2-butoxyethanol in water suggests that
volatilization from water, adsorption, and bioconcentration are not
important fate processes and that the chemical should not
bioconcentrate in aquatic organisms. Based upon aerobic
biodegradation rates, the half-life of 2-butoxyethanol in water is
estimated to range from 1 to 4 weeks (Howard et al., 1991).
2-Butoxyethanol is not likely to undergo direct hydrolysis in the
aquatic environment, and it is likely readily biodegraded (ATSDR,
1996). Five-day theoretical biological oxygen demand values range
from 5% (without acclimation) to 73% (with acclimation); 10-day
theoretical biological oxygen demand values range from 57% to 74%.
The maximum theoretical biological oxygen demand value reported is 88%
for 20 days (US NLM, 1997). Biodegradation is likely to be the most
important mechanism for the removal of 2-butoxyethanol from aerobic
soil and water.
6. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
6.1 Environmental levels
Limited data are available on the concentration of
2-butoxyethanol in environmental media. Reported levels of
2-butoxyethanol in samples of ambient air taken from Nepal and Europe
and from Antarctica have ranged from 0.1 to 1.59 µg/m3 and from 1.26
to 14.85 µg/m3, respectively (Ciccioli et al., 1993, 1996).
2-Butoxyethanol was detected at a concentration of 23 µg/litre in one
of seven groundwater samples collected near the Valley of Drums,
Kentucky, USA (ATSDR, 1996). Additional monitoring data on the
concentration of 2-butoxyethanol in surface waters and information on
levels in soils or sediments have not been identified. Levels below
100 µg 2-butoxyethanol/litre have been reported in samples of
industrial wastewater effluents in the USA (ATSDR, 1996). Water
samples obtained from a highly polluted site on the Hayashida River in
Japan, where effluent entered the river from the leather industry,
contained 1310 and 5680 µg 2-butoxyethanol/litre (Yasuhara et al.,
1981).
6.2 Human exposure
Quantitative information on levels of 2-butoxyethanol in
drinking-water and foodstuffs has not been identified, although the
chemical has been detected (levels not specified) in drinking-water in
six US cities, and there is the potential for the presence of
2-butoxyethanol in foods arising from labelling or packaging
materials. Data on concentrations of 2-butoxyethanol in indoor air in
the USA are limited to one report, in which the daily arithmetic mean
concentration was 0.214 ppbv (1 µg/m3) for samples obtained from 14
non-industrial offices. 2-Butoxyethanol at a concentration of 8
µg/m3 was detected in one of six samples of indoor air collected from
14 homes in northern Italy (ATSDR, 1996).
2-Butoxyethanol is present in a variety of consumer products,
including cleaning agents and surface coatings, such as paints,
lacquers, and varnishes. The average concentration of 2-butoxyethanol
in household products marketed in the USA in 1977 was 2.8%. Levels of
2-butoxyethanol in industrial and household window-cleaning agents
have been reported to range from 1% to 30% (v/v) (ATSDR, 1996). Based
upon available data, indirect exposure of the general population to
2-butoxyethanol is most likely via inhalation and dermal absorption
during the use of products containing this chemical.
Based on information from the National Occupational Exposure
Survey (NIOSH, 1983), the number of workers potentially exposed to
2-butoxyethanol in the workplace in the USA during 1981-1983 was
estimated at about 1.7 million, although it has probably increased
since then. Data on the occurrence of airborne 2-butoxyethanol in the
workplace obtained from facilities in the USA indicate that, in
general, most mean time-weighted-average exposures are below 7 ppm
(33.8 mg/m3) (NIOSH, 1990; ATSDR, 1996). Time-weighted average
2-butoxyethanol exposures have ranged from 1.1 to 5.4 ppm (5.3-26.1
mg/m3), with an average of 3.5 ppm (16.9 mg/m3), for silk screening;
average exposures of 6.8 ppm (32.8 mg/m3) for silk screeners, 2.6 ppm
(12.6 mg/m3) for silk screen spray painters, and 1.8 ppm (8.7 mg/m3)
for printing have also been reported (NIOSH, 1990; ATSDR, 1996). In a
study of various industrial operations, geometric mean atmospheric
exposures to 2-butoxyethanol ranged from 1.5 to 17.7 mg/m3 for
printing, from 3.4 to 93.6 mg/m3 for painting, and from 0.2 to 1774
mg/m3 in a mirror manufacturing plant (Veulemans et al., 1987).
Workers employed in varnish production facilities have been reported
to have individual exposures ranging from <0.1 to 8.1 ppm
(<0.5-39.1 mg/m3) (Angerer et al., 1990; Sohnlein et al., 1993). In
a study of automobile cleaners using products containing
2-butoxyethanol, time-weighted-average personal exposures ranged from
<0.1 to 7.33 ppm (<0.5-35.4 mg/m3) (Vincent et al., 1993).
7. COMPARATIVE KINETICS AND METABOLISM IN LABORATORY ANIMALS AND
HUMANS
Results of animal and human studies (most of the available data
are from studies conducted with rats) indicate that 2-butoxyethanol is
readily absorbed following inhalation, oral, and dermal exposure and
is oxidized to 2-butoxyacetic acid (Jonsson & Steen, 1978).
2-Butoxyethanol is metabolized primarily via alcohol and aldehyde
dehydrogenases, with the formation of 2-butoxyacetaldehyde and
2-butoxyacetic acid, the principal metabolite (Ghanayem et al., 1987b;
Medinsky et al., 1990). This is the favoured metabolic pathway for
lower systemic doses of 2-butoxyethanol. Alternative pathways include
O-dealkylation to ethylene glycol and conjugation to 2-butoxyethanol
glucuronide and/or 2-butoxyethanol sulfate (Medinsky et al., 1990).
In the study conducted by Medinsky et al. (1990), higher relative
concentrations of 2-butoxyacetic acid and ethylene glycol were
obtained at lower vapour concentrations of 2-butoxyethanol; higher
2-butoxyethanol glucuronide levels were observed at the high exposures
to 2-butoxyethanol, possibly owing to saturation of the pathways
leading to the formation of 2-butoxyacetic acid and ethylene glycol.
In human but not animal studies, the amino acid conjugate of
2-butoxyethanol, N-butoxyacetylglutamine, has been identified as a
metabolite (Rettenmeier et al., 1993).
In general, the metabolism of 2-butoxyethanol to 2-butoxyacetic
acid is linearly related to exposure concentration up to levels
causing mortality. In one study, after inhalation exposure in rats,
2-butoxyethanol and 2-butoxyacetic acid were analysed in blood,
muscle, liver, and testes. The kinetic profile of 2-butoxyacetic acid
tissue concentrations was similar to that of 2-butoxyethanol tissue
concentrations. Sixty-four per cent of the inhaled dose of
2-butoxyacetic acid was eliminated in urine, and the rate of urinary
excretion of 2-butoxyacetic acid was dose-dependent (Johanson, 1994).
In humans exposed to 20 ppm (96.6 mg/m3) 2-butoxyethanol for 2 h
via inhalation, the concentration of 2-butoxyethanol in the blood
reached a plateau of 7.4 µmol/litre within 1-2 h, and the chemical
could no longer be detected in the blood 2-4 h after exposure. The
mean elimination half-time was 40 min. Less than 0.03% of the total
uptake of 2-butoxyethanol was excreted in the urine, whereas urinary
excretion as 2-butoxyacetic acid ranged from 17% to 55% (Johanson et
al., 1986). Similarly, after percutaneous uptake of 2-butoxyethanol,
the urinary excretion of 2-butoxyacetic acid peaked 3 h after exposure
and subsequently declined, with an average half-life of 3.1 h. The
accumulated excretion of 2-butoxyacetic acid ranged from 8.7 to 313
µmol, corresponding to 2.5-39% of uptake (Johanson et al., 1988).
Several physiologically based pharmacokinetic (PBPK) models of
2-butoxyethanol absorption, metabolism, disposition, and excretion
have been developed. One model examined human inhalation exposures
during rest and exercise (Johanson et al., 1986; Johanson & Boman,
1991), whereas another addressed high-to-low-dose extrapolation and
route of administration extrapolation based on animal data (Shyr et
al., 1993). In the Shyr et al. (1993) model, 2-butoxyethanol is
metabolized to 2-butoxyacetic acid and ethylene glycol. An additional
model combined aspects of the preceding models and addressed the
disposition of 2-butoxyacetic acid in rats and humans (Corley et al.,
1994). The Corley et al. (1994) PBPK model describes the uptake,
distribution, metabolism, and elimination of 2-butoxyethanol and its
metabolite 2-butoxyacetic acid. It was developed by expanding a
previous inhalation model for 2-butoxyethanol (Johanson et al., 1986)
and is composed of two separate models for 2-butoxyethanol and
2-butoxyacetic acid that are joined through metabolism in the liver.
Both the 2-butoxyethanol and 2-butoxyacetic acid models have the same
eight compartments, with an additional kidney compartment in the
2-butoxyacetic acid model. Unlike the original model of Johanson et
al. (1986), the muscle and skin compartments have been separated.
Corley et al. (1994) also incorporated protein binding and saturable
elimination of 2-butoxyacetic acid by the kidneys. Equations for
additional routes of exposure (oral, dermal, and intravenous infusion)
were also added. Physiological and biochemical parameters were
allometrically scaled rather than using standard values for a 70-kg
human. This allows simulations to be conducted for specific data
sets. A rat version of the model was also developed.
The Corley et al. (1994) model accurately predicted animal data
at dose levels that did not cause haemolysis, the principal effect
exerted by 2-butoxyethanol (see below). At dose levels causing
haemolysis, the model overpredicted the amount of 2-butoxyacetic acid
excreted in the urine. This overprediction is assumed to be caused by
toxicity in the kidneys that is secondary to haemolysis. The model
does not accommodate toxicity in the kidneys and assumes that the
kidneys will continue to function as normal, thereby leading to the
overprediction of 2-butoxyacetic acid levels in the urine. The
results of the Johanson & Boman (1991) study indicated that during
whole-body exposure to 2-butoxyethanol vapour, dermal uptake accounted
for approximately 75% of the total uptake of the chemical. The Corley
et al. (1994) model was able to accurately predict the Johanson &
Boman (1991) human whole-body exposure blood data when it was assumed
that the sampled blood did not represent systemic venous blood but
instead represented venous blood draining from the skin compartment.
This blood had not yet been diluted by the venous blood pool. Corley
et al. (1994, 1997) suggested that the blood samples collected by
Johanson & Boman (1991) were not representative of systemic blood
concentrations and that dermal uptake is approximately 21% of the
total, rather than the 75% suggested by Johanson & Boman (1991). An
additional study further addresses dermal uptake in humans from the
vapour phase but does not address direct skin contact with liquid
containing 2-butoxyethanol (Corley et al., 1997).
8. EFFECTS ON LABORATORY MAMMALS AND IN Vitro TEST SYSTEMS
8.1 Single exposure
Many acute toxicity studies of 2-butoxyethanol have led to the
establishment of LC50s or LD50s in a variety of species by
inhalation, oral, and dermal exposure. Inhalation LC50s for
2-butoxyethanol of 486 ppm (2347 mg/m3) (male rats, 4 h), 450 ppm
(2174 mg/m3) (female rats, 4 h), 700 ppm (3381 mg/m3) (mice, 7 h),
and >650 ppm (3140 mg/m3) (guinea-pigs, 1 h) have been reported.
Oral LD50s for rats (2500 mg/kg body weight), mice (1400 mg/kg body
weight), guinea-pigs (1200 mg/kg body weight), and rabbits (320 mg/kg
body weight) have also been reported. Dermal LD50s of 404-502 and
2000 mg/kg body weight have been reported for rabbits and guinea-pigs,
respectively. Effects observed in rats, mice, and guinea-pigs exposed
by inhalation to the LC50 or by ingestion to the LD50 include loss of
coordination, ataxia, sluggishness, muscular flaccidity, enlarged
kidney, blood in the bladder, haemoglobinuria, splenic lesions, and
pulmonary congestion (Werner et al., 1943a; Carpenter et al., 1956;
Dodd et al., 1983; Gingell et al., 1997). Inhalation exposures of
female rats to 62 ppm (299 mg/m3) 2-butoxyethanol for 4-h periods
resulted in increased osmotic fragility of erythrocytes (Carpenter et
al., 1956).
Ghanayem et al. (1987a) indicated that the haemolytic activity of
2-butoxyethanol in rats is age-dependent, with older rats being more
susceptible than younger animals. In their study, 2-butoxyethanol (0,
125, or 500 mg/kg body weight) was administered orally to young (4- to
5-week-old) and adult (9- to 13-week-old) male F344 rats, and
haematotoxicity was assessed from 2 to 48 h later. A decrease in red
blood cells, haemoglobin, and haematocrit was accompanied by a
significant ( p < 0.05) dose-dependent increase in free haemoglobin
plasma levels in both age groups administered 500 mg
2-butoxyethanol/kg body weight; in both groups, there was a gradual
recovery after 48 h. Although no significant haematotoxic effects
were observed in the younger rats administered 125 mg
2-butoxyethanol/kg body weight, effects in older animals administered
this dose included a significant decrease ( p < 0.05) in the
number of red blood cells, haematocrit, and haemoglobin. Free
haemoglobin plasma levels were significantly increased ( p < 0.05)
in adult rats 8 h after oral administration of 125 mg/kg body weight;
there was no effect on free haemoglobin plasma levels in young
animals. Histopathological evaluation of tissues collected 24 h after
2-butoxyethanol administration to rats of various ages revealed dose-
and age-dependent liver and kidney changes. These histopathological
changes exhibited signs of regression when examined 48 h following
exposure. Severe acute haemolytic anaemia was evidenced by a decrease
in circulating red blood cells, an increase in the concentration of
free haemoglobin in plasma, and the development of haemoglobinuria.
Using a laser-based haematology analyser, Ghanayem et al. (1987a)
indicated that the acute haemolysis in 2-butoxyethanol-exposed rats
was caused by a time- and dose-dependent decrease in the number of red
blood cells, in haemoglobin concentrations, and in haematocrit, with
little or no change in mean cell volume. In a follow-up study in
which the authors used both a laser-based haematology analyser and an
impedance-based analyser, haematology profiles from the
impedance-based haematology analyser revealed a time- and
dose-dependent increase in haematocrit and mean cell volume; the
laser-based analyser was unable to detect early increases in
haematocrit and mean cell volume in the exposed animals. Based on
these data, Ghanayem et al. (1990) concluded that 2-butoxyethanol
causes spherical swelling of red blood cells followed by haemolysis.
To investigate the induction of tolerance, Ghanayem et al. (1992)
assessed haematological parameters in naive or previously bled rats
administered a single dose of 125 or 250 mg 2-butoxyethanol/kg body
weight. The bled/recovered rats were less sensitive to
2-butoxyethanol than the naive animals. In vitro incubations with
2-butoxyacetic acid revealed that red blood cells from the
bled/recovered rats were less sensitive than those cells from naive
animals. Ghanayem et al. (1992) concluded that young red blood cells
formed during the regeneration process were less sensitive to
2-butoxyacetic acid than older red blood cells. Chronic exposure to
2-butoxyethanol would be expected to result in tolerance to
2-butoxyethanol-induced haemolytic anaemia. The mechanism is probably
related to the greater susceptibility of older cells to 2-butoxyacetic
acid; haemolysis of these cells during the initial exposure followed
by their replacement with less susceptible younger cells may account
for the development of tolerance.
Toxic effects in the kidneys have been observed in rabbits
exposed percutaneously to 2-butoxyethanol (Carpenter et al., 1956).
Necropsy of rabbits exposed for 24 h to undiluted 2-butoxyethanol
(0.48-0.64 ml/kg body weight) revealed congestion of the kidneys,
haemoglobinuria, pale livers, and engorged spleens (Carpenter et al.,
1956).
When 2-butoxyethanol (200, 260, 320, 375, or 500 mg/kg body
weight) was applied to the shaved dorsal skin of groups of female
rats, increased mean cell volume, a lowered erythrocyte count and
haemoglobin level, and haemoglobinuria were observed within 6 h of
exposure to the highest dose; no haemolytic effects were observed at
the lowest dose tested (Bartnik et al., 1987). 2-Butoxyethanol at
doses of 260, 320, and 375 mg/kg body weight produced similar effects
in at least some animals in each group; however, there was no
discernible dose-response relationship, which was attributed to the
inherent biological variation in percutaneous absorption and
haemolytic susceptibility and to the small number of animals ( n = 3)
in these dose groups.
8.2 Irritation and sensitization
2-Butoxyethanol is irritating to the eyes and skin. In rabbits,
instillation of an unspecified amount of undiluted 2-butoxyethanol
caused severe eye irritation, including conjunctival hyperaemia and
oedema (von Oettingen & Jirouche, 1931). More recent ocular tests in
rabbits revealed that 30% and 70% concentrations of 2-butoxyethanol
were moderately irritating (Kennah et al., 1989). When applied to the
skin of rabbits for 4 h, 2-butoxyethanol caused mild irritation;
extending the period of contact increased the severity of irritation
(Tyler, 1984). 2-Butoxyethanol was classified as a severe cutaneous
irritant when the Draize method was used (Zissu, 1995).
2-Butoxyethanol did not induce skin sensitization in guinea-pigs
(Unilever, 1989, as cited in ECETOC, 1994; Zissu, 1995).
8.3 Short-term exposure
In older studies, haematotoxic effects (e.g. increased osmotic
fragility, decreased haemoglobin, decreased numbers of red blood
cells) have been observed in rats (54-320 ppm; 261-1546 mg/m3), dogs
(200-385 ppm; 966-1860 mg/m3), and monkeys (210 ppm; 1014 mg/m3)
exposed repeatedly via inhalation to 2-butoxyethanol for up to
approximately 30-35 days (Werner et al., 1943b; Carpenter et al.,
1956).
Dodd et al. (1983) exposed Fischer 344 rats of both sexes to 0,
20, 86, or 245 ppm (0, 97, 415, or 1183 mg/m3) 2-butoxyethanol, 6
h/day for 9 days in total (5 consecutive days of exposure, followed by
2 days of no exposure, then 4 additional consecutive days of
exposure). In both sexes, exposure to 245 ppm (1183 mg/m3) was
associated with a significant reduction in red blood cell counts
( p < 0.001), haemoglobin levels ( p < 0.001), and mean cell
haemoglobin concentration ( p < 0.01), as well as a significant
increase ( p < 0.001 in all cases) in mean cell volume, nucleated
red blood cells, and reticulocytes. Fourteen days post-exposure, a
substantial recovery of the affected erythroid parameters was
observed; however, statistically significant differences from controls
were still observed for the males (i.e. red blood cell count
[ p < 0.01], mean cell volume [ p < 0.001], and mean cell
haemoglobin [ p < 0.001]). Exposure of both sexes to 86 ppm (415
mg/m3) 2-butoxyethanol was associated with a significant but less
profound effect on erythroid parameters. The no-observed-adverse-
effect level (NOAEL) in this study is 20 ppm (97 mg/m3).
In a study designed primarily to assess developmental effects,
Tyl et al. (1984) exposed pregnant Fischer 344 rats (36 per group) and
New Zealand white rabbits (24 per group) to 2-butoxyethanol (0, 25,
50, 100, or 200 ppm; 0, 121, 242, 483, or 966 mg/m3) for 6 h/day on
days 6-15 of gestation for the rats and on days 6-18 of gestation for
the rabbits. In rats, there were significant reductions in red blood
cell count and significant increases in haemoglobin and haematocrit at
200 ppm (966 mg/m3) ( p < 0.001); the red blood cell count was also
reduced at 100 ppm (483 mg/m3) ( p < 0.001). In dams exposed to
100 or 200 ppm (483 or 966 mg/m3) 2-butoxyethanol, mean cell volume
and mean cell haemoglobin were significantly increased relative to
controls; the mean cell haemoglobin concentration was significantly
reduced at 100 ppm (483 mg/m3) 2-butoxyethanol ( p < 0.01) and 200
ppm (966 mg/m3) 2-butoxyethanol ( p < 0.001), relative to controls.
In the rabbits, statistically significant increases in haemoglobin
content and haematocrit were observed at 100 ppm (483 mg/m3)
( p < 0.01) but not at 200 ppm (966 mg/m3) 2-butoxyethanol. The
results of this study indicate that rats are more sensitive than
rabbits to the haemolytic effects of 2-butoxyethanol. The NOAEL in
this study is 50 ppm (242 mg/m3) 2-butoxyethanol.
The oral administration of 500 or 1000 mg 2-butoxyethanol/kg body
weight per day for 4 consecutive days to male F344 rats produced a
pronounced dose-dependent effect on circulating red and white blood
cells (Grant et al., 1985); however, some effects were reversible
following the end of exposure. Reduced erythrocyte counts,
haematocrit, haemoglobin levels, and leukocyte counts and elevated
mean cell volume, reticulocyte counts, and mean cell haemoglobin
concentration ( p < 0.001) were observed in animals in the high-dose
group. Similar, although less severe, effects were observed in the
low-dose group.
To assess the development of tolerance to the haemolytic effects
of 2-butoxyethanol exposure in laboratory animals, male F344 rats were
administered (by gavage) 125 mg 2-butoxyethanol/kg body weight per day
for 0, 1, 2, 3, 6, and 12 days, and haematological parameters (red
blood cell counts, haemoglobin content, haematocrit) were determined
after exposure (Ghanayem et al., 1987a). Administration of
2-butoxyethanol for 2 and 3 days caused significant haemolysis of red
blood cells, although after the third day there was a gradual increase
in the number of red blood cells and haemoglobin content. After 12
days, red blood cells and haemoglobin approached pre-exposure levels,
indicative of the development of tolerance to the haemolytic effects
of 2-butoxyethanol. In a follow-up study, Ghanayem et al. (1992)
assessed the haemolytic effects of 2-butoxyethanol (administered as a
single dose of 0, 125, or 250 mg/kg body weight) in untreated
(control) or 2-butoxyethanol-pretreated male F344 rats. The
pretreated animals were administered (by gavage) 125 mg
2-butoxyethanol/kg body weight per day for 3 days and then allowed to
recover for 7 days prior to study. The pretreated animals were less
sensitive to the haemolytic effects of subsequent exposure to
2-butoxyethanol than the untreated controls. In vitro incubations
with 2-butoxyacetic acid revealed that red blood cells from the
2-butoxyethanol-pretreated group were less sensitive than cells from
the untreated controls. The authors suggested that the development of
tolerance to the haemolytic effects of 2-butoxyethanol might be due in
part to the reduced sensitivity of young erythrocytes formed during
the blood regeneration process.
In mice orally administered 500 or 1000 mg 2-butoxyethanol/kg
body weight per day, 5 days/week for 5 weeks, no effect upon white
blood cell counts, mean cell volume, or haemoglobin levels was
observed; however, red blood cell counts were reduced at both doses
(Nagano et al., 1979). The oral administration of 222, 443, or 885 mg
2-butoxyethanol/kg body weight per day, 5 days/week for 6 weeks, to
male rats principally affected red blood cells, whereas white blood
cell counts were unaffected (Krasavage, 1986).
In a study in which F344/N rats and B6C3F1 mice were
administered 2-butoxyethanol in drinking-water daily for 2 weeks,
estimates of 2-butoxyethanol intake by rats and mice ranged from 70 to
300 mg/kg body weight per day and from 90 to 1400 mg/kg body weight
per day, respectively (NTP, 1993). Survival of both species was not
affected by exposure to 2-butoxyethanol. Statistically significant
decreases ( p < 0.05) in relative and absolute thymus weights were
noted in male mice receiving 400 or 650 mg 2-butoxyethanol/kg body
weight per day. No haematological tests were conducted in this study.
8.4 Long-term exposure
8.4.1 Subchronic exposure
In older studies, haematotoxic effects (e.g. increased osmotic
fragility, decreased haemoglobin, decreased red blood cell numbers)
have been observed in mice (100-400 ppm; 483-1932 mg/m3), dogs (415
ppm; 2004 mg/m3), and monkeys (100 ppm; 483 mg/m3) exposed
repeatedly by inhalation to 2-butoxyethanol for up to approximately 90
days (Werner et al., 1943c; Carpenter et al., 1956). More recent
studies on effects associated with the subchronic exposure of
laboratory animals to 2-butoxyethanol are limited.
Dodd et al. (1983) exposed Fischer 344 rats of both sexes (16 per
group) to 0, 5, 25, or 77 ppm (0, 24, 121, or 372 mg/m3)
2-butoxyethanol by inhalation, 6 h/day, 5 days/week, for 13 weeks.
After 6 weeks, animals exposed to 77 ppm (372 mg/m3) 2-butoxyethanol
had a slight but statistically significant decrease in red blood cell
counts ( p < 0.01) and haemoglobin level (statistics not reported),
accompanied by an 11% increase in mean cell haemoglobin concentration
( p < 0.001). At the end of the study, these effects had either
lessened or returned to the ranges of control values. The only
significant haemolytic effect for male rats in the 77 ppm (372 mg/m3)
2-butoxyethanol exposure group was a 5% decrease in red blood cell
count after 66 exposures to 2-butoxyethanol (statistics not provided).
The NOAEL in this study is 25 ppm (121 mg/m3).
Groups of F344/N rats and B6C3F1 mice (10 per sex per
concentration) were administered 2-butoxyethanol in drinking-water (0,
750, 1500, 3000, 4500, or 6000 mg/litre) daily for 13 weeks; estimated
intakes by rats and mice ranged from 70 to 500 mg/kg body weight per
day and from 100 to 1300 mg/kg body weight per day, respectively (NTP,
1993). Effects observed in both species included decreased body
weight gain and water consumption. In rats, reduced red blood cell
counts and histopathological lesions in the liver, spleen, and bone
marrow were observed in males and females (at concentrations of
3000-6000 mg/litre and 750-6000 mg/litre, respectively). Reduced
thymus weights (at concentrations of 4500 and 6000 mg/litre in males
and females, respectively), diminished uterine size (at 4500 and 6000
mg/litre in females), and diminished sperm concentration (750-6000
mg/litre in males) were also noted. A NOAEL could not be identified
owing to a mild to moderate anaemia present in most dose groups of
rats. In mice, the only effect observed was reduced body weight gain
in males and females at concentrations of 3000-6000 mg/litre.
8.4.2 Chronic exposure and carcinogenicity
Published information on effects associated with the chronic
exposure of laboratory animals to 2-butoxyethanol was not
identified.1
8.5 Genotoxicity and related end-points
2-Butoxyethanol has been tested for genotoxicity in a range of
in vitro and in vivo assays (see Elliott & Ashby, 1997, for a
recent review). In standard tests in bacteria, 2-butoxyethanol was
not mutagenic in Salmonella typhimurium strains TA1535, TA1537,
TA97, TA98, TA100, and TA102 (Zeiger et al., 1992; Hoflack et al.,
1995; Gollapudi et al., 1996). However, the results for strain TA98a
were inconsistent, with one report of mutagenicity observed in both
the presence and absence of metabolic activation (Hoflack et al.,
1995) and another report of no mutagenicity (Gollapudi et al., 1996).
2-Butoxyethanol was not mutagenic at the HPRT locus in Chinese
hamster ovary cells in either the presence or absence of metabolic
activation (McGregor, 1984; Chiewchanwit & Au, 1995). However, there
was evidence that it caused gene mutations at the HPRT locus in
Chinese hamster lung (V79) cells (Elias et al., 1996). An in vitro
assay for unscheduled DNA synthesis in rat hepatocytes yielded
equivocal results (Elliott & Ashby, 1997). 2-Butoxyethanol produced
sister chromatid exchanges in human peripheral lymphocytes but not in
Chinese hamster lung (V79) or ovary cells. In vitro cytogenetic
assays conducted with human peripheral lymphocytes, Chinese hamster
lung (V79) cells, and Chinese hamster ovary cells revealed no
induction of chromosomal aberrations. An in vitro micronucleus
assay in Chinese hamster lung (V79) cells, which incorporated a test
for aneuploidy, yielded equivocal results (Elliott & Ashby, 1997).
In vivo mutagenicity tests have yielded uniformly negative
results for 2-butoxyethanol. These assays have included three bone
marrow micronucleus tests utilizing intraperitoneal injection in rats
and mice (Elias et al., 1996; Elliott & Ashby, 1997); a
[32P]post-labelling assay for DNA adducts in the brain, kidney,
liver, spleen, and testes of orally dosed rats (Keith et al., 1996);
an assay for DNA methylation in the brain, kidney, liver, spleen, and
testes of rats and in FVB/N transgenic mice carrying the v-Ha- ras
1 Results of a US National Toxicology Program 2-year carcinogenesis
bioassay completed in July 1995 were not available at the time this
CICAD was prepared.
oncogene (Keith et al., 1996); as well as a test for tumour formation
in FVB/N transgenic mice (Keith et al., 1996). Although the results
of in vitro tests for mutagenicity of 2-butoxyethanol are
inconsistent, the absence of structural alerts as well as the negative
results from in vivo studies are sufficiently reassuring to allow
the conclusion that 2-butoxyethanol is not mutagenic.
Mutagenicity studies have also been performed on two metabolites
of 2-butoxyethanol -- 2-butoxyacetic acid and 2-butoxyacetaldehyde.
2-Butoxyacetic acid was not mutagenic in a series of in vitro
assays, in addition to an in vivo micronucleus assay in mice
administered the chemical by intraperitoneal injection (Hoflack et
al., 1995; Elias et al., 1996; Elliott & Ashby, 1997).
2-Butoxyacetaldehyde exhibited mutagenic potential in several
in vitro studies (including tests for HPRT gene mutation,
chromosomal aberrations, micronuclei, aneuploidy, and sister chromatid
exchange); however, in the absence of data from in vivo studies, it
is not possible to reach a final conclusion concerning the possible
mutagenic hazard of this metabolite (Chiewchanwit & Au, 1995; Hoflack
et al., 1995; Elias et al., 1996; Elliott & Ashby, 1997).
8.6 Reproductive and developmental toxicity
Effects on the testes were not observed in studies in which
Alpk/Ap (Wistar-derived) rats were exposed by inhalation to 800 ppm
(3864 mg/m3) 2-butoxyethanol for 3 h (Doe, 1984), JCL-ICR mice were
orally administered 2-butoxyethanol at doses ranging from 500 to 2000
mg/kg body weight per day, 5 days/week, for 5 weeks (Nagano et al.,
1979), or rats were administered 2-butoxyethanol (by gavage) at doses
ranging from 222 to 885 mg/kg body weight per day, 5 days/week, for 6
weeks (Krasavage, 1986). Testicular damage was not observed in groups
of Alpk/Ap (Wistar-derived) rats administered a single oral dose of
174, 434, or 868 mg 2-butoxyacetic acid/kg body weight (Foster et al.,
1987).
No adverse effects were observed in either the dams or pups
(number of resorptions, fetal weights, and incidence of malformations)
in a study in which Sprague-Dawley rats were exposed by inhalation for
7 h/day on days 7-15 of gestation to 150 or 200 ppm (725 or 966
mg/m3) 2-butoxyethanol (Nelson et al., 1984); exposure to 250 or 500
ppm (1208 or 2415 mg/m3) 2-butoxyethanol caused death in the dams.
Tyl et al. (1984) exposed pregnant Fischer 344 rats (36 per
group) and New Zealand white rabbits (24 per group) to 0, 25, 50, 100,
or 200 ppm (0, 121, 242, 483, or 966 mg/m3) 2-butoxyethanol for 6
h/day on days 6-15 of gestation for the rats and on days 6-18 of
gestation for the rabbits. No adverse reproductive or developmental
effects were observed in rats or rabbits exposed to 25 ppm or 50 ppm
(121 or 242 mg/m3) 2-butoxyethanol. In rats, exposure to 200 ppm
(966 mg/m3) 2-butoxyethanol was associated with a reduction in
maternal weight gain, a significant ( p < 0.01) increase in the
number of totally resorbed litters, and a reduction in the number of
viable implants ( p < 0.001) and in the percentage of live fetuses
( p < 0.01) per litter. However, there were no statistically
significant increases in incidences of external, visceral, skeletal,
or total malformations associated with exposure to 2-butoxyethanol.
Exposure to 200 ppm (966 mg/m3) 2-butoxyethanol was also associated
with a significant increase ( p < 0.05) in the number of litters
with one or more fetuses with unossified skeletal elements and poorly
ossified skeletal elements. There was a decreased incidence of
bilobed cervical centrum 5, bilobed thoracic centra 9 and 13, as well
as poorly ossified proximal phalanges of the hindlimb. Following
maternal exposure to 100 ppm (483 mg/m3) 2-butoxyethanol, skeletal
ossification in the fetuses was retarded, with a significant
( p < 0.05) decreased (primarily because at these exposure
concentrations this skeletal element was largely unossified) incidence
of bilobed cervical centrum 5 and an increased incidence ( p < 0.05)
of unossified cervical centrum 6. In rabbits, exposure to 200 ppm
(966 mg/m3) 2-butoxyethanol produced a significant reduction in
maternal body weight, gravid uterine weight, and numbers of total
implants and viable implants. No significant increases in the number
of fetuses or litters with malformations were observed in any
treatment group; however, exposure to 200 ppm (966 mg/m3)
2-butoxyethanol was associated with a significant ( p < 0.05)
reduction in unossified sternebra 6 and in rudimentary rib at the
first lumbar rib. The occurrence of unossified skeletal elements in
both rats and rabbits was an indication of delayed development in rats
and rabbits exposed to 2-butoxyethanol under maternally toxic
conditions (Tyl et al., 1984).
Maternal deaths and a reduction in the number of viable litters
were observed in a study in which CD-1 mice were orally administered
4000 mg 2-butoxyethanol/kg body weight per day on days 7-14 of
gestation (Schuler et al., 1984).
Heindel et al. (1990) used a continuous breeding protocol
(Heindel et al., 1989) to assess the reproductive toxicity of
2-butoxyethanol. Male and female Swiss CD-1 mice were administered
2-butoxyethanol in drinking-water (0, 0.5, 1.0, or 2.0%; equivalent to
0, 0.7, 1.3, and 2.1 g/kg body weight per day) 7 days prior to and
during a 98-day cohabitation period (20 pairs of mice per dose).
Exposure to 1.0% or 2.0% 2-butoxyethanol in drinking-water was
associated with increased mortality in the females and a significant
reduction ( p < 0.05) in the number of live pups per litter, the
proportion of pups born alive, and the live pup weights (both absolute
and adjusted). The authors noted that these effects occurred in the
presence of maternal toxicity, as evidenced by decreased body weight,
decreased water consumption, and increased kidney weight in the female
mice. Necropsy revealed that testes and epididymis weights were
normal, as were sperm number and motility. The reproductive toxicity
of 2-butoxyethanol was evident only in female mice, at doses that also
elicited general toxicity (Heindel et al., 1990).
No maternal, embryotoxic, fetotoxic, or teratogenic effects were
detected when 2-butoxyethanol (106 mg) was applied to the shaved
interscapular skin of female Sprague-Dawley rats, four times daily on
days 7-14 of gestation (Hardin et al., 1984).
8.7 Immunological and neurological effects
Effects on the immune system were examined in two studies in
which 2-butoxyethanol was administered orally, by drinking-water or
gavage. In the first study, Sprague-Dawley rats were administered
2-butoxyethanol at 0, 2000, or 6000 mg/litre (males) or 0, 1600, or
4800 mg/litre (females) in drinking-water for 21 consecutive days.
Exposure to 2-butoxyethanol had no effect on antibody production,
delayed-type hypersensitivity reactions, and interferon or
interleukin-2 production. However, natural killer cell cytotoxicity
responses were enhanced ( p < 0.05) in rats receiving the lowest
concentrations of 2-butoxyethanol (Exon et al., 1991). In the second
study, male Fischer rats were administered (by gavage) 0, 50, 100,
200, or 400 mg 2-butoxyethanol/kg body weight per day for 2
consecutive days, following immunization with
trinitrophenyl-lipopolysaccharide. A reduction ( p < 0.05) in the
serum haemagglutination titre was observed 3 days later in rats
administered 200 mg 2-butoxyethanol/kg body weight per day. All
animals in the highest dose group died (Smialowicz et al., 1992).
No specific investigations on potential neurological effects
associated with exposure to 2-butoxyethanol were identified. However,
adverse effects on the central nervous system associated with exposure
to 2-butoxyethanol have been observed. These included loss of
coordination, sluggishness, narcosis, muscular flaccidity, and ataxia
(Carpenter et al., 1956; Dodd et al., 1983; Hardin et al., 1984;
Krasavage, 1986).
8.8 In vitro haemolytic effects
Bartnik et al. (1987) examined the effects of 2-butoxyethanol and
2-butoxyacetic acid on human (from healthy males) and rat (four male
Wistar) erythrocytes in vitro. Under these conditions 175, 200, 225,
and 250 mmol 2-butoxyethanol/litre induced complete lysis of rat
erythrocytes, whereas only 200, 225, and 250 mmol
2-butoxyethanol/litre induced complete lysis of human erythrocytes.
Although 3.75-7.5 mmol 2-butoxyacetic acid/litre caused complete lysis
of rat erythrocytes, lysis of human erythrocytes was not observed at
these concentrations. These results indicate that rats may be more
susceptible than humans to the haemolytic effects of 2-butoxyethanol
and its metabolite 2-butoxyacetic acid (Bartnik et al., 1987).
Ghanayem (1989) examined the effect of 2-butoxyethanol and
2-butoxyacetic acid on blood collected by cardiac puncture from male
F344 rats. The addition of 2-butoxyethanol to whole blood to
concentrations of 5 or 10 mmol/litre had no effect on haematocrit,
whereas a concentration of 20 mmol/litre caused significant haemolysis
( p < 0.05). The addition of 2-butoxyacetic acid to rat
erythrocytes to concentrations of 0.5 or 1 mmol/litre caused a time-
and concentration-dependent increase in haematocrit followed by
haemolysis. Incubation with 2 mmol 2-butoxyacetic acid/litre caused a
faster time-dependent increase in haematocrit, with the haematocrit
reaching a maximum after 2 h, followed by nearly complete haemolysis
after 4 h. Also examined was the effect of 2-butoxyacetic acid (0.5,
1, 2, 4, or 8 mmol/litre) on human blood obtained from healthy young
male and female volunteers (Ghanayem, 1989). No significant changes
in haematocrit or haemolysis were observed at 2-butoxyacetic acid
concentrations of 4 mmol/litre or less; at 8 mmol 2-butoxyacetic
acid/litre, there was a slight but significant increase in haematocrit
( p < 0.05), followed by a slight but significant haemolysis
( p < 0.05) of erythrocytes.
In a subsequent study, Ghanayem & Sullivan (1993) assessed the
haemolytic activity of 2-butoxyacetic acid (1 or 2 mmol/litre) in
blood collected from a variety of species (i.e. rats, mice, hamsters,
baboons, rabbits, pigs, guinea-pigs, dogs, cats, and humans).
2-Butoxyacetic acid caused a time- and concentration-dependent
increase in mean cell volume and haematocrit of blood from rats,
rabbits, hamsters, mice, and baboons. However, no or minimal effects
were observed on blood from humans, guinea-pigs, dogs, cats, and pigs
(Ghanayem & Sullivan, 1993), demonstrating the sensitivity of rat
erythrocytes and the relative insensitivity of human erythrocytes to
the haemolytic effects of 2-butoxyacetic acid.
The effect of 2-butoxyacetic acid on red blood cells from healthy
young and older individuals (Udden & Patton, 1994) and individuals
with a possible susceptibility to 2-butoxyethanol-induced haemolysis
(i.e. sickle cell and spherocytosis patients) (Udden, 1994, 1996) has
also been examined. Along with haemolysis, 0.2 and 2 mmol
2-butoxyacetic acid/litre caused decreased red blood cell
deformability and increased mean cellular volume in rat red blood
cells (Udden & Patton, 1994). However, none of the human blood
samples exhibited prehaemolytic changes (i.e. decreased deformability
and increased mean cellular volume) or haemolysis after treatment with
2 mmol 2-butoxyacetic acid/litre for up to 4 h (Udden, 1994, 1996;
Udden & Patton, 1994). The results of these in vitro studies
provide further evidence that rat erythrocytes are more susceptible
than human erythrocytes to 2-butoxyacetic acid-induced haemolysis.
9. EFFECTS ON HUMANS
Information on human health effects associated with exposure to
2-butoxyethanol are limited to a few case reports and one laboratory
investigation; epidemiological studies have not been identified. The
principal human health effects attributed to 2-butoxyethanol exposure
have involved the central nervous system, the blood, and the kidneys
(ATSDR, 1996).
In one report involving a number of small studies, the exposure
of two males to 113 ppm (546 mg/m3) 2-butoxyethanol for 4 h produced
nose and eye irritation as well as disturbed taste, but there was no
evidence of haemolytic effects. Similar effects were observed in a
second study in which two males and one female were exposed to 195 ppm
(942 mg/m3) 2-butoxyethanol for two 4-h periods, separated by a
30-min period of no exposure. When two males and two females were
exposed to 100 ppm (483 mg/m3) 2-butoxyethanol for 8 h, the effects
included vomiting and headache. No clinical signs of haemolysis were
observed in any of the subjects; however, following exposure to 195
ppm (942 mg/m3) 2-butoxyethanol, increased osmotic fragility of
erythrocytes was observed in vitro (Carpenter et al., 1956).
Haemoglobinuria, erythropenia, and hypotension (Rambourg-Schepens
et al., 1988; Gijsenbergh et al., 1989), metabolic acidosis, shock,
non-cardiogenic pulmonary oedema, and albuminuria (Bauer et al.,
1992), and metabolic acidosis, hepatic laboratory abnormalities, and
haematuria (Gualtieri et al., 1995) have been reported in case-studies
of individuals who had attempted suicide by ingesting 2-butoxyethanol-
containing cleaning solutions (involving an estimated ingestion of
25-60 g 2-butoxyethanol). In two of the cases, haemodialysis was
employed, and all patients recovered fully with appropriate treatment.
A survey of paediatric poisonings identified 24 children who had
ingested 5-300 ml of glass cleaners containing 2-butoxyethanol (Dean &
Krenzelok, 1992). The two children with the highest intake exhibited
no evidence of haemolytic effects. 2-Butoxyethanol is reportedly not
a skin sensitizer in humans (Greenspan et al., 1995).
10. EFFECTS ON OTHER ORGANISMS IN THE LABORATORY AND FIELD
10.1 Aquatic environment
Results of acute and long-term studies on toxicity to aquatic
organisms are summarized in Table 1. Long-term studies are restricted
to microorganisms and unicellular algae, for which 72 h is the cut-off
point for the designation of acute/long-term studies.
10.2 Terrestrial environment
Information on the toxicological effects of 2-butoxyethanol on
terrestrial organisms was not identified.
Table 1: Acute and long-term studies on toxicity to aquatic organisms.
Species End-pointa Concentration Reference
(mg/litre)
Freshwater
Bacterium (Pseudomonas putida) 16-h LOEC (growth) 700 Bringmann & Kuhn, 1980a
Sewage sludge bacteria 16-h IC50 >1000 Union Carbide, 1989
Protozoan (Entosiphon sulcatum) 72-h LOEC (growth) 91 Bringmann & Kuhn, 1980a
Protozoan (Chilomonas paramecium) 48-h EC5 (growth) 911 Bringmann & Kuhn, 1980b
Protozoan (Uronema parduczi) 48-h EC5 (growth) 463 Bringmann & Kuhn, 1980b
Cyanobacterium (Microcystis aeruginosa) 8-day LOEC (growth) 35 Bringmann & Kuhn, 1980a
Green alga (Scenedesmus quadricaudata) 7-day LOEC (growth) 900 Bringmann & Kuhn, 1980a
Green alga (Selenastrum capricornutum) 7-day NOEC 125 Dow, 1988
7-day EC50 >1000
Water flea (Daphnia magna) 24-h LC50 1720 Bringmann & Kuhn, 1977
24-h LC50 1698-1940 Bringmann & Kuhn, 1982
24-h LC50 5000 CMA, 1994
48-h LC50 835 Dow, 1979
Guppy (Poecilia reticulata) 7-day LC50 982 Koenemann, 1981
Golden ide (Leuciscus idus melanotus) 48-h LC50 165-186 Junke & Ludemann, 1978
48-h LC50 1880 CMA, 1994
Bluegill sunfish (Lepomis macrochirus) 96-h LC50 1490 Dawson et al., 1977
Goldfish (Carassius auratus) 24-h LC50 1700 Bridie, 1979
24-h LC50 1650 Verschueren, 1983
Fathead minnow (Pimephales promelas) 96-h LC50 2137 Dow, 1979
Emerald shiner (Notropus atherinoides) 72-h LC50 >500 Dill, 1995
Rainbow trout (Oncorhynchus mykiss) 96-h LC50 >1000 Environment Canada, 1997
Estuarine/marine
Oyster (Crassostrea virginica) 96-h LC50 89 US EPA, 1984
White shrimp (Penaeus setiferus) 96-h LC50 130 OECD, 1997
Grass shrimp (Palaemonetes pugio) 96-h LC50 5.4 Environment Canada, 1997
Brown shrimp (Crangon crangon) 48-h LC50 600-1000 Verschueren, 1983
96-h LC50 550-950
Table 1: (continued)
Species End-pointa Concentration Reference
(mg/litre)
Brine shrimp (Artemia salina) 24-h LC50 1000 Price et al., 1974
Inland silverside (Menidia beryllina) 96-h LC50 1250 Dawson et al., 1977
Sheepshead minnow (Cyprinodon variegatus) 96-h LC50 116 OECD, 1997
a NOEC = no-observed-effect concentration; LOEC = lowest-observed-effect concentration.
11. EFFECTS EVALUATION
11.1 Evaluation of health effects
11.1.1 Hazard identification and dose-response assessment
In general, effects associated with exposure to 2-butoxyethanol
have been identified from studies in animals. 2-Butoxyethanol has
moderate acute toxicity following inhalation, ingestion, or dermal
exposure. It is an eye and skin irritant, but it is not a skin
sensitizer. 2-Butoxyethanol is readily absorbed via inhalation,
dermal exposure, and ingestion. The pharmacokinetic models of Corley
et al. (1994, 1997) and Johanson & Boman (1991) consider dermal
absorption from the vapour phase to account for approximately 21-75%
of the total uptake. Pathways for the metabolism of 2-butoxyethanol
are similar in animals and humans; the principal metabolite is
2-butoxyacetic acid.
The principal effect exerted by 2-butoxyethanol and its
metabolite 2-butoxyacetic acid is haematotoxicity, with rats being the
most sensitive species. Older rats are more sensitive than younger
animals to the haemolytic effects of 2-butoxyethanol and
2-butoxyacetic acid. Critical effects observed in inhalation studies
conducted with rats were decreased haemoglobin and mean cell
haemoglobin; increased haematocrit and mean cell volume (NOAEL = 20
ppm [97 mg/m3]; lowest-observed-adverse-effect level [LOAEL] = 86 ppm
[415 mg/m3] in animals exposed for 9 days; Dodd et al., 1983);
decreased red blood cells and haemoglobin (NOAEL = 25 ppm [121
mg/m3]; LOAEL = 77 ppm [372 mg/m3]) in animals exposed
subchronically (Dodd et al., 1983); and decreased red blood cells and
increased mean cell volume (NOAEL = 50 ppm [242 mg/m3]; LOAEL = 100
ppm [483 mg/m3]) in pregnant animals exposed on days 6-15 of
gestation (Tyl et al., 1984). The results of in vitro studies
indicate that human red blood cells are not as sensitive as rat red
blood cells to the haemolytic effects of 2-butoxyethanol and
2-butoxyacetic acid and also that red blood cells are more sensitive
to haemolysis by 2-butoxyacetic acid than to haemolysis by
2-butoxyethanol (Bartnik et al., 1987; Ghanayem, 1989; Ghanayem &
Sullivan, 1993; Udden, 1994; Udden & Patton, 1994).
In rats, adverse effects on the central nervous system, kidneys,
and liver occur at higher exposure concentrations than do haemolytic
effects. 2-Butoxyethanol (and, in one study, 2-butoxyacetic acid) did
not cause adverse reproductive or developmental effects in either sex
at less than toxic doses (Nagano et al., 1979; Doe, 1984; Hardin et
al., 1984; Nelson et al., 1984; Tyl et al., 1984; Foster et al., 1987;
Heindel et al., 1990). Although sperm concentration was reduced in
rats administered drinking-water containing 2-butoxyethanol (NTP,
1993), the reduction was not dose-dependent, and no change in sperm
cell morphology was observed. Although the results of in vitro
tests for mutagenicity of 2-butoxyethanol were inconsistent, the
absence of structural alerts and the negative findings from in vivo
studies are sufficiently reassuring to allow the conclusion that
2-butoxyethanol is not mutagenic. 2-Butoxyethanol has not been found
to have an adverse effect on the immune system (Exon et al., 1991;
Smialowicz et al., 1992).
Based on limited data from case reports and one laboratory study,
similar acute effects -- including haemolytic effects as well as
effects on the central nervous system -- are observed in humans and
rats exposed to 2-butoxyethanol, although the effects are observed at
much higher exposure concentrations in humans than in rats.
11.1.2 Criteria for setting guidance values for 2-butoxyethanol
The following guidance is provided as a possible basis for
derivation of limits of exposure and for judgement of the quality of
environmental media by relevant authorities. Available data indicate
that the haematotoxicity associated with exposure to 2-butoxyethanol
is similar in laboratory animals and humans, although available data
do not permit quantification of dose-response for the latter. The
guidance value provided here is derived, therefore, on the basis of
studies conducted in animals. Based on limited data in humans, the
rat is likely more sensitive to the haemolytic effects of
2-butoxyethanol exposure (Carpenter et al., 1956; Bartnik et al.,
1987).
The dose-response for haematotoxicity in rats has been consistent
in an inhalation study of developmental toxicity, in which pregnant
animals were exposed to 2-butoxyethanol on days 6-15 of gestation (Tyl
et al., 1984), and in a subchronic inhalation toxicity study (Dodd et
al., 1983). In the developmental study, the NOAEL and LOAEL in the
dams were 50 ppm (242 mg/m3) and 100 ppm (483 mg/m3), respectively
(Tyl et al., 1984). In the subchronic inhalation study, the NOAEL and
LOAEL were 25 ppm (121 mg/m3) and 77 ppm (372 mg/m3), respectively
(Dodd et al., 1983). A tolerable concentration (TC) has been derived
as follows:
TC = [(242 mg/m3)/10] × [6/24] × [(0.16 m3 per day/0.215
kg)/(22 m3 per day/64 kg)]
= 13.1 mg/m3
where:
* 242 mg/m3 (50 ppm) is the NOAEL from the study (Tyl et al.,
1984) providing the best bounding of dose-response in the most
sensitive species;
* 10 is the uncertainty factor to account for intraspecies
variability in humans. No additional factor was incorporated to
address interspecies variability on the basis of limited data in
humans and several in vitro studies that indicate that rat
erythrocytes are far more sensitive than human erythrocytes to
the haemolytic effects associated with exposure to
2-butoxyethanol (and its metabolite 2-butoxyacetic acid). No
additional factor was incorporated to account for the short
duration of exposure in the critical study, as there is no
indication that effect levels vary with increased exposure
duration;
* 6/24 is the conversion from 6 h/day to continuous exposure; and
* [(0.16 m3 per day/0.215 kg)/(22 m3 per day/64 kg)] is the
scaling factor from rats to humans, based on the assumed
inhalation volume and body weight for rats (0.16 m3 per day and
0.215 kg, respectively) and humans (22 m3 per day and 64 kg,
respectively). The PBPK model of Corley et al. (1994, 1997)
would not result in an appreciably different TC at this level of
exposure.
It should be noted that the TC was based on a study involving the
whole-body exposure of rats and an assumption that 100% of the inhaled
2-butoxyethanol was retained. The extent of skin absorption has not
been formally taken into account in the development of this TC and may
be greater than intake via inhalation.
11.1.3 Sample risk characterization
The extremely limited nature of the available data to serve as a
basis for estimation of exposure should be borne in mind in
interpreting the comparisons presented here for indirect exposure of
the general population to 2-butoxyethanol. The concentration of 8 µg
2-butoxyethanol/m3 measured in a sample of indoor air collected in
northern Italy is approximately 1600-fold lower than the TC developed
in the preceding section. Levels of 2-butoxyethanol are considerably
higher in some occupational settings. In addition, skin absorption
may be of greater importance than respiratory absorption.
11.2 Evaluation of environmental effects
11.2.1 Aquatic environment
Data on measured levels of 2-butoxyethanol in surface waters are
insufficient for risk characterization. However, a sample risk
characterization for the aquatic environment is presented in which the
ratio between a predicted (local) environmental concentration
(PEClocal) and a predicted no-effect concentration (PNEC) is
calculated.
PEClocals for surface waters have been derived based upon data
from Australia (OECD, 1997) as well as information on all reported
releases to the environment in 1993 from individual industrial plants
in the USA (Staples et al., 1998). Calculations of expected surface
water concentration were based on worst-case scenarios for local river
flows identified from a US Geological Survey database. Site-specific
estimates were made for 36 industrial plants, of which 26 discharged
through sewage treatment plants and 10 discharged directly to rivers.
Both studies relied on fugacity modelling to predict the environmental
distribution of 2-butoxyethanol, yielding slightly different results.
However, both approaches indicated that most (84-96%) of the chemical
will partition to water, with almost all of the remainder volatilizing
to air. There is negligible binding of 2-butoxyethanol to
particulates, and no bioconcentration in organisms is expected. In
addition, 2-butoxyethanol is readily degraded by microorganisms.
A PEC for surface water in Sydney, Australia, based on the
assumption that all local usage passes through a single sewage
treatment plant and releases at a point source to a river, was
calculated as follows:
PEClocal (water) = Ceffluent/[(1 + Kp(susp) × C(susp)) × D]
= 50.4 µg/litre
where:
* Ceffluent is the concentration (g/litre) of the chemical in the
sewage treatment plant effluent, calculated as Ceffluent = W ×
(100 × P)/(100 × Q)
where:
W = emission rate: 1400 kg/day (OECD, 1997)
P = % removal by biodegradation in the sewage
treatment plant (modelled as 91% using the
SIMPLETREAT model)
Q = volume of wastewater: 250 000 m3/day (OECD, 1997)
* Kp(susp) is the suspended matter/water adsorption coefficient),
calculated as Kp(susp) = Foc(susp) × Koc
where:
Foc(susp)= the fraction of organic carbon in suspended
matter (0.01)
Koc = 0.411 × Kow
where:
Kow = the octanol/water partition coefficient
(6.76)
* C(susp) is the concentration of suspended matter in river water
(default value = 15mg/litre)
* D is the dilution factor for river flow (default value = 10)
As degradation in the sewage treatment plant is a large component
of the assumptions, and as it cannot be assumed that this level of
sewage treatment occurs in all countries globally, this calculation
can be revised assuming no sewage treatment (i.e. P = 0), yielding a
PEC of 560 µg/litre. This value assumes that all local release is
diluted with general wastewater from the urban centre. No values were
available for individual industrial plants in Sydney, Australia, and
therefore concentrations released directly to rivers cannot easily be
calculated.
Using the other approach of site-specific estimation (Staples et
al., 1998), 36 industrial plants in the USA were selected from 814
reporting emissions, on the basis of availability of river flow values
and worst-case releases. Calculations were based on local stream
flows, taking a value for the lowest flow expected over any single
7-day period once in 10 years. For plants emitting via a sewage
treatment system, degradation rates of 90% were assumed. Calculated
concentrations are "instantaneous," assuming no dilution by the
receiving stream, no degradation in the receiving waters, and no
distribution to media other than water. These are conservative
assumptions. Calculated in-stream concentrations ranged from 0.0002
to 21.7 mg/litre for emissions via sewage treatment (annual release
ranged from 18 000 to 974 000 kg for the 26 plants with sewage
treatment) and from 0.000 01 to 4.66 mg/litre for untreated emissions
(annual release ranged from 1870 to 35 000 kg for the 10 plants with
no sewage treatment). The highest reported concentration of
2-butoxyethanol in surface waters was 5.7 mg/litre following release
by the leather industry into the Hayashida River in Japan, before
treatment was introduced (Yasuhara et al., 1981). These measured and
estimated surface water concentrations are summarized in Table 2.
Table 2: PEC/PNEC ratios.
Location Sewage Highest PEC/PNEC
treatment concentration ratioa
(mg/litre)
Australiab Yes 0.05 0.3
(Sydney) No 0.56 3.4
USA (site Yes 21.7 131.5
specific)c No 4.66 28.2
Japand No 5.7 34.5
a Based on a PNEC of 165 µg/litre (see text).
b Modelled.
c Modelled, but based on known annual release for each site.
d Measured.
As a guide for those wishing to perform similar calculations
using local use/release figures, the Staples et al. (1998) study
estimates that the annual release of total glycol ethers (assuming
that 50% of released compounds would be 2-butoxyethanol) leading to
instantaneous 2-butoxyethanol concentrations in surface waters of 1
mg/litre would be 18 000 kg with sewage treatment and 1800 kg without
sewage treatment for streams with very low flow at 0.03 m3/s
(equivalent to 2.5 million litres/day).
A PNEC for surface waters may be calculated as follows:
PNEC = (165 mg/litre)/1000
= 165 µg/litre
where:
* 165 mg/litre is the lowest reported effect level for a lethality
end-point in aquatic species (48-h LC50 in the golden ide
[ Leuciscus idus melanotus], a freshwater fish); and
* 1000 is the uncertainty factor. The range of organisms tested in
short-term tests would justify application of an uncertainty
factor of 100, yielding a PNEC of 1.65 mg/litre, based on the
lowest reported LC50 in fish. However, there is some indication
that estuarine species may be more sensitive, although the lowest
reported LC50 for the grass shrimp ( Palaemonetes pugio) (96-h
LC50 = 5.4 mg/litre) is such an extreme outlier compared with
the range of other data that it is difficult to justify its use
as the basis for the PNEC calculation. Application of an
uncertainty factor of 1000 to the lowest freshwater value would
be protective for both freshwater and estuarine environments,
yielding margins relative to the 96-h LC50s for the grass shrimp
(5.4 mg/litre) and the oyster ( Crassostrea virginica) (89
mg/litre), the most sensitive of the estuarine invertebrates, of
33 and 540, respectively. For freshwater organisms, the
threshold concentration for inhibition of growth in algae
(long-term effect) cannot be justified as the basis for
application of uncertainty factors to establish a PNEC.
As the highest measured concentration in surface waters (at 5.7
mg/litre) is almost identical to the lowest reported LC50
concentration (at 5.4 mg/litre for the grass shrimp), it is not
surprising that high risk factors are generated. High-volume usage
and emissions to surface waters in a range of industries would lead to
locally high concentrations, principally where sewage treatment was
not in operation and river flow was low. It can be expected that
concentrations would exceed those likely to produce effects in some
aquatic species under these circumstances. However, the majority of
reported acute toxicity effect levels are 100 mg/litre or higher, and
most exceed 800 mg/litre. Four of 38 estimated surface water
concentrations exceed 2 mg/litre, with the remainder less than, and
usually substantially less than, 1 mg/litre (Figure 1). Most of these
estimates also fail to account for dilution in rivers. Using an
uncertainty factor of 100, justified by the range of toxicity data, on
the lowest reported freshwater LC50 and typical estimates of water
concentrations yields PEC/PNEC ratios of <1. Therefore, for most
releases to surface waters, the risk is considered to be low. It is
also unlikely that 2-butoxyethanol would be toxic to sewage treatment
plant bacteria, as the only reported effect level for bacteria is an
IC50 of >1000 mg/litre (Union Carbide, 1989).
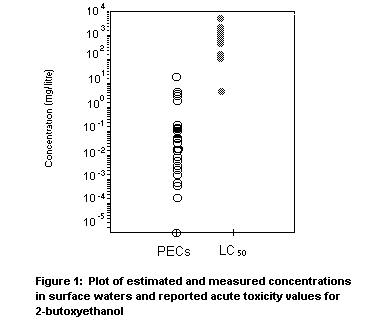 11.2.2 Terrestrial environment
Data are inadequate to characterize the risks to terrestrial
organisms of exposure to 2-butoxyethanol. A PEClocal(air) of 537 µg/m3,
based upon the use patterns of this chemical in Australia, has been
reported (OECD, 1997). Although available monitoring data are
limited, this predicted concentration is much higher than levels
measured in ambient air (see section 6). As 2-butoxyethanol is
expected to have a half-life in the atmosphere of less than 1 day,
these concentrations are considered to have no environmental
significance.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Previous evaluations of 2-butoxyethanol published by WHO, the
International Agency for Research on Cancer (IARC), the Joint FAO/WHO
Expert Committee on Food Additives (JECFA), or the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) were not identified. A
Screening Information Dataset (SIDS) Initial Assessment Report has
been prepared under the Organisation for Economic Co-operation and
Development (OECD) High Production Volume (HPV) Chemicals Programme
(OECD, 1997). Information on international hazard classification and
labelling is included in the International Chemical Safety Card that
has been reproduced in this document.
13. HUMAN HEALTH PROTECTION AND EMERGENCY ACTION
Human health hazards, together with preventative and protective
measures and first aid recommendations, are presented in the
International Chemical Safety Card (ICSC 0059) reproduced in this
document.
13.1 Human health hazards
2-Butoxyethanol is toxic to humans. Following long-term or
repeated exposure, effects on the blood may be observed.
13.2 Advice to physicians
In case of intoxication, immediate supportive measures should be
given, as central nervous system depression, respiratory paralysis,
hypotension, and metabolic acidosis have been observed in the few
hours post-exposure. Close monitoring for renal toxicity and possible
haemodialysis are mandatory in the subsequent days (renal
insufficiency may develop 2-3 days post-exposure) until recovery is
achieved, on average by the second week post-exposure.
13.3 Health surveillance advice
Periodic medical examination of the haematopoietic system should
be included in a health surveillance programme.
13.4 Spillage
As 2-butoxyethanol is toxic and absorbed through the skin,
emergency crews need to wear proper equipment, including a mask with
cartridge for organic vapour, for handling spills. The chemical
should not be allowed to enter drains or watercourses.
14. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Information on national regulations, guidelines, and standards
can be found in the International Register of Potentially Toxic
Chemicals (IRPTC), available from UNEP Chemicals (IRPTC), Geneva.
The reader should be aware that regulatory decisions about
chemicals taken in a certain country can be fully understood only in
the framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
INTERNATIONAL CHEMICAL SAFETY CARD
2-BUTOXYETHANOL ICSC: 0059
26.04.193
CAS # 111-76-2 Ethylene glycol monobutyl ether
RTECS # KJ8575000 Monobutyl glycol ether
UN # 2369 C6H14O2/CH3(CH2)2CH2OCH2CH2OH
EC # 603-014-00-0 Molecular mass: 118.2
TYPES OF HAZARD/ ACUTE HAZARDS/ PREVENTION FIRST AID/FIRE FIGHTING
EXPOSURE SYMPTONS
FIRE Combustible. NO open flames. NO contact with Powder, alcohol-resitant foam, water spary,
oxidizing agents. carbon dioxide.
EXPLOSION Above 61°C explosive vapour/air Above 61°C closed system, In case of fire: keep drums, etc., cool by
mixtures may be formed. ventilation. spraying with water.
EXPOSURE PREVENT GENERATION OF IN ALL CASES CONSULT A DOCTOR!
MISTS!
Inhalation Cough, drowsiness, headache, Ventilation, local exhaust, or Fresh air, rest, and refer for medical
nausea. breathing protection. attention.
Skin MAY BE ABSORBED! Dry skin Protective gloves, protective Remove contaminated clothes, rinse skin with
(further see Inhalation). clothing. plenty of water or shower, and refer for
medical attention.
Eyes Redness, pain, blurred vision. Safety goggles or eye protection First rinse with plenty of water for several
combination with breathing minutes (remove contact lenses if easily
protection. possible), then take to a doctor.
(continued)
TYPES OF HAZARD/ ACUTE HAZARDS/ PREVENTION FIRST AID/FIRE FIGHTING
EXPOSURE SYMPTONS
Ingestion Abdominal pain, diarrhoea, Do not eat, drink, or smoke during Rinse mouth, give plenty of water to drink,
nausea, vomiting (further see work. induce vomiting (ONLY IN CONSCIOUS PERSONS),
inhalation). and refer for medical attention.
SPILLAGE DISPOSAL PACKAGING & LABELLING
Collect leaking and spilled liquid in sealable containers as far as possible, Xn Symbol
wash away remainder with plenty of water. R: 20/21/22-37
S: 24/25
UN Hazard Class: 6.1
UN Subsidiary Risks:
UN Pack Group: III
Airtight. Do not transport with food and feedstuffs.
EMERGENCY RESPONSE STORAGE
NFPA Code: H 2; F 2; R 0 Separated from strong oxidants, food and feedstuffs;
keep in the dark.
IMPORTANT DATA
PHYSICAL STATE: APPEARANCE: ROUTES OF EXPOSURE:
COLOURLESS LIQUID, WITH CHARACTERISTIC ODOUR. The substance can be absorbed into the body by inhalation
and through the skin, and by ingestion.
CHEMICAL DANGERS:
The substance can form explosive peroxides. The substance INHALATION RISK:
decomposes, producting toxic fumes. Reacts with strong A harmful contamination of the air will be reached rather
oxidants, causing fire and explosion hazard. slowly on evaporation of this substance at 20°C.
(continued)
OCCUPATIONAL EXPOSURE LIMITS: EFFECTS OF SHORT-TERM EXPOSURE:
TLV: 25 ppm; 121 mg/m3 (as TWA) (skin) (ACGIH 1992-1993) The substance irritates the eyes, the skin, and the
respiratory tract. Exposure could cause central nervous
system depression and liver and kidney damage.
EFECTS OF LONG-TERM OR REPEATED EXPOSURE:
The liquid defats the skin. The substance may have effects
on the haematopoietic system, resulting in blood disorders.
PHYSICAL PROPERTIES
Boiling point: 171°C Flash point: (c.c.) 61°C
Melting point: -75°C Auto-ignition temperature: 238°C
Relative density (water = 1) 0.90 Explosive limits, vol% in air: 1.1-12.7
Solubility in water: miscible Octanol/water partition coefficient as log Pow: 0.830
Vapour pressure, kPa at 20°C: 0.10
Relative vapour density (air = 1); 4.1
Relative density of the vapour/air-mixture at 20°C (air=1): 1.00
ENVIRONMENTAL DATA
This substance may be hazardous to the environment; speical attention should be given to the water environment and aquifer.
NOTES
Depending on the degree of exposure, periodic medical examination is indicated. Check for peroxides prior to distillation,
render harmless if positive. Keep in dark because of possible formation of explosive peroxides.
ADDITIONAL INFORMATION
LEGAL NOTICE: Neither the CEC or the IPCS no any person acting on behalf of the CEC or the IPCS is responsible for the use
which might be made of this information
(c) IPCS CEC 1996
REFERENCES
Amoore JE, Hautala E (1983) Odor as an aid to chemical safety: odor
threshold compared with threshold limit values and volatilities for
214 industrial chemicals in air and water dilution. Journal of
applied toxicology, 36(6):272-290.
Angerer J, Lichterbeck E, Begerow J, Jekel S, Lehnert G (1990)
Occupational chronic exposure to organic solvents: XIII. Glycol ether
exposure during the production of varnishes. International archives
of occupational and environmental health, 62(2):123-126.
ATSDR (1996) Toxicological profile for 2-butoxyethanol and
2-butoxyethanol acetate (August 1996 draft). Atlanta, GA, US
Department of Health and Human Services, Public Health Service, Agency
for Toxic Substances and Disease Registry.
Bartnik FG, Reddy AK, Klecak G, Zimmermann V, Hostynek JJ, Kunstler K
(1987) Percutaneous absorption, metabolism, and hemolytic activity of
n-butoxyethanol. Fundamental and applied toxicology, 8:59-70.
Bauer PH, Weber M, Mur JM, Protois JC, Bollaert PE, Condi A, Larcan A,
Lambert H (1992) Transient noncarcinogenic pulmonary edema following
massive ingestion of ethylene glycol butyl ether. Intensive care
medicine, 18:250-251.
Bormett GA, Bartels MJ, Markham DA (1995) Determination of
2-butoxyethanol and butoxyacetic acid in rat and human blood by gas
chromatography-mass spectrometry. Journal of chromatography,
B665:315-325.
Bridie AL (1979) The acute toxicity of some petrochemicals to fish.
Water research, 13:623-626.
Bringmann G, Kuhn R (1977) Results of the damaging effects of water
pollutants on Daphnia magna. Zeitschrift fuer Wasser und Abwasser
Forschung, 10:161-166.
Bringmann G, Kuhn R (1980a) Comparison of the toxicity thresholds of
water pollutants to bacteria, algae, and protozoa in the cell
multiplication test. Water research, 14:231-241.
Bringmann G, Kuhn R (1980b) Determination of the toxicity of water
pollutants to protozoa. II. Bacteriovorous ciliates. Zeitschrift
fuer Wasser und Abwasser Forschung, 13:26-31.
Bringmann G, Kuhn R (1982) Results of the toxic action of water
pollutants on Daphnia magna Straus tested by an improved
standardized procedure. Zeitschrift fuer Wasser und Abwasser
Forschung, 15:1-6.
Canadian Chemical Producers' Association (1996) Reducing emissions.
A Responsible Care initiative. 1994 emissions inventory and five
year projections. Ottawa, 36 pp.
Carpenter CP, Pozzani UC, Woil CS, Nair JH, Keck GA, Smyth HF Jr
(1956) The toxicity of butyl cellosolve solvent. American Medical
Association Archives of industrial health, 14:114-131.
CEFIC (1995) CEFIC Document 486/95/7/stat/year, Oxygenated Solvents
S.G. -- Statistical Investigation 1994 (23 February 1995). Brussels,
European Chemical Industry Council [cited in OECD, 1997].
Chiewchanwit T, Au WW (1995) Mutagenicity and cytotoxicity of
2-butoxyethanol and its metabolite, 2-butoxyacetaldehyde, in Chinese
hamster (CHO-AS52) cells. Mutation research, 334(13):341-346.
Ciccioli P, Brancaleoni E, Cecinato A, Sparapani R, Frattoni M (1993)
Identification and determination of biogenic and anthropogenic
volatile organic compounds in forest areas of northern and southern
Europe and a remote site of the Himalaya region by high-resolution gas
chromatography-mass spectrometry. Journal of chromatography,
643:55-69.
Ciccioli P, Cecinato A, Brancaleoni E, Frattoni M, Bruner F, Maione M
(1996) Occurrence of oxygenated volatile organic compounds (VOC) in
Antarctica. International journal of analytical chemistry,
62:245-253.
CMA (1994) HEDSET for ethylene glycol butyl ether. Prepared for the
European Union Existing Substances Programme. Washington, DC, Chemical
Manufacturers Association.
Corley RA, Bormett GA, Ghanayem BI (1994) Physiologically-based
pharmacokinetics of 2-butoxyethanol and its major metabolite,
2-butoxyacetic acid, in rats and humans. Toxicology and applied
pharmacology, 129(1):61-79.
Corley RA, Markham DA, Banks C, Delorme P, Masterman A, Houle JM
(1997) Physiologically-based pharmacokinetics and the dermal
absorption of 2-butoxyethanol vapor by humans. Fundamental and
applied toxicology, 39:120-130.
Dawson GW, Jennings AL, Drozdowski D, Rider E (1977) The acute
toxicity of 47 industrial chemicals to fresh and saltwater fishes.
Journal of hazardous materials, 1:303-318.
Dean BS, Krenzelok EP (1992) Clinical evaluation of pediatric ethylene
glycol monobutyl ether poisonings. Journal of toxicology and
clinical toxicology, 30(4):557-563.
Dill DC (1995) Environmental summary for Dowanol EB and DB glycol
ethers. Unpublished report. Midland, MI, Dow Chemical Company.
Dodd DE, Snelling WM, Maronpot RR, Ballentyne B (1983) Ethylene glycol
monobutyl ether: acute 9-day and 90-day vapor inhalation studies in
Fischer 344 rats. Toxicology and applied pharmacology, 68:405-414.
Doe JE (1984) Further studies on the toxicology of the glycol ethers
with emphasis on rapid screening and hazard assessment.
Environmental health perspectives, 57:199-206.
Dow (1979) Toxicity of Dowanol EB to freshwater organisms.
Unpublished report. Midland, MI, Dow Chemical Company (Report No.
ES-330).
Dow (1988) Dowanol EB glycol ether: evaluation of the toxicity to
the green alga, Selenastrum capricornutum. Unpublished report.
Midland, MI, Dow Chemical Company.
ECETOC (1994) Butoxyethanol criteria document. Including a
supplement for 2-butoxyethyl acetate. Brussels, European Chemical
Industry, Ecology and Toxicology Centre (Special Report No. 7, April
1994).
Elias Z, Daniere MC, Marande AM, Poirot O, Terzelti F, Schneider O
(1996) Genotoxic and/or epigenetic effects of some glycol ethers:
results of different short-term tests. Occupational hygiene,
2:187-212.
Elliott BM, Ashby J (1997) Review of the genotoxicity of
2-butoxyethanol. Mutation research, 387:89-96.
Environment Canada (1997) Canadian Environmental Protection Act.
Priority Substances List Supporting Documentation --
2-Butoxyethanol. Vol. 2 (draft). Ottawa, pp. 187-212.
Exon JH, Mather GG, Bussiere JL, Olson DP, Talcott PA (1991) Effects
of subchronic exposure of rats to 2-methoxyethanol or 2-butoxyethanol:
thymic atrophy and immunotoxicity. Fundamental and applied
toxicology, 16(4):830-840.
Foster PMD, Lloyd SC, Blackburn DM (1987) Comparison of the in vivo
and in vitro testicular effects produced by methoxy-, ethoxy- and
n-butoxy acetic acids in the rat. Toxicology, 43:17-30.
Ghanayem BI (1989) Metabolic and cellular basis of 2-butoxyethanol-
induced hemolytic anemia in rats and assessment of human risk in
vitro. Biochemical pharmacology, 38(10):1679-1684.
Ghanayem BI, Sullivan CA (1993) Assessment of the haemolytic activity
of 2-butoxyethanol and its major metabolite, butoxyacetic acid, in
various mammals including humans. Human experimental toxicology,
1214:305-311.
Ghanayem BI, Blair PC, Thompson MB, Maronpot RR, Matthews HB (1987a)
Effect of age on the toxicity and metabolism of ethylene glycol
monobutyl ether (2-butoxyethanol) in rats. Toxicology and applied
pharmacology, 91:222-234.
Ghanayem BI, Burka LT, Sanders JM, Matthews H (1987b) Metabolism and
disposition of ethylene glycol monobutyl ether (2-butoxyethanol) in
rats. Drug metabolism and disposition, 15:478-484.
Ghanayem BI, Ward SM, Blair PC, Matthews HB (1990) Comparison of the
hematologic effects of 2-butoxyethanol using two types of hematology
analyzers. Toxicology and applied pharmacology, 106(2):341-345.
Ghanayem BI, Sanchez IM, Matthews HB (1992) Development of tolerance
to 2-butoxyethanol-induced hemolytic anemia and studies to elucidate
the underlying mechanisms. Toxicology and applied pharmacology,
112(2):198-206.
Gijsenbergh FP, Jenco M, Veulemans H, Groesenken D, Verberckmoes R,
Delooz HH (1989) Acute butyl-glycol intoxication: a case report.
Human toxicology, 8:243-245.
Gingell R, Boatman RJ, Lewis S (1997) Comparative acute toxicity
of ethylene glycol mono-n-butyl ether in several species. Arlington,
VA, Chemical Manufacturers Association.
Gollapudi BB, Barber ED, Lawlor TE, Lewis SA (1996) Re-examination of
the mutagenicity of ethylene glycol monobutyl ether to Salmonella
tester strain TA97a. Mutation research, 370:61-64.
Grant D, Slush S, Jones HB, Gangolli SD, Butler WH (1985) Acute
toxicity and recovery in the hemopoietic system of rats after
treatment with ethylene glycol monomethyl and monobutyl ethers.
Toxicology and applied pharmacology, 77:187-200.
Greenspan AH, Reardon RC, Gingell R, Rosica KA (1995) Human repeated
insult patch test of 2-butoxyethanol. Contact dermatitis, 33:59-60.
Groeseneken D, Van Vlem E, Veulemans H, Masschelein R (1986) Gas
chromatographic determination of methoxyacetic and ethoxyacetic acid
in urine. British journal of industrial medicine, 43:62-65.
Groeseneken D, Veulemans H, Masschelein R, Van Vlem E (1989) An
improved method for the determination in urine of alkoxyacetic acids.
International archives of occupational and environmental health,
61:249-254.
Gualtieri J, Harris C, Roy R, Corley R, Manderfield C (1995) Multiple
2-butoxyethanol intoxications in the same patient: clinical findings,
pharmacokinetics, and therapy. Journal of toxicology and clinical
toxicology, 33(5):550-551.
Hardin BD, Goad PT, Burg JR (1984) Developmental toxicity of four
glycol ethers applied cutaneously to rats. Environmental health
perspectives, 57:69-74.
Heindel JJ, Lamb JC IV, Chapin RE, Gulati DK, Hope E, George J,
Jameson CW, Teague J, Schwetz BA (1989) Reproductive toxicity
testing by continuous breeding: Test protocol in Swiss (CD-1) mice.
Available from National Technical Information Service, Springfield, VA
(NTIS No. PB89152451AS).
Heindel JJ, Gulati DK, Russell VS, Reel JR, Lawton AD, Lamb JC IV
(1990) Assessment of ethylene glycol monobutyl and monophenyl ether
reproductive toxicity using a continuous breeding protocol in Swiss
CD-1 mice. Fundamental and applied toxicology, 15(4):683-696.
Hoflack JC, Lambolez L, Elias Z, Vasseur P (1995) Mutagenicity of
ethylene glycol ethers and of their metabolites in Salmonella
typhimurium his-. Mutation research, 341(4):281-287.
Howard PH, Boethling RS, Jarvis WF, Meylan WM, Michalenko EM (1991)
Handbook of environmental degradation rates. Chelsea, MI, Lewis
Publishers Inc. [cited in ATSDR, 1996].
US NLM (1997) Hazardous substances data bank. Last revision on
7/11/96. Bethesda, MD, National Library of Medicine, National
Toxicology Information Program.
IPCS (1993) International Chemical Safety Card -- 2-Butoxyethanol.
Geneva, World Health Organization, International Programme on Chemical
Safety (No. 0059).
Johanson G (1994) Inhalation toxicokinetics of butoxyethanol and its
metabolite butoxyacetic acid in the male Sprague-Dawley rat.
Archives of toxicology, 68(9):588-594.
Johanson G, Boman A (1991) Percutaneous absorption of 2-butoxyethanol
vapor in human subjects. British journal of industrial medicine,
48(11):788-792.
Johanson G, Kronborg H, Naslund PH, Nordqvist MB (1986) Toxicokinetics
of inhaled 2-butoxyethanol (ethylene glycol monobutyl ether) in man.
Scandinavian journal of work and environmental health,12:594-602.
Johanson G, Boman A, Dynesius B (1988) Percutaneous absorption of
2-butoxyethanol in man. Scandinavian journal of work and
environmental health, 14:101-109.
Jonsson AK, Steen G (1978) n-Butoxyacetic acid, a urinary metabolite
from inhaled n-butoxyethanol (butylcellosolve). Acta
Pharmacologica et Toxicologica, 42:354-356.
Junke I, Ludemann D (1978) Results of the examination of the effects
of 200 chemical compounds on fish toxicity using the golden orfe test.
Zeitschrift fuer Wasser und Abwasser Forschung, 11:161-164.
Keith G, Coulais C, Edoh A, Botlin MC, Rihn B (1996) Ethylene glycol
monobutyl ether has neither epigenetic nor genotoxic effects in acute
treated rats and in subchronic treated v-Ha- ras transgenic mice.
Occupational hygiene, 2:237-249.
Kennah HE II, Hignet S, Laux PE, Dorko JD, Barrow CS (1989) An
objective procedure for quantitating eye irritation based upon changes
of corneal thickness. Fundamental and applied toxicology,
12(2):258-268.
Kennedy ER, O'Connor PF, Grote AA (1990) Application of
multidimensional gas chromatography-mass spectrometry to the
determination of glycol ethers in air. Journal of chromatography,
522:303-333.
Koenemann H (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part 1. Relationships for 50 industrial
pollutants. Toxicology, 19:209-221.
Krasavage WJ (1986) Subchronic oral toxicity of ethylene glycol
monobutyl ether in male rats. Fundamental and applied toxicology,
6:349-355.
Leaf DA (1985) Glycol ethers: an overview. Washington, DC, US
Environmental Protection Agency, Office of Pesticides and Toxic
Substances.
McGregor DB (1984) The genotoxicity of glycol ethers. Environmental
health perspectives, 57:97-103.
Medinsky MA, Singh G, Bechtold WE, Bond JA, Sabourin PJ, Birnbaum LS,
Henderson RF (1990) Disposition of three glycol ethers administered in
drinking water to male F344/N rats. Toxicology and applied
pharmacology, 102(3):443-455.
Nagano K, Nakayama E, Koyano M, Oobayashi H, Adachi H, Yamada T (1979)
Mouse testicular atrophy induced by ethylene glycol monoalkyl ethers.
Japanese journal of industrial health, 21:29-35.
Nelson BR, Setzer JV, Brightwell WS, Mathinos PR, Kuczuk MH, Weaver
TE, Goad PT (1984) Comparative inhalation teratogenicity of four
glycol ether solvents and an amino derivative in rats.
Environmental health perspectives, 57:261-271.
NIOSH (1983) National Occupational Exposure Survey (NOES), 1981-83:
estimated total and female employees, actual observation and
trade-named exposure to EGEE, EGHE, EGBE, and their acetates.
Unpublished database. Cincinnati, OH, US Department of Health and
Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, Division of
Surveillance, Hazard Evaluations, and Field Studies, Surveillance
Branch.
NIOSH (1990) Criteria for a recommended standard. Occupational
exposure to ethylene glycol monobutyl ether and ethylene glycol
monobutyl ether acetate. Cincinnati, OH, US Department of Health and
Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, Division of
Standards Development and Technology Transfer (DHHS [NIOSH]
Publication No. 90-118).
NIOSH (1994) Manual of analytical methods, 4th ed. Cincinnati, OH,
US Department of Health and Human Services, Public Health Service,
Centers for Disease Control and Prevention, National Institute for
Occupational Safety and Health (DHHS [NIOSH] Publication No. 94-113).
NTP (1993) NTP technical report on toxicity studies of ethylene
glycol ethers 2-methoxyethanol, 2-ethoxyethanol, 2-butoxyethanol
administered in drinking water to F344/N rats and B6C3F1 mice.
Research Triangle Park, NC, US Department of Health and Human
Services, Public Health Service, National Institutes of Health,
National Toxicology Program (NIH Publication No. 93-3349).
OECD (1997) Screening Information Dataset (SIDS) initial assessment
report on 2-butoxyethanol. 6th SIDS Initial Assessment Meeting.
Paris, Organisation for Economic Co-operation and Development.
OSHA (1990) 2-Butoxyethanol (butyl cellosolve) and 2-butoxyethyl
acetate (butyl cellosolve acetate). Salt Lake City, UT, US
Department of Labor, Occupational Safety and Health Administration,
Organic Methods Evaluation Branch, OSHA Analytical Laboratory.
Price KS, Waggy GT, Conway RA (1974) Brine shrimp bioassay and
seawater BOD of petrochemicals. Journal of the Water Pollution
Control Federation, 46:63-77.
Rambourg-Schepens MD, Buffet M, Bertault R, Jaussaud M, Journe B, Fay
R, Lamiable D (1988) Severe ethylene glycol butyl ether poisoning.
Kinetics and metabolic pattern. Human toxicology, 7:187-189.
Rettenmeier AW, Hennigs R, Wodarz R (1993) Determination of
butoxyacetic acid and n-butoxyacetylglutamine in urine of lacquerers
exposed to 2-butoxyethanol. International archives of occupational
and environmental health, 65(1) (Suppl.):S151-S153.
Rowe VK, Wolf MA (1982) Derivatives of glycols. In: Clayton GD,
Clayton EF, eds. Patty's industrial hygiene and toxicology, 3rd rev.
ed. Vol. 2. New York, NY, John Wiley and Sons, pp. 3909-4052.
Sakai T, Araki T, Masuyama Y (1993) Determination of urinary
alkoxyacetic acid by rapid and simple method for biological monitoring
of workers exposed to glycol ethers and their acetates.
International archives of occupational and environmental health,
64:495-498.
Sakai T, Araki T, Morita Y, Masuyama Y (1994) Gas chromatographic
determination of butoxyacetic acid after hydrolysis of conjugated
metabolites in urine from workers exposed to 2-butoxyethanol.
International archives of occupational and environmental health,
66:249-254.
Sax NI, Lewis RJ (1987) Hawley's condensed chemical dictionary, 11th
ed. New York, NY, Van Nostrand Reinhold Company, pp. 488-489.
Schuler RL, Hardin BD, Niemeier RW, Booth G, Hazelden K, Piccirillo V
(1984) Results of testing 15 glycol ethers in a short-term in vivo
reproductive toxicity assay. Environmental health perspectives,
57:141-146.
Shyr LJ, Sabourin PJ, Medinsky MA, Birnbaum LS, Henderson RF (1993)
Physiologically-based modeling of 2-butoxyethanol disposition in rats
following different routes of exposure. Environmental research,
63(2):202-218.
Smallwood AW, DeBord KE, Lowry LK (1984) Analyses of ethylene glycol
monoalkyl ethers, and their proposed metabolites in blood and urine.
Environmental health perspectives, 57:249-253.
Smallwood AW, DeBord KE, Burg J, Moseley C, Lowry LK (1988)
Determination of urinary 2-ethoxyacetic acid as an indicator of
occupational exposure to 2-ethoxyethanol. Applied industrial
hygiene, 3(2):47-50.
Smialowicz RJ, Williams WC, Riddle HH, Andres DL, Luebke RW, Copeland
CB (1992) Comparative immunosuppression of various glycol ethers
orally administered to Fischer 344 rats. Fundamental and applied
toxicology, 18(4):621-627.
Sohnlein B, Letzel S, Weltle D, Rüdiger HW, Angerer J (1993) XIV.
Examinations concerning the evaluation of a limit value for
2-ethoxyethanol and 2-ethoxyethylacetate and the genotoxic effects of
these glycol ethers. International archives of occupational and
environmental health, 64(7):479-484.
Staples CA, Boatman RJ, Cano ML (1998) Ethylene glycol ethers: An
environmental risk assessment. Chemosphere, 36:1585-1613.
Tyl RW, Millicovsky G, Dodd DE, Pritts IM, France KA, Fisher LC (1984)
Teratologic evaluation of ethylene glycol monobutyl ether in Fischer
344 rats and New Zealand white rabbits following inhalation exposure.
Environmental health perspectives, 57:47-68.
Tyler TR (1984) Acute and subchronic toxicity of ethylene glycol
monobutyl ether. Environmental health perspectives, 57:85-191.
Udden MM (1994) Hemolysis and deformability of erythrocytes exposed to
butoxyacetic acid, a metabolite of 2-butoxyethanol: II. Resistance in
red blood cells from humans with potential susceptibility. Journal
of applied toxicology, 14(2):97-102.
Udden MM (1996) Effects of butoxyacetic acid on human red cells.
Occupational hygiene, 2:283-290.
Udden MM, Patton CS (1994) Hemolysis and deformability of erythrocytes
exposed to butoxyacetic acid, a metabolite of 2-butoxyethanol: I.
Sensitivity in rats and resistance in normal humans. Journal of
applied toxicology, 412:91-96.
Union Carbide (1989) Ecological fate and effects data on four
selected glycol ether products. Unpublished report. South
Charleston, WV, Union Carbide Chemicals and Plastic Co. Inc.
US EPA (1984) Acute toxicity studies on Wellaid 31. Study submitted
to the US Environmental Protection Agency by Amoco Corporation [cited
in OECD, 1997].
US ITC (1996) Preliminary report on U.S. production of selected
synthetic organic chemicals (including synthetic plastics and
resin materials). Fourth Quarter and Preliminary International Trade
Commission, Series C/P-96-2; No. 26, pp. 2-12. Totals, 1995.
Washington, DC, US International Trade Commission.
Verschueren K (1983) Handbook of environmental data on organic
chemicals, 2nd ed. New York, NY, Van Nostrand Reinhold Company, 1310
pp.
Veulemans H, Groeseneken D, Masschelein R, Van Vlem E (1987) Survey of
ethylene glycol ether exposures in Belgian industries and workshops.
American Industrial Hygiene Association journal, 48(8):671-676.
Vincent R, Cicolella A, Subra I, Rieger B, Parrot P, Pierre F (1993)
Occupational exposure to 2-butoxyethanol for workers using window
cleaning agents. Applied occupational and environmental hygiene,
8(6):580-586.
von Oettingen WF, Jirouche EA (1931) The pharmacology of ethylene
glycol and some of its derivatives in relation to their chemical
constitution and physical chemical properties. Journal of
pharmacology and experimental therapeutics, 42(3):355-372.
Werner HW, Mitchell JL, Miller JW, von Oettingen WF (1943a) The acute
toxicity of vapors of several monoalkyl ethers of ethylene glycol.
Journal of industrial hygiene and toxicology, 25:157-163.
Werner HW, Nawrocki CZ, Mitchell JL, Miller JW, von Oettingen WF
(1943b) Effects of repeated exposure of rats to monoalkyl ethylene
glycol ether vapors. Journal of industrial hygiene and toxicology,
25:374-379.
Werner HW, Mitchell JL, Miller JW, von Oettingen WF (1943c) Effects of
repeated exposure of dogs to monoalkyl ethylene glycol ether vapors.
Journal of industrial hygiene and toxicology, 25:409-414.
Yasuhara A, Shiraisi H, Tsuji M, Okuno T (1981) Analysis of organic
substances in highly polluted river water by mass spectrometry.
Environmental science and technology, 15:570-573.
Zeiger E, Anderson B, Haworth S, Lawlor T, Mortelmans K (1992)
Salmonella mutagenicity tests: V. Results from the testing of 311
chemicals. Environmental and molecular mutagenesis, 19 (Suppl.
21):2-141.
Zissu D (1995) Experimental study of cutaneous tolerance to glycol
ethers. Contact dermatitis, 32(2):74-77.
APPENDIX 1 -- SOURCE DOCUMENTS
NIOSH (1990)
Copies of this source document ( Criteria for a recommended
standard. Occupational exposure to ethylene glycol monobutyl ether
and ethylene glycol monobutyl ether acetate; NIOSH Publication No.
90-118) are available from:
Publications Office
National Institute for Occupational Safety and Health
4676 Columbia Parkway
Cincinnati, OH 45226
USA
(513) 533-8471
This document was prepared by Joann Wess and reviewed internally
by staff of the National Institute for Occupational Safety and Health.
The draft document was reviewed externally by Dr F. Mirer, United Auto
Workers; Mr M. Gillen, Workers' Institute for Safety and Health; Mr F.
Burkhardt, International Brotherhood of Builders and Allied Trades; Dr
J. McCuen, ARCO Chemical Company; Mr W. Lypka, Graphic Communications
International Union; Dr H. Veulemans, Laboratorium voor
arbeidshygienne en-toxicologie; Dr E.M. Johnson, Jefferson Medical
College; Dr J.V. Rodricks, Dr J.S. Ferguson, Dr R.M. Putzrath, Mr M.
Fitzgerald, Chemical Manufacturers Association; Dr L. Welch, George
Washington University; Dr P. Sharma, Utah State University; Dr R.
Elves, Department of the Air Force; and Dr F. Welsch, Chemical
Industry Institute of Toxicology.
ATSDR (1996)
Copies of the ATSDR's Toxicological profile for
2-butoxyethanol and 2-butoxyethanol acetate (draft for public
comment) may be obtained from:
Agency for Toxic Substances and Disease Registry
Division of Toxicology
1600 Clifton Road NE, E-29
Atlanta, GA 30333
USA
This ATSDR draft document has undergone internal ATSDR review.
The document has also been reviewed by an expert panel of
nongovernmental reviewers consisting of the following members: Dr W.
Decker, Private Consultant, El Paso, TX; Dr A. Gregory, Private
Consultant, Sterling, VA; and Dr R. Rubin, Johns Hopkins School of
Public Health, Baltimore, MD.
APPENDIX 2 -- CICAD PEER REVIEW
The draft CICAD on 2-butoxyethanol was sent for review to
institutions and organizations identified by IPCS after contact with
IPCS national Contact Points and Participating Institutions, as well
as to identified experts. Comments were received from:
BASF, Ludwigshafen, Germany
Chemical Manufacturers Association, Arlington, USA
Department of Health, London, United Kingdom
Environment Canada, Ottawa, Canada
Health and Safety Executive, Liverpool, United Kingdom
Health Canada, Ottawa, Canada
Ministry of Health and Welfare, Government of Japan, Tokyo, Japan
National Chemicals Inspectorate (KEMI), Solna, Sweden
National Institute of Occupational Health, Budapest, Hungary
National Institute of Public Health and Environmental Protection,
Bilthoven, The Netherlands
National Occupational Health & Safety Commission, Sydney,
Australia
Oxygenated Solvents Producers Association, Brussels, Belgium
United States Department of Health and Human Services (National
Institute of Environmental Health Sciences, Research Triangle
Park)
United States Environmental Protection Agency (National Center
for Environmental Assessment, Washington, DC)
APPENDIX 3 -- CICAD FINAL REVIEW BOARD
Berlin, Germany, 26-28 November 1997
Members
Dr H. Ahlers, Education and Information Division, National Institute
for Occupational Safety and Health, Cincinnati, OH, USA
Mr R. Cary, Health Directorate, Health and Safety Executive, Bootle,
United Kingdom
Dr S. Dobson, Institute of Terrestrial Ecology, Huntingdon, United
Kingdom
Dr R.F. Hertel, Federal Institute for Health Protection of Consumers &
Veterinary Medicine, Berlin, Germany (Chairperson)
Mr J.R. Hickman, Health Protection Branch, Health Canada, Ottawa,
Ontario, Canada
Dr I. Mangelsdorf, Documentation and Assessment of Chemicals,
Fraunhofer Institute for Toxicology and Aerosol Research, Hanover,
Germany
Ms M.E. Meek, Environmental Health Directorate, Health Canada, Ottawa,
Ontario, Canada (Rapporteur)
Dr K. Paksy, Department of Reproductive Toxicology, National Institute
of Occupational Health, Budapest, Hungary
Mr V. Quarg, Ministry for the Environment, Nature Conservation &
Nuclear Safety, Bonn, Germany
Mr D. Renshaw, Department of Health, London, United Kingdom
Dr J. Sekizawa, Division of Chemo-Bio Informatics, National Institute
of Health Sciences, Tokyo, Japan
Prof. S. Soliman, Department of Pesticide Chemistry, Alexandria
University, Alexandria, Egypt (Vice-Chairperson)
Dr M. Wallen, National Chemicals Inspectorate (KEMI), Solna, Sweden
Ms D. Willcocks, Chemical Assessment Division, Worksafe Australia,
Camperdown, Australia
Dr M. Williams-Johnson, Division of Toxicology, Agency for Toxic
Substances and Disease Registry, Atlanta, GA, USA
Dr K. Ziegler-Skylakakis, Senatskommission der Deutschen
Forschungsgemeinschaft zuer Pruefung gesundheitsschaedlicher
Arbeitsstoffe, GSF-Institut fuer Toxikologie, Neuherberg,
Oberschleissheim, Germany
Observers
Mrs B. Dinham,1 The Pesticide Trust, London, United Kingdom
Dr R. Ebert, KSU Ps-Toxicology, Huels AG, Marl, Germany (representing
ECETOC, the European Centre for Ecotoxicology and Toxicology of
Chemicals)
Mr R. Green,1 International Federation of Chemical, Energy, Mine and
General Workers' Unions, Brussels, Belgium
Dr B. Hansen,1 European Chemicals Bureau, European Commission, Ispra,
Italy
Dr J. Heuer, Federal Institute for Health Protection of Consumers &
Veterinary Medicine, Berlin, Germany
Mr T. Jacob,1 DuPont, Washington, DC, USA
Ms L. Onyon, Environment Directorate, Organisation for Economic Co-
operation and Development, Paris, France
Dr H.J. Weideli, Ciba Speciality Chemicals Inc., Basel, Switzerland
(representing CEFIC, the European Chemical Industry Council)
Secretariat
Dr M. Baril, International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland
Dr R.G. Liteplo, Health Canada, Ottawa, Ontario, Canada
Ms L. Regis, International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland
Mr A. Strawson, Health and Safety Executive, London, United Kingdom
Dr P. Toft, Associate Director, International Programme on Chemical
Safety, World Health Organization, Geneva, Switzerland
1 Invited but unable to attend.
RÉSUMÉ D'ORIENTATION
Ce CICAD relatif au 2-butoxyéthanol a été rédigé sur la base
d'évaluations préparées par le National Institute for Occupational
Safety and Health (NIOSH, 1990) et l'Agency for Toxic Substances and
Disease Registry (ATSDR, 1996). Une étude de la littérature publiée
jusqu'en mai 1997 a fourni des données complémentaires, à quoi se sont
ajoutés les éléments d'information obtenus lors de l'évaluation par
des pairs du présent CICAD. Des informations concernant la nature de
l'évaluation par les pairs et la disponibilité des documents originaux
figurent à l'appendice 1. Des informations sur cette évaluation sont
données à l'appendice 2. Ce CICAD a été approuvé en tant
qu'évaluation internationale lors d'une réunion du Comité d'évaluation
finale qui s'est tenue à Berlin (Allemagne) du 26 au 28 novembre 1997.
La liste des participants à la réunion du Comité d'évaluation finale
figure à l'appendice 3. La fiche d'information sur la sécurité
chimique (ICSC 0059), préparée par le Programme international sur la
sécurité chimique (IPCS, 1993), est également reproduite dans le
présent document.
Le 2-butoxyéthanol (CAS N° 111-76-2) est un éther du glycol
produit en quantités industrielles. C'est un liquide incolore
miscible à l'eau et soluble dans la plupart des solvants organiques.
Il est très largement utilisé dans les enduits, les laques à séchage
rapide, les émaux, les vernis, les dissolvants et les peintures au
latex. On l'utilise également dans les nettoyants pour métaux et
certains produits ménagers. Dans l'atmosphère, il est présent
entièrement à l'état gazeux et comme sa demi-vie à ce niveau est
d'environ 17 heures, il n'y a guère de risque de transport
atmosphérique. Dans l'eau, on estime sa demi-vie à environ 1-4
semaines. Il est probablement décomposé sans difficulté en aérobiose
dans le sol et dans l'eau. Son potentiel de bioaccumulation est
faible. Selon les données limitées dont on dispose, les
concentrations auxquelles on peut être exposé dans l'air sont de
l'ordre du µg/m3. S'il y a exposition indirecte de la population,
c'est très probablement par inhalation ou absorption percutanée lors
de l'utilisation de produits qui contiennent du 2-butoxyéthanol. La
concentration du 2-butoxyéthanol sur le lieu de travail est de l'ordre
du mg/m3.
Après exposition par la voie respiratoire, buccale ou percutanée,
le 2-butoxyéthanol est facilement résorbé. La métabolisation s'opère
essentiellement sous l'action de l'alcool- et de l'aldéhyde-
déshydrogénase et conduit à la formation de 2-butoxyacétaldéhyde et
d'acide 2-butoxyacétique, le principal métabolite. Il existe
toutefois d'autres voies métaboliques.
Le 2-butoxyéthanol présente une toxicité aiguë modérée et peut
irriter la peau et les yeux; il n'entraîne aucune sensibilisation
cutanée. Les principaux effets du 2-butoxyéthanol et de son principal
métabolite, l'acide 2-butoxyacétique, sont dus à l'hématotoxicité de
ces composés. Le rat est l'espèce la plus sensible. Les résultats
des études in vitro montrent que les hématies humaines ne sont pas
aussi sensibles que celles du rat aux effets hémolytique du
2-butoxyéthanol et de l'acide 2-butoxyacétique et que l'effet
hémolytique de ce dernier est plus prononcé. Chez le rat, l'action
toxique se manifeste aussi au niveau du système nerveux central, des
reins et du foie, mais à une concentration plus élevée que dans le cas
des effets hémolytiques. On n'a pas observé chez l'animal d'effets
toxiques sur la reproduction à des doses inférieures aux doses
toxiques. Les épreuves de mutagénicité in vitro ont donné des
résultats irréguliers mais en l'absence d'indices structuraux et
compte tenu des résultats négatifs obtenus in vivo, on peut avec une
confiance suffisante, considérer que le 2-butoxyéthanol n'est pas
mutagène. Les données limitées que l'on a pu tirer d'un certain
nombre de cas d'intoxication de même que les résultats d'une étude en
laboratoire, montrent que des effets analogues -- notamment des effets
hémolytiques et des effets au niveau du système nerveux central -- se
produisent chez l'homme comme chez le rat, mais à des concentrations
beaucoup plus élevées. Compte tenu des effets hémolytiques observés
chez des rattes gravides exposées pendant la période de gestation, on
a estimé à 13,1 mg/m3 la concentration tolérable pour l'homme.
En s'en tenant à des hypothèses extrêmement prudentes, on peut
considérer que la concentration estimative maximale de 2-butoxyéthanol
dans les eaux de surface très proches des effluents est susceptible de
dépasser parfois la valeur prévisible de la concentration maximale
sans effet observable. Toutefois, selon des hypothèses plus réalistes
fondées sur les données disponibles, il semblerait que ce composé ne
soit que faiblement toxique pour les organismes aquatiques. Comme la
demi-vie atmosphérique du 2-butoxyéthanol est brève, on estime que la
concentration mesurée ou calculée de cette substance dans l'air ne
pose pas de problème écologique.
RESUMEN DE ORIENTACION
El presente documento abreviado de evaluación internacional de
productos químicos (CICAD) sobre el 2-butoxietanol se basó en los
exámenes preparados por el Instituto Nacional para la Seguridad y
Salud del Trabajo (NIOSH, 1990) y la Agencia para el Registro de
Sustancias Tóxicas y Enfermedades (ATSDR, 1996). Se identificaron
datos adicionales en una investigación de publicaciones actualizada
hasta mayo de 1997, así como en el curso del examen por homólogos del
presente CICAD. En el apéndice 1 se halla información sobre la
naturaleza del examen por homólogos y la disponibilidad de los
documentos de origen. En el apéndice 2 se presenta información sobre
el análisis por homólogos del presente CICAD. Este CICAD fue aprobado
como evaluación internacional en una reunión de la Junta de Examen
Final, celebrada en Berlín (Alemania) los días 26-28 de noviembre de
1997. En el apéndice 3 se halla la lista de los participantes en la
reunión de la Junta de Examen Final. También se ha reproducido en el
presente documento la ficha internacional de seguridad química (ICSC
0059) producida por el Programa Internacional de Seguridad de las
Sustancias Químicas (IPCS, 1993).
El 2-butoxietanol (CAS N° 111-76-2) es un éter glicólico de alto
volumen de producción. Es un líquido incoloro miscible en agua y
soluble en la mayor parte de los disolventes orgánicos. El
2-butoxietanol se utiliza ampliamente como disolvente en
revestimientos de superficies, y en lacas en nebulización, lacas de
secado rápido, esmaltes, barnices, eliminadores de barnices y pintura
látex. También se utiliza en productos limpiadores de metales y
domésticos. El 2-butoxietanol existe en la atmósfera casi totalmente
en forma de vapor; dado que el producto químico tiene una semivida
atmosférica de unas 17 horas, el riesgo de transporte por la atmósfera
debe ser pequeño. La semivida estimada del 2-butoxietanol en agua es
aproximadamente de 1-4 semanas; probablemente experimenta una
biodegradación rápida en el suelo aerobio y en el agua. La capacidad
de acumulación es baja. Basándose en datos limitados puede indicarse
que la exposición ambiental en el aire se halla en general en la gama
de µg/m3. La exposición indirecta de la población general al
2-butoxietanol se produce muy probablemente por inhalación y absorción
cutánea durante el empleo de productos que contienen la sustancia
química. Las concentraciones del 2-butoxietanol en el aire en
entornos laborales se hallan típicamente en la gama de mg/m3.
El 2-butoxietanol se absorbe fácilmente después de la exposición
por inhalación o por vías oral y cutánea. El producto químico es
metabolizado principalmente por la deshidrogenasa de alcoholes y
aldehídos, con formación de 2-butoxiacetaldehído y de ácido
2-butoxiacético, el principal metabolito, aunque también se han
identificado otras vías metabólicas.
El 2-butoxietanol presenta una moderada toxicidad aguda y es
irritante para los ojos y la piel; no es un sensibilizador cutáneo.
El principal efecto del 2-butoxietanol y de su metabolito, el ácido
2-butoxiacético, es la hematotoxicidad, siendo la rata la especie más
sensible. Los resultados de estudios in vitro muestran que los
eritrocitos humanos no son tan sensibles como los eritocitos de rata a
los efectos hemolíticos del 2-butoxietanol y del ácido
2-butoxiacético, y también que los eritrocitos son más sensibles a la
hemólisis por el ácido 2-butoxiacético que por el 2-butoxietanol. En
la rata, los efectos adversos sobre el sistema nervioso central, los
riñones y el hígado se producen con concentraciones de exposición más
altas que los efectos hemolíticos. En animales no se han observado
efectos adversos sobre la reproducción y el desarrollo con dosis
inferiores a las tóxicas. Aunque los resultados de las pruebas
in vitro de mutagenicidad del 2-butoxietanol son incoherentes, la
ausencia de elementos estructurales de alerta y los resultados
negativos de los estudios in vivo son suficientemente alentadores
para permitir llegar a la conclusión de que el 2-butoxietanol no es
mutagénico. Basándose en datos limitados procedentes de estudios de
casos y de un estudio de laboratorio, se han señalado efectos agudos
análogos (incluidos efectos hemolíticos y otros sobre el sistema
nervioso central) en personas y ratas expuestas al 2-butoxietanol,
aunque los efectos se observaron con concentraciones de exposición
mucho más altas en personas que en ratas. Basándose en la aparición
de efectos hemolíticos en ratas grávidas expuestas durante la
gestación, se ha deducido una concentración tolerable de muestra para
las personas de 13,1 mg de 2-butoxietanol/m3.
Sobre la base de supuestos extremadamente conservadores, las
concentraciones previstas máximas de 2-butoxietanol en aguas
superficiales situadas cerca de corrientes de efluentes pueden, en
algunos casos, exceder de las concentraciones previstas de efectos no
observados. Sin embargo, supuestos más realistas basados en los datos
disponibles permiten indicar que el riesgo para los seres acuáticos es
escaso. Debido a la corta semivida del 2-butoxietanol en la
atmósfera, las concentraciones medidas o previstas de este producto
químico en el aire se consideran exentas de importancia ambiental.
11.2.2 Terrestrial environment
Data are inadequate to characterize the risks to terrestrial
organisms of exposure to 2-butoxyethanol. A PEClocal(air) of 537 µg/m3,
based upon the use patterns of this chemical in Australia, has been
reported (OECD, 1997). Although available monitoring data are
limited, this predicted concentration is much higher than levels
measured in ambient air (see section 6). As 2-butoxyethanol is
expected to have a half-life in the atmosphere of less than 1 day,
these concentrations are considered to have no environmental
significance.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Previous evaluations of 2-butoxyethanol published by WHO, the
International Agency for Research on Cancer (IARC), the Joint FAO/WHO
Expert Committee on Food Additives (JECFA), or the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) were not identified. A
Screening Information Dataset (SIDS) Initial Assessment Report has
been prepared under the Organisation for Economic Co-operation and
Development (OECD) High Production Volume (HPV) Chemicals Programme
(OECD, 1997). Information on international hazard classification and
labelling is included in the International Chemical Safety Card that
has been reproduced in this document.
13. HUMAN HEALTH PROTECTION AND EMERGENCY ACTION
Human health hazards, together with preventative and protective
measures and first aid recommendations, are presented in the
International Chemical Safety Card (ICSC 0059) reproduced in this
document.
13.1 Human health hazards
2-Butoxyethanol is toxic to humans. Following long-term or
repeated exposure, effects on the blood may be observed.
13.2 Advice to physicians
In case of intoxication, immediate supportive measures should be
given, as central nervous system depression, respiratory paralysis,
hypotension, and metabolic acidosis have been observed in the few
hours post-exposure. Close monitoring for renal toxicity and possible
haemodialysis are mandatory in the subsequent days (renal
insufficiency may develop 2-3 days post-exposure) until recovery is
achieved, on average by the second week post-exposure.
13.3 Health surveillance advice
Periodic medical examination of the haematopoietic system should
be included in a health surveillance programme.
13.4 Spillage
As 2-butoxyethanol is toxic and absorbed through the skin,
emergency crews need to wear proper equipment, including a mask with
cartridge for organic vapour, for handling spills. The chemical
should not be allowed to enter drains or watercourses.
14. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Information on national regulations, guidelines, and standards
can be found in the International Register of Potentially Toxic
Chemicals (IRPTC), available from UNEP Chemicals (IRPTC), Geneva.
The reader should be aware that regulatory decisions about
chemicals taken in a certain country can be fully understood only in
the framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
INTERNATIONAL CHEMICAL SAFETY CARD
2-BUTOXYETHANOL ICSC: 0059
26.04.193
CAS # 111-76-2 Ethylene glycol monobutyl ether
RTECS # KJ8575000 Monobutyl glycol ether
UN # 2369 C6H14O2/CH3(CH2)2CH2OCH2CH2OH
EC # 603-014-00-0 Molecular mass: 118.2
TYPES OF HAZARD/ ACUTE HAZARDS/ PREVENTION FIRST AID/FIRE FIGHTING
EXPOSURE SYMPTONS
FIRE Combustible. NO open flames. NO contact with Powder, alcohol-resitant foam, water spary,
oxidizing agents. carbon dioxide.
EXPLOSION Above 61°C explosive vapour/air Above 61°C closed system, In case of fire: keep drums, etc., cool by
mixtures may be formed. ventilation. spraying with water.
EXPOSURE PREVENT GENERATION OF IN ALL CASES CONSULT A DOCTOR!
MISTS!
Inhalation Cough, drowsiness, headache, Ventilation, local exhaust, or Fresh air, rest, and refer for medical
nausea. breathing protection. attention.
Skin MAY BE ABSORBED! Dry skin Protective gloves, protective Remove contaminated clothes, rinse skin with
(further see Inhalation). clothing. plenty of water or shower, and refer for
medical attention.
Eyes Redness, pain, blurred vision. Safety goggles or eye protection First rinse with plenty of water for several
combination with breathing minutes (remove contact lenses if easily
protection. possible), then take to a doctor.
(continued)
TYPES OF HAZARD/ ACUTE HAZARDS/ PREVENTION FIRST AID/FIRE FIGHTING
EXPOSURE SYMPTONS
Ingestion Abdominal pain, diarrhoea, Do not eat, drink, or smoke during Rinse mouth, give plenty of water to drink,
nausea, vomiting (further see work. induce vomiting (ONLY IN CONSCIOUS PERSONS),
inhalation). and refer for medical attention.
SPILLAGE DISPOSAL PACKAGING & LABELLING
Collect leaking and spilled liquid in sealable containers as far as possible, Xn Symbol
wash away remainder with plenty of water. R: 20/21/22-37
S: 24/25
UN Hazard Class: 6.1
UN Subsidiary Risks:
UN Pack Group: III
Airtight. Do not transport with food and feedstuffs.
EMERGENCY RESPONSE STORAGE
NFPA Code: H 2; F 2; R 0 Separated from strong oxidants, food and feedstuffs;
keep in the dark.
IMPORTANT DATA
PHYSICAL STATE: APPEARANCE: ROUTES OF EXPOSURE:
COLOURLESS LIQUID, WITH CHARACTERISTIC ODOUR. The substance can be absorbed into the body by inhalation
and through the skin, and by ingestion.
CHEMICAL DANGERS:
The substance can form explosive peroxides. The substance INHALATION RISK:
decomposes, producting toxic fumes. Reacts with strong A harmful contamination of the air will be reached rather
oxidants, causing fire and explosion hazard. slowly on evaporation of this substance at 20°C.
(continued)
OCCUPATIONAL EXPOSURE LIMITS: EFFECTS OF SHORT-TERM EXPOSURE:
TLV: 25 ppm; 121 mg/m3 (as TWA) (skin) (ACGIH 1992-1993) The substance irritates the eyes, the skin, and the
respiratory tract. Exposure could cause central nervous
system depression and liver and kidney damage.
EFECTS OF LONG-TERM OR REPEATED EXPOSURE:
The liquid defats the skin. The substance may have effects
on the haematopoietic system, resulting in blood disorders.
PHYSICAL PROPERTIES
Boiling point: 171°C Flash point: (c.c.) 61°C
Melting point: -75°C Auto-ignition temperature: 238°C
Relative density (water = 1) 0.90 Explosive limits, vol% in air: 1.1-12.7
Solubility in water: miscible Octanol/water partition coefficient as log Pow: 0.830
Vapour pressure, kPa at 20°C: 0.10
Relative vapour density (air = 1); 4.1
Relative density of the vapour/air-mixture at 20°C (air=1): 1.00
ENVIRONMENTAL DATA
This substance may be hazardous to the environment; speical attention should be given to the water environment and aquifer.
NOTES
Depending on the degree of exposure, periodic medical examination is indicated. Check for peroxides prior to distillation,
render harmless if positive. Keep in dark because of possible formation of explosive peroxides.
ADDITIONAL INFORMATION
LEGAL NOTICE: Neither the CEC or the IPCS no any person acting on behalf of the CEC or the IPCS is responsible for the use
which might be made of this information
(c) IPCS CEC 1996
REFERENCES
Amoore JE, Hautala E (1983) Odor as an aid to chemical safety: odor
threshold compared with threshold limit values and volatilities for
214 industrial chemicals in air and water dilution. Journal of
applied toxicology, 36(6):272-290.
Angerer J, Lichterbeck E, Begerow J, Jekel S, Lehnert G (1990)
Occupational chronic exposure to organic solvents: XIII. Glycol ether
exposure during the production of varnishes. International archives
of occupational and environmental health, 62(2):123-126.
ATSDR (1996) Toxicological profile for 2-butoxyethanol and
2-butoxyethanol acetate (August 1996 draft). Atlanta, GA, US
Department of Health and Human Services, Public Health Service, Agency
for Toxic Substances and Disease Registry.
Bartnik FG, Reddy AK, Klecak G, Zimmermann V, Hostynek JJ, Kunstler K
(1987) Percutaneous absorption, metabolism, and hemolytic activity of
n-butoxyethanol. Fundamental and applied toxicology, 8:59-70.
Bauer PH, Weber M, Mur JM, Protois JC, Bollaert PE, Condi A, Larcan A,
Lambert H (1992) Transient noncarcinogenic pulmonary edema following
massive ingestion of ethylene glycol butyl ether. Intensive care
medicine, 18:250-251.
Bormett GA, Bartels MJ, Markham DA (1995) Determination of
2-butoxyethanol and butoxyacetic acid in rat and human blood by gas
chromatography-mass spectrometry. Journal of chromatography,
B665:315-325.
Bridie AL (1979) The acute toxicity of some petrochemicals to fish.
Water research, 13:623-626.
Bringmann G, Kuhn R (1977) Results of the damaging effects of water
pollutants on Daphnia magna. Zeitschrift fuer Wasser und Abwasser
Forschung, 10:161-166.
Bringmann G, Kuhn R (1980a) Comparison of the toxicity thresholds of
water pollutants to bacteria, algae, and protozoa in the cell
multiplication test. Water research, 14:231-241.
Bringmann G, Kuhn R (1980b) Determination of the toxicity of water
pollutants to protozoa. II. Bacteriovorous ciliates. Zeitschrift
fuer Wasser und Abwasser Forschung, 13:26-31.
Bringmann G, Kuhn R (1982) Results of the toxic action of water
pollutants on Daphnia magna Straus tested by an improved
standardized procedure. Zeitschrift fuer Wasser und Abwasser
Forschung, 15:1-6.
Canadian Chemical Producers' Association (1996) Reducing emissions.
A Responsible Care initiative. 1994 emissions inventory and five
year projections. Ottawa, 36 pp.
Carpenter CP, Pozzani UC, Woil CS, Nair JH, Keck GA, Smyth HF Jr
(1956) The toxicity of butyl cellosolve solvent. American Medical
Association Archives of industrial health, 14:114-131.
CEFIC (1995) CEFIC Document 486/95/7/stat/year, Oxygenated Solvents
S.G. -- Statistical Investigation 1994 (23 February 1995). Brussels,
European Chemical Industry Council [cited in OECD, 1997].
Chiewchanwit T, Au WW (1995) Mutagenicity and cytotoxicity of
2-butoxyethanol and its metabolite, 2-butoxyacetaldehyde, in Chinese
hamster (CHO-AS52) cells. Mutation research, 334(13):341-346.
Ciccioli P, Brancaleoni E, Cecinato A, Sparapani R, Frattoni M (1993)
Identification and determination of biogenic and anthropogenic
volatile organic compounds in forest areas of northern and southern
Europe and a remote site of the Himalaya region by high-resolution gas
chromatography-mass spectrometry. Journal of chromatography,
643:55-69.
Ciccioli P, Cecinato A, Brancaleoni E, Frattoni M, Bruner F, Maione M
(1996) Occurrence of oxygenated volatile organic compounds (VOC) in
Antarctica. International journal of analytical chemistry,
62:245-253.
CMA (1994) HEDSET for ethylene glycol butyl ether. Prepared for the
European Union Existing Substances Programme. Washington, DC, Chemical
Manufacturers Association.
Corley RA, Bormett GA, Ghanayem BI (1994) Physiologically-based
pharmacokinetics of 2-butoxyethanol and its major metabolite,
2-butoxyacetic acid, in rats and humans. Toxicology and applied
pharmacology, 129(1):61-79.
Corley RA, Markham DA, Banks C, Delorme P, Masterman A, Houle JM
(1997) Physiologically-based pharmacokinetics and the dermal
absorption of 2-butoxyethanol vapor by humans. Fundamental and
applied toxicology, 39:120-130.
Dawson GW, Jennings AL, Drozdowski D, Rider E (1977) The acute
toxicity of 47 industrial chemicals to fresh and saltwater fishes.
Journal of hazardous materials, 1:303-318.
Dean BS, Krenzelok EP (1992) Clinical evaluation of pediatric ethylene
glycol monobutyl ether poisonings. Journal of toxicology and
clinical toxicology, 30(4):557-563.
Dill DC (1995) Environmental summary for Dowanol EB and DB glycol
ethers. Unpublished report. Midland, MI, Dow Chemical Company.
Dodd DE, Snelling WM, Maronpot RR, Ballentyne B (1983) Ethylene glycol
monobutyl ether: acute 9-day and 90-day vapor inhalation studies in
Fischer 344 rats. Toxicology and applied pharmacology, 68:405-414.
Doe JE (1984) Further studies on the toxicology of the glycol ethers
with emphasis on rapid screening and hazard assessment.
Environmental health perspectives, 57:199-206.
Dow (1979) Toxicity of Dowanol EB to freshwater organisms.
Unpublished report. Midland, MI, Dow Chemical Company (Report No.
ES-330).
Dow (1988) Dowanol EB glycol ether: evaluation of the toxicity to
the green alga, Selenastrum capricornutum. Unpublished report.
Midland, MI, Dow Chemical Company.
ECETOC (1994) Butoxyethanol criteria document. Including a
supplement for 2-butoxyethyl acetate. Brussels, European Chemical
Industry, Ecology and Toxicology Centre (Special Report No. 7, April
1994).
Elias Z, Daniere MC, Marande AM, Poirot O, Terzelti F, Schneider O
(1996) Genotoxic and/or epigenetic effects of some glycol ethers:
results of different short-term tests. Occupational hygiene,
2:187-212.
Elliott BM, Ashby J (1997) Review of the genotoxicity of
2-butoxyethanol. Mutation research, 387:89-96.
Environment Canada (1997) Canadian Environmental Protection Act.
Priority Substances List Supporting Documentation --
2-Butoxyethanol. Vol. 2 (draft). Ottawa, pp. 187-212.
Exon JH, Mather GG, Bussiere JL, Olson DP, Talcott PA (1991) Effects
of subchronic exposure of rats to 2-methoxyethanol or 2-butoxyethanol:
thymic atrophy and immunotoxicity. Fundamental and applied
toxicology, 16(4):830-840.
Foster PMD, Lloyd SC, Blackburn DM (1987) Comparison of the in vivo
and in vitro testicular effects produced by methoxy-, ethoxy- and
n-butoxy acetic acids in the rat. Toxicology, 43:17-30.
Ghanayem BI (1989) Metabolic and cellular basis of 2-butoxyethanol-
induced hemolytic anemia in rats and assessment of human risk in
vitro. Biochemical pharmacology, 38(10):1679-1684.
Ghanayem BI, Sullivan CA (1993) Assessment of the haemolytic activity
of 2-butoxyethanol and its major metabolite, butoxyacetic acid, in
various mammals including humans. Human experimental toxicology,
1214:305-311.
Ghanayem BI, Blair PC, Thompson MB, Maronpot RR, Matthews HB (1987a)
Effect of age on the toxicity and metabolism of ethylene glycol
monobutyl ether (2-butoxyethanol) in rats. Toxicology and applied
pharmacology, 91:222-234.
Ghanayem BI, Burka LT, Sanders JM, Matthews H (1987b) Metabolism and
disposition of ethylene glycol monobutyl ether (2-butoxyethanol) in
rats. Drug metabolism and disposition, 15:478-484.
Ghanayem BI, Ward SM, Blair PC, Matthews HB (1990) Comparison of the
hematologic effects of 2-butoxyethanol using two types of hematology
analyzers. Toxicology and applied pharmacology, 106(2):341-345.
Ghanayem BI, Sanchez IM, Matthews HB (1992) Development of tolerance
to 2-butoxyethanol-induced hemolytic anemia and studies to elucidate
the underlying mechanisms. Toxicology and applied pharmacology,
112(2):198-206.
Gijsenbergh FP, Jenco M, Veulemans H, Groesenken D, Verberckmoes R,
Delooz HH (1989) Acute butyl-glycol intoxication: a case report.
Human toxicology, 8:243-245.
Gingell R, Boatman RJ, Lewis S (1997) Comparative acute toxicity
of ethylene glycol mono-n-butyl ether in several species. Arlington,
VA, Chemical Manufacturers Association.
Gollapudi BB, Barber ED, Lawlor TE, Lewis SA (1996) Re-examination of
the mutagenicity of ethylene glycol monobutyl ether to Salmonella
tester strain TA97a. Mutation research, 370:61-64.
Grant D, Slush S, Jones HB, Gangolli SD, Butler WH (1985) Acute
toxicity and recovery in the hemopoietic system of rats after
treatment with ethylene glycol monomethyl and monobutyl ethers.
Toxicology and applied pharmacology, 77:187-200.
Greenspan AH, Reardon RC, Gingell R, Rosica KA (1995) Human repeated
insult patch test of 2-butoxyethanol. Contact dermatitis, 33:59-60.
Groeseneken D, Van Vlem E, Veulemans H, Masschelein R (1986) Gas
chromatographic determination of methoxyacetic and ethoxyacetic acid
in urine. British journal of industrial medicine, 43:62-65.
Groeseneken D, Veulemans H, Masschelein R, Van Vlem E (1989) An
improved method for the determination in urine of alkoxyacetic acids.
International archives of occupational and environmental health,
61:249-254.
Gualtieri J, Harris C, Roy R, Corley R, Manderfield C (1995) Multiple
2-butoxyethanol intoxications in the same patient: clinical findings,
pharmacokinetics, and therapy. Journal of toxicology and clinical
toxicology, 33(5):550-551.
Hardin BD, Goad PT, Burg JR (1984) Developmental toxicity of four
glycol ethers applied cutaneously to rats. Environmental health
perspectives, 57:69-74.
Heindel JJ, Lamb JC IV, Chapin RE, Gulati DK, Hope E, George J,
Jameson CW, Teague J, Schwetz BA (1989) Reproductive toxicity
testing by continuous breeding: Test protocol in Swiss (CD-1) mice.
Available from National Technical Information Service, Springfield, VA
(NTIS No. PB89152451AS).
Heindel JJ, Gulati DK, Russell VS, Reel JR, Lawton AD, Lamb JC IV
(1990) Assessment of ethylene glycol monobutyl and monophenyl ether
reproductive toxicity using a continuous breeding protocol in Swiss
CD-1 mice. Fundamental and applied toxicology, 15(4):683-696.
Hoflack JC, Lambolez L, Elias Z, Vasseur P (1995) Mutagenicity of
ethylene glycol ethers and of their metabolites in Salmonella
typhimurium his-. Mutation research, 341(4):281-287.
Howard PH, Boethling RS, Jarvis WF, Meylan WM, Michalenko EM (1991)
Handbook of environmental degradation rates. Chelsea, MI, Lewis
Publishers Inc. [cited in ATSDR, 1996].
US NLM (1997) Hazardous substances data bank. Last revision on
7/11/96. Bethesda, MD, National Library of Medicine, National
Toxicology Information Program.
IPCS (1993) International Chemical Safety Card -- 2-Butoxyethanol.
Geneva, World Health Organization, International Programme on Chemical
Safety (No. 0059).
Johanson G (1994) Inhalation toxicokinetics of butoxyethanol and its
metabolite butoxyacetic acid in the male Sprague-Dawley rat.
Archives of toxicology, 68(9):588-594.
Johanson G, Boman A (1991) Percutaneous absorption of 2-butoxyethanol
vapor in human subjects. British journal of industrial medicine,
48(11):788-792.
Johanson G, Kronborg H, Naslund PH, Nordqvist MB (1986) Toxicokinetics
of inhaled 2-butoxyethanol (ethylene glycol monobutyl ether) in man.
Scandinavian journal of work and environmental health,12:594-602.
Johanson G, Boman A, Dynesius B (1988) Percutaneous absorption of
2-butoxyethanol in man. Scandinavian journal of work and
environmental health, 14:101-109.
Jonsson AK, Steen G (1978) n-Butoxyacetic acid, a urinary metabolite
from inhaled n-butoxyethanol (butylcellosolve). Acta
Pharmacologica et Toxicologica, 42:354-356.
Junke I, Ludemann D (1978) Results of the examination of the effects
of 200 chemical compounds on fish toxicity using the golden orfe test.
Zeitschrift fuer Wasser und Abwasser Forschung, 11:161-164.
Keith G, Coulais C, Edoh A, Botlin MC, Rihn B (1996) Ethylene glycol
monobutyl ether has neither epigenetic nor genotoxic effects in acute
treated rats and in subchronic treated v-Ha- ras transgenic mice.
Occupational hygiene, 2:237-249.
Kennah HE II, Hignet S, Laux PE, Dorko JD, Barrow CS (1989) An
objective procedure for quantitating eye irritation based upon changes
of corneal thickness. Fundamental and applied toxicology,
12(2):258-268.
Kennedy ER, O'Connor PF, Grote AA (1990) Application of
multidimensional gas chromatography-mass spectrometry to the
determination of glycol ethers in air. Journal of chromatography,
522:303-333.
Koenemann H (1981) Quantitative structure-activity relationships in
fish toxicity studies. Part 1. Relationships for 50 industrial
pollutants. Toxicology, 19:209-221.
Krasavage WJ (1986) Subchronic oral toxicity of ethylene glycol
monobutyl ether in male rats. Fundamental and applied toxicology,
6:349-355.
Leaf DA (1985) Glycol ethers: an overview. Washington, DC, US
Environmental Protection Agency, Office of Pesticides and Toxic
Substances.
McGregor DB (1984) The genotoxicity of glycol ethers. Environmental
health perspectives, 57:97-103.
Medinsky MA, Singh G, Bechtold WE, Bond JA, Sabourin PJ, Birnbaum LS,
Henderson RF (1990) Disposition of three glycol ethers administered in
drinking water to male F344/N rats. Toxicology and applied
pharmacology, 102(3):443-455.
Nagano K, Nakayama E, Koyano M, Oobayashi H, Adachi H, Yamada T (1979)
Mouse testicular atrophy induced by ethylene glycol monoalkyl ethers.
Japanese journal of industrial health, 21:29-35.
Nelson BR, Setzer JV, Brightwell WS, Mathinos PR, Kuczuk MH, Weaver
TE, Goad PT (1984) Comparative inhalation teratogenicity of four
glycol ether solvents and an amino derivative in rats.
Environmental health perspectives, 57:261-271.
NIOSH (1983) National Occupational Exposure Survey (NOES), 1981-83:
estimated total and female employees, actual observation and
trade-named exposure to EGEE, EGHE, EGBE, and their acetates.
Unpublished database. Cincinnati, OH, US Department of Health and
Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, Division of
Surveillance, Hazard Evaluations, and Field Studies, Surveillance
Branch.
NIOSH (1990) Criteria for a recommended standard. Occupational
exposure to ethylene glycol monobutyl ether and ethylene glycol
monobutyl ether acetate. Cincinnati, OH, US Department of Health and
Human Services, Public Health Service, Centers for Disease Control,
National Institute for Occupational Safety and Health, Division of
Standards Development and Technology Transfer (DHHS [NIOSH]
Publication No. 90-118).
NIOSH (1994) Manual of analytical methods, 4th ed. Cincinnati, OH,
US Department of Health and Human Services, Public Health Service,
Centers for Disease Control and Prevention, National Institute for
Occupational Safety and Health (DHHS [NIOSH] Publication No. 94-113).
NTP (1993) NTP technical report on toxicity studies of ethylene
glycol ethers 2-methoxyethanol, 2-ethoxyethanol, 2-butoxyethanol
administered in drinking water to F344/N rats and B6C3F1 mice.
Research Triangle Park, NC, US Department of Health and Human
Services, Public Health Service, National Institutes of Health,
National Toxicology Program (NIH Publication No. 93-3349).
OECD (1997) Screening Information Dataset (SIDS) initial assessment
report on 2-butoxyethanol. 6th SIDS Initial Assessment Meeting.
Paris, Organisation for Economic Co-operation and Development.
OSHA (1990) 2-Butoxyethanol (butyl cellosolve) and 2-butoxyethyl
acetate (butyl cellosolve acetate). Salt Lake City, UT, US
Department of Labor, Occupational Safety and Health Administration,
Organic Methods Evaluation Branch, OSHA Analytical Laboratory.
Price KS, Waggy GT, Conway RA (1974) Brine shrimp bioassay and
seawater BOD of petrochemicals. Journal of the Water Pollution
Control Federation, 46:63-77.
Rambourg-Schepens MD, Buffet M, Bertault R, Jaussaud M, Journe B, Fay
R, Lamiable D (1988) Severe ethylene glycol butyl ether poisoning.
Kinetics and metabolic pattern. Human toxicology, 7:187-189.
Rettenmeier AW, Hennigs R, Wodarz R (1993) Determination of
butoxyacetic acid and n-butoxyacetylglutamine in urine of lacquerers
exposed to 2-butoxyethanol. International archives of occupational
and environmental health, 65(1) (Suppl.):S151-S153.
Rowe VK, Wolf MA (1982) Derivatives of glycols. In: Clayton GD,
Clayton EF, eds. Patty's industrial hygiene and toxicology, 3rd rev.
ed. Vol. 2. New York, NY, John Wiley and Sons, pp. 3909-4052.
Sakai T, Araki T, Masuyama Y (1993) Determination of urinary
alkoxyacetic acid by rapid and simple method for biological monitoring
of workers exposed to glycol ethers and their acetates.
International archives of occupational and environmental health,
64:495-498.
Sakai T, Araki T, Morita Y, Masuyama Y (1994) Gas chromatographic
determination of butoxyacetic acid after hydrolysis of conjugated
metabolites in urine from workers exposed to 2-butoxyethanol.
International archives of occupational and environmental health,
66:249-254.
Sax NI, Lewis RJ (1987) Hawley's condensed chemical dictionary, 11th
ed. New York, NY, Van Nostrand Reinhold Company, pp. 488-489.
Schuler RL, Hardin BD, Niemeier RW, Booth G, Hazelden K, Piccirillo V
(1984) Results of testing 15 glycol ethers in a short-term in vivo
reproductive toxicity assay. Environmental health perspectives,
57:141-146.
Shyr LJ, Sabourin PJ, Medinsky MA, Birnbaum LS, Henderson RF (1993)
Physiologically-based modeling of 2-butoxyethanol disposition in rats
following different routes of exposure. Environmental research,
63(2):202-218.
Smallwood AW, DeBord KE, Lowry LK (1984) Analyses of ethylene glycol
monoalkyl ethers, and their proposed metabolites in blood and urine.
Environmental health perspectives, 57:249-253.
Smallwood AW, DeBord KE, Burg J, Moseley C, Lowry LK (1988)
Determination of urinary 2-ethoxyacetic acid as an indicator of
occupational exposure to 2-ethoxyethanol. Applied industrial
hygiene, 3(2):47-50.
Smialowicz RJ, Williams WC, Riddle HH, Andres DL, Luebke RW, Copeland
CB (1992) Comparative immunosuppression of various glycol ethers
orally administered to Fischer 344 rats. Fundamental and applied
toxicology, 18(4):621-627.
Sohnlein B, Letzel S, Weltle D, Rüdiger HW, Angerer J (1993) XIV.
Examinations concerning the evaluation of a limit value for
2-ethoxyethanol and 2-ethoxyethylacetate and the genotoxic effects of
these glycol ethers. International archives of occupational and
environmental health, 64(7):479-484.
Staples CA, Boatman RJ, Cano ML (1998) Ethylene glycol ethers: An
environmental risk assessment. Chemosphere, 36:1585-1613.
Tyl RW, Millicovsky G, Dodd DE, Pritts IM, France KA, Fisher LC (1984)
Teratologic evaluation of ethylene glycol monobutyl ether in Fischer
344 rats and New Zealand white rabbits following inhalation exposure.
Environmental health perspectives, 57:47-68.
Tyler TR (1984) Acute and subchronic toxicity of ethylene glycol
monobutyl ether. Environmental health perspectives, 57:85-191.
Udden MM (1994) Hemolysis and deformability of erythrocytes exposed to
butoxyacetic acid, a metabolite of 2-butoxyethanol: II. Resistance in
red blood cells from humans with potential susceptibility. Journal
of applied toxicology, 14(2):97-102.
Udden MM (1996) Effects of butoxyacetic acid on human red cells.
Occupational hygiene, 2:283-290.
Udden MM, Patton CS (1994) Hemolysis and deformability of erythrocytes
exposed to butoxyacetic acid, a metabolite of 2-butoxyethanol: I.
Sensitivity in rats and resistance in normal humans. Journal of
applied toxicology, 412:91-96.
Union Carbide (1989) Ecological fate and effects data on four
selected glycol ether products. Unpublished report. South
Charleston, WV, Union Carbide Chemicals and Plastic Co. Inc.
US EPA (1984) Acute toxicity studies on Wellaid 31. Study submitted
to the US Environmental Protection Agency by Amoco Corporation [cited
in OECD, 1997].
US ITC (1996) Preliminary report on U.S. production of selected
synthetic organic chemicals (including synthetic plastics and
resin materials). Fourth Quarter and Preliminary International Trade
Commission, Series C/P-96-2; No. 26, pp. 2-12. Totals, 1995.
Washington, DC, US International Trade Commission.
Verschueren K (1983) Handbook of environmental data on organic
chemicals, 2nd ed. New York, NY, Van Nostrand Reinhold Company, 1310
pp.
Veulemans H, Groeseneken D, Masschelein R, Van Vlem E (1987) Survey of
ethylene glycol ether exposures in Belgian industries and workshops.
American Industrial Hygiene Association journal, 48(8):671-676.
Vincent R, Cicolella A, Subra I, Rieger B, Parrot P, Pierre F (1993)
Occupational exposure to 2-butoxyethanol for workers using window
cleaning agents. Applied occupational and environmental hygiene,
8(6):580-586.
von Oettingen WF, Jirouche EA (1931) The pharmacology of ethylene
glycol and some of its derivatives in relation to their chemical
constitution and physical chemical properties. Journal of
pharmacology and experimental therapeutics, 42(3):355-372.
Werner HW, Mitchell JL, Miller JW, von Oettingen WF (1943a) The acute
toxicity of vapors of several monoalkyl ethers of ethylene glycol.
Journal of industrial hygiene and toxicology, 25:157-163.
Werner HW, Nawrocki CZ, Mitchell JL, Miller JW, von Oettingen WF
(1943b) Effects of repeated exposure of rats to monoalkyl ethylene
glycol ether vapors. Journal of industrial hygiene and toxicology,
25:374-379.
Werner HW, Mitchell JL, Miller JW, von Oettingen WF (1943c) Effects of
repeated exposure of dogs to monoalkyl ethylene glycol ether vapors.
Journal of industrial hygiene and toxicology, 25:409-414.
Yasuhara A, Shiraisi H, Tsuji M, Okuno T (1981) Analysis of organic
substances in highly polluted river water by mass spectrometry.
Environmental science and technology, 15:570-573.
Zeiger E, Anderson B, Haworth S, Lawlor T, Mortelmans K (1992)
Salmonella mutagenicity tests: V. Results from the testing of 311
chemicals. Environmental and molecular mutagenesis, 19 (Suppl.
21):2-141.
Zissu D (1995) Experimental study of cutaneous tolerance to glycol
ethers. Contact dermatitis, 32(2):74-77.
APPENDIX 1 -- SOURCE DOCUMENTS
NIOSH (1990)
Copies of this source document ( Criteria for a recommended
standard. Occupational exposure to ethylene glycol monobutyl ether
and ethylene glycol monobutyl ether acetate; NIOSH Publication No.
90-118) are available from:
Publications Office
National Institute for Occupational Safety and Health
4676 Columbia Parkway
Cincinnati, OH 45226
USA
(513) 533-8471
This document was prepared by Joann Wess and reviewed internally
by staff of the National Institute for Occupational Safety and Health.
The draft document was reviewed externally by Dr F. Mirer, United Auto
Workers; Mr M. Gillen, Workers' Institute for Safety and Health; Mr F.
Burkhardt, International Brotherhood of Builders and Allied Trades; Dr
J. McCuen, ARCO Chemical Company; Mr W. Lypka, Graphic Communications
International Union; Dr H. Veulemans, Laboratorium voor
arbeidshygienne en-toxicologie; Dr E.M. Johnson, Jefferson Medical
College; Dr J.V. Rodricks, Dr J.S. Ferguson, Dr R.M. Putzrath, Mr M.
Fitzgerald, Chemical Manufacturers Association; Dr L. Welch, George
Washington University; Dr P. Sharma, Utah State University; Dr R.
Elves, Department of the Air Force; and Dr F. Welsch, Chemical
Industry Institute of Toxicology.
ATSDR (1996)
Copies of the ATSDR's Toxicological profile for
2-butoxyethanol and 2-butoxyethanol acetate (draft for public
comment) may be obtained from:
Agency for Toxic Substances and Disease Registry
Division of Toxicology
1600 Clifton Road NE, E-29
Atlanta, GA 30333
USA
This ATSDR draft document has undergone internal ATSDR review.
The document has also been reviewed by an expert panel of
nongovernmental reviewers consisting of the following members: Dr W.
Decker, Private Consultant, El Paso, TX; Dr A. Gregory, Private
Consultant, Sterling, VA; and Dr R. Rubin, Johns Hopkins School of
Public Health, Baltimore, MD.
APPENDIX 2 -- CICAD PEER REVIEW
The draft CICAD on 2-butoxyethanol was sent for review to
institutions and organizations identified by IPCS after contact with
IPCS national Contact Points and Participating Institutions, as well
as to identified experts. Comments were received from:
BASF, Ludwigshafen, Germany
Chemical Manufacturers Association, Arlington, USA
Department of Health, London, United Kingdom
Environment Canada, Ottawa, Canada
Health and Safety Executive, Liverpool, United Kingdom
Health Canada, Ottawa, Canada
Ministry of Health and Welfare, Government of Japan, Tokyo, Japan
National Chemicals Inspectorate (KEMI), Solna, Sweden
National Institute of Occupational Health, Budapest, Hungary
National Institute of Public Health and Environmental Protection,
Bilthoven, The Netherlands
National Occupational Health & Safety Commission, Sydney,
Australia
Oxygenated Solvents Producers Association, Brussels, Belgium
United States Department of Health and Human Services (National
Institute of Environmental Health Sciences, Research Triangle
Park)
United States Environmental Protection Agency (National Center
for Environmental Assessment, Washington, DC)
APPENDIX 3 -- CICAD FINAL REVIEW BOARD
Berlin, Germany, 26-28 November 1997
Members
Dr H. Ahlers, Education and Information Division, National Institute
for Occupational Safety and Health, Cincinnati, OH, USA
Mr R. Cary, Health Directorate, Health and Safety Executive, Bootle,
United Kingdom
Dr S. Dobson, Institute of Terrestrial Ecology, Huntingdon, United
Kingdom
Dr R.F. Hertel, Federal Institute for Health Protection of Consumers &
Veterinary Medicine, Berlin, Germany (Chairperson)
Mr J.R. Hickman, Health Protection Branch, Health Canada, Ottawa,
Ontario, Canada
Dr I. Mangelsdorf, Documentation and Assessment of Chemicals,
Fraunhofer Institute for Toxicology and Aerosol Research, Hanover,
Germany
Ms M.E. Meek, Environmental Health Directorate, Health Canada, Ottawa,
Ontario, Canada (Rapporteur)
Dr K. Paksy, Department of Reproductive Toxicology, National Institute
of Occupational Health, Budapest, Hungary
Mr V. Quarg, Ministry for the Environment, Nature Conservation &
Nuclear Safety, Bonn, Germany
Mr D. Renshaw, Department of Health, London, United Kingdom
Dr J. Sekizawa, Division of Chemo-Bio Informatics, National Institute
of Health Sciences, Tokyo, Japan
Prof. S. Soliman, Department of Pesticide Chemistry, Alexandria
University, Alexandria, Egypt (Vice-Chairperson)
Dr M. Wallen, National Chemicals Inspectorate (KEMI), Solna, Sweden
Ms D. Willcocks, Chemical Assessment Division, Worksafe Australia,
Camperdown, Australia
Dr M. Williams-Johnson, Division of Toxicology, Agency for Toxic
Substances and Disease Registry, Atlanta, GA, USA
Dr K. Ziegler-Skylakakis, Senatskommission der Deutschen
Forschungsgemeinschaft zuer Pruefung gesundheitsschaedlicher
Arbeitsstoffe, GSF-Institut fuer Toxikologie, Neuherberg,
Oberschleissheim, Germany
Observers
Mrs B. Dinham,1 The Pesticide Trust, London, United Kingdom
Dr R. Ebert, KSU Ps-Toxicology, Huels AG, Marl, Germany (representing
ECETOC, the European Centre for Ecotoxicology and Toxicology of
Chemicals)
Mr R. Green,1 International Federation of Chemical, Energy, Mine and
General Workers' Unions, Brussels, Belgium
Dr B. Hansen,1 European Chemicals Bureau, European Commission, Ispra,
Italy
Dr J. Heuer, Federal Institute for Health Protection of Consumers &
Veterinary Medicine, Berlin, Germany
Mr T. Jacob,1 DuPont, Washington, DC, USA
Ms L. Onyon, Environment Directorate, Organisation for Economic Co-
operation and Development, Paris, France
Dr H.J. Weideli, Ciba Speciality Chemicals Inc., Basel, Switzerland
(representing CEFIC, the European Chemical Industry Council)
Secretariat
Dr M. Baril, International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland
Dr R.G. Liteplo, Health Canada, Ottawa, Ontario, Canada
Ms L. Regis, International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland
Mr A. Strawson, Health and Safety Executive, London, United Kingdom
Dr P. Toft, Associate Director, International Programme on Chemical
Safety, World Health Organization, Geneva, Switzerland
1 Invited but unable to attend.
RÉSUMÉ D'ORIENTATION
Ce CICAD relatif au 2-butoxyéthanol a été rédigé sur la base
d'évaluations préparées par le National Institute for Occupational
Safety and Health (NIOSH, 1990) et l'Agency for Toxic Substances and
Disease Registry (ATSDR, 1996). Une étude de la littérature publiée
jusqu'en mai 1997 a fourni des données complémentaires, à quoi se sont
ajoutés les éléments d'information obtenus lors de l'évaluation par
des pairs du présent CICAD. Des informations concernant la nature de
l'évaluation par les pairs et la disponibilité des documents originaux
figurent à l'appendice 1. Des informations sur cette évaluation sont
données à l'appendice 2. Ce CICAD a été approuvé en tant
qu'évaluation internationale lors d'une réunion du Comité d'évaluation
finale qui s'est tenue à Berlin (Allemagne) du 26 au 28 novembre 1997.
La liste des participants à la réunion du Comité d'évaluation finale
figure à l'appendice 3. La fiche d'information sur la sécurité
chimique (ICSC 0059), préparée par le Programme international sur la
sécurité chimique (IPCS, 1993), est également reproduite dans le
présent document.
Le 2-butoxyéthanol (CAS N° 111-76-2) est un éther du glycol
produit en quantités industrielles. C'est un liquide incolore
miscible à l'eau et soluble dans la plupart des solvants organiques.
Il est très largement utilisé dans les enduits, les laques à séchage
rapide, les émaux, les vernis, les dissolvants et les peintures au
latex. On l'utilise également dans les nettoyants pour métaux et
certains produits ménagers. Dans l'atmosphère, il est présent
entièrement à l'état gazeux et comme sa demi-vie à ce niveau est
d'environ 17 heures, il n'y a guère de risque de transport
atmosphérique. Dans l'eau, on estime sa demi-vie à environ 1-4
semaines. Il est probablement décomposé sans difficulté en aérobiose
dans le sol et dans l'eau. Son potentiel de bioaccumulation est
faible. Selon les données limitées dont on dispose, les
concentrations auxquelles on peut être exposé dans l'air sont de
l'ordre du µg/m3. S'il y a exposition indirecte de la population,
c'est très probablement par inhalation ou absorption percutanée lors
de l'utilisation de produits qui contiennent du 2-butoxyéthanol. La
concentration du 2-butoxyéthanol sur le lieu de travail est de l'ordre
du mg/m3.
Après exposition par la voie respiratoire, buccale ou percutanée,
le 2-butoxyéthanol est facilement résorbé. La métabolisation s'opère
essentiellement sous l'action de l'alcool- et de l'aldéhyde-
déshydrogénase et conduit à la formation de 2-butoxyacétaldéhyde et
d'acide 2-butoxyacétique, le principal métabolite. Il existe
toutefois d'autres voies métaboliques.
Le 2-butoxyéthanol présente une toxicité aiguë modérée et peut
irriter la peau et les yeux; il n'entraîne aucune sensibilisation
cutanée. Les principaux effets du 2-butoxyéthanol et de son principal
métabolite, l'acide 2-butoxyacétique, sont dus à l'hématotoxicité de
ces composés. Le rat est l'espèce la plus sensible. Les résultats
des études in vitro montrent que les hématies humaines ne sont pas
aussi sensibles que celles du rat aux effets hémolytique du
2-butoxyéthanol et de l'acide 2-butoxyacétique et que l'effet
hémolytique de ce dernier est plus prononcé. Chez le rat, l'action
toxique se manifeste aussi au niveau du système nerveux central, des
reins et du foie, mais à une concentration plus élevée que dans le cas
des effets hémolytiques. On n'a pas observé chez l'animal d'effets
toxiques sur la reproduction à des doses inférieures aux doses
toxiques. Les épreuves de mutagénicité in vitro ont donné des
résultats irréguliers mais en l'absence d'indices structuraux et
compte tenu des résultats négatifs obtenus in vivo, on peut avec une
confiance suffisante, considérer que le 2-butoxyéthanol n'est pas
mutagène. Les données limitées que l'on a pu tirer d'un certain
nombre de cas d'intoxication de même que les résultats d'une étude en
laboratoire, montrent que des effets analogues -- notamment des effets
hémolytiques et des effets au niveau du système nerveux central -- se
produisent chez l'homme comme chez le rat, mais à des concentrations
beaucoup plus élevées. Compte tenu des effets hémolytiques observés
chez des rattes gravides exposées pendant la période de gestation, on
a estimé à 13,1 mg/m3 la concentration tolérable pour l'homme.
En s'en tenant à des hypothèses extrêmement prudentes, on peut
considérer que la concentration estimative maximale de 2-butoxyéthanol
dans les eaux de surface très proches des effluents est susceptible de
dépasser parfois la valeur prévisible de la concentration maximale
sans effet observable. Toutefois, selon des hypothèses plus réalistes
fondées sur les données disponibles, il semblerait que ce composé ne
soit que faiblement toxique pour les organismes aquatiques. Comme la
demi-vie atmosphérique du 2-butoxyéthanol est brève, on estime que la
concentration mesurée ou calculée de cette substance dans l'air ne
pose pas de problème écologique.
RESUMEN DE ORIENTACION
El presente documento abreviado de evaluación internacional de
productos químicos (CICAD) sobre el 2-butoxietanol se basó en los
exámenes preparados por el Instituto Nacional para la Seguridad y
Salud del Trabajo (NIOSH, 1990) y la Agencia para el Registro de
Sustancias Tóxicas y Enfermedades (ATSDR, 1996). Se identificaron
datos adicionales en una investigación de publicaciones actualizada
hasta mayo de 1997, así como en el curso del examen por homólogos del
presente CICAD. En el apéndice 1 se halla información sobre la
naturaleza del examen por homólogos y la disponibilidad de los
documentos de origen. En el apéndice 2 se presenta información sobre
el análisis por homólogos del presente CICAD. Este CICAD fue aprobado
como evaluación internacional en una reunión de la Junta de Examen
Final, celebrada en Berlín (Alemania) los días 26-28 de noviembre de
1997. En el apéndice 3 se halla la lista de los participantes en la
reunión de la Junta de Examen Final. También se ha reproducido en el
presente documento la ficha internacional de seguridad química (ICSC
0059) producida por el Programa Internacional de Seguridad de las
Sustancias Químicas (IPCS, 1993).
El 2-butoxietanol (CAS N° 111-76-2) es un éter glicólico de alto
volumen de producción. Es un líquido incoloro miscible en agua y
soluble en la mayor parte de los disolventes orgánicos. El
2-butoxietanol se utiliza ampliamente como disolvente en
revestimientos de superficies, y en lacas en nebulización, lacas de
secado rápido, esmaltes, barnices, eliminadores de barnices y pintura
látex. También se utiliza en productos limpiadores de metales y
domésticos. El 2-butoxietanol existe en la atmósfera casi totalmente
en forma de vapor; dado que el producto químico tiene una semivida
atmosférica de unas 17 horas, el riesgo de transporte por la atmósfera
debe ser pequeño. La semivida estimada del 2-butoxietanol en agua es
aproximadamente de 1-4 semanas; probablemente experimenta una
biodegradación rápida en el suelo aerobio y en el agua. La capacidad
de acumulación es baja. Basándose en datos limitados puede indicarse
que la exposición ambiental en el aire se halla en general en la gama
de µg/m3. La exposición indirecta de la población general al
2-butoxietanol se produce muy probablemente por inhalación y absorción
cutánea durante el empleo de productos que contienen la sustancia
química. Las concentraciones del 2-butoxietanol en el aire en
entornos laborales se hallan típicamente en la gama de mg/m3.
El 2-butoxietanol se absorbe fácilmente después de la exposición
por inhalación o por vías oral y cutánea. El producto químico es
metabolizado principalmente por la deshidrogenasa de alcoholes y
aldehídos, con formación de 2-butoxiacetaldehído y de ácido
2-butoxiacético, el principal metabolito, aunque también se han
identificado otras vías metabólicas.
El 2-butoxietanol presenta una moderada toxicidad aguda y es
irritante para los ojos y la piel; no es un sensibilizador cutáneo.
El principal efecto del 2-butoxietanol y de su metabolito, el ácido
2-butoxiacético, es la hematotoxicidad, siendo la rata la especie más
sensible. Los resultados de estudios in vitro muestran que los
eritrocitos humanos no son tan sensibles como los eritocitos de rata a
los efectos hemolíticos del 2-butoxietanol y del ácido
2-butoxiacético, y también que los eritrocitos son más sensibles a la
hemólisis por el ácido 2-butoxiacético que por el 2-butoxietanol. En
la rata, los efectos adversos sobre el sistema nervioso central, los
riñones y el hígado se producen con concentraciones de exposición más
altas que los efectos hemolíticos. En animales no se han observado
efectos adversos sobre la reproducción y el desarrollo con dosis
inferiores a las tóxicas. Aunque los resultados de las pruebas
in vitro de mutagenicidad del 2-butoxietanol son incoherentes, la
ausencia de elementos estructurales de alerta y los resultados
negativos de los estudios in vivo son suficientemente alentadores
para permitir llegar a la conclusión de que el 2-butoxietanol no es
mutagénico. Basándose en datos limitados procedentes de estudios de
casos y de un estudio de laboratorio, se han señalado efectos agudos
análogos (incluidos efectos hemolíticos y otros sobre el sistema
nervioso central) en personas y ratas expuestas al 2-butoxietanol,
aunque los efectos se observaron con concentraciones de exposición
mucho más altas en personas que en ratas. Basándose en la aparición
de efectos hemolíticos en ratas grávidas expuestas durante la
gestación, se ha deducido una concentración tolerable de muestra para
las personas de 13,1 mg de 2-butoxietanol/m3.
Sobre la base de supuestos extremadamente conservadores, las
concentraciones previstas máximas de 2-butoxietanol en aguas
superficiales situadas cerca de corrientes de efluentes pueden, en
algunos casos, exceder de las concentraciones previstas de efectos no
observados. Sin embargo, supuestos más realistas basados en los datos
disponibles permiten indicar que el riesgo para los seres acuáticos es
escaso. Debido a la corta semivida del 2-butoxietanol en la
atmósfera, las concentraciones medidas o previstas de este producto
químico en el aire se consideran exentas de importancia ambiental.
 1. EXECUTIVE SUMMARY
This CICAD on 2-butoxyethanol was based upon reviews prepared by
the National Institute for Occupational Safety and Health (NIOSH,
1990) and the Agency for Toxic Substances and Disease Registry (ATSDR,
1996). Additional data were identified through an updated literature
search to May 1997, as well as during the peer review of this CICAD.
Information on the nature of the peer review and availability of the
source documents is presented in Appendix 1. Information on the peer
review of this CICAD is presented in Appendix 2. This CICAD was
approved as an international assessment at a meeting of the Final
Review Board, held in Berlin, Germany, on 26-28 November 1997.
Participants at the Final Review Board meeting are listed in Appendix
3. The International Chemical Safety Card (ICSC 0059) produced by the
International Programme on Chemical Safety (IPCS, 1993) has also been
reproduced in this document.
2-Butoxyethanol (CAS no.
1. EXECUTIVE SUMMARY
This CICAD on 2-butoxyethanol was based upon reviews prepared by
the National Institute for Occupational Safety and Health (NIOSH,
1990) and the Agency for Toxic Substances and Disease Registry (ATSDR,
1996). Additional data were identified through an updated literature
search to May 1997, as well as during the peer review of this CICAD.
Information on the nature of the peer review and availability of the
source documents is presented in Appendix 1. Information on the peer
review of this CICAD is presented in Appendix 2. This CICAD was
approved as an international assessment at a meeting of the Final
Review Board, held in Berlin, Germany, on 26-28 November 1997.
Participants at the Final Review Board meeting are listed in Appendix
3. The International Chemical Safety Card (ICSC 0059) produced by the
International Programme on Chemical Safety (IPCS, 1993) has also been
reproduced in this document.
2-Butoxyethanol (CAS no.  11.2.2 Terrestrial environment
Data are inadequate to characterize the risks to terrestrial
organisms of exposure to 2-butoxyethanol. A PEClocal(air) of 537 µg/m3,
based upon the use patterns of this chemical in Australia, has been
reported (OECD, 1997). Although available monitoring data are
limited, this predicted concentration is much higher than levels
measured in ambient air (see section 6). As 2-butoxyethanol is
expected to have a half-life in the atmosphere of less than 1 day,
these concentrations are considered to have no environmental
significance.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Previous evaluations of 2-butoxyethanol published by WHO, the
International Agency for Research on Cancer (IARC), the Joint FAO/WHO
Expert Committee on Food Additives (JECFA), or the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) were not identified. A
Screening Information Dataset (SIDS) Initial Assessment Report has
been prepared under the Organisation for Economic Co-operation and
Development (OECD) High Production Volume (HPV) Chemicals Programme
(OECD, 1997). Information on international hazard classification and
labelling is included in the International Chemical Safety Card that
has been reproduced in this document.
13. HUMAN HEALTH PROTECTION AND EMERGENCY ACTION
Human health hazards, together with preventative and protective
measures and first aid recommendations, are presented in the
International Chemical Safety Card (ICSC 0059) reproduced in this
document.
13.1 Human health hazards
2-Butoxyethanol is toxic to humans. Following long-term or
repeated exposure, effects on the blood may be observed.
13.2 Advice to physicians
In case of intoxication, immediate supportive measures should be
given, as central nervous system depression, respiratory paralysis,
hypotension, and metabolic acidosis have been observed in the few
hours post-exposure. Close monitoring for renal toxicity and possible
haemodialysis are mandatory in the subsequent days (renal
insufficiency may develop 2-3 days post-exposure) until recovery is
achieved, on average by the second week post-exposure.
13.3 Health surveillance advice
Periodic medical examination of the haematopoietic system should
be included in a health surveillance programme.
13.4 Spillage
As 2-butoxyethanol is toxic and absorbed through the skin,
emergency crews need to wear proper equipment, including a mask with
cartridge for organic vapour, for handling spills. The chemical
should not be allowed to enter drains or watercourses.
14. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Information on national regulations, guidelines, and standards
can be found in the International Register of Potentially Toxic
Chemicals (IRPTC), available from UNEP Chemicals (IRPTC), Geneva.
The reader should be aware that regulatory decisions about
chemicals taken in a certain country can be fully understood only in
the framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
INTERNATIONAL CHEMICAL SAFETY CARD
2-BUTOXYETHANOL ICSC: 0059
26.04.193
CAS #
11.2.2 Terrestrial environment
Data are inadequate to characterize the risks to terrestrial
organisms of exposure to 2-butoxyethanol. A PEClocal(air) of 537 µg/m3,
based upon the use patterns of this chemical in Australia, has been
reported (OECD, 1997). Although available monitoring data are
limited, this predicted concentration is much higher than levels
measured in ambient air (see section 6). As 2-butoxyethanol is
expected to have a half-life in the atmosphere of less than 1 day,
these concentrations are considered to have no environmental
significance.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
Previous evaluations of 2-butoxyethanol published by WHO, the
International Agency for Research on Cancer (IARC), the Joint FAO/WHO
Expert Committee on Food Additives (JECFA), or the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) were not identified. A
Screening Information Dataset (SIDS) Initial Assessment Report has
been prepared under the Organisation for Economic Co-operation and
Development (OECD) High Production Volume (HPV) Chemicals Programme
(OECD, 1997). Information on international hazard classification and
labelling is included in the International Chemical Safety Card that
has been reproduced in this document.
13. HUMAN HEALTH PROTECTION AND EMERGENCY ACTION
Human health hazards, together with preventative and protective
measures and first aid recommendations, are presented in the
International Chemical Safety Card (ICSC 0059) reproduced in this
document.
13.1 Human health hazards
2-Butoxyethanol is toxic to humans. Following long-term or
repeated exposure, effects on the blood may be observed.
13.2 Advice to physicians
In case of intoxication, immediate supportive measures should be
given, as central nervous system depression, respiratory paralysis,
hypotension, and metabolic acidosis have been observed in the few
hours post-exposure. Close monitoring for renal toxicity and possible
haemodialysis are mandatory in the subsequent days (renal
insufficiency may develop 2-3 days post-exposure) until recovery is
achieved, on average by the second week post-exposure.
13.3 Health surveillance advice
Periodic medical examination of the haematopoietic system should
be included in a health surveillance programme.
13.4 Spillage
As 2-butoxyethanol is toxic and absorbed through the skin,
emergency crews need to wear proper equipment, including a mask with
cartridge for organic vapour, for handling spills. The chemical
should not be allowed to enter drains or watercourses.
14. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
Information on national regulations, guidelines, and standards
can be found in the International Register of Potentially Toxic
Chemicals (IRPTC), available from UNEP Chemicals (IRPTC), Geneva.
The reader should be aware that regulatory decisions about
chemicals taken in a certain country can be fully understood only in
the framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
INTERNATIONAL CHEMICAL SAFETY CARD
2-BUTOXYETHANOL ICSC: 0059
26.04.193
CAS # 